5
Surveillance, Spectrum, and Burden of Tick-Borne Disease, and At-Risk Populations
An understanding of the science of Lyme disease and other tick-borne diseases (TBDs) begins with the surveillance, spectrum, and burden of disease.
In addition to case definitions used for surveillance purposes, even terminology used to describe a condition can have an impact on the perceptions and recognized burden of a disease. As an example, what some refer to as “post-Lyme syndrome” others call “chronic Lyme disease.” The lack of consistency, understanding, and application of accepted definitions and terms are major obstacles to a better understanding of this disease and its long-term outcomes.
The availability, use, and interpretation of diagnostic tests also influence the validity and accuracy of surveillance findings. This can substantially impact the reported patterns and burden of disease. Diagnostic methods now used for most tick-borne illness are antibody based and have not improved much throughout the past two decades.
Ticks can often transmit more than one pathogen. For example, Ixodes ticks can simultaneously or sequentially infect their hosts with Borrelia burgdorferi, Anaplasma phagocytophilum, and Babesia microti. How often this occurs—and what it means for the presentation and severity of tick-borne diseases—is not well understood. As a result of changes in climate, the geographic distribution of tick vectors may also change the currently recognized demographic patterns, seasonality, and, ultimately, the incidence of tick-borne diseases.
In this chapter, five researchers explored the current state of knowledge of the incidence, patterns, and severity of key tick-borne diseases in the
United States and their impact on patients. These researchers also explain what efforts to track these diseases among people, and the movement of the pathogens in the environment, reveal about how infection moves from animals to people, especially among vulnerable populations.
LANDSCAPE OF LYME DISEASE: CURRENT KNOWLEDGE, GAPS, AND RESEARCH NEEDS
Gary P. Wormser, M.D., New York Medical College
Lyme disease is the most commonly reported vector-borne infection in the United States. B. burgdorferi is the only recognized pathogen to cause Lyme disease in the United States, and can be differentiated into 16 to 45 subtypes that may vary in infectivity and/or in pathogenicity (Wormser et al., 2008a; Crowder et al., 2010). In Europe, B. burgdorferi also causes Lyme disease, but other species of Borrelia, such as Borrelia afzelii and Borrelia garinii, also are responsible for Lyme disease. Because different species are responsible for infection in the two locations, the clinical syndromes associated with Lyme disease also differ between the United States and Europe.
The reported incidence rate of Lyme disease has increased steadily from 10,000 cases in 1992 to approximately 30,000 cases in 2009 (CDC website) (Figure 5-1). Twelve states in the Northeast, Mid-Atlantic, and North Central regions of the United States account for nearly 95 percent of these reported cases. New Hampshire (a 37-fold increase) and Maine (a 19-fold increase) have seen the largest proportionate increases in the number of cases during the past 10 years. New York has the largest absolute number of reported cases of Lyme disease, but is only fifth in the incidence of Lyme disease—the number of cases per 100,000 residents. Connecticut has the highest reported incidence.
In the United States, B. burgdorferi are transmitted exclusively by Ixodes ticks, which may transmit pathogens that cause other infections as well, including babesiosis, human granulocytic anaplasmosis, and flavivirus Powassan-like encephalitis. Of the diseases transmitted by I. scapularis, flavivirus Powassan-like encephalitis virus is the least well characterized. One recent study suggested that 2–5 percent of adult Ixodes scapularis ticks collected from two sites in Westchester County, New York, in 2008 contained Powassan virus (Tokarz et al., 2010). Approximately 4 percent of those ticks also contained B. miyamotoi, a relapsing fever-like Borrelia. Whether Borrelia miyamotoi causes human infection is unknown, and the clinical manifestations, if it does, are likewise unknown. Both Powassan virus and B. miyamotoi deserve of additional research efforts.
A number of animals act as reservoirs for Borrelia species, including mice, other small mammals, and some birds. Although deer serve as hosts
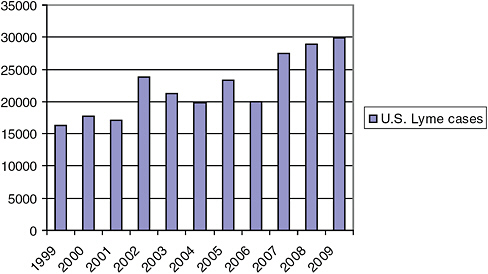
FIGURE 5-1 The reported incidence rate of Lyme disease has steadily increased in the United States since its emergence in the early 1980s.
SOURCE: Gary Wormser, unpublished.
for Ixodes ticks, they are not competent reservoirs for B. burdorferi. A number of prevention strategies have been demonstrated to decrease the incidence of Lyme disease. Reducing the number of ticks through the use of acaricides on land, mice, or deer is one approach for preventing Lyme disease. Modifying landscapes and building fences to keep deer away from inhabited areas can also reduce human exposure to ticks. Some investigators have reported that simply clearing leaf litter can reduce the number of ticks by approximately 90 percent (Schulze et al., 1995). Personal protective measures have been shown to reduce exposure to ticks. These measures include covering up as much as possible when outdoors, using insect repellents on exposed skin (Vasquez et al., 2008), bathing within 2 hours of tick exposure, and performing daily tick checks (Connally et al., 2009).
Absent from this arsenal of personal protective measures is a human Lyme disease vaccine. A first generation Lyme disease vaccine was introduced in 1998 by GlaxoSmithKline and was approximately 80 percent effective against Lyme disease. The reasons for the withdrawal of the vaccine from the market in 2002 are multifactorial and would be difficult to enumerate in this section. Subsequently, little work has been done to develop a new human vaccine. There is interest in developing a Lyme disease vaccine for mice, because they are the host reservoir for this infection. In laboratory experiments, Borrelia infection rates can be substantially reduced by feeding antibacterial compounds to mice (Dolan et al., 2008). Limiting the
pathogen burden in the host reservoir, either through vaccination or antibacterial treatment, would likely reduce the proportion of ticks that become infected and therefore are capable of transmitting Lyme disease to humans. However, the long-term feasibility of such an approach is unknown.
Lyme disease has several stages. Early localized Lyme disease manifests as a single skin lesion known as erythema migrans (EM). Early disseminated Lyme disease consists of multiple erythema migrans skin lesions in addition to possible cardiac and neurological manifestations, such as seventh cranial nerve palsy and meningitis. Late Lyme disease is most often associated with arthritis in large joints, less commonly with neurological and cardiac manifestations, and in Europe, acrodermatitis, a chronic skin condition.
From 1992 to 2006, 248,074 cases of patients with Lyme disease were reported to the U.S. Centers for Disease Control and Prevention (CDC). Of these patients, 69 percent had EM, 32 percent had arthritis, and 12 percent had neurological manifestations (Bacon et al., 2008). In contrast, during a vaccine trial that monitored 267 patients before and after they became ill, approximately 73 percent had erythema migrans, approximately 1.5 percent had arthritis, and 18 percent had developed a nonspecific viral-like syndrome (Steere et al., 1998). Other studies have suggested that 10 percent of patients believed to be infected with B. burgdorferi present with a viral-like syndrome, but the clinical manifestations and patterns of disease progression have not been well characterized (Aucott et al., 2009). Furthermore, approximately 7 percent of individuals in the vaccine trial study developed an asymptomatic infection based on documented seroconversion. At this time, the natural history for asymptomatic infection, and whether it is the same as that for untreated erythema migrans, is unknown. This area needs further study.
In 2008, the CDC/Council of State and Territorial Epidemiologists (CSTE) surveillance case definition for Lyme disease was made more encompassing to allow the documentation and tabulation of other manifestations reportedly associated with Lyme disease. The definition of a confirmed case remained the same: either erythema migrans, or late manifestations of the disease with laboratory evidence of infection. However, a definition for a probable case was added of physician-diagnosed Lyme disease with laboratory evidence of infection using a two-tier test. That is, any disease a physician designates as Lyme disease is a probable case if supported by laboratory test results. In 2009, clinicians reported 8,500 probable cases of infection, for a confirmed-to-probable ratio of 3.5 to 1.
CDC and CSTE also added two definitions for a suspected case: (1) EM with no known exposure to ticks and no laboratory evidence of infection, and (2) laboratory evidence of infection in the absence of clinical information. These changes were a constructive attempt to build the knowledge
base regarding the spectrum of illness among persons that the community considers to have Lyme disease.
Current serological testing for Lyme disease presents a number of challenges. First, serology testing using Western blots during early Lyme disease and in patients with erythema migrans is not sensitive. Second, the over-reading of weak bands on a Western blot by many commercial laboratories results in a high percentage of false-positive reports, although no studies have been done to document the percentage. Third, residents of some high-risk regions may demonstrate background seropositivity, leading them to test positive for Lyme disease on IgG Western blots even though they are completely well or have confirmed illness due to other etiologies. In one study, more than 50 percent of individuals who tested positive for Lyme disease on Western blot had no history of having had Lyme disease (Hilton et al., 1999). Finally, there are some patients who, after successful treatment and resolution of early Lyme disease symptoms, maintain persistent antibodies to B. burgdorferi.
Serologic findings lag the presence of erythema migrans. In a study of 252 patients in the United States with erythema migrans, serologic testing, including whole cell sonicate enzyme-linked immunosorbent assay (ELISA), two-tier testing, and the second generation serologic assay, C6 showed low sensitivity during the first 7 days of infection when erythema migrans is present. By 20–30 days after the onset of illness, the frequency of a positive C6 serologic test rises to approximately 100 percent of all patients (Wormser et al., 2008b). Although the dissociation of symptoms and laboratory results may appear insignificant, erythema migrans can resemble southern tick-associated rash illness (STARI), which appears after the bite of a lone-star tick (Amblyomma americanum). In fact, cases of STARI have been reported in Maryland and New Jersey, where B. burgdorferi infection is also common. The ability to differentiate STARI from B. burgdorferi infection clinically and in the laboratory would be helpful. Currently, the etiology, disease burden and patterns, and clinical manifestations (other than a rash) of STARI are unknown.
