7
A New Regulatory Framework for the Nation’s Investment in Academic Research
Having completed, in the preceding three chapters, its analyses of several policies and regulations, the committee offers its overarching findings, principles to guide the partnership, and recommends a new regulatory framework to govern the government-academic research partnership.
An immensely productive research enterprise emerged following World War II from the decision of the federal government to support basic research by flowing funds through academic research institutions, thereby creating what has often been termed the federal-academic research partnership. This partnership has been built on the belief that each of the partners would fulfill its roles and obligations with honesty, integrity, and credibility, and with the public good always in mind. The compact has produced a national research enterprise that engages in a constant process of discovery, creating new knowledge and advancing our understanding of human health and disease, our world, and our universe, while simultaneously training the next generation of researchers.
Research fuels the economy by generating new products, processes, and services; creating jobs; enabling new means of communication and commerce; and founding entirely new industries, such as biotechnology and information technology. Research contributes to national security through the development of weapons and defense systems and by strengthening the security of our national communication, transportation, financial, and public health and safety systems. Research has improved the quality of life and the overall health and well-being of the population. Research in the social and behavioral sciences has provided novel insights into human behaviors and into the social, political, and economic problems facing the nation. Scholarship in the humanities has enriched our understanding of our own culture and the cultures of others. Importantly, the remarkable growth and success of this enterprise has created a mutual interdependence between the federal government and the academic research community.
Despite the achievement of these extraordinary benefits, the partnership has come under stress from increasingly numerous and complex federal regulations and reporting requirements. While began as a means of exercising responsible oversight, regulations and reporting requirements have grown such that they now
unduly encumber and strain the very research enterprise they were intended to facilitate. The accumulation and complexity of regulations have required ever-greater commitments of time and resources from investigators. Indeed, they have generated a new category of university administrators: research compliance officers. Regulations, reporting requirements, and congressional mandates often overlap, resulting in duplication of effort, multiple reporting of the same information in different formats, and multiple submissions of information on different schedules. Conflicting guidance on compliance requirements has created uncertainty and confusion, often leading universities to implement overly prescriptive procedures in an effort to avoid penalties and thereby adding additional burden.1 The bottom line for the nation’s research enterprise is that we may be increasingly funding researchers to perform administrative tasks at the expense of research and teaching. It is time for a reaffirmation of the partnership and the development of a sensible regulatory framework adapted to the current needs of research enterprise.
OVERARCHING FINDINGS
As noted throughout this report, the research performed at research institutions by individual investigators and research teams, selected on the basis of scientific merit and capability, fuels economic growth, strengthens national security, enhances the overall health, education, and well-being of U.S. citizens, and often, of all humanity, and greatly contributes to U.S. leadership in science, technology, and social and behavioral sciences. Thus, federal investment in such research serves the interests of the nation. With the importance of this investment to the well-being of the nation as its backdrop, the committee noted nine overarching findings that characterize the current climate for federal support of research at academic research institutions:
- Effective regulation is essential to the overall health of the research enterprise, protecting both national investment and the various parties in the partnership (research participants, investigators, universities, and agencies).
- Continuing expansion of the federal regulatory system and its ever-growing requirements are diminishing the effectiveness of the nation’s research investment by directing investigators’ time away from research and training toward overlapping and incongruent administrative matters that do not take into consideration the environment under which research is conducted at academic institutions today. Our understanding of the cumulative effect of regulations is, however, constrained by a lack of empirical data.2
___________________
1Universities may also impose additional requirements in order to comply with state and local regulations or because of institutional approach.
2Particularly quantitative data.
- Most federal regulations, policies, and guidance, in and of themselves, are efforts to address important issues of accountability and performance associated with scientific integrity, the stewardship of federal funds, and the well-being of the people and animals involved in research. But these well-intended efforts often result in unintended consequences that needlessly encumber the nation’s investment in research.
- Many regulations fail to recognize the significant diversity of academic research institutions (e.g., in geographic location, public or private, size, legal structure, missions, financial and physical resources, and research capability). This diversity translates into widely varying capabilities to respond to increasing and overlapping research regulations.
- When regulations are inconsistent, duplicative, or unclear, universities may place additional requirements on research investigators, thereby diminishing the effectiveness of the national investment in research.
