8
Insect Societies as Divided Organisms: The Complexities of Purpose and Cross-Purpose
JOAN E. STRASSMANN and DAVID C. QUELLER
Individual organisms are complex in a special way. The organization and function of their parts seem directed toward a purpose: the survival and reproduction of that individual. Groups of organisms are different. They may also be complex, but that is usually because their parts, the individual organisms, are working at cross-purposes. The most obvious exception to this rule is the social insects. Here, the individuals cooperate in complex ways toward the common goal of the success of the colony, even if it means that most of them do not reproduce. Kin selection theory explains how this can evolve. Nonreproductive individuals help in the reproduction of their kin, who share and transmit their genes. Such help is most favored when individuals can give more to their kin than they give up by not reproducing directly. For example, they can remain at their natal site and help defend a valuable resource (“fortress defenders”), or they can ensure that at least one adult survives to care for helpless young (“life insurers”). Although kin selection explains the extensive cooperation and common purpose of social insect colonies, it also predicts a certain amount of cross-purpose and conflict behavior. Kin selection has predicted how workers and queens disagree over sex ratios, how potential queens struggle to be the colony’s head, how workers try to produce sons, and how other workers often prevent them. Kin
Department of Ecology and Evolutionary Biology, Rice University, Houston, TX 77005.
selection analysis of cooperation and conflict in social insects is one of the outstanding achievements of evolutionary theory.
THE ROCK, THE CLOCK, AND ORGANISMAL COMPLEXITY
Darwin built his theory of descent with modifications from many quarters. He took uniformitarianism from the geologist Charles Lyell, the struggle for existence from the economist Thomas Malthus, and homology from a number of continental biologists. Perhaps most surprising is his debt to a theologian, William Paley. At university, Darwin had Paley’s Natural Theology (Darwin, 1887b) almost by heart.
Paley pointed to the complexity of organisms and claimed that such complexity required a supernatural intelligence. Darwin’s chief achievement was to provide a scientific explanation for adaptive complexity. Paley had famously built his argument from a rock and a clock (Paley, 1802). A stone, he argued, did not beg for any special explanation. It was simple, predictable, unchanging, devoid of obvious purpose. It might have been put there by some intelligence, but nothing about it begged for that explanation. A watch told a different story. The gears, levers, and springs work together in intricate harmony, causing the hands to move across the labeled face and measure time. Such complexity of design or purpose could not arise by chance. The watch must have had a designer, a watchmaker. Paley then applied the argument to organisms and their parts. The eye has a complex arrangement of parts that have a clear purpose, endowing its bearer with sight, and such complexity of purpose seemed to imply a designer and a maker. Throughout the rest of the book, Paley polishes the argument and applies it to other cases, including the sting of the worker honey bee, which he called a neutral bee.
Darwin won the argument with Paley long ago. Both had candidate explanations for complexity, but only Darwin also described a natural mechanism for adaptation and a natural explanation for the changes observed in fossils. Only Darwin explained aspects of biology that were nonadaptive consequences of history, from vestigial organs and other homologies to biogeographical patterns. Our understanding that organisms are a mix of historical constraint and adaptation by natural selection has led to many successful predictions about the natural world, whereas Paley’s theory stands mute about the details. In other words, Darwin’s theory is much richer than a simple explanation for design; it makes many further extensions and predictions. Some of these extensions and predictions were not fully appreciated in Darwin’s time. The last several decades have seen increased attention to a further important question
about the apparent design of organisms. A good theory of design also ought to explain what kinds of entities are adapted and what kinds of complexity they show.
Organisms, together with man-made machines, seem to show a unique kind of complexity. We will call this the “complexity of purpose.” “Purpose,” as used here, is a metaphor, just as “natural selection” is a metaphor and has no real selector. This kind of complexity can even be used to define biological organisms. The organism is the consolidated unit of design or adaptation; almost everything in the organism seems built to further the individual’s survival and reproduction (Queller, 1997). Few parts of the organism are organized to gain at the expense of other parts, and few parts of the organism are organized to benefit other organisms (the chief exception being offspring).
The same cannot generally be said about groups of organisms. How does a flock of birds compare with the rocks and clocks? The parts of a flock of birds, the individual birds themselves, do not generally appear organized to benefit the flock. To the contrary, the members compete for food and mates, sometimes by physical fights, and they hide behind each other as shields against predation. Groups of organisms, e.g., flocks, populations, species, and communities, are not themselves clock-like or organismal.
Neither are they like the rock, because they are far more complex. But in contrast to the complexity of purpose shown by organisms, these aggregates have what we call the “complexity of cross-purpose.” The behavior of flocks, populations, and communities is extraordinarily rich, but not in a predictable and unified manner like the meshing of gears in a watch. Instead, much of the complexity stems from indifference of the parts to other parts and the apparent striving of each part to further its own survival and reproduction, if necessary at the expense of other parts.
