Health care in America presents a fundamental paradox. The past 50 years have seen an explosion in biomedical knowledge, dramatic innovation in therapies and surgical procedures, and management of conditions that previously were fatal, with ever more exciting clinical capabilities on the horizon. Yet, American health care is falling short on basic dimensions of quality, outcomes, costs, and equity. Available knowledge is too rarely applied to improve the care experience, and information generated by the care experience is too rarely gathered to improve the knowledge available. The traditional systems for transmitting new knowledge—the ways clinicians are educated, deployed, rewarded, and updated—can no longer keep pace with scientific advances. If unaddressed, the current shortfalls in the performance of the nation’s health care system will deepen on both quality and cost dimensions, challenging the well-being of Americans now and potentially far into the future.
Consider the impact on American services if other industries routinely operated in the same manner as many aspects of health care:
- If banking were like health care, automated teller machine (ATM) transactions would take not seconds but perhaps days or longer as a result of unavailable or misplaced records.
- If home building were like health care, carpenters, electricians, and plumbers each would work with different blueprints, with very little coordination.
- If shopping were like health care, product prices would not be posted, and the price charged would vary widely within the same store, depending on the source of payment.
- If automobile manufacturing were like health care, warranties for cars that require manufacturers to pay for defects would not exist. As a result, few factories would seek to monitor and improve production line performance and product quality.
- If airline travel were like health care, each pilot would be free to design his or her own preflight safety check, or not to perform one at all.
The point is not that health care can or should function in precisely the same way as all other sectors of people’s lives—each is very different from the others, and every industry has room for improvement. Yet, if some of the transferable best practices from banking, construction, retailing, automobile manufacturing, flight safety, public utilities, and personal services were adopted as standard best practices in health care, the nation could see patient care in which
- records would be immediately updated and available for use by patients;
- care delivered would be proven reliable at the core and tailored at the margins;
- patient and family needs and preferences would be a central part of the decision process;
- all team members would be fully informed in real time about each other’s activities;
- prices and total costs would be fully transparent to all participants;
- payment incentives were structured to reward outcomes and value, not volume;
- errors would be promptly identified and corrected; and
- results would be routinely captured and used for continuous improvement.
Unfortunately, these are not features that would describe much of health care in America today. Health care can lag behind many other sectors with respect to its ability to meet patients’ specific needs, to offer choice, to adapt, to become more affordable, to improve—in short, to learn. Americans should be served by a health care system that consistently delivers reliable performance and constantly improves, systematically and seamlessly, with each care experience and transition.
In the face of these realities, the Institute of Medicine (IOM) convened the Committee on the Learning Health Care System in America to explore
the most fundamental challenges to health care today and to propose actions that can be taken to achieve a health care system characterized by continuous learning and improvement. This study builds on earlier IOM studies on various aspects of the health care system, from To Err Is Human: Building a Safer Health System (1999), on patient safety; to Crossing the Quality Chasm: A New Health System for the 21st Century (2001a), on health care quality; to Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care (2003), on health care disparities. The study process was also facilitated and informed by the published summaries of workshops conducted under the auspices of the IOM Roundtable on Value & Science-Driven Health Care. Over the past 6 years, 11 workshop summaries have been produced, exploring various aspects of the challenges and opportunities in health care today, with a particular focus on the foundational elements of a learning health system.
Meeting the challenges discussed at those workshops has taken on great urgency as a result of two overarching imperatives:
- to manage the health care system’s ever-increasing complexity, and
- to curb ever-escalating costs.
The convergence of these imperatives makes the status quo untenable. At the same time, however, opportunities exist to address these problems—opportunities that did not exist even a decade ago:
- vast computational power that is affordable and widely available;
- connectivity that allows information to be accessed in real time virtually anywhere;
- human and organizational capabilities that improve the reliability and efficiency of care processes; and
- the recognition that effective care must be delivered by collaborations between teams of clinicians and patients, each playing a vital role in the care process.
The committee undertook its work to consider how these opportunities for best care at lower cost can be leveraged to meet the challenges outlined above. The committee, whose work was supported by the Blue Shield of California Foundation, the Charina Endowment Fund, and the Robert Wood Johnson Foundation, was charged with (1) identifying how the effectiveness and efficiency of the current health care system can be transformed through tools and incentives for continuous assessment and improvement and (2) developing recommendations for actions that can be taken to that end. This report explores the imperatives for change, describes the emerging tools that make transformation possible, sets forth a vision
for a continuously learning health care system, and delineates a path for achieving this vision. Detailed findings are presented throughout the report, together with the conclusions and recommendations they support, which are also highlighted in this summary.
The title of the report underscores that care that is based on the best available evidence, takes appropriate account of individual preferences, and is delivered reliably and efficiently—best care—is possible today. When such care is routinely implemented, moreover, it is generally less expensive than the less effective, less efficient care that is now too commonly provided. Moreover, the transition to best care envisioned in this report is urgently needed given the budgetary, economic, and health pressures facing the nation’s health care system.
Decades of rapid innovation and technological improvement have created an extraordinarily complex health care system. Clinicians and health care staff work tirelessly to care for their patients in an increasingly complex, inefficient, and stressful environment. Certain breakthrough innovations have benefited millions of patients, but the aggregate impact of the flood of new interventions has introduced challenges for both clinicians and patients in treating and managing health conditions. In addition to the challenge of complexity, and in part because of it, health care often falls short of its potential in the quality of care delivered and the patient outcomes achieved. These shortfalls are occurring even as costs are rising to unsustainable levels. Additionally, new opportunities emerging from technology, industry, and policy can be leveraged to help mold the system into one characterized by continuous learning and improvement. In this context, the committee identified three imperatives for achieving a continuously learning health care system that provides the best care at lower cost: (1) managing rapidly increasing complexity; (2) achieving greater value in health care; and (3) capturing opportunities from technology, industry, and policy.
Managing Rapidly Increasing Complexity
The complexity of health care has increased in multiple dimensions—in the ever-increasing treatment, diagnostic, and care management options available; in the rapidly rising levels of biomedical and clinical evidence; and in administrative complexities, from complicated workflows to fragmented financing. The complexity due to ever-increasing treatment options can be illustrated by the evolution of care for two common conditions—heart disease and cancer. During much of the twentieth century, heart attacks commonly were treated with weeks of bed rest. Today, advanced diagnostics allow for customized treatments for patients; interventions
such as percutaneous coronary interventions and coronary artery bypass grafts can reopen blocked vessels and restore blood flow to the heart; and pharmaceutical therapies, such as thrombolytics and beta-blockers, improve survival and reduce the chances of subsequent heart attacks (Certo, 1985; Nabel and Braunwald, 2012). Similarly, five decades ago, breast cancer was detected from a physical exam, and mastectomy was the recommended treatment. Today, multiple imaging technologies exist for the detection and diagnosis of the disease, and once diagnosed, the cancer can be further classified and treated according to genetic characteristics and hormone receptor status (Harrison, 1962; IOM, 2001b; Kasper and Harrison, 2005).
As a result of improved scientific understanding, new treatments and interventions, and new diagnostic technologies, the U.S. health care system now is characterized by more to do, more to know, and more to manage than at any time in history. As one quantification of this increase, the volume of the biomedical and clinical knowledge base has rapidly expanded, with research publications having risen from more than 200,000 per year in 1970 to more than 750,000 in 2010 (see Figure S-1). The result is a
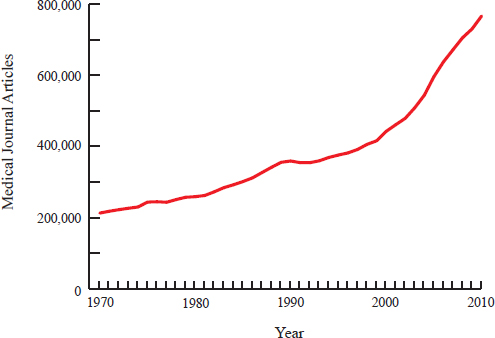
FIGURE S-1 Number of journal articles published on health care topics per year from 1970 to 2010. Publications have increased steadily over 40 years, with the rate of increase becoming more pronounced starting approximately in 2000.
SOURCE: Data obtained from online searches at PubMed: http://www.ncbi.nlm.nih.gov/pubmed/.
paradox: advances in science and technology have improved the ability of the health care system to treat diseases, yet the sheer volume of new discoveries stresses the capabilities of the system to effectively generate and manage knowledge and apply it to regular care. These advances have occurred at the same time as, and sometimes have contributed to, challenges in health care quality and value.
Conclusion: Diagnostic and treatment options are expanding and changing at an accelerating rate, placing new stresses on clinicians and patients, as well as potentially impacting the effectiveness and efficiency of care delivery.
Beyond the increasing stores of biomedical and clinical knowledge, changes in disease prevalence and patient demographics have altered the landscape for care delivery. The prevalence of chronic conditions, for example, has increased over time. In 2000, 125 million people suffered from such conditions; by 2020, that number is projected to grow to an estimated 157 million (Anderson, 2010). The role of chronic diseases has changed as the demographics of the population have shifted. In general, the population has gotten older; in the past decade, the portion of the population over age 65 has increased at 1.5 times the rate of the rest of the population (Howden and Meyer, 2011). Almost half of those over 65 receive treatment for at least one chronic disease, and more than 20 percent receive treatment for multiple chronic diseases (Schneider et al., 2009); fully 75 million people in the United States have multiple chronic conditions (Parekh and Barton, 2010).
Managing these multiple conditions requires a holistic approach, because the use of various clinical practice guidelines developed for single diseases may have adverse effects (Boyd et al., 2005a; Parekh and Barton, 2010; Tinetti et al., 2004). For example, existing clinical practice guidelines would suggest that a hypothetical 79-year-old woman with osteoporosis, osteoarthritis, type 2 diabetes, hypertension, and chronic obstructive pulmonary disease should take as many as 19 doses of medication per day. Such guidelines might also make conflicting recommendations for the care. If she had peripheral neuropathy, guidelines for osteoporosis would recommend that she perform weight-bearing exercise, while guidelines for diabetes would recommend that she avoid such exercise (Boyd et al., 2005a). These situations create uncertainty for clinicians and patients as to the best course of action to pursue as they attempt to manage the treatments for multiple conditions.
