3
Low CD4 Count as an Indicator of Disability
The committee sought to identify a laboratory marker or set of markers that could be used to make decisions about functional impairment and disability. However, after carefully reviewing data from several large multinational HIV/AIDS cohorts and related publications, the committee found no direct associations between laboratory measures and the level of disability. In the absence of such evidence, the committee considered measures predictive of disease progression, morbidity, and mortality, which, in the expert assessment of the committee, could be taken as surrogate markers of disability.
Two laboratory markers, CD4 count and HIV-1 viral load, are a routine part of HIV clinical care and recommended by all primary care guidelines. They are inexpensive and widely available. The results are present in medical records and available in the disability determination process. Of the two, CD4 count is most appropriate for consideration in the determination of HIV disability. The CD4 count is a direct marker of the stage of HIV-induced immune deficiency and of the near-term risk of morbidity and mortality.
INDICATORS OF DISEASE PROGRESSION
AIDS was first recognized when clinicians diagnosed previously healthy individuals with rare cancers or opportunistic infections such as Kaposi’s sarcoma or Pneumocystis jirovecii pneumonia. In these early cases, it was noted that patients had severe depletion of the helper or CD4+ subtype of T-lymphocytes (CD4 cells), which in part explained their susceptibility to
opportunistic infections and cancers. Shortly thereafter a novel retrovirus, now called HIV, was discovered as the cause of AIDS. Laboratory measures were later determined to correspond with severity of disease and the rate of clinical progression. The two measures most commonly used to stage HIV infection are the absolute number of CD4 cells per cubic millimeter (mm3) of blood and the quantity of HIV RNA molecules per milliliter (mL) of plasma, also known as viral load.
The CD4 count indicates the degree of immune depletion or immunodeficiency. The remaining immunologic reserve, reflected in the CD4 count, is highly predictive of near-term risk of opportunistic diseases and mortality. The viral load, on the other hand, indicates the production rate of HIV virions and expected rate of subsequent CD4 cell destruction. Although CD4 count is the central means of staging the disease in HIV infection, the viral load is the most accurate means of following the success or failure of antiretroviral therapy. Researchers and clinicians have used both of these tests to determine prognosis. In the absence of combination antiretroviral therapy, Mellors et al. (1996) demonstrated that the combination of the viral load and CD4 count could be used together to determine prognosis and survival and to some extent as indicators guiding the optimal point to recommend the initiation of antiretroviral therapy (see Figure 3-1).
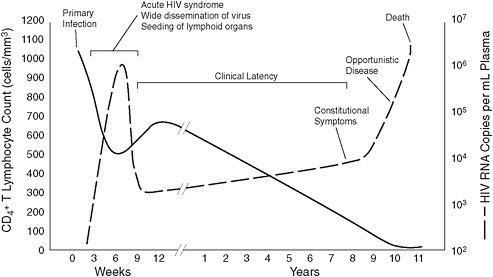
FIGURE 3-1 Time-based progression of untreated HIV infection demonstrated by CD4 count and viral load.
SOURCE: Image reprinted with permission from eMedicine.com, 2010, http://emedicine.medscape.com/article/211316-overview. Adapted from Fauci and Pan-taleo, 1996.
CD4 Count
The CD4+ T-cell count is the single best laboratory determinant of clinical outcomes (Egger et al., 2002). It is an important prognostic indicator of the remaining degree of immune function and is the single most important laboratory criterion for initiation of antiretroviral therapy in the patient with HIV infection. A typical healthy, HIV-negative person has a CD4 count greater than 500 cells/mm3 (Klimas et al., 2008). CD4 cells are directly and indirectly destroyed by HIV infection, and a lower number or percentage correlates with increasing risk of morbidity and mortality. In the early days of the HIV epidemic, CD4 less than 200 cells/mm3 was quickly established as the threshold below which serious or fatal opportunistic infections became common. The risk for certain opportunistic infections can be reduced by the prophylactic use of antibiotics. This use is recommended based on specific CD4 counts. For example, the risk of Pneumocystis jirovecii pneumonia rises steeply when the CD4 falls below 200 cells/mm3, and daily treatment with trimethoprim/sulfamethoxazole is recommended for these individuals (Meyers et al., 2008).
