2
Background
This chapter provides background and information on several aspects of the committee’s work. It begins with an overview of malaria as a disease and of the need for antimalarial drugs for prophylaxis. It then provides an overview of how the antimalarials under consideration interrupt the life cycle of the Plasmodium parasites and discusses the differences among causal prophylaxis, suppressive prophylaxis, presumptive anti-relapse therapy, and the treatment of malaria. An overview of differences among the classes of antimalarial drugs and their mechanisms of action is also provided. The next part of the chapter focuses on the use of prophylactic antimalarial drugs within military populations, including adherence and concurrent exposures that could occur during military service.
MALARIA IN HUMANS
The World Health Organization (WHO) has estimated that in 2018 there were 228 million cases of malaria (range, 206 million to 258 million) occurring in 87 countries and that 405,000 of these cases resulted in death. Nearly 50% of the world’s population live in malaria-risk areas. WHO’s Africa region carries the highest global burden of malaria, with 93% of the world’s cases and almost 50% of its deaths occurring there. More than 60% of the malaria deaths in Africa are estimated to occur in children under 5 years of age. In 2018, just six countries in Africa accounted for more than half of all malaria cases worldwide: Nigeria (25%), the Democratic Republic of the Congo (12%), Uganda (5%), Côte d’Ivoire (4%), Mozambique (4%), and Niger (4%). By contrast, WHO’s South-East Asia Region and Eastern Mediterranean Region accounted for 3.4% and 2.1% of cases, respectively (WHO, 2019). In 2016, 2,078 confirmed cases of (and 7 deaths from)
malaria, nearly all imported, occurred in the United States, the majority of which (n = 1,729) originated in Africa (Mace et al., 2019). Increasing opportunities for international travel creates the risk of contracting malaria in populations that would otherwise not be exposed (Lalloo and Magill, 2019).
The vast majority of people residing in endemic areas experience malaria multiple times over their lifetimes, and very often the disease involves two or more species and stages of parasite. Consequently, as they age, these people often develop a partial immunity to each malaria species that they were infected with and subsequently experience less severe illness when infected with any of the species to which they have previously been exposed (Baird, 2012). WHO has made malaria case reduction a priority with the goal of reducing morbidity and mortality associated with malaria by 90% by the year 2030. Such an endeavor will require a multipronged approach to greatly reduce the transmission of malaria in endemic populations, primarily through the use of drugs for prophylaxis or treatment, vector control, and early diagnosis, and it will require improvements in access to and availability of antimalarial drugs for both prophylaxis and treatment, political leadership, increased resources, new tools (such as an efficacious vaccine), and education about antimalarial drugs and the need for increased drug adherence. Resistance to efficacious antimalarial drugs is a major concern, and this has been observed for several of the available drugs. Improvements in the availability of high-quality, correctly dosed drugs is particularly important, as antimalarial drugs of substandard quality or even falsified contents have been reported in endemic areas (Kaur et al., 2015; Newton et al., 2017). Low-quality drugs and falsified drugs contribute to drug resistance and higher levels of morbidity and mortality.
Disease
Infection with the Plasmodium parasite occurs after an infected female Anopheles mosquito takes a blood meal from a human host. Once the parasite infects a human, it migrates to the liver and enters into an incubation period during which the parasite establishes itself within the body and continues its life cycle. Depending on the Plasmodium species, the incubation period lasts from 7 to 30 days, and no symptoms of malaria are present during this time. For P. falciparum, typically about 10 to 15 days after the infective bite occurs, the first signs of disease will manifest. Most patients with uncomplicated malaria present with some combination of common symptoms including fever, chills, sweats, headaches, nausea, vomiting, body aches, or general malaise. Additional symptoms may include abdominal cramping, cough, muscle pains, and varying levels of mental disorientation. These symptoms are typically the result of the human immune response to massive hemolysis and malaria parasites being released into the bloodstream (Moss and Morrow, 2014).
If uncomplicated malaria is not treated in a timely manner, severe malaria can develop. Features of severe malaria generally appear 3 to 7 days after the onset
of the symptoms associated with uncomplicated malaria. According to WHO, the case definition of severe malaria includes one or more of the following symptoms or clinical findings that occurs in the absence of an identified alternative cause and in the presence of P. falciparum asexual parasitemia: impaired consciousness, acidosis, hypoglycemia, severe malarial anemia, renal impairment or acute kidney injury, jaundice, pulmonary edema, significant bleeding, shock, or hyperparasitemia (WHO, 2014).
The current case definition of severe malaria by WHO no longer includes neurologic symptoms or abnormalities outside of those associated with a coma. Prior to the 2014 malaria case definition revision, cerebral malaria was defined as severe malaria in which patients who were not comatose also exhibited neurologic symptoms (e.g., headache, neck stiffness, drowsiness, agitation, delirium, febrile convulsions, focal neurologic signs, or behavioral disturbances). Neurologic symptoms were eliminated from the case definition because high fever alone, which is a common symptom of malaria, is known to produce mild impairment of consciousness (sometimes referred to as delirium, obtundation, obnubilation, confusion, and psychosis) (WHO, 2014). The removal of other neurologic symptoms or abnormalities from the case definition also allowed for comparability of clinical findings associated with severe malaria. Because it is difficult to differentiate between the symptoms caused by high fever and those caused by severe malaria, removing fever from the case definition allows for more precise diagnostic criteria in which fever is removed as a potential confounder.
Although WHO’s case definition for severe malaria no longer includes neurologic symptoms not directly associated with coma, several studies have found that many survivors of severe malaria can develop long-term physiologic damage resulting in neurologic and cognitive deficits (Idro et al., 2006, 2010, 2016; John et al., 2008). Studies examining the relationship between uncomplicated malaria and long-term neurologic and psychiatric effects have been inconclusive (Dugbartey et al., 1998; Fernando et al., 2003).
In addition to neurologic and cognitive deficits that may be caused by clinical malaria, the Centers for Disease Control and Prevention (CDC) has reported that it has potential for other long-term health consequences as well. Serious long-term health effects can include severe anemia, rupture of the spleen, nephrotic syndrome, hyperreactive malarial splenomegaly, severe disease in a pregnant mother, premature birth or low-birth-weight infants, and recurrence of malaria infection. These symptoms can lead to severe disability and may even result in death if malaria infections remain untreated. Prompt and adequate treatment can prevent the development of these more serious health consequences (CDC, 2019a).
Infectious Agents
There are five species of the Plasmodium parasite that are known to cause disease in humans: Plasmodium falciparum, Plasmodium vivax, Plasmodium ovale,
Plasmodium malariae, and Plasmodium knowlesi. Figure 2-1 summarizes the life cycle of all Plasmodium parasites, although the specific details of the progression through the cycle vary among the five species. A result of this variation is that the timelines for symptom presentation and the targets of drug actions differ among the species (CDC, 2019a).
After the Plasmodium parasite enters the human body, it migrates to the liver and begins invading hepatocyctes; this is the exo-erythrocytic phase of the Plasmodium life cycle. Once in the hepatocytes, the incubation period varies, resulting in different timelines being observed between infection and the presentation of symptoms for different infective species. The P. falciparium, P. malariae, and P. knowlesi species enter the incubation period, and over the next several days replicate thousands of times inside the hepatocytes. The increasing parasitic load inside liver cells eventually causes the hepatocyte to swell and rupture, releasing thousands of parasites into the bloodstream. Once in the bloodstream, the parasite enters into the erythrocytic phase of its life cycle and continues replicating within the body (Moss and Morrow, 2014).
By contrast, P. vivax and P. ovale can, after invading the hepatocytes, either continue to progress through the life cycle (like P. falciparum, P. malariae, or P. knowlesi) or become hypnozoites and lie dormant within the hepatocyte for up to several years before reactivating and resuming their development, subsequently causing clinical symptoms of malaria. During this dormant period P. vivax and P. ovale hypnozoites remain undetectable by the human immune system or any current diagnostic techniques. These hypnozoites can differentiate at any time into the next stage of the life cycle, at which point they are released into the bloodstream (Moss and Morrow, 2014). Importantly, not all dormant parasites differentiate at the same time. This means some of the dormant parasites may differentiate and continue through the parasite life cycle, while others may remain dormant and undetectable. This complicates the prophylaxis and treatment needed for these two Plasmodium species; therefore, prophylactics that target the exo-erythrocytic stage of the Plasmodium life cycle are critical for preventing infection with P. vivax and P. ovale.
PROPHYLACTIC ANTIMALARIAL DRUGS
As described in Chapter 1, the antimalarial drugs covered by this report are those that are currently available and approved by the Food and Drug Administration (FDA) as of 2019 for malaria prophylaxis in adults and that are currently being used, or that have been used in the past 25 years by U.S. military personnel for malaria prophylaxis. Specifically, they are mefloquine, tafenoquine, atovaquone/proguanil (A/P), doxycycline, primaquine, and chloroquine.
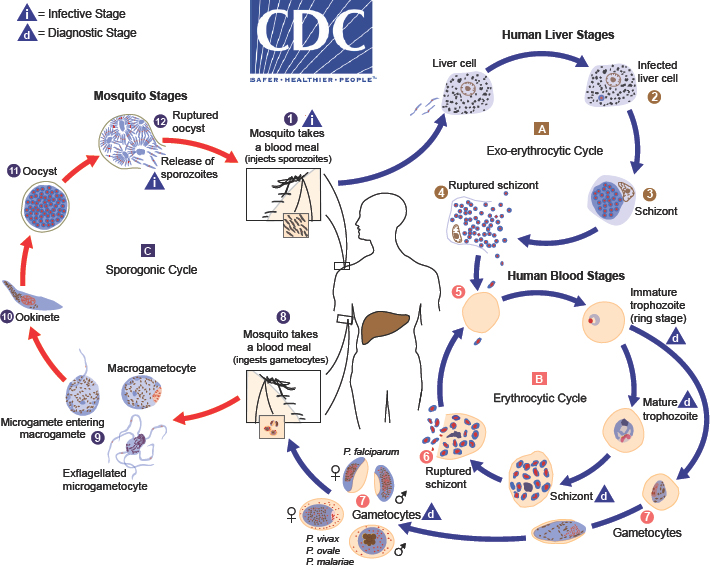
 . Sporozoites infect liver cells
. Sporozoites infect liver cells  and mature into schizonts
and mature into schizonts 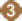 , which rupture and release merozoites
, which rupture and release merozoites  . (Of note, in P. vivax and P. ovale a dormant stage [hypnozoites] can persist in the liver [if untreated] and cause relapses by invading the bloodstream weeks, or even years later.) After this initial replication in the liver (exo-erythrocytic schizogony
. (Of note, in P. vivax and P. ovale a dormant stage [hypnozoites] can persist in the liver [if untreated] and cause relapses by invading the bloodstream weeks, or even years later.) After this initial replication in the liver (exo-erythrocytic schizogony 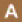 ), the parasites undergo asexual multiplication in the erythrocytes (erythrocytic schizogony
), the parasites undergo asexual multiplication in the erythrocytes (erythrocytic schizogony  ). Merozoites infect red blood cells
). Merozoites infect red blood cells  . The ring stage trophozoites mature into schizonts, which rupture releasing merozoites
. The ring stage trophozoites mature into schizonts, which rupture releasing merozoites  . Some parasites differentiate into sexual erythrocytic stages (gametocytes)
. Some parasites differentiate into sexual erythrocytic stages (gametocytes)  . Blood stage parasites are responsible for the clinical manifestations of the disease. The gametocytes, male (microgametocytes) and female (macro-gametocytes), are ingested by an Anopheles mosquito during a blood meal
. Blood stage parasites are responsible for the clinical manifestations of the disease. The gametocytes, male (microgametocytes) and female (macro-gametocytes), are ingested by an Anopheles mosquito during a blood meal  . The parasites’ multiplication in the mosquito is known as the sporogonic cycle
. The parasites’ multiplication in the mosquito is known as the sporogonic cycle  . While in the mosquito’s stomach, the microgametes penetrate the macrogametes generating zygotes
. While in the mosquito’s stomach, the microgametes penetrate the macrogametes generating zygotes  . The zygotes in turn become motile and elongated (ookinetes)
. The zygotes in turn become motile and elongated (ookinetes)  , which invade the midgut wall of the mosquito where they develop into oocysts
, which invade the midgut wall of the mosquito where they develop into oocysts  . The oocysts grow, rupture, and release sporozoites
. The oocysts grow, rupture, and release sporozoites  , which make their way to the mosquito’s salivary glands. Inoculation of the sporozoites
, which make their way to the mosquito’s salivary glands. Inoculation of the sporozoites  into a new human host perpetuates the malaria life cycle” (CDC, 2017).
into a new human host perpetuates the malaria life cycle” (CDC, 2017).SOURCE: CDC, 2017.
