5
A Continuously Learning Health Care System
In 1982, results of the Beta-Blocker Heart Attack Trial were published, showing that the use of beta-blockers after a heart attack reduced mortality by at least 25 percent (Beta-Blocker Heart Attack Trial Research Group, 1982). Further studies validated these results (Yusuf et al., 1985). Yet, by the mid-1990s, beta-blockers were being prescribed after a heart attack only 30 to 50 percent of the time (Brand et al., 1995; Burwen et al., 2003; Gottlieb et al., 1998; Krumholz et al., 1998). Even as utilization remained low, trials in the 1990s showed that the mortality reduction from beta-blocker use was as high as 40 percent and that more patients benefited from the treatment than had originally been estimated (Gottlieb et al., 1998). The use of this treatment was encouraged in the 1990s by its inclusion in professional guidelines and by efforts to measure the extent of its use. The American College of Cardiology and the American Heart Association recommended beta-blocker treatment after heart attack in their guidelines (Ryan et al., 1996, 1999). On the measurement front, the Joint Commission established a performance measurement program for hospitals, including in its measures the level of prescribing of beta-blockers after heart attack hospitalizations; the Health Care Financing Administration (now the Centers for Medicare & Medicaid Services) began collecting similar data for Medicare patients (Krumholz et al., 1998; Marciniak et al., 1998), and the National Committee for Quality Assurance (NCQA) included beta-blocker usage in its Healthcare Effectiveness Data and Information Set (HEDIS) measures (Health
Plan Employer Data and Information Set) (Bradley et al., 2001; Lee, 2007). Beyond guidelines and measures, some health plans offered financial incentives under pay-for-performance contracts to increase the rates at which beta-blocker therapy was delivered (Lee, 2007). In addition to developing guidelines, the American College of Cardiology and American Heart Association both created programs to encourage clinicians to implement these guidelines in their practices. And the Institute for Healthcare Improvement included beta-blocker use as one component of its 100,000 Lives Campaign (Gosfield and Reinertsen, 2005). After this considerable amount of effort, on May 8, 2007, NCQA retired the use of a beta-blocker measure. The measure finally was no longer necessary because most patients under most health plans were now receiving this therapy for heart attack care (Lee, 2007).
Advances in science and technology have allowed health care to make great strides in treating diseases. Some diseases considered fatal just a generation ago are now routinely managed. Despite this progress, however, health care today displays notable shortcomings on each of the six aims for high-quality care identified in the Institute of Medicine (IOM) report Crossing the Quality Chasm: safety, effectiveness, efficiency, equity, timeliness, and patient-centeredness (2001). Care varies significantly from one part of the country to another and even from one town to another, with some areas offering high-quality, high-value care and others falling short of their potential. Substantial variations exist as well in the dissemination and adoption of new innovations. Some interventions and treatments with little evidence for superior outcomes spread rapidly, while others with a strong evidence base languish in obscurity. The shortfalls of the current health care system are captured by this simple fact: fully 160 years after Semmelweis discovered the importance of hand hygiene, many American health care institutions are finding it necessary to mount campaigns to encourage providers to wash their hands (Chassin and Loeb, 2011).
The health care environment itself places unnecessary burdens on health care professionals, siloing care activities, insufficiently meeting patient needs, and failing to disseminate knowledge broadly. The “system” has few elements that are systematic. Patients often report their frustration with a health care delivery enterprise that is fragmented, uncoordinated, and diffusely organized. As a result, they often are lost in the gaps and frustrated in trying to access the care they need.
Further, as discussed in Part I of this report, evidence on what is effective for a given patient under specific clinical circumstances often is lacking, poorly disseminated, or inconsistently implemented. The sheer volume of
new clinical trials, journal articles, clinical guidelines, and other medical information far exceeds individual human cognitive capacity—no clinician can read, process, and apply all of this constantly emerging information to regular patient care. Future developments in genomics, proteomics, informatics, and technology will only exacerbate these challenges.
