4
Upper-Extremity Prostheses
This chapter provides an overview of the various levels of amputation and congenital limb absence, their prevalence, and the types of prosthetic devices commercially available for each level. In addition, it reviews relevant clinical considerations pertaining to upper-extremity prostheses (UEPs), including their role in helping to mitigate the effects of impairments due to missing limbs. The evaluation and monitoring of individuals who need UEPs, the training and adaptation required to use such a device, and considerations of access and availability are addressed in turn. The chapter ends with findings and conclusions. Note that the discussion of prostheses and amputation, unless otherwise specified, applies to persons with congenital absence of a limb as well as to those with acquired amputation.
OVERVIEW OF CONDITIONS BENEFITING FROM UPPER-EXTREMITY PROSTHESES
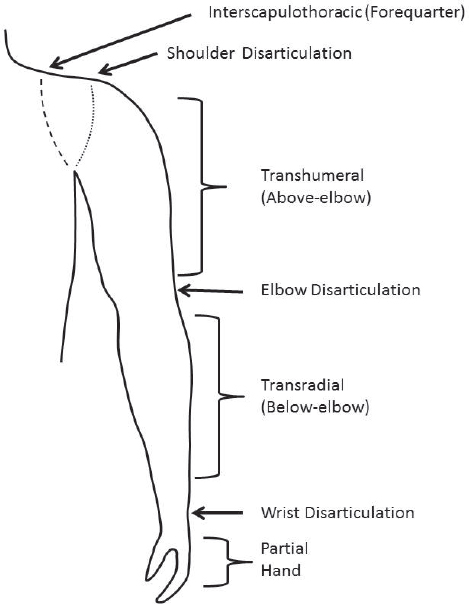
Upper-extremity (upper-limb) amputations are defined by the level at which they occur (see Figure 4-1). Although congenital limb absence is properly defined with a slightly altered taxonomy, individuals are fit with prosthetic devices according to the corresponding amputation level regardless of whether the limb deficiency is congenital or acquired (Schuch and Pritham, 1994). The remaining part of the limb is often referred to as the “stump”; however, the more accepted term in the United States and the term used in this report is “residual limb.”
Amputation of one limb is termed “unilateral amputation,” while amputation of both the right and the left limb is termed “bilateral amputation.”
SOURCE: Adapted from Schuch and Pritham, 1994.
Given the added challenges in performing tasks with bilateral as opposed to unilateral amputations, prostheses used for bilateral amputations often have additional features. For example, wrist flexion units and a variety of components (e.g., two different hook styles) may increase the range of functional movement for people with these amputations and allow for improved performance of activities of daily living (Lehneis and Dickey, 1992; Uellendahl and Heckathorne, 1989).
A “transradial amputation” (or “below-elbow amputation”) occurs through the long bones of the radius and ulna, while a “transhumeral amputation” (or “above-elbow amputation”) occurs through the humerus. An amputation through a joint is called a “disarticulation.” Therefore, an individual may have a wrist disarticulation amputation (between the carpals and radius and ulna), an elbow disarticulation (between the radius and ulna and humerus), or a shoulder disarticulation (between the humerus and scapula). Amputations proximal (closer to the center of the body) to the shoulder are termed “interscapulothoracic amputations” (sometimes called “forequarter amputations”). Partial hand amputations are amputations
distal to the wrist joint, best described by the bones that are involved using the International Organization for Standardization terminology (ISO, n.d.): (phalangeal, metacarpo-phalangeal disarticulation, metacarpal, carpometacarpal disarticulation, and carpal).
In the United States in 2005, it is estimated that 41,000 people experienced upper-limb loss at levels at or above the wrist (Ziegler-Graham et al., 2008), with upper-limb amputations accounting for the majority of trauma-related amputations (68.6 percent) (Dillingham, 1998). According to data available for 2013, more than 10 times more lower- than upper-limb amputations were performed (O&P Almanac, 2016). During this year, 10,365 upper-limb amputations were performed. Of these, 5.2 percent occurred at the wrist disarticulation and transradial level, 6.1 percent were elbow disarticulation or transhumeral, and 2.3 percent were at the shoulder or higher. The majority (75.6 percent) were finger amputations (O&P Almanac, 2016). Similar percentages by level have been documented by the National Trauma Data Bank and by surveys of individuals with amputation (Inkellis et al., 2015).
The majority of all upper-limb amputations are the result of trauma (Raichle et al., 2008; Ziegler-Graham et al., 2008). Other common causes of upper-limb loss are vascular/infection, congenital absence, and cancer (Raichle et al., 2008). In general, the distribution across levels is the same for all causes, with the exception of cancer, which is more likely to result in a higher level of amputation (Dillingham, 1998). Partial hand amputations are the most common, accounting for 75 percent of all traumatic amputations (O&P Almanac, 2016). An estimated 500,000 people were affected by amputation of the hand or fingers in the United States in 2005 (Ziegler-Graham et al., 2008).
The function of the upper extremities is far more difficult to replace than that of the lower extremities. This is the case because the primary functions of the lower limbs are more limited and concern primarily maintenance and achievement of upright stance and various types of locomotion (e.g., walking, running, hopping, jumping, stair climbing). In contrast, the primary functions of the upper extremities include not only gross and fine motor activities but also more complex combinations of activities, such as self-care, interaction with the environment and others, and self-expression. An upper extremity is a high degree-of-freedom system, allowing for great mobility to move the hand into a range of positions around the body. The complexity of the human upper extremity is illustrated by the massive proportion of space within the motor and sensory areas of the brain (the motor and sensory homunculi) dedicated to the motor control and sensation of the hand and fingers. Thus, it is not surprising that persons with upper-limb amputation are generally less satisfied with the restoration of function provided by their prostheses relative to those with lower-limb amputation (Raichle et al., 2008).
TAXONOMY1
There are four main categories of prostheses, defined by the method used to control the device.
The most basic is the passive prosthesis. A passive prosthesis allows for no active movement of any of the joints. These are the lightest-weight devices because they contain no motors and few mechanical systems. Although these devices are often termed “cosmetic,” they can provide function by assisting the intact hand/arm with bimanual tasks. For example, a passive prosthesis can be used to hold down papers when writing, to help carry items, or to stabilize objects held in the intact hand. They can also fill out and support clothing. Since they provide for no other controlled “function,” however, these devices are not discussed in detail in the remainder of this chapter.
Next is the body-powered prosthesis (see Figure 4-2), so called because it is moved by the individual’s remaining body. Typically, a harness with a strap that lies over the lower third of the scapula connects to a cable that operates the device. With biscapular abduction, the cable is pulled in the same way that pulling the handle on a bicycle break pulls on the cable that closes the brakes. The benefits of body-powered prostheses are that they are relatively lightweight and durable, can be made to be waterproof, and can provide feedback to the user based on the tension in the control cable. The disadvantages are that they require harnessing, and the user must have the strength and range of motion to pull the cable sufficiently to make the device work in all positions, particularly overhead.
Third is the externally powered prosthesis (see Figure 4-3). Whereas a body-powered system’s power comes from the user’s movements, an externally powered prosthesis is powered by batteries contained within the system. The device can be controlled with various inputs, including electromyographic (EMG) signals, force-sensing resistors, pull switches, and push switches (Esquenazi, 2015). Physical switches are a good complement to EMG signals for high-level amputees when multiple motors must be controlled or when the EMG signal is insufficient. Given that the most prevalent type of externally powered device is a myoelectric prosthesis, the following description focuses on those devices.
A typical myoelectric control scheme, direct control, uses EMG signals from two antagonist muscle contractions to operate two directions of movement. With a simple two-site direct control system, for example, wrist extensor EMG signals control opening of the hand, while wrist flexor EMG signals control closing of the hand. Thresholds are set for each muscle to
___________________
1 The images in this section serve as examples of device categories only and should not be considered an endorsement of specific products or manufacturers.
allow some contraction without the occurrence of inadvertent movement. Most modern systems provide proportional control, in which the magnitude of the signal above the threshold is proportional to the speed of device movement and the generated grip force.
The benefits of myoelectric prostheses are that they typically use no harnessing or less harnessing than body-powered systems; they often can be operated in more planes of movement; the terminal devices (i.e., hands/hooks) can generate more force; and because there are no cables and straps on the outside of the device, they can appear more cosmetic. The disadvantages are that the batteries and motors make them heavier than body-powered systems; they can be water-resistant but not waterproof; they need to be charged daily; they require more maintenance than body-powered devices; and they are more expensive than those devices. Because of their complexity, they are also more prone to break and need repair (Biddiss and Chau, 2007b). In addition, myoelectric prostheses require that the electrode sensors that record signals from the muscles to control the device maintain contact with the skin. Thus, they require an intimate fit that may be uncomfortable or not tolerated by fragile skin or may be impeded or disrupted by scar tissue or excessive sweating. Contact with the electrodes also can be disrupted if the prosthesis is donned inappropriately or if changes occur in residual limb size or shape. Moreover, if more than two controlled motions (degrees of freedom) are available in the limb (e.g., elbow flexion and extension, as well as hand open and close), the same two antagonist muscles will be used for controlling each movement. In such cases, various strategies must be used to alternate control between the multiple movements in a sequential manner. The user must generate a trigger, such as co-contraction of both muscles or quick contraction of one muscle, to switch between the various movements, such as elbow and hand or various hand grip patterns. If the system is extremely complex (elbow, wrist rotation, various hand grasp patterns), the user may need to generate multiple types of triggers in a specific order to accomplish the various movements. Although there are many options for control with a trigger-controlled system, a user in a typical scenario would co-contract the wrist flexors and extensors to alternate control of wrist flexion between hand close and pronation or wrist extension between hand open and supination (Williams, 2004). When a multifunction hand is used, the various switching mechanisms, or triggers, such as co-contraction, impulse, and the like, can be used to alternate between the various hand grasps. These “Morse code” systems of control can be difficult to learn and cognitively burdensome to use.
A number of emerging technologies are being developed to overcome some of the above limitations of myoelectric prostheses. For grip selection there are devices that employ the use of gesture control by using imbedded accelerometers to switch grasp (Touch Bionics, n.d.-a). There are also
passive radio frequency identification tags that can be placed in the environment, and when the device approaches these tags, it will automatically switch to the preconfigured grip needed in that location (Infinite Biomedical Technologies, n.d.; Touch Bionics, n.d.-b). With pattern recognition control, individuals do not use triggers to alternate the motors being controlled by the same two antagonist muscles (Chicoine et al., 2012; Englehart and Hudgins, 2003; Lock and Hargrove, 2014; Scheme and Englehart, 2011). Instead, numerous sensors are placed on the residual limb, and a microprocessor is “trained” to recognize the various patterns of movement, with the goal of making movements more intuitive and less mentally taxing (Deeny et al., 2014). For example, individuals with a transradial amputation can control wrist rotation and hand open/close by trying to contract the muscles in a way that feels more like they are performing those movements. Another emerging control option is inertial measurement unit (IMU) control, which will be available with the upcoming commercial release of the DEKA arm (anticipated in 2017) (see Figure 4-4) (Resnik et al., 2014b). With this technology, IMUs worn on the top of the shoes detect the speed and direction of foot movements, which can be programmed to control specific functions of the device, and the EMG sensors are implanted within the muscle tissue instead of being on the surface of the residential limb. Since a conventional myoelectric device requires that the sensors maintain contact with the skin in order to record accurate EMG signals to control

SOURCE: Courtesy of the Providence VA Medical Center.
the device, a tight-fitting socket is necessary so that the sensors do not shift or pull away from the skin even as the user carries items, extends the arm in various planes, or perspires. Although not yet commercially available, methods such as IMUs worn on the feet (Resnik et al., 2014b) and EMG sensors implanted within the muscle tissue (Weir et al., 2003) avoid the use of surface EMG sensors and may allow a looser-fitting, and possibly more comfortable, socket fit.
The final category of devices is hybrid prostheses, which combine body-powered components and myoelectric/externally powered components in one device (see Figure 4-5). They are used for transhumeral and shoulder disarticulation prostheses and most commonly include a body-powered elbow and a myoelectric terminal device (hook or hand). This configuration allows both components to be operated simultaneously and provides the increased force of the powered hand for gripping, which is not required by the lightweight body-powered elbow. Annex Table 4-1 at the end of this chapter summarizes some of the features of body-powered, myoelectric, and hybrid prostheses for different levels of upper-limb amputation.
Prosthetic Components
For each missing joint, various options are available. For replacement shoulder function, there are only passive components, either friction or locking. Currently, no shoulder joints are commercially available that are motorized or body-powered; the same is true for replacement of humeral rotation. However, the commercial release of the DEKA arm will offer both powered shoulder movement and humeral rotation. At the elbow, there are motorized joints (myoelectric/externally powered) as well as body-powered joints. The motorized elbows have passive lift (how much can be carried) of approximately 50 pounds, which is comparable to body-powered elbows, although none of the motorized joints are able to flex actively with more than a few pounds held in the hand (Heckathorne, 1992). Available at the wrist are friction/locking and motorized wrist rotators; at this time, however, there are only friction/locking wrist flexion units.
Many varieties of terminal devices (hooks or hands) are available for both myoelectric and body-powered prostheses. There are body-powered hook options—called voluntary opening (VO) devices—in which pulling on the cable opens the device, and a spring or rubber bands cause it to close (see Figure 4-6). The hooks can be aluminum, titanium, or steel, depending on the durability needed, although the more durable ones are often heavier. One limitation of VO devices is that grip strength is limited by the rubber bands or springs used. To achieve a higher grip strength, the user must pull against this force every time the device is opened. Another variation is voluntary closing (VC) devices, in which pulling on the cable causes the

SOURCE: Courtesy of Motion Control division of Fillauer.
device to close, and a lightweight spring causes it to open. The user can generate much higher grip forces with a VC than with a VO device, but unless using some kind of cleat or cable lock, must maintain tension on the harness to keep the device closed. The amount of force required varies across types of VC terminal devices (Smit and Plettenburg, 2010). Both VO and VC hands also exist. A body-powered prosthesis can be provided with a hook, a hand, or both to interchange in specific situations, most often for cosmesis or for use in social settings. Specialized work or recreation devices may also be interchanged on a body-powered system to maximize function during tasks when a traditional hook or hand device is not optimal (Texas Assistive Devices, 2016; TRS Prosthetics, 2016).
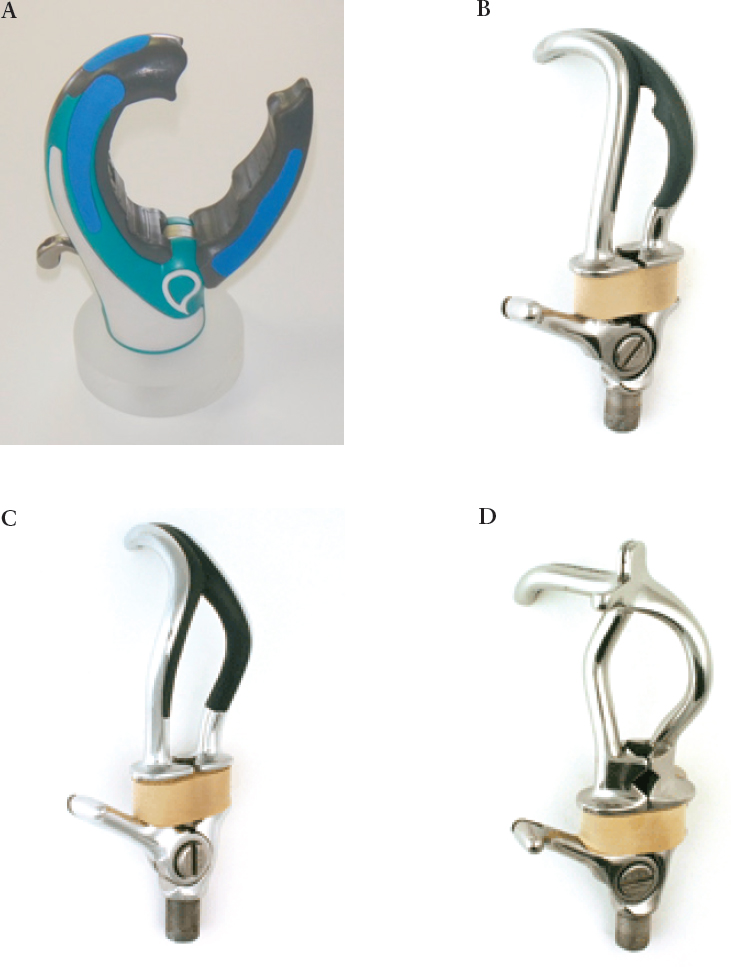
NOTE: A. TRS GRIP 5, Voluntary Closing, Body-Powered Prehensor; B. Hosmer 5X Hook, canted-shape voluntary opening hook; C. Hosmer 555 Hook, lyre-shape voluntary opening; D. Hosmer 7 Work Hook, voluntary opening.