In 1993, the Centers for Disease Control and Prevention added a CD4 of less than 200 cells/mm3 as an AIDS-defining condition, which became a key issue before the development of effective antiretroviral therapy when the current HIV Infection Listings were published. As the CD4 count continues to fall, which it does inevitably in the absence of antiretroviral therapy, HIV-infected persons have an accelerated risk of a wide range of opportunistic diseases, all with serious morbidity, many rapidly fatal, unless immune deficiency is reversed with HIV suppression. In the time before current combination antiretroviral therapy, an HIV-infected adult with a CD4 ≤ 50 cells/mm3 had approximately a 45 percent 12-month mortality rate without therapy (Yarchoan et al., 1991).
Table 3-1 shows dramatic improvements in 12-month mortality rates before and after combination antiretroviral therapy for three multinational cohorts (U.S. Military HIV Natural History Study [Department of Defense, or DoD], EuroSIDA [SIDA is the Spanish acronym for AIDS], and the North American AIDS Cohort Collaboration on Research and Design [NA ACCORD]), supporting the notion that mortality rates have changed over time with the introduction of combination antiretroviral therapy. More importantly, these data show a substantial difference in mortality between CD4 ≤ 50 cells/mm3 and CD4 50–100 cells/mm3, indicating that a CD4 threshold (at or below 50 cells/mm3) independently predicts increased mortality.
The CD4 count usually rises with the initiation of potent combination antiretroviral therapy. The extent and rate of recovery may be limited in some individuals with a low CD4 cell nadir, which is associated with an
TABLE 3-1 12-Month Mortality Rates for Patients With HIV Infection as a Function of CD4 Strata, Before or After Combination Antiretroviral Therapy
increased incidence of the potentially disabling immune reconstitution inflammatory syndrome (IRIS). IRIS may develop in patients with HIV who also have coinfections or inflammatory diseases when beginning therapy (Hoffman et al., 2010). As the immune system responds to treatment and begins to restore to healthy levels, the first weeks on therapy typically include an increase in CD4 count and decrease in viral load (Hoffman et al., 2010). IRIS develops in 15 to 25 percent of patients when they start combination antiretroviral therapy (French et al., 2004; Jevtovi et al., 2005; Shelburne et al., 2006) and can be associated with substantial morbidity and even mortality. Research has shown CD4 of less than 100 cells/mm3 to be an independent risk factor for the development of IRIS (Manabe et al., 2007).
When the CD4 count is low, patients remain at an increased risk of morbidity and mortality from AIDS-related conditions. Close monitoring is needed during this vulnerable period of at least 12 months or until successful antiretroviral management is established. The probability of achieving HIV suppression is reduced in those with a CD4 ≤ 50 cells/mm3 at treatment initiation (Knobel et al., 2001) and at 48 weeks following the initiation of therapy. Patients who initiated therapy with CD4 ≤ 50 cells/mm3 have an average CD4 of 167 cells/mm3, compared to an average CD4 of 281 cells/mm3 for those who started therapy with CD4 in the 51–200 cells/ mm3 range (Robbins et al., 2009). Because of its prognostic significance, CD4 count has become the central criterion for deciding when to recommend beginning antiretroviral therapy, and, along with HIV viral load, is universally used to monitor the outcomes of this treatment.
CD4 count determination is recommended by all guidelines of HIV management and essentially all patients diagnosed with HIV infection have results of this test available in their medical records. CD4 count is an inexpensive ($50 or less in most laboratories)1 and reproducible test performed in numerous hospital and commercial laboratories. The significance of the CD4 count in staging and monitoring HIV disease is that it is a low-cost method of allowing clinicians to chart progression of disease and response to treatment. These measurements are easily obtained from medical records.
HIV-1 Viral Load
The number of copies of HIV-1 virions in the circulating blood (viral load) can be accurately measured by polymerase chain reaction or other nucleic acid detection methods. Most assays can accurately detect and reproducibly quantitate viral loads greater than 50 copies/mL. The viral load
in chronic infection is in the range of 8,000 to 60,000 copies/mL (Mellors et al., 2007). Each infected person has a relatively characteristic viral load or set point over the course of infection. Although the viral load has been used in the diagnosis of acute HIV infection before the appearance of serum HIV antibodies, its primary application is in monitoring response to antiretroviral therapy. Viral load is expected to decline rapidly once therapy is initiated and to drop to levels of less than 50 copies/mL after 12 to 24 weeks of treatment. The poorest responses to combination antiretroviral therapy are in those patients with CD4 less than 50 cells/mm3, viral load greater than 100,000 copies/mL, and age greater than 50 when treatment is initiated (Egger et al., 2002). Once a goal of suppression of viral load ≤ 50 copies/mL is achieved, the test is repeated (every 3 to 6 months is recommended in most guidelines) to detect subsequent virologic failure. This is often a consequence of poor adherence to the prescribed medication.
