4
Vaccine Distribution and Delivery
Following the Chapter 3 review of issues related to maintaining the supply chains needed to provide the critical components, this chapter turns to the logistical challenges involved in the worldwide distribution of finished vaccines. Those challenges include country preparedness, vaccine characteristics, data systems, and the physical supply chain required for various vaccine types.
Given the annual need to update a seasonal influenza vaccine and the limited period of use, poorly managed logistics systems can disrupt vaccination campaigns and lead to inequitable vaccine distribution. Some U.S. regions receive more vaccines than needed while others receive an insufficient supply, both of which can increase the costs of a vaccination program (Li et al., 2018). Data are critical to establishing and maintaining a well-managed logistics system, but the currently available tools to gather data and assess the global supply of influenza vaccine fall short of providing those data. In the United States, few states have systems in place that provide the necessary visibility into how many doses of vaccine have been administered, how much inventory is left, and where it needs replenishing. The same is true globally, where gaps in countrywide assessments create the same problem regarding visibility. In this chapter the committee discusses how vaccine characteristics and design can affect distribution, the need for diversified portfolios of influenza vaccines, gaps in obtaining supply and demand data, and vaccine information systems and coordination.
Table 4-1 summarizes the recommendations in this chapter, delineated by the U.S. and global or regional actors identified for their implementation.
TABLE 4-1 Summary of Recommendations on Vaccine Distribution and Delivery
| Global/Regional Actor | Recommendation | Domestic Actor(s) |
|---|---|---|
|
Recommendation 4-1: Systems approach to vaccine design and development |
U.S. Department of Health and Human Services, including
|
|
Recommendation 4-2: Global vaccine portfolio rollout | |
|
Recommendation 4-3: Commission studies in forecasting and demand uptake |
|
| Recommendation 4-4: Improved models for vaccine cost effectiveness |
|
|
|
Recommendation 4-5: Global tools for vaccination planning |
|
DESIGN FOR DISTRIBUTION
Looking to the future and the likely development of new types of vaccines for seasonal and pandemic influenza, it is important to understand that the characteristics of a vaccine have implications for its distribution, acceptance, and appropriate use, particularly in low-resource settings. Vaccines may have different requirements for cold chain logistics, vial size, shelf life, dose volume and regimen, route of administration (i.e., injection, oral), among others. For example, a vaccine that requires sub-zero cold chain temperatures (less than 0°C) along with various ancillary supplies will place greater stains on supply chains than a vaccine that can be stored at standard refrigeration temperatures (2°–8°C) or can be administered with fewer ancillary supplies.
These considerations have been underscored by the mRNA vaccines developed against SARS-CoV-2. While highly efficacious, the ultra-cold temperature requirement is a significant barrier to administration in limited--
resource contexts (Crommelin et al., 2021). Manufacturers need encouragement to do stability studies to reduce the need for ultra-cold chains and to improve vaccine stability outside of cold chains. However, there are barriers to conducting such studies: since it is in the best interest of manufacturers to have vaccines in the market as quickly as possible, nonessential research, such as stability improvement studies, are not prioritized. A complex array of considerations need to be accounted for when encouraging manufacturers to improve existing vaccine platforms.
Other specifications have led to a more complicated rollout for some vaccines. For example, the Pfizer mRNA vaccine has a set of unique specifications that require additional steps in administration. While the industry standard for a vaccine dose is 0.5 mL, the Pfizer vaccine dose is 0.3 mL. This vaccine also requires ultra-cold storage for transportation, along with dilution before administration, which is another nonstandard practice (Pharmacy Today, 2021) that presents challenges in administration. Timing between shots has also caused some confusion, as the recommended interval between doses is 21 days for the Pfizer vaccine and 28 days for the Moderna vaccine (CDC, 2021c). In contrast to these two, the Janssen vaccine from Johnson & Johnson requires only one dose. Though a one-dose regimen provides significant advantages in reduced needs for supplies, storage, and transportation, this vaccine has shown somewhat lower efficacy than the other two.
The fact that different vaccines have varying efficacies can lead to confusion and the perception that some vaccines are superior to others (Harvard University, 2021). The development of multiple COVID-19 vaccine candidates demonstrates the tradeoffs between vaccines with high efficacy and those that can be distributed easily and quickly. Though no currently available influenza vaccines require ultra-cold supply chains, these barriers must be considered in the event that mRNA technology is someday used for influenza vaccines. There is a tradeoff between vaccine design and speed and the efficiency of distribution, delivery, and administration. Manufacturers need to consider mass-scale distribution (especially in low-resources settings) and reverse logistics when developing vaccines, accounting for end-to-end supply chain needs. Desirable vaccine characteristics for distribution, delivery, and administration need to be considered by vaccine manufacturers and public health officials.
At a minimum, vaccines must meet criteria for quality, safety, and efficacy (Dellepiane and Wood, 2015). Though available influenza vaccines meet these standards, the World Health Organization (WHO) recognizes the need for improved vaccines that provide longer-lasting protection across a variety of strains (Neuzil et al., 2017). Many low- and middle-income countries (LMICs) do not have national seasonal influenza vaccination programs, in part due to the challenges associated with current vaccines,
which need to be tailored each year to match the anticipated circulating strains, are generally of moderate efficacy against influenza, and provide limited duration of protection. Improved influenza vaccines that provide broader and longer protection against severe illness endpoints are needed to address public health needs in LMIC settings. Even in the United States, for the 2009–2019 period, effectiveness for seasonal vaccines ranged only from 19 to 60 percent (CDC, 2020a). The committee recognizes that higher vaccine efficacy could increase confidence in influenza vaccines and lead to greater integration into immunization schedules.
Beyond vaccine quality and efficacy, there is a need to develop vaccines that account for global logistical constraints. To encourage innovation in influenza vaccine development to address these needs, WHO has developed preferred product characteristics1 for next generation influenza vaccines (WHO, 2017). These characteristics describe preferred parameters of vaccines that balance efficacy with ease of administration and delivery and result in vaccine formulations for both high-income countries and LMICs. WHO has also published guidance on the programmatic suitability of vaccine candidates, accounting for such considerations as dose volume, dose number, and administration device (WHO, 2012). Improved vaccines should allow for needle-free administration and greater thermostability to reduce requirements on supply chains (Neuzil et al., 2017). The existing WHO prequalification mechanism (see Chapter 3) also contains a description of “critical” criteria that, although not required, are “recommended to increase feasibility of administration” (Ortiz and Neuzil, 2019, p. S101). The WHO criteria state that vaccines should be compatible with existing vaccine vial monitors, packaged with materials that can be disposed of at the administration sites, and “have auto-disable needles if prefilled in syringes” (Ortiz and Neuzil, 2019). Until more broadly effective vaccines are available, certain product characteristics can be achieved in the short term to facilitate global access to influenza vaccines (Neuzil et al., 2017).
Target Product Profiles and Operational Considerations
Target product profiles (TPPs) established by WHO are intended to stimulate improvements to existing vaccines or the development of new products to further expand programmatic suitability. TPPs are documents that contain descriptions of desirable features for future prequalified vac-
___________________
1 WHO’s preferred product characteristics are similar to the target product profiles that the pharmaceutical industry develops, except they are tailored to the public health perspective. Industry target product profiles “specify minimally acceptable criteria in addition to preferred criteria” (WHO, 2017, p. 4).
cines and are targeted at manufacturers and stakeholders to identify needed areas of improvement for vaccines (WHO, 2021b). TPPs are non-obligatory and intended as guidance, but they need to incentivize manufacturers by guiding the development of a more successful product (WHO, 2021a). Considerations for program requirements need to be addressed early in vaccine development stages to ensure suitability for LMICs (Zehrung et al., 2017).
In addition to TPPs, tools are available to identify operational considerations, which are assessed on an implementation level. Operational considerations are intended to inform country offices and immunization program partners of factors that affect vaccine rollout, which are separate from the characteristics of the vaccine itself. In the United States, the Centers for Disease Control and Prevention (CDC) established a series of operational considerations for continuing routine immunizations in LMICs during the COVID-19 pandemic (CDC, 2021d). These include recommendations for health care worker engagement with communities and considerations for setting up vaccination sites that reduce risk of SARS-CoV-2 transmission. They also advocate for “catch-up vaccinations”—special efforts to roll out routine immunizations that have been disrupted due to the COVID-19 pandemic. These operational considerations acknowledge that communities with higher risk of vaccine-preventable disease outbreak, such as those experiencing low vaccine coverage, poverty, or displacement, should be prioritized for catch-up efforts (CDC, 2021d). By accounting for local contexts and logistical features, operational considerations can contribute to equitable vaccine rollouts. Operational and programmatic characteristics need to be given higher consideration and brought to the attention of manufacturers earlier in the development cycle, especially in low-resource countries, to improve uptake, which will affect forecasting and address hesitancy and can minimize the operational costs.
The committee finds that the process of consulting guidance from TPPs and operational considerations is currently disjointed; they need to be aligned for priority vaccines, such as those for seasonal and pandemic influenza. There is a need for additional focus on operational considerations in the TPPs and to upgrade minimum requirements in the TPPs with some preferred vaccine characteristics. Such in-country operational, distribution, and delivery considerations need to be included further upstream in the development process at the policy, manufacturing, and regulatory levels before a vaccine becomes available. Greater integration of operational considerations and TPPs can push manufacturers to develop vaccines that are better suited for low-resource contexts, in addition to preparing for mass vaccination and minimizing wastage. As the costs of accounting for all of these considerations is likely to be high, the adoption of a systems approach to product discovery and development, which incorporates the downstream challenges of distribution and delivery as outlined above, may have to be
incentivized. In addition, countries need to better understand how these various considerations will affect their rollout and uptake.
Vaccine Labels During Pandemics
Vaccine labeling is an approval task conducted by regulatory agencies, such as the U.S. Food and Drug Administration (FDA) in the United States, other national regulatory agencies, and WHO. In a pandemic with ongoing assessments, expiration dates may have to be updated, so it is part of a design characteristic. Countries with less robust medical regulatory systems may not understand that new knowledge will develop after labeling, particularly in areas of vaccine stability. During the COVID-19 pandemic, for example, an adenovirus-based recombinant vaccine that received an approval for emergency use was labeled with a particular expiration date based on data available at the time those vials were filled with vaccine. Over time, however, the manufacturer of this vaccine continued assessing vaccine stability, and based on the newer data, regulators extended the expiration date of the vaccine from 3 to 9 months. However, public health authorities in Sudan, the Democratic Republic of the Congo, Malawi, and perhaps other countries rejected and destroyed vials with the original expiration date on the label, even after being told about the new, extended expiration date. At least in part, their actions reflect the significant time and effort regulators have taken to ensure vaccinators look at the vial label and discard it if expired. During the unique circumstances of a pandemic, an innovative approach to labeling may be needed to prevent vaccine destruction or the need to recall for relabeling. Currently, regulatory agencies do not authorize relabeling products, but rather issue a letter that the product’s expiration date has been extended or storage requirements have changed (FDA, 2021a).
