1
Introduction
Separation, as the term is used in this report, is the division of a chemical mixture (a mixture of molecules or colloidal particles) into its constituent or distinct elements. The purpose of a chemical separation process is usually to enrich a product stream in one or more of the components of the original mixture. In some cases, the purpose of the separation process is to divide the mixture fully into its pure components. At the simplest level, a chemical separation might involve boiling and condensing water to eliminate salts or preferentially evaporating a more volatile component from a mixture. At a more sophisticated level, a chemical separation might involve passing a mixture through a specialized membrane that interacts with specific constituents to affect their permeabilities and thus separates them.
Separations are an integral component of a wide array of technologies that are necessary to meet societal needs. The goal can vary from dividing a complex mixture into thousands of fractions to extracting a single chemical from a highly dilute solution. The scale of a separation process can vary from less than a microgram to megatons of material. Regardless of the goal and the scale, separation processes are vital to numerous industries, including the oil and gas industry, chemical and pharmaceutical manufacturing, biotechnology, food production, water desalination and purification, and waste management and treatment. They are critical for providing clean water and air to the world’s population, extracting natural resources for energy storage and use, and delivering effective and affordable health care.
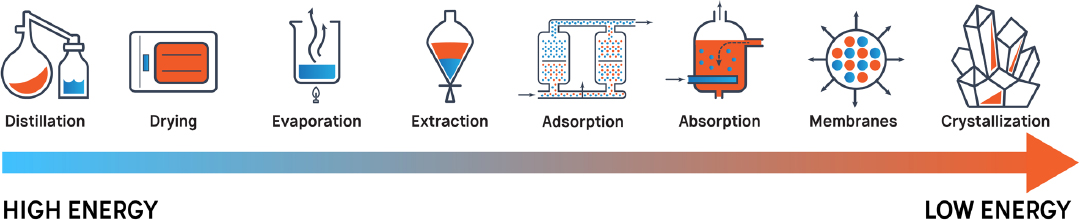
Although separation processes are used throughout many industries, there are challenges, and current methods are often costly. For example, most industrial separations are energy-intensive (see Box 1-1, Figure 1-1) and by some estimates account for about half the energy used in U.S. industry and 10–15% of total U.S. energy consumption (Humphrey and Keller, 1997; ORNL, 2005). Many traditional separation processes use larger amounts of energy than emerging technologies that do not rely on the vaporization of one or more of the chemical components and instead rely primarily on separation materials (see Figure 1-2). Although design and operation improvements have reduced the energy use of distillation processes, energy-intensive separations are reported to account for 80% of industrial separations (ORNL, 2005). Some separation processes generate large waste streams that are expensive to manage. For those reasons, advances in separation science are critical both for future U.S. economic competitiveness and for improving living standards globally.
Given the scientific and technological advances that have occurred in the decades since the publication of Separation and Purification: Critical Needs and Opportunities (NRC, 1987), the Department of Energy (DOE), the National Science Foundation (NSF), and the National Institute of Standards and Technology (NIST) recognized that there are important opportunities for improving separation processes and asked the National Academies of Sciences, Engineering, and Medicine (the National Academies) to develop an agenda for fundamental research in separation science. As a result of that request, the National Academies convened the Committee on a Research Agenda for a New Era in Separation Science, which prepared this report.
MOTIVATIONS
There were several compelling reasons to undertake the committee’s task; some are described below.
Developing a Sustainable Chemical Enterprise to Drive the Economy. Advances in separation science and technology are critical for developing a sustainable chemical enterprise. As noted, separations in industrial processes can be extremely energy-intensive. Reducing energy consumption in industrial separation processes
was identified as one of the grand challenges in Sustainability in the Chemical Industry: Grand Challenges and Research Needs (NRC, 2006, p. 86), which stated the following:
The energy efficiency of chemical separations is a key research component of this grand challenge. Finding effective alternatives to distillation are especially needed. While membrane separations, adsorption, and extractions tend to be less energy intensive, significant technical challenges must be overcome in the development of these alternatives in order to realize any significant reductions in the energy intensity of the [chemical processing industry].
