7
Selected Impairments and Limitations in Functional Abilities Relevant to Work
The committee’s Statement of Task (see Box 1-1 in Chapter 1) includes “in the context of disability assessment, describ[ing] the spectrum of changes to functional abilities relevant to work requirements related to the progression of common disease processes in example impairments,” which “could include, but are not limited to back disorders, cardiac impairments, or depression.” Chapter 7 addresses this charge for several common conditions. These illustrations of disease trajectory, treatment, and related disability are provided as though each condition is present in isolation. Yet more commonly, these pure trajectories are modified by their intersections with each other, by aging, and by comorbidities that influence the impact of disease even when not themselves a major cause of limitation.
For each condition addressed, the committee was asked to
- Identify where along the spectrum an individual’s ability to perform functions relevant to work requirements is affected;
- Describe whether SSA [U.S. Social Security Administration] could expect improvement, no improvement, or progressive worsening in the example impairments;
- Describe the efficacy of medications and other treatments on an individual’s ability to perform functional abilities relevant to work requirements for these examples, and whether that treatment causes its own subset of medical and/or psychological problems that negatively affect an individual’s functioning and how SSA could request an appropriate assessment of functional changes;
- Describe when significant changes in functional abilities relevant to work requirements may occur through the aging process for these examples, such as for adults with common age-related physical and mental impairments; and
- Describe how the examples are similar to or different from other impairments. (See Box 1-1 in Chapter 1.)
In addition to back disorders, cardiac impairments, and depression, the committee elected to address traumatic brain injury (TBI) because of its prevalence and complexity and the associated high rates of cognitive impairment and work disability. In this chapter, the committee discusses for each condition the above five bullet points under three headings: (1) effects on the ability to perform work and expectations for improvement, (2) effects of treatment, and (3) effects of aging and comorbidities. This discussion is followed by a summary and comparison of the trajectories of the four conditions, the effects of treatment, and the effects of aging and comorbidities. The chapter ends with findings and conclusions.
BACK DISORDERS
Effects on the Ability to Perform Work and Expectations for Improvement
As the number of persons reporting disability among adults in the United States continues to rise (CDC, 2009), musculoskeletal conditions such as chronic back pain are among the most common associated conditions (U.S. Burden of Disease Collaborators, 2018). Only ischemic heart disease, lung cancer, chronic obstructive pulmonary disease, and diabetes mellitus surpass chronic low back pain with respect to the number of years lived with a disability (U.S. Burden of Disease Collaborators, 2018). Adults in the United States with chronic back pain are more likely to be socioeconomically disadvantaged, to be covered by government-sponsored health insurance, and to have more frequent health care visits (Shmagel et al., 2016). Early predictors for work-related disability for adults with chronic back pain include injury severity, recent prior job-related injury, and length of time off work (Turner et al., 2008).
Most individuals with back pain improve substantially over the course of a few weeks (Pengel et al., 2003). Gurcay and colleagues (2009) evaluated 91 participants with acute back pain using multiple instruments assessing severity of pain, specific and general health, and depression. At 2 weeks, 57 percent had fully recovered, and only 9 percent went on to develop chronic back pain (Gurcay et al., 2009).
The initial presentation to a health care provider may determine the short-term outcome for individuals with acute back pain. Positive prognostic
factors for persons with chronic nonspecific low back pain (CNSLBP) include younger age, lack of comorbid clinical conditions, and a lower level or shorter duration of pain intensity at initial assessment. Verkerk and colleagues (2013) evaluated 1,760 individuals with CNSLBP (defined as back pain without a specific etiology, such as radiculopathy, infection, or trauma) who received multidisciplinary therapy. Participants were evaluated for their course of disability and prognostic factors at baseline and at 2-, 5-, and 12-month follow-up evaluations. At the 12-month follow-up evaluation, prognostic factors for recovery included younger age, shorter duration of pain complaint at baseline evaluation, and lack of underlying mental health comorbidity.
Menezes and colleagues (2012) conducted a meta-analysis of inception cohort studies to evaluate the prognosis for acute and chronic back pain. They demonstrated that most individuals with back pain were able to realize substantial improvement in approximately 6 weeks, with more gradual improvement over the next 10 to 11 months. Measures of pain intensity and disability indicate gradual improvement in low back pain. Steffens and colleagues (2014) evaluated 118 consecutive individuals with CNSLBP who were enrolled in a group exercise program that also incorporated cognitive-behavioral therapy. Outcome measures of pain intensity (based on visual analogue scale pain scores of 0–10) and disability (based on the Roland Morris Disability Questionnaire) were made at 12 months. These measures showed that pain intensity had decreased by 39 percent, and disability and function had improved by 60 and 72 percent, respectively.
Although the majority of patients with a new diagnosis of back pain improve substantially, some go on to experience chronic back pain, which can lead to substantial functional incapacity and disability. In one study, approximately 500 participants were evaluated in a primary care setting. Multiple potential predictive factors in relation to low back pain were evaluated (demographic, physical, psychological, and occupational), and participants were again evaluated at 6 months and 5 years. Baseline pain intensity and the individuals’ belief that their back pain would be longstanding were the factors most associated with poor outcomes (Campbell et al., 2013). Other factors found to be associated with a poor prognosis for recovery from back pain include older age, sciatica, functional disability, poor general health, increased psychological stress, negative cognitive characteristics, poor colleague relations, heavy physical work demands, and the presence of compensation (Hayden et al., 2010). Heymans and colleagues (2010) found that having no clinically relevant change in pain intensity and disability status and a high level of pain after the first 3 months were strongly related to developing chronic back pain. In a more recent study, Steenstra and colleagues (2017) evaluated prognostic factors for returning to work after 12 weeks of sick leave among individuals with subacute and
chronic back pain. Such factors as age, functional status, delay in treatment referral, receipt of workers’ compensation, and attorney involvement were negatively associated with returning to work.
Persistent back pain is clearly associated with developing chronic functional disability. Mehling and colleagues (2012) initially evaluated 605 individuals who consulted their primary care providers about acute low back pain. The participants had no history of back pain in the prior 12 months and no history of spine surgery. Follow-up telephone interviews regarding current symptoms and work status were conducted at 6 months and 2 years. The McGill Pain Questionnaire and Roland-Morris Disability Questionnaire were used to assess the participants. The prevalence of chronic back pain and disability was higher than would be expected based on the published medical literature, with 19 percent of the 443 participants who remained in the study reporting chronic back pain and disability. According to Chou and Shekelle (2010), negative predictors for developing disabling chronic back pain are maladaptive pain coping behaviors, nonorganic signs (physical findings without an underlying physiologic or anatomic etiology), functional impairment, poor general health status, and the presence of psychiatric comorbidities.
Effects of Treatment
Treatment for back pain depends on the individual’s clinical presentation, associated findings on physical examination, and pathology as demonstrated by radiological and/or electrodiagnostic tests. Treatment varies from noninvasive approaches such as nonsteroidal anti-inflammatory medications and physical therapy to more aggressive treatments, such as spine injection procedures and surgical intervention (Patrick et al., 2014). Initial treatment for individuals with acute back pain (without neurological deficits or acute radiographic pathology) is typically noninvasive, consisting primarily of nonpharmacologic treatments such as exercise; general physical rehabilitation; and such modalities as acupuncture, progressive relaxation, and biofeedback. Nonpharmacologic treatments for chronic low back pain, including exercise, yoga, psychological therapies, and acupuncture, have demonstrated clinical effectiveness (Chou et al., 2017). Multidisciplinary rehabilitation treatment for persons with low back pain has been shown to be particularly effective (Stein and Miclescu, 2017), with demonstrated improvements for pain management, functional restoration, and quality-of-life measures (Moradi et al., 2012; Morone et al., 2011).
Nonsteroidal anti-inflammatory and muscle relaxant medications are a first-line choice for pharmacologic management (Chung et al., 2013; Qaseem et al., 2017). The use of opioid analgesic medications for the treatment of acute and chronic back pain remains controversial (Deyo et al.,
2015). Recently, prescription of opioid analgesic medications in hospital emergency departments has been discouraged (Lee et al., 2016). Indeed, Krebs and colleagues report that treatment with opioids for individuals with back pain was not superior to treatment with nonopioid medications for improving pain-related function (Krebs et al., 2018), and individuals with a higher rate of prescribed opioid medications are more likely to have associated depression (Smith et al., 2017).
Spine injection procedures utilizing corticosteroids are usually reserved for individuals whose clinical presentation is consistent with radiculopathy (Benoist et al., 2012). However, the benefits of this treatment are often limited and short term (Choi et al., 2013; Chou et al., 2015). Epidural steroid injections may increase function and reduce the need for surgical intervention for individuals with back pain (Bicket et al., 2015; Choi et al., 2016). Surgical intervention is reserved for individuals with structural spinal pathology and associated clinical presentations. It is more likely to be effective if an individual’s symptoms are associated with a structural abnormality, such as spondylolisthesis, stenosis, or disk herniation, and surgical intervention for radiographically demonstrated intervertebral disc herniation, degenerative spondylolisthesis, and spinal stenosis has been shown to be clinically appropriate, with demonstrated efficacy (Abraham et al., 2016), as well as superior to nonsurgical medical management for appropriately selected individuals (Jacobs et al., 2013; Parker et al., 2014).
Multiple factors can determine when and/or whether an individual will return to work after spine surgery for back pain. Huysmans and colleagues (2018a) conducted a systematic review of the medical literature to identify the factors that influence the duration of an individual’s absence from and eventual return to work. They found that important factors diminishing the likelihood of returning to work include older age, female gender, longer duration of preoperative symptoms, and shorter time at preoperative employment. They also found that symptoms of depression after surgery prolong the time before resuming work. Anderson and colleagues (2015) confirmed these predictors, specifically emphasizing clinical depression as a strong predictor of both the outcome of lumbar spine surgery and eventual return to work. Lee and colleagues (2017) reviewed individuals receiving workers’ compensation prior to lumbar spine surgery, and found that patients who had been working at least 3 months prior to their surgery were more likely to resume working within 1 year. Other factors, such as lower education, more physically demanding work, and low income, were also negative predictors of return to work after lumbar spine surgery (Truszczy´ nska et al., 2013). Although many patients do return to work, prediction of postsurgical disability is limited even with extensive evaluation. A detailed model using 39 clinical assessment variables and 38 clinical questionnaire items,
for example, explained only half of the observed variation in postsurgical outcomes (McGirt et al., 2015).
Return to work after an acute episode of low back pain that resolves is generally quite straightforward. Indeed, the individual may never have been removed from work, depending on the severity of symptoms, and likely symptoms resolved with conservative therapy or none at all. However, in cases where back pain is persistent and the patient is removed from the work environment, there are opportunities for intervention beyond the pharmacologic, nonpharmacologic, and surgical interventions discussed above. Physical therapy as a modality toward strengthening the core and other muscle groups, as well as reducing pain and increasing flexibility, has met with success (Shipton, 2018).
In the work arena, work hardening, also known as work conditioning, is a further extension of physical therapy and is often utilized in the work setting. Work conditioning is usually employed when the injured worker remains unable to work because of deconditioning and/or functional deficits that remain despite physical therapy and other noninvasive therapy. Work conditioning involves simulation of the work environment and job tasks in which the individual will be engaged. This is carried out in a highly structured environment with the end goal of functional restoration and return to work (Schonstein et al., 2003). The degree of physicality of the job and the age of the worker are among the factors that can influence the ability to return to work (Huysmans et al., 2018b).
The biopsychosocial approach has also been employed in this arena, based on the tenet that the relationship between pain and disability is not predictive and that an individual’s reaction to injury is mediated to some extent by psychosocial factors (Schultz et al., 2000, 2007). Cognitive-behavioral therapy has also been used to address some of the other factors affecting return to work, including fear avoidance and pain catastrophizing (Besen et al., 2015).
Returning an individual to work in some capacity before full recovery in the work environment has been found to be associated with more rapid improvement in acute low back pain and functional recovery (Shaw et al., 2018). If modified duty is available, it can be used to return the individual to work early with resultant reduced number of days of disability and earlier return to function both at home and at work. A Cochrane database of systematic reviews by Schonstein and colleagues (2003) found evidence that physical conditioning programs that included cognitive-behavioral therapy coupled with intensive physical training by a physiotherapist or a multidisciplinary team encompassing aerobic capacity, muscle strength and endurance, and coordination was associated with reduced number of sick days due to chronic low back pain.
Effects of Aging and Comorbidities
Low back pain is the most common musculoskeletal condition affecting older adults. Approximately 36 percent of individuals aged 65 and older living in the community experience an episode of back pain each year (Cayea et al., 2006), which in the majority of cases is thought to be of mechanical or soft tissue etiology (Weiner et al., 2006). As previously noted, most cases of low back pain resolve over several weeks. However, low back pain tends to be more persistent in older adults. Cassidy and colleagues (2005) conducted a survey by mail to estimate the prevalence of severity-graded low back pain in the general adult population and reported the results after a 1-year follow-up. They found that most episodes of low back pain were mild and tended to resolve in a matter of weeks, but older individuals were more likely to have persistent low back pain and only partial resolution of their symptoms.
