The External Environment Influencing Diagnosis: Reporting, Medical Liability, and Payment
This chapter focuses on the external environment and how it contributes to the diagnostic process and the occurrence of diagnostic errors (see Figure 7-1). The category of external environmental factors is quite broad and may include: error reporting, medical liability and risk management, payment and care delivery, and oversight processes (such as accreditation, certification, and regulatory requirements). While the committee does consider oversight processes to be external environmental factors, they are discussed in the sections on health care professional education and competency in Chapter 4 and in the section on the oversight of health care organizations in Chapter 6.
In this chapter the committee emphasizes the need for safe environments for voluntary error reporting, without the threat of legal discovery or disciplinary action, where health care organizations can analyze and learn from diagnostic errors in order to improve diagnosis. The role of medical liability reform is also described as an opportunity to increase the disclosure of diagnostic errors as well as to promote improved reporting, analysis, and learning from diagnostic errors. The committee highlights the potential for payment models—both current and new—to incentivize improved diagnostic performance. Importantly, this chapter reflects the committee’s commitment to consider recommendations from both a pragmatic and an aspirational perspective. The committee’s recommendations balance the urgent need to improve diagnosis by identifying immediate opportunities for improvement while also considering more fundamental changes that are likely to take significant effort and time to achieve. As noted elsewhere in the report, the committee’s recommenda-
tions to improve diagnosis in this chapter may also improve patient safety and health care more generally. For example, the evaluation of the Patient Safety Organization (PSO) program is likely to be informative for error reporting broadly; adoption of communication and resolution programs (CRPs) has the potential to improve disclosure and error analysis for all types of errors in health care; and reforming fee-for-service (FFS) payment and documentation guidelines could also benefit the health care system more broadly.
REPORTING AND LEARNING FROM DIAGNOSTIC ERRORS
The committee concluded that there need to be safe, confidential places for health care organizations and professionals to share and learn from their experiences of diagnostic errors, adverse events, and near misses. Conducting systems-based analyses of these events presents the best opportunity to learn from such experiences and to implement changes to improve the diagnostic process. The Institute of Medicine’s (IOM’s) To Err Is Human: Building a Safer Health System (2000) report recommended
that reporting systems be used to collect this information. Various groups, including individual states, The Joint Commission, the Department of Veterans Affairs, and PSOs, have developed a number of reporting systems which collect different types of information for different purposes. Characteristics of successful reporting systems include: reporting is safe for those individuals who report; reporting leads to constructive responses; adequate expertise and resources enable learning from reporting; and the results of reporting can be disseminated (Barach and Small, 2000; WHO, 2005). In contrast, if health care organizations focus on punishing individuals who make mistakes, it will prevent people from reporting because they fear that a report may be used as evidence of fault, could precipitate lawsuits, or could result in disciplinary action by state professional licensing boards and employers (IOM, 2012; WHO, 2005). Thus, there is a need for safe environments in which there is not the threat of legal discovery or disciplinary action, where diagnostic errors, adverse events, and near misses can be analyzed and learned from in order to improve the quality of diagnosis and prevent future diagnostic errors. In line with the To Err Is Human report, the committee recommends that the Agency for Healthcare Research and Quality (AHRQ) or other appropriate agencies or independent entities should encourage and facilitate the voluntary reporting of diagnostic errors and near misses.
Unfortunately, it is often difficult to create environments where diagnostic errors, adverse events, and near misses can be shared and discussed. Health care organizations and clinicians have been challenged by the limitations of inconsistent and individual state-enacted peer review and quality improvement processes for the protection of information relating to adverse events and medical errors, the external use of such information, and what benefits the health care organizations and clinicians receive from reporting. In response to this challenge, the To Err Is Human report recommended that “Congress should pass legislation to extend peer review protections to data related to patient safety and quality improvement that are collected and analyzed by health care organizations for internal use or shared with others solely for purposes of improving safety and quality” (IOM, 2000, p. 10). In 2005 the Patient Safety and Quality Improvement Act (PSQIA) was passed by Congress; the act confers privilege and confidentiality protections to health care organizations that share specific types of patient safety information with federally listed PSOs (HHS, 2015). According to AHRQ, which shares responsibility for implementing PSQIA with the Office for Civil Rights, “The Act promotes increased patient safety event reporting and analysis, as adverse event information reported to a … PSO is protected from disclosure in medical malpractice cases. This legislation supports and stimulates advancement of a culture of safety in health care organizations across the country,
leading to provision of safer care to patients” (AHRQ, 2015a, p. 23). The PSO program provides an important national lever to increase voluntary error reporting and analysis which is well aligned with the committee’s recommendation. However, progress in implementing the PSO program has been slow (AHRQ, 2015a; GAO, 2010). The committee is concerned that a number of challenges with the current program may limit the extent to which it can facilitate much-needed voluntary reporting, analysis, and learning of diagnostic errors and near misses (see section below on the evaluation of the PSO program).
Due to this concern, the committee’s recommendation recognizes that additional federal efforts across the Department of Health and Human Services (HHS), as well as the involvement of other independent entities, need to be considered in order to prioritize voluntary event reporting for diagnostic errors and near misses. Support of this recommendation can be found in the IOM report Health IT and Patient Safety: Building Safer Systems for Better Care, which reviewed the existing reporting systems in health care and concluded that despite the various reporting systems and numerous calls for change, adverse event reports are not being collected and analyzed in a comprehensive manner (IOM, 2012). The report concluded that “learning from these systems is limited because a multitude of different data is collected by each system, hampering any attempt to aggregate data between reporting systems” (IOM, 2012, p. 152). After reviewing the opportunities to improve adverse event reporting, the committee that produced the 2012 report made a recommendation for a new entity, akin to the National Transportation Safety Board, that could investigate “patient safety deaths, serious injuries, or potentially unsafe conditions” and report results of these activities (IOM, 2012, p. 11). That committee suggested that this entity’s purview could include (1) collecting reports of adverse events; (2) analyzing collected reports to identify patterns; (3) investigating reports of patient deaths or serious injuries related to health IT; (4) investigating trends of reports of unsafe conditions; (5) recommending corrective actions; (6) providing feedback based on these investigations; and (7) disclosing the results of the investigations to the public.
Because efforts to improve voluntary reporting and analysis at the national level have been slow, the current committee also recognized the potential for more localized efforts that could be carried out while national efforts continue to be developed and improved. In the interim, smaller-scale efforts to improve voluntary reporting and learning from diagnostic errors, adverse events, and near misses may be helpful for generating and sharing the lessons learned from such efforts. For instance, at the level of health care organizations, quality and patient safety committees can incorporate the analysis of and learning from diagnostic errors, and these activities may be protected from disclosure by state statutes. In
an integrated delivery system in Maine, for example, a surgical quality collaborative was established to review the quality and safety of surgical care, compare results to national and regional data, and provide feedback to participating organizations.1 Another option that some organizations (including PSOs) are incorporating is the use of “safe tables” forums (WSHA, 2014), which are “members-only, shared learning meeting[s] of healthcare peers to exchange patient safety experiences, discuss best practices and learn in an open, uninhibited and legally protected environment” (MHA PSO, 2015). The limitation to this approach is that the best practices and lessons learned cannot be shared beyond the participants.
Evaluation of the PSO Program
The PSO program enables public or private organizations to be listed as a PSO provided that they meet certain qualifications articulated in the patient safety rule (AHRQ, 2015e). PSO designation indicates that an organization is “authorized to serve providers as independent patient safety experts and to receive data regarding patient safety events that will be considered privileged and confidential” (GAO, 2010, p. 2). PSOs do not receive federal funding, but they can recruit health care organizations and clinicians to join their PSO. When health care organizations or health care professionals join a PSO, they can then voluntarily send patient safety data to the PSO for analysis and feedback on how to improve care. Additionally, PSOs can send de-identified patient safety data to the Network of Patient Safety Databases (NPSD) overseen by AHRQ. The intent of the program is that AHRQ will then analyze the aggregated data and publish reports (GAO, 2010). A provision in the Affordable Care Act will likely increase the number of hospitals who join PSOs; per the HHS 2015 Payment Notice, hospitals with more than 50 beds will be required to join a PSO by January 2017 in order to contract with health plans in insurance exchanges (CFPS, 2015).2 There is very limited information about the impact that PSOs have on learning and improving the quality and safety of care. The Government Accountability Office concluded in 2010 that it was too early to evaluate the effectiveness of the PSO program (GAO, 2010). AHRQ is still in the process of implementing the NPSD, and to this point aggregated information collected from PSOs has not been analyzed or shared.
Currently there are more than 80 listed PSOs (AHRQ, 2015c), and the
______________
1 The MaineHealth Surgical Quality Collaborative was developed in accordance with the provisions of the Maine Health Security Act, 24 Me.Rev.Stat.Ann. § 2501, et seq to maintain the confidentiality of information and data reviewed.
2 See www.federalregister.gov/articles/2013/12/02/2013-28610/patient-protection-and-affordable-care-act-hhs-notice-of-benefit-and-payment-parameters-for-2015 (accessed December 6, 2015).
PSO network is active in sharing information with their members about strategies to mitigate patient safety events, as evidenced by PSO websites. AHRQ has also developed “Common Formats,” or generic- and event-specific forms, to encourage standardized event reporting among PSOs (AHRQ, 2015b). However, use of the Common Formats is voluntary, and some organizations are implementing these variably or using legacy reporting formats (ONC, 2014). To facilitate the aggregation of patient safety data, AHRQ established the PSO Privacy Protection Center (PSO PPC). The PSO PPC receives data from PSOs, facilitates use of the Common Formats, de-identifies data in a standardized manner, validates the quality and accuracy of the data, provides technical assistance to PSOs and other Common Formats users, and transmits non-identifiable information to the NPSD (AHRQ, 2015a).
The PSO PPC works with individual PSOs that wish to submit de-identified patient safety event information. In order to submit reports, PSOs are required to sign a data use agreement with the PSO PPC. By the end of fiscal year (FY) 2014, 20 of 76 listed PSOs had established data use agreements with the PSO PPC (AHRQ, 2015a). AHRQ reports that while these data use agreements “grew in number in FY 2014, and some data were transmitted to the PSO PPC, none have been of sufficient quality and volume to ensure that data transmitted to the NPSD is both accurate and non-identifiable” (AHRQ, 2015a, p. 53). For FY 2015, AHRQ expects the volume of data submission to the PSO PPC and the quality of the data submitted to increase significantly. AHRQ’s goal is to gather “sufficient patient safety event reports to transmit to the NPSD,” and the FY 2015 target is to transfer 25,000 patient safety event reports to the NPSD (AHRQ, 2015a, p. 53).
