CHAPTER 40
ARE THERE ALTERNATIVES TO DESTRUCTION?
MICHAEL H.ROBINSON
Director, National Zoological Park, Washington, D.C.
Any consideration of the forces tending to produce drastic reductions in biodiversity must concentrate on habitat destruction. To slow down, ameliorate, or prevent habitat destruction we need to understand the forces that cause it. There is a widespread assumption that habitat destruction, particularly in the Third World, is caused by ignorance or stupidity or both. This assumption is reflected in the philanthropic funding of environmental education programs and in attempts to seed environmental defense organizations in Africa, Latin America, and Asia. It is often assumed that these efforts are constructive. The contrary viewpoint is that environmental destruction results from economic pressures that have nothing to do with stupidity or ignorance. It is certainly arguable that given the present international distribution of wealth, the mainly tropical less-developed countries will be forced to exploit their natural resources on a massive scale in order to try to raise living standards quickly. A discussion of whether they can do this is beyond the scope of this chapter and beyond my competence; the majority of the countries involved are certainly trying.
In this volume are a number of analyses of the causes of habitat destruction and the economic realities facing the Third World. Most tropical deforestation, and with it the major threat to biological diversity, comes from efforts to increase the level of subsistence and to generate foreign exchange for the purchase of goods manufactured in the developed world. To halt destruction we must find alternative means of providing subsistence goods (food, fuel, and construction materials) and alternative commodities to replace those resulting from environmental rape. There is a third logical possibility that is almost certainly not a real one. That is for the entire world to adopt voluntary restraints on resource exploitation and living standards. This is the pathway of so-called green politics and the small-is-beautiful
concept. On this there seems to be unanimity of opposition from all conventional political movements from the capitalist right to the Marxist left.
I would contend that the discovery of alternatives to destruction can come from the results of purely basic (i.e., nonapplied) studies. I have developed this theme in detail elsewhere (Robinson, 1985, 1986). In Chapter 42 of this volume, Rubinoff deals in detail with the scheme that we developed in Panama—a scheme based on the principle that native species of plants and animals should be considered for domestication and cultivation, as opposed to the use of exotics introduced at the behest of the developed world. In Panama, the animal studies include the green iguana (Iguana iguana), a large folivorous lizard, and the paca (Cuniculus paca), a caviomorph rodent widely esteemed for its succulence. Both these animals had been the subject of numerous academic studies before being considered as potential alternatives to destruction. The National Research Council’s Board on Science and Technology for International Development, on the other hand, has deliberately set out to search for animals that have a high potential for domestication or husbandry (NRC, 1983). This approach was thus project-oriented from the start. Both approaches are valid and potentially productive. The purist approach has some exciting implications. Tropical regions are often the scene of highly coadapted/coevolved communities of plants and animals. It would seem that these communities should, a priori, be the place to search for exploitable bioresources that can be used by humans. They can be regarded as having a high potential for containing species that are preadapted for human use.
As an example of this we can briefly consider the paca. This rodent lives on the floor of rain forests in Central and South America. Its activity is nocturnal. These facts set the scene for its potential utility. Because it lives on the forest floor, does not climb trees, and is a rodent, it has a proscribed food source. It feeds on fruits that fall to the ground from the trees and to a much lesser extent on roots and seedlings. It utilizes the secondary products of the forest that are unavailable to humans and most grazing animals (exotic or indigenous). Fruitfall in the humid tropics is sporadic, but the paca subsists on an intermittent food supply by scatter hoarding during times of plenty (intermittent fruitfall may be an interspecific adaptation to ensure dispersal; Smythe, 1970). Thus pacas can prosper without destroying forest. They are almost tailor-made for domestication. Being nocturnal they do not need the capacity to run long distances to escape predators. They can be fat, unlike their diurnal complementary species the agouti (Dasyprocta spp.). Their disadvantages are a low reproductive rate, small families, and solitary social life.
In other parts of the world, there have been similar adaptations in response to similar environmental and biological pressures. The bearded pig (Sus barbatus), found in Borneo, Java, and the Philippines, is a forest floor fruit eater, grows to a substantial size, and copes with intermittent or seasonal fruit supplies by migration from area to area. It is clearly a candidate for exploitation within the forest (NRC, 1983; Robinson, 1986). There are similar adapted animals in Australasia, despite the absence of large placental animals there. The tree kangaroo (Dendrolagus spp.) is a leaf-eating arboreal animal that might prove to be as suitable for domestication as the iguana. In Malaysia and throughout the Indian subcontinent and Southeast
Asia there are giant squirrels, and so on. As we contemplate the mass extinction of tropical animals, we can speculate that if it were not for a dramatic historical extinction we might be considering the dodo (Raphus cuculatus) as a candidate for domestication and transtropical exploitation. This giant (25-kilogram) pigeon was flightless and probably subsisted on fruits, just like the paca! Just as the dispersal strategies of some Neotropical trees seem to have coevolved with the pacas, so, it seems, have some of the Mauritian fruits coevolved with the dodo. Temple (1977) suggested that the hard, nutlike seed of Calvaria major depended for its germination on its abrasion in the gizzard of the dodos.
Janzen and Martin (1982) suggested that at least 30 species of trees in Costa Rica depended for their dispersal on the digestive processes of some now extinct, large (sometimes giant), herbivores of the Neotropics. These, like the dodo, were probably eminently edible. Similarly, the giant ground sloths of South America and the giant birds of New Zealand, known from historic times, could have been candidates for domestication. This is not fruitless speculation but a notice that our regrets about these extinctions are certainly likely to be matched soon by more massive regrets about the ongoing and imminent extinctions.
Our knowledge of plants with a high potential for future exploitation is very sketchy indeed. The Board on Science and Technology for International Development within the National Research Council has highlighted some examples (NRC, 1975, 1979, 1981, 1984, 1985). In Chapter 10 of this volume, Iltis has drawn attention to some other dramatic examples. Again on a priori grounds we can predict that there must be many plants that have or are in themselves valuable, undiscovered products.
The tropical forests of the world are the most intense battlegrounds for species competition on the face of the Earth. This intensity is a direct result of extraordinary biodiversity and the continuingly ferocious evolutionary arms race. Huge assemblages of animal species eat leaves, and it is no exaggeration to say that tropical plants must have evolved a formidable array of insecticidal compounds and insect deterrents. We already use some of these. The picture is similar in the case of fungicides. The moist tropical forest is a superb environment for fungi of all kinds. Fungi relish warmth, wetness, and an abundance of organic substrates. The leaves of tropical trees are not only assaulted by insects and other animals but also by fungi and epiphyllic plants. What a place to look for fungicides and herbicides!
An illustration of how fundamental studies lead to discoveries in this field comes from a report of the food preferences of leaf-cutter ants by Hubbell et al. (1983). They found that leaf-cutter ants (Atta sp.) rejected the leaves of the leguminous tree Hymenaea courbaril. Such leaves kill the fungus that the ants grow in their nests as a food resource. It is entirely improbable that the tree evolved the fungicide to deter leaf-cutter ants. Rather, it probably had to cope with a broad spectrum of epiphyllic fungi and evolved a broad-spectrum fungicide to (literally) keep its leaves clean. Laboratory tests on the fungicide, a terpene-caryophyllene epoxide, show antifungal activity against a wide range of pathogenic fungi. This precisely illustrates the points I make above. It surely means that we can confidently predict, because of the nature of tropical forest ecology, that its plants should be a source of a wide range of useful products. These await discovery. There is little doubt that
among them are further alternatives to destruction. In terms of food sources we already exploit tropical plants as sources of oils, fats, carbohydrates (sugars as well as starches), and proteins, but the current number of species that we use for food is very small. Discovering potential forest crops is going to require a massive research effort, but we can expect some shortcuts. These are outlined below.
First, we have a great potential source of insights in ethnobotany and ethno-zoology. Populations of humans throughout the world have existed in essentially harmonious relationships with tropical rain forests for centuries. Many of these relationships involved sustained yield subsistence use of the forests, and the Mayans produced a surplus of foodstuffs without destroying vast areas of forest. There are thus a wide range of peoples with invaluable knowledge about plants and animals. This knowledge may not be expressed in the terminology of modern science; it may be interlaced with magic, myth, and superstition, but it is certainly extractable. Just as certainly, its extraction is a matter of extreme urgency, since the folkways of forest peoples are disappearing more rapidly than the plants and animals that they have learned to exploit. In Part 2 of this volume, Nations, Farnsworth, Iltis, and Plotkin address various aspects of our need to utilize traditional knowledge—knowledge that is threatened and fragile. It is not merely knowledge of species but also knowledge of practices of husbandry, gardening, and agriculture. In Chapter 41, Altieri argues persuasively for the modern applications of ancient systems of mixed cropping as means of ex situ preservation of diversity. In addition to traditional knowledge, there is another shortcut to research into alternatives. This second method is implied in much of what I have already argued above. We have, in my opinion, a virtually untapped resource in the insights of tropical biologists. Experienced tropical biologists are potentially a source of major advances; they should be able to identify systems that are sources of evolutionary strategies that are preadapted to nondestructive parasitization by humans.
