2
Context for the Use of Marmosets as Animal Models
Suzette Tardif, associate director of Research for the Southwest National Primate Research Center, began the workshop with a keynote address describing the history of marmosets for research, how and why they are currently used, and the challenges of working with them.
HISTORY
Marmosets have been kept in captivity for centuries, but they only began to be used as biomedical research subjects in the 1960s. They were appealing to researchers because they were so small, widely available (at the time), and relatively inexpensive. For many decades, the term “marmosets” was used interchangeably to describe tamarins and marmosets, and that is how they are described in the older literature. For example, Callithrix jacchus was called “cotton-eared marmoset,” but today it is labeled “common marmoset.” Similarly, Saguinus oedipus, today’s “cotton-top tamarin,” was labeled “cotton-top marmoset.” Both tamarins and marmosets belong to the Callitrichinae sub-family. It is important to keep this history in mind when reviewing older studies.
Marmosets used in U.S. research facilities today followed a convoluted path to get here. In the 1960s and 1970s, it was mostly tamarins, not marmosets, that came to U.S. research facilities from wild populations in South American countries such as Brazil, Colombia, Peru, and Venezuela. Marmosets from the same areas were more commonly imported to the
United Kingdom and Europe and from there eventually made their way to research facilities in Japan and the United States.
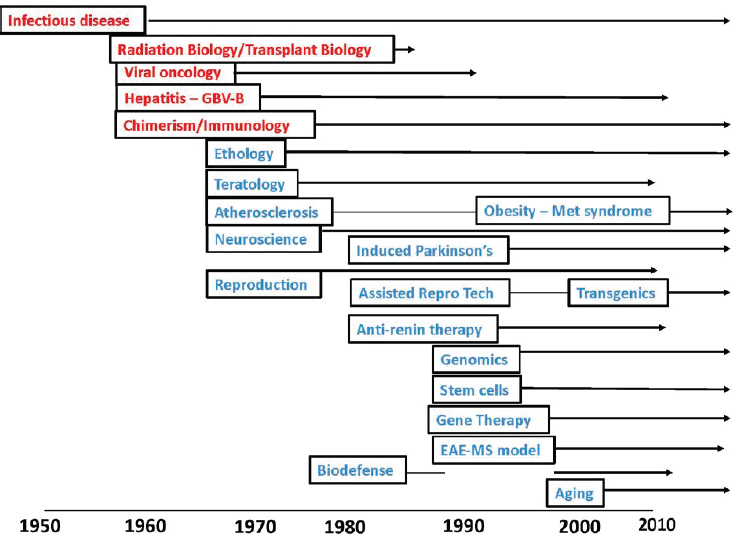
At that time, Oak Ridge Associated Universities had the largest tamarin/marmoset population in the United States, housing about 550–650 animals, and also served as the national distribution center. The animals were being used to study a wide variety of medical conditions (see Figure 2-1). In those first few decades, tamarin research focused on infectious diseases, radiation and transplant biology, viral oncology, and immunology. Marmosets were used for studies in a wide range of areas including reproduction, biodefense, and gene therapy.
CURRENT USE AND KEY ADVANTAGES
In the 1980s and 1990s, researchers began to use marmosets more and more in neuroscience, behavior, and physiology inquiries, as well as stem cell research and assisted reproductive technologies. Researchers successfully petitioned to have the marmoset genome sequenced, and more
Abbreviations: EAE-MS = experimental autoimmune encephalomyelitis-multiple sclerosis; GBV-B = GB virus B; Met = metabolic.
SOURCE: Suzette Tardif.
SOURCE: Suzette Tardif.
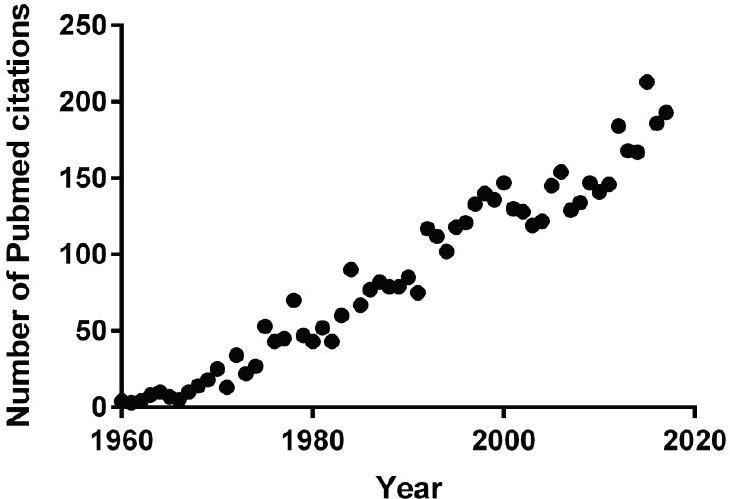
recent years have seen a growing emphasis on using them in gene therapy, and aging studies. Today, marmosets as model organisms are attracting so much research interest (see Figure 2-2) that demand is far outstripping an already limited supply. One of the main drivers of this increased demand is neuroscience studies. Marmosets are excellent models for studies of visual systems, auditory systems, cognitive function, and connectivity. They have two key advantages over other primates: the lissencephalic1 cortex, which is easier to measure and manipulate in a marmoset than a macaque; and the fact that they are small enough to be housed in a conventional vivarium, whereas larger primates need more space. Currently, 10–15 institutions are developing small marmoset colonies (of 20–60 animals each) for neuroscience studies.