There are concerns that the federal privilege protections extended by PSQIA are not shielding organizations from state reporting requirements; a recent ruling by the Kentucky Supreme Court found that the information a hospital is required to generate under state law is not protected by PSQIA, even if it is shared with a PSO.3 This type of court decision could undermine the creation of safe environments for sharing this information and thus make voluntary submissions to PSOs much less likely.
Given that the PSO program has the potential to improve learning
______________
3Tibbs v. Bunnel, Ky., 2012-SC-000603-MR (August 21, 2014). The Kentucky Supreme Court held that the incident report developed by the University of Kentucky Hospital’s patient safety evaluation system (PSES), following the death of a patient, was not protected as patient safety work product (PSWP) under PSQIA. While this case may not set any official precedent in other states, it will be considered persuasive case law. Organizations that have established a PSES for reporting to a PSO need to explore any state-mandated safety and quality regulations to ensure that the collection of such information is conducted in harmony with the PSES to ensure protection as PSWP.
about diagnostic errors and to expedite the implementation of solutions and adoption of best practices, it is important to evaluate whether the program is meeting the statutory objectives of PSQIA—namely, that the PSO program is creating opportunities to examine and learn from medical errors, including diagnostic errors. Thus, the committee recommends that AHRQ should evaluate the effectiveness of PSOs as a major mechanism for voluntary reporting and learning from these events. Given the concern over the erosion of PSQIA privilege protections at the state level, the evaluation could also focus on whether these protections are consistent with Congress’s intent in enacting the legislation. While the evaluation of the PSO program is ongoing, PSOs can help support voluntary reporting efforts by educating their members about the applicable state peer review protections as well as about the PSQIA privilege protections. Health care organizations participating in PSOs can also take steps to ensure that any information and data shared with PSOs are protected by defining their patient safety evaluations systems broadly and by carefully analyzing the information they intend to submit to a PSO in order to minimize the chance that the PSQIA privilege is abrogated (or invalidated) at the state level.
The evaluation of the PSO program could also explore how the PSO program influences efforts to improve transparency within health care organizations. According to a recent report, “PSOs have the potential to foster transparency through increased reporting of complications and errors, and identification and sharing of learning and best practices; however, it remains to be seen how successfully these groups can balance the need for a protected space to which organizations can voluntarily report errors and the need for open sharing of information outside the organization” (National Patient Safety Foundation’s Lucian Leape Institute, 2015, p. 16). The committee recognizes that efforts to improve diagnosis can include both a focus on improving the disclosure of medical errors to patients and their families (see discussion on CRPs) and efforts to improve voluntary reporting and learning.
In addition, AHRQ’s evaluation needs to focus on how AHRQ and PSOs can improve the voluntary reporting of diagnostic errors and learning from those errors, which have not been a major focus within PSOs to date. The committee recommends that AHRQ should modify the PSO Common Formats for reporting of patient safety events to include diagnostic errors and near misses. To implement Common Formats specific to diagnostic error, AHRQ could begin with high-priority areas (such as the most frequent diagnostic errors or “don’t miss” health conditions that may result in significant patient harm, such as stroke, acute myocardial infarction, and pulmonary embolism). AHRQ could also consider whether
other PSO activities, such as discussions during annual PSO meetings, could focus attention on diagnostic errors.
The two core functions of the medical liability system are to compensate negligently injured patients and to promote quality by encouraging clinicians and organizations to avoid medical errors. Although the medical liability system may act as a generalized deterrent to medical errors, it is not well aligned with the promotion of high-quality safe care (Mello et al., 2014b). Concerns about medical liability prevent clinicians from disclosing medical errors to patients and their families, despite calls from numerous groups that full disclosure is an ethical necessity (Hendrich et al., 2014; Sage et al., 2014) and despite the fact that such disclosures are a requirement for The Joint Commission accreditation. Clinicians often struggle to fulfill this responsibility: There is limited guidance for clinicians about how to disclose this information effectively, and a number of factors, including embarrassment, inexperience, lack of confidence, and mixed messages from risk managers and health care organizations’ senior leadership, can thwart disclosures to patients and their families (Gallagher et al., 2007, 2013; The Joint Commission, 2005; Schiff et al., 2014).
The current tort-based judicial system for resolving medical liability claims creates barriers to improvements in quality and patient safety and stifles continuous learning. Medical malpractice reform could be designed to permit patients and health professionals to become allies in trying to make health care safer by encouraging transparency about errors. Such an approach would allow patients to be promptly and fairly compensated for injuries that were avoidable, while at the same time turning errors into lessons to improve subsequent performance (Berenson, 2005; Mello and Gallagher, 2010; Mello et al., 2014a).
The IOM report Fostering Rapid Advances in Health Care: Learning from System Demonstrations concluded that there are numerous challenges to the current medical liability system, including the many instances of negligence that do not result in litigation and, conversely, malpractice claims that are not the result of negligent care, judgments that are inconsistent with the evidence base, and highly variable compensation for similar medical injuries (IOM, 2002). Patients and their families are poorly served by the current system; only a fraction of negligently injured patients receive compensation, typically after a protracted and adversarial litigation process (AHRQ, 2014; Kachalia and Mello, 2011). One analysis found that fewer than 2 percent of patients who experienced adverse events due to medical negligence actually filed malpractice claims (Localio et al., 1991);
another analysis found that the rates of paid medical malpractice claims have steadily declined since the early 2000s (Mello et al., 2014b).
An ongoing medical liability concern is the practice of defensive medicine. Defensive medicine “occurs when doctors order tests, procedures, or visits, or avoid high-risk patients or procedures, primarily (but not necessarily soley) to reduce their exposure to malpractice liability” (OTA, 1994, p. 13). The practice of defensive medicine is a barrier to high-quality care because it can lead to overly aggressive and unnecessary care. For example, clinicians who practice defensive medicine may order more diagnostic tests than are necessary (Hoffman and Kanzaria, 2014; Kessler et al., 2006; Mello et al., 2010). Overtesting in the diagnostic process has the potential to cause patient harm—both from the risk of the diagnostic test itself as well as from the resulting cascade of diagnostic and treatment decisions that stem from the test result (Hoffman and Kanzaria, 2014) (see also Chapter 3).
Diagnostic errors are a leading cause of malpractice claims, and these claims are more likely to be associated with patient deaths than other types of medical errors (Tehrani et al., 2013). Reforming the medical liability system, therefore, has the potential to improve learning from diagnostic errors, to facilitate the disclosure of diagnostic errors to patients and their families, and may produce fairer outcomes in the medical injury resolution processes. The committee recommends that states, in collaboration with other stakeholders (health care organizations, professional liability insurance carriers, state and federal policy makers, patient advocacy groups, and medical malpractice plaintiff and defense attorneys), should promote a legal environment that facilitates the timely identification, disclosure, and learning from diagnostic errors.
There have been many calls for changes to the medical liability system. Traditional mechanisms to reform the liability system—such as imposing barriers to bringing lawsuits, limiting compensation, and changing the way that damage awards are paid—have not resulted in improvements in either compensating negligently injured patients or deterring unsafe care (Mello et al., 2014b). Thus, the committee concluded that these stakeholders need to consider alternative approaches to improving the legal environment and promoting learning from diagnostic errors. The To Err Is Human report concluded that alternative approaches to the resolution of medical injuries could reduce the incentive to hide medical injuries, and in 2002 the IOM proposed state-level demonstration projects to explore alternative approaches to the current liability system that are patient-centered and focused on patient safety (IOM, 2000, 2002). In 2010, AHRQ allocated approximately $23 million in funding for demonstration and planning grants aimed at finding ways to improve medical injury compensation and patient safety (AHRQ, 2015d; Kachalia and Mello,
2011). Five of the seven demonstration grants (totaling $19.7 million in awarded funds) that were funded by AHRQ focused on CRPs, one on safe harbors for following evidence-based clinical practice guidelines, and one on judge-directed negotiation. The 13 planning grants (totaling $3.5 million) were diverse and included CRPs, safe harbors, and other formats (AHRQ, 2015d). These demonstration and planning grants were somewhat limited, however, because they could not involve approaches that required legislative changes (such as administrative health court demonstrations) (Bovbjerg, 2010). Furthermore, while the Affordable Care Act authorized $50 million to test new approaches to the resolution of medical injury disputes, this funding was never appropriated.
Although enthusiasm for alternative approaches to the current medical liability system is growing, in general the progress toward such approaches has been slow, especially for those that involve more fundamental changes to the medical liability system. Thus, the committee took both a pragmatic and an aspirational approach to considering which changes to medical liability could promote improved disclosure of diagnostic errors and opportunities to learn from these errors. A number of alternative approaches to the current medical liability system were evaluated, and the committee concluded that the most promising approaches included CRPs, the use of clinical practice guidelines as safe harbors, and administrative health courts (see Box 7-1). CRPs represent a more pragmatic approach in that they are more likely to be implemented in the current medical liability climate, and they have a strong focus on improving patient safety as well as on reducing litigation. Thus, the committee recommends that states, in collaboration with other stakeholders (health care organizations, professional liability insurance carriers, state and federal policy makers, patient advocacy groups, and medical malpractice plaintiff and defense attorneys), should encourage the adoption of CRPs with legal protections for disclosures and apologies under state laws.
Safe harbors for adherence to clinical practice guidelines may also help facilitate improvements in diagnostic accuracy by encouraging clinicians to follow evidence-based diagnostic approaches; however, most clinical practice guidelines address treatment, not diagnosis. Moreover, implementing safe harbors for adherence to these guidelines will be administratively complex. Administrative health courts offer a fundamental change that would promote a more open environment for identifying, studying, and learning from errors, but their implementation will be a major challenge due to operational complexity and to resistance from stakeholders who are strongly committed to preserving the current tort-based system. Thus, the committee concluded that these changes are more aspirational, and recommends that states and other stakeholders should conduct demonstration projects of alternative approaches to the
resolution of medical injuries, including administrative health courts and safe harbors for adherence to evidence-based clinical practice guidelines. The following sections describe the alternative approaches, the challenges influencing their implementation, and the potential benefits for improving diagnosis.
