4
AFRRI Programs, Research, and Resources
This chapter addresses the second element of the committee’s statement of task, assessing how Armed Forces Radiobiology Research Institute (AFRRI) programs are advancing research in radiobiological science related to human health risks from exposures to low-dose ionizing radiation. The chapter presents a brief history of AFRRI and its role within the Military Services. The Institute’s physical plant, staff, budget, current research, collaborations, and educational efforts are also described.
Material in this chapter is derived in part from a presentation to the committee by AFRRI Director L. Andrew Huff, Col. USAF, MC, SFS (Huff, 2013a) and on responses that AFRRI provided to questions posed by the committee (AFRRI, 2013a,b; Huff, 2013b, 2014). Citations for these and other sources are made as appropriate.
AFRRI HISTORY AND BACKGROUND
The Department of Defense (DoD) has been interested in the health effects of exposure to radiological agents since at least the initiation of the Manhattan Project. That interest became operational in 1958 when the U.S. Navy Bureau of Medicine and Surgery proposed that a bionuclear research facility be established to study such issues (DTRA, 2002; Tenforde, 2011). Public Law 86-500 (June 8, 1960) subsequently authorized construction of a laboratory and vivarium under the auspices of the Defense Atomic Support Agency (DASA) and, on December 2, 1960, the Military Services surgeons general and DASA approved a charter for AFRRI (DTRA, 2002;
Solyan, 2004). The Institute was formally established on May 12, 1961, when DoD Directive 5154.16 was issued.
Research at AFRRI began in January 1962, although the laboratory was not fully operational until September 1963 (DTRA, 2002; Solyan, 2004). At that time, the research facility included a Training, Research, Isotopes, General Atomics (TRIGA®) Mark F nuclear reactor, laboratory space, and an animal facility. The TRIGA reactor, a unit specifically designed for research, teaching, and commercial applications, allowed studies of radiation characteristics relevant to nuclear weapons that were not available at other DoD or Department of Energy facilities at that time. A high-dose cobalt-60 (60Co) facility, a 54-megaelectron volt (MeV) linear accelerator (LINAC), and a low-level 60Co irradiation facility were subsequently added (Solyan, 2004).
The Institute operated as a joint agency of the U.S. Army, Navy, and Air Force under the command and administrative control of the Office of the Secretary of Defense. In July 1964, responsibility for AFRRI was assigned to DASA, and the Chief of DASA was designated as the chair of its Board of Governors. AFRRI was identified as an operational field element of DASA while essentially functioning as an independent institute (AFRRI, 1968).
In the 1960s, the Institute’s research enterprise was partitioned into five departments—Experimental Pathology, Behavioral Sciences, Physical Sciences, Chemistry, and Radiation Biology—and focused on biological responses, with an emphasis on high doses of external radiation. Animal studies were an integral part of this work and were used to establish the effects of radiation on the central nervous and circulatory systems and on other tissues and organs (DTRA, 2002). AFRRI collaborated through memoranda of understanding and interagency agreements with universities, government agencies, and corporations (DTRA, 2002).
DASA was disestablished in 1971, and its responsibility for AFRRI was assumed by the newly formed Defense Nuclear Agency (DTRA, 2002). At around the same time, concerns were emerging about the possible relationship between exposure to radiation during military service and the occurrence of cancer. AFRRI was involved in the process that resulted in the establishment of the Nuclear Test Personnel Review program in January 1978. This program, which is still in operation, determines or estimates the radiation dose of veterans who participated in U.S. atmospheric nuclear tests or in the occupation forces of Hiroshima and Nagasaki, Japan, immediately after the atomic bombings, information that is used in compensation determinations for long-term radiation-related illnesses (DTRA, 2010). The experience and expertise developed by AFRRI in dealing with accidents, hazardous materials, and radiological cleanup issues were used in the international arena when AFRRI staff formed part of the International Chernobyl Site Restoration Assistance Team after the 1986 accident. They
also provided assistance to the environmental cleanup efforts at the closed Soviet test site at Semipalatinsk, Kazakhstan (DTRA, 2002).
The Defense Nuclear Agency transferred control of AFRRI to the Uniformed Services University of the Health Sciences (USUHS) in 1993 (DTRA, 2002). As the Cold War wound down, resources shifted to concentrate on peaceful activities, including social and nondefense programs. AFRRI’s funding and personnel levels diminished, and proposals were made to close the facility. However, military leaders indicated that there were no alternative sources for the information that the Institute developed, and these proposals were not acted on (Solyan, 2004).
U.S. interest in nuclear preparedness again increased in the late 1990s in response to India’s and Pakistan’s nuclear testing and the suspected development of nuclear weapons by Iraq and North Korea. AFRRI’s mission became more important to DoD in part because private companies lacked the incentive to develop radioprotectants and countermeasures for the military (Solyan, 2004). In response to this renewed interest, funding for AFRRI increased in 2000 (Assistant Secretary of Defense, 2004).
Increased awareness of terrorist threats in the wake of the attacks on U.S. sites on September 11, 2001, also stimulated support for the Institute (AFRRI, 2009). These events helped shape a change in AFRRI’s scope of work to include minimizing the effects of radiological dispersal devices (RDDs), terrorist access to radiation sources, and sabotage of nuclear reactors (Solyan, 2004). However, AFRRI’s readiness and capabilities were limited by their facilities and staffing level. At the time, the Institute faced the challenge of deteriorating mechanical and structural systems. Extra support granted in 2003–2004 allowed for infrastructural upgrades and the development of a radioprotective drug (5-androstenediol, HE 2100).
Today, AFRRI is DoD’s only medical research and development initiative dedicated to nuclear and radiological defense. It serves the military by performing medical research and development, education, and advisory and consultative functions for the purposes of understanding, preventing, preparing for, and responding to releases of ionizing radiation (AFRRI, 2011a). Box 4-1 delineates AFRRI’s mission, responsibilities, and assigned functions.
Management Structure
AFRRI functions as a joint entity of the Military Services under the authority, direction, and control of the president of USUHS, the Assistant Secretary of Defense for Health Affairs, and the Under Secretary of Defense for Personnel and Readiness (DoD, 2006). DoD Initiative 5105.33 (§4.2) specifies that it is to be led by a director who is a military officer with a doctoral degree in one of the life sciences. The director is nominated by the
BOX 4-1
AFRRI’s Mission, Responsibilities, and Functions as Delineated in DoD Instruction 5105.33, Issued on March 29, 2006
Mission (§3)
The mission of the AFRRI shall be to conduct research in the field of radiobiology and related matters essential to the operational and medical support of the Department of Defense and the Military Services. The AFRRI may provide services and perform cooperative research with other Federal and civilian agencies and institutions with the approval of the Assistant Secretary of Defense for Health Affairs.
Responsibilities and Functions (§5.1)
- Operate research facilities for the study of radiobiology and ionizing radiation bioeffects and for the development of medical countermeasures against ionizing radiation, and the results shall be disseminated.
- — The scope of this research shall reflect requirements identified by the DoD Components for support of military operational planning and employment (current and future), and shall put special emphasis on individual and organizational performances under nuclear and radiological combat conditions in realistic operational and force protection scenarios.
- — The AFRRI program shall consider present and projected threats, Service and joint operational concepts and weapons, and defense systems developments.
- Provide analysis, study, and consultation on the impact of the biological effects of ionizing radiation on the organizational efficiency of the Military Services and their members.
- Conduct cooperative research with the Military Medical Departments in those aspects of military operational and medical support considerations related to nuclear weapons effects and the radiobiological hazards of space operations.
- Conduct advanced training in the field of radiobiology and the biological effects of nuclear and radiological weapons to meet the internal requirements of the AFRRI, the Military Services, and other DoD Components and organizations.
- Participate in cooperative research and other enterprises, consistent with the AFRRI mission and applicable authorities, with other Federal agencies involved in homeland security and emergency medical preparedness.
- Perform such other functions as may be assigned by the Assistant Secretary of Defense for Health Affairs.
SOURCE: DoD, 2006.
surgeons general of the U.S. Army, Navy, and Air Force and appointed for a 4-year term. It is the director’s responsibility (§6.1) to act as liaison to the heads of DoD’s components and other governmental and nongovernmental agencies and to ensure that the DoD components are informed of AFRRI’s activities. No scientific duties are assigned to the post.
Scientific leadership is exercised by a scientific director, who is tasked with the administration and supervision of the Institute’s research-oriented departments, overall scientific and technical planning of the research program, and service as the scientific liaison with the outside world (AFRRI, 1968). However, this position has not been filled since 2012. There is, at present, a scientific advisor, who counsels the director and acts as a liaison with outside agencies but is not a part of the chain of command (Huff, 2013b). Box 4-2 lists AFRRI’s directors and scientific directors since its inception.
AFRRI is currently made up of Radiation Sciences, Scientific Research, Military Medical Operations, Veterinary Sciences, Facilities Management, Good Laboratory Practice/Test Facility, and Administration Support departments, which are led by department heads or managers (see Figure 4-1). Four primary research areas are identified on the Institute’s website: biodosimetry, combined injury (radiation with other insults), internal contamination and metal toxicity, and countermeasure development (AFRRI, 2014g).
Capability and Infrastructure
AFRRI’s dedicated radiation sources and specialized facilities are summarized in Table 4-1 and are described in the following sections.
TRIGA Reactor
AFRRI’s TRIGA reactor is 1 of 66 worldwide (General Atomics, 2014). These research reactors are used in university and government laboratories and medical centers for applications that include production of radioisotopes for medicine and treating tumors, nondestructive testing, basic science research, education, and training. They operate at thermal power levels of <0.1–16 megawatts (MW) and may be pulsed up to 22,000 MW.
