3
The Elimination of Hepatitis C
In its deliberations, the committee analyzed the problem of hepatitis C in the United States and concluded that eliminating the public health problem of this disease is feasible with certain conditions. This chapter will lay out the committee’s logic in coming to this conclusion. First, the chapter describes the epidemiology and natural history of hepatitis C virus (HCV), followed by a brief statement on the committee’s conclusion regarding elimination. Next, it discusses ending transmission of HCV and eliminating infection in chronic hepatitis C patients. The following section describes the challenges of preventing deaths among people with chronic infection. The last section presents critical factors that would influence the success of any HCV elimination program. A discussion of the strategies that might be employed to eliminate hepatitis C is outside the scope of this report, but can be expected from phase two of this project.
EPIDEMIOLOGY OF HEPATITIS C
In 1989, a small enveloped RNA virus was discovered to be the cause of hepatitis C, a transfusion-associated condition that had until then been called non-A non-B hepatitis (after etiologic exclusion of hepatitis A virus and hepatitis B virus [HBV]) (Choo et al., 1989). HCV infects 2.5-4.7 million people in the United States, and more than 185 million people worldwide (Edlin et al., 2015; Mohd Hanafiah et al., 2013). In addition to contaminated blood transfusions, HCV is primarily spread from person to person by injection drug use, and, less commonly, through sex (particularly
among HIV positive men who have sex with men) or from mother to child (Ray and Thomas, 2015).
Most HCV infected people are unaware of their condition. About one-third of infections resolve spontaneously in the first year, the rest continue and become chronic (Cox et al., 2005; Mosley et al., 2005). Both people with resolved infection and those with chronic infection test positive for antibodies to HCV, whereas only those with chronic hepatitis C have consistently detectable HCV RNA in the blood (a test often called viral load) at a level of about 3.2 million IU/mL with 96 percent having a viral load greater than 400,000 IU/mL (Afdhal et al., 2014a; Ferenci et al., 2014; Kowdley et al., 2014; Thomas et al., 2000b; Zeuzem et al., 2014). Chronic HCV infection can progress to end-stage liver disease, including liver cancer. HCV can also cause serious problems in organ systems other than the liver, including cryoglobulinemic vasculitis, metabolic bone disease, kidney disease, cardiovascular disease, and hematologic malignancies (Cacoub et al., 2015; Lee et al., 2012). In the United States, HCV is the leading cause of end-stage liver disease requiring liver transplantation, and mortality attributed to HCV is expected to continue rising during the next 10 years (Luu, 2015; McNamara et al., 2014). Though effective treatment is now available, its impact will be muted without expanded measures for screening, disease surveillance, enrolling and retaining patients in care, and patient management (Durham et al., 2016).
The Hepatitis C Virus
In human blood, HCV exists as a lipo-viro-particle containing cholesterol-laden lipoproteins. The lipoproteins facilitate the virus binding to low-density lipoprotein receptors on liver cells, followed by more specific interactions between the viral envelope proteins with an array of cell surface proteins (Andre et al., 2002; Ray et al., 2013). Internalization of the virion is followed by release of the viral RNA genome into the cytoplasm where it functions as a messenger RNA. This viral RNA is translated by ribosomes to produce HCV’s polyprotein, which is cleaved by cellular and viral proteases (e.g., NS3/NS4A protease) to form viral proteins that manufacture viral RNA genomes (via the NS5B RNA polymerase) and reprogram the cell’s lipid export pathway to assemble infectious HCV particles (dependent on NS5A phosphoprotein), completing the cycle.
HCV’s unique life cycle offers several targets for treatment and prevention of infection. The NS3/NS4A protease, the NS5B RNA polymerase, and the NS5A phosphoprotein are essential, and each of these is targeted by one or more of the direct-acting antiviral drug combinations, therapies
that make cure or sustained virologic response1 possible in more than 95 percent of patients (Zoulim et al., 2015). Viral proteins are potential targets of cellular immunity, and abundant human and animal research evidence supports a critical role for T-cell responses in spontaneous resolution of acute HCV infection (Klenerman and Thimme, 2012). B-cell responses to HCV control the infection in an animal model and may have therapeutic or prophylactic potential, especially as the B-cell response tends to appear early in people who are clearing their acute infection (de Jong et al., 2014; Osburn et al., 2014). Spontaneous control of HCV, which occurs in about a third of acutely infected individuals, is associated with cellular and humoral responses that appear to protect against HCV persistence after repeated infection, further evidence that prophylactic vaccination is possible (Lauer, 2013; Osburn et al., 2010).
The Natural History of Hepatitis C Virus Infection
The National Health and Nutrition Examination Survey (NHANES) conducted between 2003 and 2010 reported that approximately 1 percent of the US population, or about 2.7 million people, are infected with chronic hepatitis C virus (HCV) (Denniston et al., 2014). This is likely an underestimate, as the NHANES survey does not include people who are incarcerated, hospitalized, homeless, in the military, or living in nursing homes—groups thought to account for at least 800,000 additional cases (Armstrong et al., 2006; CDC, 2016; Holmberg et al., 2013). More recent estimates peg the US prevalence at approximately 2.5-4.7 million people (Edlin et al., 2015). Because of the large burden of undiagnosed cases, the Centers for Disease Control and Prevention (CDC) estimates that for each new, symptomatic infection reported, an estimated 3.3 cases of symptomatic, acute hepatitis C actually occur (University of Washington, 2016). Furthermore, because of under-reporting and nonspecific symptoms, for every new, acute, symptomatic infection reported there are an estimated 12.3 actual new HCV infections in the population (Klevens et al., 2014). This ratio suggest about 17,000 people are infected every year, considerably less than the peak new infections of about 230,000 per year seen in the 1980s (CDC, 2010; Klevens et al., 2014; Smith et al., 2012). Effective screening of blood and tissue donors beginning in 1992 vastly decreased the estimated annual incidence of HCV (Alter and Houghton, 2000; Selvarajah and Busch, 2012).
___________________
1Sustained virologic response and cure are used synonymously. When interferon treatments were standard of care for hepatitis C, sustained virologic response was defined as negative viral load 24 weeks after cessation of therapy. With direct-acting antivirals, this timeframe is shortened to 12 weeks. The 12-week mark is recognized as the endpoint for cure by the Food and Drug Administration (FDA) because of the high concordance between sustained virologic response at 12 and 24 weeks (FDA, 2013).
But because of high transmission during the 1970s and 1980s, people born between 1945 and 1965 account for more than three-quarters of chronic hepatitis C in the United States (Smith et al., 2012).
Only about half of chronically HCV-infected people are aware of their condition (Volk et al., 2009). Over time, chronic HCV infection initiates a cascade of inflammation and progressive hepatic fibrosis, which can lead to cirrhosis (D’Amico et al., 2006; Ray and Thomas, 2015). The risk of liver fibrosis increases with duration of infection. Given the aging of the Baby Boomers, by 2020 there should be about one million cases of cirrhosis attributable to chronic hepatitis C in the United States (Davis et al., 2010).
The most recent models suggest that the 2,138 confirmed, acute HCV infections reported to the CDC in 2013 indicate 29,700 incident cases, a 180 percent increase over 2011 estimates (CDC, 2015). Such estimates may be artificially low (Onofrey et al., 2015). As with HBV, surveillance for HCV in the United States is limited; nine states and the District of Columbia do not report acute cases to the CDC (CDC, 2013). There is also a problem of identifying acute infections for a largely asymptomatic disease. Disease elimination programs generally rely on clinically apparent signs of infection or complications. Identifying acute HCV infection requires active testing to identify seroconversion and repeated testing over time, unless a cross-sectional incidence assay becomes available (Patel et al., 2016). Verifying chronic hepatitis C requires attention to a high volume of cases and laboratory test results, exceeding the capacity of the surveillance infrastructure in most jurisdictions. Furthermore, the people at greatest risk for HCV are often marginalized in other ways; they tend to be undercounted in surveys and outside the reach of the formal health system.
THE FEASIBILITY OF ELIMINATING THE PUBLIC HEALTH PROBLEM OF HEPATITIS C
As the previous chapter describes, disease elimination is a matter of reducing the reproductive number (abbreviated R0) of a pathogen to a value less than one and maintaining this value until new infections cease. HCV has an R0 of about 2, which is to say its transmission is not especially efficient, and a modest reduction in transmission efficiency could end the public health problem of HCV (Pybus et al., 2001). If a quarter of a million people were treated for HCV in the United States in 2015, the treatment rate would exceed the annual infection rate by about 10-fold (Durham et al., 2016). But as the disease burden falls more on difficult-to-treat populations, this could change. Already the rate of new infections appears to be rising substantially, probably driven by increasing injection drug use among young people in at least seven states (CDC, 2013; Zibbell et al., 2015).
Biologically, HCV is a tractable target for eradication. There is no known non-human reservoir and, unlike HIV and HBV, there is no latent cellular reservoir (Ray et al., 2013). Pharmacologic treatment with direct-acting antivirals can truly cure an infected person, treatment-based elimination is an option (Pawlotsky, 2014). At the same time, reinfection is a possibility until either risk of transmission can be eliminated or prophylaxis is available via antiviral drug or vaccine. Such risk undermines progress toward HCV elimination (Simmons et al., 2016). Therefore, the committee concludes that elimination of hepatitis C as a public health problem in the United States is feasible, though there are several conditions on this statement.