Borrelia burgdorferi spirochetes are thought to move from the site of a tick bite to other parts of the skin and organs through hematogenous dissemination. Blood cultures of approximately 40 percent of untreated patients in one study, tested up to 1 month after erythema migrans first appeared, yielded B. burgdorferi regardless of the size or duration of the rash (Wormser et al., 2005).
Approximately 73 percent of Lyme disease patients have symptoms in addition to a skin lesion when they first seek treatment, including arthralgias (joint pain), myalgias (muscle pain), fatigue, malaise, neck pain, and headache. Approximately 25 percent of patients continue to report milder symptoms even after treatment, although their skin lesions have long since
resolved. The median frequency of reported symptoms at 6 months is approximately 11.5 percent in eight U.S. randomized treatment trials (Cerar et al., 2010).
Three long-term outcome studies of patients with Lyme disease found that among healthy controls, who did not have Lyme disease, 15–43 percent reported fatigue, 16–20 percent reported headaches, up to 27 percent reported joint pain, and 19 percent reported muscle aches. These findings call into question whether the percentages of Lyme disease patients who continue to have symptoms more than 6 months after treatment exceed the percentages of the general population reporting the same symptoms. Furthermore, if the percentages among those reported to have Lyme disease are above the background rates, the causes of the long-term symptoms among Lyme disease patients remain unknown. A number of factors are associated with long-term symptoms, including how severely ill patients are when they first seek care (Nowakowski et al., 2003), the presence of neurologic manifestations (Eikeland et al., 2011), prior or current psychiatric conditions (Solomon et al., 1998), and greater sensitivity to symptoms (i.e., patients who are more aware of their symptoms will report them over a longer length of time). There are limited data suggesting that coinfection with untreated babesiosis and autoimmune events, such as the production of antineural antibodies, are correlated with long-term symptoms.
Some of the controversies surrounding Lyme disease reflect the fact that the disease means different things to different people. To some, Lyme disease is insidious and ubiquitous: Such patients commonly present with nonspecific symptoms, are diagnosed based on clinical judgment because diagnostic tests are insensitive, and require antibiotic treatment for months to years. To others, Lyme disease occurs focally, depends on exposure to infected ticks, and usually is linked to objective clinical manifestations. Positive laboratory tests are needed to support a diagnosis for symptoms other than EM, and the disease typically responds to antibiotic treatment.
Knowledge Gaps and Research Opportunities
There are a number of research opportunities to begin to answer questions regarding outstanding issues associated with Lyme disease:
-
Create a network of investigators and clinical trials for Lyme disease and other TBDs, and promote opportunities for collaborative research.
-
Create a repository for specimens of serum and cerebrospinal fluid from patients with tick-borne diseases.
-
Formalize definitions of tick-borne diseases and instruments for evaluating and following patients in different clinical groups.
-
Conduct broad-based studies of chronic Lyme disease, fibromyalgia, chronic fatigue syndrome, and other medically unexplained syndromes, free of any preconceived ideas on cause, perhaps led by the Institute of General Medical Sciences.
LYME DISEASE: APPROACHES TO UNDERSTANDING A MULTIDIMENSIONAL DISEASE
Benjamin J. Luft, M.D., State University of New York–Stony Brook
Lyme disease is an emerging and diverging disease. Increasingly, clinicians are starting to recognize that it is a disease that may affect many organ systems in subtle ways and have a biological, social, and societal impact for the patient. The acknowledgment that Lyme disease may be a complex and chronic illness requires a comprehensive, multidisciplinary and patient-centered perspective. Patients are not interested in whether their illness is caused by Borrelia burgdorferi or another genotype of Borrelia. They want to be well again.
Clinicians and researchers need to understand that the disease and its impact may intimately affect the severity and progression of symptoms. Because of the complexity of this disease, there is a need to develop better biological and clinical instruments to evaluate and measure the effectiveness of outcomes of treating its various manifestations. Furthermore, there is a need to develop a universally accepted phenotype of the disease. More than a quarter century after the discovery of Lyme disease, infectious disease specialists, neurologists, and psychiatrists still hold different conceptions of the disease. This may be due in part because they are focused on a particular organ system (i.e., their specialty), or they may be seeing the patient at a different phase of the illness. The natural history varies greatly from person to person, leading to an absence of consensus about what is “active” disease and what is disease impact.
The management of chronic illness, with its waxing and waning symptoms, poses a challenge to our traditional office-based, single-specialty approach to management. Furthermore, few centers are equipped to address the full gamut of medical, psychological, and social aspects of Lyme disease in a coordinated fashion. Because of the complexities and unknowns, third-party payers are not responsive to the needs of Lyme disease patients. As a result, reimbursement for disease management is denied because the symptoms are not accepted as the “disease,” and the patient is presented with a significant bill and marginalized from standard medical care.
The complexity of Lyme disease is unraveling. Currently, 37 species of Borrelia have been identified throughout the world, 12 of which are believed to cause Lyme disease. Some have been cultivated in the lab, while
others have been detected only by using polymerase chain reaction (PCR). In general, B. burgdorferi is the sensu stricto species that results in Lyme disease in the United States, while B. afzelii, B. garinii, and B. burgdorferi cause Lyme disease in Eurasia. The species diversity of Borrelia has important research implications, such as the appropriate number of the patients needed for studies, the geographic locations of study sites, and the appropriate power for meaningful statistical analysis.
The structure of OspA and OspC, the major outer surface proteins of Borrelia burgdorferi, and epitopes—the part of B. burgdorferi antigens recognized by the immune system—have been mapped. These data have allowed for further characterization of the pathogen at different stages along the life cycle of the tick and disease transmission. In an early study (Wang et al., 1999; Qiu et al., 2002), OspC variation was used to determine the variants of B. burgdorferi. Most ticks were host to between one and nine variants of B. burgdorferi. In a subsequent study (Seinost et al., 1999) that looked at patients and their skin isolates, ticks transmitted only a subset of B. burgdorferi variants, and even fewer of these variants actually made their way into the blood of patients. That means that only a relatively small number of B. burgdorferi strains actually cause invasive disease. The structure of OspC did not differ significantly among strains except that the electrostatic force at the head of the protein appears to be much stronger in invasive strains (Kumaran et al., 2001). The VMP protein in Borrelia hermsii strains that become invasive, and can cause relapsing fever, has a similar characteristic. These findings deserve further study.
At least 46 genotypes of B. burgdorferi have been identified and more than 34 percent of ticks carry at least 2 of the genotypes, while 5 percent have more than 3 (Qiu et al., 2002, 2008; Crowder et al., 2010). Some genotypes persist longer in mice and affect the severity of disease in humans. However, it is not known whether there is synergy or antagonism among a patient’s immune responses to different genotypes. Using a combination of broad-range PCR and mass spectrometry, Borrelia genotypes were identified in New York, Connecticut, Indiana, and California (Coulter et al., 2005). The genotypes varied among the four states. For example, 31 genotypes were found to occur in New York and 19 in Connecticut (see Figure 5-2). Although the two states are neighbors, they shared only 15 of the genotypes, with the remainder being found in only one of the states. Such variation suggests that people can contract different types of Lyme disease depending on where they live, and they may respond differently to antibiotic treatment.
Although there is no current research that indicates antibiotic resistance varies from strain to strain, multicentered therapeutic trials will be required to assess the efficacy of treatment over a broad array of patients exposed to various genotypes and their variants, as well as other concomitant
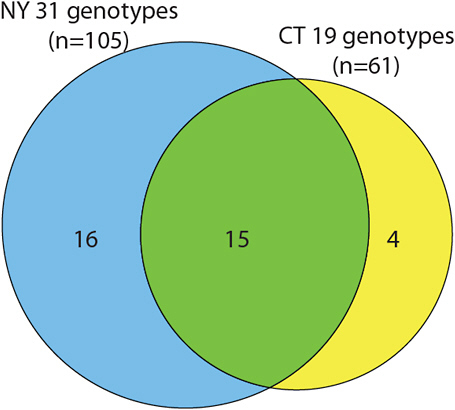
FIGURE 5-2 Borrelia burgdorferi genotype variation between regions. New York has 31 genotypes, but only 16 of them are unique to that state. Connecticut has 19 genotypes, with 4 unique to that state.
SOURCE: Coulter et al., 2005.
pathogens. These studies will need to be double blinded, placebo controlled, and amply powered to make a definitive assessment of efficacy. Furthermore, diagnostic tests for Lyme disease will need to consider genotypic variants of B. burgdorferi, as well as whether concomitant infection with other pathogens can lead to false-positive or false-negative results.
Recently, multilocus genetic sequencing was used to produce a family tree of Borrelia to determine whether B. burgdorferi could continually acquire new genetic information from other species and strains. After sequencing 17 B. burgdorferi genotypes, the organism’s core genome (i.e., the genes that occur in all strains of the organism) and its pangenome (i.e., the full complement of genes) could be characterized. From these results, it appears that the genome is open to a large gene repertoire, can adapt to evade host immunity and vaccination, and had the ability to develop antibiotic
resistance. In addition, there is the opportunity for lateral gene transfer between genotypes, which will determine the effectiveness of any treatment.
Understanding the genome of B. burgdorferi is a potential avenue to refine diagnostic tools. In one experiment, researchers identified B. burgdorferi genes that were positively selected (varied from strain to strain) as a result of immune pressure. From this information, an array that included more than 500 recombinant proteins was developed. Sera from patients tested using the two-tiered testing protocol found that approximately 54 percent of the patients sera yielded results that were diagnostic for Lyme disease (Coulter et al., 2005). When the protein microarray was used to test the patients’ sera, all patients tested positive for antibodies to Borrelia. These results suggest that seronegative Lyme disease, whether early or late, does not exist, and the findings simply reflect inadequate diagnostic tests.
