4
Mental Health Disorders
Mental health disorders affect a person’s thinking, feeling, mood, or behavior and include conditions such as depression, anxiety, bipolar disorder, and schizophrenia (CDC, 2018). The Diagnostic and Statistical Manual for Mental Disorders, Fifth Edition (DSM-5) defines mental disorder as
a syndrome characterized by clinically significant disturbance in an individual’s cognition, emotion regulation, or behavior that reflects a dysfunction in the psychologic, biological, or developmental processes underlying mental functioning. Mental disorders are usually associated with significant distress or disability in social, occupational, or other important activities. (APA, 2013, p. 20)
The committee selected mental health disorders for inclusion in the report in accordance with the stipulations in the Statement of Task—that is, that the committee consider conditions that last 12 months or longer and can be disabling for a period of time but that may not result in permanent disability. The decision on which specific mental health disorders to include was based on the prevalence of the disorders in the United States and on their potential responsiveness to treatment. The committee selected the mental disorders listed in Table 4-1, recognizing that others might also meet its criteria.
The chapter begins with a description of the prevalence of mental disorders in the United States, followed by a discussion of cross-cutting issues that affect individuals with the selected mental health disorders. The
TABLE 4-1 Selected Mental Health Conditions
| SSA Listing | DSM-5 Diagnosis | DSM-5 Classification |
|---|---|---|
| Depressive, bipolar, and related disorders | Major depressive disorder | Depressive disorders |
| Bipolar I disorder | Bipolar and related disorders | |
| Bipolar II disorder | ||
| Anxiety and obsessive compulsive disorders | Panic disorder | Anxiety disorders |
| Generalized anxiety disorder | ||
| Social anxiety disorder (social phobia) | ||
| Obsessive compulsive disorder | Obsessive compulsive and related disorders | |
| Trauma and stress-related disorders | Posttraumatic stress disorder | Trauma and stress-related disorders |
NOTE: DSM-5 = Diagnostic and Statistical Manual for Mental Disorders, Fifth Edition.
remainder of the chapter consists of discussions of the issues noted in the Statement of Task and ends with a summary and conclusions.
The committee collected information from a variety of sources. The DSM-5 (APA, 2013) provides the professionally accepted diagnostic criteria for mental health disorders used by health care professionals in the United States and was the source for the diagnostic criteria described in this chapter. Pertinent modifications to those criteria that occurred over time are noted where relevant. Selected clinical practice guidelines from professional organizations were used to identify evidence-based treatments for each disorder. The guidelines used were selected based on their comprehensiveness and relevance to the questions, their transparency and clarity about literature search strategies and approaches to evidence-based decisions, recently updated information, and whether the guideline had had an external peer review. See Tables A-1 through A-5 in the Appendix for details on the guidelines used for each disorder. Other sources of scientific information include recent systematic reviews, meta-analyses, and notable publications.
It is estimated that in 2018, 19.1 percent of U.S. adults experienced mental illness (47.6 million people), and 4.6 percent experienced serious mental illness (11.4 million people) (SAMHSA, 2018). For the specific disorders of interest, the best data available suggest that the 12-month prevalence for anxiety disorders is 18.1 percent among U.S. adults (estimated 23 million people) (Kessler et al., 2005a); for major depressive episodes, 7.2 percent (17.7 million people) (SAMHSA, 2018); for posttraumatic stress disorder (PTSD), 3.5 percent (7 million people) (Kessler et al., 2005a); for any bipolar disorder, 2.8 percent (estimated 6 million people) (Merikangas et al., 2007); and for obsessive compulsive disorder (OCD), 1.2 percent (estimated 2.5 million people) (Ruscio et al., 2010).
CROSS-CUTTING ISSUES FOR SELECTED MENTAL HEALTH DISORDERS
Cross-cutting issues include the types of medical professionals involved in mental health treatment, responses to treatment in the context of disability, and the complexity of measuring the improvement from treatment in terms of functional outcomes, as required by the Social Security Adminstration (SSA) in determining entitlement. Pain is also a cross-cutting feature in the mental health conditions discussed. Chapter 2 includes a discussion of approaches to pain and pain treatment in general.
How People Are Identified for Treatment and the Types of Medical Professionals Involved in Care
People are identified for mental health treatment through a number of different mechanisms. Some people self-refer because they or their family members or friends become concerned about certain behaviors; some people are identified through screenings in a given health care setting; and others are referred through medical professionals during an encounter for another condition. Less commonly, people may be identified as needing mental health care because of a risk to self or others. Individuals are then directed to a specific treatment depending on the treatment guidelines, their treatment history, patient preference, and treatment availability, among other factors.
Non-mental health specialists may be involved in the treatment of people with mental health disorders. However, for those individuals with severe, persistent, and treatment-resistant disease, which might be disabling, the expectation is that they would be receiving psychiatric services from a variety of mental health professionals, including a prescriber (e.g., psychiatrist, advanced practice nurse), psychologist, or licensed clinician with a social work, counseling, or rehabilitation degree. For some populations (e.g., those in rural areas or small towns), however, care from qualified mental health professionals (e.g., specialized in evidence psychotherapy) might not be available.
Response to Treatments for Mental Health Disorders in the Context of Disability
Most of the mental disorders under consideration can occasionally result in SSA-defined disability. Individuals meeting the criteria for disability are likely to have a severe form of the disorder or a significant comorbidity that affects response to treatment and the potential for remission. Thus, the applicability of usual treatment algorithms is uncertain. It is possible
that individuals who are determined to be disabled by SSA may require second- and third-line treatment or even be non-responders to treatment. Additionally, most of the mental health disorders being considered can be chronic and relapsing. Although an individual could respond to treatment in the acute phase and remain well (with ongoing treatment or even in the absence of treatment) for a period of time, episodes of illness can recur, sometimes with greater severity and reduced response to treatment. Finally, the presence of comorbidities may limit treatment choices because of the presence of or risk for side effects.
Measuring Improvement of Functional Outcomes
The SSA definition of disability requires that a person have limitations in function that prevents that person from engaging in substantial gainful activity (SGA). Under SSA’s Medical Improvement Review Standard, benefits may not be stopped unless there is medical improvement related to the ability to perform SGA, as determined in the disability decision that entitled the person to benefits. Determining how impairments affect occupational function has been a longstanding challenge. Because of its importance within the SSA review standard, SSA asked the committee to describe time to improvement in functioning to the point where the condition is no longer disabling.
In response to that issue, the committee acknowledges important challenges, which are common to the mental health disorders discussed in the chapter. First, the treatments in research and in clinical practice guidelines for mental health disorders target symptoms and do not clearly focus on occupational function as an outcome. Second, psychiatric disorders are often recurrent and, therefore, the time until improvement is not adequately captured as a linear process. Third, psychiatric disorders are often comorbid with other psychiatric disorders, pain, and other medical conditions, and thus the time to improvement will depend on those and other factors. Any estimates of time to improvement need to consider the fact that clinical trials generally exclude participants with comorbidities. Fourth, symptom improvement and functional improvement may represent separate aspects of recovery, and one cannot assume that improvement in symptoms results in improvements in functioning. For the mental health conditions described in this chapter, the discontinuity between symptoms and functioning as well as the multiple, inter-related domains that measure function are not well understood. The 2019 National Academies report Functional Assessment for Adults with Disabilities found that when assessing the functional abilities of individuals with mental health disorders, the following domains are important: general cognitive/intellectual ability, language and communication, learning and
memory, attention and vigilance, processing speed, executive functioning, adaptability, and work-related personal interactions. The report concluded that because of the fluctuating nature of the disease course, it is important to understand the relationship between mental illness and functioning and to perform frequent assessments of disability in applicants with mental health disorders. The report also concluded that there is no single measure that captures all important aspects of the mental abilities needed for work. Only some measures include occupational functioning as one of the domains to measure improvement for psychiatric disorders. Some of the measures developed for assessing the occupational domain, which are described further in the 2019 National Academies report and in the 2016 Canadian Network for Mood and Anxiety Treatments (CANMAT) guidelines, are the Multidimensional Scale of Independent Functioning, the World Health Organization (WHO) Disability Assessment Schedule 2.0, the Social and Occupational Functioning Assessment Scale, the Work Disability Functional Battery Mental Health Measures, the University of California, San Diego, Performance-Based Skills Assessment, and the Occupational Functioning Scale.
Bearing in mind those challenges, the committee now discusses the limited research base related to treatment and functional improvement within each mental health disorder section.
Mental Health Conditions and Pain
It is well known that mental conditions and chronic pain often occur together, but the causal pathway or direction of the association is still debated. Chronic pain may contribute to mental conditions, and, vice versa, mental conditions may result in an increased risk of chronic pain (Velly and Mohit, 2018). According to data from the World Mental Health Survey Initiative, individuals with mental conditions comorbid with chronic pain conditions (back or neck pain) were more likely to be among the most disabled, as measured by the WHO Disability Assessment Schedule (WHODAS 2.0), than those with neither mental disorder nor chronic pain (Scott et al., 2009). As reported by Gureje et al. (1998) and Velly and Mohit (2018), individuals with chronic pain are four times more likely to have anxiety or depression than those without chronic pain (Gureje et al., 1998).
Regardless of the causal pathway, mental conditions with comorbid chronic pain result in more work-loss days than do the individual conditions by themselves. The effect is additive except for chronic back pain, where the interaction with mental conditions has been found to be synergistic with respect to work loss (Buist-Bouwman, 2005; NASEM, 2019). Additionally, a systematic review found no evidence of a difference in return-to-work rates between patients with chronic back pain and mental comorbidities
(depression, bipolar disorder, panic disorder [PD]) and chronic back pain patients without mental comorbidities (Baumeister et al., 2012). The same review, however, reported more work absence and more work-related disability in depressed chronic back pain patients than in patients without depression.
The types of chronic pain that commonly co-occur with mental conditions include migraine headaches (Fishbain et al., 2017; Goldstein, 2009; Katzman et al., 2014), neck and back pain (Fishbain et al., 2017; Goldstein, 2009; Katzman et al., 2014; Kroenke et al., 2013; Velly and Mohit, 2018), fibromyalgia (Fishbain et al., 2017; Kroenke et al., 2013; Velly and Mohit, 2018), rheumatoid arthritis (Goldstein, 2009; Katzman et al., 2014; Velly and Mohit, 2018), and abdominal pain (Velly and Mohit, 2018). The treatment of comorbid chronic pain and mental conditions varies by the type of chronic pain and the specific mental condition. The treatment for both conditions should be managed by a team of specialists and monitored cautiously to avoid adverse outcomes or addictions (see Chapters 2 and 5 for further discussion of treatments for chronic pain management).
MAJOR DEPRESSIVE DISORDER
The adult depressive disorders listed in the DSM-5 have common features, such as sad, empty, or irritable mood, and they include major depressive disorder (MDD) (including major depressive episode), persistent depressive disorder (dysthymia), premenstrual dysphoric disorder, substance/medication-induced depressive disorder, depressive disorder due to another medical condition, other specified depressive disorder, and unspecified depressive disorder. The disorders differ from each other in their duration, timing, and presumed etiology.
This section focuses on MDD. It is both the most prevalent mental health disorder in the United States and a major cause of disability, and it exhibits many of the common features noted above. Major depression affects multiple domains, which results in important economic burdens for individuals and countries (Salomon et al., 2015). Its adverse effects can be observed in physical health problems and include heart disease, arthritis, asthma, back pain, chronic pulmonary disease hypertension, and migraine. In addition, as with other mental disorders, MDD is associated with negative functional outcomes such as social and occupational dysfunction (Deschenes et al., 2015) and reduced quality of life (Burgel et al., 2013; Faller et al., 2015; Schowalter et al., 2013). WHO reported that depression accounts for greater than 5 percent of population illness-related productivity loss. MDD is associated with 65.5 excess lost days per worker with depression and with $31 billion to $51.5 billion in annual workplace costs. In the areas of social and work functioning, the impairments range from the
mild to complete incapacity, and three factors are associated with higher work disability: the severity of the illness, concurrent medical conditions, and anxiety as a comorbidity (Kessler et al., 2005a).
Professionally Accepted Diagnostic Criteria for Major Depressive Disorder
The DSM-5 describes MDD as involving discrete episodes of at least 2 weeks’ duration involving clear-cut changes in affect, cognition, and neurovegetative functions and inter-episode remissions (i.e., when the symptom severity is within the normal, nondepressed range). In the most common presentation of major depression, a single episode is typically longer than 2 weeks, and recurrent episodes are frequent. According to the DSM-5 there is no specific age of onset, but puberty seems to be a critical period, and the incidence is highest at approximately age 20. Table 4-2 includes criteria for the diagnosis of major depressive episode. A more chronic form of depression, persistent depressive disorder (defined as dysthymia and chronic major depression in the Diagnostic and Statistical Manual for Mental Disorders, 4th Edition [DSM-IV]), is diagnosed when the mood disturbance continues for at least 2 years in adults.
Developmental Course, Gender Distribution, and Comorbidities
MDD presents in various ways, and it is recommended that a diagnosis be accompanied by “specifiers” that can guide the selection of an approach to management and monitoring tools. These specifiers might include the presence of anxiety, melancholic features, atypical features, psychotic features, catatonia, peripartum onset, and seasonal pattern. Often the severity, which is an important aspect of a disability determination, is also included in the diagnosis as a specifier (see Table 4-2).
Although each person will experience major depression differently, Figure 4-1 is widely recognized as a representation of the typical course of MDD. Because of its episodic nature, a patient’s remission status needs to be included as a specifier when diagnosing, managing, and monitoring the disease. For example, a patient in remission might consider an adjuvant treatment to minimize the risk of relapse. This course, where an acute episode is followed by a maintenance phase and a continuation phase, corresponds chronologically with the treatment phases that have been proposed, and it has implications for how to identify improvements in functioning.
The DSM-5 states that although the 12-month prevalence of MDD in the United States is about 7 percent, females experience it at a much higher rate, specifically 1.5–3 times more frequently than in males. MDD often occurs with other medical or mental health comorbidities. In one study
TABLE 4-2 DSM-5 Criteria for Major Depressive Disorder and Severity Specification
| Criterion/Symptom Description | |
|---|---|
|
|
| Note: This exclusion does not apply if all of the manic-like or hypomanic-like episodes are substance-induced or are attributable to the physiological effects of another medical condition. | |
| Severity Specification | |
| Mild | Few, if any, symptoms in excess of those required to make the diagnosis are present; the intensity of the symptoms is distressing but manageable; and the symptoms result in minor impairment in social or occupational functioning. |
| Moderate | The number of symptoms, intensity of symptoms, and/or functional impairment are between those specified for “mild” and “severe.” |
| Severe | The number of symptoms is substantially in excess of that required to make the diagnosis, the intensity of the symptoms is seriously distressing and unmanageable, and the symptoms markedly interfere with social and occupational functioning. |
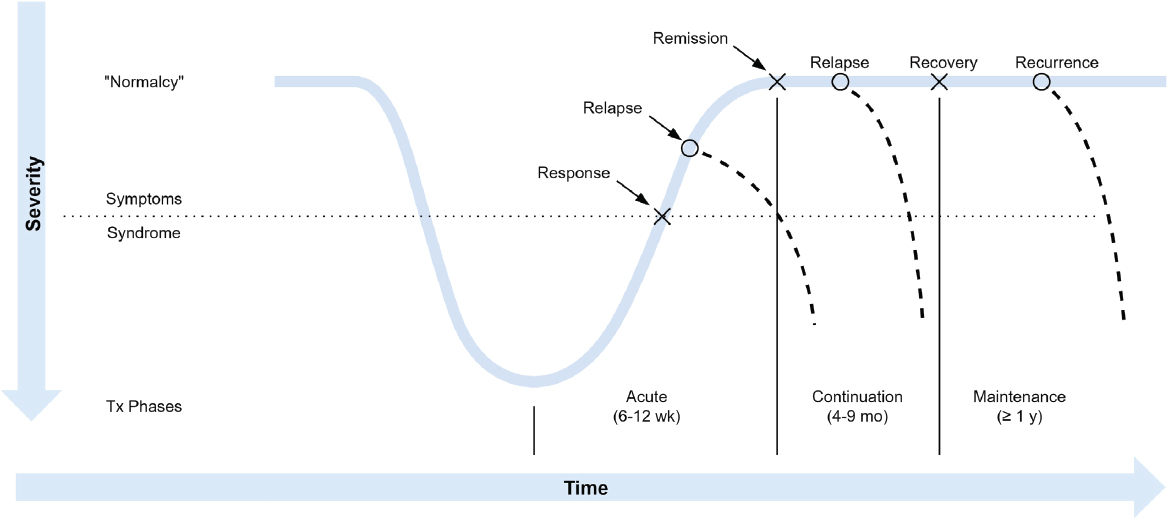
NOTES: Response represents a clinically significant reduction in symptom severity relative to baseline status (usually 50 percent). Remission is when symptom severity is within the normal, nondepressed range. Relapse is the reemergence of symptoms of major depression following some level of remission, but preceding recovery. Recovery is a prolonged period of remission that marks the end of the index episode (e.g., 6–12 months). Recurrence is the onset of a new episode of depression following recovery.
SOURCE: Kupfer, 1991.
almost 75 percent of individuals with lifetime MDD met the criteria for another psychiatric disorder (Kessler et al., 2003), especially an anxiety disorder (59 percent). Moreover, 31.9 percent of the persons with MDD had at least one other International Statistical Classification of Diseases and Related Health Problems (ICD) diagnosis, and 24 percent had at least one substance use disorder (SUD). With regard to medical comorbidities, depression can be either a primary condition or due to another medical condition. Some medical conditions with high rates of comorbid depression include cancer, cardiovascular disease, multiple sclerosis, traumatic brain injury, HIV, epilepsy, migraines, Parkinson’s disease, hepatitis C, and chronic pain.
Standard Measures of Outcomes for Major Depressive Disorder
The field still lacks reliable and valid biomarkers for depression, recovery, and prognosis. The most recent systematic review of predictors of depression treatment outcomes concluded that despite a vast amount of literature on the topic, there are still no reliable or valid biomarkers or other predictors that can be recommended for use in the clinic (Perlman et al., 2019).
This section describes two sets of patient-reported outcome measures, symptom measures and functional measures. Measures of depression symptom severity are the most widely used types of outcome assessment.
The Hamilton Depression Rating Scale (HDRS, also known as HAM-D) is one of the oldest and most widely used depression symptom severity measures, though it has declined in use in recent years (Bagby et al., 2004). The original HDRS is a multidimensional 21-item clinician-rated measure with variable item scaling that covers symptom severity over the past week (Hamilton, 1967). However, items do not correspond to DSM criteria, limiting the validity of the measure (Furukawa, 2010). Although the original HAM-D has only adequate internal and inter-rater reliability (Furukawa, 2010), it has been the benchmark to which other measures are compared in term of sensitivity to change, clinically significant response to treatment, and remission (Reeves et al., 2012). The Montgomery Asberg Depression Rating Scale (MADRS) is a clinician-rated measure designed to be sensitive to antidepressant-versus-placebo effects rather than following DSM criteria (Furukawa, 2010). It has excellent inter-rater reliability, good concurrent validity compared with the HDRS, and sensitivity to change similar to the HDRS (Furukawa, 2010). The Quick Inventory of Depressive Symptomology is a measure that covers DSM-IV and DSM-5 criteria. It has good internal consistency, high correlations with the HDRS, and a sensitivity to change similar to the HDRS. The Beck Depression Inventory II (BDI-II) contains most DSM MDD criteria. It has good internal consistency and good test–retest reliability. The sensitivity to change of the BDI-II is
comparable to that of MADRS (Furukawa, 2010). The Patient Health Questionnaire-9 (PHQ-9) parallels DSM-IV and DSM-5 MDD criteria. It has excellent internal consistency and test–retest reliability (Furukawa, 2010). In contrast with most other measures, it is a valid screener for MDD with a pooled sensitivity of 0.80 and a pooled specificity of 0.92, generally using a cutoff of 10 or higher (Gilbody et al., 2007). The PHQ-9 is at least as sensitive to change as other measures (Furukawa, 2010).
Historically, response to treatment (typically defined as at least a 50 percent reduction in symptom severity compared with baseline) has been the most widely used standard for judging symptomatic outcomes by clinicians and researchers. However, remission (the virtual absence of depressive symptoms as defined by the measures described above) has been adopted as the target of treatment because patients who achieve it have better functioning, better prognosis, and a more stable state (Frank et al., 1991; Paykel et al., 1995) and are less likely to relapse into another MDD episode (Rush et al., 2006a).
Although reduced depression symptom severity can be associated with improved functioning, there is not always improvement even with adequate depression treatment (McKnight and Kashdan, 2009; Sheehan et al., 2017; Trivedi, 2013). Clinicians have recommended that measures of daily functioning be included in depression treatment trials (Rush et al., 2006a). The topic of measuring functional outcomes is complicated by the fact that major depressive episodes can have both acute and chronic effects on cognition. A recent review by Summit Consulting, commissioned by SSA (Mosbach et al., 2018), found that depression can significantly affect attention, concentration, learning, and memory as well as executive functioning (Lee et al., 2012; Rocca et al., 2015; Rock et al., 2014; Snyder et al., 2013). There is evidence that impaired cognition can begin with the first depressive episode (Lee et al., 2012) and that cognitive deficits continue even after remission (Bhalla et al., 2006). A high-enough degree of residual cognitive impairments can result in work disability.
Treatments for Major Depressive Disorder
Depression treatment can be divided into two phases. The goal of the acute phase, which typically lasts 8–12 weeks, is to achieve symptom remission. The goals of the maintenance phase, which can last 6–24 months or longer, are to restore full functioning and prevent relapse. Detailed treatment algorithms are available to help guide clinical decision making. An example of such an algorithm for managing depression is presented in Figure A-1 of the Appendix.
Table 4-3 lists the first- and second-line psychologic and pharmacologic treatments both for acute episodes and for maintenance, based on the
TABLE 4-3 Treatments for Major Depressive Disorder
| Treatment | Psychotherapy | Pharmacotherapy |
|---|---|---|
| First-line |
Cognitive behavioral therapy (CBT) Interpersonal therapy (IPT) (for acute) Behavioral activation (for acute) Mindfulness-based cognitive therapy (for maintenance) |
Agomelatine, bupropion, citalopram, escitalopram, fluoxetine, fluvoxamine, mianserina, mirtazapine, paroxetine, sertraline, vortioxetine |
| Second-line |
IPT (for maintenance) Behavioral activation (for maintenance) Mindfulness-based cognitive therapy (for acute) Cognitive behavioral analysis system of psychotherapy Problem-solving therapy (acute) Short-term psychodynamic psychotherapy (acute) Telephone-delivered CBT and IPT (acute) Internet- and computer-assisted therapy (acute) |
Amitriptyline, clomipramine, levomilnacipran, moclobemide, quetiapine, selegiline, transdermala, trazodone, vilazodone |
SOURCES: CANMAT, 2016; VA/DoD, 2016.
CANMAT1 guidelines and the U.S. Department of Veterans Affairs/U.S. Department of Defense (VA/DoD) guidelines.2 Complementary and alternative medicine treatments are not included in the table because of the more limited clinical evidence regarding their effectiveness.
The overall effectiveness of antidepressants and of evidence-based psychotherapies are equivalent (Cuijpers et al., 2014a). An initial treatment with antidepressant medication results in remission in 30–50 percent of cases (Rush et al., 2006b; Thase et al., 2005, 2010). Treatment may take as long as 10–12 weeks to be effective (Rush et al., 2006b). The overall remission rate associated with psychotherapies is 43 percent versus 27 percent for all control groups (Cuijpers et al., 2014a). Psychotherapy, especially cognitive behavioral therapy (CBT) and interpersonal therapy, results in persistent benefit after the treatment is over, whereas with antidepressant medications the benefits of treatment are often lost once the drug is stopped (APA, 2010; Parikh et al., 2009). It is important to note that combined
___________________
1 The Canadian Network for Mood and Anxiety Treatments (CANMAT) is a network of academic and clinical experts dedicated to improving clinical care for people with mood and anxiety disorders.
2 The 2016 U.S. Department of Veterans Affairs and the U.S. Department of Defense Clinical Practice Guideline for the Management of Major Depressive Disorder provide evidence-based recommendations that have been peer reviewed. The committee assumes those guidelines are applicable to the civilian population.
antidepressant medication and psychotherapy is more effective than either treatment alone (Cuijpers et al., 2009a,b). CANMAT (2016) or VA/DoD (2016) guidelines recommend a combination of antidepressant medication and a psychologic intervention for moderate to severe major depression (Cuijpers et al., 2009a,b).
Studies have shown that combining medical treatments with psychotherapy is effective for treatment-resistance depression. However, less than 50 percent of patients respond to first-line antidepressant treatment or psychotherapy. A meta-analysis and meta-regression analysis conducted to investigate the effectiveness of psychotherapy for treatment-resistant depression found that adding psychotherapy (CBT, interpersonal therapy, mindfulness-based cognitive therapy, and cognitive behavioral analysis) to treatment as usual had a moderate effect on treatment-resistance depression (van Bronswijk et al., 2019). A 2018 meta-analysis reported that there was moderate-quality evidence that adding psychotherapy to usual care significantly increased remission from treatment-resistant depression (Ijaz et al., 2018). Finally, a meta-analysis of treatment augmentation showed that adding mood stabilizers, antipsychotics, and N-methyl-D-aspartate (NMDA)targeting drugs to current treatment significantly reduced symptom severity (Strawbridge et al., 2019). Augmentation, however, is not always acceptable to the patient, given the potential for added interactions and side effects.
Alternative medications represent another option for treating treatment-resistant depression. The VA/DoD guideline (VA/DoD, 2016) on treating depression recommends monoamine oxidase inhibitors (MAOIs) (e.g., isocarboxazid, phenelzine, and tranylcypromine) and tricyclic antidepressants (e.g., amitriptyline, imipramine, desipramine, nortriptyline) for people who have failed first-line treatments. However, MAOIs require dietary restrictions on tyramine. Both MAOIs and tricyclics have worse adverse-effect profiles than first-line antidepressant medications. Lithium and triiodothyronine can be used to augment selective serotonin reuptake inhibitors (SSRIs) or serotonin-norepinephrine reuptake inhibitors (SNRIs). The VA/DoD guideline also recommends neurostimulation for treatment-resistant depression. Electroconvulsive therapy (ECT) is an effective treatment for persons with severe MDD or treatment-resistance depression. Indications for ECT include catatonia, psychosis, and severe suicidality as well as poor response to multiple medical treatments. Repetitive transcranial magnetic stimulation is also potentially effective for treatment-resistant depression.