Although the biology of the virus and medicines available to treat it would seem to favor the feasibility of elimination, there are serious barriers to this goal. Like hepatitis B, Hepatitis C is generally asymptomatic both during the acute and chronic phase. This epidemiological challenge is aggravated by the heavy concentration of HCV infections in difficult-to-reach populations, including people who inject drugs and the homeless, as well as marginalized groups such as the incarcerated. These groups are not generally represented, sometimes systematically under-represented, in national surveillance surveys such as NHANES (Edlin et al., 2015). Also, the curative treatment that holds the best promise to destroy the viral reservoir is exceedingly expensive. This expense raises concerns about the feasibility of widespread treatment (Durham et al., 2016). In any case, a prophylactic vaccination would make elimination more manageable, especially for a disease that disproportionately effects people who have limited access to medical care. A prophylactic vaccine may be an essential missing piece of the effort to eliminate hepatitis C. Similarly, if current trends in incidence continue, a single-dose curative treatment (as opposed to the current 8- to
TABLE 3-1 The Feasibility of Eliminating Hepatitis C as a Public Health Problem in the United States with Critical Factors for Success and Crosscutting Problems
| Goal | Feasibility | Critical Factors | Crosscutting Barriers | ||||||
| Ending Transmission | Feasible |
|
|
||||||
| Eliminating Chronic Infection | Feasible |
|
|||||||
| Reducing morbidity and mortality attributable to ongoing infection | Slowing progression to cirrhosis | Feasible |
|
||||||
| Reducing deaths | |||||||||
NOTE: HCV, hepatitis C virus.
12-week therapy) could be invaluable in ending chronic infection among patients often out of contact with the health system.
As with hepatitis B, eliminating the public health problem of hepatitis C in the United States is a matter of ending transmission and reducing the burden of disease among people already infected. The curative treatments available for HCV make the elimination of chronic infection another possibility. This chapter discusses each of these goals in turn and identifies factors crucial to the success of each goal. After analyzing the problem of hepatitis C in the United States, the committee concluded that control is feasible in the relatively short term. Eliminating the public health problem of hepatitis C will take more time, and require considerable public will, resources, and attention to the barriers mentioned in Table 3-1. Past success with other infectious diseases suggest that a prophylactic vaccine would greatly increase the likelihood and durability of progress toward elimination, especially given the disproportionate burden of HCV among people with the least engagement with the health system. Table 3-1 gives an overview of the committee’s main points and the conclusions presented in this chapter.
ENDING TRANSMISSION OF HEPATITIS C
Eliminating hepatitis C will require stopping transmission of the virus. In the United States, people who inject drugs have the highest risk of transmitting the virus (Alter, 1997). A strategy to stop transmission in this group should give attention to both reducing the risk of contracting HCV among people who are not infected and to reducing the likelihood of transmission among those who are (Edlin and Winkelstein, 2014; Grebely and Dore, 2014; Hellard et al., 2014). Preventing infection among people who use drugs was key to reducing incidence of HIV, but appears less effective for HCV (Palmateer et al., 2010; Wright and Tompkins, 2006). This is because HCV is more transmissible than HIV (Alter, 2006). Even without sharing
a needle, HCV can be spread through shared equipment such tourniquets (Hagan et al., 2001).
Nevertheless, there is some evidence that HCV transmission can be reduced through primary prevention efforts. In a 2011 review, Hagan and colleagues found little evidence that behavioral interventions, substance-abuse treatment, opioid-replacement therapy, or syringe programs reduced incidence of HCV when any one program was implemented alone (Hagan et al., 2011). However, two studies that employed multiple strategies (both including opioid replacement, one with counseling, the other with syringe exchange) reduced HCV incidence among drug injectors by about 75 percent (Hagan et al., 2011). A pooled analysis from the United Kingdom, however, suggested that syringe exchange and opioid replacement could each be effective in reducing infection by about 50 percent when implemented independently, provided the syringe exchange reached a large amount of the target population (Turner et al., 2011). When these programs were combined, their reduction on HCV incidence was near 80 percent, similar to the estimate in the Hagan review (Turner et al., 2011).
There is reason to believe that needle and syringe exchange programs, counselling, and opioid replacement therapy (collectively called harm reduction services) can help reduce the transmission of HCV especially among younger people and those who have more recently begun injecting (Mehta et al., 2011; Tseng et al., 2007). Cities with strong needle and syringe exchange programs, including New York and San Francisco, have seen significant declines in HCV prevalence (Des Jarlais et al., 2005; Tseng et al., 2007). The higher prevalence of HCV among older injectors and those who have been injecting longer suggests that the programs delayed, but did not prevent, the infection (Mehta et al., 2011). The longer time to infection may also prove a valuable window in which to reach young people who inject drugs with information about preventing infection or treating substance use disorders (Mehta et al., 2011). The recent lifting of Congress’ ban on use of federal funds for syringe exchange (with the caveat that the funds cannot be used on needles or syringes) may allow for expanding harm reduction programs.
Comprehensive drug and alcohol programs will be critical to any HCV elimination program. Such programs may be complicated. Injecting drug use is becoming more common in rural areas and small towns, not the just densely populated cities where such programs have a history (Suryaprasad et al., 2014; Zibbell et al., 2015). Adapting harm reduction strategies to rural and suburban areas could be challenging. Mathematical models suggest that high-coverage—referring to both the number of injectors reached in a geographical area and the number of syringes exchanged relative to injections made—is important in needle and syringe exchange programs (Day and Topp, 2011). Furthermore, needle and syringe exchange pro-
grams in combination with opioid replacement therapy can reduce HCV prevalence, but it takes many years to see these effects (Vickerman et al., 2012). In areas where harm reduction programs have been going on for many years, additional reductions in prevalence may be difficult without the introduction of new, more effective interventions, such as widespread treatment or use of a vaccine.
Treatment as Prevention and Reducing Reinfection
The simple, safe, and curative HCV treatments that came onto the market in 2014 changed not only the prognosis for individual hepatitis C patients, but also the strategy for interrupting transmission. Treating infected patients as a way to prevent disease spread is an established clinical and epidemiological principle in HIV (Cohen et al., 2011; Tanser et al., 2013). The promise of direct-acting, combination antiviral treatments for hepatitis C is even better, as these therapies can cure the vast majority of infections and can do so in relatively short time.
Modelling studies, many based on populations outside the United States, indicate that treating hepatitis C in people who inject drugs would reduce disease prevalence by 20 to 80 percent (Durier et al., 2012; Hellard et al., 2012; Martin et al., 2011). In Vancouver, where about 5 out of 1,000 drug-injecting hepatitis C patients are treated per year, treating an additional 71 patients per year would halve the burden of hepatitis C (Martin et al., 2013b). More recent models suggest that a fourfold increase in treatment of hepatitis C (from about 100,000 to 400,000 patients per year) could prevent more than 250,000 deaths and more than 500,000 cases of cirrhosis per year (Durham et al., 2016). As the burden of disease is heaviest among people who inject drugs, a 90 percent reduction in prevalence in the United States would require screening 20 percent of injection drug users and treating about a third of those infected (Durham et al., 2016) (see Box 3-1).
Translating these models’ promise into tangible results is complicated for several reasons. First, there may be error in the models introduced by the assumption of random mixing2 among people in a population. In reality, only a fraction of hepatitis C patients are actively transmitting the virus. The most infectious candidates are those currently using injection drugs, a relatively young and frequently incarcerated population (Hagan et al., 2004; Zeiler et al., 2010). Though hepatitis C passes only rarely from mother to child, pregnant women with HCV could transmit it to their children. Among people infected with both HCV and HIV the otherwise low risk of sexual transmission rises, so HIV+ people are also seen as higher
___________________
2 Meaning, for example, that a teenager who injects drugs is as likely to trasnmit the virus to a person of middle age as to another teenager.
risk for transmission. Treating hepatitis C in high-risk transmitters, an otherwise healthy group who could become reinfected, would prevent no immediate deaths. Preventing deaths means treating the patients at greatest risk of cirrhosis and end-stage liver disease: older people, often no longer sharing needles or injecting drugs at all, beyond child bearing age, and with fewer sexual partners (Hagan et al., 2004; Kirk et al., 2013). So while the direct-acting antiviral treatments can both prevent hepatitis C deaths and interrupt new infections, meeting these goals requires attention to widely different patient populations. Furthermore, treatment models assume that cases identified in screening programs would complete a course of treatment under appropriate medical supervision, but national data suggest that only about one-third of people who screen positive for HCV antibody receive
any follow-up care (Holmberg et al., 2013). Patients themselves may be reluctant to pursue treatment. Hepatitis C infection is mostly asymptomatic, encouraging the perception that it is not serious especially when weighed against the common and severe side effects of pre-2012 pegylated interferon and ribavirin treatment (Edlin et al., 2005; Mehta et al., 2008). The new direct-acting antiviral treatments are far more easily tolerated, but the misconception may persist, or concern with HCV be simply overshadowed by poverty and the comorbidities of addiction (Grebely et al., 2008; Mehta et al., 2008; Treloar et al., 2010). Providers may share in the reluctance to treat or even screen people actively using injection drugs out of a belief that they would not cooperate with treatment or be quickly reinfected (Lambert et al., 2011; Osilla et al., 2011).
The Threat of Reinfection
Reinfection is a barrier to using hepatitis C treatment as a tool to end transmission. Curative treatments are new, and research on reinfection after direct-acting antiviral therapy is limited (van de Ven et al., 2015). There is some evidence that risk of reinfection is relatively low, however. A 2013 analysis pooled data from seven studies on reinfection with HCV following treatment-induced sustained virologic response in men who have sex with men and people who inject drugs (Grady et al., 2013). The overall pooled estimate or risk of reinfection was between 0.91 and 6.12 percent per 100 person-years (Grady et al., 2013). The authors observed that the baseline incidence of HCV in the population appeared to influence this rate. So in places where HCV infection was rare, the risk of reinfection in high-risk patients was lower than in places where the background incidence of HCV was high (Grady et al., 2013). It is also not easy to distinguish between relapse after sustained virologic response and true reinfection. Patients from high-risk groups may be more likely to be infected with more than one strain of HCV. In these cases one strain may respond to treatment, while another persists (Grady et al., 2013).
