Tobacco use is the leading cause of preventable death in the United States, causing more than 440,000 deaths annually and resulting in $193 billion in health-related economic losses each year—$96 billion in direct medical costs and $97 billion in lost productivity (CDC, 2008). Since the first U.S. Surgeon General’s report on smoking in 1964, more than 29 Surgeon General’s reports, drawing on data from thousands of studies, have documented “the overwhelming and conclusive biologic, epidemiologic, behavioral, and pharmacologic evidence that tobacco use is deadly” (HHS, 2010b, p. iii). This evidence base links tobacco use to the development of multiple types of cancer1 and other life-threatening conditions, including cardiovascular and respiratory diseases (HHS, 2004). Smoking accounts for at least 30 percent of all cancer deaths, and 80 percent of lung cancer deaths (ACS, 2012). Despite widespread agreement on the dangers of tobacco use and considerable success in reducing tobacco use prevalence from more than 40 percent at the time of the 1964 Surgeon General’s report to less than 20 percent today, recent progress in reducing tobacco use has slowed. An estimated 18.9 percent of U.S. adults (45.3 million) smoke cigarettes, nearly
________________
1 Smoking heightens the risk of up to 18 different types of cancers, including head and neck cancers, leukemia, and cancers of the esophagus, bladder, pancreas, kidney, liver, stomach, colorectum, cervix, uterus, and ovaries (ACS, 2012; HHS, 2004; Secretan et al., 2009).
one in four high school seniors smoke, and 13 percent of high school males use smokeless tobacco products (CDC, 2012b,g; HHS, 2012).
In recognition that progress in combating cancer will not be fully achieved without addressing the tobacco problem, the National Cancer Policy Forum of the Institute of Medicine (IOM) convened a public workshop, Reducing Tobacco-Related Cancer Incidence and Mortality, June 11–12, 2012, in Washington, DC.2 In opening remarks to the workshop participants, planning committee chair Roy Herbst, professor of medicine and of pharmacology and chief of medical oncology at Yale Cancer Center and Smilow Cancer Hospital, described the goals of the workshop, which were to examine the current obstacles to tobacco control and to discuss potential policy, outreach, and treatment strategies that could overcome these obstacles and reduce tobacco-related cancer incidence and mortality. Experts explored a number of topics, including
• the changing demographics of tobacco users and the changing patterns of tobacco product use;
• the influence of tobacco use on cancer incidence and cancer treatment outcomes;
• tobacco dependence and cessation programs;
• federal- and state-level laws and regulations to curtail tobacco use;
• tobacco control education, messaging, and advocacy;
• financial and legal challenges to tobacco control efforts; and
• research and infrastructure needs to support tobacco control strategies, reduce tobacco-related cancer incidence, and improve cancer patient outcomes.
During the workshop, individual workshop participants raised a number of potential action items to reduce tobacco use and the associated health consequences, including the higher rates of cancer incidence and mortality, as well as suggestions to improve tobacco control policy, research, and advocacy. These suggestions focused on a number of key audiences, includ-
________________
2 This workshop was organized by an independent planning committee whose role was limited to the identification of topics and speakers. This workshop summary was prepared by the rapporteurs as a factual summary of the presentations and discussions that took place at the workshop. Statements, recommendations, and opinions expressed are those of individual presenters and participants, are not necessarily endorsed or verified by the Institute of Medicine or the National Cancer Policy Forum, and should not be construed as reflecting any group consensus.
ing the general public, cancer patients, clinicians, policy makers, advocacy groups, health researchers, and insurers. An overview of key discussion points raised by individual presenters is provided below.
The workshop agenda, statement of task, and speaker biographies are in Appendixes A and B. The speakers’ presentations (PDF and audio files) have been archived at www.iom.edu/Activities/Disease/NCPF/2012-JUN-11.aspx.
Overview of Key Points Highlighted by Individual Participants
Clinicians can improve their patients’ health by
• Recognizing and treating nicotine dependence as a serious chronic medical problem.
• Incorporating tobacco assessment and cessation support as a standard part of clinical care for all patients.
• Discussing the immediate and long-term cardiovascular, pulmonary, cancer, and other related health benefits associated with tobacco cessation at every patient encounter.
• Recommending and/or providing evidence-based tobacco cessation therapy, including counseling and medication, for all patients who use tobacco.
• Providing consistent and repeated counseling for tobacco cessation at every patient encounter.
Cancer care could be improved by
• Accurately identifying tobacco use in cancer patients during and following cancer treatment using structured tobacco assessments and/or biochemical confirmation methods.
• Incorporating the treatment of tobacco dependence into the standard of care for all cancer patients who use tobacco products, to improve treatment outcomes and reduce treatment complications and toxicity.
• Ensuring that all institutions that treat cancer patients have evidence-based tobacco cessation programs as a requirement for accreditation.
• Mandating dedicated cessation support as a standard requirement for National Cancer Institute (NCI) Cancer Center Designation.
Tobacco cessation therapy could be advanced and made more accessible by
• Mandating tobacco assessment and cessation referrals for payment incentives or as a condition of reimbursement for standard medical procedures.
• Ensuring tobacco cessation programs have adequate resources to achieve their missions.
• Ensuring all insurance plans provide coverage for evidence-based tobacco cessation therapy.
• Standardizing electronic medical record fields to document tobacco use status, cessation referrals, and cessation therapy.
• Ensuring all tobacco cessation therapy is personalized to prioritize patient preferences and needs.
• Training health care professionals in evidence-based tobacco cessation therapy and encouraging use of available cessation resources.
• Enhancing referral programs and other partnerships between clinicians/health systems and cessation therapy providers in the public health sector (e.g., state quitlines).
Tobacco control policy and advocacy could be improved by
• Coordinating institutional, local, and national tobacco control efforts and oversight.
• Ensuring tobacco control programs have sufficient resources and funding to achieve their missions.
• Engaging clinicians and clinician societies to join with the public health community to advance tobacco control efforts.
• Anticipating legal challenges from the tobacco industry, and ensuring that communities have the financial resources to defend them.
• Aligning policies and advocacy efforts with the trends of tobacco product use, including dual use of noncombustible and combustible products, and the use of new tobacco products.
Reductions in tobacco use could be facilitated by
• Using Food and Drug Administration (FDA) regulatory authority to reduce the amount of nicotine in tobacco products to nonaddicting levels so that consumers who wish to discontinue use can do so easily.
• Assessing and communicating the relative health risks of new, combined, and alternative tobacco products in a rigorous, evidence-based manner with FDA oversight, so consumers can make informed decisions about the products they use.
• Advancing effective policies and advocacy efforts such as taxes, smoke-free laws, and media campaigns.
• Increasing referrals from clinicians/health systems to public health cessation resources such as quitlines.
Health research could be improved and the evidence base could be expanded by
• Including measures to assess tobacco use and cessation in all cancer clinical trials.
• Prioritizing behavioral and social science research on tobacco use and cessation in conjunction with other health research efforts.
• Evaluating communication strategies to determine the most effective ways to inform the public about the risks of tobacco use and to encourage quit attempts.
• Conducting research to maximize the impact of FDA oversight, such as informing product standards (e.g., with regard to nicotine).
• Assessing the roles that may be played by evidence-based and emerging technologies such as cell phone apps and video games in reducing tobacco use, facilitating cessation, and enhancing cancer care.
CHANGING DEMOGRAPHICS OF TOBACCO USE
Several speakers noted that the average person who smokes today tends to have a different educational and economic background from the typical person who smoked decades ago, when cigarette smoking was more popular, with fewer known risks. People who smoke now tend to have a lower economic and educational status than those who do not smoke, according to Kenneth Warner, the Avedis Donabedian Distinguished University Professor of Public Health at the University of Michigan School of Public Health. He noted that less than 10 percent of college graduates smoke, while those with a high school diploma/GED or less have a smoking
prevalence of 27 to 28 percent. In some blue-collar occupations, smoking prevalence is around 30 percent (CDC, 2011a).
Michele Bloch, acting chief of the Tobacco Control Research Branch at NCI, also stressed that many people who smoke today “live in communities where smoking is normative, and we haven’t done enough to think about how to reach folks that live in [that] environment.” Otis Brawley, chief medical and scientific officer and executive vice president of the American Cancer Society, cautioned against viewing smoking as a racial disparities issue, and instead emphasized that smoking is a socioeconomic and education issue. Brawley noted that some surveys have shown that black high school students have a lower prevalence of tobacco use compared with white students (CDC, 2012b). “I tend to focus … on the fact that this is a problem [and] that everybody needs to stop smoking. This is not just a black problem or a white problem,” he said. “It is an American problem.”
In addition, some studies indicate that individuals with mental illness and/or substance abuse are twice as likely to smoke compared to the general population, and consume nearly half of the cigarettes smoked in the United States (Lasser et al., 2000). Warner noted that about 60 percent of schizophrenic patients smoke—three times the national average (McClave et al., 2010). This high rate of tobacco use has led some to suggest that these individuals may be self-medicating with tobacco, and led to a suggestion by Brawley for mental health and tobacco control advocates to join together to tackle this issue of severe addiction coupled with a high prevalence of mental illness or substance abuse.
Warner also noted that “there is a raging debate about whether many [of the people who smoke today] are hardcore smokers—people who can’t quit or never will quit. That is a very different image [than] smoking as a freely chosen adult pleasure.” Some have contended this could explain, in part, why the decline in smoking prevalence has slowed almost to the point of stagnation. However, others disagree with the premise that people who smoke today are more hard core, noting that many people who currently smoke consume fewer cigarettes and smoke far less regularly.
Jamie Ostroff, attending psychologist, director of the Tobacco Cessation Program for Memorial Hospital, and chief of the Behavioral Sciences Service at the Memorial Sloan-Kettering Cancer Center, emphasized recent studies suggest that evidence-based approaches to tobacco cessation are also safe and effective for people with mental illness or substance abuse disorders (Fiore et al., 2008; Williams and Ziedonis, 2004). Michael Fiore, professor of medicine and director of the University of Wisconsin Center for Tobacco
Research and Intervention, noted that a bigger challenge is getting health care professionals to overcome their reluctance to treat their psychiatric patients’ tobacco dependence because of unwarranted concern that such treatment will worsen their underlying psychiatric condition.
Several speakers and attendees also pointed out the heightened prevalence of tobacco use in the military compared to the overall civilian population, with nearly a third of military personnel reporting they have smoked in the past 30 days and 14 percent reporting smokeless tobacco use (TRICARE, 2009). According to a Centers for Disease Control and Prevention (CDC) study, close to 45 percent of U.S. service members deployed to Iraq and Afghanistan smoke, roughly double the rate of nonmilitary Americans (CDC, 2012e). Warner pointed out that cigarettes are sold at a discount rate on military installations (IOM, 2009a). Brawley added that his impression from interactions with military personnel is that the military medical officers would like to ban cigarette sales from military bases and would like to have the ability to order soldiers not to smoke. But some battle frontline officers argue this is an imposition on the American soldier who is exposed to combat and other stressful conditions. However, Brawley noted that smoking is no longer allowed on submarines, and Fiore added that all basic training facilities must be smoke free (IOM, 2009a; Shanker, 2010).
Richard Hurt, professor of medicine and director of the Nicotine Dependence Center at the Mayo Clinic, suggested that the issue of banning smoking in military facilities should be extended not only to active duty facilities but also to those operated by the U.S. Department of Veterans Affairs (VA). Fiore concurred, noting that the smoking rate of veterans from the Gulf War is higher than that of the same soldiers prior to going to battle (Bastian and Sherman, 2010; Brown, 2009; IOM, 2009a). “This is an important consideration—these soldiers are surviving [the battlefield] only to return with this enormous risk to their future health,” Fiore said. He also called attention to the recent report by the IOM on tobacco use in the military and the challenges tied to this issue (IOM, 2009a).
Several speakers addressed the current smoking behavior of youth. Terry Pechacek, associate director for science in the Office on Smoking and Health at CDC, pointed out that after years of sustained progress in reducing smoking in youth, the decline in smoking appears to be slowing for cigarette use, and is at a standstill for smokeless tobacco, since about 2003 (HHS, 2012). There was a 40 percent decline in youth smoking between 1997 and 2003, but only about half as much of a decline in youth
smoking between 2003 and 2011 (see Figure 1). Pechacek stressed that there are now 3 million more youth and young adults in the United States who smoke than there would have been if progress from 1999 to 2003 in preventing smoking initiation had been sustained (HHS, 2012). Danny McGoldrick, vice president for research at the Campaign for Tobacco-Free Kids, blamed this on a lack of a substantial price increase in cigarettes since 2003. McGoldrick and Tim McAfee, director of the Office of Smoking and Health at CDC, emphasized that although cigarette smoking is still declining slowly among youth (as shown in Figure 1), other tobacco product use is up (see section on Changing Patterns of Tobacco Use), so the progress in stemming overall use of tobacco products may be overestimated.
“The [tobacco industry] is getting all the replacement smokers that it needs,” Pechacek said. “Since the birth cohort of about 1980, we have not really been cutting down the number of smokers being fed into the epidemic.” While the percentage of young people who start smoking has decreased and is lower than it would have been had tobacco control measures not been adopted, when one includes the use of cigars, the progress made in stemming smoking initiation is even less.
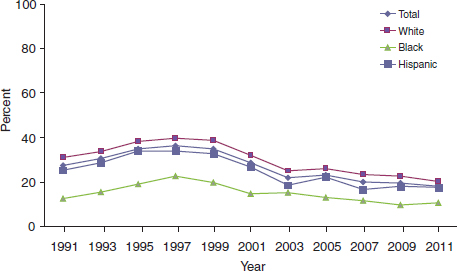
FIGURE 1 Percentage of high school students who reported current cigarette use, 1991 to 2011. For all high school students, cigarette smoking increased from 1991 to 1997 and decreased from 1997 to 2011. The rate of decline in smoking has slowed down from 2003 to 2011 compared to 1997 to 2003.
NOTE: Current cigarette use is defined as smoking at least 1 day during the 30 days prior to the survey.
SOURCES: Pechacek presentation (June 11, 2012); CDC (2012f).
Pechacek noted that the 2012 Surgeon General’s report Preventing Tobacco Use Among Youth and Young Adults stated that prevention efforts must focus on adolescents and young adults through age 25, because data indicate that few start smoking after age 25, whereas nearly 9 out of 10 who smoke start by age 18, and 99 percent start by age 26 (HHS, 2012). Progression from occasional to daily smoking almost always occurs by age 26 (HHS, 2012). However, David Abrams, executive director of the Schroeder Institute for Tobacco Research and Policy Studies at Legacy, said that among a young adult cohort they have been studying, 32 percent of people who have ever used tobacco reported initiation of use after age 18 and 39 percent of people who regularly use tobacco reported progressing to regular use during young adulthood (Rath et al., 2012).
Pechacek pointed out that although around 20 percent of the general population smokes, recent data from the 2011 Youth Risk Behavior Survey found about 40 percent of young adults have tried smoking (CDC, 2012d). “We are underestimating the burden of smoking that is being passed forward into the future if we focus only on the current 20 percent prevalence,” said Pechacek. He added that under current tobacco exposure patterns, about two-thirds of preventable cancers in children born today will not be averted. Abrams also stressed the need to focus on the smoking behavior of young adults “because they are the pattern of use of the future.” Legacy found that 30 percent of 18- to 34-year-olds are dual cigarette and other tobacco product users (Rath et al., 2012). “That is of huge concern,” he said.
There has also been a slowing of progress in reducing the use of tobacco products in older adults, Pechacek noted. He pointed out that the projected prevalence of smoking for 2020, based on current smoking patterns, will be around 17 percent (CDC, 2011d). Howard Koh, assistant secretary for health at the U.S. Department of Health and Human Services (HHS), noted that the perception among too many is that “the tobacco problem has been solved and it is time to move on to something else. But it is time to accelerate and reinvigorate our efforts and reaffirm that tobacco use is the premier public health challenge of our time.” Michael Cummings, professor and codirector of the Tobacco Policy and Control Program at the Medical University of South Carolina’s Hollings Cancer Center, added, “The time to act is sooner rather than later. If the population ceased smoking, this would be tantamount to an effective vaccine against a third of all cancers.” McGoldrick stated it more bluntly: “The goal is to have fewer dead people—this is the bottom line.”
CHANGING PATTERNS OF TOBACCO USE
Although tobacco use has been a common practice for centuries, the development of the modern cigarette in the early part of the 20th century made it easier to inhale tobacco smoke and is driving the current epidemic of smoking, Pechacek noted. Introduction of the cigarette also drove down the use of other less inhalable forms of tobacco smoking, including pipe and cigar smoking (see Figure 2).
Although the cigarette is the most prominent tobacco product currently on the market, several speakers pointed out that the use of different types of tobacco products have increased in recent years. These products include cigars, a type of smokeless tobacco called snus, spit tobacco, dissolvable tobacco products, and the e-cigarette, which is a device that resembles a cigarette and converts a nicotine-laden liquid into vapor.
Often noncombustible tobacco products are viewed as being less hazardous to health than combustible ones, but that thinking may be misguided, according to Pechacek. The use of noncombustible tobacco products is often in addition to cigarette or cigar smoking and not instead of such smoking. He reported that a current survey indicates that nearly 40 percent
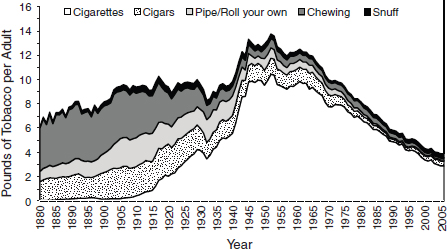
FIGURE 2 Trends in per capita consumption of various tobacco products—United States, 1880–2005.
SOURCES: Pechacek presentation (June 11, 2012); adapted from Giovino (2007).Reprinted with permission from Elsevier.
of all smokeless tobacco users also use cigarettes.3 Among youth and young adults, the reported dual use of cigarettes and smokeless tobacco is even greater; nearly 60 percent among 18- to 25-year-olds and 48 percent among 12- to 17-year-olds. Many “smokeless tobacco users are smoking cigarettes,” Pechacek stressed. “We are not saying that noncombustible sources don’t have a role, but watch out because noncombustible tobacco products are adding on to, rather than replacing, cigarette smoking.”
Pechacek noted that cigar smoking is assumed to be less dangerous than cigarette smoking because it is often not inhaled, but this may not be the case for current or former cigarette smokers who do inhale cigar smoke. Studies have shown that former cigarette smokers who switched to cigars had potentially higher levels of exposure to toxic chemicals and risk of disease than people who smoke cigarettes (NCI, 2012a). McAfee added that the increased consumption of cigars seen in the past few years is being influenced strongly by the growing availability of cigars that are only slightly larger than cigarettes, with their weight adjusted in order to be eligible for preferential tax treatment (CDC, 2012a). He added that these cigars appear to be used functionally more like cigarettes than traditional large cigars. Another factor that may be contributing to cigarette-like cigar uptake is differential FDA regulatory authority for cigars versus cigarettes.
Margaret Foti, chief executive officer of the American Association for Cancer Research (AACR), stressed that the use of the hookah pipe is also increasing in the United States, and that 1 hour of using a hookah pipe can equate to inhaling 100 to 200 times the volume of smoke inhaled from a single cigarette (CDC, 2011b). Brawley agreed that the use of hookah is growing, especially on college campuses, and its use should be addressed. Pechacek confirmed that the prevalence of hookah smoking is growing rapidly, and public health officials need to pay attention to the problem. Several speakers emphasized the need to increase national surveillance for all forms of tobacco use, with emphasis on the need to assess the effects of combined tobacco product use. Warner added that the University of Michigan’s Monitoring the Future study, which traces use of drugs by children, is now tracking the use of hookah.
McAfee discussed the emergence of new noncombustible products, such as refrigerated snus and dissolvables in tablet or stick form that have been introduced in the United States during the past 5 years, as well as the availability of e-cigarettes. Some of these products are being marketed as
________________
3 National Survey on Drug Use and Health, 2010 (unpublished data).
providing “cleaner tobacco enjoyment,” or as products that can be used where smoking is not allowed or accepted. He noted that the Blu e-cigarette has an ad that states, “Why quit? Switch to Blu,” and another ad pictures an e-cigarette user showing a vulgar gesture with the words “Dear Smoking Ban,” suggesting that the use of e-cigarettes evades smoke-free policies.
Several speakers emphasized that the health effects of many new alternative tobacco products have not been tested or monitored, and thus are unknown. Although individual risk for the health consequences of tobacco use may be reduced if individuals replace combustible tobacco products with smokeless tobacco products, the potential for harm at the level of population health may be significant. “A product like snus or even an e-cigarette could potentially cause harm because they introduce kids to a tobacco product that they [might be able to] use in school and [may be more] socially acceptable, or encourage long-term dual use in established smokers rather than quitting or switching completely,” said McAfee. In addition, the presence of e-cigarettes, which resemble cigarettes, may renormalize smoking in public places, and have negative consequences for public health. McAfee stressed that the public health effects of noncombustible products such as snus, dissolvables, and e-cigarettes will depend not only on their physical characteristics, but also on how they are marketed by companies, regulated by the FDA, and used by consumers. He also mentioned the new product Verve, a nicotine disc manufactured by the smokeless tobacco company Altria. “If this was coming off a pharmaceutical company’s manufacturing line, it would have been considered a nicotine replacement product. But nobody knows exactly yet how it is going to be marketed or what its effect will be,” he said.
Linda Sarna, professor and Lulu Wolf Hassenplug Endowed Chair of the University of California, Los Angeles, School of Nursing, summarized the likely impact of these new products by stating, “The tobacco industry clearly has a business model in which someday the combusted cigarette is going [away], so they need to maximize their profit for as long as possible, while looking more like the good guys by offering ‘harm-reduced’ alternative bridge products. But they don’t tell the public that the more they use those noncombustible bridge products and buy their message, the longer they are also going to use some form of a combustible product. We have to be out there with a public education campaign that frames that debate differently.”
