1
Introduction
When the Institute of Medicine (IOM)1 issued its 1975 report on the public health impact of legalized abortion, the scientific evidence on the safety and health effects of legal abortion services was limited (IOM, 1975). It had been only 2 years since the landmark Roe v. Wade decision had legalized abortion throughout the United States and nationwide data collection was just under way (Cates et al., 2000; Kahn et al., 1971). Today, the available scientific evidence on abortion’s health effects is quite robust.
In 2016, six private foundations came together to ask the Health and Medicine Division of the National Academies of Sciences, Engineering, and Medicine to conduct a comprehensive review of the state of the science on the safety and quality of legal abortion services in the United States. The sponsors—The David and Lucile Packard Foundation, The Grove Foundation, The JPB Foundation, The Susan Thompson Buffett Foundation, Tara Health Foundation, and William and Flora Hewlett Foundation—asked that the review focus on the eight research questions listed in Box 1-1.
The Committee on Reproductive Health Services: Assessing the Safety and Quality of Abortion Care in the U.S. was appointed in December 2016 to conduct the study and prepare this report. The committee included 13 individuals2 with research or clinical experience in anesthesiology,
___________________
1 In March 2016, the IOM, the division of the National Academies of Sciences, Engineering, and Medicine focused on health and medicine, was renamed the Health and Medicine Division.
2 A 14th committee member participated for just the first 4 months of the study.
obstetrics and gynecology, nursing and midwifery, primary care, epidemiology of reproductive health, mental health, health care disparities, health care delivery and management, health law, health professional education and training, public health, quality assurance and assessment,
statistics and research methods, and women’s health policy. Brief biographies of committee members are provided in Appendix A.
This chapter describes the context for the study and the scope of the inquiry. It also presents the committee’s conceptual framework for conducting its review.
ABORTION CARE TODAY
Since the IOM first reviewed the health implications of national legalized abortion in 1975, there has been a plethora of related scientific research, including well-designed randomized controlled trials (RCTs), systematic reviews, and epidemiological studies examining abortion care. This research has focused on examining the relative safety of abortion methods and the appropriateness of methods for different clinical circumstances (Ashok et al., 2004; Autry et al., 2002; Bartlett et al., 2004; Borgatta, 2011; Borkowski et al., 2015; Bryant et al., 2011; Cates et al., 1982; Chen and Creinin, 2015; Cleland et al., 2013; Frick et al., 2010; Gary and Harrison, 2006; Grimes et al., 2004; Grossman et al., 2008, 2011; Ireland et al., 2015; Kelly et al., 2010; Kulier et al., 2011; Lohr et al., 2008; Low et al., 2012; Mauelshagen et al., 2009; Ngoc et al., 2011; Ohannessian et al., 2016; Peterson et al., 1983; Raymond et al., 2013; Roblin, 2014; Sonalkar et al., 2017; Upadhyay et al., 2015; White et al., 2015; Wildschut et al., 2011; Woodcock, 2016; Zane et al., 2015). With this growing body of research, earlier abortion methods have been refined, discontinued, and new approaches have been developed (Chen and Creinin, 2015; Jatlaoui et al., 2016; Lichtenberg and Paul, 2013). For example, the use of dilation and sharp curettage is now considered obsolete in most cases because safer alternatives, such as aspiration methods, have been developed (Edelman et al, 1974; Lean et al, 1976; RCOG, 2015). The use of abortion medications in the United States began in 2000 with the approval by the U.S. Food and Drug Administration (FDA) of the drug mifepristone. In 2016, the FDA, citing extensive clinical research, updated the indications for mifepristone for medication abortion3 up to 10 weeks’ (70 days’) gestation (FDA, 2016; Woodcock, 2016).
Box 1-2 describes the abortion methods currently recommended by U.S. and international medical, nursing, and other health organizations that set professional standards for reproductive health care, including the American College of Obstetricians and Gynecologists (ACOG), the Society of Family Planning, the American College of Nurse-Midwifes, the National Abortion Federation (NAF), the Royal College of Obstetricians and Gynaecologists (RCOG) (in the United Kingdom), and the World
___________________
3 The terms “medication abortion” and “medical abortion” are used interchangeably in the literature.
Health Organization (ACNM, 2011, 2016; ACOG, 2013, 2014; Costescu et al., 2016; Lichtenberg and Paul, 2013; NAF, 2017; RCOG, 2011; WHO, 2014).
A Continuum of Care
The committee views abortion care as a continuum of services, as illustrated in Figure 1-1. For purposes of this study, it begins when a woman, who has decided to terminate a pregnancy, contacts or visits a provider seeking an abortion. The first, preabortion phase of care includes an initial clinical assessment of the woman’s overall health (e.g., physical examination, pregnancy determination, weeks of gestation, and laboratory and other testing as needed); communication of information on the risks and benefits of alternative abortion procedures and pain management options; discussion of the patient’s preferences based on desired anesthesia and weeks of gestation; discussion of postabortion contraceptive options if desired; counseling

and referral to services (if needed); and final decision making and informed consent. The next phases in the continuum are the abortion procedure itself and postabortion care, including appropriate follow-up care and provision of contraceptives (for women who opt for them).
A Note on Terminology
Important clinical terms that describe pregnancy and abortion lack consistent definition. The committee tried to be as precise as possible to avoid misinterpreting or miscommunicating the research evidence, clinical practice guidelines, and other relevant sources of information with potentially significant clinical implications. Note that this report follows Grimes and Stuart’s (2010) recommendation that weeks’ gestation be quantified using cardinal numbers (1, 2, 3...) rather than ordinal numbers (1st, 2nd, 3rd...). It is important to note, however, that these two numbering conventions are sometimes used interchangeably in the research literature despite having different meanings. For example, a woman who is 6 weeks pregnant has completed 6 weeks of pregnancy: she is in her 7th (not 6th) week of pregnancy.
This report also avoids using the term “trimester” where possible because completed weeks’ or days’ gestation is a more precise designation, and the clinical appropriateness of abortion methods does not align with specific trimesters.
Although the literature typically classifies the method of abortion as either “medical” or “surgical” abortion, the committee decided to specify methods more precisely by using the terminology defined in Box 1-2. The term “surgical abortion” is often used by others as a catchall category that includes a variety of procedures, ranging from an aspiration to a dilation and evacuation (D&E) procedure involving sharp surgical and other instrumentation as well as deeper levels of sedation. This report avoids describing abortion procedures as “surgical” so as to characterize a method more accurately as either an aspiration or D&E. As noted in Box 1-2, the term “induction abortion” is used to distinguish later abortions that use a
medication regimen from medication abortions performed before 10 weeks’ gestation.
See Appendix B for a glossary of the technical terms used in this report.
Regulation of Abortion Services
Abortion is among the most regulated medical procedures in the nation (Jones et al., 2010; Nash et al., 2017). While a comprehensive legal analysis of abortion regulation is beyond the scope of this report, the committee agreed that it should consider how abortion’s unique regulatory environment relates to the safety and quality of abortion care.
In addition to the federal, state, and local rules and policies governing all medical services, numerous abortion-specific federal4 and state laws and regulations affect the delivery of abortion services. Table 1-1 lists the abortion-specific regulations by state. The regulations range from prescribing information to be provided to women when they are counseled and setting mandatory waiting periods between counseling and the abortion procedure to those that define the clinical qualifications of abortion providers, the types of procedures they are permitted to perform, and detailed facility standards for abortion services. In addition, many states place limitations on the circumstances under which private health insurance and Medicaid can be used to pay for abortions, limiting coverage to pregnancies resulting from rape or incest or posing a medical threat to the pregnant woman’s life. Other policies prevent facilities that receive state funds from providing abortion services5 or place restrictions on the availability of services based on the gestation of the fetus that are narrower than those established under federal law (Guttmacher Institute, 2017h).