Differences Between Causal Prophylaxis, Suppressive Prophylaxis, Presumptive Anti-Relapse Therapy, and Treatment of Malaria
There are two types of prophylaxis used to prevent the development of clinical malaria: causal and suppressive. Briefly, causal prophylaxis is begun in persons free of infection, and it prevents the formation of both tissue schizonts in the liver and hypnozoites of the malaria parasite. Suppressive prophylaxis refers to drugs that act only on parasites within the red blood cells (Schwartz, 2012). Neither type of prophylaxis prevents human infection with Plasmodium parasites; instead, the drugs inhibit the Plasmodium parasite’s ability to further establish infection, replicate, and cause clinical disease; the drugs are also used to prevent recurrence of malaria. Two other categories of antimalarial drugs are approved for use by FDA: those for presumptive anti-relapse therapy (PART) and drugs for treatment of malaria. Precise definitions that clearly explain the differences between the different types of prophylaxis were not available from CDC, WHO, or FDA. As a result, the following definitions were compiled from other sources.
Richter et al. (2016) defines recurrence of malaria as a clinical malaria attack after it has been treated. Recurrence is further differentiated into recrudescence and relapse. Recrudescence is defined as “malaria recurrence originating from subclinical low-level circulating asexual erythrocytic stages, and the Plasmodium species associated with recrudescence are genetically identical to the ones of the first attack” (Richter et al., 2016, p. 2140). Relapse is defined as “malaria recurrence originating from the latent Plasmodium tissue stages (hypnozoites) associated with P. vivax and P. ovale species, and the Plasmodium species associated with relapse are heterologous and differ from those causing the first malaria episode” (Richter et al., 2016, p. 2140); however, relapses may be genetically identical, if the initial infection was monogenomic. Recrudescence is often linked to the failure or low efficacy of suppressive antimalarial prophylaxis, whereas relapse can be associated with the inappropriate use of suppressive prophylaxis for the prevention of hypnozoites from P. vivax or P. ovale infection or with the failure of causal prophylaxis.
Causal Prophylaxis
Of the six drugs included in this report, three exhibit causal prophylactic activity: A/P, primaquine, and tafenoquine. A/P exhibits causal prophylactic activity only against P. falciparum, and it is not effective against hypnozoites associated with P. ovale or P. vivax. Causal prophylaxis, also called exo-erythrocytic stage prophylaxis, kills the Plasmodium parasite before it can complete its development in the liver, thereby inhibiting the parasite’s ability to replicate or cause clinical disease (60 Degrees Pharmaceuticals, 2018). The use of causal prophylaxis to prevent infection with P. vivax and P. ovale is critical; these species can remain dormant in the liver for long periods (up to several years) after an infection occurs and can result in relapse of disease. Causal prophylaxis should be taken for 7–14 days after returning from
an area with endemic malaria because the parasites are killed early in their life cycle and they never enter the bloodstream. Figure 2-1 illustrates the way in which causal prophylaxis interferes with the exo-erythrocytic phase of the Plasmodium life cycle (A; steps 1–4). Because causal prophylaxis acts on the exo-erythrocytic stage of the life cycle, it can prevent human infection by all Plasmodium parasites. It is believed that causal prophylactic drugs work by interfering with some key cellular processes necessary for replication and cell survival, including deoxyribonucleic acid (DNA) replication and mitochondrial function (Schwartz, 2012).
Suppressive Prophylaxis
Whereas some of the drugs used for causal prophylaxis also demonstrate suppressive prophylactic activity (e.g., A/P, primaquine, and tafenoquine), mefloquine, chloroquine, and doxycycline are defined exclusively as suppressive antimalarial prophylactic drugs. Suppressive prophylaxis has no effect on Plasmodium parasites until the liver phase of the life cycle is complete and the parasite has invaded red blood cells. Suppressive prophylaxis is ineffective against hypnozoites of P. vivax or P. ovale, but it will kill P. vivax or P. ovale parasites that have entered the bloodstream (Schwartz, 2012). Figure 2-1 shows how suppressive prophylaxis interferes with the erythrocytic phase of the Plasmodium life cycle (B; steps 5–7). Taking suppressive prophylaxis as directed will suppress the symptoms of malaria in individuals who are infected with the malaria parasite. However, if suppressive prophylaxis is not taken as directed, symptoms of the disease will likely appear. Because these drugs act only on parasites in red blood cells, an individual must take them for 4 weeks after leaving an area with endemic malaria in order to eliminate parasites that may appear in the bloodstream during that time (Moss and Morrow, 2014).
The mechanisms of action for suppressive prophylactic drugs vary but are known to include blocking the production of hemozoin, which is a chemical that the parasite produces to protect itself from the toxic products produced as a result of its digestion of hemoglobin by turning them into a non-toxic compound; inhibiting vesicle functions that may interfere with phospholipid metabolism; binding to and altering the parasite’s DNA; blocking transcription and translation of DNA into RNA and proteins; and impairing the expression of the apicoplast (a collection of structures present in Plasmodium parasites that allows for the invasion of host cells and the establishment of the parasite–host interaction) genes in the parasite, resulting in the inability of the parasite to replicate its DNA (Parhizgar and Tahghighi, 2017).
Presumptive Anti-Relapse Therapy
PART, also known as terminal prophylaxis, is the use of an antimalarial drug toward the end of the exposure period (or immediately thereafter) to prevent relapses
or the delayed onset of clinical malaria caused by hypnozoites (dormant exo-erythrocytic stages) of P. vivax or P. ovale. PART is generally indicated for people who have had prolonged exposure in malaria-endemic areas (such as military personnel, Peace Corps volunteers, or missionaries) (CDC, 2019a). Two of the drugs included in this report, primaquine and tafenoquine, can be used both as primary prophylaxis and as PART. When used for PART, these drugs are often given in combination with chloroquine or another blood schizonticide. PART is an important factor in preventing relapse of malaria. Because PART agents act on both the exo-erythrocytic and erythrocytic stages, they affect the malaria life cycle at the same points as causal and suppressive prophylactic agents (see A and B in Figure 2-1).
Treatment
An inherent distinguishing factor between the prophylaxis and the treatment of malaria is how the antimalarial drugs are used: prophylaxis prevents disease, while the goal of treatment is to cure infection. Each of the drugs included in this report can be used for either prophylaxis or treatment; however, the dosage at which each drug is used for the treatment of malaria is significantly higher than when it is used for prophylaxis. For example, the treatment dose of A/P is four times higher than its prophylactic dose. As a result of the higher blood concentrations achieved with treatment regimens, adverse events may occur with treatment that are not observed when the drug is used for prophylaxis, or adverse events may be more severe in their presentation (Arguin and Magill, 2017; CDC, 2019b). Whereas the FDA package insert for A/P lists diarrhea, dreams, oral ulcers, and headache as common adverse events (≥4% of adults) when A/P is used as directed for prophylaxis, the common (≥5% of adults) adverse events when A/P is used for treatment include abdominal pain, nausea, vomiting, headache, diarrhea, asthenia, anorexia, and dizziness (FDA, 2019).
Distinguishing Between the Different Classes of Antimalarial Drugs
The antimalarial drugs under consideration in this report belong to several drug classes (based on chemical structure): 8-aminoquinolines (primaquine and tafenoquine), 4-aminoquinolines (chloroquine), tetracyclines (doxycycline), and quinoline methanols (mefloquine). A/P is a combination drug: atovaquone belongs to the class of naphthoquinones and proguanil is a synthetic arylbiguanide (antifolate). The adverse events of a given drug are often difficult to predict a priori, but sometimes the compounds of a certain drug class result in common adverse events. However, even though numerous non-antimalarial drugs contain quinoline (or quinolone) substructures, these structurally diverse drugs have adverse event profiles that are distinct from those of the antimalarial quinolines discussed in this report. The following text explains the different mechanisms of action that each drug class exhibits on the Plasmodium parasites; however, it is important
to note that each drug class also exhibits unique pharmacokinetic and pharmacodynamic properties once inside the human body. The differences are not limited to the characteristics observed at a drug class level, but are also observed between individual drugs within the same drug class. Detailed information on the different pharmacokinetic and pharmacodynamic properties of each drug are presented in each drug-specific chapter.
8-Aminoquinolines
The 8-aminoquinoline class of antimalarials is unique in that it owes its ability to prevent the relapsing forms of malaria by acting against the malaria hypnozoites that lie dormant in the liver (Marcsisin et al., 2014). Primaquine, the prototype 8-aminoquinoline, was developed in 1945 and has prophylactic activity against the liver stage of all malaria parasites, against the asexual and sexual stages of P. vivax, P. ovale, P. malariae, and P. knowlesi, and the sexual stages of P. falciparum (it is only weakly active against the asexual blood stages of P. falciparum), and it has radical curative activity in P. vivax and P. ovale malaria. It is the most widely used 8-aminoquinoline for malaria prophylaxis, but its exact mechanism of action is still unknown. Primaquine localizes within the Plasmodium mitochondria and impairs the mitochondrial metabolism, which suggests drug-induced mitochondrial dysfunction as a potential mechanism of action (Schlagenhauf et al., 2019).
In general, 8-aminoquinolines are metabolized by the cytochrome P450 CYP2D family (particularly CYP2D6), which is required for both their antimalarial activity and their toxicity. Several possible modes of action on the parasite have been proposed. One hypothesis is that active metabolites of 8-aminoquinolines may lead to mitochondrial dysfunction (Schlagenhauf et al., 2019) and the alteration of intracellular membrane structures in both erythrocytic and pre-erythrocytic stages of the parasite. Another hypothesis is that the highly reactive metabolites generate intracellular reactive species, which cause oxidative damage. The primaquine metabolite, 5-hydroxyprimaquine, and its downstream oxidation products such as the corresponding 5,6-orthoquinone (Fasinu et al., 2019) cause a substantial generation of reactive oxygen species, most notably hydrogen peroxide (Camarda et al., 2019). This leads to the killing of the malaria parasite (Camarda et al., 2019), methemoglobinemia (Liu et al., 2011), and oxidative damage to the erythrocyte cytoskeleton (Bowman et al., 2005).
Tafenoquine was first identified in 1978, but it was only recently approved (2018) for the prophylaxis and radical cure of malaria. Clinical trials of tafenoquine have not definitively determined whether it works via a causal or a suppressive prophylaxis mechanism (Baird, 2018). Tafenoquine is substantially more active against the erythrocytic stages of the Plasmodium life cycle than primaquine, and it is more slowly metabolized with a terminal elimination half-life of 14–17 days. Tafenoquine is active against all pre-erythrocytic and erythrocytic forms of
human malaria as well as against the gametocytes of P. falciparum and P. vivax (FDA, 2018a).
Neither the precise mechanism of action of tafenoquine nor its molecular target have been determined. In vitro studies with the erythrocytic forms of P. falciparum suggest that tafenoquine may exert its effect by inhibiting hemozoin formation and inducing apoptotic-like death of the parasite (FDA, 2018b; Vennerstrom et al., 1999). This may explain why tafenoquine is active against the asexual blood stage of parasites, unlike primaquine, which does not inhibit hemozoin formation (Ebstie et al., 2016). Otherwise, the mechanism of action of tafenoquine is similar to that of primaquine (Ebstie et al., 2016) in which the spontaneous oxidation of metabolites generates hydrogen peroxide and hydroxyl radicals. The reactive oxygen species generated through P. falciparum ferredoxin-nicotinamide adenine dinucleotide phosphate (NADP+) reductase and diflavin reductase enzymes are thought to result in parasite death, a theory that is supported by the upregulation of these enzymes in tafenoquine-sensitive stages of the parasite (Ebstie et al., 2016).