Communication and Resolution Programs
CRPs have recently garnered significant attention as a means of improving the disclosure and resolution of medical injuries and improving patient safety. A number of the AHRQ demonstration projects focused on CRPs, and organizations such as the American College of Physicians and the American College of Surgeons have called for continued experimentation (ACP, 2014; ACS, 2015). At 14 hospitals in 3 health care systems across the country, AHRQ is currently developing and field-testing an educational toolkit on CRPs which teaches about the best practices from the CRP-focused demonstration projects (AHRQ, 2015a). CRPs offer a principled, comprehensive, and systematic approach to responding to patients who have been harmed by their health care. They are an integral component of a larger commitment to quality and patient safety. CRPs seek to meet the needs of the affected patient and his or her family; it is the health care organization’s responsibility to address the quality issues and safety gaps that caused the event. While some of the specifics related to CRP implementation may vary based on an organization’s circumstances, Box 7-1 describes the essential components of a CRP.
CRPs could improve patient safety generally and reduce diagnostic errors in several ways. CRPs rely on creating transparent health care cultures in which the early reporting of adverse events is the norm and is coupled with systems-based event analysis designed to understand the root causes of the event and to aid in the development of plans for preventing recurrences. Increased transparency surrounding diagnostic errors can help foster an improved culture of reporting, which in turn can promote learning about and identifying interventions to improve the safety and quality of diagnosis (Mello et al., 2014a). CRPs also emphasize remaining transparent about adverse events—including diagnostic errors—with patients and their families.
The disclosure of medical errors also can also improve outcomes for patients, their families, and health care professionals (Delbanco and Bell, 2007; Helmchen et al., 2010; Hendrich et al., 2014; Lopez et al., 2009). In some cases, clinician disclosure of medical errors to patients is associated with higher ratings of quality care by patients (Lopez et al., 2009). When a CRP was implemented at the University of Michigan Health System (UMHS), it was associated with fewer malpractice claims, faster claims
BOX 7-1
Description of Alternative Approaches to the Medical Liability System
-
Communication and resolution programs (CRPs) are principled comprehensive patient safety programs in which health care professionals and organizations openly discuss adverse outcomes with patients and proactively seek resolution while promoting patient-centeredness, learning, and quality improvement. CRPs typically incorporate the following elements:
- Early reporting of adverse events to the health care organization or liability insurer for rapid analysis using human factorsa and other advanced event analysis techniques
- Developing plans for preventing recurrences and communicating these plans to patients and their families
- Open communication with patients and their families about unanticipated care outcomes and adverse events
- Proactively seeking resolutions, including offering an explanation as to why the event occurred and an acknowledgment of responsibility and/or an apology
- Initiating support services, both emotional and other types of support, for the patient, family, and care team
- Where appropriate, offering timely reimbursement for medical expenses not covered by insurance or compensation for economic loss or other remedies
- Safe harbors for adherence to evidence-based clinical practice guidelines are laws that provide health care professionals and organizations a defense against a malpractice claim if they can show that they followed a clinical practice guideline in providing care for a patient. Safe harbors:
processing times, and reduced liability costs and settlement amounts (Boothman et al., 2009, 2012; Kachalia et al., 2010). Safety culture scores at UMHS also improved with the implementation of the CRP; however, it is difficult to attribute causation to the CRP program (Boothman et al., 2012). CRPs “appear to be effective in improving communication with patients and families. Disclosure reportedly became more routine and robust in implementing hospitals after clinicians were given disclosure training and risk managers began more closely monitoring whether and how disclosures were carried out” (Mello et al., 2014b, pp. 2150–2151).
CRPs continue to expand in the United States. For example, the Massachusetts Alliance for Communication and Resolution following Medical Injury (MACRMI) is committed to spurring adoption of CRPs and sharing lessons learned to improve the dissemination of CRPs throughout Massachusetts (MACRMI, 2015). MACRMI supported enabling legislation
-
- May create a rebuttable presumption (i.e., it is introduced as evidence of the standard of care, but is not dispositive) or irrefutable presumption of nonnegligence
-
Administrative health courts offer a system of administrative compensation for medical injuries which has the following components:
- Injury compensation decisions are made outside the regular court system by specially trained judges
- Compensation decisions are based on a standard of avoidability of medical injuries rather than a standard of negligence—claimants must show that the injury would not have occurred if best practices had been followed or an optimal system of care had been in place, but they need not show that care fell below the standard expected of a reasonably prudent health care professional
- Compensation decisions are guided by previous determinations about the preventability of common medical adverse events; this knowledge, coupled with precedent, is converted to decision aids that allow fasttrack compensation decisions for certain types of injury
- Previous determinations also inform decisions about the amount of the award for economic and noneconomic damages
——————
a Human factors (or ergonomics) is: “the scientific discipline concerned with the understanding of interactions among humans and other elements of a system, and the profession that applies theory, principles, data and methods to design in order to optimize human well-being and overall system performance. Practitioners of ergonomics and ergonomists contribute to the design and evaluation of tasks, jobs, products, environments and systems in order to make them compatible with the needs, abilities and limitations of people” (IEA, 2000).
SOURCES: Chow, 2007; Jost, 2006; Mello et al., 2014b; Peters, 2008; Timm, 2010.
that adopted the UMHS CRP model, including a 6-month pre-litigation period and protections for disclosures and apologies (MACRMI, 2015).
Although establishing CRPs does not require legislative changes, CRP adoption could be facilitated by changes to state laws, such as laws protecting disclosures and apologies (Sage et al., 2014). For example, the American College of Physicians has called for “strong, broad legal protections that ensure apologies from physicians and other health care professionals are inadmissible” in a subsequent medical malpractice action (ACP, 2014). Though more than two-thirds of states have apology laws, the majority only protect the clinician’s voluntary expression of sympathy from use by a patient in malpractice litigation (Mastroianni et al., 2010). A small number of states also protect explanations of the event or expressions of fault, or both; however, Sage and colleagues concluded that no states protect “the full scope of information that patients report needing
when an unexpected outcome arises: a preliminary explanation of what happened; an expression of sympathy; an admission of responsibility; and a final analysis of the causes and consequences of the event, with information about remedial actions taken to prevent such incidents in the future” (Sage et al., 2014, p. 14). Of the nine states that have disclosure laws, a majority require health care organizations to notify patients when an event has caused serious harm: “States vary on whether the disclosure receives protection from subsequent use by a plaintiff in malpractice litigation. For the most part, states provide limited, if any, procedural guidance; some states require written—versus oral—communication or timely communication” (Mastroianni et al., 2010, p. 1614).
The implementation of CRPs face a number of challenges. One challenge is HHS’s recent interpretation of the reporting requirements to the National Practitioner Data Bank (NPDB). Federal law requires that medical liability insurers report malpractice payments to the NPDB, which was initially established to prevent clinicians from concealing disciplinary and malpractice histories as they moved across state lines (Sage et al., 2014). An Oregon law attempted to assert that the NPDB reporting was not required if a settlement resulted from a mediation mechanism, such as a CRP (Robeznieks, 2014), but HHS concluded that any payments stemming from written demands (whether part of mediation mechanisms or not) are required to be submitted to the NPDB (HHS, 2014). There are concerns that these reporting requirements will prevent participation in CRPs: “Physicians worry that CRPs will offer compensation when the physician was not at fault, either as a compassionate gesture or because the hospital or insurer deems it prudent to settle, and that, as a result, physicians will be reported to the NPDB more often” (Sage et al., 2014, p. 16). The reporting of settlements arising from mediation mechanisms to the NPDB could have negative effects on clinicians’ reputations, credentialing, or disciplinary actions, and at least one medical specialty society, the American College of Physicians, recommends that the reporting requirement be altered to encourage CRP participation (ACP, 2014).
Other considerations will influence the implementation and effectiveness of CRPs, including the presence of organizational champions and a culture that supports the reporting of medical errors; a focus on coaching and support services to help clinicians participate in disclosures and the CRP processes; and buy-in from and coordination with health care organizations and professional liability insurance carriers (Mello et al., 2014a).
Of particular interest is the potential for CRPs to promote widespread learning following adverse events. As growing numbers of health care organizations and professional liability insurers adopt CRPs, close collaboration among these programs and between these programs and PSOs could help ensure that the lessons learned from adverse events are shared
widely within and outside the organizations where the events occurred. The establishment of a national collaborative of CRPs could be one way to accelerate the spread of CRPs and to fully realize the quality and safety benefits of these programs.
Safe Harbors for Adherence to Evidence-Based Clinical Practice Guidelines
Safe harbors for following evidence-based clinical guidelines have the potential to raise the quality of health care by creating an incentive—liability protection—for clinicians to follow evidence-based clinical practice guidelines.4 Safe harbors can create an affirmative defense for health care professionals who adhered to accepted and applicable clinical practice guidelines. Input to the committee suggested that safe harbors, unlike other approaches to improving the medical liability environment, offer direct opportunities to improve diagnosis (Kachalia, 2014). While other approaches to improving medical liability focus on improving learning through improved disclosure, safe harbors focus on aligning clinical care with best practices.
Available evidence suggests that creating national standards of care against which clinicians are judged in malpractice claims can improve quality of care. Providing standardized guidelines for certain diagnostic work-ups and holding these to be the standard of care has the potential to reduce diagnostic error. Despite calls for safe harbors (ACP, 2014; Mello et al., 2014b), there is limited information about how effective safe harbors are in minimizing medical errors, partly because there have been relatively few pilot programs and those programs have had poor clinician participation (Kachalia et al., 2014; Mello et al., 2014b). A recent simulation analysis evaluated the potential impact of safe harbors and concluded that they constitute a promising approach to driving improvements in the quality of patient care, but their impact on liability costs and patient outcomes is likely to be minimal (Kachalia et al., 2014).
______________
4 Safe harbors for adherence to clinical practice guidelines differ from the current use of clinical practice guidelines in the courts. Typically, malpractice litigation uses expert testimony to determine whether the care provided by a clinician fell below the standard of care (what would be expected of a reasonably prudent clinician). Expert witnesses can introduce clinical practice guidelines as legal evidence, but many states permit defendants to escape liability if they demonstrated customary care, even if it is not considered optimal care (IOM, 2011). This is partly due to variability in how states define the standard of care. Some states employ a national standard (clinicians would be held to the same degree of care and skill that a reasonably competent health care professional in the same field would exercise under similar circumstances). Other states use a local standard of care (clinicians would be held to the degree of knowledge and skill that is generally exercised by the same professionals in the community where they practice).
