3
Using Insights About Learning to Inform Teaching
“I think it’s about putting yourself in the students’ shoes and seeing how a first-time student, maybe someone who hasn’t even taken chemistry before, is looking at it.”
—Valerie Taraborelli, undergraduate chemistry student, University of Arizona1
“In some ways, I think the people who are the most successful as teachers are the ones who are able to remember what it was like being uncertain and not knowing. When you become an expert, things are easy. So the idea is to try and see where [students] are coming from and why they’ve developed this misconception and what you can do to specifically address it.”
—Dee Silverthorn, biology professor, University of Texas2
These two quotations underscore a point made by research—that effective science and engineering instruction involves much more than conveying to students what you, as an expert, already know and what you think they should know. Rather, effective teaching in these disciplines involves ascertaining what students know, what they don’t know, and what they think they know but do not really understand accurately or fully. Using that information, you can help students establish a solid framework of understanding that can better support new knowledge.
While teaching and learning are often seen as distinct processes—one controlled by the teacher and the other by the student—they are really intertwined aspects of a complex process shaped by both teacher and student. Understanding
________________
1 Interview, April 24, 2013.
2 Interview, June 25, 2013.

how students learn is a key aspect of good teaching.
This chapter describes some of the main insights from research on how people learn in general and how undergraduate students learn science and engineering in particular. Rather than being a comprehensive review of evidence from discipline-based education research (DBER), the findings and examples highlighted in this chapter are intended to illustrate how an understanding of learning can lead you to think differently about instruction and design more effective approaches. Additional information from research on how undergraduate students learn science and engineering is available in the 2012 National Research Council (NRC) report on DBER.
General Insights About How Students Learn
Karl Wirth, a geosciences professor at Macalester College, is one of many instructors who have been strongly influenced by research on how people learn.
In designing new instructional strategies, Wirth and others have drawn on four decades of findings from cognitive sciences, neurosciences, and related fields. This body of scholarship comprises “an extraordinary outpouring of scientific work on the mind and brain, on the processes of thinking and learning, on the neural processes that occur during thought and learning, and on the development of competence” (National Research Council, 2000, p. 3).
Helping Students Become Intentional Learners
In the 1990s, Macalester College professor Karl Wirtha realized that although he thought he had been teaching his geosciences students effectively, “when they did senior capstone projects, they really weren’t very well prepared. That came as sort of a shock to me.” This realization led him to seek out ways to help his students develop deeper understanding and critical-thinking skills and become more strategic, self-motivated learners. He attended workshops and read the research literature on learning, including How People Learn: Brain, Mind, Experience, and School, a seminal study by the National Research Council (2000).
Drawing on this body of research, Wirth incorporated activities into all of his courses to develop students’ skills in metacognition, or thinking about their own thinking and learning. The first reading assignment in his courses is a paper called Learning to Learn, prepared by Wirth and University of North Dakota professor Dexter Perkins (2008), which highlights research on learning and metacognition and signals his expectation that students will learn in ways that go far beyond memorizing content.
Each time Wirth’s students complete an out-of-class reading assignment, for example, they do a short writing assignment, or “reading reflection,” in which they answer questions like these: What is the main point of this reading? What information did you find surprising and why? What did you find confusing and why? Similarly, after completing an in-class activity, students write a brief “learning reflection,” in which they respond to questions like these: What are the three most important things you have learned? Why? Describe the learning strategies that you are using. How might they be adapted for more effective learning? How does learning in this course relate to other courses?
These reflection exercises prompt students to monitor their own understanding and progress and motivate them to come to class better prepared, says Wirth. In addition, the exercises provide the instructor with frequent feedback about students’ learning that can be used to guide improvements in instruction, a function known as formative assessment. “They give me a sense of where the sticky points are in their content learning,” he says. “More than anything, it puts me in touch with what’s going on in their brains.” Often Wirth shows students graphs of the positive correlation between completion of the reading reflection assignments and their course grades. “When they see that, they begin to realize that the people who are doing well are doing all the reading reflections.” The reflection process “not only leads to deeper and more effective learning, but also lays the groundwork for being a self-directing learner” (Wirth and Perkins, 2008).
Spurred by research on the value of active student engagement and student collaboration, Wirth has adopted a so-called studio format in his classes that makes no distinction between lecture and lab. Students spend the better part of each two-hour class period conducting hands-on lab activities and solving problems.
One such activity is the M&M® magma chamber, which Wirth designed to help students better understand how magma changes in composition through the effects of crystallization and gravitational settling.
________________
a Except where noted, the information in this example comes from an interview with Karl Wirth, July 8, 2013.
This is one of the most important processes in geology, and one that students often have difficulty understanding, says Wirth. The activity is also intended to provide students with an opportunity to use and reinforce concepts learned in a mineralogy or petrology course, such as stoichiometry or classification and chemical variation diagrams, and to give them practical experience in designing and using spreadsheets (Wirth, n.d.).
Using different colors of M&Ms to represent different elements, students work through a 10-step simulation of the crystallization process, in which they remove the M&Ms in specific proportions to create representations of rock layers formed by the accumulation of crystals at the bottom of a “magma chamber” drawn on butcher paper. After each step, they calculate and record on spreadsheets the relative percentages of each element remaining, as well as the proportion of magma remaining as a fraction of the original magma. They graph the changing composition in the layers. The diagram below shows the M&M magma chamber at four stages of evolution (Wirth, 2003, p. 4).
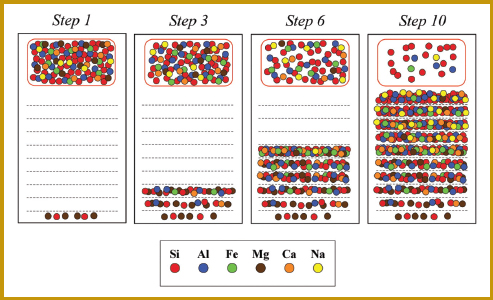
The M&M magma chamber at four stages of evolution.
SOURCE: Wirth, 2003, p. 4.
As a wrap-up, students apply and reflect on what they have learned from the activity by answering a series of questions about changes in minerals and the composition of the magma at various stages, the reasons why certain transformations occur, and which aspects of the simulation are realistic and which are not. They apply their understanding of magma differentiation by crystal fractionation to predict what would happen if certain circumstances were changed and to determine the approximate volume of basaltic magmas needed to produce the lavas of the Yellowstone Plateau.
“It’s fun, it’s colorful, it’s kinesthetic, they’re moving things around—it’s chaos,” says Wirth of the exercise, which has been widely disseminated as part of the Science Education Resource Center (SERC) collection of exemplary teaching activities.b This exercise also uses a familiar analogy of sorting candies by color as a “bridge” to help students understand a complex concept—a strategy consistent with research on learning. After students work through the M&M model, says Wirth, they understand and remember the underlying concept much better than they did when he taught it by lecturing for 20 minutes and drawing with colored markers on the board. Since he incorporated this exercise into the curriculum, “student knowledge of fractional crystallization and magmatic differentiation, as indicated by exam results and course projects, has increased significantly,” he reports (Wirth, 2003, p. 1).
________________
b See http://serc.carleton.edu/NAGTWorkshops/petrology/teaching_examples/24646.html.
Following is a summary of general findings about learning with the greatest relevance to undergraduate science and engineering education. This information is drawn mainly from How People Learn: Brain, Mind, Experience, and School: Expanded Edition (National Research Council, 2000); a companion volume, How People Learn: Bridging Research and Practice (National Research Council, 1999); and the sections on metacognition and knowledge transfer in Chapter 7 of the 2012 NRC report on DBER.
Prior knowledge shapes learning
Learners do not come to a new topic knowing nothing, particularly by the time they are undergraduates. Learners of all ages possess understandings, skills, and beliefs that significantly influence how they remember, reason, solve problems, and acquire new knowledge.
