3
Relevant Advances in Intersecting Disciplines
As described in Chapter 2, much progress has been made in separation science in the last 30 years, but many of the motivations for improvement in chemical separations remain. The new era of separation science will require working at the intersection of numerous fields of chemistry, engineering, materials science, physics, and information science. It will require understanding of advances in those disciplines and their application to separation challenges. This chapter discusses some of the advances in intersecting disciplines that present opportunities for separation science. The committee highlights advances in five categories: materials synthesis, systems-engineering approaches, using external stimuli, instrumentation and characterization, and data analytics and science.
ADVANCES IN MATERIALS SCIENCE FOR MATERIALS SYNTHESIS
The design and realization of materials with targeted applications have historically relied largely on trial and error.1 Driven by advances in theory and computation and new characterization tools, materials synthesis is rapidly transforming into a more science-based approach (DOE, 2016). For example, current capabilities are enabling the prediction of optimal molecular-scale structures for selective metal binding that are finding use in the design of new extractants (Bryantsev and Hay, 2015). Retrosynthetic techniques, combined with emerging machine-learning capabilities, are providing guidance in synthesizing predicted organic molecules (Szymkuć et al., 2016). But rational approaches to synthesis of even simple inorganic or hybrid materials are still in their infancy.
In early developmental stages in the materials community are the theoretical, computational, and experimental components necessary for design and synthesis of many of the complex, hierarchical materials in use and envisioned for advanced separations. Those components include capabilities for prediction of optimal pore sizes and their distribution in framework structures, of porous polymer monoliths, and of structured fluid components, such as reverse micelles and biphasic emulsions. Efforts are under way to merge theories that span diverse length scales and experimental approaches to synthesize complex materials. Progress will require coupling atomistic simulation techniques, such as density functional theory and molecular dynamics simulations, with aggregate interactions (coarse-grained molecular dynamics), lattice-Boltzmann methods, and computational fluid dynamics to provide predictive materials requirements that span the multiple scales of the interactions that drive many separation processes.
The general inability to synthesize materials predicted by theory and computation constitutes a bottleneck to progress (DOE, 2016). The most promising avenues for advancement include new capabilities for monitoring syntheses as they proceed. Combining multiple experimental tools that probe structural and electronic signatures in situ is expected to provide a perspective that is not now available for synthesis design. Use of that information, combined with emerging machine-learning tools, will allow operando control over reaction outcome. It will also provide information about potential material instabilities, competing phases, and potential failure or decomposition modes and lead to insights into how to minimize energy and waste costs related to targeted materials synthesis. Those efforts, which span the broad materials community and are discussed in detail in a recent U.S. Department of Energy report, Basic Research Needs for Synthesis Science (2016), will have a direct effect on the synthesis opportunities available to the separations community.
___________________
1 The word materials in this report includes both hard and soft materials that are crystalline, amorphous, composite, or liquid.
ADVANCES IN SYSTEMS-ENGINEERING APPROACHES
In the last 3 decades, the number of new materials synthesized has greatly increased, owing in part to advances in materials science, as discussed in the previous section. Some of the new materials have found applications in separations. Examples include organic-templated high-silica zeolites, which found their way to such applications as NOx-emission abatement (Lobo et al.,1993; Zones et al., 1998); aluminosilicate zeolite membranes that are used for dehydration applications (Kita et al., 2001); polymeric and carbon molecular sieve membranes that are being considered for gas- and liquid-based (non–phase change) separations (Koh et al., 2016); ionic liquids (Hong et al., 2016); and metal-organic frameworks (MOFs) (Bachman et al., 2016; Denny et al., 2016; Yaghi et al., 2003) that are considered selective adsorbents. More recently, various compositions of two-dimensional materials—such as zeolites, graphene, graphene oxide (Li et al., 2013), MXenes, and polymeric brushes (Moses et al., 2014; Keating et al., 2016)—have been proposed as promising membrane materials. Finally, biomimetic materials are being developed. An example is an aquaporin protein complex that has been used to prepare pseudo-synthetic membranes by embedding the complex into a synthetic porous membrane for desalination of brackish water (Zhao et al., 2012). Long-term stability of the protein aquaporin separating machines in vitro has been reported to be about 100 days with good performance (Qi et al., 2016), although using protein-based systems under environmental conditions will still be a challenge. There are also synthetic mimics of aquaporin channels with self-assembled imidazole molecules that have formed oriented chiral water wires (Kocsis et al., 2018). Producing mimics of aquaporin channels that have the required salt selectivity, sufficient permeation fluxes (volume flow rate per unit area) and extended lifetimes, however, will involve substantial effort.
Despite the growth in new materials, the corresponding effect on separation applications has lagged because of such difficulties as cost-effective manufacturing and scale-up, performance and stability limitations, and, most important, the inability of relating the structure of a material to its performance. More specifically, a single metric is not sufficient to determine whether a material is ready for industrial use. A complex compromise is often desirable and is dictated by process design, robustness, controllability, material cost, scalability, and performance stability. Therefore, an integrated approach that combines process synthesis2 and emerging materials synthesis and characterization technologies would improve the connection of separation materials and performance.
One can envision that materials development using process-driven materials discovery will become feasible. In that approach, one would set separation performance metrics on the basis of process needs, and design and synthesize or select materials that can optimally meet the performance targets. That integrated approach relies on large-scale process synthesis computation methods and accurate structure–property relations that are based on simulations, in situ and operando characterization, and performance measurements (see Figure 3-1). Excellent examples of such an integrated approach for separation problems are found in Pereira et al. (2011) and Hasan et al. (2013).
