4
Functional and Economic Impact of Sleep Loss and Sleep-Related Disorders
CHAPTER SUMMARY Sleep loss and sleep disorders affect an individual’s performance, safety, and quality of life. Almost 20 percent of all serious car crash injuries in the general population are associated with driver sleepiness, independent of alcohol effects. Further, sleep loss and sleep disorders have a significant economic impact. The high estimated costs to society of leaving the most prevalent sleep disorders untreated are far more than the costs that would be incurred by delivering adequate treatment. Hundreds of billions of dollars a year are spent on direct medical costs associated with doctor visits, hospital services, prescriptions, and over-the-counter drugs. Compared to healthy individuals, individuals suffering from sleep loss, sleep disorders, or both are less productive, have an increased health care utilization, and an increased likelihood of accidents.
The public health consequences of sleep loss, night work, and sleep disorders are far from benign. Some of the most devastating human and environmental health disasters have been partially attributed to sleep loss and night shift work-related performance failures, including the tragedy at the Bhopal, India, chemical plant; the nuclear reactor meltdowns at Three Mile Island and Chernobyl; as well as the grounding of the Star Princess cruise ship and the Exxon Valdez oil tanker (NCSDS, 1994; NTSB, 1997; Moss and Sills, 1981; United States Senate Committee on Energy and National Resources, 1986; USNRC, 1987; Dinges et al., 1989). Each of these incidents not only cost millions of dollars to clean up, but also had a significant impact on the environment and the health of local communities.
Less visible consequences of sleep conditions take a toll on nearly every key indicator of public health: mortality, morbidity, performance, accidents and injuries, functioning and quality of life, family well-being, and health care utilization. This chapter begins with an overview of the consequences of sleep loss and sleep disorders on an individual’s performance, safety, and quality of life. Drawing on the available body of evidence, the chapter then describes the economic impact of sleep loss and sleep disorders.
PERFORMANCE AND COGNITION DEFICITS
Nearly all types of sleep problems are associated with performance deficits in occupational, educational, and other settings. The deficits include attention, vigilance, and other measures of cognition, including memory and complex decision making. This section addresses sleep loss and then turns to sleep-disordered breathing and other sleep disorders.
Sleep Loss Affects Cognitive Performance
Sleep loss had been largely dismissed as the cause of poor cognitive performance by early, yet poorly designed, research. The prevailing view until the 1990s was that people adapted to chronic sleep loss without adverse cognitive effects (Dinges et al., 2005). More recent research has revealed sleep loss-induced neurobehavioral effects, which often go unrecognized by the affected individuals. The neurobehavioral impact extends from simple measures of cognition (i.e., attention and reaction time) to far more complex errors in judgment and decision making, such as medical errors, discussed below and in Box 4-1. Performance effects of sleep loss include the following:
-
Involuntary microsleeps occur.
-
Attention to intensive performance is unstable, with increased errors of omission and commission.
|
BOX 4-1 Reducing Interns’ Work Hours in Intensive Care Units Lowers Medical Errors The longstanding debate over medical residents’ lengthy work hours pits patient safety advocates against those who view the practice as necessary for continuity of care, preparation for medical practice, and cost containment (Steinbrook, 2002). After years of debate, and the threat of federal regulations, the Accreditation Council for Graduate Medical Education changed its requirements in 2003 to restrict residents’ work hours to about 80 hours per week (ACGME, 2003). The policy permits no more than a maximum shift duration of 24 hours and overnight call no more than every third night. Does this revised policy protect patients? The Harvard Work Hours, Health and Safety Study compared a schedule of about 80 hours per week (termed the traditional schedule) with a reduced schedule that eliminated shifts of 24 hours or more and kept work hours under 63 per week. The trial was conducted in intensive care units because they typically have the longest hours and the highest rates of errors. The intervention schedule not only enhanced interns’ sleep duration and lowered their rate of attentional failures, but also reduced the rate of serious medical errors, according to two articles published in 2004 in the New England Journal of Medicine. In the first article, the investigators used a within-subjects design (n = 20 interns) and validated sleep duration by polysomnography and attentional failures by slow-rolling eye movements recorded during continuous electro-oculography. Under the intervention schedule, the article reported that residents slept nearly 6 more hours per week, and they experienced half the rate of attentional failures during on-call nights than under the traditional schedule (Lockley et al., 2004). The second article on medical errors reported results after randomizing interns to either the traditional or reduced schedule (Landrigan et al., 2004). Two physicians who directly observed the interns without awareness of their schedules identified serious medical errors, defined as causing or having the potential to cause harm to a patient. Errors were recorded by type (medication, diagnosis, and procedure) and in terms of number, or rate per 1,000 patient days. The study covered a total of 2,203 patient-days involving 634 admissions. Under the traditional schedule, interns made nearly 21 percent more medication errors and at least five times more diagnostic errors. Overall, the unitwide rate of serious medical errors was 22 percent higher in the traditional versus the intervention schedule (P < .001) as shown in the table below. The investigators concluded that reducing interns’ hours can lower the occurrence of serious medical errors in the intensive care unit. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-
Cognitive slowing occurs in subject-paced tasks, while time pressure increases cognitive errors.
-
Response time slows.
-
Performance declines in short-term recall of working memory.
-
Performance requiring divergent thinking deteriorates.
-
Learning (acquisition) of cognitive tasks is reduced.
-
An increase in response suppression errors in tasks requiring normal primarily prefrontal cortex function.
-
The likelihood of response preservation on ineffective solutions is increased.
-
Compensatory efforts to remain behaviorally effective are increased.
-
Although tasks may be done well, performance deteriorates as tasks duration increases (Durmer and Dinges, 2005).
Attention and reaction time are altered by experimental sleep loss, which leads to cumulative, dose-dependent deterioration of attention and reaction time (Figure 4-1). Deterioration is measured in part using the psy-
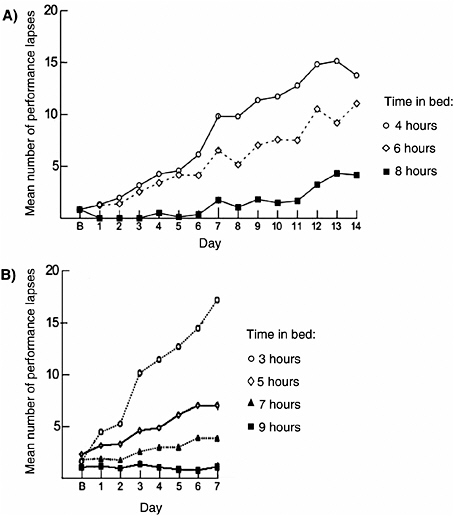
FIGURE 4-1 Repeated nights of sleep loss have cumulative cognitive impairment.
NOTE: B, baseline day.
SOURCES: (A) Van Dongen et al. (2003); (B) Belenky et al. (2003).
chomotor vigilance task (PVT), a test that requires continuous attention to detect randomly occurring stimuli and that is impervious to aptitude and learning effects. In one study 48 healthy subjects were randomized to 4, 6, or 8 hours time in bed for 14 days (Van Dongen et al., 2003). Investigators found a dose-dependent effect, which increased over time (Figure 4-1A). Performance deficits in individuals who slept 6 hours or less per night were similar to those observed in individuals after two nights of total sleep deprivation. Most striking was that study subjects remained largely unaware of their performance deficits, as measured by subjective sleepiness ratings. A second study (Belenky et al., 2003) showed a similar dose-dependent, cumulative effect over 7 days of sleep loss in 66 healthy volunteers (Figure 4-1B). Subjects were followed for 3 days after the period of sleep restriction, during which time they recovered, but not enough to return to their baseline levels. Imaging studies have demonstrated a physiological basis for cognitive impairments with sleep loss that has been linked with metabolic declines in the frontal lobe of the brain (Thomas et al., 2000). Although there is not a large body of evidence, associations are also likely between sleep loss and increased risk taking (Roehrs et al., 2004).
Sleep Loss in Adolescents and Academic Performance
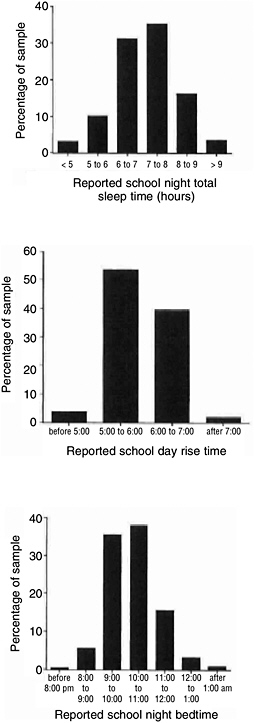
Sleep loss in adolescence is common and grows progressively worse over the course of adolescence, according to studies from numerous countries (Wolfson and Carskadon, 2003; Howell et al., 2004). Average sleep duration diminishes by 40 to 50 minutes from ages 13 to 19. Despite the physiological need for about 9 hours of sleep, sleep duration, across this age span, averages around 7 hours and about a quarter of high school and college students are sleep deprived (Wolfson and Carskadon, 1998). Research indicates that patterns of shortened sleep occur in the preadolescent period and may be most marked in African American boys, compared to white children or African American girls (Spilsbury et al., 2004). The decline in adolescent sleep duration is attributed to psychological and social changes, including growing desire for autonomy, increased academic demands, and growing social and recreational opportunities, all of which take place in spite of no change in rise time for school (Figure 4-2) (Wolfson and Carskadon, 1998). Furthermore, the need to earn income adds to the burden. Students who worked 20 or more hours weekly, compared with those who worked less than 20 hours, were found to go to bed later, sleep fewer hours, oversleep, and fall asleep more in class (Millman et al., 2005).
Sleep loss affects alertness, attention, and other cognitive functions in adolescents (Randazzo et al., 1998), but demonstrating a causal relationship between sleep loss and academic performance has been difficult. Most studies attempting to link the two are cross-sectional in design, based on
self-reporting of grades and sleep times, and lack a control for potential confounders (Wolfson and Carskadon, 2003). An association between short sleep duration and lower academic performance has been demonstrated (Wolfson and Carskadon, 1998; Drake et al., 2003; Shin et al., 2003), but the question of causality has not been resolved by longitudinal studies. A 3-year study of 2,200 middle school students did not find that sleep loss resulted in lower academic performance. It only found a cross-sectional association at the beginning of the study. However, by the end of the study, as sleep time worsened, grades did not proportionately decrease (Fredriksen et al., 2004). A study of the Minneapolis School District, which delayed start times for its high schools by almost 1.5 hours (from 7:15 a.m. to 8:40 a.m.), found significant improvements in sleep time, attendance, and fewer symptoms of depressed mood (Wahlstrom et al., 2001). Further, there was a trend toward better grades, but not of statistical significance. The study compared grades over the 3 years prior to the change with grades 3 years afterwards.
Much of the difficulty in studying sleep loss and its relation to academic performance stems from multiple, often unmeasured, environmental factors that affect sleep (such as school demands, student employment after school, family influences, TV viewing, and Internet access). These are set against the rapid developmental and physiological changes occurring in adolescence. Another difficulty is the challenge of objectively assessing school performance (Wolfson and Carskadon, 2003).
