CHAPTER 47
DIVERSE CONSIDERATIONS
THOMAS E.LOVEJOY*
Executive Vice President, World Wildlife Fund, Washington, D.C.
Before dwelling on the economic, social, and political problems that are fundamental to present problems and future prospects, there are two aspects of natural science that require attention but have not yet been mentioned in this volume: the abundance of relatively few of the many species on Earth and the limitations deriving from our shallow knowledge of diversity.
SPECIES ABUNDANCE
One of the great questions of biological science arises when biological diversity is viewed through ecological glasses: Why are ecosystems generally made up of a large number of species of which only a few are abundant? While the roster of rarer species in an ecosystem is much longer in tropical regions than at higher latitudes, there is a general tendency to accumulate large numbers of species in all but the most simple ecosystems.
This pattern can be generally portrayed by graphing the relative abundance (for example, the percentage of total individuals or of total biomass) of species against the order of species from most to least abundant (Figures 47–1 and 47–2). In early successional communities, there is a smaller number of species and the most abundant ones constitute a larger fraction of the community, i.e., are more dominant.
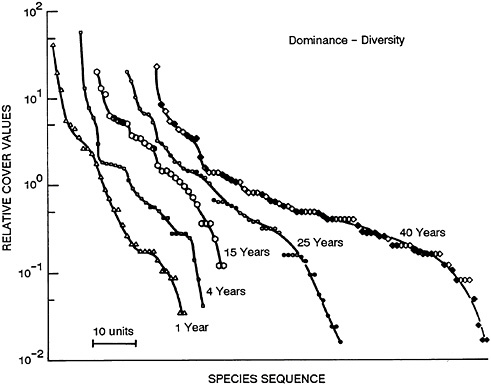
FIGURE 47–1 Patterns of the relative abundance of species at five different stages of abandonment in old fields in southern Illinois. The patterns are expressed as the percentage that a given species contributes to the total area covered by all species in a community, plotted against the species’ rank and ordered from most to least abundant. The symbols are open for herbs, half open for shrubs, and closed for trees. From May, 1985, with permission.
Figure 47–1 shows such an accumulation of species after an old field was abandoned in southern Illinois. Conversely, if a community is subjected to stress, e.g., from temperature, toxic substances, or the arrival of an alien species, this process is reversed: diversity declines and a small number of species become dominant and are often called nuisance growths. Figure 47–2 shows such a response to heavy fertilizer applications in experimental grass plots in England. There are excellent aquatic analogs to these patterns, many of which have emanated from the work of Ruth Patrick and colleagues at the Philadelphia Academy of Natural Sciences (Patrick, 1949, 1975; Patrick et al., 1969).
The meaning of pattern in relative abundance and diversity in communities has only begun to be initially understood and remains largely tantalizing (Fisher et al., 1943; Hutchinson, 1958; MacArthur, 1957, 1965; May, 1975; Patrick 1961, 1984; Preston, 1948). Furthermore, it is very difficult to understand the function of any of the large number of rare species in an ecosystem precisely because they are so rare. Indeed, there has been a tendency when considering endangered vertebrate species to think of them as ecologically nonfunctional or even ecologically extinct. I believe this is a highly dangerous and inappropriate attitude not only because
rarity in an ecosystem is in fact the common condition but also because the true meaning of rare species in the system cannot easily be assessed for a moment but only when viewed over periods of environmental change.
The role of an obscure species of yeast in the genus Cryptococcus (Brunner and Bott, 1974) is very telling in this regard. This species is rare in the aquatic communities of eastern Pennsylvania, presumably because of competition from other species. Its role and value are not immediately apparent. When either natural or human-generated mercury contamination occur, this yeast suddenly is at great advantage, because it is able to short-circuit a particular metabolic pathway along which mercury has toxic effects for organisms generally. The yeast is actually able to reduce methyl mercury to the elemental state and store the quicksilver in a vacuole that it subsequently deposits on a convenient surface such as a rock. Under conditions of elevated mercury concentration, the yeast temporarily becomes very abundant, while many previously abundant species in the community are depressed and diversity declines. The yeast literally cleans up the mercury contamination, thereby making itself rare again.
The number of species that respond in this fashion to environmental change in a way that keeps them rare is probably not great. Nonetheless, all rare species in ecosystems are likely to be able to respond with population increases, given the
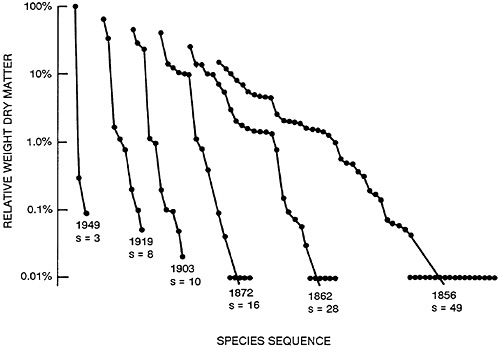
FIGURE 47–2 Changes in the patterns of relative abundance of species in an experimental plot of permanent pasture at Parkgrass, Rothamsted, England, following continuous application of nitrogen fertilizer since 1856. (Species with abundance less than 0.01% were recorded as 0.01%.) Note that here time runs from right to left, so that the patterns look like the successional patterns of Figure 47–1 running backward in time. From May, 1985, with permission.
right changes in environmental conditions. Many of these rare species are likely to be part of local habitats at any given moment, because the ecological conditions under which they flourish have occurred sufficiently frequently or recently for them to be represented. This means that the rare species in a sense reflect a variety of past conditions and confer some measure of ability for the ecosystem to respond to changing conditions.
Survival strategies of this kind have as yet only been superficially explored. Nevertheless, the role rare species may play under changing conditions suggests that the accumulation of species in an ecosystem, while perhaps only an accident of the history of any particular system, can turn out to be of value. We thus have the beginning of an argument in favor of rare species in general—species attuned to conditions that are not prevalent at the moment but that may well return.
At the present time, we are faced with a situation in which not just local ecosystems respond to stress by losing their diversity and by simplification; rather, the entire biosphere is subject to impoverishment. This inevitably means that individual ecosystems in a number of instances will be less diverse, because some of the species that would otherwise form part of the long list of rare species are doomed to extinction.
LIMITATIONS OF OUR KNOWLEDGE
To approach the global problem from a scientific perspective, we are immediately confronted by the second problem commanding attention, namely, the limitations deriving from our relatively shallow knowledge of flora and fauna. Recent discoveries of insect diversity in the canopy of South American tropical forests (Erwin, 1982, 1983, and Chapter 13 of this volume; May, 1986) warn us that we do not even know the extent of biological diversity on our planet to the nearest order of magnitude (Wilson, 1985, and Chapter 1 of this volume). Given such a poor inventory of life on Earth, biologists can say relatively little about which species occur where, which are in danger of extinction, where protected areas should be established, and where heavy environmental modification for development is permissible.
What is desperately required is a revitalization of the science of biological systematics, with all the ancillary strength modern technology and molecular biology can provide, combined with a crash program of biological exploration. A decade or two of intensive biological mapping is needed while development is halted, or at least severely curtailed, in areas that are evidently the richest but least explored.
SOCIOLOGICAL ISSUES
I would now like to switch very abruptly to matters of the social sciences, which drive our societies in a multiplicity of little-appreciated ways. Consider the price of shoes, which on the face of it, seems but a matter of domestic detail far removed from the threat of massive extinctions in the tropics. Yet the U.S. shoe industry, far from strong, has been seeking tariff protection from the Brazilian shoe industry. If the U.S. government were to respond in a protectionist manner, what might
be the consequences? Brazil’s struggles with its international debt would be the more difficult. Furthermore, its drive to increase exports would be even stronger, and as already apparent, the emphasis on export crops would lead to the intrusion of small farmers into the vital remaining natural areas.
Shoe prices and similar isolated indices may not be a large factor. They might even prove to have no measurable impact on conservation, or they could even be a positive increment, if decreased Brazilian shoe exports reduced incentives for cattle ranchers. The point, however, is that a myriad of such unrelated issues swirl about our everyday lives and together with decisions made on Wall Street and in Washington, have a great deal to do with fueling the biological diversity crisis. Each and every one of us is more tightly connected to the global conservation problem than generally realized. This Gordian Knot of economic and political linkages deserves far greater attention than it has received up to the present time.
Of course, there remain the great overarching issues such as population growth. I feel this acutely as I write from the great Indian subcontinent. When Indira Gandhi came to power in 1966, there were 480 million Indians, and when she left in violence in 1984, there were 250 million more. Since this trend continues, my environmental colleagues in New Delhi worry deeply about the long-term security of parks and reserves.
The problem of population growth sometimes seems so enormous as to be intractable. Even with stringent birth control, future population growth is inevitable because of the very youthful age structure of populations in developing countries. Short of coercion, the only permanently ameliorative approach is to improve living standards while providing birth control devices and aggressive public education programs. That is not an instant solution, because human conditions cannot be improved overnight and a response in the form of reduced fertility does not happen right away either. There is no question that the association between development, population control, and conservation needs our continuing preoccupation.
ECONOMIC PRESSURES
Looming large in the picture is the massive international debt, now expressed in trillions of dollars. This economic goliath binds the industrialized and developing nations as surely as their shared interest in the protection of biological diversity. In many instances, all earned foreign exchange must be devoted to interest payments. This leaves little capacity to use foreign exchange to invest in the economic growth so necessary for improved living standards and all that goes with them, including a greater awareness of, and willingness to do something about, conservation problems.
For some time, I have advocated the notion that some portion of the debt be allocated to these pressing problems of biological diversity. The money is already in the developing nations where the greatest conservation problems are, and much of it is unlikely to be returned to the lenders. Why not then arrange for some form of conservation credits where tropical debtor nations can use their own local currencies for national conservation projects? Already there is interest in buying debt at market-dollar values considerably less than face value and redeeming the
paper in local currencies. Legislation is developing to encourage donation of such debt by commercial banks for international purposes. Whatever form these activities take, it must of course be fully sensitive to national sovereignty, but this should not be difficult to achieve since conservationists in the developing nations are as ardent as conservationists anywhere.
Perhaps the most important point about the possibility of exploiting the international debt is not so much the example itself but rather the need to constantly seek new ways to elevate and expand the conservation effort. The scale of the problem is so great compared to the conservation efforts now under way that we will not succeed in safeguarding the majority of biological diversity without major innovation and major infusion of resources. It is all too easy to feel content with some hard-won conservation victory when problems crop up all around us like warriors sown from dragon’s teeth.
From time to time in a flight of apparent silliness, I advance the notion of buying a major piece of west African real estate with substantial populations of gorillas and chimpanzees and setting up a new political entity, the “Kingdom of the Apes.” I do so not because such a possibility really exists. Rather, I do so because however fanciful the idea of a great silverback male gorilla being present at the UN General Assembly, it does suggest how apart we have set ourselves from other living things, and how awry our system of governing the world really is.
We and our fellow vertebrates are largely along for the ride on this planet. If we want to perpetuate the dream that we are in charge of our destiny and that of our planet, it can only be by maintaining biological diversity—not by destroying it. In the end, we impoverish ourselves if we impoverish the biota.