Conclusion: Chronic diseases and comorbid conditions are increasing, exacerbating the clinical, logistical, decision-making, and economic challenges faced by patients and clinicians.
Care delivery also has become increasingly demanding. It would take an estimated 21 hours per day for individual primary care physicians to provide all of the care recommended to meet their patients’ acute, preventive, and chronic disease management needs (Yarnall et al., 2009). Clinicians in intensive care units, who care for the sickest patients in a hospital, must manage in the range of 180 activities per patient per day—from replacing intravenous fluids, to administering drugs, to monitoring patients’ vital signs (Donchin et al., 2003). In addition, rising administrative burdens and inefficient workflows mean that hospital nurses spend only about 30 percent of their time in direct patient care (Hendrich et al., 2008; Hendrickson et al., 1990; Tucker and Spear, 2006). These pressures are not limited to clinicians; patients often find the health care system uncoordinated, opaque, and stressful to navigate. One study found that for 1 of every 14 tests, either the patient was not informed of a clinically significant abnormal test result, or the clinician failed to record reporting the result to the patient (Casalino et al., 2009).
With specialization, moreover, clinicians must coordinate with multiple other providers; for their health care, Medicare patients now see an average of seven physicians, including five specialists, split among four different practices (Pham et al., 2007). One study found that in a single year, a typical primary care physician coordinated with an average of 229 other physicians in 117 different practices just for Medicare patients (Pham et al., 2009). The involvement of multiple providers tends to blur accountability. One survey found that 75 percent of hospital patients were unable to identify the clinician in charge of their care (Arora et al., 2009).
Conclusion: Care delivery has become increasingly fragmented, leading to coordination and communication challenges for patients and clinicians.
Achieving Greater Value in Health Care
In addition to, and sometimes as a result of, the challenge of complexity, health care quality and outcomes often fall short of their potential. A decade after the IOM (1999) estimated that 44,000 to 98,000 patients died each year from preventable medical errors, recent studies have reported that as many as one-third of hospitalized patients may experience harm or an adverse event, often from preventable errors (Classen et al., 2011; Landrigan et al., 2010; Levinson, 2010). While infections and complications once were viewed as routine consequences of medical care, it is now recognized that strategies and evidence-based interventions exist that can significantly reduce the incidence and severity of such events.
Similarly, medical care often is guided insufficiently by evidence, with Americans receiving only about half of the preventive, acute, and chronic care recommended by current research and evidence-based guidelines (McGlynn et al., 2003). Sometimes this occurs because available evidence is not applied to clinical care, while in other cases evidence is not available.
As a result of all of these factors, the nature and quality of health care vary considerably among states, with serious health and economic consequences. If all states could provide care of the quality delivered by the highest-performing state, an estimated 75,000 fewer deaths would have occurred across the country in 2005 (McCarthy et al., 2009; Schoenbaum et al., 2011).
Conclusion: Health care safety, quality, and outcomes for Americans fall substantially short of their potential and vary significantly for different populations of Americans.
These deficiencies in care quality have occurred even as expenses have risen significantly. Health care costs1 have increased at a greater rate than the economy as a whole for 31 of the past 40 years, and now constitute 18 percent of the nation’s gross domestic product (CMS, 2012; Keehan et al., 2011). The growth in health care costs has contributed to stagnation in real income for American families. Although income has increased by 30 percent over the past decade, these gains have effectively been eliminated by a 76 percent increase in health care costs (Auerbach and Kellermann, 2011). These high costs have strained families’ budgets and put health insurance coverage out of reach for many, contributing to the 50 million Americans without coverage (DeNavas-Walt et al., 2011).
In addition to unsustainable cost growth, there is evidence that a substantial proportion of health care expenditures is wasted, leading to little improvement in health or in the quality of care. Estimates vary on waste and excess health care costs, but they are large. The IOM workshop summary The Healthcare Imperative: Lowering Costs and Improving Outcomes contains estimates of excess costs in six domains: unnecessary services, services inefficiently delivered, prices that are too high, excess administrative costs, missed prevention opportunities, and medical fraud (IOM, 2010). These estimates, presented by workshop speakers with respect to their areas of expertise and based on assumptions from limited observations, suggest the
___________________________________________________
1In this report, price refers to the amount charged for a given health care service or product. It is important to note that there are frequently multiple prices for the same service or product, depending on the patient’s insurance status and payer, as well as other factors. Cost is the total sum of money spent at a given level (episodes, patients, organizations, state, national), or price multiplied by the volume of services or products used.
substantial contribution of each domain to excessive health care costs (see Table S-1).
Although these estimates have unknown overlap, the sum of the individual estimates—$765 billion—suggests the significant scale of waste in the system. Two other independent and differing analytic approaches—considering regional variation in costs and comparing costs across countries—produce similar estimates, with total excess costs approaching $750 billion in 2009 (Farrell et al., 2008; IOM, 2010; Wennberg et al., 2002).
TABLE S-1 Estimated Sources of Excess Costs in Health Care (2009)
| Category | Sources | Estimate of Excess Costs |
| Unnecessary Services |
|
$210 billion |
| Inefficiently Delivered Services |
|
$130 billion |
| Excess Administrative Costs |
|
$190 billion |
| Prices That Are Too High |
|
$105 billion |
| Missed Prevention Opportunities |
|
$55 billion |
| Fraud |
|
$75 billion |
SOURCE: Adapted with permission from IOM, 2010.
While there are methodological issues with each method for estimating excess costs, the consistently large figures produced by each signal the potential for reducing health care costs while improving quality and health outcomes.
At this level, unnecessary health care costs and waste exceed the 2009 budget for the Department of Defense by more than $100 billion (OMB, 2010). Health care waste also amounts to more than 1.5 times the nation’s total infrastructure investment in 2004, including roads, railroads, aviation, drinking water, telecommunications, and other structures.2 To put these estimates in the context of health care expenditures, the estimated redirected funds could provide health insurance coverage for more than 150 million workers (including both employer and employee contributions), which exceeds the 2009 civilian labor force.3 And the total projected amounts could pay the salaries of all of the nation’s first response personnel, including firefighters, police officers, and emergency medical technicians, for more than 12 years.4
Conclusion: The growth rate of health care expenditures is unsustainable, with waste that diverts major resources from necessary care and other priorities at every level—individual, family, community, state, and national.
In sum, as illustrated in Figure S-2, each stage in the processes that shape the health care received—knowledge development, translation into medical evidence, application of evidence-based care—has prominent shortcomings and inefficiencies that contribute to a large reservoir of missed opportunities, waste, and harm. The threats to the health and economic security of Americans are clear, present, and compelling.
___________________________________________________
2The Department of Defense budget was calculated from the fiscal year 2009 outlays listed in the Fiscal Year 2011 U.S. Government Budget (OMB, 2010); the comparison of health care waste with the national infrastructure investment was drawn from a Congressional Budget Office analysis, with inflation adjusted according to the Consumer Price Index (CPI) (Congressional Budget Office, 2008).
3The average premiums for a single worker were calculated using the Kaiser Family Foundation’s 2009 Employer Health Benefits survey, with the size of the civilian labor force drawn from Bureau of Labor Statistics estimates for 2009 (Kaiser Family Foundation and Health Research & Educational Trust, 2009; U.S. Bureau of Labor Statistics, 2012).
4The comparison with expenditures on first responders was calculated from the annual salary data for firefighters, police officers, and emergency medical technicians provided in the 2009 National Compensation Survey, while the total number of individuals in those occupations was drawn from the 2009 Occupational Employment Statistics (U.S. Bureau of Labor Statistics, 2010a,b).
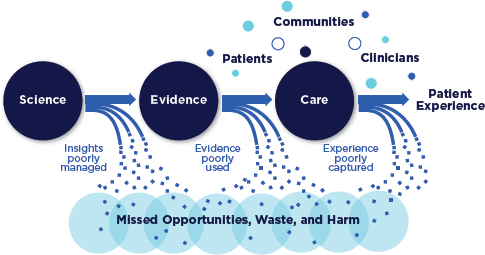
FIGURE S-2 Schematic of the health care system today.
Capturing Opportunities from Technology, Industry, and Policy
As noted earlier, new opportunities exist to address the challenges outlined above. Just as the information revolution has transformed many other fields, growing stores of data and computational abilities hold the same promise for improving clinical research, clinical practice, and clinical decision making. In the past three decades, for example, computer processing speed has grown by 60 percent per year on average, while the capacity to share information over telecommunications networks has risen by an average of 30 percent per year (Hilbert and López, 2011). These advances in computing and connectivity have the potential to improve health care by expanding the reach of knowledge, increasing access to clinical information when and where needed, and assisting patients and providers in managing chronic diseases. Studies also have found that using such electronic systems can improve safety—one study reported a 41 percent reduction in potential adverse drug events following the implementation of a computerized patient management system (computerized physician order entry, or CPOE), while another estimated that overall medication error rates dropped by 81 percent (Bates et al., 1998, 1999; Potts et al., 2004). Projections are for 90 percent of office-based physicians to have access to fully operational electronic health records by 2019, up from 34 percent in 2011 (Congressional Budget Office, 2009; Hsiao et al., 2011). Because these capacities are relatively early in their development in the health care arena, there is substantial room for progress as they are implemented in the field. However,
multiple nontechnological developments, such as supportive care processes, governance, and patient and public engagement, will be necessary if these technologies are to reach their full potential.
Conclusion: Advances in computing, information science, and connectivity can improve patient-clinician communication, point-of-care guidance, the capture of experience, population surveillance, planning and evaluation, and the generation of real-time knowledge—features of a continuously learning health care system.
In addition to advances in computing and connectivity, new organizational capabilities have been developed in diverse industries to improve safety, quality, reliability, and value. Advances in safety alone, for instance, enabled domestic commercial commuter airlines to report no fatalities from 2007 to 2010 (Bureau of Transportation Statistics, 2011). New capabilities in systems engineering, operations management, and production can be adapted to health care settings to improve performance. In one study, the use of checklists inspired by the aviation industry eliminated catheter-related bloodstream infections in the intensive care units of most hospitals in the study and resulted in an 80 percent decrease in infections per catheter-day (Pronovost et al., 2006, 2009). Commercial strategies to improve the reliability of the delivery of goods and services have potential applicability to health care as well. A pharmacy unit, for example, undertook systematic problem solving and reduced the time spent searching for medications by 30 percent and the frequency of out-of-stock medications by 85 percent (Spear, 2005).