Participant Gruen Von Behrens reminded fellow participants that noncombustible tobacco products are not safe alternatives to smoking. As
a cancer survivor and former spit tobacco user, Von Behrens stressed that “there is no safe tobacco. No matter what we use, how it is ingested, it is going to hurt you. I am living and breathing proof of that.” He noted that state governments tend to focus on raising taxes on cigarettes, but not on spit tobacco. Benjamin Toll, assistant professor of psychiatry at the Yale University School of Medicine, member of Yale Cancer Center, and program director of the Smoking Cessation Service for Smilow Cancer Hospital at Yale-New Haven, concurred that much of the research is focused on smoking and how to curb it, and there is a need to study use of all tobacco products.
A portion of the workshop was devoted to the effects of tobacco smoke on cancer, including how it increases cancer risk and worsens cancer treatment outcomes.
Cigarette smoke contains more than 7,000 compounds, at least 60 of which are known carcinogens, said Pechacek and Graham Warren, a clinical radiation oncologist who treats cancer patients and director of the Tobacco Assessment and Cessation Program at Roswell Park Cancer Institute (HHS, 2010b). Approximately 600 compounds are added to tobacco to enhance flavor or nicotine absorption. Inhaling this mix of chemicals through smoke induces tissue injury and changes in the cellular environment that foster proliferation and transformation into cancer cells (HHS, 2004, 2010b).
Fadlo Khuri, professor and chair of hematology and medical oncology at Emory University, deputy director for the Winship Cancer Institute, and Roberto C. Goizueta Distinguished Chair in Cancer Research, showed the major pathways by which cigarette smoke causes cancer, including the ability of carcinogens in tobacco smoke to latch onto DNA, leading to mutations in critical genes. This results in a loss of normal growth control mechanisms, precancerous tumor growth, and the accumulation of additional mutations that cause those cells to become malignant (see Figure 3). Khuri said several environmental factors, including tobacco smoke, and specific genetic mutations are linked to carcinogenesis, from precancerous abnormal growth of cells to metastatic lung cancer (HHS, 2010b). He pointed out that compounds in tobacco smoke can also silence tumor sup-
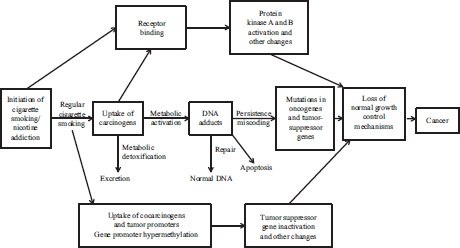
FIGURE 3 Link between cigarette smoking and cancer through carcinogens in tobacco smoke.
SOURCES: Khuri presentation (June 12, 2012) and HHS (2010b).
pressor genes that normally prevent tumor growth. Khuri added that there is a strong correlation with lifetime cigarette consumption and the genetic changes that are a signature for the loss of this critical tumor suppressor mechanism (Takeshima et al., 1993). “We now have the imprint of tobacco on DNA showing carcinogenesis,” he said.
Even after a cell becomes malignant, “tobacco is able to turn on a symphony of bad actors in a very organized manner,” Khuri said, including changes in the activity of many genes that are targeted by new cancer treatments. Mutations in these genes are known as driver mutations because tumor growth depends on them.
Nicotine and activation of systemic nicotinic acetylcholine receptors by various products in tobacco can trigger cell survival pathways that prevent the death of mutated cells. Warren noted several studies demonstrating that nicotine can increase cancer proliferation, angiogenesis, migration, and invasion, and decrease the effectiveness of conventional cancer treatments such as chemotherapy and/or radiotherapy (Warren et al., 2008, 2012c).
Khuri also stressed that “tobacco-related carcinogenesis is something of a race because you have multiple tumors emerging so even if you treat one, another will emerge to take its place. Continuation of smoking enhances the likelihood of second primary tumors.” Such tumors are likely to emerge in
part because of the overexpression of some genes, such as cyclooxygenase-2, that don’t directly cause tumors, but regulate other genes that do, Khuri added.
Khuri noted that the 2010 Surgeon General’s report “put to bed any lingering doubt as to whether tobacco had left its fingerprints all over these diseases” (HHS, 2010b). This report also concluded that tobacco cessation is the only proven strategy for reducing the pathogenic processes leading to cancer. There is insufficient evidence that modifying tobacco products can reduce cancer risk, the report clearly stated.
Impact of Smoking on Cancer Incidence and Treatment Outcomes
The impact of smoking on cancer is substantial. Smoking accounts for at least 30 percent of all cancer deaths and 80 percent of lung cancer deaths (ACS, 2012). Lung cancer, for which smoking is the primary risk factor, is the leading cause of cancer death in both men and women (ACS, 2012). However, smoking heightens the risk of up to 18 types of cancers, including head and neck cancers, leukemia, and cancers of the esophagus, bladder, pancreas, kidney, liver, stomach, colorectum, cervix, uterus, and ovaries (ACS, 2012; HHS, 2004; Secretan et al., 2009).
Smoking not only raises the risk of developing various cancers, but it worsens cancer outcomes (NCI, 2012b). Worse survival among patients who smoked or continue to smoke is seen not only among patients with cancers strongly linked to smoking (lung, esophageal, or head and neck), but also in patients with breast, prostate, and other cancers (Warren et al., 2012a). Warren highlighted more than 100 studies demonstrating that cancer patients who are or were smokers were also more likely to have decreased therapeutic responses, increased cancer recurrences, and increased cancer treatment complications, including problems with wound healing, infections, cardiovascular complications, and the development of a secondary malignancy.
The risk of developing secondary malignancies in cancer patients who smoke is very high, Warren said. He noted studies demonstrating that smoking substantially increased the risk of developing lung cancer in patients with breast cancer or Hodgkin’s disease treated with radiotherapy and/or chemotherapy (Kaufman et al., 2008; Lorigan et al., 2005; Travis et al., 2002). In addition, smoking substantially increased the risk of bladder cancer in patients treated with radiotherapy (Boorjian et al., 2007). The increased risk of developing a second malignancy provides strong support
for the synergistic tumorigenic activities of tobacco with conventional cancer treatments.
Warren pointed out that the difference in survival outcomes between patients who do not smoke and those who do can be much greater than the difference in survival associated with various treatment regimens. For example, there is about a 15 percent improvement in survival in breast cancer patients who are nonsmokers as compared with those who smoke (Warren et al., 2012a), but there is a less than 4 percent difference in survival between breast cancer patients who are treated with cyclophosphamide and doxorubicin versus cyclophosphamide, methotrexate, and fluorouracil chemotherapies (Breast Cancer Trialists Group, 1998). This 4 percent improvement led to a change in treatment guidelines for breast cancer. Unfortunately, current nationally funded actively accruing cooperative group clinical trials do not capture information on tobacco use or cessation. A new study demonstrates that only 29.4 percent of cooperative group trials assess any form of tobacco use at study entry, only 4.5 percent assess tobacco at follow up, and no trials offer cessation support (Peters et al., 2012). “We are changing our standard [of care] recommendations for a treatment modality for breast cancer, yet we are ignoring tobacco,” Warren said.
Warren, Cummings, Toll, and others noted that one significant limitation to accurately understanding the effects of tobacco on cancer treatment is the lack of well-defined tobacco assessments in clinical practice. In order to fully understand the effects of tobacco on cancer treatment, we must ask about former and current tobacco use prior to diagnosis, during treatment, and during follow-up. In addition, mandatory cessation efforts should be implemented and tracked to understand the effects of cessation as well as to identify risks of continued tobacco use in cancer patients. Warren further discussed biologic models showing that exposure to tobacco products [nicotine] specifically during the time of cancer treatment is the primary determinant of changes in therapeutic response (Warren et al., 2012c). “This is the reason that it is critically important for cancer patients to stop using tobacco as soon as possible to get the most benefit from cancer treatment,” Warren said.
Warren stressed that encouraging cancer patients to quit smoking may have a marked effect on their survival, as some studies have indicated for head, neck, and lung cancers (Browman et al., 1993; Herbst et al., 2005; Thatcher et al., 2005). “If you have a current smoker, you might still be able to help them,” he said. Data demonstrate that having quit tobacco use within the past year results in a significant improvement in survival for
head and neck as well as lung cancer, suggesting that some of the effects of tobacco on survival may be reversible (Warren et al., 2012a). Pechacek pointed out that inhaled smoke is particularly damaging to health and, as Warren noted, inhaling even secondhand smoke can increase the risk of lung cancer, as was seen in nearly 37 studies of spouses of people who smoke (Hackshaw et al., 1997). Pechacek stressed that the duration of smoking is more important for cancer risk than level of exposure (Flanders et al., 2003). He added that cancer risk increases much more for each additional year of smoking than it does for higher average number of cigarettes smoked.
Pechacek also pointed out that among people who currently smoke, about 70 percent smoke 10 to 30 cigarettes per day (CDC, 2011d), which makes them well within the intensity levels needed to boost their cancer risk. He added that “people are far from understanding that infrequent, but still regular, exposure, is very risky to health, particularly to cardiovascular health. Over half of tobacco-related health effects are likely to be maintained at low levels of cigarette use,” Pechacek said. In light of these risks, Abrams expressed concern that noncombustible tobacco products merely lower the number of cigarettes smoked and deter people from quitting smoking completely. “Substantial population risks may be associated with any product that delays complete cessation among people who smoke,” Pechacek added.
What makes tobacco use especially difficult to combat is its addictive nature due to nicotine and other compounds and additives that tobacco products contain. “Nicotine addiction is not just a habit,” said Cheryl Healton, founding president and chief executive officer of Legacy. “It is an extremely uncomfortable addiction for most people, which makes it very difficult to quit.”
Several speakers noted that tobacco companies specifically add compounds to their tobacco products to enhance nicotine absorption and make their products more addictive. Hurt said that tobacco companies add ammonia to tobacco to free-base the nicotine, resulting in high doses reaching the brain quickly (Hurt and Robertson, 1998). He said nicotine from smoking cigarettes reaches the brain faster than if it were injected intravenously.
Once in the brain, nicotine increases the number of the brain’s nico-
tine receptors, probably for months, according to Hurt. “If you don’t feed these receptors what they want, then you have severe withdrawal. Plus these receptors have a memory, so even though a person may have stopped smoking for many months and the receptors are downregulated, if they are in a situation which would prompt the trigger to smoke, the cravings can come back,” he said. As Healton succinctly put it, nicotine “hijacks the brain.” Hurt also stressed that positron emission tomography and magnetic resonance imaging (MRI) scans of the brain showed that smoking one cigarette resulted in more than 88 percent receptor occupancy, while smoking three cigarettes during a scanning session could fully saturate the brain with nicotine (Brody et al., 2006). The half-life of nicotine is only 120 minutes.
Because nicotine is so addictive, many people who smoke cigarettes have to make numerous attempts at quitting before they are successful, and overall quitting success is minimal. According to Warner, surveys have found that 70 percent of people who smoke want to quit and about 50 percent make a sincere effort to quit each year. However, only about 6 percent of individuals successfully quit in the past year (CDC, 2011c). Cummings noted that that number of people who make quit attempts each year may be much higher; many try to quit but never reach the 24-hour mark reflected in the estimate (CDC, 2011c). “The problem isn’t the motivation to quit—it’s the ability to sustain quitting,” he said.
Given the addictive nature of nicotine and the difficulties individuals face in trying to quit, several speakers discussed long-term treatment with nicotine as a potential therapeutic strategy. Abrams noted that nicotine use by itself may not be as hazardous to health as tobacco use, and that nicotine treatment might be a way to avoid the health problems associated with tobacco products. “Nicotine has a bad reputation because it is in bed with burnt tobacco in a product that is carcinogenic, but nicotine can be a useful drug for society if it is properly framed, administered in a safe form, and perhaps even marketed,” Abrams said. He stressed that the brains of people addicted to nicotine may need the compound to function properly just like people with a chronic mental illness like schizophrenia need their medications to function properly. “You wouldn’t say to someone with schizophrenia ‘you can’t take your meds for more than three months because we don’t want you to get addicted to them,’” Abrams said.
Warner noted that the tobacco company Philip Morris purchased rights to a patent for a nicotine inhaler that might be an alternative to tobacco products for those already addicted to nicotine (Felberbaum, 2011). Warner further noted that pharmaceutical companies may be reluctant to pursue
alternative tobacco products because they do not want to sell a product that would be addictive for life. “But some of us take blood pressure and cholesterol medications every day to save our lives and it is not clear that this is fundamentally different,” Warner said. Hurt noted that a metered-dose nicotine inhaler was developed by 3M about 15 years ago and produces high spikes of nicotine akin to what cigarettes provide. When 3M presented the inhaler to FDA, the agency declined to consider approving it because it would be an addictive product. This FDA stance has discouraged pharmaceutical companies from developing such a product, he said. Carolyn Dresler, medical director for the Tobacco Prevention and Cessation Program at the Arkansas Department of Health, added that GlaxoSmithKline explored whether to pursue developing a nicotine inhaler and declined to do so mainly because it would not be marketable nor much of a profit maker because it would have to be prescribed by a physician.
Hurt and others stressed that health practitioners need to treat nicotine addiction as a serious medical problem. Fiore noted that the recently updated U.S. Public Health Service (PHS) clinical practice guidelines released in 2008 (Fiore et al., 2008) for the first time referred to tobacco dependence as a chronic disease that warrants treatment. Hurt further emphasized that treatment and cessation support should be repeated at every visit, including counseling and pharmacotherapy when appropriate. He also said that insurers need to recognize tobacco use as a chronic health condition and that cessation is a cost-effective treatment. Several speakers pointed out that given the highly addictive nature of nicotine, people who smoke should not be looked down upon or blamed. “The patient [who smokes] is not to be criticized or treated with condescension, but treated the same way we treat people with other ailments,” Brawley said, adding he is concerned about a movement under way to raise insurance rates for people who smoke. “We need to get treatment to those people who have an illness as opposed to penalizing them for having an illness,” he said. Dresler concurred, adding, “We should be careful not to blame people who smoke for the disease they have, because it is an industry-caused disease and it is the industry we should be focusing on.”
Ellen R. Gritz, professor and chair of the department of behavioral science and Olla S. Stribling Distinguished Chair for Cancer Research at the University of Texas MD Anderson Cancer Center, noted that surveys find that cancer patients, especially lung cancer patients, who have a history of smoking have increased levels of guilt and shame compared those who did not smoke. Bloch added that a 2008 Gallup poll found that 66 percent of
people agreed that people who smoke are completely or mostly to blame for the health problems they face, and only 24 percent blamed tobacco companies (Gallup, 2008). “I think the vast majority of Americans, including health professionals, think that smoking is a personal weakness,” she said. Gritz added that when she talks to such patients she tries to turn their burden of guilt and shame into anger at the tobacco industry for facilitating tobacco addictions.
Bloch further elaborated on the of role tobacco companies in the current epidemic of smoking-related cancers. Bloch noted that the 2006–2007 annual report of the President’s Cancer Panel described the tobacco industry as a vector in the development of cancer, akin to mosquitoes in the transmission of malaria (NCI, 2007). She also pointed out that in 2006, following a 9-month trial, U.S. district judge Gladys Kessler found that the major U.S. cigarette manufacturers engaged in racketeering, fraud, and conspiracy. Judge Kessler’s final opinion noted that the cigarette industry “survives, and profits, from selling a highly addictive product which causes diseases that lead to a staggering number of deaths per year, an immeasurable amount of human suffering and economic loss, and a profound burden on our national health care system.” Her ruling further stated that “defendants have marketed and sold their lethal product with zeal, with deception, with a single-minded focus on their financial success, and without regard for the human tragedy or social cost that success exacted.”4
Judge Kessler found that the cigarette companies falsely denied, distorted, and minimized the significant adverse health consequences of smoking for decades, including smoking’s causal link to cancer; falsely denied that they control the level of nicotine delivered in cigarettes in order to create and sustain addiction; and falsely marketed and promoted low-tar cigarettes as less harmful. In addition, from the 1950s to the present, tobacco companies intentionally marketed to young people to recruit what they termed “replacement smokers” to ensure the companies’ economic future. They also publicly denied that secondhand smoke is hazardous to
________________
4 United States of America, Plaintiff, and Tobacco-Free Kids Action Fund, American Cancer Society, American Heart Association, American Lung Association, Americans for Nonsmoker Rights, and National African American Tobacco Prevention Network, Intervenors, v. Philip Morris USA, Inc. (f/k/a Philip Morris, Inc.), et al., Defendants. United States District Court for the District of Columbia Civil Action No. 99-2496 (GK). Final Opinion (2006).
nonsmokers, despite internally acknowledging this fact (Tobacco Control Legal Consortium, 2006).
A substantial portion of the workshop was devoted to tobacco cessation programs, including how to boost referrals to such programs, the role of counseling and pharmacotherapy, the need for relapse prevention, pharmacotherapy options and challenges, and targeting tobacco cessation programs to cancer patients.
Overview of Tobacco Cessation Therapy
The recent tobacco cessation treatment guideline notes that tobacco dependence treatments are effective across a wide range of populations (Fiore et al., 2008). “We have an extraordinary evidence base to treat tobacco dependence,” Fiore said, noting that the current guideline was based on nearly 9,000 studies and about 35 meta-analyses. In addition, an independent review of the meta-analysis performed in the 2008 PHS guideline and analysis of Cochrane reviews on treating tobacco dependence found a high rate of concordance on the evidence for efficacy of tobacco cessation therapy (Hughes, 2009). Unfortunately, Fiore also noted that tobacco use and cessation are linked by an interesting confluence of situations: a large health threat, effective treatment, and a disinclination for clinicians to intervene consistently.
The PHS guideline found a powerful dose-response effect; spending more time counseling patients led to higher quit rates. But even group counseling or telephone-based counseling, including quitlines, are effective interventions, as well as brief interventions as short as 3 minutes. (See Box 1 for more information on quitlines.) Practical counseling, such as problem-solving and skills training, as well as social support delivered during treatment, were found to be especially effective, the guideline notes (Fiore et al., 2008). However, Fiore added that “there is not going to be a one-size-fits-all treatment for tobacco cessation. What works best is a menu of options that are available without barriers for people who want to quit.” That menu of options is large, and includes seven firstline medications that help stem nicotine cravings (see Table 1). Although counseling or medications are each effective singly, combining the two is especially effective, Fiore stressed. “There’s an additive effect of combining counseling and medica-
A quitline is a telephone counseling service for individuals who are trying to quit tobacco use (Fiore et al., 2008). Quitline services can include coaching and counseling, referrals, mailed materials, training to health care providers, Web-based services, and in some circumstances, free medicines, such as nicotine replacement therapy (NAQC, 2012). The 2008 clinical practice guideline concluded that “telephone quitline counseling is effective with diverse populations and has broad reach. Therefore, both clinicians and health care delivery systems should ensure patient access to quitlines and promote quitline use” (Fiore et al., 2008).
In the United States, individuals looking for tobacco cessation counseling can call 1-800-QUIT-NOW or visit www.smokefree.gov. This national network of state quitlines is supported by a combination of funding from states, CDC, and NCI. Although quitlines are available in every state, Linda Sarna, professor and Lulu Hassenplug Endowed Chair of the University of California, Los Angeles, School of Nursing, and Colleen Stevens, branch chief of the California Department of Public Health’s Tobacco Control Program, emphasized that quitlines are an underutilized resource, and there is often a lack of awareness about their availability. Carolyn Dresler, medical director for the Tobacco Prevention and
TABLE 1 Tailoring Pharmacotherapy for Tobacco Cessation Therapy
| Long-Acting Options | Short-Acting Options |
| Pick 1 or 2: | Add 1 or 2 from here: |
| Nicotine patch | Nicotine gum |
| Bupropion | Nicotine inhaler |
| Varenicline | Nicotine lozenge |
| Nicotine nasal spray | |
SOURCE: Hurt presentation (June 11, 2012).
tion that should be the basis of an intervention for every patient, absent contraindications,” he said.
Fiore noted that although 25 to 35 percent of smokers who visit primary care settings are willing to make a quit attempt, research shows that
Cessation Program at the Arkansas Department of Health, encouraged clinicians to call a quitline in order to have a better understanding of the services that a quitline provides.
Several speakers discussed some innovative approaches using quitlines to improve quitting success. According to Thomas Land, director of the Office of Statistics and Evaluation for the Bureau of Community Health and Prevention at the Massachusetts Department of Public Health, an integrated electronic medical record (EMR) system can send electronic referrals to the state’s quitline, and then receive information on a patient’s progress from the quitline (see the section on Successful State and Local Efforts for more information). Stevens noted that a federal grant supporting California’s quitline is assessing whether incentivizing individuals to quit will improve quitting success, and is also attempting to remove barriers to quitting by sending tobacco cessation medicines directly to an individual’s home.
Quitlines are a major component of the CDC’s media campaign, Tips from Former Smokers (see Box 3). “Behind the scenes of the media campaign, the states played a very important role—the 1-800-QUIT-NOW action line,” said Howard Koh, assistant secretary for health at the U.S. Department of Health and Human Services. “Much of the funding to support [the national network of state quitlines] comes from state funding, and that has really been the safety net that tobacco users have gone to during the 12 weeks of this campaign. It is very important that that capacity be continued,” he said.
number can be increased to as much as 50 to 70 percent, if they are asked if they are willing to try cutting down on how much they smoke on the way to quitting, or if they are willing to try a nicotine replacement product. There is evidence that such prequit treatment can be effective, especially if nicotine replacement treatment is given for periods of time up to 6 months, and if it is linked with a recommendation to reduce the number of cigarettes smoked per day (Fiore and Baker, 2011). Fiore added that evidence also shows that motivational interviewing practices can also boost the likelihood of a quit attempt by a patient. “There are simple strategies that allow us to counsel our patients who are not yet ready to quit,” Fiore said. Although the guideline recommended counseling for users of smokeless tobacco, it did not recommend pharmacotherapy for these tobacco users because there was not yet a sufficient evidence base for that recommendation, Fiore reported.