Evolutionary theory has been addressing this issue of what kinds of units are adapted, and as it has done so, an interesting puzzle has emerged. The entities that we recognize as individual organisms actually originated as groups of lower-level units (Buss, 1987; Maynard Smith and Szathmáry, 1995). Somehow, the first cell assembled a group of components sufficient to sustain replication. The eukaryotic cell began as an assemblage of several prokaryotic cells, with at least the mitochondria and chloroplasts having independent origins. Larger organisms are groupings of cells. If groups show cross-purpose, how did they combine and make the transition to the unity of purpose of a single organism?
Social insect groups can give us special insight into this question. We will argue that social insect colonies are much like organisms, and we will show how their unity of purpose can arise through kin selection. We will also show that some cross-purpose remains, that colonies are not perfectly coherent. These remaining conflicts might be viewed as
compromising the organismal nature of the colony. But a closer look at traditional individuals shows that they too have some internal conflicts (Burt and Trivers, 2006; Haig, 1996; Hurst et al., 1996). For example, selfish genetic elements such as transposons can not only make up large parts of genomes, using expensive resources and extending replication times, but they can also interfere with the functioning of the individual (Burt and Trivers, 2006). The conflicts within cooperative social insect colonies have helped biologists to identify conflict in other cooperative entities, for example the conflicts between maternal and paternal genes mediated by genomic imprinting (Haig, 2000).
A HOUSE DIVIDED
A small stingless bee out in a tropical forest might seem like any other animal as it searches for food to survive and reproduce. Upon closer inspection, a more complex picture would emerge. The foraging bee is a member of a complex colony inside a tree hollow. Within, there is a citadel of wax with a smooth protective skin surrounding fat peripheral cells that contain honey and pollen, and central combs of smaller brood cells, all held together and supported by a lacy network of wiry wax struts. Small female worker bees are busy everywhere, bringing in food and propolis, adding to the structures, cleaning, and guarding. But the focus of their attention is a single female, the queen, with a greatly distended abdomen and worn, useless wings. A throng of workers surrounds the queen so closely as to slow her approach to an empty cell. At the empty cell, the queen antennates the inside, as if checking its construction. The workers dart in and out, at one time crouching before the queen, at other times rearing up before her. This agitated ballet ends with the queen stroking the workers, who then regurgitate larval food into the cell, one after the other, until it is full. Then the queen lays a single egg that floats on the provisions. When she leaves, the workers carefully bend the collar of wax over and close the cell. The egg will hatch and grow to adulthood undisturbed but benefiting from the workers’ attention to climate control and defense (Zucchi, 1993).
This scene summarizes what is special about social insects: complex communication and integration of behavior, and individuals caring for the offspring of another. The colony as a whole appears to have the kind of integration and common purpose normally associated with individual organisms, with the parts subservient to the whole. The stingless bee colony is highly organized, both structurally and behaviorally. The provisioning and oviposition process seems to have an almost clock-like precision, with elaborate coordination between the queen who lays the egg and the workers who build and provision the cell. This process only
works as a whole. If any step in the provisioning and oviposition process is omitted, the whole operation may fail.
For societies with this level of organization, it is no wonder that the claim for organismal status of groups has sometimes been made (Seeley, 1989; Wheeler, 1911). If this claim stands up to scrutiny, it is extraordinary in two ways because we think of organisms as consolidated units in two senses: they are both physically contiguous and genetically uniform.
An organism is typically one solid, connected mass. If it is a single cell, it is bounded by a membrane; if it is multicellular, the cells abut one another and form a discrete larger unit. If a social insect colony is an organism, however, it is a divided one, with parts (the individuals) freely moving past each other and only occasionally coming into direct contact. Other organisms with separated parts are known. A lizard may detach its tail to save itself from a predator, and the tail continues to twitch, distracting the predator long after the main body of the lizard has escaped. Similarly, when a honey bee worker stings a foe, the barbed sting can easily detach from the honey bee’s body, and when it does, the sting continues to dig into the victim’s skin and the poison sac continues to contract and deliver more of its venom. But these detached organs act independently for only a brief time before expiring. Moreover, these parts are clearly secondary, in the sense that a joining of trunk and tail did not form the lizard. Instead, tails are normally attached parts of the organism, both in the lizard itself and in its relatives with nondetachable tails. A social insect superorganism, on the other hand, is built from the very beginning of detached parts. Physical attachment is rare and ephemeral, such as when army ant workers interlock to form a sheltering bivouac.
A typical organism is also genetically homogeneous. Again, social insect colonies differ from this standard. In the simplest colony structure, all members are offspring of a single queen and her mate, so they share many genes, but each receives its own unique combination of parental genes. In other species, this genetic distinctness is exacerbated by the presence of multiple queens or multiple mates. This genetic structure is utterly different from the clonal, mitotically derived set of cells that constitute a typical multicellular organism (Maynard Smith, 1988). Given that natural selection operates by favoring genes that pass copies into the next generation, it is little surprise that a clonal entity can evolve cooperation. If social insect colonies lack this unity of genotype, what gives them the unity of purpose that makes them an organism rather than a contentious flock?
HOW ARE COLONIES ORGANISMAL?
