9
Health Monitoring and Research Programs
Veterans of all eras are concerned that their environmental exposures during deployment may have ramifications for their reproductive health and for the health of their children and grandchildren. The convergence of veteran concerns with the rise of epigenetics has fueled interest in the study of the role of gene x environment interactions in mediating adverse reproductive and transgenerational health outcomes. Acknowledging those concerns, the Department of Veterans Affairs (VA) requested that the Volume 11 committee update what is currently known about the reproductive and developmental effects associated with the exposures potentially experienced by Gulf War and Post-9/11 veterans. In addition, VA also asked the committee to identify areas requiring further study in order to assess the potential health effects in the descendants of veterans of any era and to provide guidance on how such research might be conducted. To address the latter task, the committee was requested to do the following:
- develop a framework for a health monitoring or screening program that would be applicable to all veterans and their descendants;
- determine the feasibility of conducting an epidemiologic study on generational health effects in veterans and their descendants; and
- assess the scope and methodology for conducting research, including the use of animal models, on generational health effects.
The committee posits that the implementation of a comprehensive health monitoring and research program (HMRP) that involves veterans, their partners,1 and their descendants could help to identify areas of priority for VA and provide an appropriate framework for addressing areas of concern in the future. Because a health monitoring program and an epidemiologic research program need to work together to inform each other, this chapter discusses the committee approach to both aspects of an HMRP. Chapter 10 provides guidance and an overview on the basic and translational research studies that can
___________________
1 The committee uses this term to capture male or female spouses, the child’s other biological parent, or caregivers who can respond to questions about the child’s health and development.
be considered for the study of generational health effects in animal and cellular models. The proposed HMRP should be structured not only to provide a long-term mechanism for health monitoring but also to create the infrastructure and data resources required to design epidemiologic studies to appropriately examine specific veteran and descendant populations for the health outcomes of concern; it should also establish a strong basic and translational research program that can help fill current and future gaps in knowledge. The proposed HMRP should build on the health outcomes identified in Chapters 4 through 7, whether to expand the evidence base for relatively well-studied toxicants (e.g., chlorpyrifos and trichloroethylene) or to identify possible reproductive and developmental effects for poorly studied toxicants (e.g., depleted uranium and jet fuels). It should be tailored to enroll participants who are representative of all conflicts as well as appropriate comparison groups of nondeployed members of the military and civilian populations. By design, the HMRP should create an infrastructure for health monitoring at health care facilities within VA as well as seek participation of health care organizations and providers outside of VA that are involved in providing care to veterans, their partners, and their descendants. This is particularly important given the necessary involvement of partners and children of veterans who may not be receiving care within VA facilities.
Epidemiology studies provide a unique opportunity to study the distribution and determinants of health-related states or events (including disease) and also to apply these studies to the control of diseases and other health problems (WHO, 2018). Epidemiology is often integrated with other scientific disciplines, including biostatistics, the social and behavioral sciences, and environmental health, to study the extent to which a particular exposure might be associated with a health outcome. Epidemiologic studies provide the opportunity for the monitoring and screening of the health of populations over time as well as the testing of hypothesis-driven research to address specific questions—the functions that together constitute an HMRP. These two aspects of epidemiology are not mutually exclusive, but rather work to complement each other and to enhance the value of the research findings. The proposed research program also emphasizes the importance of using experimental animal or cell culture models to help elucidate the biological processes that may be responsible for the generational health effects that appear after deployment exposures (see Chapter 10). As such, specific epidemiology and basic and translational research programs should be established to take advantage of the availability of data collected through the HMRP. Future studies may be nested within the HMRP. These studies may make direct or indirect use of the data collected by the HMRP, identify cases among HMRP participants, or focus on specific exposures or outcomes of interest that have been identified through the HMRP.
Below, the committee provides a framework for the development of an HMRP and ways to optimize current research efforts to conduct longitudinal generational studies. The logistic, organizational, technological, laboratory, and statistical considerations needed to support these efforts are also presented.
OVERVIEW OF VA AND DOD HEALTH CARE SYSTEMS
The Military Health System (MHS) operates within the Department of Defense (DoD) to provide comprehensive health care to active-duty military personnel. The MHS also provides, when space is available, health care to dependents of active-duty service members, to retirees and their dependents, and to some former partners. Such care has been made available since 1966 through TRICARE and its predecessors. In October 2001, TRICARE benefits were extended to retirees and their dependents ages 65 and over. Since 2014 TRICARE has been managed by the Defense Health Agency.
The TRICARE network represents a major component of the MHS. Although TRICARE is sometimes used to describe only purchased care, the committee uses the term in a broader sense: as a wide-reaching health care provider for DoD beneficiaries, including service members, retirees, and their
families that delivers direct care through military treatment facilities as well as purchased care through network and nonnetwork civilian health professionals, hospitals, and pharmacies (DoD, 2013). The DoD TRICARE Management Activity contracts with community purchased-care providers when direct-care providers are not available or when supplemental services are required.
In 2013 an estimated 9.4 million beneficiaries were enrolled in the TRICARE system, including 1.37 million active-duty service members, 1.71 million active-duty family members, 170,000 active and inactive National Guard and reserve members, 750,000 family members of National Guard and reservists, and 5.42 million retirees and their family members (DoD, 2018). According to TRICARE Management Activity, the TRICARE network includes 56 hospitals and medical centers and 361 ambulatory health clinics that provide direct care in the MHS, and more than 3,300 network acute-care hospitals and 914 behavioral health facilities provide purchased care (DoD, 2013).
Once a service member separates from the military and becomes an eligible veteran, he or she may receive health care from VA through a network of hospitals, clinics, centers, and long-term care facilities located around the country. VA also has contracts with many health care providers who may treat veterans when they do not have ready access to a VA facility. VA does not typically provide direct health care to veterans’ dependents; however, the dependents of veterans who are permanently and totally disabled or who died of a service-connected disability may qualify for CHAMPVA, a health insurance program that relies on private doctors and hospitals for the provision of care. As of 2014, 42% of the total veteran population receives its health care directly from VA or through a VA contract with a private health care provider (Bagalman, 2014).
Although some veterans receive health care within VA, many do not. Veterans who are eligible for VA health benefits are assigned to one of eight priority groups based on need, period of service, and other considerations such as a service-connected disability and being a Medal of Honor recipient (VA, 2018a). Reserve or National Guard members on active duty for training purposes only do not meet the basic eligibility requirement. Veterans who are not eligible for VA health care may receive care from a variety of other health care providers outside the VA system. Those outside the VA health care system may be difficult to identify as discussed in the Study Populations section later in the chapter. For example, the 2010 National Survey of Veterans found that fewer than 30% of Vietnam veterans had ever enrolled in VA health care services (Westat, 2010). The health care facilities and providers for veterans and their descendants are essential elements of the HMRP.
FRAMEWORK FOR A HEALTH MONITORING AND RESEARCH PROGRAM
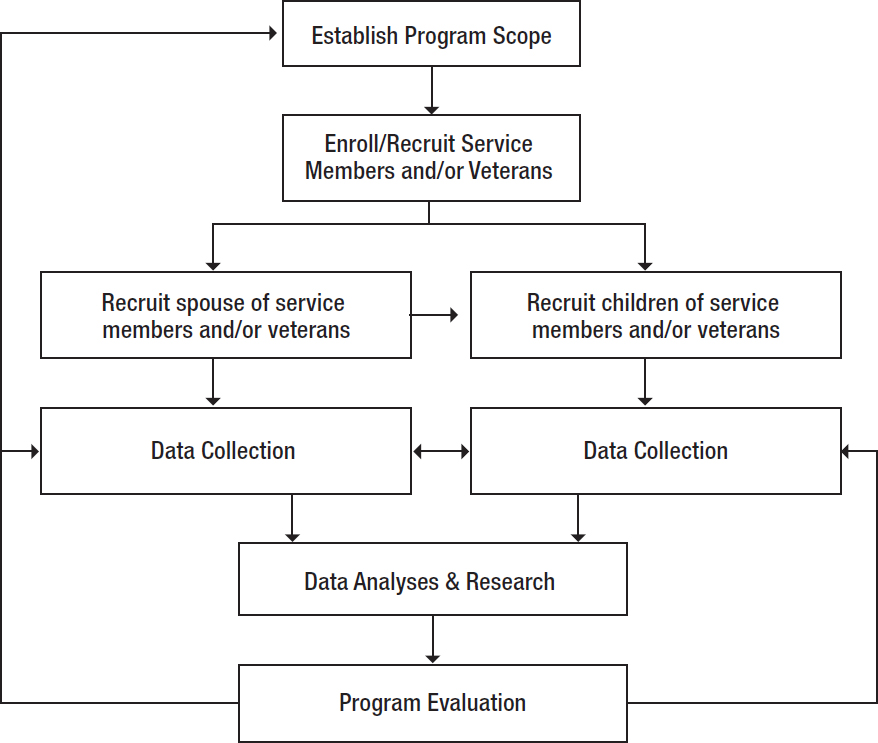
The committee designed a general framework (see Figure 9-1) applicable to any veteran era that can serve as a starting point for a research program. As science evolves, modifications will likely be implemented to improve the framework. An HMRP is an ongoing initiative designed to assess a wide range of health effects in veterans, their partners, and their children and grandchildren, including reproductive and development outcomes. Participants should be assessed periodically using a systematic, longitudinal approach that includes service members and veterans—both men and women—and even those who have yet to join the military. Veterans of the Vietnam War era, the 1990–1991 Gulf War era, and the Post-9/11 era should all be adequately represented.
The framework in Figure 9-1 can be tailored to the specific research questions and the specific veteran cohorts being evaluated, but the basic elements of the framework should remain consistent. To ensure success, a strong program management team will be critical. The lead organization for any given project within the HMRP needs to be specified a priori and the collaborators promptly identified. Inherent in effective program management are communication strategies for both internal and external
audiences (as discussed in more detail later in the chapter), particularly program participants (i.e., service members and veterans, and their descendants) and researchers; the assessment of resources that are available or that will be necessary to conduct the program; the development and implementation of standard operating procedures for data collection, storage, and analysis; and a rigorous process for ensuring that all ethical and legal requirements for human subjects are strictly adhered to. Information technology (IT) will also be an essential aspect of program management since the data to be collected on participants will likely be extensive, as will the links necessary between federal and private (and possibly state) databases. Thus, there is a complexity to VA and DoD health care studies that will require careful scoping and planning in order to conduct the committee’s recommended HMRP; this planning should include the specification of methods to track program participants longitudinally in such a way that family-based links can be made and followed.
The program should begin by defining the scope of the health monitoring efforts. This scope will guide the recruitment, enrollment, and engagement of participants—active-duty service members, reservists, veterans, partners, and their descendants—now and in the future, as well as the collection and maintenance of the data for each participant. As these data will not only be electronic but also biological, it will be important for the program to be adequately prepared to receive, catalog, and maintain multiple forms of information. Analyzing data will also require linking databases and statistical software. Biological samples provide one of the keys to understanding a physiological response. Given the essential
nature of this component, it will be important to identify appropriate sample collection and storage processing systems and equipment as well as suitable laboratory facilities and biorepositories, and it will be necessary to develop standardized protocols. Among the details of the methodology, emphasis should be placed on the frequency and duration of the monitoring program and also the assessment of any laboratory technologies that might be used in the process.
Publishing program results in a systematic manner, perhaps as a series of papers from program researchers, will ensure that veterans and their descendants are aware of the findings, both positive and negative, that may affect their health and well-being. Furthermore, dissemination to a broader audience, including researchers and the general public, can spur interest in the program and its activities. Finally, the health monitoring effort needs to be evaluated, modified, and updated on a continuing and regular basis, possibly with the oversight of an external advisory board (see Collaboration and Coordination later in this chapter). Without adequate review of all aspects of the program, its resources may not be efficiently used, data collection and analysis may lag, and veterans and their descendants may be participating in a program that does not serve their needs. In the sections below, the committee considers each of these elements in greater detail.
Of critical importance for a successful health monitoring effort will be close and continuous collaboration among and between the various government and private entities that are responsible for the health of veterans and their descendants. As will become evident in the following sections, no one agency is likely to have all the necessary data and resources to implement any part of the program on its own. Therefore, the committee encourages VA, DoD, and the National Institutes of Health (NIH) to work together with other stakeholders, particularly veterans and their families, and with academia to address the veterans’ concerns about the possible effects that their war zone exposures may have on their children and future generations.
PROGRAM SCOPE AND STUDY DESIGN
Scope Definition
The first priority of the health monitoring part of an HMRP is to define the program scope, including its goals, priority data measurements, and which health outcomes should be monitored. A second priority might be to decide on the epidemiologic studies that are to be carried out with data from the health monitoring data repositories or that address other critical research questions. The scope of the HMRP will be a major determinant of the overall value of the data and the costs of recruiting and following up on participants over the long term. Establishing the scope of the program and the methodology to be used must be done with care and with input from stakeholders, including veterans and their descendants along with governmental and private entities.
The committee believes that the most effective approach will be to tailor the management of each HMRP for the particular veteran cohort under study. An effective management strategy will require an initial designated lead organization and management team, although the lead organization may change over time as the cohort population evolves or as the research questions change. For example, conducting a study of Vietnam veterans may require VA to serve as the lead organization with support from DoD and other agencies, whereas a study of future veterans may be led by DoD when service members and their families receive care from the MHS and then later transition to VA as service members and their families leave the MHS. Establishing the program scope and goals early in the process will not only provide the overarching framework for the program, but will also help to provide a platform with which to establish communication with veterans and collaborating entities, thus helping to promote clarity and transparency and optimize the use of resources.
One program goal might be to identify the reproductive health effects of veterans that may be the result of specific military exposures, particularly deployments to a war zone. For example, a small health monitoring program is being conducted by the Baltimore VA medical center to assess the health of male Gulf War veterans exposed to depleted uranium through embedded fragments (continuous exposure); however, while the reproductive status of these veterans is being assessed, the health of their descendants is not (McDiarmid et al., 2015).
The program scope should embrace continuous improvement as the program matures and data are analyzed and new health concerns emerge. Focus groups and other exploratory strategies with both veterans and health experts can help identify the specific exposures, health effects, populations, data sets, logistical considerations, and study designs that are most appropriate. These exploratory strategies could answer such questions as:
- Which cohort of veterans is to be studied and why?
- What exposures are most relevant to the specific veteran cohort?
- Are exposures comparable among different cohorts that might help substantiate or provide additional information?
- What does the current scientific and medical literature or other data resources suggest are the health outcomes that should be monitored in veterans and their descendants?
- How long and how often will the monitoring be conducted?
- How and where will biospecimens be collected, processed, catalogued, and maintained?
- What are the deliverables of the program?
- How will success be determined, and what metrics will be used for evaluation?
- What partnerships are needed to carry out the tasks?
Once these and other relevant questions are addressed, pilot studies could be conducted before larger studies are initiated. The committee recommends that VA or other lead organizations design and manage a pilot program that can be used to determine the feasibility of collection strategies, the necessary linkages for the appropriate use of the data, an assessment of response rates among veterans and their descendants, and the testing of whether a survey or other data collection methods can be scaled up and made operable in the field. The results from a pilot program may help to further tailor the scope of the program. The results from the pilot studies should be disseminated to interested parties and the public to provide the basis for a decision on whether to implement a large-scale set of specific studies.
Implementing a pilot study may be the first tier in a multitiered approach to an HMRP. Building on the information gathered in the pilot study, the next tier might encompass a large epidemiologic study that looks more broadly at additional reproductive or developmental health outcomes, a larger cohort of veterans or descendants, or some other variable. Each tier should build on the previous one in terms of scope, study parameters, duration, and resource needs. The committee notes that in Chapter 4, Deployment-Related Exposures, VA and DoD have already conducted epidemiologic studies that may be considered as pilot studies that provide useful information for designing more comprehensive studies. For example, to assess whether exposure to burn pits resulted in adverse birth outcomes, Conlin et al. (2012) used the DoD Birth and Infant Health Registry to identify live infants born to deployed men or women between 2004 and 2007 and categorized veterans as being within a specified distance of a burn pit. This might be considered to be a pilot study. Building on this study, VA and DoD collaborators might follow the infants as they grow and assess long-term effects and distinguish between maternal and paternal exposures. A further tier may be prospective and assess the health of children born to men and women after they return from deployment and even after they leave the military. Again, the cohorts
would be categorized as exposed or unexposed to burn pits during deployment. For each tier, feasibility must be evaluated with respect to the strength of the associations being seen in the prior tier and the availability of data and resources to conduct the next tier.
The committee makes no assumptions about VA’s goals for an HMRP because the questions to be addressed depend on how VA chooses to prioritize potential deployment-related health effects. For example, questions may be prioritized based on gaps in existing knowledge, concerns raised by veterans, the burden of disease, frequent or widespread exposures, the level of risk or change to be observed, or other issues. Furthermore, it is expected that these goals may change over time as more information on generational effects becomes known. Important to an HMRP is that baseline parameters be established for all participants against which future comparisons can be made. Depending on specific goals, collecting baseline data from active-duty service members may begin when these individuals enter the military and before orders are presented, before and after deployment, or upon the completion of specific assignments. The system should be capable of providing data on the incidence and prevalence of veterans’ reproductive health outcomes as well as on the health of their children as they mature. For descendants, baseline data may be collected at birth or after enrollment in the HMRP. For parents who are not active-duty service members or veterans, baseline data may be collected when they enter the program or when they (if women) become pregnant.
Study Design
Various methods can be used to study human health outcomes, including screening, health monitoring, and research. Screening is used to identify the presence of a disease in a target population. The disease is usually known to be an important health problem and one for which there is an effective treatment if detected at an early stage (e.g., screening for colon cancer in individuals 50 years or older) (Andermann et al., 2008). In contrast, health monitoring is the continuous, systematic collection, analysis, and interpretation of health-related data that are needed for the planning, implementation, and evaluation of programs to improve the health of a population. A health monitoring initiative may be used to identify and characterize health problems in a population, to aid in setting priorities for interventions, and to inform future research efforts. Thus, the HMRP outlined here must be distinguished from a surveillance program that passively acquires data and that often lacks participant input. Surveillance programs—such as those that assess health effects in workers who handle hazardous substances—or registries—such as state-sponsored cancer registries that collect data without the active participation of the patients—may be considered as options for assessing veterans’ health, but they were deemed by the committee to be unsuitable for the purpose it envisions, although they may be used to provide supplemental information.
