Comparative Exposure Assessment
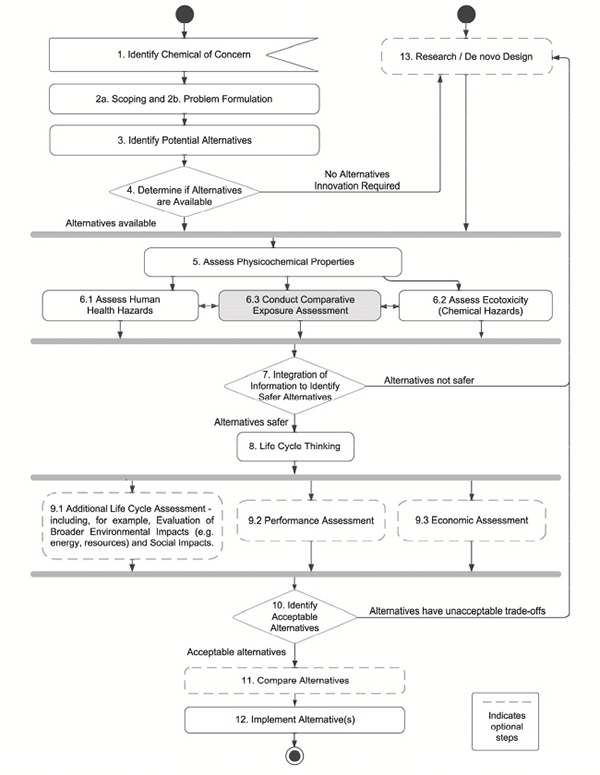
Exposure assessment is the process of considering and estimating the extent of exposure of human and ecological receptors.29Comparative exposure assessment plays an important role in the committee’s alternatives assessment process in understanding the overall safety of alternatives (Figure 6-1). The committee’s approach to exposure is to: a) consider the potential for reduced exposure due to inherent properties of the alternative chemicals; b) ensure that any substantive changes to the routes and any substantive increases in the levels of exposure are identified; and c) allow for consideration of the routes (dermal, oral, inhalation, etc.), patterns (acute, chronic), and levels of exposure (irrespective of any exposure controls) when integrating the evidence related to human and ecological toxicity among alternatives (Step 7 in Chapter 9).
In this chapter, the committee provides an overview of the approach to exposure assessment employed in other alternatives assessment frameworks. The committee then focuses on its framework and expands on the earlier discussion of exposure as it relates to scoping and bounding the assessment (Step 3; see Chapter 4). It is important to note that the consideration of exposure in the committee’s framework is not to demonstrate “safe” levels of exposure. Instead, it is comparative and is focused on the intrinsic potential for exposure without physical or administrative controls. In this way, the committee’s approach is different than most other approaches outlined in the frameworks reviewed.
The final section of the chapter goes through the sub-steps to be taken to complete Step 6.3, the comparative exposure assessment. Box 6-1 presents the elements of the committee’s suggested approach.
EXPOSURE ASSESSMENT IN EXISTING FRAMEWORKS
The committee considered the role of exposure assessment in existing frameworks. The role of exposure and how it is determined varies significantly from framework to framework, and depends on several factors. These include the objective or focus of the alternatives assessment, regulatory requirements, framework policies and procedures, and how the alternatives assessment results are used. Many of the existing frameworks only consider exposure to a cursory degree, such as considering intrinsic properties that influence persistence or bioaccumulation. This is often because exposures of alternatives are assumed to be the same, or “substantially equivalent” to each other and/or an original chemical of concern. This assumption allows the user of these frameworks to primarily focus on reducing toxicological indicators of hazard. Therefore, when an exposure assessment is included, it may be used in a secondary role, to confirm that the alternatives that appear acceptable or preferable to the chemical of concern from a toxicological perspective are not clearly worse from an exposure perspective. Some frameworks (e.g., TURI 2006a) include exposure potential (environmental, occupational, and public health) in their preliminary prioritization of chemical alternatives. Information such as the mobility of the chemical for a particular use, and potential for user exposure when the chemical is in a product, is used to determine the exposure potential of a chemical. In addition, TURI uses occupational exposure limits as measures of acute toxicity in comparative chemical hazard assessments.
