5
Research and Development Actions for Reducing the Need for Antimicrobials
Session III of the workshop, moderated by Kent Kester, vice president and head of translational science and biomarkers at Sanofi Pasteur, focused on research and development actions aimed at reducing the need for antimicrobials. The human health perspective on effective scientific advances and promising research was provided by L. Clifford McDonald, associate director for science in the Division of Healthcare Quality Promotion at the U.S. Centers for Disease Control and Prevention (CDC). Timothy Johnson, associate professor of microbiology at the University of Minnesota, described alternatives to antibiotic use in animal health and production agriculture. Keith Klugman, director for pneumonia at the Bill & Melinda Gates Foundation (BMGF), discussed the potential for vaccination to reduce the burden of antimicrobial resistance. Ellen Jo Baron, professor emerita at Stanford University and executive director of medical affairs at Cepheid, described research and development actions in the area of diagnostics. Gregory Daniel, deputy director and clinical professor at the Duke-Margolis Center for Health Policy, explored economic strategies for accelerating research and development for new antimicrobials.
SCIENTIFIC ADVANCES AND PROMISING RESEARCH TO REDUCE THE NEED FOR ANTIMICROBIALS FROM A HUMAN AND AN ANIMAL HEALTH PERSPECTIVE
Human Health Perspective: Research on the Microbiome
“New drugs alone aren’t enough to protect Americans,” said McDonald. He explained that combating antimicrobial resistance will require comprehensive, aggressive action across the U.S. government and around the globe on multiple fronts to prevent the spread of infectious disease, to track the spread of resistance and disease, to improve stewardship, and to develop new drugs and diagnostics.
CDC has gained useful experience in addressing health care–associated infections (HAIs), said McDonald. Many HAIs have been prevented by mobilizing grassroots patient support, he said, to spur local governments to mandate state reporting and action. He emphasized the importance of patient-centered messaging about antibiotic-resistant infections: When messaging to the public, he said, CDC emphasizes that “you don’t become resistant to antibiotics, but the bacteria in and on your body can.” While many more infections are now preventable using existing technologies and better stewardship, he said, discovering how to prevent other types of HAIs and antibiotic-resistant infections will require new approaches and innovation.
CDC’s Approach to Combating Antibiotic Resistance
McDonald provided an overview of CDC’s approaches to combating antimicrobial resistance. Work with academic and health care partners includes 28 collaborative groups of hospitals, he said, that are devising innovative ways to protect patients and to scale up effective interventions across health systems. Eleven prevention epicenters, which are academic centers of excellence, are identifying new prevention strategies to guide clinical practice and to maximize public health impact, he said. CDC’s Broad Agency Announcement is an innovative funding contract mechanism that invites applicants from academia, government, and industry to work with CDC. To date, he reported, this has generated 260 white paper submissions as well as 14 1-year studies exploring the gut–drug relationship and patients’ microbiomes. CDC is also working with industry partners in several initiatives including the Antibiotic Resistance Isolate Bank, which is a collaboration activity with the U.S. Food and Drug Administration (FDA) that supports the development of new drugs and diagnostics. To further spur innovation, he said, CDC is making its sequencing data from antibiotic-resistant pathogens publicly available at the National Center for Biotechnology Information.
Antibiotic Resistance and the Microbiome
In recent years, CDC has made significant investment in research on the microbiome, said McDonald, because it considers it integral to antibiotic resistance. The microbiome is a community of organisms in and on a body: the skin, gut, oral, respiratory systems, and urogenital tract are examples. When communicating to the public, he suggested using the analogy of a forest to describe how a healthy microbiome is an ecology with microorganisms that exist in complex communities. Although a single microorganism can be isolated in the laboratory, he said,
Antibiotic pressure on the community is like a forest fire that disrupts that microbiome and it wipes out all that normal flora. . . . It’s not just about selection within a species. It’s selection across species.
A healthy microbiome protects people and their communities against infection, he said, but antibiotics disrupt the microbiome by eliminating both good and bad bacteria. Resistant bacteria can take advantage of this disruption and multiply, he said, and this overgrowth can prime a person’s body for infection. Once a person is colonized, the resistant bacteria can spread easily to other people. He noted that the term antibiotic pressure more aptly refers to pressure on the individual microbiome and collectively on the human population microbiome, the environmental microbiome, and the animal microbiome.
CDC’s key premise, according to McDonald, is that an intact human microbiome is a primary host defense for preventing antibiotic-resistant outcomes of human health importance. Colonization and dominance often precede transmission, he said; dominance with a high organism load is a precedent risk factor for becoming infected with opportunists or pathobionts such as Clostridium difficile, Salmonella, Shigella, and Campylobacter as well as multidrug-resistant organisms (vancomycin-resistant Enterococci, carbapenem-resistant Enterobacteriaceae [CRE], and the extended spectrum beta-lactamases [ESBL]).1
To facilitate research on the microbiome, McDonald said, CDC is developing microbiome indices (MIs) for use in clinical medicine and public health. MIs can be used to monitor patients who are receiving antibiotics, he said, and to detect when disruption to the microbiome reaches a level that can promote antibiotic-resistant outcomes. He explained that MIs can also be used to gauge the relative risks of different antibiotics and other agents
___________________
1 The organisms are often called opportunists, but McDonald suggested that the term places too much emphasis on the immune system, when the problem actually lies with the microbiome. The term pathobiont implies an organism that causes inflammation after the microbiome has been disrupted in some way (e.g., Clostridium difficile, Salmonella, Campylobacter, CRE, and ESBLs are all pro-inflammatory).