Frequency of infectious contacts after being cured and the background HCV prevalence in the subpopulation both influence the risk of reinfection (Grebely et al., 2012). Phylogenetic analysis indicates that HCV transmission clusters among people with a reported injecting relationship (Sacks-Davis et al., 2012). Treating all infections within an injecting group—accomplished by encouraging patients to bring their friends for treatment—has promise to reduce prevalence among the drug-injecting subpopulation by 7 percentage points more than traditional treatment strategies and is currently being tested in Australia (Hellard et al., 2014, 2015).
Harm Reduction
Active drug users drive the bulk of hepatitis C transmission in the United States (Alter, 2002, 2007; Bruggmann and Grebely, 2015). The expansion of harm reduction services holds particular promise to interrupt transmission in this group. Recent models indicate that preceding hepatitis C therapy with a 40 percent increase in harm reduction services could decrease the treatment rate required to halve chronic hepatitis C prevalence over 10 years (Martin et al., 2013a).
The people most likely to transmit the virus are young and injecting drugs—people who are not well connected to health or social services. Reaching this group will be challenging, but there may be transferrable strategies from HIV (e.g., mobile testing and treatment, community and peer support, cash incentives) to encourage compliance with treatment
(Chang et al., 2013). There is also the matter of addressing the root causes of drug addiction, and linking hepatitis C treatment to social and mental health services.
ELIMINATING CHRONIC HEPATITIS C
There are 2.7 to 4.7 million people in the United States with chronic hepatitis C (Ward and Mermin, 2015). The disease disproportionately affects people aged 40 to 59, the same age group already experiencing the increased burden of chronic disease brought on by age. The burden of HCV is also felt more among the poor and less educated; fewer than half of people with HCV infection have incomes greater than twice the poverty level or education beyond high school (Denniston et al., 2014). Even these estimates might be biased toward the relatively stable and affluent, as prisoners and homeless people, known risk groups for HCV, are not included in national surveys (Denniston et al., 2014).
Managing chronic disease is challenging for reasons discussed later in this chapter. Both the chronic disease patient and his or her providers need to be involved in an ongoing process from diagnosis through continued monitoring and treatment. This process is sometimes called the care cascade or treatment cascade (HHS, 2015b). For people living with chronic hepatitis C, the care cascade starts with diagnosis and links to care. Once in care, patients should have HCV RNA confirmatory testing and genotyping and undergo liver fibrosis staging to help inform their prognosis and make decisions regarding antiviral therapy. Lastly, they must be prescribed HCV
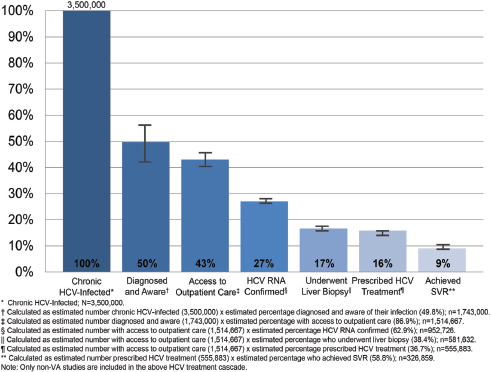
NOTE: HCV, hepatitis C virus; RNA, ribonucleic acid; SVR, sustained virologic response; VA, Department of Veterans Affairs.
SOURCE: Yehia et al., 2014.
treatment and adhere to that treatment to the point of sustained virologic response (Yehia et al., 2014). National guidelines outline the appropriate care for patients along this continuum (Ghany et al., 2009, 2011; Yee et al., 2012).
The concept of a care cascade as a diagram showing the proportion of patients accounted for at every step of treatment, provides a framework for evaluating hepatitis C services over time and within patient subgroups. The effects of new screening efforts or antiviral drugs can be shown clearly on such a diagram (Moyer, 2013; Smith et al., 2012; Thomas, 2012). One recent meta-analysis made clear that the sharpest drop in the continuum from infection to sustained virologic response was in the diagnosis of HCV infection (Yehia et al., 2014) (see Figure 3-1).
In 2012, the CDC recommended one-time HCV testing for everyone born between 1945 and 1965, regardless of other risk factors, because this group is thought to account for three-fourths of all HCV infections in the
country (Smith et al., 2012). The same recommendation called for referral of newly identified HCV-infected people to care (Smith et al., 2012). This recommendation may be better observed in the breach than in practice. CDC and NHANES research suggests that only about half of HCV infected people are aware of their condition (Denniston et al., 2012). Among those who are aware, about 57 percent carry health insurance, compared to 43 percent of those unaware of their infection (Denniston et al., 2012).
As Figure 3-1 illustrates, the prescription of antiviral therapy is another drop-off on the HCV care cascade, though recent developments in treatment may be reducing this decline. A retrospective analysis of Department of Veterans Affairs data found that the 2011 introduction of boceprevirbased combination therapies nearly doubled the monthly volume of patients treated (Gidwani et al., 2015). The advent of all-oral, highly efficacious, and tolerable direct-acting antivirals has shortened treatment duration. Shorter treatment means less opportunity to lose track of patients during therapy; the better tolerability of the treatments should allow for improved compliance. The best strategies for retaining patients in care are still unclear, however.
Access to Direct-Acting Antiviral Therapies
Direct-acting antiviral drugs make cure possible in 95 percent of chronic HCV infections, but their use is limited. The cost of these treatments is discussed later in this chapter, but briefly, the high cost and anticipated demand for these drugs have led many insurers to restrict access to these medications. Before approving a prescription some insurers need evidence of advanced liver fibrosis or consultation with a specialist (Brennan and Shrank, 2014; Saag, 2014; Steinbrook and Redberg, 2014). Many insurers require patients abstain from alcohol and illicit drugs for a set time before beginning treatment; some request drug screening to confirm sobriety (Grebely et al., 2015). Two recent reports analyzed how these restrictions are put into practice across different state Medicaid programs. They revealed considerable variation in different states’ restrictions on treatment, despite the federal Medicaid law’s requirement that all states cover drugs in accordance with the FDA label (Barua et al., 2015; Canary et al., 2015).
Wide inconsistencies in insurers’ restrictions on access to hepatitis C treatments have created extra work for providers. Insurers now require that the prescribing clinician or collaborating pharmacy submit a request for prior authorization documenting that the patient meets the insurer’s criteria for HCV treatment. The insurer reviews the request and either approves or denies it. If prescription is denied, the prescriber can appeal the decision; the insurer will uphold or overturn the denial based on the supporting information presented during the appeal. It is unclear what toll the
denial and appeal process might take on clinical outcomes, to say nothing of patient–provider relationships. Further research examining the consequences of treatment denial could inform a more complete understanding of the direct and opportunity costs of treatment.
Adherence to Antiretroviral Therapy
Clinical trials have shown direct-acting antivirals to be highly efficacious and tolerable, but their real-life effectiveness depends on the patient’s adherence to treatment (Afdhal et al., 2014b; Feld et al., 2014; Sulkowski et al., 2014). As with other chronic viral illnesses, particularly HIV, poor adherence reduces the likelihood of treatment response (Bangsberg et al., 2001; Gross et al., 2001; Nachega et al., 2007). Thus, understanding patients’ actual adherence to treatment is crucial when considering the possible elimination of chronic HCV infection.
Before the direct-acting agents were available, better adherence to treatment pegylated interferon and ribavirin was associated with greater HCV RNA declines and higher rates of early and sustained virologic response (Lo Re et al., 2009, 2011, 2013). But these treatments were difficult for patients to tolerate, and adherence tended to decline over the course of therapy. The direct-acting antivirals are easier to tolerate, but to date no studies have evaluated the effects of adherence on serological markers of infection. Given the costs of these therapies and the possible consequences of non-adherence (i.e., drug resistance, continued progression of liver disease, potential to transmit HCV infection), it is important to understand the relationship between adherence and sustained virologic response, the patterns of adherence to different regimens, and changes in adherence over the 8- to 12-week course of therapy. Such analyses could help clinicians ensure the best possible clinical response (Weiss et al., 2009).
Threat of Resistance to Direct-Acting Antiviral Therapies
When patients do not respond to direct-acting antiviral therapy, there is a threat that unsuccessful treatment can select for HCV variants resistant to these agents (Poveda et al., 2014). The selection of mutations at different positions in the NS3/4a protease, NS5B polymerase, and NS5A replication complex proteins drive the development of resistance to direct-acting antivirals (Sarrazin and Zeuzem, 2010). With the exception of NS5B nucleos(t)ide inhibitors, most direct-acting antivirals have a low genetic barrier to resistance, with significant cross-resistance between agents in the same drug class. Cross-resistance between drugs in the same class seems to be of greatest concern with use of NS3/4A protease inhibitors and NS5A inhibitors (Poveda et al., 2014). A specific mutation profile may be associ-
ated with each medication or class and can vary by genotype or subtype (Poveda et al., 2014; Wyles, 2013). There are also resistant mutations existing as natural polymorphisms in certain genotypes and subtypes. In the past, testing prior to antiviral treatment aimed to identify these mutations before treatment. For example, the Q80K polymorphism is frequently found among patients with genotype 1a3 and is associated with resistance to the NS3/4A protease inhibitor simeprevir (Kieffer et al., 2012; Poveda et al., 2014; Trevino et al., 2011; Trimoulet et al., 2011).
Cure rates for current direct-acting antiviral agents exceed 90 percent, but these agents have only been used for a few years. Many questions regarding resistance remain. Current data suggest that preexisting mutations have a negligible impact on HCV treatment response, and retreatment of patients with treatment-acquired resistance variants can be effective (Afdhal et al., 2014a; Feld et al., 2014; Sulkowski et al., 2014). The clinical significance of preexisting resistance mutations for these therapies is largely unknown. Moreover, it is not clear how long existing resistant mutations persist or if NS3/4A or NS5A cross-resistance might be a concern for patients who failed prior treatment regimens with these agents. These are important questions, as the threat of resistance needs to be considered in the management of hepatitis C and the eventual elimination of the disease.