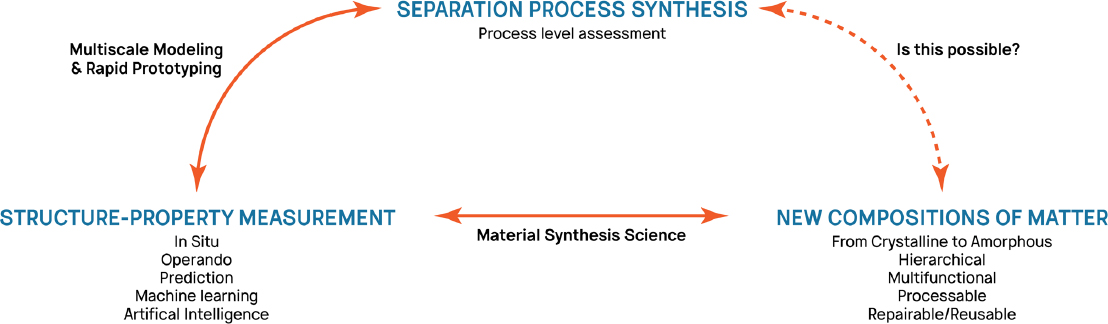
___________________
2 Process synthesis is a technique for developing a chemical process from concept to flowsheet. It encompasses an experimental program, modeling of experimental results, choice of processing units, how they are interconnected, and optimization of the proposed plant (Noble and Agrawal, 2005).
ADVANCES IN USING EXTERNAL STIMULI
There are opportunities to achieve separations by using unconventional stimuli. In general, controlling field gradients to achieve separations is a nascent field. The committee reviewed the use of electric and magnetic fields, which are both unconventional stimuli. Field-flow fractionation is an established use of a field for separations but has seen some recent advances. In addition, photoregulation is an important tool that can be used in separations. Four examples of the use of these unconventional drivers are discussed below.
Electric Fields
Recent work has shown that an external electric field can be applied to a separation system to control specific properties (such as transport and structure), mitigate fouling, and preconcentrate samples. For example, structural transformations of porous materials were experimentally observed after an electric field was applied (Knebel et al., 2017). Zeolitic imidazolate framework-8 (ZIF 8) crystals under an external electric field were reversibly transformed to stiffer, less permeable, but (for some mixtures) more selective polymorphs. It is not fully understood, but electric field–induced switching appears to be widely applicable in MOFs (Fernandez et al., 2014) and extends the range of materials with porosity—beyond amorphous polymers and modified mesoporous materials—that can be reversibly tuned by external fields (Fu et al., 2003; Cohen Stuart et al., 2010; Theato et al., 2013).
On a different scale, membrane fouling has been mitigated experimentally by an electric potential applied on the surface of conductive carbon membranes. The reduced fouling was attributed in some cases to field-induced changes in structure and surface tension (Zhu et al., 2018) or to repulsive forces between the charged membrane surface and charged organic foulants (Dudchenko et al., 2014).
Another application of electric fields is in controlling sample concentration and sample cleanup. Enrichment factors as high as 100 have been obtained by using microfluidic preconcentration devices via dielectrophoresis or electrodialysis (Wuethrich et al., 2016). Capacitive deionization is an example of a novel means of desalination (Ahmed and Tewari, 2018); it uses two porous electrodes with a low applied potential. More studies of the fundamental processes involved in such separations will likely provide enhanced enrichment and scaling of preconcentration approaches.
Magnetic Fields
Magnetoelectrophoresis and magnetophoresis have recently been applied in separation science. Magnetoelectrophoresis uses a combination of electric and magnetic fields and has been used in cell sorting and particle sorting by attaching magnetic beads to the cells or particles of interest. Magnetophoresis involves separation of a magnetized material induced by the presence of an external magnetic field. Recent studies showed that paramagnetic ions in the entire lanthanide series can be separated selectively by exposing concentrated droplets of the ions to an external magnetic field (Rodrigues et al., 2018); this discovery might lead to further enhanced selective separations of paramagnetic ions.
Field-Flow Fractionation
Field-flow fractionation (FFF) involves influencing components flowing in a channel by subjecting them to a field—which could be another flow field—that affects component mobility. Although it was invented in the 1970s (Giddings et al., 1976), recent advances have expanded its potential. High-temperature, asymmetric FFF has permitted the analysis of polymers; for example, thermal FFF has exhibited high resolution in the separation of polymers that have molecular weights over 100 kDa (Gunderson and Giddings, 1986). Two-dimensional separations that combine FFF with size-exclusion chromatography or mass spectrometry have been successful (Van Asten et al., 1995; Basile et al., 2005; Messaud et al., 2009). In addition, advances in instrumentation and theoretical developments might drive researchers to apply FFF to other separation challenges, particularly in the analysis of nanomaterials (Williams et al., 2011).
Photoregulation
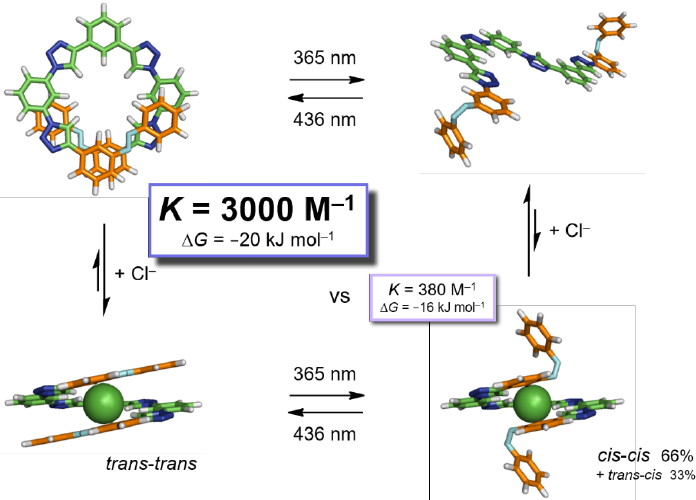
Photoregulation has recently been used to control pore shape and size of porous materials, and this could enable more selective separations. Covalent organic frameworks with integrated thermally reversible stimulus-responsive π-units exhibit structural transformations on photostimulation, which leads to tunable gas storage capacity (Lyndon et al., 2013; Huang et al., 2015). Similarly, a pore entrance can be functionalized with a photoswitchable molecular gate that can permit selective molecular sieving during adsorption but allows easy desorption on switching the gate to the open configuration (Zhu et al., 2016). As Figure 3-2 shows, light can also trigger the binding and release of ions.