Finally, a recent systematic review and network meta-analysis3 of the comparative efficacy and tolerability of pharmacologic and somatic
___________________
3 Network meta-analyses use all available data from randomized clinical trials to estimate the effect of each intervention relative to other interventions, even those that have not been compared directly.
interventions for treatment-resistant depression found that ketamine demonstrated superior efficacy for treatment-resistant depression when compared with other pharmacologic and somatic treatments at 2 weeks after treatment began (Papadimitropoulou et al., 2017). Data comparing ketamine to other treatments beyond 2 weeks are limited. At 4, 6, and 8 weeks, quetiapine augmentation and risperidone augmentation were found to be the first and second best treatments, respectively.
Because symptomatic recovery from depressive illness does not guarantee recovery of function, including occupational functioning, investigators have increasingly advocated for the use of combined treatments that target both depressive symptoms and functional impairments, including work disability (Adler et al., 2015; Trivedi, 2018). That combined treatment approach parallels what has been used to reduce work disability in other populations, such as those suffering from chronic pain (Costa-Black et al., 2010). Furthermore, there is mixed evidence concerning whether adding work-focused interventions (e.g., assisting the depressed person with return to work) to a standard depression treatment improves work-related outcomes (Adler et al., 2015; Hellstrom et al., 2017; Lerner et al., 2012; Nieuwenhuijsen et al., 2014). However, the science of using cognitive rehabilitation strategies to treat cognitive dysfunction in persons with major depression is still being explored, and findings are inconclusive (Porter et al., 2014).
More pertinent to this report is the limited treatment literature on fostering return to employment in persons who are already on Social Security Disability Insurance (SSDI) due to mental illness. Drake and colleagues (2013) randomized 2,059 SSDI recipients with depression, schizophrenia, or bipolar disorder to a multifaceted intervention consisting of individual placement and support, medication management, other behavioral health services, health insurance coverage with no copay, and the suspension of disability reviews compared with usual services. The treatment group achieved greater paid employment (60.3 versus 40.2 percent) as well as improved mental health and quality of life relative to the controls. However, according to the authors, only 14 percent of eligible beneficiaries joined the study (perhaps because of fears about potential loss of benefits). Moreover, among those who returned to work, less than 3 percent earned at or above the threshold for SGA established by SSA.
Length of Time to Improvement for Major Depressive Disorder
Estimating time to symptom improvement is complicated by numerous factors, including under-treatment, the need for multiple sequential treatment episodes to achieve remission of symptoms, treatment-resistance
depression, the presence of comorbidities, and the chronic relapsing course of major depression. Additionally, the resolution of functional impairments likely lags behind the remission of depressive symptoms. For example, depression-related cognitive impairment might persist despite depression remission or recovery. Residual functional and cognitive impairments might require supplementary cognitive- or work-focused treatments to optimize improvement.
The American College of Neuropsychopharmacology recommends that clinical trials last 12–20 weeks to optimize the detection of remission (Rush et al., 2006b). However, the STAR*D4 study, a trial that includes patients with medical and psychiatric comorbidities, reported less optimistic results. The trial, which included a sample of 4,000 adults receiving various successive treatments for their current depression, found that only 36.8 percent of the patients remitted within the first 12- to 14-week treatment phase and that even the overall cumulative remission rate, which included all four acute treatment steps, was 67 percent over 48–56 weeks. In the overall group of remitters, 33–50 percent relapsed within the 1-year study followup period. The trial also found that even in cases when the second, third, or fourth step resulted in remission, the outcome might be temporary and that remission was less likely for those with a comorbid anxiety disorder.
On the other hand, among SSA beneficiaries, improvement in employment rate began within 6 months and peaked at 19 months post-treatment initiation (Drake et al., 2013). However, almost no one achieved the threshold for substantial gainful employment; functional improvement is thought to occur more slowly than symptom improvement (McKnight and Kashdan, 2009; Mintz et al., 1992). Thus, the expected times for improvement mentioned above are likely underestimates for the purpose of predicting functional improvement. In addition, the course of major depression is not linear and residual symptoms during a temporary period of remission may lead to relapse (Conradi et al., 2011; Judd et al., 2000; Nierenberg et al., 2010). Given the evidence about predicting final response to treatment (Steidmann et al., 2013; Wagner et al., 2017), assessing early response (after 4–8 weeks) during the course of treatment could be helpful in predicting the likelihood of improvement.
Finally, there appears to be the potential for depression improvement among adults of any age. A recent systematic review of predictors of antidepressant efficacy reported that antidepressants are effective across a broad age range and that any age effects were inconsistent and depended on the type of treatment (Perlman et al., 2019). Systematic and other reviews of psychosocial treatments for depression in older adults indicate they are effective (Huang et al., 2015; Renn and Arean, 2017).
BIPOLAR DISORDERS
Bipolar disorders are mood disorders that manifest as episodes of mania, hypomania, and major depression (APA, 2013). They include bipolar I disorder, bipolar II disorder, cyclothymic disorder, substance/medication-induced bipolar and related disorder, bipolar and related disorder due to another medical condition, other specified bipolar and related disorder, and unspecified bipolar and related disorder. This section includes discussions of bipolar I and bipolar II disorders.
The diagnostic guidelines indicate that as many as 30 percent of individuals diagnosed with bipolar I disorder have been estimated to exhibit occupational impairments. The DSM-5 indicates that bipolar II disorder can be as severe and disabling as bipolar I disorder because those individuals with bipolar II disorder experience more depression, sometimes co-occurring with hypomanic symptoms (APA, 2013). In addition, although bipolar I disorder has more severe symptoms, individuals with bipolar II disorder experience episodes with more frequency and with higher rates of comorbidities and recurrent suicidal behaviors (Vieta and Suppes, 2008). Furthermore, their executive functioning can be as impaired or more so (Dickinson et al., 2017) compared with those with bipolar I disorder. Even the functional consequences of individuals with cyclothymia can be significant as a result of the mood disturbances. Patients with bipolar disorder have the potential to be impaired in all functional domains (e.g., social, occupational, and general function) (Baune and Malhi, 2015; Dickinson et al., 2017; Gitlin and Miklowitz, 2016; Raucher-Chene et al., 2017; Szmulewicz et al., 2017), although the factors that cause impairment have not been clearly specified.
Professionally Accepted Diagnostic Criteria for Bipolar Disorders
Bipolar disorder can be experienced in various forms and is typically accompanied by serious impairments in work and social functioning. Individuals with bipolar I disorder may experience manic episodes with inflated self-esteem and a decreased need to sleep, for example. The majority of those individuals also experience major depressive episodes during the course of their lives. Individuals diagnosed with bipolar II disorder present with at least one major episode of depression and one of hypomania, but not manic episodes.
According to the DSM-5 there is no specific age of onset for bipolar I disorder and bipolar II disorder, although the average age of onset is earlier for bipolar I disorder—specifically, around 18 years of age for bipolar I and the mid-20s for bipolar II disorder (APA, 2013). Women with bipolar II appear to be more likely than those with bipolar I disorder to experience hypomania with mixed depressive features and a rapid-cycling course.
Table 4-4 lists criteria for the diagnosis of bipolar I disorder and bipolar II disorder as described in DSM-5.
Developmental Course, Gender Distribution, and Comorbidities
Recent studies confirm that bipolar disorder is a recurrent disorder, with greater than 90 percent of individuals having recurrent mood episodes. There is a great variability in the presentation, sequence, and length of episodes in bipolar disorders, but patterns are repeated for a given patient. For example, some patients tend to experience a manic episode followed by a depressive episode, whereas the reverse pattern is typical for other patients. A manic episode with psychotic features is more likely to be followed by more episodes with psychotic features (APA, 2013).
In patients undergoing usual care for bipolar I disorder, the median length of mood episodes (manic or depressed) is 13 weeks, and 75 percent recover within 1 year (Solomon et al., 2010). Although depressive episodes vary, they typically last longer than manic episodes, as depicted in Figure 4-2. For example, one study found that patients spent longer periods in depression (on average three times longer) than in the mania/hypomania states (Baldessarini et al., 2010). Despite the episodic nature of the disease, which implies periods with no symptoms and normal functioning, studies have shown that bipolar patients experience symptoms the majority of the time (Judd et al., 2002, 2003). People with rapid cycling bipolar disorder I or II will experience a minimum of four episodes of mania/hypomania or depression each year (Carvalho et al., 2014).
The National Comorbidity Survey-Replication (NCS-R) study, a nationally representative survey of mental disorders in adults in the United States, found that the 12-month prevalence of bipolar I disorder was 0.6 percent, with no difference by gender. For bipolar II disorder, the 12-month prevalence was 0.8 percent in the United States, with some indication of higher rates in females (Merikangas et al., 2007). There are also gender-related differences in the presentation of bipolar disorders. Females experience the depressive symptoms more frequently than males as well as rapid cycling and mixed states as specifiers. Lifetime eating disorders and a higher risk of alcohol use disorder are often comorbid with bipolar disorder (APA, 2013).
Several factors can modify the course of the disorder. For those with psychosis or severe psychosocial impairment at an episode’s onset, the probability of recovery is diminished (Solomon et al., 2010). Those with more total years spent in manic or depressed episodes are less likely to recover. Each year spent in a major or minor depression episode reduces the likelihood of recovery from subsequent major depressive episodes by 7 percent (Solomon et al., 2013). As the number of mood disordered episodes increase, there is greater risk of recurrence, longer duration of
TABLE 4-4 DSM-5 Criteria Bipolar I Disorder and Bipolar II Disorder
| Diagnosis | Criterion/Symptom Description | |||
|---|---|---|---|---|
| Bipolar I disorder |
For a diagnosis of bipolar I disorder it is necessary to meet the following criteria for a manic episode. The manic episode may have been preceded by and may be followed by hypomanic or major depressive episodes.
Note: A full manic episode that emerges during antidepressant treatment (e.g., medication, electroconvulsive therapy) but persists at a fully syndromal level beyond the physiological effect of that treatment is sufficient evidence for a manic episode and, therefore, a bipolar I diagnosis. |
|||
| Diagnosis | Criterion/Symptom Description | |||
|---|---|---|---|---|
| Bipolar II disorder |
For a diagnosis of bipolar II disorder it is necessary to meet the following criteria for a current or past hypomanic episode and the following criteria for a current or past major depressive episode: Hypomanic Episode
Note: A full hypomanic episode that emerges during antidepressant treatment (e.g., medication, electroconvulsive therapy) but persists at a fully syndromal level beyond the physiologic effect of that treatment is sufficient evidence for a hypomanic episode diagnosis. However, caution is indicated so that one or two symptoms (particularly increased irritability, edginess, or agitation following antidepressant use) are not taken as sufficient for diagnosis of a hypomanic episode, nor necessarily indicative of a bipolar diathesis. |
|||
| Major Depressive Episode (see Table 4-2 under Depression section) Specifiers to Help with Diagnosis and Treatment (CANMAT, 2018). |
||||
| Specifier | Manic Episode | Depressive Episode | Illness Course | |
| Anxious distress | X | X | ||
| Mixed features | X | X | ||
| Rapid cycling | X | |||
| Melancholic features | X | |||
| Atypical features | X | |||
| Psychotic features | X | X | ||
| Catatonia | X | X | ||
| Peripartum onset | X | X | ||
| Seasonal pattern | X | |||
| Remission | X | X | ||
| Current episode severity | X | X | ||
episodes, increased symptom severity during episodes, decreasing threshold for developing recurrent episode, and increased likelihood of dementia (Kessing and Andersen, 2017).
Bipolar disorder often co-occurs with comorbidities that complicate the course of the disorder. The existence of comorbidities is an important consideration when making decisions about a treatment plan. The most frequent comorbid psychiatric disorders are anxiety disorders, OCD, attention deficit hyperactivity disorder (ADHD), SUD, eating disorders, and personality disorders. Common medical comorbid disorders include metabolic syndrome and migraine headache.
Standard Measures of Outcomes for Bipolar Disorder
A recent systematic review examined the clinical utility of patient-reported and clinician-rated measures of mania and depression for the management of bipolar disorder (Cerimele et al., 2019). The authors found that the Altman Self-Rating Mania Scale4 had high clinical utility in that it
___________________
4 The Altman Self-Rating Mania Scale is a short, five-item self-assessment questionnaire that can be used in assessing the presence and severity of manic or hypomanic symptoms. This scale is compatible with other diagnostic scales and the DSM-IV diagnostic criteria.
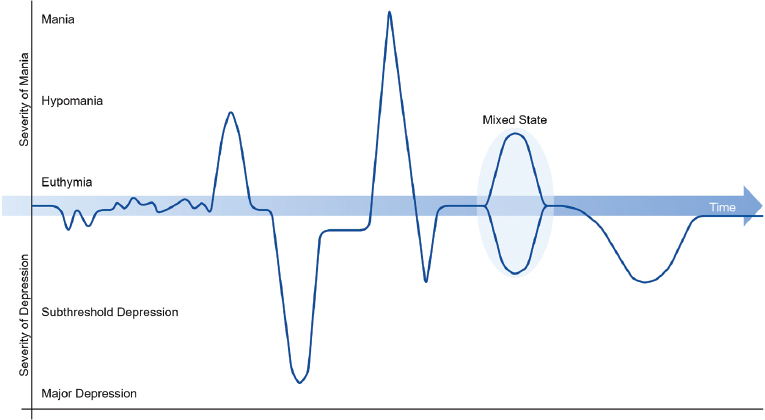
NOTE: As explained in the text, patients with bipolar I disorder experience mania while those with bipolar II disorders experience hypomanic and depression episodes, and patients with cyclothymia do not experience mania or major depression episodes.
SOURCE: Grande et al., 2016.
is brief, easy to score, and has good reliability and validity. No clinician-rated measure of mania had clinical utility that was as good or better. With regard to depression measures, the researchers concluded that the self-report and clinician-observed version of the Quick Inventory of Depressive Symptomatology both had high clinical utility scores. The five-item version of the HDRS, a clinician-rated measure, also had high clinical utility scores. The Internal State Scale and the Affective Self-Rating Scale had moderately good clinical utility scores for patient-reported measures. Two measures that clinicians use to rate both mania and depression, the Bipolar Inventory of Symptoms and the Life Chart Methodology—Clinician version, had moderately high clinical utility scores.
In addition to symptom measures, measures of daily functioning and cognition are also pertinent to people with bipolar disorder. Correspondingly, a few tools have been developed and used to measure the functional outcomes of bipolar disorder. Some of the most widely used tools were developed for the general measurement of function (e.g., the Global Assessment of Functioning, Functioning Assessment Short Test, and WHODAS 2.0, which is directly linked to the International Classification of Functioning, Disability and Health). Those tools have been
adapted and validated with core sets for bipolar disorder. In the domain of cognitive functioning, the International Society for Bipolar Disorders’ Targeting Cognition Task Force recommends the Battery for Assessment of Neurocognition composite (Yatham et al., 2010); an alternative measure that has been validated in bipolar disorder is the Measurement and Treatment Research to Improve Cognition in Schizophrenia Consensus Cognitive Battery (Burdick et al., 2011).
Treatments for Bipolar Disorders I and II
The first step in deciding on a treatment for bipolar disorder is to determine whether the patient is experiencing mania, hypomania, or depression as the therapeutic plan will vary depending on the state of the individual. Other factors that will require assessment are the presence of comorbidities, previous treatments, and the patient’s response to and adverse effects of prior treatments. As with other mental disorders, the patient’s preferences for treatment need to be taken into consideration.
As with major depression and other mental disorders, guidelines recognize the need to treat both the acute and maintenance phases (Bauer and Gitlin, 2016). Treating the acute phase, which typically lasts 6–12 weeks, involves establishing the correct diagnosis; initiating treatment; monitoring efficacy, safety, and tolerability; and achieving symptom remission as well as functional improvements. If remission is achieved, the patient enters the maintenance phase, which lasts 6–24 months or longer. During that period the focus is on returning to baseline functioning and quality of life, treating comorbidities, and preventing relapse or recurrence. Though most studies have been short term and have been conducted in individuals experiencing acute episodes, the same therapies are typically recommended for both the acute and the maintenance phases. An example of an algorithm that clinicians use to guide decisions, depending on the patients’ status (i.e., whether the patient is in a mania/hypomania, depressive, or mixed episode) can be found in Figure A-2 in the Appendix.
Pharmacotherapy recommendations for bipolar I–related depression are summarized in Table 4-5. The CANMAT guideline states that the general principles for assessing depression in patients with bipolar I disorder also apply to patients with bipolar II disorder. The evidence-based treatments for mania are exclusively pharmacologic and not psychologic. For clarity and efficiency, only first- and second-line treatments for mania are listed in Table 4-6.
Bipolar disorders cause a wide range of functional impairments that can be lasting and merit treatment in an effort to improve overall outcomes. Persistent functional impairment is predicted by the number of manic/hypomanic episodes and is even more strongly correlated with subsyndromal
TABLE 4-5 Pharmacologic Treatments for Acute Bipolar Depression: Level of Evidencea and Line of Treatmentb
| Level of Evidence and Line of Treatment | |||||
|---|---|---|---|---|---|
| Acute | Maintenance | Acute | |||
| Treatment | Acute Depression | Prevention of Any Mood Episode | Prevention of Depression | Prevention of Mania | Acute Mania |
| First-line treatments | |||||
| Quetiapine | Level 1 | Level 1 | Level 1 | Level 1 | Level 1 |
| Lurasidone + Li/DVP | Level 1 | Level 3 | Level 3 | Level 4 | |
| Lithium | Level 2 | Level 1 | Level 1 | Level 1 | Level 1 |
| Lamotrigine | Level 2 | Level 1 | Level 1 | Level 2 | Negative evidence |
| Lurasidone | Level 2 | Level 4 | Level 4 | Level 4 | nd |
| Lamotrigine | Level 2 | Level 4 | Level 4 | Level 4 | Negative evidence |
| Second-line treatments | |||||
| Divalproex | Level 2 | Level 1 | Level 2 | Level 3 | Level 1 |
| SSRIs/bupropion | Level 1 | nd | Level 4 | nd | |
| ECT | Level 4 | Level 4 | Level 4 | Level 4 | Level 3 |
| Cariprazine | Level 1 | nd | nd | nd | Level 1 |
| Olanzapine-fluoxetine | Level 2 | nd | nd | nd | nd |
NOTE: ECT = electroconvulsive therapy; Li/DVP = lithium/divalproex; nd = no data; SSRI = select serotonin reuptake inhibitor.
a Levels 1 and 2 evidence refer specifically to treatment studies in which randomized comparisons are available. Recommendations involving epidemiological or risk factors primarily arise from observational studies, and hence the highest level of evidence is usually Level 3.
b First line: Level 1 evidence or Level 2 plus clinical support; Second line: Level 3 or higher plus clinical support; Third line: Level 4 or higher, plus clinical support. Clinical support refers to the application of expert opinion of the CANMAT committees to ensure that evidence-supported interventions are feasible and relevant to clinical practice. Therefore, treatments with higher levels of evidence may be downgraded to lower lines of treatment due to clinical issues such as side effects or safety profile.
TABLE 4-6 Pharmacologic Treatments for Acute Mania: Level of Evidencea and Line of Treatmentb
| Level of Evidence and Line of Treatment by Phase | |||||
|---|---|---|---|---|---|
| Acute | Maintenance | Acute | |||
| Acute Mania | Prevention of Any Mood Episode | Prevention of Depression | Prevention of Mania | Acute Depression | |
| First-line treatments: Monotherapies | |||||
| Lithium | Level 1 | Level 1 | Level 1 | Level 1 | Level 2 |
| Quetiapine | Level 1 | Level 1 | Level 1 | Level 1 | Level 1 |
| Divalproex | Level 1 | Level 1 | Level 3 | Level 2 | Level 2 |
| Asenapine | Level 1 | Level 2 | Level 2 | Level 2 | nd |
| Aripiprazole | Level 1 | Level 2 | Level 2 | nd | Negative evidence |
| Paliperidone | Level 1 | Level 2 | Level 2 | nd | nd |
| Risperidone | Level 1 | Level 4 | Level 4 | nd | nd |
| Cariprazine | Level 1 | nd | nd | nd | Level 1 |
| First-line treatments: Combination therapies | |||||
| Quetiapine + Li/DVP | Level 1 | Level 1 | Level 1 | Level 1 | Level 4 |
| Aripiprazole + Li/DVP | Level 2 | Level 2 | Level 2 | nd | Level 4 |
| Risperidone + Li/DVP | Level 1 | Level 4 | Level 4 | nd | Level 4 |
| Asenapine + Li/DVP | Level 3 | Level 4 | Level 4 | nd | Level 4 |
| Second-line treatments | |||||
| Olanzapine | Level 1 | Level 1 | Level 1 | Level 1 | Level 1 |
| Carbamazepine | Level 1 | Level 2 | Level 2 | Level 2 | Level 3 |
| Olanzapine + Li/DVP | Level 1 | Level 4 | Level 4 | Level 4 | nd |
| Li + DVP | Level 3 | Level 3 | Level 3 | nd | nd |
| Ziprasidone | Level 1 | Level 4 | Level 4 | nd | Negative evidence |
| Level of Evidence and Line of Treatment by Phase | |||||
|---|---|---|---|---|---|
| Acute | Maintenance | Acute | |||
| Acute Mania | Prevention of Any Mood Episode | Prevention of Depression | Prevention of Mania | Acute Depression | |
| Haloperidol | Level 1 | nd | Level 4 | Negative evidence | nd |
| ECT | Level 3 | Level 4 | Level 4 | Level 4 | Level 4 |
NOTE: ECT = electroconvulsive therapy; Li/DVP = lithium/divalproex; nd = no data.
a Levels 1 and 2 evidence refer specifically to treatment studies in which randomized comparisons are available. Recommendations involving epidemiologic or risk factors primarily arise from observational studies, and hence the highest level of evidence is usually Level 3.
b First line: Level 1 evidence or Level 2 plus clinical support; Second line: Level 3 or higher plus clinical support; Third line: Level 4 or higher, plus clinical support. Clinical support refers to application of expert opinion of the CANMAT committees to ensure that evidence-supported interventions are feasible and relevant to clinical practice. Therefore, treatments with higher levels of evidence may be downgraded to lower lines of treatment because of clinical issues such as side effects or safety profile.
mood symptoms, especially mood instability and mild depressive symptoms (Gitlin and Miklowitz, 2017). Cognitive impairment can be caused by depression, mania, or hypomania and also persists after symptomatic recovery. Cognitive impairment represents an independent impediment to functional recovery (Baune and Malhi, 2015; Gitlin and Miklowitz, 2016). Cognitive impairment may stem from medication side effects and experiencing psychosis during mood episodes (Gitlin and Miklowitz, 2016). Psychosocial stress (e.g., deterioration of social and financial supports) as well as personality factors and substance dependence can play a role in functional outcomes (Gitlin and Miklowitz, 2017). A number of other factors are associated with worse functional outcomes, such as lower education, being unmarried, poor sleep quality, receiving multiple psychiatric medications, and hospitalization (APA, 2013; Bonnin et al., 2019).
Bipolar-related impaired ability to work is especially pertinent to this report. As is the case with other mental disorders, functional recovery, including return to work, may lag behind or not occur despite a recovery from symptoms (Gitlin and Miklowitz, 2017). As a result, multiple investigations have examined the efficacy of treatments that combine medical management and psychosocial treatments in order to improve residual functional outcomes, including cognitive impairment and work disability. There is some evidence that work-focused interventions such as Vocational Case Management, a multifaceted supported employment intervention (Abdel-Baki et al., 2013; Drake et al., 2013), and cognitive remediation
plus supported employment (Ikebuchi et al., 2017) improve work outcomes in people with severe mental illness, including bipolar disorder. Evidence indicates that for persons on SSDI, robust return-to-work efforts rarely result in substantial gainful employment (Drake et al., 2013).
Length of Time to Improvement for Bipolar Disorders
The committee could not find any evidence to indicate clearly what the time to functional improvement in bipolar disorders is. In one large, longitudinal study of bipolar disorder under usual care conditions, 25 percent of individuals recovered from the onset of a mood episode within 5 weeks, 50 percent recovered within 13 weeks, 75 percent recovered within 38 weeks, and 85–89 percent recovered within 2 years (Solomon et al., 2010). Functional recovery is thought to lag behind symptomatic recovery and potentially depends on additional specific treatments to promote improved functioning in bipolar disorder (Gitlin and Miklowitz, 2017). Therefore, the rates of recovery suggested by Solomon and colleagues likely underestimate the time to functional improvement. Finally, a study of Social Security beneficiaries with bipolar, schizophrenia, or depression found that in the treatment arm of the study, employment increased from 5 to 30 percent by the end of 16 months, but the results may not be representative of these populations because of the low participation (Drake et al., 2013).
Both pharmacologic and psychologic treatments have a role in managing bipolar disorders. The 2018 CANMAT guideline on managing bipolar disorder concludes that despite limited research on treatment efficacy in older adults, medications that are efficacious in adults are likely to be efficacious in older adults (Yatham et al., 2018); that conclusion was thought to be true for managing acute mania, bipolar depression, and maintenance therapy. Therefore, improvement should be possible across all adult ages. With regard to psychologic treatments for bipolar-related depression, there is research demonstrating the efficacy of CBT for treating depression in both the acute and the maintenance phases of bipolar I disorder. Psycho-education is considered to be an evidence-based treatment only for the acute phase (see CANMAT guidelines).
OBSESSIVE COMPULSIVE DISORDER
OCD is a relatively common and potentially highly disabling disorder with substantial impacts on multiple domains of functioning. The illness is generally characterized by the presence of unwanted, recurring thoughts (obsessions) that generate compulsions, which are thoughts or acts performed to reduce distress or prevent an undesirable outcome related to the obsessions.
According to the NCS-R, OCD has a lifetime prevalence of 2.3 percent and a 12-month prevalence of 1.2 percent (Ruscio et al., 2010). The mean age of onset is 19.5 years old, and new onset rarely occurs after age 30 (Ruscio et al., 2010). The NCS-R study also found that males are more likely to develop early onset, with almost one-quarter of males developing the illness before age 10. In contrast, females tend to have higher rates of onset during adolescence. Overall, several epidemiologic studies of children and adolescents reported equal rates in boys and girls (Flament et al., 1988; Heyman et al., 2001). The disorder is evenly distributed across socioeconomic strata in most studies (Karno et al., 1988).