An epidemiologic research study addresses a specific hypothesis about whether or how an exposure contributes to the risk of a particular disease or other health outcome. Box 9-1 highlights some existing HMRPs and epidemiologic studies that have assessed health effects in various military and civilian populations and that serve as examples of potential approaches for examining health effects in veterans and generational health effects in their descendants. In addition to the large, longitudinal studies presented in Box 9-1, there are several ongoing studies that may be used by VA and other organizations as examples of how children may be evaluated for health effects as they grow. Several of these studies are discussed in Chapters 5 through 7 and include the assessment of children in eight towns on Cape Cod with drinking water contaminated by tetrachloroethylene (Janulewicz et al., 2008), the Center for the Health Assessment of Mothers and Children of Salinas study in California (Eskenazi et al., 2004), and the Longitudinal Study of Early Development (Stingone et al., 2016). Each of these epidemiologic studies has its strengths and weaknesses and together they illustrate some of the issues that the Volume
11 committee believes may be addressed by the HMRP framework presented in this chapter. These issues include but are not limited to the following: funding issues (e.g., Air Force Health Study), imprecise or changing program goals (e.g., National Children’s Study), poor response rates (National Health Survey of Gulf War Veterans and Their Families), and poor retention rates (Millennium Cohort Study), or they did not collect sufficient data to be applicable to other goals (Millennium Cohort Study). Most of the studies were conducted by one government organization, such as DoD, VA, or NIH, which may make it difficult to leverage resources from other organizations. The committee believes that the HMRP described in this chapter can help overcome some of the challenges inherent in conducting large-scale prospective or retrospective monitoring programs and epidemiologic studies.
There are a number of different epidemiologic study designs which vary in cost, time frame, the selection of study subjects, data collection methods, sample size, causal inference, and feasibility. The choice of study design often depends on the hypothesis being tested and on the study population of interest. The current gold standard for epidemiologic study design is the randomized controlled trial (RCT) in which individuals are randomly assigned to either receive or not receive a given exposure in order to determine whether the exposure increases or decreases the risk of a given outcome. RCTs are
commonly used to assess the effectiveness of drugs, but they are difficult to conduct because of their cost, the length of time necessary to see differences in the populations, and, in some instances, the ethical implications of withholding or providing an intervention versus the current standard of care.
Because of these constraints, most epidemiologic studies are observational in nature: that is, the exposures occur through environmental or occupational scenarios or by accident and are therefore not intentional. Most assessment of the effects of deployment-related exposures will be from observational studies. The most commonly used observational studies are cohort studies, case-control studies, and cross-sectional studies. VA has extensive experience in exposure cohort study designs and has used them to assess health outcomes in different cohorts of veterans. For example, VA has conducted three waves for its National Health Survey of Gulf War Era Veterans and Their Families (Dursa et al., 2016), has launched the Health Study for a New Generation of U.S. Veterans, and is currently recruiting participants for its Vietnam Era Health Retrospective Observational Study (VA, 2017a). Each of these cohort studies compares deployed (exposed) veterans with veterans of the same era who did not deploy. Some VA cohort studies stratify veterans based on the number of deployments they have experienced; although veterans of the 1990–1991 Gulf War deployed only once, Post-9/11 veterans have often had more than one deployment.
TABLE 9-1 Epidemiologic Study Designs: Definitions, Advantages, and Disadvantages
| Type of Study | Advantages | Disadvantages |
|---|---|---|
| Cohort (specifically prospective) Subjects are defined according to their exposure levels and followed over time for outcome occurrence. |
|
|
| Case-control Subjects are defined as cases and controls, and exposure histories are compared. |
|
|
| Nested Case-Control Subjects with a disease and controls are sampled from a larger cohort study with prospective data collection. |
|
|
| Cross-sectional Examines the relationship between exposure and outcome prevalence in a defined population at a single point in time. |
|
|
Table 9-1 lists the advantages and disadvantages of each of the major study designs. Other study designs, such as the case-cohort approach and novel study designs that address lifecourse considerations (see Chapter 3), are less common and are not included in the table. Study designs that have particular relevance for the HMRP include perinatal and birth cohorts such as the National Collaborative Perinatal Project conducted by NIH between 1959–1974, which obtained information on pre- and postnatal child health for 58,000 pregnancies in the United States (NARA, 2015), and high-risk cohorts. High-risk cohorts are selected on the basis of their exposure history, which may be particularly useful for studying veterans and their descendants where one study variable might be a deployment exposure such as hexavalent chromium or sarin compared to a cohort of nonexposed veterans. See Gulf War and Health, Volume 8 (IOM, 2010) and Buka et al. (2018) for more detailed descriptions of epidemiologic study types and their applications. In the design of a particular study, the collection of covariates that may be assessed as potential confounders, effect modifiers, and mediators for that study need to be considered. The researcher should also consider potential sources of bias and guard against these in the study
design. Researchers also need to consider whether and how study participants vary from veterans and descendants who choose not to participate and whether those factors influence the results. Details are provided below.
In addition to the types of epidemiologic studies presented in Table 9-1, ecologic studies may also provide valuable information about the possible health effects of various exposures. In these studies, a population or community is observed, and disease rates and exposures are measured in each of a series of populations. Trends in exposure and disease and their correlation are assessed at the population level. A weakness of the ecologic study design is that it cannot discern whether the exposed individuals are the same as those who get the disease, as there are no measures conducted at the individual level. Ecologic studies may use abstracted data from published statistics and therefore do not require expensive or time-consuming data collection.
STUDY POPULATIONS
An HMRP that includes studies of generational effects requires that an appropriate study population (service members, veterans, descendants, and control populations, if applicable) be identified, recruited to participate in the HMRP, and encouraged to continue their participation over the course of the program and that efficient and effective sampling be conducted from the set of participants. In the sections below, the committee discusses the necessary elements of each of these activities.
Identification
An HMRP cannot be conducted without appropriate definition, selection, and recruitment of veterans, partners, and veterans’ children (and where relevant, grandchildren). All these populations will require appropriate participant consent, which may be modeled after the National Health and Nutrition Examination Survey (NHANES), Environmental Influences on Child Health Outcomes (ECHO), or the All of Us studies. Ethical considerations are discussed later in this chapter.
The population of concern (e.g., Gulf War veterans) and the sample to be assessed (e.g., a representative selection of 30,000 Gulf War veterans) will depend on the scope of the HMRP and the questions being asked. The population may range from all veterans, which could be required in order to study a wide range of questions, to smaller groups defined on the basis of various parameters, such as specified times or locations of deployment or exposures or perhaps particular occupations.
It will be necessary to distinguish veterans and their descendants by era. For example, the Vietnam War era is considered to be 1961 to 1975. Vietnam veterans are thus at least 62 years of age, assuming they were at least 18 years old at the time they enlisted or were drafted, and thus they are old enough to have children, grandchildren, and even great-grandchildren. The veterans of the 1990–1991 Gulf War in Iraq are likely to be at least 45 years of age and may have underage children (possibly still in the TRICARE system) and young grandchildren. Post-9/11 veterans may include new enlistees who are as young as 18 years of age, with or without children, but other veterans still serving may be older and may have children, including infants, as well as grandchildren.
DoD conducts regular health surveillance of all military personnel and their dependents through the Defense Medical Surveillance System (DMSS). However, the committee notes that National Guard and reservists who have been federalized and deployed, particularly those who have served in Iraq and Afghanistan since 2001, will not be captured in the DoD MHS after they return home from deployment.
Veterans could be identified using DoD and VA administrative records. One concern is that not all service members represented in DoD databases are represented in VA databases. VA has a congressionally
mandated priority system designed to provide health care to those most in need, with eight groups and rankings based on health status, age, and income. As such, not all service members who have a DoD electronic health record (EHR) will necessarily be eligible for care in VA, and even those that are eligible may choose not to receive care at VA health care facilities. Those who do not receive care from VA will not be captured in VA databases. These challenges are compounded by the fact that the MHS records of children of military personnel are not represented in VA medical databases at all. Children born to veterans after they leave the military will also not be in the MHS, further compounding issues concerning the availability of EHRs for veterans’ descendants.
Veterans who enter the VA health care system will be easy to capture via VA databases, whereas veterans who do not enter the system may be difficult to locate and may be less inclined to participate. Efforts to identify these men and women may require using other resources such as Social Security records and working with the DoD Defense Manpower Data Center (DMDC) to locate last known addresses. Furthermore, veterans who do not use or are not eligible for the VA system may differ from veterans in the system in important ways such as income and health status. Any such differences should be identified and assessed in any epidemiologic research study.
VA has implemented several programs, particularly registries, to collect health data on defined groups of veterans based on exposure: for instance, the Agent Orange or Airborne Hazards and Open Burn Pit registries. The registries include data on veterans who are not in the VA health care system, but the utility of such registries in investigating exposure–disease relationships is limited (NASEM, 2017).
The focus of an HMRP on assessing reproductive outcomes and generational health effects adds complexity to estimating the size of the population to be monitored. Examples of questions that need to be considered for each HMRP for a veteran cohort include the following:
- What is the size of the veteran cohort that will be representative of all branches of military service?
- What is the appropriate control population and how will they be identified and recruited?
- How many children and grandchildren need to be enrolled for adequate statistical analyses?
- What is the availability of baseline data already collected on all service members?
- Is there a need to oversample women across the branches of military service, and how feasible is it to do so?
- How many pregnancies and births are anticipated for the cohort?
- Do cost or feasibility concerns require that only a subsample of the cohort be studied?
- What are the ethical issues that need to be considered for the veterans and their descendants?
Recruitment and Engagement
In Figure 9-1, the Volume 11 committee provides a framework for assessing the generational health effects in service members, veterans, and their descendants, including men and women who enlist in the military after an HMRP is established. The HMRP begins recruitment at enlistment to provide a baseline, then continues throughout the veterans’ lives. This will ensure that a sufficient sample is available with which to observe small effects in veterans and will provide a mechanism to contact dependents (partners and children). The recruitment of veterans, their partners, and their children outside of the military system will pose additional challenges, as discussed below.
To study effects in children and grandchildren, it will be best to recruit, enroll, and collect data from both the partners and the children themselves. Data collected directly from children are important for documenting outcomes and also for assessing risk factors that appear throughout the child’s life—for
example, with older children, substance use and occupation. Access to the health records of partners will facilitate the study of confounders, such as a partner’s occupational exposures, existing health concerns (e.g., obesity, diabetes, cancer), and lifestyle factors such as smoking and nutrition that might affect a pregnancy and the developing fetus. The greater the number of partners and children in the program, the greater the statistical power of the program will be, and the less impact future attrition will have. In the sections below, the committee examines how participants ranging from newly enlisted service members to the children of Vietnam veterans may be recruited into an HMRP.
The Volume 11 committee notes that recruitment and retention are critical aspects of the HMRP. Although the VA has extensive experience in large veteran cohort studies, there have been concerns that the response rate for many of its epidemiologic studies is low. Therefore, the committee suggests that VA explore innovative methods to increase recruitment and retention. It is possible that compensation for completing surveys or providing biospecimens, reimbursement for travel costs to a clinic when necessary, or recognizing HMRP participants in publications such as newsletters may serve as inducements to participate and remain in the program. For example, the MVP Insider: A Newsletter for Million Veteran Program Participants highlights stories about individual veterans and why they are participating in the VA Million Veteran Program (MVP). VA or another lead organization will also need to consider whether and how to compensate veterans or their descendants for providing biospecimens, particularly if they are to be collected at medical clinics which may require that veterans or their descendants incur expenses to travel to a clinic. The ability to provide biospecimens through the mail at no cost to veterans and their descendants may also reduce barriers to participation.
Active-Duty Service Members
The Volume 11 committee believes that the most expedient way to observe possible generational health effects associated with military exposures will be to enroll new active-duty service members into an HMRP at the time they enter the military. By enrolling all new recruits, a baseline of health status that includes information on lifestyle and environment can be established prior to any deployment. This preemptive approach will also facilitate the tracking of exposures associated with each deployment or occupational assignment. There is precedent for automatically enrolling and collecting data on military personnel: the DoD Serum Repository collects serum samples from all personnel at regular intervals, for example. Even though the Serum Repository has inconsistent collection procedures (which are being rectified), the availability of these serum samples has been useful in pilot studies (Lushniak et al., 2016). Recruits would receive information on the value of participation in a program that monitors their exposures and health and that of their descendants. Newly recruited participants would be introduced to the concept of monitoring their future reproductive health and the health of future generations and would be informed about the importance of participation by their partners and children. Opportunities to discuss details of the program with partners should be offered. The program would follow service members throughout their military careers to the point of separating from service. If a service member prefers not to participate in the HMRP after having the goals of the program explained, they should be able to decline to participate or to participate at a later date.
Although continued participation in the HMRP would be voluntary once an individual had separated from military service, assessing the long-term sequelae from potential exposures during service requires that there be some sort of follow-up. The importance of such a monitoring program and the value to each participant would be emphasized. Currently there are approximately 2 million active-duty service members, only a portion of whom have been or will be deployed to a war zone or other theater of operation. Given the retention rates of existing programs, the committee considers it likely that a substantial
proportion of service members would not agree to be followed indefinitely. However, developing the baseline and other samples would be essential in assessing the effects of any exposure to individuals for which symptoms present after discharge. The need to maximize retention in the HMRP in turn points to the importance of having an overarching communication program so that participating individuals understand the purpose of the program and embrace its goals and potential benefits. A further consideration is that among the veterans who might agree to participate in the HMRP into the future, only a proportion will receive care at VA after they separate from the military; it will be essential to get these individual veterans to agree to permit access to their health care records from private providers.
Veterans
Recruiting veterans from earlier conflicts poses a different set of challenges. Some veterans receive health care within VA, but many do not. As noted earlier, eligibility to receive health care in VA is based on a priority system. Recruiting veterans who are already enrolled in VA does not guarantee that they will participate in ongoing surveys. For example, the annual Survey of Veteran Enrollees’ Health and Use of Health Care had a response rate of between 27–36% for 2015 to 2017 using both Web-based and mailed questionnaires (Huang et al., 2017). Veterans who receive care outside the VA system may be difficult to identify and follow, particularly if they move. Veterans who are disabled (not as a result of their military service) or otherwise qualify may be enrolled in Medicaid and those from earlier eras such as Vietnam may be enrolled in either Medicare or Medicaid. Locating those veterans in the Centers for Medicare & Medicaid Services (CMS) system would require cross-referencing the DoD DMDC files with those of CMS, a difficult and time-consuming task. Other approaches may be more fruitful, such as partnering with veterans service organizations (VSOs) for outreach efforts. VSOs may also be helpful as part of a public communication campaign to generate interest and enhance recruitment efforts.
Partners
Establishing a comprehensive HMRP for generational health effects cannot be based on the deployment exposures and reproductive health of active-duty service members and veterans alone. As described in Chapter 2, deployment exposures occur within the larger context of lifecourse exposures; an additional complexity is the fact that any exposures experienced by the child’s other parent prior to conception and during pregnancy will also play a role in generational health effects. For this reason, it will be important to recruit and engage the partners of active-duty service members and veterans. Partners of interest would be those who are the other parent, biological or not, of any children born to the active-duty service member or veteran after deployment. Nonbiological parents living with a child may provide information on the child’s environment and health as well as on any of their own exposures that may affect the child.
The enrollment of active-duty service members’ partners could begin at the time of their entry into the MHS/TRICARE system as a beneficiary, with follow-up for the duration of their eligibility for TRICARE. Only spouses of active-duty service members (whether men or women) are eligible for TRICARE benefits; unmarried partners are not. Special attention and outreach will be needed to identify and recruit the unmarried partners of service members whenever possible. Should a partner for some reason not receive health care through MHS/TRICARE, he or she could be encouraged to provide the necessary information and biological samples. The committee acknowledges that it might be difficult to maintain long-term contact with partners outside the MHS, particularly if the partner is divorced from the service member or is otherwise not covered by the MHS. The need to recruit and maintain partners
in the program reinforces the importance of an effective communication strategy that emphasizes the health and well-being of children and future generations.
It will be necessary to develop approaches to identifying and following the nonmilitary parents of children of military personnel over time, regardless of divorce and death, in order to determine preconception and prenatal exposures and to assess such risk factors as lifestyle, occupation, and nutrition that might be related to developmental and health effects in children. After the children reach the legal age of consent, they would provide their own information, as discussed below.
Children and Grandchildren
As the overarching goal of any veteran HMRP focused on generational health effects is to determine if those effects occur in the descendants of active-duty service members potentially exposed to toxicants during deployment, it is critical to follow these descendants for at least two generations—that is, at least through the service members’ children and grandchildren. Recruiting children may begin with those who are born to active-duty service members and covered under TRICARE, which would facilitate access to each child’s EHR. For example, DoD already has the DoD Birth and Infant Health Registry that may be used to access to these records, as discussed later in the chapter. For the small number of children who have an active-duty parent and who are not covered by TRICARE, DoD should encourage those parents to enroll their children in the HMRP. Additionally, there is a need for VA to identify and recruit children born to veterans after they have separated from the military, regardless of whether the veteran is enrolled in VA services.
The committee notes that birth and health information on an active-duty service member’s children, including those born before and after the service member was deployed, will be captured in the TRICARE system. Information on any children born before a service member is deployed would provide a comparison group for any children born after deployment (similar to a case cross-over study design). The HMRP would need to consider that some service members have multiple deployments and that they may conceive one or more children before and after each deployment (and possibly during deployment). Any one of these scenarios would complicate associating exposures across deployments with potential health outcomes in their children: that is, it may be difficult to link a developmental effect to a deployment exposure immediately prior to conception rather than an earlier exposure. Multiple deployments argue for collecting biospecimens from service members before and immediately after deployment to help make such distinctions.