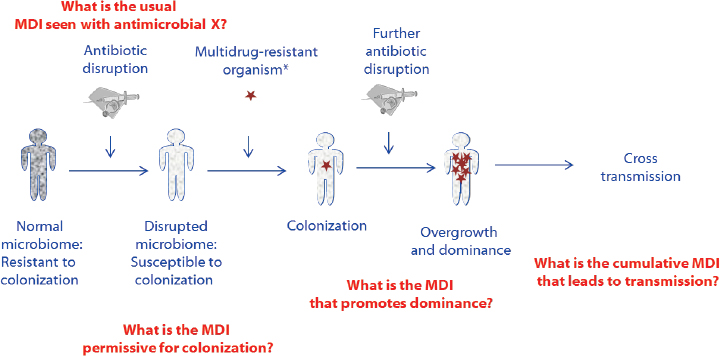
NOTE: MDI = microbiome disruption indices. * Examples include carbapenem-resistant enterobacteriaceae, vancomycin-resistant enterococci, extended-spectrum beta-lactamase producing enterobacteriaceae. May also include transfer of genetic transfer of resistance determinants.
SOURCES: McDonald presentation, June 21, 2017; adapted from Halpin et al., 2016. Reprinted from American Journal of Infection Control, Vol. 4/Edition 7, Halpin et al., “Intestinal microbiome disruption in patients in a long-term acute care hospital: A case for development of microbiome disruption indices to improve infection prevention,” Pages 830–836, Copyright (2016), with permission from Elsevier.
disruptive to the microbiome, and to develop microbiome protectants and restoratives, such as probiotics. The broad concept of MIs is to align the sciences around antibiotic-resistant outcomes (see Figure 5-1) by establishing which measures of microbiome disruption are the best risk markers for colonization, expansion, transmission, and infection with antibiotic-resistant organisms. MI research can be used to tailor antibiotic stewardship to the microbiome of a patient or patient population, said McDonald, as well as to develop better microbiome diagnostics and therapeutics.
Therapeutic developments of note, McDonald said, include using fecal microbiota transplantation to reduce resistant bacteria in patients with significantly disrupted microbiomes, such as people with multiple recurrent Clostridium difficile infections and hematopoietic stem cell transplant patients colonized with CRE (Bilinski et al., 2017). Phage therapy is also very promising for treating chronic infections involving biofilms, he said, although they are cleared quickly from the bloodstream. McDonald predicted that phage therapy will play a large role in microbiome remediation
as well as dealing with CRE hospital outbreaks that are very difficult to eradicate.
Animal Health Perspective: Alternative Approaches to Antibiotic Use
Although there is no single replacement for antibiotics in animal agriculture, Johnson said, the emergence of antibiotic alternatives on the market represents a promising development. He noted that the concept of using the self-microflora to combat pathogens has existed since 1973, when researchers discovered that feeding adult gut content from a hen to newly hatched chickens inhibits Salmonella colonization (Nurmi and Rantala, 1973). Today, there are dozens of alternative product choices on the market, he said, but veterinarians lack a platform or central resource to learn about how alternative products work in different situations. Johnson said that common alternative approaches to antibiotic use include prebiotics and probiotics to “seed” the gut with healthy flora, vaccinations, and products such as oregano, saponins, garlic, thyme, organic acids, cinnamaldehyde, and carvacrol (see Figure 5-2).
A prebiotic called XPC, a combination of extracted nutrients, is widely used across the animal agriculture industry, said Johnson. The product was initially geared toward improving feed efficiency, but it turned out also to help reduce the colonization of Salmonella in the animal’s gut. According
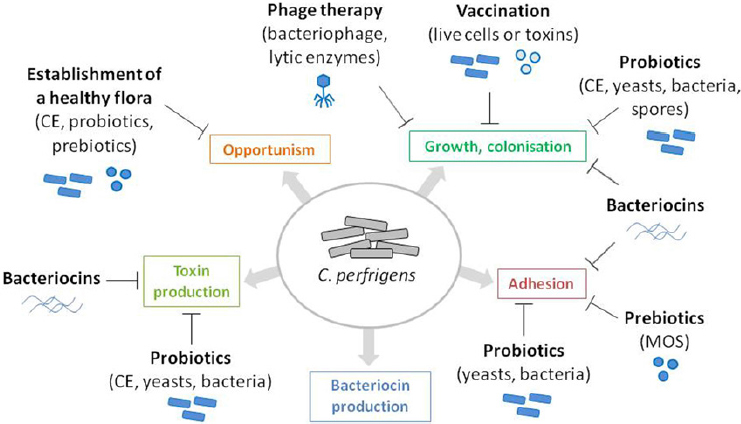
NOTE: CE = competitive exclusion; MOS = mannan-oligosaccharides.