The prevalence of chronic low back pain increases with age, with the highest likelihood in the fifth and sixth decades of life (Shmagel et al., 2016). However, the intensity of low back pain has a stronger correlation with disability in younger adults (Houde et al., 2016). In the study by Houde and colleagues (2016), pain measurements were obtained during the initial physician evaluation using a visual analog scale (0 = no pain; 10 = worst pain imaginable) for individuals who presented to a spine center for treatment. Disability also was assessed during the initial visit using the Oswestry Disability Index (ODI). The authors found a significant, positive association between reported pain intensity as measured on the visual analog scale and disability as measured with the ODI for both age groups. The correlation was stronger in the younger (r = 0.66; p < 0.01) than in the older (r = 0.44; p < 0.01) group (Fisher Z = 2.03; p < 0.05), and the linear regression model showed the slope of the relationship to be steeper in the younger group (p < 0.05) (Houde et al., 2016).
CARDIAC IMPAIRMENTS
Effects on the Ability to Perform Work and Expectations for Improvement
Heart Failure
The term “heart failure” refers to any primary heart muscle disease (cardiomyopathy) or secondary impairment of heart function that can arise from multiple causes, most commonly coronary artery disease. To encourage awareness, the current American College of Cardiology/American Heart Association staging system for heart failure defines an asymptomatic Stage B that can be detected as decreased heart function, usually left
ventricular ejection fraction seen on echocardiography or nuclear imaging, but has not yet resulted in clinical symptoms (Yancy et al., 2013). This extension of the term “heart failure” has enhanced recognition and treatment of early disease to prevent progression, but can create unnecessarily negative expectations about the imminence and inevitability of disability and death (Cleland et al., 2017; Stevenson, 2017).
Individuals may occasionally be diagnosed in the asymptomatic stage (Stage B) when decreased cardiac function is recognized incidentally during general screening, during evaluation of a family with known genetic disease, or during evaluation of an acute symptom such as chest pain or palpitations. At this early stage, it would be uncommon for usual activity to be limited, except perhaps severe sustained exertion, such as at the level of competitive sports. Occasionally, asymptomatic cardiomyopathy associated with a risk of sudden life-threatening arrhythmias may warrant prohibition of sudden strenuous exertion or employment requiring commercial vehicles, machinery, or working at heights (Banning and Ng, 2012). Otherwise, Stage B heart failure would not be expected to limit employment. Progression of disease from Stage B is uncertain and unpredictable, with fewer than 5 percent of cases progressing to major symptomatic limitation during the next 4 years (Young et al., 2017).
At the time of first heart failure diagnosis, many individuals experience shortness of breath occurring at rest or upon modest exertion (Class III or IV). These symptoms often result from an accumulation of excess body fluid and improve dramatically within days of instituting diuretic therapy, usually during hospitalization (Kato et al., 2012). Improvement of the underlying cardiac condition over the next few months can represent spontaneous resolution of a reversible cardiomyopathy, such as a viral-triggered inflammation of the heart or the cardiomyopathy associated with pregnancy. Improvement also can occur with correction of a structural abnormality, such as heart valve disease, or a functional abnormality, such as a persistently rapid heart rhythm. Even without such correctable causes, establishment of the recommended regimen of medications and pacing devices can improve heart function and functional capacity in some individuals even after years of heart failure symptoms, with about 15 percent improving to near-normal levels of function (Cleland et al., 2017).
Most issues regarding employability with cardiomyopathy or heart failure are likely to arise in Stage C, which encompasses individuals who have ever had symptoms of heart failure but whose symptoms are not yet considered to be refractory. It is in this stage that most individuals are first diagnosed with heart failure and that much of the impact of modern therapies has been achieved. Within Stage C, symptom severity varies markedly, as described by the New York Heart Association’s (NYHA’s) classification
ranging from Class I, with no limitation on activity, to Class IV, with limitation on any physical movement (see Figure 7-1).
Early in the course of symptomatic heart disease, at the level of NYHA Class II symptoms only with “more than usual activity,” individuals would generally be unable to sustain strenuous physical labor (e.g., moving furniture or leading exercise classes), associated on exercise testing with 7 or more metabolic equivalents (METS) of energy expenditure (see Figure 7-1 and Chapter 5). Current SSA listing criteria for cardiac functional impairment for both heart failure and ischemic heart disease can be met with inability to perform 5 METS of activity (IOM, 2010, pp. 86, 118). The energy
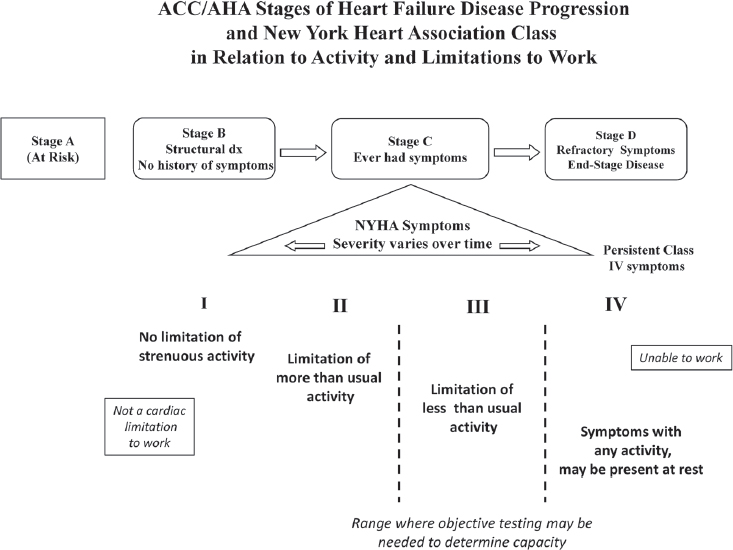
NOTES: Stages A to D describe patients in terms of disease progression from no history of symptoms through refractory symptoms, with the general implication that most patients move only from left to right. In contrast, the New York Heart Association’s (NYHA’s) symptom classification (Roman numerals) is dynamic, such that patients frequently shift from no limitation to severe limitation and back to mild limitation, usually as the result of changes in therapy. NYHA class is defined in terms of severity of activity limitation. The range between Classes II and III is where objective testing may be needed to determine actual functional capacity, as an individual in Class I would not be impaired on a cardiac basis, and Class IV symptoms would preclude usual employment. ACC = American College of Cardiology; AHA = American Heart Association; dx = diagnosis.
level of 5 METS is the estimated requirement for doing, for example, light work in a bakery or small carpentry at a bench. This leaves only a narrow, uncertain margin within which typical cardiac conditions would limit an individual’s ability to perform common indoor occupations without reaching the SSA listing criteria for disability.
Commonly cited is the statistic that 50 percent of individuals hospitalized with heart failure will die within 5 years, but national data in the United States are dominated by Medicare populations, in which the average age of individuals with heart failure is more than 75. By contrast, in the nationwide Danish registry of 21,455 individuals aged 18 to 60 at the time of their first hospitalization for heart failure, 55 percent were working, and 68 percent of them had returned to the workforce within 12 months (Rørth et al., 2016). Return to work was more common with younger age, male gender, and higher education, but it was substantially less common in the presence of comorbidities, particularly kidney or lung disease (Rørth et al., 2016). In general, the more severe the clinical symptoms and impairment of cardiac function at presentation, the less likely it is that substantial improvement will occur. A recent large prospective 3-year study following Stage C heart failure typical of individuals under age 70 demonstrated that most individuals remained stable without progressive deterioration of symptoms during the 3 years. Each year, about 5 percent died without prior change in their disease stage, and 5 percent deteriorated to Stage D (Kalogeropouolos et al., 2017), the end stage of heart failure.
Individuals with Class IV symptoms that no longer improve with therapy but have become refractory are considered to have progressed to Stage D, with expected survival of less than 1 year (Yancy et al., 2013). Highly selected patients with good self-care and without comorbidities may be appropriate for consideration of cardiac transplantation, durable mechanical circulatory support devices, palliative care, home intravenous inotropic therapy, or hospice care (Yancy et al., 2013). As contemporary therapies for heart failure have slowed the progression of heart failure, they have also markedly reduced unexpected sudden deaths prior to deterioration to Stage D disease, such that the prevalence and duration of severely symptomatic heart failure continue to increase (Udelson and Stevenson, 2016).
Coronary Artery Disease
Symptoms of angina during exercise or other stress arise most commonly from narrowing of the coronary arteries due to atherosclerosis. Most individuals with potentially limiting symptoms resulting from coronary artery disease undergo procedural interventions to enlarge significantly narrowed coronary arteries or surgery to bypass them, assuming they have no comorbidities that limit eligibility for major cardiac surgery.
Evaluation of functional capacity usually occurs following initial appropriate interventions.
Many individuals with coronary artery disease present first with myocardial infarction (heart attack). A previous myocardial infarction that was small or that was effectively treated early is a common cause for Stage B heart failure. Even before the modern era of effective early intervention, the majority of individuals returned to work within 3 months following myocardial infarction (Froelicher et al., 1994). Nonetheless, while coronary artery disease can lead to functional impairment due to angina or myocardial infarction or after cardiac surgery, much of the disability resulting from coronary artery disease relates to a progressive decrease in heart function after myocardial infarction, which is the most common cause for heart failure (Cahill and Kharbanda, 2017).
Of interest, a strong predictor of return to work was an individual’s prediction of return to work made before coronary artery bypass surgery or in the hospital early after myocardial infarction (Shanfield, 1990; Sivarajan and Newton, 1984). Indeed, the capacity to continue or return to work with disease does not correlate well with the measured severity of the disease (Sullivan et al., 1997). For patients at multiple stages of coronary artery disease, depression is a key factor predicting impairment and disability (Papakostas, 2009). Depression is one of the most intensely studied predictors of outcome, present in about one of every five patients hospitalized with myocardial infarction, most of whom continue to have depression months later (Bush et al., 2005). Depression after myocardial infarction is associated with poorer quality of life and increased risk of death, with a less consistent relationship being demonstrated with nonfatal cardiac events.
For patients with coronary artery disease, concern may arise regarding the effect of perceived stress in the work environment. Work stress has been associated with a 10 to 40 percent increase in the incidence of a first heart attack, but work stress is difficult to isolate as patients likely to perceive work stress may have other factors that increase their cardiac risk. This association has not been considered sufficient to warrant any targeted recommendations for patients at risk for cardiac disease (Kivimäki and Kawachi, 2015). Patients who perceived major job stress were less likely to return to work after a myocardial infarction, but it was not possible to isolate that effect as it was inversely correlated with job satisfaction. A large prospective study continued over 2 years demonstrated that perception of high stress at the time of initial return to work after a heart attack did not have a major impact unless stress was very high. However, the maintenance of stress over time was associated with almost twice the risk of recurrent heart attack, and almost eight-fold higher risk in patients who also had major impairment in cardiac function after the initial heart attack (Aboa-Éboulé et al., 2007).
Effects of Treatment
Current assessment of physical function and survival with heart failure and coronary artery disease reflects widespread use of medications, devices, and procedures recommended for these diagnoses (Fihn et al., 2014; Yancy et al., 2013). Many medications prescribed for heart failure and coronary artery disease overlap, particularly for individuals with hypertension. Individuals with recent myocardial infarction receive therapies to delay or prevent progression to heart failure. As most of these therapies improve disease prognosis and many help decrease the symptoms of heart failure, their use is expected to improve rather than limit the ability to work. Medications may occasionally cause a transient drop in blood pressure for a few seconds upon standing after prolonged sitting, which may impair the ability to move rapidly at short notice. Individuals with implantable pacemakers and/or defibrillators in their shoulder area will have some restriction on vigorous pulling and stretching with their arm on that side. The most common treatment requiring work adaptation is the use of diuretics by individuals with fluid retention in heart failure. The appropriate timing of diuretic administration may require individuals to use the bathroom several times during the morning, and the rapidity of drug action may require that they be located close to bathroom facilities during these periods. In terms of the conceptual framework described in Chapter 2 (see Figure 2-3), these factors associated with the use of diuretics would be considered interrupters, because they interfere with the ability to perform sustained work activities on a regular and continuing basis.
Return to work after revascularization procedures and cardiac surgery is less frequent than would be expected given the favorable impact of these treatments on reducing cardiac symptoms. Following revascularization with either surgery or catheter-based procedures as performed before 2000, almost one-third of individuals did not return to work, and about one in five rated their health as fair or poor (BARI, 1997), this despite the relief of angina in more than 70 percent of individuals having undergone these procedures to improve coronary artery blood flow (BARI, 2007). In another report on coronary artery bypass surgery, 90 percent of individuals described symptom relief, but only 50 percent returned to work. Of those who did not return to work, only 30 to 40 percent gave “heart problems” as the reason. The likelihood of returning to work increased with higher socioeconomic status, more education, and “looking forward to returning to work” (Mital et al., 2004).
Cardiac rehabilitation is routinely recommended for patients after myocardial infarction or coronary artery bypass surgery and more recently approved for patients with heart failure. Despite the clear benefits to improve survival and quality of life, fewer than one-third of eligible patients
participate in cardiac rehabilitation programs (Jolliffe et al., 2001). Some of the limitations are insurance coverage and access to nearby facilities. Exercise capacity and quality of life have frequently improved in large studies. Return to work has occasionally been documented in the United States (Williams et al., 2006), but many patients are already past retirement age at the time they acquire a cardiac indication for rehabilitation. Rates of return to work are more often reported for studies outside the United States, and are higher with earlier enrollment in rehabilitation (Samkange-Zeeb et al., 2006). As with other populations, return to work is more common with younger age and higher patient expectations to return to work.