It is not hard to view a termite castle, an army ant bivouac, or a wasp colony as a single, coordinated organism. Each shows division of labor,
with specialization for reproduction, nutrition, communication, defense, and often thermoregulation. Seemingly autonomous individuals are actually workers whose function appears directed entirely to the whole, such as a worker who fans the colony to cool it or one who lives her life as a living honey storage pot (Seeley, 1989). The earliest analogies with multicellular organisms focused on these physiological processes and led to Wilson’s physiologically oriented definition of a superorganism (Wheeler, 1911; Wilson, 1971; Hölldobler and Wilson, 1990; Bonner, 2006). But the analogy could not be pushed too far, perhaps because of fundamental differences between the physiology of a divided organism (with separately mobile individuals) and a multicellular organism. The mobility of individuals means information and resources can be walked throughout the colony with no need for specialized structures.
Mobility may therefore underlie the relatively small number of castes in social insects. Castes are in some ways analogous to cell types in multicellular organisms. Each caste or cell type specializes in certain tasks, with the division of labor aiding the whole. All social insects have functional reproductive and worker roles, but only some are morphologically differentiated into queen and worker castes. A fraction of these species have multiple worker castes, with the primary distinction being between small foragers and large soldiers (Wilson, 1971). Even highly specialized functions, such as being a honey storage vessel in honeypot ants or using one’s head to block the colony entrance in Colobopsis ants, are usually performed by castes that also have more general functions.
Fig. 8.1 shows the complexity, measured as the number of types of subunits, of social insect colonies, compared with multicellular individuals. In one sense, of course, social insect colonies are more complex than multicellular individuals because the colonies include all of the complexity of their constituent individuals and then add more complexity at the colony level. But it is still interesting to compare the degree of complexity added by the specialization of parts in the two cases. Following Bonner (2006), as a measure of the complexity of specialization, we use cell-type number and caste number to represent the complexity of individuals and colonies, respectively. We are unable to use phylogenetically independent contrasts, but Fig. 8.1 well illustrates how depauperate in specialized castes social insects are compared with cell specialization in organisms, a pattern that is unlikely to disappear when analyses are performed with accurate phylogenies. Complexity increases with the number of units, the units being cells for organisms and individuals for colonies (Fig. 8.1). The lower complexity of colonies can be explained partly by size. On average, social insect colonies do not have as many units as multicellular animals; colonies rarely have more than a million individuals, whereas large organisms have billions of cells. But that is not the complete explanation. The
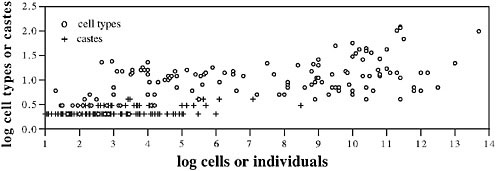
FIGURE 8.1 The number of specialized types (cell types or worker castes) as a function of the number of units (cells in an organism or number or individuals in a colony). Cell data (open circles) are from Bell and Mooers (1997). Caste data (crosses) are for ants and were compiled by Bonner (1993) from data in Hölldobbler and Wilson (1990) and from judgments of caste number by E. O. Wilson (Bonner, 1993). Mean colony sizes were used when available; when they were not available, we used the midpoint of the range.
specialization complexity of insect societies is lower for a given number of units than the complexity of multicellular organisms. This conclusion seems fairly clear despite the difficulties inherent in defining the number of castes or cell types, for which we have relied on the judgments of others (see Fig. 8.1). Social insects almost never have even as many as five castes, whereas many small multicellular organisms attain 10, or many more, cell types. We suggest that the mobility of the separate parts of a social insect colony reduces the need for specialized types at the level of the colony.
Despite the limitations of the physiological superorganism model, the superorganism view can be useful, not only for understanding divided organisms but also for stimulating new ways to view traditional organisms. Colonies may not have the same kinds of systems as animals and plants for fulfilling colony functions, just as in typical organisms the parts of a colony are subordinate to the whole. As Darwin himself noted, “… if on the whole the power of stinging be useful to the social community, it will fulfill all of the requirements of natural selection, although it may cause the death of some few members” (Darwin, 1872, p. 163). A colony can be viewed as an organism simply because it is highly adapted at the colony level. Of course, this logic does not apply to just any social community, so the social insects force the question of how some entities become organismal while others do not.
Although social insect colonies may not have physiologies that closely match those of multicellular organisms, they do have their own systems for
defense, nutrition, and reproduction. Close study of any social insect species would reveal examples of individual traits that have evolved for colony function, but the honey bee is the best-studied case. For example, honey bees use floral food sources, and they need to track this ever-changing resource. Their famous dance language allows workers to communicate the quality and location of a resource to nestmate workers. But they also have mechanisms to effectively allocate work among multiple food sources, even when each individual has quite limited information. Fidelity to a good food source is not absolute; a fraction of workers always seeks to discover new food sources. Returning foragers adjust the intensity of their waggle dancing (the number of waggle runs) according to the profitability of their trip. Foragers from better sources therefore recruit more followers, so the colony concentrates on the better food source (Seeley, 1997).