The reactor is licensed by the U.S. Nuclear Regulatory Commission (License R-84). As of 2005, it was 1 of 18 TRIGA reactors in the United States and the only one dedicated to applied medical radiobiology research (Dix, 2005). It is a medium-sized unit that generates neutrons and gamma rays for radiation experiments. The reactor can produce a controlled, self-sustaining fission chain reaction in the reactor core which, in addition to the fuel elements and control rods (containing boron carbide), includes a neutron start-up source (americium/beryllium). It is suspended under 16 feet (~4.9 m) of water within a pool (an effective radiation shield) in a
BOX 4-2
AFRRI Leadership
AFRRI Directors
1961–1966: James T. Brennan, COL, MC, USA
1966–1967: Joseph S. Burkle, CAPT, MC, USN
1967–1971: Hugh B. Mitchell, Col, MC, USAF
1971–1975: Myron I. Varon, CAPT, MC, USN
1975–1977: LaWayne R. Stromberg, Col, MC, USAF
1977–1979: Darrell W. McIndoe, Col, MC, USAF
1979–1982: Paul E. Tyler, CAPT, MC, USN
1982–1985: Bobby R. Adcock, COL, MSC, USA
1985–1986: James J. Conklin, Col, MC, USAF
1986–1987: Richard I. Walker, CAPT, MSC, USN
1987–1991: George W. Irving III, Col, BSC, USAF
1991–1995: Robert L. Bumgarner, CAPT, MC, USN
1995–1997: Eric E. Kearsley, CAPT, MSC, USN
1997–2003: Robert R. Eng, COL, MS, USA
2003–2006: David G. Jarrett, COL, MC, USA
2006–2010: Patricia K. Lillis-Hearne, COL, MC, USA
2010–2012: Mark A. Melanson, COL, MSC, USA
2012–present: L. Andrew Huff, Col, MC, SFS, USAF
AFRRI Scientific Directors
1966–1971: Harold O. Wyckoff, Ph.D.
1982–1987: Lawrence S. Myers, Ph.D.
1987–1989: Richard I. Walker, Ph.D., CAPT, MSC, USN
1989–1998: E. John Ainsworth, Ph.D.
2002–2008: Terry C. Pellmar, Ph.D.
2008–2012: Christopher R. Lissner, Ph.D.
NOTE: This position was not filled in some periods.
SOURCES: AFRRI, 2010, 2013b; Solyan, 2004.
carriage assembly that allows movement of the core between two exposure rooms for experimental work with large-animal or other studies (Dix, 2005). The advantages of such a movable reactor core are that the quantity and character of the radiation that reaches the exposure facilities can be controlled, and more than one exposure facility can be used during reactor operations.
The reactor can operate in steady-state as well as pulse mode. The maximum allowed steady-state power level is 1.0 MW. Its pulse mode can produce a short peak (from a prompt critical excursion) of up to 2,500 MW occurring in about 0.1 second. The neutrons and gamma rays produced
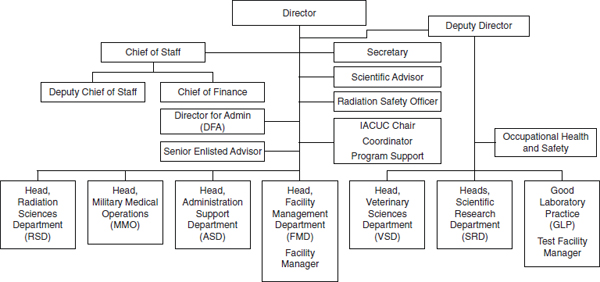
FIGURE 4-1 AFRRI chain of command and organizational structure.
NOTE: IACUC = Institutional Animal Care and Use Committee.
SOURCES: Derived from AFRRI, 2013c, and Huff, 2013b.
TABLE 4-1 AFRRI Radiation Sources
|
|
|
| Source | Activity or Energy |
|
|
|
| TRIGA® Mark F reactor | 1-megawatt (MW) steady-state or 2,500-MW pulse, mixed beam—neutron, photon |
| 60Co facility (panoramic irradiator) | 450,000-Curie (Ci) 60Co source |
| Chronic irradiation facility | 100-Ci 60Co source |
| Philips industrial X-ray machine* | 40-320 peak kilovoltage (kVp) |
| Cesium (Cs) calibration facility | 100-Ci 137Cs source |
| Elekta Infinity™ linear accelerator (LINAC) with Synergy® image-guided workflow and Philips Brilliance CT Big Bore | LINAC operations up to 15 megaelectron volt (MeV) System was acquired in August 2012 but was not operational as of January 2014 |
|
|
|
* This source was replaced by an Xstrahl Small Animal Radiation Research Platform in 2014. SOURCES: AFRRI, 2011a; Huff, 2014; Kang et al., 2011.
in the reactor pass (as a unique mixed field) to exposure facilities, where biological systems are irradiated for studies. The facilities include two large exposure rooms and a core experiment tube, each with a distinctive radiation field and setup characteristics that allow for studies of a variety of conditions. The gamma:neutron ratio can be varied from 1:20 to 20:1 through the use of shields and absorbers placed in the exposure rooms (AFRRI, 1993). Special setups (in-pool portable beam tubes, a pneumatic transfer system, and in-core grid-location tubes) and custom radiation beams are also available. Exposure rates can be varied from about 0.1 rad/min (0.06 gray [Gy]/hr) to 1,000 rad/min/pulse (600 Gy/hr/pulse). Although primarily used for biological studies, the unit may also be used for transient radiation–electronic effects (TREE) studies and the production of isotopes.
Cobalt-60 Facility
The 60Co facility at AFRRI first opened in 1969. The facility is located below ground in the AFRRI complex, with shielding provided by massive reinforced concrete and earth fill. Its panoramic irradiator is a wet-source storage unit consisting of a 450,000 Ci (at installation) 60Co source, water trench, source and storage racks, elevator mechanism, and associated equipment. The exposure room is 35 ft × 35 ft and 25 ft, 8 in. high (10.7 m × 10.7 m × 7.6 m = 870 m3) (AFRRI, 1993). The irradiator produces monoenergetic gamma rays at variable dose rates with flexible configurations in both unilateral and bilateral irradiation modes and may be used for acute and chronic studies of materials, biologic specimens, and small and large animals (Carter and Verrelli, 1973; Naquin et al., 2001). It
has been employed in a variety of applications, including investigations of the effects of ionizing radiation exposure on cells (McKinney et al., 1998), prognostic indicators of survival in a variety of mammals (Moroni, et al. 2011), and the efficacy of radioprotective agents (Landauer et al., 2001; Singh et al., 2010).
In 2013, AFRRI contracted to replace the facility’s existing, decaying sources with new 60Co sources (FedBizOps.gov, 2013).
Chronic-Irradiation Facility
AFRRI has a second 60Co radiation source that provides low-dose rate gamma-photon radiation to simulate chronic exposure to low doses and is used to study early and late effects in biological samples. This 100-Ci chronic-irradiation facility (Dix, 2005) is sometimes called the low-level irradiation facility (Solyan, 2004).
Earlier AFRRI reports (AFRRI, 1993; Zeman and Dooley, 1984) describe a 4,200-Ci therapeutic irradiator (AECL Theratron-80) capable of providing from 1 to several hundred rad/hr (0.01 to several Gy/hr) over limited field sizes, and a uniform field. It was primarily used to conduct cellular studies. However, this source was decommissioned in the 1990s.
Linear Accelerator
AFRRI’s first LINAC was designed and assembled between 1965 and 1968; it provided a powerful, flexible source of high-energy electrons, high-energy bremsstrahlung (X-rays), and neutrons (AFRRI, 1993). It was used for a broad range of applications, including radiobiology and radiochemistry studies (AFRRI, 1993). Various machine configurations were used to provide electron energies continuously variable from 10 to 54 MeV (Dix, 2005).
This device was retired and, in August 2012, AFRRI acquired a new LINAC and computed tomography (CT) unit: an Elekta Infinity LINAC capable of operations up to 15 MV and a Philips Brilliance CT Big Bore (Huff, 2014). These devices will be used for research purposes only. Neither was operational at the time the committee completed its work in late 2013.
Other Radiation Sources
AFRRI also has a Philips industrial X-ray machine that is a water-cooled device with peak kilovoltage (kVp) that ranges from 40 to 320 kVp (AFRRI, 2011a).1 This machine is used mainly for cellular work and, depending
_________________
1 After the report was completed, the committee learned that this source has been replaced by an Xstrahl Small Animal Radiation Research Platform.
on the field size, the output can be varied from a few to 7,000 rad/min (4,200 Gy/hr). The machine also serves as a backup for the 60Co facility (AFRRI, 1993).
The Institute also maintains a cesium (Cs) calibration facility that consists of a 100-Ci 137Cs source and associated equipment.
Usage of Radiation Facilities
The committee asked AFRRI to delineate how often its radiation facilities are being used by AFRRI investigators and by any outside investigators or collaborators.2 AFRRI’s response, dated October 15, 2013, is summarized in Table 4-2 (AFRRI, 2013b).
Animal Facility
An animal-research facility is an important resource for understanding basic radiobiology and for developing medical countermeasures against radiation injuries. AFRRI’s facility is organized within its Veterinary Science Division. The 28,565 ft2 (~2,650 m2) space is designed to support radiation and surgical studies and includes environmental controls and monitoring, histopathology, microbiology, and clinical pathology laboratories. It comprises
- Two large-animal surgery suites,
- One radiology suite,
- One large-animal treatment room,
- One large-animal necropsy room,
- Ultrasound and electrocautery equipment for diagnostic and surgical purposes, and
- Two rodent-procedure rooms.
AFRRI is one of the few DoD laboratories capable of housing a variety of animals. In response to an inquiry from the committee, AFRRI indicated that the facility commonly maintains 4,000–5,000 mice and rats, 8–20 minipigs, and 60–80 nonhuman primates (rhesus macaques) (AFRRI, 2013b).
In late 2013, the Veterinary Sciences Department comprised 26 staff members: 5 veterinarians (Department Head, Deputy Head, Contract Clinical Veterinarian, Veterinarian, and Veterinary Pathologist), 6 veterinary technicians (5 military and 1 civilian), 11 government animal-husbandry
_________________
2 Outside investigators may use the facilities for studies performed in collaboration with an AFRRI project or other projects funded by a government agency.
