Science Preparedness: Conducting Research During Public Health Emergencies
The H1N1 influenza pandemic in 2009 highlighted the challenges of doing scientific research in the face of a public health crisis, said Assistant Secretary for Preparedness and Response Nicole Lurie in her keynote address. She highlighted several successes, such as the rapid characterization of the virus, determining the vaccine dose for children, surveillance for antigenic drift, and standardized data collection using existing adult and pediatric critical care research networks. However, there were also real shortcomings, for example, delays in human subjects review that limited real-time data sharing and analysis, important research questions that were not considered until it was too late to act on them, and limited biospecimen collection. Other disasters in years following 2009 also illustrated the need for a robust science response, and multiple federal agencies have begun partnering on projects to accomplish this in a coordinated and streamlined manner.
Lurie also shared her personal experience with the challenges of data sharing and research during H1N1 and other disasters. After speaking with intensive care physicians, Lurie sought to collect data to develop practice guidelines for managing critical patients with H1N1 influenza. She approached the Acute Respiratory Distress Syndrome Research Network (ARDSNet), which had an organized research network and was able to rapidly modify its data collection protocol to gather data during the pandemic. Unfortunately, no data were submitted, as participating institutions had to get IRB approval to change their protocols to be able
to submit deidentified data, a process that took up to 6 months. Eighteen months later it was learned that 40 percent of the children who died succumbed to resistant Staphylococcus infections, not H1N1—critical information that could have guided practice had it been known in a timely fashion.
Many similar research challenges were present in subsequent disasters. For example, real-time collection and analysis of data during the January 2010 earthquake in Haiti would have allowed for development of more granular clinical guidelines on treating complex fractures in austere environments, Lurie said. Following the Deepwater Horizon oil spill in the Gulf of Mexico in April 2010, the IOM convened a science workshop in the Gulf at the request of HHS to consider how to assess the effects of the spill on human health1 (IOM, 2010). At that workshop it was discussed that despite nearly 40 major oil spills in the past 50 years, all involving some kind of oil and some kind of dispersant, data remain very limited on the impacts of oil and dispersants on health. Each of those oil spills also had important behavioral health sequelae, yet there was little information on preventive behavioral health interventions. During the 2011 Japan nuclear disaster, it became clear that the United States has a limited number of experts in radiation exposure, limited laboratory capacity to handle radiation disasters, and inconsistent guidance on the use of potassium iodide.
BUILDING A ROBUST SCIENCE RESPONSE
Together, the after-action reports for each of these events demonstrate the need to do a better job of “scientific research in response,” Lurie said. This means strengthening the evidence base to inform preparedness and making decisions with the best available science. In this regard, the process for expert analysis and advice should be formally integrated into the command structure for response, she said. A process is also needed that links that best available science with research needs, a process to rapidly prioritize gaps and execute the research to resolve critical questions before the next disaster event.
To begin to address these gaps, Lurie and colleagues from NIH and CDC set out to define the basic components required to build a robust
_______________
1Additional information on this workshop can be found at http://iom.edu/Reports/2010/Research-Priorities-for-Assessing-Health-Effects-from-the-Gulf-of-Mexico-Oil-Spill.aspx (accessed September 14, 2014).
science response and what would be needed to make them operational (see Table 2-1), publishing an article in the New England Journal of Medicine. Lawrence Taybak, principal deputy director of NIH, quoted from the article that “public health emergencies, especially those that result from natural disasters, are inevitable. The failure to use research to improve our response to future disasters is not” (Lurie et al., 2013, p. 1255). Tayback added that Francis Collins, director of NIH and an author on the paper, has stressed the urgent need to develop a multifaceted plan that allows for a swift and flexible response to disasters of the future. Lurie highlighted progress in implementing some of the components, including the development of rosters of experts for some threat areas and a pilot of the process for rapid identification of research priorities during the emergence of Middle East respiratory syndrome coronavirus (MERS-CoV).