Vaccines, like all medicinal products, have an expiration date and shelf life that is determined by the manufacturer and approved by regulatory authorities (WHO Africa, 2021). During public health crises, approval for emergency use is at times granted by regulators when there is only 6 months of data available for determining an appropriate expiration date (WHO Africa, 2021). Regulators approve the drugs for emergency use with the understanding that more stability data will be forthcoming. This means some profile characteristics, such as the expiration date, will not be fully established at the time the product is being labeled for use. For example, on May 19, 2021, FDA announced additional information had been obtained from the COVID-19 vaccine manufacturer extending the time undiluted vaccine vials could be stored in the refrigerator from 5 days to up to 1 month (FDA, 2021b). This guidance superseded their previous recommendations, which had been released only a few months earlier, in
February. An extended shelf life would decrease wasted medicine, which is especially critical when a product is in short supply, as is often the case during a public health crisis. Understandably, however, frontline workers are appropriately trained to carefully review packaging labels and discard any expired drug product that, with additional data, might actually be safe and effective for an extended period.
Quick response (QR) codes are one way that manufacturers are providing more up-to-date expiration information to vaccinators rather than printing an expiration date on products (CDC, 2021b). Unlike traditional linear barcodes that require specific scanning engines to be read, QR codes can be read using a smartphone, making the information more accessible to frontline workers in low-resource environments. These codes can be placed on both primary packaging (individual vials) and secondary packaging (cases of vials) (Vander Stichele et al., 2021). The codes can be checked at a district level rather than at individual points of administration. Internet access may be a limiting factor for some areas on an implementation level, but, overall, the use of QR codes could be broadly applicable and provide a pathway to quickly update expiration dates. Despite the value and modest cost to manufacturers, few vaccines supplied to LMICs currently use the technology. While there appear to be discussions that Gavi, The Vaccine Alliance (known as Gavi), and UNICEF will require QR codes in the future to track and trace vaccines, the technology is not currently being used by the vast majority of COVID-19 vaccines (Gavi, 2019b; Vander Stichele et al., 2021). The need for multiple languages on limited label spacing and the use of different expiration date formats on labels are challenges for their adoption (Ramakanth et al., 2021). However, the committee finds that these considerations further strengthen the appeal for greater use of QR codes during public health crises. During a pandemic when there is limited initial stability data for novel vaccines, and potentially even for well-accepted vaccines (i.e., strategic national stockpile), QR codes are the best option for manufacturers to use that provide access to the latest data on vaccine stability, shelf life, and safety.
RECOMMENDATION 4-1: The U.S. Department of Health and Human Services (HHS), in partnership with its counterparts in other countries and relevant global stakeholders and funders, should ensure a systems approach to the design and development of vaccines for feasible distribution and delivery in various global contexts and support relevant innovations.
RECOMMENDATION 4-1(a): HHS, its global counterparts, and relevant global funders and stakeholders should encourage attention to operational considerations up-front when funding vaccine development.
RECOMMENDATION 4-1(b): The U.S. Food and Drug Administration (FDA), along with the World Health Organization (WHO), should encourage manufacturers to consider including WHO’s preferred characteristics in their submissions for clinical trials. The National Institutes of Health (NIH) should prioritize including these preferred characteristics in its influenza vaccine research program.
RECOMMENDATION 4-1(c): HHS offices and relevant agencies, including the Assistant Secretary for Preparedness and Response and NIH, should make sustained global investments in novel vaccine end-to-end technologies, including stabilization and delivery platforms, that will improve equitable access and adaptation for vaccines to be used in various temperature settings.
RECOMMENDATION 4-1(d): WHO and FDA, after issuance of a WHO emergency use listing procedure or an FDA Emergency Use Authorization, should require digital packaging labels during a pandemic so that changes in vaccine stability and shelf life can be immediately understood and easily accessed by end users. These labels could also serve as a verification mechanism against counterfeit vaccines.
VACCINE PORTFOLIOS
The committee notes that a variety of factors, including vaccine characteristics and physical supply chain capacity, significantly affect the feasibility and efficiency of distribution, which has implications for uptake and administration (Songane, 2018). The committee finds that a lack of capacity of (1) personnel and cold chains, (2) funded national pandemic preparedness and response plans, and (3) supplies needed for successful vaccination both internationally and in-country are significant challenges, as well as a lack of personal protective equipment, delivery devices, and waste management. These challenges also hinder equitable vaccine distribution. Certain vaccines, such as the mRNA COVID-19 vaccines, can strain a country’s vaccine distribution by increasing the need for cold chain maintenance, specific delivery devices, or repeated doses. As a result, researchers and vaccine manufacturers need to consider the effect of these issues on mass-scale distribution.
Vaccine “portfolios,” lists of priority vaccine candidates, have previously been funded by institutions that advocate for pandemic preparedness. The Coalition for Epidemic Preparedness Innovations funds 19 vaccine candidates that use different platforms, targeting a variety of diseases. As it is difficult to determine which technologies will be most successful while vaccines are in the clinical stages, funding a portfolio increases the likelihood of
identifying successful candidates (Bernasconi et al., 2020). These portfolios support the development of vaccines that cover a variety of diseases. Before COVID-19 vaccines were widely available, a study developed a model to identify optimal portfolios that allowed users to explore candidates while accounting for research and development and manufacturing timelines (McDonnell et al., 2020). Similar tools could be applied in the future to assist funders and policy makers in preparedness planning.
A key initiative, the Vaccine Innovation Prioritization Strategy (VIPS) alliance2 has been recently established to drive priority vaccine product innovations forward and address key roadblocks and gaps to innovation development and uptake. The VIPS alliance is developing a single integrated framework to evaluate and prioritize vaccine product innovations and focus on needs of LMICs, in consultation with wider stakeholders, including end users from those countries. It is crucial to take stock of the current vaccine innovations implemented in response to COVID-19 vaccines and further support end-to-end vaccine innovations covering the entire product development to simplify logistics, improve equitable access, increase the acceptability and safety of immunization, allow vaccine interchangeability, and facilitate outreach during future pandemic preparedness and response.
The committee supports the idea of a new vaccine portfolio specifically for vaccines targeting influenza. A balanced portfolio of vaccines is needed so that different vaccines can be selected for different country contexts to ensure better reach and uptake. Portfolios need to be balanced considering the tradeoff between access and affordability and the additional complexity of managing distribution, management, and administration of multiple vaccines. The tradeoff of vaccine efficacy and speed of production is another important consideration. If higher efficacy vaccines that, from a total cost perspective, are expensive to purchase, distribute, and administer are unavailable to some countries, it could create the perception that LMICs are receiving lower-quality vaccines. To counteract this perception, the committee finds it important to ensure that all vaccines in a portfolio are available to all countries that want them. In developing such a portfolio, agencies should refrain from providing normative guidance to countries about which “approved” vaccines to use.3
The committee recognizes that sources of financing for vaccines may vary. All countries need to have the opportunity to purchase any “approved” vaccine. When there is scarce global funding to buy into a portfolio, interna-
___________________
2 The alliance is comprised of the Bill & Melinda Gates Foundation, Gavi, PATH, UNICEF, and WHO. It built on an existing foundation established by WHO’s Immunization Practices Advisory Committee and the Product Development for Vaccines Advisory Committee.
3 For example, the WHO Emergency Use Listing Procedure and WHO prequalification may be used to identify vaccines that meet designated criteria.
tional agencies could use the lens of program suitability and cost efficiency to determine which vaccines to supply to which regions or countries. Global financing and distribution mechanisms can offer the full portfolio but structure a full or partial subsidy to a country for different vaccines. These mechanisms could be adjusted as platforms and vaccine characteristics evolve over time. Having country access to a full portfolio of vaccines available is an equity issue to avoid the appearance of some countries receiving subpar vaccines. This approach requires collaboration with regional structures to ensure they can support the use of vaccines. When adopting multiple vaccines, a country needs to determine its allocation strategy. Choices include allocation of vaccines by geography (based on infrastructure, scale, cold chain and other handling requirements, characteristics of the vaccine) or providing multiple options to every service delivery point. Ethical considerations must also play a role in the decision process (WHO, 2020a).
RECOMMENDATION 4-2: The World Health Organization, its partners, and its funders should facilitate a global vaccine portfolio rollout to ensure the development and access to a broad portfolio of influenza vaccines.
DEMAND FORECASTING AND GENERATION
Demand Forecasting
As governments worldwide increase their focus on initiating large-scale immunization drives to tackle influenza outbreaks, it becomes increasingly important to have tools available to forecast the demand for influenza vaccine, particularly given the long time currently needed for vaccine production. One estimate projects the influenza vaccines market to rise at a 7.7 percent compounded annual growth rate (Fortune Business Insights, 2020), but this type of projection does not provide the concrete guidance that can help manufacturers or those managing the supply chains for vaccine production to prepare for an upcoming influenza season or pandemic. Unlike childhood vaccines, whose demand is relatively simple for vaccine manufacturers to predict, seasonal influenza vaccine demand is more uncertain given the seasonality of influenza outbreaks, their changing nature from year to year, varying and uncertain vaccine uptake due to multiple factors, and vaccine spoilage and wastage. The result is that organizations that purchase vaccine are loath to order more than they think they can use in a given season, leaving open the possibility that an unexpected increase in demand by the public can cause shortages.
The challenges are different when considering pandemic influenza vaccines. During the H1N1 pandemic of 2009, demand significantly exceeded
supply as batches were released. Prioritizing populations to receive the vaccine was a critical step in allocation (Huang et al., 2017), yet identifying cohort groups based on priority was difficult even in developed nations. There is a tradeoff between risk-based prioritization policies for vaccination and efficiency. Implementing sophisticated prioritization policies is also challenging due to information gaps and cohort group identification. There is a need to better understand how prioritization policies affect demand. The committee agrees that better tools for demand forecasting are needed and that countries need technical assistance in implementing them. Identifying priority populations for vaccination will be important when addressing future pandemic scenarios to better match supply and demand.
No one factor drives or fully predicts country demand for influenza vaccine. Many factors affect demand, including price elasticity, convenience, knowledge of influenza’s impact, severity and timing of the influenza season, demographics, severity of the previous year’s season, providers’ recommendations, and perception of need (Layton et al., 2005). One group has created a model to forecast demand for maternal influenza vaccination in LMICs (Debellut et al., 2018). These investigators assessed potential demand for seasonal influenza vaccine, and the results led them to identify the ways that immunization programs may be affected by availability gaps in supply linked to current vaccine production cycles and shelf life duration. The models projected that demand for seasonal influenza vaccination in pregnant women could increase from 7.7–16.0 million doses in 2020 to 27.0–61.7 million doses in 2029. The reason for this increase is attributed to WHO efforts to expand seasonal influenza immunization in countries that had not yet adopted maternal influenza immunization plans. Although the global capacity for seasonal influenza vaccine production is projected to meet this demand, current vaccine production cycles are likely to leave gaps in vaccine availability and coverage as demand increases (Debellut et al., 2018). Demand forecasting—aggregate as well as disaggregate—is affected by many factors and remains a challenge for most countries. Factors affecting forecasts include the challenge of identifying cohorts and accessing them, operational costs, access to finance, and human behavior. There is a need for better understanding of these factors that affect forecasts and their accuracy.