More energy-efficient chemical separations in U.S. industry could save an estimated $4 billion in energy costs a year (DOE, 2015) and make industry more sustainable.
Creating a more sustainable chemical enterprise will also require optimizing separations so that useful materials can be recovered and waste generation minimized. For example, rare-earth metals are important resources that are used regularly in magnets, in renewable-energy technologies, and as catalysts in petroleum refining. However, separation of rare-earth metals from ores or used electronic components is inefficient and produces large amounts of waste and unwanted byproducts. New approaches would improve recovery of these valuable materials. Likewise, more mass- and energy-efficient separations are needed for a wide variety of chemical recycling applications. For instance, depolymerization of mixed plastics followed by easy separation into purified monomer streams could contribute to a more sustainable chemical enterprise.
Reducing Adverse Effects of Industrial Activities. Some industrial activities result in the discharge of pollutants and have adverse effects on human health and the environment. An integral component of developing a sustainable chemical enterprise is reduction of those effects. Creation of more efficient and selective separation processes can play a key role in this endeavor. For example, minimization of solvent use in separations, use of more sustainable solvents, and removal of heavy metals and other contaminants from wastewater before discharge all hold tremendous potential for reducing the amount of toxic and waste substances generated or released and thus reducing effects on human health and the environment.
Another grand challenge identified by the National Academy of Engineering (NRC, 2008, updated 2017) is the development of carbon-sequestration methods. The most energy-intensive and capital-intensive part of carbon sequestration is separation of carbon dioxide from a gas mixture or potentially separation of oxygen from air for use in oxyfuel combustion. In both cases, the main difficulty is to separate compounds that are extremely similar in size. More energy-efficient separations could reduce carbon dioxide emissions by 100 million tons a year (Sholl and Lively, 2016)—an important reduction in carbon emissions relative to climate-change initiatives.
Improving Human Health. Separation science is key to many aspects of improvement of the standard of living and quality of life of billions of people. For example, one-sixth of the world’s population do not have access to potable water. There is no lack of water on the planet, but only 3% is freshwater. Providing access to clean water is one of the 14 grand challenges identified by the National Academy of Engineering (NAE, 2008, updated 2017) and constitutes a separation challenge.
Improved separation technologies are also critical for other aspects of human health. For example, monoclonal antibodies (mAbs) are the most widely produced and most important biopharmaceutical products. Numerous types of separation methods are necessary in the production of these compounds. Their commercial-scale manufacture is based on batch processing in which each unit operation is completed in sequence, and a long-term challenge is to convert batch processing to continuous synthesis and purification (Zydney, 2016). To be used as pharmaceuticals, mAbs have to be safe for patients, and multiple quality attributes must be maintained. For example, the removal of compounds of lower and higher molecular weight than a given pharmaceutical mAb is important. Higher-molecular-weight species might cause unwanted immunogenic responses, such as anaphylaxis, and lower the efficacy of the drug. Lower-molecular-weight fragments often have lower activity than the original mAb, and immune responses might be elicited by exposure to unique epitopes. The exact glycosylation distribution and charge-state distribution of an approved mAb also must be maintained, and post-translation modifications might induce changes.
New and Challenging Separations. Many industrial processes—such as in the electronics, solar energy, and pharmaceutical and biotechnology industries—have much higher purity requirements than in past decades. The constant pressure to produce higher-purity products creates a demand for new separation processes or technologies. Furthermore, scientists are identifying challenging separations that could yield valuable products or resources. For example, although uranium concentrations in seawater are extremely low (parts per billion), some estimate that more than 4 billion tons of uranium could be extracted from seawater and used for nuclear power if efficient separation techniques could be developed (Sholl and Lively, 2016).