There are a number of implementation challenges related to safe harbors for adherence to clinical practice guidelines. For example, it requires state endorsement of specific clinical practice guidelines for use in malpractice litigation. Furthermore, safe harbor programs may be administratively complex because they require determining which clinical practice guidelines apply, when they apply, and who makes the determination. Also, given the constantly changing evidence base, ensuring the timely updating of approved guidelines and making clinicians aware of the updates could be challenging (Bovbjerg and Berenson, 2012). Clinician acceptability is another concern. Clinicians may find it burdensome to have to comply with additional clinical practice guidelines for improving diagnostic performance and avoiding liability. Clinicians already encounter multiple guidelines from specialty associations, insurers, health care organizations, hospitals, and others, and these guidelines are likely not all in alignment. Additionally, recent policy changes add to the resistance of using clinical practice guidelines for legal purposes. The legislation that repealed the sustainable growth rate included a provision that prevents the use of guidelines or standards used in federal programs as proof of negligence: The “development, recognition, or implementation of any guideline or other standard” under the Medicare and Medicaid programs and any provision in the Affordable Care Act “shall not be construed to establish the standard of care or duty of care owed by a health care provider to a patient in any medical malpractice or medical product liability action or claim.”5
Administrative Health Courts
Administrative health courts have been proposed as a way to provide injured patients with expedited compensation decisions for certain types of medical errors and to promote the disclosure of medical errors (such as diagnostic errors). Administrative health courts are a nonjudicial way of handling medical injuries, in which cases are filed through an administrative process. The goal in using these courts is to quickly and equitably compensate patients who have experienced avoidable injuries without requiring them to become plaintiffs within the medical liability system who must prove negligence in an adversarial proceeding (Berenson, 2005).
There are various versions of how such an approach might work. In one version, specially trained judges preside and are assisted by investigations and opinions provided by neutral experts on the matter under consideration. Administrative health courts also take fault—or negligence—terminology out of the determination of liability and sub-
______________
5 Medicare Access and CHIP Reauthorization Act of 2015. P.L. 114-10. (April 16, 2015).
stitute it with the concept of avoidability (IOM, 2002; Mello et al., 2006). “[A] system based on an avoidability standard would award compensation to claimants who could show that their injury would not have occurred in the hands of the best practitioner or system” (Kachalia et al., 2008, p. 388). Proving negligence requires evidence that a clinician failed to meet a standard of care, is very fact-specific, and is more challenging to demonstrate; on the other hand, avoidability represents complications that generally should not occur under competent medical care (Berenson, 2005). Although substituting the negligence standard with an avoidability standard will lower the threshold for making these determinations, claimants will still have to establish cause—that their injuries were the result of their care rather than their underlying illnesses (Kachalia et al., 2008).
The establishment of administrative health courts could help to reduce process inefficiencies and inequities in compensation caused by shortcomings in the current system of tort liability, and adjudicated cases could be used to inform and foster the development of mechanisms to identify and mitigate medical errors (IOM, 2002; Mello et al., 2006). Administrative health courts have been described as holding theoretic appeal because “the model addresses some of the most important problems with the U.S. medical malpractice system, including the difficulty that patients have filing and prevailing in claims, the duration of litigation, the substantial overhead costs, the unpredictability of damages awards, and the punitive effect felt by physicians” (Mello et al., 2014b). Health courts have been used in other countries, including Denmark, New Zealand, and Sweden, and evidence suggests that they provide compensation to a greater number of claimants and are able to reach conclusions more quickly and at lower costs than tort-based mechanisms (ACP, 2006; Bovbjerg and Sloan, 1998; Mello et al., 2011).
Health courts appear to have bipartisan support in the United States: A nationwide poll conducted in 2012 found that 68 percent of Republicans, 67 percent of Democrats, and 61 percent of independents surveyed support the creation of health courts (Howard, 2012). Legislation to experiment with, or create, health courts has been proposed in a number of states—including Georgia, Maryland, New York, Oregon, and Virginia—but none has passed (Peters, 2008). Several organizations and experts have recommended pilot-testing or using health courts in the United States, but very few systems have been implemented or even tested (ACP, 2014; Howard and Maine, 2013; IOM, 2002; Mello et al., 2014b; Peters, 2008). There are only two state systems that implement the principles of health courts, and these uses are confined to cases involving neurological birth injury (Howard and Maine, 2013; Mello et al., 2014b).
There are several challenges associated with health courts, including the need for legislative action, which has been difficult to achieve (Mello
et al., 2014b; Peters, 2008). As mentioned earlier, resistance from stakeholders strongly committed to preserving the current tort-based system will be a major challenge to overcome. Another issue that needs to be considered is how a health court should make information on paid claims of avoidable injuries available to state professional licensing boards, state hospital licensing agencies, medical specialty boards, and the NPDB. Such reporting could have a chilling effect on clinician disclosure of diagnostic errors; however, there is a competing concern about limiting the transparency of information on potentially substandard care practices.
Professional liability insurance carriers and health care organizations that participate in captive or other self-insurance arrangements have an inherent interest in improving diagnosis. Many of these organizations are actively exploring opportunities to improve diagnosis and reduce diagnostic errors. According to input the committee received, “[M]edical liability serves as a rich training area for reducing diagnostic error” (Lembitz and Boyle, 2014, p. 1). Given the expertise of professional liability insurance carriers and captive insurers in understanding the contributing factors to diagnostic errors, they can bring an important perspective to efforts to improve diagnosis, both those focused on individual health care professionals and those focused on the work system components that may contribute to diagnostic errors. Thus, the committee recommends that professional liability insurance carriers and captive insurers should collaborate with health care professionals on opportunities to improve diagnostic performance through education, training, and practice improvement approaches and they should increase participation in such programs.
One way in which these groups are helping improve diagnosis is by conducting data analyses that characterize the reasons that diagnostic errors occur. PIAA, the industry trade association representing companies in the medical liability insurance field, has a data sharing project that gathers and analyzes data on medical professional liability claims submitted by its members (Parikh, 2014).6 The project’s findings are used to identify opportunities to reduce risk and improve patient safety in health care organizations. Individual carriers can also provide information to help improve the understanding of diagnostic errors that lead to medical
______________
6 As discussed in Chapter 3, one of the limitations of malpractice claims data is that these data are not necessarily representative of diagnostic error in clinical practice; in one analysis, fewer than 2 percent of patients who experienced adverse events due to medical negligence filed malpractice claims (Localio et al., 1991).
liability claims. For example, Physician Reciprocal Insurers (PRI), CRICO, and The Doctors Company have gathered data on submitted and paid malpractice claims that suggest that diagnostic errors are the cause of around 20 percent of all submitted claims and 52 percent of all paid claims (CRICO, 2014; Donohue, 2014; Troxel, 2014). CRICO synthesizes information on important issues in medical injury claims and produces reports on these issues (such as a report on diagnostic errors in ambulatory care settings) (CRICO, 2014). Professional liability insurers often have rich data because they have collected a variety of information (e.g., information from electronic health records [EHRs], statements from various participants in the diagnostic process, and information from court documents) in the course of preparing for medical malpractice lawsuits. This information can lead to important, albeit potentially nonrepresentative, insights about the vulnerabilities in the diagnostic process and about potential areas on which to focus in order to improve care. Improved voluntary participation in malpractice claims databases among all professional liability insurance carriers and captive insurers could be helpful for aggregating information and sharing lessons learned.
Many professional liability insurers offer risk management educational services that are designed to improve diagnostic performance. The associated activities include seminars, workshops, team training, residency training programs, and newsletters (Donohue, 2014; Lembitz and Boyle, 2014). COPIC, a provider of medical liability insurance, reported that it conducts more than 2,000 practice site visits each year, in which specially trained nurses use explicit criteria to identify patient safety and risk issues, including vulnerability to systems errors, communication failures, information transfer, EHR issues, and standardized processes (Lembitz and Boyle, 2014). In some cases, incentives such as discounted insurance premiums are offered to individuals to induce participation (Donohue, 2014; Lembitz and Boyle, 2014). Surveys suggest that clinicians perceive these educational and training approaches as beneficial; for example, PRI reported that 94 percent of the clinicians participating in their case review exercise believe that it will reduce the risk of diagnostic errors occurring in their practice (Donohue, 2014). Unfortunately, because of measurement difficulties, there is little information on the impact of these educational approaches on the occurrence of diagnostic error (Donohue, 2014; Lembitz and Boyle, 2014). However, the committee concluded that the expertise of health professional liability insurance carriers should be leveraged to improve the diagnostic process. Improved collaboration between health professional liability insurance carriers and health care professionals and organizations could help to identify resources, prioritize areas of concern, and devise interventions. Collaboration among health care professional educators and professional liability insurance carriers also could be help-
ful in developing interventions for trainees. An example of collaborative efforts among medical liability insurers and educators is the recent grant from The Doctors Company Foundation to the Society to Improve Diagnosis in Medicine (SIDM, 2015; TDCF, 2015). This grant will provide funding for diagnostic training, with a focus on clinical reasoning and methods to communicate with patients about diagnostic errors (SIDM, 2015).
FFS payment, the predominant form of payment for health care services in the United States, pays health care professionals for each service they provide. FFS payment has long been recognized for its inability to incentivize well-coordinated, high-quality, and efficient health care (Council of Economic Advisors, 2009; IOM, 2001, 2013a; National Commission on Physician Payment Reform, 2013). There is relatively little information about the impact of payment on the diagnostic process. However, the committee concluded that payment is likely to have an impact on the diagnostic process, and several payment experts who provided input to the committee helped elaborate on some of these consequences (Miller, 2014; Rosenthal, 2014; Wennberg, 2014).
In general, FFS payment may not incentivize a high-quality, efficient diagnostic process because the more services the diagnostic process entails, the more remuneration will result. There is no disincentive for ordering unnecessary diagnostic testing that could lead to false positive results and diagnostic errors (Miller, 2014; Wennberg, 2014). There is also a financial incentive to provide treatment to patients rather than determining that patients do not have health problems; thus, inappropriate diagnoses are better compensated than determining that a patient does not have a health problem. Likewise, accuracy in the diagnostic process is not explicitly rewarded by FFS payment: Clinicians who interpret diagnostic testing or provide a diagnosis during a patient visit receive payment whether or not the work was done adequately to support accurate interpretation and diagnosis and whether or not the interpretations and diagnoses are accurate (Miller, 2014).