- Academic research institutions often receive research funding from multiple federal agencies, but approaches to similar shared goals and requirements (formats of grant proposals and biosketches, animal care, financial conflicts of interest, etc.) are not harmonized across these agencies. Consequently, investigators and administrative staff spend unnecessary time, energy, and resources complying with different sets of rules, regulations, and policies that address common core issues and concerns.
- Some academic research institutions have failed to respond appropriately to investigators’ transgressions or failed to use effectively the range of tools available to create an environment that strongly discourages, at both the institutional and individual level, behaviors in conflict with the standards and norms of the scientific community.
- Academic research institutions may be audited by any agency’s Inspector General office, many of which have very different approaches that in some cases are incongruent with stated policies of their agency.
- The relationship between federal research funding agencies and academic research institutions has for the past seven decades been considered a partnership. Yet, there exists no formal entity, mechanism, or process by which senior stakeholders from both partners, dedicated to fostering, sustaining, and strengthening our nation’s unique research partnership, can consider the effectiveness of existing research policies and review proposed new policies needed to sustain a maximally dynamic, efficient, and effective research enterprise. Further, no entity exists that can collect the data necessary to provide a true measure of the effectiveness and unintended consequence of existing research regulations.
As the committee learned, stresses in the federal-academic partnership diminish returns on the nation’s investment in academic research. The current structure of the regulatory regime needs to be recalibrated in order to best serve the nation’s interests.
A NEW REGULATORY FRAMEWORK FOR THE NATION’S INVESTMENT IN ACADEMIC RESEARCH
With these findings and in accord with its explicit charge, the committee sought to develop a new federal framework that, in conjunction with academic research institutions, allows for the conceptualization, development, harmonization, and reconsideration of research policy and regulation across federal agencies. The committee agrees on the importance of the following provision in the Statement of Task:
“Develop a framework and supporting principles for the Federal regulation of research universities in the 21st century, taking into account (a) the purposes, costs, benefits, and reporting requirements of regulation, (b) the processes used to promulgate regulations and reporting requirements, (c) the roles of Congress, Offices of Inspectors General and Federal agencies, including the Office of Science and Technology Policy and Office of Management and Budget, and (d) the missions of research universities.”
Throughout its study, the committee heard that the current volume of regulation steals from the nation’s investment in research and has become self-defeating. Inefficient and over-scaled regulation diverts, at the very least, researchers’ attention from research and must, as a consequence, reduce not just output, but also creativity and innovation. The effect of regulatory overburden is inevitably a less ambitious national research agenda.
Over the course of the committee’s deliberations, it became evident that to achieve a more efficient and effective research enterprise—one that maximizes the social benefits resulting from deployment of its intellectual capital and public and private investment of funds—it is essential to establish a much more focused, integrated, and forward-looking framework for managing the research partnership. The committee recognizes, as have others, “the importance of ensuring that policies have strong empirical foundations, both through careful analysis in advance and through retrospective review of what works and what does not.”3 In this report, the committee aims to articulate a framework that can meet the complexity and scale of 21st-century issues and that can adapt to the
___________________
3Cass Sunstein, Simpler: The Future of Government (New York, NY: Simon & Schuster, 2013), p. 41.
challenges that will arise inevitably from the results of research and from social change during decades ahead.
Background and Analysis
A distinguishing feature of the U.S. research enterprise is that a large, central part, including most fundamental and much applied research, operates as a partnership among federal agencies and academic research institutions and is built on the mutual investment of public and private funds. Historically, the largest share has derived from federal and state appropriations, although more recently, the second largest share of funds has come from the research universities themselves (see Figure 2-2). The public investment flows through mission-based research agencies that provide programmatic leadership and oversee processes to identify the very best research talent and meritorious ideas. Research institutions provide, in addition to intellectual capital, state-of-the-art facilities and infrastructure necessary for the safe and efficacious conduct of cutting-edge research performed by outstanding faculty, students, and trainees. Moreover, these institutions anchor local, regional, and national scientific and technological ecosystems that have profound and positive economic effects and that foster the development of hundreds of thousands, if not millions, of jobs. This research partnership, involving the mutual investment of talent and resources, has for the past seven decades produced the world’s most successful national research enterprise, an enterprise that has been and continues to be widely emulated around the world.