Foragers can also tune the intensity of their recruitment dances according to how much the colony needs food, but this requires coordination with the workers that specialize in nectar processing. If a colony needs more nectar, the nectar-processing bees that have this information crowd closer to the hive entrance. This means that a returning forager unloads her nectar quickly, which cues her to intensify her dancing to recruit more foragers (Seeley, 1997) and allay the shortage of nectar. On the other hand, there may sometimes be more nectar coming into the colony than the nectar processors can handle, resulting in inefficiently long unloading times. In this event, foragers perform the tremble dance, which is different from the waggle dance in that it stimulates other workers to become nectar processors (Seeley, 1997).
Such integrated behaviors of many workers in a honey bee colony allow the colony to find and exploit food efficiently, to alter group foraging based on individual information, and to adjust the number of foragers and nectar processors to meet changing needs. No individual is doing anything that by itself would be very useful; instead, each is performing a role in a process that only makes sense in terms of increasing colony function. Such a smooth coordination among workers in finding, harvesting, and processing food makes the argument for the colony as an organism compelling. Similar kinds of coordination are found wherever they are looked for in social insects, for example, in nest construction in Polybia wasps (Jeanne, 1986), in nest-finding in honey bees (Seeley and Visscher, 2004) and Leptothorax ants (Mallon et al., 2001), and in the establishment of foraging trails by army ants (Franks et al., 1991).
THE SUCCESS OF SOCIAL INSECTS
If colonies have found ways to be more efficient than separate individuals, one might expect social insects to be particularly successful. In
fact, they are fantastically successful, particularly the ants and termites, but also the bees and wasps (Wilson, 1987). Success can be measured as current ecological success, e.g., geographic diversity, species richness, and biomass. Ants are very speciose and are native to all terrestrial habitats except Antarctica, Iceland, Greenland, and a few remote islands (Wilson, 1987). At one thoroughly studied location in the Amazonian forest, social wasps, ants, bees, and termites make up four-fifths of all insect biomass and over a fifth of all animal biomass (Fittkau and Klinge, 1973).
Success can also be measured in evolutionary antiquity, staying power, and diversity. Social insects are represented in the fossil record by impressions of their bodies and their nests. The fossils of bodies are most useful for tying these insects to extant lineages, whereas the fossil nests demonstrate clear evidence of ancient sociality. Body fossils of presumably social termites, ants, bees, and wasps are found in the Cretaceous, whereas nest fossils are found for some lineages as early as the Triassic (Grimaldi et al., 1997; Hasiotis, 2003; Bordy et al., 2004). Only an origin before the breakup of Pangaea in the late Triassic is consistent with the worldwide extent of major social insect lineages. The dispersion of major social insect lineages was essentially complete before the high sea levels of the Late Cretaceous isolated many land masses 100 Mya (Hasiotis, 2003; Bordy et al., 2004). Another indication of the evolutionary robustness of the social habit is the absence of clear evidence for major lineages of social insects that subsequently went extinct (Wilson, 1987).
AN INORDINATE FONDNESS FOR KINSHIP
Any theory of adaptation or design ought to explain why social insect groups are so well adapted while most groups of multicellular organisms are not adapted. Perhaps the most common feature of insect societies, aside from their cooperation, is that they are family groups. In some species, colonies are headed by one singly mated queen (or in termites, a queen and a king), and all other colony members are full siblings. In others, the degree of relatedness is lower, but it is nevertheless substantial in all but a few species of unicolonial insects that are recently derived from those with higher relatedness. The high relatedness within colonies is often enforced by overt kin recognition: nonrelatives are recognized and rejected. Haldane once quipped that nature suggests that the creator must have had an inordinate fondness for beetles. With respect to superorganisms, there also seems to be an inordinate fondness for kinship ties among cooperators.
Darwin had at least an inkling of this: “As with the varieties of the stock, so with social insects, selection has been applied to the family, and not to the individual, for the sake of gaining a serviceable end” (Darwin, 1872, p. 230). But the idea was not formalized until W. D. Hamilton’s work
in the 1960s (Hamilton, 1964a,b). Taking a gene’s-eye view, Hamilton reasoned that a gene could spread in future generations not only by contributing to the production of offspring, but also by aiding reproduction of other relatives who might share the gene. Genetic relatedness specifies the comparative values of different kin. Hamilton’s rule predicts what behaviors will be favored by selection. A particularly useful form of Hamilton’s rule, for behaviors that exert a fitness cost, c, on some relatives and give a fitness benefit, b, to others, is rbb > rcc. The two fitness effects are scaled by the relatedness of the actor to those benefited, rb, and to those harmed, rc. Crucially, individuals can be selected to give up their own offspring (rc = 1/2) to help other relatives, provided the benefit b is sufficiently higher than the cost c. Genetic relatedness among individuals is essential, for without it no value of b, the benefit to cooperation, will favor giving up reproducing oneself.