Opportunities for a Paradigm Shift. As will be described in Chapter 3, important advances have occurred in the last 30 years in molecular modeling and simulation, machine learning and data analytics, analytical techniques, and characterization, especially for short time and length scales. Those advances tantalize us with the prospect of a greatly improved theoretical and mechanistic understanding of separation processes, improved modeling and pre-
diction of chemical behaviors, and the exploration and development of new chemicals, materials, and approaches. Thus, separation science is poised for a paradigm shift in which the brightest minds in chemistry, chemical engineering, materials science, and other fields will be vying to achieve the breakthroughs in the next generation of separation science, and conventional separation technology will be replaced with high-throughput, energy-efficient, and exquisitely selective separation systems.
THE 1987 REPORT
Given the motivations described above, it is time for a compelling vision and strategy for separation science. Such a vision was last offered in Separation and Purification: Critical Needs and Opportunities (NRC, 1987).1 That report highlighted several key technological challenges: commercializing biotechnology, reducing dependence on foreign sources of critical and strategic metals, protecting the environment, meeting the demands for ultrapure materials, and developing alternative energy sources and feedstocks. The report noted that those endeavors had many features in common, such as the need to isolate products or contaminants from dilute solutions, the need to separate complex mixtures, and the need to replace energy-intensive processes. Given the commonalities, the report concluded that separations could be substantially improved by focusing on six generic research themes in which concentrated efforts could “lead to clearer insights into fundamental principles and major opportunities for technological innovations” (NRC, 1987, p. 44); the research themes are listed in Box 1-2 and discussed in more detail in Chapter 2 of the present report.
The 1987 report highlighted the idea that research activities are fragmented and that better communication, idea exchange, and technology transfer among the disciplines engaged in separation science research are needed. The report recommended a conference on separation science or formation of a new professional society for separation research to facilitate collaborative efforts. It also recommended modification of educational courses in traditional undergraduate disciplines (chemical engineering, chemistry, and biochemistry) and development of a cross-disciplinary curriculum in separation science and technology. And it recommended increased efforts to collect, evaluate, correlate, and disseminate physicochemical data so that more powerful predictive models could be developed. The report concluded that “the importance and pervasiveness of separations throughout the U.S. economy indicate that a federal program of generic research… will have significant benefits to our economic competitiveness… A timely response is required if these opportunities are not to slip from our grasp” (NRC, 1987, p. 6).
CURRENT LANDSCAPE OF SEPARATION SCIENCE
Since publication of the 1987 report, separation science and technology have seen substantial advances. Perhaps the most dramatic example is the widespread commercial adoption of membrane-based separations for water desalination. Salt can be removed from water by evaporation, but that process requires enormous amounts of energy. Reverse-osmosis technology lowers the energy requirement substantially and therefore now dominates new commercial desalination installations (Elimelech and Phillip, 2011). Thousands of facilities now use that technology. A remarkable observation about modern reverse-osmosis membranes for desalination is that they require only twice the thermodynamic minimum energy. That implies that additional improvements in the technology will need to focus on such issues as the longevity of membranes, particularly with respect to fouling, pretreatment, and post-treatment conditions (Imbrogno et al., 2017). A second example of commodity-scale separations in which nonthermal methods are becoming widespread is the use of adsorption or membranes for separating oxygen or nitrogen from air. Cryogenic distillation is still the most economically competitive technology for these separations
___________________
1Separation and Purification: Critical Needs and Opportunities is referred to hereafter as the 1987 report.
at the largest scales and highest purities, but nonthermal separation methods are now favored in many settings, such as preservation of fruits, vegetables, and flowers and oxygen enrichment for health applications.
Important advances in separation technologies have occurred in applications in which the total feed volume is modest by commodity chemical standards but exquisite selectivity and the ability to manage extremely complex feeds are critical. An example is the tailoring of materials to capture trace radionuclides from nuclear waste (Wilmarth et al., 2011). It is characterized by radionuclides at trace concentrations in competition with bulk ions at 100,000-fold higher concentrations—which demands recovery requirements of greater than 99.9975% for Cs-137—and by extreme environments with high radiation fields and highly corrosive conditions. Designer solvents that use calixarene-crown molecules as highly specific sequestering agents (Dozol and Ludwig, 2010) reject almost all competing ions and deliver a nearly pure cesium borate product stream from the complex waste for vitrification in borosilicate glass (Moyer et al., 2005).
