If the insights about learning in Chapter 3 have spurred you to think in new ways about improving your instruction, the question remains about which instructional strategies to try. This chapter describes a variety of practical strategies for instruction that are informed by evidence about how students learn best. The strategies discussed here are broader than those highlighted in Chapter 3 and are not discipline specific, although some instructors have given them a discipline-specific spin. The chapter first presents the kinds of student learning outcomes that these strategies are designed to produce. Next is a brief explanation of the characteristics of student-centered instruction, a pedagogical idea that undergirds the strategies in this chapter. The remainder of the chapter describes some of the most common research-based strategies for making lectures more interactive, promoting student collaboration and peer interactions, supplementing instruction with tutorials, and providing students with authentic experiences and instruction that engages them in the practices of science and engineering. Scattered throughout the chapter are examples of how skilled instructors have successfully implemented these strategies.
The strategies highlighted here are by no means the only research-based approaches for improving science and engineering education. Several more are described in Chapter 6 of the 2012 National Research Council (NRC) report on discipline-based education research (DBER), the source of much of the information in this chapter. In addition, information about tested and documented teaching methods can be obtained from professional organizations, resource centers, and other sources. The Pedagogy in Action Web portal of the Science Education Resource Center (SERC),1 for example, offers descriptions, course activities, and other information and materials for more than 50 such teaching strategies.
________________
When deciding on strategies to pursue, you might consider which ones will align well with the learning goals you established and the assessments you are using. You will also need to consider logistical issues such as class size, schedule, and classroom space, and your own teaching preferences and comfort level, among other factors. As the authors of How People Learn point out, “Asking which teaching technique is best is analogous to asking which tool is best—a hammer, a screwdriver, a knife, or pliers” (National Research Council, 2000, p. 22).
Goals of Research-Based Instruction
As mentioned in Chapter 2, decisions about how to teach should be based in large part on goals for what students should learn. These goals include general outcomes, derived from research on how students learn, that apply across science and engineering disciplines, as well as more specific goals for a particular course or program of study.
In engineering, the accreditation criteria for degree-granting programs developed by ABET, the accrediting agency, specify 11 general student learning outcomes. These outcomes address the specific knowledge and skills, as well as the more general habits of mind and professional conduct, that undergraduate students of engineering are expected to learn (ABET, 2009). (Chapter 7 of this book discusses the ABET criteria in more detail.) Many of these outcomes are best met through instructional methods that emphasize problem solving, communication, and teamwork, as well as other skills. Engineering instructors can use the program-level ABET criteria to guide their development of course-specific learning goals.
Across the science disciplines there is no consensus on a single set of learning goals for undergraduate education. Still, several general learning outcomes are largely consistent across the various fields of DBER. One useful list of general learning outcomes emerged from a series of workshops on promising practices held by the NRC (2011). Participants in these workshops identified these expectations for what undergraduate science, technology, engineering, and mathematics (STEM) students should eventually be able to do:
- Master a few major concepts well and in depth
- Retain what is learned over the long term
- Build a mental framework that serves as a foundation for future learning

- Develop visualization competence, including the ability to critique, interpret, construct, and connect with physical systems
- Develop skills (analytic and critical judgment) needed to use scientific information to make informed decisions
- Understand the nature of science
- Find satisfaction in engaging in real-world issues that require knowledge of science
The research-based instructional strategies presented in this chapter typically address several or most of these outcomes. You may need to combine more than one strategy to meet the specific learning goals you expect your students to achieve.
An Emphasis on Student-Centered Instruction
The chemistry curriculum developed by Vicente Talanquer and John Pollard, chemistry professors at the University of Arizona, encompasses several of the outcomes listed above from the NRC STEM workshop.
Shifting Instruction from What Chemists Know to How Chemists Think
An effort to redesign an undergraduate chemistry curriculum around findings about how students learn began as a conversation over lunch, says University of Arizona professor John Pollard.a Around 2006, Pollard was chatting with his Arizona chemistry colleague Vicente Talanquer, and they discovered they shared similar frustrations with the lack of depth and focus on breadth in the traditional undergraduate chemistry curriculum. Talanquerb had become dissatisfied with the lack of intellectual demand in undergraduate courses and wanted to shift the focus “from presenting what chemists know to illustrating how chemists think.” Both professors saw a need to revamp a curriculum that was “fact-based and encyclopedic … focused too much on abstract concepts and algorithmic problem solving, and detached from the practices, ways of thinking, and applications of both chemistry research and chemistry education research” (Talanquer and Pollard, 2010, p. 43).

Students work in groups using a computer simulation to investigate the properties of gases in John Pollard’s general chemistry class.
As a chemistry education researcher, Talanquer was already familiar with the literature on learning. He and Pollard began thinking about how to transform the curriculum and adapt the Process Oriented Guided Inquiry Learning (POGIL) approach, in which students work in small groups, to their own large introductory courses with enrollments of roughly 300 students. “We started, little by little, changing the emphasis of the curriculum; we created activities and pilot-tested them,” Talanquer explains. They obtained grants to implement their ideas from the National Science Foundation (NSF) and the State of Arizona and assessed the effects on student learning of the changes they were making.
The curriculum they developed, called Chemical Thinking (2012), is intended to “promote deeper conceptual understanding of a minimum core of fundamental ideas instead of superficial coverage of multiple topics” and to connect ideas from one topic to another through a progression of learning experiences (Talanquer and Pollard, 2010, p. 43). Students “learn how to approach realistic problems from a chemical perspective, using the powerful and
________________
a Interview, April 30, 2013.
b Interview, April 3, 2013.
productive models, techniques, and ways of thinking developed in the field” (p. 44).
Although Talanquer and Pollard still lecture for short periods, they have moved from presenting a video or a simulation of a chemical process to having students create their own simulations and models. Students often work in pairs on what Talanquer and Pollard call “Let’s Think” activities that require them to make observations, build models, make predictions and decisions, and construct explanations. Here is a Let’s Think activity from a unit on predicting the physical properties of chemical compounds using their molecular structure (Talanquer and Pollard, 2012):
Let’s Think—A Linear Molecule?
- Imagine for a second, that the water molecule was linear and not bent.
- How would this change the strength of the different contributions (i.e., dispersion, dipole-dipole, and H-bonding) to the IMFs [intermolecular forces] between water molecules?
- What would be the impact of this change on (a) the physical properties of water, (b) Earth’s climate, and (c) life in our planet?
- Share and discuss your ideas with a classmate.
At the end of every learning module (the equivalent of about one week), students are given an interesting problem to solve, called “Let’s Apply,” which helps the instructor and the students themselves determine whether they have achieved the main performance outcome for that module. Based on a review of students’ work, “we can get a sense of whether we need to emphasize something or go back and talk about other things,” says Talanquer. Here is a Let’s Apply task from the module on analyzing molecular structure:
Let’s Apply—Water Repellent?
Some textiles are frequently treated with organofluorines to make them more hydrophobic. A layer of the fluorine compound reduces the ability of the fibers to absorb water.

- Build a reasonable explanation to justify why some organofluorine compounds may reduce water absorption by textiles.
