GEORGE HERMANN BÜCHI
August 1, 1921–August 28, 1998

BY GLENN A. BERCHTOLD AND LOUISE H. FOLEY
MASSACHUSETTS INSTITUTE OF TECHNOLOGY Professor Emeritus George H.Büchi of Cambridge, Massachusetts, and Jackson, New Hampshire, one of this century’s foremost organic chemists, died of heart failure while hiking with his wife in his native Switzerland on August 28, 1998, at the age of 77. Throughout his life he was an avid hiker, hunter, skier, and fisherman whose fondness and appreciation of the outdoors rivaled his love of science.
Büchi was born in Baden, Switzerland, on August 1, 1921. He received a diploma in chemical engineering from the Eidgenössische Technische Hochschule (ETH) in Zürich in 1945. He received the D.Sc. in organic chemistry from the ETH in 1947, while working in the laboratory of Professor Leopold Ruzicka. Even in the 1940s the ETH was the center of organic chemistry in Europe, and Büchi’s education and love for the chemistry of natural products was heavily influenced by close association with distinguished faculty, including not only Ruzicka but also V.Prelog, O. Jeger, and P.A.Plattner. Early in his graduate days George Büchi completed a synthesis of 1-methylazulene. The area of research was of active interest to Professor Plattner, and it was agreed that the work would be published with Plattner
as coauthor.1 This one publication became the source of an error begun in Topics in Organic Chemistry by L.F.Fieser and M.Fieser,2 which persisted most of his career. Because of this one paper Büchi was often introduced as a doctoral student of Plattner and, as was his style, he never corrected the introducer.
Büchi left the ETH and came to the United States as a Firestone postdoctoral fellow in the laboratory of Professor M.Kharasch at the University of Chicago. During his three-year tenure in Kharasch’s laboratory Büchi’s systematic investigations of free-radical chemistry augmented his curiosity in the potential utility of organic photochemistry.
In 1951 Büchi accepted an offer from the late Arthur C. Cope to join the faculty of the Chemistry Department at the Massachusetts Institute of Technology. He was promoted to associate professor in 1956 and to full professor in 1958. He was appointed the Camile and Henry Dreyfus professor of chemistry in 1969, a position he held until his retirement in 1991. During his 40 years of service at MIT, Büchi trained 70 doctoral students and more than 130 postdoctoral students. Many of his former coworkers have gone on to leadership positions in academia and industry throughout the world. George Büchi’s research accomplishments, reported in over 200 publications, made significant contributions in diverse areas of organic chemistry, including organic photochemistry, structure elucidation of natural products, synthesis of natural products, toxicology, and the development of new synthetic methods. What makes the number of publications even more impressive was that George’s high standards did not allow him to publish anything but a complete work: a complete structure, a total synthesis, a novel synthetic method. He often suggested to the students whose synthetic work did not lead to the total
synthesis but contained interesting chemistry that they publish the work on their own.
In addition to contributions from his own research and teaching, George’s originality, thorough knowledge of organic chemistry, and its literature made him a highly respected and prized consultant to Pfizer (1956–63) and Hoffmann-La Roche (Nutley, New Jersey, and Basle, Switzerland, 1963–91) and throughout his career to Firmenich, S.A. (Geneva, Switzerland, 1954–91). It was rare that he could not recite on the spot where, when, what journal, and the author for a critical reference or procedure. His consulting contributions, like his research, were not just to reproduce the literature but rather to come up with novel uses of a method or a totally new method to accomplish a critical step in a synthesis. One of his suggestions resulted in a new route to vitamin K patented by Roche and Büchi. Many of his contributions to flavor and fragrance chemistry have been patented by Firmenich, S.A., and Büchi, and most with coinventor and nearly career-long coworker Hans Wüest.
Prior to the 1950s, the photochemistry of organic compounds was a rarely investigated and poorly understood field of endeavor. Büchi’s accomplishments in this area in the 1950s were instrumental in converting this latent field into an understandable and useful synthetic tool, and they laid the groundwork for what is now modern organic photochemistry. The light-catalyzed addition of carbonyl compounds to olefins had been observed by E.Paterno in 1909, but the structure of the oxetane products remained a mystery until elucidated by Büchi and coworkers in 1954. The reaction is now known as the Paterno-Büchi reaction, and its scope was extended in Büchi’s laboratories to include the addition of carbonyl compounds to alkynes and alkenes. Over a 10-year period new photochemical reactions and structural work in Büchi’s laboratory, coupled with his
mechanistic insights provided a detailed understanding of numerous fundamental photoreactions, all of which were unprecedented at the time.