SOURCES: A. Courtesy of TRS Inc.; B-D. Photos for Hosmer Hooks provided by Fillauer LLC.
For body-powered systems, it is important that the individual have the range of motion and strength to operate the device. To fully open a standard VO hook, 2 inches of cable excursion is required. For a body-powered elbow, an additional 2.5 inches is required. To enable this full range of movement, the socket must fit well, and the harness must be properly fit to the user—a potentially time-consuming task. Proper fitting of the harness also often requires that it fit very tightly on the body, which can cause discomfort and lead to abandonment.
Motorized hooks and hands also are available (see Figures 4-7 and 4-8, respectively). They are much heavier (approximately 1-1.5 pounds) than the body-powered hooks (0.25-0.5 pound) and hands (0.75 pound) (Belter et al., 2013; Fillauer Companies, Inc., 2016; Hosmer, 2017; Steeper, 2016). The advantage of motorized hooks is that they are more durable than a motorized hand and can generate much more force than the frequently used VO hooks. Motorized hands can be divided into two categories: single-degree-of-freedom and multiarticulating. Single-degree-of-freedom hands have one motor and only open and close. They are the most durable type of myoelectric hand and can generate the most pinch force (Belter et al., 2013).
Multiarticulating hands have more than one motor to generate multiple grasp patterns (see Figure 4-9). The most complex have a motor in each digit. Because the motors are smaller, these hands cannot generate the same pinch force as a single-motor hand; by individually powering each digit, however, a more conforming grasp can be achieved.

NOTE: A. Electronic Terminal Device (ETD) from Motion Control; B. Greifer from Ottobock.
SOURCES: A. Courtesy of Motion Control division of Fillauer; B. Courtesy of Ottobock.
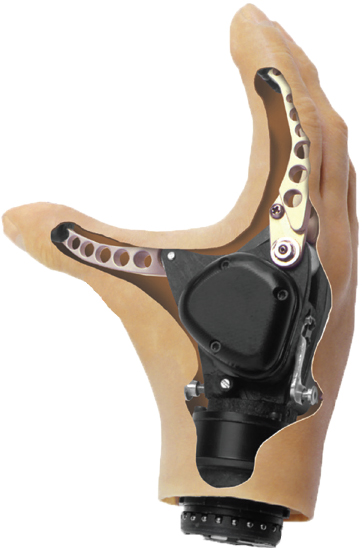
SOURCE: Courtesy of Motion Control division of Fillauer.

NOTE: A. Michelangelo hand; B. i-limb quantum XS; C. bebionic (small).
SOURCES: A. Ottobock, 2016a, courtesy of Ottobock; B. Courtesy of Touch Bionics by Össur; C. bebionic, courtesy of Steeper Group.
Prosthetic Sockets
Although component selection is important for prosthetic fitting, the aspect that most often impacts ultimate prosthesis use is the user interface, or socket. The socket is the framework that holds the device onto the remaining limb, and the comfort of the fitting, along with skin integrity, has a direct impact on acceptance of the device (Biddiss and Chau, 2007b,c; Schultz et al., 2007). During the initial fitting with a new device, an individual is typically fit first with a diagnostic or test socket. This type of socket often is made out of a clear material that allows the prosthetist to evaluate interface pressure and the presence of total contact and make adjustments as needed. Components often are connected to the check socket to confirm a comfortable and appropriate fit as the user operates the device in multiple planes. The check socket is then completed in the final materials, which may be of multiple types—for example, rigid laminations, flexible materials, and gel inserts. To improve socket comfort and therefore acceptance of the devices, prosthetists and engineers have continued to develop new materials, new socket designs, and designs that allow the user to customize the socket by tightening or loosening it for various functions and needs (Alley et al., 2011; Baschuk, 2016; Miguelez and Miguelez, 2003; Miguelez et al., 2016).
Obtaining a well-fitting socket can be challenging because the residual limb is often irregular in shape and density, with scarring, irregular soft tissue coverage, or the presence of bony projections. The prosthetic socket must optimize stability while controlling for movement such as slippage, translation, and rotation of the soft tissues and the socket itself (Resnik et al., 2014a). Even when the socket fits well, maintaining a consistent fit over time can be challenging because the shape and volume of the residual limb may change with changes in weight and fluid volume. Additionally, some people experience volume changes in their limb throughout the day, causing variability in socket fit and comfort even without long-term overall changes in body weight that may occur more slowly over time.
Osseointegration is an emerging surgical technique designed to bypass the impact of the socket on the success of the fitting. This technique, used clinically outside of the United States, was only recently approved by the U.S. Food and Drug Administration (FDA) for lower-limb applications (FDA, 2015) and has not yet been approved for upper-limb applications. The surgery entails connecting a metal rod to the remaining bone. The rod then protrudes through the skin (Aschoff et al., 2010; Branemark et al., 2001; Jönsson et al., 2011; Muderis et al., 2016; Palmquist et al., 2008; Pitkin, 2013), and the user can connect the prosthesis to the rod as desired. Although this technique does eliminate the need for a socket, challenges remain in interfacing with the residual limb to record EMG signals for a myoelectric prosthesis or to connect the control straps for a body-powered
system. For myoelectric devices, research is ongoing to record EMG signals more directly through implanted myoelectric sensors (as discussed above) and through the osseointegration abutment (Ortiz-Catalan et al., 2012, 2014).
Box 4-1 provides a summary of definitions relevant to the taxonomy of UEPs.
CLINICAL CONSIDERATIONS
Mitigating the Effects of Impairment
Upper-limb amputation and limb deficiency are associated with self-reported disability and diminished functional performance (James et al., 2006; Lerman et al., 2005; Postema et al., 2012; Resnik et al., 2012; Tennent et al., 2014). These limitations and restrictions are greater with bilateral than with unilateral limb loss (Davidson, 2004). Disability also
is typically greater with more proximal-level (transhumeral and shoulder-level) than with transradial-level amputation (Lerman et al., 2005; Resnik and Borgia, 2015), although people with partial hand amputation report greater disability than those with either transradial or transhumeral amputation (Davidson, 2004). Additionally, some evidence suggests that the impact on functional performance is greater for acquired than for congenital limb loss (James et al., 2006), although the evidence for this finding is mixed (Lerman et al., 2005).
Several small studies have found that persons with upper-limb amputation who use a prosthesis report less disability, as measured by the Disabilities of the Arm, Shoulder, and Hand (DASH) and QuickDASH questionnaires, relative to those who do not use such a device (Lifchez et al., 2005; Resnik and Borgia, 2015). However, as many as one-third of prosthesis users report that they do not find their device to be useful to them when performing daily activities or work functions (Datta et al., 2004). Prosthesis users report dissatisfaction with their ability to use their devices in such daily tasks as food preparation, eating, and self-care (Ritchie et al., 2011). And some research focused on pediatric congenital amputees has found no differences in functional performance between children who utilize a prosthesis and those who do not (James et al., 2006), suggesting that children adapt to limb deficiency at a young age and maximize function without the use of a prosthetic limb.
It is clear that currently available upper-limb prostheses cannot replace the complex functions of the missing upper limb because of limitations inherent in their control and design, their lack of sensory feedback, and the methods required to suspend them onto the residual limb. The human hand, wrist, elbow, and shoulder complex together comprises 32 bones, with dozens of movable joints and muscles, which together enable a wide field of active movement around the body, sometimes called the “functional envelope” (Harrtigan and Serrafin, 2004).
Although prosthesis use may in some instances partially mitigate the impact of upper-limb amputation on impairments in body functions and limitations in activity, the amount of mitigation provided by the prosthesis varies by the type of device used, as well as the level of amputation (see Annex Table 4-2) and, as discussed later in the chapter, of user training and ability. The findings in Annex Table 4-2 were determined through the consensus of committee members based on their expert knowledge, review of selected manufacturers’ specifications, and review of the scientific literature. These findings are summarized below.
Body Image
The experience of acquired amputation is disruptive to one’s body image and sense of self-esteem (Morris, 2008; Racey, 1992; Winchell, 1996). Body image includes physical, psychological, and social components. The upper extremities are an integral part of a person’s self-identity, communication, and social interactions with others (Desteli et al., 2014), and acquired amputation, as well as congenital limb absence, can be associated with significant psychological distress, embarrassment, and shame due to perceived social stigma. Some persons with amputation may experience prejudice from others and a sense of injustice as a result of the visibility of their disability. For some people with limb loss, cosmetic restoration is highly valued, or even preferred over functional restoration, because of its mitigating effect on the disruption to body image. People with upper-limb loss in particular report high levels of distress about body image, and for them, the cosmetic aspects of the prosthesis are considered more important than is the case for people with lower-limb loss (Desteli et al., 2014). Thus, some people with limb loss may prefer terminal devices that resemble more closely the shape of the hand than a hook because they appear more like an unimpaired hand, and so may draw less attention to the prosthesis (Flannery and Faria, 1999; Hanson, 2003). Indeed, concerns about body image and the desire to improve cosmesis with prosthetic devices can result in rejection of prosthetic devices that may be more functional and may impact the choice of prosthetic devices, particularly for activities, such as work, that involve interaction with others (Morris, 2008; Racey, 1992; Winchell, 1996). Completely passive prostheses provide the best mitigation of cosmetic impairment. Cosmetic coverings can approximate the skin tone of the lost limb, and custom coverings may provide high-definition, cosmetic restoration that fully mirrors skin coloring, as well as anatomical details such as freckles or even hair of the lost limb. As discussed later in the chapter, however, access to such custom coverings varies because of limitations in reimbursement and the availability of providers who can make them.
Sensation
Research is under way on means of providing sensory function to prosthesis users (Carlsen et al., 2014; Chaubey et al., 2014; Cipriani et al., 2011, 2012; Clippinger et al., 1974; Hebert et al., 2014, 2016; Marasco et al., 2009; Nghiem et al., 2015; Rager et al., 2013; Schiefer et al., 2016; Schofield et al., 2014; Tabot et al., 2013; Tan et al., 2015; Tyler, 2015; Witteveen et al., 2014, 2015). Some existing devices have crude sensors for grip pressure or other features intended to provide the user with
information on touch or object slip. However, all currently available devices fall short of restoring the sense of touch or proprioception of the missing appendage. Limited proprioception is available because users can feel the weight of the socket on the residual limb as the device is positioned in space and the muscular energy associated with the body movements required to pull the cable (for body-powered devices) or contraction of the musculature required to operate the myoelectric controls (for myoelectric devices). Loss of sensation makes control of grip force problematic, as prosthesis users cannot tell how hard they are gripping objects; thus, they are susceptible to dropping items because of too little or improper grip or to crushing fragile items by using a stronger grip than intended (Schiefer et al., 2016). To ensure that an object is held firmly, the user may be able to listen for the sound of the motors stalling or just hold a contraction after contacting an object so a large grip force is generated. For the most part, however, the user must rely on vision to modulate grip force and object placement (Sobuh et al., 2014), and so may avoid handling delicate, fragile, or slippery objects with the prosthesis. Lack of sensation also impairs the performance of fine motor activity.
Passive Range of Motion
No currently available upper-limb prosthesis can execute all of the passive movements of the human upper extremity. Although some advances have been achieved in improving the movements of multiarticulating prosthetic hands, even the most advanced of these devices, which may allow passive movement of the thumb or other digits, do not possess all of the passive range of motion of the human hand. The most commonly used terminal devices—body-powered and myoelectric hooks—have only a single degree of freedom, meaning that they can be moved only in two opposing directions—open and closed. Other myoelectric devices, such as the i-limb and Michelangelo hands, have a movable thumb and compliance in the digits that enable some degree of passive movement to conform to grasped objects.
Some existing prosthetic wrists allow passive wrist pronation and supination. However, prosthetic wrists lack full passive flexion/extension and radial/ulnar deviation, although there are some compliant wrists that allow a small amount of passive wrist movement in these directions. In contrast, most prosthetic elbows possess good passive flexion and extension movement, although some myoelectric elbows can be positioned only by motor action. Some shoulder-level prostheses have a fixed shoulder joint that does not move at all, while others can be manually positioned and then locked in a limited set of positions or held in place with friction.
Active Range of Motion
Active control of the movements of upper-limb prostheses is limited by both hardware constraints and currently available prosthetic control options. Even when passive range of motion is available (as described above), the user may not have active range of motion or control of that range. This may be because the device lacks the actuators and motors to move the joint. Some hand designs, for example, are under actuated, meaning that joints are linked together so they can be powered by a single motor or action. As another example, certain multiarticulating hands that have passive thumb movement require the user to position the thumb manually to change the thumb alignment. Further, no currently available devices allow active control over wrist flexion and extension or radial and ulnar deviation, humeral rotation, or powered shoulder movement in any direction.
Limitations in the active range of motion of any joint negatively impact the size of the functional envelope and are considered a contributing factor in the compensatory movements of the trunk observed in kinematic studies of upper-limb prosthesis users (Carey et al., 2008). This abnormal movement may, in turn, be associated with overreliance and overuse injuries of the sound side that have been reported (for people with unilateral amputation) and the more proximal joints and/or the neck and back (Burger and Vidmar, 2016; Østlie et al., 2011; Postema et al., 2012, 2016a).
The current standard for controlling prostheses is direct control, meaning that the user directly operates a specific control to activate a single intended movement, such as wrist extension to control hand opening (Phillips et al., 2013). In most cases, each intended prosthesis movement must be activated by the user in a sequential fashion, and it is difficult, if not impossible, to engage available powered movements simultaneously. Users of traditional two-site EMG myoelectric devices who may have powered elbow flexion/extension, wrist pronation/supination, and terminal device open/close cannot operate any of these movements simultaneously. Instead, they must use an alternate signal (frequently a muscular co-contraction) to switch control between joints (also termed switch modes). Another challenge for users of myoelectric prostheses is that active range of movement may not always function reliably. It is well known that myoelectric controls may function erratically if the surface electrode contact is disrupted. As discussed earlier, this can sometimes happen if the socket fit changes as a result of changes in residual limb volume or if the residuum becomes excessively sweaty.
Activities
Grasp, fine motor use, dexterous activities, and handling objects The human hand has more than 30 distinct grasp (grip) patterns (Cutosky, 1989), which are sometimes grouped into 6 or more major categories based on the shape of the grasping hand. Commercially available prostheses have between 1 and 14 (Ottobock, 2016b; Steeper, 2016; Touch Bionics, 2016). However, the most commonly used single-degree-of-freedom terminal device, the split hook, has a single grasp, although there are various styles of hooks. Grip strength and speed vary widely across these devices and type of grip (Belter et al., 2014; van der Niet et al., 2013).
Studies reporting the overall functionality of commercially available devices compared with limbs of intact subjects, as measured by the overall index-of-function score of the Southampton Hand Assessment Procedure (SHAP), show wide variation in mitigation of impairments of grasp. To date, no terminal device tested has been shown to restore function completely to all major grasp patterns. Body-powered terminal devices may be VO or VC, with some evidence that VC devices are associated with faster task performance as measured by the SHAP (Berning et al., 2014). SHAP index-of-function scores for several two-joint, single-degree-of-freedom terminal devices have been reported to be about 74 percent and 43-84 percent, respectively, compared with the functionality typical of an intact hand (Dalley et al., 2012; Luchetti et al., 2015; van der Niet et al., 2013).2 The SHAP index-of-function scores for several commercially available multiarticulating hands have been reported in multiple studies: 52-76 percent for the i-limb Hand (Dalley et al., 2012; van der Niet et al., 2013), 87-88 percent for the i-limb Pulse (van der Niet et al., 2013), and 75-89 percent for the Michelangelo hand (Luchetti et al., 2015). Other research has found that myoelectric prosthesis users had average index-of-function scores of 43-50 percent compared with an intact hand (Bouwsema et al., 2012).
Few studies have directly compared the dexterity of myoelectric and body-powered terminal devices or multiarticulating and conventional myoelectric terminal devices. In one study, the functionality of a myoelectrically controlled single-degree-of-freedom terminal device was reported to be lower than that of a single-degree-of-freedom body-powered device as measured by the SHAP (Kyberd, 2011). Another study, comparing the performance of six transradial amputees using both a multiarticulating (Michelangelo) hand and a standard myoelectric hand, found better dexterity (box and blocks test and SHAP tests) using the multiarticulating hand (Luchetti et al., 2015).
___________________
2 The index of functionality is the percent of function compared with an index hand as measured by the SHAP test.
Partial hand amputations range from single-digit amputations to those that involve multiple digits or part of the hand itself. Although single-digit amputations typically do not result in significant functional deficits, partial hand amputations that involve multiple digits and/or the thumb, compromising prehension force and sensation, can result in significant disability. Many people with partial hand amputations lose the opposition of digits necessary for an effective grasp, particularly if the amputation involves the thumb. Restoration of grasp to persons who have lost part but not all of their hand has been particularly challenging, with little research having been conducted in this area. A single study of 10 people with partial hand amputations showed that prosthesis use improved the strength of some but not all grips and also improved self-reported difficulty in the performance of specific activities (Lifchez et al., 2005).