REDUCING MORBIDITY AND MORTALITY FROM HEPATITIS C
People living with HCV—especially those with cirrhosis—are at a substantially increased risk of death. HCV-infected people participating in the NHANES survey between 1988 and 1994 and followed though 2006 had a 2.4 times higher all-cause mortality rate and a 26.5 times higher liver-related mortality rate ration compared to HCV negative people (El-Kamary et al., 2011). Models that assume absence of treatment suggest that incident cases of end stage liver disease among HCV-infected people will peak
___________________
3 19 to 48 percent prevalent.
between 2030 and 2035 with 379,600 cumulative deaths by 2030 and 1,071,229 cumulative deaths by 2060 (Rein et al., 2011).
HCV related cirrhosis is also a leading indication for liver transplantation in the United States. The Scientific Registry of Transplant Recipients from 2012 gives HCV-related cirrhosis as the underlying cause for listing 4,612 patients (about 30 percent of the list), resulting in 1,402 liver transplants (OPTN/SRTR, 2014). If untreated prior to liver transplantation, HCV will reinfect the transplanted organ resulting in decreased patient and graft survival. Efforts to cure HCV in patients with decompensated cirrhosis may help avoid need for liver transplantation. If transplantation is still required, HCV cure after transplant prolongs graft survival (Joshi et al., 2014).
HCV also causes considerable morbidity, including complications not related to liver disease. HCV-related cryoglobulinemia has been associated with cutaneous and visceral vasculitis, and glomerulonephritis, as well as B-cell non-Hodgkin’s lymphoma (Negro et al., 2015). Because the liver and heart function together to ensure a healthy blood flow, liver damage can cause cardiovascular problems. HCV patients have increased risk of cardiovascular ailments such as carotid plaques, insulin resistance, diabetes, and hypertension (Petta et al., 2012; Younossi et al., 2013).
HCV also hurts patients through less tangible reductions to quality of life. When compared to uninfected and HBV-infected controls, HCV patients reported significantly more fatigue, low energy, and body pain (Foster et al., 1998). Fatigue and cognitive impairment are common in people with hepatitis C (Cacoub et al., 2002; Weissenborn et al., 2004). Though sickness can itself be a cause of depression, when compared to other people with liver disease, hepatitis C patients had worse quality of life and more depressive feelings, even after viral clearance (Tillmann et al., 2011).
The Staging and Progression of Hepatitis C
The progression of HCV infection is closely linked to the severity of fibrosis. Liver biopsy is considered the standard way to assess fibrosis, but the procedure has many limitations. Only a small sliver of liver tissue is drawn in biopsy. A specimen that accounts for only about 1/50,000th of the organ may not be representative of the liver as a whole. Even experienced pathologists can disagree on the stage of a sample; inter- and intra-observer variability of 10 to 20 percent is not unusual (Bravo et al., 2001). The specialists needed to draw and read the specimen drives up the cost of the procedure, which can also be painful, and carries a risk of bleeding and drop in blood pressure that can require hospitalization in 1 to 3 percent of patients (Bravo et al., 2001). For these reasons, less invasive methods are needed to stage fibrosis. Certain panels of serum markers and non-
invasive imaging techniques such as transient elastography and magnetic resonance elastography can also be used to assess fibrosis, and have promise for describing the disease burden in the growing population of hepatitis C patients (Poynard et al., 2007; Vallet-Pichard et al., 2007; Venkatesh et al., 2013; Wai et al., 2003).
Twenty to 40 percent of chronic hepatitis C patients will develop cirrhosis; the median duration of time from infection to cirrhosis is around 30 years (Poynard et al., 1997). Age, sex, and lifestyle appear to influence disease progression: Men infected after age 40 progress about 13 years faster than the median, while non-drinking women infected before age 40 might be expected to live 42 years before the onset of cirrhosis (Poynard et al., 1997). Estimating any patient’s risk of fibrosis is complicated, however, because of often unidentifiable interactions between the virus and the host. Among strains of HCV, genotype 3 appears more virulent; infection with HBV or HIV speeds the progression of fibrosis in HCV patients, though interestingly, neither viral load nor means of infection consistently appear to have an effect on the progression of the disease (AASLD-IDSA, 2015c; Poynard et al., 2001). Other predictors of disease progression include fibrosis stage, inflammation grade, male sex, past organ transplantation, and age at time of HCV infection.
Fibrosis does not generally progress in a predictable or linear way, however. After 20 years of HCV infection the overall risk of cirrhosis is about 16 percent; after 30 years, 41 percent (Thein et al., 2008). Age at the time of exposure and duration of infection are established risk factors for development of advanced fibrosis and cirrhosis. The process does accelerate at the patient ages, however; fibrosis progresses most quickly after age 50 (Poynard et al., 2001). Therefore, both the patient’s age and the duration of his or her infection should be taken into account when identifying those at high risk of illness or death from advanced fibrosis and cirrhosis.
Still, certain patient characteristics seem to predict faster progression of fibrosis, some of these are within the patient’s ability to control. Alcohol consumption, for example, increases the HCV-infected person’s odds of developing cirrhosis fourfold, and appears to speed the path to cirrhosis (Harris et al., 2001; Thomas et al., 2000a). HCV patients who drink alcohol are hospitalized and die younger than those who abstain (Kim et al., 2001). Given these risks, abstinence from alcohol is the safest strategy for preventing cirrhosis in HCV-infected people, and support for alcohol cessation in heavy drinkers might influence the success of an elimination program.
The co-morbidities of obesity aggravate the natural course of HCV. Dyslipidemia and type 2 diabetes can develop as both cause and an effect of a non-alcoholic fatty liver disease, a condition of fatty infiltration in the liver found in about 20 percent of American adults (Chalasani et al., 2012; Vernon et al., 2011). When HCV patients also have fatty liver disease,
they appear more likely to develop advanced fibrosis (Sanyal et al., 2003). Fatty liver and other obesity-related metabolic conditions are common in the United States; they could influence the speed at which HCV patients develop complications.
Co-infection with HIV, though found in less than 20 percent of people with HCV, can do the same (AASLD-IDSA, 2015b; Rotman and Liang, 2009). Infection with both HIV and HCV causes more aggressive progression to fibrosis, and speeds the onset of cirrhosis by 12 to 16 years (Di Martino et al., 2001; Rockstroh and Spengler, 2004). A prospective study comparing HCV patients with and without HIV found higher rates of hepatic decompensation in the patients with both infections (hazard ratio accounting for competing risks, 1.56 [95% confidence interval (CI), 1.31-1.86]). Even when the patient’s HIV was well-controlled, the risk of hepatic decompensation persisted (Lo Re et al., 2014). Given the particularly aggressive course of liver disease in people infected with both HIV and HCV, their treatment with direct-acting antivirals for HCV should be a priority. Curing infection in this group could prevent the complications of advanced liver disease (Naggie et al., 2015).
Prevention of Cirrhosis and Hepatocellular Carcinoma Through Treatment
The curative treatments available for HCV make it different from other chronic viral infections. The new drugs have made cure or sustained virologic response possible in more than 95 percent of patients with relatively short courses of treatment with few or mild side effects (Afdhal et al., 2014a; Feld et al., 2014). Sustained virological response is usually described as durable, meaning that it persists for years after cure. In follow-up studies over 5 years and longer, patients with sustained virologic response have undetectable HCV RNA; relapse is rare and associated with pre-treatment cirrhosis (George et al., 2009; Maylin et al., 2009)
Eradicating the virus before the patient starts progression to advanced fibrosis can also largely eliminate the risk of hepatic complications. HCV treatment can also improve liver histology. In a pooled analysis of 3,010 treatment-naïve patients who underwent liver biopsy before and after interferon based therapy, patients who achieved sustained virologic response had a significant improvement in disease activity (Poynard et al., 2002). Another study on paired biopsies on 49 patients who achieved sustained virologic response, 82 percent had a decrease in fibrosis score and 20 percent had normal or near normal histology after 5 years (George et al., 2009). The clinical and histologic evidence that disease progression stops in patients with early stage fibrosis who are cured of HCV infection informed the current American Association for the Study of Liver Disease (AASLD)
and Infectious Disease Society of America (IDSA) recommendation that no further monitoring of these patients is warranted after cure (AASLD-IDSA, 2015a). In short, curing HCV before the disease progresses to advanced fibrosis is the most efficient way to prevent the complications of the infection.
Prevention of Complications in Patients with Advanced Fibrosis
HCV patients who already have cirrhosis are at risk of decompensated liver disease, hepatocellular carcinoma, and death. In those with compensated cirrhosis, portal pressures remain relatively low; despite histologic evidence of cirrhosis, clinical presentations are largely asymptomatic. The compensated cirrhosis of chronic HCV infection can progress to decompensated cirrhosis, marked by increase in portal pressure, which causes ascites, portal hypertensive gastrointestinal bleeding, hepatic encephalopathy, and jaundice. Portal hypertensive complications significantly increase risk of death. For example, 1-year mortality risk in a patient with non-bleeding esophageal varices and without ascites is 3.4 percent, but with bleeding esophageal varices, the 1-year mortality jumps to 57 percent (D’Amico et al., 2006). One-year survival in patients with decompensated cirrhosis is approximately 82 percent, and 5-year survival was near 51 percent (Planas et al., 2004).
Direct-acting antiviral therapy has been shown to be safe and highly effective in achieving sustained virologic response among cirrhotic patients (Afdhal et al., 2014a; Charlton et al., 2015). Patients with compensated cirrhosis who undergo curative therapy are unlikely to progress to a decompensated state. In patients with cirrhosis or advanced fibrosis, sustained virologic response has been associated with improvement in histology as well as decreased liver-related death, incidence of liver failure, development of hepatocellular carcinoma and all-cause mortality rates (Backus et al., 2011; Veldt et al., 2007). Antiviral treatment has also recently been shown to restore liver function in decompensated cirrhosis. HCV patients with Child4 B and C cirrhosis not only had high response rates to direct-acting antiviral therapy, with 86 to 89 percent showing sustained virologic response, but also had improvements in Childs score, MELD score,5 albumin and bilirubin levels (Charlton et al., 2015). In some ways, cirrhotics have the most to gain from curative HCV treatment, as the nature of their disease puts them at the most immediate risk of death.