Trends and Demographics
National- and state-level abortion statistics come from two primary sources: the Centers for Disease Control and Prevention’s (CDC’s) Abortion
___________________
4 Hyde Amendment (P.L. 94-439, 1976); Department of Defense Appropriations Act (P.L. 95-457, 1978); Peace Corps Provision and Foreign Assistance and Related Programs Appropriations Act (P.L. 95-481, 1978); Pregnancy Discrimination Act (P.L. 95-555, 1977); Department of the Treasury and Postal Service Appropriations Act (P.L. 98-151, 1983); FY1987 Continuing Resolution (P.L. 99-591, 1986); Dornan Amendment (P.L. 100-462, 1988); Partial-Birth Abortion Ban (P.L. 108-105, 2003); Weldon Amendment (P.L. 108-199, 2004); Patient Protection and Affordable Care Act (P.L. 111-148 as amended by P.L. 111-152, 2010).
5 Personal communication, O. Cappello, Guttmacher Institute, August 4, 2017: AZ § 15-1630, GA § 20-2-773; KS § 65-6733 and § 76-3308; KY § 311.800; LA RS § 40:1299 and RS § 4 0.1061; MO § 188.210 and § 188.215; MS § 41-41-91; ND § 14-02.3-04; OH § 5101.57; OK 63 § 1-741.1; PA 18 § 3215; TX § 285.202.
| Type of Regulationa | States | Number of States |
|---|---|---|
| An ultrasound must be performed before all abortions, regardless of method | AL, AZ, FL, IA, IN, KS, LA, MS, NC, OH, OK, TX, VA, WI | 14 |
| Clinicians providing medication abortions must be in the physical presence of the patient when she takes the medication | AL, AR, AZ, IN, KS, LA, MI, MO, MS, NC, ND, NE, OK, SC, SD, TN, TX, WI, WV | 19 |
| Women must receive counseling before an abortion is performed | AL, AK, AR, AZ, CA,b CT,b FL, GA, IA, ID, IN, KS, KY, LA, ME,b MI, MN, MO, MS, NC, ND, NE, NV,b OH, OK, PA, RI,b SC, SD, TN, TX, UT, VA, WI, WV | 35 |
| Abortion patients are offered or given inaccurate or misleading information (verbally or in writing) onc | ||
|
AR, SD, UT | 3 |
|
AZ, KS, NC, NE, SD, TX | 6 |
|
AK, KS, MS, OK, TX | 5 |
|
ID, KS, LA, MI, NC, ND, NE, OK, SD, TX, UT, WV | 12 |
| All methods of abortion are subject to a mandatory waiting period between counseling and procedure | ||
|
IN | 1 |
|
AZ, GA, ID, KS, KY, MI, MN, MS, ND, NE, OH, PA, SC, TX, VA, WI, WV | 17 |
|
AL, AR, TN | 3 |
|
MO, NC, OK, SD, UT | 5 |
| Preabortion counseling must be in person, necessitating two visits to the facility | AR, AZ, IN, KY, LA, MO, MS, OH, SD, TN, TX,d UT,e VA, WI | 14 |
| Type of Regulationa | States | Number of States |
|---|---|---|
| All abortions, regardless of method, must be performed by a licensed physician | AL, AK, AR, AZ, DE, FL, GA, IA, ID, IN, KS, KY, LA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NV, OH, OK, PA, SC, SD, TN, TX, UT, VA, WI, WY | 34 |
| Clinicians performing any type of abortion procedures must have hospital admitting privileges or an agreement with a local hospital to transfer patients if needed | AL, AZ, IN, LA, MS, ND, OK, SC, TX, UT | 10 |
| Abortion facilities must have an agreement with a local hospital to transfer patients if needed | FL, KY, MI, NC, OH, PA, TN, WI | 8 |
| All abortions, regardless of method, must be performed in a facility that meets the structural standards typical of ambulatory surgical centers | AL, AR, AZ, IN, KY, LA, MI, MO, MS, NC, OH, OK, PA, RI, SC, SD, UT | 17 |
| Procedure room size, corridor width, or maximum distance to a hospital is specified | AL, AR, AZ,f FL, IN, LA, MI, MS, ND, NE, OH, OK, PA, SC, SD, UT | 16 |
| Public funding of abortions is limited to pregnancies resulting from rape or incest or when the woman’s life is endangeredg | AL, AR, CO, DC, DE, FL, GA, IA, ID, IN, KS, KY, LA, ME, MI, MO, MS, NC, ND, NE, NH, NV, OH, OK, PA, RI, SC, SD, TN, TX, UT, VA, WI, WY | 34 |
| Insurance coverage of abortion is restricted in all private insurance plans written in the state, including those offered through health insurance exchanges established under the federal health care reform lawh | ID, IN, KS, KY, MI, MO, ND, NE, OK, TX, UT | 11 |
| Insurance coverage of abortion is restricted in plans offered through a health insurance exchangeh | AL, AR, AZ, FL, GA, ID, IN, KS, KY, LA, MI, MO, MS, NC, ND, NE, OH, OK, PA, SC, SD, TN, TX, UT, VA, WI | 26 |
| Type of Regulationa | States | Number of States |
|---|---|---|
| No abortions may be performed after a specified number of weeks’ gestation unless the woman’s life or health is endangered | ||
|
AL, AR, GA, IA, IN, KS, KY, LA, MS, NC, ND, NE, OH, OK, SC, SD, TX, WI, WV | 19 |
|
FL, MA, NV, NY, PA, RI, VA | 7 |
| Dilation and evacuation (D&E) abortions are banned except in cases of life endangerment or severe physical health risk | MS, WV | 2 |
| Abortions cannot be performed in publicly funded facilities | AZ, GA, KS, KY, LA, MO, MS, ND, OH, OK, PA, TX | 12 |
a Excludes laws or regulations permanently or temporarily enjoined pending a court decision.
b States have abortion-specific requirements generally following the established principles of informed consent.
c The content of informed consent materials is specified in state law or developed by the state department of health.
d In-person counseling is not required for women who live more than 100 miles from an abortion provider.
e Counseling requirement is waived if the pregnancy is the result of rape or incest or the patient is younger than 15.
f Maximum distance requirement does not apply to medication abortions.
g Some states also exempt women whose physical health is at severe risk and/or in cases of fetal impairment.
h Some states have exceptions for pregnancies resulting from rape or incest, pregnancies that severely threaten women’s physical health or endanger their life, and/or in cases of fetal impairment.
Surveillance System and the Guttmacher Institute’s Abortion Provider Census (Jatlaoui et al., 2016; Jerman et al., 2016; Jones and Kavanaugh, 2011; Pazol et al., 2015). Both of these sources provide estimates of the number and rate of abortions, the use of different abortion methods, the characteristics of women who have abortions, and other related statistics. However, both sources have limitations.
The CDC system is a voluntary, state-reported system;6,7 three states (California, Maryland, and New Hampshire) do not provide information (CDC, 2017). The Guttmacher census, also voluntary, solicits information from all known abortion providers throughout the United States, including in the states that do not submit information to the CDC surveillance system. For 2014, the latest year reported by Guttmacher,8 information was obtained directly from 58 percent of abortion providers, and data for nonrespondents were imputed (Jones and Jerman, 2017a). The CDC’s latest report, for abortions in 2013, includes approximately 70 percent of the abortions reported by the Guttmacher Institute for that year (Jatlaoui et al., 2016).