Viral load, along with the CD4 count, is considered a routine test and part of the standard of care in all published guidelines for patients with HIV/AIDS. Like the CD4 count, this test is performed regularly throughout a patient’s treatment regimen to determine treatment efficacy. The viral load is also relatively inexpensive (about $100 in most labs)2 and is widely available. Unlike the CD4 count, the viral load does not directly indicate disease stage. Although the loss of CD4 cells is somewhat faster in those with very high viral loads, even that relationship is weak. The viral load is of crucial importance in monitoring response to antiretroviral therapy, but is not needed as part of disability assessment.
CD4 COUNT AS AN INDICATOR OF DISABILITY
Unfortunately, reproducible, standardized, and widely accepted measures of HIV functional impairment are limited. However, a strong relationship exists between advanced immune impairment and clinical outcomes, including mortality, although exceptions can occur. Laboratory markers of HIV infection are important indicators of the severity of disease. In the absence of direct measures of disability, the committee reviewed a variety of measures, notably CD4 count and viral load, to evaluate their efficacy in predicting morbidity, mortality, and treatment response. The committee concluded that viral load is a strong indicator of success of antiretroviral therapy, while CD4 is a strong indicator of disease progression. Therefore, CD4 is more appropriate in assessing disability than viral load.
Based on this review, the CD4 count was found to meet many of the principles identified in Chapter 1 for a listing and is already fully incorporated in current medical practice. Of the predictive measures considered, CD4 count is part of a routine standard of care for patients with HIV/AIDS,
commonly used, and an objective measure found easily in the medical record. All patients being medically managed for HIV infection should have serial CD4 count determinations in their medical records.
Findings from the literature and the data cohorts led the committee to find that a threshold can be drawn at CD4 ≤ 50 cells/mm3 that is directly associated with morbidity and mortality. The committee concludes that CD4 ≤ 50 cells/mm3 is a significant prognostic marker of poor outcome and, in the absence of direct data on functioning, of disability. CD4 ≤ 50 cells/mm3 is associated with functional impairments severe enough to warrant it as a listings-level impairment. Such a low level of CD4 cells indicates a very advanced stage of HIV infection and increased morbidity and mortality over time. It is important to note that this does not require a claimant to have symptomatic HIV infection.
A claimant with a CD4 ≤ 50 cells/mm3 ideally would receive treatment as part of his disability benefits, potentially improving his immunologic and functional status. After a period of time, if the claimant’s CD4 count is greater than 50 cells/mm3, the claim should be reevaluated to determine whether the person continues to be disabled according to other components of the HIV Infection Listings, including sublistings estimating functional status. Since antiretroviral treatment often allows clinical improvement over a period of 1 or 2 years, the committee believes claimants allowed under such a listing should be reevaluated periodically for disability status. The committee believes 3 years would allow for a sustained response and is the maximum practical period for Social Security Administration (SSA) reassessment. If the claimant’s CD4 count exceeds the minimum threshold and the claimant is not disabled according to other sublistings, he should no longer receive disability benefits. However, in the event that the CD4 count drops below 50 cells/mm3, his disability benefits should be reinstated.
RECOMMENDATION 1. SSA should use CD4 count as an indicator of disability. Specifically, CD4 ≤ 50 cells/mm3is an indicator that a claimant’s HIV infection is disabling. This allowance should be reevaluated periodically by SSA.
REFERENCES
Agan, B., B. Hale, N. Crum-Cianflone, T. Whitman, A. Ganesan, G. Wortmann, A. Weintrob, V. Barthel, V. Marconi, M. Landrum, T. Ferguson, L. Eberly, A. Lifson, K. H. Hullsiek, G. Grandits, and N. Michael for the U.S. Military (Department of Defense) HIV Natural History Study. 2010 (unpublished). Mortality by CD4 count or clinical diagnosis.
