3
Brown Tree Snake
NATURAL HISTORY
The brown tree snake, Boiga irregularis, like other members of the genus Boiga, is a slender, arboreal, nocturnal snake with grooved venom fangs at the posterior margin of the maxilla. It subdues prey by biting, constriction, and injection of a mildly toxic venom. It grows up to 3.1 m long on Guam (2.3 m in many parts of its native range), but the total length of most specimens encountered on Guam is less than 1.2 m. Juveniles constitute a greater proportion of the population on Guam than in the snake's native range, where juvenile mortality is likely to be much higher. This snake's ability to swallow exceptionally large prey items—up to 70% of the snake's body weight (Chiszar 1990)—suggests that its predatory capabilities are broad and therefore can affect prey species of a large range of sizes. Adult snakes can take birds the size of aga adults, but juvenile snakes might be restricted to eggs and small nestlings. Males attain greater sizes than females, so the largest males might be less frequently found in arboreal habitats.
Distribution
The species is extremely adaptable and occupies a wide range of tropical and subtropical environments among the hundreds of islands and continental locales within its native distribution. It has the most southern and eastern distribution of the 25 species of Boiga, which range from southwestern Asia through the Philippines and Indonesia, extending from Sulawesi in Indonesia through New Guinea
and the Solomon Islands and along a narrow subxeric to humid coastal areas of northern and eastern Australia as far south as Sydney. In many parts of its range in Australia, the brown tree snake is the most-abundant arboreal snake and indeed the most-proficient arboreal predators of birds (Shine 1991a). It tolerates disturbed habitats, forages both on the ground and in trees, and is a common inhabitant of human dwellings, where it can play a role as a predator of rodents.
Active at night, it seeks refuge from high temperatures and bright light during daylight hours in the forest canopy, in dense vegetation, and even on the forest floor under logs and other structures, in limestone crevices, and in a multitude of sites made by humans, including buildings, vehicles, and shipping containers. The use of the latter daytime refuges places the brown tree snake in contact with cargo moving from Guam to other geographic areas. The activity of the snake is influenced by both humidity and temperature in the most-temperate parts of its range, but in moist tropical regions, it is active around the year and might have a nonseasonal reproductive cycle. Snakes are active year-round, but differences in detectability, frequency of power outages due to snakes, and frequency of snakebites exhibit seasonal components. On Guam, snake activity seems to be greatest at the transition from the dry to the wet season (May and June) and lowest during extremely dry seasons (Fritts 1988).
Diet
The brown tree snake has an extremely broad dietary range, including frogs, lizards, other snakes, birds, rodents, and bird eggs. On Guam, it is known to consume lizards, birds, eggs, insects, mammals, and human refuse, including picnic scraps, chicken bones, and even meat wrappers, although such scavenging might involve only snakes that are starving. Lizards constitute the most common prey of snakes on Guam and of juvenile and subadult snakes in the native range; larger snakes tend to take proportionally more birds and mammals than smaller snakes do (Greene 1989; Savidge 1987; Shine 1991a). On Guam, lizards predominate in analyses of stomach contents in all sizes of snakes (Rodda and others in press c; Savidge 1988); both terrestrial diurnal species and arboreal nocturnal species occur in the diet. That pattern suggests that snakes on Guam undergo an active search for sleeping prey and ambush foraging for active prey. The dietary flexibility of the brown tree snake contributes to its ability to remain abundant even after large segments of the prey base dropped precipitously. This characteristic explains why the brown tree snake has been able essentially to extirpate most of an island's avifauna and still remain relatively abundant.
Predatory Mechanisms
To find prey, the brown tree snake depends on its acute night vision when visual stimuli are present, but uses chemical cues (especially nonvolatile molecules)
when deprived of visual cues (Chiszar and others 1988; Kardong and Smith 1991). Its abilities to use volatile cues and heat as predatory cues remain to be determined. When active, the snake moves slowly in the vegetation or on the ground and responds to both prey and potential threats that it detects visually. When closely approached by a human or illuminated by a strong light, it temporarily freezes and then flees within 10–60 seconds if the stimulus is not removed. There is evidence that the snake alters its foraging strategies, moving from forest canopy to the forest floor or cleared road sides, as arboreal lizard prey is depleted (Rodda and others in press a). Thus, in some situations when snakes are found on the ground, the number of snakes is inversely correlated with the abundance of lizards detected in the foliage, and the proportion of arboreal lizards in the snakes' stomachs is lower than that in snakes collected from trees or in habitats where arboreal lizards are more abundant. Whether snakes are able to assess prey availability on the basis of chemical cues or are merely foraging in habitats on the basis of proximal capture success and search images is unknown.