The expertise for a robust science response exists, but bureaucratic, logistical, and financial obstacles can impede strategic science. NIH has
TABLE 2-1 Components of a Robust Science Response
| Components | Making It Operational |
| Rostered experts in research design, technology, and topical areas of concern | Identify and roster experts; plan for “ready reserve” of citizen scientists and clinicians |
| Scientific research is part of core response plans | Make formal part of planning documents and incident command structure |
| Identification of knowledge gaps and research questions | Explicitly review, prioritize, and recommend which research to pursue |
| Generic and scenario-specific templates and protocols | Preapproved core survey documents; prescripted clinical protocols; minimum dataset |
| Rapid-review mechanisms for human subjects research | Advance approval, national review board for emergencies |
| Rapid funding | Implement administrative mechanisms to enable |
| Registries and networks for studies | Preprepared registries |
| Components | Making It Operational |
| Involvement of affected communities | Establish mechanisms to directly engage community to discuss concerns; share findings |
SOURCE: Lurie presentation, June 12, 2014, adapted from Lurie et al. (2013).
been working on several pilot policies to enable scientific research in response. For example, the United States has more than 138 clinical research networks. With the appropriate level of guidance and infrastructure support, Lurie said, some of these networks could be used to support collaborative medical and public health research during an emergency. The PHERRB is another mechanism that NIH is developing. It is a rapid, centralized IRB, an intramural entity positioned to review protocols quickly during an emergency. NIH is working with 20 select institutions in these clinical networks to develop “reliance agreements,” a legal agreement under which the institution agrees to rely on the PHERRB in an emergency. Lurie also suggested that the terms and conditions of awards could be used to facilitate rapid research. If a research network receives federal funding, it could, for example, be required to commit to the IRB review within 72 hours in the event of a disaster. Another policy approach is to expand access of the research community to datasets after an emergency event. For example, deidentified data from the Centers for Medicare & Medicaid Services (CMS) claims could be linked with data from the Federal Emergency Management Agency (FEMA) about who received what kind of assistance, or with data from the Department of Housing and Urban Development about housing, Census data, or other data to enable research.
Lurie described a pilot of the process to identify a research agenda, secure rapid funding, and work collaboratively to initiate the research. Following Hurricane Sandy in 2012, HHS asked the IOM and the New York Academy of Medicine (NYAM) to convene stakeholders in New York to define priority areas for recovery research less than 2 weeks after the storm.2 Participants helped identify priorities in health system response research, community resilience, mold mitigation and related health issues, morbidity and mortality among at-risk and general populations, workforce health and response, evacuation and policy decision making and health outcomes, and mental health outcomes.
_______________
2Additional information on the NYAM meeting can be found at http://www.nyam.org/news/docs/pdf/NYAM-Disaster-Research-Priorities-Meeting-Summary.pdf (accessed October 13, 2014).
Funding was secured through the emergency supplemental appropriations for Sandy disaster relief,3 and a collaborative effort among NIEHS, CDC, and ASPR awarded a series of grants for research in the identified priority areas (CDC, 2014). Although this was unique and fast moving for federal-level funding, grantees did not receive awards until 9 months after the hurricane, so there is still room for improvement.
Going forward, Lurie stressed the importance of continuing to build partnerships and relationships with local science research responders and clinical networks and to work toward prescripted clinical research protocols. To operationalize science preparedness and response efforts, she highlighted the need to; build research response into the incident command framework for disasters; test, train, and exercise plans (e.g., PHERRB); implement plans and policy tools that support research into real-world responses; develop and test tracking and exposure technologies; and develop and test capabilities for biospecimen collection.4
FEDERAL DISASTER RESEARCH INITIATIVES AND RESOURCES
As background for the panel discussions, speakers representing the federal sponsors of the workshop provided brief overviews of several current federal disaster research initiatives and resources, including the NIH Disaster Research Response Project, the National Institute for Occupational Safety and Health (NIOSH) Emergency Responder Health Monitoring and Surveillance System (ERHMS), the NIOSH Disaster Science Research Initiative, and the NLM Disaster Information Management Research Center.