Given that tobacco cessation therapy is effective, and even highly
TABLE 2 The 5 A’s Model for Treating Tobacco Dependence
| Ask about tobacco use | Identify and document tobacco use status for every patient at every visit. |
| Advise to quit | In a clear, strong, and personalized manner, urge every tobacco user to quit. |
| Assess willingness to make a quit attempt | Is the tobacco user willing to make a quit attempt this time? |
| Assist in quit attempt | For the patient willing to make a quit attempt, offer medication and provide or refer for counseling or additional treatment to help the patient quit. For patients unwilling to quit at the time, provide interventions designed to increase future quit attempts. |
| Arrange follow-up | For the patient willing to make a quit attempt, arrange for follow-up contacts, beginning with the first week after the quit date. For patients unwilling to make a quit attempt at the time, address tobacco dependence and willingness to quit at next clinic visit. |
SOURCES: Fiore presentation (June 11, 2012) and Fiore et al. (2008).
cost-effective, the guideline recommends that insurance plans include the counseling and medication identified as effective in the guideline as covered benefits. “We need to pay our clinicians to deliver smoking cessation treatments just as we pay them to deliver other treatments, and we need to expand our insurance packages to include evidence-based treatments for tobacco dependence in the basic benefits package for all insurance plans,” Fiore said. This strategy was advocated by several other speakers during the workshop, and several speakers noted that the Patient Protection and Affordable Care Act (ACA) will help expand coverage of tobacco cessation therapy (see section on Antismoking Laws and Regulations).
The tobacco cessation treatment guideline recommends that every health practitioner use the “5 A’s intervention” model for their patients, including asking patients about tobacco use, advising them to quit, assessing their willingness to quit, assisting them in their effort to quit, and arranging
for follow-up (see Table 2). The model also specifies offering motivational interviewing if the patient is not willing to quit.
Fiore views the guideline as aiming “to change the architecture of a clinical encounter from one in which we ignore smoking with impunity, to one in which there is no possibility for a person who smokes to visit that clinic without his or her smoking status being identified and a specific plan in place to assist his or her efforts to quit.”
Hurt stressed integrating behavioral treatment, addiction treatment, pharmacotherapy, and relapse prevention with motivational interviewing. The Mayo Clinic, he reported, uses higher nicotine patch doses for heavy smokers and combination pharmacotherapy as the rule rather the exception, especially combinations of long-acting medications with short-acting medications to treat nicotine withdrawal. They also make sure to ask their patients open-ended questions, such as “How are you doing?” to assess if the treatment provided is sufficient. If the patient is still having substantial nicotine cravings, another medication may be given or the dosage of a medication may be increased. To assess the proper dosage, Mayo Clinic also routinely measures patients’ serum cotinine5 levels, with levels over 300 ng/ml indicating a heavy nicotine exposure that may warrant two 21-mg nicotine patches, for example.
Abrams noted that the pharmacotherapy for tobacco cessation should be akin to the step-up care approach taken to treat asthma, with higher doses, multiple medications, or different medications given until the patient is comfortable, and that physicians avoid the same treatment that has failed in the past. He suggested modifying the tobacco dependence treatment guideline so it has more of a step-up sequential approach that indicates the next option if the previous treatment option fails. He noted that “we don’t have research [results] to inform this type of approach,” but he added that failure to treat people who are severely addicted with high enough doses or sufficient medicine combinations initially will lead them to think it is not possible for them to quit smoking and deter further quit attempts.
Hurt emphasized that the longer the treatment, the better the results will be. The length of treatment is determined by how long it takes patients to be comfortable without smoking. Once a sufficient comfort level is achieved, medication levels are tapered off.
Hurt also stressed that “patient involvement is absolutely critical,”
________________
5 Cotinine, a metabolite of nicotine, is measured in blood, saliva, or urine to assess nicotine exposure.
because almost all tobacco users have had some experience trying to quit smoking. “You can use that experience to really figure out what might be the best treatment for this person at this particular time,” he said. Hurt added that patient preferences for specific treatments should be honored, unless there is a contraindication, because it will facilitate patient compliance, which is often a challenge. “We have more trouble keeping people on medications long enough than we do [with] any other factor,” he said. Sarna added that electronic medical records (EMRs) could help prompt physicians to determine the best drug combinations and doses to use to support an individual’s quit attempt.
Use of Tobacco Cessation Therapy
Fiore pointed that even though 80 percent of all people who smoke see a clinician each year, and 70 percent of smokers report that they want to quit, only about 32 percent attempted to do so using evidence-based counseling and/or medication (CDC, 2011c). Geoffrey Fong, professor of psychology and public health and health systems at the University of Waterloo and senior investigator at the Ontario Institute for Cancer Research, added that the number of smokers referred to a cessation program is even smaller, and that many physicians do not assess a patient’s smoking status. He suggested mandating smoking assessments and referrals to cessation programs for Medicaid recipients or as a national policy consideration for reimbursement in health care. “If we refer all people who smoke to cessation programs, we will have a greater impact on reducing smoking,” he said.
Warren added that “we are not getting enough referrals from our physicians,” noting several studies that documented this finding. Some found that fewer than half of all community physicians refer people who smoke to cessation programs, and many physicians who conduct cancer-related clinical trials also fail to refer (Fiore et al., 2008; Peters et al., 2012; Warren et al., 2012b). Fiore noted, “I would never let a patient with high blood sugar leave my clinic without addressing diabetes, irrespective of what brought her to the clinic that day. Similarly, if I have a patient with elevated blood pressure, it would be malpractice to let that patient leave the office without addressing [it]. Yet people come in and out of clinics across America today without tobacco use being addressed,” he said.
Lawrence Deyton, director of FDA’s Center for Tobacco Products, added that when he worked as a physician at the VA, the EMR there would remind him to make a referral to a tobacco cessation program once he noted
in the medical record that a patient smoked. “I suggest looking at the VA system, learn from it what worked and didn’t work, and try to replicate its best practices into a referral strategy,” he said.
Fiore concurred, pointing out that the PHS 2008 clinical practice guideline on treating tobacco use and dependence stressed the critical importance of identifying and intervening with every person who smokes at every health visit (Fiore et al., 2008). “Smoking status as a vital sign was recommended about 20 years ago, but with the advent of the EMR, we have electronic systems that allow this to happen even more systematically. We need to hold accountable our health systems that aren’t effectively doing this because everything follows from that identification,” he said. But Sarna noted that not all EMRs have the appropriate fields to assess proper adherence to the PHS tobacco dependence treatment guideline (Conroy et al., 2005). “The EMR provides a lot of opportunities, but it is not perfect. We have to look carefully at the fields that are available,” she said. McAfee added that “changing health care systems and individual provider behavior is at least as challenging as getting people to quit smoking.”
Models for Clinician Referrals
The need to involve more clinicians in assessing and treating their patients’ tobacco use was stressed by several speakers. Not only do clinicians make a difference, Sarna pointed out, but the number of clinicians involved in quit attempts makes a difference in how likely patients are to stop smoking (see Figure 4). “It doesn’t have to be the physician who is involved—it can be the nurse, respiratory therapist, psychologists, social workers, etc. The more [types of] clinicians involved, the better,” she said.
Sarna said that compared to smokers who receive usual care, those who receive assistance from a nurse have a 28 percent greater probability of successfully quitting for at least 5 months (Rice and Stead, 2008). She added that helping patients with tobacco cessation is within a nurse’s scope of practice, and that nurses are the largest health care workforce so they should be leveraged more in tobacco cessation efforts. Hurt noted that at Mayo Clinic, a tobacco use intervention protocol empowers nurses to provide a brief intervention for every smoker identified, and also provides referrals to tobacco treatment specialists and nicotine replacement therapy for every individual who wants it. However, such a model seems to be the exception rather than the rule, Sarna pointed out. Studies show a lack of referral of patients to smoking cessation programs by both nurses and
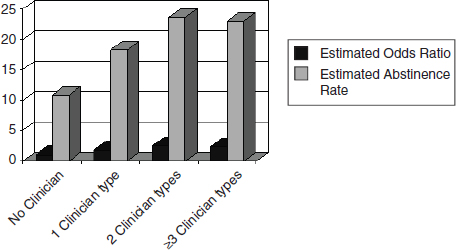
FIGURE 4 Effectiveness of and estimated abstinence rates for interventions delivered by various numbers of clinician types. Individuals are more likely to stop smoking if more clinicians are involved in a quit attempt.
NOTE: Abstinence rate = percentage of people who achieve long-term abstinence from smoking.
SOURCES: Sarna presentation (June 11, 2012) and Fiore et al. (2008).
physicians (Sarna et al., 2009, 2012; Tong et al., 2010). “Clinicians are not doing enough, rapidly enough and we need to change that,” she concluded.
Warren suggested institutions mandate assessment of patient smoking status and referral to a cessation program. This was done successfully at his institution (Roswell Park Cancer Institute) and patients were highly receptive to interventions offered by trained cessation personnel, he said. Others have explored whether lung cancer screening prompts more people to seek smoking cessation therapy. Low-dose computed tomography (CT) scanning of the lungs can detect lung tumors at an early stage, and Ostroff reported that most people enrolling in screening programs do appear to be motivated to quit and have higher quit rates than generally observed in studies of healthy or asymptomatic smokers (Anderson et al., 2009; Hahn et al., 2006; Ostroff et al., 2001). But she added that the results are inconclusive as to whether the screening results (positive or negative findings) impact actual cessation outcomes, with a number of conflicting or statistically insignificant findings reported in the literature. “More work is needed on this,” Ostroff said, adding that one advantage of lung cancer screening is that it offers the opportunity not only for a patient-focused intervention,
but also a provider-delivered intervention because the results are sent back to the primary care physician who orders the screening.
Abrams added that research he is involved in suggests that the cost effectiveness of lung cancer screening improves with the inclusion of aggressive smoking cessation therapy, and that such treatment should be mandated in the care model for lung cancer screening.6 “It’s a missed opportunity to let anyone through repeated [lung cancer] screening without aggressive cessation treatment,” he said. Hurt added that several screening facilities conduct ultrafast CT scans of the lungs inexpensively, yet have no intention of providing any cessation therapy to those being screened. Several speakers criticized CT screening protocols that inadequately address cessation.
Gritz emphasized that at MD Anderson Cancer Center, patients who are directly referred to tobacco cessation therapy by their health care provider are more likely to attend a visit than those proactively identified through the EMR. However, the volume of referrals through EMRs are substantially greater overall, resulting in a higher absolute level of attendees via proactive EMR identification and referral. Sarna suggested that financial incentives may help reward practitioners, including nurses, for providing care consistent with clinical guidelines, including documenting smoking status and increasing referrals to tobacco cessation therapy.
Tobacco Cessation Therapy for Cancer Patients
Warren stressed that because tobacco use increases the relapse and treatment complication rate and decreases survival for a number of cancers, tobacco cessation therapy for cancer patients could have a major impact. “This is a single treatment that could potentially benefit 500,000 cancer patients a year and it is something cancer patients can do themselves to improve their outcomes. I would challenge anyone else to find a therapeutic modality that benefits 500,000 cancer patients a year,” he said.
Gritz noted that a sizable portion of cancer patients continue to smoke, even if diagnosed with a smoking-related cancer. One study at MD Anderson found that 37 percent of patients with early-stage non-small-cell lung cancer were smoking 1 year after diagnosis, and overall, up to 30 to 50 percent of cancer patients who are smoking at diagnosis do not quit or relapse following initial attempts to quit (Cooley et al., 2009; Gritz et al.,
________________
6 Abrams noted that this research has been submitted for publication and is currently under review.
2011; Tseng et al., 2012; Walker et al., 2006). Gritz noted that smoking also continues to be prevalent in those diagnosed with other smoking-related diseases, such as emphysema and chronic bronchitis (CDC, 2007a).
Gritz suggested tailoring tobacco cessation therapy to cancer patients by educating them about the links between cancer outcomes and smoking, by being sensitive to certain physical limitations imposed by the disease, the treatment, and medical contraindications. She also suggested being sensitive to the psychological issues cancer patients have related to their smoking, including guilt, depression, anxiety, and stressors related to cancer itself as well as to smoking behavior.
Gritz added that cessation interventions that are initiated closer to a cancer diagnosis tend to be more successful. One study found that when smoking, depression, and alcohol use were treated in head and neck cancer patients, there was a higher 6-month quit rate than among those treated with standard care (Duffy et al., 2006). She also stressed that once cancer patients quit smoking, it is a good opportunity for clinicians to reinforce the message to abstain from tobacco use.
Gritz pointed out that the negative stigma of smoking was stronger for lung cancer patients than those with other cancers. Even lung cancer patients who never smoked felt blamed for their illness, and this stigma had an effect on relationships with the patients’ families, friends, and physicians, even when patients had stopped smoking years ago (Chapple et al., 2004).
Khuri pointed out that practitioners are often reluctant to treat cancer patients for tobacco dependence if they are actively undergoing chemotherapy because they are concerned that such treatment will interfere with their cancer therapy. But Hurt countered that he has no reluctance in using any of the medications that relieve nicotine cravings, especially for the nicotine replacement products because they are already getting the equivalent nicotine by smoking. He added that bupropion has been used for decades in cancer patients and that varenicline is not likely to interfere because it is a highly focused treatment. “If you can help [cancer patients] stop smoking, your cancer treatments are going to work better and there will be fewer side effects from them. I think it is incumbent upon every oncologist to help patients stop smoking so as to get better benefits from the treatments you are giving them,” Hurt said.
Even though many cancers are caused by smoking, oncologists have one of the lowest rates of addressing tobacco dependence and offering their patients cessation therapy, according to Fiore. A recent survey of NCI-designated cancer centers found that only 62 percent of centers reported
routinely providing tobacco education materials to patients, while just more than half of cancer centers reported effective assessment of patient tobacco use (Goldstein et al., 2013). Twenty percent of cancer centers reported that they had no tobacco cessation services, and less than half of cancer centers had personnel designated for tobacco cessation therapy (Goldstein et al., 2013). Given the lack of focus on tobacco assessment and cessation therapy at cancer centers, Gritz emphasized that “we need more resources and top-down leadership and commitment, and we need funding and personnel.”
There are no standards for cancer centers requiring tobacco cessation programs, Cummings pointed out (Morgan et al., 2011). He suggested that NCI and accrediting organizations should make the presence of an active tobacco use treatment program for cancer patients a standard of care, with quality indicators to document delivery of such services to cancer patients. He also recommended that NCI and accrediting organizations require designated cancer centers to implement interventions to reduce tobacco use in their catchment areas, with quality indicators to demonstrate that these efforts are lowering tobacco use rates. Sarna noted that the Joint Commission, an accrediting body for more than 19,000 health care organizations and programs, recently released voluntary measures to assess tobacco cessation therapy performance (Fiore et al., 2012). “In our national cancer centers, the lack of expectations for tobacco dependence treatment after cancer diagnosis and treatment as part of quality care has got to change,” concurred Sarna, adding that both the American Society of Clinical Oncology (ASCO) and the Oncology Nursing Society have policy statements that reaffirm the role of clinicians who treat cancer patients in addressing tobacco dependence (ASCO, 2003; Oncology Nursing Society, 2008). Several speakers noted the increasing advocacy by national cancer organizations such as ASCO, AACR, and NCI, but substantial dedicated efforts will be required to address the burden of tobacco use in cancer patients.
Several speakers noted that health practitioners need to become more educated about tobacco dependence and its treatment. Sarna said that many health practitioners are still unaware of the tobacco dependence clinical practice guidelines and the availability of quitlines. “[Practitioners] also have inappropriate attitudes and beliefs about the patient’s willingness to quit and their obligation to perform the smoking cessation intervention. There are a lot of myths and misperceptions about tobacco dependence,” she said.
Many nurses are reluctant to discuss tobacco cessation with their patients, Sarna pointed out, because they are concerned about adding guilt and stress to already vulnerable patients. “They don’t want them to suffer, not understanding the power of tobacco dependence and suffering,” Sarna said.
She noted that surveys assessing prelicensure education in both medical and nursing schools indicated that there is good coverage of the health risks of tobacco use, but much less on the health benefits of quitting or tobacco dependence assessment and treatment (Ferry et al., 1999; Wewers et al., 2004). “Health care professional schools in the United States should not get accreditation unless they cover tobacco dependence as a leading cause of preventable death,” she said. Sarna also suggested that credentialing of medical institutions include a requirement for adequate tobacco dependence treatment.
She added that there are abundant educational resources on treating tobacco dependence; in addition to the PHS tobacco cessation guideline (Fiore et al., 2008), ASCO is planning a tobacco cessation guide for oncology clinicians.7 The Rx for Change curriculum,8 hosted by the University of California, San Francisco, School of Pharmacy, contains a module on tobacco cessation therapy tailored to patients with cancer. In addition, there are online resources to support clinician interventions, including Helping Smokers Quit: A Guide for Clinicians (HHS, 2008). Additional resources can be found on the websites for the Smoking Cessation Leadership Center, the Agency for Healthcare Research and Quality, NCI, and the American Cancer Society.9 “There should be no more excuses that we don’t have knowledge and skills,” she said.
Hurt noted that the Mayo Clinic trains practitioners on tobacco dependence treatment, as do many other institutions, including the University of Massachusetts, and Ohio State University. “There are many places that are training tobacco treatment specialists who are beginning to populate medical centers around the country,” he said. Hurt pointed out that the state of New York is innovative in requiring substance abuse treatment programs to treat tobacco use and dependence and maintain tobacco-free environments
________________
7 See http://chicago2012.asco.org/ASCODailyNews/Tobacco.aspx (accessed December 10, 2012).
8 See http://rxforchange.ucsf.edu (accessed December 10, 2012).
9 See http://smokingcessationleadership.ucsf.edu (accessed December 10, 2012); http://www.ahrq.gov/path/tobacco.htm (accessed December 10, 2012); http://www.smokefree.gov (accessed December 16, 2012); and http://www.cancer.org/healthy/stayawayfromtobacco/index (accessed December 10, 2012).
(New York State Office of Alcoholism and Substance Abuse Services, 2008; New York State Tobacco Control Program, 2010).
OVERVIEW OF TOBACCO CONTROL POLICY
The need for strong government tobacco control policy and advocacy was stressed at the workshop by several speakers. “Compared to corporate or institutional policy, government policy affects the most people. It is permanent and it changes social norms,” said Russ Sciandra, director of advocacy for the American Cancer Society in New York State. Colleen Stevens, branch chief of the California Department of Public Health’s Tobacco Control Program, agreed that policy is an integral part of social norm change. “It reinforces behavior change and permanently institutionalizes change so you don’t have to keep going back and reinventing the wheel.” Stevens noted that when policy makers in California originally committed to reducing tobacco use, there was not a large evidence base for changing social norms. However, the states, counties, and cities that had very low smoking rates all had a social norm that reinforced no smoking. There is now a strong evidence base showing that statewide efforts can reduce tobacco use and tobacco-related disease. These data can be used to help facilitate future efforts to change social norms.
Sciandra stressed that although government employees are not allowed to lobby, they can still educate about the benefits of policy change. Stevens agreed. “We are a government organization so we never advocated for specific policies. Instead, we educated California, both smokers and nonsmokers, about the impact of secondhand smoke and the tobacco industry’s role in promoting initiation and discouraging cessation all with the goal of denormalizing tobacco use.” For example, her state office developed ads that detailed the harms of secondhand smoke in multiunit housing. “By educating residents on how smoke drifts between apartments, we built the norm or expectation that you wouldn’t want to live in a place where smoking could be drifting from one place to another,” said Stevens.
Tobacco control policy options outlined by Warner include informing and educating the public and health practitioners; instituting laws, regulations, taxes, and other disincentives; and promoting or supporting tobacco cessation programs. Tobacco control policy is interactive at both the federal and state levels, as well as more locally, and includes planning, infrastructure, and financial resources.
Howard Koh, assistant secretary of HHS, discussed the national strate-
BOX 2
Major Tobacco Control Activities at the U.S. Department of Health and Human Services (HHS)
HHS provides national leadership for comprehensive tobacco prevention and control. Its tobacco control activities include
• Expanding the evidence base for effective tobacco control and current tobacco use. The National Institutes of Health (NIH) and Centers for Disease Control and Prevention (CDC) lead HHS’s efforts on research and dissemination of evidence-based findings to prevent, treat, and control tobacco use. HHS maintains state and federal surveillance and evaluation systems, including the Behavioral Risk Factor Surveillance System, the National Health and Nutrition Examination Survey, National Health Interview Survey, National Survey on Drug Use and Health, Monitoring the Future, and the National Youth Tobacco Survey. These surveys and systems monitor the prevalence of tobacco use and exposure to its constituents, the knowledge, attitudes, and behaviors related to tobacco use, and the effectiveness of tobacco prevention and control interventions. In addition, the Agency for Healthcare Research and Quality led a consortium of federal and nonfederal groups that sponsored the development and release of the U.S. Public Health Service Clinical Practice Guideline Treating Tobacco Use and Dependence: 2008 Update, which is the gold standard for evidence-based clinical treatments for tobacco dependence.
• Building sustainable capacity and infrastructure for a comprehensive tobacco control program. Through the National Tobacco Control Program (NTCP), CDC coordinates national efforts to reduce tobacco-related diseases and deaths. NTCP funds all 50 states, the District of Columbia, 8 U.S. territories, 8 tribal support centers, and 6 national networks devoted to reducing tobacco-related morbidity and mortality and tobacco-related disparities among vulnerable populations, including African Americans, Latinos, Asian and Pacific Islanders, Native American tribes, lesbian, gay, bisexual, and transsexual communities, and those of low socioeconomic status. The funding leverages significant state investments to implement comprehensive, evidence-based tobacco control interventions. Both NTCP and state funding is provided to the National Network of Tobacco Cessation Quitlines, a collaborative effort between NCI and states that makes free telephone cessation counseling available in the United States.
• Regulating the manufacture, marketing, and distribution of tobacco products. The 2009 Family Smoking Prevention and
Tobacco Control Act provides the Food and Drug Administration (FDA) with the authority to regulate the manufacture, distribution, advertising, and promotion of tobacco products. Some of FDA’s responsibilities under the law include setting tobacco product standards, reviewing premarket applications for new and modified-risk tobacco products, requiring new health warnings, and establishing and enforcing advertising and promotion restrictions. The Substance Abuse and Mental Health Services Administration enforces the Synar Amendment, which requires states, the District of Columbia, and the eight U.S. jurisdictions to enact and enforce laws prohibiting the sale of tobacco products to individuals younger than 18 years of age.
