




Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
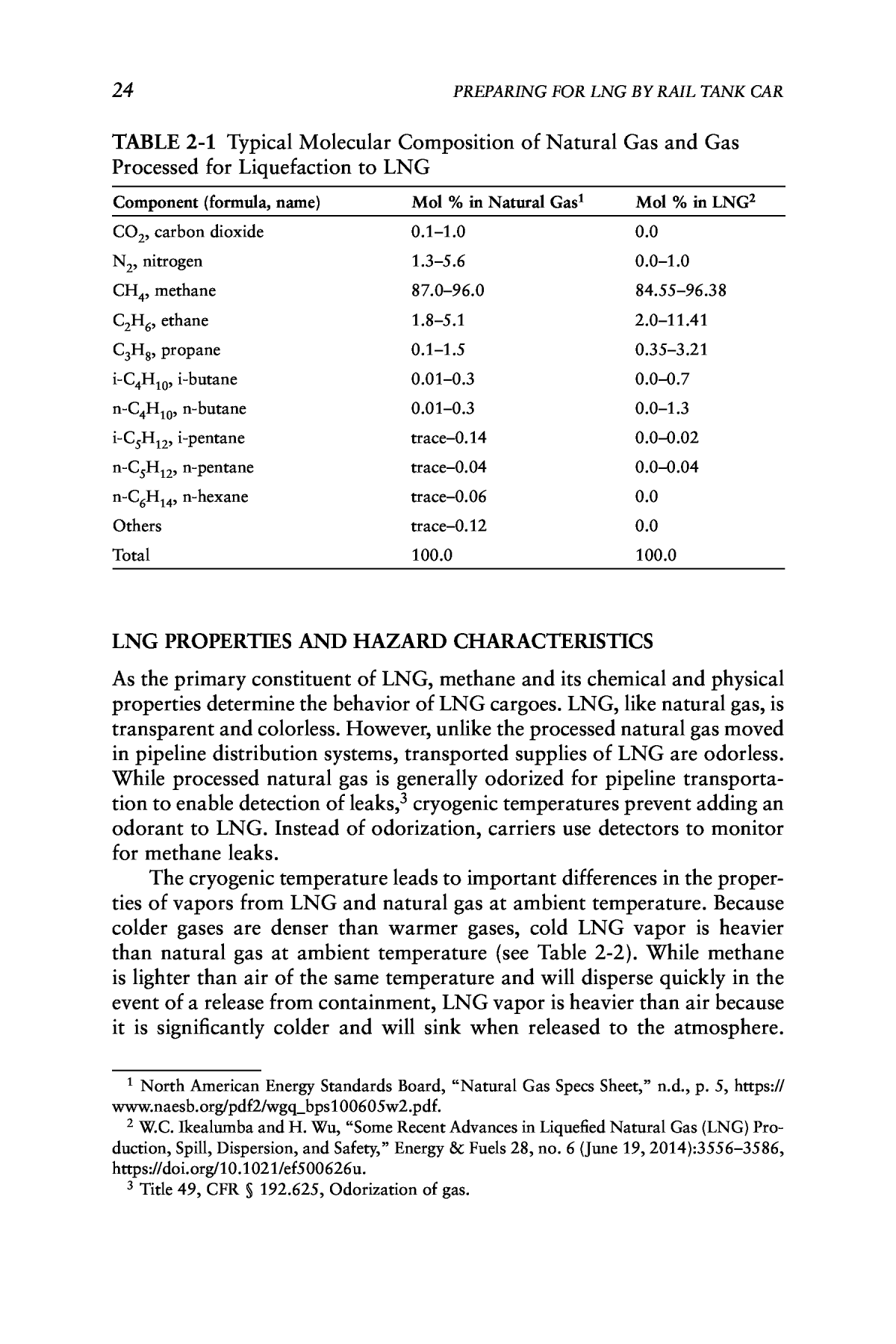
23 This chapter provides background on liquefied natural gas (LNG), its chemical and physical properties, and hazard characteristics. The chapter then discusses these hazards in the context of rail transportation. WHAT IS LNG? Natural gas is a mixture of hydrocarbons extracted from underground reservoirs that consists primarily of methane (CH4) and small amounts of ethane, propane, and other heavier hydrocarbons. Natural gas extractions can also include trace amounts of nitrogen, helium, carbon dioxide, sulfur compounds, water, and in some cases, mercury. As noted in the previous chapter, natural gas can be liquefied to reduce its volume by a factor of about 600. Liquefaction allows for the transportation of supplies overseas and to domestic markets lacking gas transmission pipelines or requiring additional supplies to supplement pipeline gas. However, liquefaction alone does not transform natural gas into LNG. Before natural gas is lique- fied, it is processed to remove impurities and meet end-user specifications. Table 2-1 lists the typical components of natural gas and supplies of LNG readied for transport. Natural gas processed to create LNG contains a higher percentage of methane and smaller amounts of other elements and compounds than unprocessed natural gas. 2 Background on Liquefied Natural Gas and Its Transport
24 PREPARING FOR LNG BY RAIL TANK CAR TABLE 2-1 Typical Molecular Composition of Natural Gas and Gas Processed for Liquefaction to LNG Component (formula, name) Mol % in Natural Gas1 Mol % in LNG2 CO2, carbon dioxide 0.1â1.0 0.0 N2, nitrogen 1.3â5.6 0.0â1.0 CH4, methane 87.0â96.0 84.55â96.38 C2H6, ethane 1.8â5.1 2.0â11.41 C3H8, propane 0.1â1.5 0.35â3.21 i-C4H10, i-butane 0.01â0.3 0.0â0.7 n-C4H10, n-butane 0.01â0.3 0.0â1.3 i-C5H12, i-pentane traceâ0.14 0.0â0.02 n-C5H12, n-pentane traceâ0.04 0.0â0.04 n-C6H14, n-hexane traceâ0.06 0.0 Others traceâ0.12 0.0 Total 100.0 100.0 LNG PROPERTIES AND HAZARD CHARACTERISTICS As the primary constituent of LNG, methane and its chemical and physical properties determine the behavior of LNG cargoes. LNG, like natural gas, is transparent and colorless. However, unlike the processed natural gas moved in pipeline distribution systems, transported supplies of LNG are odorless. While processed natural gas is generally odorized for pipeline transporta- tion to enable detection of leaks,3 cryogenic temperatures prevent adding an odorant to LNG. Instead of odorization, carriers use detectors to monitor for methane leaks. The cryogenic temperature leads to important differences in the proper- ties of vapors from LNG and natural gas at ambient temperature. Because colder gases are denser than warmer gases, cold LNG vapor is heavier than natural gas at ambient temperature (see Table 2-2). While methane is lighter than air of the same temperature and will disperse quickly in the event of a release from containment, LNG vapor is heavier than air because it is significantly colder and will sink when released to the atmosphere. 