4
Addressing Continuous Threats: HIV/AIDS, Tuberculosis, and Malaria
Although emerging and immediate infectious diseases often dominate media attention and captivate much of the dialogue around global health threats, the global community must not forget the continuing, persistent global health priorities the world has been addressing for several decades: human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS), tuberculosis (TB), and malaria. In 2002, AIDS was the leading cause of death worldwide among people ages 15–59, and more than 30 million people worldwide were infected (UNAIDS, 2016a). Although HIV/AIDS is no longer necessarily a death sentence, and people living with HIV/AIDS are able to treat it as a chronic disease, there is still no vaccine and no cure. With 2 million new infections, 1.1 million deaths, and 36.7 million people living with HIV in 2015, there is a clear need for heightened attention (UNAIDS, 2015d). TB, often an overlooked danger, saw 10.4 million new cases in 2015 and 1.4 million deaths1 worldwide (WHO, 2016c). In the United States alone, 9,557 cases of TB occurred in 2015. Although this was a 64.2 percent decrease from 1992, it was a 1.6 percent increase from 2014 (CDC, 2016a). Multiple strains of TB are resistant to one or more medications, with very few new drugs under development to address them. Finally, malaria continues to plague many countries, with 212 million new cases causing 429,000 deaths globally in 2015 (WHO, 2016d). Although
___________________
1 There were 1.8 million TB deaths worldwide in 2015, but 0.39 million of these occurred among people with an HIV infection. When an HIV-positive person dies from TB, the underlying cause is classified as HIV in the International Classification of Disease Systems (ICD-10th Revision) (WHO, 2016c).
malaria is rarely carried by mosquitoes in the United States, it is a constant threat to many U.S. travelers, international employees, and military members visiting and working in other countries. HIV/AIDS, TB, and malaria have a huge detrimental effect on the afflicted countries, making it difficult to foster growing economies and progressive societies.
This chapter begins by exploring the disease burden of HIV/AIDS and the progress made on reducing that burden in recent decades through bilateral and multilateral programs. It then provides future opportunities for programs and focus areas given the characteristics of the disease and patient populations now understood. Next, this chapter reviews the historic lack of funding toward tuberculosis and the danger it presents as many resistant strains continue to circulate. It also discusses the favorable reductions in malaria incidence and prevalence over the past 20 years, thanks to many dedicated programs. However, though much success has been achieved in all three disease areas, the chapter closes by cautioning against complacency—as all of these diseases are capable of resistance and resurgence.
HIV/AIDS
Beginning in the early 2000s, the HIV/AIDS pandemic and the global attention it received resulted in a great deal of funding and research toward finding drugs and other treatments and helping those with HIV improve their quality of life. Because of these concerted efforts by the global community, about 18.2 million people living with HIV now have access to antiretroviral therapy—a major feat, considering that just 16 years ago, fewer than 1 million people had access (HHS, 2016). This increased access is a testament to the power of collective action, as the global target of 15 million people receiving HIV treatment by 2015 was reached 9 months ahead of schedule (UNAIDS, 2015c). As a result of these efforts, new infections decreased globally by 35 percent between 2000 and 2015, and AIDS-related deaths decreased by 42 percent between 2005 and 2015 (UNAIDS, 2015a). Such an accomplishment is due in large part to the availability of sustainable funding, which is infrequently accessible for infectious disease efforts.
The story of the HIV/AIDS pandemic is still fraught with tragedy. Despite this success, people in sub-Saharan Africa continue to face the highest burden of disease worldwide, with 69.5 percent2 of people living with HIV and 65.2 percent3 of new infections occurring in that region in
___________________
2 In 2015, 36.7 million people were living with HIV globally, 25.5 million of whom were in sub-Saharan Africa. These figures were obtained by selecting “People living with HIV” as an indicator (UNAIDS, 2016a).
3 In 2015, 2.1 million new HIV infections occurred globally, 1.37 million of which were in sub-Saharan Africa. These figures were obtained by selecting “New HIV infections” as an indicator (UNAIDS, 2016a).
2015 (UNAIDS, 2016a). Additionally, progress within vulnerable populations has been slower in sub-Saharan Africa than other parts of the world, with adolescent girls and young women at disproportionate risk: AIDS is the leading cause of death among women of reproductive age (UNAIDS, 2015c). Global progress is also precariously at risk, as only 60 percent of people with HIV know their status, and the remaining 40 percent still need access to HIV testing services (HHS, 2016). That people under the age of 25 frequently do not know their HIV status and that there are now 200 million people between 15 and 24 years of age in sub-Saharan Africa creates the potential for the pandemic to surge out of control again (PEPFAR, 2016a).
The global community and U.S. government’s enthusiasm to end the AIDS pandemic has led to the establishment of lofty targets. Current global strategy is guided by the 90-90-90 agenda of the Joint United Nations Programme on HIV/AIDS, which, by 2020 aims to have 90 percent of people living with HIV knowing their status; 90 percent of people with diagnosed HIV infection receiving sustained antiretroviral treatment; and 90 percent of all people on antiretroviral treatment having viral suppression (UNAIDS, 2014a). To support these goals, funders have crafted their own strategies. For example, the Global Fund to Fight AIDS, Tuberculosis and Malaria Strategy 2017–2022 to End Epidemics aims to maximize the impact of its investments through finding differentiated approaches for diverse country contexts, increased alignment, and planning for sustainability of programs that tackle the disease (Global Fund, 2016).
The global monetary support for HIV/AIDS programs now totals about $19 billion annually, dwarfing funding provided for any other single disease. In 2015, $10.9 billion—57 percent of the total—came from domestic, in-country sources (AVERT, 2017; UNAIDS, 2016b). However, donor sources contribute in a variety of ways, especially for low-income countries that still heavily rely on external aid to finance their HIV response, with 44 countries relying on 75 percent or more of international assistance in 2014 (AVERT, 2017). Private funding accounted for $618 million in fiscal year (FY) 2014 (Kates et al., 2016). The U.S. President’s Emergency Plan for AIDS Relief (PEPFAR), which is responsible for the U.S. government’s response to global HIV, contributed $5.22 billion in FY2016 to bilateral HIV/AIDS programs,4 $1.35 billion to the Global Fund5 (PEPFAR, 2016c), and $117.9 million toward TB/HIV programs (PEPFAR, 2016b). This funding has remained stagnant for several years, forcing the PEPFAR program
___________________
4 Funding for bilateral HIV/AIDS programs covers funding for bilateral funding programs and regional programs in addition to contributions to the Joint United Nations Programme on HIV/AIDS, the International AIDS Vaccine Initiative, microbicides, and National Institutes of Health AIDS research (PEPFAR, 2016c).
5 This is the total U.S. contribution to the Global Fund.
to make difficult decisions in priorities as more patients are put on lifelong treatment regimens.
PEPFAR’s Progress and Potential
The PEPFAR program, began under the Bush administration in 2003, has seen tremendous success during its 13 years of existence (see Box 4-1), and has crossed through multiple phases of varying focus. Initially, it was a true emergency response at a time when entire generations in Africa were dying, leaving behind 14 million orphans and vulnerable children (PEPFAR, 2016a). One of PEPFAR’s strengths from its inception has been its use of senior-level leadership to monitor and coordinate its interagency efforts to drive change.
The program shifted to Phase II under the Obama administration once the crisis had abated; it maintained a focus on saving lives but placed greater emphasis on sustainability through partnership frameworks and mutual accountability. PEPFAR most recently shifted to Phase III, directing attention to what is likely to be the most challenging phase: getting the pandemic under sustainable control (PEPFAR, 2012a). As of September 30, 2015, PEPFAR has supported antiretroviral treatment for 9.5 million people and has reduced the number of new infections in its focus countries from 2.58 million in 2003 to 1.48 million in 2015 (PEPFAR, 2017a). With so many individuals dependent on the PEPFAR program for life-saving treatment and the dangerous potential for regression given the rapid youth population growth in many PEPFAR countries, there is an ethical imperative that PEPFAR be continued. Furthermore, more people are living with HIV/AIDS today than when the program started in 2003 (UNAIDS, 2016a), demonstrating the need for continued investment. The sustainability of an ongoing program like PEPFAR demands occasional realignment and shifting of priorities, as well as changes in the way it operates while maintaining its prior humanitarian commitments. Such changes should reflect the changes in the pandemic and various microepidemics (i.e., small-scale, community-level epidemics) as the disease continues to evolve.
PEPFAR has recently increased its use of data-driven programming. This change has allowed the program to report statistics on the HIV/AIDS burden in countries and on antiretroviral coverage and patients’ viral loads at the district level. The data-driven focus has improved the measurement of the number of patients on treatment. Increased data collection has been accompanied by a concomitant increase in understanding the socioeconomic influences of virus transmission, which should continue to be examined and analyzed to determine the best course of action. For example, although the incidence rates of HIV in most PEPFAR countries have declined, the rates have increased for populations most at risk, such as young women. As many as 380,000 adolescent girls and young women (ages 10–24) are infected each year globally, often due to gender-based violence (GBV) and exploitation by older men (UNAIDS, 2014b). In fact, women who experience GBV are three times more likely to contract HIV than women who do not (PEPFAR, 2015). Additionally, this group of women acquires HIV 5 to 7 years earlier than men (UNAIDS, 2014b) and is three times more likely to be HIV positive than men of the same age (PEPFAR, 2015).
One of the main future challenges for the PEPFAR program will be ascertaining the amount of money it can spend on direct treatment for patients versus the amount it can spend on prevention. When the PEPFAR program was created, the initial authorization from Congress called for 55 percent of funding to be spent on treatment. That requirement was carried over in the reauthorization legislation in 2008 but was relaxed slightly in
2013, when the PEPFAR Stewardship and Oversight Act of 20136 called for just half of bilateral funds to be directed toward treatment and care versus 55 percent (KFF, 2017c; PEPFAR, 2017a).
Next Generation of PEPFAR
The clear returns from PEPFAR investments show that its involvement contributes to better overall health outcomes in a country. Studies have indicated that PEPFAR focus countries have higher male employment rates than similar non-PEPFAR focus countries7 (Wagner et al., 2015) and have improved opinions of the United States (Daschle and Frist, 2015). Further enabling PEPFAR platforms to serve other country-specific health needs could increase this return on investment. Doing so is critical for driving future U.S. global health efforts and for establishing global partnerships to help build robust, broad, and efficient in-country health systems that ensure equitable access to the quality health care needed to help end the AIDS pandemic.