Developing appropriate vaccination strategies also requires information about anticipated demand. Demand forecasting provides decision makers with data on how much vaccine is likely to be taken up in a given region, which informs vaccine ordering and development of allocation procedures. Forecasting models have been successfully implemented in LMIC supply chains to project vaccination needs. An impact assessment in Niger revealed that, when combined with increased transport frequency, use of a demand forecasting system “increased the number of successfully administered vac-
cine doses and lowered the logistics cost per dose up to 34%” (Mueller et al., 2016, p. 3663). When combined with assessments and improvements in the physical supply chain, demand forecasting can lead to tangible improvements in supply chain efficiency. Although the importance of demand forecasting is understood at a global level, few tools exist to aid countries in making these assessments. Several challenges that are unique to influenza have to be accounted for to develop accurate and timely estimates for vaccine demand, which will then inform allocation and distribution strategies.
Demand Generation
Demand generation and demand forecasting are two distinct gaps affecting vaccine supply chains. Generating demand is a critical consideration that is often neglected. Producing vaccines, that is, generating supply, is rightfully a focal point in a pandemic scenario; less focus is placed on building demand for a vaccine among the public. As seen in the COVID-19 vaccine rollout, building demand has become a priority in countries with high vaccine supply, as vaccine hesitancy is a more significant barrier than vaccine availability. In countries such as the United States, most of those who remain unvaccinated are vaccine hesitant. Vaccine hesitancy is also a concern in countries with low vaccine supply, though hesitant populations represent a smaller percentage of the unvaccinated (Evans and French, 2021).
As vaccine supplies become more available, hesitancy will likely emerge as a primary factor in influencing demand, and therefore uptake (Evans and French, 2021). When addressing vaccine hesitancy, it is important to identify what factors affect public perception. A meta-analysis of community perceptions of the Ebola vaccine in 2019 revealed that a misunderstanding of side effects and limited vaccine information led to hesitancy. Poor engagement of local leaders and low trust in health care workers also contributed to hesitancy (Carter et al., 2020). The fact that the Ebola vaccine was investigational (i.e., not fully licensed) and excluded pregnant women and children also contributed to mistrust. Proper communication is critical, and has to be developed with localized contexts to build trust (Hasan, 2019). Vaccine hesitancy will be most effectively addressed with a variety of interventions together with “communication plans, educational efforts, community engagement strategies, and vaccine delivery systems” to address both behavioral and structural drivers of low vaccine uptake (Evans and French, 2021, p. 326). These strategies address matters of not only hesitancy, but also information on how to access vaccines. Public outreach and vaccine delivery need to be developed to build confidence and trust in the vaccines while addressing structural issues, such as access to information and vaccination sites.
In order to generate demand, the root causes of low vaccination coverage need to be addressed. This has been a challenge in the United States, as demand for COVID-19 vaccines decreased after the initial months of availability to the general public. Surgo Ventures, a U.S.-based research institution, developed the COVID-19 Vaccine Coverage Index, which was designed to reveal underlying barriers to vaccination on a community level. This tool not only identified communities at risk, but also determined causal factors, such as socioeconomic characteristics, vaccine hesitancy, constrained resources, and health care inaccessibility. These results can lead to better-informed policy decisions that address the drivers of low vaccine uptake (Surgo Ventures, 2021). Similar analyses conducted in other countries, particularly LMICs, would be beneficial in revealing drivers of low vaccine coverage in different contexts.
Worldwide, vaccine hesitancy has emerged as a significant barrier to vaccine coverage. In countries such as the United States, much of the unvaccinated population is wary of the COVID-19 vaccine due to perceived risk of severe adverse events. Communication and public education interventions are important for increasing public trust, which can, in turn, result in higher uptake, and thereby increase vaccination coverage. There are multiple factors that affect demand uptake including access, hesitancy, supply availability, and prior experience with health systems. Overall, there is a need for better understanding of these factors and development of country- and culture-appropriate mechanisms to address them.
RECOMMENDATION 4-3: The Centers for Disease Control and Prevention should work with the World Health Organization, Gavi, and global counterparts to commission studies in demand forecasting and demand uptake. The National Vaccine Advisory Committee should be augmented to engage in this task.
Seasonal influenza presents a distinct challenge with demand generation, as general acceptance and uptake is low. Especially in LMICs, seasonal influenza is not considered a priority health concern, largely because of a lack of studies that assess the local burden of disease (Bresee et al., 2018) and a lack of local influenza epidemiological data (Bhan and Sinha, 2019). These lacks hinder data-driven decision making to promote influenza vaccine uptake. Low efficacy of the seasonal vaccine is a poor incentive to increase integration into immunization schedules.
The committee distinguishes two factors that affect demand generation for seasonal influenza vaccines: a country’s willingness to include seasonal influenza vaccines in its vaccination programs, and uptake by the public once a vaccine is in the program. Country willingness is largely driven by cost-effectiveness analyses. A lack of cost-effectiveness analyses in LMICs
is a significant barrier to implementing seasonal influenza vaccination programs (Kraigsley et al., 2021). Though seasonal influenza is often not considered a high-priority disease, the Global Burden of Disease Study revealed that influenza is responsible for a high burden of disease when considering comorbidities, particularly in LMICs. In 2017, lower respiratory tract infections resulting from seasonal influenza led to 145,000 deaths and nearly 9.5 million hospitalizations worldwide (GBD 2017 Influenza Collaborators, 2019). Seasonal influenza clearly has a significant global health and economic impact. Influenza vaccines remain underutilized because there are relatively few economic evaluations done on seasonal influenza vaccination, leading to poor understanding of the benefits of vaccination to guide decision makers (Newall et al., 2018).
A seasonal influenza vaccine program is also crucial in demand generation for both seasonal and pandemic influenza. An evaluation of a program conducted by WHO during the 2009 H1N1 influenza pandemic to increase equitable access through donations found that countries with existing seasonal influenza programs were better prepared for a pandemic. The countries with robust seasonal influenza vaccination programs were more likely to apply for and receive the vaccine. The existence of a program ensured the infrastructure for transportation, storage, and administration of the vaccine, along with trained staff and a population that were familiar with influenza vaccine in the country (Porter et al., 2020). Robust seasonal influenza vaccine programs increase national familiarity with the management of influenza vaccine and therefore enhance pandemic preparedness. Therefore, investment in seasonal influenza programs can result in broader global health security.
RECOMMENDATION 4-4: The Centers for Disease Control and Prevention and the National Institutes of Health should support the development of better models for influenza vaccine cost effectiveness. The U.S. Agency for International Development should support technical assistance to strengthen country systems for vaccine uptake.
GLOBAL DISTRIBUTION AND TRANSPORTATION
Transporting vaccines to the point of administration is subject to bottlenecks with transport capacity and cold chain requirements at both global and local levels. The cold chain requires significant inputs of energy and resources, along with expensive equipment, to ensure that vaccine integrity is maintained to the point of administration. This section discusses vaccine supply chain and logistics considerations at both the global and country levels.
Transportation
The committee identified transport capacity and cold chain considerations as primary logistical constraints in the transportation of vaccines from a manufacturer to the recipient country. Multilateral organizations, such as Gavi and UNICEF, assist in this transport. Transport capacity is often strained during a public health emergency and can delay the delivery to a country’s point of entry. As seen in the COVID-19 pandemic, a drastic reduction of commercial flights significantly reduced the available volume of cargo space, thereby limiting vaccine transport (Kiernan, 2021). In addition, personal protective equipment, ancillary supplies, treatment products, and other materials needed for emergency response all compete for limited space in transport systems.
Getting influenza vaccines from manufacturers to recipients while maintaining the cold chain is a logistical challenge. Limited storage capacity and inefficient distribution and logistics systems have long been bottlenecks in the supply chain, particularly for LMICs (Zaffran et al., 2013). The development of mRNA vaccines for COVID-19 brought this challenge to the fore, and in response, the manufacturers of those vaccines have developed detailed logistics plans and special insulated containers to ensure that their vaccines get to the intended recipients (Pfizer, 2020). In addition, major logistics companies, including FedEx, UPS, and DHL, invested in new storage facilities for cold chain management that are expected to prove useful after the COVID-19 pandemic is over. Many major cargo handling airports already had extensive temperature-controlled handling facilities with direct apron access. Frankfurt airport, for example, has approximately 12,000 square meters of temperature-controlled handling capacity, including 8,000 square meters at the Lufthansa Cargo Pharma Hub, that meet all international standards (International Airport Review, 2020). Abu Dhabi has also made significant investments for cold chain transportation in its international airport, with the intent to serve as a hub for global pharmaceutical logistics (Etim, 2020). The International Air Transport Association has a program for certifying airports as pharmaceutical freight hubs capable of safely and efficiently handling temperature-sensitive and time-sensitive pharmaceuticals, including vaccines. In June 2021, the association updated its guidance for vaccine and pharmaceutical logistics and distribution (IATA, 2021).
Various international aid organizations have invested substantial funds in developing global cold chain equipment and services for distributing vaccines. UNICEF, for example, procured close to $100 million worth of cold rooms, refrigerators, cold boxes, and insulated vaccine carriers, primarily to deliver vaccines for administration to children. Gavi’s cold chain equipment optimization platform provides funding to procure cold chain equipment and also provides market shaping strategies to make equipment more accessible (Gavi, 2019a). The UN Environment Programme notes, however,
that even for countries with effective childhood vaccine distribution, “the sheer scale and urgency of mass … vaccination” during a pandemic, such as COVID-19, would be very difficult for “countries with large rural populations” (UNEP, 2020, para. 9). Transportation considerations are also important in planning for reverse logistics, when unused vaccines are returned in order to be redistributed (if appropriate) or destroyed. This process requires supply management and storage at every level of the supply chain (WHO and UNICEF, 2021a). Stringent procedures in place for redistributing or destroying vaccines is important for maintaining public safety.
Product and ancillary supplies could follow multiple pathways to a country. Global logistics (from manufacturer to country port of entry) requires complex coordination of multiple agencies, receiving countries, manufacturers, and logistics providers. A pandemic context places strains on transport capacity that could affect flow of materials and products across the end-to-end supply chain. Better pre-pandemic planning is required to anticipate issues and address them.
Trade Barriers
As the world rallied around the need to equitably distribute vaccines to fight the COVID-19 pandemic, it became clear that vaccine nationalism was a threat to achieving that goal (Evenett et al., 2021). As the World Bank has noted, vaccine nationalism can take the form of overt export bans or limits that are designed to increase domestic vaccine supplies at the expense of foreign supplies, or it can use more subtle mechanisms, such as delaying promised vaccine shipments (Evenett, 2021; Evenett et al., 2021). Perception of an action’s intent may vary greatly depending on where one sits in relation to the outcome. Some notable examples of perceived vaccine nationalism were the European Union’s move to prohibit export of COVID-19 vaccines and India’s curb of vaccine exports, the latter of which had a severe impact on 91 nations that were depending on vaccines produced in India (Peters and Prabhakar, 2021; Som, 2021). However, both of these export restrictions were implemented after devastating second waves hit each region, and the countries aimed to secure vaccines for their own populations who were in need of vaccines (Kar et al., 2021). The United States leveraged the Defense Production Act, which requires U.S. manufacturers to prioritize fulfilling contracts with the U.S. government (White House, 2021) (see Box 3-1 in Chapter 3). While the United States did not restrict exports of COVID-19 vaccines, it did impose restrictions on the export of key raw materials needed to manufacture COVID-19 vaccines. There are concerns that this could limit the supply of these components to global manufacturers (Jain and Rocha, 2021).