In some frameworks (e.g., BizNGO (Rossi et al. 2012), an exposure assessment is performed only after alternatives are first identified based on hazard assessments. In this instance, the exposure assessment may be initiated based on the results of applying Life Cycle Thinking (see Chapter 10), meaning that potential impacts to human health or the environment across the life cycle of the alternative are considered. If the exposure assessment identifies concerns, then a partial or full risk assessment would be conducted, (depending on resources) to assess health effects. Similarly, a full or partial life cycle assessment (depending on resources and needs) would be conducted to assess remaining environmental impacts. It is unclear, however, how
_____________
29 Ecological receptors can include tissues, organisms, populations, communities, and ecosystems (EPA 2014d).
BOX 6-1
COMPARATIVE EXPOSURE ASSESSMENT AT A GLANCE (STEP 6.3)
- Determine if the alternatives would be expected to result in substantially equivalent exposures (Step 6.3). This can be accomplished by looking at outputs of simple exposure models (especially those considering estimates based on observed use patterns), comparing key physicochemical properties of alternatives (considered and compiled in Step 5), or, in some cases, applying knowledge about use scenarios and material properties.
- If alternatives are substantially equivalent in their expected exposure, then the assessment can be mainly hazard based (i.e., based on inherent hazard).
- If an alternative is deemed to have a substantially higher potential for exposure than the chemical of concern, then a more detailed exposure assessment may be appropriate. But a more detailed exposure assessment should only be performed if the toxicological and other advantages of the alternative are found, after analysis in later steps, to be attractive enough to warrant this additional effort.
- If the exposure potential of an alternative is preferable due to its inherent properties, this should be noted. It may add further weight to the choice of the alternative.
- To focus the consideration of alternatives on the inherent properties of substances, exposure estimates should be derived in the absence of assumptions about reliance on alternative-specific administrative, engineering, or personal protective equipment (PPE) controls.
For the required elements of Step 6.3, the exposure considerations are limited to the stage at which the chemical is used for human exposures and the use and disposal stages for ecological exposures. Broader upstream and downstream exposures that need to be considered may result from Life Cycle Thinking (Step 8) and life cycle analysis (Step 9.1).
these frameworks assess exposure and define and identify exposure concerns.
While exposure assessment is a module in the IC2 framework (IC2 2013), it is conducted after the hazard, performance evaluation, and cost and availability modules are completed. In the IC2 framework, chemical hazard reduction is viewed as a first step, and exposure is considered when examining potential trade-offs with identified alternatives.
Exposure also has a major role in the CA SPC (CA DTSC 2013b) and the REACH frameworks (ECHA 2011). For example, quantitative risk assessments30 and, as a consequence, exposure assessments, are required components of alternatives assessment in the second tier of the REACH framework (after the first tier of replacing high-concern chemicals with those of lower hazard).
As this discussion shows, there is considerable variation in the way exposure is considered in existing frameworks. This variability may be partly explained by the principles that frameworks have adopted to guide the consideration of exposure in an alternatives assessment. Many frameworks have a stated principle to prevent harm by focusing first on inherent toxicity rather than relying on downstream controls of exposure to mitigate the risk. This approach is consistent with the industrial hygiene hierarchy of controls, which prefers to completely prevent exposure from a hazardous chemical, rather than control exposure, because exposure controls can fail (Schulte et al. 2013). In addition to the principles, other factors may limit the user from performing an exposure assessment. For example, some users may not have enough detailed knowledge about how downstream product developers or end users use the chemical. The added cost, time, or expertise requirements needed to perform an exposure assessment may also be a consideration for some users.
