Oil Spill Response Technologies
INTRODUCTION
In addition to exploring the viability of an ecosystem services approach to damage assessment and approaches to measuring and increasing the resiliency of the Gulf of Mexico (GoM) ecosystem, the committee was also charged with evaluating the effectiveness of the various spill response technologies used during the Deepwater Horizon (DWH) oil spill and, where possible, the effect of those technologies on GoM ecosystem services (Task 6 of the Statement of Task; see Chapter 1). Figure 4.1 illustrates the U.S. government’s estimate of the fate of the spilled oil.
Given the magnitude, location, and depth of the DWH oil spill, multiple response technologies were applied at scales never before attempted. The goal of response efforts was to contain, remove, and remediate the oil at sea prior to its transport into sensitive wetland and intertidal regions (where cleanup is particularly difficult and costly) by employing a wide array of technologies, including mechanical operations to contain and recover the spilled oil, chemical treatments to disperse oil before it reached sensitive coastal habitats, and a number of shore-based mitigation strategies. In the case of chemical dispersants, the 1.8 million U.S. gallons used in surface and subsurface applications (Federal Interagency Solutions Group, 2010) elicited public concerns about their potential impacts on ecosystems and the services they provide.
To prevent oil from making landfall, several berms and reaches were constructed near shore in Louisiana at a cost of $220 million (National Commission on the BP Deepwater Horizon Oil Spill and Offshore Drilling, 2011). To clean oil from beaches, sand washing and surf washing were performed. To physically remove oil from marshes, manual labor, floating machinery, and manual cropping of oiled vegetation were employed.
Natural processes, such as abiotic weathering and microbial degradation, also played a major role in oil removal. The monitoring of natural recovery processes after an oil spill without active intervention (natural attenuation) must be considered a valid response option under certain circumstances. If oil has impacted a sensitive shoreline such as a wetland, which precludes the use of invasive cleanup operations, then natural attenuation would be the response of choice once accessible floating oil has been removed using absorbents. For areas in which a spill is logistically inaccessible for reasons of remoteness (e.g., the Arctic), stormy weather, or lack of equipment and manpower, natural attenuation might be the only option available.
This chapter reviews the suite of spill response technologies used during the DWH oil spill event, discusses how they were applied in response to the spill, presents the effectiveness and risks associated with their use, and, where possible, assesses their potential impacts on the GoM ecosystem and its services. The discussion begins with potential responses at sea that can
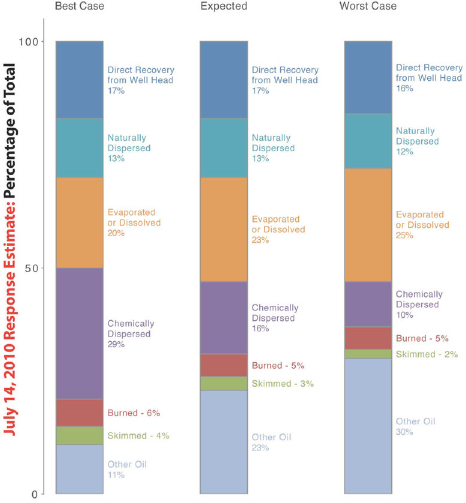
FIGURE 4.1 Deepwater Horizon oil budget calculator. SOURCE: Federal Interagency Solutions Group, 2010.
prevent adverse impacts, moves to potential responses on and near the shore that can prevent adverse impacts, and then moves to methods to treat impacted shoreline habitats. The final section deals with natural attenuation—the natural fate of oil in the marine environment.
Understanding of the efficacy of response technologies used during the DWH oil spill is rapidly evolving, and papers and reports continue to be published. The results presented in this
chapter may be verified, challenged, or negated by further findings. The extrapolation of results from lab-based studies to ecological relevance is tenuous at times, but lab-based studies do provide information about the potential cause and level of effect under certain circumstances. Studies of long-term changes in populations and community structure and function under the Natural Resource Damage Assessment (NRDA) remain undisclosed or have not been completed. Given these constraints, the committee acknowledges that it may never be possible to fully understand how some technologies have affected ecosystem processes and functions.
CHEMICAL DISPERSANTS
Chemical oil dispersants are proprietary mixtures of surfactants and solvents that are applied directly to an oil spill in order to reduce the natural attractive forces within the oil. The use of chemical oil dispersants offshore can be effective in preventing heavy oiling of sensitive coastal environments, such as beaches and wetlands, and in mitigating risks associated with marine and terrestrial wildlife coming into direct contact with a surface oil slick. Some researchers (Lehr et al., 2010) have estimated that the use of dispersants helped to keep 500,000 barrels of the DWH oil away from the highly productive and sensitive coastal areas, but there is no feasible way to verify this estimate.
When an oil slick is sprayed with dispersant and exposed to mixing energy, typically from wave action, some of the oil is broken up into small droplets, which may then become entrained in the water column (Li et al., 2008, 2009a,b; Lunel, 1995). Because the chemically dispersed oil droplets may be small enough to be neutrally buoyant, diffusion and advection transport processes are expected to dilute the plume over a large volume to concentrations below toxicity threshold limits—a key objective for the use of dispersants. Furthermore, as microbial degradation of oil spilled at sea primarily occurs at the oil-water interface, oil biodegradation rates should be enhanced, provided that the dissolved oil concentration is not so large as to be toxic to the microbes. At the large scale, the overall biodegradation rate is increased when dispersants are used effectively (Venosa and Holder, 2007; Venosa and Zhu, 2003). In addition, small droplets enhance dissolution of soluble and semivolatile compounds into surrounding waters, resulting in fewer airborne volatiles, which enhances safety for cleanup workers. However, one consequence of dispersant use is that oil concentrations may increase in the water column and potentially increase exposure for pelagic species.
Finding 4.1. Chemical dispersants have long been applied to oil spills to break up oil into smaller droplets. From a purely physical perspective, chemical oil dispersants can reduce oil concentrations at the surface by increasing horizontal and vertical mixing of the oil. This tends to enhance biodegradation and mitigate the adverse effects of the oil at the surface including the reduction of vapors (thus enhancing the safety of response personnel). However, the overall volume of ocean impacted by the oil will increase, which may have adverse effects especially for subsurface biota.
Use of Dispersants During the DWH Oil Spill Response
Approval and Monitoring of Dispersant Application
About 1.8 million gallons of dispersants were used during the DWH oil spill response. Both product formulations used in the GoM—Corexit 9527 and 9500—were approved by the U.S. Environmental Protection Agency (EPA) under the National Contingency Plan for the treatment of oil spills. A Centers for Disease Control and Prevention report (CDC, 2010) concluded that “because of the strict guidelines that must be followed to utilize dispersants, it is unlikely that the general public will be exposed (directly) to (the) product.” The report further stated that “ingredients are not considered to cause chemical sensitization; the dispersants contain proven, biodegradable and low toxicity surfactants.”
The use of chemical dispersants at the DWH oil spill site was regulated under the Clean Water Act. During the spill response, approximately 1.8 million gallons of Corexit brand chemical oil dispersant were applied (Figure 4.2): 1.07 million gallons of Corexit 9527 and 9500A at the surface and 772,000 gallons of Corexit 9500 via subsurface injection (Federal Interagency Solutions Group, 2010). Figure 4.3 shows the geographical area where approximately 90 percent of the aerial surface dispersant spraying occurred.
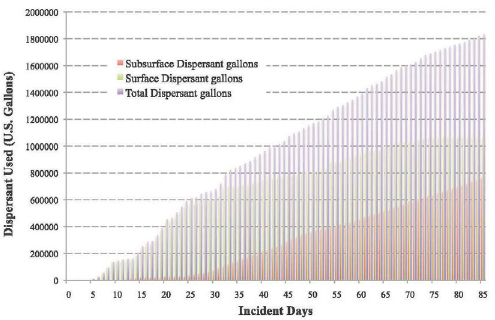
FIGURE 4.2 Cumulative surface and subsurface dispersant use by day of spill response.
SOURCE: Federal Interagency Solutions Group, 2010.
FIGURE 4.3 Region in which 90 percent of aerial dispersant spray passes occurred. SOURCE: Created by O’Brien’s Response Management.
Dispersant Application Methods
Chemical dispersants are most often sprayed directly onto a surface oil slick from an airplane or ship. During the DWH oil spill response, chemical oil dispersants were also injected directly into the stream of oil and gas flowing from the damaged wellhead located 1,522 m below the surface of the ocean. The impacts of these application methods are discussed in detail below.
Subsurface Injection of Dispersant at the Wellhead One of the most innovative and controversial aspects of the immediate response to the DWH oil spill was the injection of 770,000 gallons of Corexit 9500 at the wellhead. Some experts questioned the necessity of injecting dispersants at the wellhead to keep some of the oil from reaching the surface. A plume of oil formed at depths between 1,100 and 1,200 m below the surface (Reddy et al., 2011). Some researchers (e.g., Peterson et al., 2012) have suggested that the subsurface dispersants had little impact in dispersing the spill or preventing oil from reaching the surface. They argue that the turbulence
in the oil jet was sufficient to induce massive dispersion. They offer anecdotal evidence from the BP videos taken at the wellhead during the DWH spill that the dispersant injection wand sometimes missed the main oil plume, therefore making the dispersant ineffective.
Paris et al. (2012) argue that the application of subsurface dispersants did little to alter the vertical distribution of oil. However, their argument depends heavily on the use of an algorithm to estimate the oil droplet size that is based on oil-in-water emulsions with debatable relevance to the blowout problem. Furthermore, their calculated droplet size for the DWH oil spill in the absence of dispersant is so small that it could not explain the observed surface slick that formed immediately above the well.
The American Petroleum Institute (2012) provides an argument for the efficacy of subsurface dispersants. The Institute provides aerial photographs taken during the DWH oil spill that show that the surface slick nearly disappeared when subsurface dispersant injection was initiated, then reappeared when the injection was stopped. Other evidence for the efficacy of subsurface dispersant use is provided by Johansen et al. (2013) and Brandvik et al. (2013), who describe studies of droplet size in simulated blowout plumes conducted in a 6-m laboratory tank. Their results show roughly a threefold decrease in modal droplet size when dispersant is applied at ratios used in the DWH oil spill. From the laboratory data, they develop an algorithm for droplet size based on a modified Weber law that accounts for the effects of dispersant. The algorithm gives a reasonable fit to their laboratory studies as well as several other relevant studies. This algorithm suggests that dispersant application in the DWH oil spill dramatically reduced the droplet size. As discussed earlier in this chapter, the formation of smaller oil droplets would enhance oil biodegradation rates.