At about the same time, Büchi began structural work in natural products that led to the structure determination of more than 55 natural products. During this era, prior to the routine use of X-ray crystallographic methods for organic structure determination, his accomplishments have been described by his peers as among the finest examples of structure elucidation by classic degradation and spectrometric techniques. The structures of the sesquiterpenes patchouliol (with J.Dunitz), maaliol, aromadendrene, valerenic acid, calarene, and copaene (with P.de Mayo), all containing novel skeletons, were disclosed in succession. Work on the complex alkaloids (uleine, flavocarpine and aconitine), the latter with K.Weisner, led to the structures through ingeniously conceived degradative studies. A study on the bis-indole alkaloid voacamine suggested to Büchi that the antitumor alkaloids vinblastine and vincristine were structurally similar bis-indoles. This suggestion was confirmed in collaboration with Klaus Biemann (MIT) and N.Neuss and M.Gorman (both of Eli Lilly).
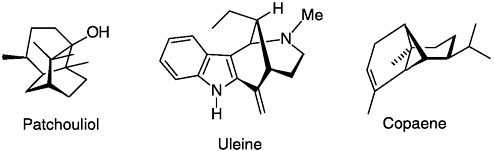
The synthesis of over 75 complex natural products came from Büchi’s laboratory. His peers considered his syntheses creative, elegant, and original. Typically his syntheses were
very efficient in producing quantities of the target compound in very few steps. The first syntheses of many sesquiterpenes (e.g., patchouliol, maaliol, aromadendrene, agarofuran), iboga alkaloids, iridoid glucosides (loganin), fulvoplumierin, aflatoxins, and their metabolites were
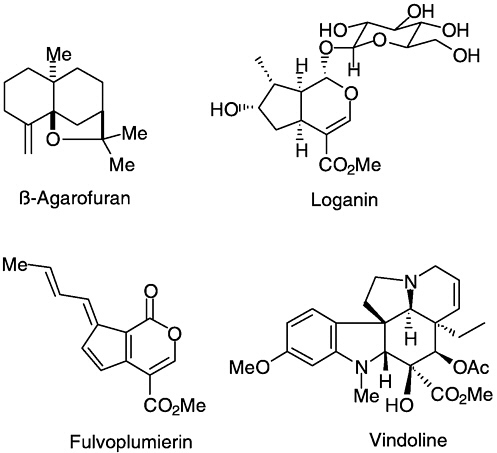
accomplished. Both vindoline and catharanthine first yielded to synthesis in his laboratory; these two alkaloids were subsequently combined by Potier to produce the antitumor drug, vinblastine. Neolignans (burchellin, guianin, futoenone) and several marine natural products, including parazoanthoxanthin and dibromophakellin, were synthesized
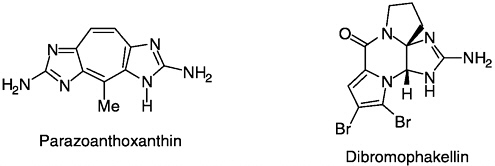
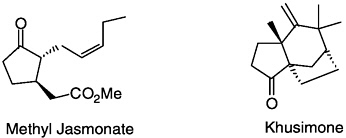
by elegant biomimetic routes. Throughout his career Büchi maintained a strong interest in flavor and fragrance principles, and many were synthesized in his laboratory. They include damascenes (rose), methyl jasmonate (jasmine), muscone (musk deer), sinensals (orange), khusimone (vetiver), khaweofuran (coffee), furaneol (strawberry), and muscopyridine.
Many important contributions to synthetic methodology were developed in Büchi’s laboratory. They include numerous methods for new and convenient preparations of olefins, carbonyl compounds, macrocycles, tropolones, and heterocycles. A new synthesis of allylic sulfones and their conversion to polyolefins provided for an elegant synthesis of ß-carotene from vitamin A.
