The composition and viability of indoor microbial communities and their metabolic products are determined by the characteristics of the building they inhabit, the building’s occupants and their behaviors, and the surrounding external environment. The intricate, dynamic interplay of these elements affects human health in both positive and negative ways that are, at least currently, poorly understood.
This chapter focuses on buildings and how their characteristics and occupants shape the indoor microbiome. The chapter characterizes indoor microbial sources and reservoirs associated with air, water, and building surfaces; examines how features of the building and the environment impact indoor microbial communities and occupant exposures; considers how microbial communities affect material degradation and energy use; and identifies research needs. Subsequent chapters address how changes in one component of the built environment–microbiome–occupant nexus, intentional or otherwise, affect the others (see Chapter 5) and describe the research toolkit available for studying these interactions (see Chapter 4). This chapter is focused on microbes that have demonstrated adverse health effects; however, much of the discussion is likely applicable as well to (the far less-studied) potentially beneficial microbes.
INTRODUCTION TO MICROBIAL RESERVOIRS IN COMMERCIAL AND RESIDENTIAL BUILDINGS
There are three primary reservoirs1 for microbes found indoors: air, water, and surfaces. The primary sources for these microbes are outdoor
___________________
1 A reservoir is any person, animal, plant, material, or particle on which a microbe lives and multiplies. The reservoir typically harbors the microbe without injury to itself and serves as a source from which it may be spread (adapted from MedicineNet.com, 2017).
microbes carried indoors carried by air, water, or occupants and microbial growth that occurs indoors.
The sources and reservoirs of microbes that can be found in the air include
- mechanical heating, ventilation, and air conditioning (HVAC) systems;
- airborne particles that have been aerosolized via HVAC operation or occupant activities, such as walking or cleaning;
- outdoor air that enters through infiltration and natural or mechanical ventilation; and
- reservoirs in unfinished spaces such as crawl spaces, basements and attics, and concealed spaces (defined in Box 3-1) that are linked to occupied spaces via a range of airflow pathways.
Microbes are also found in water sources and on moist surfaces and materials, including
- municipal or well water supplies, harvested rainwater, recycled water, and drinking fountain water;
- roof, foundation, and plumbing leaks;
- condensation on or in walls and on cold water pipes;
- mechanical equipment drain pans, coils, insulation, and filters;
- cooling towers and ponds;
- whole-house or room humidifiers;
- hot water storage tanks, with subsequent aerosolization through plumbing fixtures;
- moisture generated by household appliances and food preparation;
- aerosolized water from personal hygiene practices (showering, bathing, and the like) and toilet flushing; and
- water features, including fountains, pools, hot tubs, whirlpool baths, and spas.
Finally, microbes and microbial products linked to human health may exist on surfaces of objects and materials that serve as transmission sources (called “fomites”), including microbes found in or on
- dust from floors, mattresses, furniture, and other surfaces that is resuspended in the air and inhaled;
- surfaces (doorknobs, faucets, remote control devices, keyboards, light switches) that are touched and thereby allow for dermal absorption or ingestion via hand-to-mouth;
- materials and objects that are used (toothbrushes), touched (pillows, textiles), or worn (clothing), leading to ingestion either directly or via hand to mouth; and
- soil floors and rain- or pest-damaged surfaces in crawl spaces or basements.
In addition, indoor plants, pests (such as rodents and cockroaches), and pets carry bacteria, fungi, and viruses that are then inhaled, ingested, or transmitted dermally through touch. Building occupants also may bring in or store food and beverage products that contain associated microorganisms or can support microbial growth. This report does not attempt to draw a sharp distinction between flourishing microbial communities and transient microbial presence in air, in water, and on surfaces. Built environments contain both established and transient microorganisms in different spaces and at different times, and both are affected by such practices as cleaning and remediation. However, there are still significant gaps in knowledge about the factors that shape their persistence, growth, evolution, transmission, and dynamics.
This chapter distinguishes the indoor air, water, and surface microbial reservoirs and transmission routes and details how they may lead to human exposure and are affected by building design and operation and by occupant actions. Where appropriate, it notes the distinctive features of building systems and management that lead to unique issues for residential buildings and for small and large commercial buildings.
THE DIVERSITY OF BUILDINGS AND ITS IMPACT ON THEIR MICROBIOMES
Buildings are as diverse as living things: they are differentiated by geographic location and the associated climatic conditions; their type, age, and occupancy; their HVAC and other systems; the investments made in their ongoing operations and maintenance; and the expertise of their operations staff. Like living things, moreover, they change over time. The commonalities and differences among the various types of buildings and how elements of their design, construction, and operation affect the indoor microbial environment2 are briefly discussed below.
In single-family residential buildings there is little systematic or institutional control over the introduction and management of sources of microbes, and the indoor microbiome tends to reflect the actions or inactions of the building’s occupants within the context of geographic location and seasonal variations. Mechanical, plumbing, and other systems reflect the choices of the designers, builders, and occupants; the use, maintenance, and condition of this equipment depend primarily on the owners and occupants and are highly variable. A wide range of biocides and antifungal and antibacterial chemicals of highly varying composition and inadequately studied efficacy may be used to control microbial presence in such buildings.
HVAC systems in U.S. single-family residential buildings rarely incorporate outdoor air intake but instead recirculate interior air primarily for temperature control, typically with low-efficiency particle filtration. These buildings generally have local exhaust fans in kitchens and bathrooms to remove moisture and odor, but the effectiveness and use of these systems vary greatly. As a result, these residences are ventilated primarily by weather-driven infiltration through unintentional building leakage, supplemented by the opening of windows based on outdoor weather conditions and occupant preferences. High-rise, multifamily residential buildings are more likely to incorporate some amount of mechanical outdoor air intake, often supplied to hallways, but the HVAC and outdoor air systems in such buildings vary greatly.
While the microbial environments of commercial buildings—including offices, schools, and other nonindustrial workplaces—share some similarities with residences, there are a number of important differences. Relative to residences, for example, commercial buildings tend to have
___________________
2 The discussion in this chapter is focused on building types and climate conditions found in North America and, to a lesser extent, Europe, reflecting the environments where much of the English-language research in this field has been conducted. Its applicability to building types in other parts of the world and in different climates will vary.
- greater density of occupants in contact with shared surfaces through which microbes can be transferred via doorknobs, handrails, faucets, remotes, keyboards, counters, light switches, elevator buttons, and the like;
- central HVAC systems with particle filtration and with liquid and aerosolized water associated with air conditioning coils and humidification systems that may be sources of microbes distributed to occupied spaces via mechanical ventilation;
- rooftop HVAC components (including outdoor air intakes and cooling towers) that may be exposed to standing water that supports microbial growth;
- intentional outdoor air intakes in HVAC systems in larger commercial buildings3; and
- higher pressure differentials than those in low-rise residential buildings, created by outdoor wind, the propensity for warmer air to rise (“stack effect”), and mechanical exhausts, all of which may increase the entry of outdoor air and microbes, as well as air and microbial migration between interior spaces.
Both residential and commercial buildings are characterized by widespread use of carpeting and textiles—known reservoirs of microbes—as well as maintenance and cleaning practices that may limit or promote the accumulation of microbial material and microbial growth, depending on frequency and the methods and materials used.
Characteristics of commercial buildings vary considerably by the structure’s size and use. Small and medium-sized buildings constitute the vast majority of U.S. commercial and institutional building stock, although they do not contain the majority of occupants or floor area. Such buildings—restaurants, office parks, gas stations, hair salons, bodegas, and dental offices, for example—seldom have on-site engineering staff and often rely on sometimes-distant owners or real estate management companies to manage HVAC, water intrusion, cleaning, and indoor air quality. Smaller commercial buildings tend to be designed similarly to single-family residences in terms of the building envelope and HVAC systems and are given a similar level of attention to operations and maintenance. A field study of small and medium-sized buildings in California prepared for the California Energy Commission (Bennett et al., 2011, p. 3) found that
sixteen of the thirty-seven buildings [examined in the study] did not have mechanically supplied outdoor air, including all the buildings built before
___________________
3 In contrast, many small commercial buildings rely on a combination of infiltration, local exhaust, and windows, similar to single-family residences.
1980 and 19 percent of the buildings built after 1980. In some cases, the air handling unit was generally a residential model rather than a commercial model, and thus did not have the capability to bring outdoor air inside. Air filters used in the buildings’ ventilation systems generally had low efficiency, with 56 percent having a Minimum Efficiency Reporting Value rating of four or lower. Only a quarter of the buildings had a ventilation maintenance contractor that inspected regularly. Buildings with regular contractor visits had HVAC systems that were better maintained than buildings that did not have regular inspections.
Larger commercial buildings are more likely to have dedicated in-house operations and maintenance staff. The tendency of larger buildings to have more complex HVAC systems makes the systems’ presence important, as their performance will degrade over time if they are not properly maintained, increasing the likelihood of poor control of temperature, humidity, and ventilation. However, it is not enough to have an operations and maintenance staff. A separate question is whether the budget allotted to operations and maintenance allows such tasks as filter changing, system inspections and repair, and control sensor calibration to be carried out in the manner recommended by system manufacturers.
Schools are a significant exposure environment for those 6–18 years of age. American children spend an average of 180 days per year in school, and a little more than one-quarter of each of those days is spent in the school environs (6.64 hours/day on average) (ED, 2008). A 2006 National Research Council report on “green schools” includes the following findings:
- A robust body of scientific evidence indicates that the health of children and adults can be affected by indoor air quality. A growing body of evidence suggests that teacher productivity and student learning may also be affected by indoor air quality.
- Well-designed, -constructed, and -maintained building envelopes are critical to the control and reduction of excess moisture and mold growth. (NRC, 2006, p. 6)
This report offers a number of recommendations regarding these findings, including that “future green school guidelines should emphasize the control of excess moisture, dampness, and mold to protect the health of children and adults in schools,” and that “such guidelines should specifically address moisture control as it relates to the design, construction, operation, and maintenance of a school building’s envelope (foundations, walls, windows, and roofs) and related items such as siting and landscaping” (NRC, 2006, p. 6). The report further notes that “the survival, dispersal, and removal of airborne pathogens are affected by relative humidity, ventilation rate, and the percentage of recirculated air in the air supply” and recommends “addi-
tional research . . . to determine the optimal infection-control interventions in terms of measurable outcomes such as absenteeism and academic achievement” (p. 119).
In multiunit residential (apartments, dormitories) and mixed-use buildings with domiciliary activities (hotels, firehouses), all of the sources and building features detailed above affect the overall microbiome. Within these categories of buildings, hospitals, nursing homes, and other inpatient health care facilities represent particular challenges because the sources of microbes—viruses in particular—and occupant vulnerabilities are greatest.
Only a limited literature addresses the microbiome of buildings—commercial or mixed-use—that contain retail operations. Hoisington and colleagues (2016) examined HVAC filter dust in a total of 13 electronics, furniture, grocery, home improvement, office supply, and general merchandise stores located in Pennsylvania and Texas. They found that, for this admittedly limited sample,
the indoor environment in retail stores may offer a variety of niches for microbial populations that support a diverse community as compared to other built environment studies.…The microbiome was significantly influenced by several parameters including human microbiota (most notably to oral and skin bacterial communities) and the outdoor environment [but only a] tangential relationship between the bacterial community present and factors such as season, store location, and store type. (p. 685)
There are also specialized types of living and working environments, such as aircraft, submarines, and spacecraft, that pose special challenges for the management of microbes; Box 3-2 provides an example.
In some cases, buildings fail to deliver acceptable indoor environmental quality even at the time of their initial occupation. This issue has led, especially in the case of larger-scale commercial and other buildings, to a growing commitment to “building commissioning”4—a process intended to ensure that “systems are designed, installed, functionally tested, and capable of being operated and maintained according to the owner’s operational needs” (DOE, 1999, p. 9). In the past, testing, adjusting, and balancing (TAB) of HVAC components were performed only once, after construction was completed. In commissioning, this process is expanded to include dynamic testing of multiple systems, including plumbing, lighting, and the building envelope, in all modes of building operation to capture seasonal changes. Most recently, this process has been extended to include “retrocommissioning”—the same systematic process applied to existing
___________________
4 A more complete discussion of commissioning may be found in the Transportation Research Board report Optimizing Airport Building Operations and Maintenance Through Retrocommissioning: A Whole-Systems Approach (TRB, 2015).
buildings that have never been commissioned; “recommissioning”—the reevaluation of a previously commissioned building, sometimes in conjunction with changes in use or renovations involving upgrades to the physical plant; and “continuous commissioning”5—an ongoing process that uses technology to identify and address performance problems, enhance occupant comfort, and optimize energy use via monitoring and dynamic adjustment of building systems (DOE, 2007; TEES, 2017). The negative consequences of understaffing and underfunding building operations and maintenance activities and the growing complexity of both building systems and environmental factors of concern highlight the importance of ongoing commissioning and draw attention to the need to incorporate into the commissioning process consideration of the effects of the indoor microbiome on occupants. Such knowledge may one day lead to the use of measures of the state of the indoor microbiome as part of the commissioning process.
The next three sections of this chapter identify indoor air, water, and surface microbial reservoirs and transmission routes and detail how they may lead to human exposure and how they are affected by occupant actions.
INDOOR AIR SOURCES AND RESERVOIRS OF MICROBES
Air is a critical transport vehicle of microbes and their metabolites in the built environment because it connects surfaces, water, and dust to what occupants inhale, inadvertently ingest, or absorb through their skin. The sources of the microbial communities that make up the microbiome of the built environment include indoor and outdoor sources of bacteria, fungi, and viruses, and the movement of air is a significant factor in their distribution. Indeed, the impact of these microbes on human health is often a function of how they become airborne and subsequently move through a building. This section reviews four key factors in the air transport of indoor and outdoor microbes: air leakage through unintentional openings in the building air envelope, internal migration of air between zones, mechanical ventilation, and natural ventilation.
In practice, most residential and commercial buildings are ventilated through a combination of envelope infiltration and mechanical and natural ventilation. HVAC systems in commercial buildings are designed and operated to maintain temperature and humidity within a comfortable range and to ensure the delivery of outdoor air for ventilation. In residential buildings, until recently, mechanical systems rarely incorporated outdoor air intake, relying on infiltration and operable windows to provide outdoor air. Engineered natural ventilation systems involving, for example, thermal chimneys and stack effect (discussed below) with carefully designed and located
___________________
5 Also known as “ongoing commissioning” or “monitoring-based commissioning.”
inlet vents are now becoming prevalent in northern Europe and Asia. The combination of these natural ventilation strategies with mechanical ventilation with or without mechanical cooling is termed “hybrid” or “mixed-mode” ventilation, and interest has been growing in using this approach to achieve energy savings while maintaining a healthy and comfortable indoor environment (Chenari et al., 2016; Heiselberg, 2006).
Increased attention has been paid to indoor air chemistry in recent years, revealing many important mechanisms affecting the fate and transport of airborne chemical contaminants within buildings (Morrison, 2015; Nazaroff and Goldstein, 2015; Weschler, 2011, 2016). This work, however, has to date not focused on impacts of indoor microbiomes. Accordingly, one of the areas for future investigation suggested by Adams and colleagues (2016, p. 227) is “how does the microbiome affect indoor chemistry, and how do chemical processes and the composition of building materials influence the indoor microbiome?” The authors’ summary of available information on the topic notes that while the chemical metabolites produced by microbes may affect indoor chemistry, evidence suggests that their impact may be weak. Chemical agents in the environment may influence the microbiome, though, with research finding that differences in growth substrate lead to differences in microbial composition and metabolite production on wetted materials, and source strength may drive microbial community structure (Adams et al., 2016). A greater understanding of the extent to which indoor chemistry influences and is influenced by the building microbiome should result from the research recommended in the present report and from work being conducted under the Alfred P. Sloan Foundation’s Chemistry of Indoor Environments6 initiative.