Of course, even if subsurface dispersant application does keep more oil below the surface, the concern is that dispersing the oil will simply increase the concentration of bioavailable oil in one environmental compartment, the water column, and decrease it in another, the shoreline. Despite this obvious tradeoff, the Presidential Oil Spill Commission concluded that dispersant application had reduced the overall risks associated with the spill (National Commission on the BP Deepwater Horizon Oil Spill and Offshore Drilling, 2011). The alternative to using dispersants would have been to allow additional oil from the Macondo wellhead to impact GoM coastal marshes and beaches.
Dispersant Effectiveness The effectiveness of a dispersant for the treatment of oil spilled at sea is largely dependent on a number of physicochemical factors such as oil properties, turbulence (e.g., waves for dispersant applied at the surface), temperature, oil weathering, salinity of the sea water, and the hydrophilic-lipophilic balance of the dispersant (Chandrasekar et al., 2005, 2006; Mukherjee and Wrenn, 2009).
Biodegradation of Chemically Dispersed Oil Dispersants can enhance the initial rate of petroleum hydrocarbon degradation by being the first substrate utilized by the hydrocarbon-degrading bacteria to grow and colonize dispersed oil droplets (Varadaraj et al., 1995). However, the observed effects of chemical dispersants on the rate of oil biodegradation have varied among studies (NRC, 2005a). Chemical dispersion stimulated biodegradation rates in some
studies (Swannell and Daniel, 1999; Traxler and Bhattacharya, 1978), but chemical dispersion had no effect or inhibited biodegradation in others (Foght and Westlake, 1982; Lindstrom and Braddock, 2002).
Corexit concentrations of 1 to 10 milligrams per milliliter, which were likely encountered during the actual dispersant application, promoted the biodegradation of DWH oil in laboratory tests, but also caused significant reductions in production and viability of the hydrocarbon degraders, Acinetobacter and Marinobacter (Hamdan and Fulmer, 2011). Marinobacter was most sensitive to the dispersant, with nearly 100 percent reduction in viability and production. Dispersant concentrations within the immediate vicinity of the wellhead may have reduced the capacity of some microorganisms in those waters to bioremediate the dispersed oil because of the elevated concentrations of dispersant and oil above toxicity threshold limits, which became reduced as the plume moved farther away.
Attempts have been made to predict the rate of oil biodegradation in the environment from the results of kinetic models based on laboratory data (Venosa and Holder, 2007; Zahed et al., 2011). Lee et al. (2011) highlighted two important challenges to consider when designing experiments. The first challenge is to conduct tests at the low dispersed oil concentrations representative of field conditions where rapid dilution occurs. Many previous biodegradation studies were conducted at unrealistically high concentrations of dispersed oil in closed microcosms. Studying dispersed oil biodegradation at concentrations several orders of magnitude above expected at-sea concentrations in closed systems could limit biodegradation rates and total degradation by exhausting the available nutrients. Some researchers have attempted to address this by adding nutrients to the system, but that action can lead to unrepresentative modification of the microbial community.
The second challenge is to maintain a stable dispersion within the test system. Under normal sea-state conditions, oil droplets have a strong tendency to become more dispersed over time, especially for subsurface plumes where droplets can freely move in all three dimensions. Even for surface slicks, Langmuir cells (Langmuir, 1938; Leibovich, 1983) and the action of waves (Boufadel et al., 2006; Elliot et al., 1986) can temporarily overpower the oil buoyancy and drive the droplets several meters down into the water column, thus dramatically reducing the concentration and the chance of coalescence. It is difficult to simulate these kinds of spreading mechanisms in a laboratory setting. Until these issues are addressed in experimental research, actual rates for the biodegradation of chemically dispersed oil at sea will remain unknown.
Biodegradation of Dispersants The biodegradation of dispersants and the surfactants used in them are relatively well studied (Baumann et al., 1999; Garcia et al., 2009; Lindstrom and Braddock, 2002; Liu, 1983; Odokuma and Okpokwasili, 1992; Una and Garcia, 1983). In general, results indicate that most surfactant formulations are readily biodegraded under aerobic conditions by marine bacteria (Lee et al., 1985; Liu, 1983; Una and Garcia, 1983), with the rate of biodegradation under anaerobic conditions being much lower (Berna et al., 2007; Ying, 2006).
Different types of surfactants, and even individual surfactants of the same class, can biodegrade at different rates, depending on environmental conditions and the structural complexity of a dispersant’s chemical branching. For example, Una and Garcia (1983) observed extensive
biodegradation of the ethoxylated nonionic surfactants used in Corexit 9527 and 9500 in pure cultures of bacteria isolated from an estuary. These nonionic surfactants are sorbitan mono- and tri-(9Z)-9-octadecenoate poly(oxy-1,2-ethanediyl) derivatives, known commercially as Tween 80 and Tween 85. The authors also noted that poor biodegradation (<20 percent) of Span 80, the unethoxylated nonionic surfactant constituent in Corexit products, may have been caused by substrate inhibition due to the extremely high surfactant concentration (5 grams per liter), which likely would have impacted the integrity of bacterial membranes (Garcia et al., 2009). Campo et al. (2013) analyzed biodegradation of Corexit 9500 and Louisiana sweet crude (similar to the Macondo oil in properties and hydrocarbon constituents) at both sea surface and seafloor temperatures, and they observed complete degradation of specific surfactants such as dioctyl sodium sulfosuccinate (DOSS) within 28 days.
Finding 4.2. Existing studies of the biodegradation rate of dispersants alone show that they do degrade, although at rates that depend strongly on the concentration of dissolved oxygen and the composition of the surfactant. The biodegradation rate of chemically dispersed oil can also vary widely, but some, if not all, of this variation reflects the lack of realism in the underlying experiments. Further studies are needed that use realistic initial dispersed oil concentrations and avoid boundary effects that increase oil-dispersant concentrations.
Toxicity and Bioaccumulation of Dispersed Oil, Dispersants, and Surfactants
Although a great deal of work has been done under controlled laboratory conditions to evaluate the toxicity of chemical oil dispersants and dispersed oil in the marine environment, differences in results have been observed because of factors such as species-specific sensitivity, the particular dispersant formulation used, and experimental conditions (George-Ares and Clark, 2000). For example, with exposure of Atlantic herring (Clupea harengus) to dispersed oil, toxicity has been found to be related to concentration, duration of exposure, and life stage exposed (McIntosh et al., 2010). Some controversy remains over the use of dispersants (Chapman et al., 2007) because they may elicit toxic responses. Corexit 9500 and Corexit 9527 result in low to moderate toxicity to most aquatic species in laboratory tests (George-Ares and Clark, 2000). However, there is evidence that oil dispersed as small droplets in the water column might be more bioavailable, and therefore more toxic, than the oil alone. For example, fish studies with newly hatched mummichog (Fundulus heteroclitus) in 96-hour static renewal tests have shown that dispersant additions induced changes in aqueous concentrations of polycyclic aromatic hydrocarbons (PAHs) that affect larval survival, body length, or ethoxyresorufin-O-deethylase activity, an indicator of contaminant uptake (Couillard et al., 2005). At 0.2 gram per liter, the addition of dispersant caused twofold and fivefold increases in the concentrations of total PAHs and high-molecular-weight PAHs (three or more benzene rings), respectively. Recent studies have focused on the evaluation of chronic effects from short-term exposures to dispersants and dispersed oil (Hannam et al., 2009; McFarlin et al., 2011b). Much consideration has also
been given to the environmental persistence of dispersants, dispersed oil, or dispersant byproducts at low concentrations (Lee et al., 2011).
Fuller et al. (2004) conducted a study using two fish species, Cyprinodon variegatus and Menidia beryllina, and one shrimp species, Americamysis bahia (formerly Mysidopsis bahia), and the luminescent bacterium, Vibrio fischeri, to evaluate the relative toxicity of test media prepared with dispersant, weathered crude oil, and weathered crude oil plus dispersant. The study results indicated that the toxicity of chemically dispersed oil preparations was equal to or less than that of the oil alone. However, the real source of toxicity was a function of the soluble crude oil components, which would likely not persist over time. An EPA study by Hemmer et al. (2010) of the toxicity of Louisiana sweet crude (LSC), chemical oil dispersants, and chemically dispersed LSC on Menidia beryllina and Americamysis bahia (two species used by Fuller et al., 2004) revealed that the toxicity of the dispersant alone was lower than that of LSC or dispersed LSC, which both showed moderate to high toxicity. Similarly, Milinkovitch et al. (2011) found no significant difference between the toxicity of naturally and chemically dispersed oil when looking at a series of biomarker responses in the gills of golden grey mullet (Liza aurata).
Judson et al. (2010) used in vitro (cell-based) assays and concluded that, although some dispersants showed low potential for endocrine disruption, most (including Corexit) did not show any significant effect. Research into the toxicity of a series of chemical oil dispersants (the Corexit series of dispersants were not tested) on two Indo-Pacific branching coral species found that the two corals were sensitive to chemical oil dispersants and chemically dispersed oil, but that toxicity decreased sharply with dilution (Shafir et al., 2007). During this study, the water-soluble oil fraction was also tested and showed relatively low toxicity, but naturally dispersed crude oil was not evaluated. Using the frog (Xenopus) embryo teratogenesis assay, Smith et al. (2012) reported a range of abnormalities due to Corexit 9500A (concentration range: 1 to 1,000 microliters [µL] per liter) that included edemas of the pharynx, thorax, and eye, and incomplete and reversed gut coiling.
Fish are highly mobile and will encounter dispersed oil. Greer et al. (2012) showed that brief exposures of Atlantic herring (Clupea harengus) embryos to low concentrations of chemically dispersed Alaskan North Slope and Arabian light crude oils for 2.4 hours could induce blue-sac disease and reduce the percentage of normal embryos at hatching. Furthermore, toxicity increased with exposure time. However, fish are not expected to be exposed for long time periods to dispersed oil, because of natural dilution processes under dynamic open ocean conditions (Lee et al., 2011).