Almost 30 percent of individuals without OCD report experiencing obsessions or compulsions at some point in their lives. That finding underscores the general problem in obtaining an accurate understanding of the epidemiology of OCD. Prevalence estimates have varied widely, perhaps due to the differing definitions of the disorder and shifts in understanding of the threshold for having a diagnosis as opposed to subsyndromal symptoms (Ruscio et al., 2010). Notably, diagnoses of OCD obtained in the Epidemiological Catchment Area Study5 were found to have poor validity (Nelson and Rice, 1997), leaving estimates of the prevalence of OCD uncertain at that time. The NCS-R study found that individuals with lifetime OCD who had symptoms within the past year reported spending an average of 5.9 hours each day coping with obsessions and 4.6 hours per day engaging in compulsions (Ruscio et al., 2010). Such intense engagement would likely interfere with functioning.
The disability and reduced quality of life associated with a diagnosis of OCD is substantial. WHO has ranked OCD as the 10th leading cause of disability of all health conditions in the industrialized world. Specifically, the NCS-R found that nearly two-thirds (65.3 percent) of people who had been diagnosed with OCD and who had experienced symptoms for 12 months reported severe impairment. Just more than half (53.6 percent) reported any work-related impairment (Ruscio et al., 2010). In a large study of outpatients, Yaryura-Tobias and colleagues (2000) found that increased OCD symptoms were associated with reduced occupational functioning. A loss of work, reduction to part-time status, or work in occupations unrelated to professional training (i.e., nonprofessional work when trained for professional career) were common.
___________________
5 The Epidemiologic Catchment Area program of research was initiated in response to the 1977 report of the President’s Commission on Mental Health. The purpose was to collect data on the prevalence and incidence of mental disorders and on the use of and need for services by the mentally ill.
Professionally Accepted Diagnostic Criteria for Obsessive Compulsive Disorder
The diagnostic criteria for OCD changed to some extent between the DSM-IV, which was used before 2013, and DSM-5. Table A-6 in the Appendix shows the differences between DSM-IV and DSM-5. In DSM-IV, OCD was considered in the class of anxiety disorders. In DSM-5 it was considered in the class of obsessive compulsive and related disorders. DSM-5 also allows the specification of the presence of a current or a past tic disorder.
The professionally accepted current diagnostic criteria for OCD are described within the most recent DSM-5 and listed in Table 4-7.
Developmental Course, Gender Distribution, and Comorbidities
OCD is often chronic if left untreated. Remission rates for treated individuals are more promising. In a meta-analysis examining long-term remission in adults with OCD, remission rates were found to be 53 percent (Sharma et al., 2014). In that study, remission was defined as a Yale–Brown Obsessive Compulsive Scale (Y-BOCS) rating of less than 16 at the longest follow-up. The meta-analysis included 17 studies with a mean follow-up time of 4.91 years and was composed primarily of naturalistic studies. Being of female gender, experiencing a shorter duration of untreated illness, having had an onset in late adolescence or early adulthood, and having a lower baseline symptom severity were associated with better outcomes. Poorer insight has been linked to worse long-term outcome (APA, 2013). The DSM-5 also includes gender differences on symptomatology (APA, 2013) for OCD.
OCD can be comorbid with other psychiatric disorders. The most common comorbidities are depression (41 percent) and anxiety disorders, such as PD, generalized anxiety disorder (GAD), specific phobias, or social anxiety disorder (SAD) (76 percent) (DSM-5). Childhood onset is associated with high rates of ADHD, oppositional defiant disorder, and tic disorders (Janardhan Reddy, 2017). Tic disorder is most common in males with childhood onset (APA, 2013).
Additional comorbidities may include bipolar disorder, with a prevalence of 17 percent (Amerio et al., 2014), and schizophrenia, with a prevalence of 12.1 percent (Achim, 2009; Janardhan Reddy, 2017). Prevalence estimates for comorbid OCD and bipolar disorder are higher in children and adolescents than in adults (24.2 to 13.5 percent, respectively) (Amerio et al., 2014). OCD that is comorbid with bipolar disorder tends to be episodic in nature, with worsening symptoms during depressive periods and improvement in symptoms during manic or hypomanic periods
TABLE 4-7 DSM-5 Criteria for Obsessive Compulsive Disorder
| Criterion/Symptom Description |
|---|
|
A. Presence of obsessions, compulsions, or both: Obsessions are defined by (1) and (2):
Compulsions are defined by (1) and (2):
Note: Young children may not be able to articulate the aims of these behaviors or mental acts. |
| B. The obsessions or compulsions are time-consuming (e.g., take more than 1 hour per day) or cause clinically significant distress or impairment in social, occupational, or other important areas of functioning. |
| C. The obsessive compulsive symptoms are not attributable to the physiological effects of a substance (e.g., a drug of abuse, a medication) or another medical condition. |
| D. The disturbance is not better explained by the symptoms of another mental disorder (e.g., excessive worries, as in generalized anxiety disorder; preoccupation with appearance, as in body dysmorphic disorder; difficulty discarding or parting with possessions, as in hoarding disorder; hair pulling, as in trichotillomania [hair-pulling disorder]; skin picking, as in excoriation [skin-picking] disorder; stereotypies, as in stereotypic movement disorder; ritualized eating behavior, as in eating disorders; preoccupation with substances or gambling, as in substance-related and addictive disorders; preoccupation with having an illness, as in illness anxiety disorder; sexual urges or fantasies, as in paraphilic disorders; impulses, as in disruptive, impulse-control, and conduct disorders; guilty ruminations, as in major depressive disorder; thought insertion or delusional preoccupations, as in schizophrenia spectrum and other psychotic disorders; or repetitive patterns of behavior, as in autism spectrum disorder). |
| Specify if: With good or fair insight: The individual recognizes that obsessive compulsive disorder beliefs are definitely or probably not true or that they may or may not be true. With poor insight: The individual thinks obsessive compulsive disorder beliefs are probably true. With absent insight/delusional beliefs: The individual is completely convinced that obsessive compulsive disorder beliefs are true. Tic-related: The individual has a current or past history of a tic disorder. |
(Janardhan Reddy, 2017). Mood symptoms influence the type of treatment used because the SSRIs commonly used to treat OCD might induce mania or lead to rapid cycling (Janardhan Reddy, 2017). OCD symptoms are reported in approximately one-third of schizophrenia patients (Janardhan Reddy, 2017). Antipsychotics such as clozapine, risperidone, and olanzapine may cause or worsen obsessive compulsive symptoms (Janardhan Reddy, 2017).
Standard Measures of Outcomes for Obsessive Compulsive Disorder
The primary outcome measure used to evaluate improvements in symptoms is the Y-BOCS; there are versions for children and adults (Janardhan Reddy, 2017; Sharma et al., 2014). The Y-BOCS is a 10-item scale measuring symptom severity during the previous week. Scores greater than or equal to 16 are considered clinically significant (Janardhan Reddy, 2017). The Y-BOCS asks about time spent dealing with compulsions, impact on functioning, distress, and resistance against and degree of control over both obsessions and delusions. The Y-BOCS scores are correlated with the Quality of Life scale, and Y-BOCS scores greater than 20 have an impact on functioning (as cited in Sharma et al., 2014). The Y-BOCS functioning items are listed in Box 4-1.
A positive response to treatment that lasts for a week is indicated by a 35 percent or greater reduction in Y-BOCS scores and a Clinical Global Impression–Improvement score of 1 or 2 (1=very much improved, 2=much improved) (Janardhan Reddy, 2017). Remission is present when a patient no longer meets the DSM criteria for the disorder or has a score of 12 or less on the Y-BOCS and a Clinical Global Impression–Severity score of 1 or 2 (1=normal, not at all ill, 2=borderline mentally ill) for 1 week (Janardhan Reddy, 2017). During that time, residual obsessive compulsive symptoms might be present, but they are not time consuming or life interfering. To be considered to be in recovery, patients must meet those same criteria for remission for at least 1 year (Janardhan Reddy, 2017).
Treatments for Obsessive Compulsive Disorder
To the extent that the Y-BOCS, which covers both symptom severity and functioning, has been used in studies on which the practice guidelines are based, the treatments should have implications for both symptom reduction and functioning. At the same time, because functioning is embedded in the Y-BOCS, an overall score might not provide specific information about functioning. Scores of 3 or 4 on Y-BOCS items 2 and 7 would be most
associated with functional impairment. The committee did not find evidence suggesting that treatment recommendations vary by age in adults.
Psychotherapy for Obsessive Compulsive Disorder
The American Psychiatric Association (APA) practice guidelines indicate that CBT consisting of exposure and response prevention (ERP) should be a first-line treatment for patients who are not too severely ill, anxious, or depressed to comply with treatment (Koran et al., 2007). The recommended duration includes 13–20 weekly sessions or daily sessions for 3 weeks (Koran et al., 2007). Successful treatment should be followed by monthly boosters for 3–6 months to prevent relapse (Janardhan Reddy 2017; Koran et al., 2007).
Although the APA guidelines recommend ERP as a first-line treatment, the Canadian Clinical Practice Guidelines for Management of Anxiety, Post Traumatic Stress, and Obsessive Compulsive Disorders suggest that
outcomes are similar between interventions that focus on exposure and those that focus on cognitive interventions (Katzman et al., 2014). In one study, a cognitive intervention (danger ideation reduction therapy) without exposure was found to be more effective than the intervention with exposure (as cited in Katzman et al., 2014). Katzman and colleagues (2014) suggest that cognitive interventions may be more helpful for patients without overt compulsions, because exposure is more difficult with these patients. A review article on OCD (Grant, 2014) found that approximately 60–85 percent of patients have a reduction in symptoms after exposure therapy and that the improvement in symptoms is maintained for up to 5 years after treatment discontinuation. After cognitive therapy, about 60–80 percent of patients improve, but there is a 20–30 percent dropout rate (Grant, 2014).
With new advances in technology, there is a question regarding whether technology-delivered CBT is as effective as in-person CBT. The British Association for Psychopharmacology (BAP) guidelines indicate that although Internet-delivered CBT is better than online supportive therapy, therapist-led CBT is more effective than computerized CBT (Baldwin et al., 2014). A recent meta-analysis (Dèttore et al., 2015), examining the effectiveness of technology-delivered CBT versus control and therapist-administered CBT, found technology-delivered CBT was better than control on OCD symptoms but not on comorbid depression. The differences between the two methods of delivering treatment were not significant, even though there was a trend favoring therapist-administered CBT.
The effectiveness of individual versus group CBT is unknown (Baldwin et al., 2014). However, the National Institute for Health and Care Excellence (NICE) 2013 guidelines indicate that more people have clinical remission with group CBT rather than with sertraline medication. If patients are non-responsive to CBT, it is recommended that SSRIs or CBT plus SSRIs be used (Janardhan Reddy, 2017). Combined psychological and pharmacologic treatment has been shown to be superior to medication alone, but not to CBT alone (Katzman et al., 2014).
Medications for OCD
Serotonin reuptake inhibitors (SRIs) are recommended for patients who are unable to comply with CBT, prefer medication, or have previously responded well to medication (Koran, 2007). Sertraline, clomipramine, fluoxetine, fluvoxamine, and paroxetine are approved by the U.S. Food and Drug Administration (FDA) for the pharmacologic treatment of OCD (Koran et al., 2007). Although research suggests that clomipramine may have greater efficacy than other SSRIs, it is not typically recommended because of its side effects; therefore other SSRIs are recommended as a
first-line treatment (Baldwin et al., 2014; Bandelow et al., 2008; Koran et al., 2007). Clomipramine and other SSRI treatments result in a 20–40 percent symptom improvement in about 40–65 percent of patients (Grant, 2014).
During SRI treatment, the APA guidelines recommend increasing the dosage on a weekly basis to the maximum tolerated and FDA-approved dosage during the first month of treatment and continuing at that dose for at least 6 weeks (Koran et al., 2007). Other guidelines, such as those from NICE and BAP, recommend starting at lower doses and only increasing the dose if there is an insufficient response (Baldwin et al., 2014; NICE, 2006). It is recommended that effective treatment should be continued for at least 12 months to prevent relapse. APA guidelines recommend continuing successful medication treatment for 1–2 years before tapering by 10–25 percent every 1–2 months (Koran et al., 2007). Relapse rates are 25–40 percent if treatment is discontinued after 2 years and as high as 80 percent if treatment is discontinued earlier (Grant, 2014). The probability of full remission with pharmacotherapy alone is 11 percent (Grant, 2014). An early age of onset, severe OCD, tics, or hoarding symptoms are associated with poor response to pharmacotherapy (Grant, 2014).
The BAP guidelines also suggest that increasing the dose of an SSRI beyond formulary limits may be helpful or, alternatively, considering the augmentation of an SSRI or clomipramine with an antipsychotic (Baldwin et al., 2014). That is supported by evidence from a meta-analysis comparing antipsychotic augmentation and placebo augmentation in treatment-resistant OCD (Dold et al., 2013). In that study the antipsychotics aripiprazole, haloperidol, and risperidone were significantly more effective than placebo at reducing Y-BOCS scores (Dold et al., 2013). However, olanzapine, paliperidone, and quetiapine were not significantly different from placebo. Another potential treatment is electroconvulsive therapy, but the World Federation of Societies of Biological Psychiatry (WFSBP) guidelines state that this should be limited to treatment-refractory OCD (Bandelow et al., 2008).
Other Treatments
Most guidelines, such as those from NICE and WFSBP, indicate that transcranial magnetic stimulation (TMS)6 might not be an effective treatment for OCD (Bandelow et al., 2008; NICE, 2013). However, the Canadian clinical practice guidelines suggest that it may be helpful for improving comorbid depressive symptoms (Katzman et al., 2014). TMS is typically used when other depression treatments have not been effective.
___________________
6 Transcranial magnetic stimulation (TMS) is a non-invasive procedure that uses magnetic fields to stimulate nerve cells in the brain to improve symptoms of depression.
Deep high-frequency TMS was found to be more effective than sham TMS for individuals whose OCD did not satisfactorily respond to pharmacologic and psychologic interventions.
The Canadian clinical practice guidelines also indicate that deep brain stimulation might improve symptoms and functioning in up to two-thirds of patients with treatment-refractory OCD, based on data from small studies (Katzman et al., 2014). A meta-analysis of 31 deep brain stimulation studies for severe treatment-resistant OCD in 116 adults found a Y-BOCS reduction of 45.1 percent after treatment (Alonso et al., 2015). Better response was associated with an older age at onset and the presence of sexual or religious obsessions and compulsions.
Brown et al. (2016) conducted a systematic review of the literature and found supporting evidence for the efficacy of both dorsal anterior cingulotomy and anterior capsulotomy in highly treatment-refractory populations. Other experimental treatments that may improve symptoms include adjunctive moderate intensity aerobic exercise and herbal therapies such as milk thistle, valerian root, and St. John’s wort (Katzman et al., 2014).
Length of Time to Improvement for Obsessive Compulsive Disorder
According to the Canadian clinical practice guidelines, the optimal duration and intensity of treatment is a persistent question (Katzman et al., 2014). An intensive ERP therapy program provides 15 2-hour sessions delivered daily (5 days per week) over 3 weeks (Foa et al., 2005; Kuzak and Foa, 2004). Interestingly, a comparable twice-weekly program was similarly effective as the intensive 5 days per week strategy (Abramowitz et al., 2003). Another study that provided a step-care approach consisting of 6 weeks of low-intensity counseling with ERP bibliotherapy (i.e., reading materials relevant to the individual with OCD) and standard ERP for those who did respond was generally as effective as an initial therapy with standard ERP (17 sessions twice weekly) (Tolin et al., 2011). Accordingly, the APA practice guidelines indicate that the number of treatment sessions, their length, and the duration of an adequate trial have not been firmly and consistently established; nevertheless, expert consensus is that for most patients, treatment should consist of 13–20 weekly sessions. A number of studies that followed patients longitudinally have found that the benefits of CBT may last from 1–5 years (Braga et al., 2010; Jaurrieta et al., 2008; van Oppen et al., 2005).
In contrast to psychotherapy, psychopharmacologic treatments will not produce substantial improvement until patients have received 4–6 weeks of medication. Furthermore, in some patients treatment for as much as 10–12 weeks is necessary to see meaningful improvement.
Relapse prevention and naturalistic follow-up studies have provided
information about the long-term benefits of therapy. Generally speaking, such studies compare patients who have responded to medication and who are then randomized to placebo or continued active treatment. Overall, a meta-analysis of six relapse-prevention studies provided support for the ongoing benefits of treatment in reducing risk of relapse with SSRIs over 6–12 months (Donovan et al., 2010). A number of medications have shown benefits in randomized controlled trials (RCTs), including escitalopram (Fineberg et al., 2007), paroxetine (Hollander et al., 2003), sertraline (Koran et al., 2002), and high-dose fluoxetine (Romano et al., 2001). In addition, studies that have tested mirtazapine (Koran et al., 2005) and clomipramine (Katz et al., 1990) have demonstrated continued improvement compared with placebo in RCT discontinuation studies over approximately 6–12 months. Other studies provide evidence that fluoxetine, fluvoxamine XR, and sertraline are efficacious over 6–24 months (Bergeron et al., 2002; Koran et al., 2010; Rasmussen et al., 1997; Ravizza et al., 1996).
It is notable that complete relief from all OCD symptoms is uncommon with first treatments. If 13–20 weeks of weekly outpatient CBT treatment, 3 weeks of daily CBT, or 8–12 weeks of SRI treatment (including 4–6 weeks at the highest comfortably tolerated dose) does not produce an adequate response, it is reasonable for the clinician to consider, with the patient, whether to enhance or alter the treatment (see the APA guidelines).
Given that OCD can be a relapsing disorder, another set of challenges and decisions confronts patients who have responded to treatment and their clinicians: How long should such treatments be continued? APA guidelines suggest that successful medication treatment should be continued for 1–2 years. At that point it is reasonable to consider a gradual taper by decrements of 10–25 percent every 1–2 months with close monitoring of the possibility of symptom return or exacerbation. Regarding ERP, it is recommended that patients receive monthly booster sessions for 3–6 months following an initial successful course of ERP. Follow-up could be more intensive for individuals who have achieved only a partial improvement. It is difficult to know the relapse rates in discontinuation trials of medications because of methodologic differences across studies. Thus, a continued treatment of some form is recommended for most patients. The benefits of ERP may be more durable than those of some SRIs after discontinuation, but it is also possible that the observed differences in relapse rates across the two treatment types could be explained by other factors.
Relationship Between Symptomatic and Functional Improvement in Obsessive Compulsive Disorder
As previously mentioned, the treatment for OCD very likely improves functional outcomes based on improvements in the Y-BOCS. However,
there is scant information from treatment trials on the specific impact of different treatments on occupational functioning. A recent randomized trial comparing CBT (exposure and ritual prevention [EX/RP]) to risperidone and pill placebo found greater functional improvements among patients receiving EX/RP than among those who received risperidone or the pill placebo (Asnaani et al., 2017). Steketee (1997) noted that few treatment outcome studies have examined functional outcome; a few studies of ERP did establish improvement in occupational functioning with treatment and 1 year after treatment completion. A review evaluating the impact of in vivo exposure in treatment of individuals with anxiety disorders did find some benefits of such exposure on work-related outcomes for individuals with OCD (Noordik et al., 2010). Specifically, the authors found low- to high-quality evidence that exposure in vivo can reduce adverse work-related outcomes with a medium to large effect in different modalities and comparisons (group CBT versus SSRIs, group CBT plus SSRIs versus SSRIs, clinician-guided CBT versus systematic self-relaxation, exposure homework combined with clomipramine versus clomipramine with anti-exposure homework). They also found moderate evidence that exposure in vivo failed to improve adverse work-related outcomes in workers with OCD in three other modalities and comparisons (computer CBT at home via telephone versus systematic self-relaxation, exposure at home versus response prevention, and exposure at home plus response prevention versus response prevention). Additionally, Noordik et al. (2010) summarized a meta-analysis of two OCD studies representing the net contribution of exposure in vivo; their overall conclusion from those meta-analyses is that there is moderate evidence that anxiety treatments, including exposure in vivo, can improve work-related outcomes in workers with OCD.
POSTTRAUMATIC STRESS DISORDER
PTSD is a potentially chronic and disabling condition associated with significant morbidity and mortality as well as with disruptions in family, work, and social relationships (APA, 2013). Extensive research has documented the negative consequences of PTSD, including presence of other forms of psychophathology (Brown et al., 2001; Gradus et al., 2015a; Kessler et al., 1995; O’Donnell et al., 2004; Pietrzak et al., 2011), poor physical health (Gradus et al., 2015a, 2017; Hoge et al., 2007; Pacella et al., 2013; Schnurr and Jankowski, 1999), poor health-related quality of life (Fang et al., 2015: Goldberg et al., 2014; Schnurr et al., 2006, 2009), and mortality, specifically death by suicide (Boscarino, 2006; Gradus et al., 2015a,b, 2018; Kessler et al., 1999a). Thus, it is not surprising that PTSD has been described as conferring an important burden to individuals and society (Kessler, 2000).
Professionally Accepted Diagnostic Criteria for Posttraumatic Stress Disorder
The professionally accepted diagnostic criteria for PTSD are described within the DSM-5 (APA, 2013) and listed in Table 4-8. It is critical to note that the diagnostic criteria for PTSD have changed in significant ways in the past few years (VA/DoD, 2017). Prior to the current diagnostic criteria in the DSM-5, from 2004–2013, the DSM-IV was used to diagnose PTSD (APA, 2000). One of the more substantive changes is its new classification as a trauma- and stress-related disorder (versus an anxiety disorder in the DSM-IV). In addition, the definition of trauma has been clarified and narrowed. The number of symptom groups increased from three to four, and the number of symptoms increased from 17 to 20. Furthermore, acute and chronic PTSD specifiers were eliminated in the DSM-5, and the concept of “delayed-onset PTSD” was replaced with “delayed expression.” These changes have the potential for significant impact on the screening and diagnosis of PTSD. One study has estimated that a significant number of people (~50 percent) would be diagnosed under one set of criteria but not the other (Hoge et al., 2016), while other experts do not support that conclusion (Friedman et al., 2016). An additional important implication, particularly with regard to our understanding of the effect of available treatments, is that almost all research to date on recommended treatments for PTSD was performed among persons meeting the criteria for the previous DSM-IV definition and not the current DSM-5 definition of PTSD. Until more is known about the implications of the change in diagnostic criteria on PTSD screening, diagnosis, and treatment, this report must be read and interpreted with appropriate caution.
Developmental Course, Gender Distribution, and Comorbidities
According to the National Comorbidity Survey, the lifetime prevalence of PTSD in the U.S. general population is 8 percent, making it the fifth-most prevalent mental disorder in the United States (Kessler et al., 1995). The 12-month prevalence is 3.5 percent (Kessler et al., 2005a). More recent data from the National Epidemiologic Survey on Alcohol and Related Conditions reported a PTSD prevalence of 6.4 percent overall in the United States, with a prevalence of 8.6 percent in women and 4.1 percent in men (Pietrzak et al., 2011). PTSD is a common issue among U.S. veterans—approximately 10 percent of VA health care users have a PTSD diagnosis (Bernardy and Friedman, 2017). According to the DSM-5, PTSD can occur at any point from 1 year of age onward (APA, 2013).
Almost all people who experience a trauma will experience at least some symptoms consistent with PTSD in the immediate aftermath of the
TABLE 4-8 DSM-5 Criteria for Posttraumatic Stress Disorder
| Criterion/Symptom Description |
|---|
| Note: The following criteria apply to adults, adolescents, and children older than 6 years. |
A. Exposure to actual or threatened death, serious injury, or sexual violence in one (or more) of the following ways:
Note: Criterion A4 does not apply to exposure through electronic media, television, movies, or pictures, unless this exposure is work related. |
B. Presence of one (or more) of the following intrusion symptoms associated with the traumatic event(s), beginning after the traumatic event(s) occurred:
Note: In children older than 6 years, repetitive play may occur in which themes or aspects of the traumatic event(s) are expressed.
Note: In children, there may be frightening dreams without recognizable content.
Note: In children, trauma-specific reenactment may occur in play.
|
C. Persistent avoidance of stimuli associated with the traumatic event(s), beginning after the traumatic event(s) occurred, as evidenced by one or both of the following:
|
| Criterion/Symptom Description |
|---|
D. Negative alterations in cognitions and mood associated with the traumatic event(s), beginning or worsening after the traumatic event(s) occurred, as evidenced by two (or more) of the following:
|
E. Marked alterations in arousal and reactivity associated with the traumatic event(s), beginning or worsening after the traumatic event(s) occurred, as evidenced by two (or more) of the following:
|
| F. Duration of the disturbance (Criteria B, C, D, and E) is more than 1 month. |
| G. The disturbance causes clinically significant distress or impairment in social, occupational, or other important areas of functioning. |
| H. The disturbance is not attributable to the physiological effects of a substance (e.g., medication, alcohol) or another medical condition. |
|
Specify whether: With dissociative symptoms: The individual’s symptoms meet the criteria for posttraumatic stress disorder and, in addition, in response to the stressor, the individual experiences persistent or recurrent symptoms of either of the following:
Note: To use this subtype, the dissociative symptoms must not be attributable to the physiological effects of a substance (e.g., blackouts, behavior during alcohol intoxication) or another medical condition (e.g., complex partial seizures). |
| Specify if: With delayed expression: If the full diagnostic criteria are not met until at least 6 months after the event (although the onset and expression of some symptoms may be immediate). |
event. For many people the symptoms will resolve over time, usually in 1–3 months (APA, 2017). A recent meta-analysis of the natural course of PTSD remission found that without specific treatment, 44 percent of individuals with PTSD at baseline no longer met criteria for the diagnosis at approximately 10 months (Morina et al., 2014). Persons with PTSD symptoms that do not resolve over time, or that worsen, become eligible for a diagnosis of PTSD. While most people with PTSD experience an onset of symptoms immediately after a trauma and it is the lack of the resolution of these symptoms that ultimately leads to a diagnosis, research has shown that a significant proportion (approximately 15–25 percent) of the population may experience delayed onset PTSD in which the diagnostic criteria are not met until 6 months after the traumatic event (VA/DoD, 2017). The symptoms of PTSD can cause significant distress and impairment in functioning, yet many people with PTSD do not present for treatment for months or sometimes years after the symptoms begin (likely owing at least in part to the nature of the disorder, which involves avoidance of reminders of the event). Studies have shown that approximately 42.6 percent of adults with PTSD do not seek treatment and that the treatment is often inadequate among those persons who do seek treatment (Forman-Hoffman et al., 2018). According to a 2014 systematic review and meta-analysis of 42 observational prospective studies including 81,642 persons, 44 percent of individuals with PTSD no longer met criteria for the diagnosis after 10 months, but overall PTSD remission rates varied greatly across studies (from 8 to 89 percent) (Morina et al., 2014). It is important to note that the previous work was based on observational research and, in these studies, remission was not linked to any specific treatment. Therefore, an average remission rate cannot be implied because the remission rates in individual studies are not comparable.
