2
An Overview of Tick-Borne Diseases
A SYSTEMS APPROACH TO UNDERSTANDING TICK-BORNE DISEASES: PEOPLE, ANIMALS, AND ECOSYSTEMS
Richard S. Ostfeld, Ph.D., Cary Institute of Ecosystem Studies
Throughout the 20th and 21st centuries, the number of infectious diseases in humans has been increasing as approximately 335 human infectious diseases have emerged since 1940 (Jones et al., 2008; Figure 2-1). Approximately 60 percent of those diseases are zoonotic, of which 72 percent are transmitted from wildlife and the remainder are transmitted from domestic animals. Furthermore, approximately 30 percent of emerging infectious diseases are vector-borne, which include tick-borne diseases (TBDs). Currently, there is incomplete and inadequate knowledge about key factors pertaining to persistence of reservoir, transmission, and host responses. More research is needed to better understand these diseases and to improve strategies to protect human health.
Lyme disease, one of the tick-borne diseases in the United States, emerged in the later half of the 20th century. It was first described in United States in the mid-1970s, although cases were reported in Europe in the late 1800s and early 1900s. The annual incidence of reported cases of Lyme disease has grown significantly from its initial recognition through 2008. By 1982, the Centers for Disease Control and Prevention designated Lyme disease as a notifiable disease, but even with this designation, an unknown number of cases remain unreported. Lyme disease is also found in Europe, where it is one of the fastest growing zoonotic diseases.
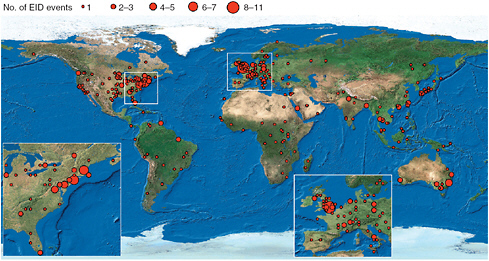
FIGURE 2-1 Global richness map of emerging infectious diseases from 1940 through 2004 showing clustering in the northeastern United States, western Europe, Japan, and southeastern Australia.
SOURCE: Reprinted by permission from Macmillian Publishers LTD: Nature, Jones et al., copyright 2008.
Reducing the burden of Lyme disease and other TBDs requires two main strategies: treatment of currently infected patients and prevention of transmission. Prevention is the ultimate goal to reduce the number of infections and clinical manifestations of TBD. Critical to any prevention measure is a fundamental understanding of the tick, its hosts, the pathogen, and the dynamic interplay of these components. Armed with that understanding, we can target the life stages, habitats, and other features of the organisms that confer a high risk of Lyme disease and other TBDs. Because effective vaccines are not currently available to humans, prevention strategies can be grouped into two approaches. The first focuses on human behavior such as the use of repellants and protective clothing, avoidance of risky activities and habitats, and so forth. The second is environmental and includes interventions that target ticks, their hosts, and the pathogens they transmit.
In most of northeastern United States, the black-legged tick, or Ixodes scapularis, is the primary vector for the transmission of Borrelia burgdorferi, the spirochete bacterium that causes Lyme disease. The Ixodes tick is a three-host tick, and its life cycle includes three post-egg stages: larva, nymph, and adult. At each stage, the tick takes a single blood meal from a vertebrate host. The tick then drops off the host and molts into the next stage: larvae into nymph, nymph into adult (see Figure 2-2). After a single No. of EID events 1 2–3 4–5 6–7 8–11
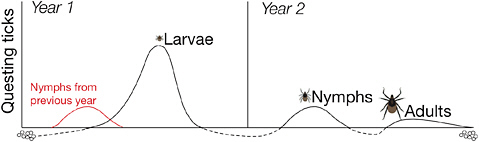
FIGURE 2-2 In its 2-year life cycle, the black-legged tick develops from egg to adult, taking a single blood meal at each stage in its development.
SOURCE: Unpublished, reprint courtesy of Dr. Jesse Brunner.
blood meal during which the males and females copulate, the adults also drop off, the females lay eggs, and both adults die to complete the life cycle.
Larval ticks hatch uninfected with Lyme disease spirochetes because of the lack of transovarial transmission of the pathogen from ticks through their eggs. However, the larval ticks will feed on virtually any warm-blooded vertebrate they encounter while questing on the forest floor. If they happen to feed on a host infected with Lyme disease spirochetes, the larvae may become infected. In that case, they will molt into an infected nymph capable of transmitting the infection to its next host, including humans.
