COMMUNITY ECOLOGY AND THE VAGINAL MICROBIOME
Larry J. Forney43,44and Jacques Ravel45,46
Abstract
Human vaginas are home to complex communities of microorganisms that are partners in mutualistic associations with their hosts that are critical to maintaining health and protecting the host from infectious urogenital diseases. Recent studies have begun to shed light on how vaginal microbial communities vary among and within individuals at various stages of a woman’s life span. However, relatively little is known about the function of these communities or how their constituent members interact with each other and the host to form a dynamic ecosystem that responds to environmental disturbances. To better define variation in the species composition of these communities we conducted a cross-sectional study in which the vaginal bacterial communities of 396 asymptomatic North American women that represented four ethnic groups (white, black, Hispanic, and Asian) were sampled and the species composition was characterized by pyrosequencing of barcoded 16S rRNA genes. The communities clustered into five groups; four were dominated by Lactobacillus iners, L. crispatus, L. gasseri, or L. jensenii, while the fifth had lower proportions of lactic acid bacteria and higher proportions of anaerobic organisms. Although it appears that a key ecological function—the production of lactic acid—is conserved, there are statistically significant differences in the proportions of each community group in the four ethnic groups. Moreover, the vaginal pH of women in different ethnic groups also differed and was higher in Hispanic (pH 5.0±0.59) and black (pH 4.7±1.04) women as compared to Asian (pH 4.4±0.59) and white (pH 4.2±0.3) women. In a longitudinal study we investigated the temporal dynamics of vaginal bacterial communities in 32 reproductive age women over a 16-week period. The dynamics were highly personalized and generally varied both within and between women over time, but five major classes of community dynamics could be discerned. These vaginal communities sometimes changed markedly over short periods of time, while others were relatively stable. It is not yet known whether community-level functions varied as the species
________________
43 Institute for Bioinformatics and Evolutionary Studies.
44 Department of Biological Sciences, University of Idaho, Moscow, Idaho.
45 Institute for Genome Sciences.
46 Department of Microbiology and Immunology, University of Maryland School of Medicine, Baltimore, Maryland.
composition of communities changed. Modeling community stability indicated that deviation from stability was correlated with time in the menstrual cycle, community composition, and sexual activity. The women studied were healthy; thus, it appears that neither variation in community composition per se, nor higher levels of observed diversity (codominance) are necessarily indicative of dysbiosis. It is envisioned that better knowledge of vaginal community dynamics and the mutualism between the host and indigenous bacterial communities will lead to the development of strategies to manage the vaginal ecosystem in a way that promotes health and minimizes the use of antibiotics. These efforts should take into account the temporal dynamics of vaginal communities and the potential problems that might arise from diagnostic and therapeutic strategies based on cross-sectional studies.
Introduction
The female lower reproductive tract is home to diverse microbial communities that are thought to play a critical role in maintaining health and protecting individuals from infectious disease. These communities and the host are an example of a finely balanced mutualistic association in which the microbes confer a protective advantage upon their hosts in exchange for a nutrient rich, oxygen-depleted habitat (Danielsson et al., 2011). In reproductive age women, this protective effect is assumptively due to the production of lactic acid and other antimicrobial substances with the former reducing the pH of the vaginal environment making it inhospitable to other invading organisms and precluding unwanted microbial growth (Linhares et al., 2010; O’Hanlon et al., 2011). Lactic acid–producing bacteria (LAB) are thought to be primarily responsible for creating and maintaining this characteristic low-pH environment through the fermentation of glucose liberated from the vaginal epithelium during glycogenolysis (Danielsson et al., 2011). Consequently, high proportions of LAB, particularly Lactobacillus species have been paradigmatically considered the hallmark of vaginal health, while the lack thereof is typically associated with a “disturbance” or a “disease” (O’Hanlon et al., 2011).
Coevolutionary processes between the human host and specific microbial partners have shaped the vaginal ecosystem, although the selective forces driving this mutualistic association are not known. The specificity of this host–microbe association is exemplified by the vaginal lactobacilli. Although these organisms are ubiquitous in the environment, they inhabit only three regions of the human body: the oral cavity, the vagina, and the intestines (Pavlova et al., 2002). Of the more than 100 of species of lactobacilli known to exist, only four species are commonly found as dominant members of vaginal microbial communities, namely L. crispatus, L. iners, L. gasseri, and L. jensenii (Ravel et al., 2011; Zhou et al., 2007). This suggests these species possess certain characteristics that allow them to outcompete others in the vaginal environment for resources or to actively
exclude nonindigenous species by producing antimicrobial compounds and creating an inhospitable environment.
Lactobacilli have been considered the keystone species47 of the normal postpubertal vaginal community since 1892, when Professor Albert Döderlein, a renowned German gynecologist, first cultured the organism from vaginal secretions obtained from healthy, pregnant women (Döderlein, 1892; Martin, 2011). These long, slender rods—originally called “Döderlein’s bacillus”—were renamed Lactobacillus acidophilus in 1928 after their ability to produce lactic acid and reduce the vaginal pH (Danielsson et al., 2011). In the 1980s, it was later discovered that L. acidophilus was not a single species, but a group of closely related, obligately homofermentative species collectively known as the Lactobacillus acidophilus complex (Lauer et al., 1980). Since species in this complex are difficult to differentiate phenotypically or biochemically (Johnson et al., 1980), they have been classified on the basis of DNA homology (Du Plessis and Dicks, 1995; Schleifer and Ludwig, 1995). All species of Lactobacillus recovered from the vagina to date belong to this complex, including L. iners, L. crispatus, L. jensenii, L. gasseri, L. casei, L. plantarum, L. fermentum, L. cellobiosus, L. brevis, L. minutus (now classified as Atopobium minutum) (Collins and Wellbanks, 1992), and L. salivarius (Antonio et al., 1999; Levison et al., 1977; Reid et al., 1996; Rogosa and Sharpe, 1960).
Lactic acid bacteria that predominate in the vaginas of reproductive-age women also metabolize extracellular glycogen into lactic acid by anaerobic glycolysis, and also release the acid into the vagina. Epithelial cells produce only the L-lactate isomer, while many bacteria are capable of producing both D- and L-lactate. Moreover, many organisms produce organic acids such as formic acid, acetic acid, propionic acid, succinic acid and others through fermentative metabolism and thereby contribute to acidification of the vaginal environment. The environmental pH of the vagina, traditionally considered to be around 4.0–4.5 in healthy women, is presumably determined by the organic acid contributions from both epithelial cells, resident LAB, and other anaerobic and strictly anaerobic bacteria (Linhares et al., 2010). In addition to lactic acid, vaginal lactobacilli are also capable of producing other antimicrobial compounds, such as antibiotics, target-specific bacteriocins—proteinaceous substances capable of permeabilizing target cell membranes (Aroutcheva, 2001; Oscariz and Pisabarro, 2001)—and the broad-spectrum antimicrobial, hydrogen peroxide (Hawes et al., 1996; Klebanoff et al., 1991). The vagina is, however, a virtually anoxic environment, and as a result it is unlikely that any significant amounts of hydrogen peroxide would be produced and allowed to accumulate to a toxic antimicrobial level in vitro (O’Hanlon et al., 2011).
________________
47 A keystone species is one that plays a unique and crucial role in the way an ecosystem functions.
Common Wisdom of the Vaginal Microbiota
Vaginal microbial communities undergo significant structural changes at various stages in a woman’s life span that are directly linked to the level of estrogen in the body (Farage and Maibach, 2006). While numerous studies have been done to understand the vaginal microbiology of reproductive age women, far fewer studies have been done on the vaginal microbiota48 of premenarcheal, perimenopausal, and postmenopausal females. Despite the paucity of data a plausible sequence of successional events can be described that occur in the vaginal microbiota over a woman’s life span.
Initial colonization of the vagina occurs at birth when the infant is first exposed to the mother’s vaginal microbiota via passage through the birth canal, or by the skin bacteria of persons handling infants delivered via Caesarian section (Domingez-Bello, 2010). This initial colonization event is believed to establish the gut, skin, and vaginal microbiota, allowing them to differentiate into habitat-specific communities in the weeks and months following birth (Koenig et al., 2011). During the first 2–4 weeks of life, maternal estrogen mediates maturation of the vaginal epithelium and the accumulation of glycogen that is fermented by indigenous bacteria resulting in a lowering of the vaginal pH (~5.0). This effect is transitory, however, as waning maternal estrogen, subsequent thinning of the vaginal mucosa, and a concomitant increase in vaginal pH occur. During childhood the vagina is colonized by diverse assemblages of aerobic, strictly anaerobic, and enteric species of bacteria and the pH is nearly neutral (Farage and Maibach, 2006). At the onset of puberty, adrenal and gonadal maturation causes an increase in estrogen production, which once again causes thickening of the vaginal epithelium and accumulation of glycogen. These environmental conditions selectively favor the proliferation of glycogen-fermenting LAB and the concomitant acidification of the vaginal environment (pH~4.5), which is sustained throughout the reproductive years. The vaginal microbiota of adolescent girls (13–18 years) are comparable to those found in adults, but less is known whether this is also the case for premenarcheal or perimenarcheal girls (Farage and Maibach, 2006). During menopause, a decrease in estrogen levels and the cessation of menstruation are accompanied by atrophy of the vaginal epithelium and reduced cervicovaginal secretions (Farage and Maibach, 2006). In many women this is accompanied by a shift in the vaginal microbiota from populations of lactic acid-producing bacteria to an assortment of species that include anaerobic bacteria comparable to those found during childhood or bacterial vaginosis. Likewise, vaginal pH typically rises to near-neutral levels (6.5–7.0) in
________________
48 The term microbiota refers collectively to all the microorganisms present in a given environment without regard to whether they interact with one another. This is in contrast to the ecological concept of a “microbial community,” which refers to a group of actually or potentially interacting species that inhabit a place. These terms are distinct from the broader term “microbiome” that is more holistic and includes the microorganisms as well as the environment in which they are found.
women who do not use hormone-replacement therapy (HRT), whereas women who do use HRT typically maintain a vaginal pH comparable to that of reproductive age women (4.5–5.0) and a Lactobacillus dominated microbiota (Danielsson et al., 2011). The dynamic nature of this ecosystem underscores the importance of resolving its microbial constituents at different stages of human development and the important role of estrogen on the vaginal environment.