Several studies have found substantially lower dexterity in users of upper-limb prostheses of all amputation levels compared with age-matched norms (Resnik and Borgia, 2012), as well as slower time to complete movements and activities (Bouwsema et al., 2010b; Cowley et al., 2016). Slower speeds are attributable, in part, to the fact that prosthesis users must perform more discrete submovements to perform basic tasks (Doeringer and Hogan, 1995; Fraser and Wing, 1981). In addition, grasping is uncoupled from reaching when one is using a prosthesis, which makes reaching for and grasping an object take longer (Blough et al., 2010; Bouwsema et al., 2012; Cowley et al., 2016).
Lifting, carrying, and reaching overhead Lifting and carrying objects with a prosthesis can be limited by the design restrictions of the prosthetic components, the method used to attach the prosthesis to the socket, or the socket and its suspension system. Lifting and carrying can be limited if the prosthesis hardware will not support the weight of the object, if the device detaches from the socket, or if the socket slips from the residuum. The functional envelope, or the space around the body in which the prosthesis can be used reliably, is often limited. Even when individuals with amputation have use of their own shoulder joint for reaching activities, they may be unable to use their prosthesis when reaching overhead because of the constraints of the harnessing that operates the cable. Users of myoelectric devices may also have difficulty operating their device in a full range of body positions because involuntary co-contraction of residual limb muscles that often occurs when stabilizing the limb against gravity can interfere with voluntary control of residual musculature.
Effects of Multiple Impairments on Impairment Mitigation
Acquired upper-limb amputations and congenital limb deficiencies are often associated with other impairments and comorbidities that affect overall function and can impact the use of prosthetic devices to mitigate impairment. The function of the upper extremities is complex and requires coordinated movements of multiple joints, visual feedback, sensation, and proprioception to accomplish even simple tasks.
Persons with amputation often have comorbid injuries, such as burns, nerve damage, or muscular loss, that interfere with the skin integrity, range of motion, strength, and/or sensation of the residual limb. Weakness of the proximal arm or trunk musculature and/or pain in the proximal joints, neck, or back can make it difficult to lift the weight of the prosthetic limb or to tolerate wearing a prosthesis for long periods. Thus, even those who wish to wear an upper-limb prosthesis may be unable to do so to the extent that they desire. Comorbid injuries and conditions that affect vision, cognitive ability, or the upper extremity/trunk proximal to the limb deficiency will make using a prosthesis more challenging and will limit the impairment mitigation effects of a prosthetic device. In addition, comorbid conditions that cause limb volume fluctuations, such as diabetes or renal or cardiovascular disease, can impact a person’s ability to fit comfortably and consistently in a prosthetic device.
As noted earlier, beyond the physical conditions that often accompany upper-limb deficiency, the impact of limb loss or deficiency on body image, social role, and psychological health is substantial (Gallagher et al., 2007). In addition to the body image issues discussed above, it is estimated that at least 27 percent of people with limb loss experience symptoms of depression and that the risk for developing depression is greater for people with comorbid conditions (Desmond, 2007; McKechnie and John, 2014; Perkins et al., 2012). People with traumatic amputations are also at increased risk of developing posttraumatic stress disorder (PTSD), with prevalence rates estimated at 17-77 percent (Copuroglu et al., 2010; Tennent et al., 2014; Vincent et al., 2015).
In addition to psychological distress, upper-limb loss is frequently associated with chronic pain (Hanley et al., 2009; Kooijman et al., 2000). Several studies have found that people with upper-limb amputation report more postamputation pain relative to people with lower-limb amputation (Bosmans et al., 2010; Davidson et al., 2010). It is important to consider this high prevalence of postamputation pain, including phantom limb pain, when examining the use of prosthetic devices, as pain is a common reason why people with limb loss are not able to wear prosthetic devices successfully or restrict their use of the devices (Desmond et al., 2012).
Effects of User’s Age on Prosthesis Use
Little is known about the association between age and use of upper-extremity prosthetic devices, particularly as children transition to adulthood and enter the workforce. A number of studies have examined factors that affect the use of prosthetic devices in children with either congenital limb deficiencies or acquired amputation. In general, these studies have found that young children with acquired amputation are more likely to become users of upper-extremity prosthetic devices if they begin using them at a younger age (Dabaghi-Richerand et al., 2015; Meurs et al., 2006). One study of 218 children aged 2-20 years in the Netherlands found that the majority preferred the use of unaffected parts of the body to accomplish tasks (>60 percent) over adaptive devices (<48 percent) and prostheses (<9 percent) (Vasluian et al., 2013). In this study, 27 percent of children reported having difficulties with work (the majority of these were children aged 13-20, and work for them was defined as any part-time, summer, or full-time job), yet none of these children reported using adaptive or prosthetic devices for their work. The children in this study who used both adaptive devices and prostheses reported higher satisfaction with the use of adaptive devices for specific activities compared with prosthetic devices because of the former devices’ dimensions, weight, adjustability, and ease of use.
Another survey conducted in Sweden (Sjoberg et al., 2014) found that a majority of adults with congenital limb deficiencies (68 percent) used assistive devices, including prosthetic devices, but there were gender differences in the types of devices used: those using body-powered prostheses were exclusively men, and women were more likely to use cosmetic prostheses. Among those with upper-limb deficiencies, the vast majority were either working or in school (93/108); however, 24 percent of people reported that their self-assessed work capacity was reduced because of their upper-limb deficiency.
A qualitative focus group study of children and adolescents (aged 8-20 years) with unilateral transradial congenital limb deficiencies found that the limitations they experienced were typically environmental—related to attitudes or lack of accommodations—rather than being due to the limb deficiency itself (de Jong et al., 2012). Study participants transitioning to adulthood and thus more dependent on adults reported that they were more limited in their function relative to those who had already transitioned to independence. This finding suggests that the transition period itself is challenging for adolescents with limb deficiency, a phenomenon that has been recognized in other populations of people with disabilities (Donkervoort et al., 2009). This qualitative study also highlights that children and adolescents with upper-limb deficiency use many different and creative strategies
to accomplish activities that do not necessarily rely on adaptive or prosthetic devices.
Prosthesis Use and Prognosis for Occupational/Educational Success
As noted earlier, congenital limb absence and acquired upper-limb amputation can significantly affect a person’s ability to work. Although a number of studies have reported return-to-work rates of 50-75 percent following lower-limb amputation (Dajpratham et al., 2008; Fisher et al., 2003; Schoppen et al., 2001a,b), the committee found limited data, particularly from the United States, documenting work reintegration following acquired upper-limb amputation. Several studies specifically examining return to work after upper-limb amputation have found employment rates of approximately 57-85 percent; however, none of these studies were conducted in the United States (Datta et al., 2004; Fernandez et al., 2000; Jones and Davidson, 1995; Millstein et al., 1986; Postema et al., 2016b). One of the studies found that a majority of people who returned to work changed their employment because of limitations associated with their amputation (Datta et al., 2004). Another study, conducted in the Netherlands, found that individuals with amputation were employed at lower rates (57 percent for acquired amputation, 74 percent for congenital limb absence) than their age- and sex-matched peers (82 percent) but reported similar work productivity. This study also found that prosthesis use was a predictor of employment (Postema et al., 2016b). Other studies similarly found a positive association between prosthesis use and return to work following amputation (Fernandez et al., 2000; Millstein et al., 1985, 1986). The precise relationship between prosthesis use and return to work is unclear (Fernandez et al, 2000), although some have speculated that positive attitude and motivation may contribute to both (Millstein et al., 1985).
A study of people with partial hand amputation found that fewer than half were able to return to the same job, and most found prosthetic devices insufficient to meet the demands of their work, although cosmetic prostheses were important to their work success (Burger et al., 2007). While a limited sample, a recent survey of people with major limb amputation in Ireland found that 92 percent (n = 11/12) of those with upper-limb amputation experienced difficulty with employment or job seeking, even more so than people with lower-limb amputation (69 percent, n = 96) (Gallagher et al., 2011). Another European study found that 38 percent of people with upper-limb amputation required modifications to their job duties but that people with amputation reported job satisfaction similar to that of age- and gender-matched controls (van der Sluis et al., 2009). One small case series found that of 13 civilians with upper-limb amputation in the United States, 4 were able to return to work in any capacity, although none were able
to return to their specific preinjury employment (Livingston et al., 1994). These findings are consistent with those of international studies, which found that upper-limb amputees typically returned to work in jobs that had fewer physical demands but required greater intellectual skills (e.g., clerical, service, managerial work) (Millstein et al., 1985). Similar findings also emerged from a study showing higher rates of return to work following upper-limb amputation in the building industry than in agriculture, presumably because agriculture offers fewer job opportunities compatible with a missing upper limb (Fernandez et al., 2000). Higher levels of employment for people with upper-limb amputation in more skilled jobs may explain the positive association found between postinjury employment and higher levels of education (Postema et al., 2016b), as well as receipt of vocational services (Millstein et al., 1985). Younger age at amputation and male sex also have been positively associated with return to work (Postema et al., 2016b).
In the United States, most of the studies examining the relationship between upper-extremity amputation and work duties were conducted in military populations. Although return-to-duty rates remain low for both upper- and lower-limb amputations, return to active duty is particularly challenging for people with upper-limb amputation (Belisle et al., 2013; Hurley et al., 2015). Roughly 75 percent of combat amputees from the global wars on terror were retired because of the amputation (Belisle et al., 2013). A recent study of all U.S. military amputations from 2001 to 2011 identified 153 people with upper-extremity loss (Tennent et al., 2014). In this population, no upper-extremity amputees were found to be fit for full duty, although 12 percent were allowed to continue on active duty (Tennent et al., 2014). Earlier studies had found higher return-to-duty rates for upper-limb amputees (17-22 percent), only slightly lower than the rates for lower-limb amputees (18-25 percent) (Stinner et al., 2010). Although military amputees are a unique population, and these findings may not be applicable to the general civilian population with upper-extremity amputation, they highlight the difficulties associated with return to work following upper-extremity loss. Further, upper-extremity amputees were more likely than lower-extremity amputees to experience PTSD and comorbid nerve injuries that impacted function (Tennent et al., 2014).
There are many reasons why employment may be difficult after upper-limb amputation, including limited ability to perform tasks repetitively (either with or without a prosthesis). One study found that bilateral amputees were less successful in returning to work relative to unilateral amputees (Millstein et al., 1985). Associated musculoskeletal complaints, including overuse injuries of the remaining limb, and pain also are negatively associated with return to work (Millstein et al, 1985; Postema et al., 2016b), and the impact of limb loss on psychological health, body image, and
social role may negatively affect return to work as well (Saradjian et al., 2008). Depending on the type of work, each of these issues may impact one’s ability to perform repetitive tasks involving the upper extremities and consequently, the quality of work and work productivity (Postema et al., 2016b). In addition, professions in which interactions with the public or social interactions are integral to job function may be more challenging for people who experience perceived social stigma or public self-consciousness due to their upper-limb loss (Saradjian et al., 2008).
Employment has been associated with decreased rejection rates of upper-limb prosthetics (Postema et al., 2016b), although this association depends in part on the level of amputation. Studies suggest greater employment among individuals with transradial relative to transhumeral amputations (Fernandez et al., 2000; Millstein et al., 1985), presumably because retention of one’s elbow helps with carrying out work-related tasks (Fernandez et al., 2000). Some studies suggest that people with more proximal amputations use prostheses primarily for cosmesis while at work, whereas people with transradial amputations use prostheses for functional activities, and people with partial hand amputations more frequently choose not to use such devices at all (Burger et al., 2007; Postema et al., 2016b).
In summary, although research in the United States is lacking, international studies suggest that male sex, younger age, medium or high level of education, prosthesis use, good general health (fewer comorbidities), and positive attitude are predictors of work participation among upper-limb amputees (Fernandez et al., 2000; Millstein et al., 1985; 1986; Postema et al., 2016b; Raichle et al., 2008). Importantly, issues of prosthesis reliability and the need for repair also impact device use in the work environment and may contribute to work interruptions.
Prosthesis Wear-Time
Few statistics on prosthesis wear-time are available. Unlike a lower-limb prosthesis that is often required for ambulation, an upper-limb prosthesis can be donned and used when necessary, either for a small portion of the day or for the entire day. However, a few studies do report on the number of individuals studied who use an upper-limb prosthesis daily. An Israeli study of 42 people with upper-limb amputation fit with a combination of body-powered, myoelectric, and passive devices found that 21 (50 percent) used the prosthesis daily, 9 (21.43 percent) used it intermittently, and 12 (28.57 percent) did not use it at all (Dudkiewicz et al., 2004). Millstein and colleagues (1986) found higher daily usage results: 89 percent of below-elbow amputees, 76 percent of above-elbow amputees, and only 60 percent of higher-level amputees. Raichle and colleagues (2008) surveyed 107 people with upper-limb amputation and found that having a
proximal amputation was related to greater use in terms of hours per day, while having a distal amputation was associated with greater use in terms of days per month.
Prosthesis Acceptance and Rejection
In addition to issues of comfort and body image discussed earlier, the prevalence of prosthesis use is associated with the level of amputation, loss of the dominant versus nondominant hand, bilateral versus unilateral amputation, and time since amputation. The term rejection is used in this chapter because it encompasses but is broader than abandonment. In some cases, amputees reject use of a certain type of prosthesis from the start, which is different from abandonment of a device later. Primary rejection rates (rejecting any use of a prosthesis) appear to be related to the level of amputation, age at the time of amputation, gender, and discrepancies between perceived needs and the availability of prosthetic devices that will meet those needs (Burger and Marinček, 1994; Dougherty et al., 2010; McFarland et al., 2010; Østlie et al., 2012a). Additionally, a large proportion of people who use upper-limb prostheses use a combination of devices (body-powered, myoelectric, and cosmetic), depending on their activities and goals (Crandall and Tomhave, 2002). And as noted earlier, even when functional devices are provided, many individuals may still choose to wear a device only for cosmetic purposes (Burger and Marinček, 1994).
Amputation Level
Rates of rejection of prosthesis use for people with upper-limb amputation range from 30 to 80 percent, with the rates typically being lowest for those with transradial amputations (Tintle et al., 2010; Wright et al., 1995) and highest for those with transhumeral or shoulder-level amputations as well as partial hand amputations (Burger and Marinček, 1994; Kruger and Fishman, 1993; Østlie et al., 2012a; Silcox et al., 1993). Research by Biddiss and Chau (2007a,b) supports the finding that individuals with a more proximal level of amputation compared with those with a more distal level of amputation are more likely to reject use of a prosthetic device. Rejection of prosthesis use in people with shoulder- or transhumeral-level amputations likely reflects the increased weight of the device, increased energy expenditure necessary to control the device, and more limited ability to improve function compared with prosthetic devices for transradial-level amputations (Østlie et al., 2012a). Higher rejection rates in people with partial hand amputations likely reflect the limited ability of partial hand prostheses to restore grasp ability adequately or to improve function
compared with use of the limb without a device (Burger and Marinček, 1994; Burger et al., 2007).
Loss of Dominant Versus Nondominant Hand and Bilateral Versus Unilateral Amputation
Findings are mixed with respect to loss of the dominant or nondominant hand. Some studies have found increased wear-time when the dominant hand has been amputated (Burger and Marinček, 1994; Dudkiewicz et al., 2004; Hacking et al., 1997), while others have found little correlation been loss of the dominant hand and prosthetic use (Gaine et al., 1997).
The most thorough report of bilateral usage is a survey of 242 individuals, 15 percent of whom had bilateral amputations. Overall, those with unilateral and bilateral acquired amputation had similar rejection rates. However, those with congenital bilateral limb absence had significantly higher rejection rates (75 percent) than those with congenital unilateral absence (28 percent) (p = 0.004) (Biddiss and Chau, 2007a).
Time Since Amputation
Multiple studies have found that early prosthetic fitting and rehabilitation positively impacted prosthesis success (Biddiss and Chau, 2007b, 2008; Gaine et al., 1997; Kejlaa, 1993; Malone et al., 1984; Roeschlein and Domholdt, 1989). And in a clinical review of 23 cases involving traumatic amputation, none who were fit after 12 weeks returned to gainful employment (Gaine et al., 1997).
Age
Some studies have found that age is not a factor in prosthesis success (Burger and Marinček, 1994; Hacking et al., 1997; Roeschlein and Domholdt, 1989; Wright et al., 1995). Conversely, others have found an increased risk of rejection among the elderly (Biddiss and Chau, 2007a,b; Østlie et al., 2012a) and at certain life stages (adolescence and early adulthood) (Biddiss and Chau, 2007a).