___________________
4 Officially, Child-Pugh or Child-Turcotte-Pugh score, a classification used to assess the prognosis of chronic liver disease. Child class B and C have 3-year survival rate of 59 and 46 percent, respectively (Angermayr et al., 2003).
5 The Model for End-Stage Liver Disease, or MELD score, rates the severity of chronic liver disease on a scale of 1 to 40 with higher scores indicating higher 3-month mortality (Cholongitas et al., 2005).
When cirrhosis is advanced, viral eradication alone may not restore synthetic dysfunction and portal pressures. Today, these patients require liver transplantation. Transplantation is an expensive procedure. The scarcity of donor organs motivates requirements that candidates be otherwise healthy and have strong social support. In the future, transplant might be avoided with the use of anti-fibrotic and portal pressure lowering therapies now in development (Friedman, 2015). In the meantime, antiviral therapy is an effective means to prevent progression to severely decompensated cirrhosis.
In patients with chronic hepatitis C, risk of hepatocellular carcinoma is closely linked to development of fibrosis (Yoshida et al., 1999). Because hepatocellular carcinoma in the absence of advanced fibrosis is rare, halting progression of fibrosis though therapy will be an effective means of cancer prevention. For those who already have advanced fibrosis, sustained virologic response is still an effective means to reducing cancer risk. A recently published meta-analysis showed that among 2,649 patients with advanced fibrosis only 4.2 percent of patients who achieved sustained virologic response developed hepatocellular carcinoma, compared to 17.8 percent of patients who did not respond to therapy (Morgan et al., 2013).
Need for Further Research
Preventing complications of advanced liver disease in people with HCV requires reliable predictive models for disease progression. Although risk factors such as HIV and alcohol use have been well described, most of the factors influencing disease progression are not understood well, making it difficult to predict any patient’s likely outcome. More research into host and viral genomics might help advance such understanding, as would better predictive models for progression of fibrosis (Harouaka et al., 2016). Ultimately, such tools would help clinical teams identify patients who should be the first priority for therapy.
Clinical management of HCV patients would also benefit from better therapy to avoid the complications of portal hypertension among cirrhotic patients. Anti-fibrotic therapies and better pharmacologic treatment for portal hypertension are possibilities currently under investigation (Friedman, 2015). Although successful HCV therapy allows for regression of disease in most patients, these treatments will be necessary for those who still progress or continue to have significant amount of fibrosis after treatment with direct-acting antivirals.
CROSSCUTTING BARRIERS TO HEPATITIS C ELIMINATION
Any progress toward reducing transmission of HCV, toward eliminating chronic infection, or to reducing the morbidity and mortality attributable to chronic infection could be impeded by certain systemic barriers. Problems with disease surveillance, enrolling and managing patients in care, the cost of the curative treatment necessary to eliminate chronic infection, stigma, and motivation to eliminate the disease could also pose serious impediments to progress against the infection.
Surveillance
Chapter 2 describes problems with viral hepatitis surveillance in the United States. A 2010 Institute of Medicine (IOM) report on hepatitis and liver cancer recommended improving national surveillance for these viruses, but to date the CDC funds such programs in only seven jurisdictions (HHS, 2015a; IOM, 2010).
Surveillance for viral hepatitis could improve understanding of the true burden of disease across the care cascade. Identifying acute cases is essential to identifying an outbreak and instituting control measures, especially for nosocomial outbreaks or ones connected to medical care (Apostolou et al., 2015). Follow-up on acute cases can also help identify changes in the pattern of the HCV epidemic. If, for example, the public health officer sees a spike in infections among young people who inject drugs or among HIV+ men who have sex with men then they can adapt preventative measures accordingly (Breskin et al., 2015; Onofrey et al., 2011).
Challenges to conducting acute surveillance for HCV abound, however.
First of all, the unsafe injection of drugs drives most transmission in the United States (Alter, 1997). People who inject drugs are less likely to receive medical care, and are therefore less commonly reported to the health department. Identifying acute HCV cases is also an imprecise process. The Massachusetts health department recently found that of 183 patients clinically diagnosed with HCV over ten years, only one would have met the case definition required for inclusion in national statistics (Onofrey et al., 2015). (See Box 3-2 for case definitions.) As the CDC bases its estimates of HCV incidence on reported acute cases, overly restrictive definitions and inconsistencies in clinical or laboratory reporting could lead to an amplified underestimate of true disease burden (Onofrey et al., 2015). Without a simple test for acute cases this problem will likely persist, as will the challenge of identifying asymptomatic cases.
Identifying spikes in acute infection is important, and a reasonable
expectation for a surveillance system. But a good understanding of chronic hepatitis C prevalence in an area is essential for allocating resources for prevention and treatment. Estimates of national prevalence are derived from the NHANES, Aside from the sampling limitations of the NHANES discussed earlier, the survey does not distinguish between differences in disease burden among different jurisdictions (Edlin et al., 2015). A more accurate response to the epidemic would require better local data, which could give a more accurate picture of the full, local care cascade, not just the acute infections.
Surveillance for HCV requires a strong public health infrastructure; chronically infected people often need multiple assays every year for years. The volume of testing needs to move through the system promptly to allow for real-time follow-up on potential outbreaks. A proper understanding of HCV burden would also attempt to capture the consequences of chronic infection, linking surveillance to health insurance claims, electronic health records, and birth, death, and cancer registries. There is also a need for deliberate attention to those populations at high risk for the disease: people who inject drugs, the homeless, and incarcerated. A system that can promptly identify seroconversion in these groups has the chance to react to new outbreaks and spot trends in how the disease is transmitted. Contact tracing, a common practice in outbreaks of sexually transmitted disease such as syphilis and HIV, may be a useful practice for HCV as well.
Screening
Even the best disease surveillance cannot change the fact that about half the chronically infected people in the United States are undiagnosed (Smith et al., 2012). In 2012 the CDC expanded HCV screening guidelines; later the US Preventive Services Task Force recommended testing everyone born between 1945 and 1965 (often called the Baby Boomers) (Smith et al., 2012; USPSTF, 2013). Baby Boomers account for about three-quarters of the country’s chronic hepatitis C (Ward, 2013). When screening this birth cohort became the default at the University of Alabama Hospital emergency room (meaning that anyone born in these years was tested unless he or she declined testing), they identified chronic infection in one out of every nine people screened (Feld et al., 2015). This prevalence is considerably higher than what would be expected, even among Baby Boomers, possibly because poor and uninsured people are both more likely to seek care at emergency departments and more likely to have hepatitis C (Galbraith et al., 2015). The greatest incidence of HCV is in younger age groups, driven largely by drug injection among whites in rural areas and small towns (Suryaprasad et al., 2014). This changing pattern of disease complicates the protocol for screening. There might be transferable lessons from HIV screening, which
has established methods of linking HIV+ patients to treatment and social support, and some overlapping target populations. There is also room to make better use of screening opportunities in prisons and in drug treatment and harm reduction programs. Research in sexually transmitted disease clinics found that HCV counselling, testing, and referral for people who inject drugs cost about $25 per patient tested (Honeycutt et al., 2007).6 About 45 percent of participants who inject drugs returned to the clinic for their results, a far better rate of return that found among the non drug-using patients of whom less than 13 percent returned (Honeycutt et al., 2007).
About half of the chronically HCV-infected people in the United States are unaware of their condition (Denniston et al., 2014; Holmberg et al., 2013). The first steps to preventing deaths in these people is diagnosing and retaining them in care. Most hepatitis C screenings are done in clinical settings, which can ease the patient’s referral to care and provide better assurance that the test result will be documented (CDC, 2013). But those at highest risk for HCV, including people who inject drugs, the homeless, and the incarcerated, can remain outside the reach of clinical screenings (Larney et al., 2014). Community screening might be a way to reach more patients, provided there is a strong referral system in place (Trooskin et al., 2015). Correctional facilities pose a similar opportunity. Maintaining contact with patients met through community screening is challenging, however. A team working in New Jersey and New York City found that screening at community organizations allowed for diagnosis of new patients, most of whom had no traditional risk factor for HCV, but 36 percent of these newly diagnosed people were lost to follow-up and never evaluated for treatment (Perumalswami et al., 2013). The method of screening might contribute to the attrition. Research in mobile clinics in New Haven found that HCV-infected patients diagnosed with point-of-care tests were significantly more likely to link to care and get treatment then those tested with traditional and slower phlebotomy tests (Morano et al., 2014).
Enrolling and Retaining Patients in Hepatitis C Care
Once patients are referred to care, the challenge of managing them over time remains. HCV might be particularly difficult to manage because of the demands it puts on primary care. A 2014 survey found that about half of primary care physicians were already at full capacity, about a third reported being overextended and overworked (The Physicians Foundation, 2014). In general these providers are obliged to give more attention to acute and urgent problems than to the management of chronic disease (Bodenheimer
___________________
6 In 2006 dollars.
et al., 2002). This burden on the health system could impede progress toward the elimination of hepatitis C.
A concern with redesigning the health system to meet the needs of the chronically ill is the essence of Edward Wagner’s chronic care model described in Box 3-3 (Glasgow et al., 2001; Wagner et al., 2001). These principles have been shown to be useful in clinical practice. A 2009 review found that the chronic care model improved patient outcomes or the care process; when diabetic care conformed to the model, for example, HbA1c score and risk of heart disease decreased (Coleman et al., 2009; Parchman et al., 2007). Granted, research on the chronic care model often has many component interventions. It can be difficult to separate the benefits directly due to the chronic care model from those due to an observer bias, but the
research suggests that the model has promise to greatly improve outcomes for patients with chronic disease (Coleman et al., 2009; Landon et al., 2004).