Effects of Aging and Comorbidities
In considering the impact of cardiac conditions on exercise capacity, it should be noted that the expected “normal” levels of exercise capacity decline substantially with older age. For instance, an exercise performance of 7 METS, needed for heavy carpentry, would be 50 percent of normal for a 40-year-old man and 90 percent of normal for a 70-year-old man (Froelicher et al., 1992). Most cardiovascular exercise impairments are described in terms of proportional decrease such that the absolute diminution is greater from a higher predicted normal value. However, little work has been done to compare cardiac impairment in exercise capacity across age groups.
In general, the limitation imposed by the severity of the cardiac condition outweighs the contribution from chronologic age. However, age is associated with increasing comorbidities, which substantially increase the physical limitation and likelihood of disability due to heart disease (Forman et al., 2018). Atrial fibrillation is a cardiac comorbidity that increases dramatically with age, present in about 0.1 percent of patients aged 40 to 50 and 1.8 percent of those aged 60 to 70, commonly aggravating symptoms of both heart failure and coronary artery disease (Feinberg et al., 1995). Kidney disease and pulmonary disease are the most common medical comorbidities diminishing physical function in people with cardiac disease. The increasing prevalence of diabetes and obesity in older age is anticipated to further limit functional capacity and prognosis for patients with heart failure (Christiansen et al., 2017).
A study following patients after cardiac catheterization showed that physical function as assessed by functional status questionnaires converged to the same level by 6 months regardless of the number of diseased vessels, but remained strongly correlated separately with depression and anxiety (Sullivan et al., 1997). Depression occurs in 20 to 40 percent of patients with heart failure and in a similar proportion of individuals with coronary artery disease (Bush et al., 2005; Mbakwem et al., 2016). The combination is correlated with increased mortality for both conditions. Intervention to
relieve depression has been more effective after myocardial infarction than for heart failure, but has not had demonstrable benefit for survival. Unlike most comorbidities, however, concomitant depression with heart failure may be reported less commonly by older individuals than by those who are younger (Gottleib et al., 2004).
DEPRESSION
Effects on the Ability to Perform Work and Expectations for Improvement
Extensive evidence indicates that depression negatively affects functioning relevant to work (Adler et al., 2006; Kessler et al., 2003b; Lerner et al., 2004; Mosbach et al., 2018). In understanding where along the spectrum of depression severity an individual’s ability to perform functions relevant to work requirements is affected, it is important to consider how depression can affect job performance. Symptoms of depression include not only sadness but also symptoms that directly reduce functioning, such as inability to attend and concentrate, psychomotor agitation or retardation, and fatigue or loss of energy (Appelbaum, 2018). Preoccupation with death or suicide, guilt, and inability to experience pleasure also may reduce work functioning (Appelbaum, 2018).
Contributors/Correlates of Depression-Related Disability
Given the relatively high prevalence of depressive disorders, it is important to note that the vast majority of individuals experiencing a depressive disorder do not develop severe work-related disability that results in an inability to work. It is thus important to understand and identify the critical factors that contribute to depression-related disability. To this end, one can ask about (1) factors predisposing to depression-related functional impairment, (2) factors related to the depression itself that increase vulnerability to functional impairment, (3) important co-occurring disorders, and (4) factors related to the nature of the job that interact with depression-related work disability.
Predisposing factors A variety of sociodemographic factors are associated with work-related disability among individuals who are depressed. Ervasti and colleagues (2013) found that work-related disability from depression was associated with lower level of education, lower position in job, and renting versus owning a home (Mosbach et al., 2018). Elinson and colleagues (2004) found that among individuals with depression, those aged 18 to 24 were more likely to work than those aged 55 to 69; men and white individuals were more likely to work than women and black individuals; and those with
higher levels of education were more likely to work than those with lower levels. The Finnish Vantaa Depression Study (VDS) examined factors related to social and occupational disability, social adjustment, and work disability among individuals with major depressive disorder (MDD) (Rytsälä et al., 2005). Severity and recurrence of depression were the most important factors associated with level of social, functional, and work disability; older age and current Axis I and II comorbidity contributed significantly as well. The study also found that older age, greater sense of hopelessness, worse social and occupational functioning, and persistence of depression predicted receipt of a disability pension (Rytsälä et al., 2007). Sorvaniemi and colleagues (2003) conducted a retrospective study of 213 adult psychiatric outpatients with first-time documented major depression as defined in the Diagnostic and Statistical Manual of Mental Disorders, Third Edition Revised (DSM-III-R) and found that older age, comorbidity, and lowered self-esteem were strongly associated with being granted a pension. Taken together, these findings indicate that older age is the most consistently observed factor contributing to work-related disability in the context of depression.
Depression-related factors Not surprisingly, overall severity of depression has been associated with loss and impairment of productivity. According to Lerner (2008), severity of depression symptoms has repeatedly been shown to account for some of the variation in work outcomes. A study of the employed baseline sample in the Depression Improvement Across Minnesota: Offering a New Direction (DIAMOND) study (N = 771) found that for every increase of 1 point on the Patient Health Questionnaire-9 (PHQ-9), productivity as measured by the Work Productivity and Activity Impairment Questionnaire declined by an additional 1.65 percent (Beck et al., 2011). Consistent with that result, individuals with MDD are most at risk for disability days due to depression, although minor depression can also lead to disability days (Broadhead et al., 1990). As noted above, the VDS found that severity and recurrence of depression were critical predictors of occupational disability and that greater sense of hopelessness and persistence of depression predicted receipt of a disability pension (Rytsälä et al., 2005, 2007). McKnight and Kashdan (2009) conducted an extensive review of the correlation between clinical measures of depression among individuals with MDD and overall functional impairment. They found moderate to strong correlations across clinical samples of all ages. Occupational impairment (Aikens et al., 2008; Hannula et al., 2006; Hirschfield et al., 2002) showed similarly robust correlations with depression severity. Importantly, the association between depression and occupational impairment was found to be greater from assessments obtained during treatment (b = 0.22; t = 3.27, p < 0.008) and posttreatment (b = 0.33; t = 4.16, p < 0.002) than from those obtained at baseline (McKnight and Kashdan, 2009).
Previous research has examined whether specific symptoms of depression negatively affect the degree of work impairment. Studies have found low energy or fatigue, psychomotor disturbance, and low interest or pleasure (Sanderson et al., 2007); difficulty concentrating or being fidgety, feeling tired, or sleep disturbance (Lerner et al., 2004); and memory problems, anxiety, and irritability (Lam, 2012) to be associated with depression-related work outcomes.
Depression severity appears to be the most important clinical depression-related factor associated with disability (Lerner, 2008). It is also clear, however, that levels of depression severity that fail to meet the threshold for a diagnosis can be associated with marked role impairment (Wells et al., 1989).
Co-occurring disorders An emerging body of research documents the combined effects of mental health disorders such as depression and physical health disorders on work-related disability (Kessler and Frank, 1997; Rytsälä et al., 2005; Wells et al., 1989). Buist-Bouwman and colleagues (2005) analyzed data from the Netherlands Mental Health Survey and Incidence Study and found that all physical disorders, except injury caused by accident, were significantly related to anxiety and mood disorders. Both physical and mental disorders were significantly related to work loss; notably, the physical–mental comorbidity was largely additive except for chronic back pain and hypertension, which interacted with mental disorders synergistically. Other studies have documented the association of comorbidity upon receipt of disability pensions (Rytsälä et al., 2007; Sorvaniemi et al., 2003).
Nature of the job In addition to factors relating to the individual, the effects of depression vary by job type. For example, Lerner and colleagues (2004) studied factors related to productivity among employed individuals with depression. They found that reduced productivity was most influenced by depression severity (p < 0.01 in 5/5 models); however, deficits increased when employees had occupations requiring proficiency in decision making and communication and/or frequent customer contact (p < 0.05 in 3/5 models).
Measuring Depression and Work Impairment
Understanding of where along the spectrum of depression severity an individual’s ability to work is affected is limited by the fact that most accepted clinical measures of depression do not assess an individual’s functional capacity (Harvey et al., 2017). As is true for all mental disorders, the DSM-5 diagnosis of both major and persistent depressive disorders requires
that the “symptoms cause clinically significant distress or impairment in social, occupational, or other important areas of functioning” (APA, 2013, pp. 95, 98). (Persistent depressive disorder, previously called dysthymia, requires fewer depressive symptoms relative to MDD, and is sometimes termed “minor” depression [APA, 2013].) Therefore, functional impairment is a component of the diagnosis of depression, distinct from clinical symptoms. Understanding the extent to which commonly used depression measures provide relevant information about work-related impairment and whether there are measures of work-related functional ability that are useful for individuals experiencing major depression should provide a starting point for understanding where along the spectrum of depression severity work-related disability may occur.
A review by McKnight and Kashdan (2009) identifies three studies that provide correlations of the Hamilton-Depression Rating Scale (HAM-D) (Hirschfeld et al., 2002), the Symptoms Checklist Depression Scale (SCL)-20 (Aikens et al., 2008), and the SCL-90 (Hannula et al., 2006) with functional measures related to occupational functioning. There does not appear to be a consistent correlation or cutpoint over time for the HAM-D, and insufficient data are available to comment on the SCL-90 and SCL-20. Regarding the PHQ-9, the general standard is that a cutpoint of 15 or above is indicative of moderately severe depression, which generally corresponds with work impairment (Kroenke et al., 2001; Lerner et al., 2004, 2012, 2015). Studies by Adler and colleagues (2006) and Lerner and colleagues (2015) have found that a cutpoint of 13 captures additional people with substantial work impairment from depression.
A number of other scales have been used in a limited way to assess the impact of depression on work-related impairment instead of measuring depression per se. None of these scales has achieved widespread use, and all have limited utility. The Lam Employment Absence and Productivity Scale is a measure of work functioning that has been validated for individuals with MDD. It has been shown to have good internal consistency validity and demonstrated to measure “work productivity and troublesome symptoms” among individuals with depression (Lam et al., 2009). The Depression Prognosis Index (DPI) takes various factors into account to predict depression outcomes. Similarly, the 36-Item Short Form Health Survey (SF-36) takes account of social functioning among other factors, within which work functioning is assessed. The DPI has been found to be a valid measure of depression outcomes, including work functioning as measured by the SF-36 (Rubenstein et al., 2007). The Endicott Work Productivity Scale (EWPS) also has been used to assess work function among depressed individuals. Hellerstein and colleagues (2014) used the EWPS to assess the impact of behavioral activation on work productivity among individuals with depression.
A recent study compared individuals with MDD and healthy controls using the World Health Organization Disability Assessment Schedule 2.0 (WHODAS 2.0), which provides measures of “real-world functioning” relevant to work (Milanovic et al., 2018). The “life activities” subscale of the WHODAS was used to measure functional disability and the “getting along with other people” subscale to measure interpersonal functioning. This study evaluated task performance as well as self-rated competence. The MDD group performed worse than controls on competence tasks, and reported greater functional disability and lower self-perception of competence.
Effects of Treatment
A critical factor in addressing the question of expected improvement in depression is the receipt of appropriate treatment. While data vary, a consistent finding across multiple studies is that fewer than half of people with depression receive treatment, and even fewer receive evidence-based treatment (Stewart et al., 2003). Kessler and colleagues (2003a) estimated that only 21.7 percent of Americans with depression received adequate treatment in the National Comorbidity Study Replication. Therefore, the assumption of treatment in determining the expected course of depression-related impairment is not concordant with the average person’s experience.
What happens to untreated depression? Stegenga and colleagues (2012) followed a cohort of individuals with MDD in primary care. They found that 17 percent had a chronic and 40 percent had a fluctuating course, while 43 percent remitted. Individuals with chronic courses had more severe depressive symptoms (mean difference 6.54; 95% confidence interval [CI] 4.38–8.70), somatic symptoms (mean difference 3.31; 95% CI: 1.61–5.02), and greater mental dysfunction (mean difference 10.49; 95% CI: –14.42 to –6.57) at baseline relative to those who remitted from baseline, independent of age, sex, level of education, presence of a chronic disease, and a lifetime history of depression (Stegenga et al., 2012).
Schoenbaum and colleagues (2002) underscore the importance of treatment. They evaluated the effects of depression treatment in primary care on individuals’ clinical status and employment over 6 months. Data were drawn from a randomized controlled trial of quality improvement for depression that included 938 adults with depressive disorder in 46 managed primary care clinics in five states. At 6 months, individuals with appropriate care had lower rates of depressive disorder (24 versus 70 percent), better mental health–related quality of life, and higher rates of employment (72 versus 53 percent) (each p < 0.05) compared with those without such care.
At the same time, symptomatic improvement does not lead automatically to functional improvement. Lerner and colleagues (2015) emphasize
the value of high-quality depression care in reducing unemployment, absenteeism, and presenteeism (i.e., at-work performance deficits) (Rost et al., 2004, 2005; Schoenbaum et al., 2002). However, they note that treating depression symptoms does not always restore the ability to work. Studies have shown that even after improvement in depressive symptoms, residual functional limitations persist, including limitations in performance of work activities (Buist-Bouwman et al., 2004; Hirschfeld et al., 2002; Judd et al., 2000; Lerner et al., 2011). The large Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, for example, found that individuals who responded to first-line antidepressants had reduced work-related impairments. At the same time, however, individuals who did not respond to first-line antidepressants had persistent work-related impairment even when second-line antidepressants eventually reduced their symptoms (Trivedi et al., 2013). Furthermore, other factors, such as chronic general medical and mental comorbidities, influence work-related impairment (Adler et al., 2006; Lerner et al., 2010, 2012).