Asking, urging, or demanding that clinicians keep pace with new clinical knowledge will not improve the quality of care. Such an approach would only impose unnecessary and demoralizing stress on these health care providers and associated professionals. Indeed, the problems described here persist even as individual physicians, nurses, technicians, pharmacists, and others involved in patient care work diligently at performing difficult health care tasks and at providing high-quality, compassionate care to their patients. Yet, they work within a system that lags far behind other industries in the ability to assimilate and disseminate information in real time and useful form—a system impaired by the weight of its own complexity. The path to improvement, then, is to transform the current environment into a coordinated system of care. This new environment would provide tools and resources, actionable real-time information, and appropriate incentives to help providers successfully manage the increasing complexity of medical care. In short, by making the right thing easy to do, systemwide change can be achieved.
The example at the beginning of this chapter of the diffusion of the use of beta-blockers after heart attack is a success story in many ways: high-quality evidence was produced; it was incorporated into clinical care guidelines, quality improvement initiatives, and quality-of-care measures; and several health plans offered financial incentives for its uptake. Yet even with this level of effort, it took 25 years from the time the initial results were published until the time the treatment saw general use in clinical practice. This example speaks to the need to create infrastructure that makes the process of learning and improvement easier, so that the next discovery does not require 25 years of sustained effort before it is widely used to help patients.
Improving quality and controlling costs requires moving from this unsustainable and flawed organizational arrangement to a system that gains knowledge from every care delivery experience and is engineered to promote continuous improvement. In short, the nation needs a health care system that learns, and the committee believes a learning health care system is both possible and necessary for the nation today. This chapter outlines the vision for such a system, highlighting specific characteristics and aims for improvement.
A Learning Health Care System
A learning health care system is one in which science, informatics, incentives, and culture are aligned for continuous improvement and innovation, with best practices seamlessly embedded in the care process, patients and families active participants in all elements, and new knowledge captured as an integral by-product of the care experience. (Roundtable on Value & Science-Driven Health Care, 2012)
As noted in Part I of this report, the supply of knowledge currently available to health care providers and patients has several deficiencies. Providers and patients often lack reliable evidence on the effectiveness of different treatment options, interventions, and technologies and on how the effectiveness of treatments varies for different patients. Moreover, the quality of care depends not only on the effectiveness of a given treatment but also on the way that treatment is delivered. Thus it is necessary to build knowledge about different methods of delivering care and provide clinicians and health care organizations with tools to improve care processes.
Learning processes must also be tailored to the circumstances and needs of the various stakeholders in the health care system. Each stakeholder has a different role in the generation and dissemination of knowledge, so each will need different tools to support continuous learning and improvement. Furthermore, organizations and individuals are at different stages in their learning journey; some have developed advanced systems for continuously improving care (see Chapter 9 for examples), while others are just starting out. New opportunities, such as digital technologies with which to share information and measure progress, can increase the learning potential of every stakeholder. Figure 5-1 illustrates the committee’s vision of how systematically capturing and translating information generated from clinical research and from care delivery can close now open-ended learning loops.
To foster transition to a health care system characterized by continuous learning and improvement, public and private purchasers, health care organizations, clinicians, patients, and other stakeholders should focus their efforts on the foundational elements of a learning health care system, as detailed below and summarized in Table 5-1.
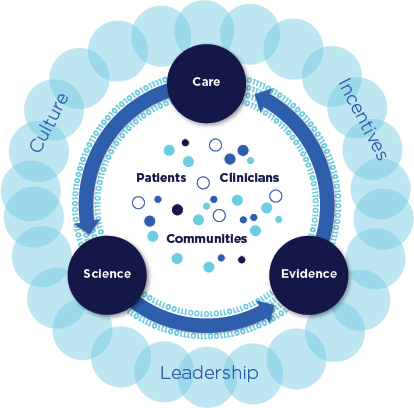
FIGURE 5-1 Schematic of a learning health care system.