It is also possible that patient navigators, lay health workers who can help patients overcome barriers in the health system, can help HCV-infected people stay in care. The patient navigator model has been shown effective in managing chronic conditions such as HIV and cancer (Vargas and Cunningham, 2006; Wells et al., 2008). The strategy has not yet been fully evaluated for HCV, though. Recent work in Philadelphia found that patient navigators can greatly facilitate diagnosis and retention in care of patients identified through community screening (Trooskin et al., 2015). Another Philadelphia group demonstrated that routine HCV testing and referral could be successfully integrated with ambulatory care for poor and homeless people (Coyle et al., 2015). Medical assistants helped relieve the burden on clinicians and other health center staff by guiding patients through their appointments and follow up. They also used reflex HCV testing technology to ensure that anti-HCV-positive patients received HCV RNA testing; the reflex technology also has the advantage of providing HCV antibody and RNA test results in one visit. Intensive services carried out by a patient navigator further increased the number of HCV-infected patients who received their results and were referred and seen by a specialist (Coyle et al., 2015).
Cost
An emphasis on diagnosis and bringing patients to care will do much to reduce the burden of disease from HCV, but the most promising tool to eliminate chronic disease is the direct-acting antiviral drugs that elicit sustained virologic response in more than 95 percent of patients (Kohli et al., 2015). These response rates are similar across genotypes, with the exception of genotype 3, and for treatment-naïve patients as well as those who failed prior treatments.
Although effective treatment is now a reality, access to treatment remains poor. Estimates that predate the development of curative HCV drugs suggested that about a third of people aware of their chronic HCV infection were on treatment (Kanwal et al., 2010; Moorman et al., 2013). No recent population-based studies have examined the fraction of people with HCV infection on direct-acting agents, but some inferences may be drawn from sales data of pharmaceutical firms selling these agents. In 2014, US sales of Harvoni®7 and Sovaldi®8—the two blockbuster direct-acting agents mar-
___________________
7 The US proprietary name of 90 mg of the viral NS5A inhibitor ledipasvir and 400 mg of sofosbuvir, a nucleotide inhibitor of the viral RNA polymerase.
8 The US proprietary name of sofosbuvir.
keted by Gilead—totaled $10.5 billion, and an estimated 140,000 patients initiated HCV treatment using one of these two drugs (Gilead Sciences, 2015a; Pollack, 2015). So the revenue per patient initiating therapy was about $75,000.9 In the first three-quarters of 2015, US sales of these drugs totaled $10.1 billion, the latest sales data available at the time of this report, and another ~200,000 patients initiated treatment with Harvoni® or Sovaldi® (Gilead Sciences, 2015b,c,d). So the revenue per patient initiating therapy in 2015 was $52,000. The decline in revenue per patient or discounted price might reflect competition from new HCV therapies such as AbbVie’s Viekira®10 approved in December 2014. AbbVie reported US sales of Viekira® of roughly $600 million in the first three quarters of 2015 (AbbVie, 2015). Assuming that revenue per patient initiating therapy was $50,000, it follows that roughly 12,000 patients initiated treatment with Viekira® in 2015. Taken together these estimates imply that roughly 350,000 patients have initiated treatment with direct-acting agents since their introduction in late 2013. Given the estimated HCV prevalence in the United States, one can assume between seven and fourteen percent of people with HCV infection have initiated treatment with direct-acting agents (Edlin et al., 2015).
So despite the impressive effectiveness of direct-acting agents only about 1 in 10 chronically infected people is given curative treatment. Admittedly, part of the problem is the asymptomatic nature of HCV, leaving about half of chronically infected people unaware of their condition. The high prevalence of HCV among marginalized and difficult to reach people also complicates the problem. Still, the price of direct-acting agents is a major barrier to eliminating the disease. The first direct-acting agent introduced in the market (Gilead’s Sovaldi®) was launched at a price of $1,000 per pill or about $84,000 for a 12-week course of treatment (Sanger-Katz, 2014). Subsequent introductions of similar drugs were priced between $54,000 and $168,000 per course of treatment (Bickerstaff, 2015; Loria, 2016). Treating all diagnosed HCV infections in the United States with these drugs would cost about $175 billion up front, and would be unmanageable in other ways; treating millions of hepatitis C patients would exceed the capacity of available providers (Van Nuys et al., 2015a,b). Even treating only 5 percent of known infections would cost about $25 billion up front (Van Nuys et al., 2015a).
___________________
9 Revenue per patient is simply the $10.5 billion in revenue divided by the 140,000 patients initiating therapy. Revenue per patient is a good approximation of the discounted price for a full course of treatment received by the manufacturer from health plans and other payers. It is lower than the list price for these drugs as it includes the discounts offered by the manufacturer to payers.
10 The US proprietary name of ombitasvir, paritaprevir, and ritonavir tablets co-packaged with dasabuvir tablets.
These prices have met widespread criticism for being too high (Hoofnagle and Sherker, 2014; Pollack, 2015). The Senate Finance Committee’s 18-month investigation into the pricing of Sovaldi® and Harvoni® concluded that Gilead priced the drug to maximize profits without consideration of affordable access (US Senate Committee on Finance, 2015). The committee also recognized that Gilead’s pricing analyses had not accounted for the restrictions Medicaid and insurance companies would place on access (US Senate Committee on Finance, 2015).
There are two primary arguments to support the claim that the prices of direct-acting agents are too high. First, is that the high prices are unaffordable—not only for the uninsured patient, but also for insured patients and their health plans. Roughly one-third of the people with HCV infection lack insurance coverage (Ditah et al., 2015; Stepanova et al., 2011). Lack of insurance coverage is highly predictive of not receiving follow-up care or treatment after diagnosis with HCV infection (Ditah et al., 2015). And even if the uninsured were all to enroll in care, the financial burden of paying for treatment would be tremendous. Insurers have responded to the drugs’ prices by imposing clinical restrictions on access. As the previous section mentions, most Medicaid programs restrict access to direct-acting antivirals by limiting eligibility for treatment to those with advanced fibrosis or cirrhosis or by limiting coverage to prescriptions from a specialist (Barua et al., 2015; Canary et al., 2015). The drug and alcohol testing described earlier in this chapter pose further barriers. Finally, for people co-infected with HIV and HCV, some states’ Medicaid programs limit access to individuals receiving antiretroviral therapy for HIV or to those who have suppressed HIV RNA levels (Barua et al., 2015). Three-quarters of states’ Medicaid programs limit access to persons with advanced liver disease, two-thirds of states have restrictions based on prescriber type, and half require a period of abstinence (Barua et al., 2015). These restrictions in access to direct-acting agents are not just limited to Medicaid. Several private insurers, including Aetna, BlueCross, and United Healthcare, have instituted similar controls (Aetna, 2015; BlueCross BlueShield of Mississippi, 2015; United Health Care, 2015). No clinical evidence or treatment guidelines support the restrictions; they appear to be motivated only by concerns about cost (Harvard Law School, 2013). A national analysis of prescription drug spending found that expenditures on prescription drugs increased by 12.2 percent in 2014—a sharp acceleration from growth of 0.2 percent in 2012 and 2.4 percent in 2013 (Martin et al., 2015). The introduction of direct-acting antivirals drove most of this acceleration and accounted for one-third of the 2014 spending increase (Martin et al., 2015). Another recent analysis found that unrestricted coverage of direct-acting agents would increase California’s Medicaid budget by about 5 percent per member per month (Institute for Clinical and Economic Review, 2015).
Such dramatic budget increases to cover just one therapy are not common, making insurers reluctant to provide unrestricted coverage for these drugs.
At the same time, not everyone agrees that the prices of direct-acting agents are too high or unsustainable. There are three arguments that support their case. First, despite the high prices, direct-acting agents are cost-effective for treating patients with genotype 1 HCV, the most prevalent genotype (Bickerstaff, 2015; Chhatwal et al., 2015; Najafzadeh et al., 2015; Younossi et al., 2015). That is, even at the high prices, the benefits from treatment outweigh the costs (Van Nuys et al., 2015a). For example, Chhatwal and colleagues (2015) found that for treatment-naïve patients with genotype 1 HCV, the incremental cost-effectiveness ratio11 for sofosbuvir-based therapies compared to older interferon therapies ranged from $31,452 to $9,703 per quality-adjusted life year.
It is difficult to say how much society is willing to pay for one quality-adjusted life year; $100,000 is a common, if conservative, threshold (Hirth et al., 2000; Neumann et al., 2014). By this measure, sofosbuvir-based therapies are highly cost-effective. The incremental cost-effectiveness for patients with genotype 1 who had already tried other treatments were higher (ranging from $35,853 to $79,238), but well below the $100,000 threshold (Giovanni et al., 2016). Sofosbuvir-ledipasvir treatments show similar cost-effectiveness for patients with genotype 1 HCV (Najafzadeh et al., 2015; Younossi et al., 2015). But there are some patients for whom sofosbuvir-based treatments might not be cost-effective, such as people with genotype 2 or 3 HCV who had failed earlier treatment (Chhatwal et al., 2015). Treating 1.6 million cases of chronic hepatitis C with direct-acting agents would cost about $49 billion ($65 billion in direct costs, less $16 billion in offsets) (Chhatwal et al., 2015). Assuming society is willing to pay $100,000 for a quality-adjusted year of life, the benefits of treatment are $88 billion, exceeding the costs by $39 billion (Chhatwal et al., 2015).