REFERENCES
Brunner, R.L., and T.L.Bott. 1974. Reduction of mercury to the elemental state by a yeast. Appl. Microbiol. 27(5):870–873.
Erwin, T.L. 1982. Tropical forests: Their richness in Coleoptera and other arthropod species. Coleop. Bull. 36(1):74–75.
Erwin, T.L. 1983. Beetles and other insects of tropical forest canopies at Manaus, Brazil, sampled by insecticidal fogging. Pp. 59–75 in S.L.Sutton, T.C.Whitmore, and H.C.Chadwick, eds. Tropical Rain Forest: Ecology and Management. Blackwell Science Publishers, Palo Alto, Calif.
Fisher, R.A., A.S.Corbett, and C.B.Williams. 1943. The relation between the number of species and the number of individuals in a random sample of an animal population. J. Anim. Ecol. 12:42–58.
Hutchinson, G.E. 1958. Concluding remarks. Cold Spring Harbor Symp. Quant. Biol. 22:415–427.
MacArthur, R.H. 1957. On the relative abundance of bird species. Proc. Natl. Acad. Sci. USA 43:293–295.
MacArthur, R.H. 1965. Patterns of species diversity. Cambridge Philos. Soc. Biol. Rev. 40:510–533.
May, R.M. 1975. Patterns of species abundance and diversity. Pp. 81–120 in M.L.Cody and J. M.Diamond, eds. Ecology and Evolution of Communities. Belknap Press, Harvard University, Cambridge, Mass.
May, R.M. 1985. Population dynamics: Communities. Pp. 31–44 in H.Messell, ed. The Study of Populations. Pergamon Press, Rushcutters Bay, Australia.
May, R.M. 1986. How many species are there? Nature 324:514–515.
Patrick, R. 1949. A proposed biological measure of stream conditions based on a survey of the Conestoga Basin, Lancaster County, Pennsylvania. Proc. Acad. Nat. Sci. Phila. 101:277–341.
Patrick, R. 1961. A study of the number and kinds of species found in rivers in eastern United States. Proc. Acad. Nat. Sci. Phila. 113(10):215–258.
Patrick, R. 1975. Structure of stream communities. Pp. 445–459 in M.L.Cody and J.M.Diamond, eds. Ecology and Evolution of Communities. Belknap Press, Harvard University, Cambridge, Mass.
Patrick, R. 1984. Some thoughts concerning the importance of pattern in diverse systems. Proc. Am. Philos. Soc. 128:48–78.
Patrick, R., B.Crum, and J.Coles. 1969. Temperature and manganese as determining factors in the presence of diatom or blue-green algal floras in streams. Proc. Natl. Acad. Sci. U.S.A. 64:472–478.
Preston, F.W. 1948. The commonness, and rarity, of species. Ecology 29:254–283.
Wilson, E.O. 1985. The biological diversity crisis: A challenge to science. Issues Sci. Technol. 2(1):20–29.
CHAPTER 48
THE CONSERVATION OF BIODIVERSITY IN LATIN AMERICA
A Perspective
MARIO A.RAMOS
Director, Wildlife Program, Institute Nacional de Investigaciones, Sobre Recursos Bioticos, San Cristobal Las Casas, Chiapas, Mexico
The increasing awareness of the need to preserve biological diversity in the world is demonstrated by the meetings that have been convened in developed countries. In the United States, for example, there were the Smithsonian Institution meeting in December 1985, the World Wildlife Fund meeting in September 1986, the National Academy of Sciences and Smithsonian Institution joint meeting in September 1986, and the meeting of the New York Zoological Society in October 1986. All are clear examples of this general trend. Although this awareness has come slowly to countries in the Third World, some important steps have been taken. In Latin America, for example, there has been a dramatic increase in the number of established protected areas (Harrison et al., 1984).
What has been slow to come, in my opinion, is recognition that the preservation of the biological diversity in the world is a shared commitment between rich and poor countries and that major responsibilities fall into the hands of the countries where this diversity is found. Since the greatest diversity exists in the tropical areas of the world, these responsibilities generally lie within the developing countries. In these countries, however, social, economic, and political problems often make conservation of their diversity very difficult. The riddle of balancing development; stability in economic, social, and political terms; and conservation of their natural resources is difficult for any of the countries to solve by themselves. Because Mexico provides an instructive example of these problems in the Latin American region, I will summarize the general conditions that determine the context in which this
conservation is now and will be taking place in the near future. I will also consider means to achieve the sharing of responsibilities among countries in the world. Here I pull together the knowledge and experience of many other people working in the region who have expressed their views in the recent meeting during the World Wildlife Fund’s Twenty-fifth Anniversary.
CONSERVATION AWARENESS
In Mexico, there has been a growing awareness of conservation issues, and new leadership has emerged within the scientific community and nongovernment organizations (NGOs) such as Monarca A.C. and Pronatura A.C. The new leaders have been responsible for the inclusion of environmental considerations in part of the government planning process and in political agendas. NGOs have flourished in the last 5 to 10 years. At present, there are probably 200 of these organizations located primarily in major cities throughout the country. In 1985, 22 NGOs established a federation in Mexico City. These organizations, supported by the scientific community, have pushed for improving environmental legislation and the establishment and protection of the conservation units within different habitats. In addition, they have discussed wildlife trade and have helped to organize and spread the conservation message among citizens in the country. A striking example is the controversial establishment and management of the Montes Azules Biosphere Reserve in Chiapas. International organizations such as the World Wildlife Fund/ Conservation Foundation and the Nature Conservancy International Program are supporting this grass-roots movement through direct economic support, official recognition, training of personnel, and infrastructure to help NGOs carry out their functions.
THE SOCIOECONOMIC ENVIRONMENT
Mexico is a clear example of a country in the process of development. It has slowed down its population growth from 3.3 to 2.3% per year, and it has two basic faces: an industrial one and a rural one. In addition to the problems stemming from industrial development, it has large rural and urban populations living in very marginal conditions. A comparison between 1940 and 1980 figures is revealing. There were about 19 million Mexicans in 1940, and in 1980, there were 68 million. By 1985, the population exceeded 79 million. Four million people lived in urban areas in 1940 and 41 million by 1980—a 10-fold increase, whereas rural populations changed from 15 to 27 million during the same period. Now, 65% of the population is concentrated in the central and southern part of the Mexican plateau (altiplano), where 80% of the total industry is also located.
In addition, Mexico probably has the largest city in the world, Mexico City, which is home to about 18 million people. The city faces many environmental problems related to its size. Among the more pressing ones are air pollution control, solid waste and sewage disposal, water supply, encroachment on green areas, and the movement of rural populations into urban areas, where they live under extremely marginal conditions in huge slum areas within and around the city.
The most important and fastest-growing industry has been the petrochemical industry, which generates not only oil, plastics, pesticides, and fertilizers but also air, water, and soil pollution. Modern industry generates large quantities of toxic materials, commonly storing them on site, dumping them down sewers or landfills, or burning them without regard for environmental concerns.
The country also faces enormous economic problems. Most of the capital is concentrated in a few hands, leaving the majority of people living under substandard conditions. Inflation has increased tremendously; consumer prices rose no less than 1,822% between late 1979 and February 1986 (Story, 1986), and real salaries dropped 27% in 1985 alone (Tello, 1986). Unemployment and underemployment have both soared, making it difficult for an important segment of the population to cope with the economic crisis. Devaluation of the Mexican currency has been rapid during the last 10 years, becoming critical in the last 4. A comparison between 1976 and 1986 figures is revealing. Our currency was worth 19.95 pesos to 1 U.S. dollar at the end of 1976, 473.60 to 1 in March 1986, approximately 700 to 1 in August 1986, and still continues to drop.
The international economic crisis has brought additional problems to Mexico. It was because of the impacts in Mexico that the international community first recognized the major problems confronting Third-World countries in relation to the international debt. A great part of the borrowing from the international community was wasteful or unjustified; it was not used for financing investment but for consumption and government budget deficits. Capital flight increased. Estimates from the 1979–1982 period suggest extraordinary amounts of capital flight in Mexico—about US$55 billion (Dornbusch and Fischer, 1986). The issue of the foreign debt has become politicized, partly because of the stagnation of the economy, which has a growth rate of less than 1%, and because the inflation appears to have become structural. Inflation in 1986 reached 105%, and hyperinflation seems to be knocking at the door. The situation has been exacerbated by trade deficits, showing the need for deep and significant structural changes.
In general terms, the foreign debt is being serviced but at increasing social costs (Lustig, 1986). The international commercial banks played a significant part in causing the economic crisis by imprudent lending to imprudent government and private-sector borrowers in Mexico, but they have paid a relatively low price for doing so. The country now suffers from a drop in economic activity, a worsening of the standards of living, and such a large increase in the foreign debt that it can not even pay the interest. Estimates for the foreign debt servicing bill reached US$11.5 billion in 1986. There have been some structural adjustments, but they have not gone as far as the international community requests. However, the Mexican government should get credit for taking some initiatives, such as the privatization of decentralized national companies. Changes of this magnitude, as requested by the International Monetary Fund, the World Bank, and other banking organizations, do not happen overnight.
A major concern of environmentalists has been the possibility that payment of the foreign debt may generate additional environmental problems and that the environmental costs may be too high. The reaction of the government so far has been similar to the strategy for payments of the foreign debt: negotiating resched-
ulings for future payments and hoping that someone will then solve the problem. Present administrators do worry about the environmental problems, but not at the high level or with the priority required to solve them. They plan and carry out economic and social development activities without major environmental constraints, letting others worry about them in the future.
Of special environmental concern is the production of oil—the main Mexican export product. For years, the Mexican government has depended on oil export revenues to strengthen its economy. Major loans were given and are still coming in now from the international banking community to support exploration, infrastructure building, and exploitation of oil wells, without apparent concern for environmental issues. However, oil prices keep dropping—from more than US$30 per barrel in 1984 to US$20 in early January 1986, approximately US$12 in late 1986 and US$15 in mid-1987. Story (1986, p. 30) reported, “Revenues from oil exports declined by $2 billion (in 1985) compared with a 1984 total of 16.5 billion….” The total loss to Mexico due to the decline in oil prices and lower sales volume (in 1985) was US$3.3 billion. The situation became even worse in 1986 and 1987.
There has also been increased pressure from the developed world to obtain as many natural resources from Third-World countries as cheaply as possible. This has generated conflicts that are building and will continue to do so if present conditions prevail. The trade of wild flora and fauna at the international level is a clear example. Although Mexicans try to control the supply of natural resource goods, the international community must help to control the demand from their countries. Prices soar into the thousands of dollars for rare and endangered species of wild animals and plants, and while the demand exists, someone in the developing world will find, trap, and sell them.
THE POLITICAL ENVIRONMENT
The politics of the environment and its conservation is poorly defined in Mexico. There are no clear policies. Legislation is frequently a response to conflicting priorities and emergency situations. In some areas where there are laws (e.g., pollution control), there are no regulations to implement them (Albert, 1985). In other areas, the law is rarely applied, and when it is, the sanctions and fines are ridiculously low (Ramos, 1985). Government officers are politically appointed, and communication among them or their agencies is limited, because of strong territoriality at that level.