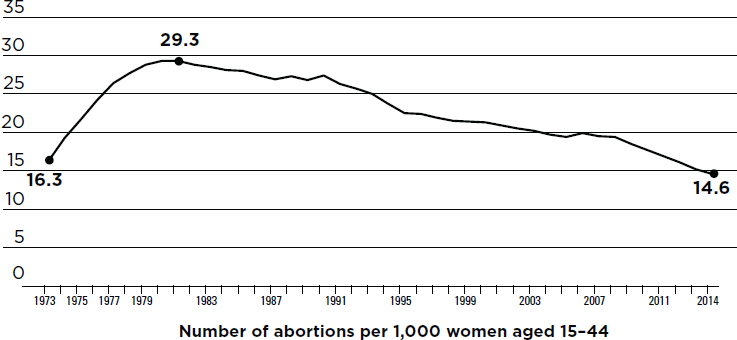
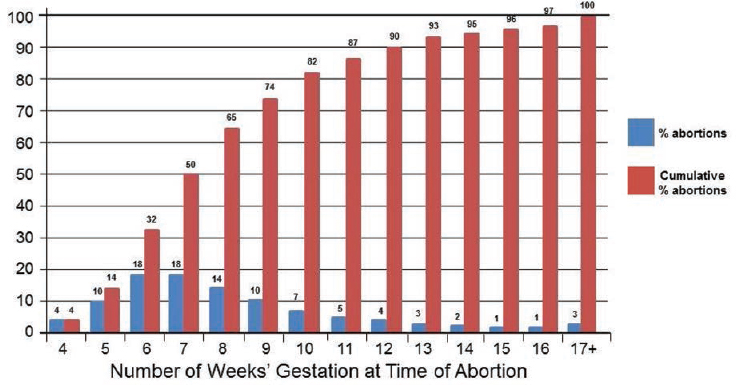
Both data collection systems report descriptive statistics on women who have abortions and the types of abortion provided, although they define demographic variables and procedure types differently. Nevertheless, in the aggregate, the trends in abortion utilization reported by the CDC and Guttmacher closely mirror each other—indicating decreasing rates of abortion, an increasing proportion of medication abortions, and the vast majority of abortions (90 percent) occurring by 13 weeks’ gestation (see Figures 1-2 and 1-3) (Jatlaoui et al., 2016; Jones and Jerman, 2017a).9 Both data sources are used in this chapter’s brief review of trends in abortions and throughout the report.
Trends in the Number and Rate of Abortions
The number and rate of abortions have changed considerably during the decades following national legalization in 1973. In the immediate years after
___________________
6 In most states, hospitals, facilities, and physicians are required by law to report abortion data to a central health agency. These agencies submit the aggregate utilization data to the CDC (Guttmacher Institute, 2018a).
7 New York City and the District of Columbia also report data to the CDC.
8 Guttmacher researchers estimate that the census undercounts the number of abortions performed in the United States by about 5 percent (i.e., 51,725 abortions provided by 2,069 obstetrician/gynecologist [OB/GYN] physicians). The estimate is based on a survey of a random sample of OB/GYN physicians. The survey did not include other physician specialties and other types of clinicians.
9 A full-term pregnancy is 40 weeks.
SOURCE: Guttmacher Institute, 2017a. Used with permission.
NOTE: n = 8,105.
SOURCE: Adapted from Jones and Jerman, 2017b.
national legalization, both the number and rate10 of legal abortions steadily increased (Bracken et al., 1982; Guttmacher Institute, 2017a; Pazol et al., 2015; Strauss et al., 2007) (see Figure 1-2). The abortion rate peaked in the
___________________
10 Reported abortion rates are for females aged 15 to 44.
1980s, and the trend then reversed, a decline that has continued for more than three decades (Guttmacher Institute, 2017a; Jones and Kavanaugh, 2011; Pazol et al., 2015; Strauss et al., 2007). Between 1980 and 2014, the abortion rate among U.S. women fell by more than half, from 29.3 to 14.6 per 1,000 women (Finer and Henshaw, 2003; Guttmacher Institute, 2017a; Jones and Jerman, 2017a) (see Figure 1-2). In 2014, the most recent year for which data are available, the aggregate number of abortions reached a low of 926,190 after peaking at nearly 1.6 million in 1990 (Finer and Henshaw, 2003; Jones and Jerman, 2017a). The reason for the decline is not fully understood but has been attributed to several factors, including the increasing use of contraceptives, especially long-acting methods (e.g., intrauterine devices and implants); historic declines in the rate of unintended pregnancy; and increasing numbers of state regulations resulting in limited access to abortion services (Finer and Zolna, 2016; Jerman et al., 2017; Jones and Jerman, 2017a; Kost, 2015; Strauss et al., 2007).
Weeks’ Gestation
Length of gestation—measured as the amount of time since the first day of the last menstrual period—is the primary factor in deciding what abortion procedure is most appropriate (ACOG, 2014). Since national legalization, most abortions in the United States have been performed in early pregnancy (≤13 weeks) (Cates et al., 2000; CDC, 1983; Elam-Evans et al., 2003; Jatlaoui et al., 2016; Jones and Jerman, 2017a; Koonin and Smith, 1993; Lawson et al., 1989; Pazol et al., 2015; Strauss et al., 2007). CDC surveillance reports indicate that since at least 1992 (when detailed data on early abortions were first collected), the vast majority of abortions in the United States were early-gestation procedures (Jatlaoui et al., 2016; Strauss et al., 2007); this was the case for approximately 92 percent of all abortions in 2013 (Jatlaoui et al., 2016). With such technological advances as highly sensitive pregnancy tests and medication abortion, procedures are being performed at increasingly earlier gestational stages. According to the CDC, the percentage of early abortions performed ≤6 weeks’ gestation increased by 16 percent from 2004 to 2013 (Jatlaoui et al., 2016); in 2013, 38 percent of early abortions occurred ≤6 weeks (Jatlaoui et al., 2016). The proportion of early-gestation abortions occurring ≤6 weeks is expected to increase even further as the use of medication abortions becomes more widespread (Jones and Boonstra, 2016; Pazol et al., 2012).
Figure 1-3 shows the proportion of abortions in nonhospital settings by weeks’ gestation in 2014 (Jones and Jerman, 2017a).
Abortion Methods
Aspiration is the abortion method most commonly used in the United States, accounting for almost 68 percent of all abortions performed in 2013 (Jatlaoui et al., 2016).11 Its use, however, is likely to decline as the use of medication abortion increases. The percentage of abortions performed by the medication method rose an estimated 110 percent between 2004 and 2013, from 10.6 to 22.3 percent (Jatlaoui et al., 2016). In 2014, approximately 45 percent of abortions performed up to 9 weeks’ gestation were medication abortions, up from 36 percent in 2011 (Jones and Jerman, 2017a).
Fewer than 9 percent of abortions are performed after 13 weeks’ gestation; most of these are D&E procedures (Jatlaoui et al., 2016). Induction abortion is the most infrequently used of all abortion methods, accounting for approximately 2 percent of all abortions at 14 weeks’ gestation or later in 2013 (Jatlaoui et al., 2016).
Characteristics of Women Who Have Abortions
The most detailed sociodemographic statistics on women who have had an abortion in the United States are provided by the Guttmacher Institute’s Abortion Patient Survey. Respondents to the 2014/2015 survey included more than 8,000 women who had had an abortion in 1 of 87 outpatient (nonhospital) facilities across the United States in 2014 (Jerman et al., 2016; Jones and Jerman, 2017b).12Table 1-2 provides selected findings from this survey. Although women who had an abortion in a hospital setting are excluded from these statistics, the data represent an estimated 95 percent of all abortions provided (see Figure 1-3).
The Guttmacher survey found that most women who had had an abortion were under age 30 (72 percent) and were unmarried (86 percent) (Jones and Jerman, 2017b). Women seeking an abortion were far more likely to be poor or low-income: the household income of 49 percent was below the federal poverty level (FPL), and that of 26 percent was 100 to 199 percent of the FPL (Jerman et al., 2016). In comparison, the
___________________
11 CDC surveillance reports use the catchall category of “curettage” to refer to nonmedical abortion methods. The committee assumed that the CDC’s curettage estimates before 13 weeks’ gestation refer to aspiration procedures and that its curettage estimates after 13 weeks’ gestation referred to D&E procedures.
