2
Role of Topical Pain Creams in Pain Management
Pain is a global problem affecting approximately 1 in 5 adults across the world, with nearly 1 in 10 adults newly diagnosed with chronic pain each year (Goldberg and McGee, 2011). The effective treatment of pain has therefore been viewed with increasing importance in clinical practice, leading to the consideration of pain as the “fifth vital sign” (Baker, 2017). General consensus among pain researchers and clinicians holds that optimal pain management plans require patient-centered care involving clinicians from multiple disciplines, including application of integrative treatments to enhance patient outcomes, relieve suffering, and restore function (AAPM, 2020; Gordon et al., 2005; Hsu et al., 2019; Koele et al., 2014; Scascighini et al., 2008). An increasingly aging and diversifying American population is bringing limits of current pain management therapies to the forefront of consciousness, with patients and clinicians eager for better pain management strategies.
This chapter examines the complexities inherent in both the individual experience of pain and in its effective management. It also situates topical pain creams within the spectrum of integrative approaches to treating pain, with particular attention paid to special populations that may benefit from alternatives to FDA-approved pain treatments.
COMPLEXITY OF PAIN
Categories of Pain
Pain is an unpleasant sensory and emotional experience associated with actual or potential bodily damage or injury (IASP, 2019a). The experience
of pain is subjective, complex, and multifactorial (IASP, 2019a). Pain is commonly categorized broadly into three major categories: nociceptive, neuropathic, or nociplastic pain.1 Nociceptive pain arises from actual or threatened damage to nonneural tissue (IASP, 2019a). Visceral and nonvisceral nociceptive pain is commonly associated with noxious stimuli to internal organs, the musculoskeletal system, soft tissues, or skin (Leppert et al., 2018).2 Neuropathic pain—which is commonly associated with damage or disease of the somatosensory nervous system—is often described as sharp, burning, or shooting pain (Colloca et al., 2017) and may occur even in the absence of an identified condition or disease.3 Nociplastic pain may arise without any clear evidence of actual or threatened tissue damage (Trouvin and Perrot, 2019).4 In certain instances, there is overlap in the experience of these different pain types, as seen in people with chronic low back pain and cancer patients with bone metastases (Leppert et al., 2018).
The major categories of pain are often further delineated by etiology and time duration (IASP, 2019b). Examples of pain defined in terms of etiology or cause includes cancer pain (attributable to cancer or its treatment), diabetic neuropathy, postherpetic neuralgia, regional pain syndrome, erythromelalgia, and site pain. In contrast, referred pain is perceived at a site other than where the pain originates, because of interconnecting sensory nerves (Murray, 2009). As both a symptom and a disease, pain is further categorized with respect to time duration. Acute pain is generally confined to a given period of time and severity, typically lasting no more than 3 months. Chronic pain is defined by a duration persisting longer than 3 months or beyond the time for normal tissue healing (IASP, 2019b). Acute pain is often the progenitor of chronic pain, although little is known about this relationship (Gerbershagen et al., 2014). Some studies suggest that inadequate management of acute pain may increase risks for development of chronic pain (Clarke et al., 2012; Sinatra, 2010); current research efforts are examining potential strategies to decrease such risks (McGreevy et al., 2011) and help mitigate pain’s negative effect on overall health and well-being.
___________________
1 Additional pain types, including central types of pain phenomena, such as fibromyalgia syndrome, also exist.
2 Nociceptive pain is associated with conditions such as pancreatitis (visceral pain), postoperative surgery on nonneural tissue (visceral pain), fractures (nonvisceral pain), or muscle sprains (nonvisceral pain), and is generally described as aching or dull pain (Reeves and Swenson, 2008).
3 Peripheral neuropathic pain conditions include painful diabetic peripheral neuropathy and complex regional pain syndrome; central neuropathic pain conditions include pain resulting from spinal injury or stroke (Nicholson, 2006).
4 Nociplastic pain conditions include central sensitization symptoms, such as those symptoms associated with fibromyalgia syndrome.