Prior knowledge can either facilitate or interfere with new learning. When students’ prior knowledge is accurate, it can provide a foundation for constructing new knowledge. When students’ existing ideas, or preconceptions, are basically correct but incomplete, instructors can use what students do know as a bridge to help them fill in gaps in understanding. But when students have misconceptions—ideas, beliefs, and understandings that differ from accepted scientific and engineering explanations—they may have difficulty integrating new knowledge with their inaccurate notions. Misconceptions can persist through the undergraduate years, even when students have been taught accurate explanations in their earlier science classes. In your own courses, you may have encountered students who cling to misconceptions like these: Individuals can evolve during a single lifespan. Chemical bonds store energy that is used to make them. As discussed later in this chapter, DBER has identified prevalent misconceptions in specific science and engineering disciplines.
Rather than simply telling students the right explanation, good instructors make an effort to elicit students’ prior knowledge and use it to help students construct a more complete and accurate understanding.
Learning is a process of actively constructing knowledge
Learning is not simply the accrual of information; rather, it involves a process of conceptual reorganization. The brain is a “dynamic organ”; even a mature brain is structurally altered during learning (NRC, 2000, p. 235). The brain actively seeks to make sense of new knowledge by connecting it with prior knowledge and experience. Through this process, the learner “constructs” new understanding and meaning.
Constructing new knowledge is easier when a student has a strong foundation of sufficient, well-organized, and accurate knowledge on which to build. It becomes more difficult, however, when it requires an upending or complete restructuring of students’ current understanding. In this case, instructors will need to address inaccurate or incomplete preconceptions and guide students in reorganizing their thinking in more fruitful ways, as discussed later in this chapter.
A related idea from research on cognition emphasizes that meaningful learning occurs when students select, organize, and integrate information, either independently or in groups, and take control of their own learning (National Research Council, 2000, 2012). This principle of “active learning” has strongly influenced many DBER scholars and studies and undergirds many research-based strategies for teaching science and engineering.
This view of a student as an active constructor of knowledge does not mean that instructors should never tell students anything directly. In some situations, “teaching by telling” can be an effective part of a broader instructional design, but only after students have been primed for this “telling” process by grappling with the ideas on their own in a carefully structured way (Schwartz and Bransford, 1998). Even during “times for telling,” instructors still need to attend to students’ interpretations and provide guidance when necessary.
Experts organize knowledge and approach problems differently from novices
The work of a science or engineering instructor might be seen as a process of moving students from novice toward more expert-like understanding in a discipline. Undergraduates cannot be expected to develop the expertise that it has taken you, as a professional, many years of dedicated practice to attain. Your goal, then, is to help move students farther along this continuum. Instructors sometimes have difficulty with this process because of blind spots—for instance, they fail to see that a step in problem solving that is automatic to them as an expert may be a substantial challenge for novices.
Research provides insights about differences between how novices and experts think and perform. Acquiring a rich body of knowledge in a discipline is a necessary starting point for developing expertise. To become an expert, one must spend enough time studying and working in a discipline to master its content. The more one knows about a subject, the easier it is to learn still more.
But expertise consists of more than just knowing an impressive array of facts. What truly distinguishes experts from novices is experts’ deep understanding of the concepts, principles, and procedures of inquiry in their field, and their framework for organizing this knowledge. Experts also know when and how to apply particular
aspects of their knowledge, and their mental organizations make it easier for them to remember and access relevant knowledge. This depth and organization of knowledge enables experts to notice patterns, relationships, and discrepancies that elude novices. It allows them to quickly identify the relevant aspects of a complex problem or situation, make inferences, and draw conclusions.
Meaningful learning occurs when students select, organize, and integrate information, either independently or in groups, and take control of their own learning.
The knowledge that novices possess, by contrast, is often disconnected, unorganized, and therefore less usable. Novices do not always connect the relevant knowledge they do have to new tasks. And they may focus on aspects of a problem, such as superficial details, that make it more difficult rather than easier to solve.
Thus, while students need to acquire a foundation of knowledge in a discipline, this is not enough to become competent. Students also need to be able to affix their knowledge to a coherent mental framework. Instructors can help students develop more expert-like understanding by emphasizing organized bodies of knowledge and embedding specific ideas, principles, and concepts within these structures. Because knowledge is organized differently for different disciplines, attaining expertise in an area requires knowledge of both its content and its broader structural organization. More detailed findings from DBER about how novices and experts solve problems and approach other aspects of learning science and engineering are discussed in a later section of this chapter.
Metacognition can help students learn
Metacognition—the mind’s ability to monitor and control its own activities—is an essential competency for learning. Students who have greater metacognitive capacity are better learners overall. They monitor their comprehension as they learn: for example, by asking themselves when they encounter a new concept whether they truly understand it, or by pausing to consider whether their strategy is working when they tackle a problem. If they find they do not understand or are not making headway, they can take corrective steps.
Although relatively few students report using metacognitive strategies when studying on their own (Karpicke, Butler, and Roediger, 2009), research suggests that students can develop metacognition over time when metacognitive strategies
are embedded into instruction (Weinstein, Husman, and Dierking, 2000). The reflection exercises in Wirth’s classroom, described above, are one way to build metacognitive activities into science teaching.
In chemistry, students who took laboratories designed specifically to prompt metacognitive activity showed significant gains on the Metacognitive Activities Inventory, which measures students’ monitoring of their own thinking during problem solving (Sandi-Urena, Cooper, and Stevens, 2011). Consistent gains on this inventory were also found among students who participated in a workshop designed to promote metacognition (Sandi-Urena, Cooper, and Stevens, 2011). Studies in engineering education have found that incorporating reflection steps and self-explanation prompts into instruction can improve students’ problem solving (Svinicki, 2011).
Activities that require students to generate their own explanations of concepts or explain a concept to another person also have a metacognitive element. Studies indicate that these “self-explanation” strategies can enhance learning more than just having students read a passage or examine the diagrams in a textbook (National Research Council, 2012).
Students who can transfer their knowledge to new situations learn more readily
If students can apply what they have learned only in conditions that are exactly the same as those in which they learned it, their education will have little practical value. The ability to “transfer” knowledge to new contexts inside and outside the classroom helps students learn related information more quickly. Knowledge transfer is a mark of a well-educated person and an ultimate goal of education—but it is often an elusive goal.
There are different degrees of transfer. “Near transfer” occurs among highly similar tasks in the same setting, such as using knowledge learned from one type of problem to solve similar problems in the same course or a sequential course in the same discipline. For example, physics students who have learned to apply Newton’s second law of motion to a problem involving a block on an inclined plane should be able to recognize that they can apply the same law to understanding the data collected in a physics lab experiment.
“Far transfer” takes place when knowledge learned in one setting is applied in a distinctly different setting, such as transferring what has been learned in one course to a course in a different discipline or using what has been learned in the classroom in a new professional context or everyday situation outside of school. Far transfer is more challenging for students to master.
Students often have difficulty applying their knowledge in a new context, according to DBER studies. Research in chemistry, for example, has demonstrated that while students can memorize how to solve problems that require them to manipulate symbols and chemical formulas, they typically cannot transfer these skills to a similar problem involving drawings of atoms and molecules (see, for example, Nakhleh and Mitchell, 1993). As discussed later in this chapter, students’ difficulties in transferring knowledge across various types of problems are often related to their inability to distinguish the critical features of a problem.
Findings from How People Learn and the 2012 NRC report on DBER shed light on the kinds of learning that support knowledge transfer. Students must have a body of sufficient knowledge about a particular topic, but this should consist of more than a collection of facts. For students to be able to transfer what they have learned, they need to understand the core concepts related to that topic that can serve as a structure for organizing their knowledge. In biology, for example, students would be expected to know the facts that arteries are thicker and more elastic than veins and carry blood away from the heart, while veins carry blood back to the heart. But to be able to apply their knowledge of the circulatory system to a new problem, students must also understand why arteries and veins have these different properties and how these properties are integral to their distinct functions (National Research Council, 2000, p. 9).