Field studies in the brown tree snake's native range strongly suggest that hatchling and juvenile survivorship is low because of the lack of prey small enough for it (Rodda and others in press c). Such reduced recruitment into the adult population thereby limits population densities of the snake in its native range. In contrast, oceanic islands (such as Guam and other Mariana islands) have higher numbers of native and introduced species of lizards that are small enough to be suitable as prey for juvenile snakes. These islands might therefore be able to support high populations of snakes if snakes are introduced. Also, small birds poorly equipped with antipredator defenses against snakes were prevalent on Guam and undoubtedly contributed to the buildup of snake populations there; indeed, the smallest and most-abundant birds on Guam disappeared first as a result of snake predation (Aguon 1983; Engbring and Fritts 1988; Pratt and others 1979). Malaita and the Solomon Islands are examples of the disparity of snake densities in native range versus oceanic islands. The same predators—mammals, birds, or reptiles—of the snake on Malaita are present on Guam. The average sighting rate for brown tree snakes on Maliata was 5% of the average for the same searchers on Guam (Rodda and others in press c). Shine (1991b) concluded that neither habitat structure nor predation was a limiting factor for other snakes; Rodda and others (in press c) conducted statistical analyses of faunal similarities that supported this conclusion.
In Australia and New Guinea, some antipredator defenses might partially protect birds from snake predation, but no specific research has been done on this. However, the lower populations of snakes in their native range (limited by availability of food for juveniles) than on Guam could be the primary factor in reducing risk of predation on bird populations. Armstrong and Pyke (1991) pointed to the lack of correlation between successful nesting efforts of a nectivorous bird in Australia and the peak of nectar resources. They concluded that nesting success was related to reduced activity of the brown tree snake during the coolest months.
Nesting failure during warmer months was related to egg and nestling predation and to a lesser extent to nest abandonment presumably because of attack by predators. In the warmer months, nestlings disappeared between dusk and dawn; the disappearance rate was halved during the coolest months, when the brown tree snake was least active (Armstrong and Pyke 1991). Bell (1966) observed similar patterns of nest predation, presumably by reptiles—with decreased nestling loss during cooler months and increased predation in warmer months—in an area inhabited by the brown tree snake.
Most studies of nest failure in birds underestimate the potential predatory impact of snakes. Snakes can engulf eggs and young and leave no trace of the predatory act; therefore losses may be higher. For example, Ricklefs (1969) recorded nearly half as many losses of yellow-headed blackbird eggs because of disappearance from nests for unknown reasons (42) as because of known predation (102) and almost equal numbers of disappearances of young for unknown reasons (22) and known predation (21). Snake (or bird) predation could have been a major factor in the disappearance of both eggs and young; if so, nest predation for this data set was more than 50% higher than reported by Ricklefs (1969).
EFFECTS OF THE PRESENCE OF THE BROWN TREE SNAKE ON GUAM
The brown tree snake was first reported in Guam in 1953, but reports of unidentified snakes in the vicinity of the military port in preceding years suggest that it arrived and became established in the late 1940s. The snake is likely to have arrived in military surplus equipment shipped to and through Guam for the Lend Lease Program in China, salvage and smelting, or return to the US mainland. Structural variation in body color and markings throughout the snake's native range provides a basis for establishing the source population as that on the Admiralty Islands (Rodda and others 1992), which coincides with the location of the large aggregation of Allied troops and material on Manus, the Pacific command headquarters before the capture of Guam in 1944.
A large volume of military traffic and cargo flowed from the Admiralties to Guam, and so too moved the snakes that colonized Guam. Within a decade after the species' arrival on Guam, large brown tree snakes were increasingly encountered by residents near the harbor and central Guam; by the 1960s, bird populations in the central part of the island began to disappear (Rodda and Fritts 1992; Savidge 1987). In Guam's environment of abundant lizards, birds, and introduced rodents, the snake thrived and attained large populations; by 1969, the species had spread throughout the island, even though the birds of northern Guam did not decline noticeably until more than 10 years later (Rodda and others 1992; Savidge 1987).