Adler and colleagues (2006) found further evidence of the persistent impact of depression severity on job performance among employed primary care patients. The study groups included 286 individuals with DSM-IV MDD and/or dysthymia, 93 individuals with rheumatoid arthritis, and 193 depression-free healthy control subjects. Measurements form the Work Limitations Questionnaire showed the depression group had significantly greater deficits in managing mental-interpersonal, time, and output tasks. The rheumatoid arthritis group had significant deficits in managing physical job demands relative to other comparison groups. Although symptom severity helped to predict improvements in job performance, the “job performance of even the clinically improved subset of depressed individuals remained consistently worse than that of the control groups” (Adler et al., 2006).
An additional question regarding the effects of depression treatment is the extent to which treatment improves cognition, which may improve work-related impairment. A large European study found that cognitive symptoms were problematic in depressed persons but did see some improvement with treatment, albeit to a lesser degree than was the case for depressive symptoms (Hammer-Helmich et al., 2018). This large 2-year European observational study of 1,159 outpatients treated with antidepressants found immediate improvement in depressive symptoms and functional impairment following initiation or switch of antidepressant monotherapy, followed by more gradual improvement and long-term stabilization. Improvements in cognitive symptoms were less marked during the acute treatment phase. Functional impairment in individuals with MDD was found not only to be associated with severity of depressive symptoms but also to be independently associated with subjective cognitive symptoms
after adjustment for depression severity throughout the 2 years of followup. The authors emphasize the importance of cognitive impairment as a limiting factor in recovery of functioning from depression. As discussed further below, Drake and colleagues (2013) found that supported employment services are associated with improved work functioning in people with major depression. Such services can help compensate for the impairing effects of symptoms on the ability to function at work and improve quality of life (McGurk et al., 2018). A recent large randomized controlled trial considered the question of whether cognitive problems improve with antidepressant treatment (Shilyansky et al., 2016). The international Study to Predict Optimized Treatment in Depression (iSPOT-D) assessed the effects of acute antidepressant treatment among 1,008 outpatients, across clinical remission outcomes, on a range of cognitive domains. Impairment in the five domains of attention, response inhibition, verbal memory, decision speed, and information processing speed showed “no relative improvement with acute treatment, irrespective of antidepressant treatment group, even in individuals whose depression remitted acutely according to clinical measures” (Shilyansky et al., 2016, p. 2). In addition, while broader cognitive impairment was associated with greater illness chronicity, it was not associated with symptom severity or previous antidepressant failures. Timing and potential lag as well as extent of improvement are important factors in the impact of depression treatment on functional outcomes. According to McKnight and Kashdan (2009), functioning tends to be less responsive than symptoms to treatment, and therefore functional improvement may take longer than symptom improvement.
In the case of MDD without comorbidity, it appears that improvement can be expected with treatment, although the extent of the improvement and whether it would allow an individual to return to work are uncertain. Persistent symptoms, particularly cognitive symptoms, may impact work-related functioning even in cases of improvement or remission in depressive symptoms. The limited available evidence suggests that the impact of comorbid illnesses is likely to be marked. Evidence suggests that comorbid conditions will increase both the likelihood of depression-related disability and the extent of the work impairment, although the effect in most cases is additive. Without treatment for those conditions, reduction of overall work-related disability is unlikely.
In cases of comorbidity, it is critical to consider the relationship between the comorbid condition and depression. To the extent that depression is exacerbated by or due to another condition, treatment of the second (or third) condition is likely to reduce the severity of depressive symptoms. Notably, the National Comorbidity Study found that almost 80 percent of individuals with 12-month Composite International Diagnostic Interview (CIDI) MDD had comorbid CIDI DSM mental disorders, with MDD only
rarely being primary (Kessler et al., 2003a). A recent study of the costs of MDD for treated employed individuals in the United States found that only 40 percent of the incremental costs was due to MDD. The remainder was due to the costs of treatment for other mental health diagnoses (9 percent), as well as non–mental health conditions (29 percent) and associated prescription costs (13 percent) (Greenberg et al., 2015).
Individuals with treatment-resistant depression (including those with persistent depressive disorders) are a key example of how the associated cognitive impairments (i.e., concentration, attention), as well as the physical/somatic manifestations (i.e., disturbed sleep, fatigue, lack of energy), impair individuals’ work (and social) functioning, in some cases leading to disability claims. Instruments such as the Work Limitations Questionnaire can be used to assess the specific functional work impairments, and treatments that address these deficits exist (Lerner et al., 2004).
Regarding side effects of antidepressants, Lam (2012) found that individuals reported the medication side effects interfering most with work functioning to be daytime sedation, insomnia, headache, and agitation/anxiety.
As noted above, there is evidence of benefits of vocational rehabilitation, and in particular, supported employment, the only evidence-based practice for improving the capacity for competitive work in people with serious mental illnesses (Drake et al., 2012), including major depression (Drake et al., 2013). Principles of supported employment emphasize rapid deployment of services for the job search, a focus on competitive work, job support services that are consistent with client preferences, integrated mental health and employment services, and time-unlimited follow-along supports. Supported employment produces superior competitive work outcomes as compared with every other form of vocational services. Longer-term outcomes of supported employment do not include routine disengagement from receipt of Social Security Disability Insurance as full-time work is rarely achieved, including in people with severe mental illness, with fear of loss of benefits being a contributor to the predominant part-time work outcomes of supported employment.
Effects of Aging and Comorbidities
Advanced age appears to be a relatively consistent correlate of disability related to depression. This relationship may be due to the additive effects of each episode on work functioning rather than the aging process and its functional decrements, and is independent of other measured factors. As individuals with depressive disorders age, those who have certain chronic comorbid conditions (particularly pain, insomnia, musculoskeletal disorders, and diabetes) find that their depressive symptoms further impair their capacity to function. The key issue is whether the association of increased
impairment with depression as people age is due to comorbidities rather than age per se.
MDD occurs more frequently among individuals with chronic medical conditions than among those in the general population (Kang et al., 2015). According to Kessler and colleagues (2005), the depression rate for individuals with medical disorders is approximately two to three times greater than the depression rate for those in the general population. In a 1-year prevalence study conducted by the World Health Organization (WHO), which included 245,400 individuals from 60 countries, 9.3 to 18 percent of those with a single physical disorder experienced depression. Among those with two or more medical conditions, 23 percent experienced depression, compared with just 3.2 percent of medically well individuals (WHO, 2008). According to the Centers for Disease Control and Prevention (CDC, 2012), individuals with the following chronic health disorders experience MDD at higher rates than those in the general population: Alzheimer’s disease (11 percent), cardiovascular disease (17 percent), cerebrovascular disease (23 percent), diabetes (27 percent), cancer (42 percent), and Parkinson’s disease (51 percent). Strokes are also highly correlated with depression, which is experienced by approximately 31 percent of stroke survivors (Hackett and Pickles, 2014).
While depression often co-occurs with physical health conditions, depression and other mental health disorders also co-occur at high rates. According to Kessler and colleagues (2003a), data from a large-scale national survey revealed that 72.1 percent of individuals who met criteria for lifetime MDD also met criteria for another mental health disorder. The most common of these comorbidities were anxiety disorder (59.2 percent), substance use disorder (24 percent), and impulse control disorder (30 percent).
TRAUMATIC BRAIN INJURY
Traumatic brain injury (TBI) is a leading cause of death and disability worldwide, with estimates of 1.6–3.8 million new cases per year in the United States alone (Shames et al., 2007, as cited in Scaratti et al., 2017). TBI is defined as damage to the brain caused by external mechanical force to the head resulting, for example, from falls, being struck by or against objects, motor vehicle accidents, assaults, sports-related concussions, and blast injuries (Little et al., 2015). Returning to the same occupation where the brain injury occurred may be unsafe. This most commonly occurs in military veterans and first responders, and in such cases, consideration should be given to skill development for a different occupation (Little et al., 2015). The risk of TBI is age dependent, as is the impact on work functioning (Little et al., 2015). For children aged 0–4, TBI is typically due to falls and abuse;
for adolescents, the risk is associated with the legal age of driving; and occupationally related TBI is associated with professional drivers, construction workers, first responders, professional athletes, and military personnel, with the presence of extreme stress at the time of injury diminishing injury recovery in first responders and military personnel (Little et al., 2015). The most prevalent group experiencing TBI is children and youth from infancy through adolescence as the result of falls, abuse, and accidents. TBI is approximately three times more common in men than in women. Sports- and occupationally related TBI is associated with multiple lifetime TBIs. With the exception of blast injuries, TBIs are typically due to acceleration–deceleration, with additional risk of rotational components associated with optic nerve and hippocampal damage and cerebral vasculature bleeds.
TBI severity is classified as mild (less than 30 minutes loss of consciousness [LOC], 13–15 Glasgow Coma Score [GCS], less than 24 hours posttraumatic amnesia [PTA]); moderate (30 minutes–24 hours LOC, 9–12 GCS, 24 hours–7 days PTA); or severe (greater than 24 hours LOC, 3–8 GCS, more than 7 days PTA). TBI severity classification based on these acute TBI variables is only grossly predictive of outcome. In general, persons sustaining milder injuries achieve better recovery and less long-term impairment relative to those with more severe injuries.
Mild TBI, or concussion, is the most common type of TBI, accounting for 70–90 percent of all cases (CDC, 2007). These estimates are imprecise, however, because mild TBI cases often are not documented or consistently diagnosed (Cancelliere et al., 2014).
Effects on the Ability to Perform Work and Expectations for Improvement
TBI is associated with a broad range of motor, sensory, emotional, and cognitive impairment. Symptoms include fatigue, irritability, impulse control problems, self-centeredness, headaches, dizziness, sleep disturbance, problems with balance and coordination, anxiety, and depression, as well as impairment in memory, attention, information processing speed, planning, and problem solving (Little et al., 2015). Most individuals with mild TBI will recover within 1 year, but 5–20 percent of individuals may have persistent problems (Losoi et al., 2016; Rao et al., 2010). Symptoms can resolve in months, but in some cases, sequelae can last years. The course and prognosis of TBI are complicated by multiple trauma, such as damage to the spinal cord, orthopedic injuries, and chest trauma. Cognitive impairment may resolve for people with mild TBI within 3 months, but up to 18 percent of those with mild TBI experience persistent symptoms and functional deficits that can last from months to years postinjury, with as many as 18 percent of those who were employed premorbidly remaining unemployed at 12 months post-TBI (Cancelliere et al., 2014). Individuals
with psychiatric comorbidities prior to experiencing TBI were found to be at increased risk for relapse and to have lower return-to-work rates following their injury (Garrelfs et al., 2015).
A substantial literature exists on the relationship between TBI and work functioning, but this literature does not yield definitive conclusions about the impact of TBI on work because of the heterogeneity of injuries, definitions of TBI and its degrees of severity, demographic characteristics of the injured individuals, methods used to measure both the injury and employment, lengths of follow-up, treatments received, and systems of care involved in assessment and treatment (Cancelliere et al., 2014). The WHO Collaborating Center Task Force for Mild Traumatic Brain Injury determined that the studies reviewed addressing return-to-work rates in people with TBI were of low scientific quality, and no acceptable studies on return to work addressed physical, cognitive, or emotional barriers (Cancelliere et al., 2014).
A recent systematic review of the literature from 1993 to 2015 addressed specific work-related difficulties associated with TBI, defined as job instability over 5 years postinjury, as well as determinants of these difficulties, focusing primarily on return to work (Scaratti et al., 2017). The authors first conducted a systematic assessment of the quality of studies, judging them to be poor, acceptable, good, or excellent based on National Institute for Health and Care Excellence guidelines, and excluding poor-quality studies from the review. They identified 42 papers meeting their inclusionary criteria, encompassing a total of 25,756 people: 29 percent were female, the average age was 34.9 years, mean time from acute event was 14.6 months, 51.4 percent had severe TBI, 14 percent had moderate TBI, and 33.5 percent had mild TBI, with multitrauma information being available in 9 of the 42 studies. The mean employment rate postinjury was 42.5 percent for all TBI, although information on employment in these studies was quite limited (Scaratti et al., 2017).
Demographic variables that predicted work-related difficulties included older age at the time of injury (older age being defined as 35–54 versus younger than 34); female gender (females being found to have lower workforce participation, including reduced hours or unemployment); lower education (those without a high school diploma having the greatest difficulties compared with those with a high school diploma, and the least difficulty occurring in those with a college degree); preinjury unemployment status; preinjury unmarried status; being a manual worker; and having a minority status. Features of the injury associated with work-related difficulties 5 years postinjury included its severity as indicated by a low GCS and lengthy PTA; a violent cause of the TBI (as compared with a car accident); and polytrauma (such as spinal cord injury). Functional status at discharge had prognostic implications for return to work, including requiring assistance
with functioning and lower scores on measures of motor and cognitive functioning (Scaratti et al., 2017).
The importance of cognition is highlighted by studies indicating that employment at 1 year postinjury was 82 percent lower for those with lower cognitive scores, as determined by the Functional Independence Measure. The presence of factors directly or indirectly related to cognitive functioning, such as self-centeredness, inappropriate social behavior, impulsivity, and irritability, predicted poorer work functioning 1 year postinjury (Scaratti et al., 2017). In addition to cognitive impairment, other factors found to be related to lower rates of return to work were behavioral problems such as confusion, agitation, inappropriate behavior at admission, and discharge to rehabilitation. Individuals with personality changes in particular were found to be 10 times less likely relative to those without such changes to return to work at 18 months postinjury. Apathy, depression, agitation, posttraumatic stress disorder, and psychosis also have been identified as common behavioral sequelae of TBI, with apathy and depression occurring in more than 50 percent of cases (Scaratti et al., 2017).
