7
Digital Health and Primary Care
Digital technology is an essential tool necessary for primary care to carry out its basic functions. Without high-functioning digital technologies, many of the aspirations of this report are not possible. Technology in other sectors outpaces its performance in health care. The financial industry, for example, has free flow of information and complete interoperability between business silos. Smartphones are customizable with robust “app stores” to easily add and remove features. More importantly, people do not need intensive training to learn how to use smartphones, which are intuitive, recognize users’ needs, and can prompt support.
For routine use in primary care, technology has not fundamentally expanded beyond electronic health records (EHRs), registration systems, and patient portals created two decades ago. More concerning, technology remains a leading cause of clinician burnout (NASEM, 2019). In an average day, clinicians spend 6 hours documenting care in an EHR (Arndt et al., 2017). EHRs continue to require dozens of clicks, unique to each individual system and far from intuitive, in a structured format designed for meeting billing and coding requirements rather than enhancing clinical care and relationships. These excessive requirements mean that U.S. clinicians spend significantly more time working in an EHR per day than clinicians using the same EHR in other countries (Holmgren et al., 2020). Interoperability requires that the clinician significantly review and correct information from multiple sources (e.g., specialists, hospitals, vaccine registries, pharmacies), or, even worse, call and fax other care team members to find information that is sitting in a silo elsewhere and then decide if it is current and correct. National efforts to make the use of digital health more meaningful put the
majority of the burden on end users, requiring health systems and primary care practices to substantially modify and implement rudimentary base systems and adding to their workload.
The experience for people seeking care is no better. They have lost the attention of their personal clinician, who is distracted by a computer screen (O’Malley et al., 2010; Street et al., 2014). Limited interoperability introduces errors as outdated information is populated in systems, which can lead to clinicians refilling or continuing out-of-date medications. Conversely, overuse occurs when information is not transferred, as clinicians may repeat recent tests or procedures from elsewhere. Adding to this, people have limited access to their own health information through patient portals that present information in medical language. In addition, an individual’s medical history is not linked to person-centered educational material or more than a few actionable, relevant steps they can take. Finally, the experience is burdened by the very design of systems that are not created with the needs of the most underserved populations in mind. Patients with limited English language proficiency, health and digital literacy, and access to high-speed Internet often cannot gain the full benefit of technology.
This is the lived experience of health information technology (HIT) for clinicians and patients. The real potentials of digital health to aggregate a wide array of medical, environmental, biological, and social data; make meaningful sense of information; automate care; make care proactive and not reactive; enhance health equity; and enable population health monitoring and management have been barely explored. The concept of “digital capitalism” in the health sector creating a dichotomy of public benefit versus private gain offers a compelling explanation for the current lack of innovation, exploration, and interoperability compared to other sectors (Sharon, 2018). Furthermore, the lack of adequate regulation of digital health companies to ensure innovation, interoperability, and support of the functions of high-quality primary care continues to put solutions out of reach for health care organizations and individual clinicians.
The committee broadly refers to digital health as the use of HIT to care for individuals and communities. Digital health has many uses—documenting care, collecting and storing information, understanding information, delivering care, and communicating. At its best, though, digital health should make it easier for people to receive and clinicians to know how to deliver the right care at the right time, while also supporting relationships between individuals, families, clinicians, and communities. In primary care, digital health can include tools such as EHRs, patient portals, mobile applications, telemedicine platforms, electronic registries, analytic systems, remote monitoring, wearable technology, care-seeker and care team communication support, and geographical and population health displays. Digital health
tools used for diagnosis and treatment are beyond the scope of this report, which is focused on implementing high-quality primary care.
Over the past 20 years, rapid improvements in computing power, software development, data storage, and Internet bandwidth, as well as smartphone proliferation, have supported the growth of a digital health industry that has developed products and technologies that are ubiquitous in health care delivery today. Undoubtedly, future technology disruption will continue to change health care and improve primary care. This chapter will discuss digital health’s role in delivering care in a way that is convenient, accessible, and efficient, the key attributes of a well-functioning digital health infrastructure, and the barriers to fully benefiting primary care. This chapter will also describe how the recent COVID-19 pandemic has forced many primary care practices to rapidly transform their processes to make virtual care and population care a new norm and how the lessons learned from this experience can enhance equity. These lessons need to be applied to future digital health advances and the policies that enable them. Finally, the chapter will present design principles for digital health systems and implementation strategies for different actors to consider in strengthening the role of digital health to support high-quality primary care.
THE GROWTH OF DIGITAL HEALTH
The digital health landscape has advanced dramatically in the years following Primary Care: America’s Health in a New Era (IOM, 1996a). In 2004, the Office of the National Coordinator for Health Information Technology (ONC) was established, followed by Congress passing the Health Information Technology for Economic and Clinical Health (HITECH)1 Act (part of the American Recovery and Reinvestment Act) in 2009. The HITECH Act mandated ONC to regulate and create standards for HIT and create incentives for its adoption (Washington et al., 2017). Congress also passed the Health Insurance Portability and Accountability Act (HIPAA)2 in 1996, to promote and support health information exchange. However, HIPAA has largely been a barrier to information exchange and is badly in need of updating (NCVHS, 2019).
Largely spurred by the HITECH Act, some digital health care applications, specifically EHRs, are commonplace today and have been widely adopted by a vast majority of health care delivery systems, large and small. By 2017, 86 percent of all office-based physicians were using an EHR and
___________________
1 American Recovery and Reinvestment Act of 2009, § 13001, Public Law 111-5 (February 17, 2009).
2 Health Insurance Portability and Accountability Act of 1996, Public Law 104-191 (August 21, 1996).
80 percent were using a certified EHR product3 (ONC, 2019). Primary care has been an early adopter of HIT, often leading the way in EHRs and advocating for greater functionality (Phillips et al., 2015; Rittenhouse et al., 2017). In a 2019 survey, a majority of physicians across specialties felt that technology enabled them to provide better care. Among primary care physicians, 40 percent reported that technology gave them a “definite” advantage, and 46 percent reported that it gave them “somewhat” of an advantage in providing care (AMA, 2020).
Electronic Health Records and Meaningful Use
The HITECH Act established programs through the Centers for Medicare & Medicaid Services (CMS), known collectively as Meaningful Use, to incentivize EHR adoption with the goal of improving the quality and coordination of care, patient engagement, and population health, while ensuring the privacy of patients and their personal information (CDC, 2019). Meaningful Use awarded financial incentives if practices satisfied a set of core objectives and several optional objectives (Fernald et al., 2013). Meaningful Use stage 1 focused on data capture and sharing, stage 2 on advanced clinical processes, and stage 3 on improved outcomes (Ornstein et al., 2015), but the EHR functionality and use requirements only supported very basic functions of primary care (Krist et al., 2014). For example, one stage 1 objective was to maintain an up-to-date problem list for 80 percent of patients and a stage 2 objective was to use secure messaging for 10 percent of patient communications.
Meaningful Use did catalyze the uptake of HIT in primary care, and it also created burdens for which the incentives only partially compensated. EHRs were and remain expensive, with most practices paying more for one than they were paid through Meaningful Use incentives (Fleming et al., 2011). In addition, clinicians and staff received inadequate training and support to use EHR functionality to its full potential, and the EHRs had many technical limitations that required extra staff work to fulfill the Meaningful Use criteria (Fernald et al., 2013). Meaningful Use did result in some improvements in quality, safety, and outcomes (Kruse and Beane, 2018), but it also contributed to physician burnout, EHR market oversaturation, and data obfuscation (Colicchio et al., 2019). CMS and ONC had limited authority to enforce Meaningful Use standards and primarily acted through incentive and penalty payments to clinicians. It did not provide a way to incentivize or force EHR vendors to change and improve their systems,
___________________
3 A certified EHR product has received certification from the Office of the National Coordinator for Health Information Technology that the system supports all of the required Meaningful Use functionalities (2020).
beyond certification. This put the burden to adopt, modify, and implement base EHR products to meet certification standards on clinicians and health systems. In April 2018, CMS renamed this incentive program to “Promoting Interoperability Programs” and shifted priorities to include patient access to information, interoperability, and e-prescribing (CMS, 2020a).
Today, the global EHR market is estimated to be worth $25 billion and projected to reach $37 billion by 2025 (Medgadget, 2020). Three vendors dominate, with Epic, Cerner, and Meditech controlling 75 percent of the U.S. hospital market share (Landi, 2020). Among large health systems, Epic and Cerner dominate (Tate and Warburton, 2020). For such a large market that has such a large influence on health care, the current landscape of a vendor-dominated economy locks customers into systems and stifles innovation while contributing to rising and unsustainable health care costs (Fisher et al., 2009).
Telehealth, mHealth, and Other Patient-Facing Systems
Other types of digital health, such as telemedicine, remote monitoring applications and devices, patient engagement tools, decision support technology, patient portals, data-sharing tools, and chat bots driven by artificial intelligence or ambient computing to automate care, are not used as widely as EHRs, but their adoption has increased significantly in recent years across all specialties, age groups, and genders (AMA, 2020). Today, many patients and clinicians strive for health care to be less transactional and based more on relationships and partnerships (see Chapter 4). More patients now expect to own their health information, be included with their clinicians in the health care decision-making process, and have their care be collaborative, convenient, and accessible (Meskó et al., 2017). Well-designed, person-centered technology, such as patient portals that allow patients and clinicians to communicate via secure messaging, can fill a need by facilitating efficient communication outside of the office setting and giving people easier access to their medical information (Friedberg et al., 2014; Hoonakker et al., 2017; NASEM, 2019). However, current payment systems do not reimburse clinicians for providing this critical service.