On the basis of patterns of the disappearance of Guam's avifauna and the conspicuousness of snakes, peaks in the eruption of the snake population moved
progressively toward the north end of Guam until the 1980s, when most of the remnants of Guam's bird populations disappeared or were most severely depressed (Fritts 1988; Savidge 1987). Savidge demonstrated high predation rates using several species of birds in snake traps spaced in forest areas of Guam where native birds were declining. Snake populations declined by as much as 50% within 3 years after the primary elements of Guam's forest bird fauna disappeared from northern Guam (Rodda and others 1992). Predation on aga eggs by the brown tree snakes has been observed (Campbell 1996b), and the loss of nestlings and adults has been inferred from disappearances from nests and territories studied by personnel of the Guam Department of Agriculture Division of Aquatic and Wildlife Resources (DAWR).
Since the arrival of the snake on Guam, most of Guam's indigenous forest vertebrates have been extirpated. Too-few baseline data are available to reveal unequivocally the degree to which the snake was responsible for those losses, but, although the exact sequence of extirpations and their causes are incompletely known, several lines of evidence point to the snake as the primary cause of the loss of many of Guam's birds and bats and several lizards species (Rodda and others in press a). Guam's 3 bat species (Wiles 1987) might have been prey for the snake, but data on 2 bats that disappeared before 1980 are lacking, and human hunting has obscured the cause of the decline of the remaining species (Pteropus marianamus) and its failure to recover. However, the snake appears to prey on young bats, inhibiting recruitment into an otherwise-protected population (Wiles 1987).
Evidence that the snake played a major role in the extirpation of the vertebrates of Guam (Rodda and others in press a) includes geographic patterns of bird and lizard losses concordant with population eruptions of the snake; predation of snakes on the vertebrates that declined, including lizards, birds, and mammals; lack of indications of substantial habitat loss, disease, pesticide contamination, or other causal factors; vulnerability of, and recorded impact on, both native and introduced species; unprecedently high populations of the snake coinciding with the disappearance of the avifauna; reductions in snake populations on northern Guam after the birds disappeared; and lack of comparable extirpations on nearby islands and islets where the snake is not present or conspicuous.
Estimates of snake density suggest unprecedented numbers of snakes on Guam (Rodda and others 1992, in press a). Snake densities in 1985, when the native forest birds of northern Guam disappeared or were reduced to only a few individuals, are estimated to have been about 100 per hectare (Rodda and others 1992). That estimate is based on trapping results and the calculated abundance of snakes in night surveys relative to results of work from 1987 through 1992, when snake populations were judged to have dropped to 35–50 snakes/ha—still 10 times greater than in its native range (Rodda 1996)—after the virtual collapse of most bird populations and the depression of rat, shrew, and some lizard populations. That the populations of the snake were exceptionally large and remain relatively
large can be inferred from the frequency of power outages on Guam attributable to snakes' coming into contact with high-voltage conductors (Fritts and others 1987); the frequency with which snakes, potentially motivated by hunger, invaded homes and attacked sleeping humans (Fritts and others 1994); and the high incidence of snakes discovered in extraliminal situations as a result of surfaceship and air traffic from Guam (Fritts 1987; Fritts and others in press).
Population estimates for snakes at individual locales have varied from 1988 through 1995; both increases and decreases have been noted, but the mean population estimates aggregate in the range of 35–58 snakes/ha. That the brown tree snake is not likely to exhaust its lizard prey base is suggested by 1995 studies of densities in forested areas (Campbell 1996a)—although there were 36/ha more, there were nearly 400 lizards and one rat for each snake. Thus, there is no scarcity of small prey, although the scarcity of moderate-size to large prey in many situations on Guam could place dietary limits on large snakes. The brown tree snake can survive (or even thrive) on a diet of small lizards and reach reproductive size (980 mm snout-to-vent length), thereby ensuring continuous recruitment into the breeding population.