Air Leakage Through Unintentional Openings in the Building Envelope
Airflow into and within a building can bring outdoor microbes indoors and transport them throughout the structure, and it can also transport microbes from interior sources from one part of a building to another. Although certain airflows are established by design (e.g., via mechanical ventilation systems), others are unintended and uncontrolled. Air leakage through unintentional openings in the building envelope7—known as “air infiltration”—is an important pathway for bioaerosols to enter buildings. Leaky building envelopes in both residential and commercial buildings lead
___________________
6 See https://sloan.org/programs/science/chemistry-of-indoor-environments (accessed July 14, 2017).
7 “Building envelope” is the collective term for the physical separators between the interior and exterior of a building, comprising such components as walls, floors, roofs, windows, skylights, and doors.
to considerable outdoor air entry, and even very “tight” buildings have non-negligible infiltration levels (Ng et al., 2015). Leakage alone can result in outdoor air ventilation rates equal to the lower range of mechanical ventilation rates (Grot et al., 1989). Research suggests that in naturally ventilated buildings nearly all particles in the diameter range 0.1–10 μm can flow through leaks in the building envelope with no significant losses (Liu and Nazaroff, 2001; Nazaroff, 2016).
Infiltration is driven by differences between indoor and outdoor air pressures, which vary with outdoor weather conditions, including wind velocity and direction relative to building exterior surfaces and their exposure to the wind, as well as differences between outdoor and indoor air temperatures. It is also affected by the operation of building equipment, including furnaces and boilers, local exhaust fans in bathrooms and kitchens, and mechanical ventilation systems that may have an imbalance between outdoor air intake and exhaust. Such infiltration and exhaust airflows often are highly complex, even in apparently simple buildings, and may be subject to significant short-term variations. These effects need to be considered to understand the impacts of airflow on microbial transport and growth.
Internal Migration of Air Between Zones
Building envelope construction, mechanical infrastructure, and interior layout define important physical features of a building that impact how airborne microbes may come into contact with occupants. These features define a building’s major space conditioning (heating and cooling) zones. Pressure differences among zones combined with airflow pathways lead to interzone airflow and movement of airborne contaminants. In turn, these features drive airflow and surface and material moisture levels in relation to the available sites for potential microbial growth.
Building height plays an important role in creating indoor–outdoor pressure differences, especially when it is colder outside the building than inside. A phenomenon known as the stack or chimney effect results in significant airflow into lower floors of a building that then moves upward and flows out of the building at higher levels. During summer cooling periods, when it is colder inside than out, these airflow directions are reversed. The stack effect in a building is enhanced by its vertical shafts—elevators, stairwells, plumbing, and other service chases—providing important paths for airflows that can transport microbes. Variations in space or zone temperatures and their humidity levels—whether they are conditioned or unconditioned, occupied or unoccupied, or directly connected to the outdoors—play a role in microbial growth and movement of airborne microbes to other building spaces.
The height and shape of a building also will impact its exposure to wind. That exposure can in turn influence infiltration of outdoor air and
microbes into the building, as well as amplify air pressure differences among building zones that affect indoor microbial migration.
Mechanical Ventilation
While HVAC systems meet important thermal and ventilation needs in buildings, these systems also provide routes for the entry of outdoor bioaerosols into buildings, as well as a means of circulating and dispersing indoor airborne contaminants. HVAC systems may themselves harbor microbial reservoirs, especially when water is involved (as described in the following section). These systems also affect temperature and moisture conditions throughout a building’s interior spaces and within interior and exterior walls—conditions that affect the state of the indoor microbiome.
The delivery of outdoor air, or ventilation, through HVAC systems varies greatly among buildings and at different times within the same building, variations that are compounded by different approaches to HVAC control and operation. Most ventilation systems incorporate particle filtration, either of the outdoor air intake or of mixed airstreams of outdoor and recirculated air from occupied spaces. The efficiency with which particles are removed is a function of filter type, particle size, and airflow rate. The most common air filter effectiveness classification system in the United States is the MERV (Minimum Efficiency Reporting Value) rating,8 with higher numbers corresponding to a higher fraction of particle removal. However, the MERV rating for filters commonly chosen for residential and commercial buildings indicates that they are not very efficient at removing submicron particles (<1.0 μm in mean aerodynamic diameter), which limits their effectiveness in reducing some airborne microbes.
In addition to the effectiveness of filters relative to microbial management, it is also important to gain a greater understanding of the effect of operating HVAC systems intermittently—for example, turning systems off over weekends and holidays—and the extent to which this intermittent operation enables microbes to collect on the filter medium, metabolize and multiply, and then be released when the system restarts (ASHRAE, 2009).
The design and placement of outdoor air intakes for mechanical ventilation systems can also influence the indoor microbial environment. The U.S. Environmental Protection Agency (EPA) guidance for schools suggests that “intakes should not be placed within 25 feet of any potential sources of air contaminants, including . . . mist from cooling towers”; that they should be screened to prevent birds and rodents from fouling and intro-
___________________
8 American National Standards Institute (ANSI)/American Society of Heating, Refrigerating and Air-Conditioning Engineers (ASHRAE) Standard 52.2-2012. Method of Testing General Ventilation Air-Cleaning Devices for Removal Efficiency by Particle Size.
ducing microbes into HVAC systems; and that systems should be designed to cause “moisture to flow to the outside or to a drain if intake grilles are not designed to completely eliminate the intake of rain or snow” (EPA, 2017b). Similar requirements are contained in American National Standards Institute (ANSI)/American Society of Heating, Refrigerating and Air-Conditioning Engineers (ASHRAE) Standard 62.1-2016 (Ventilation for Acceptable Indoor Air Quality),9 with the goal of keeping organic materials and moisture out of HVAC systems.
Studies have shown associations between increased ventilation rates and improved health outcomes, including reduced incidence of influenza and asthma and allergy symptoms10 (Seppänen and Fisk, 2004; Sundell et al., 2011). Although higher prevalence of sick building syndrome symptoms is seen in air-conditioned buildings relative to naturally ventilated buildings (Finnegan et al., 1984), studies relating ventilation rates and health generally fail to describe how, when, and where ventilation rates were measured (Persily and Levin, 2011), and they often ignore buildings with strong indoor sources of air pollutants and locations with poor outdoor air quality (Sundell et al., 2011).
Existing Ventilation Standards and Measured Performance
Existing standards and building regulations include requirements for outdoor air ventilation rates and exhaust airflow rates for different building types and space uses intended to provide standards for model code requirements (ASHRAE, 2016a,b). For example, ASHRAE Standards 62.1, 62.2, and 170 contain minimum ventilation requirements for commercial and institutional buildings, residential buildings, and health care facilities, respectively.
ASHRAE’s ventilation standard defines minimum values for acceptable HVAC system performance and is used in building design and construction when required by code. However, actual ventilation and exhaust airflow rates usually are quite different from those specified by codes or design documents as the result of a range of shortcomings in system installation, commissioning, operation, and maintenance. In some cases, building uses (and therefore, pollution sources) change after the system was designed,
___________________
9 As defined by the standard, acceptable indoor air quality is “air in which there are no known contaminants at harmful concentrations as determined by cognizant authorities and with which a substantial majority (80% or more) of the people exposed do not express dissatisfaction” (ASHRAE, 2016a, pp. 6–7). Outside air used in ventilation also must meet National Ambient Air Quality Standards or be filtered, and local sources of concern may be identified. However, these evaluations are unlikely to specifically consider microorganisms.
10 Factors other than ventilation also influence these respiratory health outcomes.
so that the design values are no longer relevant to the building as it exists (Persily et al., 2005).
Mechanical ventilation systems use a variety of approaches to control outdoor air entry and ventilation air distribution within buildings to achieve adequate minimum ventilation air delivery while saving energy. Minimum outdoor air intake rates are specified in the system design, based on standards and regulations. Ideally, these rates are verified during system commissioning and monitored occasionally during the life of the building, but this is rare in practice. Some buildings modulate the rate of outdoor air intake based on indicators of building occupancy (e.g., air temperature and carbon dioxide concentrations), providing less outdoor air during times of low occupancy as an energy-efficiency measure. This approach, referred to as “demand-controlled ventilation,” may employ occupancy sensors, indoor carbon dioxide level detectors, or other strategies. Ventilation systems also may be configured to increase outdoor air intake when the outdoor air is cool and dry as an energy-efficient cooling mechanism, referred to as an “economizer cycle,” replacing use of a mechanical system to lower the air temperature. Research to advance the development of real-time indoor and outdoor microbial sensors could be valuable, with data from such sensors being integrated into dynamic HVAC control systems.
Most studies of building ventilation performance to date have considered a small, nonrepresentative set of buildings, and authors often provide incomplete descriptions of the ventilation measurement methods employed (Persily, 2016). The most recent comprehensive studies yielded mixed results as to whether building ventilation systems actually meet current standards. The Building Assessment Survey and Evaluation (BASE) study, conducted in the 1990s by EPA, included ventilation and other indoor air quality measurements for 100 randomly selected large U.S. office buildings (Persily and Gorfain, 2008; Persily et al., 2005). The mean measured outdoor air ventilation rate was 49 L/s per person based on the occupant densities during the time of the ventilation measurements, a rate that exceeds the minimum requirements of ventilation standards. These high air change rates occurred in part because the systems often operated in economizer (energy-saving, free cooling) mode or because the actual space occupancies were, on average, 80 percent of the design values. Considering only minimum outdoor air intake operation and accounting for the lower occupancy levels, the mean ventilation rate was about 11 L/s per person at default occupancy values in ASHRAE Standard 62.1, which is based on achieving <20 percent dissatisfaction with perceived indoor air quality (ASHRAE, 2016a). Approximately one-half of the values were below the minimum requirements in Standard 62-1999 or the designed ventilation rates.
In low-rise residential buildings, uncontrolled air leakage across the building envelope can also be an important source of outdoor air ventila-
tion, and these air change rates vary with geography and such factors as the age and size of the home (Koontz and Rector, 1995). A field study of 108 new single-family homes in California included measurements of outdoor air change rates (Offermann, 2009). The median 24-hour outdoor air change rate was 0.26 h–1, with a range of 0.09 to 5.3 h–1. Sixty-seven percent of the homes had outdoor air change rates below the minimum California Building Code requirement of 0.35 h–1. The author notes that the combination of relatively tight envelope construction and “the fact that many people never open their windows for ventilation . . . resulted in many homes with low outdoor air exchange rates” (p. 210).
Mechanical Conditioning and Ventilation Systems and Indoor Microbiomes
Because building mechanical conditioning and ventilation systems are designed primarily for thermal environmental control (i.e., air temperature and relative humidity), conditions relevant to airborne microbes generally are affected by system status, design, and operation and other internal building conditions (including occupant numbers and activities). Where heating demand dominates design considerations, indoor air will be less humid than colder outdoor air because of the reduced relative humidity from warming of the air.11 Where air conditioning is dominant, indoor air will be cooler than the air outdoors, and management of the air conditioning system will determine the moisture content of the indoor air and its potential effect on airborne microbes. Of special interest is the effect of air humidity on the survival and pathogenicity of infectious airborne agents, such as viruses and bacteria associated with seasonal flu, the common cold, and other diseases.
Mechanical systems may distribute microbes to occupants or disperse microbes from humans with bacterial or viral infections. HVAC design employs a range of ductwork configurations and components to distribute air to occupied zones and control the airflow as a function of thermal requirements in the zones and other factors. In many commercial buildings, zoned air distribution systems are designed to mix the ventilation air with the conditioned room air based on thermal comfort goals, a system known as “mixing ventilation.” Displacement ventilation systems and task air systems that deliver air directly to occupants are also being implemented in some buildings, and research suggests that such systems could be more effective than mixing systems in creating healthier indoor environments (Kong et al., 2017). Also seeing increasing application are 100 percent outside air systems, also known as dedicated outdoor air systems (DOASs), with many such buildings being designed to separate ventilation from heating and
___________________
11 The reduction in humidity from warming of the air will be offset by such indoor sources as cooking, showering, and occupant metabolism.
cooling. Such systems are more typically found in hospitals and are often combined with a means of heat recovery.
Natural Ventilation
Natural ventilation has been used for centuries to bring outdoor air into buildings and to circulate it within the building interior. It offers the benefits of resiliency in the face of power outages and the potential for higher levels of outside air to purge indoor pollutants when wind speeds or indoor–outdoor temperature differentials are sufficiently high. However, natural ventilation can lead to the entry of outdoor contaminants and moisture. It is limited by the driving pressures that induce airflows into the building, and the ventilating air entering the building may not be well distributed among occupied spaces or the rates of entry well controlled. As a result, to ensure a given continuous minimum rate of ventilation, natural ventilation systems increasingly are being designed and integrated with mechanical ventilation on a climate-specific basis. Critical design factors include such parameters as the ratio of window opening size to floor area; the ratio and locational relationships of openings to each other, as well as their relative positions in the room; and the influence of wind velocity and direction on air distribution in the room (Levin, 2010). Door openings also may have a significant influence on the amount of outside air that enters a building, especially in some types of commercial buildings. A Pacific Northwest National Laboratory report notes that “restaurants, strip-mall stores, retail stores, supermarkets, offices and hospitals are likely to have high door-opening frequency, either at certain time periods of day or in some cases throughout the occupied hours” (Cho et al., 2010, p. 1) that lead to high infiltration rates. Such airflows may raise or lower the concentration of indoor pollutants, including microbial agents, depending on their outdoor concentrations (Zaatari et al., 2014).
Design guidance and tools for natural ventilation include the Chartered Institution of Building Services Engineers’ Applications Manual AM10 (CIBSE, 2014) and the National Institute of Standards and Technology’s LoopDA (NIST, 2017).
Indoor Air Sources and Reservoirs of Microbes: Summary of Findings
Air leakage through unintentional openings in the building envelope, internal migration of air between zones, the distribution of air by mechanical conditioning and ventilation systems, and natural ventilation affect the association between air and the indoor microbiome. These interrelationships involve bioaerosol and moisture generation, contaminant transport by
natural and mechanical ventilation elements, and human behaviors that resuspend particles or modify ventilation. Building airflow, which constitutes an important transport mechanism for indoor air contaminants, is complex and often poorly controlled. Building ventilation systems—mechanical, natural, and hybrid—do not necessarily perform as intended or expected. These facts highlight the importance of considering microbial growth in relation to air-related transport in a building and the interplay of human behaviors and use patterns related to the indoor microbiome.
Ventilation and filtration have the potential to control the quality of airflow and indoor microbial conditions, but exercising this control requires sound maintenance practices and proper operation, especially the management of wet environments that may contribute to microbial growth. Generally speaking, over the past several decades commercial buildings in the United States have employed mechanical conditioning and ventilation systems along with tighter building envelopes without operable windows. This “sealed” approach to building ventilation is quite distinct from the earlier use of operable windows and natural ventilation (Banham, 1984). The implications of the shift to sealed buildings for indoor air quality, occupant satisfaction and performance, and indoor microbiomes have yet to be thoroughly researched.
INDOOR WATER SOURCES AND RESERVOIRS OF MICROBES
Water is essential to the viability (growth and survival) of microbes. The periodic or episodic presence of water can sustain some microbial life and even support reproduction and growth. As noted in Chapter 2, there is general agreement that dampness and mold within buildings are associated with unfavorable health outcomes, although many studies examining this issue have important methodological considerations that limit their generalizability. Water that enters a building from public or private supply sources can also contain pathogens, including Legionella pneumophila and Mycobacterium avium, that present health risks.
While buildings generally are intended to be and stay dry, water still can be present in four forms—liquid, vapor, adsorbed moisture, and ice—depending on air and surface temperatures, physical characteristics of surfaces and materials, and water concentration. Adsorbed moisture is water that is held on the surfaces of a material by intermolecular forces. It has inherently different properties from those of moisture in its liquid and frozen states, as well as from those of moisture that has been absorbed and chemically bound by a material, becoming part of its chemical structure. These different states influence how efficiently the water can play a role in biologic processes or chemical reactions; the more tightly bound it is, the less of it is available for such processes or reactions.
