Session 1: What Do We Know?
During this workshop session, speakers and participants were asked to consider the information that is already known about the pace of permafrost thaw and where it is most prevalent, what can be expected to emerge from the thawed permafrost (e.g., animals, corpses, ancient pathogens), and the existing research efforts to help determine threats. Speakers shared information about the microbial ecology of permafrost and ice environments and their sensitivity to climatic changes as well as the viability of microorganisms in these environments.
Viruses in Permafrost
Dr. Jean-Michel Claverie, Aix-Marseille University & CNRS Mediterranean Institute of Microbiology, provided insights on viruses in permafrost. Dr. Claverie reemphasized that 24 percent of the northern hemisphere contains permafrost and most of this is very old and contains ancient frozen materials. The Arctic is warming faster than the rest of the world and leads to widespread permafrost thaw (Tollefson, 2017). This thaw may lead to additional natural consequences such as extremely large holes in the ground where soil is sinking and may even trigger spontaneous explosions.1 Dr. Claverie also pointed out that warmer temperatures and open shipping lanes have made large industrial ventures and resource mining viable in the Arctic region, which further disrupts permafrost and associated bacteria and microbes.
Dr. Claverie discussed the potential for permafrost thaw and changes to the active layer to enable the return of anthrax outbreaks, traces of smallpox virus, and other pathogens from people and materials buried in the permafrost (Theves et al., 2014). One example is the regeneration of 30,000-year-old plants that were buried in Siberian permafrost (Yashina et al., 2012). Given that plants can be revived from the permafrost, Dr. Claverie challenged participants to consider if viruses could be revived as well. He discussed work at the Stanchikovsky Yar exposure in Chersky, Russia and the ancient permafrost that is exposed and can be accessed without digging through more recent materials. In sampling this material, it can be tested for acanthamoeba-killing viruses through a process that subjects the sample to antibiotic-adapted acanthamoeba cultures, seeks cell lysis (or breakdown of the cell), isolates the agent, and characterizes the infectious cycle, genomics, transcriptomics, and proteomics. These viruses can survive over long time periods in permafrost. Two new, previously unknown viruses were isolated from a single sample from this process (Legendre et al., 2014, 2015).
Dr. Claverie highlighted another example where a research team was given access to a skin and hair sample from frozen mammoth remains recovered from the Yana Rhinoceros Horn Site, Siberia (Pitulko et al., 2004). The sample revealed four new
___________________
1 See for example: https://www.nature.com/news/mysterious-siberian-crater-attributed-tomethane-1.15649.
viruses: (1) ancient Megavirus, (2) ancient Pandoravirus, (3) ancient Pithovirus, and (4) Asfar-like virus. Another example involving metagenomic2 sequencing of cores from the Yakutia region, revealed a large diversity of bacteria as well as β-lactamas genes, a potential threat related to bacterial resistance to antibiotics. These genes could potentially be transmitted to modern bacteria as a new way of resisting antibiotics. Most of the viruses detected were already known and expected, but other viruses, less well known and in smaller quantities, were detected as well. A final example shared by Dr. Claverie illustrates the new, viable viruses discovered from Kolyma river exposures and thawing permafrost in the region near Duvanny Yar and Chersky, in addition to three new viruses discovered from the Lena river, one new virus from the thawed material, and four new viruses from the Pleistocene (or >11,700 years ago). He noted that these discoveries provide independent confirmation that some DNA viruses can remain infectious since the end of the last glacial period.
A key issue that arises from the types of discoveries shared by Dr. Claverie is the need to ensure that the samples (and associated data) are not contaminated by modern materials. Understanding the possibility of a real threat from pathogens in permafrost is crucial, especially when the topic receives a great deal of media attention. Confirmation of the results can be achieved through different sample sites, sample types, and investigation methods. Dr. Claverie concluded his remarks by emphasizing that hazards from thawing permafrost depend on the presence, viability, and resilience of known and unknown viruses pathogenic for humans, animals, and plants. However, exposure will grow due to increased human presence in the warming Arctic and potentially through increased permafrost disruption. Therefore, it could be useful for large-scale Arctic industrial projects to include on-site medical surveillance as well as on-site quarantine or isolation facilities and protocols. He also suggested that research on pathogenic permafrost microbes including protozoa is important to move forward, yet it can be extremely difficult to export potentially infectious samples out of the region. This highlights the importance of international research partnerships.
2016 Anthrax Outbreak
A deeper look at the 2016 anthrax outbreak in the Yamal peninsula was provided by Dr. Alexander Volkovitskiy, Russian Academy of Sciences and Yamal Expedition. The peninsula is the site for several gas extraction facilities, including sites at Bovanenkovo and Sabetta, but it is also the world’s largest center of reindeer pastoralism. This is the traditional way of life for the indigenous people of Yamal. The Nenets tundra nomads work to preserve their traditional practices and the model of family migration through a synthesis of traditions and modern innovations. Approximately 6000 people live in the tundra and depend on fishing and reindeer
___________________
2 “Metagenomics is the study of a collection of genetic material (genomes) from a mixed community of organisms. Metagenomics usually refers to the study of microbial communities.” Source: https://www.genome.gov/genetics-glossary/Metagenomics.