Myriad health information resources are easily accessible to the consumer via the Internet, mobile applications, and patient-facing technologies (e.g., patient portals) that allow people to more easily interact directly with their clinicians. In 2018, for example, a nationally representative survey found that 35 percent of respondents reported owning an electronic monitoring device, such as a smart watch or blood glucose monitor, and 84 percent reported owning a smartphone or tablet. Of the latter, 49 percent reported they used a health or wellness application on their device to track health goals (Patel and Johnson, 2019). However, the corresponding change
among many clinicians to adopt technologies that could make care more accessible and convenient has lagged because of a combination of factors, such as burdensome regulatory restrictions, resistance to change from a traditional hierarchical model that is less person centered, and technology-related professional burnout (Meskó et al., 2017; NASEM, 2019). For example, in 2018, only about half of patients reported being offered access to their online medical records, the same as in 2017. Of those who were, 30 percent viewed their records at least once in the past year; the impact of viewing the information on their health and engagement is unknown (Patel and Johnson, 2019).
Telemedicine4 can make care more accessible to people who have access to the required technology, such as a device with broadband Internet connectivity, by sparing them an often time-consuming and burdensome trip to a medical office. One study of telemedicine trends among a large, commercially insured population showed that primary care was the most frequently delivered form (Barnett et al., 2018). Still, the overall percentage of primary care via telemedicine was low, and it was most commonly used by people in urban locations, who typically have greater access in general compared to those in more rural locations (Barnett et al., 2018).
Personal monitoring devices, such as wearable devices (e.g., smart watch), Internet-connected scales, and glucometers, are used by individuals and clinicians to monitor health goals and vital signs between direct encounters. Wearable devices are generally not remotely integrated with an EHR, yet these same devices are widely integrated across a range of smartphone applications (Dinh-Le et al., 2019). The direct integration of glucometers within the EHR is in the early stages of development and can improve monitoring of people with diabetes between encounters (Weatherly et al., 2019). This type of digital health integration can facilitate engagement from people seeking care and make monitoring and care more efficient and less burdensome for clinicians, but only if the information is shared simply and meaningfully.
Population Health
Population health, an essential component of effective primary care that is most enabled by digital health and currently neglected, encompasses strategies to make the delivery of evidence-based care easy. It shifts care from being reactive (e.g., someone has to schedule an appointment) to being proactive (e.g., identify and reach out to those in need); and it expands
___________________
4 Telemedicine is “the use of electronic information and communications technologies to provide and support health care when distance separates the participants” usually via video conference, telephone, or mobile application (IOM, 1996b, p. 1).
care beyond individuals to caring for entire communities. For example, alerts, reminders, and quality or health maintenance tabs5 built into EHRs increase guideline-based care (Alagiakrishnan et al., 2016). Similarly, safety alerts have been shown to decrease prescribing contraindicated medications. Despite a strong evidence base demonstrating the effectiveness of these features (Bright et al., 2012), EHRs are not preprogrammed with functional and up-to-date, evidence-based guidelines, and each health system or practice must create its own quality or health maintenance tabs. Programmed medication safety alerts are more commonplace, but most systems provide too many such alerts that are not prioritized and often clinically inappropriate, resulting in alert fatigue and reducing their effectiveness (Hussain et al., 2019). Community data or “community vital signs” to consider risks for social needs and environmental exposures are emerging as a promising tool that could notify clinicians of care-seekers’ needs (Hughes et al., 2016; Liaw et al., 2018). However, all of these types of functions still occur primarily at the point of care, requiring a visit rather than being true proactive population health care features.
Patient registries are more of a true population health tool. Practices can use registries to identify all patients in their panel who have gaps in care, are overdue for care, or have risks and conditions needing additional attention (AMA, 2015, 2016). Care teams can use these tools to develop intervention strategies that target those with a range of needs to prevent negative health events (ONC, 2013). Registries are an essential function for many practices and health systems to participate in Medicare Shared Savings Programs, accountable care organizations, and other value-based programs. Some EHRs allow clinicians to generate patient lists around basic elements, such as everyone with diabetes, those needing an annual wellness visit, children in need of vaccinations, and patients overdue for colon cancer or developmental screening. However, registries are often an optional add-on to EHRs that practices need to purchase, and many are limited to basic functionality and cannot perform more sophisticated population queries or queries on nontraditional medical values. Few EHR-based registries have automated functionality, requiring users to manually create and run queries (Nelson et al., 2016).
Other population health tools can include geographic information systems (GISs) to understand a practice’s footprint and the communities that it serves (Rock et al., 2019). GIS tools can support community-based interventions and collaboration building to better care. Alternatively, clinicians can use information about a patient’s place of residence to understand
___________________
5 Quality or health maintenance tabs are sections of EHRs dedicated to displaying evidence-based alerts about preventive or chronic care recommendations that a patient may or may not need (Schellhase et al., 2003).
environmental factors that may contribute to health or even use place-based data as a surrogate for health risks (DeVoe et al., 2016; Hughes et al., 2016; Liaw et al., 2018; Tong et al., 2019).
Interoperability
Effective, functional, and automated interoperability is an essential lynchpin to ensure the success of digital health. The free flow of health information between settings and systems does not easily occur in today’s health care system because of a lack of interoperability between different technologies. This is an issue for all of health care, but it is particularly relevant for primary care, which needs complete data for whole-person care and is currently responsible for curating a person’s “complete” health record. The HITECH Act offered grants and incentives to states and municipalities for developing regional health information exchange (HIE) initiatives. These initiatives have met with variable success. Problems included technical, policy, governance, funding, and security and privacy concerns (Adler-Milstein et al., 2013; Vest and Gamm, 2010).
The ONC National Interoperability Roadmap has set 2024 as a goal for universal interoperability (HealthIT, 2020). Yet, Taking Action Against Clinician Burnout: A Systems Approach to Professional Well-Being showed that clinicians are “increasingly frustrated that the digital transition in health care has not translated into having the information necessary for patient care when and where patients need it” (NASEM, 2019, p. 207). The 21st Century Cures Act of 2016 (Cures Act)6 defines an interoperable HIT system as one that
(a) enables the secure exchange of electronic health information with, and use of electronic health information from, other health information technology without special effort on the part of the user;
(b) allows for complete access, exchange, and use of all electronically accessible health information for authorized use under applicable State or Federal law; and
(c) does not constitute information blocking.
However, the ONC Roadmap appears to be off track for achieving its goal for universal interoperability by 2024, and the ONC does not have any authority over EHR vendors to enforce the Cures Act requirements.
Most current EHRs and other IT systems do not meet the interoperability standard of the Cures Act for a variety of reasons (Blumenthal, 2009; NASEM, 2019; Pronovost et al., 2018), such as the proprietary policies of
___________________
6 21st Century Cures Act of 2016, Public Law No. 114-255 (December 13, 2016).
EHR vendors and health care organizations and ineffective incentives (Ratwani et al., 2018a). The lack of interoperability, and the restriction of the flow of information by some vendors, negatively affects care. Even within a single health system, information is often siloed in different HIT systems that may not be interoperable. For example, one specialty may use an EHR that is different from and not interoperable with the EHR used by the rest of the system (Friedberg et al., 2014). As noted in Taking Action Against Clinician Burnout, a lack of interoperability “can increase administrative and clerical efforts to ensure that all tests results, scheduling updates, orders, and clinical notes are accurate and consistent across the different systems” (NASEM, 2019, p. 207). Similarly, even if two different health systems use EHRs from the same vendor, they may not be compatible. As a result, when patients move among health systems, transferring their medical information may require manual input, a time-consuming process prone to human error (Smith et al., 2018). Thus, “the lack of interoperability compounds the administrative burden placed on clinical staff, which erodes efficiency and contributes to fatigue and dissatisfaction” (NASEM, 2019, p. 208).
Several efforts are under way to improve interoperability through the Cures Act. For example, Draft 2 of the Trusted Exchange Framework and Common Agreement, released in April 2019, “outlines a common set of principles, terms, and conditions to facilitate interoperability and information exchange across disparate HIE platforms and help enable the nationwide exchange of electronic health information” (NASEM, 2019, p. 208). When fully implemented, the framework should help achieve seamless access to information across different HIT platforms and health care systems (Rucker, 2018).
Recent mandates to use the Substitutable Medical Applications and Reusable Technologies on Fast Health Interoperability Resources platform may help with data sharing. It is designed to enable medical applications to run unmodified across different HIT systems (Mandel et al., 2016), providing a common format for sharing health data in the EHR across applications and facilitating the flow of data across otherwise incompatible systems. Merely adopting these standards will not ensure success. How they are implemented will also be critical. Whether this translates into true interoperability for care-seekers and clinicians remains to be seen.
Usability
While technology can make it easier compared with paper records to review a medical history, make diagnoses, generate orders and prescriptions, and document treatment plans, poorly designed systems may introduce frustrating processes into the care delivery experience and even make the experience more difficult and error prone (NASEM, 2019). Health IT
and Patient Safety: Building Safer Systems for Better Care describes key attributes of safe digital health (IOM, 2011) (see Box 7-1). Most systems today do not meet these basic standards.
Taking Action Against Clinician Burnout documented many of the usability problems of current EHRs, particularly during a clinical encounter (NASEM, 2019). Poorly designed visual displays, for example, may contribute to prescribing errors (Moacdieh and Sarter, 2015; Ratwani et al., 2018a). Part of the reason for poor usability of EHRs is that the initial motivation behind their design and implementation was to help facilitate billing, reporting, and fulfilling regulatory requirements, not necessarily to improve clinical workflow. Thus, the user interface and standard menus often do not accurately reflect the uniqueness of individual clinical situations or allow the flexibility to adapt (NASEM, 2019). This, in turn, forces clinicians to make unnecessary clicks to move through the EHR, which is one factor contributing to clinician dissatisfaction (Friedberg et al., 2014). Primary care teams who care for children face additional challenges in the usability of EHRs (Ratwani et al., 2018b,c). Children require weight-based medication dosing and age-specific screenings and interventions. EHRs are often not designed with child health–focused usability, and thus errors in medication dosing and in missed or inappropriate care have occurred.
Usability may be even more important for patient-facing digital health systems. While it may be feasible to train clinicians to use an EHR or other digital health tool, patient-facing systems need to be “plug and play”: easy to access or download, intuitive to use, and understandable even for those with lower health literacies. Adding to this, some health information can be sensitive (e.g., self-reported substance use or sexual history) or
worrisome (e.g., a new diagnosis or abnormal results). While vendors for direct-to-consumer digital health products have paid attention to usability, the science around usability for the products that primary care clinicians share with those they serve is limited, with the available evidence focused more on individuals’ desired access to health information (Kerns et al., 2013). Studies have shown that people generally have trouble navigating patient portals, frequently make operational errors, and expect nonexistent functionalities (Baldwin et al., 2017; Yen et al., 2018).