One limitation of antimalarial 8-aminoquinolines is that they are contraindicated in people who have the X-linked glucose-6-phosphate dehydrogenase (G6PD) genetic defect. G6PD deficiency is the most common genetic human enzyme disorder, with 186 genetic variants that have been described. It is estimated to affect more than 400 million people worldwide, most of them in malaria-endemic areas and most commonly in males (Nkhoma et al., 2009). G6PD is the key enzyme in the oxidative pentose phosphate pathway. It converts NADP+ into its reduced form, NADPH. NADPH is essential for protection against oxidative stress in erythrocytes. G6PD deficiency causes an increased susceptibility of erythrocytes to hydrogen peroxide and other reactive oxygen species which can lead to hemolysis (the rupture of red blood cells and release of their contents into the plasma) and hemolytic anemia (red blood cells being destroyed faster than they can be replaced), which in turn can lead to other serious complications, including arrhythmias, cardiomyopathy, heart failure, and death (NIH, n.d.; Peters and Van Noorden, 2009). The extent of hemolysis depends on the dose and duration of drug exposure and the degree of G6PD deficiency. Persons with >80% of normal red-blood-cell G6PD activity are considered to be G6PD normal (WHO, 2016a). Males with <10% normal red-blood-cell G6PD activity are regarded as G6PD deficient; females with 30–80% of normal red-blood-cell G6PD activity are considered to be G6PD intermediate, and those with <30% are regarded as G6PD deficient. A study of more than 63,000 U.S. military personnel determined that 2.5% of men and 1.6% of women were G6PD deficient, with most of them having a moderate level of deficiency (Chinevere et al., 2006). The highest rates of deficiency were seen in African American men (12.2%) and women (4.1%) and Asian men (4.3%).
WHO recommends the use of ultraviolet spectrophotometry as the gold standard for measuring G6PD activity; however, this technique requires technology that is not suitable for field development or point-of-care testing. As a result,
the most commonly used field test for G6PD deficiency is the fluorescent spot test (FST), a semi-quantitative assay that requires minimal laboratory supplies and does not require expertise for result interpretation. Several qualitative tests have been recently introduced that have better operational characteristics and similar detection capabilities as the FST. These tests can only distinguish G6PD homozygous females and hemizygous males with intermediate enzyme activities above 30% of normal from G6PD-normal individuals. Because G6PD deficiency is linked to the X chromosome, females can present with homozygous, heterozygous, or normal G6PD gene expression. The commonly used field tests are insufficient for detecting G6PD activity in heterozygous females with intermediate enzyme activities that fall outside of the 30% of normal threshold. As a result, heterozygous females may express G6PD-deficient characteristics that are not detected by the currently available field testing procedures (Ley et al., 2017); the presentation of these false-negative results may lead to inadvertent exposure to 8-aminoquinoline antimalarial drugs and their subsequent adverse events (Peters and Van Noorden, 2009). Recently, researchers have also begun exploring quantitative testing that can be administered in resource-limited settings; however, many of these tests are still in development or are undergoing evaluation (Pal et al., 2019).
Methemoglobinemia, usually mild and reversible, is a well-characterized feature in recipients of 8-aminoquinolines at therapeutic dosing (Baird, 2019). Methemoglobinemia occurs when the level of methemoglobin in red blood cells exceeds 1%, which can lead to decreased availability of oxygen to tissues (Denshaw-Burke et al., 2018). Severe methemoglobinemia can lead to complications, including abnormal cardiac rhythms, altered mental status, delirium, seizures, coma, and profound acidosis; if the level of methemoglobin in red blood cells exceeds 70%, death can result.
4-Aminoquinolines
As reviewed by Foley and Tilley (1998) and O’Neill et al. (2006), chloroquine, which was first discovered in 1934, is the prototype 4-aminoquinoline antimalarial drug. Both enantiomers of the racemic chloroquine have equivalent antimalarial activity. Chloroquine is active against the erythrocytic stages of all species of malaria, and it is also active against the gametocytes of P. vivax, P. malariae, and P. ovale. It is a diprotic weak base, and it works by concentrating in the parasite food vacuole and binding tightly to hematin as it is formed during the digestion of hemoglobin by the parasite. The binding of chloroquine to hematin interferes with the assembly of hematin into the non-toxic hemozoin, or malaria pigment, and this may increase the intrinsic toxicity of hematin to the parasite. As reviewed by Ecker et al. (2012), drug resistance to chloroquine in P. falciparum is mediated primarily by mutant forms of the chloroquine resistance transporter (PfCRT). These mutant forms of PfCRT effectively efflux chloroquine from the parasite digestive vacuole, the site of the drug’s action. Mutations in PfCRT allow the parasite to persist at
drug levels that kill chloroquine-sensitive parasites. Some researchers have suggested that chloroquine-sensitive P. falciparum is returning in parts of Africa due to the discontinuation of the widespread use of chloroquine in the early 1990s, which may have resulted in the parasite’s reversal to a chloroquine-sensitive state (Schlagenhauf et al., 2019). Individuals who have G6PD deficiency should be closely monitored while receiving chloroquine because of the potential for hemolysis to occur; however, significant hemolysis is rare when the drug is given at prophylactic and therapeutic doses (Schlagenhauf et al., 2019).
Quinoline Methanols
Mefloquine is a synthetic structural analog of quinine (Hellgren et al., 1997). Mefloquine, as well as the other antimalarial quinolines such as chloroquine, primaquine, and tafenoquine, differs considerably from quinine with respect to both its mechanistic biology and the adverse events associated with its use. Furthermore, as numerous non-antimalarial drugs also contain quinoline substructures, overgeneralizations about adverse events of quinolines as a group are unwarranted (Dorwald, 2012). Mefloquine, a quinoline methanol, was first identified as a compound with antimalarial activity in animal models in the 1960s and was approved for prophylaxis and the treatment of malaria in humans in 1984. Mefloquine consists of a 50:50 racemic mixture of the erythro isomers available as tablets containing 250 mg of mefloquine salt. The formulation of mefloquine available in the United States contains 250 mg of mefloquine hydrochloride.
Mefloquine is a potent, long-acting blood schizonticide that is effective against all malarial species that infect humans (Schlagenhauf et al., 2010); however, it has no activity against the liver stages of parasite development (Palmer et al., 1993). Both the (+) and the (–) enantiomers are active against P. falciparum, but a higher activity for (+) mefloquine has been reported (Hellgren et al., 1997). One major reason for its importance in the malaria-prophylaxis armamentarium is its efficacy against chloroquine-sensitive and chloroquine-resistant P. falciparum, (Palmer et al., 1993), although resistance of P. falciparum to mefloquine is known to exist in parts of Cambodia, Laos, Myanmar, Thailand, and Vietnam (CDC, 2019a). The exact mechanism of action is unclear, but it is thought that inhibiting hemozoin formation in the P. falciparum food vacuole causes a toxic accumulation of the highly reactive hematin moiety, which in turn kills the parasite; oxidative damage is believed to play a role (Ridley et al., 1997; Sullivan et al., 1998). One study has found that in addition to inhibiting hemozoin, mefloquine can induce apoptosis in Plasmodium species by activating metacaspase and reactive oxygen species production (Gunjan et al., 2016). More recently, mefloquine was found to act by targeting the P. falciparum 80S ribosome to inhibit protein synthesis (Wong et al., 2017).
Antibiotics
Tetracyclines are a class of broad-spectrum antibiotic drugs that are used to treat a wide range of illnesses. Tetracyclines were first investigated for their antimalarial potential in the 1960s after the emergence of chloroquine-resistant P. falciparum parasites. The tetracycline drug doxycycline is a slow-acting schizonticidal agent. In addition to its activity against the erythrocytic stage of the parasite, doxycycline is thought to possess some pre-erythrocytic (causal) activity, but efficacy studies found unacceptably high failure rates for its use as a causal prophylactic. There is very limited evidence concerning doxycycline’s effect on gametocytes of Plasmodium species, and it has been shown to have no effect on the hypnozoites. In one study that examined gametocytemia and doxycycline, P. vivax gametocytemia increased from 32% pre-treatment to 44% immediately post-treatment, and the median gametocyte clearance time was 62 hours. Gametocytemia has no clinical implications, but malaria may still be transmitted through mosquitoes if they bite an individual being treated for malaria with doxycycline (Tan et al., 2011).
Although doxycycline is known to be a blood schizontocide, the exact mechanism of its action is not well defined (Schlagenhauf et al., 2019). In P. falciparum, doxycycline impairs the expression of apicoplast genes, leading to nonfunctional apicoplasts in subsequent progeny, and it impedes the development of viable parasites. Doxycycline’s antimalarial actions may be similar to its bacteriostatic actions of binding to ribosomal subunits and inhibiting protein synthesis, but this has only been observed in suprapharmacologic doses (Tan et al., 2011).
Combinations (Antifolates)
Atovaquone/proguanil (A/P) is a fixed drug combination made from atovaquone and proguanil for the prophylaxis of P. falciparum malaria. Atovaquone is a hydroxynaphthoquinone, and proguanil is a synthetic arylbiguanide (antifolate); the two drugs work synergistically against the erythrocytic stages of all the Plasmodium parasites and against the liver stage (causal prophylaxis) of P. falciparum. A/P is not active against hypnozoites in P. vivax or P. ovale, and it does not prevent relapse infections (Nixon et al., 2013).
Atovaquone acts by inhibiting the Plasmodium species’ mitochondrial electron transport at the cytochrome bc1 complex, which collapses mitochondrial membrane potential. The electron transport system of the Plasmodium species is 1,000 times more sensitive to atovaquone than this system in mammals, which is thought to explain the selective action and limited adverse events of the drug (Schlagenhauf et al., 2019). Inhibition of the Plasmodium bc1 complex by atovaquone affects the concentrations of metabolites in the pyrimidine bio-synthetic pathway and in the biosynthesis of purine, both of which are required for DNA replication in the Plasmodium parasite (Boggild et al., 2007). Proguanil is
metabolized to cycloguanil, which inhibits dihydrofolate reductase, resulting in an impeding of the synthesis of folate cofactors required for parasite DNA synthesis.
When atovaquone and proguanil are given in combination, both in vitro and in vivo studies have found the mechanism of action to be synergistic between the two of them (Canfield et al., 1995; Looareesuwan et al., 1999). This leads to high cure rates of P. falciparum malaria, even in cases where the parasites have developed a resistance to cycloguanil conferred by DHFR mutations (Gay et al., 1997). The cause of the synergy between proguanil and atovaquone is thought to be found in the biguanide mode of action, not in the action of its metabolite(s) (Srivastava et al., 1999). Proguanil acts synergistically with atovaquone in individuals with proguanil-resistant parasites or in those who are unable to metabolize proguanil to cycloguanil because of CYP450 enzyme deficiencies (Helsby et al., 1990; Looareesuwan et al., 1999).
MILITARY USE OF ANTIMALARIALS
Malaria has affected almost all U.S. military deployments, actions, and overseas exercises since the American Civil War (see Table 2-1), and despite advances in antimalarial drugs and improvements in preventive equipment and supplies, it remains an ongoing threat (IOM, 2006). The number of malaria cases in U.S. service members varies each year and recently has ranged from a high of 124 cases in 2011 to a low of 30 cases in 2013 and 2015 (AFHSC, 2013, 2014, 2015, 2016; WHO, 2016b). A 65% increase in reported cases of malaria in military service members was reported between 2017 (35 cases) and 2018 (58 cases), and more than 25% of the cases in 2018 were due to P. falciparum, the most severe species of malaria (AFHSB, 2019). Successful control of malaria in the military demands effective prophylactic interventions, force-wide education about malaria and prevention, and prophylactic adherence by individual service members.