Diagnoses of comorbid psychiatric disorders and physical health conditions are common among persons with PTSD (Forman-Hoffman et al., 2018). According to the APA publication Clinical Practice Guideline for the Treatment of Posttraumatic Stress Disorder in Adults (2017), commonly co-occurring psychiatric disorders and conditions include substance use and abuse, depression, anxiety, dissociation and dissociative disorders, personality disorders, psychosis, cognitive impairment, violence toward self and others, non-suicidal self-injury, and suicide. PTSD has also been shown to be associated with poorer physical health, morbidity, and increased health care use (VA/DoD, 2017). While PTSD itself is in part defined by compromised functional status, the co-occurrence of other disorders with PTSD can lead to further functional challenges and a decreased quality of life (VA/DoD, 2017). The adverse consequences of PTSD and comorbid disorders can be seen over the life course with impacts on many domains of functioning, including educational attainment, work stability, marriage, and family life.
Although comorbidity should not prevent patients from receiving PTSD treatment (VA/DoD, 2017), complications due to multiple comorbidities can make PTSD difficult to treat (APA, 2017).
Standard Measures of Outcomes for Posttraumatic Stress Disorder
In almost all studies of PTSD treatment, the main outcome of interest is PTSD symptom reduction (other secondary outcomes typically assessed include the loss of the PTSD diagnosis or improvement in functioning) as a marker of treatment efficacy (Forman-Hoffman et al., 2018). Both clinician-administered and patient-rated diagnostic instruments can be used for measuring outcomes. Among the clinician-administered measures, the Clinician-Administered PTSD Scale (CAPS) is a commonly used 30-item scale that assesses past-week, past-month, or lifetime PTSD and PTSD severity (Weathers et al., 2001). Versions of the CAPS have been developed that correspond to both the DSM-IV and the DSM-5 diagnostic criteria. Other clinician-administered PTSD scales include the PTSD Symptom Scale Interview, the Posttraumatic Diagnostic Scale, and the Structured Interview for PTSD (Forman-Hoffman et al., 2018). Among patient-rated or self-report measures, the PTSD checklist (PCL-5) is a commonly used 20-item measure (note that a previous version of the PCL that corresponded with the 17 items of the DSM-IV diagnostic criteria for PTSD was also commonly used, particularly in the research that generated the treatment results described below) (Weathers et al., 1993). Furthermore, a four-item primary care PTSD screening tool is often recommended and has demonstrated good reliability and validity (Spoont et al., 2015). Other patient-rated PTSD measures include: the PTSD Symptom Scale Self-report Version and the Impact of Events Scale (Forman-Hoffman et al., 2018).
Because compromised functioning is an essential criterion of PTSD diagnosis, scales related to the assessment of functioning have also been used in PTSD treatment studies. Outcome scales in this domain include the Global Assessment of Functioning, the Sheehan Disability Scale, and the Work and Social Adjustment Scale (Forman-Hoffman et al., 2018).
Treatments for Posttraumatic Stress Disorder
Although often thought of as a chronic condition, PTSD is treatable, even many years after a traumatic event. The committee did not find evidence suggesting that treatment recommendations vary by age in adults. As with many disorders, an early diagnosis and appropriate treatment of PTSD are crucial to ameliorating its symptoms and shortening the course of the disorder, as well as reducing functional impairment (Forman-Hoffman
et al., 2018). Despite this, there is no one universally preferred method for treating PTSD, and the various treatment guidelines that exist offer somewhat contradictory recommendations (Forman-Hoffman et al., 2018). Combine that with treatment complications that arise as a result of high levels of comorbid psychiatric disorders and variability in PTSD presentation, and it becomes clear why clinical uncertainty remains about which treatment to select for patients. A recent report from the Agency for Healthcare Research and Quality (AHRQ) provides the following broad guidance on the length of adequate treatment: “receiving either appropriate pharmacotherapy for 2 or more months for the focal disorder plus more than four visits to any type of physician or eight or more psychotherapy visits with any health care or human services professional lasting an average of 30 minutes or more” (Forman-Hoffman et al., 2018). Manualized trauma-focused psychotherapies for PTSD (described below) mostly involve 8 to 16 sessions with a therapist (VA/DoD, 2017).
Below is summary of the PTSD treatment recommendations from three clinical practice guidelines—specifically, the APA, the International Society for Traumatic Stress Studies (ISTSS), and the VA and DoD (APA, 2017; VA/DoD, 2017)—and the 2018 systematic review from the AHRQ (Forman-Hoffman et al., 2018). These recommendations fall broadly within two categories: psychologic and pharmacologic interventions.
Psychotherapy for Posttraumatic Stress Disorder
According to the VA/DoD joint document Clinical Practice Guideline for the Management of Posttraumatic Stress Disorder, the first-line recommendation for PTSD is psychotherapy with treatments that involve cognitive restructuring or exposure (as compared with non-trauma-focused psychotherapy or pharmacotherapy) (VA/DoD, 2017). The recommendation of trauma-focused psychotherapy as preferable to pharmacotherapy was based on the results of two meta-analyses that demonstrated that psychotherapy has greater and longer-lasting benefits on PTSD symptoms than pharmacotherapy (Lee et al., 2016; Watts et al., 2013). The trauma-focused psychotherapies generally recommended in the VA/DoD guideline include cognitive processing therapy (CPT), prolonged exposure (PE), and eye movement desensitization and reprocessing therapy (EMDR), specific CBTs for PTSD, brief eclectic psychotherapy (BEP), narrative exposure therapy (NET), and written narrative exposure. According to the VA/DoD guideline, the strongest evidence is shown for CPT, PE, and EMDR, with evidence of efficacy on amelioration of PTSD symptoms across trials and under varying methods and settings, while the other therapies listed above have sufficient (although potentially less strong) evidence to warrant their
use. Within the VA/DoD guideline, second-line psychotherapy treatments include non-trauma-focused interventions such as stress inoculation training, present-centered therapy (PCT), and IPT. In general, those treatments are described as likely not having as large an effect as trauma-focused psychotherapy, but they are useful options when trauma-focused therapy is not available or when preferred by the patient (VA/DoD, 2017).
The VA/DoD guideline is generally consistent with the ISTSS Posttraumatic Stress Disorder Prevention and Treatment Guidelines, which strongly recommends CBT, cognitive therapy, EMDR, PE, and trauma-focused CBT (Hoskins et al., 2015). The guideline further provides a recommendation of non-trauma-focused CBT, NET, and PCT (Hoskins et al., 2015). In contrast, guidelines from the APA do not distinguish any form of psychotherapy as a first-line treatment for PTSD, relative to pharmacotherapy, likely due to the few studies comparing the effectiveness of psychotherapy and pharmacotherapy for PTSD directly (APA, 2017). Overall, the APA treatment guidelines strongly recommend CBT, CT, and PE for the treatment of PTSD (APA, 2017). Those recommendations were largely upheld by a more recent (2018) systematic review from AHRQ (Forman-Hoffman et al., 2018). Although the term “first-line treatments” was purposely omitted from the APA guidelines, CBT, CPT, cognitive therapy, and PE were recommended more strongly than other forms of psychotherapy (APA, 2017).
Medications for Posttraumatic Stress Disorder
Within the VA/DoD guidelines (2017), pharmacotherapy is recommended as a second-line treatment only, when trauma-focused psychotherapy is either unavailable or unwanted by the patient. The VA/DoD guideline recommends three SSRIs—sertraline, paroxetine, and fluoxetine—and one SNRI—venlafaxine—as second-line treatments for PTSD (VA/DoD, 2017). Evidence for the efficacy of those medications comes from both systematic reviews and meta-analyses showing that these have a larger impact on PTSD symptoms than do other SSRIs and SNRIs (Davidson et al., 2006; Lee et al., 2016; Watts et al., 2013).
While the recommendations about psychotherapy vary somewhat among the VA/DoD, ISTSS, and APA guidelines, the recommendations for pharmacotherapy are more consistent with two important exceptions. First, the APA guidelines describe insufficient evidence for considering pharmacotherapy as a second-line treatment to psychotherapy, based on insufficient evidence from studies that directly compare the two (APA, 2017). However, when pharmacotherapy will be used for the treatment of PTSD, the APA guidelines recommend fluoxetine, paroxetine, sertraline, and venlafaxine, consistent with the VA/DoD guidelines, and describe these medications as all having moderate evidence for a small effect on PTSD
symptom reduction (APA, 2017). The AHRQ systematic review generally corroborates this viewpoint, with the exception of sertraline, for which the strength of evidence was reported as low (Forman-Hoffman et al., 2018). This is consistent with the ISTSS treatment guidelines, in which fluoxetine, paroxetine, sertraline, and venlafaxine are all described as having a “low” effect (Hoskins et al., 2015). The APA guidelines further recommend topiramate as having moderate evidence for a medium to large effect on symptoms (APA, 2017), but this finding was not mirrored in the AHRQ systematic review or the ISTSS treatment guidelines, which described the strength of the evidence for topiramate as limited (Forman-Hoffman et al., 2018; Hoskins et al., 2015).
Other Treatments
Various treatments have been used for PTSD that have insufficient evidence for their efficacy in treating PTSD symptoms. In psychotherapy, dialectical behavior therapy, skills training in affect and interpersonal regulation, acceptance and commitment therapy, and Seeking Safety need further research before use in the treatment of PTSD (VA/DoD, 2017). Additionally, the ISTSS treatment guidelines include couples trauma-focused CBT, group trauma-focused CBT, reconsolidation of trauma memories, single session CBT, written exposure therapy, and the medication quetiapine as treatments with emerging evidence (Hoskins et al., 2015).
Length of Time to Improvement for Posttraumatic Stress Disorder
People with PTSD may face functional deficits related to education, socioeconomic status, social relationships, and employment (APA, 2017). Studies of specific psychotherapy modalities, such as CT, PE, CBT, BEP, and IPT, have examined improvement in functioning as an outcome. For CT and CBT, the evidence generally indicates that these therapies are associated with an improvement in functioning over approximately 8 to 16 weeks of treatment (Forman-Hoffman et al., 2018). The evidence is less clear for improvements in functioning associated with PE, BEP, or IPT (Forman-Hoffman et al., 2018). Two studies have documented a benefit of venlafaxine on functioning, while other comparative effectiveness studies of venlafaxine and sertraline have found that sertraline may be more effective, but neither drug had a large impact on functioning outcomes (Forman-Hoffman et al., 2018). Similarly Forman-Hoffman et al. (2018) have found that paroxetine may improve functional status over a period of 12 weeks, while studies of other SSRIs have provided little evidence of efficacy with regard to improving functioning (Forman-Hoffman et al., 2018).
Relationship Between Symptoms and Functional Improvement in Posttraumatic Stress Disorder
A 2019 National Academies report reviewed the literature on PTSD and work-related functioning specifically (NASEM, 2019). Although research in this area is more limited, the report states that PTSD may reduce work functioning, particularly if the trauma occurred in the workplace. The few studies that exist have generally found that increased PTSD symptomatology is associated with unemployment and have also documented, more specifically, that the hyperarousal and reexperiencing symptoms of PTSD were most associated with receiving PTSD-related disability benefits. No studies have examined the impact of the specific PTSD treatments described above on improvements in work functioning specifically.
ANXIETY DISORDERS
This section focuses on the three most common clinically presented anxiety disorders: SAD, GAD, and PD (Andrews et al., 2018). Although there is considerable overlap between effective therapies for those anxiety disorders, there are also differences and, for the most part, separate evidence bases for treating each disorder. For these reasons the committee addresses each of those anxiety disorders separately.
The treatment effectiveness for anxiety disorders is evaluated in terms of symptomatic improvement (McKnight et al., 2016). Generally, treatment response is operationalized in terms of a certain magnitude of improvement (e.g., 30 to 50 percent) on symptom measures, and remission is judged in terms of a predetermined cutoff score (which focuses primarily on the frequency and severity of symptoms specific to each diagnosis). Thus, in keeping with the nature of the treatment literature for anxiety disorders, the committee describes anxiety disorder treatments that improve clinical outcomes (e.g., symptom response or remission) and time to improvement in clinical outcomes, which is generally the duration of effects for a specific intervention in clinical trials. The complexity of the relationship between symptomatic and functional improvement cuts across anxiety disorders and is discussed at the end of this section.
Social Anxiety Disorder
Professionally Accepted Diagnostic Criteria for Social Anxiety Disorder
SAD, also known as social phobia, is a mental disorder characterized by excessive and persistent fears of scrutiny, embarrassment, and humiliation in social or performance situations, leading to significant distress
or impairment in functioning. Individuals with SAD avoid feared social situations or endure them with intense distress. They often experience anticipatory anxiety, worrying for hours or days prior to a feared event. In social or performance situations, persons with SAD may also experience physical manifestations of anxiety, such as blushing, sweating, trembling, and palpitations, which sometimes can take the form of a full panic attack. People with SAD often fear that others will notice that they are irrationally anxious.
The DSM-5 criteria for SAD are listed in Table 4-9. Compared with the DSM-IV, Text Revision (TR), changes to the diagnostic criteria for SAD have been minimal. The DSM-5 includes SAD and a performance-only subtype (APA, 2013). This replaces the DSM-IV-TR subtypes of generalized and nongeneralized SAD. Persons with performance-only SAD have performance fears that are most impairing in their professional lives; they do not fear or avoid nonperformance social situations. Persons diagnosed with SAD not limited to performance situations have more severe symptoms and impairment than individuals with the performance-only subtype of SAD (Baldwin et al., 2014; Bögels et al., 2010). SAD that is not limited to performance situations is the more relevant disorder to general psychiatric clinical work and thus is considered for this statement of work. Furthermore, clinical trials of treatments for SAD were based on samples primarily composed of patients with the generalized form of the disorder.
Developmental Course, Gender Distribution, and Comorbidities
SAD is one of the most common types of anxiety disorder, with a 12-month prevalence of about 7 percent and a lifetime prevalence of about 12 percent in the United States (Ruscio et al., 2008). SAD typically begins in childhood or adolescence (median age of onset is 13 years), although it can occur as early as age 5 years (Grant et al., 2005; Kessler et al., 2005b). It often arises as an intensification of non-impairing shyness, although some patients will identify an unusually stressful social experience as a precipitant. New onset after age 30 years is uncommon (Beesdo et al., 2007). Left untreated, SAD has a chronic and unremitting course (Wittchen and Fehm, 2003). Risk factors for SAD include female gender (the female-to-male ratio is approximately 2:1), a family history of SAD, and early childhood shyness or behaviorally inhibited temperament (Grant et al., 2005; Hirshfeld-Becker et al., 2008; Kessler et al., 2005b; Low et al., 2008). SAD is associated with low socioeconomic status and being single or divorced (Acarturk et al., 2009; Schneier et al., 1992). SAD can be associated with extensive functional impairment, economic burden, and reduced quality of life (Katzman et al., 2014; Ruscio et al., 2008). Despite its early onset and the extent of distress and impairment, many individuals with SAD never seek treatment, and
TABLE 4-9 DSM-5 Criteria for Social Anxiety Disorder
| Criterion/Symptom Description |
|---|
| A. Marked fear or anxiety about one or more social situations in which the individual is exposed to possible scrutiny by others. Examples include social interactions (e.g., having a conversation, meeting unfamiliar people), being observed (e.g., eating or drinking), and performing in front of others (e.g., giving a speech). Note: In children, the anxiety must occur in peer settings and not just during interactions with adults. |
| B. The individual fears that he or she will act in a way or show anxiety symptoms that will be negatively evaluated (i.e., will be humiliating or embarrassing: will lead to rejection or offend others). |
| C. The social situations almost always provoke fear or anxiety. Note: In children, the fear or anxiety may be expressed by crying, tantrums, freezing, clinging, shrinking, or failing to speak in social situations. |
| D. The social situations are avoided or endured with intense fear or anxiety. |
| E. The fear or anxiety is out of proportion to the actual threat posed by the social situation and to the sociocultural context. |
| F. The fear, anxiety, or avoidance is persistent, typically lasting for 6 months or more. |
| G. The fear, anxiety, or avoidance causes clinically significant distress or impairment in social, occupational, or other important areas of functioning. |
| H. The fear, anxiety, or avoidance is not attributable to the physiological effects of a substance (e.g., a drug of abuse, a medication) or another medical condition. |
| I. The fear, anxiety, or avoidance is not better explained by the symptoms of another mental disorder, such as panic disorder, body dysmorphic disorder, or autism spectrum disorder. |
| J. If another medical condition (e.g., Parkinson’s disease, obesity, disfigurement from burns or injury) is present, the fear, anxiety, or avoidance is clearly unrelated or is excessive. |
| Specify if: Performance only: If the fear is restricted to speaking or performing in public. |
those who do generally only seek treatment after 15–20 years of symptoms (Wang et al., 2005). Among those presenting for clinical care, SAD tends to be particularly persistent (APA, 2013).
SAD generally co-occurs with other psychiatric disorders (Grant et al., 2005; Kessler et al., 2005b). Rates of comorbidity with depressive (30–50 percent) and other anxiety disorders (50–60 percent) are particularly high (NICE, 2013). Comorbidity with other anxiety disorders may reflect shared diagnostic features and possibly shared higher-order traits, such as harm avoidance, anxiety sensitivity, or neuroticism. SAD also increases the risk of other mental disorders, including depression and substance use disorders (SUDs) (Beesdo et al., 2007; Schneier et al., 2010). In patients with severe SAD, symptoms are pervasive and overlap with those of avoidant personality disorder (Friborg et al., 2013). About 15 percent of those with schizophrenia also have SAD (Achim et al., 2011). Persons with medical conditions that include highly visible symptoms, such as tremulousness from Parkinson’s disease, stuttering, facial disfigurement, and hyperhidrosis, may develop excessive social anxiety and meet DSM-5 criteria for SAD (Schneier et al., 2001).
Standard Measures of Outcomes for Social Anxiety Disorder
The Liebowitz Social Anxiety Scale (LSAS) was the first instrument developed specifically for SAD (Liebowitz, 1987). The original clinician-administered version and the self-report version have comparable and strong psychometric properties (Baker et al., 2002; Fresco et al., 2001). The other commonly used clinician-administered scale for SAD symptom assessment is the Brief Social Phobia Scale (Davidson et al., 1997). A 2015 systematic review of the psychometric properties of 22 self-report measures (including shortened derivatives) found that there was no “gold standard” self-report measure for social anxiety (Modini et al., 2015). Furthermore, other than the LSAS (Mennin et al., 2002), it is unknown which measures of outcome in SAD are commonly used in routine care. To evaluate outcomes, clinicians may use measures of anxiety that are not specific to social phobia, such as the Hamilton Anxiety Scale (Hamilton, 1959) or global measures of improvement such as the 7-point Clinical Global Impression (CGI) (Guy, 1976). Previous meta-analyses have shown that changes in the CGI among patients with anxiety disorders are broadly similar to changes in the other clinician-rated and patient-rated instruments (e.g., Bandelow et al., 2007).
Treatments for Social Anxiety Disorder
All clinical treatment guidelines for SAD recommend CBT, medications (usually antidepressants), or their combination for optimal management
(Andrews et al., 2018; Baldwin et al., 2014; Bandelow et al., 2012; Katzman et al., 2014; NICE, 2013). There is no consistent evidence that the efficacy of pharmacotherapy or psychotherapy for SAD varies by age (Davis et al., 2014; Eskildsen et al., 2010; Schneider et al., 2015). Fifty to 70 percent of patients respond to treatments for SAD in clinical trials (Andrews et al., 2018; Eskildsen et al., 2010); however, remission rates are lower (e.g., Leichsenring et al., 2013). Response and remission rates may be even lower in routine practice settings where patients, therapists, and clinical support systems are more variable. In general, clinical trials have not found one modality to be superior to the other (Canton et al., 2012). The onset of symptom response may be faster with pharmacotherapy, although CBT results in a more durable improvement (Baldwin et al., 2014; Canton et al., 2012; Haug et al., 2003; Liebowitz et al., 1999). In most studies, combined medications and psychotherapy have not resulted in improved patient outcomes relative to first-line monotherapy (Baldwin et al., 2014; Katzman et al., 2014; Mayo-Wilson et al., 2014). D-cycloserine, a partial agonist at the glycine recognition site of the glutamatergic NMDA receptor, seems to accelerate treatment response when used as an adjunct to exposure therapy for SAD—but within a narrow therapeutic window and only when exposure sessions are successful (Hofmann, 2014). The 2013 NICE guideline recommends CBT over pharmacologic interventions for SAD and the combination of psychotherapy and pharmacotherapy for individuals who are partial responders to either monotherapy after an adequate treatment course (Baldwin et al., 2014; NICE, 2013).
Psychotherapy for Social Anxiety Disorder
CBT developed for SAD is the best studied and most efficacious of the psychotherapies (Canton et al., 2012; Katzman et al., 2014). In CBT tailored for SAD, the therapist works with the patient to identify and challenge maladaptive cognitions associated with social situations and to confront these feared situations through in vivo exposure. A network meta-analysis including 101 clinical trials found CBT to be superior to no treatment, to pill placebo, to psychologic control conditions, and to several psychotherapies including psychodynamic psychotherapy, interpersonal psychotherapy, mindfulness, and supportive therapy (Mayo-Wilson et al., 2014). Network meta-analyses use all available data from randomized clinical trials to estimate the effect of each intervention relative to other interventions, even those that have not been compared directly. A 2019 meta-analysis concluded that CBT maintained superiority to control conditions for SAD beyond the 12-month period following treatment (van Dis et al., 2019).
CBT for SAD can be provided in individual or group formats. In the above-referenced network meta-analysis, individual CBT for SAD was the
most effective psychotherapy (Mayo-Wilson et al., 2014), and the NICE (2013) guideline recommends individual over group CBT for SAD. However, no statistical difference was reported between group and individual therapy for SAD, and a 2016 meta-analysis of 36 RCTs reported equivalence between group and individual therapies for SAD (Barkowski et al., 2016). Evidence is also accumulating to support self-administered (e.g., Internet-based or through printed material) CBT for SAD (e.g., Andrews et al., 2011; Hedman et al., 2011). It is, however, unclear whether therapist contact and guidance is a necessary component of self-administered CBT for SAD (Katzman et al., 2014), and effect sizes are generally larger for therapist-assisted self-help than for pure self-help (Andrews et al., 2018).
Exposure therapy is as effective as CBT for SAD at post-treatment, but evidence suggests that CBT results in better maintenance of treatment gains (Hofmann et al., 2004). Other therapies that have been found to be more effective than waitlist control7 but less effective than CBT include manual-based psychodynamic psychotherapy for SAD (NICE, 2013), interpersonal therapy (Stangier et al., 2011), and mindfulness-based therapy (Koszycki et al., 2007). Those therapies, therefore, may be useful for patients who do not want, cannot access, or do not fully benefit from CBT. Attention retraining, an intervention that modifies the attentional bias underlying many forms of psychopathology by training patients to attend to nonthreatening rather than threatening stimuli, may have benefits for individuals with SAD (Katzman et al., 2014; Li et al., 2008; Schmidt et al., 2009), though more robust trials are needed.
CBT has shown evidence of persistent benefits for as long as 5 years after treatment (Mörtberg et al., 2011; Powers et al., 2008), although some patients require longer-term treatment or subsequent “booster” sessions to maintain gains. A meta-analysis of RCTs of variants of CBT found that significant effects at post-treatment were maintained at follow-up, with no drop in effect sizes. However, less than half of SAD patients achieve remission from SAD even with CBT (Springer et al., 2018). Furthermore, many patients do not have access to quality CBT or are hesitant to enter psychotherapy (Wolitzky-Taylor et al., 2015).
Medications for Social Anxiety Disorder
Table 4-10 lists recommendations for pharmacotherapy for SAD from the 2014 Canadian clinical practice guidelines (Katzman et al., 2014). Those recommendations are largely consistent with other clinical guidelines
___________________
7 Waitlist control group in psychotherapy research is a group of participants who do not receive the experimental treatment, but who are put on a waiting list to receive the intervention after the active treatment group does.
TABLE 4-10 Recommendations for Pharmacotherapy for Social Anxiety Disorder
| First-linea | Escitalopram, fluvoxamine, fluvoxamine CR, paroxetine, paroxetine CR, pregabalin, sertraline, venlafaxine XR |
| Second-lineb | Alprazolam, bromazepam, citalopram, clonazepam, gabapentin, phenelzine |
| Third-linec | Atomoxetine, bupropion SR, clomipramine, divalproex, duloxetine, fluoxetine, mirtazapine, moclobemide, olanzapine, selegiline, tiagabine, topiramate |
| Adjunctive therapy | Third-line: pregabalin Not recommended: clonazepam, pindolol |
| Not recommendedd | Atenolol, buspirone, imipramine, levetiracetam, propranolol, quetiapine |
| Biological and alternative emerging therapies for which more data are needed | Repetitive transcranial magnetic stimulation, herbal preparations such as silexan, Galphimia glauca extract, passiflora, valerian; resistance training (weightlifting), aerobic exercise, acupuncture, meditation, and yoga-based treatments |
a First-line treatment recommendations derived from Levels 1 and 2 evidence plus clinical support for efficacy and safety.
b Second-line treatment recommendations derived from Level 3 evidence or higher plus clinical support for efficacy and safety.
c Third-line treatment recommendations derived from Level 4 evidence or higher plus clinical support for efficacy and safety.
d Not recommended treatments derived from Level 1 or Level 2 evidence for lack of efficacy.
SOURCE: Adapted from Katzman et al., 2014.
(Baldwin et al., 2014; Bandelow et al., 2012). There is, however, a paucity of studies examining pharmacotherapy for SAD in individuals with comorbid mental disorders, including SUDs.