Given the importance of team-based care in the diagnostic process, the lack of financial incentives in FFS payment to coordinate care may contribute to challenges in diagnosis and diagnostic errors, particularly delays in diagnosis (Rosenthal, 2014). FFS Medicare and most commercial payers do not pay for time that a clinician spends contacting other clinicians by phone or e-mail to facilitate the diagnostic process: for example, by helping determine the appropriate diagnostic tests for a patient. In addition, clinicians are not reimbursed for proactive outreach to patients to obtain diagnostic testing, to schedule visits with specialists, or
to make follow-up appointments (Miller, 2014). To improve teamwork and care coordination in the diagnostic process, the committee recommends that the Centers for Medicare & Medicaid Services (CMS) and other payers should create current procedural terminology (CPT) codes and provide coverage for additional evaluation and management activities not currently coded or covered, including time spent by pathologists, radiologists, and other clinicians in advising ordering clinicians on the selection, use, and interpretation of diagnostic testing for specific patients. New CPT codes can help incentivize communication and collaboration among treating clinicians and clinicians who conduct diagnostic testing in order to improve the diagnostic testing process for patients (Allen and Thorwarth, 2014; Kroft, 2014; Miller, 2014). These codes could be modeled on current CPT codes that compensate coordination and planning activities that are recognized for payment by Medicare and some other payers (e.g., CPT codes for radiation therapy planning, post-discharge transitional care coordination, and complex chronic care coordination) (AAFP, 2013; ASTRO, 2014; Bendix, 2013; Blue Cross Blue Shield of North Carolina, 2015; CMS, 2013, 2014b; Edwards and Landon, 2014; Nicoletti, 2005; Texas Medical Association, 2013). The proposed new codes are not meant to capture every discussion among clinicians; rather they are meant to capture discrete work that does not occur routinely in normal interactions to encourage more collaborative activity in the diagnostic process.
The Medicare physician fee schedule sets payment rates based on relative value units that are meant to reflect the level of time, effort, skill, and stress associated with providing each service (MedPAC, 2014). Fee schedule services can include evaluation and management services (“E&M services,” such as office, inpatient, or emergency department visits), diagnostic testing, and other procedures. For all medical specialties, there are well-documented fee schedule distortions that result in more generous payments (in relation to the costs of production) being made for procedures and diagnostic testing interpretations than for E&M services (Berenson, 2010; National Commission on Physician Payment Reform, 2013). The existence of these distortions has coincided with a large growth in diagnostic testing in health care (see Figure 7-2); for example, the percent of patients presenting to the emergency department with dizziness who underwent computed tomography (CT) scans rose from 9 percent in 1995 to 40 percent in 2013, but this has not increased diagnoses of stroke or other neurologic diseases (Iglehart, 2009; Newman-Toker et al., 2013).
The lower relative value afforded to E&M services versus procedure-oriented care is problematic for improved diagnostic performance. E&M services reflect the cognitive expertise and skills that all clinicians have
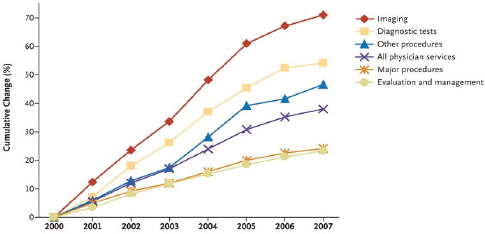
FIGURE 7-2 Rates of use of medical imaging services and diagnostic testing compared with rates of other clinician-ordered services, per Medicare Beneficiary (2000–2007).
SOURCE: J. K. Iglehart, Health insurers and medical-imaging policy—A work in progress. New England Journal of Medicine 360(10), 1030–1037. Copyright 2009 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
and use in the diagnostic process, and these distortions may be diverting attention and time from important tasks in the diagnostic process, such as performing a patient’s clinical history and interview, conducting a physical exam, and decision making in the diagnostic process. Thus, the committee recommends that CMS and other payers reorient relative value fees to more appropriately value the time spent with patients in evaluation and management activities. Realigning relative value fees to better compensate clinicians for cognitive work in the diagnostic process has the potential to improve accuracy in diagnosis while also reducing incentives that drive the inappropriate utilization of diagnostic testing.
E&M payment policies and documentation guidelines also are misaligned with the goal of accurate and timely diagnosis. E&M payments penalize clinicians for spending extra time on the diagnostic process for an individual patient. There are different levels of E&M visits based on time and complexity, and practices receive better compensation if they see more patients with shorter appointment lengths. For example, in Medicare, if a clinician spends 20 minutes with a patient who is billed as a level 3 E&M visit rather than spending just 15 minutes, the clinician’s practice will receive 25 percent less revenue per hour; if a clinician spends 25 minutes for a level 4 E&M visit instead of 15 minutes for a level 3 visit, the practice will receive 11 percent less revenue per hour (Miller, 2014).
Time pressures in clinical visits can contribute to various challenges in clinical reasoning and to the occurrence of errors (Durning, 2014; Kostis et al., 2007; Sarkar et al., 2012, 2014; Schiff et al., 2009; Singh et al., 2013). Although there is evidence that the lengths of clinical appointments have not generally declined,7 there are concerns that the rising complexity of health care, the growth in patients with complicated health conditions, and increased EHR-related tasks are contributing to increased time pressures. The aging U.S. population contributes to added complexity for patient care decisions due to the need to understand the various factors that may be contributing to an older adult’s health problem, such as multiple comorbidities and polypharmacy (IOM, 2008, 2013b). While unlimited time is neither the objective nor realistic, it is important to make time for effectively addressing these complex care decisions. Making more effective use of the time available will be critical, as will making improvements to the work system in which the diagnostic process occurs (such as disseminating an organizational culture that is supportive of teamwork in the diagnostic process, the better allocation of tasks, and ensuring that health information technology [health IT] is supportive of the diagnostic process).
In addition to modifying payment policies, the documentation guidelines for E&M services could also be improved to support the diagnostic process. Documentation guidelines for E&M services were created to ensure that the services performed were consistent with insurance coverage; to validate specific information, such as the site of service, the appropriateness of the care, and the accuracy of the reported information; and to prevent fraud and abuse (Berenson, 1999; CMS, 2014a). Documentation guidelines specify the extent of a patient’s clinical history and interview, the physical exam, and the complexity of medical decision making involved in the E&M visit (Berenson et al., 2011; HHS, 2010). There are a number of criticisms of the documentation guidelines; the primary argument is that the level of detail required is onerous, is often irrelevant to patient care, and shifts the purpose of the medical record toward billing rather than facilitating clinical reasoning (Berenson et al., 2011; Brett, 1998; Kassirer and Angell, 1998; Kuhn et al., 2015; Schiff and Bates, 2010) (see the discussion of clinical documentation in Chapter 5).
The documentation guidelines have become an even greater concern with the broad implementation of EHRs because EHR design has focused on fulfilling documentation and legal requirements and not on facilitating the diagnostic process (Berenson et al., 2011; Schiff and Bates, 2010). EHRs
______________
7 For example, the National Ambulatory Medical Care Survey found that in 1992 most visits lasted 15 minutes or less; by 2010, only half of clinician visits were that short (Rabin, 2014).
tend to lack a cohesive patient narrative, which include nuance, details, and important contextual information that help clinicians make accurate and timely diagnoses. The orientation of EHRs to documentation, their overreliance on templates, and the copy and paste functionalities within EHRs have resulted in “EHR-generated data dumps, including repetitive documentation of elements of patients’ histories and physical examinations, that merely result in electronic versions of clinically cumbersome, uninformative patient records” (Berenson et al., 2011, p. 1894). Generating documentation to support E&M coding (or higher levels of E&M coding than are warranted, which is called “upcoding”) can result in inaccuracies in the patient’s EHR that can contribute to diagnostic errors.
A number of payment and care delivery reforms aimed at countering the limitations of the FFS payment system are actively being considered, implemented, and evaluated (see Box 7-2). These include capitation/global payments, shared savings, bundled episodes of care, accountable care organizations, patient-centered medical homes, and pay for performance (which Medicare refers to as “value-based purchasing”). Box 7-2 includes both potential benefits of new payment models on improving diagnosis as well as some of the potential drawbacks (see also Himmelstein and Woolhandler [2014] for a discussion of the potential limitations of new payment models). Salary is not described as a payment model because the committee focused on third-party payments rather than provider organization compensation.
CMS recently announced that it plans to have 30 percent of Medicare payments based on alternative models by the end of 2016 and 50 percent of payments by the end of 2018 (Burwell, 2015). The Medicare Access and CHIP Reauthorization Act of 2015 (which repealed the sustainable growth rate) continues down the path toward alternative payment models, particularly for the payment of Medicare clinicians.8 While the impact of alternative payment and delivery systems on quality are actively being investigated (e.g., the Blue Cross Blue Shield of Massachusetts Alternative Quality Contract, as well as patient-centered medical homes), there is very limited evidence on what impact such payment and delivery models will have on the diagnostic process and on the accuracy of diagnosis, and this represents a fundamental research need. Thus, the committee recommends that CMS and other payers should assess the impact of payment and care delivery models on the diagnostic process, the occurrence of diagnostic errors, and learning from these errors. Assessing the impact of payment and care delivery models, including FFS, on the diagnostic process, diagnostic errors, and learning are critical areas of focus as these models are evaluated more broadly. CMS’s Innovation Center is testing
______________
8 Medicare Access and CHIP Reauthorization Act of 2015. P.L. 114-10. (April 16, 2015).
BOX 7-2
Payment and Care Delivery Reforms and Their Potential Impact on Diagnosis
-
Global Payment, Capitation, and Per-Member Per-Month
- Definition: “A single per-member per-month payment is made for all services delivered to a patient, with payment adjustments based on measured performance and patient risk” (Schneider et al., 2011, p. 13).
- Potential impact on diagnosis: Broader adoption could enhance provider activities that improve diagnostic accuracy and reduce diagnostic errors because the capitated, at-risk organization bears the cost of diagnostic error if there are immediate costs associated with the error. For diagnostic errors that do not necessarily lead to higher costs for the organization, investment in lowering these errors (e.g., more vigilant evidence-based cancer screening which could increase costs due to treatment of newfound cancers) may be suboptimal. The use of quality measures and reporting may incentivize organizations to detect the underuse of these screening activities, to reengineer care, to invest in electronically based decision support and artificial intelligence which could improve accuracy, to engage clinicians in ongoing activities to improve diagnostic skills, and to engage in systems approaches to mitigating harm from potential diagnostic errors.
