The Scientific Examination of Works of Art on Paper
Paul M. Whitmore
Research Center on the Materials of the Artist and Conservator
Carnegie Mellon University
Pittsburgh, Pennsylvania
ABSTRACT
The scientific examination of works of art on paper utilizes tools from the very simple to state-of-the-art analytical instrumentation, depending in large part on the question that is the objective of the investigation. Identifying pigments or paper fibers is straightforward, constrained only by the size of the samples that can be removed for destructive analysis. Inks are more difficult because of the lack of pronounced chemical differentiation between the ink types and because of possible interferences in the analyses from the paper substrate. Paper can be characterized easily to an extent, in identifying a watermark or the risk of deterioration from a high acid content, but the monitoring of the condition and degradation of paper remains an extremely difficult challenge. The assessment of light sensitivity, which is not easy to determine by merely identifying material composition, has been made straightforward by the development of a device that allows rapid, essentially nondestructive fading tests. Those tests are now being exploited to survey groups of objects to determine whether one may make generalizations about their exhibition needs. The further adaptation of nondestructive or micro-scale destructive analytical tools in the study of works of art on paper promises to allow even more extensive investigations of the creation and preservation of these objects.
INTRODUCTION
The scientific study of works of art on paper shares common objectives with the technical studies of any work of art. Artifacts are examined in order to answer art historical questions about the origin of a work, namely, where, when, and by whom a work was created. The scientific examinations seeking to answer these questions generally require identification of the materials and working methods used to craft the object. Other studies seek to answer basic questions about the care of the artifact: its physical and chemical condition, causes for deterioration, and vulnerability to storage or exhibition conditions.
Technical studies of paper-based artifacts tend to resemble the study of paintings, because many paper objects actually are paintings that just happen to be executed on a paper support. Manuscript illuminations, watercolors, lithographic prints—these objects could easily be viewed as paintings, amenable to analyses of the colorants, paint media, or layer structure of paints observable in cross-sections. Apart from the occasional thinness of the paint layer itself, as in watercolor paintings, or binder-poor paint layers, such as in pastels, these paper-based paintings can often be studied as one would study any other painting.
Despite this similarity, many works of art on paper present special circumstances that constrain analyses or warrant unusual examination techniques. Paper artifacts tend to be small: The sheets were traditionally made in molds that could be manipulated by people, and these sheets were then cut down for use. Thus, books, prints, watercolors, and other paper-based art are relatively small, meant for close-up viewing within an arm’s length. For this reason, analytical methods that require removal of paint samples are often not feasible, for the damage to the artifact can sometimes be visible upon close inspection. Nondestructive tools, particularly optical spectroscopic or imaging techniques, are more widely used to study these objects.
Another distinction between paper-based objects and traditional paintings is the use of the paper substrate as part of the image itself. Particularly with such graphic art as drawings and prints but also with printed text or even thinly painted watercolors, the paper substrate is exposed and is part of the image. Thus, the color of the paper and its surface texture are important contributors to the appearance and visual appeal of the object, and study of the paper and its preservation is of great importance. (Occasionally in historical times and more frequently in the twentieth century, paintings too have been created with unpainted canvas as part of the image. For these objects the concern about the appearance and stability of the canvas is of course shared.) A complication in studying objects in which the paper is so intimately associated with the drawing media is the discrimination between the two, so that many analyses must have very small spatial or depth resolution, or contributions to the detected signal from the paper must be subtracted.
Paper-based collections in museums are known to pose some of the most
common preservation problems because many of the artifacts that are now prized were not created as lasting works of art but as more utilitarian objects. Because paper was inexpensive and widely available through much of history, it has seen use for many purposes, a primary one being for communication and recording of information. Some of these artifacts, such as books, were meant to last for a long time, but others, such as newspapers, announcements, or letters, were often not created with posterity in mind. Thus, it is not uncommon for museums and archives to have paper artifacts that are delicate or deteriorating because of their creation with impermanent materials or techniques. Preservation problems are common, particularly with those objects that were not made as art objects.
This review will survey the examination techniques of paper-based objects that are used both for art historical investigations as well as for preservation studies. Some of those techniques are routine and can be found in many well-equipped museum laboratories; others are less widely available and have not found widespread use. This survey will conclude with a description of a relatively new tool developed to detect a particular vulnerability, the susceptibility of colored materials to fade from light exposure, and illustrate its use for the study of Japanese woodblock prints.