Traditionally, the management of environmental issues in Mexico was dispersed among different ministries. For example, pollution and its control were under the Health Ministry and fisheries under the Fisheries Ministry, while some protected areas (e.g., parks) were under the Agriculture Ministry and others under the Urban Development Ministry. Since late 1982, most of the government’s environmental considerations were brought under a single ministry—the Urban Development and Ecology Ministry (SEDUE). Although the authorities of SEDUE have tried to cope with Mexico’s environmental problems, they have had little success. Unfortunately, authorities at the cabinet level and below have changed three to five times since
SEDUE’s establishment, producing inevitable instability in the ministry. All ministries are under the control of the Budget and Planning Ministry (SSP), which does not have staff well trained in environmental issues and thus faces serious conflict when trying to establish priorities. At the budgetary level, ecological issues are at the bottom of the agenda, if they are included at all. Changes in priorities need to start at this level.
During the past 65 years, the Mexican government has essentially been run by the official party—Partido Revolucionario Institucional (PRI). The environmental platform of the different political parties, including the official one, are not well known and in some cases, probably do not exist. Recently, before the 1982 presidential elections, Mexico’s current president, Miguel de la Madrid Hurtado, did make references to environmental policy during his political campaign; he included them in his National Development Plan for 1982–1988. An evaluation of his accomplishments on the subject is still to be made. The democratization process now occurring in the official political party may facilitate the inclusion of more environmental considerations in the official party platform in the years to come. Environmental policy statements were also issued by the left-wing political parties in late 1982. It seems likely, however, that environmental policy and its implications will play a more powerful role in the elections to come in 1988 and beyond. The environment is becoming an economic issue politicians cannot afford to ignore. The health impact of the thermal inversions during the winters of 1986 and 1987, for example, are just now being appreciated. Environmentalists should work to improve their performance and relations with political groups now in order to be effective in the near future.
In terms of international affairs, our foreign policy has not been very active. In fact, it was more rhetorical than real until the 1970s when it began changing. Unfortunately, with the current economic crisis, the Mexican international profile has in general been diminishing. Furthermore, Mexico’s definition of foreign policy depends on the president in office, and it changes with every new president elected. The situation is improving, however. The new leadership emerging from the official political party seems to be more technology oriented and apparently better able to cope with international agreements and relations.
Mexican international environmental policy is not well defined. Mexico has signed some international conventions, e.g., the Convention on Nature Protection and Wildlife Preservation in the Western Hemisphere (the Washington Convention) and the Convention Concerning the Protection of the World Cultural and Natural Heritage. However, it has not signed other, very important ones, e.g., the Convention on Wetlands of International Importance especially as Waterfowl Habitat (Ramsar Convention), the Convention on International Trade in Endangered Species of Wild Fauna and Flora (Cites Convention), and the Convention on the Conservation of Migratory Species of Wild Animals (Bonn Convention) (UNEP, 1984). The acceptance of many of these conventions generally depends on favorable opinions from different agencies within the government (e.g., SEDUE, the Foreign Relations Ministry, and the Mexican Senate), which have trouble reaching agreement. Analysis of the conventions has not been completed, and their value is not well understood.
THE TECHNICAL ENVIRONMENT
From the scientific and technical standpoint, Mexico has limited capabilities for environmental protection. Of primary importance, its scientific community is slowly eroding because of attrition. Salaries in this sector have dropped in real value to 30% of what they were 7 years ago. Operating budgets for research institutions have dropped dramatically too. Although these budgets for 1986 were the same as those for 1985, inflation has increased to about 65 to 70%, and because of dropping international oil prices, they suffered additional cutbacks of 23 to 27% for the whole year. This left most research institutions stranded. The personnel and infrastructure budgets have not been funded for the last 3 years, except in very selective cases approved by the Planning and Budget Ministry (SSP). This means that there are no possibilities for hiring personnel or replacing old infrastructure. Job openings are frozen as soon as they become vacated so that funds for those personnel costs can be used for other government expenditures. All these cutbacks are especially critical in research centers outside the main cities. The government has tried to decentralize some areas, science included, but the present economic and structural crisis has not helped this process.
Traditionally, most of the scientific endeavor in Mexico has been oriented toward basic sciences. Applied science does not carry the prestige or the curricular weight when academic evaluations are made. As in other countries, basic research tends to follow international academic fashions, and at present, molecular triumphalism overrides systematics and taxonomy, biogeography, and paleontology—some of the sciences necessary for mapping biological diversity on Earth.
In addition, Mexican scientists have commonly remained outside the political sphere and to some extent have neglected their roles as citizens. Scientists have not taken serious and clear stands on political issues or on policy related to the environment. Most of the technical work in this area has been designed or implemented by professionals in other areas such as economics, sociology, agriculture, or policy, without a clear understanding of the biologist’s role and the importance of adequate scientific expertise in the decision-making process.
Moreover, only a few institutions are qualified to address issues of conservation and development, topics that commonly require interdisciplinary work and the infrastructure to carry basic and applied research to develop new technologies and apply the new knowledge to human communities. Among the most prominent institutions with these capabilities are the Instituto Nacional de Investigaciones sobre Recursos Bioticos (INIREB), the Centro de Investigaciones de Quintana Roo (CIQRO), the Instituto de Ecologia (IE), and to some extent, the Laboratorio Natural las Joyas de la Sierra de Manantlan in the University of Guadalajara (LNLJ). These organizations have been conducting research concerning conservation and development issues in well-defined natural areas. They are all very young organizations but have promising, capable people on their staffs.
The role of environmental education has been underestimated at both professional and nonprofessional levels. University curricula lag behind technical knowledge, and academic standards have been dropping for the last two decades. Conservation as a subject has not been commonly included in university curricula
either in biology or in the social sciences. Important changes are necessary in this area too.
Amidst all this difficulty, we should recognize that as correctly pointed out by the International Union for the Conservation of Nature and Natural Resources in its World Conservation Strategy (IUCN, 1980), the way out is the achievement of a proper balance between conservation of natural resources and socioeconomic development. Economic development needs to offer alternative options for the human populations living under marginal conditions without damaging the natural resource base. Examples of this kind come from ancient Indian cultures, such as the Mayans in southern Mexico and Central America and many of the Indian groups in Brazil. Recent examples are difficult to find, but important efforts are being made in the Biosphere Reserve in Sian Ka’an in Quintana Roo, Mexico, and in the Planada project in Colombia. We have to recognize that there must be a sustainable balance between human populations and natural resources.
CONSIDERATIONS FOR SHARED RESPONSIBILITIES
Conservation of biological diversity on a global scale cannot take place only through the efforts of the developed world. Moreover, the general conditions of countries like Mexico and the real possibilities of preserving biological diversity under those conditions appear very grim. Major changes would have to be made within Mexico and in the international community to produce well-defined strategies designed to avoid or diminish the possibility of failure and the duplication of efforts. Both need to concentrate on specific plans of action for successful conservation and development efforts. Thus, although there is a tremendous need to address the issues of conservation more globally, it is essential to include the Third-World countries as equal partners in the process.
Mexico will have to respond effectively to the great challenge that includes the need for structural changes, constituency building, legislative reforms, and bureaucratic implementation. We will also require strong support from foreign and international groups. There are many ways countries like Mexico could be helped. Economic support is critical but is not the only important factor. Following are some of the basic ways developed nations and international organizations can provide the greatest assistance.
Developed Nations
-
Developed nations should be aware of the issues involved in conserving biological diversity and their magnitude at the local, national, regional, and international levels. There is an extreme need to understand more globally the problems faced by Third-World countries. Money alone will not lead to the successful conservation of biological diversity in those regions.
-
They must also be aware of our partnership and shared responsibilities as well as the need for mutual respect. For example, the conservation of migratory species of wildlife requires collaboration, understanding, and mutual respect.
-
They must be cognizant of the need to spread the conservation message and educate citizens of developed nations on environmental issues. The international
-
trade of wildlife demonstrates the urgent need to educate people where the demand is greatest.
-
They must be sensitive to the need for a more balanced exchange in all interactions with the Third World, be it in policy, economy, or scientific expertise. Examples of such exchanges can be found in multilateral and transnational organizations.
International Organizations
International organizations can help in the following ways:
-
They can facilitate the flow of information at all levels, as is done now by the Food and Agriculture Organization’s Latin-American network of protected areas, other reserves, and wildlife.
-
They can cooperate among themselves when working within certain countries. For example, close collaboration between the World Wildlife Fund and the Conservation Foundation with the Nature Conservancy International Program in Latin America would benefit them as well as the region.
-
They can support leadership development in all fields related to the environment, considering local needs within a particular country or geographical region. Local needs should be specifically addressed with local vision. Train (1986, p. 1) wrote, “We often are so busy developing our own solutions to other peoples’ problems as we see them that we sometimes become a bit deaf and a bit insensitive to the ideas of those very people whose problems we are trying to solve.”
-
They can cooperate in the definition of plans to coordinate conservation and development programs. The large international banks such as the World Bank or the Inter-American Development Bank would benefit a great deal from a closer association with local, national, and international conservation organizations.
-
They can cooperate in the identification of alternative ways natural resources can be used that contribute to economic growth without compromising their survival. Projects such as the Green Iguana Project of the Smithsonian Tropical Research Center in Panama (see Chapter 42) and INIREB’s captive-breeding programs for crocodiles and white-collared peccaries in Chiapas, Mexico, should be encouraged.
-
They can collaborate in the application of ecological concepts, such as sustainability and carrying capacity, to development programs.
-
They can help to build local capabilities with well-defined strategies. It is cheaper and ethically more correct in the long run. Good examples are the Conservation Data Centers of the Nature Conservancy International Program in Latin America and the support given to Restauracion Ambiental, Inc., in Mexico by the Conservation Foundation and the World Wildlife Fund.
-
They can support the establishment of functional conservation units in every country within the region. Many protected areas in those countries are protected only on paper. Biological diversity will not survive under these conditions.
-
They can promote research on basic and applied sciences related to environmental issues in the Third World. Support should be given preferentially to interdisciplinary groups that address topics on conservation and development. These
-
groups should include qualified nationals from the country in which the work is performed.
-
They can help to increase the prestige of particular research areas such as taxonomy, systematics, biogeography, and paleontology. This could be done by awarding prizes, fellowships, and scholarships and establishing exchange programs between research centers and universities.
We face enormous tasks that in the long run can be performed only by reaching a basic understanding of our shared responsibilities. Time is running out. The challenges to biodiversity can only be met with mutual understanding, help, and respect.
ACKNOWLEDGMENTS
I would like to acknowledge the assistance of Curtis Freese, Robert Healy, Martha Hays-Cooper, Oscar Flores, Patricia Gerez, and Alexander and Barbara Stevenson for reviewing early drafts of the manuscript.
REFERENCES
Albert, L.A. 1985. Legislacion y Plaguicidas. Jan. 4, 1985. Subcomite de Plaguicidas, Comite de Ecologia, Consejo General de Salubridad. Mexico, D.F.
Dornbusch, R., and S.Fischer. 1986. Third World debt. Science 234:836–841.