Spending a lot of time studying material and practicing its application is not sufficient to promote transfer of knowledge; what matters is how this time is spent. The goal is to spend time on activities that promote deeper learning. Students are more likely to develop the kind of flexible understanding that supports transfer if they learn how to extract themes and principles from their learning activities. Some instructors address this by calling attention to underlying principles and designing activities in which students explicitly practice transfer.
Moreover, if students learn a concept mostly by working on problems and examples that are similar in context—such as problems involving balls that are thrown upward or dropped from buildings—their knowledge can become “context-bound” (National Research Council, 2000, p. 236). This can be addressed by using different kinds of problems and examples that encourage students to extract the relevant features of a concept—to think in terms of problems of gravitational force and energy rather than problems involving balls. Giving students complex, realistic problems can also provide them with practice in transferring their knowledge to a new situation.
Interactions with others can promote learning
Much in the way that children learn to talk by hearing the people around them converse or that adults acquire new skills by working alongside colleagues, students construct understanding through social interactions, such as talking about and collaborating on meaningful learning activities (Vygotsky, 1978). The evidence is very strong that collaborative activities enhance the effectiveness of student-centered learning over traditional instruction and improve retention of content knowledge (see, for example, the meta-analyses by Johnson, Johnson, and Smith, 1998, 2007, and numerous other studies cited in Chapter 6 of the 2012 NRC report on DBER).

When students work together on well-designed learning activities, they establish a community of learners that provides cognitive and social support for the efforts of its individual members. In such a community, students share the responsibility for thinking and doing. They can help each other solve problems by building on each other’s knowledge, asking each other questions, and suggesting ideas that an individual working alone might not have considered (Brown and Campione, 1994). By challenging each other’s thoughts and beliefs, they compel the members of the group to be explicit about what they mean and to negotiate any conflicts that arise, which in turn fosters metacognition. Social interactions also have a positive effect on motivation by making individuals feel they are contributing something to others (Schwartz et al., 1999).
Much in the way that children learn to talk by hearing the people around them converse or that adults acquire new skills by working alongside colleagues, students construct understanding through social interactions, such as … collaborating on meaningful learning activities.
Instructors can help create this sense of community by designing learning activities that encourage this type of intellectual camaraderie and by creating classroom environments in which all students, including those from groups underrepresented in science, feel safe about sharing their ideas. To be effective, these approaches must be carefully selected and implemented and well aligned with student learning outcomes and assessment procedures. Chapter 4 gives some examples of effective collaborative approaches.
![]()
Many DBER studies are grounded in general findings about learning from cognitive science and related fields. But DBER goes deeper by looking at how students learn the knowledge, practices, and ways of thinking in a science or engineering discipline. Much of this body of work focuses on three aspects of learning that are central to developing competency in these disciplines at the undergraduate level:
- Understanding and applying the fundamental concepts of a discipline
- Framing and solving problems with greater expertise
- Using visual and mathematical representations, such as graphs, models, and equations, of important ideas and situations in a discipline
While other aspects of learning science and engineering have also received scholarly attention, these three have been studied the most extensively. Across disciplines, DBER has identified common challenges in these three areas that can impede students’ learning, as well as approaches that can further learning. The core findings in these three areas, which are discussed in the sections that follow, are a good entry point for science and engineering instructors who want to use research to improve their teaching.
Understanding and Applying the Fundamental Concepts of a Discipline
Each of the disciplines discussed in this book is built on a set of fundamental concepts—ideas that can be applied in multiple contexts to explain and predict scientific phenomena. To become competent in biology, for example, students
need to understand the concept that species arise, change, and become extinct over time. Students of chemistry must comprehend that the atoms of a compound are held together by chemical bonds formed by the interaction of electrons from each atom.
Students often have difficulty mastering the fundamental concepts of a discipline. These concepts tend to be abstract, and students may fail to recognize their value as keys to thinking about the discipline.
DBER has helped to elucidate how students develop an understanding of central science and engineering concepts and where they run into difficulty. An extensive body of DBER scholarship has identified and analyzed common student misconceptions in specific disciplines. DBER studies have also examined the effectiveness of strategies for promoting conceptual change. This research is reviewed at length in Chapter 4 of the 2012 NRC report on DBER; the major findings are summarized below.
Misconceptions
In every science and engineering discipline, undergraduates harbor misconceptions. These misconceptions are often derived from what students have observed in their own experience or what seems to be common sense. In physics, for example, students may think that denser objects fall more quickly than lighter objects in a vacuum because they have seen a rock plummet to the ground while a leaf wafts slowly downward. In biology, many students have an inaccurate “round-trip” notion of the human circulatory system in which they envision blood flowing in a continuous circle from the heart around the body before returning to the heart (Pelaez et al., 2005). Incorrect ideas may also arise from inaccurate instruction in the K–12 grades or be influenced by cultural or religious beliefs.
Across disciplines, some of the most difficult concepts for students to grasp are those for which they have no frame of reference, especially those that involve very large or very small scales of space or time. In chemistry, for example, the idea that all matter is composed of particles too small to be seen with a microscope—molecules, atoms, and subatomic particles—is one of three main domains of knowledge students are expected to master. This “particulate” domain is often represented as one corner of “Johnstone’s triangle”; the other two corners are the macroscopic domain, or entities and properties that can be perceived with the human senses, and the symbolic domain, such as the letters and numbers used to represent compounds (Johnstone, 1991). While students struggle to comprehend all three domains, understanding the particulate nature of matter is one of their greatest barriers to learning chemistry (Gabel, Samuel, and Hunn, 1987; Yezierski and Birk, 2006).
In geosciences, biology, and astronomy, the concept of “deep time” often confounds students. Deep time refers to the age of Earth or the universe and involves time scales spanning billions of years (see Box 3.1).
Misconceptions about scientific and engineering concepts do not always surface during traditional instruction. Moreover, deeply rooted misconceptions can be hard to change. For example, even some students who have completed undergraduate chemistry
BOX 3.1 HOW LONG IS 4.5 BILLION YEARS, REALLY?:
COMPREHENDING DEEP TIME
Most of us have seen a chart in the form of a metaphorical calendar that compresses the entire history of Earth into the scale of one single year and places the first appearance of humans in the final minutes of the last day of that year. This notion of “deep time,” or geologic time—the well-established concept that Earth is billions of years old—is one of the revolutionary ideas in geology and a framework for understanding evolutionary biology (Catley and Novick, 2008). In astronomy, deep time extends back even further, across several billion years to the Big Bang and the origin of the universe. In all three disciplines, the very large scales make deep time a difficult concept for students to grasp.
Research has found that although most undergraduate students place significant events in geologic history—such as the formation of Earth, the first appearance of life, and the arrival of dinosaurs—in the right order, they misunderstand the scale of time between events (Libarkin, Kurdziel, and Anderson, 2007). Very few students produce estimates that are close to the scientifically accepted timeline. Students also sometimes conflate events that are far apart, such as the age of the dinosaurs and the age of humans. While the majority of students recognize that Earth is very old, some hold a “young Earth” perspective, including some who explicitly embrace creationist beliefs (Cervato and Frodeman, 2012; Libarkin, Kurdziel, and Anderson, 2007).
An analysis of 79 peer-reviewed studies of geosciences misconceptions (Cheek, 2010) notes that a poor understanding of large numbers could partially account for students’ difficulty in understanding geologic time. In some of these studies, participants seemed to pick the largest number they could think of but showed no real sense of how much time it represented.