The Volume 11 committee acknowledges that monitoring the health of veterans’ children and grandchildren will be difficult. VA (2018a) estimates that there are more than 6 million veteran households with dependent children (and more than 13 million without children). Veterans may elect to continue to provide health information to the HMRP on minor children. However, once children are legally of age, this is no longer possible unless the child or grandchild willingly consents to participate in the HMRP; the adult child may also need to provide consent for the use of previous data and samples collected while he or she was a minor. Thus, continuing encouragement for service members and veterans and their adolescent children to remain in the HMRP will be critical to collecting the data necessary to understand the health effects that may occur as these children grow from infancy through childhood and adolescence and then into adults. Having veterans’ children enroll their own children (i.e., the veterans’ grandchildren) in the program will be necessary for determining if any adverse health effects are seen in the third generation. For the grandchildren, collecting data (e.g., fertility or pregnancy history and family medical history) from partners who are not biologically related to the veteran would potentially
require consent. This would add an additional layer of complexity. Some key considerations in recruiting children into the HMRP are:
- What is the expected number of pregnancies among the male and female registry participants from the time of enrollment until the end of reproductive years? If 10,000 active service personnel were enrolled, what is the reasonable number of pregnancies expected over the next two decades?
- How many children or grandchildren will be required in a birth cohort to provide meaningful data on generational studies? This is dependent on the endpoints of concern and, in particular, on the incidence rates in the population and on retention rates. For example, childhood cancer would require a much larger cohort than an examination of attention deficit–hyperactivity disorder (ADHD) or even autism. Once the number of births projected from the enrolled cohort is determined, what is a reasonable estimate of the number of children who can be monitored prospectively into the future?
- What are the appropriate informed consent strategies and data boundaries during the lifecourse of each participant?
- What are the best approaches for maintaining consent as children age from minors to adults?
- What strategies should be developed for recruiting and gaining consent from adult children and their children after the parent is no longer an active-duty service member?
Sampling of Study Subjects
Sampling is used as a selection process to identify a representative population of the target group because costs can limit the feasibility of carrying out a hypothesis-driven research study in the entire target population. The selection of the study population and comparison population is an important consideration in an epidemiologic study. Different sampling schemes can be used, including a simple random sample, systematic sampling, and stratified sampling. For systematic sampling, individuals are selected from a list at repeated intervals, while for stratified sampling, individuals are first divided into subgroups and then sampled. These methods can often be used to ensure that the study population includes adequate numbers of people or subgroups of people to answer specific health outcome questions. Other methods of sampling include convenience sampling, clustered sampling, snowball sampling, and quota sampling.
Sampling should be done carefully because an improper sampling of a study population can result in selection bias. Selection bias means that the probability of an individual’s participation in the study is associated with both the exposure and the outcome. For example, if an investigator sought to obtain a random sample of all military personnel deployed to the Gulf War in 1990, but instead sampled only Air Force personnel, this could result in bias for the particular study under question, as Air Force personnel may have different baseline health/demographic characteristics, exposure levels, exposures to important confounders, and health outcomes than those from other branches of DoD. Biased sampling can lead to biased results in one direction or another (i.e., a false positive or a false negative finding). The tracking of study participants over a period of time should include an analysis of whether the retention rates are nonrandom; if the retention is not random, it can introduce selection bias into the final analysis. For example, if a study of Gulf War veterans was evaluating long-term health effects that were more common in women (e.g., iron deficiency anemia), but had a loss to follow-up rate in which the losses of men and women veterans differed, then the estimates of an association with a deployment exposure that increased the risk of iron deficiency could be significantly biased. This would also be true if the
exposure itself caused a health effect that reduced subsequent follow-up. A classic example of this is an exposure in pregnancy that results in an increased risk of early-term spontaneous abortion, an outcome which may occur prior to a woman’s awareness of her pregnancy.
Comparison groups may be unexposed military or veteran populations (and their descendants) which would help reduce selection bias for the healthy warrior effect. Whether military, veteran, or civilian, comparison groups should be sampled and surveyed in the same manner (timing and method) as the target population (IOM, 2008). Nevertheless, identifying, recruiting, and retaining HMRP participants will be a substantial task. Researchers need to factor potential loss to follow-up among all HMRP subgroups (e.g., exposed veterans, unexposed, veterans, children) as well as attrition into the determination of how many veterans and their descendants to recruit into the program and recognize that attrition may begin enrollment. Furthermore, there may be phases of the study when loss to follow-up or attrition are more likely to occur such as when service members separate from the military, with the birth of a child, or when a child reaches an age when he or she can decide whether to continue in the program. Consideration also needs to be given to appropriate sampling of minority groups such as women, blacks, Hispanics, and other relevant subpopulations.
Minimizing attrition will be an important aspect of the HMRP. As noted in the later section on Communication, attrition may be reduced by collecting, retaining, and updating participating contact information, sending reminders via the participant’s preferred method (email, texts, telephone calls) about follow-up activities, building rapport between participants and researchers, and sharing study findings or new activities with participants on an ongoing basis (Buka et al., 2018).
DATA COLLECTION
Integral to any HMRP is the collection, processing, and maintenance of data from all the participants. An HMRP should be designed to collect data in three key dimensions:
- biological samples to include a person’s genome, epigenome, proteome, microbiome, physiology, metabolome/exposome, and health status;
- environmental factors to include exposures to biological, physical, and chemical agents in air, water, and soil; and
- personal and social factors to include occupations, lifestyle habits, education, life events, and neighborhood.
Together these factors constitute the foundation for understanding the impact of potential deployment exposures associated with service on the health of the individual and of his or her descendants. Therefore, collecting data on each of these components is integral to a successful HMRP or epidemiologic study.
The committee encourages HMRP managers to standardize reporting mechanisms, whether for biospecimens or questionnaires and surveys, and to include the resulting information in the individuals’ EHRs in order to minimize the reporting burden on both veterans and health care providers as well as to ensure that research can be conducted in a valid way when the HMRP is used as a source of research study populations. The committee notes that an important consideration for any large-scale data collection effort on health effects is a process for validating self-reported health effects and exposure information. Although EHRs are a good source of medical diagnoses, they can be subject to errors in diagnosis and data entry; therefore, an HMRP should include disease diagnosis verification on subsets of participants.
The frequency of monitoring should be increased if exposure to deployment toxicants is suspected on the basis of environmental sampling, and biological samples (e.g., blood, urine, semen) should be
collected as close to the time of exposure as possible or at the time of anticipated physiological impact. For women, collecting data after their childbearing years may not be necessary to identify generational effects in descendants. However, data collection for the adult descendants of deployed men and women will be important for understanding developmental impacts on adult disease. The committee envisions the HMRP as providing an opportunity to collect biological, personal, and environmental data for veterans of all eras, which, combined with data from partners, and descendants, could be used to establish a longitudinal program to evaluate transgenerational inheritance.
Electronic Health Records and Other Data Sources
An advantage of the DoD MHS is that some of the critical information that needs to be collected is already found in the EHRs of the service members and, for the most part, of the partners and children who are beneficiaries while the service member is on active duty. The anticipated synchronization of the DoD and VA EHRs may also make it easier to capture health effects for veterans who receive care from VA after they separate from the military.
A service member’s EHR can be used to track his or her health from the time of entry into the military until separation. As such, the EHR is a valuable resource for health monitoring which provides baseline data and health effects that may be recorded before (each service member must have a physical prior to deployment) and after a service member deploys. Information from the EHR may also indicate if the service member has reproductive issues before or after deployment as well as how many children the service member had prior to entry into the military and during service.
Numerous data sources for veterans may be accessed as part of the HMRP. These data sources include the DMSS for active-duty service members and the VA EHR for veterans who are separated from the military and are in the VA health care system. The DoD DMDC provides administrative and logistical data (including deployment locations). At present, little information on reproductive effects is collected for male veterans, although male reproductive history is often a bellwether of disease.
VA is encouraged to expand the reproductive effects information that it collects on the veterans in its system. In addition to reproductive effects in male veterans, the EHR should also capture information on pregnancy outcomes or time to pregnancy in the male veterans’ partners as well as information on health effects in their children. Information should also be collected on the maternity care that female veterans receive from non-VA providers, including those who are covered by VA benefits if they are eligible for them. This pregnancy health information should be collected from the veteran’s EHR since VA tracks this information. Time to pregnancy information could also be helpful if collected in a systematic manner for both men and women. The committee notes that veteran status is not necessarily captured in the medical records of private health care providers, and even if veteran status is noted, the era in which the veteran served and deployment status may not be included. There is a need to recruit and retain non-VA veterans by partnering with private health care providers (e.g., participants in the VA Choice Program) since if only VA health care users were included in a study, selection bias would be a major concern (e.g., biases in age, veteran era, socioeconomic status, and health status).
With regard to children, particularly those born in the TRICARE system, an EHR can provide data on birth outcomes and health status. Many of these data will be contained in the child’s medical record, but some health effects may not be captured, such as behavioral and cognitive deficits. Thus, an in-person assessment of the health and development of children and grandchildren should occur at least once during each life stage (e.g., infancy, early childhood, middle childhood, adolescence), and then every 5–10 years or so in adulthood. During these visits, children or their parents (depending on age of the child) might be interviewed, and the children could undergo a brief medical examination (e.g.,
anthropometry, spirometry, etc.) and a neurodevelopmental assessment (e.g., computerized assessments such as NIH toolbox are possible), with a biospecimen collection as well.
Medical Data Linkages
At present, the medical data linkages between DoD and VA are minimal, largely due to lack of interoperability between the two departments’ IT platforms. The DoD EHR is a clinical information management system that provides access to health care records for MHS beneficiaries. Since 2004, DoD has used the Armed Forces Health Longitudinal Technology Application (AHLTA) system for its EHRs.
In contrast, VA uses the Veterans Information Systems and Technology Architecture (VISTA) as its nationwide system for EHRs. VISTA includes more than 180 applications for clinical, financial, and administrative functions within a single integrated database. Of special note is that in June 2017, the Secretary of VA announced that VISTA will be subsumed into the AHLTA system.
The integration of the DoD and VA EHR systems, once it has occurred, may help address data gaps. The Interagency Clinical Informatics Board has endorsed prioritized data domains to support the continuity of care, but to date several domains have no structure to map the data that will be required for the migration of clinical notes stored in the legacy systems. A 2022 completion date has been predicted for the availability of an interoperable DoD and VA EHR system, but the degree to which the DoD/VA system will be interoperable with the EHR systems used by the private sector remains a major unknown.
In order to develop rapid and convenient monitoring of the health of military personnel and veterans that uses real-time hospital discharge or medical data that can be linked with deployment or exposure information captured within the theater of operation, the MHS and VA health care systems must be appropriately linked. Such a link will allow health care professionals and researchers to track veterans longitudinally and to report results in a timely manner.
The Defense Occupational and Environmental Health Readiness System (DOEHRS), a database system managed by the Defense Health Services Systems, includes data in four different categories: industrial hygiene, environmental health, radiation, and general incident reporting. DOEHRS is intended for use by both garrison and deployed operations as the system of record for the individual longitudinal exposure record (ILER). However, as of 2015 DoD did not expect the ILER system to be operative for at least 6 to 8 years (GAO, 2015). For the immediate future, since personalized exposures are not yet available, ILERs will be based primarily on time-weighted average exposure concentrations for a large geographical area. At present the exposure data appears to be limited to chemicals or materials that have been predetermined to be of importance. Until new technologies are developed, this will preclude the identification of potential new threats. Once the system is operational, a veteran’s ILER should be available to DoD and VA researchers and help them link veterans’ exposures to their symptoms.
More than 50% of military personnel do not enter the VA health care system after they leave the military; instead they enter the private health care system or CMS or else they have no health care. Although HMRP participants may sign medical release forms that allow researchers to access their health care records from multiple private health care providers, the data may be in a format that cannot be efficiently handled, such as existing in different EHR operating systems or as hard copies of test results or as large hospitalization records that to date are available only as paper; the latter may be particularly true for older records, such as health records from the Vietnam War era. The fragmentation of the private health care system in the United States adds to the complexity of designing health monitoring systems for service members and veterans.
HMRP managers will face several hurtles in trying to establish reliable and comprehensive IT systems for collecting and analyzing health data on service members and veterans; including their children and grandchildren will offer even more challenges. Harmonizing or integrating the databases will be
difficult if they are incomplete or use coding systems that deviate significantly from those used for data acquisition (e.g., the use of International Classification of Diseases [ICD] codes that may not be useful for effects other than diagnoses). It will be necessary to develop database ontologies to harmonize data from different sources in which the same variable may have different names (e.g., sex in one database, gender in another). IT efforts to overcome these challenges to ensure interoperability and that the data are accessible and protected will be critical. As noted, newer medical informatics methods for mining the EHR, including natural language processing, have been shown to be superior to the use of ICD codes in retrieving data from the EHR (Wang et al., 2017).
Surveys
Both DoD and VA have experience using surveys to collect both health status and personal and socioeconomic information from veterans and, in some cases, from their partners. DoD requires that all service members complete a pre-deployment health assessment and as well as post-deployment health assessment and reassessment questionnaires. VA has conducted numerous studies of veterans and of their use of VA services, including the National Survey of Veterans to ascertain who uses VA health care and other services and the 2016 Survey of Veteran Enrollees’ Health and Use of Health Care (Huang et al., 2017). VA has also conducted three surveys for the Longitudinal Health Study of Gulf War Era Veterans (in 1995, 2005, and 2012), and in 2016 VA carried out the Vietnam Era Health Retrospective Observational Study (VE-HEROeS), a follow-up study to the 1988 National Vietnam Veterans’ Readjustment Study. VA has also conducted separate surveys of women veterans. Several of these surveys have relied on paper documentation, which included the use of mailed invitation letters and surveys to collect participant-specific data. Such an approach is expensive, time-consuming, and inefficient, and generally the response rates have been low (Dursa et al., 2016; NASEM, 2016). VA is moving toward greater use of electronic survey instruments and has used online surveys for epidemiologic studies such as the 2012 Follow-up Study of a National Cohort of Gulf War and Gulf Era Veterans (VA, 2016).
To follow veterans after their separation from active service, it will be necessary to design a systematic process for ongoing data collection from these veterans. The process might include regular, periodic surveys of veterans’ and descendants’ health status along with permission to access medical records in the event of reported adverse health effects. The committee recognizes that regardless of which veteran cohort is monitored, data collection after a veteran separates from the military may provide a challenge because veterans may receive health care in different places, including DoD, VA, private health care providers, and government providers (e.g., state or local health clinics), or they may not be receiving care at all. Veterans who do receive health care could provide a biological sample at a VA or other medical facility or by mail. Questionnaires could be supplied to primary care physicians (from DoD, VA, or elsewhere), or they may be made available online for the veteran to complete.
Although VA has collected data on veterans’ partners, it has not traditionally collected information on the health of veterans’ children. The information necessary for an HMRP that assesses generational effects may extend beyond a child’s disease status to areas that may not be captured in the health record, such as educational attainment and behavioral concerns. Therefore, other approaches and tools may be required to capture different aspects of children’s health outcomes, such as questionnaires, environmental and geographic data collection, and long-term follow-up. The committee encourages VA, in its efforts to capture information on children’s health, to collaborate with other organizations, such as the National Institute of Child Health and Human Development, which have more experience with questionnaires and surveys of children and their parents.
New methodologies and efficiencies are available for identifying the population to be sampled, for the issuance of invitations, for the what and how of data collection and analysis, for the use of incentives for recruiting and retaining participants, and for the use of reminders to increase response rates (Toledano et al., 2015). The use of social media may be a novel way to capture cognitive health via games and tests administered via cell phones or other devices as they are developed. Standardized questionnaires will facilitate comparisons across populations.
Biorepository
The utility of DoD and VA biobanks was reviewed in special supplemental issues of the Journal of Occupational and Environmental Medicine (Vol. 5, No. 7S, July 2016) and Military Medicine (Vol. 180, No. 10, October 2015), both of which described a series of pilot studies that used biological samples for exposure assessments. The biological archives consist primarily of serum collections that have been cross-referenced with other samples to maximize the amount of information associated with any one individual as well as with other datasets (such as DoD Serum Repository, DMDC, and EHR). Of note is that current storage conditions may lessen the potential use of these biological samples in future genomic and epigenomic research.
At a minimum, blood, urine, and semen2 samples should be collected at entry into the military, prior to each deployment, and after each deployment with the optional collection of saliva and hair. Upon separation from the military, veterans participating in the health monitoring system could be requested to provide additional samples, including hair and saliva, depending on the research questions that are being addressed. The expanded use of omic technologies as they become available may enhance exposure assessment, even those that occur during deployment (Bradburne et al., 2015).
Sample collection from service members should occur at specified intervals from enlistment to separation, perhaps during service members’ annual physical (see Table 9-2). Blood samples are now collected by DoD at deployment and then serially every 2 years as part of an HIV screening program; the samples are stored in the DoD Serum Repository.
There is precedent for VA collecting biospecimens from veterans. The VA MVP is assembling a well-curated collection of DNA and other samples paired with health and exposure information (VA and DoD Resources for Study of Generational Health Effects). Veterans using the VA health care system complete surveys about their health and health-related behavior, provide a blood sample, allow researchers to access their VA and VA-linked medical records, and agree to future contact. To date, more than 625,000 veterans have participated in the program (see Leveraging Existing Resources), although complete data are available on fewer than half of them.
It should be noted that a “blood” collection can refer to three types of specimens: whole blood, serum, and plasma. Different types of biomarkers can be measured in each, and ideally all three would be collected.
The biospecimens required to study generational effects should be collected from preconception to birth. The child’s health and development should then be monitored postnatally as part of the yearly wellness assessment to at least year 5, at which point the child will generally enter the school system. Health assessments and biological sampling of the child should occur with subsequent follow-up. At a minimum, the follow-up should include visits to a doctor at each life stage (school age, preadolescence, post pubertal and late adolescence, and even into adult life). To capture prenatal exposures, samples
___________________
2 The committee notes that the collection of oocytes from female service members would also be appropriate whenever semen is collected from men; however, it may not be practical or acceptable to do so in most military situations, e.g., prior to deployment.