According to the American Society of International Law, the treaties that form the basis for the multinational trade system—the General Agreement on Tariffs and Trade, the General Agreement on Trade in Services,
the Agreement on Trade-Related Aspects of Intellectual Property Rights, the Agreement on the Application of Sanitary and Phytosanitary Measures, and the Agreement on Technical Barriers to Trade—provide exceptions that allow countries to put export restrictions in place when contracts are already signed during a global pandemic (Ibrahim, 2021). However, given the near universal condemnation of these export restrictions, several organizations and authors have called for negotiations to reduce these exceptions to effectively address the next pandemic (Pauwelyn, 2020). One specific proposal, for example, calls for importers to eliminate import tariffs in exchange for a guarantee that exporters drop export restrictions and only impose new ones under strict conditions (Evenett and Winters, 2020). This latter approach could be workable given the strong trade interdependencies in the goods needed to produce, distribute, and administer vaccines: see Figure 4-1.
The World Trade Organization (WTO) has noted that many LMICs depend on imported vaccines that are either donated or sold at reduced prices, which may increase diversion of these vaccines into higher-priced markets. WTO clearly states that
to ensure that vaccines reach and stay in a destination country, appropriate measures, including but not limited to those required under the Agreement on TRIPS [Trade-Related Aspects of Intellectual Property] Rights, need to be taken by the importing country against unlawful diversion to prevent re-exportation of the vaccines, and by all WTO members to prevent the importation and sale of diverted vaccines in their territories. (WTO, 2020, p. 26)
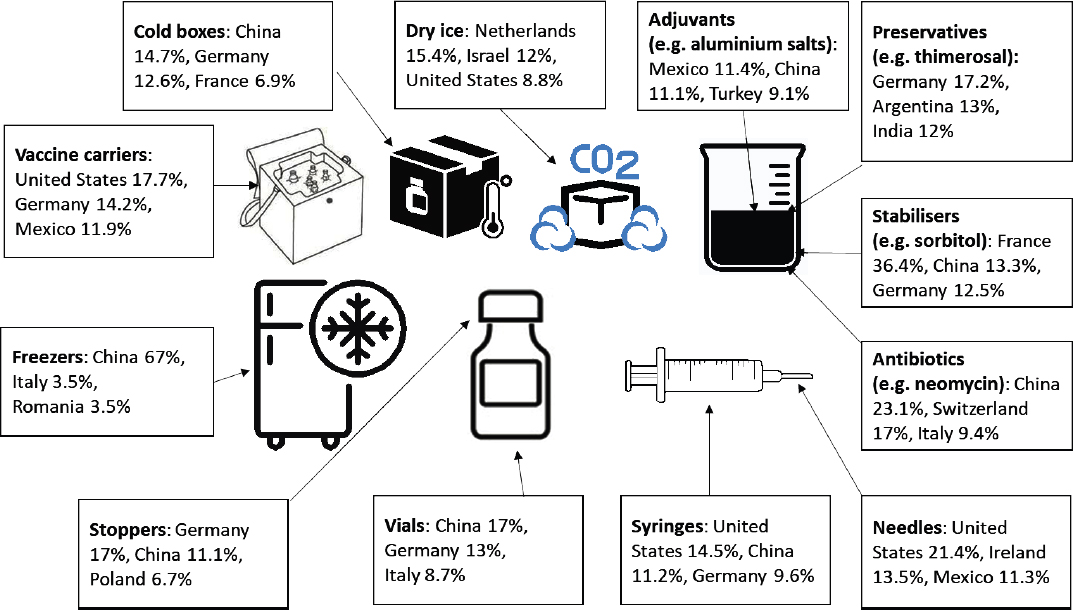
SOURCE: Andrenelli et al., 2021, p. 6.
The Organisation for Economic Co-operation and Development has noted that “while tariffs on vaccines are unlikely to pose a major challenge” to vaccine distribution, high tariffs on vaccine-related inputs could present a problem (Andrenelli et al., 2021, p. 9). Average world tariffs on vaccine ingredients, such as preservatives, adjuvants, stabilizers, and antibiotics range between 2.6 and 9.4 percent. Tariffs on materials to administer vaccines, such as syringes and needles, are in the 4.4–4.5 percent range, while tariffs for primary packaging, including vials and stoppers or distribution materials, including cold boxes, freezers, and dry ice, can hit 12.7 percent. The need to maximize the availability of vaccines during a pandemic may be considered as justification for reducing or waiving such tariffs (Andrenelli et al., 2021). Absent the ability to drop the tariffs, additional subsidies may be needed from aid donors to cover the additional cost or risk that some countries may be unable to afford to meet their vaccine needs (Gavi, 2021).
IN-COUNTRY STORAGE AND DISTRIBUTION
The committee finds that the physical supply chain, from cold chain considerations to transport capacity, is already stressed in LMICs. Once vaccines reach a recipient country, it is that country’s responsibility to transport vaccines to points of distribution and administration. Transport capacity in LMICs is highly variable, which has significant effects on speed and efficiency of vaccine rollout. Throughout this process, optimal temperatures, which vary depending on the type of vaccine, have to be maintained.
Current capacity is already insufficient to cover pediatric immunization platforms, which, in the committee’s experience, are the most globally developed and reliable vaccine supply chains. Since 2010, the number of vaccines introduced into LMICs has increased due to programs such as the Expanded Program on Immunization (EPI) to increase global access to vaccines. Despite the efforts to improve vaccination programs in LMICs, many countries have struggled to achieve target levels of vaccine introduction. A primary factor limiting vaccination success is that new vaccines increase logistical complexity, as new vaccines may not fit into established EPI schedules and require additional steps for delivery (Guignard et al., 2019). Concurrently, these introductions often require changes to immunization programs, which may alter other logistical and transport needs in order to achieve vaccination targets. The growing complexity of these systems has to be considered when incorporating new vaccines (Guignard et al., 2019).
The addition of pandemic vaccines into immunization schedules can result in further burdening of supply chains. COVID-19 vaccination in
Africa is predicted to require increased storage capacity at national levels (Ortiz et al., 2021). These dynamics reveal that addition of new vaccines must be implemented with consideration for financial and structural resource constraints, highlighting the importance of careful planning. Failure to address these dynamics may perpetuate low uptake of influenza vaccines into national vaccine schedules due to the high burden on local supply chains and competition for limited resources with other priority vaccines.
Cold Chain Considerations
The temperature instability of vaccines can emerge as a key challenge to distributing an approved vaccine for widespread use. Vaccine manufacturers attempt to minimize temperature instability using various techniques, but influenza vaccines, like many other vaccines, must be kept within a narrow temperature range—cold, but not frozen—throughout manufacturing, distribution, storage, and administration. The vaccine cold chain ensures they retain their potency and remain safe for human use. A single transport leg or storage port that is not temperature controlled will break the cold chain and can decrease the potency of the vaccine to the point it is rendered ineffective. Vaccine wastage rates in LMICs are highly variable among countries, and wastage data are not readily available for many countries (Parmar, 2010). A study by Karp et al. (2015) estimated that around 8 percent of the total cost of immunization programs in LMICs is used on vaccines that are eventually wasted. Wastage is primarily attributed to improper temperature storage and expiration (Songane, 2018). Breaking the cold chain can also mean that patients will need to be revaccinated (Kroger et al., 2021), representing another waste of vaccine and the potential that patients may refuse to be revaccinated, thereby remaining unprotected.
According to guidance from CDC (2021f), maintaining an effective cold chain relies on having a well-trained staff, reliable storage and temperature monitoring equipment, and accurate vaccine inventory management. CDC recommends that every facility that receives, distributes, or administers influenza vaccines maintain clearly written, detailed, and up-to-date storage and handling standard operating procedures and train staff on those procedures. All currently approved influenza vaccines have to be stored between 2° and 8° and never frozen. Should an mRNA vaccine for influenza be approved for human use in the future, it will likely need to be stored at much lower temperatures: the Pfizer/BioNTech COVID-19 vaccine, for example, requires storage at –70°C (±10°C) (Pfizer, 2020), while storage of the Moderna mRNA COVID-19 vaccine for longer than 30 days requires a temperature of –20°C (Moderna, 2020).
While there are pharmaceutical-grade, purpose-built refrigerators designed specifically to store vaccines, household-grade refrigerators can be an acceptable option for vaccine refrigeration under the right conditions. The advantage of purpose-built devices is that they include microprocessor-based temperature control with a digital temperature sensor along with fan-forced air circulation using powerful fans or multiple cool air vents that promote uniform temperature and fast temperature recovery from an out-of-range temperature. These specially designed refrigeration units may be cost-prohibitive in LMICs.
Temperature monitoring is essential to ensure that proper temperatures have been maintained throughout storage. CDC says that every vaccine storage unit must have a temperature monitoring device. Digital data loggers are the most accurate of such devices, according to CDC, as they detail how long a unit has been operating outside the recommended temperature range. Simple thermometers only show the highest and lowest temperatures that were reached in a unit, but a digital data logger provides detailed information on all temperatures recorded at preset intervals (CDC, 2021f). Other technologies, such as stickers that change color when exposed to temperatures outside of the recommended range, have also been used with previous vaccine introduction efforts (Zipursky et al., 2014).
One of the challenges of distributing influenza vaccines in remote or rural communities in LMICs is the unavailability or unreliability of electricity to power refrigeration units. Ultra-cold freezers that reach temperatures of –70°C require high energy inputs and costly equipment to maintain the required temperatures. Cold boxes that are capable of operating at temperatures from –70°C to –80°C cost from $10,000 to $20,000 each. This was a particular challenge for the mRNA-based COVID-19 vaccines, and experts have assessed that vaccines requiring ultra-cold storage are unlikely to be widespread solutions in some LMICs (Sustainable Energy for All, 2021). To address this challenge, PATH has a program to evaluate and design technologies and systems that can extend the cold chain beyond the power grid, including containers that will prevent vaccines from freezing as they are transported in ice-packed carriers to remote locations (PATH, 2018). Since 2017, UNICEF, partnering with WHO and Gavi, has installed more than 40,000 solar-powered cold chain refrigerators, mostly in Africa. UNICEF is also promoting solar technologies to help countries maintain their vaccine supply chains. For example, in South Sudan, the world’s least electrified country, more than 700 health facilities have been equipped by UNICEF with solar power refrigerators, accounting for approximately 50 percent of all facilities nationwide (UN, 2020). Pfizer has also developed innovative thermal shipping containers for COVID-19 vaccines that ensure temperature control during the transit of vials and can be used as temporary storage units for up to 10 days. For longer shelf life, ultra-low-temperature
freezers are needed, where vaccines can be stored until their expiration dates. Prior to mixing, the Pfizer vaccine can be stored at standard refrigeration temperatures (2°–8°C) for 31 days (CDC, 2021e).