In addition to having tunable pore shape and size, separation materials can be designed to exhibit a photo-response on interaction with light. Nayak et al. (2006) demonstrated the reversible conversion of a photografted, photoresponsive, polymeric synthetic membrane surface from polar to nonpolar. A moiety with switchable ultraviolet-visible characteristics, such as a spiropyran, was grafted onto a commercially available photoactive poly(ether)sulfone polymer by using a simple photograft polymerization method.
ADVANCES IN INSTRUMENTATION AND CHARACTERIZATION
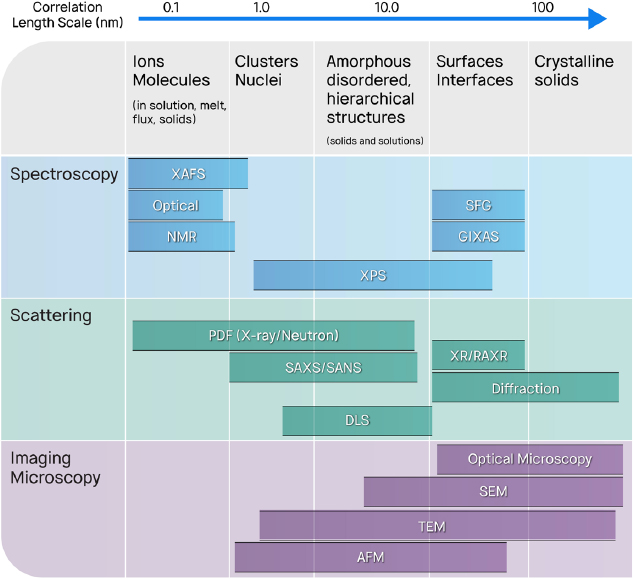
Generally, three classes of experiments based on spectroscopic, scattering, and imaging (including microscopy) techniques are commonly deployed to characterize structures on length scales from the molecular to the nanoscale to the microscopic. As briefly discussed below, each class of experiments has the potential to elucidate fundamental insights on cutting-edge scientific questions, which are needed to advance the understanding of separation systems. Relevant examples illustrate the kinds of information now obtainable with advanced instrumentation.
This instrumentation is constantly under development. Many instruments are at and enabled by investments in U.S. national user facilities. As mentioned in Chapter 2, user facilities are important resources that are available for external use to advance scientific or technical knowledge.
Spectroscopic Techniques
Spectroscopic techniques rely on the response of a system to variable wavelength probes, with energies spanning from the infrared to x-ray. Many of the techniques, including conventional optical spectroscopy, have long been used as indirect probes of changing electronic and structural speciation. The results of those studies have been used to populate a variety of databases, notably those devoted to chemical speciation and thermodynamic stabilities. The availability of tunable x-ray sources through national user programs presents an underused opportunity to expand on the analytical and structural information to probe separation systems. Simplest among these capabilities is x-ray fluorescence (XRF) spectroscopy, which provides analytical information down to low concentrations by measuring the characteristic emission line of a targeted element. The recent availability of high-flux, widely tunable x-rays provided by a synchrotron source at a national facility, with in-house XRF instruments, allows access to a wider array of elements than those available previously (West et al., 2009). Additional advantages of national facilities are time and resource savings through the use of energy-dispersive techniques that permit the simultaneous acquisition of data from different elements. Researchers can also do spatial distribution mapping, including depth profiling. Generally, such capabilities are continually expanding to allow measurements in increasingly complex and relevant chemical environments, as illustrated by the emergence of such techniques as ambient-pressure x-ray photoelectron spectroscopy. Collectively, these capabilities could be used, for example, to address issues that limit capacity, such as membrane fouling.
Other uses of x-ray sources include x-ray absorption spectroscopy (XAS or XAFS), which combines both x-ray near edge structure and extended x-ray absorption fine structure (EXAFS). These techniques scan the incident photon energy about a selected element’s K or L absorption edge such that the resulting standing-wave interference pattern and how it changes with energy provide quantitative information about the absorbing ion’s near-neighbor number and distance.
EXAFS as part of XAS has recently become a go-to spectroscopic technique for determining a selected ion’s speciation (for example, coordinating ligands and their number and distance) at concentrations as low as parts per million in gases, liquids, or solids. It provides direct structural information on noncrystalline systems with an approximate accuracy of 10% of the coordination number and 1% of interatomic distances, even for species as far as 0.6 nm from the absorbing ion. The technique has received increasing use in separation-related research, providing information critical for increasing selectivity through modeling and simulation. For example, EXAFS was used in a fundamental study to elucidate competitive coordination of lanthanides with neutral and acidic organophosphorus extractants and revealed that their ratios depend on solution conditions (Braatz et al., 2017). From a more practical standpoint, speciation is important for the selective removal of contaminants, as exemplified by the information obtained about vanadium speciation in heavy oils and bitumens, the elucidation of which played an important role in understanding impediments to its effective removal (Dechaine et al., 2010). Unfortunately, XAFS spectroscopy is generally limited to absorption edges for elements heavier than Si (that is, with atomic number greater than 14). Thus, speciation information on lighter elements, such as C (atomic number 6), can be only indirectly probed by using their interactions with neighboring heavier elements that absorb.