TABLE 9-2 Veteran Data and Biospecimens to Be Collected for an HMRP
| Pre-Deployment | During Deployment | Post-Deployment (days, months, years following deployment) |
|---|---|---|
| Military Exposures | ||
| Biomarkers (e.g., blood, urine, semen) | Frequent and consistent exposure assessments of the environment, which may include the collection of additional biospecimens and additional sampling during or following an incident such as the release of chemical warfare agents | Biomarkers (e.g., blood, urine, semen) |
| Documentation of vaccines | Documentation of vaccines | Documentation of vaccines |
| Infections (vector borne, bacterial, or viral) | Infections (vector borne, bacterial, or viral) | Infections (vector borne, bacterial, or viral) |
| Military occupation specialty and any employment prior to joining military | Military occupation specialty | Military occupation specialty and other employment after separation |
| Exposures Over the Lifecourse | ||
| Dietary and lifestyle data (e.g., smoking, alcohol, use of dietary supplements) | Dietary and lifestyle data (e.g., smoking, alcohol, use of dietary supplements) | Dietary and lifestyle data (e.g., smoking, alcohol, use of dietary supplements) |
| Measures of mental health (e.g., cognitive issues, depression, anxiety, PTSD) | Measures of mental health (e.g., cognitiv issues, depression, anxiety, PTSD) | e Measures of mental health (e.g., cognitive issues, depression, anxiety, PTSD) |
| Health Effects | ||
| Comprehensive assessments of health status (vital signs, weight) | Comprehensive assessments of health status (vital signs, weight) | Comprehensive assessments of health status (vital signs, weight) |
| Reproductive status and history | Reproductive status and history | Reproductive status history |
| Socioeconomic Factors | ||
| Demographic data, marital status, education, number and gender of children | Changes to socioeconomic factors (e.g., birth of a child, divorce) | Demographic data, marital status, education, number and gender of children |
NOTE: PTSD=posttraumatic stress disorder.
from women should be collected within 5 weeks after conception and upon the child’s birth or as soon as possible thereafter. The obstacles to blood draws, saliva, and even urine collection in very young children need to be considered. Capturing as many life stages as possible would enable researchers to encompass all the periods in which an immediate generational response may present, for example, within the first trimester when most birth defects appear and potentially are diagnosed.
Preconception samples from veterans may also be available from the DoD Serum Repository, but these could be supplemented with samples from those couples who seek fertility care. Additional opportunistic samples that are indicative of early development may be available when assisted reproductive technology (in vitro fertilization) is used.
Exposure Assessment
The primary reason to distinguish among veteran cohorts is that deployment exposures and health outcomes or status may be vastly different from one cohort to another. For example, many Vietnam veterans were exposed to a number of herbicides that included the contaminant 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD, or dioxin), although there is little information on the actual exposures that veterans experienced during the war, and these herbicides were not used in subsequent conflicts. Veterans of the 1990–1991 Gulf War and the Post-9/11 conflicts in Afghanistan and Iraq had different exposures, which included severe dust storms, burn pits, and chemical warfare agents (e.g., sarin at the Khamisiyah munitions depot in Iraq, and for Gulf War veterans, burning oil wells in Kuwait).
Deployment and occupational exposures may result in or contribute to a biological response and the manifestation of adverse health effects in not only the veterans but also their descendants. Exposure to stressors—nutritional, psychological, physical, radiological, and chemical—during the preconception period and in utero may affect the health of descendants. In addition to the more typical stressors associated with veterans, the stressors may also include vaccines, infectious diseases, smoking, and drug and alcohol use.
Currently, one major limitation of conducting a study using available data on veterans is the lack of adequate exposure information. Validated individual measures of exposure are not available for veterans of any era, and estimates of exposure often rely on self-reported information or military records (e.g., whether a veteran was deployed to a base that had an operating burn pit, or associating exposures with a specific military occupational specialty code). Few environmental sampling studies have been conducted (e.g., see IOM, 2011). The Volume 11 committee recognizes that it may be impossible to determine veterans’ deployment exposures retroactively. Therefore, to better identify deployment exposures in the future, two different types of studies might be helpful. One series of studies would describe the quality (i.e., reliability and validity) of both retrospective and prospective exposure assessment data available for the toxicants of interest for a given veteran cohort—for instance, air sampling at burn pits for Post-9/11 veterans—and make recommendations about the use of such data in future studies. This type of study might demonstrate that some exposures are measured so poorly that future studies of these exposures would be of questionable value. In such situations, cohort studies of deployed versus nondeployed veterans may be the best assessment of exposure. A second series of studies could identify the extent and combinations of exposure to toxicants of interest. Such descriptive epidemiology studies of exposure prevalence, timing, frequency, and combinations might inform future studies to identify mixture effects among veterans in different exposure scenarios.
Exposure misclassification is possible when relying on self-reports of exposure or when measurements of exposure are not available. For example, Gulf War veterans who were originally determined to have been exposed to chemical warfare agents at the Khamisiyah munitions depot in Iraq on the basis of
environmental modeling were later reclassified as unexposed when the model was corrected. Misclassification may also occur if a veteran is unaware of what toxicants he or she was exposed to in a certain situation, such as what materials were being disposed of in a burn pit. Exposure misclassification may be reduced by using more than one source of information to assess exposure, such as environmental sampling and an inventory of toxicants that were used. As noted in the 2011 National Academies (IOM, 2011) report on the health effects associated with exposure to burn pits
Compared with personal monitoring, use of environmental concentrations loses information because personnel at a given site may experience varied levels of exposure based on their activities, personal characteristics, and day-to-day differences in ambient pollutant concentrations; use of environmental concentrations assumes personnel are exposed to the average exposure at each site. Any approach that blurs the distinction between individual exposures while maintaining the collection of individual health outcomes will reduce the estimated variation in exposure and lead to exposure misclassification of some individuals. These issues bias the study, increasing the chance that any association between exposure and health outcome will not be detected.
Role of Partners and Descendants in an HMRP
Partners
The participation of partners will be critical to the success of an HMRP designed to study health outcomes in the children, grandchildren, and future generations of veterans. To monitor the effects of preconception or prenatal exposures, it will be essential for the HMRP to also enroll the partners (men and women) of the service members, whether active-duty or veterans. Biological samples from male partners of study participants that may be used to assess the genetic and chemical risk factors for reproductive and developmental effects include blood and urine samples, which can be linked to the timing of other required pregnancy specimen collections, and a semen sample when pregnancy is first suspected or, at the latest, during the first month of pregnancy. From female partners, the specimens to be collected include blood and urine and cord blood obtained at the time of birth and a heel stick (blood spot) sample from the newborn, which is collected as part of all newborn screening programs. Other information to be collected from partners includes the same lifecourse exposure data collected for service members and veterans, occupational and recreational/environmental exposures, a comprehensive assessment of health status (e.g., vital signs, weight, presence of disease), and a reproductive history.
Optimally, if both parents are members of the military, their health records will be in the MHS, and both will agree to the use of their health information for the monitoring program. Although the DoD EHRs may include health information for partners if they are beneficiaries, it may be difficult to obtain health information for those individuals who do not use TRICARE. Issues of consent may also arise if the parents are not married, get divorced while in TRICARE, or otherwise have objections to the HMRP.
Descendants
Obtaining health effects information from the children of service members and veterans will be challenging, but various approaches may be used to reduce the burden of sample collection. Data collection will depend on whether a parent is active-duty, enrolled in VA, or a civilian in another health care system. Medical data for children who are TRICARE beneficiaries will be available through the MHS until they are no longer eligible for TRICARE, either because they are no longer dependents of a military parent or because their parents have separated from the military. The TRICARE system can
provide information on a child’s birth and health as long as the child is covered. Should a child leave the TRICARE system, further monitoring will be challenging, especially if the child receives health care from private providers. The child/adult would need to authorize the release of his or her medical information. With the proper permission, health information from pediatric visits might be obtained.
In addition to retrieving the information from a child’s medical record, it will also be useful to collect and store a child’s blood spot taken at birth, which can be used for genomic testing and other omics testing. Deciduous, or “baby,” teeth might also be collected from those children ages 5–12 years who were in utero at the time of maternal deployment in order to reconstruct exposures in pregnancy. New technologies have been developed that can reconstruct certain chemical exposures (e.g., metals) experienced in pregnancy and early childhood from deciduous teeth (Andra et al., 2016). Table 9-3 identifies the data and biospecimens that should be collected for an HMRP from children and grandchildren.
Several health outcomes should be assessed for veterans’ children—specifically, prenatal and neonatal health (e.g., fetal growth, preterm birth, birth defects, sex ratio); neurodevelopment; growth (e.g., weight, height, head and waist circumference); metabolic, endocrine (e.g., thyroid hormone), and immunologic function; respiratory health; reproductive health (e.g., age of onset of puberty and menopause, fertility); behavioral health (e.g., depression, anxiety, and other mental health disorders), and chronic disease in childhood and adulthood (e.g., cardiovascular and metabolic diseases, cancer). Many of these endpoints are routinely collected as part of standard medical care. For example, it is usual for states to conduct a neonatal screen for thyroid-stimulating hormone, in which case it is important to ensure that the authorities record the actual level and time of postnatal collection and not just whether the level was normal. Grandchildren should also be assessed for these outcomes, if possible. For practical purposes, the diseases of childhood will be easier to study than the adult-onset diseases in any generational research
TABLE 9-3 Descendant Data and Biospecimens to Be Collected for an HMRP
| Birth | Childhood | Adolescence | Adulthood |
|---|---|---|---|
| Growth (e.g., weight, height, head circumference) Anogenital distance Vital signs (e.g., Apgar) Biomarkers (e.g., cord blood, blood spot) State mandated neonatal screen Feeding (e.g., breastfeeding) |
Growth (e.g., weight, height, waist circumference) Vital signs (e.g., blood pressure) Neurodevelopment Biomarkers (e.g., blood, urine, hair) Baby teeth Behavioral health Physical health and chronic illnesses School performance Physical activity and diet |
Growth (e.g., weight, height, waist circumference) Vital signs (e.g., blood pressure) Neurodevelopment Biomarkers (e.g., blood, urine, hair) Onset of puberty Other reproductive status Physical health and chronic illnesses Lifestyle (e.g., smoking, drugs) and diet Behavioral health School performance |
Growth (e.g., weight, height, waist circumference) Vital signs (e.g., blood pressure) Neurodevelopment Biomarkers (e.g., blood, urine, hair) Semen/oocytes Reproductive status (fertility, time to pregnancy, number of children, etc.) Physical health and chronic illnesses Lifestyle (e.g., smoking, drugs, diet, physical activity) Behavioral health Education and socioeconomic information |
since waiting for the F1 or F2 generation to age into adulthood will delay the results. In addition, the field is still too young to clearly delineate which health outcomes are likely to be generationally transmitted.
Veteran subpopulations, such as children born to women who became pregnant while deployed or to veterans who had specific hazardous duties or exposures, should be studied more extensively. Collections of data on pregnancy complications (e.g., gestational diabetes and hypertension disorders of pregnancy such as preeclampsia) and birth outcomes (e.g., low birth weight, preterm birth, birth defects, sex ratio, thyroid levels) will be important. The grandchildren of the veterans should also be followed in order to ascertain their health and development.
DoD currently collects data on all births covered by TRICARE. For VA, it might be possible to capture pregnancies and birth with periodic surveys of veterans. In addition to the blood spot that the committee suggests be collected at birth and be maintained by the states’ newborn screening programs, and in addition to any project-specific aliquots, other additional biological samples (e.g., saliva or meconium collected from offspring) would depend on the specific goals and questions asked. Other data resources for children’s health, such as state and DoD registries of birth defects and cancer registries and also national vital statistics for birth and death certificates, should be considered.
DATA ANALYSES
Studies looking for associations between veterans’ exposures and specific outcomes in those veterans and their children should be focused on and driven by exposures that were incurred during deployment (suspected or measured), by the emerging literature on genetics and epigenetics, and by the reproductive and developmental effects identified in Chapters 4 through 7. The analytic techniques that can be used in assessing generational health outcomes include epidemiologic studies and experiments with animal and cellular models; these techniques are briefly described below.
It is essential that the analysis include certain critical elements: proper adjustments for confounders; consideration of mediators and modifiers; identification of appropriate comparison groups; timeliness of the analysis and its reporting; and making the data used in the analysis accessible to outside researchers for their evaluation and further analysis. Each of these elements is discussed below.
The amount of information that will be collected for such an analysis will be substantial, and it will continue to grow as veterans and their descendants age. Current statistical methodology and computing resources permit the screening of a large number of health outcomes in order to determine whether particular diseases are over-represented in the descendants of veterans.
VA or other organizations maintaining the HMRP will need a systematic process for analyzing the data and disseminating results. Technical personnel should regularly review the data to ensure its quality and optimize its effectiveness. VA, DoD, and other collaborators should work together to standardize the data-reporting tools and the analytic programs to be used by each organization to collect, store, and present the information. HMRP data should be analyzed and the results should be reported on a regular basis to aid in the detection of even subtle changes in health over time. For example, DoD regularly analyzes DMSS data and reports on various health statistics of service members in its Medical Surveillance Monthly Report.
Where HMRP data suggest an association, more in-depth investigations in the form of hypothesis-driven epidemiologic studies should be considered to verify or further explore the association. Animal or cellular models may also be used to examine the potential mechanisms and biological underpinnings of the findings.
The data collected as part of the HMRP should be made available for extramural research, but it will also be crucial to include appropriate human subject protection as part of the standardized data
access; this can be done by making the data available through such resources as dbGAP, a NIH database developed to archive and distribute the data and results from human studies on genotype and phenotype interactions. In addition, the NIEHS Children’s Health Exposure Analysis Resource (CHEAR) seeks to provide the “extramural research community access to laboratory and data analyses that add or expand the inclusion of environmental exposures in children’s health research” (NIEHS, 2018) (see Box 9-2). Allowing outside investigators to address additional research questions makes the task of conducting a myriad of potential analyses much more manageable. Analyses by outside investigators may target areas of research not yet prioritized by VA, such as rare diseases. The results from such analyses may help redirect the priority health outcomes selected; the methods used to assess exposures, outcomes, or confounders; and additional research priorities for VA and DoD.
Biospecimen and Environmental Analyses
Blood must be processed shortly after collection in the case of both serum and plasma, as freezing a whole blood sample and processing later would destroy the integrity of the serum/plasma due to hemolysis. Infrastructure to process samples shortly after collection will be needed. Spot cards may be an alternative for some measures, but their applicability is not universal.
The committee notes, however, that sera may not be adequate for detecting many of the biomarkers of exposure, response, and susceptibility. Some chemicals or their metabolites are best measured in different biological matrices. For example, exposure to chlorpyrifos might be determined by measuring its metabolite 3,5,6-trichloro-2-pyridinol in urine, whereas organochlorine pesticides such as lindane
would best be measured in serum and dioxins in adipose tissue. Thus, it is important that at least blood and urine be collected for each veteran; blood samples alone will not be sufficient for some toxicants.
A further consideration in biospecimens analysis is the need to conserve individual specimens while still being able to use them for research assays. One approach to this is to use specimen pooling. In case-control studies, cases and controls may be separately pooled to permit an estimation of individual-level exposure effect using modified logistic regression analysis. This approach allows the study of biomarkers while reducing assay costs. Saha-Chaudhuri and Weinberg (2013) have demonstrated how pooling specimens can also be used to screen for discrete exposure effects such as time to pregnancy, although they acknowledge that the approach may produce slightly biased odds ratios. Pooling also helps permit sampling when individual samples may be at or below the level of detection, which may be particularly important when assessing veteran biospecimens for toxicants such as dioxins, although this approach is not recommended for use when more than half of the samples are below the level of detection (Mumford et al., 2006). Schisterman and Vexler (2008) have proposed a combined design, which uses pooled and unpooled biospecimens to capture the strengths of the different sampling strategies and overcome limitations such as levels of detection.
Driven by such issues, the sample-processing and analytic capabilities used to develop biomarkers are advancing at a rapid pace. The capacity to generate a wealth of data is growing at an exponential rate, and this information is being integrated into various precision medicine initiatives, including the NIH All of Us program. Calls to incorporate exposome analyses (i.e., untargeted chemical screens using high-resolution mass spectrometry) as a discovery approach to understanding deployment exposures have already been made (Accardi et al., 2016; Liu et al., 2016; Walker et al. 2016). These specimens can be used to carry out a comprehensive screen of biological systems.
To identify emerging health problems as well as to pinpoint the possible generational effects associated with various exposures, omics data generated from biospecimens can, in the context of the child’s and the parents’ health status, provide a resource with which to evaluate biomarkers of exposure and mechanisms of action and to develop a personalized response profile. The committee recognizes that the number of complete sets of data on children is likely to be limited, certainly in the early years of any HMRP. But as the program matures, particularly for Post-9/11 veterans and future veterans, these data could provide a robust resource that is well-suited to the growing omics field of personalized and precision medicine.
Because it is not clear which health effects are most likely to be generationally transmitted, a broad spectrum of health effects will need to be assessed. Recent advances in phenomics—the study of all health outcomes—may be adapted in a future HMRP. Hebbring (2014) suggests that phenome-wide association studies that use electronic medical record systems to conduct a genotype-to-phenotype approach when studying human disease may be a complementary approach to genome-wide association studies. Phenomic advances in image analysis, the use of machine learning to process biological lab data, and the use of natural language processing to perform computational searches of the EHR may be key to reducing the number of phenotypes to be studied to a more manageable number.