SURVEY OF EXAMINATION AND MATERIAL IDENTIFICATION TECHNIQUES
The most common technical investigation for paintings or colored prints on paper involves identification of the pigments in the paint. For this, the routine analytical tools of polarizing-light microscopy, X-ray diffraction, and elemental analyses by X-ray fluorescence are commonly employed, usually on samples of the paint that have been removed from the artifact. Descriptions of these tools can be found in accounts of painting examinations, or in reference books devoted to pigment identification (Feller, 1986; Roy, 1993; FitzHugh, 1997). Nondestructive techniques can also sometimes be used to identify pigments on paper objects. Open-air X-ray fluorescence is used for elemental analyses of pigments, and Raman spectroscopy and Raman microscopy have been found useful for examining both pigments in paints and dyes in colored paper (Bell et al., 2000; Best et al., 1995). Some pigments have distinctive features in the visible spectrum (Schweppe and Roosen-Runge, 1986; Leona and Winter, 2001), while others, like Indian yellow, can be detected by their peculiar fluorescence observable under ultraviolet light illumination (Baer et al., 1986).
Drawing materials can also be studied, although they present some difficulties. Early drawings were created using metal tools or wires as drawing implements (thus the name “metalpoints” for these drawings), and they can be analyzed by measuring the elemental composition of the metals in the lines (by X-ray fluorescence, typically). Inks are more problematic, with the exception of iron gall inks, which can be distinguished by the presence of iron in X-ray fluorescence or
in more unusual techniques such as Mössbauer spectroscopy (Rusanov et al., 2002) or PIXE (Budnar et al., 2001). Inks can also be analyzed for the trace elements they contain, introduced in the ink ingredients or as residues from the printing process. Inks in early books (such as a Gutenberg Bible) have been examined for these trace elements by synchrotron-excited X-ray fluorescence in the hope of distinguishing books produced in the early German printing shops (Mommsem et al., 1996). Other organic inks, such as sepia (cuttlefish ink), bistre (from soot), or such black drawing media as charcoal, bone black, lamp black, ivory black, or graphite cannot usually be distinguished by their elemental composition (although bone black is often detected by the presence of phosphorus), nor do the infrared spectra of these inks usually present characteristic features useful for their identification. Polarizing-light microscopy remains a common tool to discriminate between inks on the basis of their particle morphologies. The media used as pigment binders for drawing and painting materials can be identified by analyzing the organic composition of micro-samples. Of the various methods available the most useful are the gas chromatography/mass spectroscopy analyses that have been developed for oils and resins used as paint binders (Mills and White, 2000; Schilling and Khanjian, 1996) and more recently adapted for the study of gums used in watercolors or gouaches (Vallance et al., 1998).
In addition to the study of the image-forming materials, the examination of the paper itself is also often a clue to the artifact’s origin. Paper is usually differentiated by its fiber composition, its physical characteristics, and its manufacturing method. The fibers can be studied with optical microscopy, and the plant origin of the component fibers can be determined by appearance or by reaction to certain stains, such as Hertzberg or Graff C stains. The fiber type, length, and heterogeneity can all be distinctive, as can such physical dimensions as sheet thickness. The evidence of manufacture is most easily detected in the pattern left by the papermaking mold, typically a pattern of lines called chain and laid lines in so-called “laid” paper. Watermarks, the decorative patterns often woven into the wire molds or embossed on the cast sheets, are also the most obvious characteristic patterns of the paper manufacture. The evidence of chain and laid lines and watermarks can be captured in any of a number of ways, with transmitted light photography or with beta or soft X-ray radiography, and various image processing tools have been applied to enhance such records (erasing interferences from the printing, for example) and making them more useful for indexing and retrieval for comparison in a reference database (Brown and Mulholland, 2002). The presence of sizing (a water-resistant finish on the surface of the paper) can be determined by infrared spectroscopy or colorimetric methods (Barrett and Mosier, 1995), and fillers (typically finely ground minerals or clays added for increased opacity of the sheet) can be identified by optical or electron microscopies (Browning, 1969). The fiber, finish, or watermark, along with other historical evidence, can assist in tracing a paper’s origin (Hunter, 1978), yet many challenges remain (Slavin et al., 2001).