TABLE 4-2 Usage and Availability of AFRRI Radiation Sources, January 2012–October 2013
|
|
||
| Device | Usage | Availability* |
|
|
||
| TRIGA reactor Accessible 293 days total |
51 days training; 17% of available time 10 days in-house use; 3% of available time | 79% unused |
| Cobalt-60 facility Accessible 2,300 hr total |
1,058 hr in-house use; 46% of available time 82 hr outside use; 4% of available time | 50% unused |
| Chronic-irradiation (low-level) facility |
“Several days” | Nearly 100% unused |
|
|
||
* These numbers include time when sources were unavailable due to equipment failure or maintenance.
SOURCE: AFRRI, 2013b.
personnel, 3 pathology lab staff (2 military and 1 civilian), and 1 administrative person (military, on loan) (AFRRI, 2013b).
The Veterinary Sciences Department has maintained an American Association for Accreditation of Laboratory Animal Care–accredited animal care and use program since 1984. All research protocols require review and approval from the organization’s Institutional Animal Care and Use Committee. The program supports not only AFRRI research but also USUHS and Walter Reed National Military Medical Center studies.
CURRENT RESEARCH PRIORITIES AND PORTFOLIO
AFRRI’s research agenda has evolved over time with U.S. defense needs, changes in funding, and scientific advancements. In the 1960s, the focus was on the effects of high doses of external radiation and the development of causality criteria for radiation illnesses. However, as military and defense priorities changed, it has expanded to include nuclear-weapons effects, trauma, toxicology, nonionizing-radiation effects, cancer markers, and drug toxicity, as well as specific needs for solutions to casualty problems that may be associated with multiple insults from exposure to radiation and other battlefield hazards such as biological and chemical agents as well as wounds, infection, and diseases (Solyan, 2004).
AFRRI’s current efforts concentrate on minimizing the health effects of exposure to high-dose ionizing radiation in combat and military environments through prevention of hazards, assessment, and medical treatments of injuries relating to radiation both alone and with other chemical or biological hazardous agents. These efforts are summarized in the following sections.
Radiobiology Research
In accordance with its charter, AFRRI conducts research in the field of radiobiology and related matters essential to the operational and medical support of DoD and the Military Services. This includes evaluation of threats (the threat spectrum, defining the source and risk, operating in a contaminated environment, consequence management, modeling and decision support, and the like); triage (methods, tools, techniques, and biodosimetric models); and treatment (both prophylactic and mitigative, as well as evaluating off-label and investigative drug use in DoD). AFRRI research has concentrated on military concerns such as preparation, consequence management, and mitigation, and it extends from acute event–response readiness to actual radiation exposures of DoD personnel. Such work is also relevant to civilian exposures resulting from accidents, terrorist activities, or war.
The AFRRI research program is aimed directly at current research gaps in medical preparedness for responding to such events. For example, current countermeasures (amifostine, the only U.S. Food and Drug Administration [FDA]-approved radioprotectant) may have effects that make them inappropriate for military use. Available biodosimetric tools for triage are limited in speed and physiological predictive power. There is also a need for mitigation and therapeutic agents approved for radiation-induced hematopoietic or gastrointestinal injury. Therefore, the AFRRI research and development goals concentrate on the following: pursuing new drugs that will prevent the life-threatening and health-degrading effects of ionizing radiation; developing methods for rapidly assessing radiation exposure to ensure appropriate medical treatment; investigating the effects of radiation injury combined with other challenges such as trauma, disease, and chemical exposures; and contributing to the radiobiological knowledge base.
Biological Dosimetry
AFRRI has been engaged in developing rapid, high-precision analytical methods that can be used to assess radiation-exposure doses from clinical samples to aid in the triage and medical management of radiological casualties. The specific objectives of this research include automating field-deployable biological dosimetry capabilities for rapid dose assessment on the battlefield, establishing reference biological dosimetry for definitive analysis of biological samples from military theater operations, and identifying and validating early-phase radiation-specific biomarkers of late-radiation effects.
AFRRI’s research in the biological dosimetry arena has centered on the development of integrated biodosimetry and diagnostic systems (see Figure 4-2). Triage, clinical, and definitive radiation biodosimetry require multiple bioassays and specific analytic technologies designed for chemical,
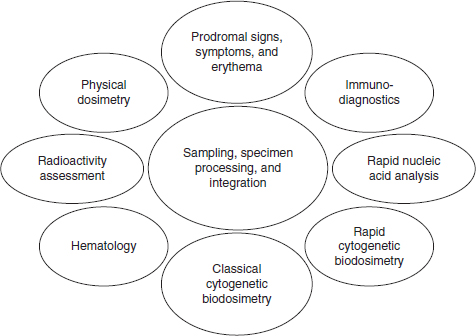
FIGURE 4-2 The components of an integrated biodosimetry and diagnostic system.
SOURCE: AFRRI, 2013d.
biological, radiological, nuclear, and explosive (CBRNE) exposures because no single assay or technique is sufficient. The aims are to create protocols and analytical systems for high-throughput applications, to identify bio-indicator assays for rapid assessment over a broad dose range, to refine hematological and molecular protocols and analytical systems, to validate the systems using in vivo model studies, and to develop software for integrated biodosimetry data management.
The appropriate use of medical resources for personnel exposed to ionizing radiation depends on timely, accurate dose information. To assist in meeting this specific and often complex need, AFRRI developed the Biodosimetry Assessment Tool (BAT) (AFRRI, 2013e), a computer-based software system for use by health care providers responding to a radiation incident. The BAT assists providers in identifying individuals who have significant radiation exposures and then in making appropriate treatment decisions using AFRRI-developed, radiation dose–predicting algorithms (single lymphocyte count, lymphocyte-depletion rate, and time from exposure to time of onset of emesis). The BAT algorithms are also available at the Radiation Event Medical Management website (www.remm.nlm.gov/ars_wbd.htm), an integrated educational and response tool supported
by the U.S. Department of Health and Human Services (HHS) and the National Library of Medicine.
The AFRRI biodosimetry research program also provides assessment instrumentation, discussed later in this chapter in the section called Publications, Guidance, and Tools.
AFRRI is working toward integrating the various tools with a deployable laboratory system for use in the field, and it holds seminars concentrating on ongoing and future advancements in the area of biodosimetry.
Internal Contamination and Metal Toxicity
Research on the chemical and radiological toxicity of radiologic materials is applicable to a variety of battlefield scenarios and possible terrorism events. AFRRI scientists have been at the forefront of research into the health effects of embedded fragments of depleted uranium (DU), a component of some munitions and armor. A specific aim of the AFRRI internal contamination and metal toxicity program is to determine whether the short- and long-term radiological and toxicological risks of embedded metals warrant changes in the current combat and postcombat fragment-removal policies for military personnel and (in the case of internalized radiological hazards) to investigate treatment strategies to enhance elimination of these metals from the body. To that end, AFRRI is developing models for assessing the health effects of embedded metal fragments, for investigating new decorporation protocols for the elimination of internalized radionuclides, and for studying the long-term health effects resulting from exposure to DU as well as for identifying biomarkers that can distinguish this exposure from other toxic insults. Such studies are also relevant to the isotopes that could be used in RDDs.
Research conducted at AFRRI was instrumental in the formulation of the U.S. Army policy dealing with injuries from DU. As munitions developers shift away from the use of DU in armor-piercing shells, AFRRI has also examined replacement metals such as tungsten alloys. Much of the work on the development of an in vivo model for investigating the health effects of embedded fragments (and protocols for dealing with such wounds), the toxicological properties and health effects of embedded DU, the refinement of analytical procedures for determining DU levels in biological samples, and the mechanism of damage resulting from DU exposures was first conducted at AFRRI (AFRRI, 2013f).
Radiation Countermeasures
AFRRI maintains a program to study, evaluate, and develop pharmacological countermeasures to radiation injury that can be used by military per-
sonnel and emergency responders. This research program aims to develop a better understanding of the biology of radiation injury and the radiation-countermeasure drugs, to identify and assess novel drug candidates, and to collaborate with others to develop and obtain approval for such drugs for use in the field and the clinic (AFRRI, 2014b).
Possible countermeasures to ionizing radiation can be broadly categorized into three groups: drugs that prevent the initial radiation injury (for example, free-radical scavengers; antioxidant, anti-inflammatory, and anti-fibrotic agents; and hypoxia-, enzymatic detoxification-, or oncogene-targeting agents), drugs that repair the molecular damage caused by radiation (hydrogen transfer, enzymatic repair), and drugs that stimulate proliferation of surviving stem and progenitor cells (immunomodulators, growth factors, and cytokines). Nontoxic or extremely low-toxicity countermeasures to ionizing radiation are urgently needed by both military personnel and emergency responders, and much of the current focus is on drug candidates that are easy to administer and therefore suitable for use outside of specific clinics without close physician supervision.
The AFRRI radiation-countermeasures program has been involved in the development of several drugs for acute radiation syndrome (ARS) that now have FDA Investigational New Drug status, allowing their testing for safety in humans (AFRRI, 2014b). Several of the countermeasures were conceived by or initially developed at AFRRI or were developed in collaboration with AFRRI: granulocyte–colony stimulating factor (G-CSF), Neupogen®, 5-androstenediol, genistein, Ex-Rad®, and CBLB502. Other countermeasure candidates are ready for advanced development.
Besides its development work, AFRRI has an ongoing in vivo efficacy screening and mechanistic research program (ranging from standard models to a newly developed minipig large-animal model). AFRRI is also frequently approached by outside organizations requesting collaboration or consultation on their countermeasure candidates. The available radiation facilities at AFRRI—especially the mixed neutron–gamma fields that mimic those produced by nuclear detonations—has afforded AFRRI’s radiation-countermeasures scientists the opportunity to assess agents for efficacy more broadly across a spectrum of possible needs and uses. In addition to evaluating the toxicity of amifostine, AFRRI’s work on cytokines as a radiation countermeasure resulted in the inclusion of Neupogen® in the Strategic National Stockpile3 (CDC, 2012).
_________________
3 The Centers for Disease Control and Prevention’s Strategic National Stockpile comprises caches of medicine and medical supplies intended for use in case of a public health emergency (for example, a terrorist attack, flu outbreak, or earthquake) that is severe enough to deplete local supplies.