Regardless of their deployment or occupational status, humans are affected by environmental exposures from countless sources during their lifetimes, including parental exposures that may occur even before the conception of an individual. Accordingly, one must not only consider exposures that occur during deployment, including multiple deployments to the same or different locations, but also take into account how previous or subsequent exposures throughout a lifetime affect a person’s health and that of subsequent generations. Some environmental exposures, such as tobacco use, may cause adverse effects, but many exposures will not—or they may even be beneficial (e.g., vitamins, medicines). As a result, deployment-related exposures cannot be examined in isolation; rather, they must be considered
in the context of the many other exposures that occur over the lifecourse. For example, if the veteran is a smoker, what effect might smoking have on the veteran’s response to an additional exposure such as particulate matter during a dust storm in Iraq? Thus, it becomes difficult to know with certainty whether a health effect can be attributed to an exposure during deployment or to an exposure prior to or after deployment or to a combination of all three. Furthermore, as is evident from prior Gulf War and Health volumes, a toxicant may be associated with multiple health outcomes making it difficult to attribute any given health effect to a single toxicant. For example, polycyclic aromatic hydrocarbons (PAHs) have been associated with reproductive effects in men as well as adverse pregnancy outcomes. Because so little is known about the generational effects of deployment-related exposures, it is difficult to answer questions about how exposures over the lifecourse, including deployment exposures, may affect subsequent generations. However, the use of appropriate epidemiological methods and robust data could assist in determining the associations between potential deployment-related exposures and the health effects across multiple generations.
A well-conducted epidemiologic study will need estimates of environmental exposures during deployment, including during incidents of special concern such as the demolition of the Khamisiyah weapons depot. This will require the use of state-of-the-art exposure assessment methods. For example, recently developed wearable monitoring devices could be employed by either all deployed personnel or to a generalizable subset. Wearable devices are a rapidly evolving field; thus, the program should stay abreast of developments as they occur because improvements over current methods are likely to arise. As observed at Khamisiyah in 1991, researchers need to be aware of the limits of the detection of exposures and where additional methods for detecting exposures should be deployed. This is particularly important for exposures such as Khamisiyah where the exposure was to a chemical warfare agent where air sampling at the time of the demolition or shortly afterward would have helped to determine the extent and severity of the sarin plume and thus the number of service members who were potentially exposed. Such monitoring by DoD can facilitate risk communication and help identify a cohort of exposed individuals. For most veterans, however, exposures will need to be modeled from environmental data, lists of chemicals used on-site, and other geospatial information. Other deployment exposures that should be captured include the use of pesticides (such as permethrin-impregnated uniforms), the application of chemical agent-resistant coatings, and the use of weapons containing depleted uranium. The 2000 NRC report Strategies to Protect the Health of U.S. Deployed Forces: Analytical Framework for Assessing Risks provided a risk assessment framework that might be used by DoD to assess threats to service members by establishing strategic baseline preparations based on military activities (rather than on specific threats), characterizing activities during deployment (including environmental sampling and information on unit activities), and monitoring veterans for effects from those activities after deployment.
Statistical Considerations
Applying the results of a health monitoring effort to the design and implementation of an epidemiological research study requires that the researchers have the appropriate sample size so that the study has sufficient power to detect an effect. Without a sufficiently large population, it is difficult for researchers to know if their study will answer the questions they developed when establishing the goals of the study. The size of the health monitoring effort, the amount of data collected on the specified populations (including comparison groups), and the manner in which exposures are measured or estimated and health outcomes assessed will all contribute to determining the statistical methodology used for the epidemiologic research study, including sample size and power.
Sample Size and Power
Choosing a targeted sample size is an important step in designing a hypothesis-driven epidemiologic study. Sample-size calculations are based on the expected magnitude of the difference between the exposed and unexposed groups, the relative sizes of the groups to be compared, and specified levels for type I error (the error of rejecting the null hypothesis when it is true) and type II error (the error of failing to reject the null hypothesis when the alternative hypothesis is true). To calculate the minimum required sample size, the number of subjects per group must be multiplied by the number of groups used for comparison (IOM, 2008). Although not specific to a reproductive or developmental effect, a prior Institute of Medicine (IOM) committee (IOM, 2008) did calculate sample sizes for a statistically significant difference in mean serum creatinine concentration, a biomarker for renal injury, between depleted uranium-exposed and -unexposed subjects, setting a type I error (that is a false positive) at 5% and the power at 80%. To detect a risk ratio of 1.50, approximately 9,000 participants (1,760 exposed and 7,038 unexposed) would be required.
A sample size that is too small will not provide estimates that are precise enough to reliably answer the questions of interest, whereas a sample size that is too large may not make optimal use of time and resources. Sample size is often chosen on the basis of a power calculation. The power of a study is the probability that it can detect an association between an exposure and an outcome if there really is such an association. If the statistical power is high, the probability of concluding that there is no association when in fact such an association does exist goes down. In clinical trials, a power of at least 80% is typically required; however, this threshold should depend on context. Knowing the power of a study helps with the interpretation of negative results.
To perform a power calculation and use it to determine the sample size needed to achieve a given level of power, three critical inputs are needed: (1) the size of effect that is to be detected (intuitively, to detect a smaller effect with high power, a larger sample size is needed); (2) the variability of the outcome—for example, the standard deviation of the outcome variable in the population (i.e., with higher variability, a larger sample size is needed); and (3) the range of the exposure (i.e., with a smaller range, it is harder to detect effects of the exposure, and a larger sample size is again needed). Reviews of power calculations and sample size determination are provided by Hajian-Tilaki (2011), Lenth (2001), and Liu et al. (2005).
In cohort studies, many exposures and outcomes may be of interest, and the power will differ for each exposure/outcome combination. It may then be advisable to assess power calculations for a few representative exposure/outcome combinations. The needed sample size will be larger if the researcher wants to gain information about specific subpopulations, such as those carrying certain genotypes, and for the power calculations to be correct, these subpopulations will need to be defined and fixed before the study is conducted. Thus, it may be advisable to expand targeted sample sizes to compensate for exposure-classification issues, attrition of subjects by dropout, other losses to follow-up, and deaths of veterans and their descendants over the lengthy follow-up period.
When planning a generational cohort study, there are additional considerations for power calculations that will need to be taken into account. Since the initial sample size needs to be determined based on the generation receiving the initial exposure, one needs to consider what the effect of the initial exposure will be on the survival and fecundity of subsequent generations in order to estimate the sample size that will be available for subsequent generations, given the initial generation sample size. The committee is not aware of any substantial research on this topic, and further research would be valuable.
Bias
An important contribution of statistical theory to epidemiology is a method to understand the statistical uncertainty associated with effect size estimates. In particular, this theory provides an approach to computing the expected values and standard errors of estimates. By increasing the sample size, estimates can be made to approach the population quantity being estimated. However, the theory assumes that there is no bias or systematic error in the approach. In the presence of bias, this theory is no longer valid. When an estimate of an effect size is consistently different from the actual effect size in the population of interest, regardless of the sample size, the estimate is said to be biased. For example, consider a survey that seeks to measure the proportion of individuals in a population with hypertension. Due to a flaw in the methodology, older individuals are more likely to be included in the sample. This would result in a biased estimate: no matter how large the sample size for the study, the study would be expected to produce a higher estimate than the real proportion of the population with hypertension.
Reducing bias is therefore an important part of epidemiologic studies. Bias can arise from a number of sources. Below the committee describes some common examples: selection bias, information bias, confounders, mediators, and effect modifiers.
Selection Bias
As noted in Chapter 2 and in the section on Sampling of Study Populations earlier in this chapter, bias can result when study participants are selected in such a way that they do not represent the target population or if the probability of selection is related to exposure or disease status. This may be due to a poor definition of the eligible population or a failure to obtain a random sample. This is an important consideration when surveying veterans or any volunteer population where those who agree to participate may be different from those who decline to participate. For example, VA has determined that male veterans who use VA benefits tend to be older than those who do not (64 versus 57 years of age), whereas among female veterans, VA beneficiaries are slightly younger than non-beneficiaries (46 versus 48 years of age) (VA, 2017b). Furthermore, veterans who are more concerned about their children’s health, possibly because the child has a health problem, may be more inclined to participate in an HMRP than veterans with healthy children (i.e., a volunteer bias). Other types of selection bias include nonresponse bias and the healthy-warrior effect, which is similar to the healthy-worker effect. Such biases can be acknowledged and adjusted for in the statistical analyses of study data.
In some cases, selection bias can be introduced if subject inclusion correlates with disease and exposure. For example, autistic children might be excluded from an analysis that required a blood-based biomarker. Newer technologies—such as blood spot–based biomarkers and volumetric absorptive microsampling (a technology that enables quantitative blood volume collection from a capillary prick)—will reduce, but probably not eliminate, selection bias from biomarker sample collection. Both the MVP (discussed later in the chapter) and the proposed HMRP will collect extensive demographic data on correlates of exposure such as socioeconomic status, location of residence, education levels (see Tables 9-2 and 9-3), and disease. Modern methods of EHR mining, such as machine learning–based methods, will allow more complete covariate and phenotype data to be collected. In all cases when studies are conducted on subsets of cohort subjects, investigators may compare the included versus excluded groups using sensitivity analyses to determine whether there is selection bias. With respect to measurement error, the committee recommends that researchers consult the growing body of literature on applicable measurement corrections (e.g., Bennett et al., 2017; Keogh and White, 2014).
Information Bias
When a study misclassifies the exposure, outcome, or both, it is said that there is information bias. These errors can occur either differentially or non-differentially. For non-differential misclassification, an error in the measurement of the exposure or outcome has occurred evenly across these two groups. With differential misclassification, an error in the measurement has occurred in the exposure group that differs according to the outcome or else an error in measurement has occurred in the outcome that differs between the members of the exposed and unexposed groups. These different types of misclassification result in different forms of bias. Other types of information bias include recall bias and interviewer bias.
Confounders
In epidemiological studies, there are variables that can be associated with both the exposure of interest and the outcome of interest. When not considered in the study, these factors can lead to bias. For example, cigarette smoking could be a confounder of a particular association of interest, such as ambient air pollution and respiratory conditions, because cigarette smoke contains constituents of air pollution and can cause adverse respiratory effects. In this case, if the analysis does not take into account individuals’ cigarette smoking, it can end up overestimating or underestimating the association of interest. Because of this, it is essential that confounders be measured and adjusted for in the statistical models in order to reduce bias and obtain valid estimates of the association of interest. Much work has been dedicated to issues of validity in epidemiologic studies.
Mediators
These variables are often confused with confounders because they are associated both with exposure and with outcome. But unlike confounders, mediators are on the pathway between the exposure and the outcome and are part of the reason the exposure causes the outcome. These variables are therefore not confounders, but rather are considered to be mediators of the exposure-outcome association. Mathematically, they would behave similarly to confounders in a regression analysis. Special statistical methods are used to address mediators. Multiple mediators can be on a given pathway of interest. Information on mediators can be highly informative and can provide key information about the mechanisms involved in a given exposure-outcome association, especially in the context of generational health effects. However, limitations in the power of the analysis or in the data can make it difficult to detect mediators.
Effect Modifiers
Variables that modify the association between an exposure and an outcome of interest are defined as effect modifiers. In biological terminology, synergism and potentiation are two concepts that are similar to effect modification. In effect modification, the association between the exposure and the outcome is amplified in the presence of another variable. For example, it may be hypothesized that posttraumatic stress disorder (PTSD) could be an effect modifier of the effect of ambient PAHs on asthma. In this case, individuals who have PTSD and who are exposed to PAHs are much more likely to develop asthma than individuals who are exposed to PAHs but who do not have PTSD. Because the effect modification says something about the biology underlying the exposure and the outcome, findings are reported stratified by the modifier. Often, effect modification is used synonymously with interaction. In statistical models, interaction terms can account for the presence of effect modification. Effect modification is different
from confounding. It provides important information about potentially vulnerable or resilient populations because it relates to the exposure and outcome associations. To avoid a biased sample, it is necessary to account for such interactions in the data.
Directed acyclic graphs (DAGs) require that the temporal relationship of covariates be considered as well as whether the covariates are confounders or mediators of the association of interest. Over extended periods of time, it may be difficult to determine whether a variable is a fixed confounder or mediator. This is particularly true when considering research questions that involve multiple generations. Even in cases where a shorter period of time is considered for the mediating mechanism, such as maternal exposure during deployment and fetal growth, it is important to note the non-static nature of the exposures and time-varying confounders as they relate to the outcomes of interest. In this example, there may be uncertainty about the impact of confounding on the associations of interest. As such, DAGs should be used with caution in situations with multiple variables.
Appropriate statistical methods should address the temporality of exposure and its potential confounders and mediators as it relates to a given outcome across the lifecourse. As the field of generational health effects develops, it will likely require both current methods, such as the use of DAGs, and new statistical approaches that have yet to be developed (Hernan and Robins, 2018; Pearl et al., 2016). Statistical methods for mediation analysis distinguish between pre-exposure confounders and potential post-exposure mediators. Instead of adjusting for the post-exposure variable as one would for a confounder, the variable is treated as an intermediate outcome in the analysis. Reviews of considerations and methods for mediation analysis and of the analysis of post-exposure variables are provided by Bellavia and Valeri (2018), Rosenbaum and Rubin (1984), Taguri et al. (2018), Ten Have and Joffe (2012), and VanderWeele (2015).
Challenges with Mixtures
As discussed in Chapter 2, a major challenge in assessing the effect of human exposure to a specific chemical agent is that humans are usually exposed to many environmental agents at once and these exposures are often highly correlated (i.e., colinear). Many health effect studies focus on a single agent or else consider simple two-agent interactions. However, the results of such studies can be very misleading when the agent is highly correlated with multiple biologically active agents. An example of this would be the way that exposures to ambient particulate matter may be highly correlated with exposures to NO2 because they are both components of fossil fuel combustion. The complexity of assessing mixtures, particularly poorly defined mixtures such as combustion products and their implications for epidemiologic studies of veterans was discussed in the IOM report on the long-term health effects of exposure to burn pits (IOM, 2011).
Several methods have been developed for dealing with exposures to mixtures. Billionnet et al. (2012) reviewed the statistical methods used to assess the health effects associated with the multiple chemicals that constitute air pollution and other researchers have explored the challenges of assessing exposures to mixtures for epidemiological studies. Braun et al. (2016) acknowledge that measuring exposure to a mixture requires the accurate measurement of the individual components of the mixture, which may be difficult given within-person variability and misclassification of exposures, reverse causality due to pharmacokinetic factors, and the validity of the biomarker for assessing exposure over time. Nevertheless, it may be possible to identify and prioritize important mixtures which pose greater risks or are more common in certain situations (e.g., the mixture of pyridostigmine bromide, permethrin, and DEET that along with stress are used to mimic Gulf War exposures suspected of causing Gulf War illness). The committee recognizes, however, that there are no standard analytical methods for the mixtures that may
be encountered during deployment. Braun et al. (2016) present several methods to quantify the association between individual exposures and health effects and apply those to mixtures, examine interactions between chemicals in a mixture, and assess the cumulative chemical exposures with a health effect by quantifying the “summary effect of a class or multiple classes of exposure.”
New methods and statistical approaches may also be useful when assessing mixtures. Hamra and Buckley (2018) state that the research question to be answered by the assessment of a mixture will drive the parameterization of the exposure variables. The committee encourages VA to follow scientific advances in mixture assessment, including both measuring environmental concentrations of individual components of mixtures as well as new techniques for sampling mixtures themselves and new toxicologic information on the effects of mixtures in animal models (see Chapter 10). Some of the issues with assessing exposure to Gulf War mixtures in animal models of Gulf War illness are reviewed in Volume 10, Chapter 5, Animal Studies (NASEM, 2016).
LEVERAGING EXISTING RESOURCES
Thus far, the committee has laid out the basic elements of an HMRP. These elements may be perceived as establishing a contemporary gold standard for answering questions about generation health effects. But the committee realizes that time and resources are finite, and while it may be possible to implement parts of the proposed health monitoring effort and hypothesis-driven epidemiologic studies, it may be more feasible and expedient to leverage ongoing programs such as the VA MVP. Regardless of the approach used, the utility of the results of any program will hinge on the availability and quality of the data on the veterans’ exposures and of the data on their health and the health of their partners and descendants. In the sections below, the committee provides guidance on how VA and DoD might improve the collection of information to determine possible generational health effects using existing infrastructure. The committee examined a number of ongoing research programs from the perspective of which offered the best opportunity for answering questions about the health of veterans’ descendants. These research programs include the VA-sponsored MVP and the DoD-sponsored Millennium Cohort (MilCo) and MilCo Family Study as well as the DoD Birth and Infant Health Registry.
Million Veteran Program3
The Million Veteran Program is a VA initiative aimed at developing a better understanding of how genes affect health, with the ultimate goal of improving health care for male and female veterans of any era as well as for the general population. The MVP was established in 2011 to serve as a resource for precision medicine by collecting biological samples that may be used to develop prevention interventions, diagnostic tools, and treatments to improve health care. Although the goal of the MVP is to have a representative sample of all veterans who use VA health care across the nation, participation in the program is voluntary and by invitation only (i.e., not a self-selected population). As of October 2017, 49% of the 625,000 veterans enrolled in the MVP were from the 1961–1975 Vietnam era, 23% served from 1975 to 1990, 23% served from 1990 to 2001 (of whom 5% are Gulf War veterans), and 12% have served since 2001 (of whom 6% are Operation Enduring Freedom/Operation Iraqi Freedom veterans). Veterans currently enrolled in the VA health care system complete surveys about their health and health-related behavior, provide a blood sample, allow researchers to access their VA and VA-linked medical
___________________
3 References: https://www.research.va.gov/mvp/veterans.cfm#6; https://www.nih.gov/sites/default/files/research-training/initiatives/pmi/pmi-presentation-million-veteran-program-cohort-example-large-scale-health-system-based-cohort.pdf; and https://www.nihcollaboratory.org/Pages/GR-Slides-09-09-16.pdf.
records, and agree to future contact (VA, 2017c). Data for the MVP can be extracted from VA clinical and administrative databases, including the National Patient Care Database, VA Managerial Cost Accounting National Data Extracts, the Corporate Data Warehouse (Health Data Repository), Medical Domain Web Services, the Patient Care Services Clinical Data Warehouse, the Veteran Informatics and Computing Infrastructure, the Veterans Health Administration Support Service Center, the VA Cancer Registry, and the VA Vital Status File. VA has recently partnered with the Department of Energy (DOE) for the MVP’s Computational Health Analytics for Medical Precision to Improve Outcomes (CHAMPION) initiative. The goal of the initiative is to bring the Department of Energy’s state-of-the-art expertise and capabilities in a secure computing environment to analyze large amounts of health care and genetic data from VA and other federal sources, beginning with MVP data (VA, 2017c).