In 1962 Büchi and MIT colleague Professor Gerald N. Wogan initiated a collaborative effort that ultimately established molecular toxicology as an important scientific discipline. Their experimental evidence concerning carcinogens
provided fundamental chemical, biological, and epidemiological correlations that are a paradigm for modern toxicological studies. They were first concerned with establishing the structures of the aflatoxins, fungal metabolites that had been isolated from moldy peanuts and found to be responsible for a mass outbreak of poultry disease. Aflatoxin B1, the major metabolite, is a potent carcinogen, and its consumption is associated with primary liver cancer. Büchi deduced the structure of aflatoxin B1 by ingenious application of spectroscopic information. Subsequent work resulted
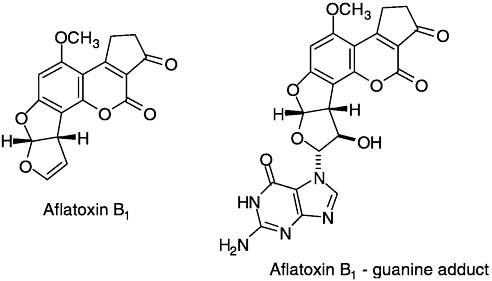
in the isolation and structure identification of other aflatoxin metabolites (M1, P1, Q1) and a number of other mycotoxins (rubatoxins, tryptoquivalines, mollicellins, malformin C). In a series of brilliant synthetic studies, he devised methods for their total synthesis. Several of these syntheses provided the quantities of aflatoxin metabolites needed for essential toxicological investigations. Later work established the structure of the major adduct formed between aflatoxin B1 and DNA. The isolation and identification of the DNA-
aflatoxin-derived adduct as a 3-hydroxyaflatoxin linked via C-9 to the N-7 atom of a guanine of DNA was a milestone in toxicology. It provided the first definitive molecular structure for a carcinogen-induced covalent modification of a mammalian DNA.
George did not become the “head” of organic chemistry at MIT; academic administration was not his style. Yet he influenced organic chemistry at MIT in many ways. In the 1970s he believed that organic chemistry at MIT should develop with a more significant bioorganic component. He and a few colleagues led the department in that direction and thus had an impact on the future thrust of the department. George nurtured the highly talented young faculty in organic chemistry and biochemistry. He was excited by the arrival of new faculty members, and he made a significant effort to help their careers both inside and outside MIT. His presence provided a magnet that attracted excellent graduate students and young as well as established organic faculty to MIT.
One of us (L.H.F.) was drawn to the Büchi group as a
graduate student after reading his papers and seeing the insights and knowledge each paper contained. Clearly here was someone with knowledge to share, and share he did with his students and other coworkers. Up until the late 1970s he could be expected to come in to talk with his students and coworkers several times a day and during nights and weekends. How many of us recall working long hours trying unsuccessfully to determine a side product’s structure only to have GB or “the boss,” both of which his students called him, come in, sit down, and in minutes (if not seconds) figure it out and then share with all how he had deduced it? In their first years, graduate students could count on George for numerous suggestions of things to try when problems occurred. In the last years of their studies, when things went wrong, students often found themselves on their own for a period of time. During this time George watched from a distance to see if the students were searching for solutions on their own. If he saw they were and still were not making progress, he would step back in with suggestions of things to try or papers to read. This approach made his students very self-sufficient and ready for the “real world,” where he knew they would be expected to “sink or swim” on their own.
George’s high standards also applied to his teaching. In his early career at MIT he taught a basic organic chemistry course and a graduate course in natural products. It was the natural products course that was his course and the course for which most MIT organic graduate students and postdocs will remember George. All who attended will recall the impeccable chalk talks; each structure was drawn with care and clarity and a lecture was full of insights on structure determination and synthetic methods. Each year George analyzed recent publications in structure elucidation and synthesis of natural products, and in this course
he provided his evaluation of the work reported. This course taught one how to read the literature and how to analyze what was presented, not to just accept it as fact; no better education was available anywhere.
George had a wonderful sense of humor, and as a graduate student I (L.H.F.) was continually coming up with tricks to play on him, often enlisting other graduate students to share in the fun. One could be sure that he would both enjoy the joke and at some later point extract some revenge. This continued even after I left MIT. On one occasion I gave him a $1.99 bottle of New Hampshire wine saying that I thought as a wine connoisseur and a New Hampshire resident he should have at least one bottle of New Hampshire wine in his wine cellar. Sometime later I was invited to share one of Anne Büchi’s fine dinners, and I alone was served the New Hampshire “fine wine.” The face I made on taking the first sip was sufficient to make George’s day.