The media have raised concerns about the potential effects of the crude oil and dispersants on the food web dynamics in the GoM (Mascarelli, 2010). Microcosm experiments performed in situ in North Inlet Estuary near Georgetown, South Carolina, using DWH oil and a Texas crude oil noted a decrease in chlorophyll a in phytoplankton as crude oil concentration increased from 10 to 100 microliters per liter (Gilde and Pinckney, 2012). Researchers interpreted this change as a decrease in biomass rather than in the pigment itself, because of the short duration of the experiments. Diatom, cyanobacteria, euglenophyte, and chlorophyte abundances were unaffected or increased with increased oiling, whereas cryptophyte abundance decreased. The authors suggested that oiling could result in changes in phytoplankton
community composition within salt marsh estuaries impacted by the DWH oil, thereby affecting higher trophic levels such as zooplankton, which selectively feed on phytoplankton that might be killed by the oil (Gilde and Pinckney, 2012). Graham et al. (2010) found that δ13C depletion, used as a tracer of oil-derived carbon in mesozooplankton and suspended particulate samples in near-surface and bottom waters from four stations in 8- to 33-m water depth in the northern GoM, corresponded with the arrival of surface slicks from the DWH oil spill and demonstrated that carbon from the spill was incorporated into both trophic levels of the planktonic food web.
Ortmann et al. (2012) conducted mesocosm experiments to determine the response of the surface microbial community off Alabama to Macondo well oil and Corexit 9500A. The results suggest that the oil alone increased the abundance of ciliates (predators of the carbondigesting bacteria), and therefore the likelihood of carbon being transferred up the food web. However, when dispersant or dispersed oil was used, an increase in heterotrophic prokaryotes with a significant inhibition of ciliates ensued, which suggests that the prokaryotes were able to use the dispersant and dispersed oil as a carbon source and that a decrease in the number of grazing microbes was occurring, with a corresponding decrease in carbon transfer to higher trophic levels (although the carbon transfer was not measured). The researchers hypothesized that the addition of dispersant and dispersed oil to the northern GoM from the DWH incident may have reduced the carbon transfer to higher organisms and potentially decreased zooplankton and fish production on the shelf. However, in a previous mesocosm study conducted on the Pacific coast off Canada, Lee et al. (1985) noted that the presence of crude oil dispersed with Corexit 9527 may have reduced the grazing pressure by bacterivores such as ciliates on potential oil-degrading bacteria.
Satellite measurements of algal blooms in the GoM suggest that the spill may have triggered an increase in these blooms. Initial results of changes in the Moderate Resolution Imaging Spectroradiometer (MODIS) fluorescence line height as a proxy for phytoplankton biomass and a source of carbon for trophic transfer1 showed a statistically significant chlorophyll a anomaly in an area exceeding 11,000 km2 in August 2010, about 3 weeks after the Macondo well was capped (Hu et al., 2011). The values of fluorescence line height were higher than in any similar August imagery from 2002 to 2009 (Hu et al., 2011). A second bloom from December 2010 to January 2011 seemed to have been from strong upwelling and mixing events during late fall, but data were insufficient to support or reject the hypothesis that subsurface oil from the DWH oil spill may have contributed to its occurrence (Hu et al., 2011). These studies highlight the need to fully understand the long-term effects of residual oil and dispersed oil in the marine environment.
Dispersant application during the DWH oil spill occurred at a time when birds in the Gulf region were nesting. Some birds and their eggs may have been exposed to oil, Corexit, or dispersed oil. To test the embryotoxicity of Macondo well oil dispersed with Corexit 9500 to a model bird species, 50:1 and 10:1 mixtures of weathered Macondo well crude oil (collected from the GoM) and Corexit were applied to the shells of mallard duck (Anas platyrhynchos)
_________________
1 National Aeronautics and Space Administration (NASA), http://modis.gsfc.nasa.gov/about/.
eggs in ranges of 0.1 to 59.9 mg for the 50:1 mixture, and 0.1 to 44.9 mg for the 10:1 mixture (Finch et al., 2012). No deformities were observed, which is consistent with a previous study using weathered MC252 oil (Finch et al., 2011). It appears that the weathering of the oil reduces the potential toxic effects on bird eggs; in previous studies, fresh crude oils have been shown to be embryotoxic and teratogenic (Hoffman and Albers, 1984), because they contain lighter-molecular-weight components, including PAHs (Barron et al., 1999), that can penetrate the eggshell and membrane.
After the DWH oil spill, the EPA issued a number of directives (EPA, 2010) regarding the monitoring of the efficacy of subsurface oil dispersant injection and the fate and transport of dispersed oil and operational shutdown criteria. Based on the results of daily field data reports, the EPA had the authority to suspend the application of dispersants because of a significant reduction in dissolved oxygen (National Incident Command Joint Analysis Group, 2010), high mortality in rotifer toxicity tests, and other indicators related to human health. In support of these mandates, the EPA conducted a series of toxicity experiments to determine the potential threat posed to marine organisms by the large volumes of dispersant being applied during the spill response (Hemmer et al., 2010). The acute toxicity and capacity for endocrine disruption and other biological activity of eight chemical oil dispersant formulations in the presence of Louisiana Sweet Crude oil, at recommended application rates, were evaluated with GoM mysid shrimp, Americamysis bahia, and the inland silverside fish, Menidia beryllina. Acute toxicity testing was conducted using the LC50 metric, which identifies the lethal concentration (LC) at which 50 percent of the test species die over a given period of time. The capacity of the dispersants to act as endocrine disruptors, or engage in other biological activity, was determined using a series of standardized in vitro (cell-based) assays. Overall, the dispersant-LSC oil mixtures were considered to be highly to moderately toxic, depending on the test species and dispersant. Corexit 9500A, which was used for both surface and subsurface application during the DWH oil spill response, was considered moderately toxic for both species. The toxicity of the dispersant alone was lower than that of LSC or dispersed LSC.
In light of the varied and somewhat circumstantial results being obtained for toxicity studies of chemical oil dispersants, George-Ares and Clark (2000) suggested that factors such as toxicity of dispersed oil, dilution and degradation in the environment, presence of species or resources requiring priority protection, potential adverse effects of all response options, and the potential for recovery of sensitive habitats and populations should weigh more heavily in the decision-making process than dispersant toxicity alone.
Some consideration has also been given to the bioaccumulation of surfactants. There is evidence that surfactants of all classes are readily taken up across the gills of some marine animals (EOSCA, 2000) and that nonionic and ionic surfactants (such as those found in most oil dispersants) may be effectively biotransformed and eliminated via the gall bladder (Tolls et al., 1994). A review of the bioaccumulation potential of surfactants conducted by the European Oilfield Speciality Chemicals Association (EOSCA) concluded that although surfactants and their metabolites can be found in aquatic organisms following exposure, there was no evidence to support biomagnification of surfactants through the food web (EOSCA, 2000).
Numerous studies of the toxicity of dispersants and dispersed oil, some with conflicting
or inconclusive results, are ongoing. The body of evidence, however, implies that, in general, dispersant and dispersed oil biodegrade in the marine environment. The fate of dispersant surfactants in laboratory studies is highly dependent on the concentration and chemical characteristics of the surface-active compounds, the microbes available, and the methods used to monitor biodegradability. The degradation of surfactants in combination with the crude oil hydrocarbons remains a challenge in analytical chemistry.
Finding 4.3. There is some evidence that chemically dispersed oil and some dispersant compounds are toxic to some marine life, especially those in early life stages. There is contradictory evidence as to whether chemically dispersed oil is more or equally toxic to marine life compared to undispersed oil. The use of dispersants does reduce the amount of oil available to reach shorelines and shallow water environments and impact additional marine life. These facts may be weighed when considering the net environmental benefit to having used dispersants to respond to the DWH oil spill.
Dispersant Degradation During the DWH Oil Spill
The Operational Science Advisory Team (OSAT, 2010) chose the chemical indicators 2-butoxyethanol, dipropylene glycol n-butyl ether (DPnB), propylene glycol, and dioctyl sodium sulfosuccinate (DOSS) of Corexit dispersants 9500 and 9527 to monitor the expanse of Corexit and the DWH dispersed oil. Corexit chemical indicators were observed in 79 percent of offshore water column samples in a wide range of concentrations, but only in a small fraction (<10 percent) of all other (near-shore and inland) samples. Propylene glycol was the only dispersant indicator detected in the near-shore sediments (OSAT, 2010). A research team studying the distribution of propylene glycol, 2-butoxyethanol, and DOSS found at near-shore and inland water sampling sites of Orange Beach, Alabama, between September 2010 and January 2011 concluded that these Corexit constituents probably did not originate from dispersant use during the DWH oil spill, but were likely related to stormwater discharge (Hayworth and Prabakhar Clement, 2012).
DPnB was the most commonly detected dispersant indicator. It was found in 57 of the 60 water samples collected offshore, and its concentrations decreased over time. Peaks in DPnB were found in surface waters and also between 1,100 and 1,300 m. Water samples from 200-m depths had DPnB concentrations ranging from 0.0170 to 113.4 micrograms per liter, with a mean of 4.3 micrograms per liter (OSAT, 2010). Concentrations of DPnB decreased over time after dispersant application stopped in mid July; all values were less than 5 micrograms per liter by July 30 (OSAT, 2010).
Detection of the DOSS Corexit indicator was measured during dispersant injection and up to 300 km from the wellhead 64 days after injection at the wellhead had ceased (Kujawinski et al., 2011). The majority of the DOSS associated with the subsurface injection of Corexit 9500 moved into 1,000- and 1,200-m depth rather than rising to the surface. Kujawinski et al. (2011) concluded that although biodegradation might have occurred, the most significant factor
causing a decrease in DOSS concentration at depth was dilution. Campo et al. (2013) observed rapid biodegradation of DOSS with and without the presence of LSC by microbial cultures isolated from the surface of the GoM, but they also observed a significant lag in the biodegradation response from the cultures isolated from deep GoM. The authors hypothesized that, because DOSS is a xenobiotic compound in that environment, they would not find cultures able to degrade DOSS rapidly at 5°C, except after a long adaptation period (Campo et al., 2013).
The Fate of Chemically Dispersed Macondo Well Oil
Other than the study of the fate of DOSS (Kujawinski et al., 2011), no studies have specifically investigated the fate of chemically dispersed Macondo well oil (see Box 4.1 for the fate of oil from a comparable spill event). Several studies, however, have investigated the submerged plume of dispersed oil and gas droplets less than several millimeters in size that extended southwest from the wellhead between about 1,000 and 1,200 m below the surface (Camilli et al., 2010; Diercks et al., 2010; Hazen et al., 2010; Kessler et al., 2011; Valentine et al., 2010, 2012). Modeling predicted that the volume of hydrocarbons released by the Macondo well was sufficient to double the bacterial population in the deep plume over an area of 30,000 km2 within the deep-water layer. The modeling results suggested that mixing processes recirculated water containing microbial oil degraders around the wellhead and spill site, causing rapid biodegradation of the suspended oil (Valentine et al., 2012) and decreased oxygen concentrations due to respiratory processes. Chakrabarty et al. (2012) reported that the concentrations of Corexit 9500 used during the DWH response were not toxic to indigenous microbes in the deep-water plume that included bacterial species capable of degrading the dispersant components, which include hydrocarbons, glycols, and DOSS.
