Under the Endangered Species Act (ESA), the US Fish and Wildlife Service (FWS) and the National Marine Fisheries Service (NMFS)—herein called the Services—are responsible for listing species as endangered or threatened and for designating critical habitats that are essential for their conservation. Furthermore, in consultation with the Services, federal agencies must ensure that their actions are not likely to jeopardize listed species or adversely affect critical habitats. Under the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA), the US Environmental Protection Agency (EPA) is responsible for registering or reregistering pesticides and must ensure that pesticide use does not cause any unreasonable adverse effects on the environment, which is interpreted to include listed species and their critical habitats. The agencies have developed their own approaches to evaluating environmental risk, and their approaches differ because their responsibilities, institutional cultures, and expertise differ. Over the years, the agencies have tried to resolve their differences but have been unsuccessful in reaching a consensus regarding their assessment approaches. As a result, FWS, NMFS, EPA, and the US Department of Agriculture asked the National Research Council (NRC) to examine scientific and technical issues related to determining risks posed to listed species by pesticides. Specifically, the NRC was asked to evaluate methods for identifying the best scientific data available; to evaluate approaches for developing modeling assumptions; to identify authoritative geospatial information that might be used in risk assessments; to review approaches for characterizing sublethal, indirect, and cumulative effects; to assess the scientific information available for estimating effects of mixtures and inert ingredients; and to consider the use of uncertainty factors to account for gaps in data. The present report, which was prepared by the NRC Committee on Ecological Risk Assessment under FIFRA and ESA, is the response to that request.
THE FEDERAL INSECTICIDE, FUNGICIDE, AND RODENTICIDE ACT AND THE ENDANGERED SPECIES ACT
FIFRA is the federal statute that governs the sale, distribution, and use of pesticides in the United States; it assigns EPA the authority to issue pesticide registrations or reregistrations, which are required for use of the pesticides. To obtain a registration, an applicant must demonstrate that a pesticide will perform
its intended function and will not cause unreasonable adverse environmental effects. Once granted, the registration requires that the pesticide be labeled with specific product information, directions for use, and hazard information; the label specifies legal use of the pesticide.
The ESA is the federal statute that assigns FWS and NMFS the authority to designate species as threatened or endangered—that is, to “list” species—and governs the activities that might affect listed species. Under the ESA, federal agencies must ensure that their actions do not harm listed species or jeopardize their existence. Accordingly, if EPA is deciding whether to register a pesticide, it must determine whether the action “may affect” a listed species. If the answer is yes, EPA has the option of initiating a formal consultation or conducting further analysis to determine whether the action is “likely to adversely affect” listed species. If EPA determines that the action is not likely to affect listed species adversely—and FWS or NMFS, as appropriate, agrees—no further consultation is required. However, if EPA determines that the action is likely to affect a listed species adversely, a formal consultation is required, and FWS or NMFS must determine whether the proposed action is likely to jeopardize the existence of the listed species. The product of that determination is called a biological opinion (BiOp) and is issued by FWS or NMFS.
Compliance with the ESA in registering pesticides creates some challenges. First, pesticides are intended to harm target organisms and are intentionally released into the environment. Other species that are in an area where a pesticide is applied could be exposed to and harmed by the pesticide. Second, FIFRA requires that EPA must determine before registering a pesticide that the use of the pesticide will not cause an unreasonable adverse effect on the environment, taking into account economic and social benefits associated with its use. That is, EPA weighs the costs to human health and the environment that could result from pesticide use against social and economic benefits, such as the benefits of mitigating disease vectors and reducing crop damage. The ESA prohibits jeopardizing listed species or adversely affecting their critical habitats but does not generally consider economic and social costs and benefits. Third, FIFRA creates a national registration process in which pesticides are registered on a nationwide basis, but the ESA calls for evaluating effects on specific species and their critical habitats and thus is geographically and temporally focused. The differences between the statutes have led to conflicting approaches in evaluating risks and have contributed to the current inability to reach consensus on assessing risks to listed species from pesticides.