Cultivation-Dependent and Cultivation-Independent Studies
More recently cultivation-independent methods reliant on DNA sequencing of 16S rRNA genes have provided a means to study fine-scale variation in host-associated microbial communities within and among individuals and exploration of ecological relationships between bacterial species and the host. Typically, partial 16S rRNA gene sequences are amplified from total genomic DNA from a sample, and the resulting amplicons are sequenced. Phylogenetic analyses of the sequences allows for classification of phylotypes and determination of the numerically dominant taxa in a community. Major advances in DNA sequencing technologies over the last decade have fundamentally changed the way we assess microbial community structure and composition, and this has facilitated more expansive and intensive studies of vaginal community structure and dynamics. Below we summarize the results of studies done by our group using this approach and the insights to the structure, function, and dynamics of vaginal microbial communities that have been obtained.
Results
Vaginal Bacterial Community Composition and Structure
We characterized the vaginal microbiota and vaginal pH of 396 asymptomatic, sexually active women that fairly equally represented four self-reported ethnic groups: Caucasian (N = 98), black (N = 104), Asian (N = 97), and Hispanic (N = 97) (Ravel et al., 2011). Self-collected midvaginal samples were obtained and used to determine the species composition and structure of the resident bacterial communities by phylogenetic analysis of 16S rRNA gene sequences (Hamady and Knight, 2009). Whole genomic DNA was extracted from each swab and variable regions 1 and 2 (V1–V2) of 16S rRNA genes were PCR amplified using the barcoded universal primers. The resulting amplicons were pyrosequenced, and the 897,345 high-quality sequences (~2,200 reads per sample) were classified using the RDP Naïve Bayesian Classifier (Wang et al., 2007). Species-level taxonomic assignments of Lactobacillus sp. was done using a bioinformatics algorithm based on a combination of species-level Hidden Markov Models (HMM) and clustering as described by Ravel et al. (2011). Overall, a total of 282 taxa were observed in the vaginal communities of these women. The depth of coverage
for each community was sufficient to detect taxa that constitute ~0.1% of the community. While taxa present at less than this level are often referred to as low abundance or “rare” taxa, they are only rare in the context of sampling depth. If a vaginal bacterial community has ~108 cells per ml of vaginal secretion, then high numbers (105 cells per ml) of “rare” members are present in the community, while phylotypes present at densities less than 105 cells per ml would remain undetected. These rare taxa could play major roles in the ecology of a community while undetected members may constitute a seed bank of species whose numbers increase under conditions that favor their growth.
The vaginal bacterial communities were grouped based on community composition (Figure A11-1, panel A), and the phylotypes were clustered based on their correlation profiles (Figure A11-1, panel C). The heat map in Figure A11-1 shows the results obtained using log10 transformed percent abundance of each taxon. It highlights the diversity found in all vaginal bacterial communities, even those where the phylotypes abundance is highly skewed and dominated by a single phylotype, and identifies taxa with similar correlation profiles. The analysis revealed five major groups of microbial communities, which we named community state types (CST) and is reminiscent of previously published studies on microbial diversity in the human vagina (Zhou et al., 2007, 2010). The five CSTs, designated CST I, CST II, CST III, CST IV, and CST V, contained 104, 25, 135, 108, and 21 taxa, respectively. The most diverse communities were those of CST IV, and this was reflected in the Shannon diversity indices of the communities (Figure A11-1).
Unlike any other anatomical site on the human body, most vaginal communities (73%) were dominated by one or more species of Lactobacillus that constitute more than 50% of all sequences obtained. Communities in CST I, which occurred in 26.2% of the women sampled were dominated by L. crispatus, while CST II (6.3%), CST III (34.1%), and CST V (5.3%) were dominated by L. gasseri, L. iners, and L. jensenii, respectively. The skewed rank abundances of species in these communities leaves the impression that these communities are species poor, but this may not be the case since there are an unknown number of rare species. The remaining communities found in 27% of the women formed a large heterogeneous group (CST IV), and were typified by higher proportions of anaerobic and strictly anaerobic bacteria, including Prevotella, Dialister, Atopobium, Gardnerella, Megasphaera, Peptoniphilus, Sneathia, Eggerthella, Aerococcus, Finegoldia, and Mobiluncus. This finding is consistent with those of previous studies wherein the species composition of vaginal communities was investigated by cloning and sequencing of 16S rRNA genes (Ferris et al., 2004, 2007; Fredricks et al., 2005; Srinivasan and Fredricks, 2008; Verhelst et al., 2004; Verstraelen et al., 2004; Zhou et al., 2007, 2010). It should be pointed out that while communities in CST IV appear to be diverse relative to the other CST, it could simply reflect greater species evenness. Although communities in CST IV were not dominated by Lactobacillus sp., L. iners, and L. crispatus were detected
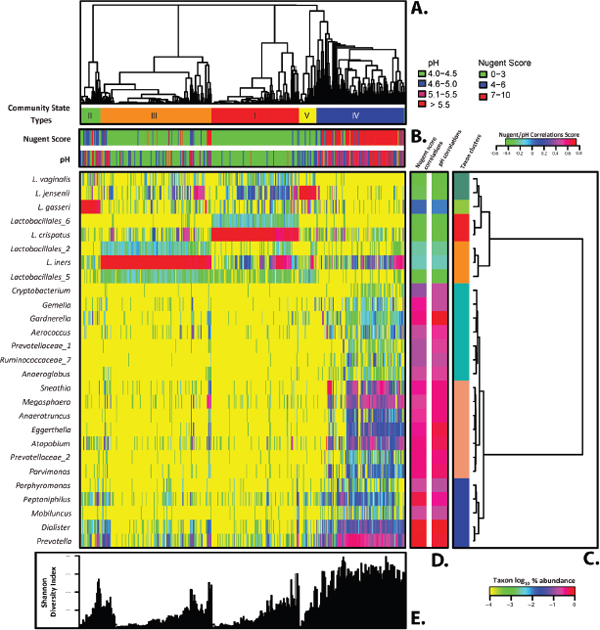
FIGURE A11-1 Heat map of log10 transformed proportions of microbial taxa found in the vaginal bacterial communities of 394 women of reproductive age (color key is indicated in the lower-right corner). (A) Complete linkage clustering of samples based the species composition and abundance of vaginal bacterial communities that defines community groups I to V. (B) Nugent scores and pH measurements for each of the 394 community samples (color key is indicated above panel C). (C) Complete linkage clustering of taxa based on Spearman’s correlation coefficient profiles, which were defined as the set of Spearman’s correlation coefficients calculated between one taxon and all the other taxa. (D) Spearman’s correlation coefficients between the presence of a taxon and the Nugent score or pH of a sample. (E) Shannon diversity indices calculated for 394 vaginal communities. SOURCE: Modified from Ravel et al. (2011).
in 78.7% and 51.9% of CST IV communities, respectively. Only four subjects in CST IV lacked detectable Lactobacillus sp. in their vaginas, and those communities were dominated by Prevotella, Sneathia, Megasphaera, or Streptococcus. Interestingly, all communities contained members that have been assigned to genera known to produce lactic acid including Lactobacillus, MegasphaeraStreptococcus, and Atopobium. This suggests that an important catabolic function, namely the production of lactic acid, might be conserved among each community’s state types despite differences in the species composition.
Core Microbiomes of the Human Vagina
One objective of studies on the human microbiome is to determine if there is a core set of microbial species associated with the bodies of all humans. It is postulated that changes to this “core microbiome” may be correlated with changes in human health or risk to disease. The results from this study suggest that for the human vagina there is no single core microbiome. Instead, it appears there are multiple core microbiomes that can be defined by CST I–V depicted in Figure A11-1. As noted above, these groups can be readily distinguished on the basis of two criteria: (a) whether the constituent communities are dominated by Lactobacillus or not, and (b) the particular species of Lactobacillus present. The vast majority of communities in CST I, CST II, CST III, and CST V had more than one phylotype of lactic acid bacteria suggesting a degree of functional redundancy, but they differed widely in abundance. Although no core microbiome can be identified based on the taxa found in these communities, we posit that core functions are conserved among communities despite differences in their species composition, and that functional redundancy would be associated with increased community reliability in the face of environmental changes (Konopka, 2009).
Differences in the Vaginal Microbiomes of Ethnic Groups
The study cohort consisted of roughly equal numbers of four self-described ethnicities (Caucasian, Asian, black, and Hispanic), and this offered the opportunity to assess the relationship of ethnic background on vaginal bacterial community composition. The proportions of each CST varied among the four ethnic groups (Figures A11-2 and A11-3), and these differences were statistically significant (χ2 = 36.8 on 10 df, P < .0001). No statistically significant associations were observed between age and community types within or across ethnic groups.
