1
Introduction
Over the past 20 years, significant changes have taken place in the policy landscape surrounding cannabis legalization, production, and use. To date, 28 states and the District of Columbia have legalized cannabis for the treatment of medical conditions (NCSL, 2016). Eight of these states and the District of Columbia have also legalized cannabis for recreational use. These landmark changes in policy have markedly changed cannabis use patterns and perceived levels of risk. Based on a recent nationwide survey, 22.2 million Americans (12 years of age and older) reported using cannabis in the past 30 days, and between 2002 and 2015 the percentage of past month cannabis users in this age range has steadily increased (CBHSQ, 2016).
Despite this reported rapid rise in the use of cannabis, both for medical purposes and for recreational use, conclusive evidence regarding the short- and long-term health effects of cannabis use remains elusive. While a myriad of studies have examined cannabis use in all its various forms (Calabria et al., 2010; Whiting et al., 2015, 2016; WHO, 2016), often these research conclusions are not appropriately synthesized, translated for, or communicated to policy makers, health care providers, state health officials, or other stakeholders who have been charged with influencing and enacting policies, procedures, and laws related to cannabis use. Unlike other substances whose use may confer risk, such as alcohol or tobacco, no accepted standards for the safe use or appropriate doses are available to help guide individuals as they make choices regarding the issues of if, when, where, and how to use cannabis safely and, in regard to therapeu-
tic uses, effectively (Freeman et al., 2014; Marsot et al., 2016). Moreover, studying the potential health impacts of cannabis presents its own set of unique challenges. Current challenges include the existence of certain regulations and policies that restrict access to cannabis products suited for research purposes (e.g., Schedule 1 status; regulatory approvals), the limited availability of funding for comprehensive cannabis research, and crosscutting methodological challenges. Additionally, researchers are often unable to obtain the necessary quantity, quality, or type of cannabis product to address cutting-edge public health research questions.
STUDY CHARGE
Shifting public sentiment, conflicting and impeded scientific research, and legislative battles have fueled the debate about what, if any, harms or benefits can be attributed to the use of cannabis or its derivatives. In March 2016 the Health and Medicine Division1 of the National Academies of Sciences, Engineering, and Medicine (the National Academies) was asked to convene a committee of experts to conduct a comprehensive review of literature regarding the health consequences of using cannabis or its constituents that had appeared since the publication of the 1999 Institute of Medicine (IOM) report Marijuana and Medicine (IOM, 1999). In addition, the committee was asked to make recommendations for a research agenda that will identify the most critical research questions regarding the association of cannabis use with health outcomes (both harms and benefits) that can be answered in the short term (i.e., within a 3-year time frame), as well as steps that should be taken in the short term to ensure that sufficient data are being gathered to answer long-term questions. Of note, throughout this report the committee has attempted to highlight research conclusions that affect certain populations (e.g., pregnant women, adolescents) that may be at greater risk for potential adverse effects of cannabis use. The committee’s full statement of task is presented in Box 1-1.
The resulting Committee on the Health Effects of Marijuana included experts in the areas of marijuana, addiction, oncology, cardiology, neurodevelopment, pulmonary, pediatric and adolescent health, immunology, toxicology, preclinical research, epidemiology, systematic review, and public health. (See Appendix E for the biographical sketches of committee members.)
In conducting its work, the committee met six times from March 2016 through December 2016. In conjunction with two of those meet-
___________________
1 As of March 2016, the Health and Medicine Division continues the consensus studies and convening activities previously carried out by the Institute of Medicine (IOM).
ings, the committee held half-day public information-gathering sessions which allowed the committee to hear from study sponsors, experts, and other stakeholders. These discussions helped to inform the committee’s deliberations.