Posttraumatic pain and headache also have been identified as impacting return to work (Little et al., 2015). Headache is reported as the most common symptom after TBI and is one of a constellation of symptoms of postconcussive syndrome, with highest rates in mild TBI. Because headache is self-rated, episodic, and variable in course within and among individuals, its impact on return to work is largely unknown. Chronic posttraumatic headache (CPTH) reportedly resolves in a few months postinjury, with 25 percent of individuals still having symptoms after 4 years. CPTH—which has been explained by insufficient treatment; analgesic rebound; and psychosocial comorbidities such as anxiety, depression, and insomnia—is associated with occupational disability. Among people with CPTH, 12 percent report missed workdays and annual absenteeism of an average of 27 days, with greater reduced activity level, more disability, and lower physical function relative to those with nontraumatic chronic headache (Little et al., 2015).
Effects of Treatment
Returning to work promotes recovery from TBI and is associated with improved well-being, health status, and quality of life (Little et al., 2015). Successful return to work is aided by having health insurance; receipt of supported employment; tailoring of work duties based on preserved abilities; a socially inclusive work environment; and reduced stress at work, such as avoiding frustration associated with having to relearn prior performed tasks, avoiding the need to multitask, and diminishing the need to learn new skills (Little et al., 2015). Reviews consistently mention the need for
vocational rehabilitation to help people with TBI return to work. Supported employment is also encouraged given its supports and services for the range of physical, psychological, and cognitive sequelae associated with TBI. A few studies have shown enhanced return-to-work rates in people receiving such services. However, randomized controlled studies are needed to further understand whether and how vocational rehabilitation enhances return to work in people with TBI (Mani et al., 2017; Scaratti et al., 2017).
Effects of Aging and Comorbidities
Individuals who are older at the time of their TBI have worse outcomes (e.g., are less likely to return to work) compared with younger people with similar TBI, with “older” being defined in some studies as ages 35–54 (Scaratti et al., 2017). The physical effects of aging also may make older workers more vulnerable to injury, including head injury (Kristman et al., 2010). In addition, older people are more likely to have progressive age-related cognitive decline, which also contributes to diminished rates of return to work, as well as greater difficulties on the job, following TBI (Marquez de la Plata et al., 2008).
SUMMARY AND COMPARISON OF COMMON IMPAIRMENTS
Trajectories
The trajectories of disease are variable for most conditions that can severely impair ability to work, as in the examples of back pain, depression, cardiovascular disease, and TBI discussed in this chapter. Figure 7-2 illustrates the effects of a variety of factors on the trajectory of ability to work over time.
Both back pain and depression can improve, worsen, or stabilize, but their course can also be characterized by episodes of exacerbation followed by return to a previous plateau. Back pain, depression, and cardiac disorders can improve markedly with appropriate treatment even late after diagnosis. A different course is tracked for TBI, which, like stroke, often improves early to a plateau with little chance of further improvement, and associated limitations often fall into other specific categories, such as disorders of cognition, coordination, and mood.
For cardiac conditions, the severity of deviation from normal capacity can be measured more objectively than for the other conditions discussed in this chapter, as is also the case for pulmonary disorders. Even for cardiopulmonary disease, however, the associated physiologic decrements do not translate directly to work impairment, as multiple other factors affect the ability and the motivation to work with limitations. In addition, the
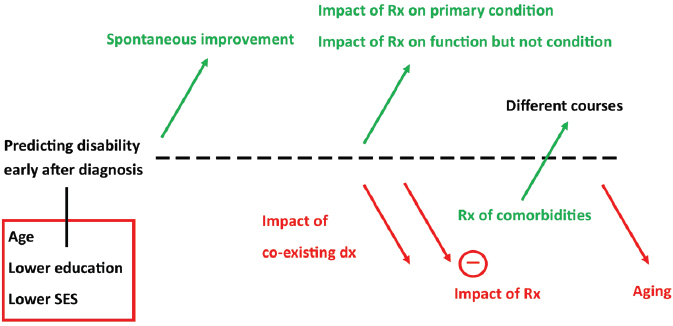
NOTES: Age, lower education, and lower socioeconomic status at the time of diagnosis predict lower likelihood of return to work. After diagnosis, increase in functional capacity can result from spontaneous improvement in the condition, from the impact of therapy on the condition itself, or from the impact of therapy on the ability to adapt to work despite continued presence of the condition. Coexisting diagnoses of comorbidities often limit improvement over time, but treatment of these comorbidities may improve overall function. Aging in general is associated with less improvement over time and greater burden of comorbidities limiting improvement in function. dx = diagnosis; Rx = treatment; SES = socioeconomic status.
multiple physical demands of a work environment are difficult to quantitate, the exception being strictly repetitive physical exertions, which often are not required by the dominant tasks of contemporary employment. Following initiation of therapies, heart failure can also be characterized by episodes of treatable exacerbation, but these usually herald a progressive downhill course. In contrast with the course of musculoskeletal limitations, depression, or most other impairments, the progression of cardiac disease to become the primary limitation to employment is often associated with a decreasing likelihood of surviving the next 2 years.
Impacts of Treatment
Therapies can treat a condition directly or aid adaptation to the work environment without changing the severity of the condition itself. For example, medications can often delay or prevent deterioration of cardiac and peripheral vascular function, while exercise training can improve functional capacity without improving the underlying organ system disease. Likewise, multiple medications can improve the ability to function with
major psychiatric diagnoses without necessarily altering the underlying pathology or trajectory of the condition. Diseases differ in the degree to which access to appropriate therapy determines the trajectory of functional capacity. Cardiovascular disease and depression are profoundly influenced by the availability of and compliance with optimal therapy. However, therapies for chronic back pain and for TBI have a limited effect on the condition’s trajectory. For conditions, such as cardiac disease and back pain, that may necessitate major surgical procedures, recovery of employability is often influenced by factors other than the primary diagnosis for which the surgery was performed. The efficacy of treatments for comorbidities can also influence the ability to work. In addition, it is important to consider the impact health disparities can have on access to treatment. Factors related to socioeconomic status, race/ethnicity, and language may limit access to treatment, symptom improvement, and thus return to work.
Aging and Comorbidities
Aging affects the physical capacity to repair, recover from, and adapt to most specific conditions that can affect employment. In addition, the age-related decline in reserve capacity for both physical and cognitive tasks decreases the margin for performance of many work-related tasks. Aging is strongly associated with increasing numbers and severity of comorbidities, including physical, mental, and cognitive limitations.
Depression
Depression is the most common comorbidity limiting employment for individuals with a wide range of physical and mental impairments, as well as rehabilitation from such events as surgeries and myocardial infarction. For all of the conditions considered in this chapter, co-occurring depression is frequent and associated with poor outcomes. Of major concern, moreover, it is underrecognized both as a primary diagnosis and as a powerful contributor to impairment from other diagnoses. The impact of treatment is clear for depression as the central diagnosis, but less is known about how to identify and address it as a complicating factor (Anderson et al., 2015; Scarrati et al., 2017; Sullivan et al., 1997).
Other Comorbidities
Although this chapter focuses on several common conditions in isolation, it is increasingly clear that these and other functional limitations often cannot be envisioned as a single impairment, but share common comorbidities that must be considered in evaluation. The landmark Medical
Outcomes Study of ambulatory office visits focused on combinations rather than single diagnoses of physical conditions, including angina, heart failure, myocardial infarction, back problems, gastrointestinal disorders, chronic lung problems, arthritis, and diabetes. Two or more conditions were present in the majority of patients with any one condition, and the number of conditions strongly influenced limitations on physical, role, and social functioning (Stewart et al., 1989). The declining trajectory of physical function in patients with these multiple conditions was documented over a 4-year longitudinal follow-up (Bayliss et al., 2004).
As the prevalence of people with multiple chronic conditions increases, health care delivery is facing new challenges in their evaluation and treatment. A major initiative for 2020 focuses on the need for health services research devoted specifically to “addressing the health care needs of Americans with [multiple chronic conditions] or disabilities” (Iezzoni, 2010, p. 1524).
FINDINGS AND CONCLUSIONS
Findings
7-1. The course of an impairment and where along the trajectory of a condition an individual becomes unable to work vary tremendously and are affected by symptom severity, comorbidities, demographic characteristics, and the job demands associated with the work itself.
7-2. Although the likelihood of return to work for a population is correlated with multiple factors that can be ascertained (as is the case for low back surgery and traumatic brain injury), return to work has not been reliably modeled or predicted with high accuracy.
7-3. Increasing age compounds the effects of the conditions discussed herein, both directly and in association with increasing burden of comorbidities.
7-4. Individuals with a primary cardiac limitation will often meet the U.S. Social Security Administration (SSA) cardiac listings for disability by the time they need to limit their work activities in occupations other than those requiring sustained heavy physical labor.
7-5. Rehabilitation has consistently been shown to improve exercise capacity and quality of life, with frequent benefits for improved mood, but has varying impacts on return to work.
7-6. Individuals with low back pain are more likely to return to work if they are of younger age, have fewer comorbid conditions, and perform less physically demanding work.
7-7. Depression frequently compounds work-related functional limitation in the context of other primary impairments.
7-8. Fewer than half of people with depression receive treatment, and even fewer receive evidence-based treatment.
Conclusions
7-1. Consideration of age and comorbidities is critically important in both the evaluation and the likely trajectory of common conditions and their effects on work-related impairment.
7-2. Depression is particularly important to consider and incorporate into evaluations regarding return to work with a chronic condition that limits physical function.
7-3. Age, presurgical employment, the physical exertion involved in work, and comorbidities are key factors in evaluating individuals’ functional abilities relevant to work requirements.
7-4. Evaluation of cardiac limitation to employment may focus primarily on determination of presence of SSA listing criteria for cardiac conditions.
7-5. Rehabilitation offers multiple benefits and is best offered early, but its impact on return to work varies widely.
7-6. Even with extensive assessment in multiple different domains, it is unlikely that multivariate models could be constructed to reliably answer the question of whether an individual will be able to return to work in the future.
7-7. It is advisable to address treatment of comorbidities during treatment of the primary impairment identified, in order to maximize functional capacity and ability to work.
7-8. Given the prevalence and underdetection of depression in general medical care and its impact on functioning, particularly when comorbid with other conditions, it is valuable to screen all disability applicants for depression. Such screening is standard practice in many primary care settings.
REFERENCES
Aboa-Éboulé, C., C. Brisson, E. Maunsell, B. Mâsse, R. Bourbonnais, M. Vézina, A. Milot, P. Theroux, and G. R. Dagenais. 2007. Job strain and risk of acute recurrent coronary heart disease events. JAMA 298(14):1652–1660.
Abraham, P., R. C. Rennert, J. R. Martin, J. Ciacci, W. Taylor, D. Resnick, E. Kasper, and C. C. Chen. 2016. The role of surgery for treatment of low back pain: Insights from the randomized controlled Spine Patient Outcomes Research Trials. Surgical Neurology International 7:38. http://surgicalneurologyint.com/surgicalint-articles/the-role-of-surgery-for-treatment-of-low-back-pain-insights-from-the-randomized-controlled-spine-patient-outcomes-research-trials (accessed April 4, 2019).
Adler, D. A., T. J. McLaughlin, W. H. Rogers, H. Chang, L. Lapitsky, and D. Lerner. 2006. Job performance deficits due to depression. American Journal of Psychiatry 163(9):1569–1576.
Aikens, J., K. Kroenke, D. E. Nease, M. S. Klinkman, and A. Sen. 2008. Trajectories of improvement for six depression-related outcomes. General Hospital Psychiatry 30(1):26–31. doi: 10.1016/j.genhosppsych.2007.10.003.
Anderson, J. T., A. R. Haas, R. Percy, S. T. Woods, U. M. Ahn, and N. U. Ahn. 2015. Clinical depression is a strong predictor of poor lumbar fusion outcomes among workers’ compensation subjects. Spine 40(10):748–756. doi: 10.1097/BRS.0000000000000863.
APA (American Psychiatric Association). 2013. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: APA.
Appelbaum, P. S. 2018. Functional assessment of depression and anxiety disorders relevant to work requirements. Presentation to the Committee on Functional Assessment for Adults with Disabilities, Washington, DC, April 19. http://nationalacademies.org/hmd/~/media/Files/Agendas/Activity%20Files/SelectPops/Functional%20Assessment/Meeting%203/Functional%20Assessment%20of%20Depression%20%20Anxiety%20%20Appelbaum.pdf (accessed April 4, 2019).
Banning, A. S., and A. Ng. 2012. Driving and arrhythmia: A review of scientific basis for international guidelines. European Heart Journal 34(3):236–244. doi.org/10.1093/eurheartj/ehs356.
BARI (Bypass Angioplasty Revascularization Investigation). 1997. Five-year clinical and functional outcome comparing bypass surgery and angioplasty in patients with multivessel coronary disease. Writing group for BARI investigators. JAMA 277(9):715–722.
BARI. 2007. Final 10-year follow-up results from the BARI randomized trial. Journal of the American College of Cardiology 49(15):1600–1606. doi: 10.1016/j.jacc.2006.11.048.
Bayliss, E. A., M. S. Bayliss, J. E. Ware, and J. F. Steiner. 2004. Predicting declines in physical function in persons with chronic medical conditions. Health and Quality of Life Outcomes 2:47–55. doi: 10.1186/1477-7525-2-47.