To apply such intuitions is going to require a change of attitude on the part of many academic biologists. They will have to recognize that elegant theoretical generalizations, intellectually exciting and satisfying as they may be, are not their only responsibility to science and society. We must become concerned about the future of tropical mankind, even if only because this is the only way we can preserve tropical nature. Janzen’s application of his extensive theoretical insights to the practical problems involved in regrowing tropical forests is a shining example to us all (see Chapter 14). The involvement of several Smithsonian biologists in research on alternatives to destruction is another excellent sign. We should also realize that sciences other than biology may be able to make substantial contributions to halting environmental destruction. An incalculable pressure is exerted on forests worldwide by fuelwood gathering. Eckholm (1975) has estimated that 1.5 billion people derive more than 90% of their fuel needs from wood, while another billion derive at least 50% from wood. Most of these people use extremely inefficient stoves, many with efficiencies less than 10%. The invention of an efficient and inexpensive wood-burning stove could greatly reduce the subsistence pressures on tropical forests.
While all the initiatives and possibilities mentioned above give cause for hope, although certainly not cause for optimism, there is still another area from which
alternatives to destruction might be derived. This is the area of radical innovation. Instead of trying to solve problems by applying existing techniques or improving them, it is possible that we can discover new approaches that are so disjoined from present approaches that they qualify as breakthroughs or new scientific revolutions. The woodgrass system proposed by Shen in Chapter 43 has all the earmarks of a revolutionary approach. It is very simple in concept: in trees, there is a comparatively large increase of biomass in the first years of growth. The growth curve is most efficiently cropped at its maximum angle. At this stage, trees are thin and useless as sources of board timber. But they can be harvested by techniques more appropriate to hay production and the product then subjected to treatment as cattle fodder, composition boards and beams, fuelwood, and so on. The agricultural management of trees as if they were essentially grasses is surely revolutionary. This kind of approach needs to be applied to our whole approach to tropical problems.
Finally, there must be a realization that present levels of research into all the matters relating to tropical ecosystems, natural and man-made, terrestrial and marine, is totally and fundamentally inadequate. It is scientifically invidious to compare research expenditures on astronomy and tropical biology, but this does serve to point to the neglect of studies of life on Earth. Astronomical studies are important; they are contributing fundamentally to knowledge, but the stars are not about to disappear. A world expenditure on fundamental studies of tropical biology that is less than half the cost of a Boeing 747 airliner is a sad reflection on both our priorities and our values.
REFERENCES
Eckholm, E. 1975. The Other Energy Crisis: Firewood. Worldwatch Paper 1. Worldwatch Institute, Washington, D.C. 20 pp.
Hubbell, S.P., D.F.Wiemer, and A.Adejare. 1983. An antifungal terpenoid defends a neotropical tree (Hymenaea) against attack by fungus-growing ants (Atta). Oecologia 60:321–327.
Janzen, D.H., and P.S.Martin. 1982. Neotropical anachronisms: The fruits the gomphotheres ate. Science 215:19–27.
NRC (National Research Council). 1975. Underexploited Tropical Plants with Promising Economic Value. Board on Science and Technology for International Development Report 16. National Academy Press, Washington, D.C. 187 pp.
NRC (National Research Council). 1979. Tropical Legumes: Resources for the Future. Board on Science and Technology for International Development Report 25. National Academy Press, Washington, D.C. 331 pp.
NRC (National Research Council). 1981. The Winged Bean: A High Protein Crop for the Tropics. Board on Science and Technology for International Development Report 37. National Academy Press, Washington, D.C. 49 pp.
NRC (National Research Council). 1983. Little-Known Asian Animals with a Promising Economic Future. Board on Science and Technology for International Development Report 46. National Academy Press, Washington, D.C. 133 pp.
NRC (National Research Council). 1984. Amaranth: Modern Prospects for an Ancient Crop. Board on Science and Technology for International Development Report 47. National Academy Press, Washington, D.C. 76 pp.
NRC (National Research Council). 1985. Jojoba: New Crop for Arid Lands. Board on Science and Technology for International Development Report 53. National Academy Press, Washington, D.C. 102 pp.
Robinson, M.H. 1985. Alternatives to destruction: Investigations into the use of tropical forest resources with comments on repairing the effects of destruction. Environ. Prof. 7:232–239.
Robinson, M.H. 1986. The Biological Resources of Southeast Asia and Future Development. Paper presented at ASEAN Science and Technology Conference, Kuala Lumpur, Malaysia, 29 April 1986.
Smythe, N. 1970. Relationships between fruiting seasons and seed dispersal methods in a neotropical forest. Am. Nat. 104(935):25–35.
Temple, S.A. 1977. Plant-animal mutualism: Coevolution with Dodo leads to near extinction of plant. Science 197:885–886.
CHAPTER 41
AGROECOLOGY AND IN SITU CONSERVATION OF NATIVE CROP DIVERSITY IN THE THIRD WORLD
MIGUEL A.ALTIERI
Associate Professor, Division of Biological Control, University of California, Berkeley
LAURA C.MERRICK
Graduate Research Assistant, Department of Vegetable Crops, University of California, Davis, and L.H.Bailey Hortorium, Cornell University, Ithaca, N.Y.
Today, the foundation and health of agriculture in industrial countries largely depend on their access to the rich crop genetic diversity found in Third-World countries. Yet the very same germplasm resources most sought after for their potential applications in biotechnology are constantly threatened by the spread of modern agriculture. On the one hand, the adoption of high-yielding, uniform cultivars over broad areas has resulted in the abandonment of genetically variable, indigenous varieties by subsistence farmers (Frankel and Hawkes, 1975; Harlan, 1975). The new varieties are often less dependable than the varieties they have replaced when grown under traditional agricultural management (Barlett, 1980). On the other hand, the planting of vast areas with monocultures of genetically uniform cultivars makes agricultural productivity extremely vulnerable to yield-limiting factors, as illustrated by the southern corn leaf blight epidemic in the United States in 1969–1970 (Adams et al., 1971). Agroecosystems established far from centers of origin tend to have simpler genetic defenses against pathogens and insect pests, rendering crops more vulnerable to epidemic attack—a situation that rarely occurs in an unmodified traditional agroecosystem (Segal et al., 1980).
Concern for this rapid loss of genetic resources and crop vulnerability consolidated at the international level about 13 years ago with the establishment of the International Board for Plant Genetic Resources (IBPGR), which coordinates a global network of gene banks to provide plant breeders with the genetic resources
necessary to develop better crops. International efforts have so far placed more emphasis on increasing yield than on maintaining stable harvests (Plucknett et al., 1983)—an emphasis that has provided the justification for technological innovation and transfer in a manner not reflecting indigenous social, ecological, and ethnobotanical considerations. Landraces1 and wild relatives of major crops are collected from their native habitats, and the seed or vegetative material is placed in gene banks for storage or breeding collections for evaluation and potential use (Frankel and Bennett, 1970). Although ex situ conservation methods have contributed to the improvement of certain crops and the storage of the germplasm of a variety of major crops (Frankel and Bennett, 1970), they do not provide a panacea for conserving natural sources of crop genetic resources (Oldfield, 1984). A major problem is that seed storage freezes the evolutionary processes by preventing new types or levels of adaptations or resistance to evolve, because plants are not allowed to respond to the selective pressures of the environment (Simmonds, 1962). In addition, ex situ methods remove crops from their original cultural-ecological context (Nabhan, 1985)—the human-modified systems in which they evolved.
As Wilkes (1983, p. 136) stated, “The centers of genetic variability are moving from natural systems and primitive agriculture to gene banks and breeders’ working collections with the liabilities that a concentration of resource (power) implies.” Controversy has already erupted around the control of gene banks, since countries such as Colombia, Cuba, Libya, and Mexico question the free access to genetic resources by industrial countries. In the industrial countries, breeders develop new commercial varieties, often using valuable genes derived from landraces or wild species originally collected in the Third World. Then, the new commercial varieties are sold back to the Third World at considerable profit (Wolf, 1985).
A number of scientists have emphasized the need for in situ conservation of crop genetic resources and the environments in which they occur, since in situ conservation allows for continued, dynamic adaptation of plants to the environment (Nabhan, 1985; Prescott-Allen and Prescott-Allen, 1982; Wilkes, 1983). For agriculture, this phenomenon is particularly important in areas under traditional farming, where crops are often enriched by gene exchange with wild or weedy relatives (Harlan, 1965). However, most researchers consider that in situ preservation of landraces would require a return to or the preservation of microcosms of primitive agricultural systems—to many, an unacceptable and impracticable proposition (Ingram and Williams, 1984). Nevertheless, we contend that maintenance of traditional agroecosystems is the only sensible strategy to preserve in situ repositories of crop germplasm. Although most traditional agroecosystems are undergoing some process of modernization or drastic modification, conservation of crop genetic resources can still be integrated with agricultural development, especially in regions where rural development projects preserve the vegetational diversity of traditional agroecosystems and depend upon the peasants’ rationale to utilize local resources and their intimate knowledge of the environment (Alcorn, 1984; Nabhan, 1985; Sarukhan, 1985).
In alternative strategies, the conservation of plant genetic resources and agricultural development by peasants can be considered simultaneously. In a recent article (Altieri and Merrick, 1987), we suggested the best ways in which traditional varieties, agroecological patterns, and management systems can be integrated into rural development programs to salvage crop genetic resources. These are reviewed below.
