Chronic Illnesses and
the People Who Live with Them
Some chronic diseases are well known as “causes” of mortality. Cardiovascular disease, many cancers, stroke, and chronic lung disease are the most common causes of death in the United States (Mokdad et al., 2004; Thacker et al., 2006). There are many other chronic illnesses, however, that may or may not directly cause death but may have multiple effects on quality of life. The quality of life impact of these chronic illnesses is not as widely appreciated in public health, clinical practice, or health policy planning. Chronic illnesses often cause bothersome health problems for those affected and/or those around them, problems that persist over time. These include problems with physical health (e.g., distressing symptoms, physical functional impairment), mental health (e.g., emotional distress, depression, anxiety), or social health (e.g., social functional impairment), all of which are associated with lower quality of life (Cella et al., 2010). In many people with chronic illnesses, a mild impairment in any single one of these aspects of health leads to impairments in other aspects and may progress further to disability.
There is, in fact, a spectrum of chronic diseases that are in some ways quite disparate, yet they share certain commonalities that merit their being listed together. They are disparate in that they affect different organ systems and are frequently characterized by different time courses and the severity of disease burden. They are similar in that their effects on health and individual functioning share common pathways and outcomes. This chapter explores the differences and similarities among many chronic diseases,
considers several exemplar diseases, health conditions, and impairments in more detail, and examines the people living with these illnesses and the ways in which they are affected.1
In this section, we first consider the nature of chronic diseases, including their similarities and differences. We then discuss the effects of these illnesses on the ability to live well with them.
The National Center for Health Statistics has defined chronic diseases as those that persist for 3 months or longer or belong to a group of conditions that are considered chronic (e.g., diabetes), regardless of when they began. Although some (e.g., polymyalgia rheumatica, depression) may resolve, most are lifelong diseases. Chronic diseases can vary in multiple ways, including their stage at presentation and characteristic clinical symptoms and their natural history (time course). Some specific conditions have typical time courses for clinical progression. Other chronic diseases, such as treated breast or prostate cancers, may follow a quiescent pattern for many years. Similarly, the health burden in terms of symptoms and functional impairment, requirements for self-management, effects on significant others, and individual economic impact vary. This results in disparate patterns of human suffering across the spectrum of chronic illnesses. Table 2-1 displays selected patterns of chronic illnesses along important dimensions. For example, some illnesses (e.g., diabetes) have high self-management requirements, whereas others (e.g., Alzheimer’s disease) may require substantial care from others. Age of onset may also influence complications and burden; for example, older onset rheumatoid arthritis is associated with more shoulder involvement and symptoms of polymyalgia rheumatica and less frequent hand deformities compared with younger onset disease (Turkcapar et al., 2006). The stability of the condition over time is also an important determinant of overall health burden.
Below we summarize the spectrum of chronic diseases as early, moderate, and late stage. As highlighted in Table 2-1, individuals with certain chronic illnesses, such as congestive heart failure, chronic obstructive pulmonary disease (COPD), Parkinson’s disease, and diabetes mellitus, may
![]()
1Some chronic illnesses have a recognized precursor state (e.g., osteopenia, hyperlipidemia, ductal carcinoma in situ) that may or may not progress to a chronic condition that people sense and suffer from. Although these presymptomatic states, if diagnosed, may cause symptoms (e.g., worry) or socioeconomic consequences (e.g., inability to obtain insurance), this report focuses on persons who actually have and are living with a chronic illness, not just a precursor state. Thus, such states as asymptomatic hypothyroidism or stage 3 chronic kidney disease are not considered.
present at various stages during the course of their illness with different health and economic consequences.
Chronic illnesses can be characterized by the stage (i.e., clinical severity), pattern (i.e., continuous versus intermittent symptoms), and anticipated course (i.e., stable, fixed deficit versus progressive). Because the stage of the condition has the largest impact on health and social consequences, we have organized this section around condition stages.
Early-Stage Chronic Illnesses
We define early-stage chronic illnesses as ones that cause little or no functional impairment and impose a low burden on others. This often characterizes certain chronic illnesses early after their diagnosis or in their uncomplicated stages. For example, such illnesses as benign prostatic hypertrophy (BPH) or early Parkinson’s disease have mild symptoms and burden. Some chronic early-stage illnesses, such as uncomplicated diabetes or New York Heart Association stage I (i.e., individuals with heart disease with no physical limitations) or II heart failure (i.e., individuals with heart disease with slight physical activity limitations), although associated with low functional impairment and burden to others, are associated with a high self-management burden (e.g., the need to monitor sodium and fluid intake and daily weight in heart failure, the need for self-monitoring of blood glucose in diabetes). Other early-stage chronic illnesses, such as mild asthma or osteoarthritis, may cause physical symptoms and functional limitation only intermittently, with asymptomatic periods in between, requiring a low to moderate degree of self-management.
Moderate-Stage Chronic Illnesses
Moderate-stage illnesses can be characterized by moderate, as opposed to low, degree of functional impairment and disability and moderate to high self-management and caregiver burden. At this stage, symptoms often interfere with usual lifestyles. Examples include painful hip or knee osteoarthritis and stage 2 or 3 Parkinson’s disease.
Several illnesses are associated with disabling episodic flares, although they may have low burden between flares. They are distinguished from early-stage illnesses following this pattern in that they cause moderate to severe, episodic disability (e.g., hospitalization for a flare of COPD), increased self-management and caregiver burden, and moderate to high economic impact. COPD, rheumatoid arthritis, depression, and migraine headache are conditions that often follow this pattern. Some people with complicated diabetes may have functional impairment due to peripheral neuropathy or a lower extremity amputation yet remain stable for some years, despite high
TABLE 2-1 Selected Patterns of Chronic Illnesses: Stage, Chronicity, Burden, and Example Illnesses
| Health Burden and Consequences (not including economic) | ||||||
| Stage | Chronicity/ Time Course |
Symptomsa | Functional impairment/ disability |
Self-management burden |
Burden to others | Example Illnesses |
| Early | Chronic with episodic flares |
Minimal or none between flares |
Low | Variable | Low | Asthma in adults, mild degenerative joint disease |
| Chronic | Mild | Low | Low | Low | BPH, mild Parkinson's disease | |
| Chronic | Mild | Low | High | Low | Uncomplicated but symptomatic diabetes, NYHA 1 or II heart failure |
|
| Moderate | Chronic with episodic flares |
Mild or minimal between flares, moderate or severe during flares |
Moderate | Moderate | Modcrat | COPD, RA, depression, migraine headache |
| Chronic, quiescent |
None to moderate |
Low | Low | Low | Breast or prostate cancer in remission |
|
| Chronic, stable | Moderate | Moderate | High | Moderate | Complicated diabetes, mild to moderate stroke, mild to moderate posttraumatic states, RA with some joint deformities |
|
| Chronic, pcogreiiive |
Moderate | Moderate | Moderate | High | Severe ostcoarthritis, server Parkinson's disease. progressive Alzheimer's disease, progressive macular degeneration, progressive hearing impairment |
|
| Late | Chronic, pepgreiiive |
Moderate or server |
High | Variable | High | Severe dementia, severe diabetes with eitensiie vascular disease |
| Chronic, slowly progresive |
Moderate or server |
High | High | High | NYHA Class III or IV heart failure, COPD with chronic respiratory failure, end-stage renal disease en dialysis |
|
| Terminal | Severe | High | High | High | Metastatic cancer; patients in hospice |
NOTE: BPH = benign prostatic hyperplasia; COPD = chronic obstructive pulmonary disease; NYHA = New York Heart Association; RA = rheumatoid arthritis.
aSpecific symptoms vary by condition
self-management burden, moderate caregiver burden, and moderate to high economic impact on the individual. Similarly, people with a posttraumatic disabling condition or previous mild to moderate stroke may have a chronic pattern that remains stable over some time despite having moderate functional impairment and disability and moderate to high self-management and caregiver burden and individual economic impact.
Another pattern shown by moderate-stage chronic illnesses is more progressive. Alzheimer’s disease typically begins with memory loss and is later associated with functional impairment and behavioral and psychological complications, leading to moderate to high self-management and caregiver burden and individual economic impact. People with Parkinson’s disease and some with macular degeneration or hearing impairment may also experience this time course and burden. Amyotrophic lateral sclerosis (ALS) often begins with milder symptoms and burden but may progress rapidly to severe disability and death.
Late-Stage Chronic Illnesses
We define late-stage chronic illnesses as those that are slowly or rapidly progressive or terminal and are characterized by high functional impairment and disability and self or caregiver management burden. People with late-stage chronic illnesses often have multiple chronic conditions (MCCs) and may suffer a rapidly progressive decline in multiple functions. For example, people with severe dementia or people with diabetes and severe vascular disease often have a progressive course with high burden on significant others. In its terminal stage, metastatic cancer is often accompanied by a rapidly progressive, downhill course. In contrast, some people with late-stage chronic illnesses progress more slowly. For example, some people with end-stage renal disease who are on dialysis or some people with severe COPD and require chronic oxygen may remain stable for years. Other chronic conditions (e.g., those with spinal cord injuries) may result in high functional impairment and remain stable for many years.
Variation in a Chronic Illness in Time Course,
Health Burden, and Consequences
Although Table 2-1 indicates differences in commonly encountered patterns among chronic illnesses, it also highlights the marked variation within them. A single chronic illness may, in different people, demonstrate its own range of time course and burden. Some people with the same condition may progress from mild burden to severe limitation to disability or death at a constant, rapid rate, and others may progress slowly or not at all. For example, although the median survival for a person younger than
age 75 with Alzheimer’s disease is 7.5 years, a quarter do not survive 4.2 years and another quarter live beyond 10.9 years (Larson et al., 2004). Similarly, some people with diabetes progress inexorably to severe visual impairment, and others show little evidence of severe ocular complications or retinopathy regression even years after diagnosis (Klein et al., 1989). Only a few illnesses have a “typical” type of progression in that the vast majority of affected people show the same rate of worsening status. Most chronic illnesses are more variable, with different individuals with the same illness progressing at widely varying rates. The variation in progression rates is often independent of medical treatment. As a result of the variability of the natural history of individual illnesses, comorbidity, interactions between illness and environment, and adverse effects of treatments, the true burden of chronic illness in an individual is inconsistent and sometimes unpredictable. Thus, typical illness patterns of consequences are only rough guides. Any individual person may have a health burden that varies from the typical situation.
THE SPECTRUM OF CHRONIC ILLNESSES:
COMMON CONSEQUENCES
In addition to demonstrating differences among chronic illnesses, Table 2-1 also displays their common consequences. It is useful to consider that all of these illnesses create a common human burden of suffering. Although these illnesses have multiple mechanisms leading to suffering with variable time courses and severity, they all affect the same aspects of health: physical, mental, and social (Cella et al., 2010). A variety of models have been used to describe the process leading from disease to consequences in these aspects, including the Disablement Model that includes pathology; impairment at the tissue, organ, or body level, functional limitations; and disability (Nagi, 1976). More recently, the World Health Organization’s (WHO’s) International Classification of Functioning, Disability and Health (known as ICF) has classified health and health-related domains from “body, individual and societal perspectives by means of two lists: a list of body functions and structure, and a list of domains of activity and participation. Since an individual’s functioning and disability occurs in a context, the ICF also includes a list of environmental factors” (WHO, [a]). Regardless of the model used to explain the pathway from disease to consequences, chronic illnesses all lead, in their own ways, to human suffering (Cassell, 1983). In Table 2-1, we have rated the health burden and consequences of chronic illnesses along four dimensions: functional impairment/disability, self-management burden, and burden to others. The economic impact of chronic illness to the individual is described separately later in the chapter.
Below we discuss important dimensions of the health burden of chronic
illnesses and mention a measurement approach developed by the Patient Reported Outcomes Measurement Information System (PROMIS). The PROMIS instruments also measure related constructs of social support, interpersonal attributes, and global health but do not include management burden directly or caregiver burden (Cella et al., 2010). In a pilot study of a large but unrepresentative sample of the general population, PROMIS selected five domains to assess health-related quality of life in people with chronic illnesses: physical function, fatigue, pain, emotional distress, and social function (Rothrock et al., 2010). They found that people with chronic illnesses reported poorer scores on these domains than did people without such illnesses and that people with two or more chronic illnesses had poorer scores than people with only one had.
Symptoms
These are medical or psychiatric symptoms that can be measured quantitatively and/or qualitatively. Examples include pain, fatigue, immobility, dyspnea on exertion, claudication (lameness), foot dysesthesia (numbness), depressive symptoms, seizures, and behavioral and psychological symptoms of dementia. The PROMIS approach measures physical symptoms, emotional distress, cognitive function, and positive psychological function (Cella et al., 2010).
Functional Impairment/Disability
Functional impairment can relate to restrictions in physical, mental, or social function. Disability is a more severe impairment that limits the performance of functional tasks and fulfillment of socially defined roles (handicap). For example, physical disability is the inability to complete specific physical functional tasks, called activities of daily living (ADLs) and instrumental activities of daily living (IADLs), that are important to daily life. The PROMIS measures assess both physical function and social function.
Chronic illnesses can cause functional impairment or disability through any of the three following health pathways:
1. Directly causing impairment or disability
2. Causing other medical complications that lead to impairment and disability
3. Causing mental health complications that lead to impairment and disability
Below we consider examples of each.
![]()
FIGURE 2-1 Osteoarthritis.
Chronic Illnesses Directly Causing Disability
Osteoarthritis causes impairment or disability directly through reduced mortality or pain in such joints as the knee or hip. Knee osteoarthritis results in 25 percent of affected individuals having difficulty performing activities of daily living due to pain and limited mobility (CDC, [c]). Knee and hip osteoarthritis are the third leading cause of years lived with disability in the United States (Figure 2-1) (Michaud et al., 2006).
Chronic Illnesses Leading to Other Medical Conditions
Diabetes can lead to impairment and disability indirectly, such as its effects on blood vessels. For example, visual impairment and end-stage renal disease are often microvascular complications, and coronary heart and cerebrovascular disease are frequently macrovascular complications (Figure 2-2).
Data from the National Health and Nutrition Examination Survey show that cardiovascular disease (i.e., coronary heart disease or chronic
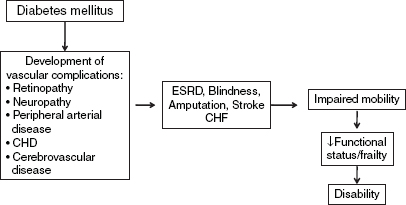
FIGURE 2-2 Diabetes mellitus and coronary heart disease as examples of complications leading to disability.
NOTE: CHD = chronic heart disease; CHF = chronic heart failure; ESRD = end stage renal disease.
heart disease [CHD], heart failure, and stroke) and obesity among older adults with diabetes were associated with greater disability in several areas, including lower extremity mobility, general physical activity, activities of daily living, and instrumental activities of daily living (Kalyani et al., 2010). Data from the Women’s Health and Aging Study show that women with diabetes had a higher prevalence of mobility disability and severe walking limitation and that this was partially explained by peripheral arterial disease and peripheral nerve dysfunction (Volpato et al., 2002).
Chronic Illnesses Leading to Mental Health Conditions
Chronic medical illnesses, such as diabetes, may also lead to mental health illnesses, such as depression and dementia, which have an adverse effect on health behaviors, leading to increased risk of clinical complications (Figure 2-3).
Both diabetes and cardiovascular disease are associated with an increased risk of developing depression (Mezuk et al., 2008; Rugulies, 2002). Conversely, depressive disorders in persons with diabetes are also associated with poor adherence to therapy (Gonzalez et al., 2008), worse control of glycemia and cardiovascular risk factors (Lustman et al., 2000), and greater diabetes complications (De Groot et al., 2001). Thus, individuals who develop depression are at higher risk of disability secondary to their greater propensity to develop vascular complications. Similarly, population-based studies indicate that type 2 diabetes is a risk factor for age-related cognitive decline (Biessels et al., 2008) with a 1.5- to 2.0-fold increased
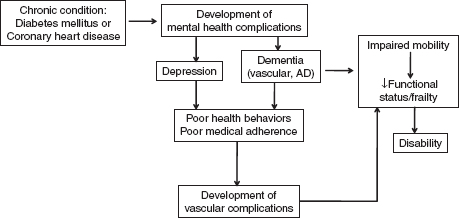
FIGURE 2-3 Association of chronic illnesses with mental health consequences. NOTE: AD = Alzheimer’s disease.
risk of all-cause dementia (Cukierman et al., 2005). Studies also show that cognitive impairment is associated with poor diabetes self-management behaviors (Sinclair et al., 2000; Thabit et al., 2009) hyperglycemia (Munshi et al., 2006), and higher prevalence of diabetes complications (Roberts et al., 2008), which are predicted to contribute to functional, in addition to cognitive, impairment in this population.
In 2011, the Centers for Disease Control and Prevention’s (CDC’s) National Center for Chronic Disease Prevention and Health Promotion released a public health action plan on mental health promotion and chronic disease prevention, which contains eight strategies to integrate mental health and public health programs that address chronic disease (CDC, 2011c). The eight strategy categories include surveillance, epidemiology research, prevention research, communication, education of health professionals, program integration, policy integration, and systems to promote integration. In recognizing the complexity of living well and effectively managing a chronic illness when a serious mental health condition is present, the committee has included a separate article highlighting depression care in patients with medical chronic illness (see Appendix A).
Chronic Illness Management Burden
In many cases, patients themselves must deliver their own care to effectively manage the chronic illnesses they live with, demanding consistent participation from patients and caregivers (Bayliss et al., 2003). In doing so, patients put forth substantial time, effort, and inconvenience that accompany day-to-day management of the illness. To properly manage their condition, patients typically run through the process of joining in physically and psychologically beneficial activities, working with health professionals to ensure adherence to treatment guidelines, monitoring health and making appropriate care decisions, and managing the effects of the illness on their physical, psychological, and social well-being (Bayliss et al., 2003). Any disruption to this process can have negative consequences on an individual’s health and livelihood (Bayliss et al., 2003).
To effectively address the multiple determinants behind almost all chronic illnesses, self-management regimens dictate appropriate medical guidelines as well as psychological and social functioning (Newman et al., 2004). Chronic illnesses factor into patient lifestyle choices, such as diet, level of physical activity, and suitable living environments, forcing self-management regimens for those illnesses to cross over multiple domains and affect the quality of a patient’s life (Newman et al., 2004). Patients with diabetes, for example, maintain day-to-day self-management routines typically including multiple components (e.g., self-monitoring of blood glucose, carbohydrate counting/awareness, home dialysis, home oxygen use, and
daily weights and check-ins with disease management programs). With all these activities, diabetes patients understandably perceive management of their condition as burdensome, frustrating, and overwhelming, which can have further negative consequences on their health (Weijman et al., 2005).
As Weijman et al. (2005) found, adherence to self-management activities has strong ties to the perceived burden. Patients who do not see these activities as burdensome perform them more frequently with close regard to proposed guidelines and reported better health outcomes in relation to their diabetes (Weijman et al., 2005). In contrast, patients who saw these activities as burdensome reported poorer health outcomes in relation to their diabetes, higher rates of depression and fatigue, and overall poorer quality of life (Weijman et al., 2005). Despite consistent evidence in support of self-management (Warsi et al., 2004), barriers still exist and complicate the self-care strategy. Many patients, such as those living with heart failure, are elderly, highly symptomatic with frequent hospitalizations, and without strong financial and social support, making self-management regimens difficult to maintain (Gardetto, 2011). In addition, issues with physical and financial limitations, health literacy, logistical complications, and lack of social and financial support interrupt and prevent effective progression through the self-management process (Bayliss et al., 2003). Without greater investment in addressing these barriers, patients will continue to face the burden behind self-management regimens designed to promote living well with chronic illness.