Children, adolescents, and their parents are less likely to use patient portals for information or communication, compared to adults, in large part due to the inadequate usability of EHRs for this population (Sharko et al., 2018; Webber et al., 2019). There are no consistent or widely used standards adopted by EHRs for how (e.g., technical design for proxy access) or when (e.g., automated access offered at age 13) to grant adolescents independent access to their own data and an ability to communicate with the care team, and EHRs largely are not designed to allow care teams to filter sensitive versus non-sensitive data to allow both parents and teens to have access to the EHR simultaneously, without jeopardizing either teen confidentiality and privacy or parental access to important, non-sensitive health information (Society for Adolescent Health Medicine et al., 2014). The result is that many care teams are left with only the options of denying portal access to parents at a pre-specified child age or not using the portal at all once children reach adolescence. These challenges were further exposed during the COVID-19 pandemic (The Larry A. Green Center and PCC, 2020). Usability for patients will likely require a significant implementation element, including training on the system, more flexibility in privacy based on age and status (e.g., adolescents whose parents may have primary access to the portal), assistance and support as users encounter difficulties, and personnel and strategies to keep them engaged in meaningfully using the system to improve their health.
Lessons from the COVID-19 Pandemic
The COVID-19 pandemic forced rapid changes in digital health across all primary care throughout the nation. Within the span of weeks, many practices converted from almost all in-person care to near complete telehealth (Wosik et al., 2020), to reduce the spread of COVID-19 and protect care-seekers, staff, and clinicians while remaining connected to people and communities that continued to need medical care (Krist et al., 2020). However, COVID-19 has also revealed and amplified the growing breadth of the digital divide and added to health inequities (Woolf et al., 2020).
Three policy changes enabled this rapid transformation. First, CMS relaxed strict regulations about which telehealth platforms were HIPAA
compliant. This change allowed patients and clinicians to easily adopt tools, such as video calling, that they were already using in their work and personal lives, rather than requiring people to download complex applications or clinicians to purchase applications for use within EHRs. Second, CMS relaxed strict regulations regarding documentation of telehealth visits and rules about who could be seen by which doctors. This allowed clinicians to offer a telehealth visit to someone with whom they had an established relationship but who lived in or was traveling to another state. Third, payment for telehealth visits, including audio-only visits via telephone, was made equal to in-person visits—payment parity. Previously, many payers did not reimburse for telehealth visits at all; if they did, it was at a lower rate and excluded some visit types (e.g., wellness) (Verma, 2020). These policy changes have been transformative (Contreras et al., 2020; Mann et al., 2020) and some, but not all, were made permanent in late 2020 (CMS, 2020b). Combined with collective fears of catching and spreading COVID-19 and the need for everyone to practice social distancing, telehealth, which had limited low use, was widely and rapidly implemented. Adding allowances for telephone visits has provided remote access for those without smartphones, tablets, or computers, which are needed for video-based telehealth applications.
The U.S. telehealth COVID-19 experience highlights the demand for digital health innovations, the need for policy changes to better support digital health while ensuring digital health equity, and the potential for advancement if the health care technology marketplace can be more open. Assessing the global perspective in comparison to the U.S. response makes it evident that quick, responsive, and adequately applied digital health technology has enabled multiple countries to contain the spread of COVID-19 while leveraging a robust primary care and public health infrastructure. Many of these same countries continue to be front-runners in surveillance, testing, contract tracing, quarantine, individual clinical management and effectively managing the burden of COVID-19 across their populations (Whitelaw et al., 2020).
DESIGNING DIGITAL HEALTH FOR PRIMARY CARE: WHAT DOES SUCCESS LOOK LIKE?
Useful digital health systems for primary care need to support the core functions of primary care. An overarching principle is that systems should aggregate information and make that information usable by clinicians, patients, families, and community members to carry out the core functions of primary care, including promoting access to care, coordinating care, ensuring care is integrated across settings, and allowing for high-quality population health (see Figure 7-1). Based on the function and design of primary
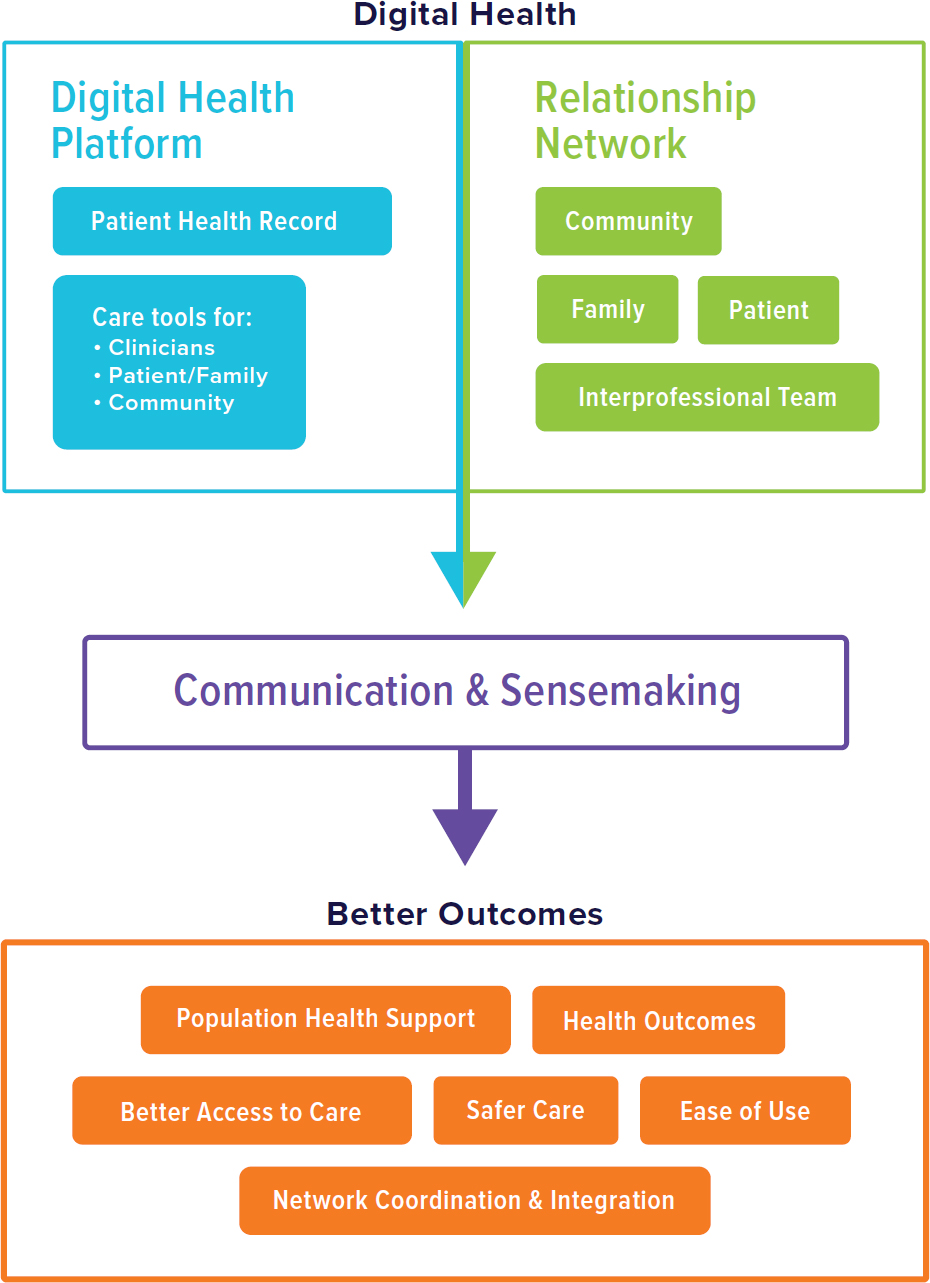
NOTES: Digital health needs to support relationships between members of the care team with patients, families, and the community to improve the essential components of high-quality primary care. These components include care access, coordination, integration, and safety and population health support. Digital health should be easy to use for all team members and care-seekers.
care articulated in this chapter, effective digital health systems will need to (1) support relationships; (2) support high-functioning interprofessional teams to engage in sensemaking, decision making, and action; (3) integrate care delivery across systems and communities; (4) reduce workload; and (5) make care more equitable. Both setting up digital health and using digital health to carry out these functions must occur in a way to reduce workload on primary care and health systems.
Supporting Relationships
Health care is fundamentally relational. This is especially true in primary care, as individuals, families, care teams, and communities collaborate to co-create care plans that evolve over time (Finley et al., 2013). Relationships provide the platform from which all care activities occur, as positive relationships support effective communication. Traditionally, individual–interprofessional team interactions have been episodic and dependent on the person making a burdensome trip to a clinic setting for care and to “engage” in the relationship. Digital health presents an opportunity to increase the frequency and depth of interactions in a manner that is convenient and empowering for the person seeking care and integrates care across health care systems and within communities (Lanham et al., 2016). These increased contacts, in turn, support relationships with patients and care team members across the care and community continuums and improve the quality of care (Lanham et al., 2009). Digital tools are not meant to replace face-to-face visits with a care team but rather to enhance interactions, so that in-person visits become richer in meaning, with deeper interactions resulting from the frequency and level of communication being supported by digital tools. Patient portals, telehealth platforms, health apps, remote monitoring devices, integrated EHRs, and other technologies already exist that can enhance and develop deep interpersonal relationships with the care team.
Person-centered digital health will also enable care to be more transparent, providing information that is understandable and appropriate to the person’s unique circumstances. This will promote trust and relationships between patients and their interprofessional care teams, increasing the likelihood of effectively co-created care plans, consistent engagement with the health care system, engagement with community resources, and improvement in health outcomes.