12 Participating facilities were randomly selected and excluded hospitals. All other types of facilities were included if they had provided at least 30 abortions in 2011 (Jerman et al., 2016). Jerman and colleagues report that logistical challenges precluded including hospital patients in the survey. The researchers believe that the exclusion of hospitals did not bias the survey sample, noting that hospitals accounted for only 4 percent of all abortions in 2011.
TABLE 1-2 Characteristics of Women Who Had an Abortion in an Outpatient Setting in 2014, by Percent
| Characteristic | Percent |
|---|---|
| Age (a) | |
| <15–17 | 3.6 |
| 18–19 | 8.2 |
| 20–24 | 33.6 |
| 25–29 | 26.3 |
| 30–34 | 16.0 |
| 35+ | 12.2 |
| Race/Ethnicity (a) | |
| Asian/Pacific Islander | 4.7 |
| Black | 24.8 |
| Hispanic | 24.5 |
| Multiracial | 4.5 |
| Other | 2.5 |
| White | 39.0 |
| Prior Pregnancies (a) | |
| No prior pregnancies | 29.2 |
| Prior birth only | 26.0 |
| Prior abortion only | 11.7 |
| Prior birth and abortion | 33.1 |
| Prior Births (b) | |
| None | 40.7 |
| 1 | 26.2 |
| 2+ | 33.1 |
| Education (a) | |
| Not a high school graduate | 12.2 |
| High school graduate or GED | 29.0 |
| Some college or associates degree | 39.2 |
| College graduate | 19.7 |
| Family Income as a Percentage of Federal Poverty Level (b) | |
| <100 | 49.3 |
| 100–199 | 25.7 |
| ≥200 | 25.0 |
| Payment Method (a) | |
| Private insurance | 14.1 |
| Medicaid | 21.9 |
| Financial assistance | 13.2 |
| Out of pocket | 45.4 |
| Other/unknown | 5.4 |
NOTE: Percentages may not sum to 100 because of rounding.
SOURCES: (a) Jones and Jerman, 2017b (n = 8,098); (b) Jerman et al., 2016 (n = 8,380).
corresponding percentages among all women aged 15 to 49 are 16 and 18 percent.13 Women who had had an abortion were also more likely to be women of color14 (61.0 percent); overall, half of women who had had an abortion were either black (24.8 percent) or Hispanic (24.5 percent) (Jones and Jerman, 2017b). This distribution is similar to the racial and ethnic distribution of women with household income below 200 percent of the FPL, 49 percent of whom are either black (20 percent) or Hispanic (29 percent).15 Poor women and women of color are also more likely than others to experience an unintended pregnancy (Finer and Henshaw, 2006; Finer et al., 2006; Jones and Kavanaugh, 2011).
Many women who have an abortion have previously experienced pregnancy or childbirth. Among respondents to the Guttmacher survey, 59.3 percent had given birth at least once, and 44.8 percent had had a prior abortion (Jerman et al., 2016; Jones and Jerman, 2017b).
While precise estimates of health insurance coverage of abortion are not available, numerous regulations limit coverage. As noted in Table 1-1, 33 states prohibit public payers from paying for abortions and other states have laws that either prohibit health insurance exchange plans (25 states) or private insurance plans (11 states) sold in the state from covering or paying for abortions, with few exceptions.16 In the Guttmacher survey, only 14 percent of respondents had paid for the procedure using private insurance coverage, and despite the disproportionately high rate of poverty and low income among those who had had an abortion, only 22 percent reported that Medicaid was the method of payment for their abortion. In 2015, 39 percent of the 25 million women lived in households that earned less than 200 percent of the FPL in the United States were enrolled in Medicaid, and 36 percent had private insurance (Ranji et al., 2017).
Number of Clinics Providing Abortion Care
As noted earlier, the vast majority of abortions are performed in nonhospital settings—either an abortion clinic (59 percent) or a clinic offering a variety of medical services (36 percent) (Jones and Jerman, 2017a) (see Figure 1-4). Although hospitals account for almost 40 percent of facilities offering abortion care, they provide less than 5 percent of abortions overall.
___________________
13 Calculation by the committee based on estimates from Annual Social and Economic Supplement (ASEC) to the Current Population Survey (CPS).
14 Includes all nonwhite race and ethnicity categories in Table 1-2. Data were collected via self-administered questionnaire (Jones and Jerman, 2017b).
15 Calculation by the committee based on estimates from Annual Social and Economic Supplement (ASEC) to the Current Population Survey (CPS).
16 Some states have exceptions for pregnancies resulting from rape or incest, pregnancies that endanger the woman’s life or severely threaten her health, and in cases of fetal impairment.
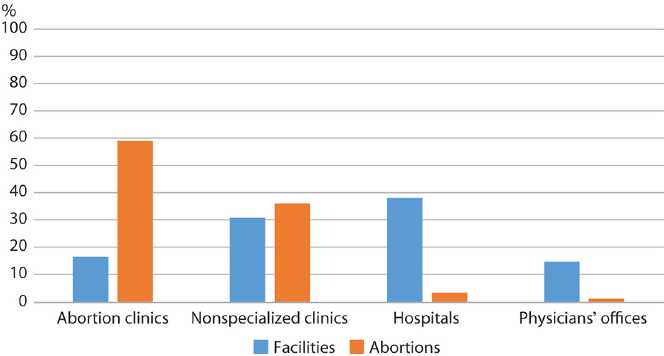
NOTE: Abortion clinics are nonhospital facilities in which 50 percent or more of patient visits are for abortion services. Nonspecialized clinics are nonhospital facilities in which fewer than 50 percent of patient visits are for abortion services.
SOURCE: Jones and Jerman, 2017a.
The overall number of nonhospital facilities providing abortions—especially specialty abortion clinics—is declining. The greatest proportional decline is in states that have enacted abortion-specific regulations (Jones and Jerman, 2017a). In 2014, there were 272 abortion clinics in the United States, 17 percent fewer than in 2011. The greatest decline (26 percent) was among large clinics with annual caseloads of 1,000–4,999 patients and clinics in the Midwest (22 percent) and the South (13 percent). In 2014, approximately 39 percent of U.S. women aged 15 to 44 resided in a U.S. county without an abortion provider (90 percent of counties overall) (Jones and Jerman, 2017a). Twenty-five states have five or fewer abortion clinics; five states have one abortion clinic (Jones and Jerman, 2017a). A recent analysis17 by Guttmacher evaluated geographic disparities in access to abortion by calculating the distance between women of reproductive age (15 to 44) and the nearest abortion-providing facility in 2014 (Bearak et al., 2017). Figure 1-5 highlights the median distance to the nearest facility by county.
___________________
17 The analysis was limited to facilities that provided at least 400 abortions per year and those affiliated with Planned Parenthood that performed at least 1 abortion during the period of analysis.
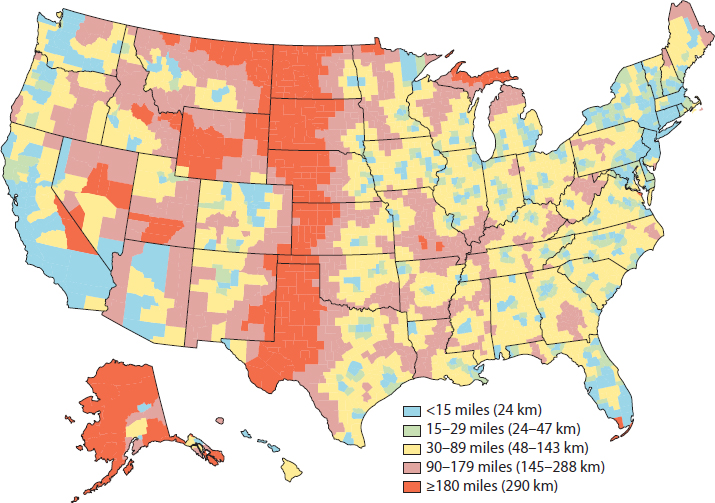
NOTE: Analysis is limited to facilities that had caseloads of 400 abortions or more per year and those affiliated with Planned Parenthood that performed at least 1 abortion in the period of analysis.