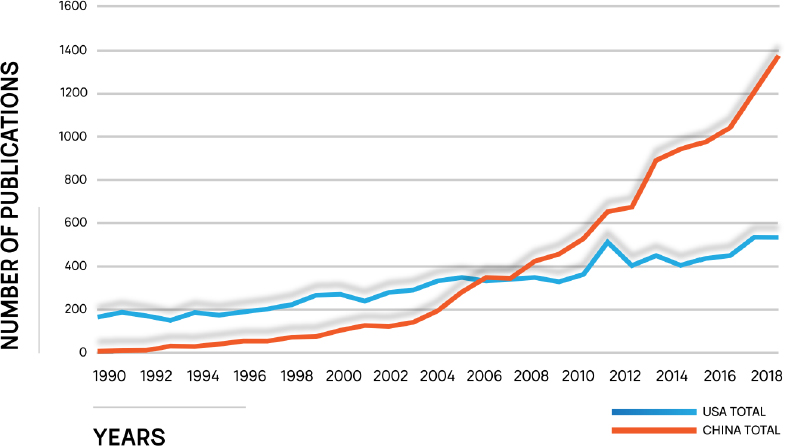
Regardless of the important advances that have occurred over the last few decades, there is a concern that the United States is losing a competitive edge in science and technology to China and India and this trend also is a concern for separation science. Among many possible metrics, the number of publications focused on separations2 by U.S. researchers has increased modestly. From 1990 to 2006, the United States outnumbered most countries in number of publications, but China has been the leading country in publications focused on separations since 2006 (see Figure 1-3). The U.S. loss of leadership in number of publications in the separation field might be caused by the number and size of programs that clearly target separations research.
Separation science is not highlighted in many chemistry and chemical engineering courses; thus, fewer scientists are exposed to the fundamentals of separation science and how the fundamentals can be applied. In a survey conducted by the American Institute of Chemical Engineers in 2018, respondents indicated that U.S. chemical engineering programs typically offer a dedicated class in separations to their undergraduates but that the class focuses primarily on distillation and other traditional separation technologies rather than on newer separation methods (such as use of membranes) or on the fundamental principles needed to understand separations.3 That approach might lead students to view separations as established technology and devoid of opportunities for exciting research advances. That idea is supported by the fact that only a minority of the top 10 chemical engineering PhD programs in the United States have any research efforts in separations. Graduate chemical engineering courses related to separations tend to be very specific (for example,
___________________
2 Key search terms used in SCOPUS were gas separations, liquid separations, separations and adsorption, separations and chromatography, separations and extraction, separations and purification, and separations and membranes.
3 The survey was conducted by the AIChE Education Division. Data were solicited from 166 chemical engineering programs in the United States that are accredited by the Accreditation Board for Engineering and Technology, Inc. The response rate was 36%, and the data are posted on the AIChE web site (see https://www.aiche.org/community/sites/divisions-forums/education-division/how-we-teach-surveys).
bioseparations or use of membranes) and in many cases are not offered regularly. Chemists in the United States usually take a separation course as part of the graduate curriculum but only if the school has a sufficiently large and active analytical chemistry program. The courses typically begin by studying equilibrium driving forces and thermodynamics, then move to studying flow and mass transport, and conclude by studying chromatography. Additional separation courses are normally not available or offered to chemistry students.
Furthermore, the number of academic chemists who have separation science as a major portion of their research portfolio has dropped by nearly 40% since the 1987 report;4 this demonstrates that there is a strong need to attract young colleagues to the field. Likewise, the number of chemical engineering faculty in top departments who can be clearly identified as separation researchers has decreased by 30% despite the large increase in the total number of faculty in those departments. The publication trends in separation science, the results of the AIChE survey, and the number of faculty currently in the field raise questions about whether the training and exposure of students to the fundamentals of separations needs to be re-evaluated and restructured and about whether current funding is sufficient for separation science. Those questions and others are discussed further in Chapter 6 of this report.