Additional robust intervention studies are needed to determine the effect of having later school start times on student performance. However, a confounder to later school start times is the potential onset of sleep phase delay during middle school (seventh and eighth grade). Moving middle school start time early to compensate for later high school start time may be problematic for the middle school children. There have been no studies that have examined effects of early start time on elementary-aged children (Wolfson and Carskadon, 2003). An alternative to changing the school starting times might be to implement bright light therapy in early morning classes for high school students as a means to change the circadian timing system of these students and thereby enable earlier sleep schedules (Wolfson and Carskadon, 2003).
Sleep Loss and Medical Errors
The Institute of Medicine’s report To Err Is Human estimated that as many as 98,000 deaths—due to medical errors—occur annually in United States hospitals (IOM, 2000). Long work hours and extended shifts among hospital workers are now known to contribute to the problem. Since the report’s release, several new studies, discussed below, have found strong
relationships between sleep loss, shift duration, and medical errors among medical residents.
Medical residents work longer hours than virtually all other occupational groups (Steinbrook, 2002). During the first year, medical residents frequently work a 24-hour shift every third night (i.e., 96-hours per week). Two studies found that sleep-deprived surgical residents commit up to twice the number of errors in a simulated laparoscopic surgery (Grantcharov et al., 2001; Eastridge et al., 2003). In a survey of 5,600 medical residents, conducted by the Accreditation Council for Graduate Medical Education, total work time was inversely correlated with reported sleep time. Residents who worked more than 80 hours per week were 50 percent more likely than those working less than 80 hours to report making a significant medical error that led to an adverse patient outcome (Baldwin and Daugherty, 2004). The strongest evidence tying medical errors to sleep-related fatigue from extended work hours comes from an intervention trial designed to limit residents’ work hours (Box 4-1). Earlier attempts to demonstrate patient safety benefits by reducing resident hours were beset by methodological problems (Fletcher et al., 2004).
Residents are not the only health professionals to report medical errors in association with short sleep. Nurses who completed logbooks recording their schedule length, sleep, and errors, reported 3.3 times more medical errors during 12.5 hour shifts than 8.5 hour shifts (Rogers et al., 2004). Nearly 40 percent of the nurses reported having 12-hour shifts; and although their sleep duration was not directly studied, the findings suggest that fatigue is a major factor.
Obstructive Sleep Apnea Is Associated with Development, Cognition, and Behavior in Children
Children with obstructive sleep apnea (OSA) often have problems in development, cognition, behavior, and academic performance, according to detailed reviews of the evidence (Schechter, 2002; Bass et al., 2004). The risk of neurobehavioral abnormalities in children with severe OSA is about three times greater than in children without OSA (Schechter, 2002). The contribution of overnight reduction of oxygen levels in the blood (hypoxemia) in comparison to sleep disruption is unclear. One study shows an association with the lowest level of oxygen during sleep and scores in arithmetic (Urschitz et al., 2005), but other studies show cognitive or behavioral deficits in children who snore without severe sleep apnea (Kennedy et al., 2004; Rosen et al., 2004; Gottlieb et al., 2004; O’Hara et al., 2005). Outcome measures used in numerous studies include intelligence quotient, learning and vocabulary, attention, symptoms of attention deficit hyperactivity disorder (ADHD), and academic performance. For example, two historical
cohort studies found decrements in intelligence quotient, impaired learning, and vocabulary in children with polysomnography-confirmed cases (Rhodes et al., 1995; Blunden et al., 2000). A study of younger children with sleep apnea also did not find a relationship with academic performance, after adjusting for the effects of socioeconomic status (Chervin et al., 2003). O’Brien and colleagues (2004) found that 35 children with sleep-disordered breathing, compared with matched controls, showed significant deficits in neurocognition, including overall cognitive ability, as well as attention and executive function, but the study did not find behavioral differences. A previous study by the same researchers found higher symptoms of ADHD, according to parents’ reports, in children with OSA (O’Brien et al., 2003). Several other studies have found greater symptoms of ADHD in children with OSA than controls (Weissbluth and Liu, 1983; Stradling et al., 1990; Chervin et al., 1997).
The neurobehavioral effects of OSA may be partially reversible with tonsillectomy and adenoidectomy, a surgical procedure that opens the airway. Treatment is related to partial improvement in school performance, cognition, or behavior (Ali et al., 1996; Friedman et al., 2003). A limitation to this work is that it is often difficult to control for the many confounders that influence cognitive function, with a recent study showing that after robustly adjusting for neighborhood socioeconomic status (Emancipator et al., 2006), effects were much attenuated, although they persisted in a subgroup of children who had been born prematurely. No randomized controlled study has been conducted to address the potential reversibility of cognitive deficits with sleep-disordered breathing; such data would more definitively address this situation. Gozal (1998) studied 54 children with sleep-disordered breathing and low school performance. Half of them underwent surgical tonsillectomy and adenoidectomy to treat OSA. Children undergoing the interventions improved their academic performance, compared to untreated children. One problem with the study design; however, was that surgical treatment was not randomly assigned (parents elected whether or not their children could receive surgery). Given the high proportion of children with sleep-disordered breathing, especially in vulnerable groups such as children in minority populations and those born prematurely, there is a large need to address the role of sleep-disordered breathing and its reversibility in these important outcomes.
Sleep-Disordered Breathing and Cognitive Impairment in Adults
Several cross-sectional studies indicate that sleep-disordered breathing in adults is associated with impaired cognitive function (Greenberg et al., 1987; Bedard et al., 1991; Naegele et al., 1995; Redline et al., 1997; Kim et al., 1997). Cognitive deficits, in turn, partially contribute to poorer work
performance (Ulfberg et al., 1996), accidents and injuries, and deterioration of the quality of life (see later sections).
A meta-analysis of the case-control studies found that the magnitude of the cognitive disturbance was greatest in individuals with severe OSA. Cognitive domains most affected were attention and executive function (the capacity to plan and organize complex tasks) with only milder effects on memory (Engleman et al., 2000). The meta-analysis also found some cognitive benefit associated with continuous positive airway pressure (CPAP) treatment. In a series of randomized, placebo-controlled crossover trials, people with mild OSA exhibited a trend toward better performance. The failure to detect a robust effect may have been due to the fact that the patients had mild disease, were nonadherent to therapy, or that they had a possibly irreversible component to the cognitive impairment. The cognitive deficits with sleep-disordered breathing are thought to be related to both sleep fragmentation and hypoxemia (Weaver and George, 2005). However, one study showed no clear threshold level between level of hypoxia and performance deficits (Adams et al., 2001). Animal models of chronic episodic hypoxia have led to the hypothesis that cognitive deficits in humans result from injury of nerve cells in the pre-frontal cortex (Beebe and Gozal, 2002), the area of the brain responsible for problem solving, emotion, and complex thought.
MOTOR VEHICLE CRASHES AND OTHER INJURIES
Motor Vehicle Crashes
Sleepiness is a significant, and possibly growing, contributor to serious motor vehicle injuries. Almost 20 percent of all serious car crash injuries in the general population are associated with driver sleepiness, independent of alcohol effects (Connor et al., 2002). Driver sleepiness is most frequently a manifestation of sleep loss, as discussed below, but other sleep disorders, which have lower prevalence, contribute to the problem, including sleep-disordered breathing, restless legs syndrome, and narcolepsy.
The 20 percent figure, cited above, is the population-attributable risk, which is a key public health measure indicating what percentage of car crash injuries, including fatal injuries of passengers, could be avoided by eliminating driver sleepiness. The finding was based on a population-based case-control study in a region of New Zealand in which 571 car drivers and a matched control sample were asked detailed questions about measures of acute sleepiness while driving (Connor et al., 2002). The study adjusted for potential confounding factors, including alcohol. Crashes examined in this study involved a hospitalization or death. The greatest risk factor for the crashes was sleep loss and time of day (driving between 2:00 a.m. to 5:00 a.m.), but sleep apnea symptoms were not risk factors.
Indications are that the public health burden of sleepiness-related injuries is likely increasing, given recent trends in drowsy driving. The National Sleep Foundation found that self-reported drowsy driving has increased significantly over the past years, from 51 percent of respondents in 2001 to 60 percent in 2005 (NSF, 2005). Similarly striking was that more than 10 percent of the entire sample reported nodding off or falling asleep while driving at least 1 to 2 days per month.
The impact of driver sleepiness is similar in magnitude to that of alcohol consumption. A study of all crashes between 1990 to 1992 reported to North Carolina’s uniform reporting system found that fall-asleep crashes (ones in which a law officer determines the driver to be asleep or fatigued) and alcohol-related crashes were similar in terms of serious injuries (13.5 and 17.8 percent of crashes from all causes, respectively) and fatalities (1.4 and 2.1 percent of all fatalities, respectively) (Pack et al., 1995). In actual driving performance on a closed course, sleep-deprived adults performed as poorly as did alcohol-challenged adults (Powell et al., 2001). After a night of total sleep deprivation, impairments in lane-keeping ability were similar to those found with blood alcohol content of 0.07 percent (Fairclough and Graham, 1999).
Fall-asleep crashes have distinct patterns by type, age, and time of day. According to the North Carolina study, fall-asleep crashes are largely off-the-road and at higher speeds (in excess of 50 mph) (Pack et al., 1995). Adolescents and young adults between the ages of 16 and 29 are the most likely to be involved in crashes caused by the driver falling asleep (Horne and Reyner, 1995; Pack et al., 1995). They account for about 50 percent of all crashes (Horne and Reyner, 1995; Pack et al., 1995). Fall-asleep crashes occur at two periods of day that coincide with circadian variation in sleepiness, in the early morning (2:00 a.m. to 8:00 a.m.) (Pack et al., 1995; Connor et al., 2002) and during the midafternoon (Horne and Reyner, 1995; Pack et al., 1995; Carskadon, 2004). The most common reasons behind fall-asleep crashes are working multiple jobs, night shift work, and sleep duration of less than 5 hours (Connor et al., 2002; Stutts et al., 2003).
Sleep apnea accounts for a small, but measurable percentage of motor vehicle crashes, primarily in drivers above the age of 25 (Sassani et al., 2004). Individuals with sleep apnea are at twice the risk of having a traffic accident as unaffected individuals (Teran-Santos et al., 1999)—the higher the apnea-hypopnea index, the higher the risk (Young et al., 1997a). Sleepy drivers tend to display reduced vigilance, slow reaction times, and loss of steering control. Steering impairment in OSA, sleep deprivation, and alcohol intoxication was compared in a controlled clinical trial. Untreated OSA and sleep deprivation were similar in producing progressive steering deterioration throughout the drive, whereas alcohol-impaired individuals steered equally throughout the drive (Hack et al., 2001). Occupational groups at
high risk of sleep-related crashes are night shift workers (Horne and Reyner, 1995; Ohayon et al., 2002; Drake et al., 2004), medical residents and house staff (Marcus and Loughlin, 1996; Barger et al., 2005), and commercial truck drivers (Walsh et al., 2005).
Commercial truck drivers have attracted the most study because of the prevalence, severity, and public health impact of crashes involving commercial trucks. There are an estimated 110,000 injuries and 5,000 fatalities each year in motor vehicle accidents involving commercial trucks (CNTS, 1996). The National Transportation Safety Board (NTSB) determined that fatigue (including sleepiness) was the probable cause of 57 percent of crashes leading to a truck driver’s death (NTSB, 1990a,b). Although this figure is not universally accepted, the definition of fatigue by the NTSB is equivalent to the term sleepiness or sleep-related fatigue used by sleep experts (i.e., fatigue that results in human performance failure) (Walsh et al., 2005). For each truck driver fatality, another three to four people are killed (NHTSA, 1994).