Buildings contain many different water sources and reservoirs. The most obvious is municipal or well water brought into the building deliberately through premise plumbing. Unwanted liquid water results from plumbing leaks and leakage through the building envelope (exterior walls, roofs, floor slabs, crawl space, or basement) or when capillary action draws groundwater through pores in building materials (so-called rising damp). Water vapor commonly migrates when air pressure differences between connected spaces result in transport of air and its contents from the region of higher pressure to that of lower pressure (although diffusion due to humidity differences can also be important). In addition to high outdoor vapor pressure, water vapor is also produced by such activities as showering, cooking, and washing clothes. Water vapor and adsorbed moisture and ice can enter the building enclosure when air pressures and temperatures and humidities inside and outside a building change, causing transfer of heat and moisture between the air and the building’s materials. Adsorbed moisture in building materials, such as gypsum panels or rain-soaked construction lumber, can influence mold growth on surfaces, on interior walls, and in the concealed spaces within walls. Recycled water—which may be used indoors in such applications as toilet and urinal flushing under certain circumstances (Los Angeles County Department of Public Health, 2016)—can harbor microbial contaminants if not properly managed (Toze, 2006). Finally, green building practices such as rainwater harvesting or wastewater treatment and reuse can be a source of microbes in the indoor environment (Ghaitidak and Yadav, 2013).
The following sections address six examples of water-related sources of potentially problematic microbes:
- premise plumbing
- hot water heaters
- cooling towers, cooling coils, and drain pans
- leakage, flooding, and wet building materials
- indoor water sources and airborne moisture generators
- indoor humidity
Premise Plumbing
Water piped into buildings can come from municipal water treatment plants, wells, groundwater, or surface water sources. Water entering a building commonly contains bacteria, and some potable water supplies can contain fungi as well. Premise plumbing—a building’s plumbing systems and equipment—can affect the concentrations of these microbes before they reach such outlets as faucets, spigots, showerheads, and other appliances, which often aerosolize microbes directly into the breathing zone of building occupants.
Showerheads, for example, have been identified as a source of exposure to aerosolized nontuberculous mycobacteria (Falkinham, 2011; Feazel et al., 2009). The type of showerhead, the frequency and duration of its use, and the duration of stagnancy affect the amount of aerosol generated and thus the total exposure to microbes, with those showerheads that produce a lower-pressure stream generally also producing less aerosolization.
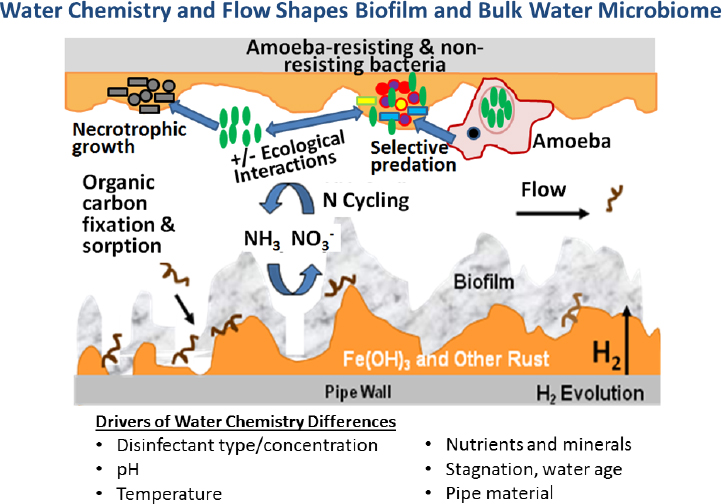
Premise plumbing pathogens are responsible for a significant number of infections whose origins have been traced to drinking water (Beer et al., 2015). These opportunistic pathogens represent an emerging waterborne disease problem with a major economic cost of at least $1 billion annually (Falkinham, 2015). Water is the source of Legionella pneumophila, Mycobacterium avium, and Pseudomonas aeruginosa, which are ubiquitous in water systems and are estimated to be responsible for tens of thousands of infections each year (Collier et al., 2012; Falkinham, 2015). The design, operation, and maintenance of premise plumbing systems are critical to controlling exposures. Figure 3-1 illustrates how water chemistry and flow shape biofilms12 in pipes and the resulting microbiome.
Premise water filters can themselves become sites for biofilms and microbial growth. Maintenance of filters and the frequency of their replacement are important factors in filter performance and the presence of microbes found in potable water at the point of use. Filters are used to remove inorganics (such as lead) or objectionable chemical agents (such as chlorine, benzene, or trichloroethylene) from the tap where water for cooking or drinking is drawn. Such filtering may leave little or no residual chlorine, which can result in growth of microbes within the premise plumbing system. The application of such filtration is an example of human behaviors that affect the microbial content (and its pathogenicity) of water in the premise plumbing system.
Building features and human behaviors that increase the likelihood of stagnant water are important to consider because stagnant water supports microbial growth. The frequency with which various components of the plumbing system are used and the design and materials of these systems determine both the locations of a building’s stagnant water and the amount of time the water will remain stagnant. During the off season, for example, showers and water features of hotels and vacation resorts may see little use, thus allowing for more microbial growth. A similar problem can occur in vacation homes.
___________________
12 Biofilm is defined as “a thin, normally resistant, layer of microorganisms such as bacteria that form on and coat various surfaces” (https://www.merriam-webster.com/dictionary/biofilm [accessed July 17, 2017]).
SOURCE: Courtesy of Drs. Amy Pruden and Hong Wang, Virginia Polytechnic Institute and State University.
Hot Water Heaters
Researchers have long known that commercial and residential hot water heaters are reservoirs for thermophilic bacteria (Brock and Boylen, 1973) and are thus a primary source of waterborne pathogens in buildings in the United States (Brazeau and Edwards, 2013). As already noted, Legionella pneumophila is a particular concern in this regard, but Brazeau and Edwards (2013, pp. 617–618) note that “Acanthamoeba, Mycobacterium avium complex, and Pseudomonas aeruginosa can also grow within water heating systems and cause thousands of cases of infections annually.”
Investigators have studied microbial communities in domestic hot water heated to different temperatures in systems with varying levels of use (Ji et al., 2017; Rhoads, 2017; Rhoads et al., 2015). The practice of lowering water heater temperatures to 120°F (49°C) to conserve energy and limit the possibility of scalding creates a circumstance whereby tap water is warm enough to support the growth of L. pneumophila but not to inactivate microbes in the biofilm normally found on the bottom of hot water tanks. And while 140°F (60°C) is hot enough to inactivate L. pneumophila in the
tank, it may not eliminate the bacterium in the pipes of preheated water for infrequent hot water users. As noted earlier, the frequency of use of hot water fixtures (such as showers) determines the length of time the water remains stagnant and the corresponding potential for microbial growth. Furthermore, Rhoads and colleagues (2015) found that under experimental conditions, 124°F (51°C) may represent a “sweet spot” for L. pneumophila in conditions of low water use enriching the concentration at the tap.
Cooling Towers, Cooling Coils, and Drain Pans
Air conditioning system cooling towers are a documented source of community outbreaks of Legionnaires’ disease, with higher rates of infection occurring closer to the source tower (Addiss et al., 1989; Weiss et al., 2017). Indeed, Legionella pneumophila have been identified as the main microbial risk in cooling towers (Torvinen et al., 2014), where they often are found growing in the warmer water associated with discharge from refrigeration and air conditioning systems. The cooling tower’s role is heat rejection to the atmosphere, with the water being cooled by evaporation or exposure to air that is cooler than the water. Because of their rooftop location, which may be near or upwind of outdoor air intakes, cooling towers can release L. pneumophila into the airstream taken in by the ventilation systems, leading to illness in the buildings where they are located. The aerosol emitted into the air stream can also be carried to nearby and even distant locations, leading to community outbreaks (Weiss et al., 2017).
Testing of cooling tower water for L. pneumophila is not required. Reported concentrations vary quite widely. There is no official guidance on treatment requirements based on concentrations. The dominant form of treatment is the addition of chemicals, although cleaning of cooling towers to remove protozoans growing on surfaces is associated with reduced concentrations of L. pneumophila in the water (Pagnier et al., 2009).
Conventional HVAC system design in air-conditioned buildings—which involves frequently wet surfaces on cooling coils, drain pans, and sometimes humidifiers—may lead to as yet uncharacterized microbiologic exposures and consequent illness (Mendell et al., 2008; Menzies et al., 2003). Poor system condition and poor maintenance increase the risk of such problems. Accumulated dust and dirt and moisture in HVAC systems provide a nutrient source and growth medium for microorganisms (Morey et al., 2009; West and Hansen, 1989).
ASHRAE has a guideline—12-2000, Minimizing the Risk of Legionellosis Associated with Building Water Systems—and a standard—188-2015, Legionellosis: Risk Management for Building Water Systems—that address the minimization of Legionella contamination in building water systems. Separately, ANSI/ASHRAE Standard 62.1-2016, Ventilation for Acceptable
Indoor Air Quality, contains several general requirements related to moisture management in HVAC systems that include, for example, the cleanability of cooling coils, the ability of condensate drain pans to collect moisture effectively and for it to be removed from the system, the specification of duct surfaces to reduce microbial growth, and the provision of access to systems for inspection and maintenance. However, many HVAC systems still have drain pans with inadequate slope so that condensate remains stagnant and microbial growth, including biofilm formation, is facilitated. The standard also contains requirements designed to minimize the likelihood of envelope-related moisture problems via air leakage and building pressure control. ASHRAE/Illuminating Engineering Society (IES)/U.S. Green Building Council (USGBC) Standard 189.1 has analogous requirements, but they are more stringent than those in Standard 62.1 given that 189.1 is a high-performance standard. These ASHRAE standards are incorporated in some “model” codes, but widespread adoption of up-to-date standards is elusive given the thousands of local code agencies and shortage of trained inspectors or enforcement agents. The building codes are intended to govern design and issuance of building permits, but not operating conditions in occupied buildings.
Leakage, Flooding, and Wet Building Materials
Water can also enter a building from a host of unintentional sources, including enclosure leakage and flooding, plumbing leakage, rising damp, condensation, and human activities. Leakage through roofs, foundations, and walls during rain is common and often results in wetting of building materials and sometimes in the accumulation of moisture. During floods or other ground saturation conditions, hydrostatic pressure (the pressure exerted by a fluid at equilibrium at a given point within the fluid, due to the force of gravity13) can force groundwater in through cracks in the slab or foundation of a building. The increased frequency and intensity of floods and hurricanes have created extreme conditions of water intrusion that need to be followed by remediation to remove or limit future microbial growth (the installation of perimeter drains, for instance) and additional measures to reduce problems with hydrostatic pressure.
Building materials also may be damaged by rain or excessive moisture prior to or during construction, which changes their physical properties and potentially contaminates them with mold spores that grow when water is subsequently reintroduced. Andersen and colleagues (2017) found fungal species that had become embedded in gypsum wallboard during the manufacturing process, before the material reached retailers or construction
___________________
13 See http://www.dictionary.com/browse/hydrostatic-pressure (accessed May 1, 2017).
sites. Keeping building materials dry prior to construction has long been recognized as a key strategy for limiting the likelihood of mold growth in buildings (EPA, 2013a).
Any excess liquid water or water vapor within a building will contribute to absorption by adjacent materials. The amount of absorption depends in part on the porosity of the material. Most building materials are hydrophilic, absorbing water readily (Straub, 2006). When water flooding a floor comes in contact with gypsum board, for example, it is slowly absorbed and by capillary action can saturate the board. The rate of drying of wet gypsum board14 depends on several factors that interact dynamically, including the porosity, temperature, and moisture content of the gypsum board; its surface coating, if any; the absolute and relative humidity of the surrounding air; the velocity at which that drying air flows across the surface; and the temperature of the air compared with that of the wet board (Dedesko and Siegel, 2015).
Water in interstitial or concealed spaces in walls, crawl spaces, attics, and HVAC chases15 supports the growth of mold. Mold and bacteria metabolism produces microbial volatile organic compounds (MVOCs) that can reach occupied spaces and be detected by the compounds’ characteristic “earthy,” “moldy,” or “musty” odors. The presence of these odors is strong evidence of microbial growth resulting from persistent dampness in concealed spaces or inside HVAC systems (Mendell and Kumagai, 2017).
Standing water in crawl spaces beneath buildings—including single-family homes and portable classrooms—may come from rainwater, groundwater, water vapor in the soil, or soil gas (DOE, 2013). Such moisture can lead to high levels of mold growth within the crawl space from which mold spores and MVOCs can be carried into occupied spaces via airflow. There are also multiple pathways through which such moisture itself can enter buildings and contribute to mold growth within occupied spaces. These mechanisms include capillary action, bulk airflow through holes and other penetrations, and vapor diffusion (DOE, 2013).
Indoor Water Sources and Airborne Moisture Generators
In addition to rain penetration with the possibility of both direct wetting of materials and standing water, microbial growth can be supported
___________________
14 Gypsum board is “the generic name for a family of panel products that consist of a noncombustible core, composed primarily of gypsum, and a paper surfacing on the face, back and long edges” (https://www.gypsum.org/technical/using-gypsum-board-for-walls-andceilings/using-gypsum-board-for-walls-and-ceilings-section-i [accessed July 17, 2017]). It may also be called drywall, wallboard, or plasterboard.
15 A chase is a wall or a ceiling feature through which ducts, electrical wires, pipes, and the like are run.
by indoor and outdoor moisture migration and condensation. Microbial growth on surfaces and within building assemblies depends on moisture availability at the surface rather than on air humidity itself—high air humidity can support high surface water activity. Human activities that generate water vapor—cooking, showering, clothes and dish washing and drying, and occupant metabolism (which is an issue in densely occupied spaces), as well as use of humidifiers—are significant sources of indoor humidity. The frequency and duration of these activities determine the release of moisture into air, and the presence or absence of effective exhaust ventilation is critical.
The human activities that impact the water vapor emission intensity indoors include the
- frequency and duration of personal hygiene practices, including showers, baths, and toilet flushing16;
- use of water for cleaning hard surfaces, such as flooring;
- frequency and duration of washing dishes and clothes;
- frequency and use of clothes dryers, with or without the exhaust of moisture to the outdoors;
- use of an exhaust hood to remove moisture emitted during food preparation; and
- use of exhaust fans or window opening during and after showering.
Hot tubs, whirlpool baths, and other spas and water features can aerosolize water and microbes and result in very high levels of exposure to aerosolized bacteria (e.g., Legionella pneumophila and Mycobacteria spp.). These water-based recreational and therapeutic systems often involve water temperatures in the range of 104–110°F (40–43°C), and they produce aerosolized bacteria in the surface water close to the head of the human occupant/user of the spa. In addition, increased humidity levels indoors can lead to higher water vapor on interior surfaces and within building materials.
Water vapor condenses more readily on cold surfaces, so interior surfaces of exterior walls and roofs in cold weather or the interior surfaces or layers of exterior walls and roofs of air-conditioned buildings in warm, humid climates increase the occurrence of unwanted condensation. Water supply pipes, wastewater pipes, and fire suppression system water pipes also tend to have colder temperatures than the air around them, which can cause condensation on these pipes. In addition, building materials that are
___________________
16 The position of the toilet seat cover also affects the amount of aerosols released into the room air.
saturated with moisture themselves possess high water activity levels that can result in fungal growth.
The indoor moisture content of air and materials varies greatly depending on the season of the year. Inside walls, attics, and roofing assemblies, the moisture content of materials is affected by seasonal surface temperatures, with cooler internal surfaces collecting and retaining more moisture than warm surfaces. Air-conditioned interiors have increased moisture absorption and therefore higher surface water activity and more potential to support microbial growth.
Biowalls—also called green or living walls—have garnered attention as a possible means of promoting a healthy indoor environment through the introduction of plants that are intended to clean the air or create a “green” atmosphere. Very little research has been done on their effectiveness in this regard, however, and because they use water in liquid and aerosolized form as part of their operation and because plants loose water into the air as a result of metabolism (transpiration), they could elevate humidity and harbor pathogens (Girman et al., 2009; Waring, 2016).