Students’ problems with the concept of deep time have wide-ranging implications for learning because so many areas of geology, as well as biology and astronomy, are premised on this idea. Concepts such as plate tectonics, rock layering, and sedimentation, among others, all depend to some extent on an understanding of geologic time. Libarkin, Kurdziel, and Anderson (2007) suggest that instructors incorporate a thorough discussion of the basic concept of deep time early in their introductory courses and reiterate its effects on other aspects of geology throughout the course. Dodick (2012) proposes that instead of simply focusing on the raw numbers in the chronology of deep time, instructors direct students’ attention to the significance of the numbers by anchoring a specific time period with key events they can visualize, much in the way that people might associate a particular month with their birthday.
courses still stand by the misconception that chemical bonds release energy when they break (Sözbilir, 2004). If instruction does not address misconceptions like these, students may fail to grasp new concepts and information. Or, they may learn them well enough to pass a test but go back their old, inaccurate ways of thinking outside the classroom.
It’s especially important to probe the reasoning that underlies misconceptions, says Lillian McDermott,3 a physics professor at the University of Washington who has designed tutorials and other interventions to facilitate conceptual change. “The problem with the term ‘misconceptions research’ is that it seems to imply that all that needs to be done is to identify a mistaken interpretation of a concept and replace it with a correct one. It is the reasoning in physics, however, that distinguishes related concepts from one another, identifies their relationship, and makes possible their correct application,” writes McDermott in a forthcoming manuscript.4 “Our emphasis has therefore not been on the eradication of misconceptions, but rather on the development of reasoning skills necessary for the proper application of concepts.”
Assessing students’ conceptual understanding
To improve conceptual understanding, instructors first need to determine what students know, what they understand incompletely, and where they have misconceptions. With this information in hand, instructors can then help students replace or refine misconceptions and use what they already know as a framework for building a more complete and accurate understanding.
As discussed in more detail in Chapter 5, scholars have designed various tools to assess students’ conceptual understanding. These range from formal instruments like concept inventories to everyday classroom methods like ConcepTests in the form of clicker questions. It is often necessary to use more than one type of assessment. Pelaez and colleagues (2005) found that their essay exams were insufficient to expose the extent of common student misconceptions about the circulatory system and that other assessment methods, including drawings and individual interviews, were required to discover how and what students thought.
A relatively simple way of regularly assessing how well students understand the concepts being taught is to have them reflect in writing on the concepts that confuse them—soliciting the “muddiest points,” as Patricia Cross and Thomas Angelo (1988) called them in their widely used handbook on classroom assessment techniques. Stephen Krause, an engineering professor at Arizona State University, has adapted this approach in his courses.
________________
3 Interview, April 13, 2013.
4 Unpublished manuscript by Lillian McDermott titled A View from Physics: Discipline-Based Education Research in a University Physics Department, 1973–2013.
Clarifying the Muddiest Points in an Engineering Class
How do instructors know which ideas in their course are misunderstood by or confusing to students? And once they know, how do they address that?
One quick and simple way of obtaining feedback is to ask students to reflect anonymously on what they found to be the “muddiest points” discussed during each class—the concepts or other issues that remain unclear to them or that they feel uncertain about. Stephen Krausea uses this approach in his introductory engineering courses at Arizona State and is part of a group of engineering faculty members who have studied the impact. While the idea of soliciting muddiest points from students was first mentioned by Cross and Angelo (1988), Krause and others have refined the approach in various ways, such as using Web software to collect and display students’ reflections and adding YouTube tutorials that explain difficult concepts.
At the conclusion of each class, Krause’s students take a few minutes to fill out a worksheet that asks them to identify the concepts and topics they had trouble understanding and to rate the degree of difficulty they experienced with a particular concept. Their responses are catalogued in a spreadsheet that the instructor and assistants review. This direct feedback enables Krause to readily gauge how well

Word cloud in which the size of type reflects the degree of students’ confusion about the term.
________________
a Except where noted, the information in this case study comes from an interview with Stephen Krause, July 9, 2013.
students understand the course content and address misunderstandings in the next class. In addition, students can anonymously access a running catalogue of their own responses to see how their thinking has progressed during the semester.
At the beginning of each class, Krause displays a selection of the muddiest points from the previous session using the students’ own words. (Krause employs a slide or the online Blackboard® software to display the results; some other instructors use the online Concept Warehouse system to generate a word cloud of the muddiest points.) For example, after a lesson on crystallographic planes—geometric planes linking the nodes (atoms, ions, molecules) of a crystal—one student raised this question: Why are the crystallographic planes important? Following a lesson on phase diagrams, which represent the various phases of a substance under different pressure and temperature conditions, a student was confused about this point: How do I find chemical composition and phase fractions from a phase diagram? Bethany Smith,b who served as Krause’s undergraduate assistant in her junior year of 2012–2013, admits that she herself was initially confused by phase diagrams when she took the course as a sophomore.
Krause uses the first 5 or 10 minutes of class to address the most common muddiest points. Smith says this type of review discussion helped her to better understand phase diagrams. In fact, she adds, much of what she learned in the sophomore materials class has proved to be “definitely useful” in later classes in the materials science sequence.
After clarifying the muddiest points, Krause moves on to a mini-lecture to prepare students for the activities they will do that day. “The nice thing about the muddiest points is that they activate the knowledge from the previous class and provide a connection to the current class,” he says. “They can provide segues into that day’s mini-lecture.”
Krause and Smith have also produced a series of YouTube tutorials (https://www.youtube.com/user/MaterialsConcepts) that list a few of the muddiest points cited by students for a particular topic and explain each one using voiceover narration and visual aids. Students view the videos to clarify difficult concepts and help them with homework. Preliminary results have found significant gains in achievement on the content included in the YouTube tutorials, compared with test results for previous classes (Krause et al., 2013a).
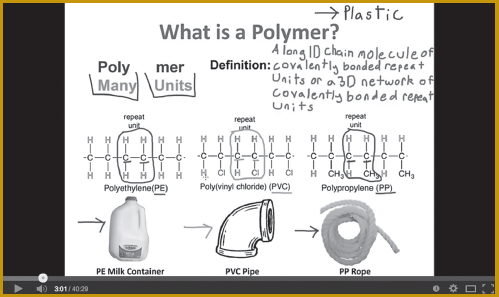
Screen capture from a YouTube tutorial.
The muddiest point reflections are just one part of Krause’s student-centered instructional approach, which he calls Just-in-Time Teaching with Interactive Learning (JiTTIL). This approach is described in Chapter 4.
________________
b Interview, July 11, 2013.
Teaching for conceptual understanding
DBER has identified various instructional strategies that can help students develop a deeper and more accurate understanding of important concepts and, where necessary, promote conceptual change. This type of change depends on students recognizing that their preconceptions are not facts but hypotheses or models that must be evaluated in light of empirical evidence (National Academy of Sciences, National Academy of Engineering, and Institute of Medicine, 2005). Many of these strategies seek to create situations in which students realize that their preconceptions conflict with new evidence and that they must change their thinking to fit with new knowledge.
To adapt their thinking to new evidence, students may need to add, remove, or revise elements of an existing mental model; create a new model where there was none before; or replace a preconception with a different and better one—or make each of these changes at different times (Clement, 2008). Because students’ preconceptions are nonscientific in different ways, instructors may need to use a variety of approaches, possibly even in the same class, to help students refine or replace these nonscientific ideas and beliefs. And students will need multiple exposures to the same concept in different contexts before they begin to really understand it.
David Sokoloff5 has seen these kinds of change occur in students’ conceptual understanding in his physics lecture and laboratory courses at the University of Oregon. To foster conceptual change and increase student participation in a lecture course, Sokoloff and Ron Thornton, a physics professor at Tufts University, developed a curriculum built around Interactive Lecture Demonstrations (ILDs)—physical demonstrations of scientific phenomena that the instructor conducts in class. In the approach used by Sokoloff and Thornton, students first predict what will happen before the instructor does the demonstration. Students next discuss their predictions in small peer groups and explain their predictions to the whole class. Then the class observes the instructor conducting the demonstration. In the final stage, students compare their observations to their predictions (Sokoloff and Thornton, 2004).
Sokoloff, Thornton, and Priscilla Laws at Dickinson College have also developed a related curriculum for active learning laboratories called Real Time Physics. In these labs, students do experiments supported by real-time, computer-based tools. But before conducting an experiment, students make predictions about the outcome and discuss their predictions in small groups.