Conclusion: Systematic, evidence-based process improvement methods applied in various sectors to achieve often striking results in safety, quality, reliability, and value can be similarly transformative for health care.
Across the United States, moreover, there is growing momentum to implement novel partnerships and collaborations to test delivery system innovations aimed at high-value, high-quality health care. In many settings, stakeholders at all levels—federal, state, and local governments; public and private insurers; health care delivery organizations; employers; patients and consumers; and others—are working together with the shared objectives of controlling health care costs and improving health care quality. States ranging from Massachusetts to Utah to Vermont have introduced new initiatives aimed at expanding health insurance coverage, improving care quality and value, and advancing the overall health of their residents. Multiple initiatives by employers, specialty societies, patient and consumer
groups, health care delivery organizations, health plans, and others—such as the American Board of Internal Medicine (ABIM) Foundation’s Choosing Wisely® campaign and the Good Stewardship project—are focused on improving the health care system. Other initiatives currently under way range from the Patient-Centered Primary Care Collaborative, which seeks to spread patient-centered medical homes; to community-based initiatives, such as the Aligning Forces for Quality program and the Chartered Value Exchange project; to all-payer databases being established in various states around the country. And drawing on their experiences in improving outcomes and lowering costs through initiatives in their own institutions, a group of health care delivery leaders has developed “A CEO Checklist for High-Value Health Care,” which describes system-change approaches that can be adopted in most health care settings to improve outcomes and reduce costs of care (Cosgrove et al., 2012) (see Appendix B). The convergence of these novel partnerships, a changing health care landscape, and investments in knowledge infrastructure has created a unique opportunity to achieve continuously learning health care.
Conclusion: Innovative public- and private-sector health system improvement initiatives, if adopted broadly, could support many elements of the transformation necessary to achieve a continuously learning health care system.
The committee believes that achieving a learning health care system—one in which science and informatics, patient-clinician partnerships, incentives, and culture are aligned to promote and enable continuous and real-time improvement in both the effectiveness and efficiency of care—is both necessary and possible for the nation. Table S-2 lists the fundamental characteristics of such a system, according to the major dimensions in play.
There are challenges to implementing this vision in real-world clinical environments. Clinicians routinely report moderate or high levels of stress, feel there is not enough time to meet their patients’ needs, and find their work environment chaotic (Burdi and Baker, 1999; Linzer et al., 2009; Trude, 2003). Furthermore, they struggle to deliver care while confronting inefficient workflows, administrative burdens, and uncoordinated systems. These time pressures, stresses, and inefficiencies prevent clinicians from focusing on additional tasks and initiatives, even those that have important goals for improving care. Similarly, professionals working in health care organizations are overwhelmed by the sheer volume of initiatives currently under way to improve various aspects of the care process, initiatives that appear to be unconnected with the organization’s priorities. Often, these
TABLE S-2 Characteristics of a Continuously Learning Health Care System
|
Science and Informatics |
|
Real-time access to knowledge—A learning health care system continuously and reliably captures, curates, and delivers the best available evidence to guide, support, tailor, and improve clinical decision making and care safety and quality. |
|
Digital capture of the care experience—A learning health care system captures the care experience on digital platforms for real-time generation and application of knowledge for care improvement. |
|
Patient-Clinician Partnerships |
|
Engaged, empowered patients—A learning health care system is anchored on patient needs and perspectives and promotes the inclusion of patients, families, and other caregivers as vital members of the continuously learning care team. |
|
Incentives |
|
Incentives aligned for value—A learning health care system has incentives actively aligned to encourage continuous improvement, identify and reduce waste, and reward high-value care. |
|
Full transparency—A learning health care system systematically monitors the safety, quality, processes, prices, costs, and outcomes of care, and makes information available for care improvement and informed choices and decision making by clinicians, patients, and their families. |
|
Continuous Learning Culture |
|
Leadership-instilled culture of learning—A learning health care system is stewarded by leadership committed to a culture of teamwork, collaboration, and adaptability in support of continuous learning as a core aim. |
|
Supportive system competencies—A learning health care system constantly refines complex care operations and processes through ongoing team training and skill building, systems analysis and information development, and creation of the feedback loops for continuous learning and system improvement. |
initiatives may be successful in one setting yet may not translate to other parts of the same organization.
Given such real-world impediments, initiatives that focus merely on incremental improvements and add to a clinician’s daily workload are unlikely to succeed. Just as the quantity of clinical information now available exceeds the capacity of any individual to absorb and apply it, the number of tasks needed for regular care outstrips the capabilities of any individual. Significant change can occur only if the environment, context, and systems in which these professionals practice are reconfigured so that the entire health care infrastructure and culture support learning and improvement.
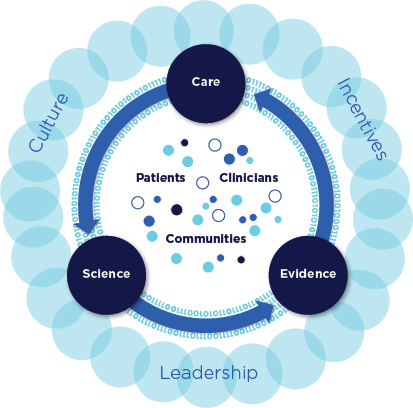
FIGURE S-3 Schematic of the continuously learning health care system.
Figure S-3 illustrates the committee’s vision of how systematically capturing and translating information generated by clinical research and care delivery can close now open-ended learning loops.
The path to achieving the vision of a learning health care system entails generating and using real-time knowledge to improve outcomes; engaging patients, families, and communities; achieving and rewarding high-value care; and creating a new culture of care.
Generating and Using Real-Time Knowledge to Improve Outcomes
Although unprecedented and increasing levels of information are available in journals, guidelines, and other sources, patients and clinicians often
lack practical access to guidance that is relevant, timely, and useful for the circumstances at hand. For example, fewer than half of the clinical guidelines for the nine most common chronic conditions consider older patients with multiple comorbid chronic conditions, even though, as noted earlier, 75 million Americans fall in that category (Boyd et al., 2005b; Parekh and Barton, 2010). In the case of localized prostate cancer, for instance, which treatment works best for a given patient—from watchful waiting, to radical prostatectomy, to radiation and chemotherapy—is unknown. Furthermore, the evidence base for clinical guidelines and recommendations needs to be strengthened. In some cases, 40 to 50 percent of the recommendations made in guidelines are based on expert opinion, case studies, or standards of care rather than on more systematic trials and studies (Chauhan et al., 2006; IOM, 2008, 2011a; Tricoci et al., 2009).
New methods are needed to address current limitations in clinical research. The cost of current clinical research methods averages $15-$20 million for larger studies—and much more for some—yet there are concerns about generalizing study results to all practice conditions and patient populations (Holve and Pittman, 2009, 2011). Given the increasing number of new medical treatments and technologies, the complexity of managing multiple chronic diseases, and the growing personalization of treatments and diagnostics, the challenge is to produce and deliver practical evidence that clinicians and patients can apply to clinical questions.
Conclusion: Despite the accelerating pace of scientific discovery, the current clinical research enterprise does not sufficiently address pressing clinical questions. The result is decisions by both patients and clinicians that are inadequately informed by evidence.
Meeting this challenge will require new approaches for generating clinical evidence that reduce the expense and effort of conducting research and improve the clinical applicability of research findings while retaining the rigorous reliability of the process. The issue is not determining which research method is best for a particular condition, but which method provides the information most appropriate to a particular clinical need. Each study must be well tailored to provide useful, practical, and reliable results for the condition at hand.
Opportunities for achieving these aims leverage the expanded capacity of the digital infrastructure along with new statistical and research techniques. Computational capabilities present promising, as yet unrealized, opportunities for care improvement, while advances in statistical analysis, simulation, and modeling can supplement traditional methods for conducting trials. The application of computing capacity and new analytic approaches enables the development of real-time research insights from
patient populations. For example, one study found that real-time analysis of clinical data from electronic health records could have identified the increased risk of heart attack associated with one diabetes drug within 18 months of its introduction, as opposed to the 7-8 years between the medication’s introduction and the point at which concerns were raised publicly (Brownstein et al., 2010). Computational capabilities also hold promise for hastening the derivation of important new insights from the care experience. A comprehensive disease registry for heart attack patients in Sweden, for example, has contributed to a 65 percent reduction in 30-day mortality and a 49 percent decrease in 1-year mortality from heart attacks (Larsson et al., 2012).
Conclusion: Growing computational capabilities to generate, communicate, and apply new knowledge create the potential to build a clinical data infrastructure to support continuous learning and improvement in health care.
Harnessing this potential for care improvement will require systematic approaches that address the regulatory, commercial, communications, and technological challenges involved. Results of surveys of health researchers suggest that the current formulation and interpretation of privacy rules have increased the cost and time to conduct research, impeded collaboration, and hampered the recruiting of subjects (IOM, 2009; Ness, 2007). Privacy is a highly important societal and personal value, but the current rules, with their inconsistent interpretation, offer a relatively limited security advantage to patients while impeding the pace and scope of new insights from health research and care improvement.
Conclusion: Regulations governing the collection and use of clinical data often create unnecessary and unintended barriers to the effectiveness and improvement of care and the derivation of research insights.
The current system for capturing and using new knowledge is already flawed and, absent change, is likely to be overwhelmed by the pace of knowledge growth. The diffusion of new evidence can take considerable time; in the case of thrombolytic drugs for heart attack treatment, for example, 13 years elapsed between when they were shown to be effective and when most experts recommended the treatment (Antman et al., 1992). Substantial work is required to identify high-quality evidence that minimizes the risk of contradiction by later studies and is sufficiently robust to provide insight on application to a particular patient’s clinical circumstances. This is time-consuming work, which goes on while clinical patterns are being formed.