Prevalence of Chronic Pain
A 2016 U.S. Department of Health and Human Services (HHS) survey estimated that 50 million U.S. adults are living with chronic pain, making it one of the most common reasons adults seek medical care (Dahlhamer et al., 2018). Causes of chronic nociceptive pain include musculoskeletal pain conditions including neck, back, and knee pain (Pitcher et al., 2019). Chronic neuropathic pain includes neuralgia, diabetic neuropathy, HIV infection, amputation, peripheral nerve injury pain, and stroke (Colloca et al., 2017). Conditions associated with nociplastic pain, include fibromyalgia and complex regional pain syndrome type 1 (Trouvin and Perrot, 2019). A recent survey of pain characteristics found the most common locations for chronic pain are headache/migraine, leg, low back, joints, neck, and jaw (Pitcher et al., 2019). Importantly, people living with chronic pain may experience overlapping pain conditions such as fibromyalgia, low back pain, headache, neck ache, and complex regional pain syndrome (Peppin et al., 2015). It has been proposed that chronic pain may itself be considered a disease syndrome because it leads to changes in the nervous system over time (IOM, 2011).
Chronic pain has reached epidemic portions and has become a national public health problem (NCCIH, 2018) (see Box 2-1). It is receiving a great
deal of attention owing to its cost and myriad accompanying symptoms and comorbidities, including impaired memory, cognition, and attention; sleep disturbances; reduced physical functioning; and reduced overall quality of life. Pain can interfere with all aspects of life—physical functioning, psychological functioning, economic productivity, and family and social roles—thereby impairing an individual’s overall health and well-being, while also negatively affecting the individual’s caregivers, family, and community (Dahan et al., 2014; Fine, 2011; IOM, 2011).
PHARMACOLOGICAL APPROACHES TO TREATING PAIN
A wide range of pharmacological therapies are available to treat acute and chronic pain conditions (Turk et al., 2011). The most common U.S. Food and Drug Administration (FDA)-approved medications include nonopioid analgesic medications (e.g., acetaminophen; nonsteroidal antiinflammatory drugs [NSAIDs], including COX-2 inhibitors; ibuprofen; aspirin), opioid agonists, and adjuvant analgesic drugs (IOM, 2011). Other classes of medications, some of which are not FDA approved to treat pain indications, have limited evidence to suggest effectiveness in off-label use to alleviate pain. These therapies include but are not limited to medications within the classes of antidepressants, anticonvulsants, glucocorticoids, muscle relaxants, and cannabinoids.
Topical Pain Creams
Although there are multiple routes of administration for analgesics,5 the committee focused on topical pain creams in this report. For many years, topical pain creams have augmented pain management. In theory, topical pain creams provide clinicians with the ability to treat pain through multiple mechanisms of action and offer a level of versatility potentially greater than that of oral dosage forms. For example, topical creams can be
___________________
5 Routes of administration include oral, sublingual, buccal, intranasal, inhaled, subcutaneous, intravenous, intramuscular, rectal, intramedullary, intrathecal, transdermal, and topical (Leppert et al., 2018).
formulated to support the penetration of drugs into the skin’s subcutaneous skin tissue to produce intended local, regional, and/or systemic effects to alleviate pain symptoms.6 Local effects occur when the drug is applied and absorbed into the skin at the site of pain (Leppert et al., 2018). Systemic effects occur when a drug is absorbed through the skin and accesses the blood stream for distribution throughout the body by the systemic circulation. In this circumstance, the drug does not have to be applied on the skin at the site of pain. Regional effects are experienced when a drug is applied to the skin near the intended site of action. In this setting, a small concentration of the drug may enter into the systemic circulation, even though this may not be the intended action of the drug.