Sponsors of this report include federal, state, philanthropic, and nongovernmental organizations. These include the Alaska Mental Health Trust Authority; Arizona Department of Health Services; California Department of Public Health; CDC Foundation; Centers for Disease Control and Prevention (CDC); The Colorado Health Foundation; Mat-Su Health Foundation; National Highway Traffic Safety Administration;
National Institutes of Health/National Cancer Institute; National Institutes of Health/National Institute on Drug Abuse; Oregon Health Authority; the Robert W. Woodruff Foundation; Truth Initiative; U.S. Food and Drug Administration; and the Washington State Department of Health.
STUDY CONTEXT AND APPROACH
Over the past 20 years the IOM has published several consensus reports that focused on the health effects of marijuana or addressed marijuana within the context of other drug or substance abuse topics.2 Two IOM reports that most prominently informed the committee’s work were Marijuana and Health (IOM, 1982), and the 1999 report Marijuana and Medicine: Assessing the Science Base (IOM, 1999). Although these reports differed in scope, they were useful in providing a comprehensive body of evidence upon which the current committee could build.
Marijuana and Health (IOM, 1982) was commissioned by the former Secretary of Health and Human Services and the former director of the National Institutes of Health, Joseph Califano, Jr., and Donald S. Fredrickson, respectively. The study’s committee was appointed to (1) analyze the potential hazards of marijuana use on user safety and health, (2) analyze data concerning the therapeutic value of marijuana, (3) assess the federal research programs, (4) identify new research directions, and (5) draw conclusions that would assist future policy decision making. The authoring committee concluded that there was evidence indicating that marijuana has a broad range of psychological and biological effects, some of which under certain conditions are harmful to human health, but there was a substantial lack of definitive evidence to characterize the seriousness of harm. The committee’s major conclusion was that “what little we know for certain about the effects of marijuana on human health—and all that we have reason to suspect—justifies serious national concern” (IOM, 1982, p. 5). The committee’s major recommendation called for an intensification and more comprehensive research effort into the effects of marijuana on the health of the American people.
In 1997 the White House Office of National Drug Control Policy contracted with the IOM to conduct a scientific review of available literature to determine the potential health benefits and risks of marijuana and its constituent cannabinoids. The resulting report, Marijuana and Medicine (IOM, 1999), offered several conclusions and recommendations (see Box 1-2) on the effects of isolated cannabinoids, the efficacy of cannabinoid drugs, the influence of psychological effects on therapeutic effects,
___________________
2 See https://www.nap.edu/search/?year=1995&rpp=20&ft=1&term=marijuana (accessed July 2016).
physiological risks, marijuana dependence and withdrawal, marijuana as a “gateway drug,” and the use of smoked marijuana.
The scientific literature on cannabis use has grown substantially since the publication of Marijuana and Medicine in 1999. The current committee conducted an extensive search of relevant databases, including Medline, Embase, the Cochrane Database of Systematic Reviews, and PsycINFO,
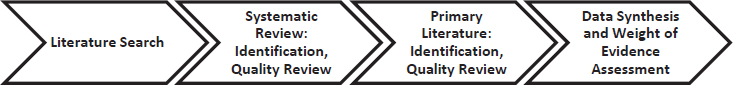
and they initially retrieved more than 24,000 abstracts for articles published since the 1999 report that could potentially be relevant to this study. These abstracts were reduced by limiting articles to those published in English and removing case reports, editorials, studies by “anonymous” authors, conference abstracts, and commentaries. In the end, the committee considered more than 10,700 abstracts for their relevance to this report. (See Appendix B for details.)