PEASANT AGRICULTURE AND CROP GERMPLASM RESOURCES
The stability and sustainability of traditional agriculture are based on crop diversity (Altieri and Merrick, 1987; Chang, 1977; Clawson, 1985; Egger, 1981; Harwood, 1979). The peasant’s strategy of hedging against risk by planting several species and varieties of crops in different spatial and temporal cropping systems designs is the most effective long-lasting means of stabilizing yields. Although improved varieties are distributed throughout Third-World countries, they have made serious inroads in areas strongly linked to commercial agriculture and the national market, where they have hastened the disappearance of wild relatives and traditional varieties of crops (Brush, 1980). Thus today, the rural landscapes consist of mosaics of modern and traditional varieties and technologies (Figure 41–1). As areas become more marginal in natural resources and in infrastructural support, however, the use of improved varieties declines; farmers abandon them because of

FIGURE 41–1 A traditional small farm system in Tlaxcala, Mexico, exhibiting a corn-alfalfa strip-cropping pattern, borders of Maguey and Capulin trees, and a number of wild plants both within and around the crop area. Photo by M.A.Altieri.
the risk and expense and rely on their century-tested, regionally adapted stocks (Figure 41–2). In Peru, for example, as altitude increases, the percentage of native potatoes in the field increases steadily (Brush, 1980). In Thailand, rice farmers plant the modern semidwarf varieties in part of their land during the dry season and sow traditional varieties during the monsoon season. They have thus established a system that allows them to take advantage of the productivity of irrigated modern varieties during dry months and the stability of the traditional varieties in the wet season when pest outbreaks are common (Grigg, 1974).
As the economic crisis deepens in most developing countries, and rural populations become increasingly impoverished, a sizeable portion of the peasantry is renewing use of the traditional varieties and low-input management practices needed for subsistence agriculture (Altieri and Anderson, 1986). Opting for less crop uniformity may mean lower yields for farmers, but it gives them the extra margin of resistance to pests, diseases, and other environmental hazards—an important

FIGURE 41–2 Bean seeds of different colors expressing high genetic diversity. Harvested from a single field in a rain-fed traditional cropping system in Tlaxcala, Mexico. Photo by M.A.Altieri.
consideration when working under conditions of economic uncertainty. In many areas, unfortunately, the return of some peasant communities to native varieties has been difficult because of genetic erosion.
Several factors have contributed to this loss of crop genetic resources (Nabhan, 1986): decrease in the number of growers, decrease in crop size per field, decrease in planting frequency, loss of seed-saving and seed-collection skills, and changes in the crop’s vulnerability to pests and weeds. In such regions, the implementation of in situ crop genetic conservation will be more complex, since both the folk science and the genetic heritage necessary to nurture such programs can only be retrieved very slowly (Altieri and Merrick, 1987).
TOWARD A STRATEGY FOR IN SITU CROP GENETIC CONSERVATION
Recommendations for in situ conservation of crop germplasm have emphasized the development of a large system of village-level landrace custodians (a farmer-curator system) whose purpose would be to continue to grow a limited sample of endangered landraces native to the region (Nabhan, 1985). To preserve crop-plant diversity, Wilkes (1983) has suggested that the governments set aside carefully chosen 5-by-20-kilometer strips at as few as 100 sites around the world where native agriculture is still practiced, areas where both indigenous crops and their close wild relatives may interbreed periodically. The idea of setting aside parks for crop relatives and landraces is obviously a luxury in countries where farmland is already at a premium, but to some this may be less costly than allowing native crop varieties to disappear (Brush, 1980; Wolf, 1985). In many areas, the urgent short-term issue is survival, and it would therefore be totally inappropriate to divert the limited land available to peasants for conservation purposes per se so that the germplasm could be used by industrialized nations. Such a position could be viewed as an undesirable form of neocolonialism.
Supporters of in situ strategies argue that farmers should be incorporated into conservation programs by creating biosphere reserves where peasants can continue their traditional agriculture under subsidy (Wilkes, 1983). Once identified, these areas would be designated as germplasm centers and would qualify for special agricultural assistance aimed at promoting the cultivation of native varieties. Industrialized countries using this germplasm would subsidize farmers cultivating native varieties and would help them in marketing the produce. Brush (1980) believes that these support programs could also include the machinery and financial aid needed to compensate for monetary losses incurred by farmers who maintain germplasm and do not reap the rewards of growing improved varieties. Some means of computing the opportunity cost of maintaining these cultivars must be established.
Although Nabhan (1985) agrees with the creation of centers of traditional agriculture, he believes that conservation measures will be most effective when native farmers are cognizant of, and involved in, their planning and implementation. Such efforts are likely to succeed, he argues, as long as members of a culture identify their own reasons for maintaining their crop heritage and persevere in conducting the practices for nurturing these plants. An obvious incentive for
resource-poor peasants is to enhance harvest security and to make them independent of the market for seeds and inputs. It is for this type of farmer that preservation efforts should be linked to the overall rural development agenda. Design of sustainable farming systems and appropriate technologies aimed at upgrading peasant food production for self-sufficiency should incorporate locally adapted, native crops, and wild and weedy relatives, to complement the various production processes (Altieri and Merrick, 1987).
At present, there are a number of assistance programs temporarily directed at meeting the subsistence needs of peasants (Altieri and Anderson, 1986; Altieri and Merrick, 1987). These efforts are intended to minimize dependency on purchased inputs and industrialized technology; improve the efficiency with which local resources, including local vegetation, is used; achieve the production goals needed to satisfy home consumption; and favor peasant organization to enhance their capacity for economic reproduction (de Janvry, 1981). The approaches consist generally of taking existing peasant production systems and technologies as starting points and then using modern agricultural science to improve, progressively and carefully, on the productivity of these systems (Altieri, 1983).
Thus, proposed agricultural models are based on the peasants’ skills in utilizing the environment and their ability to cope with change, as well as their knowledge of the plant resources and the general biology of the area. The programs have a definite ecological bent and rely on resource-conserving and yield-sustaining production technologies. Through the design of crop associations and regionally adapted patterns, functions of nutrient recycling, natural pest control, and soil conservation can be optimized (Altieri, 1983; Gliessman et al., 1981). As subsistence needs are met, most programs emphasize channeling of excess production to local markets. Income generation is also achieved by promoting nonagricultural activities (e.g., basketry) within the villages.
When valuable crop genetic resources are incorporated into farming systems designed to encourage self-sufficiency of the rural poor, important conservation gains can be achieved. This is illustrated by the efforts by Nabhan (1984) and associates to improve arid land agriculture for native Americans in the U.S.-Mexico borderlands and by a number of groups in Latin America (Altieri and Anderson, 1986; Altieri and Merrick, 1987).
In Mexico, for example, researchers designed for peasants production modules based on the pre-Hispanic traditional chinampas (a meadow or garden developed from a reclaimed lake or pond) and multilayered, species-rich kitchen gardens that once characterized the original agroecosystems of Tabasco, Mexico. Diverse arrays of crop and noncrop species were utilized in the various modular systems. In a parallel project, integrated farms were established in Veracruz to help farmers make better use of their local resources (Morales, 1984). In unique designs based on the chinampas and on Asiatic systems, vegetable production and animal husbandry, including aquaculture, were integrated through the management and recycling of organic matter. The intensive cultivation of corn, beans, and squash for local consumption and of vegetables with high commercial value, e.g., Swiss chard (Beta vulgaris cicla), cilantro (Cordiandrum sativum), chilies (Capsicum spp.), and cabbage (Brassica oleracea capitata), provided abundant plant wastes and cuttings used as
cattle and horse feed; all animal wastes were reintegrated as fertilizer for the fields (Altieri and Merrick, 1987).
In the highlands of Bolivia, the project AGRUCO is attempting to maintain the ecological diversity of the Andean agropastoral economy by helping peasants recover their production autonomy. To replace the use of fertilizers and meet the nitrogen requirements of potatoes and cereals, intercropping and rotational systems utilizing a native species, wild lupin (Lupinus mutabilis), have been designed. Wild lupin has been cultivated in the high Andes for several thousand years (Smith, 1976). It can fix 200 kilograms of nitrogen per hectare, part of which becomes available to the associated or subsequent potato crop, thus significantly minimizing the need for fertilizers (Augstburger, 1983).
In Chile, where lately the peasantry has been subjected to a process of systematic impoverishment, the Centro de Educacion y Tecnologa (CET) is helping peasants become self-sufficient, thus reducing their dependence on credit demands and fluctuating market prices. CET’s approach has been to establish several 0.5-hectare model farms where most of the food requirements for a family with scarce capital and land can be met (Altieri, 1983). Peasant community leaders live in CET farms for variable periods, thus learning through direct participation farm design, management technologies, and resource allocation recommendations. CET farms are composed of a diversified combination of crops, trees, and animals. The main components are vegetables, staple crops (corn, beans, potatoes, fava beans), cereals, forage crops, fruit trees, forest trees (e.g., Robinia, Gleditsia, Salix), and domestic animals all assembled in a 7-year rotational system designed to produce the maximum variety of basic crops in six plots, taking advantage of the soil-restoring properties of the legumes included in the rotation (Altieri, 1983). Most species are locally adapted varieties traditionally grown and consumed by rural populations (Altieri and Merrick, 1987).
THE FUTURE
A number of people have stressed the importance of in situ preservation of crop genetic resources but have failed to suggest practical avenues to achieve this goal in Third-World countries (Prescott-Allen and Prescott-Allen, 1982). If the conservation of crop genetic resources is indeed to succeed among small farmers, the process must be linked to rural development efforts that give equal importance to local resource conservation and to food self-sufficiency and market participation. Any attempt at in situ crop genetic conservation must struggle to preserve the agroecosystem in which these resources occur (Nabhan, 1985, 1986). In the same vein, preservation of traditional agroecosystems cannot succeed if not tied to the maintenance of the sociocultural organization of the local people (Altieri, 1983). The few examples of grassroots rural development programs currently functioning in the Third World suggest that the process of agricultural betterment must utilize and promote autochthonous knowledge and resource-efficient technologies; emphasize the use of local and indigenous resources, including valuable crop germplasm and essentials such as firewood resources and medicinal plants; and be based on self-contained villages and the active participation of the peasants (Altieri and
Anderson, 1986; Altieri and Merrick, 1987). The subsidizing of a peasant agricultural system with external resources (e.g., pesticides, fertilizers, improved seeds, irrigation water) can bring high levels of productivity through dominance of the production system, but these systems are sustainable only at high external cost and depend on the uninterrupted availability of commercial inputs. An agricultural strategy based on native crop diversity can bring moderate to high levels of productivity through manipulation and exploitation of the resources internal to the farm and can be sustainable at a much lower cost and for a longer period.