To look to the future in phasing down direct PEPFAR costs and incorporating health needs of countries, a priority for PEPFAR should be to continue supporting countries in their transition from bilateral aid to domestic financing by aiding in the mobilization of domestic resources for HIV/AIDS (and health more broadly). This shift would inevitably also allow more country ownership and decision making in their planning of national AIDS programs. This effort will require realistic country-specific assessments and strategies for building each national health system’s capacity to assume financial and operational responsibilities for service delivery. A caveat is that domestic resource mobilization may be a more realistically viable option for middle-income countries than for many low-income countries, which may have very little fiscal space in which to maneuver. In those cases, low-income countries will continue to rely on support from donors like the United States in order to keep HIV infection rates from surging out of control. A range of opinions exists about how to best support national governments in this transition, but many emphasize the need to incentivize and encourage country-driven funding by devising innovative, creative, and flexible financing strategies (Oomman et al., 2007; PEPFAR, 2009; Resch et al., 2015). Since meeting with other partners in 2015, PEPFAR and the U.S. Treasury are working with ministries of finance in recipient countries to create HIV expenditure committees and improve coordination
___________________
6 PEPFAR Stewardship and Oversight Act of 2013, Public Law 113-56.
7 PEPFAR focus countries include Botswana, Côte d’Ivoire, Ethiopia, Guyana, Haiti, Kenya, Mozambique, Namibia, Nigeria, Rwanda, South Africa, Tanzania, Uganda, Vietnam, and Zambia (KFF, 2017c).
of funding sources to increase efficiency and use domestic resources most effectively (PEPFAR, 2016a). Under these partnerships, the U.S. Department of the Treasury may provide technical assistance to finance ministries to strengthen public financial management of health resources. Another means for decreasing recipient countries’ reliance on PEPFAR for AIDS treatment is for the U.S. government to direct more of its AIDS budget to the Global Fund, which utilizes a unique e-marketplace8 to create a competitive market for specific health products and a Pooled Procurement Mechanism by which the Global Fund negotiates favorable procurement terms for health products (Global Fund, 2017). As a way to make current taxpayer funds go further, PEPFAR should also continue to employ key partnerships with the private sector focused on prevention, treatment, and care and support. In the case of supply chain management, for example, one partnership implemented new approaches to several steps in the supply chain, ensuring inventory was used before expiration and managing costs through pooling procurement (Sturchio and Cohen, 2012). Partially because of this infrastructure, PEPFAR was able to increase its proportion of generic drugs and contribute to reducing the annual cost of treatment per patient by nearly $700 (PEPFAR, 2012b).
The committee solicited information from the U.S. Agency for International Development (USAID) and the U.S. Centers for Disease Control and Prevention (CDC) field staff in multiple countries on PEPFAR’s successes and challenges.9 Overall, respondents reported that the PEPFAR program was generally well-received in countries and was significantly increasing the number of patients on treatment and decreasing HIV infection rates. Some respondents noted that country leaders understandably appreciated the technical and financial support provided by PEPFAR but wanted a greater degree of responsibility in decision making and prioritizing. In some cases, the priority of the U.S. program did not reflect the health priority of the host government. To alleviate this mismatch, some respondents recommended a shift in focus toward health systems strengthening or capacity building.
Broadening the Approach
There has been a growing understanding—both in the United States and internationally—of the need to broaden the spectrum of health care in a country by adopting a “development lens” that addresses the social and economic vulnerabilities of families and individuals multisectorally to achieve better outcomes in health, education, and livelihood (UNAIDS,
___________________
8 See more at http://www.wambo.org/home (accessed March 8, 2017).
9 See Chapter 1 for the full description of the information-gathering process.
2015c). Field staff respondents supported this approach by noting that although epidemic control is important, thousands of children and adults dying from preventable causes such as lack of access to care or safe water demonstrates the program’s need to broaden. Offering various types of interventions through the PEPFAR platform can ensure that patients are being viewed holistically while they are interacting with the health care system. For example, when HIV patients visit a clinic for treatment or testing, they could also be provided with education on nutrition or blood pressure screening and hypertension awareness, as some pilot projects are beginning to do through PEPFAR. Anecdotal results report that a patient with HIV who is on antiretroviral therapy can now receive care for hypertension or diabetes at the same clinic (Green, 2016). These program innovations are still somewhat limited in their design and depth, however, and funding is difficult. With all these considerations in mind, alternative methods of operation could be explored that can complement innovative financing strategies.
When considering new, broader approaches, there is a clear objective to couple PEPFAR’s services with those of other U.S. agencies and nongovernmental organizations (NGOs) working on nutrition efforts. People who take antiretroviral therapy often encounter food insecurity as a critical barrier to linkage to care, treatment adherence, retention in care, and viral load suppression. Malnourished people living with HIV are two to six times more likely to die within the first 6 months of treatment than similar patients who are not malnourished (UNAIDS, 2015b). Currently, nutrition assessment, counseling, and support are classified as “near core” interventions based on country context, but nutrition should be prioritized for all PEPFAR patients (PEPFAR, 2016a).10
Leveraging Partnerships
Although PEPFAR has had informal partnerships with host-country stakeholders (i.e., civil society), it has now begun to incorporate these relationships into all aspects of its programming in order to achieve sustainability and self-sufficiency. PEPFAR’s 2017 Annual Report to Congress states that now more than ever it encourages full participation of civil society to ensure they “have a voice at the decision-making table” (PEPFAR, 2017a). Partner governments and civil society organizations are now keenly in-
___________________
10 PEPFAR defines “core” interventions as activities central to HIV/AIDS that are critical to saving lives and preventing new infections and are grounded in science. “Near-core” interventions are defined as activities that directly support HIV/AIDS goals and cannot yet be done well by other partners or the host government, and “non-core” interventions are activities that do not directly affect HIV/AIDS goals and/or can be taken on by other partners or the host government (PEPFAR, 2016a).
volved in all aspects of the country operational plan process, which for the first time includes representatives from ministries of health and a large number of civil society organizations. This is a much different approach to country operational plan development than in previous years, and it has been extremely well-received by country stakeholders. The committee fully supports this new direction for PEPFAR and encourages more partnerships like the Determined, Resilient, Empowered, AIDS-free, Mentored, and Safe women (DREAMS) initiative (described in Box 4-2) and other engagements with community-level organizations to help drive sustainability, implementation, and effectiveness.
PEPFAR funding rules require that money is spent only on activities that focus on narrowly defined indicators related to scaling up treatment and decreasing HIV infection. Although the committee agrees these goals are important, addressing more general health goals through the same PEPFAR intervention would be easier if the indicators were defined a little more broadly. For example, “capacity building” could be added to the 50 percent on treatment and care allocation. Local workforce capacity is an ongoing challenge in many of these countries. The Medical Education Partnership Initiative (MEPI) and Nursing Education Partnership Initiative (NEPI) were created in response to the poor treatment outcomes resulting from the paucity of trained medical professionals in PEPFAR countries in 2010. These programs allowed PEPFAR to fund African medical institutions to increase the number of high-quality, in-country nurses and doctors (OARAC, 2015).
In their 5 years of partnerships, MEPI and NEPI have significantly improved capacity for education and research at in-country universities. For instance, at the University of Botswana, the MEPI program helped to establish and strengthen teaching sites, which led to the first-ever graduation of 80 locally trained doctors. The successes of the MEPI and NEPI partnerships mean the progress made possible by PEPFAR interventions will not be lost because there are too few medical professionals to treat patients. In FY2017, this partnership will be reinstated under a new name and, in learning from past MEPI and NEPI programs, will be more focused on medical and nursing education. Awardee institutions are encouraged to collaborate with the newly formed African Forum for Research and Education in Health, international partners that share the same goals, and successful MEPI and NEPI programs (NIH, 2016). This updated program will emphasize interdepartmental collaboration within awardee universities, as well as the development of partnerships with other universities and schools in their country to further expand the research workforce and resources. These goals advance the Fogarty International Center work by expanding research capacity among individuals, institutions, and larger networks and strengthening partnerships to advance global health research.
Understanding some of these broader determinants, PEPFAR’s DREAMS project began in 2014 as a $385 million partnership to reduce HIV infection in young women in 10 sub-Saharan African countries. Recognizing that HIV infection vulnerabilities in this population extend far beyond the health sector, DREAMS addresses the structural drivers of HIV infection, such as poverty, gender-based violence and inequality, and
education (see Box 4-2) (PEPFAR, 2017b). The program is still young and represents only 5 percent of the PEPFAR budget. The committee believes that more cross-sector and multidisciplinary programs like this should be explored not only to reduce HIV infection rates, but also to more holistically address social and development challenges and have a greater effect overall while leveraging the strengths of other stakeholders.
A Continued Focus on Data and Metrics
Despite the major progress made in addressing the HIV/AIDS pandemic, serious barriers prevent ending the pandemic. Most importantly, as noted previously, nearly half of all people living with HIV are unaware of their status, underscoring the urgency of closing the testing gap. Late diagnosis of HIV infection is the most substantial barrier to scaling up HIV treatment (UNAIDS, 2015c), but it also presents continuing challenges in keeping the epidemic under control if those who are infected are unaware. With the increase in data collection, PEPFAR has now been able to undertake public health impact assessments in 13 countries to measure progress toward epidemic control, with three completed so far (PEPFAR, 2017a). These public health impact assessments are comprehensive and measure prevalence, incidence, historic mortality, and service coverage down to the household level (PEPFAR, 2016a). Starting in FY2017, PEPFAR will also require implementing partners to provide yearly differentiated technical and direct support to sites and patients.
New infections still outpace the number of patients on treatment, so HIV prevention, and not just treatment, must be a focus of the next phase of the program. The pandemic cannot be solved through treatment alone, yet it will play a key role given the significant gains realized through using treatment as prevention. Recognizing this, PEPFAR increased funding for HIV prevention in young women in 2015 through the DREAMS initiative, expanded voluntary medical circumcision to prevent infections in young men, and in 2017 expanded Prevention for Treatment efforts to decrease transmission in young men (PEPFAR, 2017a). Additionally, with new guidelines from the World Health Organization (WHO) on antiretroviral therapy–based prevention, including pre-exposure prophylaxis (PrEP) (WHO, 2016a), PEPFAR is supporting a scale-up of PrEP in key populations, including adolescent girls and young women, which is the first time girls have had access to PrEP outside of a research setting. This scale-up is being supported by public–private partnerships such as DREAMS and through Gilead, which is financially supporting purchasing and operational expenses for PrEP for young women (PEPFAR, 2017a). However, there is still much to be done to prevent new infections. Prevention requires a comprehensive package that includes behavioral and structural interven-
tions and condom programming in addition to PrEP, HIV testing, voluntary male circumcision for HIV-negative men, and prevention of mother-to-child transmission, necessitating a varied approach to financing (UNAIDS, 2015c).