________________
5 Except where noted, the information in this example comes from an interview with David Sokoloff, July 10, 2013.
In both the ILDs and the lab activities, the prediction and discussion phases are essential to the process of conceptual change, says Sokoloff. In many cases, “students are confronted with the fact that the predictions don’t describe the situation,” he explains. This creates a moment of “psychological disequilibrium” in which students recognize they have to restructure their thinking to integrate new information. “For many years, the majority of physics instructors did demonstrations in their classes,” says Sokoloff. “They would do some kind of physics experiment at the front of the room—hopefully as dramatic as possible, because they thought that the more dramatic it is, the more likely that students will learn from it. There’s now some very significant research that shows that unless you do demonstrations in the context of asking students to make predictions about them before you do them, they won’t learn anything from them.” Research by Sokoloff and Thornton (1997) indicates that ILDs can improve students’ understanding of foundational physics concepts as measured by the Force and Motion Conceptual Evaluation. For example, after physics students experienced a sequence of ILDs related to Newton’s third law, they retained an appropriate understanding of the law months later.
The ILDs developed by Sokoloff and Thornton are also sequenced in a way that gradually leads students toward a better understanding of concepts like Newton’s laws. In lessons on kinematics and dynamics, for example, the initial demonstrations are designed to solidify students’ understanding of very basic concepts. Later demonstrations introduce more complex concepts (Sokoloff and Thornton, 1997). (See Box 3.2.)
This progression of ILDs is an example of “scaffolding”—a term first used by Wood, Bruner, and Ross (1976) to describe the process of starting with what students know and providing them with carefully structured support to move them toward more accurate understanding. Scaffolding provides successive levels of temporary support that allow learners to accomplish a task and reach a level of understanding that they would otherwise be unable to achieve without assistance. The idea is that eventually the instructor will systematically remove the scaffolding supports so that students will use the newly acquired concepts and skills on their own.
Another example of scaffolding is the use of “bridging analogies” that connect a situation that students understand correctly with another situation about which they harbor a misconception. For example, students in a physics class may realize that when they hold a book, both the book and their hands are exerting forces, and that the forces are balanced if the book does not move. But if a book is placed on a table, many students fail to understand that an upward force from the table is balancing the downward force of the book. To help students understand
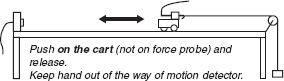
BOX 3.2 INTERACTIVE LECTURE DEMONSTRATION SEQUENCE FOR NEWTON’S FIRST AND SECOND LAWS
Demonstration #1:
The cart (with very small friction) is pulled with a constant force so that it moves away from the motion detector, speeding up at a steady rate.
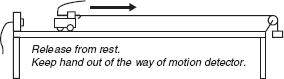
Demonstration #2:
The cart with larger friction (friction pad in contact with ramp) is pulled with a constant force so that it still moves away from the motion detector, speeding up at a steady rate.
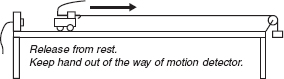
Demonstration #3:
Show that cart accelerates in either direction when only one fan unit is on (as seen in previous demos). With both fans on (balanced), the cart does not move. Now push and release and observe velocity and acceleration.

Demonstration #4:
Cart with very small frictional force is given a brief pull away from the motion detector and released.

Demonstration #5:
The cart (with very small friction) is given a push toward the motion detector and released. A constant force acts in the direction away from the motion detector. The cart moves toward the motion detector, slowing down at a steady rate.

Demonstration #6:
The cart (with very small friction) is given a push toward the motion detector and released. A constant force acts in the direction away from the motion detector. The cart moves toward the motion detector, slowing down at a steady rate, comes to rest momentarily, and then moves away from the motion detector.
that the table is applying a force, an instructor might take students through a series of bridging situations—putting their hand on a spring, placing a book on a foam pad, setting a book on a flexible board—and ask them to discuss similarities and differences with the earlier situations. As part of this lesson about force, the instructor might show students a microscopic model of a rigid object (like a table) that is composed of atoms connected by spring-like bonds. The lesson might conclude with an experiment in which a mirror is placed on the table and someone stands on the table; when a light beam is reflected off the mirror and onto the wall, the beam is deflected downward, indicating that the table has been compressed ever so slightly (Clement, 1993).
In geosciences, evidence on the effectiveness of instructional strategies in fostering conceptual change is derived mostly from studies of individual courses during brief periods. One such study by Rebich and Gautier (2005) found large increases in knowledge and a decrease in misconceptions among students who had participated in a three-week mock summit on climate change; this approach used role-playing, debate, and discussion to heighten awareness of the concepts underlying climate change.
Changing students’ conceptual understanding can be difficult. An example comes from a study of a two-semester sequence of introductory geology in which researchers examined students’ concept maps—diagrams that typically use words or phrases, along with boxes and lines, to connect and show the relationships among a list of concepts (Englebrecht et al., 2005). Although students were able to identify more geological concepts after a sequence of instruction, they showed only small improvement in their ability to integrate those concepts into a framework of understanding.
In sum, improving students’ conceptual understanding takes time and a range of instructional techniques. Chapter 4 describes additional approaches in a variety of disciplines that hold promise for accomplishing this goal.
Framing and Solving Problems with Greater Expertise
Learning how to solve problems is an important part of developing competency in science and engineering. Whether in an educational or a professional setting, the ability to solve problems is central to the practice of science and engineering. Problem solving also comes into play in other areas of everyday life when one needs to reach a goal but is uncertain how to attain it. To solve problems effectively, students must not only have the types of conceptual understanding discussed above,
but they must be able to apply those concepts correctly. They must also bring to bear other sophisticated thinking skills that go beyond rote memory.
Problem solving is a significant focus of DBER in physics, chemistry, and engineering, and it is an emerging area of study in biology and geosciences. This line of research has found that students, as novices, tend to approach, organize, and go about solving problems differently than experts. DBER studies have identified the particular difficulties students experience with various aspects of problem solving. In addition, the research literature offers insights about instructional approaches that can help students develop greater expertise with problem solving. The sections that follow summarize the main findings about problem solving from DBER; a more complete treatment of this topic can be found in Chapter 5 of the 2012 NRC report on DBER.
Focusing on superficial features instead of underlying principles
When people set about solving a problem, they construct a model of how they might approach the problem—in their minds and sometimes in a tangible form like a drawing. These models, whether arrived at deliberately or with little forethought, will guide the steps people take to solve it.
As novices, students approach problems in ways that are consistently and identifiably different from those used by experts. Students typically focus unduly on the superficial features of a problem, such as the specific objects, terms, and phrasing used in a question. Experts, by contrast, look at the deeper structure of the problem—the underlying principles that are required to solve it. In one interview study, for example, undergraduate biology students grouped classical genetics problems according to their surface features, such as whether the problem concerned humans or fruit flies and how it was worded, whereas biology professors grouped them according to key underlying concepts, such as the mechanism of genetic inheritance (Smith, 1992).
When confronted with two problems that have the same underlying structure, experts easily recognize these as the same kind of problem “deep down.” This understanding of the essential features of a problem leads to better reasoning and problem solving. Students, however, may assume the problems are distinctly different due to superficial variations and may construct very different models of the two problems. For example, students may not recognize that a problem involving discs of different sizes stacked on a peg has the same structure as a problem involving acrobats of different sizes standing on one another’s shoulders.
Research from physics, and to a lesser extent from chemistry and biology, supports this finding. When asked to categorize physics problems according to how they are solved, experts grouped them according to the major concepts or principles that could be applied to solve them, such as determining that a problem relates to Newton’s second law. Novices relied much more on surface features, such as whether the problem mentions pulleys versus inclined planes versus springs (de Jong and Ferguson-Hessler, 1986). A study in biology (Kindfield, 1993/1994) compared the diagrams of chromosomes drawn during problem solving by two groups: a group with more knowledge of meiosis and chromosomes and a group with less knowledge. The drawings of the less knowledgeable participants often more literally resembled the way chromosomes looked under a light microscope and included features like dimensionality and shape that were irrelevant to the solution. The more knowledgeable participants included chromosome features that were biologically relevant to the problem.