EXAMINATIONS TO ASSESS CONDITION AND PRESERVATION PROBLEMS
As with all works of art, preserving works of art on paper focuses on maintaining both the physical integrity of the artifact and its appearance. The physical integrity is derived mainly from the paper sheet itself, and preservation of the sheet’s cohesive strength is of paramount importance. (For books and archival materials that are handled by users, other sheet properties, such as flexibility, are also important, but for works of art that are usually mounted in a frame, the physical stresses are usually merely the tensile stresses from the paper’s weight and from its reaction to temperature and humidity.)
The cohesive strength of a sheet of paper is derived from the strength of its constituent fibers and of the bonds between the fibers. Aging tends to reduce the fiber strength, and old weak papers are usually seen to fail from broken fibers rather than by unraveling from weakened interfiber bonds. The reduction of fiber strength is in turn a result of the breakdown of cellulose, the natural polymer of glucose that composes plant fibers. Chemical degradation breaks cellulose chains, which reduces the average molecular weight but more importantly also breaks the connections between the highly crystalline cellulose zones. This progressive rupture of the tie chains, the amorphous cellulose chains connecting the crystallites and imparting the cohesive strength to the fiber, is the underlying aging chemistry leading to physical failure of the paper sheet.
Unfortunately, there are no analytical tools that can allow detection of such deterioration in a paper artifact without destructive analysis of unacceptably large portions of paper. Typically for degrading polymers, nondestructive tools such as infrared spectroscopy do not have the sensitivity to detect the production of the very small concentrations of new chain ends in the degrading cellulose. Recent studies suggest that production of glucose or xylose residues (Erhardt and Mecklenberg, 1995) or low molecular weight acids (Shahani and Harrison, 2002) may be easier to track as some measure of cellulose reaction, but these techniques have not yet been applied to artifacts. Other efforts to develop micro-scale molecular weight analyses for cellulose have reduced the amount of paper needed (Rohrling et al., 2002), but a recent molecular weight analysis of cellulose in a single paper fiber, while successful, also suggests that such small sample sizes may not be typical of the other fibers or representative of the average molecular weight of larger samples (Stol et al., 2002). Thus, even if the analytical procedure can be adapted, the slow deterioration of the cellulose may not be easily tracked by successive measurements of individual fibers over time.
While the deterioration of paper artifacts may be difficult to detect directly, many years of investigation of cellulose degradation have clearly indicated that there are other materials that may be reliable indicators of instability in the paper. Acidity is well established as a catalyst for the hydrolytic breakdown of cellulose, the most important of the known degradation chemistries. Lignin is primarily
responsible for the discoloration of groundwood papers, and iron and copper impurities can also act both as acid catalysts for hydrolysis and as catalysts for oxidative breakdown of cellulose. It is much easier to determine the presence of these sensitizing agents in paper than to track the slow deterioration of the cellulose, so the study of artifact materials often does not go beyond a pH measurement, the detection of lignin with phloroglucinol stain or an infrared spectrum, or the analysis for iron or copper impurities by a technique such as electron spin resonance spectroscopy (Attanasio et al., 1995). Iron present not as a paper impurity but as an ink component is also a well-known and easily identifiable risk factor for the preservation of manuscript and print materials.
The maintenance of the image-forming materials, particularly the colored paints and inks used to create the image, is another objective of preservation strategies. Light exposure is the most common hazard to the pigments and dyes used on art objects. In contrast to preserving the paper support, in which the deterioration is difficult to monitor and easier to predict by detecting the presence of destabilizing components, the loss of color is easy to monitor by periodic color measurements but difficult to predict. The light stability of a pigment depends not only on the material but also on its preparation, particle size, and prior fading history. None of these is easy to determine from study of the pigments, and until recently the only means to detect light sensitivity was to monitor the damage inflicted by light exposure.
Recently a new device has been developed to determine the risk of future fading from light exposure (Whitmore et al., 1999). That device operates as a reflectance spectrophotometer using a very intense focused beam from a xenon lamp as the illumination for the measurement (see Figure 1). By making rapid repeated spectral measurements while the material is illuminated by the intense light, very slight degrees of fading can be detected in light-sensitive materials in only a few minutes (see Figure 2). Because of the high precision of the spectrum acquisition, extremely small amounts of fading are easily recorded, and the test can be stopped before perceptible changes to the art object have been produced. All of the different color areas on a work of art can be tested, and the overall sensitivity of the object can be judged by the fading rate of the most light-sensitive color (see Figure 3). These tests can be used to develop exhibition requirements that are tailored to the needs of the object, with the very light-sensitive objects receiving greater care (less frequent exhibition at lower light levels) so that they do not suffer from fading damage caused by inappropriate display. These same tests, done with filtered illumination, can also be used to test the effectiveness of different lighting in reducing fading rates. By performing the tests in air or under an inert gas, the efficacy of oxygen-free housings for slowing the fading of works of art can also be assessed (see Figure 4).