Harrison, J., K.Miller, and J.McNeeley. 1984. The world coverage of protected areas: Development goals and environmental needs. Pp. 24–33 in J.McNeeley and K.Miller, eds. National Parks: Conservation and Development. Proceedings of the 2nd World Conference on National Parks. Smithsonian Institution Press, Washington, D.C.
IUCN (International Union for Conservation of Nature and Natural Resources). 1980. World Conservation Strategy: Living Resource Conservation for Sustainable Development. International Union for Conservation of Nature and Natural Resources, Gland, Switzerland.
Lustig, N. 1986. El precio social del ajuste Mexicano: Balance de sombras. Nexos 106(Oct.):27–31.
Ramos, M.A. 1985. Problems hindering the conservation of tropical forest birds in Mexico and Central America and steps toward a conservation strategy. Pp. 305–318 in Technical Publication No. 4. International Council on Bird Preservation, Cambridge, England.
Story, C., ed. 1986. News and commentaries on Latin America. The Latin American Times (London) 73:29–31.
Tello, C. 1986. La deuda externa. Nexos 106(Oct.):19–26.
Train, R. 1986. Closing remarks, Partners in Conservation. Twenty-fifth Anniversary Conference of the World Wildlife Fund, September 17, 1986, Mayflower Hotel, Washington, D.C.
UNEP (United Nations Environment Programme). 1984. Register of International Treaties and Other Agreements in the Field of the Environment. UNEP/GC/Information/11. United Nations Environment Programme, Nairobi, Kenya. 205 pp.
CHAPTER 49
A MAJOR NEW OPPORTUNITY TO FINANCE THE PRESERVATION OF BIODIVERSITY
ROBERT J.A.GOODLAND
Ecologist, Office of Environmental Affairs, The World Bank, Washington, D.C.
In June 1985, the World Bank1 promulgated a major new policy (World Bank, 1986) regarding wildlands, which are defined as natural land and water areas in a state virtually unmodified by human activity. This policy focuses mainly on the preservation of wild plants and animals and their habitats. It is very significant to the financing of the preservation of biological diversity.2 There is no need to justify this policy to those committed to biological diversity, although such justification has been provided by the World Bank (1986, 1987), by Ehrlich and Ehrlich (1981), by Goodland (1985), and by Norton (1986).
The World Bank’s general policy is to avoid eliminating wildlands—following the first injunction of Hippocrates 2,000 years ago: “Non noli nocere” (First do no harm). Rather, the Bank will assist in the preservation of wildlands. This translates into six specific policy elements that are addressed at various stages of the Bank’s project cycle (see Appendix A at the end of this chapter).
The first policy element states that the Bank normally declines to finance projects involving the conversion of wildlands of special concern, which are areas recognized as exceptionally important in conserving biological diversity or perpetuating en-
|
1 |
Includes the International Bank for Reconstruction and Development (IBRD), the International Development Association (IDA), and the International Finance Corporation (IFC). |
|
2 |
Preservation of genetic diversity, particularly varieties of economic plants and animals, is acknowledged to be important but is not discussed separately from biotic diversity in this chapter. The necessary complement, reducing poverty, population growth, and other pressures forcing people to destroy wildlands, is being addressed by, for example, the World Bank and World Resources Institute. See Chapter 44 by Spears and Chapter 45 by Burley. |
vironmental services. They can be classified into two types. First are wildlands officially designated as protected areas by governments, sometimes in collaboration with the United Nations or the international scientific community. These are national parks, biosphere reserves, world heritage sites, wetlands of international importance, areas designated for protected status in national conservation strategies or master plans, and similar wildland management areas (WMAs), i.e., areas where wildlands are protected and managed to retain a relatively unmodified state. Second are wildlands not yet protected by legislation but recognized by the national or international scientific and conservation communities, often in collaboration with the United Nations, as exceptionally endangered ecosystems, known sites of rare or endangered species, or important wildlife breeding, feeding, or staging areas. These include certain types of wildlands that are threatened throughout much of the world, yet are biologically unique, ecologically fragile, or especially important for local people or for environmental services. Tribal people almost always manage their areas sustainably and in a manner entirely compatible with environmental conservation; this important link is discussed by the World Bank (1982).
Wildlands of special concern often occur in tropical forests, Mediterranean-type brushlands, mangrove swamps, coastal marshes, estuaries, sea grass beds, coral reefs, small oceanic islands, and certain tropical freshwater lakes and riverine areas. Within the spectrum of tropical forests, lowland moist or wet forests are the richest in species and are often the most vulnerable. Wildlands of special concern also occur in certain geographical regions where they have been reduced to comparatively small patches and continue to undergo rapid attrition. As a result, these regions harbor some of the most threatened species in the world. Following is a list of some tropical wildlands of special concern:
EAST AFRICA
Madagascar: significant proportions of the northern and eastern moist forests.
Ethiopia: much of the remaining highland forest.
Tanzania: Usambara, Pare, and Uluguru Mountains.
Rwanda: mountain forests along the Zaire and Uganda borders.
Kenya: Kakamega, Nandi, and Arabuko-Sokoke forests.
WEST AFRICA
Cameroon: particularly Cameroon Mountain and the moist forested area extending into Gabon and to the Cross River in southeastern Nigeria, including the Oban Hills.
Ivory Coast: southwestern forests (including the Tai forest) and adjacent parts of Liberia and Sierra Leone.
EAST ASIA AND PACIFIC
The Malay Peninsula (including parts of Thailand): lowland forests, especially along the northwestern and eastern coasts.
Indonesia: much of the remaining lowland forests of Kalimantan, Sumatra, Sulawesi (especially the two southern peninsulas), and many smaller islands (e.g., Siberut).
Philippines: much lowland forest on all larger islands.
SOUTH ASIA
Sri Lanka: the coastal hills of the southwest and the Sinharaja forest of the wet zone.
India: most of the forests remaining on the Western Ghats.
Burma: the untouched teak in the northern regions.
LATIN AMERICA AND CARIBBEAN
Ecuador: lowland coastal forests.
Mexico: Lacandon forest in Chiapas.
Honduras-Nicaragua border: Mosquitia forest.
Panama: Darien province.
Colombia: the Choco region adjacent to Darien province.
Brazil: coastal forests of the Cocoa Region in the southeastern extension of Bahia, between the coast and an area located 41° 30′ W and somewhere between 13° 00′ and 18° 15′ S, and an outlier near Linhares, Espiritu Santo.
Brazil: parts of the eastern and southern Amazon region.
TROPICAL AQUATIC AREAS
Brazil, Peru, Colombia, Ecuador, and Bolivia: Amazon River and associated wetlands (including varzea forests); Mato Grosso, Pantanal Swamp.
Venezuela and Colombia: Orinoco River and Delta.
Papua New Guinea: Purari River.
Sumatra, Indonesia: Musi River and Lake Toba.
Malawi: Lake Malawi and other Rift Valley lakes.
Sudan: Sudd Swamp.
Guatemala: Lake Atitlan.
Before a project is begun, a brief wildland survey is needed in order to determine whether the proposed project area fits one of these categories (see sample form on p. 445). This means that more tropical biologists in applied ecology and more biodiversity data will be needed. The World Bank normally declines to finance projects within wildlands of special concern even if they were partly converted prior to the Bank being invited to consider financing. The phrase “normally declines” may appear to be a glaring deficiency, but it provides for mutually useful trade-offs in certain cases. For example, for a request that a typical 1% of a desert national park be inundated by a reservoir, the quid pro quo or trade-off would normally be financing for the whole park plus the addition of an equivalent 1% area. This should be equivalent both biologically and in size. A needed, under-represented habitat could be the compensatory tract if that is the national priority. Furthermore, the desert national park would get a permanent aquatic habitat and a source of perennial water. Financing the elevation of unprotected or unmanaged conservation units into well-managed parks can often be part of a mutually acceptable trade-off.
The second policy element pertains to wildlands other than those of special concern. The Bank prefers projects on lands already converted (e.g., logged over, abandoned, degraded, or already cultivated) some time in the past rather than in anticipation of a Bank project. This means that biotic surveys are necessary in
order to select logged-over sites rather than intact sites. Rehabilitation of degraded sites therefore must take preference over conversion of wildlands.
The third element concerns deviations from these policies. All deviations must be explicitly justified. This need not necessarily include public hearings or town meetings, which are required in applications of the U.S. National Environmental Policy Act (CEQ, 1978). However, explicit justification implies a more open decision-making process than has occurred in the past. The options, particularly site selection, should be discussed outside the main ministry responsible for implementation and with the ministry of the environment (or equivalent), academia, and environmental groups.
The fourth element applies where development of wildlands is justified. In this case, the conversion of less-valuable wildlands is preferable to the conversion of more-valuable ones. This also means that detailed biotic inventory and resource surveys are essential for two reasons: to avoid more-valuable wildlands such as centers of endemism and to locate less-valuable wildlands. At the very least, such essential surveys will strengthen the environmental data base on wildlife and the nation’s biological diversity. The need for experienced tropical biologists in applied ecology will therefore increase.
The fifth element applies when significant conversion of wildlands is explicitly justified, as described in the third element. Somewhat arbitrarily, “significant” conversion is defined as the conversion of 100 square kilometers (which is smaller than many development projects). “Significant” conversion also includes tracts smaller than 100 square kilometers when they are a large proportion of the remaining wildland area of a specific ecosystem. The loss of such wildlands must be compensated by including a wildlands management component in the project concerned—usually direct financing for the preservation of an area similar to that converted for the project, both ecologically and in size (it must not be smaller than the area converted by the project). Thus, biotic surveys are essential to define the area slated for conversion and to find an ecologically similar area. Occasionally, the compensatory wildland may be an underprotected habitat rather than another sample of an already adequately protected habitat. These policies pertain to any project in which the Bank is involved, irrespective of whether the Bank is financing the project component that affects wildlands.
The sixth element concerns projects in which conversion of wildlands is not contemplated. The policy is to preserve wildlands for the value of their environmental service alone. The success of projects that do not eliminate any wildlands often depends on the environmental services that those wildlands provide. In such cases, the Bank’s policy is to include a project component to conserve the relevant land in a wildland management area (WMA) rather than to leave its preservation to chance. The best example of an environmental services component is in Indonesia, where the water supply for a 197-square kilometer irrigated rice-growing scheme in Sulawesi is protected by designating an entire watershed outside the rice fields as a national park. This 3,200-square-kilometer area, now called Dumoga Bone National Park, received $1.1 million (1.5% of project costs) from the Bank-assisted loan. Since then, the park has become the site of the world’s biggest-ever scientific expedition, supported by the government of Indonesia and the Royal
Entomological Society (UK). This expedition, called Operation Wallace, lasted 3 years and involved over 150 scientists of many disciplines, mainly taxonomists and ecologists.
Four types of economic development projects need compensatory wildland management components, as required under the fifth policy element:
-
Agriculture and livestock projects involving wildland clearing, wetland elimination, wildland inundation for irrigation storage reservoirs, watershed protection for irrigation, displacement of wildlife by fences or domestic livestock; fishery projects involving elimination of important fish nursery, breeding, or feeding sites; over-fishing or introduction of ecologically risky exotic species within aquatic wildlands; forestry projects involving access roads, clear-cutting or other intensive logging of wildlands, wildland elimination. Plantations of fast-growing tree species are often an important complement to more direct wildland management activities by reducing the economic pressures for cutting the remaining forest wildland. They should be sited preferentially on already deforested land.