SOURCES: Johnson presentation, June 21, 2017; Caly et al., 2015.
to Johnson, studies now show that XPC makes the remaining Salmonella in the gut less resistant by expelling one of the genomic islands that contains multidrug resistance in that Salmonella. He surmised that more products may have similar potential to affect resistance, but their mechanisms are not yet understood.
Understanding the potential effect of an antibiotic alternative requires understanding the animal microbiome, said Johnson. To work toward defining the microbiome, he said, gut samples from thousands of animals have been analyzed. He reported that predictable patterns have been exposed in the succession of the microbiome over time and in tissues from different parts of the gut. This predictability, he said, has been helpful in identifying the dominant bacteria to modulate in the microbiome. However, he reported that so far, microbiome analysis by different types of alternative treatment products (versus control) have indicated that around 95 percent of those products do not have much effect on the gut microbiome.
Custom-Tailored Vaccines and Probiotics
Vaccines are widespread in animal agriculture, Johnson noted, referring to commercial subunit vaccines based on iron-receptor proteins in bacteria that have been very effective in chicken cholera and cow mastitis. But when there is no effective commercial vaccine to use against a specific disease, he said, veterinarians can make their own “autogenous” vaccine by identifying and sending the particular flock’s strain to a company that will produce the vaccine directly. While this is highly effective, he added, the main challenge has been surveying the pathogen ecology to identify which strains would make the most effective vaccines. Developments in genomic technology are enabling more detailed surveillance of pathogen isolates, he continued. Phylogenetic analysis of different vaccine strains for the same bacteria will help to determine the best combination of strains to vaccinate with, he said, as well as serving as a diagnostic tool to understand the overall pathogen population in a flock.
Probiotics custom-tailored to a specific flock or herd will eventually dominate the market in animal agriculture, predicted Johnson. This involves cross-referencing a library of potential probiotic strains against a library of flock-specific pathogens, he explained, to identify the appropriate combination of probiotic pathogens. He noted that this technique is already highly effective in poultry flocks with chronic Escherichia coli infection. However, he added that evidence already indicates that resistance to the probiotic strains can develop over time, even if the strains are cycled.
A new area of research is taking the probiotic approach a step further to host-specific probiotics, Johnson said, such as turkey-specific products containing combinations of strains from turkeys only. He presented evi-
dence showing that when comparing controls, nonhost-specific commercial probiotics, antibiotics, and a turkey-specific probiotic, only the turkey-specific probiotic had an effect on performance and was associated with increased bacterial diversity in the turkey gut.2 He said that this underscores the need to further explore the microbiome–host connection. He also suggested conducting mechanistic studies about how the effective products work, as well as comparing different combinations of products. Johnson warned that it is still not possible to compare the effects of alternative products in different types of animals—or even among different breeds of the same animal. He also suggested researching the effects of timing and animal stress on the effectiveness of alternative products.
VACCINATION TO REDUCE THE BURDEN OF ANTIMICROBIAL RESISTANCE
Vaccines have been underresearched, argued Klugman, and they have not been sufficiently prioritized as a potential modality for reducing antimicrobial resistance in both humans and animals. Evidence demonstrates that existing vaccines designed to target antimicrobial-resistant bacterial pathogens can affect both the use of antibiotics and the burden of resistance, he said. Other types of vaccines given to humans may also affect antibiotic resistance and/or use, he said, but many lack published data on resistance. Vaccines that warrant further study, according to Klugman, include pneumococcal conjugate vaccine (PCV), Haemophilus influenzae type B vaccine, cholera vaccine, meningococcal vaccines, tuberculosis (TB) vaccines, and vaccines under development for typhoid and nontyphoid Salmonella and group B strep.3
Competing Selection of Vaccines and Antibiotic Use on Antimicrobial Resistance
Good data on vaccine impact on resistance comes from the PCV model, Klugman said, because PCV has reduced the burden of antibiotic-resistant pneumococcal disease globally. He reported that when given to children, PCV successfully eliminates between 10 and 13 of 98 known pneumococcal serotypes, including 90 percent of antibiotic-resistant strains. PCV also reduces resistance in adults, he said, by interrupting the transmission of
___________________
2 For more information on Johnson’s paper presented at the 2018 Midwest Poultry Federation Convention, see midwestpoultry.com/wp-content/uploads/Johnson-Tim.pdf (accessed July 30, 2017).
3 Given that one-third of pregnant women get antibiotics for fear of group B strep to prevent transmission, a vaccine could have a huge effect on use, even though it is not particularly a resistant pathogen, Klugman said.
antibiotic-resistant vaccine-type strains. Direct evidence from large randomized trials has demonstrated that PCV can reduce resistance in the Pneumococcus bacteria among children in South Africa (Klugman et al., 2003), and that PCV can prevent a total of 35 antibiotic prescriptions per 100 children vaccinated in California (Fireman et al., 2003). Recent evidence from Finland shows that rolling out the PCV vaccine reduced the use of antibiotics for respiratory infections in children, as well as reducing the proportion of resistant Pneumococcal strains (Sihvonen et al., 2017).