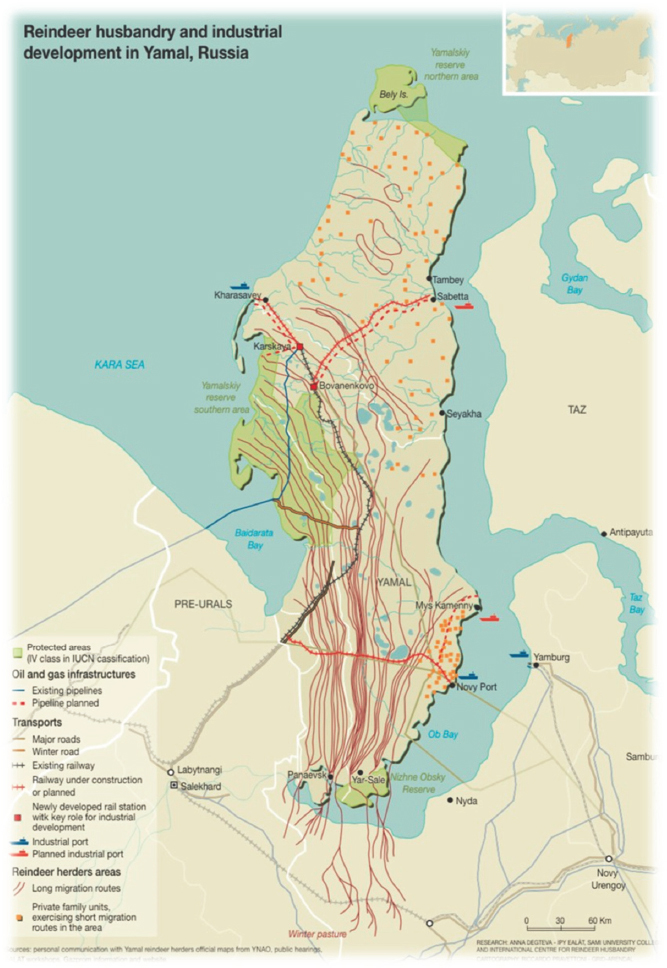
herding. They graze roughly 300,000 reindeer, about 90 percent of which are privately owned, and the other 10 percent are municipal property. Estimates of the actual number of reindeer and migration patterns are made because accurate accounting of reindeer data and tracking does not exist. As noted, these traditional practices occur within the context of intensive industrial development (Figure 7; Degteva and Nellemann, 2013).
Three groups of reindeer herders are divided into southern, middle, and
northern zones, excluding the northern most part of Yamal. Dr. Volkovitskiy provided historical background and noted that the first recorded suspected outbreak of anthrax in the Urals region was discovered in 1848 and led to the total loss of reindeer herds in the region. Indigenous community members consumed the reindeer and became sick as well. Another suspected outbreak was recorded in 1876, closer to the Yamal region, and in 1911, anthrax officially reached the Yamal peninsula. This was the first attempt by regional officials to identify the disease. Since then, there have been several documented cases of anthrax outbreaks. Vaccination campaigns were established in 1930 and the last case of officially recorded anthrax in Yamal was reported in 1941. By the 1960s, 82.5 percent of potentially receptive reindeer were vaccinated and thus mass vaccination was stopped in 2007. However, many parts of the migration routes and reindeer herding corridors pass directly through the known anthrax areas of the past. In the summer of 2016, the air temperature and the dynamics of temperature in the soil, combined with the lack of mass vaccination, allowed anthrax to emerge. Temperatures in July are most often studied, but it is important to note that the temperatures in June, as well as July, are key for understanding the processes of active layer thawing and potential release of anthrax.
Dr. Volkovitskiy noted that 2,649 reindeer perished and 36 cases of anthrax were diagnosed in people because of the 2016 anthrax outbreak. Since then, mass vaccination has resumed and discussions are ongoing with respect to the conditions of pastures and the problem of overgrazing. According to Dr. Volkovitskiy, there is unsupported blame placed on the indigenous people in the region for the anthrax outbreak coupled with resistance within the herding community to vaccinate themselves and the reindeer.
Panel on Ecosystem Changes and Microbial Threats in the Environment
Dr. Birgitta Evengård, Umeå University, moderated a panel discussion on ecosystem changes and microbial threats that are known in the environment. The first speaker, Dr. Thomas A. Douglas, of the U.S. Army Cold Regions Research and Engineering Laboratory, provided additional information about permafrost and illustrated an example research site in Utqiaġvik, Alaska where the seasonally thawed “active layer” can be rich in peat. He noted there may be a potential role of peat carbon in the biological systems of interest to the workshop. In areas of mixed forest where disturbances can occur, or wetlands where water has a near constant presence, the depth of the active layer increases, providing some protection against thaw (Douglas et al., 2020). Dr. Douglas also highlighted the dynamic nature of the “transition zone” between the active layer and the frozen permafrost below, which may be of particular interest to those researching emerging materials from the permafrost (Figure 8). This zone changes throughout the seasonal cycles of freeze and thaw and may contribute to changing soil conditions, microbes, or biogeochemical processes. As noted earlier in the

workshop, rapid thermokarst3 landscape events can happen on the order of days to weeks and often involve water and slumps or cavities in the ground. He also highlighted ongoing and potential future work at the Permafrost Tunnel facility in Fairbanks, AK.