-
Transportation projects involving construction of highways, rural roads, railways, or canals that penetrate wildlands or that facilitate access and spontaneous settlement in or near wildlands; channelization of rivers for fluvial navigation; dredging and filling of coastal wetlands for port projects.
-
Hydroelectric projects involving large-scale water development, including reservoirs and power and water diversion schemes; inundation or other major transformation of aquatic or terrestrial wildlands; watershed protection for enhanced power output; construction of power transmission corridors. Wetlands (such as ponds, marshes, swamps, flood plain forests, estuaries, and mangroves) can be eliminated inadvertently through water diversions upstream or deliberately through drainage, diking, or filling.
-
Industry projects involving chemical and thermal pollution, which may damage wildlands; wildland loss from large-scale mining; wildland conversion for industrial fuels or feedstocks.
Essentially, any project affecting wildlands, even indirectly, is a candidate (Ledec and Goodland, in press). The policy also states that projects with wildland management as the sole objective should be encouraged. This does not mean the Bank has changed from being an economic development lender to an environmental granting agency. The preservation of wildlands can often be justified with economic criteria alone, such as the income generated by tourism or scientific and educational activities. Moreover, it means that conversion of wildlands will be increasingly unlikely in Bank-financed projects. More importantly, it means that where some conversion will occur, then the trade-off translates into major financing for the preservation of biological diversity.
TYPES OF WILDLAND MANAGEMENT COMPONENTS
The most effective component of wildland management is financial support for the conservation of ecologically similar wildlands in one or more WMA. Where a WMA has already been established in the same type of ecosystem that is to be converted by a Bank-supported project, then it may be preferable, for administrative
or biological conservation reasons, to enlarge the existing WMA rather than to establish a new one. Biological conservation is usually more effective in one large WMA than in several small ones that combined have the same total size and the same types of natural habitats. The government’s wildland agencies, local university wildlife departments, and various international organizations can often assist in making such judgments.
A wildland management component could also involve the creation of a new wildland habitat rather than the preservation of existing habitat. For example, marginal land on the fringes of irrigation projects could be converted to wildlife reserves by taking advantage of the water supply created by the projects. Natural depressions or seasonal swamps could be exploited by diverting water from the canal systems (probably a very small part of the total supply). Such reserves could attract significant numbers of migratory and resident waterfowl with minimal additional project costs and land. The Wildfowl Trust in Slimbridge, England, has set up such reserves, ranging in size from 5 to 8 square kilometers.
A useful option is to improve the quality of the management of existing WMAs. Many WMAs in Bank-member countries receive insufficient on-the-ground management due to a lack of adequately paid staff; deficiencies in training, staff housing, equipment, spare parts, and fuel; or the absence of a well-developed management plan through which efficient resource-allocation decisions can be made. Small components can often help correct these deficiencies. In countries where effective management is clearly lacking, it is generally preferable to improve the management of existing WMAs than to create new units on paper, thereby further overextending the limited capabilities of the responsible agencies. Whenever a new WMA is established as a project component, provisions are needed to ensure effective management. Since many wildland agencies (e.g., departments of national parks or wildlife) are not as operationally effective as necessary, institutional strengthening (particularly support for training) should be an important element of Bank-supported wildland management components.
The establishment or strengthening of WMAs is particularly effective when the government includes these wildland areas in a national conservation or land-use plan. A growing number of Bank-member governments have undertaken some type of systematic land use planning for wildland management. Such planning can take various forms, ranging from master plans for a system of national parks and other WMAs to National Conservation Strategies, which address wildland management as only one component of a broad range of natural-resource planning concerns, and in which policy interventions such as economic incentives are used to influence resource utilization. Bank assistance with such planning efforts greatly strengthens wildland management at the national level. When member governments agree to develop appropriate land-use plans, the Bank refrains from supporting projects that involve the elimination of wildlands and run counter to these plans.
SIGNIFICANCE FOR BIOLOGICAL DIVERSITY
The significance of these new policies for biological diversity is that the World Bank will finance the preservation of wildlands. The World Bank is owned by 152 developed and developing member nations, so it is likely that the species or habitats with which most readers are concerned occur in a country that is a member of the Bank. This demonstrates a clear need for nongovernment organizations (NGOs). Communications between governmental agencies, NGOs, and academia are not yet efficient and systematic in the environmental arena. The Bank seeks to foster such communications.
The Bank lends money mainly for specific economic development projects—more than US$16 billion in fiscal 1986. This means that the country must be willing to borrow at near-prevailing global interest rates. If World Bank lending conditions become too onerous, a borrower may seek less-conditional financing elsewhere (and there are indications that this has already started to occur). Also, if the project does not have an impact on a wildland or is not influenced by a wildland, financing for wildlands becomes more difficult. If a project needs only 1% of a wildland, for example, a mutually beneficial trade-off can be negotiated. If you hear of a project needing such wildland financing, you may want to contact the government concerned or the Bank’s project officer. Improved cooperation between government, academia, NGOs, and financing agencies is a key element in preserving biological diversity. Although this is not easy, the alternative is accelerating extinctions.
REFERENCES
CEQ (Council on Environmental Quality). 1978. Regulations (for Implementing the Procedural Provision of the) National Environmental Policy Act. U.S. Government Printing Office, Washington, D.C. 44 pp.
Ehrlich, P.R., and A.Ehrlich. 1981. Extinction: The Causes and Consequences of the Disappearance of Species. Random House, New York. 305 pp.
Goodland, R. 1985. Wildlands Management in Economic Development. First International Wildlife Symposium. IX World Forestry Congress and the Wildlife Society of Mexico, Maria Isabel Sheraton Hotel, Mexico, 14 May 1985. The World Bank, Office of Environmental and Scientific Affairs, Washington, D.C. 34 pp.
Ledec, G., and R.Goodland. In press. An environmental perspective on tropical land settlement. In D.A.Schumann and W.L.Partridge, eds. Human Ecology of Tropical Land Settlement in Latin America. Westview Press, Boulder, Colo.
Norton, B.G., ed. 1986. The Preservation of Species. Princeton University Press, Princeton, N.J. 305 pp.
World Bank. 1982. Tribal Peoples and Economic Development, Human Ecologic Considerations. The World Bank, Washington, D.C. 111 pp.
World Bank. 1984. Environmental Policies and Procedures of the World Bank. The World Bank, Office of Environmental and Scientific Affairs, Washington, D.C. 8 pp.
World Bank. 1986. The World Bank’s Operational Policy on Wildlands: Their Protection and Management in Economic Development. The World Bank, Washington, D.C. 21 pp.
World Bank. 1987. Wildlands: Their Protection and Management in Economic Development. The World Bank, Washington, D.C. 300 +pp.
APPENDIX A: THE WORLD BANK’S PROJECT CYCLE
Responsibility for implementing wildland management projects or components within the World Bank rests primarily with the Bank’s regional operations staff, which receives advice and operational support from the Office of Environmental and Scientific Affairs, Projects Policy Division (PPDES). The key contact person is the project officer.
Projects under consideration are reviewed by the Bank’s regional staff in conjunction with PPDES to identify, as early as possible, the need to avoid converting a wildland tract or to preserve such a tract as part of the project. To determine whether a proposed project will have an impact on or be in close proximity to environmentally important wildlands, Bank staff consult those government agencies with jurisdiction over wildland management authority. PPDES maintains contacts with such agencies and will assist upon request. Additional sources of information on ecologically important wildlands include computerized data bases maintained by some nongovernment organizations (NGOs) and several published directories, which are available from PPDES. In this manner, it will often be possible to learn quickly whether a proposed project site contains existing or proposed WMAs; known endangered species; major wildlife or fish breeding, feeding, or staging-areas; important watershed catchments; or living resources of major importance to local people. If none of these mechanisms reveal the existence of ecologically important wildlands in the project area, a brief preproject field survey is necessary since many important wildlands have not yet been identified. This field survey should be undertaken by relevant specialists from the government’s environmental ministry, wildlife agency, national university, or similar institution. This brief survey indicates the nature and extent of impacts on critical wildlands that would result from the implementation of the project and puts the information in a national context. The results should be recorded on the sample form provided below.3
During project preparation (or feasibility study stage), the Bank’s project staff (or their consultants) may assist the borrower (e.g., the Ministry of Highways) or the project sponsor in carrying out the necessary environmental studies, including those pertaining to wildlands. PPDES can recommend consultants or other experts who can identify important wildland areas, carry out necessary field surveys, or help design appropriate wildland management project components. At the completion of any necessary studies, information on whether the project involves the conversion or disintegration of a relatively unmodified ecosystem and alternative suggestions for achieving the goals of the government should be added to the project brief. If conversion is justified, the brief should explain why and should identify the wildland management components needed.
As part of the appraisal process, the Bank’s project staff assesses the planned wildland management and other environmental measures. The Staff Appraisal Report specifically describes any planned wildland management measures, including budgets and agency responsibilities. Once wildland measures are identified as necessary, timely action should be ensured by loan conditionality such as loan effectiveness or disbursement. Since wildlands must be managed in perpetuity to be effective, the loan agreement should specify long-term measures that the borrowing nation has agreed to implement.
Supervision should routinely review implementation of the wildland component with the borrower. As a general principle, the wildland component of the project should be well under way before major land clearing or construction are allowed to proceed.
|
WILDLAND SURVEY AND MANAGEMENT FORM (Sample Only) Name of project: _________________________________________ Expected appraisal (or other) date: ____________________________ Date of this survey: _______ Surveyor: ________ Affiliation: _______ Methodology(ies) (circle one): site inspection/library research/both/other (specify)
Maps and more complete reports used or available should be appended or cited. |
CHAPTER 50
AND TODAY WE’RE GOING TO TALK ABOUT BIODIVERSITY…THAT’S RIGHT, BIODIVERSITY
LESTER R.BROWN
President, Worldwatch Institute, Washington, D.C.
In Chapter 1, E.O.Wilson says that we are locked into a race concerning biodiversity. He implies that we are in a race against time and that the “we” is humanity. Unfortunately, the only people who are actively engaged in the race to preserve our rich evolutionary inheritance of plant and animal life are a handful of concerned scientists and environmentalists. To make the sort of headway that the situation calls for will require a lot more people expressing concern and working on the issue.
The problem is difficult in that the loss of biodiversity is largely the indirect result of other activities, such as producing food and using energy. It is similar to carbon dioxide-induced climate change, the depletion of the ozone layer, and the acidification of lakes and forests. Often there are no obvious bad guys. And the issue is not new: The Sinking Ark was published in 1979 (Myers, 1979); Extinction came out in 1981 (Ehrlich and Ehrlich, 1981). Looking at the problem and comparing it with other issues that Worldwatch has looked at over the years, I can identify three things that might help in generating response on the scale needed: better information, internationalization of the issue, and heightened public awareness.