Vaccines may reduce resistance, Klugman warned, but continued use drives resistance in residual strains. When PCV was introduced in the United States in 2000, incidence rates decreased both for penicillin-susceptible disease and for penicillin-nonsusceptible disease among children under 2 years of age (Kyaw et al., 2006). Resistant infection rates also decreased among unvaccinated adults older than 65 years of age through herd immunity, because the vaccine interrupts transmission in the community. However, he noted, after the initial drop in incidence, a slight uptick in resistance occurred only among children less than 2 years of age. The selection of strains resistant to trimethoprim-sulfamethoxazole and penicillin was occurring only in the children and not the adults, he added (see Figure 5-3). He explained that antibiotic use continues to select resistance in the remaining 85 to 88 serotypes that PCV does not cover (in this case, it was serotype 19A). A similar trend is occurring for another strain—serotype 15A—for which there is not yet a pneumococcal vaccine. Between 1999 and 2007, susceptibility to penicillin among isolates collected by CDC surveillance dropped from 75 percent to 15 percent (Gertz et al., 2010). He warned that if the vaccine used does not prevent the major resistant strains, while an initial effect on resistance is observed, nonvaccine strains may become increasingly antibiotic resistant.
Impact of Viral Vaccines on Antibiotic Use
Vaccines that affect viral pathogens instead of bacterial pathogens can also affect antibiotic use, said Klugman. For example, influenza vaccination can reduce antibiotic use for otitis media, the leading indication for antibiotics in children, he reported. A large multinational study on the effect of the influenza vaccine on antibiotic use in healthy children has reported a 50 percent reduction in antibiotic use among children who received the vaccine compared to controls (Dbaibo et al., 2017). Klugman suggested that the measles vaccine also has the potential to prevent both antibiotic use for pneumonia complications and the need for antibiotic prophylaxis. Future viral vaccines, such as one for respiratory syncytial virus, may also affect antibiotic resistance simply because it reduces antibiotic use and therefore the attendant selection of strains in the flora, he suggested.
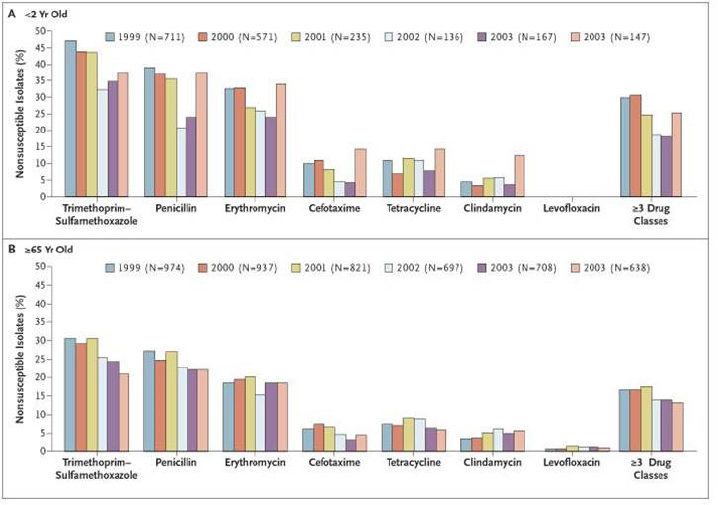
SOURCES: Klugman presentation, June 21, 2017; Kyaw et al., 2006. From the New England Journal of Medicine, Kyaw et al., “Effect of Introduction of the Pneumococcal Conjugate Vaccine on Drug-Resistant Streptococcus pneumoniae,” Vol. 354, Page 1461. Copyright © 2006 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
Klugman also recommended that new human vaccines should be developed to combat the key current antimicrobial-resistant pathogens, known as ESKAPE pathogens, which include Enterococcus faecalis, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumanii, Pseudomonas aeruginosa, and Enterobacter. These pathogens lend themselves to vaccines, which could provide short-term protection for hospitalized patients against nosocomial infection by these pathogens, as well as short-term protection against neonatal sepsis, which is increasingly becoming untreatable, particularly in developing countries, he said. Klugman suggested that alternative approaches to antibiotics such as monoclonal antibodies, which are cells that are derived from a single ancestral cell, can play a major role. Maternal immunization could also protect neonates from resistant pathogens in the first 3 months of life. Finally, Klugman offered the perspective of a human infectious disease specialist on the potential effect of the use of animal vaccines to reduce antibiotic use and resistance, citing several examples on
fish and poultry where vaccines have been an essential part of controlling antibiotic use.
RESEARCH AND DEVELOPMENT ACTIONS IN DIAGNOSTICS
To illustrate the value of investing in diagnostics, Baron described the effect that a new rapid diagnostic test (developed by Cepheid) had during the Ebola outbreak. Before the test, patients suspected of having Ebola were quarantined in a holding area for up to 8 days while waiting for results from a distant reference laboratory. When the rapid test became available, it reduced the time to an accurate diagnosis at point of care to within 2 hours. Patients not infected with Ebola were no longer at risk of exposure to other patients who were infected in the quarantine area, and health care workers—also at high risk of infection—were able to collect samples in a safer and more expeditious way. The rapid test offered increased specificity as well as the capability for ongoing monitoring by testing of semen and breast milk over the longer term.