- Share and discuss your ideas with a classmate. Do not forget to clearly justify your reasoning.
Talanquer and Pollard also administer formal exams, which they have redesigned to align better with their instructional approaches. “All of our exams are topical,” says Talanquer. For example, the first unit of the course deals with how chemistry makes it possible to identify substances in one’s surroundings. One of the exams designed for this unit focused on the composition of Titan, a moon of Saturn. Students were given data about Titan and asked to answer

Students complete topical exams in which they are asked to integrate their knowledge and apply it to the analysis of a relevant system.
questions about the types of substances found on this moon’s surface and atmosphere.
Carly Schnoebelen,c who took Pollard’s course as a freshman in 2009–2010 and became an undergraduate preceptor in the course the next year, found the active learning approach “absolutely more effective” than the lecture-based science courses she took. “A lot of times in lecture students memorize something for a test and immediately forget it,” she says. “But if you’re more active, you understand and remember it better.”
Talanquer and Pollard are also evaluating the impact of their instructional approaches. On a traditional standardized exam that includes a heavy dose of algorithmic problem solving, their students perform the same as those in a traditionally taught class, but on an assessment of conceptual understanding, their students do significantly better (Talanquer, 2012). In addition, survey data show that considerably higher percentages of students taught with the redesigned curriculum find the course “challenging but rewarding,” report that they were interested to learn about the material in the course, express interest in doing a chemistry research project, and agree that the teaching approach in the course motivated them to learn (Talanquer, 2012). Furthermore, says Talanquer, students who took an introductory course with the redesigned curriculum get better grades in advanced chemistry courses than those taught with a traditional curriculum. “That has become a powerful tool for changing people’s minds,” he adds.
________________
c Interview, April 25, 2013.
Most of the strategies described in this chapter, like those used by Talanquer and Pollard, are student-centered. Student-centered approaches place less emphasis on the instructor transmitting factual information by lecturing and more emphasis on students building their own understanding with careful structuring and guidance from the instructor. As noted in the 2012 NRC report on DBER, these approaches include these characteristics drawn from evidence about how people learn:
- More time spent engaging students in active learning during class
- Frequent formative assessment of students’ levels of conceptual understanding
- Attention to students’ metacognitive strategies
In this type of instruction, students are expected to be actively and cognitively engaged in their coursework. Rather than being told what they should learn, students often do activities designed to help them discover key concepts or draw their own conclusions before the instructor explains a concept. In addition, students may be expected to acquire at least some of the basic knowledge for the course by doing assignments outside of class so they can come to class ready to apply and build on that knowledge.
Instructors still play several vital roles in student-centered classrooms. Instructors carefully design activities to support students in developing the desired knowledge and skills. They structure the activities to provide “scaffolding” during the learning process that builds on what students know and challenges them to gradually work beyond their current level. Instructors also assess students’ prior understanding and frequently check how students’ knowledge is progressing throughout the course. And while students are doing activities, the instructor observes, listens, asks questions, and guides students when they get off track. An understanding of research on learning can help with all of these roles.
Active learning is a critical element of virtually all of the research-based instructional strategies described in this chapter. Active learning engages students in answering and asking questions, thinking about and solving problems, and explaining and reflecting on what they are learning instead of just sitting and listening.
Some student-centered approaches differ radically from traditional lecture-based instruction. Observers of a classroom that uses formal cooperative learning, such as the problem-based learning approach or the Student-Centered Active Learning Environment with Upside-down Pedagogies (SCALE-UP) model, discussed later in this chapter and in Chapter 5, may wonder where the lecture went. Students work in small groups throughout most of the class period, tackle challenging and
authentic problems, or do hands-on activities. The instructor circulates among the student groups, providing guidance and pausing the action only briefly to explain something to the whole class.
The “flipped classroom” approach, a term much in vogue, describes instructional designs in which students are responsible for learning most of the basic course content through out-of-class reading, research, or pre-recorded video lectures in order to free up class time for problem solving and other substantive activities. While a flipped classroom approach can be a dramatic departure from traditional instruction, this depends on how it is implemented. Flipped classroom models vary in the extent to which they use student-centered approaches or rely on lectures (albeit, prerecorded) to convey information.
In any case, you can implement active learning without devoting the entire class to cooperative work, or flipping the classroom, or eliminating your lectures. Less sweeping approaches can be effective, as explained in the next section.
Making Lectures More Interactive
DBER provides a strong base of evidence in multiple disciplines of the value of making lectures more interactive. Several research-based strategies intersperse lecture with shorter student-centered activities, such as interactive exercises or student discussions about probing questions intended to spur deeper thinking. Karl Smith (2000) has employed an approach in which he organizes a class by alternating between short, 10- to 12-minute lectures and even briefer, 3- to 4-minute opportunities for student discussion, “bookending” these activities with an introduction and summary. This is just one example. Other research-based methods, discussed below, use varying amounts of lecture and different types of interactive exercises.
Think-Pair-Share
Think-Pair-Share is an informal type of collaborative learning that can be implemented relatively easily in a lecture or a lab of any size in a fairly short time. This strategy engages every student in talking out loud about her or his ideas but requires less formal monitoring from the instructor than is recommended when students work in groups for longer periods.
The idea of Think-Pair-Share is an old one (Lyman, 1981), and its procedures have been refined over the years (Johnson, Johnson, and Smith, 1991). The instructor follows these basic steps (Allen and Tanner, 2002):
- Pose a question during class, often one with many possible answers.
- Give all students one or two minutes to THINK about their individual answer by having them jot down their ideas on a piece of paper.
- Give all students a chance to discuss their answer and ideas with a neighbor in a PAIR or a small group. (This may take a few to several minutes depending on the complexity of the question.)
- Invite pairs to SHARE what they discussed with the whole class.
- Wrap up with a summary that emphasizes the main learning points.
The following examples illustrate how Think-Pair-Share questions can vary in topic, complexity, length of time required to answer, and presentation format:
Biology: Watch the video clip about the Human Genome Project and the designer baby. Work in pairs to answer the following questions: (1) What is the basis of making a designer baby? Think about the biology of DNA, genes, and traits. (2) What are the advantages and disadvantages of having a designer baby? (3) Do you agree or disagree with the idea of a designer baby? Why? (Chen and Ray, n.d.)
Geosciences: Look at this graph of satellite measurements of ozone concentration above Antarctica from 1979–1992. Put yourself in the mind of a scientist seeing this data for the first time. What complexities, patterns, or trends would be important to develop a theory for how this pattern has been generated? (Hancock, 2010)
Astronomy: Rigel is much more luminous than Sirius B. Rigel and Sirius B have the same temperature. Which star has the greatest surface area?: (a) Rigel, (b) Sirius B, (c) They have the same surface area, or (d) There is insufficient information to answer this question. (Forestell et al., 2008)
While students are discussing the question(s), the instructor can walk around the room and listen in on their conversations. This enables the instructor to assess what students do and do not yet understand, identify misconceptions, and note how students explain ideas to their peers. After the pairs’ discussion, the instructor can acknowledge the good ideas heard and perhaps invite students who used insightful ways of explaining a concept or correcting a misconception to share their thoughts with the whole class.