Vaginal bacterial communities dominated by species of Lactobacillus (CSTs I, II, III, and V) were found in 80.2% and 89.7% of Asian and white women, respectively, but in only 59.6% and 61.9% of Hispanic and black women, respectively. The higher median pH values in Hispanic (pH 5.0±0.59) and black (pH 4.7±1.04) women reflects the higher prevalence of communities not dominated by Lactobacillus sp. (CST IV) in these two ethnic groups when compared
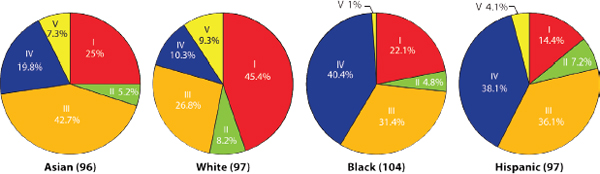
FIGURE A11-2 Representation of vaginal bacterial community groups within each ethnic group of women.
NOTE: The number of women from each ethnic group is in parentheses.
SOURCE: Modified from Ravel et al. (2011).
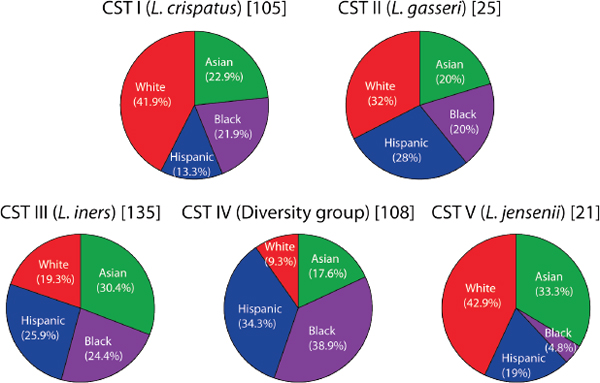
FIGURE A11-3 Contribution of ethnicity to each of the five vaginal community groups expressed as percentage.
NOTE: Sectors are colored according to ethnicity and labeled accordingly. The percentage represents the proportion of subjects of each ethnicity divided by the total number of subjects assigned to a community group (indicated in square brackets).
The dominant species for each community group is indicated in parentheses. SOURCE: Modified from Ravel et al. (2011).
to Asian (pH 4.4±0.59) and white (pH 4.2±0.3) women (Table A11-1). This is significant because the occurrence of high numbers of lactobacilli and pH < 4.5 have become synonymous with :healthy.” If accepted at face value, this common wisdom suggests that while most Asian and white women are healthy, a significant proportion of asymptomatic Hispanic and black women are “unhealthy”; a notion that seems implausible. It also begs the question of what kinds of bacterial communities should be considered normal in Hispanic and black women. We found that CST IV was overrepresented in Hispanic (34.3%) and black (38.9%) women as compared to Asian (17.6%) and white (9.3%) women (Figure A11-3). From these data we conclude that vaginal bacterial communities not dominated by species of Lactobacillus are common and normal in black and Hispanic women. The data from this study are in accordance with the results of Zhou et al. (2007, 2010) who studied the vaginal bacterial communities of white, black, and Japanese women. The reasons for these differences among ethnic groups are unknown, but it is tempting to speculate that the species composition of vaginal communities could be governed by genetically determined differences between hosts. These might include differences in innate and adaptive immune systems, the composition and quantity of vaginal secretions, and ligands on epithelial cell surfaces, among others. While these may be key to shaping vaginal communities, previous studies have also shown that human habits and practices including personal hygiene, methods of birth control, and sexual behaviors also exert strong influences (Schwebke, 2009).
The small number of different kinds of vaginal communities is somewhat surprising given that these communities are probably assembled independently after birth. The repeatability of community assembly suggests that a host exerts strong selection for a rather limited number of different kinds of bacteria. This is especially evident in the limited number of Lactobacillus phylotypes and other lactic acid producing bacteria that are abundant in these communities. The prominence of these populations and their important role in modulating vaginal pH suggests they might be drivers in these communities and thought of in terms of Walker’s driver-passenger model (Peterson et al., 1998, Walker, 1992). This model posits that ecological function resides in “driver” species or in functional groups of such species that have key ecological functions that significantly structure ecosystems, while passenger species are those that have minor ecological impact. Studies done to tease out the influence of these various factors on vaginal community ecology will be important to understanding community stability, resistance, and resilience so that strategies can be developed to maintain human vaginal health and prevent disease.
Vaginal Community Space
The relationships among communities were visualized by principal component analysis and displayed in three-dimensional (3-D) space. The three principle
TABLE A11-1 Median Vaginal pH by Community State Types Within Ethnic Background
| Community State Typesa | ||||||
| CST I (L. crispatus) | CST II (L. gasseri) | CST III (L. iners) | ||||
| Ethnic groups | Subjectsb | Median pH ±MADc | Subjectsb | Median pH ±MADc | Subjectsb | Median pH ±MADc |
| Asian | 24 | 4.4 ± 0.52 | 5 | 4.4 ± 0.44 | 41 | 4.0 ± 0.0 |
| White | 44 | 4.0 ± 0.0 | 8 | 4.7 ± 0.44 | 26 | 4.3 ± 0.30 |
| Black | 23 | 4.0 ± 0.0 | 5 | 5.0 ± 0.0 | 33 | 4.0 ± 0.0 |
| Hispanic | 14 | 4.0 ± 0.0 | 7 | 4.7 ± 0.22 | 35 | 4.4 ± 0.59 |
aCommunity groups are defined as in Figure A11-1.
bTotal number of subjects within a CST.
cMedian absolute deviation.
components explained 82% of the variance. Each point in Figure A11-4 represents the vaginal community of an individual. Communities dominated by species of Lactobacillus and representing CSTs I, II, III, and V are shown at each of the four outer vertices of the tetrahedron, with CST IV at the inner vertex. Communities found on the edges joining two vertices are mixtures of the two Lactobacillus species that dominate the communities found at the corresponding vertices, with an equal proportion of each species at the midpoint of the edge. We refer to each location in this 3-D space as a community state, and one can consider the entire space to represent the plausible alternative community states, or vaginal bacterial community space.
The cross-sectional design of this study with only one sample from each subject precludes knowing whether the locations of these communities in vaginal community space vary over time. Nonetheless, at this stage we can propose four distinct conceptual models for the variation of community composition over time. The first is the “dynamic equilibrium hypothesis” in which the composition of a community is comparatively invariant over time and exists in a single dynamic equilibrium. A second “community space hypothesis” is the opposite of the first, and each community can and does occupy any position in community space over time and throughout a woman’s lifetime. These changes are postulated to occur in response to hormonal cycles, an individual’s habits and practices, changes in diet, or some other ecological force. A third model is an “alternative equilibrium states hypothesis” wherein a woman’s community can change over time, but the number of alternative states are limited in number and governed by unknown factors. A fourth possibility is a “community resilience hypothesis” in which a
| CST IV (Diversity group) | CST V (L. jensenii) | All CSTs | |||
| Subjectsb | Median pH ±MADc | Subjectsb | Median pH ±MADc | Subjectsb | Median pH ±MADc |
| 19 | 5.5 ± 0.44 | 7 | 5.0 ± 0.89 | 96 | 4.4 ± 0.59 |
| 10 | 5.5 ±.74 | 9 | 4.85 ± 0.22 | 97 | 4.2 ± 0.30 |
| 42 | 5.3 ± 0.44 | 1 | 4.7 ± 0.44 | 104 | 4.7 ± 1.04 |
| 37 | 5.3 ± 0.44 | 4 | 5.0 ± 0.59 | 97 | 5.0 ± 074 |
community normally resides in a single region of space. Under this scenario the composition and structure of a vaginal community can change to a transitional state in response to disturbance, but the resistance and resilience of a community determine the extent and duration of a change, while homeostatic mechanisms drive communities back to their “ground state.” We expect that no single hypothesis will explain the dynamics of all communities.
The pH and Nugent scores of each community are depicted in the 3-D community space on Figures A11-4B and A11-4C. The figures show a strong correlation between high pH and high Nugent scores.49 The lowest pH values were associated with community states dominated by L. iners and L. crispatus, and the highest pH values were associated with community states not dominated by species of Lactobacillus. Both Nugent scores and pH values increased as the proportion of non-Lactobacillus sp. increased. This was most readily seen in communities that contained a decreasing proportion of L. iners. Interestingly, elevated pH and high Nugent scores were observed in some communities that have high proportions of Lactobacillus species (also shown in Figure A11-1) suggesting that these metrics cannot be predicted with absolute certainty based solely on the proportion of Lactobacillus in a community. Clearly additional research is needed to understand the various factors that govern vaginal pH.
________________
49 Nugent scores are used to diagnose bacterial vaginosis. The scores range from 0–10 and are based on the weighted counts of different bacterial cell morphologies observed in a Gram-stained smear of a vaginal sample. For further information see Nugent, P. et al. (1991) J Clin Microbiol 29(2), 297–301.
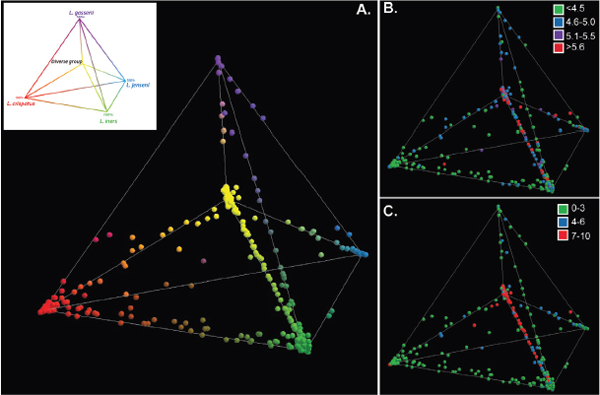
FIGURE A11-4 Relationships among vaginal bacterial communities visualized by principal component analysis in which the relative abundances are expressed as proportions of the total community and displayed in three-dimensional (3-D) space. Communities dominated by species of Lactobacillus and representing CST I, II, III, and V are shown at each of the four outer vertices of the tetrahedron, with CST IV at the inner vertex and shown in the inset. Panel A: Each point corresponds to a single subject and was colored according to the proportions of phylotypes in each community. Panel B: The pH of each vaginal community shown in Panel A. Panel C: The Nugent score category of each vaginal community shown in Panel A.