It has been found that this fading test can also be used to identify pigments, not by their elemental or chemical constitution but rather on the basis of their photochemical reaction. Prussian blue, a ferric ferrocyanide complex used in art
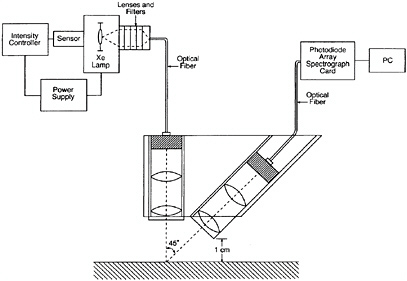
FIGURE 1 Schematic of fading tester. Reprinted from the Journal of the American Institute for Conservation, vol. 38, no. 3, with the permission of the American Institute for Conservation of Historic and Artistic Works, 1717 K St., NW, Suite 200, Washington, D.C. 20006.
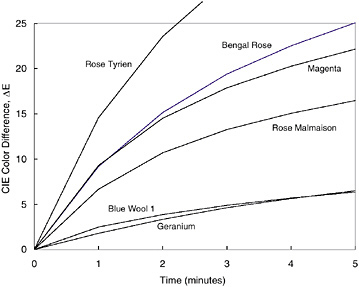
FIGURE 2 Fading test results for selected Winsor & Newton gouache paints. “Blue Wool 1” designates fading test results for the ISO Blue Wool no. 1, the most light-sensitive of the standard cloths. Reprinted from the Journal of the American Institute for Conservation, vol. 38, no. 3, with the permission of the American Institute for Conservation of Historic and Artistic Works, 1717 K St., NW, Suite 200, Washington, D.C. 20006.
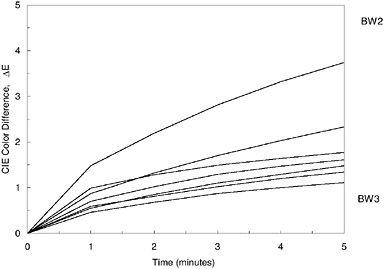
FIGURE 3 Fading test results for all the different color areas on a Japanese woodblock print (Yoshitoshi, Carnegie Museum of Art No. 89.28.1516). “BW2” and “BW3” designate the degree of color difference produced after five minutes in fading tests of ISO Blue Wool fading standards nos. 2 and 3.
since the early eighteenth century, is known to fade reversibly during light exposure, with the blue color being recovered in a subsequent dark reaction (Ware, 1999). The fading tests of Prussian blue using the tester described above demonstrated this peculiar reversible fading behavior on a cyanotype, an early photographic process used to create blueprints (see Figure 5) (Whitmore et al., 2000).
In addition to these fading tests designed to evaluate individual artifacts, current studies are measuring the fading rates of particular colorants in Japanese woodblock prints from different eras, printed at different depths of color, and of varying degrees of prior fading. Results of such a population study will reveal whether there is general consistency or a wide variation in light sensitivity for particular materials. If there is widely varying behavior, fading tests must be performed on each object in order to determine the sensitivity and light exhibition needs. If there are very similar fading rates among the different applications of a pigment, one need not test every object and can instead safely use a rule of thumb to make such judgments of the object’s required care. This formulation of new rules of thumb, based on actual fading sensitivities observed in a large population of objects, will bring a new level of intuition about how to preserve objects. The results of tests on a large number of Japanese woodblock prints indicate that the fading of the colorant dayflower blue (aobana) is very regular, and its sensitivity can probably be safely estimated without individual fading tests (see Figure 6a). By contrast, yellow passages on the Japanese prints vary greatly in their light
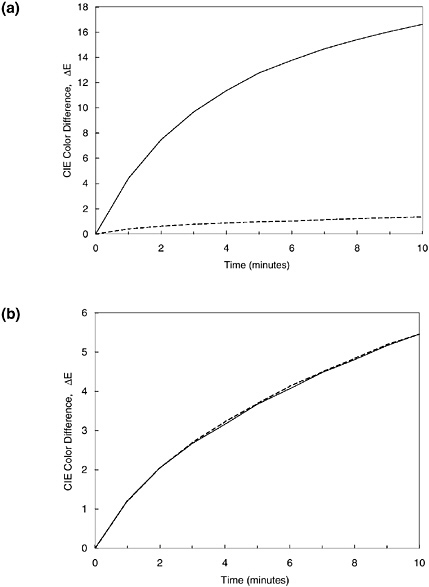
FIGURE 4 Fading test results in air (solid lines) and under nitrogen (dashed lines). (a) Results for a gouache paint (Winsor & Newton Rose Bengal), showing slower fading in anoxic environment. (b) Results for ISO Blue Wool cloth no. 1, showing no difference in fading rate in anoxic environment. Reprinted from the Journal of the American Institute for Conservation, vol. 38, no. 3, with the permission of the American Institute for Conservation of Historic and Artistic Works, 1717 K St., NW, Suite 200, Washington, D.C. 20006.