The second panelist, Dr. Emily Jenkins, University of Saskatchewan, discussed microbial threats at the interface of humans, animals, and the Arctic environment. She approached her remarks through the context of microbial risk assessment, including hazard identification, hazard characterization (or pathogenicity), exposure assessment, and risk characterization. Risk management and communication are also important, as is the inclusion of indigenous communities in associated research efforts. Dr. Jenkins provided several examples of known zoonoses in the Arctic, including the emerging prions pathogen that has only been recognized in recent years. Challenges in terms of hazard identification include gaps in surveillance, especially of wildlife, that limit the ability to determine which pathogens are circulating; lack of information about which pathogens have been there in the past; the yearly reintroduction of seasonal migratory wildlife; and other dynamics that contribute to these difficulties. Dr. Jenkins highlighted zoonoses that are particularly likely to survive for years in this environment: spore--
___________________
3 Thermokarst refers to the process by which characteristic landforms result from the thawing of ice-rich permafrost or the melting of massive ice. Source: https://nsidc.org/cryosphere/glossary/term/thermokarst.
forming bacteria, Mycobacterium species, protozoan cysts/oocysts, some helminth eggs, prions, non-enveloped viruses, and pox viruses. On the other hand, many types of vectorborne disease pathogens are not resistant to the Arctic environment and are unlikely to survive for long periods of time in these regions. Similarly, obligate intracellular pathogens and many viruses are dependent on host cell infrastructure and may not be resistant to this environment.
In the Canadian Arctic, bison are a source of anthrax, Mycobacterium bovis, and Brucella abortus, introduced by domestic livestock and now established in wild populations. In Saskatchewan, chronic wasting disease-infected deer are located at the fringes of the boreal caribou habitat (Figure 9), and this can be a serious challenge from a food security perspective for subsistence populations. Another example is a parasite,
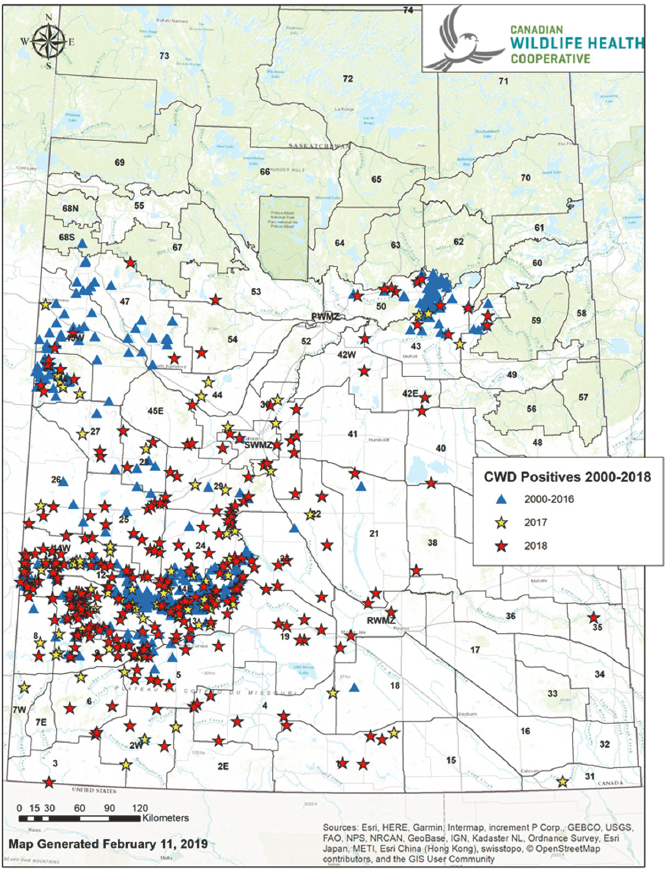
Echinococcus, of Arctic fox that can survive for years in cold environments of the north. Dr. Jenkins highlighted the need to understand actual microhabitat conditions, the probability of survival (including environmental stages and stages inside the carcasses), and the probability of transmission (including host specificity, transmission routes, and minimum infectious doses). She noted that gaps in exposure assessment include short time spans in empirical studies, lack of information on how to preserve microbes, and differences between the freeze tolerances of Arctic-adapted strains compared to temperate and lab strains.
The final panel speaker, Dr. Aleksandr Sokolov, Russian Academy of Sciences, discussed terrestrial ecosystems specifically in Yamal. He highlighted three major drivers of change in the Arctic including climate change, industrial development, and herbivory grazing, all of which are occurring in Yamal. In addition, studies in Yamal allow for research into all three of these drivers in different types of geography or subzones of tundra. Logistically, it can be easier to travel to remote areas of Yamal due to the presence of a railway system and an airport, resources that are not common in other high latitude regions. Ongoing support for research stations allows for the collection of long-term data and the study of biodiversity and abundance of birds, mammals, fish, plants, and other organisms. Another key aspect of scientific research in this region involves the local communities, and he stressed the importance of interacting with the people in meaningful and robust ways to ensure true involvement in the work happening there.