As novices, students approach problems in ways that are consistently and identifiably different from those used by experts. Students typically focus unduly on the superficial features of a problem, such as the specific objects, terms, and phrasing used in a question. Experts, by contrast, look at the deeper structure of the problem—the underlying principles that are required to solve it.
Research on human cognition (see, for example, Bassok and Novick, 2012) has found that for some problems, getting the right mental model of the problem is a key to finding a solution. Neither the model nor the process is fixed; each influences the other. A process for solving a problem may change as the solver’s model of the problem changes, which in turn may lead to changes in the solutions one tries. However, students need to acquire sufficient expertise before they can recognize when they need to change their strategy instead of moving down the same dead ends, as novices tend to do.
By focusing on superficial aspects, students miss the essence of a problem, which makes it much harder to solve. This approach is also less efficient. Because experts can recognize structural relationships and patterns, they can tap into their long-term memory about what to do when certain patterns are present and can readily see solutions.
A failure to recognize the most salient features of a problem also makes it difficult for students to apply what they have learned from one problem to new problems that are similar in structure but different in context. This type of knowledge transfer is crucial to becoming a more expert-like problem solver.
These findings also suggest that students need a solid grounding in the core principles of a science or engineering discipline—the kind of conceptual understanding discussed above—in order to determine the “deep structure” of a problem. Research in engineering education, for example, indicates that a lack of understanding of fundamental concepts impedes students’ ability to solve problems (see, for example, Baillie, Goodhew, and Skryabina, 2006).
Working “backward” instead of working “forward”
Another difference between novices and experts relates to how much time they spend creating a model of the problem versus working to find a solution. Novices often jump immediately to the end goal of a problem and start looking for an equation that might help them solve it. Then they must use another equation to calculate an unknown quantity in the first equation, and so on, until they find an equation that includes all the necessary quantities. This “working backward” strategy puts a heavy load on their working memory—their ability to hold key information in mind temporarily while they do the work of problem solving. This burden leaves little room for them to learn general strategies for solving similar problems. It also makes it easy for them to forget crucial elements of the problem at hand. And when they get stuck, they lack strategies to proceed.
Experts spend more time analyzing the nature of a problem from the outset and creating a coherent solution strategy. Experts go on to enrich their model of the problem with information from what they know and remember, such as procedures they have used to solve similar problems in the past. In this “working forward” approach, they start with the information given, make inferences based on that information, and continue refining their inferences until they have reached their goal. Experts monitor their progress as they solve a problem and evaluate whether an answer is reasonable.
This difference between the working backward approach of novices and the working forward approach of experts has been documented in numerous studies. In physics, for example, research has shown that expert problem solvers typically begin by considering the qualitative aspects of a problem and using that information to decide on a solution strategy before taking the quantitative step of writing
down equations. Beginning physics students typically start by writing down equations that match the quantities provided in the problem statement and then work backward to find an equation for which the unknowns are supplied directly in the problem (see, for example, Larkin et al., 1980).
Research in chemistry education has found that students tend to rely more on algorithms to solve problems—stuffing numbers into a formula that worked with a very similar problem or applying memorized chemical reactions—than on a logical problem-solving process (Bhattacharyya and Bodner, 2005). Sometimes students can solve problems with these less skillful approaches, even though they have a shallow understanding of the underlying concept (see, for example, Gabel and Bunce, 1994). But these methods will fail when students are confronted with problems that don’t fit the mold.
In order to work forward, problem solvers must have an organized framework of disciplinary knowledge, as well as experience in solving problems in that discipline. A command of basic facts and conceptual knowledge is a necessary part of this framework, but other elements are also critical. The framework should include discipline-specific models for approaching problems, as well as criteria for selecting the model appropriate to a context, determining which information in the problem is relevant and which is not, and evaluating whether an answer makes sense (Mayer and Wittrock, 2006).
Helping students to improve problem-solving skills
Taken together, these findings suggest that it is important for science and engineering instructors to help students recognize the need for both a good mental model of a problem and a sound method to solve it. When students run into difficulties in solving problems, they first need to consider alternate ways of representing the problem and then contemplate possible methods for figuring out an answer. Some instructors ask students to justify why their proposed procedures for solving a problem are reasonable. Toward this end, instructors might provide examples of how a good model can make it easier to find a solution, while a flawed one can make it harder.
A body of research indicates that problem-solving skills can be taught and that carefully designed forms of scaffolding appear to benefit students. The learning gains from any one type of support appear to be small and difficult to measure, however. These findings point to the wisdom of using multiple forms of scaffolding within a systematic approach to improving students’ problem-solving skills.
Research has investigated a variety of strategies for moving students from novice toward expert problem-solving approaches. Promising strategies include the following (for specific references, see Chapter 5 of the 2012 NRC report on DBER):
- Teaching specific, organized methods for solving problems
- Explaining the different problem types
- Providing examples of problems with the solutions worked out
- Providing guidance and greater classroom interaction
- Having students solve problems in collaborative groups
- Making symbols more transparent to students by using more explicit labeling
- Assigning authentic (real-world) problems
- Assigning open-ended problems that encourage students to invent and test various models to solve them
- Incorporating prompts for students to reflect on and explain their approaches to solving problems
Chapter 4 of this book discusses several broader instructional approaches aimed at improving problem solving and other aspects of science and engineering learning.
Using Visual and Mathematical Representations
In every science or engineering discipline, visual, spatial, and mathematical representations are essential tools for communicating and remembering ideas and solving problems. A map of a rock outcrop; the formula for determining frictional force; a graph of the density of different species in a habitat; a chemical structure that shows the shape of a molecule and the bonding between its atoms; an engineer’s free-body diagram that depicts all of the forces acting on an object—these are just a few of the myriad representations that are common in science and engineering disciplines.
Representations serve several purposes. They enable people to communicate ideas within a discipline in a shorthand way. By storing information succinctly,
they free up working memory that can be devoted to other thinking processes. In some cases, representations can simplify the nature of a task. Consider, for example, how most people can estimate proportions more easily by looking at a pie chart than by studying a numerical table. Representations also assist in problem solving and other types of critical thinking. Some representations are created to analyze a phenomenon in research; these may be quite complicated and targeted mainly at other researchers in the same field. Other representations are intended to convey information to someone else; these may omit the complexities in order to better communicate the central idea (Dutrow, 2007).
Each discipline has its own common ways of representing key concepts that are easily recognizable to experts. For students to communicate conversantly in a
discipline, they need to be able to interpret and use the major types of representations for that discipline. Just as importantly, they need to understand the concept a particular representation is intended to convey and know why both the representation and the underlying concept are important.
Research suggests that when students construct their own representations, in addition to interpreting those produced by experts, they are often more engaged and learn better (Ainsworth, Prain, and Tytler, 2011). When instructors observe how their students interpret, use, create, and translate among different types of
representations, they can gain insights about how well students understand important concepts. When modeling—a pervasive but infrequently taught aspect of engineering—is taught explicitly, students gain a better understanding of how to use models and why they are important (Carberry and McKenna, 2014).
Representations that instructors and other experts can easily interpret may completely befuddle undergraduates, however. DBER studies and cognitive science research highlight the challenges students face in mastering representations. Findings from this body of research are described in Chapter 5 of the 2012 NRC report on DBER; the main points are summarized below.
Interpreting and constructing representations
Across disciplines, students often have difficulty interpreting representations and constructing their own from existing information. In physics, a field with a strong research base on this topic, students struggle to interpret representations that are common in introductory courses (Rosengrant, Etkina, and Van Heuvelen, 2007). Students often misunderstand the quantities and concepts being represented in diagrams, according to some research, and they shy away from using them because they have few opportunities to practice the skills needed to construct diagrams (Van Heuvelen, 1991).