It has been impressive and confusing to see the number of estimates on the rate of future extinction—just looking to the end of the century. The estimates, from reputable scientists who are concerned about the issue, vary not only widely, but wildly. This uncertainty provides an excuse for inaction by those who prefer not to be doing anything. It might be useful to bring scientists together to try to narrow the range of estimates, to clarify the assumptions, and to provide some numbers that can be used. In addition to the rate of species loss, we need a better understanding than what we now have of the economic implications.
The question concerning internationalization of the issue is how. One way would be to look to the United Nations (UN) and its special conferences, a procedure that has been followed for the last 15 years with other emerging global problems. The 1972 UN conference on the environment in Stockholm gave the environmental issue international legitimacy. Then came the UN conference on population in Bucharest and one on food in Rome, both in 1974. Following this came conferences on habitat in Vancouver, on water in Rio de Plata, on deserts in Nairobi, and on technology in Vienna.
Maybe the UN should have a conference on biodiversity. Not because the conferences themselves achieve great things in their own right but because they do serve important educational functions. Their first function is education of the bureaucracy. This occurs when governments have to analyze their own national situation and prepare their position papers for the conference. Their second important educational function is that they provide an excuse for Time magazine to carry a story on biodiversity. And that’s important.
Another option related to the UN is a possible special session at the UN General Assembly. Special sessions in the last few years have addressed disarmament and Africa. Perhaps biodiversity should also be an integral part of the new, major, international initiative that the International Council for Scientific Unions (ICSU) will be orchestrating on global habitability.
These activities can raise public awareness. There is a long-standing principle in Washington, D.C., that you can convince everyone within the beltway to move in a particular direction on a particular issue, but if no one outside the beltway knows the issue exists, nothing will happen. To illustrate, the United States over the last half century has had two soil erosion crises. The first was the Dust Bowl in the thirties—a highly visible crisis, which is now an era in U.S. history. The clincher in getting a response came when Congress left a session late one afternoon in 1934 and discovered their cars covered with dust. Someone pointed out that the soil came from Oklahoma and Kansas. Only then did the urban Congressmen from the East, who previously had been unmoved by the issue, take action by creating the Soil Conservation Service (SCS), which launched an effective response to that threat.
The more recent erosion problem unfolded in the late 1970s. This resulted from the doubling of grain prices in 1972 after the Soviet wheat purchase and the fencerow-to-fencerow planting, which increased the amount of cultivated land by about 10%. Much of that land should not have been brought under the plow. Coupled with this was a second issue: the need for information. Congress passed the legislation requiring a natural resources inventory. The SCS conducted two inventories of soil erosion, the first in 1977 and a second in 1982. With the two points, we could begin to get a fix on trends. Over a million readings were involved in the 1982 inventory, literally almost down to the individual farm. The inventory told us precisely where the soil erosion was occurring, under what conditions, and how much soil was being lost. We also knew from a number of research projects what that loss translated into economically. On both corn land and wheat land, a loss of one inch of topsoil reduces yields by 6%. For corn, the loss was about 6
bushels per acre; for wheat, about 2 bushels per acre. Thus erosion and economics were indisputably linked.
Many other studies followed. The American Farmland Trust (1984) did an excellent study using the data from the National Resources Inventory. The National Audubon Society made soil erosion one of their projects. At the Worldwatch Institute, our very first book over a decade ago was entitled Losing Ground (Eckholm, 1976). We’ve since done two Worldwatch Papers, and each State of the World (Brown et al., 1984, 1985, 1986) report has had either a chapter or a section of a chapter dealing with the issue. We have in our files something close to a thousand articles generated or bylined by staff members in periodicals, newspapers, magazines, and journals around the world. It has been an enormous educational process involving many groups, of which we are one.
I remember being invited to appear on the Today Show to talk about this issue. The lead that Jane Pauley used beginning at 7:00 a.m. was, “And today we’re going to have an author who’s written a book about soil erosion. THAT’S RIGHT, soil erosion.” They repeated this in incredulous tones as though it was not an issue that would be found on the Today Show. For weeks after that show, my two children entertained themselves around the house saying, “And our dad’s written a book on soil erosion. THAT’S RIGHT, soil erosion.”
Once public concern was aroused and there was a public awareness of the issue, it became possible for politicians to act. In addition to the traditional organizations (e.g., the Soil Conservation Service of America), environmental groups, such as Audubon and Sierra—large membership groups with skilled lobbyists who write position papers and work with members of Congress—became active. Suddenly it became politically the thing to do, and both Houses voted overwhelmingly for a new farm bill that called for taking 45 million acres (18 million hectares) of our most highly erodible cropland out of production over the next 5 years and putting it into either grass or trees under 10-year contracts. The second thing they did would have been anathema only a few years ago. The farmers who were cropping highly erosive land or managing it badly were told they would not be eligible for farm support programs! That was new.
We are in a race. Maybe we should call it a contest. A lot of the things that are happening, like the threats to biodiversity, are the products of rather innocent actions. Population growth is the result of reproductive patterns that were an integral part of our survival as a species. It has now become a real threat. In July our number reached 5 billion. There was no dancing in the streets: rather, there was a profound sense of unease, not only because we had reached 5 billion but also because there is so much momentum inherent in population growth now. With the young age structure, there will be 3 billion people entering the reproductive age group over the next generation—an enormous push. Nonetheless, the U.S. government recently announced that it was withdrawing all support for the UN Fund for Population Activities—the major agency responsible for working with over 199 countries to try to get the brakes on population growth.
One of the lessons I draw from this sort of exercise is that scientists are going to have to become activists. The recently formed Club of Earth has pointed out
that some of the leading scientific minds working on biodiversity are deeply concerned about the issue and believe that we need to take action on a number of fronts. Maybe the Club of Earth,1 the purpose of which is to bring scientific attention more quickly to important but neglected environmental problems, should write the White House and explain why it is important to restore full support for the UN Fund for Population Activities in order to protect biodiversity.
We’ve got to move the issue from the scientific journals into the magazines and the popular press, so that maybe someday Jane Pauley will say, “And today we have a scientist who’s going to discuss biodiversity. THAT’S RIGHT, biodiversity.”
REFERENCES
American Farmland Trust. 1984. Soil Conservation in America: What Do We Have to Lose? American Farmland Trust, Washington, D.C. 133 pp.
Brown, L.R., W.U.Chandler, C.Flavin, J.Jacobson, C.Pollock, S.Postel, L.Starke, E.C. Wolf. 1984. State of the World 1984. W.W. Norton, New York. 250 pp.
Brown, L.R., W.U.Chandler, C.Flavin, J.Jacobson, C.Pollock, S.Postel, L.Starke, E.C. Wolf. 1985. State of the World 1985. W.W. Norton, New York. 301 pp.
Brown, L.R., W.U.Chandler, C.Flavin, J.Jacobson, C.Pollock, S.Postel, L.Starke, E.C. Wolf. 1986. State of the World 1986. W.W. Norton, New York. 263 pp.
Eckholm, E.P. 1976. Losing Ground. Environmental Stress and World Food Prospects. W.W. Norton, New York. 223 pp.
Ehrlich, P.R., and A.Ehrlich. 1981. Extinction. The Causes and Consequences of the Disappearance of Species. Random House, New York. 305 pp.
Myers, N. 1979. The Sinking Ark. Pergamon, Oxford. 307 pp.
CHAPTER 51
THE EFFECT OF GLOBAL CLIMATIC CHANGE ON NATURAL COMMUNITIES
ROBERT L.PETERS II
Research Associate, World Wildlife Fund’Conservation Foundation, Washington, D.C.
Current human population and development pressures are breaking wild biological communities into fragments surrounded by human-dominated urban or agricultural lands. The result is that many wild species, perhaps hundreds of thousands by the end of this century, will be lost because of habitat disturbance (Lovejoy, 1980; Myers, 1979). Recent advances in conservation biology have demonstrated that even some species we thought would be protected within reserves may still be lost because the reserves are too small to maintain viable populations of all the species within them (Frankel and Soulé, 1981; Schonewald-Cox et al., 1983; Soulé, 1986; Soulé and Wilcox, 1980). To this daunting picture must be added a newly recognized threat, one with potentially disastrous consequences for biological diversity. This threat is global warming, commonly called the greenhouse effect.
It now seems very likely that ecologically significant climate change will occur within the next century and that many natural populations of wild organisms will be unable to exist within their present ranges. They will be lost, unless they are able to colonize new habitat where the climate is suitable, either on their own or with human help. Simply because many species survived past natural climate changes does not mean that they will survive this one without aid. The coming change promises to be very big and very fast, and because human activities will increasingly fragment and isolate populations, it will be more difficult for many species to successfully colonize new habitat when the old one becomes unsuitable.
FUTURE CLIMATE
What do we know about the climatic future? In the last several years a virtual consensus has been reached among atmospheric scientists that the planet will warm significantly during the next hundred years as the result of the production of carbon dioxide and other so-called greenhouse gases by humans (NRC, 1983; Schneider and Londer, 1984). Because molecules of these gases absorb infrared radiation, preventing it from radiating into space, increases in their concentration will cause increases in average global temperature. Exactly how large the warming will be and how fast it will come are still uncertain, but best estimates are of a magnitude sufficient to have profound effects on natural biological systems. In 1983 the National Research Council concluded that 3±1.5°C of warming by the end of the next century was most likely (NRC, 1983), based on effects due to carbon dioxide concentration alone (see Figure 51–1A for one model’s predictions). More recent analyses of the contributions of other greenhouse gases, including methane and the chlorofluorocarbons, suggest that the total greenhouse effect may be double that of carbon dioxide alone—cutting in half the amount of time necessary to reach a particular level of warming (Machta, 1983; Ramanathan et al., 1985). In short, warming of several degrees is likely within the next 100 years, perhaps the next 50 years. While a warming of this amount may seem small, it is not. Even a 2°C change is very large compared to normal fluctuations and would leave us with a planet warmer than at any time in the past 100,000 years (Schneider and Londer, 1984).
In thinking about the effects of climate change on natural communities, it is important to realize that the effects do not suddenly begin at some arbitrary threshold, such as the commonly used benchmark of doubled carbon dioxide concentration. Rather, ecological responses will begin with small amounts of warming and will increase as the warming does. Thus, a species like the dwarf birch (Betula nana), which exists in Britain only at sites where the temperature never exceeds 22°C (Ford, 1982), might begin retracting the southernmost portion of its range as soon as the local temperature climbs over 22°C.
In the long term, temperatures may rise above the several degrees predicted for a doubled carbon dioxide scenario, for there is no reason that concentrations of carbon dioxide and other greenhouse gases, and hence warming, should stabilize when the benchmark of doubled carbon dioxide is reached. If people continue to put more gases into the atmosphere, temperatures will continue to climb.
At least as important as temperature rise itself in affecting the distributions of species and the stability of biological communities will be the widespread changes in precipitation it causes (Hansen et al., 1981; Manabe et al., 1981; Wigley et al., 1980). Thus, the southern limit of the European beech tree (Fagus sylvatica) is determined by the point at which rainfall is less than 600 millimeters annually (Seddon, 1971), and a change in rainfall would be expected to cause a change in range. Although models of future rainfall distribution based on projected temperature increases are still rough, their implications are cause for concern. One model predicts that global warming will cause rainfall decreases of up to 40% for the American Great Plains by the year 2040 (Figure 51–1B; Kellogg and Schware,
FIGURE 51–1 (a) Global patterns of surface temperature increase, as projected by the Goddard Institute for Space Studies (GISS) model (Hansen et al., in press). Numbers are in degrees Celsius, (b) Global changes in moisture patterns. After Kellogg and Schware (1981).