A COMMON APPROACH
Compliance with the ESA in the context of pesticide registration requires EPA and the Services to determine the probability of adverse effects on listed species and their critical habitat when a pesticide is used according to its label requirements. Clearly, there are tensions among the agencies in making that de-
termination, many of which seem to result from different assumptions, technical approaches (data and models used), and risk-calculation methods. What is needed is a common, scientifically credible approach that is acceptable to EPA and the Services. The committee concludes that the risk-assessment paradigm that traces its origins to the seminal NRC report Risk Assessment in the Federal Government: Managing the Process1 and more recently to the NRC report Science and Decisions: Advancing Risk Assessment2 provides such an approach. After 30 years of use and refinement, this risk-assessment paradigm has become scientifically credible, transparent, and consistent; can be reliably anticipated by all parties involved in decisions regarding pesticide use; and clearly articulates where scientific judgment is required and the bounds within which such judgment can be applied. The process is used for human-health and ecological risk assessments and is used broadly throughout the federal government. Thus, the committee concludes that the risk-assessment paradigm reflected in the ecological risk assessment (ERA) process is singularly appropriate for evaluating risks posed to ecological receptors, such as listed species, by chemical stressors, such as pesticides.
Figure S-1 shows the three major steps in the ESA process in connection with the ERA framework. As illustrated in the figure, the framework is the same at each step, but the contents of each element (problem formulation, exposure and effects analysis, and risk characterization) are expected to change as the focus shifts from assessing whether a pesticide “may affect” a listed species (Step 1) to whether it is “likely to adversely affect” a listed species (Step 2) to whether it is likely to jeopardize the continued existence of a listed species (Step 3). That is, the assessment becomes more focused and specific to the chemicals, species, and habitats of concern as it moves from Step 1 to Step 3. If the Services can build on the EPA assessment conducted for Steps 1 and 2 rather than conducting a completely new analysis for Step 3, the ERA will likely be more effective and scientifically credible. Although the committee does not expect the basic risk-assessment framework to change, it recognizes that risk-assessment approaches and methods for determining, for example, what is hazardous, what concentration or quantity is hazardous, what end points constitute an adverse effect, and when, where, and how much exposure is occurring will continue to evolve.
Given the changing scope of the ERA process from Step 1 to Step 3, EPA and the Services need to coordinate to ensure that their own technical needs are met. One approach is to use problem formulation, conducted as part of the ERA process, as an effective means for agencies to coordinate and reach agreement
______________________
1NRC (National Research Council). 1983. Risk Assessment in the Federal Government: Managing the Process. Washington, DC: National Academy Press.
2NRC (National Research Council). 2009. Science and Decisions: Advancing Risk Assessment. Washington, DC: National Academies Press.
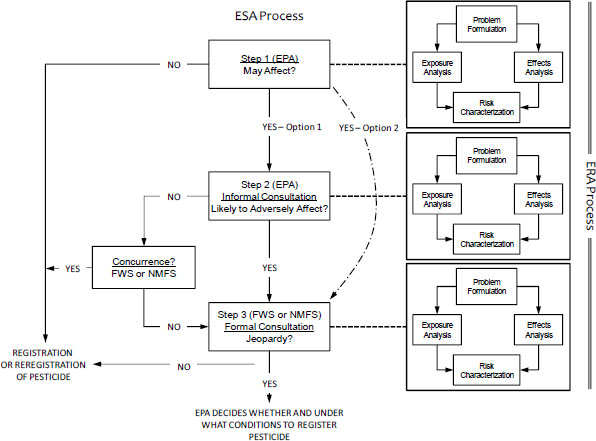
FIGURE S-1 Relationship between the Endangered Species Act (ESA) process and the ecological risk assessment (ERA) process. Each step answers the question that appears in the box.
on many of the key technical issues involved in assessing risks posed to listed species by pesticides. Another approach would be to use technical working groups that address technical details of the assessment approach and other working groups to address policy-based issues. Regardless of the approach, the committee views coordination among EPA and the Services as a collegial exchange of technical and scientific information for the purpose of producing a complete and representative assessment of risk that includes a discussion on the types and depths of analyses needed for the decision and on the time and resources available.
BEST DATA AVAILABLE
One of the critical tasks in any risk assessment is to identify the data that will be used. The ESA directs the Services to conduct assessments on the basis of the “best scientific and commercial data available.” However, the ESA, its legislative history, the rules and policies of the Services, and court cases contain little guidance for elaborating the meaning of that mandate, and the agencies do not appear to have formal protocols that define “best data available.” Consequently, there have been some conflicts about what data to include in the assessments. EPA and the Services do have information-quality guidelines, and each appears to use assessment factors that include data-quality and datarelevance criteria.
Regardless of the breadth of the data collection, some guidelines—such as those listed below—need to be followed in identifying and selecting data for a credible assessment.