Despite the trend of many existing frameworks to only minimally address exposure, this approach may not be appropriate in some cases. Chemical alternatives can have different chemical structures that influence their toxicity and exposure. The presence of different functional groups and physicochemical properties may increase (or decrease) the likelihood of chemical exposure to humans or ecological receptors, thus negating benefits derived from selecting a chemical alternative on the basis of relative hazard alone. In many cases, the greater the difference in the chemical structure, the more likely that the exposures will not be equivalent.
The committee also observed that in some frameworks, the role of exposure differed between human health and ecological assessments—a difference that appears difficult to justify. Within the ecological component of alternatives assessment, the
_____________
30Within risk assessment, exposure assessment serves the function of providing an estimate of dose that, when combined with dose-response assessment, converts the potential for harm into a probability of harm.
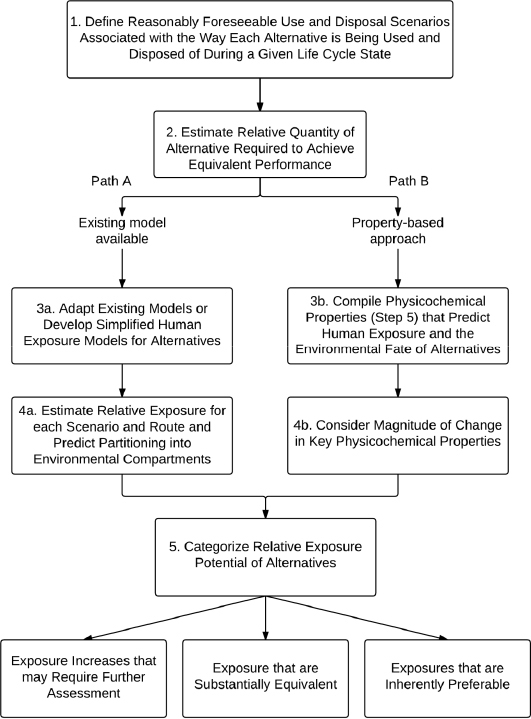
FIGURE 6-2 An approach to comparative exposure assessment within the committee’s chemical alternatives assessment framework. Exposure potential could be derived from either the outputs of simple exposure models or the comparison of key physicochemical properties to arrive at one of three determinations with respect to an alternative, as compared to a chemical of concern.
extent that potential exposure of ecological receptors (given degradation, bioaccumulation, persistence, and other processes related to a chemical’s fate in the environment) is considered stems from inherent properties of the chemicals, but consideration of the impact of these inherent exposure-related chemical properties on human health seems absent.
THE COMMITTEE’S APPROACH TO EXPOSURE ASSESSMENT
The role of exposure assessment needs to be carefully considered during an alternatives assessment. The committee notes that its role is very context-dependent, and may range from minimal to greater importance. In the following section, the committee describes its approach to exposure assessment in its framework, which is comparative and focused on the evaluation of intrinsic potential for exposure, in the absence of any physical or administrative controls.
To conserve assessment resources and still facilitate informed and efficient decision making, the committee’s framework describes a staged approach to the assessment of comparative exposure, with exposure being considered to different degrees at different points in the framework:
- In the problem formulation step (Step 2b, Chapter 4), the exposure pathways of the chemical of concern are considered early, during the problem formulation step, to focus the effort. Expected patterns (acute versus chronic) and routes (oral, dermal, inhalation) of exposure likely to be important were also identified during Step 2b, assuming there are intended and reasonably foreseeable exposure scenarios. Further, a qualitative consideration of a chemical’s use (in a formulated or dispersive product, locked in a polymer matrix, etc.) and physicochemical properties provides important information in Step 2b.
- A comparative exposure assessment (Step 6.3, described in this chapter) to estimate relative exposure differences between potential alternatives and the original chemical of concern. This step can be done in parallel with the assessment of toxicological evidence for both ecological receptors and human populations. At this stage, the only human exposures that should be considered are those that occur while using the chemical of concern.31 Ecological exposures to consider are those related to use and disposal, the areas of the most immediate interest. The procedure for completing the comparative exposure assessment is described in Sub-steps 1-7 of Step 6.3.