Baron described another example of the effect of diagnostics: the Xpert MTB/RIF assay, which can detect both Mycobacterium tuberculosis (MTB) and resistance to rifampicin (RIF). Xpert MTB/RIF is now used in 122 of 145 developing countries with high burdens of TB, she said. The rollout of the test has increased RIF-resistant TB case detection by over fivefold (Sachdeva et al., 2015), and the detection of multidrug-resistant TB has increased by threefold to eightfold (Albert et al., 2016). The rollout of the test has changed the worldwide diagnostic landscape of TB, she said, and it has attracted researchers and stakeholders to the TB field. However, most studies have found no difference in TB mortality after the rollout, said Baron, which demonstrates that a diagnostic test is not the only answer to these complex problems.
Potential Prioritization of Diagnostic Tests
According to Baron, certain types of diagnostic tests should be prioritized. The first are tests for conditions in which immediate patient management decisions will affect both public health and individual patient outcomes, she suggested. For example, a study from France on intrapartum group B strep polymerase chain reaction screening for pregnant women reported that the screening is cost neutral and achieves a 50 percent reduction in the probability of group B strep disease in neonates (El Helali et al., 2012). Baron argued that knowing which patients are colonized with group B strep at the time of delivery will not only decrease the use of intrapartum prophylactic antibiotics, but will also help with the infant obesity
epidemic.4 Another example of diagnostics with both public and individual benefit concerns sexually transmitted infections, she said. One study found that using a rapid polymerase chain reaction test for chlamydia/gonorrhea in an emergency department reduced empiric treatment by 35 percent and increased notified results by 56 percent (May et al., 2016).
Baron suggested prioritizing the development of three other types of diagnostic tests. The first is any test to rapidly identify patients eligible for clinical trials of new antimicrobial agents at enrollment, such as Ebola tests for Ebola vaccine trials. The second are rapid tests that can definitively rule out bacterial infection at patient presentation, which she predicted would prevent the unnecessary use of antibiotics. The third are rapid tests to detect resistance factors directly from patient samples for screening and surveillance. She said that this type of test could inform the type of prophylaxis used for surgery, as well as helping to prevent transmission of antibiotic resistance in a hospital setting. Connectivity to enable shared knowledge of resistance factors in real time across sectors will also be critical, she said; for example, South Africa has a host information system that aggregates diagnostic test results and presents organisms, diseases, and resistance factors on a geopositioned map.
Accelerating the Development of Diagnostics
Lack of predictable profit is a barrier to the development of diagnostics, said Baron, who suggested that the Ebola diagnostic test would not have been developed by Cepheid today if the BMGF had not supported them financially. Many countries require their own clinical trials before a diagnostic can be introduced, she noted, but clinical trials are costly, lengthy, and impeded by regulatory issues. The product’s final cost and market acceptability can be a barrier to scaling up and maintaining quality, she warned. Changing microbial genetics and epidemiology are huge challenges, she said, because tests must be updated and go through new clinical trials for approval.
To achieve the greatest short-term impact, she suggested several immediate steps. First, she said that target product profiles should be publicized and made very specific, so that companies do not make a product that will not be accepted in the marketplace. Her second suggestion was to create a rapid, inexpensive regulatory path to market, which may include abbreviated clinical trials up front and then extensive post-market review and surveillance to ensure it is performing as expected. To address some of those regulatory barriers, she suggested that rather than adding stringency,
___________________
4 Baron characterized this infant obesity epidemic as a function of disturbing the microbiome of newborns who come through the birth canal in a penicillin-laden environment.
FDA should transition toward the CE marking process,5 which is required for certain products sold within the European Economic Area and allows a company to self-register a test. Finally, Baron advised that companies have no incentive to develop diagnostics without a guaranteed market, so companies should be given prepurchase agreements or sufficient sales guarantees to offset their development costs.
ECONOMIC STRATEGIES FOR ACCELERATING RESEARCH AND DEVELOPMENT FOR NEW ANTIMICROBIALS
Very few antibiotic drugs are currently in the pipeline, said Daniel, who cited data from The Pew Charitable Trusts that show that only 41 antibiotics are in some phase of clinical testing (The Pew Charitable Trusts, 2017). This is a relatively low number compared to other therapeutic areas as he explained that only a couple of those would be expected to make it to market, and it is unlikely that any of those would have efficacy against the most concerning types of multidrug-resistant infections. Large pharmaceutical companies are moving out of the antibiotic development space, he said, because of low use, very slow uptake, and low revenues.
Economic Challenges to Antimicrobial Development
Daniel explained that appropriate antibiotic stewardship presents an economic challenge for antimicrobial development, especially in the United States. New antibiotic drugs on the market have limited use, so sales volumes and revenue are very low. Furthermore, the lack of rapid diagnostic tools leads to empiric treatment with broad-spectrum antibiotics, he said, which tends to involve low-cost generic antibiotics and contributes to inappropriate use. Daniel added that the strong and effective generic market also limits the cases in which new antibiotics are needed, thus driving down prices. He noted that reimbursement for antibiotics almost never reflects the public health benefit that the antibiotics bring to the whole of society; for example, they enable invasive medical interventions and help prevent the spread of rampant infections that are multidrug resistant. Daniel argued that new models are urgently needed to reward the development of innovative antimicrobial products, while supporting stewardship and appropriate use.