Cold chain capacity is a persistent bottleneck, as it requires financing for complexities, such as data loggers and other equipment, and requires accurate reporting. Adding new vaccines without adequate increase in capacity puts a strain on already limited systems, and these strains are magnified for vaccines requiring ultra-cold storage. Good data-logging ability is needed to ensure that vaccines were not compromised in order to move vaccines between different countries. Monitoring temperatures and collecting data needs to be applied to vaccine storage, not just shipment. Up-front investment and increased support for cold chains (including off-grid solutions) need to be highlighted ahead of time during a pandemic preparedness phase.
Vaccine Delivery and Rollout
Vaccine delivery is the final step in distribution, where vaccines will be administered (CDC, 2021a). Delivery systems vary based on the source of the vaccines and the location of delivery. In the United States, routine vaccines, and recently COVID-19 vaccines, are ordered from the federal government by states, territories, and local jurisdictions, which are fulfilled through a federal system. The Vaccine Tracking System is used to make orders and log data related to vaccine delivery (CDC, 2021a). Even for this system there is an exception, as Pfizer has its own system to deliver vaccines. Global entities, such as UNICEF and COVAX,4 also have their own delivery systems to bring vaccines to countries. Entities that are filling vaccine orders establish allocation criteria for prioritized delivery when demand outpaces supply, such as for COVID-19. Considerations include the number and distribution of at-risk populations, such as health care workers and individuals with compromised immune systems (WTO, 2021).
Once vaccines from a multilateral distributor arrive in the target country, the national government becomes responsible for delivery to the final destination. In LMICs, vaccines are often transported from the country point of entry to a distribution center, where vaccines are transported to other local distribution points or to the vaccination site. This “last mile” delivery is subject to several challenges, particularly in limited resource settings. Lack of funding to deliver vaccines is a critical issue. Personnel, cold chain requirements, and transportation costs can all serve as added challenges to vaccine transportation funding, particularly in a pandemic scenario when a significant portion of the population is targeted for vac-
___________________
4 COVAX is the acronym for COVID-19 Vaccines Global Access; see fn. 9 in Chapter 2.
cination. Additional delivery costs for COVID-19 vaccines for the average LMIC are estimated to represent nearly 20 percent “of the entire preCOVID-19 health budget” (UNICEF, 2021). In addition to the cost, poor infrastructure often serves as a barrier to scale up vaccine rollout efforts. In LMICs, primary care centers often serve as the primary access point for vaccines. Vaccine uptake in these settings may be influenced by the relationship between the health care facilities and the local community. Health care workers commonly engage in community outreach activities, such as school programs, which can bolster trust and increase knowledge of vaccines (Guignard et al., 2019). Efforts to increase vaccine access must account for these structural and financial barriers to last-mile delivery.
The operational cost of getting influenza vaccines to people is significant, placing a high burden on some LMIC country budgets. Beyond standard transportation and distribution costs, human resource capacity for distribution and delivery is a major challenge. There are significant gaps in funding of these operational expenses. There is a need to determine effective ways to resource in-country distribution and delivery.
Vaccination Strategies
Various kinds of locations can serve as pandemic vaccination sites, and there are benefits and drawbacks of different options. Mass vaccination sites, as were established during COVID-19, serve as central points of administration that can potentially have higher throughput, vaccinating more people per day (Everett et al., 2013). The benefits of this model include easier delivery of vaccines and critical components to one centralized location, rather than dividing the same volume of resources among multiple, smaller health care settings, which can translate to easier management of the cold chain and less wastage of vaccine doses. It is also easier to schedule appointments with one large facility than seeking limited appointment slots from numerous smaller sites. Despite these benefits, access remains a key issue with mass vaccination. Communities that are distant from mass vaccination sites may face challenges in access. Also, establishing a mass vaccination site requires significant investment and time. A lack of health care personnel available to meet the needs of a mass vaccination site is also a limitation, especially in LMICs (He, 2021).
Other methods of vaccination may fit more localized contexts. Outreach strategies often require health care workers to travel to the vaccine recipients. Vaccine dissemination through local clinics and health care centers has the advantage of being more accessible to geographically dispersed communities. In addition, mobile vaccination units have been used effectively in settings where local health care access is especially limited, such as rural and remote areas. Mobile vaccination has been found to be effective in
advancing vaccine coverage in adults living in remote regions in India (Ghia et al., 2021). In LMICs, vaccination occurs mostly in primary care settings, not hospitals. Outreach strategies that involve health care workers traveling to the vaccine recipients often require real-time, flexible systems that connect public health, high operational cost, logistics, and medical experts.
Various vaccination strategies need to be evaluated and applied based on the localized context, accounting for availability of resources and needs of the community (He, 2021). Guidance documents, such as the WHO Framework for Decision-Making (WHO, 2020b), can assist countries in selecting appropriate methods for vaccination campaigns. The committee finds that these considerations for vaccination methods are generally lacking from country vaccination plans: they need to be considered earlier on in the pandemic preparedness process to be fully integrated into preparedness activities. Different types of vaccination strategies (including clinic-based, mass vaccination sites, mobile sites) create different transport, logistics, and operational challenges. Country planning needs to determine the appropriate mix of these modalities, perform better cost estimation, seek appropriate funding, and solicit private-sector expertise for participation in design logistics, distribution, and operational systems.
Ancillary Supplies
Vaccine delivery strategies are also linked to the supplies required to conduct vaccinations. Ancillary supplies, such as syringes, and workforce capacity are critical components of the vaccination effort (Hatchett et al., 2021). However, these supplies represent additional items and logistical constraints that have to be accounted for when developing vaccine delivery strategies. Procurement of ancillary supplies also varies by country. In Kenya, although the national government is responsible for vaccine purchases, counties are responsible for procuring other supplies, such as syringes, and data collection tools. Counties often lack funding to procure these supplies, which puts vaccination programs at risk (VxDel, 2020).
Globally, the COVID-19 pandemic has resulted in a syringe shortage, which may have hindered vaccine rollouts. Extracting the maximum number of doses from multi-dose vials, which are used for the Pfizer and Moderna vaccines, requires a specialized “low dead volume” syringe that minimizes the amount of vaccine left in the syringe. These syringes are in short supply even in countries that have greater access to vaccines and supplies, such as the United States. Many of the syringes supplied by the U.S. government during Operation Warp Speed were not low dead volume syringes and were therefore not used. Some vaccination sites were able to procure their own syringes, but others that could not saw vaccine doses go to waste. When standard syringes are used, doses in vials can still be admin-
istered, but fewer doses can be extracted from each vial due to the amount of vaccine that is retained by the syringe itself (Moutinho, 2021). Both the availability of syringes and the types of syringe used have implications for vaccine wastage and efficacy of vaccination systems.
Vaccines that can use novel routes of administration would have significant impacts on the supply chain. For example, microneedle patch technologies are being developed, in which a patch containing the vaccine is applied to the skin without need for syringes or trained health care personnel for administration (Wedlock et al., 2019). Models of widespread use of patch technologies for some routine vaccinations revealed a significant decrease in vaccine wastage, although specific effects on the supply chain varied depending on context-specific cold chain capacities (Wedlock et al., 2019). It is important to invest in new technologies for administration due to the potential benefits to the supply chain and reduced strain on a limited workforce, even though the variety in ancillary supplies (e.g., different types of syringes and delivery devices) creates complexity in distribution and logistics.
MATCHING SUPPLY AND DEMAND
In a pandemic scenario, the goal of vaccine rollout should be to have a population that becomes widely immunized. In order to achieve this, decision makers need “evidence-based vaccine delivery strategies that generate demand, allocate and distribute vaccines, and verify coverage” (Weintraub et al., 2021, p. 1). Data on vaccine availability and uptake is foundational for successful policy development. Visibility on the location of the finished product is essential for knowing how much has been administered and how much remains in storage or in transit.
As of the 2021 vaccine rollout, there is a lack of end-to-end visibility on the full vaccine supply chain (IATA, 2021). Vaccine management databases in health care facilities track local use and allocation, but these systems are not integrated. As a result, there is a general lack of awareness regarding the location of vaccine batches, which proves challenging for determining where vaccines would be best distributed, and how much should be distributed (Iwu et al., 2019). A study on vaccine inventory visibility revealed that vaccine allocation strategies that account for both population and inventory information were significantly more efficient than strategies based on population alone. Simulated population and inventory-based models resulted in lower infection rates in a given region and significantly lower inventory leftover (Li et al., 2018). Sending vaccines to areas where they will not be used or failing to distribute vaccines to areas where they are being used will result in wasted efforts in supply chain management and transit. The issue of low visibility for vaccines points to a larger issue of limited data availability for health care in general. Information capture and analytic
capability is needed to assess visibility and understanding of product movement. Lack of visibility is most evident for distribution. There is a need to carefully balance supply with capacity so as not to overwhelm systems.
On the demand side, the complicated and rapid vaccine rollout for COVID-19 resulted in confusion among the general public regarding visibility of vaccine access. Appointment systems have not been standardized across districts, resulting in unclear messaging and potentially lower vaccine uptake. When administering vaccines that require two doses, there is the additional challenge of ensuring that each recipient receives both of them for full protection (Sellers, 2020). The lack of data regarding vaccine availability has led to unclear communication and identification of vaccination sites. In the United States during times of high demand for COVID-19 vaccines, complicated online appointment systems led to challenges in vaccine access among the general public. Appointment websites experienced crashes, and there was a lack of a unified appointment system (Weise, 2021). Individuals with lower technological access or competency struggled to navigate online appointment systems, leading to lower vaccination rates among some populations (Solaiman, 2021). Low visibility on both the global supply and local demand side ultimately leads to lower uptake and improperly distributed resources.
In planning and monitoring a vaccination campaign, whether for seasonal or pandemic influenza, information systems need to be able to provide data on the uptake of vaccines, both by targeted population groups and by regions, as well as data that public health officials can use to monitor and evaluate the logistical chain that supplies vaccines where they are needed. Currently, “there is no global monitoring system for influenza vaccination coverage,” making it difficult to match vaccine supply and demand (Palache et al., 2021, p. 6082). The International Federation of Pharmaceutical Manufacturers & Associations has developed a survey method to assess global distribution of influenza vaccine as a proxy for vaccination coverage rates. This information, while a significant contribution to vaccine monitoring, does not account for vaccination policies or directly measure vaccine uptake (Palache et al., 2021). In the United States, CDC uses data from two telephone surveys, National Immunization Survey-Flu5 and the Behavioral Risk Factor Surveillance System6 to estimate influenza vaccination coverage for the U.S. population during each influenza season (CDC, 2020b). For COVID-19 vaccinations in the United States, data on administered doses are reported to CDC by various vaccination providers (CDC, 2021a).