- Additional exposure assessment consideration, if concerns are identified when applying Life Cycle Thinking or examining the synthetic history of the alternatives in Steps 8 or 9.1.
- How to conduct a fuller, quantitative exposure assessment is explained in optional Sub-step 6.
Conduct Comparative Exposure Assessment (Step 6.3)
The committee’s framework includes specific exposure considerations alongside the hazard assessment. The consideration of exposure assessment concepts at this point in the framework aims to determine whether exposure to alternative chemical(s) might be decreased or increased compared to the original chemical of concern. The committee’s approach focuses on factors that are intrinsic either to the chemical alternatives or are inherent to the product into which the substance will be integrated. Therefore, extrinsic factors that may mitigate exposure (e.g., labeling, training, and a variety of engineering, administrative, or PPE controls) are not considered, which is consistent with the industrial hierarchy of controls (Schulte et al. 2013).
Figure 6-2 describes the committee’s approach to comparative exposure assessment. This approach allows for the use of either available exposure models or comparison of critical physicochemical properties. If these are not readily available, other information on use and chemical and material properties can be used as a way to estimate the relative exposure potential of alternatives. 32 Each of
_____________
31 Or its disposal if it is being disposed of as it is used.
32 It is important to note that there are often significant uncertainties in exposure estimates that can lead to an underestimate of potential exposures. This can happen because of assumptions about the behavior of certain chemicals (how they might partition), misunderstanding of use scenarios (unexpected uses), or how the chemical might “escape” a particular matrix like a polymer. This does not, however, minimize the importance of considering potential exposure, but rather points to the importance of a broad exploration of exposure potential, as well as ensuring stakeholder involvement and a
the numbers in the diagram refers to a sub-step of Step 6.3.
All alternative chemicals need to go through Sub-steps 1 and 2 of the comparative exposure assessment (Figure 6-2). If existing models are available, the assessment follows Path A, Sub-steps 3a and 4a. If no models are available, then the assessment follows Path B, Sub-steps 3b and 4b. Both paths converge at Sub-step 5, from which it is possible to arrive at one of three possible outcomes shown at the bottom of the diagram.
Comparative Exposure Assessment
Sub-step 1. Define reasonably foreseeable use and disposal scenarios associated with the way each alternative is being used and disposed of during a given life cycle stage: During this step, a set of reasonably foreseeable use scenarios, such as how the chemical is used in cleaning products, fuels, cosmetics, or personal care products, as well as corresponding routes of human exposure, are identified for each alternative. These can be derived from knowledge about functional use, or the behavior of a chemical during a particular activity, such as manufacturing, and the application of a chemical in a process or product (for example, is the chemical bound in matrix or dispersive in its application?), as well as physicochemical properties. Stakeholders can be helpful in identifying these exposures in that they can provide important input and data that the assessor may not have access to.
Sub-step 2. Estimate relative quantity of alternative required to achieve equivalent performance: Given that the alternatives may have very different properties, it is reasonable to assume that the mass of each alternative required to achieve the same performance per unit of product as the chemical of concern may be highly variable. It follows that the relative amount of exposure to both humans and ecological receptors from the alternatives may partly depend on the amount of the chemical required to achieve the functional requirements identified in Step 2 (Chapter 4). Therefore, in some cases, the relative quantity required would need to be considered in more detail, as described in Sub-steps 3 and 4. In other cases, however, completion of these first two steps may be sufficient to determine if an alternative presents substantially equivalent exposure.
Path A
Sub-step 3a. Adapt existing models or develop simplified exposure models for alternatives: For some exposure scenarios, the chemical of concern identified in Sub-step 1 might have an established exposure model describing the range of expected human exposures expected during its use. Accordingly, it may be possible to modify the existing models to compare the relative exposure expected from the alternative chemicals based on their physicochemical or other properties. If an established model for the chemical of concern is not immediately available, a simplified human exposure model may be developed using a variety of modeling approaches. For example, publicly available exposure models that address common exposure scenarios and the associated routes of concern (e.g., dermal exposures from chemicals in contact with skin, inhalation of chemicals in indoor air) may be used (Delmaar et al. 2005). A wide variety of exposure models that address common exposure scenarios and exposure routes (OECD, 2012a) are also available. Exposure models from publicly available exposure estimates for similar uses and chemicals may be an additional source. For example, manufacturers have developed and submitted models and estimates under REACH.