SOURCE: Modified from Ravel et al. (2011).
By analogy with other ebiological communities, it is reasonable to assume that vaginal microbial communities exist in a state of dynamic equilibrium, and that homeostatic mechanisms exist to provide resilience. Given the fundamental differences in the species composition of these communities, one can speculate that they will differ in terms of number and strength of interspecies interactions. This will in turn have implications for the relative resistance and resilience of each community type to disturbances. If that is the case then invasive species, including both opportunistic and overt pathogens, are more likely to become established in communities that exhibit low stability while the converse will also be true (Hobbs and Huenneke, 1992). This has direct implications for the assessment of susceptibility to infectious disease. Importantly, it also suggests that differences in vaginal bacterial community composition should be taken into account in the estimation of disease risks. This would constitute the first
step toward personalized medicine for women’s reproductive health, wherein differences between the vaginal microbiomes of individuals would be taken into account in risk assessment, and for disease diagnosis and treatment.
Temporal Dynamics of the Vaginal Microbiome
To evaluate the hypotheses described above, we determined the temporal dynamics of vaginal bacterial communities in healthy reproductive-age women in a longitudinal study that was conducted in which 32 women self-collected midvaginal swabs twice-weekly for 16 weeks using a validated self-collection protocol (Gajer et al., 2012). The bacterial diversity in the vaginal communities sampled was determined by pyrosequencing variable regions 1 and 2 (V1–V2) of bacterial rRNA genes. A dataset of 2,522,080 high-quality classifiable 16S rRNA gene sequences was obtained from 937 samples with an average of 2,692±910 (SD) sequences per sample. Using the methods described above the bacterial communities sampled were classified into one of five community state types based on differences in species composition and their relative abundances (Figure A11-5). Three of these CSTs (I–III), were dominated by L. crispatus, L. gasseri, or L. iners, respectively. Unlike the study described above none of the communities examined in this study were dominated by L. jensenii, probably because the low prevalence of this CST and too few women were sampled. Communities that clustered in CST IV-A and IV-B were heterogeneous in composition, lacked significant numbers of Lactobacillus sp. but differed in species composition (Figure A11-5). Communities in CST IV-A were generally characterized by modest proportions of either L. crispatus, L. iners, or other Lactobacillus spp., along with low proportions of various species of anaerobic and strictly anaerobic bacteria such as Anaerococcus, Corynebacterium, Finegoldia, or Streptococcus. In contrast, CST IV-B had higher proportions of the genus Atopobium, in addition to Prevotella, Parvimonas, Sneathia, Gardnerella, Mobiluncus, or Peptoniphilus and several other taxa. Some of the taxa in CST IV-B have previously been shown to be associated with bacterial vaginosis (Fredricks et al., 2005). Overall, high Nugent scores were most often associated with CST IV-B, while low Nugent scores were associated with CST IV-A (Figure A11-6).
Community State Types Transitions
Profiles of community state types were derived from time series of community samples and clustered into five classes of longitudinal dynamics, which we refer to as community classes (Figure A11-6), designated LC, LG, LI, DA, and DB. These classes reflect similarities in how community composition changed over time (Figure A11-6). Nearly all the temporal profiles were complex and somewhat individualized. Not all kinds of transitions between community states were equally likely to occur within a time series, and some were not
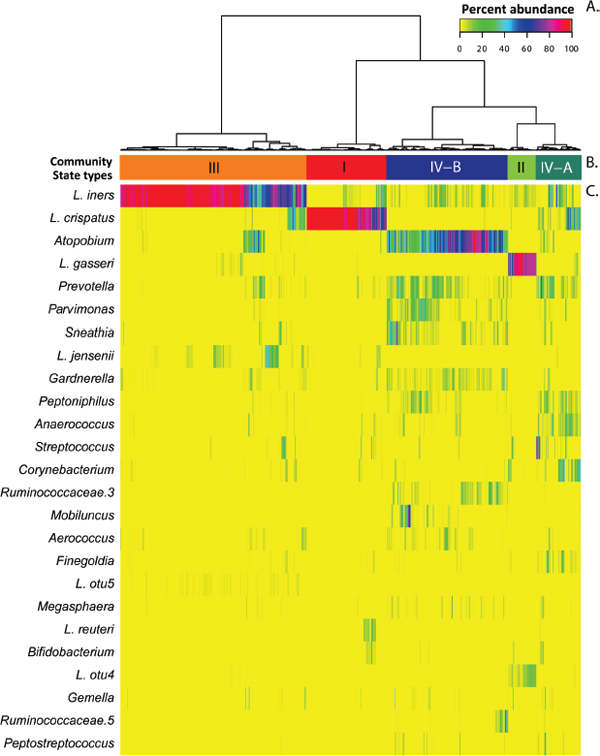
FIGURE A11-5 Assignment of vaginal community state types. (A) Dendogram of Jensen-Shannon distances between bacterial community states in all samples collected twice weekly for 16 weeks among all 32 women. (B) Community state types resulting from clustering analysis wherein each sample was assigned to one of the five community state type (CST I, CST II, CST III, CST IV-A, and CST IV-B), and these are color coded. (C) Heat map of phylotype relative abundances. The color key for abundance is in the top-right corner. NOTE: The 25 most prevalent phylotypes in all communities are listed on the left side of the heat map.
SOURCE: Modified from Gajer et al. (2012).
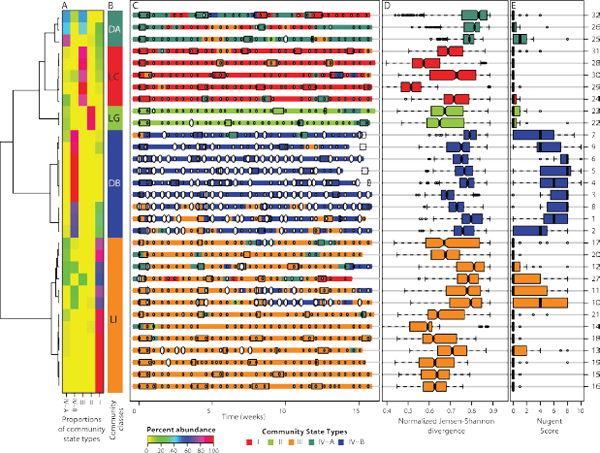
FIGURE A11-6 Dynamics of vaginal community state types in 32 women over 16 weeks. (A) Heat map showing the proportions of community state types (I, II, III, IV-A and IV-B) observed within a woman over time (color key is indicated below panel C) that were used to generate the dendogram that depicts distances between proportions of the five community state types identified. (B) Color bar indicating community class (DA, LC, LG, DB, and LI) as defined by clusters of proportions of community state types within a woman over time. (C) Profiles of community state types, Nugent scores, and menses for 32 women over a 16-week period. Each dot (white or black) represents one sample in the time series. The absence of a dot indicates missing samples. The community state types in the time series for each woman are color-coded according to the schema shown below panel C. Nugent scores is indicated: high Nugent score (7–10, large ovals; intermediate Nugent score (4–6; medium ovals); and low Nugent score (1–3; small ovals). Menses for each woman are indicated by boxes. (D) Box plot of normalized Jensen-Shannon distances between all pairs of community states within each subject. Community deviation from constancy is represented by the Jensen-Shannon Index (black bar, see Supplementary Online Material), with higher values reflecting decreased constancy. (E) Box plot of average Nugent scores for each woman over 16 weeks. Panel D and E, the whiskers represent the lowest and highest datum still within 1.5 interquartile range (IQR) of the lower and upper quartile. The middle 50% of the data is represented by the height of the box.
SOURCE: Modified from Gajer et al. (2012).
observed (Figure A11-7). For example, vaginal communities of CST IV-B often transitioned to CST III, but only rarely to CST I. Likewise, the results show that vaginal communities of CST I, which are dominated by L. crispatus, most often transition to become community CST III, which are dominated by L. iners, or CST IV-A. Of note, CST III were two times more likely to switch to CST IV-B than to CST IV-A. The community states of CST II are dominated by L. gasseri and rarely underwent transitions to other CST. Conversely, we observed notransitions from CST I to CST II, and there were only two transitions from CST III to CST II.
Index of vaginal bacterial community stability By definition, communities that persist in the same state type over time display a high level of stability, while those that often transition to different state types have low levels of stability. We introduce the normalized Jensen-Shannon divergence index,50 defined as the median of Jensen-Shannon distances between all pairs of community states, which provides a quantitative measure of community stability (Figure A11-6E). As the index becomes smaller less change is observed between different community states, and the community is more constant over time. The vaginal bacterial communities of subjects 3, 4, 5, and 6 were of class DB, and displayed higher levels of overall constancy and persistently high Nugent score even though the subjects did not report any BV symptoms. This calls into question current widely held views on what is considered to be a “normal” vaginal bacterial community that are founded on the premise that the communities of healthy women must contain high proportions of Lactobacillus species (Srinivasan and Fredricks, 2008). This premise is also undermined by the observation that vaginal bacterial communities of subjects 32, 26, and 25 of class DA and subjects 30, 24, and 31 of class LC, which had high Jensen-Shannon indices yet persistently low Nugent scores. Thus, highly variable vaginal bacterial communities do not always have persistently high Nugent scores, so variation per se does not always equate with disease. These results suggest that neither variation in community composition, nor constantly high levels of apparent diversity (codominance) are necessarily indicative of dysbiosis. The meaning and implications of these observations will remain unknown until molecular characterization of vaginal microbiota is applied to studies of adverse outcomes.