Radiation Injury Combined with Other Trauma
Recognizing that combined injuries can complicate effective treatment and mitigation activities, AFRRI established a specific research program area in 2007 to address radiation injury combined with other trauma (AFRRI, 2013g). The program’s aims include the development of medical treatments for irradiated personnel who also have traumatic wounds, burns, hemorrhage, or infection. AFRRI researchers seek to develop a comprehensive understanding of the biology of radiation injury combined with traumatic injury and to establish an understanding of countermeasure drugs (including novel drug candidates for prophylaxis, mitigation, or therapy). Treatment strategies under investigation include the use of biological-response modifiers, new antimicrobial agents, probiotics, and stem cells. Another aim is to collaborate proactively with other research institutions, pharmaceutical firms, and government agencies to further develop and gain approval of promising drugs for use in the field or the clinic. Little research is being conducted in this area, so AFRRI’s work fills an otherwise unmet need.
Other Research Areas
AFRRI formerly conducted research on the effects of radiation on behavior (DTRA, 2002). Its Behavioral Sciences Department was responsible for investigating the psychological effects of radiation by using animal models. Studies included the effect of ionizing radiation on stimulation-response rates in rats (Mele et al., 1990) and the behavioral effects of a medication used as a radiation countermeasure (Landauer et al., 1997). AFRRI published results on behavioral effects as early as 1969 (Thorp and Germas, 1969) and released a database of its findings in primates in 1981. That department was disbanded, however, and the last publications from it are from the late 1990s.
The committee is not aware of any human epidemiological research conducted by AFRRI. However, results of studies conducted by AFRRI researchers of Helicobacter pylori gastric infection in rhesus monkeys were asserted to have implications for epidemiological investigations in humans (Dubois et al., 1995).
EDUCATION, TRAINING, AND EMERGENCY-RESPONSE EFFORTS
AFRRI has long maintained a mission related to education and emergency response. Some major components of this responsibility—the Institute’s Medical Effects of Ionizing Radiation (MEIR) course, the Medical Radiobiology Advisory Team (MRAT), and production and dissemi-
nation of publications, guidance, and tools for managing nuclear and radiation-exposure incidents—are described in the following sections.
Medical Effects of Ionizing Radiation Course
AFRRI originally developed a course on the pathophysiology, diagnosis, and treatment of radiation effects in the 1970s. That course, then called the Medical Effects of Nuclear Weapons, was designed to enhance the capability of a military medical community that was inexperienced in radiation matters. Its content was expanded in the 1990s to include the medical response to radiation accidents, and the course took its current name.
MEIR is now offered regularly at military bases in the United States and abroad as a 3-day graduate-level course (AFRRI, 2014d). The course is presented by AFRRI subject-matter experts using up-to-date information (AFRRI, 2013h). It is available without cost to military personnel and DoD civilian employees; members of the general public may attend for a fee. A 6-hour online refresher course is also offered. Some 800–1,000 people take the course each year (Parde, 2012).
The course concentrates on the biomedical consequences of radiation exposure, the reduction of effects, and the medical management of casualties. It covers health-physics aspects, biological effects of radiation, and both the physical and psychological health effects.
Medical Radiobiology Advisory Team
DoD Directive 3150.08 specifies that AFRRI is “the DoD lead on medical and radiobiological matters for radiological-incident response and training” and that as part of this responsibility it shall maintain, in coordination with DTRA, a medical radiobiology advisory team (MRAT) “to support the medical and radiobiological aspects of the response to a U.S. nuclear-weapon incident and other nuclear or radiological incidents” (DOD, 2010a, p. 8). Depending on need, the team may be composed of radiation-medicine physicians, health physicists, radiobiologists, biodosimetrists, and other subject specialists. The MRAT may deploy either independently or as part of a larger team to provide guidance and advice on potential health hazards, decontamination, medical response, exposure modeling, population monitoring, risk assessment, and the like, and it participates in military training exercises that have a nuclear or radiological component (AFRRI, 2013i). This cadre of experts augments the on-call quick response of DTRA’s Consequence Management Advisory Teams (CMATs) for dealing with worst-case nuclear- and radiological-emergency scenarios worldwide. Its personnel also support other DoD activities, including the Nuclear Weapon
Incident Response Group out of the Office of the Assistant Secretary of Defense for Nuclear, Chemical, and Biological Defense Programs and the Joint Chiefs of Staff Joint Nuclear Accident and Incident Response Team (DoD Manual 3150.08 [DOD, 2013]).
The MRAT’s emergency-response mission also extends to the domestic civilian sector under the Nuclear/Radiological Incident Annex (NRIA) of the National Response Framework (NRF) (FEMA, 2013a). The NRF defines the roles and responsibilities of different federal agencies during an emergency. It may be invoked only when that emergency overwhelms the state, local, and regional capabilities.4 Its NRIA “describes the policies, situations, concepts of operations, and responsibilities of the federal departments and agencies governing the immediate response and short-term recovery activities for incidents involving release of radioactive materials to address the consequences of the event” (FEMA, 2013b).
Publications, Guidance, and Tools
In accordance with its charter and mission, AFRRI produces a number of products intended to educate medical and emergency-response professionals and facilitate the management of nuclear- and radiological-exposure incidents. These are compiled from the outcomes of more than five decades of research and development and experience.
One handbook, Medical Management of Radiological Casualties, developed by the AFRRI Military Medical Operations staff, is designed to be used by responders who may have minimal knowledge of the effects of ionizing radiation on the human body. It addresses such topics as ARS, biodosimetry, skin injury, internal contamination, psychological support, delayed effects, and decontamination. The first edition was released in 1999. At the time this report was completed, it was in its fourth edition, published in July 2013; it is available in print, PDF, and online5 formats (AFRRI, 2014e).
In 2008, AFRRI first produced its Pocket Guide—Emergency Radiation Medicine Response. As the name suggests, it is intended to be a quick reference used to help assess and control an emergency situation. The guide contains a radiation patient-treatment decision tree and summarizes steps to be taken in diagnosis, treatment, decontamination, and reporting. Its most current version is dated April 2011, and it is available in both print and PDF format (AFRRI, 2011b).
As part of the DoD series, Textbooks of Military Medicine, AFRRI—in
_________________
4 Robert T. Stafford Disaster Relief and Emergency Assistance Act, Public Law 100-707, signed into law November 23, 1988; amending the Disaster Relief Act of 1974, Public Law 93-288.
5 See http://www.usuhs.edu/afrri/outreach/4thEdition.html (accessed May 15, 2014).
conjunction with the Borden Institute, U.S. Army, Office of the Surgeon General—produced the book Medical Consequences of Radiological and Nuclear Warfare. It was initially published in 1999 and was updated in 2012 (AMEDD, 2013). This volume addresses nuclear events and their consequences for the medical community, including ARS; triage and treatment of radiation and combined-injury mass casualties; treatment of internal radionuclide contamination; behavioral and neurophysiological consequences of radiation exposure; cytogenic dosimetry; and other related topics. One chapter is devoted to late and low-level effects of ionizing radiation.
Further, AFRRI has published several specific policy guidelines for military-specific activities. Among these are a guideline on the use of insoluble Prussian blue (Radiogardase®) for treatment of internal radiocesium contamination, and an associated worksheet has a decision tree to guide response to an RDD event that involves the dispersal of radioactive cesium or thallium (AFRRI, 2014e). Both documents are in print and PDF format and were last updated in 2005. AFRRI’s guidance on the management of embedded DU and on the use of potassium iodide (KI) for the protection of U.S. service personnel and family members6 has resulted in specific Department of the Army and DoD Health Affairs policies.
AFRRI has also produced a number of publications as part of its scientific program and guidance-and-outreach mission. These include books and book chapters, contract reports, scientific journal articles, reports and report chapters, scientific abstracts, and technical documents. The AFRRI website maintains lists of these with links to PDFs of some publications. In response to a question posed by the committee, AFRRI indicated that the lists are in various stages of completeness, with the most recently released materials being the best documented (AFRRI 2013a). Table 4-3 enumerates these publications.
A number of forms and software tools useful in the response to a nuclear or radiological emergency have also been developed and are disseminated by AFRRI (2013e). These include the following:
- AFRRI Adult/Pediatric Field Medical Record (AFRRI Form 330), a one-page form for gathering emergency medical information in the field applicable to both adult and pediatric cases.
- AFRRI Biodosimetry Worksheet (AFRRI Form 331), which provides a place for recording the facts about a case of radiation exposure, including the source and type of radiation, the extent of exposure, and the nature of the resulting injuries.
_________________
6 Potassium iodide protects the thyroid gland against internal uptake of radioiodines that could be released in certain nuclear accidents or terrorist events (NRC, 2004).
TABLE 4-3 Publications Produced by AFRRI
|
|
||
| Publication Type | Years Covered | Number of Publications* |
|
|
||
| Books/book chapters | 1976–2013 | 285 |
| Contract reports | 1965–2012 | 44 |
| Journal articles | 1961–2014 | 1,585 |
| Reports and report chapters | 1993–2012 | 105 |
| AFRRI scientific reports | 1965–1979 | 264 |
| AFRRI special publications | 1965–2009 | 25 |
| AFRRI technical reports | 1965–1997 | 130 |
|
|
||
* As of April 2014, as compiled by AFRRI.
SOURCE: AFRRI, 2014f.
- AFRRI Radiocesium Worksheet (AFRRI Form 335), a flowchart, which details steps for screening patients suspected of having been or confirmed to have been exposed to radiocesium during an RDD event.
- Biodosimetry Assessment Tool (BAT), a computer-based software diagnostic tool for use by healthcare providers shortly after a radiation incident to help them identify individuals with significant radiation exposure and make appropriate treatment decisions.
- WinFRAT (First-responders Radiological Assessment Triage), a software tool that enables first responders to triage suspected radiation casualties on the basis of the initial, or prodromal, features listed in the Emergency Radiation Medicine Response pocket guide.
At present, the forms exist as PDFs and the software tools as Windows operating system programs. All are applicable to low-dose exposure circumstances.
AFRRI formerly provided information on its operations, research initiatives, publications, and other accomplishments in its annual reports. These contained detailed descriptions of work in each of the Institute’s major research areas and of the Institute’s outreach, research support and extramural activities. Annual reports were discontinued after FY 1992, but some of the same material—including a comprehensive listing of publications—is posted on the Institute’s website. However, neither that material nor information on budgets, research sponsors, staff biographies, or the like is currently available in a consolidated form.