Although large snakes can subsist on small prey, they might alter their foraging patterns in search of prey of more suitable size (by foraging over larger distances and using habitats where endotherms remain). Evidence suggests that snakes prolong foraging and movements into the early daytime hours: more electric outages have occurred in morning hours (6 AM to noon) since 1985 than occurred previously, when more than 95% of outages caused by snakes occurred at night (6 PM to 6 AM). The increase in morning activity without a comparable increase in afternoon activity suggests that snakes are prolonging nighttime activity rather than merely becoming more diurnal (Fritts and Chiszar in press). The dependence of large snakes (over 2 m long) on endothermic prey is evident in the scarcity of large snakes in forested areas and their prominence in areas around ranches and residential areas, where poultry, rodents, and introduced birds persist.
Thus, the decline of endotherms in native forests might have affected the size structure of the snake population without lessening the predatory pressure on the remaining endotherms, especially such forest inhabitants as the aga, the Mariana fruit bat, and the introduced black drongo and Philippine turtle dove. An incident in which a relatively small snake was discovered leaving an aga nest with an egg in its mouth suggests that three-quarters of male and more than half of female snakes are large enough to consume crow eggs and that the predatory pressure is high on nests in situations where snake densities are 30–50 snakes/ha. The density of snakes might have fallen more in natural habitats than in disturbed ones close to development: It is these disturbed habitats that produce snakes most likely to enter the transportation network, interrupt electric transmissions, and attack humans.
DECLINE OF BIRDS ON GUAM IN RELATION TO THE BROWN TREE SNAKE
Of the 22 native birds known to be resident on Guam, 13 have been extirpated (including 2 endemic species and 3 endemic subspecies) solely or primarily because of snake predation and related factors. Two endemic taxa (a rail and a kingfisher) have been extirpated from Guam but exist in captive situations. A few aga are also in captivity.
Four bird taxa are presumed to have disappeared before the arrival of the brown tree snake; at least, their absence cannot be directly linked to snake predation (Engbring and Fritts 1988). They include a single forest bird (megapode), which disappeared before the turn of the 20th century, and 3 oceanic or wetland birds (shearwater, Mariana mallard, and white-browed crake). Nine native forest birds apparently disappeared because of the snake. Another 3 oceanic birds (tern, noddy, and tropicbird) listed by Engbring and Fritts (1988) are biologically extirpated (infrequent sightings over the last decade involve birds arriving from Cocos or other islands); all formerly bred on Guam and no longer do so.
The 3 surviving native forest birds (aga, swiftlet, and starling) that remain on Guam occur in extremely reduced populations; of the 3 extant wetland birds that remain (bittern, reef heron, and moorhen), only the bittern appears common.
The aga is presumed to number between 20 and 40 in 1996. The swiftlet occurs as a regular inhabitant of a single cave, but recent estimates of swiftlet numbers suggest a decline from 3 years ago. An additional colony of fewer than 50 birds might exist periodically. A third colony is rumored to exist, but its size and location are unknown.
Assessing the impact of snake predation with indirect evidence on the 8 introduced species is more difficult because it is less evident what the densities and distribution of these species would be without snake predation. Over the last decade, all introduced species have been reduced, but some have periodically shown signs of population increase of succeeded in special situations, and that poses a dilemma in assessing their role as prey for the snake. Introduced species surviving on Guam could provide crucial information on methods of protecting native species, but of the introduced taxa only the black drongo (Maben, 1982) and Philippine turtle dove (Conry 1988) have been studied.
At present, introduced species are the only birds seen with any frequency on Guam, and even these are infrequent enough to be noteworthy for the lay observer. Four of the 8 introduced species of birds are reduced in numbers. The Eurasian tree sparrow is found primarily in urban, commercial, and roadside situations but has not successfully invaded forests. The black drongo and the Philippine turtle dove are conspicuous birds, but drongos are substantially reduced on the Northwest Field of Andersen Air Force Base in comparison with densities in 1985. Both drongos and turtle doves are present in low numbers over large areas of Guam, but they are obviously rather than they would be if snake predation were
not high (Conry 1988). Other introduced species are restricted in numbers and are found only in specialized situations affording partial protection from snake predation. For example, the francolin, mannikin, and quail are all typically grassland inhabitants, and the rock dove, which is an urban dweller, is extremely rare. Mannikins are infrequently encountered but only in grassy habitats. The status of the introduced coturnix quail is unknown; Engbring and Fritts (1988) considered it to be common. The status of feral populations of the jungle fowl is uncertain because of the prevalence of free-ranging chickens associated with ranch and suburban settlements, but, in direct contrast with other islands where wild populations of jungle fowl persist, it is likely that the jungle fowl is extinct on Guam except for recent escapees.