NIH Disaster Research Response Project
As summarized by Linda Birnbaum, director of NIEHS and the National Toxicology Program, the main questions to be answered by research during and after a disaster are, What are the health implications of the exposures and stressors, both acute and long term, especially
_______________
3For the full text of the Disaster Relief Appropriations Act, see http://www.gpo.gov/fdsys/pkg/PLAW-113publ2/pdf/PLAW-113publ2.pdf (accessed September 9, 2014).
4For further information about the ASPR science preparedness initiative, see http://www.phe.gov/Preparedness/planning/science/Pages/default.aspx (accessed November 3, 2014).
among those who are most vulnerable (e.g., the young, old, sick, poor, disadvantaged)? Are the impacted areas safe places in which to live and work? What do we need to know to protect the public, both in the short and the long terms and to prepare for the future? One of the key challenges is getting information in a timely way.
Birnbaum introduced the NIH Disaster Research Response Project5 (DR2), a pilot project initiated, in part, in direct response to the previously mentioned article by Lurie and colleagues in the New England Journal of Medicine (Lurie et al., 2013). The goal of the project, developed by NIEHS in collaboration with NLM, is to create a disaster research system consisting of research data collection tools and a network of trained research responders. NIEHS and NLM are developing a central repository for data collection tools and research protocols, available on the NLM Disaster Lit website.6 NIEHS is developing an Intramural Fast Data Collection Team that is ready to mobilize in the event of a disaster to collect baseline, epidemiology, and clinical data, as well as biospecimens, using “plug and play” preexisting IRB-approved protocols. The NIEHS Worker Education and Training Program (WETP) is also working on developing training materials that can be used by NIH intramural and extramural researchers on how to safely conduct research in an emergency situation.
Birnbaum described a DR2 tabletop exercise held at the port of Los Angeles in April 2014, involving about 140 participants from federal, state, and local government; academia (NIEHS-funded centers and grantees); and community partners. The scenario was an earthquake-induced tsunami approaching the ports of Los Angeles and Long Beach, which are very industrialized areas with a large number of ships, tankers, oil storage areas, and train depots, as well as homes and schools. The exercise included a bus tour through the port, where it became clear that people who were in the port area when a tsunami hit could have great difficulty escaping. Participants then discussed how to incorporate health researchers into response and recovery efforts in the hypothetical disaster scenario, including barriers to data collection and ways to overcome them. Birnbaum showed a brief video of highlights from the exercise and referred participants to a report of the findings on the NIEHS website.7
_______________
5For more on this multiagency project, see http://sis.nlm.nih.gov/dimrc/dr2/disasterresearch.html (accessed on December 12, 2014).
6See http://disasterlit.nlm.nih.gov (accessed on December 12, 2014).
7The report and additional information about the exercise are available at http://tools.niehs.nih.gov/wetp/events.cfm?id=2537 (accessed December 12, 2014).
Looking forward, Birnbaum said there are opportunities to build on DR2 and other efforts, and she encouraged participants to look for opportunities for rapid collection of environmental data to go with health data and to continue to develop collaborations with federal, state, academic, and community partners, as well as industrial partners who have a great stake in the communities in which they are located. She also noted the need to explore the role of new technologies (e.g., mobile health, social media) and use the “citizen science” approach in research. NIEHS intends to hold future exercises to test the research response strategies and protocols being developed to be able to implement them in the field, and to continue to improve training.
NIOSH Emergency Responder Health Monitoring and Surveillance System
Based on experiences with a number of disasters—Hurricane Katrina in particular—NIOSH saw the need to develop a systematic approach to health monitoring and surveillance across all phases of responder involvement in a disaster (predeployment, deployment, and postdeployment). John Howard, director of NIOSH, explained that ERHMS was developed to provide tools and guidance focused on ensuring that only qualified, trained, and properly equipped personnel (employees, contractors, and volunteers) are selected for deployment and that all responders receive sufficient health and exposure monitoring. Activities addressed by ERHMS in each phase of deployment, as listed by Howard, are shown in Figure 2-1.8 The ERHMS technical assistance document was approved by the National Response Team9 for use by responders and those involved in the deployment of responders, including incident commanders, emergency managers, and agency heads.