THE COMMITTEE AND ITS TASK
Given the need to provide a fresh vision for separation science, DOE, NIST, and NSF asked the National Academies to develop an agenda for fundamental research in separation science. Box 1-3 provides the verbatim statement of task. The committee convened as a result of the request included experts in chemistry and chemical engineering with specialties in materials science, analytical chemistry, computational and theoretical chemistry, interfacial chemistry, liquid and gas-phase separations, and industrial separation processes (see Appendix A for biographic information on the committee).
COMMITTEE’S INTERPRETATION OF ITS TASK
To accomplish its task, the committee held six meetings, including three data-gathering sessions during which it heard from experts and stakeholders in government agencies, industry (including trade associations), and the academic community. Topics explored during the data-gathering sessions included synthesis of new materials, external field effects, instrumentation, computational chemistry, and educational and societal needs. The committee also met with AltSep representatives during one of
___________________
4 These data were collected by committee members by identifying separation scientists and engineers in analytical chemistry and chemical engineering departments as listed in the 1987 American Chemical Society Directory of Graduate Research and current departmental Web sites. Separation scientists at all R1 (highest research activity) analytical chemistry programs were counted. Chemical engineering departments considered were those listed in 2018 by US News & World Report as the top 20 chemical engineering graduate programs.
the data-gathering sessions; AltSep is an initiative created to accelerate industrial adoption of less energy-intensive separation processes and is developing an industry-driven technology roadmap for separation alternatives to distillation.5 Agendas for the data-gathering sessions are provided in Appendix B. The committee also consulted published references from public sources, including the scientific literature and government reports.
The main topics discussed in this report are chemical, analytical, and biological separations, including those involving proteins and nucleic acids but excluding organelles, cells, and viruses. The committee considers a variety of separation techniques in this report but excludes mechanical or physical separations, such as centrifugation or depth filtration. The committee does not discuss distillation and evaporation in great detail because it considers those techniques to be mature technology and out of the scope of this research agenda.
The committee emphasizes that it was specifically tasked with developing a fundamental research agenda as opposed to undertaking a focused empirical investigation of particular processes. Understanding the fundamentals will lead to increased knowledge of how complex mixtures can be separated in a controlled manner and, therefore, advance the science. Advancing separation science will then enable solutions to many vexing societal problems. Although the time lag between fundamental research discovery and industrial commercialization might be several decades, fundamental discoveries are likely to benefit all aspects of chemical separations.
The use of separations spans many communities, each using a specific terminology. Accordingly, many of the key terms used in this report are defined in different ways by different communities. The committee notes that using different terminology to describe the same or similar phenomena creates roadblocks to translating advances from one field to another, and researchers need to improve understanding of terminology across the disciplines. In an effort to provide clarity to all readers, the committee defines in Box 1-4 key terms that it uses in this report.
___________________
5 AltSep is an initiative of the American Chemical Society Green Chemistry Institute Chemical Manufacturers Roundtable that was created with funding from the NIST Advanced Manufacturing Technology Consortia Program. See http://altsep.org/ for further details.
ORGANIZATION OF THE REPORT
This report is organized into six chapters and three appendixes. Chapter 2 discusses the efforts to advance separation science in the last 30 years. The committee describes advances made in the six primary research themes outlined in the 1987 report (see Box 1-2), discusses advances that were not anticipated by the 1987 report, and touches on workforce development and educational and industry needs. In a similar manner, Chapter 3 discusses relevant advances in the last 30 years in intersecting disciplines; it describes key fields of research that can provide insight and knowledge to advance separation science: materials synthesis, systems engineering, responses to external stimuli, instrumentation and characterization tools, and data science and analytics. Although much progress has been made, gaps and challenges remain, and Chapter 4 discusses the obstacles and sets the stage for the committee’s research agenda. Chapter 5 presents a research agenda that researchers and their funders can use to develop research programs in separation sciences. The committee’s research agenda outlines two high-priority research areas and two cross-cutting topics and describes the significance of moving research forward in those areas. Experimental, theoretical, and computational approaches and potential barriers are also discussed. Chapter 6 describes what it will take to implement the research agenda presented in Chapter 5. Specifically, it addresses educational needs in separation science, the need to create collaboration opportunities, the importance of access to tools at the national level, and the influence that progress in the research agenda will have on industry. It emphasizes the need to repopulate and rejuvenate the separations community to address the challenges detailed in Chapter 4 by implementing the research agenda offered in Chapter 5.