Keeping abreast of malaria medically and technologically has been a continuing effort for the U.S. military. During World War II the Japanese blockade of Javanese and Philippine quinine sources, Germany’s monopoly on manufacturing available quinine and the antimalarial quinicrine (also known as mepacrine and under the trade name Atabrine), and intelligence that Germany was synthesizing new antimalarials all served to compel the United States to attempt to synthesize quinicrine based on a drug sample and to spur the United States and its allies to focus research into new synthetic antimalarials (IOM, 2006; Kitchen et al., 2006).
By 1942, the United States had successfully synthesized quinicrine, and U.S. service members were receiving it. The antimalarial research program, a collaboration among the military, scientific institutions, universities, and pharmaceutical firms, was established in 1941. Two of its early discoveries were chloroquine, which is effective against P. falciparum and which the U.S. military began using in 1945, and primaquine, which is effective against P. vivax and was first used on
TABLE 2-1 Major U.S. Military Actions, Deployments, or Overseas Exercises in Locations with a Malaria Threat, 1861–2003
| Location | Year | Threat | Morbidity and Mortality |
|---|---|---|---|
| Civil War | 1861–1865 (Union) | P. vivax, P. falciparum |
1.3 million cases, 10,000 deathsa |
| Panama Canal | 1904–1914 | P. vivax, P. falciparum P. vivax |
1906 malaria rate 1,263/1,000/year 1913 malaria rate 76/1,000/yearb Estimated 5,000 cases overseas |
| WWI | 1914–1918 | P. falciparum | 1917: 7.5/1,000/year in United Statesc |
| WWII | 1939–1945 | P. vivax | 600,000 cases mostly in Pacific theater. In some areas of South Pacific malaria rates were 4,000/1,000/year (4 cases per person per year) (Downs et al., 1947) |
| Korean War | 1950–1953 | P. vivax | Malaria rate 611/1,000/year 3,000 cases in troops returning to United Statesd |
| Vietnam War | 1962–1975 | P. falciparum, P. vivax |
100,000 casese 1.7/1,000 case fatality rate Hospital admissions 27/1,000/year 1965 malaria rate for U.S. Army forces: 98/1,000/year 1970: 2,222 cases (mostly P. vivax) Treated in United States |
| Panama | 1988–1989 | P. falciparum | Action primarily in Panama City |
| Persian Gulf | 1991 | P. vivax | Few cases in northern Iraq, Kurdish area |
| Somalia | 1992–1994 | P. falciparum, P. vivax |
48 cases; 243 cases in forces on return homef (CDC, 1993) |
| Nigeria | 2001 | Chloroquine-resistant P. falciparum |
Special forces 7 cases (2 deaths in 300 men) |
| Afghanistan | 2003 | P. vivax, chloroquine-resistant P. falciparum |
8 cases in 725 Ranger task force membersg (Kotwal et al., 2005) |
| Liberia | 2003 | P. falciparum | U.S. marines 80/290 (28% attack rate) with 40 Marines evacuated by air to Germany |
| Iraq War | 2003– | P. vivax | Few cases |
a Records for the Confederate forces were difficult to find (probably not kept). One example in South Carolina was 42,000 cases in 18 months in 1862–1863. (Malaria was endemic in the United States until the late 1940s.)
b 1913 malaria rate drop was due to control measures enforced by Colonel Gorgas.
c Malaria rate for troops training and/or garrisoned in southern states.
d In troops returning home there were at one point 629 cases/week.
e Some operational areas were intense: Ia Drang Valley (1966) malaria rate 600/1,000/year, equivalent of 2 maneuver battalions rendered inoperative.
f In Bardera in 1993 where malaria is hyperendemic: 53/490 cases in Marines.
g Attack rate (June–September 2002) 52.4/1,000/year.
SOURCE: IOM, 2006.
U.S. ships returning from Korea in 1951 (Brundage, 2003; IOM, 2006). By the early 1960s, P. falciparum resistance to chloroquine had been reported in South America, Southeast Asia, and Oceania (CDC, 2018; Kitchen et al., 2006). By 1962, the “C-P pill,” a combination tablet of chloroquine and primaquine, had become the standard prophylactic regimen for soldiers in Vietnam. In 1963, however, the increasing toll of chloroquine-resistant P. falciparum in service members in Vietnam led to the launch of the U.S. Army Medical Research Program on Malaria at the Walter Reed Army Institute of Research (Brundage, 2003; IOM, 2006; Ockenhouse et al., 2005). Within 10 years, 27 new drugs or drug combinations had been developed, including dapsone, mefloquine, and halofantrine, which appeared to be the answers to chloroquine-resistant P. falciparum (Brundage, 2003; IOM, 2006). In 1966 dapsone was added to the C-P tablet given to troops at high risk of chloroquine-resistant P. falciparum (Brundage, 2003); hydroxychloroquine may also have also been used in Vietnam and during the Korean War, although records are unclear.1
In the late 1960s mefloquine was developed by F. Hoffmann-LaRoche in collaboration with the Walter Reed Army Institute and WHO (Kitchen et al., 2006). It was approved in 1989, was likely used by the military as early as 1990, and was used during Operation Restore Hope in Somalia (1992–1993) and Operation Iraqi Freedom (OIF; 2003–2011) (Kitchen et al., 2006, Sánchez et al., 1993).2 Mefloquine was used as a first-line prophylactic agent only for deployments to certain high-malaria-risk areas in sub-Saharan Africa, such as for the Liberian Task Force in 2003, and it was used as a second-line agent in OIF and Operation Enduring Freedom (OEF) (Wiesen, 2019). The military began testing doxycycline for malaria prophylaxis in 1985, but it was not used routinely for prophylaxis until 1992 in Somalia (Sánchez et al., 1993; Wallace et al., 1996).3 Doxycycline was used as the first-line agent in OIF (2003–2007) and OEF (2001–present), and it continues to be used in deployments to malaria-endemic regions (DoD, 2013).
A 2009 Department of Defense (DoD) memorandum advised that in chloroquine-resistant areas where doxycycline and mefloquine are equally efficacious, and when personnel have a history of neurobehavioral disorders, doxycycline should be the first-line agent and A/P the second-line agent, and in those who cannot take doxycycline or A/P, mefloquine should be used very cautiously and with clinical follow-up (DoD, 2009). The memo also stated, presumably regard-
___________________
1 Personal communication to the committee, COL Andrew Wiesen, M.D., M.P.H., Director, Preventive Medicine, Health Readiness Policy and Oversight, Office of the Assistant Secretary of Defense (Health Affairs), April 16, 2019.
2 Personal communication to the committee, COL Andrew Wiesen, M.D., M.P.H., Director, Preventive Medicine, Health Readiness Policy and Oversight, Office of the Assistant Secretary of Defense (Health Affairs), April 16, 2019.
3 Personal communication to the committee, COL Andrew Wiesen, M.D., M.P.H., Director, Preventive Medicine, Health Readiness Policy and Oversight, Office of the Assistant Secretary of Defense (Health Affairs), April 16, 2019.
ing personnel with no history of neurobehavioral disorders, that mefloquine should only be used by those with contraindications to doxycycline and without contraindications to mefloquine. A 2013 DoD memo stated that doxycycline and A/P were to be considered first-line agents in chloroquine-resistant areas, with mefloquine reserved for use by those intolerant to or with contraindications to both doxycycline and A/P (DoD, 2013). A/P was approved in 2000, but its use in military service members was not significant until 2013, when it joined doxycycline as a first-line choice for chloroquine-resistant areas (DoD, 2013). The military medicine concept of force health protection is defined as “all measures taken by commanders, supervisors, individual service members, and the military health system to promote, protect, improve, conserve, and restore the mental and physical well-being of service members” (Raczniak et al., 2019). Force health protection policy positions in DoD are issued as directives and instructions and include the use of antimalarial drugs for prophylaxis. Although policy may be made at higher levels, the final decision to use malaria prophylaxis under force health protection is made by commanders in the field, guided by their medical staff (Raczniak et al., 2019). Final decisions regarding malaria prophylaxis policy can be directed by authorities at other levels as well.
When malaria prophylaxis is indicated, service members are required to receive it under proper medical supervision. If a drug is medically contraindicated, alternative agents may be used if they are available, and the unit medical officer is to document those who have not received standard preventive measures so that they may receive additional monitoring or treatment if they become ill. However, while ordinary travelers are encouraged to adhere to malaria prophylaxis, military personnel are required to do so. Moreover, military personnel often use malaria prophylaxis for longer periods than travelers (many deployments are for 1 year or more), and they do so under demanding, stressful, and dangerous circumstances (Fukuda et al., 2018).
Unlike the case with individual travelers, large military operations have operational constraints related to their forward planning. It is not easy to make rapid changes in policy concerning the widespread use of new medications, particularly when large numbers of personnel are deployed at short notice from a number of locations around the world. Moreover, many military missions to endemic areas are in places of conflict, where malaria control measures have been interrupted or impaired (Pergallo, 2001).
Current DoD policy requires that troops sent to endemic areas use personal protective measures, such as sleeping under mosquito nets, wearing insecticide-impregnated uniforms, using insect repellent (i.e., DEET [N,N-diethyl-3-metatoluamide]), and taking malaria prophylaxis drugs as prescribed. Although these requirements are particularly important for troops stationed in endemic areas for long periods of time, some individuals with certain military occupations, such as pilots and aircrews who transport goods and people and make short trips (generally less than 24 hours) to malaria-endemic areas, are subject to different
requirements. For example, mefloquine is not approved for prophylactic use in pilots (DeJulio, 2016).
Adherence to Malaria Prophylaxis
One of the most important factors in choosing among the several drugs available for malaria prophylaxis is understanding the dosing regimen required to effectively prevent the development of clinical malaria. Efficacy rates are lower for individuals who incorrectly use antimalarial drugs than for those who use them correctly (Cunningham et al., 2014; Goodyer et al., 2011; Saunders et al., 2015). Although the terms adherence and compliance are often used interchangeably, the two terms are not synonymous. Medication adherence, as defined by WHO, is “the degree to which the person’s behavior corresponds with the agreed recommendations from a health care provider.” To assess adherence, investigators typically consider whether individuals actively fill or pick up newly prescribed medications from a pharmacy, or instead refill existing prescriptions on time (National Stroke Association, 2012). Compliance refers to the extent to which a person’s behavior matches the prescriber’s advice (Horne, 2006). Therefore, compliance refers to how much and how often an individual ingests a medication compared with the dosing regimen dictated on the medication’s prescription label, packaging, or FDA package insert. It is important to distinguish between drug adherence and drug compliance because they may affect not only the efficacy of a drug but also the adverse events that may be associated with the drug’s use. Despite this, many publications mix the terms. To avoid confusion, the committee preferentially uses the term “adherence” throughout the report when referring to behaviors regarding use of the antimalarial drugs of interest.
Adherence to prophylactic antimalarial drug regimens is often suboptimal. Studies specifically examining adherence in the case of drugs used for malaria prophylaxis have reported several reasons for the low rates of adherence, including forgetfulness, fear of adverse events, discomfort of swallowing or of swallowing too many pills, receiving inaccurate pre-travel advice from nonmedical or medical professionals, incorrect risk perception, failure to take any prophylaxis, and inaccurate understanding of malaria transmission (Adshead, 2014; Behrens et al., 1998; Cunningham et al., 2014; Goodyer et al., 2011; Hopperus Buma et al., 1996; Huzly et al., 1996; Landman et al., 2015; Laver et al., 2001; Ollivier et al., 2008; Phillips and Kass, 1996; Ropers et al., 2008; Saunders et al., 2015). In studies of people who are employed by or participate in organizations in which the use of antimalarial drugs for prophylaxis is required (e.g., military, Peace Corps, Department of State), reported adherence rates may be inflated. The dosing regimens vary for each antimalarial drug, so that adherence is more achievable for some drugs than others. For example, doxycycline and A/P must be taken every day, whereas mefloquine and tafenoquine only need to be taken once a week. One study of U.S. soldiers serving in Afghanistan in 2007 found that 60% were fully
adherent with daily doxycycline and 80% reported full adherence with weekly mefloquine (Saunders et al., 2015). Similar rates of low adherence have been reported in other international military forces. In 2006 a study of French troops using daily doxycycline for prophylaxis found that 63.4% were nonadherent, based on measured plasma concentrations of doxycycline (Ollivier et al., 2008), while Dutch marine battalions stationed for 6 months in Cambodia reported 86.3% fully adhered with weekly mefloquine prophylaxis (Hopperus Buma et al., 1996). FDA and drug manufacturers note that even for individuals who adhere completely to drug dosing regimens, no drug is 100% efficacious, and it is still possible to develop clinical symptoms of malaria. Therefore, in addition to using antimalarial prophylaxis, the use of other preventative measures is recommended.