Exposure modeling tools are often deliberately structured in tiers of complexity (Tier 1 being the simplest, Tier 2 more complicated, and so on), to accommodate variety in the amount and types of information available to the user. The simplest tier can be applied to determine if substantially equivalent, substantially more, or substantially less exposure levels could be expected from different alternative chemicals. Even simpler qualitative assessments of exposure may suffice in some cases where models are not available.
Sub-step 4a. Estimate relative exposure for each scenario and route and predict partitioning into environmental compartments: Depending on the results from the simplified exposure models, the relative human exposure (taking into account the relative quantity of the substance required to achieve the required function) can be estimated. Table 6-1 shows an example of the use of a simplified exposure model to compare two alternative chemicals with respect to exposure. The level of detail in modeling and characterization of relative exposure can be limited to the extent needed to classify the exposure as either substantially more, less, or equivalent (i.e., it may be sufficient to say that exposure will be at least 10 times more, due to the relative quantities needed, without the need to
_____________
multidisciplinary approach to the exposure assessment process that enhances the information and input that goes into the assessment.
TABLE 6-1 Comparison of Exposure Potential Using Simple Exposure Models. The ConsExpo model (Delmaar et al. 2005) used to compare hypothetical fragrance data.
| Inputs: | Fragrance A | Fragrance B |
| Use frequency: | 365 days/yr | 365 days/yr |
| Product amount: | 1000 mg | 1000 mg |
| Weight fraction compound: | 0.0001 | 0.001 |
| Exposure duration: | 10 hr | 10 hr |
| Room volume: | 50 m3 | 50 m3 |
| Ventilation rate: | 0.5 rooms/hr | 0.5 rooms/hr |
| Inhalation rate: | 5 m3 | 5 m3 |
| Uptake fraction: | 100% | 100% |
| Body weight: | 60 kg | 60 kg |
| Outputs: | ||
| Acute Internal Dose: | 330 μg/kg | 33 μg/kg |
| Daily Chronic Dose: | 330 μg/kg | 33 μg/kg |
be more precise in the exact value of the relative exposure). The relative exposure assessment should also consider the potential for bioaccumulation or persistence of the chemical, as revealed by physicochemical properties. Steps 1-4, the comparison of potential exposure using simple exposure models, can be illustrated with an air freshener comparison. As shown on Table 6-1, Fragrance A is used in an air freshener. Fragrance B is less hazardous than Fragrance A, but more of Fragrance B is required to achieve the same effect. The ConsExpo model (Delmaar et al. 2005) was used to evaluate both exposures. Based on the ConsExpo model outputs (estimated acute internal dose and daily chronic dose), the assessor could determine whether the exposure potential of Fragrance B, because more must be used in the product, is substantially equivalent to Fragrance A, or whether the exposure differences need to be taken into account when considering hazard and other data.
Path B
Sub-step 3b. Compile physicochemical properties (see Step 5) that predict human exposure and the environmental fate of alternatives: For those exposure scenarios and routes for which there are no available models, the critical physicochemical properties can be considered to predict potential exposure. This sub-step relies on the information compiled during Step 5 in the committee’s framework (see Chapter 5 for more details). The exposure scenario and route of exposure will most often indicate which of these properties will be more or less relevant to evaluating whether an alternative is likely to lead to substantially more, substantially less, or substantially equivalent exposures by each route and scenario.
Sub-step 4b. Consider magnitude of change in key physicochemical properties: Comparing physicochemical properties that relate to ecological exposures should result in a qualitative indication of each chemical’s potential for partitioning to various media. The comparative ecological exposure assessment should begin by a direct comparison of those physicochemical parameters that are most likely to describe the persistence of a chemical in environmental media (e.g., Kow); partitioning of a chemical into the environmental media (water, soil, sediment, air); and potential for bioaccumulation of the chemical into biological tissue through direct contact with environmental media or through food chain exposures (see Chapters 5 and 7 for more detail).