While the relationships between moisture and microbial growth have been studied extensively—especially with respect to mold’s association with allergy, asthma, and other health endpoints of interest (see Chapter 2)—the differences among geographic locations, building types, construction practices, and climates are too great to enable refinement of generalized design solutions or the development of broadly applicable guidelines or code requirements to ensure the elimination of surface and hidden mold in building assemblies.
Indoor Humidity
The moisture content of air is important to fungal viability and growth and to virus and bacteria survival and virulence. Mold growth on building materials depends on a variety of parameters, including moisture, temperature, time, and the presence of nutrients on the substrate and its pH level, but moisture is the primary driving factor (Haverinen, 2002). The relationships between air humidity and bacteria show different patterns of survival and pathogenicity depending on the species of concern.
Moisture in buildings can be characterized by various means. Relative humidity is perhaps used most commonly, both because it is comparatively easy to measure and because its levels are related to perceived occupant comfort. It is defined as the water vapor pressure of the air, expressed as a percentage of the saturated water vapor pressure at the same temperature, and thus reflects both the amount of water vapor in the air and the air temperature. However, airborne relative humidity by itself is not, in general,
predictive of mold growth on indoor surfaces,17 and it may vary considerably in an interior space depending on where it is measured.
Humidity level is thus only a part of the larger, more complex issue of how moisture affects the composition and viability of indoor microbiomes. Different humidity levels in combination with other parameters, such as ventilation and temperature, promote or suppress different viruses, bacteria, and fungi, and a microbe that thrives on one indoor surface may waste on another under the same humidity conditions. Therefore, generic advice about humidity levels needs to be viewed with skepticism.
Indoor Water Sources and Reservoirs of Microbes: Summary of Findings
Where there is water, there are likely to be microbial organisms. While few bacteria and molds have demonstrated adverse health effects, opportunistic pathogens and allergenic species are commonly found among indoor microbial contaminants. On the basis of its review of the literature and the prior findings of the Institute of Medicine (IOM) reports Damp Indoor Spaces and Health (IOM, 2004) and Climate Change, the Indoor Environment, and Health (IOM, 2011), the committee identified the need for more research on water quality supplied and delivered by premise plumbing; microbial management in building hot water heaters, cooling towers, cooling coils, and drain pans; leakage and flooding that results in moisture damage, especially in houses; indoor water fixtures, features, and airborne moisture generators; and the detection of mold and moisture inside building assemblies, especially walls and roof assemblies. More research is also needed on how to interpret moisture measurements in terms of the risk of mold and bacterial growth and the role of viruses in the evolution of the total indoor microbiome over time.
BUILDING SURFACES AND RESERVOIRS OF MICROBES
Microbes are introduced to and released from surfaces inside buildings through a number of mechanisms, including deposition of airborne microbes; transfer via occupants’ direct contact with surfaces; the tracking and deposition of dirt, dust, pests, and water into buildings from the outdoors; resuspension of deposited microbes due to a variety of activities; and bodily emissions from exhalation, expectoration, skin shedding, cuts in the skin, and bladder and bowel waste. Occupants are exposed to these microbes when they touch the surfaces (via hand-to-mouth ingestion or
___________________
17 The exception to this is high relative humidity conditions, which lead to damp indoor surfaces that are conducive to microbial growth.
direct dermal transmission) or when the surfaces are disturbed, aerosolizing the microbes and particles that may be attached to them, which are then inhaled. Indoor surfaces that can support microbial growth—including floor, wall, and ceiling materials, as well as plumbing and HVAC components—are important in designing and maintaining buildings to manage microbial communities to human advantage.
This section focuses on the relationship of building surfaces to microbes that impact human health. While the presence of fungi, bacteria, and viruses on surfaces or suspended in dust may be most medically serious in hospitals, important findings also have resulted from studies of homes, offices, fire stations, schools and kindergartens, gymnasiums, food service facilities, and dormitories. Because extensive interaction occurs among indoor air, water, and surfaces, some of the issues salient here relate to those introduced earlier in this chapter.
Direct Contact with Surfaces by Occupants
Bacterial, viral, and fungal communities are transferred to building surfaces by direct contact with occupants’ skin, saliva, and mucosa. Recent advances in DNA sequencing analysis have facilitated research on bacterial and fungal communities on surfaces in classrooms, offices, homes, gymnasiums, and other building types (Barberán et al., 2015; Chase et al., 2016; Dunn et al., 2013; Flores et al., 2013; Kelley and Gilbert, 2013; Meadow et al., 2014a,b; Yamamoto et al., 2015). A study by Haleem and colleagues (2013) yielded information about numerous bacterial and fungal taxa from surface samples collected at a university, including Bacillus sp., Candida albicans, E. coli, Fecal streptococcus, Pseudomonas aeruginosa, Klebsiella pneumonia, Staphylococcus sp., Streptococcus sp., and Trycophyton sp.
Evidence indicates that contaminated surfaces also play a role in the spread of viral infections. Table 3-1 presents the results of a compilation by Boone and Gerba (2007) of buildings and surfaces where viruses have been detected or survived.
Understanding of human interactions with surfaces has advanced sufficiently to demonstrate that each human leaves a specific microbial signature on surfaces. It has been shown that bacterial communities on a surface can be traced back to an individual for forensic purposes (Fierer et al., 2010). When new occupants enter a home, their distinct microbes can be detected in the building’s surface bacteria within days (Lax et al., 2014). This line of research demonstrates that bacterial communities on surfaces as distinct as floors, walls, chairs, tables, doorknobs, elevator buttons, keyboards, and other shared equipment contain human microbiota.
In addition to their ability to populate fomites, bacteria and viruses can remain infectious on surfaces for hours to days, and they have variable
TABLE 3-1 Buildings and Surfaces Where Viruses Have Been Detected or Survived
| Virus | Location of Virus | |
|---|---|---|
| Buildings (reference[s]) | Surfaces (reference[s]) | |
| Respiratory syncytial virus | Hospitals (23) | Countertops, cloth gowns, rubber gloves, paper facial tissue, hands (33) |
| Rhinovirus | Not found | Skin, hands (30), doorknob, faucet (52) |
| Influenza virus | Day care centers, homes, nursing home (51) | Towels, medical cart items (51) |
| Parainfluenza virus | Offices (data not published), hospitals (23) | Desks, phones, computer mouse (Boone and Gerba, submitted) |
| Coronavirus | Hospitals (23), apartments (62) | Phones, doorknobs, computer mouse, toilet handles (23), latex gloves, sponges (68) |
| Norovirus | Nursing home (6), hotels, hospital wards, cruise ships, recreational camps (22, 38, 61) | Carpets, curtains, lockers, bed covers, bed rails, drinking cup, water jug handle, lampshade (6, 38) |
| Rotavirus | Day care centers, pediatric ward (8) | Toys, phones, toilet handles, sinks, water fountains, door handles, play areas, refrigerator handles, water play tables, thermometers, play mats (8, 15, 38, 70), paper, china (2), cotton cloth, latex, glazed tile, polystyrene (1) |
| Hepatitis A virus | Hospitals, schools, institutions for mentally handicapped, animal care facilities, bars (72) | Drinking glasses (72), paper, china (2), cotton cloth, latex, glazed tile polystyrene (1) |
| Adenovirus | Bars, coffee shops (7, 24) | Drinking glasses (24), paper, china (2), cotton cloth, latex, glazed tile, polystyrene (1) |
| Astrovirus | Schools, pediatric wards, nursing homes (39) | Paper, china (2) |
NOTE: References (in parentheses) listed in source document.
SOURCE: Boone and Gerba, 2007, Table 1.
die-off responses to disinfectants and other cleaning products. For example, one study of children’s desks after cleaning found that fungal and bacterial communities recovered to precleaning loading levels after approximately 3 days (Kwan et al., 2016). In public restrooms, microbiota on floors that had been cleaned with bleach redeveloped within 5–8 hours and “showed remarkable stability over weeks to months” (Gibbons et al., 2015, p. 765). Other investigators have shown not only that school restrooms are dominated by microorganisms associated with the gastrointestinal and urogenital tracts (Flores et al., 2011) but also that microbes from these tracts are predominant on classroom chairs, along with those from the skin (Meadow et al., 2014b). Bean and colleagues (1982) found that viable influenza A and B virus could be transferred from such nonporous surfaces as steel and plastic to hands for 24 hours and from such porous surfaces as tissues to hands for 15 minutes. These and other data support the notion that viruses may be spread by indirect contact via fomites (Weinstein et al., 2003).
Identifying the surfaces of greatest significance for improved design or maintenance practices aimed at reducing bacterial and viral microbes continues to be a research challenge. In a study of intensive care units (ICUs) by Bures and colleagues (2000), the colonization rate for keyboards and faucet handles by “novel and unrecognized” taxa was greater than that for other surfaces in rooms with patients testing positive for methicillin-resistant Staphylococcus aureus (MRSA), revealing that a variety of surfaces may serve as reservoirs of pathogens and vectors for cross-transmission. A study of Boston-area homes found that the highest bacterial counts were associated with wet surfaces that are often touched, such as tubs, sinks, and faucet handles. These surfaces were found to be contaminated with bacteria, including Enterobacteriaceae, Pseudomonas, methicillin-sensitive Staphylococcus aureus (MSSA), and MRSA (Scott et al., 2009).
Of greatest concern are surfaces touched by multiple occupants—so-called high-touch surfaces—including both dry sites, such as door handles, elevator buttons, keyboards, light switches, and television remote controls, and wet sites, such as food preparation areas and kitchens, water fountains, bathroom faucets and counters, toilet seats and handles, and soap dispensers. Tables 3-2 and 3-3 illustrate the commonalities and differences among fomites of concern in residential buildings versus commercial buildings and hospitals. These tables also list surfaces of concern on which occupants sit and lie, from chairs to couches and bedding, where hand-to-mouth and mouth-to-mouth transfer is also possible. Exposure to bacterial, viral, or fungal fomites and their impact on human health depends on the sources and human susceptibility, as well as a host of physical and environmental conditions that need to be recorded in parallel with surface sampling to gain a more complete understanding of influences on microbial populations and communities (see Table 3-4). This topic is revisited in Chapter 4.
TABLE 3-2 Home High-Touch Surfaces and Bacterial Reservoirs
| All rooms | Light switches, air, dust, floor, rugs, door knobs |
| Kitchen | Countertop, sink, faucet handles, drain, u-pipe, refrigerator handle, refrigerator shelves, microwave buttons, dish sponge, drying towels, drying rack |
| Bathroom | Countertop, sink, u-pipes, shower floor, shower curtain, showerhead, shower poufs, bar soap, toilet bowl, toilet water, toilet seat, toilet flush handle, hand towels |
| Bedroom | Pillows, sheets |
| Living room | Seats, arm rests, head rests, pillows, blankets, remote controls |
| Office, etc. | Keyboard, mouse, water from water heater, mop head, HVAC filters |
SOURCE: Smith et al., 2013, Table A5-1.
TABLE 3-3 Hospital High-Touch Surfaces and Bacterial Reservoirs
| Patient area | Bed rails, tray table, call boxes, telephone, bedside tables, patient chair, intravenous (IV) pole, floor, light switches, glove box, air, air exhaust filter |
| Patient restroom | Sink, faucet handles, inside faucet head, hot tap water, cold tap water, light switches, door knob, handrails, toilet seats, flush lever, bed pan cleaning equipment, floor, air, air exhaust filter |
| Additional equipment | IV pump control panel, monitor control panel, monitor touch screen, monitor cables, ventilator control panel, blood pressure cuff, janitorial equipment |
| Water | Cold tap water, hot tap water, water used to clean floors |
| Patient | Stool sample, nasal swab, hand |
| Staff | Nasal swab, bottom of shoe, dominant hand, cell phone, pager, iPad, computer mouse, work phone, shirt cuff, stethoscope |
| Travel areas | Corridor floor, corridor wall, steps, stairwell door knobs, stairwell door kick plates, elevator buttons, elevator floor, handrails, air |
| Lobby | Front desk surface, chairs, coffee tables, floor, air |
| Public restroom | Floor, door handles, sink controls, sink bowl, soap dispenser, towel dispenser, toilet seats, toilet lever, stall door lock, stall door handle, urinal flush lever, air, air exhaust filter |
SOURCE: Smith et al., 2013, Table A5-2.
A range of solutions are used to reduce the opportunity for infection through fomites from human contact with surfaces, including
- hand hygiene—washing with soap and water or hand sanitizers;
- surface washing with disinfectants;
| Building | Room | Surface |
|---|---|---|
|
|
|
SOURCE: Smith et al., 2013, Table A5-3.
- surface sterilization with antimicrobial agents, such as bleach, ethanol, and peroxide;
- hands-free lights, doors, and elevators;
- protective covers, easy-to-disinfect surfaces, and built-in periodic cleaning reminders for keyboards and computer mouses in medical environments;
- ultraviolet (UV) light irradiation via lamps or sunlight; and
- architectural design that is mindful of the potential for microbial contamination, such as separate wet and dry walking areas in gyms and pools or not placing restrooms next to areas where food is prepared.
While some of these approaches are marketed as effective tools for infection control, evidence of their efficacy often is sparse or nonexistent, and improper use of some interventions can result in the development or promotion of disinfectant-resistant microbes. Research thus is needed to better understand the determinants of effective infection control. Such research might include, for example, examining whether biofilm-resistant or antimicrobial compounds and materials actually reduce the accumulation of bacterial, viral, or fungal microorganisms on a surface and further
experimentation with articulated surface topography as an alternative to chemistry to reduce biofilm development (Xu and Siedlecki, 2012).
Surfaces That Support Microbes Tracked in from Outdoor Sources
While deposition of airborne microbes via settling is a primary means of populating indoor surfaces, occupants also affect the indoor microbiome by tracking dirt, dust, pests, and water into buildings from the outdoors. Fungal and bacterial microorganisms are brought indoors on shoes and clothing, along with pests and flora that carry microbes of concern. Clothing and shoes are a source of moisture, dirt, pollen, and mold from soil, plants, pests, and animals outdoors, along with fungi and bacteria that can be aerosolized into the breathing zone or contacted directly from surfaces. In a chamber bioaerosol study, Adams and colleagues (2015) compared the relative abundance of bacterial and fungal taxa in indoor air, outdoor air, and dust. The authors concluded that “the microbial communities observed in the indoor air samples largely tracked those simultaneously measured outdoors, and taxa known to be associated with the human body played a secondary although important role” (p. 5 of 18). Meadow and colleagues (2014b) found that the floors and walls of a university building were dominated by species from the outdoors that may have been introduced via foot traffic or the HVAC system.
Simple modifications may be effective at reducing the transport of outdoor microbes indoors, although the support for such measures is derived largely from common sense rather than data. For example, Simcox and colleagues (2012) identify several steps they deem “prudent” for reducing MRSA contamination in Washington State firehouses from sources external to the building, including placing multilevel scraper walk-off mats at entrances,18 leaving work boots outside of living quarters, vacuuming frequently, and replacing cloth surfaces with hard surfaces wherever possible. However, the investigators acknowledge that one research study on the issue19 found that the use of walk-off mats was statistically significantly associated with the presence of MRSA on surfaces (p = 0.02), a counter-intuitive result they suggest could be due to a statistical issue known as the multiple comparisons or multiplicity problem. They do, however, deem that finding worthy of further investigation. Research on such related issues as the effect of removing shoes upon entering a home on indoor microbial communities would be valuable as well.
___________________
18 Such mats are required by green building standards such as Leadership in Energy and Environmental Design (LEED).
19 Later expanded on by Roberts and No (2014).
Surfaces That Suspend Dust and Enable Resuspension
Humans also alter the indoor microbiome by shedding microbes onto receptive surfaces and by engaging in activities that resuspend existing surface microbes, which expose occupants to bacteria or fungi on dust particles. Depending on their aerodynamic diameters, aerosolized microbes and particles can be deposited on surfaces as a result of Brownian motion (below 0.1 μm); accumulate in the air and be inhaled directly (0.1–1 μm); or settle on surfaces as a result of gravity, impaction, and interception (above 1 μm).