Lactobacillus sp. and Vaginal Bacterial Community Stability
The stability of vaginal communities was associated with certain species of lactic acid bacteria that dominated a community. For example, the vaginal communities of women in community classes LC (five women) and LG (two women)
________________
50 The Jensen-Shannon divergence index reflects the similarity between two probability distributions. In this case it was used to quantitatively compare the kinds and abundances of bacterial phylotypes in consecutive pairs of samples.
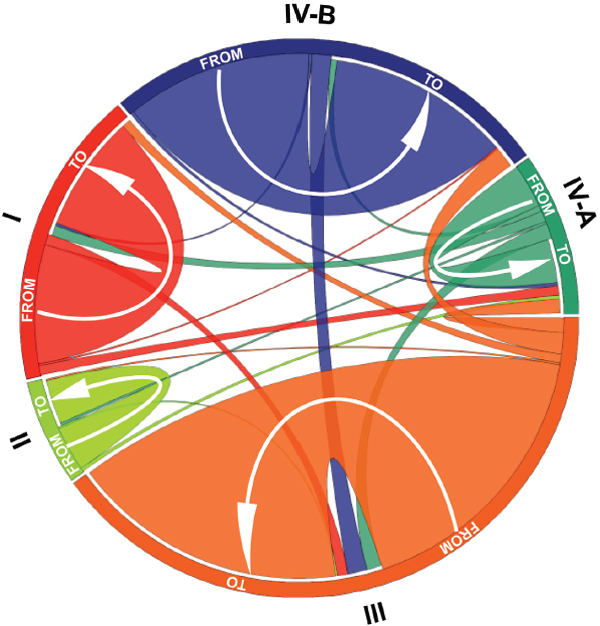
FIGURE A11-7 Graphical representation of community state type transitions observed between all consecutive pairs of time points (905 transitions) and their frequencies among all women. For each community state type, the length of the outer arcs represents the proportion of each community state type among all women combined. The width of the inner arcs (at its base) represents the frequency of transition to the same community state type or to another community state type. The directionality of the transition is indicated by the labels FROM and TO as well by an arrow for transition to the same community state type. SOURCE: Modified from Gajer et al. (2012).
were most often dominated by L. crispatus and L. gasseri, respectively. These communities were rather stable, exhibited fewer transitions between community states, and typically had low Nugent scores. In these women most transitions between community states were associated with menses (Figure A11-6). For example, in subject 28 (Figure A11-8A), a community dominated by L. crispatus was resilient, being replaced by a community dominated by L. iners during
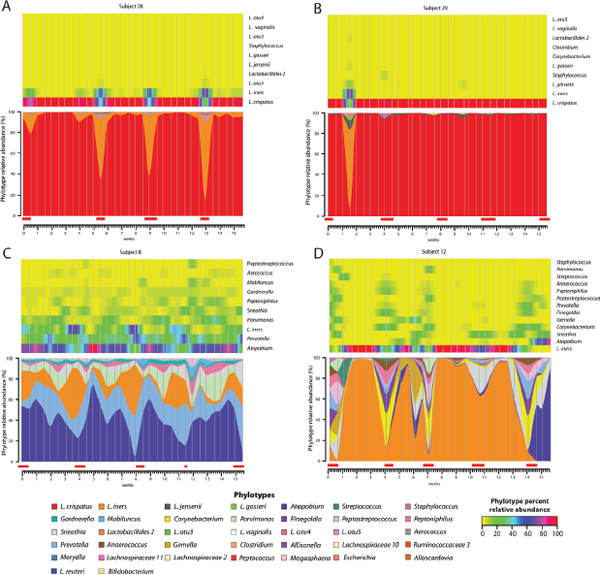
FIGURE A11-8 (A–D) Heat maps (top) and interpolated bar plots (bottom) of phylotype relative abundance observed in four selected subjects over 16 weeks (heat map color key is indicated in the lower-right corner).
NOTE: Color-codes for each phylotype represented in the interpolated bar plots are shown below the figure. Red dots below the interpolated bar graphs represent menstruation days.
SOURCE: Modified from Gajer et al. (2012).
menses, which then reverted to a community dominated by L. crispatus at the end of menses. Communities of class LI (14 women) were typically dominated by L. iners, but varied widely in terms of species composition and stability. This is illustrated by the communities of subjects 14, 15, 16, 18, and 19 that appeared rather stable over time, while others such as 11 and 27 commonly shifted to different community types that were more often associated with higher Nugent scores. These differences might reflect genomic heterogeneity in the dominating Lactobacillus sp.
The rapid and sometimes extensive turnover of human vaginal communities was visualized by mapping temporal changes in community composition onto the 3-D community space defined previously in a cross-sectional study described above of vaginal communities in 396 women (Ravel et al., 2011) (Figure A11-9). These illustrations show that while the vaginal community composition of many
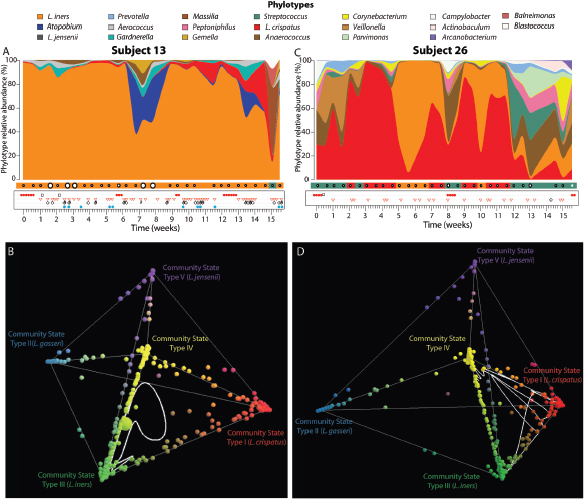
FIGURE A11-9 Temporal dynamics of vaginal bacterial communities in two women over 16 weeks. (A, C) Interpolated bar graph of phylotype relative abundance for subjects 13 (A) and 26 (C). Profiles of community state types in which Nugent scores have been superimposed (high Nugent score, 7–10, large open circles; intermediate Nugent score, 4–6, medium open circles; and low Nugent score, 1–3, small open circles) are shown below the interpolated bar graphs. Daily metadata is represented by the following: red balls, menstruation; black open square, douching; open red triangle, vaginal intercourse; open black diamond, oral sex; vertical black bar, digital penetration (insertion of finger(s) in the vagina); light blue closed circle, lubricant use. (B, D) Representation of vaginal community dynamics in 3-D community space (3) for subjects 13 (B) and 26 (D). Communities dominated by species of Lactobacillus and representing CST I (red), II (light blue), III (green), and V (purple) are shown at each of the four outer vertices of the tetrahedron, with CST IV (yellow) at the inner vertex. The white line represents the succession of community states for each subject over time.
SOURCE: Modified from Gajer et al. (2012).
individuals changed over time the alternatives were often restricted to specific community states. This is consistent with the notion that vaginal communities can exist in alternative equilibrium states. In contrast, communities of most other women evinced dynamics consistent with the “community resilience hypothesis,” in which a community, though dynamic, normally resides in a single region of space. In these cases the composition and structure of a community occasionally changed to a transition state, but they were resilient, and homeostatic mechanisms returned them to their “ground state.”
In many women the bacterial species composition and rank abundances of bacterial species changed markedly over short periods of time (Figure A11-8D for example). Consistent with this, the communities of some women seemed resilient and showed simple and predictable changes between community states that occurred only during menses (e.g., subjects 28, 12, and 19). In contrast the communities of other women were almost invariant during menses (e.g., subject 29), while still others changed states continuously over time regardless of whether the subjects were menstruating or not (e.g., subjects 1, 2, 7, and 27). The latter might be examples of communities transitioning to alternative equilibrium states. Most of the transitions to other state types were, however, transient in nature with 35% of all state types persisting for not more than a week. This is consistent with previous studies that showed significant yet short-lived changes in Nugent scores (Brotman et al., 2010; Schwebke et al., 1999) and with daily fluctuations in the composition of the vaginal microbiota that have been previously documented by microscopy (Brotman et al., 2010; Hay et al., 1997; Keane et al., 1997; Schwebke et al., 1999).
Identifying Factors of Stability Disturbance
In an effort to evaluate the dependence of stability of vaginal bacterial communities on the time in the menstrual cycle and other time-varying factors, we modeled the log Jensen-Shannon divergence rate of change over normalized menstrual cycles using a linear mixed effects model with a Fourier polynomial of the normalized menstrual time component adjusted for hormonal contraception, community type, sexual activities, lubricant use and douching (with the last three evaluated 1 day prior to sampling), and with subject-based degree two polynomial random effects to account for correlation in repeated measurements on each subject (Figure A11-10). The Fourier polynomial captured population-level dependence of the log Jensen-Shannon divergence rate of change (i.e., community deviation from constancy) on the time in the menstrual cycle. The lowest constancy was associated with menses. Interestingly, the two minima of the population-level Jensen-Shannon divergence rate of change (indicating highest community constancy) coincided with the two maxima of estradiol concentration as reported by Minassian et al. (1993), while the maximum of progesterone concentration coincided with the second minimum of the population-level constancy
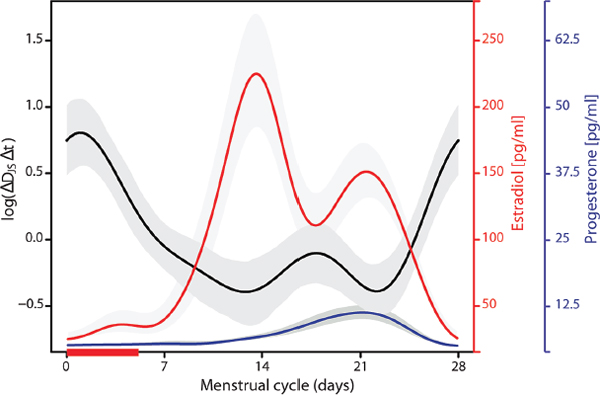
FIGURE A11-10 Modeling the dependence of the log of Jensen-Shannon divergence rate of change over the menstrual cycle. The length of menstrual cycles within and between women were normalized to 28 days with day 1 to 5 corresponding to menses (red bar). The red line shows concentrations of estradiol as a function of menstrual time (data from Brotman et al., 2008) and the blue line shows concentrations of progesterone (data from Brotman et al., 2008). The shaded areas around each curve show 95% point-wise confidence bands.