A congressionally mandated study of 80 long-haul truck drivers in the United States and Canada found that drivers had short sleep duration, averaging 5.2 hours in bed and 4.8 hours of sleep per day (Federal Motor Carrier Safety Administration, 1996). Sleep duration was verified electro-physiologically over the 5-day study. Further, commercial drivers have a high prevalence of sleep apnea (Stoohs et al., 1995). Recent studies have found that sleep apnea affects 8 to 15 percent of commercial drivers in the United States and Australia (Gurubhagavatula et al., 2004; Howard et al., 2004).
Work-Related Injuries
Sleep-related fatigue is an independent risk factor in work-related injuries and fatalities, according to two large and well-designed studies (Akerstedt et al., 2002; Swaen et al., 2003). Swaen and coworkers prospectively studied a cohort of more than 7,000 workers in numerous industries in the Netherlands over a 1-year period before studying the occurrence of occupational accidents. During the year they collected information about sleep patterns and other potential risk factors for work-related injuries. The 108 employees who reported being injured during the next year could be assessed for risk factors without recall bias affecting the results. The study found a dose-response relationship between two sleep-related fatigue measures and injuries. For example, highly fatigued workers were 70 percent more likely to be involved in accidents than were workers reporting low fatigue levels, after adjustment for other risk factors. Workers with chronic insomnia were also far more likely than those who were good sleepers to report industrial accidents or injuries (Leger et al., 2002). Finally, disturbed
sleep plays a role in occupational fatalities. In a large 20-year prospective study in Sweden of nearly 50,000 individuals, those reporting disturbed sleep were nearly twice as likely to die in a work-related accident (OR = 1.89, 95% CI 1.22–2.94) (Akerstedt et al., 2002). Similarly, workers who report snoring and excessive daytime sleepiness, indications of sleep apnea, are twice as likely to be involved in workplace accidents, as verified by registry data and after adjusting for all potential confounders (Lindberg et al., 2001).
Falls in Older People
Falls are a common and costly problem in older people (65 years and older), whether in the community or in long-term care facilities. Each year, more than 30 percent of older people fall (Hausdorff et al., 2001). Falls are the leading cause of death for this particular age group (Murphy, 2000). Although most falls are not directly fatal, they are a leading cause of injuries and trauma-related hospital admissions (Alexander et al., 1992).
Insomnia increases the risk of falling (Brassington et al., 2000). One of the major questions raised by this finding is what is responsible for the increased risk of falls—the underlying insomnia or the use of medication to treat it? Until recently, most of the studies addressing this question were not large enough to yield an answer. In 2005, a large, prospective study of 34,000 nursing home residents across the state of Michigan ruled out use of hypnotic medications as a risk factor for falls (Avidan et al., 2005). In fact, the study found that treated insomnia, and untreated insomnia, but not hypnotic medications, were predictors of falls. Although the results of this study did not find that insomnia increased the risk of hip fractures, other studies have found an association (Fitzpatrick et al., 2001). Preliminary data from the Study of Osteoporosis in Women also indicate an increased risk of falls associated with decreased sleep efficiency and sleep time (as measured objectively using actigraphy) in a large group of older women, with effects persisting after adjustment of health status and mood and other confounders (Stone et al., 2004).
Interventions
There have been a few studies that have examined the effect of interventions on improving the outcomes associated with sleepiness. A range of regulatory, technological, and therapeutic approaches are possible to ameliorate the problem of sleepiness among commercial drivers (Walsh et al., 2005). However, there has been limited study of the benefit of these strategies. Thus, before additional rules and regulations are developed, analysis of the effec-
tiveness of the current regulations and statutory items is needed. This analysis will help the establishment of much-needed future rules and regulations pertaining to sleep loss and fatigue. Preplanned naps have been successfully tested in crew members on transmeridian flights; the findings show that safe and feasible rotations occurred as crew members took brief, 40-minute nap periods, and the naps improved alertness (Graeber et al., 1986a,b). Similarly, a study of Italian policemen who patrol highways found that prophylactic naps before a night shift can lower the risk of motor vehicle accidents during the shift, according to a combination of retrospective questionnaire, prospective analysis, and mathematical modeling (Garbarino et al., 2004)
The strongest evidence for the public health benefits of treatment comes from clinical trials and retrospective studies of the impact of CPAP therapy for sleep apnea. These studies also dispel any doubt of the causal relationship between sleep disorders and accidents. In a randomized, controlled clinical trial, 59 men with sleep apnea were assigned either to therapeutic CPAP or subtherapeutic CPAP. The latter does not deliver enough pressure to open the pharynx and achieve a therapeutic effect. One month later the men were placed in a steering simulator. Therapeutic CPAP significantly improved their steering performance and reaction time relative to sub-therapeutic CPAP (Hack et al., 2000). Previous clinical trials had also shown CPAP to be effective in terms of reducing the rate of self-reported automobile crashes and performance on driving tests, but they were uncontrolled (Cassel et al., 1996; Krieger et al., 1997). A review and meta-analysis estimates that nearly 1,000 lives would be saved annually if all drivers with OSA were treated with CPAP (Sassani et al., 2004).
IMPACT ON FUNCTIONING AND QUALITY OF LIFE
Sleep problems, difficulty initiating and maintaining sleep, nonrestorative sleep, and excessive daytime sleepiness are associated with adverse effects on well-being, functioning, and quality of life, according to numerous studies covering the general population (Baldwin et al., 2001, 2004; Hasler et al., 2005; Strine and Chapman, 2005), working people (Kuppermann et al., 1995), and clinical populations (Simon and VonKorff, 1997), including pediatric samples (Rosen et al., 2002). Studies have used various measures of quality of life and functional status, the most common of which is a validated questionnaire known as the SF-36, a 36-item measure that asks about eight domains: (1) physical functioning; (2) role limitation due to physical health problems (role physical); (3) bodily pain; (4) general health perceptions; (5) vitality; (6) social functioning; (7) role limitations due to emotional health problems (role emotional); and (8) mental health. A similar measure is the health-related quality of life survey, which asks fewer questions. Individuals who suffer from primarily sleep apnea, narcolepsy,
restless legs, primary parasomnias, and insomnia constantly report poorer quality of life compared to population norms (Reimer and Flemons, 2003).
Using health-related measures of quality of life, the functional impact of sleep loss was assessed by a large and nationally representative survey, the United States Behavioral Risk Factor Surveillance System (Strine and Chapman, 2005). The study focused on nocturnal sleep time in nearly 80,000 respondents. About 26 percent of the respondents reported obtaining insufficient sleep on a frequent basis (not enough sleep on 14 days or more over the past 30 days). This group was significantly more likely than those without frequent sleep insufficiency to report poorer functioning and quality of life on each of the eight items of the health-related quality of life.
Several studies have dealt with insomnia and its adverse impact on quality of life (Zammit et al., 1999; Leger et al., 2001; Katz and McHorney, 2002). People with severe insomnia reported lower quality of life on all eight domains of the SF-36 (Leger et al., 2001). Their low quality-of-life ratings were similar to ratings by patients with congestive heart failure and depression, according to a study of nearly 3,500 primary care patients (Katz and McHorney, 2002) (Figure 4-3). About 16 percent of the sample had severe insomnia, and the study adjusted for numerous factors including health habits, obesity, other chronic conditions, and severity of disease. A study of a large health maintenance organization population (n = 2,000) found that insomnia (versus no current insomnia) was associated with significantly greater impairment, as measured by the self-rated Social Disability Schedule and the interviewer-rated Brief Disability Questionnaire. Individuals with insomnia also had more days of restricted activity due to illness
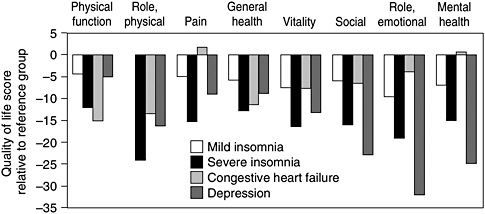
FIGURE 4-3 Severe insomnia affects quality of life.
SOURCE: Edinger and Means (2005).
and more days spent in bed (Simon and VonKorff, 1997). One study revealed a dose-response relationship, with higher levels of insomnia being associated with greater impairments in the ability to accomplish daily tasks and decreased enjoyment of interpersonal relationships (Roth and Ancoli-Israel, 1999).
Individuals with severe OSA also report significantly poorer quality of life, and mild OSA is also associated with reduced vitality (Baldwin et al., 2001). These effects are similar to those of other chronic diseases in the general population in the United States. Individuals with OSA who are compliant with CPAP treatment report improved changes in vitality and quality of life 2 months after the onset of CPAP treatment (Redline et al., 1998).
Symptoms of restless legs syndrome are associated with lower quality of life (Unruh et al., 2004), similar to the quality of life of individuals with type 2 diabetes mellitus and acute heart attack (Allen et al., 2003). Restless legs syndrome also affects marital relationships. Approximately one-third of couples sleep in separate beds due to the discomfort of their partner’s repetitive leg movements (Montplaisir et al., 2005).
Approximately a quarter of children and adolescents report difficulty with sleep (Stein et al., 2001; Archbold et al., 2002). However, very few studies have assessed the association between sleep loss and sleep disorders and health-related quality of life in children. Analysis of a widely used parent report measure of children’s physical, emotional, and social functional status and well being—the CHQ-PF50—found the quality of life of their children deteriorated with the severity of OSA (Rosen et al., 2002). This is consistent with a negative association between sleep difficulties and health-related quality of life that was observed a similar analysis of 80 parents of children referred to a pediatric sleep disorders clinic (Hart et al., 2005). Thus, sleep difficulties may broadly affect a child’s development through its impact on children’s social, emotional, and physical functioning.
Family and Community Function
The consequences of sleep loss and sleep disorders are not restricted to affected individuals; they also disrupt families and communities. Although relatively sparse, the research described in this section points to widespread impact on the health and well-being of sleep partners and/or other family members. Their sleep quality and health can be disrupted, as can their wellbeing, income, and capacity to care for children or ill family members. Adverse effects on family cohesiveness, in turn, can lead to severe family turmoil and divorce. Similarly, sleep disruption of family caregivers has broader societal effects by contributing to hospitalization or nursing home placement of ill family members for whom they provide care.
Most research on families and communities deals with bed partners of individuals with a sleep problem. Bed partners of individuals with sleep-disordered breathing report a lower quality of life, based on the SF-36 survey than the sleep-disordered breathing patients (Breugelmans et al., 2004). Further, in a large population-based sample of older individuals, bed partners report poor health, depressed mood, poor mental health, and marital unhappiness (Strawbridge et al., 2004).
Does CPAP therapy improve bed partners’ sleep? At least four studies have addressed this question, with three showing improvement. Two of the studies that demonstrated a benefit were nonrandomized and used a before versus after study design. After approximately one month of CPAP therapy, partners experienced less daytime sleepiness as measured by the Epworth Sleepiness Scale and improved quality of life as measured by the SF-36 scale (Doherty et al., 2003; Parish and Lyng, 2003). A small, polysomnographic study of individuals using CPAP found that their partners show fewer arous-als and greater sleep efficiency in the hours after CPAP’s introduction versus the hours before (Beninati et al., 1999). The improvement in sleep efficiency (percentage of time asleep while in bed) translated to an extra hour of sleep per night. The only placebo-controlled study found that CPAP is associated with subjective improvement in bed partners’ sleep (via the Pittsburgh Sleep Quality Inventory), but no objective improvement, as measured by polysomnography (McArdle et al., 2000).