Realizing the prospect of faster, deeper knowledge bases will require parallel advances in the approaches to gathering and assessing evidence, making evidence-based recommendations, translating those recommendations to practice, and reinforcing their use through relevant policies. Computing capacity can help with assessment as well as dissemination. Technological tools, such as decision support tools that can be broadly embedded in electronic health records, hold promise for improving the application of evidence. One study found that digital decision support tools helped clinicians apply clinical guidelines, improving health outcomes for diabetics by 15 percent (Cebul et al., 2011).
Conclusion: As the pace of knowledge generation accelerates, new approaches are needed to deliver the right information, in a clear and understandable format, to patients and clinicians as they partner to make clinical decisions.
Engaging Patients, Families, and Communities5
The structure, incentives, and culture of the health care system are poorly aligned to engage patients and respond to their needs. While clinicians supply information and advice based on their scientific expertise in treatment and intervention options, as well as potential outcomes, patients, their families, and other caregivers bring personal knowledge regarding the suitability—or lack thereof—of different treatments for the patient’s circumstances and preferences. Information from both sources is needed to select the right care option, particularly because studies have found that patients and clinicians have differing views on the importance of different health goals and health care risks (Lee et al., 2010a,b). At the same time, it is important to note that patient-centered care does not mean simply agreeing to every patient request. Rather, it entails meaningful awareness, discussion, and engagement among patient, family, and clinician on the evidence, risks and benefits, options, and decisions in play.
Currently, patients often are insufficiently involved in their care decisions. Even when they are encouraged to play a role in decisions about their care, they often lack understandable, reliable information—from evidence on the efficacy and risks of different treatment options to information on the quality of different providers and health care organizations—that is customized to their needs, preferences, and health goals. Fewer than half
___________________________________________________
5While the term patients is used in this report for brevity, it always refers to patients, families and other caregivers, and the public. Similarly, the term communities includes all forms of community, such as those defined by geography, culture, disease or condition, occupation, and workplace.
of patients receive clear information on the benefits and trade-offs of treatments for their condition, and fewer than half are satisfied with their level of control in medical decision making (Degner et al., 1997; Fagerlin et al., 2010; IOM, 2011b; Lee et al., 2011, 2012; Sepucha et al., 2010).
To improve patients’ involvement in their care decisions, communication tools need to be developed and customized to patient circumstances. Given the complexity of health care, even highly educated people may have difficulty finding and understanding health information and applying it to their own care or that of their loved ones (IOM, 2004), and those who produce health care information need to consider how that information will be received and used by patients (Maurer et al., 2012). Technology offers opportunities for clinicians to engage patients by meeting with them where they are. These opportunities include improving communications outside of traditional clinical visits by providing new venues for care; assisting patients in managing their own health; and explaining options for shared clinical decisions, a capability that highlights health professionals’ need to assume new roles in partnering with patients in the use of reliable online sources of health information (Brach et al., 2012).
Patient-centered care takes on increasing importance in light of research linking such care to better health outcomes, lower costs, an enhanced care experience, better quality of life, and other benefits. Patient and family involvement in health care decisions has been associated in primary care settings with reduced pain and discomfort, faster recovery in physical health, and improvements in emotional health (Stewart et al., 2000). Well-informed patients also often choose less aggressive and costly therapies. For example, it has been reported that informed patients are up to 20 percent less likely than other patients to choose elective surgery (O’Connor et al., 2009; Stacey et al., 2011). Similarly, patient-centered communication in primary care visits has been correlated with fewer diagnostic tests and referrals (Epstein et al., 2005; Stewart et al., 2000), as well as with annual charges in the range of 33 percent lower (Bertakis and Azari, 2011a,b).
Not all care delivered in the name of patient-centeredness reduces costs or improves outcomes. For example, one study found that patient-centeredness was associated with better outcomes but also higher costs (Bechel et al., 2000). Other studies have yielded mixed results with respect to cost, quality, and value for care models that aim to implement different aspects of patient-centeredness, such as disease management and care coordination programs (Nelson, 2012; Peikes et al., 2009). This may be related in part to the difficulty of identifying what truly constitutes patient-centered care, with well-meaning but poorly informed efforts producing changes that are superficial and adding little value to the experience. In the name of patient-centeredness, for example, some health care organizations have adopted luxury, hotel-like amenities or renovated their facilities. Although
some of these initiatives may appeal to patient tastes, they do not achieve the true goals of patient-centered care and may increase costs while not directly addressing the patient’s needs, preferences, or goals most important to improving quality, health, and value.
This report builds on the definition of patient-centered care offered in Crossing the Quality Chasm: “providing care that is respectful of and responsive to individual patient preferences, needs, and values and ensuring that patient values guide all decisions” (IOM, 2001a). The concept encompasses multiple dimensions, including respect for patients’ values, preferences, and needs; coordination and integration of care; information, communication, and education; physical comfort; emotional support; and involvement of family and friends. This definition provides a framework for care to be fully patient-centered.
Conclusion: Improved patient engagement is associated with better patient experience, health, and quality of life and better economic outcomes, yet patient and family participation in care decisions remains limited.
Given the increasing incidence of chronic diseases, the complexity of modern health care, and the multiple determinants of health, the challenges facing the health care system cannot be met by any individual or organization acting alone. Yet care often is poorly coordinated among clinicians both within and across settings. In one survey, roughly 25 percent of patients noted that a test had to be repeated, often because the results had not been shared by another provider (Stremikis et al., 2011). This inadequate, sometimes absent, continuity of care endangers patients and contributes to system waste. For example, almost one-fifth of Medicare patients are rehospitalized within 30 days, often without seeing their primary care provider in the interim (Jencks et al., 2009). Comprehensive health care also requires accounting for factors typically outside of the traditional health care system. Most determinants of the health status of individuals and populations lie not in health care—medical care accounts for only 10 to 20 percent of overall health prospects—but in such factors as behavior, social circumstances, and environment. Thus, close clinical-community coordination is required to protect and improve health (McGinnis et al., 2002).
Conclusion: Coordination and integration of patient services currently are poor. Improvement in this area will require strong and sustained avenues of communication and cooperation between and among clinical and community stewards of services.
Achieving and Rewarding High-Value Care
Health care payment policies strongly influence how care is delivered, whether new scientific insights and knowledge about best care are diffused broadly, and whether improvement initiatives succeed. Clinicians reimbursed for each service tend to recommend more visits and services than clinicians who are reimbursed under other payment methods. In one study, initiation of encounter- and procedure-based reimbursement for primary care led to an increased number of encounters and procedures, with visits increasing from 11 to 61 percent depending on the specialty (Helmchen and Lo Sasso, 2010). As with most aspects of health care, a variety of financial incentives and payment models currently are in use. However, most of these models tend to pay clinicians and health care organizations without a specific focus on patient health and value, which has contributed to waste and inefficiency. One study found, on average, only a 4.3 percent correlation between the quality of care delivered and the price of the medical service, with higher prices often being associated with lower quality (Office of the Attorney General of Massachusetts, 2011).
Conclusion: The prevailing approach to paying for health care, based predominantly on individual services and products, encourages wasteful and ineffective care.
Given the clear need for change, several health care organizations and health insurers across the nation have been testing new models of paying for care and organizing care delivery. While many individual initiatives have demonstrated success, evidence is conflicting on which payment models might work best and under what circumstances. Yet, it is clear that high-value care—the best care for the patient, with the optimal result for the circumstances, delivered at the right price—requires that payment and practice incentives be structured to reward the best outcomes for the patient.
To transition to a health care payment system that rewards value, assessment techniques are needed to identify and encourage high-value care. In part, this is a clinical effectiveness issue. Unnecessary and marginal treatments and tests have the potential for side effects and harm. But at its core, health care value is a basic representation of the efficient use of individual and societal resources—time, money—for individual and societal benefit. Because measures of value must fundamentally balance the results of care with the costs required to achieve the results, accurate information is needed on the various dimensions of cost, as well as the various dimensions of health—health status, quality of life, quality of care, satisfaction, and population health.
Measurement itself is only part of the improvement process. Transparency on results produces data that clinicians can use for improvement initiatives, provides information that patients and consumers can use to select care and providers, and draws attention to high-value health care providers and organizations. Several transparency initiatives have been correlated both with improving performance on those measures reported and with encouraging organizations to undertake improvement activities. Following public reporting of pneumonia care measures, for example, rates of compliance with the measures rose from 72 percent to 95 percent in 8 years (Joint Commission, 2011). Results from another initiative showed that providing financial incentives together with helping clinicians monitor their practice patterns against those of others decreased spending by 2 percent per quarter while improving the overall quality of care (Chernew et al., 2011; Mechanic et al., 2011; Song et al., 2011). While further work is needed to improve the practical implementation of transparency and minimize negative consequences, greater transparency is necessary to provide the information needed to promote continuous learning and improvement.
Conclusion: Transparency of process, outcome, price, and cost information, both within health care and with patients and the public, has untapped potential to support continuous learning and improvement in patient experience, outcomes, and cost and the delivery of high-value care.
Creating a New Culture of Care
Although financial incentives can be important to the pace at which change occurs, they do not operate in a vacuum. The culture of health care is central to promoting learning at every level. Continuous improvement requires systematic problem solving, the application of systems engineering techniques, operational models that encourage and reward sustained quality and improved patient outcomes, transparency on cost and outcomes, and strong leadership with a vision devoted to improving health care processes. The goal is to create continuously learning organizations that generate and transfer knowledge from every patient interaction to yield greater performance predictability and reliability.
As with many other aspects of the health care enterprise, there is great diversity in the organizations that deliver care, from small group practices, to independent practice associations, to individual hospitals, to large integrated delivery systems. Each brings different strengths and weaknesses, and each plays a significant and important role in delivering high-quality, high-value care. Given the dramatic differences in local health care infrastructures, substantial heterogeneity will persist for the foreseeable future.
Yet, the need for a new culture of care is common to all types of health care organizations; all need to build their capabilities to continuously learn and improve.
Most vital to building a continuously learning organization is leadership and governance that defines, disseminates, and supports a vision of continuous improvement (Cosgrove et al., 2012). One study found that hospitals ranking in the top 5 percent for heart attack outcomes had a strong leadership and governance commitment to improvement, good communication and coordination, shared values and culture, and experience with problem solving and learning (Curry et al., 2011). An organization’s leadership—and that leadership’s visible priorities—sets its tone, defines and communicates its goals, motivates its staff, and marshals the necessary resources. By defining and visibly emphasizing a vision that encourages and rewards learning and improvement, leadership at all levels of the organization prompt its disparate elements to work together toward a common end.