Dr. Sokolov discussed the liquefied natural gas project in Sabetta above the Arctic Circle and highlighted it as an example of the logistical challenges associated with bringing thousands of people into the area. A study of the rodent and fox abundance in the area showed significant changes year to year, including a drop in both populations in 2016, followed by a rebound of the foxes but not the rodents (Figure 10). This could be related to human interventions such as providing food for the foxes, but the key
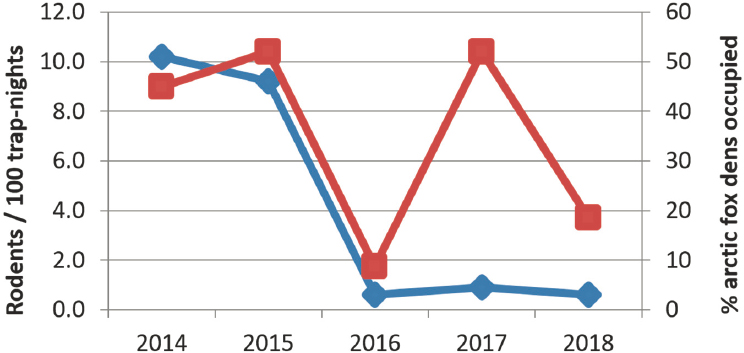
message is that there will be associated changes throughout the ecosystem. He also illustrated the scale of migration patterns of birds that originate in Yamal and the global reach that these migrations can have. As additional examples, he highlighted the increased northern range of the Gyrfalcon, assisted by human-built railroad infrastructure where this species has increasingly built their nests. Also, increased numbers of walrus observed in Yamal, where they were seldom seen before, means increasing changes to the ecosystem.
Environmental and Climatic Determinants of Infectious Disease
Exposures to a number of climate change-related events (e.g., severe weather, air pollution, water contamination, changes in vector ecology, environmental degradation, rising sea levels, food supply and safety, and others) can lead to selected health risks (Semenza and Ebi, 2019). Dr. Jan Semenza, European Centre for Disease Prevention and Control (ECDC), outlined the climate impacts on two infectious diseases, Chikungunya and Dengue. Europe has experienced increased outbreaks of these tropical diseases in the past decade (Lillepold et al., 2019), and researchers have begun to explore any potential links between climate change and the emergence or recurrence of these diseases. In looking at the climatic suitability for these outbreaks, the vectoral capacity (or the temperature-dependent climatic suitability) for transmission has increased in Europe since the 1970s, and the countries of the European Union may be considered a “hot spot” for the emergence of communicable disease (Suk and Semenza, 2011). Given this context, Dr. Semenza posed a number of questions: Is there a cause and effect relationship between the climatic suitability and the reoccurrence of the epidemics in Europe? What are the infectious disease threat events in Europe? What are the underlying drivers, determinants, and risk factors for these epidemic events? Is it possible to monitor upstream drivers of epidemics throughout Europe to potentially intervene in epidemic events and prevent them from happening? Can epidemic events that occur on a regular basis be predicted? Although it is extremely challenging to predict highly unlikely but catastrophic “black swan” events (e.g., HIV, SARS, MERS, etc.), perhaps improved monitoring of epidemic events will allow for more accurate prediction.
A “foresight study” outlined by Dr. Semenza attempted to anticipate future threats of infectious diseases in Europe (Suk and Semenza, 2011). This involves consideration of changes in disease drivers, which requires a systems perspective. Interactions between underlying drivers can be examined in the context of potential scenarios and subsequently into the development of public health actions to intervene against the public health threats. The study identified three main categories of drivers of epidemic events in Europe: globalization and environmental change, social and demographic change, and public health systems. Each of these categories contains sub-drivers (e.g., the globalization and environmental change categories contains sub-drivers such as climate change, travel and tourism, migration, and global trade). The drivers and sub-drivers interact, combine, and therefore precipitate specific scenarios such as shifts
in the transmission patterns of vector-borne diseases, drug-resistant bacteria, sexually transmitted infections, food-borne infections, resurgence of vaccine-preventable diseases, and more. Public health experts can help prepare for these potential scenarios and other potentially serious public health events.