A second driver of increased demand for marmosets is transgenics and gene editing, in particular gene editing techniques such as clustered regularly interspaced short palindromic repeats/CRISPR-associated protein 9 (CRISPR/Cas9). Marmosets’ reproductive capabilities—they can gestate
___________________
1 Lissencephaly (i.e., “smooth brain”) is a condition characterized by the absence of normal folds in the cerebral cortex (see https://www.ninds.nih.gov/Disorders/All-Disorders/Lissencephaly-Information-Page).
up to four embryos at once, have two pregnancies per year, and become physically mature in only 18 months—imply that in only a few years, a facility breeding marmosets can have a dramatically bigger population than one breeding macaques. The latter exhibit breeding seasons, produce one offspring per year at maximum and are not sexually mature until 3 years old. Marmoset litters are chimeric, in terms of their hematopoietic cells and possibly other cells as well. The growing focus on transgenic work has led to the development of some larger colonies (250–350 animals) already; if the field continues to grow, some facilities may establish much larger colonies (up to 1,000 animals) for line maintenance and characterization.
KEY CHALLENGES
Working with marmosets, researchers face three main challenges: low supplies, a lack of standard practices for their use and care, and a lack of appropriate antibody reagents.
Marmosets are hard to acquire. The Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) regulates the international trade of wild species, including all primates. In the 1950s and 1960s it is estimated that more than 200,000 wild primates were exported from Central and South America (Mittermeier et al. 1989). Since the enactment of CITES in 1975 that number has dropped dramatically to an estimated 5,000 primates of captive status (de Souza Fialho et al. 2016). CITES is undoubtedly beneficial to animals and ecosystems. While deforestation is probably the main driver of animal endangerment, exports for biomedical research have likely played an unfortunate role in the declines of some species, such as the cotton-top tamarin. Thus, current import controls will favor wild populations, even though it does make it harder for researchers to acquire marmosets.
The second major challenge, relating to the care and use of marmosets, stems from key differences between marmosets and more well-established model species. They have unique requirements in terms of housing, feeding, social interactions, and other facets, many of which remain poorly understood. There is no standardized diet for captive marmosets, and there are few people who have expertise in working with them. Marmosets in captivity are susceptible to a range of diseases and are particularly prone to marmoset wasting syndrome, which is not one disease but a perplexing composite of multiple conditions and etiologies that could be due to poor nutrition, stress, infection, or a combination of these factors. Their breeding and parenting behavior is also poorly understood, and although marmosets are easier to handle than tamarins (as they tend to be less easily stressed and are more easily habituated to handling), their multiple births can lead to poor parenting performance.
A third major challenge is that available antibody reagents often do not work with marmoset tissues. While this will likely change as more laboratories start to work with marmosets and as more emphasis is placed on mass spectrometry, currently this lack of reagents is an impediment to marmoset research.
Dr. Tardif concluded her presentation focusing on several important considerations regarding improved support of the marmoset as model organism. There is a crucial need to develop evidence-based standards for the management of marmosets in captivity to support their care, welfare, and ethical use in research. In addition, to improve the quality of research there is a need for molecular, genomic, and imaging tools developed specifically for marmosets as research models (rather than developed for other model organisms and adapted for marmosets). Finally, there is a need to establish clear and ethical approaches to managing marmosets throughout the entire research life cycle, including after animals are no longer useful for research. This is particularly important as interest in transgenic work grows and larger colonies are developed.
REFERENCES
de Souza Fialho M, Ludwig G, Valenca-Montenegro MM. 2016. Legal international trade in live neotropical primates originating from South America. Primate Conservation 30:1–6.
Mittermeier RA, Kinzey WG, Mast RB. 1989. Neotropical primate conservation. J Hum Evol 18(7):597–610.
This page intentionally left blank.