Appendix A provides the biographic information on the committee members, Appendix B provides the agendas of the committee’s open sessions, and Appendix C provides further detail on characterization and instrumentation.
REFERENCES
DOE (Department of Energy). 2015. Bandwidth Study on Energy Use and Potential Energy Saving Opportunities in U.S. Petroleum Refining. Washington, DC: U.S. Department of Energy.
Dozol, J.-F., and R. Ludwig. 2010. Extraction of radioactive elements by calixarenes. Ion Exchange and Solvent Extraction, edited by B. A. Moyer, Vol. 19. Boca Raton, Florida: CRC Press, 195–318.
Elimelech, M., and W. A. Phillip. 2011. The future of seawater desalination: Energy, technology, and the environment. Science 333(6043):712. doi: 10.1126/science.1200488.
Gulf Publishing. 2010. Hydrocarbon Processing: Petrochemical Processes 2010. Houston, TX: Gulf Publishing Company.
Humphrey, J., and G. E. Keller. 1997. Separation Process Technology. New York: McGraw-Hill.
Imbrogno, J., J. J. Keating IV, J. E. Kilduff, and G. Belfort. 2017. Critical aspects of RO desalination: A combination strategy. Desalination 401:68–87.
Moulijn, J. A., M. Makkee, and A. Van Diepen. 2001. Chemical Process Technology. Hoboken, NJ: John Wiley & Sons, Ltd., 109–123.
Moyer, B. A., J. F. Birdwell Jr., P. V. Bonnesen, and L. H. Delmau. 2005. Use of macrocycles in nuclear-waste cleanup: A real-world application of calixcrown in technology for the separation of cesium. Macrocyclic Chemistry—Current Trends and Future, edited by K. Gloe. Dordrecht, Netherlands: Springer, 383–405.
Neelis, M., Worrell., and E. Masane. 2008. Energy Efficiency Improvement and Cost Saving Opportunities for the Petrochemical Industry. Berkeley, CA: Lawrence Berkeley National Laboratory.
NRC (National Resource Council). 2008. Updated 2017. NAE Grand Challenges for Engineering. Washington, DC.
NRC (National Resource Council). 1987. Separations and Purification: Critical Needs and Opportunities. Washington, DC: National Academy Press.
NRC. 2006. Sustainability in the Chemical Industry: Grand Challenges and Research Needs. Washington, DC: The National Academies Press.
ORNL (Oak Ridge National Laboratories). 2005. Materials for Separation Technologies: Energy and Emission Reduction Opportunities.
Sholl, D., and R. Lively. 2016. Seven chemical separations to change the world. Nature 532:435–437. doi: 10.1038/532435a.
Wilmarth, W. R., G. J. Lumetta, M. E. Johnson, M. R. Poirier, M. C. Thompson, P. C. Suggs, and N. P. Machara. 2011. Review: Waste-pretreatment technologies for remediation of legacy defense nuclear wastes. Solvent Extraction and Ion Exchange 29:1–48. doi: 10.1080/07366299.2011.539134.
Worrell, E., L. Price, M. Neelis, C. Galitsky, and Z. Nan. 2007. World Best Practice Energy Intensity Values for Selected Industrial Sectors Berkeley, CA: Lawrence Berkeley National Laboratory.
Zydney, A. L. 2016. Continuous downstream processing for high value biological products: A review. Biotechnology and Bioengineering 113(3):465–475. doi: 10.1002/bit. 25695.