Historically, this system was based primarily on investigator-initiated project proposals, which, if deemed meritorious by anonymous peer review, were funded by grants to the successful investigators’ institutions to be used for research by the applicants. Under this system, the research institutions became legally responsible for overseeing the safe conduct of the research, as well as the legal and appropriate expenditure of the awarded funds. Initially, it was tacitly, if not explicitly, agreed that while expenditures of federal funds would comply with applicable regulations, the institutions would continue to be responsible for overseeing their faculty members’ conduct of research and training. During the “founding era” in the 1950s and early 1960s, an assurance system was implemented, by which institutional officials would certify for each research proposal that their institutions were in compliance with applicable federal regulations, largely related to expenditures of research funds, but also covering such topics as radiation and chemical safety. In the 1960s and 1970s, concerns for the rights and safety of human research participants and for the humane care of research animals led to regulations addressing these activities. During those years, each research funding agency promulgated its own individual requirements and formats regarding progress reports, financial reports, and invention reports. The basic system of research administration was project-centered and was reflected in a host of reports over the lifetime of each funding award.
The research partnership has grown immensely and is today far more complex. What began as a few hundred applications, awards, and reports, now numbers over 100,000 proposals annually with associated awards and reports. What was once an investment of millions of dollars in the 1950s now involves over $65 billion of public, private, and institutional resources. What could once be managed largely on a grant-by-grant basis through individualized, transaction-based applications and reports, can no longer be so managed. Although it was once sufficient for an institution to provide a few assurances regarding its conformity with applicable federal rules, now there is a need for a sophisticated infrastructure of compliance systems and safeguards to ensure the protection of human subjects, the humane care and use of animals, the appropriate use of taxpayer funds, the management of the potential for financial conflicts of interest, the safe storage and handling of potentially hazardous materials, and the appropriate recognition and management of biosafety and national security concerns. Today, government and academic research institutions expend substantial resources on the implementation of these requirements, on information systems for tracking transactions to effectively manage and report on these matters, and on training for faculty and staff to fulfill these requirements. All of this requires significant additional staff, as well as sophisticated facilities and information systems. Most research today must be conducted in institutional environments that have increasingly expensive and complicated physical facilities, as well as a complex infrastructure of procedural and physical safeguards.
The point here is that, for very good reasons, regulatory activities within the research partnership have grown dramatically in scale and sophistication. Activities that once required relatively minor costs and time commitments, and that could be managed fairly simply, now require large commitments of time, staff, and money; consequently, they entail large opportunity costs—so large that intelligent management of the cumulative regulatory load is important to the overall effectiveness of the nation’s investment in research.
An optimal regulatory framework must focus on the competence, efficiency, and harmonization of the entire system and the interdependence of the component parts, that is, agencies, institutions, faculty investigators, administrators, and electronic infrastructure. The entire system must work effectively to advance new knowledge and to move enabling ideas and innovations rapidly into practice.
Every transaction—proposal, progress report, financial transaction, and audit—must either contribute to positive outcomes and innovation or run the risk of detracting from and undermining the system. Friction and inefficiency in these transactions consumes time and funds that would otherwise be devoted to research, so that the entire system becomes less effective in producing outcomes and improving the well-being of the American public. Considered individually, many of these transactions are well intentioned and appear appropriate, but when considered holistically, they create unnecessary, conflicting, and duplicative efforts.
The maturation and continued success of the American research enterprise, founded on the basis of a federal-academic partnership, calls for a new framework, one that operates to ensure that the entire system, while ensuring integrity and safety, is focused on the identification of scientific talent and expertise, promising new ideas, innovative resources, and the optimal investment of public funds. The goal of the new framework should be the development of a holistic rather than piecemeal approach to the regulatory system so as to harmonize regulatory requirements across research funding agencies and to create a more effective and efficient partnership between agencies and research institutions. Another goal of the new framework is the routine exchange of information regarding safeguards, financial transactions, reports of inventions, and other matters in federal-wide standard systems such as e-commerce solutions to facilitate standard investigator- and project-specific exchanges of applications, biosketches, and progress reports.
A successful framework must ensure that investigators can conduct research in an environment that aims to ensure safety, efficiency, and integrity while facilitating scientific progress and the optimal use of researchers’ time. Each party in the enterprise must have a clear role in the effective operation of the system of requirements. Investigators should be provided with the administrative and project assistance to successfully navigate institutional and agency systems, thereby facilitating the appropriate use of their time on the conduct of research and training and the exchange of essential information.