-
Accountable Care Organizations
- Definition: “Groups of providers that voluntarily assume responsibility for the care of a population of patients” (Schneider et al., 2011, p. 13).
- Potential impact on diagnosis: The quality of care in accountable care organizations (ACOs) is assessed through a set of quality measures, but none of them involve accuracy or timeliness of diagnosis, for the reasons described in Chapter 3. ACOs have the potential infrastructure to provide a base of activity to improve diagnostic accuracy for their constituent or affiliated clinicians. So far, most ACOs do not accept risk, so the potential of non-fee-for-service financial incentives has not yet been realized. Nevertheless, the structure of an ACO and its need to credential its members and engage in quality and safety improvement programs can provide a new source of interest and provider expertise in engaging in the problem of diagnostic errors. To date, payers have not determined that diagnostic errors are a priority quality and safety problem that needs attention. ACOs, for example, would be well positioned to administer and promote follow-up and feedback approaches and to develop a culture in which these approaches are welcomed and routine.
-
Bundled Payment or Episode-Based Payment
- Definition: A “single ‘bundled’ payment, which may include multiple providers in multiple care settings, is made for services delivered during an episode of care related to a medical condition or procedure” (Schneider et al., 2011, p. 13).
-
- Potential impact on diagnosis: By definition, bundled payment would seem to apply mostly to welle-stablished, “correct” diagnoses, for which efficiencies of care can be further gained, and it remains volume-based (i.e., the financial incentive is to produce more, efficiently provided episodes). This raises the importance of addressing appropriateness of the bundled episode procedure being performed. Appropriateness is relevant to the topic of diagnostic error in the sense of needing to determine acuity of the condition as part of the diagnostic process. For chronic conditions, episode-based payment runs the risk of non-holistic care. For example, the clinicians receiving the episode-based payment for a condition such as diabetes may not be as attuned to diagnosis and management of comorbidities that may arise in the course of management of the index condition.
-
Pay for Performance or Value-Based Purchasing
- Definition: “[P]hysicians receive differential payments for meeting or missing performance benchmarks” (Schneider et al., 2011, p. 14).
- Potential impact on diagnosis: Theoretically this can be a useful payment tool for focusing provider attention on important quality problems that can be measured accurately and then financially rewarded and penalized. Overall, the effects of pay for performance on outcomes remain unsettled, with concerns about the effects on important elements of care that are not being measured. Current pushes for accountability neglect performance measures for diagnosis, and that is a major limitation of these approaches.
-
Patient-Centered Medical Homes
- Definition: “[A] physician practice or other provider is eligible to receive additional payments if medical home criteria are met. Payment may include calculations based on quality and cost performance using a [pay for performance]-like mechanism” (Schneider et al., 2011, p. 13). Although not an inherent part of the definition, most medical home initiatives are taking place in primary care practices.
many of the alternative payment models and is well suited to evaluate the impact of these models on the diagnostic process and the occurrence of diagnostic errors.
While new payment models have the potential to reduce diagnostic errors, the committee also recognized that these models may also create incentives for clinicians and health care organizations that could reduce use of appropriate testing and clinician services (e.g., specialty consultations) that may inadvertently lead to greater diagnostic errors. To address these possibilities, the committee recognized that not only is direct evaluation of the impact of payment models on diagnostic errors important but
-
- Potential impact on diagnosis: A well-functioning medical home, teamwork, longstanding relationships with patients as the center for care and care coordination, and ultimately, reliance on improved electronic health records and interoperability of patient information to inform clinical decision making has the potential to improve diagnostic performance. There are concerns, however, that medical home performance will be assessed using measures that do not include those related to diagnostic performance, although it is known that there is a significant problem of diagnostic error in primary care (Ely et al., 2012; Singh et al., 2013).
-
Shared Savings
- Definition: “[A] payment strategy that offers incentives for providers to reduce health care spending for a defined patient population by offering them a percentage of net savings realized as a result of their efforts” (Bailit and Hughes, 2011, p. 1).
- Potential impact on diagnosis: As a payment method, there are no direct incentives to focus on improving diagnostic accuracy. The impact depends largely on the objectives of the underlying organization to which the payment is being applied. For example, shared savings has become the primary method for rewarding ACOs for spending less than a target spending amount. Theoretically, at least, the ACO should be interested in diagnostic accuracy if by getting the diagnosis correct, subsequent spending can be promptly reduced. So the focus would be on efforts to make correct diagnoses of acute, urgent presentations of illness in emergency departments and primary care practices and for commonly misdiagnosed conditions such as stroke and congestive heart failure. Conversely, based on incentives alone, the organization might be less interested in efforts to make accurate and timely diagnoses of conditions whose costs would not be borne for many months or years. To date, there seems to be little attention paid to diagnostic accuracy as a mechanism for achieving savings.
also there is a need for better measurement tools to identify diagnostic errors in clinical practice (see Chapters 5 and 6).
Additionally, the committee asked for input from payment and delivery experts about the potential effects of new models on diagnosis and diagnostic error. Rosenthal (2014) suggested that global payment and meaningful use incentives have the potential to improve diagnosis by promoting the adoption of diagnostic test and referral tracking systems that better connect health care professionals throughout the continuum of care. Miller (2014) suggested that the development of measures for diagnostic accuracy be developed to provide feedback and reward clinicians
for diagnostic accuracy. Wennberg (2014) suggested that population-based payment models, including capitation and global budgets, have the greatest potential to reduce diagnostic errors.
Even when alternate payment and care delivery approaches to FFS are employed, they are often based on or influenced by existing coding and payment rules (Berenson et al., 2011). For example, bundled payments are combinations of current codes. Thus, the current distortions in the fee schedule and other volume-based payment approaches, such as diagnosis-related group coding, will remain a dominant component of payment and care delivery models in the near future and need to be addressed. As long as fee schedules remain a predominant mechanism for determining clinician payment, the committee recommends that CMS and other payers should modify documentation guidelines for evaluation and management services to improve the accuracy of information in the EHR and to support decision making in the diagnostic process.
Goal 6: Develop a reporting environment and medical liability system that facilitates improved diagnosis by learning from diagnostic errors and near misses
Recommendation 6a: The Agency for Healthcare Research and Quality (AHRQ) or other appropriate agencies or independent entities should encourage and facilitate the voluntary reporting of diagnostic errors and near misses.
Recommendation 6b: AHRQ should evaluate the effectiveness of patient safety organizations (PSOs) as a major mechanism for voluntary reporting and learning from these events and modify the PSO Common Formats for reporting of patient safety events to include diagnostic errors and near misses.
Recommendation 6c: States, in collaboration with other stakeholders (health care organizations, professional liability insurance carriers, state and federal policy makers, patient advocacy groups, and medical malpractice plaintiff and defense attorneys), should promote a legal environment that facilitates the timely identification, disclosure, and learning from diagnostic errors. Specifically, they should:
- Encourage the adoption of communication and resolution programs with legal protections for disclosures and apologies under state laws.
- Conduct demonstration projects of alternative approaches to the resolution of medical injuries, including administrative health courts and safe harbors for adherence to evidence-based clinical practice guidelines.
Recommendation 6d: Professional liability insurance carriers and captive insurers should collaborate with health care professionals on opportunities to improve diagnostic performance through education, training, and practice improvement approaches and increase participation in such programs.
Goal 7: Design a payment and care delivery environment that supports the diagnostic process
Recommendation 7a: As long as fee schedules remain a predominant mechanism for determining clinician payment, the Centers for Medicare & Medicaid Services (CMS) and other payers should:
- Create current procedural terminology codes and provide coverage for additional evaluation and management activities not currently coded or covered, including time spent by pathologists, radiologists, and other clinicians in advising ordering clinicians on the selection, use, and interpretation of diagnostic testing for specific patients.
- Reorient relative value fees to more appropriately value the time spent with patients in evaluation and management activities.
- Modify documentation guidelines for evaluation and management services to improve the accuracy of information in the electronic health record and to support decision making in the diagnostic process.
Recommendation 7b: CMS and other payers should assess the impact of payment and care delivery models on the diagnostic process, the occurrence of diagnostic errors, and learning from these errors.
AAFP (American Association of Family Physicians). 2013. Frequently asked questions: Transitional care management. www.aafp.org/dam/AAFP/documents/practice_management/payment/TCMFAQ.pdf (accessed June 8, 2015).
ACP (American College of Physicians). 2006. Exploring the use of health courts—Addendum to “Reforming the medical professional liability system.” www.acponline.org/acp_policy/policies/health_courts_reform_medical_liability_2006.pdf (accessed May 24, 2015).
ACP. 2014. Medical liability reform: Innovative solutions for a new health care system: A position paper. Philadelphia: American College of Physicians. www.acponline.org/acp_policy/policies/medical_liability_reform_2014.pdf (accessed June 8, 2015).
ACS (American College of Surgeons). 2015. Statement on medical liability. http://bulletin.facs.org/2015/03/statement-on-medical-liability-reform (accessed May 16, 2015).
AHRQ (Agency for Healthcare Research and Quality). 2014. Medical liability reform and patient safety initiative. www.ahrq.gov/professionals/quality-patient-safety/patient-safety-resources/resources/liability (accessed April 10, 2015).
AHRQ. 2015a. Agency for Healthcare Research and Quality: Justification of estimates for appropriations committees. www.ahrq.gov/sites/default/files/wysiwyg/cpi/about/mission/budget/2016/cj2016.pdf (accessed May 3, 2015).
AHRQ. 2015b. Common formats. www.pso.ahrq.gov/common (accessed March 28, 2015).
AHRQ. 2015c. Federally listed PSOs. www.pso.ahrq.gov/listed (accessed May 3, 2015).
AHRQ. 2015d. Medical liability reform and patient safety initiative progress report. www.ahrq.gov/professionals/quality-patient-safety/patient-safety-resources/resources/liability/medliabrep.html (accessed May 24, 2015).
AHRQ. 2015e. Patient safety organization (PSO) program: Frequently asked questions. www.pso.ahrq.gov/faq#WhatisaPSO (accessed May 3, 2015).
Allen, B., and W. T. Thorworth. 2014. Comments from the American College of Radiology. Input submitted to the Committee on Diagnostic Error in Health Care, November 5 and December 29, 2014, Washington, DC.