Lyme disease and other TBDs are most likely to be transmitted to humans during the nymphal stage. The primary reasons are the frequent high prevalence of infection among nymphal ticks, the very small size of the nymphs, and the fact that nymphs reach their peak activity in late spring and early summer when human outdoor activity also peaks. The size of the nymphal population peak and the prevalence of infected nymphs are critical in determining the risk of human exposure to Lyme disease and other TBDs.
After Lyme disease was discovered in the 1970s, conventional wisdom held that only a few hosts determine how many infected nymphs would appear in a given year, with white-tailed deer being the predominant one. Even today, many research articles suggest that white-tailed deer are the definitive host of the black-legged tick. This theory is true for some environments.1 For example, in a recent study on Monhegan Island, Maine, Rand and colleagues (2004) hunted to reduce a deer herd from approximately
100 to zero in a few years. By the end of the study, larval and nymphal tick populations had also declined to near zero. However, this is likely because humans and their pets were the only host species available on the island for adult black-legged ticks after the removal of the deer.
Other studies that have controlled deer populations and monitored tick populations have found a different outcome when other host species for the black-legged tick were present. For example, Stafford et al. (2003) significantly reduced a deer herd at two sites in southern Connecticut. At one site, the nymphal tick population declined steadily, and the researchers found a significant correlation in the size of the deer and nymphal tick populations. However, they found no significant correlation between deer and nymphal tick populations at the other site. Similarly, Deblinger et al. (1993) reduced the deer population on the northern coast of Massachusetts by 40 percent per year. Initially, the nymphal tick population declined significantly. However, by the end of the study the nymphal tick population had recovered and returned to the population level at the beginning of the study. Other studies have found no correlation between deer density and nymphal tick density in New York (Ostfeld et al., 2006) or between deer density and Lyme disease incidence in New Jersey (Jordan et al., 2007).
There are three primary reasons why the association between deer populations and black-legged ticks is often weak or variable. First, the black-legged tick is a host generalist in all three of its host-seeking life stages. Larvae and nymphs are known to feed on 41 species of mammals, 57 species of birds, and 14 species of lizards, while adults are known to feed on at least 27 species of mammals and 1 species of lizard. Second, when the population of a host species drops, ticks can aggregate on the remaining hosts. In the study reported by Deblinger et al. (1993) the number of ticks per deer rose as the researchers reduced the deer population. The same phenomenon may occur with other (non-deer) hosts for adult ticks as well, but has yet to be studied. Third, there is no correlation between the abundance of larval ticks in one year and the abundance of nymphal ticks (Ostfeld et al., 2006). A disconnect is apparent between the factors affecting larval tick populations and those affecting nymph populations—and therefore the risk of Lyme disease. So even if deer abundance determines subsequent larval abundance, this might not be relevant to Lyme disease risk.
An important note is that all three life stages feed on a number of different hosts. Rather than making assumptions about which hosts are fed upon by black-legged ticks, scientists need to determine empirically the role that the different host species play in producing the nymph population. The size of the larval cohort does not predict the size of the nymphal cohort—the cohort that is responsible for transmission of the pathogen. The critical issues are how many of the larvae are able to find a host that will support successful feeding and how many hosts will infect the larval tick so that it
becomes an infected nymph. Understanding the interactions between the various host species and the larval tick is critical.
A particular host species might encounter ticks at a typical rate based on its body size, the way it uses space, or some other factor that scientists do not yet understand. However, some ticks that encounter a host will be unable to feed because they will be groomed off and killed in the process—host permissiveness. The combination of encounter rates and permissiveness determines the number of larvae on a host—known as body burden—during the larval period. Furthermore, each host species may provide a different quality or quantity of blood to feeding larval ticks, affecting their rate of molting success and over-the-winter survival. Different host species also have different reservoir competence levels: that is, different probabilities that they will infect feeding larvae with a tick-borne pathogen.