SSRIs and the SNRI venlafaxine extended release (XR) are considered first-line treatments for SAD. Systematic reviews and a network meta-analysis confirmed that the classes of drugs that include SSRIs and SNRIs have a greater effect on outcomes than placebo (Mayo-Wilson et al., 2014). A 2017 Cochrane systematic review of 66 RCTs of medications versus placebo in the treatment of SAD found that SSRIs were the only medications that proved effective in reducing relapse based on moderate quality evidence and the only medication that demonstrated evidence of a reduction in functional disability (Williams et al., 2017).
Second-line medications that show efficacy in SAD have side effect profiles (MAOIs and benzodiazepines) or show less consistent effects (fluoxetine, moclobemide) in comparison with placebo (Katzman et al., 2014). Evidence for the effectiveness of adjunctive medications in partial responders and nonresponders comes from open trials and case series (Katzman et
al., 2014). Third-line agents are recommended for patients who have been found to be refractory to first- and second-line monotherapies and adjunctive therapies.
The findings of randomized placebo-controlled relapse-prevention studies in patients who have responded to previous acute treatment for SAD reveal a significant advantage for staying on active medication for at least 6 months (Baldwin et al., 2014; Blanco et al., 2013; NICE, 2013). Most people who respond to a SSRI will relapse within a few months if the drug is discontinued, and about 25 percent of those who continue will relapse within 6 months (e.g., Montgomery et al., 2005).
Length of Time to Improvement for Social Anxiety Disorder
Improvement in social anxiety symptoms, not function, is the primary outcome in clinical trials of interventions for SAD. Specific domains of functioning may be included as secondary outcomes, but functional outcomes are not reviewed or summarized in clinical treatment guidelines, systematic reviews, and meta-analyses of treatments for SAD. Furthermore, most studies assess only short-term outcomes and do not provide information on the durability of the treatment effects. Initial improvements in symptoms may not be maintained over time (Baldwin et al., 2014) and may not translate into improved functioning and reduced disability. Those characteristics of the extant literature presented a significant challenge for an evaluation of time to improvement in functioning in SAD. However, on average the duration of treatment in both medication and psychotherapy trials is 12 weeks (Mayo-Wilson et al., 2014).
A standard course of CBT for SAD involves approximately 14 1.5-hour sessions delivered over 3 to 4 months (Baldwin et al., 2014; NICE, 2013). Psychodynamic psychotherapy for SAD is of longer duration—it consists of 25–30 50-minute sessions over 6–10 months (Leichsering et al., 2013). However, the time to improvement in functioning may be considerably longer in routine practice due to various factors, including the characteristics of the patients, the skills of the therapists, and logistic considerations that preclude scheduling sessions at the same frequency used in RCTs.
Generally, a response to antidepressants is seen within 12 weeks of beginning treatment, which is the limit of most studies (Baldwin et al., 2014; Canton et al., 2012; Ipser et al., 2008). However, non-responders at 8–12 weeks may become responders with continuation of the same medication for 6–12 months (Ipser et al., 2008).
There is no consistent evidence that the efficacy of pharmacotherapy or psychotherapy for SAD varies by age (Davis et al., 2014; Eskildsen et al., 2010; Schneider et al., 2015).
Panic Disorder
Professionally Accepted Diagnostic Criteria for Panic Disorder
PD refers to the experiencing of recurrent panic attacks, with one or more attacks followed by at least 1 month of fear of another panic attack or significant maladaptive behavior related to the attacks. The DSM-5 criteria for PD are listed in Table 4-11. It should be noted that agoraphobia (i.e., where an individual avoids situations for fear of developing a panic attack) was considered to be a complication of PD in DSM-IV. The unlinking of PD and agoraphobia in DSM-5 reflects the current conceptualization that agoraphobia is a distinct disorder (APA, 2013; Wittchen et al., 2010). Because that change occurred relatively recently, much of the existing literature is based on mixed samples (i.e., those diagnosed with agoraphobia with or without panic attacks). Research suggests that agoraphobia in the absence of PD is relatively rare in treatment-seeking samples (Wittchen et al., 2010). PD frequently co-occurs with agoraphobia (Bienvenu et al., 2006), particularly in treatment-seeking samples (APA, 2010; Weissman et al., 1997).
Developmental Course, Gender Distribution, and Comorbidities
In the general U.S. population, the 12-month prevalence of PD approaches 3 percent when using the DSM-IV criteria (Grant et al., 2006; Kessler et al., 2005a, 2006). PD is approximately twice as common in women as in men (Kessler et al., 1994). Panic attacks (which can occur in disorders other than PD) are much more common than PD, occurring in up to one-third of individuals at some point in their lifetime (Kessler et al., 2006). Rates of PD gradually increase during adolescence, particularly among females, and peak during adulthood. Rates also decline in older (>60 years of age) adults (Kessler et al., 2005a). The median age of onset is 24 years (Kessler et al., 2005a). Generally, PD is a recurrent or chronic disorder with a waxing and waning course (APA, 2013; Batelaan et al., 2010).
Comorbidity with other anxiety, PTSD, depressive disorders, and SUDs is common (Grant et al., 2006; Kessler et al., 2006). The presence of agoraphobia in individuals with PD is associated with increased severity and worse outcomes (Bruce et al., 2005; Kessler et al., 2006; Porter and Chambless, 2015). PD is also more prevalent in individuals with medical conditions, including thyroid disease, hypoglycemia, seizure disorders, chronic pain, and cardiac conditions, among others (APA, 2010; Katzman et al., 2014). Some evidence suggests that the presence of medical comorbidity is associated with a greater severity of PD symptoms and functional limitations (Marshall et al., 2008).
TABLE 4-11 DSM-5 Criteria for Panic Disorder
| Criterion/Symptom Description |
|---|
|
A. Recurrent unexpected panic attacks. A panic attack is an abrupt surge of intense fear or intense discomfort that reaches a peak within minutes and during which time four (or more) of the following symptoms occur: Note: The abrupt surge can occur from a calm state or an anxious state.
Note: Culture-specific symptoms (e.g., tinnitus, neck soreness, headache, uncontrollable screaming, or crying) may be seen. Such symptoms should not count as one of the four required symptoms. |
B. At least one of the attacks has been followed by 1 month (or more) of one or both of the following:
|
| C. The disturbance is not attributable to the physiological effects of a substance (e.g., a drug of abuse, a medication) or another medical condition (e.g., hyperthyroidism, cardiopulmonary disorders). |
| D. The disturbance is not better explained by another mental disorder (e.g., the panic attacks do not occur only in response to feared social situations, as in social anxiety disorder; in response to circumscribed phobic objects or situations, as in specific phobia; in response to obsessions, as in obsessive compulsive disorder; in response to reminders of traumatic events, as in posttraumatic stress disorder; or in response to separation from attachment figures, as in separation anxiety disorder). |
Standard Measures of Outcomes for Panic Disorder
A complete psychiatric examination, including a medical history, thorough physical and neurologic examination, and standard laboratory testing, is needed to rule out organic causes of the symptoms and establish an accurate diagnosis of PD. Medical conditions such as angina, arrhythmias, asthma, chronic obstructive pulmonary disease, pulmonary embolus, thyroid disease, and, very rarely, temporal lobe epilepsy or pheochromocytoma may mimic panic attacks. Excessive caffeine or the use of other stimulants may trigger or worsen PD.
Clinician-administered and patient self-assessment instruments can be used to monitor changes in severity of PD. These measures assess the frequency of panic attacks and panic-related distress or impairment. The gold standard instrument for the disorder in the United States is the Panic Disorder Severity Scale, which has both clinician-administered and self-report versions (Shear et al., 1997, 2001; Wuyek et al., 2011). Each of the seven items in that scale (attack frequency, attack intensity, anticipatory anxiety, phobic avoidance, avoidance of internal bodily sensations, relationship impairment, work impairment) covers a key clinical aspect of the syndrome. To monitor treatment response, clinicians may use other interview or self-report scales specific to PD, general anxiety measures, or global rating scales that are not specific to any one type of psychiatric disorder.
Treatments for Panic Disorder
Clinical practice guidelines indicate that PD can be effectively treated with psychotherapy, medications (usually antidepressants), or a combination of the two (Andrews et al., 2018; APA, 2009; Katzman et al., 2014; NICE, 2011). A 2016 Cochrane systematic review concluded that there was no evidence of a difference between psychologic therapies and medications used to treat PD in terms of short-term remission, short-term response (defined as substantial improvement), or treatment acceptability as measured using dropouts for any reason (Imai et al., 2016). Therefore, the initial selection between antidepressant medication and therapy for most patients can be made on the basis of patient preference, motivation, and ability to engage in the treatment; prior treatment response; comorbidities; and treatment availability. Notably, there is wide variation across the United States in the availability of therapists trained to provide evidence-based psychotherapies for anxiety disorders (APA, 2010; Weissman et al., 2006), and a significant proportion (about 40 percent) of U.S. patients diagnosed with anxiety disorders remain untreated (Kroenke et al., 2007). Age is not associated with improvement in PD from CBT (Porter
and Chambliss, 2015). However, improvement may be related to age at the onset of PD. Specifically, a late age of onset of PD but not agoraphobia may be associated with improvement. Relatedly, a shorter duration of PD, but not agoraphobia, predicts greater improvement at post-treatment.
Psychotherapy for Panic Disorder
Among the psychosocial treatments for PD, CBT is most extensively supported by research. While there is no high-quality, unequivocal evidence to support one psychologic therapy over the others for the treatment of PD with or without agoraphobia, a 2016 network meta-analysis concluded that CBT is often superior to other psychotherapies (Pompoli et al., 2016). A 2019 meta-analysis concluded that CBT was superior to control conditions for PD until 12 months following treatment, after which it was equivalent to active comparison conditions (van Dis et al., 2019).
The central focus of CBT for PD is teaching patients a set of cognitive and somatic coping skills in an effort to manage anxiety as repeated exposures to feared situations and sensations are presented. Through repeated exposures patients learn that panic-related sensations are not harmful, that panic and anxiety can be managed or tolerated, and that they are able to accomplish tasks that were previously avoided.
CBT for PD can be effectively delivered in group or individual format as well as via self-help books, virtual reality, and Internet-based programs (Katzman et al., 2014). CBT treatment sessions are accompanied by homework assignments, usually daily, to be conducted between sessions. Thus, the individuals for whom CBT works best are generally highly motivated and value a problem-solving approach. Not all patients are able or willing to do the homework associated with CBT for PD (APA, 2010).
Other psychotherapies may also improve PD symptoms, although their efficacy is less well established. In the 2016 network meta-analysis (Pompoli et al., 2016), psychodynamic psychotherapies and supportive psychotherapy were found to have promising results, although further research is needed, particularly for supportive psychotherapy. Newer therapies for which evidence is accruing include third-wave therapies,8 which include features of CBT but focus on mindfulness, acceptance, and patients’ values. There were fewer dropouts in psychodynamic therapy and third-wave therapies than in CBT, suggesting that people with PD may tolerate these therapies particularly well compared with other forms of psychotherapy (Pompoli et al., 2016).
___________________
8 Third-wave therapies prioritize the promotion of psychologic and behavioral processes associated with health and well-being over the reduction or elimination of psychologic and emotional symptoms, which can be a side benefit.
Clinical trials have generally found CBT’s effects to be sustained over time, although relapse can occur (APA, 2009). A 2006 review of meta-analyses for CBT across disorders concluded that the evidence for the maintenance of treatment gains was particularly strong for PD, where the rate of relapse was almost half the rate of relapse following pharmacotherapy (Butler et al., 2006).
Chronic life stress, or episodic stressful events, can interfere with CBT for PD and agoraphobia. Relationship problems, job stress, financial hardship, and medical problems are examples of stressful situations that can both exacerbate panic symptoms and make it difficult for patients to engage in therapy (Sanderson and Bruce, 2007). A 2015 systematic review found that functional impairment at baseline was consistently related to decreased improvement in CBT for PD and agoraphobia (Porter and Chambless, 2015).
Medications for Panic Disorder
Table 4-12 lists recommendations for pharmacotherapy for PD from the 2014 Canadian clinical practice guidelines (Katzman et al., 2014). These recommendations are largely consistent with other clinical guidelines for PD. SSRIs and SNRIs are considered first-line agents for panic disorder. Second-line treatments are those that are less well tolerated and have higher discontinuation rates (as is the case with tricyclic antidepressants), present a risk for dependence (as is the case with benzodiazepines), or are less well researched (as in the case with mirtazapine). Although benzodiazepines are second-line options in these guidelines because of their addiction potential, they may be useful for the short-term management of acute or severe agitation or anxiety or at the initiation of SSRI treatment to hasten response (Katzman et al., 2014). The NICE Clinical Guideline for Generalized Anxiety Disorder and Panic Disorder in Adults (2011) recommends against the use of benzodiazepines for PD, noting less favorable long-term outcomes.
Patients who do not respond to first- or second-line agents are considered to have treatment-refractory illness. Third-line agents, adjunctive therapies, and biological and alternative therapies may be useful when patients fail to respond to first- and second-line therapies used alone and in combination.
Most studies that have examined maintenance effects report continued benefits of ongoing pharmacotherapy for PD (Katzman et al., 2014). Adding CBT to pharmacotherapy either from the start or at some later point in treatment may enhance long-term outcomes by reducing the likelihood of relapse when pharmacologic treatment is stopped (APA, 2010; Bruce et al., 1999; Katzman et al., 2014; Otto et al., 2010).
TABLE 4-12 Recommendations for Pharmacotherapy for Panic Disorder
| First-linea | Citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, paroxetine CR, sertraline, venlafaxine XR |
|---|---|
| Second-lineb | Alprazolam, clomipramine, clonazepam, diazepam, imipramine, lorazepam, mirtazapine, reboxetine |
| Third-linec | Bupropion SR, divalproex, duloxetine, gabapentin, levetiracetam, milnacipran, moclobemide, olanzapine, phenelzine, quetiapine, risperidone, tranylcypromine |
| Adjunctive therapy | Second-line: alprazolam ODT, clonazepam Third-line: aripiprazole, divalproex, olanzapine, pindolol, risperidone |
| Not recommendedd | Buspirone, carbamazepine, propranolol, tiagabine, trazodone |
| Biological and alternative emerging therapies for which more data are needed | Non-invasive brain stimulation using a radioelectric asymmetric conveyor, repetitive transcranial magnetic stimulation, capnometry-assisted respiratory training, acute aerobic exercise |
NOTE: ODT = orally disintegrating tablet.
a First-line treatment recommendations derived from Levels 1 and 2 evidence plus clinical support for efficacy and safety.
b Second-line treatment recommendations derived from Level 3 evidence or higher plus clinical support for efficacy and safety.
c Third-line treatment recommendations derived from Level 4 evidence or higher plus clinical support for efficacy and safety.
d Not recommended treatments derived from Level 1 or Level 2 evidence for lack of efficacy.
SOURCE: Adapted from Katzman et al., 2014.
Length of Time to Improvement for Panic Disorder
CBT PD protocols usually involve 12–14 weekly sessions and sometimes include booster sessions following treatment. However, briefer treatment courses and compressing the duration of therapy by administering multiple sessions per week have also been shown to be effective (Katzman et al., 2014). Some evidence suggests that the less intensive protocols do not work as well with patients with more severe PD (Dow et al., 2007). The 2011 NICE clinical guideline specifies that for most people CBT should take the form of weekly sessions of 1–2 hours and should be completed within a maximum of 4 months of commencement.
Although the time to onset of clinically meaningful action for a pharmacologic agent varies by patient, the treatment is often associated with a delay of about 2–8 weeks in the onset of symptom relief, with full response taking up to 12 weeks or more (Katzman et al., 2014). Longer-term therapy has been associated with continued symptomatic improvement and the prevention of relapse. Clinical guidelines for PD recommend that pharmacotherapy be continued for 6–24 months or more after the desired level of
improvement (Andrews et al., 2018; APA, 2010; Bandelow et al., 2012; Katzman et al., 2014; NICE, 2011).
Despite promising findings in clinical trials, a substantial minority of patients who receive evidence-based medications for PD or agoraphobia, or both, fail to show significant symptom improvement. In clinical trials, about one-third of patients are classified as non-responders (Taylor et al., 2012; Westen and Morrison, 2001). Evidence from naturalistic follow-up studies of patients in a tertiary-care setting suggests that at 4–6 years post-treatment about 30 percent of individuals are well, 40–50 percent are improved but symptomatic, and the remaining 20–30 percent have symptoms that are the same or slightly worse (Katschnig et al., 1996; Roy-Byrne and Cowley, 1994).
Age has not been associated with improvement in studies of CBT for PD (Porter and Chambless, 2015). However, improvement may be related to age of onset of PD; specifically, a late age of onset of PD—but not agoraphobia—may be associated with improvement. Relatedly, shorter duration of panic, but not of agoraphobia, predicts greater improvement post treatment.
Generalized Anxiety Disorder
Professionally Accepted Diagnostic Criteria for Generalized Anxiety Disorder
GAD refers to excessive anxiety and worry for more days than not regarding multiple events or activities and lasting for at least 6 months. That apprehension is associated with somatic symptoms. Since its introduction into the DSM classification in 1980, the conceptualization of GAD has evolved. However, there was not a major change in the diagnostic criteria for GAD in the DSM-5 compared with the DSM IV-TR (APA, 2000). The DSM-5 (APA, 2013) criteria for GAD are listed in Table 4-13.
Developmental Course, Gender Distribution, and Comorbidities
In the general population in the United States, the 12-month prevalence of GAD is approximately 3 percent (Kessler et al., 2005a). GAD is approximately twice as common in women as in men (Kessler et al., 1994). The age of onset is variable, with a median of approximately 31 years (Kessler et al., 2005b). There is an increase of new onset cases in older adults, often in the context of chronic physical health conditions (Byers et al., 2010; Mackenzie et al., 2011). GAD is frequently underrecognized (Baldwin et al., 2014).
GAD is associated with high rates of psychiatric comorbidity, particularly with other anxiety disorders, depression (Grant et al., 2005; Kessler et
TABLE 4-13 DSM-5 Criteria for Generalized Anxiety Disorder
| Criterion/Symptom Description |
|---|
|
al., 2005a) and SUD (Robinson et al., 2011). The risk of co-occurring medical conditions is also elevated, including pain syndromes, hypertension, and cardiovascular and gastric conditions (Comer et al., 2011). Comorbidity with depression or depression and pain is associated with a more severe and prolonged course of illness and with greater functional impairment (Kessler et al., 1999b). GAD has a low likelihood of spontaneous remission (Lenze et al., 2005). A longitudinal study found that the likelihood of recovery from GAD is significantly less than that of recovering from major depression (Yonkers et al., 2000). Among those who do recover, relapse is common. Based on data from the Harvard/Brown Anxiety Disorders Program, Bruce and colleagues (2005) found that nearly half of patients with GAD who recovered had a recurrence over a 12-year follow-up period.
Standard Measurement of Outcomes in Generalized Anxiety Disorder
An in-depth, structured interview by a medical specialist is the first step in establishing the diagnostic features and details of associated behaviors in GAD. An instrument such as the Anxiety Disorders Interview Schedule for DSM-5 (Brown and Barlow, 2014) can be used to collect data on the domains of worry and the somatic symptoms.
Clinician-administered and patient self-assessment instruments can be used to monitor changes in severity of GAD. These measures assess the cognitive and somatic symptoms of anxiety. Clinical trials often use the Hamilton Anxiety Rating Scale, which is a clinician-rated 14-item measure (Hamilton, 1959). Psychotherapists most commonly use self-report measures to assess GAD symptoms over the course of treatment (Szkodny et al., 2014). The Generalized Anxiety Disorder seven-item scale is a commonly used brief self-report measure (Spitzer et al., 2006). Global measures are also used, such as the CGI scale (Guy, 1976), which consist of two items, one assessing illness severity and the other assessing change from the initial treatment. In practice, clinicians often base their assessments of improvement on subjective impressions rather than on validated measures.
Treatments for Generalized Anxiety Disorder
Clinical practice guidelines indicate that GAD can be effectively treated with psychotherapy or medications (usually antidepressants) (Andrews et al., 2018; Baldwin et al., 2014; Katzman et al., 2014; NICE, 2011). Although few studies have compared psychotherapy and pharmacotherapy in the same trial, the magnitude of benefit appears comparable (Katzman et al., 2014). Few data are available on the use of combined psychologic and pharmacologic treatment for GAD. A meta-analysis concluded that combination pharmacotherapy and CBT was more effective than CBT alone at post-treatment but not at the 6-month follow-up (Hofmann et al., 2009). There is no current evidence to support the routine combination of CBT and pharmacotherapy (Baldwin et al., 2014; Katzman et al., 2014). Age does not appear to moderate treatment effects in individuals with GAD. A systematic review and meta-analysis of the efficacy of controlled interventions for GAD in adults 55 years and older concluded that older adults with GAD benefited from both pharmacologic and psychotherapeutic interventions (Goncalves and Byrne, 2012). A separate 2017 meta-analysis comparing the efficacy of CBT for GAD between working-age and older adults found that there was no statistically significant difference in the effect size for outcomes between the two groups (Kishita and Laidlaw, 2017).
Psychotherapy for Generalized Anxiety Disorder
A 2014 meta-analysis concluded that psychotherapies, especially CBT, are effective in the treatment of GAD in adults and in reducing depression symptoms in individuals with GAD (Cuijpers et al., 2014b). Because of the strength of the evidence, CBT is considered the first-line psychotherapy for GAD (Katzman et al., 2014). CBT for GAD is based on evidence that shows that individuals with GAD tend to overestimate the likelihood of negative events, have low confidence in their problem-solving abilities, and have low tolerance of uncertainty as well as other maladaptive cognitions. CBT for GAD involves challenging and disrupting the misconceptions that maintain worry; actively testing the validity of erroneous beliefs; improving skills to manage worry and anxiety; and developing more adaptive ways of responding to neutral and ambiguous situations (Katzman et al., 2014; Szkodny et al., 2014).
CBT for GAD can be effectively delivered in both individual and group format. However, individual CBT may lead to earlier improvements (see Katzman et al., 2014). Internet- and computer-based CBT programs have also been found to be effective for relieving GAD symptoms (Cuijpers et al., 2014b; Olthuis et al., 2016).
Other psychotherapies may also improve GAD symptoms, though their efficacy has been less well established. These include applied relaxation therapy (Siev and Chambless, 2007), psychodynamic psychotherapy (Leichsenring et al., 2009), and third-wave CBT, including acceptance-based behavioral therapy (Roemer et al., 2008) and metacognitive therapy (van der Heiden et al., 2012). The 2011 NICE clinical guideline recommends applied relaxation along with CBT for individuals with GAD and marked functional impairment.
Meta-analyses suggest that the benefits of psychologic treatments for patients with GAD are maintained after treatment (Covin et al., 2008; Cuijpers et al., 2014b). On average, approximately 50 percent of individuals who undergo treatment for GAD meet the criteria for responder status at 6- and 12-month follow-ups (Szkodny et al., 2014). CBT is associated with moderate GAD symptom reduction relative to control conditions beyond 12 months following treatment (van Dis et al., 2019). Relapse rates may also be lower with CBT than with other forms of psychologic treatment (Baldwin et al., 2014).
Medications for Generalized Anxiety Disorder
Table 4-14 lists recommendations for pharmacotherapy for GAD as presented in the Canadian clinical practice guidelines (Katzman et al.,
TABLE 4-14 Recommendations for Pharmacotherapy for Generalized Anxiety Disorder
| First-linea | Agomelatine, duloxetine, escitalopram, paroxetine, paroxetine CR, pregabalin, sertraline, venlafaxine XR |
|---|---|
| Second-lineb | Alprazolam, bromazepam, bupropion XL, buspirone, diazepam, hydroxyzine, imipramine, lorazepam, quetiapine XR, vortioxetine |
| Third-linec | Citalopram, divalproex chrono, fluoxetine, mirtazapine, trazodone |
| Adjunctive therapy | Second-lineb: pregabalin Third-linec: aripiprazole, olanzapine, quetiapine, quetiapine XR, risperidone Not recommended: ziprasidone |
| Not recommendedd | Beta blockers (propranolol), pexacerfont, tiagabine |
| Biological and alternative emerging therapies for which more data are needed | Repetitive transcranial magnetic stimulation, herbal preparations such as silexan, Galphimia glauca extract, passiflora, valerian; resistance training (weightlifting), aerobic exercise, acupuncture, meditation, and yoga-based treatments |
a First-line treatment recommendations derived from Levels 1 and 2 evidence plus clinical support for efficacy and safety.
b Second-line treatment recommendations derived from Level 3 evidence or higher plus clinical support for efficacy and safety.
c Third-line treatment recommendations derived from Level 4 evidence or higher plus clinical support for efficacy and safety.
d Not recommended treatments derived from Level 1 or Level 2 evidence for lack of efficacy.
SOURCE: Adapted from Katzman et al., 2014.
2014). These recommendations are largely consistent with other clinical guidelines. SSRIs, SNRIs, and pregabalin are first-line agents (Bandelow et al., 2012; Katzman et al., 2014). Response rates to these medications range from 30 to 56 percent (Kapczinski et al., 2003). Some agents, such as benzodiazepines, imipramine, and quetiapine XR, have demonstrated efficacy for the treatment of GAD but have been classified as second-line agents because of their risk profiles. Third-line agents are those with limited data or significant side effects. Adjunctive therapies can be considered for patients with treatment-resistant GAD. Biological and alternative therapies are emerging treatments for which more data are needed. One other clinical guideline specifies that benzodiazepines should only be offered as a short-term measure during crises (NICE, 2011).
Relapse prevention studies indicate an advantage for responders to staying on active medication for at least 6 months, although there are additional benefits of treatment continuation beyond 1 year (Donovan et al., 2010; Mochovitch et al., 2017). It is common in clinical practice for both
the doctor and patient to agree to maintain medication across the lifespan. Adjunctive CBT was shown to facilitate benzodiazepine tapering (Gosselin et al., 2006) and may reduce relapse rates (Baldwin et al., 2014).
Length of Time to Improvement for Generalized Anxiety Disorder
The optimal duration of treatment for GAD has not been determined. CBT for GAD protocols usually involve 10–16 weekly 1-hour sessions and sometimes include booster sessions over the phone or in person following treatment. More sessions may be required for partial responders or after relapse.
Although the time to onset of clinically meaningful action for a pharmacologic agent varies by patient, signs of improvement may begin within 4 weeks and continue to increase through months 4–6 of treatment. Longer-term therapy has been associated with continued symptomatic improvement and the prevention of relapse (Montgomery et al., 2005).