ASTRO (American Society for Radiation Oncology). 2014. Basics of RO coding. www.astro.org/Practice-Management/Reimbursement/Basics-of-RO-Coding.aspx (accessed March 26, 2015).
Bailit, M., and C. Hughes. 2011. Key design elements of shared-savings payment arrangements. The Commonwealth Fund Publication No. 1539. www.commonwealthfund.org/~/media/Files/Publications/Issue%20Brief/2011/Aug/1539_Bailit_key_design_elements_sharedsavings_ib_v2.pdf (accessed June 8, 2015).
Barach, P., and S. D. Small. 2000. Reporting and preventing medical mishaps: Lessons from non-medical near miss reporting systems. BMJ 320(7237):759–763.
Bendix, J. 2013. Making sense of the new transitional care codes. Medical Economics, March 10. http://medicaleconomics.modernmedicine.com/medical-economics/news/user-defined-tags/99495/making-sense-new-transitional-care-codes?page=full (accessed March 26, 2015).
Berenson, R. A. 1999. Evaluation and management guidelines. New England Journal of Medicine 340(11):889–891.
Berenson, R. A. 2005. Malpractice makes perfect. The New Republic, October 10. www.newrepublic.com/article/health-care-malpractice-bush-frist (accessed November 7, 2015).
Berenson, R. A. 2010. Out of whack: Pricing distortions in the medicare physician fee schedule. Expert Voices, September. www.nihcm.org/pdf/NIHCM-EV-Berenson_FINAL.pdf (accessed June 8, 2015).
Berenson, R. A., P. Basch, and A. Sussex. 2011. Revisiting E&M visit guidelines—A missing piece of payment reform. New England Journal of Medicine 364(20):1892–1895.
Blue Cross Blue Shield of North Carolina. 2015. Corporate medical policy: E-visits (online medical evaluations). www.bcbsnc.com/assets/services/public/pdfs/medicalpolicy/evisits_online_medical_evaluation.pdf (accessed June 8, 2015).
Boothman, R. C., A. C. Blackwell, D. A. Campbell Jr., E. Commiskey, and S. Anderson. 2009. A better approach to medical malpractice claims? The University of Michigan experience. Journal of Health & Life Sciences Law 2(2):125–159.
Boothman, R. C., S. J. Imhoff, and D. A. Campbell, Jr. 2012. Nurturing a culture of patient safety and achieving lower malpractice risk through disclosure: Lessons learned and future directions. Frontiers of Health Services Management 28(3):13–28.
Bovbjerg, R. R. 2010. Will the Patient Protection and Affordable Care Act address problems associated with medical malpractice? Urban Institute. www.urban.org/research/publication/will-patient-protection-and-affordable-care-act-address-problems-associated-medical-malpractice (accessed May 3, 2015).
Bovbjerg, R. R., and R. Berenson. 2012. The value of clinical practice guidelines as malpractice “safe harbors.” Urban Institute. www.rwjf.org/content/dam/farm/reports/issue_briefs/2012/rwjf72667 (accessed June 8, 2015).
Bovbjerg, R. R., and F. A. Sloan. 1998. No fault for medical injury: Theory and evidence. University of Cincinnati Law Review 67. http://ssrn.com/abstract=147016 (accessed June 6, 2015).
Brett, A. S. 1998. New guidelines for coding physicians’ services—A step backward. New England Journal of Medicine 339(23):1705–1708.
Burwell, S. M. 2015. Setting value-based payment goals—HHS efforts to improve U.S. health care. New England Journal of Medicine 372(10):897–899.
CFPS (Center for Patient Safety). 2015. National health reform provisions and PSOs. www.centerforpatientsafety.org/nationalhealthreformprovisionsandpsos (accessed November 7, 2015).
Chow, E. 2007. Health courts: An extreme makeover of medical malpractice with potentially fatal complications. Yale Journal of Health Policy, Law, and Ethics 7(2):387–427.
CMS (Centers for Medicare & Medicaid Services). 2013. Telehealth services. Department of Health and Human Services. www.cms.gov/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNProducts/downloads/TelehealthSrvcsfctsht.pdf (accessed June 8, 2015).
CMS. 2014a. Evaluation and management services guide: Official CMS information for Medicare fee-for-service providers. Washington, DC: Centers for Medicare & Medicaid Services.
CMS. 2014b. Policy and payment changes to the Medicare physician fee schedule for 2015. www.cms.gov/newsroom/mediareleasedatabase/fact-sheets/2014-Fact-sheets-items/2014-10-31-7.html (accessed March 26, 2015).
Council of Economic Advisors. 2009. The economic case for health care reform. www.whitehouse.gov/assets/documents/CEA_Health_Care_Report.pdf (accessed March 17, 2015).
CRICO. 2014. 2014 annual benchmarking report: Malpractice risks in the diagnostic process. CRICO Strategies. www.rmfstrategies.com/benchmarking (accessed June 8, 2015).
Delbanco, T., and S. K. Bell. 2007. Guilty, afraid, and alone—struggling with medical error. New England Journal of Medicine 357(17):1682–1683.
Donohue, G. 2014. PRI medical liability insurance. Input submitted to the Committee on Diagnostic Error in Health Care, November 25, 2014, Washington, DC.
Durning, S. J. 2014. Input submitted to the Committee on Diagnostic Error in Health Care, October 25, 2014, Washington, DC.
Edwards, S. T., and B. E. Landon. 2014. Medicare’s chronic care management payment—Payment reform for primary care. New England Journal of Medicine 371(22):2049–2051.
Ely, J. W., L. C. Kaldjian, and D. M. D’Alessandro. 2012. Diagnostic errors in primary care: Lessons learned. Journal of the American Board on Family Medicine 25(1):87–97.
Evans, W. N., and B. Kim. 2006. Patient outcomes when hospitals experience a surge in admissions. Journal of Health Economics 25(2):365–388.
Gallagher, T. H., C. Denham, L. Leape, G. Amori, and W. Levinson. 2007. Disclosing unanticipated outcomes to patients: The art and practice. Journal of Patient Safety 3(3):158–165.
Gallagher, T. H., M. M. Mello, W. Levinson, M. K. Wynia, A. K. Sachdeva, L. Snyder Sulmasy, R. D. Truog, J. Conway, K. Mazor, A. Lembitz, S. K. Bell, L. Sokol-Hessner, J. Shapiro, A.-L. Puopolo, and R. Arnold. 2013. Talking with patients about other clinicians’ errors. New England Journal of Medicine 369(18):1752–1757.
GAO (Government Accountability Office). 2010. Patient Safety Act: HHS is in the process of implementing the act, so its effectiveness cannot yet be evaluated. Washington, DC: Government Accountability Office.
Helmchen, L. A., M. R. Richards, and T. B. McDonald. 2010. How does routine disclosure of medical error affect patients’ propensity to sue and their assessment of provider quality? Evidence from survey data. Medical Care 48(11):955–961.
Hendrich, A., C. K. McCoy, J. Gale, L. Sparkman, and P. Santos. 2014. Ascension health’s demonstration of full disclosure protocol for unexpected events during labor and delivery shows promise. Health Affairs 33(1):39–45.
HHS (Department of Health and Human Services). 2010. Improper payments for evaluation and management services cost Medicare billions in 2010. Washington, DC: HHS Office of Inspector General.
HHS. 2014. Decision on appropriate medical malpractice payment reporting. www.citizen.org/documents/2211%20Enclosure.pdf (accessed May 16, 2015).
HHS. 2015. Patient safety and quality improvement act of 2005 statute and rule. www.hhs.gov/ocr/privacy/psa/regulation (accessed March 29, 2015).
Himmelstein, D. U., and S. Woolhandler. 2014. Global Amnesia: Embracing Fee-For-Non-Service—Again. Journal of General Internal Medicine 29(5):693.
Hoffman, J. R., and H. K. Kanzaria. (2014). Intolerance of error and culture of blame drive medical excess. BMJ 349:g5702.
Howard, P. 2012. The growing bipartisan support for health courts. Health Affairs Blog, October 2. http://healthaffairs.org/blog/2012/10/02/the-growing-bipartisan-support-for-health-courts (accessed June 8, 2015).
Howard, P., and R. G. Maine. 2013. Health courts may be best cure for what ails the liability system. Bulletin of the American College of Surgeons, March 2. http://bulletin.facs.org/2013/03/health-courts-best-cure (accessed June 8, 2015).
IEA (International Ergonomics Association). 2000. The discipline of ergonomics. www.iea.cc/whats/index.html (accessed April 10, 2015).
Iglehart, J. K. 2009. Health insurers and medical-imaging policy—A work in progress. New England Journal of Medicine 360(10):1030–1037.
IOM (Institute of Medicine). 2000. To err is human: Building a safer health system. Washington, DC: National Academy Press.
IOM. 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
IOM. 2002. Fostering rapid advances in health care: Learning from system demonstrations. Washington, DC: National Academy Press.
IOM. 2008. Retooling for an aging America: Building the health care workforce. Washington, DC: The National Academies Press.
IOM. 2011. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
IOM. 2012. Health IT and patient safety: Building safer systems for better care. Washington, DC: The National Academies Press.
IOM. 2013a. Best care at lower cost: The path to continuously learning health care in America. Washington, DC: The National Academies Press.
IOM. 2013b. Delivering high-quality cancer care: Charting a new course for a system in crisis. Washington, DC: The National Academies Press.
The Joint Commission. 2005. Health care at the crossroads: Strategies for improving the medical liability system and preventing patient injury. The Joint Commission. www.jointcommission.org/assets/1/18/Medical_Liability.pdf (accessed June 8, 2015).
Jost, T. 2006. Health courts and malpractice claims adjudication through Medicare: Some questions. Journal of Health Care Law & Policy 9(2):280–290.
Kachalia, A. 2014. Legal issues in diagnostic error. Presentation to the Committee on Diagnostic Error in Health Care, August 7, 2014, Washington, DC.
Kachalia, A., and M. M. Mello. 2011. New directions in medical liability reform. New England Journal of Medicine 364(16):1564–1572.
Kachalia, A. B., M. M. Mello, T. A. Brennan, and D. M. Studdert. 2008. Beyond negligence: Avoidability and medical injury compensation. Social Science and Medicine 66(2):387–402.