Ostfeld and colleagues (Keesing et al., 2009) captured six types of birds and mammals—representing a range of taxonomic groups and body sizes—in August, when larval black-legged ticks were feeding. The animals were held in the laboratory for about 4 days, until all naturally acquired ticks had dropped off; the researchers then placed 100 larval ticks on each host and followed their fate. There was significant variation in permissiveness among the host species. Approximately 50 percent of larval ticks that attempted a blood meal on white-footed mice succeeded, and dropped off in a replete state. Only 3.5 percent of larval ticks attempting a blood meal on an opossum succeeded, however, with the rest killed while trying to feed. Similarly, there was a significant variation in larval tick burden among species. When species were captured from the wild and the number of attached ticks determined, it was found that the average mouse hosts about 25 larval ticks, the average gray squirrel about 150 larval ticks, and the average opossum about 250 larval ticks. From these data, the encounter rate of larval ticks with hosts and the proportion of ticks that do not feed successfully as a result of low permissiveness can be estimated. The white-footed mouse grooms off and kills an average of 50 larval ticks per week, while gray squirrels groom off and kill approximately 843 larval ticks and opossums 5,686 larval ticks.
These species also vary in reservoir competence, with infected white-footed mice infecting approximately 90 percent of larval ticks that feed on them, and the other species, such as the white-tailed deer, raccoons, and opossums, infecting very few larval ticks (see Figure 2-3). Although white-footed mice, and secondarily eastern chipmunks, are ideal hosts for both feeding and infecting larval ticks with tick-borne pathogens, opossums, gray squirrels, and probably other hosts are not, which reduces the risk of human exposure to Lyme disease from these hosts. Thus, the composition of the host community for black-legged ticks in nature may determine risk for human Lyme disease.
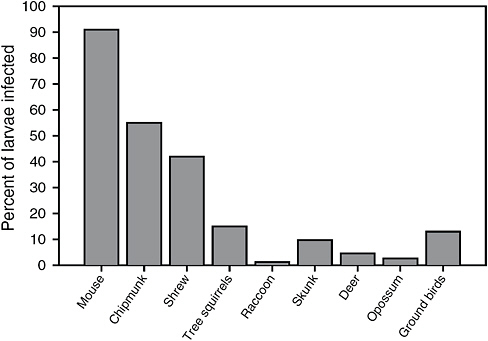
FIGURE 2-3 Reservoir competence of common Ixodes ticks in potential hosts in which high rates of larval infection reflect a greater risk of pathogen transmission during subsequent feeding cycles.
SOURCE: Updated from LoGiudice et al., 2003.
Vertebrate host populations are reduced when land is developed and habitats become fragmented. Primarily the large body animal species, which require large amounts of space to live, and the predatory species disappear. Small, omnivorous species, such as the white-footed mouse, tend to dominate small forest fragments. Based on the host competence data and host permissiveness studies, one likely outcome is an elevated risk of Lyme disease transmission in small forest fragments but not in larger forest fragments. In suburban Duchess County in southeastern New York, there was a significant negative correlation between the size of a forest fragment and the prevalence of Lyme disease infection in the nymphal tick population (Allan et al., 2003). In forest fragments smaller than about two or three hectares, the risk of human exposure to B. burgdorferi rose by a factor of three or four. Public health officials could use this information to develop avoidance and intervention mechanisms by identifying landscapes that are likely to be the riskiest for transmission to humans.
Scientists do not yet know whether these findings apply to other tick-borne diseases such as human babesiosis, which emerged in 1966, and
granulocytic anaplasmosis, which emerged in 1994. Hampering the research is the lack of a complete list of the natural reservoirs for the pathogens that cause those tick-borne diseases. However, initial work is beginning to test the role of various mammalian and avian hosts in infecting feeding larval ticks with the pathogens of these two emerging TBDs.
Knowledge Gaps and Research Opportunities
Ostfeld identified a number of key questions that remain for future study:
-
Which factors other than the size of forest fragments predict the abundance of ticks and the prevalence of Lyme disease? Some studies show that certain types of edges between forest and non-forest habitats influence the risk of Lyme disease transmission. Other studies show that the types of matrixes surrounding forest patches are important. The degree of isolation of these fragments can also influence the vertebrate host community. Understanding these factors and their impact will require much more research.
-
Do Lyme disease, anaplasmosis, and babesiosis share common risk factors? If different hosts play different roles in infecting ticks with the pathogens that cause these diseases, that would suggest that the environmental determinants of risk of Lyme disease versus other tick-borne diseases are decoupled.
-
Which animal hosts of Ixodes scapularis ticks are of critical importance in determining the tick population density?