Interest in human shedding of microbes from skin (Noble et al., 1976), as well as through coughing, sneezing, and talking (Morawska, 2006), is long-standing. Researchers have found shed or flaked (desquamated) human skin cells in aerosols in occupied indoor environments (Clark, 1974; Fox et al., 2008) and routinely have noted the presence of human microbiota such as Staphylococcus, Propionibacteria, Corynebacteria, and enteric bacteria (Täubel et al., 2009). A 2015 study found that human “bacterial clouds” are distinct with respect to their community structure and that humans carry personalized microbial clouds (Meadow et al., 2015). Findings of statistical associations and material balance–based studies suggest that resuspension can be a significant source of airborne bacteria and fungi; in densely occupied settings, it is the primary source (Hospodsky et al., 2012; Qian et al., 2012).
While shed skin can be inhaled directly by other building occupants, it is more typically shed into dust on floors and building surfaces and then resuspended through walking, vacuuming, dusting, and other activities (Meadow et al., 2015). Resuspension most critically exposes those crawling or seated, who are closer to the highest concentrations (Täubel, 2016). The emission and aerosolization of dust during vacuuming can potentially spread bacteria, including Salmonella spp. and Clostridium botulinum (Veillette et al., 2013). In the absence of combustion, cooking, and smoking, resuspension is a major source of total airborne particulate matter in occupied indoor environments, suggesting it is an important mechanism for the aerosolization of microbes (Qian et al., 2014). Yamamoto and colleagues (2015, p. 5104) indicate that research on this topic highlights “the importance of reducing indoor emissions associated with occupancy, potentially through more regular and effective floor cleaning and through the choice of flooring materials that limit particle resuspension.”
There is strong evidence that human occupancy increases fungal and bacterial concentrations in indoor air through resuspension. Dust is hygroscopic and contains a substantial portion of microbes. Luoma and Batterman (2001) found that resuspension due to walking explained 24–55 percent of variation in 1–25 μm diameter particle concentrations in
homes. Similarly, a study of six school classrooms found that human occupancy resulted in significantly elevated airborne bacterial (by a factor of 81, on average) and fungal (factor of 15) concentrations (Hospodsky et al., 2015). Microbial community analyses conducted in schools demonstrate that resuspended floor dust is enriched in bacteria and fungi associated with human skin and that surface-based resuspension may be a major source of airborne fungal allergens, rather than infiltration of these microbes into buildings from outdoor air (Hospodsky et al., 2015; Yamamoto et al., 2015). Other research revealed that classroom emission rates from shedding and resuspension were on the order of 10 million bacteria or fungi per person per hour (Hospodsky et al., 2015). If total particles are used as a measure of human impact on the indoor microbiome, the effect of resuspension appears to be much stronger than the effect of direct human emissions (Hospodsky et al., 2012). Importantly, it has been demonstrated that particles larger than 5 μm are suspended more easily than smaller ones, indicating a differential impact of human occupancy on this aspect of indoor microbial exposure (Thatcher and Layton, 1995).
Beds represent a significant but underrecognized exposure micro-environment because they are a place where people spend large amounts of time in close proximity to sources. Boor and colleagues (2015, p. 442) note that “mattresses are possible sources of a myriad of chemical species, such as volatile organic compounds (VOCs), plasticizers, flame retardants, and unreacted isocyanates (Boor et al., 2014; Stapleton et al., 2011) [and] mattresses, pillows, and bedding serve as an accumulation zone for a diverse spectrum of particles, many of which are of biological origin.” The biologic matter includes multiple allergens, fungi, and bacteria. A chamber study conducted by the authors of that study revealed that resuspension from typical sleep activities was an important source mechanism.
It is not clear how best to address shedding and resuspension as sources of microbes that may be harmful to human health. Studies suggest that rates of resuspension of microbes are influenced by flooring material type (Qian and Ferro, 2008), floor dust loading (Qian et al., 2014), human activity level (Ferro et al., 2004), and particle size (Qian and Ferro, 2008). Other studies reveal the benefits of hard surfaces over carpet and upholstery in reducing microbial levels in resuspended dust (Buttner et al., 2002) and of UV-C equipped vacuum cleaners in reducing the viable microbial load in carpets (Lutz et al., 2010). However, substantial gaps remain in research on the determinants of and the most effective way of reducing the resuspension of microbes in the full range of building types and indoor environmental conditions.
As previously discussed in this chapter, humans also affect the indoor microbiome through bodily emissions from exhalation, expectoration, skin shedding, cuts in the skin, and bladder and bowel waste. Through such
actions as coughing and sneezing, the body emits aerosols that become incorporated into a building’s microbiome. Such factors as the diameter of the viral-, bacterial-, or fungal-bearing particles; occupant density; occupant proximity (e.g., face-to-face exchange); and ventilation system configurations affect how these particles are distributed once they are emitted from the body (Li et al., 2007; Liu et al., 2017).
Surfaces That Sustain Dampness and Mold
The final surface design and maintenance issue of concern is surfaces that support growth of mold because they are or may become damp or wet. These include surfaces found in the living spaces of buildings—particularly the floor, wall, and ceiling materials—and those that are in concealed spaces, such as HVAC components and insulation within framed walls (Mensah-Attipoe et al., 2015).
Building materials, furnishings, and other surfaces differ in their ability to sustain damp conditions and their susceptibility to deterioration resulting from the presence of those conditions. A key factor in this regard is water activity (aw) or equilibrium relative humidity (ERH), which indicates whether a particular material is damp enough to support microbial growth. ERH characterizes the relative humidity of the atmosphere in equilibrium with a material that has a particular moisture content. Different materials used indoors, such as brick, concrete, drywall, textiles, and wood, may have very different ERHs in the presence of the same airborne relative humidity level. ERH is difficult to measure accurately in situ, however, with results varying depending on how the sample is taken (Dedesko and Siegel, 2015).
The implications of damp surfaces are discussed in detail in the IOM report Damp Indoor Spaces and Health (IOM, 2004), which addresses the interactions among moisture, materials, and environmental conditions within and outside a building that determine whether the building may become a source of potentially harmful dampness-related microbial and chemical exposures. In brief, that report notes that mold spores are found regularly on indoor surfaces and materials, and their growth, which usually is accompanied by bacterial growth, is determined primarily by the availability of moisture since the nutrients and temperature range they need to grow are usually present. The primary risk factors for the dampness that supports microbial growth differ across climates, geographic areas, and building types. This dampness can also damage building materials and furnishings, causing or exacerbating their release of chemicals and nonbiologic particles.
More recently, Adams and colleagues (2015) examined how the composition of building materials influences the indoor microbiome. Their overview of the literature finds that “different building materials and envi-
ronmental conditions (e.g., temperature, available water, cleaning chemicals and frequency, light intensity at certain wavelengths, and carbon sources) can create different selective pressures for microorganisms if varied over wide ranges, which can result in differential survival and persistence rates” (p. 226). They note, for example, that “wooden materials show greater fungal diversity than plasterboard or ceramics, and cellulose-based materials are more sensitive to contamination by fungal growth than inorganic materials such as gypsum, mortar, and concrete” (p. 228). Verdier and colleagues’ (2014) review of the literature on indoor microbial growth across building materials and sampling and analysis methods finds that the bioreceptivity of materials is determined most strongly by their water activity, chemical composition (in particular, nutrient sources), pH, and surface physical properties (porosity, surface roughness, and the like). The presence of fungi, bacteria, and viruses and their by-products in materials has led some manufacturers to introduce antimicrobial agents into their products. However, a white paper by the design firm Perkins+Will (2017) asserts that there is no evidence that these additives result in a healthier indoor environment, and they may in fact have negative effects on occupants and the environment through, for instance, the promotion of antibiotic-resistant species.
Several factors have led to increases in indoor dampness problems in the United States in the past several years. These include the construction of air-conditioned buildings in hot, humid regions and in areas previously considered to be wetlands, and the greater use of moisture-retaining gypsum board (Weschler, 2009). The health implications of damp indoor spaces—which are addressed in Chapter 2 of this report—are illustrated by a study by Sordillo and colleagues (2013) in 376 Boston area homes, in which visible water damage and mold or mildew were associated with a 20–66 percent increase in levels of Gram-negative bacteria associated with childhood asthma. This and other studies reveal the importance of designing building surfaces and systems so as to limit the opportunity for water damage.
Such interventions are especially important at a time when increases in the frequency and geographic spread of deluge rain, high temperatures, and high humidity conspire to speed the growth and migration of mold spores. A number of IOM and National Research Council reports have identified the importance of managing dampness and mold to improve human health and have listed critically needed research (IOM, 1993, 2000, 2004, 2011; NRC, 2006).
Numerous means exist for controlling moisture in buildings and thus limiting the growth of mold and reducing the spread of mold spores. EPA (2013a, p. 1) identifies the two key principles as
- preventing water intrusion and condensation in areas of a building that must remain dry; and
- limiting the areas of a building that are routinely wet because of their use (e.g., bathrooms, spas, kitchens, and janitorial closets) and drying them out when they do get wet.
Voluntary standards specify actions that can achieve these goals, but few of these standards have made their way into building regulations and other enforceable instruments that would lead to their widespread implementation. And research to evaluate the effectiveness of these interventions—especially in the long term—is lacking.
Building Surfaces and Reservoirs of Microbes: Summary of Findings
Surfaces constitute a critical reservoir for microbial growth and transfer in the built environment. The specification, design, and maintenance of building surfaces need to be critically evaluated to reduce microbial sources and transfers that occur by (1) direct contact with building surfaces; (2) tracking of dirt, dust, pests, and water into buildings from the outdoors; (3) activities that resuspend existing, surface-bound microbes; (4) bodily emissions from exhalation, expectoration, skin shedding, cuts in the skin, and bladder and bowel waste; and (5) surfaces that engender dampness and mold. With advances in microbial field research, the solution sets and priorities for the design community will become more evident.
IMPACTS OF MICROBES ON DEGRADATION OF BUILDING MATERIALS AND ENERGY USAGE
In addition to the important connections between building air, water, and surfaces and human exposures to indoor microorganisms, microorganisms within the built environment can impact building systems and materials in ways that can have associated sustainability, energy usage, and economic effects. For example, the formation of biofilms in water systems can lead to corrosion in pipes and holding tanks (Liu et al., 2016), which carries economic costs when the corrosion requires remediation or the infrastructure must be replaced. Also, biofilms and fungi frequently collect on damp cooling coils (Hugenholtz and Fuerst, 1992; Levetin et al., 2001), which can impair effective heat transfer (Wang et al., 2016). This reduction in heat transfer efficiency means that the cooling coils will consume more energy to accomplish their desired effects, with both economic and sustainability consequences. Microbial decay of building materials, which can include fungal decay of wood, microbial decay of paint, and more general processes known as biodeterioration, can significantly shorten the life of building materials, which also has direct economic and sustain-
ability impacts (Viitanen et al., 2010). Biodeterioration is linked most commonly to moisture in a building and can therefore be managed, but other microbial impacts on building materials and structures are less well understood. Moisture within a building also frequently leads to the growth of molds or fungi that can decay wood and is linked to the failure or rapid deterioration of interior finishes and coatings (EPA, 2013a). Improved frameworks are needed for assessing the effects of microorganisms on and in the built environment and the benefits and costs of potential interventions to manage microbial communities.
BUILDING CODES AND STANDARDS THAT MAY AFFECT THE MICROBIOME
Regulations and Guidance
The regulatory environment for indoor environmental conditions in the United States is defined by several different federal agencies, as well as by state and local authorities. The Occupational Safety and Health Administration has purview over worker health and safety in all workplaces, but it has no specific requirements related to indoor air quality in nonindustrial workplaces such as offices and schools. The General Services Administration, the U.S. Department of Defense, and other federal agencies maintain requirements for the design and operation of their own facilities, some of which are related to indoor environmental conditions. U.S. Department of Housing and Urban Development regulations cover public housing, as well as manufactured homes, some of which again affect the indoor environment. EPA does not have the authority to regulate indoor air quality, although it has the authority to issue reporting, record-keeping, and testing requirements and restrictions related to some chemicals and materials used indoors, such as pesticides and biocides. EPA does, however, produce a wide range of guidance documents20 for homeowners and commercial building designers, owners, and operators on a range of indoor air issues, including radon, asthma, moisture, and exposure to particulate matter.
Many states and local governments have their own health, safety, and environmental quality agencies and building regulations or codes that contain minimum requirements designed to protect occupant health and safety and that are enforced by local building officials as part of the process of obtaining a permit to construct a new building or make significant renovations. These local regulations historically have been focused on structural, electrical, and fire safety issues, although more recently they have also en-
___________________
20 EPA guidance documents on indoor environments are available via links found at https://www.epa.gov/indoor-air-quality-iaq (accessed July 25, 2017).
compassed building energy-efficiency requirements and, to a lesser degree, indoor air quality. These local building regulations are a key mechanism for effecting change in how buildings are designed and built. ASHRAE also offers guidelines—0-2013, The Commissioning Process; 1.1-2007, HVAC&R Technical Requirements for the Commissioning Process—and standards—202-2013, Commissioning Process for Buildings and Systems—that could influence the indoor microbiome through their intent and focus on ensuring that building systems are performing as designed and in a manner that promotes occupant comfort and health.
Sustainable, Green, and Healthy Building Standards and Certifications
A number of sustainable, green, and healthy building standards and certification programs address indoor environmental quality, as well as other aspects of building performance. EPA (2016) defines green or sustainable design as “the practice of creating and using healthier and more resource-efficient models of construction, renovation, operation, maintenance and demolition,” and the agency’s Sustainable Design and Green Building Toolkit for Local Governments offers advice on how to achieve such design within the context of permitting processes (EPA, 2013b). The Office of the Federal Environmental Executive21 defines green building as the practice of (1) increasing the efficiency with which buildings and their sites use energy, water, and materials, and (2) reducing building impacts on human health and the environment through better siting, design, construction, operation, maintenance, and removal—the complete building life cycle (OFEE, 2003).
The relationship of green design standards to both healthy and harmful microbial communities within and near buildings is a subject of significant importance for ongoing research and the identification of opportunities to improve practice based on new research findings. Table 3-5 lists some of the green design standards, guidelines, and certifications used in the United States that are aimed at improving site, energy, water, materials, and indoor environmental quality and address microbial communities.
These standards and guidelines promote building features that reduce adverse exposures to microbes, including increased ventilation rates with better filtration of outside air to remove particulate matter, commissioning and continuous commissioning of building mechanical systems, design for cleanability and quality cleaning practices, walk-off mats for reducing particulate matter and pest intrusion, cooling coil and cooling tower management, and increased access to the outdoors for occupants. These standards
___________________
21 Currently called the Office of Federal Sustainability (https://sustainability.gov/home.html [accessed September 22, 2017]).
| Code, Standard, Guideline, or Certification | Source |
|---|---|
| ANSI/ASHRAE/IES/USGBC Standard 189.1-2014, Standard for the Design of High-Performance Green Buildings | ASHRAE, 2014 |
| Federal Green Construction Guide for Specifiers | EPA, 2010 |
| Fitwel System | HHS, 2017 |
| Green Guide for Healthcare | CMPBS and HCWH, 2007 |
| International Green Construction Code (IgCC) | ICC, 2012 |
| LEED (Leadership in Energy and Environmental Design) | USGBC, 2017 |
| Living Building Challenge Standard | ILFI, 2016 |
| WELL Building Standard | IWBI, 2014 |
NOTE: ANSI = American National Standards Institute; ASHRAE = American Society of Heating, Refrigerating and Air-Conditioning Engineers; IES = Illuminating Engineering Society; USGBC = U.S. Green Building Council.
and guidelines also, in some cases, raise issues related to microbial communities indoors, such as the use of natural materials and indoor landscape features; prohibition of the use of selected cleaning, pesticide, and disinfectant agents; the use of antimicrobials; and rain capture and grey and black water22 systems. However, there are opportunities to improve how some of these standards and guidelines address the building envelope and plumbing or mechanical design as they relate to mold and moisture management. For example, one of the newer efforts, the WELL Building Standard, includes several design “features” that address microbiome-related issues (see Table 3-6). It is essential for professionals in sustainable, green, and healthy design to update these standards regularly in accordance with changes in the knowledge base on good and bad microbial communities and to promote making such voluntary standards mandatory when appropriate.