If leadership provides the top-down mission of an organization, the organization’s culture represents the social scaffolding that empowers system transformation. Organizational culture can encourage strong communication and coordination among clinicians, provide psychological safety that encourages open communication, and support innovation and creativity. This culture of care considers the needs and abilities of individual patients and how they can be engaged as members of the care team. Further, an organization’s commitment to teaming, partnership, and continuity is fundamental in fostering a culture of continuous learning and improvement. In a large, multifacility integrated health system, for example, an intervention that focused on teamwork training, coaching, and communication skills saw an 18 percent reduction in annual mortality among participating facilities, with adverse events continuing to decrease, versus only a 7 percent reduction among nonparticipating facilities (Neily et al., 2010, 2011).
Continuous learning requires dedicated learning processes—mechanisms that help the organization constantly capture knowledge and implement improvements. Achieving systems-based problem solving requires an organizational culture that incentivizes experimentation among staff—one that recognizes failure as key to the learning process and does not penalize employees if their experiments are unsuccessful. These processes can take many forms, yet they share certain essential elements: systematic problem solving and experimentation, learning from past experience and from others, and the use of internal transparency as a tool to motivate further improvement. Beyond systems-based problem solving, systems that continuously learn and improve also need to be adept at transferring the knowledge they gain throughout the organization. While each of these factors is important, it is the organization’s operational model—the way it aligns goals, resources, and incentives—that makes learning actionable. An
organization’s operational model can incentivize continuous learning, help control variability and waste that do not contribute to quality care, recoup savings to invest in improving care processes and patient health, and make improvement sustainable.
Conclusion: Realizing the potential of a continuously learning health care system will require a sustained commitment to improvement, optimized operations, concomitant culture change, aligned incentives, and strong leadership within and across organizations.
ACTIONS FOR CONTINUOUS LEARNING, BEST CARE, AND LOWER COSTS
Based on the findings and conclusions derived in the course of its work, the committee offers recommendations for specific actions that would accelerate progress toward continuous learning, best care, and lower costs. As displayed in Box S-1, these recommendations can be grouped into three categories: foundational elements, care improvement targets, and a supportive policy environment.
Following are the committee’s recommendations, which are supported by the material presented in the full report; also identified are the stakeholders whose engagement is necessary for the implementation of each recommendation. Each recommendation describes the core improvement aim for the area, followed by specific strategies representing initial steps that stakeholders should take in acting on the recommendation. Additional activities will have to be undertaken by numerous stakeholder groups to sustain and advance the continuous improvement required.
Foundational Elements
Recommendation 1: The Digital Infrastructure
Improve the capacity to capture clinical, care delivery process, and financial data for better care, system improvement, and the generation of new knowledge. Data generated in the course of care delivery should be digitally collected, compiled, and protected as a reliable and accessible resource for care management, process improvement, public health, and the generation of new knowledge.
Strategies for progress toward this goal:
- Health care delivery organizations and clinicians should fully and effectively employ digital systems that capture patient care
- experiences reliably and consistently, and implement standards and practices that advance the interoperability of data systems.
BOX S-1
Categories of the Committee’s Recommendations
Foundational Elements
Recommendation 1: The digital infrastructure. Improve the capacity to capture clinical, care delivery process, and financial data for better care, system improvement, and the generation of new knowledge.
Recommendation 2: The data utility. Streamline and revise research regulations to improve care, promote the capture of clinical data, and generate knowledge.
Care Improvement Targets
Recommendation 3: Clinical decision support. Accelerate integration of the best clinical knowledge into care decisions.
Recommendation 4: Patient-centered care. Involve patients and families in decisions regarding health and health care, tailored to fit their preferences.
Recommendation 5: Community links. Promote community-clinical partnerships and services aimed at managing and improving health at the community level.
Recommendation 6: Care continuity. Improve coordination and communication within and across organizations.
Recommendation 7: Optimized operations. Continuously improve health care operations to reduce waste, streamline care delivery, and focus on activities that improve patient health.
Supportive Policy Environment
Recommendation 8: Financial incentives. Structure payment to reward continuous learning and improvement in the provision of best care at lower cost.
Recommendation 9: Performance transparency. Increase transparency on health care system performance.
Recommendation 10: Broad leadership. Expand commitment to the goals of a continuously learning health care system.
- The National Coordinator for Health Information Technology, digital technology developers, and standards organizations should ensure that the digital infrastructure captures and delivers the core data elements and interoperability needed to support better care, system improvement, and the generation of new knowledge.
- Payers, health care delivery organizations, and medical product companies should contribute data to research and analytic consortia to support expanded use of care data to generate new insights.
- Patients should participate in the development of a robust data utility; use new clinical communication tools, such as personal portals, for self-management and care activities; and be involved in building new knowledge, such as through patient-reported outcomes and other knowledge processes.
- The Secretary of Health and Human Services should encourage the development of distributed data research networks and expand the availability of departmental health data resources for translation into accessible knowledge that can be used for improving care, lowering costs, and enhancing public health.
- Research funding agencies and organizations, such as the National Institutes of Health, the Agency for Healthcare Research and Quality, the Veterans Health Administration, the Department of Defense, and the Patient-Centered Outcomes Research Institute, should promote research designs and methods that draw naturally on existing care processes and that also support ongoing quality improvement efforts.
Recommendation 2: The Data Utility
Streamline and revise research regulations to improve care, promote the capture of clinical data, and generate knowledge. Regulatory agencies should clarify and improve regulations governing the collection and use of clinical data to ensure patient privacy but also the seamless use of clinical data for better care coordination and management, improved care, and knowledge enhancement.
Strategies for progress toward this goal:
- The Secretary of Health and Human Services should accelerate and expand the review of the Health Insurance Portability and Accountability Act (HIPAA) and institutional review board (IRB) policies with respect to actual or perceived regulatory impediments to the protected use of clinical data, and clarify regulations and their interpretation to support the use of clinical data as a resource for advancing science and care improvement.
- Patient and consumer groups, clinicians, professional specialty societies, health care delivery organizations, voluntary organizations, researchers, and grantmakers should develop strategies and outreach to improve understanding of the benefits and importance of accelerating the use of clinical data to improve care and health outcomes.
Care Improvement Targets
Recommendation 3: Clinical Decision Support
Accelerate integration of the best clinical knowledge into care decisions. Decision support tools and knowledge management systems should be routine features of health care delivery to ensure that decisions made by clinicians and patients are informed by current best evidence.
Strategies for progress toward this goal:
- Clinicians and health care organizations should adopt tools that deliver reliable, current clinical knowledge to the point of care, and organizations should adopt incentives that encourage the use of these tools.
- Research organizations, advocacy organizations, professional specialty societies, and care delivery organizations should facilitate the development, accessibility, and use of evidence-based and harmonized clinical practice guidelines.
- Public and private payers should promote the adoption of decision support tools, knowledge management systems, and evidence-based clinical practice guidelines by structuring payment and contracting policies to reward effective, evidence-based care that improves patient health.
- Health professional education programs should teach new methods for accessing, managing, and applying evidence; engaging in lifelong learning; understanding human behavior and social science; and delivering safe care in an interdisciplinary environment.
- Research funding agencies and organizations should promote research into the barriers and systematic challenges to the dissemination and use of evidence at the point of care, and support research to develop strategies and methods that can improve the usefulness and accessibility of patient outcome data and scientific evidence for clinicians and patients.
Recommendation 4: Patient-Centered Care
Involve patients and families in decisions regarding health and health care, tailored to fit their preferences. Patients and families should be given the opportunity to be fully engaged participants at all levels, including individual care decisions, health system learning and improvement activities, and community-based interventions to promote health.
Strategies for progress toward this goal:
- Patients and families should expect to be offered full participation in their own care and health and encouraged to partner, according to their preference, with clinicians in fulfilling those expectations.
- Clinicians should employ high-quality, reliable tools and skills for informed shared decision making with patients and families, tailored to clinical needs, patient goals, social circumstances, and the degree of control patients prefer.
- Health care delivery organizations, including programs operated by the Department of Defense, the Veterans Health Administration, and the Health Resources and Services Administration, should monitor and assess patient perspectives and use the insights thus gained to improve care processes; establish patient portals to facilitate data sharing and communication among clinicians, patients, and families; and make high-quality, reliable tools available for shared decision making with patients at different levels of health literacy.
- The Agency for Healthcare Research and Quality, partnering with the Centers for Medicare & Medicaid Services, other payers, and stakeholder organizations, should support the development and testing of an accurate and reliable core set of measures of patient-centeredness for consistent use across the health care system.
- The Centers for Medicare & Medicaid Services and other public and private payers should promote and measure patient-centered care through payment models, contracting policies, and public reporting programs.
- Digital technology developers and health product innovators should develop tools to assist individuals in managing their health and health care, in addition to providing patient supports in new forms of communities.
Recommendation 5: Community Links
Promote community-clinical partnerships and services aimed at managing and improving health at the community level. Care delivery and community-based organizations and agencies should partner with each other to develop cooperative strategies for the design, implementation, and accountability of services aimed at improving individual and population health.
Strategies for progress toward this goal:
- Health care delivery organizations and clinicians should partner with community-based organizations and public health agencies to leverage and coordinate prevention, health promotion, and community-based interventions to improve health outcomes, including strategies related to the assessment and use of Web-based tools.
- Public and private payers should incorporate population health improvement into their health care payment and contracting policies and accountability measures.
- Health economists, health service researchers, professional specialty societies, and measure development organizations should continue to improve measures that can readily be applied to assess performance on both individual and population health.
Recommendation 6: Care Continuity
Improve coordination and communication within and across organizations. Payers should structure payment and contracting to reward effective communication and coordination between and among members of a patient’s care team.
Strategies for progress toward this goal:
- Health care delivery organizations and clinicians, partnering with patients, families, and community organizations, should develop coordination and transition processes, data sharing capabilities, and communication tools to ensure safe, seamless patient care.