An estimated $30 billion will be needed to meet the ambitious 90-90-90 targets by 2020 (UNAIDS, 2014a, 2015c); currently, the world is short $19 billion. Moving forward, there is an opportunity to approach the next phase of PEPFAR in a multisectoral and multifaceted manner. Because results vary by country and interventions do not have the same effectiveness in all places, continued granular-level data collection and community-level understanding of community-level epidemics will be critical. Moreover, broad prevention efforts will need to be implemented at the community level and across sectors—with the support and involvement of country and community leaders—to truly reduce HIV infection rates.
SUMMARY AND RECOMMENDATION
PEPFAR is in great part responsible for the tremendous success achieved in slowing the HIV/AIDS pandemic globally since 2003. In addition to this progress on its primary goal, studies have shown that the countries in which PEPFAR is active had better opinions of the United States (Daschle and Frist, 2015) and also saw a 13 percent increase in employment rates among men compared to non-PEPFAR countries (Wagner et al., 2015). A truly bipartisan, collaborative program that has undergone transitions and shifts throughout the past 15 years, PEPFAR has shifted its focus from responding to an emergency to sustaining care while targeting at-risk populations, reflecting corresponding changes in the pandemic. However, the successes achieved should not be taken as a rationale for downscaling PEPFAR. The need for the program remains: Approximately 9.5 million people currently receive antiretroviral treatment through PEPFAR support, while 2.1 million new HIV infections still occur each year (PEPFAR, 2017a). PEPFAR’s next phase will continue to require cross-sector and data-driven efforts, as well as strong country partnerships, if the number of new HIV infections and AIDS-related deaths is to be dramatically reduced in as many countries as possible by 2030.
Conclusion: Now that the pandemic has evolved from requiring an emergency response to requiring a more sustained response, with patients needing chronic care, the PEPFAR platform needs to evolve accordingly. Given the substantial reduction in drug costs, the impact of treatment on prevention, and the ease of measurement of treatment outcomes, program ownership needs to shift to countries. At the same time, PEPFAR needs to leverage its existing
structures and platforms to address patient and population health issues more comprehensively, while continuing to fulfill the ethical imperative of providing life-saving treatment to those already covered by its programming.
Recommendation 4: Envision the Next Generation of PEPFAR
With its next reauthorization, Congress should fund The U.S. President’s Emergency Plan for AIDS Relief (PEPFAR) at current levels, and allow for more flexibility within the PEPFAR program by continuing to relax specific funding targets for all program areas. Continued accountability, efficiency, and measurement of results should be emphasized. In the future, moreover, PEPFAR should focus on the following key areas:
- Ensure that national governments assume greater ownership of national HIV/AIDS programs through joint planning and decision making, and that they increase domestic funding to help cover the costs of prevention and treatment.
- Adapt its delivery platform to become more of a cost-effective, chronic care system that is incorporated into each country’s health system and priorities.
- Continue to support the Global Fund to Fight AIDS, Tuberculosis and Malaria (Global Fund), and rely on it for specific functions where it has the comparative advantage. Such functions could include the Global Fund’s efficient procurement of products and multipartner efforts to encourage countries to transition to domestic sources of funding.
- Enhance emphasis on primary prevention through multisector efforts, including strong interventions against gender-based violence, given that many new HIV infections are occurring in adolescent girls.
TUBERCULOSIS
TB, an airborne disease, has been historically underprioritized. Although TB was placed on the international agenda after WHO declared it to be a global emergency in 1993—which only occurred after public health complacency in the 1970s and 1980s led to its resurgence (Ogden et al., 2003)—the world has struggled to reduce the global burden of TB, facing significant challenges in tackling the disease in the world’s poorest regions. Although the progress made in reducing TB incidence and mortality should be celebrated (including an approximate 1.4 to 2.3 percent reduction per
year between 2000 and 2015 [WHO, 2016c]), progress has nonetheless been slow. In fact, TB has now surpassed HIV/AIDS as the leading cause of death by an infectious disease worldwide. In 2015 there were 10.4 million new cases of TB and 1.4 million deaths attributed to TB (WHO, 2016c),11 as opposed to 1.1 million deaths attributed to HIV/AIDS (UNAIDS, 2016b).
TB is truly a global disease, with the highest absolute burden during 2015 being in Southeast Asia, Africa, and the Western Pacific12 (WHO, 2016c). The current global approach on TB is driven by WHO’s End TB Strategy, which provides goals for the Sustainable Development Goal era. These goals include achieving a 95 percent reduction in TB deaths and a 90 percent reduction in TB incidence by 2035. An additional goal is reducing the percentage of TB-affected households experiencing catastrophic medical expenditures to zero by 2030 (WHO, 2016c). Meeting these goals requires an acceleration of effort, costing an estimated $8.3 billion in global resources in 2016. Unfortunately, global financing for these goals was 30 percent short of what was required. As a testament to poor international assistance for TB, 84 percent of the $6.6 billion available in 2016 for TB care and prevention in low- and middle-income countries (LMICs) came from domestic resources. Weak international support particularly affected national TB programs in low-income countries, which required foreign assistance for about 90 percent of their program funding (WHO, 2016c).
Epidemiological Complexity of Tuberculosis
This lack of support for TB control is exacerbated by the complexities of TB infection and epidemiology, which become especially problematic in low-resource settings. First, TB is difficult to diagnose, and the resulting inadequate case finding is an obstacle to global TB control (WHO, 2016c). As a bacterial infection with an especially long growth cycle, TB cannot be easily detected with rapid diagnostic tests as is done for other diseases relevant to global health. The predominant method for diagnosing TB in LMICs is direct sputum smear microscopy,13 which is quick but limited in its ability to detect pediatric TB,14 drug-resistant TB, and TB/HIV
___________________
11 In 2015, an additional 0.39 millions deaths were caused by HIV/TB coinfection.
12 A reason Southeast Asia and Africa rank so high is that China, India, Indonesia, Nigeria, Pakistan, and South Africa accounted for 60 percent of new cases in 2015 (WHO, 2016c).
13 Direct sputum smear microscopy uses laboratory techniques to analyze a sample of sputum (a mixture of fluids coughed up from the respiratory tract) for the TB bacteria (Singhal and Myneedu, 2015). Serial samples may be required to confirm TB diagnoses, but many patients may fail to appear for a follow-up appointment, often due to an inability to afford repeat visits to a health facility (Harries et al., 1998; Kemp et al., 2007).
14 Because of limitations of direct sputum smear microscopy among children, pediatric TB is often called a “hidden epidemic,” an issue that is worsened by lack of treatment options.
coinfection (the latter two are discussed in later sections) (Desikan, 2013; Keshavjee and Farmer, 2012). Second, treatment length and complexity add to the diagnostic challenge. Due to the long growth cycle of TB, treatment can often take 6 to 9 months. Furthermore, treatment relies on combination chemotherapy, which uses multiple drugs (Laurenzi et al., 2007). Although TB drug regimens are highly effective when provided under strictly regulated clinical trial conditions (above 90 percent)15 (Laurenzi et al., 2007), adherence outside of these settings is lower due to physical barriers to access and lengthy treatment, which results in average global success rates of about 83 percent (Laurenzi et al., 2007; WHO, 2016c).
Drug-Resistant Tuberculosis
Poor adherence to TB treatment has resulted in a rapid increase of monoresistant TB, multidrug-resistant TB (MDR-TB), and even extensively drug-resistant TB (XDR-TB).16 First recognized in 1948, drug-resistant TB steadily grew but was more or less ignored (similar to nonresistant TB) until the early 1990s (Nachega and Chaisson, 2003). This lack of attention was partly driven by the emergence of combination chemotherapy in the mid-1900s, which led to the belief that TB would be conquered, and consequently resulted in the elimination of federal funding for TB control in the United States by 1972 (Cegielski, 2010). Similarly, funding for TB research dropped to just $514,000 in the United States in 1979 (Petrakos, 1993). Just as TB resurged during the 1970s and 1980s, so too did MDR-TB, with outbreaks in multiple U.S. states17 (Cegielski, 2010; Petrakos, 1993) and epidemics reported across the globe (Cegielski, 2010). In 2015 alone there were approximately 480,000 new cases of MDR-TB, 9.5 percent of which were XDR-TB cases. Furthermore, 3.9 percent of all new TB cases in 2015 were MDR-TB cases (WHO, 2016c). While these numbers seem small, they may underestimate the true burden of drug-resistant bacteria as the method for determining resistance (sputum culture) can take up to 6 weeks (CDC, 2013) and requires sophisticated laboratory capacity that is lacking in many LMICs.
The rise of drug resistance is a blow to global TB control efforts, as monoresistant TB and MDR-TB also can be spread from person to person (CDC, 2016b). It was previously thought that XDR-TB only was acquired
___________________
15 The studies referenced are clinical trials conducted in the United States and the United Kingdom, settings that do not apply to everyday circumstances in LMICs.
16 Monoresistant TB is TB that is resistant to one TB drug. MDR-TB is TB that is resistant to at least two commonly used TB drugs. XDR-TB is TB that is resistant to two or more first-line drugs in addition to at least two second-line drugs used to treat MDR-TB (WHO, 2012a).
17 New York City alone spent more than $1 billion to curb a TB epidemic in the early 1990s (Frieden et al., 1995).
due to repeated treatment failure. However, recent findings indicate that even XDR-TB can be spread from person to person (Shah et al., 2017). To make matters worse, length of treatment for these drug-resistant strains is even longer—up to 20 months18—and their success rates even lower: 52 percent for MDR-TB and 28 percent for XDR-TB (WHO, 2016c). Low treatment success rates make XDR-TB especially lethal, with the long-term chances of survival being only 15 percent (Pietersen et al., 2014). Although preventing these resistant strains is vital from a security standpoint, it is also very cost-effective. The cost to treat TB ranges from $100 to $500 per person in LMICs, yet the costs for treating MDR-TB and XDR-TB can reach up to $10,000 and $26,000, respectively (Laurence et al., 2015; Nieburg et al., 2015; Pooran et al., 2013).