A good example of the difficulties students confront in interpreting and creating representations comes from chemistry, another discipline with a considerable research base on representations. As an initial step toward understanding the relationships between the molecular structure of a material and its properties, chemistry students are often taught to draw and manipulate diagrams called Lewis structures. Many students struggle with this task (see Box 3.3).
In a related vein, many students have difficulty extracting the most salient information from representations. As in problem solving, novices often have trouble seeing beyond superficial but irrelevant features of a representation to grasp the abstract idea being represented (Hegarty, 2011). Even when students know the conventions for how a diagram is meant to represent reality, they tend to miss important patterns that experts pick up.
Students also have difficulty processing diagrams that violate familiar conventions. For example, students often try to process visual representations from left to right because that’s the direction in which they read text (Nachshon, 1985). In diagrams where this left-to-right processing makes it difficult to interpret relationships, simply shifting the diagram 180 degrees above the vertical axis can improve comprehension (Novick, Stull, and Catley, 2012). Similarly, circles and
BOX 3.3 “LOST IN LEWIS STRUCTURES”:
STUDENTS STRUGGLE WITH A BIG IDEA IN CHEMISTRY
To understand chemistry, students must understand that matter is made up of atoms bonded together into molecules and that the properties of a material can be predicted from its molecular structure (and vice versa). As an initial step toward comprehending the relationships between molecular structure and properties, students are often taught to draw and manipulate Lewis structures. These diagrams, which are common in chemistry, use atomic symbols, lines, and dots to show the arrangement of atoms and electrons in a molecule and the bonds between atoms. While concise in design, Lewis structures are packed with important information that can be used to predict and explain the physical and chemical structure of a substance (Cooper et al., 2010). Knowing how to construct them is an essential skill in chemistry.
For chemists, drawing a Lewis structure is second nature. Rules for how to do this are found in most chemistry textbooks. Yet many students struggle with this task—they get “lost in Lewis structures,” as Melanie Cooper and her colleagues have described the problem (Cooper et al., 2010). “What may appear to the expert to be a simple task is, in fact, inherently difficult, complex, counterintuitive, and all too often meaningless to many students” (p. 869).
Cooper and her colleagues (2010) tracked the processes used by undergraduate students in general and organic chemistry, as well as by graduate students and faculty members, as they drew Lewis structures. Many students, and even a few faculty members, were confused about how to draw valid Lewis structures. As the number of atoms in the diagram increased from six to seven or more, the percentage of students who drew accurate representations plummeted. The increase from six to seven atoms represents a shift to a molecular structure with more than one carbon atom. Indeed, students had difficulty drawing even one-carbon compounds if they were not given structural clues. Students’ success in producing the correct representation depended a great deal on how the formula was initially presented to them, “suggesting that they were relying on memorized cues rather than an understanding of the rules involved” (Cooper et al., 2010, p. 871).
Students often assume, incorrectly, that all Lewis structures must be symmetrical, or “balanced.” Many Lewis structures are symmetrical, such as the correct structure of dimethyl ether shown on the left in the figure in this box. But this misconception about symmetry may lead students to produce incorrect structures, such as the one for methanethiol on the right in the figure in this box (Cooper et al., 2010, p. 871).
A reason for students’ confusion, note Cooper and colleagues, is that conventional approaches to teaching Lewis structures conflict with findings about how people learn. The rules for drawing
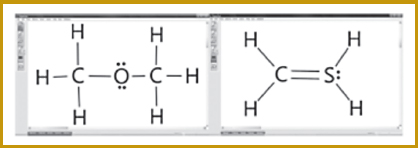
Examples of symmetrical Lewis structures produced by students. The structure on the left for C2H6O is correct; the one on the right for CH4S is incorrect.
these structures are presented to students without connecting them to concepts that students already understand, which makes them seem mysterious. The rules also include numerous exceptions, but students are not given meaningful criteria for deciding when they apply.
Interviews conducted for the study revealed that most students did not understand the kinds of chemical information that can be inferred from Lewis structures. Many students emerge from chemistry courses with a “fractured and muddled” understanding of not only how they should create Lewis structures, but also why they should do it (Cooper et al., 2010, p. 872).
Based on this research, Cooper and colleagues (2012) have developed and evaluated a chemistry curriculum that emphasizes the critical connection between energy changes and atomic interactions as a core concept. Within this curriculum, students learn about key concepts, such as the properties of materials and the different models of bonding, before they are asked to draw Lewis structures. To gain familiarity with the structures involved, students work with physical and computer-based three-dimensional models of simple molecules. At that point, students are introduced to Lewis structures as convenient two-dimensional “cartoons” that represent three-dimensional structures rather than as an end in themselves (p. 1,352). After students have practiced going back and forth between two- and three-dimensional representations, more complex structures are introduced. Once students are able to draw simple structures from a given molecular formula, they move on to the task of decoding the information contained in the formula.
Students taught with this curriculum show marked improvements in their ability to create structures, compared with a control group of students (Cooper et al., 2012). They also do significantly better at decoding the information contained in these structures.
lines seem more naturally suited to representing physical objects or locations than relationships or motion (Tversky et al., 2000). When diagrammatic representations are consistent with these conventions, college students are able to make appropriate inferences more quickly and accurately (Hurley and Novick, 2010).
Translating among different representations of the same thing
Undergraduates also struggle to see similarities among different representations that describe the same phenomenon. In chemistry, for example, students have difficulty translating among alternative ways of representing the same set of relationships, such as videos, graphs, animations, equations, and verbal descriptions (Kozma and Russell, 1997).
Take, for example, the diagrams used in biology to represent the evolutionary relationships among groups of organisms. These diagrams, called cladograms, typically take the form of a tree or a ladder, or in some cases circles nested within larger circles. Biology students have trouble understanding these diagrams and translating among alternative formats that show the same set of relationships (see, for example, Novick, Stull, and Catley, 2012).
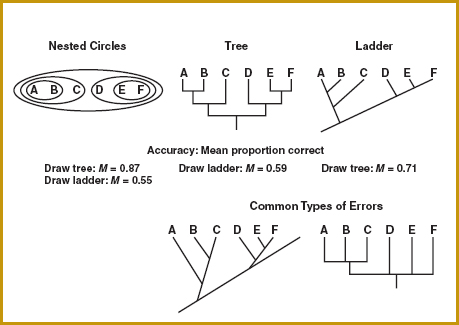
Diagrams representing evolutionary relationships.
A study by Novick and Catley (2007) asked students who had taken at least one semester of an introductory biology class for majors to transfer a hierarchy of relationships from the nested circles format to the tree and ladder formats, and from the tree format to the ladder format and vice versa. Students’ diagrams were less accurate whenever the ladder format was involved. One factor that appeared to confuse students was the use of the long, slanted “backbone” line of the ladders. Consistent with a principle from psychology known as “good continuation,” students interpret this continuous line as a single entity—in this case, a single level in the hierarchy. In follow-up work, students were given ladder diagrams that departed from the traditional format by “breaking” good continuation at exactly the points in the diagram that mark a new hierarchical level, and their performance improved greatly (Novick, Catley, and Funk, 2010).
Spatial ability
Using representations effectively requires a reasonable level of spatial ability; that is, competence in mentally manipulating two- and three-dimensional objects. In engineering design, for example, the abilities to visualize a three-dimensional image from a two-dimensional representation and to mentally rotate three-dimensional objects are important to success. Spatial ability is also an essential skill in geosciences: practitioners and students must envision what lies behind, beneath, or between rock outcrops and make inferences about rock deformation, temperature trends, and other events from the shape of natural objects like minerals and fossils. To understand chemistry, students must learn how to create visual representations of the unseen molecular level and translate those representations into the format of an equation.