___________________
5 “CE,” which originated as an abbreviation of Conformité Européenne (meaning European Conformity), is a certification mark that indicates conformity with health, safety, and environmental protection standards for products sold within the European Economic Area.
National Efforts to Combat Antimicrobial Resistance
Legislative efforts to stimulate more interest in developing antibiotics include the Generating Antibiotic Incentives Now Act of 2012, said Daniel, which effectively adds additional years of patent exclusivity. However, he explained that extending a patent does not significantly improve revenues, because appropriate stewardship limits the use of brand-new drugs on the market. Daniel said that the 21st Century Cures Act of 2016 shortened the regulatory pathway for antibiotics to enable products to reach the market sooner, but it does not affect the way that antibiotics are used or reimbursed. Daniel explained that other U.S. government efforts have included the White House National Strategy for Combating Antibiotic-Resistant Bacteria, released in 2014, which was followed by the President’s Advisory Council on Combating Antibiotic-Resistant Bacteria. The latter formed in 2015 to address the concerns about low returns on investment and recommended a combination of general and targeted incentives, Daniel said. He added that CARB-X is a public–private partnership designed to provide significant research and development funding in very early stages of antibiotic development.
Push and Pull Incentives
Daniel clarified the distinction between push and pull financial incentives. Push incentives are provided during clinical development to help stimulate research and development through the basic research, preclinical, and clinical trial phases. He said that these may include grant funding, public–private partnerships, and tax credits, for example. Pull incentives, he explained, are provided during the approval process and the post-market period and include add-on payments, market exclusivity, and delinking reimbursement from use to encourage sustainable, appropriate use. Daniel explained that in the United States, where there has been more activity on the push side without effective pull incentives, “it’s like running into a brick wall.” Few companies will enter the market without significant pull incentives, he warned.
Global efforts and expertise have converged on a fundamental principle of delinking revenues for antibiotics from the volume of use, said Daniel, to ensure return on investment regardless of how often the antibiotic is used. Market entry reward is a pull incentive that is an example of removing the “link” between development costs and revenues by paying for vital antibiotic drugs with public funds, he said. It provides substantial additional revenues to reflect public health value quickly after approval and removes the need for volume sales, explained Daniel, which helps the developer recoup investments (see Figure 5-4). Once an eligible antibiotic gets to market, the
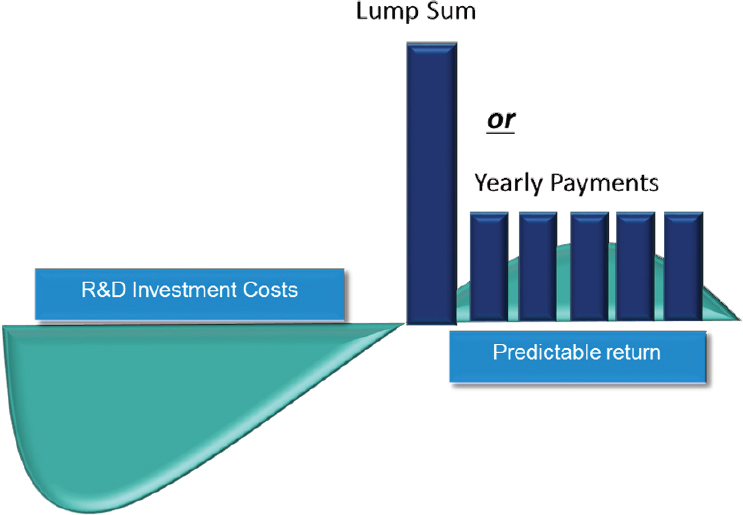
SOURCES: Daniel presentation, June 21, 2017; adapted from Drive-AB, 2016.
company either gets a lump sum or gets some yearly payment over 5 or 6 years that, in aggregate, results in a positive net present value, which he estimated at between $1 and $2 billion.
Duke-Margolis Approach to Reinvigorating the Antimicrobial Pipeline
Daniel explained that the Duke-Margolis Center for Health Policy has analyzed economic pull incentives to outline a path for feasible implementation of economic incentives in the U.S. health care system. The project generated eight core principles for antimicrobial economic incentives:
- Form part of a comprehensive strategy to provide pull incentives in combination with other push incentives.
- Promote and reward innovation.
- Promote stewardship so antibiotics are sustainable over generations.
- Promote access.
- Provide public funding leveraged by private payments.
- Provide developers with rapid access to funds upon market entry.
- Be sustainable and dependable over the entire drug development cycle.
- Support and align with broader shifts in payment models toward value and quality and away from volume and intensity.
Priority Antimicrobial Value and Entry Award Proposal
Daniel cautioned that the traditional volume-based, fee-for-service payment scheme for drugs does not support high-value, appropriate use of antibiotics. He noted that the U.S. health care system is transitioning away from this model to place more emphasis on value and quality in care delivery, which creates new opportunities for how antibacterial drugs are reimbursed. To capitalize on this opportunity, the Duke-Margolis Center for Health Policy has developed the Priority Antimicrobial Value and Entry (PAVE) Award proposal, which combines market entry reward with a shift to value-based reimbursement for antimicrobials to incentivize innovation. In the proposal, he explained, market entry reward payments phase down over time and continued eligibility for payment requires adherence to a set of conditions: reliable availability of the drug; continued sensitivity of priority organisms; tracking and reporting appropriate prescribing; data collection for post-market studies, if needed; and shifting to alternative payment models that are not volume based. He explained that the PAVE Award model resolves the current conflict between the drivers of return on investment and strong stewardship programs by leveraging private financing to reinforce the “volume to value” shift.