In summary, 4 native species are gone, and their disappearance cannot be attributed to the snake. However, 12 species probably have been extirpated by snake predation, and 6 are extant, but only one (the yellow bittern) is considered common. Snake predation has probably reduced populations of at least half the introduced species; grassland and urban species remain more abundant than forest dwellers. Of the introduced birds, only the Philippine turtle dove, black francolin, and Eurasian tree sparrow can be judged common.
It is perhaps notable that of 21 avian taxa endemic to the Marianas listed by Pratt and others (1979), 10 have disappeared from Guam and only 4 (nightingale reed warblers [Acrocephalus luscinia nijoi and A. luscinia yamashinae], collared kingfisher, and cardinal honeyeater) are now found north of Saipan. Another 11 taxa will be lost if the brown tree snake eliminates the forms now limited to the southern Northern Mariana Islands (Rota, Agiguan, Tinian, and Saipan). The threat of the snake's colonizing Saipan, Tinian, and Rota is related to increasing surface and air traffic between Guam and these islands. Agiguan is uninhabited and relatively inaccessible to humans, but its small size would increase the vulnerability of its bird fauna if the island were colonized by snakes.
EFFORTS TO CONTROL THE BROWN TREE SNAKE ON GUAM
The recognition that the brown tree snake was causing serious problems on Guam led to the development of means to control it. No easy solutions were apparent, nor was one agency or government solely responsible for leading or funding snake-control efforts. This section reviews the types of control methods and their effectiveness.
Trapping
Biological research by DAWR, the National Biological Service (NBS), and the US Department of Agriculture Animal Damage Control (ADC) led to the development of an effective snake-trapping protocol. Since its development, trapping has been the primary control strategy. DAWR biologists have trapped hundreds
of snakes in the vicinity of endangered-bird habitats. The ADC program has trapped thousands of snakes annually (for example, 6,134 were trapped in 1995) primarily in or around transportation facilities (ports and airports). However, even a large number like the 1995 yield would theoretically account for the population of snakes from an area of only 123 ha (assuming 50 snakes/ha in forested habitats), but this same number applied to thousands of hectares represents only a small fraction of the snake population and is of negligible ecological benefit. Traps might be appropriate in small areas but are inadequate by themselves for larger areas because of manpower limitations and large populations and seasonal mobility of snakes.
Nest Protection with Barriers on Trees
Aguon and others (in press) have protected individual nests of aga by encircling the trunks of the nest trees with electric barriers (Figure 3-1) and pruning canopies of adjacent trees and vegetation to prevent snakes from crossing into the nest trees via canopies. Initiating control only after identification of an active nesting attempt can lead to human disturbance of nest-building, egg-laying, and incubation; however, DAWR staff report that aga have renested in trees that were protected by barriers the previous breeding season suggesting that the human disturbance of emplacing the barriers might not cause abandonment of the nest. Such a risk must be considered in decisions as to how much trimming is needed to separate canopies and the extent of active trapping to remove snakes that might already be present in the canopies.
Such protection may not be adequate in itself to allow recovery of the aga in the face of large populations of snakes. Despite increasing egg protection, fledging success has been poor—possibly not because barriers on trees failed but potentially because canopy separation was inadequate, especially during windy periods, or because snakes remained in the trees despite efforts to trap them. Conclusive data are lacking, and few hypotheses have been tested adequately, because there are so few nests. Isolation of crows from snakes must be increased to increase survival of crow eggs, nestlings, fledglings, young birds, and adults.
Snake Exclosures
The only adequate devices for controlling snake populations in natural areas are experimental snake exclosures (area barriers) constructed in a cooperative effort involving NBS and Ohio State University (Figure 3-2). An 18-month field study demonstrated the efficacy of a snake exclosure; snakes were removed from the exclosure by trapping and hand capture. Lizards, snakes, and birds were monitored in exclosures and in comparable sham plots from which snakes were not removed. Results detailed by Campbell (1996) suggested that exclosures facilitated removal of snakes from 2 1-ha plots (in 17 and 78 days) and allowed
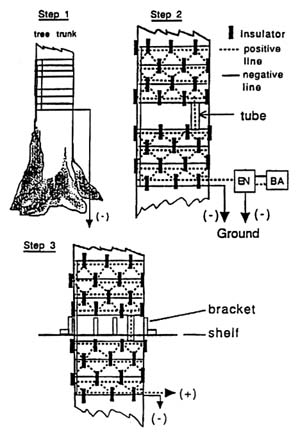
FIGURE 3-1 Electric barrier. 1) Galvanized cable wrapped around tree trunk. Steel rod inserted into ground forming negative line. 2) Insulators nailed to trunk in staggered pattern. Separate length of cable placed on insulators forming positive line. 3) Positive line connected to electric fence energizer (EN) powered by 12v DC lead acid battery (BA). Metal shelf attached to tree trunk between upper and lower electrified sections. Source: Aguon and others, in press.