Knowledge Gaps and Research Opportunities
A number of areas are important for future research, including
-
Developing accurate methods for identifying B. burgdorferi phenotypes;
-
Using appropriate animal models, such as the C3H persistent infection mouse model, to assess new approaches to diagnosis and treatment;
-
Developing better biological and clinical instruments to evaluate and measure the effectiveness of outcomes of treating its various manifestations;
-
Establishing standard operating procedures for developing criteria for acute and chronic Lyme disease; and
-
Gathering information and biological samples from patients at various stages of their disease, and using technology to evaluate those samples.
DISCUSSION SESSION
One clinician participant noted that chronic fatigue syndrome is a clinical condition of prolonged and severe fatigue of at least 6 months’ duration for which other causes have been excluded. He stated that in his clinical experience there is a relationship between chronic fatigue syndrome and Lyme disease in which some patients who meet the case definition for chronic fatigue syndrome may have seronegative Lyme disease. This was identified as a research gap.
Another clinician participant noted that in his practice there is a spectrum of acute and chronic Lyme disease, and, although work on acute disease is important, there needs to be a further emphasis on the research needs
of chronic illness. For example, the National Institutes of Health (NIH) trials showed 22 standardized measures of fatigue, pain, mental health, and physical health with patients with chronic illness that was not being adequately studied. The Zhang et al. study suggests that individuals with chronic manifestations of Lyme disease have costs of approximately $16,000 a year. He concluded by noting the need for a multidimensional program to help individuals with chronic manifestations. This discussion was expanded by other participants who suggested that research on this issue has not made substantial progress and a mechanism is needed to allow for innovative approaches to investigating the various stages of Lyme disease.
Another discussion focused on whether a protein assay could be produced to detect B. burgdorferi in all stages of disease for the general market. Luft commented that the technology could be developed to identify seroreactive proteins and detect disease in all stages. He also noted that, in addition to the development of such technology, a disease phenotype needs to be developed that would unify the research as technology moves forward.
THE INCREASING HEALTH BURDEN OF HUMAN BABESIOSIS: CLINICAL MANIFESTATIONS, COINFECTION, AND RESEARCH NEEDS
Peter J. Krause, M.D., Department of Epidemiology and School of Public Health, Yale School of Medicine
Babesiosis is an infection caused by intraerythrocytic protozoa of the genus Babesia. Transmission is primarily through the tick, Ixodes scapularis, but also may occur via blood transfusion, and rarely from mother to child perinatally. The health burden of the disease is significant and increasing in the United States (Vannier et al., 2008).
Babesiosis diagnosis is made on the basis of epidemiological, clinical, and laboratory information (Vannier et al., 2008). A person must live or have recently traveled to an endemic area or have recently received a blood transfusion. Babesiosis-compatible symptoms include fever, chills, sweats, headache, and fatigue. Most patients become ill approximately 1 to 2 weeks after a tick bite, although symptoms may appear up to 9 weeks after Babesia transmission through blood transfusions (Ruebush et al., 1981; Gubernot et al., 2009; Leiby, 2011). Symptoms usually last 1 to 2 weeks but can persist much longer in immunocompromised individuals (Krause et al., 2008). Laboratory confirmation of the diagnosis usually is made by microscopic identification of the organism on a thin blood smear supplemented with amplification of Babesia DNA using PCR and detection of antibody with immunofluorescence assay (IFA) or Western blot assays (Figure 5-3) (Krause et al., 2002; Vannier et al., 2008).
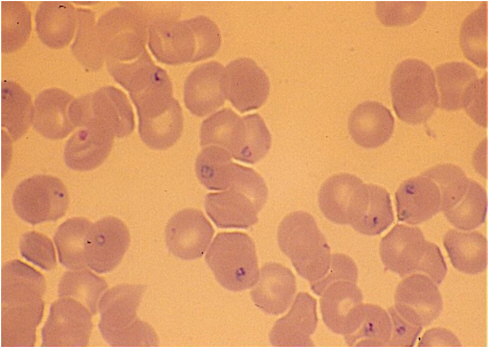
FIGURE 5-3 Ring forms of Babesia microti in human blood smear (× 1,000).
SOURCE: Courtesy of Peter Krause.
More than 100 Babesia species have been documented in a wide variety of wild and domestic animals; however, only a few of these species are known to infect humans (Vannier et al., 2008). Babesia microti is the most important of these because it causes endemic disease in the northeastern and northern Midwest United States (Vannier et al., 2008). Several cases of WA-1 (Babesia duncani) and CA-1 have been reported in Washington state and California (Persing et al., 1995; Conrad et al., 2006). Babesia divergens–like human illness has been reported in Kentucky, Missouri, and Washington state (Herwaldt et al., 1996; Beattie et al., 2002; Vannier et al., 2008). Both B. microti and B. duncani have been transmitted through transfusions of whole blood, packed red blood cells, and platelets (Ruebush et al., 1981; Gubernot et al., 2009).
In Europe, babesiosis follows a more sporadic pattern, with fewer cases being reported than in the United States, although a higher proportion of these cases are severe (Zintl et al., 2003; Vannier et al., 2008). Nearly all have occurred in people who are asplenic, raising the possibility that milder cases in immunocompetent people are not being identified. The primary agent is B. divergens, a cattle Babesia, but B. microti and Babesia venatorum (EU-1) also have been described (Herwaldt et al., 2003; Zintl
et al., 2003; Hildebrandt et al., 2007). In Asia, B. microti–like organisms have been reported to cause human infection in Japan, Korea (KO-1), and Taiwan (TW-1) (Shih et al., 1997; Wei et al., 2001; Kim et al., 2007).
The National Academy of Sciences cited babesiosis, ehrlichiosis, and Lyme disease as emerging threats to human health in the United States in a 1992 report (Lederberg et al., 1992). From 1991 to 2009, Connecticut experienced a marked increase in the number of cases of babesiosis (Connecticut State Department of Health) (Figure 5-4). Although similar trends have been reported in Massachusetts, New Jersey, New York, and Rhode Island, the actual numbers of infections are underestimated (Krause et al., 2003; Vannier et al., 2008). This increase is likely due to the expansion of the Babesia endemic range, increased recognition of babesiosis by clinicians and the public, and more widely available diagnostic testing. In a 10-year prospective study (1991–2000) of residents of Block Island, Rhode Island, case finding and serosurvey were conducted to determine the number of people with symptomatic and asymptomatic B. microti and Borrelia burgdorferi infection. About 50 B. microti and 75 B. burgdorferi infections per year were diagnosed for a ratio of 1 to 1.5 (Krause et al., 2003), reflecting the proportion of local ticks infected by these pathogens. Similar rates of hospital admissions for babesiosis were recorded for Block Island and southeastern Connecticut. These results suggest that in some areas endemic for both pathogens, the incidence of Lyme disease and that of babesiosis
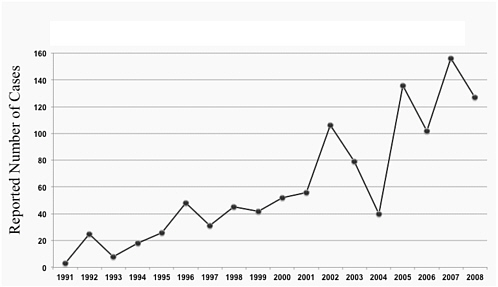
FIGURE 5-4 The reported incidence of babesiosis in Connecticut has increased from 1991 until 2008.
SOURCE: Peter Krause, unpublished.
are comparable. In recognition of the growing importance of babesiosis, the CDC and CSTE designated babesiosis as a national notifiable disease beginning in January 2011.
The health burden of disease includes both the number and severity of cases. Babesiosis is characterized by a wide range of disease severity, from asymptomatic to fatal. Approximately 25 percent of adults and 50 percent of children are asymptomatic when infected with B. microti (Krause et al., 2003). Typical clinical symptoms include chills, sweats, headache, fatigue, myalgia, arthralgia, anorexia, nausea, and cough (Krause et al., 2002, 2008; Vannier et al., 2008). Fulminant illness and death occur in 3–5 percent of otherwise healthy people and up to 25 percent of those who are immunocompromised or suffering from certain premorbid conditions (see discussion later in the chapter) (Hatcher et al., 2001; Vannier et al., 2008; Gubernot et al., 2009; Leiby et al., 2011). Healthy individuals with mild to moderate babesiosis can clear the infection without treatment, although antibiotic therapy can shorten the duration of illness (Krause et al., 1998).
Babesiosis is currently the most commonly reported transfusion-associated pathogen in the United States (FDA, 2008). More than 70 cases have been reported since the first report in 1979, with associated mortality of 10–28 percent (Gubernot et al., 2009; Leiby et al., 2011). Severity of transfusion-transmitted disease is presumably greater than tick-transmitted disease because blood recipients are more likely to be immunocompromised than those who acquire infection through tick transmission. Estimates of risk of transfusion transmission vary widely depending on geographic location, from approximately 1 case per million units of blood administered to as high as 1 in 604 units (Gerber et al., 1994; Tonnetti et al., 2009).
Human coinfection with B. microti and B. burgdorferi can occur because both pathogens share a common tick vector and mouse reservoir. Several studies suggest B. microti may increase B. burgdorferi disease severity, but B. burgdorferi appears to have little effect on the severity of B. microti infection. In a prospective study of 240 patients with Lyme disease, 26 (11 percent) were coinfected with B. microti (Krause et al., 1996). Patients experiencing both Lyme disease and babesiosis had significantly more symptoms than those with Lyme disease alone, and the duration of illness was longer. Borrelia burgdorferi DNA was detected in the blood of a higher percentage of coinfected individuals than in the blood of people infected with B. burgdorferi alone, suggesting that coinfected patients may be more likely to experience disseminated complications of Lyme disease, such as arthritis and neurologic problems, than those with Lyme disease alone. This study only assessed acute disease and did not include long-term follow-up of these patients, however. Although similar results are noted with other coinfections, including Lyme disease and human granulocytic anaplasmosis (HGA) (Belongia et al., 1999; Krause et al., 2002; Steere et al., 2003), further studies are needed regarding long-term complications of coinfection.