Sleep-disordered breathing has also been found to heighten the rate of divorce and the use of paid personal leave, among other effects, according to a study of obese individuals with OSA. A team of Swedish researchers, studying a large registry of obese subjects, found that individuals with OSA (as defined by symptoms of snoring and daytime sleepiness) report about three times the rate of divorce of those without OSA and/or daytime sleepiness (Grunstein et al., 1995). The effects are even more pronounced among the women in the sample with OSA (n = 155). Men with OSA (n = 338) reported less income, and both genders reported more sick leave and disturbed work performance. These effects were independent of the effects of obesity and other health factors. In a separate study, 60 percent of bed partners reported that they slept apart versus 20 percent of controls. Although the partners’ level of marital satisfaction was similar to controls’, the partners reported greater dissatisfaction with the sleep behaviors of their apneic spouses (Billmann and Ware, 2002).
A common complaint of parents is being awakened by a young child with a sleep problem. Sleep loss is indeed reported more frequently by parents after the birth of a child than during pregnancy (Gay et al., 2004). Improvement in parents’ sleep quality, as well as improvement of family well-being, occurs after the introduction of a behavioral intervention designed to train parents to overcome sleep problems in young children
through a graduated conditioning program known as extinction (Eckerberg, 2004). Previously, controlled clinical trials had shown that parent training and extinction are effective for treating young children (Mindell, 1999; Ramchandani et al., 2000), but the trials had not measured the impact on sleep and well-being of parents and families.
Sleep disturbances in chronic illness, whether in the affected individual or in the caregiver, affect decisions about hospital or nursing home placement. This is especially true for patients with Alzheimer’s disease, considering that up to 44 percent of them have sleep disturbances (Ritchie, 1996; McCurry et al., 1999). Indeed, sleep disturbance in Alzheimer’s disease is a common risk factor for nursing home placement (Chenier, 1997; Hope et al., 1998). Sleep hygiene training, targeted at both Alzheimer’s disease patients and the caregivers, can improve sleep quality in patients (McCurry et al., 1998, 2003, 2005). One area of future study is whether treating sleep problems (in either the patient or the caregiver) can delay institutionalization. Counseling of caregivers—although not explicitly targeted to their sleep disturbance or that of the patient—has been shown, in a separate randomized trial, to delay nursing home placement (Mittelman et al., 1996). Within nursing homes, behavioral and pharmacological therapies are effective at improving sleep problems (Alessi et al., 1999; Naylor et al., 2000).
ECONOMIC IMPACT OF SLEEP LOSS AND SLEEP DISORDERS
Although problems falling asleep or daytime sleepiness affect 35 to 40 percent of the population (Hossain and Shapiro, 2002), the full economic impact of sleep loss and sleep disorders on individuals and society is not known. There are limited data on the economic impact of insomnia, sleep-disordered breathing, and narcolepsy; the economic impact of other sleep disorders has not been analyzed. As will be discussed in further detail in Chapters 5 and 8, the lack of sufficient data result from inadequate reporting and surveillance mechanisms.
Increased Health Care Utilization
Daytime sleepiness, inadequate sleep time, insomnia, and other sleep disorders place a significant burden on the health care system through increased utilization of the health care system (see below). Patients in the highest quartile of the Epworth Sleepiness Scale are associated with an 11 percent increase in health care utilization, and individuals with sleep-disordered breathing or sleepiness and fatigue are associated with a 10 to 20 percent increase in utilization (Kapur et al., 2002b).
Insomnia
Individuals suffering from insomnia place an increased burden on the health care system (Ohayon and Roth, 2003). Their activity is more limited (Simon and VonKorff, 1997), and they are significantly more likely to access medical and psychiatric care than are individuals that do not have a sleep or psychiatric disorder (Weissman et al., 1997). Individuals with insomnia who also have an associated psychiatric disorder are more likely to seek treatment for emotional problems (14.9 percent versus 8 percent) (Weissman et al., 1997), have a greater number of physician visits, and be admitted to a hospital twice as often (Leger et al., 2002). The burden insomnia place on the health care system is long-term—the majority of individuals with either mild (59 percent) or severe (83 percent) insomnia continue to suffer symptoms of insomnia 2 years after initial diagnosis (Katz and McHorney, 1998). Consequently, individuals suffering from insomnia place a significant economic burden on society resulting in increased health care costs (see below).
Obstructive Sleep Apnea
Individuals with OSA also place a significant burden on the health care system. In the year prior to diagnosis, the medical expenses of individuals with OSA were almost two times as much as control individuals not diagnosed with OSA ($2,720 vs. $1,384) (Kapur et al., 1999). Around 80 to 90 percent of OSA cases remain undiagnosed and untreated, which increases the burden of this disorder (Young et al., 1997b; Kapur et al., 2002a). Analysis of health care utilization in Canadians with severe OSA found that during the year prior to diagnosis, individuals with severe OSA spent more than twice the number of days in the hospital compared to controls (251 versus 90). This was associated with an increase in cost of services—$49,000 to $99,000 (Kryger et al., 1996). This figure is likely greater in the United States, which also has 10 to 15 percent higher health care utilization associated with severe OSA (Kapur et al., 2002b)—due to higher health care costs compared to Canada. OSA also affects a child’s health care utilization. A survey of 287 children with OSA found that in the year prior to diagnosis, children with OSA had a 226 percent increase in health care utilization and had significantly more visits to emergency departments (Reuveni et al., 2002).
A retrospective observational cohort study demonstrated that CPAP treatment reversed the trend of increasing health care utilization observed prior to OSA diagnosis (Bahammam et al., 1999; Albarrak et al., 2005). In a Canadian study, physician visits decreased during the 5 years following CPAP treatment, compared to the 5-year period prior to diagnosis, resulting in lower physician fees (Figure 4-4). After converting to American dol-
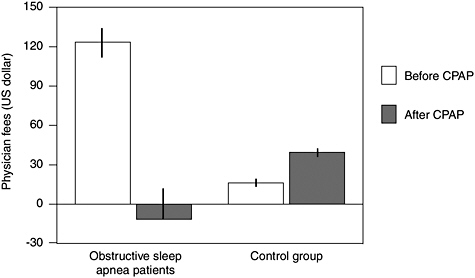
FIGURE 4-4 Effect of CPAP treatment on physician fees.
SOURCE: Albarrak et al. (2005).
lars, mean total fees were greater the year prior to OSA diagnosis ($179.09 ± $32.85) compared to the fifth year after diagnosis ($16.77 ± $33.66) in individuals who were compliant with CPAP treatment (Albarrak et al., 2005). The realized savings would likely be much larger in the United States due to higher associated health care costs.
Direct Costs of Sleep Loss and Sleep Disorders
Billions of dollars are spent each year in the United States on the direct costs of sleep loss and sleep disorders. These medical costs include expenses associated with doctor visits, hospital services, prescriptions and over-the-counter medications. In 1995 the direct cost of insomnia in the United States was estimated to be $13.9 billion (Walsh and Engelhardt, 1999). Further, based on the costs associated with a laboratory-based polysomnogram, it would cost over $17.5 billion to test and $3 billion to treat every person in the United States who has sleep apnea1 (Sassani et al., 2004). Although it is predicted that the advent of more effective portable monitoring devices (Chapter 6) will decrease the costs associated with testing and diagnosis of sleep disorders, the total direct costs will still remain high and be a burden.
Indirect Costs of Sleep Loss and Sleep Disorders
The indirect costs associated with sleep loss and sleep disorders also result in billions of dollars of annual expenditures, including costs associated with illness-related morbidity and mortality, absenteeism, disability, reduction or loss of productivity, industrial and motor vehicle accidents, hospitalization, and increased alcohol consumption (Hossain and Shapiro, 2002). As is the case with direct costs, for each of these categories further analysis is required to determine the complete indirect costs of sleep loss and sleep disorders. The annual economic impact of sleep problems relating to workers inability to adjust to late shifts are estimated to be at minimum over $60 billion (Table 4-1) (Moore-Ede, 1993). In addition, it has been estimated that sleep-related fatigue costs businesses $150 billion a year in absenteeism, workplace accidents, and other lost productivity (Sleep Disorders Create Growing Opportunities for Hospitals, 2001).
A 1994 analysis of automobile accidents estimated the cost of accidents attributed to sleepiness to be between $29.2 to $37.9 billion (Leger, 1994). Over 50 percent of automobile crashes involving a truck, where a fatality occurred, were caused by sleep-related fatigue, costing approximately $2.7 million and 4,800 lives (NTSB, 1990a, 1990b; USDOT, 1991; Mitler et al., 2000). However, there is no standardized mechanism to record fatigue- and sleep-related accidents; therefore, these figures are likely underestimates of the total cost of automobile accidents.
Although the complete economic impact of sleep disorders and sleep loss is limited, the available data demonstrates the high burden that inadequate sleep has on the economy. With the average age of the population rising, incidence of sleep disorders is likely to rise, leading to increased costs (Phillips, 2005).
TABLE 4-1 Annual Economic Impact of Sleep Problems Due to Late Shifts
|
|
Cost (billions $) |
|
Reduced manufacturing productivity |
50.0 |
|
Increased motor vehicle accidents |
5.7 |
|
Increased industrial accidents |
4.0 |
|
Increased accidents, injuries, and deaths at work |
2.5 |
|
Increase in other medical and psychiatric illnesses |
2.0 |
|
Personnel turnover and retraining |
1.0 |
|
Total |
65.2 |
|
SOURCE: Moore-Ede (1993). |
|
Economic Impact of Insomnia
The total cost estimates of insomnia range from $30 billion (Walsh and Engelhardt, 1999) to $107.5 billion (Stoller, 1994). The large variation in the range is attributed to the underlying assumptions about the prevalence of insomnia in the United States, which range from 10 to 33 percent. However, it is evident that even using more conservative prevalence estimates the total annual costs in the United States exceeds tens of billions of dollars.
Direct Cost
In 1995 the direct costs of insomnia totaled approximately $13.96 billion (Table 4-2). However, this is an underestimate of the total costs, as approximately 12 percent of all physicians, including hospital-based or government employed physicians (including doctors in VA hospitals), were not included in these estimates. In 2002 it was estimated that in the United States 27 million prescriptions were filled for hypnotics, worth about $1.2 billion (Mendelson, 2005). Calculations based on medical claims showed that increased physician fees and medical expenses for elderly and non-elderly patients with insomnia were respectively $5,580 and $4,220 higher
TABLE 4-2 The Direct Costs of Insomnia in the United States for 1995
|
|
Costs (millions $) |
|
Substances used for insomnia |
|
|
Prescription medications |
809.92 |
|
Nonprescription medications |
325.80 |
|
Alcohol |
780.39 |
|
Melatonin |
50.00 |
|
Total Cost of Substances |
1,966.11 |
|
Health care services for insomnia |
|
|
Outpatient physician visits |
660.00 |
|
Psychologist visits |
122.40 |
|
Social working visits |
75.30 |
|
Sleep specialist visits |
18.20 |
|
Mental health organizations |
153.00 |
|
In-patient hospital care |
30.80 |
|
Nursing home care |
10,900.00 |
|
Total |
11,960.70 |
|
Total direct costs |
13,926.11 |
|
SOURCE: Walsh and Engelhardt (1999). |
|
than match controls (Ozminkowski et al., 2004), demonstrating the expense incurred by individuals with insomnia.