The use of Think-Pair-Share and similar types of informal groupings has been associated with a variety of desirable outcomes, including improved achievement, critical thinking, and higher-level reasoning; better understanding of others’ perspectives; and positive attitudes about their fellow students, instructors, and the subject matter at hand (Johnson, Johnson, and Smith, 2007).
Peer Instruction and ConcepTests
If you have ever given a traditional lecture, you have probably experienced that moment when you scan the rows of students—some listening attentively, some taking notes robotically, others yawning, whispering, or surreptitiously texting—and wonder, How much of this are they really getting? If you try to check students’ comprehension by asking a question (hopefully something more pointed than the generic Any questions?), a motivated few will respond. As for the rest, you still do not know how well they understand.
One of the most influential, widely used, and time-tested strategies for making lectures more interactive builds on Think-Pair-Share but adds a more formal process for using carefully crafted questions to shed light on students’ conceptual understanding. That strategy is Peer Instruction, developed by Harvard University physics professor Eric Mazur. As explained in Chapter 1, Mazur was motivated to change his teaching after recognizing that many of his students did not truly understand core concepts in physics.
In Peer Instruction, the instructor gives a short presentation focused on a particular topic and then asks all students in the class a multiple-choice question, or ConcepTest, designed to reveal common student misunderstandings about a central concept related to that topic. Students are given one minute to arrive at an individual answer and report their responses to the instructor; often students respond with clickers, but this can also be done with colored cards or other means. For the next few minutes, as the instructor circulates and listens, students discuss their answers and their reasoning with their peers in adjacent seats and try to convince each other why their answer is correct. At the end of the peer discussions, students are again polled for their answers, which often have changed based on the discussion. The instructor reviews the correct answer and addresses questions that arose during discussion, and then moves on to the next topic (Crouch and Mazur, 2001). To make time for the ConcepTests, students are expected to complete a reading assignment on the topic covered before class.
Since first introducing Peer Instruction, Mazur and his colleague Catherine Crouch have taken ideas from different sources to refine the approach. For example,
before students come to class, they must do warm-up exercises in the form of Web-based, open-ended questions about their reading assignments. Tutorials (explained later in this chapter) and group problem-solving activities have been incorporated into a weekly discussion section that supplements the course. Evaluations of Peer Instruction show improvements in students’ understanding of the course material as measured by their performance on the ConcepTests and on two standardized tests: the Force Concept Inventory and the Mechanics Baseline Tests (Crouch and Mazur, 2001).
For many instructors, one of the most appealing aspects of Peer Instruction is that it can be done with large courses in a lecture auditorium. “This is a great way for people to add an interactive component in inadequate spaces,” says Mazur.2 “Not many people may have the opportunity to redesign both the space and the course.” The case study in Chapter 2 of David McConnell’s geosciences course at North Carolina State University describes an effective way to implement ConcepTests with peer interaction in a large enrollment course; hundreds of ConcepTests in geosciences are available online (Science Education Resource Center, n.d.). Peer Instruction has been adapted in biology (Knight and Wood, 2005), chemistry, and other disciplines, and in a range of instructional settings and class sizes. Peer Instruction is also an attractive option because it can be incorporated into a standard lecture.
It is important, however, that any adaptations maintain the crucial element of peer interaction. Simply asking students to respond to clicker questions without this element loses the benefits of having students articulate a rationale for their answer, listen to their peers’ reasoning, and try to reach consensus. “At times, it seems that students are able to explain concepts to one another more effectively than are their teachers,” writes Mazur (1997, p. 13). This is likely because students who understand the concept “have only recently mastered the idea and are still aware of the difficulties involved in grasping that concept. Consequently, they know precisely what to emphasize in their explanations.”
Designing good ConcepTest questions takes some thought. Crouch and Mazur (2001) recommend that the questions focus on important concepts, include incorrect answers based on common student misunderstandings, and be challenging enough that between 35 percent and 70 percent of the students answer correctly before the peer discussion (Mazur, 1997). Although multiple-choice questions are the easiest type to administer in a large class, ConcepTests can also take the form of open-ended questions or quantitative problems, or the students themselves can generate the answer choices that the entire class then votes on.
________________
2 Interview, April 13, 2013.
Here is an example of a ConcepTest question from Mazur (1997):
A SAMPLE CONCEPTEST QUESTION ON ARCHIMEDES’ PRINCIPLE OF BUOYANCY
Imagine holding two identical bricks under water. Brick A is just beneath the surface of the water, while brick B is at a greater depth. The force needed to hold brick B in place is:
- larger than
- the same as
- smaller than
the force required to hold brick A in place.
Correct answer: 2
Beth Simon,3 a computer science and engineering instructor at the University of California, San Diego, organizes her 80-minute classes around a series of four to six multiple-choice clicker questions, administered using Mazur’s model of individual answers, peer discussion, and a final vote. Students are expected to learn basic content before they come to class through reading assignments and prerecorded lectures, and they take a quiz on this homework at the beginning of each class. Designing clicker questions that target potential difficulties is a critical part of the instructor’s role. “It’s kind of like being a sports coach. If you hire a tennis coach for your kid, you don’t want them to just assign the kids to play a game of tennis. Instead, they have them do little drills with particular things that they know are problematic. I think of my clicker questions as the exact same thing,” says Simon.
“People say, where’s the lecture? I do have some ‘lectures,’ but they generally come in response to questions,” Simon explains. After completing a clicker question, for example, Simon might show a slide summarizing the most important things that students were expected to learn from that question. Or she might go into a deeper explanation of a topic that was not well explained in the textbook or do a demonstration of a process the students just studied.
________________
3 Interview, August 20, 2013.
Ongoing adjustments to lectures based on formative feedback
Some instructors adjust the content of their lectures based on the homework that students submit before class or on formative assessments they do in class. These strategies are informed by findings from research about the need for instruction to build on students’ prior knowledge.
In Just-in-Time Teaching (JiTT), the most familiar of these methods, students do homework in the form of written responses to questions about reading assignments or solutions to problems, and then they submit their work on deadline via the Web, in time for the instructor to review it before the next class. The instructor can then modify the next lecture to clarify common misunderstandings or accommodate students’ interests (Novak, 1999). A moderate amount of evidence suggests that JiTT is effective in teaching some physics concepts (Formica, Easley, and Spraker, 2010), is associated with positive attitudes about introductory geology (for example, Luo, 2008), and improves students’ preparation for class and study habits in biology (Marrs and Novak, 2004). In its emphasis on learning certain material before class, JiTT might be seen as a precursor to the flipped classroom model.
In an approach called Just-in-Time-Teaching with Interactive Learning (JiTTIL), Stephen Krause and his engineering colleagues at Arizona State University and elsewhere have expanded on this basic idea by adding interactive classroom exercises and various forms of fast and frequent feedback (Krause, Kelly, and Baker, 2012b). In Krause’s classes that use the JiTTIL approach, students spend much of the class time working in small groups at round tables on activities based on real-world situations. In one such activity, students select from a list of options the best materials to use to construct various components of a bicycle, based on their understanding of the properties desired in a particular component. To improve students’ ability to transfer their learning to new contexts, certain activities include an “extension” question that challenges students to apply what they just learned to a new situation. In the bicycle example, the extension activity asks students to come up with substitute materials other than the ones listed for each bicycle component.