In conclusion, the health burden of babesiosis may be approaching that of Lyme disease. The incidence and geographic dispersion of Lyme disease are greater than those of babesiosis, but the differences are smaller than generally recognized and appear to be decreasing. Babesiosis is the most commonly reported transfused pathogen in the United States. At certain endemic sites, the incidence of Lyme disease and babesiosis are similar. Babesiosis causes more life-threatening disease than Lyme disease does, and coinfection may increase the severity of Lyme disease.
Knowledge Gaps and Research Opportunities
A number of questions offer research opportunities pertaining to babesiosis:
Epidemiology
-
Where is babesiosis enzootic and endemic now and in the future?
-
What is the incidence of babesiosis at the state, national, and international levels?
-
How fast is the incidence of babesiosis increasing?
-
What is the frequency of transfusion-transmitted babesiosis?
-
How often and where do pathogens other than B. microti cause babesiosis?
Pathogenesis
-
What is the pathogenesis of babesial illness?
-
Are there B. microti substrains with varying pathogenic potential?
-
What are the primary immune factors responsible for clearing babesiosis?
-
What are the mechanisms that worsen the severity of babesiosis with aging?
Clinical Manifestations
-
What are the long-term complications of babesiosis?
-
How does coinfection with babesiosis and other co-transmitted pathogens influence transmission in the reservoir host and disease in humans?
Diagnosis
-
Can scientists develop better biomarkers for babesiosis, including antibody, nucleic acid amplification test technologies, and culture?
Treatment/Prevention
-
How can transfusion-transmitted babesiosis be prevented?
-
What new antiparasitic therapies are available for babesiosis?
-
Is the use of partial-exchange transfusion to treat severe cases of babesiosis equivalent to full-exchange transfusion?
EHRLICHIA AND ANAPLASMA: SURVEILLANCE, COINFECTION, AND RESEARCH NEEDS
J. Stephen Dumler, M.D., Johns Hopkins School of Medicine
Ehrlichia and Anaplasma are pathogens within the Anaplasmataceae family; they are obligate intracellular bacteria similar to Rickettsia. At least three species of Ehrlichia and Anaplasma are known to cause human disease. Ehrlichia chaffeensis causes human monocytic ehrlichiosis (HME) by infecting monocytes in peripheral blood and macrophages in tissues. Anaplasma phagocytophilum causes HGA by infecting neutrophils. Ehrlichia ewingii, the cause of ewingii ehrlichiosis, resembles Ehrlichia chaffeensis genetically and serologically, but resembles Anaplasma phenotypically because it also lives inside neutrophils. HGA occurs predominantly in northern states where the black-legged tick, Ixodes scapularis, serves as the vector for both Anaplasma phagocytophilum and Borrelia burgdorferi. HME occurs predominantly across the South Central and Southeast United States, mirroring the range of the lone-star tick, Amblyomma americanum, the vector for the pathogen that causes the disease.
All these pathogens cause an undifferentiated febrile illness with typical laboratory findings. The incidence of ehrlichiosis is less than that of Lyme disease; however, the disease still carries a substantial burden. Cases of all forms of ehrlichiosis have been increasing since HME was first identified in the mid-1980s (Figure 5-5). Since 2000, there has been a significant spike in the number of reported ehrlichiosis cases, although, as with other tick-borne diseases, they are underreported.
HME and HGA often present with severe headache, myalgias, and a variety of other constitutional symptoms, such as nausea and vomiting. Rashes occur relatively infrequently, differentiating these illnesses from Rocky Mountain spotted fever and Lyme disease. Many patients-especially those with HME-also have some degree of central nervous system involvement. Approximately 20 percent of patients with HME have meningoencephalitis. Some immunocompromised patients, such as those with HIV, develop overwhelming infection.
Common laboratory findings include leukopenia (low white blood cell count) and/or thrombocytopenia (low platelet count). Furthermore, there is
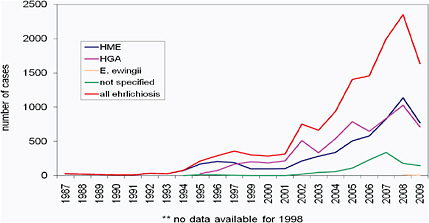
FIGURE 5-5 The incidence of reported ehrlichiosis and anaplasmosis in the United States has increased since 1986 when HME was first identified.
SOURCE: Steve Dumler, unpublished.
often evidence of mild hepatic (liver) injury, based on elevated serum levels of the hepatic enzymes ALT and AST.
Nearly half (48 percent) of the patients reported with HME from 2000 to 2007 were admitted to a hospital, while approximately 36 percent of patients with HGA were hospitalized during that period (CDC, unpublished data). The case fatality rate was approximately 2 percent for HME, and 0.5 percent for HGA. Life-threatening complications reported among these patients include acute respiratory distress syndrome, disseminated intravascular coagulation, meningitis, meningoencephalitis, and renal failure.
One way to analyze how often people are infected is to look at cross-sectional seroprevalence studies. The seroprevalence for HME in Tennessee ranges from 3.3 percent to 12.5 percent, depending on the diagnostic test used to detect the pathogen, while it is substantially lower in Arkansas at 1.3 percent. For HGA, the seroprevalence ranges from 0.6 to 0.9 percent in Connecticut to 14.9 percent in northwest Wisconsin. These results suggest a significant number of individuals are becoming infected in these locations. In contrast, the number of infections based on disease surveillance data is significantly lower. For HME, they range from 300 cases per 100,000 persons in Tennessee to 400 cases per 100,000 persons in Missouri (Standaert et al., 1995; Olano et al., 2003). For HGA, they range from 50 cases per 100,000 persons in Connecticut to 60 cases per 100,000 persons in northwest Wisconsin (Bakken et al., 1998). Based on these figures, the ratio of
symptomatic disease to infection ratio is 3–30:100 for HME and .4–6:100 for HGA. Even if the seroprevalence data reflect old and new infections, the difference between the two findings suggests either that many infected individuals are asymptomatic or subclinical, or that the diagnostic tests used to classify them are problematic.
Globally, Anaplasma seroprevalence is high: 8.9 percent in Asia, 7.2 percent in Europe, and 4.5 percent in North America, although these studies were conducted in endemic or high-risk regions (Dumler, 2005). The seroprevalence among individuals with confirmed Lyme disease who are also seropositive for Anaplasma is much higher than in cross-sectional studies. That raises two questions: Are these patients being appropriately diagnosed, and/or are there problems with serologic testing?
There are a number of common pathologic features among patients with HME and HGA. There is activation of the mononuclear phagocyte system, with macrophage infiltrates in tissues and hemophagocytosis; individuals with HME often have granulomas in the bone marrow. Pancytopenia—reflecting low leukocyte counts and low platelet counts—is likely due to consumption, sequestration, or destruction of these elements in the periphery, because bone marrow production tends to be normal in these patients. Pancytopenia can predispose patients to hemorrhage or opportunistic infections, both of which occur in HME and HGA. Some HME patients have significant hepatitis and with histopathologic appearance of apoptotic hepatocytes, a feature observed with cytokine-mediated diseases. Less frequent complications, such as acute respiratory distress syndrome, stem from interstitial pneumonitis and diffuse alveolar damage—probably from systemic proinflammatory response. Such inflammatory responses also appear with meningoencephalitis in patients with severe HME and among those who experience a toxic-shock–like syndrome with either disease.
It is not clear to what degree disease severity can be attributed to the host or the pathogen. HGA patients have a mean white blood cell count of 3.7 × 103 per microliter, yet the average proportion of infected leukocytes in less than 1 percent. In contrast, the mean leukocyte count among healthy adults is 7.8 × 103per microliter (Dumler, 2005). Thus, HGA patients lose many more white blood cells than can be accounted for by infection alone. On the other hand, the severity of HGA and HME is linked to the amount of time that elapses between the onset of illness and treatment, and the infections respond rapidly to antimicrobial therapy, suggesting that the microbe is responsible for at least some components of the disease.
Based on animal models, a consensus is beginning to emerge that these diseases are immunopathologic. In a mouse model using Ehrlichia muris or Ixodes ovatus ehrlichia, natural killer T cells mediate Ehrlichia-induced toxic-shock–like syndrome, likely via interactions with antigen-presented cells (Mattner et al., 2005). In a second mouse model study, natural killer
cells promote immunopathology and defective anti-Ehrlichial immunity, possibly by decreasing the protective immune response (Ismail et al., 2007). Similarly, mice infected with Anaplasma phagocytophilum develop lesions as a result of immunopathological mechanisms from natural killer and natural killer T cells rather than bacteria-mediated injury (Martin et al., 2001).
The hypothesis that HGA may be an immunopathologic disease as a result of macrophage activation and hemophagocytic syndromes was tested in a cohort of 42 well-corroborated HGA patients. High levels of triglycerides, significantly elevated levels of ferritin, and a variety of cytokines classically seen with hemophagocytic syndromes and macrophage-activation syndromes were found. Furthermore, severity was directly related to the levels of triglycerides, ferritin, and interleukin-12 in these patients, as well as to the the ratio of IL-10 to interferon gamma as a reflection of the interplay between the TH1 and TH2 axis.
The immunologic tests used to diagnose HME and HGA are problematic. They rely on specific antigens to detect antibodies to the pathogens, and the presence of disease is considered confirmed if there is seroconversion or a fourfold rise in antibody titer. However, patients typically do not have detectable antibodies at the time they are diagnosed, so clinicians must decide to treat before the diagnosis has been confirmed. In fact, according to the CDC, only 11 percent of reported HME cases and only 8 percent of reported HGA cases were based on either seroconversion or a fourfold rise in antibody titers. Instead, 73 percent of reported HME cases and 53 percent of reported HGA cases relied on a single serum antibody test result as the laboratory basis for the diagnosis.