Engaged, empowered public and patients. The people served by the health care system—patients, caregivers, and the public—must serve as both the system’s unwavering focus and its fully engaged agents for change. This implies that patient perspectives and needs should be fundamental in the design of health care delivery and in its daily operations. Further, patients and the public should be active contributors, supporters, and actors in the learning process. Yet, currently, the notion of patient-centeredness feels unfamiliar, even disruptive, and the health care culture is not conducive to patient involvement in care—this despite the evidence for positive benefits of such involvement (Berwick, 2009). As noted in prior IOM publications, patients often are limited in their ability to participate as full partners in their health care (IOM, 2001, 2011). Few patients receive clear information on the benefits and potential adverse effects of screenings, tests, treatments, and interventions under consideration for their condition.
TABLE 5-1 Characteristics of a Continuously Learning Health Care System
|
Science and Informatics |
|
Real-time access to knowledge—A learning health care system continuously and reliably captures, curates, and delivers the best available evidence to guide, support, tailor, and improve clinical decision making and care safety and quality. |
|
Digital capture of the care experience—A learning health care system captures the care experience on digital platforms for real-time generation and application of knowledge for care improvement. |
|
Patient-Clinician Partnerships |
|
Engaged, empowered patients—A learning health care system is anchored on patient needs and perspectives and promotes the inclusion of patients, families, and other caregivers as vital members of the continuously learning care team. |
|
Incentives |
|
Incentives aligned for value—A learning health care system has incentives actively aligned to encourage continuous improvement, identify and reduce waste, and reward high-value care. |
|
Full transparency—A learning health care system systematically monitors the safety, quality, processes, prices, costs, and outcomes of care, and makes information available for care improvement and informed choices and decision making by clinicians, patients, and their families. |
|
Continuous Learning Culture |
|
Leadership-instilled culture of learning—A learning health care system is stewarded by leadership committed to a culture of teamwork, collaboration, and adaptability in support of continuous learning as a core aim. |
|
Supportive system competencies—A learning health care system constantly refines complex care operations and processes through ongoing team training and skill building, systems analysis and information development, and creation of the feedback loops for continuous learning and system improvement. |
In contrast, the central focus of a learning health care system is those it serves—patients, their families and caregivers, and the broader public. In a learning health care system, patient needs and perspectives are factored into the design of health care processes, the creation and use of technologies, and the training of clinicians.
To increase the engagement of patients and consumers in health care, it will be necessary to develop new communication strategies that provide understandable evidence on care options and account for individual patient needs, preferences, and capabilities. In addition, tools that allow the patient to be a partner in clinical decisions need to be diffused widely. One way to disseminate these communication strategies and tools is through changes in
clinician education and training. In addition, several new initiatives, such as those centered on participatory medicine, shift the model of health care to one in which patients are key actors in their health and full partners with clinicians in their care. The vision for engaging and empowering patients and the public in a learning health care system is discussed in detail in Chapter 7.
Data infrastructure. The current methods for generating clinical knowledge, while effective in many ways, are slow, cumbersome, and expensive. Yet, the need for knowledge to guide clinical and policy decisions has never been greater. The increasing rate at which new interventions and medical technologies are developed, along with new data on individual variations in conditions and their optimum treatment, requires the development of a new engine for generating clinical knowledge.
An array of clinical effectiveness research strategies, ranging from controlled clinical trials to research drawn from clinical practice, can provide the evidence needed to guide high-quality patient care. Today, more evidence exists about the effectiveness of different treatments and interventions than at any other time in history. But clinical trials often are not structured in a way that delivers the most meaningful results for general clinical use. Despite being expensive and lengthy, large experimental trials frequently generate evidence that may not be applicable to all practice circumstances or patient populations. Trials routinely focus on younger and healthier patients, which introduces uncertainty when the results are extrapolated to real-world patient populations.