Collaborative Educational Efforts
The USUHS Department of Radiation Biology within the School of Molecular and Cell Biology has several AFRRI principal investigators (PIs) serving as primary or adjunct faculty members. The department offers graduate training and research opportunities that are complementary to AFRRI’s work (USUHS, 2012). According to information gathered in July 2013, one graduate course in radiation biology is offered each spring. For more than 10 years, the course has been taught by an AFRRI PI, with four to eight students in attendance. In addition, staff members participate in teaching USUHS classes in biology, physiology, toxicology, and military response to nuclear and radiation threats and also serve on doctoral-dissertation committees. Plans for a graduate program in radiation biology as a part of the USUHS Molecular and Cell Biology Department have been developed but not implemented (AFRRI, 2013a).
AFRRI PIs also participate in educational activities outside of the Institute. They have served as experts and lecturers for the International Atomic Energy Agency (IAEA) cytogenic biodosimetry courses, the Oak Ridge Institute for Science and Education (ORISE) Radiation Emergency Assistance Center/Training Site (REAC/TS) courses, and the National Aeronautics and Space Administration (NASA) Space Radiation Summer School (AFRRI, 2013a).
Further, the Institute conducts periodic and special educational symposia, including seminars related to their research programs and overall mission, strategic plans, and objectives.
AFRRI Internships and Fellowships
In addition to the formal educational activities of its staff members, AFRRI sponsors internships and fellowships. Generally, the Institute hosts one to three postdoctoral scholars at a time through the Research Associateship Program of the National Research Council (NAS, 2014); eight participants in this program subsequently became AFRRI staff members (AFRRI, 2013a; Gamble, 2013). AFRRI has also hosted IAEA fellows studying biodosimetry. For more than a decade, AFRRI has worked with the American Society for Engineering Education’s Scientist and Engineering Apprenticeship Program to encourage high school students to pursue careers in laboratory research and science and technology fields (SEAP, 2014). In 2013, the Office of Naval Research provided funding for eight summer interns within AFRRI (AFRRI, 2013a). Further, some military institutions send students to be educated at AFRRI. The Institute hosts one to two second- or third-year midshipmen from the U.S. Naval Academy for 3–4 weeks each summer and, in 2013, it trained a West Point cadet for
2 weeks. Finally, AFRRI reports that starting in 2014, it plans to host two or three undergraduate summer interns though the Naval Research Enterprise Internship Program (AFRRI, 2013a; NREIP, 2014). Overall, these efforts typically account for fewer than 10 students per year.
BUDGET
AFRRI functioned on a budget of $2–$3M/year in the 1960s (DTRA, 2002). After the Cold War years, funding decreased as national spending priorities concentrated on peacetime activities, including social and nondefense programs, in the 1990s. The budget dropped from $17.9M in FY 1992 to $10.1M in FY 1999, resulting in the loss of personnel and delays in routine facility maintenance and upgrades (Assistant Secretary of Defense, 2004).
As priorities changed to focus on radiological emergency preparedness, the Institute’s budget stabilized in the early 2000s at $11.3M, with yearly supplements to address infrastructural needs and specific research opportunities (Assistant Secretary of Defense, 2004). Since 2006, it has fluctuated from year to year because of one-time investments made in the physical plant and infrastructure and variations in research funding. Table 4-4 delineates allocations and amounts for FY 2005–FY 2014 (AFRRI, 2013a).
AFRRI’s DoD-supplied research funding is currently derived from two accounts: research development testing and evaluation (RDT&E)—so-called Program 6.X funding—and operations and maintenance (O&M). RDT&E funding supports basic research; applied research; technology, component, and systems development; and management support of such work (DoD, 2010b). Within DoD’s Defense Health Program, O&M funding is divided into seven major areas: in-house health care, private-sector health care, information management, education and training, management activities, consolidated health support, and base operations (Defense Health Program, 2014). AFRRI also receives external research funding from other government entities and from university and private sector collaborations.
In 2005, AFRRI was funded solely through RDT&E program funding. However, in more recent years, its funding has been divided between RDT&E and O&M allocations. O&M funding represented more than 70% of all research support (including staff salaries) and more than half of the total budget in FY 2013. Additional O&M and, to a much smaller extent, RDT&E money has been devoted to facilities sustainment, restoration, and modernization (FSR&M) and infrastructure. The amount allocated for this purpose has varied considerably between FY 2005 and FY 2013, ranging between 5% and 61% of the total budget. Extramural funding has also been variable, depending on the availability of sponsored research monies and the success of investigators’ applications for it.
Figure 4-3 illustrates AFRRI’s budgets for FY 2005 through FY 2014, separating O&M research funding (all intramural), RDT&E intramural and extramural research funding, and FSR&M and infrastructure allocations. O&M funding after FY 2005 has remained relatively stable at ~$10M/year, whereas overall RDT&E funding has fluctuated between $6M and $22M/year. The six highest budget years depicted in the figure—FY 2006–FY 2008 and FY 2010–FY 2012—were all the result of one-time infusions of facilities-related money.
Research Grants and Contracts
In recent years, the public push for greater government transparency has given rise to several sources of information that detail grants, contracts, procurement, and other expenditures. To try to assess the support that AFRRI receives in the form of research grants or other awards, several online databases were searched in May 2013. Generally, few entries reflected research support given to AFRRI either directly or through the Henry M. Jackson Foundation for the Advancement of Military Medicine (HJF).7
The search indicated that AFRRI was neither the sponsor of nor the investigator on any clinical trials registered on the National Institutes of Health (NIH) website www.ClinicalTrials.gov. The Institute was also not associated with any grants accessible on the www.Grants.gov clearinghouse site, and no awards were found in the System for Award Management (SAM, 2013), a searchable database covering several federal government procurement systems.
The awards database of DoD’s Congressionally Directed Medical Research Programs (CDMRP, 2013) indicates that AFRRI has received monies totaling ~$3.6M since 1995. The funded projects include two that are associated with low-dose radiation exposure: Evaluation of the Health Risks of Embedded Depleted Uranium (DU) Shrapnel on Pregnancy and Offspring Development in 1995 and Preconceptional Paternal Exposure to Embedded Depleted Uranium Fragments: Transmission of Genetic Damage to Offspring in 2007.
A search of HHS’s Tracking and Accountability in Government Grants System website (tags.hhs.gov) did not identify any grants directly awarded to AFRRI. However, from 2008 through 2012, HJF received awards totaling ~$1.5M for the study “Ciprofloxacin Enhances DNA Repair Capacity After Radiation Combined Injury” from the National Institute of Allergy and Infectious Diseases (NIAID).
_________________
7 The Henry M. Jackson Foundation is a not-for-profit organization approved by Congress to support USUHS and military medical research (HJF, 2013).
TABLE 4-4 AFRRI Budgets (in $K) for Fiscal Years 2005–2014 by Funding Category, Form of Appropriation, and Source
| Funding Category | Appropriation | Source |
| Research | ||
| Applied Research (6.2) | RDT&E | intramural |
| Advanced Technology Development Research (6.3) | RDT&E | intramural |
| DMRDPGuidance for Development of the Force | RDT&E | extramural |
| Congressional funds | RDT&E | extramural |
| Other DoD Components, Federal Agencies, etc. | RDT&E | extramural |
| All RDT&E research | ||
| Operations & Maintenance: Direct Research Support | O&M | intramural |
| Operations & Maintenance: Indirect Research Support | O&M | intramural |
| All O&M research | ||
| All research related | ||
| Infrastructure and Facilities | ||
| Research Infrastructure (onetime) | RDT&E | intramural |
| Facilities Sustainment, Restoration & Modernization | O&M | intramural |
| All infrastructure and facilities related | ||
| Total Budget: (Intramural and Extramural) | ||
NOTES: DMRDP = Defense Medical Research and Development Program; DoD = Department of Defense; FY = fiscal year; RDT&E = research development testing and evaluation funds; O&M = operations and maintenance funds. FY 2013 and FY 2014 funding allocations may have been affected by cuts implemented as a result of the Budget Control Act of 2011.
* Estimated July 2013.
SOURCE: AFRRI, 2013a.
| FY05 | FY06 | FY07 | FY08 | FY09 | FY10 | FY11 | FY12 | FY13 | FY14* |
| 9,427 | 3,166 | 3,236 | 3,223 | 2,997 | 3,239 | 3,464 | 3,558 | 1,139 | 1,216 |
| 2,127 | 783 | 799 | 797 | 736 | 699 | 733 | 739 | 250 | 304 |
| 0 | 0 | 0 | 0 | 0 | 3,589 | 1,026 | 762 | 849 | 3,813 |
| 1,350 | 3,352 | 1,682 | 3,705 | 6,975 | 800 | 0 | 0 | 0 | 0 |
| Not avail. | Not avail. | Not avail. | 3,214 | 4,327 | 7,772 | 5,372 | 4,939 | 2,442 | 3,487 |
| 12,904 | 7,301 | 5,717 | 10,939 | 15,035 | 16,099 | 10,595 | 9,998 | 4,680 | 8,820 |
| 0 | 7,964 | 5,826 | 5,321 | 5,043 | 6,054 | 5,954 | 5,418 | 7,078 | 7,558 |
| 0 | 1,723 | 3,566 | 4,214 | 4,571 | 4,047 | 4,124 | 4,203 | 4,285 | 4,366 |
| 0 | 9,687 | 9,392 | 9,535 | 9,614 | 10,101 | 10,078 | 9,621 | 11,363 | 11,924 |
| 12,904 | 16,988 | 15,109 | 20,474 | 24,649 | 26,200 | 20,673 | 19,619 | 16,043 | 20,744 |
| 0 | 0 | 0 | 0 | 0 | 6,000 | 1,984 | 3,600 | 0 | 0 |
| 0 | 26,672 | 17,636 | 17,922 | 1,285 | 19,495 | 13,583 | 8,197 | 4,686 | 6,363 |
| 0 | 26,672 | 17,636 | 17,922 | 1,285 | 25,495 | 15,567 | 11,797 | 4,686 | 6,363 |
| 12,904 | 43,660 | 32,745 | 38,396 | 25,934 | 51,695 | 36,240 | 31,416 | 20,729 | 27,107 |
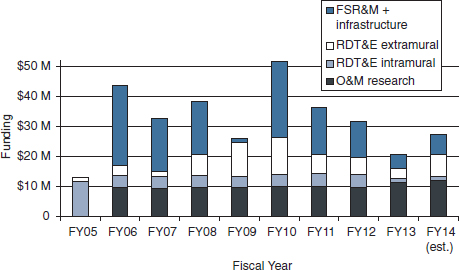
FIGURE 4-3 AFRRI funding FY 2005–FY 2014 (all sources).