1981). Other factors associated with rising temperatures that have biological implications include the direct physiological effects of rising atmospheric carbon dioxide concentration itself on plants (in Lemon, 1983) and a moderate sea level rise, variously estimated to be between 144 and 217 centimeters by 2100, according to the U.S. Environmental Protection Agency (EPA) (Hoffman et al., 1983). Plants will vary according to the way carbon dioxide concentrations affect their photosynthetic efficiencies and water requirements, thus altering interspecific relationships. In addition, changes in both precipitation and elevated carbon dioxide levels would alter soil chemistry (Emanuel et al., 1985; Kellison and Weir, in press).
SPECIES RANGES SHIFT IN RESPONSE TO CLIMATE CHANGE
By using the fossil record to study past responses of communities to similar climate changes, we can get some idea of how species ranges might respond to the physiological and competitive stresses imposed by future change. The most important observation is that, not surprisingly, species tend to track their climatic optima, retracting their ranges where conditions become unsuitable while expanding them where conditions improve (Ford, 1982; Peters and Darling, 1985). A general observation is that during past warming trends, species have shifted both toward higher latitudes and higher elevations (Baker, 1983; Bernabo and Webb, 1977; Flohn, 1979; Van Devender and Spaulding, 1979). During several Pleistocene interglacial periods when the temperature in North America was only 2° to 3°C higher than at present, osage oranges (Maclura sp.) and pawpaws (Asimina sp.) grew near Toronto, several hundred miles north of their present distribution; manatees (Trichechus sp.) swam off the New Jersey shore; tapirs (Tapirus sp.) and peccaries (Tayassu sp.) foraged in Pennsylvania; and Cape Cod had a forest like that of present-day North Carolina (Dorf, 1976). As to altitudinal shifting, during the middle Holocene when temperatures in eastern North America were 2°C warmer than at present, hemlock (Tsuga canadensis) and white pine (Pinus strobus), for example, were found 350 meters higher on mountains than they are today (Davis, 1983). In general, a short climb in altitude corresponds to a major shift in latitude, so that 3°C of cooling may be found by traveling either 500 meters up a mountain or 250 kilometers toward a pole (MacArthur, 1972).
Evidence of such range shifts during periods of warming in the past, together with projections of range shifts based on physiological tolerances and computer-modeled future climatic conditions, suggest that in the United States, the oncoming warming trend may shift the area within which a particular species may flourish by as much as several hundred kilometers to the north. A projection for loblolly pine (Pinus taeda), for example, suggests that the southern limit of this species in the United States may shift more than 300 kilometers to the north by the year 2080 because of moisture stress (Miller et al., in press). Another simulation indicates that the doubling of atmospheric carbon dioxide concentrations expected by the early part of the next century would result in elimination of Douglas fir (Pseudotsuga taxifolia) from the lowlands of California and Oregon, because rising temperatures would preclude the seasonal chilling this species requires for seed germination and shoot growth (Leverenz and Lev, in press). On a larger scale, other simulations indicate that projected temperature changes (exclusive of changes in precipitation and soil characteristics) caused by a doubling of carbon dioxide concentration would result in the shifting of entire ecosystem complexes, including the loss of as much as 37% of boreal forest (Emanuel et al., 1985).
Because each species disperses at a different rate, major climatic changes typically result in a resorting of the species constituting natural communities and the creation of new plant and animal associations (e.g., Van Devender and Spaulding, 1979; see also Figure 51–2), thereby causing new, sometimes stressful interactions among species.
FIGURE 51–2 (a) Initial distribution of two species, A and B, whose ranges largely overlap, (b) In response to climatic change, latitudinal shifting occurs at species-specific rates and the ranges disassociate.
LOCALIZED SPECIES MAY NOT BE ABLE TO COLONIZE NEW HABITAT
If the entire range occupied by a species becomes unsuitable because of climate change, the species must either colonize a new, more suitable habitat or become extinct. The smaller the present range, the more likely it will be that the species will find the entire habitat unsuitable and therefore that extinction will occur. As discussed below, the vulnerability of many species will be increased by human encroachment that restricts them to small areas. Species restricted to reserves, like the one illustrated in Figure 51–3, are good examples.
Imagine a restricted population like that represented in Figure 51–3. What is the chance that colonists, such as seeds or migrating animals, from the original population will find new habitat before the parent population becomes extinct? It will, of course, depend upon a number of factors: how much suitable area there is (i.e., the size of the target the colonists must reach), how far away the suitable area is, how many potential colonists are sent out (which will be a function of how large the original population is and the reproductive strategy of the species), how efficient these colonists are at dispersing themselves, how many physical barriers to dispersal exist, and how long some individuals within the original population can survive to reproduce.
Although the number of colonists produced per parent and their intrinsic dispersal ability are likely to be essentially the same as during past times when species had to respond to climate change, this is not so for the other variables. For many species, the target areas to be reached will be reduced by development, the number of potential colonists will be reduced through reduction of the parent population, the length of time the parent population is allowed to exist may be reduced both through the rapidity of the climate change and development pressures, and, im-
portantly, many more barriers to dispersal in the form of agriculture, urbanization, and other types of habitat degradation will be added to the natural physical barriers of mountains, oceans, and deserts. The predicament faced by a species in this situation is illustrated in Figure 51–4 for the Engelmann spruce (Picea ccengelmanni).
For a plant, the Engelmann spruce is probably a moderate disperser. It has small, wind-dispersed seeds, and its natural dispersal rate, in the absence of barriers, has been estimated to be between 1 and 20 kilometers per century (Seddon, 1971). If we assume that climate change will cause a several-hundred kilometer shift in the potential range of many species in the United States during the next century, say 30 kilometers per year, a plant with the 1- to 20-kilometer per century rate of the Engelmann spruce would be in trouble. Although some species, such as plants propagated by spores, may be able to match the 30 kilometers per year needed, many other species could not disperse fast enough to compensate for the expected climatic change without human assistance. Even some large animals that are physically capable of rapid dispersal do not travel far for behavioral reasons. Rates for several species of deer, for example, have been observed to be less than 2 kilometers per year (Rapoport, 1982).
FIGURE 51–3 How climatic warming may turn biological reserves into former reserves. Hatching indicates: (a) species distribution before human habitation; (b) fragmented species distribution after human habitation; (c) species distribution after warming. SL indicates the southern limit of species range.
FIGURE 51–4 Obstacle course to be run by species facing climatic change in a human-altered environment. To win, a population must track its shifting climatic optimum and reach suitable habitat north of the new southern limit of the species range. SL1 is the species southern range limit under initial conditions. SL2 is the southern limit after climate change. The model assumes a plant population consisting of a single species, whose distribution is determined solely by temperature. After a 3°C rise in temperature, the population must have shifted 250 kilometers to the north to survive, based on Hopkins bioclimatic law (MacArthur, 1972). Shifting will occur by simultaneous range contraction from the south and expansion by dispersion and colonization to the north. Progressive shifting depends upon propagules that can find suitable habitat in which to mature and in turn produce propagules that can colonize more habitat to the north. Propagules must pass around natural and artificial obstacles like mountains, lakes, cities, and farm fields. The Engelmann spruce has an estimated, unimpeded dispersal rate of 20 kilometers/100 years (Seddon, 1971). Therefore, for this species to win by colonizing habitat to the north of the shifted hypothetical limit would require a minimum of 1,250 years.
We know these threats are more than speculation, because the fossil record provides evidence that not only have ranges shifted in response to climate change, but in some cases their total extent was drastically reduced. For example, a large and diverse group of plant genera, including watershield (Brasenia), sweetgum (Liquidambar), yellow poplar (Liriodendron), magnolia (Magnolia), moonseed (Menispermum), hemlock (Tsuga), cedar (Thuja), and cypress (Chamaecyparis), were
found in both Europe and North America during the Tertiary period. But during the Pleistocene ice ages, these all became extinct in Europe, presumably because the east-west orientation of such barriers as the Pyrenees, the Alps, and the Mediterranean blocked southward migration, while they persisted in North America, which has longitudinally oriented mountain ranges (Tralau, 1973).
MANAGEMENT IMPLICATIONS
How might the threats posed by climatic change to natural communities be mitigated? One basic truth is that the less populations are reduced by development now, the more resilient they will be to climate change. Thus, an excellent way to start planning for climate change would be sound conservation now, in which we try to conserve more than just the minimum number of individuals of a species necessary for present survival.
In terms of responses specifically directed at the effects of climate change, the most environmentally conservative action would be to halt or slow global warming. Granted, this would be difficult, not only because fossil fuel use will probably increase as the world’s population grows but also because effective action would demand a high degree of international cooperation. If efforts to prevent global warming fail, however, and if global temperatures continue to rise, then amelio-rating the negative effects of climatic change on biological resources will require substantially increased investment in the purchase and management of reserves.
To make intelligent plans for siting and managing reserves, we need more knowledge. We must refine our ability to predict future conditions in reserves. We also need to know more about how temperature, precipitation, carbon dioxide concentrations, and interspecific interactions determine range limits (see, for example, Picton, 1984, and Randall, 1982) and, most important, how they can cause local extinctions.
Reserves that suffer from the stresses of altered climate will require carefully planned and increasingly intensive management to minimize species loss. To preserve some species, for example, it may be necessary to modify conditions within reserves, such as irrigation or drainage in response to new moisture patterns. Because of changes in interspecific interactions, competitors and predators may need to be controlled and invading species weeded out. The goal would be to stabilize the composition of existing communities, much as the habitat of Kirtland’s warbler (Dendroica kirtlandii) is periodically burned to maintain pine woods (Leopold, 1978).
In attempting to understand how climatically stressed communities may respond and how they might be managed to prevent the gradual depauperization of their constituents, restoration studies, or more properly, community creation experiments can help. Communities may be created outside their normal climatic ranges to mimic the effects of climate change. One such relocation community is the Leopold Pines experimental area at the University of Wisconsin Arboretum in Madison, where there is periodically less rainfall than in the normal pine range several hundred kilometers to the north (W.R.Jordan III, University of Wisconsin, Madison, personal communication, 1985). Researchers have found that although the pines themselves do fairly well once established at the Madison site, many of
the other species that would normally occur in a pine forest, especially the various herbs and small shrubs, have not flourished, despite several attempts to introduce them.
If management measures are unsuccessful, and old reserves do not retain necessary thermal or moisture characteristics, individuals of disappearing species might be transferred to new reserves. For example, warmth-intolerant ecotypes or subspecies might be transplanted to reserves nearer the poles. Other species may have to be periodically reintroduced in reserves that experience occasional climate extremes severe enough to cause extinction, but where the climate would ordinarily allow the species to survive with minimal management. Such transplantations and reintroductions, particularly involving complexes of species, will often be difficult, but some applicable technologies are being developed (Botkin, 1977; Lovejoy, 1985).