The concept involves three parts:
- A forum – the proposed Research Policy Board (RPB).
- A responsible federal officer – the proposed White House Office of Science and Technology Policy (OSTP) Associate Director, Academic Research Enterprise.
- A set of underlying principles to guide the partnership.
Each of these components is described below.
Research Policy Board
The need for an analytical, anticipatory, and coordinating forum on regulatory matters seems clearly evident to the committee; however, its constitution, financing, and most effective connection to federal processes are far from obvious. The partnership involves quite diverse agencies and institutions and bridges the public-private boundary. As the committee contemplated organizational possibilities, it found useful analogues in what the federal government has already done in four different arenas.4 In each of those cases, tailored entities were cre-
___________________
4Established models for coordination of complex federal partnerships include (a) the Advisory Committee on Intergovernmental Relations, which, by congressional authoriza-
ated to facilitate partnerships involving multifaceted external institutional partners or constituencies.
The committee considered the National Science Board (NSB) as a home for the entity, but found that while the board “serves as advisors to both the President and Congress on policy matters related to science and engineering,” its responsibility to and alignment with the National Science Foundation limits its ability to provide the comprehensive approach to government-wide regulation that is needed to foster a sensible regulatory system. In addition, the NSB has other responsibilities and does not have the strong relationship to the Office of Information and Regulatory Affairs (OIRA) that the committee believes to be necessary.
The committee judges that the most relevant model for a research policy board is that used by the Securities and Exchange Commission (SEC) for the operation of the Financial Accounting Standards Board (FASB), which has functioned successfully for over four decades. FASB’s authority is derived entirely from the SEC. Membership is defined through formal processes approved and overseen by the SEC. However, FASB operates on private-sector funding raised by assessments on gross income of all public companies. It is a government-enabled, private-sector entity having a staff capable of coordinating the flow of business and supporting project teams assembled from time to time to address extant policy matters.
This model should be adapted to establish an RPB. The RPB can best function as a government-enabled, government-linked, private-sector entity, supported by assessments on academic research institutions to provide it the ability to support needed expert teams and future-oriented work. 5 The assessments should be mandatory and based on total volume of federally funded research. Given the scope and importance of the RPB’s mission, the institutions should perceive the assessment mechanism as a cost-effective, practical provision to optimize the efficient functioning of the research partnership under federal regulatory oversight. The RPB will provide research institutions a formal mechanism by which they can participate in the development of new regulations, the harmonization of existing regulations, review of the effectiveness and efficiency of the existing regulatory burden, and proposals for modification of existing regulations to minimize their
___________________
tion, addressed the interfaces and linkages among federal, state, and local government; (b) the Small Business Regulatory Enforcement Fairness Act (SBREFA), which calls for the creation, under law, of individual, issue-oriented, representative panels to assess the impact on small business of new regulatory proposals and requires agencies to address the concerns raised by these panel members; (c) the Base Realignment and Closure Commission (BRAC), charged to develop recommendations for packages of closings and realignments of military bases for action by Congress on an up-or-down basis, and (d) Financial Accounting Standards Board (FASB) and Public Company Accounting Oversight Board (PCAOB), established by the Securities and Exchange Commission for policy making and regulation relevant to public accounting and auditing in the United States.
5The committee recognizes that Federal Advisory Committee Act considerations will need to be resolved by Congress.
burden while maintaining and enhancing their effectiveness. Additionally, the RPB offers the opportunity to collect data and empirically test regulations.
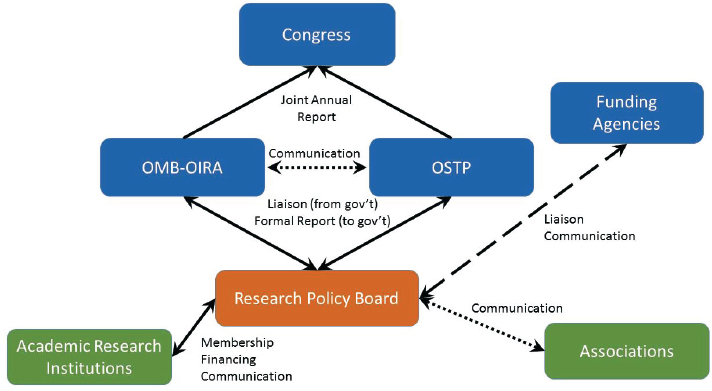
The RPB should connect formally to government through both the proposed Associate Director, Academic Research Enterprise, in OSTP and the Administrator of OIRA. (Relationships involving the RPB are laid out schematically in Figure 7-1.) These two officials should, in turn, have the obligation to report annually and jointly to Congress on regulatory issues affecting the research partnership and suggested steps to create a more effective regulatory environment.