In malaria-endemic areas, DoD policy dictates that such personal protective measures as insect repellent (most commonly DEET), bed nets, and permethrin-impregnated uniforms be used in addition to a malaria prophylactic drug. Although DEET is known to be effective against mosquitos and other insects, during some deployments, such as Somalia, troops did not like that DEET caused dust to cake on the areas of exposed skin where it was used (Ledbetter et al., 1995). Similarly, the operational work and living environments of military personnel do not always lend themselves to the appropriate use of protective measures. For example, bed nets were reportedly not used in some combat environments for fear that the poles that bed nets were suspended on made the troops larger targets to the enemy (Ledbetter et al., 1995). One survey of U.S. soldiers who served in Afghanistan found that only 1% reported consistent use of bed nets, 4% reported consistent use of mosquito repellent, and 31% reported that all of their uniforms had been treated with permethrin; however, 44% and 20% of the survey respondents reported that bed nets and skin repellents, respectively, had not been issued during the deployment (Saunders et al., 2015).
While real or perceived side effects and adverse events of drugs used for malaria prophylaxis are common reasons given for the lack of adherence to them, other factors may contribute, especially during deployments. Forgetfulness, especially when troops have irregular schedules, have little or disrupted sleep, or go on leave, is common (Hopperus Buma et al., 1996; Ledbetter et al., 1995; Mayet et al., 2010; Saunders et al., 2015). Other reasons for a lack of adherence that have been reported in the published literature include not believing malaria is a serious threat or that the threat was “over,” or accidently laundering pills (Hopperus Buma et al., 1996; Ledbetter et al., 1995; Saunders et al., 2015). Unit commanders may require that the taking of antimalarials is directly observed by unit medical personnel to improve adherence.
Concurrent Exposures of Military Service
This section focuses on the many natural and anthropogenic exposures that U.S. service members and veterans may have experienced that may confound
associations between the use of antimalarial drugs and long-term health outcomes. As those antimalarial drugs that have been used in the past 25 years are of highest interest to the Department of Veterans Affairs (VA), this would include their use in service members who were deployed in support of OEF, OIF, and Operation New Dawn (OND) in Iraq and Afghanistan as well as surrounding areas included in the Southwest Asia Theater of Operations4 and also peacekeeping, humanitarian, and engineering activities in Haiti, Liberia, Somalia, and other malaria-endemic areas around the world. Concurrent military exposures of the 1990–1991 Persian Gulf War (Operation Desert Storm and Operation Desert Eagle) are also considered because the antimalarials used for prophylaxis were chloroquine (DoD, 1993; VA, 1993) and doxycycline (Thornton et al., 2005) depending on the service branch and unit commander, with some reports of mefloquine use as well (see Kotwal et al., 2005), and these are antimalarial drugs that VA considers to be of high interest. Some of the potentially confounding exposures were unique to specific conflicts, such as the numerous oil-well fires and their smoke, the release of the nerve agents sarin and cyclosarin, and the use of pyridostigmine bromide as a prophylaxis for the nerve agents in the 1990–1991 Persian Gulf War. Other exposures, such as vaccinations against anthrax and botulinum, while uncommon, were used in the Persian Gulf War as well as the recent conflicts in Iraq and Afghanistan.
Understanding the adverse health effects of military service requires examining a combination of many complex issues, some of which may occur simultaneously for an individual. Some of these issues are explored below (and are based on information found in the National Academies Gulf War and Health report series as well as in several other reports that examined the health effects related to myriad exposures that service members received during their deployments). These issues include exposure to multiple biologic and chemical agents, combat and other psychologic stressors, the deployment environment, and individual variability factors.
Environmental and Chemical Exposures
During deployments, service members may have had a variety of environmental exposures related to their deployment including solvents, fumes from kerosene heaters in unvented tents, and exposure to petroleum-based combustion products including diesel fuel and leaded gasoline that were used in cooking stoves and portable generators and to suppress sand and dust in desert environments and aid in the burning of waste and trash in open air burn pits. Such combustion products may contain many hazardous agents such as polyaromatic hydrocarbons, dioxins, furans, and methane.
___________________
4 VA defines the Southwest Asia Theater of operations to include the following locations: Iraq, Kuwait, Saudi Arabia, Bahrain, Gulf of Aden, Gulf of Oman, Oman, Qatar, and the United Arab Emirates; the waters of the Persian Gulf, Arabian Sea, and the Red Sea; and the airspace above these regions.
In areas of conflict, pesticide exposures are generally widespread among troops as their units attempt to resist the local insect and rodent populations. DEET and permethrin-impregnated uniforms are nearly ubiquitous in deployments to Southeast Asia, Southwest Asia, and Africa. Other pesticides that have been reportedly used, particularly in Iraq and Afghanistan, include methyl carbamates (e.g., proxpur, carbaryl), organophosphates (e.g., chlorpyrifos, diazinon, malathion), pyrethroids, lindane, and chlorinated hydrocarbons (DoD, 2001; RAND, 2000). However, objective information regarding individual levels of pesticide exposure is generally not available, and reports by individual veterans as to their use of and possible exposure to pesticides are subject to considerable recall bias.
Many environmental and chemical exposures could be related to particular activities related to a service member’s or veteran’s military occupational specialty. In the conflicts in Iraq and Afghanistan, the majority of occupational chemical exposures appear to have been related to repair and maintenance (chlorinated hydrocarbons), sandblasting (abrasive particles), vehicle repair (carbon monoxide and organic solvents), weapon repair (lead particles), and welding and cutting (chromates, nitrogen dioxide, and heated metal fumes). In addition, troops painted vehicles and other equipment used in the Persian Gulf region with a chemical-agent-resistant coating either before they were shipped to the Persian Gulf or while they were at ports in Saudi Arabia. Working conditions in the field were not ideal, and recommended occupational-hygiene standards might not have been followed at all times (NASEM, 2016).
In addition to the requirements for service members to be up to date on standard vaccines, certain military deployments require additional vaccines or prophylactic agents, such as for cholera, meningitis, and typhoid. Deployment to malaria-endemic locations, such as Southwest Asia, Southeast Asia, and Africa, require the issue of an approved antimalarial drug. In some combat theaters additional protective measures may be needed. For example, during the 1990–1991 Persian Gulf War, about 150,000 troops received anthrax vaccine and about 8,000 troops received botulinum toxoid vaccine, although medical records from this period are notably lacking information regarding who received these vaccines, how frequently the vaccines were administered, or the timing of vaccinations relative to other putative exposures (IOM, 2000).
Some environmental exposures resulted from the conflict itself, such as exposures to the depleted uranium used in munitions, excessive heat or humidity, additional vaccines administered, and smoke from open burn pits. Some of the exposures could be constant, such as dust, heat, and pesticides, while other exposures were intermittent or infrequent.
Combat Exposures
Although modern warfare has resulted in fewer deaths and casualties than earlier conflicts—those in Vietnam and Korea, World War I and World War II—
there are numerous opportunities for exposure to potentially harmful situations during deployment. Combat is widely acknowledged to be one of the most intense experiences that a person can have and may include many threatening situations such as killing or attempting to kill an enemy; being shot at by others; exposure to dead and wounded comrades, enemy combatants, and civilians; and being injured. For the 1990–1991 Gulf War and the OEF/OIF/OND conflicts, these situations included being in the vicinity of Scud missile explosions, contact with improvised explosive devices, contact with prisoners of war, direct combat duty, coming under small-arms fire, having artillery close by (Hoge et al., 2004; Kang et al., 2000; Unwin et al., 1999), and the fear of terrorist or chemical attacks. Many surveys have been conducted to assess veterans’ combat experiences and exposures (e.g., Millennium Cohort Study, National Health Survey of Gulf War Era Veterans and Their Families, National Health Study for a New Generation of U.S. Veterans), and in nearly all of them, veterans have reported exposure to a wide variety of threatening or harmful situations during their deployments. In one study that conducted a survey of the combat experiences and mental health problems of Army or Marine Corps service members deployed to either OEF or OIF, researchers found that many of the respondents indicated having had several of these experiences. For example, among the marines deployed to Iraq (n = 815), 97% reported being shot at or receiving small arms fire, 95% reported being attacked or ambushed, 94% reported seeing dead bodies or human remains, and 92% reported receiving incoming artillery, rocket, or mortar fire. Although the percentages were slightly lower, soldiers deployed to Iraq (n = 894) also indicated having had similar combat experiences (Hoge et al., 2004).
Psychological Stressors
Deployment to a war zone in general, and combat exposure in particular, may result in psychiatric and physical sequelae among service members. In addition to the cramped and uncomfortable living conditions, the many potential environmental and chemical exposures, and the threat of combat, a variety of other stressors may also exert psychologic effects. Rapid mobilization may exert additional substantial pressure on those who are deployed, disrupting lives and separating families. Uncertainty about the duration of deployment was a continuing concern for U.S. troops during the Gulf War, OEF, and OIF, particularly during the early phases of the buildup. For the large numbers of reservists and National Guard members who were mobilized, there was added uncertainly about whether their jobs would be available when they returned to civilian life (VA, 2011). Although better mechanisms for and access to communication with family in the United States exist than was the case for earlier conflicts such as Vietnam, deployment can still add to the stress of maintaining family relationships, particularly for reserve and National Guard personnel who may not have deployed with a familiar or cohesive unit. Surveys of both active-duty and reserve and National Guard soldiers deployed to
Iraq in support of OIF found that the most important noncombat stressors were deployment length and family separation (MHAT, 2006a,b). Stressful working conditions, disrupted sleep patterns, and prolonged separation from families may exacerbate the psychological stressors (Adshead, 2014).
Although historically women who served in the military were not allowed to serve in direct combat specialties, they were deployed in combat support roles as administrators, air traffic controllers, logisticians, ammunition technicians, engineering equipment mechanics, ordnance specialists, communicators, radio operators, drivers, law enforcement specialists, aviators, and guards. Still others served on hospital, supply, oiler, and ammunition ships or served as public affairs officers and chaplains (DoD, 2004), and they experienced many of the same exposures and stressors as men when deployed. In addition, several studies of deployment experiences found that female military personnel were more likely to experience sexual harassment and assault than male personnel (Goldzweig et al., 2006; Kang et al., 2005; Vogt et al., 2005; Wolfe et al., 1998).
Following deployment, many veterans experience high levels of stress during the transition to civilian life (IOM, 2008, 2013; Mobbs and Bonanno, 2018). In particular, coming-home experiences may be challenging, with numerous stressors including relationships with spouses and family, and parenting roles (Mobbs and Bonanno, 2018; Steenkamp et al., 2017). Some of these coming-home stressors may be related to the military experience itself. Vietnam veterans, for example, frequently experienced social rejection and were stereotyped for being perceived as harming vulnerable Vietnamese populations, which placed them in a difficult position on top of other mental health issues they were experiencing (Marmar et al., 2015; Steenkamp et al., 2017). With a better understanding of those experiences by policy makers and professionals, Iraq and Afghanistan service members were treated more positively; however, a host of highly needed resources, such as access to mental health care, legal help, and vocational opportunities, has been limited (IOM, 2013). Whereas the lifetime prevalence of posttraumatic stress disorder (PTSD) in U.S. adults was estimated to be 6.8% according to the National Comorbidity Survey–Replication (Harvard Medical School, 2007), the prevalence of PTSD among post-9/11 veterans is much higher. Among post-9/11 veterans using VA health services, nearly one-quarter of them had a diagnosis of PTSD (IOM, 2014). Similarly, a meta-analysis of PTSD prevalence in post-9/11 veterans that included 33 studies published between 2007 and 2013 involving 4,945,897 OEF/OIF veterans estimated the overall prevalence of PTSD among these veterans to be 23% (Fulton et al., 2005). Despite some resources and programs available for returning veterans, the stresses of deployment and reintegration elevate their risk for a host of military-related psychiatric problems, including PTSD, depression, anxiety disorder, substance abuse, and suicidal ideation and attempts (Pietrzak et al., 2009; Thomas et al., 2017).