Information on the sensitivity and specificity of serologic tests is limited. For HME, the Immunoglobulin G (IgG) or IFA has greater than 80 percent sensitivity, but information on the test’s specificity is lacking. For HGA, the sensitivity of IgG IFA is 82–100 percent, but that of immunoglobulin M (IgM) IFA is just 27–37 percent, which makes it less reliable as a diagnostic test. The specificity of these assays in detecting HGA can be as low as 82 percent, although this improves if patients infected with Ehrlichia chaffeensis are omitted because of serologic cross-reactivity.
Knowledge Gaps and Research Opportunities
A number of questions require additional research, including
-
What is the natural history of HGA? Do patients with HGA have persistent health problems after recovering from acute infection? A retrospective study in Wisconsin looked at individuals during the 12 months after they had been diagnosed with HGA. HGA patients had significantly more fever, shaking chills, sweats, and fatigue, as well as
-
higher levels of bodily pain and lower levels of relative health than did individuals in a control group.
-
Clinical study groups are needed to obtain a critical mass of patients, standardize approaches, and develop sample repositories.
-
Why does the severity of HGA vary so widely? Is the severity of disease determined by host or pathogen? What are the microbial determinants and human genetic predispositions? What is the true pathogenesis and immunopathogenesis of HGA and pathogen-defined injury to tissues and organs?
-
How accurately are HGA cases identified? Are existing acute-phase diagnostics sufficient? Does the poor predictive value of serology hamper effective diagnosis? How often does coinfection with other tick-transmitted pathogens occur? What are the short- and long-term consequences of proven coinfection?
RICKETTSIA DISEASES: SPECTRUM OF DISEASE, SPATIAL CLUSTERING, AT-RISK POPULATIONS, AND RESEARCH NEEDS
Captain Jennifer H. McQuiston, D.V.M., M.S., U.S. Centers for Disease Control and Prevention
Worldwide, numerous Rickettsia species are human pathogens that can be transmitted to humans by ticks, other arthropods, and fleas. In the United States, Rickettsia rickettsii, which causes Rocky Mountain spotted fever, is the primary agent for human rickettsial disease. Increasingly, other Rickettsia pathogens are being reported, including Rickettsia parkeri and Rickettsia phillipi; the latter causes 364D rickettsiosis. Rickettsia massiliae is also found in the United States, but there is no current evidence for transmission to humans. Travelers may bring infections caused by other Rickettsia species into the country as well, particularly Rickettsia africae and Rickettsia conorii.
In the United States, several ticks serve as vectors of spotted fever group Rickettsia: the American dog tick, Dermacentor variabilis, in most of the eastern half of the United States; the Rocky Mountain wood tick, Dermacentor andersoni; in the west, is the vector for R. rickettsii. The brown dog tick, Rhipicephalus sanguineus, where it is increasingly appearing as a vector of R. rickettsii. The Gulf Coast tick, Amblyomma maculatum, transmits R. parkeri in the Southeast. Finally, the Pacific Coast tick, Dermacentor occidentalis, which lives primarily in the Pacific Northwest, is associated with 364D rickettsiosis.
Rickettsia pathogens cause a spectrum of illnesses. Rocky Mountain spotted fever is the most documented Rickettsia infection in the United States and was first recognized in the late 1800s. It infects endothelial cells,
which line blood vessels, causing damage to the vasculature. Patients with Rocky Mountain spotted fever typically present with fever and headache, and approximately 90 percent of them also have a rash.
Rickettsial infections generally respond to treatment with tetracycline-based antibiotics, such as doxycycline, if administered in a timely manner. However, if untreated, the disease progresses, affecting most organ systems and causing death in up to 10 percent of cases. Furthermore, patients who recover may experience long-term health effects, including impaired hearing from neurologic damage; cognitive deficits; and gangrene of fingers and toes caused by damage to blood vessels.
Rocky Mountain spotted fever is the most potentially fatal known rickettsial illness. Prior to the advent of antibiotic therapy, the fatality rate ranged from 20 to 90 percent in published case series. The fatality rate in more recent studies ranges from 3 to 20 percent (Conlon et al., 1996; Buckingham et al., 2007; Martinez-Medina et al., 2007). In national surveillance summaries, fatality rates decreased from about 4 percent (Dalton et al., 1995) in the 1980s to 0.5 percent during 2000 to 2007 (Openshaw, 2010). This decrease may in part be due to changes in the national surveillance system and the differences in how physicians are diagnosing Rocky Mountain spotted fever. Among confirmed cases, the mortality rate from 2000–2007 remained at 3 percent. Reducing these deaths is important because it is a wholly preventable infectious disease, and prompt antibiotic treatment will further reduce fatalities.
Other spotted fever rickettsial diseases generally result in less severe illness. Rickettsia parkeri and 364D Rickettsia cause an eschar-associated illness, fever, and a rash. These patients, however, may not come to a public health official’s attention unless an individual grows concerned or a physician pursues additional testing, for example, to rule out cutaneous anthrax or black widow spider bites.
Testing for Rickettsia relies on detection of antibodies. As Rickettsia infect the endothelial cells lining blood vessels and do not circulate in the blood, PCR using whole blood is not often useful to detect the pathogen except during severe, fulminant infections. During the early stages of illness, PCR can detect Rickettsia in skin biopsy tissue taken from a rash site, but most physicians are reluctant to take such a biopsy. IFA is the predominant diagnostic test used by healthcare providers and relies on R. rickettsii antigens, but cross-reacts with antibodies to other Rickettsia species. Single serum samples cannot be used to confirm infection because an antibody is long lived, and single positive tests may reflect past infection. Furthermore, a single serologic test may appear negative in the first week of infection, before an antibody has had time to rise to a detectable level. Infection is best confirmed using two serologic tests spaced a few weeks apart, and documenting a rise in antibody titers associated with acute infection.
Serologic tests also have less specificity than PCR, meaning that a positive serologic test indicates only that the patient is infected with a spotted fever Rickettsia, but does not identify which one. In 2010, the CTSE changed the name of the reporting category to spotted fever rickettsioses to more accurately describe the data being collected relative to this lack of ability of the serologic test to determine the specific Rickettsia.
The CTSE, in collaboration with the CDC and public health officials from state heath departments, determines which diseases to include in the national notifiable disease list and the criteria for inclusion of individual cases. Cases reported to the CDC represent only the tip of the iceberg because cases can be missed at the physician, local, or state level. What these reports have shown, however, is that the reported incidence of spotted fever rickettsiosis has increased steadily (Figure 5-6), although some of the increase may reflect a change in case definition in 2004 to include cases diagnosed by serologic tests such as enzyme immunoassays for IgM antibodies in addition to IFA. However, there are other possible explanations, including environmental changes, increasing exposure to ticks, or a rising number of ticks. The parameters of the surveillance system do not allow a determination of the contribution of each factor to the reported increase in incidence.
Although people living anywhere in the continental United States are at risk for Rocky Mountain spotted fever, there is spatial clustering of cases in the South Central and Southeast regions. Five states—Arkansas, Missouri, North Carolina, Oklahoma, and Tennessee—account for more than 64 percent of reported cases (Openshaw, 2010). These cases can be analyzed
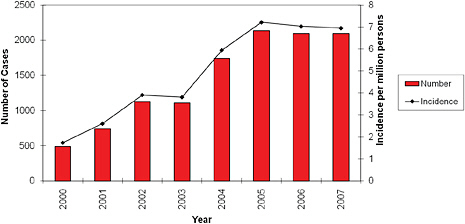
FIGURE 5-6 The number of cases and the incidence rate of spotted fever rickettsiosis has increased since 2000.
SOURCE: Jennifer McQuiston, unpublished.
further to determine whether there is a geographic difference in the severity of the disease. West Tennessee reports the highest proportion of patients who are hospitalized with complications or die from Rocky Mountain spotted fever. North Carolina, by contrast, sees less hospitalization and lower case fatality rates than the national average. Current information is insufficient to determine the cause of the geographic differences. For example, the differences could result from a different strain of Rickettsia or from differences in diagnosing or reporting. This represents a current research gap.
Risk factors for Rocky Mountain spotted fever include poverty and age. Although many individuals associate tick-borne diseases with higher socioeconomic status, because persons in this group engage more frequently in outdoor leisure activities, states with the highest incidence of Rocky Mountain spotted fever fall below the median socioeconomic status for the United States. This may reflect a lack of education about these diseases and prevention strategies, inability to treat their dogs for ticks, and lack of access to health care. The influence of poverty on incidence rates remains a major research gap.
Although the incidence of Rocky Mountain spotted fever increases with age, it is worth noting that the case fatality rates are highest among children ages 9 or younger. One reason for the higher number of fatalities may be the reluctance of physicians to prescribe doxycycline, a tetracycline-based antibiotic that greatly reduces mortality rates, to children. In a survey of healthcare providers in Tennessee, more than half reported that they would prescribe a non-tetracycline antibiotic for suspected Rocky Mountain spotted fever in a child under age 8. This may be due to the idea that tetracycline-based antibiotics stain developing teeth. Current research suggests that doxycycline does not stain teeth, and even if it does, the parents and the patients might well decide that the risk is outweighed by the decreased risk of death.
American Indian populations are also at higher risk of contracting Rocky Mountain spotted fever, with approximately 16 cases per million people versus a national average of 7 cases per million. The rate among American Indians is also about four times that of other racial groups, and American Indians die from their infections at four times the rate of other racial groups. The reasons for these high rates are unclear. Two theories are that cultural differences could put American Indians at higher risk of exposure to ticks, or underlying health disparities could predispose them to severe disease.