Nuclear magnetic resonance (NMR) spectroscopic techniques rely on the interaction of an external magnetic field with the spins of nuclei in analyte molecules. It provides considerable information on the functionalities present in analytes and is capable of providing information on dynamics (for example, from relaxation times or deuterium NMR). It can also be used to measure diffusion coefficients with pulsed field gradient NMR (PFG-NMR); PFG-NMR provides information on length scales up to 1 micron (Pagès et al., 2017). High temperature in situ NMR is increasingly used to improve understanding of catalysis (Zhang and Weitz, 2012), and high-pressure, continuous-flow NMR has been used to detect analyte species that have been separated with high-pressure liquid chromatography (Anders et al., 2018). However, in situ, continuous-flow, high-pressure systems that can monitor interactions in separation systems are needed to provide more detailed information on changing speciation and for simultaneous monitoring of flow dynamics caused by ordering within separation media (Teisseyre et al., 2011).
Scattering Techniques
In contrast with spectroscopic studies, scattering techniques generally involve treatment of light–matter interactions as momentum changes of a scattered photon (particle) as opposed to the energy changes seen in spectroscopy. Scattering techniques probe structural correlations, ordering, and morphology over a broad range of distances, from atom–atom interactions (as described by pair distribution functions) to bulk single-crystal structuring to macromolecular and aggregate structures. The common techniques include dynamic light scattering, as well as small-angle x-ray scattering (SAXS), small-angle neutron scattering (SANS), wide-angle scattering, and high-energy scattering (HEXS). The singular advantage of scattering techniques is their ability to quantify interaction with high accuracy. For example, in dealing with solutions, only HEXS data combine accuracy in ligating-species number with structural detail sufficient for use in directly determining thermodynamic stability constants and free energies (Soderholm et al., 2011). This approach provides critical experimental information, needed by theorists and modelers, that directly links structure with energetics in solution and is important for the understanding of energy landscapes. That capability is particularly important in developing new scenarios for solvent extraction. With a multimodal approach, SAXS has been combined with EXAFS to provide unprecedented insights into hierarchical solvent structuring (Motokawa et al., 2019). Short- and long-range solute and solvent correlations have been used to inform understanding of third-phase formation, a common problem that limits capacity in solvent extraction processes.
Imaging Techniques
Imaging techniques provide information about structuring on nanoscopic to microscopic length scales.3 Separation materials, particularly porous polymers and membranes, have been widely studied with atomic-force microscopy (AFM) and optical and electron microscopy. AFM relies on the displacement of a cantilever as a result of its interaction with the surface to deflect a laser beam. AFM can image surfaces at atomic-scale resolutions (Binnig, 1988) and has been exceedingly important in probing the structure of solid separation materials.
Likewise, transmission electron microscopy (TEM) in ultrahigh vacuum has been widely used in nanoscience. TEM involves transmitting a beam of electrons through a sample to form an image. The last decade has seen dramatic advances in the use of TEM in the liquid phase (liquid-cell TEM). Nanofabricated liquid cells, surrounded by high vacuum, allow penetration of an electron beam with minimal scattering. Figure 3-3 depicts the imaging of individual nanocrystals of platinum as they grow in a 20:1 mixture of o-dichlorobenzene and oleylamine
___________________
3 The information is rendered in real Cartesian-coordinate space, as opposed to the reciprocal or momentum space data that are available from scattering experiments. Reciprocal space data must be mathematically manipulated into real space coordinates by using Fourier transform.
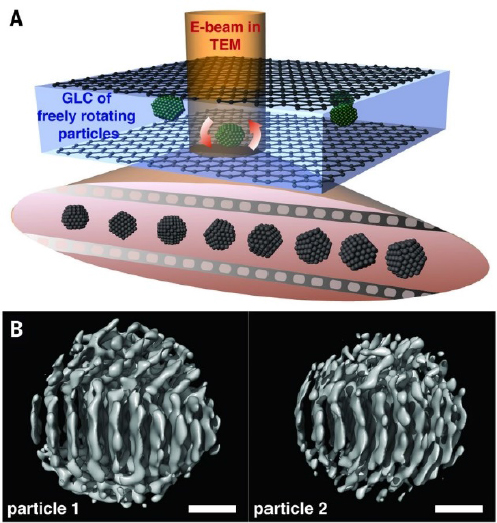
between graphene sheets (Park et al., 2015). Advantages of TEM over AFM include its high speed with 0.56 Å resolution, which images many particles simultaneously, and the three-dimensional information gained from the electron diffraction, which enables the probing of the bulk nanocrystal structure. Liquid-cell TEM has also been used to study the nucleation and growth of two MOFs: ZIF-8 and UiO-66 (Patterson et al., 2015).
Imaging that uses beams from national synchrotron and neutron user facilities has two advantages over many other available techniques: the beams’ penetrating power allows them to provide information about the bulk sample, and their shorter wavelengths allow better resolution, currently within the low-nanometer regime. Accessible information includes pore size and dispersity and preferences in pore loading and analyte distribution. The use of monolithic polymers as stationary phases in separations is a case in which advances in performance require knowledge of structure and chemistry. Initial characterization by neutron-scattering experiments was able to determine pore characteristics (Ford et al., 2011). Follow-up experiments on similar samples with scanning transmission x-ray microscopy revealed the capability of obtaining the spatial distribution of chemical constituents within the material (Arrua et al., 2014). As a result, the potential new opportunities for combining scattering and imaging techniques are now beginning to be appreciated in the separations community, and their use is expected to grow (Da Silva et al., 2017).