Gender
Surveys generally have found an association between increased prosthesis rejection rates and gender, with women more likely to reject the devices (Kyberd and Hill, 2011; Østlie et al., 2012a), although other research has not found this relationship (Raichle et al., 2008). Secondary rejection of
prosthetic devices (rejection after trial use) is associated with female gender and proximal upper-extremity amputation.
Level of Education
Level of education has not been associated with prosthesis use among individuals with upper-limb amputation (Raichle et al., 2008).
Perceived Needs
Individuals who wear their prostheses generally express satisfaction with their devices, while those who reject their devices express dissatisfaction (Biddiss et al., 2007). Addressing user desires therefore has the potential to reduce rejection rates. The most important aspects of prosthetic design that users consistently desire are comfort and reduced weight (Atkins et al., 1996; Biddiss and Chau, 2007c; Biddiss et al., 2007; Kyberd and Hill, 2011). Yet, while most users desire a device that is lighter-weight, that desire often is in conflict with the desire for more function (see the discussion of device taxonomy earlier in this chapter).
Various surveys of prosthesis users reveal similar user desires but no single design factor that would address the functional needs of all users (Kyberd and Hill, 2011). In general, body-powered prosthesis users desire additional wrist movement and better control mechanisms, as well as better cables and harness comfort (Atkins et al., 1996). Users of myoelectric devices desire components that have additional degrees of freedom (e.g., powered wrist movement, additional grips) and that are more durable, quieter, lighter-weight, and more cosmetic, with an improved control system and longer-lasting batteries (Atkins et al., 1996; Kyberd and Hill, 2011). Users also identify glove durability, improved sensory feedback, and increased dexterity as design priorities for myoelectric devices (Biddiss et al., 2007).
Overall, given the advantages and disadvantages of the various prosthetic devices, trade-offs are entailed in the prescription and fitting of the devices. Therefore, a key component of acceptance of a prosthetic device will be careful consideration of the individual’s needs and goals and whether the device meets the expectations of the user (Burger and Marinček, 1994; Østlie et al., 2012a).
EVALUATION AND MONITORING
The need for a multidisciplinary team approach is acknowledged in the first evidence-based clinical practice guidelines for the rehabilitation of persons with upper-limb amputation, released in 2014 (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014). Nonetheless,
there are still no clear universally accepted guidelines for prosthesis prescription or prosthetic training. Given the myriad factors that influence the type of device prescribed and the type and amount of prosthetic training received, a comprehensive clinical assessment by a trained (multidisciplinary) clinical team (including physician(s), occupational and physical therapists, experts in prosthetics and orthotics) can help in assessing the appropriateness and readiness for use of an upper-limb prosthesis, and it can guide the prescription of an appropriate device or (devices) and a training program to meet an individual’s needs. It is important to remember that the team centers around the individual, his or her capacity and needs. Therefore, this comprehensive assessment, performed in the outpatient setting during the preprosthetic phase, ideally will incorporate the following components that can impact prosthesis use and outcomes: current health status, current function and future functional goals and preferences, pain, social and cultural context, residual limb assessment, contralateral limb and trunk assessment, neurologic assessment, behavioral and cognitive assessment, lifestyle and occupational demands, and insurance status. Such an assessment, however, is frequently not available (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014).
Lifelong Care
A person with an upper-limb amputation requires lifelong care, including at least annual rechecks with the clinical team to ensure both that his or her needs are being met by the rehabilitation process and the device and that functional abilities have been optimized (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014). That said, there are no data on the prevalence of this practice pattern, and it is not clear that persons with amputation have access to regular annual amputation care. Studies on the frequency of upper-limb prosthetic replacements suggest that persons with upper-limb amputation receive a replacement device every 1 to 5 years (Blough et al., 2010; Dudkiewicz et al., 2004; Etter et al., 2014). A single study showed that provision of a new prosthesis was associated with an immediate decline in function (likely due to the acclimation required to become familiar with a new device), which could be remedied by prosthetic training (Dromerick et al., 2008). Indeed, it is widely recognized that prosthetic training needs to occur whenever a person with amputation, even an experienced prosthesis user, receives a new type of device (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014). However, experienced prosthesis users may require fewer training sessions than inexperienced users to learn to use a new type of device (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014).
Considerations for Prescription of Body-Powered and Myoelectric Devices
A recent systematic review of the literature (Carey et al., 2015) found that evidence was insufficient to determine the functional superiority of myoelectric versus body-powered prostheses. Each type of device has its merits and drawbacks. Body-powered devices are generally more robust and reliable, can be used in wet or dirty environments, and may require shorter training periods. In contrast, research suggests that myoelectric devices provide better cosmesis, improve phantom-limb pain, and are more accepted for light-intensity work (Carey et al., 2015). However, myoelectric devices require more training to use, are more prone to breakage, may become unreliable in hot and humid environments and in cases of weight fluctuation, and cannot be used in a wet environment. In a long-term follow-up study of pediatric patients fit at one center, a majority of the patients were provided with both myoelectric and body-powered devices, as well as a passive cosmetic prosthesis. A passive cosmetic hand was the prosthesis of choice for 44 percent of the individuals, and only 15 percent chose the myoelectric device (Crandall and Tomhave, 2002). In this study, 41 percent of those who used a prosthesis used multiple types of devices on a daily basis. Likewise, a survey of 50 service members from the Operation Enduring Freedom/Operation Iraqi Freedom era with unilateral upper-limb amputation, all of whom were provided with three types of devices (passive, body-powered, and myoelectric) within 1 year of their amputation, found that many used more than one type of device (McFarland et al., 2010).
TRAINING AND ADAPTATION
Therapy services and a team approach to amputation care are necessary throughout all phases of prosthetic rehabilitation (Atkins, 2004; Bowers, 2004; MaGuire, 2008; Management of Upper Extremity Amputation Rehabilitation Working Group, 2014; Resnik et al., 2012). Prosthetic training can improve skill in prosthesis use and help those with upper-limb amputation make better functional use of their prostheses (Atkins, 2004; Management of Upper Extremity Amputation Rehabilitation Working Group, 2014; Silcox et al., 1993). Multiple studies have found an association between long-term prosthetic use and receipt of prosthetic training (Biddiss and Chau, 2007a; Davids et al., 2006; Egermann et al., 2009), in particular, individualized and “sufficient” prosthetic training (Østlie et al., 2012a,b). In contrast, other studies have found that the quality or amount of prosthetic training was not strongly associated with prosthesis use (Biddiss and Chau, 2007b; Burger and Marinček, 1994; Hacking et al., 1997; Silcox et al., 1993). This finding is consistent with that of prior research showing that lack of technical skill in using a myoelectric prosthesis
in and of itself is not predictive of prosthesis rejection (Herberts et al., 1980). It is likely that other factors, such as the comfort and fit of the device, are more influential than skill in use in determining device adoption.
The source of most evidence on the recommended type and amount of training in use of upper-limb prostheses is expert opinion (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014), with training protocols being published largely in seminal textbooks (Atkins, 2004; Dillingham, 1998) and a handful of peer-reviewed papers (Johnson and Mansfield, 2014; Smurr et al., 2008) and available through prosthetic device manufacturers. Several recent papers focus on training for new special segments of the population, including those who have undergone targeted muscle reinnervation (Stubblefield et al., 2009), those using EMG pattern recognition control (Powell and Thakor, 2013; Powell et al., 2014; Scheme and Englehart, 2011; Simon et al., 2012), and those using the newly FDA-approved DEKA arm (Resnik et al., 2014b). Although training in prosthesis use is widely recognized as a key component of amputation rehabilitation, the relative value of intensive training or of specific training protocols for users of body-powered or myoelectric devices has not been well established (Davids et al., 2006).
The recently published comprehensive evidence-based clinical care guidelines for amputation rehabilitation (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014) describe four phases of rehabilitation for the upper-limb amputee: perioperative care, preprosthetic training, prosthetic training, and lifelong care (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014). Lifelong care was discussed above; the following sections focus on two other phases that are particularly pertinent to this report—preprosthetic and prosthetic training.
Preprosthetic Training
The preprosthetic training phase begins after acute wound healing, but it may begin even prior to amputation, once the decision to amputate has been made. This phase focuses on the skills needed to be independent in basic self-care activities, without a prosthesis. One-handed skills training also includes techniques to minimize overuse injuries of the intact limb and to prevent changes in posture that may occur as a result of upper-limb loss. If the dominant hand has been lost, training to alter hand dominance, including handwriting training, may also occur, since the prosthesis will only act as an assist. Additionally, preprosthetic training typically involves training in care of the residual limb (e.g., hygiene, shaping, desensitization, scar management) and development of an exercise program to maintain normal range of movement and to increase muscle strength that may be needed for
operation of a prosthesis. If myoelectric controls are anticipated, myosite training may begin in this phase.
Prosthetic Training
The average amount of time needed to train patients with upper-limb amputation varies by level of amputation, type of device, and such other factors as the presence of comorbid conditions. The optimal frequency of training visits, the intensity and duration of training, and the time needed to acclimate fully to a new prosthesis have not been studied. Some have suggested that persons with transradial amputation require, on average, 3-5 weeks of training (Dakpa and Heger, 1997); others have suggested that 5 hours of training is sufficient for people with transradial amputation, 10 hours for those with transhumeral amputation, and 12-20 hours for those with bilateral amputation (Atkins, 2004); and still others have suggested that gaining proficiency can require from a few days to several months (Johnson and Mansfield, 2014). Although the committee suspects that skills in prosthesis use may improve over time, with greater experience, no studies evaluating the length of time required to become a fully proficient, “expert” prosthesis user could be found. Although some detailed protocols are available in the professional and scientific literature (Atkins, 2004; Resnik et al., 2014c; Smurr et al., 2008), few studies have examined the effectiveness of specific approaches to prosthetic training. Given the small numbers of upper-limb amputees, most of these studies were conducted with able-bodied participants instead (Bouwsema et al., 2008, 2010a, 2014; Clingman and Pidcoe, 2014; Lake, 1997). One such study demonstrated that those with 8 hours of training on a prosthesis simulator had better performance than those who were not trained (Lake, 1997).
The current evidence-based guidelines state that prosthetic training should include the following components: education, controls training, and functional training (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014). Education includes instruction in putting on and taking off the prosthesis, caring for the residual limb, caring for and understanding the components of the prosthesis, and safely using the device. Generally speaking, most people require an adjustment period to acclimate to wearing a prosthesis and to learn to use it before they can wear the device full-time or for extended periods. Users need to acclimate to wearing a prosthesis through a graduated wear schedule (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014), and the fit and function of the device must be reassessed periodically during prosthetic training. Prosthetic fit (i.e., the socket and harnessing) may need to be modified by the prosthetist to address such issues as pain in the residuum, skin breakdown, or changes in socket fit and comfort.
Controls training teaches the patient how to operate each aspect of the prosthesis while avoiding unnecessary compensatory movements and using good body mechanics. Users of body-powered devices must learn the movements needed to operate the harness and to position any movable joints (e.g., wrist rotators, elbow) manually, while users of myoelectric devices must learn to isolate and control specific muscles and, in some instances, switch between modes of control. Several studies of able-bodied subjects have examined the content of prosthetic controls training and found that use of a myoelectric trainer improved myoelectric signal control (Clingman and Pidcoe, 2014). However, a recent study of able-bodied subjects found no differences in skill acquisition among three approaches to myoelectric controls training: use of a prosthesis simulator, a virtual prosthesis on a computer screen, or an isolated prosthetic hand (Bouwsema et al., 2010a). Nevertheless, use of a virtual trainer has become a widely accepted method of myoelectric controls training (Johnson and Mansfield, 2014).
Controls training typically includes repetitive drills to enable the user to practice specific prosthesis actions and commonly used movements, such as grasp and release of objects. Preliminary evidence gathered on able-bodied subjects suggests that the structure of the training approach (blocked practice versus random practice of skills) does not impact the rate of acquisition of prosthetic skills (Bouwsema et al., 2008). A study of four approaches to grasp training for users of myoelectric prostheses recommends that training programs begin with indirect grasping tasks but ultimately emphasize fine motor tasks (Bouwsema et al., 2014).
Functional skills training is designed to help individuals integrate use of their prostheses into their everyday tasks and to teach them strategies for maximizing their functional abilities. Because most prostheses lack movable wrists and have limited grasp positions, functional training often focuses on teaching prepositioning of the device in a functional position prior to utilization. Functional training typically progresses from using the prosthesis in unilateral activities to engaging it during bimanual activities. Ultimately, training should advance to engagement in community activities to increase the user’s confidence in utilizing the device in public and in real-world situations (Johnson and Mansfield, 2014).
ACCESS AND AVAILABILITY
Access to and Availability of Devices
Data are scarce on the prevalence of use or incidence of prescription of upper-limb prostheses across the United States, largely because no single nationally representative source of data contains this information. Thus, most studies on the prevalence of use of these devices, as well as the rates
of prosthetic prescription and repair, have used self-report surveys. Results of such surveys suggest that persons with major upper-limb amputation receive a new device every 1-5 years (Blough et al., 2010; MacKenzie et al., 2007).
A recent, large study on prosthesis prescription patterns in the Veterans Health Administration (VHA) used 10 years of data from the VHA’s National Prosthetic Patient Database to calculate annual prescription and repair rates by level of amputation and type of device (Etter et al., 2014). The rate of prescription of upper-limb prostheses was 0.20/year for persons with shoulder-level amputation, 0.27/year for those with transhumeral amputation, and 0.26/year for those with transradial amputation (Etter et al., 2014). These data suggest that overall, veterans with major upper-limb amputation received a new prosthesis once every 3.6 years. The prescription rate for myoelectric devices, however, was 0.38/year compared with 0.23/year for body-powered devices, indicating that the former devices were being prescribed or replaced more frequently than the latter. Repair rates varied by level of amputation, with the least frequent repairs provided for persons with shoulder-level amputation (0.02/year) and more frequent repairs for those with transhumeral and transradial amputations (0.26/year and 0.21/year, respectively). Furthermore, compared with veterans over age 65, those under age 65 had higher incidence rates of both prescription of new devices and repair of existing devices. The results of this study are limited to the prescription and repair patterns within the VHA and likely are not generalizable beyond that system of care.
It should be noted that issues with the reliability of a prosthesis may impact vocational use. Particularly with myoelectric devices, which need more frequent repairs/replacement, not having a back-up device can be a barrier to successful participation at work.
As described in Appendix C and Chapter 3, the committee examined data for a 5 percent random sample of beneficiaries from the Medicare Durable Medical Equipment (DME) files for the years 2013-2014 to gain information on the frequency with which UEPs were prescribed within the 20- to 67-year-old population of beneficiaries. Although the population represented in the Medicare data is not necessarily representative of the overall population using UEPs, these data were available to the committee. The 5 percent sample is commonly used in research as the 100 percent dataset is so large that its use for research purposes is not feasible. Because of the underrepresentation of the 20- to 67-year-old cohort in Medicare claims data and the relatively low frequency of UEP claims, the 5 percent sample yielded a small study sample, which limited the presentation of granular information based on individual-level characteristics. Use of a 20 percent random sample and/or more years of data would improve future analyses. The Medicare DME data were linked to data from the Master Beneficiary
files to obtain information on the demographic and clinical characteristics of device recipients. (See Appendix C for a detailed description of methods.) The committee identified all upper-limb prostheses provided by Medicare to persons aged 20-67 and then categorized the types of devices provided, as well as the characteristics of device recipients. Given the small sample size (fewer than 150 persons) and the Centers for Medicare & Medicaid Services’ (CMS’s) restrictions on reporting values for categories of 20 or fewer persons, the analyses reported here show proportions rather than absolute values.
Of the prosthetic components and devices provided to the 5 percent random sample of Medicare beneficiaries aged 20 to 67 in 2013, 43 percent were classified as body-powered, 27.6 percent as myoelectric, and the remainder as other (see Table 4-1). Table 4-2 shows the demographic characteristics of those Medicare recipients who received a device, including age, sex, and race/ethnicity. Most recipients were under age 65, suggesting that these were individuals with long-term disability. The majority of recipients were male (58.2 percent) and white (68.4 percent). Table 4-3 shows the distribution of amputation diagnoses among this sample. More than half (54.3 percent) had amputation at the transradial level; 23.9 percent had amputation at the transhumeral level; almost 20 percent had unspecified amputation diagnoses; and only 2.2 percent had partial hand amputation.