The methodological approach taken by the committee to conduct this comprehensive literature review and meet the objectives outlined in the Statement of Task is detailed in Appendix B and briefly described here. Given the large scientific literature on cannabis, the breadth of the statement of task, and the time constraints of the study, the committee developed an approach that resulted in giving primacy to recently published systematic reviews (since 2011) and high-quality primary research that studied 1 or more of 11 groups of health endpoints (see Figure 1-1 and Box 1-3). For each health endpoint, systematic reviews were identified and assessed for quality using methods adapted from published criteria (Whiting et al., 2016); only reviews that were assessed by the committee as being of good or fair quality were considered in this comprehensive review. The committee’s conclusions are based on the findings from the most recently published systematic review and all relevant primary literature that was determined to be fair and good quality that was published after the most recent systematic review. Where no systematic review existed, the committee reviewed all relevant primary research from January 1, 1999, through August 1, 2016. Primary research was evaluated using global assessments of the quality of available studies guided by standard approaches and methodologies (Cochrane Quality Assessment [Higgins et al., 2011], Newcastle–Ontario scale [Wells et al., 2014]). Any deviations from this approach are noted in the relevant chapters. For a comprehensive description of the committee’s approach to evaluating the available literature, please refer to Appendix B.
Informed by the reports of previous IOM committees,3 the committee developed standard language to categorize the weight of evidence regarding whether cannabis or cannabinoid use (for therapeutic purposes) is an effective or ineffective treatment for the prioritized health endpoints of interest, or whether cannabis or cannabinoid use (primarily for recreational purposes) is statistically associated with the prioritized health endpoints of interest. Box 1-4 describes these categories and the general parameters for the types of evidence supporting each category. The committee used these weight-of-evidence categories in their conclusions.
___________________
3Adverse Effects of Vaccines: Evidence and Causality (IOM, 2012); Treatment of Posttraumatic Stress Disorder: An Assessment of the Evidence (IOM, 2008); Veterans and Agent Orange: Update 2014 (NASEM, 2016).
The search strategies and processes described above were developed and adopted by the committee in order to adequately address a broad statement of task in a limited time frame while adhering to the National Academies high standards for the quality and rigor of committee reports.
First, the committee was not tasked with conducting multiple systematic reviews, which would have implied a lengthy and robust series of processes. The committee adopted key features of that process; however, a comprehensive literature search; assessments by more than one person of the quality (risk of bias) of key literature and the conclusions; prespecification of the questions of interest before conclusions were formulated; standard language to allow comparisons between conclusions; and
declarations of conflict of interest via the National Academies’ conflict-of-interest policies.
Second, there is a possibility that some literature was missed because of the practical steps taken to narrow a very large literature to one that was manageable within the time frame available to the committee. Furthermore, some very good research may not have been reviewed in this report because it did not directly address the specific health endpoint questions formulated by the committee.
Special Considerations for the Report
Biological Plausibility
After careful consideration, the committee chose not to attempt to review basic, nonhuman research in order to attempt to bolster evidence for identified health outcomes from cannabis exposure. This policy was, in part, dictated by the time constraints available for crafting this report. Also, while basic research is in the end critical for understanding health outcome mechanisms and suggesting new and innovative interventions,
it often cannot explain the large number of null findings, the frequent variation among human study outcomes, the adverse clinical effects seen in some studies, nor the diversity in host susceptibility to cannabis exposure. Given the methodologic variation in the studies reviewed, as well as potential deficiencies in study design and execution, the committee focused its attention and energy on identifying high-quality studies with the best information and lowest risk of bias as the way to ensure that report findings and conclusions were as informative and relevant as possible. In those instances where cannabis-disease associations seemed relatively secure and evidence-based, the committee believed that the findings would have clinical and public health importance even in the absence of supporting basic studies. Similarly, for those experimental studies where causation could be more explicitly determined—mostly in the area of therapeutics—these findings, if sufficiently robust and replicable, were deemed to stand on their own whether or not bolstered with mechanistic or biologically plausible underpinnings.
Considerations of Observational Studies
The vast majority of the systematic reviews, meta-analyses, and primary literature reviewed in Part III: Other Health Effects consists of observational studies. This is in contrast to the literature base in other fields such as therapeutics (discussed in Part II: Therapeutic Effects). As such, it was not possible to restrict the literature reviews to those that synthesized evidence from randomized clinical trials (RCTs). The methodology used for systematic reviews and meta-analysis originates in the synthesis of data from RCTs, where methodology is highly standardized and structured. The synthesis of observational studies presents some challenges that have not been fully met, arising, in part, out of the greater variety in study design.