SOURCE: Bearak et al., 2017.
The majority of facilities offer early medication and aspiration abortions. In 2014, 87 percent of nonhospital facilities provided early medication abortions; 23 percent of all nonhospital facilities offered this type of abortion (Jones and Jerman, 2017a). Fewer facilities offer later-gestation procedures, and availability decreases as gestation increases. In 2012, 95 percent of all abortion facilities offered abortions at 8 weeks’ gestation, 72 percent at 12 weeks’ gestation, 34 percent at 20 weeks’ gestation, and 16 percent at 24 weeks’ gestation (Jerman and Jones, 2014).
STUDY APPROACH
Conceptual Framework
The committee’s approach to this study built on two foundational developments in the understanding and evaluation of the quality of health
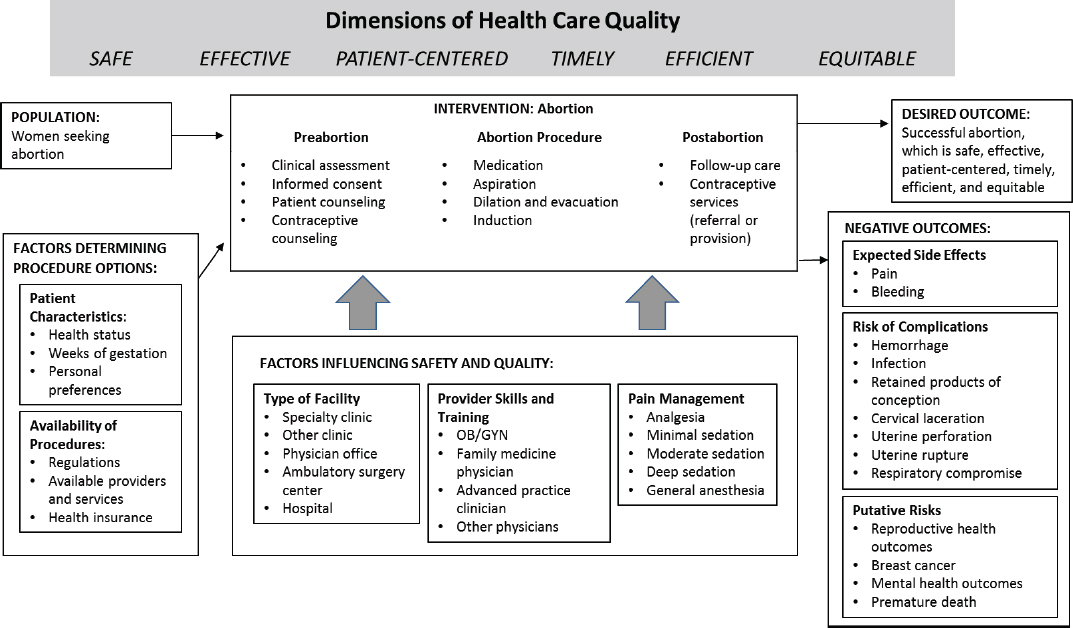
NOTE: OB/GYN = obstetrician/gynecologist.
care services: Donabedian’s (1980) structure-process-outcome framework and the IOM’s (2001) six dimensions of quality health care. Figure 1-6 illustrates the committee’s adaptation of these concepts for this study’s assessment of abortion care in the United States.
Structure-Process-Outcome Framework
In seminal work published almost 40 years ago, Donabedian (1980) proposed that the quality of health care be assessed by examining its structure, process, and outcomes (Donabedian, 1980):
- Structure refers to organizational factors that may create the potential for good quality. In abortion care, such structural factors as the availability of trained staff and the characteristics of the clinical setting may ensure—or inhibit—the capacity for quality.
- Process refers to what is done to and for the patient. Its assessment assumes that the services patients receive should be evidence based and correlated with patients’ desired outcomes—for example, an early and complete abortion for women who wish to terminate an unintended pregnancy.
- Outcomes are the end results of care—the effects of the intervention on the health and well-being of the patient. Does the procedure achieve its objective? Does it lead to serious health risks in the short or long term?
Six Dimensions of Health Care Quality
The landmark IOM report Crossing the Quality Chasm: A New Health System for the 21st Century (IOM, 2001) identifies six dimensions of health care quality—safety, effectiveness, patient-centeredness, timeliness, efficiency, and equity. The articulation of these six dimensions has guided public and private efforts to improve U.S. health care delivery at the local, state, and national levels since that report was published (AHRQ, 2016).
In addition, as with other health care services, women should expect that the abortion care they receive meets well-established standards for objectivity, transparency, and scientific rigor (IOM, 2011a,b).
Two of the IOM’s six dimensions—safety and effectiveness—are particularly salient to the present study. Assessing both involves making relative judgments. There are no universally agreed-upon thresholds for defining care as “safe” versus “unsafe” or “effective” versus “not effective,” and decisions about safety and effectiveness have a great deal to do with the context of the clinical scenario. Thus, the committee’s frame of reference for evaluating safety, effectiveness, and other quality domains is of necessity a
relative one—one that entails not only comparing the alternative abortion methods but also comparing these methods with other health care services and with risks associated with not achieving the desired outcome.
Safety—avoiding injury to patients—is often assessed by measuring the incidence and severity of complications and other adverse events associated with receiving a specific procedure. If infrequent, a complication may be characterized as “rare”—a term that lacks consistent definition. In this report, “rare” is used to describe outcomes that affect fewer than 1 percent of patients. Complications are considered “serious” if they result in a blood transfusion, surgery, or hospitalization.
Note also that the term “effectiveness” is used differently in this report depending on the context. As noted in Box 1-3, effectiveness as an attribute of quality refers to providing services based on scientific knowledge to all who could benefit and refraining from providing services to those not likely to benefit (avoiding underuse and overuse, respectively). Elsewhere in this report, effectiveness denotes the clinical effectiveness of a procedure, that
is, the successful completion of an abortion without the need for a follow-up aspiration.
Finding and Assessing the Evidence
The committee deliberated during four in-person meetings and numerous teleconferences between January 2017 and December 2017. On March 24, 2017, the committee hosted a public workshop at the Keck Center of the National Academies of Sciences, Engineering, and Medicine in Washington, DC. The workshop included presentations from three speakers on topics related to facility standards and the safety of outpatient procedures. Appendix C contains the workshop agenda.
Several committee workgroups were formed to find and assess the quality of the available evidence and to draft summary materials for the full committee’s review. The workgroups conducted in-depth reviews of the epidemiology of abortions, including rates of complications and mortality, the safety and effectiveness of alternative abortion methods, professional standards and methods for performing all aspects of abortion care (as described in Figure 1-1), the short- and long-term physical and mental health effects of having an abortion; and the safety and quality implications of abortion-specific regulations on abortion.
The committee focused on finding reliable, scientific information reflecting contemporary U.S. abortion practices. An extensive body of research on abortion has been conducted outside the United States. A substantial proportion of this literature concerns the delivery of abortion care in countries where socioeconomic conditions, culture, population health, health care resources, and/or the health care system are markedly different from their U.S. counterparts. Studies from other countries were excluded from this review if the committee judged those factors to be relevant to the health outcomes being assessed.
The committee considered evidence from randomized controlled trials comparing two or more approaches to abortion care; systematic reviews; meta-analyses; retrospective cohort studies, case control studies, and other types of observational studies; and patient and provider surveys (see Box 1-4).