In 2002, an outbreak of Rocky Mountain spotted fever began in eastern Arizona—a region where the disease had never before occurred. Although ticks are not often associated with desert climates, this outbreak was linked to the brown dog tick, which can survive in hotter, drier climates. The outbreak was associated with tribal lands with large numbers of stray dogs, which supported large numbers of ticks. Since 2002, there
have been 95 human cases of Rocky Mountain spotted fever (and climbing) and 9deaths among about 30,000 people. Compared to the national case rate, this translates to 527 cases per million on the reservation versus the national rate of 7 per million. The epidemiology of Rocky Mountain spotted fever also differs markedly in this region. The median patient age is only 8 years old, and more than half the fatalities have occurred among children younger than age 4. That makes Rocky Mountain spotted fever a significant contributor to childhood mortality on affected reservations.
The brown dog tick is of continued interest to public health officials because it has also been associated with urban outbreaks of tick-borne diseases. For example, in 2009, more than 1,000 suspected cases of Rocky Mountain spotted fever occurred in urban Mexicali, Mexico. As in Arizona, this outbreak was associated with the brown dog tick, high numbers of stray dogs, and intense transmission in home and peridomestic environments. It is of interest to know whether the brown dog tick might be contributing to Rocky Mountain spotted fever in other parts of the country as well, but just not recognized as the vector in these cases.
Knowledge Gaps and Research Opportunities
Areas for future study are described below:
Diagnostics
-
Develop diagnostic assays, both serologic and molecular, that can identify patients during acute illness and differentiate the causative Rickettsia species.
-
Better assess the sensitivity and specificity of new commercial serologic assays, including the IgM enzyme immunoassay.
-
Improve surveillance and research on human infections caused by tick-borne Rickettsia species other than R. rickettsii.
Emergence
-
Explore the role of the brown dog tick in the ecology of Rocky Mountain spotted fever.
-
Understand the ecological cycle of R. rickettsii in the brown dog tick, including the role of dogs as a possible reservoir.
-
Improve surveillance of infections caused by other species of spotted fever Rickettsia.
-
Given that ticks are acutely sensitive to moisture, precipitation, and temperature, investigate the role of climate change in the emergence of spotted fever rickettsiosis.
Special Populations
-
Investigate preventable factors that contribute to higher mortality from Rocky Mountain spotted fever among children and American Indian populations, and in certain geographic areas.
-
Clarify links between Rickettsia infection and low socioeconomic status.
-
Investigate differences in pathogenicity among Rickettsia strains in different geographic areas.
-
Develop new antibiotics and prevention strategies specifically for rickettsial infections, particularly for pregnant women and high-risk populations for whom antibiotics are contraindicated.
DISCUSSION SESSION
In the discussion session following the presentations of Krause, Dumler, and McQuiston, participants focused primarily on three areas: surveillance, chronic disease, and ongoing research into other potential pathogens.
The differences in severity of Rickettsia spotted fever diseases are an understudied area. One participant noted that the distribution map of Rocky Mountain spotted fever bears a remarkable resemblance to the distribution of Amblyomma americanum, the Lone Star tick, and asked whether there is any new information about the relationship of the Lone Star and R. amblyommii to human disease. Dr. McQuiston noted that there is increased interest in the Rickettsia species that are found in Lone Star ticks. No cases of R. amblyommii infection have been confirmed in humans, but some serologic data suggest it could play a role in mild human illness. CDC and other investigators are currently interested in whether the milder form of Rickettsia spotted fever in North Carolina could be related to R. amblyommii infections.
A number of participants commented on the problems with diagnostics, clinician education, and surveillance. One participant noted that many physicians with whom she interacts know very little about TBDs and therefore are not ordering appropriate diagnostic tests. This lack of knowledge, according to the participant, results in the underreporting of surveillance data. Krause concurred and noted that one of the greatest challenges for diagnosis is awareness of these diseases. In regions where babesiosis has been present for many years, physicians are more familiar with the symptoms and usually order the appropriate diagnostic testing, but even here, Babesia testing may not be ordered in a timely fashion because of the nonspecific nature of the symptoms. Physicians practicing in areas where the pathogen recently has been introduced are unlikely to have the same knowledge of babesiosis and may not order the appropriate testing.
Another participant noted the lack of confidence among the public about the serological tests for diagnosing tick-borne infections. Dumler noted that in Rickettsial and Babesia there are a few laboratories that are working on serodiagnostics to clarify the confusing issues, such as cross-reactions between E. chaffeensis and A. phagocytophilum. Dumler stressed that in the future there will be good methods that could be validated using well-corroborated patient serum samples.
Weber furthered the discussion on surveillance by commenting that research at the 11 largest hospitals in North Carolina indicates that in some instances only 15 percent of reportable diseases are actually reported to state agencies and the CDC. In light of this, he suggested that, in addition to other methods targeted toward underreporting, active surveillance methods are considered to strengthen and improve knowledge of incidence of tick-borne diseases.
Another participant shifted the discussion from surveillance to research on disease mechanisms by noting that there is considerable research on acute infection, but less on chronic infection, such as that reported for babesiosis. Krause agreed with the need for more research on long-term effects of babesiosis, but he indicated that these studies are more difficult to do. A number of unanswered questions also remain about acute infections. Dr. Krause noted the need for more information on pathogenesis of acute infections, such as exploring the role of cytokine expression in immunopathogenesis (Krause et al., 2007).
Another participant was interested in whether Babesia could be transmitted through organ transplantation and whether a person who had been cured of Babesia should be an organ donor. Although the panel was unable to cite a report that demonstrated this type of transmission, Krause commented that the possibility of transmission through organ donation is likely and worthy of further research.
Another clinician participant questioned the length of persistent PCR positivity in Babesia infection and its relationship to Lyme disease. Krause noted that the presence of amplifiable babesial DNA may persist for longer than 2 years, but such persistence was not necessarily indicative of active parasitemia (Krause et al., 1998). The question that still needs to be answered is whether amplifiable Babesia DNA from blood correlates directly with active infection.
PANEL DISCUSSION SESSION
The panel focused on three main themes: surveillance, coinfection, and reinfection and relapse. Although the topics varied, overall the panel indicated that research lagged far behind the needs of the community, and that further research was at the core of addressing these deficiencies.
Surveillance systems, such as the National Notifiable Disease List, are passive in nature. One participant asked whether there is a need to enhance the surveillance for tick-borne diseases by supplementing the current efforts with an active surveillance program. McQuiston suggested an enhanced surveillance model that would focus on specific geographic regions and use data obtained from active surveillance to extrapolate and more accurately quantify the national disease burden. Such surveillance methods, although promising, are not without barriers. Such barriers included inadequate physician education and knowledge of disease presentation and the availability of funding to adequately implement such programs. The panel recognized that an adequate active surveillance model would require physicians to be able to identify and diagnose illness. For this system to be effective, physicians, patients, and, potentially, patient advocacy organizations would need to be able to identify classical and less common manifestations of the respective diseases. This ability was seen as especially important in cases of Lyme disease where the classic presenting signs and symptom of erythema migrans may be absent in up to 20 percent of infected individuals. Inadequate funding was mentioned as a barrier to research, particularly for research on Rickettsia, Ehrlichia, Babesia, and Anaplasma infections, which generally receive less funding than Lyme disease research does. For example, an active surveillance program in Tennessee has an estimated cost of $120,000 for four counties for a year.
The panel also discussed the usefulness of actively surveying the disease vectors and their hosts. McQuiston indicated that, in terms of Rickettsia, the level of antibody in semi-domesticated dogs could indicate areas that pose a higher risk for human disease and that these data currently are being used to encourage surveillance in these areas. For example, it was noted that in eastern Arizona, when there is a seroprevalence of approximately 5 percent among the canine population, there may not be a significant rate of transmission to humans. Cases of human infection begin to present when host seroprevalence rates rise above this baseline (in the affected region of Arizona, human cases were noted with a canine seroprevalence background of more than 70 percent (McQuiston, 2011). Krause mentioned a survey designed to test ticks for Anaplasma, Babesia, and Borrelia and to create national prevalence maps that highlight areas with a high concentration of infected ticks. He stated that these types of studies would be very helpful in understanding where Babesia and Anaplasma infections were occurring. He also noted that testing of the ticks will result in public health officials having a better understanding of the geographic distribution of pathogens and how the disease is changing over time as it is not a one-to-one correlation between ticks and pathogens.
The use of surveillance data to identify potential at-risk populations was briefly discussed. There do not appear to be significant racial differences
among infected persons, except for Native Americans who appear to be at greater risk for Erlichia chaffeensis and Rocky Mountain spotted fever. In addition, McQuiston commented that some data show race-related factors and disease severity, such as more severe Rocky Mountain spotted fever disease in African-Americans with a glucode-6-phosphate dehydrogenase (G6PD) deficiency and more severe babesiosis in some children with sickle-cell disease. Because data on disease in minorities is lacking, however, correlating these associations on a national scale is difficult.
Because many ticks are coinfected with more than one pathogen, human coinfection is of growing concern. Luft noted the need to broadly examine the issue, pointing out that some data suggest that pathogens may act synergistically to worsen health outcomes. Wormser said that his laboratory is interested in coinfection, but there is difficulty determining which patients are coinfected, have sequential infection, or simply have an abnormal laboratory test. He noted there is a need for research to improve diagnostic assays to distinguish different pathogens and the immunologic responses they invoke. The issue of coinfection within the tick and tick biology is an area for further research, noted Luft. Currently, it is difficult to discern which pathogen has infected an individual. He noted that the technology available today should help researchers study these coinfections systematically. A participant noted that there are interactions among microbes within ticks, including the pathogenic and nonpathogenic microbes. In ticks infected with related Rickettsia pathogens, nonpathogenic Rickettsia agents may inherit more pathogenic properties.