Digital health may also have a unique benefit to adolescents. It provides another avenue for them to build a confidential, trusting relationship with a clinician and care team and a venue in which they can access that team without relying on a parent or guardian to grant access via transportation or even permission. Virtual visits may also be perceived as more confidential
than an in-person visit where the parent steps out of the room but is still within earshot.
Supporting High-Functioning Interprofessional Teams
The path from relationships and communication to care plan co-creation and subsequent action requires partnership between interprofessional teams and care-seekers to make sense of information and create mental models that are shared across the relationship network. This process is active—it unfolds over time and is constantly updated (Leykum et al., 2015). Digital health can support the ability of interprofessional teams to make sense of health data. In fact, given the vast amounts of information available about not only each person and their context but an ever-growing evidence base related to health and disease, digital health has become a necessary adjunct and support for teams’ effective decision making. The prodigious literature on adverse events, missed diagnoses, and health disparities demonstrates the negative consequences of ineffective sensemaking, and digital health can prevent poor outcomes through several mechanisms (Jordan et al., 2009).
At the individual level, improved safety and harm reduction can happen at the point of care through alerts, reminders, and the prevention of repeat and unnecessary testing, as well as by supporting effective and timely communication among and between care-seekers and the interprofessional team. Digital health provides a common information platform for interprofessional teams, and digital health tools promote effective use of that information by patients and teams to make the most appropriate and effective decisions. At the population level, improved safety and harm reduction can occur by identifying people at risk for poor outcomes. While alerts and registries are already available, the ability to include patients and caregivers, incorporate information across organizational networks, and customize tools to meet local needs and workflows would increase their effectiveness and usefulness.
Digital health tools can also improve health outcomes by promoting prevention, facilitating good health behaviors, improving diagnostic capability, and promoting evidence-based care, again at the individual and population levels. These include the decision support and artificial intelligence tools that are already being implemented to support interprofessional teams and community partners and sometimes also patient monitoring, engagement, and partnership tools that include activity app integration and virtual support communities.
As discussed in Taking Action Against Clinician Burnout, HIT (i.e., digital health tools) needs to be optimized to support the clinician (and health team) in providing high-quality care (NASEM, 2019). A properly developed
and organized digital health infrastructure will support high-quality care by creating conditions that will nurture deep patient relationships, limit redundant data, optimize data analysis for care and population health, and assist in data interpretation for both the clinician/team and the patient, all while forecasting needs and reducing clinician burnout. Recommendation 4C from Taking Action Against Clinician Burnout notes the following:
This would be an electronic interface that gives the entire care team, including the patient, the ability to collect and use timely and accurate data to achieve high-quality care. A major goal of this new health information system should be to allow clinicians to focus on optimizing patient and population health, while adjuvant processes and technologies derive, to the extent possible, the essential business, administrative, and research data necessary to deliver high-value care efficiently and effectively. (NASEM, 2019, p. 17)
Supporting Integrated Care Delivery
Effective relationships in primary care need to encompass not only the care-seeker, family, and clinician but also potentially other caregivers, the full interprofessional care team, and the community in which care occurs, in an integrated fashion. This integration creates a network around each patient, one that is involved in care plan co-creation and enactment. Integrating care this way occurs in a local context that shapes that care and is shaped by the needs of individuals in the community.
Digital health can support this network by (1) integrating the individuals and organizations in the network and (2) supporting the ties between them. This linkage relies on accurate information transfer and creating a common information platform that enables decision making by the individual and care team. Digital health can ensure that everyone is working with the same set of information to create shared mental models, which requires that information be equally accessible to everyone, when desired by the care-seeker, and it must authentically reflect each patient. Additionally, digital health can support integration across health care systems and communities by facilitating communication that meets a wide spectrum of needs. Some communication, for example, is urgent and requires interpretation, such as that regarding a new, concerning symptom. This scenario requires real-time, person-to-person, direct communication. In other instances, time sensitivity or immediate interpretation may not be necessary, such as notifying about the need for annual preventive care. For this situation, asynchronous distributed information transfer is sufficient. Digital health needs to be nimble enough to support communication and integration across all aspects of the communication spectrum.
Digital health is designed to be person centered and to help individuals get the right care at the right time and can promote integrated care, but only if the systems are easy for people and their families to access and use. A portal that requires downloading software or using specific programs or operating systems will not facilitate information integration. Digital health tools should facilitate the right care at the right time. Tools such as patient portals that help people access integrated information have five essential features. They should (1) link to existing and comprehensive clinical information, (2) allow patients and families to enter information that only they know (values, preferences, behaviors, goals), (3) present content in lay language and the preferred language, (4) interpret content by applying health information to guidelines to say what it means, and (5) make information actionable to allow people to get care and make changes to improve health and well-being (Krist and Woolf, 2011).
There are benefits to standardizing both the function and content of digital health systems to promote integration. For data to be shared across systems, a common data architecture and nomenclature are necessary. Additionally, standardization can help to promote desired care. Alerts, reminders, templates, order entry systems, and educational tools can all be linked to evidence-based guidelines to promote recommended care and deter unnecessary or harmful care.
However, locally tailoring digital health systems is also necessary to support integration in local contexts with the specific interprofessional team and community resources that best partner with each care-seeker. Different populations may have different needs, and different communities may have different resources to deliver care. Each primary care practice will have different workflows depending on staff and skills, and practices engaging in quality improvement will want to use their digital health systems to innovate and redesign care. Having the flexibility to use their systems in novel ways will be essential to continually advance and improve an integrated practice. Finally, digital health provides the means to develop technical support so that someone can take a proactive role in their own health management and care integration, whether this involves medication adherence, preventive screening tests, lifestyle changes, community resources, understanding of chronic conditions and how to manage them, or recognition of when to seek care for a change in their health status.
Making Care More Equitable
As made clear by patients’ experiences using telehealth to access care during the COVID-19 pandemic, digital health can increase existing disparities if it is not implemented intentionally to address barriers related to lack of community trust, language needs, Internet access, e-mail use,
device capabilities, and an individual’s comfort with using digital health and electronic communication platforms (NASEM, 2016; Nouri et al., 2020; Whitelaw et al., 2020). At the same time, digital health represents an opportunity to actively address long-entrenched inequities to assist marginalized populations in achieving more equitable health outcomes. High-quality primary care can use digital health tools to create more equitable care via three main pathways: (1) improved communication, (2) increased access, and (3) reduced disparities in clinical practice.
Improved communication can be achieved through community outreach in conjunction with applying an understanding of local culture to modify digital health interfaces and access points so that digital health can meet the information needs of a marginalized and underserved community (NASEM, 2016). However, simply offering online access to information through a patient portal in a person’s native language does nothing to bridge the growing digital divide. Until systemic inequalities and discrimination related to jobs, housing, education, and access to resources are resolved, improving communication with underserved communities will require unique solutions that provide affordable access to high-speed Internet connections and high-impact digital tools. The crux of the problem then becomes how to reduce disparities in digital access and digital health literacy in a manner that will enable culturally appropriate communication, outreach, and education through community partnerships with interprofessional teams to improve access to care for marginalized and underserved communities.
In terms of increased access, a “digital dilemma” now clearly exists in which improved communication and access to digital health resources (i.e., telemedicine) relies on physical access to digital and technological infrastructure. Without that access—whether that involves the Internet, computers, or mobile phone technology—a growing digital divide in access to digital health care and communications will persist. To achieve digital health equity, the “digital dilemma” must be solved. Only then can digital health tools be fully leveraged to improve communications, increase access to care, and reduce health care disparities.
In terms of reducing health care disparities in clinical practice, access to care and culturally appropriate communication are essential starting points. Digital health tools can be used to aggregate and analyze collected information to personalize communication and increase access to care (NASEM, 2016). However, it can also be used to go one step farther—to improve clinical outcomes and reduce health disparities by decreasing implicit bias and improving clinical care (Lau et al., 2015). Theoretically, EHRs and digital health technology can be a great tool for eliminating health care disparities and ensuring equal treatment despite race, gender, or socioeconomic differences. A patient’s race, in particular, has been demonstrated as
a predictor of health care quality and outcomes in the United States, due to institutional and systemic racism, and ample evidence shows that unconscious or implicit bias among clinicians may influence clinical decision making and lead to disparities in outcomes. Digital health tools represent a unique opportunity to leverage technology to reduce bias, improve clinical decision making, and increase equity in clinical care, if the appropriate policies are implemented to compel stakeholders to leverage digital health tools to reduce health care disparities.
NEEDED FUNCTIONS OF PRIMARY CARE DIGITAL HEALTH SYSTEMS TO ACHIEVE VISION OF SUCCESS
Primary care has a unique need for the most comprehensive access to patients’ health information. Starfield and colleagues (2005) described primary care as needing to provide the four Cs—first contact, comprehensive care, coordinated care, and continuous care. Adding to these roles, primary care is the only function in health care responsible for all aspects of a person’s health. Succeeding at these tasks depends on comprehensive information. Accordingly, a key responsibility for primary care clinicians is to collect and aggregate health information. Both Meaningful Use certification and the patient-centered medical home recognition mandated them to collect, enter, and manage patient health information within EHR information systems that could then be freely used by all clinicians (Blumenthal and Tavenner, 2010; NCQA, 2020). This is a tremendous burden on primary care that places primary care clinicians in more of an administrative role, detracting from truly helping patients. Additionally, given the volume and breadth of necessary data to inform care, automated tools are needed to make sense of data, identify clinically important data, and improve care. More than any specialty, primary care needs for this information aggregation and analysis to be automated. The current digital health environment makes this an impossible task.
The overarching functions of digital health for primary care include (1) collecting health information (creating the platform), (2) aggregating and making sense of health information to create a complete health record and highlight critical health information, and (3) applying health information to improve health in ways that promote person-centered care, support care teams, span settings of care, and generally make life easier (see Figure 7-2).