In 1995 over 78 percent of direct costs associated with insomnia, $11.96 billion, was spent on nursing home care (Walsh and Engelhardt, 1999), a 132 percent increase since 1990 (Walsh et al., 1995). Although this proportion may seem high, almost half of the population over 65 years of age report difficulty with sleep (Mellinger et al., 1985), and 20.4 percent of admissions to a nursing home were attributed to sleep disturbances (Walsh and Engelhardt, 1999).
Two factors contribute to these higher costs associated with insomnia. First, the general population is typically reluctant to consult doctors about their sleep problems, and second, inadequate physician training prevents proper recognition, diagnosis, and treatment of patients with insomnia (see Chapters 5 and 7) (Walsh and Engelhardt, 1999; Benca, 2005; NIH, 2005).
Indirect Cost
To date there has not been a detailed analysis assessing the total indirect costs associated with insomnia. A 1988 study estimated that productivity loss resulting from insomnia cost $41.1 billion (Stoller, 1994). Absenteeism cost more than $57 billion (Walsh, 2004). Therefore, once the costs of industrial and motor vehicle collisions and related morbidities are included, the indirect cost of insomnia could top $100 billion.
Insomnia places a greater burden on individuals of lower socioeconomic status (Gellis et al., 2005), those who are less educated, and those who are more likely to be unemployed (Bixler et al., 1979; Karacan et al., 1983; Frisoni et al., 1993; Kim et al., 2000; Li et al., 2002). Falls caused by insomnia also contribute to its economic burden. A greater risk for falls was associated with both hypnotic use (29 percent, OR = 1.29) and insomnia (90 percent, OR = 1.90) (Avidan, 2005). Like other sleep disorders, insomnia is more prevalent in the elderly (Mellinger et al., 1985); therefore, as the United States population continues to age it is expected that the cost associated with falls caused by insomnia will also continue to rise.
Economic Impact of Obstructive Sleep Apnea
Direct Cost
Similar to other sleep disorders, there is very limited data on the direct costs associated with obstructive sleep apnea. Most of the analysis has explored the impact of OSA. The average costs of diagnosis and treatment over five years for an individual is over $4,000 (Table 4-3) (Chervin et al., 1999; Wittmann and Rodenstein, 2004). An analysis of 97 obese individuals with
TABLE 4-3 Cost of Diagnosis and Treatment of OSA
|
Polysomnogram |
$1,190 |
|
CPAP titration |
$1,190 |
|
CPAP equipment and setup |
$1,290 |
|
Initial office visits |
$210 |
|
Annual follow-up |
$330 |
|
Total |
$4,210 |
|
SOURCE: Chervin et al. (1999). |
|
OSA in Canada found that over a 2-year period they had almost $30,000 in expenditures from physician claims and utilized $49,000 to $99,000 more in services than their control counterparts (Kryger et al., 1996).
Indirect Cost
There is also limited analysis of the total indirect costs associated with OSA. Based on estimates of from the Sleep Heart Health Study, only 10 to 20 percent of individuals with OSA are estimated to have been diagnosed (Kapur et al., 2002a). The annual medical costs resulting from untreated OSA was $3.4 billion (Kapur et al., 1999).
Automobile collisions attributed to OSA also contribute to the large economic burden of the syndrome. Sassani and colleagues performed a meta-analysis of PubMed from 1980 to 2003 and investigated the relationship between collisions and OSA (2004). This information was then combined with data from the National Safety Council to estimate OSA-related collisions, costs, and fatalities. Based on this analysis, it was estimated that in the year 2000 more than 800,000 drivers were involved in OSA-related motor vehicle collisions (Sassani et al., 2004). These collisions resulted in loss of life to 1,400 individuals and cost $15.9 billion. The authors calculated that annually it would cost $3.18 billion to provide CPAP treatment to all drivers who suffer from OSA, saving 980 lives and $11.1 billion to $7.9 billion if the cost of CPAP treatment is taken into account. For every dollar spent on CPAP, $3.49 would be saved in reduced collision costs. This savings does not include the presumed reduction in the number of accidents at work, decreased health care costs, or improved quality of life (Sassani et al., 2004).
Relationship Between Socioeconomic Status, Race, and Obstructive Sleep Apnea
The relationship between socioeconomic status, race, and obstructive sleep apnea is not well understood. There are limited data that suggest that
the prevalence and severity of OSA is higher in African Americans compared to whites (Scharf et al., 2004), especially in adults under 25 years of age (Redline et al., 1994; Rosen et al., 2002). Compared to whites, African Americans with OSA are more likely to have a higher body mass index (Redline et al., 1994) and a lower mean income (Scharf et al., 2004). Analysis performed between Asians and whites found that OSA in Asians was significantly more severe compared to whites (Ong and Clerk, 1998). However, differences in age, gender, body mass index, or neck circumference did not account for these differences.
Economic Impact of Narcolepsy
The impact of narcolepsy on the economy is also not well understood. A review of the PubMed database through May of 2005 found only one relevant report. It examined narcolepsy’s effect on 75 individuals in Germany (Dodel et al., 2004). After converting to American dollars the annual total costs to an individual were $15,410. The average direct costs accounted for 21 percent of the total expenditures ($3,310 total), $1,260 for hospital care, and $1,060 for medications. However, these figures have been extrapolated from a single German cohort and differences in the organization of their respective health care systems have not been taken into account. Therefore, improved surveillance data are needed to determine the actual economic impact of narcolepsy on the American population.
The socioeconomic status of an individual does not affect the prevalence and severity of narcolepsy; however, narcolepsy may worsen an individual’s socioeconomic standing. In Germany individuals with narcolepsy have a significantly higher unemployment rate than average, 59 percent compared to the national average of 9 percent (Dodel et al., 2004). Similarly, studies performed in the United Kingdom (Daniels et al., 2001) and the United States (Goswami, 1998) found that 30 to 37 percent of respondents had lost their job due to narcolepsy.
Summary
Although the data are limited, the effect of sleep disorders, chronic sleep loss, and sleepiness on accident rates, performance deficits, and health care utilization on the American economy is significant. The high estimated costs to society of leaving the most prevalent sleep disorders untreated are far more than the costs that would be incurred by delivering adequate treatment. Hundreds of billions of dollars are spent and/or lost annually as a result of poor or limited sleep. However, greater surveillance and analysis are required to estimate the full economic implications of these problems.
REFERENCES
ACGME (Accreditation Council for Graduate Medical Education). 2003. Common Program Requirements (Resident Duty Hours). [Online]. Available: http://www.acgme.org/dutyhours/dutyhourscommonpr.pdf [accessed May 13, 2005].
Adams N, Strauss M, Schluchter M, Redline S. 2001. Relation of measures of sleep-disordered breathing to neuropsychological functioning. American Journal of Respiratory and Critical Care Medicine 163(7):1626–1631.
Akerstedt T, Fredlund P, Gillberg M, Jansson B. 2002. A prospective study of fatal occupational accidents–relationship to sleeping difficulties and occupational factors. Journal of Sleep Research 11(1):69–71.
Albarrak M, Banno K, Sabbagh AA, Delaive K, Walld R, Manfreda J, Kryger MH. 2005. Utilization of healthcare resources in obstructive sleep apnea syndrome: A 5-year follow-up study in men using CPAP. Sleep 28(10):1306–1311.
Alessi CA, Yoon EJ, Schnelle JF, Al-Samarrai NR, Cruise PA. 1999. A randomized trial of a combined physical activity and environmental intervention in nursing home residents: Do sleep and agitation improve? Journal of the American Geriatrics Society 47(7):784–791.
Alexander BH, Rivara FP, Wolf ME. 1992. The cost and frequency of hospitalization for fall-related injuries in older adults. American Journal of Public Health 82(7):1020–1023.
Ali NJ, Pitson D, Stradling JR. 1996. Sleep-disordered breathing: Effects of adenotonsillectomy on behaviour and psychological functioning. European Journal of Pediatrics 155(1): 56–62.
Allen RP, Picchietti D, Hening WA, Trenkwalder C, Walters AS, Montplaisir J, et al. 2003. Restless legs syndrome: Diagnostic criteria, special considerations, and epidemiology. A report from the restless legs syndrome diagnosis and epidemiology workshop at the National Institutes of Health. Sleep Medicine 4(2):101–119.
Archbold KH, Pituch KJ, Panahi P, Chervin RD. 2002. Symptoms of sleep disturbances among children at two general pediatric clinics. Journal of Pediatrics 140(1):97–102.
Avidan AY. 2005. Sleep in the geriatric patient population. Seminars in Neurology 25(1):52–63.
Avidan AY, Fries BE, James ML, Szafara KL, Wright GT, Chervin RD. 2005. Insomnia and hypnotic use, recorded in the minimum data set, as predictors of falls and hip fractures in Michigan nursing homes. Journal of the American Geriatrics Society 53(6):955–962.
Bahammam A, Delaive K, Ronald J, Manfreda J, Roos L, Kryger MH. 1999. Health care utilization in males with obstructive sleep apnea syndrome two years after diagnosis and treatment. Sleep 22(6):740–747.
Baldwin CM, Griffith KA, Nieto FJ, O’Connor GT, Walsleben JA, Redline S. 2001. The association of sleep-disordered breathing and sleep symptoms with quality of life in the Sleep Heart Health Study. Sleep 24(1):96–105.
Baldwin DC Jr, Daugherty SR. 2004. Sleep deprivation and fatigue in residency training: Results of a national survey of first- and second-year residents. Sleep 27(2):217–223.
Barger LK, Cade BE, Ayas NT, Cronin JW, Rosner B, Speizer FE, Czeisler CA, Harvard Work Hours HaS Group. 2005. Extended work shifts and the risk of motor vehicle crashes among interns. New England Journal of Medicine 352(2):125–134.
Bass JL, Corwin M, Gozal D, Moore C, Nishida H, Parker S, Schonwald A, Wilker RE, Stehle S, Kinane TB. 2004. The effect of chronic or intermittent hypoxia on cognition in childhood: A review of the evidence. Pediatrics 114(3):805–816.
Bedard MA, Montplaisir J, Richer F, Rouleau I, Malo J. 1991. Obstructive sleep apnea syndrome: Pathogenesis of neuropsychological deficits. Journal of Clinical and Experimental Neuropsychology 13(6):950–964.
Beebe DW, Gozal D. 2002. Obstructive sleep apnea and the prefrontal cortex: Towards a comprehensive model linking nocturnal upper airway obstruction to daytime cognitive and behavioral deficits. Journal of Sleep Research 11(1):1–16.
Belenky G, Wesensten NJ, Thorne DR, Thomas ML, Sing HC, Redmond DP, Russo MB, Balkin TJ. 2003. Patterns of performance degradation and restoration during sleep restriction and subsequent recovery: A sleep dose-response study. Journal of Sleep Research 12(1):1–12.
Benca RM. 2005. Diagnosis and treatment of chronic insomnia: A review. Psychiatry Services 56(3):332–343.
Beninati W, Harris CD, Herold DL, Shepard JW Jr. 1999. The effect of snoring and obstructive sleep apnea on the sleep quality of bed partners. Mayo Clinic Proceedings 74(10): 955–958.
Billmann SJ, Ware JC. 2002. Marital satisfaction of wives of untreated sleep apneic men. Sleep Medicine 3(1):55–59.