The understanding of separations is ultimately limited by the unknown molecular-scale structures of the materials. Separations have extensively used nanomaterials for over a half century without the benefit of imaging on the scale of angstroms. For example, liquid chromatography uses nanoparticles of silica with diameters of 5–500 nm, which aggregate to form the porous silica microparticles that dominate the industry. The speed of liquid chromatography, especially size-exclusion chromatography, depends on the molecular-scale pore structures formed by the aggregated nanoparticles, but these structures have been beyond the limits of direct observation and characterization. With MOFs, the growth process itself determines the performance of the material, but there has been no method to observe growth directly without seeding the growth mixture and without sacrificing measurement speed.
A wide array of experimental techniques of spectroscopy, scattering, and imaging have recently become available to the separations community to characterize structural features over a broad range of lengths. Figure 3-4 outlines the techniques available on different length scales, ranging from 0.1 to 100 nm. Detailed examples of the use of the various techniques on various scales are described in Appendix C. Combining the techniques in a multimodal approach offers an emerging opportunity to link structure with separation efficacy, including selectivity, capacity, and robustness.
ADVANCES IN DATA SCIENCE AND ANALYTICS
Data science is a broad and historically important field that incorporates robust statistical methods and more recently developed techniques that leverage modern computing architectures to learn from large and varied datasets (Figure 3-5). Much of the motivation for advances in data science results from the dramatic increase in data collected from experiment and simulation. “Big data” is certainly relevant to separation science and can be the output of operando or in situ measurement over long periods or can consist of combined data from multimodal approaches to instrumentation.
However, not all data-science challenges are related to big data. Indeed, the merger of different types of data into heterogeneous sets that span different time and length scales, or even different types of observables, leads to substantial challenges and to opportunities in the realm of separations. As noted throughout this report, no single metric defines the efficacy of a separation system. In the context of data science, researchers must be prepared to explore and determine complex correlations in structural, dynamic, and performance assessments. The data-science approaches needed to tackle such challenges extend beyond traditional machine learning methods and might require a combination of approaches that include clustering or topological analysis used independently or combined with learning algorithms.
The last decade has seen vast increases in the amount of data produced by experiment and simulation. Although the data contain valuable information that can provide scientific insight and uncertainty quantification, data-science methods need to be used to refine and optimize experimental approaches and elucidate critical data gaps. However, elucidating multicomponent interactions, for example, greatly expands the need for data. Given constrained resources, conditions chosen for study need to be identified rationally to maximize the amount of useful information generated for the effort expended.
At national user facilities, new capabilities have increased the number of scientists that are performing time-resolved, combinatorial, or high-throughput experiments. In situ, time-resolved experiments need real-time feedback; it is now possible to perform real-time, large-scale simulations to interpret results of some techniques and allow real-time decision-making. From a simulations per-
spective, major investments in the user-facility infrastructure are propelling modeling from idealized model systems to models of chemistry and materials that approach experimental complexity. In combination, this data revolution now lets the broader scientific community consider mechanisms for exploring data in new ways, merge and analyze heterogeneous datasets (with different time or length scales or resolution), and migrate from the realm of data for analysis to data for prediction. Concomitantly, massive research efforts have been dedicated to fundamental data-science problems associated with uncertainty quantification, sensitivity analysis, and the development of new numerical models from simulation and experimental outputs. Although basic data-science algorithms as deployed by the statistics community are well established, their application to complex chemical problems is now accelerating materials design, materials synthesis, and the understanding of complex hierarchical correlations, from molecular-scale interactions up to transport phenomena.
Data analytics is still in its infancy as it pertains to applications in separations from the molecular scale to process scale. For example, consider the potential of dimensionality reduction as it pertains to identifying the essential or top characteristics of massively successful materials and failed materials. Persistent homology has not been widely used to identify the shared structural features of phases of matter and could reflect trends in intermolecular interactions. And there are important opportunities to expand the scope of learning algorithms to mapping energy landscapes for the chemical and materials transformations that occur during aging, which will also advance separation science.
CONCLUSION
The advances in intersecting disciplines that are described in this chapter, when applied to separation science, will move the field forward. The advent of powerful computational and experimental tools and approaches in various disciplines provide both an opportunity and a dilemma for separation scientists. The opportunity is to gain previously unattainable understanding and control on scales ranging from molecular to systems. The dilemma is two-fold. Exploiting new techniques requires a level of expertise that is often beyond the capability of individuals or research groups. Many of the tools are currently too sophisticated for separation scientists to be simply “users” of the tools, so there are challenges in training, accessibility and collaboration. The committee also emphasizes that often the terminology to describe the same or similar phenomena differs in ways that create roadblocks to translating advances from one field to another. Researchers need to improve the understanding of terminology across the disciplines. Finally, new discoveries often yield more unsolved gaps and challenges. Currently identifiable gaps and challenges in separation science are discussed in Chapter 4.
REFERENCES
Ahmed, M. A., and S. Tewari. 2018. Capacitive deionization: Processes, materials and state of the technology. Journal of Electroanalytical Chemistry 813:178–192. doi: https://doi.org/10.1016/j.jelechem.2018.02.024.
Anders, J., J. G. Korvink, and A. Webb. 2018. Small-volume hyphenated NMR techniques. Micro and Nano Scale NMR, edited by J. Anders and J. G. Korvink. Hoboken, NJ: John Wiley & Sons.