Because topical medications provide a noninvasive method to administer pain medication, this may support patient compliance (Pickering et al., 2017). Topical application of pain medications for local or regional pain may also potentially avoid adverse side effects that occur when analgesics are administered by other routes that result in higher systemic levels (e.g., oral) (Leppert et al., 2018). The complexity of pain management and the many proposed advantages of topical pain creams have fueled increased interest in personalized pain treatment options, including compounded topical preparations created to address a patient’s specific needs.
Potential Role of the Placebo Effect in Topical Pain Creams
The placebo effect is an important consideration that pertains to pain treatments, including topical modalities. A placebo is an inactive treatment or sham procedure. The placebo effect occurs when the therapeutic benefit reported by a control group (i.e., a group that received a treatment with no active ingredients) was the same or greater than groups that received the experimental drug or treatment (NASEM, 2017; NIA, 2020). This difference is attributable to more than just the sham treatment, medicine, or procedure. It can be influenced by any of the various factors that contribute to the overall therapeutic context of the situation, such as the patient–physician relationship and emotional factors (IOM, 2011). Placebo effects are a known element of pain treatments in particular because it has been shown in research and clinical settings that the expectation of pain relief can induce an analgesic effect. The extent of a placebo effect is highly variable and is influenced by prior experiences and verbal suggestions. In fact, the effect of any pain
___________________
6 Drugs intended for systemic absorption are placed in more complex transdermal delivery systems to better control their release. Such systems are not the focus of this study. In addition, there are a number of critical clinical factors that attribute to the penetration and absorption of drugs through skin and are recognized as a highly complex phenomenon (see Law et al., 2020, and Chapter 5 for additional details).
treatment may be a combination of the therapeutic agent and the placebo effect (IOM, 2011). Given this background, pharmacological treatments must show an effect over and above the placebo effect in randomized controlled trials (RCTs) to be approved by FDA for human use (Vase and Wartolowska, 2019). Meta-analyses of RCTs for FDA-approved chronic pain treatments report small, but significant, improvements in pain and physical functioning when compared with placebo (Busse et al., 2018; IOM, 2011).
The placebo effect has further complicated the search for effectiveness and mechanisms of action in compounded topical pain creams, owing to a long-held belief that the majority of pain relief from topical applications comes from cutaneous stimulation or rubbing of the painful area. While many RCT studies have disproved this by having the control group rub a placebo (Creamer, 2000; Derry et al., 2017), it remains true that rubbing an area often relieves pain (Field et al., 2014; Lee et al., 2015; Satran and Goldstein, 1973). Placebo effect also indicates that elements of care other than the drug can also have beneficial effects on alleviating pain.
COMPLEXITY OF PAIN MANAGEMENT
General consensus across research efforts and medical professional guidelines holds that optimal pain management plans require early access to high-quality patient-centered care involving clinicians from multiple disciplines (e.g., dentistry, medicine, nursing, occupational therapy, physical therapy, psychology, social work) and the application of integrative treatments (e.g., cognitive-behavioral, physical/rehabilitation, interventional strategies, and pharmaceutical strategies) to enhance patient outcomes, relieve suffering, and restore function (AAPM, 2020; Gordon et al., 2005; Hsu et al., 2019; Koele et al., 2014; Scascighini et al., 2008). A 2019 report from the HHS Pain Management Best Practices Inter-Agency Task Force also promotes the use of multiple approaches to pain management in order to act synergistically on different aspects of an individual’s pain (HHS, 2019). Figure 2-1 provides illustrative examples of how different patients may interact with health services to arrive at an individualized (personalized) pain management plan, as well as depicting the wide variety of treatment options in a clinician’s toolbox.