BOX 4.1
Fate of Chemically Dispersed Oil from the Ixtoc I Oil Spill
The Ixtoc I well blowout in the Bay of Campeche in the southern GoM off Mexico in 1979-1980 in 50-m water depth expelled about 150 million gallons of oil (more than half of that attributed to the DWH oil spill), yet 5 years later there appeared to be little evidence of residual toxicological impacts (Aldhous and McKenna, 2010). It took almost 10 months to cap the Ixtoc I wellhead, during which time the oil was transported along the shore and as far north as Galveston, Texas. Bulk oil incorporated into the burrows of burrowing beach organisms remained largely intact (undegraded) and reappeared in successive years from wave action in the surf zone (Amos et al., 1983), and asphaltenes, an oil fraction with limited bioavailability, can still be uncovered along the Mexican coastline (Wes Tunnell, unpublished data).
The submerged plume was identified based on fluorescence (Camilli et al., 2010; Diercks et al., 2010; Hazen et al., 2010), light scattering (Diercks et al., 2010), or the concentrations of specific hydrocarbons (Camilli et al., 2010; Diercks et al., 2010; Kessler et al., 2011; Valentine et al., 2010) and was detectable up to 35 km from the MC252 wellhead (Camilli et al., 2010).
Most of these studies also observed a local dissolved oxygen (DO) decrease in the vicinity of the submerged hydrocarbon plume. Camilli and colleagues attributed this decrease, in part, to hydrocarbon interference with the in situ DO probes that were used because data from the Winkler titration tests for DO did not show oxygen depletion within the plume at the time of their study (Camilli et al., 2010). Earlier and later studies in the region showed good agreement between data from the in situ DO probe and Winkler titrations (Kessler et al., 2011; Valentine et al., 2010), suggesting that the rate of aerobic microbial metabolism within the plume was higher than in the surrounding water despite its average temperature of about 5°C (Camilli et al., 2010; Hazen et al., 2010).
Evidence of lower DO concentrations in support of biodegradation of gaseous alkanes (Kessler et al., 2011; Valentine et al., 2010) was obtained based on compositional changes that reflected preferential loss of specific compounds and changes in temperature (for ethane and propane) (Valentine et al., 2010). One study estimated that about 70 percent of the oxygen depletion observed within the plume was due to microbial metabolism of ethane and propane (Valentine et al., 2010). Microbial degradation of other hydrocarbons, including butane and longer-chain alkanes, was modeled and thought to be responsible for the additional oxygen depletion.
Although the media expressed fears that the oxygen depletion within the water column in the vicinity of the Macondo wellhead might cause hypoxic conditions or anoxic “dead zones” and threaten marine organisms, the maximum depletion observed was 40 percent, whereas 60 percent is considered detrimental to fish (Walsh, 2011). Studies are now under way to assess the potential long-term ecological impact of MC252 residual oil buried in deep-sea sediments.
Half-lives for higher-molecular-weight normal alkanes within the plume were estimated to be between about 1.2 and 6.1 days, based on in situ and microcosm data (Hazen et al., 2010). Flocculant material isolated from samples collected within the plume between May 25 and June 2, 2010, were rich in microbes (Passow et al., 2012), oil, and oil degradation products, and bacterial counts were elevated within the plume (Hazen et al., 2010). Microbial genes involved in hydrocarbon degradation were significantly increased (p < 0.05 or p < 0.01) in plume samples, and the relative abundance was correlated with the concentrations of some low-molecular-weight components of the oil, suggesting that the composition of the bacterial community changed in response to the presence of oil (Hazen et al., 2010).
The most abundant bacterial species in samples from within the plume (comprising about 90 percent of sequences) belonged to a single operational taxonomic unit that was closely related to the order Oceanospirillales (Hazen et al., 2010). The species dominance of this simple aliphatic hydrocarbon degrader was observed to shift to Colwellia and a PAH degrader, Cycloclasticus (Abbriano et al., 2011), in samples collected from the same area by another research group about 2 weeks later while oil was still being released from the wellhead (Valentine et al.,
2010). The putative hydrocarbon-degrading bacteria in these later samples were thought to be growing on propane, ethane, and butane (Valentine et al., 2010).
Lu et al. (2011) showed that the DWH oil spill dramatically altered the functional composition and structure of the microbial community in the deep sea. A variety of metabolic genes involved in aerobic and anaerobic hydrocarbon degradation were more highly enriched inside than outside the plume. Various other microbial functional genes that are associated with carbon, nitrogen, phosphorus, sulfur, and iron cycling, metal resistance, and bacteriophage replication were also enriched within the plume. The authors suggested that the indigenous marine microbial communities could have a significant role in biodegradation of oil spills in deep water.
Microbial activity is essential to the maintenance of marine ecosystem health because it controls primary processes—for example, nutrient regeneration, the production of biomass, and the biodegradation/biotransformation of contaminants. Thus, changes in the structure and function of microbial communities may have had a profound effect on the recovery and maintenance of ecosystem services within the GoM. Soon after the spill, the microbial community responded by becoming dominated by hydrocarbon degraders, resulting in reduced community diversity. DNA surveys for bacterial 16S rRNA genes from samples collected in June revealed the dominance of Cycloclasticus and Colwellia, likely degrading propane and ethane preferentially (Kessler et al., 2011; Valentine et al., 2010). Among these were Oleispira antarctica, Thalassolituus oleivorans, and Oliphilus messinensis, all of which are bacteria known to degrade hydrocarbons and tolerate low temperatures that occur in the deep sea. Samples collected later (September 2010) indicated a shift away from these hydrocarbon degraders to methanotrophs, including Methylococcaceae, Methylophaga, and Methylophilaceae. The enhanced abundance of methanotrophs and bacteria containing the particulate methane monooxygenase gene indicated that methane was consumed later in the spill sequence by a different bacterial assemblage (Kessler et al., 2011).
Summary of Dispersant Use During the DWH Oil Spill Response
About 1.8 million gallons of dispersant were used during the DWH oil spill response, of which about 42 percent was injected at the wellhead (Federal Interagency Solutions Group, 2010). The size and composition of the GoM microbial community was altered as microbes responded to the presence of DWH crude oil and dispersed oil, which contained light, readily biodegradable hydrocarbons (Atlas and Hazen, 2011). Bacterial cell density was significantly higher within the deep subsurface plume at 5.51 x 104 cells per milliliter than outside of it at 2.73 x 104 cells per milliliter, when measurements were taken 5–7 weeks after the start of the spill (Hazen et al., 2010). Valentine et al. (2010) used measured oxygen and hydrocarbon component anomalies in the plume several weeks later, along with sample incubation experiments that included dissolved inorganic carbon (DIC) measurements, to deduce that 70 percent of the depleted oxygen was due to microbial respiration (not necessarily growth) of two components of the plume: propane and ethane. They presumed that respiration of other components could
account for the full amount of depleted oxygen, as others demonstrated later (Kessler et al., 2011). The occurrence of natural seeps in the area of the spill may have supported the development and persistence of microbial communities capable of degrading hydrocarbons.
The short- and long-term impacts of dispersants and dispersed oil from the DWH oil spill on the food web and other GoM ecosystem services are still undetermined (see Chapter 5 for discussion of specific examples). Additional research is recommended to enhance understanding of the overall effectiveness and fate of dispersants and chemically dispersed oil. Of particular interest is the role that subsurface dispersant use played in generating smaller oil droplets, which ultimately controlled the volume of oil in the subsurface oil plume. This information is required for the determination of the overall effectiveness of the dispersants and the establishment of operational guidelines (e.g., establishment of optimal dispersant-oil ratios) for their use. The work of Brandvik et al. (2013) and Johansen et al. (2013), specifically focusing on this issue, offers strong evidence that subsurface dispersants can greatly decrease droplet size and the volume of surfacing oil. However, questions still remain concerning the effects of oil temperature and high pressure and the scaling of laboratory results to the real world.
There is also a need for additional research on the potential impacts from the various dispersant remediation strategies available. At this time, impacts to ecosystem services of the deep GoM, such as chemosynthetic productivity and microbial carbonate sequestration as affected by the deep dispersed oil plume (see Chapter 5), and the fate and effect of the major dispersant constituents on deep-water species, remain unresolved.
Finding 4.4. The use of dispersants helped to keep an estimated 500,000 barrels of the oil away from the highly productive and sensitive coastal areas. The use of chemical dispersants was controversial from a public point of view because of concerns about dispersant toxicity, the potential for dispersants to make oil more bioavailable (thus making dispersed oil more toxic than nondispersed oil), and the persistence of dispersants, dispersed oil, or dispersant byproducts. The long-term impacts of dispersants and dispersed oil from the DWH spill on the food web and other ecosystem services of the GoM are still undetermined, and additional research is necessary in this area.
IN SITU BURNING
Burning oil at sea (in situ burning, ISB) is a well-established practice and is considered to be a relatively low-cost, simple method to rapidly remove large quantities of oil at sea without the generation of excessive wastes that would require further disposal (NOAA, 2011a). However, there is limited scientific data and knowledge of the effects of burning oil and how best to perform in situ burns (Fingas, 2011a). ISB can be an efficient mode of removing oil from the ocean, thus reducing the risk of an oil slick impacting vulnerable habitats such as wetlands and other high-amenity shoreline sites (Valentukevičienė and Brandvall, 2008) that provide valuable ecosystem services by habitats such as mangrove stands, salt marshes, and beaches.
ISB has also been shown to be capable of restoring salt marsh ecological structure and
function (Baustian et al., 2010), but it was not employed for this purpose during the DWH oil spill. As of August 2010, little free-floating oil was remaining, and most of the residual oil was found along the marsh fringe, where low-pressure flushing was deemed more appropriate (see Appendix D in SCAT, 2010). ISB is not appropriate for mangroves, and it was not used on sandy beaches, because it “would result in significant adverse habitat impact” (see Appendixes C and D in SCAT, 2010).