Arrua, R. D., A. P. Hitchcock, W. B. Hon, M. West, and E. F. Hilder. 2014. Characterization of Polymer Monoliths Containing Embedded Nanoparticles by Scanning Transmission X-ray Microscopy (STXM). Analytical Chemistry 86(6):2876-2881. doi: 10.1021/ac403166u.
Bachman, J. E., Z. P. Smith, T. Li, T. Xu, and J. R. Long. 2016. Enhanced ethylene separation and plasticization resistance in polymer membranes incorporating metal–organic framework nanocrystals. Nature Materials 15:845. doi: 10.1038/nmat4621.
Banerjee, D., C. M. Simon, A. M. Plonka, R. K. Motkuri, J. Liu, X. Chen, B. Smit, J. B. Parise, M. Haranczyk, and P. K. Thallapally. 2016. Metal–organic framework with optimally selective xenon adsorption and separation. Nature Communications 7. doi: 10.1038/ncomms11831.
Basile, F., G. E. Kassalainen, and S. K. R. Williams. 2005. Interface for direct and continuous sample-matrix deposition onto a MALDI probe for polymer analysis by thermal field flow fractionation and off-line MALDI-MS. Analytical Chemistry 77(9):3008–3012. doi: 10.1021/ac048391d.
Belfort, G. 2019. Membrane filtration with liquids: A global approach with prior successes, new developments and unresolved challenges. Angewandte Chemie International Edition 58(7):1892–1902. doi: 10.1002/anie.201809548.
Binnig, G. K. Atomic force microscope and method for imaging surfaces with atomic resolution. U.S. Patent 4724318 filed August 4, 1986 and issued February 9, 1988.
Braatz, A. D., Antonio, M. R., Nilsson, M. 2017. Structural study of complexes formed by acidic and neutral organophosphorus reagents. Dalton Transactions 46(4): 1194-1206.
Brand, S. K., J. E. Schmidt, M. W. Deem, F. Daeyaert, Y. Ma, O. Terasaki, M. Orazov, and M. E. Davis. 2017. Enantiomerically enriched, polycrystalline molecular sieves. Proceedings of the National Academy of Sciences 114(20):5101–5106. doi: 10.1073/pnas.1704638114.
Bryantsev, V. S., and B. P. Hay. 2015. Theoretical prediction of Am(iii)/Eu(iii) selectivity to aid the design of actinide-lanthanide separation agents. Dalton Transactions 44(17):7935–7942. doi: 10.1039/C4DT03275F.
Cohen Stuart, M. A., W. T. S. Huck, J. Genzer, M. Müller, C. Ober, M. Stamm, G. B. Sukhorukov, I. Szleifer, V. V. Tsukruk, M. Urban, F. Winnik, S. Zauscher, I. Luzinov, and S. Minko. 2010. Emerging applications of stimuli-responsive polymer materials. Nature Materials 9:101. doi: 10.1038/nmat2614.
Da Silva, C. G. A., C. B. G. Bottoli, and C. H. Collins. 2017. 3-Dimensional X-ray microtomography methodology for characterization of monolithic stationary phases and columns for capillary liquid chromatography—A tutorial. Analytica Chimica Acta 991:30–45. doi: 10.1016/j. aca.2017.08.040.
Dechaine, G. P. and Gray, M. R. 2010. Chemistry and Association of Vanadium Compounds in Heavy Oil and Bitumen, and Implications for Their Selective Removal. Energy Fuels 24(5):2795-2808.
Denny Jr., M. S., J. C. Moreton, L. Benz, and S. M. Cohen. 2016. Metal–organic frameworks for membrane-based separations. Nature Reviews Materials 1:16078. doi: 10.1038/natrevmats.2016.78.
DOE. 2016. Basic Research Needs for Synthesis Science. Report of the Basic Energy Sciences Workshop on Basic Research Needs for Synthesis Science for Energy Relevant Technology, Washington, DC, May 2–4, 2016.
Dudchenko, A. V., J. Rolf, K. Russell, W. Duan, and D. Jassby. 2014. Organic fouling inhibition on electrically conducting carbon nanotube–polyvinyl alcohol composite ultrafiltration membranes. Journal of Membrane Science 468:1–10. doi: 10.1016/j.memsci.2014.05.041.
Fernandez, C. A., P. C. Martin, T. Schaef, M. E. Bowden, P. K. Thallapally, L. Dang, W. Xu, X. Chen, and B. P. McGrail. 2014. An electrically switchable metal-organic framework. Scientific Reports 4:6114. doi: 10.1038/srep06114.
Ford, K. M., B. G. Konzman, and J. F. Rubinson. 2011. A More Informative Approach for Characterization of Polymer Monolithic Phases: Small Angle Neutron Scattering/Ultrasmall Angle Neutron Scattering. Analytical Chemistry 83(24):9201-9205. doi: 10.1021/ac202238r.
Fu, Q., G. V. R. Rao, L. K. Ista, Y. Wu, B. P. Andrzejew-ski, L. A. Sklar, T. L. Ward, and G. P. López. 2003. Control of molecular transport through stimuli-responsive ordered mesoporous materials. Advanced Materials 15(15):1262–1266. doi: 10.1002/adma.200305165.
Giddings, J. C., F. J. Yang, and M. N. Myers. 1976. Flow-field-flow fractionation: A versatile new separation method. Science 193(4259):1244–1245. doi: 10.1126/science.959835.
Gunderson, J. J., and J. C. Giddings. 1986. Comparison of polymer resolution in thermal field-flow fractionation and size-exclusion chromatography. Analytica Chimica Acta 189(C):1–15. doi: 10.1016/S0003-2670(00)83710-1.
Hasan, M. M. F., E. L. First, and C. A. Floudas. 2013. Cost-effective CO2 capture based on in silico screen of zeolites and process optimization. Physical Chemistry Chemical Physics 15(40):17601–17618.