From male veterans the MVP questionnaire collects basic information on reproductive health, fertility, and the number of children they have fathered. More detailed information is collected from female veterans, including age at menarche, age at menopause, reproductive health (hysterectomy, ovary removal), and fertility; the use of contraceptives, hormone replacement therapy, and fertility treatments; and details about pregnancies (number, stillbirths and miscarriages, age at first and last births, and breast-feeding). Women are also asked about the birth weight of their first child, but no other data are gathered on the health status of the veterans’ children.4
The only biological specimen that is collected is a blood sample that is processed into plasma, buffy coat, and DNA. Samples are to be stored indefinitely under liquid nitrogen unless the veteran withdraws from the program and the samples are destroyed.
The strengths of the MVP study are its large sample size, the fact that it uses a representative sample of the VA population, its collection of blood samples and establishment of the genetic biorepository, its ability to link together and merge other VA databases, its collection of vital statistics data, and its use of a physical examination. The MVP can be an important resource for the study of inter- and transgenerational effects because participating veterans are willing to be contacted again in order to provide additional information on the course of each generation.
The MVP is the largest existing VA genomic biobank with sufficient data depth to be used to assess genetic susceptibility to certain exposures, particularly those that can be measured in plasma samples, such as exposures to lindane and dioxins. Its DNA resource could be used to assess genome methylation status with the goal of identifying DNA methylation patterns that arise following chemical and other exposures. This is a potential way to reconstruct past exposure such as is currently done with the methylation patterns in the aryl hydrocarbon receptor repressor gene to identify smoking status. As of 2015, genotyping data were available on 199,348 veterans in the program (92% men), of whom 77% were white, 13.5% were black, the remainder were “other” or not reported (Gaziano et al., 2016).
Augmenting the MVP could make possible the study of multiple generations. The committee identified several strategies that could be adopted, including conducting regular participant and offspring follow-up sessions. Information on parental reproductive history, such as time to pregnancy (fecundability) and clinical infertility, might also be collected. The frequency of this follow-up would be determined by logistic and economic factors, but the committee suggests that information on children be collected at least once in every life stage, as outlined in the section on Health Outcome Data on Children and Grandchildren. Data from annual medical visits to primary care providers could be merged with the MVP data. Biological samples (e.g., urine, saliva, and blood) from the children and grandchildren might be collected and stored for future use.
___________________
4 Personal communication from Victor Kalasinsky, Office of Research and Development, VA, October 17, 2017.
Because the MVP does not include veterans who do not use the VA health care system, information on how MVP participants differ from veterans who do not use VA (demographics, deployment, and military characteristics) would add to the usefulness of information on veterans that do use it and would strengthen MVP estimates of health risk for all veterans. The committee is encouraged by the way in which VA and the NIH All of Us program entered into a partnership for leveraging data collection, and the committee expects that this partnership will provide a sound mechanism for helping VA collect data on veterans who do not use the VA health care system and for recruiting those veterans into an HMRP.
If such additions were made to the MVP, the data could be used for a variety of future epidemiologic and other experimental research studies. Because the MVP records can be linked to deployment history and other military exposures, adopting the committee’s recommendation to include enhanced exposure data collected during military service in a veteran’s ILER would allow researchers to conduct nested case-control studies on, for example, the prevalence of a reproductive health outcome in veterans.
Millennium Cohort
In response to concerns about the health effects of deployments following the 1990–1991 Gulf War, Congress and IOM recommended that DoD conduct prospective epidemiological research to evaluate the impact of military exposures, including deployment, on the long-term health outcomes of veterans. DoD responded by establishing the MilCo, the largest prospective health study service ever conducted with members from the Army, Navy, Air Force, Marine Corps, and Coast Guard; at present it has more than 200,000 participants, with more than 31% of them women (Smith, 2009).5 The study is being conducted by the Naval Health Research Center. The eligible population consists of active-duty personnel deployed since 2001 in support of the operations in Iraq and Afghanistan, including reservists and National Guardsmen; participants are followed after they leave the service or are deactivated.
MilCo contains data collected via questionnaires and from DoD databases. No biological specimens are collected, and no physical examinations are conducted. The questionnaires have self-reported outcomes, and participants are asked to complete them every 3 years through 2022. The questionnaire covers the following topics: general health (functional status, including physical and mental; well-being; height and weight); health behaviors (alcohol and tobacco use, eating behaviors/disorders, sleep duration and quality, physical activity); health conditions and symptoms (provider-diagnosed medical conditions); physical symptoms (e.g., pain, headaches, fatigue, shortness of breath, cough, forgetfulness); mental health (PTSD, depression, anxiety); pregnancy and infertility; potential exposures (combat experiences; injuries, including head injuries; motor vehicle accidents; burn pits/smoke; chemicals/pesticides/depleted uranium; assault, sexual and physical; adverse childhood experiences); and military life (deployments; military occupation specialty; perception of the military; status, active duty, reserve/National Guard, separated/retired).
Health data from the questionnaires can be linked to the DMSS and the DMDC for demographic, deployment, and occupational data. Service data, including deployment dates and locations, are supplied by the DMDC, and environmental exposure data for Army personnel are described in U.S. Army Public Health Command reports. Self-reported exposures are incorporated from the pre- and post-deployment health assessments.
The strengths of MilCo include its large sample of military personnel, including those specifically deployed to war zones, and its use of regular follow-up surveys to monitor for the onset of new health problems. Although the data are collected by questionnaire, this is not a weakness for social/behavioral/
___________________
5 See https://www.millenniumcohort.org/about (accessed June 13, 2018).
dietary and environmental factors, many of which cannot be measured by biomarkers. In addition, questionnaire data on chemical exposures are collected prior to the development of health effects in the participants’ children, which may reduce recall bias. The cohort may therefore play a critical role in evaluating the role of these types of exposures during deployment as predictors of inter- and transgenerational health effects from deployment.
A limitation of the MilCo is its lack of biospecimens and physical examinations. Selection bias is also a potential issue because military personnel who choose to participate may differ from nonparticipants in various ways. In addition, those who continue to participate may differ from those who drop out (e.g., those with medical issues may be retained, but healthy individuals may drop out). Mortality rates also need to be carefully considered in the interpretation of findings. Other limitations to using the MilCo data include: lack of confirmation of self-reported medical issues for participants who are not active-duty, allowing for disease misclassification; and exposure misclassification is also possible as individual exposure information is not available.
DoD has extended the MilCo to include the partners and children of MilCo participants in the Millennium Cohort Family Study (the Family Study). In the section below, the committee discusses modifications to the Family Study that would aid in studying the generational effects of deployment.
Millennium Cohort Family Study
The Family Study is a DoD-sponsored research project nested within MilCo that is intended to study the impact of deployment on family functioning. The participants are partners of service members from the Army, Navy, Air Force, Marine Corps, and Coast Guard who have served since 2000 and are enrolled in MilCo. The Family Study began in 2011 and is intended to follow partners for 21 years or longer, regardless of whether their marriage to the service member continues. Follow-up surveys are conducted every 3 years. The surveys collect information on partners’ physical and mental health, their reports of their children’s physical and mental health and functioning, and family functioning. As of 2014, 9,921 partners of MilCo participants had enrolled in the Family Study, with 8,152 children represented as well. Male partners make up 12% of the participants, and 63% of all participants have children.6
Parents report on their children’s health concerning physician-diagnosed conditions, behavioral problems, and the use of medical, psychological, and social services. The screening questionnaire for behavioral problems among children ages 3 to 17 years includes the Strengths and Difficulties Questionnaire. Additional health data on the children are obtained from DoD sources, including the Birth and Infant Health Registry (described in the next section), and medical and pharmaceutical records from military medical facilities and civilian facilities covered by TRICARE.
The Volume 11 committee finds that there are both strengths and limitations to this study. The strengths of this study are that it actively enrolls partners and children, collects some health information on children, and follows children regardless of whether their parents remain on active duty or separate from the military and regardless of whether the partners separate. The main limitations of this study are that physical exams are not conducted, nor are biospecimens collected. Furthermore, it does not include partners and children of service members who are not MilCo participants. In addition, a child’s behavior is based on only the SDQ screener, which does not allow for the assessment of cognition and other aspects of neurodevelopment.
To enhance the value of the Family Study for investigating generational health effects, the committee has several suggestions. First, it would be advantageous to have MilCo data link with other resources,
___________________
6 See http://www.familycohort.org/whatsnew (accessed May 10, 2018).
such as biospecimens collected as part of the DoD Serum Repository and VA health care records, for continued follow-up. These links would facilitate conducting in-depth investigations of possible associations between deployment exposures and health outcomes. A linkage to DoD and VA medical records would also allow other health outcomes to be assessed and self-reported outcomes to be validated.
Second, because the Family Study is relatively small, with only about 10,000 partners, the committee suggests increasing the sample size; this would permit researchers to look for rare outcomes in children about which veterans are concerned. As an example, if a relatively rare outcome in the offspring such as cleft palate (incidence ~1 in 1,000 births) or childhood cancer (incidence ~18 in 100,000 births) were to be assessed, it would be difficult to identify any such outcomes in the current study population.
Third, the follow-up of the children is limited to questionnaires completed by a parent. The committee suggests that an in-person evaluation be conducted with a brief medical exam (e.g., anthropometry, blood pressure, spirometry in older children) and a developmental examination (a computerized assessment could be done with older children using, e.g., the NIH Toolbox).
In addition, participant permission should be obtained to store and access biospecimens collected as part of routine health care visits. For example, urine samples and blood samples are routinely collected during most pregnancies, but after clinical tests are completed, they are discarded. The committee recommends that the residual sample be aliquoted and saved for those receiving care through TRICARE facilities. These samples would be extremely valuable to use in studying the genetics and epigenetics of participants and in examining prenatal exposures. Parental permission could be obtained to acquire a punch sample from newborn screening cards for genetic information and for assessing in utero exposures. Finally, permission should be obtained to merge data from other state registries, including neonatal screening data (e.g., neonatal thyroid hormone), birth defects and cancer registries, educational records (e.g., state examinations), child special services (e.g., for autism), and other vital statistics data such as the DoD Birth and Infant Health Registry as described below.
DoD Birth and Infant Health Registry
In response to the growing number of women on active duty and the diverse hazardous exposures associated with military service, DoD created the Birth and Infant Health registry in 1998, housed at the Naval Health Research Center in San Diego, to address women’s reproductive health concerns, primarily birth defects. The registry collects comprehensive health care data on live births and infant health outcomes through the first year of life for infants born to TRICARE beneficiaries. Recorded outcomes include preterm birth, pregnancy outcomes, birth weight, growth problems in utero and in infancy, and birth defects. The population-based electronic surveillance system, which contained more than 1.6 million births as of 2017 (NHRC, 2017), draws from hospitalizations at military medical facilities (including those overseas), outpatient encounters at DoD facilities, and civilian medical encounters paid for by TRICARE; the system is supplemented by case validation. Links to demographic and military service information, including information on occupational and military exposures, are available through DMDC (Conlin et al., 2012; Ryan et al., 2001).
The strengths of the registry include its ability to link with other DoD databases and the large number of births that are recorded in it. A limitation of the registry is its lack of a mechanism for following children after 1 year of age even if they continue to be in the TRICARE system or for following children under 1 year if their parents separate from the military. The registry does not capture the children of military personnel who receive care through other insurers, and, finally, the registry does not capture data on pregnancy terminations, miscarriages, or stillbirths (Ryan et al., 2001). Both of these are limitations.
The registry, linked to health care data supplied by TRICARE and the MHS, could be used to compare health conditions in children beyond the first year of life and make comparisons based on known exposures or occupations of the parent. For example, surveillance data from the registry describing pregnancy outcomes and birth defects among women of childbearing age have been published (Bukowinski et al., 2012; Conlin et al., 2012). Several published studies, as discussed in Chapter 4, have described birth outcomes in female veterans who were exposed to burn pit emissions or who received vaccines during their pregnancies (Conlin et al., 2012, 2013, 2015, 2017).
The committee suggests that other useful studies would be feasible with this database. For example, the health status of children conceived within a specific time frame relative to a service member’s return from deployment—say, 90 days for men to cover one spermatogenesis cycle—could be compared with the health status of children of nondeployed service members. Or, children who were conceived shortly before or during deployment, as determined from their mother’s administrative data, could be followed to ascertain their health status, at least while in the TRICARE system.
Other Resources
As noted earlier in the chapter, in addition to the programs discussed above, there are a number of large-scale health-monitoring and epidemiologic studies that may be useful for answering questions about the reproductive health of veterans and the health of their children. The committee believes that these programs and studies, while not sufficient to answer those questions on their own, may provide ancillary resources for VA and DoD efforts. In addition, they also demonstrate the many advantages and challenges faced by VA and other researchers when studying cohorts of veterans. They include
- The National Health Survey of Gulf War Era Veterans and Their Families compares the mental and physical health status of a cohort of 15,000 Gulf War veterans from each branch of the military, including active-duty, reserve, and National Guard, with 15,000 of their nondeployed military peers. Three phases of the study have been conducted: 1995 (n=11,441; response rate not reported), 2005 (n=9,970; response rate 34%), and 2012–2013 (n=14,252; response rate 50%). A questionnaire package was mailed to invitees with follow-up reminder postcards; computer-assisted telephone interviewing calls were made to veterans who had not responded. Respondents received $10. Data collected included sociodemographic information health information on chronic medical conditions, chronic fatigue syndrome-like illness and unexplained multi-symptom illness, mental health, health behaviors, general health, functional health, women’s health, use of complementary and alternative medicine, and VA health care utilization and satisfaction (VA, 2016).
- The National Health Study for a New Generation of U.S. Veterans began as a pilot study in 2009 with data collection concluding in 2010. The goal of the study was to assess the health of Post-9/11 veterans, help identify VA health services that veterans may need, and improve the quality of care offered by VA (VA, 2018b). About 30,000 veterans who were deployed to Iraq and Afghanistan were randomly selected from DoD records and compared with 30,000 veterans who served elsewhere during the same time. More than 20,500 veterans participated in the study with representation from active-duty, reserve, and National Guard members; women comprised 20% of the participants. Data collection included questionnaires with topics that focused on health risk behaviors, health conditions, general health, health care utilization, and potential exposures. No biological samples were collected; however, medical records were reviewed for about 2,000 veterans.
- The Vietnam Era Health Retrospective Observational Study (VE-HEROeS) is designed to evaluate the current health and well-being of 43,000 veterans who served in Vietnam, in the Blue Water Navy, and those who served elsewhere from 1961 to 1975. Veterans’ health will be compared with about 11,000 civilians within the same age groups who never served in the military. Special emphasis is given to assessing any neurologic conditions and hepatitis C infections. Participants complete a questionnaire pertaining to military service, general health, aging, and lifestyle. Data for this study are still being analyzed as well as a review of medical records for participants (VA, 2017a).
-
The Armed Forces Health Surveillance Branch conducts medical surveillance of and for the U.S. service members. It has four components:
- Data Management and Technical Support, which manages the databases to conduct surveillance and analysis of health-related information of U.S. service members.
- Epidemiology and Analysis includes the DMSS, which contains up-to-date and historical data on diseases and medical events (e.g., vaccines, hospital visits) and longitudinal data on more than 7 million personnel and deployments, including pre- and post-deployment health assessments; and the Department of Defense Serum Repository, which contains more than 62 million serologic specimens.
- Global Emerging Infections Surveillance develops, implements, supports, and evaluates an integrated global emerging infections surveillance and response system.
- Integrated Biosurveillance supports many worldwide biosurveillance and public health activities to improve force health protection.
- The Air Force Health Study, begun in 1979, was a longitudinal, prospective cohort study of more than 2,700 men who had participated in Operation Ranch Hand, the wartime herbicide spraying in Vietnam. Participants were followed for approximately 20 years, and they received up to six intensive physical examinations that included clinical measurements and observations, the collection of serum and other biological samples, and responses to questionnaires addressing health, lifestyle, socioeconomic status, employment, families and offspring, and potential sources of environmental exposures (Styka and Butler, 2015).
The DoD Serum Repository, the Joint Pathology Center Biorepository, and the Air Force Health Study all maintain biospecimens of military personnel or veterans. The National Health Survey of Gulf War Era Veterans and Their Families and the Health Study for a New Generation of U.S. Veterans and their follow-up survey have longitudinal designs, representative samples, and questions about reproductive health. DMSS is a set of relational databases that integrate service members’ deployment, health, and exposure data for DoD health surveillance purposes.
PROGRAM MANAGEMENT
Program management is a critical, integral, and continuous function of any HMRP that includes epidemiologic studies. Addressing the many complementary requirements and opportunities of an HMRP will require the identification of lead organizations to design and implement the overall program and specific research studies. This lead organization may be VA, DoD, NIH, or another organization, such as an academic research center or consortium. Close coordination and collaboration among the program partners is critical for accountability.
Once the lead organization for the HMRP has been identified, an organizational structure of the program that includes partners and collaborators along with the responsibilities for each partner will
need to be delineated. The chains of command, personnel, and lines of communication will need to be agreed upon. The strengths and limitations of each collaborator will need to be weighed so that activities are assigned to the most appropriate affiliate. The strengths of the partners may include the availability of subject matter experts, access to relevant data or records, and strong IT capabilities (e.g., DOE). Limitations might reflect the lack of a legislative mandate to participate in the program, financial constraints, and a lack of familiarity with program data requirements, each of which can be understood and addressed through a true multi-organization, cross-country collaborative effort among all participants.