TB/HIV Coinfection
The frequent coinfection of TB with HIV poses another epidemiological challenge. In fact, the risk of developing TB is 26 to 31 times greater for those who have HIV/AIDS than those who do not (WHO, 2017b). In 2015, one-third of those living with HIV were infected with TB, and of the 10.4 million new cases of TB, 1.2 million occurred in people living with HIV (WHO, 2016c). Furthermore, for those living with HIV in resource-poor settings, TB is the largest single cause of death (UNAIDS, 2016b), accounting for 34.45 percent of HIV deaths.19 Coinfection has been found to result in a dangerous interaction in the body, with HIV being linked to the progression of a TB infection and TB being reported to worsen HIV infection (Getahun et al., 2010; Whalen et al., 1995). Further exacerbating this issue is that HIV coinfection is associated with the malabsorption of anti-TB drugs (Patel et al., 1995) and has been found to be a risk factor for acquiring MDR-TB (Suchindran et al., 2009). The extensive treatment required to care for co-infected patients exerts enormous pressure on health systems found in LMICs, where 99 percent of TB-related HIV deaths occur (Pawlowski et al., 2012).
Paradigm Shift for Tuberculosis
The complexity of TB, in terms of its epidemiology, rise in drug-resistant strains, and its interaction with HIV, poses a major problem for global efforts directed toward both TB and HIV. That these diseases frequently occur in the same country (see Figure 4-1) sheds light on the challenges that country governments, multilateral organizations, and U.S.-led
___________________
18 New WHO recommendations have shortened regimens for MDR-TB to 9–12 months.
19 This percentage is derived from dividing the number of HIV deaths due to TB (0.39 million) by the total number of HIV-related deaths (1.1 million) in 2015 (UNAIDS, 2016b).
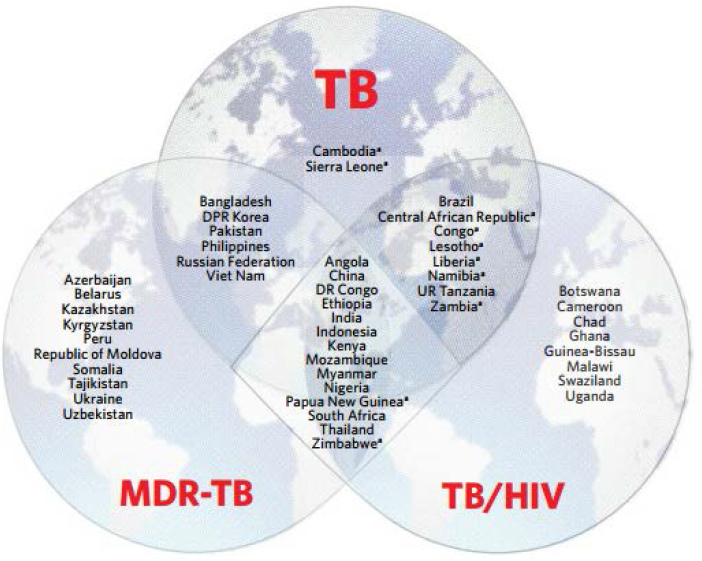
NOTES: Includes top 20 by absolute number of cases and an additional 10 countries that have the highest incidence rates per capita. HIV = human immunodeficiency virus; MDR-TB = multidrug-resistant tuberculosis; TB = tuberculosis; TB/HIV = tuberculosis/HIV coinfection.
a Indicates countries that are included in the list of 30 high-burden countries for TB on the basis of the severity of their TB burden (i.e., TB incidence per 100,000 population), as opposed to the top 20, which are included on the basis of their absolute number of incident cases per year.
SOURCE: Global Tuberculosis Report 2016 http://apps.who.int/iris/bitstream/10665/250441/1/9789241565394-eng.pdf?ua=1
ISBN 978 92 4 156539 4
Figure 2.2: Countries in the three TB high-burden country lists that will be used by WHO during the period 2016–2020, and their areas of overlap. Page 12.
bilateral programs face. The center diamond in Figure 4-1 highlights where TB, MDR-TB, and TB/HIV coinfection occur at high rates.
Clearly, the current strategies used to treat and combat TB have not kept pace with the burden it poses. The mainstay of TB treatment is the directly observed treatment, short-course (DOTS) strategy,20 which
___________________
20 Though DOTS contains five elements (sustained political and financial commitment, diagnosis by quality-ensured sputum smear microscopy, standardized short-course anti-TB
was adopted by WHO in the mid-1990s largely as a branding strategy for global TB control. The evidence for the effectiveness of short-course therapy, which served as the foundation of DOTS, was limited to Malawi, Mozambique, and Tanzania in a 1991 study (Ogden et al., 2003). By 2001, 127 countries had adopted the DOTS policy (WHO, 2001). The logic behind DOTS is that through direct observation treatment failure would occur less frequently. However, meta-analyses of the literature have shown that DOTS (the directly observed component) is no more effective than self-administration of treatment (Karumbi and Garner, 2015; Pasipanodya and Gumbo, 2013).
Given the resource intensiveness of directly observed treatment and its relative ineffectiveness compared to self-administration, policy makers need to reconsider TB control strategies and a redirection of resources toward other causes of poor treatment outcomes. As TB is a disease of poverty, one option could be targeting the social determinants of TB, which include social circumstances and material resources, misunderstandings of the etiology of TB, stigma, fear of punitive sanctions due to a positive TB diagnosis, and a lack of social support during treatment (Noyes and Popay, 2007). However, this option would not target the fundamental problems that LMICs face in diagnosing TB and its drug-resistant strains and managing the complexities of treatment. Addressing these issues would require a substantial investment in TB diagnostics and drugs.
Research and Development for Tuberculosis Diagnostics and Drugs
Development of new diagnostic tools for TB has seen some progress. The most notable example is the GeneXpert molecular test, which can detect monoresistant, specifically Rifampicin-resistant, TB strains within 2 hours (Lawn et al., 2013; Sharma et al., 2015). Although this tool is highly accurate and has the potential to improve the accuracy and speed of TB diagnosis, it is also costly, which poses a hurdle for its implementation. WHO endorsement of the GeneXpert tool has resulted in donor support, but sustaining its use will fall to national governments. Regardless, rapid diagnostic tests are urgently needed to combat TB (Lawn et al., 2013). Similar to diagnostics, the development of new TB drugs also is needed to improve the outcomes of TB therapy in terms of shortening the length and improving therapy for MDR-TB and XDR-TB. Unfortunately, the new TB drug pipeline is almost empty. Only two new TB drugs (bedaquiline and delamanid) have been approved to treat MDR-TB, which marks the first
___________________
treatment given under direct and supportive observation, a regular supply of high-quality anti-TB drugs, and standardized recording and reporting), it is mostly known for its direct observation of treatment.
time in more than 50 years that a new drug has been approved for this use (Zumla et al., 2015). Though 16 additional drugs are in preclinical and clinical development (Zumla et al., 2015), research and development for TB drugs is particularly complex due to the need to establish efficacy of a new agent in the context of combination therapy, which can make the clinical phase of development last up to two decades (Ginsberg and Spigelman, 2007). Streamlining the development of TB drugs will require novel research strategies, possibly including the use of biomarkers (discussed in Chapter 7).
U.S. Strategy for Tuberculosis
The U.S. government, through USAID, started its TB program in 1998. Global TB funding has increased since then, and the United States is now the largest international funder for TB, with its bilateral programs reaching more than 50 countries (KFF, 2017b). Since the initiation of its TB program, the U.S. government has contributed to the gains made against TB, saving 37 million lives between 2000 and 2013 (USAID, 2015). The current U.S. policy for global TB is guided by the Global Tuberculosis Strategy: 2015–2019, which assigns clear roles to U.S. government agencies for TB activities. USAID is designated as the lead agency in international TB control, including MDR-TB21 and XDR-TB; PEPFAR is established as the lead actor for U.S. government response to TB/HIV coinfection; CDC is responsible for providing technical support (surveillance and laboratory networks) to ministries of health and U.S. agencies, in addition to conducting operational research; and the National Institute of Allergy and Infectious Diseases is responsible for U.S. international TB-related research (USAID, 2015). However, the current strategy failed to designate a single entity to direct and coordinate resources. This U.S. TB strategy mirrors WHO’s End TB Strategy and has ambitious goals of achieving a 95 percent reduction in TB deaths and a 90 percent reduction in TB incidence by 2035 (USAID, 2015). The U.S. government has also created an MDR-TB–specific strategy, the National Action Plan for Combating Multidrug-Resistant Tuberculosis,22 which emphasizes improving access to diagnostics and treatment internationally and accelerating research and development for MDR-TB (White House, 2015).
Unfortunately, U.S. funding for global TB has not been commensurate
___________________
21 USAID has recently entered into a partnership with Janssen, the company that developed bedaquiline, to guarantee that the company’s donations of the MDR-TB drug are implemented effectively into treatment programs in its partner countries (USAID, 2016).
22 The National Action Plan was developed in response to President Obama’s Executive Order 13676 and creates an interagency collaboration between USAID, PEPFAR, CDC, the National Institutes of Health, and the U.S. Department of Defense. It also includes specific goals to curb the burden of MDR-TB in the United States.
with its ambitious goals. Despite $4 billion being authorized under the 2008 Lantos-Hyde Act for FY2009–FY2013 for global TB programs, only 40 percent ever became appropriated (Nieburg and Jackson, 2016) in recent years, and the TB budget has hovered around $230–$240 million annually (KFF, 2017b). Furthermore, funding was contracted in the FY2017 budget request for TB, which was only $195 million, a 19 percent decrease from FY2016 (KFF, 2017a). Although this decrease in funding could be particularly detrimental for low-income countries that rely on international donors for 90 percent of their national TB program financing (WHO, 2016c), it is symptomatic of a broader issue: an underprioritization of TB. Underprioritization is not a sustainable course of action, as it will threaten gains made in key U.S. programs, particularly PEPFAR. Figure 4-1 displays those countries that have a high burden for TB, MDR-TB, and TB/HIV in the center diamond. Of the 14 countries listed, 11 are PEPFAR-supported countries. PEPFAR has allocated an average of $138 million toward integrated TB/HIV treatment programs, but this amounts to only 3 percent of its expenditures (Morrison and Neiburg, 2014). If additional resources and operational changes are not directed toward this need, PEPFAR could lose hard-won gains in the clinical management of HIV (IOM, 2013). To meet the goals set in the current U.S. strategy for TB, a fundamental rethinking is needed regarding the treatment programs deployed, the social interventions used, and the investment in research and development for diagnostics and drugs.