SOURCE: From Gajer et al. (2012).
function (Figure A11-10). Of all the metadata evaluated in the model, sexual activity was the only one that had a significant (negative) effect on constancy independent of time in the menstrual cycle, but its effect was rather weak in comparison to that of community class.
Discussion
A Need for Personalized Medicine?
The species that make up vaginal microbial communities vary between individuals as do the dynamics of these communities. The differences between individuals can be demonstrated by what has been learned about the vaginal microbiome wherein at least five bacterial community state types are common in healthy reproductive age women, and the frequencies of these types vary among women of different ethnic groups. These communities remain dynamic within an individual,
and dramatic changes, which seem highly individualized in the species composition, can occur over short time scales. Because differences exist among individuals, different individuals can be affected differently by common risks. Hence, the rate of disease occurrence and the efficacy of various treatments may not be pertinent to some individuals in a population since the conclusions are often drawn from studies based on a population as a whole. The reasons for these differences in composition and variance in stability are unknown. Among the possible causes are differences that exist among host’s medical history, diet, genotype, ecological interactions of bacteria within hosts, and a plethora of other factors just now being discovered, as well as purely stochastic factors. Further research is needed to characterize heterogeneity in the microbiomes of the vagina, especially at a functional level, which will lead to a better understanding of disease risks for specific individuals and avoid the potential pitfalls of population-based studies. By doing so it will be possible to more reliably identify individuals at risk to bacterial vaginosis, various sexually transmitted diseases, and ascending urogenital infections.
In the data described here and reported previously (Ravel et al., 2011; Gajer et al., 2012), considerable variability exists within the definition of a “normal” vaginal microbiome. When surveyed cross-sectionally, it appears that several distinct “community state types” exist in normal, otherwise healthy women, each with a markedly different bacterial species composition and pH profile, and that the prevalence of each varies with ethnicity (Ravel et al., 2011). Nonetheless, the vaginal communities of women in the four different ethnic groups studied (Asian, Hispanic, white, and black) could be clustered into five different CSTs based on the numerically dominant species in the vaginal communities. Four of the CSTs were dominated by one of four Lactobacillus species (L. iners, L. crispatus, L. gasseri, and L. jensenii), while communities in the fifth CST included a diverse array of strict and facultative anaerobes and lower proportions of lactobacilli (Ravel et al., 2011). This group accounted for slightly over 25% of the women studied, a notable finding given the paradigmatic view that high proportions of Lactobacillus are prerequisite for vaginal health and “normality.” Indeed, the inherent differences within and between women in different ethnic groups strongly argues for a more refined definition of the kinds of bacterial communities normally found in healthy women, and the need to appreciate differences between individuals so they can be taken into account in risk assessment and disease diagnosis. Indeed the variability seen in the composition of the vaginal microbiome between healthy women and over a woman’s life span beg for us to reassess and broaden our thinking of what constitutes “normal” or at the very least to accept that “normal” is a continuum and not a categorical variable.
If the maintenance of a low pH is indeed a key function of the vaginal microbial community, then perhaps it may be more appropriate to consider all lactic acid bacteria found in vaginal communities as members of the same ecological “guild” (Jaksic, 1981) since they all rely on the same resource pool to fulfill the same ecological niche. If this is the case, then the prevailing view that
species of Lactobacillus are both necessary and sufficient for maintaining health may be overly simplistic because functionally equivalent species may “substitute” for one another. With this in mind these types of vaginal bacterial communities might be considered normal and healthy if signs and symptoms of disease are absent even though the composition of these communities closely resembles those associated with symptomatic bacterial vaginosis.
Ecological Dynamics of the Vaginal Microbiota
The dynamics of vaginal communities were found to vary widely among women even among those that clustered in the same community class, and the communities of some women were shown to have low constancy and high levels of species turnover. These findings suggest that point estimates of community composition that arise from cross-sectional studies could be misleading for most women because they experience a distribution of community states over time I. This calls into question whether data from cross-sectional studies can be reliably used to link vaginal bacterial community composition to health outcomes. Intervals of increased susceptibility to disease may occur because vaginal microbiota fluctuate.
Fluctuation in community composition and constancy appear to be mainly affected by time in the menstrual cycle, community class, and to a certain extent by sexual activity. Other unknown or less well-studied factors such as host diet and the state of the innate immune system are almost certainly at play as well. It should be noted that in many cases community function is probably maintained despite changes in community composition, because shifts in community structure often only involved changes in the relative dominance of a limited number of different lactic acid bacterial species. Such fluctuations could occur while maintaining community performance (e.g., lactic acid production) when there is functional redundancy among community members and shifts in the relative abundances of guild members occur due to changes in environmental conditions that favor one population over another. This coupled with the observation that a limited number of community types are found in women of different ethnicities suggest that deterministic processes driven by interspecies interaction, host–microbe interaction, and niche partitioning account for vaginal community composition. In some cases changes in community performance occur as a result of changes in community composition. These are reflected in the shift in composition of the metabolome,51 which in this strictly anaerobic environment is dominated by fermentation products such as lactic, succinic, and acetic acids that accumulate in vaginal secretions (Gajer et al., 2012).
________________
51 A metabolome is comprised of all the intermediates and products of metabolism that are present in a biological sample (cell, tissue, organ, or organism).
The causes of shifts in community structure and function are unknown. However the common occurrence of highly abundant lactic acid bacteria and the pronounced effect they have on the vaginal environmental in terms of pH is consistent with Walker’s driver-passenger model (Walker, 1995). If the role of lactic acid bacteria is to create and maintain the low pH environment to protect from unwanted microbial colonization, then these lactic acid bacteria could be considered the “drivers” of the vaginal environment because they set the stage for other microbial populations who must be able to withstand a pH of around 4.0 to 4.5. In this way the lactic acid bacteria act to stabilize communities. Anaerobic non-lactic acid bacteria would be considered the “passengers,” as their presence would depend on their ability to conform to the environment created by the “drivers.” These members, typically present at much lower abundance, would have much less influence on the ecosystem, and may even be lost over time without markedly affecting the community’s function. At least from a numerical perspective vaginal communities seem to conform to this model since the rank abundance of species is highly skewed, and lactic acid bacteria often outnumber others by at least an order of magnitude. However, changes in environmental conditions that favor the outgrowth of “passengers” might place them in the driver’s seat, and they might cause the demise of lactic acid bacteria via their metabolic activity or the production of antimicrobial compounds. Such marked changes in community structure might be precipitated by changes in the kinds and amounts of resources available to the bacteria by the host, disturbances of the community induced by various habits and practices (e.g., douching), or changes in host immune status. Changes in the microbiome of this sort might account for some of the symptoms classically associated with bacterial vaginosis, which include an elevated pH and the malodor of vaginal secretions that result from metabolic by-products such as amines and short-chain fatty acids (Amsel et al., 1983). The change in environmental pH and accumulation of metabolic by-products may make the environment less hospitable for lactic acid bacteria allowing anaerobic bacteria to displace them as dominant community members.
Community Stability: Resistance Versus Resilience
Vaginal bacterial communities that differ in species composition are expected to differ in their resilience and resistance to environmental change, and this is manifest in the degree of stability that each exhibits over time. The resistance of a community is a measure of its ability to resist changes in response to a disturbance event, and it is measured by the amount of change the community can withstand without having an impact on community function (McCann, 2000). Resilience, on the other hand, refers to a community’s ability to recover and return to a quasi-stable state following a disturbance. It is a measure of how often or how strong a disturbance must be to actually alter community function (Gunderson, 2000; McCann, 2000). Both aspects contribute to overall community stability,
and are heavily dependent on various characteristics of the underlying microbial network, such as their ability to tolerate various types of stresses, the strength and types of interactions present, and the degree of functional redundancy existing within the community (Hobbs and Huenneke, 1998). Those systems with greater degrees of functional redundancy in roles known to stabilize or “drive” the community, tend to have greater buffering to disturbances. This insurance hypothesis (McCann, 2000) suggests that more diverse communities are more likely to exhibit functional redundancy and be more stable in terms of their response to environmental perturbations.
Ecologists have long known that the biological communities of disturbed ecosystems are more susceptible to invasion by nonindigenous species, and the bacterial communities of the vaginal microbiota are probably no exception. Hence if the resistance or resilience of a vaginal community are low, then transitory changes to the structure of these communities may occur more readily in response to disturbances making these disturbed communities more susceptible to invasion by nonindigenous species that might include transient species of fecal origin and opportunistic pathogens (Hobbs and Huenneke, 1992). It should be kept in mind that disturbance events that cause shifts in population densities with concomitant changes in community function can vary in intensity, frequency, and duration (White and Jentsch, 2001), and whether an event will actually alter community composition or structure depends on characteristics of the community itself. For instance, communities with low levels of resistance and resilience may be disturbed by a single but intense event of relatively short duration that results in a transient shift in community structure that temporarily increases the community’s susceptibility to invasion. More robust communities, however, may be able to retain community structure and function, despite more frequent events of low to moderate intensity. Given this, we postulate that stability and resilience of vaginal bacterial communities is likely to vary widely since the species composition and structure of these communities differs among women and this variance in turn may account for differences in the susceptibility of individuals to urogenital infectious diseases. This is important since vaginal communities are continually subjected to a wide range of potential acute and chronic disturbances related to human activities, such as the use of various birth control methods, antibiotics, and sexual intercourse, as well as natural “disturbances” such as hormonal fluctuations (Eschenbach et al., 2000), aging (Larsen et al., 1982), and stress (Culhane et al., 2002; Nansel et al., 2006) of which may in turn affect their ability to maintain human health.