• Document the strategy for all data searches and retrieval. For example, if a repository database is searched, the date that the search was conducted and all search terms and search criteria should be documented. The content and scope of the repository, its criteria for inclusion and exclusion of data, the periodicity of its updates, and its quality-control measures also should be documented.
• To ensure that the best data available are used, screen the data first for relevance.3 Information that is not relevant clearly should not be used in assessing risk. Data should be from studies of the species and chemicals being assessed, or there should be a reasonable theoretical basis for data extrapolation. The data should also be applicable to the locations being considered and should be recent.
• Review the quality of the relevant data before they are used in a risk assessment.4 Sufficient information should be included to enable an independent
______________________
3 Relevance refers to the applicability of the data for the intended purpose.
4 Quality refers to the scientific adequacy of the design and execution of data collection, the analyses that use the data, and the data reporting.
evaluation of data quality. Data sources that lack sufficient details for adequate scientific evaluation—such as poster presentations, abstracts, anecdotal or personal communications, and secondary sources—might provide useful background knowledge or support an overall weight-of-evidence evaluation but should not be the sole basis of conclusions in an assessment.
• For transparency, document the evaluation of all data used with particular attention to sources, relevance, and quality and describe any issues associated with those data attributes in the discussion of uncertainty in the risk characterization.
Given that various stakeholders are aware of and can provide relevant and high-quality data, the committee encourages provision for their involvement in the early stages and throughout the risk-assessment process. The committee notes that stakeholder data are expected to meet the same standards of relevance and quality as all other data.
EXPOSURE
Exposure analysis is a principal component of ERA and involves estimating the concentrations of various chemicals released into the environment and their breakdown products of toxicological significance. The following sections discuss exposure-modeling practices and the criteria for authoritative geospatial data and highlight the committee’s conclusions on those topics.
Exposure-Modeling Practices
To determine whether a pesticide will adversely affect or jeopardize the existence of a listed species or its critical habitat, one must estimate the concentration to which the species might be exposed or the concentration that might result in the ecosystem. To accomplish that task, chemical fate and transport models are used. Because the pathways by which pesticides move from their points of application to habitats of listed species can involve a complex sequence of transfers with diverse degradation processes, it is common to use a linked series of models to estimate exposure.
The committee acknowledges that the models used for exposure analyses have several strengths but emphasizes that a model’s limitations need to be recognized and the model used in the appropriate context. As noted above, the committee has suggested a common approach that involves more refined and sophisticated modeling and analysis as one moves from Step 1 to Step 3 in the ESA process. Given the current practices in exposure analysis and the need to estimate pesticide exposures and the associated spatial-temporal variations experienced by listed species and their habitats, the committee envisions the following stepwise approach to exposure modeling.
• Step 1 (EPA). Initial exposure modeling would answer the question, Do the areas where the pesticide will be used overlap spatially with the habitats of any listed species? The Services, which have extensive knowledge of the natural history of listed species, could help EPA to identify overlaps of areas where a pesticide might be used and the habitats of listed species.
• Step 2 (EPA). If area overlaps are identified in Step 1, EPA would confer with the Services to identify relevant environmental compartments (water, soil, air, and biota), associated characteristics, and critical times or seasons in which environmental exposure concentrations need to be estimated. If the models indicate that substantial amounts of pesticides move off the application site and into the surrounding ecosystems, more sophisticated fate and transport processes could be used. At that point, the fate model could be simplified to remove processes that are unimportant in the specific regions where the listed species are and set up to estimate time-varying and space-varying pesticide concentrations in typical habitats with associated uncertainties. On the basis of the modeling results, EPA could then make a decision about the need for formal consultation with the Services.
• Step 3 (Services). During a formal consultation, the Services would further refine the exposure models to develop quantitative estimates of pesticide concentrations and their associated distributions for the particular listed species and their habitats. To that end, the models would use site-specific input values, such as actual pesticide application rates, locally relevant geospatial data, and time-sensitive life stages of listed species.
The committee emphasizes that many parameters are used in chemical fate and transport models, and their accuracy is important ultimately to the concentrations estimated in the modeling efforts. Little effort has been expended in evaluating the data inputs relevant to particular ESA evaluations. Therefore, if the agencies want to obtain more accurate modeling results, a subset of case-specific exposure estimates should be evaluated by pursuing a measurement campaign specifically coordinated with several pesticide field applications. The committee notes that field studies need to be distinguished from general monitoring studies. General monitoring studies provide information on pesticide concentration on the basis of monitoring of specific locations at specific times and are not associated with specific applications of pesticides under well-described conditions. Therefore, general monitoring data cannot be used to estimate pesticide concentrations after a pesticide application or to evaluate the performance of fate and transport models.