Containment and Ignition of Oil
Burning oil at sea may appear to be an easy method to implement, but it is actually a complex process (Fingas, 2011b). A number of tools and techniques aid in the ignition process, such as a simple rag soaked with diesel or other flammable fluid dispensed by a Heli-torch flame thrower suspended from a helicopter. Volatile compounds that evaporate from the slick provide the fuel, and they must be in sufficient concentrations to maintain the burn. The ignitability of the oil on the sea surface is reduced if the oil is emulsified and has high water content; <25 percent water is typically required for ignition (Walton and Jason, 1999). As the lighter fractions of oil evaporate, leaving asphaltenes and heavier fractions that are harder to ignite (Putorti et al., 1994), the slick may become fragmented and spread over a wider area, further complicating the scenario. Containment booms can minimize the spreading and thinning of the oil on the sea surface. Offshore booms usually consist of a flotation cavity (bladder) that protrudes from the water and a skirt that hangs down in the water column. Fire-resistant booms are the obvious choice to contain the oil.
In Situ Burning During the DWH Oil Spill
ISB was selected as a response technology during the DWH event because of the need to supplement skimming operations to capture more oil, the fact that the oil slick was initially far from populated areas, and the understanding that controlled ISB could be conducted safely and effectively to eliminate large volumes of oil with minimal environmental impact (Allen et al., 2011). Oil was contained within a variety of fire-resistant booms, then ignited with a handheld igniter containing gelled diesel in a plastic half-gallon bottle attached to a float and road flare. The application of an open-apex deflection (“funnel”) technique facilitated the burning of oil within a fire boom as newly captured oil was fed into the burn. New, improved techniques and equipment were able to match the changing oil and weather conditions. For example, controlled burning of oil immediately outside the fire boom was found to be advantageous in extending burn duration, while maintaining a reasonably sized burn area (Allen et al., 2011).
The Occupational Safety and Health Administration/National Institute for Occupational Safety and Health air monitoring specialists evaluated the air quality on vessels working near burn sites to safeguard the health and safety of responders. In addition, an EPA Aerostat team evaluated emissions in and around smoke plumes (Aurell and Gullett, 2010). No cases of harmful or prolonged smoke exposure were reported.
Effectiveness of In Situ Burning During the DWH Oil Spill Response
The Controlled In Situ Burn Group conducted 411 oil collection and ignition attempts between April 28 and July 19, 2010, of which 376 burns were large and long enough to be included in the oil budget estimates (OSAT, 2011a). About 220,000 to 310,000 barrels of oil were burned (Allen et al., 2011) within 5 to 24 km from the wellhead, and therefore, that oil did not impact shallow waters, shorelines, and sensitive coastal resources. In comparison to other technologies, according to the oil budget calculator (Figure 4.1), the amount of oil released from the well was estimated at 4.9 million barrels, and 5–6 percent of all of the spilled oil was burned, about double the removal of oil with skimmers (see “Skimmers” section below).
Byproducts Associated with In Situ Burning
Contaminants and byproducts resulting from the ISB were released into the atmosphere. Black particulate carbon in the atmosphere (Perring et al., 2011) was an issue for coastal populations downwind from burn sites. The emissions plume also contained light alkanes and aromatics, but no large concentrations of PAHs (Middlebrook et al., 2011), and were measured across 4 km up to 47 km downwind. The light alkanes and aromatics consumed by ISB were one-tenth of the total evaporated fraction from all the oil spilled (Middlebrook et al., 2011).
Dioxins, which can be detrimental to human health, are formed from the incomplete combustion of organic matter in the presence of chlorine; therefore, they are a possible compound of emissions from ISB of oil at sea. The EPA measured dioxin levels between July 13 and 16, 2010, using the Aerostat protocol involving a sampling device attached to a balloon released into the air above burning oil (Aurell and Gullett, 2010). Dioxin levels were above ambient background concentrations and were similar to a value slightly higher than that associated with residential woodstoves. The risk to workers exposed to dioxins near the burn sites was estimated to be below the level of concern of 1 in 1 million—an EPA risk threshold of one cancer case for every 1 million people exposed. Numerical models (AERMOD) showed air concentrations of dioxins lower onshore than at sea near the burn sites.
Dioxins can settle into sediments, where they degrade very slowly and can be ingested by organisms. Most human exposure to dioxins occurs through dietary intake of animal fats, where the chemicals accumulate. Regarding the risk of exposure from consuming contaminated seafood, Schaum et al. (2010) calculated the maximum factor by which dioxin concentrations in fish could increase due to the in situ burns to be approximately 0.024 pg TEQ/g, which is less than 5 percent of the background levels of dioxin in marine fish (0.5 pg TEQ/g). On the basis of this analysis, even the most at-risk population of subsistence fish consumers who eat on average 300 g of fish per day would not have an increased cancer risk exceeding 1 in 1 million, the top of the acceptable range of risk according to the EPA (Schaum et al., 2010).
Ecological Impacts of In Situ Burning
At present, there are no known effects on marine organisms resulting from ISB during the DWH event.
Finding 4.5. Several technical innovations led to in situ burning being the most successful response method in terms of removing oil from the sea surface offshore (estimated to be as high as 300,000 barrels or 6 percent of the total volume of oil spilled) and thus keeping oil away from sensitive coastal habitats. Although black particulate carbon clouds were clearly visible and subsistence consumers of local fish may have had a slight increase in the risk of cancer (1:1 million), there are no other documented negative impacts at this time. Further long-term research is needed to continue to look for future potential impacts.
SKIMMERS
Skimmer Technology
A skimmer is a mechanical device that removes oil from the surface of the water without the addition of chemicals. Fingas (2011b) provides a recent and detailed overview of skimmers and categorizes them into five types, depending on the physical mechanism employed: sorbent surface, weir, suction, elevating, and submersion. Of these, the skimmers using an oleophilic sorbent surface in the form of discs, drums, belts, brushes, or ropes are most suitable for rougher water that is characteristic of the open sea and coastlines. Skimmers can use many different oleophilic sorbent surfaces. Once the oil is attached to the sorbent surface, a wiper blade or pressure roller removes the oil, after which it is deposited into a barge or other type of container.
The recovery rates achieved by skimmers depend on oil properties, slick thickness, weather (especially wave conditions), and the size of the skimmer. At the large end, a skimmer like the HOSS Barge2 can recover about 30 barrels per minute under ideal conditions. Sustained over a full 24-hour day, this would amount to 40,000 barrels per day, but conditions are rarely perfect, so the recovery rates in practice are usually considerably less. To increase their efficiency, skimmers are almost always joined to booms deployed in the shape of a “V” to funnel and concentrate the oil.
Booming and recovery of oil with skimmers has been considered the first option of choice in response operations because this approach can often recover substantial volumes of oil before it has a chance to strand onshore or spread. Furthermore, unlike some other cleanup methods, skimmers have little potential negative environmental impact. They do not introduce any chemicals into the environment, nor do they physically disturb the seabed or coastlines under normal operations. Some skimmers recover a little seawater along with oil. There are potential
_________________
2http://www.myfloridahouse.gov/FileStores/Web/HouseContent/Approved/Web%20Site/uploads/documents/energy_exploration/Clean%20Gulf%20Associates.pdf.
risks of entrapping smaller marine creatures and colliding with or physically damaging larger species; however, such risks are small, particularly in light of the fact that the creatures would have to be in close contact with the oil and would already be at much higher risk of acute or toxic oil impacts. Some skimmers have trained observers onboard to recover any entrained sea turtles and to report impacts.
Skimmers in the DWH Oil Spill
During the DWH oil spill, skimmers were operated offshore, in bays, marshes, protected waters, and along beaches (USCG, 2011). Details regarding skimmer operations during the DWH oil spill are difficult to obtain because no accurate records were kept of the number or types of skimmers deployed or the volume of oil recovered. The only readily available information comes from the federal government’s Restore the Gulf website,3 which includes occasional press releases and summaries known as “Operations and Ongoing Response” reports. The official government estimate of recovery by the skimmers is 2–4 percent of the total 4.9 million barrels of oil released (Figure 4.1), which translates to an estimated 147,000 barrels. This is a calculated value because the Incident Command only tracked the total volume of recovered oily water.
Taking into account errors in the assumptions used in the data analysis, skimmers likely recovered somewhere between 75,000 to 300,000 barrels of oil during the DWH oil spill (Graham et al., 2011a). Several reasons likely contribute to this low oil recovery rate, including the following: the encounter rate was low due to the expanse of the spill, the oil was likely diluted and dispersed by the time it reached the sea surface, cleanup vessels had to be directed by spotter aircraft, it was difficult to distinguish reasonably thick slicks from oil sheen, and containing and concentrating a slick with booms for skimming are time-consuming tasks.
Finding 4.6. Skimmers can be an effective means of recovering oil with little impact on the environment. However, given the diluted and dispersed nature of the DWH oil spill, skimmers were only able to capture between 75,000 to 300,000 barrels of oil. Nonetheless, skimming prevented this oil from reaching sensitive coastal ecosystems and impacting the services they provide.
NEAR-SHORE AND ONSHORE PROTECTION FOR SHORELINES
The construction of obstacles to prevent oil from reaching inland of shorelines is a common practice in oil spills (Davidson et al., 2008) and generally falls into three subcategories of fixed barriers: sand barriers (berms), sediment-filled containers, and rigid structures (booms). Diversion of a watercourse to repel oil from the shoreline was also a response strategy implemented during the DWH oil spill.
_________________
Berms in Response to the DWH Oil Spill
The western side of Dauphin Island, Alabama, is of low elevation (less than 2 m above sea level), and a 2.5 km portion of it (known as the Katrina Cut) was eroded by hurricane Katrina. It was argued that filling the 2.5-km stretch would stem the influx of oil from the GoM into Mississippi Sound, while decreasing the salinity in the sound, which would help to maintain the oyster population (Webb et al., 2011). Approximately 235,000 tons of sediment were used during the project between July 2010 and April 2011. The inlet to Little Lagoon was filled with 4,500 m3 of sand. This manipulation of the shoreline at Dauphin Island prevented oil from washing over the island, prevented oil deposition under elevated houses, and blocked the oil from entering the Mississippi Sound (Douglass et al., 2011). Two additional sand berms were built. The first one was approximately 4 km long and was completed in several days. It was not extended over roads or driveways, but enough material was put in place so that bulldozers could fill these gaps, if necessary. The second berm was south of the first, at the highest elevation of the dry beach, with the goal of confining any stranded oil to the beach. A total of 267,000 m3 of sand was placed on the island between May and July 2010. Weather conditions during the summer of 2010 were such that sand accretion occurred south of the second barrier.