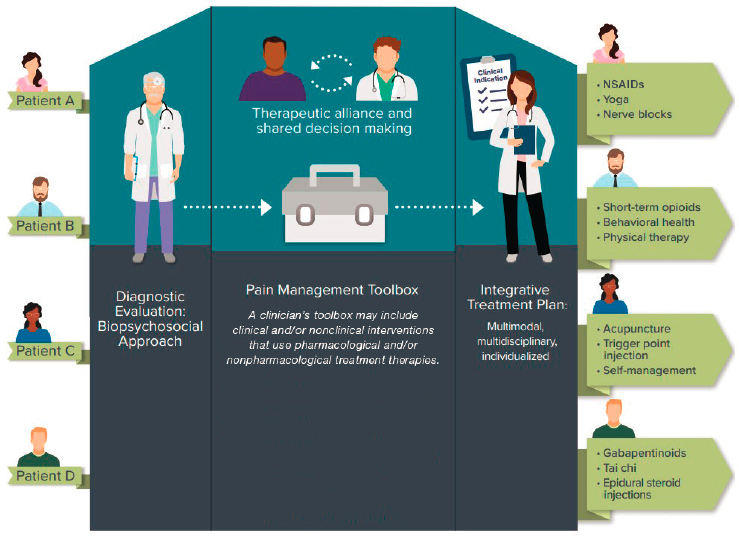
SOURCE: Adapted from HHS, 2019.
Pain treatments range from simple and inexpensive (e.g., massage) to complicated and expensive (e.g., surgical interventions, nerve blocks); pain treatment also incorporates shared decision making (Tick et al., 2018). An overview of nonpharmacological approaches to treating pain is provided in Box 2-2. Although progress has been made, additional clinical studies are needed to help clinicians determine the optimal sequence of therapies for each patient (Colloca et al., 2017). Given the broad spectrum of available pain therapies and treatment approaches, certain considerations need to be made prior to implementing an integrative pain treatment plan. These considerations include the clinical status of the patient, comorbidities, age, socioeconomic status, gender, race, and ethnicity (McCleane, 2008). In addition, the development of a pain management plan should consider patient adherence—meaning, whether they will reliably take the medicines as prescribed—and the need to minimize potential adverse side effects of treatment (e.g., caused by the route of analgesic administration and drug dose) (Leppert et al., 2018). In certain situations, prescribing clinicians may consider including compounded topical pain creams in the treatment plans of patients whose needs cannot be fully addressed by FDA-approved topical pain products.
Drug–Drug Interactions
Populations who are more likely to use pharmacological pain treatments (e.g., geriatrics, cancer patients) are also more likely to be using other medications concurrently. It is therefore important to consider the potential risks and adverse events associated with polypharmacy and drug–drug interactions. Such interactions occur when a drug, supplement, or food affects a medication’s effectiveness, often through enzyme induction or inhibition. For example, if an individual is taking two drugs that are both metabolized by the same enzyme pathway, the drugs could initially be limited in their effectiveness because they are in competition for limited enzymes. However, if the medications are taken for a long period of time, they could cause the body to overproduce those enzymes, thus increasing the body’s capacity to metabolize those drugs and reduce the medications’ potential effectiveness. One study found that each additional prescription dispensed increases the potential risk of a drug–drug interaction by 138 percent (Taylor et al., 2013). Little is known about the full gamut of potential drug–drug interactions, and even less about the potential interactions that arise from exposure to topical medications. See Chapter 6 for an additional discussion on the dearth of information available on the safety and effectiveness of ingredients commonly used in compounded topical pain creams containing multiple active pharmaceutical ingredients, and see Chapter 7 for additional discussion on risk of potential drug–drug interactions to patients.