In an attempt to quantify this information, a validation study was undertaken to explore epidemic intelligence data collected by the ECDC through global event monitoring that could potentially pose a threat to Europe. Dr. Semenza noted that each day, a roundtable meeting in ECDC’s Emergency Operations Centre assesses threats, official alerts, and epidemic intelligence from around the EU and the world. As part of the quantification study, threat data and information on drivers between July 2008 and December 2013 were derived from ECDC databases and reports including a Threat Tracking Tool, Communicable Disease Threat Report, Threat Assessment, and Rapid Risk Assessment as well as mission reports, scientific articles, and expert consultation (Semenza et al., 2016). A database of events with the associated determinants was
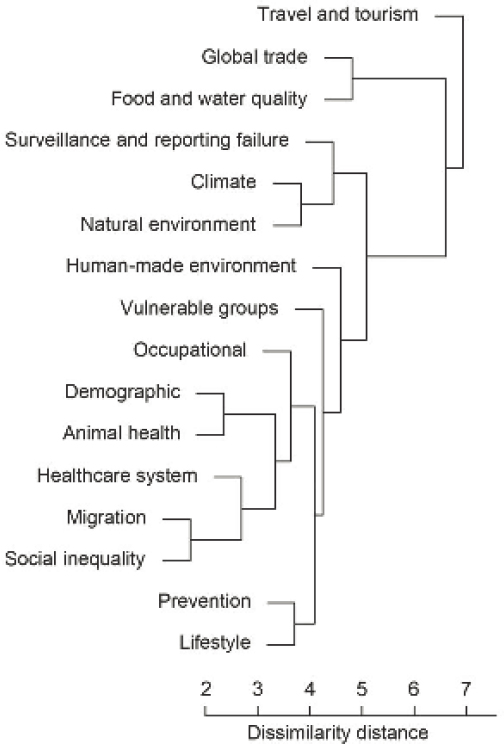
developed. A minority of these events are caused by one single driver; interactions between drivers causes the majority of events. For each disease category, the drivers can be identified and then pooled into one analysis to create a cluster dendrogram that shows how multiple drivers contribute to epidemic events. For example, climate and the natural environment drivers tend to cluster together to contribute to an epidemic event. Importantly, travel and tourism are the most significant and frequent drivers of epidemic events in Europe (Figure 11).
Restricting tourism and travel is not considered a viable option to reduce the occurrence of infectious disease events in the long-run. However, Dr. Semenza noted that the relationship between diseases, like Chikungunya and Dengue, and climatic suitability could be examined together with air passenger volume for disease active areas worldwide; air passenger volume plays a key role in the occurrence of these epidemics. In 2017, there was an outbreak of Chikungunya in France and Italy. Information on global air passenger volume was used to analyze risk zones, assess importation risk and transmission potential, as well as track the local spread using unidirectional mobility of geo-located, repeated Twitter feeds (Rocklöv et al., 2019). This analysis shows that there are specific areas within Europe that are of greater concern in terms of risk of importation, including areas where the vector is present and could transmit the virus. Further, adding the climatic suitability, the risk is higher in July and August compared to other parts of the year. Combining the climatic suitability with data derived from Twitter (mobility proximity estimates) can help determine short-distance mobility and the connections between specific risk areas with the associated spread of disease (Rocklöv et al., 2019). By focusing surveillance and control strategies in high-risk areas, this information can be part of public health practices to intervene.
Dr. Semenza also considered the contribution of climate change impacts on infectious disease burden in Europe. A study ranked infectious diseases by the strength of their link to climate change as well as the potential severity of consequence to society (Lindgren et al., 2012). This weighted risk analysis revealed that some of the diseases of greatest concern did not have surveillance systems to monitor the potential pathogens. Since the study was published, some monitoring systems have been put into place, but there are still infectious diseases that are not being monitored. An improved monitoring system could take advantage of the strong climatic links established and institute monitoring mechanisms further “upstream” in the process (i.e., before the types of surveillance that are typically done for health outcomes and after direct and indirect exposures have taken place) (Semenza et al., 2015). Dr. Semenza noted that the ECDC developed the European Environment and Epidemiology Network that aims to anticipate epidemics using a climatic or environment signal to initiate active surveillance that will help to identify the first case, initiate response activities, and therefore minimize the disease burden and public health impact (Semeza and Menne, 2009). As an example, Dr. Semenza illustrated the Baltic Sea as a suitable environment for Vibrio species, especially given increasingly warm water temperatures and low salinity (Levy, 2015). Severe health outcomes are seen in years with heat waves, due to increased
exposure to these bacteria. Early warning systems at ECDC use in situ and climatological data, remote sensing data, and models to forecast environmental suitability for vibrio growth and high-risk areas (Semenza et al., 2017). Given currently estimated climate change scenarios, this analysis predicts that cases of vibrio will increase in the Baltic region and further north into the Arctic as well.
At the conclusion of his remarks, Dr. Semenza noted that international health regulations set forth by the World Health Organization illustrate capacity in member states to advance surveillance, response, and preparedness measures for health security. Analyzing the relationship between public health capacities against infectious disease threats in Europe shows the ability of countries to respond to cross-border threat events. For example, a 10 percent increase in public health core capacities is associated with a 20 percent decrease in disease threat events (Semenza et al., 2019), and this helps to validate efforts and investments to advance public health core capacities in Europe. Returning to the earlier discussion of drivers, Dr. Semenza noted that global environmental change contributes to 61 percent of infectious disease threat events (Semenza et al., 2016), illustrating the complexity of the actual disease scenarios.