• Communicating timely, relevant information about tobacco issues to policy makers, health professionals, business leaders, partners, and the public. HHS, through its agencies and offices, conducts a national public affairs program and provides guidance for public affairs activities (including Web, new media, and broad-cast communications) on the risks of tobacco use and the benefits of quitting. Key elements include the Surgeon General’s reports on tobacco, considered to be the most authoritative scientific publications produced by the federal government regarding tobacco and health, and NCI’s Tobacco Control Monograph series, which provides ongoing and timely information about emerging public health issues in tobacco control.
• Delivering tobacco cessation therapy through direct health care services and health insurance. The Centers for Medicare & Medicaid Services includes coverage for smoking and tobacco cessation therapy for certain beneficiaries. Medicare coverage involves all beneficiaries who use tobacco. For Medicaid, the Patient Protection and Affordable Care Act (ACA) provided coverage for tobacco cessation therapy, without cost sharing, for pregnant women. In 2014, as a provision of the ACA, state Medicaid programs will be forbidden from excluding from drug coverage any pharmaceutical agents for tobacco cessation therapy, including over-the-counter medications approved by FDA. The Indian Health Service Cancer Program seeks to reduce tobacco use by promoting clinical cessation efforts, encouraging community-based education and policy interventions, and providing technical assistance to existing surveillance efforts. The Health Resources and Services Administration’s Bureau of Primary Health Care health center grantees have begun to implement tobacco cessation counseling services that have the potential to affect more than 17 million people, the majority of whom are children and women of childbearing age.
SOURCES: Adapted from HHS (2010a); Koh and Sebelius (2010).
gic action plan to reduce tobacco use, Ending the Tobacco Epidemic, which was launched in 2010 (HHS, 2010a). In addition to delineating the major tobacco control activities of HHS (see Box 2), the strategic plan outlined the four pillars that coordinate and integrate the tobacco control activities of all federal agencies. These pillars are to
1. lead by example,
2. improve the public’s health by strengthening state and local tobacco control,
3. engage the public to change social norms, and
4. advance knowledge in the battle against tobacco.
Koh described several accomplishments since the plan was unveiled. Leading by example, in 2011, HHS declared itself a tobacco-free campus, and all Federal Employees Health Benefit Plan insurers now provide comprehensive cessation therapy benefits that meet the tobacco cessation clinical practice guideline.
He added that during 2011, to strengthen state and local tobacco control, FDA provided new grants to states to improve compliance checks of retail locations. These checks ensure that retailers are abiding by legal restrictions, including not selling cigarettes to minors, not selling single cigarettes or certain candy and fruit-flavored cigarettes, and not offering self-service displays and vending machines in retail establishments where minors are present or permitted to enter. He said FDA has conducted over 60,000 retail inspections and issued nearly 2,600 warning letters and 140 fines to retailers.
Koh noted that HHS is coordinating tobacco control efforts at CDC with those at CMS, which recently expanded its coverage of tobacco cessation therapy to all its Medicare beneficiaries (CMS, 2012) and to pregnant women who are Medicaid beneficiaries (Koh and Sebelius, 2010).
CDC is the lead agency for comprehensive tobacco prevention and control. Its goals are to eliminate exposure, promote quitting, prevent initiation, and identify and eliminate disparities related to tobacco use. In 2010, CDC allocated $108 million to support these efforts, 75 percent of which supported 5-year cooperative agreements in all 50 states, the District of Columbia, 8 U.S. territories, 8 tribal centers, and 6 national networks. The Program Services Branch has a project consultant designated for each of the grantees of the cooperative agreements, who primarily supports infrastructure for comprehensive programs, including training and technical
assistance. These programs include state and community-based interventions, cessation systems change, health communication, and surveillance and evaluation.
“We do need to pay attention to building strong infrastructures,” said Karla Sneegas, chief of the Program Services Branch in the Office on Smoking and Health at CDC. “State and community tobacco control infrastructures ebb and flow over time; consequently, robust training plans are a critical aspect of our program.”
Several speakers stressed the importance of the state programs CDC supports. “In order for us to really make a significant difference nationally, we have to support state programs. These programs enable us to have our ear to the ground so we know what really is going on in communities, and how the tobacco industry is working to market to their customers in these communities,” said Sneegas. Sciandra added, “State programs are the ground troops. Their contractors have an important agenda-setting function. They set the tone to make tobacco something people are talking about in the local community.”
State efforts can also be remarkably effective, as McAfee illustrated by the greater decline in lung and bronchus cancer deaths in California from 1988 to 2009, compared with those nationwide (see Figure 5, and also the section on Successful State and Local Government Antismoking Efforts). “It was [legislative] policies and aggressive media campaigns that California ran every year that resulted in this decline,” he said. Despite their importance, state tobacco control programs have had their funding drastically cut in recent years (see the Financial and Legal Challenges section). Several speakers said that if the majority of tobacco taxes and settlement money were spent on tobacco control efforts, then substantial advances could be made in curbing the tobacco epidemic by helping support statewide programs such as in California. Notably, committing more resources from revenues such as tobacco taxes would help facilitate the minimum financial expenditures recommended by CDC for statewide tobacco control efforts.
CDC also runs its own antismoking media campaigns, including its most recent, Tips from Former Smokers (see the Media Campaign section). A key component of the CDC’s media campaign is the national network of state quitlines (1-800-QUIT-NOW) that it funds. These quitlines offer evidence-based tobacco cessation counseling, supported by a combination of state, CDC, and NCI funding.
The resources that CDC provides “enable states to do something cre-
FIGURE 5 Long-term impact of comprehensive tobacco control in California. With a 15-year investment of $1.8 billion in tobacco control, lung and bronchus cancer incidence rates declined faster in California compared to the rest of the United States from 1988 to 2009 (Lightwood et al., 2008).
NOTE: X-axis includes years (1988–2009, represented as 88–09) and incidence per 100,000 individuals for each year.
SOURCE: Stevens presentation (June 11, 2012).
ative and effective when they may not have the money to run a full-fledged campaign,” said Sneegas. CDC reports also provide the scientific findings that inform local and state tobacco control policies.
ANTISMOKING LAWS AND REGULATIONS
Legislators have passed a number of federal, state, and local antismoking laws and regulations, which have been shown to be effective in helping stem tobacco smoking. Recent federal laws and regulations include, for example, the Children’s Health Insurance Plan and Reauthorization Act (CHIPRA) of 2009, the Family Smoking Prevention and Tobacco Control Act of 2009, and the ACA of 2010.10 CHIPRA raised the federal tax rate for cigarettes on April 1, 2009, from $0.39 per pack to $1.01 per pack.
Family Smoking Prevention and Tobacco Control Act
The Tobacco Control Act11 provides FDA with the authority to regulate the manufacture, distribution, and marketing of tobacco products to protect public health, including a broad set of sanctions for violations of the law, and the ability for FDA to contract with states to conduct retailer inspections. The Tobacco Control Act retailer provisions aim to stem youth smoking by restricting cigarette and smokeless tobacco retail sales to individuals younger than 18 years old and requiring proof of age to purchase tobacco products. FDA has invested in local enforcement of such retail restrictions, creating a tobacco retail inspection program with contracts in 37 states and the District of Columbia. As noted previously, FDA has completed more than 60,000 inspections of tobacco product retailers, resulting in more than 2,600 warning letters and 140 fines. Also prohibited is the sale of tobacco products in vending machines, self-service displays, or other impersonal modes of sale that youth could access. In addition, FDA has used its authority to restrict the sale of certain tobacco products, including cigarettes with fruit and candy flavor additives, and tobacco product
________________
10 Children’s Health Insurance Program Reauthorization Act of 2009, H.R. 2, 111th Congress. 2009, 1st Sess. (February 4, 2009); Patient Protection and Affordable Care Act of 2010, H.R. 3590, 111th U.S. Congress. 2nd Sess. (March 23, 2010); Family Smoking Prevention and Tobacco Control Act, H.R. 1256, 111th Congress. 1st Sess. (June 22, 2009).
11 See http://www.fda.gov/TobaccoProducts/GuidanceComplianceRegulatoryInformation/ucm246129.htm (accessed December 10, 2012).
advertising and marketing to youth. The Tobacco Control Act also prohibits tobacco brand name sponsorship of athletic, musical, or other social events, and of teams and entries in those events, as well as the sale or distribution of items, such as hats and tee shirts, that have tobacco brand names, logos, or selling messages. Some restrictions on product advertising have been challenged successfully in the courts, however.
The act gave FDA direct authority to regulate cigarettes, cigarette tobacco, roll-your-own tobacco, and smokeless tobacco. The Tobacco Control Act also gave the agency jurisdiction, through rulemaking or developing new regulations, over other tobacco products such as cigars, pipe tobacco, hookah, and e-cigarettes that do not make therapeutic claims. Deyton said that FDA intends to extend its authority to these products.
The Tobacco Control Act also bans the sale of packages of fewer than 20 cigarettes, free samples of cigarettes, the sale or distribution of items with cigarette and smokeless tobacco brands or logos, and prohibits reduced harm claims, such as the terms “light” cigarettes or cigarettes that are “low” in tar without prior FDA review to ensure the scientific evidence supports such claims.
The Act has also enabled FDA to establish new smokeless tobacco health warning labels, and required bigger and more prominent health warnings for cigarettes. The law specified that these labels must take up half of the tops of both the front and back panels of cigarette packages, 30 percent of the two principal display panels of smokeless tobacco products, and 20 percent of advertising for both types of products (see the Cigarette Packaging Warning Labels section). Tobacco companies sued FDA over the cigarette health warning labels, and a federal appeals court ruled that the warnings as promulgated were unconstitutional (Bayer et al., 2012; Dooren, 2012). In March 2013, FDA said that it will revise the cigarette health warning labels (Dennis, 2013).
Deyton also described the limitations to FDA’s regulation of tobacco products, including its inability to set nicotine levels to zero, to ban certain classes of tobacco products, or to require prescriptions for purchase of tobacco products unless they are for therapeutic intent. FDA also does not regulate tobacco growing.
The Tobacco Control Act gives FDA authority to conduct research to support its regulation of tobacco products, which FDA has exercised, in part, by launching the Population Assessment of Tobacco and Health study with the National Institute on Drug Abuse. The aim of this large, national,
longitudinal study of both tobacco users and nonusers is to understand the determinants of tobacco use. The study will also facilitate an assessment of how consumers use multiple products together or sequentially, which will be very important for understanding the impact of new products, modified risk claims, etc. Studies will also identify and assess markers for tobacco use in biologic specimens.
FDA also supports the development of better methods to determine the levels of harmful and potentially harmful constituents in tobacco products so that regulations could be developed that reduce health risks by lowering the levels of harmful constituents in tobacco products. “FDA can issue standards appropriate for the protection of public health, including making regulated products less addictive or less harmful,” Deyton stressed.
In addition, FDA is conducting or supporting research on the toxicity or carcinogenicity of tobacco products, as well as research on tobacco dependence conducted by NIH and other institutions to understand the threshold for nicotine addiction. This information will help the agency to elucidate appropriate nicotine levels to set for tobacco products or determine the levels of other compounds that foster nicotine addiction.
Several workshop speakers discussed how the public may perceive the role of FDA in regulating products (such as tobacco) that are known to cause cancer, heart disease, and other health effects. Fong noted that traditionally, FDA approval or premarket clearance of the products it regulates indicates that the products are safe for public consumption, and given this history, the public may misinterpret FDA regulatory decisions about tobacco products. He suggested giving careful thought to how FDA communicates risk about tobacco products. “We don’t want to give people false impressions about what FDA clearance or approval of those products might mean,” he said.
The Tobacco Control Act also gives FDA authority to require tobacco companies to report the levels of the harmful or potentially harmful constituents in their products, and the agency plans to educate the public about this information, Deyton said. In addition, FDA plans to conduct or support media campaigns directed at educating youth about the dangers of tobacco products to help prevent tobacco product use initiation and encourage cessation. McGoldrick noted that the Tobacco Control Act enables FDA to give states and localities the authority to restrict time, place, and manner of tobacco marketing (Campaign for Tobacco-Free Kids, 2012a), although he noted current court challenges may limit those powers to some degree.
Patient Protection and Affordable Care Act
The ACA expands coverage for tobacco cessation therapy, but will not guarantee that all patients will have access to these services and medications. The ACA specifies that all new private health insurance plans must provide beneficiaries with high-value preventative services that receive an “A” or “B” rating from the U.S. Preventive Services Task Force (USPSTF) at no cost to plan members, Koh noted. USPSTF has recommended (with an “A” rating) that clinicians screen all adults for tobacco use and provide tobacco cessation interventions for tobacco users (USPSTF, 2009).
Under Medicaid, the ACA provides coverage of tobacco cessation therapy, without cost sharing, to pregnant women, and in 2014, will forbid state Medicaid programs from excluding from coverage tobacco cessation medication. McGoldrick pointed out that currently, only six states cover a comprehensive tobacco cessation therapy benefit for all Medicaid enrollees, and two do not cover it for any of their Medicaid beneficiaries. Likewise, only five states completely cover tobacco cessation therapy for their state employees, and nine states require private insurance plans12 to cover this treatment (American Lung Association, 2011).
In 2014 the ACA will require certain insurance plans to cover a comprehensive package of items and services called the essential health benefits package. Koh noted that HHS has put forward a bulletin that give states flexibility in selecting a benchmark plan to determine the essential health benefits package (CMS, 2011), and added that the Federal Employee Health Benefits Plan, which covers comprehensive tobacco cessation therapy, could serve as such a benchmark. “This is the benchmark that the federal government has put forward as we move into determining essential health benefits, and we look forward to the states following in that example,” Koh said. McGoldrick added that some insurance companies are expected to offer tobacco cessation therapy coverage once this provision of the ACA is instituted, but will not be required to do so, as each state will define its own essential health benefits package.
McGoldrick also noted that there are some new comprehensive provider organizations that are forming in response to the ACA that might make tobacco cessation therapy a covered benefit, especially because such
________________
12 These include nongrandfathered plans in the individual and small group markets, both inside and outside of the exchanges, Medicaid benchmark and benchmark-equivalent, and basic health programs (CMS, 2011).
coverage is likely to reduce their costs associated with providing medical services to address the health effects of tobacco use. In addition, the ACA provides a new prevention fund and other new resources to support CDC’s tobacco control efforts.
McGoldrick noted that tobacco companies spend more than $10 billion, or 92 percent of their total marketing dollars, at the point of sale of their products13 (Campaign for Tobacco-Free Kids, Counter Tobacco, and the American Heart Association, 2012). “They are making tobacco available, attractive, and affordable for kids and for people trying to quit. We haven’t yet done a whole lot at point-of-sale regulation to try to affect that marketing,” he said. He pointed out that the Village of Haverstraw, New York, had recently passed a ban on visually displaying tobacco products at the town’s 11 retail outlets (ACSCAN, 2012). After the law was passed, however, tobacco companies and convenience stores filed civil suit against the ban, and the Village of Haverstraw voted to rescind the ban, citing concerns over legal costs (Gilbert, 2012). Although every state forbids sale of tobacco products to minors, most of these laws do not seem to affect smoking in that population unless they are severely enforced, Warner noted. However, the Tobacco Control Act makes selling cigarettes to youth a federal violation (Campaign for Tobacco-Free Kids, 2012c; FDA, 2012).
Warner reported that about 30 states ban smoking in workplaces, including restaurants and bars. These laws reduce worker exposure to secondhand smoke (Callinan et al., 2010), decrease daily consumption of cigarettes, and increase the likelihood to quit smoking (Bauer et al., 2005; Fichtenberg and Glantz, 2002). The restriction also decreases employer costs by requiring fewer cleanings and lengthening the life span of certain electronic equipment, said Warner. There is also substantial evidence that adopting smoke-free workplace laws reduces the number of heart attacks
________________
13 Point of sale marketing expenditures include point-of-sale advertising; price discounts; promotional allowances to retailers, wholesalers, and others; coupons; and retail value added (bonus and nonbonus) (Campaign for Tobacco-Free Kids, Counter Tobacco, and the American Heart Association, 2012).
(IOM, 2009b). “Is there anything else in the entire field of medicine or public health that can have that much of a health impact with that modest of a policy change?” Warner asked. The benefits of smoke-free laws and other tobacco restriction efforts have been clearly demonstrated in New York and California. Stevens showed how tobacco control efforts such as smoke-free laws saved more than 1 million lives14 with reductions in tobacco-related diseases and $86 billion in health care savings (Lightwood et al., 2008).
Even within states that have not adopted smoke-free laws, many cities, towns, and municipalities have adopted such ordinances, at least in bars and restaurants, according to McGoldrick. About two-thirds of the U.S. population reside in a state, town, or city that have smoke-free restaurants and bars (see Figure 6), and many of these have smoke-free workplaces as well.
Many states and localities, including New York City and a number of jurisdictions in the state of California, have also instituted smoke-free parks and beaches. The entire campuses of many universities, such as the University of Michigan, are now smoke- or tobacco-free, with no smoking and/or tobacco use allowed both inside and outside of campus buildings. “I have people say to me, ‘This policy is a failure because I have seen people smoking on campus,’ and I respond, ‘We have laws against murder and some murders are still committed. It is widely accepted as a good policy,’” Warner said.
Many hospitals are also becoming smoke free, even limiting smoking on the entire health science campus, according to Sarna (Sarna et al., 2005). “This is making a difference. Some nurses told us it was really annoying when they have to use their entire break to walk a block so they can smoke,” she said. Warren, Cummings, and others attribute these types of behaviors to a remarkable addiction caused by tobacco that compels health care workers to travel off-site to smoke during work breaks.
Some states, such as Arkansas and California, also have laws mandating smoke-free cars in which children are passengers. But McAfee noted more could be done in the smoke-free arena, including instituting smoke-free multiunit housing. Nearly one-third of homes in the United States are rental properties, 65 percent of which are multiunit housing (representing 22.5 million households). Although homes, especially multiunit housing that may be sharing air space, are a significant source of secondhand smoke exposure, efforts to implement smoke-free policies in these areas have been limited (King et al., 2010).
________________
14 California Department of Public Health (unpublished data).
FIGURE 7 Real cigarette prices and per capita consumption, United States, 1970–2004.
SOURCES: Warner presentation (June 11, 2012) and Chaloupka (2010).
increasing state budgets, some of which may be earmarked for tobacco control programs. “Taxation is the single most important policy that we have, in terms of trying to change smoking quickly and substantially,” said Warner (see Figure 7).
This pattern has been consistent in at least 15 countries, he noted, and indicates that price increases of 10 percent decrease cigarette consumption by 3 to 5 percent. Half of that reduced consumption is due to quitting smoking, he said, while the remaining half is due to a reduction in daily consumption.
Warner added that many people who smoke have low levels of income and education, and this same group tends to be more responsive to price increases. Children are two to three times more price responsive than adults, “which tells us that a cigarette tax is by far the most important thing we can do if we want to reduce youth smoking,” he said. Unfortunately, most tobacco taxes are not spent on prevention, however. McGoldrick reported that though state tobacco tax and settlement revenues in 2012 are estimated at $25.6 billion, only $457 million was spent on tobacco control, which is far less than the $3.7 billion recommended by the CDC and $10.5 billion spent on advertising by the tobacco industry (Campaign for Tobacco-Free Kids, 2012b,d; CDC, 2007d). This relationship was also noted by McAfee.
Warner suggested several innovative laws that might help stem the use of tobacco products. He suggested controlling the supply of tobacco products, noting that a New Zealand parliamentary committee suggested banning the availability of tobacco products by 2025, and doctors in Singapore have proposed a ban on tobacco for people born after 2000 (Stark, 2011). Warner suggested that a less restrictive ban on tobacco could be instituted, such as banning combusted tobacco products. Tobacco control advocates in Australia are pushing for licensing people who smoke; people would pay for the license to smoke each year, and if they quit, they would be returned the money they spent on such licensing, with interest (Stark, 2011). Warner also suggested that FDA should require the reduction of nicotine to nonaddicting levels in tobacco products, an action that is within FDA’s regulatory authority.
McGoldrick noted some innovative restrictions on tobacco products that New York City tried to institute, but these are currently being challenged in court by tobacco companies. These restrictions include warning signs at the point of sale and bans on all flavored tobacco products, to complement the federal ban on flavored cigarettes. Also being challenged in court are bans on tobacco product coupons and other price discounting that Providence, Rhode Island, instituted.
McGoldrick also suggested that a nontax method to influence the price of tobacco products could be minimum price laws on cigarettes that are updated regularly so they “do a better job of keeping price up to overcome some of the price discounting that the companies spend billions of dollars on to undercut tobacco taxes,” he said.
One topic that generated debate at the workshop is a policy that would allow employers to legally refuse to hire anyone who smokes. The Cleveland Clinic has instituted such a policy, said Warren. According to the policy, applicants take a cotinine test during their pre-placement physical exam; those who test positive are referred to a tobacco cessation program which is paid for by the Cleveland Clinic. If after 3 months, an applicant has a negative test result, he/she may be reconsidered if the position remains unfilled (Cleveland Clinic, 2012a,b). The World Health Organization also has an antismoking hiring policy, according to Dresler. Some employers are instituting a ban on hiring people who smoke as a way of saving on health insurance costs, she added.
But Healton thought that not hiring people who smoke was not a good
policy to pursue because tobacco dependence is a medical problem that disproportionately affects low-income people. She also expressed concern about the precedent such a policy could set. “The next thing will be weighing workers to see whether they can stay employed,” she said.
SUCCESSFUL STATE AND LOCAL GOVERNMENT ANTISMOKING EFFORTS
A number of state and local governments have successfully used a combination of laws, regulations, and media campaigns to lower their prevalence of smoking, several speakers reported at the workshop. For example, between 2001 to 2012, there have been more than 100 state tobacco tax increases, 46 of which resulted in a price hike of 50 cents or more. Approaches to tobacco control in New York State and New York City, Massachusetts, and California were discussed.
New York State and New York City
The state and city of New York have passed laws mandating smoke-free workplaces and public places and increased funding for tobacco prevention and cessation programs, including a media campaign to help people quit. These initiatives led to dramatic declines in smoking among youth, McGoldrick reported. From 2002 to 2007, there was a 50 percent decline in the rate of youth smoking, and a 19 percent decline in adult smoking (see Figure 8) (CDC, 2007b; New York City Department of Health and Mental Hygiene, 2012).