___________________
5 NIS-Flu is a national random-digit-dialed cellular telephone survey of households.
6 BRFSS is a state-based random-digit-dialed cellular and landline telephone survey that collects information on a variety of health conditions and risk behaviors from one randomly selected adult ≥18 years in a household.
WHO and UNICEF, with input from experts and stakeholders, have developed an assessment tool based on WHO’s effective vaccine management framework that countries can use to generate evidence about bottlenecks in the immunization supply chain (WHO and UNICEF, 2016). This tool can assess primary stores of vaccines—the quantities received directly from manufacturers—as well as at the level of provinces and districts, local distributors, and health facilities. Based on this assessment, countries can develop and finance comprehensive improvement plans to strengthen immunization supply chains to help ensure that vaccines are available, that vaccines remain potent, and that resources are efficiently used. This tool also assesses information systems and supportive management functions involved in vaccine storage and distribution. Data generated using this tool have shown, for example, that only about one-third of LMICs in 2014 were in compliance with WHO-recommended vaccine management policies, which were designed to minimize vaccine waste and increase the odds that supply and demand are in balance (WHO and UNICEF, 2016). The Americas was the only region in which those systems met the WHO standard, with the information systems of every other global region being far from the effective vaccine management standard (WHO and UNICEF, 2021b).
Logistics management is another critical component of visibility. The UN Development Programme developed the Logistic Management Information System (LMIS) to assist countries in collecting and reporting data related to health systems management, with a focus on maintaining supply chains (UNDP, 2021). In addition to this suite of tools, there are numerous information systems available that can track regional vaccine availability or facilitate vaccine management and ordering for health care and vaccination sites. These systems are critical, as a lack of visibility in vaccine management can lead to vaccine stockouts in health facilities. A study in Tanzania revealed that implementation of LMIS facilitated vaccine availability better than paper or Excel-based systems, reducing events of vaccine stockouts (Gilbert et al., 2020).
Immunization information systems are public health data systems for tracking vaccinations. These tools were developed for public health authorities to track and measure the incidence of vaccination within their jurisdiction and assist with distribution and inventory management. A typical system can be used to track immunizations given and collect data on vaccine coverage to inform immunization campaigns. Several immunization information systems tools exist and serve a range of functions, including vaccine planning, delivery and administration, and monitoring. However, these systems operate in silos and lack integration. Also, most immunization information systems are designed for childhood immunization schedules and are not tailored to adults (Gilbert, 2021), and lack the capability
to identify individuals or population segments that may be particularly at risk or require targeted outreach for vaccination.
Immunization information systems can also be used to help monitor vaccine safety and effectiveness, but they have to be used in tandem with other data routinely contained in patients’ individual medical records and health payer claims data (Lenert et al., 2021). In LMICs, the systems have not changed much since they were adopted in the 1970s by the WHO Expanded Program on Immunizations. Data are often still collected on paper and compiled into reports to evaluate performance indicators. Data quality is often poor, incomplete, or gathered too late to make timely decisions. The introduction of newer, often more costly vaccines creates additional challenges in tracking and maintenance (PATH and WHO, 2013). Even in developed countries, such the United States, these systems are disparate: for example, there are approximately 60 systems at state and local levels, connected only at the national level (CDC) to information national policy. The individual systems are developed to meet national reporting requirements, but they are often built on different platforms that cannot share information easily. In the absence of a uniform patient identifier, cross-referencing immunization information system data with patient outcomes, including death records, can be extremely difficult (Lenert et al., 2021).
During the 2009 H1N1 influenza pandemic, a large-scale U.S. study using health insurance claims and immunization systems data demonstrated the potential for understanding vaccine safety, while also highlighting some of the major challenges and gaps in the systems (Yih et al., 2012). COVID-19 has further shown the need and potential for more advanced systems that provide real-time dating, incorporate immunization campaigns, offer better capacity to identify and report adverse events, and interact with primary care providers. Greater public awareness of the immunization process following COVID-19 could be leveraged to develop support for these systems (Atkinson et al., 2020).
Though these management systems are useful on local levels, variability in systems across localities leads to limited interoperability and challenges with communication at higher levels (Atkinson et al., 2020). This lack of integration among vaccine data networks leads to low visibility on where vaccines are sent. Data systems also do not capture demographic information on the vaccine recipients. In the state of Georgia, there was high variability in vaccination rates among counties and demographic groups. Studies revealed that people were traveling across county lines in order to receive a COVID-19 vaccine. In addition, white populations were significantly more likely than other groups to have received a vaccine, and vaccine coverage varied significantly on a local level (Wainscott-Sargent, 2021). These findings reveal that high-level data on vaccine administration often provides an
incomplete picture. Data need to be collected on a community level to assess vaccine uptake (Wainscott-Sargent, 2021). In these situations, there is a need for additional data on why residents of the counties with low demand are less likely to get the vaccine. Accessibility, time off work, or hesitancy could all be contributing factors. Interventions can be developed, but the primary reasons for low uptake have to be identified and addressed (Surgo Ventures, 2021). Within a country, there is a need to identify areas where inventory is available but demand is weak. In areas with less demand, efforts should be made to determine the drivers of low uptake. Resources may need to be allocated to generate demand, which includes advocacy for vaccine uptake and addressing vaccine hesitancy.
Immunization information systems for routine immunization are relevant to planning, delivery, and administration in supply chain logistics and procurement (delivery, cold chain monitoring). They also facilitate monitoring and evaluation of health information systems, adverse events, and digital vaccine certificates. Integration and interoperability of different information systems (e.g., LMIS and immunization information systems) is needed for efficient flow, combatting vaccine wastage, and informing better allocation in general, and given their scale, especially for influenza and adult immunization programs. The value of globally aggregated data is widely accepted (PATH and WHO, 2013). Organizations need to work with country governments to build trust. A tiered process could work to build trust, where lower-level data are first shared and evolve to more sensitive data.
As observed during COVID-19, vaccine distribution has not been efficient. Allocation based solely on population has led to vaccine scarcity in some areas and overabundance in others, as vaccine uptake varies. Both COVID-19 and influenza are best prevented by equitable vaccine distribution and uptake. Distributing influenza vaccines during the COVID-19 pandemic has been hindered by additional strain on health services and a decline in people’s seeking health care. However, developing tools that account for some key factors can facilitate development of equitable distribution plans. A study on vaccine allocation in Iran developed a model that accounted for location of vaccine distribution points, cost of transportation to administration sites, distribution center capacity, per-dose holding and transport costs, and the coverage rate of the target population. It also accounted for the priority vaccination groups, including health care workers, the elderly, and pregnant women (Rastegar et al., 2021). This model provided insight on the best distribution center in terms of cost, accounting for local budget constraints. Factors affecting allocation and distribution are similar for both influenza and COVID-19 vaccines. Insight into best practices in allocation for one yields valuable insight that can be transferred to the other.
Efficient global distribution of vaccines in a pandemic relies on the existence, coordination, and successful operation of a collection of robust systems. These systems include physical infrastructure for transportation, storage, distribution, and delivery to individuals. They also include information systems that help to coordinate demand and delivery of products, as well as information systems that provide comprehensive information on populations, vaccine coverage, safety, and effectiveness. Furthermore, the necessary systems include a planning and organization structure at the global and local levels with the principal objective of delivering vaccines. Tools need to be available to assist countries and global institutions with planning vaccination strategies, including selection of sites, supply and logistics, and operational setup of vaccination sites for efficiency. Tools are also needed to lower the cost of ownership for cold chain equipment, which serves as a bottleneck in vaccine transportation. Enhanced forecasting capabilities will also facilitate countries’ abilities to anticipate demand and plan to meet needs for seasonal and pandemic vaccination. Improved models for demand forecasting are also needed that are geographically grounded and account for human behavior. To facilitate each of these components, tools for more accurate operational distribution and delivery cost estimates to ensure necessary financing cannot be overlooked.
RECOMMENDATION 4-5: The Office of Global Affairs, in partnership with the World Health Organization, Gavi, UNICEF, and relevant global funders, should facilitate the development of global tools to help countries with better supply planning for vaccines and ancillary supplies planning, allocation, and rollout decisions, and with obtaining necessary funding for operations.
REFERENCES
Andrenelli, A., J. L. Gonzalez, and S. Sorescu. 2021. Using trade to fight COVID-19: Manufacturing and distributing vaccines. Paris, France: Organisation for Economic Co-operation and Development.
Atkinson, K. M., S. S. Mithani, C. Bell, T. Rubens-Augustson, and K. Wilson. 2020. The digital immunization system of the future: Imagining a patient-centric, interoperable immunization information system. Therapeutic Advances in Vaccines and Immunotherapy 8.
Bernasconi, V., P. A. Kristiansen, M. Whelan, R. G. Román, A. Bettis, S. A. Yimer, C. Gurry, S. R. Andersen, D. Yeskey, H. Mandi, A. Kumar, J. Holst, C. Clark, J. P. Cramer, J. A. Rottingen, R. Hatchett, M. Saville, and G. Norheim. 2020. Developing vaccines against epidemic-prone emerging infectious diseases. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 63(1):65–73.
Bhan, M. K., and B. Sinha. 2019. Immunisation against influenza in low-income and middle-income countries. Lancet Global Health 7(7):e827–e828.
Bresee, J., J. Fitzner, H. Campbell, C. Cohen, V. Cozza, J. Jara, A. Krishnan, V. Lee, and WHO Working Group on the Burden of Influenza Disease. 2018. Progress and remaining gaps in estimating the global disease burden of influenza. Emerging Infectious Diseases 24(7):1173–1177.
Carter, S., L. Mobula, H. Samaha, and S. M. Ahuka. 2020. Community engagement and vaccinations during the Ebola outbreak in Democratic Republic of Congo. In World Bank Blogs: Investing in health. https://blogs.worldbank.org/health/community-engagement-and-vaccinations-during-ebola-outbreak-democratic-republic-congo (accessed October 14, 2021).
CDC (Centers for Disease Control and Prevention). 2020a. CDC seasonal flu vaccine effectiveness studies. https://www.cdc.gov/flu/vaccines-work/effectiveness-studies.htm (accessed July 7, 2021).
CDC. 2020b. Flu vaccination coverage, United States, 2019–20 influenza season. https://www.cdc.gov/flu/fluvaxview/coverage-1920estimates.htm (accessed July 12, 2021).
CDC. 2021a. About COVID-19 vaccine delivered and administration data. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/distributing/about-vaccine-data.html (accessed August 9, 2021).
CDC. 2021b. Immunization information systems (IIS). https://www.cdc.gov/vaccines/programs/iis/2d-barcodes/shipping-table.html (accessed August 9, 2021).
CDC. 2021c. Interim clinical considerations for use of COVID-19 vaccines currently approved or authorized in the United States. https://www.cdc.gov/vaccines/covid-19/clinical-considerations/covid-19-vaccines-us.html (accessed July 12, 2021).
CDC. 2021d. Operational considerations for routine immunization services during the COVID-19 pandemic in non-U.S. settings focusing on low- and middle-income countries. https://www.cdc.gov/coronavirus/2019-ncov/global-covid-19/maintaining-immunizationservices.html (accessed July 5, 2021).