Panel on the Potential Risk of Human and Animal Exposure to Threats
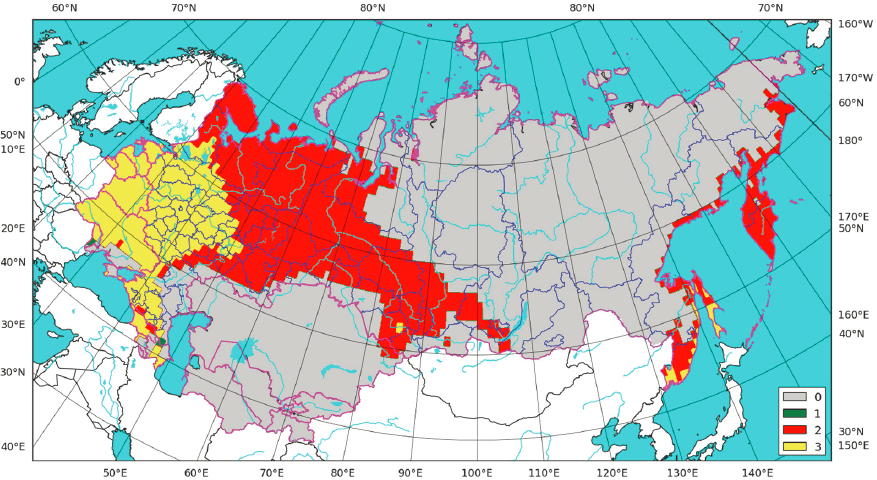
Dr. Charles Haas, Drexel University, facilitated panel remarks on potential risks associated with human and animal exposure to threats. During the panel discussion, Dr. Natalia Pshenichnaya, Central Research Institute of Epidemiology, Moscow, Russia, noted that climate change and associated increasing air and water temperature trends are taking place throughout Russia and the circumpolar Arctic region. Permafrost thaw and degradation poses risks to human health, transportation interruptions, difficulty in accessing health care, insulation, destruction of infrastructure, and crucial for this workshop, an increased risk of anthrax (Belousov et al., 2018). There is also an increased risk of release of dangerous known microorganisms (e.g., Influenza H1N1 and anthrax) from graves. Some studies have shown that viable prokaryotic and eukaryotic microorganisms can persist in permafrost from several thousand to three million years. Examples of microorganisms isolated from permafrost include aerobes (Vorobyova et al., 1997; Zvyagintsev et al., 1985); anaerobes (Rivkina, 2007); green and blue-green microalgae (Vorobyova et al., 1997); yeast (Faizutdinova et al., 2005); mycelial fungi (Kochkina et al., 2001); and viable cysts of free-living protozoa (Shatilovich et al., 2010). Some of these may be resistant to natural antibiotics, and the released bacteria can produce carbon dioxide, methane, and other gases that may lead to additional climate warming. Dr. Pshenichnaya outlined a number of infectious diseases that are climate-dependent and are of concern in the Arctic; she used examples of tick-borne encephalitis, Ae. albopictus, and Dirofilariasis in Russia to illustrate current and expected expansion of disease threat to the Arctic region. In the Ixodes ricinus (tick) example, expanded habitat changes are estimated in relation to climate warming scenarios (Figure 12; Popov, 2016), especially in the Arkhangelsk region (Balaeva et al., 2012).
Dr. Pshenichnaya closed her remarks by noting that several other factors influence infectious diseases in the Arctic. These include the occurrence and expansion of ozone holes, increased air pollution, the emergence of new species of flora, increasing duration of high temperatures, and the increasing number of heat waves. These factors could lead to additional impacts such as increased allergies, reduction of innate immunity, and stress that may increase susceptibility to infectious diseases of the respiratory tract. She suggested that adaptation measures, mitigation of climate change, monitoring and prognosis of infectious threats, and preventive measures for Northern populations may be needed.
Dr. Anne Jensen, University of Alaska Fairbanks, noted that there are sites throughout the Arctic where large numbers of people with infectious diseases have been buried, including some relatively recent gravesites. Very few people know where these, often nonobvious, mass graves are located. When these graves are being excavated by archeologists, precautions are taken, but are usually intended to keep the researchers DNA off the buried remains rather than to protect the researcher from infectious disease. Dr. Jensen remarked that other (non-burial) excavation sites might not prompt the same level of precaution, because the contents of the soil are largely unknown. For example, excavation of a site from the 1940s revealed a mummified ring
seal containing domoic acid (a neurotoxin associated with harmful algal blooms) in its intestines, which extended the known existence of harmful algal blooms back in time. During this study, one of the researchers contracted an infection that responded to antibiotics. This highlights gaps in our current diagnostics and surveillance and raises the question of protection against infectious disease at these sites. This is also an example that illustrates how a One Health approach may be warranted, where medical experts, animal health experts, social scientists, and community based researchers would likely benefit from sharing protective knowledge.
In terms of traditional knowledge, Dr. Jensen confirmed that many hunters examine their catches for diseases or infected areas, and the North Slope Department of Wildlife Management often takes samples when there appears to be something wrong with an animal. She noted that many traditional foods are prepared using fermentation. However, rather than using more traditional containers such as wood or hide, there has been an increased use of plastic containers for fermentation, potentially leading to anaerobic conditions and botulism. In addition, ice cellars are commonly used for food storage and are an important part of the culture, but could potentially become a method of exposure to harmful bacteria as permafrost warms and the temperatures in the cellars rise. Another example of potential exposure comes from permafrost slumps or collapse sites where ancient organic materials such as whale bone and other artifacts are exposed. It is possible for people to encounter these materials and increase their risk of exposure to diseases.