The Materials Concept Inventory, a standardized assessment in engineering developed by Krause and his Arizona State colleagues, provides information about students’ conceptual knowledge at the beginning of a course. Day-to-day feedback comes from the “muddiest point” reflections described in Chapter 3 and from homework problems that emphasize the key concepts for the topic being studied. Immediate feedback comes from in-class ConcepTests, administered through the

Students work in small groups to tackle real-world problems in an activity aimed at applying and extending newly acquired knowledge.
Web-based Concept Warehouse tool, and this information can be used to make on-the-spot adjustments in instruction.
These different types of formative assessments reflect the fact that students’ prior knowledge changes as they progress through a course. “When you first walk into a class, it’s your prior academic experience and life experience. But then after you’ve been in the class for a while, it’s all the knowledge you’ve acquired in class,” says Krause.4 “Then there’s the previous day’s prior knowledge, which is critical, especially if there’s a progression of three to five classes for a particular topic.”
Evaluations of students taught with the JiTTIL pedagogy show greater gains in conceptual understanding than those in traditionally taught classes, as measured by the Materials Concept Inventory. The percentage of students who persist in the
________________
4 Interview, July 9, 2013.
course after the second week has also increased from 85 percent to 95 percent in the JiTTIL classes, and the withdrawal rate of female students has decreased from 40 percent to 10 percent (Krause, Kelly, and Baker, 2012b).
The most recent iteration of this pedagogy developed by Krause and other colleagues uses an even wider range of cyber-enabled tools to generate rapid and frequent feedback about student learning and includes more active learning exercises (Krause et al., 2013b).
Alternating lectures with interactive exercises
Although a few students may engage in meaningful conceptual reorganization during a lecture, the lecture mode of instruction is a fundamentally passive one. Interactive approaches are much more effective at promoting the mental processes involved in constructing new knowledge. Recognizing this, many skilled instructors have progressed from lecturing for the whole class period to alternating shorter lecture segments with various types of exercises that promote student interaction.
Clicker questions and other forms of ConcepTests are one way to do this, as are the Interactive Lecture Demonstrations described in the Sokoloff example in Chapter 3. Some instructors integrate group problem solving, “mini-labs,” or other types of hands-on activities into their lectures. Often students work collaboratively on these activities, a form of instruction explored later in this chapter. According to a study of an approach to teaching introductory geology that alternated lectures with mini-lab activities, students who did a mini-lab with petrified wood and discussed their observations in online discussion groups showed statistically greater gains than did students in two control groups (Clary and Wandersee, 2007).
Mark Leckie,5 a geosciences professor at the University of Massachusetts Amherst, describes his 75-minute oceanography classes as “part lecture, part student active learning.” Every class includes a 15- to 20-minute collaborative activity, based on a set of exercises he developed with his colleague Richard Yuretich. (See Chapter 1 for examples of their exercises.) “In-class activities can be effectively used to ‘set the hook,’ to get students interested in what you have to teach, to challenge misconceptions, to initiate discussion of a new topic, to provide reinforcement of material presented in your lecture, to assess student understanding, or to practice critical thinking and problem-solving skills,” writes Leckie (n.d.).
________________
5 Interview, March 22, 2013.
Student-to-Student Interaction
Drawing from research on the benefits of learning through social interactions, many forms of student-centered instruction—including some described above—incorporate activities in which students work together in groups. In well-designed activities of this type, students learn from interacting with each other; they build on one another’s knowledge, ask questions and provide explanations, and come up with ideas that might not occur to an individual working alone. These activities can also help students develop the kinds of teamwork, decision-making, and interpersonal skills that are integral to the practice of science and engineering. These elements require students, both individually and collectively, to engage in the kind of thinking that leads to deep learning, to articulate their thinking, and to reflect on feedback from others in a way that causes them to reexamine and extend their thinking. Thus, while the social aspects of working in groups are important, the cognitive aspects of this process are also important.
Cooperative learning is a highly structured approach to learning through student-to-student interactions with a strong base of evidence of its effectiveness (see, for example, Smith, 2011). In a well-structured cooperative learning environment, students work together in small groups to accomplish a common goal. The activities foster interdependence by requiring the cooperation of all members of the group and hold both the individuals and the collective group accountable for successfully completing the work.
Collaborative learning is a more generic form of learning through peer interactions. Collaborative learning assumes that students learn best by constructing knowledge within a social context (see Chapter 3) and encourages students to coalesce into a “learning community.” While collaborative learning, like cooperative learning, is intended to foster interdependence among students, it is less structured than cooperative learning and does not necessarily combine individual and collective accountability (Smith, 2011).
Research has shown that activities in which students collaborate with each other can be more effective than traditional instruction and can improve students’ retention of content knowledge, according to several studies referenced in Chapter 6 of the 2012 NRC report on DBER and other literature. But results depend greatly on the care with which these activities are implemented. Collaborative approaches are not inherently effective and can be poorly implemented. The evidence is not conclusive about the conditions under which these strategies work.
Group activities often involve answering questions, solving problems, or conducting investigations. These activities can be as brief as 10 or 15 minutes or can last an entire class period or even longer. They can be integrated into a lecture format or can serve as the predominant form of instruction. While many instructors assert that they cannot do collaborative activities because their classes are too large, the experiences of expert practitioners prove otherwise. In large classes, the groups typically consist of students seated next to each other. In smaller classes, instructors use a variety of options to group students. Some assign students to groups to achieve a mix of personalities, abilities, or other characteristics, while other instructors let students form their own groups. Some instructors switch the group composition periodically to ensure that students get to know different people, while others keep the same groups to encourage bonding.
Grading group activities is tricky. Individual grades are critical to ensure that each student learns; in addition, Smith suggests that basing a small portion of a student’s grade on group performance can be beneficial if done carefully (Smith, 1998).
Some common approaches to such student-to-student interaction as cooperative learning and collaborative learning are described below.
The jigsaw technique
In the “cooperative jigsaw” approach first described by Aronson and colleagues (1978) and refined later by other practitioners, each student member of a cooperative learning group “is responsible for learning a portion of the material and conscientiously teaching it to the rest of the group” (Smith, 2000, p. 32). The instructor chooses the material, structures the groups, provides guidance about student roles, monitors their functioning, and helps students summarize, synthesize, and integrate the material. Faculty who have used this technique find that it can foster students’ interdependence and help them learn conceptual material. “Although it takes preparation and time to set up the jigsaw, students usually learn more material and remember it longer,” Smith concludes (p. 32).
Barbara Tewksbury, a Hamilton College professor, often uses the jigsaw technique in her geosciences classes. Over the years, this technique has been the most popular cooperative learning strategy among participants in the On the Cutting Edge workshops in course design offered by Tewksbury and colleagues. As the example below demonstrates, the jigsaw technique emphasizes both individual accountability and achievement of group goals.