Second, proponents of the current pricing model maintain that high prices are needed to encourage innovation. Most economists agree that there is a trade-off in lowering prices; such action will improve access to existing treatments but reduce the pace of innovation or access to future treatments (Lakdawalla and Sood, 2012). This is why most developed countries have patent regimes, to encourage innovation by allowing the patent holders to charge higher prices and increase revenues from new products for the length of the patent. Research on the effects of patent length on innovation is scant, but several studies show that pharmaceutical research and develop-
___________________
11 The incremental cost-effectiveness ratio is the cost of adding one quality-adjusted life year due to an intervention. When the incremental cost-effectiveness ration is $50,000, for example, it can be interpreted as costing society $50,000 to gain one quality-adjusted life year.
ment are responsive to revenues or market size (Acemoglu and Linn, 2004; Blume-Kohout and Sood, 2013). By some estimates, a 10 percent change in market size leads to about a 30 percent change in innovation measured as new drug approvals (Blume-Kohout and Sood, 2013). Third, the initial high prices on the drug are temporary; ultimately, such prices are thought to encourage competition. For example, as new therapies have come on the market, discounted prices or revenue per patient initiating therapy with Sovaldi® and Harvoni® has fallen from $75,000 in 2014 to $52,000 in 2015 (Gilead Sciences, 2015a,d).
Eliminating HCV infection would require near universal access to treatment, something that appears infeasible given the current pricing and policy environment. The committee believes, however, that there is room for creative solutions that promote universal access to treatment while maintaining incentives for innovation. Potential solutions might include licensing deals where insurers provide universal access and pharmaceutical firms offer volume discounts or rebates to offset some of the costs. Or, as in the Vaccines for Children program, the federal government could serve as the negotiator of a discounted price for poor patients. There might also be need for regulations to correct for market failures in private insurance markets. For example, private insurers might be reluctant to cover new treatments due to fear of adverse selection: HCV-infected clients dropping coverage from stricter plans and enrolling in more generous ones. Before the Affordable Care Act, exclusions on insurance coverage for preexisting conditions made such moves impossible; now insurers have reason to fear them.
It might also be possible to expand insurance coverage and safety net programs for uninsured HCV patients as the Ryan White Program and AIDS Drug Assistance Program did for poor HIV patients (HRSA, 2016). The parallels between response to HIV and HCV should not be overstated, however. HCV is more than twice as common in the United States as HIV (Edlin et al., 2015). As of 2007, it also causes more annual deaths (Ly et al., 2012). But far fewer resources are allocated to HCV prevention, testing, treatment, and research efforts than to HIV. Unless the disease is a public priority, as HIV was from the 1990s on, it will be difficult to marshal funds for any elimination program.
Stigma, Injection Drug Use, and Corrections
HIV was not always the global priority it is today. Like HCV, HIV disproportionately affects marginalized people whose circumstances carry a stigma that isolates them. For HIV in the United States the group most affected was men who have sex with men; for HCV it is people who inject drugs. The HIV epidemic encouraged men who have sex with men
to overcome stigma and advocate for people with the disease, improving worldwide access to treatment and services (Sengupta et al., 2011). The same transition is unlikely for HCV. People imprisoned or injecting drugs are, almost by definition, not in good position to rally public support for their health problems.
The social stigma that accompanies HCV can impair the HCV-infected person’s mental health and sense of well-being (Marinho and Barreira, 2013). These feelings in turn can contribute to poor treatment uptake and retention in care (Grebely and Dore, 2014; Zeremski et al., 2013). Providers might also be driving the stigma that keeps HCV-infected people from care (Ahern et al., 2007; Paterson et al., 2007; Valdiserri, 2002). Doctors may feel uncomfortable or uncertain when treating people who inject drugs, something often interpreted as intentional mistreatment (Merrill et al., 2002) Stigmatized people tend to internalize perceived judgments, developing a self-image of incompetence and dangerousness; perceptions of stigma can lead the HCV infected person to avoid medical care entirely (Link and Phelan, 2006; Miller et al., 2012). So stigma could undermine the best hepatitis C elimination program by keeping the population at greatest risk for transmitting the virus away from treatment (Treloar et al., 2013).
About 60 percent of prevalent hepatitis C cases in the United States are associated with injection drug use (McGowan and Fried, 2012). Drug abuse, in turn, can be both a cause and an effect of mental health problems. Prior illicit drug use in HCV patients has been linked to lower levels of social support and increased self-report of depression and anxiety (Blasiole et al., 2006). Almost 30 percent of chronic hepatitis C patients in a cohort study met the criteria for depression, though sustained virologic response post-treatment was found to improve these symptoms (Boscarino et al., 2015). Good-quality, nonjudgmental medical care can have benefits beyond improvement in markers of HCV. Such treatment can improve social and mental stability and decrease high-risk behaviors in people who inject drugs (Newman et al., 2013).
The Opportunity to Treat HCV in Corrections
Injection drug use is also a strong predictor of lifetime likelihood of incarceration (DeBeck et al., 2009; Milloy et al., 2008). By a conservative reckoning, between 24 and 36 percent of people addicted to heroin pass through the American prison system each year (Boutwell et al., 2007). Drug users are frequently and repeatedly incarcerated. The number of people incarcerated grew by 33 percent in the decade ending in 2006, but the proportion of prisoners with a drug problem grew by 10 percentage points more (Dolan et al., 2015; Walmsley, 2014). Sixty-five percent of inmates in the United States meet diagnostic criteria for substance abuse disorder;
though incarceration does not appear to reduce drug use (DeBeck et al., 2009; The National Center on Addiction and Substance Abuse at Columbia University, 2010). Only 11.2 percent of inmates with addiction receive treatment for their condition while incarcerated (The National Center on Addiction and Substance Abuse at Columbia University, 2010).
Of the roughly 6.9 million adults12 estimated to be involved in the correction system, over a third are in state or federal prisons or county jails; the rest are in community corrections including probation and parole (Glaze and Kaeble, 2014). Prisons are generally larger than jails and sentence inmates for extended periods of time, though some large jails (including Rikers, Cook, and Los Angeles County facilities) hold 10,000 people or more (NY DOC, 2016). Jails have rapid population turnover with an average length of stay around 23 days (Subramanian et al., 2015). Though prisons are managed by state governments, jails are usually in domain of county and municipal authorities, though six states13 have unified management of all corrections in one system (Krauth, 1997).
Prisoners are legally entitled to medical care while incarcerated, though there are concerns about the quality of the care provided (Estelle v. Gamble, 1976; Wilper et al., 2009). As state and local taxes support the prison system, there is also a wide variability in what different prisons can afford to provide. Medicaid and federal matching funds are not generally available for inmates unless they are admitted to community hospital for more than 24 hours. Still, prisons present an excellent venue for HCV elimination and control programs. Prison inmates account for between 28.5 and 32.8 percent of the national burden of hepatitis C (Varan et al., 2014). Prevalence in different prisons systems tends to vary between 9.6 and 41.1 percent, with the average national prison prevalence between ten and 20 percent (American Correctional Association, 2015; Larney et al., 2014; Varan et al., 2014).
Prison health systems could be instrumental in hepatitis C elimination, as the prison health authorities have access to an otherwise difficult to reach patient population. Prisons are obligated to provide care, and if scheduled properly, the prison system would allow for treatment with very little loss to follow-up and few missed doses. On the other hand, treating hepatitis C is expensive. Prison health systems are not generally large enough to negotiate a substantial discount on direct-acting agents. As the director of the Rhode Island Department of Corrections explained in an interview with National Public Radio, treating HCV can be assumed to cost roughly $150,000 per patient and hundreds of Rhode Island inmates have chronic
___________________
12 2013 estimate.
13 Alaska, Connecticut, Delaware, Hawaii, Rhode Island, and Vermont.
hepatitis C, but the maximum drug budget for the whole system is $19 million (Gourlay, 2014).
Direct-acting agents have also caused prisons to confront various patient management challenges. Older hepatitis C treatments, which needed to be given with certain foods and several times a day, were logistically incompatible with the prison timetable, but simpler direct-acting regimens are changing that (Hepatitis C Online, 2016). Furthermore, pegylated interferon therapy took about 48 weeks to complete, but a full course of direct-acting agents can be finished in 8 or 12 weeks, making treatment in jails as well as prisons an option (Hepatitis C Online, 2016). Unplanned transfers and releases can disrupt treatment, however (Liu et al., 2014).
Prisons may also be a good venue for drug and alcohol treatment programs. In the first 2 weeks after release, former inmates have more than 12.7 times greater risk of death then other people (95 percent CI: 9.2 to 17.4 percent); their risk of death from drug use is 129 times greater (95 percent CI: 89-186 percent) (Binswanger et al., 2007). Opioid replacement therapy is not typically offered in corrections, despite being the UN and WHO recommended standard of care for all populations (Kastelic et al., 2008; Pecoraro and Woody, 2011).
Prisoners account for almost a third of all hepatitis C cases in the United States (Varan et al., 2014). Treating this population is essential to the success of any elimination program; treating substance use disorder in the same patients could help reduce their risk of reinfection after release. Nevertheless, treating HCV would require more staff and budget that prison health systems can allocate to any one problem.
The questions of logistics and competing priorities that make hepatitis C a challenge for the prison system are found in other parts of society as well. Eliminating the public health problem of HCV in the United States will require attention to the medical management of the disease and the distribution of infection in the population, as well the social factors that put people at risk for infection and those that keep them in care. These are important barriers, and the strategy to overcome them will be discussed in the phase two report.
REFERENCES
AASLD-IDSA (American Association for the Study of Liver Diseases-Infectious Disease Society of America). 2015a. Monitoring patients who are starting hepatitis C treatment, are on treatment, or have completed therapy. http://hcvguidelines.org/full-report/monitoring-patients-who-are-starting-hepatitis-c-treatment-are-treatment-or-have (accessed January 28, 2016).
AASLD-IDSA. 2015b. Unique patient populations: Patients with HIV/HCV coinfection. http://www.hcvguidelines.org/full-report/unique-patient-populations-patients-hivhcv-coinfection (accessed January 28, 2016).