Relationship Between Symptomatic and Functional Improvement in Anxiety Disorders
The available literature does not permit making strong conclusions about the influence of treatments for anxiety disorders on functioning or work-related disability, let alone about the length of time to treatment-related improvement in functioning to the point where the condition is no longer disabling. In general, evidence-based anxiety disorder treatments target the core symptoms of the specific anxiety disorder, and symptomatic improvement is the primary endpoint and focus of meta analyses and systematic reviews of treatments. Thus, there is much less evidence on the functional outcomes associated with psychologic and pharmacologic treatments than on clinical response and remission. Furthermore, because the relationship between anxiety disorder symptoms and functioning appears to be weak, one cannot draw conclusions about functioning based on information about symptom improvement. A systematic review of the relationship between symptoms and functioning in individuals with common anxiety disorders found a modest overall relationship (McKnight et al., 2016), which was smaller than had been observed between symptoms of depression and functioning in an earlier study (McKnight and Kashdan, 2009). Of particular relevance to SSA, the average correlations for GAD, SAD, and PD symptoms and occupational functioning were 0.20, 0.12, and 0.01, respectively.
The finding of weak correlations between symptoms and functioning in individuals with anxiety disorders is consistent with research demonstrating that remission in anxiety disorders does not necessarily translate
into the levels of functioning seen in healthy controls. Iancu and colleagues (2014) studied trajectories of functioning after remission from anxiety disorders and found that while anxiety disorders’ symptom remission was accompanied by improvements in functioning, impairments persisted in all functional areas except self-care. Furthermore, the magnitude of improvement varied by domain—those with remitting anxiety disorders had moderate improvement in interpersonal functioning, participation in society, and cognition but not in work or household functioning. Possible reasons for the persistence of functional impairment among those with remitting anxiety disorders include comorbidities, premorbid functional limitations that are not eradicated through mental health treatments, and subthreshold symptoms that affect functioning (Iancu et al., 2014). Mental scarring in which the psychiatric disorder causes irreversible damage may also affect postmorbid functioning in those with a history of severe or recurrent anxiety disorders (Schopman et al., 2018). Importantly, impaired functioning appears to predispose individuals for an onset or recurrence of anxiety disorders (Rodriguez et al., 2005; Saris et al., 2017; Scholten et al., 2013; Schopman et al., 2018). In sum, even after treatment response or remission from anxiety disorder, individuals may continue to have significant functional impairments, which in turn may predispose them to a relapse of the anxiety disorder.
SUMMARY AND CONCLUSIONS
The committee selected eight mental health disorders for inclusion in the report: MDD, bipolar I disorder, bipolar II disorder, OCD, PTSD, PD, GAD, and SAD. Those mental health disorders are highly prevalent, are associated with significant functional impairment, and may respond to treatment. Professionally accepted diagnostic criteria for these conditions are detailed in the DSM-5.
People diagnosed with a mental health disorder are directed to a specific treatment depending on clinical practice guidelines for treatment, their treatment history, their treatment preference, and treatment availability, among other factors. The committee expects that most patients who are disabled by psychiatric disorders are receiving psychiatric services by combinations of mental health professionals, including a prescriber (e.g., psychiatrist, advanced practice nurse), psychologist, licensed clinician social workers, and individuals with counseling or rehabilitation degrees. The conclusions here, however, should be interpreted with the caveat that for some populations (e.g., those in rural areas or small towns), care from qualified mental health professionals (e.g., specialized in evidence psychotherapy) might not be available. Importantly, the committee cautions that even under ideal treatment, full remission of mental health
disorders, particularly when already determined as disabling, is seldom achieved. Disorder-specific clinical practice guidelines detail evidence-based treatments for the eight disorders that the committee reviewed. Generally, those mental health disorders can be treated effectively with psychotherapy, pharmacotherapy or other biologic treatments, or a combination of both. There is no indication that improvement varies with age. However, some individuals do not improve after receiving evidence-based treatments, and among those who do improve, some will relapse. Furthermore, it is uncertain whether the rates of remission and response observed in the scientific literature can be generalized to those receiving SSDIs or SSIs on the basis of a mental health disorder.
For the most part, in the clinical trials of treatments for mental health disorders improvement is defined in terms of disorder-specific symptoms, not functioning. Work-related disability is rarely assessed as an outcome. Furthermore, because there are no evidence-based laboratory tests for mental health disorders, mental health outcomes are assessed using patient self-report measures or clinician assessments.
There is a dearth of data on the length of time from start of treatment until the person’s functioning improves to the point where the mental health disorder is no longer disabling. Attempting to accurately describe time to functional improvement by drawing from the existing data has important limitations. First, as mentioned above, treatment efficacy in research trials is generally defined in terms of symptomatic improvement, not functional outcomes, and time to symptomatic improvement is restricted to the duration of the trials. Second, psychiatric disorders are often recurrent, so time until improvement cannot be adequately captured as a linear process. Thus, individuals may have periods of remission during which they no longer meet the criteria for disability and later have an exacerbation of illness and associated functional limitations during which they again meet the criteria for disability. Third, the relationship between changes in symptoms and functioning is complex, and symptomatic improvement may not correspond to contemporaneous improvements in functioning. Fourth, psychiatric disorders generally occur with other psychiatric disorders, chronic pain, and medical conditions, and time to improvement will depend on those and other factors. Any estimates of time to improvement needs to consider the fact that clinical trials generally exclude participants with comorbidities. Fifth, the mental health disorders discussed in the report are under-recognized and effective treatments, particularly evidence-based psychotherapies, are often unavailable. That is particularly true for OCD. However, based on the limited evidence, the committee made the following conclusions regarding time from the start of treatment to improvement in functioning.
With regard to MDD, functional improvement may lag behind or not occur even when a person is in symptomatic remission and may require rehabilitation that targets a return to work. Even then, recovery of occupational functioning, if it occurs, may take 1–2 years and may be limited by environmental contingencies. Early response to treatment might predict likelihood of improvement.
For bipolar I disorder, the acute phase of treatment lasts 6–12 weeks, while the maintenance phase treatment, which focuses on functional recovery, lasts 6–24 months. Caveats include the fact that improvement in social and occupational functioning may be limited or delayed and require targeted rehabilitation efforts. High-quality research shows that even with the addition of vocational rehabilitation, the potential for return to gainful employment may be limited due in part to financial and other environmental disincentives.
Time to improvement can range from 12 to 24 weeks in OCD. Individuals requiring higher doses of medication or more complex cases may take 1 year or more to receive the full treatment benefit. For PTSD, there is some evidence from clinical trials indicating that general functioning improves in response to psychotherapy modalities. The length of time to improvement in functioning varies across psychotherapy modalities and usually corresponds to clinical trial follow-up endpoints (e.g., 8 or 16 weeks). Evidence regarding improvement in functioning from pharmacotherapy studies is less convincing, and the literature on improvements in work functioning specifically following PTSD treatment is scant.
For PD, GAD, and SAD, the time to improvement in symptoms in clinical trials is generally about 3 months. A longer treatment period may be required for partial responders or after relapse. Time to improvement in routine practice is likely considerably longer than in randomized controlled trials because of patient clinical complexity, treatment history, psychosocial factors, and variability in treatment delivery. Notably, the relationship between symptoms and functioning in individuals with anxiety disorders is weak. Even after treatment response or remission from an anxiety disorder, individuals may continue to have significant functional impairments, which in turn may predispose them to a relapse of the anxiety disorder.
The committee notes that all of those conditions may be associated with chronic pain, which may contribute to increased risk for mental health disorders, and mental health disorders may result in an increased risk of chronic pain. The types of chronic pain that commonly co-occur with mental health conditions include migraine headaches, neck and back pain, fibromyalgia, and abdominal pain.
REFERENCES
Abdel-Baki, A., G. Letourneau, C. Morin, and A. Ng. 2013. Resumption of work or studies after first-episode psychosis: The impact of vocational case management. Early Intervention in Psychiatry 7(4):391–398.
Abramowitz, J. S., E. B. Foa, and M. E. Franklin. 2003. Exposure and ritual prevention for obsessive–compulsive disorder: Effects of intensive versus twice-weekly sessions. Journal of Consulting and Clinical Psychology 71(2):394–398.
Acarturk, C., F. Smit, R. de Graaf, A. van Straten, M. ten Have, and P. Cuijpers. 2009. Incidence of social phobia and identification of its risk indicators: A model for prevention. Acta Psychiatrica Scandinavica 119(1):62–70.
Achim, A. M., M. Maziade, E. Raymond, D. Olivier, C. Merette, and M. A. Roy. 2011. How prevalent are anxiety disorders in schizophrenia? A meta-analysis and critical review on a significant association. Schizophrenia Bulletin 37(4):811–821.
Adler, D. A., D. Lerner, Z. L. Visco, A. Greenhill, H. Chang, E. Cymerman, F. Azocar, and W. H. Rogers. 2015. Improving work outcomes of dysthymia (persistent depressive disorder) in an employed population. General Hospital Psychiatry 37(4):352–359.
Alonso, P., D. Cuadras, L. Gabriels, D. Denys, W. Goodman, B. D. Greenberg, F. Jimenez-Ponce, J. Kuhn, D. Lenartz, L. Mallet, B. Nuttin, E. Real, C. Segalas, R. Schuurman, S. T. du Montcel, and J. M. Menchon. 2015. Deep brain stimulation for obsessive–compulsive disorder: A meta-analysis of treatment outcome and predictors of response. PLoS One 10(7):e0133591.
Amerio, A., A. Odone, C. Marchesi, and S. N. Ghaemi. 2014. Treatment of comorbid bipolar disorder and obsessive–compulsive disorder: A systematic review. Journal of Affective Disorders 166:258–263.
Andrews, G., M. Davies, and N. Titov. 2011. Effectiveness randomized controlled trial of face to face versus internet cognitive behaviour therapy for social phobia. Australian and New Zealand Journal of Psychiatry 45(4):337–340.
Andrews, G., C. Bell, P. Boyce, C. Gale, L. Lampe, O. Marwat, R. Rapee, and G. Wilkins. 2018. Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for the treatment of panic disorder, social anxiety disorder, and generalised anxiety disorder. Australian and New Zealand Journal of Psychiatry 52(12):1109–1172.
APA (American Psychiatric Association). 2000. Diagnostic and statistical manual of mental disorders, 4th ed., text revision (DSM-IV-TR). Washington, DC: American Psychiatric Publishing.
APA. 2010. Practice guideline for the treatment of patients with panic disorders. Washington, DC: American Psychiatric Publishing.
APA. 2013. Diagnostic and statistical manual of mental disorders, 5th ed. (DSM-5). Washington, DC: American Psychiatric Publishing.
APA. 2017. Clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. Washington, DC: American Psychiatric Publishing.
Asnaani, A., A. N. Kaczkurkin, E. Alpert, C. P. McLean, H. B. Simpson, and E. B. Foa. 2017. The effect of treatment on quality of life and functioning in OCD. Comprehensive Psychiatry 73:7–14.
Bagby, R. M., A. G. Ryder, D. R. Schuller, and M. B. Marshall. 2004. The Hamilton Depression Rating Scale: Has the gold standard become a lead weight? American Journal of Psychiatry 161(12):2163–2177.
Baker, S. L., N. Heinrichs, H. J. Kim, and S. G. Hofmann. 2002. The Liebowitz Social Anxiety Scale as a self-report instrument: A preliminary psychometric analysis. Behaviour Research and Therapy 40(6):701–715.
Baldessarini, R. J., P. Salvatore, H. M. Khalsa, P. Gebre-Medhin, H. Imaz, A. Gonzalez-Pinto, J. Perez, N. Cruz, C. Maggini, and M. Tohen. 2010. Morbidity in 303 first-episode bipolar I disorder patients. Bipolar Disorders 12(3):264–270.
Baldwin, D. S., I. M. Anderson, D. J. Nutt, C. Allgulander, B. Bandelow, J. A. den Boer, D. M. Christmas, S. Davies, N. Fineberg, N. Lidbetter, A. Malizia, P. McCrone, D. Nabarro, C. O’Neill, J. Scott, N. van der Wee, and H. U. Wittchen. 2014. Evidence-based pharmacologic treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: A revision of the 2005 guidelines from the British Association for Psychopharmacology. Journal of Psychopharmacology 28(5):403–439.
Bandelow, B., U. Seidler-Brandler, A. Becker, D. Wedekind, and E. Ruther. 2007. Meta-analysis of randomized controlled comparisons of psychopharmacologic and psychologic treatments for anxiety disorders. World Journal of Biological Psychiatry 8(3):175–187.
Bandelow, B., J. Zohar, E. Hollander, S. Kasper, H. J. Moller, J. Zohar, E. Hollander, S. Kasper, H. J. Moller, B. Bandelow, C. Allgulander, J. Ayuso-Gutierrez, D. S. Baldwin, R. Buenvicius, G. Cassano, N. Fineberg, L. Gabriels, I. Hindmarch, H. Kaiya, D. F. Klein, M. Lader, Y. Lecrubier, J. P. Lepine, M. R. Liebowitz, J. J. Lopez-Ibor, D. Marazziti, E. C. Miguel, K. S. Oh, M. Preter, R. Rupprecht, M. Sato, V. Starcevic, D. J. Stein, M. van Ameringen, and J. Vega. 2008. World Federation of Societies of Biological Psychiatry (WDSBP) guidelines for the pharmacologic treatment of anxiety, obsessive–compulsive and post-traumatic stress disorders—First revision. World Journal of Biological Psychiatry 9(4):248–312.
Bandelow, B., L. Sher, R. Bunevicius, E. Hollander, S. Kasper, J. Zohar, and H. J. Moller. 2012. Guidelines for the pharmacologic treatment of anxiety disorders, obsessive–compulsive disorder, and posttraumatic stress disorder in primary care. International Journal of Psychiatry in Clinical Practice 16(2):77–84.
Barkowski, S., D. Schwartze, B. Strauss, G. M. Burlingame, J. Barth, and J. Rosendahl. 2016. Efficacy of group psychotherapy for social anxiety disorder: A meta-analysis of randomized controlled trials. Journal of Anxiety Disorders 39:44–64.
Batelaan, N. M., R. de Graaf, B. W. Penninx, A. J. van Balkom, W. A. Vollebergh, and A. T. Beekman. 2010. The 2-year prognosis of panic episodes in the general population. Psychologic Medicine 40(1):147–157.
Bauer, M., and M. Gitlin. 2016. Natural course of bipolar disorder and implications for treatment. In M. Bauer and M. Gitlin (eds.), The essential guide to lithium treatment. Basel, Switzerland: Springer International Publishing. Pp. 11–23.
Baumeister, H., A. Knecht, and N. Hutter. 2012. Direct and indirect costs in persons with chronic back pain and comorbid mental disorders—A systematic review. Journal of Psychosomatic Research 73(2):79–85.
Baune, B. T., and G. S. Malhi. 2015. A review on the impact of cognitive dysfunction on social, occupational, and general functional outcomes in bipolar disorder. Bipolar Disorders 17(Suppl 2):41–55.
Beesdo, K., A. Bittner, D. S. Pine, M. B. Stein, M. Hofler, R. Lieb, and H. U. Wittchen. 2007. Incidence of social anxiety disorder and the consistent risk for secondary depression in the first three decades of life. Archives of General Psychiatry 64(8):903–912.
Bergeron, R., A. V. Ravindran, Y. Chaput, E. Goldner, R. Swinson, M. A. van Ameringen, C. Austin, and V. Hadrava. 2002. Sertraline and fluoxetine treatment of obsessive–compulsive disorder: Results of a double-blind, 6-month treatment study. Journal of Clinical Psychopharmacology 22(2):148–154.
Bernardy, N. C., and M. J. Friedman. 2017. Pharmacological management of posttraumatic stress disorder. Current Opinion in Psychology 14:116–121.
Bhalla, R. K., M. A. Butters, B. H. Mulsant, A. E. Begley, M. D. Zmuda, B. Schoderbek, B. G. Pollock, C. F. Reynolds, 3rd, and J. T. Becker. 2006. Persistence of neuropsychologic deficits in the remitted state of late-life depression. American Journal of Geriatric Psychiatry 14(5):419–427.
Bienvenu, O. J., C. U. Onyike, M. B. Stein, L. S. Chen, J. Samuels, G. Nestadt, and W. W. Eaton. 2006. Agoraphobia in adults: Incidence and longitudinal relationship with panic. British Journal of Psychiatry 188:432–438.
Blanco, C., L. B. Bragdon, F. R. Schneier, and M. R. Liebowitz. 2013. The evidence-based pharmacotherapy of social anxiety disorder. International Journal of Neuropsychopharmacology 16(1):235–249.
Bögels, S. M., L. Alden, D. C. Beidel, L. A. Clark, D. S. Pine, M. B. Stein, and M. Voncken. 2010. Social anxiety disorder: Questions and answers for the DSM-5. Depression and Anxiety 27(2):168–189.
Bonnin, C. D. M., M. Reinares, A. Martinez-Aran, E. Jimenez, J. Sanchez-Moreno, B. Sole, L. Montejo, and E. Vieta. 2019. Improving functioning, quality of life, and well-being in patients with bipolar disorder. International Journal of Neuropsychopharmacology 22(8):467–477.
Boscarino, J. A. 2006. Posttraumatic stress disorder and mortality among U.S. Army veterans 30 years after military service. Annals of Epidemiology 16(4):248–256.
Braga, D. T., G. G. Manfro, K. Niederauer, and A. V. Cordioli. 2010. Full remission and relapse of obsessive–compulsive symptoms after cognitive–behavioral group therapy: A two-year follow-up. Brazilian Journal of Psychiatry 32(2):164–168.
Brown, L. T., C. B. Mikell, B. E. Youngerman, Y. Zhang, G. M. McKhann, II, and S. A. Sheth. 2016. Dorsal anterior cingulotomy and anterior capsulotomy for severe, refractory obsessive-compulsive disorder: A systematic review of observational studies. Journal of Neurosurgery 124(1):77–89.
Brown, T. A., and D. H. Barlow. 2014. Anxiety and related disorders interview schedule for DSM-5 (ADIS-5)—Adult and lifetime version: Clinician manual. New York: Oxford University Press.
Brown, T. A., L. A. Campbell, C. L. Lehman, J. R. Grisham, and R. B. Mancill. 2001. Current and lifetime comorbidity of the DSM-IV anxiety and mood disorders in a large clinical sample. Journal of Abnormal Psychology 110(4):585–599.
Bruce, S. E., K. A. Yonkers, M. W. Otto, J. L. Eisen, R. B. Weisberg, M. Pagano, M. T. Shea, and M. B. Keller. 2005. Influence of psychiatric comorbidity on recovery and recurrence in generalized anxiety disorder, social phobia, and panic disorder: A 12-year prospective study. American Journal of Psychiatry 162(6):1179–1187.
Bruce, T. J., D. A. Spiegel, and M. T. Hegel. 1999. Cognitive–behavioral therapy helps prevent relapse and recurrence of panic disorder following alprazolam discontinuation: A long-term follow-up of the Peoria and Dartmouth studies. Journal of Consulting and Clinical Psychology 67(1):151–156.
Buist-Bouwman, M. A., R. De Graaf, W. A. M. Vollebergh, and J. Ormel. 2005. Comorbidity of physical and mental disorders and the effect on work-loss days. Acta Psychiatrica Scandinavica 111(6):436–443.
Burdick, K. E., T. E. Goldberg, B. A. Cornblatt, R. S. Keefe, C. B. Gopin, P. Derosse, R. J. Braga, and A. K. Malhotra. 2011. The MATRICS consensus cognitive battery in patients with bipolar I disorder. Neuropsychopharmacology 36(8):1587–1592.
Burgel, P. R., R. Escamilla, T. Perez, P. Carre, D. Caillaud, P. Chanez, C. Pinet, G. Jebrak, G. Brinchault, I. Court-Fortune, J. L. Paillasseur, and N. Roche. 2013. Impact of comorbidities on COPD-specific health-related quality of life. Respiratory Medicine 107(2):233–241.
Butler, A. C., J. E. Chapman, E. M. Forman, and A. T. Beck. 2006. The empirical status of cognitive–behavioral therapy: A review of meta-analyses. Clinical Psychology Review 26(1):17–31.
Byers, A. L., K. Yaffe, K. E. Covinsky, M. B. Friedman, and M. L. Bruce. 2010. High occurrence of mood and anxiety disorders among older adults: The National Comorbidity Survey Replication. Archives of General Psychiatry 67(5):489–496.
CANMAT (Canadian Network for Mood and Anxiety Treatments). 2016. CANMAT 2016 clinical guidelines for the management of adults with major depressive disorder. Canadian Journal of Psychiatry 61(9):504–603.
Canton, J., K. M. Scott, and P. Glue. 2012. Optimal treatment of social phobia: Systematic review and meta-analysis. Neuropsychiatric Disease and Treatment 8:203–215.
Carvalho, A. F., D. Dimellis, X. Gonda, E. Vieta, R. S. McIntyre, and K. N. Fountoulakis. 2014. Rapid cycling in bipolar disorder: A systematic review. Journal of Clinical Psychiatry 75(6):e578–e586.
CDC (Centers for Disease Control and Prevention). 2018. Learn about mental health. https://www.cdc.gov/mentalhealth/learn/index.htm (accessed April 12, 2019).
Cerimele, J. M., S. B. Goldberg, C. J. Miller, S. W. Gabrielson, and J. C. Fortney. 2019. Systematic review of symptom assessment measures for use in measurement-based care of bipolar disorders. Psychiatric Services 70(5):396–408.
Comer, J. S., C. Blanco, D. S. Hasin, S. M. Liu, B. F. Grant, J. B. Turner, and M. Olfson. 2011. Health-related quality of life across the anxiety disorders: Results from the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC). Journal of Clinical Psychiatry 72(1):43–50.
Conradi, H. J., J. Ormel, and P. de Jonge. 2011. Presence of individual (residual) symptoms during depressive episodes and periods of remission: A 3-year prospective study. Psychological Medicine 41(6):1165–1174.
Costa-Black, K. M., P. Loisel, J. R. Anema, and G. Pransky. 2010. Back pain and work. Best Practice & Research: Clinical Rheumatology 24(2):227–240.
Covin, R., A. J. Ouimet, P. M. Seeds, and D. J. Dozois. 2008. A meta-analysis of CBT for pathological worry among clients with GAD. Journal of Anxiety Disorders 22(1):108–116.
Cuijpers, P., J. Dekker, S. D. Hollon, and G. Andersson. 2009a. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: A meta-analysis. Journal of Clinical Psychiatry 70(9):1219–1229.
Cuijpers, P., A. van Straten, L. Warmerdam, and G. Andersson. 2009b. Psychotherapy versus the combination of psychotherapy and pharmacotherapy in the treatment of depression: A meta-analysis. Depression and Anxiety 26(3):279–288.
Cuijpers, P., E. Karyotaki, E. Weitz, G. Andersson, S. D. Hollon, and A. van Straten. 2014a. The effects of psychotherapies for major depression in adults on remission, recovery and improvement: A meta-analysis. Journal of Affective Disorders 159:118–126.
Cuijpers, P., M. Sijbrandij, S. Koole, M. Huibers, M. Berking, and G. Andersson. 2014b. Psychologic treatment of generalized anxiety disorder: A meta-analysis. Clinical Psychology Review 34(2):130–140.
Davidson, J. R., C. M. Miner, J. De Veaugh-Geiss, L. A. Tupler, J. T. Colket, and N. L. Potts. 1997. The Brief Social Phobia Scale: A psychometric evaluation. Psychologic Medicine 27(1):161–166.
Davidson, J., B. O. Rothbaum, P. Tucker, G. Asnis, I. Benattia, and J. J. Musgnung. 2006. Venlafaxine extended release in posttraumatic stress disorder: A sertraline- and placebo-controlled study. Journal of Clinical Psychopharmacology 26(3):259–267.
Davis, M. L., J. A. Smits, and S. G. Hofmann. 2014. Update on the efficacy of pharmacotherapy for social anxiety disorder: A meta-analysis. Expert Opinion on Pharmacotherapy 15(16):2281–2291.
Deschenes, S. S., R. J. Burns, and N. Schmitz. 2015. Associations between depression, chronic physical health conditions, and disability in a community sample: A focus on the persistence of depression. Journal of Affective Disorders 179:6–13.
Dèttore, D., A. Pozza, and G. Andersson. 2015. Efficacy of technology-delivered cognitive behavioural therapy for OCD versus control conditions, and in comparison with therapist-administered CBT: Meta-analysis of randomized controlled trials. Cognitive Behaviour Therapy 44(3):190–211.
Dickinson, T., R. Becerra, and J. Coombes. 2017. Executive functioning deficits among adults with bipolar disorder (types I and II): A systematic review and meta-analysis. Journal of Affective Disorders 218:407–427.
Dold, M., M. Aigner, R. Lanzenberger, and S. Kasper. 2013. Antipsychotic augmentation of serotonin reuptake inhibitors in treatment-resistant obsessive–compulsive disorder: A meta-analysis of double-blind, randomized, placebo-controlled trials. International Journal of Neuropsychopharmacology 16(3):557–574.
Donovan, M. R., P. Glue, S. Kolluri, and B. Emir. 2010. Comparative efficacy of antidepressants in preventing relapse in anxiety disorders—A meta-analysis. Journal of Affective Disorders 123(1–3):9–16.
Dow, M. G., J. A. Kenardy, D. W. Johnston, M. G. Newman, C. B. Taylor, and A. Thomson. 2007. Prognostic indices with brief and standard CBT for panic disorder: II. Moderators of outcome. Psychologic Medicine 37(10):1503–1509.
Drake, R. E., W. Frey, G. R. Bond, H. H. Goldman, D. Salkever, A. Miller, T. A. Moore, J. Riley, M. Karakus, and R. Milfort. 2013. Assisting Social Security Disability Insurance beneficiaries with schizophrenia, bipolar disorder, or major depression in returning to work. American Journal of Psychiatry 170(12):1433–1441.
Eskildsen, A., E. Hougaard, and N. K. Rosenberg. 2010. Pre-treatment patient variables as predictors of drop-out and treatment outcome in cognitive behavioural therapy for social phobia: A systematic review. Nordic Journal of Psychiatry 64(2):94–105.
Faller, H., E. Brahler, M. Harter, M. Keller, H. Schulz, K. Wegscheider, J. Weis, A. Boehncke, M. Richard, S. Sehner, U. Koch, and A. Mehnert. 2015. Performance status and depressive symptoms as predictors of quality of life in cancer patients. A structural equation modeling analysis. Psycho-Oncology 24(11):1456–1462.