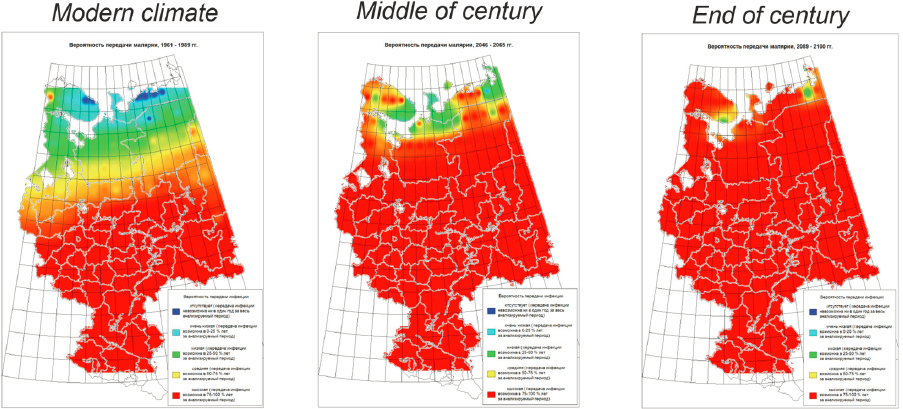
During his remarks Dr. Dmitry Orlov, Lomonosov Moscow State University, shared information about recently published medico-geographical research, which includes mapping of Russia’s natural-focal diseases, distribution of diseases, and morbidity and its dynamics (Malkhazova et al., 2019). Dr. Orlov’s work on nosogeography (the distribution of diseases) in Russia produces these types of maps of infectious disease hosts and vectors using a variety of techniques. For example, detailed maps can be produced that show areas at risk for tick-borne encephalitis throughout Russia including distribution of potential vectors. He noted that climate change and associated increases in mosquito and tick spread as well as infections transmitted by rodents are already having an impact on population health. To provide another example, Dr. Orlov shared a prognostic model of malaria risk based on climate scenarios from the Intergovernmental Panel on Climate Change (IPCC, 2000). The results illustrate an increased probability of malaria transmission in Russia by the end of the century (Figure 13). He also pointed out that West Nile has been detected in Russia relatively recently and maps have been produced to estimate potential distribution within specific regions. These mapped projections have largely been shown to be accurate. Dr. Orlov concluded his remarks by sharing a list of currently important natural focal diseases for the Russian Arctic including anthrax, tularemia, leptospiroses, opisthorchiasis, diphyllobothriases, trichinosis, echinococcosis, and toxocariasis.
Dr. Jay Butler, US Centers for Disease Control and Prevention, shared several observations on current infectious disease threats in Alaska related to higher
previous presenters, Dr. Butler noted the increased air temperatures in Alaska associated with climate warming and specifically highlighted the expanded range of Ixodes scapularis and I. pacificus in the United States (Eisen et al., 2016). Increasing northward spread of ticks has been reported anecdotally, including observations of ticks on animals as well as humans. There is some concern that previously unobserved species may be emerging in Alaska, and the State Division of Environmental Health has initiated a “submit-a-tick program”4 online as a method for sentinel surveillance for tick species previously not observed in Alaska. As another example, Dr. Butler discussed the outbreak of Vibrio parahaemolyticus in oysters that was observed in Prince William Sound following a prolonged period of water temperature above 15 degrees Celsius (McLaughlin et al., 2005). The research indicated that a general upward trend in surface water temperature has been observed in Prince William Sound and in the open ocean at Gulf of Alaska National Oceanic and Atmospheric Administration buoys. This warming likely contributed to the outbreak of foodborne disease caused by a pathogen not previously detected this far north in Alaska.