NOTES: No information on extramural funding from DoD components, other federal agencies, and outside sources is available for FY 2005–FY 2007.
FSR&M: facilities sustainment, restoration, and modernization; O&M: operations and maintenance; RDT&E: research development testing and evaluation.
SOURCE: AFRRI, 2013a.
Other Agency Support
A May 15, 2013, search of USAspending.gov8 using “AFRRI” as a keyword identified 135 contracts since 2006. These accounted for $74.9M and were contracted through the Army, Navy, TRICARE, and USUHS (see Table 4-5). Contracts were mainly awarded for maintenance, materials, and, in a few cases, staffing. The largest one reported was for building repair, contracted through the Army for $13.3M in 2006. Also included was $0.5M to the National Academy of Sciences’ Research Associateship Program for 2010–2012, contracted through USUHS.
It should be noted that the accuracy of these data is subject to timely and accurate reporting by each agency and to the frequency of updates to the online database. Changes may occur as errors are corrected and more information is submitted, and contract amounts may change as modifications are made and other contracting issues occur (USAspending.gov, 2013).
_________________
8USAspending.gov fulfills a provision of the Federal Funding Accountability and Transparency Act of 2006 (Public Law 109-282) requiring the Office of Management and Budget to maintain a publicly accessible database of all federal contracts.
TABLE 4-5 Base and Exercised Options Contract Amounts (in $M) to AFRRI by Contracting Agency and Year
| Contracting Agency | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | Total |
| Navy | 1.1 | 0.3 | 0.8 | 0.5 | 1.9 | 0.0 | 4.6 | ||
| Army | 20.2 | 11.6 | 12.6 | 0.1 | 11.2 | 7.5 | 63.2 | ||
| TRICARE | 3.9 | 0.1 | 4.0 | ||||||
| USUHS | 0.3 | 0.4 | 1.1 | 0.7 | 0.5 | 3.1 | |||
| Total | 20.2 | 13.0 | 13.3 | 1.9 | 0.8 | 12.2 | 13.3 | 0.1 | 74.9 |
SOURCE: USASpending.gov data as of May 15, 2013.
STAFF AND CAPABILITIES
AFRRI’s current and future research is dependent on its staff and resources. The Institute’s labor force grew from 254 in 1965 to 285 in 1990, followed by budgetary cuts that reduced personnel in the 1990s. In 2004, the staff comprised 154 employees, 97 of whom were civilian (Assistant Secretary of Defense, 2004; Solyan, 2004), and in 2011, there were 183 employees, of whom 130 were civilian (AFRRI, 2011a).
As of July 2013, AFRRI’s Scientific Research Department comprised 68 employees:
- 19 PIs: 17 civilian federal employees and 2 military personnel,
- 14 Research Associates: 13 contractors and 1 military personnel member, and
- 35 Technicians: 22 contractors, 9 civilian federal employees, and 4 military personnel.
The AFRRI scientific staff is interdisciplinary and diverse. Its PIs come from a range of educational backgrounds. Two have doctoral degrees in the field of Radiation Biology or Radiobiology, a third has a master’s-level degree in the same field, and three others have received specialized training or completed postdoctoral work directly related to the field (radiation countermeasures, radiation biodosimetry, and cell radiobiology). Other fields of doctorate study among the PIs include biodefense, bioinformatics, bio-inorganic chemistry, biology, chemistry, medicinal chemistry and molecular pharmacology, medicine, microbiology, nutritional chemistry, nuclear particle physics, physiology, space life sciences, toxicology, and zoology. Their specialized training includes algorithm development, programming and micro-array, cell and molecular biology, immunology, oxidative stress and cancer research, molecular biology or developmental biology, hematology, B-complex vitamins, neurophysiology or neurophamacology, neuroendocrinology, DNA
radiochemistry, neurotechnology, pharmacology, toxicology, transport physiology, and image analysis and microscopy (AFRRI, 2013a).
The PIs earned their degrees from both domestic and international institutions. Seven PIs received doctoral degrees in countries other than the United States (India [3]; China, Germany, Italy, and Russia [1 each]) (AFRRI, 2013a). The most recent doctorate among the AFRRI PIs was attained in 2011; the range of years when the doctorates were received extends back to 1974, indicating that more than half of the PIs with doctoral degrees have at least 30 years’ experience since completing their degree (median year of attainment, 1983) (AFRRI, 2013a).
These PIs participate in AFRRI’s key research areas: six conduct biodosimetry research, eight conduct countermeasures research, four conduct research on combined injuries, two investigate internal contamination and heavy-metal toxicity; some participate in two research areas. However, not everyone fits into these categories. For example, one PI focuses on radiation neutralization to better understand radiation’s effect on microbial infection and the immune system and how radiation can be an effective tool to inactivate microbial threats (sterilization, sanitation, remediation of contaminated sites, and the like) (AFRRI, 2013a). Nine PIs serve as USUHS faculty members in the Departments of Preventive Medicine and Biometrics; Radiation Biology; and Anatomy, Physiology, and Genetics (AFRRI, 2013a).
Two PIs list low-dose research topics among their interests (AFRRI, 2013a). One PI conducts work on low-dose radiation carcinogenesis models (Miller, 2011), was lead author of a literature review on late and low-level effects of ionizing radiation published in the 2013 Textbook of Military Medicine (Miller et al., 2013), and carries out studies on the effects of DU exposure (for example, Miller et al., 2010). A second PI notes an interest in the effects of low-dose-rate radiation that models the fallout environment (AFRRI, 2014a). In addition—as noted in Chapter 5—some of the other work conducted by investigators has potential low-dose applications.
AFRRI posts on its website a list of journal articles produced by its staff (AFRRI, 2014c). Of the 127 articles published or in press from 2010 through May 15, 2014,9 only two explicitly mention doses below 1 Gy. Both of these relate to biodosimetry: One examined the utility of giant magnetoresistive nanosensors for measuring protein concentrations in blood for medical diagnosis (Kim et al., 2013), and the other evaluated the minipig for its potential in γ-H2AX-based biodosimetry after exposure to ionizing radiation from 137Cs and 60Co sources (Moroni et al., 2013).
As already noted, the Veterinary Sciences Department, which manages the animal facility, is staffed with specially trained individuals, including
_________________
9 This averages to nearly two papers per year per PI over this time period, including papers authored by more than one PI.
veterinarians, veterinary technicians, animal husbandry personnel, pathology staff, and administrators, who are Army, Air Force, federal, and contract employees.
COLLABORATIONS AND REPRESENTATION IN SCIENTIFIC GROUPS
As of July 2013, AFRRI listed a number of current and recent collaborations on various research projects, listed in Box 4-3. Collaborative projects reflect all of the Institute’s key areas: radiation biology, countermeasures, biodosimetry, combined injury, and heavy-metal toxicity (AFRRI, 2013a).
Three of the projects are specified as investigations of low-dose radiation exposure. Two of these three are with USUHS investigators—one on the development of a low-dose radiation-induced skin cancer model and the other on low-dose radiation cancer risks; the third is a collaboration with a NASA researcher on low-dose-rate radiation effects.
Earlier, AFRRI collaborated with various other academic and governmental organizations for DU research: Columbia University, NIH and NCI, University of Paris, United Kingdom Medical Research Council, French Institute of Nuclear Security, Memorial Sloan Kettering Cancer Center, New York University, University of Maine, Armed Forces Institute of Pathology, and the University of Maryland School of Medicine (Kalinich et al., 2005).
AFRRI seeks extramural support for a portion of its research. In July 2013, the Institute reported 21 extramural projects. Sponsors include the Biomedical Advanced Research and Development Authority (BARDA) (three awards), Cleveland BioLabs (to evaluate a countermeasure), the Defense Medical Research and Development Program (DMRDP) (four awards, all for countermeasures research), DTRA (six awards: five for countermeasures research and one for triage biodosimetry), NASA (the previously mentioned low-dose-rate study), NIAID (five awards for countermeasures, one to develop a pediatric radiation injury model), and Xavier University (for a countermeasures study). These extramural awards sponsor the research of 10 different AFRRI PIs (AFRRI, 2013a).
AFRRI personnel also participate in a number of committees and working groups associated with DoD, the Military Services, other government agencies (including HHS, U.S. Department of Homeland Security, Environmental Protection Agency, National Institute of Standards and Technology, and Office of Science and Technology Policy), and national and international organizations (including the National Council on Radiation Protection and Measurements, International Atomic Energy Agency, International Organization for Standardization, North Atlantic Treaty Organization, and World Health Organization) (AFRRI, 2013a).
BOX 4-3
Current and Recent AFRRI Research Collaborators as of Mid-2013
Governmental Agencies
Army Research Labs, Department of Veterans Affairs, Lawrence Berkeley National Laboratory, NASA, National Cancer Institute (NCI), National Institute on Alcohol Abuse and Alcoholism (NIAAA), Sandia National Laboratory
Medical and Academic Institutions
Albert Einstein College of Medicine, Columbia University Center for Radiological Research, Indiana School of Medicine, National Space Biomedical Research Institute, New York University Medical School, Roswell Park Cancer Institute, Sloan Kettering Institute, Southwest Research Institute, Stanford University, Tulane University, University of Arkansas, University of California, Los Angeles, University of Maryland School of Medicine, University of New Hampshire, University of New Mexico, USUHS, Wake Forest University, Xavier University
Private-Sector Entities
Cellerant Therapeutics, Cleveland BioLabs, Eukarion, Hollis-Eden Pharmaceuticals, Humanetics Pharmaceuticals, LaMotte Corporation, Meso Scale Diagnostics, Tech Micro Services
International Collaborations
Commonwealth Scientific and Industrial Research Organization (Australia), Defence Research and Development Canada, Health Canada, University of Western Ontario (Canada), Commission of Atomic Energy (France), University of Paris (France), Hannover Medical School (Germany), Bhabha Atomic Research Centre (India), Institute of Nuclear Medicine and Allied Sciences (India), ENEA National Institute of Ionizing Radiation Metrology (Italy), Hirosaki University (Japan), National University of Singapore, World Health Organization
The Institute indicates that “[a]dditionally, more than 200 companies and researchers have engaged AFRRI in collaborations or discussions regarding novel radiation countermeasure candidates.”