In a learning health care system, nimble and efficient approaches, including emerging statistical techniques, research designs, and analytic models that can be applied across all population groups, drive the creation of clinical knowledge. As clinical datasets expand and become more numerous, the potential for generating new insights on the effectiveness of interventions through data mining approaches becomes greater (IOM, 2010). Further, in a true learning system, information is developed as a natural by-product of the care process; knowledge on effectiveness, quality, and value is gained from each patient experience. Increased use of data collected and measured at the point of care, of clinical datasets, and of emerging research techniques in conjunction with traditional research methods can help ensure that research informs the real-world settings of clinical practice. The vision of a robust data infrastructure in a learning health care system is discussed in detail in Chapter 6.
Real-time access to knowledge. Information has transformed modern life. Most individuals are bombarded with information throughout the day, every day. The increasing availability of information has led to widespread societal changes, altered the way governments interact with their citizens, and resulted in extraordinary changes in the way most industries do
business. Many industries have used this access to information to increase their productivity and develop new ways of delivering services.
In the health care sector, a dynamic biomedical research enterprise produces some of the world’s most advanced and innovative clinical discoveries. Unfortunately, important knowledge produced by this research often is not applied to clinical decision making. Recommended practices are delivered only approximately half of the time (McGlynn et al., 2003). One of the major barriers to the consistent application of evidence is the overwhelming quantity of knowledge that is produced every year. The volume and complexity of medical evidence are beyond the capabilities of any individual to aggregate, synthesize, and interpret for clinical practice. The result is uneven quality of care and patient health outcomes.
In a learning health care system, the data, information, and knowledge produced from both biomedical research and clinical encounters is captured, stored, exchanged, and managed using tools that are reliable and secure, and that support continuous quality improvement and health management for a population of patients. New technological tools are used to translate evidence and guidelines into a format that is usable by clinicians and integrated seamlessly at the point of care, such as through clinical decision support software. Finally, patients and their caregivers are engaged in knowledge generation and dissemination through privacy and security policies that build and maintain public trust while incorporating patient-generated data and improving patients’ access to their health information. The vision for increasing clinicians’ and patients’ real-time access to data, information, and knowledge in a learning health care system is discussed in detail in Chapter 6.
Leadership-instilled culture of learning. Strong, visible leadership will be necessary from all sectors of the health care system if the vision of a learning health care system is to be realized (NRC, 2011). For individual health care organizations, leadership has a special significance because it establishes the organization’s vision, communicates its core values, and makes learning and improvement a priority. In addition, health care system leaders help guide the culture of their organization, which has a substantial impact on health outcomes, patient experience, and the satisfaction of employees. A poor culture can present barriers to learning, while a strong culture can drive change (IOM, 2001; Schein, 2004). In promoting safety, for example, the culture must encourage coordination and teamwork among clinicians, as well as promote a nonpunitive environment in which health care professionals feel free to report potential problems. In contrast, the current health care culture is centered on the autonomy of the individual health professional. Clinician expertise is crucial, but this type of culture often leads to a system in which each individual pursues his or her own judgment instead of collaborating to provide the best care for the patient.
The culture of a learning health care system emphasizes teamwork, adaptability, and coordination and strives for continuous learning and improvement. To promote such an environment, health care leaders must know how to influence, support, and measure their organization’s culture. Further, leadership must require visible accountability for improved performance in such areas as quality and safety. This does not mean that leaders must personally spearhead improvement initiatives, but that they must be responsible for devoting resources to such initiatives and supporting the individuals involved. These leadership qualities are not innate to every health care leader and worker; they must be actively taught and reinforced if strong leadership is to become widely available throughout the system.
This is not to say that all elements of the current culture must be reworked. Most physicians, nurses, and other health care professionals are passionate about their work. Every day, in every hospital or clinic across the country, individuals go above and beyond to care for patients, regardless of the system’s limitations. However, changes are necessary to support and augment that passion and dedication. The vision of a leadership-instilled culture of learning in a learning health care system is discussed in detail in Chapter 9.