- Health economists, health service researchers, professional specialty societies, and measure development organizations should develop and test metrics with which to monitor and evaluate the effectiveness of care transitions in improving patient health outcomes.
- Public and private payers should promote effective care transitions that improve patient health through their payment and contracting policies.
Recommendation 7: Optimized Operations
Continuously improve health care operations to reduce waste, streamline care delivery, and focus on activities that improve patient health. Care delivery organizations should apply systems engineering tools and process improvement methods to improve operations and care delivery processes.
Strategies for progress toward this goal:
- Health care delivery organizations should utilize systems engineering tools and process improvement methods to eliminate inefficiencies, remove unnecessary burdens on clinicians and staff, enhance patient experience, and improve patient health outcomes.
- The Centers for Medicare & Medicaid Services, the Agency for Healthcare Research and Quality, the Patient-Centered Outcomes Research Institute, quality improvement organizations, and process improvement leaders should develop a learning consortium aimed at accelerating training, technical assistance, and the collection and validation of lessons learned about ways to transform the effectiveness and efficiency of care through continuous improvement programs and initiatives.
Supportive Policy Environment
Recommendation 8: Financial Incentives
Structure payment to reward continuous learning and improvement in the provision of best care at lower cost. Payers should structure payment models, contracting policies, and benefit designs to reward care that is effective and efficient and continuously learns and improves.
Strategies for progress toward this goal:
- Public and private payers should reward continuous learning and improvement through outcome- and value-oriented payment models, contracting policies, and benefit designs. Payment models should adequately incentivize and support high-quality team-based care focused on the needs and goals of patients and families.
- Health care delivery organizations should reward continuous learning and improvement through the use of internal practice incentives.
- Health economists, health service researchers, professional specialty societies, and measure development organizations should partner with public and private payers to develop and evaluate metrics, payment models, contracting policies, and benefit designs that reward high-value care that improves health outcomes.
Recommendation 9: Performance Transparency
Increase transparency on health care system performance. Health care delivery organizations, clinicians, and payers should increase the availability of information on the quality, prices and cost, and outcomes of care to help inform care decisions and guide improvement efforts.
Strategies for progress toward this goal:
- Health care delivery organizations should collect and expand the availability of information on the safety, quality, prices and cost, and health outcomes of care.
- Professional specialty societies should encourage transparency on the quality, value, and outcomes of the care provided by their members.
- Public and private payers should promote transparency in quality, value, and outcomes to aid plan members in their care decision making.
- Consumer and patient organizations should disseminate this information to facilitate discussion, informed decision making, and care improvement.
Recommendation 10: Broad Leadership
Expand commitment to the goals of a continuously learning health care system. Continuous learning and improvement should be a core and constant priority for all participants in health care—patients, families, clinicians, care leaders, and those involved in supporting their work.
Strategies for progress toward this goal:
- Health care delivery organizations should develop organizational cultures that support and encourage continuous
- improvement, the use of best practices, transparency, open communication, staff empowerment, coordination, teamwork, and mutual respect and align rewards accordingly.
- Leaders of these organizations should define, disseminate, support, and commit to a vision of continuous improvement; focus attention, training, and resources on continuous learning; and build an operational model that incentivizes continuous improvement and ensures its sustainability.
- Governing boards of health care delivery organizations should support and actively participate in fostering a culture of continuous improvement, request continuous feedback on the progress being made toward the adoption of such a culture, and align leadership incentive structures accordingly.
- Clinical professional specialty societies, health professional education programs, health professions specialty boards, licensing boards, and accreditation organizations should incorporate basic concepts and specialized applications of continuous learning and improvement into health professions education; continuing education; and licensing, certification, and accreditation requirements.
Given the interconnected nature of the problems to be solved, it will be important to take the actions identified above in concert. To elevate the quantity of evidence available to inform clinical decisions, for example, it is necessary to increase the supply of evidence by expanding the clinical research base; make the evidence easily accessible by embedding it in clinical technological tools, such as clinical decision support; encourage use of the evidence through appropriate payment, contracting, and regulatory policies and cultural factors; and assess progress toward the goal using reliable metrics and appropriate transparency. The absence of any one of these factors will substantially limit overall improvement. To guide success, progress on the recommendations in this report should be monitored continuously.
Implementing the actions detailed above and achieving the vision of continuous learning and improvement will depend on the exercise of broad leadership by the complex network of decentralized and loosely associated individuals and organizations that make up the health care system. Given the complexity of the system and the interconnectedness of its different actors and sectors, no one actor or sector alone can bring about the scope and scale of transformative change necessary to develop a system that continuously learns and improves. Each stakeholder brings different strengths, skills, needs, and expertise to the task of improving the system, faces unique challenges, and is accountable for different aspects of the system’s success. There is a distinct need for collaboration between and among stakeholders to produce effective and sustainable change.
As the end users of all health care services, patients are central to the success of any improvement initiative. Any large-scale change will require the participation of patients as partners, with the system building trust on every dimension. Patients can promote learning and improvement by engaging in their own care; setting high expectations for their care in terms of quality, value, and the use of scientific evidence and selecting clinicians, organizations, and plans that meet those expectations; sharing decision making
with their clinicians; and, with the help of their caregivers, directly applying evidence to their self-care and self-management on an ongoing basis.
Partnering with patients are the health care professionals who deliver care. Physicians, nurses, pharmacists, and other health professionals represent the front lines of health care delivery and the primary interface for patients and consumers. Expanding the supply of clinical information, promoting the use of evidence, and better involving patients in their care are all contingent upon the engagement and teaming of health professionals.
By convening their constituent professionals and providing a forum for action, professional societies have important roles in achieving the vision of a learning health care system. Through guidelines, performance measures, quality improvement initiatives, and data infrastructures for assessing performance with respect to specific procedures or conditions, these societies can take a leadership role in improving quality, safety, and efficiency.
Health care delivery organizations, because of their size and care capacities, have several levers by which they can steward progress toward a continuously improving system, such as using new practice methods, setting standards, and sharing resources and information with other care delivery organizations. Furthermore, through investments in health information technology, these organizations can build their capacity to perform near-real-time research, speeding the generation of practical evidence and its translation to the bedside.
Those who finance care also have opportunities to leverage their unique position to improve the quality and efficiency of care. As organizations that interact directly with patients, public and private payers can support patients as they seek to maintain healthy behaviors and access quality health care services, while their payment and contracting policies have a strong influence on how clinicians practice. Similarly, employers can support efforts to improve quality and value by using their purchasing power to drive improvement efforts through contracts with providers and insurers, the design of benefit plans, and the provision of incentives and information for employees.
Digital technology developers, health product innovators, and regulators are additional stakeholders that need to be engaged in achieving the vision of a learning health care system. Digital technology developers create the products and infrastructure necessary to meet the growing demand for capturing, storing, retrieving, and sharing information in virtually every aspect of health care. Continuous improvement in diagnostic and treatment options is contingent on a safe and innovative product development enterprise. Health product innovators, by conducting clinical research and devising new treatments and interventions, can develop novel products for diagnosis and treatment. Essential partners in this arena are regulators, including the Food and Drug Administration, who can work to develop
streamlined methods for ensuring that safe, effective products are brought to market without delay.
A learning health care system depends on evidence to promote improvements in care delivery processes and patient care and overall system improvement. Consequently, health researchers are critical partners in generating knowledge on the effectiveness and value of interventions and care protocols. A commitment to practical and efficient research methods across the spectrum of the research enterprise—the design and operation of clinical trials, the development of clinical registries and clinical databases, the creation of standards and metrics, modeling and simulation studies, studies of health services and care delivery processes, and the aggregation of study results into systematic reviews and clinical guidelines—is foundational for a learning system. Through their programmatic and funding activities, private philanthropies, as well as agencies and organizations such as the Agency for Healthcare Research and Quality, the National Institutes of Health, and the Patient-Centered Outcomes Research Institute have a central role to play in the stewardship and strategic direction of these activities.
Missed opportunities for better health care have real human and economic impacts. If the care in every state was at the quality delivered by the highest performing state, there would have been an estimated 75,000 fewer deaths across the country in 2005 (McCarthy et al., 2009; Schoenbaum et al., 2011). Current waste diverts resources from productive use—an estimated $750 billion lost (IOM, 2010). It is only through shared commitments, in alignment with a supportive policy environment, that the opportunities offered by science and information technology can be captured to address the health care system’s growing challenges and to ensure that it reaches its full potential to provide the best care for each patient. The nation’s health and economic futures—best care at lower cost—depend on the ability to steward the evolution of a continuously learning health care system.
Anderson, G. F. 2010. Chronic care: Making the case for ongoing care. Princeton, NJ: Robert Wood Johnson Foundation.
Antman, E. M., J. Lau, B. Kupelnick, F. Mosteller, and T. C. Chalmers. 1992. A comparison of results of meta-analyses of randomized control trials and recommendations of clinical experts. Treatments for myocardial infarction. Journal of the American Medical Association 268(2):240-248.
Arora, V., S. Gangireddy, A. Mehrotra, R. Ginde, M. Tormey, and D. Meltzer. 2009. Ability of hospitalized patients to identify their in-hospital physicians. Archives of Internal Medicine 169(2):199-201.
Auerbach, D. I., and A. L. Kellermann. 2011. A decade of health care cost growth has wiped out real income gains for an average US family. Health Affairs 30(9):1630-1636.
Bates, D. W., L. L. Leape, D. J. Cullen, N. Laird, L. A. Petersen, J. M. Teich, E. Burdick, M. Hickey, S. Kleefield, B. Shea, M. Vander Vliet, and D. L. Seger. 1998. Effect of computerized physician order entry and a team intervention on prevention of serious medication errors. Journal of the American Medical Association 280(15):1311-1316.
Bates, D. W., J. M. Teich, J. Lee, D. Seger, G. J. Kuperman, N. Ma’Luf, D. Boyle, and L. Leape. 1999. The impact of computerized physician order entry on medication error prevention. Journal of the American Medical Informatics Association 6(4):313-321.
Bechel, D. L., W. A. Myers, and D. G. Smith. 2000. Does patient-centered care pay off? Joint Commission Journal on Quality Improvement 26(7):400-409.
Bertakis, K. D., and R. Azari. 2011a. Determinants and outcomes of patient-centered care. Patient Education and Counseling 85(1):46-52.