Ecologists, agronomists, anthropologists, and ethnobotanists have an important, as yet unrealized role in agricultural development and genetic resource conservation (Alcorn, 1984). Through interdisciplinary efforts they can assess traditional know-how to guide the use of modern agricultural science in the improvement of small farm productivity. Ethnobotanists and ecologists can provide critical information for policy makers about resources needing protection and about the ecological and management factors that determine the persistence of natural vegetation in the traditional agroecosystems (Alcorn, 1984).
It is time to recognize the active role of peasants in genetic resource conservation (Alcorn, 1984), and it behooves the industrial nations interested in the germplasm to provide fair subsidies to the peasants for their ecological service of maintaining native cultivars. Farmers should be made aware that reciprocal exchanges of seeds with gene banks are possible and that they have unconditional access to seed held in gene banks, which they can tap if they lose their remaining seeds. Farmers must also know the reasons why others are interested in their seeds. Agricultural education programs including information from elders on traditional planting techniques, seed saving, and seed selection should also be established (Nabhan, 1985).
Incorporation of indigenous crops and other native plant germplasm in the design of self-sustained agroecosystems should ensure the maintenance of local genetic diversity available to farmers. This approach sharply contrasts with current efforts by international centers that tend to concentrate on fewer varieties, potentially eroding genetic diversity and making farmers increasingly dependent on seed companies for their seasonal seed supply. A major concern is that when impoverished peasants become dependent on distant institutions for inputs, rural communities tend to lose control over their production systems (Altieri and Merrick, 1987).
REFERENCES
Adams, M.W., A.H.Ellingbae, and E.C.Rossineau. 1971. Biological uniformity and disease epidemics. BioScience 21:1067–1070.
Alcorn, J.B. 1984. Development policy, forests and peasant farms: Reflections on Haustec-managed forests’ contributions to commercial production and resource conservation. Econ. Bot. 38:389–406.
Altieri, M.A. 1983. Agroecology: The Scientific Basis of Alternative Agriculture. Division of Biological Control, University of California, Berkeley. 162 pp.
Altieri, M.A., and M.K.Anderson. 1986. An ecological basis for the development of alternative agricultural systems for small farmers in the Third World. Am. J. Alt. Agric. 1:30–38.
Altieri, M.A., and L.C. Merrick. 1987. In situ conservation of crop genetic resources through maintenance of traditional farming systems. Econ. Bot. 41(1):86–96.
Augstburger, F. 1983. Agronomic and economic potential of manure in Bolivian valleys and highlands. Agric. Ecosys. Environ. 10:335–346.
Barlett, P.F. 1980. Adaptation strategies in peasant agricultural production. Annu. Rev. Anthropol. 9:545–573.
Brush, S.B. 1980. The environment and native Andean agriculture. Am. Indigena 40:161–172.
Chang, J.H. 1977. Tropical agriculture: Crop diversity and crop yields. Econ. Geogr. 53:241–254.
Clawson, D.L. 1985. Harvest security and intraspecific diversity in traditional tropical agriculture. Econ. Bot. 39:56–67.
de Janvry, A. 1981. The Agrarian Question and Reformism in Latin America. Johns Hopkins University Press, Baltimore. 311 pp.
Egger, K. 1981. Ecofarming in the tropics—characteristics and potentialities. Plant Res. Devel. 13:96–106.
Frankel, O.H., and E.Bennett, eds. 1970. Genetic Resources in Plants—Their Exploration and Conservation. International Biological Programme Handbook #11. Blackwell, Oxford, United Kingdom. 554 pp.
Frankel, O.H., and J.G.Hawkes, eds. 1975. Crop Genetic Resources for Today and Tomorrow. Cambridge University Press, Cambridge, United Kingdom. 422 pp.
Gliessman, S.R., R.E.Garcia, and M.A.Amador. 1981. The ecological basis for the application of traditional agricultural technology in the management of tropical agro-ecosystems. Agro-Ecosystems 7:173–185.
Grigg, D.B. 1974. The agricultural systems of the world: An evolutionary approach. Cambridge University Press, Cambridge. 358 pp.
Harlan, J.R. 1965. The possible role of weed races in the evolution of cultivated plants. Euphytica 14:173–176.
Harlan, J.R. 1975. Our vanishing genetic resources. Science 188:618–622.
Harwood, R.R. 1979. Small Farm Development—Understanding and Improving Farming Systems in the Humid Tropics. Westview Press, Boulder, Colo. 160 pp.
Ingram, F.B., and J.T.Williams. 1984. In situ conservation of wild relatives of crops. Pp. 163–178 in J.H.W.Holden and S.T.Williams, eds. Crop Genetic Resources. Conservation & Evaluation. G.Allen and Unwin, London.
King, F.H. 1927. Farmers of Forty Centuries; or, Permanent Agriculture in China, Korea and Japan. Cape, London. 379 pp.
Morales, H.L. 1984. Chinampas and integrated farms: Learning from the rural traditional experience. Pp. 188–195 in F.De Castri, F.W.G.Baker, and M.Hadley, eds. Ecology in Practice. Vol. 1. Ecosystem Management. Tycooly, Dublin.
Nabhan, G.P. 1984. Replenishing desert agriculture with native plants and their symbionts. Pp. 172–182 in W.Jackson, W.Berry, and B.Colman, eds. Meeting the Expectations of the Land. North Point Press, San Francisco.
Nabhan, G.P. 1985. Native crop diversity in Aridoamerica: Conservation of regional gene pools. Econ. Bot. 39:387–399.
Nabhan, G.P. 1986. Native American crop diversity, genetic resource conservation and the policy of neglect. Agric. Human Val. 2:14–17.
Oldfield, M.L. 1984. The Value of Conserving Genetic Resources. U.S. Department of the Interior, National Park Service, Washington, D.C. 360 pp.
Plucknett, D.L., N.J.H.Smith, J.T.Williams, and N.M.Anishetty. 1983. Crop germplasm conservation and developing countries. Science 220:163–169.
Prescott-Allen, R., and C.Prescott-Allen. 1982. The case for in situ conservation of crop genetic resources. Nat. Resour. 23:15–20.
Sarukhan, J. 1985. Ecological and social overviews of ethnobotanical research. Econ. Bot. 39:431–435.
Segal, A., J.Manisterski, G.Fischbeck, and I.Wahl. 1980. How plant populations defend themselves in natural ecosystems. Pp. 75–102 in J.G.Horsfall and E.B.Cowling, eds. Plant Disease: An Advanced Treatise. Academic Press, New York.
Simmonds, N.W. 1962. Variability in crop plants, its use and conservation. Biol. Rev. 37:422–465.
Smith, P.M. 1976. Minor crops. Pp. 312–313 in N.W.Simmonds, ed. Evolution of Crop Plants. Longman, London.
Wilkes, H.G. 1983. Current status of crop plant germplasm. CRC Crit. Rev. Plant Sci. 1:133–181.
Wolf, E.C. 1985. Conserving biological diversity. Pp. 124–126 in The State of the World. A Worldwatch Institute Report on Progress Toward a Sustainable Society. W.W. Norton, New York.
CHAPTER 42
ALTERNATIVES TO DESTRUCTION
Research in Panama
GILBERTO OCANA
Conservation Resources Manager
IRA RUBINOFF
Director
NICHOLAS SMYTHE
Staff Scientist
DAGMAR WERNER
Research Supervisor, Iguana Management Project Smithsonian Tropical Research Institute, Balboa, Republic of Panama
In 1983, the Smithsonian Tropical Research Institute (STRI) in the Republic of Panama initiated a program called Alternatives to Destruction to develop practices that could help to feed human populations and to provide some alternatives to cattle rearing and other extractive practices that are responsible for much of the destruction of tropical forests in Central and South America. STRI has approached the problem in ways involving both plants and animals with grants from the W.Alton Jones Foundation, the Exxon Corporation, the James Smithson Society, and the World Wildlife Fund. Its goals are either to enhance production in association with native vegetation or to enhance vegetation development and soil stabilization in deforested areas. Animal species selected for management projects feed directly on forest vegetation, so there is an incentive to native populations to preserve some of the forest. STRI is using knowledge derived from basic research to explore approaches that will not only yield harvestable products but will also protect natural forest.
Tropical deforestation and the concomitant loss of biological diversity in tropical rain forests have received much recent attention. The causes of tropical deforestation are complex, and no single response will be sufficient to counteract its effects. The problem has been identified, but solutions will have to incorporate sound biological principles and must be reasonable within a social and economic context. A research organization devoted to tropical biology cannot directly resolve
social and economic problems, but an understanding of the biological functioning of the system is a prerequisite to amelioration of such problems.
The slash-and-burn farmer is perhaps the principal agent of rain forest destruction, and it is commonplace to hear that he must be stopped. The key question is: “How to stop him?” Simply enacting legislation that would prevent him from further cutting cannot work, for the need for food grows with the blossoming population, and slash-and-burn is the only proven way of exploiting the land. It will continue until suitable alternatives are provided.