SUMMARY AND RECOMMENDATION
TB has historically been an underprioritized disease, and accordingly has not shown the same progress as HIV/AIDS. In fact, between 2000 and 2015, there was only a 1.4 percent reduction per year in the global incidence of TB and a 2.3 percent reduction per year in global mortality from the disease (WHO, 2016c). TB caused 1.4 million deaths in 2015, thus surpassing HIV/AIDS as the leading cause of death due to an infectious disease. Further complicating global efforts is a rise in drug resistance, with 3.9 percent of all new TB cases being drug-resistant. Treatment for TB, MDR-TB, and XDR-TB is lengthy and complex, and frequently fails. Furthermore, treatment for MDR-TB and XDR-TB can cost up to 100 times as much as treatment for nonresistant TB (Laurence et al., 2015; Nieburg et al., 2015; Pooran et al., 2013). With few drugs available to treat these resistant strains and little research and development on new treatment options, TB and its drug-resistant strains pose a growing threat to the health and health security of all countries, including the United States. Yet while TB has been a priority for the United States and was a focal point in a 2015 National Action Plan for Combating MDR-TB, it has been underprioritized financially.
Of the $4 billion authorized over 5 years under the 2008 Lantos-Hyde Act to implement the strategy, only 40 percent was ever appropriated, with the most recent budget request being reduced even further than previous years to just $195 million (KFF, 2017a; Nieburg and Jackson, 2016).
Conclusion: The threat of TB has been rising in recent decades, and levels of funding have been far short of the amounts needed to address this threat adequately. There are very few drugs or vaccines for TB in the development pipeline, and the growing number of resistant strains will make this threat even more alarming and complex to address if sufficient action is not taken. The U.S. government’s underprioritization of TB undercuts its capability to reduce the burden of TB. Given that current strategies for combating the disease are not keeping pace with its burden and complexity, the U.S. government needs to significantly reevaluate its investment in and strategies for dealing with TB.
Recommendation 5: Confront the Threat of Tuberculosis
The U.S. Centers for Disease Control and Prevention, the National Institute of Allergy and Infectious Diseases, and the U.S. Agency for International Development should conduct a thorough global threat assessment of rising tuberculosis (TB) levels, including multidrug-resistant TB and extensively drug-resistant TB. They should then execute a plan of action, including governance structure and priority activities, for developing and investing in new diagnostics, drugs, vaccines, and delivery systems.
MALARIA
Malaria, one of the world’s most widespread parasitic diseases, places approximately 3.2 billion people at risk of infection at all times (CDC, 2016c) and resulted in 212 million new cases and 429,000 deaths in 2015 (WHO, 2016d). The WHO African region faces the highest global burden, with 90 percent of cases and 92 percent of deaths. Furthermore, malaria disproportionately affects the poorest and most vulnerable populations (WHO, 2014), which includes pregnant women, infants, and children under 5 years old (WHO, 2017a). In fact, nearly 70 percent of malaria deaths worldwide in 2015 were children under the age of 5 (WHO, 2017a), with a child dying every 30 seconds. Malaria also poses an economic burden both to families and endemic countries. Families can lose more than one quarter of their income due to treatment costs and lost days of work, and malaria costs the African region upward of $12 billion each year in lost productivity (European Alliance against Malaria, 2007; UNICEF, 2004).
Global Commitment to Malaria
Although efforts to control malaria date back to the 19th century, true global commitment began in 1955 when WHO formed the Global Malaria Eradication Program (GMEP), which had ambitious goals to interrupt malaria transmission in all endemic areas, with the exception of sub-Saharan Africa. GMEP’s expansive efforts resulted in 143 malaria-endemic countries becoming classified as malaria free in 1978 (Nájera, 2001; RBM, 2011). However, public support waned throughout the 1960s, which resulted in tightening of financial resources for global malaria control. Global support for GMEP became officially withdrawn at the 22nd World Health Assembly in 1969 (Nájera et al., 2011). The lack of public support and financial resources resulted in worldwide resurgence of malaria during the 1980s (RBM, 2011). The resurgence contributed to malaria climbing back up the global agenda, formalized with the launch of the Roll Back Malaria (RBM) partnership in 1998 and the adoption of the Millennium Development Goals (MDGs) in 2000. The political commitment and subsequent influx of funding have contributed to remarkable gains in malaria control. Between 2000 and 2015, 57 countries achieved the MDG of reducing the number of new malaria cases by at least 75 percent (WHO, 2015a). At the same time, the global incidence of malaria declined by 41 percent and global malaria mortality rates declined by 62 percent (WHO, 2016d).
Global Financing of Malaria Control and Elimination
The progress made toward malaria control and elimination since the turn of the century was possible in large part due to increases in financial support, which grew from under $100 million in 2000 (WHO, 2013) to $2.9 billion in 2015 (WHO, 2016d). Despite these increases, global funding for malaria still falls 46 percent short of the $6.4 billion needed to achieve the 2020 goals established by WHO’s Global Technical Strategy. In 2015 the majority (68 percent) of funding for global malaria was provided by multilateral and bilateral programs, with the Global Fund accounting for nearly 45 percent of all international assistance. The United States continues to be the single largest international funder for malaria, accounting for approximately 35 percent of total malaria funding in 2015, including both the President’s Malaria Initiative (PMI) and contributions to the Global Fund (WHO, 2016d). The remaining 32 percent of funding was provided by governments of endemic countries. Although investing in malaria may seem costly, the cost–benefit analysis modeling the effect of global malaria reduction and elimination found that the net gains in economic output would be worth $208.6 billion (Purdy et al., 2013). These gains would positively affect international travelers and endemic populations alike.
Malaria Elimination and Shrinking the Malaria Map
The success of global malaria programs has raised interest in the concept of malaria elimination, or the reduction of incidence of locally acquired malaria infection to zero as a result of deliberate efforts. This has become a focal point of WHO’s Global Technical Strategy 2016–2030 and RBM’s Action and Investment to Defeat Malaria 2016–2030. Both of these programs inform the current global malaria strategy, which uses 2015 as a baseline and has the goal of reducing malaria mortality and incidence by 40 percent and eliminating malaria from 10 countries by 2020. The final target of the global malaria agenda is to reduce malaria mortality and incidence by 90 percent and eliminate malaria from 35 countries by 2030 (RBM, 2015; WHO, 2015b). Elimination now is considered to be a realistic goal. In fact, between 2007 and 2013 alone, four countries have been declared malaria free (Newby et al., 2016). At the core of elimination is the idea of “shrinking the malaria map,” which stipulates that as the incidence of malaria is reduced at its natural borders,23 reducing incidence—and thereby achieving elimination—becomes easier in the endemic heartland (Feachem and MEG, 2009; Feachem and Sabot, 2008). That 35 countries have made political commitments toward malaria elimination serves as a testament to its feasibility (Newby et al., 2016).
Challenges
Malaria has suffered from the “out of sight, out of mind” perspective. Past investments in malaria led to reduced deaths, costs, and illness for millions, yet as discussed in Chapter 3, relaxation of mosquito control efforts in several countries resulted in a resurgence of the disease. A severe example occurred in Venezuela, which had eliminated malaria in 1961—one of the first countries to do so (WHO, 2012b)—but experienced a resurgence recently due to its collapsing economy and relaxation of spraying interventions. In addition to maintaining coverage of interventions, it is vital for the global community to maintain a vigilant eye on the growing challenge of drug resistance. As explained in the next paragraph, countries have begun to observe strains of malaria resistant to baseline therapy. If these strains are not contained, they threaten global successes in controlling and eliminating malaria.
___________________
23 The endemic borders of malaria depend on a country’s latitude, altitude, and climate. The edges of these borders have lower probability of reintroduction.
The epicenter of drug resistance is the Greater Mekong subregion in Southeast Asia,24 but it could spread to India and Africa (where the largest burden lies) if governments, funders, and program implementers do not maintain vigilance (Hanboonkunupakarn and White, 2016). In addition to treatment resistance, insecticide resistance has also grown. Although insecticide control relies on four classes of compounds,25 the pyrethroids are the most commonly used but unfortunately have the most widespread resistance—an issue that especially threatens sub-Saharan Africa (Hemingway et al., 2016; WHO, 2016d). The growing challenge of both types of resistance poses a significant obstacle toward the integrated management of malaria. Unless commitment is maintained and new tools and strategies are developed, further resistance is likely to spread.
Corporate Investments in Malaria
The private sector has played a particularly important role in malaria control and elimination, appreciating that malaria places a tremendous burden on the businesses and workforce in many LMICs. Corporate investments in malaria, which range from direct financing of interventions, to supply-chain strengthening, to workforce capacity support, occur through two approaches: (1) protecting employees and operational site communities and (2) forming broad partnerships with national programs (Brieger, 2013). The majority of direct financing and in-kind donations for malaria have been provided by the oil/gas and minerals industries. For example, ExxonMobil donated $500,000 to establish a malaria diagnostic laboratory in the HopeXchange Medical Center in Kumasi, Ghana (Menka, 2011), and it has also contributed to reducing malaria cases among workers through a chemoprophylaxis compliance testing program (Diara et al., 2012). AngloGold Ashanti developed its own malaria control program focused on indoor spraying in the Obuasi District in Ghana in 2005 that contributed to a 74 percent reduction in malaria cases (Brieger, 2013). Similarly, Kinross Gold Corporation launched a $3.2 million 4-year malaria program in the western region of Ghana to provide vector control interventions and treatment services that resulted in a 45 percent reduction of malaria incidence in the community (Stiles-Ocran, 2013).
___________________
24 The Greater Mekong subregion, which comprises Cambodia, China (Yunnan Province), Lao PDR, and Myanmar (Burma), experiences multidrug resistance and insecticide resistance. Due to its geographical heterogeneity and the presence of multiple malaria parasites, the region is notoriously complex for malaria control and elimination. As a result, the Greater Mekong subregion is considered to be one of the most dangerous foci of malaria in Southeast Asia (Cui et al., 2012; WHO, 2016a).
25 The four classes of insecticides are organochlorides, organophosphates, carbamates, and pyrethroids (WHO, 2016d).
Although multinational companies may have made such investments in isolation at first, now they often invest in coordination through involvement with the Global Business Coalition for Health, which has three key initiatives: leading the Corporate Alliance on Malaria in Africa, which has a membership of 15 companies (CAMA, 2016); serving as the focal point for the Global Fund’s Private Sector Delegation (GBCHealth, 2014b); and the Private Sector Constituency of RBM (GBCHealth, 2014a). Because of their affiliation with the Global Business Coalition for Health, member organizations of the Corporate Alliance on Malaria in Africa have a unique interface with the global malaria space. Many of these corporate investments are examples of both social goals and corporate goals being achieved. For diseases such as malaria, which have commitment from multiple angles across the global landscape and can provide opportunity for private-sector involvement both to address the disease burden and help corporations’ bottom line, the committee sees a continuing and important role for the private sector in this next stage of global health.