Beck, A., A. L. Crain, L. I. Solberg, J. Unützer, R. E. Glasgow, M. V. Maciosek, and R. Whitebird. 2011. Severity of depression and magnitude of productivity loss. Annals of Family Medicine 9(4):305–311. doi: 10.1370/afm.1260.
Benoist, M., P. Boulu, and G. Hayem. 2012. Epidural steroid injections in the management of low-back pain with radiculopathy: An update of their efficacy and safety. European Spine Journal 21(2):204–213.
Besen, E., A. E. Young, and W. S. Shaw. 2015. Returning to work following low back pain: Towards a model of individual psychosocial factors. Journal of Occupational Rehabilitation 25(1):25–37.
Bicket, M. C., J. M. Horowitz, H. T. Benzon, and S. P. Cohen. 2015. Epidural injections in prevention of surgery for spinal pain: Systematic review and meta-analysis of randomized controlled trials. Spine 15(2):348–362.
Broadhead, W. E., D. G. Blazer, L. K. George, and C. K. Tse. 1990. Depression, disability days, and days lost from work in a prospective epidemiologic survey. JAMA 264(19):2524–2528.
Buist-Bouwman, M. A., J. Ormel, R. de Graaf, and W. A. Vollebergh. 2004. Functioning after a major depressive episode: Complete or incomplete recovery? Journal of Affective Disorders 82(3):363–371. doi: 10.1016/j.jad.2004.02.007.
Buist-Bouwman, M. A., R. de Graaf, W. A. M. Vollebergh, and J. Ormel. 2005. Comorbidity of physical and mental disorders and the effect on work loss days. Acta Psychiatrica Scandinavica 111(6):436–443.
Bush, D. E., R. C. Ziegelstein, U. V. Patel, B. D. Thombs, D. E. Ford, J. A. Fauerbach, U. D. McCann, K. J. Stewart, K. K. Tsilidis, A. L. Patel, C. J. Feuerstein, and E. B. Bass. 2005. Post-myocardial infarction depression: Summary. AHRQ Evidence Report Summaries 123. https://www.ncbi.nlm.nih.gov/books/NBK11913 (accessed April 4, 2019).
Cahill, T. J., and R. K. Kharbanda. 2017. Heart failure after myocardial infarction in the era of primary percutaneous coronary intervention. World Journal of Cardiology 9(5):407–415. doi: 10.4330/wjc.v9.i5.407.
Campbell, P., N. E. Foster, E. Thomas, and K. M. Dunn. 2013. Prognostic indicators of low back pain in primary care: Five-year prospective study. The Journal of Pain 14(8):873–883. doi: 10.1016/j.jpain.2013.03.013.
Cancelliere, C., V. L. Kristman, J. D. Cassidy, C. A. Hincapié, P. Côté, E. Boyle, L. J. Carroll, B-M. Stålnacke, C. Nygren-de Boussard, and J. Borg. 2014. Systematic review of return to work after mild traumatic brain injury: Results of the International Collaboration on Mild Traumatic Brain Injury Prognosis. Archive of Physical Medicine and Rehabilitation 95(3 Suppl. 2):S201–S209.
Cassidy, J. D., P. Côté, L. J. Carroll, and V. Kristman. 2005. Incidence and course of low back pain episodes in the general population. Spine 30(24):2817–2823.
Cayea, D., S. Perera, and D. K. Weiner. 2006. Chronic low back pain in older adults: What physicians know, what they think they know, and what they should be taught. Journal of the American Geriatrics Society 54(11):1772–1777.
CDC (U.S. Centers for Disease Control and Prevention). 2007. Rates of hospitalization related to traumatic brain injury—Nine states, 2003. Morbidity and Mortality Weekly Report 56(8):167–170.
CDC. 2009. Prevalence and most common causes of disability among adults—United States, 2005. Morbidity and Mortality Weekly Report 58(16):421–426.
CDC. 2012. Mental health and chronic diseases. https://www.cdc.gov/workplacehealthpromotion/tools-resources/pdfs/issue-brief-no-2-mental-health-and-chronic-disease.pdf (accessed April 4, 2019).
Choi, H. J., S. Hahn, C. H. Kim, B. H. Jang, S. Park, S. M. Lee, J. Y. Park, C. K. Chung, and B. J. Park. 2013. Epidural steroid injection therapy for low back pain: A meta-analysis. International Journal of Technology Assessment in Health Care 29(3):244–253.
Choi, J. H., J. Y. Hong, S. W. Suh, J. H. Yang, S. Y. Park, J. H. Park, and S. J. Hong. 2016. What is the role of epidural steroid injections in lumbar spinal disease with moderate disability? Pain Physician 19(4):293–298.
Chou, R., and P. Shekelle. 2010. Will this patient develop persistent disabling low back pain? JAMA 303(13):1295–1302. doi: 10.1001/jama.2010.344.
Chou, R., R. Hashimoto, J. Friedly, R. Fu, T. Dana, S. Sullivan, C. Bougatsos, and J. Jarvik. 2015. Pain management injection therapies for low back pain. Rockville, MD: Agency for Healthcare Research and Quality.
Chou, R., R. Deyo, J. Friedly, A. Skelly, R. Hashimoto, M. Weimer, R. Fu, T. Dana, P. Kraegel, J. Griffin, S. Grusing, and E. D. Brodt. 2017. Nonpharmacologic therapies for low back pain: A systematic review for an American College of Physicians Clinical Practice Guideline. Annals of Internal Medicine 166(7):493–505.
Christiansen, M. N., L. Kober, P. Weeke, R. S. Ramachandran, J. L. Jeppesen, J. G. Smith, G. H. Gislason, C. Torp-Pederson, and C. Anderson. 2017. Age specific trends in incidence, mortality, and comorbidities of heart failure in Denmark 1995–2012. Circulation 137(25):1214–1223. doi: 10.1161/CIRCULATIONAHA.116.025941.
Chung, J. W., Y. Zeng, and T. K. Wong. 2013. Drug therapy for the treatment of chronic nonspecific low back pain: Systematic review and meta-analysis. Pain Physician 16(6):E685–E704.
Cleland, J. G. F., P. Pellicori, A. L. Clark, and M. C. Pettrie. 2017. Time to take the failure out of heart failure. Journal of the American College of Cardiology 5(7):538–540. doi: 10.1016/j.jchf.2017.04.003.
Deyo, R. A., M. Von Korff, and D. Duhrkoop. 2015. Opioids for low back pain. British Medical Journal 350:g6380. doi: 10.1136/bmj.g6380.
Drake, R. E., G. R. Bond, and D. R. Becker. 2012. Individual placement and support: An evidence-based approach to supported employment. New York: Oxford University Press.
Drake, R. E., W. Frey, G. R. Bond, H. H. Goldman, D. Salkever, A. Miller, T. A. Moore, J. Riley, M. Karakus, and R. Milfort. 2013. Assisting Social Security Disability Insurance beneficiaries with schizophrenia, bipolar disorder, or major depression in returning to work. American Journal of Psychiatry 170(12):1433–1441.
Elinson, L., P. Houck, S. C. Marcus, and H. A. Pincus. 2004. Depression and the ability to work. Psychiatric Services 55(1):29–34.
Ervasti, J., J. Vahtera, J. Pentti, T. Oksanen, K. Ahola, M. Kivimäki, and M. Virtanen. 2013. Depression-related work disability: Socioeconomic inequalities in onset, duration and recurrence. PLoS ONE 8(11):e79855. doi: 10.1371/journal.pone.0079855.
Feinberg, W. M., J. L. Blackshear, A. Laupacis, R. Kronmal, and R. G. Hart. 1995. Prevalence, age distribution, and gender of patients with atrial fibrillation. Archives of Internal Medicine 155(5):469–473.
Fihn, S. D., J. C. Blankenship, K. P. Alexander, J. A. Bittl, J. G. Byrne, B. J. Fletcher, G. C. Fonarow, R. A. Lange, G. N. Levine, T. M. Maddox, S. S. Naidu, E. M. Ohman, and P. K. Smith. 2014. 2014 ACC/AHA/AATS/PCNA/SCAI/STS focused update of the guideline for the diagnosis and management of patients with stable ischemic heart disease: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines, and the American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Journal of the American College of Cardiology 64(18):1929–1949. doi: 10.1016/j.jacc.2014.07.017.
Forman, D. E., M. S. Maurer, C. Boyd, R. Brindis, M. Salive, and M. Rich. 2018. Multimorbidity in older adults with cardiovascular disease. Journal of the American College of Cardiology 71(19):2149–2161. doi: 10.1016/j.jacc.2018.03.022.
Froelicher, V. F., C. K. Morris, A. Hideg, T. Kawaguchi, J. Myers, and K. Ueshima. 1992. Nomogram for exercise capacity using METs and age. American College of Cardiology Learning Highlights 8:2–5.
Froelicher, E. S., L. L. Kee, M. Newton, B. Lindskog, and M. Livingston. 1994. Return to work, sexual activity, and other activities after acute myocardial infarction. Heart and Lung 23(5):423–435.
Garrelfs, S. F., B. H. Donker-Cools, H. Wind, and M. H. Frings-Dresen. 2015. Return-to-work in patients with acquired brain injury and psychiatric disorders as a comorbidity: A systematic review. Brain Injury 29(5):550–557.
Gottleib, S. S., M. Khatta, E. Friedmann, L. Einbinder, S. Katzen, B. Baker, J. Marshal, S. Minshall, S. Robinson, M. L. Fisher, M. Potenza, B. Sigler, C. Baldwan, and S. A. Thomas. 2004. The influence of age, gender, and race on the prevalence of depression in heart failure patients. Journal of the American College of Cardiology 43(9):1542–1549.
Greenberg, P. E., A. A. Fournier, T. Sisitsky, C. T. Pike, and R. C. Kessler. 2015. The economic burden of adults with major depressive disorder in the United States (2005 and 2010). Journal of Clinical Psychiatry 76(2):155–162. doi: 10.4088/JCP.14m09298.
Gurcay, E., A. Bal, E. Eksioglu, A. E. Hasturk, A. G. Gurcay, and A. Cakci. 2009. Acute low back pain: Clinical course and prognostic factors. Disability and Rehabilitation 31(10):840–845.
Hackett, M. L., and K. Pickles. 2014. Part I: Frequency of depression after stroke: An updated systematic review and meta-analysis of observational studies. International Journal of Stroke 9(8):1017–1025. doi: 10.1111/ijs.12357.
Hammer-Helmich, L., J. M. Haro, B. Jönsson, M. A. Tanguy, S. Di Nicola, J. Chollet, D. Milea, B. Rive, and D. Saragoussi. 2018. Functional impairment in patients with major depressive disorder: The 2-year PERFORM study. Neuropsychiatric Disease and Treatment 14:239–249. doi: 10.2147/NDT.S146098.
Hannula, J. A., K. Lahtela, A. Järvikoski, J. Salminen, and P. Mäkelä. 2006. Occupational Functioning Scale (OFS)—An instrument for assessment of work ability in psychiatric disorders. Nordic Journal of Psychiatry 60(5):372–378. doi: 10.1080/08039480600937140.
Harvey, P. D., E. W. Twamley, A. E. Pinkham, C. A. Depp, and T. L. Patterson. 2017. Depression in schizophrenia: Associations with cognition, functional capacity, everyday functioning, and self-assessment. Schizophrenia Bulletin 43(3):575–582.
Hayden, J. A., K. M. Dunn, D. A. van der Windt, and W. S. Shaw. 2010. What is the prognosis of back pain? Best Practice & Research: Clinical Rheumatology 24(2):167–179. doi: 10.1016/j.berh.2009.12.005.
Hellerstein, D. J., G. Erickson, J. W. Stewart, P. J. McGrath, K. Hunnicutt-Ferguson, S. K. Reynolds, D. O’Shea, Y. Chen, A. Withers, and Y. Wang. 2014. Behavioral activation therapy for return to work in medication-responsive chronic depression with persistent psychosocial dysfunction. Comprehensive Psychiatry 57:140–147. doi: 10.1016/j. comppsych.2014.10.015.
Heymans, M. W., S. van Buuren, D. L. Knol, J. R. Anema, W. van Mechelen, and H. C. de Vet. 2010. The prognosis of chronic low back pain is determined by changes in pain and disability in the initial period. Spine 10(10):847–856. doi: 10.1016/j.spinee.2010.06.005.
Hirschfeld, R. M., D. L. Dunner, G. Keitner, D. N. Klein, L. M. Koran, S. G. Kornstein, J. C. Markowitz, I. Miller, C. B. Nemeroff, P. T. Ninan, A. J. Rush, A. F. Schatzberg, M. E. Thase, M. H. Trivedi, F. E. Borian, P. Crits-Christoph, and M. B. Keller. 2002. Does psychosocial functioning improve independent of depressive symptoms? A comparison of nefazodone, psychotherapy, and their combination. Biological Psychiatry 51(2):123–133. doi: 10.1016/S0006-3223(01)01291-4.
Houde, F., F. Cabana, and G. Léonard. 2016. Does age affect the relationship between pain and disability? A descriptive study in individuals suffering from chronic low back pain. Journal of Geriatric Physical Therapy 39(3):140–145. doi: 10.1519/JPT.0000000000000055.
Huysmans, E., L. Goudman, G. Van Belleghem, M. De Jaeger, M. Moens, J. Nijs, K. Ickmans, R. Buyl, C. Vanroelen, and K. Putman. 2018a. Return to work following surgery for lumbar radiculopathy: A systematic review. Spine 18(9):1694–1714. doi: 10.1016/j. spinee.2018.05.030.