THE INFLUENCE OF CLIMATE AND CLIMATE CHANGE ON THE BUILT ENVIRONMENT AND MICROBIAL COMMUNITIES
The indoor microbiome depends strongly on climate, and a better understanding of how buildings are designed and used in different climates is essential for improving understanding of the relevant issues. Different
___________________
22 “Grey water” and “black water” are both forms of wastewater generated from human activities. Black water (toilet water, for instance) is presumed to be contaminated with fecal and organic matter that could carry or promote disease, while grey water (drainage water from a sink and the like) is presumed not to contain such contaminants.
TABLE 3-6 WELL Building Standard Features That Address Microbiome-Related Issues
| Section/Feature | No. | Title |
| Air | 06 | Microbe and Mold Control |
| 08 | Healthy Entrance | |
| 09 | Cleaning Protocol | |
| 12 | Moisture Management | |
| 16 | Humidity Control | |
| 27 | Antimicrobial Surfaces | |
| 28 | Cleanable Environment | |
| 29 | Cleaning Equipment | |
| Water | 30 | Fundamental Water Quality |
| 34 | Public Water Additives | |
| 35 | Periodic Water Quality Testing | |
| 36 | Water Treatment | |
| Nourishment | 41 | Hand Washing |
| 42 | Food Contamination |
NOTE: There are also a number of features that address ventilation and air cleaning: 03, 05, 14, 15, 17, 19, 21, 23.
SOURCE: Table created using data from IWBI (2014).
climates have diverse impacts on how water behaves in buildings. In cold winter climates, for example, the cold outdoor air contains relatively little water vapor. When this air infiltrates a heated building, the relative humidity declines, and the indoor air is perceived to be very dry. Although mold growth may be limited by the low moisture content, occupants can experience discomfort from dry skin, eyes, and mucous membranes and rapid evaporation of moisture from the skin. These conditions may lead to the use of humidifiers, which have been shown to be sources of microbial exposure.
Climatic conditions and their changes also have an effect on building ventilation and space conditioning. Outdoor weather conditions are major determinants of envelope infiltration and natural ventilation rates, given their strong dependence on indoor–outdoor temperature differences, as well as on wind speed and direction. In the United States, these conditions often lead to sealing the building windows, eliminating the possibility of natural ventilation and demanding effective design and maintenance of the mechanical system.
Local climate is a major factor in the design and operation of HVAC systems, as their capacity is based on expected heating and cooling loads—-
both the so-called sensible load, which is related to temperature control, and the latent load, which is related to humidity control. Many systems modulate the rate of outdoor air intake as a function of outdoor air temperature and humidity to minimize energy use when a building is being cooled mechanically. As noted in the discussion of interventions in Chapter 5, some systems are designed to increase the outdoor air intake rate to cool the building when the conditions of the outdoor air are conducive to cooling without air tempering by mechanical equipment (air conditioning). Changes in outdoor air temperature and humidity over time impact the ability of the system to maintain desirable indoor air temperatures and to control indoor humidity, and if these changes are large enough, the system may not be able to provide the desired indoor conditions.
Seasonal differences in climatic conditions and occupant behavior also have an effect on the indoor microbiome. The water inside buildings in areas with hot, humid summers and cold, dry winters will behave differently at different times of the year. In these areas, the indoor moisture content of air and materials varies greatly depending on the season. Inside walls, attics, and roofing assemblies, the moisture content of materials is affected by seasonal surface temperatures. In these spaces, cooler internal surfaces collect and retain more moisture than when the same surfaces are warm. In cold climates or at colder times of the year, moisture levels and relative humidity due to condensation on cold surfaces close to the exterior walls tend to increase. In hot and humid climates, moisture inside walls tends to be higher in the summer as a result of infiltration of humid air into the walls from outside the structure if indoor spaces are being cooled mechanically.
It is challenging to design and maintain buildings in climates that feature significant seasonal variation because air (along with the moisture it contains) is influenced by temperature differences among spaces. Thus, attic air can become very hot and humid in the summer, and that air can flow into the occupied zone, carrying with it microbes and their metabolites that may have grown therein. Microbes and their metabolic products in crawl spaces can then enter the living area as a result of the stack effect.
Climate change23 has the potential to affect the indoor environment and thus the indoor microbiome. Outdoor air temperature, humidity, air quality, precipitation, wind direction and velocity, land surface wetness, and catastrophic weather events all can influence the indoor environment, depending on such factors as the integrity of a building’s envelope, the design and conditions of its HVAC systems, the microbial composition of the outdoor ecosystem, and the characteristics of the surrounding buildings. If climatic conditions in a particular area change—for example, if the climate
___________________
23 The text regarding climate change in this section is excerpted or derived from the IOM report Climate Change, the Indoor Environment, and Health (IOM, 2011).
becomes warmer or if there are more severe or more frequent episodes of high heat or intense precipitation—buildings (and other infrastructure) that were designed to operate under the previous conditions may not function well under the new ones. Furthermore, in responding to climate change, people and societies will seek to mitigate undesirable changes and adapt to changes that cannot be mitigated. Some of their responses will play out in how buildings are designed, constructed, used, maintained, and in some cases retrofitted, and the actions taken may well have consequences for indoor environmental quality and public health.
The IOM report Climate Change, the Indoor Environment, and Health (IOM, 2011) addresses this topic in detail. The present committee did not attempt to review the literature in this area as many of details are outside the statement of task for this study (see Box 1-1 in Chapter 1). Instead, it draws on the research and conclusions contained in the 2011 report, which include the following:
There is inadequate evidence to determine whether an association exists between climate-change–induced alterations in the indoor environment and any specific adverse health outcomes. However, available research indicates that climate change may make existing indoor environmental problems worse and introduce new problems by
- Altering the frequency or severity of adverse outdoor conditions that affect the indoor environment.
- Creating outdoor conditions that are more hospitable to pests, infectious agents, and disease vectors that can penetrate the indoor environment.
- Leading to mitigation or adaptation measures and changes in occupant behavior that cause or exacerbate harmful indoor environmental conditions. (p. 241)
Opportunities exist to improve public health while mitigating or adapting to alterations in indoor environmental quality induced by climate change. (IOM, 2011, p. 243)
Supporting literature and additional details may be found in the 2011 report.
SUMMARY OBSERVATIONS AND KNOWLEDGE GAPS
Summary Observations
The composition and viability of indoor microbial communities are dependent on the physical attributes and environmental conditions of the buildings in which they are located. Understanding the conditions in which microbial communities form and are maintained requires consideration of
the variability of building systems, their management, and the surrounding climates.
The building characteristics that affect microbial sources in the air, in water, and on surfaces are an interconnected system that also interacts with occupants. Viewing indoor microbiomes—and by extension, the buildings they inhabit—as part of a dynamic system rather than a collection of individual components will help better define a research agenda that can identify the key drivers of harmful indoor microbial communities. At the most fundamental level, it will be important to recognize that steps taken to address one source of these communities may affect others in ways that are not necessarily easy to anticipate. To take one simple example, limiting outdoor air sources by tightening the building envelope without otherwise providing adequate ventilation could increase indoor moisture and airborne microbial levels, resulting in enhanced microbial growth on damp interior surfaces.
Little is known about what constitutes a “good” indoor microbiome and even less about which building characteristics might foster one. As the literature reviewed in this chapter makes clear, advances have been achieved in the understanding of how building and environmental characteristics influence the presence, abundance, and transmission indoors of microbes known to have adverse health effects. While much remains to be learned, there is an information base on which interventions can be built. Evidence is also starting to emerge for those microbes that either are benign in isolation24 or may have beneficial effects, but this knowledge remains preliminary and mostly speculative. Research addressing good versus bad microbial communities will need to include an examination of the building factors that support each.
Building operations and maintenance are critical contributors to the condition of indoor microbiomes. The roles of building operations and maintenance in determining the health-supporting aspects of the indoor environment often are overlooked. Yet such easily neglected elements as failing to replace air filters regularly can have a large effect. Building operations and maintenance are affected not only by the funds devoted to these activities but also by myriad factors related to the design, age, and use of the building’s enclosure and systems, as well as the actions of the building’s staff and occupants. Tracking the performance of buildings over time compared with design intent, investigating the effectiveness of various commissioning strategies, and examining the payback generated by various maintenance investments is essential when formulating research strategies.
Climatic conditions—which influence such factors as indoor water,
___________________
24 This may have indirect beneficial effects if they displace or hinder microbes that have adverse effects.
relative humidity, and the use of natural ventilation and HVAC systems—strongly affect the survival of bacteria, fungi, and viruses. The building is a mediator of these effects. The literature on the myriad ways in which changes in the outdoor environment affect the conditions inside buildings is summarized and reviewed in the IOM report Climate Change, the Indoor Environment, and Health (IOM, 2011).
In the future, it may be possible to design buildings that sustain healthy microbiomes. While research has not advanced nearly far enough to inform the intentional design of buildings that can maintain a healthy indoor microbiome, the knowledge base needed to accomplish this goal is becoming progressively broader. At this point in time (mid-2017), it is possible to formulate and test healthy microbial designs that include such features as moisture and dirt management, microbial and pest management, and reduced exposure to “bad” microbes found in the air and water and on surfaces. Such research will need to reflect cognizance of how differences in building type, location, use, and occupants influence microbial communities and health outcomes. The information gained by evaluating what does and does not work can then be used to develop design strategies for built environments that sustain healthy microbiomes.
Knowledge Gaps
On the basis of the above summary observations and the information developed in this chapter connecting the built environment and microbial communities, the committee identified the following goals for research to address knowledge gaps and advance the field:
- Improve understanding of how building attributes are associated with microbial communities, and establish a common set of building and environmental data for collection in future research efforts. The building attributes that are associated with various microbial communities need to be gathered for the full range of building types in different climates to foster a better understanding of the differences between how buildings are designed and how they actually function—an important element of determining how to achieve healthy indoor microbiomes. For example, information is lacking on how to interpret test results for water samples from premise plumbing or cooling towers for the purpose of improving building management practices. Such research could be employed to develop guidelines that are test- and climate-specific. There is also a lack of guidance on how to interpret results of microbial air or surface samples. Future research on the indoor microbiome would benefit greatly from the systematic collection of a common set of data on
-
building attributes and indoor environmental conditions so that these factors can be taken into account and examined across studies. This could be achieved by refining the information covered by such existing instruments as EPA’s Building Assessment Survey and Evaluation (BASE) study (EPA, 2017a), the U.S. Centers for Disease Control and Prevention’s MicrobeNet25 survey (CDC, 2017), and the U.S. Department of Energy’s Commercial Buildings Energy Consumption Survey (CBECS) (DOE, 2017a) and Residential Energy Consumption Survey (RECS) (DOE, 2017b), as well as such efforts as MIxS-BE (see Chapter 4). Data on air source–related features, such as HVAC system design information, building ventilation rates, filter efficiencies, and filter replacement practices, are important, as is documentation concerning such water sources and features as premise plumbing material, filtration, water treatment, water use patterns, cooling towers and coils, hot water heater types and temperature settings, building enclosure materials, and moisture damage.
- Collect better information on air, water, and surface microbiome sources and reservoirs in the built environment. The committee’s literature review identified a number of knowledge gaps associated with indoor microbial sources and reservoirs. For air sources, the implications of the shift to more completely sealed buildings for indoor air quality, occupant satisfaction and performance, and indoor microbiomes have yet to be thoroughly researched. For water sources, better means of detecting mold and moisture inside building assemblies, interpreting water activity and moisture measurements in terms of the risk of fungal and bacterial growth, and responding to water damage and subsequent mold growth are needed, as well as more complete understanding of the role of viruses in the evolution of the total indoor microbiome over time. And for surfaces, research is required to determine the relative importance of surfaces occupants touch, sit on, and lie on; surfaces that store and suspend dust and enable resuspension; and surfaces that support dampness and mold.
- Clarify the association of building attributes and conditions with the presence of indoor microorganisms that have beneficial effects. There is a need to understand when the outdoor environment and its microbial communities may have beneficial effects indoors. Examples include examining both whether natural ventilation reduces or
___________________
25 MicrobeNet is an online database containing genetic sequencing, biochemical and morphological characterization, and antibiotic resistance profile information on more than 2,400 rare disease-causing microbes (https://www.cdc.gov/microbenet [accessed May 11, 2017]).
-
increases indoor harmful microbes compared with mechanical ventilation or air conditioning and whether sunlight entering through windows reduces indoor harmful microbes.
- Develop means to better monitor and maintain the built environment, including for concealed spaces, to promote a healthy microbiome. Concealed spaces in buildings play roles in the growth and transport of indoor microorganisms, but they are not easily accessed and thus typically are left unmonitored or unmaintained. Design advances are needed to address monitoring and long-term maintenance in concealed spaces. Maintenance practices in the built environment also will need to avoid investment in unsubstantiated remedies or preventive measures and in interventions that cause, however unintentionally, more problems than they address.
- Deepen knowledge on the impact of climate and climate variations on the indoor environment. Further studies to explore the impact of climate on the survival of bacteria, fungi, and viruses will be useful, especially with the potential of climate change to affect such factors as relative humidity, outdoor microbial communities, the frequency of water penetration into buildings, whether and when windows are opened for ventilation, and how often air conditioning is used. Research on human responses to climate change—from “tight” buildings to the use of biocides, humidifiers, and dehumidifiers—will also need to be pursued to examine the potential for unintended adverse health effects and changes to the microbiome.
REFERENCES
Adams, R. I., S. Bhangar, W. Pasut, E. A. Arens, J. W. Taylor, S. E. Lindow, W. W. Nazaroff, and T. D. Bruns. 2015. Chamber bioaerosol study: Outdoor air and human occupants as sources of indoor airborne microbes. PLOS ONE 10(5):e0128022.
Adams, R. I., S. Bhangar, K. C. Dannemiller, J. A. Eisen, N. Fierer, J. A. Gilbert, J. L. Green, L. C. Marr, S. L. Miller, J. A. Siegel, B. Stephens, M. S. Waring, and K. Bibby. 2016. Ten questions concerning the microbiomes of buildings. Building and Environment 109:224-234.
Addiss, D. G., J. P. Davis, M. LaVenture, P. J. Wand, M. A. Hutchinson, and R. M. McKinney. 1989. Community-acquired Legionnaires’ disease associated with a cooling tower: Evidence for longer-distance transport of Legionella pneumophila. American Journal of Epidemiology 130(3):557-568.
Andersen, B., I. Dosen, A. M. Lewinska, and K. F. Nielsen. 2017. Pre-contamination of new gypsum wallboard with potentially harmful fungal species. Indoor Air 27(1):6-12.
ASHRAE (American Society of Heating, Refrigerating and Air-Conditioning Engineers). 2009. Indoor air quality guide: Best practices for design, construction, and commissioning. Atlanta, GA: ASHRAE.
ASHRAE. 2014. Standard for the design of high-performance green buildings. ANSI/ASHRAE/ USGBC/IES Standard 189.1-2014. https://www.ashrae.org/resources--publications/bookstore/standard-189-1 (accessed April 18, 2017).
ASHRAE. 2016a. ANSI/ASHRAE Standard 62.1: Ventilation for acceptable indoor air quality. Atlanta, GA: ASHRAE.