Egger, M., M. May, G. Chêne, A. N. Phillips, B. Ledergerber, F. Dabis, D. Costagliola, A. D’Arminio Monforte, F. de Wolf, P. Reiss, J. D. Lundgren, A. C. Justice, S. Staszewski, C. Leport, R. S. Hogg, C. A. Sabin, M. J. Gill, B. Salzberger, and J. A. C. Sterne. 2002. Prognosis of HIV-1-infected patients starting highly active antiretroviral therapy: A collaborative analysis of prospective studies. The Lancet 360(9327):119–129.
Fauci, A. S., and G. Pantaleo. 1996. Immunopathogenic mechanisms of HIV infection. Annals of Internal Medicine 124(7):654–663.
French, M. A., P. Price, and S. F. Stone. 2004. Immune restoration disease after antiretroviral therapy. AIDS 18(12):1615–1627.
Hoffman, J., J. van Griensven, R. Colebunders, and M. McKellar. 2010. Role of the CD4 count in HIV management. HIV Therapy 4(1):27–39.
Jevtovi, D. J., D. Salemovi, J. Ranin, I. Pei, S. Derjav, and O. Djurkovi-Djakovi. 2005. The prevalence and risk of immune restoration disease in HIV-infected patients treated with highly active antiretroviral therapy. HIV Medicine 6(2):140–143.
Justice, A., S. J. Gange, J. P. Tate, L. P. Jacobson, K. Gebo, K. Althoff, M. Kitahata, M. Saag, M. Horberg, and R. Moore for the NA-ACCORD and VACS Project Teams. 2010 (unpublished). Report to the Institute of Medicine committee evaluating disability criteria for those with HIV infection.
Klimas, N., A. O’Brien Koneru, and M. A. Fletcher. 2008. Overview of HIV. Psychosomatic Medicine 70(5):523–530.
Knobel, H., A. Guelar, A. Carmona, M. Espona, A. González, J. L. López-Colomés, P. Saballs, J. L. Gimeno, and A. Díez. 2001. Virologic outcome and predictors of virologic failure of highly active antiretroviral therapy containing protease inhibitors. AIDS Patient Care and STDs 15(4):193–199.
Manabe, Y., J. Campbell, E. Sydnor, and R. Moore. 2007. Immune reconstitution inflammatory syndrome: Risk factors and treatment implications. Journal of Acquired Immune Deficiency Syndromes 46(4):456–462.
Mellors, J. W., C. R. Rinaldo Jr., P. Gupta, R. M. White, J. A. Todd, and L. A. Kingsley. 1996. Prognosis in HIV-1 infection predicted by the quantity of virus in plasma. Science 272(5265):1167–1170.
Mellors, J. W., J. B. Margolick, J. P. Phair, C. R. Rinaldo, R. Detels, L. P. Jacobson, and A. Munoz. 2007. Prognostic value of HIV-1 RNA, CD4 cell count, and CD4 cell count slope for progression to AIDS and death in untreated HIV-1 infection. Journal of the American Medical Association 297(21):2349–2350.
Meyers, D., T. Wolff, K. Gregory, L. Marion, V. Moyer, H. Nelson, D. Petitti, and G. F. Sawaya. 2008. USPSTF recommendations for STI screening. (Prepared for the U.S. Preventive Services Task Force.) American Family Physician 77:819–824, http://www.ahrq.gov/clinic/uspstf08/methods/stinfections.htm (accessed June 18, 2010).
Mocroft, A., and the EuroSIDA Study Group. 2010 (unpublished). The incidence and probability of death in HIV-infected persons pre- and post-cART. University College London.
Robbins, G. K., J. G. Spritzler, E. S. Chan, D. M. Asmuth, R. T. Gandhi, B. A. Rodriguez, G. Skowron, P. R. Skolnik, R. W. Shafer, and R. B. Pollard. 2009. Incomplete reconstitution of T-cell subsets on combination antiretroviral therapy in the AIDS Clinical Trials Group Protocol 384. Clinical Infectious Diseases 48(3):350–361.
Shelburne, S. A., M. Montes, and R. J. Hamill. 2006. Immune reconstitution inflammatory syndrome: More answers, more questions. Journal of Antimicrobial Chemotherapy 57(2):167–170.
Yarchoan, R., D. J. Venzon, J. M. Pluda, J. Lietzau, K. M. Wyvill, A. A. Tsiatis, S. M. Steinberg, and S. Broder. 1991. CD4 count and the risk for death in patients infected with HIV receiving antiretroviral therapy. Annals of Internal Medicine 115(3):184–189.