The proposed new entity, the RPB, bridges the governmental organizations (shown in blue) and the private institutions and associations involved in the partnership (shown in green). Details of the bridging relationship are described in the report. The arrows show only those reporting and communication channels relevant to the operation of the RPB. There is no effort here to show operational channels within the government or the academic communities, which would not be altered in the proposed structure.
The RPB should manifest the following characteristics and roles:
- Its mission should be to improve and maintain a regulatory environment that is conducive to optimal performance of the research partnership by providing necessary data-driven information about regulatory benefits and burdens to the government, as gathered from the nation’s research institutions.
SOURCE: Courtesy of Larry R. Faulkner.
- It should be composed of 9 to 12 members from academic research institutions and 6 to 8 liaisons from federal agencies involved in the partnership, with members and liaisons being designated through formal processes of nomination and selection. Participants on both sides should be high-level leaders capable of addressing the broad range of policy issues relevant to the partnership.
- It should become the primary policy forum relating to the regulation of federal research programs in academic research institutions, with its members and liaisons serving as principal contacts for communication, both within their respective communities and across the boundary between the federal government and the research institutions.
- It should have general ability and responsibility to make recommendations concerning the conception, development, and harmonization of policies having similar purposes across research funding agencies. Toward fulfillment of this general purpose, the RPB should have these particular responsibilities:
- To provide thorough and informed analysis in the regulatory and policy-making processes.
- To identify negative or adverse consequences of existing policies and to make actionable recommendations regarding their possible improvement.
- To facilitate efforts within the government to coordinate research policy mechanisms, for example, via regulation, agency policy, agency application, and report formats, or audit standards and criteria.
- To create a forum for discussion of patterns in audit findings, compliance gaps, need for policy clarification or harmonization, and best practices.
- To conduct ongoing assessment and evaluation of regulatory burden, including development of metrics, periodic measurement, and identification of process improvements and policy changes to ensure that the administrative burden of research policy is minimized to the greatest extent possible consistent with maintaining responsible oversight of federally funded research.
- The RPB should be future oriented. It should be cognizant of trends affecting the overall regulatory load, and it should anticipate future regulatory challenges, especially those emerging from new science, such as synthetic biology, gene editing, and autonomous technology. It should organize expert project teams, as needed, to develop timely analysis on matters under consideration.
- The RPB should become a more systematic, integrated, and effective operational forum than any or all of the professional associations that have historically spoken for academic institutions on research-related matters. The committee recognizes and appreciates the excellent work often done by these organizations, but also believes that a more inte-
grated entity formally connected to the federal policy-making process is necessary to address the scale and complexity of current and future regulatory needs. Indeed, an effective RPB would become a means for leveraging continued work by these professional organizations.
Associate Director, Academic Research Enterprise, OSTP
While the RPB, as conceived, would fulfill the need for an active forum bridging the public-sector and private-sector partners, there remains a need for a federal officer with a focus on the healthy functioning of the government-academic research partnership.
The mission for the proposed OSTP Associate Director, Academic Research Enterprise, should be:
To coordinate the federal research policy and regulatory process and to routinely integrate and organize input in a broadly representative fashion among federal research agencies, the RPB, and other representatives of institutions of higher education and their representative associations.
This officer would routinely coordinate with senior agency staff including those in the Office of Management and Budget (OMB); research funding agencies; NSB, Chief Financial Officers Council; Council of Inspectors General on Integrity and Efficiency; President’s Council of Advisors on Science and Technology; National Science and Technology Council (NSTC), and other agencies as appropriate.
The Associate Director should address his or her mission through the following specific roles:
- To serve as the principal federal official responsible for coordination of federal agency policy and regulation relating to federally funded research in academic institutions, including policies in other areas, such as national security or immigration, that affect either academic research institutions or the conduct of research.