Bertakis, K. D., and R. Azari. 2011b. Patient-centered care is associated with decreased health care utilization. Journal of the American Board of Family Medicine 24(3):229-239.
Boyd, C. M., J. Darer, C. Boult, L. P. Fried, L. Boult, and A. W. Wu. 2005a. Clinical practice guidelines and quality of care for older patients with multiple comorbid diseases. Journal of the American Medical Association 294(6):716.
Boyd, C. M., J. Darer, C. Boult, L. P. Fried, L. Boult, and A. W. Wu. 2005b. Clinical practice guidelines and quality of care for older patients with multiple comorbid diseases: Implications for pay for performance. Journal of the American Medical Association 294(6):716-724.
Brach, C., B. Dreyer, P. Schyve, L. M. Hernandez, C. Baur, A. J. Lemerise, and R. Parker. 2012. Attributes of a health literate organization. Discussion Paper, Institute of Medicine, Washington, DC. http://www.iom.edu/Global/Perspectives/2012/Attributes.aspx (accessed May 27, 2012).
Brownstein, J. S., S. N. Murphy, A. B. Goldfine, R. W. Grant, M. Sordo, V. Gainer, J. A. Colecchi, A. Dubey, D. M. Nathan, J. P. Glaser, and I. S. Kohane. 2010. Rapid identification of myocardial infarction risk associated with diabetes medications using electronic medical records. Diabetes Care 33(3):526-531.
Burdi, M. D., and L. C. Baker. 1999. Physicians’ perceptions of autonomy and satisfaction in California. Health Affairs 18(4):134.
Bureau of Transportation Statistics. 2011. National transportation statistics. Washington, DC: Research and Innovation Technology Administration, U.S. Department of Transportation.
Casalino, L. P., D. Dunham, M. H. Chin, R. Bielang, E. O. Kistner, T. G. Karrison, M. K. Ong, U. Sarkar, M. A. McLaughlin, and D. O. Meltzer. 2009. Frequency of failure to inform patients of clinically significant outpatient test results. Archives of Internal Medicine 169(12):1123-1129.
Cebul, R. D., T. E. Love, A. K. Jain, and C. J. Hebert. 2011. Electronic health records and quality of diabetes care. New England Journal of Medicine 365(9):825-833.
Certo, C. M. 1985. History of cardiac rehabilitation. Physical Therapy 65(12):1793-1795.
Chauhan, S. P., V. Berghella, M. Sanderson, E. F. Magann, and J. C. Morrison. 2006. American College of Obstetricians and Gynecologists practice bulletins: An overview. American Journal of Obstetrics and Gynecology 194(6):1564-1572.
Chernew, M. E., R. E. Mechanic, B. E. Landon, and D. G. Safran. 2011. Private-payer innovation in Massachusetts: The “alternative quality contract.” Health Affairs (Millwood) 30(1):51-61.
Classen, D. C., R. Resar, F. Griffin, F. Federico, T. Frankel, N. Kimmel, J. C. Whittington, A. Frankel, A. Seger, and B. C. James. 2011. “Global trigger tool” shows that adverse events in hospitals may be ten times greater than previously measured. Health Affairs (Millwood) 30(4):581-589.
CMS (Centers for Medicare & Medicaid Services). 2012. National health expenditures summary and GDP: Calendar years 1960-2010. http://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/downloads/tables.pdf (accessed August 31, 2012).
Congressional Budget Office. 2008. Issues and options in infrastructure investment. Washington, DC: Congressional Budget Office.
Congressional Budget Office. 2009. Health Information Technology for Economic and Clinical Health Act. Washington, DC: Congressional Budget Office.
Cosgrove, D., M. Fisher, P. Gabow, G. Gottlieb, G. C. Halvorson, B. James, G. Kaplan, J. Perlin, R. Petzel, G. Steele, and J. Toussaint. 2012. A CEO checklist for high-value health care. Discussion Paper, Institute of Medicine, Washington, DC. http://www.iom.edu/ CEOChecklist (accessed August 31, 2012).
Curry, L. A., E. Spatz, E. Cherlin, J. W. Thompson, D. Berg, H. H. Ting, C. Decker, H. M. Krumholz, and E. H. Bradley. 2011. What distinguishes top-performing hospitals in acute myocardial infarction mortality rates? A qualitative study. Annals of Internal Medicine 154(6):384-390.
Degner, L. F., L. J. Kristjanson, D. Bowman, J. A. Sloan, K. C. Carriere, J. O’Neil, B. Bilodeau, P. Watson, and B. Mueller. 1997. Information needs and decisional preferences in women with breast cancer. Journal of the American Medical Association 277(18):1485-1492.
DeNavas-Walt, C., B. D. Proctor, and J. C. Smith. 2011. Income, poverty, and health insurance coverage in the United States: 2010. Washington, DC: U.S. Census Bureau.
Donchin, Y., D. Gopher, M. Olin, Y. Badihi, M. Biesky, C. L. Sprung, R. Pizov, and S. Cotev. 2003. A look into the nature and causes of human errors in the intensive care unit. Quality & Safety in Health Care 12(2):143-147.
Epstein, R. M., P. Franks, C. G. Shields, S. C. Meldrum, K. N. Miller, T. L. Campbell, and K. Fiscella. 2005. Patient-centered communication and diagnostic testing. Annals of Family Medicine 3(5):415-421.
Fagerlin, A., K. R. Sepucha, M. P. Couper, C. A. Levin, E. Singer, and B. J. Zikmund-Fisher. 2010. Patients’ knowledge about 9 common health conditions: The decisions survey. Medical Decision Making 30(Suppl. 5):S35-S52.
Farrell, D., E. Jensen, B. Kocher, N. Lovegrove, F. Melhem, L. Mendonca, and B. Parish. 2008. Accounting for the cost of US health care: A new look at why Americans spend more. Washington, DC: McKinsey Global Institute.
Harrison, T. R. 1962. Principles of internal medicine. 4th ed. New York: Blakiston Division, McGraw-Hill.
Helmchen, L. A., and A. T. Lo Sasso. 2010. How sensitive is physician performance to alternative compensation schedules? Evidence from a large network of primary care clinics. Health Economics 19(11):1300-1317.
Hendrich, A., M. P. Chow, B. A. Skierczynski, and Z. Lu. 2008. A 36-hospital time and motion study: How do medical-surgical nurses spend their time? The Permanente Journal 12(3):25-34.
Hendrickson, G., T. M. Doddato, and C. T. Kovner. 1990. How do nurses use their time? Journal of Nursing Administration 20(3):31-37.
Hilbert, M., and P. López. 2011. The world’s technological capacity to store, communicate, and compute information. Science 332(6025):60-65.
Holve, E., and P. Pittman. 2009. A first look at the volume and cost of comparative effectiveness research in the United States. Washington, DC: AcademyHealth.
Holve, E., and P. Pittman. 2011. The cost and volume of comparative effectiveness research. In Learning what works: Infrastructure required for comparative effectiveness research: Workshop summary. Institute of Medicine. Washington, DC: The National Academies Press. Pp. 89-96.
Howden, L. M., and J. A. Meyer. 2011. Age and sex composition: 2010. Washington, DC: U.S. Census Bureau, U.S. Department of Commerce.
Hsiao, C.-J., E. Hing, T. C. Socey, and B. Cai. 2011. Electronic health record systems and intent to apply for meaningful use incentives among office-based physician practices: United States, 2001-2011. Hyattsville, MD: National Center for Health Statistics.
IOM (Institute of Medicine). 1999. To err is human: Building a safer health system. Washington, DC: National Academy Press.
IOM. 2001a. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
IOM. 2001b. Mammography and beyond: Developing technologies for the early detection of breast cancer. Washington, DC: National Academy Press.
IOM. 2003. Unequal treatment: Confronting racial and ethnic disparities in health care. Washington, DC: The National Academies Press.
IOM. 2004. Health literacy: A prescription to end confusion. Washington, DC: The National Academies Press.
IOM. 2008. Knowing what works in health care: A roadmap for the nation. Washington, DC: The National Academies Press.
IOM. 2009. Beyond the HIPAA privacy rule: Enhancing privacy, improving health through research. Washington, DC: The National Academies Press.
IOM. 2010. The healthcare imperative: Lowering costs and improving outcomes: Workshop series summary, Learning health system series. Washington, DC: The National Academies Press.
IOM. 2011a. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
IOM. 2011b. Patients charting the course: Citizen engagement in the learning health system: Workshop summary. Washington, DC: The National Academies Press.
Jencks, S. F., M. V. Williams, and E. A. Coleman. 2009. Rehospitalizations among patients in the Medicare fee-for-service program. New England Journal of Medicine 360(14):1418-1428.
Joint Commission. 2011. Improving America’s hospitals: The Joint Commission’s annual report on quality and safety. http://www.jointcommission.org/assets/1/6/TJC_Annual_Report_2011_9_13_11_.pdf (accessed September 25, 2011).
Kaiser Family Foundation and Health Research & Educational Trust. 2009. Employer health benefits: 2009 annual survey. Menlo Park, CA: Kaiser Family Foundation and Health Research & Educational Trust.
Kasper, D. L., and T. R. Harrison. 2005. Harrison’s principles of internal medicine. 16th ed., 2 vols. New York: McGraw-Hill, Medical Publications Division.
Keehan, S. P., A. M. Sisko, C. J. Truffer, J. A. Poisal, G. A. Cuckler, A. J. Madison, J. M. Lizonitz, and S. D. Smith. 2011. National health spending projections through 2020: Economic recovery and reform drive faster spending growth. Health Affairs (Millwood) 30(8):1594-1605.
Landrigan, C. P., G. J. Parry, C. B. Bones, A. D. Hackbarth, D. A. Goldmann, and P. J. Sharek. 2010. Temporal trends in rates of patient harm resulting from medical care. New England Journal of Medicine 363(22):2124-2134.
Larsson, S., P. Lawyer, G. Garellick, B. Lindahl, and M. Lundström. 2012. Use of 13 disease registries in 5 countries demonstrates the potential to use outcome data to improve health care’s value. Health Affairs (Millwood) 31(1):220-227.