While alternatives are not expected to be identical, they may be considered substantively equivalent, by virtue of having broadly similar patterns and numerical values for various key properties. What level of change in a key property indicates a chemical as “better,” “equivalent,” or “worse” with respect to exposure should be determined in advance and may be established through expert judgment.
Box 6-2 provides an example of how to use relationships related to physicochemical properties
BOX 6-2
COMPARISON OF EXPOSURE POTENTIAL USING PHYSICOCHEMICAL PROPERTIES FOR DERMAL EXPOSURE
Chemical A is an antimicrobial incorporated into metal-working fluid. Chemical B is less hazardous than Chemical A, but differs in its physicochemical properties such that the amount of dermal exposure can be expected to be higher than Chemical A.
For a surface area of 1000 cm2, an exposure time of 8 hours, and a body weight of 60 kg:
| Chemical A | Chemical B | |
| Physicochemical inputs: | ||
| Molecular weight: | 150 Da | 150 Da |
| Octanol-water partition coefficient: | 100.5 | 102.5 |
| Concentration: | 0.01 mg/mL | 0.01 mg/mL |
| Outputs: | ||
| Permeability coefficient (kp): | 0.0005 cm/hr | 0.0135 cm/hr |
| Predicted amount absorbed per kg bw: | 0.0007 cm/hr | 0.0180 cm/hr |
The permeability coefficient and amount absorbed is derived from models for dermal absorption available in the literature (Potts and Guy 1992; Cleek and Bunge 1993; McDougal and Boeniger 2002). Based on these calculations, an assessor may determine that dermal exposure potentials of Chemicals A and B are not substantially equivalent and that this difference should be considered during the Integration step (Step 7) and may require a more complete exposure assessment.
to compare human exposure potential among alternatives.
Categorizing Exposure
At this point in the process, regardless of whether the alternative chemical has been assessed along Path A or Path B, it should now be categorized, as explained under Sub-step 5.
Sub-step 5. Categorize relative exposure potential of alternatives: The inference of exposure potential could be derived from either the outputs of simple exposure models or the comparison of key physicochemical properties to arrive at one of three determinations. The determinations are comparisons of the alternative to the chemical of concern or other baseline as follows.
- Exposures that are substantially equivalent: An alternative may be considered substantially equivalent in that the differences in exposure are considered to be minor, perhaps in comparison to what may be significant differences in hazard. The notion of substantial equivalence is not strictly defined and is considered to be context-dependent. The primary purpose of this determination is to simplify the subsequent assessment of alternatives so that the determination of relative safety of alternatives can be limited to a discussion of their relative hazard.
- Exposures that are inherently preferable: A second possibility is that the alternative is actually preferable to the baseline chemical due to its inherent properties or the specific way it is being used in a product. Inherently preferable exposures are those that substantially reduce the potential for human or environmental exposure. Alternatives with inherently preferable exposure profiles might be considered safer, especially if there are uncertainties in hazard or exposure potential. If any alternatives are preferable because of their inherent properties, that should be noted for further consideration in Steps 7 and 10 of the overall framework (Chapter 9).
- Exposure increases that may require further assessment: This refers to an alternative determined to have potentially higher exposures than the baseline chemical. If, after further steps and analysis are completed, the alternative is found to be preferable for other reasons (e.g., reduced hazard in Steps 6.1 or 6.2 or additional considerations from Step 8), it may be worthwhile to conduct further exposure assessment efforts to arrive at a more quantitative estimate. This optional analysis is described as Sub-step 6. It is not intended to be a requirement of the alternatives assessment process. Furthermore, because additional effort is required, it is to
be expected to be conducted after further analysis justifies the additional work.