maintenance of effectively snake-free plots; about 5% of the number of snakes recorded in comparable habitats without controls were recorded in plots during the experiment, and a substantial increase in lizard abundance was evident in snake exclosures relative to that in plots with snakes. Problems of maintaining such exclosures were identified: damage by rats and large mammals, plant control, and the potential for typhoon damage. A second phase of exclosure design and testing with materials resistant to those problems was initiated in a laboratory setting where snake performance was monitored with an infrared-sensitive video
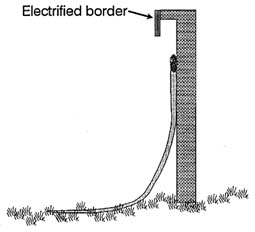
FIGURE 3-2 Snake exclosure barrier. Side view of concrete barrier wall. Source: committee generated.
recorder. New exclosure designs incorporating the following features were considered:
-
Strength to resist or ability to shed wind loads during periods of high wind velocity;
-
Surfaces smooth enough to discourage climbing; and
-
Ledges, overhangs, or electric skirts to discourage overreaching (especially by large snakes).
One exclosure design, which uses masonry materials with a height of 1.15 m and an overhanging ledge fringed by an electrified metal strip 6 cm wide, has withstood over 1,000 attempts to enter by snakes of various sizes without being breached (Figure 3-2). Even without electrification of the 6-cm metal strip, 96% of snakes were unable to cross the same exclosure in hundreds of trials. An alternative design involving retrofitting of existing Cyclone fences is being tested and developed (Campbell 1996b).
Rodda and others (in press b) probed the critical elements of controlling snakes in small plots. Recent results suggest that snake exclosures (1–50 ha) applicable to conditions on Guam are feasible. Testing of larger-scale applications of exclosures under outdoor conditions is planned, and engineering-fabrication suppliers are involved in estimating costs of prototypes and applications.
As envisioned by Campbell (1996), snake exclosures have applications in a variety of contexts:
-
to prevent snake from entering high-priority locations, such as endangered species habitats (to protect all species present), cargo storage or dispatch areas (to reduce export of snakes), electric substations and generating facilities (to reduce power outages), and residential neighborhoods and poultry farms (to protect humans and domesticated animals);
-
to facilitate detection, capture, and containment of snakes arriving at extraliminal sites in cargo (to interdict snakes) by constructing enclosure barriers that prevent snakes from leaving an area; and
-
and to deflect movements of snakes toward traps or bait stations (to enhance control efficacy), especially in destinations judged to be at risk.
PROGNOSIS FOR CONTROL ON GUAM
The ability to control snake populations throughout Guam depends on the future development of the control tools identified in the integrated pest-management plan (BTSCC 1996; Campbell and others in press), which integrates large-scale control (such as biological or toxicologic methods) with other tools available now (trapping, hand capture, habitat management, repellents, fumigants, barriers, exclosures, and so on). Biological control and toxicants are not likely to be available within 3–5 years, and even then only if funding called for in the Aquatic Nuisance Species Task Force's Brown Tree Snake Control Plan (BTSCC 1996) is continued or expanded. However, the application of combinations of exclosures, barriers, traps, and other available techniques could be initiated in the near future to protect sites that have high priority for conserving endangered species. The size, location, and intensity of management of such sites should be determined carefully. Protection of areas, rather than solitary nest sites, has an added benefit of enhancing viability of several species, instead of a single taxon of special interest. In as much as eradication of snakes on Guam is distant or even unattainable, management of smaller areas offers the only in situ method for protection of endangered species. Exclosure barriers—whether physical, ecological, or geographic—will be important in reducing the required effort and increasing the efficacy of localized control programs.