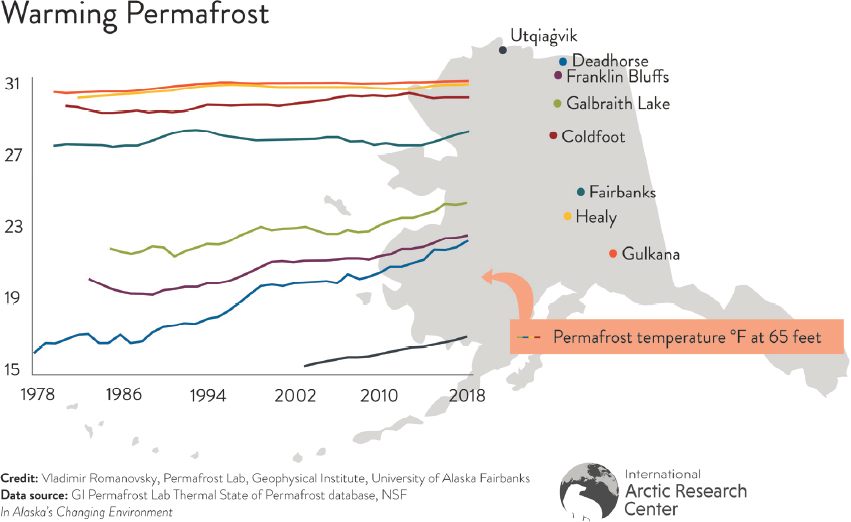
Two final examples illustrate the importance of permafrost thaw and loss of sea ice. Warming and loss of permafrost has been observed throughout Alaska (Thoman and Walsh, 2019, Figure 14), and Dr. Butler reiterated earlier concerns about food safety and thawing ice cellars, a traditional method of preserving food. Sea ice loss and an increasing number of days with open water has the potential to affect coastlines, especially during storms, which may damage crucial infrastructure such as sewage
___________________
treatment plants. Data are available that show a relationship between limited access to wash water and higher rates of respiratory tract infections and skin infections (Hennessy et al., 2008), and there is therefore some risk for these types of events to have implications for human disease. Dr. Butler also noted the increasing impact of tourism in these remote areas, and shared information about a Health Impact Assessment for the State of Alaska.5
Dr. William Bower, US Centers for Disease Control and Prevention, discussed concerns related to anthrax emergence in the Arctic. It is a naturally occurring zoonotic disease caused by Bacillus anthracis. This spore forming bacteria can survive in a temperate environment for decades, and it can presumably survive in permafrost indefinitely. It is also a pathogen of economic and public health concern as it has a high mortality rate in domesticated animals and wildlife. Humans can become secondarily infected by contact with infected animals or contaminated animal products, and there are currently an estimated 2,000 to 20,000 cases per year worldwide. Herbivores ingest spores when grazing or drinking contaminated water. Infection results in rapid death with a large amount of bacteria circulating in the blood stream at the time of death. The bacteria can be shed in blood and discharges, or openings in the carcasses that expose the bacteria to the environment. The bacteria shed into the environment form spores.
___________________
5 See http://epibulletins.dhss.alaska.gov/Document/Display?DocumentId=1962.
Certain environmental conditions, such as a high soil pH, and high calcium, moisture, and high organic matter content are more favorable for long-term spore survival. Spores can be found in the vegetation where they are ingested by grazing animals, starting the cycle again. There is some evidence to suggest that insects may serve as mechanical vectors as well.
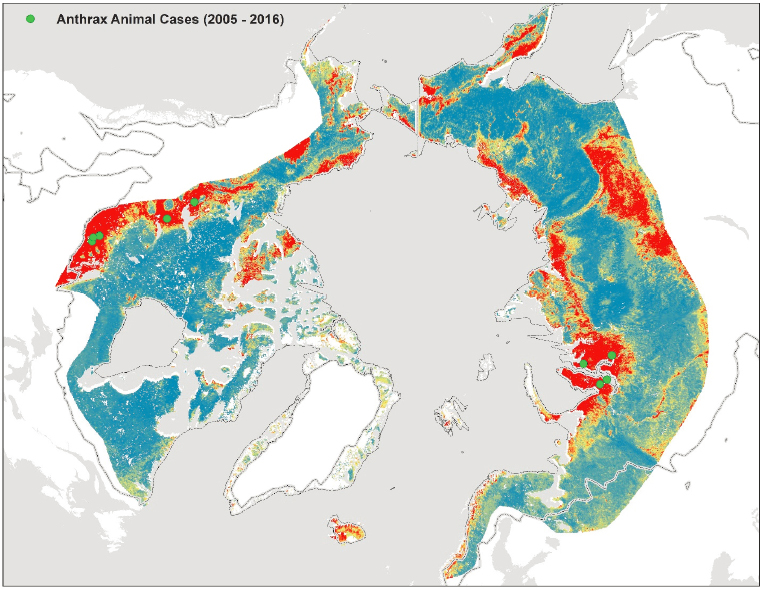
Dr. Bower illustrated the potential risk of anthrax in the Arctic region based on ecological niche modeling. Ecological niche modeling is the process of using computer algorithms to predict the geographic distribution of pathogens based on ecologic and environment variables. Variables used to create these models include environmental measurements (e.g., temperature and precipitation), ecological inputs (e.g., soil characteristics), and locations of previous outbreaks. These models can reveal areas where anthrax is more likely to occur. For example, combining the predicted areas suitable for anthrax spore survival with the distribution of permafrost and previous anthrax cases reveals areas where ecologic conditions are most suitable for anthrax and thus where anthrax is most likely to persist or reemerge in the Arctic region (Figure 15). Access to this information can increase the ability to prepare for anthrax reemergence. Capacity building is an important part of developing surveillance and detection capabilities in these areas. This likely involves input from both veterinary and human health-care providers as well as appropriate laboratory support and efficient reporting
mechanisms at regional, local, and national levels. Examples of effective local capacity include the ability to detect and diagnose potential cases, and capacity at the regional level may consist of providing surge capacity to support outbreak response and confirmatory diagnostic testing. At the national level, an example of effective capacity is the formulation of policies to encourage a One Health approach. Dr. Bower noted that this might include allocation of resources to support prevention and control through vaccination programs, carcass disposal and quarantine; providing technical support; and reporting mechanisms to international authorities.
This page intentionally left blank.


