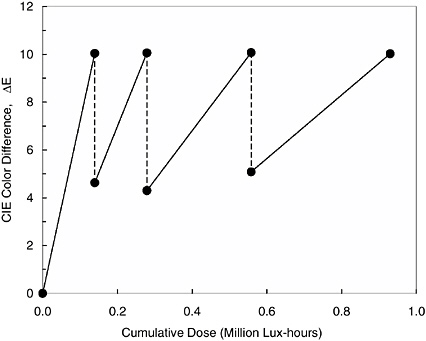
FIGURE 5 Reversible fading of Prussian blue under exposure in fading tester. Solid lines are fading measured during light exposure; dashed lines represent period of recovery, and return of blue color (smaller color difference) in the dark. Reprinted from Tradition and Innovation: Advances in Conservation, eds. A. Roy and P. Smith, with the permission of the International Institute for Conservation of Historic and Artistic Works, 6 Buckingham St., London WC2N 6BA, UK.
sensitivity, probably because many different kinds of natural colorants were used in the printing (see Figure 6b). These materials will require individual testing in order to assess their fading risks.
CONCLUSION
The scientific examination of works of art on paper utilizes tools from the very simple to state-of-the-art analytical instrumentation, depending in large part on the question that is the objective of the investigation. Identifying pigments or paper fibers is relatively easy, while inks are more challenging because of the lack of pronounced chemical differentiation between the ink types and because of possible interferences in the analyses from the paper substrate. Paper can be characterized easily to an extent, in identifying a watermark or the risk of deterioration from a high acid content, but the monitoring of the condition and degradation of paper—or for that matter, any polymeric material—remains an ex-
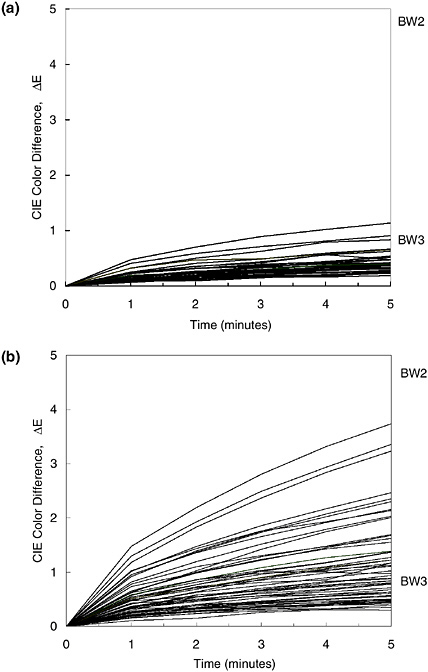
FIGURE 6 Fading test results for (a) 36 dayflower blue (aobana) passages on 25 different Japanese woodblock prints in the collection of the Carnegie Museum of Art; and (b) 55 yellow passages on 48 prints from that collection. “BW2” and “BW3” denote the color change produced in ISO Blue Wool fading standards nos. 2 and 3, respectively, after a five-minute exposure in the fading tester.
tremely difficult challenge. The assessment of light sensitivity, which is not easy to determine by merely identifying material composition, has been made straightforward by the development of a device that allows rapid, essentially nondestructive fading tests. Those tests are now being exploited to survey groups of objects to determine whether one may make generalizations about their exhibition needs. The further adaptation of nondestructive or micro-scale destructive analytical tools in the study of works of art on paper promises to allow even more extensive investigations of the creation and preservation of these objects.
REFERENCES
Attanasio, D., D. Capitani, C. Federici, and A. L. Segre. 1995. Archaeometry 37:377-384.