Social Isolation and Chronic Illness
The social consequence of chronic illness is a significant burden and impacts the ability to live well, especially when a chronic illness presents a visible functional impairment or limitation. In Social Isolation: The Most Distressing Consequence of Chronic Illness (Royer, 1998), the author eloquently describes the essence of social isolation as experienced by many individuals living with disabling chronic illnesses. Individuals living “with long-term health problems are at high risk for lessened and impaired social interactions and social isolation.” Lessened and impaired social contact and a sense of social isolation are among the more detrimental consequences of chronic illness (Royer, 1998):
Impaired social interaction relates to the state in which participation in social exchanges occurs but is dysfunctional or ineffective because of discomfort in social situations, unsuccessful social behaviors, or dysfunctional communication patterns. Indeed, social relationships are frequently disrupted and usually disintegrate under the stress of chronic illness and its management because chronic illnesses often involve disfigurement, limitations in mobility, the need for additional rest, loss of control of some body
functions, and an inability to maintain steady employment. These factors tend to reduce a person’s ability to develop and maintain a network of supportive relationships. As the illness takes up more and more of a person’s time and energy, only the most loyal family members and friends persist in offering support…. [T]he worse the illness (and/or its phases), then the more probability exists that the ill persons will feel or become isolated. Social isolation probably also occurs because family and friends need to withdraw from the ill person to gain emotional distance and protect themselves from a painful situation, particularly if they are unable to help in alleviating the problems of the sufferer. Thus, social isolation can happen in two ways: either the ill person, given the symptoms, unexpected crises, lengthy hospitalizations and convalescence, additional financial burdens, difficult regimens and loss of energy, withdraws from most social contact, or the ill person is avoided or even abandoned by friends and relatives.
The committee thinks that social isolation is not only an important consequence of long-term debilitating chronic illnesses; it is also a burden that cuts across a host of chronic illnesses, thus highlighting the commonality among many of them and presenting an opportunity to develop, disseminate, and evaluate relevant community-based interventions to help people with chronic illness.
Caregivers of Individuals with Chronic Illness
The burden of chronic illness reaches beyond the person with the illness, affecting family members as well, particularly those involved in caregiving. The National Alliance for Caregiving (NAC) and AARP conducted a national survey of caregivers in the United States to assess the issues they faced in 1997, 2004, and 2009 (NAC and AARP, 2009). The 2009 survey indicated that approximately 28.5 percent—or an estimated 65.7 million people in the United States—served as a family caregiver to an ill or disabled child or adult in the past 12 months. Caregivers of adults spend an average of 18.9 hours per week providing care. And 66 percent of caregivers are women, and women caregivers report more time spent in caregiving than men caregivers.
The burden on informal caregivers is highly variable (see Table 2-1), but as the severity of illness-related impairment increases, caregiver burdens increase as well. Research has documented numerous physical and mental health effects of caregiving. The NAC and AARP report (2009) documents that 17 percent of caregivers consider their health to be fair or poor compared with 13 percent of the general population. Health is particularly affected among low-income caregivers, 34 percent of whom report fair or poor health (NAC and AARP, 2009). Female caregivers in the Nurses’ Health Study were more likely to report a history of hypertension, diabetes,
high cholesterol, and poorer health behaviors (more likely to smoke, eat more saturated fat, and have a higher body mass index). When controlling for these factors, the study found an 82 percent higher incidence of CHD in those who cared for a spouse than in noncaregivers. There was no increased CHD risk among those providing care for an ill parent (Lee et al., 2003). The Caregiver Health Effects Study (CHES) study categorized approximately 800 spouses on the basis of their level of caregiving demand: those with disabled spouses for whom they do not provide care; those who provide care to a disabled spouse but report no caregiver strain; and those who provide care for a disabled spouse and report either physical or emotional strain. These groups were compared with spouses whose partners were not disabled, reporting no difficulty with activities of daily living. After controlling for the presence of illness and subclinical cardiovascular disease in the spouse, those spouses who provided care for a disabled partner and reported caregiver strain had 63 percent higher 4-year mortality than those whose spouses were not disabled (Schulz and Beach, 1999).
Caregivers also report increased symptoms of psychological distress. A meta-analysis of differences between caregivers of older adults with various illnesses and noncaregivers found the largest differences were in depression, stress, self-efficacy, and subjective well-being (Pinquart and Sörensen, 2003). For example, depression among caregivers was higher than in comparable groups of noncaregivers. Depression was higher among caregivers of people with dementia and more common in women than men, spouses than other family caregivers, and caregivers for whom both the perceived and the actual workload are greater (Pinquart and Sörensen, 2003; Schoenmakers et al., 2010). More time spent in caregiving is associated with higher levels of depressive symptoms (Cannuscio et al., 2004).
Caregiving can have an economic impact as well. Caregivers have a lower labor force participation rate than do adults not involved in caregiving. Effects seem particularly pronounced among women, caregivers who are in poor health themselves, older caregivers, those with more caregiving involvement, immediate family members, caregivers with young children at home, those who cared for people with more limitations, caregivers with lower incomes, and those with less education (Lilly et al., 2007). In all, 58 percent of caregivers of adults are currently employed, with 48 percent working full-time and 10 percent working part-time. And 69 percent report making work changes to accommodate caregiving, such as going in late or leaving early (65 percent), taking a leave of absence (18 percent), turning down a promotion (5 percent), losing job benefits (4 percent), giving up work entirely (7 percent), or retiring early (3 percent) (NAC and AARP, 2009). Caregiving can affect productivity through both absenteeism and presenteeim (decreased productivity while at work) (Giovannetti et al., 2009). Time spent in the physical care of the ill person or in helping them
access health care may increase absenteeism at work. Even when the caregiver is at work, he or she may be distracted by worries about the family member or by spending time dealing with insurance companies, health care records, etc. Furthermore, caregivers may be locked into jobs or prevented from advances or job transfers because of fear of loss of insurance and the need to stay in geographic proximity to the person for whom they provide care.
Economic Consequences of Chronic Illness on the Individual
Chronic illness can wreak havoc on the socioeconomic standing of an individual and his or her family (Jeon et al., 2009). Overwhelming evidence connects lower socioeconomic status with poorer health, putting a large portion of the worldwide population at risk for developing one or more chronic illnesses and further financial hardship (Jeon et al., 2009). The prevalence of chronic illness increases with age, increasing the likelihood of developing a health-related financial and economic burden as an individual gets older (Woo et al., 1997). This burden includes both direct (e.g., out-of-pocket costs of health care) and indirect (e.g., loss of work income) consequences for the individual and/or his or her caregiver or families. In terms of direct consequences, taking a microeconomic approach, a strong association exists between financial stress, disability, and poor physical and mental health and between poverty rates and chronic illness (Jeon et al., 2009). The estimated costs of addressing disability consumed approximately 29 percent of household income and 49 percent for those with severe restrictions (Jeon et al., 2009). Based on these estimates, those with one or more chronic illnesses are six times more likely to sink down to the poverty line than are those without one (Jeon et al., 2009). One Australian study interviewed 52 patients with one or more chronic illnesses and 14 caregivers (or spouses or offspring) of those patients and found that 60 percent of the patients and 79 percent of the caregivers reported experiencing financial difficulties associated with the patients’ chronic illness (Jeon et al., 2009). In all, 84 percent of both groups identified the basic cost of disease management as a primary financial challenge, and 64 percent of both groups reported experiencing financial difficulty related to addressing the patients’ chronic illness and believing that it negatively affected their quality of life (Jeon et al., 2009). Overall, both groups reported financial stress related to affordability of treatment, including out-of-pocket expenses for medications, regular check-ups, and lack of support resources, and affordability of other things, including healthy food, exercise and gym membership, and partaking in social activities (Jeon et al., 2009). In another study, conducted by Teo et al. (2011), 42 percent of the estimated cost burden of COPD was attributed to medical management alone, an expense put in different
weights on the shoulders of the patients and their caregivers. For every dollar spent on fibromyalgia-related health care expenses for its employees, certain employers spent an additional $57 to $143 on direct and indirect costs, masking any evidence of successful treatment (Robinson et al., 2003). For indirect costs alone, Ivanova et al. (2010) compared a group of employees with treatment-resistant depression (TRD) and nontreatment-resistant major depressive disorder and found TRD-likely employees were more likely to have a disability and go through more disability days. Furthermore, although TRD-likely employees had lower rates of medical-related absenteeism, they did go through a higher number of medical-related absenteeism days (Ivanova et al., 2010). From that, TRD-likely employees have more days away from work, creating a loss in productivity for the employee and extra cost for the employer (Ivanova et al., 2010). The indirect consequences of chronic illness, like missing multiple days from work and reduced productivity, increases the risk of losing employment, an event that reinforces financial pressures. Without substantial caregiver, family, or employer support, individuals with one or more chronic illnesses may sink into financial hardship beyond repair.
Effects of Comorbidity
The burden of chronic illness is often compounded by multiple chronic conditions, a situation that is often referred to as multimorbidity or comorbidity. Typically, the term comorbidity is used in the context of an index condition (e.g., cancer) to reflect the impact of other (comorbid) conditions (e.g., heart failure) on prognosis, quality of life, and treatment. Multimorbidity is used to describe MCCs that in aggregate may affect prognosis, quality of life, and treatment. Although most important conditions begin as single diagnostic entities, they may vary in their rate of progression for many reasons other than the primary pathological process. For example, prior conditions may already be present at the time of the occurrence of the new condition, leading to an increased burden for these “new” index conditions. Multimorbidities can contribute to worse outcomes because of complications that affect multiple organ systems, either individually (e.g., macular degeneration may affect vision and osteoarthritis may affect mobility in the same person) or synergistically (e.g., diabetes and hypertension together may accelerate atherosclerotic coronary, cerebrovascular, and peripheral vascular disease). Multimorbidities can also complicate treatment regimens, including competing guidelines for care that may confuse people, decreasing adherence or leading to conflicting therapeutic regimens (Boyd et al., 2005; Tinetti et al., 2004). One condition can also interfere with the ability to adhere to treatment for another condition, such as osteoarthritis occurring in individuals with diabetes or cardiovascular disease inhibiting
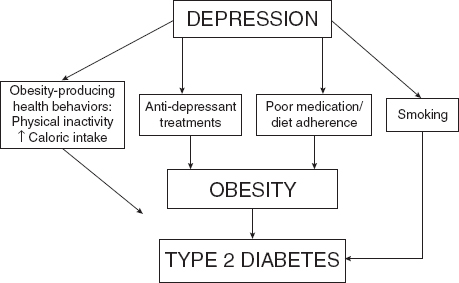
FIGURE 2-4 Depression and the risk of diabetes.
participation in physical activity (Bolen et al., 2009). Primary mental illnesses, such as depression, can increase the risk for medical conditions and the adverse outcomes associated with them (Figure 2-4).
In addition, comorbid depression or anxiety is associated with higher numbers of medical symptoms across a wide variety of illnesses (Katon et al., 2007), in part because of their association with poor adherence to self-care regimens (Lin et al., 2004) and heightened awareness of symptoms (Katon et al., 2001).
Finally, secondary conditions of varying importance and impact can occur because of the debilitating effects of the primary illness. These secondary conditions can take various forms depending on the primary condition and the nature of care, including falls, fractures, depression and other mental consequences, constipation, bedsores, anemia, obesity, sleep disorders, social dysfunction, spasticity, and injuries from various medical devices. These are important not only for their health impact but also because many can be prevented or mitigated with optimal care. Thus, they are also important objects of surveillance in order to define the population burden of chronic disease. This understanding that functional limitation due to one chronic condition may lead to disability through the development of other chronic illnesses provides an opportunity for the prevention of disability. If prevention approaches for people with chronic illness can reduce the risk of developing additional ones, the risk of disability may be reduced as well.
Illness-Environment Interactions
The interaction between persons with chronic illness and their environments can also contribute to the burden and consequences they may experience. For example, a person with late-stage Alzheimer’s disease who has a family caregiver or has the resources to hire a paid caregiver may be able to remain at home, whereas a similar person without this support system is likely to be institutionalized. Similarly, a person with severe rheumatoid arthritis who works in the service industry may be able to continue working by use of voice recognition technology and telecommuting from home, whereas someone who works in construction would be unable to work.
Adverse Effects of Clinical Treatment
Another reason for variation in the rate of development of disability is adverse effects of treatment. Some illnesses may lead to less physical fitness, as with fatigue and muscle atrophy. Moreover, it is well described that patients undergoing varying kinds of clinical care are subject to the adverse effects of that care (IOM, 1999). Adverse effects occur in all elements of care, including medications (Kongkaew et al., 2008); institutionalization, such as hospitalizations and surgical procedures (Michel et al., 2004); and long-term care in various settings (Dhalla et al., 2002). Patients with chronic illnesses, because of extensive and often intensive care experiences, are thus particularly likely to experience adverse effects, even if, in general, their health is better off with the care than without it. Although the severity of adverse effects is sometimes difficult to characterize in detail, care surveillance systems and quality improvement programs clearly demonstrate the general scope of the problem and the need for remediation whenever possible. It is very difficult to identify studies that summarize the net health impact of adverse effects across common chronic illnesses. In complex illnesses, it may be difficult to distinguish between an effect of the illness and the effect of the treatment. Nonetheless, there is an important need to understand the role of adverse effects in affecting the health trajectories of those with chronic illness.
One of the charges to the committee was to suggest a new set of diseases for which to provide increased emphasis in terms of surveillance and chronic disease control efforts. As always, such programmatic emphases may change over time, in part because of the advent of new community or clinical interventions that can improve the lives of individuals with chronic
illness. There are many illnesses from which to choose—in many ways, almost an endless menu of conditions that can lead to suffering and disability.
In addressing the challenges of living well with chronic illness, priorities must be established. Although priority setting in public health and health care is not a new concept, it is a matter of growing importance (Ham, 1997). The combination of constrained resources and increasing demands has led policy makers to address priority setting more directly than in the past. In particular, an explicit part of the committee’s task asked “Which chronic diseases should be the focus of public health efforts to reduce disability and improve functioning and quality of life?”
Fundamentally, the determination of priorities for public health intervention begins with the burden of disease and preventability (Sainfort and Remington, 1995). Other considerations include size of the chronic disease problem, perceptions of urgency, severity of the problem, potential for economic loss, impact on others, effectiveness, propriety, economics, acceptability, legality of solutions, and availability of resources (Vilnius and Dandoy, 1990).
Although there is no correct approach to setting priorities, it is beneficial to have a common planning framework. The framework should
• include multiple perspectives, including patients, providers, employers, and community members;
• use clear and consistent criteria for selecting priorities, whenever possible;
• result in aims and objectives that are clear and feasible;
• consider at what level the decisions are being made (e. g., federal, state, local); and
• include the values of these involved in the decisions.
Despite the challenges involved in setting programmatic priorities, a number of organizations have used these measures and approaches to set health priorities. The Oxford Health Alliance based in the United Kingdom convened a group from around the world of academics, nongovernmental organizations, activists, corporate and industry executives, patients’ rights advocates, health professionals, and others to focus on preventing the worldwide epidemic of chronic diseases (http://www.oxha.org). In 2006, they launched the “3four50” effort (http://www.3four50.com/). This “open space for health” promotes chronic disease prevention by focusing on the three risk factors (poor diet, lack of physical activity, and tobacco use) that lead to four chronic diseases (cardiovascular disease, diabetes, chronic lung diseases, and some cancers) contributing to more than 50 percent of deaths worldwide.
CDC has not set priorities explicitly but has developed the approach
called Winnable Battles to describe public health priorities with large-scale impact on health and with known, effective strategies to intervene (CDC, [d]). The charge under Winnable Battles is to identify optimal strategies and to rally resources and partnerships to accelerate a measurable impact on health. The priority areas for CDC include some that relate directly to chronic disease, including physical activity promotion, obesity elimination, and tobacco control.
Although the federal Healthy People 2010 did not explicitly set national priorities, it established leading health indicators to reflect major public health concerns in the United States (CDC, [b]). These leading health indicators were selected on the basis of their capacity to motivate action, the availability of data to measure progress, and their importance as public health issues. The Institute of Medicine (IOM) published a report recommending leading health indicators for Healthy People 2020 (2011a). These also include several that pertain to living well with chronic illness. In this chapter, we have explained the additional framework used to select paradigm diseases based on the great variation in their causes, onset, clinical patterns, and outcomes (see Table 2-1). These highlight some of the important dimensions and variations in chronic illnesses that are relevant to patients, the health care system, and the nation, including
a. time course, chronicity, and downstream consequences;
b. enormous variation in etiology and pathogenesis;
c. late-stage manifestations;
d. symptom patterns;
e. functional impairment and disability;
f. secondary consequences, such as falls, sleep disorders, pressure sores;
g. multimorbidity associated with several coexisting chronic illnesses;
h. management burden, both to the patient, the family, and other caregivers and to the health care system;
i. social consequences, such as isolation;
j. economic consequences to the patient and society;
k. impact on the environment; and l. important adverse effects of therapy.
Given such great diversity and a real absence of population data for these dimensions (except possibly in some instances for the most common diseases), the committee took the exemplar approach to highlight disease complexity, diversity, cross-cutting commonalities, and the implications for multidimensional approaches to chronic disease surveillance and control.
The multidimensional approach to selecting the exemplars was derived from the committee’s view that an additional approach to chronic disease
was needed to supplement current approaches for selecting the most common, high-mortality diseases for public health control efforts. The committee’s approach, while appreciating the wisdom and practicality of current approaches, is grounded in other considerations:
1. Current approaches to selecting diseases for control activity based on such criteria as prevalence, mortality, disability, and economic cost to the care system are useful, but these criteria are often orthogonal to each other, and thus the selection algorithm is in several ways arbitrary.
2. Current approaches to selecting diseases for public health focus inadequately address the great variation in clinical manifestations and trajectories that make public health approaches complex and challenging.
3. Current approaches are not inclusive of the large number of less common illnesses that impact individuals and communities in important ways.
4. The recognized problem of MCCs has not been adequately addressed in current disease control activities.
For these reasons, the committee recommended an “exemplar” approach to address some of these perceived inadequacies. This approach starts with a framework, presented in this chapter, that begins not with a specific set of conditions or criteria for them but with a broad set of clinical manifestations and other consequences experienced by individuals with chronic illness. The committee thinks that this framework highlights a new and alternative approach to public health chronic disease control. The exemplars did not come from a list. Rather, they come from the clinical and research experience of committee members and were chosen to highlight some important features of chronic diseases that have received less emphasis in the past, including
1. Great diversity in clinical manifestations within and among chronic diseases and the great variation in their manifestations as illnesses continue their natural histories.
2. The inclusion of illnesses that can be manifest across the life course, raising the possibility of public health interventions that may be effective at various life stages of disease. The life course approach also more effectively deals with the occurrence of recurrent or additional different conditions (MCCs).
3. The highlighting of important psychological and social consequences that come with many chronic illnesses, including individuals
with primary mental illnesses and those that are secondary to other conditions.
4. The highlighting of the chronic, multiple, degenerative age-related conditions, for which public health approaches are perhaps less well developed.
In addition, the committee endorses CDC’s emphasis on “winnable battles” and thinks that the exemplar approach will help identify new types of battles and population-based interventions in the management and control of chronic diseases. Accordingly, the committee has selected nine emblematic diseases, health conditions, and impairments, because together they encompass and flesh out the range of key issues that affect the quality of life of patients with the full spectrum of chronic illnesses. More importantly, if interventions, policies, and surveillance were developed to address these nine diseases, they would also address diseases similar to them. The exemplar approach also avoids the trap of pitting one disease against another in competing for resources and attention. Rather, it conceptualizes the commonalities across diseases with the intent of developing strategies that benefit all affected by the vast array of chronic diseases.
Thus, we have sampled from the different patterns (clinical manifestations and trajectories) of chronic diseases in order to represent the important dimensions of varying chronic disease manifestations. The nine clinical clusters—not all specific and individual diseases and conditions in the literal sense—are described below, with brief comments on their epidemiology and community impact. Each represents an important challenge to public health, in addition to those diseases that have received more attention, namely, the diseases responsible for much of morbidity and mortality and significantly add to health care cost in the United States and other developed countries. The nine are arthritis, cancer survivorship, chronic pain, dementia, depression, diabetes mellitus type 2, posttraumatic disabling conditions, schizophrenia, and vision and hearing loss.
Arthritis
Arthritis is the term used to describe more than 100 rheumatic diseases and conditions that affect joints, tissues surrounding the joints, and other connective tissue.