Special Populations to Consider in Pain and Pain Management
The individual experience of pain and response to painful stimuli is highly variable. A key factor in this variability may be the way the pain message is modulated in an individual’s central nervous system (Ossipov et al., 2010), suggesting that clinicians’ ability to diagnose and treat pain depends on their understanding of the various processes involved in experiencing pain (Sluka et al., 2009). Mechanisms underlying individual differences in pain response include the following:
- Biological factors: genetics, developmental stage, gonadal (sex) hormones, endogenous pain inhibition and modulation7
- Psychological factors: pain coping, anxiety, depression, cognitive factors, behavioral factors8
- Sociocultural factors: social determinants of health, culture, discrimination, gender roles, family history, work roles9 (Sluka et al., 2009)
Accordingly, there is great variability in patients’ response to specific pharmacological and nonpharmacological pain treatments (Skelly et al., 2018; Twycross et al., 2015). Pain management decision making is also variable and complicated by disparities in access to care driven by sociodemographic and economic factors. Many patients report that their pain complaints are often unheard (or dismissed) and therefore undertreated10 (Baratta et al., 2014; Sinatra, 2010). Pain care is further complicated by insufficient clinician and patient education, misinformation, poor communication between clinicians and patients, and variability in clinician attitudes.11
Complexities of pain and its management are further intensified in special populations. The confluence of biological, psychosocial, and sociodemographic factors that drive wide variability in individual-level responses
___________________
7 Twin studies have demonstrated that genetic influences account for approximately 50 percent of the variance in chronic pain (Fillingim et al., 2008).
8 There is a high prevalence of psychiatric illnesses in chronic pain patients, which are associated with increased pain perception, as well as impairment in activity and function (Rajmohan and Kumar, 2013).
9 Differences in pain sensitivity between women and men are partly attributable to social conditioning and to psychosocial factors (Wiesenfeld-Hallin, 2005).
10 Undertreatment of pain can lead to decreased physical activities, diminished mental health, weakened social interactions, missed workdays, and lower quality of life while also placing an increased financial burden on the patient and health care system as a whole.
11 Physicians report lesser goals for treating chronic pain and less satisfaction with their care when compared to other conditions, including acute and cancer pain, but they report greater goals for pain management in the palliative care arena (Green et al., 2003).
to pain also contribute to substantial between-group differences (Coghill, 2010; Fillingim et al., 2008). Social determinants of health—including age, race, ethnicity, gender, place of residence, and socioeconomic status—profoundly influence pain measurement, coping, response, and overall disability (Green et al., 2003). Factors that mediate treatment differences among special populations remain poorly understood, affecting the quality of pain care received by those groups (Green et al., 2003). Furthermore, the safety and efficacy of FDA-approved pain treatments typically are not studied in representative populations stratified by age, race, gender, or other sociodemographic factors (Green et al., 2003; McBane et al., 2019). As a result, there is much to learn regarding the potential clinical usefulness of topical pain creams and other alternative modalities such as compounded topical pain creams to address the complexities of pain management in these special populations. See Appendix H for an additional discussion on the understudied populations highlighted above.
REFERENCES
AAPM (American Academy of Pain Medicine). 2020. What is pain medicine? https://painmed.org/about/what-is-pain-medicine (accessed March 18, 2020).
Baker, D. 2017. The Joint Commission’s pain standards: Origins and evolution. Oakbrook Terrace, IL: The Joint Commission.
Baratta, J. L., E. S. Schwenk, and E. R. Viscusi. 2014. Clinical consequences of inadequate pain relief: Barriers to optimal pain management. Plastic and Reconstructive Surgery 134(4 Suppl 2):15s–21s.
Busse, J. W., L. Wang, M. Kamaleldin, S. Craigie, J. J. Riva, L. Montoya, S. M. Mulla, L. C. Lopes, N. Vogel, E. Chen, K. Kirmayr, K. De Oliveira, L. Olivieri, A. Kaushal, L. E. Chaparro, I. Oyberman, A. Agarwal, R. Couban, L. Tsoi, T. Lam, P. O. Vandvik, S. Hsu, M. M. Bala, S. Schandelmaier, A. Scheidecker, S. Ebrahim, V. Ashoorion, Y. Rehman, P. J. Hong, S. Ross, B. C. Johnston, R. Kunz, X. Sun, N. Buckley, D. I. Sessler, and G. H. Guyatt. 2018. Opioids for chronic noncancer pain: A systematic review and meta-analysis. JAMA 320(23):2448–2460.