THE HAPLODIPLOID HYPOTHESIS
Hamilton (1964b) also noticed a special feature of social ants, bees, and wasps: they share a haplodiploid sex determination mechanism in which haploid males arise from unfertilized eggs and diploid females arise from normal fertilized eggs (Normark, 2003). This is an ancestral trait in Hymenoptera that arose long before sociality (Hamilton, 1967), but it affects relatednesses in ways that could favor sociality. What makes it significant for sociality is that sisters are related by 3/4 because they share all their genes from their haploid father. Other things being equal, a sister would therefore pass on more genes by rearing sisters (r = 3/4) than by rearing her own progeny (r = 1/2), favoring daughters who remain with their mothers to rear additional sisters. Hamilton noted that this observation could potentially explain at a stroke at least two salient feature of social insects (Hamilton, 1964b). First, there have been many origins of sociality in the haplodiploid Hymenoptera and few elsewhere, termites being the most notable exception. Second, only females work in the Hymenoptera, whereas both sexes work in diploid termites.
However, this haplodiploid hypothesis is debatable, for a variety of reasons (Alexander et al., 1991; Queller and Strassmann, 1998). First, most haplodiploids have not evolved sociality, whereas a few diploids have. Another issue is that relatedness is elevated only to full sisters and is lowered to brothers (r = 1/4) (Crozier, 1970). Thus, if a female helps to rear an equal mixture of sisters and brothers, the average relatedness (1/2) is exactly the same as to her own offspring and exactly the same as full siblings in diploids. Haplodiploidy can still help if workers can concentrate on rearing sisters (Hamilton, 1972), but the advantage is transitory and disappears at sex ratio equilibrium (Crozier and Pamilo, 1996).
Also, the special 3/4 relatedness applies only in colonies with a single, once-mated queen. Multiple queens and queen replacement reduce relatedness, as does multiple mating. Accurate estimates of relatedness among colony members are now available for hundreds of species, typically based on inherited variation in DNA microsatellite loci (Gadagkar, 1990b; Crozier and Pamilo, 1996; Peters et al., 1999). Although relatedness among female colony members in many species is near the full-sister value of 3/4, in many other species it is lower.
Finally, other possible explanations, some of them noted by Hamilton himself (Hamilton, 1964b), have been proposed for the facts the haplodiploid hypothesis seems to explain. Specifically, the high incidence of sociality in the Hymenoptera and the all-female workforce may relate to preadaptations involving parental care (Alexander et al., 1991). The solitary Hymenoptera have an unusually high level of parental care, meaning that adaptations for nest-building, prey capture, brood care, sanitation, and defense are already in place. It must be much easier to evolve alloparental care in groups that already have parental care. And because it is females that provide the parental care in solitary Hymenoptera, with special adaptations such as the sting, it is not surprising that females provide the care in social Hymenoptera (Alexander et al., 1991).
KIN SELECTION AND SYNERGISM: LIFE INSURANCE AND FORTRESS DEFENSE
Kin selection has been so closely identified with the haplodiploid hypothesis that concerns with the latter have caused some to question kin selection in general. But of course Hamilton’s rule does not require that relatedness to beneficiaries must be higher than relatedness to one’s own offspring. If rb = rc, or even if rb < rc, Hamilton’s rule can still favor altruism if the benefit is sufficiently greater than the cost (b > c) (West-Eberhard, 1975). The question then concerns how it is possible to rear more young by aiding the beneficiary than by reproducing independently. Synergies from division of labor between helpers and reproducers are easy to see after sociality has evolved, but this kind of specialization seems unlikely to be present at the beginning of sociality.
Two kinds of factors seem especially likely to provide the necessary advantage to helping: “fortress defense” and “life insurance” (Queller and Strassmann, 1998). Fortress defenders live in protected, expandable sites that generally include food (Andersson, 1984; Alexander et al., 1991; Crespi, 1994), such as the wood galleries of termites and the plant galls of social aphids and thrips. An offspring can gain by remaining at the natal site, even if she has to rear less-related collateral relatives, because she avoids risking death by migrating to a new site (see Fig. 8.2a). Because
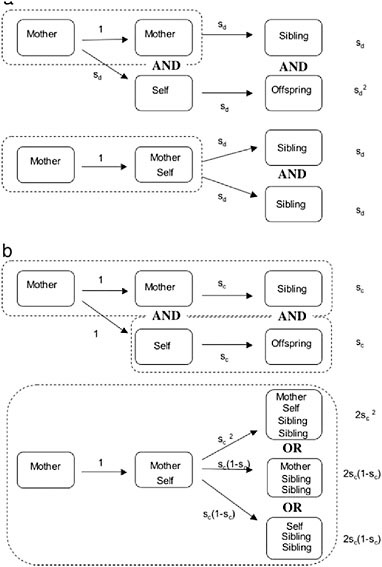
FIGURE 8.2 Fitness advantages of sociality when siblings and offspring are equally valuable. Arrows show transitions from one time period to another, and enclosure within dashed rectangles indicates the same nest. (a) Leaving vs. Fortress Defense. If an individual (self) leaves her natal site, she survives dispersal with probability sd, whereas her mother survives in the safe natal site. Both then produce offspring. Alternatively, if self stays in the safe site with her mother, she avoids a bout of dispersal mortality and doubles her mother’s output. (b) Leaving vs. Life Insurance. Here we assume that dispersal carries no cost but that a parent survives the period of parental care with probability sc. If she dies during this period, her offspring also die. When self stays with her mother, there are three ways for the offspring to survive: if both adults survive, if only the mother survives, and if only self survives.
food is available at the home site, little feeding care is needed, and the first worker specialists are generally soldiers specialized for defense.