Huysmans, E., K. Ickmans, D. Van Dyck, J. Nijs, Y. Gidron, N. Roussel, A. Polli, M. Moens, L. Goudman, and M. De Kooning. 2018b. Association between symptoms of central sensitization and cognitive behavioral factors in people with chronic nonspecific low back pain: A cross-sectional study. Journal of Manipulative and Physiological Therapeutics 41(2):92–101.
Iezzoni, L. I. 2010. Multiple chronic conditions and disabilities: Implications for health services research and data demands. Health Services Research 45(5):1523–1540.
IOM (Institute of Medicine). 2010. Cardiovascular disability. Washington, DC: The National Academies Press.
Jacobs, W. C., S. M. Rubinstein, B. Koes, M. W. van Tulder, and W. C. Peul. 2013. Evidence for surgery in degenerative lumbar spine disorders. Best Practice & Research: Clinical Rheumatology 27(5):673–684.
Jolliffe, J. A., K. Rees, R. S. Taylor, D. Thompson, N. Oldridge, and S. Ebrahim. 2001. Exercise-based rehabilitation for coronary heart disease. The Cochrane Database of Systematic Reviews (1):CD001800. doi: 10.1002/14651858.CD001800.
Judd, L. L., H. S. Akiskal, P. J. Zeller, M. Paulus, A. C. Leon, J. D. Maser, J. Endicott, W. Coryell, J. L. Kunovac, T. I. Mueller, J. P. Rice, and M. B. Keller. 2000. Psychosocial disability during the long-term course of unipolar major depressive disorder. Archives of General Psychiatry 57(4):375–380.
Kalogeropouolos, A. P., A. Samman-Tahhan, J. S. Hedley, A. A. McCue, J. B. Bjork, D. W. Markham, K. N. Bhatt, V. V. Georgiopoulou, A. L. Smith, and J. Butler. 2017. Progression to Stage D heart failure among outpatients with Stage C heart failure and reduced ejection fraction. Journal of the American College of Cardiology 5(7):528–537. doi: 10.1016/j.jchf.2017.02.020.
Kang, H.-J., S.-Y. Kim, K.-Y. Bae, S.-W. Kim, I.-S. Shin, J.-S. Yoon, and J.-M. Kim. 2015. Comorbidity of depression with physical disorders: Research and clinical implications. Chonnam Medical Journal 51(1):8–18.
Kato, M., L. W. Stevenson, M. Palardy, P. M. Campbell, C. W. May, N. K. Lakdawala, G. Stewart, A. Nohria, J. G. Rogers, J. T. Heywood, M. Gheorghiade, E. F. Lewis, X. Mi, and S. Setoguchi. 2012. The worst symptom as defined by patients during heart failure hospitalization: Implications for response to therapy. Journal of Cardiac Failure 18(7):524–533. doi: 10.1016/j.cardfail.2012.04.012.
Kessler, R. C., and R. G. Frank. 1997. The impact of psychiatric disorders on work loss days. Psychological Medicine 27(4):861–873.
Kessler, R. C., P. Berglund, O. Demler, R. Jin, D. Koretz, K. R. Merikangas, A. J. Rush, E. E. Walters, and P. S. Wang. 2003a. The epidemiology of major depressive disorder: Results from the National Comorbidity Survey Replication (NCS-R). JAMA 289(23):3095–3105.
Kessler, R. C., C. Barber, A. Beck, P. Berglund, P. D. Cleary, D. McKenas, N. Pronk, G. Simon, P. Stang, T. B. Üstün, and P. Wang. 2003b. The World Health Organization Health and Work Performance Questionnaire (HPQ). Journal of Occupational and Environmental Medicine 45(2):156–174.
Kessler, R. C., W. T. Chiu, O. Demler, K. R. Merikangas, and E. E. Walters. 2005. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Archives of General Psychiatry 62(6):617–627.
Kivimäki, M., and I. Kawachi. 2015. Work stress as a risk factor for cardiovascular disease. Current Cardiology Reports 17(9):630.
Krebs, E. E., A. Gravely, S. Nugent, A. C. Jensen, B. DeRonne, E. S. Goldsmith, K. Kroenke, M. J. Bair, and S. Noorbaloochi. 2018. Effect of opioid vs. nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: The SPACE randomized clinical trial. JAMA 319(9):872–882.
Kristman, V. L., P. Côté, S. Hogg-Johnson, J. D. Cassidy, D. Van Eerd, M. Vidmar M. Rezai, and R. A. Wennberg. 2010. The burden of work disability associated with mild traumatic brain injury in Ontario compensated workers: A prospective cohort study. The Open Occupational Health & Safety Journal 2:1–8. https://benthamopen.com/contents/pdf/TOOHSJ/TOOHSJ-2-1.pdf (accessed April 4, 2019).
Kroenke, K., R. L. Spitzer, and J. B. Williams. 2001. The PHQ-9: Validity of a brief depression severity measure. Journal of General Internal Medicine 16(9):606–613.
Lam, R. W. 2012. Which depressive symptoms and medication side effects are perceived by patients as interfering most with occupational functioning? Depression Research and Treatment 2012, Article 630206. doi: 10.1155/2012/630206.
Lam, R. W., E. E. Michalak, and L. N. Yatham. 2009. A new clinical rating scale for work absence and productivity: Validation in patients with major depressive disorder. BMC Psychiatry 9:78. doi: 10.1186/1471-244X-9-78.
Lee, S. S., Y. Choi, and G. S. Pransky. 2016. Extent and impact of opioid prescribing for acute occupational low back pain in the emergency department. Journal of Emergency Medicine 50(3):376–384. doi: 10.1016/j.jemermed.2015.10.015.
Lee, Y. P., S. D. Farhan, D. Kiester, C. Rosen, A. Pendi, and N. Bhatia. 2017. Variables affecting return to work after spinal surgery in a non-workers’ compensation population: A retrospective cohort study. Journal of the American Academy of Orthopaedic Surgeons 25(12):e282–e288. doi: 10.5435/JAAOS-D-17-00098.
Lerner, D., D. A. Adler, H. Chang, E. R. Berndt, J. T. Irish, L. Lapitsky, M. Y. Hood, J. Reed, and W. H. Rogers. 2004. The clinical and occupational correlates of work productivity loss among employed patients with depression. Journal of Occupational and Environmental Medicine 46(6 Suppl.):S46–S55.
Lerner, D., D. A. Adler, W. H. Rogers, H. Chang, L. Lapitsky, T. McLaughlin, and J. Reed. 2010. Work performance of employees with depression: The impact of work stressors. American Journal of Health Promotion 24(3):205–213. doi: 10.4278/ajhp.090313-QUAN-103.
Lerner, D., D. A. Adler, R. C. Hermann, W. H. Rogers, H. Chang, P. Thomas, A. Greenhill, and K. Perch. 2011. Depression and work performance: The work and health initiative study. In Work accommodation and retention in mental health, edited by I. Z. Schultz and E. S. Rogers. New York: Springer. Pp. 103–120. doi: 10.1007/978-1-4419-0428-7_6.
Lerner, D., D. A. Adler, R. C. Hermann, H. Chang, E. J. Ludman, A. Greenhill, K. A. Perch, W. C. McPeck, and W. H. Rogers. 2012. Impact of a work-focused intervention on the productivity and symptoms of employees with depression. Journal of Occupational and Environmental Medicine 54(2):128–135.
Lerner, D., D. A. Adler, W. H. Rogers, H. Chang, A. Greenhill, E. Cymerman, and F. Azocar. 2015. A randomized clinical trial of a telephone depression intervention to reduce employee presenteeism and absenteeism. Psychiatric Services 66(6):570–577.
Lerner, M. 2008. What does research tell us about depression, job performance, and work productivity? Journal of Occupational and Environmental Medicine 50(4):401–410.
Little, D. M., A. J. Cook, S. B. Morissette, and J. W. Klocek. 2015. Considerations for return to work following traumatic brain injury. Handbook of Clinical Neurology 131:465–479. doi: 10.1016/B978-0-444-62627-1.00027-5.
Losoi, H., N. D. Silverberg, M. Wäljas, S. Turunen, E. Rosti-Otajärvi, M. Helminen, T. M. Luoto, J. Julkunen, J. Öhman, and G. L. Iverson. 2016. Recovery from mild traumatic brain injury in previously healthy adults. Journal of Neurotrauma 33(8):766–776. doi: 10.1089/neu.2015.4070.
Mani, K., B. Cater, and A. Hudlikar. 2017. Cognition and return to work after mild/moderate traumatic brain injury: A systematic review. Work 58(1):51–62.
Marquez de la Plata, C., T. Hart, F. M. Hammond, A. Frol, A. Hudak, C. R. Harper, T. O’Neil-Pirozzi, J. Whyte, M. Carlile, and R. Diaz-Arrastia. 2008. Impact of age on long-term recovery from traumatic brain injury. Archives of Physical Medicine and Rehabilitation 89(5):896–903.
Mbakwem, A., F. Aina, and C. Amadi. 2016. Depression in patients with heart failure: Is enough being done? Cardiac Failure Review 2(2):110–112. doi: 10.15420/cfr.2016:21:1.
McGirt, M. J., A. Sivaganesan, A. L. Asher, and C. J. Devin. 2015. Prediction model for outcome after low-back surgery. Neurosurgical Focus 39(6):1–10. doi: 10.3171/2015.8.FOCUS15338.
McGurk, S. R., R. E. Drake, H. Xie, J. Riley, R. Milfort, T. W. Hale, and W. Frey. 2018. Cognitive predictors of work among Social Security Disability Insurance beneficiaries with psychiatric disorders enrolled in IPS supported employment. Schizophrenia Bulletin 44(1):32–37.
McKnight, P. E., and T. B. Kashdan. 2009. The importance of functional impairment to mental health outcomes: A case for reassessing our goals in depression treatment research. Clinical Psychology Review 29(3):243–259.
Mehling, W. E., V. Gopisetty, E. Bartmess, M. Acree, A. Pressman, H. Goldberg, F. M. Hecht, T. Carey, and A. L. Avins. 2012. The prognosis of acute low back pain in primary care in the United States: A 2-year prospective cohort study. Spine 37(8):678–684. doi: 10.1097/BRS.0b013e318230ab20.
Menezes Costa, L. C., C. G. Maher, M. J. Hancock, J. H. McAuley, R. D. Herbert, and L. O. P. Costa. 2012. The prognosis of acute and persistent low-back pain: A meta-analysis. CMAJ: Canadian Medical Association Journal 184(11):E613–E624. doi: 10.1503/cmaj.111271.
Milanovic, M., K. Holshausen, R. Milev, and C. R. Bowie. 2018. Functional competence in major depressive disorder: Objective performance and subjective perceptions. Journal of Affective Disorders 234:1–7. doi: 10.1016/j.jad.2018.02.094.
Mital, A., A. Desai, and A. Mital. 2004. Return to work after a coronary event. Journal of Cardiopulmonary Rehabilitation and Prevention 24(6):365–373.
Moradi, B., S. Hagmann, A. Zahlten-Hinguranage, F. Caldeira, C. Putz, N. Rosshirt, E. Schönit, A. Mesrian, M. Schiltenwolf, and E. Neubauer. 2012. Efficacy of multidisciplinary treatment for patients with chronic low back pain: A prospective clinical study in 395 patients. Journal of Clinical Rheumatology 18(2):76–82.
Morone, G., T. Paolucci, M. R. Alcuri, M. C. Vulpiani, A. Matano, I. Bureca, S. Paolucci, and V. M. Saraceni. 2011. Quality of life improved by multidisciplinary back school program in patıents with chronic non-specific low back pain: A single blind randomized controlled trial. European Journal of Physical and Rehabilitation Medicine 47(4):533–541.
Mosbach, P., S. Campanelli, A. D’Anello, L. Klaus, E. Aberger, J. Pelletier, P. Quinn, V. Sundar, A. Houtenville, B. Ogut, H. Brotsos, and O. Urdapilleta. 2018. Mental cognitive demands. Contract No. 28321318A00040029/28321318FA0011262. Summit Consulting.
Papakostas, G. I. 2009. Major depressive disorder: Psychosocial impairment and key considerations in functional improvement. American Journal of Managed Care 15(11 Suppl.):S316–S321.
Parker, S. L., S. S. Godil, S. K. Mendenhall, S. L. Zuckerman, D. N. Shau, and M. J. McGirt. 2014. Two-year comprehensive medical management of degenerative lumbar spine disease (lumbar spondylolisthesis, stenosis, or disc herniation): A value analysis of cost, pain, disability, and quality of life: Clinical article. Journal of Neurosurgery: Spine 21(2):143–149.
Patrick, N., E. Emanski, and M. A. Knaub. 2014. Acute and chronic low back pain. Medical Clinics of North America 98(4):777–789.
Pengel, L. H. M., R. D. Herbert, C. G. Maher, and K. M. Refshauge. 2003. Acute low back pain: Systematic review of its prognosis. British Medical Journal 327(7410):323. doi: 10.1136/bmj.327.7410.323.
Qaseem, A., T. J. Wilt, R. M. McLean, and M. A. Forciea. 2017. Noninvasive treatments for acute, subacute, and chronic low back pain: A clinical practice guideline from the American College of Physicians. Annals of Internal Medicine 166(7):514–530.