Figure 5-5 describes how the PAVE Award would be phased in, said Daniel. The top figure represents the first 5 years a drug is on the market, he explained. The publicly funded market entry reward (blue) starts off very high as the source of most of the company’s revenue, he said, but it phases down drastically in subsequent years and is contingent on the company demonstrating that it is increasing its revenue with alternative payment models from the private side. By year 5, he added, the company is expected to have 75 to 80 percent of its revenue coming from the value-based payments, with full delinkage in year 6. He added that all of the revenue going back to that company is borne by the private sector and is dependent on population sizes that are covered by these payers—not dependent on how often patients are using the drug—so the company has a strong incentive to maintain a healthy supply chain.
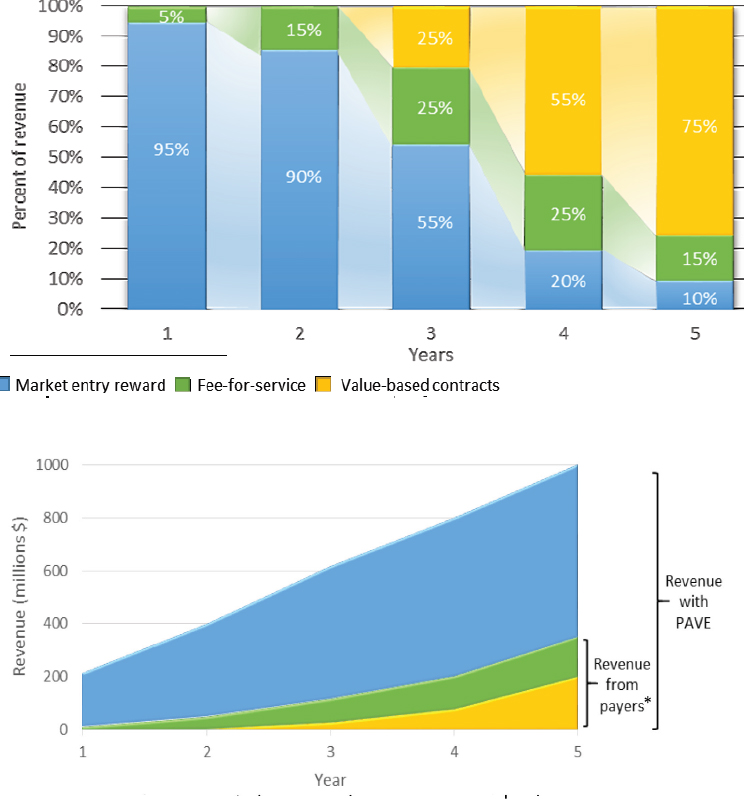
Top: The distribution of the company’s revenue includes a high percentage from the publicly funded market entry reward at the start but decreases subsequently over the 5 years a drug is on the market.
Bottom: The cumulative revenue for new antimicrobials would increase with PAVE, but the level of revenue from payers is the same as under the current fee-for-service model. * Level of revenue from payers is the same under current fee-for-service model and new PAVE model.
SOURCES: Daniel presentation, June 21, 2017; Duke-Margolis Center for Health Policy, 2017. Reprinted with permission from Duke University, Copyright (2017).
DISCUSSION
Jeffrey Duchin, health officer and chief of Communicable Disease Epidemiology and Immunization Section for Public Health in Seattle and King County, Washington, asked about the relationship between the mechanism of action of the pneumococcal vaccine and the mechanism of action of resistance, as well as the implications of using vaccines against antimicrobial drug-resistant pathogens in the future. Klugman replied that the ecology of resistance is located within the nasopharynx; the duration of time that invasive strains spend in the nasopharynx correlates with their exposure to antibiotics and their likelihood of acquiring resistance genes. He added that there are now strains that are less adapted to the nasopharynx and because there is less competition, they can colonize and acquire resistance. Fortunately, he said, those strains appear to be less invasive than the original strains, but there is also an increasing population of individuals who are able to get invasive pneumococcal disease because of immune issues, such as diabetes. However, he noted that in the United States today, there is generally much less antibiotic use, fewer antibiotic-resistant strains, and the total number of infections is far below baseline. On the other hand, in the United Kingdom, he reported, the number of nonvaccine strains is expanding rapidly and acquiring resistance, particularly in adults, for reasons that are unclear. David Relman, professor of medicine at Stanford University, asked what it takes to preempt or displace a resistant organism from one of those ecological niches. Klugman replied that the low-hanging fruit is to develop a vaccine that protects against invasive disease, but the ultimate goal would be to interrupt transmission. He added that the notions are complementary within the context of changing the complex environment of the microbiome through multiple strategies.