Sneegas added that smoke-free initiatives also reduced hospital admissions from heart attacks by 8 percent, saving the state $56 million (Juster et al., 2007). “This is a shining example of what we can do,” she said. “The only place this doesn’t work is where you don’t do it,” McGoldrick added.
Sciandra noted that the antismoking campaigns in New York are based on effective presentation of evidence, building diverse political coalitions, media advocacy, and direct lobbying with legislators. He posited that the reason why past campaigns in New York and other states were so effective is because there was an office in each of the states completely dedicated to antismoking efforts that was funded by the Robert Wood Johnson Foundation SmokeLess States program. This office helped keep all relevant voluntary agencies and officials focused on antismoking advocacy efforts. “Today a robust, active, evidence-driven tobacco control program is the single most
SOURCES: McGoldrick presentation (June 11, 2012); CDC (2007b); and New York City Department of Health and Mental Hygiene (2012).
important element helping frame tobacco as a public policy issue. But we still need a well-coordinated aggressive nongovernmental coalition to advocate for those policies,” he said.
Massachusetts was an early adopter of strong tobacco control efforts, including raising tobacco taxes, funding media campaigns, and passing smoke-free ordinances. More recently, Massachusetts dramatically decreased tobacco use among Medicaid (MassHealth) participants by providing tobacco cessation therapy benefits to all its Medicaid beneficiaries and publicizing that benefit. This benefit was mandated under the Massachusetts health reform legislation passed in April 2006. Although planned as just a 2-year pilot, within a year the program was made a permanent benefit, due in part to its high use, reported Thomas Land, director of the Office of Statistics and Evaluation for the Bureau of Community Health and Prevention at the Massachusetts Department of Public Health. The legislation also
required annual evaluations of the benefit in regard to its use, cost savings, and clinical outcomes.
The cessation therapy benefit covers 16 counseling sessions and all FDA-approved medications with copays as low as $1 to $3. “This is essentially a no-hoops approach that reduces the barriers as much as possible,” Land said. He added that the benefit was designed to recognize that tobacco dependence is a chronic relapsing condition and supports two quit attempts annually.
The state promoted the benefit to both providers and consumers through many media venues, including radio, professional and consumer newsletters, the transit system, television, and brochures. Simultaneously, major pharmaceutical companies conducted media campaigns to promote their tobacco cessation medicines. According to Land, this publicity boosted consumer awareness of the benefit from 31 percent when the campaign began to 75 percent 18 months later. By May 2009, 37 percent of MassHealth smokers utilized the tobacco cessation therapy benefit, and Land added that currently use is up to 55 percent (Land et al., 2010b).
This initiative was highly effective, fostering a decline in smoking prevalence among those with MassHealth insurance by 10 percent in just 2.5 years after the benefit was implemented, while no decline was seen in a comparable population without insurance (Land et al., 2010b). A decline in smoking continues in the MassHealth population, whose smoking prevalence rate is as low as it has been in two decades, with significant decreases being maintained for nearly 4 years, Land reported.
By focusing on heart disease, researchers were able to document immediate health benefits of the MassHealth beneficiaries who used the cessation therapy benefit. Land noted that the health benefits of quitting smoking are seen much sooner in heart disease as compared to cancer. Following first use of the cessation medications, there was a sharp reduction in hospital claims for heart attacks among MassHealth beneficiaries—a 46 percent drop annually. There also was a sharp reduction in claims for acute coronary heart disease or atherosclerosis (Land et al., 2010a).
Other researchers who evaluated the MassHealth benefit estimated a return on investment of $2.12 for every dollar spent in program costs (Richard et al., 2012). “That is an impressive number, but what is even more impressive is that the bulk of savings associated with quitting smoking may actually be in the future, as all the respiratory illnesses, diabetic complications, and cancer are added into the calculation,” Land said. These results were so favorable, he added, that the Massachusetts Department of
Public Health is now working with a major insurer in the state to develop cost-sharing models for employers who provide cessation therapy benefits that parallel or possibly even expand what was done at MassHealth.
Massachusetts has also fostered declines in smoking by innovatively applying the benefits of EMRs and other electronically integrated systems, Land said. As part of the Massachusetts health reform law, a Massachusetts All Payer Claims Database was launched. The law specified that most medical claims be prepared in standard formats and submitted to a common registry. When operational, the registry will be available for use by government and nongovernment researchers, and will enable the tracking of the impact of public health programs much more easily.
PopMedNet is another electronic innovation being put to use by the state. This tool can conduct regular surveillance of ambulatory care settings. It is a distributed query system that accesses clinical data onsite and returns summary results to agencies, such as the Department of Public Health. PopMedNet will enable analysts to conduct population health studies without taking possession of clinical records. It is also aiding assessments of provider adherence to core meaningful use measures, such as tobacco use. In addition, Massachusetts is developing the technical capability to link EMRs to nonclinical resources, such as tobacco cessation counseling. With the commitment of partner health centers, these referrals will be electronic and updates from community partners will be added directly into patient EMRs.
According to Land, Atrius Health is demonstrating the usefulness of integrated electronic systems in delivering tobacco cessation therapy. Atrius is a multisite primary care provider in the suburban Boston area. It uses an EMR capable of sending electronic referrals to the state’s tobacco quitline, and receives electronic information back to assess patient progress. “Within the realm of primary care, Atrius is putting together a lot of previously unconnected pieces,” Land said. A study of 2.6 million clinical encounters at Atrius Health found that once tobacco interventions with patients become routine, there are significant reductions in self-reported smoking and smoking-related disease office visits compared to what was achieved at Atrius sites before the electronic integration was instituted (Land et al., 2012).
Prior to these innovations at Atrius, doctors spoke with patients about smoking and some were offered cessation medications or were referred to a tobacco quitline, according to Land, but “the difference is that the component parts didn’t function well together. I believe that linking these elec-
tronic pieces together into a whole may take years, but the impact will be felt by nearly every citizen for decades to come because the whole is greater than the sum of the parts.” Warren also confirmed that he and others have successfully instituted an EMR-based assessment and cessation program at Roswell Park Cancer Institute for cancer patients during treatment, and have demonstrated feasibility, efficacy, and clinical efficiency.
Between 1988 and 2009, the incidence of lung and bronchus cancers in the state of California fell by nearly 30 percent—about twice as much the decline in such cancers nationwide during the same time period, Stevens said (ACS, CDPH, and CCR, 2011; see also Figure 5). “California used to have a higher lung cancer incidence, [but] now it is nearly four times lower than the rest of the United States,” she said. The number of packs of cigarettes sold in California has also declined substantially from 2.4 billion in 1988 to under 1 billion in 2011 (California State Board of Equalization, 2012), despite an increase in population during that time. Stevens attributed much of that decline to state policies, including a tax on cigarettes instituted in 1988. She added that 20 percent of the income generated from that tax is dedicated to state tobacco control efforts.
The state also used media to change social norms and build awareness of the hazards of secondhand smoke, which created an environment that led to laws requiring smoke-free environments, including indoor work sites, schools, cars with children, beaches, parks, apartment buildings, and condominiums. Between 1990 and 1994, nearly 300 counties and cities had implemented smoke-free policies in the state. By 1995, there was a state law banning smoking in indoor work sites. In 1998, the state focused on making bars smoke free. “Before the law went into effect, people thought it was going to be a calamity, but people didn’t stop going to bars. In fact, bar revenues continued to go up after the ban went into effect,” Stevens noted.
Stevens said that California’s success has been based on the ability of these efforts to change the social norms about smoking. For example, shortly after the state focused on educating the public about the hazards of secondhand smoke, there was a marked increase in the percentage of smokers who voluntarily banned smoking in their own homes, with nearly 60 percent of all smokers instituting such bans by 2008 (California Tobacco Control Program, 2010). “They don’t want their kids exposed to smoke and
they don’t want other people to know they smoke so they ban smoking in their own homes,” Stevens said.
The state and local communities have also instituted policies on cigarette vending machines, tobacco displays, retail licensing, and bans on samples of tobacco products. “Once you start implementing policies, people are open … to implementing more policies,” Stevens noted.
In California, the process for tobacco control reforms has been goal-oriented and coordinated among media, local health departments, and community programs, said Stevens. The state develops and airs supportive media; provides training and technical assistance to local partners; supports local education; and qualitatively and quantitatively evaluates these efforts. An example of its training was a state conference on smoking in multiunit housing. At the conference, scientists described how smoke travels from one apartment to another, and detailed the health hazards of such secondhand smoke.
Studies illustrate the positive outcomes of California’s tobacco control efforts, including $86 billion saved in health care dollars (Lightwood et al., 2008); 1 million lives saved,15 mostly from young people who never started smoking; major declines in heart disease; and a decline in cancers of the mouth, throat, bronchi, and lungs (ACS, CDPH, and CCR, 2011). In addition, fewer children in California start smoking or become regular smokers. Less than 3 percent of Californians currently smoke more than 20 cigarettes per day (Pierce et al., 2011).
EDUCATION, OUTREACH, AND MESSAGING
The Surgeon General reports that have focused on the health effects of tobacco use have been integral to tobacco control efforts from the very beginning. Warner noted that such reports can have powerful effects, as indicated by the 1964 Surgeon General’s report linking smoking to lung cancer. This report prompted a 15 percent drop in cigarette consumption within just three months of release, although recidivism pushed consumption levels back up somewhat (Warner and Mendez, 2010). More recently, the Surgeon General has released a report on how tobacco smoke causes the various illnesses linked to it (HHS, 2010b), and a report on preventing
________________
15 California Department of Public Health (unpublished data).
tobacco use among youth and young adults (HHS, 2012). As Koh noted, these reports and other recent federal efforts “reinvigorate the national conversation” about the health hazards of tobacco use and how to combat them.
According to Warner, tobacco control educational efforts that are school based have been less effective. “That is not to say that it is not theoretically possible to do something in schools that is effective. But as it is implemented now by teachers, who are overburdened and do not provide this kind of education every year, it has no impact,” he said.
Alexander Prokhorov, professor and director of the Tobacco Outreach Education Program at MD Anderson Cancer Center, agreed. “School-based programs don’t work when we don’t have continuity—unfortunately, we have sporadic use of these programs in our schools. Do we expect them to learn anything about tobacco use if we just have a few hours somewhere in the middle of the school year? Probably not. We need to do a much better job in that regard,” he said.
Researchers are using a number of approaches to improve tobacco prevention and cessation education for youth (Dobbins et al., 2008; Grimshaw and Stanton, 2006; Sherman and Primack, 2009). According to Prokhorov, these approaches include both school-based settings as well as other settings in order to expand the reach among youth who may be at risk for tobacco use. Prokhorov’s research has focused on incorporating the newer technologies adolescents use, including the Internet, cell phones and cell phone apps, social networking sites, video game kiosks, and computer tablets to encourage tobacco prevention and cessation. “We live in an era of technology,” Prokhorov said, noting that there has been a 57 percent increase in smart phone users in the United States between 2010 and 2011, and that nearly all public schools have Internet access (CTIA, 2011; Gray and Lewis, 2009). “This technology gives us a lot of opportunity to do our job, in terms of smoking prevention and cessation education,” he said. Technology also can improve data collection and management of educational projects, he added.
A Smoking Prevention Interactive Experience (ASPIRE) is the flagship tobacco control educational project that Prokhorov developed. It is aimed at smoking prevention and cessation among middle and high school students. This program is computer based and interactive, and is now being disseminated nationwide with 24 states enrolled. In addition to school districts, juvenile justice systems, health plans, and other entities are using ASPIRE. A major component of this program shows teenagers what smoking tobacco can do to their bodies.
ASPIRE was aimed at and tested among urban-based, economically
disadvantaged high school students. Additionally, Prokhorov and his colleagues developed another program, CURBING, that targets rural and suburban children, and addresses both smoking and smokeless tobacco. It includes a game-like format, an interactive blog, and the ability to interview Jeff Bagwell, a famous baseball player, who recounts a story about smokeless tobacco.
A study found that ASPIRE substantially reduced smoking initiation at 18 months after having five 45-minute computer sessions, compared to a control group given a booklet about smoking health effects (Prokhorov et al., 2008). Among teenagers considered at highest risk of starting smoking because their parents smoke, they confront significant peer pressure, or other factors, smoking initiation was less than 3 percent among those who participated in ASPIRE, compared to more than 15 percent among those who did not. In addition, teenagers seem to appreciate the program, with about three-quarters of those surveyed saying they would recommend ASPIRE to others. The program was recently translated into Spanish, and there are plans to translate it into Vietnamese, Portuguese, and other languages, Prokhorov reported.
Prokhorov is currently developing a website-based tobacco control program for college students that is about to be tested. He also developed a video game kiosk for young adults not in school, called Health Scare General Hospital. Pretesting of this video game showed it increased smoking knowledge in 94 percent of participants, and inspired 82 percent to try quitting smoking or never to start smoking. One kiosk is now housed in a detention center and the other is in a recovery center in Texas. Prokhorov noted a recent study that showed video games have the potential to reach a substantial number of people who smoke (Raiff et al., 2012).
Responding to both the surge in use of computer tablets and the high smoking rates in the military, Prokhorov also developed a tablet computer-based video that he is testing with 2,000 soldiers at Fort Hood, Texas. He is also currently developing a program called Project ACTION, which sends antismoking text messages to economically disadvantaged adults who are trying to quit smoking. A study found that such messaging improved the 6-month quit rate by 50 percent (Free et al., 2011).
Prokhorov noted that only 2 percent of all consumer health cell phone apps are focused on smoking cessation, and a review of those available found most did not adhere to current cessation therapy guidelines (Abroms et al., 2011). He suggested researchers develop more evidence-based apps for smoking cessation, such as QuitSTART, developed by NCI, and My Quit
Coach, developed by LIVESTRONG. “They are free and very good,” he said and can be accessed at smokefreeapps@mmgct.com. In addition to showing quitters’ charts that mark their progress, My Quit Coach provides access to the LIVESTRONG community, a virtual community that works like an Alcoholics Anonymous sponsor to whom a person’s quitting progress is announced.
Prokhorov also recently developed an educational app on secondhand smoke aimed at Mexican Americans, as well as a cessation educational app for health care professionals called QuitMedKit. The latter provides a tool that follows clinical practice guidelines, which in turn makes counseling of patients more efficient and sustainable (Prokhorov et al., 2010). Both apps will soon be submitted to the Apple App Store for review.
He noted that interactivity and connectedness to online social media still appears to be in the early stages of development among tobacco control-relevant sites, as is use of social networking (Freeman and Chapman, 2012). Twitter is often used to build quit-smoking social networks, but one study found the content on these networks is often not consistent with clinical guidelines, and that most of these Twitter accounts are no longer active (Prochaska et al., 2012). “There are social networks out there, but we still don’t know how to use them correctly,” he said. One possible cost-effective use of Facebook could be to recruit young people who smoke to participate in surveys of their tobacco use, he added. The potential benefit of preventing tobacco use in youth is tremendous, he said. Using advances in technology and adapting to social media used by youth is a potentially powerful method to prevent future tobacco use.
YouTube is another widespread medium that could be used to provide education about tobacco products and cessation, but one study found that although there is information on smoking cessation on YouTube, much of that information is not evidence-based (Richardson et al., 2011). Prokhorov said a number of videos on the site do promote smoking. For example, one popular video, called True Hookah King, discounts the evidence that hookah smoking can be harmful to health. “Kids do watch it—it gets lots of hits—so we need to find a way to counter that,” he said. Fong added that tobacco companies already have quite a presence on the Internet, where their promotions have prompted the registration of tens of millions of smokers with whom the companies can directly communicate.
Prokhorov noted that a major challenge in developing tobacco control educational programming is the rapid evolution of tobacco products, and innovative ways to promote and distribute them, compared to the slow
pace of evidence-based research to find out what educational programming about these products is effective. “The industry is like a sports car, and we are like a bicycle. We won’t be able to catch up to industry, so we have to come up with a different way of getting evidence,” he said.
Tobacco control media campaigns include messages delivered via television, the Internet, and print media. A 2008 NCI monograph summarized the extensive evidence regarding the ability of media campaigns to influence tobacco use (NCI, 2008). The monograph described how the tobacco industry has been effective at encouraging tobacco use through its advertising. Tobacco control media campaigns have also been an effective mechanism to discourage tobacco use. Subsequent studies have further strengthened the evidence base (for example, see Farrelly et al., 2009; Vallone et al., 2011a,b; Wakefield et al., 2010). Tobacco control ads can have powerful effects, studies document. For example, the Fairness Doctrine ads16 from mid-1967 to 1971 led to 4 consecutive years of decline in adult per capita cigarette consumption, Warner pointed out (Warner and Mendez, 2010). McAfee added that more than one-fifth of the decline in youth smoking that occurred from 1999 to 2002 could be attributed to the Legacy truth® campaign ads (Farrelly et al., 2005), described in more detail in Box 3. He also noted that exposure to state and Legacy tobacco control advertising was linked to less smoking and increased intention to quit or making a quit attempt in the past year (Emery et al., 2011).
In addition, CDC’s Tips from Former Smokers campaign, described in Box 3, was linked to a doubling of calls to quitlines as well as a tripling of unique visits to the website www.smokefree.gov listed in the ads. McAfee estimates that the campaign led to more than 700,000 people clicking on the website or calling a quitline. In addition, there were more than 2.8 million visitors to the campaign website.
Comprehensive restriction of ads for tobacco products can also be effective. One study found that elimination of all forms of advertising and promotion of tobacco products could reduce smoking by about 7 percent, whereas partial bans do not demonstrate this effect (Saffer and Chaloupka,
________________
16 The Fairness Doctrine required broadcasters to ensure fair and balanced coverage of controversial matters, and stations that aired cigarette commercials were required to donate airtime to antismoking messaging (CDC, 2010).
BOX 3
Successful Media Campaigns
CDC’s Tips from Former Smokers
This national campaign consisted of paid advertising and public service announcements placed in/on television, radio, magazines, newspapers, billboards, bus shelters, theaters, and online through digital video, search, and mobile channels. Additional information and resources were made available to the public through the Internet, including Facebook, Twitter, and YouTube.
The campaign included videos featuring real people with smoking-related medical conditions, such as cancer, stroke, asthma, and amputated limbs. “These are real people engaging in their communities,” McAfee said, noting that the states in which the featured people live have had follow-up ads and great media coverage. Some of these videos have had over a million hits on YouTube.
The Tips campaign emphasizes the immediate damage and suffering smoking-related conditions can cause to the body, rather than the deaths that may ensue. The campaign featured a number of individuals who experienced smoking-related diseases at a relatively young age, some of whom were diagnosed before they were 40 years old.
The campaign also features three former smokers who provide tips on how they successfully quit. These tips include throwing away cigarettes and ashtrays, exercising, identifying a strong reason to quit, and “just keep trying,” all strategies that have been shown to help people quit.
2000). Fong pointed out the need to reach youth with messages that appear on the Internet and other new media channels to which they tend to be drawn. “We need to figure out how to make our presence known in the channels in which our young people and others are inhabiting,” he said.
Healton noted that when Legacy first launched its truth® campaign in 2000, 90 percent of youth said they got their health information from television. Now 90 percent of youth receive their health information from the Internet or similar venues. Consequently, the truth® campaign is in the process of a massive rebranding effort and will relaunch with a heavy focus
The ads provide an encouraging message that it is possible to quit smoking, and resources are available to help (1-800-QUIT-NOW and www.smokefree.gov).
Legacy truth® Campaign
Launched in February 2000, the truth® campaign is a national youth smoking prevention campaign that exposes the tactics of the tobacco industry, the truth about addiction, and the health effects and social consequences of smoking to enable teens to make informed choices about tobacco use.
One popular video featured in the campaign shows emergency vehicles unloading 1,200 body bags in front of a tobacco company, illustrating the number of people killed by tobacco each day. According to Legacy President Cheryl Healton, this ad featured young people making an “in your face” sarcastic statement against the tobacco industry.
The truth® campaign has placed ads and videos in a wide range of media sources, including television, radio, print, online advertising, websites, social networking sites, video-sharing sites, branded entertainment and clothing apparel, and theater advertising.
Healton said that the truth® campaign has not only fostered an increased call volume on quitlines, but also has worked on a population level to reduce smoking initiation.
SOURCES: CDC (2012c); Farrelly et al. (2005, 2009); Legacy (2012); YouTube (2006).
on the Internet. Stevens pointed out that although young people are more likely to use the Internet, certain population groups, such as people who smoke and those with low incomes, tend to watch more television and suggested, “Whenever you can afford to do a television ad, it is still the most effective way to get your message out.”
Cigarette Packaging Warning Labels
In the United States, warning labels on cigarette packaging could expose people to more than 8 billion ads a year, the number of cigarette packages sold each year, said David Hammond, associate professor in the School of Public Health and Health Systems at the University of Waterloo.
“The health warning is a traveling billboard that appears in essentially every corner store, travels around in people’s pockets, and ends up being placed on tables and other sites,” he said. Hammond suggested linking the messaging in the warning labels to other media campaigns.
FDA developed graphic warning labels for cigarettes that, unlike previous warning labels, would take up a sizable portion of the package and visually display the hazards of smoking. However, a federal appeals court ruled that the warnings as promulgated were unconstitutional,17 and FDA said that it will revise the warnings (see section on the Family Smoking Prevention and Tobacco Control Act). More than 50 other countries have instituted such labeling (Hammond, 2011).18 In most of these countries, the labels constitute 50 percent of the pack size, and depict disturbing images, such as cancerous lungs, corpses, mouth discoloration, and people in mourning. An advantage of these graphic depictions is that they are understood by people who may have low literacy and may be unable to read the writing that accompanies the graphics, Hammond said. Another approach to making cigarettes less marketable was taken by Australia, which required plain packaging in an unappealing color (Kirby, 2012).