CDC. 2021e. Pfizer-BioNTech COVID-19 vaccine storage and handling summary. https://www.cdc.gov/vaccines/covid-19/info-by-product/pfizer/downloads/storage-summary.pdf (accessed September 5, 2021).
CDC. 2021f. Vaccine storage and handling toolkit. https://www.cdc.gov/vaccines/hcp/admin/storage/toolkit/index.html (accessed September 1, 2021).
Crommelin, D. J. A., T. J. Anchordoquy, D. B. Volkin, W. Jiskoot, and E. Mastrobattista. 2021. Addressing the cold reality of mRNA vaccine stability. Journal of Pharmaceutical Sciences 110(3):997–1001.
Debellut, F., N. Hendrix, J. R. Ortiz, P. Lambach, K. M. Neuzil, N. Bhat, and C. Pecenka. 2018. Forecasting demand for maternal influenza immunization in low- and lower-middle-income countries. PLoS One 13(6):e0199470.
Dellepiane, N., and D. Wood. 2015. Twenty-five years of the WHO vaccines prequalification programme (1987–2012): Lessons learned and future perspectives. Vaccine 33(1):52–61.
Etim, T. 2020. Abu Dhabi brings hope to global vaccine distribution. Air Cargo Eye, November 27.
Evans, W. D., and J. French. 2021. Demand creation for COVID-19 vaccination: Overcoming vaccine hesitancy through social marketing. Vaccines 9(4):319.
Evenett, S. J. 2021. Export controls on COVID-19 vaccines: Has the EU opened Pandora’s box? Journal of World Trade 55(3):397–408.
Evenett, S. J., and L. A. Winters. 2020. Preparing for a second wave of COVID-19: A trade bargain to secure supplies of medical goods. UK Trade Policy Observatory. https://blogs.sussex.ac.uk/uktpo/files/2020/04/BP40.pdf (accessed October 14, 2021).
Evenett, S. J., B. Hoekman, N. Rocha, and M. Ruta. 2021. The COVID-19 vaccine production club: Will value chains temper nationalism? Washington, DC: World Bank.
Everett, K. H., M. A. Potter, W. D. Wheaton, S. M. Gleason, S. T. Brown, and B. Y. Lee. 2013. Geospatial analytics to evaluate point-of-dispensing sites for mass immunizations in Allegheny County, Pennsylvania. Journal of Public Health Management and Practice 19(Suppl 2):S31–S36.
FDA (U.S. Food and Drug Administration). 2021a. Expiration dating extension. https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policyframework/expiration-dating-extension (accessed August 20, 2021).
FDA. 2021b. FDA in brief: FDA authorizes longer time for refrigerator storage of thawed Pfizer-BioNTech COVID-19 vaccine prior to dilution, making vaccine more widely available. https://www.fda.gov/news-events/press-announcements/fda-brief-fda-authorizes-longer-time-refrigerator-storage-thawed-pfizer-biontech-covid-19-vaccine (accessed October 14, 2021).
Fortune Business Insights. 2020. Influenza vaccines market to rise at 7.7% CAGR till 2026; increasing demand for effective flu vaccines to aid market expansion: Fortune business insights. Pune, India. https://www.globenewswire.com/en/news-release/2020/06/08/2044825/0/en/Influenza-Vaccines-Market-to-Rise-at-7-7-CAGR-till-2026-Increasing-Demand-forEffective-Flu-Vaccines-to-Aid-Market-Expansion-Fortune-Business-Insights.html (accessed October 14, 2021).
Gavi (Gavi, The Vaccine Alliance). 2019a. CCEOP market shaping evaluation. Gavi and John Snow, Inc. https://publications.jsi.com/JSIInternet/Inc/Common/_download_pub.cfm?id=23762&lid=3 (accessed October 14, 2021).
Gavi. 2019b. Gavi announcement: Vaccine manufacturer GS1 compliance. https://www.unicef.org/supply/stories/gavi-announcement-vaccine-manufacturer-gs1-compliance (accessed September 23, 2021).
Gavi. 2021. COVAX explained. https://www.gavi.org/vaccineswork/covax-explained (accessed September 20, 2021).
GBD (Global Burden of Disease) 2017 Influenza Collaborators. 2019. Mortality, morbidity, and hospitalisations due to influenza lower respiratory tract infections, 2017: An analysis for the Global Burden Of Disease Study 2017. Lancet Respiratory Medicine 7(1):69–89.
Ghia, C. J., and G. S. Rambhad. 2021. Developing adult vaccination ecosystem in India: Current perspective and the way forward. Health Services Research and Managerial Epidemiology 8.
Gilbert, S. 2021. Overview of vaccine information systems, current use in routine immunization and future for adult vaccinations (e.g. Influenza). Seattle, WA: PATH.
Gilbert, S. S., N. Bulula, E. Yohana, J. Thompson, E. Beylerian, L. Werner, and J. C. Shearer. 2020. The impact of an integrated electronic immunization registry and logistics management information system (EIR-eLMIS) on vaccine availability in three regions in Tanzania: A pre-post and time-series analysis. Vaccine 38(3):562–569.
Guignard, A., N. Praet, V. Jusot, M. Bakker, and L. Baril. 2019. Introducing new vaccines in low- and middle-income countries: Challenges and approaches. Expert Review of Vaccines 18(2):119–131.
Harvard University. 2021. Public perceptions of “good” and “bad” COVID-19 vaccines pose challenges. https://www.hsph.harvard.edu/news/hsph-in-the-news/public-perceptions-of-good-and-bad-covid-19-vaccines-pose-challenges (accessed August 19, 2021).
Hasan, M. M. 2019. Missing the mark? People in eastern DRC need information on Ebola in a language they understand. Danbury, CT: Translators Without Borders.
Hatchett, R., M. Saville, M. Downham, T. Cueni, L. Bigger, P. Arthur, R. Suri, S. Prasad, and R. Bech. Hansen. 2021. Towards vaccinating the world: Landscape of current COVID-19 supply chain and manufacturing capacity, potential challenges, initial responses, and possible “solution space”—A discussion document. Coalition of Epidemic Preparedness Innovations. https://www.ifpma.org/wp-content/uploads/2021/03/Summit_Landscape_Discussion_Document.pdf (accessed October 1, 2021).
He, N. 2021. Rapid evolution of the COVID-19 pandemic calls for a unified public health response. BioScience Trends 15(4):196–200.
Huang, H., B. Singh, D. P. Morton, G. P. Johnson, B. Clements, and L. A. Meyers. 2017. Equalizing access to pandemic influenza vaccines through optimal allocation to public health distribution points. PLoS ONE 12(8):e0182720.
IATA (International Air Transport Association). 2021. Guidance for vaccine and pharmaceutical logistics and distribution. https://www.iata.org/contentassets/028b3d4ec3924cb393155c84784161ac/guidance-for-vaccine-and-pharmaceutical-logistics-and-distribution--extract.pdf (accessed October 14, 2021).
Ibrahim, I. A. 2021. Overview of export restrictions on COVID-19 vaccines and their components. American Society of International Law Insights 25(10).
International Airport Review. 2020. Frankfurt airport and Lufthansa well prepared for handling COVID-19 vaccine. https://www.internationalairportreview.com/news/125453/frankfurt-airport-handling-covid-19-vaccine (accessed June 15, 2021).
Iwu, C. J., A. Jaca, L. H. Abdullahi, N. J. Ngcobo, and C. S. Wiysonge. 2019. A scoping review of interventions for vaccine stock management in primary health-care facilities. Human Vaccines & Immunotherapeutics 15(11):2666–2672.
Jain, R., and E. Rocha. 2021. U.S. curbs on raw material exports could dent new Quad alliance’s vaccine push. Reuters, March 11. https://www.reuters.com/article/us-healthcoronavirus-vaccines-quad/u-s-curbs-on-raw-material-exports-could-dent-new-quadalliances-vaccine-push-idUSKBN2B31M1 (accessed October 12, 2021).
Kar, S. K., R. Ransing, S. M. Y. Arafat, and V. Menon. 2021. Second wave of COVID-19 pandemic in India: Barriers to effective governmental response. EClinicalMedicine 36.
Karp, C. L., D. Lans, J. Esparza, E. B. Edson, K. E. Owen, C. B. Wilson, P. M. Heaton, O. S. Levine, and R. Rao. 2015. Evaluating the value proposition for improving vaccine thermostability to increase vaccine impact in low and middle-income countries. Vaccine 33(30):3471–3479.
Kiernan, K. 2021. COVID-19 vaccine delivery: Can air cargo meet the challenge? Forbes, January 14.
Kraigsley, A. M., K. A. Moore, A. Bolster, M. Peters, D. Richardson, M. Arpey, M. Sonnenberger, M. McCarron, P. Lambach, H. C. Maltezou, and J. S. Bresee. 2021. Barriers and activities to implementing or expanding influenza vaccination programs in low- and middle-income countries: A global survey. Vaccine 39(25):3419–3427. https://doi.org/10.1016/j.vaccine.2021.04.043.
Kroger, A., L. Bahta, and P. Hunter. 2021. General best practice guidelines for immunization. Atlanta, GA: Centers for Disease Control and Prevention.
Layton, C., T. Robinson, and A. Honeycutt. 2005. Influenza vaccine demand: The chicken and the egg. Research Triangle Park, NC: Research Triangle Institute.
Lenert, L. A., W. Ding, and J. Jacobs. 2021. Informatics for public health and health system collaboration: Applications for the control of the current COVID-19 pandemic and the next one. Journal of the American Medical Informatics Association 28(8):1807–1811.
Li, Z., J. L. Swann, and P. Keskinocak. 2018. Value of inventory information in allocating a limited supply of influenza vaccine during a pandemic. PLoS ONE 13(10):e0206293.
McDonnell, A., S. Lloyd, K. Chalkidou, J. Li, D. Reader, R. V. Exan, and P. Yadav. 2020. Launching an interactive webtool to analyse the COVID-19 vaccine portfolio. https://www.cgdev.org/blog/covid-19-vaccine-predictions-tool-launch-blog (accessed August 27, 2021).
Moderna. 2020. Moderna announces longer shelf life for its COVID-19 vaccine candidate at refrigerated temperatures. Cambridge, MA.
Moutinho, S. 2021. Syringe size and supply issues continue to waste COVID-19 vaccine doses in United States. Science, March 26.
Mueller, L. E., L. A. Haidari, A. R. Wateska, R. J. Phillips, M. M. Schmitz, D. L. Connor, B. A. Norman, S. T. Brown, J. S. Welling, and B. Y. Lee. 2016. The impact of implementing a demand forecasting system into a low-income country’s supply chain. Vaccine 34(32):3663–3669.
Neuzil, K. M., J. S. Bresee, F. de la Hoz, K. Johansen, R. A. Karron, A. Krishnan, S. A. Madhi, P. Mangtani, D. J. Spiro, and J. R. Ortiz. 2017. Data and product needs for influenza immunization programs in low- and middle-income countries: Rationale and main conclusions of the WHO preferred product characteristics for next-generation influenza vaccines. Vaccine 35:5734–5737.