Competencies that promote continuous improvement. Given the complexity of the health care system and the limits of human capacity, human errors are inevitable. Yet many health care systems are designed and operated under the mistaken assumption that their workers will never err. Adverse events result in part from the variability in the flow of patients through the health care delivery system. And the siloed nature of health care, which boasts hundreds of specialties and often is marked by a lack of communication within and among providers and health care organizations, leads to quality lapses during transitions in patient care.
In a learning health care system, health care organizations design care delivery with an understanding of these limitations. System analysis tools such as root cause analyses and standard protocols for clinical processes are used to identify and overcome human error and support consistent performance. Teamwork and coordination among professionals help integrate care and reduce adverse events at the interfaces between different care processes. Variations in care quality are reduced through the use of variability methodologies and operations management. This type of deliberate system design allows health care providers to harness their strengths—compassion and an emphasis on meeting individual patient needs—more effectively instead of focusing on factors beyond their control. The vision of continuous improvement in a learning health care system is discussed in detail in Chapter 9.
Alignment of incentives. The current health care system fails to support high-value care, and the result has been serious long-term fiscal challenges
for the nation. Health care costs consistently outpace inflation rates; squeeze the budgets of states, employers, and individuals; and reduce individual and family income—all without commensurate health improvements. Medical practice varies significantly from state to state, hospital to hospital, and clinician to clinician, degrading patient care and resulting in uneven quality and safety. Counteracting these trends will require a stronger focus on ways to enhance both health and economic returns from health care investments.
In a learning health care system, the best practices, drawn from research and experience, are the starting point for care. Reliably employing established best practices and building them into routine care leads to system excellence. New technologies provide new opportunities for reducing variations in care, such as through decision support tools. Incentives also are powerful agents for change. To support the transition to a learning health care system, payment incentives must be directly aligned with the goals of a high-quality health care system; promote a focus on the needs of patients and families; and provide the resources and time necessary to support a culture of continuous improvement in the effectiveness, efficiency, and safety of care. Further, a learning health care system fosters value by advancing the science of value incentives so the effects of different payment and incentive models can be better understood. The vision of alignment of incentives in a learning health care system is discussed in detail in Chapter 8.
Transparency. While many definitions of transparency exist, in its basic sense transparency means ensuring that complete, timely, and understandable information is available to support wise decisions. Such information often is missing in the modern health care environment. Yet transparency can be a powerful motivator for change, encouraging providers and organizations to reassess their own practices in order to improve. Most clinicians lack critical data on their own performance and how it relates to that of their peers. Transparency in this regard empowers providers to improve their performance and helps organizations eliminate waste and improve care processes.
Further, patients and consumers lack the information they need to make health care decisions, from which course of medical treatment to pursue to the selection of health care providers. While there are unanswered questions about the best way to present this information to a public audience, the current opacity of the health care system prevents people from discovering basic information, from the cost of a proposed treatment to the average outcomes for a particular intervention. Without meaningful and trustworthy sources of information on costs and outcomes of care, patients and consumers cannot make fully informed decisions. The vision of increased transparency in a learning health care system is discussed in detail in Chapter 8.
THE PATH TO A CONTINUOUSLY LEARNING HEALTH CARE SYSTEM
On each of the dimensions discussed above, the current health care system falls short of its potential. Achieving the core aims of the health care system—better patient health, enhanced experience of care, and improved value from care—will require a fundamental transformation on all these fronts. As outlined in Part I, the imperatives are clear. Too much is spent on health care without concomitant benefits. Equally clear is the path to improvement. Even as the health care system struggles in the face of increasing complexity and costs, it can achieve its potential by transforming into a system that continuously learns and improves. The goal of such a system is to draw on the best evidence in providing care, emphasize prevention and health promotion, continuously improve in value and care quality, and foster advances in the nation’s health.