The influence of multiple pathogens within the host vector has been understudied. Krause noted that in experiments in the Peromyscus leucopus mouse, the effect of coinfection can vary according to the timing with which these organisms are introduced (e.g., simultaneous infection by a single coinfected tick or sequential infection from a second tick bite). He further noted that at least Babesia and Borrelia may affect each other in the natural reservoir host, which has implications in terms of the transmissions of these diseases. Some evidence shows that Borrelia may enhance the transmission or the infection intensity of Babesia in the reservoir mouse. Understanding the various interactions of multiple pathogens, both in the vector and in the pathogen reservoir hosts, could provide insight into the natural history of the disease, disease epidemiology, and disease transmission.
Finally, the panel discussed its opinions on reinfection and relapsing disease. In terms of Lyme disease, the panelists held divergent views on whether the disease was capable of relapsing or whether persistent symptoms following treatment indicated subsequent reinfection following a cure. Wormser noted that between 1 to 4 percent of patients become reinfected with the B. burgdorferi. In a study of 17 patients from whom the pathogens were cultured and genotyped, it was found that none of the new pathogens
were the same genotype as those that caused the prior infection. Luft agreed that reinfection does occur, but given the nature of Lyme disease, it was likely the disease was a relapsing process characterized by periods of active and inert disease. Krause noted that in his study on the Block Island cohort, he did not find evidence of relapsing disease (Krause et al., 2006). Approximately 10 percent of the population reported a repeat episode, and several lines of evidence indicated that it was due to reinfection as opposed to relapse. First, the subsequent reported infection episodes occurred during the tick transmission season and not during the winter when tick transmission is greatly reduced. Second, people with repeated infections had a high degree of exposure to ticks. Third, the location of repeated erythema migrans rash lesions were at different body sites than the initial infection. Fourth, repeated episodes did not occur within a year of the initial episode and usually were separated from the first by several years. For most relapsing illnesses, subsequent episodes occur in the first month or two after the initial illness. Finally, one component of the study was an annual serosurvey. Most of the patients had clearance of their initial antibody test, and one would not expect this if there was a persistence of infection. Furthermore, when patients presented with a second infection, their responses were amnestic immunologic responses with a high IgG concentration and an absence of an IgM response.
With the exception of babesiosis in immunocompromised individuals (Krause et al., 2008), little evidence supports the idea of relapse in tick-borne diseases. Dumler noted that there is not clear evidence of persistent infection of Ehrlichia and Anaplasma in humans. Most infection in humans with either Ehrlichia or Anaplasma are either self-limited or are cured with appropriate treatment. Relapses of either agent appear to be a very rare occurrence, and most subsequent presentations of disease are the result of new infections. McQuiston commented that there is also a lack of data in terms of Rickettsia infection, but most Rocky Mountain spotted fever infections are acute infections that either are self-limited, and fatal, or resolve with antibiotic treatment. She indicated that although rare, most permanent or long-term consequences are the result of damage sustained by the body during the course of the acute infection. However, it was mentioned that difficulty in isolating the pathogen does not indicate an absence of persistent infection. There is a Rickettsia species, Rickettsia prowazekii, that causes epidemic typhus and has been shown to persist in the body for very long periods of time and to reappear decades later in a form of disease called Brill-Zinsser disease. This type of event has not been observed with Rickettsia rickettsii infections, however.
In closing, some members of the panel noted the need for a consensus on the terminology and case definitions used to report and discuss tick-borne diseases. Some members of the panel indicated that many surveillance
efforts were hindered not only because many physicians did not understand the clinical manifestations of disease but also because there was a lack of commonality in defining these diseases.
GENETIC AND ACQUIRED DETERMINANTS OF HOST SUSCEPTIBILITY AND VULNERABLE POPULATIONS
David Jay Weber, M.D., M.P.H., University of North Carolina School of Medicine and School of Pubic Health
Tick-borne diseases result from the interaction of tick biology (host[s], climate, and species), tick exposure (e.g., residence, occupation, and recreation activities), and human biology (e.g., age, gender, and treatment). Acquiring a tick-borne disease is the result of a two-pronged process of exposure and infection. Each of these processes plays a role in acquisition of disease and determination of risk.
The risk of exposure is dependent on two variables: proximity to the tick environment and barriers to tick interaction. Proximity to the tick environment is a measure of the distance to and amount of time in the tick environment. Living in a rural area puts one at greater risk than does living in an urban area; however, this risk is not equal across all areas. Wooded areas, for example, carry greater risk of exposure to ehrlichiosis than pastures because they have a higher prevalence of host animals (Standaert et al., 1995). The geographic region in which persons reside is crucially important because the presence of tick-borne diseases is highly geographically dependent. In the United States, Lyme disease, babesiosis, and Rocky Mountain spotted fever (RMSF) all have important geographic predilections.
In terms of occupation, people who work outdoors are at higher risk for tick exposure than are office workers, although different outdoor occupations vary in their risk for tick-borne diseases. For example, general outdoor laborers were shown to be at a greater risk for Lyme disease (Schwartz et al., 1994), farmers for tick-borne encephalitis (Cisak et al., 1999), and forestry workers for Lyme disease, tick-borne encephalitis, and anaplasmosis (Cisak et al., 1999, 2005; Adamek et al., 2006). Recreational activities may also increase one’s risk of exposure because many outdoor activities place participants within the tick environment. For example, an increased risk of Lyme disease has been demonstrated for gardeners and orienteers (Fahrer et al., 1991, 1998) whose activities take place in open, outdoor areas. In some cases, one’s ability level as well as one’s recreational preference may influence the level of risk associated with a particular activity. For example, Standaert and colleagues (1995) found that poor golfers are at an increased risk of ehrlichiosis due to the greater amount of time spent in the higher, thicker grass of the rough compared with those who
play primarily from the manicured grass of the fairway and green. Finally, interspecies relationships also can influence one’s exposure to disease. Pet owners may be at increased risk of Lyme disease (Schwartz et al., 1994) and RMSF (Demma et al., 2006) because of ticks that have fallen from domesticated animals or household pets.
The relative effectiveness of barriers to tick interaction varies with proximity to the tick environment. In low-proximity areas, these barriers are less important because the overall exposure risk is low. For example, among those in wooded rural areas (high proximity), those with unfenced backyards are at greater risk than those whose property is at least partially enclosed (Connally et al., 2009). Moreover, evidence has shown that practicing personal, protective measures, such as wearing long sleeves and pants, taping pants or tucking them into boots, and the use of insect repellents or insecticides such as permethrin-treated clothing can reduce risk of tick exposure by up to 93 percent (Vaughn and Meshnick, 2011).
Although exposure to a tick is a necessary part of the development of tick-borne disease, it is not sufficient to produce disease as an individual needs to be infected by the pathogenic agent. Like exposure, many factors can affect one’s risk of becoming infected with a pathogen and developing severe or fatal disease. Many risk factors are associated with behavioral and biological indicators of the exposed individual or of the tick environment. Behavioral factors associated with increased risk of infection include failure to perform regular tick checks after interacting in the tick environment (Smith et al., 2001; Connally et al., 2009) and lack of use of tick insect repellents (Standeart et al., 1995; Smith et al., 2001). Biological risk factors that have been identified for RMSF include being Caucasian (Dalton et al., 1995) or of Native American descent (Holman et al., 2009), being male (Dalton et al., 1995), and being between the ages of 5 and 9 years old (Dalton et al., 1995). Age has also been indicated as a risk factor for Lyme disease particularly for individuals ages 10 to 19 (Smith et al., 2001). In addition to behavioral and biological indicators, the nature of some infections may play a role in the risk of infection. For instance, because Babesia species primarily infect red blood cells, there has been evidence indicating a higher risk of infection among blood transfusion recipients than the general population (White et al., 1998; Cable and Leiby, 2003).
Beyond the risk for infection, some factors have been associated with the likelihood of severe disease and hospitalization. For RMSF, risk factors for severe disease and hospitalization include being American Indian (McQuiston et al., 2000); the geographic location in which the infection occurs, particularly in North Carolina and Oklahoma (Adjemian et al., 2009); and the use of chloramphenicol instead of tetracycline-based antibiotics (Dalton et al., 1995). In addition, in terms of Babesia species, splenectomy (Sun et al., 1983; White et al., 1998) and immunosuppression from HIV or cancer (Vannier et
al., 2008) have been shown to increase one’s risk of severe disease. Research indicates that genetics also may predispose individuals to severe disease. Genetic predispositions include the presence of a toll-like receptor (TLR)-9 in Crimean-Congo fever (Engin et al., 2010a), homozygosity for CCR5 delta 32 in tick-borne encephalitis (Kindberg et al., 2008), and HLA-DR4 and DR2 in Lyme disease (Steere et al., 1990; Kalish et al., 1993).
Research further indicates that some tick-borne diseases carry additional risk factors for fatal disease. In the case of RMSF, anything that delays treatment by as little as 4–5 days, including the absence of rash or headache and the presence of fever, results in a higher risk of fatal disease. In addition to the fact that many doctors are not trained to recognize early-stage disease, off-season presentation and no history of tick attachment contribute to delayed diagnosis and treatment and may further increase the risk of fatalities (Hattwick et al., 1978; Dalton et al., 1995; Kirkland et al., 1995; Holman et al., 2001). Age also seems to be a factor in determining the risk of fatal infection. Individuals younger than age 5 and older than 40 appear to be at increased risk of dying from RMSF than are healthy individuals between 6 and 39 years old (Dalton et al., 1995; Holman et al., 2001; Chapman et al., 2006b). In addition, a high risk of fatal disease has been associated with increased serum creatinine on presentation and the presence of neurological involvement (Conlon et al., 1996) as well as with being African American with a G6PD deficiency (Walker et al., 1983). This is not to say that all genetic predispositions are indicative of severe or fatal disease. For example, TLR-2 heterozygous for SNP Arg753Gln may provide increased protection against late-stage Borrelia burgdoferi infection (Schroder et al., 2005).