Systems that collect health information can be patient facing, clinician facing, automated, or any combination of these. Patient-facing systems allow patients to report information that only they know, such as health behaviors, mood, feelings, quality of life, self-reported outcomes, and goals, and aggregate this information. These systems can include patient portals, smartphone applications, and Web-based surveys. Clinician-facing systems
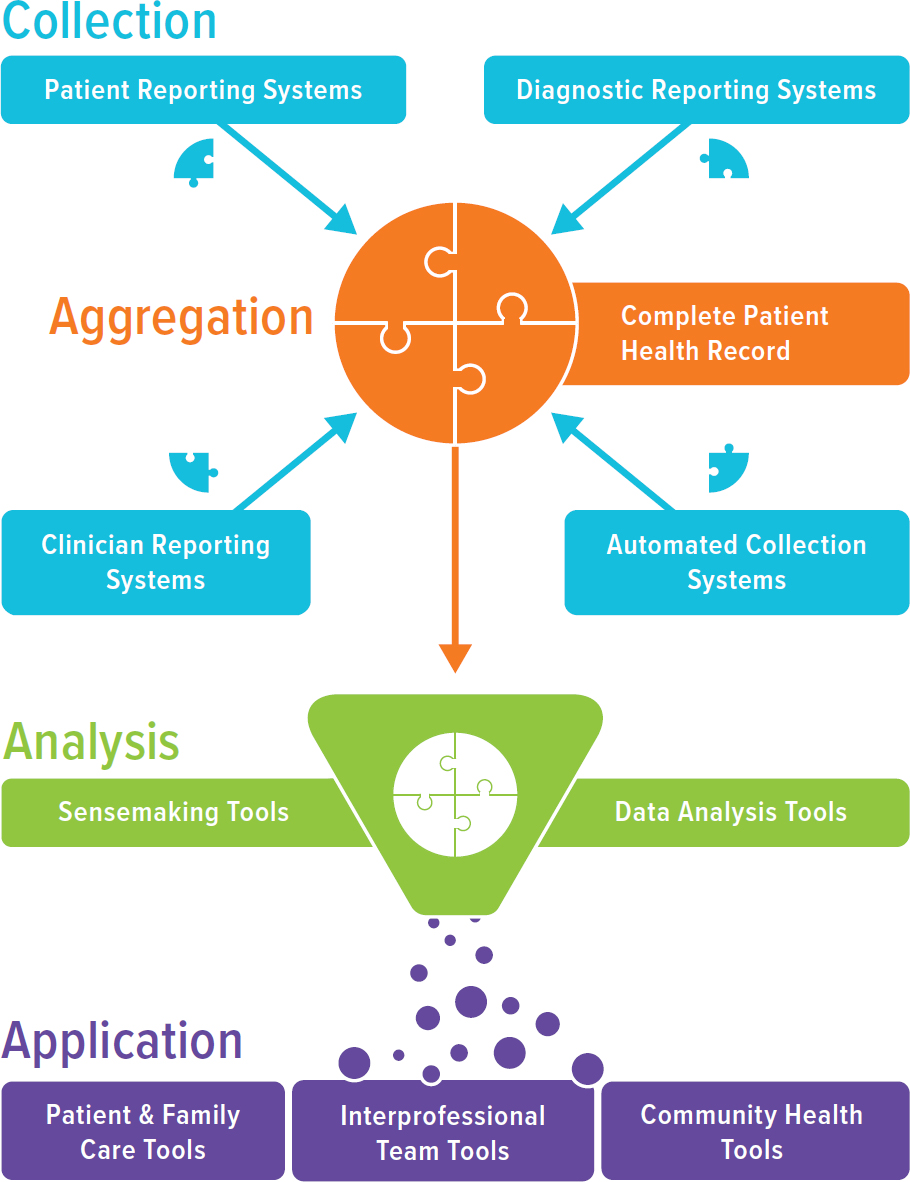
NOTES: From a primary care perspective, digital health helps clinicians with collecting information, aggregating and analyzing information, and applying information to decision making and clinical care. Multiple digital health tools can collect information from different audiences (patients, clinicians, diagnostic tests, and automated tools, such as wearables). Once information is aggregated, automated systems are needed to analyze the information to make it usable by patients, families, and care teams.
allow the interprofessional care team to enter information such as medical history, exam findings, diagnoses, treatments, and care plans; the classic such system is the EHR. Automated systems, not currently in routine use, include any system that can collect and aggregate important health-related information, such as about biometrics, behaviors, environment, or exposures, without effort from the clinician, staff, or person seeking care. These automated systems may be important sources of community and contextual information. In the future, automated systems may even act as “scribes,” collecting and documenting clinician–patient interactions and allowing clinicians to truly focus on the person. Multiple collection systems can be used simultaneously, and future versions may even replace aspects of existing clinician and patient-facing systems to automate data collection.
Once collected, information needs to be aggregated and analyzed, creating the information platform that patients, families, clinicians, and communities can use to make sense of what is happening and take action. Both functions need to be automated and not dependent on clinicians, staff, or care-seekers to collect, re-enter, or analyze. The information must be comprehensive and not siloed, and while it could be distributed (i.e., stored in multiple systems), it should be connected to yield an immediately comprehensive and complete record when needed. Patients must be in control of who has access to which elements of their information and when. Supporting the comprehensive information, analytics and sensemaking tools are required to sort information in ways that are valuable to users. This is particularly important as these systems incorporate more raw data (e.g., daily weights, smart watch measurements, and environmental data). Tools are needed to identify clinically meaningful data, overdue care, and potential safety issues, aid in diagnosis and care delivery, and even inform population health activities. Existing tools include alerts and reminders, drug interactions, quality measurements, and patient registries, though these are just a starting point if digital health is to truly aid clinicians and individuals in providing and accessing care.
Ultimately, the purpose of digital health is to help patients, families, clinicians, and care teams to improve health. The same tools that collect health information should enable all involved parties to access health information through analytics and tools that promote the ability of patients, families, care teams, and communities to make sense of the available information and take action to improve health. This includes being able to make diagnoses, see what care is needed, deliver care, communicate among team members, coordinate care, and track progress.
To meet these collective needs, the committee has identified the following high-level primary care digital health functions required for success:
Collect Information
- Complete information. Primary care needs systems to collect information from all health care sources (primary care, specialists, community providers, and care-seekers) and non–health care sources that affect health (environmental data, social descriptors) and include all potential settings (inpatient, outpatient, and communities). Nontraditional health care team members will need access to and will generate health information.
- Automate information collection. Information collection should not depend on primary care clinicians entering or reconciling data; it needs intelligent automation.
- Ownership of information. People need to own their health data and be able to grant care team members access to their information. As children age into adolescents, they should become owners of their health information, independent of the parent or guardian. Similarly, if primary care clinicians spend 6 hours per day documenting care, they own those data—not the health system or EHR vendor. Once generated, clinicians should have indefinite access to the data they created.
Aggregate and Analyze Information
- Create a comprehensive record. Primary care needs a comprehensive record that includes all individual health information but is not responsible for creating it; rather, digital health should be a resource that supports primary care. Comprehensive information could exist in distributed sources, simultaneously and seamlessly accessed.
- True interoperability. To aggregate health information and create a comprehensive record, primary care needs digital health systems to be functionally interoperable. The requirements for digital health systems should measure “lived interoperability,” not whether systems can theoretically be interoperable. The metric of success for lived interoperability is the daily transfer of health information from one system to another and the amount of data that fails to transfer in a completely automated way.
- Information sensemaking. For a comprehensive health record to be useful, tools such as artificial intelligence are needed to parse relevant data, understand implications and interrelationships of data, and aid decision making and health promotion.
Apply Information
- Engage care-seekers in action. Digital health systems should engage and activate individual patients and populations in their care by translating medical content into lay language, allowing patients to clearly state and communicate their goals of care and providing them with logic, educational support, and tools to facilitate their action. Systems need to work with a broad range of audiences with diverse needs and account for patient confidentiality and privacy.
- Promote evidence-based care and safety. Digital health systems need to promote national quality and safety standards and to include and make usable the most up-to-date national guidelines and quality measures.
- Make care proactive. Registries, alerts, reminders, and other population health tools are needed to identify and target persons who require care.
- Automate more care. Technology disruption is needed to automate some elements of care delivery through artificial intelligence, chat bots and avatars, and ambient computing.
- Coordinate care teams. Integrated communication tools can help teams to better coordinate around care.
- Allow local tailoring. Not all patients, practices, and communities are the same. Local adaptation is necessary to accommodate variations in their needs, workflows, and resources.
Payment and care models also need to change, as described in Chapter 9. Changing digital health without providing resources for primary care to carry out these functions would not result in change and even exacerbate clinician burnout.
HEALTH DISPARITIES AND DIGITAL HEALTH
While digital health is transforming the health care landscape, it is not immune to the pervasive systemic inequalities that have contributed to long-entrenched health care disparities. The COVID-19 pandemic has shed light on these disparities, which have been further amplified by the policy response to the crisis, exacerbating the entrenched inequities in the U.S. health care system, in general, and the primary care system, in particular. The pandemic also showcased how digital health initiatives can further escalate socioeconomic, racial, and geographic inequalities that directly influence health care disparities (Nouri et al., 2020; Woolf et al., 2020). As telehealth rapidly expands and ensures access to care even when
primary care practices are closed and people are self-isolating, many older Americans, low-income families, rural communities, and racial and ethnic minorities are unable to access care and suffering from the consequences of delayed treatment (Hirko et al., 2020; Kim et al., 2020; Nouri et al., 2020; Verma, 2020).
In October 2014, the National Academies of Sciences, Engineering, and Medicine held a workshop (Promotion of Health Equity and the Elimination of Health Disparities) (NASEM, 2016) focused on reviewing examples and models of digital health technologies to improve health outcomes for underserved populations. Key themes that emerged include the importance of community engagement to adopt digital health tools; leveraging mobile technology to reach underserved populations; the impact of infrastructure and systemic inequities on the access to these technologies; and the marginalization of minority communities by the current market forces driving digital health innovation. Box 7-2 shows one example of the type of local approach and resources needed to operationalize these themes.