Caroline Harwood, professor of microbiology at University of Washington, asked McDonald to elaborate about phage therapy and in what setting it would be most effective. McDonald responded that there is natural resistance, although “cocktails” against a variety of strains in biofilm applications seem most promising, both environmentally and for patients. He noted that strains will need to be shifted, because the strains that transmit the best are the ones that tend to pick up the most resistance. The aim is to target the ones that transmit the best or are most virulent, he explained. He suggested that this may lead to targeting against colonization in cases where it is known that something less virulent and less resistant will take its place (which is true with Streptococcus pneumococcus). The worry had been that something like methicillin-resistant Staphylococcus aureus might occupy the niche in the nasopharynx after the decolonization, he continued, but that does not seem to be the case. He suggested that in the future, vaccines, tools like phage, and the microbiome will work in tandem to eliminate the most
virulent and resistant strains. He said there is also potential to manipulate phage to render antibiotic-resistant strains ecologically disadvantaged.
Emily Erbelding, deputy director of the Division of AIDS at the National Institutes of Health, asked Baron if specimen repositories would be useful for diagnostic companies to help in getting tests cleared for additional indications, such as gonorrhea and chlamydia from the oropharynx and the rectum. Baron responded that access to more clinical samples would be extremely helpful for manufacturing approval testing. She explained that they currently create artificial matrices to simulate clinical samples, because acquiring the number of real samples needed from commercial providers is cost prohibitive. Jesse Goodman, professor of medicine and infectious diseases at Georgetown University, suggested that a promising target is the tremendous need for new drugs for gonorrhea.
Rima Khabbaz, deputy director for infectious diseases at CDC, asked about incentives for modalities other than new antibiotics, such as vaccines, diagnostics, phages, and manipulating the microbiome. Daniel said that incentives are definitely needed, but the delinkage model will not necessarily work for modalities like diagnostics. He explained that the fundamental challenge with antibiotics is the need to use less of them, whereas the use of diagnostics needs to be increased. As the health care system shifts toward value and outcomes for patients, he said, it will incentivize development of diagnostics and vaccines given their fundamental effect on quality for patients. Daniel suggested focusing on the unique economic consequences and post-market issues that drive the use of those products.
McDonald wondered whether there is potential for pharmaceutical companies to branch out beyond drug development and adopt an integrated approach to managing the overall health of animals or the health of humans, such as by acquiring diagnostic technology expertise, information technology expertise, and other proprietary knowledge. Daniel agreed that the combination of antibiotics, vaccines, and diagnostics makes sense. He cited Medtronic as an example of a company that is shifting from being a medical device manufacturer to a services provider and is currently operating surgical suites in hospitals to provide end-to-end care for patients. Daniel noted that the regulatory approach with companion diagnostics is starting to address this, but a challenge is that the diagnostic itself is literally tied to a particular drug. Baron added that commercial companies are working on companion diagnostics with drug manufacturers or vaccine manufacturers (for example, on the Ebola vaccine). However, she cautioned that having a coproduct limits the use and does not provide much financial incentive. Baron warned that because policies have not changed to keep up with technology, billions of dollars are being wasted in the United States alone. For example, she said, rapid TB tests can pull TB-negative patients
out of airborne isolation faster, but jurisdictions still follow very old rules that require three negative smears to pull patients out of airborne isolation.
Kumanan Rasanathan, chief of the Implementation Research and Delivery Science Unit at the United Nations Children’s Fund, asked about evidence comparing vaccinations in low-income countries with poor access to higher-income countries with good access. Klugman replied that PCV appears to interrupt transmission, so it is well suited to developing countries with poor access and coverage. The percentage of coverage required to interrupt transmission is likely to be between 50 and 70 percent, he estimated, so even in areas without access, half of the community will be covered. Klugman argued that vaccine prevention should be introduced before antibiotics:
If a kid has three or four episodes of life-threatening pneumonia in the first year of life and doesn’t get antibiotics for all three of them, that’s it. [However], if they get vaccines very early on, it could have saved their lives.
He added that vaccination programs are vertical systems that are easier to establish in-country than programs to increase antibiotic exposure. Rasanathan followed up by asking if the effect on antimicrobial resistance has been incorporated into the investment case for vaccines. Not currently, replied Klugman, because investment decisions are being related to the disease burden and mortality prevention, but the BMGF intends to add the effect on antimicrobial resistance to make the value proposition for vaccines even more attractive.
Rasanathan asked Baron whether advance market commitments for diagnostics should be delinked in emergency situations of immediate need or if there should be other mechanisms in place. Baron responded that the current system both is and is not working. For example, there is an emergency use authorization quick route through FDA for a crisis-situation drug or diagnostic, for very specific utilizations during that emergency, which goes away after a certain number of years. But that in itself is not a sufficient incentive to develop a diagnostic because after the emergency authorization ends, Baron explained, the diagnostic must go through the normal regulatory process. Goodman cautioned that if the approval process is too lax, it could affect public health as well as the industry, because it disincentivizes the quality players to make good products. He considers the diagnostic approval process in the United States to be relatively well balanced. Goodman continued by observing that the transition to value-based health care could offer opportunities around drug pricing, as it has happened in oncology where you pay sufficiently for a drug that you only use when you truly need it. The pricing of the drug can serve as an important disincentive to inappropriate use of antibiotics.
This page intentionally left blank.




