Community Dynamics and Stability
Most vaginal microbiome studies done to date have employed cross-sectional designs, wherein samples are obtained from some number of individuals at a single point in time or with multiple sampling points separated by relatively
long intervals (weeks or months). While these studies have yielded valuable information in terms of species composition, they do not allow for any assessment of community stability, therefore painting an incomplete picture of vaginal microbial ecology. This reliance on data from cross-sectional studies is surprising since daily fluctuations in the composition of the vaginal microbiota have been previously documented by microscopy and other means. The data presented here shows that nearly all the vaginal communities studied exhibit some degree of variability, with some changing markedly over a short time and others remaining relatively constant. Usually these shifts involved changes in the relative proportions of species present, but in some cases, a distinct and persistent turnover in species composition occurred, marking the presence of an apparent alternative equilibrium state. Several factors may have contributed to these differing levels of community stability. For instance, menses was identified as having the largest effect on changes in community composition, while communities were less variable during periods of the menstrual cycle marked by high levels of estrogen (late follicular phase) or estrogen and progesterone (luteal phase; Gajer et al., 2012). These findings highlight the potential of prospective longitudinal studies to identify causes of community instability that are associated with increased risk to infectious disease and the community disturbances that give rise to bacterial vaginosis.
Prospective
Host–microbe interactions are notoriously complex, yet in the case of the vaginal microbiome there has been a willingness to accept that highly abundant lactobacilli and an acidic environment (< 4.5) are both necessary and sufficient for vaginal health. However, the evidence from recent studies suggests there is a need to critically reassess this common wisdom because the vaginal communities of a large fraction of asymptomatic, healthy, reproductive-age women (~25%) are not dominated by species of Lactobacillus; an observation that is equally true for apparently the majority of healthy premenarcheal and postmenopausal females. Likewise, the vaginal pH of a large proportion of reproductive age women (especially Hispanics and blacks) as well as that of premenarcheal and postmenopausal females is greater than 5.0. These facts indicate that our understanding of other processes and mechanisms that contribute to avoiding infectious disease and the maintenance of health is incomplete.
Certainly there is compelling evidence that the low pH resulting from lactic acid production is important to precluding the colonization of nonindigenous organisms in the vagina. However, lactic acid may have other roles as well. For example, lactic acid itself is known to antimicrobial activity and has been shown to be more effective than acidity alone as a microbicide against HIV and pathogens like Neisseria gonorrhea (Graver and Wade, 2011). Furthermore, Witkin et al. and others have shown that L-lactic acid, in addition to its role in influencing
vaginal acidity, stimulates the IL-23/IL-17 T lymphocyte pathway (Shime et al., 2008; Witkin et al., 2011), and induces the production of pro-inflammatory cytokines by vaginal epithelial cells in the presence of a synthetic viral RNA (Mossop et al., 2011), induction of tumor angiogenesis (Vegran et al., 2011), lymphocyte activation (Murray et al., 2005), and inhibition of bacterial growth (Alakomi et al., 2000; O’Hanlon et al., 2011). Witkin et al. suggest that lactic acid may have stimulatory effects on the host innate defense system by enhancing cytokine release in response to lipopolysaccharide (LPS) exposure (Witkin et al., 2011). In these studies, lactic acid enhanced the release of IL-23 by peripheral monocytes and macrophages in response to LPS exposure (Witkin et al., 2011). Given that IL-23 is one of the primary cytokines involved in neutrophil recruitment, mobilization, and activation at mucosal surfaces, this is thought to translate to better immune surveillance of mucosal surfaces characterized by high lactic acid levels in vivo (Witkin et al., 2011). This is consistent with the findings of Fichorova et al. (2011) that colonization of vaginal epithelial cell monolayers with common bacteria such as L. crispatus, Prevotella bivia, and Atopobium vaginae may regulate the epithelial innate immunity in a species-specific manner. Finally, recent studies by Witkin et al. (mBio, in press) provided evidence that the ratio of D- and L-lactate isomers influences the expression of matrix metalloproteinase (MMP-8) production by controlling extracellular matrix metalloproteinase inducer (EMMPRIN) levels. Based on these studies it is plausible to suggest that in addition to lowering the environmental pH lactic acid, it also serves as a signaling molecule that influences host gene expression.
It seems likely that products of bacterial species other than lactobacilli will also contribute to the maintenance of host vaginal health in humans as well as in other host species by influencing influence host gene expression. These might include other low molecular weight metabolites, including various short-chain fatty acids that are produced by strict anaerobes that inhabit the vagina. This is an appealing idea because vaginal bacterial species have coevolved with their hosts over time, and it seems likely that they collectively provide benefits to the host as part of a complex mutualistic relationship. Perhaps future studies should move away from the perhaps overly simplistic notion that lactobacilli are “good” simply because they lower the vaginal pH, and all other species are inconsequential at best, or at worst opportunistic pathogens.
Acknowledgments
This work was in part supported by the National Institute of Allergies and Infectious Diseases, National Institutes of Health (grant numbers U19 AI084044, UO1 AI070921 and UH2AI083264) and a Center of Biomedical Research Excellence grant (P30 GM103324) from the National Institute for General Medical Sciences.
References
Alakomi, H-L., E. Skytta, M. Saarela, T. Mattila-Sandholm, K. Latva-Kala, and I. M. Helander. 2000. Lactic acid permeabilizes gram-negative bacteria by disrupting the outer membrane. Applied and Environmental Microbiology 66:2001-2005.
Amsel, R., P. A. Totten, C. A. Spiegel, K. C. Chen, D. Eschenbach, and K. K. Holmes. 1983. Nonspecific vaginitis. Diagnostic criteria and microbial and epidemiologic associations. American Journal of Medicine 74(1):14-22.
Antonio, M. A., S. E. Hawes, and S. L. Hillier. 1999. The identification of vaginal Lactobacillus species and the demographic and microbiologic characteristics of women colonized by these species. Journal of Infectious Diseases 180(6):1950-1956.
Aroutcheva, A., D. Gariti, M. Simon, S. Shott, J. Faro, J. A. Simoes, A. Gurguis, and S. Faro. 2001. Defense factors of vaginal lactobacilli. American Journal of Obstetrics and Gynecology 185(2):375-379.
Brotman, R. M., K. G. Ghanem, M. A. Klebanoff, T. E. Taha, D. O. Scharfstein, and J. M. Zenilman. 2008. The effect of vaginal douching cessation on bacterial vaginosis: A pilot study. American Journal of Obstetrics and Gynecology 198(6):628.e1–628.e7.
Brotman, R. M., J. Ravel, R. A. Cone, and J. M. Zenilman. 2010. Rapid fluctuation of the vaginal microbiota measured by Gram stain analysis. Sexually Transmitted Infections 86(4):297-302.
Collins, M. D., and S. Wallbanks. 1992. Comparative sequence analyses of the 16S rRNA genes of Lactobacillus minutus, Lactobacillus rimae and Streptococcus parvulus: proposal for the creation of a new genus Atopobium. FEMS Microbiology Letters 74(2-3):235-240.
Culhane, J. F., V. Rauh, K. F. McCollum, I. T. Elo, and V. Hogan. 2002. Exposure to chronic stress and ethnic differences in rates of bacterial vaginosis among pregnant women. American Journal of Obstetrics and Gynecology 187(5):1272-1276.
Danielsson, D., P. K. Teigen, and H. Moi. 2011. The genital econiche: focus on microbiota and bacterial vaginosis. Annals of the New York Academy of Sciences 1230:48-58.
Döderlein A. 1892. Das scheidensekret und seine bedeutung fur puerperalfieber. Zentralblatt fur Bakteriology 11:699.
Dominguez-Bello, M. G., E. K. Costello, M. Contreras, M. Magris, G. Hidalgo, N. Fierer, and R. Knight. 2010. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proceedings of the National Academy of Sciences of the United States of America 107(26):11971-11975.
Du Plessis, E. M., and L. M. Dicks. 1995. Evaluation of random amplified polymorphic DNA (RAPD)-PCR as a method to differentiate Lactobacillus acidophilus, Lactobacillus crispatus, Lactobacillus amylovorus, Lactobacillus gallinarum, Lactobacillus gasseri, and Lactobacillus johnsonii. Current Microbiology 31(2):114-118.
Eschenbach, D. A., S. S. Thwin, D. L. Patton, T. M. Hooton, A. E. Stapleton, K. Agnew, C. Winter, A. Meier, and W. E. Stamm. 2000. Influence of the normal menstrual cycle on vaginal tissue, discharge, and microflora. Clinical Infectious Diseases 30(6):901-907.
Farage, M., and H. Maibach. 2006. Lifetime changes in the vulva and vagina. Archives of Gynecology and Obstetrics 273(4):195-202.
Ferris, M. J., A. Masztal, K. E. Aldridge, J. D. Fortenberry, P. L. Fidel Jr., and D. H. Martin. 2004. Association of Atopobium vaginae, a recently described metronidazole resistant anaerobe, with bacterial vaginosis. BMC Infectious Disease 4:5.