Arthritis is a highly prevalent condition. It is estimated that 50 million adults in the United States (approximately one in five) report doctor-diagnosed arthritis (CDC, 2011a). Arthritis is more prevalent in older age groups, women, individuals who are overweight, and individuals with lower socioeconomic status. It affects members of all racial and ethnic groups (AAOS, 2008; CDC, 2011a; Dalstra et al., 2005). Although arthritis
is more prevalent in older age groups, with half of adults age 65 and older reporting arthritis, nearly two-thirds of the adults reporting doctor-diagnosed arthritis are younger than age 65 (AAOS, 2008). As the U.S. population ages, the prevalence of arthritis is projected to increase over current levels to 67 million by 2030 (CDC, 2011a; Hootman and Helmick, 2006).
In addition to being one of the most prevalent chronic illnesses, arthritis is the leading cause of disability (McNeil and Binette, 2001) and one of the leading causes of work limitations (Stoddard et al., 1998). In 2008, 29 million persons over age 18, 13 percent of all adults in the United States, had self-reported activity limitations attributable to arthritis (AAOS, 2008). As with the frequency of arthritis, the prevalence of arthritis-attributable activity limitations increases as people age. Among adults age 65 and older, 28 percent reported activity limitations attributed to arthritis in 2008 (AAOS, 2008). In terms of work disability, 5.3 percent of all U.S. working-age adults (age 18 to 64) reported work limitations due to arthritis (CDC, 2011a).
Significant personal and societal burdens result from the high prevalence of arthritis and limitations and disability associated with it. In 2004, the estimated annual cost of medical care for arthritis and joint pain was $281.5 billion (AAOS, 2008). Of this amount, $37.3 billion is estimated to be incremental cost that can be directly attributed to arthritis and joint pain (AAOS, 2008). The indirect cost of arthritis and related rheumatic conditions due to lost earnings was estimated to be $54.3 billion in 2004 (AAOS, 2008). This includes an estimated $22 billion as a result of OA, $17.1 billion from RA, and $15.2 billion from gout (AAOS, 2008). These costs do not include the intangible costs of an individual forgoing the activities that they and society value.
Arthritis, in particular, is often comorbid with other conditions. A total of 24 percent of adults with arthritis have heart disease, 19 percent have chronic respiratory illnesses, and 16 percent have diabetes (CDC, [a]). Conversely, 57 percent of people with heart disease and 52 percent of people with diabetes have arthritis.
The most commonly occurring type of arthritis is osteoarthritis (OA), characterized by progressive damage to the cartilage and other joint tissues (AAOS, 2008). OA frequently affects the hands, knees, and hips. Other forms of arthritis that occur frequently include rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), fibromyalgia, and gout (CDC, 2011a). Pain, stiffness, and swelling are common symptoms for these conditions, and some forms of arthritis, such as RA and SLE, also have a systemic component whereby multiple organs can be affected (Arthritis Foundation, 2008). The prevalence of OA can be estimated in terms of either radiographic changes related to the presence of OA or as symptomatic
OA, which includes having pain, aching, or stiffness in the same joint that shows radiographic OA (AAOS, 2008). More than 27 million U.S. adults have OA, and it is estimated that half of all adults will develop symptomatic OA of the knee at some point their lives (Arthritis Foundation and CDC, 2010; Murphy et al., 2008). In addition to being more common in women and obese individuals, OA is more common in certain occupations, including mining, construction, agriculture, and certain segments of the service industry (Arthritis Foundation and CDC, 2010). Approximately 25 percent of people with knee OA have difficulty performing activities of daily living and also have pain on ambulation (Arthritis Foundation and CDC, 2010). OA interferes with working adults’ (age 18 to 64) work productivity, and their employment rates are lower than among adults without arthritis (Arthritis Foundation and CDC, 2010). It is estimated that $3.4 to 13.2 billion is spent on job-related OA costs per year (Arthritis Foundation and CDC, 2010). In terms of direct medical costs, in 2004, OA resulted in more than 11 million physician and outpatient visits, 662,000 hospitalizations, and more than 632,000 total joint replacements (Arthritis Foundation and CDC, 2010).
RA, the second most common type of arthritis, is a chronic autoimmune disease that causes pain, stiffness, swelling, and limitation in the motion and function of multiple joints. The prevalence of RA is estimated to be around 0.6 percent of the population over the age of 17, approximately 1.3 million adults in 2005 (AAOS, 2008). RA is twice as common in women as in men. In 2006, RA accounted for 2.9 million ambulatory care visits and 15,400 short-stay hospitalizations (AAOS, 2008). This estimate does not account for hospitalizations related to arthritis treatment complications, such as gastrointestinal bleeding related to the use of nonsteroidal anti-inflammatory drugs, and it does not account for hospitalizations related to orthopedic procedures (AAOS, 2008).
In summary, arthritis and related rheumatic conditions have a significant impact on the quality of life of affected individuals, with substantial physical, psychosocial, and economic consequences.
Cancer Survivorship
The number of cancer survivors in the United States is on the rise; in 2007 there were nearly 12 million people alive in the United States with a previous cancer diagnosis, up from approximately 3.5 million in 1971 (NCI, 2011; Rowland et al., 2004). Survivors older than 65 comprise 7 million of the 12 million survivors, the largest survivor age group (NCI, 2011). With the aging of the U.S. population, this group of cancer survivors 65 is projected to grow faster than other age groups (Smith et al., 2009). In addition, cancer is expected to increase more rapidly in all nonwhite racial
and ethnic groups; between 2000 and 2030, cancer cases are expected to increase by 31 percent in whites, and by 99 percent in nonwhite racial and ethnic groups (Smith et al., 2009).
Cancer is a serious and often life-threatening disease, requiring difficult and intensive treatments that may leave survivors with lasting negative health consequences, despite a stabilization or elimination of their cancer. Cancer treatment can affect the health, functioning, and well-being of survivors. These can be divided into long-term effects (side effects/complications that begin during treatment and persist beyond the end of treatment) or late effects (side effects/treatment toxicities that are unrecognized or subclinical at the end of treatment but emerge later because of developmental processes), decreased ability to compensate as the survivor ages, or organ senescence (IOM and NRC, 2006). Nearly every organ system and tissue has the potential to be affected by cancer treatment, including cardiovascular, pulmonary, neurological, lymphatic, bone, endocrine, gastrointestinal, hematologic, hepatic, immune, ophthalmologic, and renal systems. A thorough description of the medical and psychosocial effects of cancer can be found in the IOM report From Cancer Patient to Cancer Survivor: Lost in Transition (IOM and NRC, 2006), but some examples of lasting and late effects are described below.
Highly effective and frequently used anthracycline chemotherapy can cause left ventricular dysfunction and heart failure (Pinder et al., 2007; Towns et al., 2008). For example, Pinder et al. (2007) found a 26 percent increased risk of congestive heart failure in breast cancer survivors between the ages of 66 and 70 who received anthracycline-based chemotherapy, compared with those who did not receive adjuvant chemotherapy. Newer targeted therapies, such as trastuzumab (Herceptin), bevacizumab, and sunitinib, also can have detrimental effects on the heart (Chu et al., 2007; Floyd et al., 2005).
Cancer surgery that removes lymph nodes (as well as radiation therapy to the nodes) can lead to lymphedema, the collection of fluid in a limb or other body part due to impedance of the flow of fluid in the lymphatic system, leading to swelling, pain, and loss of function. Lymphedema is frequently a concern for breast cancer survivors (NCI, [a]); it can also affect survivors of melanoma, gynecologic, genitourinary, and head and neck cancers (Cormier et al., 2010).
Radiation therapy can damage healthy tissue as well as tumor cells; effects on healthy tissue may involve cell killing through DNA double-strand breaks but also increased risk of fibrosis and impaired function in blood and lymph vessels. The effects of the damage depend on the area that was irradiated; for example, survivors who have radiation treatment for gynecologic cancers report 12 times the risk of bowel incontinence compared with controls who have not had cancer (Lind et al., 2011).
Other aftereffects of cancer are prevalent but are more difficult to tie to specific treatment toxicities. Nevertheless, cancer survivors report persistent problems with fatigue, sleep difficulties, and psychological distress, particularly anxiety about recurrence (Bower et al., 2008). Furthermore, survivors are at increased risk of second primary tumors, either because of host susceptibility or treatment effects, necessitating careful surveillance for cancer recurrence and detection of new cancer (IOM and NRC, 2006).
More than ever before, cancer is being managed like a chronic disease. In part this is due to the late effects described above. However, it is also because the treatment of cancer has been extended for many cancer sites. For example, women with estrogen-receptor positive breast cancer receive the recommendation to take estrogen-suppressing therapy for 5 years, and in some cases survivors experience troublesome side effects, such as joint and muscle pain (Mao et al., 2009). The treatment of chronic myelogenous leukemia has been revolutionized by the use of imatinib, a targeted agent that has relative low toxicity but is taken for an indefinite period of time to keep the disease at bay. Even metastatic disease, which has historically resulted in a rapid decline and death, has more treatment options, so that for certain disease sites, such as breast and colon, survivors with metastatic disease are living longer. Survivors with metastatic illness often stay on a therapy until it stops working or the side effect burden becomes too great, when they may switch to another therapy.
Lasting and late effects, as well as side effects from continuous treatment, have negative repercussions for health and functioning in a range of areas. Results from analyses of the National Health Interview Survey show that cancer survivors are more likely to rate their health as fair or poor (31 percent) than the noncancer controls (17.9 percent). They also are more likely than controls to report functional limitations, including needing help with ADL (cancer survivors, 4.9 percent; controls, 3 percent), instrumental activities of daily living (cancer survivors, 11.4 percent; controls, 6.5 percent), and any limitation (cancer survivors, 36.2 percent; controls, 23.8 percent). Survivors are more likely to report being unable to work and being more limited in the amount of type of work they can do because of health (Yabroff et al., 2004).
These functional limitations persist long after diagnosis; one study found that the odds of having a functional limitation in cancer survivors versus controls was similar for survivors within 5 years of diagnosis and more than 5 years after diagnosis; in an analysis of data from the National Health and Nutrition Examination Survey, Ness et al. (2006) found that the odds of physical performance limitations were 85 percent higher in survivors within 5 years of diagnosis compared with adults who had not had cancer, and by 49 percent among those who were 5 or more years from diagnosis after controlling for sex, age, race/ethnicity, and annual house-hold
income. Age and comorbid health problems also complicate the health status of cancer survivors.
Because age is one of the strongest risk factors for cancer, most cancer survivors are older (60 percent are age 65 or older; NCI, 2011), and 42.1 percent have one or more chronic illnesses other than their cancer (compared with 19.7 percent among those who have not had cancer (Hewitt et al., 2003). Approaches to living well need to take into account issues of aging and MCCs.
Chronic Pain
Pain varies in severity and locale. It can be mild or acute, but in many cases it can be chronic. Some of the most commonly occurring chronic pain originates from headaches, the lower back, cancer, arthritis, peripheral nerve damage, and an unknown source (NINDS, [a]). Approximately 100 million adults within the United States suffer from chronic pain (IOM, 2011b). The different forms and origins of pain vary in prevalence. As various studies have shown, however, chronic pain is on the rise, continuing to affect both men and women and individuals of all races and ethnicities. The level of chronic pain experienced worldwide is expected to continue to increase as the population ages and rates of obesity and physical inactivity leading to pain-related conditions soar (Phillips and Harper, 2011). For example, a survey of North Carolina residents found that the prevalence of chronic low back pain increased from 3.9 to 10.2 percent between 1992 and 2006 (Freburger et al., 2009). Similarly, the number of cancer diagnoses continues to rise, with 50 to 90 percent of patients suffering from cancer- and treatment-related pain (WHO, 2008; Zaza and Baine, 2002). Recent literature suggests that racial and ethnic minorities, including African Americans and Hispanics, have greater chances of going undertreated for pain than white Americans (Green et al., 2003).
Chronic pain may result from a previous injury or medical condition, or it may have no known cause (NINDS, [a]). It can be considered a disease, as it has the potential to increasingly damage the nervous system over time (IOM, 2011b). Chronic pain often occurs with a variety of comorbidities. In many instances, it occurs in conjunction with other pain-inducing conditions, such as chronic fatigue syndrome, fibromyalgia, and vulvodynia (NINDS, [a]). Furthermore, it often occurs in conjunction with other mental conditions, such as depression and multiple mood and anxiety disorders, including panic disorder and posttraumatic stress disorder (Bair et al., 2003; McWilliams et al., 2003).
Chronic pain and musculoskeletal disorders typically score lowest in terms of quality of life (Phillips and Harper, 2011). Depending on the type and severity of pain experienced, chronic pain can cause a substantial
amount of disablement. Even differing levels of pain with the same origin, such as the low back, can lead to differing levels of disablement. Low back pain symptoms range from being specific and part of a specific pathology to being localized or part of a widespread, unknown pathology (Wormgoor et al., 2006). As pain decreases in specificity, patients often focus on it more, resulting in greater distress and dissatisfaction with life factors (Wormgoor et al., 2006). It has also been found, however, that as pain increases in specificity, loss of function and activity limitations increase (Wormgoor et al., 2006). In either form, the studied group illustrates that pain leads to negative consequences in functioning. In another study, individuals who suffer from chronic daily headaches demonstrated significant decreases in all health-related markers on the SF-36 health survey compared with healthy individuals, with the highest decreases found in role, physical, bodily pain, vitality, and social functioning (Guitera et al., 2002). In the population studied, chronicity of pain had greater influence than intensity of pain on quality of life (Guitera et al., 2002). A review of 52 studies conducted by Jensen and colleagues (2007) found solid evidence that the presence and severity of chronic neuropathic pain is associated with impairments in physical, emotional, role, and social functioning.
The burden associated with chronic pain reaches far beyond the individual suffering from it (Phillips and Harper, 2011). Significant functional disablement translates into substantial financial outcomes, reaching beyond the individual to the individual’s caretaker and family, community, and country. Evidence shows that chronic pain has a substantial impact on productivity levels, as it results in higher rates of absenteeism and the likelihood of leaving the workforce (Phillips and Harper, 2011). One study showed that, among spouses of individuals suffering from chronic pain, 35 percent had to take on extra work to support the family, 43 percent had to take time off to care for the pain sufferer, 37 percent had to assume greater financial-related task responsibility, and 89 percent had to assume greater household responsibility (Hahn et al., 2001). Mechanical low back pain ranks fourth out of the top 10 most costly physical health conditions affecting American businesses today in terms of total medical expenses, medical-related absences, and short-term disability payments (Goetzel et al., 2003). Ricci and colleagues (2005) estimated the annual lost productive work time cost due to arthritis in the U.S. workforce at around $7.11 billion, with 65.7 percent attributable to the 38 percent of workers with pain exacerbations. In a previous IOM report, it was estimated that the annual cost of chronic pain in the United States runs anywhere from $560 to $635 billion (IOM, 2011b).
In the battle against the development of chronic pain, a myriad of primary preventive interventions have been tested. Psychological factors are tightly connected to the development of costly disability (Linton and
BOX 2-1
Findings from Relieving Pain in America
• Need for interdisciplinary approaches. Given chronic pain’s diverse effects, interdisciplinary assessment and treatment may produce the best results for people with the most severe and persistent pain problems.
• Importance of prevention. Chronic pain has such severe impacts on all aspects of the lives of its sufferers that every effort should be made to achieve both primary prevention (e.g., in surgery for broken hip) and secondary prevention (of the transition from the acute to the chronic state) through early intervention.
• Wider use of existing knowledge. While there is much more to be learned about pain and its treatment, even existing knowledge is not always used effectively, and thus substantial numbers of people suffer unnecessarily.
SOURCE: IOM, 2011b.
Ryberg, 2001). Because of this, cognitive-behavioral interventions often have positive results in preventing further disability (Linton and Ryberg, 2001). Linton (2002) showed that it is possible to identify patients who suffer from musculoskeletal pain at high risk for developing pain-related disability and to successfully lower their risk of work disability through cognitive-behavioral intervention. Once disability appears, however, similar therapy methods still appear successful. Linton and Ryberg (2001) provided evidence of this as study participants suffering from chronic neck and back pain undergoing cognitive-behavioral group intervention showed significantly better results in terms of fear-avoidance beliefs, number of pain-free days, and use of sick leave.
Relevant findings from the IOM’s report Relieving Pain in America are presented in Box 2-1.
Dementia
Dementia affects 13 percent of persons age 65 and older and up to 43 percent of persons age 85 and older (Alzheimer’s Association, 2011a). In the United States, an estimated 5.4 million persons are affected by Alzheimer’s disease (Alzheimer’s Association, 2011b). Moreover, the burden of dementia is even higher, as Alzheimer’s disease accounts for only 60 to 80 percent of cases of dementia. Although dementia is commonly thought of as a condition of the elderly, an estimated 220,000 to 640,000 persons under age 65 are also affected (Alzheimer’s Association, 2006). Studies in
nursing homes indicate that 26 to 48 percent of residents have dementia (Magaziner et al., 2000; O’Brien and Caro, 2001).
These patients and their families have needs far beyond those of healthier older persons and those who have chronic illnesses that do not affect memory. In many respects, dementia is a prototypic chronic disease that requires both medical and social services to provide a high quality of care and to prevent complications, including repeated hospitalizations (Chodosh et al., 2004) and high care costs. In 2011, Medicare and Medicaid programs for people with Alzheimer’s disease were estimated at $130 billion (Okie, 2011). The clinical manifestations of dementia are protean and devastating and include cognitive impairment, immobility and falls, swallowing disorders and aspiration pneumonia, urinary and fecal incontinence, and behavioral disturbances (e.g., agitation, aggression, depression, hallucinations), which lead to caregiver stress and burnout.
Most cases of dementia start insidiously, often beginning with mild memory symptoms and progressing to mild cognitive impairment when deficits can be demonstrated on clinical examination. By the time of diagnosis of dementia, there are deficits in other dimensions of cognition (e.g., language, visual-spatial, executive function) in addition to memory that interfere with functioning. As the illness progresses, patients progressively lose memory and function and, at the late stages, may have no or unintelligible speech. Patients spend more years with severe dementia than in earlier stages (Arrighi et al., 2010). Almost all patients with dementia have at least one coexisting medical illness, especially coronary heart disease (26 percent), diabetes (23 percent), congestive heart failure (16 percent), and cancer (13 percent). Persons with dementia and these illnesses have more hospital stays than those with the same illnesses without dementia (Alzheimer’s Association, 2011a). Although dementia has variable rates of progression and lengths of survival after diagnosis, the median is 4 to 8 years (Brookmeyer et al., 2002; Ganguli et al., 2005; Helzner et al., 2008; Larson et al., 2004).
Dementia is a particularly devastating illness because the clinical manifestations affect the ability to maintain function and manage other chronic illnesses. Moreover, as dementia progresses, its complications often result in caregiving needs that may overwhelm the care of other preexisting and new chronic illnesses.
Nationwide in 2010, an estimated 15 million caregivers provided 17 billion hours of care worth $202 billion (Alzheimer’s Association, 2011a). And 80 percent of care provided in the home for patients with dementia is delivered by family caregivers who provide ADL and IADL functions, manage safety issues and behavioral symptoms, and coordinate medical and supportive care. Although these caregivers report positive feelings about this role, 61 percent rated the emotional stress of caregiving as high or very
high (Alzheimer’s Association, 2011a), and approximately one-third report symptoms of depression (Taylor et al., 2008; Yaffe et al., 2002). The physical health of caregivers may also be affected. For example, caregivers of dementia patients have increased rates of coronary heart disease (Vitaliano et al., 2002).
Current medications can sometimes slow the course of decline of Alzheimer’s disease and some other dementias, but they do not cure the disorder. The addition of a dementia care manager to primary care practices can improve quality of care, reduce complications of aggression and agitation, and prevent caregiver depression (Callahan et al., 2006). Similarly, a disease management program led by care managers has been shown to improve patient health-related quality of life, overall quality of patient care, caregiving quality, social support, and level of unmet caregiving assistance needs (Vickrey et al., 2006). In addition, partnering with local Alzheimer’s Association chapters can improve the quality of dementia care (Reuben et al., 2010).
Research is needed on models of care that link health care systems with community-based organizations to provide the wide range of services needed by patients with dementia. This research needs to include developing payment structures for community-based social services that are necessary to provide comprehensive care for persons with dementia. As stated in the IOM report Retooling for an Aging America: Building the Health Care Workforce (2009), “research is needed for the development and promulgation of technological advancements that could enhance an individual’s capacity to provide care for older adults including the use of ADL technologies and information technologies that increase the efficiency and safety of care and caregiving.”