Clarke, H., R. P. Bonin, B. A. Orser, M. Englesakis, D. N. Wijeysundera, and J. Katz. 2012. The prevention of chronic postsurgical pain using gabapentin and pregabalin: A combined systematic review and meta-analysis. Anesthesia & Analgesia 115(2):428–442.
Coghill, R. C. 2010. Individual differences in the subjective experience of pain: New insights into mechanisms and models. Headache 50(9):1531–1535.
Colloca, L., T. Ludman, D. Bouhassira, R. Baron, A. H. Dickenson, D. Yarnitsky, R. Freeman, A. Truini, N. Attal, N. B. Finnerup, C. Eccleston, E. Kalso, D. L. Bennett, R. H. Dworkin, and S. N. Raja. 2017. Neuropathic pain. Nature Reviews Disease Primers 3:17002.
Creamer, P. 2000. Osteoarthritis pain and its treatment. Current Opinion Rheumatology 12(5):450–455.
Dahan, A., M. van Velzen, and M. Niesters. 2014. Comorbidities and the complexities of chronic pain. Anesthesiology 121(4):675–677.
Dahlhamer, J., J. Lucas, C. Zalaya, R. Nahin, S. Mackey, L. DeBar, R. Kerns, M. Von Korff, L. Porter, and C. Helmick. 2018. Prevalence of chronic pain and high-impact chronic pain among adults—United States, 2016. Morbidity and Mortality Weekly Report 67:1001–1006.
Derry, S., P. J. Wiffen, E. A. Kalso, R. F. Bell, D. Aldington, T. Phillips, H. Gaskell, and R. A. Moore. 2017. Topical analgesics for acute and chronic pain in adults—An overview of Cochrane Reviews. Cochrane Database of Systematic Reviews 5:CD008609.
Field, T., M. Diego, and L. Solien-Wolfe. 2014. Massage therapy plus topical analgesic is more effective than massage alone for hand arthritis pain. Journal of Bodywork and Movement Therapies 18(3):322–325.
Fillingim, R. B., M. R. Wallace, D. M. Herbstman, M. Ribeiro-Dasilva, and R. Staud. 2008. Genetic contributions to pain: A review of findings in humans. Oral Diseases 14(8):673–682.
Fine, P. G. 2011. Long-term consequences of chronic pain: Mounting evidence for pain as a neurological disease and parallels with other chronic disease states. Pain Medicine 12(7):996–1004.
Gaskin, D. J., and P. Richard. 2012. The economic costs of pain in the United States. The Journal of Pain 13(8):715–724.
Gerbershagen, H. J., E. Pogatzki-Zahn, S. Aduckathil, L. M. Peelen, T. H. Kappen, A. J. van Wijck, C. J. Kalkman, and W. Meissner. 2014. Procedure-specific risk factor analysis for the development of severe postoperative pain. Anesthesiology 120(5):1237–1245.
Goldberg, D. S., and S. J. McGee. 2011. Pain as a global public health priority. BMC Public Health 11:770.
Gordon, D. B., J. L. Dahl, C. Miaskowski, B. McCarberg, K. H. Todd, J. A. Paice, A. G. Lipman, M. Bookbinder, S. H. Sanders, D. C. Turk, and D. B. Carr. 2005. American Pain Society recommendations for improving the quality of acute and cancer pain management: American Pain Society Quality of Care Task Force. Archives of Internal Medicine 165(14):1574–1580.
Gureje, O., M. Von Korff, G. E. Simon, and R. Gater. 1998. Persistent pain and well-being: A World Health Organization study in primary care. JAMA 280(2):147–151.
HHS (U.S. Department of Health and Human Services). 2019. Pain Management Best Practices Inter-Agency Task Force report: Updates, gaps, inconsistencies, and recommendations. https://www.hhs.gov/ash/advisory-committees/pain/reports/index.html (accessed March 18, 2020).
Hsu, J. R., H. Mir, M. K. Wally, R. B. Seymour, and Orthopaedic Trauma Association Musculoskeletal Pain Task Force. 2019. Clinical practice guidelines for pain management in acute musculoskeletal injury. Journal of Orthopaedic Trauma 33(5):e158–e182.