Lee, C. N., R. Dominik, C. A. Levin, M. J. Barry, C. Cosenza, A. M. O’Connor, A. G. Mulley, Jr., and K. R. Sepucha. 2010a. Development of instruments to measure the quality of breast cancer treatment decisions. Health Expectations 13(3):258-272.
Lee, C. N., C. S. Hultman, and K. Sepucha. 2010b. Do patients and providers agree about the most important facts and goals for breast reconstruction decisions? Annals of Plastic Surgery 64(5):563-566.
Lee, C. N., J. Belkora, Y. Chang, B. Moy, A. Partridge, and K. Sepucha. 2011. Are patients making high-quality decisions about breast reconstruction after mastectomy? Plastic and Reconstructive Surgery 127(1):18-26.
Lee, C. N., Y. Chang, N. Adimorah, J. K. Belkora, B. Moy, A. H. Partridge, D. W. Ollila, and K. R. Sepucha. 2012. Decision making about surgery for early-stage breast cancer. Journal of the American College of Surgeons 214(1):1-10.
Levinson, D. R. 2010. Adverse events in hospitals: National incidence among Medicare beneficiaries. Washington, DC: U.S. Department of Health and Human Services, Office of Inspector General.
Linzer, M., L. B. Manwell, E. S. Williams, J. A. Bobula, R. L. Brown, A. B. Varkey, B. Man, J. E. McMurray, A. Maguire, B. Horner-Ibler, M. D. Schwartz, and MEMO (Minimizing Error, Maximizing Outcome) Investigators. 2009. Working conditions in primary care: Physician reactions and care quality. Annals of Internal Medicine 151(1):28-36, W26-W29.
Maurer, M., P. Dardess, K. L. Carman, K. Frazier, and L. Smeeding. 2012. Guide to patient and family engagement: Environmental scan report. Rockville, MD: Agency for Healthcare Research and Quality.
McCarthy, D., S. How, C. Schoen, J. Cantor, and D. Belloff. 2009. Aiming higher: Results from a state scorecard on health system performance. New York: Commonwealth Fund Commission on a High Performance Health System.
McGinnis, J. M., P. Williams-Russo, and J. R. Knickman. 2002. The case for more active policy attention to health promotion. Health Affairs (Millwood) 21(2):78-93.
McGlynn, E. A., S. M. Asch, J. Adams, J. Keesey, J. Hicks, A. DeCristofaro, and E. A. Kerr. 2003. The quality of health care delivered to adults in the United States. New England Journal of Medicine 348(26):2635-2645.
Mechanic, R. E., P. Santos, B. E. Landon, and M. E. Chernew. 2011. Medical group responses to global payment: Early lessons from the “alternative quality contract” in Massachusetts. Health Affairs (Millwood) 30(9):1734-1742.
Nabel, E. G., and E. Braunwald. 2012. A tale of coronary artery disease and myocardial infarction. New England Journal of Medicine 366(1):54-63.
Neily, J., P. D. Mills, Y. Young-Xu, B. T. Carney, P. West, D. H. Berger, L. M. Mazzia, D. E. Paull, and J. P. Bagian. 2010. Association between implementation of a medical team training program and surgical mortality. Journal of the American Medical Association 304(15):1693-1700.
Neily, J., P. D. Mills, N. Eldridge, B. T. Carney, D. Pfeffer, J. R. Turner, Y. Young-Xu, W. Gunnar, and J. P. Bagian. 2011. Incorrect surgical procedures within and outside of the operating room: A follow-up report. Archives of Surgery 146(11):1235-1239.
Nelson, L. 2012. Lessons from Medicare’s demonstration projects on disease management and care coordination. Washington, DC: Congressional Budget Office.
Ness, R. B. 2007. Influence of the HIPAA privacy rule on health research. Journal of the American Medical Association 298(18):2164-2170.
O’Connor, A. M., C. L. Bennett, D. Stacey, M. Barry, N. F. Col, K. B. Eden, V. A. Entwistle, V. Fiset, M. Holmes-Rovner, S. Khangura, H. Llewellyn-Thomas, and D. Rovner. 2009. Decision aids for people facing health treatment or screening decisions. Cochrane Database of Systematic Reviews (3):CD001431.
Office of the Attorney General of Massachusetts. 2011. Examination of health care cost trends and cost drivers pursuant to G.L. c. 118g, ![]() 6½(b). http://www.mass.gov/ago/docs/healthcare/2011-hcctd-full.pdf (accessed August 30, 2012).
6½(b). http://www.mass.gov/ago/docs/healthcare/2011-hcctd-full.pdf (accessed August 30, 2012).
OMB (Office of Management and Budget). 2010. Fiscal year 2011 budget of the U.S. government. Washington, DC: OMB.
Parekh, A. K., and M. B. Barton. 2010. The challenge of multiple comorbidity for the US health care system. Journal of the American Medical Association 303(13):1303-1304.
Peikes, D., A. Chen, J. Schore, and R. Brown. 2009. Effects of care coordination on hospitalization, quality of care, and health care expenditures among Medicare beneficiaries: 15 randomized trials. Journal of the American Medical Association 301(6):603-618.
Pham, H. H., D. Schrag, A. S. O’Malley, B. Wu, and P. B. Bach. 2007. Care patterns in Medicare and their implications for pay for performance. New England Journal of Medicine 356(11):1130-1139.
Pham, H. H., A. S. O’Malley, P. B. Bach, C. Saiontz-Martinez, and D. Schrag. 2009. Primary care physicians’ links to other physicians through Medicare patients: The scope of care coordination. Annals of Internal Medicine 150(4):236-242.
Potts, A. L., F. E. Barr, D. F. Gregory, L. Wright, and N. R. Patel. 2004. Computerized physician order entry and medication errors in a pediatric critical care unit. Pediatrics 113(1 Pt. 1):59-63.
Pronovost, P., D. Needham, S. Berenholtz, D. Sinopoli, H. T. Chu, S. Cosgrove, B. Sexton, R. Hyzy, R. Welsh, G. Roth, J. Bander, J. Kepros, and C. Goeschel. 2006. An intervention to decrease catheter-related bloodstream infections in the ICU. New England Journal of Medicine 355(26):2725-2732.
Pronovost, P. J., C. A. Goeschel, K. L. Olsen, J. C. Pham, M. R. Miller, S. M. Berenholtz, J. B. Sexton, J. A. Marsteller, L. L. Morlock, A. W. Wu, J. M. Loeb, and C. M. Clancy. 2009. Reducing health care hazards: Lessons from the commercial aviation safety team. Health Affairs (Millwood) 28(3):w479-w489.
Schneider, K. M., B. E. O’Donnell, and D. Dean. 2009. Prevalence of multiple chronic conditions in the United States’ Medicare population. Health and Quality of Life Outcomes 7:82.
Schoenbaum, S. C., C. Schoen, J. L. Nicholson, and J. C. Cantor. 2011. Mortality amenable to health care in the United States: The roles of demographics and health systems performance. Journal of Public Health Policy 32(4):407-429.
Sepucha, K. R., A. Fagerlin, M. P. Couper, C. A. Levin, E. Singer, and B. J. Zikmund-Fisher. 2010. How does feeling informed relate to being informed? The decisions survey. Medical Decision Making 30(Suppl. 5):S77-S84.
Song, Z., D. G. Safran, B. E. Landon, Y. He, R. P. Ellis, R. E. Mechanic, M. P. Day, and M. E. Chernew. 2011. Health care spending and quality in year 1 of the alternative quality contract. New England Journal of Medicine 65(10):909-918.
Spear, S. J. 2005. Fixing health care from the inside, today. Harvard Business Review 83(9):78.
Stacey, D., C. L. Bennett, M. J. Barry, N. F. Col, K. B. Eden, M. Holmes-Rovner, H. Llewellyn-Thomas, A. Lyddiatt, F. Legare, and R. Thomson. 2011. Decision aids for people facing health treatment or screening decisions. Cochrane Database of Systematic Reviews (10):CD001431.
Stewart, M., J. B. Brown, A. Donner, I. R. McWhinney, J. Oates, W. W. Weston, and J. Jordan. 2000. The impact of patient-centered care on outcomes. Journal of Family Practice 49(9):796-804.
Stremikis, K., C. Schoen, and A.-K. Fryer. 2011. A call for change: The 2011 Commonwealth Fund survey of public views of the U.S. health system. New York: Commonwealth Fund.
Tinetti, M. E., S. T. Bogardus, and J. V. Agostini. 2004. Potential pitfalls of disease-specific guidelines for patients with multiple conditions. New England Journal of Medicine 351(27):2870-2874.
Tricoci, P., J. M. Allen, J. M. Kramer, R. M. Califf, and S. C. Smith, Jr. 2009. Scientific evidence underlying the ACC/AHA clinical practice guidelines. Journal of the American Medical Association 301(8):831-841.
Trude, S. 2003. So much to do, so little time: Physician capacity constraints, 1997-2001. Tracking report/Center for Studying Health System Change (8):1.
Tucker, A. L., and S. J. Spear. 2006. Operational failures and interruptions in hospital nursing. Health Services Research 41(3 Pt. 1):643-662.
U.S. Bureau of Labor Statistics. 2010a. May 2009 national occupational employment and wage estimates. http://www.bls.gov/oes/2009/may/oes_nat.htm (accessed May 22, 2012).
U.S. Bureau of Labor Statistics. 2010b. National compensation survey: Occupational earnings in the United States, 2009. http://www.bls.gov/ncs/ocs/sp/nctb1346.pdf (accessed May 22, 2012).
U.S. Bureau of Labor Statistics. 2012. Labor force statistics from the current population survey. http://data.bls.gov/pdq/SurveyOutputServlet?request_action=wh&graph_name=LN_cpsbref1 (accessed May 23, 2012).
Wennberg, J. E., E. S. Fisher, and J. S. Skinner. 2002. Geography and the debate over Medicare reform. Health Affairs (Millwood) (Suppl. Web Exclusives):W96-W114.
Yarnall, K. S. H., K. Krause, K. Pollak, M. Gradison, and J. Michener. 2009. Family physicians as team leaders: “Time” to share the care. Preventing Chronic Disease 6(2).








