ASHRAE. 2016b. ANSI/ASHRAE Standard 62.2: Ventilation and acceptable indoor air quality in low-rise residential buildings. Atlanta, GA: ASHRAE.
Banham, R. A. 1984. The architecture of the well-tempered environment. Chicago, IL: University of Chicago Press.
Barberán, A., R. R. Dunn, B. J. Reich, K. Pacifici, E. B. Laber, H. L. Menninger, J. M. Morton, J. B. Henley, J. W. Leff, S. L. Miller, and N. Fierer. 2015. The ecology of microscopic life in household dust. Proceedings of the Royal Society B: Biological Sciences 282(1814):20151139. doi:10.1098/rspb.2015.1139.
Bean, B., B. M. Moore, B. Sterner, L. R. Peterson, D. N. Gerding, and H. H. Balfour, Jr. 1982. Survival of influenza viruses on environmental surfaces. The Journal of Infectious Diseases 146(1):47-51.
Beer, K. D., J. W. Gargano, V. A. Roberts, H. E. Reses, V. R. Hill, L. E. Garrison, P. K. Kutty, E. D. Hilborn, T. J. Wade, K. E. Fullerton, and J. S. Yoder. 2015 Outbreaks associated with environmental and undetermined water exposures—United States, 2011–2012. Morbidity and Mortality Weekly Report 64(31):849-851.
Bennett, D., M. Apte, X. Wu, A. Trout, D. Faulkner, R. Maddalena, and D. Sullivan. 2011. Indoor environmental quality and heating, ventilating, and air conditioning survey of small and medium size commercial buildings: Field study. CEC-500-2011-043. Sacramento, CA: California Energy Commission.
Boone, S. A., and C. P. Gerba. 2007. Significance of fomites in the spread of respiratory and enteric viral disease. Applied and Environmental Microbiology 73(6):1687-1696. http://aem.asm.org/content/73/6/1687.full (accessed May 1, 2017).
Boor, B. E., H. Järnström, A. Novoselac, and Y. Xu. 2014. Infant exposure to emissions of volatile organic compounds from crib mattresses. Environmental Science & Technology 48(6):3541-3549.
Boor, B. E., M. P. Spilak, R. L. Corsi, and A. Novoselac. 2015. Characterizing particle resuspension from mattresses: Chamber study. Indoor Air 25(4):441-456.
Brazeau, R. H., and M. A. Edwards. 2013. Role of hot water system design on factors influential to pathogen regrowth: Temperature, chlorine residual, hydrogen evolution, and sediment. Environmental Engineering Science 30(10):617-627.
Brock, T. D., and K. L. Boylen. 1973. Presence of thermophilic bacteria in laundry and domestic hot-water heaters. Applied Microbiology 25(1):72-76.
Bures, S., J. T. Fishbain, C. F. Uyehara, J. M. Parker, and B. W. Berg. 2000. Computer keyboards and faucet handles as reservoirs of nosocomial pathogens in the intensive care unit. American Journal of Infection Control 28(6):465-471. http://www.sciencedirect.com/science/article/pii/S0196655300906552 (accessed May 1, 2017).
Buttner, M. P., P. Cruz-Perez, L. D. Stetzenbach, P. J. Garrett, and A. E. Luedtke. 2002. Measurement of airborne fungal spore dispersal from three types of flooring materials. Aerobiologia 18(1):1.
CDC (U.S. Centers for Disease Control and Prevention). 2017. MicrobeNet. https://www.cdc.gov/microbenet (accessed March 16, 2017).
Chase, J., J. Fouquier, M. Zare, D. L. Sonderegger, R. Knight, S. T. Kelley, J. Siegel, and J. G. Caporaso. 2016. Geography and location are the primary drivers of office microbiome composition. mSystems 1(2):e00022-16.
Checinska, A., A. J. Probst, P. Vaishampayan, J. R. White, D. Kumar, V. G. Stepanov, G. E. Fox, H. R. Nilsson, D. L. Pierson, J. Perry, and K. Venkateswaran. 2015. Microbiomes of the dust particles collected from the International Space Station and Spacecraft Assembly Facilities. Microbiome 3:50.
Chenari, B., J. D. Carrilho, and M. G. da Silva. 2016. Towards sustainable, energy-efficient and healthy ventilation strategies in buildings: A review. Renewable and Sustainable Energy Reviews 59:1426-1447.
Cho, H., Liu, B., and K. Gowri. 2010. Energy saving impact of ASHRAE 90.1 vestibule requirements: Modeling of air infiltration through door openings. No. PNNL-20026. Richland, WA: Pacific Northwest National Laboratory.
CIBSE (Chartered Institution of Building Services Engineers). 2014. Natural ventilation in non-domestic buildings—CIBSE applications manual AM10. http://www.cibse.org (accessed May 1, 2017).
Clark, R. P. 1974. Skin scales among airborne particles. Journal of Hygiene 72(1):47-51.
CMPBS (Center for Maximum Potential Building Systems) and HCWH (Health Care Without Harm). 2017. Green guide for healthcare, version 2.2. http://www.gghc.org (accessed August 7, 2017).
Collier, S. A., L. J. Stockman, L. A. Hicks, L. E. Garrison, F. J. Zhou, and M. J. Beach. 2012. Direct healthcare costs of selected diseases primarily or partially transmitted by water. Epidemiology and Infection 140(11):2003-2013.
Dedesko, S., and J. A. Siegel. 2015. Moisture parameters and fungal communities associated with gypsum drywall in buildings. Microbiome 3(1):71.
DOE (U.S. Department of Energy). 1999. Building commissioning. The key to quality assurance. https://www.michigan.gov/documents/CIS_EO_commissioningguide_75698_7.pdf (accessed March 18, 2017).
DOE. 2007. Chapter 5. Retrocommissioning. In ENERGY STAR building upgrade manual. https://www.energystar.gov/sites/default/files/buildings/tools/EPA_BUM_CH5_RetroComm.pdf (accessed March 19, 2017).
DOE. 2013. Guide to closing and conditioning ventilated crawlspaces. http://www.nrel.gov/docs/fy13osti/54859.pdf (accessed June 14, 2017).
DOE. 2017a. Commercial Buildings Energy Consumption Survey (CBES). https://www.eia.gov/consumption/commercial (accessed March 18, 2017).
DOE. 2017b. Residential Energy Consumption Survey (RECS). https://www.eia.gov/consumption/residential (accessed March 18, 2017).
Dunn, R. R., N. Fierer, J. B. Henley, J. W. Leff, and H. L. Menninger. 2013. Home life: Factors structuring the bacterial diversity found within and between homes. PLOS ONE 8(5):e64133.
ED (U.S. Department of Education). 2008. National Center for Education Statistics, Schools and Staffing Survey (SASS), “Public School Data File,” 2007–08. “Average number of hours in the school day and average number of days in the school year for public schools, by state: 2007–08.” https://nces.ed.gov/surveys/sass/tables/sass0708_035_s1s.asp (accessed June 7, 2017).
EPA (U.S. Environmental Protection Agency). 2010. Federal green construction guide for specifiers. https://www.wbdg.org/ffc/epa/federal-green-construction-guide-specifiers (accessed August 8, 2017).
EPA. 2013a. Moisture control guidance for building design, construction and maintenance. https://www.epa.gov/sites/production/files/2014-08/documents/moisture-control.pdf (accessed March 16, 2017).
EPA. 2013b. Sustainable design and green building toolkit for local governments. EPA 904B10001. https://archive.epa.gov/greenbuilding/web/pdf/sustainable-design-permitting-toolkit-06_27_13_formatted.pdf (accessed April 18, 2017).
EPA. 2016. Green building. https://archive.epa.gov/greenbuilding/web/html (accessed April 18, 2017).
EPA. 2017a. Building Assessment Survey and Evaluation Study. https://www.epa.gov/indoor-air-quality-iaq/building-assessment-survey-and-evaluation-study (accessed March 16, 2017).
EPA. 2017b. Location of outdoor air intakes and exhaust. In Heating, ventilation and air-conditioning systems, part of indoor air quality design tools for schools. https://www.epa.gov/iaq-schools/heating-ventilation-and-air-conditioning-systems-part-indoor-air-quality-design-tools#Location (accessed June 15, 2017).
Falkinham, J. O. 2011. Nontuberculous mycobacteria from household plumbing of patients with nontuberculous mycobacteria disease. Emerging Infectious Disease 17(3):419-424.
Falkinham, J. O. 2015. Common features of opportunistic premise plumbing pathogens. International Journal of Environmental Research and Public Health 12(5):4533-4545.
Feazel, L. M., L. K. Baumgartner, K. L. Peterson, D. N. Frank, J. K. Harris, and N. R. Pace. 2009. Opportunistic pathogens enriched in showerhead biofilms. Proceedings of the National Academy of Sciences of the United States of America 106(38):16393-16399.
Ferro, A. R., R. J. Kopperud, and L. M. Hildemann. 2004. Source strengths for indoor human activities that resuspend particulate matter. Environmental Science & Technology 38(6):1759-1764.
Fierer, N., C. L. Lauber, N. Zhou, D. McDonald, E. K. Costello, and R. Knight. 2010. Forensic identification using skin bacterial communities. Proceedings of the National Academy of Sciences of the United States of America 107(14):6477-6481.
Finnegan, M. J., C. A. Pickering, and P. S. Burge. 1984. The sick building syndrome: Prevalence studies. British Medical Journal (Clinical Research Edition) 289(6458):1573-1575.
Flores, G. E., S. T. Bates, D. Knights, C. L. Lauber, J. Stombaugh, R. Knight, and N. Fierer. 2011. Microbial biogeography of public restroom surfaces. PLOS ONE 6(11):e28132.
Flores, G. E., S. T. Bates, J. G. Caporaso, C. L. Lauber, J. W. Leff, R. Knight, and N. Fierer. 2013. Diversity, distribution and sources of bacteria in residential kitchens. Environmental Microbiology 15(2):588-596.
Fox, K., E. Castanha, A. Fox, C. Feigley, and D. Salzberg. 2008. Human K10 epithelial keratin is the most abundant protein in airborne dust of both occupied and unoccupied school rooms. Journal of Environmental Monitoring 10(1):55-59.
Ghaitidak, D. M., and K. D. Yadav. 2013. Characteristics and treatment of greywater—A review. Environmental Science and Pollution Research 20(5):2795-2809.
Gibbons, S. M., T. Schwartz, J. Fouquier, M. Mitchell, N. Sangwan, J. A. Gilbert, and S. T. Kelley. 2015. Ecological succession and viability of human-associated microbiota on restroom surfaces. Applied and Environmental Microbiology 81(2):765-773.
Girman, J. R., T. Phillips, and H. Levin. 2009. Critical review: How well do house plants perform as indoor air cleaners? Proceedings: Healthy Buildings 2009 667-671.
Grot, R. A., A. Persily, A. T. Hodgson, and J. M. Daisey. 1989. Environmental evaluation of the Portland East federal office building preoccupancy and early occupancy results. NISTIR 89-4066. Gaithersburg, MD: National Institute of Standards and Technology.
Haleem, A. M., D. M. Hassan, and S. A. Al-Hiyaly. 2013. Comparative assessment of microbial contamination from swabs collected within university facilities. Journal of Health Science 3(2):25-28. http://www.sapub.org/global/showpaperpdf.aspx?doi=10.5923/j.health.20130302.04 (accessed May 1, 2017).
Haverinen U. 2002. Modeling moisture damage observations and their association with health symptoms. Ph.D. dissertation. Kuipio, Finland: National Public Health Institute, Department of Environmental Health.
Heiselberg, P. 2006. Hybrid ventilation in non-residential buildings. In Building ventilation: The state of the art, edited by M. Santamouris and P. Wouters. London, UK: Earthscan Publications, Ltd. Pp. 191-216.
HHS (U.S. Department of Health and Human Services). 2017. The Fitwel System. https://fitwel.org/system (accessed April 18, 2017).
Hoisington, A., J. P. Maestre, K. A. Kinney, and J. A. Siegel. 2016. Characterizing the bacterial communities in retail stores in the United States. Indoor Air 26(6):857-868.
Hospodsky, D., J. Qian, W. W. Nazaroff, N. Yamamoto, K. Bibby, H. Rismani-Yazdi, and J. Peccia. 2012. Human occupancy as a source of indoor airborne bacteria. PLOS ONE 7(4):e34867.
Hospodsky, D., N. Yamamoto, W. W. Nazaroff, D. Miller, S. Gorthala, and J. Peccia. 2015. Characterizing airborne fungal and bacterial concentrations and emission rates in six occupied children’s classrooms. Indoor Air 25(6):641-652.
Hugenholtz, P., and J. A. Fuerst. 1992. Heterotrophic bacteria in an air-handling system. Applied and Environmental Microbiology 58(12):3914-3920.
ICC (International Code Council). 2012. 2012 International green construction code. https://www.iccsafe.org/codes-tech-support/international-green-construction-code-igcc/international-green-construction-code (accessed April 18, 2017).
ILFI (International Living Future Institute). 2016. Living Building Challenge 3.1. Seattle, WA: ILFI. https://living-future.org/lbc (accessed August 7, 2017).
IOM (Institute of Medicine). 1993. Indoor allergens: Assessing and controlling adverse health effects. Washington, DC: National Academy Press.
IOM. 2000. Clearing the air: Asthma and indoor air exposures. Washington, DC: National Academy Press.
IOM. 2004. Damp indoor spaces and health. Washington, DC: The National Academies Press.
IOM. 2011. Climate change, the indoor environment, and health. Washington, DC: The National Academies Press.
IWBI (International WELL Building Institute). 2014. The WELL Building Standard, version 1.0. New York: Delos Living, LLC.
Ji, P., W. J. Rhoads, M. A. Edwards, and A. Pruden. 2017. Impact of water heater temperature setting and water use frequency on the building plumbing microbiome. The ISME Journal 11(6):1318.
Kelley, S. T., and J. A. Gilbert. 2013. Studying the microbiology of the indoor environment. Genome Biology 14(2):202.
Kong, M., T. Q. Dang, J. Zhang, and H. E. Khalifa. 2017. Micro-environmental control for efficient local cooling. Building and Environment 118:300-312.
Koontz, M. D., and H. E. Rector. 1995. Estimation of distributions for residential air exchange rates: Final report. Document No. 600R95180. Washington, DC: EPA.
Kwan, S., R. Shaughnessy, U. Haverinen-Shaughnessy, and J. Peccia. 2016. Exploring the rate of surface microbial redevelopment after cleaning in schools; DNA-sequencing and ATP analysis. Presented at Indoor Air 2016, The 14th International Conference of Indoor Air Quality and Climate, Ghent, Belgium, July 3-8.
Lax, S., D. P. Smith, J. Hampton-Marcell, S. M. Owens, K. M. Handley, N. M. Scott, S. M. Gibbons, P. Larsen, B. D. Shogan, S. Weiss, and J. L. Metcalf. 2014. Longitudinal analysis of microbial interaction between humans and the indoor environment. Science 345(6200):1048-1052.
Levetin, E., R. Shaughnessy, C. A. Rogers, and R. Scheir. 2001. Effectiveness of germicidal UV radiation for reducing fungal contamination within air-handling units. Applied Environmental Microbiology 67(8):3712-3715.
Levin, H. 2010. Natural ventilation: A sustainable solution to infection control in healthcare settings? In Proceedings, ASHRAE IAQ 2010. Atlanta, GA: ASHRAE. http://www.buildingecology.com/articles/natural-ventilation-a-sustainable-solution-to-infection-control-in-healthcare-settings/at_download/file (accessed April 14, 2017).
Li, Y., G. M. Leung, J. W. Tang, X. Yang, C. Y. Chao, J. Z. Lin, J. W. Lu, P. V. Nielsen, J. Niu, H. Qian, and A. C. Sleigh. 2007. Role of ventilation in airborne transmission of infectious agents in the built environment—a multidisciplinary systematic review. Indoor Air 17(1):2-18.