In the search for alternatives to destruction, four major projects were selected by STRI researchers, who believe that their knowledge of the species involved combined with the possibilities of finding less destructive crops could be productive. These projects are management of the green iguana, captive breeding of the paca, forest gardening or experimentation with crops that cause minimum perturbation in the existing forest, and management of game mammals. It was, of course, realized that quick fixes can not be expected in the face of so many complex problems, but success in even one or two projects could contribute greatly toward decreasing the seriousness of those problems.
MANAGEMENT OF THE GREEN IGUANA (IGUANA IGUANA)
For over two decades, basic research at STRI has led to great increases in knowledge of the biology of the green iguana (Burghardt and Rand, 1982). Formerly a wide-ranging reptile, and prized as a protein source for more than 7,000 years, this animal is now drastically decreasing in numbers due to habitat destruction from slash-and-burn farming, conversion of forested lands to pasture, increased use of biocides, and uncontrolled hunting. Through much of the natural range of the green iguana, there is a widespread belief that the about-to-be-laid eggs have aphrodisiac properties, and the gravid females, which are easily discovered as they converge on traditional, communal nesting areas, are especially vulnerable.
An early goal in this project was to reduce egg and hatchling mortality. Eggs laid in the wild have about 50% hatching success, and only 5% of the hatchlings survive their first year. Techniques developed at STRI have yielded captive hatching success in excess of 95%. Moreover, survival to the yearling stage is near 100%. Captive-raised hatchlings are fed a low-cost, high-protein diet and gain weight twice as fast as wild iguanas. An artificial nest developed by STRI to facilitate egg collection is preferred by both wild and captive females over nests of their own construction. Techniques developed at STRI ensure production of a predictable number of young iguanas and their regulated multiplication in captivity (Werner, 1986).
Several new directions, including studies of nutrition and disease control, as well as experimental releases of yearlings into the wild, are now being explored. In December 1985, reintroduction experiments using 1,200 7- to 10-month-old, captive-raised iguanas were begun in appropriate habitats in rural communities. Their survival from the age at which they were released appears to be comparable to that of wild-born animals. The repopulation experiment is accompanied by an education project designed to encourage rational resource management and to
introduce the techniques to the rural communities. Results to date indicate that wild iguana populations can be reestablished in 3 years, after which harvesting can begin.
Our calculations show that meat production from iguanas matches that of cattle. Beef production may be higher in the first years after the forest is cleared to create pasture. However, the annual cattle yield drops to 15 kilograms per hectare after 10 to 15 years because the quality of the pasture is lower. By comparison, the results of the Iguana Management Project indicate that iguanas could provide a sustainable yield of more than 230 kilograms per hectare annually. Moreover, iguana management has the potential to maintain or to improve soils on land degraded by intensive cultivation or cattle ranching. Production costs are estimated at $0.66 per kilogram, and the meat is presently sold (albeit illegally) for between $1 and $6 per kilogram. A secondary benefit of iguana management is that simultaneous reforestation with high-quality lumber and fruit trees could also become attractive. Development of a reforestation scheme including plant species that also support iguana populations could yield a harvest of iguanas after 5 to 6 years and thus provide an early return that compensates for the reforestation investment.
CAPTIVE BREEDING OF THE PACA (CUNICULUS PACA)
The paca is a large nocturnal rodent related to the guinea pig and is native to the broad-leaved forests of Central and South America. Its meat is very highly prized by people of both rural and urban populations. An adult paca weighs about 10 kilograms, 60 to 70% of which is edible meat. Domestication of pacas could provide an inexpensive supply of high-quality meat for local consumption or for use as a cash crop. The purposes of this project are to evaluate the possibilities of breeding pacas in captivity and to develop techniques that rural people can use for captive management. Once regular captive breeding is established, the process of domestication should follow as a matter of course. Some characteristics of the paca favor domestication; some do not (Smythe, 1987). Precocial young undergo a period of early learning that makes individuals very easy to tame. In addition, the animals subsist on food that comes from the forest and thus would be inexpensive to acquire.
Among the characteristics that do not favor domestication are the fact that pacas naturally live in pairs that defend territories and are thus highly intolerant of other pacas. They are also strictly nocturnal, retiring to burrows or retreats, which are aggressively defended against other pacas or other potential intruders in the daytime. Furthermore, pacas generally produce only one or two young per year. (The fact that their natural reproductive rate is low does not necessarily indicate a low reproductive potential. Artificial selection to achieve the reproductive potential would be expected to occur early in the domestication process. The reproductive potential of a female paca should be between 12 and 16 per year.)
All young mammals are more socially tolerant than adults of the same species. By taking young pacas away from their mothers when they are only a few days old and raising them in contact with other pacas, animals have been taught to be nonaggressive toward other members of their own group. The goal is to raise them
in groups of five females per male. At present, we have two groups of two females with one male, and two of three females with one male. None of these animals has shown any aggression toward other group members, and all are easier to handle than those raised by their mothers.
It might be expected that denial of a retreat to a naturally burrowing animal would give rise to pathological trauma, but researchers in comparative psychology have shown with other burrowing mammals (Price, 1984) that raising them in cages without any form of retreat causes the animals to mature faster and show less stress than those raised with retreats. Pacas raised without burrows are definitely more tranquil and are also more diurnal, which facilitates observation of their health and behavior. Psychologists have also demonstrated that handling makes an animal’s subsequent behavior toward its handlers less aggressive. The different groups of pacas have been treated according to different regimens, and all are easier to handle than animals left alone. The ease of manipulation is inversely proportional to the age at which they were removed from their mothers. It is hoped that the young born to human-acclimated females will learn their attitudes from their parents.
Pacas born early in the project gained weight at an average rate of 13 grams per kilogram of body weight per day (g/kg bw/day) and reached a weight of 6 kilograms (approximately the weight at maturity) at 18 months. As a result of improved nutrition and a rigorous antiparasite program, those most recently born gained 20 g/kg bw/day, and some have reached 6 kilograms at 4 months. By comparison, cattle in temperate regions gain about 7 to 10 g/kg bw/day during the same age span (Seigmund, 1979).
Pacas are naturally frugivorous. The gathering of wild fruits is not only highly laborious but is also subject to the seasonality of production. Many forest leaves are as nutritious as fruit and may be higher in proteins. One of the benefits of hand-rearing pacas is that they can be taught to eat diets that they would not encounter in the natural state and that adults only learn to eat reluctantly. Experiments are presently under way to develop a diet that consists of a high proportion of easily obtained leaves supplemented with a readily available, aseasonal, high-carbohydrate food such as manioc (Manihot esculentia), which is locally called yucca.
Only a few wild species have been truly domesticated, and most of these have been social species such as dogs and ungulates. Thus, the attempt to domesticate (or even semidomesticate) the paca is faced with long odds. But none of the traditional domestic meat animals thrive in the lowland tropical areas; thus there is a need for a domestic species that will do well in the area. In the 3.5 years since the inception of this project, significant progress has been made.
FOREST GARDENING
This 3-year project has several objectives. Using species that are not traditional in Panama, STRI hopes to restore and maintain soil fertility by selecting and establishing highly adaptable, hardy, and competitive leguminous plant species with emphasis on multipurpose shrubs and trees that can be used for green manure, forage, firewood, and timber production; to evaluate the adaptability of potential
food crops and of native fruit and leguminous trees to poor soil and other conditions found in small forest clearings; and to transfer field plots and plant material to Panamanian government agencies responsible for the development of agricultural technology to enable them to continue with the most promising results obtained by this project.
Soils in the project area are very poor, like much soil under tropical forests. They are characterized by very low to medium pH values (4.3–5.4); a low to high aluminum content (traces-7.4 ppm), and low to extremely low phosphorus concentrations (traces-9.5 ppm).
The quality of these soils did not seem to vary with successional stage of vegetation. Seeds of leguminous shrubs, trees, and vines with the required adaptation and performance potential (NRC, 1979) were obtained from several sources. Promising results in terms of vigor, competitiveness, and stress tolerance against drought or impeded drainage were obtained with fast-growing trees, e.g., Sabah salwood (Acacia mangium) in Malaysia, wattle (A. auriculiformis) in Papua New Guinea, and mata raton, madre del cacao (Gliricidia sepium) in Latin America; shrubs, e.g., Desmodium gyroides, Tephrosia candida, and Townsville clover (Stylosanthes guianensis) in Trinidad; and creeping vines, e.g., tropical kudzu (Pueraria phaseoloides) and Desmodium ovalifolium. Of these, only G. sepium and S. guianensis are native to the Neotropics.
The response of Acacia mangium was impressive under the local conditions; no species hitherto proposed for lumber production in similar soil in Panama even approaches the growth figures obtained with this species. The trees showed a striking response to the addition of phosphorus, and rhizobium-induced root nodules were large and prolific. A symbiotic relationship with mycorrhizal fungi was clearly indicated in growth trials using forest and pasture soils. The performance of A. mangium and of other leguminous species under severely adverse soil conditions opens up a wide range of options for the restoration and maintenance of soil fertility and for the development of agroforestry systems.
Results of trials in small forest clearings showed that the following species could constitute the core of a highly productive forest garden in tropical soils of low fertility: arrow root (Maranta arundinacea), tuber-bearing yam (Dioscorea bulbifera), Mexican yam bean (Pachyrhizus erosus), American peach palm (Bactris gasipaes), hardy banana clones (Musa spp.), a shade-tolerant, soil-covering vine (Desmodium ovalifolium), and leguminous shade trees (Erythrina spp. and Gliricidia sepium) (cf. Huxley, 1983).