Geospatial Data, Habitat Delineation, and Exposure Analysis
Habitat includes all environmental attributes present in an area that allow an organism to survive and reproduce, and habitat delineation is necessary for determining where a pesticide and a species might co-occur, for calculating spa-
tially explicit estimates of pesticide exposure, and for specifying the spatial structure of population models used in effects analysis (see below). Several methods for identifying and statistically modeling associations between species and their environment exist; although some caveats and uncertainties are associated with them, quantitative statistical habitat delineation is typically objective and more reliable than qualitative and subjective habitat descriptions.
The accuracy and reliability of habitat delineation and exposure analysis are increased substantially by the use of authoritative geospatial data. To be considered authoritative, geospatial data on any scale need to meet three criteria: availability from a widely recognized and respected source, public availability, and inclusion of metadata5 that are consistent with the standards of the National Spatial Data Infrastructure—a federal interagency program to organize and share spatial data and to ensure their accuracy. The geospatial data that are most useful for delineating habitat and estimating exposure are data on topography, hydrography, meteorology, solar radiation, soils, geology, and land cover. Table S-1 provides some examples of authoritative sources of those data. In many cases, there are multiple authoritative sources for each type of data on different spatial and temporal scales. Although it would be ideal to be able to identify specific authoritative sources, no one source will be best for all habitat delineations, exposure analyses, or other applications. However, accuracy assessments that generally are available for authoritative data sources might allow one to gauge which source is likely to be the most reliable for a particular objective.
EFFECTS
Pesticides are designed to have biological activity; specifically, they are “intended for preventing, destroying, repelling or mitigating” pests. Consequently, they have the potential to cause a variety of effects on nontarget organisms, including listed species. Determining the potential for and possible magnitude of effects is a process known as effects analysis. The following sections consider various topics on effects analysis as they are related to the committee’s task and highlight the committee’s conclusions on the topics.
Sublethal, Indirect, and Cumulative Effects
Pesticides can kill organisms but can also affect reproduction or growth or make organisms less competitive. Although EPA and the Services agree that those sublethal (less-than-lethal) effects should be considered in the assessment process, they disagree on the extent to which they can be included. To address
______________________
5Metadata document the fundamental attributes of data, such as who collected them, when and where they were collected, what variables were measured, how and in what units measurements were taken, and the coordinate system used to identify locations.
that issue, the committee first considered how to define objectively the degree to which observed effects are adverse. Defining adversity is essential for ERA because the mere existence of an effect is not sufficient to conclude that it is adverse. The committee concluded that the only way to determine whether an effect is adverse and how adverse it might be is to assess the degree to which it affects an organism’s survival and reproductive success; any effect that results in a change in either survival or reproduction is relevant to the assessment, and any effect that does not change either outcome is irrelevant with respect to a quantitative assessment of population effects. Thus, EPA in Step 2 (see Figure S-1) should conduct a broad search to identify sublethal effects of pesticides and any information on concentration-response relationships. In Step 3, the Services should then show how such effects change probability of survival or reproduction of the listed species and incorporate such information into the population viability analyses or state that such relationships are unknown but possible and include a qualitative discussion in the uncertainty section of the BiOp. The inability to quantify the relationship between a sublethal effect and survival or reproductive success does not mean that the sublethal effect has no influence on population persistence; but in the absence of data, the relationship remains a hypothesis that can be discussed only qualitatively with reference to the scientific literature to explain why such a hypothesis is tenable.
TABLE S-1 Examples of Authoritative Sources of Geospatial Data
| Data Type | Examples of Authoritative Data Sources | |
| Topography | Topographic features can be derived from elevation data in the National Elevation Dataset, the Shuttle Radar Topography Mission, and the Global Digital Elevation Map. | |
| Hydrography | Watershed data are available on line from EPA; watersheds are referred to by hydrologic unit codes of the US Department of Agriculture Natural Resources Conservation Service. | |
| Meteorology | Data are available from the National Oceanographic and Atmospheric Administration National Climatic Data Center. | |
| Solar radiation | Solar-radiation data are available from the National Aeronautics and Space Administration Earth Observing System Solar Radiation and Climate Experiment;a solar insolation can be estimated by using the on-line calculator of the Photovoltaic Education Network. | |
| Soils | Soil surveys are available from the US Department of Agriculture Natural Resources Conservation Service. | |
| Geology | Geological data are available from the US Geological Survey Mineral Resources Online Spatial Data. | |
| Land cover | Land-cover data are available from the National Agricultural Statistics Service. | |
aSolar-radiation measurements are taken at the top of Earth’s atmosphere. Computer modeling is required to estimate solar radiation at Earth’s surface.