Baer, N. S., A. Joel, R. L. Feller, and N. Indictor. 1986. In Artists’ Pigments, vol. 1, ed. R. L. Feller, pp. 17-36. Cambridge, U.K.: Cambridge University Press.
Barrett, T., and C. Mosier. 1995. Journal of the American Institute for Conservation 34:173-186.
Bell, S. E. J., E. S. O. Bourguignon, A. C. Dennis, J. A. Fields, J. J. McGarvey, and K. R. Seddon. 2000. Analytical Chemistry 72:234-239.
Best, S. P., R. J. H. Clark, M. A. M. Daniels, C. A. Porter, and R. Withnall. 1995. Studies in Conservation 40:31-40.
Brown, A. J. E., and R. Mulholland. 2002. In Works of Art on Paper, Books, Documents, and Photographs: Techniques and Conservation, eds. V. Daniels, A. Donnithorne, and P. Smith, pp. 21-26. London: International Institute for Conservation.
Browning, B. L. 1969. Analysis of Paper. New York: Marcel Dekker.
Budnar, M., J. Vodopivec, P. A. Mando, F. L. G. Casu, and O. Signorini. 2001. Restaurator 22:228-241.
Erhardt, D., and M. F. Mecklenberg. 1995. In Materials Issues in Art and Archaeology IV, eds. P. B. Vandiver, J. R. Druzik, J. L. G. Madrid, I. C. Freestone, and G. S. Wheeler, pp. 247-270. Pittsburgh: Materials Research Society.
Feller, R. L., ed. 1986. Artists’ Pigments, vol. 1. Cambridge, U. K.: Cambridge University Press.
FitzHugh, E. W., ed. 1997. Artists’ Pigments, vol. 3. Washington, D.C.: National Gallery of Art.
Hunter, D. 1978. Papermaking: The History and Technique of an Ancient Craft. New York: Dover.
Leona, M., and J. Winter. 2001. Studies in Conservation 46:153-162.
Mills. J. S., and R. White. 2000. The Organic Chemistry of Museum Objects, 2nd ed. London: Butterworth-Heinemann.
Mommsem, H., T. Beier, H. Dittmann, D. Heimermann, A. Hein, A. Rosenberg, M. Boghardt, E.-M. Hanebutt-Benz, and H. Halbey. 1996. Archaeometry 38:347-357.
Rohrling, J., A. Potthast, T. Rosenau, T. Lange, G. Ebner, H. Sixta, and P. Kosma. 2002. Biomacromolecules 3:959-968.
Roy, A., ed. 1993. Artists’ Pigments, vol. 2. Washington, D.C.: National Gallery of Art.
Rusanov, V., K. Chakarova, and T. Madolev. 2002. Applied Spectroscopy 56:1228-1236.
Schilling, M. R., and H. P. Khanjian. 1996. In Preprints of the 11th Triennial Meeting of the ICOM (International Council of Museums) Committee for Conservation, ed. J. Bridgland, pp. 220-227. London: James & James Ltd.
Schweppe, H., and H. Roosen-Runge. 1986. In Artists’ Pigments, vol. 1, ed. R. L. Feller, pp. 17-36. Cambridge, U.K.: Cambridge University Press.
Shahani, C. J., and G. Harrison. 2002. In Works of Art on Paper, Books, Documents, and Photographs: Techniques and Conservation, eds. V. Daniels, A. Donnithorne, and P. Smith, pp. 189-192. London: International Institute for Conservation.
Slavin, J., L. Sutherland, J. O’Neill, M. Haupt, and J. Cowan, eds. 2001. Looking at Paper: Evidence and Interpretation. Ottawa: Canadian Conservation Institute.
Stol, R., J. L. Pedersoli, Jr., H. Poppe, and W. T. Kok. 2002. Analytical Chemistry 74:2314-2320.
Vallance, S. L., B. W. Singer, S. M. Hitchen, and J. H. Townsend. 1998. Journal of the American Institute for Conservation 37:294-311.
Ware, M. 1999. Cyanotype: The History, Science, and Art of Photographic Printing in Prussian Blue. London: Science Museum; Bradford, West Yorkshire, U.K.: National Museum of Photography, Film, and Television.
Whitmore, P. M., C. Bailie, and S. Connors. 2000. In Tradition and Innovation: Advances in Conservation, eds. A. Roy and P. Smith, pp. 200-205. London: International Institute for Conservation.
Whitmore, P. M., X. Pan, and C. Bailie. 1999. Journal of the American Institute for Conservation 38:395-409.