Exposure measurement is always an additional concern when evaluating comprehensive reviews of observational studies. Assessment of cannabis exposure is particularly challenging because of its illegal status (in most settings) and the reliance on self-report. Inherent difficulties in accurately assessing the exposure in terms of dose, specific type of cannabis product used, mode of intake, duration, frequency, and other variables result in the variability in definitions used to operationalize cannabis exposure. Additionally, observational studies often have to contend with confounders related to polysubstance use, which obscures the ability to answer questions about the effects of “cannabis only” on the health effects. Moreover, in some cases, samples included different populations (i.e., adolescents versus adults), cannabis-use history (i.e., chronic versus
acute), and patterns of use (i.e., frequency, dose, quantity)—all of which provide mixed or inconsistent evidence as to the effects of cannabis on a specific outcome. Additional limitations include a lack of longitudinal assessments and small study cohorts.
There is also a concern about the broad reporting standards across cannabis research fields. For example, several systematic reviews on cognition discussed in the report’s Psychosocial chapter did not consistently describe the methods for scoring the evidence for each endpoint. That is, the reviews include scores of the strength and consistency of the evidence for each outcome, but they provided less information about issues such as study design and statistical analyses. As a result, the committee found that the reviews did not include the conventional data generally found within quantitatively-based systematic examinations of a topic, or such as would be found in meta-analytic reviews. Reasons for this may include variations in study methodologies, instrumentation, populations, or research designs.
Despite these special considerations regarding the use of systematic reviews, meta-analyses, and primary literature of observational studies, the committee determined that using recent good- or fair-quality systematic reviews was the most appropriate approach to adequately address the committee’s broad statement of task and comprehensive, prioritized research questions while maintaining a high standard for quality and rigor. For additional information on these considerations, please see Chapter 11 (Psychosocial) and Box 12-2 in Chapter 12 (Mental Health).
Comparing Harms and Benefits of Cannabis Use
Several health endpoints are discussed in multiple chapters of the report (e.g., cancer, schizophrenia); however, it is important to note that the research conclusions regarding potential harms and benefits discussed in these chapters may differ. This is, in part, due to differences in the study design of the reviewed evidence, differences in characteristics of cannabis or cannabinoid exposure (e.g., form, dose, frequency of use), and the populations studied. As such, it is important that the reader is aware that this report was not designed to reconcile the proposed harms and benefits of cannabis or cannabinoid use across the report’s chapters. In drafting the report’s conclusions, the committee made an effort to be as specific as possible about the type and/or duration of cannabis or cannabinoid exposure and, where relevant, cross-referenced findings from other report chapters.
Key Definitions
The terms “marijuana” and “cannabis” are often used interchangeably, particularly within the United States; however, these are two separate entities. Cannabis is a broad term that can be used to describe organic products (e.g., cannabinoids,4 marijuana,5 hemp6) derived from the Cannabis sativa plant. These products exist in various forms and are used for a number of different purposes (e.g., medical, industrial, recreational). Given its broad potential, the all-encompassing word “cannabis” has been adopted as the standard terminology within scientific and scholarly communities. The committee uses the term “cannabis” rather than “marijuana” throughout this report.
The committee notes the existence of “cannabimimetic agents” (often referred to as “K2” or “Spice”) which are made up of dried plant matter sprayed with synthetic chemicals that mimic the effect of THC by interacting with cannabinoid receptors in the brain (King, 2014). At the request of the study sponsors, nontherapeutic synthetic cannabinoids are not considered in this study.