IASP (International Association for the Study of Pain). 2019a. Introduction. In Classification of chronic pain, 2nd ed. (revised). https://www.iasp-pain.org/Education/Content.aspx?ItemNumber=1698&navItemNumber=576#Pain (accessed March 18, 2020).
IASP. 2019b. IASP Terminology. In Classification of Chronic Pain, 2nd ed. (revised). https://s3.amazonaws.com/rdcms-iasp/files/production/public/Content/ContentFolders/Publications2/ClassificationofChronicPain/Introduction.pdf (accessed March 18, 2020).
IOM (Institute of Medicine). 2011. Relieving pain in America: A blueprint for transforming prevention, care, education, and research. Washington, DC: The National Academies Press.
Koele, R., G. Volker, F. van Vree, M. van Gestel, A. Koke, and T. Vliet Vlieland. 2014. Multidisciplinary rehabilitation for chronic widespread musculoskeletal pain: Results from daily practice. Musculoskeletal Care 12(4):210–220.
Law, R. M., M. A. Ngo, and H. I. Maibach. 2020. Twenty clinically pertinent factors/observations for percutaneous absorption in humans. American Journal of Clinical Dermatology 21(1):85–95.
Lee, S. H., J. Y. Kim, S. Yeo, S. H. Kim, and S. Lim. 2015. Meta-analysis of massage therapy on cancer pain. Integrative Cancer Therapies 14(4):297–304.
Leppert, W., M. Malec-Milewska, R. Zajaczkowska, and J. Wordliczek. 2018. Transdermal and topical drug administration in the treatment of pain. Molecules (Basel, Switzerland) 23(3):681.
McBane, S. E., S. A. Coon, K. C. Anderson, K. E. Bertch, M. Cox, C. Kain, J. LaRochelle, D. R. Neumann, and A. M. Philbrick. 2019. Rational and irrational use of nonsterile compounded medications. Journal of the American College of Clinical Pharmacy 2(2):189–197.
McCleane, G. 2008. Pain management: Expanding the pharmacological options. Hoboken, NJ: Wiley-Blackwell.
McGreevy, K., M. M. Bottros, and S. N. Raja. 2011. Preventing chronic pain following acute pain: Risk factors, preventive strategies, and their efficacy. European Journal of Pain Supplements 5(2):365–372.
Murray, G. M. 2009. Guest editorial: Referred pain. Journal of Applied Oral Science: Revista FOB 17(6):i.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2017. Pain management and the opioid epidemic: Balancing societal and individual benefits and risks of prescription opioid use. Washington, DC: The National Academies Press.
NCCIH (National Center for Complementary amd Integrative Health). 2018. Chronic pain: In depth. https://www.nccih.nih.gov/health/chronic-pain-in-depth (accessed April 3, 2020).
NIA (National Institute on Aging). 2020. Placebos in clinical trials. https://www.nia.nih.gov/health/placebos-clinical-trials (accessed February 5, 2020).
Nicholson, B. 2006. Differential diagnosis: Nociceptive and neuropathic pain. American Journal of Managed Care 12(9 Suppl):S256–S262.
Ossipov, M. H., G. O. Dussor, and F. Porreca. 2010. Central modulation of pain. Journal of Clinical Investigation 120(11):3779–3787.
Peppin, J. F., M. D. Cheatle, K. L. Kirsh, and B. H. McCarberg. 2015. The complexity model: A novel approach to improve chronic pain care. Pain Medicine 16(4):653–666.
Pickering, G., E. Martin, F. Tiberghien, C. Delorme, and G. Mick. 2017. Localized neuropathic pain: An expert consensus on local treatments. Drug Design, Development and Therapy 11:2709–2718.
Pitcher, M. H., M. Von Korff, M. C. Bushnell, and L. Porter. 2019. Prevalence and profile of high-impact chronic pain in the United States. The Journal of Pain 20(2):146–160.