Depression
Major depression is a common chronic illness that causes a substantial degree of impairment and disability (Michaud et al., 2006). National studies in the United States found a point prevalence of about 7 percent in 2001 and 2002 (Compton et al., 2006). Cohort studies found that the lifetime prevalence of major depression is 17 percent (National Comorbidity Survey Replication, 2007). The prevalence among women is about twice that among men (Murphy et al., 2000), and the lifetime prevalence is higher for whites than for African Americans (Williams et al., 2007). Both point prevalence and lifetime prevalence of major depression is higher for younger than for older persons (Kessler et al., 2010). However, depression is more common in older persons with a greater number of chronic illnesses, including those with disabilities (Charney et al., 2003; Lebowitz et al., 1997; Lyness et al., 2006).
Major depression causes a large burden of suffering on both individuals and society. One extensive study of the burden of chronic illnesses in the United States for 1996 found that major depression was the leading cause of lost disability-adjusted life-years (DALYs) for people age 25 to 44 (Michaud et al., 2006). Another study of a nationally representative sample of people age 18 and older investigated the association between life role disability in the previous 30 days and 30 different chronic illnesses. Musculoskeletal illnesses and depression had the largest effects on disability of any of the other illnesses (Merikangas et al., 2007). Depression is also a frequent complicating factor for many other chronic illnesses. It frequently accompanies such illnesses as diabetes, disabling osteoarthritis, and cognitive impairment. One study found that 71 percent of Medicare recipients with depression have four or more other chronic illnesses (Wolff and Boult, 2005).
Multiple studies and meta-analyses have found that collaborative care—including depression screening, assessment, enhanced patient education, use of allied health professionals to provide close follow-up, a consultant psychiatrist as backup, and stepped-care treatment approaches with incremental increases in treatment for people with persistent symptoms—is effective in reducing depression and increasing function (Gilbody et al., 2006; Katon et al., 2010). Screening for depression is recommended by the U.S. Preventive Services Task Force (USPSTF, 2009).
Despite effective management options, few physician organizations use evidence-based programs for patients with depression. One study of 1,040 physician organizations found that only 29 (3.2 percent) used four effective organized care management processes for patients with depression (Casalino et al., 2003).
Given the overlap between depression and MCCs, other interventions that are complex approaches to integrating community and clinical resources may be considered. One systematic review and meta-analysis of 89 randomized controlled trials of community-based complex interventions found reductions in nursing home admissions (RR 0.87; 95 percent CI 0.83–0.90), risk of hospital admission (0.94; 0.91–0.97), and falls (0.90; 0.86–0.95) (Beswick et al., 2008). One randomized controlled trial, for example, examined the effect of geriatric care management, which included home-based care by a nurse practitioner and a social worker collaborating with a primary care physician and a geriatrics interdisciplinary team, on low-income people age 65 and older with MCCs. After 2 years, the study found significant improvements for patients receiving the intervention in four of eight SF-36 quality of life scales, including general health, vitality, social functioning, and mental health (Counsell et al., 2007).
The CDC’s National Center for Chronic Disease Preventions and Health Promotion, Division of Adult and Community Health recently published
a Public Health Action Plan to Integrate Mental Health Promotion and Mental Illness Prevention with Chronic Disease Prevention, 2011–2015 (CDC, 2011c). This plan recognizes the interconnection between chronic disease and mental health, including major depression, and outlines the goal to include the promotion of mental health as part of efforts to prevent chronic disease. The committee commissioned a paper by Wayne J. Katon on improving recognition and depression care in individuals with common chronic illnesses (see Appendix A).
Type 2 Diabetes
Diabetes mellitus is defined as a group of metabolic diseases characterized by hyperglycemia resulting from defects in insulin secretion from the pancreatic beta (β) cells; insulin action at the level of skeletal muscle, liver, and fat; or both (American Diabetes Association, 2010). It is estimated that 25.6 million, or 11.3 percent of adults age 20 and older in the United States, have diagnosed and undiagnosed diabetes, and 90 to 95 percent of diagnosed cases involve type 2 diabetes (CDC, 2011b). The prevalence of diabetes is similar in men and women (CDC, 2011b). Diabetes is a particular public health burden among the elderly: 26.9 percent of adults age 65 and older have diabetes (10.9 million individuals) (CDC, 2011b). And non-Hispanic blacks and Mexican Americans have twice the age- and sex-standardized prevalence of diagnosed diabetes compared with non-Hispanic whites (Cowie et al., 2009). Risk factors for type 2 diabetes include increasing age, obesity, physical inactivity, having a prior history of gestational diabetes, having hypertension or dyslipidemia, being a member of a high-risk racial/ethnic group (i.e., African, Hispanic, Asian, Native American, or Pacific Islander), or having a family history of type 2 diabetes, particularly in first-degree relatives (American Diabetes Association, 2010, 2011).
The onset of type 2 diabetes is often insidious and asymptomatic; a preclinical stage of prediabetes is defined as having impaired fasting glucose, impaired glucose tolerance, or a high risk hemoglobin A1c (HbA1c) value of 5.7 to 6.4 percent (American Diabetes Association, 2010). Approximately 35 percent of U.S. adults over age 20 have prediabetes, and 50 percent of elderly individuals have prediabetes (CDC, 2011b). Type 2 diabetes often develops with obesity, which induces insulin resistance. Although the β cell attempts to compensate for insulin resistance by secreting increasing amounts of insulin, this compensatory mechanism eventually fails with progressive β cell dysfunction, resulting in hyperglycemia and development of type 2 diabetes (American Diabetes Association, 2010). There is quite a bit of variability in the degree of β cell dysfunction along the spectrum of insulin resistance, which is why some patients with type 2 diabetes require more aggressive pharmacological intervention earlier than others. The resultant
hyperglycemia, if untreated, can lead to long-term complications, including microvascular complications (i.e., retinopathy, nephropathy, and peripheral and autonomic neuropathy) and macrovascular complications (i.e., coronary heart disease, cerebrovascular disease, and peripheral arterial disease). Diabetes is the leading cause of incident blindness in adults age 20 to 74 and the leading cause of end-stage renal disease (CDC, 2011b). Mild to severe nervous system damage occurs in 60 to 70 percent of individuals with diabetes and includes peripheral neuropathy, gastroparesis, and erectile dysfunction, among others (CDC, 2011b). Peripheral neuropathy is a major contributor to lower extremity amputation. Other complications include periodontal disease, increase in susceptibility to infectious diseases, decrease in functional status, and depression (CDC, 2011b).
There are several evidence-based therapies that can prevent development of complications. Intensive control of hyperglycemia, with a target HbA1c of 7 percent, has been shown to reduce the risk of microvascular complications for individuals with type 2 diabetes (UK Prospective Diabetes Study Group, 1998). Interventions to reduce hypertension have been shown to reduce the risk of both cardiovascular disease and retinopathy for people with type 2 diabetes; aggressive lowering of LDL cholesterol with HMG-CoA reductase inhibitors reduces the risk of cardiovascular disease, the leading cause of death among those with diabetes (American Diabetes Association, 2011). Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers have been shown to reduce the risk of incident nephropathy and progression to end-stage renal disease in type 2 diabetes (American Diabetes Association, 2011). For individuals with diabetes and macular edema or severe nonproliferative or high-risk proliferative diabetic retinopathy, laser photocoagulation therapy reduces the risk of vision loss (American Diabetes Association, 2011).
Diabetes is associated with limitations in physical functioning and the ability to perform ADLs (De Rekeneire et al., 2003; Gregg et al., 2000; Maty et al., 2004; Ryerson et al., 2003; Volpato et al., 2002). Potential mediators of this association include diabetes complications and comorbidities (Kalyani et al., 2010; Volpato et al., 2002), hyperglycemia (De Rekeneire et al., 2003; Kalyani et al., 2010), and depression (Egede, 2004; Volpato et al., 2002); however, in some populations, diabetes remains associated with functional disability even after accounting for these factors (Maty et al., 2004). Diabetes also carries a high patient self-management burden due to the need for daily self-monitoring of blood glucoses by patients on insulin therapy and/or oral diabetes medications, carbohydrate intake, medication administration, avoidance of hypoglycemia, and maintenance of an exercise routine.
Diabetes is also associated with reduced quality of life, particularly among individuals with multiple and/or severe complications (Rubin and
Peyrot, 1999). Patients with type 2 diabetes who are diet-controlled or whose hyperglycemia is managed with oral antidiabetic agents report better quality of life than those managed with insulin (Bradley et al., 2011; Rubin and Peyrot, 1999); however, individuals with type 2 diabetes poorly controlled on oral agents report improved quality of life following transition to insulin therapy (Bradley et al., 2011; Jennings et al., 1991; Wilson et al., 2004), a result probably related to improved glycemic control. Quality of life can also be influenced by the type of insulin used to treat hyperglycemia (Bradley and Gilbride, 2008; Bradley and Speight, 2002). However, the majority of pharmacological intervention studies that included clinical outcomes for type 2 diabetes have not concurrently measured quality of life outcomes. The United Kingdom Prospective Diabetes Study assessed a measure of health status rather than quality of life, so it remains unclear if quality of life was different between the intensive and conventional therapy treatment groups at the end of the study (Bradley et al., 2011). The PANORAMA Study in Europe will shed further light on the impact of various diabetes treatment regimens and glycemic control on patient-reported outcomes, including quality of life (Bradley et al., 2011). This observational study will recruit 5,000 individuals with type 2 diabetes in nine European countries and investigate the association between treatment regimens, levels of glycemia, quality of life, treatment satisfaction, fear of hypoglycemia, and health status (Bradley et al., 2011). Because pharmacological therapies that prevent diabetes complications are administered within the clinical health care system, future studies are needed to determine whether the interface between the health care and public health systems and community-based organizations can improve adherence to these treatments while also improving patient-reported outcomes.
Posttraumatic Disabling Conditions
Posttraumatic disabling conditions (PTDCs) are a diverse group of conditions with heterogeneous causes and outcomes that cumulatively can yield a substantial amount of short- and long-term morbidity, mortality, and permanent disability. It is very difficult to define the population rates of such conditions because they are not easy to define or detect in population surveys. Outcome studies generally come from institutional registries and may underestimate the total community burden of PTCDs, which have always been considered a group from a public health perspective. Although primary injury prevention for some types of community-acquired trauma has been a public health priority for many years, individual PTCDs are not as common as naturally occurring conditions. With a few general policy exceptions, such as the Americans with Disabilities Act, ameliorating the
chronic disability and reducing the secondary conditions associated with these disabilities has never been a public health priority.
As noted, PTCDs are extremely diverse. Knee meniscus injuries from many causes can lead to chronic degenerative arthritis years or decades after the injury or repair, although some early interventions can mitigate some of this adverse long-term outcome (Zafagnini et al., 2011). It is estimated that about 235,000 Americans are admitted to hospitals each year with nonfatal traumatic brain injuries (TBIs) (Corrigan et al., 2010). Long-term improvement with rehabilitation can be obtained for TBI patients with disordered consciousness, but this process can be prolonged over many years (Nakase-Richardson et al., 2011). Late deaths more than 30 days after hospital discharge from a variety of causes occur commonly among trauma patients (Claridge et al., 2010), suggesting substantial community experiences with posttraumatic states. Trauma patients who survive surgical intensive care units for more than 3 years have substantial long-term disability rates (Livingston et al., 2009). Severe burn injuries may lead to substantial disability and disfigurement, restricted movement, and long-term metabolic abnormalities (Jeschke et al., 2011). Falls and fractures are very common among older adults, leading to increased disability and joint replacement. For example, on the basis of emergency room visits, it was estimated that in excess of 1 million fall-related wrist and forearm injuries occurred over a 7-year period in the United States (Orces and Martinez, 2011). Taken together, a substantial segment of the U.S. population is living with the varying but sometimes severe consequences of a variety of traumatic events. There are many other important sources of trauma with long-term consequences, such as work and home implements and firearms. As other patient groups have done, posttrauma patients have organized to improve their circumstances (Bradford et al., 2011).
A clear public health recognition of the cumulative importance of posttrauma patients is worthy of consideration. There is an important need to create a public health taxonomy of PTDCs that encompasses commonalities and assesses long-term health outcomes, allowing for more precise population surveys and more effective population surveillance of the burdens of trauma. Also, as with other chronic illnesses, monitoring for secondary disease and dysfunctions associated with PTDCs and for a community-oriented research program that attempts to minimize long-term adverse outcomes and promote improved prevention could be valuable.
Schizophrenia
Schizophrenia is a severe, chronic, and disabling mental disorder. Individuals with schizophrenia often experience terrifying symptoms, such as auditory and visual hallucinations and illusions, or believing that other
people are reading their minds, controlling their thoughts, or plotting to harm them. These symptoms may leave them fearful and withdrawn. “Their speech and behavior can be so disorganized that they may be incomprehensible or frightening to others” (http://www.schizophrenia.com/family/sz.overview.htm).
About 1 percent of the Americans develop schizophrenia over their lifetime, affecting more than 2 million Americans in a given year. Although schizophrenia appears equally frequently among men and women, the onset of the illness is earlier in men, usually in the late teens or early twenties; women typically experience the onset of illness in their twenties to early thirties (http://www.schizophrenia.com/family/szfacts.htm).
Available treatments can relieve many symptoms, but most people with schizophrenia continue to experience some symptoms throughout their lives. Medication compliance with this population is difficult, and it has been estimated that no more than one in five individuals fully recovers (http://www.schizophrenia.com/family/sz.overview.htm). Homelessness also makes it difficult to provide consistent and effective treatment to many individuals with schizophrenia. It is estimated that 6 percent of homeless individuals have schizophrenia. Homeless individuals with schizophrenia may experience a worsening of their symptoms caused by the stress of living on the streets. Poor hygiene, lack of sleep, and the threat of violence may accelerate a person’s decline into psychosis (http://www.health.am/psy/more/homelessness-schizophrenia/).
Schizophrenia is considered the most chronic, disabling, and costly mental illness. The indirect excess cost due to unemployment is the chief contributor to overall schizophrenia excess annual costs. In 2002, the cost of schizophrenia in the United States was about $62.7 billion, including $22.8 billion in excess direct health care costs: $7.0 billion for outpatient services, $5.0 billion for drugs, $2.8 billion for inpatient services, and $8.0 billion for long-term care. The total direct non–health care excess costs, including living cost offsets, were estimated at about $7.6 billion, and the total indirect excess costs were estimated at about $32.4 billion (Wu et al., 2005).
The management and consequences of living with schizophrenia are numerous and difficult. For example, people who have schizophrenia abuse alcohol, illicit drugs, and nicotine more than the general population does (http://www.schizophrenia.com/family/sz.overview.htm). In addition to reducing the effect of antipsychotic treatment, substance abuse is a health hazard that places the schizophrenic person at increased risk for MCCs over time. About one-third of the excess mortality in schizophrenics is due to unnatural causes, such as suicide, whereas two-thirds is due to natural causes (Lawrence et al., 2010). The largest number of deaths is due to cardiovascular disease (Lawrence et al., 2010). Research has also shown that
persons with schizophrenia and substance abuse are also at an increased risk for committing a violent crime (Fazel et al., 2009).
The burden of schizophrenia on families and caregivers is significant. A shift toward a community-based approach to mental illness management and the increased role of family in the daily care of mentally ill persons has also had a psychosocial, physical, and financial impact on families. Caregiver burden associated with mental illness refers to the “negative responses that occur when caregivers assume unpaid and unanticipated responsibility for the person for whom they are caring who has a disabling mental health problem” (Schulze and Rössler, 2005). In addition, the concept of “burdens of care” involves “subtle but distressing notions such as shame, embarrassment, feelings of guilt and self-blame” (Awad and Voruganti, 2008). In the United States, 40 to 80 percent of persons with schizophrenia, depending on the subgroup, live with a relative or spouse (UNC Center for Excellence in Community Mental Health, [a]).
Vision and Hearing Loss
Visual and hearing losses are common disorders, especially among the elderly. In 2008, 15 percent of Americans age 18 and older were estimated to have hearing difficulty (without a hearing aid) and 11 percent had visual impairment (defined as trouble seeing, even with glasses or contact lenses) (NCHS, 2009). The rates of both hearing and vision problems increase with age, rising to 43 and 21 percent, respectively, among those age 75 and older (NCHS, 2009). Moreover, each of the four major eye diseases that cause visual impairment (cataract, age-related macular degeneration, glaucoma, and diabetic retinopathy) is more common with advancing age. Women are more likely to have vision problems than are men, but men are more likely than are women to have hearing problems (NCHS, 2009). Asian adults and black adults are less likely to have hearing difficulty than are white adults (NCHS, 2009). Poorer adults and those with Medicaid coverage are also more likely to have sensory problems than those who are wealthier or have private insurance or Medicare-only coverage (NCHS, 2009).
Subjectively reported or objectively measured visual impairment is predictive of decline in ADL and IADL function at 10 years and over 10 years (Reuben et al., 1999). Bilateral noncorrectable vision loss leads to dependence, nursing home placement, and worse emotional well-being (Horowitz, 2003; Vu et al., 2005). Noncorrectable unilateral visual loss is associated with increased risk of falling (Vu et al., 2005). Hearing loss has been associated with anxiety, social isolation, and depressive symptoms (NCOA, 1999). Self-reported or objectively measured hearing impairment predicts impairment in walking a quarter-mile, climbing up and down steps, and performing heavy chores (e.g., yard work, washing windows)
(Reuben et al., 1999). Combined objectively measured hearing and visual impairment has the highest risk (relative risk 8.03) for subsequent ADL impairment (Reuben et al., 1999).
Sensory impairment also results in a high economic burden. The annual cost of visual impairment and blindness was estimated in 2002 at $5.5 billion (Frick et al., 2007), and a cohort-survival study estimated that $4.6 billion will be spent over the lifetime of persons who acquired their impairment in 1998 (Mohr et al., 2000).
The treatment of sensory impairment depends on the cause. For visual impairment, cataracts can be cured by surgery with intraocular lens implantation. The other eye disorders are managed with a variety of medications and surgical procedures aimed at preventing further visual impairment. In addition, visual assistive devices, including eyeglasses, electronic video magnifiers, spectacle-mounted telescopes for distance vision, and closed-circuit television to enlarge text, are useful, as well as technologies like talking books. Community-based organizations (e.g., Braille Institute, the Lighthouse) provide direct services in addition to counseling and adaptive equipment. Insurance coverage for these services and equipment is variable.
For hearing impairment, the primary treatment approach is amplification, either by hearing aids or assistive listening devices (e.g., devices that have a microphone and headphones that facilitate hearing). In addition, text telephones (TTY) and telephone devices for the deaf (TDD) are often available at no cost to hearing-impaired persons. Other technology, such as FM loop systems, can be used for groups of people who have FM receivers or telecoil switches in their hearing aids. Infrared group listening devices can also be useful. Medicare does not cover amplification devices, including hearing aids. Selected groups of hearing-impaired persons may benefit from cochlear implants. A major barrier to treatment for persons with hearing impairment is denial of the problem or its importance by affected persons (NCOA, 1999).
A North Carolina study identified the common barriers to hearing-impaired persons accessing basic services as lack of access to communication, lack of understanding of the indicators and consequences of hearing loss, insufficient resources to effectively advocate for themselves in obtaining services, and lack of knowledge of existing resources available (North Carolina Department of Health and Human Services, 2009). There are community-based organizations and groups that facilitate living with hearing loss (e.g., Better Hearing Institute, Association of Late-Deafened Adults, Hearing Loss Association of America). Many states sponsor programs for hearing loss that offer various type of counseling and educational services (North Carolina Department of Health and Human Services, 2009).
Vision impairment is associated with considerable caregiver burden. A French study found that a quarter of caregivers of persons with blindness
could not go out for an entire day and better than half reported that the caregiving burden affected their physical and emotional health and mental welfare and that they needed to modify their work (Brézin et al., 2005). A Japanese study found hearing impairment to be associated with increased caregiver burden (Kuzuya and Hirakawa, 2009).