One of the fascinating delights of being a colleague of George Büchi was to experience his expression of the unique insight into structure and synthesis that he possessed. His critical evaluation of the works of others, not only in his graduate course but also during the daily course of events, was truly a work of art. He genuinely praised the work of high intellectual talent; he quickly discovered, and often corrected, the mistaken approach or conclusions. Although others grasped the opportunity to correct even the smallest errors in recent literature, to his credit, he was above this. His published corrections were only those of major works, and they were done in a gentlemanly fashion—a characteristic of the man.
Yes, we all knew and respected George as a gentleman and a perfectionist in everything with which he was associated. Dinner with George was always a special treat. The
food, the wine, the ambiance had to be perfect—it was special. But then again, a late evening dinner on the banks of the Stewart River in the Yukon Territory, where the appetizer was fire-roasted beaver tail on a stick followed by duck or goose prepared and presented in similar fashion was, I (G.A.B.) believe, one of the most enjoyable meals George and I shared: not the ambiance of Lock-Ober but a gastronomical delight. He knew how to get the most out of life, and he certainly did that.
George’s love of the outdoors began in early life and continued throughout his life with alpine skiing up until 1995 and cross-country skiing, hiking, hunting, and fly fishing up until the time of his death. He began alpine skiing (often using skins to climb uphill) as a youngster in Switzerland. His characteristic limp was the result of a collision he had as a teenager. This collision resulted in a number of broken bones and damage to his hip, the pain from which he endured up until its replacement in 1980. Following hip replacement he was told he should not alpine ski, but since it was one of his loves, he continued to ski. At each checkup his orthopedic surgeon would report the hip was as good as when it was put in. This prompted the doctor to say, according to George, that he was going to suggest that all his hip replacement patients should continue to ski and those who did not already ski should take it up: a story told with George’s characteristic humor.
George’s insights into organic chemistry continued right up until his death. One of us (L.H.F.) had the pleasure to spend George’s last birthday with him and Anne in New Hampshire. Along with hiking and wonderful cuisine that weekend, there was a discussion of a natural product synthesis. As was so characteristic of George, midway through our discussion he stopped and began to write down a new
route to the final compound involving a novel rearrangement.
George’s contributions to chemistry were recognized by election to the National Academy of Sciences in 1965, and numerous awards, honorary degrees, and over 30 honorary lectureships around the world. Among the awards he received were the J.R.Killian Faculty Achievement Award (MIT’s highest faculty award), the Order of the Rising Sun from the Government of Japan, the American Chemical Society Award for Creative Work in Synthetic Organic Chemistry, the first Ruzicka Prize, and the Fritzche Award of the American Chemical Society.
One of his former colleagues in describing George wrote, “He was one of the most balanced and influential chemists of his day. More to the point, he embodied a style of chemistry—excellence in science, breadth, curiosity, and enormous joie de vivre—that made him a symbol for organic synthesis at its best and most sane. He was a remarkable man, who embodied a remarkable era, and did so in a way that made the whole enterprise human.”3 George died as he would have preferred: out-of-doors doing something he loved, but for his wife and his many friends it was much too soon.
George is survived by his wife of 43 years, Anne Barkman Büchi of Cambridge, Massachusetts, and Jackson, New Hampshire; a brother, Heinrich, of Berne, Switzerland; and three nephews, all of whom live in Switzerland.
THE AUTHORS GRATEFULLY acknowledge Anne Büchi, Satoru Masamune, Gerald Wogan, and Hans Wüest for their help in the preparation of this memoir.
NOTES
SELECTED BIBLIOGRAPHY
1954 With C.Inman and E.S.Lipinsky. Light catalyzed organic reactions. 1. The reaction of carbonyl compounds with 2-methylbutene-2. J. Am. Chem. Soc. 76:4327.
1956 With J.T.Kofron, E.Koller, and D.Rosenthal. Addition of aromatic carbonyl compounds to a disubstituted acetylene. J. Am. Chem. Soc. 78:876.
