
On a Manhattan street corner in April 1973, Motorola engineer Martin Cooper made history. Holding the world’s first cell phone—a 9 in. long, 2.5 lb brick of beige plastic—he strode out across Sixth Avenue and made the world’s first cell phone call.
Until that point, most cell phone research was focused on developing car phones, which were powered by heavy equipment that could be stored in the car’s trunk. But Cooper had other ideas. “People want to talk to other people—not a house, or an office, or a car. Given a choice, people will demand the freedom to communicate wherever they are, unfettered by the infamous copper wire,” he explained.16

The main challenge lay in developing a suitable energy source: To be truly portable, a mobile phone would need a small, light, and quickly rechargeable battery. The 1973 prototype cell phone contained a large and heavy battery that took 10 hours to charge and provided just 35 minutes of talk time. A breakthrough came in 1976, with the development of the lithium-ion battery, which boasted a high energy-to-weight ratio and could hold a charge for a long time. The lithium-ion battery helped bring down the weight of the first commercially available cell phone—the 1983 DynaTAC—to 1.75 lb. Since then, continued improvements to lithium-ion batteries have allowed cell phones—and a host of other modern devices such as tablets, laptops, and digital cameras—to become ever smaller, lighter, and more portable.

The size of mobile phones decreased drastically from the time that the first commercially available cell phones were released in the mid-1980s through the mid-2000s, owing in large part to improvements in battery energy density. Further improvements in batteries and other technological advances have led to the introduction of phones with greater capabilities (accompanied by growth in size to accommodate those capabilities). Shown are (from left to right): (1) Motorola DynaTEC (1983), (2) Nokia Mobira cityman 900 (1987), (3) Motorola MicroTAC (1994), (4) Motorola StarTAC (1996), (5) Nokia 7110 (1999), (6) Samsung SCH-A650 (2004), (7) Nokia E71 (2008), and (8) Samsung Galaxy S4 (2013). SOURCES: 1, 3, and 4, courtesy of Motorola; 2, 5, and 7, courtesy of Nokia; 6 and 8, courtesy of Samsung.
So, what makes a good battery and why is lithium the battery of choice for so many devices? Among the most important criteria used for evaluating the effectiveness of a battery are (1) its (weight-based) energy density (the amount of energy deliverable per unit volume or per unit weight), (2) its (weight-based) power density (the rate of energy deliverable per unit of time and weight), (3) safety, (4) costs, and (5) reliability. It turns out that lithium offers significant advantages judged by some of these criteria but drawbacks as judged by others. Because lithium, with a density of 0.53 g cm-3, is the lightest of metals—even lighter than water, which has a density of 1.00 g cm-3 at room temperature—it allows the building of batteries with a high energy density. Lithium is also among the most electropositive of metals, a measure of its ability to donate electrons and therefore form positive ions. Consequently, the change in the potential energy of lithium ions as they undergo chemical reactions at the anode and cathode is significant, translating into a higher voltage across the battery than in batteries that use other elements. However, a characteristic of lithium closely related to its electropositive nature (tendency to release an electron) is that it is highly reactive and flammable, which must be considered in the safe design of batteries that use lithium as a component.
The exploitation of lithium for use in batteries can be traced directly back to a materials discovery, the so-called “lithium intercalation compound,” which accommodates lithium atoms into its structure without tightly binding them so that they can be released during the charging process.17 This scientific discovery was followed by more discoveries, such as the high-cell-voltage cathode material LiCoO2, developed by John Goodenough,18 which is still one of the most common cathode materials in portable consumer electronics today.
How Do Batteries Work?
Batteries are made up of cells that contain two electrodes—a negatively charged anode and positively charged cathode—and an electrolyte that separates the electrodes and allows for the flow of charged atoms (ions) between them. Once the electrodes are connected to an external circuit, chemical reactions take place at the two electrodes, resulting in the release of electrons able to travel along the connected path and ions that travel through the electrolyte from one electrode to the other.
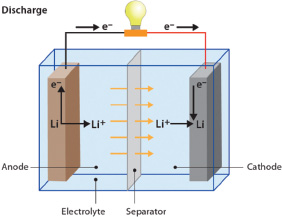
During charging, lithium is brought to the anode, where it is stored. When electricity is needed, lithium ions flow from the anode (negative electrode in which lithium is in a higher energy state) to the cathode (positive electrode, where lithium is in a lower energy state) just as water would flow from higher to lower ground. As a lithium ion leaves the anode, an electron is collected and then flows through the device (the light bulb, in this schematic).
In these compounds, lithium and other elements (such as cobalt and oxygen in LiCoO2) are combined in such a way that the atoms form a layered structure in which the lithium is loosely bound and therefore is easily available to participate in the chemical reactions and transportation that make the battery work. The architecture of the material is designed so that the material’s structure remains unaffected as the lithium atoms engage in the repetitive process of releasing electrons, traveling out of the material as ions and being replaced by lithium atoms from the electrolyte, allowing the process to be repeated many times.
Much of the materials research that has taken place over the last 35 years in refining the lithium battery (or trying to replace it with something even better) has focused on taking advantage of what lithium has to offer while satisfying the other needs for a good battery. Cobalt, for example, is a relatively expensive mineral and so materials researchers have worked to find alternatives that still meet standards of safety, reliability, and high energy densities.19
The payoff for significant improvements in energy and power densities extends well beyond personal electronic devices.
Other efforts have gone into developing alternative architectural structures that provide even greater energy densities or allow for cheaper synthesis and even replacing lithium itself with elements having similar chemical properties.20
The payoff for significant improvements in energy and power densities extends well beyond personal electronic devices. To take advantage of renewable solar or wind energy, we need to store electricity collected when the sun shines or the wind blows, for use when night falls or the wind calms. Battery-driven electric vehicles could replace foreign oil with domestic electricity and reduce our emission of greenhouse gases. Scientists are now working toward the next-generation batteries for these energy applications with up to five times better performance and costing one fifth the cost of the best batteries available today. Discovering novel materials is key to achieving this visionary goal.
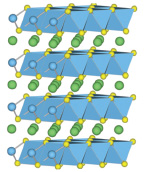
A schematic of the crystal structure of lithium cobalt oxide, LiCoO2, where single layers of lithium atoms (green balls) are loosely arrayed between cobalt oxide layers, made up of cobalt atoms (blue balls) and oxygen atoms (yellow balls). SOURCE: Courtesy of Anton Van der Ven, University of Michigan.

Renewable energy such as wind and solar requires storage of energy to be used when the sources are not available.