Sub-step 6. Quantitative comparative exposure assessment (optional): This chapter has focused on qualitative comparative exposure assessment, but for a number of reasons, a more quantitative or expansive exposure assessment may be required. The reasons this may be needed are: 1) toxicity is similar enough that exposure is a tiebreaker (as explained in Chapter 9); 2) the alternative is considered favorable for other reasons than exposure; or 3) the implications of Life Cycle Thinking or analysis (Steps 8 and 9.1) expand the number of chemicals or chemical use patterns that need to be evaluated. As a result, the alternative chemicals may have to undergo assessment of use patterns and exposure pathways to further examine how exposure might change.
Quantitative comparative exposure assessment is not considered a simple task. A useful reference for exposure assessment is the report, “Descriptions of Existing Models and Tools Used for Exposure Assessment, Results of OECD Survey” (OECD 2012a), which includes a table of available exposure models and tools with descriptions and links for each tool. Table 6-2 is an excerpt from this survey. It highlights models that are useful when considering human exposure. They may be suitable in the committee’s alternatives assessment process.
Another source of commonly used tools is the EPA’s “EXPOsure toolBOX” (EPA-Expo-Box), which was publicly released in 2013 (EPA 2014d). EXPOsure toolBOX is a compendium of exposure assessment tools and contains links to guidance documents, databases, models, reference materials, and other resources, including an “Exposure Factors” module designed to facilitate use of the 2011 Exposure Factors Handbook data (EPA 2014e).
Integration of Exposure Assessment into Subsequent Steps
The result of the required sub-steps of Step 6.3 is to identify and categorize the potential exposure for each alternative, in a relative sense, as being a) substantially equivalent, b) inherently preferable, or c) potentially worse (higher). In most cases, Step 6.3 can help identify differences in exposure that should be considered when integrating information in Steps 7 (Chapter 9) and 10 (Chapter 11) of the committee’s framework. If the extent, pattern, and degree of exposure are considered to be substantially equivalent between an alternative and the chemical of concern, then the determination of “safer” can be limited to the relative hazard of the chemicals. Where one or more of the exposure scenarios is inherently preferable due to intrinsic properties of the chemical or its integration into the actual product, then this can be noted as a further contribution to the relative safety of this alternative.
In the case where substantially increased human or ecological exposure is predicted for an alternative, then more detailed or rigorous exposure assessment may be called for. Rather than proceed immediately to a more complete exposure analysis, this assessment can be delayed until it has been determined that the alternatives in question have sufficient merit to justify the effort and the broader life cycle consequences have been explored in Step 8.
TABLE 6-2 Selected Human Health Comparative Exposure Assessment Tools
| Model name | Owner | Description |
| ART (Advanced Reach Tool) |
TNO (Netherlands Organisation for Applied Scientific Research) |
Advanced worker inhalation exposure assessment. |
| CALENDEX | Exponent | Estimates human exposure to chemical residues in foods and home-based chemical treatments, such as pest control and turf treatments (subscription required). |
| CALTOX | Lawrence Berkeley National Laboratory | A risk assessment model that calculates the distribution of a chemical in the environment and the risk of an adverse health effect due to a chemical. It also evaluates the distribution among different environmental compartments. |
| CARES (Cumulative and Aggregate Risk Evaluation System) |
US-EPA (U.S. Environmental Protection Agency) |
Databases to evaluate potential risk from dietary, drinking water, and residential sources and from oral, dermal, and inhalation routes of exposure. |
| ChemSTEER (Chemical Screening Tool For Exposures & Environmental Releases) |
US-EPA | Model for estimating (1) occupational inhalation and dermal exposures and (2) environmental releases to air, water, and land for chemicals during manufacturing, processing, and use. |
| CHESAR (Chemical Safety Assessment and Reporting) |
ECHA (European Chemicals Agency) |
REACH specific model to predict the concentration in environmental compartments, exposure of workers, and exposure of consumers via food and environment. Consumer exposure to be added soon. |