Medicare and most state Medicaid agencies and private insurance companies utilize the CMS DME, Prosthetics/Orthotics, and Supplies fee schedules for all procedure codes. In contrast with most medical care, time typically is not billed separately. Instead, the billing of a code (L–code in the case of prosthetic devices) is assumed to include the time needed to evaluate, fit, deliver, and follow up on the given device. Later modifications, adjustments, and repairs past the warranty period may be billed
TABLE 4-1
Upper-Limb Prostheses Provided to Beneficiaries by Type
| Type of Device | Percentage | Cumulative Percentagea |
|---|---|---|
| Body-Powered | 42.9 | 42.9 |
| Myoelectric | 27.6 | 70.4 |
| Other | 29.6 | 100.0 |
NOTE: Information from 5 percent random sample of 2013–2014 Durable Medical Equipment (DME) Medicare beneficiaries aged 20–67.
a Percentages may not total because of rounding.
SOURCE: DME Research Identifiable File, Centers for Medicare & Medicaid Services.
TABLE 4-2
Demographic Characteristics of Recipients of Upper-Limb Prostheses
| Category | Percentage | Cumulative Percentagea |
|---|---|---|
| Age | ||
|
20–45 |
20.4 | 20.4 |
|
46–55 |
25.5 | 45.9 |
|
56–64 |
26.5 | 72.4 |
|
65–67 |
27.6 | 100.0 |
| Sex | ||
|
Male |
58.2 | 58.2 |
|
Female |
41.8 | 100.0 |
| Race/Ethnicity | ||
|
White |
68.4 | 68.4 |
|
Black |
18.4 | 86.7 |
|
Hispanic |
8.2 | 94.9 |
|
Other |
5.1 | 100.0 |
NOTE: Information from 5 percent random sample of 2013–2014 Durable Medical Equipment (DME) Medicare beneficiaries aged 20–67.
a Percentages may not total because of rounding.
SOURCE: DME Research Identifiable File, Centers for Medicare & Medicaid Services.
TABLE 4-3
Estimated Percentage of Upper-Limb Prosthetic Devices by Amputation Category
| Amputation Category | Percentage | Cumulative Percentagea |
|---|---|---|
| Transradial/wrist disarticulation | 54.3 | 54.3 |
| Transhumeral | 23.9 | 78.3 |
| Level/side unspecified | 19.6 | 97.8 |
| Partial hand | 2.2 | 100.0 |
NOTE: Information from 5 percent random sample of 2013–2014 Durable Medical Equipment (DME) Medicare beneficiaries aged 20–67.
a Percentages may not total because of rounding.
SOURCE: DME Research Identifiable File, Centers for Medicare & Medicaid Services.
as repair time and parts. When devices and components are provided for patients through private payment or workers’ compensation insurance, payment can vary widely based on the selected codes and provider customary fee schedules, which may or may not cover the cost of the components. Medicare fee schedules are shown in Annex Table 4-1; however, they may not be representative of practice outside of Medicare. The existence of a device-specific code does not guarantee coverage, which, depending on the source of coverage, may require preauthorization or predetermination prior to delivery. Additionally, some recommended items may be noncoded, noncovered items to which no specific fee has been assigned.
Medicare does not provide preauthorization and instead may later request documentation to justify medical necessity. If the claim of medical necessity is not met, a claim will not be paid to a provider. In some situations, the provider anticipates that a device may be determined not medically necessary and therefore may be denied, and so they may ask a patient to sign an advance beneficiary notice (ABN). The ABN allows the patient to make an informed decision about whether to receive an item or service that may be determined noncoverable by Medicare and to accept financial responsibility should that be the case. An example is a device or parts “not otherwise specified” (code L7499), which includes any new technology. The challenge in these cases is that the new technology may be very expensive (>$10,000). A clinical facility, especially a smaller one that is less able to absorb the cost of an unpaid device or less confident in the justification for a complex upper-limb device, may be very concerned about assuming the risk of purchasing and delivering such an item for which payment is not assured. Likewise, a patient may be very hesitant to accept financial responsibility in case Medicare does not. In addition, Medicare, as well as other insurance providers, requires a copayment for service, which may represent a significant financial burden for individuals without secondary insurance coverage.
The committee recognizes that limited or lack of evidence about the impact of upper-limb loss, prosthesis use, and amputation rehabilitation on activity and participation may affect decisions by funding sources about which devices and services to cover. Such information on outcomes could contribute to studies on the effectiveness or cost-effectiveness of different types of prostheses and help inform the development of rational resource utilization, including use by insurers and other funding sources to inform their coverage decisions.
Distribution of Experienced and Knowledgeable Clinicians
It is widely recognized among those who work frequently with individuals with upper-limb amputation that this population is best served by knowledgeable, trained clinical providers who work together as a team to
provide amputation care. Yet, while limited data are available on utilization of rehabilitation services in the U.S. population, one study examining practice patterns found that only 27 percent of the people who had a unilateral amputation and used a prosthesis had received training from an occupational or a physical therapist, and only 22 percent had received more than 10 hours of training from either a therapist or a prosthetist (Kestner, 2006).
Requirements for becoming a certified prosthetist/orthotist have evolved to an entry-level master’s degree from a school accredited by the Commission on Accreditation of Allied Health Education Programs. Students receive training in upper- and lower-limb prosthetic fittings. To achieve certification for each discipline, all students must also complete a 1-year residency (per discipline) working under a certified clinician at an American Board for Certification in Orthotics, Prosthetics and Pedorthics (ABC)-accredited residency location, followed by written and practical qualifying exams. The experience the student receives in upper-limb fittings will depend on the patients seen at the residency facility. Only 15 states have licensure for prosthetics and orthotics (ABC, 2017b). Those that do typically rely on the ABC certification examination as a requirement. There are approximately 14,000 ABC-certified clinicians (prosthetists, orthotists, and pedorthists) (ABC, 2017a).
Prosthesis users receive training from occupational or physical therapists, depending on the clinical environment and team. Both occupational and physical therapy training programs require training in upper-limb prosthetics as part of the accreditation process. However, there are no mandates regarding the extent or content of this training.
Given the relative rarity and geographic dispersion of persons with upper-limb amputation in the United States, it is difficult for physicians, prosthetists, and therapists to acquire expertise in working with this population. Although limited data on the workforce are available, a recent survey of ABC members found that only 150 of 1,145 prosthetist members (13 percent) would consider fitting a patient with a UEP (Stark, 2016). Nearly 72 percent of respondents to a survey of upper-limb practitioners categorized themselves as novices in upper-extremity prosthetics and indicated that they treated an average of three such patients per year. The 26.2 percent who considered themselves experts in the field treated an average of 25 such patients per year. Many prosthetists endorsed the idea that the fitting process for patients with upper-limb amputation is particularly complex and that few prosthetists are being educated in this practice specialty. The areas of greatest challenge in upper-limb prosthetics were identified as knowledge of componentry, patient training, patient variation, and functional adjustment.
Given the scarcity of clinicians, including physicians, prosthetists, and occupational therapists, who are specialists in caring for persons with
upper-limb amputation, a skilled team approach to care may not be widely available across the United States. Because the typical clinician has minimal education specific to upper-limb amputation and may see very few of these patients over the course of his or her career (Alley, 2004), most have little opportunity to develop expertise with this patient population (Stark, 2016). In addition to having limited experience in prosthetic fitting, clinicians would also have limited experience in justifying the need for and benefits of certain devices to insurers, which also could negatively impact delivery and outcomes. Because of their lack of expertise, many prosthetists rely on consultations with upper-limb experts to treat their patients. Some clinicians and patients must travel long distances to deliver or receive care. This necessity may delay care and negatively impact outcomes (Stark, 2016).
In 2007, the VHA launched an Amputation System of Care (ASoC) to improve access to expertise and specialized training for veterans with amputation (VA, 2016). Seven geographically dispersed VHA medical centers were designated as Regional Amputation Care Centers (RACs). RACs provide interdisciplinary care and prosthetic fabrication facilities, and they serve as resources to other VHA medical centers by offering telerehabilitation, consultation, and education. In addition, 18 VHA sites were designated Polytrauma/Amputation Network Sites (PANS), which offer inpatient and outpatient amputation rehabilitation services and have prosthetic labs. The remaining VHA medical centers were designated Amputation Care Centers. These centers have interdisciplinary amputation care teams but may lack the resources of RACs or PANS; or they may be designated amputation points of contact for assessment and refer patients to sites with requisite resources. The goal of the ASoC is to offer a coordinated system of high-quality, multidisciplinary, lifelong care that can provide the “most advanced and appropriate prosthetic components to Veterans across the system of care regardless of geographic location” (VA, 2012).
FINDINGS AND CONCLUSIONS
Findings
Overview, Taxonomy, Prosthetic Components, Prosthetic Sockets
4-1. A variety of different types of upper-extremity prostheses (UEPs) are available, the major categories being cosmetic, body-powered, hybrid, and myoelectric devices. There also exist a range of socket interfaces, suspension methods, terminal devices, and other components for each of these categories.
4-2. Regardless of the type of prosthetic device used, a well-fitting and comfortable socket is essential to successful use of a prosthesis.
4-3. For some people with limb loss, cosmetic restoration is highly valued or even preferred over functional restoration because of its mitigating effect on the disruption to body image.
4-4. The complex function of an upper extremity is far more difficult to replace with a prosthesis relative to the function of a lower extremity.
Impairment Mitigation
4-5. Upper-extremity limb loss or deficiency results in a wide range of significant impairments to body functions and resulting limitations on activities and participation.
4-6. Currently available UEPs cannot replace the complex functions of the missing limb because of limitations inherent in their control and design, their lack of sensory feedback, and the methods required to suspend them onto the residual limb.
4-7. The extent of impairment mitigation provided by a prosthesis varies by the type of device, the level of amputation, and the user’s training in using the device.
4-8. Relative to the upper extremities, the primary functions of the lower limbs are more limited and concern primarily maintenance and achievement of upright stance and various types of locomotion (walking, running, hopping, jumping, stair climbing). In contrast, the primary functions of the upper extremities include self-care, interaction with the environment and others, self-expression, and fine and gross motor activities.
Acceptance/Rejection of Prostheses and Factors Affecting Device Use
4-9. The fit and function of the prosthesis and thus its impairment-mitigating effects may be impacted by environmental factors that may change over time, such as exposure to moisture, heat, and dirt. The consistency of impairment mitigation also depends on the condition and volume of the residual limb, which impact socket fit and comfort.
4-10. Skin integrity and a well-fitting prosthetic socket are key factors for successful use of a UEP.
4-11. People with transradial amputations are more likely to use prosthetic devices relative to those whose amputations are either more proximal (transhumeral or shoulder-level) or more distal (partial hand amputations).
4-12. Overall rejection rates of currently available types of UEPs are high. Understanding of all factors related to successful prosthesis adoption is limited, although primary rejection rates appear to be related to
comfort of the prosthetic socket, level of amputation, age at the time of amputation, gender, and discrepancies between users’ perceived needs and the availability of prosthetic devices to meet these needs.
4-13. Data on durability and repair rates for UEPs are limited, but clinical experience indicates that these devices require frequent maintenance and repair that can interfere with their consistent use.
Prognosis for Occupational Success
4-14. The extent of impairment mitigation provided by a prosthesis varies by the type of device and the level of amputation. In addition to design selection, impairment mitigation is dependent on socket fit, which may change in various environments and over time.
4-15. The only U.S. data available on vocational reintegration following upper-extremity amputation come from the military health care system. The committee could find no other studies examining the impact of prosthetic devices and rehabilitation strategies on work participation in the United States.
4-16. International studies and studies of U.S. military personnel demonstrate that a significant percentage of people with acquired limb loss have difficulty returning to work at all, and those who do usually require job modifications and adaptations to be successful.
4-17. International studies suggest that use of UEPs is a predictor of work participation and that employment is associated with decreased prosthesis rejection rates.
Access and Availability
4-18. Users need training to utilize UEPs effectively. Prosthetic training begins in the preprosthetic phase and continues upon device acquisition.
4-19. Persons with amputation require lifelong care with annual rechecks to ensure that their prosthetic needs are being met.
4-20. Recent evidence-based clinical practice guidelines (Management of Upper Extremity Amputation Rehabilitation Working Group, 2014) acknowledge the importance of taking a multidisciplinary team approach to the rehabilitation of people with upper-limb amputation.
4-21. There are no universally accepted guidelines for prescription of prosthetics and training in their use.
4-22. Data on the prevalence of use and prescription of UEPs are fragmented and limited.
4-23. Within the United States, people with limb loss or limb deficiency experience significant barriers to accessing and successfully using prosthetic devices.
4-24. Medicare and other insurers may reject payment for devices and components that are new technologies or that they do not consider “medically necessary” even if prescribed by a trained professional.
4-25. Evidence suggests that prosthesis users may not receive adequate training in using their devices.
4-26. Data do not exist on the location and distribution of expertise, such as centers of excellence, in the care of persons with upper-limb amputation across the United States. There is no standardization for UEP centers of excellence except in the Veterans Health Administration (VHA), where access to services is limited to veterans.
Conclusions
Overview, Taxonomy, Prosthetic Components, Prosthetic Sockets
4-1. The type of UEPs provided, including the choice of terminal device and socket design, needs to be customized to meet individual needs. [Findings 4-1, 4-2, 4-3]
Impairment Mitigation
4-2. Despite advances in prosthetic designs and research, currently available UEPs are limited in their ability to mitigate impairments related to limb loss. [Findings 4-4, 4-5, 4-6, 4-8]
4-3. Many individuals would benefit from more than one type of prosthetic device and/or terminal device to mitigate their impairments. [Findings 4-1, 4-4, 4-8]
4-4. In the future, emerging technology, if made available to people with limb loss, may improve the ability of UEPs to mitigate impairments. [Findings 4-6, 4-9]
Acceptance/Rejection of Prostheses and Factors Affecting Device Use
4-5. Even for people who are able to obtain UEPs, rejection rates are high, in part because of discomfort with wearing the devices, limited ability of the devices to meet their needs, a lack of training in their use, and limited durability. [Findings 4-2, 4-4, 4-6, 4-7, 4-9 through 4-14, 4-18, 4-19, 4-25]
Prognosis for Occupational Success
4-6. In selected cases, UEPs could significantly improve the ability to work, depending on the specific job requirements and environmental and personal factors. [Findings 4-9, 4-11, 414, 4-16 through 4-18]
4-7. Comprehensive efforts to study the impact of upper-limb loss, prosthesis use, and amputation rehabilitation on activity and participation, including work participation, are needed. Such research may not only enhance knowledge in these areas but also inform the development of rational resource utilization, including informing cost-benefit analyses and coverage for devices and related services. [Findings 4-13, 4-15, 4-16, 4-22, 4-26]
Access and Availability
4-8. It is important that UEP prescriptions be generated by qualified providers and customized to individuals’ functional goals and clinical conditions. [Findings 4-1, 4-2, 4-7, 4-9, 4-10, 4-12, 414, 4-19, 4-20]
4-9. Reimbursement for UEPs and related rehabilitation services is highly variable, which creates unequal access, particularly for new technologies. [Finding 4-24]
4-10. The provision of UEPs is contingent largely on reimbursement policy rather than patient need. In many cases, a mismatch exists between the UEPs covered by Medicare and other insurers as medically necessary and the products or technologies that would best meet the needs of users to enhance their participation in life roles. [Finding 4-24]
4-11. There is a need for more qualified providers (including physicians, prosthetists, and therapists) and clinics with the knowledge, skill, and expertise to properly evaluate, prescribe, fit, and train people in the use of UEPs. [Findings 4-21, 4-25, 4-26]
4-12. Many civilian users of UEPs could benefit from the establishment of regional amputation centers of excellence incorporating a specialty multidisciplinary team rehabilitation approach, similar to those in the VHA amputation system of care. [Finding 4-26]
General Conclusion
4-13. At this time, it is difficult to quantify fully the impact of UEPs on impairment mitigation and employability because of a lack of research on contextual/environmental factors that impact device use and job function and a lack of data on occupational success. Even as UEPs have the potential to improve functional capacity in work
participation, their impact is limited by unequal access to the devices and training in their use.
REFERENCES
ABC (American Board for Certification in Orthotics, Prosthetics and Pedorthics). 2017a. Learn who we are. https://www.abcop.org/WhoWeAre/Pages/default.aspx (accessed January 5, 2017).
ABC. 2017b. State licensure. https://www.abcop.org/State-Licensure/Pages/state-licensure.aspx (accessed January 18, 2017).
Alley, R. D. 2004. The prosthetist’s evaluation and planning process with the upper extremity amputee. In Functional restoration of adults and children with upper extremity amputation. New York: Demos Medical Publishing, Inc. Pp. 125-132.
Alley, R. D., T. W. Williams, III, M. J. Albuquerque, and D. E. Altobelli. 2011. Prosthetic sockets stabilized by alternating areas of tissue compression and release. Journal of Rehabilitation Research & Development 48(6):679-696.