Putting Together the Pieces of a Geosciences Puzzle
In the early 1990s, Hamilton College professor Barbara Tewksburya was considering ways to place more responsibility on students for learning the content in her geosciences courses. She came up with a plan to have individual teams learn a portion of the material about a particular topic and then reconfigure into new groups and share what they had learned—an approach she later realized had already been developed by others and dubbed the jigsaw technique. “I had no idea it had been formally invented and named in 1978 by Aronson,” she says. “But it was one of these situations where I had a pedagogical problem to solve and thought, ah, this would be a good way to solve it. And it turned out I wasn’t the only one who had done that,” she says, adding that this “Lone Ranger” mentality was typical of geosciences faculty at that time, before geosciences education research had coalesced as an area of scholarship. (Today, she adds, instructors have no need to reinvent the wheel with all of the resources available.)
In Tewksbury’s course on geology and human events in North Africa and the Middle East, one of the topics students study is the impact of climate changes on the development of Egyptian civilization around 3000 B.C. She uses the jigsaw technique as a way for students to analyze geological data on sediment accumulation in the basins of the Sahara in order to determine prehistoric changes in rainfall.
To prepare for the in-class jigsaw, students do homework assignments that help them understand the extremely arid climate of the modern Sahara. These assignments include looking at Google images of the Sahara. Students also read historical accounts of traverses across the Sahara, a report from a British army expedition trapped in the desert, and descriptions of the modern salt mines operating in Mali.

The White Desert near Farafra Oasis, Egypt.
When the students arrive in class, they are divided into four teams of roughly four people per team. Every team receives a set of published data on the sediments in a particular paleolake; each of the four lakes is located in a different part of the Sahara and has a distinctive stratigraphic record. Their task as a team is to analyze the sediment record and figure
________________
a Except where noted, the information in this case study comes from an interview with Barbara Tewksbury, March 28, 2013.
out when the lake first appeared, when it dried up, and how the rainfall changed over thousands of years. “By the time the team is done analyzing, everybody has to be prepared to tell somebody else what they learned about their particular data. They have to be able to paint a picture—here’s what I think is going on, and here’s the evidence,” Tewksbury explains.
At that point, she creates new groups composed of a member from each of the former teams. Each member of the new group shares the findings from the previous team’s analysis of one particular lake. “They each teach each other what their data suggest,” says Tewksbury. The new groups then do a two-part assignment: first, to determine what they can say about the timing and amount of rainfall changes in the Sahara as a whole based on sediment data from four lakes; and second, to analyze additional data about worldwide climate change and predict what will happen in the Sahara with future global climate change. “Of course, their intuition is that if it warms up, it’s going to get drier, but the geologic record shows just the opposite: when it gets hotter, it rains more in the Sahara. So it ties back into the modern world,” she says. In later class meetings, the students study other aspects of the impact of climate and geological events in order to understand why Egyptian civilization developed when it did.
“It’s called a jigsaw because everybody has a piece of the puzzle,” Tewksbury explains. “Each team has one stratigraphic column, so that means that four people in that team have the same jigsaw puzzle piece.” When the groups are reconfigured, all four puzzle pieces are represented by each new member contributing one piece. As the students teach the members of their new group about what they learned, “that’s equivalent to putting the piece down on the table.” The final group assignment—to look across all four pieces and arrive at conclusions about Sahara rainfall trends and their implications for global climate change—“puts the whole picture together.” To instill individual accountability, each student must do a major analysis of the timing of the rise of Egyptian civilization that incorporates the earlier paleolakes analysis and other data.

This approach of having students reach their own conclusions based on data provides them with a much better learning experience than if she were to lecture on the topic, says Tewksbury. “They’re perfectly capable of drawing the conclusions themselves.”
It took Tewksbury time and professional development to arrive at an understanding of effective strategies for learning. Early in her career, she taught as she had been taught—by lecturing. “I had never heard of the word pedagogy,” she says. Her “transformative” moment came in the late 1980s, she says, when she became involved as a content expert in a workshop on active learning and other strategies for middle and high school teachers. “I think I probably learned more about pedagogy and had my eyes opened a lot wider by what I learned from the Earth science teachers than they did from me…. That got me thinking for the very first time about what I was trying to accomplish in my classes,” she says.
This example of a jigsaw in a geosciences class includes several elements associated with improvements in student motivation and learning:
- Students actively construct conceptual knowledge by analyzing data, seeing how it fits into a larger framework, and reaching conclusions.
- Students take control of their learning by doing advance reading, studying a variety of information, and explaining what they learned to their peers.
- The interactive element provides students with an incentive to do the work so they will not let down their peers or themselves.
- Students use the tools and practices of geology, much as a professional scientist would, by analyzing maps and data.
- The activity has relevance to the real-world problem of global climate change.
Natalie Yeo,6 who was a student at the Community College of Rhode Island in 2013, participated in jigsaws and other cooperative learning approaches in a geology class with professor Karen Kortz. She describes the jigsaw groups as “kind of a mini–think tank because you’re all trying to bolster the same idea.” The variety of small group work in Kortz’s classes keeps students constantly engaged, she says. “Sometimes I leave a lab and say, ‘What a think marathon—my brain hurts, in a good way.’”
Group work on problems, experiments, and projects
In other types of cooperative or collaborative learning, students work in groups to solve problems, conduct investigations or experiments, or carry out projects. Some of these strategies involve more extensive changes in classroom organization and instructional practices than a short activity incorporated into a lecture would.
Process Oriented Guided Inquiry Learning (POGIL) began in college chemistry departments but has since been adopted by instructors in a variety of disciplines in numerous postsecondary institutions and high schools. Students in a POGIL classroom learn through a guided inquiry process in which they are presented with data or information, followed by leading questions designed to guide them in formulating their own conclusions (Process Oriented Guided Inquiry Learning, 2014a). Students work in small groups as the instructor observes and facilitates, stepping in as needed to address questions and provide guidance.
________________
6 Interview, April 10, 2013.
The following high school POGIL activity shows how the guided inquiry process works (Process Oriented Guided Inquiry Learning, 2014b):
LEARNING ABOUT ENZYMES THROUGH GUIDED INQUIRY
In a biology classroom that uses the PoGIL “guided inquiry” approach, students explore this question: What are the factors that regulate the rate at which enzymes catalyze reactions? Students read a paragraph explaining how enzymes affect their daily lives and why the preceding question matters. In the specific activity, students look at the optimal conditions for two different enzymes: lipase, which breaks down lipids in the small intestine, and pepsin, which breaks down proteins in the stomach. They are presented with an initial model consisting of (a) a simple diagram showing that lipase breaks down triglycerides into glycerol and fatty acids and that pepsin breaks down large polypeptides into smaller polypeptides and amino acids, and (b) a graph of the effect of ph on enzyme activity. Students study the model and compare data in order to answer a series of questions of increasing complexity, including questions that require them to explain an answer in detail, justify their reasoning, and add a line to the graph. The final question in the series requires them to use their knowledge of protein structure to explain in detail the effect of exposing an enzyme to a pH outside of its optimal range. Include the effect on both enzyme structure and function.