If policies impacting digital health are not changed to create equitable access and outcomes, the nation will be left facing unnecessary and
premature deaths within its most vulnerable populations. Additionally, without stronger accountability and oversight, the U.S. health care system will continue to provide the most expensive and advanced digital health technology to those who can afford it rather than to those who would most benefit.
DIGITAL HEALTH IMPLEMENTATION NEEDS
This chapter presents a bold future vision for how digital health can support primary care. Achieving this vision requires (1) buy-in from clinicians and care-seekers who use digital health systems, (2) support from practice and health system informaticists who field these systems, (3) a willingness on the part of digital health vendors to transform their systems, (4) disruption of the existing centralized marketplace to allow for innovation, (5) new authorities and policies to enforce digital health meets standards, and (6) policy makers’ commitment to implement rules, regulations, and metrics that assess the lived experiences of digital health users. For successful implementation, all requirements must be satisfied concurrently and satisfying requirements cannot add burden to primary care. Failure from any sector will result in continuing with the status quo.
As the end users, clinicians and those seeking care need to demand digital health tools that meet their needs. While they have the least power to effect change, they suffer the most when digital health fails to meet their needs and bear the greatest burden when changing from one digital health system to another. Accordingly, they must be protected throughout the transformation process. Practice and health system informaticists often decide about digital health systems to adopt; many of them are not clinicians, and few are routine end users. These decision makers need to understand the lived experience of those they serve.
While Meaningful Use incentives stimulated the national adoption of EHRs, it also consolidated the market, creating powerful, resourced, and established vendors. Rather than merely adding code or database architecture to their existing systems or acquiring and integrating a new system, vendors need to move past the 1990s and 2000s and create new systems. To support this, the digital health marketplace needs to be a free marketplace that supports innovation. Clinicians and health systems should not be bound to existing systems because transitioning is prohibitively labor intensive, resulting in losing data that they spent years entering. The linchpin to transforming the marketplace and promoting innovation will be true interoperability, not the checkbox interoperability that exists today.
To achieve true interoperability, there must be a common health information database available to all health care clinicians to support data sharing while ensuring local control of the data to ensure security and
meet privacy requirements. One option is a centralized national medical database. This model has been successfully deployed in other countries, as exemplified by the Historia Clínica Digital del Sistema Nacional de Salud (the National Health System Electronic Health Records Project) in Spain (Huerta et al., n.d.). Another option that would optimally support high-quality primary care is a distributed data network capable of transferring information between EHR databases in response to health information queries that can provide aggregated data to the end user or data requestor. In 2010, the Agency for Healthcare Research and Quality published a report providing a blueprint for a distributed research network to conduct population studies and safety surveillance (Brown et al., 2010). The report highlighted a distributed architecture, scalability, query distribution, data holder autonomy, and privacy protection as key attributes that would be needed to successfully implement a centralized distributed data network to support health information. This model can be adapted nationwide to create a digital health backbone that supports delivering high-quality primary care to people, families, and communities who ultimately retain control of their own data. An obvious first step would be to create a digitally encoded card with individual health data for those with state and federally funded health insurance coverage that would allow secure transfer, queries, and analysis. If successful, the concept can be expanded broadly to all people regardless of insurance type. However, to build the distributed database of the future, federal government support is required, including new legislation authorizing its creation, regulatory oversight, and funding to design and support the system.
Policy makers are well positioned to catalyze and ensure that the needed transformation of digital health occurs, although they need greater authority over digital health vendors to ensure their systems meet requirements. Doing so requires fundamentally prioritizing patients and clinicians over existing businesses and focusing on creating the next generation of measures and standards that track and assess users’ lived experience, not merely digital health’s potential to meet standards in an ideal and even theoretical way. The next phase of digital health standards should focus on measuring in real time the transfer of data, the integrity of data, how often data need manual reconciliation, the number of clicks to perform tasks, data entry time, and use of the system by clinicians and patients that can be directly linked to improved health and well-being.
DIGITAL HEALTH RESEARCH NEEDS
Evidence should guide digital health development and use for primary care. Both digital health functionality and implementation need to be studied using improvements in the quadruple aim as the desired outcome
(Bodenheimer and Sinsky, 2014). Digital health applications need to show they enhance the care experience, improve population health, reduce costs, achieve equitable outcomes, and improve the work life of clinicians and staff.
While relationships, interprofessional care teams, comprehensive care, and health equity are critical elements of effective primary care, the direct evidence about how digital health can best support these functions is still evolving. This understanding will require work that goes beyond the current examination of clinician use of features or decision support tools, or individual logins to health portals or use of secure messages, and a rich, mixed methods, ethnographic research agenda that engages patients, families, interprofessional care teams, and community partners to understand optimal use over time. This work needs to be in partnership with groups engaged in developing digital health tools to ensure that they create the most effective tools.
Disruptive digital health advances that transform aspects of future health care delivery are inevitable. Innovations using artificial intelligence and avatars can automate care. Ambient computing can collect, aggregate, and analyze information. New unforeseen technologies will bring advances to diagnosis, treatment, and delivery of care. Research is needed to develop these disruptive technologies, to assess their impact on health outcomes, to evaluate them for unforeseen complications, and to determine how to implement and integrate them into future care delivery models.
FINDINGS AND CONCLUSIONS
Digital health, particularly EHRs that serve as the hub of patient information, is an essential tool to improve systems of care. It is also the major source of professional dissatisfaction and clinician burnout (NASEM, 2019). The committee supports three major informatics changes needed to advance digital health for primary care—changes to the marketplace, aggregated comprehensive patient data, and new federal standards to drive meaningful change. This chapter describes the principles needed for these changes.
The current dominance of the market by a few informatics vendors coupled with limited interoperability has locked clinicians and practices into existing systems and stifled innovation. Switching from one EHR to another is a tremendous effort and sacrifices essential data. While many EHRs technically meet interoperability standards, they are not functionally interoperable. Because the privatization and monetization of health information is how vendors maintain the market share of their products, they are not incented to be truly interoperable. Vendor policies, inconsistent data
storage and architecture, and limited mechanisms for efficient data transfer all contribute to limited interoperability.
A key action that will change the marketplace, catalyze innovation, and advance care is to create a national comprehensive and aggregated patient data system, which would enable primary care clinicians, interprofessional teams, patients, and families to easily access the comprehensive data needed to provide whole-person care. It could be used by any certified digital health vendor to create innovations, and patients could control who has access to their health information. There are several ways this could be achieved. It could be set up as either a centralized data warehouse or individual health card or distributed sources connected by a real-time functional HIE. Access to centralized comprehensive data would represent an essential innovation for primary care teams responsible for whole-person care. It shifts the burden that national quality metrics and performance payments currently impose on primary care clinicians to manually enter patient information to create a comprehensive record, placing it on an automated system that would allow clinicians and teams to focus on care. The committee recognizes that these changes will require innovation from vendors and state and national support agencies and that accomplishing these goals will not be easy to ascertain. However, this is an essential need.
The committee supports federal standards setting for this field but has determined that the past Meaningful Use requirements inadequately met the needs of primary care and unacceptably put the burden of meeting the requirements onto primary care. A new phase of federal standards is needed to ensure that HIT aligns with primary care functions, makes it easy to deliver the right care at the right time, is designed to support equitable access, can help clinicians make sense of complex information, and fundamentally reduces clinician and patient workload. The lived experience of clinicians and care-seekers should be measured and used to assess whether HIT is meeting expected standards, not the theoretical ability of systems, as previously done. Vendors and state and national support agencies should be charged with designing base digital health systems to meet these requirements, and should be held accountable when systems fail to meet benchmarks.
REFERENCES
Adler-Milstein, J., D. W. Bates, and A. K. Jha. 2013. Operational health information exchanges show substantial growth, but long-term funding remains a concern. Health Affairs 32(8):1486–1492.
Alagiakrishnan, K., P. Wilson, C. A. Sadowski, D. Rolfson, M. Ballermann, A. Ausford, K. Vermeer, K. Mohindra, J. Romney, and R. S. Hayward. 2016. Physicians’ use of computerized clinical decision supports to improve medication management in the elderly—the Seniors Medication Alert and Review technology intervention. Clinical Interventions in Aging 11(1):73–81.
AMA (American Medical Association). 2015. Panel management: Provide preventative care and improve patient health. https://edhub.ama-assn.org/steps-forward/module/2702192 (accessed May 6, 2020).
AMA. 2016. Point-of care registries: Proactively manage chronic care conditions. https://edhub.ama-assn.org/steps-forward/module/2702745 (accessed May 6, 2020).
AMA. 2020. AMA digital health research: Physicians’ motivations and requirements for adopting digital health and attitudinal shifts from 2016–2019. Chicago, IL: American Medical Association.
Arndt, B. G., J. W. Beasley, M. D. Watkinson, J. L. Temte, W. J. Tuan, C. A. Sinsky, and V. J. Gilchrist. 2017. Tethered to the EHR: Primary care physician workload assessment using EHR event log data and time-motion observations. Annals of Family Medicine 15(5):419–426.
Baldwin, J. L., H. Singh, D. F. Sittig, and T. D. Giardina. 2017. Patient portals and health apps: Pitfalls, promises, and what one might learn from the other symptoms. Healthcare 5(3):81–85.
Barnett, M. L., K. N. Ray, J. Souza, and A. Mehrotra. 2018. Trends in telemedicine use in a large commercially insured population, 2005–2017. JAMA 320(20):2147–2149.
Blumenthal, D. 2009. Stimulating the adoption of health information technology. New England Journal of Medicine 360(15):1477–1479.
Blumenthal, D., and M. Tavenner. 2010. The “Meaningful Use” regulation for electronic health records. New England Journal of Medicine 363(6):501–504.
Bodenheimer, T., and C. Sinsky. 2014. From triple to quadruple aim: Care of the patient requires care of the provider. Annals of Family Medicine 12(6):573–576.
Bright, T. J., A. Wong, R. Dhurjati, E. Bristow, L. Bastian, R. R. Coeytaux, G. Samsa, V. Hasselblad, J. W. Williams, M. D. Musty, L. Wing, A. S. Kendrick, G. D. Sanders, and D. Lobach. 2012. Effect of clinical decision-support systems: A systematic review. Annals of Internal Medicine 157(1):29–43.