U.S. Commitment: President’s Malaria Initiative
As noted, the United States is the single largest international funder for malaria, and in 2016 contributed $861 million for global malaria control, with the majority of the activities being conducted by PMI. Launched in 2005 to assist a set of 15 focus countries that had a high burden of malaria (PMI, 2017a), PMI is directed by the Global Malaria Coordinator within USAID. PMI now provides surveillance, prevention, treatment, research, and health system–strengthening services to 20 partner countries in Africa and Southeast Asia (PMI, 2017a). Through a focus on four key proven interventions,26 PMI has been able to contribute to a global decline of malaria mortality by 48 percent, a decline of malaria cases by 37 percent, and an avoidance of 6.2 million deaths of children under five in sub-Saharan Africa (PMI, 2016b).
From many accounts, PMI is considered a successful program not only because of its coordination with global partners, but also because of its
___________________
26 PMI focuses on four proven interventions: indoor residual spraying, artemisinin-based combination therapy, insecticide-treated mosquito nets and long-lasting insecticide-treated nets, and intermittent preventative treatment for pregnant women (PMI, 2016a). As PMI’s approach is focused on achieving results, almost 50 percent of its funding is directed toward purchasing these commodities, including rapid diagnostic tests (Simon et al., 2011). In 2015 alone, PMI procured more than 42 million long-lasting insecticide-treated nets, conducted indoor residual spraying on more than 4 million houses (protecting more than 16 million people), conducted more than 21 million intermittent preventative treatments for pregnant women, and administered more than 57 million artemisinin-based combination therapies and 54 million rapid diagnostic tests (PMI, 2016a).
strong interagency coordination (Simon et al., 2011). Unlike PEPFAR, PMI was launched when the global community had already established a framework for global malaria control and strong institutions, such as RBM and the Global Fund, were already in existence. Thus, PMI was never meant to lead the global response to malaria but rather had donor coordination as a key priority at its inception (Simon et al., 2011). In fact, PMI’s decision to partner with its initial 15 focus countries was made because the Global Fund had functioning grants in place (Simon et al., 2011).
PMI has also been successful in coordinating the interagency27 programs involved in global malaria control. Although this success is partly due to the leadership of the Global Malaria Coordinator, which has been Admiral Timothy Ziemer since PMI’s inception, it is also due to the mechanism by which funding is allocated to the agencies involved (Simon et al., 2011). PMI funding decisions start at the country level, where a detailed malaria operation plan is prepared for each year of funding that describes what inputs and activities a national malaria program needs to reach targets. Each malaria operation plan covers the four priority interventions, but the balance among them is determined by the local epidemiology of malaria. After a malaria operation plan is reviewed by the PMI leadership,28 funds are moved to the implementation level. The process by which this occurs improves the interagency coordination, as allocations are made not only based on the needs of the country but also on the capability of each partner.29
Thus, just as with PEPFAR, much of PMI’s successes can be linked to the influence of its “presidential” aspect, showing how effective U.S. government programs can be when there is political commitment at the highest level of government, clear targets are set, and agency responsibility and authority are clearly designated.
SUMMARY AND RECOMMENDATION
Remarkable progress has been made in reversing the tide of malaria. From 2000 to 2015, the global community was able to reduce the number
___________________
27 PMI is housed within USAID and coordinates both USAID and CDC for implementing programs. However, it is overseen by an Interagency Advisory Group comprising representatives from USAID, CDC and the U.S. Department of Health and Human Services, the U.S. Department of State, the U.S. Department of Defense, the National Security Council, and the Office of Management and Budget (PMI, 2017a).
28 The leadership involved with the malaria operation plan review is the PMI Interagency Technical Working Group, the PMI Coordinator, and the PMI Interagency Steering Group (Simon et al., 2011).
29 Personal communication with Michael Miller, The Kyle House Group, November 11, 2016.
of deaths due to malaria by 62 percent and the incidence of the disease by 41 percent (WHO, 2016d). This progress has been possible in large part as a result of increases in global programmatic and financial support—from $100 million in 2000 to $2.9 billion in 2015 (WHO, 2013, 2016d). As a result, elimination of malaria has become a realistic goal, with many countries not only making commitments to elimination but also declaring themselves malaria-free. Much of this progress has been possible through U.S. support, notably through the PMI, which has been praised for its ability to coordinate the interagency response to global malaria; collaborate with global partners; and, most important, deliver results. However, malaria still imposes a major burden of disease, with 212 million cases occurring in 2015 and enormous financial costs for affected families and countries’ economies (WHO, 2016d). Furthermore, the global community is witnessing a rise in malaria’s resistance to drugs and insecticides, which threatens the hard-won gains that have been achieved.
Conclusion: As a result of the continuing threat of malaria, it is vital for the United States to remain engaged in the global fight against the disease. The President’s Malaria Initiative (PMI) program has been highly successful within the U.S. Agency for International Development under the leadership of the PMI coordinator. The committee concludes that PMI is a well-designed interagency model that warrants continuous and sustainable funding.
Recommendation 6: Sustain Progress Toward Malaria Elimination
Relevant agencies of the U.S. government should continue their commitment to the fight against malaria through the President’s Malaria Initiative and collaborative work with all partners toward elimination of the disease.
PUBLIC HEALTH COMPLACENCY AND RESISTANCE: A RATIONALE FOR CONTINUED INVESTMENT
There is no doubt that gains have been made against HIV/AIDS, TB, and malaria. Because of the investments by the United States and the global community, HIV is no longer necessarily a death sentence for the poorest in the world, millions of deaths due to TB have been averted, and malaria elimination has become a real possibility for many countries. Maintaining programmatic and financial commitment toward these diseases is vital if these gains are to be sustained. All too often, a reduction of the burden can result in complacency and cause prioritization to subside. Lapses in commitment occurred with both TB and malaria in the mid-1900s, followed by
outbreaks and resurgence of both diseases worldwide in the 1980s. These disastrous results were incredibly expensive to reverse, especially for TB. New York City alone spent $1 billion to curb a TB epidemic in the 1990s (Frieden et al., 1993). The ability of these diseases to rebound should serve as a cautionary tale for public health vigilance and as a rationale for continued U.S. investments in PEPFAR, global TB control, and PMI.
HIV/AIDS has thus far not suffered from this cycle of neglect and panic. However, if interest wanes, it is likely that resurgence of HIV will also occur, a predictable but avoidable result. With 9.5 million people directly benefitting from PEPFAR-provided antiretroviral therapy in 2015, and many more secondarily benefitting through decreased rates of HIV transmission, a decrease in PEPFAR funding could be disastrous to the communities that PEPFAR serves. PEPFAR acknowledged this possibility in its 2016 Annual Report to Congress, noting that countries like Uganda demonstrate how easily progress can be reversed if continual focus and pandemic control efforts are not maintained (PEPFAR, 2016a).
In addition to the risk of resurgence, sustained involvement is important to contain the spread of antimicrobial resistance. As discussed in the previous sections, resistance has created a global emergency for TB and significantly threatens the progress of integrated malaria control. As national borders become more porous and globalization increases global travel, coordination among neighboring countries becomes vital to contain resistance. This need is most apparent for malaria, as indicated by the massive efforts by PMI and global partners to contain artemisinin and insecticide resistance in the Greater Mekong subregion in Southeast Asia (PMI, 2017b; WHO, 2016b). If resistance in this region were to spread to nearby countries, such as India, or the African continent, progress toward global malaria control efforts could be not only halted but also reversed (Cui et al., 2012). This concern goes beyond just neighboring countries as well, as four patients in the United Kingdom recently recorded failure of frontline drug treatment after returning home from visiting countries in Africa in 2015–2016 (UCLH, 2017).
To finish the work that began decades ago and build the capacity of so many countries, it is important to address the three unfinished agendas for HIV/AIDS, TB, and malaria. However, the rise in resistance for all three diseases threatens the completion of these agendas. Although recent advancements in research and development, such as bedaquiline for MDR-TB, are praiseworthy, a sustained, concerted effort to contain resistance is crucial to ensure that the global community’s hard-won gains are not lost. Public health complacency has reversed progress in the past. If the United States and its partners do not want to see history repeat itself, then a continued investment in the agendas of HIV, TB, and malaria is imperative.
REFERENCES
AVERT (AVERTing HIV and AIDS). 2017. Funding for HIV and AIDS. http://www.avert.org/professionals/hiv-around-world/global-response/funding (accessed November 15, 2016).
Brieger, W. 2013. The corporate challenge: Help defeat malaria. Cambridge, UK: Africa Health.
CAMA (Corporate Alliance on Malaria in Africa). 2016. Corporate Alliance on Malaria in Africa. New York: GBCHealth.
CDC (U.S. Centers for Disease Control and Prevention). 2013. Chapter 4: Diagnosis of tuberculosis disease. In Core Curriculum on Tuberculosis: What the Clinician Should Know. Atlanta, GA: Centers for Disease Control and Prevention.
CDC. 2016a. Reported tuberculosis in the United States, 2015. Atlanta, GA: Centers for Disease Control and Prevention.
CDC. 2016b. Multidrug-resistant tuberculosis (MDR-TB). https://www.cdc.gov/tb/publications/factsheets/drtb/mdrtb.htm (accessed March 3, 2017).
CDC. 2016c. Impact of malaria. https://www.cdc.gov/malaria/malaria_worldwide/impact.html (accessed March 7, 2017).
Cegielski, J. P. 2010. Extensively drug-resistant tuberculosis: “There must be some kind of way out of here.” Clinical Infectious Diseases 50(Supplement_3):S195-S200.
Cohen, J. 2016. AIDS epidemic nears control in three African countries. Science 354(6317): 1213.
Cui, L., G. Yan, J. Sattabongkot, Y. Cao, B. Chen, X. Chen, Q. Fan, Q. Fang, S. Jongwutiwes, D. Parker, J. Sirichaisinthop, M. P. Kyaw, X. Z. Su, H. Yang, Z. Yang, B. Wang, J. Xu, B. Zheng, D. Zhong, and G. Zhou. 2012. Malaria in the greater Mekong subregion: Heterogeneity and complexity. Acta Tropica 121(3):227-239.