An extensive literature documents the biases common in published research on the effectiveness of health care services (Altman et al., 2001; Glasziou et al., 2008; Hopewell et al., 2008; Ioannidis et al., 2004; IOM, 2011a,b; Plint et al., 2006; Sackett, 1979; von Elm et al., 2007). Thus, the committee prioritized the available research according to conventional principles of evidence-based medicine intended to reduce the risk of bias in a study’s conclusions, such as how subjects were allocated to different types of abortion care, the comparability of study populations, controls
for confounding factors, how outcome assessments were conducted, the completeness of outcome reporting, the representativeness of the study population compared with the general U.S. population, and the degree to which statistical analyses helped reduce bias (IOM, 2011b). Applying these principles is particularly important with respect to understanding abortion’s
long-term health effects, an area in which the relevant literature is vulnerable to bias (as discussed in Chapter 4).
The committee’s literature search strategy is described in Appendix D.
ORGANIZATION OF THE REPORT
Chapter 2 of this report describes the continuum of abortion care including current abortion methods (question 1 in the committee’s statement of task [Box 1-1]); reviews the evidence on factors affecting their safety and quality, including expected side effects and possible complications (questions 2 and 3), necessary safeguards to manage medical emergencies (question 6), and provision of pain management (question 7); and presents the evidence on the types of facilities or facility factors necessary to provide safe and effective abortion care (question 4).
Chapter 3 summarizes the clinical skills that are integral to safe and high-quality abortion care according to the recommendations of leading national professional organizations and abortion training curricula (question 5).
Chapter 4 reviews research examining the long-term health effects of undergoing an abortion (question 2).
Finally, Chapter 5 presents the committee’s conclusions regarding the findings presented in the previous chapters, responding to each of the questions posed in the statement of task. Findings are statements of scientific evidence. The report’s conclusions are the committee’s inferences, interpretations, or generalizations drawn from the evidence.
REFERENCES
ACNM (American College of Nurse-Midwives). 2011. Position statement: Reproductive health choices. http://www.midwife.org/ACNM/files/ACNMLibraryData/UPLOADFILENAME/000000000087/Reproductive_Choices.pdf (accessed August 1, 2017).
ACNM. 2016. Position statement: Access to comprehensive sexual and reproductive health care services. http://www.midwife.org/ACNM/files/ACNMLibraryData/UPLOADFILENAME/000000000087/Access-to-Comprehensive-Sexual-and-Reproductive-Health-Care-Services-FINAL-04-12-17.pdf (accessed August 1, 2017).
ACOG (American College of Obstetricians and Gynecologists). 2013. Practice Bulletin No. 135: Second-trimester abortion. Obstetrics & Gynecology 121(6):1394–1406.
ACOG. 2014. Practice Bulletin No. 143: Medical management of first-trimester abortion (reaffirmed). Obstetrics & Gynecology 123(3):676–692.
AHRQ (Agency for Healthcare Research and Quality). 2016. The six domains of health care quality. https://www.ahrq.gov/professionals/quality-patient-safety/talkingquality/create/sixdomains.html (accessed May 3, 2017).
Altman, D. G., K. F. Schulz, D. Moher, M. Egger, F. Davidoff, D. Elbourne, P. C. Gøtzsche, and T. Lang. 2001. The revised CONSORT statement for reporting randomized trials: Explanation and elaboration. Annals of Internal Medicine 134(8):663–694.
Ashok, P. W., A. Templeton, P. T. Wagaarachchi, and G. M. Flett. 2004. Midtrimester medical termination of pregnancy: A review of 1002 consecutive cases. Contraception 69(1):51–58.
Autry, A. M., E. C. Hayes, G. F. Jacobson, and R. S. Kirby. 2002. A comparison of medical induction and dilation and evacuation for second-trimester abortion. American Journal of Obstetrics and Gynecology 187(2):393–397.
Bartlett, L. A., C. J. Berg, H. B. Shulman, S. B. Zane, C. A. Green, S. Whitehead, and H. K. Atrash. 2004. Risk factors for legal induced abortion-related mortality in the United States. Obstetrics & Gynecology 103(4):729–737.
Bearak, J. M., K. L. Burke, and R. K. Jones. 2017. Disparities and change over time in distance women would need to travel to have an abortion in the USA: A spatial analysis. The Lancet Public Health 2(11):e493–e500.
Borgatta, L. 2011. Labor induction termination of pregnancy. Global library for women’s medicine. https://www.glowm.com/section_view/heading/Labor%20Induction%20Termination%20of%20Pregnancy/item/443 (accessed September 13, 2017).
Borkowski, L., J. Strasser, A. Allina, and S. Wood. 2015. Medication abortion. Overview of research & policy in the United States. http://publichealth.gwu.edu/sites/default/files/Medication_Abortion_white_paper.pdf (accessed January 25, 2017).
Bracken, M. B., D. H. Freeman, Jr., and K. Hellenbrand. 1982. Hospitalization for medical-legal and other abortions in the United States 1970–1977. American Journal of Public Health 72(1):30–37.
Bryant, A. G., D. A. Grimes, J. M. Garrett, and G. S. Stuart. 2011. Second-trimester abortion for fetal anomalies or fetal death: Labor induction compared with dilation and evacuation. Obstetrics & Gynecology 117(4):788–792.
Cates, Jr., W., K. F. Schulz, D. A. Grimes, A. J. Horowitz, F. A. Lyon, F. H. Kravitz, and M. J. Frisch. 1982. Dilatation and evacuation procedures and second-trimester abortions. The role of physician skill and hospital setting. Journal of the American medical Association 248(5):559–563.
Cates, Jr., W., D. A. Grimes, and K. F. Schulz. 2000. Abortion surveillance at CDC: Creating public health light out of political heat. American Journal of Preventive Medicine 19(1, Suppl. 1):12–17.
CDC (Centers for Disease Control and Prevention). 1983. Surveillance summary abortion surveillance: Preliminary analysis, 1979–1980—United States. MMWR Weekly 32(5): 62–64. https://www.cdc.gov/mmwr/preview/mmwrhtml/00001243.htm (accessed September 18, 2017).
CDC. 2017. CDC’s abortion surveillance system FAQs. https://www.cdc.gov/reproductivehealth/data_stats/abortion.htm (accessed June 22, 2017).
Chen, M. J., and M. D. Creinin. 2015. Mifepristone with buccal misoprostol for medical abortion: A systematic review. Obstetrics & Gynecology 126(1):12–21.
Cleland, K., M. D. Creinin, D. Nucatola, M. Nshom, and J. Trussell. 2013. Significant adverse events and outcomes after medical abortion. Obstetrics & Gynecology 121(1):166–171.
Costescu, D., E. Guilbert, J. Bernardin, A. Black, S. Dunn, B. Fitzsimmons, W. V. Norman, H. Pymar, J. Soon, K. Trouton, M. S. Wagner, and E. Wiebe. 2016. Medical abortion. Journal of Obstetrics and Gynaecology Canada 38(4):366–389.
Donabedian, A. 1980. The definition of quality and approaches to its assessment. In Explorations in quality assessment and monitoring. Vol. 1. Ann Arbor, MI: Health Administration Press.
Edelman, D. A., W. E. Brenner, and G. S. Berger. 1974. The effectiveness and complications of abortion by dilatation and vacuum aspiration versus dilatation and rigid metal curettage. American Journal of Obstetrics and Gynecology 119(4):473–480.
Elam-Evans, L. D., L. T. Strauss, J. Herndon, W. Y. Parker, S. V. Bowens, S. Zane, and C. J. Berg. 2003. Abortion surveillance—United States, 2000. MMWR Surveillance Summaries 52(SS-12):1–32. https://www.cdc.gov/mmwr/preview/mmwrhtml/ss5212a1.htm (accessed September 18, 2017).
FDA (U.S. Food and Drug Administration). 2016. MIFEPREX®: Highlights of prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/020687s020lbl.pdf (accessed September 11, 2017).