A second berm-related project involved the placement of massive berms in Louisiana that consisted of six “reaches” totaling 64.5 km in length. By the time BP capped the well on July 15 (day 44 of the berm construction project), Louisiana’s contractor estimated that 6 percent of the total project had been completed. It took 5 months to build roughly 16 km of berms, at a cost of about $220 million (Rudolf, 2010). The more than 15 million m3 of dredged sand came from sources or locations that were not proposed for future coastal restoration, and of this, more than 9.2 million m3 came from the Mississippi River.4 Other sources were Pass a Loutre Hopper Dredge Disposal Area, Pilottown Anchorage, Cubit’s Gap, South Pass, and Hewes Point (north of Chandeleur Islands). Estimates of how much oil the berms collected vary, but none collected much more than 200 tons. On November 1, the state of Louisiana announced plans to convert the berms into part of a long-term coastal restoration project.
The installation of berms changes barrier island sediment transport, causes a loss of sand, and adversely impacts benthic and pelagic flora and fauna (Martínez et al., 2012). The extent of impacts from the berm operations conducted during the DWH oil spill is unknown.
Dynamic barrier islands shift with sediment transport mechanisms, especially those associated with summer tropical storms. As a part of a long-term investigation of the area, the U.S. Geological Survey conducts periodic surveys of Chandeleur Islands, Louisiana (Miselis et al., 2012). The most recent survey of the area was completed just 1 month prior to Hurricane Isaac in 2012. Subsequent data from airborne optical sensors that determine volume of lost sand indicated that most of the oil spill mitigation sand berm created in 2010–2011 was swept away in the storm.5 A seafloor survey planned for 2013 will help to pinpoint where lost sediment
_________________
4 Coastal Protection and Restoration, State of Louisiana, http://coastal.louisiana.gov/index.cfm?md=pagebuilder&tmp=home&pid=131.
5http://coastal.er.usgs.gov/hurricanes/isaac/phtot-comparisons/ and http://www.nola.com/hurricane/index.ssf/2012/09/isaacs_surge_waves_wiped_out_b.html.
was deposited and whether or not it will be naturally available to the islands for post-storm recovery.
An evaluation of the efficacy of the Louisiana berms project (and the other oil spill response strategies used during the DWH oil spill) by the staff of the Oil Commission found that the berms were not an effective response technology based on a cost-benefit analysis. They concluded that the scope and scale of the berms proposed were far too extensive and could not be designed, approved, and built in the time needed to be effective. The value of the berms as a spill response technology depends on how much oil they trapped. Estimates vary, but none of the estimates is much greater than 1,000 total barrels (Graham et al., 2011b).
Booms
Onshore booms create barriers that keep oil from affecting sensitive areas and from intruding further inland. There are three main types. A shore-sea deflection boom for highcurrent areas has an air tube that rides on one or two water-filled tubes. Once deployed and anchored at the selected location, water is pumped into the lower tubes to form a skirt. This water acts as ballast when the boom is floating. When the boom is grounded, these flexible tubes follow the sediment surface to form a seal. Oil is deflected toward pooling areas where it can be recovered from land (USCG, 2011). The second type is a containment boom that is used to surround pooled oil so it can be removed by skimmers. This type of boom can also be used as a barrier (protection) when laid parallel to the shore to prevent oiling of sensitive areas (USCG, 2011). The third type is a snare (sorbent) boom, which is constructed of a long fabric sock filled with oleophilic material, or a series of oleophilic polypropylene “pompoms” tied to a line. Unlike hard booms, sorbent booms do not have an attached skirt. A sorbent boom is typically anchored parallel to the shoreline to absorb oil slicks as they wash toward the shore.
Use of Booms During the DWH Oil Spill
During the DWH oil spill, booms were extensively used to protect sensitive coastal marshes and were later removed. About 1.16 million m of hard (containment) boom and 2.95 million m of sorbent boom were deployed6 at critical points to protect wildlife refuges, bird nesting areas, estuaries, beaches, marshes, and other environmentally sensitive and economically significant lands throughout the Gulf Coast. All of the hard booms on shorelines in Mississippi, Alabama, and the Florida Panhandle were removed by September 7, 2010, and they were inspected, cleaned, repaired if possible, and stored at sites along the Gulf Coast for further redeployment should the need arise.7
Response crews deployed more than 2 million feet of containment boom in Louisiana, secured by Danforth-style anchors, which were designed to embed in the sediment and collapse
_________________
6http://www.restorethegulf.gov/release/2011/08/19/operations-and-ongoing-response-august-17-2011.
7http://www.restorethegulf.gov/release/2010/09/07/all-operational-hard-boom-removed-mississippi-alabama-and-florida-panhandle.
flat when not in use. Upon boom retrieval, it was discovered that some of the anchors remained embedded in the sediment. The U.S. Coast Guard commissioned a net environmental benefit analysis (NEBA) to determine the tradeoffs, including the potential loss of ecosystem services, associated with the recovery of the anchors. The June 2011 Orphan Anchor report (Gulf Coast Incident Management Team, 2011) noted that 46 anchors were buried at an average depth of 1.9–2.1 m, where the risk of damage to personal and commercial vessels was extremely low. There had been no reports of accidents involving these anchors, and decomposition of the steel and galvanized steel components was not considered to be a risk to humans, aquatic life, or the environment. The NEBA team recommended that leaving the remaining buried anchors in place to naturally degrade was the best option (Gulf Coast Incident Management Team. 2011). Apart from the buried anchor information, no additional data on the impact of booms on local habitat or ecosystem services exist.
Hydrology Modification via Mississippi River Diversions
The Davis Pond Freshwater Diversion was built in 2004 to reduce salinity throughout the length of Barataria Bay, enhance fisheries, and add additional nutrients and sediment to the area wetlands.8 The diversion was opened by the state of Louisiana on April 30, 2010, during the DWH oil spill to prevent oil from contaminating the marshes of Barataria Bay (Bianchi et al., 2011). The initial flow of 113 m3/s was increased to 212 m3/s on May 7 and reached its full capacity of about 300 m3/s on May 10—21 days after the DWH explosion.9 In July, the discharge rate was generally maintained above 200 m3/s. By September, discharge was reduced to <25 m3/s. The rapid and extreme changes in salinity caused 50 percent mortality in oyster beds in Louisiana (Food Channel Editor, 2011). Reductions of oyster recruitment and harvest were projected to continue over the next 3 years (Martínez et al., 2012). Mortality of the oyster beds and a decline in the oyster catch for 2012 through 2013 resulted in decreased income for oyster fisherman.
By mid May 2010, in an effort to prevent the intrusion of oil from the DWH spill into fragile coastal ecosystems, a number of freshwater diversions were in effect, many of them to the east of the Mississippi River. The total measurable flow from these diversions was 29,550 cubic feet per second (cfs) (Louisiana Office of Coastal Protection and Restoration, 2010).
• Bayou Lamoque Diversion: Plaquemines Parish, 7,500 cfs (capacity 12,000)
• Davis Pond Diversion: St. Charles Parish, 10,650 cfs (capacity 10,650)
• Violet Siphon: St. Bernard Parish, 200 cfs (capacity 200)
• Caernarvon Diversion: St. Bernard Parish, 8,000 cfs (capacity 8,800)
• Whites Ditch Siphon: Plaquemines Parish, 200 cfs (capacity 200)
• Naomi Siphon: Plaquemines Parish, 1,500 cfs (capacity 1,500)
• West Pointe a la Hache Siphon: Plaquemines Parish, 1,500 cfs (capacity 1,500)
_________________
8http://www.lca.gov/Projects/14/Default.aspx.
9 USGS Davis Pond Freshwater Diversion: http://waterdata.usgs.gov/usa/nwis/uv?295501090190400.
The total water diverted to east of the river was 17,400 cfs, which amounts to less than 1 percent of the water flow down the lower river during flood years 2008–2010 (Allison et al., 2012). Dubravko Justić’s FVCOM model grid (personal communication) was applied to Breton Sound, with incorporation of state-designed freshwater diversions to keep oil from entering Louisiana estuaries.
Finding 4.7. The effectiveness of mechanisms for near-shore and onshore protection of shorelines was negligible in terms of berms and freshwater diversions, but somewhat effective in terms of booms. Well-formed scientific advice was provided in advance that sand berms and diversions would be ineffective in keeping the oil from reaching the wetlands. Moving forward, decision analyses may be needed to refine guidelines and to optimize the effectiveness of various technologies and protocols. Additional research is needed to monitor the overall impacts of these response technologies on ecosystems and the services they provide.
TREATMENT OF SHORELINE OILED BY THE DWH OIL SPILL
Technological efforts offshore do not entirely contain the oil. Oil at sea very often will impact coastal shores if sensitive shorelines are not far away, and if ocean currents are likely to move oil slicks or dispersed oil to those areas. When shorelines and costal habitats are oiled, as they were in the DWH oil spill, shoreline cleanup processes are invoked.
DWH Shoreline Cleanup Assessment Technique (SCAT)
In response to the DWH oil spill, NOAA formed a shoreline response team, Shoreline Cleanup Assessment Technique (SCAT), which relied upon the creation of a consistent data and knowledge base of the extent of oiling along types of shoreline habitat (Santner et al., 2011). Between 15 and 20 SCAT teams were deployed each day after May 4, 2010, and eventually determined the total length of oiled shoreline to be 1,053 miles. Teams of archaeologists (often as a part of the SCAT teams) also conducted field surveys of the entire impacted shoreline to locate known cultural resources, identify sites of potential cultural concern, and monitor operations to provide direction on minimizing impacts to important ecosystem services related to cultural resources.
The shoreline response team also created multiagency Core Groups, one for Louisiana and one for Mississippi, Alabama, and Florida to (a) set treatment priorities, (b) develop treatment recommendations for the various habitat types, (c) monitor the cleanup process, and (d) determine the effectiveness and impacts of treatment. Technical Working Groups were established within the Core Groups to deal with particular habitat issues and treatment recommendations and to establish “no further treatment” (NFT) criteria for sandy shorelines, coastal marshes and mangroves, and human-constructed shorelines. Their reports serve as appendixes to the Core Groups’ final reports, “The Stage III SCAT-Shoreline Treatment Implementation Framework” (Santner et al., 2011). Their findings are described below.
Shoreline Cleanup Strategies After the DWH Oil Spill
Barrier island shoreline treatment following the DWH oil spill typically involved manual or mechanical removal, because mixing, sediment relocation, washing, or chemical treatment each requires some form of regulatory approval or permitting (Owens et al., 2011). The differences in oiling conditions (surface and buried oil or oil layers, stained sand, and sunken oil in the adjacent subtidal waters) and sediment characteristics of the central Louisiana coast and the Gulf beaches of Alabama, Florida, and Mississippi precluded the use of a single technique as a panacea (Owens et al., 2011; Santner et al., 2011). Key considerations in choosing the cleanup methods for beaches were minimization of sand removal and therefore waste generation, minimization of restoration time for amenity beaches used for recreation, and maintenance of beach stability against storms.