-
Which local and landscape features affect human use of forests and other habitats, and hence their contact with ticks? Although the density of infected nymphs in small forest fragments might be high, this finding may not be tremendously important for Lyme disease if people prefer to use more extensive forests for recreation. The decoupling of entomological risk and human behavior will mean that different educational and environmental interventions are needed to reduce risk.
DISCUSSION
During the discussion, the participants and Ostfeld focused on the roles of various hosts, ticks, and habitats in the transmission of B. burgdorferi to humans and how this knowledge can be used to predict the occurrence of new areas for tick populations and to develop public health strategies aimed at reducing transmission of the disease.
The Role of Migratory Birds in Tick Distribution
Several studies (Klich et al., 1996; Smith et al., 1996) have shown the nymphal tick burdens on migratory birds in the northeastern and midwestern United States and in Canada, where researchers have hypothesized that migratory birds are responsible for moving significant numbers of ticks long distances into areas where populations might not otherwise occur or for increasing the number of ticks in an already populated area. Ostfeld noted that this is another emerging frontier for research. Some evidence suggests that the ticks that are moved around by migratory birds tend to have low infection prevalence because, with a few exceptions, migratory birds are not highly competent reservoirs. However, the research is not complete and the data is limited.
Reservoir–Competent Hosts
The need for ongoing research to identify why some host species have low or zero reservoir competence was suggested by some participants who cited the fact that lizards are not competent hosts. Ostfeld noted that the reason for low reservoir competence can vary by species. In the case of lizards, circulating proteins prevent infection. In other cases, such as opossums or gray squirrels, it is unknown whether low reservoir competence is a function of that complement system, which is innate or of induced immunity, including antibody production.
Environmental Factors Affecting Tick Populations
One participant questioned the relationship between the population size of larval ticks and the opportunities for feeding on hosts. Ostfeld noted that researchers have found that acorn abundance is a reliable predictor of infected nymphal ticks 2 years later. In studies in New York, increased acorn production both attracts white-tailed deer and boosts populations of white-footed mice, with a subsequent increase in the abundance of larval ticks (Ostfeld et al., 1996; Jones et al., 1998).
Transovarial Transmission
Studies in the late 1980s assessed questing larval ticks for prevalence of infection with spirochetes. The rate of infected larval ticks was approximately 1 percent, but the techniques used were not highly specific to B. burgdorferi (Piesman et al., 1986). One participant questioned whether the larvae’s role in transmitting disease to humans has been underrecognized.
Ostfeld noted that other spirochetes are known to have a more efficient transovarial transmission, and the 1 percent rate noted in these studies may be these other spirochetes. The evidence of the lack of transovarial transmission comes from experimental studies that were done with adult ticks feeding on hosts known to be infected with B. burgdorferi spirochetes. The larval ticks that hatched from the eggs of infected ticks were not infected.
Land-Use and Public Health Strategy
The relationship of tick and host habitat to the transmission of disease to humans was an area of considerable interest in which participants and Ostfeld discussed land-use strategies and the potential influence on public health. One participant asked whether there is an inherent geographic or landscape scale limit on the spread of Lyme disease. Ostfeld noted that it is possible to sample various areas to estimate tick abundance and to use Landsat imagery to examine the correlates. With these estimates, scientists can make models of how the ticks will spread. The problem is that the risk maps are created on a dynamic system, which results in underestimating the suite of potentially favorable conditions under which ticks can occur. As a result, ticks may inhabit areas that the risk maps do not indicate as favorable.
The research presented suggests an opportunity for engaging with regional planners in terms of forest fragmentation, observed one participant. Ostfeld concurred that engaging various county- and city-level government agencies is an area for future collaboration. The challenge lies in translating ecological observations into actual policy at the local, regional, and county levels, and more discussion is needed to determine how to integrate these observations into zoning and planning.
Other participants focused on specific strategies such as placing signage in high-risk areas or using pesticides. Ostfeld agreed that both of these strategies had promise. For example, the Tick Task Force in Duchess County has placed signs at trail heads and in public parks. The signs not only point out that the danger of exposure to pathogen-bearing ticks is high but also offer advice on how to reduce risk through personal protection measures. Targeted pesticide usage is an area of further discussion, Ostfeld noted. This strategy would benefit public health by targeting application in areas where both the incidence of infected ticks and human use are high—such as school fields—while reducing the collateral damage from overuse of pesticides.