To the extent that we can still establish reserves, pertinent information about changing climate and subsequent ecological response should be used in deciding how to design and locate them to minimize the effects of changing temperature and moisture. Considerations include:
-
The existence of multiple reserves for a given species or community type increases the probability that if one reserve becomes unsuitable for climatic reasons, the organisms may still be represented in another reserve.
-
Reserves should be heterogeneous with respect to topography and soil types, so that even given climatic change, remnant populations may be able to survive in suitable microclimatic areas. Species may survive better in reserves with wide variations in altitude, since from a climatic point of view, a small altitudinal shift corresponds to a large latitudinal one. Thus, to compensate for a 2°C rise in temperature, a Northern Hemisphere species can achieve almost the same result by increasing its altitude only some 500 meters as it would by moving 300 kilometers to the north (MacArthur, 1972).
-
As models of climate become more refined, pertinent information should be considered in making decisions about where to site reserves in order to minimize the effects of temperature and moisture changes. In the Northern Hemisphere, for example, where a northward shift in climate zones is likely, it makes sense to locate reserves as near the northern limit of a species’ or community’s range as possible, rather than farther south, where conditions are likely to become unsuitable more rapidly.
-
Maximizing the size of reserves will increase long-term persistence of species by increasing the probability that suitable microclimates exist, by increasing the probability of altitudinal variation, and by increasing the latitudinal distance available to shifting populations.
-
In the future, flexible zoning around reserves could allow us to move reserve boundaries in response to changing climatic conditions. Also, as habitat inside a reserve becomes unsuitable for the species or communities within, reserve land might be traded for nonreserve land that either remains suitable or becomes so as the climate changes. The success of these strategies, however, would depend on a highly developed restoration technology that is capable of guaranteeing, in effect, the portability of species and whole communities.
ACTIONS THAT CAN BE TAKEN
The best solutions to the ecological upheaval resulting from climatic change are not yet clear. In fact, little attention has been paid to the problem. What is clear, however, is that these changes in climate would have tremendous impact on communities and populations isolated by development and that by the middle of the next century, they may dwarf any other consideration in planning for reserve management. The problem may seem overwhelming. One thing is worth keeping in mind, however: the more fragmented and smaller populations of species will be less resilient to the new stresses brought about by climate change. Thus, one of the best things that can be done in the short term is to minimize further encroachment of development upon existing natural ecosystems. Furthermore, we must refine our climatological predictions and increase our understanding of how climate affects species, both individually and in their interactions with each other. Such studies may allow us to identify those areas where communities will be most stressed as well as alternative areas where they might best be saved. Meanwhile, efforts to improve techniques for managing communities and ecosystems under stress and for restoring them when necessary must be carried forward energetically.
ACKNOWLEDGMENTS
Ideas presented in this paper are based in part on an article, “The Greenhouse Effect and Nature Reserves,” by R.L.Peters II and J.D.S.Darling, published in BioScience 35(11):707–717, 1985. Research was supported by the Conservation Foundation. I wish to thank Joan Darling for her thoughtful collaboration, and I am grateful to Kathy Freas, James Hansen, Bill Jordan, William Kellogg, Thomas Lovejoy, Norman Myers, Elliott Norse, Pamela Parker, Christine Schonewald-Cox, James Titus, and Bruce Wilcox for encouragement, helpful comments, and review of manuscript drafts during the development of these ideas.
REFERENCES
Baker, R.G. 1983. Holocene vegetational history of the western United States. Pp. 109–125 in H. E.Wright, Jr., ed. Late-Quaternary Environments of the United States. Volume 2. The Holocene. University of Minnesota Press, Minneapolis.
Bernabo, J.C., and T.Webb III. 1977. Changing patterns in the Holocene pollen record of northeastern North America: A mapped summary. Quat. Res. 8:64–96.
Botkin, D.B. 1977. Strategies for the reintroduction of species into damaged ecosystems. Pp. 241–260 in J.Cairns, Jr., K.L.Dickson, and E.E.Herricks, eds. Recovery and Restoration of Damaged Ecosystems. University Press of Virginia, Charlottesville, Va.
Davis, M.B. 1983. Holocene vegetational history of the eastern United States. Pp. 166–181 in H.E.Wright, Jr., ed. Late-Quaternary Environments of the United States. Volume 2. The Holocene. University of Minnesota Press, Minneapolis.
Dorf, E. 1976. Climatic changes of the past and present. Pp. 384–412 in C.A.Ross, ed. Paleo-biogeography: Benchmark Papers in Geology 31. Dowden, Hutchinson, and Ross, Stroudsburg, Pa.
Emanuel, W.R., H.H.Shugart, and M.P.Stevenson. 1985. Response to comment: Climatic change and the broadscale distribution of terrestrial ecosystem complexes. Clim. Change 7:457–460.
Flohn, H. 1979. Can climate history repeat itself? Possible climatic warming and the case of paleo-climatic warm phases. Pp. 15–28 in W.Bach, J.Pankrath, and W.W.Kellogg, eds. Man’s Impact on Climate. Elsevier Scientific Publishing, Amsterdam.
Ford, M.J. 1982. The Changing Climate: Responses of the Natural Fauna and Flora. George Allen and Unwin, London. 190 pp.
Frankel, O.H., and M.E.Soulé. 1981. Conservation and Evolution. Cambridge University Press, Cambridge. 327 pp.
Hansen, J., D.Johnson, A.Lacis, S.Lebedeff, P.Lee, D.Rind, and G.Russell. 1981. Climate impact of increasing atmospheric carbon dioxide. Science 213:957–966.
Hansen, J., A.Lacis., D.Rind, G.Russell, I.Fung, and S.Lebedeff. In press. Evidence for future warming: How large and when. In W.E.Shands and J.S.Hoffman, eds. The Greenhouse Effect, Climate Change, and U.S. Forests. Conservation Foundation, Washington, D.C.
Hoffman, J.S., D.Keyes, and J.G.Titus. 1983. Projecting Future Sea Level Rise: Methodology, Estimates to the Year 2100, and Research Needs. Strategic Studies Staff, Office of Policy Analysis, Office of Policy and Resource Management, U.S. Environmental Protection Agency, Washington, D.C. 121 pp.
Kellison, R.C., and R.J.Weir. In press. Selection and breeding strategies in tree improvement programs for elevated atmospheric carbon dioxide levels. In W.E.Shands and J.S.Hoffman, eds. The Greenhouse Effect, Climate Change, and U.S. Forests. Conservation Foundation, Washington, D.C.
Kellogg, W.W., and R.Schware. 1981. Climate Change and Society: Consequences of Increasing Atmospheric Carbon Dioxide. Westview, Boulder, Colo. 178 pp.
Lemon, E.R. 1983. CO2 and Plants: The Response of Plants to Rising Levels of Atmospheric Carbon Dioxide. Westview, Boulder, Colo. 280 pp.
Leopold, A.S. 1978. Wildlife and forest practice. Pp. 108–120 in H.P.Brokaw, ed. Wildlife and America. Council on Environmental Quality, U.S. Government Printing Office, Washington, D.C.
Leverenz, J.W., and D.J.Lev. In press. Effects of CO2-induced climate changes on the natural ranges of six major commercial tree species in the western U.S. In W.E.Shands and J.S.Hoffman, eds. The Greenhouse Effect, Climate Change, and U.S. Forests. Conservation Foundation, Washington, D.C.
Lovejoy, T.E. 1980. A projection of species extinctions. Pp. 328–331 in The Global 2000 Report to the President: Entering the Twenty-First Century. Council on Environmental Quality and the U.S. Department of State. U.S. Government Printing Office, Washington, D.C.
Lovejoy, T.E. 1985. Rehabilitation of Degraded Tropical Rainforest Lands. Commission on Ecology Occasional Paper No. 5. International Union for the Conservation of Nature and Natural Resources, Gland, Switzerland.
MacArthur, R.H. 1972. Geographical Ecology. Harper & Row, New York. 269 pp.
Machta, L. 1983. Effects of non-CO2 greenhouse gases. Pp. 285–291 in Changing Climate: Report of the Carbon Dioxide Assessment Committee. National Academy Press, Washington, D.C.
Manabe, S., R.T.Wetherald, and R.J.Stouffer. 1981. Summer dryness due to an increase of atmospheric CO2 concentration. Clim. Change 3:347–386.
Miller, W.F., P.M.Dougherty, and G.L.Switzer. In press. Effect of rising CO2 and potential climate change on loblolly pine distribution, growth, survival, and productivity. In W.E.Shands and J.S.Hoffman, eds. The Greenhouse Effect, Climate Change, and U.S. Forests. Conservation Foundation, Washington, D.C.
Myers, N. 1979. The Sinking Ark. Pergamon Press, New York. 307 pp.
NRC (National Research Council). 1983. Changing Climate: Report of the Carbon Dioxide Assessment Committee. National Academy Press, Washington, D.C. 496 pp.
Peters, R.L., II, and J.D.S.Darling. 1985. The greenhouse effect and nature reserves: Global warming would diminish biological diversity by causing extinctions among reserve species. BioScience 35(11):707–717.
Picton, H.D. 1984. Climate and the prediction of reproduction of three ungulate species. J. Appl. Ecol. 21:869–879.
Ramanathan, V., R.J.Cicerone, H.B.Singh, and J.T.Kiehl. 1985. Trace gas trends and their potential role in climate change. J. Geophys. Res. 90:5547–5566.
Randall, M.G.M. 1982. The dynamics of an insect population throughout its altitudinal distribution: Coleophora alticolella (Lepidoptera) in northern England. J. Anim. Ecol. 51:993–1016.
Rapoport, E.H. 1982. Areography. Geographical Strategies of Species. Pergamon Press, New York. 269 pp.
Schneider, S.H., and R.Londer. 1984. The Coevolution of Climate and Life. Sierra Club Books, San Francisco. 563 pp.
Schonewald-Cox, C.M., S.M.Chambers, B.MacBryde, and W.L.Thomas. 1983. Genetics and Conservation. A Reference for Managing Wild Animal and Plant Populations. Benjamin-Cummings Publishing, Menlo Park, Calif. 722 pp.
Seddon, B. 1971. Introduction to Biogeography. Barnes and Noble, New York. 220 pp.
Soulé, M.E. 1986. Conservation Biology: The Science of Scarcity and Diversity. Sinauer Associates, Sunderland, Mass. 584 pp.
Soulé, M.E., and B.A.Wilcox. 1980. Conservation Biology: An Evolutionary-Ecological Perspective. Sinauer Associates, Sunderland, Mass. 395 pp.
Tralau, H. 1973. Some Quaternary plants. Pp. 499–503 in A.Hallam, ed. Atlas of Palaeobiogeography. Elsevier Scientific Publishing, Amsterdam.
Van Devender, T.R., and W.G.Spaulding. 1979. Development of vegetation and climate in the southwestern United States. Science 204:701–710.
Wigley, T.M.L., P.D.Jones, and P.M.Kelly. 1980. Scenario for a warm, high CO2 world. Nature 283:17.













