Bixler EO, Kales A, Soldatos CR, Kales JD, Healey S. 1979. Prevalence of sleep disorders in the Los Angeles metropolitan area. American Journal of Psychiatry 136(10):1257–1262.
Blunden S, Lushington K, Kennedy D, Martin J, Dawson D. 2000. Behavior and neurocognitive performance in children aged 5-10 years who snore compared to controls. Journal of Clinical and Experimental Neuropsychology 22(5):554–568.
Brassington GS, King AC, Bliwise DL. 2000. Sleep problems as a risk factor for falls in a sample of community-dwelling adults aged 64-99 years. Journal of the American Geriatrics Society 48(10):1234–1240.
Breugelmans JG, Ford DE, Smith PL, Punjabi NM. 2004. Differences in patient and bed partner-assessed quality of life in sleep–disordered breathing. American Journal of Respiratory and Critical Care Medicine 170(5):547–552.
Carskadon MA. 2004. Sleep deprivation: Health consequences and societal impact. Medical Clinics of North America 88(3):767–776.
Cassel W, Ploch T, Becker C, Dugnus D, Peter JH, von Wichert P. 1996. Risk of traffic accidents in patients with sleep-disordered breathing: Reduction with nasal CPAP. European Respiratory Journal 9(12):2606–2611.
Chenier MC. 1997. Review and analysis of caregiver burden and nursing home placement. Geriatric Nursing (London) 18(3):121–126.
Chervin RD, Dillon JE, Bassetti C, Ganoczy DA, Pituch KJ. 1997. Symptoms of sleep disorders, inattention, and hyperactivity in children. Sleep 20(12):1185–1192.
Chervin RD, Murman DL, Malow BA, Totten V. 1999. Cost-utility of three approaches to the diagnosis of sleep apnea: Polysomnography, home testing, and empirical therapy. Annals of Internal Medicine 130(6):496–505.
Chervin RD, Clarke DF, Huffman JL, Szymanski E, Ruzicka DL, Miller V, Nettles AL, Sowers MR, Giordani BJ. 2003. School performance, race, and other correlates of sleep-disordered breathing in children. Sleep Medicine 4(1):21–27.
CNTS (Center for National Truck Statistics). 1996. Truck and Bus Accident Factbook—1994. UMTRI-96-40. Washington, DC: Federal Highway Administration Office of Motor Carriers.
Connor J, Norton R, Ameratunga S, Robinson E, Civil I, Dunn R, Bailey J, Jackson R. 2002. Driver sleepiness and risk of serious injury to car occupants: Population-based case control study. British Medical Journal 324(7346):1125.
Daniels E, King MA, Smith IE, Shneerson JM. 2001. Health-related quality of life in narcolepsy. Journal of Sleep Research 10(1):75–81.
Dinges DF, Graeber RC, Carskadon MA, Czeisler CA, Dement WC. 1989. Attending to inattention. Science 245(4916):342.
Dinges D, Rogers N, Baynard MD. 2005. Chronic sleep deprivation. In: Kryger MH, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 4th ed. Philadelphia: Elsevier/ Saunders. Pp. 67–76.
Dodel R, Peter H, Walbert T, Spottke A, Noelker C, Berger K, Siebert U, Oertel WH, Kesper K, Becker HF, Mayer G. 2004. The socioeconomic impact of narcolepsy. Sleep 27(6):1123–1128.
Doherty LS, Kiely JL, Lawless G, McNicholas WT. 2003. Impact of nasal continuous positive airway pressure therapy on the quality of life of bed partners of patients with obstructive sleep apnea syndrome. Chest 124(6):2209–2214.
Drake CL, Roehrs T, Roth T. 2003. Insomnia causes, consequences, and therapeutics: An overview. Depression and Anxiety 18(4):163–176.
Drake CL, Roehrs T, Richardson G, Walsh JK, Roth T. 2004. Shift work sleep disorder: Prevalence and consequences beyond that of symptomatic day workers. Sleep 27(8):1453–1462.
Durmer JS, Dinges DF. 2005. Neurocognitive consequences of sleep deprivation. Seminars in Neurology 25(1):117–129.
Eastridge BJ, Hamilton EC, O’Keefe GE, Rege RV, Valentine RJ, Jones DJ, Tesfay S, Thal ER. 2003. Effect of sleep deprivation on the performance of simulated laparoscopic surgical skill. American Journal of Surgery 186(2):169–174.
Eckerberg B. 2004. Treatment of sleep problems in families with young children: Effects of treatment on family well-being. Acta Paediatrica 93(1):126–134.
Edinger JD, Means MK. 2005. Overview of insomnia: Definitions, epidemiology, differential diagnosis, and assessment. In: Kryger MH, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 4th ed. Philadelphia: Elsevier/Saunders. Pp. 702–713.
Emancipator JL, Storfer-Isser A, Taylor HG, Rosen CL, Kirchner HL, Johnson NL, Zambito AM, Redline SR. 2006. Variation of cognition and achievement with sleep-disordered breathing in full-term and preterm children. Archives of Pediatrics and Adolescent Medicine 160(2):203–210.
Engleman HM, Kingshott RN, Martin SE, Douglas NJ. 2000. Cognitive function in the sleep apnea/hypopnea syndrome (SAHS). Sleep 23(suppl 4):S102–S108.
Fairclough SH, Graham R. 1999. Impairment of driving performance caused by sleep deprivation or alcohol: A comparative study. Human Factors 41(1):118–128.
Federal Motor Carrier Safety Administration. 1996. Commercial Motor Vehicle/Driver Fatigue and Alertness Study. Washington, DC: Office of Research and Technology.
Fitzpatrick P, Kirke PN, Daly L, Van Rooij I, Dinn E, Burke H, Heneghan J, Bourke G, Masterson J. 2001. Predictors of first hip fracture and mortality post fracture in older women. Irish Journal of Medical Science 170(1):49–53.
Fletcher KE, Davis SQ, Underwood W, Mangrulkar RS, McMahon LF Jr, Saint S. 2004. Systematic review: Effects of resident work hours on patient safety. Annals of Internal Medicine 141(11):851–857.
Fredriksen K, Rhodes J, Reddy R, Way N. 2004. Sleepless in Chicago: Tracking the effects of adolescent sleep loss during the middle school years. Child Development 75(1):84–95.
Friedman BC, Hendeles-Amitai A, Kozminsky E, Leiberman A, Friger M, Tarasiuk A, Tal A. 2003. Adenotonsillectomy improves neurocognitive function in children with obstructive sleep apnea syndrome. Sleep 26(8):999–1005.
Frisoni GB, De Leo D, Rozzini R, Bernardini M, Buono MD, Trabucchi M. 1993. Night sleep symptoms in an elderly population and their relation with age, gender, and education. Clinical Gerontology 13(1):51–68.
Garbarino S, Mascialino B, Penco MA, Squarcia S, De Carli F, Nobili L, Beelke M, Cuomo G, Ferrillo F. 2004. Professional shift-work drivers who adopt prophylactic naps can reduce the risk of car accidents during night work. Sleep 27(2):1295–1302.
Gay CL, Lee KA, Lee SY. 2004. Sleep patterns and fatigue in new mothers and fathers. Biological Research for Nursing 5(4):311–318.
Gellis LA, Lichstein KL, Scarinci IC, Durrence HH, Taylor DJ, Bush AJ, Riedel BW. 2005. Socioeconomic status and insomnia. Journal of Abnormal Psychology 114(1):111–118.
Goswami M. 1998. The influence of clinical symptoms on quality of life in patients with narcolepsy. Neurology 50(2 suppl 1):S31–S36.
Gottlieb DJ, Chase C, Vezina RM, Heeren TC, Corwin MJ, Auerbach SH, Weese-Mayer DE, Lesko SM. 2004. Sleep-disordered breathing symptoms are associated with poorer cognitive function in 5-year-old children. Journal of Pediatrics 145(4):458–464.
Gozal D. 1998. Sleep-disordered breathing and school performance in children. Pediatrics 102(3 Pt 1):616–620.
Graeber RC, Dement WC, Nicholson AN, Sasaki M, Wegmann HM. 1986a. International cooperative study of aircrew layover sleep: Operational summary. Aviation Space and Environmental Medicine 57(12 Pt 2):B10–B13.
Graeber RC, Lauber JK, Connell LJ, Gander PH. 1986b. International aircrew sleep and wakefulness after multiple time zone flights: A cooperative study. Aviation Space and Environmental Medicine 57(12 Pt 2):B3–B9.
Grantcharov TP, Bardram L, Funch-Jensen P, Rosenberg J. 2001. Laparoscopic performance after one night on call in a surgical department: Prospective study. British Medical Journal 323(7323):1222–1223.
Greenberg GD, Watson RK, Deptula D. 1987. Neuropsychological dysfunction in sleep apnea. Sleep 10(3):254–262.
Grunstein RR, Stenlof K, Hedner JA, Sjostrom L. 1995. Impact of self-reported sleep-breathing disturbances on psychosocial performance in the Swedish Obese Subjects (SOS) study. Sleep 18(8):635–643.
Gurubhagavatula I, Maislin G, Nkwuo JE, Pack AI. 2004. Occupational screening for obstructive sleep apnea in commercial drivers. American Journal of Respiratory and Critical Care Medicine 170(4):371–376.
Hack M, Davies RJ, Mullins R, Choi SJ, Ramdassingh-Dow S, Jenkinson C, Stradling JR. 2000. Randomised prospective parallel trial of therapeutic versus subtherapeutic nasal continuous positive airway pressure on simulated steering performance in patients with obstructive sleep apnoea. Thorax 55(3):224–231.
Hack MA, Choi SJ, Vijayapalan P, Davies RJO, Stradling JR. 2001. Comparison of the effects of sleep deprivation, alcohol and obstructive sleep apnoea (OSA) on simulated steering performance. Respiratory Medicine 95(7):594–601.
Hart CN, Palermo TM, Rosen CL. 2005. Health-related quality of life among children presenting to a pediatric sleep disorders clinic. Behavioral Sleep Medicine 3(1):4–17.
Hasler G, Buysse DJ, Gamma A, Ajdacic V, Eich D, Rossler W, Angst J. 2005. Excessive daytime sleepiness in young adults: A 20-year prospective community study. Journal of Clinical Psychiatry 66(4):521–529.
Hausdorff JM, Rios DA, Edelberg HK. 2001. Gait variability and fall risk in community-living older adults: A 1-year prospective study. Archives of Physical Medicine and Rehabilitation 82(8):1050–1056.
Hope T, Keene J, Gedling K, Fairburn CG, Jacoby R. 1998. Predictors of institutionalization for people with dementia living at home with a carer. International Journal of Geriatric Psychiatry 13(10):682–690.
Horne JA, Reyner LA. 1995. Sleep-related vehicle accidents. British Medicine Journal 310(6979):565–567.
Hossain JL, Shapiro CM. 2002. The prevalence, cost implications, and management of sleep disorders: An overview. Sleep and Breathing 6(2):85–102.
Howard ME, Desai AV, Grunstein RR, Hukins C, Armstrong JG, Joffe D, Swann P, Campbell DA, Pierce RJ. 2004. Sleepiness, sleep-disordered breathing, and accident risk factors in commercial vehicle drivers. American Journal of Respiratory and Critical Care Medicine 170(9):1014–1021.
Howell AJ, Jahrig JC, Powell RA. 2004. Sleep quality, sleep propensity and academic performance. Perceptual and Motor Skills 99(2):525–535.