1 North American Energy Standards Board, âNatural Gas Specs Sheet,â n.d., p. 5, https:// www.naesb.org/pdf2/wgq_bps100605w2.pdf. 2 W.C. Ikealumba and H. Wu, âSome Recent Advances in Liquefied Natural Gas (LNG) Pro- duction, Spill, Dispersion, and Safety,â Energy & Fuels 28, no. 6 (June 19, 2014):3556â3586, https://doi.org/10.1021/ef500626u. 3 Title 49, CFR § 192.625, Odorization of gas.
BACKGROUND ON LIQUEFIED NATURAL GAS AND ITS TRANSPORT 25 TABLE 2-2 Select Properties of Methane4 Methane Chemical formula CH4 Initial boiling point â258.7°F Vapor pressure, in mm Hg At 100°F At â258.7°F 258,574 760 Relative vapor density (air = 1) 0.55 Flash point â306°F Auto-ignition temperature 1,004°F Flammability limits, lower 5% Flammability limits, upper 15% If a sufficient amount of LNG is spilled, the cryogenic liquid can form a pool that will be accompanied by a dense cloud of vapors that will flow along the ground before dispersing as its temperature increases. Skin contact with an LNG pool or vapor cloud will cause cryogenic burns. Cryogenic damage to materials is also a concern because some exposed materials can become brittle and crack. In addition, like the vapors from other cryogenic liquids, a sufficiently high concentration of LNG vapor, especially in an enclosed area, can cause asphyxiation because it displaces oxygen in the area. LNG is flammable as it returns to a gaseous state and mixes with air at ambient temperature, which poses a combustion hazard once its concentra- tion in the air reaches 5 percent and continues to do so up to 15 percent (see Table 2-2). LNG rapidly vaporizes into natural gas when it is spilled onto land or water to form a dense vapor cloud that has a very low height relative to its horizontal dimensions because the cold LNG vapor density is initially >1.5 times that of air. Owing to its low temperature, the cloud will not become immediately buoyant but will spread much farther and persist longer than hydrocarbon gases at ambient temperature that are lighter than air when immediately released. A low lying, dense cloud has a much greater chance of reaching an ignition source than one that readily disperses into the atmosphere. Moreover, once ignited, the heat radiated to objects in proximity to an LNG pool fire5 is more extreme than the 4 National Oceanic and Atmospheric Administration, âChemical Datasheet: Methane,â CAMEO Chemicals, n.d., https://cameochemicals.noaa.gov/chemical/8823. 5 A pool fire can occur when a flammable liquid spills, spreads, mixes with air as it vapor- izes, and finds a source of ignition. The fire is fueled by the continuing vaporization of the volatile liquid such that the mixture with air remains within its flammability limit.