Collaboration and Coordination
The development and implementation of an HMRP to assess generational health effects in veterans and their descendants will require the joint efforts of a number of governmental and nongovernmental organizations. Although the statutory language in P.L. 114-315 Section 362 implies that VA will lead the effort to conduct the HMRP, the scope of the mandate will require well-focused collaborative efforts to achieve success. Although VA may lead the assessment of reproductive effects in veterans, it has limited experience in monitoring the health of nonveterans, especially children. VA will need to work closely with other organizations, public and private—and potentially other countries—to implement a program that includes assessing the health of descendants and partners in order to avoid duplicating resources or parts of programs to minimize costs while maximizing benefits.
Collaborators and affiliates for the HMRPs might include DoD; DOE; the Department of Health and Human Services, including NIH (e.g., National Institute of Child Health and Development and the National Institute of Environmental Health Sciences [NIEHS]) and the Centers for Disease Control and Prevention; the Agency for Toxic Substances and Disease Registry (ATSDR); states’ departments of health, including newborn screening and vital statistics; VSOs; foundations with an interest in the health and well-being of service members, veterans, and families (e.g., the Robert Wood Johnson Foundation, Henry M. Jackson Foundation); and academic institutions, all of which may provide guidance on, and resources for, specific aspects of the HMRP. Furthermore, it may be advantageous to consider working with large health care organizations in the private sector, as it is likely that many veterans and their children and grandchildren may receive medical care from such providers. The collection of health data, access to those data, and the sharing of information among collaborators and affiliates and with the public would be addressed by the HMRP management team. In addition, VA may want to consider working with other countries, such as the United Kingdom or Australia, that were part of the coalition forces that fought in the Gulf War and the Post-9/11 conflicts. Service members from those countries had many exposures similar to U.S. forces and they have the advantage of national health systems that include all citizens, including veterans and their descendants. The United Kingdom Medical Research Council and Wellcome Trust established the UK Biobank prospective cohort in 2002 to help investigate risk factors for the major diseases of middle and old age. The biobank recruited more than 500,000 men and women ages 40–69 years between 2006–2010 and their health is being followed long term (Collins, 2012). Should veteran status be included, this might provide a valuable resource even for U.S. researchers.
Each collaborator and affiliate should bring different yet complementary strengths and resources to the program. For example, VSOs may be able to reach out to their members to encourage participation and retention in the program and to assist in the dissemination of surveys. Their expertise in crafting surveys and identifying research priorities should also be solicited. They may also serve as a staging platform for the collection of biological specimens such as blood, saliva, and urine samples. Academic institutions, particularly those affiliated with medical centers, may provide guidance concerning program
design, the selection of biomarkers, statistical and bioinformatics assessments, and innovative approaches for data collection and analysis, in addition to access to experts in reproductive and child health and genetics and epigenetics. State health departments and other state programs may provide access to state registries and other data, whereas local city and county agencies can provide supportive efforts.
In P.L. 114-315, Section 633, Congress called on the Secretary of VA to establish an “Advisory Board On Research Relating To Health Conditions Of Descendants Of Veterans With Toxic Exposure While Serving In The Armed Forces.” The Advisory Board is to “advise the Secretary in the selection of a research entity or entities under section 634, advise such entity or entities in conducting research under such section, and advise the Secretary with respect to the activities of such entity or entities under such section.” The committee is to be composed of 13 members with expertise in birth defects, developmental disabilities, epigenetics, public health, reproduction, environmental exposure assessment, the science of toxic substances, and medical and research ethics. The Volume 11 committee strongly endorses the creation of such a term-limited rotating advisory board to energize the HMRP. Such an advisory board may serve as a catalyst for promoting collaboration and coordination among participants, including both governmental and nongovernmental organizations. To ensure interoperability, this oversight body can provide critical feedback for each veteran cohort–specific program as it is established and can ensure periodic evaluations so that progress toward program goals is monitored and necessary adjustments to the program are implemented in a timely manner. As cohorts are delineated, membership on the oversight committee should adapt to meet the needs of the program. This would inform the rotation of, and membership to, the Scientific Advisory Board on Research Relating to Health Conditions of Descendants of Veterans with Toxic Exposure While Serving in the Armed Forces. For example, an advisory board may recommend new biomarkers of effect as they come into use or provide input on specifics for the number of veterans or their descendants that will need to be included in each cohort-specific program.
Data Resource Needs
The data resources that will be required to conduct a comprehensive and longitudinal HMRP will be substantial. The committee considers that, at a minimum, the lead and collaborating organizations should have access to distributed IT systems capable of handling a large amount of data. When an interface with other IT systems is necessary, program managers will also need to ensure that the appropriate data privacy safeguards are in place, including processes to ensure compliance with the Health Insurance Portability and Accountability Act (also known as HIPAA). The “NIH Security Best Practices for Controlled-Access Data Subject to the NIH Genomic Data Sharing (GDS) Policy” (NIH, 2018a) is one resource for guidance on ensuring the security of human data from which the specifications could be drawn. Other resource needs include personnel (e.g., scientific, technological, administrative, logistical), facilities, and equipment.
The committee considers IT to be one of the pillars of the HMRP. Independent of the particular veteran cohort to be studied, the data needs of the program will be voluminous, and the management of these data will be critical to all aspects of the program. The extensive interoperable database created during the course of the program data collection will likely include, for example, detailed data from DoD regarding deployments, dependents, medical care, occupation and duties; VA medical data; contact information; updated dependent information; biospecimen data (collection dates, status, location, etc.); genomic data; and responses to surveys. Thus, the scope of the technological requirements for the HMRP will include substantial storage capabilities; linkage of EHRs for veterans and their descendants as they move from DoD to VA and other health care providers; and maintenance of a secure cloud by
IT specialists who can work with program managers and analysts to support databases, interfaces, websites, and other electronic information exchange platforms. This integral component that will ensure the veracity, accessibility, and usefulness of the data collected. Furthermore, processes and protocols to protect sensitive personal information will be essential to ensure secure data collection, maintenance, and use. Although the committee realizes that VA and DoD already maintain extensive and well-protected databases on veterans and service members, the necessary collaborations will require that each partner (e.g., NIH, private foundations, state cancer registries) has data privacy safeguards and appropriate IT capabilities in place. Some aspects of medical data linkages for DoD and VA were discussed earlier.
COMMUNICATION
Communication activities will be essential for a successful HMRP and its associated epidemiologic studies. As described above in Collaboration and Coordination, lines of communication and authority will need to be established among the HMRP collaborators and affiliates. The HMRP will need to communicate effectively with all stakeholders, the general public, Congress, and other researchers. This can be achieved as the HMRP management team formalizes bidirectional communication networks with veterans and their descendants. This communication pathway should engage veterans in a dialogue and involve them in the program from start-up to evaluation.
Return of Results
Researchers and veterans and their families together should develop clear guidelines on the communication of individual and group-level study results since these results may have marked ramifications for each participant. For example, researchers and veterans and their families together should consider what health or developmental information should be returned to individuals, carefully weighing the benefits versus the risks of receiving this information. In most cases, individuals should be able to opt in to or opt out of receiving individual-level results. It should also be determined a priori what information will be given back to health care providers with the individual’s permission and which data will under law need to be reported to authorities (e.g., suspected child abuse or neglect or reportable diseases), and under what criteria. In addition, the overall study results should be regularly reported to participants, and the best vehicle for this report should be discussed with participant groups. The return of results is governed by well-established principles of ethical conduct in research, and some of these key elements are discussed below within the context of the proposed HMRP to evaluate reproductive and generational health effects.
Veterans and Their Descendants
Achieving high levels of participation (recruitment and retention) in the HMRP will require purposeful, consistent, and comprehensive communication among the leadership of the HMRP, the U.S. military, and the veteran populations and their descendants. A communication strategy should be developed early that will include the recruitment and enrollment of service members and veterans and their descendants into the program. As noted earlier, the strategy will need to facilitate the retention of HMRP participants through the ongoing reporting of results to stakeholders. Transparency and the inclusion of the public will be essential attributes of the program. DoD, VA, and other collaborators should have a well-developed and validated communication mechanism for engaging service members and their children in the MHS. Engaging children from an early age using age-appropriate methods may encourage their participation as
they mature and after their parents separate from the military or no longer receive TRICARE coverage. These attitudes may also translate to the children’s children—that is, the veteran’s grandchildren—as it becomes part of a family tradition. Given that many children of military parents join the military themselves, this may be an important group to identify and engage to assess for generational effects.
The committee suggests that in order to better recruit and retain participants, activities that have been demonstrated to be successful by other longitudinal studies should be considered, modified, and adopted as appropriate by the HMRP. For example, participants in the National Children’s Study felt pride in identifying themselves as part of the study cohort, and their interest was encouraged with such meaningful incentives from the researchers as networking, priority newsletters, and the occasional special event that prompted group recognition of the participants’ continuation in the program. The Nurses’ Health Study Growing Up Today Study (GUTS), has employed newsletters, emails, and birthday cards to promote a sense of community and create a sense of added value in belonging to the program among participants. Communication with the participants of the HMRP will have to be in multiple forms (e.g., social media, Internet, mailings, media, and messaging) that reflect the preferred methods of communication for different groups of participants, factoring in considerations such as age, which often predicts social media usage.
The use of electronic surveys for collecting information and the ability to rapidly analyze that information is essential. For example, to reduce the time and effort for data collection on the part of both the HMRP staff and the program participants, the program could quickly disseminate reminders and notices regarding the availability of surveys to large numbers of recipients by using electronic databases such as voter rolls to locate the descendants of veterans. The MilCo program offers a good example of how electronic surveys and data analysis can be used with a veteran population. Interactive websites, mobile apps, electronic reminders, and other electronic approaches may optimize veteran and public interest in and response to the program.
Communication campaigns must be tailored to the target constituency. For example, Vietnam and Gulf War veterans and their adult descendants may respond better to the use of telephone landlines and the U.S. Postal Service than to electronic forms of communication, including texting, emails, and social media campaigns, which may be of more appeal to Post-9/11 veterans. This should also be a consideration for reaching out to the children and grandchildren of the veterans. Mass media communications such as brief public television announcements and AM/FM radio might also be helpful in some instances. The program must keep pace with new trends in technology that will most likely reach the early adopters—the children and grandchildren of the veterans—who will be a key group in the generational and transgenerational study.
In-person meetings might be held at VSO facilities, if appropriate, or at local VA medical facilities, including Vet Centers, around the country. Virtual connectivity would allow veterans and family members to engage if they are unable to attend in person. The use of VA facilities might serve the dual purpose of engaging HMRP participants and familiarizing the veterans with VA if they have not already been receiving VA medical care. HMRP staff needs to be culturally aware of the veteran communities with whom they are trying to engage. Messages may need to be in the language the veteran uses at home, such as Spanish or Vietnamese. Public advertisements might also be needed to inform veterans and the public about the program. Regular updates on the implementation of the HMRP need to be presented in an array of national scientific meetings in addition to meetings attended by military and veteran populations.
As noted earlier, the return-of-results policy developed for the HMRP can be based on recently developed protocols by the All of Us research program. This is an important issue for any longitudinal
study, and documents are being developed for communicating child health outcomes by the NIH ECHO consortium.
Other Key Stakeholders
Information about the HMRP needs to be disseminated and to reach all stakeholders, such as members of the scientific community, health care systems, government officials, veterans and their descendants, and the general public, as discussed above. Communication among governmental and nongovernmental agencies will need to be efficient and effective in order to ensure that program goals are achieved. A biennial report detailing what has been discovered, the status of recruitment efforts, and participation statistics might be an effective communication mechanism for enhancing public awareness of the program. Publicity and media releases might include widely accessible videos, such as podcasts and TED Talks.
A comprehensive and user-friendly webpage for the HMRP would be an important communication tool for all stakeholders as well as a way of preparing for the future with virtual reality and other forms of media that are being developed and gaining audiences as communication media. VA is familiar with designing such webpages for stakeholder information such as the one for the MVP.7 Information on the webpage could be divided into information useful for the veterans and their descendants, for researchers, and for the general public, yet still remain accessible to all. New research may be highlighted; new procedures or surveys may be announced; basic background information on toxicants and their effects may be explained; and appropriate links to other agencies with additional information, such as ATSDR, could be provided with annotation.
PROGRAM EVALUATION
A well-conducted HMRP would include an iterative evaluation process and guidance from a veterans advisory committee and scientific advisory board to provide important feedback on priorities and evaluation metrics. For example, as the program matures and information is analyzed, it will likely be necessary to adjust the data collection efforts to capture new exposures for veterans and to more closely examine outcomes in descendants. As new sampling techniques and instruments become available for environmental toxicants, it may be advantageous to collect new or different types of information on deployment exposures. As the science of omics advances, it may become easier to identify biomarkers of exposure or biomarkers of effect and susceptibility in both veterans and their descendants. Furthermore, as more information is gathered, it may become evident which exposures and effects are most likely to be associated with each other, and this would allow HMRP leadership to narrow or broaden the scope of the program.
The scope and complexity of the program and the research to be proposed will require repeated feedback from researchers and veterans and their descendants to help modify the HMRP to address new information and processes. Such evaluations should be conducted on a regular basis with a mechanism for communicating with program administrators, researchers, and the public. VA is already familiar with many aspects of program evaluation, particularly those related to improving patient care. Should the need arise, there are other organizations that have developed relevant metrics that may be used by VA and its collaborators in assessing the value of the program to governmental organizations as well
___________________
7 See https://www.research.va.gov/MVP/veterans.cfm (accessed February 7, 2018).
as to veterans and their descendants; one example is the Partnerships for Environmental Public Health Evaluation Metrics Manual from NIEHS (2012).
To ensure a successful program there will also be a need to regularly assess the efficiency of the HMRP’s processes, its methods to recruit and retain participants, and the appropriateness of its data collection efforts. Those challenges and limitations that are identified throughout the process should be used to inform and modify the HMRP. For example, if administrators notice a particularly low participation rate for a certain demographic group, efforts should be made to adjust recruitment efforts in a timely manner.
ETHICAL CONSIDERATIONS
There are important ethical considerations in designing and conducting an investigation on the health of veterans, their partners, children, and grandchildren. Voluntary participation in the HMRP requires developing informed consent procedures as defined in protocols reviewed and approved by the institutional review boards of the lead and collaborating institutions. Such procedures typically follow the ethical principles identified in the Belmont Report of beneficence, justice, and respect (HEW, 1979). VA and DoD have already applied these principles in the MVP, MilCo, and other comparable programs. However, the recruitment, engagement, and participation of the children and grandchildren of veterans will involve unique considerations and challenges. The structuring of such programs could benefit from the procedures developed by other initiatives, such as the NIH National Children’s Study and the ECHO and All of Us studies.
While the goal of studying veterans and their partners and descendants is to generate knowledge concerning the health outcomes of these individuals as shaped by the veterans’ deployments, additional knowledge may be gathered that can improve personalized medicine and be generalized to the broader U.S. population. It is essential to secure voluntary informed consent from adults who clearly understand their involvement and the risks and benefits associated with participation. Whether and how individual and group results will be returned to them should be apparent to the participants. If results, particularly those for individuals, are to be provided to participants, they should be presented in a manner that promotes the health of the recipient. Providing results to individual participants may be difficult for the proposed HMRP, given the need for proxy consents for studies across multiple generations and the fact that much of the research to be conducted is being developed upon a very limited database that may not allow for a clear explanation of risks and benefits for participants. As such, additional levels of scrutiny will be required to assess the level of risk inherent in any one study developed under the HMRP umbrella as well as the benefits to be derived in the short term and across multiple generations. A key question to be addressed will be how the risks and benefits to participants and their descendants, as determined from the HMRP, will be communicated to them.
The participation of children and grandchildren, especially those who are minors, will require special considerations. Because children are considered uniquely vulnerable research subjects, important decisions will need to be made about what will be studied; whether there will be particular risks inherent in the research that must be shared with the children’s parents or guardians; whether the children are old enough to provide oral or written assent themselves; and what results will be shared with parents and clinicians or educators and how they will be shared. The researcher should also consider the intended and unintended consequences of the findings and which options will be made available should negative health outcomes be identified when remediation at this time is not possible. If recontact is not possible, the disposition of any associated data has to be considered with the utmost care. Careful consideration must also be given to the possible perception by authorities that the children of veterans might be
treated as a “captive population” with restricted opt-out options. It will also be important in instances when multiple follow-ups are necessary to consider how frequently adults and children are asked to re-consent or re-assent.
The legality of proxy consents for a child in the case of procedures that are not related to the child’s need for medical diagnosis or treatment will also be critical factors. Furthermore, the participation of children with preexisting conditions and the options for the return of results along with the choice to receive or not receive certain types of information are but a few of the many ethical considerations that will require careful review and consideration by VA and collaborating organizations. Thus, efforts must be made to develop studies that can be justified for specific populations, with appropriate safeguards and oversight and clear and definable benefits, especially as the children mature and begin to participate in decisions in situations where the risks and benefits to them may not be entirely clear.
When the child is of age to provide assent, the purpose and the procedures of the study should be described to the child in a developmentally appropriate manner. When the child reaches adulthood, he or she may be required to consent to the use of all previous data and samples collected when the child was a minor. This will require a special consenting procedure and the need to retain detailed contact information for recontact. If recontact is not possible, the disposition of any associated data has to be carefully considered. Sponsoring organizations will have to address issues related to inter-institutional variation in human subject procedures, the utility of centralized human subjects protocol review (with a central institutional review board), and alternatives for the completion of face-to-face, mail, telephone, or electronic consenting. Many advances have been made in this regard during the course of the implementation of several large-scale national initiatives by both VA and DoD.
The opportunity for and interest in genetic testing research as part of the proposed HMRP deserves special comment as it presents unique ethical challenges, especially those related to the testing of asymptomatic children, familial identity, incidental findings, variants of unknown significance, and carrier status, to list a few difficult issues. Several authoritative reviews and guidelines are available that can be used to guide efforts (e.g., HEW, 1979; NIH, 2018b; WHO, 2011). This can be a particularly sensitive area in situations where the investigation identifies genetic risks for which no clear intervention is available. In the context of investigations conducted over a lifetime and into future generations, the risk-benefit considerations of genetic testing will be influenced by the timing of disease onset and the availability of treatments. Similar issues arise from the collection of stem cells from parents and children and the possible future use of these specimens by participants, their family members, and society at large.