Finer, L. B., and S. K. Henshaw. 2003. Abortion incidence and services in the United States in 2000. Perspectives on Sexual and Reproductive Health 35(1):6–15.
Finer, L. B., and S. K. Henshaw. 2006. Disparities in rates of unintended pregnancy in the United States, 1994 and 2001. Perspectives on Sexual and Reproductive Health 38(2):90–96.
Finer, L. B., and M. R. Zolna. 2016. Declines in unintended pregnancy in the United States, 2008–2011. New England Journal of Medicine 374(9):843–852.
Finer, L. B., L. F. Frohwirth, L. A. Dauphinee, S. Singh, and A. M. Moore. 2006. Timing of steps and reasons for delays in obtaining abortions in the United States. Contraception 74(4):334–344.
Frick, A. C., E. A. Drey, J. T. Diedrich, and J. E. Steinauer. 2010. Effect of prior cesarean delivery on risk of second-trimester surgical abortion complications. Obstetrics & Gynecology 115(4):760–764.
Gary, M. M., and D. J. Harrison. 2006. Analysis of severe adverse events related to the use of mifepristone as an abortifacient. Annals of Pharmacotherapy 40(2):191–197.
Glasziou, P., E. Meats, C. Heneghan, and S. Shepperd. 2008. What is missing from descriptions of treatment in trials and reviews? British Medical Journal 336(7659):1472–1474.
Grimes, D. A., and G. Stuart. 2010. Abortion jabberwocky: The need for better terminology. Contraception 81(2):93–96.
Grimes, D. A., S. M. Smith, and A. D. Witham. 2004. Mifepristone and misoprostol versus dilation and evacuation for midtrimester abortion: A pilot randomised controlled trial. British Journal of Obstetrics & Gynaecology 111(2):148–153.
Grossman, D., K. Blanchard, and P. Blumenthal. 2008. Complications after second trimester surgical and medical abortion. Reproductive Health Matters 16(31 Suppl.):173–182.
Grossman, D., K. Grindlay, T. Buchacker, K. Lane, and K. Blanchard. 2011. Effectiveness and acceptability of medical abortion provided through telemedicine. Obstetrics & Gynecology 118(2 Pt. 1):296–303.
Guttmacher Institute. 2017a. Fact sheet: Induced abortion in the United States. https://www.guttmacher.org/fact-sheet/induced-abortion-united-states (accessed November 10, 2017).
Guttmacher Institute. 2017b. Bans on specific abortion methods used after the first trimester. https://www.guttmacher.org/state-policy/explore/bans-specific-abortion-methods-used-after-first-trimester (accessed September 12, 2017).
Guttmacher Institute. 2017c. Counseling and waiting periods for abortion. https://www.guttmacher.org/state-policy/explore/counseling-and-waiting-periods-abortion (accessed September 12, 2017).
Guttmacher Institute. 2017d. Medication abortion. https://www.guttmacher.org/state-policy/explore/medication-abortion (accessed September 12, 2017).
Guttmacher Institute. 2017e. An overview of abortion laws. https://www.guttmacher.org/state-policy/explore/overview-abortion-laws (accessed September 12, 2017).
Guttmacher Institute. 2017f. Requirements for ultrasound. https://www.guttmacher.org/state-policy/explore/requirements-ultrasound (accessed September 12, 2017).
Guttmacher Institute. 2017g. State funding of abortion under Medicaid. https://www.guttmacher.org/state-policy/explore/state-funding-abortion-under-medicaid (accessed September 12, 2017).
Guttmacher Institute. 2017h. State policies on later abortions. https://www.guttmacher.org/state-policy/explore/state-policies-later-abortions (accessed September 12, 2017).
Guttmacher Institute. 2017i. Targeted regulation of abortion providers. https://www.guttmacher.org/state-policy/explore/targeted-regulation-abortion-providers (accessed September 12, 2017).
Guttmacher Institute. 2018a. Abortion reporting requirements. https://www.guttmacher.org/state-policy/explore/abortion-reporting-requirements (accessed January 22, 2018).
Guttmacher Institute. 2018b. Restricting insurance coverage of abortion. https://www.guttmacher.org/state-policy/explore/restricting-insurance-coverage-abortion (accessed January 24, 2018).
Hopewell, S., M. Clarke, D. Moher, E. Wager, P. Middleton, D. G. Altman, K. F. Schulz, and the CONSORT Group. 2008. CONSORT for reporting randomized controlled trials in journal and conference abstracts: Explanation and elaboration. PLoS Medicine 5(1):e20.
Ioannidis, J. P., S. J. Evans, P. C. Gøtzsche, R. T. O’Neill, D. G. Altman, K. Schulz, D. Moher, and the CONSORT Group. 2004. Better reporting of harms in randomized trials: An extension of the CONSORT statement. Annals of Internal Medicine 141(10):781–788.
IOM (Institute of Medicine). 1975. Legalized abortion and the public health. Washington, DC: National Academy Press.
IOM. 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
IOM. 2011a. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
IOM. 2011b. Finding what works in health care: Standards for systematic reviews. Washington, DC: The National Academies Press.
Ireland, L. D., M. Gatter, and A. Y. Chen. 2015. Medical compared with surgical abortion for effective pregnancy termination in the first trimester. Obstetrics & Gynecology 126(1):22–28.
Jatlaoui, T. C., A. Ewing, M. G. Mandel, K. B. Simmons, D. B. Suchdev, D. J. Jamieson, and K. Pazol. 2016. Abortion surveillance—United States, 2013. MMWR Surveillance Summaries 65(No. SS-12):1–44.
Jerman, J., and R. K. Jones. 2014. Secondary measures of access to abortion services in the United States, 2011 and 2012: Gestational age limits, cost, and harassment. Women’s Health Issues 24(4): e419–e424.
Jerman J., R. K. Jones, and T. Onda. 2016. Characteristics of U.S. abortion patients in 2014 and changes since 2008. https://www.guttmacher.org/sites/default/files/report_pdf/characteristics-us-abortion-patients-2014.pdf (accessed October 17, 2016).
Jerman, J., L. Frohwirth, M. L. Kavanaugh, and N. Blades. 2017. Barriers to abortion care and their consequences for patients traveling for services: Qualitative findings from two states. Perspectives on Sexual and Reproductive Health 49(2):95–102.
Jones, R. K., and H. D. Boonstra. 2016. The public health implications of the FDA update to the medication abortion label. New York: Guttmacher Institute. https://www.guttmacher.org/article/2016/06/public-health-implications-fda-update-medication-abortion-label (accessed October 27, 2017).
Jones, R. K., and J. Jerman. 2017a. Abortion incidence and service availability in the United States, 2014. Perspectives on Sexual and Reproductive Health 49(1):1–11.
Jones, R. K., and J. Jerman. 2017b. Characteristics and circumstances of U.S. women who obtain very early and second trimester abortions. PLoS One 12(1):e0169969.
Jones, R. K., and M. L. Kavanaugh. 2011. Changes in abortion rates between 2000 and 2008 and lifetime incidence of abortion. Obstetrics & Gynecology 117(6):1358–1366.
Jones, R. K., L. B. Finer, and S. Singh. 2010. Characteristics of U.S. abortion patients, 2008. New York: Guttmacher Institute.
Kahn, J. B., J. P. Bourne, J. D. Asher, and C. W. Tyler. 1971. Technical reports: Surveillance of abortions in hospitals in the United States, 1970. HSMHA Health Reports 86(5):423–430.
Kelly, T., J. Suddes, D. Howel, J. Hewison, and S. Robson. 2010. Comparing medical versus surgical termination of pregnancy at 13–20 weeks of gestation: A randomised controlled trial. British Journal of Obstetrics & Gynaecology 117(12): 1512–1520.