Brown, J., B. Syat, K. Lane, and R. Platt. 2010. Blueprint for a distributed research network to conduct population studies and safety surveillance. Rockville, MD: Agency for Healthcare Research and Quality.
CDC (Centers for Disease Control and Prevention). 2019. Public health and promoting interoperability programs. https://www.cdc.gov/ehrmeaningfuluse/introduction.html (accessed May 7, 2020).
CMS (Centers for Medicare & Medicaid Services). 2020a. Promoting interoperability. https://www.cms.gov/Regulations-and-Guidance/Legislation/EHRIncentivePrograms/Stage3Medicaid_Require (accessed May 6, 2020).
CMS. 2020b. Trump administration finalizes permanent expansion of Medicare telehealth services and improved payment for time doctors spend with patients. https://www.cms.gov/newsroom/press-releases/trump-administration-finalizes-permanent-expansion-medicaretelehealth-services-and-improved-payment (accessed January 27, 2021).
Colicchio, T. K., J. J. Cimino, and G. Del Fiol. 2019. Unintended consequences of nationwide electronic health record adoption: Challenges and opportunities in the post-Meaningful Use era. Journal of Medical Internet Research 21(6):e13313.
Contreras, C. M., G. A. Metzger, J. D. Beane, P. H. Dedhia, A. Ejaz, and T. M. Pawlik. 2020. Telemedicine: Patient–provider clinical engagement during the COVID-19 pandemic and beyond. Journal of Gastrointestinal Surgery 24(7):1692–1697.
DeVoe, J. E., A. W. Bazemore, E. K. Cottrell, S. Likumahuwa-Ackman, J. Grandmont, N. Spach, and R. Gold. 2016. Perspectives in primary care: A conceptual framework and path for integrating social determinants of health into primary care practice. Annals of Family Medicine 14(2):104–108.
Dinh-Le, C., R. Chuang, S. Chokshi, and D. Mann. 2019. Wearable health technology and electronic health record integration: Scoping review and future directions. JMIR mHealth uHealth 7(9):e12861.
Fernald, D. H., R. Wearner, and W. P. Dickinson. 2013. The journey of primary care practices to Meaningful Use: A Colorado Beacon Consortium study. Journal of the American Board of Family Medicine 26(5):603–611.
Finley, E. P., J. A. Pugh, H. J. Lanham, L. K. Leykum, J. Cornell, P. Veerapaneni, and M. L. Parchman. 2013. Relationship quality and patient-assessed quality of care in VA primary care clinics: Development and validation of the work relationships scale. Annals of Family Medicine 11(6):543–549.
Fisher, E. S., J. P. Bynum, and J. S. Skinner. 2009. Slowing the growth of health care costs—lessons from regional variation. New England Journal of Medicine 360(9):849–852.
Fleming, N. S., S. D. Culler, R. McCorkle, E. R. Becker, and D. J. Ballard. 2011. The financial and nonfinancial costs of implementing electronic health records in primary care practices. Health Affairs 30(3):481–489.
Friedberg, M. W., P. G. Chen, K. R. Van Busum, F. Aunon, C. Pham, J. Caloyeras, S. Mattke, E. Pitchforth, D. D. Quigley, R. H. Brook, F. J. Crosson, and M. Tutty. 2014. Factors affecting physician professional satisfaction and their implications for patient care, health systems, and health policy. RAND Health Quarterly 3(4):1.
Ginossar, T., and S. Nelson. 2010a. La Comunidad Habla: Using internet community-based information interventions to increase empowerment and access to health care of low income Latino/a immigrants. Communication Education 59(3):328–343.
Ginossar, T., and S. Nelson. 2010b. Reducing the health and digital divides: A model for using community-based participatory research approach to e-health interventions in low-income Hispanic communities. Journal of Computer-Mediated Communication 15(4):530–551.
HealthIT. 2020. Interoperability. https://www.healthit.gov/topic/interoperability (accessed January, 26 2020).
Hirko, K. A., J. M. Kerver, S. Ford, C. Szafranski, J. Beckett, C. Kitchen, and A. L. Wendling. 2020. Telehealth in response to the COVID-19 pandemic: Implications for rural health disparities. Journal of the American Medical Informatics Association 27(11):1816–1818.
Holmgren, A. J., N. L. Downing, D. W. Bates, T. D. Shanafelt, A. Milstein, C. D. Sharp, D. M. Cutler, R. S. Huckman, and K. A. Schulman. 2020. Assessment of electronic health record use between U.S. and non-U.S. health systems. JAMA Internal Medicine. https://doi.org/10.1001/jamainternmed.2020.7071 (accessed December 29, 2020).
Hoonakker, P. L. T., P. Carayon, and R. S. Cartmill. 2017. The impact of secure messaging on workflow in primary care: Results of a multiple-case, multiple-method study. International Journal of Medical Informatics 100:63–76.
Huerta, J. E., C. A. Villar, M. C. Fernández, G. M. Cuenca, and I. A. Acebedo. n.d. NHS electronic health record system. Madrid: Health Information Institute.
Hughes, L. S., R. L. Phillips, Jr., J. E. DeVoe, and A. W. Bazemore. 2016. Community vital signs: Taking the pulse of the community while caring for patients. Journal of the American Board of Family Practice 29(3):419–422.
Hussain, M. I., T. L. Reynolds, and K. Zheng. 2019. Medication safety alert fatigue may be reduced via interaction design and clinical role tailoring: A systematic review. Journal of the American Medical Informatics Association 26(10):1141–1149.
IOM (Institue of Medicine). 1996a. Primary care: America’s health in a new era. Washington, DC: National Academy Press.
IOM. 1996b. Telemedicine: A guide to assessing telecommunications in health care. Washington, DC: National Academy Press.
IOM. 2011. Health IT and patient safety: Building safer systems for better care. Washington, DC: The National Academies Press.
Jordan, M. E., H. J. Lanham, B. F. Crabtree, P. A. Nutting, W. L. Miller, K. C. Stange, and R. R. McDaniel, Jr. 2009. The role of conversation in health care interventions: Enabling sensemaking and learning. Implementation Science 4:15.
Kerns, J. W., A. H. Krist, D. R. Longo, A. J. Kuzel, and S. H. Woolf. 2013. How patients want to engage with their personal health record: A qualitative study. BMJ Open 3(7):e002931.
Kim, J. H., E. Desai, and M. B. Cole. 2020. How the rapid shift to telehealth leaves many community health centers behind during the COVID-19 pandemic. https://www.healthaffairs.org/do/10.1377/hblog20200529.449762/full (accessed December 2, 2020).
Krist, A. H., and S. H. Woolf. 2011. A vision for patient-centered health information systems. JAMA 305(3):300–301.
Krist, A. H., J. W. Beasley, J. C. Crosson, D. C. Kibbe, M. S. Klinkman, C. U. Lehmann, C. H. Fox, J. M. Mitchell, J. W. Mold, W. D. Pace, K. A. Peterson, R. L. Phillips, R. Post, J. Puro, M. Raddock, R. Simkus, and S. E. Waldren. 2014. Electronic health record functionality needed to better support primary care. Journal of the American Medical Informatics Association 21(5):764–771.
Krist, A. H., J. E. DeVoe, A. Cheng, T. Ehrlich, and S. M. Jones. 2020. Redesigning primary care to address the COVID-19 pandemic in the midst of the pandemic. Annals of Family Medicine 18(4):349–354.
Kruse, C. S., and A. Beane. 2018. Health information technology continues to show positive effect on medical outcomes: Systematic review. Journal of Medical Internet Research 20(2):e41.
Landi, H. 2020. Epic, meditech gain U.S. hospital market share as other EHR vendors lose ground. https://www.fiercehealthcare.com/tech/epic-meditech-gain-u-s-hospital-market-share-as-other-ehr-vendors-lose-ground (accessed December 2, 2020).
Lanham, H. J., R. R. McDaniel, Jr., B. F. Crabtree, W. L. Miller, K. C. Stange, A. F. Tallia, and P. Nutting. 2009. How improving practice relationships among clinicians and nonclinicians can improve quality in primary care. Joint Commission Journal on Quality and Patient Safety 35(9):457–466.
Lanham, H. J., R. F. Palmer, L. K. Leykum, R. R. McDaniel, Jr., P. A. Nutting, K. C. Stange, B. F. Crabtree, W. L. Miller, and C. R. Jaen. 2016. Trust and reflection in primary care practice redesign. Health Services Research 51(4):1489–1514.
Lau, B. D., A. H. Haider, M. B. Streiff, C. U. Lehmann, P. S. Kraus, D. B. Hobson, F. S. Kraenzlin, A. M. Zeidan, P. J. Pronovost, and E. R. Haut. 2015. Eliminating health care disparities with mandatory clinical decision support: The Venous Thromboembolism (VTE) example. Medical Care 53(1):18–24.
Leykum, L. K., H. Chesser, H. J. Lanham, P. Carla, R. Palmer, T. Ratcliffe, H. Reisinger, M. Agar, and J. Pugh. 2015. The association between sensemaking during physician team rounds and hospitalized patients’ outcomes. Journal of General Internal Medicine 30(12):1821–1827.
Liaw, W., A. H. Krist, S. T. Tong, R. Sabo, C. Hochheimer, J. Rankin, D. Grolling, J. Grandmont, and A. W. Bazemore. 2018. Living in “cold spot” communities is associated with poor health and health quality. Journal of the American Board of Family Practice 31(3):342–350.
Mandel, J. C., D. A. Kreda, K. D. Mandl, I. S. Kohane, and R. B. Ramoni. 2016. Smart on FHIR: A standards-based, interoperable apps platform for electronic health records. Journal of the American Medical Informatics Association 23(5):899–908.
Mann, D. M., J. Chen, R. Chunara, P. A. Testa, and O. Nov. 2020. COVID-19 transforms health care through telemedicine: Evidence from the field. Journal of the American Medical Informatics Association 27(7):1132–1135.