In most cases, pesticides have the potential to affect a listed species indirectly—not through direct exposure but through effects on other species in the community. For example, the prey of a listed species might be reduced in abundance or eliminated by the pesticide, and this would affect the survival of the species. As in the case of sublethal effects, EPA and the Services differ about the degree to which indirect effects can be included in an assessment. The committee recommends that indirect effects that can be quantified relatively easily be incorporated into the effects analysis. However, determining and quantifying most indirect effects can be challenging and can require complex models. When such modeling is conducted, uncertainties should be estimated quantitatively in a realistic and scientifically defensible manner and should be propagated formally and explicitly through the analysis.
A risk assessor must also consider cumulative effects. They are defined by regulation under the ESA as “those effects of future State or private activities, not involving Federal activities that are reasonably certain to occur within the action area of the Federal action subject to consultation” (50 CFR 402.02). However, cumulative effects typically are more broadly defined as effects that interact or accumulate over time and space. The committee could not determine a scientific basis for excluding past and present conditions (the environmental baseline) from the consideration of cumulative effects and therefore used that broad definition in its evaluation. The committee concluded that population models provide a framework for incorporating baseline conditions and projected future cumulative effects into an effects analysis.
One problem that arises in an effects analysis is how to extrapolate toxicity information on tested species to listed species. Although the idea of identifying an appropriate surrogate species is appealing, the committee finds such identification problematic because different species often respond differently to chemical exposures, and the sensitivity differences can be large. Furthermore, different life histories can complicate the extrapolation. A scientifically defensible alternative approach is to define a range of sensitivities within which the sensitivity of a listed species could reasonably be expected to occur or a range of sensitivities that could be used to make reasoned extrapolations from information on species that have been tested by using inferences based on other chemicals. Further details are provided in Chapter 4 of this report.
Effects Models
EPA and the Services use different approaches to determine the potential effects of a pesticide on a listed species and its critical habitat. EPA addresses population effects simply as extensions of individual effects: if survival or reproduction is affected, EPA assumes population-level consequences and enters consultation with the Services. The Services use population models to address the question of population persistence explicitly. Population models are used to estimate population-level end points—such as population growth rate, probabil-
ity of population survival (population viability), and probability of population recovery—on the basis of individual-level effects. For purposes of population modeling, the effects must be estimated at a range of concentrations that includes all values that the populations being assessed might plausibly experience. Therefore, test results expressed only as threshold values, such as a no-observed-adverse-effect level or a lowest observed-adverse-effect level, are insufficient for a population-level risk assessment.
Because the ESA is concerned with species, population models are necessary for quantifying the effects of pesticides on populations of listed species. Population models require three basic inputs: changes in survival or reproduction as a function of pesticide concentration, exposure estimates of pesticide concentration over time and space, and demographic and life-history information. There are a variety of population models, and the choice of a model will depend on the data available. Although species-specific models that incorporate all three inputs are preferred, in the absence of detailed demographic information it is reasonable to use simple generic models that characterize the life history of a group of species to estimate the effects of a pesticide on a given species. It is important to incorporate density dependence by using models with parameter values that are functions of population density or population size, but it is not accurate to assume that mortality due to pesticide exposure will be compensated for by density dependence because it is likely that such exposure will decrease the growth rate of the population at all densities and generally depress the curve of population size vs growth rate.
MIXTURES: AN IMPORTANT CONCERN FOR EXPOSURE AND EFFECTS ANALYSIS
Assessing the risks posed by exposure to mixtures is clearly a subject of disagreement and concern for the agencies. To address the mixture issue, the committee made several distinctions. First, some pesticides might contain more than one active ingredient (a chemical that is responsible for the biological effect of the pesticide); most pesticides contain other chemicals that are typically designated as “inerts.”6 Second, pesticides are often mixed with other chemicals before their application. The resulting mixtures are referred to as tank mixture and can contain other pesticides, fertilizers, and adjuvants—materials that facilitate handling and application, such as surfactants, compatibility agents, antifoaming agents, and drift-control agents. Third, chemicals from other sources are already in the environment; unless exposure occurs only at or near the point of pesticide application, species are more likely to be exposed to environmental mixtures than to a single pesticide formulation or tank mixture. Environmental
______________________
6 The term inerts is defined by FIFRA as an ingredient that is not active. Inerts are intentionally added to pesticide products, and the term does not mean that the chemicals are nontoxic.