Koonin, L. M., and J. C. Smith. 1993. Abortion surveillance—United States, 1990. MMWR Surveillance Summaries 42(SS-6):29–57. https://www.cdc.gov/mmwr/preview/mmwrhtml/00031585.htm (accessed September 18, 2017).
Kost, K. 2015. Unintended pregnancy rates at the state level: Estimates for 2010 and trends since 2002. New York: Guttmacher Institute.
Kulier, R., N. Kapp, A. M. Gulmezoglu, G. J. Hofmeyr, L. Cheng, and A. Campana. 2011. Medical methods for first trimester abortion. The Cochrane Database of Systematic Reviews (11):CD002855.
Lawson, H. W., H. K. Atrash, A. F. Saftlas, L. M. Koonin, M. Ramick, and J. C. Smith. 1989. Abortion surveillance, United States, 1984–1985. MMWR Surveillance Summaries 38(SS-2):11–15. https://www.cdc.gov/Mmwr/preview/mmwrhtml/00001467.htm (accessed September 18, 2017).
Lean, T. H., D. Vengadasalam, S. Pachauri, and E. R. Miller. 1976. A comparison of D & C and vacuum aspiration for performing first trimester abortion. International Journal of Gynecology and Obstetrics 14(6):481–486.
Lichtenberg, E. S., and M. Paul. 2013. Surgical abortion prior to 7 weeks of gestation. Contraception 88(1):7–17.
Lohr, A. P., J. L. Hayes, and K. Gemzell Danielsson. 2008. Surgical versus medical methods for second trimester induced abortion. Cochrane Database of Systematic Reviews (1):CD006714.
Low, N., M. Mueller, H. A. Van Vliet, and N. Kapp. 2012. Perioperative antibiotics to prevent infection after first-trimester abortion. Cochrane Database of Systematic Reviews (3):CD005217.
Mauelshagen, A., L. C. Sadler, H. Roberts, M. Harilall, and C. M. Farquhar. 2009. Audit of short term outcomes of surgical and medical second trimester termination of pregnancy. Reproductive Health 6(1):16.
NAF (National Abortion Federation). 2017. 2017 Clinical policy guidelines for abortion care. Washington, DC: NAF.
Nash, E., R. B. Gold, L. Mohammed, O. Cappello, and Z. Ansari-Thomas. 2017. Laws affecting reproductive health and rights: State policy trends at midyear, 2017. Washington, DC: Guttmacher Institute. https://www.guttmacher.org/article/2017/07/laws-affecting-reproductive-health-and-rights-state-policy-trends-midyear-2017 (accessed September 21, 2017).
Ngoc, N. T., T. Shochet, S. Raghavan, J. Blum, N. T. Nga, N. T. Minh, V. Q. Phan, B. Winikoff. 2011. Mifepristone and misoprostol compared with misoprostol alone for second-trimester abortion: A randomized controlled trial. Obstetrics & Gynecology 118(3):601–608.
Ohannessian, A., K. Baumstarck, J. Maruani, E. Cohen-Solal, P. Auquier, and A. Agostini. 2016. Mifepristone and misoprostol for cervical ripening in surgical abortion between 12 and 14 weeks of gestation: A randomized controlled trial. European Journal of Obstetrics & Gynecology and Reproductive Biology 201:151–155.
Pazol, K., A. A. Creanga, and S. B. Zane. 2012. Trends in use of medical abortion in the United States: Reanalysis of surveillance data from the Centers for Disease Control and Prevention, 2001–2008. Contraception 86(6):746–751.
Pazol, K., A. A. Creanga, and D. J. Jamieson. 2015. Abortion surveillance—United States, 2012. Morbidity and Mortality Weekly Report 64(SS-10):1–40.
Peterson, W. F., F. N. Berry, M. R. Grace, and C. L. Gulbranson. 1983. Second-trimester abortion by dilatation and evacuation: An analysis of 11,747 cases. Obstetrics & Gynecology 62(2):185–190.
Plint, A. C., D. Moher, A. Morrison, K. Schulz, D. G. Altman, C. Hill, and I. Gaboury. 2006. Does the CONSORT checklist improve the quality of reports of randomised controlled trials? A systematic review. Medical Journal of Australia 185(5):263–267.
Ranji, U., A. Salganicoff, L. Sobel, C. Rosenzweig, and I. Gomez. 2017. Financing family planning services for low-income women: The role of public programs. https://www.kff.org/womens-health-policy/issue-brief/financing-family-planning-services-for-low-income-women-the-role-of-public-programs (accessed September 9, 2017).
Raymond, E. G., C. Shannon, M. A. Weaver, and B. Winikoff. 2013. First-trimester medical abortion with mifepristone 200 mg and misoprostol: A systematic review. Contraception 87(1):26–37.
RCOG (Royal College of Obstetricians and Gynaecologists). 2011. The care of women requesting induced abortion (Evidence-based clinical guideline number 7). London, UK: RCOG Press. https://www.rcog.org.uk/globalassets/documents/guidelines/abortion-guideline_web_1.pdf (accessed July 27, 2017).
RCOG. 2015. Best practice in comprehensive abortion care (Best practice paper no. 2). London, UK: RCOG Press. https://www.rcog.org.uk/globalassets/documents/guidelines/best-practice-papers/best-practice-paper-2.pdf (accessed September 11, 2017).
Roblin, P. 2014. Vacuum aspiration. In Abortion care, edited by S. Rowlands. Cambridge, UK: Cambridge University Press.
Sackett, D. L. 1979. Bias in analytic research. Journal of Chronic Diseases 32(1–2):51–63.
Sonalkar, S., S. N. Ogden, L. K. Tran, and A. Y. Chen. 2017. Comparison of complications associated with induction by misoprostol versus dilation and evacuation for second-trimester abortion. International Journal of Gynaecology & Obstetrics 138(3):272–275.
Strauss, L. T., S. B. Gamble, W. Y. Parker, D. A. Cook, S. B. Zane, and S. Hamdan. 2007. Abortion surveillance—United States, 2004. MMWR Surveillance Summaries 56 (SS-12):1–33.
Upadhyay, U. D., S. Desai, V. Zlidar, T. A. Weitz, D. Grossman, P. Anderson, and D. Taylor. 2015. Incidence of emergency department visits and complications after abortion. Obstetrics & Gynecology 125(1):175–183.
von Elm, E., D. G. Altman, M. Egger, S. J. Pocock, P. C. Gøtzsche, and J. P. Vandenbrouke. 2007. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. PLoS Medicine 4(10):e296.
White, K., E. Carroll, and D. Grossman. 2015. Complications from first-trimester aspiration abortion: A systematic review of the literature. Contraception 92(5):422–438.
WHO (World Health Organization). 2012. Safe abortion: Technical and policy guidance for health systems (Second edition). http://apps.who.int/iris/bitstream/10665/70914/1/9789241548434_eng.pdf (accessed September 12, 2017).
WHO. 2014. Clinical practice handbook for safe abortion. Geneva, Switzerland: WHO Press. http://apps.who.int/iris/bitstream/10665/97415/1/9789241548717_eng.pdf?ua=1&ua=1 (accessed November 15, 2016).
Wildschut, H., M. I. Both, S. Medema, E. Thomee, M. F. Wildhagen, and N. Kapp. 2011. Medical methods for mid-trimester termination of pregnancy. The Cochrane Database of Systematic Reviews (1):Cd005216.
Woodcock, J. 2016. Letter from the director of the FDA Center for Drug Evaluation and Research to Donna Harrison, Gene Rudd, and Penny Young Nance. Re: Docket No. FDA-2002-P-0364. Silver Spring, MD: FDA.
Zane, S., A. A. Creanga, C. J. Berg, K. Pazol, D. B. Suchdev, D. J. Jamieson, and W. M. Callaghan. 2015. Abortion-related mortality in the United States: 1998–2010. Obstetrics & Gynecology 126(2):258–265.




