MANAGEMENT OF GAME MAMMALS
Native populations in the Neotropical humid forests have traditionally relied on native wildlife for their high-quality protein. Since the advent of firearms and headlamps, more efficient hunting methods combined with habitat destruction have resulted in the extermination of game species in many areas. It is thus urgent to determine the sustainable yield of protein that can be obtained from these forests and to devise the means to manage the game populations appropriately.
In many areas, forest remains only in small patches such as on hilltops, along rivers, or in areas where the topography makes exploitation difficult. Populations of game animals are usually reduced in such areas, and since they are isolated, it is difficult or impossible for the game species to return. Wildlife populations could be maintained (or established) in these isolated patches, and in the absence of their natural predators (which the patches are too small to support), they could be cropped as a sustainable yield protein.
Most forest game animals are frugivorous, and as a result, they experience high juvenile mortality during seasonal scarcity of fruit (Smythe, 1986). Pre-Hispanic forest natives may have left waste crops in the fields, inadvertently or intentionally. Game animals that came to feed were exploited as a supplementary source of protein. The natives thus practiced provisioning of the population and may, in so doing, have reduced seasonal juvenile mortality of the game animals. A principal aim in this project is to develop methods to enhance juvenile survivorship in the field by provisioning in times of fruit scarcity.
In any wild animal management scheme, it is necessary to census the populations as accurately as possible. Since no single method of population censusing appears to be adequate in tropical forests, two different methods are used where possible. Populations of potential meat animals are being estimated with strip-census techniques in a 62-hectare peninsula adjacent to Barro Colorado Island, which is isolated from the contiguous forests by a 1-kilometer-long, five-strand electric fence. The animals being censused in this way are the agoutis (Dasyprocta punctata), pacas, spiny rats (Proechimys semispinosus), collared peccaries (Tayassu tajacu), and white-tailed deer (Odocoileus virginianus). The three species of rodents are also being censused in a trap-mark-release program. This program will be used to make more accurate estimates of the rodent populations but is confined to the period when food is scarce in the forest (between September and March) and animals are willing to enter traps.
The study area is divided into quadrants, one of which is artificially provisioned during the season of scarcity. The effect of the provisioning on the survival of juvenile animals is being determined by the censuses.
Progress in this project has been slower than in those discussed earlier, chiefly due to the unpredictable elements inherent in working with wild animals and the necessary restrictions on manipulation of predators or competing species imposed by working within a protected area. Furthermore, the electric fence is less effective than expected: predators have moved in and taken advantage of trapped animals, and unwanted animals have settled in the provisioned areas and used provisions intended for the targeted species. Nonetheless, much valuable knowledge is being gained.
STRI personnel undertook these projects several years ago with the full understanding that instant solutions to the problems that they were trying to solve were unlikely. But significant progress has been made in these high-risk ventures. Further progress, no matter how slow it is in coming, and the dedication necessary to achieve that progress are essential if conservation goals are to be achieved at the same time as the livelihood of the rural poor is improved.
REFERENCES
Burghardt, G.M., and A.S.Rand, eds. 1982. Iguanas of the World: Their Behavior, Ecology, and Conservation. Noyes, Park Ridge, N.J. 472 pp.
Huxley, P.A., ed. 1983. Plant Research and Agroforestry: Proceedings of a Consultative Meeting Held in Nairobi, 8 to 15 April, 1981. International Council for Research in Agroforestry, Nairobi, Kenya. 617 pp.
NRC (National Research Council). 1979. Tropical Legumes: Resources for the Future. National Academy of Sciences, Washington, D.C. 331 pp.
Price, E.O. 1984. Behavioral aspects of animal domestication. Q. Rev. Biol. 59(1):1–32.
Siegmund, O.H., ed. 1979. Nutrition. Pp. 1268–1269 in The Merck Veterinary Manual, Part V. Merck, Rahway, N.J.
Smythe, N. 1986. Competition and resource partitioning in the guild of Neotropical, terrestrial, frugivorous mammals. Annu. Rev. Ecol. Syst. 17:169–188.
Smythe, N. 1987. The paca (Cuniculus paca) as a domestic source of protein for the Neotropical, humid lowlands. Appl. Anim. Behav. Sci. 17(1–2):155–170.
Werner, D.I. 1986. Iguana management in Central America. BOSTID Dev. 6(1):1, 4–6.
CHAPTER 43
BIOLOGICAL ENGINEERING FOR SUSTAINABLE BIOMASS PRODUCTION
SINYAN SHEN
Program Manager, Energy and Environmental Systems Division, Argonne National Laboratory, Argonne, Illinois
Since the 1950s, the new science of molecular biology has produced a remarkable outpouring of new ideas and powerful techniques. From this revolution has sprung a new discipline called genetic engineering, which gives us the power to alter living organisms for important purposes in agriculture, energy, industry, and medicine. Applications of resulting technologies span the range from the ancient arts of fermentation to the most esoteric use of gene splicing and monoclonal antibodies. During the last decade, progress has continued with the rapid development of techniques to produce chemical feedstock using renewable sources of material. In particular, modern and traditional genetic engineering techniques have been combined with modern agricultural methods and chemical engineering know-how to produce high-volume biomass feedstocks at low cost for use as energy, chemicals, and building materials.
A recently evolved engineering discipline requires the design of biological systems that accumulate organic material photosynthetically more efficiently than those used in food production and with positive environmental impacts. In a fairly broad context, biological engineering can be defined as the engineering and use of biological systems in the production of goods and services for industry, trade, and commerce. The underlying fundamental scientific principles, engineering considerations, and government regulations dealing with the development and application of biological engineering all need to be addressed. Since this is a relatively new discipline, the best way to describe its integral components is through a number of examples. The examples discussed in this chapter include the engineering of biological production systems for biomass feedstocks with environmental benefits in mind.
Every region of the world has its own ways of managing its energy and material resources. The criteria for selecting the appropriate mixture of energy and material technologies are usually based on resource constraints imposed on the region and the structure of its industry, e.g., whether it is capital-intensive or labor-intensive. Fossil energy resources and mineral resources are accumulated over a very long time and can only be managed. Biomass resources, on the other hand, are composed of organic material accumulated by photosynthesis and can be enhanced and managed.
Very few things in this world have a global impact on us as great as the forest/ grassland ecosystem. A balanced tree and grass ecosystem controls runoff, supplies water, and supports irrigation, soil fertility, and oxygen production. For every given ecological zone, there is a minimum ratio of tree acreage to food-crop acreage that must be maintained locally. Otherwise, the long-term productivity of land and the supply of water and nutrients cannot be maintained.
Since the introduction of large-scale mechanization, plant breeding, irrigation, and fertilization, there has been a tendency to move away from the ecological balance needed to sustain productivity. We often speak of land productivity, but we are really only talking about land fertility. The overall productivity can only be measured by considering the entire ecosystem, which supports the life of all zoological species, trees, legumes, and grasses. The study of genetics and plant breeding has resulted in the production of superior varieties of trees, legumes, and grasses in terms of product yields. To increase the economic output of agriculture and forestry, however, we have often selected ways to design production systems that are not compatible with long-term productivity.
The pursuit of short-term profitability at any price, justified by the technocratic concept of consumer needs, is leading to the replacement of genetically diversified natural forest, for example, by plantations consisting of row upon row of trees belonging to a very small number of different species. These species are chosen for their rate of growth and consist mainly of conifers, poplars, or eucalyptus. The complete lack of diversity in monoculture plantations makes them particularly vulnerable to attack by pests, that is, by pathogenic insects and fungi. Proliferation of bark beetles (family Scolytidae), for example, which is generally haphazard and localized among naturally diversified conifers, assumes disastrous proportions in spruce plantations that have spread so extensively over France during the last decades.
One of the results of the systematic introduction of monoculture plantations is a significant alteration of the soil structure, including a reduction in porosity of upper soil horizons, which makes them less and less permeable. Thus, there is a twofold or threefold increase in runoff. Loss by evaporation also increases, because pine needles retain a large amount of rainwater. Coniferous forests are therefore drier than broadleaved woodlands, other conditions being equal. Soil fauna is also affected. The number of earthworms under conifers is smaller by a factor of 100 to 500 than under broadleaved trees. Decomposers are also affected, and a marked reduction in bacteria occurs, especially in the microorganisms involved in the nitrogen cycle. The current practice of reforestation with conifers in temperate, and even in tropical, regions would seem to constitute an astonishing perseverance
with mistaken methods on the part of the authorities responsible for forests in the countries involved.
There is nothing wrong with using genetically superior material, and there is also nothing wrong with increasing our economic output, as long as we fully understand what it takes ecologically to sustain long-term balance and productivity.
The horticultural agriculture of Asia provides some food for thought. The intensive agriculture of Eastern China, for example, has been sustained for thousands of years. The key to its success is genetic diversity in any one locality. With sufficient genetic diversity in the agriculture of any one locality and a balance of trees and grasses (most food crops are tall grasses), Eastern China maintains its ecological balance and long-term productivity. The indiscriminate exploitation of forests in parts of Northern China, however, has resulted in the rapid expansion of deserts there.