Kachalia, A., S. R. Kaufman, R. Boothman, S. Anderson, K. Welch, S. Saint, M. A. Rogers. 2010. Liability claims and costs before and after implementation of a medical error disclosure program. Annals of Internal Medicine 153(4):213–221.
Kachalia, A., A. Little, M. Isavoran, L. M. Crider, and J. Smith. 2014. Greatest impact of safe harbor rule may be to improve patient safety, not reduce liability claims paid by physicians. Health Affairs (Millwood) 33(1):59–66.
Kassirer, J. P., and M. Angell. 1998. Evaluation and management guidelines—Fatally flawed. New England Journal of Medicine 339(23):1697–1698.
Kessler, D. P., N. Summerton, and J. R. Graham. 2006. Effects of the medical liability system in Australia, the UK, and the USA. Lancet 368(9531):240–246.
Kostis, W. J., K. Demissie, S. W. Marcella, Y. H. Shao, A. C. Wilson, and A. E. Moreyra. 2007. Weekend versus weekday admission and mortality from myocardial infarction. New England Journal of Medicine 356(11):1099–1109.
Kroft, S. H. 2014. Statement of Steven H. Kroft, MD, FASCP, American Society for Clinical Pathology (ASCP). Input submitted to the Committee on Diagnostic Error in Health Care, April 28, 2014, Washington, DC.
Kuhn, T., P. Basch, M. Barr, and T. Yackel. 2015. Clinical documentation in the 21st century: Executive summary of a policy position paper from the American College of Physicians. Annals of Internal Medicine 162(4):301–303.
Lembitz, A., and D. Boyle. 2014. COPIC medical liability insurance. Input submitted to the Committee on Diagnostic Error in Health Care, November 25, 2014, Washington, DC.
Localio, A. R., A. G. Lawthers, T. A. Brennan, N. M. Laird, L. E. Hebert, L. M. Peterson, J. P. Newhouse, P. C. Weiler, and H. H. Hiatt. 1991. Relation between malpractice claims and adverse events due to negligence. New England Journal of Medicine 325(4):245–251.
Lopez, L., J. S. Weissman, E. C. Schneider, S. N. Weingart, A. P. Cohen, and A. M. Epstein. 2009. Disclosure of hospital adverse events and its association with patients’ ratings of the quality of care. Archives of Internal Medicine 169(20):1888–1894.
MACRMI (Massachusetts Alliance for Communication and Resolution following Medical Injury). 2015. About MACRMI. www.macrmi.info/about-macrmi/#sthash.wpQ4E35l.dpbs (accessed November 7, 2015).
Mastroianni, A. C., M. M. Mello, S. Sommer, M. Hardy, and T. H. Gallagher. 2010. The flaws in state “apology” and “disclosure” laws dilute their intended impact on malpractice suits. Health Affairs 29(9):1611–1619.
MedPAC (Medicare Payment Advisory Commission). 2014. Physician and other health professional payment systems. www.medpac.gov/-documents-/payment-basics/page/2 (accessed March 17, 2015).
Mello, M. M., and T. H. Gallagher. 2010. Malpractice reform—Opportunities for leadership by health care institutions and liability insurers. New England Journal of Medicine 362(15):1353–1356.
Mello, M. M., D. M. Studdert, A. B. Kachalia, and T. A. Brennan. 2006. “Health courts” and accountability for patient safety. Milbank Quarterly 84(3):459–492.
Mello M., A. Chandra, A. Gawande, and D. Studdert. 2010. National costs of the medical liability system. Health Affairs, 29(9):1569–1577.
Mello, M. M., A. Kachalia, and D. Studdert. 2011. Administrative compensation for medical injuries: Lessons from three foreign systems. Commonwealth Fund.
Mello, M. M., R. C. Boothman, T. McDonald, J. Driver, A. Lembitz, D. Bouwmeester, B. Dunlap, and T. Gallagher. 2014a. Communication-and-resolution programs: The challenges and lessons learned from six early adopters. Health Affairs 33(1):20–29.
Mello, M. M., D. M. Studdert, and A. Kachalia. 2014b. The medical liability climate and prospects for reform. JAMA 312(20):2146–2155.
MHA PSO (Michigan Health Hospital Association Patient Safety Organization). 2015. Safe tables. www.mhapso.org/safetables (accessed April 10, 2015).
Miller, H. D. 2014. How healthcare payment systems and benefit designs can support more accurate diagnosis. Input submitted to the Committee on Diagnostic Error in Health Care, December 29, 2014, Washington, DC.
National Commission on Physician Payment Reform. 2013. Report of the National Commission on Physician Payment Reform. http://physicianpaymentcommission.org/report (accessed March 17, 2015).
National Patient Safety Foundation’s Lucian Leape Institute. 2015. Shining a Light: Safer Health Care Through Transparency. http://c.ymcdn.com/sites/www.npsf.org/resource/resmgr/LLI/Shining-a-Light_Transparency.pdf (accessed July 24, 2015).
Newman-Toker, D. E., K. M. McDonald, and D. O. Meltzer. 2013. How much diagnostic safety can we afford, and how should we decide? A health economics perspective. BMJ Quality and Safety 22(Suppl 2):ii11–ii20.
Nicoletti, B. 2005. How to document and bill care plan oversight. Family Practice Management 12(5):23–25.
ONC (Office of the National Coordinator for Health Information Technology). 2014. Health information technology adverse event reporting: Analysis of two databases. Washington, DC: Office of the National Coordinator for Health Information Technology.
OTA (Office of Technology Assessment). 1994. Defensive Medicine and Medical Malpractice. OTA-H-602. Washington, DC: U.S. Government Printing Office.
Parikh, P. D. 2014. PIAA medical liability insurance. Input submitted to the Committee on Diagnostic Error in Health Care, February 25, 2014, Washington, DC.
Peters, P. G. 2008. Health courts? Boston University Law Review 88:227–289.
Rabin, R. C. 2014. 15-minute visits take a toll on the doctor–patient relationship. Kaiser Health News, April 21. http://khn.org/news/15-minute-doctor-visits (accessed June 8, 2015).
Robeznieks, A. 2014. Malpractice claims can be kept out of court, but not the NPDB. Modern Healthcare, August 13. www.modernhealthcare.com/article/20140813/NEWS/308139939 (accessed June 8, 2015).
Rosenthal, M. 2014. Comments to the Institute of Medicine. Input submitted to the Committee on Diagnostic Error in Health Care, December 29, 2014, Washington, DC.
Sage, W. M., T. H. Gallagher, S. Armstrong, J. S. Cohn, T. McDonald, J. Gale, A. C. Woodward, and M. M. Mello. 2014. How policy makers can smooth the way for communication-and-resolution programs. Health Affairs 33(1):11–19.
Sarkar, U., D. Bonacum, W. Strull, C. Spitzmueller, N. Jin, A. Lopez, T. D. Giardina, A. N. Meyer, and H. Singh. 2012. Challenges of making a diagnosis in the outpatient setting: A multi-site survey of primary care physicians. BMJ Quality and Safety 21(8):641–648.
Sarkar, U., B. Simchowitz, D. Bonacum, W. Strull, A. Lopez, L. Rotteau, and K. G. Shojania. 2014. A qualitative analysis of physician perspectives on missed and delayed outpatient diagnosis: The focus on system-related factors. Joint Commission Journal on Quality and Patient Safety 40(10):461–470.
Schiff, G. D., and D. W. Bates. 2010. Can electronic clinical documentation help prevent diagnostic errors? New England Journal of Medicine 362(12):1066–1069.
Schiff, G. D., O. Hasan, S. Kim, R. Abrams, K. Cosby, B. L. Lambert, A. S. Elstein, S. Hasler, M. L. Kabongo, N. Krosnjar, R. Odwazny, M. F. Wisniewski, and R. A. McNutt. 2009. Diagnostic error in medicine: Analysis of 583 physician-reported errors. Archives of Internal Medicine 169(20):1881–1887.
Schiff, G., P. Griswold, B. R. Ellis, A. L. Puopolo, N. Brede, H. R. Nieva, F. Frederico, N. Leydon, J. Ling, D. Wachenheim, L. L. Leape, and M. Biondolillo. 2014. Doing right by our patients when things go wrong in the ambulatory setting. Joint Commission Journal on Quality and Patient Safety/Joint Commission Resources 40(2):91–96.
Schneider, E. C., P. S. Hussey, and C. Schnyer. 2011. Payment reform: Analysis of models and performance measurement implications. Santa Monica, CA: RAND Corporation.
SIDM (Society to Improve Diagnosis in Medicine). 2015. $200K Breakthrough Grant Elevates Diagnostic Training. www.improvediagnosis.org/blogpost/950784/221522/200K-Breakthrough-Grant-Elevates-Diagnostic-Training (accessed July 22, 2015).
Singh, H., T. D. Giardina, A. N. Meyer, S. N. Forjuoh, M. D. Reis, and E. J. Thomas. 2013. Types and origins of diagnostic errors in primary care settings. JAMA Internal Medicine 173(6):418–425.
TDCF (The Doctors Company Foundation). 2015. Grants awarded. www.tdcfoundation.com/Grants/index.htm (accessed July 22, 2015).
Tehrani, A., H. Lee, S. Mathews, A. Shore, M. Makary, P. Pronovost, and D. Newman-Toker. 2013. 25-year summary of U.S. malpractice claims for diagnostic errors, 1986–2010: An analysis from the National Practitioner Data Bank. BMJ Quality and Safety 22:672–680.
Texas Medical Association. 2013. Coding for telephone consultations. www.texmed.org/template.aspx?id=5422 (accessed April 10, 2015).
Timm, N. T. 2010. From damages caps to health courts: Continuing progress in medical malpractice reform. Michigan State Law Review 2010:1211–1236.
Troxel, D. 2014. The Doctors Company. Input submitted to the Committee on Diagnostic Error in Health Care, April 28, 2014, Washington, DC.
Wennberg, D. 2014. Comments for the Institute of Medicine’s Committee on Diagnostic Error in Health Care. Input submitted to the Committee on Diagnostic Error in Health Care, December 29, 2014, Washington, DC.
WHO (World Health Organization). 2005. WHO draft guidelines for adverse event reporting and learning systems. Geneva, Switzerland: World Health Organization.
WSHA (Washington State Hospital Association). 2014. WSHA safe tables and webcasts. www.wsha.org/0518.cfm (accessed April 10, 2015).
This page intentionally left blank.




