_______________
8For further information on ERHMS, see http://www.cdc.gov/niosh/topics/erhms and http://nrt.sraprod.com/ERHMS (accessed October 9, 2014).
9The National Response Team is an organization of 15 federal departments and agencies responsible for coordinating preparedness and response to hazardous substance pollution incidents. See http://www.nrt.org (accessed November 10, 2014).
NIOSH Disaster Science Research Initiative
In January 2014, NIOSH launched the Disaster Science Research Initiative to develop a framework for an approach to timely, scalable, and scientifically sound research. The framework allows for research to be started quickly at the beginning, during, and after the response to a large-scale disaster. Areas of investigation include responder demographic ascertainment (i.e., rostering); exposure assessment (direct reading and sensor technology; real-time, continuous air monitoring; biomonitoring; analytics); personal protective equipment selection, use, and effectiveness; and responder mental health and resiliency. Responder resilience is the ability to rapidly adjust to adversity without physiological or psychological adverse effect, and is an integral component of health and safety (Reissman et al., 2009, 2011).
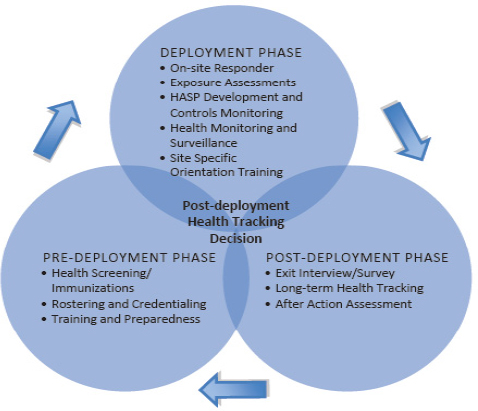
FIGURE 2-1 Emergency Responder Health Monitoring and Surveillance System comprehensive approach to responder health and safety.
NOTE: HASP = health and safety plan.
SOURCE: Howard presentation, June 12, 2014.
Howard noted that NIOSH has developed a number of direct reading instruments for workplace monitoring that have been patented and licensed. A well-known example is the personal dust monitor carried by coal miners. NIOSH is also studying biomarkers of exposure and the potential uses of in-dwelling monitors enabled by nanosensors that can send data to a central database (e.g., core temperature monitors in pill form). The goal is to make exposure detection faster so an individual can manipulate the environment and reduce exposure if necessary. Howard noted that determining when biological monitoring should be conducted can be difficult. For example, whether a scientific rationale exists for biological monitoring in a given situation, whether the monitoring results can provide meaningful and/or reliable information regarding health impact, or how such information would ultimately benefit the worker are questions to consider before implementing a monitoring system (Decker et al., 2013b).
Engaging Medical Librarians and the National Library of Medicine
Donald Lindberg, director of NLM, called attention to NLM’s vast resources, including the Disaster Information Management Re-search Center, which houses extensive health information resources and technology for disaster preparedness, response, and recovery.10 He urged participants to use these resources and those of medical libraries more generally. NLM and medical librarians should be included in planning for disaster management and recovery, he said. Specifically, he suggested that NLM could be called on to organize the published academic and noncommercial information relevant to disaster planning research and recovery for archiving and for immediate retrieval. He added that he would welcome an explicit request and the authority to organize the gray literature on disasters. Lindberg explained that many reports and resources are not as easily available as most would expect, and some can be very difficult to find, depending on how they are indexed or posted on the Internet. He also pointed out that there is an emerging specialist group of medical librarians called Disaster Information Specialists; these informaticians can be embedded in NIH research grants and paid for by NLM.
_______________
10See http://disasterinfo.nlm.nih.gov (accessed November 3, 2014).
This page intentionally left blank.