Ferris, M. J., J. Norori, M. Zozaya-Hinchliffe, and D. H. Martin. 2007. Cultivation-independent analysis of changes in bacterial vaginosis flora following metronidazole treatment. Journal of Clinical Microbiology 45:1016–1018.
Fichorova, R. N., H. S. Yamamoto, M. L. Delaney, A. B. Onderdonk, and G. F. Doncel. 2011. Novel vaginal microflora colonization model providing new insight into microbicide mechanism of action. MBio 2(6):e00168-00111.
Fredricks, D. N., T. L. Fiedler, and J. M. Marrazzo. 2005. Molecular identification of bacteria associated with bacterial vaginosis. New England Journal of Medicine 353(18):1899-1911.
Gajer, P., R. M. Brotman, G. Bai, J. Sakamoto, U. M. Schutte, X. Zhong, S. S. Koenig, L. Fu, Z. S. Ma, X. Zhou, Z. Abdo, L. J. Forney, and J. Ravel. 2012. Temporal dynamics of the human vaginal microbiota. Science Translational Medicine 4(132):132ra152.
Graver, M. A., and J. J. Wade. 2011. The role of acidification in the inhibition of Neisseria gonorrhoeae by vaginal lactobacilli during anaerobic growth. Annals of Clinical Microbiology and Antimicrobials 10:8.
Gunderson, L. H. 2000. Ecological resilience—in theory and application. Annual Review of Ecology and Systematics 31(1):425-439.
Hamady, M., and R. Knight. 2009. Microbial community profiling for human microbiome projects: Tools, techniques, and challenges. Genome Research 19(7):1141-1152.
Hawes, S. E., S. L. Hillier, J. Benedetti, C. E. Stevens, L. A. Koutsky, P. Wolner-Hanssen, and K. K. Holmes. 1996. Hydrogen peroxide-producing lactobacilli and acquisition of vaginal infections. Journal of Infectious Diseases 174(5):1058-1063.
Hay, P. E., A. Ugwumadu, and J. Chowns. 1997. Sex, thrush and bacterial vaginosis. International Journal of STD & AIDS 8(10):603-608.
Hobbs, R. J., and L. F. Huenneke. 1992. Disturbance, Diversity, and Invasion: Implications for Conservation. Conservation Biology 6(3):324-337.
Jaksić, F. M. 1981. Abuse and Misuse of the Term “Guild” in Ecological Studies. Oikos 37(3):397-400. Keane, F. E., C. A. Ison, and D. Taylor-Robinson. 1997. A longitudinal study of the vaginal flora over a menstrual cycle. International Journal of STD & AIDS 8(8):489-494.
Klebanoff, S. J., S. L. Hillier, D. A. Eschenbach, and A. M. Waltersdorph. 1991. Control of the microbial flora of the vagina by H2O2-generating lactobacilli. Journal of Infectious Diseases 164(1):94-100.
Koenig, J. E., A. Spor, N. Scalfone, A. D. Fricker, J. Stombaugh, R. Knight, L. T. Angenent, and R. E. Ley. 2011. Succession of microbial consortia in the developing infant gut microbiome. Proceedings of the National Academy of Sciences of the United States of America 108 Suppl 1:4578-4585.
Konopka, A. 2009. What is microbial community ecology? ISME J 3(11):1223-1230.
Larsen, B., and R. P. Galask. 1982. Vaginal microbial flora: composition and influences of host physiology. Annals of Internal Medicine 96(6 Pt 2):926-930.
Lauer, E., C. Helming, O. Kandler. 1980. Heterogeneity of the species Lactobacillus acidophilus (Moro) Hansen and Mocquot as revealed by biochemical characteristics and DNA-DNA hybridization. Zentbl Bacteriol Mikrobial Hyg Abt 1:150-168.
Levison, M. E., L. C. Corman, E. R. Carrington, and D. Kaye. 1977. Quantitative microflora of the vagina. American Journal of Obstetrics and Gynecology 127(1):80-85.
Linhares, I. M., P. R. Summers, B. Larsen, P. C. Giraldo, and S. S. Witkin. 2011. Contemporary perspectives on vaginal pH and lactobacilli. American Journal of Obstetrics and Gynecology 204(2):120 e121-125.
Martin, D. H. 2012. The microbiota of the vagina and its influence on women’s health and disease. American Journal of the Medical Sciences 343(1):2-9.
McCann, K. S. 2000. The diversity-stability debate. Nature 405(6783):228-233.
Minassian, S. S., and C. H. Wu. 1993. Free and protein-bound progesterone during normal and luteal phase defective cycles. International Journal of Gynaecology and Obstetrics 43(2):163-168.
Mossop, H., I. M. Linhares, A. M. Bongiovanni, W. J. Ledger, and S. S. Witkin. 2011. Influence of lactic acid on endogenous and viral RNA-induced immune mediator production by vaginal epithelial cells. Obstetrics & Gynecology 118:840-846.
Murray, C. M., R. Hutchinson, J. R. Bantick, G. P. Belfield, A. D. Benjamin, D. Brazma, R. V. Bundick, I. D. Cook, R. I. Craggs, S. Edwards, L. R. Evans, R. Harrison, E. Holness, A. P. Jackson, C. G. Jackson, L. P. Kingston, M. W. D. Perry, A. R. J. Ross, P. A. Rugman, S. S. Sdhu, M. Sullivan, D. A. Taylor-Fishwick, P. C. Walker, Y. M. Whitehead, D. J. Wilkinson, A. Wrigth, and D. K. Donald. Monocarboxylate transporter MCT1 is a target for immunosuppression. Nature Chemical Biology 1:371-376.
Nansel, T. R., M. A. Riggs, K. F. Yu, W. W. Andrews, J. R. Schwebke, and M. A. Klebanoff. 2006. The association of psychosocial stress and bacterial vaginosis in a longitudinal cohort. American Journal of Obstetrics and Gynecology 194(2):381-386.
O’Hanlon, D. E., T. R. Moench, and R. A. Cone. 2011. In vaginal fluid, bacteria associated with bacterial vaginosis can be suppressed with lactic acid but not hydrogen peroxide. BMC Infectious Diseases 11:200.
Oscáriz, J. C., and A. G. Pisabarro. 2001. Classification and mode of action of membrane-active bacteriocins produced by gram-positive bacteria. International Microbiology 4(1):13-19.
Pavlova, S. I., A. O. Kilic, S. S. Kilic, J. S. So, M. E. Nader-Macias, J. A. Simoes, and L. Tao. 2002. Genetic diversity of vaginal lactobacilli from women in different countries based on 16S rRNA gene sequences. Journal of Applied Microbiology 92(3):451-459.
Peterson, G., C. R. Allen, and C. S. Holling. 1998. Ecological Resilience, Biodiversity, and Scale. Ecosystems 1(1):6-18.
Ravel, J., P. Gajer, Z. Abdo, G. M. Schneider, S. S. Koenig, S. L. McCulle, S. Karlebach, R. Gorle, J. Russell, C. O. Tacket, R. M. Brotman, C. C. Davis, K. Ault, L. Peralta, and L. J. Forney. 2011. Vaginal microbiome of reproductive-age women. Proceedings of the National Academy of Sciences of the United States of America 108 Suppl 1:4680-4687.
Reid, G., J. A. McGroarty, L. Tomeczek, and A. W. Bruce. 1996. Identification and plasmid profiles of Lactobacillus species from the vagina of 100 healthy women. FEMS Immunology and Medical Microbiology 15(1):23-26.
Rogosa, M., and M. E. Sharpe. 1960. Species differentiation of human vaginal lactobacilli. Journal of General Microbiology 23:197-201.
Schleifer K. H. and W. Ludwig. 1995. Phylogeny of the genus Lactobacillus and related genera. Systematic and Applied Microbiology 18:461-467.
Schwebke, J. R. 2009. New concepts in the etiology of bacterial vaginosis. Current Infectious Disease Reports 11(2):143-147.
Schwebke, J. R., C. M. Richey, and H. L. Weiss. 1999. Correlation of behaviors with microbiological changes in vaginal flora. Journal of Infectious Diseases 180(5):1632-1636.
Shime, H., M. Yabu, T. Akazawa, K. Kodama, M. Matsumoto, T. Seya, and N. Inoue. 2008. Tumor-secreted lactic acid promotes IL-23/Il-17 proinflammatory pathway. Journal of Immunology 180: 7175-7183.
Srinivasan, S., and D. N. Fredricks. 2008. The Human Vaginal Bacterial Biota and Bacterial Vaginosis. Interdisciplinary Perspectives on Infectious Diseases 2008.
Vegran, F. R. Boidot, C. Michiels, P. Sonveaux, and O. Feron. 2011. Lactate influx through the endothelial cell monocarboxylate transporter MCT1 supports an NF-B/IL-8 pathway that drives tumor angiogenesis. Cancer Research 71(7):2550-2560.
Verhelst, R., H. Verstraelen, G., Claeys, G. Verschraegen, J. Delanghe, L. Van Simaey, C. De Ganck, M. Temmerman, and M. Vaneechoutte..2004. Cloning of 16S rRNA genes amplified from normal and disturbed vaginal microflora suggests a strong association between Atopobium vaginae, Gardnerella vaginalis and bacterial vaginosis. BMC Microbiology 4:16.
Verstraelen, H., R. Verhelst, G. Claeys, M. Temmerman, and M. Vaneechoutte. 2004. Culture-independent analysis of vaginal microflora: The unrecognized association of Atopobium vaginae with bacterial vaginosis. American Journal of Obstetrics and Gynecology 191:1130–1132.
Walker, B. H. 1992. Biodiversity and Ecological Redundancy. Conservation Biology 6(1):18-23.