26 PREPARING FOR LNG BY RAIL TANK CAR heat radiated from pool fires involving other hydrocarbon gases, such as liquefied petroleum gas (LPG). For example, the average surface emissive heat flux of an LPG pool fire is 48 kW/m2 compared to 153â286 kW/m2 for an LNG pool fire.6 Likewise, the radiant heat flux of an LNG fireball7 (475â540 kW/m2)8 is also much higher than the heat flux from an LPG fireball (195â287 kW/m2).9,10 For an object engulfed by an LNG fire, the heat flux is about double that for one engulfed by an LPG fire.11 HAZARDS IN THE CONTEXT OF RAIL TRANSPORTATION Despite its hazards, LNG has been safely transported by marine vessels and cargo tank trucks for several decades. The rail environment, however, is different, and therefore a central concern of the Task Force is examining these hazards in context, such as by presenting risks from a loss of contain- ment during a derailment or by an error during transfer of the product to and from a rail tank car. In a 2016 safety assessment, the Federal Railroad Administration (FRA) identified LNGâs flammability, cryogenic, and handling proper- ties as potentially creating hazards that warrant assessment for safe rail 6 G.A. Mizner and J.A. Eyre, âLarge-Scale LNG and LPG Pool Fires,â 1982, 147â163; T.K. Blanchat et al., âThe Phoenix Series Large Scale LNG Pool Fire Experiments,â December 1, 2010, https://doi.org/10.2172/1044989. For LNG, the lower value is found in the work of Mizner and Eyre, the upper value in that of Blanchat et al. In addition, the range for a 35 m LNG pool in D. Nedelka et al. (âThe Montoir 35 m Diameter LNG Pool Fire Experiments,â in Proc LNG IX, 9th Int Conf & Exp on LNG, Nice, France, 1989) is 257â273 kW/m2. 7 A fireball can occur when a dense vapor cloud is ignited, which, for example, could be precipitated by the rupture of a container caused by a boiling liquid expanding vapor explo- sion. The dynamics of a fireball result in rapid consumption of the fuel and extreme heat. 8 S. Betteridge and L. Phillips, âLarge Scale Pressurised LNG BLEVE Experiments,â January 2015, p. 9, https://www.scopus.com/inward/record.uri?eid=2-s2.0-84958212719&partnerID= 40&md5=a4a0547c56b80bd20b4ecd4305318337. 9 T. Roberts, A. Gosse, and S. Hawksworth, âThermal Radiation from Fireballs on Failure of Liquefied Petroleum Gas Storage Vessels,â Process Safety and Environmental Protection 78, no. 3 (May 1, 2000):184â192, https://doi.org/10.1205/095758200530628. 10 Roberts, Gosse, and Hawksworth; D.M. Johnson, M.J. Pritchard, and British Gas plc. Research and Technology Division, Large Scale Experimental Study of Boiling Liquid Expand- ing Vapour Explosions (BLEVEs), British Gas plc., Research and Technology Division, 1991, http://www.opengrey.eu/item/display/10068/632267. In referencing fireball tests of similar masses, the upper value of 540 kW/m2 for LNG is comparable to 195 kW/m2 for propane. The lower value of 475 kW/m2 for an LNG fireball is comparable to an average of values (287 kW/m2 and 344 kW/m2) found in studies on LPG. 11 Blanchat et al., âThe Phoenix Series Large Scale LNG Pool Fire Experimentsâ; Pipeline and Hazardous Materials Safety Administration, âUN-T75 Portable Tank Fire-Testing Task Resource,â August 2020, fig. 42. The committee compared Figure 42 in the section on âFire Performance of Cryogenic ISO UN-T75â in this resource document with the work by Blanchat et al.
BACKGROUND ON LIQUEFIED NATURAL GAS AND ITS TRANSPORT 27 transportation.12 With respect to each, FRA pointed to the following issues warranting attention: 1. FlammabilityâA spill of LNG, on land or on water, will rapidly evaporate, and the resulting vapor cloud will move via wind pat- terns before dispersing. If flammable concentrations come in con- tact with an ignition source, then it will ignite, and a flash fire will form downwind and possibly upwind; 2. Cryogenic propertiesâIn addition to the risk of cryogenic burns to people, materials such as metals that become embrittled at cryo- genic temperatures are susceptible to cracking. In rail transporta- tion, the TC-128 grade B steel outer tank shell of a DOT-113 tank has the potential for cracking from embrittlement when exposed to a release of LNG,13 which could cause a loss of vacuum; and 3. HandlingâWithout a properly functioning pressure release valve system, the expansion ratio of LNG (600 to 1) could cause over- pressurization, which could result in the rupture of the container. An overpressure can also result from a rapid phase transition, which is a mechanical explosion with a blast of enough magnitude to be a hazard to emergency response personnel working in close proximity to the release. Additionally, friction from flowing LNG may accumulate a static electric charge during the loading and unloading process without bonding and grounding. Training in the handling of LNG cargo is necessary to avoid a static discharge in the presence of flammable LNG vapor, which may be sufficient to cause ignition. These and other hazards, as they pertain to the rail environment, are the subject of the Task Forceâs activities as reviewed in the chapters that follow. 12 Federal Railroad Administration Office of Research, Development and Technology, âLNG Safety Assessment Evaluation Methods,â July 2016, Table 4, https://railroads.dot.gov/sites/fra. dot.gov/files/fra_net/16555/LNGSafetyAssessmentEvalMethods_final.pdf. 13 B.W. Williams et al., âCapturing Variability in the Fracture Response of TC128B Steel Using Damage Mechanics,â Procedia Structural Integrity, 1st Virtual European Con- ference on FractureâVECF1, 28, January 1, 2020:1024â1038, https://doi.org/10.1016/j. prostr.2020.11.118.