Aschoff, H. H., R. E. Kennon, J. M. Keggi, and L. E. Rubin. 2010. Transcutaneous, distal femoral, intramedullary attachment for above-the-knee prostheses: An endo-exo device. Journal of Bone & Joint Surgery 92(Suppl. 2):180-186.
Atkins, D. 2004. Prosthetic training. In Atlas of amputations and limb deficiencies: Surgical, prosthetic, and rehabilitation principles, edited by D. G. Smith and J. W. Michael. Rosemont, IL: American Academy of Orthopaedic Surgeons. Pp. 27-284.
Atkins, D. J., D. C. Y. Heard, and W. H. Donovan. 1996. Epidemiologic overview of individuals with upper-limb loss and their reported research priorities. Journal of Prosthetics and Orthotics 8(1):211.
Baschuk, C. 2016. Application of elevated vacuum suspension in upper-limb prosthetics. The Academy TODAY 12(3).
Belisle, J. G., J. C. Wenke, and C. A. Krueger. 2013. Return-to-duty rates among U.S. military combat-related amputees in the global war on terror: Job description matters. Journal of Trauma Acute Care Surgery 75(2):279-286.
Belter, J. T., J. L. Segil, A. M. Dollar, and R. F. Weir. 2013. Mechanical design and performance specifications of anthropomorphic prosthetic hands: A review. Journal of Rehabilitation Research & Development 50(5):599-618.
Belter, J. T., B. C. Reynolds, and A. M. Dollar. 2014. Grasp and force based taxonomy of split-hook prosthetic terminal devices. Conference of the IEEE Engineering in Medicine and Biology Society 2014:6613-6618.
Berning, K., S. Cohick, R. Johnson, L. A. Miller, and J. W. Sensinger. 2014. Comparison of body-powered voluntary opening and voluntary closing prehensor for activities of daily life. Journal of Rehabilitation Research & Development 51(2):253-261.
Biddiss, E., and T. Chau. 2007a. The roles of predisposing characteristics, established need, and enabling resources on upper extremity prosthesis use and abandonment. Disability Rehabilitation Assistive Technology 2(2):71-84.
Biddiss, E., and T. Chau. 2007b. Upper-limb prosthetics: Critical factors in device abandonment. American Journal of Physical Medicine & Rehabilitation 86(12):977-987.
Biddiss, E., and T. Chau. 2007c. Upper limb prosthesis use and abandonment: A survey of the last 25 years. Prosthetics and Orthotics International 31(3):236-257.
Biddiss, E., and T. Chau. 2008. Multivariate prediction of upper limb prosthesis acceptance or rejection. Disability Rehabilitation: Assistive Technology 3(4):181-192.
Biddiss, E., D. Beaton, and T. Chau. 2007. Consumer design priorities for upper limb prosthetics. Disability Rehabilitation: Assistive Technology 2(6):346-357.
Blough, D. K., S. Hubbard, L. V. McFarland, D. G. Smith, J. M. Gambel, and G. E. Reiber. 2010. Prosthetic cost projections for service members with major limb loss from Vietnam and OIF/OEF. Journal of Rehabilitation Research & Development 47(4):387-402.
Bosmans, J. C., J. H. Geertzen, W. J. Post, C. P. van der Schans, and P. U. Dijkstra. 2010. Factors associated with phantom limb pain: A 3 1/2-year prospective study. Clinical Rehabilitation 24(5):444-453.
Bouwsema, H., C. K. van der Sluis, and R. M. Bongers. 2008. The role of order of practice in learning to handle an upper-limb prosthesis. Archives of Physical Medicine and Rehabilitation 89(9):1759-1764.
Bouwsema, H., C. K. van der Sluis, and R. M. Bongers. 2010a. Learning to control opening and closing a myoelectric hand. Archives of Physical Medicine and Rehabilitation 91(9):1442-1446.
Bouwsema, H., C. K. van der Sluis, and R. M. Bongers. 2010b. Movement characteristics of upper extremity prostheses during basic goal-directed tasks. Clinical Biomechanics (Bristol, Avon) 25(6):523-529.
Bouwsema, H., P. J. Kyberd, W. Hill, C. K. van der Sluis, and R. M. Bongers. 2012. Determining skill level in myoelectric prosthesis use with multiple outcome measures. Journal of Rehabilitation Research & Development 49(9):1331-1348.
Bouwsema, H., C. K. van der Sluis, and R. M. Bongers. 2014. Changes in performance over time while learning to use a myoelectric prosthesis. Journal of Neuroengineering and Rehabilitation 11(1):16.
Bowers, R. 2004. The team approach to amputee rehabilitation. Senior Step 1.
Branemark, R., P. I. Branemark, B. Rydevik, and R. R. Myers. 2001. Osseointegration in skeletal reconstruction and rehabilitation: A review. Journal of Rehabilitation Research & Development 38(2):175-181.
Burger, H., and Č. Marinček. 1994. Upper limb prosthetic use in Slovenia. Prosthetics and Orthotics International 18(1):25-33.
Burger, H., and G. Vidmar. 2016. A survey of overuse problems in patients with acquired or congenital upper limb deficiency. Prosthetics and Orthotics International 40(4):497-502.
Burger, H., T. Maver, and Č. Marinček. 2007. Partial hand amputation and work. Disability and Rehabilitation 29(17):1317-1321.
Carey, S. L., M. Jason Highsmith, M. E. Maitland, and R. V. Dubey. 2008. Compensatory movements of transradial prosthesis users during common tasks. Clinical Biomechanics (Bristol, Avon) 23(9):1128-1135.
Carey, S. L., D. J. Lura, and M. J. Highsmith. 2015. Differences in myoelectric and body-powered upper-limb prostheses: Systematic literature review. Journal of Rehabilitation Research & Development 52(3):247-262.
Carlsen, B. T., P. Prigge, and J. Peterson. 2014. Upper extremity limb loss: Functional restoration from prosthesis and targeted reinnervation to transplantation. Journal of Hand Therapy 27(2):106-113.
Chaubey, P., T. Rosenbaum-Chou, W. Daly, and D. Boone. 2014. Closed-loop vibratory haptic feedback in upper-limb prosthetic users. Journal of Prosthetics and Orthotics 26(3):120-127.
Chicoine, C. L., A. M. Simon, and L. J. Hargrove. 2012. Prosthesis-guided training of pattern recognition-controlled myoelectric prosthesis. Conference of the IEEE Engineering in Medicine and Biology Society 2012:1876-1879.
Cipriani, C., M. Controzzi, and M. C. Carrozza. 2011. The smarthand transradial prosthesis. Journal of Neuroengineering and Rehabilitation 8:29.
Cipriani, C., M. D’Alonzo, and M. C. Carrozza. 2012. A miniature vibrotactile sensory substitution device for multifingered hand prosthetics. IEEE Transactions on Biomedical Engineering 59(2):400-408.
Clingman, R., and P. Pidcoe. 2014. A novel myoelectric training device for upper limb prostheses. IEEE Transactions and Neural Systems Rehabilitation Engineering 22(4):879-885.
Clippinger, F. W., R. Avery, and B. R. Titus. 1974. A sensory feedback system for an upper-limb amputation prosthesis. Bulletin of Prosthetics Research 247-258.
Copuroglu, C., M. Ozcan, B. Yilmaz, Y. Gorgulu, E. Abay, and E. Yalniz. 2010. Acute stress disorder and post-traumatic stress disorder following traumatic amputation. Acta Orthopaedica Belgica 76(1):90-93.
Cowley, J., L. Resnik, J. Wilken, L. Smurr Walters, and D. Gates. 2016. Movement quality of conventional prostheses and the DEKA Arm during everyday tasks. Prosthetics and Orthotics International 41(1):33-40.
Crandall, R. C., and W. Tomhave. 2002. Pediatric unilateral below-elbow amputees: Retrospective analysis of 34 patients given multiple prosthetic options. Journal of Pediatric Orthopaedics 22(3):380-383.
Cutosky, M. 1989. On grasp choice, grasp models, and the design of hand for manufacturing tasks. IEEE Transactions on Robotics and Automation 5(3):269-279.
Dabaghi-Richerand, A., F. Haces-Garcia, and R. Capdevila-Leonori. 2015. Prognostic factors of a satisfactory functional result in patients with unilateral amputations of the upper limb above the wrist that use an upper limb prosthesis. Revista Espanola de Cirugia Ortopedica y Traumatologia 59(5):343-347.
Dajpratham, P., S. Tantiniramai, P. Lukkapichonchut, and S. Kaewnaree. 2008. Factors associated with vocational reintegration among the Thai lower limb amputees. Journal of the Medical Association of Thailand 91(2):234-239.
Dakpa, R., and H. Heger. 1997. Prosthesis management and training of adult upper limb amputees. Current Orthopaedics 11(3):193-202.
Dalley, S. A., D. A. Bennett, and M. Goldfarb. 2012. Preliminary functional assessment of a multigrasp myoelectric prosthesis. Conference Proceedings of the IEEE Engineering in Medicine and Biology Society 2012:4172-4175.
Datta, D., K. Selvarajah, and N. Davey. 2004. Functional outcome of patients with proximal upper limb deficiency—acquired and congenital. Clinical Rehabilitation 18(2):172-177.
Davids, J. R., L. V. Wagner, L. C. Meyer, and D. W. Blackhurst. 2006. Prosthetic management of children with unilateral congenital below-elbow deficiency. Journal of Bone and Joint Surgery: Series A 88(6):1294-1300.
Davidson, J. 2004. A comparison of upper limb amputees and patients with upper limb injuries using the Disability of the Arm, Shoulder and Hand (DASH). Disability and Rehabilitation 26(14-15):917-923.
Davidson, J. H., K. E. Khor, and L. E. Jones. 2010. A cross-sectional study of post-amputation pain in upper and lower limb amputees, experience of a tertiary referral amputee clinic. Disability and Rehabilitation 32(22):1855-1862.
de Jong, I. G., H. A. Reinders-Messelink, K. Tates, W. G. Janssen, M. J. Poelma, I. van Wijk, and C. K. van der Sluis. 2012. Activity and participation of children and adolescents with unilateral congenital below elbow deficiency: An online focus group study. Journal of Rehabilitation Medicine 44(10):885-892.
Deeny, S., C. Chicoine, L. Hargrove, T. Parrish, and A. Jayaraman. 2014. A simple ERP method for quantitative analysis of cognitive workload in myoelectric prosthesis control and human-machine interaction. PLOS One 9(11):e112091.
Desmond, D. M. 2007. Coping, affective distress, and psychosocial adjustment among people with traumatic upper limb amputations. Journal of Psychosomatic Research 62(1):15-21.
Desmond, D. M., L. Coffey, P. Gallagher, M. MacLachlan, S. Wegener, and F. O’Keeffe. 2012. Limb amputation. In The Oxford handbook of rehabilitation psychology, 1st ed., edited by P. Kennedy. New York: Oxford University Press. Pp. 351-367.
Desteli, E. E., Y. Imren, M. Erdogan, G. Sarisoy, and S. Cosgun. 2014. Comparison of upper limb amputees and lower limb amputees: A psychosocial perspective. European Journal of Trauma and Emergency Surgery 40(6):735-739.
Dillingham, T. 1998. Rehabilitation of the upper limb amputee. In Textbook of military medicine part IV surgical combat casualty care: Rehabilitation of the injured combatant. Washington, DC: Office of the Surgeon General, U.S. Department of the Army. Pp. 33-77.
Doeringer, J. A., and N. Hogan. 1995. Performance of above elbow body-powered prostheses in visually guided unconstrained motion tasks. IEEE Transactions on Biomedical Engineering 42(6):621-631.
Donkervoort, M., D. J. Wiegerink, J. van Meeteren, H. J. Stam, and M. E. Roebroeck. 2009. Transition to adulthood: Validation of the Rotterdam Transition Profile for young adults with cerebral palsy and normal intelligence. Developmental Medicine and Child Neurology 51(1):53-62.
Dougherty, P. J., L. V. McFarland, D. G. Smith, A. Esquenazi, D. J. Blake, and G. E. Reiber. 2010. Multiple traumatic limb loss: A comparison of Vietnam veterans to OIF/OEF servicemembers. Journal of Rehabilitation Research & Development 47(4):333-348.
Dromerick, A. W., C. N. Schabowsky, R. J. Holley, B. Monroe, A. Markotic, and P. S. Lum. 2008. Effect of training on upper-extremity prosthetic performance and motor learning: A single-case study. Archives of Physical Medicine and Rehabilitation 89(6):1199-1204.
Dudkiewicz, I., R. Gabrielov, I. Seiv-Ner, G. Zelig, and M. Heim. 2004. Evaluation of prosthetic usage in upper limb amputees. Disability and Rehabilitation 26(1):60-63.
Egermann, M., P. Kasten, and M. Thomsen. 2009. Myoelectric hand prostheses in very young children. International Orthopaedics 33(4):1101-1105.
Englehart, K., and B. Hudgins. 2003. A robust, real-time control scheme for multifunction myoelectric control. IEEE Transactions on Biomedical Engineering 50(7):848-854.
Esquenazi, A. 2015. Upper limb amputation, rehabilitation & prosthetic restoration. In Current diagnosis & treatment: Physical medicine & rehabilitation, edited by I. B. Maitin and E. Cruz. New York: McGraw-Hill Education. Pp. 453-459.
Etter, K., M. Borgia, and L. Resnik. 2014. Prescription and repair rates of prosthetic limbs in the VA healthcare system: Implications for national prosthetic parity. Disability and Rehabilitation: Assistive Technology 10(6):1-8.
FDA (U.S. Food and Drug Administration). 2015. FDA authorizes use of prosthesis for rehabilitation of above-the-knee amputations. Silver Spring, MD: FDA.
Fernandez, A., I. Isusi, and M. Gomez. 2000. Factors conditioning the return to work of upper limb amputees in Asturias, Spain. Prosthetics and Orthotics International 24(2):143-147.
Fillauer Companies, Inc. 2016. Fillauer Companies product catalog. http://hosmer.com/products/PDFs/Fillauer%20Companies%20Catalog.zip (accessed January 24, 2017).
Fisher, K., R. S. Hanspal, and L. Marks. 2003. Return to work after lower limb amputation. International Journal of Rehabilitation Research 26(1):51-56.
Flannery, J. C., and S. H. Faria. 1999. Limb loss: Alterations in body image. Journal of Vascular Nursing 17(4):100-106.
Fraser, C., and A. W. Wing. 1981. A case study of reaching by a user of a manually-operated artificial hand. Prosthetics and Orthotics International 5(3):151-156.
Gaine, W. J., C. Smart, and M. Bransby-Zachary. 1997. Upper limb traumatic amputees: Review of prosthetic use. Journal of Hand Surgery (British and European Volume) 22(1):73-76.
Gallagher, P., O. Horgan, F. Franchignoni, A. Giordano, and M. MacLachlan. 2007. Body image in people with lower-limb amputation: A Rasch analysis of the amputee body image scale. American Journal of Physical Medicine & Rehabilitation 86(3):205-215.
Gallagher, P., M. A. O’Donovan, A. Doyle, and D. Desmond. 2011. Environmental barriers, activity limitations and participation restrictions experienced by people with major limb amputation. Prosthetics and Orthotics International 35(3):278-284.
Hacking, H. G. A., J.-P. van den Berg, K. T. Dahmen, and M. W. Post. 1997. Long-term outcome of upper limb prosthetic use in the Netherlands. European Journal of Physical Medicine and Rehabilitation 7(6):179-181.
Hanley, M. A., D. M. Ehde, M. Jensen, J. Czerniecki, D. G. Smith, and L. R. Robinson. 2009. Chronic pain associated with upper-limb loss. American Journal of Physical Medicine & Rehabilitation 88(9):742-751.
Hanson, W. 2003. Upper limb prosthetic components: Function vs. appearance. In Motion 13(6):29.
Harrtigan, B., and S. Serrafin. 2004. Kinesiology and functional characteristics of the upper limb. In Atlas of amputations and limb deficiencies: Surgical, prosthetic, and rehabilitation principles, 3rd ed., edited by D. G. Smith, J. W. Michael, and J. H. Bowker. Rosemont, IL: American Academy of Orthopaedic Surgeons. Pp. 101-116.
Hebert, J. S., J. L. Olson, M. J. Morhart, M. R. Dawson, P. D. Marasco, T. A. Kuiken, and K. M. Chan. 2014. Novel targeted sensory reinnervation technique to restore functional hand sensation after transhumeral amputation. IEEE Transactions on Neural Systems and Rehabilitation Engineering 22(4):765-773.
Hebert, J. S., K. M. Chan, and M. R. Dawson. 2016. Cutaneous sensory outcomes from three transhumeral targeted reinnervation cases. Prosthetics and Orthotics International 40(3):303-310.