Rajmohan, V., and S. K. Kumar. 2013. Psychiatric morbidity, pain perception, and functional status of chronic pain patients in palliative care. Indian Journal of Palliative Care 19(3):146–151.
Reeves, A., and R. Swenson. 2008. Evaluation and management of pain. In Disorders of the nervous system. https://www.dartmouth.edu/~dons/part_2/chapter_19.html (accessed February 5, 2020).
Satran, R., and M. N. Goldstein. 1973. Pain perception: Modification of threshold of intolerance and cortical potentials by cutaneous stimulation. Science 180(4091):1201–1202.
Scascighini, L., V. Toma, S. Dober-Spielmann, and H. Sprott. 2008. Multidisciplinary treatment for chronic pain: A systematic review of interventions and outcomes. Rheumatology (Oxford) 47(5):670–678.
Sinatra, R. 2010. Causes and consequences of inadequate management of acute pain. Pain Medicine 11(12):1859–1871.
Skelly, A., R. Chou, J. Dettori, J. Turner, J. Friedly, S. Rundell, R. Fu, E. Brodt, N. Wasson, C. Winter, and A. Ferguson. 2018. Noninvasive nonpharmacological treatment for chronic pain: A systematic review. AHRQ Publication No 18-EHC013-EF. Rockville, MD: Agency for Healthcare Research and Quality. https://effectivehealthcare.ahrq.gov/sites/default/files/pdf/nonpharma-chronic-pain-cer-209.pdf (accessed February 5, 2020).
Sluka, K., J. Turner, B. Collett, C. Miaskowski, C. Eccleston, D. Justins, H. Wittink, H. Abu-Saad, J. Castro-Lopes, M. Bond, N. Barros, P. McGrath, P. Sjogren, S. Allen, S. Mackey, T. Ushida, and Y. Shir. 2009. Pain treatment services. International Association for the Study of Pain. https://www.iasp-pain.org/Education/Content.aspx?ItemNumber=1381 (accessed February 5, 2020).
St Sauver, J. L., D. O. Warner, B. P. Yawn, D. J. Jacobson, M. E. McGree, J. J. Pankratz, L. J. Melton, 3rd, V. L. Roger, J. O. Ebbert, and W. A. Rocca. 2013. Why patients visit their doctors: Assessing the most prevalent conditions in a defined American population. Mayo Clinic Proceedings 88(1):56–67.
Taylor, R., Jr., J. V. Pergolizzi, R. A. Puenpatom, and K. H. Summers. 2013. Economic implications of potential drug-drug interactions in chronic pain patients. Expert Review of Pharmacoeconomics & Outcomes Research 13(6):725–734.
Tick, H., A. Nielsen, K. R. Pelletier, R. Bonakdar, S. Simmons, R. Glick, E. Ratner, R. L. Lemmon, P. Wayne, and V. Zador. 2018. Evidence-based nonpharmacologic strategies for comprehensive pain care: The Consortium Pain Task Force white paper. Explore (NY) 14(3):177–211.
Trouvin, A. P., and S. Perrot. 2019. New concepts of pain. Best Practice & Research Clinical Rheumatology 33(3).
Turk, D. C., H. D. Wilson, and A. Cahana. 2011. Treatment of chronic non-cancer pain. Lancet 377(9784):2226–2235.
Twycross, R., J. Ross, A. Kotlinska-Lemieszek, S. Charlesworth, M. Mihalyo, and A. Wilcock. 2015. Variability in response to drugs. Journal of Pain and Symptom Management 49(2):293–306.
Vase, L., and K. Wartolowska. 2019. Pain, placebo, and test of treatment efficacy: A narrative review. British Journal of Anaesthesia 123(2):e254–e262.
Wiesenfeld-Hallin, Z. 2005. Sex differences in pain perception. Gender Medicine 2(3):137–145.
This page intentionally left blank.