IOM (Institute of Medicine). 2000. To Err Is Human: Building a Safer Health System. Washington, DC: National Academy Press.
Kapur V, Blough DK, Sandblom RE, Hert R, de Maine JB, Sullivan SD, Psaty BM. 1999. The medical cost of undiagnosed sleep apnea. Sleep 22(6):749–755.
Kapur V, Strohl KP, Redline S, Iber C, O’Connor G, Nieto J. 2002a. Underdiagnosis of sleep apnea syndrome in U.S. communities. Sleep and Breathing 6(2):49–54.
Kapur VK, Redline S, Nieto F, Young TB, Newman AB, Henderson JA. 2002b. The relationship between chronically disrupted sleep and healthcare use. Sleep 25(3):289–296.
Karacan I, Thornby J, Williams R. 1983. Sleep disturbance: A community survey. In: Guilleminault C, Lugaresi E, eds. Sleep/Wake Disorders: Natural History, Epidemiology, and Long-Term Evolution. New York: Raven Press. Pp. 37–60.
Katz DA, McHorney CA. 1998. Clinical correlates of insomnia in patients with chronic illness. Archives of Internal Medicine 158(10):1099–1107.
Katz DA, McHorney CA. 2002. The relationship between insomnia and health-related quality of life in patients with chronic illness. Journal of Family Practice 51(3):229–235.
Kennedy JD, Blunden S, Hirte C, Parsons DW, Martin AJ, Crowe E, Williams D, Pamula Y, Lushington K. 2004. Reduced neurocognition in children who snore. Pediatric Pulmonology 37(4):330–337.
Kim HC, Young T, Matthews CG, Weber SM, Woodard AR, Palta M. 1997. Sleep-disordered breathing and neuropsychological deficits: A population-based study. American Journal of Respiratory and Critical Care Medicine 156(6):1813–1819.
Kim K, Uchiyama M, Okawa M, Liu X, Ogihara R. 2000. An epidemiological study of insomnia among the Japanese general population. Sleep 23(1):41–47.
Krieger J, Meslier N, Lebrun T, Levy P, Phillip-Joet F, Sailly J-C, Racineux JL. 1997. Accidents in obstructive sleep apnea patients treated with nasal continuous positive airway pressure: A prospective study. Chest 112(6):1561–1566.
Kryger MH, Roos L, Delaive K, Walld R, Horrocks J. 1996. Utilization of health care services in patients with severe obstructive sleep apnea. Sleep 19(9 suppl):S111–S116.
Kuppermann M, Lubeck DP, Mazonson PD, Patrick DL, Stewart AL, Buesching DP, Fifer SK. 1995. Sleep problems and their correlates in a working population. Journal of General Internal Medicine 10(1):25–32.
Landrigan CP, Rothschild JM, Cronin JW, Kaushal R, Burdick E, Katz JT, Lilly CM, Stone PH, Lockley SW, Bates DW, Czeisler CA. 2004. Effect of reducing interns’ work hours on serious medical errors in intensive care units. New England Journal of Medicine 351(18):1838–1848.
Leger D. 1994. The cost of sleep-related accidents: A report for the National Commission on Sleep Disorders Research. Sleep 17(1):84–93.
Leger D, Scheuermaier K, Philip P, Paillard M, Guilleminault C. 2001. SF-36: evaluation of quality of life in severe and mild insomniacs compared with good sleepers. Psychosomatic Medicine 63(1):49–55.
Leger D, Guilleminault C, Bader G, Levy E, Paillard M. 2002. Medical and socio-professional impact of insomnia. Sleep 25(6):625–629.
Li RHY, Wing YK, Ho SC, Fong SYY. 2002. Gender differences in insomnia—A study in the Hong Kong Chinese population. Journal of Psychosomatic Research 53(1):601–609.
Lindberg E, Carter N, Gislason T, Janson C. 2001. Role of snoring and daytime sleepiness in occupational accidents. American Journal of Respiratory and Critical Care Medicine 164(11):2031–2035.
Lockley SW, Cronin JW, Evans EE, Cade BE, Lee CJ, Landrigan CP, Rothschild JM, Katz JT, Lilly CM, Stone PH, Aeschbach D, Czeisler CA, Harvard Work Hours HaS Group. 2004. Effect of reducing interns’ weekly work hours on sleep and attentional failures. New England Journal of Medicine 351(18):1829–1837.
Marcus CL, Loughlin GM. 1996. Effect of sleep deprivation on driving safety in house staff. Sleep 19(10):763–766.
McArdle N, Grove A, Devereux G, Mackay-Brown L, Mackay T, Douglas NJ. 2000. Split-night versus full-night studies for sleep apnoea/hypopnoea syndrome. European Respiratory Journal 15(4):670–675.
McCurry SM, Logsdon RG, Vitiello MV, Teri L. 1998. Successful behavioral treatment for reported sleep problems in elderly caregivers of dementia patients: A controlled study. Journals of Gerontology Series B-Psychological Sciences and Social Sciences 53(2):122–129.
McCurry SM, Logsdon RG, Teri L, Gibbons LE, Kukull WA, Bowen JD, McCormick WC, Larson EB. 1999. Characteristics of sleep disturbance in community-dwelling Alzheimer’s disease patients. Journal of Geriatric Psychiatry and Neurology 12(2):53–59.
McCurry SM, Gibbons LE, Logsdon RG, Vitiello M, Teri L. 2003. Training caregivers to change the sleep hygiene practices of patients with dementia: The NITE-AD project. Journal of American Geriatrics Society 51(10):1455–1460.
McCurry SM, Gibbons LE, Logsdon RG, Vitiello MV, Teri L. 2005. Nighttime insomnia treatment and education for Alzheimer’s disease: A randomized, controlled trial. Journal of American Geriatrics Society 53(5):793–802.
Mellinger GD, Balter MB, Uhlenhuth EH. 1985. Insomnia and its treatment: Prevalence and correlates. Archives of General Psychiatry 42(3):225–232.
Mendelson WB. 2005. Hypnotic medications: Mechanisms of action and pharmacologic effects. In: Kryger MH, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 4th ed. Philadelphia: Elsevier/Saunders. Pp. 444–451.
Millman RP, Working Group on Sleepiness in Adolescents/Young Adults, and AAP Committee on Adolescence. 2005. Excessive sleepiness in adolescents and young adults: Causes, consequences, and treatment strategies. Pediatrics 115(6):1774–1786.
Mindell JA. 1999. Empirically supported treatments in pediatric psychology: Bedtime refusal and night wakings in young children. Journal of Pediatric Psychology 24(6):465–481.
Mitler M, Dement WC, Dinges DF. 2000. Sleep medicine, public policy, and public health. In: Kryger MH, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 3rd ed. Philadelphia: Elsevier/Saunders. Pp. 580–588.
Mittelman MS, Ferris SH, Shulman E, Steinberg G, Levin B. 1996. A family intervention to delay nursing home placement of patients with Alzheimer disease: A randomized controlled trial. Journal of the American Medical Association 276(21):1725–1731.
Montplaisir J, Allen RP, Walters AD, Lerini-Strambi L. 2005. Restless legs syndrome and periodic limb movements during sleep. In: Kryger MH, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 4th ed. Philadelphia: Elsevier/Saunders. Pp. 839–852.
Moore-Ede MC. 1993. The Twenty-Four-Hour Society: Understanding Human Limits in a World That Never Stops. Reading, MA: Addison-Wesley.
Moss TH, Sills DL. 1981. The Three Mile Island Nuclear Accident: Lessons and Implications. New York: New York Academy of Sciences.
Murphy SL. 2000. Deaths: Final data for 1998. National Vital Statistics Report 48(11):1–105.
Naegele B, Thouvard V, Pepin JL, Levy P, Bonnet C, Perret JE, Pellat J, Feuerstein C. 1995. Deficits of cognitive executive functions in patients with sleep apnea syndrome. Sleep 18(1):43–52.
Naylor E, Penev PD, Orbeta L, Janssen I, Ortiz R, Colecchia EF, Keng M, Finkel S, Zee PC. 2000. Daily social and physical activity increases slow-wave sleep and daytime neuropsy-chological performance in the elderly. Sleep 23(1):87–95.
NCSDS (National Commission on Sleep Disorders Research). 1994. Wake Up America: A National Sleep Alert. Volume II: Working Group Reports. 331-355/30683. Washington, DC: Government Printing Office.
NHTSA (National Highway Traffic Safety Administration). 1994. Crashes and Fatalities Related to Driver Drowsiness/Fatigue. Washington, DC: United States Department of Transportation.
NIH (National Institutes of Health). 2005. NIH State of the Science Conference Statement on Manifestations and Management of Chronic Insomnia in Adults Statement: Manifestations and Management of Chronic Insomnia in Adults. Journal of Clinical Sleep Medicine 1(4):412–421.
NSF (National Sleep Foundation). 2005. 2005 Sleep in America Poll. [Online]. Available: http://www.sleepfoundation.org/_content/hottopics/2005_summary_of_findings.pdf [accessed June 7, 2005].
NTSB (National Transportation Safety Board). 1990a. Safety Study: Fatigue, Alcohol, Other Drugs, and Medical Factors in Fatal-to-the-Driver Heavy Truck Crashes (Volume I). Washington, DC: National Transportation Safety Board.
NTSB. 1990b. Safety Study: Fatigue, Alcohol, Other Drugs, and Medical Factors in Fatal-to-the-Driver Heavy Truck Crashes (Volume II). Washington, DC: National Transportation Safety Board.
NTSB. 1997. Grounding of the Liberian Passenger Ship Star Princess on Poundstone Rock, Lynn Canal, Alaska June 23, 1995: Marine Accident Report. Washington, DC: National Transportation Safety Board [Online] Available: http://www.ntsb.gov/publictn/1997/MAR9702.pdf [accessed March 6, 2006].
O’Brien LM, Holbrook CR, Mervis CB, Klaus CJ, Bruner JL, Raffield TJ, Rutherford J, Mehl RC, Wang M, Tuell A, Hume BC, Gozal D. 2003. Sleep and neurobehavioral characteristics of 5- to 7-year-old children with parentally reported symptoms of attention-deficit/ hyperactivity disorder. Pediatrics 111(3):554–563.
O’Brien LM, Mervis CB, Holbrook CR, Bruner JL, Smith NH, McNally N, McClimment MC, Gozal D. 2004. Neurobehavioral correlates of sleep-disordered breathing in children. Journal of Sleep Research 13(2):165–172.
O’Hara R, Schroder CM, Kraemer HC, Kryla N, Cao C, Miller E, Schatzberg AF, Yesavage JA, Murphy GM Jr. 2005. Nocturnal sleep apnea/hypopnea is associated with lower memory performance in APOE epsilon4 carriers. Neurology 65(4):642–644.
Ohayon MM, Roth T. 2003. Place of chronic insomnia in the course of depressive and anxiety disorders. Journal of Psychiatric Research 37(1):9–15.
Ohayon MM, Lemoine P, Arnaud-Briant V, Dreyfus M. 2002. Prevalence and consequences of sleep disorders in a shift worker population. Journal of Psychosomatic Research 53(1):577–583.
Ong KC, Clerk AA. 1998. Comparison of the severity of sleep-disordered breathing in Asian and Caucasian patients seen at a sleep disorders center. Respiratory Medicine 92(6):843–848.