Students often have difficulties with spatial thinking. In geosciences, some students find it challenging to visualize what a three-dimensional volume would look like when they are presented with information in one or two dimensions. In one study (Kali and Orion, 1996), students were given a two-dimensional drawing that depicted three sides of a block of Earth’s crust and were asked to draw a slice, or vertical cross-section, of the block. Students who gave incorrect answers had difficulty “penetrating” the block, as shown in the figure below; some simply copied a pattern from one side of the block. Students who were more successful with this task used specific strategies to visually penetrate the block, such as continuing a pattern vertically or horizontally.
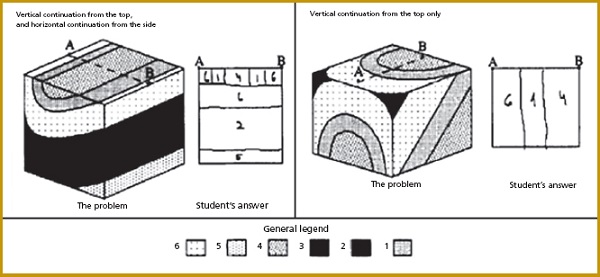
Examples of incorrect attempts by students to draw a vertical cross-section of a block of Earth’s crust.
Although the evidence is limited, some research suggests that explicit instruction can improve students’ spatial visualization skills. A review of several studies in engineering found that specially designed multimedia training courses enhanced these skills, especially for female students (Sorby, 2009). Some research indicates that teaching geosciences with visually rich materials can improve students’ spatial visualization skills and in some cases reduce gaps in this area between male and female students (see, for example, Titus and Horsman, 2009). Evidence in chemistry is mixed; some studies have concluded that teaching analytic problem-solving strategies can lead to greater improvements in students’ mental models than does emphasizing students’ natural spatial ability (Hegarty, Stieff, and Dixon, 2013).
Animations and simulations
Animations, interactive computer simulations, virtual models, and other technology-based representations are widely used in the practice of science and engineering and are becoming increasingly popular in undergraduate education. By experimenting with a computer-based simulation of a projectile fired from a cannon, for example, students can see how the path of a projectile is altered (or not altered) when they change such variables as the type of object, initial speed, mass, and diameter—something that would not be feasible in a classroom setting. By rotating a three-dimensional model of a molecule, students can get a better sense of its composition than they would from a two-dimensional representation.
Animations and simulations have considerable potential for helping students learn. They enable students to conduct experiments that would otherwise be impractical or impossible. They can slow down or speed up the time involved in a particular process and allow students to work with entities and phenomena that are too tiny or huge to observe in the real world. In these ways, they not only can help students develop competence with representations, but also can make sophisticated concepts easier to understand.
It is not a foregone conclusion, however, that animations and their kin enhance student learning or do a better job than does using other types of representations. Simply showing an animation or simulation is not sufficient; this may confuse students without drawing attention to such issues as context, assumptions behind the model, and key features to look for. For example, some research suggests that animations should be implemented in a way that encourages students to predict what will happen before they work through a simulation and to reason why certain outcomes occurred after they do it (Hegarty,
Kriz, and Cate, 2003). In addition, animations, visualizations, and related tools do not necessarily reduce the demands on students’ spatial ability; in fact, some research suggests that these tools may require greater spatial ability and visualization skills than static representations (Hegarty, 2011).
Implications for instruction
Improving students’ skills in using visual and mathematical representations is an important part of moving them toward greater expertise in science and engineering. An understanding of the research on students’ difficulties with representations and effective approaches for addressing them can inform efforts to design instruction. Some instructional issues to consider include the following:
- Instructors may need to provide students with more explicit introductions to the conventions that underlie the construction of various kinds of representations. Instructors might also highlight the relationships among alternative displays of similar information and explain how different types of representations are better suited to particular tasks.
- Providing extensive opportunities for students to practice interpreting and producing multiple types of representations can improve students’ performance, according to research from physics and chemistry.
- When instructors construct representations, they should consider how people naturally interpret symbols like circles or use conventions like left-to-right processing.
Biologists at Michigan State University have adopted a model-based instructional approach in several departmental courses. The strategies used in these courses engage all of the three main aspects of learning emphasized by DBER studies: (1) building conceptual understanding, (2) solving problems, and (3) interpreting, constructing, and understanding representations. Although the example in the following case focuses primarily on ecology, modeling will work well for any scale in biology, including the cellular level, and for other science and engineering disciplines.
Modeling in the Broadest Sense
At Michigan State, plant biology faculty have transformed their introductory courses to emphasize scientific practices. (This project also included graduate students and post-docs at Michigan State University, as well as faculty members at other institutions.) A goal of the practice-based approach to instruction is to help students conceptualize and reason about complex biological systems. Students in these courses construct, apply, and evaluate scientific models, including visual representations like graphs, diagrams, and box-and-arrow systems models. “When they are learning a topic, they are learning modeling in the broadest sense, via the practices of science” says Diane Ebert-May,a who along with Tammy Long was one of two Michigan State University plant biology faculty involved in this effort.
The authors of a paper about this course transformation effort point out that practice-based instruction is particularly well suited to the study of ecology, which emphasizes an understanding of systems and the interdependence of the elements that comprise them (Long et al., 2014). Specifically, the authors also point out that models are an authentic way of gaining insights into how students organize and connect knowledge.
A case study about the interaction of moose and gray wolves in Isle Royale National Park illustrates how students in an ecology course construct models as a way to understand the relationships among species in an ecosystem. In this park, moose are the primary diet for gray wolves, and the fitness of the wolves may be affected by congenital vertebral malformations. Students are given the following assignment:
Construct a box-and-arrow systems model that shows (a) the origin of genetic variations among wolves;

________________
a Interview, April 17, 2013.
(b) the relationship between genetic variation and phenotypic variation among wolves; (c) the consequences of phenotypic variation on wolves’ fitness. Include the following structures in your systems model but modify your language to make them specific to the case of the wolves’ vertebrae. You may use structures more than once and add additional structures not on the list.
Allele, gene, DNA, protein, phenotype, nucleotides, fitness
A representative student response is displayed in the figure below.
As another example, students in Ebert-May’s introductory biology course analyze graphs of current and projected patterns in atmospheric carbon during a unit on global climate change. Based on the data in these graphs, they determine what claims they can make about the projected consequences of global climate change. They are also asked what they can glean about the nature of science by analyzing graphs with seven different projections of future trends in carbon dioxide concentrations. “Our curriculum is based on three science practices: using data, modeling, and arguments,” says Ebert-May. “Everything is data driven.”
Faculty who teach the transformed courses note that “although many students struggle with using scientific models, particularly early in instruction, many report that over time, models help them to see ‘the big picture’ and how biological concepts are connected” (Long et al., 2014, p. 139). Models also provide a tangible way for students to assess and think critically about their own learning as they evaluate and revise their models to incorporate new knowledge.
A student-constructed model of the interactions of moose and gray wolves in the Isle Royale ecological system.
The research summarized in this chapter indicates that effective instructors start from an understanding of how students think and learn, both in general and in science and engineering disciplines in particular. They emphasize students’ prior knowledge and the role of students in constructing this knowledge instead of emphasizing the instructor’s ability to transmit knowledge. Many researchers and science and engineering instructors are putting these findings into practice in their own classrooms, in ways that can be adopted or adapted by others. More ideas for how to do this are described in Chapter 4.
Resources and Further Reading
Discipline-Based Education Research: Understanding and Improving Learning in Undergraduate Science and Engineering (National Research Council, 2012)
Chapter 4: Identifying and Improving Students’ Conceptual Understanding in Science and Engineering
Chapter 5: Problem Solving, Spatial Thinking, and the Use of Representations in Science and Engineering
Chapter 7: Some Emerging Areas of DBER
How People Learn: Brain, Mind, Experience, and School: Expanded Edition (National Research Council, 2000)




