SOURCE: AFRRI, 2013a.
SUMMARY, FINDINGS, AND CONCLUSIONS
AFRRI’s Programs, Research, and Resources
AFRRI is the only DoD entity dedicated to ionizing-radiation health-effects research. Its unique infrastructure includes a number of radiation sources that may be used to study acute and chronic effects on cells and animals, and it maintains a vivarium that houses mammals, including nonhuman primates, used in studies. The Institute’s research portfolio principally comprises work addressing biodosimetry, combined injury, internal contamination and metal toxicity, and countermeasure development. It disseminates the results of these studies in refereed journal papers, reports, books and book chapters, and other publications. Some projects are supported by and in some cases conducted at the behest of government or private-sector funders, and the remainder are initiated by PIs and supported internally. AFRRI also fulfills its mission by producing manuals and protocols on radiation-exposure response, conducting education and training in these areas, supplying nuclear and radiological emergency response assistance, and providing advice to the federal government. Approximately a third of the Institute’s 19 PIs hold appointments (primarily adjunct) at the USUHS, teaching classes or lecturing there, and mentor students. USUHS and other graduate students can perform research at the Institute. AFRRI hosts participants in fellowship programs and participates in science, technology, engineering, and mathematics outreach. Its budget—which was ~$21M in FY 2013—has fluctuated in recent years, including some large infusions to maintain and upgrade the physical plant, but overall it and the Institute’s staffing levels are lower than they were in the early 1990s.
Findings and Conclusions
AFRRI contributes to ionizing-radiation health-effects research by providing specialized expertise and abilities for evaluating, modeling, and countering the consequences of exposure to nuclear and radioactive agents. Assessing risks and creating models to predict casualties from combined injuries and internal contamination (radiation or metal poisoning resulting from embedded shrapnel made of DU or tungsten alloys) permits better military decision making. Developments in biological dosimetry increase the speed and accuracy of radiation dose assessment; these developments include assays that use blood, urine, or hair for screening, and improvements in equipment design that aid in the triage, medical management, and treatment of radiation-exposed personnel. Researchers at AFRRI continue to investigate the mechanisms of radiation damage and pursue the development of treatments for persons exposed to harmful doses of ionizing
radiation by using molecular and cellular approaches and animal models. These scientific advancements are also applicable to the aerospace industry and flight personnel exposed to cosmic and solar radiation and to the civilian population in cases of terrorism and industrial accidents that result in radiation exposure.
The second element of the statement of task called on the committee to assess how AFRRI programs are advancing research in radiobiological science related to human health risks from exposures to low-dose ionizing radiation. Although AFRRI has conducted a small number of studies at low doses, low-dose radiation exposure was not a specifically defined research area at the time this report was written. In the dose range 1 Gy and below, studies include the development of models to study carcinogenesis and non-targeted effects at the molecular level. Late effects of radiation (including internal and external contamination from DU), countermeasures to prevent those late effects, and associated biomarkers are also being studied. Areas of research that address both low- and high-level exposure include the tissue and cellular effects of combined injuries on the skeletal system (for example, bone marrow and bone loss), countermeasures to the effects of low-dose-rate gamma radiation encountered in nuclear fallout, and some of the Institute’s biodosimetry and exposure characterization work.
As noted in this chapter, the Institute’s current portfolio of studies is focused almost exclusively on exposures above 1 Gy—a range that the research community and international organizations classify as moderately high and high dose. This work is consonant with AFRRI’s mission and yields information that is vital to managing the consequences of nuclear and radiological material releases as a result of armed conflicts, terrorist actions, and accidents. It does not, though, generate knowledge that would help answer the questions identified in Chapter 2 as being important to understanding the health risks of low-dose radiation exposure. The committee thus concludes that, although AFRRI carries on a robust program of research on the biological and health effects of high-dose ionizing radiation exposure, it is not currently substantively advancing low-dose research.
Chapter 5 draws on the material presented here to offer findings, conclusions, and recommendations about opportunities for AFRRI to advance its mission for understanding human health risks from exposures to low-level ionizing radiation, with special emphasis on DoD military operations and personnel.
REFERENCES
AFRRI (Armed Forces Radiobiology Research Institute). 1968. Organization. AFRRI Staff Memorandum 10-1. https://www.osti.gov/opennet/servlets/purl/16004533-h9b0K3/16004533.pdf (accessed February 10, 2014).
AFRRI. 1993. Annual report on AFRRI research for fiscal year 1992. Bethesda, MD. http://www.dod.mil/pubs/foi/operation_and_plans/NuclearChemicalBiologicalMatters/910.pdf (accessed February 10, 2014).
AFRRI. 2009. Former AFRRI Scientific Director was pioneer in radiation research. http://www.usuhs.edu/afrri/news/pioneer.htm (accessed December 30, 2013).
AFRRI. 2010. Ceremony welcomes new AFRRI director. http://www.usuhs.edu/afrri/news/new_director2010.htm (accessed March 2, 2014).
AFRRI. 2011a. AFRRI: A unique national resource. Presented at the 2011 Society of American Federal Medical Laboratory Scientists (SAFMLS) annual meeting, New Orleans, LA, March 31. http://www.safmls.org/2011/2011%20Presentations/ST%2014/AFRRI%20-%20A%20Unique%20National%20Resource.pdf (accessed March 2, 2014).
AFRRI. 2011b. Pocket guide—emergency radiation medicine response. http://www.usuhs.edu/afrri/outreach/pdf/AFRRI-Pocket-Guide.pdf (accessed December 30, 2013).
AFRRI. 2013a. Responses to questions provided by the National Academy of Sciences concerning the statement of work for the Committee on Research Directions in Human Biological Effects of Low-Level Ionizing Radiation. Bethesda, MD, July 12, 2013.
AFRRI. 2013b. Responses to supplementary questions provided by the National Academy of Sciences concerning the statement of work for the Committee on Research Directions in Human Biological Effects of Low-Level Ionizing Radiation. Bethesda, MD, October 31, 2013.
AFRRI. 2013c. About AFRRI. http://www.usuhs.edu/afrri/organiza/about_afrri.htm (accessed October 8, 2013).
AFRRI. 2013d. Biodosimetry. http://www.afrri.usuhs.mil/research/biodos.htm (accessed February 12, 2013).
AFRRI. 2013e. Biodosimetry Assessment Tool. http://www.usuhs.edu/afrri/outreach/pdf/BAT_brochure.pdf (accessed December 30, 2013).
AFRRI. 2013f. Internal contamination and metal toxicity. http://www.usuhs.edu/afrri/research/metal-tox.htm (accessed December 30, 2013).
AFRRI. 2013g. Combined injury: Radiation with other insults. http://www.usuhs.edu/afrri/research/combined-injury.htm (accessed December 30, 2013).
AFRRI. 2013h. Medical effects of ionizing radiation (course brochure). November 2013. http://www.usuhs.edu/afrri/outreach/pdf/MEIR_brochure.pdf (accessed April 12, 2014).
AFRRI. 2013i. The Medical Radiobiology Advisory Team. http://www.usuhs.edu/afrri/outreach/pdf/describeMRAT.pdf (accessed February 2, 2014).
AFRRI, 2014a. Biography, Dr. Mark H. Whitnall, AFRRI Scientific Advisor. http://www.usuhs.edu/afrri/organiza/Bio-DrWhitnall2014.pdf (accessed May 15, 2014).
AFRRI. 2014b. Countermeasures. http://www.usuhs.edu/afrri/research/rcp.htm (accessed February 12, 2014).
AFRRI. 2014c. Journal articles 2010–2019. http://www.usuhs.edu/afrri/outreach/journal2010s.htm (accessed May 15, 2014).
AFRRI. 2014d. Medical effects of ionizing radiation (in-person) course. http://www.usuhs.edu/afrri/outreach/meir/meir.htm (accessed February 5, 2014).
AFRRI. 2014e. Medical/operational guidance for managing radiation casualties. http://www.usuhs.edu/afrri/outreach/guidance.htm (accessed February 10, 2014).
AFRRI. 2014f. Product quick list. http://www.usuhs.edu/afrri/outreach/infoprod.htm (accessed April 7, 2014).
AFRRI. 2014g. Research programs. http://www.usuhs.edu/afrri/research/research.htm (accessed February 10, 2014).
AMEDD (Army Medical Department). 2013. Medical consequences of radiological and nuclear warfare. http://www.cs.amedd.army.mil/borden/Portlet.aspx?id=b3cb37ed-08e7-4617-a40c-f148ee3d2303 (accessed December 30, 2013).
Assistant Secretary of Defense. 2004. Report to Congress: Armed Forces Radiobiology Research Institute (AFRRI). Washington, DC, September 20, 2004.
Carter, R. E., and D. M. Verrelli. 1973. AFRRI cobalt whole-body irradiation. Technical Note 73-3. Bethesda, MD: Armed Forces Radiobiology Research Institute.
CDC (Centers for Disease Control and Prevention). 2012. Strategic National Stockpile (SNS). http://www.cdc.gov/phpr/stockpile/stockpile.htm (accessed December 30, 2013).
CDMRP (Congressionally Directed Medical Research Programs). 2013. Search awards. http://cdmrp.army.mil/search.aspx (accessed December 30, 2013).