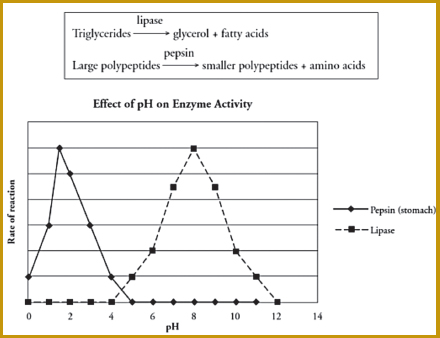
Model and graph provided to students in a POGIL guided inquiry on enzymes.
SOURCE: Flinn Scientific, Inc., 2014.
This activity continues with a second model consisting of three graphs showing the effects of various factors on the function of the enzyme amylase in the body. Students work through another series of questions about that model. The activity also includes “extension questions,” which require deeper thinking and might be used for homework or as an extra activity for groups that move quickly.
Within the basic POGIL framework, instructors use a variety of strategies to manage the groups and help students learn. Instructors might suspend the group work at various points to allow groups to report their answers to the whole class, and activities generally conclude with a whole-class discussion. At times, instructors may need to give a mini-lecture to the whole class or to an individual group to clarify an area of confusion or reinforce a vital point. Instructors may also set aside time for students to reflect on their learning and process skills. The strategies used will depend on what transpires during each class. “The principle is to have students do as much thinking and working as possible. No day is a typical day,” says Rick Moog, a Franklin & Marshall College chemistry professor and executive director of The POGIL Project. Formative assessment is a central part of the POGIL process, as explained in Chapter 5.
Although POGIL has been used most extensively in smaller classes, it can also be adapted for large classes—for example, by framing a recitation section around POGIL activities or having students use clickers to give feedback to provide responses to questions based on POGIL activities (Amaral et al., 2005). Numerous resources in a range of disciplines—curriculum, instructional guidance, professional development workshops, and more—are available for instructors who want to use the POGIL pedagogy (see www.pogil.org).
Studies of the effectiveness of POGIL at a range of institutions and for a variety of courses provide evidence that this approach can improve achievement and reduce attrition as compared with courses taught through traditional methods (for lists of specific studies, see https://pogil.org/about/effectiveness). Other research has found improvements in test scores for general chemistry students when POGIL is combined with Peer-Led Team Learning, the approach described in Chapter 1 in which trained undergraduates lead supplemental problem-solving sessions (Lewis and Lewis, 2005).
Cooperative problem solving, an instructional approach that draws from research on cooperative learning, was originally developed for physics as a way to encourage students to develop expertise in problem solving while discouraging their tendency to use novice strategies (Heller and Heller, 2010). Working in small groups, students use a specially designed strategy to solve context-rich problems. These are problems rooted in real situations that would be difficult to solve by applying a few equations and plugging in numbers, seeking the recognizable patterns found in many traditional textbook problems, or looking for physics vocabulary cues. Instead, these problems are best solved by analyzing the situation, perhaps with the help of a visual representation, and logically constructing a path to
a solution. These problems often require students to consider what they know and do not know and what assumptions they need to make.
Here is an example of a context-rich problem in one-dimensional kinematics:
You have a summer job as the technical assistant to the director of an adventure movie. The script calls for a large package to be dropped onto the bed of a fast moving pick-up truck from a helicopter that is hovering above the road. The helicopter is 235 feet above the road, and the bed of the truck is 3 feet above the road. The truck is traveling down the road at 40 miles/hour. You must determine when to tell the helicopter to drop the package so it lands in the truck. (Heller and Heller, 2010, p. 242)
A typical 50-minute class organized around cooperative problem solving might look something like this: The instructor begins by articulating the concepts to be learned during that class session. For the next 35 minutes, students work in groups to solve the problem as the instructor observes and listens, diagnosing students’ difficulties and occasionally interacting with groups that need help. At the end of the appointed time, one member from each group is randomly selected to put part of their solution on the board, with license to ask other members of the group for help. Students in other groups examine the solutions on the board to see how they parallel or differ from their own group’s result. The instructor then leads a class discussion of the possible solutions but does not give students a paper with the complete correct solution until the end of class (Heller and Heller, 2010, pp. 1–2).
Cooperative problem solving can be used as the major focus of a course, as in the example above, or to make smaller recitation or discussion sections more interactive.
Findings from a study in chemistry also indicated that cooperative problem solving improved students’ problem-solving abilities by about 10 percent, and that this improvement was retained when students returned to individual problem-solving activities (Cooper et al., 2008).
In problem-based learning (PBL), students learn disciplinary knowledge by working through problems that mirror real-world situations, most commonly in groups but occasionally individually. At the core of PBL is the notion of the “ill-structured” question, which reflects the complex, messy, and tentative nature of many of the problems facing science and society that have no simple, formulaic “right” solutions. Groups are presented with contextual situations and asked to define the problem, decide what skills and resources are necessary to investigate
the problem, and then pose possible solutions (Duch, Groh, and Allen, 2001). To solve this type of ill-structured problem, students must engage in inquiry, information-gathering, and reflection. PBL activities can take up most of the teaching and learning time in a classroom or can be combined with a lecture.
“Strategies used will depend on what transpires during each class. The principle is to have students do as much thinking and working as possible. No day is a typical day.”
—Rick Moog,
Franklin & Marshall College
Although the most striking thing about the SCALE-UP (Student-Centered Active Learning Environment with Upside-down Pedagogies) model is its redesign of the physical classroom space (described in Chapter 5), it is also a powerful example of how student collaboration can serve as the primary mode of instruction. In a SCALE-UP classroom, the lecture and lab components of a course are combined. Students learn some of the basic content through readings and homework before and after class. This arrangement makes it possible even for students in very large enrollment courses to spend most of their class time working in small groups on rich activities.
The main learning occurs through interactions with peers under the guidance of the instructor and through classroom discussions among the students and with the instructor. Students do hands-on activities, problems, simulations, and experiments that challenge them to think deeply. Brief intervals of lecture are mostly limited to providing motivation and linking the collaborative activities to the bigger course content.
As one type of SCALE-UP activity, students do 10- to 15-minute experiments in groups using a “predict-observe-explain” model. For example, groups are given the task of rolling a racquetball through a curved path between a pair of concentric quarter-circle arcs drawn on a piece of paper. Some students tip the paper or spin or blow on the ball. The instructor asks them why they need to do this (making references to Newton’s second law), with the goal of getting them to understand they are applying a force to the ball to change the direction of its motion. Once they understand, they are asked to specify the direction of the force. Through Socratic dialogue, the instructor eventually gets students to see that the force is always directed toward the center of the concentric arcs. Students recognize this as a centripetal force and then do a task in which they approximate the magnitude of the force from the mass of the ball and estimate its speed (Beichner et al., 2007).
Other SCALE-UP activities require students to think through a problem without making measurements. An example: How far does a bowling ball travel down the lane before it stops skidding and is only rolling? This difficult problem requires a great deal of estimation, but it provides students with insights about frictional force. When they have developed an answer, they can check it against a simulation of this scenario.
According to Robin Wright,7 who teaches a SCALE-UP biology course at the University of Minnesota, an overarching theme of this approach to teaching is “to have students doing biology, not learning about biology, and to have that ‘doing’ happen in the classroom where we can watch and help and coach them.”