WHO ARE THE PEOPLE WITH CHRONIC ILLNESSES?
Age and Chronic Illnesses
The relationship between aging and chronic illness is complex and variable. Differences between older and younger persons must be recognized and considered in a population-based approach to living well with chronic illness.
First, with aging, chronic diseases become more prevalent: 43 percent of Medicare beneficiaries have three or more illnesses (IOM, 2009) and 23 percent have more than five (Anderson, 2005). Moreover, the percentage of persons with MCCs rises with age. These multiple illnesses often require different and sometimes conflicting treatments (Boyd et al., 2005; Tinetti et al., 2004). As the number of medications used to treat multiple illnesses increases, the risk of adverse effects also increases (Agostini et al., 2004).
Second, the type, severity, number, and particular combination of chronic illnesses among the elderly vary. Older persons may accumulate conditions that have become inactive. For example, an 85-year-old woman with breast cancer, coronary artery disease, and chronic kidney disease may have had a mastectomy 20 years ago, had a coronary artery bypass 15 years ago, and have no restrictions from kidney disease other than dose adjustment for kidney function. Conversely, another woman with the same diagnoses may be receiving chemotherapy, taking six cardiac medications for her heart disease, and receiving dialysis. Some chronic illnesses (e.g., dementia, osteoarthritis, hypertension, sensory impairments) occur almost exclusively or at much higher prevalence among older persons. Dementia, which may affect up to 43 percent of persons age 85 and older (Alzheimer’s Association, 2011a), is a particularly devastating disease because the protean manifestations affect the ability to maintain function and manage other chronic illnesses. As this disorder progresses, it predominates with needs that often overwhelm other preexisting and new chronic illnesses.
Third, the interaction of aging and chronic illness must be considered. The physiological functional reserve decreases with aging, often referred to as “homeostenosis.” As a result, the ability to compensate for illness processes is usually lower than for younger persons. A construct termed “allostatic load” has been used to describe the burden of multiple chronic subclinical disturbances that are more common in older persons yet have
prognostic importance (Seeman et al., 2001). At a clinical level, some normal aspects of aging (e.g., changes in vision, dexterity) may affect the ability to manage chronic illnesses. Consider the elderly person with diabetes and presbyopia and impaired visual contrast sensitivity who must measure and administer variable doses of insulin. Similarly, the age-related decrease in renal function and increase in the percentage of body fat affect the dosing, toxicity, and distribution of medications.
Fourth, the interaction among socioeconomic factors and chronic illness must be recognized. With aging, a variety of social supports change. For example, older persons may retire, become widowed, cease driving, move to different housing. Any of these may affect the ability to live independently or cope with chronic illness. Many of the functional capabilities that younger persons, even with those with chronic illness, take for granted, are gone or are in jeopardy of being lost. For example, persons with postpolio syndrome who have adapted to their impairments may find that, with aging, these adaptive responses are no longer sufficient to maintain function. Similarly, the loss of a spouse who has been a caregiver for a person with Alzheimer’s disease may precipitate a crisis, even though the person’s clinical status has not changed.
Fifth, prognosis and personal goals change with aging. Even in the absence of chronic illness and disability, life expectancy declines with aging (Keeler et al., 2010). Limited life expectancy may affect choices in managing chronic illness as well as the goals of care. These goals may differ considerably from those of younger persons with chronic illnesses, who may have a much longer life expectancy. Older persons’ goals may relate to a functional or health state (e.g., being able to walk independently), symptom control (e.g., control of pain or dyspnea), living situation (e.g., remaining in one’s home), or short-term survival (e.g., living long enough to reach a personal milestone, such as a family member’s wedding) rather than long-term survival. Sometimes an older person’s physician believes that a better outcome is possible but the patient declines to follow the recommended route (e.g., physical therapy to regain mobility). In addition, patient preferences for specific treatments may lead to care that is not the best evidence-based option “(e.g., using pads to manage urinary incontinence even though effective behavioral and pharmacological therapy is available)” (Reuben, 2009).
Demographic Disparities
Health Disparities and Living Well with Chronic Illness
As noted in Chapter 1, the health of Americans is better now than at any other time in history. As compared with those living in 1900, Americans today are “healthier, live longer, and enjoy lives that are less likely
to be marked by injuries, ill health, or premature death,” according to an earlier IOM report (IOM, 2003a). However, these gains are not shared with all members of society. Health disparities exist and persist. Race, as well as income, account for the pronounced disparities in care and therefore the disparities in health status between white and minority Americans (Watson, 2003). Recent reports on health disparities document the relatively poor health of African Americans, American Indians, Native Hawaiians, and Latinos, and other underrepresented groups when compared with white Americans (IOM, 2003b). Not only are racial and ethnic groups often less healthy; they also tend to have shorter life expectancies, higher rates of chronic illnessess, worse outcomes when diagnosed with an illness, and less access to quality health care (IOM, 2003b). In 2003 the IOM produced the report entitled Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care, which highlighted how within racial and ethnic groups in the United States there are remarkably consistent racial and ethnic disparities across a range of illnesses and health care services (IOM, 2003b).
CDC defines health disparities as significant differences between one population and another that can occur by gender, race or ethnicity, education or income, disability, geographic location, or sexual orientation (CDC’s Office of Minority Health and Health Disparities, [a]). For racial and ethnic minorities, these disparities exist in a number of illnesses, including cardiovascular disease, HIV/AIDS, hypertension, diabetes, and mental illness (CDC’s Office of Minority Health and Health Disparities, [a]). The severity of health disparities among specific groups becomes stark when the total U.S. population is segmented by race and ethnicity and about a third of the U.S. population consists of minorities impacted by disparate health (CDC’s Office of Minority Health and Health Disparities, [a]; Center for Prevention and Health Services, 2009).
Health behaviors and lifestyles greatly contribute to chronic illness and health disparities. Research has demonstrated that a myriad of sources and complexities account for these disparities. However, socioeconomic status (SES), class status, lack of health insurance, and the quality of care different racial and ethnic groups receive are also powerful factors that impact the ability for people to make healthy decisions and live full and engaged lives, living well despite their chronic illness.
Specifically, SES is highly related to the presence and persistence of health disparities. Individuals with “lower socioeconomic status [SES] die earlier and have more disabilities than those with higher [SES]” (Schroeder, 2007). And the most extreme disparities in health occur among the impoverished, including individuals who are impoverished because of their health-related problems, as well as individuals whose health has suffered as a result of poverty. Environmental factors, such as lead paint, water and air pollution, dangerous neighborhoods, lack of outlets for physical activity, as
well as other health-compromising factors, contribute to single and MCCs for individuals with lower SES (Schroeder, 2007).
Class is also highly related to the prevalence of chronic illness among in racial and ethnic groups. Similar to SES in a stepwise pattern from lowest to highest, class is defined by income, total wealth, education, employment, and residential neighborhood (Schroeder, 2007). The class gradient in health means that people in the lower class gradient are more likely to practice unhealthy behaviors, partly due to inadequate grocery stores, constrained conditions to exercise (Schroeder, 2007), and the inability to secure the resources needed to support healthy living or manage chronic illnesses. Class is a determinant of the nation’s health and an important factor for public health leaders to consider in population-based efforts to help individuals living with chronic illnesses.
Lack of health insurance is a barrier to access to quality care, is a serious determinant of health, and contributes to disparities in health. As Box 2-2 shows, racial and ethnic minorities are much more likely to be uninsured than white Americans.
Inequalities in quality of care also exist and also contribute to poor health outcomes. A large body of published research revealed that racial and ethnic minorities and/or poor disadvantaged patients receive inadequate quality care (IOM, 2003b). And the differences in health care quality do not disappear when controlled for SES differences or health insurance, which means that disparities across the range of chronic illnesses and health care services cannot be attributed to economic status or access to care alone.
The concept of living well adopted by the committee in this report—the best achievable state of health that encompasses all dimensions of physical, mental, and social well-being—may be heavily clouded in the minds of individuals with single or MCCs who live in communities where complex and social inequities are deeply rooted. The committee thinks that public health action to lead and enhance efforts to help racial and ethnic groups
BOX 2-2
Access to Care: Uninsured in 2010
• 11.7 percent of Whites, not Hispanic
• 18.1 percent of Asian Americans
• 20.8 percent of African American
• 30.7 percent of Latinos
SOURCE: U.S. Census Bureau, 2011.
with chronic illness live better with better health outcomes is important and achievable.
Health Literacy
Health literacy includes general comprehension of the human body, healthy behaviors, and the workings of the health care system (HHS, 2010). It is a complex construct that measures an individual’s ability to function effectively in the health care system (Berkman et al., 2011). Today’s health care system requires a particularly sophisticated level of understanding from individuals to receive needed care, and lower health literacy is commonly found among minorities, the elderly, and patients with chronic illnesses (Schillinger et al., 2002).
The National Assessment of Adult Literacy from 2003 categorized 14 percent of adults as “below basic” in health literacy (NCES, 2006). Between socioeconomic and racial and ethnic groups, only 9 percent of white respondents were categorized as below basic compared with 24 percent of African Americans and 41 percent of Hispanics (NCES, 2006). In addition, 3 percent of respondents with a college degree and some graduate study and 49 percent of respondents with less than a high school education placed at the below-basic level (NCES, 2006). A systematic review conducted by Berkman et al. (2011) found that “low health literacy was consistently associated with more hospitalizations; greater use of emergency care; lower receipt of mammography screening and influenza vaccine; poorer ability to demonstrate taking medications appropriately; poorer ability to interpret labels and health messages; and, among elderly persons, poorer overall health status and higher mortality rates.” Previous studies among indigent and Medicare patient populations have shown that older individuals have lower health literacy (Gazmararian et al., 2003). One study found that an estimated 81 percent of English-speaking patients over age 60 treated at a public hospital had inadequate levels of health literacy (Gazmararian et al., 2003). A separate study found that all low-income, community-dwelling with adults between the ages of 60 and 94 possessed reading skills averaging at the fifth-grade level and one-fourth of the adults admitted having trouble comprehending written information from physicians (Gazmararian et al., 2003).
Individuals with poor health literacy are more likely to report having a chronic illness. In a population-based cross-sectional study of 2,923 Medicare managed care enrollees in four U.S. cities, about 22.2 percent had “inadequate” health literacy and about 11.3 percent had “marginal” health literacy (Wolf et al., 2005). In statistically significant unadjusted analyses, people with inadequate health literacy had more self-reported cases of diabetes (18.7 versus 12.8 percent, p < 0.001), heart failure (6.1 versus
3.8 percent, p = 0.05), and arthritis (57.3 versus 50.1 percent, p = 0.01) than people with adequate health literacy had. Furthermore, individuals with inadequate health literacy were more likely to report greater difficulty in completing daily activities and fewer accomplishments due to worse physical health and higher levels of pain (Wolf et al., 2005). Even after adjusting for higher prevalence of chronic illness, individuals with inadequate health literacy had worse physical and mental health (Wolf et al., 2005).
Although those with poor health literacy are more likely to report having a chronic illness, the reverse is also true. Individuals with poor health literacy often know less about any chronic illness they might have (Gazmararian et al., 2003). Previous studies have documented a weaker base of chronic illness knowledge among those with asthma, diabetes, and hypertension (Gazmararian et al., 2003). In a more recent study, Gazmararian et al. (2003) surveyed 653 newly enrolled Medicare patients age 65 and older with one or more chronic illnesses to see how much these patients knew about their own chronic illness(es). Of those surveyed, 24 percent had inadequate and 12 percent had marginal health literacy. Analysis reinforced previous study findings of higher chronic illness knowledge among those with higher health literacy (Gazmararian et al., 2003).
Studies such as the one conducted by Gazmarmarian et al. (2003) point to serious repercussions among those with chronic illnesses and lower health literacy. For example, congestive heart failure (CHF) is a common reason for hospitalizations among those age 65 and older; however, many cases for rehospitalization are preventable with proper CHF management, knowledge, and skills, which are possessed by those with higher levels of health literacy (Baker et al., 2002). Similarly, patients with type 2 diabetes and inadequate health literacy report weaker glycemic control and higher prevalence of retinopathy (Schillinger et al., 2002). Findings like this suggest that inadequate health literacy disproportionately contributes to the burden experienced by those with type 2 diabetes from disadvantaged populations (Schillinger et al., 2002).
With the passage of the Affordable Care Act, millions of new patients will gradually flood the health care system, receiving treatment for previously unaddressed or undiagnosed chronic illnesses. However, with many patients possessing weak health literacy, most health efforts will be in vain, as health literacy has a proven record with poorer health outcomes. To reverse poor health literacy and improve health outcomes among patients throughout the United States, and particularly disadvantaged populations, the 2010 National Action Plan to Improve Health Literacy was developed. It has seven goals: (1) “develop and disseminate health and safety information that is accurate, accessible, and actionable”; (2) “promote changes in the health care system that improve health information, communication, informed decision-making, and access to health services”; (3) “incorporate
accurate, standards-based, and developmentally appropriate health and science information and curricula in child care and education through the university level”; (4) “support and expand local efforts to provide adult education, English language instruction, and culturally and linguistically appropriate health information services in the community”; (5) “build partnerships, develop guidance, and change policies”; (6) “increase basic research and the development, implementation, and evaluation of practices and interventions to improve health literacy”; and (7) “increase the dissemination and use of evidence-based health literacy practices and interventions.” To translate these goals and strategies into action and effectively promote higher levels of health literacy, these actions need to be multidisciplinary, evidence-based, and evaluated, and to involve the communities and individuals most affected.
Primary and Secondary Prevention
Elsewhere in the chapter the problems of MCCs (comorbidity) are well characterized in terms of their impact and importance. Although some secondary conditions are related to progressive primary illnesses (e.g., falls and fractures associated with disabling progressive neurological illnesses), persons with chronic illnesses are also subject to additional, unrelated illnesses by virtue of aging, personal risk profiles, and perhaps other biological vulnerabilities associated with the original illness (e.g., genetic risks of multiple cancer syndromes, tobacco exposures). Although there are authoritative sources of effective primary and secondary preventive interventions for persons in clinical practice (U.S. Preventive Services Task Force) and in the community (The Community Guide), neither of these resources systematically or comprehensively addresses these important interventions for persons with overt chronic illnesses. In fact, there is good evidence that quality primary care, including preventive services, may be deficient among those with mental and disabling illnesses (Havercamp et al., 2004; Mitchell et al., 2009; Reichard et al., 2011).
Indeed, one can find expert opinion, clinical recommendations or a true evidence base related to certain primary preventive interventions (e.g., influenza vaccine for certain risk groups) for primary prevention and screening and screening in the disease-specific literature, but, after thorough literature review, the committee thinks that there are major gaps in research-based recommendations for routine preventive activities for those with common and important chronic diseases. Although there may be an abiding logic in many instances to extend preventive recommendations intended for healthy persons to those with chronic illnesses (e.g., smoking cessation, hypertension control), an enhanced research and systematic review approach to this problem is clearly indicated.
BOX 2-3
Primary Preventive Interventions
• Vaccines for adults with chronic illnesses, as recommended by the Advisory Committee on Immunization Practices, including tetanus, diphtheria, and acellular pertussis vaccine; pneumonia vaccine; zoster vaccine; and the newly developed high-dose influenza vaccine, as clinically indicated (Chen et al., 2011).
• Special food safety and food preparation instructions for persons with any conditions associated with immune-compromised states or treatments, such as occur in cancer patients (USDA, 2006).
• Education to recognize and seek care immediately when the symptoms of stroke appear in an individual (American Stroke Association, [a]).
• Personal and family monitoring of environmental alerts, such as extreme heat, cold, or air pollution conditions, all associated with increased morbidity and mortality risk among those with chronic illnesses (Wen et al., 2009).
• Preparedness education for persons with chronic illnesses when natural disasters occur. For example, maintaining electrical devices that are needed for illness management when power outages occur (Khan, 2011).
Emphasis on Primary Preventive Interventions
In addition to the need to determine the needs and outcomes of general preventive interventions for persons with chronic illnesses, there are several such interventions that may require special emphasis—interventions that have been given little attention. The level of evidence for most of these varies, but in general there have been enough studies to raise these interventions to the level of consideration for public health policy. Some of them have been assessed only in outbreak situations, and some are not subject to experimental trial interventions per se, except in the situation in which techniques for behavior modification are indicated. The list in Box 2-3 is not exhaustive, but the committee thinks these preventive efforts need some further consideration for dissemination activities that target persons with chronic illness.
Recommendations 1–4 are the result of the committee’s efforts to answer statement of task question 2—which chronic diseases should be the focus of public health efforts to reduce disability and improve functioning and quality of life?
Recommendation 1
The committee recommends that CDC select a variety of illnesses for special consideration based on a planning process that first and foremost emphasizes the inclusion of chronic illnesses with cross-cutting clinical, functional, and social implications that impact the individuals who live with them. In addition, the committee suggests that other important criteria for illness selection include
• nonduplication with major illnesses for which public health programs have already been developed (e.g. cardiovascular disease, stroke);
• those with important implications for various models of chronic illness care, such as public health, health system, and self-care programs, especially when effective health service interventions are possible;
• variation in organ systems and long-term clinical manifestations and outcomes; and
• those for which the effective public health preventive interventions are either most feasible or at least the subject of promising research.
Recommendation 2
Although research has attempted to characterize MCCs, the complexity of single chronic illnesses over time has not allowed for MCC taxonomies that will be easily applicable to public health control of chronic diseases. Thus, the committee recommends that CDC:
1. Continue to review the scientific literature to monitor for potential MCC taxonomies that are useful for planning, executing, and evaluating disease control programs of MCC occurrences.
2. Explore surveillance techniques that are more likely to capture MCCs effectively. This should include counting not merely the co-occurrence of diseases and conditions but also the order of occurrence and the impact on quality of life and personal function.
3. Emphasize MCC prevention by selecting for execution and evaluation one or more exploratory public health interventions aimed at preventing or altering the course of new disease occurrences in patients with MCCs or who are at risk for them. This might include established approaches, such as tobacco control or experimental approaches, such as metabolic or genetic screening.
4. Increase demonstration programs for chronic disease control that cut across specific diseases or MCCs and emphasize mitigating the secondary consequences of a variety of chronic conditions, such as falls, immobility, sleep disorders, and depression.
Recommendation 3
The committee recommends that the secretary of HHS support the states in developing comprehensive population-based strategic plans with specific goals, objectives, actions, time frames, and resources that focus on the management of chronic illness among their residents, including community-based efforts to address the health and social needs of people living with chronic illness and experiencing disparities in health outcomes. Such strategic plans should also include steps to collaborate with community-based organizations, the health care delivery system, employers and businesses, the media, and the academic community to improve living well for all residents with chronic illness, including those experiencing disparities in health outcomes.
Recommendation 4
The committee recommends that, in addition to addressing individual illnesses in the community, all relevant federal and state agencies charged with public health and community approaches to control chronic illness, to the extent feasible, extend surveillance, evaluation, and mitigation programs to the widest possible range of chronic illnesses. This approach recognizes the commonality of important health, functional, and social outcomes for the population of individuals who live with different chronic illnesses.
Finally, the committee offers a fifth recommendation to answer the question what is the role of primary prevention (for those at highest risk) secondary, and tertiary prevention of chronic disease in reducing or minimizing life impacts?
Recommendation 5
The committee recommends that the federal health and related agencies that create and promulgate guidelines for general and community and clinical preventive services evaluate the effectiveness of these services for persons with chronic illness, and specifically catalog and disseminate these guidelines to the public health and health care organizations that implement them.
CHRONIC ILLNESS AND THE NATION’S
HEALTH AND ECONOMIC WELL-BEING
Chronic illness imposes very considerable costs on society. This is due to many factors, including their high—and, in many cases, apparently increasing—prevalence; the aging of the population; advances in treatment that help maintain many individuals; their occurrence across the life course, despite being somewhat stereotypically associated with older ages; and the highly disabling nature of many chronic illnessess, especially when inadequately treated.