Ozminkowski R, Wang S, Trautman H, Orsini L. 2004. Estimating the cost burden of insomnia for health plans. Journal of Managed Care Pharmacy 10(5):467.
Pack AI, Pack AM, Rodgman E, Cucchiara A, Dinges DF, Schwab CW. 1995. Characteristics of crashes attributed to the driver having fallen asleep. Accident Analysis and Prevention 27(6):769–775.
Parish JM, Lyng PJ. 2003. Quality of life in bed partners of patients with obstructive sleep apnea or hypopnea after treatment with continuous positive airway pressure. Chest 124(3):942–947.
Phillips B. 2005. The Future of Sleep Medicine. Northbrook, IL: American College of Chest Physicians.
Powell NB, Schechtman KB, Riley RW, Li K, Troell R, Guilleminault C. 2001. The road to danger: The comparative risks of driving while sleepy. Laryngoscope 111(5):887–893.
Ramchandani P, Wiggs L, Webb V, Stores G. 2000. A systematic review of treatments for settling problems and night waking in young children. British Medical Journal 320(7229): 209–213.
Randazzo AC, Muehlbach MJ, Schweitzer PK, Walsh JK. 1998. Cognitive function following acute sleep restriction in children ages 10-14. Sleep 21(8):861–868.
Redline S, Kump K, Tishler PV, Browner I, Ferrette V. 1994. Gender differences in sleep-disordered breathing in a community-based sample. American Journal of Respiratory and Critical Care Medicine 149(3 Pt 1):722–726.
Redline S, Strauss ME, Adams N, Winters M, Roebuck T, Spry K, Rosenberg C, Adams K. 1997. Neuropsychological function in mild sleep-disordered breathing. Sleep 20(2):160–167.
Redline S, Adams N, Strauss ME, Roebuck T, Winters M, Rosenberg C. 1998. Improvement of mild sleep-disordered breathing with CPAP compared with conservative therapy. American Journal of Respiratory and Critical Care Medicine 157(3 Pt 1):858–865.
Reimer MA, Flemons WW. 2003. Quality of life in sleep disorders. Sleep Medicine Review 7(4):335–349.
Reuveni H, Simon T, Tal A, Elhayany A, Tarasiuk A. 2002. Health care services utilization in children with obstructive sleep apnea syndrome. Pediatrics 110(1 Pt 1):68–72.
Rhodes SK, Shimoda KC, Waid LR, O’Neil PM, Oexmann MJ, Collop NA, Willi SM. 1995. Neurocognitive deficits in morbidly obese children with obstructive sleep apnea. Journal of Pediatrics 127(5):741–744.
Ritchie K. 1996. Behavioral disturbances of dementia in ambulatory care settings. International Psychogeriatrics 8(suppl 3):439–442.
Roehrs T, Greenwald M, Roth T. 2004. Risk-taking behavior: Effects of ethanol, caffeine, and basal sleepiness. Sleep 27(5):887-893.
Rogers AE, Hwang WT, Scott LD, Aiken LH, Dinges DF. 2004. The working hours of hospital staff nurses and patient safety. Health Affairs (Millwood) 23(4):202–212.
Rosen CL, Palermo TM, Larkin EK, Redline S. 2002. Health-related quality of life and sleep-disordered breathing in children. Sleep 25(6):657–666.
Rosen CL, Storfer-Isser A, Taylor HG, Kirchner HL, Emancipator JL, Redline S. 2004. Increased behavioral morbidity in school-aged children with sleep-disordered breathing. Pediatrics 114(6):1640–1648.
Roth T, Ancoli-Israel S. 1999. Daytime consequences and correlates of insomnia in the United States: Results of the 1991 National Sleep Foundation survey. II. Sleep 22(suppl 2):S354– S358.
Sassani A, Findley LJ, Kryger M, Goldlust E, George C, Davidson TM. 2004. Reducing motor-vehicle collisions, costs, and fatalities by treating obstructive sleep apnea syndrome. Sleep 27(3):453–458.
Scharf SM, Seiden L, DeMore J, Carter-Pokras O. 2004. Racial differences in clinical presentation of patients with sleep-disordered breathing. Sleep and Breathing 8(4):173–183.
Schechter MS. 2002. Technical report: Diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics 109(4):e69.
Shin C, Kim J, Lee S, Ahn Y, Joo S. 2003. Sleep habits, excessive daytime sleepiness and school performance in high school students. Psychiatry and Clinical Neurosciences 57(4):451–453.
Simon GE, VonKorff M. 1997. Prevalence, burden, and treatment of insomnia in primary care. American Journal of Psychiatry 154(10):1417–1423.
Sleep Disorders Create Growing Opportunities for Hospitals. 2001. Health Care Strategy Management 19(2):16–17.
Spilsbury JC, Storfer-Isser A, Drotar D, Rosen CL, Kirchner LH, Benham H, Redline S. 2004. Sleep behavior in an urban U.S. sample of school-aged children. Archives of Pediatrics and Adolescent Medicine 158(10):988–994.
Stein MA, Mendelsohn J. Obermeyer WH, Amromin J, Benca R. 2001. Sleep and behavior problems in school-aged children. Pediatrics 107(4):E60.
Steinbrook R. 2002. The debate over residents’ work hours. New England Journal of Medicine 347(16):1296–1302.
Stoller MK. 1994. Economic effects of insomnia. Clinical Therapeutics: The International Peer-Reviewed Journal of Drug Therapy 16(5):873–897.
Stone KL, Schneider JL, Blackwell T, Ancoli-Israel S, Redline S, Claman D, Cauley JA, Ensrud KE, Hillier TA, Cummings SR. 2004. Impaired sleep increases the risk of falls in older women: A prospective atigraphy study. Sleep 27(276 abstract supplement):A125.
Stoohs RA, Bingham L, Itoi A, Guilleminault C, Dement WC. 1995. Sleep and sleep-disordered breathing in commercial long-haul truck drivers. Chest 107(5):1275–1282.
Stradling JR, Thomas G, Warley AR, Williams P, Freeland A. 1990. Effect of adenotonsillectomy on nocturnal hypoxaemia, sleep disturbance, and symptoms in snoring children. Lancet 335(8684):249–253.
Strawbridge WJ, Shema SJ, Roberts RE. 2004. Impact of spouses’ sleep problems on partners. Sleep 27(3):527–531.
Strine TW, Chapman DP. 2005. Associations of frequent sleep insufficiency with health-related quality of life and health behaviors. Sleep Medicine 6(1):23–27.
Stutts JC, Wilkins JW, Scott OJ, Vaughn BV. 2003. Driver risk factors for sleep-related crashes. Accident Analysis and Prevention 35(3):321–331.
Swaen GMH, Van Amelsvoort LGPM, Bultmann U, Kant IJ. 2003. Fatigue as a risk factor for being injured in an occupational accident: Results from the Maastricht Cohort Study. Occupational and Environmental Medicine 60(suppl 1):88–92.
Teran-Santos J, Jimenez-Gomez A, Cordero-Guevara J. 1999. The association between sleep apnea and the risk of traffic accidents. Cooperative group Burgos-Santander. New England Journal of Medicine 340(11):847–851.
Thomas M, Sing H, Belenky G, Holcomb H, Mayberg H, Dannals R, Wagner H, Thorne D, Popp K, Rowland L, Welsh A, Balwinski S, Redmond D. 2000. Neural basis of alertness and cognitive performance impairments during sleepiness. I. Effects of 24 h of sleep deprivation on waking human regional brain activity. Journal of Sleep Research 9(4): 335–352.
Ulfberg J, Carter N, Talback M, Edling C. 1996. Excessive daytime sleepiness at work and subjective work performance in the general population and among heavy snorers and patients with obstructive sleep apnea. Chest 110(3):659–663.
United States Senate Committee on Energy and National Resources. 1986. The Chernobyl Accident. Washington, DC: Government Printing Office.
Unruh ML, Levey AS, D’Ambrosio C, Fink NE, Powe NR, Meyer KB. 2004. Restless legs symptoms among incident dialysis patients: Association with lower quality of life and shorter survival. American Journal of Kidney Disease 43(5):900–909.
Urschitz MS, Wolff J, Sokollik C, Eggebrecht E, Urschitz-Duprat PM, Schlaud M, Poets CF. 2005. Nocturnal arterial oxygen saturation and academic performance in a community sample of children. Pediatrics 115(2):204–209.
USDOT (United States Department of Transportation). 1991. The Costs of Highway Crashes. Washington, DC: Federal Highway Administration.
USNRC (United States Nuclear Regulatory Commission). 1987. Report on the Accident at the Chernobyl Nuclear Power Station. NU-REG 1250. Washington, DC: Government Printing Office.
Van Dongen HP, Maislin G, Mullington JM, Dinges DF. 2003. The cumulative cost of additional wakefulness: Dose-response effects on neurobehavioral functions and sleep physiology from chronic sleep restriction and total sleep deprivation. Sleep 26(2):117–126.
Wahlstrom KL, Davison ML, Choi J, Rossm JN. 2001. Minneapolis Public Schools Start Time Study: Executive Summary—August 2001. Twin Cities, MN: University of Minnesota.
Walsh JK. 2004. Clinical and socioeconomic correlates of insomnia. Journal of Clinical Psychiatry 65(suppl 8):13–19.
Walsh JK, Engelhardt CL. 1999. The direct economic costs of insomnia in the United States for 1995. Sleep 22(suppl 2):S386–S393.
Walsh JK, Engelhardt CL, Hartman PG. 1995. The direct economic cost of insomnia. In: Nutt DJ, Mendelson WB, eds. Hypnotics and Anxiolytics. London: Bailliere Tindall.
Walsh JK, Dement WC, Dinges DF. 2005. Sleep medicine, public policy, and public health. In: Kryger MH, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 4th ed. Philadelphia: Elsevier/Saunders. Pp. 648–656.
Weaver TE, George CFP. 2005. Cognition and performance in patients with obstructive sleep apnea. In: Kryger MH, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 4th ed. Philadelphia: Elsevier/Saunders. Pp. 1023–1033.
Weissbluth M, Liu K. 1983. Sleep patterns, attention span, and infant temperament. Journal of Developmental and Behavioral Pediatrics 4(1):34–36.
Weissman MM, Greenwald S, Nino-Murcia G, Dement WC. 1997. The morbidity of insomnia uncomplicated by psychiatric disorders. General Hospital Psychiatry 19(4):245–250.
Wittmann V, Rodenstein DO. 2004. Health care costs and the sleep apnea syndrome. Sleep Medicine Reviews 8(4):269–279.
Wolfson AR, Carskadon MA. 1998. Sleep schedules and daytime functioning in adolescents. Child Development 69(4):875–887.
Wolfson AR, Carskadon MA. 2003. Understanding adolescents’ sleep patterns and school performance: A critical appraisal. Sleep Medicine Reviews 7(6):491–506.
Young T, Blustein J, Finn L, Palta M. 1997a. Sleep-disordered breathing and motor vehicle accidents in a population-based sample of employed adults. Sleep: Journal of Sleep Research and Sleep Medicine 20(8):608–613.
Young T, Evans L, Finn L, Palta M. 1997b. Estimation of the clinically diagnosed proportion of sleep apnea syndrome in middle-aged men and women. Sleep 20(9):705–706.
Zammit GK, Weiner J, Damato N, Sillup GP, McMillan CA. 1999. Quality of life in people with insomnia. Sleep 22 (suppl 2):S379–S385.