Newall, A. T., N. Chaiyakunapruk, P. Lambach, and R. C. W. Hutubessy. 2018. WHO guide on the economic evaluation of influenza vaccination. Influenza and Other Respiratory Viruses 12(2):211–219.
Ortiz, J. R., and K. M. Neuzil. 2019. Influenza immunization in low- and middle-income countries: Preparing for next-generation influenza vaccines. Journal of Infectious Diseases 219:S97–S106.
Ortiz, J. R., J. Robertson, J.-S. Hsu, S. L. Yu, A. J. Driscoll, S. R. Williams, W. H. Chen, M. C. Fitzpatrick, S. Sow, R. J. Biellik, and K. M. Neuzil. 2021. The potential effects of deploying SARS-CoV-2 vaccines on cold storage capacity and immunization workload in countries of the WHO African region. Vaccine 39(15):2165–2176.
Palache, A., S. Rockman, B. Taylor, M. Akcay, J. K. Billington, and P. Barbosa. 2021. Vaccine complacency and dose distribution inequities limit the benefits of seasonal influenza vaccination, despite a positive trend in use. Vaccine 39(41):6081–6087.
Parmar, D., E. M. Baruwa, P. Zuber, and S. Kone. 2010. Impact of wastage on single and multi-dose vaccine vials: Implications for introducing pneumococcal vaccines in developing countries. Human Vaccines 6(3):270–278.
PATH. 2018. First vaccine carrier approved by World Health Organization to prevent vaccine freezing during transport commercially available. Seattle, WA.
PATH and WHO (World Health Organization). 2013. A case for better immunization information systems. https://www.who.int/immunization/programmes_systems/supply_chain/optimize/better_immunization_information_systems.pdf?ua=1 (accessed October 14, 2021).
Pauwelyn, J. 2020. Export restrictions in times of pandemic: Options and limits under international trade agreements. Journal of World Trade 54:727–748.
Peters, R., and D. Prabhakar. 2021. Export restrictions do not help fight COVID-19. https://unctad.org/news/export-restrictions-do-not-help-fight-covid-19 (accessed August 19, 2021).
Pfizer. 2020. Pfizer-BioNTech COVID-19 vaccine U.S. distribution fact sheet. https://www.pfizer.com/news/hot-topics/covid_19_vaccine_u_s_distribution_fact_sheet (accessed June 9, 2021).
Pharmacy Today. 2021. Beware of errors when preparing Pfizer–BioNTech COVID-19 vaccine. Pharmacy Today 27(5):37.
Porter, R. M., S. Goldin, K. E. Lafond, L. Hedman, M. Ungkuldee, J. Kurzum, E. Azziz-Baumgartner, C. Nannei, J. S. Bresee, and A. Moen. 2020. Does having a seasonal influenza program facilitate pandemic preparedness? An analysis of vaccine deployment during the 2009 pandemic. Vaccine 38(5):1152–1159.
Ramakanth, D., S. Singh, P. K. Maji, Y. S. Lee, and K. K. Gaikwad. 2021. Advanced packaging for distribution and storage of COVID-19 vaccines: A review. Environmental Chemistry Letters 19:3597–3608.
Rastegar, M., M. Tavana, A. Meraj, and H. Mina. 2021. An inventory-location optimization model for equitable influenza vaccine distribution in developing countries during the COVID-19 pandemic. Vaccine 39(3):495–504.
Sellers, F. S. 2020. A shot. A wait. Another shot: Two-dose coronavirus vaccine regimens will make it harder to inoculate America. The Washington Post, November 27.
Solaiman, R. H. 2021. COVID-19 vaccination inequity in the United States: An intersectional issue. Journal of Health and Social Sciences 6(2):167–174.
Som, V. 2021. Indianvaccineexportbanmakes91nationsvulnerabletonewstrains:WHO. https://www.ndtv.com/india-news/indian-vaccine-export-ban-makes-91-nations-vulnerableto-new-strains-who-2453195?pfrom=home-ndtv_topscroll (accessed July 23, 2021).
Songane, M. 2018. Challenges for nationwide vaccine delivery in African countries. International Journal of Health Economics and Management 18(2):197–219.
Surgo Ventures. 2021. The U.S. COVID-19 vaccine coverage index. Press release. https://surgoventures.org/newsroom-all/new-the-us-covid-19-vaccine-coverage-index (accessed October 14, 2021).
Sustainable Energy for All. 2021. Chilling prospects: Tracking sustainable cooling for all 2021. https://www.seforall.org/chilling-prospects-2021 (accessed October 14, 2021).
UN (United Nations). 2020. COVID-19 vaccine: UNICEF to stockpile more than half a billion syringes by year’s end. UN News, October 19.
UNDP (United Nations Development Programme). 2021. Logistic management information systems (LMIS). https://www.undp-capacitydevelopment-health.org/en/capacities/focus/procurement-and-supply-chain-management/logistic-management-information-systems (accessed September 22, 2021).
UNEP (United National Environment Programme). 2020. Why optimized cold-chains could save a billion COVID vaccines. https://www.unep.org/news-and-stories/story/why-optimized-cold-chains-could-save-billion-covid-vaccines (accessed August 9, 2021).
UNICEF (United Nations Children’s Fund). 2021. The last mile: In-country vaccine delivery challenges. New York: UNICEF.
Vander Stichele, R. H., C. Hay, M. Fladvad, M. C. J. M. Sturkenboom, and R. T. Chen. 2021. How to ensure we can track and trace global use of COVID-19 vaccines? Vaccine 39(2):176–179.
VxDel. 2020. Vxdel data research: Kenya initial insights. John Snow, Inc., and Sonder Collective. https://www.sonderdesign.org/vxdel/VxDel_InittialKenyaReport_v4_2.pdf (accessed October 14, 2021).
Wainscott-Sargent, A. 2021. Georgia COVID-19 vaccine dashboard breaks down vaccination trends by race at county level. https://research.gatech.edu/georgia-covid-19-vaccine-dashboard-breaks-down-vaccination-trends-race-county-level (accessed August 22, 2021).
Wedlock, P. T., E. A. Mitgang, F. Elsheikh, J. Leonard, J. Bakal, J. Welling, J. Crawford, E. Assy, B. P. Magadzire, R. Bechtel, J. V. DePasse, S. S. Siegmund, S. T. Brown, and B. Y. Lee. 2019. The potential effects of introducing microneedle patch vaccines into routine vaccine supply chains. Vaccine 37(4):645–651.
Weintraub, R. L., L. Subramanian, A. Karlage, I. Ahmad, and J. Rosenberg. 2021. COVID-19 vaccine to vaccination: Why leaders must invest in delivery strategies now. Health Affairs (Millwood) 40(1):33–41.
Weise, E. 2021. “Vaccine angels” can only do so much: US struggles to make it easier to find COVID-19 vaccination appointments. USA Today, March 3, 2021. https://www.usatoday.com/story/news/health/2021/03/02/covid-19-vaccination-appointment-systemcoronavirus/6838918002 (accessed September 10, 2021).
White House. 2021. Background press call by senior administration officials on COVID-19 in India. Press briefing. https://www.whitehouse.gov/briefing-room/press-briefings/2021/04/26/background-press-call-by-senior-administration-officials-on-covid-19-inindia (accessed October 14, 2021).
WHO (World Health Organization). 2012. Assessing the programmatic suitability of vaccine candidates for WHO prequalification. Geneva, Switzerland: World Health Organization.
WHO. 2017. WHO preferred product characteristics for next-generation influenza vaccines. Geneva. https://www.who.int/publications/i/item/9789241512466 (accessed October 14, 2021).
WHO. 2020a. Ethics and COVID-19: Resource allocation and priority-setting. https://www.who.int/ethics/publications/ethics-covid-19-resource-allocation.pdf?ua=1 (accessed October 14, 2021).
WHO. 2020b. Framework for decision-making: Implementation of mass vaccination campaigns in the context of COVID-19. https://apps.who.int/iris/handle/10665/332159 (accessed October 14, 2021).
WHO. 2021a. PQS target product profile. https://extranet.who.int/pqweb/sites/default/files/documents/PQS%20Target%20Product%20Profile%20Definition-D.1.pdf (accessed July 7, 2021).
WHO. 2021b. Target product profiles. https://extranet.who.int/pqweb/immunization-devices/target-product-profiles (accessed July 7, 2021).
WHO Africa. 2021. Expiry date and shelf life of the Astrazeneca Covishield vaccine produced by the Serum Institute of India. https://www.afro.who.int/news/expiry-date-and-shelf-life-astrazeneca-covishield-vaccine-produced-serum-institute-india (accessed July 10).
WHO and UNICEF. 2016. Achieving immunization targets with the comprehensive effective vaccine management (EVM) framework. Geneva, Switzerland: World Health Organization.
WHO and UNICEF. 2021a. COVID-19 vaccination: Supply and logistics guidance. Geneva, Switzerland: World Health Organization. https://www.who.int/publications/i/item/who-2019-ncov-vaccine-deployment-logistics-2021-1 (accessed December 2, 2021).
WHO and UNICEF. 2021b. Effective vaccine management (EVM) global data analysis 2009-2020. Geneva, Switzerland: World Health Organization. https://cdn.who.int/media/docs/default-source/immunization/supply-chain/evm-global-data-analysis-2009-2020.pdf?sfvrsn=9113352_11 (accessed October 14, 2021).
WTO (World Trade Organization). 2020. Developing and delivering COVID-19 vaccines around the world. https://www.wto.org/english/tratop_e/covid19_e/vaccine_report_e.pdf (accessed October 14, 2021).
WTO. 2021. COVID-19 vaccine supply chain and regulatory technical symposium. https://youtu.be/RlvMoJGqoaI (accessed October 14, 2021).
Yih, W. K., G. M. Lee, T. A. Lieu, R. Ball, M. Kulldorff, M. Rett, P. M. Wahl, C. N. McMahill-Walraven, R. Platt, and D. A. Salmon. 2012. Surveillance for adverse events following receipt of pandemic 2009 H1N1 vaccine in the post-licensure rapid immunization safety monitoring (PRISM) system, 2009–2010. American Journal of Epidemiology 175(11):1120–1128.
Zaffran, M., J. Vandelaer, D. Kristensen, B. Melgaard, P. Yadav, K. O. Antwi-Agyei, and H. Lasher. 2013. The imperative for stronger vaccine supply and logistics systems. Vaccine 31(Suppl 2):B73–B80.
Zehrung, D., C. Jarrahian, B. Giersing, and D. Kristensen. 2017. Exploring new packaging and delivery options for the immunization supply chain. Vaccine 35(17):2265–2271.
Zipursky, S., M. H. Djingarey, J.-C. Lodjo, L. Olodo, S. Tiendrebeogo, and O. Ronveaux. 2014. Benefits of using vaccines out of the cold chain: Delivering meningitis A vaccine in a controlled temperature chain during the mass immunization campaign in Benin. Vaccine 32(13):1431–1435.
This page intentionally left blank.




