By contrast, life insurers cooperate to ensure that dependent young survive (Queller, 1989, 1994, 1996; Strassmann and Queller, 1989; Gadagkar, 1990a). They can live in a variety of sites, safe or unsafe, but generally have helpless young that need food and protection. The problem is that an adult must undertake dangerous foraging for her young, but if she dies during one of these trips, her still-dependent young also die. But when a daughter stays at the natal nest to help, and either the mother or the daughter dies, the survivor can take over feeding and protecting the young, giving rise to the synergistic advantage (Fig. 8.2b). Wasps, bees, and ants appear to fit this mold. The crucial assumption that dead individual’s investments can be saved by its surviving colony mates has been experimentally confirmed in a stenogastrine wasp (Field et al., 2000). Other advantages besides fortress defense and life insurance are also possible, and much work remains to be done on assessing their relative importance.
RELATEDNESS IS STILL IMPORTANT
Whatever the fitness advantages of altruism might be, they are selectively irrelevant unless they go to relatives. Hamilton’s kin selection theory (Hamilton, 1964a,b) still provides the framework for understanding altruism, even if the altruism is not driven by extra-high relatedness. As noted above, the fact that social insect colonies consist of families, and that they exclude outsiders, shows that relatedness matters. But other studies have tested more specific predictions about the importance of relatedness.
A recent comparative study of wasps and bees (Wenseleers and Ratnieks, 2006) showed how workers modulate their altruism and selfishness according to relatedness in queenless colonies. Colonies with queens removed were used because (as we will see below) worker selfishness can be repressed in colonies with the queen present, either by the queen herself or by other workers. With the queen gone, some workers develop ovaries and lay unfertilized eggs that will develop into males. If all workers ceased working and took up laying eggs, the colony’s production of males would presumably fall, because a certain number of workers are needed to feed the larvae. In fact, the queenless colonies never had more than 40% reproductive workers; at least 60% remained as helpers. Most interesting was the finding that fewer workers laid eggs from species in which relatedness among workers was high. In other words, more workers stayed in the altruistic helping mode when relatedness was high. Relatedness explained 62% of the variance in percentage of helpers (Wenseleers and Ratnieks, 2006). Variation in relatedness also predicts variation in helping behavior in birds and mammals (Griffin and West, 2003).
Unexpected and powerful evidence of the importance of relatedness has come from sex ratio studies. As noted above, haplodiploid female workers are related to full sisters by 3/4 and to brothers by 1/4. This implies that, other things being equal, these workers ought to prefer helping sisters (Hamilton, 1972). Specifically, Trivers and Hare (1976) showed that in a colony with a single once-mated queen, workers should prefer to invest three times as much in future queens as in males. They also showed that, in species of ants likely to have a single, once-mated queen, investment ratios are in fact closer to this 3:1 ratio than to the usual Fisherian 1:1.
More impressive evidence comes from species with variable relatedness. When a queen mates multiple times, workers will not favor this 3:1 ratio because the workers are equally related (by 1/4) to half sisters and brothers (because brothers do not have fathers, multiple mating by the queen does not change their relatedness). This means that these workers should rear more males than those in singly mated colonies, and the frequency-dependent nature of sex ratio selection should cause the two kinds of colonies to increasingly specialize (Boomsma and Grafen, 1990, 1991). Workers in colonies with singly mated queens should specialize largely in rearing queens, and workers in colonies with multiply mated queens should specialize in rearing males. This odd prediction of what has come to be called “split sex ratio theory” was strikingly confirmed in a study of the ant Formica exsecta (Sundström et al., 1996) and has been confirmed in many other species (Queller and Strassmann, 1998; Chapuisat and Keller, 1999; Bourke, 2005).
Besides showing that workers are indeed sensitive to relatedness, the sex ratio studies made an even more important point: There can be conflict within these apparently superorganismal colonies. Queens are equally related to their sons and daughters, so they should prefer the standard 1:1 sex investment ratio (Trivers and Hare, 1976). The resulting conflict can lead to inefficiencies that are decidedly against the interests of the superorganism as a whole. For example, the split sex ratio described above for Formica exsecta is achieved only after some waste. Queens in both singly and multiply mated colonies laid the same sex ratio of eggs, but workers in the singly mated colonies achieved their preferred investment in full sisters by killing many of the male larvae (Sundström et al., 1996; Chapuisat et al., 1997).
It seems paradoxical that this elegant evidence for kin selection theory comes from conflict rather than from cooperation, but there is really no contradiction. Kin selection theory shows how individuals can further the reproduction of their own genes, and this is sometimes achieved by cooperation and sometimes by conflict.
AN EVEN MORE INORDINATE FONDNESS FOR SELF
Conflict over sex ratios was an exciting finding, both because of the support that it provided for kin selection theory and because it showed that even sterile workers could find ways to pursue interests that were different from the queen’s. It thus poked a hole in the view of a colony as superorganismal, a small hole by itself, but one that opened up a vista of other realms of possible conflict. Hamilton’s rule predicts more extensive conflict over the question of who gets to reproduce. Although typically related, individuals are genetically separate, and each individual is usually more related to its own young than to those of a relative. If other things are equal, Hamilton’s rule predicts that each individual would prefer to take the reproductive role. Thus, even though advantages like fortress defense or life insurance select for helping instead of going it alone, the issue of helping is not completely resolved because it is often better, still, to be helped.