REPORT ORGANIZATION
This report is organized into four parts and 16 chapters. Part I: Introduction and Background (Chapters 1–3) provides an overview of the origin, purpose, and organization of the report, as well as essential information on cannabis and cannabis-derived medications and products, and the history and current state of federal and state cannabis policy. In addition to this Introduction (Chapter 1), Chapter 2 (Cannabis) reviews the biology of cannabis and its constituent compounds, exploring the biochemistry of the marijuana plant, its derivatives, and the different routes of administration. Additionally, this chapter provides an overview of synthetic versions of cannabis, including U.S. Food and Drug Administration–approved medicinal synthetics and manufactured cannabis (street drugs such as K2, Spice). Chapter 3 (Cannabis: Prevalence of Use, Regulation, and Current Policy Landscape) provides an overview of cannabis use in the United States and reviews policy related to cannabis legislation.
___________________
4 Cannabinoids are a group of active chemical compounds found in cannabis. Among the more than 100 different types of cannabinoids are tetrahydrocannabinol (THC) and cannabidiol (CBD) (Small, 2015).
5 In general, marijuana refers only to parts of the plant or derivative products that contain substantial levels of tetrahydrocannabinol (THC), the chemical compound that is found in the highest concentrations in the cannabis plant and which is primarily responsible for the plant’s intoxicative qualities (Small, 2015).
6 Under U.S. law, cannabis plants with very low levels of THC (not more than 0.3 percent) are not considered marijuana but instead “industrial hemp” (Small, 2015).
Part II: Therapeutic Effects (Chapter 4—Therapeutic Effects of Cannabis and Cannabinoids) discusses the health effects of cannabis and cannabinoids used for therapeutic purposes in relation to the most commonly reported conditions for medical cannabis use (in states where usage is legal), as well as the current qualifying ailments recognized by state medical marijuana programs. Most of the evidence reviewed in this chapter derives from clinical and basic science research conducted for the specific purpose of answering an a priori question of whether cannabis and/or cannabinoids are an effective treatment for a specific disease or health condition. The vast majority of these studies examined the potential therapeutic effect of cannabinoids (e.g., FDA-approved synthetics) rather than smoked cannabis.
Part III: Other Health Effects (Chapters 5–14) discusses the health effects of cannabis and/or cannabis-derived products used for primarily recreational and other nontherapeutic purposes. Most of the evidence reviewed in Part III derives from epidemiological research primarily focusing on smoked cannabis. It is of note that several of the prioritized health conditions discussed in Part III are also reviewed in Part II, albeit from the perspective of effects associated with using cannabis for primarily recreational, as opposed to therapeutic, purposes. A bulleted list of chapter highlights are included in the introduction of the chapters in Part II and Part III of the report.
Within Part III, the effects of cannabis use on cancer incidence are discussed in Chapter 5. Chapter 6 addresses cardiometabolic risks of cannabis use, including effects on acute myocardial infarction, stroke, and metabolic effects—metabolic dysregulation, metabolic syndrome, prediabetes, and diabetes mellitus. Respiratory disease—pulmonary function, chronic obstructive pulmonary disease, respiratory symptoms including chronic bronchitis, and asthma—are discussed in Chapter 7. Immunity and infection are discussed in Chapter 8. The effects of cannabis use on overall mortality, overdose death, employment injuries, and motor vehicle crashes are reviewed in Chapter 9 (Injury and Death). Prenatal, perinatal, and neonatal effects are discussed in Chapter 10. Psychosocial effects, including the effects of cannabis on learning, memory, attention, academic achievement, employment and income, and social relationships and social roles are discussed in Chapter 11, and mental health conditions, including schizophrenia and other psychosis, bipolar disorder, depression, suicide, anxiety, and posttraumatic stress disorder are discussed in Chapter 12. Chapter 13 discusses problem cannabis use, including cannabis use disorder, and the abuse of other substances is discussed in Chapter 14.
Part IV: Research Barriers and Recommendations (Chapters 15–16) reviews the regulatory barriers and methodological challenges that hinder cannabis research, and recommends the actions necessary to successfully
implement a comprehensive cannabis research agenda. Chapter 15 provides an overview of barriers to studying cannabis, including regulatory, policy, and financial, as well as of methodological challenges, and Chapter 16 outlines the committee’s proposed research agenda, detailing both short-term and long-term objectives.