Studies show such labels and packaging change people’s attitudes toward smoking and make them think about quitting. For example, one study found that following the release of graphic health warnings in the United Kingdom, 43 percent of people who had never smoked claimed that the health warnings persuaded them not to start smoking, and 60 percent of nonsmokers could recall a specific warning on the UK packs (European Commission, 2009). More than half of all Australians who smoke reported that the warnings made them more likely to think about quitting, and about a third said they spurred them to try quitting (Shanahan and Elliott, 2009). When graphic warning labels also include quitline numbers, the use of those quitlines dramatically increases, several studies have found. A survey of 27 European countries found that respondents who were younger, less educated, or manual workers were more likely to report that warnings on
________________
17 R.J. Reynolds Tobacco Company, et al., Appellees v. Food and Drug Administration, et al., Appellants. U.S. Court of Appeals for the District of Columbia Circuit. No. 11-5332 (August 24, 2012).
18 Hammond (2011) includes images of international graphic warning labels. Images of the FDA warning labels are available at http://www.latimes.com/health/boostershots/la-he-smoking-cigarette-warning-labels-pictures,0,6757388.photogallery (accessed September 13, 2012).
cigarette packages are effective at informing them about health hazards of smoking and keeping them from smoking (European Commission, 2009).
Warnings can increase health knowledge. After Australia instituted warnings about how smoking can cause blindness, belief in this sort of health effect of smoking increased over time (Hammond et al., 2006). Warning labels also capitalize on health concerns; 92 percent of U.S. smokers cited health concerns as their primary reason for wanting to quit. In addition, warnings reduce the impact of product branding and make cigarettes less appealing, 90 percent of Canadian youth surveyed reported. “These labels reduce the marketing value wrapped up in all those brand elements that the industry designs,” Hammond said.
Scientifically, it is harder to isolate the role of graphic warning labels on initiation and cessation, but there are data that suggest that they have a positive impact on reducing smoking. The year graphic health warning labels were implemented in Canada, Hammond said that the country experienced its single largest decrease in smoking prevalence on record over the past 40 years. However, because Canada also undertook a variety of other tobacco control measures it could not be determined with mathematical precision how much the labels contributed to that decline in prevalence.
Hammond made several suggestions for improving the impact of health warning labels, including rotating the sets of warnings. “Just like you don’t run the same advertisements for 30 years, you shouldn’t leave the same health warnings on the pack for 30 years,” he said. He added that the size of warning is very important because the larger the size, the more room to communicate information and the less room for industry branding. “When you walk into a store in Uruguay or Canada, you don’t see the Marlboro or Camel logo first. Instead you see lung cancer or emphysema,” Hammond said.
Message Content, Framing, and Placement
The content of tobacco control messaging is an important factor in its effectiveness, several speakers stressed. Hammond suggested including supportive information on quitting as well as using direct, succinct, and hard-hitting messages, such as “Smoking kills” in large letters. He stressed that pictorial warnings have greater impact than text warnings. Studies have found graphic health effects are rated as more effective and, especially among youth, those that depict the negative aesthetic health consequences
can be more effective than those that depict diseased lungs or other health effects inside the body (reviewed by Hammond, 2011).
People who smoke also rate warnings that depict the effects of smoking on loved ones as being more effective than the effects on smokers themselves. Personal testimonials are particularly effective, Hammond said, especially when they come from real people. “What people don’t really like in health warnings is the feeling that the government is telling you what to do. That is especially more so for established smokers than nonsmokers.”
Although most of the studies on what content works in warning labels rely on the reports of participants, many of their findings are confirmed by studies that track participant eye movements or functional MRI studies of brain areas activated by the ads, Hammond said.
Healton said ads that cite the health effects of smoking are not powerful for teens who have not yet started smoking, but are at risk for smoking initiation. For these adolescents, ads that depict rebellion against an adult or authority figure are more powerful and address that youthful need for rebellion that can inspire children to smoke.
“Our Legacy truth® campaign body bags ad depicts a rebellion against an evil industry that is trying to do something bad to society. That kind of channeling of rebellion is appropriate for kids open to smoking because they are risk-taking kids with a different psychometric profile than your average goody-two-shoes kid.”
Healton noted that antismoking ads developed by Philip Morris were not effective (Wakefield et al., 2006), and suggested it may be because they featured wholesome-looking children who did not fit the profile of the typical smoking teenager in the United States. “It is not only who is depicted, but what they are doing. Health effects are important and they can be in the mix, but they shouldn’t be the central thematic in our experience,” Healton concluded.
Hammond added that what seems to resonate with teenagers are warning labels that depict the aesthetic personal appearance consequences of smoking. He noted that the smoking ads in the 1920s worked because they glamorized smoking, and ads that do the opposite might deter people from smoking.
Because most people already recognize that smoking is harmful to their health, Cummings suggested devising new messages to use in media campaigns. These messages could focus on the wrongdoing of the tobacco industry, such as their manipulation of tobacco products to increase the likelihood that people will become addicted to them, he said.
Hammond countered that there has been an evolution in the ads based on the health effects of smoking. He claimed the newer ads convey the information that smoking is bad for one’s health in a more emotionally engaging way. “The key is emotional engagement. These ads strike a chord.” He pointed out that the CDC’s Tips from Former Smokers ads had many themes in addition to direct health effects, including personal loss, and the impact that smoking has on others. There also are anti-industry themes in that ad campaign. “The best ads hit on more than one level,” he said.
Cummings stressed that “we talk about reducing tobacco almost separate from reducing cancer. We have got to pair those together, because the public doesn’t really care about reducing tobacco, but they want to reduce cancer mortality. Unless those are linked, we are not going to win the war on cancer.” The complexity of the issue was noted by McAfee who stated that tobacco control efforts, including media, quit resources, comprehensive state programs and funding, are facing changing demographics.
Positive or Negative Message Framing
There was extensive discussion at the workshop about the advantages and limitations of positive and negative framing of messages in media campaigns. “There may be unintended consequences of the most shocking ads, and they may not translate into constructive behavior as opposed to more supportive media messages,” warned Abrams.
Toll noted that messages can be gain-framed, or framed in a positive way, such as “quitting smoking will make you live longer,” or they can be loss-framed, such as “if you don’t quit smoking, you will die sooner.”
One small study found teens rated the loss-framed warnings as probably working better, in terms of impeding their plans to smoke in the future (Goodall and Appiah, 2008). Another study involving tobacco control messaging in conjunction with tobacco cessation medicine found approximately a 12 percent greater sustained quitting rate in those given gain-framed messages, which was significant compared to those given loss-framed messages (Toll et al., 2007). Another study found that loss-framed messages were better received by individuals highly dependent on smoking, while individuals with lower tobacco dependence reacted more positively to the gain-framed messages (Moorman and van den Putte, 2008). But these findings contradict the results of a subsequent study (Fucito et al., 2010). Another study found that when counselors at a quitline incorporated gain-
framed messages, people were significantly more likely to quit smoking 2 weeks later than those given standard care messages (Toll et al., 2010).
Despite the mixed findings, Toll asserted, “We feel that people [with high nicotine dependence] exposed to gain-framed messages will quit smoking at greater rates because if you are highly dependent on nicotine, you may be scared you are going to get the health consequences of smoking and less confident about your ability to quit.” But he noted that graphic warning labels are harder to put into a gain-framed context. “We don’t want Marlboros to have smiley faces on their packages,” he said. Warren suggested that gain-framed messaging that promotes a positive outcome may be more appropriate for cancer patients, but he acknowledged that this approach is as yet untested.
Healton suggested the ideal messaging outreach for cessation therapy probably should have a mix of both gain- and loss-framed messages. She noted that when a gain-framed tobacco control ad campaign was run in Grand Rapids, there was an 11-fold increase in quitline call volume.19 She also pointed out that as an ex-smoker, she tended to shut down her responses to loss-framed messages because they aroused so much fear. “I think the jury is still out on what is the best mix for bringing about this type of population-based response to communications. It is intuitive that there are all sorts of people who smoke, with different motivations, so that an ideal model would be one that provides some kind of mix,” Healton said. Fong concurred. “It is important for us to be able to speak to multiple audiences using mass media channels, so for something like warning labels, there should be something for everybody in there.”
Ostroff added that the ads for tobacco cessation medications tend to be “feel-good” ads that are hopeful and gain-framed. She suggested more research could be conducted on framing messages about how easy or hard it is to quit to assess the best approach. Healton pointed out that the ads for tobacco cessation medicines are based on extensive consumer market research, with companies tracking consumer response to their ads through sales. Healton pointed out that few ads for tobacco cessation medicines focus on eliciting fear; instead, they usually are serious, supportive, or humorous.
Hammond added that the different findings on tobacco control message framing may be due to whether they are aimed at preventing smoking versus aimed at prompting people to quit smoking. Even people who smoke
________________
19 EX Pilot Evaluation, Grand Rapids, Michigan, 2007 (unpublished data).
seem to want different types of framing depending on whether they are already engaged in attempting to quit or not. “The warnings that actually motivate them to make the next quit attempt are different than the information that they want when they are ready to quit. Then they want positive encouraging information, as well as concrete information on how to quit,” he said. Toll added that it is very difficult to quit smoking and “an ad is not going to make you quit smoking forever. You also need counseling and medication. The ad is really a bridge to get people into treatment.”
Khuri suggested younger generations may respond better to positive messaging than older generations. “Some data suggest for this newer generation, the more positive, get-your-life back messaging works,” he said. Hammond responded that studies of the graphic warning labels show “what works well, works across groups. The ads that hit, hit your 16-year-old kid and they hit your 68-year-old smoker.”
Stevens concluded the discussion of gain- versus loss-framed messaging by noting, “We have seen it as sort of a push-pull. We do some ads that push you to quit and make it uncomfortable for you to smoke. Other ads say ‘we are here to help.’”
Several workshop participants stressed the important roles that clinicians, clinician organizations, and patients can play in advocating for antismoking policy. Herbst, an oncologist who treats lung cancer patients, stressed, “The best way I can help a patient is for them to never get lung cancer. Once a patient has lung cancer, about half of the time, it’s already metastatic, and despite everything we have, we still can’t cure most of these patients.” McGoldrick pointed out that the Illinois Medical Society was instrumental in the recent passage of a tobacco tax in Illinois. This society had been a reliable campaign supporter of the Illinois House Republicans and lobbied their support for the tax because the alternative was a cut in Medicaid payments to physicians. “That’s why we won in Illinois. It wasn’t because the tobacco tax was going to save lives, it was because the House GOP always sticks with the docs, no matter what,” said McGoldrick. “We have to be strong advocates for these policies. You can have all the science in the world, but if we each individually and organizationally don’t get involved in the policy process, then we are going to suffer the fate we have been suffering the last few years because we have very powerful forces working against us,” he stressed.
Many workshop participants stressed the importance of physicians providing message support in policy campaigns. “It’s not just the messaging, but the messenger,” said McGoldrick, and pointed out that when physicians deliver ad messages, it tends to give the messages more credibility, as indicated by the tobacco company ads aimed at defeating California’s recent attempt to raise the cigarette tax (YouTube, 2012). In these ads, physicians were used to deliver their message.
Sneegas concurred, noting that “defining a comprehensive tobacco control program was made tolerable by the fact that I very frequently had a physician on either side of me, backing me up—it was harder to attack the clinician than the little woman. It was harder to ignore the physician who had a command in the room and whose presence cannot be overstated.” She added that New York has been effective in instituting tobacco control policies, not just because of its state tobacco control program, but because of the strong advocacy network, which includes many physicians.
Physicians and physician organizations can aid tobacco control efforts in many ways, Sneegas pointed out. Ideas include writing letters to the editor of local newspapers; submitting comments to FDA; testifying at hearings; educating relevant boards, councils, and commissions; and passing resolutions through their professional organizations. “Clinicians have a powerful voice and can make such an incredible difference if they testify for policy, taxes, or smoke-free ordinances,” Sneegas said. “The clinician’s voice is a pure voice that is not used nearly enough,” she added.
Many physicians focus their tobacco control efforts on trying to prevent teens from smoking, and Sneegas noted, “If they really want to prevent youth initiation of cigarette smoking or other tobacco product use, then they need to focus on raising the prices of tobacco products, making school campuses tobacco free, ensuring their community is smoke free, and restricting the sale and advertising of tobacco products to children. And of course there’s nothing better to help a kid not to start smoking than to help their family quit smoking.”
She also recommended that everyone monitor tobacco industry promotion and marketing of their products in their local communities. “The next time you buy gas, go inside and look at the array of tobacco products being sold at the station and think about it as if you were looking through the eyes of a child,” Sneegas said.
Sciandra also noted that the burden of lobbying for tobacco control policies tends to fall on organizations such as the American Cancer Society, the Campaign for Tobacco-Free Kids, the American Heart Association, and
the American Lung Association. “In my experience, I am sorry to say,” the medical societies have been unreliable partners in this situation. “They were great in Illinois because it was a pocketbook issue for them and they were definitely champs when we passed the Clean Air Act in 2003, but they have been pretty much AWOL since then and that needs to change because they can be effective as part of a lobbying coalition,” Sciandra said.
Sarna added that clinicians are unclear about their role in policy making and have competing priorities, including a lack of time and a lack of financial benefit in spending time treating patients’ tobacco dependence. “But could you really say that you have no time for someone who is diabetic or suffering from significant high blood pressure?”
Sneegas also noted that nonclinicians also can make an impact in tobacco control efforts. She gave an example of a person who lost her mother to lung cancer “who goes from city to city telling people the story of her mother and why it didn’t have to happen. She does media interviews and she testifies at hearings,” Sneegas said. She also mentioned a lung cancer patient who, while her cancer was in remission, became a spokesperson for tobacco control. “These are the kinds of people in our communities who are willing to help all of us do our job and make a difference,” Sneegas added.
Toll suggested that relevant large professional organizations, such as the American Psychological Association, ASCO, and AACR have a bigger stake in trying to change tobacco policy. “We need advocacy at the highest levels from these organizations,” he said. Cummings agreed, noting that although relevant cancer professional organizations, such as ASCO and AACR have committees on tobacco control, they often do not devote many resources to tobacco control advocacy efforts. “They advocate for other issues regarding cancer treatment and research dollars and they ought to be working in partnership on this issue too,” he said. Warren further suggested that inclusion of tobacco assessment and cessation therapy in clinical practice guidelines such as those of the National Comprehensive Cancer Network (NCCN) would facilitate rapid uptake by clinical oncology providers. Coordinating efforts by a national organization such as NCI could help facilitate agreement on best practices for cancer treatment.
FINANCIAL AND LEGAL CHALLENGES
A number of financial and legal challenges are impeding tobacco control efforts, workshop participants stressed, especially the large amount of money that tobacco companies spend on marketing their products and
on lobbying and legal suits to prevent tobacco control policies from being implemented. The amount of funding for tobacco control marketing and policy is miniscule by comparison. “You have to understand where the financial levers are if you really want to make something happen here,” McGoldrick noted.
He pointed out that the tobacco companies spent $50 million on a media campaign to defeat California’s recent ballot initiative to raise the taxes on cigarettes by $1 per pack (Slosson, 2012). McGoldrick said that amount is more than the CDC spent on its national tobacco control media campaign this past year. Prior to the California antitax media campaign, he said, 65 percent of people polled in the state indicated they supported the bill. But that support dropped to 50 percent on voting day and the tax increase did not pass. Herbst added that “it is interesting to me that, even in California, which is doing so well [in terms of tobacco control policy], they still have to continue to fight and to maintain what they have.” McGoldrick added that despite the decline in state budgets in recent years, which usually inspires bills to increase cigarette taxes, there were no state tobacco tax increases in 2011, and only one passed in 2012, in part because of lobbying efforts opposing such taxes by tobacco companies.
In addition, although state revenues from tobacco taxes have increased from 2007 to 2012 ($22.3 billion to $25.6 billion), the total state spending on tobacco prevention has decreased from $597.5 million to $457 million (Campaign for Tobacco-Free Kids, 2012b,d). Sneegas added that North Carolina abolished the state agency that previously allocated $17 million annually for tobacco control. This year, the state will devote only $5 million for tobacco control, and that will not include any funding for media campaigns.
McAfee expressed concern that given the current budget crisis, states have been less inclined to fund tobacco control efforts. Even though states have increased excise taxes on tobacco products, they have allocated the tobacco product tax revenue to balance their budgets. Khuri concurred, adding, “Most states have used their tobacco settlement money to pay off other debts and are shutting down prevention and scientific programs.”
Even states that earmarked a portion of the money gained from cigarette taxes for tobacco control efforts are finding the funds insufficient. “The buying power of the 5 cents California spends on tobacco control from the cigarette tax on each pack sold is worth substantially less than it was 20 years ago,” Stevens said.
Cummings added that the amount that CDC can spend on tobacco
control efforts is declining as its budget has been decreasing in recent years. No more than 1 percent of CDC’s budget supports the Office on Smoking and Health, he said. The amount of money CDC allocates to fund state tobacco control efforts is a tiny fraction of the CDC-recommended $3.7 billion, McGoldrick stressed. “There is a dose-response relationship between resources and advocacy, just as there is for drugs. We have to do everything in our power to reinvigorate the advocacy infrastructure if we are going to keep making progress on tobacco control policy levers that we know work,” he said.
McAfee noted that the tobacco industry spends 23 times more on tobacco promotion than states do on tobacco control programs—$10.5 billion spent by the industry in 2008 compared to less than $500 million spent at the state level, despite the fact that states collect $25 billion a year from the taxes on tobacco products and the Master Settlement Agreement
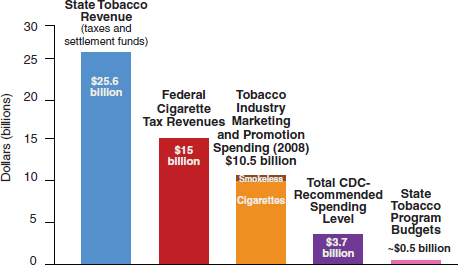
FIGURE 9 The tobacco industry outspends state tobacco prevention efforts by 23 to 1. Although tobacco-related state and federal revenues comprise around $40 billion, state tobacco control program budgets are less than $500 million, which is less than the Centers for Disease Control and Prevention (CDC)-recommended $3.7 billion, and 23 times less than the $10.5 billion spent by the tobacco industry on marketing and promotion.
SOURCES: McAfee presentation (June 11, 2012); Campaign for Tobacco-Free Kids (2012b,d); CDC (2007c).
funds, and the federal government collects an additional $15 billion per year in cigarette tax revenues (see Figure 9).
“The only way we have any chance of even staying equal in the game is if we have resources, because we are David against Goliath,” Stevens stressed, and Fong added, “You don’t want to bring a knife to a gun fight.” McGoldrick pointed out, “When we spend less money on tobacco prevention and cessation, that means more kids are starting to smoke, fewer adults are quitting, more people are dying and more health care dollars are being spent treating tobacco-caused diseases.” Sneegas added, “We know what works, and that state programs will give us a return on investment, but we need the funding in the states in order to make it happen.”
Funding for Tobacco Cessation Programs
Another major financial impediment is lack of funding for tobacco cessation programs. Gritz noted that these programs often rely on hospital budgets and clinical revenue, and therefore are underfunded or never even come into existence because of a lack of funds. Another financial barrier is the frequent lack of insurance reimbursement for tobacco cessation therapy. Although Medicare recently expanded coverage of tobacco cessation therapy to all beneficiaries who use tobacco (CMS, 2012), there is variable coverage for cessation therapy. “This is a real barrier because so many people don’t have coverage,” Hurt noted. “A gold standard institution should provide coverage for [tobacco cessation therapy] for their employees with no copays,” he argued. Given their reimbursement difficulties, Hurt’s institution, Mayo Clinic, established a charitable fund for tobacco cessation therapy that is composed of money donated by grateful patients for others who cannot afford the treatment.
Fiore pointed out that the ACA will expand coverage for smoking cessation counseling and medications20 (see section on Antismoking Laws and Regulations) (Koh et al., 2010). He added that as insurance companies are recognizing the rapid return on investment in tobacco cessation therapy, more are expanding coverage to include this service. He reiterated that Massachusetts has provided evidence that there is a $2 return on investment for every dollar spent within just a few years of implementation of a smoking cessation treatment program (Richard et al., 2012). “There is
________________
20 Patient Protection and Affordable Care Act of 2010, H.R. 3590, 111th U.S. Congress. 2nd Sess. (March 23, 2010).
a powerful evidence base that is moving the private sector to support this. Part of the progress we see in this area rests purely on economic grounds,” he said.
Cummings added, “It is cheaper to treat smoking than to provide $10,000 a month on chemotherapy. It makes you wonder why we are pinching pennies to find treatments for smoking cessation in cancer patients.” This is especially true in light of the estimate by Warner that smoking causes as much as 10 percent of all health care costs in the United States (Warner et al., 1999). Warren noted that his institution, Roswell Park, built a new tobacco cessation program that provides treatment for about 2,000 people a year at a cost of $240,000 per year. “I need to save 80 ICU days in 2,000 smokers per year to break even. I feel comfortable that I am going to reduce those hospitalizations due to fewer wound infections, pulmonary complications, and other complications of treatment,” he said. Cummings pointed out that once treatment programs are established, they can seek insurance reimbursement for their services. Cummings also stressed that until cessation therapy becomes the standard of care for all patients who smoke, insurance reimbursement will not be guaranteed.
There was some discussion on the coding of tobacco cessation therapy for the purposes of billing and reimbursement. According to Cummings, many clinicians do not document the diagnosis of a tobacco use disorder via the International Classification of Diseases, Ninth Edition (ICD-9) code, 301.5. Toll noted that the Yale Cancer Center provides psychotherapy or psychiatric codes for tobacco cessation therapy, and is reimbursed accordingly. But this requires having psychologists, psychiatrists, or other personnel who can be reimbursed for such coded treatment. The 2008 clinical practice guideline provides examples of codes used for the diagnosis and billing of tobacco dependence treatment, and the guideline recognizes that “it is difficult to accurately document and obtain reimbursement for this treatment” (Fiore et al., 2008).
A great deal of discussion focused on the legal challenges tobacco companies pose whenever state or local governments try to enact tobacco control laws. “You have to expect legal challenges and have a good defense for those tobacco control laws that you pass and be prepared for litigation, both psychologically and financially,” McGoldrick advised. He noted that
even if policies have strong legal backing, tobacco companies will still pursue lawsuits so as to stall their implementation.