Fang, S. C., P. P. Schnurr, A. L. Kulish, D. W. Holowka, B. P. Marx, T. M. Keane, and R. Rosen. 2015. Psychosocial functioning and health-related quality of life associated with posttraumatic stress disorder in male and female Iraq and Afghanistan war veterans: The valor registry. Journal of Women’s Health (Larchmont) 24(12):1038–1046.
Fineberg, N. A., B. Tonnoir, O. Lemming, and D. J. Stein. 2007. Escitalopram prevents relapse of obsessive–compulsive disorder. European Neuropsychopharmacology 17(6–7):430–439.
Fishbain, D. A., A. Pulikal, J. E. Lewis, and J. Gao. 2017. Chronic pain types differ in their reported prevalence of post -traumatic stress disorder (PTSD) and there is consistent evidence that chronic pain is associated with PTSD: An evidence-based structured systematic review. Pain Medicine (Malden, Mass.) 18(4):711–735.
Flament, M. F., A. Whitaker, J. L. Rapoport, M. Davies, C. Z. Berg, K. Kalikow, W. Sceery, and D. Shaffer. 1988. Obsessive compulsive disorder in adolescence: An epidemiological study. Journal of the American Academy of Child and Adolescent Psychiatry 27:764–771.
Foa, E. B., M. R. Liebowitz, M. J. Kozak, S. Davies, R. Campeas, M. E. Franklin, J. D. Huppert, K. Kjernisted, V. Rowan, A. B. Schmidt, H. B. Simpson, and X. Tu. 2005. Randomized, placebo-controlled trial of exposure and ritual prevention, clomipramine, and their combination in the treatment of obsessive–compulsive disorder. American Journal of Psychiatry 162(1):151–161.
Forman-Hoffman, V., J. C. Middleton, C. Feltner, B. N. Gaynes, R. P. Weber, C. Bann, M. Viswanathan, K. N. Lohr, C. Baker, and J. Green. 2018. Psychologic and pharmacologic treatments for adults with posttraumatic stress disorder: A systematic review update. AHRQ publication no. 18-ECH011-EF. Rockville, MD: Agency for Healthcare Research and Quality.
Frank, E., R. F. Prien, R. B. Jarrett, M. B. Keller, D. J. Kupfer, P. W. Lavori, A. J. Rush, and M. M. Weissman. 1991. Conceptualization and rationale for consensus definitions of terms in major depressive disorder. Remission, recovery, relapse, and recurrence. Archives of General Psychiatry 48(9):851–855.
Fresco, D. M., M. E. Coles, R. G. Heimberg, M. R. Liebowitz, S. Hami, M. B. Stein, and D. Goetz. 2001. The Liebowitz Social Anxiety Scale: A comparison of the psychometric properties of self-report and clinician-administered formats. Psychological Medicine 31(6):1025–1035.
Friborg, O., M. Martinussen, S. Kaiser, K. T. Overgard, and J. H. Rosenvinge. 2013. Comorbidity of personality disorders in anxiety disorders: A meta-analysis of 30 years of research. Journal of Affective Disorders 145(2):143–155.
Friedman, M. J., D. G. Kilpatrick, P. P. Schnurr, and F. W. Weathers. 2016. Correcting misconceptions about the diagnostic criteria for posttraumatic stress disorder in DSM-5. JAMA Psychiatry 73(7):753–754.
Furukawa, T. A. 2010. Assessment of mood: Guides for clinicians. Journal of Psychosomatic Research 68(6):581–589.
Gilbody, S., D. Richards, S. Brealey, and C. Hewitt. 2007. Screening for depression in medical settings with the Patient Health Questionnaire (PHQ): A diagnostic meta-analysis. Journal of General Internal Medicine 22(11):1596–1602.
Gitlin, M. J., and D. J. Miklowitz. 2017. The difficult lives of individuals with bipolar disorder: A review of functional outcomes and their implications for treatment. Journal of Affective Disorders 209:147–154.
Goldberg, J., K. M. Magruder, C. W. Forsberg, L. E. Kazis, T. B. Ustun, M. J. Friedman, B. T. Litz, V. Vaccarino, P. J. Heagerty, T. C. Gleason, G. D. Huang, and N. L. Smith. 2014. The association of PTSD with physical and mental health functioning and disability (VA cooperative study #569: The course and consequences of posttraumatic stress disorder in Vietnam-era veteran twins). Quality of Life Research 23(5):1579–1591.
Goldstein, B. I., P. R. Houck, and J. F. Karp. 2009. Factors associated with pain interference in an epidemiologic sample of adults with bipolar i disorder. Journal of Affective Disorders 117(3):151–156.
Goncalves, D. C., and G. J. Byrne. 2012. Interventions for generalized anxiety disorder in older adults: Systematic review and meta-analysis. Journal of Anxiety Disorders 26(1):1–11.
Gosselin, P., R. Ladouceur, C. M. Morin, M. J. Dugas, and L. Baillargeon. 2006. Benzodiazepine discontinuation among adults with GAD: A randomized trial of cognitive–behavioral therapy. Journal of Consulting and Clinical Psychology 74(5):908–919.
Gradus, J. L., S. Antonsen, E. Svensson, T. L. Lash, P. A. Resick, and J. G. Hansen. 2015a. Trauma, comorbidity, and mortality following diagnoses of severe stress and adjustment disorders: A nationwide cohort study. American Journal of Epidemiology 182(5):451–458.
Gradus, J. L., D. K. Farkas, E. Svensson, V. Ehrenstein, T. L. Lash, A. Milstein, N. Adler, and H. T. Sorensen. 2015b. Associations between stress disorders and cardiovascular disease events in the Danish population. BMJ Open 5(12):e009334.
Gradus, J. L., D. K. Farkas, E. Svensson, V. Ehrenstein, T. L. Lash, and H. Toft Sorensen. 2017. Posttraumatic stress disorder and gastrointestinal disorders in the Danish population. Epidemiology 28(3):354–360.
Grande, I., M. Berk, B. Birmaher, and E. Vieta. 2016. Bipolar disorder. Lancet 387(10027): 1561–1572.
Grant, B. F., D. S. Hasin, C. Blanco, F. S. Stinson, S. P. Chou, R. B. Goldstein, D. A. Dawson, S. Smith, T. D. Saha, and B. Huang. 2005. The epidemiology of social anxiety disorder in the United States: Results from the National Epidemiologic Survey on Alcohol and Related Conditions. Journal of Clinical Psychiatry 66(11):1351–1361.
Grant, B. F., D. S. Hasin, F. S. Stinson, D. A. Dawson, R. B. Goldstein, S. Smith, B. Huang, and T. D. Saha. 2006. The epidemiology of DSM-IV panic disorder and agoraphobia in the United States: Results from the National Epidemiologic Survey on Alcohol and Related Conditions. Journal of Clinical Psychiatry 67(3):363–374.
Grant, J. E. 2014. Clinical practice: Obsessive–compulsive disorder. New England Journal of Medicine 371(7):646–653.
Gureje, O., M. Von Korff, G. E. Simon, and R. Gater. 1998. Persistent pain and well-being: A World Health Organization study in primary care. JAMA 280(2):147–151.
Guy, W. 1976. ECDEU assessment manual for psychopharmacology. Rockville, MD: U.S. Department of Health, Education, and Welfare, Public Health Service, Alcohol, Drug Abuse, and Mental Health Administration, National Institute of Mental Health, Psychopharmacology Research Branch, Division of Extramural Research Programs.
Hamilton, M. 1959. The assessment of anxiety states by rating. British Journal of Medical Psychology 32(1):50–55.
Hamilton, M. 1967. Development of a rating scale for primary depressive illness. British Journal of Social and Clinical Psychology 6(4):278–296.
Haug, T. T., S. Blomhoff, K. Hellstrøm, I. Holme, M. Humble, H. P. Madsbu, and J. E. Wold. 2003. Exposure therapy and sertraline in social phobia: 1-year follow-up of a randomised controlled trial. British Journal of Psychiatry 182(APR.):312–318.
Hedman, E., G. Andersson, B. Ljotsson, E. Andersson, C. Ruck, E. Mortberg, and N. Lindefors. 2011. Internet-based cognitive behavior therapy vs. cognitive behavioral group therapy for social anxiety disorder: A randomized controlled non-inferiority trial. PLoS One 6(3):e18001.
Hellstrom, L., P. Bech, C. Hjorthoj, M. Nordentoft, J. Lindschou, and L. F. Eplov. 2017. Effect on return to work or education of individual placement and support modified for people with mood and anxiety disorders: Results of a randomised clinical trial. Occupational and Environmental Medicine 74(10):717–725.
Heyman, I., E. Fombonne, H. Simmons, T. Ford, H. Meltzer, and R. Goodman. 2001. Prevalence of obsessive–compulsive disorder in the British nationwide survey of child mental health. British Journal of Psychiatry 179:324–329.
Hirshfeld-Becker, D. R., J. Micco, A. Henin, A. Bloomfield, J. Biederman, and J. Rosenbaum. 2008. Behavioral inhibition. Depression and Anxiety 25(4):357–367.
Hofmann, S. G. 2004. Cognitive mediation of treatment change in social phobia. Journal of Consulting and Clinical Psychology 72(3):392–399.
Hofmann, S. G. 2014. D-cycloserine for treating anxiety disorders: Making good exposures better and bad exposures worse. Depression and Anxiety 31(3):175–177.
Hofmann, S. G., A. T. Sawyer, K. J. Korte, and J. A. Smits. 2009. Is it beneficial to add pharmacotherapy to cognitive–behavioral therapy when treating anxiety disorders? A meta-analytic review. International Journal of Cognitive Therapy 2(2):160–175.
Hoge, C. W., A. Terhakopian, C. A. Castro, S. C. Messer, and C. C. Engel. 2007. Association of posttraumatic stress disorder with somatic symptoms, health care visits, and absenteeism among Iraq war veterans. American Journal of Psychiatry 164(1):150–153.
Hoge, C. W., R. Yehuda, C. A. Castro, A. C. McFarlane, E. Vermetten, R. Jetly, K. C. Koenen, N. Greenberg, A. Y. Shalev, S. A. Rauch, C. R. Marmar, and B. O. Rothbaum. 2016. Unintended consequences of changing the definition of posttraumatic stress disorder in DSM-5: Critique and call for action. JAMA Psychiatry 73(7):750–752.
Hollander, E., A. Allen, M. Steiner, D. E. Wheadon, R. Oakes, and D. B. Burnham. 2003. Acute and long-term treatment and prevention of relapse of obsessive–compulsive disorder with paroxetine. Journal of Clinical Psychiatry 64(9):1113–1121.
Hoskins, M., J. Pearce, A. Bethell, L. Dankova, C. Barbui, W. A. Tol, M. van Ommeren, J. de Jong, S. Seedat, H. Chen, and J. I. Bisson. 2015. Pharmacotherapy for post-traumatic stress disorder: Systematic review and meta-analysis. British Journal of Psychiatry 206(2):93–100.
Huang, A. X., K. Delucchi, L. B. Dunn, and J. C. Nelson. 2015. A systematic review and meta-analysis of psychotherapy for late-life depression. American Journal of Geriatric Psychiatry 23(3):261–273.
Iancu, S. C., N. M. Batelaan, M. B. Zweekhorst, J. F. Bunders, D. J. Veltman, B. W. Penninx, and A. J. van Balkom. 2014. Trajectories of functioning after remission from anxiety disorders: 2-year course and outcome predictors. Psychologic Medicine 44(3):593–605.
Ijaz, S., P. Davies, C. J. Williams, D. Kessler, G. Lewis, and N. Wiles. 2018. Psychological therapies for treatment-resistant depression in adults. Cochrane Database of Systematic Reviews 2018(5):CD010558.
Ikebuchi, E., S. Sato, S. Yamaguchi, M. Shimodaira, A. Taneda, N. Hatsuse, Y. Watanabe, M. Sakata, N. Satake, M. Nishio, and J. I. Ito. 2017. Does improvement of cognitive functioning by cognitive remediation therapy effect work outcomes in severe mental illness? A secondary analysis of a randomized controlled trial. Psychiatry and Clinical Neurosciences 71(5):301–308.
Imai, H., A. Tajika, P. Chen, A. Pompoli, and T. A. Furukawa. 2016. Psychologic therapies versus pharmacologic interventions for panic disorder with or without agoraphobia in adults. Cochrane Database of Systematic Reviews 2016(10):CD011170.
Ipser, J. C., C. M. Kariuki, and D. J. Stein. 2008. Pharmacotherapy for social anxiety disorder: A systematic review. Expert Review of Neurotherapeutics 8(2):235–257.
Janardhan Reddy, Y. C., A. S. Sundar, J. C. Narayanaswamy, and S. B. Math. 2017. Clinical practice guidelines for obsessive–compulsive disorder. Indian Journal of Psychiatry 59(Suppl 1):S74–S90.
Jaurrieta, N., S. Jimenez-Murcia, P. Alonso, R. Granero, C. Segalas, J. Labad, and J. M. Menchon. 2008. Individual versus group cognitive behavioral treatment for obsessive–compulsive disorder: Follow up. Psychiatry and Clinical Neurosciences 62(6):697–704.
Judd, L. L., M. J. Paulus, P. J. Schettler, H. S. Akiskal, J. Endicott, A. C. Leon, J. D. Maser, T. Mueller, D. A. Solomon, and M. B. Keller. 2000. Does incomplete recovery from first lifetime major depressive episode herald a chronic course of illness? The American Journal of Psychiatry 157(9):1501–1504.
Judd, L. L., H. S. Akiskal, P. J. Schettler, J. Endicott, J. Maser, D. A. Solomon, A. C. Leon, J. A. Rice, and M. B. Keller. 2002. The long-term natural history of the weekly symptomatic status of bipolar I disorder. Archives of General Psychiatry 59(6):530–537.
Judd, L. L., H. S. Akiskal, P. J. Schettler, W. Coryell, J. Endicott, J. D. Maser, D. A. Solomon, A. C. Leon, and M. B. Keller. 2003. A prospective investigation of the natural history of the long-term weekly symptomatic status of bipolar II disorder. Archives of General Psychiatry 60(3):261–269.
Kapczinski, F., M. S. Lima, J. S. Souza, and R. Schmitt. 2003. Antidepressants for generalized anxiety disorder. Cochrane Database of Systematic Reviews 2003(2):CD003592.
Karno, M., J. M. Golding, S. B. Sorenson, and M. A. Burnam. 1988. The epidemiology of 20 obsessive–compulsive disorder in five U.S. communities. Archives of General Psychiatry 45:1094–1099.
Katschnig, H., M. Amering, J. M. Stolk, and J. C. Ballenger. 1996. Predictors of quality of life in a long-term followup study in panic disorder patients after a clinical drug trial. Psychopharmacology Bulletin 32(1):149–155.
Katzman, M. A., P. Bleau, P. Blier, P. Chokka, K. Kjernisted, M. Van Ameringen, M. M. Antony, S. Bouchard, A. Brunet, M. Flament, S. Grigoriadis, S. Mendlowitz, K. O’Connor, K. Rabheru, P. M. Richter, M. Robichaud, and J. R. Walker. 2014. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive–compulsive disorders. BMC Psychiatry 14(Suppl 1):S1.
Kessing, L. V., and P. K. Andersen. 2017. Evidence for clinical progression of unipolar and bipolar disorders. Acta Psychiatrica Scandinavica 135(1):51–64.
Kessler, R. C. 2000. Posttraumatic stress disorder: The burden to the individual and to society. Journal of Clinical Psychiatry 61(Suppl 5):4–12; discussion 13–14.
Kessler, R. C., K. A. McGonagle, S. Zhao, C. B. Nelson, M. Hughes, S. Eshleman, H. U. Wittchen, and K. S. Kendler. 1994. Lifetime and 12-month prevalence of DSM-III-R psychiatric disorders in the United States. Results from the National Comorbidity Survey. Archives of General Psychiatry 51(1):8–19.
Kessler, R. C., A. Sonnega, E. Bromet, M. Hughes, and C. B. Nelson. 1995. Posttraumatic stress disorder in the National Comorbidity Survey. Archives of General Psychiatry 52(12):1048–1060.
Kessler, R. C., G. Borges, and E. E. Walters. 1999a. Prevalence of and risk factors for lifetime suicide attempts in the National Comorbidity Survey. Archives of General Psychiatry 56(7):617–626.
Kessler, R. C., R. L. DuPont, P. Berglund, and H. U. Wittchen. 1999b. Impairment in pure and comorbid generalized anxiety disorder and major depression at 12 months in two national surveys. American Journal of Psychiatry 156(12):1915–1923.
Kessler, R. C., P. Berglund, O. Demler, R. Jin, D. Koretz, K. R. Merikangas, A. J. Rush, E. E. Walters, and P. S. Wang. 2003. The epidemiology of major depressive disorder: Results from the national comorbidity survey replication (NCS-R). JAMA 289(23):3095–3105.
Kessler, R. C., W. T. Chiu, O. Demler, K. R. Merikangas, and E. E. Walters. 2005a. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Archives of General Psychiatry 62(6):617–627.
Kessler, R. C., P. Berglund, O. Demler, R. Jin, K. R. Merikangas, and E. E. Walters. 2005b. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Archives of General Psychiatry 62(6):593–602.
Kessler, R. C., W. T. Chiu, R. Jin, A. M. Ruscio, K. Shear, and E. E. Walters. 2006. The epidemiology of panic attacks, panic disorder, and agoraphobia in the National Comorbidity Survey Replication. Archives of General Psychiatry 63(4):415–424.
Kishita, N., and K. Laidlaw. 2017. Cognitive behaviour therapy for generalized anxiety disorder: Is CBT equally efficacious in adults of working age and older adults? Clinical Psychology Review 52:124–136.
Koran, L. M., E. Hackett, A. Rubin, R. Wolkow, and D. Robinson. 2002. Efficacy of sertraline in the long-term treatment of obsessive–compulsive disorder. American Journal of Psychiatry 159(1):88–95.
Koran, L. M., N. N. Gamel, H. W. Choung, E. H. Smith, and E. N. Aboujaoude. 2005. Mirtazapine for obsessive–compulsive disorder: An open trial followed by double-blind discontinuation. Journal of Clinical Psychiatry 66(4):515–520.
Koran, L. M., G. L. Hanna, E. Hollander, G. Nestadt, and H. B. Simpson. 2007. Practice guideline for the treatment of patients with obsessive–compulsive disorder. American Journal of Psychiatry 164(7 Suppl):5–53.
Koran, L. M., D. Bromberg, C. S. Hornfeldt, J. C. Shepski, S. Wang, and E. Hollander. 2010. Extended-release fluvoxamine and improvements in quality of life in patients with obsessive–compulsive disorder. Comprehensive Psychiatry 51(4):373–379.
Koszycki, D., M. Benger, J. Shlik, and J. Bradwejn. 2007. Randomized trial of a meditation-based stress reduction program and cognitive behavior therapy in generalized social anxiety disorder. Behaviour Research and Therapy 45(10):2518–2526.
Kroenke, K., R. L. Spitzer, J. B. Williams, P. O. Monahan, and B. Lowe. 2007. Anxiety disorders in primary care: Prevalence, impairment, comorbidity, and detection. Annals of Internal Medicine 146(5):317–325.
Kroenke, K., S. Outcalt, E. Krebs, M. J. Bair, J. Wu, N. Chumbler, and Z. Yu. 2013. Association between anxiety, health-related quality of life and functional impairment in primary care patients with chronic pain. General Hospital Psychiatry 35(4):359–365.
Kupfer, D. J. 1991. Recurrent depression: Challenges and solutions. Journal of Clinical Psychiatry 52:28–34.
Lee, D. J., C. W. Schnitzlein, J. P. Wolf, M. Vythilingam, A. M. Rasmusson, and C. W. Hoge. 2016. Psychotherapy versus pharmacotherapy for posttraumatic stress disorder: Systemic review and meta-analyses to determine first-line treatments. Depression and Anxiety 33(9):792–806.
Lee, R. S., D. F. Hermens, M. A. Porter, and M. A. Redoblado-Hodge. 2012. A meta-analysis of cognitive deficits in first-episode major depressive disorder. Journal of Affective Disorders 140(2):113–124.
Leichsenring, F., S. Salzer, U. Jaeger, H. Kachele, R. Kreische, F. Leweke, U. Ruger, C. Winkelbach, and E. Leibing. 2009. Short-term psychodynamic psychotherapy and cognitive–behavioral therapy in generalized anxiety disorder: A randomized, controlled trial. American Journal of Psychiatry 166(8):875–881.
Leichsenring, F., S. Salzer, M. E. Beutel, S. Herpertz, W. Hiller, J. Hoyer, J. Huesing, P. Joraschky, B. Nolting, K. Poehlmann, V. Ritter, U. Stangier, B. Strauss, N. Stuhldreher, S. Tefikow, T. Teismann, U. Willutzki, J. Wiltink, and E. Leibing. 2013. Psychodynamic therapy and cognitive–behavioral therapy in social anxiety disorder: A multicenter randomized controlled trial. American Journal of Psychiatry 170(7):759–767.
Lenze, E. J., B. H. Mulsant, J. Mohlman, M. K. Shear, M. A. Dew, R. Schulz, M. D. Miller, B. Tracey, and C. F. Reynolds, 3rd. 2005. Generalized anxiety disorder in late life: Lifetime course and comorbidity with major depressive disorder. American Journal of Geriatric Psychiatry 13(1):77–80.
Lerner, D., D. Adler, R. C. Hermann, H. Chang, E. J. Ludman, A. Greenhill, K. Perch, W. C. McPeck, and W. H. Rogers. 2012. Impact of a work-focused intervention on the productivity and symptoms of employees with depression. Journal of Occupational and Environmental Medicine 54(2):128–135.
Li, S., J. Tan, M. Qian, and X. Liu. 2008. Continual training of attentional bias in social anxiety. Behaviour Research and Therapy 46(8):905–912.
Liebowitz, M. R. 1987. Social phobia. Modern Problems of Pharmacopsychiatry 22:141–173.
Liebowitz, M. R., R. G. Heimberg, F. R. Schneier, D. A. Hope, S. Davies, C. S. Holt, D. Goetz, H. R. Juster, S. H. Lin, M. A. Bruch, R. D. Marshall, and D. F. Klein. 1999. Cognitive–behavioral group therapy versus phenelzine in social phobia: Long-term outcome. Depression and Anxiety 10(3):89–98.
Low, N. C., L. Cui, and K. R. Merikangas. 2008. Specificity of familial transmission of anxiety and comorbid disorders. Journal of Psychiatric Research 42(7):596–604.
Mackenzie, C. S., K. Reynolds, K. L. Chou, J. Pagura, and J. Sareen. 2011. Prevalence and correlates of generalized anxiety disorder in a national sample of older adults. American Journal of Geriatric Psychiatry 19(4):305–315.
Marshall, E. C., M. J. Zvolensky, N. Sachs-Ericsson, N. B. Schmidt, and A. Bernstein. 2008. Panic attacks and physical health problems in a representative sample: Singular and interactive associations with psychologic problems, and interpersonal and physical disability. Journal of Anxiety Disorders 22(1):78–87.
Mayo-Wilson, E., S. Dias, I. Mavranezouli, K. Kew, D. M. Clark, A. E. Ades, and S. Pilling. 2014. Psychological and pharmacological interventions for social anxiety disorder in adults: A systematic review and network meta-analysis. Lancet Psychiatry 1(5):368–376.
McKnight, P. E., and T. B. Kashdan. 2009. The importance of functional impairment to mental health outcomes: A case for reassessing our goals in depression treatment research. Clinical Psychology Review 29(3):243–259.
McKnight, P. E., S. S. Monfort, T. B. Kashdan, D. V. Blalock, and J. M. Calton. 2016. Anxiety symptoms and functional impairment: A systematic review of the correlation between the two measures. Clinical Psychology Review 45:115–130.
Mennin, D. S., D. M. Fresco, R. G. Heimberg, F. R. Schneier, S. O. Davies, and M. R. Liebowitz. 2002. Screening for social anxiety disorder in the clinical setting: Using the Liebowitz Social Anxiety Scale. Journal of Anxiety Disorders 16(6):661–673.
Merikangas, K. R., H. S. Akiskal, J. Angst, P. E. Greenberg, R. M. Hirschfeld, M. Petukhova, and R. C. Kessler. 2007. Lifetime and 12-month prevalence of bipolar spectrum disorder in the National Comorbidity Survey Replication. Archives of General Psychiatry 64(5):543–552.
Mintz, J., L. I. Mintz, M. J. Arruda, and S. S. Hwang. 1992. Treatments of depression and the functional capacity to work. Archives of General Psychiatry 49(10):761–768.
Mochcovitch, M. D., R. C. da Rocha Freire, R. F. Garcia, and A. E. Nardi. 2017. Can long-term pharmacotherapy prevent relapses in generalized anxiety disorder? A systematic review. Clinical Drug Investigation 37(8):737–743.
Modini, M., M. Abbott, and C. Hunt. 2015. A systematic review of the psychometric properties of trait social anxiety self-report measures. Journal of Psychopathology and Behavioral Assessment 37:645–662.
Montgomery, S. A., R. Nil, N. Durr-Pal, H. Loft, and J. P. Boulenger. 2005. A 24-week randomized, double-blind, placebo-controlled study of escitalopram for the prevention of generalized social anxiety disorder. Journal of Clinical Psychiatry 66(10):1270–1278.
Morina, N., J. M. Wicherts, J. Lobbrecht, and S. Priebe. 2014. Remission from post-traumatic stress disorder in adults: A systematic review and meta-analysis of long term outcome studies. Clinical Psychology Review 34(3):249–255.
Mörtberg, E., D. M. Clark, and S. Bejerot. 2011. Intensive group cognitive therapy and individual cognitive therapy for social phobia: Sustained improvement at 5-year follow-up. Journal of Anxiety Disorders 25(8):994–1000.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2019. Functional assessment for adults with disabilities. Washington, DC: The National Academies Press.
Nelson, E., and J. Rice. 1997. Stability of diagnosis of obsessive–compulsive disorder in the Epidemiologic Catchment Area Study. American Journal of Psychiatry 154(6):826–831.
NICE (National Institute for Health and Care Excellence). 2006. Obsessive–compulsive disorder: Core interventions in the treatment of obsessive–compulsive disorder and body dysmorphic disorder. London, UK: National Institute for Health and Care Excellence.