In Chapter 1, the committee mentions a number of different methods for quantifying the consequences of chronic illness at a population level, including methods for assessing disability and premature mortality; “direct” costs of medical care and other services provided to prevent and/or treat chronic illness; and “indirect” costs of chronic illness, such as reduced labor output and other consequences that lie beyond the health care system. Such methods can and in many cases have been used to estimate the consequences associated with particular illnesses or categories of illness. Thus, it might be natural to ask what such methods explain in terms of which consequences of chronic illness—or even which specific chronic illnesses—are most important for the nation’s health and economic well-being. In this section, we provide some conceptual discussion of this issue, from a national population perspective. Earlier in this chapter, the committee provides additional details on the health, economic, and other consequences of chronic illnesses at a more “micro” level, from the perspective of the people who have such illnessess and others in their communities.
Most fundamentally, chronic illnesses can reduce the quality of life of the people who live with them, via the symptoms and dysfunctions they cause. In economic terms, one manifestation of this is that chronic illness degrades society’s productive capacity by reducing people’s labor output, with people withdrawing from the labor market entirely due to poor health, shifting from full-time to part-time work and/or missing work periodically, accumulating less “human capital” (i.e., knowledge and skills), and being less effective at work (“presenteeism”). At the individual level, this may be reflected in lower earnings and other negative consequences among the people who have the chronic illness(es). At the societal level, a given person’s reduced productivity may also reduce the productivity of others, such as in teamwork settings, and—very importantly for many chronic illnesses—via informal caregiving. The disease burden borne by people who would be outside the formal labor force in any case, such as retired people, is also important to consider. While lost labor earnings are irrelevant, retirees’ potential contributions to society are potentially large and not limited to their labor market participation. Indeed, most generally, a person’s suffering—or
premature mortality—has negative consequences for the person’s family, friends, and others. In all these ways, potentially preventable negative consequences of illness represent an opportunity cost to society.
These costs related to work and retirement described above generally fall in the category of “indirect” costs of illness. There are, of course, also very considerable “direct” costs associated with chronic illness—that is, the costs of health care per se—outpatient and inpatient treatment, diagnostic tests and other ancillary services, prescription and nonprescription pharmaceuticals, medical devices, therapy/rehabilitation services, and so on, as well as public health initiatives focused on primary or secondary prevention. Direct costs represent an opportunity cost for the people or institutions (e.g., insurers, employers, taxpayers) who pay for the services, in the sense that most health care is an “intermediate good” that is consumed not for its own sake but because of its (expected) effect on health; without a particular disease burden, these resources could be used for other purposes. However, direct costs also represent income/earnings for the people or institutions providing the care and are thus not entirely a deadweight loss. Despite general skepticism about the sustainability of the nation’s direct health care spending—which has risen in absolute terms and as a percentage of national income throughout recent decades (Kaiser Family Foundation, 2010)—there is no objective standard for how much health care spending is too much. Still, the direct costs associated with chronic illness have many adverse societal consequences, including that they undermine public and private health insurance programs.
It is important to recognize that there is a kind of reciprocal relationship between direct and indirect costs. For example, public health investments are specifically intended to prevent illness, which both promotes well-being and reduces the need for health care services. Thus, up-front costs of effective public health interventions can raise direct costs while being implemented but decrease indirect costs via successful disease prevention. At the same time, clinical health care services are, in no small part, intended to preserve or restore well-being, including work and social functioning and the ability to live independently. Health care can also raise the direct costs of an illness while decreasing the indirect costs. In contrast, poor or restricted access to effective preventive or curative services can lower—or increase—direct costs while increasing indirect costs, sometimes drastically. As a result, one can’t simply add direct and indirect costs for particular diseases to generate a “total” cost to society of those diseases, and the methods for determining the costs of illness are thus extremely complex. The relevant question is what would happen to the disease burden associated with a given condition if direct costs for that condition were higher, lower, or had a different composition than under the status quo.
In this sense, the most important consequences of chronic illness are
those that could be prevented efficiently. Information on current disease burden—whether in the form of direct and/or indirect costs—is not by itself sufficient information to prioritize new investments in prevention or treatment, nor in research and development. At an economic level, new spending should be relatively cost-effective, in terms of yielding as large a benefit as possible for a given cost (or, for research and development, as large an expected benefit as possible, since the outcome of such efforts is uncertain in general). There may also be ethical or other reasons to prioritize prevention and treatment of particular diseases, or for particular population subgroups, beyond their cumulative burden of illness or even the cost-effectiveness of intervention.
In Chapter 1, the committee referenced a body of research that has estimated direct health care costs in the United States overall (i.e., the national health accounts [CMS, 2009]) and for certain chronic diseases or disease categories, as well as a complementary literature that has examined indirect costs attributed to certain diseases or disease categories. In principle, such evidence could support identifying diseases with the largest economic burden in particular categories, and/or the categories of cost that are most salient across chronic diseases overall. In practice, however, we think that the available evidence is currently inadequate to support this in any robust way.
For example, while the U.S. national health accounts apply a consistent methodology across the spectrum of health and health care, these accounts cannot currently be broken out by diseases or disease category (with the exception of mental and substance use disorders), as noted in Chapter 1. Also, of particular relevance for this report, the U.S. national health accounts do not fully capture public health interventions that may be relevant for preventing or otherwise mitigating chronic disease. They do count direct costs of “publicly provided health services such as epidemiological surveillance, inoculations, immunization/vaccination services, disease prevention programs, the operation of public health laboratories, and other such functions” (CMS, 2009, p. 26). However, the committee could not determine to what extent the national health accounts capture spending on health promotion and disease prevention initiatives that are not delivered directly to individuals, such as disability-friendly urban design. Also, they specifically exclude “government spending for public works, environmental functions (air and water pollution abatement, sanitation and sewage treatment, water supplies, and so on)” (CMS, 2009, p. 26), although this includes some core aspects of public health that are intended to—and in practice do—mitigate the societal burden of many chronic diseases.
In terms of assessing the costs of specific diseases or disease categories, the cost of illness literature consists of many distinct studies of direct and—in our estimation, less commonly—indirect costs. With important exceptions, such as the Milken Institute study mentioned in Chapter 1 that
assessed costs in five major categories of illness, most condition-specific studies have been conducted independently of each other. In practice, the studies in this literature have generally been different in terms of their data sources, scope of assessed costs, period of assessment, and other aspects of methodology. This makes it difficult or impossible to compare findings across different diseases in a consistent way, even for those diseases for which data are available. The committee also notes that some studies in this literature have been sponsored and/or conducted by entities with a stake in the outcome.
Another issue with the available literature on direct and indirect costs of particular chronic diseases is that a given cost may be counted multiple times across different studies, for example, because of difficulty attributing particular direct or indirect costs to a given disease. This is a risk even across studies that have used approximately equivalent methodology. Such double-counting may particularly affect accounting for the direct costs of public health interventions, which may target multiple diseases simultaneously, or even target no condition per se but affect rates and outcomes for multiple conditions. Finally, as is generally true of all health indicators, data on direct and indirect costs at a national level may mask considerable variation across subnational areas and/or population subgroups.
There are a number of ways to improve the quality and utility of information on the economic burdens of chronic disease, and—importantly—on opportunities to prevent or reduce them. For example, disease-specific national health accounts, as proposed by Rosen and Cutler (2009), could provide useful new information by illuminating not only the total direct costs attributable to particular diseases but also the current composition of those costs across types of service. More comprehensive capture of public health programs that encompass estimates of direct costs, including such programs that focus on communities and interventions that address varied diseases as well as individuals with MCCs rather than individuals, including those that may affect many different chronic (and acute) illnesses, would also be valuable. This seems relatively straightforward in the overall national health accounts, in which attribution to specific diseases is not required, but even disease-specific estimates should explicitly address the role of general/broad public health and other interventions that affect the disease(s) of interest.
Improving population health surveillance systems at the national and the subnational level would contribute substantially to the ability to assess direct and especially indirect costs of illness along with other measures of disease burden and health status. It would also be likely to inform the development and targeting of new disease prevention and treatment programs and aid in assessing the potential costs and benefits of investments in such programs. Perhaps most valuable, however, would be a systematic effort to assess not only the burdens associated with particular diseases but also
the opportunity—and opportunity costs—of potential investments in their prevention and treatment. In this context, we endorse the concept—if not necessarily the specific methods or substantive findings—of efforts of this type conducted in other settings. For instance, the Disease Control Priorities Project (DCPP, [a]) examined a wide range of health problems affecting developing countries (including some consideration of chronic diseases). The Copenhagen Consensus Center (Copenhagen Consensus Center, [a]) has conducted analogous research on a wide range of health and nonhealth issues, including a recent program to identify investment priorities in HIV/AIDS prevention. And the new Center for Medicare and Medicaid Innovation (Center for Medicare and Medicaid Innovation, [a]) created as part of the Affordable Care Act of 2010, is specifically seeking to identify interventions to address the so-called triple aim of improving the health of the population; enhancing the patient experience of care (including quality, access, and reliability); and reducing, or at least controlling, the per capita cost of care (Institute for Healthcare Improvement, [a]). The committee thinks that a similar approach could be applied to identify high-priority opportunities to improve the lives of people living with chronic illness.
The statement of task asks the committee to consider what consequences of chronic disease are most important to the nation’s health and well-being.
Recommendation 6
The committee recommends that CDC support the greater use of new and emerging economic methods, as well as those currently in use, in making policy decisions that will promote living well with chronic illnesses, including
1. those with greater use of cost-effectiveness techniques;
2. more exploitation of methods used in determining national health accounts, but for specific and important chronic illnesses with long-term outcomes;
3. enhanced consideration of opportunity costs for various program decisions; and
4. those with a greater focus on economic evaluation of interventions that involve MCCs and cut across a variety of community settings.
AAOS (American Academy of Orthopaedic Surgeons). 2008. Chapter 4. Arthritis and related conditions. In The Burden of Musculoskeletal Diseases in the United States: Prevalence, Societal and Economic Cost. Pp. 75–102. http://www.boneandjointburden.org/pdfs/BMUS_chpt4_arthritis.pdf (accessed October 20, 2011).
Agostini, J.V., L. Han, and M.E. Tinetti. 2004. The relationship between number of medications and weight loss or impaired balance in older adults. Journal of the American Geriatrics Society 52(10):1719–1723.
Alzheimer’s Association. 2006. Early-Onset Dementia. A National Challenge, a Future Crisis. Washington, DC: Alzheimer’s Association. http://www.alz.org/national/documents/report_earlyonset_summary.pdf (accessed October 5, 2011).
Alzheimer’s Association. 2011a. Alzheimer’s disease facts and figures. Alzheimer’s and Dementia 7(2). http://www.alz.org/downloads/Facts_Figures_2011.pdf (accessed October 5, 2011).
Alzheimer’s Association. 2011b. California’s Alzheimer’s Statistics. http://www.alz.org/documents_custom/Facts_2011/ALZ_CA.pdf?type=interior_map&facts=undefined&facts=facts (accessed October 5, 2011).
American Diabetes Association. 2010. Diagnosis and classification of diabetes mellitus. Diabetes Care 33(Supplemental 1):S62–S69.
American Diabetes Association. 2011. Standards of medical care in diabetes. Diabetes Care. 34(Suppl 1):S11–S61.
American Stroke Association (a). Warning Signs. http://www.strokeassociation.org/STROKEORG/WarningSigns/Warning-Signs_UCM_308528_SubHomePage.jsp (accessed September 2, 2011).
Anderson, G.F. 2005. Medicare and chronic conditions. New England Journal of Medicine 353(3):305–309.
Arrighi, H.M., P.J. Neumann, I.M. Lieberburg, and R.J. Townsend. 2010. Lethality of Alzheimer’s disease and its impact on nursing home placement. Alzheimer Disease and Associated Disorders 24(1):90–95.
Arthritis Foundation. 2008. Primer on Rheumatic Disease, 13th ed. Edited by J.H. Klippel, J.H. Stone, L.J. Crofford, and P.H. White. New York: Springer and Arthritis Foundation.
Arthritis Foundation and CDC (Centers for Disease Control and Prevention). 2010. A National Public Health Agenda for Osteoarthritis. http://www.cdc.gov/arthritis/docs/oaagenda.pdf (accessed October 15, 2011).
Awad, A.G., and L.N. Voruganti. 2008. The burden of schizophrenia on caregivers: A review. Pharmoeconomics 26(2):149–162.
Bair, M.J., R.L. Robinson, W. Katon, and K. Kroenke. 2003. Depression and pain comorbidity: A literature review. Archives of Internal Medicine 163(20):2433–2445.
Baker, D.W., J.A. Gazmararian, M.V. Williams, T. Scott, R.M. Parker, D. Green, J. Ren, and J. Peel. 2002. Functional health literacy and the risk of hospital admission among Medicare managed care enrollees. American Journal of Public Health 92(8):1278–1283.
Bayliss, E.A., J.F. Steiner, D.H. Fernald, L.A. Crane, and D.S. Main. 2003. Descriptions of barriers to self-care by persons with comorbid chronic diseases. Annals of Family Medicine 1(1):15–21.
Berkman, N.D., S.L. Sheridan, K.E. Donahue, D.J. Halpern, and K. Crotty. 2011. Low health literacy and health outcomes: An updated systematic review. Annals of Internal Medicine 155(2):97–107.
Beswick, A.D., K. Rees, P. Dieppe, S. Ayis, R. Gooberman-Hill, J. Horwood, and S. Ebrahim. 2008. Complex interventions to improve physical function and maintain independent living in elderly people: A systematic review and meta-analysis. Lancet 371(9614):725–735.
Biessels, G.J., I.J. Deary, and C.M. Ryan. 2008. Cognition and diabetes: A lifespan perspective. Lancet Neurology 7:184–190.
Bolen, J., L. Murphy, K. Greenlund, C.G. Helmick, J. Hootman, T.J. Brady, G. Langmaid, and N. Keenan. 2009. Arthritis as a potential barrier to physical activity among adults with heart disease—United States, 2005 and 2007. MMWR 58(7):165–169. http://www.cdc.gov/mmwr/PDF/wk/mm5807.pdf (accessed November 16, 2011).
Bower, J.E. 2008. Behavioral symptoms in patients with breast cancer and survivors. Journal of Clinical Oncology 26(5):768–777.
Boyd, C.M., J. Darer, C. Boult, L.P. Fried, L. Boult, and A.W. Wu. 2005. Clinical practice guidelines and quality of care for older patients with multiple comorbid diseases: implications for pay for performance. Journal of the American Medical Association 294(6):716–724.
Bradford, A.N., R.C. Castillo, A.R. Carlini, S.T. Wegener, H. Teter, Jr., and E.J. Mackenzie. 2011. The trauma survivors network: Survive. Connect. Rebuild. The Journal of Trauma 70:1557–1560.
Bradley, C., and C.J. Gilbride. 2008. Improving treatment satisfaction and other patient-reported outcomes in people with type 2 diabetes: The role of once-daily insulin glargine. Diabetes, Obesity, and Metabolism 10(Suppl 2):50–65.
Bradley, C., and J. Speight. 2002. Patient perceptions of diabetes and diabetes therapy: Assessing quality of life. Diabetes/Metabolism Research and Reviews 18(Suppl 3):S64–S69. Bradley, C., P. de Pablos-Velasco, K.G. Parhofer, E. Eschwège, L. Gönder-Frederick, and D. Simon. 2011. PANORAMA: A European study to evaluate quality of life and treatment satisfaction in patients with type 2 diabetes mellitus—Study design. Primary Care Diabetes. 2011 Jul 11 [Epub ahead of print].
Brézin, A.P., A. Lafuma, F. Fagnani, M. Mesbah, and G. Berdeaux. 2005. Prevalence and burden of self-reported blindness, low vision, and visual impairment in the French community: A nationwide survey. Archives of Ophthalmology 123(8):1117–1124.
Brookmeyer, R., M.M. Corrada, F.C. Curriero, and C. Kawas. 2002. Survival following a diagnosis of Alzheimer’s disease. Archives of Neurology 59(11):1764–1767.
Callahan, C.M., M.A. Boustani, F.W. Unverzagt, M.G. Austrom, T.M. Damush, A.J. Perkins, B.A. Fultz, S.L. Hui, S.R. Counsell, and H.C. Hendrie. 2006. Effectiveness of collaborative care for older adults with Alzheimer disease in primary care: A randomized controlled trial. Journal of the American Medical Association 295(18):2148–2157.
Cannuscio, C.C., G.A. Colditz, E.B. Rimm, L.F. Berkman, C.P. Jones, and I. Kawachi. 2004. Employment status, social ties, and caregivers’ mental health. Social Science and Medicine 58(7):1247–1256.
Casalino, L., R.R. Gillies, S.M. Shortell, J.A. Schmittdiel, T. Bodenheimer, J.C. Robinson, T. Rundall, N. Oswald, H. Schauffler, and M.C. Wang. 2003. External incentives, information technology, and organized processes to improve health care quality for patients with chronic diseases. Journal of the American Medical Association 289(4):434–441.
Cassell, E.J. 1983. The relief of suffering. Archives of Internal Medicine 143(3):522–523.
CDC (Centers for Disease Control and Prevention) (a). Arthritis—Data and Statistics—Comorbidities. http://www.cdc.gov/arthritis/data_statistics/comorbidities.htm (accessed October 5, 2011).
CDC (b). Healthy People 2010 Leading Health Indicators at a Glance. http://www.cdc.gov/nchs/healthy_people/hp2010/hp2010_indicators.htm (accessed October 5, 2011).
CDC (c). Osteoarthritis. http://www.cdc.gov/arthritis/basics/osteoarthritis.htm (accessed February 14, 2012).
CDC (d). Winnable Battles. http://www.cdc.gov/winnablebattles/ (accessed July 4, 2011).
CDC. 2011a. Arthritis. Meeting the Challenge. At a Glance 2011. Atlanta, GA. http://www.cdc.gov/chronicdisease/resources/publications/aag/pdf/2011/Arthritis-AAG-2011-508.pdf (accessed October 5, 2011).
CDC. 2011b. National Diabetes Fact Sheet: National Estimates and General Information on Diabetes and Pre-diabetes in the United States, 2011. Atlanta, GA. http://www.cdc.gov/diabetes/pubs/pdf/ndfs_2011.pdf (accessed November 16, 2011).
CDC. 2011c. Public Health Action Plan to Integrate Mental Health Promotion and Mental Illness Prevention with Chronic Disease Prevention. Atlanta, GA. http://www.cdc.gov/mentalhealth/docs/11_220990_Sturgis_MHMIActionPlan_FINAL-Web_tag508.pdf (accessed October 15, 2011).
CDC’s Office of Minority Health and Health Disparities (a). Fact Sheet—CDC Health Disparities and Inequalities Report—U.S., 2001. http://www.cdc.gov/minorityhealth/reports/CHDIR11/FactSheet.pdf (accessed December 19, 2011).
Cella, D., W. Riley, A. Stone, N. Rothrock, B. Reeve, S. Yount, D. Amtmann, R. Bode, D. Buysse, S. Choi, K. Cook, R. Devellis, D. DeWalt, J.F. Fries, R. Gershon, E.A. Hahn, J.S. Lai, P. Pilkonis, D. Revicki, M. Rose, K. Weinfurt, R. Hays, and PROMIS Cooperative Group. 2010. The Patient-Reported Outcomes Measurement Information System (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005-2008. Journal of Clinical Epidemiology 63(11):1179–1194.
Center for Medicare and Medicaid Innovation (a). The CMS Innovation Center. http://innovations.cms.gov/ (accessed December 19, 2011).
Center for Prevention and Health Services. 2009. Eliminating Racial and Ethnic Health Disparities; A Business Case Update for Employers. Washington, DC: Center for Prevention and Health Services. http://minorityhealth.hhs.gov/Assets/pdf/checked/1/Eliminating_Racial_Ethnic_Health_Disparities_A_Business_Case_Update_for_Employers.pdf (accessed December 19, 2011).
Charney, D.S., C.F. Reynolds, III, L. Lewis, B.D. Lebowitz, T. Sunderland, G.S. Alexopoulos, D.G. Blazer, I.R. Katz, B.S. Meyers, P.A. Arean, S. Borson, C. Brown, M.L. Bruce, C.M. Callahan, M.E. Charlson, Y. Conwell, B.N. Cuthbert, D.P. Devanand, M.J. Gibson, G.L. Gottlieb, K.R. Krishnan, S.K. Laden, C.G. Lyketsos, B.H. Mulsant, G. Niederehe, J.T. Olin, D.W. Oslin, J. Pearson, T. Persky, B.G. Pollock, S. Raetzman, M. Reynolds, C. Salzman, R. Schulz, T.L. Schwenk, E. Scolnick, J. Unützer, M.M. Weissman, R.C. Young, and Depression and Bipolar Support Alliance. 2003. Depression and Bipolar Support Alliance consensus statement on the unmet needs in diagnosis and treatment of mood disorders in late life. Archives of General Psychiatry 60(7):664–672.