Cummings pointed out that it is especially difficult for small communities to drum up the financial resources to defend legal challenges to their antismoking ordinances. Some law firms have provided pro bono assistance to such communities, and the Tobacco Control Legal Consortium has provided research, analysis, and legal briefs to assist state and municipal attorneys prepare for these legal challenges, according to McGoldrick. But Sciandra questioned whether government entities can accept charity donations from outside groups to help them fight their legal battles. Matthew Myers, president of the Campaign for Tobacco-Free Kids, and Stevens suggested trying to pass only tobacco control laws in those communities that have access to the resources to defend them, because if the lawsuits prevail, they set legal precedents that can impede other communities from adopting similar laws.
RESEARCH AND INFRASTRUCTURE NEEDS
Several speakers stressed research and infrastructure needs related to reducing tobacco-related cancer incidence and mortality, including a rapid research capability to assess the use and health effects of new tobacco products, more funding for research on lung cancer and on tobacco cessation therapy, more systems integration, and coordination of tobacco assessment in clinical trials of pharmaceutical products and disease processes.
“We need rapid, rigorous, and representative research that is done on a dime,” said Abrams, who suggested more funding for behavioral and social science research on tobacco control. He noted that although it is important to understand the genetic and neurobiological basis for nicotine addiction, such information is just “the sound of one hand clapping when it comes to massive environmental pathogenic exposure. The behavioral sciences that [seek to] understand what we put inside our bodies and why is as important to fund, in terms of making a public health impact.” Stevens also suggested more research on how best to change social norms and implement policies, both of which have contributed substantially to the success of California’s tobacco control efforts. Sciandra endorsed Abrams’ call for rapid research that “can inform appropriate and effective policy responses to industry innovations—it gives us the ammunition we need to convince policy makers.”
A number of speakers suggested that additional research could improve
the delivery of tobacco cessation therapy. Abrams noted that this research could help tailor tobacco cessation medicines so that they are maximally effective for individual patients. Ostroff suggested that research could help determine how to best incorporate CT screening interventions with tobacco cessation therapy.
Several speakers pointed out the need to verify self-reports in studies of tobacco use. Such reports are often inaccurate, but can be verified by assessing cotinine levels in serum, or by taking carbon monoxide measurements with a carbon monoxide meter. Gritz and Warren cited a study demonstrating that cancer patients may not accurately report tobacco use (Warren et al., 2012d), perhaps due to the stigmatization of tobacco.
Others suggested the need to routinely collect data on tobacco use in cancer clinical trials (Gritz et al., 2005). “We need to build a large clinical trials database with the power to assess complications, toxicities, outcomes, recurrences, and second primary cancers in people who smoke so we can convince clinicians, basic scientists, and government funders on the absolute critical importance of tobacco cessation for cancer patients,” Gritz said. Several participants pointed out that NCI Cooperative Group trials often do not assess tobacco use; of 155 clinical trials reviewed, only 29.4 percent assessed any form of tobacco use at enrollment, 4.5 percent assessed tobacco use at follow-up, and no trials assessed tobacco cessation or nicotine addiction (Peters et al., 2012). “Even though tobacco changes drug metabolism, tumor physiology, and response to treatment, we will not have a good understanding of how tobacco affects the outcomes of our cutting edge cancer research,” said Warren. He suggested there may be support for determining the appropriate metrics to document in this regard from the leadership at NCI and other organizations.
Gritz also suggested conducting more prospective, randomized controlled trials to establish optimal cessation regimens and timing of treatment. Cummings noted that although tobacco use causes 30 percent of all cancer deaths, NCI devotes only about 4 percent of its budget to tobacco control research. “This is a big problem if we are going to win the war on cancer. We have to get more grants focused in this area,” he said. Khuri also called for more support for research on lung cancer. He said that even though lung cancer accounts for more deaths than colorectal, breast, and prostate cancers combined, it receives much fewer research dollars than any of these cancers (see Figure 10).
Abrams called for more integration of tobacco control efforts and oversight. He pointed out that even within FDA, one Center regulates
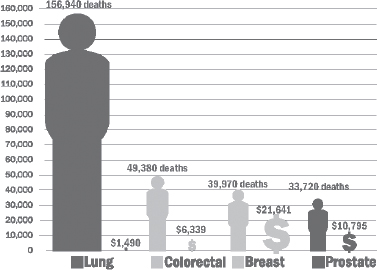
FIGURE 10 U.S. cancer deaths versus federal research funding per death. Although lung cancer accounts for more deaths than breast, prostate, and colorectal cancers combined, proportionately it receives fewer research dollars per death than these other cancers.
NOTES: Estimated deaths by cancer type in the United States for 2011 are from ACS (2011). Annual funding figures represent the National Institutes of Health (NIH, 2012) and Department of Defense (CDMRP, 2012) estimated 2011 spending. Although the Centers for Disease Control and Prevention also funds cancer research, a breakdown of spending between research and prevention is not currently available.
SOURCE: Khuri presentation (June 12, 2012); reprinted with permission from the National Lung Cancer Partnership.
tobacco products, while another regulates nicotine replacement treatments, and the degree of regulation is not equal between the two Centers. He also envisions more comprehensive treatment and continuity of care for patients who smoke, similar to the goal for patients with diabetes. But that requires integrating public health models into a health care delivery system that is largely modeled on the individual patient in the clinic, he said.
Several people spoke about the benefits of having electronic medical records. Land said he was asked by the Office of the National Coordinator for Health Information Technology to evaluate the patient-centered medical home program in Massachusetts to determine if there are demonstrable effects of having large interconnected data systems for tobacco control and beyond. He suggested stressing the more widespread benefits of such systems so they are more likely to be adopted by institutions.
Before adjourning the workshop, Herbst urged participants to take action to address the needs identified at the workshop, with both short-term and long-term strategies. Myers noted that a recurring theme was that much is known about how to effectively counter promotion of tobacco products and encourage people to quit smoking, but that knowledge is not being applied because a lack of resources and political will. He also stressed that the cancer research and clinician communities are not engaged as fully as they should be in tobacco control advocacy, as well as in assessing tobacco use in their patients and offering tobacco cessation therapy. “If we don’t engage then it is not the tobacco industry we have to blame, but rather ourselves,” he said. “Unless we can get the practitioners who see people dying of cancer every day to treat tobacco control with the seriousness it deserves, and to take advantage of the tools we have and we know work, we are not going to win the war on cancer.”
Abroms, L. C., N. Padmanabhan, L. Thaweethai, and T. Phillips. 2011. iPhone apps for smoking cessation. American Journal of Preventive Medicine 40(3):279-285.
ACS (American Cancer Society). 2011. Cancer facts & figures 2011. http://www.cancer.org/Research/CancerFactsFigures/CancerFactsFigures/cancer-facts-figures-2011 (accessed September 10, 2012).
ACS. 2012. Cancer facts & figures 2012. http://www.cancer.org/acs/groups/content/@epidemiologysurveilance/documents/document/acspc-031941.pdf (accessed August 21, 2012).
ACS, CDPH (California Department of Public Health), and CCR (California Cancer Registry). 2011. California cancer facts and figures 2012. Oakland, CA: American Cancer Society, California Division.
ACSCAN (American Cancer Society Cancer Action Network). 2012. Rockland Co. village bans tobacco marketing aimed at kids. Point of sale displays an important cause of youth smoking. http://www.acscan.org/action/ny/updates/1931/ (accessed September 7, 2012).
American Lung Association. 2011. Helping smokers quit: Tobacco cessation coverage 2011. http://www.lung.org/assets/documents/publications/smoking-cessation/helping-smokers-quit-2011.pdf (accessed September 10, 2012).
Anderson, C. M., R. Yip, C. I. Henschke, D. F. Yankelevitz, J. S. Ostroff, and D. M. Burns. 2009. Smoking cessation and relapse during a lung cancer screening program. Cancer Epidemiology Biomarkers & Prevention 18(12):3476-3483.
ASCO (American Society of Clinical Oncology). 2003. American Society of Clinical Oncology policy statement update: Tobacco control—reducing cancer incidence and saving lives. Journal of Clinical Oncology 21(14):1-10.
Bastian, L. A., and S. E. Sherman. 2010. Effects of the wars on smoking among veterans. Journal of General Internal Medicine 25(2):102-103.
Bauer, J. E., A. Hyland, Q. Li, C. Steger, and K. M. Cummings. 2005. A longitudinal assessment of the impact of smoke-free worksite policies on tobacco use. American Journal of Public Health 95(6):1024-1029.
Bayer, R., L. Gostin, and D. Marcus-Toll. 2012. Repackaging cigarettes—will the courts thwart the FDA? New England Journal of Medicine 367(22):2065-2067.
Boorjian, S., J. E. Cowan, B. R. Konety, J. DuChane, A. Tewari, P. R. Carroll, C. J. Kane, and Cancer of the Prostate Strategic Urologic Research Endeavor Investigators. 2007. Bladder cancer incidence and risk factors in men with prostate cancer: Results from Cancer of the Prostate Strategic Urologic Research Endeavor. Journal of Urology 177(3):883-887.
Breast Cancer Trialists Group. 1998. Polychemotherapy for early breast cancer: An overview of the randomised trials. Early Breast Cancer Trialists’ Collaborative Group. Lancet 352(9132):930-942.
Brody, A. L., M. A. Mandelkern, E. D. London, R. E. Olmstead, J. Farahi, D. Scheibal, J. Jou, V. Allen, E. Tiongson, S. I. Chefer, A. O. Koren, and A. G. Mukhin. 2006. Cigarette smoking saturates brain α4 β2 nicotinic acetylcholine receptors. Archives of General Psychiatry 63(8):907-915.
Browman, G. P., G. Wong, I. Hodson, J. Sathya, R. Russell, L. McAlpine, P. Skingley, and M. N. Levine. 1993. Influence of cigarette smoking on the efficacy of radiation therapy in head and neck cancer. New England Journal of Medicine 328(3):159-163.
Brown, D. W. 2009. Smoking prevalence among US veterans. Journal of General Internal Medicine 25(2):147-149.
California State Board of Equalization. 2012. Annual report 2010-2011: Appendix. http://www.boe.ca.gov/annual/2010-11/appendix.html (accessed September 4, 2012).
California Tobacco Control Program. 2010. Two decades of the California Tobacco Control Program: California Tobacco Survey, 1990-2008. http://www.cdph.ca.gov/programs/tobacco/Documents/CDPH_CTS2008%20summary%20report_final.pdf (accessed September 4, 2012).
Callinan, J. E., A. Clarke, K. Doherty, and C. Kelleher. 2010. Legislative smoking bans for reducing secondhand smoke exposure, smoking prevalence and tobacco consumption (review). Cochrane Database of Systematic Reviews 14(4):CD005992.
Campaign for Tobacco-Free Kids. 2012a. The impact of the new FDA tobacco law on state tobacco control efforts. http://www.tobaccofreekids.org/research/factsheets/pdf/0360.pdf (accessed September 7, 2012).
Campaign for Tobacco-Free Kids. 2012b. Spending vs. tobacco company marketing. http://www.tobaccofreekids.org/content/what_we_do/state_local_issues/settlement/FY2012/10_State_Tobacco_Prevention_Spending_vs_Tob_Co._Marketing.pdf (accessed September 10, 2012).
Campaign for Tobacco-Free Kids. 2012c. Enforcing laws prohibiting cigarette sales to kids reduces youth smoking. http://www.tobaccofreekids.org/research/factsheets/pdf/0049.pdf (accessed September 10, 2012).
Campaign for Tobacco-Free Kids. 2012d. Trends in state tobacco prevention spending vs. state tobacco revenues. http://www.tobaccofreekids.org/research/factsheets/pdf/0220.pdf (accessed September 11, 2012).
Campaign for Tobacco-Free Kids, Counter Tobacco, and the American Heart Association. 2012. Deadly alliance: How big tobacco and convenience stores partner to market tobacco products and fight life-saving policies. http://www.tobaccofreekids.org/content/what_we_do/industry_watch/store_report/deadlyalliance_full_report.pdf (accessed September 10, 2012).
CDC (Centers for Disease Control and Prevention). 2007a. Cigarette smoking among adults—United States, 2006. Morbidity and Mortality Weekly Report 56(44):1157-1161.
CDC. 2007b. Decline in smoking prevalence—New York City, 2002-2006. Morbidity and Mortality Weekly Report 56(24):604-608.
CDC. 2007c. Best practices for comprehensive tobacco control programs—2007. http://www.cdc.gov/tobacco/stateandcommunity/best_practices/index.htm (accessed December 10, 2012).
CDC. 2008. Smoking-attributable mortality, years of potential life loss, and productivity losses—United States, 2000-2004. Morbidity and Mortality Weekly Report 57(45):1226-1228.
CDC. 2010. Smoking & tobacco use: Regulation. http://www.cdc.gov/tobacco/data_statistics/by_topic/policy/regulation/index.htm (accessed September 5, 2012).
CDC. 2011a. Current cigarette smoking prevalence among working adults—United States, 2004-2010. Morbidity and Mortality Weekly Report 60(38):1305-1309.
CDC. 2011b. Smoking & tobacco use. Hookahs. http://www.cdc.gov/tobacco/data_statistics/fact_sheets/tobacco_industry/hookahs (accessed August 23, 2012).
CDC. 2011c. Quitting smoking among adults—United States, 2001-2010. Morbidity and Mortality Weekly Report 60(44):1513-1519.
CDC. 2011d. Vital signs: Current cigarette smoking among adults aged ≥18 years—United States, 2005-2010. Morbidity and Mortality Weekly Report 60(35):1207-1212.
CDC. 2012a. Consumption of cigarettes and combustible tobacco—United States, 2000-2011. Morbidity and Mortality Weekly Report 61(30):565-569.
CDC. 2012b. Current tobacco use among middle and high school students—United States, 2011. Morbidity and Mortality Weekly Report 61(31):581-585.
CDC. 2012c. Tips from former smokers. http://www.cdc.gov/tobacco/campaign/tips/ (accessed September 5, 2012).
CDC. 2012d. Youth risk behavior surveillance—United States, 2011. Morbidity and Mortality Weekly Report 61(SS04):1-162.
CDC. 2012e. Tobacco use among Iraq- and Afghanistan-era veterans: A qualitative study of barriers, facilitators, and treatment preferences. Preventing Chronic Disease 9:110131.
CDC. 2012f. Adolescent and school health: Youth risk behavior survey fact sheets. http://www.cdc.gov/healthyyouth/yrbs/factsheets/index.htm (accessed September 7, 2012).
CDC. 2012g. Figure 8.1. Prevalence of current smoking among adults aged 18 and over: United States, 1997-2011. http://www.cdc.gov/nchs/data/nhis/earlyrelease/earlyrelease201206_08.pdf (accessed October 5, 2012).
CDMRP (Congressionally Directed Medical Research Programs). 2012. Research programs. http://cdmrp.army.mil/researchprograms.shtml (accessed September 10, 2012).
Chaloupka, F. J. 2010. Tobacco control lessons learned: The impact of state and local policies. ImpacTeen Research Paper No. 38. Chicago, IL: Health Policy Center, Institute for Health Research and Policy, University of Illinois at Chicago.
Chapple, A., S. Ziebland, and A. McPherson. 2004. Stigma, shame, and blame experienced by patients with lung cancer: Qualitative study. British Medical Journal 328(7454):1470.
Cleveland Clinic. 2012a. A message about smoking. http://my.clevelandclinic.org/tobacco/a_message_about_smoking.aspx (accessed September 10, 2012).
Cleveland Clinic. 2012b. New nonsmoking hiring policy at Cleveland Clinic. http://my.clevelandclinic.org/Documents/Urology/Non-Smoking_Hiring_Statement.pdf (accessed September 10, 2012).
CMS (Centers for Medicare & Medicaid Services). 2011. Essential health benefits bulletin. Center for Consumer Information and Insurance Oversight. http://cciio.cgov/resources/files/Files2/12162011/essential_health_benefits_bulletin.pdf (accessed September 7, 2012).
CMS. 2012. Smoking & tobacco use cessation (counseling to stop smoking or using tobacco products). http://www.medicare.gov/coverage/smoking-and-tobacco-use-cessation.html (accessed September 7, 2012).
Conroy, M. B., N. E. Majchrzak, C. B. Silverman, Y. Chang, S. Regan, L. I. Schneider, and N. A. Rigotti. 2005. Measuring provider adherence to tobacco treatment guidelines: A comparison of electronic medical record review, patient survey, and provider survey. Nicotine and Tobacco Research 7(S1):S35-S43.
Cooley, M. E., L. Sarna, J. Kotlerman, J. M. Lukanich, M. Jaklitsch, S. B. Green, and R. Bueno. 2009. Smoking cessation is challenging even for patients recovering from lung cancer surgery with curative intent. Lung Cancer 66(2):218-225.
CTIA. 2011. CTIA—the Wireless Association semi-annual survey reveals historical wireless trend. http://www.ctia.org/media/press/body.cfm/prid/2133 (accessed September 4, 2012).
Dennis, B. 2013. Government quits legal battle over graphic cigarette warnings. http://articles.washingtonpost.com/2013-03-19/national/37845669_1_cigarette-makers-tobacco-companies-confusion-or-deception (accessed April 4, 2013).
Dobbins, M., K. DeCorby, S. Manske, E. Goldblatt. 2008. Effective practices for school-based tobacco use prevention. Preventive Medicine 46(4):289-297.
Dooren, J. C. 2012. Court strikes graphic cigarette labels. http://online.wsj.com/article/SB10000872396390444358404577609242156512180.html (accessed October 6, 2012).
Duffy, S. A., D. L. Ronis, M. Valenstein, M. T. Lambert, K. E. Fowler, L. Gregory, C. Bishop, L. L. Myers, F. C. Blow, and J. E. Terrell. 2006. A tailored smoking, alcohol, and depression intervention for head and neck cancer patients. Cancer Epidemiology, Biomarkers & Prevention 15(11):2203-2208.
Emery, S., Y. Kim, Y. K. Choi, G. Szczyka, M. Wakefield, and F. J. Chaloupka. 2011. The effects of smoking-related television advertising on smoking and intentions to quit among adults in the United States: 1999-2007. American Journal of Public Health 102(4):751-757.
European Commission. 2009. Survey on tobacco: Analytical report. http://ec.europa.eu/public_opinion/flash/fl_253_en.pdf (accessed September 5, 2012).
Farrelly, M. C., K. C. Davis, M. L. Haviland, P. Messeri, and C. G. Healton. 2005. Evidence of a dose—response relationship between “truth” antismoking ads and youth smoking prevalence. American Journal of Public Health 95(3):425-431.
Farrelly, M. C., J. Nonnemaker, K. C. Davis, and A. Hussin. 2009. The influence of the national truth campaign on smoking initiation. American Journal of Preventive Medicine 6(5):379-384.
FDA (Food and Drug Administration). 2012. Overview of the Family Smoking Prevention and Tobacco Control Act: Consumer fact sheet. http://www.fda.gov/TobaccoProducts/GuidanceComplianceRegulatoryInformation/ucm246129.htm (accessed September 10, 2012).
Felberbaum, M. 2011. Philip Morris Int’l buys rights to nicotine system. http://seattletimes.com/html/businesstechnology/2015155221_apusphilipmorrisintlnicotinedelivery.html (accessed September 13, 2012).
Ferry, L. H., L. M. Grissino, and P. S. Runfola. 1999. Tobacco dependence curricula in U.S. undergraduate medical education. Journal of the American Medical Association 282(9):825-829.
Fichtenberg, C. M., and S. A. Glantz. 2002. Effect of smoke-free workplaces on smoking behaviour: Systematic review. British Medical Journal 325(7357):188.
Fiore, M. C., and T. B. Baker. 2011. Treating smokers in the health care setting. New England Journal of Medicine 365(13):1222-1231.
Fiore, M. C., C. R. Jaen, T. B. Baker, W. C. Bailey, N. L. Benowitz, S. J. Curry, S. F. Dorfman, E. S. Froelicher, M. G. Goldstein, C. G. Healton, P. N. Henderson, R. B. Heyman, H. K. Koh, T. E. Kottke, H. A. Lando, R. E. Mechlenburg, R. J. Mermelstein, P. D. Mullen, C. T. Orleans, L. Robinson, M. L. Stitzer, A. C. Tommasello, L. Villejo, and M. E. Wewers. 2008. Treating tobacco use and dependence: 2008 update. Clinical Practice Guideline. Rockville, MD: U.S. Public Health Service.
Fiore, M. C., E. Goplerud, and S. A. Schroeder. 2012. The Joint Commission’s new tobacco-cessation measures—will hospitals do the right thing? New England Journal of Medicine 366(13):1172-1174.
Flanders, W. D., C. A. Lally, B. P. Zhu, S. J. Henley, and M. J. Thun. 2003. Lung cancer mortality in relation to age, duration of smoking, and daily cigarette consumption: Results from Cancer Prevention Study II. Cancer Research 63(19):6556-6562.
Free, C., R. Knight, S. Robertson, R. Whittaker, P. Edwards, W. Zhou, A. Rodgers, J. Cairns, M. G. Kenward, and I. Roberts. 2011. Smoking cessation support delivered via mobile phone text messaging (txt2stop): A single-blind, randomised trial. Lancet 378(9785):49-55.
Freeman, B., and S. Chapman. 2012. Measuring interactivity on tobacco control websites. Journal of Health Communication 17(7):857-865.
Fucito, L. M., A. E. Latimer, P. Salovey, and B. A. Toll. 2010. Nicotine dependence as a moderator of message framing effects on smoking cessation outcomes. Annals of Behavioral Medicine 39(3):311-317.
Gallup. 2008. Tobacco and smoking. http://www.gallup.com/poll/1717/tobacco-smoking.aspx (accessed September 7, 2012).
Gilbert, S. 2012. Village of Haverstraw passes law rescinding tobacco advertising ban, discusses zoning and other regular business. http://www.rocklandtimes.com/2012/08/16/village-of-haverstraw-passes-law-rescinding-tobacco-advertising-ban-discusses-zoning-and-other-regular-business/ (accessed September 7, 2012).
Giovino, G. A. 2007. The tobacco epidemic in the United States. American Journal of Preventive Medicine 33(6S):S318-S326.