1959 With K.Wiesner, M.Götz, D.L.Simmons, L.R.Fowler, F.W.Bachelor, and R.F.C.Brown. The structure of aconitine. Tet. Lett. 2:15.
With E.W.Warnhoff. The structure of uleine. J. Am. Chem. Soc. 81:4433.
1963 With T.Asao, M.M.Abdel-Kade, S.B.Chang, E.L.Wick, and G.N. Wogan. Aflatoxins B and G. J. Am. Chem. Soc. 85:1706.
With M.Dobler, J.D.Dunitz, B.Gubler, H.P.Weber, and J.Padilla O. The structure of patchouli alcohol. Proc. Chem. Soc., Lond. 383.
1964 With N.Neuss, M.Gorman, W.Hargrove, N.J.Cone, K.Biemann, and R.E.Manning. The structure of the oncolytic alkaloids vinblastine and vincristine. J. Am. Chem. Soc. 86:1440.
With W.D.MacLeod, Jr., and J.Padilla O.Terpenes. XIX. Synthesis of patchouli alcohol. J. Am. Chem. Soc. 86:4438.
With R.E.Manning and S.A.Monti. Voacamine and voacorine. J. Am. Chem. Soc. 86:4631.
1966 With D.L.Coffen, K.Kocsis, P.E.Sonnet, and F.E.Ziegler. The total synthesis of iboga alkaloids. J. Am. Chem. Soc. 88:3099.
1967 With D.M.Foulkes, M.Kurono, G.F.Mitchell, and R.S.Schneider. The total synthesis of racemic aflatoxin B1. J. Am. Chem. Soc. 89:6745.
1969 With J.A.Carlson. The total synthesis of fulvoplumierin. J. Am. Chem. Soc. 91:6470.
1970 With J.A.Carlson, J.E.Powell, Jr., and L.F.Tietze. The total synthesis of loganin. J. Am. Chem. Soc. 92:2165.
1971 With S.M.Weinreb. Total synthesis of aflatoxins M1 and G1 and an improved synthesis of aflatoxin B1. J. Am. Chem. Soc. 93:746.
With K.E.Matsumoto and H.Nishimura. The total synthesis of vindorosine. J. Am. Chem. Soc. 93:3299.
1974 With R.Friedinger. A new synthesis of allylic sulfones and their conversion to polyolefins, ß-carotene, and vitamin A. J. Am. Chem. Soc. 96:3332.
1975 With M.Ando and T.Ohnuma. The total synthesis of racemic vindoline. J. Am. Chem. Soc. 97:6880.
1977 With J.M.Essigmann, R.G.Croy, A.M.Nazdan, W.F.Busby, V.N. Reinhold, and G.N.Wogan. Structural identification of the major DNA adduct formed by aflatoxin B1 in vitro. Proc. Natl. Acad. Sci. U. S. A. 74:1870.
With A.Hauser and J.Limacher. The synthesis of khusimone. J. Org. Chem. 42:3323.
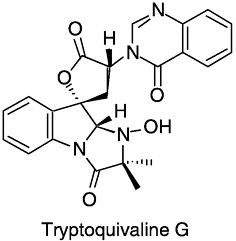
1979 With P.R.DeShong, S.Katsumura, and Y.Sugimura. Total synthesis of tryptoquivaline G. J. Am. Chem. Soc. 101:5084.
With H.Wüest. Macrocycles by olefination of dialdehydes with 1,3-bis(dimethyl-phosphone)-2-propanone. Helv. Chim. Acta. 62:2661.
With H.Wüest. New synthesis of ß-agarofuran and dihydroagarofuran. J. Org. Chem. 44:546.
1981 With P.-S.Chu. A synthesis of gymnomitrol. Tetrahedron. 37:4509.
With L.H.Foley. A biomimetic synthesis of dibromophakellin. J. Am. Chem. Soc. 104:1776.
1986 With J.C.Leung. Total syntheses of atrovenetin and scleroderodione. J. Org. Chem. 51:4813.
1989 With H.Wüest. The synthesis of racemic ambrox. Helv. Chim. Acta. 72:996.
1994 With D.A.Home and K.Yakushijin. A two-step synthesis of imidazoles from aldehydes via 4-tosyloxyazolines. Heterocycles 39:139.


