Studies of the SCALE-UP model have found that participating students have better scores on problem-solving exams and ConcepTests, slightly better attitudes about science, and less attrition than students in traditional courses (Beichner et al., 2007; Gaffney et al., 2008). Failure rates among women and students from underrepresented groups have also been substantially reduced.
Mackenzie Tilley,8 an engineering major at North Carolina State who took Robert Beichner’s SCALE-UP physics course, benefited from this type of instruction in several ways. “What’s great about the group work is that if you have a question, you don’t have to wait for the professor to finish talking,” she says. “You just turn to your partner and say, hey, how did you figure this out? Most of the time your partner will understand what you don’t understand, or you’ll understand when your partner or group members don’t.” In addition, she says, “the activities all worked in a way that clearly showed you the differences between certain principles…. You could distinguish what was going on and why it was going on that way.”
SCALE-UP is just one example of an approach that combines the lecture and lab sessions of a course into a single, longer block of time to facilitate group investigations and projects. Some of these approaches include laboratory experiences aligned with scientific practices in which, for example, students record observations, develop and test explanations, refine existing models, and build and refine their own models of a scientific process by experimenting.
Another such approach is Modeling Instruction, developed by Eric Brewe and colleagues in the physics department at Florida International University.
________________
7 Interview, April 12, 2013.
8 Interview, March 27, 2013.
Florida International University (FIU), a large, public research university with a majority Hispanic enrollment, graduates more Hispanic holders of bachelor’s degrees than any other institution in the United States. In an effort to increase the participation of Hispanic students in physics courses and improve learning for all students, Eric Brewe and other faculty members in the physics education research group at FIU have designed and implemented a version of Modeling Instruction, an approach first explored by David Hestenes (1987) at Arizona State for whom Brewe was a graduate assistant. The Modeling Instruction approach used by Brewe and his colleagues combines active student engagement, cooperative grouping, activities to foster conceptual understanding, and experience with the authentic practices of the discipline—features that can support the learning of students underrepresented in science, according to participating faculty (Brewe, Kramer, and O’Brien, 2009).

Eric Brewe engages in model building with his class at FIU.
In the physics classes taught with the modeling method, students construct and validate models that allow them to explain, describe, and predict outcomes, much as scientists do. “If we think science is about building models, then we have to focus attention on getting students into authentic practices,” says Eric Brewe,a who was instrumental in designing the modeling curriculum. The “cookbook” method of doing lab experiments fails to help students see the connection between their experiment and the theory behind it, he asserts. Instead of thinking about whether their approach to solving a problem is a useful model, students fixate on whether they arrived at the right answer. In Modeling Instruction, explains Brewe, “students think about what goes into a model…. We’re careful to get students to understand how to use representations to make predictions or draw
________________
a Except where noted, the information in this case study comes from an interview with Eric Brewe, April 16, 2013.
conclusions. A graph is only as good as the information you can draw out of it.”
During his classes, which meet three times a week for two hours at a time, Modeling Instruction students work in small groups in a studio-format classroom with whiteboards and tables instead of desks. The longer class period has made it possible to integrate the laboratory and lecture components of the course, which mirrors the practices of scientists. “No reasonable scientist says, ‘It’s our day to do lab’—they do investigations when it’s appropriate,” says Brewe. The activities and lab experiments are designed to encourage model building. The groups share their ideas on portable whiteboards and come together for discussions, with the instructor moderating.
Stephanie Castañeda, a student who took two semesters of Brewe’s course in 2012–2013, recalls a kinematics experiment. The students set up their equations and then tested whether their calculations were correct by rolling a ball down a ramp and placing a cup where they predicted the ball would hit the floor. “We basically learned that the model we had learned in class applied to real life,” she says.
Other aspects of the course also attempt to replicate authentic practices. Students are assessed on their use of scientific apparati. Unlike the traditional approach of assigning many homework problems that are variations on the same thing, the homework in the courses taught with the modeling approach includes fewer problems that require deeper understanding. “We give homework every day, but just one problem that has to be done thoroughly and explained in words,” Brewe notes.

Studies of Modeling Instruction indicate that students taught through this method outperform those taught in lecture-format classes, with benefits for Hispanic and white students and for male and female students (Brewe et al., 2010). Student attitudes about physics, as measured by a standardized survey instrument, have also improved (Brewe et al., 2013). In addition, students in Modeling Instruction sections report 10 times the number of ties among themselves than students in lecture sections. Brewe, Kramer, and O’Brien (2009) see this as evidence that students in Modeling Instruction form richer and more deeply connected learning communities, which further contribute to learning gains.
Supplementing Instruction with Tutorials
Tutorials can serve as a ready-made tool for instructors who want to supplement their lectures with carefully sequenced, research-based learning exercises that students can do in small groups or independently. The widely influential Tutorials in Introductory Physics (McDermott and Shaffer, 2002) highlights tutorials created by Lillian McDermott,9 Peter Shaffer, and the physics education group at the University of Washington that are a prototype for research-based tutorials designed to promote the kind of active intellectual engagement necessary to develop a functional understanding of physics concepts. Tutorials are often used in a recitation or discussion section in which students work in small groups, but they can also be incorporated into a lecture or assigned as homework.
The genesis of the physics tutorials, says McDermott, was her realization that even graduate students in physics had difficulties in understanding key physics concepts. “They were much more sophisticated, so they could get by with memorizing formulas; they could get by with knowing how to do things without necessarily understanding them. If you asked questions about understanding, they turned out to have the same problems as the other students.” This led McDermott to begin documenting common student difficulties and researching the impact of instructional strategies to confront them.
Typically, tutorials target critical concepts and are designed to have students “elicit, confront, and resolve” common difficulties in learning, says McDermott. The questions on the tutorial worksheets are structured to help lead students through the steps of reasoning necessary to develop and apply basic concepts.
One of the Washington tutorials, for example, uses a simple optical system consisting of a light source, a mask with a small triangular hole, and a screen to help students understand and apply two basic principles from geometrical optics: light travels in straight lines, and light rays from every point on an object travel outward in all directions. The tutorial first asks students, working in small groups, to predict the images that will appear on the screen from different types of lightbulbs when the size and shape of the aperture changes. After the students have made predictions and explained their reasoning to one another, they observe what actually happens and try to resolve any discrepancies with their predictions. They are then asked to predict and explain up-down and left-right inversions of images produced by asymmetric sources. The point is to help students understand that the size and shape of the source, the size and shape of the aperture, and the distances
________________
9 Interview, April 18, 2013.
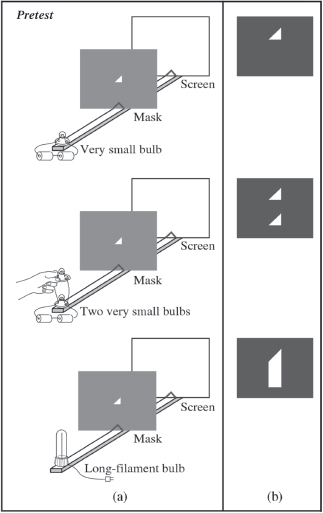
(a) Students were asked to sketch what they would see on the screen. (b) Correct answers.
involved can all have an effect on the image (Heron and McDermott, 1998).