Hong, B., L. D. Simoni, J. E. Bennett, J. F. Brennecke, and M. A. Stadtherr. 2016. Simultaneous process and material design for aprotic N-heterocyclic anion ionic liquids in post-combustion CO2 capture. Industrial & Engineering Chemistry Research 55(30):8432–8449.
Hua, Y., and A. H. Flood. 2010. Flipping the switch on chloride concentrations with a light-active foldamer. Journal of the American Chemical Society 132(37):12838–12840. doi: 10.1021/ja105793c.
Huang, N., X. Ding, J. Kim, H. Ihee, and D. Jiang. 2015. A photoresponsive smart covalent organic framework. Angewandte Chemie International Edition 54(30):8704–8707. doi: 10.1002/anie.201503902.
Keating, J. J., J. Imbrogno, and G. Belfort. 2016. Polymer brushes for membrane separations: A review. ACS Applied Materials & Interfaces 8(42):28383–28399. doi: 10.1021/acsami.6b09068.
Kita, H., K. Fuchida, T. Horita, H. Asamura, and K. Okamoto. 2001. Preparation of Faujasite membranes and their permeation properties. Separation and Purification Technology 25(1):261–268. doi: 10.1016/S1383-5866(01)00110-1.
Knebel, A., B. Geppert, K. Volgmann, D. I. Kolokolov, A. G. Stepanov, J. Twiefel, P. Heitjans, D. Volkmer, and J. Caro. 2017. Defibrillation of soft porous metal-organic frameworks with electric fields. Science 358(6361):347–351. doi: 10.1126/science.aal2456.
Kocsis, I., M. Sorci, H. Vanselous, S. Murail, S. E. Sanders, E. Licsandru, Y.M. Legrand, A. van der Lee, M. Baaden, P.B. Petersen, G. Belfort, and M. Barboiu. 2018 Oriented chiral water wires in artificial transmembrane channels. Sci Adv 4(3):eaao5603.
Koh, D.-Y., B. A. McCool, H. W. Deckman, and R. P. Lively. 2016. Reverse osmosis molecular differentiation of organic liquids using carbon molecular sieve membranes. Science 353(6301):804–807. doi: 10.1126/science.aaf1343.
Laher, M., T. J. Causon, W. Buchberger, S. Hild, and I. Nischang. 2013. Assessing the nanoscale structure and mechanical properties of polymer monoliths used for chromatography. Analytical Chemistry 85(12):5645–5649. doi: 10.1021/ac401303k.
Li, H., Z. Song, X. Zhang, Y. Huang, S. Li, Y. Mao, H. J. Ploehn, Y. Bao, and M. Yu. 2013. Ultrathin, molecular-sieving graphene oxide membranes for selective hydrogen separation. Science 342(6154):95–98. doi: 10.1126/science.1236686.
Liang, X., S.-P. Ng, N. Ding, and C.-M. L. Wu. 2018. Enhanced hydrogen purification in nanoporous phosphorene membrane with applied electric field. The Journal of Physical Chemistry C 122(6):3497–3505. doi: 10.1021/acs.jpcc.7b12283.
Lobo, R. F., M. Pan, I. Chan, H.-X. Li, R. C. Medrud, S. I. Zones, P. A. Crozier, and M. E. Davis. 1993. SSZ-26 and SSZ-33: Two molecular sieves with intersecting 10- and 12-ring pores. Science 262(5139):1543–1546. doi: 10.1126/science.262.5139.1543.
Lyndon, R., K. Konstas, B. P. Ladewig, P. D. Southon, C. J. Kepert, and M. R. Hill. 2013. Dynamic photo-switching in metal–organic frameworks as a route to low-energy carbon dioxide capture and release. Angewandte Chemie International Edition 52(13):3695–3698. doi: 10.1002/anie.201206359.
Messaud, F.A.; Sanderson, R.D.; Runyon, J.R.; Otte, T.; Pasch, H.; Williams, S.K.R. 2009. Progress in Polymer Science. 34, 351-368.
Moses, K. J., and Y. Cohen. 2014. Wettability of terminally anchored polymer brush layers on a polyamide surface. Journal of Colloid and Interface Science 436:286–295. doi: 10.1016/j.jcis.2014.08.042.
Motokawa, R., Kobayashi, T., Endo, H., Mu, J., Williams, C. D., Masters, A. J., Antonio, M. R., Heller, W. T., Nagao, M. 2019. “A Telescoping View of Solute Architectures in a Complex Fluid System.” ACS Central Science 5(1):85-96.
Murad, S., and J. Lin. 2002. Using thin zeolite membranes and external electric fields to separate supercritical aqueous electrolyte solutions. Industrial & Engineering Chemistry Research 41(5):1076–1083. doi: 10.1021/ie010425+.
Nayak, A., H. Liu, and G. Belfort. 2006. An optically reversible switching membrane surface. Angewandte Chemie International Edition 45(25):4094–4098. https://doi.org/10.1002/anie.200600581.
Noble, Richard D., and Rakesh Agrawal. 2005. Separations Research Needs for the 21st Century. Industrial & Engineering Chemistry Research 44(9):2887-2892. doi: 10.1021/ie0501475.
Pagès, G., V. Gilard, R. Martino, and M. Malet-Martino. 2017. Pulsed-field gradient nuclear magnetic resonance measurements (PFG NMR) for diffusion ordered spectroscopy (DOSY) mapping. Analyst 142(20):3771-3796. doi: 10.1039/C7AN01031A.