Yet there are challenges to implementing this vision in real-world clinical environments. Clinicians routinely report moderate or high levels of stress, feel there is not enough time to meet their patients’ needs, and find their work environment chaotic (Burdi and Baker, 1999; Linzer et al., 2009; Trude, 2003). As described in Chapter 2, clinicians struggle to deliver care while confronting inefficient workflows, administrative burdens, and uncoordinated systems. These time pressures, stresses, and inefficiencies limit clinicians from focusing on additional tasks and initiatives, even those that have important goals for improving care. Similarly, professionals working in health care organizations are overwhelmed by the sheer volume of initiatives currently under way to improve various aspects of the care process, initiatives that appear to be unconnected with the organization’s priorities. Often, these initiatives may be successful in one setting yet may not translate to other parts of the same organization.
Given such real-world impediments, initiatives that focus merely on incremental improvements and add to a clinician’s daily workload are unlikely to succeed. Just as the quantity of clinical information now available exceeds the capacity of any individual to absorb and apply it, the number of tasks needed for regular care outstrips the capabilities of any individual. Rather, significant improvements can occur only if the environment, context, and systems in which these professionals practice are reconfigured. Strategies for building this type of system that supports clinicians’ efforts focus on three major areas: providing the foundations for learning, establishing a suitable environment for improvement, and ensuring that learning focuses on the right targets. Essential as well are expanding the evidence base to ensure that clinicians have the information they need, expanding the capacity to capture patient data in digital records, and developing metrics for assessing different aspects of learning and improvement. In creating a
supportive environment, the levers for change include developing incentives that promote improvement, ensuring that payment and contracting policies support learning, promoting transparency that helps clinicians and patients make informed decisions, and building cultures that encourage improvement. Finally, focusing learning on the right targets requires approaches for engaging patients to ensure that care addresses their needs, goals, and circumstances. Part III of this report explores each of these strategies in more detail.
Berwick, D. M. 2009. What “patient-centered” should mean: Confessions of an extremist. Health Affairs (Millwood) 28(4):w555-w565.
Beta-Blocker Heart Attack Trial Research Group. 1982. A randomized trial of propranolol in patients with acute myocardial infarction. I. Mortality results. Journal of the American Medical Association 247(12):1707-1714.
Bradley, E. H., E. S. Holmboe, J. A. Mattera, S. A. Roumanis, M. J. Radford, and H. M. Krumholz. 2001. A qualitative study of increasing beta-blocker use after myocardial infarction: Why do some hospitals succeed? Journal of the American Medical Association 285(20):2604-2611.
Brand, D. A., L. N. Newcomer, A. Freiburger, and H. Tian. 1995. Cardiologists’ practices compared with practice guidelines: Use of beta-blockade after acute myocardial infarction. Journal of the American College of Cardiology 26(6):1432-1436.
Burdi, M. D., and L. C. Baker. 1999. Physicians’ perceptions of autonomy and satisfaction in California. Health Affairs 18(4):134.
Burwen, D. R., D. H. Galusha, J. M. Lewis, M. R. Bedinger, M. J. Radford, H. M. Krumholz, and J. M. Foody. 2003. National and state trends in quality of care for acute myocardial infarction between 1994-1995 and 1998-1999: The Medicare health care quality improvement program. Archives of Internal Medicine 163(12):1430-1439.
Chassin, M. R., and J. M. Loeb. 2011. The ongoing quality improvement journey: Next stop, high reliability. Health Affairs (Millwood) 30(4):559-568.
Gosfield, A. G., and J. L. Reinertsen. 2005. The 100,000 lives campaign: Crystallizing standards of care for hospitals. Health Affairs (Millwood) 24(6):1560-1570.
Gottlieb, S. S., R. J. McCarter, and R. A. Vogel. 1998. Effect of beta-blockade on mortality among high-risk and low-risk patients after myocardial infarction. New England Journal of Medicine 339(8):489-497.