Liu, D. L., and W. W. Nazaroff. 2001. Modeling pollutant penetration across building envelopes. Atmospheric Environment 35(26):4451-4462.
Liu, L., Y. Li, P. V. Nielsen, J. Wei, and R. L. Jensen. 2017. Short-range airborne transmission of expiratory droplets between two people. Indoor Air 27(2):452-462.
Liu, S., C. Gunawan, N. Barraud, S. A. Rice, E. J. Harry, and R. Amal. 2016. Understanding, monitoring, and controlling biofilm growth in drinking water distribution systems. Environmental Science & Technology 50(17):8954-8976.
Los Angeles County Department of Public Health. 2016. Guidelines for alternate water sources: Indoor and outdoor non-potable uses. http://publichealth.lacounty.gov/eh/docs/ep_cross_con_AltWaterSourcesGuideline.pdf (accessed March 20, 2017).
Luoma, M., and S. A. Batterman. 2001. Characterization of particulate emissions from occupant activities in offices. Indoor Air 11(1):35-48. http://www.aivc.org/sites/default/files/airbase_13651.pdf (accessed May 1, 2017).
Lutz, E. A., S. Sharma, B. Casto, G. Needham, and T. J. Buckley. 2010. Effectiveness of UV-C equipped vacuum at reducing culturable surface-bound microorganisms on carpets. Environmental Science & Technology 44(24):9451-9455.
Mayer, T., A. Blachowicz, A. J. Probst, P. Vaishampayan, A. Checinska, T. Swarmer, P. de Leon, and K. Venkateswaran. 2016. Microbial succession in an inflated lunar/Mars analog habitat during a 30-day human occupation. Microbiome 4(1):22.
Meadow, J. F., A. E. Altrichter, S. W. Kembel, J. Kline, G. Mhuireach, M. Moriyama, D. Northcutt, T. K. O’Connor, A. M. Womack, G. Z. Brown, and J. L. Green. 2014a. Indoor airborne bacterial communities are influenced by ventilation, occupancy, and outdoor air source. Indoor Air 24(1):41-48.
Meadow, J. F., A. E. Altrichter, S. W. Kembel, M. Moriyama, T. K. O’Connor, A. M. Womack, G. Z. Brown, J. L. Green, and B. J. Bohannan. 2014b. Bacterial communities on classroom surfaces vary with human contact. Microbiome 2(1):7.
Meadow, J. F., A. E. Altrichter, A. C. Bateman, J. Stenson, G. Z. Brown, J. L. Green, and B. J. Bohannan. 2015. Humans differ in their personal microbial cloud. PeerJ 3:e1258.
MedicineNet.com. 2017. Medical definition of reservoir of infection. http://www.medicinenet.com/script/main/art.asp?articlekey=14969 (accessed June 7, 2017).
Mendell, M. J., and K. Kumagai. 2017. Observation-based metrics for residential dampness and mold with dose-response relationships to health: A review. Indoor Air 27(3):506-517.
Mendell, M. J., Q. Lei-Gomez, A. G. Mirer, O. Seppänen, and G. Brunner. 2008. Risk factors in heating, ventilating, and air-conditioning systems for occupant symptoms in U.S. office buildings: The U.S. EPA BASE study. Indoor Air 18(4):301-316.
Mensah-Attipoe, J., T. Reponen, A. Salmela, A. M. Veijalainen, and P. Pasanen. 2015. Susceptibility of green and conventional building materials to microbial growth. Indoor Air 25(3):273-284.
Menzies, D., J. Popa, J. A. Hanley, T. Rand, and D. K. Milton. 2003. Effect of ultraviolet germicidal lights installed in office ventilation systems on workers’ health and wellbeing: Double-blind multiple crossover trial. The Lancet 362(9398):1785-1791.
Morawska, L. 2006. Droplet fate in indoor environments, or can we prevent the spread of infection? Indoor Air 16(5):335-347.
Morey, P. R., T. Rand, and T. Phoenix. 2009. On the penetration of mold into the fiberboard used in HVAC ductwork. Presented at Healthy Buildings 2009—9th International Conference and Exhibition, Syracuse, NY.
Morrison, G. 2015. Recent advances in indoor chemistry. Current Sustainable/Renewable Energy Reports 2(2):33-40.
Nazaroff, W. W. 2016. Indoor bioaerosol dynamics. Indoor Air 26(1):61-78.
Nazaroff, W. W., and A. H. Goldstein. 2015. Indoor chemistry: Research opportunities and challenges. Indoor Air 25(4):357-361.
Ng, L., A. Persily, and S. Emmerich. 2015. Infiltration and ventilation in a very tight, high performance home. Presented at 36th AIVC Conference Effective Ventilation in High Performance Buildings, Madrid, Spain, Air Infiltration and Ventilation Centre, Madrid, Spain.
NIST (National Institute of Standards and Technology). 2017. LoopDA. https://www.nist.gov/services-resources/software/loopda (accessed March 20, 2017).
Noble, W. C., J. D. Habbema, R. Van Furth, I. Smith, and C. A. De Raay. 1976. Quantitative studies on the dispersal of skin bacteria into the air. Journal of Medical Microbiology 9(1):53-61.
NRC (National Research Council). 2006. Green schools: Attributes for health and learning. Washington, DC: The National Academies Press.
OFEE (Office of Federal Environmental Executive). 2003. The federal commitment to green building: Experiences and expectations. https://archive.epa.gov/greenbuilding/web/pdf/fedcomm_greenbuild.pdf (accessed April 18, 2017).
Offermann, F. J. 2009. Ventilation and indoor air quality in new homes. CEC-500-2009-085. https://www.arb.ca.gov/research/apr/past/04-310.pdf (accessed July 17, 2017).
Pagnier, I., M. Merchat, and B. La Scola. 2009. Potentially pathogenic amoeba-associated microorganisms in cooling towers and their control. Future Microbiology 4(5):615-629.
Perkins+Will. 2017. Healthy environments: Understanding antimicrobial ingredients in building materials. http://perkinswill.com/sites/default/files/Antimicrobial_WhitePaper_PerkinsWill.pdf (accessed June 19, 2017).
Persily, A. K. 2016. Field measurement of ventilation rates. Indoor Air 26(1):97-111.
Persily, A., and J. Gorfain. 2008. Analysis of ventilation data from the U.S. Environmental Protection Agency Building Assessment Survey and Evaluation (BASE) Study. NISTIR 7145-Revised. http://ws680.nist.gov/publication/get_pdf.cfm?pub_id=916664 (accessed May 1, 2017).
Persily, A., and H. Levin. 2011. Ventilation measurements in IAQ studies: Problems and opportunities. In Proceedings of Indoor Air 2011, 12th International Conference on Indoor Air Quality and Climate. http://ws680.nist.gov/publication/get_pdf.cfm?pub_id=907718 (accessed September 22, 2017).
Persily, A., J. Gorfain, and G. Brunner. 2005. Ventilation rates in U.S. office buildings from the EPA Base Study. Indoor Air 15(11):917-922.
Qian, J., and A. R. Ferro. 2008. Resuspension of dust particles in a chamber and associated environmental factors. Aerosol Science and Technology 42(7):566-578.
Qian, J., D. Hospodsky, N. Yamamoto, W. W. Nazaroff, and J. Peccia. 2012. Size-resolved emission rates of airborne bacteria and fungi in an occupied classroom. Indoor Air 22(4):339-351.
Qian, J., J. Peccia, and A. R. Ferro. 2014. Walking-induced particle resuspension in indoor environments. Atmospheric Environment 89:464-481.
Quimby, S. C. 2016. Concealed spaces. Insurance Advocate, October 17. p. 13. http://www.msonet.com/wp-content/uploads/2016/12/Concealed-Spaces.pdf (accessed July 14, 2017).
Rhoads, W. J. 2017. Growth of opportunistic pathogens in domestic plumbing: Building standards, system operation, and design. Ph.D. dissertation. Blacksburg, VA: Virginia Tech. https://vtechworks.lib.vt.edu/bitstream/handle/10919/76653/Rhoads_WJ_D_2017.pdf (accessed May 1, 2017).
Rhoads, W. J., P. Ji, A. Pruden, and M. A. Edwards. 2015. Water heater temperature set point and water use patterns influence Legionella pneumophila and associated microorganisms at the tap. Microbiome 3(1):67. https://microbiomejournal.biomedcentral.com/articles/10.1186/s40168-015-0134-1 (accessed May 1, 2017).
Roberts, M. C., and D. B. No. 2014. Environment surface sampling in 33 Washington State fire stations for methicillin-resistant and methicillin-susceptible Staphylococcus aureus. American Journal of Infection Control 42(6):591-596.
Scott, E., S. Duty, and K. McCue. 2009. A critical evaluation of methicillin-resistant Staphylococcus aureus and other bacteria of medical interest on commonly touched household surfaces in relation to household demographics. American Journal of Infection Control 37(6):447-453.
Seppänen, O. A., and W. J. Fisk. 2004. Summary of human responses to ventilation. Indoor Air 14(S7):102-118.
Simcox, N. J., J. Camp, and M. C. Roberts. 2012. Environmental surface sampling for MRSA in Washington State fire stations. Final report. http://deohs.washington.edu/sites/default/files/images/MRSA_report_8-16-12.pdf (accessed March 16, 2017).
Smith, D. P., J. C. Alverdy, J. A. Siegel, and J. A. Gilbert. 2013. Design considerations for home and hospital microbiome studies. In The science and applications of microbial genomics: Workshop summary. Washington, DC: The National Academies Press.
Sordillo, J. E., U. K. Alwis, E. Hoffman, D. R. Gold, and D. K. Milton. 2013. Bacterial and fungal microbial biomarkers in house dust. In Environmental health: Indoor exposures, assessments and interventions. Oakville, ON: Apple Academic Press. Pp. 63-86.
Stapleton, H. M., S. Klosterhaus, A. Keller, P. L. Ferguson, S. van Bergen, E. Cooper, T. F. Webster, and A. Blum. 2011. Identification of flame retardants in polyurethane foam collected from baby products. Environmental Science & Technology 45(12):5323–5331.
Straub, J. 2006. Building Science Digests. BSD-138: Moisture and materials. https://buildingscience.com/documents/digests/bsd-138-moisture-and-materials (accessed March 5, 2017).
Sundell, J., H. Levin, W. W. Nazaroff, W. S. Cain, W. J. Fisk, D. T. Grimsrud, F. Gyntelberg, Y. Li, A. K. Persily, A. C. Pickering, J. M. Samet, J. D. Spengler, S. T. Taylor, and C. J. Weschler. 2011. Ventilation rates and health: Multidisciplinary review of the scientific literature. Indoor Air 21(3):191-204.
Täubel, M. 2016. Of house dust and a crawling baby robot—indoor microbial exposure assessment. Presented at the 5th Conference—Microbiology of the Built Environment, Boulder, CO, June 3.
Täubel, M., H. Rintala, M. Pitkäranta, L. Paulin, S. Laitinen, J. Pekkanen, A. Hyvärinen, and A. Nevalainen. 2009. The occupant as a source of house dust bacteria. Journal of Allergy and Clinical Immunology 124(4):834-840.
TEES (Texas A&M Experimental Energy Station). 2017. Continuous commissioning. College Station, TX: Energy Systems Laboratory. http://esl.tamu.edu/CC (accessed March 19, 2017).
Thatcher, T. L., and D. W. Layton. 1995. Deposition, resuspension, and penetration of particles within a residence. Atmospheric Environment 29(13):1487-1497.
Torvinen, E., S. Suomalainen, L. Paulin, and J. Kusnetsov. 2014. Mycobacteria in Finnish cooling tower waters. APMIS 122(4):353-358.
Toze, S. 2006. Water reuse and health risks—real vs. perceived. Desalination 187(1):41-51.
TRB (Transportation Research Board). 2015. Optimizing airport building operations and maintenance through retrocommissioning: A whole-systems approach. Washington, DC: The National Academies Press.
USGBC (U.S. Green Building Council). 2017. LEED v4 for building design and construction. http://www.usgbc.org/resources/leed-v4-building-design-and-construction-current-version (accessed April 18, 2017).
Veillette, M., L. D. Knibbs, A. Pelletier, R. Charlebois, P. B. Lecours, C. He, L. Morawska, and C. Duchaine. 2013. Microbial contents of vacuum cleaner bag dust and emitted bioaerosols and their implications for human exposure indoors. Applied and Environmental Microbiology 79(20):6331-6336.
Venkateswaran, K. 2016. Environmental “omics” of ISS. Presentation to the Committee on Microbiomes of the Built Environment: From Research to Application, Irvine, CA, October 17.
Verdier, T., M. Coutand, A. Bertron, and C. Roques. 2014. A review of indoor microbial growth across building materials and sampling and analysis methods. Building and Environment 80:136-149.
Viitanen, H., J. Vinha, K. Salminen, T. Ojanen, R. Peuhkuri, L. Paajanen, and K. Lähdesmäki. 2010. Moisture and bio-deterioration risk of build materials and structures. Journal of Building Physics 33(3):201-224.
Wang, Y., C. Sekhar, W. P. Bahnfleth, K. W. Cheong, and J. Firantello. 2016. Effectiveness of an ultraviolet germicidal irradiation system in enhancing cooling coil energy performance in a hot and humid climate. Energy and Buildings 130:321-329.
Waring, M. S. 2016. Bio-walls and indoor houseplants: Facts and fictions. Presentation before the Committee on Microbiomes of the Built Environment: From Research to Application, October 17. http://nas-sites.org/builtmicrobiome/files/2016/07/Michael-Waring-FOR-POSTING.pdf (accessed March 20, 2017).
Weinstein, R. A., C. B. Bridges, M. J. Kuehnert, and C. B. Hall. 2003. Transmission of influenza: Implications for control in health care settings. Clinical Infectious Diseases 37(8):1094-1101. https://academic.oup.com/cid/article/37/8/1094/2013282/Transmission-of-Influenza-Implications-for-Control (accessed May 1, 2017).
Weiss, D., C. Boyd, J. L. Rakeman, S. K. Greene, R. Fitzhenry, T. McProud, K. Musser, L. Huang, J. Kornblum, E. J. Nazarian, A. D. Fine, S. L. Braunstein, D. Kass, K. Landman, P. Lapierre, S. Hughes, A. Tran, J. Taylor, D. Baker, L. Jones, L. Kornstein, B. Liu, R. Perez, D. E. Lucero, E. Peterson, I. Benowitz, K. F. Lee, S. Ngai, M. Stripling, and J. K. Varma. 2017. A large community outbreak of Legionnaires’ disease associated with a cooling tower in New York City, 2015. Public Health Reports 132(2):241-250.
Weschler, C. J. 2009. Changes in indoor pollutants since the 1950s. Atmospheric Environment 43:153-169.
Weschler, C. J. 2011. Chemistry in indoor environments: 20 years of research. Indoor Air 21(3):205-218.
Weschler, C. J. 2016. Roles of the human occupant in indoor chemistry. Indoor Air 26(1):6-24.
West, M., and E. Hansen. 1989. Determination of material hygroscopic properties that affect indoor air quality. In Proceedings of the IAQ 89, the human equation: Health and comfort. Atlanta, GA: ASHRAE.
Xu, L.-C., and C. A. Siedlecki. 2012. Submicron-textured biomaterial surface reduces staphylococcal bacterial adhesion and biofilm formation. Acta Biomaterials 8(1):72-81.
Yamamoto, N., D. Hospodsky, K. C. Dannemiller, W. W. Nazaroff, and J. Peccia. 2015. Indoor emissions as a primary source of airborne allergenic fungal particles in classrooms. Environmental Science & Technology 49(8):5098-5106.
Zaatari, M., E. Nirlo, D. Jareemit, N. Crain, J. Srebric, and J. Siegel. 2014. Ventilation and indoor air quality in retail stores: A critical review (RP-1596). HVAC&R Research 20(2):276-294.

























