Despite the advantages of knowing the aforementioned high-risk associations, it is important to recognize that these data are not without limitations. Determining risk factors for tick-borne disease requires sufficiently sophisticated tests to detect disease accurately, and the development and conduct of surveillance studies capable of multivariate analysis to delineate independent risk factors that may be thrown together or overshadowed in case-control studies. Furthermore, despite disparate indications, it is difficult to isolate risk factors for exposure from risk factors for infection, and for this reason, many case-control studies measure exposure rate based on the number of infections reported rather than recording the total number of actual tick exposures or tick bites, an outcome measure that may not always be appropriate. In spite of these limitations, there are sufficient scientific data to develop policies that would lead to risk reduction and to design safe and cost-effective intervention studies to reduce exposure and disease. Furthermore, these data can be used to develop educational programs for healthcare providers regarding recognition and proper treatment of TBDs. In conclusion, additional research is needed, especially on genetic factors to further define risk factors for infection and severe or fatal TBDs.
AT-RISK POPULATIONS FOR BABESIA
Peter J. Krause, M.D., Department of Epidemiology and School of Public Health, Yale School of Medicine
As discussed earlier in the chapter, babesiosis is associated with a number of clinical syndromes, including asymptomatic infection; viral-like illness (fever, chills, sweats, headache, fatigue); persistent, relapsing illness; and fulminant illness and death. Adults over age 50 are at risk for more severe disease manifestations (Ruebush et al., 1981; Vannier et al, 2008). As the U.S. population ages, babesiosis will have an increasingly important impact on the health of this segment of the population. Immunological problems associated with aging or comorbid diseases, such as lung, renal, or liver disease, contribute to the difference. Patients with AIDS, malignancies, or splenectomy are at increased risk for persistent, relapsing illness (Gubernot et al., 2009). Complications of babesiosis that are seen more frequently in immunocompromised people consist of acute respiratory distress syndrome, disseminated intravascular coagulation, congestive heart failure, coma, and renal failure (Leiby et al., 2011).
The specific mechanisms that lead to increased severity of babesiosis continue to be the subject of investigation. A DBA/2 mouse model has been developed to investigate age-related severity of babesiosis (5). Edouard Vannier and I are carrying out a study to elucidate genetic factors that may account for severe babesiosis in people over age 50.
We previously carried out a retrospective case-control study of human Babesia microti infection among 14 highly immunocompromised patients who experienced persistent, relapsing babesiosis and 49 control subjects who experienced a typical course of babesiosis infection that resolved after 1 to 2 weeks (Gubernot et al., 2009). The subjects were enrolled between 1991 and 2005 and resided in Connecticut, Massachusetts, New York, Rhode Island, and Wisconsin. Eleven of the 14 patients with severe relapsing disease had malignancies, including 8 with B-cell lymphoma, while 10 were asplenic. Patients with B-cell lymphoma were given Rituximab, a monoclonal antibody directed against the protein CD20 on B cells. For the patients with relapsing illness, the percentage of infected red blood cells ranged from 2 to 75 percent, compared with 0.5 to 10 percent for the control group. The group with relapsing illness required 2–10 courses of anti-Babesia antibiotic to clear infection while the control group required only a single course of antibiotic. The median length of therapy was 13 weeks for the relapsing illness group (range 4–102 weeks) and 1 week (range 0.5–1.5 weeks) for the control group. About a quarter of the relapsing illness group died compared to none in the control group. Resolution in the immuno-compromised patients required several courses of anti-Babesia therapy, but once a therapy was found to be effective, duration was a critical factor for
success. All the patients who cleared infection were given a minimum of 6 weeks of therapy, including therapy that continued for at least 2 weeks beyond the first noted absence of parasites on thin blood smear. These results indicate that people who suffer from broad-based immunosuppression are at risk of persisting, relapsing Babesia microti infection and require prolonged antibabesial therapy. They also suggest that B cells and anti-Babesia microti antibodies are important in clearing Babesia infection, although differences in the relative contribution of T cells and B cells in clearing infection requires further study.
One of the first well-recognized risk factors for severe babesiosis was asplenia (Krause et al., 2002). The spleen removes red blood cells that are infected or senescent, and macrophages and other immune factors present in the spleen may help to eliminate intraerythrocytic pathogens. Interestingly, asplenia does not always result in severe Babesia illness. This suggests that Babesia strains with different virulence also may contribute to disease severity. An editorial in the New England Journal of Medicine suggested that individuals who lack a spleen should avoid owning property or visiting an area that is highly endemic for Babesia microti because such activities could potentially be life threatening (Persing et al., 1995). Clinicians treating patients who lack a spleen, have a malignancy, are HIV infected, or are immunocompromised for other reasons should provide information and counseling regarding the potential for life-threatening babesiosis in regions that are highly endemic.
Knowledge Gaps and Research Opportunities
There are several critical questions that offer research opportunities pertaining to the effect of babesiosis on populations at risk for severe and fatal outcome:
-
What are the causes of life-threatening babesiosis in people who are over 50 but otherwise healthy, and how can they best be treated and the problem prevented?
-
What are the causes of life-threatening babesiosis in people with asplenia, HIV, malignancy, and pre-morbid conditions, and how can they best be treated and the problem prevented?
-
Why do people who acquire babesiosis through blood transfusion experience more severe babesiosis, and how can they best be treated and the problem prevented?
DISCUSSION
During the discussion of the At-Risk Populations Panel, panelists and participants presented a number of points that addressed an array of topics,
including public health communication, research potential and barriers, transmission in at-risk communities, and disease severity.
King posed the question of how to develop public health messages to educate the public on tick-borne diseases and disease prevention strategies. Weber noted that targeted messages need to be directed to high-risk populations, but public health messages may not be very useful. Targeting the outdoor workforce population might be particularly effective because the workforce model could permit the development and enforcement of preventive policy measures. Public health messages are apt to be less effective because it is difficult to compel behavioral changes, such as wearing long sleeves even on hot days or routinely checking for ticks. Instead, Weber suggested the need to continue to develop permethrin-impregnated clothing and other technological advances to limit exposure.
One participant questioned whether it is possible for Babesia to be transmitted through national blood donation programs. Krause observed that, although the chance of acquiring babesiosis from a blood transfusion likely is very low, Babesia microti is the most common infectious agent transmitted through the blood supply in the United States (FDA, 2008). This has attracted the attention of the CDC and the NIH as a potentially significant public health problem. Screening for potential active B. microti infection in blood donors currently relies on self-reporting by the blood donor regarding a history of prior infection and results in lifetime prohibition from donation for those who report having had babesiosis.
In addition, the Food and Drug Administration has approved a screening program at the Rhode Island Blood Center that uses laboratory-based B. microti screening, including antibody analysis and PCR, to test donor blood for use in newborn infants and persons with sickle-cell disease (Gubernot et al., 2009; Young and Krause, 2009). The purpose of this study is to observe whether the screening will affect the infection rate in these populations. Although confident this type of screening will be effective in reducing the incidence of transfusion-transmitted babesiosis, Krause indicated that further assessment is necessary to develop a laboratory-based screening protocol that will optimally balance the need to minimize the risk of B. microti transmission with the need to minimize discarding blood or blood products that may not be actively infected. Any screening system developed will need to address the issue of screening in endemic and non-endemic areas. A variety of screening approaches may be necessary to meet the needs of different populations with varying levels of risk. Use of pathogen reduction technologies to destroy Babesia within donated blood units is another potential option for reducing transfusion transmitted babesiosis (Gubernot et al., 2009).
Another participant questioned the potential of the Babesia pathogen to infect cells other than erythrocytes. Although human Babesia currently
is believed to infect only red blood cells, further research may include investigation of the possibility of alternative sites of infection during various stages in the pathogen life cycle. Neurologic manifestations of Babesia, such as coma, have not been shown to be the result of Babesia infection of neuronal tissue and may occur because of electrolyte imbalance or other side effects of the overall infection.
The disease severity of Babesia duncani and its implication for treatment was another topic of discussion. In response to a participant’s question, Krause indicated that B. duncani infections that have been reported have been more severe than B. microti infections (Persing et al., 1995). He added that although it is generally true that initially the most critical manifestations of a disease receive the most attention, there is evidence in a hamster model that B. duncani is a significantly more severe disease than B. microti is (Wozniak et al., 1996). Krause advocated aggressive treatment for patients diagnosed with B. duncani, including the possibility of exchange transfusion to replace infected red blood cells and remove toxic by-products of infection.
Finally, one participant asked whether there may be some utility in investigating antibiotic alternatives to cure Babesia. Krause indicated that there is limited evidence that the anti-malarial drug Artemisinin may be useful in clearing a babesial infection, but that more research is needed to determine the effectiveness of this and other alternative therapies, including new drugs and herbal remedies (Krause et al., 2008).
Concluding Thoughts on Surveillance, Spectrum, and Burden of Tick-Borne Disease, and At-Risk Populations
Gordon Schutze, M.D., Baylor College of Medicine
Creating a repository for specimens of blood and cerebrospinal fluid from patients with tick-borne diseases can improve the accuracy of diagnostic tests and disease diagnosis. Such a repository needs to include demographic information on patients and information on their diagnosis, treatment, and outcome. A well-funded network of researchers investigating tick-borne diseases could maintain such a repository, which also would enable them to advance knowledge of these diseases.
Clinicians clearly need new methods for diagnosing tick-borne diseases, given the genetic diversity among different strains of a pathogen (e.g., Borrelia burgdoferi) and the realization that new species may be responsible for a larger burden of disease than previously recognized (e.g., Rickettsia parkeri in spotted fever rickettsiosis). Clinicians also need better tests to accurately differentiate between patients with acute tick-borne illness and those previously infected with tick-borne disease.




