Conflict over reproduction has long been apparent to researchers working on simple social insect societies that are made up of morphologically identical females, such as Polistes wasps. Colonies are initiated by single females or by groups of females, who are often sisters. They do not share reproduction equally. Instead, they form dominance hierarchies (Pardi, 1942; West-Eberhard, 1969; Strassmann, 1981) enforced by time-consuming aggression so intense it can result in death, although some species have conventions that reduce the battling (Hughes and Strassmann, 1988; Seppä et al., 2002). Once the hierarchy is set, the losers function as workers, if they choose to remain at the nest. But they still may not work optimally, instead waiting for a chance to reproduce. In Liostenogaster flavolineata, a Malaysian stenogastrine wasp that lacks morphological castes, a queue based on order of arrival determines who succeeds the dominant queen. When females reach the number two spot in the queue, they work less hard (Field et al., 2006). In other words, when the option of reproducing directly appears more likely, they decline to take as many risks on behalf of their relatives.
The success of predictions of sex ratio conflict led researchers to ask how much conflict over reproduction remains in highly eusocial insects, those with morphologically distinct queen and worker castes. Are these colonies subject to the complexities of cross-purpose?
It turns out that even the most advanced societies are not immune to this kind of conflict. When it comes time for honey bee colonies to divide, several half-sister queens are reared with special food in extra-large cells. The old queen leaves with much of the workforce to start a new colony. Then, the first of the new queens to emerge as an adult seeks out all of the other queen cells and uses her sting to kill her sister rivals (Gilley, 2001).
Why honey bees produce these extra queens is not fully clear, perhaps as insurance against one of them dying. But they do limit the conflict to a few individuals by controlling queen production through the special feeding they require. This limitation is extremely common in social insects with queen and worker castes. Queens generally require more food, offering the opportunity to control queen production (Wilson, 1971). It is instructive to see what happens in the unusual cases in which this constraint does not hold.
In one genus of stingless bees, Melipona, caste is not determined by differential feeding. Instead, workers and queens develop in cells of the same size, provisioned with the same amounts of food. This presumably leaves the choice of being queen up to each developing female larva. As a consequence, a significant fraction of females (5–20%) develop into queens, with small heads, large abdomens, and lacking the pollen baskets required to be effective workers (Wenseleers and Ratnieks, 2004). Because stingless bees reproduce by colony division, this amount of queen production is far more than the single queen they need, every once in awhile, to head a new colony. The excess queens, useless for work and a potential threat to the old queen, are slaughtered by workers (Wenseleers et al., 2004). The 5–20% reduction in worker production must constitute a significant cost to the colony and clearly shows that cross-purpose can remain important in advanced social insects.
Stingless bees other than Melipona determine caste by the usual means of feeding some larvae more, but this does not entirely solve the problem. In some species, in which brood cells are adjacent, a larvae that is supposed to be a worker can tunnel from its own cell into its neighbor’s, consume the food stores intended for its neighbor, and develop as a queen (Engels and Imperatriz-Fonseca, 1990). In other species, some individuals with worker-sized food allotments will nevertheless develop into morphological queens (Wenseleers et al., 2005). In Schwarziana quadripunctata, these dwarf queens make up only 0.6% of all individuals reared in worker-sized cells but 86% of all queens reared. These queens are less successful than normal-sized queens in attaining reproductive status and are executed more readily by the workers. Still, the strategy appears to be successful often enough to pay, inasmuch as 22% of all reproductive females are dwarf queens (Wenseleers et al., 2005). Some ant genera, such as Myrmica and Solenopsis, also have some small queens (called microgynes), which may be the result of individual larvae determining their own caste fate in colonies that are initiated by budding (Bourke and Franks, 1995; McInnes and Tschinkel, 1995).
The threat of other queens may lie behind another colony-level design flaw that is usually not obvious but is present in many species: the lack of a backup queen. Consider the fungus-growing ants of the genus Atta. A
mature Atta colony is a huge and intricate operation. Millions of ants cultivate fungi deep underground. The nest has chambers a person could stand in, gardens tended by a suite of worker castes, including leaf processors who use microbial fungicides and have specialized organs to carry these symbionts (Currie et al., 2006). However, this metropolis of ants has but a single queen, and when she dies the colony dies: the fungus gardens are overrun by rogue fungi, the workers cease to rear brood, and the galleries ultimately collapse. This disaster could be avoided by the relatively simple matter of keeping on hand one or more backup queens. But this would risk unleashing competition among the queens that might harm the interests of both the current queen and her workers.