Medgadget. 2020. Electronic health records market size 2020 industry analysis, share, growth, upcoming trends, segmentation and forecast by 2025. https://www.medgadget.com/2020/02/electronic-health-records-market-size-2020-industry-analysis-sharegrowth-upcoming-trends-segmentation-and-forecast-by-2025.html (accessed December 2, 2020).
Meskó, B., Z. Drobni, É. Bényei, B. Gergely, and Z. Győrffy. 2017. Digital health is a cultural transformation of traditional healthcare. mHealth 3:38.
Moacdieh, N., and N. Sarter. 2015. Clutter in electronic medical records: Examining its performance and attentional costs using eye tracking. Human Factors 57(4):591–606.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. The promises and perils of digital strategies in achieving health equity: Workshop summary. Washington, DC: The National Academies Press.
NASEM. 2019. Taking action against clinician burnout: A systems approach to professional well-being. Washington, DC: The National Academies Press.
NCQA (National Committee for Quality Assurance). 2020. Patient-centered medical home (PCMH). https://www.ncqa.org/programs/health-care-providers-practices/patient-centered-medical-home-pcmh (accessed June 2, 2020).
NCVHS (National Committee on Vital and Health Statistics). 2019. Additional recommendations for HHS actions to improve the adoption of standards under the Health Insurance Portability and Accountability Act (HIPAA) of 1996. Hyattsville, MD: National Committee on Vital and Health Statistics.
Nelson, E. C., M. Dixon-Woods, P. B. Batalden, K. Homa, A. D. Van Citters, T. S. Morgan, E. Eftimovska, E. S. Fisher, J. Ovretveit, W. Harrison, C. Lind, and S. Lindblad. 2016. Patient focused registries can improve health, care, and science. BMJ 354:i3319.
Nouri, S., E. C. Khoong, C. R. Lyles, and L. Karliner. 2020. Addressing equity in telemedicine for chronic disease management during the COVID-19 pandemic. NEJM Catalyst. https://catalyst.nejm.org/doi/full/10.1056/CAT.20.0123 (accessed January 8, 2021).
O’Malley, A. S., J. M. Grossman, G. R. Cohen, N. M. Kemper, and H. H. Pham. 2010. Are electronic medical records helpful for care coordination? Experiences of physician practices. Journal of General Internal Medicine 25(3):177–185.
ONC (Office of the National Coordinator for Health Information Technology). 2013. Building technology capabilities to aggregate clinical data and enable population health measurement. Washington, DC: Office of the National Coordinator for Health Information Technology.
ONC. 2019. Office-based physician electronic health record adoption. dashboard.healthit. gov/quickstats/pages/physician-ehr-adoption-trends.php (accessed December 8, 2020).
Ornstein, S. M., L. S. Nemeth, P. J. Nietert, R. G. Jenkins, A. M. Wessell, and C. B. Litvin. 2015. Learning from primary care meaningful use exemplars. Journal of the American Board of Family Practice 28(3):360–370.
Patel, V., and C. Johnson. 2019. Trends in individuals’ access, viewing and use of online medical records and other technology for health needs: 2017–2018. Washington, DC: Office of the National Coordinator for Health Information Technology.
Phillips, R. L., Jr., A. W. Bazemore, J. E. DeVoe, T. J. Weida, A. H. Krist, M. F. Dulin, and F. E. Biagioli. 2015. A family medicine health technology strategy for achieving the triple aim for us health care. Family Medicine 47(8):628–635.
Pronovost, P., M. M. E. Johns, S. Palmer, R. C. Bono, D. B. Fridsma, A. Gettinger, J. Goldman, W. Johnson, M. Karney, C. Samitt, R. D. Sriram, A. Zenooz, and Y. C. Wang, eds. 2018. Procuring interoperability: Achieving high-quality, connected, and person-centered care. Washington, DC: National Academy of Medicine.
Ratwani, R. M., M. Hodgkins, and D. W. Bates. 2018a. Improving electronic health record usability and safety requires transparency. JAMA 320(24):2533–2534.
Ratwani, R. M., B. Moscovitch, and J. P. Rising. 2018b. Improving pediatric electronic health record usability and safety through certification: Seize the day. JAMA Pediatrics 172(11):1007–1008.
Ratwani, R. M., E. Savage, A. Will, A. Fong, D. Karavite, N. Muthu, A. J. Rivera, C. Gibson, D. Asmonga, B. Moscovitch, R. Grundmeier, and J. Rising. 2018c. Identifying electronic health record usability and safety challenges in pediatric settings. Health Affairs 37(11):1752–1759.
Rittenhouse, D. R., P. P. Ramsay, L. P. Casalino, S. McClellan, Z. K. Kandel, and S. M. Shortell. 2017. Increased health information technology adoption and use among small primary care physician practices over time: A national cohort study. Annals of Family Medicine 15(1):56–62.
Rock, R. M., W. R. Liaw, A. H. Krist, S. Tong, D. Grolling, J. Rankin, and A. W. Bazemore. 2019. Clinicians’ overestimation of their geographic service area. Annals of Family Medicine 17(Suppl 1):S63–S66.
Rucker, D. 2018. Achieving the interoperability promise of 21st Century Cures. https://www.healthaffairs.org/do/10.1377/hblog20180618.138568/full (accessed May 6, 2020).
Schellhase, K. G., T. D. Koepsell, and T. E. Norris. 2003. Providers’ reactions to an automated health maintenance reminder system incorporated into the patient’s electronic medical record. Journal of the American Board of Family Practice 16(4):312–317.
Sharko, M., L. Wilcox, M. K. Hong, and J. S. Ancker. 2018. Variability in adolescent portal privacy features: How the unique privacy needs of the adolescent patient create a complex decision-making process. Journal of the American Medical Informatics Association 25(8):1008–1017.
Sharon, T. 2018. When digital health meets digital capitalism, how many common goods are at stake? Big Data & Society 5(2):1–12.
Smith, C., C. Balatbat, S. Corbridge, A. Dopp, J. Fried, R. Harter, S. Landefeld, C. Martin, F. Opelka, L. Sandy, L. Sato, and C. Sinsky. 2018. Implementing optimal team-based care to reduce clinician burnout. Washington, DC: National Academy of Medicine.
Society for Adolescent Health Medicine, S. H. Gray, R. H. Pasternak, H. C. Gooding, K. Woodward, K. Hawkins, S. Sawyer, and A. Anoshiravani. 2014. Recommendations for electronic health record use for delivery of adolescent health care. Journal of Adolescent Health 54(4):487–490.
Starfield, B., L. Shi, and J. Macinko. 2005. Contribution of primary care to health systems and health. Milbank Quarterly 83(3):457–502.
Street, R. L., Jr., L. Liu, N. J. Farber, Y. Chen, A. Calvitti, D. Zuest, M. T. Gabuzda, K. Bell, B. Gray, S. Rick, S. Ashfaq, and Z. Agha. 2014. Provider interaction with the electronic health record: The effects on patient-centered communication in medical encounters. Patient Education and Counseling 96(3):315–319.
Tate, C., and P. Warburton. 2020. U.S. hospital EMR market share 2020 report. https://klasresearch.com/resources/blogs/2020/06/15/us-hospital-emr-market-share-2020-report (accessed January 26, 2021).
The Larry A. Green Center and PCC (Primary Care Collaborative). 2020. Quick COVID-19 primary care survey: Clinician survey. https://www.green-center.org/covid-survey (accessed September 15, 2020).
Tong, S., R. A. Mullen, C. J. Hochheimer, R. T. Sabo, W. R. Liaw, D. E. Nease, Jr., A. H. Krist, and J. J. Frey, 3rd. 2019. Geographic characteristics of loneliness in primary care. Annals of Family Medicine 17(2):158–160.
Verma, S. 2020. Early impact of CMS expansion of Medicare telehealth during COVID-19. https://www.healthaffairs.org/do/10.1377/hblog20200715.454789/full (accessed December 2, 2020).
Vest, J. R., and L. D. Gamm. 2010. Health information exchange: Persistent challenges and new strategies. Journal of the American Medical Informatics Association 17(3):288–294.
Washington, V., K. DeSalvo, F. Mostashari, and D. Blumenthal. 2017. The HITECH era and the path forward. New England Journal of Medicine 377(10):904–906.
Weatherly, J., S. Kishnani, and T. Aye. 2019. Challenges with patient adoption of automated integration of blood glucose meter data in the electronic health record. Diabetes Technology & Therapeutics 21(11):671–674.
Webber, E. C., D. Brick, J. P. Scibilia, P. Dehnel, Council on Clinical Information Technology, Committee on Medical Liability and Risk Management, and Section on Telehealth Care. 2019. Electronic communication of the health record and information with pediatric patients and their guardians. Pediatrics 144(1).
Whitelaw, S., M. S. Mamas, E. J. Topol, and H. G. C. Van Spall. 2020. Applications of digital technology in COVID-19 pandemic planning and response. The Lancet. https://www.thelancet.com/action/showPdf?pii=S2589-7500%2820%2930142-4 (accessed December 2, 2020).
Woolf, S. H., D. A. Chapman, R. T. Sabo, D. M. Weinberger, and L. Hill. 2020. Excess deaths from COVID-19 and other causes, March–April 2020. JAMA 324(5):510–513.
Wosik, J., M. Fudim, B. Cameron, Z. F. Gellad, A. Cho, D. Phinney, S. Curtis, M. Roman, E. G. Poon, J. Ferranti, J. N. Katz, and J. Tcheng. 2020. Telehealth transformation: COVID-19 and the rise of virtual care. Journal of the American Medical Informatics Association 27(6):957–962.
Yen, P. Y., D. M. Walker, J. M. G. Smith, M. P. Zhou, T. L. Menser, and A. S. McAlearney. 2018. Usability evaluation of a commercial inpatient portal. International Journal of Medical Informatics 110:10–18.
Young, R. A., S. K. Burge, K. A. Kumar, J. M. Wilson, and D. F. Ortiz. 2018. A time-motion study of primary care physicians’ work in the electronic health record era. Family Medicine 50(2):91–99.


