The removal of bulk oil, mobile oil in intertidal areas that posed a threat to adjacent habitats or resources, from the intertidal areas along the northern GoM was repeated as necessary in areas of high environmental significance such as turtle-nesting areas, high-use tourist beaches, waterfront parks, and local residential areas (Santner et al., 2011). Amenity beaches that suffered recurring oiling from remobilized oil, or reworking of the shoreline by wind and wave action, were also subject to continued oil removal operations through the 2010–2011 winter (Santner et al., 2011).
Cleanup techniques for salt marshes and mangroves were natural attenuation (discussed below), low-pressure ambient-temperature flushing (to float the oil), mobile vacuum systems, securely deployed containment sorbents or snares, manual removal (on sand or shell substrates only), and vegetation cutting from boats for limited access to Roseau cane marshes only (see Appendix D in SCAT, 2010). In salt marsh habitats where there was little or no risk of repeated oiling, bulk oil removal was only done once on a limited scale, conducted from floating platforms, skiffs, or shallow-draft barges fitted with flushing and vacuum systems, adapted from concrete pump arms (Santner et al., 2011). These floating crafts were able to reach into oiled fringe wetlands to wash and recover mobile oil. When stranded oil was removed, it was primarily carried out by hand with sorbent material and by cutting oiled vegetation. The preferred oil spill response in salt marshes was natural attenuation (Santner et al., 2011).
Manual Removal of DWH Oil
Human-constructed shorelines of riprap, breakwaters, groins (low walls or timber barriers extending into the sea from a beach to check erosion), and jetties were treated after the DWH oil spill through manual removal of bulk oil, followed by washing using a range of temperatures and pressures (Santner et al., 2011). Manual equipment included long-handle hand-mesh and screens, pitchforks with screens, pool nets for surface residue balls along the water line, and mechanical adaptations such as rotary screens and welded mesh screens for extendedreach backhoes working with surface residue balls and patties in water-saturated sand (Owens et al., 2011).
Challenges to Cleaning Sandy Beaches
Re-oiling during storm events (as observed after Hurricane Isaac in August, 2012; Clement et al., 2012) exposed buried oil, and chronic deposition of new oil occurred from oily sediment mats in the lower intertidal and near-shore subtidal zones, which created challenging issues for cleanup operations (Santner et al., 2011). Oil became stranded in the supratidal zone during storms and buried up to 1.5 m deep in beaches, thus requiring extensive excavation, especially on amenity beaches. Further challenges included delineation of buried oil over large areas, both along-shore and cross-shore. Submerged oil mats were widespread in the lower intertidal zone, but they were difficult to clean because they became covered by high water and clean sand over time (OSAT, 2011b; Santner et al., 2011). The use of heavy equipment was limited because of concerns that mechanical methods would result in increased beach erosion or because of restrictions and prohibitions on the use of mechanical equipment on remote barrier islands. An NFT guideline of no visible oil above background on amenity beaches was a difficult, but required, endpoint.
Sand Washing
A fixed sand-washing system, constructed with a shaker sieve to remove large surface residue balls and patties along with debris, as well as two heated wash units, was used on Grand Isle, Louisiana, and proved to be successful (Owens et al., 2011). The small amount of residual oil remaining in the treated sediments from this procedure was removed by surf-washing operations.
Surf Washing
Three field (and several small-scale) demonstrations of surf washing (i.e., enhanced dispersion of oil by the formation of oil-mineral aggregates [OMAs]) were conducted on Grand Isle, Louisiana (Owens et al., 2011). The demonstrations included sediment and water chemistry analysis before and after the event, and sometimes beach profiling, to determine the efficacy of the technique as a sand-polishing step after the removal of bulk oil by sieving or sand washing. Surf washing by relocation of sediment to the lower intertidal zone did not cause significant sediment loss, nor did the technology increase hydrocarbon concentrations in intertidal or subtidal sediments or water. The decision was made to use this technique, and more than 30,000 cubic yards were surf washed within a week. Although one small-scale demonstration in Escambia, Florida, led to an initial decision against surf washing because of strong longshore currents, sediment drift, and little residual oil (Owens et al., 2011), operations were resumed at a later date to treat residual oil in the beach sediments.
Finding 4.8. The impacts of shoreline cleanup operations have not yet been determined. Cleanup efforts were inconsistently successful because of storms, shifting sediments, vari-
able beach habitats, and variable oiling profiles along the northern GoM coasts. Lightly oiled beaches on barrier islands were the easiest to clean and restore.
Salt Marsh Impacts from Cleanup Operations
Physical destruction of marsh habitat during cleanup operations is the most common concern, but virtually all options will cause some damage to marshes during cleanup (Aldhous and Hecht, 2010). Fertilizer, such as phosphorus to encourage regrowth of oiled Spartina alterniflora, was not applied to encourage marsh recovery, probably because of concerns about root damage as described in studies of nutrient overenrichment of marsh habitats (e.g., Darby and Turner, 2008; Deegan et al., 2012; Turner et al., 2009). Root damage would reduce the power of the marsh sod to hold together, resulting in greater erosion (Aldhous and Hecht, 2010).
However, there is evidence to support the application of nutrients to enhance the remediation of oiled wetlands dominated by Spartina alterniflora and Spartina patens (Lin and Mendelssohn, 1998). In the case of the DWH oil spill, it has been suggested that oiled Gulf Coast marshes will recover by natural attenuation because prior research has demonstrated their intrinsic resilience (DeLaune and Wright, 2011). Natural attenuation was the preferred option in the case of the DWH oil spill (Santner et al., 2011).
Aeration from tidal action, along with the addition of nitrogen in the form of ammonia, has been shown to significantly increase oil biodegradation in Louisiana salt marsh sediments (Jackson and Pardue, 1997; Shin and Pardue, 2001a,b; Shin et al., 2000, 2001). Anaerobic biodegradation of oil in marsh sediments can be enhanced in the presence of mixed electron acceptors (sulfate, nitrate). This enhancement occurs in Louisiana marsh sediments where various electron-accepting anaerobes that degrade petroleum hydrocarbons coexist (Boopathy et al., 2012). The recovery rate will depend on the extent of oiling, depth of oil penetration into the sediments, and types of plant species affected. There is already evidence that new shoots are appearing in heavily oiled areas, so the long-term effects may be limited (DeLaune and Wright, 2011). Research is under way evaluating the long-term impacts of the Macondo well oil on coastal ecosystem health and function in the GoM.10
Risk Analysis for Shoreline Cleanup Operations
The current understanding of impacts of residual near-shore oil from the spill on organisms including plants, invertebrates, fish, birds, mammals, and sea turtles has been addressed in the Operational Science Advisory Team report (OSAT, 2011a) and is summarized below. Direct exposure may occur through incidental ingestion when feeding on sand beaches, or through physical contact. Aquatic organisms can be indirectly exposed to toxins by diffusion of dissolved oil fractions across gills and cell membranes, or directly exposed by feeding on suspended oil residues.
_________________
OSAT considered the risk of acute toxicity to fish and invertebrates to be unlikely, but there is potential for chronic exposure to dibenzothiophenes and dibenzofuran from the wateraccommodated fraction. The dynamic nature of the surf zone and subtidal areas is expected to rapidly dilute petroleum concentrations to insignificant levels, and toxicologically significant petroleum concentrations would be expected to be localized in the surface microlayer adjacent to submerged oil mats or surface residue balls.
OSAT considered the risk to sea turtles from oil residues on top of the beach to be low, but it identified a potential difficulty for turtles attempting to nest by digging through subsurface tar mats (OSAT, 2011a). Beach cleanup and spill monitoring activities also impacted sea turtle nesting activities, but to what degree remains unknown. The toxicity of weathered oil to turtle eggs was assumed to be low (OSAT, 2011a). The oil is degrading and becoming less toxic, but media reports have claimed that more than 1,000 turtles, 70 marine mammals, and possibly hundreds more marine mammals (see Chapter 5), and 4,000 birds are thought to have died as a consequence of the DWH oil spill (Service, 2010). These numbers may be significantly underestimated. Neither short-term nor long-term risks nor damage to juvenile forms of these animals is known at this time. The release of the damage assessment data is expected to support more accurate quantification of these impacts.
OSAT (2011a) determined that the risks to birds are mostly applicable to their foraging along the beach and inadvertently ingesting oil. Risk appears low for the piping plover, but low to medium for the western sandpiper based on its foraging behavior. Risks to eggs of beachnesting birds are possible but unknown. Risks from cleanup activities are medium to high. Beach-dwelling mammals may be exposed to surface residue balls by incidental ingestion, or if they build burrows in the vicinity of supratidal buried oil (OSAT, 2011a). OSAT determined the risk of oil exposure for the federally endangered Alabama beach mouse (Peromyscus polionotus ammobates) to be low. That said, spill response actions, such as cleanup activities and increased human traffic on the beach, may have altered, reduced, or damaged the mouse’s habitat and therefore increased the threat to this species.
Results from the OSAT 2 NEBA analysis indicated that the environmental effects of the residual oil following cleanup are relatively minor, especially considering the pre-spill baseline level of oiling (OSAT, 2011b). Analyses of water and sediment samples along the northwestern Florida coastline have shown that swimming at or visiting the beaches poses no risk to human health from oil spill hydrocarbons (Florida Department of Environmental Protection, 2011). If cleanup were continued, then it would be expected to result in an increasingly greater extent of damage to habitats and associated resources. Consideration must also be given to the use of beaches for recreation in the spring and summer, which would conflict with cleanup operations (Florida Department of Environmental Protection, 2011).
NATURAL ATTENUATION
Natural attenuation is the “reduction in mass or concentration of a contaminant in the environment over time or distance from its source of release due to naturally occurring physical, chemical and biological processes, such as biodegradation, dispersion, dilution, adsorption,
and volatilization” (American Society for Testing and Materials, 2010). The natural attenuation of oil can be defined as the biotic and abiotic degradation and dispersion of oil that results in natural recovery of an oil-impacted environment. When oil enters the marine environment, abiotic weathering processes (evaporation to the air, dissolution in water, emulsification with water, dispersion, and photodegradation) (see Appendix G in SCAT, 2010) alter properties of the oil (density, viscosity, water content, surface and interfacial tensions), which ultimately define its fate.
In the natural environment, oil droplets do not persist intact. Some of the lighter compounds in the oil dissolve to become the water-accommodated fraction. A number of droplets will recombine or coalesce, rising back to the surface. Others undergo colonization by microbes and are biodegraded.