The initial question is framed so that many students will make an incorrect prediction, says Paula Heron,10 a professor in the physics department. When they actually try the experiment, such as turning on the lightbulb or changing the shape of the aperture, the results are not what they expected. “That’s an opportunity to take advantage of that surprise, or confusion, or dismay, whatever the emotion is,” says Heron. “It’s a moment when you’re likely to get strong intellectual engagement. By letting them see how their thinking led to one prediction and how a different way of thinking leads to a correct prediction, you can get them to not only see what the correct thing is, but give them an opportunity to reflect on the patterns of thought or beliefs they had, or the way that they were approaching the problem, that were incorrect or unproductive.”
Numerous studies have demonstrated that the University of Washington tutorials significantly improve students’ conceptual understanding and their more general scientific reasoning (see a review by Docktor and Mestre, 2011). These tutorials do require fairly extensive facilitation by teaching assistants.
Researchers and faculty have developed similar tutorials in astronomy, biology, geosciences, and other disciplines (see Chapter 2 for more information about the geosciences tutorials designed by Karen Kortz and Jessica Smay).
Science and Engineering Practices and Authentic Experiences
Think back to why you went into science or engineering. You likely became fascinated by something you saw or did that opened your mind to bigger ideas, such as your first view through a microscope of the amoebas that lived in a water puddle
________________
10 Interview, April 12, 2013.
or your realization that so much of the world around you could be explained by the principles of physics. But you probably also found satisfaction in practicing science or engineering—for example, by collecting and analyzing data, using the “tools of the trade,” or designing a device or technology that can make our lives or our world better.
For students to be competent in science and engineering, they need more than knowledge of the content of a discipline. They also need to understand the practices, tools, and ways of thinking in that discipline—and how these can be applied to solve real problems. Although most students in science or engineering courses will not pursue careers in these fields, all of them can benefit from understanding the process of science and how it can be used to evaluate and make decisions about issues facing society.
Research at the K–12 level has shown that with well-designed curricula and instruction, students can become more competent in the practice of science at the same time they learn content at a deeper level (National Research Council, 2007). Some instructional strategies at the undergraduate level combine these goals. Using the practices of a profession and working on authentic problems also makes learning more relevant in a way that many students find motivating. These approaches may have students work on authentic problems or activities, analyze published data, use tools commonly used in the practice of a discipline, and participate in research experiences, as illustrated by the following examples.
In the SCALE-UP Foundations of Biology course for majors at the University of Minnesota, the lab and lecture components are integrated into thrice-weekly sessions of almost two hours each. As part of the course, students do a 5-week and a 10-week team project that explores an authentic problem of social value that they identify themselves, says Wright, one of the instructors. Past projects have dealt with the impact of climate change on coral reefs; the biological effects of an oil spill, Alzheimer’s disease, or cancer; and even the design of camouflage clothing. Students apply their knowledge of genetics and evolution to propose solutions, says Wright.
As the culminating experience of the class, students present and discuss their proposals in a class poster session, just as biologists would. “We try in every way to represent the authentic work of the discipline,” says Wright.
A unique undergraduate engineering program in the Iron Range mining region of northern Minnesota uses authentic engineering design projects to develop students’ understanding of core competencies in engineering and professional engineering practices.
Prospective Engineers Learn Technical and Professional Competencies Through Authentic Projects
Through the Iron Range Engineering program, students learn both the technical knowledge and the professional practices of engineering by carrying out engineering design projects (Minnesota State University, Mankato, 2013). The program is a partnership between Itasca Community College in Grand Rapids, Minnesota, and Minnesota State University (MNSU), Mankato. Most of the participants are community college graduates who are completing the upper-division portion of an engineering bachelor’s degree.
The majority of students’ design projects are sponsored by the mining, milling, and manufacturing industries in Minnesota’s Iron Range, says Rebecca Bates,a an engineering professor at MNSU, Mankato, although some students do entrepreneurial projects or work with faculty on a research project. One student project, for example, examined the problem of heat loss and transfer in a paper mill. This loss affects the quality of the paper produced because the paper must go through phases of dampness and dryness. The student team analyzed changes in the temperatures and airflow in the plant and made recommendations for improving insulation, air movement, and other factors.
Rather than attending lecture-based classes, students learn by conducting their design projects, participating in seminars and conversations with faculty and external experts, receiving feedback on their projects, and engaging in self-study and peer learning. Some of the required credits are related to engineering knowledge about areas such as digital logic and thermodynamics. Others are related to professional skills, such as teamwork, ethics, individual communication, and time-management skills—“all of the things we connect with professionalism,” says Bates.
In the proposal stage, students must develop both a work plan for their project and a learning plan that outlines their learning objectives for professional skills and technical knowledge. The plan also describes the methods students will pursue to meet those learning objectives and the means that will be used to assess and reflect on their learning. Students present evidence that they have learned the requisite knowledge and skills to the faculty, who review the evidence and assess students’ competency through oral and written exams.

Students’ written reflections on what and how they are learning are an important part of the evidence used to assess their work. “When they start, they hate doing this because it’s more work,” says Bates. “They have produced a finished assignment and gotten the right answer, and then they say, ‘You want me to write about how I got it?’” But, she adds, these reflections often lead to improvements in their projects, their learning processes, and their retention of content.
________________
a Except where noted, the information in this case study comes from an interview with Rebecca Bates, July 8, 2013.
Students in Christopher Swan’s11 civil and environmental engineering and geotechnical engineering courses at Tufts University learn about engineering practices through projects with a community service angle. Working in small groups, students have tackled projects with environmental implications: for example, evaluating what happens when soils in a waste site are heated to high temperatures, as occurs in sites remediated with incineration. During the course of six to eight weeks, the students develop a research process and conduct a study of such questions as whether the resulting soil can be used as clean soil and how it differs from uncontaminated soil from the same site. The work of one student group was published as a symposium paper, says Swan. Students “get turned on by a research project—it may spark a deeper interest in engineering,” he says.
Swan also works with students participating in service learning engineering projects in developing nations through Engineers Without Borders. In one ongoing project, students work with a village in Uganda to determine the most feasible way to continuously pump drinking water from a local source.
The approaches described in this chapter are intended to give science and engineering faculty a flavor of how research on how people learn can inform instructional practices in undergraduate education. Many other strategies backed by evidence of effectiveness, and many sources of information and support, await instructors who wish to explore these ideas in their courses. If you take the next step, you will be joining a community of practitioners engaged in learning, experimenting, and sharing results.
Resources and Further Reading
Discipline-Based Education Research: Understanding and Improving Learning in Undergraduate Science and Engineering (National Research Council, 2012)
Chapter 6: Instructional Strategies
Pedagogy in Action Web portal of the Science Education Resource Center (SERC)
http://serc.carleton.edu/sp/index.html
Process Oriented Guided Inquiry Learning (POGIL)
www.pogil.org
________________
11 Interview, August 27, 2013.
