Heckathorne, C. W. 1992. Upper-limb prosthetics: Components for adult externally powered systems. In Atlas of limb prosthetics: Surgical, prosthetic, and rehabilitation principles, 2nd ed., edited by J. H. Bowker and J. W. Michael. St. Louis, MO: Mosby-Year Book, Inc. Pp. 151-174.
Herberts, P., L. Korner, K. Caine, and L. Wensby. 1980. Rehabilitation of unilateral below-elbow amputees with myoelectric prostheses. Scandinavian Journal of Rehabilitation Medicine 12(3):123-128.
Hosmer. 2017. Hosmer products. http://hosmer.com/products (accessed January 5, 2017).
Hurley, R. K., Jr., J. C. Rivera, J. C. Wenke, and C. A. Krueger. 2015. Identifying obstacles to return to duty in severely injured combat-related servicemembers with amputation. Journal of Rehabilitation Research & Development 52(1):53-61.
Infinite Biomedical Technologies. n.d. morph2. http://www.i-biomed.com/morph2.html (accessed April 5, 2017).
Inkellis, E. R., E. E. Low, and S. Morshed. 2015. Traumatic upper extremity amputations: An epidemiologic study using the National Trauma Data Bank. Paper presented at American Academy of Orthotists & Prosthetists 41st Academy Annual Meeting & Scientific Symposium, New Orleans, LA, February 18-21. http://www.oandp.org/publications/jop/2015/2015-05.pdf (accessed January 21, 2017).
ISO (International Organization for Standardization). n.d. Prosthetics and orthotics—vocabulary—part 4: Terms relating to limb amputation. https://www.iso.org/obp/ui/#iso:std:iso:8549:-4:ed-1:v1:en (accessed January 18, 2017).
James, M. A., A. M. Bagley, K. Brasington, C. Lutz, S. McConnell, and F. Molitor. 2006. Impact of prostheses on function and quality of life for children with unilateral congenital below-the-elbow deficiency. Journal of Bone & Joint Surgery: American Edition 88(11):2356-2365.
Johnson, S. S., and E. Mansfield. 2014. Prosthetic training: Upper limb. Physical Medicine and Rehabilitation Clinics of North America 25(1):133-151.
Jones, L. E., and J. H. Davidson. 1995. The long-term outcome of upper limb amputees treated at a rehabilitation centre in Sydney, Australia. Disability and Rehabilitation 17(8):437-442.
Jönsson, S., K. Caine-Winterberger, and R. Brånemark. 2011. Osseointegration amputation prostheses on the upper limbs: Methods, prosthetics and rehabilitation. Prosthetics and Orthotics International 35(2):190-200.
Kejlaa, G. H. 1993. Consumer concerns and the functional value of prostheses to upper limb amputees. Prosthetics and Orthotics International 17(3):157-163.
Kestner, S. 2006. Defining the relationship between prosthetic wrist function and its use in performing work tasks and activities of daily living. Journal of Prosthetics and Orthotics 18(3):80-86.
Kooijman, C. M., P. U. Dijkstra, J. H. Geertzen, A. Elzinga, and C. P. van der Schans. 2000. Phantom pain and phantom sensations in upper limb amputees: An epidemiological study. Pain 87(1):33-41.
Kruger, L. M., and S. Fishman. 1993. Myoelectric and body-powered prostheses. Journal of Pediatric Orthopaedics 13(1):68-75.
Kyberd, P. J. 2011. The influence of control format and hand design in single axis myoelectric hands: Assessment of functionality of prosthetic hands using the Southampton hand assessment procedure. Prosthetics and Orthotics International 35(3):285-293.
Kyberd, P. J., and W. Hill. 2011. Survey of upper limb prosthesis users in Sweden, the United Kingdom and Canada. Prosthetics and Orthotics International 35(2):234-241.
Lake, C. 1997. Effects of prosthetic training on upper-extremity prosthesis use. Journal of Prosthetics and Orthotics 9(1):3-9.
Lehneis, H. R., and R. Dickey. 1992. Special considerations: Fitting and training the bilateral upper-limb amputee. In Atlas of limb prosthetics: Surgical, prosthetic, and rehabilitation principles, 2nd ed., edited by J. H. Bowker and J. W. Michael. St. Louis, MO: Mosby-Year Book, Inc. Pp. 311-324.
Lerman, J. A., E. Sullivan, D. A. Barnes, and R. J. Haynes. 2005. The Pediatric Outcomes Data Collection Instrument (PODCI) and functional assessment of patients with unilateral upper extremity deficiencies. Journal of Pediatric Orthopaedics 25(3):405-407.
Lifchez, S. D., J. Marchant-Hanson, H. S. Matloub, J. R. Sanger, W. W. Dzwierzynski, and H. H. Nguyen. 2005. Functional improvement with digital prosthesis use after multiple digit amputations. American Society for Surgery of the Hand 30(4):790-794.
Livingston, D. H., D. Keenan, D. Kim, J. Elcavage, and M. A. Malagnoni. 1994. Extent of disability following traumatic extremity amputation. Journal of Trauma 37(3):495-499.
Lock, B. A., and L. J. Hargrove. 2014. Systems and methods for autoconfiguration of pattern-recognition controlled myoelectric prostheses (application number 13/950,000). Washington, DC: U.S. Patent and Trademark Office. http://appft1.uspto.gov/netacgi/nph-Parser?Sect1=PTO1&Sect2=HITOFF&d=PG01&p=1&u=/netahtml/PTO/srchnum.html&r=1&f=G&l=50&s1=20140032462.PGNR (accessed January 24, 2017).
Luchetti, M., A. G. Cutti, G. Verni, R. Sacchetti, and N. Rossi. 2015. Impact of Michelangelo prosthetic hand: Findings from a crossover longitudinal study. Journal of Rehabilitation Research & Development 52(5):605-618.
MacKenzie, E. J., A. S. Jones, M. J. Bosse, R. C. Castillo, A. N. Pollak, L. X. Webb, M. F. Swiontkowski, J. F. Kellam, D. G. Smith, R. W. Sanders, A. L. Jones, A. J. Starr, M. P. McAndrew, B. M. Patterson, and A. R. Burgess. 2007. Health-care costs associated with amputation or reconstruction of a limb-threatening injury. Journal of Bone & Joint Surgery American Edition 89(8):1685-1692.
MaGuire, M. T. 2008. Empowering prosthetics: A team approach to prosthetics and limb loss. REHAB Management, December 1. http://www.rehabpub.com/2008/12/empowering-prosthetics (accessed January 21, 2017).
Malone, J. M., L. L. Fleming, J. Roberson, T. E. Whitesides, Jr., J. M. Leal, J. U. Poole, and R. S. Grodin. 1984. Immediate, early, and late postsurgical management of upper-limb amputation. Journal of Rehabilitation Research & Development 21(1):33-41.
Management of Upper Extremity Amputation Rehabilitation Working Group. 2014. VA/DoD clinical practice guideline for the management of upper extremity amputation rehabilitation. Washington, DC: VA, Department of Defense (DoD).
Marasco, P. D., A. E. Schultz, and T. A. Kuiken. 2009. Sensory capacity of reinnervated skin after redirection of amputated upper limb nerves to the chest. Brain 132(Pt. 6):1441-1448.
McFarland, L. V., S. Winkler, M. W. Jones, A. W. Heinemann, G. E. Reiber, and A. Esquenazi. 2010. Unilateral upper limb loss: Satisfaction and prosthetic device use in service members from Vietnam and OIF/OEF conflicts. Journal of Rehabilitation Research & Development 47(4):275-298.
McKechnie, P. S., and A. John. 2014. Anxiety and depression following traumatic limb amputation: A systematic review. Injury 45(12):1859-1866.
Meurs, M., C. G. Maathuis, C. Lucas, M. Hadders-Algra, and C. K. van der Sluis. 2006. Prescription of the first prosthesis and later use in children with congenital unilateral upper limb deficiency: A systematic review. Prosthetics and Orthotics International 30(2):165-173.
Miguelez, J. M., and M. D. Miguelez. 2003. The microframe: The next generation of interface design for glenohumeral disarticulation and associated levels of limb deficiency. Journal of Prosthetics and Orthotics 15(2):66-71.
Miguelez, J. M., M. Lang, R. Dodson, M. Cameron, and C. Hays. 2016. Building a better arm: Design and fabrication in upper-limb prosthetics. O&P Edge, July.
Millstein, S., D. Bain D, and G. A. Hunter. 1985. A review of employment patterns of industrial amputees—Factors influencing rehabilitation. Prosthetics and Orthotics International 9(2):69-78.
Millstein, S. G., H. Heger, and G. A. Hunter. 1986. Prosthetic use in adult upper limb amputees: A comparison of the body powered and electrically powered prostheses. Prosthetics and Orthotics International 10(1):27-34.
Morris, R. 2008. Focus on upper extremity: Therapeutic influences on the upper-limb amputee. The Academy TODAY 4(3).
Muderis, M. A., K. Tetsworth, A. Khemka, S. Wilmot, B. Bosley, S. J. Lord, and V. Glatt. 2016. The Osseointegration Group of Australia Accelerated Protocol (OGAAP-1) for two-stage osseointegrated reconstruction of amputated limbs. The Bone & Joint Journal 98-B(7):952-960.
Nghiem, B. T., I. C. Sando, R. B. Gillespie, B. L. McLaughlin, G. J. Gerling, N. B. Langhals, M. G. Urbanchek, and P. S. Cederna. 2015. Providing a sense of touch to prosthetic hands. Plastic and Reconstructive Surgery 135(6):1652-1663.
O&P Almanac. 2016. Amputation data from community hospitals. O&P Almanac, April, p. 8.
Ortiz-Catalan, M., R. Branemark, B. Hakansson, and J. Delbeke. 2012. On the viability of implantable electrodes for the natural control of artificial limbs: Review and discussion. BioMedical Engineering OnLine 11:33.
Ortiz-Catalan, M., B. Håkansson, and R. Brånemark. 2014. An osseointegrated human-machine gateway for long-term sensory feedback and motor control of artificial limbs. Science Translational Medicine 6(257):257re6.
Østlie, K., R. J. Franklin, O. H. Skjeldal, A. Skrondal, and P. Magnus. 2011. Musculoskeletal pain and overuse syndromes in adult acquired major upper-limb amputees. Archives of Physical Medicine and Rehabilitation 92(12):1967-1973.
Østlie, K., I. M. Lesjø, R. J. Franklin, B. Garfelt, O. H. Skjeldal, and P. Magnus. 2012a. Prosthesis rejection in acquired major upper-limb amputees: A population-based survey. Disability and Rehabilitation: Assistive Technology 7(4):294-303.
Østlie, K., I. M. Lesjø, R. J. Franklin, B. Garfelt, O. H. Skjeldal, and P. Magnus. 2012b. Prosthesis use in adult acquired major upper-limb amputees: Patterns of wear, prosthetic skills and the actual use of prostheses in activities of daily life. Disability and Rehabilitation: Assistive Technology 7(6):479-493.
Ottobock. 2016a. Michelangelo hand. https://professionals.ottobockus.com/Prosthetics/Upper-Limb-Prosthetics/Michelangelo-Axon-Bus-System/Michelangelo-Hand/c/2001 (accessed January 24, 2017).
Ottobock. 2016b. Michelangelo prosthetic hand. http://www.ottobockus.com/prosthetics/upper-limb-prosthetics/solution-overview/michelangelo-prosthetic-hand (accessed January 24, 2017).
Palmquist, A., T. Jarmar, L. Emanuelsson, R. Branemark, H. Engqvist, and P. Thomsen. 2008. Forearm bone-anchored amputation prosthesis: A case study on the osseointegration. Acta Orthopaedica 79(1):78-85.
Perkins, Z. B., H. D. De’Ath, G. Sharp, and N. R. Tai. 2012. Factors affecting outcome after traumatic limb amputation. British Journal of Surgery Society 99(Suppl. 1):75-86.
Phillips, S., L. Resnik, and G. Latlief. 2013. Endpoint control for a powered shoulder prosthesis. Journal of Prosthetics and Orthotics 25(4):193-200.
Pitkin, M. 2013. Design features of implants for direct skeletal attachment of limb prostheses. Journal of BioMedical Materials Research: Part A 101(11):3339-3348.
Postema, S. G., C. K. van der Sluis, K. Waldenlov, and L. M. Norling Hermansson. 2012. Body structures and physical complaints in upper limb reduction deficiency: A 24-year follow-up study. PLOS One 7(11):e49727.
Postema, S. G., R. M. Bongers, M. A. Brouwers, H. Burger, L. M. Norling-Hermansson, M. F. Reneman, P. U. Dijkstra, and C. K. van der Sluis. 2016a. Musculoskeletal complaints in transverse upper limb reduction deficiency and amputation in the Netherlands: Prevalence, predictors, and effect on health. Archives of Physical Medicine and Rehabilitation 97(7): 1137-1145.
Postema, S. G., R. M. Bongers, M. A. Brouwers, H. Burger, L. M. Norling-Hermansson, M. F. Reneman, P. U. Dijkstra, and C. K. van der Sluis. 2016b. Upper limb absence: Predictors of work participation and work productivity. Archives of Physical Medicine and Rehabilitation 97(6):892-899.
Powell, M. A., and N. V. Thakor. 2013. A training strategy for learning pattern recognition control for myoelectric prostheses. Journal of Prosthetics and Orthotics 25(1):30-41.
Powell, M. A., R. R. Kaliki, and N. V. Thakor. 2014. User training for pattern recognition-based myoelectric prostheses: Improving phantom limb movement consistency and distinguishability. IEEE Transactions on Neural Systems and Rehabilitation Engineering 22(3):522-532.
Racey, J. 1992. Psychological adaptation to amputation. In Atlas of limb prosthetics: Surgical, prosthetic, and rehabilitation principles, 2nd ed., edited by J. H. Bowker and J. W. Michael. St. Louis, MO: Mosby-Year Book, Inc. Pp. 707-716.
Rager, D. M., D. Alvares, I. Birznieks, S. J. Redmond, J. W. Morley, N. H. Lovell, and R. M. Vickery. 2013. Generating tactile afferent stimulation patterns for slip and touch feedback in neural prosthetics. Annual International Conference of the IEEE Engineering in Medicine and Biology Society 2013:5922-5925.
Raichle, K. A., M. A. Hanley, I. Molton, N. J. Kadel, K. Campbell, E. Phelps, D. Ehde, and D. G. Smith. 2008. Prosthesis use in persons with lower- and upper-limb amputation. Journal of Rehabilitation Research & Development 45(7):961-972.
Resnik, L., and M. Borgia. 2012. Reliability and validity of outcome measures for upper limb amputation. Journal of Prosthetics and Orthotics 24(4):192-212.
Resnik, L., and M. Borgia. 2015. Reliability, validity, and responsiveness of the quickdash in patients with upper limb amputation. Archives of Physical Medicine and Rehabilitation 96(9):1676-1683.
Resnik, L., M. R. Meucci, S. Lieberman-Klinger, C. Fantini, D. L. Kelty, R. Disla, and N. Sasson. 2012. Advanced upper limb prosthetic devices: Implications for upper limb prosthetic rehabilitation. Archives of Physical Medicine and Rehabilitation 93(4):710-717.
Resnik, L., T. Patel, S. G. Cooney, J. J. Crisco, and C. Fantini. 2014a. Comparison of transhumeral socket designs utilizing patient assessment and in vivo skeletal and socket motion tracking: A case study. Disability and Rehabilitation: Assistive Technology 11(5):423-432.
Resnik, L., S. L. Klinger, and K. Etter. 2014b. The DEKA Arm: Its features, functionality, and evolution during the Veterans Affairs study to optimize the DEKA arm. Prosthetics and Orthotics International 38(6):492-504.
Resnik, L., S. L. Klinger, K. Korp, and L. S. Walters. 2014c. Training protocol for a powered shoulder prosthesis. Journal of Rehabilitation Research & Development 51(8):vii-xvi.
Ritchie, S., S. Wiggins, and A. Sanford. 2011. Perceptions of cosmesis and function in adults with upper limb prostheses: A systematic literature review. Prosthetics and Orthotics International 35(4):332-341.
Roeschlein, R. A., and E. Domholdt. 1989. Factors related to successful upper extremity prosthetic use. Prosthetics and Orthotics International 13(1):14-18.
Saradjian, A., A. R. Thompson, and D. Datta. 2008. The experience of men using an upper limb prosthesis following amputation: positive coping and minimizing feeling different. Disability and Rehabilitation 30(11):871-883.
Scheme, E., and K. Englehart. 2011. Electromyogram pattern recognition for control of powered upper-limb prostheses: State of the art and challenges for clinical use. Journal of Rehabilitation Research & Development 48(6):643-659.
Schiefer, M., D. Tan, S. M. Sidek, and D. J. Tyler. 2016. Sensory feedback by peripheral nerve stimulation improves task performance in individuals with upper limb loss using a myoelectric prosthesis. Journal of Neural Engineering 13(1):016001.