Living Conditions
Combat troops deployed to the conflicts in Southwest Asia were often crowded into warehouses and tents upon arrival in the Persian Gulf region and then often moved to isolated desert locations (NASEM, 2016). Most troops lived in tents and slept on cots lined up side by side, affording virtually no privacy or quiet. Sanitation was often primitive, with strains on latrines and communal washing facilities. Hot showers were infrequent, the interval between laundering uniforms was sometimes long, and desert flies were a constant nuisance, as were scorpions and snakes. Military personnel worked long hours and had narrowly restricted outlets for relaxation. Troops were ordered not to fraternize with local people, and alcoholic drinks were prohibited in deference to religious beliefs in the host countries. A mild traveler’s type of diarrhea affected more than half of the troops in some units; one study of ground forces found that 57% of those surveyed had experienced at least one episode of diarrhea within the first 2 months of deployment and of those, 20% were unable to perform their military duties while affected (Hyams et al., 1991). Among British and Australian medical teams that were part of the coalition forces, 69% of British troops and 36% of Australian troops experienced diarrhea, with some episodes lasting for several days (Rudland et al., 1996). Fresh fruits and vegetables from neighboring countries were identified as the cause and were removed from the diet. Thereafter, the diet consisted mostly of packaged foods and bottled water.
Depending on the deployment location, weather may create additional stressors. During the summer months in Iraq, the air temperatures could reach as high as 115°F and the sand temperatures as high as 150°F. Except for coastal regions, the relative humidity was less than 40%. Troops had to drink large quantities of water to prevent dehydration. Although the summers were hot and dry, temperatures in winter in Iraq and Afghanistan were low, with wind chill temperatures at night dropping to well below freezing. Wind and blowing sand made the protection of skin and eyes imperative. Goggles and sunglasses helped somewhat, but visibility was often poor.
Interindividual Variability
Differences among people in their genetic, biologic, psychologic, and social vulnerabilities add to the complexity of determining health outcomes related to specific agents (NASEM, 2016). The likelihood of observing a particular health outcome may differ for people with increased sensitivity to an agent, such as G6PD deficiency as described earlier in the chapter. For example, a person who is a poor metabolizer of a particular substance, depending on his or her genetic makeup, might be at higher or lower risk for specific health effects if exposed to the substance.
All antimalarial drugs used for prophylaxis in adults are prescribed as fixed dose regimens in which the amount of drug (e.g., one tablet) and unit of time (e.g.,
once daily, weekly) is specified. For example, the recommended dosing for A/P is one tablet (250 mg atovaquone and 100 mg proguanil) per day beginning 1–2 days before entering and continuing throughout the stay, and for 7 days after leaving an endemic area. Consistent with FDA indications, the same fixed dose is prescribed to all adult individuals, regardless of sex, weight, or age. As such, certain antimalarials may be more likely to be associated with side effects or adverse events in people with certain demographic characteristics. For example, among people who use mefloquine, more women report adverse events than men. Because in general women weigh less than men and have a smaller vascular volume, a fixed dose tablet of an antimalarial may result in higher plasma levels of the drug in people of lower weight (women) than in people of heavier weight (men). Some studies have shown that adverse events are related to the concentration of drug in the blood (Schwartz et al., 2001), not to the absolute dose of drug delivered. Therefore, if the target of drug delivery was for a specific plasma level across all adult users, then the drug would have to be dosed on a mg/kg basis. Since the goal of prophylaxis is protection against malaria, the fixed dose was determined based on the pharmacokinetic studies of a dose that offers the best combination of protection and tolerability and that was easy to mass produce.
REFERENCES
60 Degrees Pharmaceuticals. 2018. ArakodaTM (Tafenoquine Succinate) Tablets for the Prevention of Malaria in Adults. Washington, DC: 60 Degrees Pharmaceuticals.
Adshead, S. 2014. The Adverse Effects of Mefloquine in Deployed Military Personnel. J R Nav Med Serv 100(3):232-237.
AFHSB (Armed Forces Health Surveillance Branch). 2019. Update: Malaria, U.S. Armed Forces, 2018. MSMR 26(2):1-28.
AFHSC (Armed Forces Health Surveillance Center). 2013. Update: Malaria, U.S. Armed Forces, 2012. MSMR 20(1):2-5.
AFHSC. 2014. Update: Malaria, U.S. Armed Forces, 2013. MSMR 21(1):4-7.
AFHSC. 2015. Update: Malaria, U.S. Armed Forces, 2014. MSMR 22(1):2-6.
AFHSC. 2016. Update: Malaria, U.S. Armed Forces, 2015. MSMR 23(1):2-6.
Arguin, P. M., and A. J. Magill. 2017. For the record: A history of malaria chemoprophylaxis. https://wwwnc.cdc.gov/travel/yellowbook/2018/infectious-diseases-related-to-travel/emfor-the-recorda-history-of-malaria-chemoprophylaxisem (accessed December 18, 2018).
Baird, J. K. 2012. Elimination therapy for the endemic malarias. Curr Infect Dis Rep 14(3):227-237. Baird, J. K. 2018. Tafenoquine for travelers’ malaria: Evidence, rationale, and recommendations. J Travel Med 25(1):1-13.
Baird, J. K. 2019. 8-aminoquinoline therapy for latent malaria. Clin Microbiol Rev 32(4):e00011-19.
Behrens, R. H., R. B. Taylor, D. I. Pryce, and A. S. Low. 1998. Chemoprophylaxis compliance in travelers with malaria. J Travel Med 5(2):92-94.
Boggild, A., M. E. Parise, L. S. Lewis, and K. C. Kain. 2007. Atovaquone-proguanil: Report from the CDC Expert Meeting on Malaria Chemoprophylaxis (II). Am J Trop Med Hyg 76(2):208-223.
Bowman, Z. S., J. D. Morrow, D. J. Jollow, and D. C. McMillan. 2005. Primaquine-induced hemolytic anemia: Role of membrane lipid peroxidation and cytoskeletal protein alterations in the hemotoxicity of 5-hydroxyprimaquine. J Pharmacol Exp Ther 314:833-845.
Brundage, J. 2003. Conserving the fighting strength: Milestones of operational military preventive medicine research. In Military preventive medicine: Mobilization and deployment. 1st ed. Vol. 1: Office of the Surgeon General, U.S. Army.
Camarda, G., P. Jirawatcharadech, R. S. Priestley, A. Saif, et al. 2019. Antimalarial activity of Primaquine operates via a two-step biochemical relay. Nat Commun 10(3226).
Canfield, C. J., M. Pudney, and W. E. Gutteridge. 1995. Interactions of atovaquone with other antimalarial drugs against Plasmodium falciparum in vitro. Exp Parasitol 80:373-381.
CDC (Centers for Disease Control and Prevention). 1993. Malaria among U.S. military personnel returning from Somalia, 1993. MMWR 42(27):523-526.
CDC. 2017. DPDx—Laboratory identification of parasites of public health concern. https://www.cdc.gov/dpdx/malaria/index.html (accessed March 13, 2019).
CDC. 2018. Drug resistance in the malaria-endemic world. https://www.cdc.gov/malaria/malaria_worldwide/reduction/drug_resistance.html (accessed November 12, 2019).
CDC. 2019a. Chapter 4: Travel related infectious diseases. In CDC Yellowbook. https://wwwnc.cdc.gov/travel/yellowbook/2020/travel-related-infectious-diseases/malaria (accessed March 14, 2019).
CDC. 2019b. Treatment of malaria: guidelines for clinicians (United States). https://www.cdc.gov/malaria/resources/pdf/treatment_guidelines_101819.pdf (accessed December 27, 2019).
Chinevere, T. D., C. K. Murray, E. Grant, Jr., G. A. Johnson, F. Duelm, and D. R. Hospenthal. 2006. Prevalence of glucose-6-phosphate dehydrogenase deficiency in U.S. Army personnel. Mil Med 171(9):905-907.
Cunningham, J., J. Horsley, D. Patel, A. Tunbridge, and D. G. Lalloo. 2014. Compliance with long-term malaria prophylaxis in British expatriates. Travel Med Infect Dis 12:341-348.
DeJulio, P. A. 2016. You’re the flight surgeon. Aerosp Med Hum Perform 87(4):429-432.
Denshaw-Burke, M., A. L. Curran, D. C. Savior, and M. Kumar. 2018. Methemoglobinemia. Medscape. https://webcache.googleusercontent.com/search?q=cache:MoqV2XjOJHIJ:https://emedicine.medscape.com/article/204178-overview+&cd=1&hl=en&ct=clnk&gl=us (accessed December 23, 2019).
DoD (Department of Defense). 1993. United States Army Reserve in Operation Desert Storm: Reservists of the Army Medical Department (monograph, J. R. Brinkerhoff, T. Silva, and J. Seitz, September 23, 1993). https://apps.dtic.mil/dtic/tr/fulltext/u2/a277591.pdf (accessed November 15, 2019).
DoD. 2001. Environmental exposure report: Pesticides, final report. Falls Church, VA: Department of Defense.
DoD. 2004. Task force report on care for victims of sexual assault. Washington, DC: Department of Defense. https://vawnet.org/material/task-force-report-care-victims-sexual-assault (accessed November 12, 2019).
DoD. 2009. Policy memorandum on the use of mefloquine (Lariam®) in malaria prophylaxis (September 4, 2009, #HA 09-017). Provided by COL Andrew Wiesen, M.D., M.P.H., Director, Preventive Medicine, Health Readiness Policy and Oversight, Office of the Assistant Secretary of Defense (Health Affairs), DoD, January 25, 2019.
DoD. 2013. Memorandum on guidance on medications for prophylaxis of malaria (April 15, 2013). Provided by COL Andrew Wiesen, M.D., M.P.H., Director, Preventive Medicine, Health Readiness Policy and Oversight, Office of the Assistant Secretary of Defense (Health Affairs), DoD, January 25, 2019.
Dorwald, F. Z. 2012. Lead optimization for medicinal chemists: Pharmacokinetic properties of functional groups and organic compounds. Mörlenbach, Germany: Wiley-VCH.
Downs, W. G., P. A. Harper, and E. T. Lisamsky. 1947. Malaria and other insect-borne diseases in the South Pacific Campaign, 1942-1945. II Epidemiology of insect-borne diseases in Army troops. Am J Trop Med 27:69-89.
Dugbartey, A. T., M. T. Dugbartey, and M. Y. Apedo. 1998. Delayed neuropsychiatric effects of malaria in Ghana. J Nerv Ment Dis 186(3):183-186.
Ebstie, Y. A., S. M. Abay, W. T. Tadesse, and D. A. Ejigu. 2016. Tafenoquine and its potential in the treatment and relapse prevention of Plasmodium vivax malaria: The evidence to date. Drug Des Devel Ther 10:2387-2399.
Ecker, A., A. M. Leane, J. Clain, and D. A. Fidock. 2012. PfCRT and its role in antimalarial drug resistance. Trends Parasitol 28(11):504-514.
Fasinu, P. S., N. P. Dhammika Nanayakkara, Y.-H. Wang, N. D. Chaurasiya, H. M. Bandara Herath, J. D. McChesney, B. Avula, I. Khan, B. L. Tekwani, and L. A. Walker. 2019. Formation primaquine-5, 6-orthoquinone, the putative active and toxic metabolite of primaquine via direct oxidation in human erythrocytes. Malar J 18(30).