Daschle, T., and B. Frist. 2015. The case for strategic health diplomacy: A study of PEPFAR. Washington, DC: Bipartisan Policy Center.
Desikan, P. 2013. Sputum smear microscopy in tuberculosis: Is it still relevant? The Indian Journal of Medical Research 137(3):442-444.
Diara, M., A. Nowosiwsky, S. Harmen, N. Burke, and M. Alilio. 2012. Enabling factors for improved malaria chemoprophylaxis compliance. The American Journal of Tropical Medicine and Hygiene 87(5):960-961.
European Alliance against Malaria. 2007. Malaria and poverty. http://www.malariaconsortium.org/userfiles/file/Past%20events/factsheet2%20-%20malaria%20and%20poverty.pdf (accessed March 7, 2017).
Feachem, R., and O. Sabot. 2008. A new global malaria eradication strategy. The Lancet 371(9624):1633-1635.
Feachem, R. G. A., and MEG (The Malaria Elimination Group). 2009. Shrinking the malaria map: A guide on malaria elimination for policy makers. San Francisco: The Global Health Group, Global Health Sciences, University of California, San Francisco.
Frieden, T. R., T. Sterling, A. Pablos-Mendez, J. O. Kilburn, G. M. Cauthen, and S. W. Dooley. 1993. The emergence of drug-resistant tuberculosis in New York City. New England Journal of Medicine 328(8):521-526.
Frieden, T. R., P. I. Fujiwara, R. M. Washko, and M. A. Hamburg. 1995. Tuberculosis in New York City—turning the tide. New England Journal of Medicine 333(4):229-233.
GBCHealth. 2014a. Roll Back Malaria private sector constituency. http://www.gbchealth.org/focal-point-roles/roll-back-malaria-private-sector-constituency (accessed February 15, 2017).
GBCHealth. 2014b. Global Fund private sector delegation. http://www.gbchealth.org/focalpoint-roles/global-fund-private-sector-delegation (accessed February 13, 2017).
Getahun, H., C. Gunneberg, R. Granich, and P. Nunn. 2010. HIV infection–associated tuberculosis: The epidemiology and the response. Clinical Infectious Diseases 50(Supplement_3):S201-S207.
Ginsberg, A. M., and M. Spigelman. 2007. Challenges in tuberculosis drug research and development. Nature Medicine 13(3):290-294.
Global Fund (Global Fund to Fight AIDS, Tuberculosis and Malaria). 2016. The Global Fund strategy 2017-2022: Investing to end epidemics. Geneva, Switzerland: Global Fund to Fight AIDS, Tuberculosis and Malaria.
Global Fund. 2017. Sourcing & management of health products. https://www.theglobalfund.org/en/sourcing-management (accessed April 25, 2017).
Green, A. 2016. NIH project focuses on integration of HIV and NCD care. Lancet 388(10054): 1869.
Hanboonkunupakarn, B., and N. J. White. 2016. The threat of antimalarial drug resistance. Tropical Diseases, Travel Medicine and Vaccines 2(1):10.
Harries, A. D., D. Maher, and P. Nunn. 1998. An approach to the problems of diagnosing and treating adult smear-negative pulmonary tuberculosis in high-HIV-prevalence settings in sub-Saharan Africa. Bulletin of the World Health Organization 76(6):651-662.
Hemingway, J., H. Ranson, A. Magill, J. Kolaczinski, C. Fornadel, J. Gimnig, M. Coetzee, F. Simard, D. K. Roch, C. K. Hinzoumbe, J. Pickett, D. Schellenberg, P. Gething, M. Hoppé, and N. Hamon. 2016. Averting a malaria disaster: Will insecticide resistance derail malaria control? The Lancet 387(10029):1785-1788.
HHS (U.S. Department of Health and Human Services). 2016. Global statistics. https://www.aids.gov/hiv-aids-basics/hiv-aids-101/global-statistics (accessed February 1, 2017).
IOM (Institute of Medicine). 2013. Evaluation of PEPFAR. Washington, DC: The National Academies Press.
Karumbi, J., and P. Garner. 2015. Directly observed therapy for treating tuberculosis. The Cochrane Database of Systematic Reviews(5):1-56.
Kates, J., A. Wexler, and E. Lief. 2016. Financing the response to HIV in low- and middle-income countries. Washington, DC: Henry J. Kaiser Family Foundation; UNAIDS.
Kemp, J. R., G. Mann, B. N. Simwaka, F. M. Salaniponi, and S. B. Squire. 2007. Can Malawi’s poor afford free tuberculosis services? Patient and household costs associated with a tuberculosis diagnosis in Lilongwe. Bulletin of the World Health Organization 85(8):580-585.
Keshavjee, S., and P. E. Farmer. 2012. Tuberculosis, drug resistance, and the history of modern medicine. New England Journal of Medicine 367(10):931-936.
KFF (Kaiser Family Foundation). 2017a. U.S. global health funding: Bilateral tuberculosis (TB), FY2001-FY2017 request. http://kff.org/global-health-policy/slide/u-s-global-health-funding-global-tuberculosis-tb, July 20. http://www.kff.org/global-health-policy/factsheet/the-u-s-government-and-global-tuberculosis-efforts (accessed March 13, 2017).
KFF. 2017b. The U.S. government and global tuberculosis efforts. http://www.kff.org/global-health-policy/fact-sheet/the-u-s-government-and-global-tuberculosis-efforts (accessed March 13, 2017).
KFF. 2017c. The U.S. President’s Emergency Plan for AIDS Relief (PEPFAR). http://kff.org/global-health-policy/fact-sheet/the-u-s-presidents-emergency-plan-for (accessed March 3, 2017).
Laurence, Y. V., U. K. Griffiths, and A. Vassall. 2015. Costs to health services and the patient of treating tuberculosis: A systematic literature review. Pharmacoeconomics 33(9):939-955.
Laurenzi, M., A. Ginsberg, and M. Spigelman. 2007. Challenges associated with current and future TB treatment. Infectious Disorders–Drug Targets 7(2):105-119.
Lawn, S. D., P. Mwaba, M. Bates, A. Piatek, H. Alexander, B. J. Marais, L. E. Cuevas, T. D. McHugh, L. Zijenah, N. Kapata, I. Abubakar, R. McNerney, M. Hoelscher, Z. A. Memish, G. B. Migliori, P. Kim, M. Maeurer, M. Schito, and A. Zumla. 2013. Advances in tuberculosis diagnostics: The Xpert MTB/RIF assay and future prospects for a point-of-care test. Lancet Infectious Diseases 13(4):349-361.
Lima, V. D., R. Granich, P. Phillips, B. Williams, and J. S. G. Montaner. 2013. Potential impact of The U.S. President’s Emergency Plan for AIDS Relief on the tuberculosis/HIV coepidemic in selected sub-Saharan African countries. The Journal of Infectious Diseases 208(12):2075-2084.
Menka, E. 2011. ExxonMobil Foundation provides $500,000 for malaria diagnosis in Ghana. Ghana Business News, July 20. https://www.ghanabusinessnews.com/2011/07/20/exxonmobil-foundation-provides-500000-for-malaria-diagnosis-in-ghana (accessed March 13, 2017).
Morrison, J. S., and P. Neiburg. 2014. Strategic U.S. leadership—essential to address the global tuberculosis pandemic. Washington, DC: Center for Stratgic and International Studies.
Nachega, J. B., and R. E. Chaisson. 2003. Tuberculosis drug resistance: A global threat. Clinical Infectious Diseases 36(Supplement_1):S24-S30.
Nájera, J. A. 2001. Malaria control: Achievements, problems and strategies. Parassitologia 43(1-2):1-89.
Nájera, J. A., M. González-Silva, and P. L. Alonso. 2011. Some lessons for the future from the Global Malaria Eradication Programme (1955–1969). PLOS Medicine 8(1):e1000412.
Newby, G., A. Bennett, E. Larson, C. Cotter, R. Shretta, A. A. Phillips, and R. G. A. Feachem. 2016. The path to eradication: A progress report on the malaria-eliminating countries. The Lancet 387(10029):1775-1784.
Nieburg, P., and A. Jackson. 2016. U.S. government roles in control of global tuberculosis: Opportunities for strengthening program effectiveness. Washington, DC: Center for Strategic and International Studies.
Nieburg, P., T. Dubovi, and S. Angelo. 2015. Tuberculosis: A complex health threat. Washington, DC: Center for Strategic and International Studies.
NIH (National Institutes of Health) Fogarty International Center. 2016. Partnership initiative for health professional education and research in selected PEPFAR countries. https://www.fic.nih.gov/About/Advisory/Pages/funding-concepts.aspx#partnerhsip (accessed February 15, 2017).
Noyes, J., and J. Popay. 2007. Directly observed therapy and tuberculosis: How can a systematic review of qualitative research contribute to improving services? A qualitative meta-synthesis. Journal of Advanced Nursing 57(3):227-243.
OARAC (Office of AIDS Research Advisory Council). 2015. OARAC meeting minutes. https://www.oar.nih.gov/oarac/meetings/April_16_2015.asp (accessed December 10, 2016).
Ogden, J., G. Walt, and L. Lush. 2003. The politics of “branding” in policy transfer: The case of DOTS for tuberculosis control. Social Science and Medicine 57(1):179-188.
Oomman, N., M. Bernstein, and S. Rosenzweig. 2007. Following the funding for HIV/AIDS: A comparative analysis of the funding practices of PEPFAR, the Global Fund and World Bank map in Mozambique, Uganda, and Zambia. Washington, DC: Center for Global Development.
Pasipanodya, J. G., and T. Gumbo. 2013. A meta-analysis of self-administered vs. directly observed therapy effect on microbiologic failure, relapse, and acquired drug resistance in tuberculosis patients. Clinical Infectious Diseases 57(1):21-31.
Patel, K. B., R. Belmonte, and H. M. Crowe. 1995. Drug malabsorption and resistant tuberculosis in HIV-infected patients. New England Journal of Medicine 332(5):336-337.
Pawlowski, A., M. Jansson, M. Sköld, M. E. Rottenberg, and G. Källenius. 2012. Tuberculosis and HIV co-infection. PLOS Pathogens 8(2):e1002464.
PEPFAR (The U.S. President’s Emergency Plan for AIDS Relief). 2009. Guidance for PEPFAR partnership frameworks and partnership framework implementation plans. Washington, DC: The U.S. President’s Emergency Plan for AIDS Relief.