Schofield, J. S., K. R. Evans, J. P. Carey, and J. S. Hebert. 2014. Applications of sensory feedback in motorized upper extremity prosthesis: A review. Expert Review of Medical Devices 11(5):499-511.
Schoppen, T., A. Boonstra, J. W. Groothoff, J. de Vries, L. N. Goeken, and W. H. Eisma. 2001a. Employment status, job characteristics, and work-related health experience of people with a lower limb amputation in the Netherlands. Archives of Physical Medicine and Rehabilitation 82(2):239-245.
Schoppen, T., A. Boonstra, J. W. Groothoff, E. van Sonderen, L. N. Goeken, and W. H. Eisma. 2001b. Factors related to successful job reintegration of people with a lower limb amputation. Archives of Physical Medicine and Rehabilitation 82(10):1425-1431.
Schuch, C. M., and C. H. Pritham. 1994. International standards organization terminology: Application to prosthetics and orthotics. Journal of Prosthetics and Orthotics 6(1):29-33.
Schultz, A., S. Baade, and T. Kuiken. 2007. Expert opinions on success factors for upper-limb prostheses. Journal of Rehabilitation Research & Development 44(4):483-490.
Silcox, D. H., III, M. D. Rooks, R. R. Vogel, and L. L. Fleming. 1993. Myoelectric prostheses. A long-term follow-up and a study of the use of alternate prostheses. Journal of Bone & Joint Surgery: American Edition 75(12):1781-1789.
Simon, A. M., B. A. Lock, and K. A. Stubblefield. 2012. Patient training for functional use of pattern recognition-controlled prostheses. Journal of Prosthetics and Orthotics 24(2):56-64.
Sjoberg, L., Y. Nilsagard, and C. Fredriksson. 2014. Life situation of adults with congenital limb reduction deficiency in Sweden. Disability and Rehabilitation 36(18):1562-1571.
Smit, G., and D. H. Plettenburg. 2010. Efficiency of voluntary closing hand and hook prostheses. Prosthetics and Orthotics International 34(4):411-427.
Smurr, L. M., K. Gulick, K. Yancosek, and O. Ganz. 2008. Managing the upper extremity amputee: A protocol for success. Journal of Hand Therapy 21(2):160-176.
Sobuh, M. M., L. P. Kenney, A. J. Galpin, S. B. Thies, J. McLaughlin, J. Kulkarni, and P. Kyberd. 2014. Visuomotor behaviours when using a myoelectric prosthesis. Journal of NeuroEngineering and Rehabilitation 11:72.
Stark, G. 2016. Competency, risk, and acceptance of upper limb prosthetic technology. Presentation to the Committee on the Use of Selected Assistive Products and Technologies in Eliminating or Reducing the Effects of Impairments, Washington, DC, May 16.
Steeper. 2016. Bebionic technical manual. http://bebionic.com/uploads/files/RSLLIT317_-_Tech_Manual_Medium_Large_Hand_web1.pdf (accessed January 18, 2017).
Stinner, D. J., T. C. Burns, K. L. Kirk, and J. R. Ficke. 2010. Return to duty rate of amputee soldiers in the current conflicts in Afghanistan and Iraq. Journal of Trauma and Acute Care Surgery 68(6):1476-1479.
Stubblefield, K. A., L. A. Miller, R. D. Lipschutz, and T. A. Kuiken. 2009. Occupational therapy protocol for amputees with targeted muscle reinnervation. Journal of Rehabilitation Research & Development 46(4):481-488.
Tabot, G. A., J. F. Dammann, J. A. Berg, F. V. Tenore, J. L. Boback, R. J. Vogelstein, and S. J. Bensmaia. 2013. Restoring the sense of touch with a prosthetic hand through a brain interface. Proceedings of the National Academy of Sciences of the United States of America 110(45):18279-18284.
Tan, D. W., M. A. Schiefer, M. W. Keith, J. R. Anderson, and D. J. Tyler. 2015. Stability and selectivity of a chronic, multi-contact cuff electrode for sensory stimulation in human amputees. Journal of Neural Engineering 12(2):026002.
Tennent, D. J., J. C. Wenke, J. C. Rivera, and C. A. Krueger. 2014. Characterisation and outcomes of upper extremity amputations. Injury 45(6):965-969.
Texas Assistive Devices. 2016. Manufacturers of upper extremity components for persons with hand dysfunction and amputation. http://www.n-abler.org (accessed January 21, 2017).
Tintle, S. M., M. F. Baechler, G. P. Nanos, III, J. A. Forsberg, and B. K. Potter. 2010. Traumatic and trauma-related amputations: Part II: Upper extremity and future directions. Journal of Bone & Joint Surgery: American Edition 92(18):2934-2945.
Touch Bionics. 2016. Touch Bionics i-limb ultra. http://www.touchbionics.com/products/active-prostheses/i-limb-ultra (accessed January 21, 2017).
Touch Bionics. n.d.-a. Touch bionics i-limb quantum. http://www.touchbionics.com/products/active-prostheses/i-limb%E2%84%A2-quantum (accessed April 5, 2017).
Touch Bionics. n.d.-b. Grip chips demo. http://www.i-biomed.com/morph2.html (accessed April 5, 2017).
TRS Prosthetics. 2016. Sports: Prosthetics for sports and activities. http://www.trsprosthetics.com/shop-category/sports (accessed January 21, 2017).
Tyler, D. J. 2015. Neural interfaces for somatosensory feedback: Bringing life to a prosthesis. Current Opinion in Neurology 28(6):574-581.
Uellendahl, J., and C. W. Heckathorne. 1989. Creative prosthetic solutions for bilateral upper extremity amputation. In Comprehensive management of the upper-limb amputee, edited by D. J. Atkins and R. H. Meier. New York: Springer. Pp. 225-237.
VA (U.S. Department of Veterans Affairs). 2012. VHA handbook 1172.03: Amputation system of care. Washington, DC: VA.
VA. 2016. Rehabilitation and prosthetic services: Amputation system of care. http://www.prosthetics.va.gov/PROSTHETICS/asoc/index.asp (accessed January 18, 2017).
van der Niet, O., R. M. Bongers, and C. K. van der Sluis. 2013. Functionality of i-limb and i-limb pulse hands: Case report. Journal of Rehabilitation Research & Development 50(8):1123-1128.
van der Sluis, C. K., P. P. Hartman, T. Schoppen, and P. U. Dijkstra. 2009. Job adjustments, job satisfaction and health experience in upper and lower limb amputees. Prosthetics and Orthotics International 33(1):41-51.
Vasluian, E., I. G. de Jong, W. G. Janssen, M. J. Poelma, I. van Wijk, H. A. Reinders-Messelink, and C. K. van der Sluis. 2013. Opinions of youngsters with congenital below-elbow deficiency, and those of their parents and professionals concerning prosthetic use and rehabilitation treatment. PLOS One 8(6):e67101.
Vincent, H. K., M. Horodyski, K. R. Vincent, S. T. Brisbane, and K. K. Sadasivan. 2015. Psychological distress after orthopedic trauma: Prevalence in patients and implications for rehabilitation. PM&R 7(9):978-989.
Weir, R. F., P. R. Troyk, G. DeMichele, and T. Kuiken. 2003. Implantable myoelectric sensors (IMES) for upper-extremity prosthesis control—preliminary work. Paper presented at IEEE Engineering in Medicine and Biology Society, Cancun, Mexico, September 17-21.
Williams, T. W. 2004. Control of powered upper extremity prostheses. In Functional restoration of adults and children with upper extremity amputation, edited by R. H. Meier and D. J. Atkins. New York: Demos Medical Publishing, Inc. Pp. 207-224.
Winchell, E. 1996. Body image: Acceptance is step one. In Motion 6(4).
Witteveen, H. J., F. Luft, J. S. Rietman, and P. H. Veltink. 2014. Stiffness feedback for myoelectric forearm prostheses using vibrotactile stimulation. IEEE Transactions on Neural Systems and Rehabilitation Engineering 22(1):53-61.
Witteveen, H. J., H. S. Rietman, and P. H. Veltink. 2015. Vibrotactile grasping force and hand aperture feedback for myoelectric forearm prosthesis users. Prosthetics and Orthotics International 39(3):204-212.
Wright, T. W., A. D. Hagen, and M. B. Wood. 1995. Prosthetic usage in major upper extremity amputations. The Journal of Hand Surgery 20(4):619-622.
Ziegler-Graham, K., E. J. MacKenzie, P. L. Ephraim, T. G. Travison, and R. Brookmeyer. 2008. Estimating the prevalence of limb loss in the United States: 2005 to 2050. Archives of Physical Medicine and Rehabilitation 89(3):422-429.
CHAPTER 4 ANNEX TABLES
ANNEX TABLE 4-1
Upper-Limb Prosthetic Device Taxonomy
| Device | Estimated Costa | Indications/Requirements for Use | ||||
|---|---|---|---|---|---|---|
| Body-Powered | ||||||
| Transradial | $4,400 |
|
||||
| Transhumeral | $6,000 | |||||
| Shoulder disarticulation | $11,300 | |||||
| Myoelectric | ||||||
| Transradial | $15,100 |
|
||||
| Transhumeral | $56,200 | |||||
| Shoulder disarticulation | $65,700 | |||||
| Hybrid | ||||||
| Transhumeral | $19,200 |
|
||||
| Shoulder disarticulation | $32,500 | |||||
| Relative Contraindications | Benefits of Device | Limitations of Device |
|---|---|---|
|
|
|
|
|
|
|
|
|
| Device | Estimated Costa | Indications/Requirements for Use | ||||
|---|---|---|---|---|---|---|
| Terminal Devices | ||||||
| Passive hand | $500b |
|
||||
| Body-powered hook | $400–$1,420 |
|
||||
| Single-degree-of-freedom (DOF), body-powered hand | $1,100–$1,500 |
|
||||
| Single-DOF powered hand | $6,800 |
|
||||
| Multiarticulating hand powered hand | $32,000b |
|
||||
| Relative Contraindications | Benefits of Device | Limitations of Device |
|---|---|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Device | Estimated Costa | Indications/Requirements for Use |
|---|---|---|
| Specialized terminal device: work attachment | $600 |
|
NOTES:
Transradial body-powered includes codes L6100, L6615, L6660, L6675, L6680, L6687, L7400, L7403, L8415, L8435, and L8485.
Transhumeral body-powered includes codes L6250, L6615, L6620, L6635, L6660, L6665, L6676, L6682, L6688, L7401, L7404, L8415, L8435, and L8485.
Shoulder disarticulation body-powered includes codes L6300, L6615, L6620, L6635, L6641, L6646, L6647, L6660, L6665, L6672, L6684, L6689, L7402, and L7405.
Transradial myoelectric includes codes L6680, L6687, L6686, L6935, L7259, L7368, L7400, and L7403.
Transhumeral myoelectric includes codes L6682, L6688, L6955, L7181, L7259, L7368, L7401, and L7404.
Shoulder disarticulation myoelectric includes codes L6646, L6648, L6684, L6689, L6965, L7181, L7259, L7368, L7402, and L7405.
Transhumeral hybrid includes codes L6638, L6693, L6655, L6675, L6682, L6688, L6955, L7368, L7401, and L7404.
Shoulder disarticulation hybrid includes codes L6638, L6646, L6648, L6693, L6655, L6675, L6684, L6689, L6965, L7259, L7368, L7402, and L7405.
Passive hand includes codes L6703 and L6890.
Body-powered hook includes codes L6706 (low) and L6707 (high).
Body-powered hand includes codes L6708 (low) and L6709 (high) and L6890.
Sports/recreation/work attachment device includes code L6704.
Myoelectric hand (low) includes codes L6629, L6890, L7007, and L6882.
Myoelectric hand (high) includes codes L6621, L6629, L6890, L6880, L6881, and L6882.
a Estimated costs for devices include the base code and associated codes to create an example full system, excluding the terminal device (hook or hand). Codes shown are meant to be representative of a general estimate of billing for the upper-limb prosthesis design types and are not meant to be specific billing recommendations. Prices are based on the January 2015 Centers for Medicare & Medicaid Services (CMS) fee schedule for Illinois. Prices in the table have been rounded to the nearest $100.
b Any unlisted codes that might be suggested for a product (for example, advanced myoelectric devices, custom silicone gloves) are not included in these estimates.
| Relative Contraindications | Benefits of Device | Limitations of Device |
|---|---|---|
|
|
ANNEX TABLE 4-2
Ability of Upper-Limb Prosthetic Devices to Mitigate the Effects of Impairment
| Body-Powered | |||
|---|---|---|---|
| TR | TH | Shoulder | |
| Sensation | |||
| Proprioception | No | No | No |
| Touch function | No | No | No |
| Passive Range of Motion | |||
| Fingers | Partial | Partial | Partial |
| Thumb | No | No | No |
| Wrist ulnar/radial deviation | No | No | No |
| Wrist flexion/extension | Var/partial | Var/partial | Var/partial |
| Wrist pronation/supination | Variable | Variable | Variable |
| Elbow flexion/extension | NA | Variable | Variable |
| Shoulder flexion/extension | NA | NA | Var/partial |
| Shoulder abduction/adduction | NA | NA | Var/partial |
| Shoulder rotation | NA | NA | Variable |
| Shoulder horizontal ad/abduction | NA | NA | Var/partial |
| Active Range of Motion | |||
| Fingers | Partial | Partial | Partial |
| Thumb | No | No | No |
| Wrist ulnar/radial deviation | No | No | No |
| Wrist flexion/extension | No | No | No |
| Wrist pronation/supination | Var/Yes | NA | NA |
| Elbow flexion/extension | NA | Variable | No |
| Shoulder flexion/extension | NA | NA | No |
| Shoulder abduction/adduction | NA | NA | No |
| Shoulder rotation | NA | NA | No |
| Shoulder horizontal ad/abduction | NA | NA | No |
| Myoelectric | Hybrid | ||||
|---|---|---|---|---|---|
| TR | TH | Shoulder | TH | Shoulder | |
| No | No | No | No | No | |
| No | No | No | No | No | |
| Var/partial | Var/partial | Var/partial | Var/partial | Var/partial | |
| Var/partial | Var/partial | Var/partial | Var/partial | Var/partial | |
| No | No | No | No | No | |
| Var/partial | Var/partial | Var/partial | Var/partial | Var/partial | |
| Variable | Variable | Variable | Variable | Variable | |
| NA | Partial | Variable | Variable | Variable | |
| NA | NA | Var/partial | NA | Variable | |
| NA | NA | Var/partial | NA | Variable | |
| NA | NA | Variable | NA | Variable | |
| NA | NA | Var/partial | NA | Var/partial | |
| No | No | No | No | No | |
| No | No | No | No | No | |
| Var/Yes | Var/Yes | Var/Yes | Var/Yes | Var/Yes | |
| NA | Yes | Yes | No | No | |
| NA | NA | No | NA | No | |
| NA | NA | No | NA | No | |
| NA | NA | No | NA | No | |
| NA | NA | No | NA | No | |
| Body-Powered | |||
|---|---|---|---|
| TR | TH | Shoulder | |
| Activities | |||
| Grasp | Partial | Partial | Partial |
| Fine motor use | Partial | Partial | Partial |
| Dexterous activities | Partial | Partial | Partial |
| Handling objects | Partial | Partial | Partial |
| Lifting <20 pounds | Partial | Partial | Partial |
| Lifting >20 pounds | Partial | Partial | Partial |
| Carrying <20 pounds | Partial | Partial | Partial |
| Carrying >20 pounds | Partial | Partial | Partial |
| Reaching forward | Variable | Partial | Partial |
| Overhead reaching | Var/partial | Var/partial | Var/partial |
| Considerations | |||
| Indications |
|
||
| Contraindications |
|
||
| Clinical expertise/training |
|
||
| Limitations |
|
||
NOTE: NA = not applicable; TH = transhumeral; TR = transradial; Var = Variable.
| Myoelectric | Hybrid | ||||
|---|---|---|---|---|---|
| TR | TH | Shoulder | TH | Shoulder | |
| Partial | Partial | Partial | Partial | Partial | |
| Partial | Partial | Partial | Partial | Partial | |
| Partial | Partial | Partial | Partial | Partial | |
| Partial | Partial | Partial | Partial | Partial | |
| Partial | Partial | Partial | Partial | Partial | |
| Var/partial | Var/partial | Var/partial | No | No | |
| Partial | Partial | Partial | Partial | Partial | |
| Var/partial | Var/partial | Var/partial | Var/partial | Var/partial | |
| Partial | Partial | Partial | Partial | Partial | |
| Partial | Partial | Partial | Partial | No | |
|
|
||||
|
|
||||
|
|
||||
|
|
||||
This page intentionally left blank.