THREAT OF SPREAD OF THE BROWN TREE SNAKE TO OTHER ISLANDS
In addition to Guam, the brown tree snake has been reported on 10 other islands—including islands in the Northern Marianas, the Federated States of Micronesia, the Ryukyu Islands, Diego Garcia Atoll, the state of Hawaii, and Wake Island—as a result of dispersal from Guam (Fritts and others in press). A single record exists of a brown tree snake's arrival in Corpus Christi, TX, in a shipment of military household goods from Guam (McCoid and others 1994). Such sightings involve snakes in civilian and military transportation systems,
ships, and air traffic from Guam. Seven brown tree snakes have been verified in Hawaii (1981—1994); however, on the basis of captures of snakes and reports that did not result in captures, Saipan, Rota, and Tinian in CNMI are at greatest risk for infestation because of their proximity to Guam and the volume of cargo transported from and through Guam to these destinations. Since 1985, more than 30 sightings of snakes on Saipan, an island formerly lacking snakes, have been reported.
The likelihood of dispersal of the brown tree snakes as passive stowaways in ship and air traffic from Guam is increased by the central position of Guam in the transportation system of the western Pacific (especially for American flag islands). Increasing traffic volume is a direct result of commercial activity in the region. The incidence of brown tree snakes in cargo from Guam is also a result of the extremely high snake populations on Guam and the snakes' tolerance for disturbed habitats, including commercial, residential, and industrial sites from which cargo is shipped. The brown tree snake avoids high temperatures and bright sunlight by seeking daytime refugia. Vehicles, containers, construction materials, and other cargo furnish ample hiding spots and result in dispersal to and through ports and airports. Snakes can remain inactive and hidden and can survive without food for long periods. For example, the snake that crawled from household goods in Texas might have been in transit in a shipping container for up to 6 months.
As a native of hundreds of Pacific islands and a successful colonist of Guam, the brown tree snake is likely to succeed in establishing populations on other islands with similar environments if enough individuals arrive. As a habitat generalist and dietary opportunist, it might reach pest levels elsewhere in the Northern Mariana chain, elsewhere in Micronesia, in the state of Hawaii, and beyond unless dispersal from Guam is reduced, dispersing snakes are intercepted on arrival, and incipient populations are eradicated before they are widespread and firmly established. The environmental consequences on the Pacific islands that the snake has colonized might develop faster than on Guam because most of the islands are smaller than Guam. In many cases, the islands at risk hold some of the same—or at least closely related—bird and reptile taxa that were lost from Guam. Further losses would constitute a heightened threat to the biodiversity of the region. The long-term consequences of major disruption of entire bird, bat, and lizard faunas on Guam and other islands are poorly documented, but these organisms played ecological roles as insect predators, pollinators, seed dispersers, and consumers of fruit that are now only partially filled by introduced species and invertebrate organisms.
The brown tree snake constitutes a threat to the survival and conservation of the aga on two levels:
-
predation on crows and interference with recruitment in the extremely reduced population on Guam, and
-
risk of continued dispersal from Guam to other western Pacific islands, including Rota, where establishment and eruption of brown tree snakes would threaten the survival of the only other population of the aga.
Colonization of Rota would effectively nullify all management programs to protect and preserve the aga and its habitat. Approaches that will be necessary to address those two facets of the snake problem might well differ.
Efforts to Prevent Spread of the Brown Tree Snake to Other Islands
The documented dispersal of snakes from Guam to other islands and parts of the world (Fritts 1987; Fritts and others in press) resulted in the initiation of the ADC program to control snakes in the transportation system on Guam. Great strides have been made in and around ports and airports in implementing trapping, visual searches, canine inspections, and, to a lesser extent, management of habitats, but the enormity and complexity of the task remain evident. Recent discoveries (in November and December 1995) of snakes on ships and in cargo on Saipan and Pohnpei highlight the need to expand and enhance control efforts in the overall transportation network on Guam and on islands at risk of receiving snakes. Snake control focused on transportation from Guam is not complete: problems include limited ADC personnel on Guam, relying on voluntary compliance with ADC snake searches, periodic ship departures with no advance notice, and the complex dynamics of moving commercial, private, and military cargo in a heavily used port and airport system.