Park, J., H. Elmlund, P. Ercius, J. M. Yuk, D. T. Limmer, Q. Chen, K. Kim, S. H. Han, D. A. Weitz, A. Zettl, and A. P. Alivisatos. 2015. 3D structure of individual nanocrystals in solution by electron microscopy. Science 349(6245):290. doi: 10.1126/science.aab1343.
Patterson, J. P., P. Abellan, M. S. Denny, C. Park, N. D. Browning, S. M. Cohen, J. E. Evans, and N. C. Gianneschi. 2015. Observing the growth of metal–organic frameworks by in situ liquid cell transmission electron microscopy. Journal of the American Chemical Society 137(23):7322–7328. doi: 10.1021/jacs.5b00817.
Pereira, F. E., E. Keskes, A. Galindo, G. Jackson and C. S. Adjiman. 2011. Integrated solvent and process design using a SAFT-VR thermodynamic description: High-pressure separation of carbon dioxide and methane. Computers and Chemical Engineering 35:474–491.
Qi, S. R. Wang, G.K.M Chaitra, J. Torres, X. Hu, and A. G. Fane. 2016. Aquaporin-based biomimetic reverse osmosis membranes: Stability and long term performance. J. Membr. Sci. 508:94-103.
Rodrigues, I. R., L. Lukina, S. Dehaeck, P. Colinet, K. Binnemans, and J. Fransaer. 2018. Magnetophoretic sprinting: A study on the magnetic properties of aqueous lanthanide solutions. The Journal of Physical Chemistry C 122(41):23675–23682. doi: 10.1021/acs.jpcc.8b06471.
Soderholm, L., Skanthakumar, S., Wilson, R. E. 2009. Structures and Energetics of Erbium Chloride Complexes in Aqueous Solution. The Journal of Physical Chemistry A 113(22):6391-6397.
Szymkuć, S., E. P. Gajewska, T. Klucznik, K. Molga, P. Dittwald, M. Startek, M. Bajczyk, and B. A. Grzybowski. 2016. Computer-assisted synthetic planning: The end of the beginning. Angewandte Chemie International Edition 55(20):5904–5937. doi: 10.1002/anie.201506101.
Teisseyre, T. Z., J. Urban, N. W. Halpern-Manners, S. D. Chambers, V. S. Bajaj, F. Svec, and A. Pines. 2011. Remotely detected NMR for the characterization of flow and fast chromatographic separations using organic polymer monoliths. Analytical Chemistry 83(15):6004–6010. doi: 10.1021/ac2010108.
Theato, P., B. S. Sumerlin, R. K. O’Reilly, and T. H. Epps III. 2013. Stimuli responsive materials. Chemical Society Reviews 42(17):7055–7056. doi: 10.1039/C3CS90057F.
Van Asten, A. C., R. J. van Dam, W. T. Kok, R. Tijssen, and H. Poppe. 1995. Determination of the compositional heterogeneity of polydisperse polymer samples by the coupling of size-exclusion chromatography and thermal field-flow fractionation. Journal of Chromatography A 703(1–2):245–263. doi: 10.1016/0021-9673(94)01165-B.
West, M., A. T. Ellis, P. J. Potts, C. Streli, C. Vanhoof, D. Wegrzynek, and P. Wobrauschek. 2009. Atomic spectrometry update. X-Ray fluorescence spectrometry. Journal of Analytical Atomic Spectrometry 24(10):1289–1326. doi: 10.1039/B915056K.
Williams, S.K.R., Runyon, J.R.; Ashames, A.A. 2011. Field-Flow Fractionation: Addressing the Nano Challenge. Analytical Chemistry, 83:634-642.
Wuethrich, A., P. R. Haddad, J. P. Quirino. The electric-field – an emerging driver in sample preparation. TrAC Trends in Analytical Chemistry. 80”604-611. doi: 10.1016/j.trac. 2016.04.016
Yaghi, O. M., M. O’Keeffe, N. W. Ockwig, H. K. Chae, M. Eddaoudi, and J. Kim. 2003. Reticular synthesis and the design of new materials. Nature 423:705. doi: 10.1038/nature01650.
Zhang, J., and E. Weitz. 2012. An in situ NMR study of the mechanism for the catalytic conversion of fructose to 5-hydroxymethylfurfural and then to levulinic acid using 13C labeled d-fructose. ACS Catalysis 2(6):1211–1218. doi: 10.1021/cs300045r.
Zhao, Y., C. Qiu., X. Li, A. Vararattanavech, W. Shen, J. Torres, C. Helix-Nielsen, R. Wang, X. Hu, and A.G. Fane. 2012. Synthesis of robust and high-performance aquaporin-based biomimetic membranes by interfacial polymerization-membrane preparation and RO performance characterization. Journal of Membrane Science 423:422-428.
Zhu, J., J.-J. Ding, X.-Q. Liu, P. Tan, and L.-B. Sun. 2016. Realizing both selective adsorption and efficient regeneration using adsorbents with photo-regulated molecular gates. Chemical Communications 52(21):4006–4009. doi: 10.1039/C5CC10634F.
Zhu, X., A. V. Dudchenko, C. M. Khor, X. He, G. Z. Ramon, and D. Jassby. 2018. Field-induced redistribution of surfactants at the oil/water interface reduces membrane fouling on electrically conducting carbon nanotube UF membranes. Environmental Science & Technology 52(20):11591–11600. doi: 10.1021/acs.est.8b02578.
Zones, S. I., Y. Nakagawa, G. S. Lee, C. Y. Chen, and L. T. Yuen. 1998. Searching for new high silica zeolites through a synergy of organic templates and novel inorganic conditions. Microporous and Mesoporous Materials 21(4):199–211. doi: 10.1016/S1387-1811(98)00011-0.