WOOD GRASS
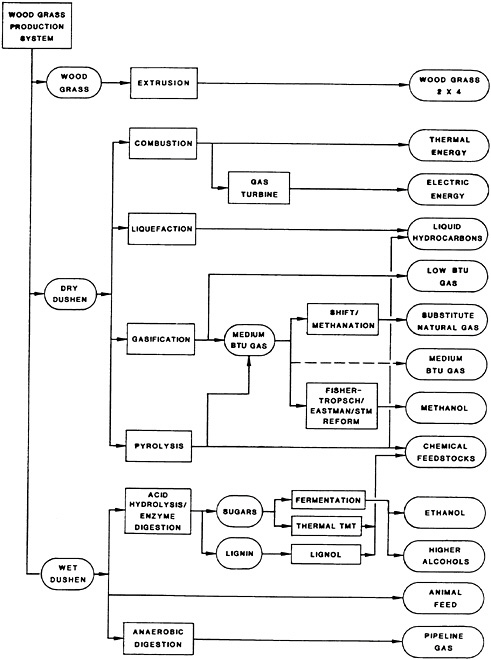
High-yield, fast-growing multipurpose trees have been considered by many countries to increase both economic and natural resource development. A biologically engineered system developed in the United States produces a large volume of woody feedstock at low cost. Modern agricultural techniques are combined with our understanding of the tree growth to produce feedstocks that are conveniently handled in conversion processes. One biologically engineered system produces two varieties of wood grass products: the dry Dushen and the wet Dushen (Shen, 1982; Shen et al., 1984).
Dry Dushen is made by chopping the wood grass after it is crushed. It is a good combustion feedstock for use in thermochemical conversion processes such as combustion, liquefaction, gasification, and pyrolysis. Wet Dushen is made by chopping wood grass into 2.5-centimeter segments as the wood grass is harvested. This product makes an excellent feedstock for such biochemical conversion processes as acid hydrolysis, enzymatic hydrolysis, and anaerobic digestion. Wood grass was given that name because its stumpage is a thin woody material (the wood grass) and it is managed by techniques used for managing grasses. The production system is biologically engineered for maximum productivity and efficiency in land utilization. Optimal plant spacing is used for the product desired, and woody biomass is harvested on an annual, biannual, or seasonal basis.
In traditional forestry, tree seedlings are spaced relatively far apart. Thus, the trees are not biologically affected by each other until they are near the end of the rotation, which could be many years after planting. The efficiency of land use is therefore low. The wood grass system uses specially selected fast-growing and coppicing species. The coppicing capability removes the need for frequent replanting, and the fast-growing characteristics are crucial since only species with a sufficiently narrow coppice growth curve can be used in the wood grass system.
Agricultural management techniques are used, optimal fertilization schedules are followed, and the soil is enriched to compensate for nutrient removal. Irrigation may be needed in certain parts of the world during root establishment. The methods of planting include vegetative propagation with unrooted cuttings, direct seeding,
and other methods. The completely engineered wood grass system includes planting and harvesting. For some countries that do not have mechanized agriculture, these activities will most likely be handled manually. In mechanized agriculture, manure spreaders have been effectively used to spread cuttings and tractors have been fitted with specially made sickle bars for use in harvesting.
Good-quality and low-cost structural building material can be made from wood grass by extrusion. In this process, the fresh lignin embodied in the wood grass is used as a primary binder. The term wood grass 2×4 is applied to the product and is used generically to refer to the extruded structural material. The shape of the die can be designed so that a variety of irregular cross-section shapes can be made in different sizes. Wood grass 2×4 utilizes the original molecular structure of wood and fuses the boundaries of adjacent wood grass. This structural material resists bending and twisting, since all joints between adjacent pieces of wood grass need to be broken before such movements are possible. Furthermore, since the extrusion process works better with thin trees and fresh lignin, 1-year-old wood grass is ideal as its feedstock. Figure 43–1 shows the feedstock applications of wood grass. In addition to wood grass 2×4, products include liquid hydrocarbons, alcohols, other chemical feedstocks, and protein. The energy products include heat, electricity, low- and medium-Btu gas, pipeline gas, and liquid fuels. The wood grass production system also stabilizes soil—a tremendous implication for erosion control and watershed management in many countries.
The biological foundation of the wood grass production system is shown in Figure 43–2. The right-hand curve indicates growth on a typical plantation planted with a certain number of trees (N) per hectare. The tree is a fast-growing species and has a narrow growth curve, which is characterized by three distinct phases:
-
an initial establishment period in which the trees are developing their root structures;
-
a growth period in which the established trees undergo steady growth—a period recognized by an almost constant rate of growth; and
-
a period in which the growth rate declines as a result of competition for sunlight and nutrients. This third phase is known as closure. A point near inflection of the growth curve indicates the onset of closure.
During closure, competition for sunlight and nutrients begins to result in a decreased mean annual increment of biomass. After the onset of closure, not only is the growth rate of each individual tree affected but the average mortality also increases. In this chapter, the term closure is used to encompass all the phenomena that affect the total biomass yield per hectare. The onset of closure is defined quantitatively as the point before which the total quantity of biomass in a given area is proportional to the number of trees in that area.
The left-hand curve in Figure 43–2 shows the growth curve if twice the number of trees (2N) are planted per hectare. Initially, the quantity of biomass from 2N trees is almost twice that from N trees, but closure begins earlier. Because of the limits on the ultimate productivity of the land, the maximum quantity of biomass produced from 2N trees appears to be similar to that achieved from N trees after total closure.
Two observations can be made from Figure 43–2. First, closure begins sooner if tree density is increased. Second, the limit on the quantity of biomass that can be obtained can be ascertained from a growth curve for any density, provided N is not so low that closure does not take place. The implications of these observations are that the mean annual increment of biomass at harvest can be enhanced by increasing the planting density and that there is an optimal planting density for each rotation (age of trees at harvest), which can be determined.
For a short-rotation forest, the optimal harvesting time is some time before the onset of closure. The exact economically optimal harvesting time is a function of the discount rate, which determines the cost of money. Higher discount rates tend to favor an earlier harvesting age, since the value of future harvests is markedly reduced.
The decision to grow a particular tree as a crop can be hindered by many considerations. For example, tree growers usually cannot realize their revenue until several (or many) years after planting, and harvested forest land is expensive to clear. Thus, the use of land for wood production involves a long-term commitment. With the current analysis, however, appropriate planting density and production methods can be engineered and trees need not have these disadvantages.
Figure 43–3 shows the results of an experiment involving determination of the closure age of trees as a function of the number of trees planted (see Shen et al., 1984). As shown in the figure, if you want to harvest trees after 4 years to obtain a sufficiently large diameter, the planting density is about 1,700 trees per hectare. If you are harvesting every 3 years, you probably want to plant 6,000 to 7,400 trees. And if you are hoping to harvest every 2 years, you need a minimum of 25,000 trees. For an annual harvest, the planting density could be as high as 120,000 trees. And that’s approaching the density of corn or rice.
The actual planting density should be based on two considerations. If one is not concerned about committing the land to trees, one could start with about one tree per thousand square centimeters. After the first cutting, the coppice ratio (the number of new stems per old stem) in the second spring could be around 5 to 1. And after the second cutting, the coppice ratio could be around 3 to 1. And the ratio approaches 1 to 1 after that. The desired steady-state wood grass density may
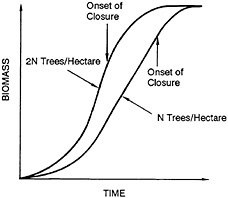
FIGURE 43–2 The onset of closure as a function of planting density for a typical plantation.
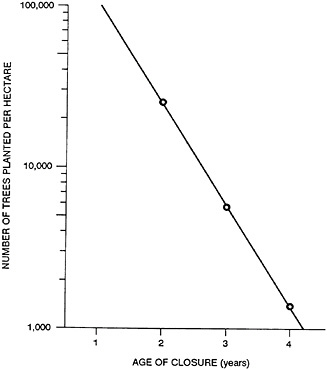
FIGURE 43–3 Closure age as a function of planting density in a forest located in the Great Lakes region of the United States.
thus be achieved after two or three cuttings. But by then the root system may be too large to be conveniently plowed under. If one is concerned about crop flexibility, the initial planting density should be chosen to control the size of the root system as well.
Wood grass production involves a degree of management as high as that in land consumption activities such as those found in urban areas or in association with roads, mining, and water impoundments. Wood grass is part of modern agriculture, except that in addition to its food and feed products, it produces energy and materials as well. Figure 43–4 shows wood grass grown at a density of one per thousand square centimeters for feed, electricity, and steam.
To determine the economics of wood grass production, we analyzed a plantation of wood grass (Populus) in the Pacific Northwest of the United States, allowing a range of uncertainties in the wood grass yield. The cost to produce the Dushen as a function of the discount rate (the cost of money) was estimated. At a 7% real interest rate, for instance, the range of uncertainties is from $12.00 to $25.00 per dry ton. At that rate, the average wood cost is $18.00 per ton, which is $1.00 per million Btu measured in energy value (Jones and Shen, 1982; Shen 1983; Shen et al., 1982; Vyas and Shen, 1982).
Which areas are best for growing wood grass? In the United States, the states bordering the five Great Lakes, the Eastern United States, and the Pacific North-

FIGURE 43–4 Wood grass in Livingston, California, grown as a low-cost source of protein for poultry feed and as a biomass fuel for cogeneration. Photo courtesy of Foster Farms.
west all have excellent soil and climate to support the wood grass production system. At the lower bound of the yield, wood grass competes economically in the Corn Belt at current prices. Its most important role, however, is its potential use in energy, materials, and feed markets of the world.