NICE. 2011. Generalised anxiety disorder and panic disorder in adults: Management. London, UK: National Institute for Health and Care Excellence.
NICE. 2013. Social anxiety disorder: Recognition, assessment and treatment. London, UK: National Institute for Health and Care Excellence.
Nierenberg, A. A., M. M. Husain, M. H. Trivedi, M. Fava, D. Warden, S. R. Wisniewski, S. Miyahara, and A. J. Rush. 2010. Residual symptoms after remission of major depressive disorder with citalopram and risk of relapse: A STAR*D report. Psychological Medicine 40(1):41–50.
Nieuwenhuijsen, K., B. Faber, J. H. Verbeek, A. Neumeyer-Gromen, H. L. Hees, A. C. Verhoeven, C. M. van der Feltz-Cornelis, and U. Bultmann. 2014. Interventions to improve return to work in depressed people. Cochrane Database of Systematic Reviews 2014(12):CD006237.
Noordik, E., J. J. van der Klink, E. F. Klingen, K. Nieuwenhuijsen, and F. J. van Dijk. 2010. Exposure-in-vivo containing interventions to improve work functioning of workers with anxiety disorder: A systematic review. BMC Public Health 10:598.
O’Donnell, M. L., M. Creamer, and P. Pattison. 2004. Posttraumatic stress disorder and depression following trauma: Understanding comorbidity. American Journal of Psychiatry 161(8):1390–1396.
Olthuis, J. V., M. C. Watt, K. Bailey, J. A. Hayden, and S. H. Stewart. 2016. Therapist-supported internet cognitive behavioural therapy for anxiety disorders in adults. Cochrane Database of Systematic Reviews 2016(3):CD011565.
Otto, M. W., R. K. McHugh, N. M. Simon, F. J. Farach, J. J. Worthington, and M. H. Pollack. 2010. Efficacy of CBT for benzodiazepine discontinuation in patients with panic disorder: Further evaluation. Behaviour Research and Therapy 48(8):720–727.
Pacella, M. L., B. Hruska, and D. L. Delahanty. 2013. The physical health consequences of PTSD and PTSD symptoms: A meta-analytic review. Journal of Anxiety Disorders 27(1):33–46.
Papadimitropoulou, K., C. Vossen, A. Karabis, C. Donatti, and N. Kubitz. 2017. Comparative efficacy and tolerability of pharmacologic and somatic interventions in adult patients with treatment-resistant depression: A systematic review and network meta-analysis. Current Medical Research and Opinion 33(4):701–711.
Parikh, S. V., Z. V. Segal, S. Grigoriadis, A. V. Ravindran, S. H. Kennedy, R. W. Lam, and S. B. Patten. 2009. Canadian Network for Mood and Anxiety Treatments (CANMAT) clinical guidelines for the management of major depressive disorder in adults. II. Psychotherapy alone or in combination with antidepressant medication. Journal of Affective Disorders 117(Suppl 1):S15–S25.
Paykel, E. S., R. Ramana, Z. Cooper, H. Hayhurst, J. Kerr, and A. Barocka. 1995. Residual symptoms after partial remission: An important outcome in depression. Psychological Medicine 25(6):1171–1180.
Perlman, K., D. Benrimoh, S. Israel, C. Rollins, E. Brown, J. F. Tunteng, R. You, E. You, M. Tanguay-Sela, E. Snook, M. Miresco, and M. T. Berlim. 2019. A systematic meta-review of predictors of antidepressant treatment outcome in major depressive disorder. Journal of Affective Disorders 243:503–515.
Pietrzak, R. H., R. B. Goldstein, S. M. Southwick, and B. F. Grant. 2011. Prevalence and axis I comorbidity of full and partial posttraumatic stress disorder in the United States: Results from wave 2 of the National Epidemiologic Survey on Alcohol and Related Conditions. Journal of Anxiety Disorders 25(3):456–465.
Pompoli, A., T. A. Furukawa, H. Imai, A. Tajika, O. Efthimiou, and G. Salanti. 2016. Psychologic therapies for panic disorder with or without agoraphobia in adults: A network meta-analysis. Cochrane Database of Systematic Reviews 2016(4):CD011004.
Porter, E., and D. L. Chambless. 2015. A systematic review of predictors and moderators of improvement in cognitive-behavioral therapy for panic disorder and agoraphobia. Clinical Psychology Review 42:179–192.
Porter, R. J., K. Douglas, J. Jordan, C. R. Bowie, J. Roiser, and G. S. Malhi. 2014. Psychologic treatments for cognitive dysfunction in major depressive disorder: Current evidence and perspectives. CNS & Neurological Disorders—Drug Targets 13(10):1677–1692.
Powers, M. B., S. R. Sigmarsson, and P. M. G. Emmelkamp. 2008. A meta-analytic review of psychologic treatments for social anxiety disorder. International Journal of Cognitive Therapy 1(2):94–113.
Rasmussen, S., E. Hackett, E. DuBoff, J. Greist, A. Halaris, L. M. Koran, M. Liebowitz, R. B. Lydiard, S. McElroy, J. Mendels, and K. O’Connor. 1997. A 2-year study of sertraline in the treatment of obsessive–compulsive disorder. International Clinical Psychopharmacology 12(6):309–316.
Raucher-Chene, D., A. M. Achim, A. Kaladjian, and C. Besche-Richard. 2017. Verbal fluency in bipolar disorders: A systematic review and meta-analysis. Journal of Affective Disorders 207:359–366.
Ravizza, L., G. Barzega, S. Bellino, F. Bogetto, and G. Maina. 1996. Drug treatment of obsessive–compulsive disorder (OCD): Long-term trial with clomipramine and selective serotonin reuptake inhibitors (SSRIs). Psychopharmacology Bulletin 32(1):167–173.
Reeves, G. M., K. J. Rohan, P. Langenberg, S. Snitker, and T. T. Postolache. 2012. Calibration of response and remission cut-points on the Beck Depression Inventory, second edition, for monitoring seasonal affective disorder treatment outcomes. Journal of Affective Disorders 138(1–2):123–127.
Renn, B. N., and P. A. Arean. 2017. Psychosocial treatment options for major depressive disorder in older adults. Current Treatment Options in Psychiatry 4(1):1–12.
Robinson, J., J. Sareen, B. J. Cox, and J. M. Bolton. 2011. Role of self-medication in the development of comorbid anxiety and substance use disorders: A longitudinal investigation. Archives of General Psychiatry 68(8):800–807.
Rocca, M. A., E. Pravata, P. Valsasina, M. Radaelli, B. Colombo, L. Vacchi, C. Gobbi, G. Comi, A. Falini, and M. Filippi. 2015. Hippocampal-DMN disconnectivity in MS is related to WM lesions and depression. Human Brain Mapping 36(12):5051–5063.
Rock, P. L., J. P. Roiser, W. J. Riedel, and A. D. Blackwell. 2014. Cognitive impairment in depression: A systematic review and meta-analysis. Psychologic Medicine 44(10):2029–2040.
Rodriguez, B. F., S. E. Bruce, M. E. Pagano, and M. B. Keller. 2005. Relationships among psychosocial functioning, diagnostic comorbidity, and the recurrence of generalized anxiety disorder, panic disorder, and major depression. Journal of Anxiety Disorders 19(7):752–766.
Roemer, L., S. M. Orsillo, and K. Salters-Pedneault. 2008. Efficacy of an acceptance-based behavior therapy for generalized anxiety disorder: Evaluation in a randomized controlled trial. Journal of Consulting and Clinical Psychology 76(6):1083–1089.
Romano, S., W. Goodman, R. Tamura, and J. Gonzales. 2001. Long-term treatment of obsessive–compulsive disorder after an acute response: A comparison of fluoxetine versus placebo. Journal of Clinical Psychopharmacology 21(1):46–52.
Roy-Byrne, P. P., and D. S. Cowley. 1994. Course and outcome in panic disorder: A review of recent follow-up studies. Anxiety 1(4):151–160.
Ruscio, A. M., T. A. Brown, W. T. Chiu, J. Sareen, M. B. Stein, and R. C. Kessler. 2008. Social fears and social phobia in the USA: Results from the National Comorbidity Survey Replication. Psychological Medicine 38(1):15–28.
Ruscio, A. M., D. J. Stein, W. T. Chiu, and R. C. Kessler. 2010. The epidemiology of obsessive compulsive disorder in the National Comorbidity Survey Replication. Molecular Psychiatry 15(1):53–63.
Rush, A. J., H. C. Kraemer, H. A. Sackeim, M. Fava, M. H. Trivedi, E. Frank, P. T. Ninan, M. E. Thase, A. J. Gelenberg, D. J. Kupfer, D. A. Regier, J. F. Rosenbaum, O. Ray, and A. F. Schatzberg. 2006a. Report by the ACNP Task Force on Response and Remission in Major Depressive Disorder. Neuropsychopharmacology 31(9):1841–1853.
Rush, A. J., M. H. Trivedi, S. R. Wisniewski, A. A. Nierenberg, J. W. Stewart, D. Warden, G. Niederehe, M. E. Thase, P. W. Lavori, B. D. Lebowitz, P. J. McGrath, J. F. Rosenbaum, H. A. Sackeim, D. J. Kupfer, J. Luther, and M. Fava. 2006b. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: A STARD report. American Journal of Psychiatry 163(11):1905–1917.
Salomon, J. A., J. A. Haagsma, A. Davis, C. M. de Noordhout, S. Polinder, A. H. Havelaar, A. Cassini, B. Devleesschauwer, M. Kretzschmar, N. Speybroeck, C. J. Murray, and T. Vos. 2015. Disability weights for the global burden of disease 2013 study. Lancet Global Health 3(11):e712–e723.
SAMHSA (Substance Abuse and Mental Health Services Administration). 2018. Key substance use and mental health indicators in the United States: Results from the 2017 National Survey on Drug Use and Health. Rockville, MD: Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration.
Sanderson, W. C., and T. J. Bruce. 2007. Causes and management of treatment-resistant panic disorder and agoraphobia: A survey of expert therapists. Cognitive and Behavioral Practice 14(1):26–35.
Saris, I. M. J., M. Aghajani, S. J. A. van der Werff, N. J. A. van der Wee, and B. Penninx. 2017. Social functioning in patients with depressive and anxiety disorders. Acta Psychiatrica Scandinavica 136(4):352–361.
Schmidt, N. B., J. A. Richey, J. D. Buckner, and K. R. Timpano. 2009. Attention training for generalized social anxiety disorder. Journal of Abnormal Psychology 118(1):5–14.
Schneider, R. L., J. J. Arch, and K. B. Wolitzky-Taylor. 2015. The state of personalized treatment for anxiety disorders: A systematic review of treatment moderators. Clinical Psychology Review 38:39–54.
Schneier, F. R., J. Johnson, C. D. Hornig, M. R. Liebowitz, and M. M. Weissman. 1992. Social phobia. Comorbidity and morbidity in an epidemiologic sample. Archives of General Psychiatry 49(4):282–288.
Schneier, F. R., L. F. Barnes, S. M. Albert, and E. D. Louis. 2001. Characteristics of social phobia among persons with essential tremor. Journal of Clinical Psychiatry 62(5):367–372.
Schneier, F. R., T. E. Foose, D. S. Hasin, R. G. Heimberg, S. M. Liu, B. F. Grant, and C. Blanco. 2010. Social anxiety disorder and alcohol use disorder co-morbidity in the National Epidemiologic Survey on Alcohol and Related Conditions. Psychologic Medicine 40(6):977–988.
Schnurr, P. P., and M. K. Jankowski. 1999. Physical health and post-traumatic stress disorder: Review and synthesis. Seminars in Clinical Neuropsychiatry 4(4):295–304.
Schnurr, P. P., A. F. Hayes, C. A. Lunney, M. McFall, and M. Uddo. 2006. Longitudinal analysis of the relationship between symptoms and quality of life in veterans treated for posttraumatic stress disorder. Journal of Consulting and Clinical Psychology 74(4):707–713.
Schnurr, P. P., C. A. Lunney, M. J. Bovin, and B. P. Marx. 2009. Posttraumatic stress disorder and quality of life: Extension of findings to veterans of the wars in Iraq and Afghanistan. Clinical Psychology Review 29(8):727–735.
Scholten, W. D., N. M. Batelaan, A. J. van Balkom, B. Wjh Penninx, J. H. Smit, and P. van Oppen. 2013. Recurrence of anxiety disorders and its predictors. Journal of Affective Disorders 147(1–3):180–185.
Schopman, S. M. E., M. Ten Have, S. van Dorsselaer, R. de Graaf, and N. M. Batelaan. 2018. Limited functioning after remission of an anxiety disorder as a trait effect versus a scar effect: Results of a longitudinal general population study. Journal of Clinical Psychiatry 79(3):16m11256.
Schowalter, M., G. Gelbrich, S. Stork, J. P. Langguth, C. Morbach, G. Ertl, H. Faller, and C. E. Angermann. 2013. Generic and disease-specific health-related quality of life in patients with chronic systolic heart failure: Impact of depression. Clinical Research in Cardiology 102(4):269–278.
Scott, K. M., M. Von Korff, J. Alonso, M. C. Angermeyer, E. Bromet, J. Fayyad, G. de Girolamo, K. Demyttenaere, I. Gasquet, O. Gureje, J. M. Haro, Y. He, R. C. Kessler, D. Levinson, M. E. Medina Mora, M. Oakley Browne, J. Ormel, J. Posada-Villa, M. Watanabe, and D. Williams. 2009. Mental–physical co-morbidity and its relationship with disability: Results from the World Mental Health Surveys. Psychologic Medicine 39(1):33–43.
Sharma, E., K. Thennarasu, and Y. C. Reddy. 2014. Long-term outcome of obsessive–compulsive disorder in adults: A meta-analysis. Journal of Clinical Psychiatry 75(9):1019–1027.
Shear, M. K., T. A. Brown, D. H. Barlow, R. Money, D. E. Sholomskas, S. W. Woods, J. M. Gorman, and L. A. Papp. 1997. Multicenter collaborative panic disorder severity scale. American Journal of Psychiatry 154(11):1571–1575.
Shear, M. K., P. Rucci, J. Williams, E. Frank, V. Grochocinski, J. Vander Bilt, P. Houck, and T. Wang. 2001. Reliability and validity of the Panic Disorder Severity Scale: Replication and extension. Journal of Psychiatric Research 35(5):293–296.
Sheehan, D. V., K. Nakagome, Y. Asami, E. A. Pappadopulos, and M. Boucher. 2017. Restoring function in major depressive disorder: A systematic review. Journal of Affective Disorders 215:299–313.
Siev, J., and D. L. Chambless. 2007. Specificity of treatment effects: Cognitive therapy and relaxation for generalized anxiety and panic disorders. Journal of Consulting and Clinical Psychology 75(4):513–522.
Snyder, H. R. 2013. Major depressive disorder is associated with broad impairments on neuropsychological measures of executive function: A meta-analysis and review. Psychological Bulletin Journal 139(1):81–132.
Solomon, D. A., A. C. Leon, W. H. Coryell, J. Endicott, C. Li, J. G. Fiedorowicz, L. Boyken, and M. B. Keller. 2010. Longitudinal course of bipolar I disorder: Duration of mood episodes. Archives of General Psychiatry 67(4):339–347.
Solomon, D. A., J. G. Fiedorowicz, A. C. Leon, W. Coryell, J. Endicott, C. Li, R. J. Boland, and M. B. Keller. 2013. Recovery from multiple episodes of bipolar I depression. Journal of Clinical Psychiatry 74(3):e205–e211.
Spitzer, R. L., K. Kroenke, J. B. Williams, and B. Lowe. 2006. A brief measure for assessing generalized anxiety disorder: The GAD-7. Archives of Internal Medicine 166(10):1092–1097.
Spoont, M. R., J. W. Williams, Jr., S. Kehle-Forbes, J. A. Nieuwsma, M. C. Mann-Wrobel, and R. Gross. 2015. Does this patient have posttraumatic stress disorder?: Rational clinical examination systematic review. JAMA 314(5):501–510.
Springer, K. S., H. C. Levy, and D. F. Tolin. 2018. Remission in CBT for adult anxiety disorders: A meta-analysis. Clinical Psychology Review 61:1–8.
Stangier, U., E. Schramm, T. Heidenreich, M. Berger, and D. M. Clark. 2011. Cognitive therapy vs. interpersonal psychotherapy in social anxiety disorder: A randomized controlled trial. Archives of General Psychiatry 68(7):692–700.
Steidtmann, D., R. Manber, C. Blasey, J. C. Markowitz, D. N. Klein, B. O. Rothbaum, M. E. Thase, J. H. Kocsis, and B. A. Arnow. 2013. Detecting critical decision points in psychotherapy and psychotherapy + medication for chronic depression. Journal of Consulting and Clinical Psychology 81(5):783–792.
Steketee, G. 1997. Disability and family burden in obsessive–compulsive disorder. Canadian Journal of Psychiatry 42(9):919–928.
Strawbridge, R., B. Carter, L. Marwood, B. Bandelow, D. Tsapekos, V. L. Nikolova, R. Taylor, T. Mantingh, V. de Angel, F. Patrick, A. J. Cleare, and A. H. Young. 2019. Augmentation therapies for treatment-resistant depression: Systematic review and meta-analysis. British Journal of Psychiatry 214(1):42–51.
Szkodny, L. E., M. G. Newman, and M. R. Goldfried. 2014. Clinical experiences in conducting empirically supported treatments for generalized anxiety disorder. Behavior Therapy 45(1):7–20.
Szmulewicz, A. G., M. P. Valerio, J. M. Smith, C. Samame, D. J. Martino, and S. A. Strejilevich. 2017. Neuropsychologic profiles of major depressive disorder and bipolar disorder during euthymia. A systematic literature review of comparative studies. Psychiatry Research 248:127–133.
Taylor, S., J. S. Abramowitz, and D. McKay. 2012. Non-adherence and non-response in the treatment of anxiety disorders. Journal of Anxiety Disorders 26(5):583–589.
Thase, M. E., B. R. Haight, N. Richard, C. B. Rockett, M. Mitton, J. G. Modell, S. VanMeter, A. E. Harriett, and Y. Wang. 2005. Remission rates following antidepressant therapy with bupropion or selective serotonin reuptake inhibitors: A meta-analysis of original data from 7 randomized controlled trials. Journal of Clinical Psychiatry 66(8):974–981.
Thase, M. E., A. A. Nierenberg, P. Vrijland, H. J. van Oers, A. J. Schutte, and J. H. Simmons. 2010. Remission with mirtazapine and selective serotonin reuptake inhibitors: A meta-analysis of individual patient data from 15 controlled trials of acute phase treatment of major depression. International Clinical Psychopharmacology 25(4):189–198.
Tolin, D. F., G. J. Diefenbach, and C. M. Gilliam. 2011. Stepped care versus standard cognitive–behavioral therapy for obsessive–compulsive disorder: A preliminary study of efficacy and costs. Depression and Anxiety 28(4):314–323.
Trivedi, D. 2018. Cochrane review summary: Interventions to improve return to work in depressed people. Primary Health Care Research & Development 19(2):107–109.
Trivedi, M. H. 2013. Evaluating and monitoring treatment response in depression using measurement-based assessment and rating scales. Journal of Clinical Psychiatry 74(7):e14.
VA/DoD (U.S. Department of Veterans Affairs and U.S. Department of Defense). 2016. VA/DoD clinical practice guideline for the management of major depressive disorder. Washington, DC: U.S. Department of Veterans Affairs and U.S. Department of Defense.
VA/DoD. 2017. VA/DoD clinical practice guideline for the management of posttraumatic stress disorder and acute stress disorder. Washington, DC: U.S. Department of Veterans Affairs.
van Bronswijk, S., N. Moopen, L. Beijers, H. G. Ruhe, and F. Peeters. 2019. Effectiveness of psychotherapy for treatment-resistant depression: A meta-analysis and meta-regression. Psychologic Medicine 49(3):366–379.
van der Heiden, C., P. Muris, and H. T. van der Molen. 2012. Randomized controlled trial on the effectiveness of metacognitive therapy and intolerance-of-uncertainty therapy for generalized anxiety disorder. Behaviour Research and Therapy 50(2):100–109.
van Dis, E. A. M., S. C. van Veen, M. A. Hagenaars, N. M. Batelaan, C. L. H. Bockting, R. M. van den Heuvel, P. Cuijpers, and I. M. Engelhard. 2019. Long-term outcomes of cognitive behavioral therapy for anxiety-related disorders. A systematic review and meta-analysis. JAMA Psychiatry, November 23 [Epub ahead of print].
van Oppen, P., A. J. van Balkom, E. de Haan, and R. van Dyck. 2005. Cognitive therapy and exposure in vivo alone and in combination with fluvoxamine in obsessive–compulsive disorder: A 5-year follow-up. Journal of Clinical Psychiatry 66(11):1415–1422.
Velly, A. M., and S. Mohit. 2018. Epidemiology of pain and relation to psychiatric disorders. Progress in Neuro-Psychopharmacology and Biological Psychiatry 87(Pt B):159–167.
Vieta, E., and T. Suppes. 2008. Bipolar II disorder: Arguments for and against a distinct diagnostic entity. Bipolar Disorders 10(1 Pt 2):163–178.
Wagner, S., A. Engel, J. Engelmann, D. Herzog, N. Dreimuller, M. B. Muller, A. Tadic, and K. Lieb. 2017. Early improvement as a resilience signal predicting later remission to antidepressant treatment in patients with major depressive disorder: Systematic review and meta-analysis. Journal of Psychiatric Research 94:96–106.
Wang, P. S., M. Lane, M. Olfson, H. A. Pincus, K. B. Wells, and R. C. Kessler. 2005. Twelvemonth use of mental health services in the United States: Results from the National Comorbidity Survey Replication. Archives of General Psychiatry 62(6):629–640.
Watts, B. V., P. P. Schnurr, L. Mayo, Y. Young-Xu, W. B. Weeks, and M. J. Friedman. 2013. Meta-analysis of the efficacy of treatments for posttraumatic stress disorder. Journal of Clinical Psychiatry 74(6):e541–e550.
Weathers, F. W., T. M. Keane, and J. R. Davidson. 2001. Clinician-administered PTSD scale: A review of the first ten years of research. Depression and Anxiety 13(3):132–156.
Weathers, P. L., M. J. Furlong, and D. Solórzano. 1993. Mail survey research in counseling psychology: Current practice and suggested guidelines. Journal of Counseling Psychology 40(2):238–244.
Weissman, M. M., R. C. Bland, G. J. Canino, C. Faravelli, S. Greenwald, H. G. Hwu, P. R. Joyce, E. G. Karam, C. K. Lee, J. Lellouch, J. P. Lepine, S. C. Newman, M. A. Oakley-Browne, M. Rubio-Stipec, J. E. Wells, P. J. Wickramaratne, H. U. Wittchen, and E. K. Yeh. 1997. The cross-national epidemiology of panic disorder. Archives of General Psychiatry 54(4):305–309.
Weissman, M. M., H. Verdeli, M. J. Gameroff, S. E. Bledsoe, K. Betts, L. Mufson, H. Fitterling, and P. Wickramaratne. 2006. National survey of psychotherapy training in psychiatry, psychology, and social work. Archives of General Psychiatry 63(8):925–934.
Westen, D., and K. Morrison. 2001. A multidimensional meta-analysis of treatments for depression, panic, and generalized anxiety disorder: An empirical examination of the status of empirically supported therapies. Journal of Consulting and Clinical Psychology 69(6):875–899.
Williams, T., C. J. Hattingh, C. M. Kariuki, S. A. Tromp, A. J. van Balkom, J. C. Ipser, and D. J. Stein. 2017. Pharmacotherapy for social anxiety disorder (SAnD). Cochrane Database of Systematic Reviews 2017(10):CD001206.
Wittchen, H. U., and L. Fehm. 2003. Epidemiology and natural course of social fears and social phobia. Acta Psychiatrica Scandinavica. Supplementum (417):4–18.
Wittchen, H. U., A. T. Gloster, K. Beesdo-Baum, G. A. Fava, and M. G. Craske. 2010. Agoraphobia: A review of the diagnostic classificatory position and criteria. Depression and Anxiety 27(2):113–133.
Wolitzky-Taylor, K., M. Zimmermann, J. J. Arch, E. De Guzman, and I. Lagomasino. 2015. Has evidence-based psychosocial treatment for anxiety disorders permeated usual care in community mental health settings? Behaviour Research and Therapy 72:9–17.
Wuyek, L. A., M. M. Antony, and R. E. McCabe. 2011. Psychometric properties of the Panic Disorder Severity Scale: Clinician-administered and self-report versions. Clinical Psychology & Psychotherapy 18(3):234–243.
Yaryura-Tobias, J. A., M. S. Grunes, J. Todaro, D. McKay, F. A. Neziroglu, and R. Stockman. 2000. Nosological insertion of axis I disorders in the etiology of obsessive–compulsive disorder. Journal of Anxiety Disorders 14(1):19–30.
Yatham, L. N., I. J. Torres, G. S. Malhi, S. Frangou, D. C. Glahn, C. E. Bearden, K. E. Burdick, A. Martinez-Aran, S. Dittmann, J. F. Goldberg, A. Ozerdem, O. Aydemir, and K. N. Chengappa. 2010. The International Society for Bipolar Disorders Battery for Assessment of Neurocognition (ISBD–BACN). Bipolar Disorders 12(4):351–363.
Yatham, L. N., S. H. Kennedy, S. V. Parikh, A. Schaffer, D. J. Bond, B. N. Frey, V. Sharma, B. I. Goldstein, S. Rej, S. Beaulieu, M. Alda, G. MacQueen, R. V. Milev, A. Ravindran, C. O’Donovan, D. McIntosh, R. W. Lam, G. Vazquez, F. Kapczinski, R. S. McIntyre, J. Kozicky, S. Kanba, B. Lafer, T. Suppes, J. R. Calabrese, E. Vieta, G. Malhi, R. M. Post, and M. Berk. 2018. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disorders 20(2):97–170.
Yonkers, K. A., I. R. Dyck, M. Warshaw, and M. B. Keller. 2000. Factors predicting the clinical course of generalised anxiety disorder. British Journal of Psychiatry 176:544–549.




















































