FDA (Food and Drug Administration). 2018a. FDA briefing document. Tafenoquine tablet, 100 mg. Meeting of the Antimicrobial Drugs Advisory Committee (AMDAC). July 26, 2018. https://www.fda.gov/media/114753/download (accessed November 11, 2019).
FDA. 2018b. Package insert for Arakoda™ (tafenoquine) tablets. https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=210607 (accessed October 29, 2019).
FDA. 2019. Package insert for Malarone (atovaquone and proguanil hydrochloride) tablets and Malarone (atovaquone and proguanil hydrochloride) pediatric tablets. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/021078s023lbl.pdf (accessed October 3, 2019).
Fernando, S. D., D. M. Gunawardena, M. R. Bandara, D. De Silva, R. Carter, K. N. Mendis, and A. R. Wickremasinghe. 2003. The impact of repeated malaria attacks on the school performance of children. Am J Trop Med Hyg 69(6):582-588.
Foley, M., and L. Tilley. 1998. Quinoline antimalarials: Mechanisms of action and resistance and prospects for new agents. Pharmacol Ther 79(1):55-87.
Fukuda, M., G. A. Raczniak, M. S. Riddle, M. Forgione, and A. J. Magill. 2018. Special considerations for U.S. military deployments. In Yellow book. Atlanta, GA: Centers for Disease Control and Prevention. https://relief.unboundmedicine.com/relief/view/cdc-yellowbook/204140/all/Special_Considerations_for_US_Military_Deployments (accessed November 12, 2019).
Fulton, J. J., P. S. Calhoun, H. R. Wagner, A. R. Schry, L. P. Hair, N. Feeling, E. Elbogen, and J. C. Beckham. 2005. The prevalence of posttraumatic stress disorder in Operation Enduring Freedom/Operation Iraqi Freedom (OEF/OIF) veterans: A meta-analysis. J Anxiety Disord 31:98-107.
Gay, F., D. Bustos, and B. Traore. 1997. In vitro response of Plasmodium falciparum to atovaquone and correlation with other antimalarials: Comparison between African and Asian strains. Am J Trop Med 56:315-317.
Goldzweig, C. L., T. M. Balekian, C. Rolon, E. M. Yano, and P. G. Shekelle. 2006. The state of women veterans’ health research: Results of a systematic literature review. J Gen Intern Med 21(Suppl 3):S82-S92.
Goodyer, L., L. Rice, and A. Martin. 2011. Choice of and adherence to prophylactic antimalarials. J Travel Med 18(4):245-249.
Gunjan, S., S. K. Singh, T. Sharma, H. Dwivedi, B. S. Chauhan, M. Imran Siddiqi, and R. Tripathi. 2016. Mefloquine induces ROS mediated programmed cell death in malaria parasite: Plasmodium. Apoptosis 21(9):955-964.
Harvard Medical School. 2007. Lifetime prevalence of DSM-IV/WMH-CIDI disorders by sex and cohort (n = 9,282). https://www.hcp.med.harvard.edu/ncs/ftpdir/NCS-R_Lifetime_Prevalence_Estimates.pdf (accessed September 25, 2019).
Hellgren, U., I. Berggren-Palme, Y. Bergvist, and M. Jerling. 1997. Enantioselective pharmacokinetics of mefloquine during long-term intake of the prophylactic dose. Br J Clin Pharmacol 44:119-124.
Helsby, N. A., S. A. Ward, R. E. Howell, and A. M. Breckenridge. 1990. The pharmacokinetics and activation of proguanil in man: Consequences of variability in drug metabolism. Br J Clin Pharmacol 30:287-291.
Hoge, C. W., C. A. Castro, S. C. Messer, D. McGurk, D. I. Cotting, and R. L. Koffman. 2004. Combat duty in Iraq and Afghanistan, mental health problems, and barriers to care. NEJM 351:13-22.
Hopperus Buma, A. P., P. P. van Thiel, H. O. Lobel, C. Ohrt, E. J. van Ameijden, R. L. Veltink, D. C. Tendeloo, T. van Gool, M. D. Green, G. D. Todd, D. E. Kyle, and P. A. Kager. 1996. Long-term malaria chemoprophylaxis with mefloquine in Dutch Marines in Cambodia. J Infect Dis 173:1506-1509.
Horne, R. 2006. Compliance, adherence, and concordance: Implications for asthma treatment. Chest 130(1):65S-72S.
Huzly, D., C. Schonfeld, W. Beuerle, and U. Bienzle. 1996. Malaria chemoprophylaxis in German tourists: A prospective study on compliance and adverse reactions. J Travel Med 3(3):148-155.
Hyams, K. C., A. L. Bourgeois, B. R. Merrell, P. Rozmajzl, J. Escamilla, S. A. Thornton, G. M. Wasserman, A. Burke, P. Echeverria, K. Y. Green, A. Z. Kapikian, and J. N. Woody. 1991. Diarrheal disease during Operation Desert Shield. NEJM 325(20):1423-1428.
Idro, R., J. A. Carter, G. Fegan, B. G. Neville, and C. R. Newton. 2006. Risk factors for persisting neurological and cognitive impairments following cerebral malaria. Arch Dis Child 91(2):142-148.
Idro, R., K. Marsh, C. C. John, and C. R. Newton. 2010. Cerebral malaria: Mechanisms of brain injury and strategies for improved neurocognitive outcome. Pediatr Res 68(4):267-274.
Idro, R., A. Kakooza-Mwesige, B. Asea, K. Sebyala, P. Bangirana, R. O. Opoka, S. K. Lubowa, M. Semrud-Clikeman, C. C. John, and J. Nalugya. 2016. Cerebral malaria is associated with long-term mental health disorders: A cross sectional survey of a long-term cohort. Malar J 15:184.
IOM (Institute of Medicine). 2000. Gulf War and health, volume 1: Depleted uranium, pyridostigmine bromide, sarin, vaccines. Washington, DC: National Academy Press.
IOM. 2006. Battling malaria: Strengthening the U.S. military malaria vaccine program. Washington, DC: The National Academies Press.
IOM. 2008. Gulf War and health, volume 6: Psychologic, psychiatric, and psychosocial effects of deployment-related stress. Washington, DC: The National Academies Press.
IOM. 2013. Returning home from Iraq and Afghanistan: Assessment of readjustment needs of veterans, service members, and their families. Washington, DC: The National Academies Press.
IOM. 2014. Treatment for posttraumatic stress disorder in military and veteran populations: Final assessment. Washington, DC: The National Academies Press.
John, C. C., A. Panoskaltsis-Mortari, R. O. Opoka, G. S. Park, P. J. Orchard, A. M. Jurek, R. Idro, J. Byarugaba, and M. J. Boivin. 2008. Cerebrospinal fluid cytokine levels and cognitive impairment in cerebral malaria. Am J Trop Med Hyg 78(2):198-205.
Kang, H. K., C. M. Mahan, K. Y. Lee, C. A. Magee, and F. M. Murphy. 2000. Illnesses among United States veterans of the Gulf War: A population-based survey of 30,000 veterans. J Occup Environ Med 42(5):491-501.
Kang, H., N. Dalager, C. Mahan, and E. Ishii. 2005. The role of sexual assault on the risk of PTSD among Gulf War veterans. Ann Epidemiol 15(3):191-195.
Kaur, H., E. L. Allan, I. Mamadu, Z. Hall, O. Ibe, M. El Sherbiny, A. van Wyk, S. Yeung, I. Swamidoss, M. D. Green, P. Dwivedi, M. J. Culzoni, S. Clarke, D. Schellenberg, F. M. Fernández, and O. Onwujekwe. 2015. Quality of artemisinin-based combination formulations for malaria treatment: Prevalence and risk factors for poor quality medicines in public facilities and private sector drug outlets in Enugu, Nigeria. PLOS ONE 10(5):e0125577.
Kitchen, L. W., D. W. Vaughn, and D. R. Skillman. 2006. Role of US military research programs in the cevelopment of US Food and Drug Administration-approved antimalarial drugs. Clin Infect Dis 43:67-71.
Kotwal, R. S., R. B. Wenzel, R. A. Sterling, W. D. Porter, N. N. Jordan, and B. P. Petruccelli. 2005. An outbreak of malaria in US Army Rangers returning from Afghanistan. JAMA 293(2):212-216.
Lalloo, D. G., and Magill. 2019. Travel medicine: 14 - malaria: Epidemiology and risk to the traveler. Edited by J. S. Keystone. 4th ed. Elsevier.
Landman, K. Z., K. R. Tan, and P. M. Arguin. 2015. Adherence to malaria prophylaxis among Peace Corps volunteers in the Africa region, 2013. Travel Med Infect Dis 12:61-68.
Laver, S. M., J. Wetzels, and R. H. Behrens. 2001. Knowledge of malaria, risk perception, and compliance with prophylaxis and personal and environmental preventive measures in travelers exiting Zimbabwe from Harare and Victoria Falls International Airport. J Travel Med 8:298-303.
Ledbetter, E., K. R. Hanson and M. R. Wallace. 1995. Malaria in Somalia: Lessons in prevention. JAMA 273(10):774-775.
Ley, B., G. Bancone, L. von Seidlein, K. Thriemer, J. S. Richards, G. J. Domingo, and R. N. Price. 2017. Methods for the field evaluation of quantitative G6PD diagnostics: A review. Malar J 16(1):361.
Liu, H., L. A. Walker, and R. J. Doerksen. 2011. DFT study on the radical anions formed by primaquine and its derivatives. Chem Res Toxicol 24(9):1476-1485.
Looareesuwan, S., J. D. Chulay, C. J. Canfield, and D. B. Hutchinson. 1999. Malarone (atovaquone and proguanil hydrochloride): A review of its clinical development for treatment of malaria. Malarone clinical trials study group. Am J Trop Med Hyg 60(4):533-541.
Mace, K. E., P. M. Arguin, N. W. Lucchi, and K. R. Tan. 2019. Malaria surveillance—United States, 2016. MMWR Surveill Summ 68(5):1-35.
Marcsisin, S. R., J. C. Sousa, G. A. Reichard, D. Caridha, Q. Zeng, N. Roncal, R. McNulty, J. Careagabarja, R. J. Sciotti, J. W. Bennett, V. E. Zottig, G. Deye, Q. Li, L. Read, M. Hickman, N. P. Dhammika Nanayakkara, L. A. Walker, B. Smith, V. Melendez, and B. S. Pybus. 2014. Tafenoquine and NPC-1161b require CYP 2D metabolism for anti-malarial activity: Implications for the 8-aminoquinoline class of anti-malarial compounds. Malar J 13(2):2.
Marmar, C. R., W. Schlenger, C. Henn-Haase, M. Qian, E. Purchia, M. Li, N. Corry, C. S. Williams, C. L. Ho, D. Horesh, K. I. Karstoft, A. Shalev, and R. A. Kulka. 2015. Course of posttraumatic stress disorder 40 years after the Vietnam War: Findings from the National Vietnam Veterans Longitudinal Study. JAMA Psychiatry 72(9):875-881.
Mayet, A., D. Lacassagne, N. Juzan, B. Chaudier, R. Haus-Cheymol, F. Berger, O. Romand, L. Ollivier, C. Verret, X. Deparis, and A. Spiegel. 2010. Malaria outbreak among French army troops returning from the Ivory Coast. J Travel Med 17(5):353-355.
MHAT (Mental Health Advisory Team). 2006a. Mental Health Advisory Team (MHAT) III. Operation Iraqi Freedom 04-06: Final Report. Washington, DC: Office of the Surgeon Multinational Force-Iraq and Office of the Surgeon General United States Army Medical Command. 29 May 2006. http://www.armymedicine.army.mil/news/mhat/mhat_iii/mhat-iii.cfm (accessed October 22, 2019).