Appendixes A–E contain the report glossary, details about the committee’s search strategy, systematic reviews considered in this report, open session agendas, and biographical sketches of committee and staff members.
REFERENCES
Calabria, B., L. Degenhardt, W. Hall, and M. Lynskey. 2010. Does cannabis use increase the risk of death? Systematic review of epidemiological evidence on adverse effects of cannabis use. Drug and Alcohol Review 29(3):318–330.
CBHSQ (Center for Behavioral Health Statistics and Quality). 2016. Behavioral health trends in the United States: Results from the 2014 National Survey on Drug Use and Health. https://www.samhsa.gov/data/sites/default/files/NSDUH-FFR1-2015/NSDUH-FFR1-2015/NSDUH-FFR1-2015.pdf (accessed January 3, 2017).
Freeman, T. P., C. J. Morgan, C. Hindocha, G. Schafer, R. K. Das, and H. V. Curran. 2014. Just say “know”: How do cannabinoid concentrations influence users’ estimates of cannabis potency and the amount they roll in joints? Addiction 109(10):1686–1694.
Higgins, J. P. T., D. G. Altman, P. C. Gøtzsche, P. Jüni, D. Moher, A. D. Oxman, J. Savović, K. F. Schulz, L. Weeks, J. A. C. Sterne, Cochrane Bias Methods Group, and Cochrane Statistical Methods Group. 2011. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928.
IOM (Institute of Medicine). 1982. Marijuana and health. Washington, DC: National Academy Press.
IOM. 1999. Marijuana and medicine: Assessing the science base. Washington, DC: National Academy Press.
IOM. 2008. Treatment of posttraumatic stress disorder: An assessment of the evidence. Washington, DC: The National Academies Press.
IOM. 2012. Adverse effects of vaccines: Evidence and causality. Washington, DC: The National Academies Press.
King, L.A. 2014. Legal controls on cannabimimetics: An international dilemma? Drug Testing and Analysis 6(1-2):80–87.
Marsot, A., C. Audebert, L. Attolini, B. Lacarelle, J. Micallef, and O. Blin. 2016. Comparison of cannabinoid concentrations in plasma, oral fluid and urine in occasional cannabis smokers after smoking cannabis cigarette. Journal of Pharmacy and Pharmaceutical Sciences 19(3):411–422.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. Veterans and agent orange: Update 2014. Washington, DC: The National Academies Press.
NCSL (National Conference of State Legislatures). 2016. State medical marijuana laws. November 9. http://www.ncsl.org/research/health/state-medical-marijuana-laws.aspx (accessed November 21, 2016).
Small, E. 2015. Evolution and classification of Cannabis sativa (marijuana, hemp) in relation to human utilization. The Botanical Review 81(3):189–294.
Wells, G. A., B. Shea, D. O’Connell, J. Peterson, V. Welch, M. Losos, and P. Tugwell. 2014. The Newcastle–Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed November 2, 2016).
Whiting, P. F., R. F. Wolff, S. Deshpande, M. Di Nisio, S. Duffy, A. V. Hernandez, J. C. Keurentjes, S. Lang, K. Misso, S. Ryder, S. Schmidlkofer, M. Westwood, and J. Kleijnen. 2015. Cannabinoids for medical use: A systematic review and meta-analysis. JAMA 313(24):2456–2473.
Whiting, P., J. Savovic, J. P. T. Higgins, D. M. Caldwell, B. C. Reeves, B. Shea, P. Davies, J. Kleijnen, R. Churchill, and the ROBIS Group. 2016. ROBIS: A new tool to assess risk of bias in systematic reviews was developed. Journal of Clinical Epidemiology 69:225–234.
WHO (World Health Organization). 2016. The Health and Social Effects of Nonmedical Cannabis Use. Geneva, Switzerland: WHO Document Production Services.
This page intentionally left blank.


