Chen, W.H., A.S. Cross, R. Edelman, M.B. Sztein, W.C. Blackwelder, and M.F. Pasetti. 2011. Antibody and Th1-type cell-mediated immune responses in elderly and young adults immunized with the standard or a high dose influenza vaccine. Vaccine 29(16):2865–2873. Chodosh, J., T.E. Seeman, E. Keeler, A. Sewall, S.H. Hirsch, J.M. Guralnik, and D.B. Reuben. 2004. Cognitive decline in high-functioning older persons is associated with an increased risk of hospitalization. Journal of the American Geriatrics Society 52(9):1456–1462.
Chu, T.F., M.A. Rupnick, R. Kerkela, S.M. Dallabrida, D. Zurakowski, L. Nguyen, K. Woulfe, E. Pravda, F. Cassiola, J. Desai, S. George, J.A. Morgan, D.M. Harris, N.S. Ismail, J.H. Chen, F.J. Schoen, A.D. Van den Abbeele, G.D. Demetri, T. Force, and M.H. Chen. 2007. Cardiotoxicity associated with tyrosine kinase inhibitor sunitinib. Lancet 370(9604):2011–2019.
Claridge, J.A., W.H. Leukhardt, J.F. Golob, A.M. McCoy, and M.A. Malangoni. 2010. Moving beyond traditional measurement of mortality after injury: Evaluation of risk factors for late death. Journal of the American College of Surgeons 210(5):788–796.
CMS (Centers for Medicare and Medicaid Services). 2009. National Health Expenditures Accounts: Definitions, Sources, and Methods, 2009. https://www.cms.gov/nationalhealthexpenddata/downloads/dsm-09.pdf (accessed December 19, 2011).
Compton, W.M., K.P. Conway, F.S. Stinson, and B.F. Grant. 2006. Changes in the prevalence of major depression and comorbid substance use disorders in the United States between 1991-1992 and 2001-2002. American Journal of Psychiatry 163(12):2141–2147.
Copenhagen Consensus Center (a). http://www.copenhagenconsensus.com (accessed December 19, 2011).
Cormier, J.N., R.L. Askew, K.S. Mungovan, Y. Xing, M.I. Ross, and J.M. Armer. 2010. Lymphedema beyond breast cancer: A systematic review and meta-analysis of cancer-related secondary lymphedema. Cancer 6(22):5138–5149.
Corrigan, J.D., A.W. Selassie, and J.A. Orman. 2010. The epidemiology of traumatic brain injury. Journal of Head Trauma Rehabilitation 25(2):72–80.
Counsell, S.R., C.M. Callahan, D.O. Clark, W. Tu, A.B. Buttar, T.E. Stump, and G.D. Ricketts. 2007. Geriatric care management for low-income seniors: A randomized controlled trial. Journal of the American Medical Association 298(22):2623–2633.
Cowie, C.C., K.F. Rust, E.S. Ford, M.S. Eberhardt, D.D. Byrd-Holt, C. Li, D.E. Williams, E.W. Gregg, K.E. Bainbridge, S.H. Saydah, and L.S. Geiss. 2009. Full accounting of diabetes and pre-diabetes in the U.S. population in 1988-1994 and 2005-2006. Diabetes Care 32(2):287–294.
Cukierman, T., H.C. Gerstein, and J.D. Williamson. 2005. Cognitive decline and dementia in diabetes—systematic overview of prospective observational studies. Diabetologia 48(12):2460–2469.
Dalstra, J.A., A.E. Kunst, C. Borrell, E. Breeze, E. Cambois, G. Costa, J.M. Geurts, E. Lahelma, H. Van Oyen, N.K. Rasmussen, E. Regidor, T. Spadea, and J.P. Mackenbach. 2005. Socioeconomic differences in the prevalence of common chronic diseases: An overview of eight European countries. International Journal of Epidemiology 34(2):316–326.
DCPP (The Disease Control Priorities Project) (a). About DCPP. http://www.dcp2.org/page/main/Home.html (accessed December 19, 2011).
De Groot, M., R. Anderson, K.E. Freedland, R.E. Clouse, and P.J. Lustman. 2001. Association of depression and diabetes complications: A meta-analysis. Psychosomatic Medicine 63(4):619–630.
De Rekeneire, N., H.E. Resnick, A.V. Schwartz, R.I. Shorr, L.H. Kuller, E.M. Simonsick, B. Vellas, T.B. Harris, and Health, Aging, and Body Composition study. 2003. Diabetes is associated with subclinical functional limitation in nondisabled older individuals: The Health, Aging, and Body Composition study. Diabetes Care 26(12):3257–3263.
Dhalla, I.A., G.M. Anderson, M.M. Mamdani, S.E. Bronskill, K. Sykora, and P.A. Rochon. 2002. Inappropriate prescribing before and after nursing home admission. Journal of American Geriatrics Society 50(6):995–1000.
Egede, L.E. 2004. Diabetes, major depression, and functional disability among U.S. adults. Diabetes Care 27(2):421–428.
Fazel, S., N. Långström, A. Hjern, M. Grann, and P. Lichtenstein. 2009. Schizophrenia, substance abuse, and violent crime. Journal of the American Medical Association 301(19): 2016–2023.
Floyd, J.D., D.T. Nguyen, R.L. Lobins, Q. Bashir, D.C. Doll, and M.C. Perry. 2005. Cardiotoxicity of cancer therapy. Journal of Clinical Oncology 23(30):7685–7696.
Freburger, J.K., G.M. Holmes, R.P. Agans, A.M. Jackman, J.D. Darter, A.S. Wallace, L.D. Castel, W.D. Kalsbeek, and T.S. Carey. 2009. The rising prevalence of chronic low back pain. Archives of Internal Medicine 169(3):251–258.
Frick, K.D., E.W. Gowe, J.H. Kempen, and J.L. Wolff. 2007. Economic impact of visual impairment and blindness in the United States. Archives of Ophthalmology 125(4):544–550.
Ganguli, M., H.H. Dodge, C. Shen, R.S. Pandav, S.T. DeKosky. 2005. Alzheimer’s disease and mortality: A 15-year epidemiological study. Archives of Neurology 62(5):779–784.
Gardetto, N.J. 2011. Self-management in heart failure: Where have we been and where should we go? Journal of Multidisciplinary Healthcare 4:39–51.
Gazmararian, J.A., M.V. Williams, J. Peel, and D.W. Baker. 2003. Health literacy and knowledge of chronic disease. Patient Education and Counseling 51(3):267–275.
Gilbody, S., P. Bower, J. Fletcher, D. Richards, and A.J. Sutton. 2006. Collaborative care for depression: A cumulative meta-analysis and review of longer-term outcomes. Archives of Internal Medicine 166(21):2314–2321.
Giovannetti, E.R., J.L. Wolff, K.D. Frick, and C. Boult. 2009. Construct validity of the Work Productivity and Activity Impairment questionnaire across informal caregivers of chronically ill older patients. Value Health 12(6):1011–1017.
Goetzel, R.Z., K. Hawkins, R.J. Ozminkowski, and S. Wang. 2003. The health and productivity cost burden of the “top 10” physical and mental health conditions affecting six large U.S. employers in 1999. Journal of Occupational and Environmental Medicine 45(1):5–14.
Gonzalez, J.S., M. Peyrot, L.A. McCarl, E.M. Collins, L. Serpa, M.J. Mimaga, and S.A. Safren. 2008. Depression and diabetes treatment nonadherence: A meta-analysis. Diabetes Care 31(12):2398–2403.
Green, C.R., K.O. Anderson, T.A. Baker, L.C. Campbell, S. Decker, R.B. Fillingim, D.A. Kaloukalani, K.E. Lasch, C. Myers, R.C. Tait, K.H. Todd, and A.H. Vallerand. 2003. The unequal burden of pain: Confronting racial and ethnic disparities in pain. Pain Medicine 4(3):277–294.
Gregg, E.W., G.L. Beckles, D.F. Williamson, S.G. Leveille, J.A. Langlois, M.M. Engelgau, and K.M. Narayan. 2000. Diabetes and physical disability among older U.S. adults. Diabetes Care 23(9):1272–1277.
Guitera, V., P. Muñoz, J. Castillo, and J. Pascual. 2002. Quality of life in chronic daily headache: A study in a general population. Neurology 58(7):1062–1065.
Hahn, B., S. Dogra, and S. King-Zeller. 2001. Impact of chronic pain on health care resource use, daily activities and family burden. Pain Medicine 1(2):195–196.
Ham, C. 1997. Priority setting in health care: Learning from international experience. Health Policy 42(1):49–66.
Havercamp, S.M., D. Scandlin, and M. Roth. 2004. Health disparities among adults with developmental disabilities, adults with other disabilities, and adults not reporting disability in North Carolina. Public Health Reports 119(4):418–426.
Helzner, E.P., N. Scarmeas, S. Cosentino, M.X. Tang, N. Schupf, and Y. Stern. 2008. Survival in Alzheimer’s disease: A multiethnic, population-based study of incident cases. Neurology 71(19):1489–1495.
Hewitt, M., J.H. Rowland, and R. Yancik. 2003. Cancer survivors in the United States: Age, health, and disability. Journals of Gerontology: Series A, Biological Sciences and Medical Sciences 58(1):82–91.
HHS (U.S. Department of Health and Human Services), Office of Disease Prevention and Health Promotion. 2010. National Action Plan to Improve Health Literacy. Washington, DC. http://www.health.gov/communication/hlactionplan/pdf/Health_Literacy_Action_Plan.pdf (accessed October 6, 2011).
Hootman, J.M., and C.G. Helmick. 2006: Projections of U.S. prevalence of arthritis and associated activity limitations. Arthritis Rheumatology 54(1):226–229.
Horowitz, A. 2003. Depression and vision and hearing impairments in later life. Generations 27:32–38.
Institute for Healthcare Improvement (a). The IHI Triple Aim. http://www.ihi.org/offerings/Initiatives/TripleAim/Pages/default.aspx (accessed December 19, 2011).
IOM (Institute of Medicine). 1999. To Err Is Human: Building a Safer Health System. Washington, DC: National Academy Press.
IOM. 2003a. The Future of the Public’s Health in the 21st Century. Washington, DC: The National Academies Press.
IOM. 2003b. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care. Washington, DC: The National Academies Press. [OU—PUB DATE IS 2003] IOM. 2009. Retooling for an Aging America: Building the Health Care Workforce. Washington, DC: The National Academies Press.
IOM. 2011a. Leading Health Indicators for Healthy People 2020 Letter Report. Washington, DC: The National Academies Press.
IOM. 2011b. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: The National Academies Press.
IOM and NRC (National Research Council). 2006. From Cancer Patient to Cancer Survivor—Lost in Transition: An American Society of Clinical Oncology and Institute of Medicine Symposium. Washington, DC: The National Academies Press.
Ivanova, J.I., H.G. Birnbaum, Y. Kidolezi, G. Subramanian, S.A. Khan, and M.D. Stensland. 2010. Direct and indirect costs of employees with treatment-resistant and non-treatment-resistant major depressive disorder. Current Medical Research and Opinion 26(10):2475–2484.
Jennings, A.M., K.S. Lewis, S. Murdoch, J.F. Talbot, C. Bradley, and J.D. Ward. 1991. Randomized trial comparing continuous subcutaneous insulin infusion and conventional insulin therapy in type II diabetic patients poorly controlled with sulfonylureas. Diabetes Care 14(8):738–744.
Jensen, M.P., M.J. Chernoff, and R.H. Dworkin. 2007. The impact of neuropathic pain on health-related quality of life: Review and implications. Neurology 68(15):1178–1182.
Jeon, Y., B. Essue, S. Jan, R. Wells, and J.A. Whitworth. 2009. Economic hardship associated with managing chronic illness: A qualitative inquiry. BMC Health Services Research 9:182. http://www.biomedcentral.com/1472-6963/9/182 (accessed November 17, 2011).
Jeschke, M.G., G.G. Gauglitz, G.A. Kulp, C.C. Finnerty, F.N. Williams, R. Kraft, O.E. Suman, R.P. Mlcak, and D.N. Herndon. 2011. Long-term persistence of the pathophysiological response to severe burn injury. PLoS One 6(7):e21245.
Kaiser Family Foundation. 2010. U.S. Health Care Costs. http://www.kaiseredu.org/Issue-Modules/US-Health-Care-Costs/Background-Brief.aspx (accessed October 12, 2011).
Kalyani, R.R., C.D. Saudek, F.L. Brancati, and E. Selvin. 2010. Association of diabetes, comorbidities, and A1C with functional disability in older adults: Results from the National Health and Nutrition Examination Survey (NHANES), 1999-2006. Diabetes Care 33(5):1055–1060.
Katon, W., M. Sullivan, and E. Walker. 2001. Medical symptoms without identified pathology: Relationship to psychiatric disorders, childhood and adult trauma, and personality traits. Annals of Internal Medicine 134(9, Part 2):917–925.
Katon, W., E.H. Lin, and K. Kroenke. 2007. The association of depression and anxiety with medical symptom burden in patients with chronic medical illness. General Hospital Psychiatry 29(2):147–155.
Katon, W., J. Unützer, K. Wells, and L. Jones. 2010. Collaborative depression care: History, evolution and ways to enhance dissemination and sustainability. General Hospital Psychiatry 32(5):456–464.
Keeler, E., J.M. Guralnik, H. Tian, R.B. Wallace, and D.B. Reuben. 2010. The impact of functional status on life expectancy in older persons. Journals of Gerontology: Series A, Biological and Medical Sciences 65(7):727–733.
Kessler, R.C., H. Birnbaum, E. Bromet, I. Hwang, N. Sampson, and V. Shahly. 2010. Age differences in major depression: Results from the National Comorbidity Survey Replication (NCS-R). Psychological Medicine 40(2):225–237.
Khan, A.S. 2011. Public health preparedness and response in the USA since 9/11: A national health security imperative. Lancet 378(9794):953–956.
Klein, R., B.E. Klein, S.E. Moss, M.D. Davis, and D.L. DeMets. 1989. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. X. Four-year incidence and progression of diabetic retinopathy when age at diagnosis is 30 years or more. Archives of Ophthalmology 107(2):244–249.
Kongkaew, C., P.R. Noyce, and D.M. Ashcroft. 2008. Hospital admissions associated with adverse drug reactions: A systematic review of prospective observational studies. Annals of Pharmacotherapy 42(7):1017–1025.
Kuzuya, M., and Y. Hirakawa. 2009. Increased caregiver burden associated with hearing impairment but not vision impairment in disabled community-dwelling older people in Japan. Journal of the American Geriatrics Society 57(2):357–358.
Larson, E.B., M.F. Shadlen, L. Wang, W.C. McCormick, J.D. Bowen, L. Teri, and W.A. Kukull. 2004. Survival after initial diagnosis of Alzheimer disease. Annals of Internal Medicine 140(7):501–509.
Lawrence, D., S. Kisely, and J. Pais. 2010. The epidemiology of excess mortality in people with mental illness. Canadian Journal of Psychiatry 55(12):752–760.
Lebowitz, B.D., J.L. Pearson, L.S. Schneider, C.F. Reynolds, III, G.S. Alexopoulos, M.L. Bruce, Y. Conwell, I.R. Katz, B.S. Meyers, M.F. Morrison, J. Mossey, G. Niederehe, and P. Parmelee. 1997. Diagnosis and treatment of depression in late life. Consensus statement update. Journal of the American Medical Association 278(14):1186–1190.
Lee, S., G.A. Colditz, L.F. Berkman, and I. Kawachi. 2003. Caregiving and risk of coronary heart disease in U.S. women: A prospective study. American Journal of Preventive Medicine 24(2):113–119.
Lilly, M.B., A. LaPorte, and P.C. Coyte. 2007. Labor market work and home care’s unpaid caregivers: A systematic review of labor force participation rates, predictors of labor market withdrawal, and hours of work. The Milbank Quarterly 85(4):641–690.
Lin, E.H., W. Katon, M. Von Korff, C. Rutter, G.E. Simon, M. Oliver, P. Ciechanowski, E.J. Ludman, T. Bush, and B. Young. 2004. Relationship of depression and diabetes self-care, medication adherence, and preventive care. Diabetes Care 27(9):2154–2160.
Lind, H., A.C. Waldenström, G. Dunberger, M. al-Abany, E. Alevronta, K.A. Johansson, C. Olsson, T. Nyberg, U. Wilderäng, G. Steineck, and E. Åvall-Lundqvist. 2011. Late symptoms in long-term gynaecological cancer survivors after radiation therapy: A population-based cohort study. British Journal of Cancer 105(6):737–745.
Linton, S.J. 2002. Early identification and intervention in the prevention of musculoskeletal pain. American Journal of Industrial Medicine 41(5):433–442.
Linton, S.J., and M. Ryberg. 2001. A cognitive-behavioral group intervention as prevention for persistent neck and back pain in a non-patient population: A randomized controlled trial. Pain 90(1–2):83–90.
Livingston, D.H., T. Tripp, C. Biggs, and R.F. Lavery. 2009. A fate worse than death? Long-term outcome of trauma patients admitted to the surgical intensive care unit. Journal of Trauma 67(2):341–348.
Lustman, P.J., R.J. Anderson, K.E. Freedland, M. De Groot, R.M. Carney, and R.E. Clouse. 2000. Depression and poor glycemic control: A meta-analytic review of the literature. Diabetes Care 23(7):934–942.
Lyness, J.M., A. Niculescu, X. Tu, C.F. Reynolds, III, and E.D. Caine. 2006. The relationship of medical comorbidity and depression in older, primary care patients. Psychosomatics 47(5):435–439.
Magaziner, J., P. German, S.I. Zimmerman, J.R. Hebel, L. Burton, A.L. Gruber-Baldini, C. May, and S. Kittner. 2000. The prevalence of dementia in a statewide sample of new nursing home admissions aged 65 and older: Diagnosis by expert panel. Epidemiology of Dementia in Nursing Homes Research Group. Gerontologist 40(6):663–672.
Mao, J.J., C. Stricker, D. Bruner, S. Xie, M.A. Bowman, J.T. Farrar, B.T. Greene, and A. DeMichele. 2009. Patterns and risk factors associated with aromatase inhibitor-related arthralgia among breast cancer survivors. Cancer 115(16):3631–3639.
Maty, S.C., L.P. Fried, S. Volpato, J. Williamson, F.L. Brancati, and C.S. Blaum. 2004. Patterns of disability related to diabetes mellitus in older women. Journals of Gerontology: Series A, Biological and Medical Sciences 59(2):148–153.
McNeil, J.M., and J. Binette. 2001. Prevalence of disabilities and associated health conditions among adults—United States, 1999. MMWR 50(7):120–125. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5007a3.htm (accessed November 16, 2011).
McWilliams, L.A., B.J. Cox, and M.W. Enns. 2003. Mood and anxiety disorders associated with chronic pain: An examination in a nationally representative sample. Pain 106(1–2):127–133.
Merikangas, K.R., M. Ames, L. Cui, P.E. Stang, T.B. Ustun, M. Von Korff, and R.C. Kessler. 2007. The impact of comorbidity of mental and physical conditions on role disability in the U.S. adult household population. Archives of General Psychiatry 64(10):1180–1188.
Mezuk, B., W.W. Eaton, S. Albrecht, and S.H. Golden. 2008. Depression and type 2 diabetes over the lifespan: A meta-analysis. Diabetes Care 31(12):2383–2390.
Michaud, C.M., M.T. McKenna, S. Begg, N. Tomijima, M. Majmudar, M.T. Bulzacchelli, S. Ebrahim, M. Ezzati, J.A. Salomon, J.G. Kreiser, M. Hogan, and C.J. Murray. 2006. The burden of disease and injury in the United States 1996. Population Health Metrics 4(11). http://www.pophealthmetrics.com/content/4/1/11 (accessed November 16, 2011).