Oil droplets can attract and incorporate suspended particulate matter from the water column (Owens and Lee, 2003), surface water outflows, or stirred up sediment. These OMAs are an important factor controlling the fate and transport of oil spilled in the marine environment (Bassin and Ichiye, 1977; Boehm, 1987; Karickhoff, 1981; Payne et al., 1989). The parameters controlling the quantity, type, and size of OMA include mineral type and surface properties, quantity of mineral fines, oil viscosity and composition, and oil/mineral ratio (Stoffyn-Egli and Lee, 2002). Water turbulence (breaking waves, strong flood currents) greatly enhances OMA formation.
The concept of using enhanced OMA formation as an operational strategy for the cleanup of oil stranded on coastal beaches came about from laboratory studies of oiled sediments from the Exxon Valdez oil spill (Bragg and Yang, 1995; Bragg et al., 1994). These studies revealed that micron-sized mineral fines, seawater, and weathered oil interacted to form clay-oil flocculations (a type of OMA) that promoted the release of oil into the water column. This process was hypothesized to be responsible for the natural cleaning in sheltered shorelines of Prince William Sound after the oil spill. Enhanced OMA production by surf-washing operations has now been recognized as a strategy to remove oil stranded within beach sediments (Lee et al., 1997, 1999; Lunel et al., 1996, 1997; Owens, 1999; Owens and Lee, 2003; Swannell et al., 1999; see section, “Treatment of Shoreline Oiled by the DWH Oil Spill,” subsection “Surf Washing”).
Biodegradation
A large number of microorganisms are capable of biodegrading hydrocarbons (Atlas, 1984; NRC, 2003: ZoBell, 1973), and bacteria are the predominant hydrocarbon degraders in the marine environment (Atlas and Hazen, 2011). Biodegradation by microbial communities is the major process controlling the eventual removal of oil that enters the marine environment from natural seeps (Atlas, 1995; Atlas and Bartha, 1992; Leahy and Colwell, 1990). Although much slower, anaerobic (oxygen absent) biodegradation of oil should not be underestimated as a strategy, because it has been shown to be a major process in anoxic marine sediments, where sulfate, nitrate, manganese (IV), and iron (III) are the primary terminal electron-acceptors (Canfield et al., 2005; Finke et al., 2007; Lovley et al., 1997). Although normally present in small numbers in pristine environments, oil-degrading microbes can multiply rapidly upon the intro-
duction of oil (Atlas, 1995; Horel et al., 2012), as was the situation in the GoM after the DWH oil spill (Hazen et al., 2010).
The individual components in crude oil do not biodegrade at the same rate. Biodegradation rates are highest for the lightest components such as alkanes, followed by light aromatics (up to three rings), with high-molecular-weight aromatics (four rings and higher) and polar compounds exhibiting extremely low rates (Prince, 2010). Because of the chemical complexity of crude oil, its biodegradation may require the cooperation of a consortium of microbes or syntrophic interspecies (McInerney et al., 2008). Some microorganisms may also naturally produce extracellular biosurfactants that promote emulsification of hydrophobic hydrocarbons for subsequent transport into the hydrophilic intracellular space for biodegradation (Desai and Banat, 1997; Southam et al., 2001). Microbes are well adapted to their surroundings, including extremely low temperatures (Delille et al., 2009; McFarlin et al., 2011a), and Hazen et al. (2010) confirmed that oil in the low-temperature, deep-water environments of the GoM may be degraded by indigenous microorganisms at higher rates than previously thought.
Finding 4.9. Abiotic weathering processes and microbial degradation are major processes controlling the eventual removal of oil that enters marine and coastal environments. Thus, natural attenuation of some oil will occur without technological interventions.
Natural Attenuation Following the DWH Oil Spill
Regardless of the response technique employed, some oil will remain in the environment. For the DWH event, because of the volume of oil spilled and the natural rates of oil weathering, oil reached near-shore waters and coastal habitats, to which a range of removal technologies were applied. With the exception of buried oil that was reexposed after storms (e.g., Hurricane Isaac in 2012) and the associated wave surge, to which additional cleanup activities were applied, the fate of this residual oil was left to natural attenuation. The DWH oil spill highlighted the significance of natural oil degradation. Marine bacteria may have been primarily responsible for the observed loss of residual oil evident in the GoM (American Society of Microbiology, 2011; Walsh, 2010).
Unlike oil spills occurring at the sea surface, much of the petroleum hydrocarbons erupting from the Macondo wellhead experienced a prolonged, buoyancy-driven ascent through the 1,500-m water column (Hazen et al., 2010). Hazen et al. (2010) reported that the disappearance of residual oil in the GoM water column from the DWH oil spill was associated with microbial degradation processes. Genes for chemotaxis, motility, and aliphatic hydrocarbon degradation were found to be elevated in samples taken from inside the DWH oil plume compared to samples taken from outside of the plume, and cells of Oceanospirillales bacteria isolated from plume samples were found to possess gene sequences for the degradation of n-alkane and near-complete oxidation of cyclohexane (Mason et al., 2012). Temperature did not appear to be a major limiting factor because significant rates of oil degradation were observed within the subsurface plume of dispersed oil at a depth of 1,100- to 1,200-m and temperatures below 4°C (Hazen et al., 2010). Some estimates indicate that as much as 30 percent of the oil and gas
never reached the sea surface, but instead formed hydrocarbon-rich plumes within the cold waters present at 1,100–1,200 m depth (Hazen et al., 2010; Valentine et al., 2010).
An examination of petroleum hydrocarbons in oil mousse (emulsion of water and oil) collected between May and July 2010 from the seafloor and the sea surface, and as close as 6 km and as distant as 185 km from the wellhead (Liu et al., 2012), showed only light to moderate degradation of oil deposited in sediments near the wellhead (Liu et al., 2012). This contrasted considerably with the extensive weathering of oil observed in mousse samples in the marshes. Trace metal concentrations, particularly of aluminum, iron, and manganese, increased significantly from DWH crude oil, to surface mousse, and then to the marsh sediment, suggesting that clay minerals and dissolved metals were aggregated during oil weathering and mousse transportation. The fate of the oil in the subsurface plume remains an active field of research.
Finding 4.10. The fate and transformations of oil from the DWH oil spill were and will continue to be influenced by natural attenuation processes. These processes may be considered the best practical methods for oil removal in sensitive habitats, where the application of technology may cause more harm, or in logistically inaccessible habitats. Continued observations, monitoring, and research of these processes, especially in relation to technological countermeasures, are warranted.
Because biodegradation of all components within crude oil is not a rapid process, reliance on natural attenuation as the sole strategy might result in reduced provisioning of finfish and shellfish because of impacts to juveniles in spawning and nursery grounds, and a corresponding loss of commercial fishery, recreational fishing, and tourism income (discussed further in Chapter 5, “Fisheries” subsection). Thus far, however, the documented losses of ecosystem services after the DWH oil spill have been due to fisheries closures and public fears of consuming contaminated seafood, rather than reduced juvenile recruitment (McGill, 2011; Upton, 2011).
The longer-term impacts of selecting natural attenuation over more proactive spill responses to the ecosystem services provided by the deep-sea GoM, such as provisioning of bluefin tuna and royal red shrimp, recreational whale watching, and the provisioning of oil and gas, are uncertain (see Chapter 5). Regulating and cultural services provided by larger animals such as turtles and bottlenose dolphins (see Chapter 5) may also be adversely impacted. In addition, wetland losses due to the inundation and smothering of marsh plants have affected the ecosystem services provided by these habitats (additional discussion found in Chapter 5).
SUMMARY
A number of spill response technologies and techniques were applied during the DWH oil spill, including direct recovery from the wellhead, skimming, in situ burning, chemical dispersion, flushing (river diversion), and erection of physical barriers. In deeper water, enhanced dispersion via the use of chemical dispersants was one of the most effective response methods in terms of impacts on total oil volume. Skimming and burning removed about 7–10 percent of total oil. Once the oil made it to shore, cleanup efforts depended upon the type of shoreline.
To clean oil from beaches, sand washing and surf washing were performed. To clean oil from marshes, physical removal of the oil by manual labor, floating machinery, and manual cropping of oiled vegetation were employed.
Almost all countermeasures and response options have limitations and environmental effects that ultimately will impact ecosystem services. If the goal of response efforts is to protect the environment that sustains the ecosystem services, then we must realize that prioritization among the services involves complex decisions and the selection and application of response technologies involves tradeoffs. Most decisions regarding the application of spill response technologies after the DWH oil spill were based on NEBA,11 and they were made in consideration of the best available information at the time.
The immediate goal of the response technologies applied during the DWH oil spill was to prevent oil from reaching sensitive coastal habitats. Although none of the response tools was completely effective, the use of in situ burning, booms, skimmers, and particularly dispersants significantly mitigated the impacts of the spill along the coast. Continued remediation efforts will eventually result in diminishing returns, at which time natural attenuation of the remaining oil may be the best option. To this end, more science needs to be incorporated into the remediation process (both on- and offshore), not only to determine the most effective methods to apply under various conditions, but also to feed into the NEBA process of ascertaining the operational endpoint. Regarding the DWH oil spill, two factors raised important research questions: (1) the depth of the spill and (2) the large-scale application of emerging new technologies, such as subsurface injection of dispersants and oceanic in situ burning.
At the time of this writing, the consequences of the technologies applied to contain the oil and to prevent its reach into sensitive coastal and wetland habitats, as well as the response of the GoM ecosystem to the spill, have not been, and may never be, fully documented. Work to understand the fate and effects of the spilled oil is ongoing. Much work to determine the long-term effects remains. Understanding of the potential impacts of response technologies to ecosystem services and of the impacts of oil on multiple layers of ecosystems and their services is limited by the availability of baseline information and of important data collected during the NRDA process. Until this massive and unprecedented collection of data is complete and the results made available, any assessment of the impacts of the response technologies will not be fully informed. These issues will be discussed further in Chapter 6.
Finding 4-11. Any conclusive assessment of the impacts of response technologies on the GoM marine ecosystem and ecosystem services would be premature at this time, given the amount of key data and analyses that is being held confidentially in the ongoing NRDA process. Nonetheless, the committee believes that the technologies applied offshore mitigated the impacts of the oil spill on sensitive coastal habitats. A further factor for consideration is that only 3 years have passed since the spill started, and many substantial impacts may not become apparent for several more years.
_________________
11http://www.crrc.unh.edu/workshops/dwh_researchneeds_11/index.html.
