The practice of protecting human participants in research has evolved, and it is expected that ethical practices in human subject research will continue to do so as new adverse events, advanced methodologies, and altered philosophies concerning the protection of human subjects emerge. Clearly, the challenge for policy makers and researchers at VA, DoD, and collaborating organizations will be to develop an HMRP that minimizes risk, provides acceptable risk-benefit ratios to participants, and ensures the engagement of parents and children over time in studies likely to generate valuable findings. In the end, VA and veterans must weigh the risk of conducting and participating in such research against the risk of not seeking answers. Only VA, in consultation with veterans and their descendants, can make this decision.
SUMMARY
Designing, implementing, maintaining, and evaluating a program to conduct research to identify current or possible health effects in veterans’ descendants is a substantial task, but many new technologies and methods are available to facilitate such a program, and the committee believes that it is an important undertaking. Understanding the possible generational health effects that may result from
veterans’ exposures during deployment begins with determining, to the extent possible, which toxicants they encountered. Newer technologies that can screen thousands of chemicals in biological samples combined with biobanks may provide an important new method for determining the relevant environmental exposures. Furthermore, developments in high-throughput computing and informatics can strengthen such a program’s ability to zero in on the most relevant health outcomes. At present, the answers to which environmental exposures and which health outcomes are most related to transgenerational inheritance are largely speculative, but advances in bioassays, computer science, and study design should help in addressing these issues, as long as they are rigorously employed. This information on exposures and outcomes should be complemented with the concurrent collecting of biological samples from service members and veterans. As children are born following their parents’ deployment, they too may be monitored during their lifetimes to determine what, if any, health effects they may have compared with children of veterans who were not deployed.
An HMRP with a structured data collection and analysis protocol is an ideal approach for framing research questions that may be addressed by epidemiologic studies. By building on existing health monitoring and epidemiologic research programs such as the DoD MilCo and the VA MVP, and taking advantage of access to databases such as the DoD Serum Repository and the DoD Birth and Infant Health Registry, it should be possible to reduce costs, expedite data collection, and provide access to already-engaged study populations.
An HMRP with epidemiologic studies will require efficient program management and access to a variety of resources. In particular, it will be critical for the program to have sufficient IT capabilities
combined with linked medical records from DoD and VA and potentially other participants. With a sustained commitment from governmental and other organizations, the generational health effects that may stem from veterans’ exposures experienced while defending their country may be illuminated.
The critical features of any HMRP devoted to veterans and their descendants are described above and listed in Box 9-3. Such a program could feasibly encompass any theater of operation, ranging from humanitarian disaster relief to a direct combat mission. A coordinated health monitoring system is needed to address these important questions. This HMRP will integrate and expand existing and future information across the multiple levels of decision making in order to generate timely and actionable knowledge for individual veterans and their children, VA, DoD, and Congress.
REFERENCES
Accardi, C.J., D. I. Walker, K. Uppal, A.A. Quyyumi, P. Rohrbeck, K.D. Pennell, C. T. Mallon, and D.P. Jones. 2016. High-resolution metabolomics for nutrition and health assessment of Armed Forces personnel. Journal of Occupational and Environmental Medicine 58(8 Suppl 1):S80–S88.
Andermann, A., I. Blancquaert, S. Beauchamp, and V. Déry. 2008. Revisiting Wilson and Jungner in the genomic age: A review of screening criteria over the past 40 years. Bulletin of the World Health Organization 86(4):241–320.
Andra, S., C. Austin, and M. Arora. 2016. The tooth exposome in children’s health research. Current Opinion in Pediatrics 28(2):221–227.
Bagalman, E. 2014. The number of veterans that use VA health care services: A fact sheet. CRS Report 7-5700. Washington, DC: Congressional Research Service. R43579.
Bellavia, A., and L. Valeri. 2018. Decomposition of the total effect in the presence of multiple mediators and interactions. American Journal of Epidemiology 87(6):1311–1318.
Bennett, D.A., D. Landry, D.J. Little, and C. Minelli. 2017. Systematic review of statistical approaches to quantify, or correct for, measurement error in a continuous exposure in nutritional epidemiology. BMC Medical Research Methodology 17:146–168.
Billionnet, C., D. Sherrill, I. Annesi-Maesano, and GERIE study. 2012. Estimating the health effects of exposure to multipollutant mixture. Annals of Epidemiology 22(2):126–141.
Bradburne, C., D. Graham, H.M. Kingston, R. Brenner, M. Pamuku, and L. Carruth. 2015. Overview of ’omics technologies for military occupational health surveillance and medicine. Military Medicine 180(10 Suppl):34–48.
Braun, J.M., C. Gennings, R. Hauser, and T.F. Webster. 2016. What can epidemiological studies tell us about the impact of chemical mixtures on human health? Environmental Health Perspectives 124(1):A6–A9.
Buka, S., S.R. Rosenthal, and M.E. Lacy. 2018. Epidemiological study designs: Traditional and novel approaches to advance life course health development research. In Handbook of Life Course Health Development, edited by N. Halfon, C. Forrest, R.M. Lerner, and E.M. Faustman. 10.1007/978-3-319-47143-3 (accessed July 18, 2018).
Bukowinski, A.T., C. DeScisciolo, A.M.S. Conlin, M.A.K. Ryan, C.J. Sevick, and T.C. Smith. 2012. Birth defects in infants born in 1998–2004 to men and women serving in the U.S. military during the 1990–1991 Gulf War era. Birth Defects Research (Part A): Clinical and Molecular Teratology 94:721–728.
Collins, R. 2012. What makes UK Biobank special? The Lancet 379:1173–1174.
Conlin, A.M., C. DeScisciolo, C.J. Sevick, A.T. Bukowinski, C.J. Phillpis, and T.C. Smith. 2012. Birth outcomes among military personnel after exposure to documented open air burn pits before and during pregnancy. Journal of Occupational and Environmental Medicine 54(6):689–697.
Conlin, A.M., A.T. Bukowinski, C.J. Sevick, C. DeScisciolo, and N.F. Crum-Cianflone. 2013. Safety of the pandemic H1N1 influenza vaccine among pregnant U.S. military women and their newborns. Obstetrics and Gynecology 121(3):511–518.
Conlin, A.M.S., A.T. Bukowinski, and G.R. Gumbs. 2015. Analysis of pregnancy and infant health outcomes among women in the National Smallpox Vaccine in Pregnancy Registry who received anthrax vaccine adsorbed. Vaccine 33(36):4387–4390.
Conlin, A.M.S., C.J. Sevick, G.R. Gumbs, Z.G. Khodr, and A.T. Bukowinski. 2017. Safety of inadvertent anthrax vaccination during pregnancy: An analysis of birth defects in the U.S. military population, 2003–2010. Vaccine 35(34):4414–4420.
DoD (U.S. Department of Defense). 2013. TRICARE management activity FY 2013 military construction, defense-wide. Chief, Acquisition and Management Office.
DoD. 2018. Patients by beneficiary category. https://www.health.mil/I-Am-A/Media/Media-Center/Patient-PopulationStatistics/Patients-by-Beneficiary-Category (accessed June 22, 2018).
Dursa, E.K., S.K. Barth, A.I. Schneiderman, and R.M. Bossarte. 2016. Physical and mental health status of Gulf War and Gulf era veterans. Journal of Occupational and Environmental Medicine 58(1):41-46.
Eskenazi, B., K. Harley, A. Bradman, E. Weltzien, N.P. Jewell, D.B. Barr, C.E. Furlong, and N.T. Holland. 2004. Association of in utero organophosphate pesticide exposure and fetal growth and length of gestation in an agricultural population. Environmental Health Perspectives 112(10):1116–1124.
GAO (U.S. Government Accountability Office). 2015. Defense health care: DOD needs to clarify policies related to occupational and environmental health surveillance and monitor risk mitigation activities. GAO-15-487. Washington, DC: U.S. Government Accountability Office.
Gaziano, J.M., J. Concatoc, M. Brophy, L. Fiore, S. Pyarajana, J. Breeling, S. Whitbourn, J. Deena, C. Shannona, D. Humphries, P. Guarino, M. Aslan, D. Anderson, R. LaFleur, T. Hammond, K. Schaaf, J. Moser, G. Huang, S. Muralidhar, R. Przygodzki, and T.J. O’Leary. 2016. Million Veteran Program: A mega-biobank to study genetic influences on health and disease. Journal of Clinical Epidemiology 70:214–223.
Hajian-Tilaki, K. 2011. Sample size estimation in epidemiologic studies. Caspian Journal of Internal Medicine 2(4):289–298.
Hamra, G.B., and J.P. Buckley. 2018. Environmental exposure mixtures: Questions and methods to address them. Current Epidemiology Reports.https://doi.org/10.1007/s40471-018-0145-0 (accessed August 5, 2018).
Hebbring, S.J. 2014. The challenges, advantages and future of phenome-wide association studies. Immunology 141(2):157–165.
Hernan, M.A., and J.M. Robins. 2018. Causal inference. https://www.hsph.harvard.edu/miguel-hernan/causal-inference-book (accessed July 27, 2018).
HEW (U.S. Department of Health, Education, and Welfare). 1979. The Belmont report: Ethical principles and guidelines for the protection of human subjects of research. Washington, DC: Office of the Secretary, The National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research. April 18.
Huang, G., S. Kim, J. Gasper, Y. Xu, T. Bosworth, and L. May. 2017. 2016 survey of veteran enrollees’health and use of health care: Main results report. Prepared for the Department of Veterans Affairs. Rockville, MD: Westat.
IOM (Institute of Medicine). 2008. Epidemiologic studies of veterans exposed to depleted uranium. Washington, DC: The National Academies Press.
IOM. 2010. Gulf War and health, volume 8: Update of health effects of serving in the Gulf War. Washington, DC: The National Academies Press.
IOM (Institute of Medicine). 2011. Long-term health consequences of exposure to burn pits in Iraq and Afghanistan. Washington, DC: The National Academies Press.
Janulewicz, P.A., R.F. White, M.R. Winter, J.M. Weinberg, L.E. Gallagher, V. Vieira, T.F. Webster, and A. Aschengrau. 2008. Risk of learning and behavioral disorders following prenatal and early postnatal exposure to tetrachloroethylene (PCE)contaminated drinking water. Neurotoxicology and Teratology 30(3):175–185.
Keogh, R.H., and I.R. White. 2014. A toolkit for measurement error correction, with a focus on nutritional epidemiology. Statistics in Medicine 33(12):2137–2155.
Lenth, R.V. 2001. Some practical guidelines for effective sample size determination. The American Statistician 55:187–193.
Liu, A., E.F. Schistermann, M. Mazumdar, and J. Hu. 2005. Power and sample size calculation of comparative diagnostic accuracy studies with multiple correlated test results. Biometrical Journal 47(2):140–150.
Liu, K.H., D.I. Walker, K. Uppal, V. Tran, P. Rohrbeck, T.M. Mallon, and D.P. Jones. 2016. High-resolution metabolomics assessment of military personnel: Evaluating analytical strategies for chemical detection. Journal of Occupational and Environmental Medicine 58(8 Suppl 1):S53–S61.
Lushniak, B., T.M. Mallon, J.C. Gaydos, and D.J. Smith. 2016. Utility of the Department of Defense Serum Repository in assessing deployment exposure. Journal of Occupational and Environmental Medicine 58(8S):S1–S2.
McDiarmid, M.A. 2015. Biologic monitoring and surveillance results for the Department of Veterans Affairs’ depleted uranium cohort: Lessons learned from sustained exposure over two decades. American Journal of Industrial Medicine 58:583–594.
Mumford, S.L., E.F. Schisterman, A. Vexler, and A. Liu. 2006. Pooling biospecimens and limits of detection: Effects on ROC curve analysis. Biostatistics 7(4):585–598.
NARA (U.S. National Archives and Records Administration). 2015. National Collaborative Perinatal Project, 1959-1974. https://www.archives.gov/research/electronic-records/nih.html (accessed September 17, 2018).
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. Gulf War and health, volume 10: Update of health effects of serving in the Gulf War, 2016. Washington, DC: The National Academies Press.
NASEM. 2017. Assessment of the Department of Veterans Affairs Airborne Hazards and Open Burn Pit Registry. Washington, DC: The National Academies Press.
NHRC (Naval Health Research Center). 2017. Research and development focus areas.https://www.med.navy.mil/sites/nhrc/Pages/Research-and-Development-Focus-Areas.aspx?Category=MILITARY-RANDDFOCUS (accessed June 4, 2018).
NIEHS (National Institute of Environmental Health Sciences). 2012. Partnerships for environmental public health evaluation metrics manual. NIH Publication No. 12-7825. Research Triangle Park, NC: National Institute of Environmental Health Sciences.
NIEHS. 2018. Children’s Health Exposure Analysis Resource (CHEAR).www.niehs.nih.gov/research/supported/exposure/chear/index.cfm (accessed June 29, 2018).
NIH (National Institutes of Health). 2018a. NIH security best practices for controlled-access data subject to the NIH genomic data sharing (GDS) policy. https://osp.od.nih.gov/wp-content/uploads/NIH_Best_Practices_for_Controlled-access_Data_Subject_to_the_NIH_GDS_Policy.pdf (accessed June 22, 2018).
NIH. 2018b. NIH human subjects policies and guidance. https://humansubjects.nih.gov/nih-human-subjects-policies-guidance (accessed July 22, 2018).
NRC (National Research Council). 2000. Strategies to protect the health of deployed U.S. forces: Analytical framework for assessing risks. Washington, DC: The National Academies Press.
Pearl, J., M. Glymour, and N. Jewell. 2016. Causal inference in statistics: A primer. Chichester, UK: Wiley.
Rosenbaum, P.R., and D.B. Rubin. 1984. Reducing bias in observational studies using subclassification on the propensity score. Journal of the American Statistical Association 79(387):516–524.
Ryan, M., M. Pershyn-Kisor, W. Honner, T. Smith, R. Reed, and G. Gray. 2001. The Department of Defense Birth Defects Registry: Overview of a new surveillance system. Teratology 64:S26–S29.
Saha-Chaudhuri, P., and C.R. Weinberg. 2013. Specimen pooling for efficient use of biospecimens in studies of time to a common event. American Journal of Epidemiology 178(1):126–135.
Schisterman, E., and A. Vexler. 2008. To pool or not to pool, from whether to when: Applications of pooling to biospecimens subject to a limit of detection. Paediatric and Perinatal Epidemiology 22:486–496.
Smith, T. 2009. The US Department of Defense Millennium Cohort Study: Career span and beyond longitudinal follow-up. Journal of Occupational and Environmental Medicine 51(10):1193–1201.
Stingone, J.A., K.H. McVeigh, and L. Claudio. 2016. Association between prenatal exposure to ambient diesel particulate matter and perchloroethylene with children’s 3rd grade standardized test scores. Environmental Research 148:144–153.
Styka, A. N., and D. A. Butler. 2015. The Air Force Health Study Data and Specimens as a Resource for Researchers. Military Medicine 180(10 Suppl):79–84.
Taguri, M., J. Featherstone, and J. Cheng. 2018. Causal mediation analysis with multiple causally non-ordered mediators. Statistical Methods in Medical Research 27(1):3–19.
Ten Have, T.R., and M.M. Joffe. 2012. A review of causal estimation of effects in mediation analyses. Statistical Methods in Medical Research 21(1):77–107.
Toledano, M.B., R.B. Smith, J.P. Brook, M. Douglass, P. Elliott. 2015. How to establish and follow up a large prospective cohort study in the 21st Century—Lessons from UK COSMOS. PLOS ONE 10(7): e0131521.
VA (U.S. Department of Veterans Affairs). 2016. Health study of Gulf War era veterans. https://www.publichealth.va.gov/epidemiology/studies/gulf-war-longitudinal-study.asp (accessed September 11, 2018).
VA. 2017a. VE-HEROeS: Studying the Health of Vietnam Veterans. https://www.publichealth.va.gov/exposures/publications/agent-orange/agent-orange-summer-2016/ve-heroes.asp (accessed August 30, 2018).
VA. 2017b. VA Utilization Profile FY 2016. National Center for Veterans Analysis and Statistics. https://www.va.gov/vetdata/docs/QuickFacts/VA_Utilization_Profile.pdf (accessed September 7, 2018).
VA. 2017c. Million Veteran Program.https://www.research.va.gov/MVP (accessed September 19, 2018).
VA. 2018a. https://www.va.gov/HEALTHBENEFITS/apply/veterans.asp (accessed September 14, 2018).
VA. 2018b. Health Study for a new generation of U.S. veterans. https://www.publichealth.va.gov/epidemiology/studies/newgeneration/index.asp (accessed September 11, 2018).
VanderWeele, T.J. 2016. Mediation analysis: A practitioner’s guide. Annual Reviews in Public Health 37:17–32.
Walker, D. I., T. Mallon, P.K. Hopke, K. Uppal, K.Y.-M. Go, P. Rohrbeck, K.D. Pennell, and D.P. Jones. 2016. Deployment-associated exposure surveillance with high-resolution metabolomics. Journal of Occupational and Environmental Medicine 58(8):S12–S21.
Wang, M., A. Cyhaniuk, D.L. Cooper, and N.N. Iyer. 2017. Identification of patients with congenital hemophilia in a large electronic health record database. Journal of Blood Medicine 8:131–139.
Westat. 2010. National Survey of Veterans, Active Duty Service Members, Demobilized National Guard and Reserve Members, Family Members, and Surviving Spouses. Final report. Deliverable 27. Prepared for the U.S. Department of Veterans Affairs. Rockville, MD: Westat.
WHO (World Health Organization). 2011. Standards and operational guidance for ethics review of health-related research with human participants. Geneva, Switzerland: World Health Organization.
WHO. 2018. Epidemiology.http://www.who.int/topics/epidemiology/en (accessed June 4, 2018).
This page intentionally left blank.




















