- To serve as an ex officio member of the NSTC and its primary committees, as appropriate.
- In partnership with OMB, to assist with coordination of the conception, development, and harmonization of regulations, policies, and proposal application formats having similar purposes across federal research agencies.
- To foster inclusion of representative input from the RPB and the university community on a routine basis in the regulatory and policy-making process, with the expectation that these comments will be given particular weight as agencies develop regulation and policy.
- To address with the RPB and the academic research community any unintended consequences of existing policy and to initiate appropriate corrective action.
- To ensure that input from the RPB and the research community is considered in the development of all policy mechanisms affecting research, for example, regulation, agency policy, agency application and report formats, and audit standards and their criteria.
- To serve as a coordinator among federal research agencies to discuss concerns identified by the RPB regarding audit findings, compliance gaps, or undue regulatory burdens. In addition, this individual will identify the need for policy clarification, harmonization, and clarification of audit standards, as appropriate.
- In partnership with the Administrator of OIRA, to report annually to Congress the results of ongoing assessment and evaluation of regulatory burden, including the development of metrics, periodic measurement, identification of process improvements, and policy changes that ensure that the administrative burden of research policy and regulation is minimized to the greatest extent possible while being mindful of the need to prevent fraud, waste, and abuse. Reports to Congress will include the results of such assessments from the RPB.
While, at times, the OSTP Associate Director for Science has taken on some of these responsibilities, it is often ad hoc and inconsistent, as the Associate Director for Science is required to address other pressing issues facing an administration. The position called for in this report would engender a consistent, long-term commitment to the overall health of the partnership. While OSTP often benefits from agency staff rotations, the position should be permanent. Federal research agencies could, however, provide the necessary funding for this OSTP Associate Director position.
Principles to Guide the Regulatory Framework
Finally, the committee offers the following principles to consistently guide the recalibration and future development of federal research regulations:
- Regulations should reflect the shared commitment of academic research institutions and federal agencies to the effective and efficient conduct of research and the maintenance of research integrity.
- Regulations should be harmonized across all federal research funding agencies. To the extent that agency-specific missions require agencies to depart from a uniform approach, agency-based deviations should be reviewed and approved by OIRA in consultation with the Associate Director, Academic Research Enterprise, OSTP.
- Regulations should be written with the input of the RPB.
- Regulations and their enforcement should take into account the risk of malfeasance and the overall cost of compliance. Before proposing any new regulation, an agency should determine whether the problem that the regulation is intended to address is systemic. Actions need to be targeted where transgressions occur. Minor issues should not become cause for disproportionate regulatory response. Egregious transgressions that are found to be isolated events should not trigger disproportionate responses.
- Regulations should be framed with the recognition that risk levels will never be reduced to zero.
- Regulations should be reviewed periodically to determine their effectiveness. If a regulation is deemed to be ineffective or excessively burdensome, it should be repealed or reformed.
- Wherever practical and appropriate, new regulations should be piloted at a small number of institutions to determine whether they efficiently accomplish the intent of regulation, and funds should be provided to pilot institutions for related personnel expenses.
- Academic research institutions must take timely and appropriate action against members of their communities who violate the values of trust and integrity to which community standards and federal funding of research, as well as academic responsibilities, require strict adherence.
RECOMMENDATIONS
The committee recommends the creation of a new mechanism, to include an active public-private forum and a designated official within government, to foster a more effective conception, development, and harmonization of research regulations of similar purposes across agencies.
7.1. Specifically, the committee recommends that Congress take the following actions:
- Establish a new entity, a Research Policy Board. The RPB would be a self-funded, government-linked entity serving as the primary policy forum for discussions relating to the regulation of federally funded research programs in academic research institutions.
- Establish a new Associate Director, Academic Research Enterprise, in the White House OSTP, having responsibilities to (a) serve as one of two principal federal contact points for the RPB; (b) oversee and facilitate the general health of the government-academic research partnership; (c) work in partnership with OMB-OIRA to manage the overall regulatory burden; and (d) jointly, with the Administrator of OIRA, issue an annual report to Congress on regulatory issues and actions affecting the research partnership.
7.2. Specifically, the committee recommends that participants in the government-academic research partnership adopt the above set of operational principles as a part of the new regulatory framework for federally funded academic research.
This page intentionally left blank.
