PEPFAR. 2012a. PEPFAR 3.0—controlling the epidemic: Delivering on the promise of an AIDS-free generation. Washington, DC: The U.S. President’s Emergency Plan for AIDS Relief.
PEPFAR. 2012b. Report on costs of treatment in the President’s Emergency Plan for AIDS Relief (PEPFAR). Washington, DC: The Office of the U.S. Global AIDS Coordinator and Health Diplomacy, U.S. Department of State.
PEPFAR. 2015. PEPFAR 2015 annual report to Congress. Washington, DC: The Office of the U.S. Global AIDS Coordinator and Health Diplomacy, U.S. Department of State.
PEPFAR. 2016a. PEPFAR 2016 annual report to Congress. Washington, DC: The Office of the U.S. Global AIDS Coordinator and Health Diplomacy, U.S. Department of State.
PEPFAR. 2016b. PEPFAR dashboards. https://data.pepfar.net/global (accessed March 16, 2017).
PEPFAR. 2016c. PEPFAR funding: Fiscal year 2014-2017. Washington, DC: The Office of the U.S. Global AIDS Coordinator and Health Diplomacy, U.S. Department of State.
PEPFAR. 2017a. PEPFAR 2017 annual report to Congress. Washington, DC: The Office of the U.S. Global AIDS Coordinator and Health Diplomacy, U.S. Department of State.
PEPFAR. 2017b. Working together for an AIDS-free future for girls and women. https://www.pepfar.gov/partnerships/ppp/dreams (accessed December 10, 2016).
Petrakos, C. 1993. Catching a killer: Before we were stalked by AIDS, we were menaced by TB. The Chicago Tribune. http://articles.chicagotribune.com/1993-08-29/entertainment/9308290161_1_tb-tuberculosis-forgotten-plague (accessed January 18, 2017).
Pietersen, E., E. Ignatius, E. M. Streicher, B. Mastrapa, X. Padanilam, A. Pooran, M. Badri, M. Lesosky, P. van Helden, F. A. Sirgel, R. Warren, and K. Dheda. 2014. Long-term outcomes of patients with extensively drug-resistant tuberculosis in South Africa: A cohort study. The Lancet 383(9924):1230-1239.
PMI (President’s Malaria Initiative). 2016a. A decade of progress: Tenth annual report to Congress. Washington, DC: The President’s Malaria Initiative.
PMI. 2016b. PMI by the numbers. https://www.pmi.gov/docs/default-source/default-document-library/pmi-reports/pmi-by-the-numbers.pdf (accessed March 15, 2017).
PMI. 2017a. About. https://www.pmi.gov/about (accessed March 8, 2017).
PMI. 2017b. Greater Mekong subregion. https://www.pmi.gov/where-we-work/mekong (accessed March 19, 2017).
Pooran, A., E. Pieterson, M. Davids, G. Theron, and K. Dheda. 2013. What is the cost of diagnosis and management of drug resistant tuberculosis in South Africa? PLOS ONE 8(1):e54587.
Purdy, M., M. Robinson, K. Wei, and D. Rublin. 2013. The economic case for combating malaria. The American Journal of Tropical Medicine and Hygiene 89(5):819-823.
RBM (Roll Back Malaria Partnership). 2011. Eliminating malaria: Learning from the past, looking ahead: Progress and impact series. Geneva, Switzerland: Roll Back Malaria Partnership.
RBM. 2015. Action and investment to defeat malaria: 2016-2030. Geneva, Swizerland: Roll Back Malaria Partnership Secretariat, World Health Organization.
Resch, S., T. Ryckman, and R. Hecht. 2015. Funding AIDS programmes in the era of shared responsibility: An analysis of domestic spending in 12 low-income and middle-income countries. The Lancet Global Health 3(1):e52-e61.
Richter, R. 2012. 740,000 lives saved: Study documents benefits of AIDS relief program. https://med.stanford.edu/news/all-news/2012/05/740000-lives-saved-study-documents-benefits-of-aids-relief-program.html (accessed January 5, 2017).
Shah, N. S., S. C. Auld, J. C. M. Brust, B. Mathema, N. Ismail, P. Moodley, K. Mlisana, S. Allana, A. Campbell, T. Mthiyane, N. Morris, P. Mpangase, H. van der Meulen, S. V. Omar, T. S. Brown, A. Narechania, E. Shaskina, T. Kapwata, B. Kreiswirth, and N. R. Gandhi. 2017. Transmission of extensively drug-resistant tuberculosis in South Africa. New England Journal of Medicine 376(3):243-253.
Sharma, S. K., M. Kohli, R. N. Yadav, J. Chaubey, D. Bhasin, V. Sreenivas, R. Sharma, and B. K. Singh. 2015. Evaluating the diagnostic accuracy of Xpert MTB/RIF assay in pulmonary tuberculosis. PLOS ONE 10(10):e0141011.
Simon, J., K. Yeboah-Antwi, A. Schapita, M. K. Cham, R. Barber-Maddan, and M. I. Brooks. 2011. External evaluation of the President’s Malaria Initiative: Final report. Washington, DC: The QED Group LLC, Camris International, Social & Scientific Systems Inc.
Singhal, R., and V. P. Myneedu. 2015. Microscopy as a diagnostic tool in pulmonary tuberculosis. International Journal of Mycobacteriology 4(1):1-6.
Stiles-Ocran, J. 2013. The Chirano Gold Standard: A successful private sector initiative against malaria. Accra: Chirano Malaria Control Programme.
Sturchio, J. L., and G. M. Cohen. 2012. How PEPFAR’s public-private partnerships achieved ambitious goals, from improving labs to strengthening supply chains. Health Affairs (Millwood) 31(7):1450-1458.
Suchindran, S., E. S. Brouwer, and A. Van Rie. 2009. Is HIV infection a risk factor for multidrug resistant tuberculosis? A systematic review. PLOS ONE 4(5):e5561.
Tarnoff, C., and M. L. Lawson. 2016. Foreign aid: An introduction to U.S. programs and policy. Washington, DC: Congressional Research Service.
UNAIDS (Joint United Nations Programme on HIV/AIDS). 2014a. 90-90-90: An ambitious treatment target to help end the AIDS epidemic. Geneva, Switzerland: Joint United Nations Programme on HIV/AIDS.
UNAIDS. 2014b. The gap report. Geneva, Switzerland: Joint United Nations Programme on HIV/AIDS.
UNAIDS. 2015a. AIDS by the numbers 2015. Geneva, Switzerland: Joint United Nations Program on HIV/AIDS.
UNAIDS. 2015b. How AIDS changed everything—MDG6: 15 years, 15 lessons of hope from the AIDS response. Geneva, Switzerland: Joint United Nations Programme on HIV/AIDS.
UNAIDS. 2015c. On the fast-track to end AIDS: UNAIDS strategy 2016-2021. Geneva, Switzerland: Joint United Nations Programme on HIV/AIDS
UNAIDS. 2015d. People living with HIV (all ages). Geneva, Switzerland: Joint United Nations Programme on HIV/AIDS.
UNAIDS. 2016a. AIDSinfo. http://aidsinfo.unaids.org (accessed March 1, 2017).
UNAIDS. 2016b. Fact sheet November 2016. http://www.unaids.org/en/resources/fact-sheet (accessed December 12, 2016).
UNICEF (United Nations International Children’s Emergency Fund). 2004. Fact sheet: Malaria, a global crisis. New York: United Nations International Children’s Emergency Fund.
University College London Hospitals. 2017. First recorded cases of frontline malaria drug failing to treat UK patients. London, UK: University College London Hospitals.
USAID (U.S. Agency for International Development). 2015. United States government Global Tuberculosis Strategy: 2015-2019. Washington, DC: United States Agency for International Development.
USAID. 2016. USAID’s bedaquiline donation program in partnership with Johnson and Johnson. https://www.usaid.gov/what-we-do/global-health/tuberculosis/technical-areas/bedaquiline-donation-program (accessed March 19, 2017).
Wagner, Z., J. Barofsky, and N. Sood. 2015. PEPFAR funding associated with an increase in employment among males in ten sub-Saharan African countries. Health Affairs (Millwood) 34(6):946-953.
Whalen, C., C. R. Horsburgh, D. Hom, C. Lahart, M. Simberkoff, and J. Ellner. 1995. Accelerated course of human immunodeficiency virus infection after tuberculosis. American Journal of Respiratory and Critical Care Medicine 151(1):129-135.
White House. 2015. National Action Plan for Combatting Multidrug-Resistant Tuberculosis. Washington, DC: The White House.
WHO (World Health Organization). 2001. Global tuberculosis control report, 2001. Geneva, Switzerland: World Health Organization.
WHO. 2012a. “Totally drug-resistant TB”: A WHO consultation on the diagnostic definition and treatment options. Geneva, Switzerland: World Health Organization.
WHO. 2012b. World malaria report 2012. Geneva, Switzerland: World Health Organization.
WHO. 2013. World malaria report 2013. Geneva, Switzerland: World Health Organization.
WHO. 2014. World malaria report 2014. Geneva, Switzerland: World Health Organization.
WHO. 2015a. World malaria report 2015. Geneva, Switzerland: World Health Organization.
WHO. 2015b. Global Technical Strategy for Malaria: 2016-2030. Geneva, Switzerland: World Health Organization.
WHO. 2016a. Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection: Recommendations for a public health approach. Geneva, Switzerland: World Health Organization.
WHO. 2016b. Eliminating malaria in the greater Mekong subregion: United to end a deadly disease. Geneva, Switzerland: World Health Organization.
WHO. 2016c. Global tuberculosis report 2016. Geneva, Switzerland: World Health Organization.
WHO. 2016d. World malaria report 2016. Geneva, Switzerland: World Health Organization.
WHO. 2017a. High-risk groups. http://www.who.int/malaria/areas/high_risk_groups/en (accessed February 10, 2017).
WHO. 2017b. Tuberculosis and HIV. http://www.who.int/hiv/topics/tb/en (accessed March 10, 2017).
Zumla, A., J. Chakaya, R. Centis, L. D’Ambrosio, P. Mwaba, M. Bates, N. Kapata, T. Nyirenda, D. Chanda, S. Mfinanga, M. Hoelscher, M. Maeurer, and G. B. Migliori. 2015. Tuberculosis treatment and management: An update on treatment regimens, trials, new drugs, and adjunct therapies. The Lancet Respiratory Medicine 3(3):220-234.


