WORKER REPRODUCTION AND POLICING
The existence of dwarf queens shows that even larvae that are fed less than the normal queen amount can reproduce. This ability can also extend to workers. Workers are females that are morphologically or behaviorally specialized to forage, care for brood, and defend the colony. None of these tasks is enhanced by egg-laying, and yet workers in nearly all species maintain some ability to lay eggs. Workers in many species regularly do so, producing males because they are uninseminated. They have considerable incentive to do this because a worker is more related to her own sons (1/2) than to brothers (1/4) produced by her mother (Trivers and Hare, 1976).
In some species, the queen “polices” these worker-laid male eggs, eating them when she finds them. Indeed the elaborate provisioning and oviposition process of stingless bees described earlier may sometimes be less a matter of cooperative communication about the filling of the cell than a contest over who gets to fill it. All that actually needs to be accomplished is regurgitation of food into the cell by workers, laying of an egg by the queen, and then sealing of the cell by workers. As it is, many more workers than necessary surround the empty cell. When the queen approaches the empty cell she can be very aggressive toward the workers, who ritually either approach her or back away. The queen often aggressively solicits food from the workers, who nearly always refuse to provide it. After the cell is filled, the queen lays an egg in it. Interestingly, the workers close the cell with their abdomen in it, a position in which they might lay an egg, something that might account for the commonness of worker male production in stingless bees (Tóth et al., 2002b). [In some species, workers return later to sealed cells to open them and oviposit (Beig, 1972; Tóth et al., 2002a), which can succeed because the worker-laid male egg hatches quickly, and the larva hunts down and kills the older female larva in the cell.] The fact that these behavior patterns differ considerably
between species is consistent with an “arms race” caused by conflict (Why would a good, efficient communication system need to change rapidly?). In other words, the complexity of the provisioning process may not be purposeful clockwork, but more like a boxing match full of punches and counterpunches. The winner varies from species to species, with queens producing all of the males in some and workers producing most in others (Tóth et al., 2004).
If we do not see these elaborate direct contests in other species, it may be because the stingless bees are rather unusual in mass-provisioning their brood cells with all of the food before oviposition. This focuses the laying process on one cell at a time and brings the contenders together. And of course, workers are favored to produce the males because colonies are headed by single-once mated queens (Peters et al., 1999).
In other social insects, workers have wider opportunities to lay eggs when the queen is absent. Policing by the queen then seems unlikely to succeed in colonies with thousands of workers, so one might expect workers to take advantage of this lack of control and produce many males. In some species they do, but in other species, like the honey bee, the queen still produces all of the males. Here, policing of worker-laid eggs is performed by other workers, in a seeming lapse of class solidarity. Each worker should prefer to lay her own male eggs, but how should she view the contest between other workers and the queen? When the queen is mated multiply, as in honey bees, and workers are usually half-sisters, each worker will prefer the queen’s males (r = 1/4) to those of other workers (r = 1/8) (Ratnieks, 1988). Again we have the inefficiencies of conflict, with some colony members negating what the others have done. Workers may be neuter but, to recall Paley’s adjective, they are anything but neutral. They would prefer to reproduce, but they also prefer not to let each other reproduce, and therefore they ensure that the queen wins. Oddly, the stronger the policing, the less actual conflict there may be. In species in which most worker-laid eggs are removed, few workers develop ovaries and lay eggs (Wenseleers and Ratnieks, 2006).
Workers can sometimes win the battle with queens in the most extreme way possible: by killing the queen (Bourke, 1994). Usually this is not a beneficial option, because the workers need the queen to produce both more workers and more reproductive females. But certain social insects have an annual colony cycle with males produced at the end, which makes the queen dispensable at the end of the season. Killing her allows workers to produce all of the males. Again, the choice appears to be affected by relatedness in ways predicted by kin selection theory: queens disappear more in singly mated species because those are the species in which workers favor production of males by other workers (Foster and Ratnieks, 2001).
CONCLUSIONS
Living things are complex, but this complexity is of two broad types. Organisms show complexity of apparent purpose, with all of the parts acting for the whole. Groups, however, are usually dominated by the complexities of cross-purpose; the parts seem goal-directed, but the goals are not shared, and the result is often anything but elegant. The most spectacular exceptions, at the group level, are social insect societies, in which the individuals usually do seem to act toward a common goal.
Any scientific theory purporting to account for biological complexity ought to account for this special nature of social insects. Why do their colonies show a degree of apparent purpose lacking in other aggregations, herds, and flocks? The kin selection extension of natural selection theory does explain this; cooperation results from the opportunity to give sufficiently large benefits to kin.
More importantly, kin selection theory has successfully predicted new findings. Although social insect colonies have clock-like design in many respects, kin selection theory predicts who is throwing sand into the clockworks, as well as which gears might be slipped and which springs sprung. Many of the predicted findings, such as sex ratio conflict and policing, were otherwise completely unexpected. The success of this approach shows that the Darwinian paradigm is capable of explaining not just the adaptations of organisms but also how new kinds of organismal entities come into being.
ACKNOWLEDGMENTS
We thank F. Ayala and J. Avise for organizing the symposium in which this work was presented, John Bonner for kindly providing original data for the social insect portion of Fig. 8.1, and J. J. Boomsma and the anonymous referees for helpful suggestions. This material is based on work supported by National Science Foundation Grant EF0626963.




