Defense Health Program. 2014. Fiscal year (FY) 2014 budget estimates: Appropriation highlights. http://comptroller.defense.gov/Portals/45/Documents/defbudget/fy2014/budget_justification/pdf/09_Defense_Health_Program/VOL_I/VOL_I_Sec_1_PBA-19_Introductory_Statement_DHP_PB14.pdf (accessed May 14, 2014)
Dix, M. A. 2005. The Armed Forces Radiobiology Research Institute. Uniformed Services University of the Health Sciences Journal 2004/5 Edition. Pp. 461-496.
DoD (Department of Defense). 2006. Instruction: Armed Forces Radiobiology Research Institute. Directive number 5105.33. http://www.dtic.mil/whs/directives/corres/pdf/510533p.pdf (accessed December 30, 2013).
DoD. 2010a. DoD response to nuclear radiological incidents. Department of Defense Directive 3150.08. January 20, 2010.
DoD. 2010b. Financial management regulation, Volume 2b: Budget formulation and presentation (Chapters 4–19). Department of Defense Directive 7000.14-R. September 2010. http://comptroller.defense.gov/Portals/45/documents/fmr/current/02b/Volume_02b.pdf (accessed May 14, 2014).
DoD. 2013. Nuclear Weapon Accident Response Procedures (NARP). Department of Defense Manual 3150.08. August 22, 2013. www.dtic.mil/whs/directives/corres/pdf/315008m.pdf (accessed February 2, 2014).
DTRA (Defense Threat Reduction Agency). 2002. Defense’s Nuclear Agency 1947–1997. Washington, DC: U.S. Department of Defense, DTRA History Series.
DTRA. 2010. Nuclear Test Personnel Review (NTPR) program fact sheet. http://www.dtra.mil/documents/ntpr/factsheets/NTPR_Program.pdf (accessed February 5, 2014).
Dubois, A., N. Fiala, R. H. Weichbrod, G. S. Ward, M. Nix, P. T. Mehlman, D. M. Taub, G. I. Perez-Perez, and M. J. Blaser. 1995. Seroepizootiology of Helicobacter pylori gastric infection in nonhuman primates housed in social environments. Journal of Clinical Microbiology 33(6):1492-1495.
FedBizOps.Gov. 2013. Cobalt-60, remove and replace. Solicitation number: HT940413-Q-0020. https://www.fbo.gov/?s=opportunity&mode=form&tab=core&id=614ea9482f61dd43ae19febece66f2e1&_cview=1 (accessed February 11, 2014).
FEMA (Federal Emergency Management Agency). 2013a. The national response framework. http://www.fema.gov/media-library/assets/documents/32230?id=7371 (accessed February 2, 2014).
FEMA. 2013b. The Nuclear/Radiological Incident Annex. http://www.fema.gov/media-library/assets/documents/25554 (accessed February 2, 2014).
Gamble, R. 2013. Personal communication, H. Ray Gamble, Director, Fellowships Office, National Research Council, May 5, 2013.
General Atomics. 2014. TRIGA nuclear reactors. http://www.ga.com/triga (accessed February 11, 2014).
HJF (Henry M. Jackson Foundation). 2013. About HJF. http://www.hjf.org/about (accessed December 30, 2013).
Huff, L. A. 2013a. Armed Forces Radiobiology Research Institute: Presentation to the Committee on Research Directions in Human Biological Effects of Low-Level Ionizing Radiation.Washington, DC, May 3, 2013.
Huff, L. A. 2013b. Personal communication, e-mail to the committee, Washington, DC, November 1, 2013.
Huff, L. A. 2014. Personal communication, e-mail to the committee, Washington, DC, January 10, 2014.
Kalinich, J. F., C. A. Emond, T. K. Dalton, S. R. Mog, G. D. Coleman, J. E. Kordell, A. C. Miller, and D. E. McClain. 2005. Embedded weapons-grade tungsten alloy shrapnel rapidly induces metastatic high-grade rhabdomyosarcomas in F344 rats. Environmental Health Perspectives 113(6):729-734.
Kang, A., D. Gibson, and M. Dempsey. 2011. AFRRI: A unique national resource. Presented at the 2011 Society of American Federal Medical Laboratory Scientists (SAFMLS), New Orleans, LA, March 31, 2011.
Kim, D., F. Marchetti, Z. Chen, S. Zaric, R. J. Wilson, D. A. Hall, R. S. Gaster, J. R. Lee, J. Wang, S. J. Osterfeld, H. Yu, R. M. White, W. F. Blakely, L. E. Peterson, S. Bhatnagar, B. Mannion, S. Tseng, K. Roth, M. Coleman, A. M. Snijders, A. J. Wyrobek, S. X. Wang. 2013. Nanosensor dosimetry of mouse blood proteins after exposure to ionizing radiation. Science Reports 3(2234).
Landauer, M. R., D. G. McChesney, and G. D. Ledney. 1997. Synthetic trehalose dicoryno-mycolate (S-TDCM): Behavioral effects and radioprotection. Journal of Radiation Research 38(1):45-54.
Landauer, M. R., C. A. Castro, K. A. Benson, J. B. Hogan and J. F. Weiss. 2001. Radio-protective and locomotor responses of mice treated with nimodipine alone and in combination with WR-151327. Journal of Applied Toxicology 21:25-31.
McKinney, L. C., E. M. Aquilla, D. Coffin, D. A. Wink, and Y. Vodovotz. 1998. Ionizing radiation potentiates the induction of nitric oxide synthase by IFN-g and/or LPS in murine macrophage cell lines: Role of TNF-α. Journal of Leukocyte Biology 64(4):459-466.
Mele, P. C., C. G. Franz, and J. R. Harrison. 1990. Effects of ionizing radiation on fixed-ratio escape performance in rats. Neurotoxicology and Teratology 12(4):367-373.
Miller, A. C. 2011. Development of models to study radiation-induced late effects. In RTG 033 Subgroup 1: Radiobiology mechanisms and late effects. HFM Panel-099 RTG-033 Activity: Radiation bioeffects and countermeasures. Bethesda, MD. http://ftp.rta.nato.int/public/PubFullText/RTO/TR/RTO-TR-HFM-099/$$TR-HFM-099-Pre-Release-ALL.pdf (accessed May 15, 2014).
Miller, A. C., M. Stewart, R. Rivas. 2010. Preconceptional paternal exposure to depleted uranium: Transmission of genetic damage to offspring. Health Physics 99(3):371-379.
Miller, A. C., M. Satyamitra, S. Kulkarni, T. Walden. 2013. Late and low-level effects of ionizing radiation. In Medical Consequences of Radiologicial and Nuclear Weapons, edited by A. B. Mickelson. Fort Detrick, MD: Office of the Surgeon General. Pp. 195-215.
Moroni, M., E. Lombardini, R. Salber, M. Kazemzedeh, V. Nagy, C. Olsen, M. Whitnall. 2011. Hematological changes as prognostic indicators of survival: Similarities between gottingen minipigs, humans, and other large animal models. PLoS ONE 6:e25210.
Moroni, M., D. Maeda, M. H. Whitnall, W. M. Bonner, C. E. Redon. 2013. Evaluation of the gamma-H2AX assay for radiation biodosimetry in a swine model. International Journal of Molecular Science 14:14119-14135.
Naquin, T. D., R. C. Bhatt, G. R. Kahles, and D. M. McKown. 2001. A low gamma dose rate cobalt-60 facility. Radiation Protection Management 18(2):31-36.
NAS (National Academy of Sciences). 2014. Associateship programs at Armed Forces Radiobiology Research Institute. http://nrc58.nas.edu/RAPLab10/Opportunity/Program.aspx?LabCode=15&ReturnURL=%2fRAPLab10%2fOpportunity%2fPrograms.aspx%3fLabCode%3d15 (accessed February 12, 2014).
NRC (National Research Council). 2004. Distribution and administration of potassium iodide in the event of a nuclear incident. Washington, DC: The National Academies Press.
NREIP (Naval Research Enterprise Internship Program). 2014. Participating labs: Armed Forces Radiobiology Research Institute. https://nreip.asee.org/labs/armed_forces_radiobiology_research_institute__bethesda__md_ (accessed February 12, 2014).
Parde, N. 2012. AFRRI: A global authority on ionizing radiation. The Journal (official newspaper for Walter Reed Army Medical Center). http://www.dcmilitary.com/ARTICLE/20120906/NEWS11/709069953/AFRRI-A (accessed February 6, 2014).
SAM (System for Award Management). 2013. System for Award Management. http://www.sam.gov (accessed December 30, 2013).
SEAP (Scientist and Engineering Apprenticeship Program). 2014. Participating labs: Armed Forces Radiobiology Research Institute. https://seap.asee.org/participating_labs/armed_forces_radiobiology_research_institute_bethesda_md (accessed February 12, 2014).
Singh, V. K., D. S. Brown, T.-C. Kao. 2010. Alpha-tocopherol succinate protects mice from gamma-radiation by induction of granulocyte-colony stimulating factor. International Journal of Radiobiology 86(1):12-21.
Solyan, D. K. 2004. Armed Forces Radiobiology Research Institute. In The Uniformed Services University of the Health Sciences: First Generation Reflections, edited by K. E. Kinnamon, pp. 309-315.
Tenforde, T. S. 2011. Tribute to AFRRI on its 50th anniversary and perspectives on the history and future of radiation biology and health protection. Presented at AFRRI Golden Jubilee Banquet, Bethesda, MD, May 12, 2011.
Thorp, J. W., and J. E. Germas. 1969. Performance of monkeys after partial body irradiation. AD699127. Bethesda, MD: AFRRI. http://oai.dtic.mil/oai/oai?verb=getRecord&metadataPrefix=html&identifier=AD0699127 (accessed February 10, 2014).
USAspending.gov. 2013. USA spending. http://usaspending.gov (accessed December 30, 2013).
USUHS (Uniformed Services University of the Health Sciences). 2012. Uniformed Services University of the Health Sciences: Learning to care for those in harm’s way. 2012 Report, 40th Anniversary Edition.
Zeman, G. H., and M. A. Dooley. 1984. Performance and dosimetry of theratron-80 cobalt-60 unit at Armed Forces Radiobiology Research Institute. Technical report. Accession Number: ADA142770. Bethesda, MD: AFRRI.




