Michel, P., J.L. Quenon, A.M. de Sarasqueta, and O. Scemama. 2004. Comparison of three methods for estimating rates of adverse events and rates of preventable adverse events in acute care hospitals. British Medical Journal 328(7433):199. http://www.bmj.com/content/328/7433/199.full (accessed November 17, 2011).
Mitchell, A.J., D. Malone, and C.C. Doebbeling. 2009. Quality of medical care for people with and without comorbid mental illness and substance misuse: Systematic review of comparative studies. The British Journal of Psychiatry: The Journal of Mental Science 194(6):491–499.
Mohr, P.E., J.J. Feldman, J.L. Dunbar, A. McConkey-Robbins, J.K. Niparko, R.K. Rittenhouse, and M.W. Skinner. 2000. The societal costs of severe to profound hearing loss in the United States. International Journal of Technology Assessment in Health Care 16(4): 1120–1135.
Mokdad, A.H., J. S. Marks, D.F. Stroup, and J.L. Gerberding. 2004. Actual causes of death in the United States, 2000. Journal of the American Medical Association 291(10):1238–1245.
Munshi, M., L. Grande, M. Hayes, D. Ayres, E. Suhl, R. Capelson, S. Lin, W. Milberg, and K. Weinger. 2006. Cognitive dysfunction is associated with poor diabetes control in older adults. Diabetes Care 29(8):1794–1799.
Murphy, J.M., N.M. Laird, R.R. Monson, A.M. Sobol, and A.H. Leighton. 2000. A 40-year perspective on the prevalence of depression: The Stirling County Study. Archives of General Psychiatry 57(3):209–215.
Murphy, L., T.A. Schwartz, C.G. Helmick, J.B. Renner, G. Tudor, G. Koch, A. Dragomir, W.D. Kalsbeek, G. Luta, and J.M. Jordan. 2008. Lifetime risk of symptomatic knee osteoarthritis. Arthritis and Rheumatism 59(9):1207–1213.
NAC (National Alliance for Caregiving) and AARP. 2009. Caregiving in the U.S. 2009. http://www.caregiving.org/data/Caregiving_in_the_US_2009_full_report.pdf (accessed October 15, 2011).
Nagi, S.Z. 1976. An epidemiology of disability among adults in the United States. The Milbank Memorial Fund Quarterly. Health and Society 54(4):439–467.
Nakase-Richardson, R., J. Whyte, J.T. Giacino, S. Pavawalla, S.D. Barnett, S.A. Yablon, M. Sherer, K. Kalmar, F.M. Hammond, B. Greenwald, L.J. Horn, R. Seel, M. McCarthy, J. Tran, and W.C. Walker. 2011. Longitudinal outcomes of patients with disordered consciousness in the NIDRR TBI model systems program. Journal of Neurotrauma 2011, Aug 4 [Epub ahead of print].
National Comorbidity Survey Replication. 2007. NCS-R Lifetime Prevalence Estimates. http://www.hcp.med.harvard.edu/ncs/ (accessed October 17, 2011).
NCES (National Center for Education Statistics). 2006. The Health Literacy of America’s Adults: Results From the 2003 National Assessment of Adult Literacy (NCES 2006-483). Washington, DC. http://nces.ed.gov/pubs2006/2006483.pdf (accessed October 6, 2011).
NCHS (National Center for Health Statistics). 2009. Summary health statistics for U.S. adults: National Health Interview Survey, 2008. Vital Health Statistics 10(242). http://www.cdc.gov/nchs/data/series/sr_10/sr10_242.pdf (accessed November 17, 2011).
NCI (National Cancer Institute) (a). Lymphedema (PDQ®). http://www.cancer.gov/cancertopics/pdq/supportivecare/lymphedema/healthprofessional/page1 (accessed February 14, 2012).
NCI. 2011. News. U.S. Cancer Survivors Grows to Nearly 12 Million. http://www.cancer.gov/newscenter/pressreleases/2011/survivorshipMMWR2011/print (accessed December 19, 2011).
NCOA (National Council on Aging). 1999. The Consequences of Untreated Hearing Loss in Older Persons. Washington, DC: National Council on Aging. http://www.hearingoffice.com/download/UntreatedHearingLossReport.pdf (accessed October 6, 2011).
Ness, K.K., M.M. Wall, J.M. Oakes, L.L. Robison, and J.G. Gurney. 2006. Physical performance limitations and participation restrictions among cancer survivors: A population-based study. Annals of Epidemiology 16(3):197–205.
Newman, S., L. Steed, and K. Mulligan. 2004. Self-management interventions for chronic illness. Lancet 364(9444):1523–1537.
NINDS (National Institute of Neurological Disorders and Stroke) (a). NINDS Chronic Pain Information Page. http://www.ninds.nih.gov/disorders/chronic_pain/chronic_pain.htm (accessed September 12, 2011).
North Carolina Department of Health and Human Services Division of Services for the Deaf and the Hard of Hearing. 2009. The Impact of Hearing Loss on Older Adults in North Carolina. Raleigh, NC: North Carolina Department of Health and Human Services Division of Services for the Deaf and the Hard of Hearing. http://www.ncdhhs.gov/dsdhh/leg_study.pdf (accessed October 6, 2011).
O’Brien, J.A., and J.J. Caro. 2001. Alzheimer’s disease and other dementia in nursing homes: Levels of management and cost. International Psychogeriatrics 13(3):347–358.
Okie, S. 2011. Confronting Alzheimer’s Disease. New England Journal of Medicine 365(12): 1069–1072.
Orces, C.H., and F.J. Martinez. 2011. Epidemiology of fall-related forearm and wrist fractures among adults treated in U.S. hospital emergency departments. Injury Prevention 17(1):33–36.
Phillips, C.J., and C. Harper. 2011. The economics associated with persistent pain. Current Opinion in Supportive and Palliative Care 5(2):127–130.
Pinder, M.C., Z. Duan, J.S. Goodwin, G.N. Hortobagyi, and S.H. Giordano. 2007. Congestive heart failure in older women treated with adjuvant anthracycline chemotherapy for breast cancer. Journal of Clinical Oncology 25(25):3808–3815.
Pinquart, M., and S. Sörensen. 2003. Differences between caregivers and noncaregivers in psychological and physical health: A meta-analysis. Psychology and Aging 18(2):250–267.
Reichard, A., H. Stolzle, and M.H. Fox. 2011. Health disparities among adults with physical disabilities or cognitive limitations compared to individuals with no disabilities in the United States. Disability and Health Journal 4(2):59–67.
Reuben, D.B. 2009. Medical care for the final years of life: “When you’re 83, it’s not going to be 20 years.” Journal of the American Medical Association 302(24):2686–2694.
Reuben, D.B., S. Mui, M. Damesyn, A.A. Moore, and G.A. Greendale. 1999. The prognostic value of sensory impairment in older persons. Journal of the American Geriatrics Society 47(8):930–935.
Reuben, D.B., C.P. Roth, J.C. Frank, S.H. Hirsch, D. Katz, H. McCreath, J. Younger, M. Murawski, E. Edgerly, J. Maher, K. Maslow, and N.S. Wenger. 2010. Assessing care of vulnerable elders—Alzheimer’s disease: A pilot study of a practice redesign intervention to improve the quality of dementia care. Journal of the American Geriatrics Society 58(2):324–329.
Ricci, J.A., W.F. Stewart, E. Chee, C. Leotta, K. Foley, and M.C. Hochberg. 2005. Pain exacerbation as a major source of lost productive time in U.S. workers with arthritis. Arthritis and Rheumatism 53(5):673–681.
Roberts, R.O., Y.E. Geda, D.S. Knopman, T.J. Christianson, V.S. Pankratz, B.F. Boeve, A. Vella, W.A. Rocca, and R.C. Peterson. 2008. Association of duration and severity of diabetes mellitus with mild cognitive impairment. Archives of Neurology 65(8):1066–1073.
Robinson, R.L., H.G. Birnbaum, M.A. Morley, T. Sisitsky, P.E. Greenberg, and A.J. Claxton. 2003. Economic cost and epidemiological characteristics of patients with fibromyalgia claims. Journal of Rheumatology 30(6):1318–1325.
Rosen, A.B., and D.M. Cutler. 2009. Challenges in building disease-based national health accounts. Medical Care 47(7 Suppl 1):S7–S13.
Rothrock, N.E., R.D. Hays, K. Spritzer, S.E. Yount, W. Riley, and D. Cella. 2010. Relative to the general U.S. population, chronic diseases are associated with poorer health-related quality of life as measured by the Patient-Reported Outcomes Measurement Information System (PROMIS). Journal of Clinical Epidemiology 63(11):1195–1204.
Rowland, J., A. Mariotto, N. Aziz, G. Tesauro, E.J. Feuer, D. Blackman, P. Thompson, and L.A. Pollack. 2004. Cancer survivorship—United States, 1971–2001. Morbidity and Mortality Weekly Report 53(24):526–529. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5324a3.htm (accessed December 19, 2011).
Royer, A. 1998. Social Isolation: The Most Distressing Consequence of Chronic Illness. http://research.allacademic.com/meta/p_mla_apa_research_citation/1/1/0/2/1/p110216_index.html?phpsessid=7fc502fc26e39b3c7a03341b5cc8d2b1 (accessed October 15, 2011).
Rubin, R.R., and M. Peyrot. 1999. Quality of Life and Diabetes. Diabetes/Metabolism Research and Reviews 15(3):205–218.
Rugulies, R. 2002. Depression as a predictor for coronary heart disease. A review and meta-analysis. American Journal of Preventive Medicine 23(1):51–61.
Ryerson, B., E.F. Tierney, T.J. Thompson, M.M. Engelgau, J. Wang, E.W. Gregg, and L.S. Geiss. 2003. Excess physical limitations among adults with diabetes in the U.S. population, 1997-1999. Diabetes Care 26(1):206–210.
Sainfort, F., and P.L. Remington. 1995. The Disease Impact Assessment System (DIAS). Public Health Reports 110(5):639–644.
Schillinger, D., K. Grumbach, J. Piette, F. Wang, D. Osmond, C. Daher, J. Palacios, G.D.
Sullivan, and A.B. Bindman. 2002. Association of health literacy with diabetes outcomes. Journal of the American Medical Association 288(4):475–482.
Schoenmakers, B., F. Buntinx, and J. Delepeleire. 2010. Factors determining the impact of care-giving on caregivers of elderly patients with dementia. A systematic literature review. Maturitas 66(2):191–200.
Schroeder, S.A. 2007. We can do better—improving the health of the American people. New England Journal of Medicine 357:1221–1228.
Schulz, R., and S.R. Beach. 1999. Caregiving as a risk factor for mortality: The Caregiver Health Effects Study. Journal of the American Medical Association 282(23):2215–2219.
Schulze, B., and W. Rössler. 2005. Caregiver burden in mental illness: Review of measurement, findings and interventions in 2004-2005. Current Opinion in Psychiatry 18(6):684–691.
Seeman, T.E., B.S. McEwen, J.W. Rowe, and B.H. Singer. 2001. Allostatic load as a marker of cumulative biological risk: MacArthur studies of successful aging. Proceedings of the National Academy of Sciences 98(8):4770–4775.
Sinclair, A.J., A.J. Girling, and A.J. Bayer. 2000. Cognitive dysfunction in older subjects with diabetes mellitus: Impact on diabetes self-management and use of care services. All Wales Research into Elderly (AWARE) Study. Diabetes Research and Clinical Practice 50(3):203–212.
Smith, B.D., G.L. Smith, A. Hurria, G.N. Hortobagyi, and T.A. Buchholz. 2009. Future of cancer incidence in the United States: Burdens upon an aging, changing nation. Journal of Clinical Oncology 27(17):2758–2765.
Stoddard, S., L. Jans, J. Ripple, and L. Kraus. 1998. Chartbook on work and disability. Washington, DC: U.S. National Institute on Disability and Rehabilitation Research. http://www.infouse.com/disabilitydata/workdisability/ (accessed November 17, 2011).
Taylor, Jr., D.H., M. Ezell, M. Kuchibhatla, T. Ostbye, and E.C. Clipp. 2008. Identifying the trajectories of depressive symptoms for women caring for their husbands with dementia. Journal of the American Geriatrics Society 56(2):322–327.
Teo, W.S., W.S. Tan, W.F. Chong, J. Abisheganaden, Y.J. Lew, T.K. Lim, and B.H. Heng. 2011. The economic burden of chronic obstructive pulmonary disease. Respirology 2011, Sep 29 [Epub ahead of print].
Thabit, H., S.M. Kennelly, A. Bhagarva, M. Ogunlewe, P.M. McCormack, J.H. McDermott, and S. Sreenan. 2009. Utilization of Frontal Assessment Battery and Executive Interview 25 in assessing for dysexecutive syndrome and its association with diabetes self-care in elderly patients with type 2 diabetes mellitus. Diabetes Research and Clinical Practice 86(3):208–212.
Thacker, S.B., D.F. Stroup, V. Carande-Kulis, J.S. Marks, K. Roy, and J.L. Gerberding. 2006. Measuring the public’s health. Public Health Reports 121(1):14–22. http://www.publichealthreports.org/issueopen.cfm?articleID=1576 (accessed November 17, 2011).
Tinetti, M.E., S.T. Bogardus, Jr., and J.V. Agostini. 2004. Potential pitfalls of disease-specific guidelines for patients with multiple conditions. New England Journal of Medicine 351(27):2870–2874.
Towns, K., P.L. Bedard, and S. Verma. 2008. Matters of the heart: Cardiac toxicity of adjuvant systemic therapy for early-stage breast cancer. Current Oncology 15(Suppl 1):S16–S29.
Turkcapar, N., O. Demir, T. Atli, M. Kopuk, M. Turgay, G. Kinikli, and M. Duman. 2006. Late onset rheumatoid arthritis: Clinical and laboratory comparisons with younger onset patients. Archives of Gerontology Geriatrics 42(2):225–231.
UK Prospective Diabetes Study Group. 1998. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 352(9131):837–853.
UNC Center for Excellence in Community Mental Health (a). Myths, Half-truths, and Common Misconceptions about Schizophrenia and Severe and Persistent Mental Illness (SPMI). http://www.unccmh.org/clients-and-families/learn-about-mental-illness/common-myths/ (accessed February 14, 2012).
U.S. Census Bureau. 2011. Current Population Reports, P60-239, Income, Poverty, and Health Insurance Coverage in the United States: 2010. Washington, DC. http://www.census.gov/prod/2011pubs/p60-239.pdf (accessed November 17, 2011).
USDA (U.S. Department of Agriculture). 2006. Food Safety for People with Cancer. http://www.fsis.usda.gov/PDF/Food_Safety_for_People_with_Cancer.pdf (accessed September 2, 2011).
USPSTF (U.S. Preventive Services Task Force). 2009. Screening for depression in adults: U.S. preventive services task force recommendation statement. Annals of Internal Medicine 151(11):784–792.
Vickrey, B.G., B.S. Mittman, K.I. Connor, M.L. Pearson, R.D. Della Penna, T.G. Ganiats, R.W. Demonte, Jr., J. Chodosh, X. Cui, S. Vassar, N. Duan, and M. Lee. 2006. The effect of a disease management intervention on quality and outcomes of dementia care: A randomized, controlled trial. Annals of Internal Medicine 145(10):713–726.
Vilnius, D., and S. Dandoy. 1990. A priority rating system for public health programs. Public Health Reports 105(5):463–470.
Vitaliano, P.P., J.M. Scanlan, J. Zhang, M.V. Savage, I.B. Hirsch, and I.C. Siegler. 2002. A path model of chronic stress, the metabolic syndrome, and coronary heart disease. Psychosomatic Medicine 64(3):418–435.
Volpato, S., C. Blaum, H. Resnick, L. Ferrucci, L.P. Fried, J.M. Guralnik, and Women’s Health and Aging Study. 2002. Comorbidities and impairments explaining the association between diabetes and lower extremity disability: The Women’s Health and Aging Study. Diabetes Care 25(4):678–683.
Vu, H.T.V., J.D. Keeffe, C.A. McCarty, and H.R. Taylor. 2005. Impact of unilateral and bilateral vision loss on quality of life. British Journal of Ophthalmology 89(3):360–363.
Warsi, A., P.S. Wang, M.P. LaValley, J. Avorn, and D.H. Solomon. 2004. Self-management education programs in chronic disease: A systematic review and methodological critique of the literature. Archives of Internal Medicine 164(15):1641–1649.
Watson, S.D. 2003. Forward. St. Louis University Law Journal 48(1).
Weijman, I., J.G. Ros Wynand, G. E.H.M. Rutten, W.B. Schaufeli, M.J. Schabracq, and J.A.M.
Winnubst. 2005. Frequency and perceived burden of diabetes self-management activities in employees with insulin-treated diabetes: Relationships with health outcomes. Diabetes Research and Clinical Practice 68(1):56–64.
Wen, X.J., L. Balluz, and A. Mokdad. 2009. Association between media alerts of air quality index and change of outdoor activity among adult asthma in six states, BRFSS, 2005. Journal of Community Health 34(1):40–46.
WHO (World Health Organization) (a). International Classification of Functioning, Disability and Health (ICF). http://www.who.int/classifications/icf/en/ (accessed December 13, 2011).
WHO. 2008. World Cancer Report. Geneva, Switzerland: World Health Organization.
Williams, D.R., H.M. González, H. Neighbors, R. Nesse, J.M. Abelson, J. Sweetman, and J.S. Jackson. 2007. Prevalence and distribution of major depressive disorder in African Americans, Caribbean blacks, and non-Hispanic whites: Results from the National Survey of American Life. Archives of General Psychiatry 64(3):305–315.
Wilson, M., M.P. Moore, and H. Lunt. 2004. Treatment satisfaction after commencement of insulin in Type 2 diabetes. Diabetes Research and Clinical Practice 66(3):263–267.
Wolf, M.S., J.A. Gazmararian, and D.W. Baker. 2005. Health literacy and functional health status among older adults. Archives of Internal Medicine 165(17):1946–1952.
Wolff, J.L., and C. Boult. 2005. Moving beyond round pegs and square holes: Restructuring Medicare to improve chronic care. Annals of Internal Medicine 143(6):439–445.
Woo, J., S.C. Ho, S.G. Chan, A.L.M. Yu, Y.K. Yuen, and J. Lau. 1997. An estimate of chronic disease burden and some economic consequences among the elderly Hong Kong population. Journal of Epidemiology and Community Health 51(5):486–489.
Wormgoor, M.E.A., A. Indahl, M.W. van Tulder, and H.C.G. Kemper. 2006. Functioning description according to the ICF Model in chronic back pain: Disablement appears even more complex with decreasing symptom-specificity. Journal of Rehabilitation Medicine 38(2):93–99.
Wu, E.Q., H.G. Birnbaum, L. Shi, D.E. Ball, R.C. Kessler, M. Moulis, and J. Aggarwal. 2005. The economic burden of schizophrenia in the United States in 2002. Journal of Clinical Psychiatry 66(9):1122–1129.
Yabroff, K.R., W.F. Lawrence, S. Clauser, W.W. Davis, and M.L. Brown. 2004. Burden of illness in cancer survivors: Findings from a population-based national sample. Journal of the National Cancer Institute 96(17):1322–1330.
Yaffe, K., P. Fox, R. Newcomer, L. Sands, K. Lindquist, K. Dane, and K.E. Covinsky. 2002. Patient and caregiver characteristics and nursing home placement in patients with dementia. Journal of the American Medical Association 287(16):2090–2097.
Zafagnini, S., G.M. Marcheggiani Muccioli, N. Lopomo, D. Bruni, G. Giordano, G. Ravazzolo, M. Molinari, and M. Marcacci. 2011. Prospective long-term outcomes of the medial collagen mescus implant versus partial medial meniscectomy: A minimum 10-year follow-up study. American Journal of Sports Medicine 39(5):977–985.
Zaza, C., and N. Baine. 2002. Cancer pain and psychosocial factors: A critical review of the literature. Journal of Pain and Symptom Management 24(5):526–542.




































