| ConsExpo | Exposure assessment of compounds in non-food consumer products. | |
| E-FAST (Exposure and Fate Assessment Screening Tool) |
US-EPA | Model for screening level estimates of chemical concentrations from releases to air, surface water, landfills, and from consumer products. Also estimates inhalation, dermal and ingestion potential dose rates and aquatic organism risks. |
| EMKG-EXPO-TOOL | BAUA (Federal Institute for Occupational Safety and Health) | Quantitative tier 1 assessment of occupational exposure (inhalation) to hazardous substances. |
| EUSES 2.1 (European Union System for the Evaluation of Substances) |
EC-JRC (European Commission Joint Research Center) |
A decision- support instrument to carry out assessments of the general risks of industrial chemicals and biocides posed by substances to people and the environment. |
| FHX (Farfield Human Exposure) |
Trent University | Holistic fate and exposure model for chemical exposure assessment of humans of different age-classes. |
| G-CIEMS (Grid-Catchment Integrated Environmental Modeling System) |
NIES (National Institute for Environmental Studies) |
Assessment of compounds in environmental and human exposure. |
| Generic Exposure Scenarios | CEFIC (European Chemical Industry Council) |
Tool (database) developed by sector associations to communicate generic exposure scenarios in that sector. |
| HERA (Human and Environmental Risk Assessment) |
AISE/CEFIC (International Association for Soaps, Detergents and Maintenance Products) |
Multiple human and environmental risk assessments on ingredients of household cleaning products according to HERA principles. |
| Model name | Owner | Description |
| IGEMS (Internet Geographical Exposure Modeling System) |
US-EPA | Includes models and data for ambient air, surface water, soil, and groundwater. |
| Industry Specific Generic Scenarios | US-EPA | Industry-specific methods and models for estimating occupational exposures and environmental releases for chemicals during industrial and commercial operations. |
| LIFELINE software suite | LifeLine Group Inc. | Addresses exposures that occur from the use of pesticides on agricultural crops and in residences, as well as pesticide residues that occur in water supplies (subscription required). |
| PROMISE (Probabilistic Methodology for Improving Solvent Exposure Assessment) |
American Chemistry Council | Designed to evaluate exposures and doses from single or multiple uses of products that contain volatile organics; not a population- based model. |
| RAIDAR (Risk Assessment, Identification and Ranking) |
Trent University | Holistic mass balance framework providing chemical exposure and risk assessments for humans and the environment. It is predominantly used as an evaluative model. |
| Risk Learning | AIST (Institute of Advanced Industrial Science and Technology) |
Estimating human health risks of a specific chemical substance in environmental media (air, water, soil, etc.) or contact media (food, etc.) using carcinogenic risk and hazard quotient as risk indices. |
| RiskCaT-LLE (Risk Calculation Took for the LLE-based Risk Estimation) |
AIST | Estimating human health risk as loss of life expectancy (LLE) from exposure to chemicals. |
| RiskOfDerm (Risk Assessment of Occupational Dermal Exposure to Chemicals) |
TNO | Worker potential dermal exposure assessment. |
| SDA (Soap and Detergent Association) |
Exponent | Exposure and risk screening methods for consumer product ingredients methodology for screening level exposure and risk assessments of chemicals used in consumer products, mainly laundry, cleaning, and personal care products. |
| SHEDS (Stochastic Human Exposure and Dose Simulation) |
US-EPA | A probabilistic human exposure model. There are currently three versions of SHEDS. SHEDS-Multimedia version 3 / 4 is a probabilistic aggregate residential exposure model. The other SHEDS models address exposures to particulate matter (SHEDS-PM), air toxics (SHEDS-ATOX), and wood (SHEDSWood). |
| Stoffenmanager | TNO | Control banding for worker dermal and inhalation exposure and quantitative exposure assessment for worker inhalation exposure. |
| USES 4.0 (Uniform System for the Evaluation of Substances) |
RIVM (National Institute of Public Health and the Environment) |
Quantitative assessment of the risks posed by new and existing chemical substances, as well as agricultural and non-agricultural pesticides to people and the environment. |
| SOURCE: OECD 2012a. | ||