mixtures are formed when a tank mixture—active ingredients, inerts, and adjuvants—combines with other chemicals in the environment from other sources. Ideally, assessments should be based on exposure to all pesticide components and to other chemicals that are present in the exposure environment. However, quantitative estimates of exposure to environmental mixtures are difficult given the dynamic state of environmental mixtures over space and time. In any given location, the amounts of pesticide active ingredients, inerts, adjuvants, and other environmental chemicals are highly variable and depend on pesticide uses and other sources of environmental contamination.
EPA recognizes the potential importance of exposure to mixtures but typically assesses only pesticide active ingredients. The Services have expressed substantial concern about the need to account for mixture exposure but have dealt with the issue only with a qualitative discussion in their assessments. The greatest concern is that a mixture component might act to enhance the toxicity of a pesticide active ingredient. The committee notes that a quantitative assessment of the risk posed by chemical mixtures requires extensive data, including data on the identity, concentration, and toxicity of mixture components. Challenges in assessing risk to listed species posed by pesticide-containing mixtures arise largely because of the lack of such data and the lack of understanding of the potential for interactions among mixture components. In the absence of such quantitative data, the possible contribution of specific mixture components to the toxicity of a pesticide active ingredient cannot be incorporated into a quantitative risk assessment. The committee, however, emphasizes that the complexity of assessing the risk posed by chemical mixtures should not paralyze the process, and it provides guidelines in Chapter 4 of its report to help in determining when and how to consider components other than a pesticide active ingredient in a risk assessment.
RISK CHARACTERIZATION AND UNCERTAINTY
Risk characterization is the final stage of a risk assessment in which the results of the exposure and effects analyses are integrated to provide decision-makers with a risk estimate and its associated uncertainty. Two general approaches have been used for risk characterization: the risk-quotient (RQ) approach, which compares point estimates of exposure and effect values, and the probabilistic approach, which evaluates the probability that exposure to a chemical will lead to a specified adverse effect at some future time.
The RQ approach does not estimate risk—the probability of an adverse effect—itself but rather relies on there being a large margin between a point estimate that is derived to maximize a pesticide’s environmental concentration and a point estimate that is derived to minimize the concentration at which a specified adverse effect is not expected. If the results raise doubts regarding the possibility of an adverse effect, the common response is to widen the margin by, for example, adding uncertainty factors or assuming more stringent, and possibly
implausible, exposure scenarios. The flaw in that approach is that there is no accounting for what the probability of an adverse effect was before the application of assumptions, and there is no calculation of how their use modifies that probability. Accordingly, the committee concludes that adding uncertainty factors to RQs to account for lack of data (on formulation toxicity, synergy, additivity, or any other aspect) is unwarranted because there is no way to determine whether the assumptions that are used overestimate or underestimate the probability of adverse effects. Furthermore, the committee concludes that RQs are not scientifically defensible for assessing the risks to listed species posed by pesticides or indeed for any application in which the desire is to base a decision on the probabilities of various possible outcomes.
Instead, the committee recommends using a probabilistic approach that requires integration of the uncertainties (from sampling, natural variability, lack of knowledge, and measurement and model error) into the exposure and effects analyses by using probability distributions rather than single point estimates for uncertain quantities. The distributions are integrated mathematically to calculate the risk as a probability and the associated uncertainty in that estimate. Ultimately, decision-makers are provided with a risk estimate that reflects the probability of exposure to a range of pesticide concentrations and the magnitude of an adverse effect (if any) resulting from such exposure.
The committee recognizes the pragmatic demands of the pesticide-registration process and encourages EPA and the Services to consider the probabilistic methods that have already been successfully applied to pesticide risk assessments, that have otherwise appeared often in the technical literature, that are familiar to many risk-assessment practitioners, that can be implemented with commercially available software, and that are most readily explicable to decision-makers, stakeholders, and the public. The committee also recognizes that administrative and other nonscientific hurdles will need to be overcome to implement this approach, but moving the uncertainty analysis from the typical narrative addendum to an integral part of the assessment is possible and necessary to provide realistic, objective estimates of risk.