Goldstein, A. O., C. E. Ripley-Moffitt, D. E. Pathman, and K. M. Patsakham. 2013. Tobacco use treatment at the U.S. National Cancer Institute’s Designated Cancer Centers. Nicotine and Tobacco Research 15(1)52-58.
Goodall, C., and O. Appiah. 2008. Adolescents’ perceptions of Canadian cigarette package warning labels: Investigating the effects of message framing. Health Communication 23(2):117-127.
Gray, L., and L. Lewis. 2009. Educational technology in public school districts: Fall 2008 (NCES 2010-003). Washington, DC: National Center for Education Statistics (accessed September 21, 2012).
Grimshaw, G.M., and A. Stanton. 2006. Tobacco cessation interventions for young people. Cochrane Database of Systematic Reviews Oct 18(4):CD003289.
Gritz, E. R., C. Dresler, and L. Sarna. 2005. Smoking, the missing drug interaction in clinical trials: Ignoring the obvious. Cancer Epidemiology, Biomarkers & Prevention 14(10):2287-2293.
Gritz, E. R., C. Y. Lam, D. J. Vidrine, and M. C. Fingeret (Eds.). 2011. Cancer prevention: Tobacco dependence and its treatment. In Cancer: Principles & practice of oncology, edited by V. T. DeVita, T. S. Lawrence, and S. A. Rosenberg. Philadelphia, PA: Wolters Kluwer Lippincott Williams & Wilkins.
Hackshaw, A. K., M. R. Law, and N. J. Wald. 1997. The accumulated evidence on lung cancer and environmental tobacco smoke. British Medical Journal 315(7114):980-988.
Hahn, E. J., M. K. Rayens, C. Hopenhayn, and W. J. Christian. 2006. Perceived risk and interest in screening for lung cancer among current and former smokers. Research in Nursing and Health 29(4):359-370.
Hammond, D. 2011. Health warning messages on tobacco products: A review. Tobacco Control 20:327-337.
Hammond, D., G. T. Fong, A. McNeill, R. Borland, and K. M. Cummings. 2006. Effectiveness of cigarette warning labels in informing smokers about the risks of smoking: Findings from the International Tobacco Control (ITC) Four Country Survey. Tobacco Control 15(Suppl III):iii19-iii25.
Herbst, R. S., D. Prager, R. Hermann, L. Fehrenbacher, B. E. Johnson, A. Sandler, M. G. Kris, H. T. Tran, P. Klein, X. Li, D. Ramies, D. H. Johnson, and V. A. Miller. 2005. TRIBUTE: A phase III trial of erlotinib hydrochloride (OSI-774) combined with carboplatin and paclitaxel chemotherapy in advanced non-small-cell lung cancer. Journal of Clinical Oncology 23(25):5892-5899.
Herbst, R. S., J. V. Heymach, and S. M. Lippman. 2008. Molecular origins of lung cancer. New England Journal of Medicine 359(13):1367-1380.
HHS (U.S. Department of Health and Human Services). 2004. The health consequences of smoking: A report of the Surgeon General. Atlanta, GA: Office on Smoking and Health.
HHS. 2008. Helping smokers quit: A guide for clinicians. http://www.ahrq.gov/clinic/tobacco/clinhlpsmksqt.pdf (accessed September 12, 2012).
HHS. 2010a. Ending the tobacco epidemic: A tobacco control strategic action plan for the U.S. Department of Health and Human Services. Washington, DC: Office of the Assistant Secretary for Health.
HHS. 2010b. How tobacco smoke causes disease: The biology and behavioral basis for smoking-attributable disease. A Report of the Surgeon General. Atlanta, GA: Office on Smoking and Health.
HHS. 2012. Preventing tobacco use among youth and young adults: A report of the Surgeon General. Atlanta, GA: Office on Smoking and Health.
Hughes, J. R. 2009. How confident should we be that smoking cessation treatments work? Addiction 104(10):1637-1640.
Hurt, R. D., and C. R. Robertson. 1998. Prying open the door to the tobacco industry’s secrets about nicotine: The Minnesota Tobacco Trial. Journal of the American Medical Association 280(13):1173-1181.
IOM (Institute of Medicine). 2009a. Combating tobacco use in military and veteran populations. Washington, DC: The National Academies Press.
IOM. 2009b. Secondhand smoke exposure and cardiovascular effects: Making sense of the evidence. Washington, DC: The National Academies Press.
Juster, H. R., B. R. Loomis, T. M. Hinman, M. C. Farrelly, A. Hyland, U. E. Bauer, and G. S. Birkhead. 2007. Declines in hospital admissions for acute myocardial infarction in New York state after implementation of a comprehensive smoking ban. American Journal of Public Health 97(11):2035-2039.
Kaufman, E. L., J. S. Jacobson, D. L. Hershman, M. Desai, A.I. Neugut. 2008. Effect of breast cancer radiotherapy and cigarette smoking on risk of second primary lung cancer. Journal of Clinical Oncology 26(3):392-398.
King, B. A., K. M. Cummings, M. C. Mahoney, H. R. Juster, and A. J. Hyland. 2010. Multiunit housing residents’ experiences and attitudes toward smoke-free policies. Nicotine & Tobacco Research 12(6):598-605.
Kirby, T. 2012. Plain cigarette packaging: Australia’s victory. Lancet Aug 24. [E-pub ahead of print.]
Koh, H. K., and K. G. Sebelius. 2010. Promoting prevention through the Affordable Care Act. New England Journal of Medicine 363(14):1296-1299.
Land, T., N. A. Rigotti, D. E. Levy, M. Paskowsky, D. Warner, J.-A. Kwass, L. Wetherell, and L. Keithly. 2010a. A longitudinal study of Medicaid coverage for tobacco dependence treatments in Massachusetts and associated decreases in hospitalizations for cardiovascular disease. PLoS Medicine 7(12):e1000375.
Land, T., D. Warner, M. Paskowsky, A. Cammaerts, L. Wetherell, R. Kaufmann, L. Zhang, A. Malarcher, T. Pechacek, and L. Keithly. 2010b. Medicaid coverage for tobacco dependence treatments in Massachusetts and associated decreases in smoking prevalence. PLoS ONE 5(3):e9770.
Land, T., N. Rigotti, D. Levy, T. Schilling, D. Warner, and W. Li. 2012. The effect of systematic clinical interventions with cigarette smokers on quit status and the rates of smoking-related primary care office visits. PLoS ONE 7(7):e41649.
Lasser, K., W. Boyd, S. Woolhandler, D. U. Himmelstein, D. McCormick, and D. H. Bor. 2000. Smoking and mental illness: A population-based prevalence study. Journal of the American Medical Association 284(20):2606-2610.
Legacy. 2012. truth. http://www.legacyforhealth.org/28.aspx (accessed September 5, 2012).
Lightwood, J. M., A. Dinno, and S. A. Glantz. 2008. Effect of the California Tobacco Control Program on personal health care expenditures. PLoS Medicine 5(8):1214-1222.
Lorigan, P., J. Radford, A. Howell, and N. Thatcher. 2005. Lung cancer after treatment for Hodgkin’s lymphoma: a systematic review. Lancet Oncology 6(10):773-779.
Lorjian, S., J. E. Cowan, B. R. Konety, J. DuChane, A.Tewari, P. R. Carroll, C. J. Kane. 2007. Cancer of the Prostate Strategic Urologic Research Endeavor Investigators. Bladder cancer incidence and risk factors in men with prostate cancer: results from Cancer of the Prostate Strategic Urologic Research Endeavor. Journal of Urology 177(3):883-887.
McClave, A. K., L. R. McKnight-Eily, S. P. Davis, and S. R. Dube. 2010. Smoking characteristics of adults with selected lifetime mental illnesses: Results from the 2007 National Health Interview Survey. American Journal of Public Health 100(12):2464-2472.
Moorman, M., and B. van den Putte. 2008. The influence of message framing, intention to quit smoking, and nicotine dependence on the persuasiveness of smoking cessation messages. Addictive Behaviors 33(10):1267-1275.
Morgan, G., R. A. Schnoll, C. M. Alfano, S. E. Evans, A. Goldstein, J. Ostroff, E. R. Park, L. Sarna, and L. S. Cox. 2011. National Cancer Institute Conference on Treating Tobacco Dependence at Cancer Centers. Journal of Oncology Practice 7(3):178-182.
NAQC (North American Quitline Consortium). 2012. What is a quitline? http://www.naquitline.org/?page=whatisquitline (accessed October 5, 2012).
New York City Department of Health and Mental Hygiene. 2012. Youth risk behavior survey. https://a816-healthpsi.nyc.gov/SASStoredProcess/guest?_PROGRAM=%2FEpiQuery%2FYRBS%2Fyrbsindex (accessed December 10, 2012).
NCI (National Cancer Institute). 2007. Promoting healthy lifestyles: Policy, program, and personal recommendations for reducing cancer risk. 2006-2007 annual report: President’s Cancer Panel. Bethesda, MD: National Cancer Institute.
NCI. 2008. Tobacco Control Monograph 19: The role of the media in promoting and reducing tobacco use. http://cancercontrol.cancer.gov/tcrb/monographs/19/index.html (accessed September 27, 2012.
NCI. 2012a. Monograph 9: Cigars: Health effects and trends. http://www.cancercontrol.cancer.gov/tcrb/monographs/9/index.html (accessed September 12, 2012).
NCI. 2012b. Smoking cessation and continued risk in cancer patients (PDQ®). http://www.cancer.gov/cancertopics/pdq/supportivecare/smokingcessation/HealthProfessional/page3 (accessed September 13, 2012).
New York State Office of Alcoholism and Substance Abuse Services. 2008. Tobacco-free services Title 14 NYCRR Part 856. http://www.oasas.ny.gov/regs/856.cfm (accessed September 7, 2012).
New York State Tobacco Control Program. 2010. Leading the way toward a tobacco-free society: 2010-2013. http://www.health.ny.gov/prevention/tobacco_control/docs/leading_the_way_2010-2013.pdf (accessed September 7, 2012).
NIH (National Institutes of Health). 2012. Estimates of funding for various research, condition, and disease categories (RCDC). http://report.nih.gov/categorical_spending.aspx (accessed September 11, 2012).
Oncology Nursing Society. 2008. Nursing leadership in global and domestic tobacco control. http://www.ons.org/Publications/Positions/Tobacco (accessed September 7, 2012).
Ostroff, J., N. Buckshee, C. A. Mancuso, D. F. Yankelevitz, C. I. Henschke. 2001. Smoking cessation following CT screening for early detection of lung cancer. Preventive Medicine 33(6):613-621.
Peters, E. N., E. Torres, B. A. Toll, K. M. Cummings, E. R. Gritz, A. Hyland, R. S. Herbst, J. R. Marshall, and G. W. Warren. 2012. Tobacco assessment in actively accruing National Cancer Institute Cooperative Group Program clinical trials. Journal of Clinical Oncology 30(23):2869-2875.
Pierce, J. P., K. Messer, M. M. White, D. W. Cowling, and D. P. Thomas. 2011. Prevalence of heavy smoking in California and the United States, 1965-2007. Journal of the American Medical Association 305(11):1106-1112.
Prochaska, J. J., C. Pechmann, R. Kim, and J. M. Leonhardt. 2012. Twitter=quitter? An analysis of Twitter quit smoking social networks. Tobacco Control 21(4):447-449.
Prokhorov, A. V., S. H. Kelder, R. Shegog, N. Murray, J. R. Peters, C. Aqurcia-Parker, P. M. Cinciripini, C. de Moor, J. L. Conroy, K. S. Hudmon, and S. Marani. 2008. Impact of A Smoking Prevention Interactive Experience (ASPIRE), an interactive, multimedia smoking prevention and cessation curriculum for culturally diverse high-school students. Nicotine and Tobacco Research 10(9):1477-1485.
Prokhorov, A. V., K. S. Hudmon, S. Marani, L. Foxhall, K. H. Ford, N. S. Luca, D. W. Wetter, S. B. Cantor, F. Vitale, and E. R. Gritz. 2010. Engaging physicians and pharmacists in providing smoking cessation counseling. Archives of Internal Medicine 170(18):1640-1646.
Raiff, B. R., B. P. Jarvis, and D. Rapoza. 2012. Prevalence of video game use, cigarette smoking, and acceptability of a video game-based smoking cessation intervention among online adults. Nicotine and Tobacco Research 14(12)1453-1457.
Rath, J. M., A. C. Villanti, D. B. Abrams, and D. M. Vallone. 2012. Patterns of tobacco use and dual use in U.S. young adults: The missing link between youth prevention and adult cessation. Journal of Environmental and Public Health May 14. [E-pub ahead of print.]
Rice, V. H., and L. F. Stead. 2008. Nursing interventions for smoking cessation. Cochrane Database of Systematic Reviews 23(1):CD001188.
Richard, P., K. West, and L. Ku. 2012. The return on investment of a Medicaid tobacco cessation program in Massachusetts. PLoS One 7(1):e29665.
Richardson, C. G., L. Vettese, S. Sussman, S. P. Small, and P. Selby. 2011. An investigation of smoking cessation video content on YouTube. Substance Use & Misuse 46(7):893-897.
Saffer, H., and F. Chaloupka. 2000. The effect of tobacco advertising bans on tobacco consumption. Journal of Health Economics 19(6):1117-1137.
Sarna, L., S. A. Bialous, M. E. Wewers, E. S. Froelicher, and L. Danao. 2005. Nurses, smoking, and the workplace. Research in Nursing & Health 28(1):79-90.
Sarna, L., S. A. Bialous, M. Wells, J. Kotlerman, M. E. Wewers, and E. S. Froelicher. 2009. Frequency of nurses’ smoking cessation interventions: Report from a national survey. Journal of Clinical Nursing 18(14):2066-2077.
Sarna, L., S. A. Bialous, M. Ong, M. Wells, and J. Kotlerman. 2012. Nurses’ treatment of tobacco dependence in hospitalized smokers in three states. Research in Nursing & Health 35(3):250-264.
Secretan, B., K. Straif, R. Baan, Y. Grosse, F. El Ghissassi, V. Bouvard, L. Benbrahim-Tallaa, N. Guha, C. Freeman, L. Galichet, and V. Cogliano. 2009. A review of human carcinogens—Part E: Tobacco, areca nut, alcohol, coal smoke, and salted fish. Lancet Oncology 10(11):1033-1034.
Shanahan, P., and D. Elliott. 2009. Evaluation of the effectiveness of the graphic health warnings on tobacco product packaging 2008. Canberra, Australia: Australian Government Department of Health and Ageing.
Shanker, T. 2010. Navy bans tobacco use on its submarine fleet. New York Times, June 21, A16. http://www.nytimes.com/2010/06/21/us/21smoking.html (accessed August 23, 2012).
Sherman, E. J., and B. A. Primack. 2009. What works to prevent adolescent smoking? A systematic review of the National Cancer Institute’s Research-Tested Intervention Programs. Journal of School Health 79(9):391-399.
Slosson, M. 2012. California tobacco tax hike narrowly defeated at polls. http://www.reuters.com/article/2012/06/22/us-usa-campaign-tobacco-idUSBRE85L1F320120622 (accessed September 7, 2012).
Stark, J. 2011. Now butt out: A new push seeks to outlaw cigarettes. http://www.theage.com.au/victoria/now-butt-out-new-push-seeks-to-outlaw-cigarettes-20110521-1ey2s.html (accessed September 10, 2012).
Takeshima, Y., T. Seyama, W. P. Bennett, M. Akiyama, S. Tokuoka, K. Inai, K. Mabuchi, C. E. Land, and C. C. Harris. 1993. p53 mutations in lung cancers from non-smoking atomic-bomb survivors. Lancet 342:1520-1521.
Thatcher, N., A. Chang, P. Parikh, J. Rodrigues Pereira, T. Ciuleanu, J. von Pawel, S. Thongprasert, E. H. Tan, K. Pemberton, V. Archer, and K. Carroll. 2005. Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: Results from a randomised, placebo-controlled, multicentre study (Iressa Survival Evaluation in Lung Cancer). Lancet 366(9496):1527-1537.
Tobacco Control Legal Consortium. 2006. The verdict is in: Findings from United States v. Philip Morris. The hazards of smoking. Saint Paul, MN: Tobacco Control Legal Consortium.
Toll, B. A., S. S. O’Malley, N. A. Katulak, R. Wu, J. Dubin, A. Latimer, B. Meandzija, T. P. George, P. Jatlow, J. L. Cooney, and P. Salovey. 2007. Comparing gain- and loss-framed messages for smoking cessation with bupropion: A randomized controlled trial. Psychology of Addictive Behaviors 21(4):534-544.
Toll, B. A., S. Martino, A. Latimer, P. Salovey, S. O’Malley, S. Carlin-Menter, J. Hopkins, R. Wu, P. Celstino, and K. M. Cummings. 2010. Randomized trial: Quitline specialist training in gain-framed vs. standard-care messages for smoking cessation. Journal of the National Cancer Institute 102(2):96-106.
Tong, E. K., R. Strouse, J. Hall, M. Kovac, and S. A. Schroeder. 2010. National survey of U.S. health professionals’ smoking prevalence, cessation practices, and beliefs. Nicotine and Tobacco Research 12(7):724-733.
Travis, L. B., M. Gospodarowicz, R. E. Curtis, E. A. Clarke, M. Andersson, B. Glimelius, T. Joensuu, C. F. Lynch, F. E. van Leeuwen, E. Holowaty, H. Storm, I. Glimelius, E. Pukkala, M. Stovall, J. F. Fraumeni, Jr., J. D. Boice, Jr., and E. Gilbert. 2002. Lung cancer following chemotherapy and radiotherapy for Hodgkin’s disease. Journal of the National Cancer Institute 94(3):182-192.
TRICARE. 2009. Department of Defense survey of health related behaviors among active duty military personnel: A component of the Defense Lifestyle Assessment Program. http://www.tricare.mil/2008HealthBehaviors.pdf (accessed August 22, 2012).
Tseng, T., H. Lin, S. Moody-Thomas, M.Y. Martin, and T. Chen. 2012. Who tended to continue smoking after cancer diagnosis: the national health and nutrition examination survey 1999-2008. BMC Public Health 12:784.
USPSTF (U.S. Preventive Services Task Force). 2009. Counseling and interventions to prevent tobacco use and tobacco-caused disease in adults and pregnant women. http://www.uspreventiveservicestaskforce.org/uspstf/uspstbac2.htm (accessed September 7, 2012).
Vallone, D. M., J. C. Duke, J. Cullen, K. L McCausland, and J. A. Allen. 2011a. Evaluation of EX: A national mass media smoking cessation campaign. American Journal of Public Health 101(2):302-309.
Vallone, D. M., J. Niederdeppe, A. K. Richardson, P. Patwardhan, R. Niaura, and J. Cullen. 2011b. A national mass media smoking cessation campaign: Effects by race/ethnicity and education. American Journal of Health Promotion 25(5 Suppl):S38-S50.
Wakefield, M., Y. Terry-McElrath, S. Emery, H. Saffer, F. J. Chaloupka, G. Szczykpa, B. Flay, P. M. O’Malley, and L. Johnston. 2006. Effect of televised, tobacco company—funded smoking prevention advertising on youth smoking-related beliefs, intentions, and behavior. American Journal of Public Health 96(12):2154-2160.
Wakefield, M. A., B. Loken, and R. C. Hornik. 2010. Use of mass media campaigns to change health behavior. Lancet 376(9748):1261-1271
Walker, M. S., D. J. Vidrine, E. R. Gritz, R. J. Larsen, Y. Yan, R. Govindan, and E. B. Fisher. 2006. Smoking relapse during the first year after treatment for early-stage non-small-cell lung cancer. Cancer Epidemiology, Biomarkers & Prevention 15(12):2370-2377.
Warner, K. E., and D. Mendez. 2010. Tobacco control policy in developed countries: Yesterday, today, and tomorrow. Nicotine & Tobacco Research 12(9):876-887.
Warner, K. E., T. A. Hodgson, and C. E. Carroll. 1999. Medical costs of smoking in the United States: Estimates, their validity, and their implications. Tobacco Control 8(3):290-300.
Warren, G. W., V. M. Rangnekar, R. McGarry, S. M. Arnold, and M. Kudrimoti. 2008. Pathways of Resistance: Potential Effects of Nicotine on Cancer Treatment Response. International Journal of Radiation Oncology Biology Physics 72:S715.
Warren, G. W., K. A. Kasza, M. E. Reid, K. M. Cummings, and J. R. Marshall. 2012a. Smoking at diagnosis and survival in cancer patients. International Journal of Cancer Apr 26. [E-pub ahead of print.]
Warren, G. W., E. N. Peters, E. Torres, K. M. Cummings, E. R. Gritz, A. Hyland, R. Herbst, J. R. Marshall, and B. A. Toll. 2012b. Abstract 648: Tobacco assessment in actively accruing Cooperative Group clinical trials. Cancer Research 72(8, S1):10.1158/1538-7445.AM2012-1648.
Warren, G. W., M. A. Romano, M. R. Kudrimoti, M. E. Randall, R. C. McGarry, A. K. Singh, and V. M. Rangnekar. 2012c. Nicotinic modulation of therapeutic response in vitro and in vivo. International Journal of Cancer 131(11):2519-2527.
Warren, G. W., S. M. Arnold, J. P. Valentino, T. J. Gal, A. J. Hyland, A. K. Singh, V. M. Rangnekar, K. M. Cummings, J. R. Marshall, and M. R. Kudrimoti. 2012d. Accuracy of self-reported tobacco assessments in a head and neck cancer treatment population. Radiotherapy and Oncology 103(1):45-48.
Wewers, M. E., K. Kidd, D. Armbruster, and L. Sarna. 2004. Tobacco dependence curricula in U.S. baccalaureate and graduate nursing education. Nursing Outlook 52(2):95-101.
Williams, J. M., and D. Ziedonis. 2004. Addressing tobacco among individuals with a mental illness or an addiction. Addictive Behaviors 29(6):1067-1083.
YouTube. 2006. The truth—body bags TV ad. http://www.youtube.com/watch?v=c4xmFcrJexk (accessed September 5, 2012).
YouTube. 2012. No on Proposition 29. http://www.youtube.com/watch?v=fNHKfj04op4 (accessed September 7, 2012).