IOM (Institute of Medicine). 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
IOM. 2010. Redesigning the clinical effectiveness research paradigm: Innovation and practice-based approaches: Workshop summary. Washington, DC: The National Academies Press.
IOM. 2011. Patients charting the course: Citizen engagement in the learning health system: Workshop summary. Washington, DC: The National Academies Press.
Krumholz, H. M., M. J. Radford, Y. Wang, J. Chen, A. Heiat, and T. A. Marciniak. 1998. National use and effectiveness of beta-blockers for the treatment of elderly patients after acute myocardial infarction: National cooperative cardiovascular project. Journal of the American Medical Association 280(7):623-629.
Lee, T. H. 2007. Eulogy for a quality measure. New England Journal of Medicine 357(12):1175-1177.
Linzer, M., L. B. Manwell, E. S. Williams, J. A. Bobula, R. L. Brown, A. B. Varkey, B. Man, J. E. McMurray, A. Maguire, B. Horner-Ibler, M. D. Schwartz, and MEMO (Minimizing Error, Maximizing Outcome) Investigators. 2009. Working conditions in primary care: Physician reactions and care quality. Annals of Internal Medicine 151(1):28-36, W26-W29.
Marciniak, T. A., E. F. Ellerbeck, M. J. Radford, T. F. Kresowik, J. A. Gold, H. M. Krumholz, C. I. Kiefe, R. M. Allman, R. A. Vogel, and S. F. Jencks. 1998. Improving the quality of care for medicare patients with acute myocardial infarction: Results from the cooperative cardiovascular project. Journal of the American Medical Association 279(17):1351-1357.
McGlynn, E. A., S. M. Asch, J. Adams, J. Keesey, J. Hicks, A. DeCristofaro, and E. A. Kerr. 2003. The quality of health care delivered to adults in the United States. New England Journal of Medicine 348(26):2635-2645.
NRC (National Research Council). 2011. Strategies and priorities for information technology at the Centers for Medicare & Medicaid Services. Washington, DC: The National Academies Press.
Roundtable on Value & Science-Driven Health Care. 2012. The Roundtable. Washington, DC: Institute of Medicine.
Ryan, T. J., J. L. Anderson, E. M. Antman, B. A. Braniff, N. H. Brooks, R. M. Califf, L. D. Hillis, L. F. Hiratzka, E. Rapaport, B. J. Riegel, R. O. Russell, E. E. Smith Iii, W. D. Weaver, J. L. Ritchie, M. D. Cheitlin, K. A. Eagle, T. J. Gardner, J. A. Garson, R. J. Gibbons, R. P. Lewis, and R. A. O’Rourke. 1996. ACC/AHA guidelines for the management of patients with acute myocardial infarction: A report of the American College of Cardiology/American Heart Association task force on practice guidelines (committee on management of acute myocardial infarction). Journal of the American College of Cardiology 28(5):1328-1419.
Ryan, T. J., E. M. Antman, N. H. Brooks, R. M. Califf, L. D. Hillis, L. F. Hiratzka, E. Rapaport, B. Riegel, R. O. Russell, E. E. Smith, W. D. Weaver, R. J. Gibbons, J. S. K. A. Eagle, T. J. Gardner, A. Garson, G. Gregoratos, and S. C. Smith. 1999. 1999 update: ACC/AHA guidelines for the management of patients with acute myocardial infarction: Executive summary and recommendations: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on Management of Acute Myocardial Infarction). Circulation 100(9):1016-1030.
Schein, E. H. 2004. Organizational culture and leadership. San Francisco, CA: Jossey-Bass.
Trude, S. 2003. So much to do, so little time: Physician capacity constraints, 1997-2001. Tracking Report/Center for Studying Health System Change (8):1.
Yusuf, S., R. Peto, J. Lewis, R. Collins, and P. Sleight. 1985. Beta blockade during and after myocardial infarction: An overview of the randomized trials. Progress in Cardiovascular Diseases 27(5):335-371.