Due to differences in agricultural systems and practices and in topographical and climatic conditions, there are different schemes for producing Dushen feedstock using the wood grass production system. There are also considerations concerning capital versus labor intensity and availability. These can be illustrated by comparing Dushen production in two countries: the United States and China. Field preparation is mostly mechanical in both the United States and China. Planting is mechanical in the United States and manual in China. Figure 43–5 shows farmers planting wood grass at a density of four per thousand square centimeters in Southern China. In both countries, the cuttings are planted vertically or are placed horizontally on the ground and covered with a moisture-preserving mulch such as wheat straw. A manure spreader has proved effective in random horizontal planting. Vertical planting is used for rice seedlings or tree cuttings. Each cutting may produce more than one tree, depending on how many buds are present on the cutting and on the survival rate. The herbicide and fertilizer applications are mostly mechanical
in the United States and manual in China. Both countries use chemicals and manure as fertilizer, but the Chinese use more manure, whereas chemicals predominate in the United States. Harvesting is mechanical in the United States and is still manual in China. The Dushen harvester chops wood grass directly into 2.5-centimeter pieces, the wet Dushen. The dry Dushen is produced if the wood grass is crushed with rollers before it is chopped in the harvesting process. A single harvester is needed to produce the Dushen.
The near-term uses of wood grass in the United States are heating, animal feed, industrial fuel, the coproduction of steam and electricity, and, most importantly, structural material—material that does not require 50 to 100 years of growing, such as pine trees. The mid-term applications include pipeline-quality gas production, which the Gas Research Institute (GRI) is developing, and liquid fuels. In his work on pipeline-quality gas performed for GRI, David Chynoweth of the University of Florida has found that wet Dushen approaches the performance of the most digestible cellulosic samples used as reference in his experiment. The long-term applications include its use as chemical feedstock, which has the highest market value.
In China, the regions of most intensive applications are located in the Northeast, where there is need for industrial fuel and heating, in Eastern China, and in Northwestern China. The near-term applications are cooking, heating, industrial

FIGURE 43–5 Establishment of a wood grass multipurpose plot in Fujian Province of Southern China. Chicken manure diluted with rice hull is used as fertilizer. The wood grass will be used as cattle feed. Photo courtesy of Yangting Farm.
fuel, cogeneration, animal feed, and construction materials. As in the United States, its long-term use is as chemical feedstock. There are tremendous possibilities here for the production of various materials, including building material, animal feed, and chemical feedstock.
At present, the Dushen products from the wood grass production system look attractive because of the system’s rapid return on investment. In the long run, the soil conservation characteristics of the production system may be the most important benefit of wood grass. We recently analyzed the impact of large-scale production in the United States and found wood grass to be a significant soil-conserving crop (Shen and Turhollow, 1983). In contrast to conventional food crops, the wood grass system provides surface coverage throughout the year. In addition, the root system provides a soil-stabilizing matrix. Furthermore, selected wood grass configurations reduce groundwater runoff, increase groundwater infiltration, and recharge groundwater reservoirs.
In designing the wood grass production system, we first applied traditional genetic selection to hybrid trees, which were cloned for a number of attributes. We focused on a narrow growth curve and a rapid closure phase. The average growth curves of course are those under selected fertilization and other soil amendment schedules for a specific species-site match. For all clones selected for these two attributes, the coppice growth curves are examined, since sustained steady-state coppice growth is our objective function. In the next step, the selection criteria include disease resistance, drought resistance, and ease of establishment. Then, depending on the desired market, additional attributes directly related to product properties are identified. In practice, this last step is performed at a very early stage for general screening, because the objective of the biological engineering is really the product. On the other hand, without the growth attributes established in the genetic selection, the biological system has no basis.
Through the wood grass production system, we have shown that biological engineering with components based on genetic selection, species propagation techniques, modern agriculture, and chemical or mechanical processing methods could produce energy and novel materials that fit within the framework of the existing market infrastructure. This applied engineering approach will allow multidisciplinary teams to produce products and technologies that can be marketed more rapidly than most new technology, while taking environmental benefits into full consideration as a primary long-term objective.
CONTOUR HEDGEROWS
Contour hedgerows proposed for the highland regions of Nepal, Pakistan, India, and other countries (Benge, 1984) constitute a biologically engineered system for the production of reforestation planting stock. The Department of Soil and Water Conservation of Nepal estimates that between 30 and 75 tons of soil are washed away annually from each hectare of deforested land. This means that Nepal altogether loses as much as 249 million cubic meters of soil per year to India (Cool, 1980). The contour hedgerows alleviate the problem of the short supply of appropriate planting stock, and at the same time effectively check erosion, reduce
FIGURE 43–6 Cross-sectional view of hedgerows.
groundwater runoff, increase ground water infiltration, recharge groundwater reservoirs, and produce the by-products of fuelwood and fodder.
A typical design of a clonal hedgerow system entails the construction of hillside ditches and the planting of two rows of trees or shrubs on the rise (bund) of the ditches, which follow the contour of the hillsides, and one row planted in the depression (Figure 43–6). The hillside ditches increase the system’s effectiveness in controlling erosion while increasing water percolation into the soil. Soil erosion control is more effective when the spacing of the plants is alternated between hedgerows (Figure 43–7). Soil could still be eroded from around the stems. It is desirable to place some type of biomass uphill and behind the live stems to make a more effective barrier to hold the soil and accelerate the formation of natural terraces. Rows of plants on the rise and in the ditch would provide greater access to the forage by browsing animals, if the functions of plant material production and the cut-and-carry system of forage production were phased into a browse-pasture system.
FIGURE 43–7 The spacing of plants is alternated between hedgerows.
According to Benge (1984), 1 hectare of contour hedgerows could provide enough plant material to reforest approximately 600 hectares (at a spacing of 4 meters between trees) the first year and 3,000 hectares the second year. To arrive at this production figure, a 6-centimeter in-row spacing of cuttings, a distance between rows of 0.5 meter and 0.75 meter, respectively (center to center—3 rows), and a spacing between hedgerows of 5 meters (edge to edge) was used (Figure 43–8). The production capacity of this contour hedgerow system used for clonal propagation of planting stock could be as high as 450,000 cuttings per hectare per year. The actual per-hectare production rate for cuttings from contour hedgerows depends upon the genetic capacity of the clones as well as the environmental conditions in which they are grown. The system benefits from high in-row density and edge effect, i.e., plants near the edge of each hedgerow benefit from additional sunlight and nutrients outside the hedgerow.
It is important to incorporate genetic diversity in contour hedgerow systems that cover large areas by the use of a wide variety of species or provenances in order to decrease the possibility of widespread disease and insect infestations and the production losses that would result. Many tree species that have been propagated from cuttings either will not develop a tap root or will develop a modified tap root with extensive lateral roots but without deep penetration into the subsoil. Therefore, these trees will draw nutrients only from the upper soil strata, which may already be nutrient poor from the mining effect of many food crops and other surface feeding plants. Thus, it may be desirable to plant between the contour hedgerows trees that have been propagated from seeds and that develop a deeper tap root system (Figure 43–8). This would increase the per-hectare productivity of the
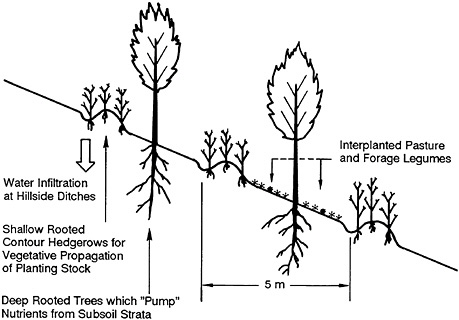
FIGURE 43–8 Complete biologically engineered system of contour hedgerows.
system, since these trees would pump from a deeper soil strata nutrients that may not be available in the shallower soil.
REFERENCES
Benge, M.D. 1984. Contour hedgerows for soil erosion control and planting stock, forage and fuelwood production in highland regions. Pp. 117–123 in Energy from Biomass: Building on a Generic Technology Base. Proceedings of the Second Technical Review Meeting. Report No. ANL/CNSV-TM-146. Argonne National Laboratory, Argonne, Ill.
Cool, J.C. 1980. Stability and Survival—The Himalayan Challenge. Ford Foundation, New York. 22 pp.
Jones, P.C., and S.Y.Shen. 1982. A Framework for Evaluating the Economics of Short-Rotation Forestry Research and Development. Report No. ANL/CNSV-35. Argonne National Laboratory, Argonne, Ill. 56 pp.
Shen, S.Y. 1982. Wood Grass Production Systems for Biomass. Paper presented at the 1982 Midwest Forest Economist Meeting, Duluth, Minn., August 17–19, 1982.
Shen, S.Y. 1983. Regional Economic Impacts of Woody and Herbaceous Biomass Production. Paper presented at the 1983 Joint National Meeting of the Institute of Management Science and the Operations Research Society of America, Chicago, April 24–27, 1983.
Shen, S.Y., and A.F.Turhollow. 1983. Regional impacts of herbaceous and woody biomass production on U.S. agriculture. Pp. 207–234 in Symposium Papers: Energy from Biomass and Wastes VII. Presented January 24–28, 1983, Lake Buena Vista, Fla. Institute of Gas Technology, Chicago.
Shen, S.Y., P.C.Jones, and A.D.Vyas. 1982. Economic Analysis of Short-Rotation Forestry. Paper presented at the 1982 Joint National Meeting of the Operations Research Society of America and the Institute of Management Sciences, San Diego, Calif. October 1982.
Shen, S.Y., A.D.Vyas, and P.C.Jones. 1984. Economic analysis of short and ultra-short rotation forestry. Resour. Conserv. 10:255–270.
Vyas, A.D., and S.Y.Shen. 1982. Analysis of Short-Rotation Forests Using the Argonne Model for Selecting Economic Strategy (MOSES). Report No. ANL/CNSV-36. Argonne National Laboratory, Argonne, Ill. 50 pp.