Rao, V., M. Bertrand, P. Rosenberg, M. Makley, D. J. Schretlen, J. Brandt, and M. M. Mielke. 2010. Predictors of new-onset depression after mild traumatic brain injury. The Journal of Neuropsychiatry and Clinical Neurosciences 22(1):100–104.
Rørth, R., C. Wong, K. Kragholm, E. L. Fosbøl, U. M. Mogensen, M. Lamberts, M. C. Petrie, P. S. Jhund, T. A. Gerds, C. Torp-Pedersen, G. H. Gislason, J. J. McMurray, L. Køber, and S. L. Kristensen. 2016. Return to the workforce after first hospitalization for heart failure. Circulation 134(14):999–1009.
Rost, K., J. L. Smith, and M. Dickinson. 2004. The effect of improving primary care depression management on employee absenteeism and productivity: A randomized trial. Medical Care 42(12):1202–1210.
Rost, K., J. Fortney, and J. Coyne. 2005. The relationship of depression treatment quality indicators to employee absenteeism. Mental Health Services Research 7(3):161–169.
Rubenstein, L. V., N. R. Rayburn, E. B. Keeler, D. E. Ford, K. M. Rost, and C. D. Sherbourne. 2007. Predicting outcomes of primary care patients with major depression: Development of a depression prognosis index. Psychiatric Services 58(8):1049–1056. doi: 10.1176/appi.ps.58.8.1049.
Rytsälä, H. J., T. K. Melartin, U. S. Leskelä, T. P. Sokero, P. S. Lestelä-Mielonen, and E. T. Isometsä. 2005. Functional and work disability in major depressive disorder. The Journal of Nervous and Mental Disease 193(3):189–195.
Rytsälä, H. J., T. K. Melartin, U. S. Leskelä, T. P. Sokero, P. S. Lestelä-Mielonen, and E. T. Isometsä. 2007. Predictors of long-term work disability in major depressive disorder: A prospective study. Acta Psychiatrica Scandinavica 115(3):206–213.
Samkange-Zeeb, F., T. Altenhöner, G. Berg, and T. Schott. 2006. Predicting non-return to work in patients attending cardiac rehabilitation. International Journal of Rehabilitation Research 29(1):43–49.
Sanderson, K., E. Tilse, J. Nicholson, B. Oldenburg, and N. Graves. 2007. Which presenteeism measures are more sensitive to depression and anxiety? Journal of Affective Disorders 101(1–3):65–74.
Scaratti, C., M. Leonardi, D. Sattin, S. Schiavolin, M. Willems, and A. Raggi. 2017. Work related difficulties in patients with traumatic brain injury: A systematic review on predictors and associated factors. Disability and Rehabilitation 39(9):847–855. doi: 10.3109/09638288.2016.1162854.
Schoenbaum, M., J. Unützer, D. McCaffrey, N. Duan, C. Sherbourne, and K. B Wells. 2002. The effects of primary care depression treatment on patients’ clinical status and employment. Health Services Research 37(5):1145–1158. doi: 10.1111/1475-6773.01086.
Schonstein, E., D. T. Kenny, J. L. Keating, and B. W. Koes. 2003. Work conditioning, work hardening and functional restoration for workers with back and neck pain. Cochrane Database of Systematic Reviews 3:CD001822. doi: 10.1002/14651858.CD001822.
Schultz, I. Z., J. Crook, K. Fraser, and P. W. Joy. 2000. Models of diagnosis and rehabilitation in musculoskeletal pain-related occupational disability. Journal of Occupational Rehabilitation 10(4):271–293.
Schultz, I. Z., A. W. Stowell, M. Feuerstein, and R. J. Gatchel. 2007. Models of return to work for musculoskeletal disorders. Journal of Occupational Rehabilitation 17(2):327–352.
Shames, J., I. Treger, H. Ring, and S. Giaquinto. 2007. Return to work following TBI: Trends and challenges. Disability and Rehabilitation 29(17):1387–1395.
Shanfield, S. B. 1990. Return to work after an acute myocardial infarction: A review. Heart and Lung 19(2):109–117.
Shaw, W. S., C. C. Nelson, M. J. Woiszwillo, B. Gaines, and S. E. Peters. 2018. Early return to work has benefits for relief of back pain and functional recovery after controlling for multiple confounds. Journal of Occupational and Environmental Medicine 60(10):901–910.
Shilyansky, C., L. M. Williams, A. Gyurak, A. Harris, T. Usherwood, and A. Etkin. 2016. Effect of antidepressant treatment on cognitive impairments associated with depression: A randomised longitudinal study. The Lancet Psychiatry 3(5):425–435.
Shipton, E. A. 2018. Physical therapy approaches in the treatment of low back pain. Pain and Therapy 7(2):127–137.
Shmagel, A., R. Foley, and H. Ibrahim. 2016. Epidemiology of chronic low back pain in U.S. adults: Data from the 2009–2010 National Health and Nutrition Examination Survey. Arthritis Care Research (Hoboken) 68(11):1688–1694.
Sivarajan, E. S., and K. M. Newton. 1984. Exercise, education, and counseling for patients with coronary artery disease. Clinics in Sports Medicine 3(2):349–369.
Smith, J. A., R. L. Fuino, I. Pesis-Katz, X. Cai, B. Powers, M. Frazer, and J. D. Markman. 2017. Differences in opioid prescribing in low back pain patients with and without depression: A cross-sectional study of a national sample from the United States. PAIN Reports 2(4):e606.
Sorvaniemi, M., H. Helenius, and R. K. Salokangas. 2003. Factors associated with being granted a pension among psychiatric outpatients with major depression. Journal of Affective Disorders 75(1):43–48.
Steenstra, I. A., C. Munhall, E. Irvin, N. Oranye, S. Passmore, D. Van Eerd, Q. Mahood, and S. Hogg-Johnson. 2017. Systematic review of prognostic factors for return to work in workers with sub acute and chronic low back pain. Journal of Occupational Rehabilitation 27(3):369–381. doi: 10.1007/s10926-016-9666-x.
Steffens, D., M. J. Hancock, C. G. Maher, J. Latimer, R. Satchell, M. Ferreira, P. H. Ferreira, M. Partington, and A. L. Bouvier. 2014. Prognosis of chronic low back pain in patients presenting to a private community-based group exercise program. European Spine Journal 23(1):113–119. doi: 10.1007/s00586-013-2846-x.
Stegenga, B. T., M. H. Kamphuis, M. King, I. Nazareth, and M. I. Geerlings. 2012. The natural course and outcome of major depressive disorder in primary care: The PREDICT-NL study. Social Psychiatry and Psychiatric Epidemiology 47(1):87–95. doi:10.1007/s00127-010-0317-9.
Stein, K. F., and A. Miclescu. 2017. Effectiveness of multidisciplinary rehabilitation treatment for patients with chronic pain in a primary health care unit. Scandinavian Journal of Pain 4(4):190–197.
Stevenson, L. W. 2017. Who would be branded with failure? Circulation 136(15):1359–1361. doi: 10.1161/CIRCULATIONAHA.117.029667.
Stewart, A., S. Greenfield, R. D. Hays, K. Wells, W. H. Rogers, S. D. Berry, E. A. McGlynn, and J. E. Ware. 1989. Functional status and well-being of patients with chronic conditions. JAMA 262(7):907–913.
Stewart, W. F., J. A. Ricci, E. Chee, S. R. Hahn, and D. Morganstein. 2003. Cost of lost productive work time among U.S. workers with depression. JAMA 289(23):3135–3144.
Sullivan, M. D., A. Z. Larois, C. Caum, L. C. Grothaus, and W. J. Katon. 1997. Functional status in coronary artery disease: 1-year prospective study of the role of anxiety and depression. American Journal of Medicine 103(5):348–356.
Trivedi, M. H., D. W. Morris, S. R. Wisniewski, I. Lesser, A. A. Nierenberg, E. Daly, B. T. Kurian, B. N. Gaynes, G. K. Balasubramani, and A. J. Rush. 2013. Increase in work productivity of depressed individuals with improvement in depressive symptom severity. American Journal of Psychiatry 170(6):633–641. doi: 10.1176/appi.ajp.2012.12020250.
Truszczy´ nska, A., K. Rapała, O. Truszczy´ nski, A. Tarnowski, and S. Łukawski. 2013. Return to work after spinal stenosis surgery and the patient’s quality of life. International Journal of Occupational Medicine and Environmental Health 26(3):394–400. doi: 10.2478/s13382-013-0105-3.
Turner, J. A., G. Franklin, D. Fulton-Kehoe, L. Sheppard, B. Stover, R. Wu, J. V. Gluck, and T. M. Wickizer. 2008. Early predictors of chronic work disability: A prospective, population-based study of workers with back injuries. Spine 33(25):2809–2818.
Udelson, J. E., and L. W. Stevenson. 2016. Future of heart failure diagnosis, therapy, and management. Circulation 133(25):2671–2686.
U.S. Burden of Disease Collaborators, A. H. Mokdad, K. Ballestros, M. Echko, S. Glenn, H. E. Olsen, E. Mullany, A. Lee, A. R. Khan, A. Ahmadi, A. J. Ferrari, A. Kasaeian, A. Werdecker, A. Carter, B. Zipkin, B. Sartorius, B. Serdar, B. L. Sykes, C. Troeger, C. Fitzmaurice, C. D. Rehm, D. Santomauro, D. Kim, D. Colombara, D. C. Schwebel, D. Tsoi, D. Kolte, E. Nsoesie, E. Nichols, E. Oren, F. J. Charlson, G. C. Patton, G. A. Roth, H. D. Hosgood, H. A. Whiteford, H. Kyu, H. E. Erskine, H. Huang, I. Martopullo, J. A. Singh, J. B. Nachega, J. R. Sanabria, K. Abbas, K. Ong, K. Tabb, K. J. Krohn, L. Cornaby, L. Degenhardt, M. Moses, M. Farvid, M. Griswold, M. Criqui, M. Bell, M. Nguyen, M. Wallin, M. Mirarefin, M. Qorbani, M. Younis, N. Fullman, P. Liu, P. Briant, P. Gona, R. Havmoller, R. Leung, R. Kimokoti, S. Bazargan-Hejazi, S. I. Hay, S. Yadgir, S. Biryukov, S. E. Vollset, T. Alam, T. Frank, T. Farid, T. Miller, T. Vos, T. Bärnighausen, T. T. Gebrehiwot, Y. Yano, Z. Al-Aly, A. Mehari, A. Handal, A. Kandel, B. Anderson, B. Biroscak, D. Mozaffarian, E. R. Dorsey, E. L. Ding, E. K. Park, G. Wagner, G. Hu, H. Chen, J. E. Sunshine, J. Khubchandani, J. Leasher, J. Leung, J. Salomon, J. Unutzer, L. Cahill, L. Cooper, M. Horino, M. Brauer, N. Breitborde, P. Hotez, R. Topor-Madry, S. Soneji, S. Stranges, S. James, S. Amrock, S. Jayaraman, T. Patel, T. Akinyemiju, V. Skirbekk, Y. Kinfu, Z. Bhutta, J. B. Jonas, and C. J. L. Murray. 2018. The state of U.S. health, 1990–2016: Burden of diseases, injuries, and risk factors among U.S. states. JAMA 319(14):1444–1472.
Verkerk, K., P. A. Luijsterburg, M. W. Heymans, I. Ronchetti, A. L. Pool-Goudzwaard, H. S. Miedema, and B. W. Koes. 2013. Prognosis and course of disability in patients with chronic nonspecific low back pain: A 5- and 12-month follow-up cohort study. Physical Therapy 93(12):1603–1614.
Weiner, D. K., S. Sakamoto, S. Perera, and P. Breuer. 2006. Chronic low back pain in older adults: Prevalence, reliability, and validity of physical examination findings. Journal of the American Geriatrics Society 54(1):11–20.
Wells, K. B., A. Stewart, R. D. Hays, M. A. Burnam, W. Rogers, M. Daniels, S. Berry, S. Greenfield, and J. Ware. 1989. The functioning and well-being of depressed patients. Results from the Medical Outcomes Study. JAMA 262(7):914–919.
WHO (World Health Organization). 2008. The global burden of disease: 2004 update. Geneva, Switzerland: WHO Press.
Williams, M. A., P. A. Ades, L. F. Hamm, S. J. Keteyian, T. P. LaFontaine, J. L. Roitman, and R. W. Squires. 2006. Clinical evidence for a health benefit from cardiac rehabilitation: An update. American Heart Journal 152(5):835–841.
Yancy, C. W., M. Jessup, B. Bozkurt, J. Butler, D. E. Casey, Jr., M. H. Drazner, G. C. Fonarow, S. A. Geraci, T. Horwich, J. L. Januzzi, M. R. Johnson, E. K. Kasper, W. C. Levy, F. A. Masoudi, P. E. McBride, J. J. V. McMurray, J. E. Mitchell, P. N. Peterson, B. Riegel, F. Sam, L. W. Stevenson, W. H. W. Tang, E. J. Tsai, and B. L. Wilkoff. 2013. 2013 ACCF/AHA Guideline for the management of heart failure. Journal of the American College of Cardiology 62(16):e150–e239.
Young, K., C. Scott, R. Rodeheffer, and H. Chen. 2017. Preclinical heart failure: Progression of Stage A and Stage B heart failure in a community population. European Heart Journal 38(Suppl. 1):ehx504.P3382. doi: 10.1093/eurheartj/ehx504.P3382.








