MHAT. 2006b. Mental Health Advisory Team (MHAT) IV Operation Iraqi Freedom 05-07: Final Report. Washington, DC: Office of the Surgeon Multinational Force-Iraq and Office of the Surgeon General United States Army Medical Command. 17 November 2006. http://www.armymedicine.army.mil/news/mhat/mhat_iv/MHAT_IV_Report_17NOV06.pdf (accessed October 22, 2019).
Mobbs, M. C., and G. A. Bonanno. 2018. Beyond war and PTSD: The crucial role of transition stress in the lives of military veterans. Clin Psychol Rev 59:137-144.
Moss, W. J., and R. H. Morrow. 2014. The epidemiology and control of malaria. In K. E. Nelson and C. William, Infectious disease epidemiology: Theory and practice. Burlington, MA: Jones & Bartlett Learning.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. Gulf War and health, volume 10: Update of health effects of serving in the Gulf War. Washington, DC: The National Academies Press.
National Stroke Association. 2012. Medication Adherence and Compliance. National Stroke Association: 1-9. https://oregon.providence.org/~/media/Files/Providence%20OR%20Migrated%20PDFs/Patients%20Toolkit/formsinstructions/nsa_med_adherencebooklet.pdf (accessed November 3, 2019).
Newton, P. N., K. Hanson, C. Goodman, and ACTwatch Group. 2017. Do anti-malarials in Africa meet quality standards? The market penetration of non-quality assured artemisinin combination therapy in eight African countries. Malar J 16:204.
NIH (National Institutes of Health). n.d. Hemolytic anemia. https://www.nhlbi.nih.gov/health-topics/hemolytic-anemia (accessed October 1, 2019).
Nixon, G. L., D. M. Moss, A. E. Shone, D. G. Lalloo, N. Fisher, P. M. O’Neill, S. A. Ward, and G. A. Biagini. 2013. Antimalarial pharmacology and therapeutics of atovaquone. J Antimicrob Chemother 68:977-985.
Nkhoma, E. T., C. Poole, V. Vannappagari, S. A. Hall, and E. Beutler. 2009. The global prevalence of glucose-6-phosphate dehydrogenase deficiency: A systematic review and meta-analysis. Blood Cells Mol Dis 42(3):267-278.
Ockenhouse, C. F., A. Magill, D. Smith, and W. Milhous. 2005. History of U.S. military contributions to the study of malaria. Mil Med 170(4):12-16.
Ollivier, L., R. Michel, M. P. Carlotti, P. Mahé, O. Romand, A. Todesco, R. Migliani, and J. P. Boutin. 2008. Chemoprophylaxis compliance in a French battalion after returning from malaria endemic area. J Travel Med 15:355-357.
O’Neill, P. M., S. A. Ward, N. G. Berry, J. P. Jeyadevan, G. A. Biagini, E. Asadollaly, B. K. Park, and P. G. Bray. 2006. A medicinal chemistry perspective on 4-aminoquinoline antimalarial drugs. Curr Top Med Chem 6(5):479-507.
Pal, S., P. Bansil, G. Bancone, S. Hrutkay, M. Kahn, G. Gornsawun, P. Penpitchaporn, C. S. Chu, F. Nosten, and G. J. Domingo. 2019. Evaluation of a novel quantitative test for glucose-6phosphate dehydrogenase deficiency: Bringing quantitative testing for glucose-6-phosphate dehydrogenase deficiency closer to the patient. Am J Trop Med Hyg 100(1):213-221.
Palmer, K. J., S. M. Holliday, and R. N. Brogden. 1993. Mefloquine: A review of its antimalarial activity, pharmacokinetic properties and therapeutic efficacy. Drugs 45(3):430-475.
Parhizgar, A. R., and A. Tahghighi. 2017. Introducing new antimalarial analogues of chloroquine and amodiaquine: A narrative review. Iran J Med Sci 42(2):115-128.
Pergallo, M. S. 2001. The Italian Army standpoint on malaria chemoprophylaxis. Med Trop 61:59-62.
Peters, A. L., and C. J. Van Noorden. 2009. Glucose-6-phosphate dehydrogenase deficiency and malaria: Cytochemical detection of heterozygous G6PD deficiency in women. J Histochem Cytochem 57(11):1003-1011.
Phillips, M. A., and R. B. Kass. 1996. User acceptability patterns for mefloquine and doxycycline malaria chemoprophylaxis. J Travel Med 3:40-45.
Pietrzak, R. H., D. C. Johnson, M. B. Goldstein, J. C. Malley, and S. M. Southwick. 2009. Perceived stigma and barriers to mental health care utilization among OEF-OIF veterans. Psychiatr Serv 60(8):1118-1122.
Raczniak, G. A., M. S. Riddle, and M. Forgione. 2019. U.S. military deployments. In Chapter 9, Travel for work & other reasons, Yellow book. Atlanta, GA: Centers for Disease Control and Prevention. https://wwwnc.cdc.gov/travel/yellowbook/2020/travel-for-work-other-reasons/us-military-deployments (accessed November 12, 2019).
RAND Corporation. 2000. Review of the scientific literature as it pertains to Gulf War illnesses. Volume 8: Pesticides. Santa Monica, CA: RAND Corporation.
Richter, J., G. Franken, M. C. Holtfreter, S. Walter, A. Labisch, and H. Mehlhorn. 2016. Clinical implications of a gradual dormancy concept in malaria. Parasitol Res 115(6):2139-2148.
Ridley, R. G., A. Dorn, S. R. Vippagunta, and J. L. Vennerstrom. 1997. Haematin (haem) polymerization and its inhibition by quinoline antimalarials. Ann Trop Med Parasitol 91(5):559-566.
Ropers, G., M. Du Ry van Beest Holle, O. Wichmann, L. Kappelmayer, U. Stüben, C. Schönfeld, and K. Stark. 2008. Determinants of malaria prophylaxis among German travelers to Kenya, Senegal, and Thailand. J Travel Med 15(3):162-171.
Rudland, S., M. Little, P. Kemp, A. Miller, and J. Hodge. 1996. The enemy within: Diarrheal rates among British and Australian troops in Iraq. Mil Med 161(12):728-731.
Sánchez, J. L., R. F. DeFraites, T. W. Sharp, and R. K. Hanson. 1993. Mefloquine or doxycycline prophylaxis in U.S. troops in Somalia. Lancet 341(8851):1021-1022.
Saunders, D. L., E. Garges, J. E. Manning, K. Bennett, S. Schaffer, A. J. Kosmowski, and A. J. Magill. 2015. Safety, tolerability, and compliance with long-term antimalarial chemoprophylaxis in American soldiers in Afghanistan. Am J Trop Med Hyg 93(3):584-590.
Schlagenhauf, P., M. Adamcova, L. Regep, M. T. Schaerer, and H. G. Rhein. 2010. The position of mefloquine as a 21st century malaria chemoprophylaxis. Malar J 9(357).
Schlagenhauf, P., M. E. Wilson, E. Petersen, A. McCarthy, L. H. Chen, J. S. Keystone., P. E. Kozarsky, B. A. Connor, H. D. Nothdurft, M. Mendelson, and K. Leder. 2019. Malaria chemoprophylaxis. Travel Med (15):145-167.
Schwartz, E. 2012. Prophylaxis of malaria. Mediterr J Hematol Infect Dis 4(1):e2012045.
Schwartz, E., I. Potasman, M. Rotenberg, S. Almog, and S. Sadetzki. 2001. Serious adverse events of mefloquine in relation to blood level and gender. Am J Trop Med Hyg 65(3):189-192.
Srivastava, I. K., and A. B. Vaidya. 1999. A mechanism for the synergistic antimalarial action of atovaquone and proguanil. Antimicrob Agents Chemother 43(6):1334-1339.
Steenkamp, M. M., W. E. Schlenger, N. Corry, C. Henn-Haase, M. Qian, M. Li, D. Horesh, K. I. Karstoft, C. Williams, C. L. Ho, A. Shalev, R. Kulka, and C. Marmar. 2017. Predictors of PTSD 40 years after combat: Findings from the National Vietnam Veterans Longitudinal Study. Depres Anxiety 34(8):711-722.
Sullivan, D. J., Jr., H. Matile, R. G. Ridley, and D. E. Goldberg. 1998. A common mechanism for blockade of heme polymerization by antimalarial quinolines. J Biol Chem 273(47):31103-31107.
Tan, K. R., A. J. Magill, M. E. Parise, P. M. Arguin, and Centers for Disease Control and Prevention. 2011. Doxycycline for malaria chemoprophylaxis and treatment: Report from the CDC expert meeting on malaria chemoprophylaxis. Am J Tropl Med Hyg 84(4):517-531.
Thomas, M. M., I. Harpaz-Rotem, J. Tsai, S. M. Southwick, and R. H. Pietrzak. 2017. Mental and physical health conditions in U.S. combat veterans: Results from the National Health and Resilience in Veterans Study. Prim Care Companion CNS Disord 19(3).
Thornton, S. A., S. S. Sherman, T. Farkas, W. Zhong, P. Torres, and X. Jiang. 2005. Gastroenteritis in US Marines during Operation Iraqi Freedom. Clin Infect Dis 40(4):519-525.
Unwin, C., N. Blatchley, W. Coker, S. Ferry, M. Hotopf, L. Hull, K. Ismail, I. Palmer, A. David, and S. Wessely. 1999. Health of UK servicemen who served in Persian Gulf War. Lancet 353(9148):169-178.
VA (Department of Veterans Affairs). 1993. Unstructured information for: Post-operations Desert Shield/Desert Storm. https://gulflink.health.mil/va/va_refs/n46en031/osd.html (accessed November 5, 2019).
VA. 2011. Caring for Gulf War I veterans. Veterans Health Administration. http://www.publichealth.va.gov/docs/vhi/caring-for-gulf-war-veterans-vhi.pdf (accessed October 20, 2015).
Vennerstrom, J. L., E. O. Nuzum, R. E. Miller, A. Dorn, L. Gerena, P. A. Dande, W. Y. Ellis, R. G. Ridley, and W. K. Milhous.1999. 8-Aminoquinolines active against blood stage Plasmodium falciparum in vitro inhibit hematin polymerization. Antimicrob Agents Chemother 43(3):598-602.
Vogt, D. S., A. P. Pless, L. A. King, and D. W. King. 2005. Deployment stressors, gender, and mental health outcomes among Gulf War I veterans. J Trauma Stress 18(2):115-127.
Wallace, M. R., T. W. Sharp, B. Smoak, C. Iriye, P. Rozmajzl, S. A. Thornton, R. Batchelor, A. J. Magill, H. O. Lobel, C. F. Longer, and J. P. Burans. 1996. Malaria among United States troops in Somalia. Am J Med 100(1):49-55.
WHO (World Health Organization). 2014. Severe malaria. Trop Med Int Health 19(Suppl 1):7-131.
WHO. 2016a. Testing for G6PD deficiency for safe use of primaquine in radical cure of P. vivax and P. ovale. Policy brief, October 2016. https://www.who.int/malaria/publications/atoz/g6pdtesting-pq-radical-cure-vivax/en (accessed September 27, 2019).
WHO. 2016b. Fact sheet: World malaria 2016. https://www.who.int/malaria/media/world-malariareport-2016/en (accessed January 12, 2019).
WHO. 2019. World Malaria Report, 2019. https://www.who.int/publications-detail/world-malaria-report-2019 (accessed December 10, 2019).
Wiesen, A. 2019. Overview of DoD antimalarial use policies. Presentation to the Committee to Review Long-Term Health Effects of Antimalarial Drugs on January 28, 2019.
Wolfe, J., E. J. Sharkansky, J. P. Read, R. Dawson, J. A. Martin, and P. C. Ouimette. 1998. Sexual harassment and assault as predictors of PTSD symptomatology among U.S. female Persian Gulf War military personnel. J Interpers Violence 13(1):40-57.
Wong, W., X. C. Bai, B. E. Sleebs, T. Triglia, A. Brown, J. K. Thompson, et al. 2017. The antimalarial mefloquine targets the Plasmodium falciparum 80S ribosome to inhibit protein synthesis. Nat Microbiol 2(17031).
