AASLD-IDSA. 2015c. When and in whom to initiate HCV treatment. http://hcvguidelines.org/full-report/when-and-whom-initiate-hcv-therapy (accessed January 28, 2016).
AbbVie. 2015. Abbvie reports third-quarter 2015 financial results.
Acemoglu, D., and J. Linn. 2004. Market size in innovation: Theory and evidence from the pharmaceutical industry. The Quarterly Journal of Economics 119(3):1049-1090.
Aetna. 2015. Pharmacy clinical policy bulletins. http://www.aetna.com/products/rxnonmedicare/data/2014/GI/hepatitis_c.html (accessed January 29, 2016).
Afdhal, N., K. R. Reddy, D. R. Nelson, E. Lawitz, S. C. Gordon, E. Schiff, R. Nahass, R. Ghalib, N. Gitlin, and R. Herring. 2014a. Ledipasvir and sofosbuvir for previously treated HCV genotype 1 infection. New England Journal of Medicine 370(16):1483-1493.
Afdhal, N., S. Zeuzem, P. Kwo, M. Chojkier, N. Gitlin, M. Puoti, M. Romero-Gomez, J.-P. Zarski, K. Agarwal, P. Buggisch, G. R. Foster, N. Bräu, M. Buti, I. M. Jacobson, G. M. Subramanian, X. Ding, H. Mo, J. C. Yang, P. S. Pang, W. T. Symonds, J. G. McHutchison, A. J. Muir, A. Mangia, and P. Marcellin. 2014b. Ledipasvir and sofosbuvir for untreated HCV genotype 1 infection. New England Journal of Medicine 370(20):1889-1898.
Ahern, J., J. Stuber, and S. Galea. 2007. Stigma, discrimination and the health of illicit drug users. Drug and Alcohol Dependence 88(2-3):188-196.
Alter, H. J., and M. Houghton. 2000. Hepatitis C virus and eliminating post-transfusion hepatitis. Nature Medicine 6(10):1082-1086.
Alter, M. J. 1997. Epidemiology of hepatitis C. Hepatology 26(3 Suppl 1):62s-65s.
Alter, M. J. 2002. Prevention of spread of hepatitis C. Hepatology 36(5 Suppl 1):S93-S98.
Alter, M. J. 2006. Epidemiology of viral hepatitis and HIV co-infection. Journal of Hepatology 44(1 Suppl):S6-S9.
Alter, M. J. 2007. Epidemiology of hepatitis C virus infection. World Journal of Gastroenterology 13(17):2436-2441.
American Correctional Association. 2015. Hepatitis C in correctional settings: Challenges and opportunities. Alexandria, VA: American Correctional Association.
Andre, P., F. Komurian-Pradel, S. Deforges, M. Perret, J. L. Berland, M. Sodoyer, S. Pol, C. Brechot, G. Paranhos-Baccala, and V. Lotteau. 2002. Characterization of low- and very-low-density hepatitis C virus RNA-containing particles. Journal of Virology 76:6919-6928.
Angermayr, B., M. Cejna, F. Karnel, M. Gschwantler, F. Koenig, J. Pidlich, H. Mendel, L. Pichler, M. Wichlas, A. Kreil, M. Schmid, A. Ferlitsch, E. Lipinski, H. Brunner, J. Lammer, P. Ferenci, A. Gangl, and M. Peck-Radosavljevic. 2003. Child-Pugh versus MELD score in predicting survival in patients undergoing transjugular intrahepatic portosystemic shunt. Gut 52(6):879-885.
Apostolou, A., M. L. Bartholomew, R. Greeley, S. M. Guilfoyle, M. Gordon, C. Genese, J. P. Davis, B. Montana, and G. Borlaug. 2015. Transmission of hepatitis C virus associated with surgical procedures—New Jersey 2010 and Wisconsin 2011. Morbidity and Mortality Weekly Report 64(07):165-170.
Armstrong, G. L., A. Wasley, E. P. Simard, G. M. McQuillan, W. L. Kuhnert, and M. J. Alter. 2006. The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Annals of Internal Medicine 144(10):705-714.
Backus, L. I., D. B. Boothroyd, B. R. Phillips, P. Belperio, J. Halloran, and L. A. Mole. 2011. A sustained virologic response reduces risk of all-cause mortality in patients with hepatitis C. Clinical Gastroenterology and Hepatology 9(6):509-516.
Baker, B. J. 2016. Making history by curing hepatitis C. Native News Online, February 22.
Bangsberg, D. R., S. Perry, E. D. Charlebois, R. A. Clark, M. Roberston, A. R. Zolopa, and A. Moss. 2001. Non-adherence to highly active antiretroviral therapy predicts progression to AIDS. AIDS 15(9):1181-1183.
Barua, S., R. Greenwald, J. Grebely, G. J. Dore, T. Swan, and L. E. Taylor. 2015. Restrictions for Medicaid reimbursement of sofosbuvir for the treatment of hepatitis C virus infection in the United States. Annals of Internal Medicine 163(3):215-223.
Bickerstaff, C. 2015. The cost-effectiveness of novel direct acting antiviral agent therapies for the treatment of chronic hepatitis C. Expert Review of Pharmacoeconomics & Outcomes Research 15(5):787-800.
Binswanger, I. A., M. F. Stern, R. A. Deyo, P. J. Heagerty, A. Cheadle, J. G. Elmore, and T. D. Koepsell. 2007. Release from prison—a high risk of death for former inmates. New England Journal of Medicine 356(2):157-165.
Blasiole, J. A., L. Shinkunas, D. R. LaBrecque, R. M. Arnold, and S. L. Zickmund. 2006. Mental and physical symptoms associated with lower social support for patients with hepatitis C. World Journal of Gastroenterology 12(29):4665-4672.
BlueCross BlueShield of Mississippi. 2015. Medical policy search: Chronic hepatitis C (HCV). https://www.bcbsms.com/index.php?q=provider-medical-policy-search.html&action=viewPolicy&path=%2Fpolicy%2Femed%2FChronic_Hepatitis_C.html (accessed February 9, 2016).
Blume-Kohout, M. E., and N. Sood. 2013. Market size and innovation: Effects of Medicare Part D on pharmaceutical research and development. Journal of Public Economics 97:327-336.
Bodenheimer, T., E. H. Wagner, and K. Grumbach. 2002. Improving primary care for patients with chronic illness. JAMA 288(14):1775-1779.
Boscarino, J. A., M. Lu, A. C. Moorman, S. C. Gordon, L. B. Rupp, P. R. Spradling, E. H. Teshale, M. A. Schmidt, V. Vijayadeva, and S. D. Holmberg. 2015. Predictors of poor mental and physical health status among patients with chronic hepatitis C infection: The chronic hepatitis cohort study (CHECS). Hepatology 61(3):802-811.
Boutwell, A. E., A. Nijhawan, N. Zaller, and J. D. Rich. 2007. Arrested on heroin: A national opportunity. Journal of Opioid Management 3(6):328-332.
Bravo, A. A., S. G. Sheth, and S. Chopra. 2001. Liver biopsy. New England Journal of Medicine 344(7):495-500.
Brennan, T., and W. Shrank. 2014. New expensive treatments for hepatitis C infection. JAMA 312(6):593-594.
Breskin, A., A. Drobnik, P. Pathela, C. Chan, S. Braunstein, K. Bornschlegel, and J. Fuld. 2015. Factors associated with hepatitis C infection among HIV-infected men who have sex with men with no reported injection drug use in New York City, 2000-2010. Sexually Transmitted Diseases 42(7):382-386.
Bruggmann, P., and J. Grebely. 2015. Prevention, treatment and care of hepatitis C virus infection among people who inject drugs. International Journal of Drug Policy 26(Suppl 1):S22-S26.
Cacoub, P., V. Ratziu, R. P. Myers, P. Ghillani, J. C. Piette, J. Moussalli, and T. Poynard. 2002. Impact of treatment on extra hepatic manifestations in patients with chronic hepatitis C. Journal of Hepatology 36(6):812-818.
Cacoub, P., C. Comarmond, F. Domont, L. Savey, and D. Saadoun. 2015. Cryoglobulinemia vasculitis. American Journal of Medicine 128(9):950-955.
Canary, L. A., R. M. Klevens, and S. D. Holmberg. 2015. Limited access to new hepatitis C virus treatment under state Medicaid programs. Annals of Internal Medicine 163(3):226-228.
CDC (Centers for Disease Control and Prevention). 2010. Viral hepatitis surveillance—United States, 2010. Atlanta, GA: CDC.
CDC. 2013. Surveillance for viral hepatitis—United States, 2013. Atlanta, GA: CDC.
CDC. 2015. Statistics and surveillance. http://www.cdc.gov/hepatitis/statistics/index.htm (accessed February 7, 2016).
CDC. 2016. Hepatitis C FAQs for health professionals. http://www.cdc.gov/hepatitis/hcv/hcvfaq.htm#section1 (accessed January 29, 2016).
Chalasani, N., Z. Younossi, J. E. Lavine, A. M. Diehl, E. M. Brunt, K. Cusi, M. Charlton, and A. J. Sanyal. 2012. The diagnosis and management of non-alcoholic fatty liver disease: Practice guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 55(6):2005-2023.
Chang, L. W., D. Serwadda, T. C. Quinn, M. J. Wawer, R. H. Gray, and S. J. Reynolds. 2013. Combination implementation for HIV prevention: Moving from clinical trial evidence to population-level effects. Lancet Infectious Diseases 13(1):65-76.
Charlton, M., G. T. Everson, S. L. Flamm, P. Kumar, C. Landis, R. S. Brown, Jr., M. W. Fried, N. A. Terrault, J. G. O’Leary, H. E. Vargas, A. Kuo, E. Schiff, M. S. Sulkowski, R. Gilroy, K. D. Watt, K. Brown, P. Kwo, S. Pungpapong, K. M. Korenblat, A. J. Muir, L. Teperman, R. J. Fontana, J. Denning, S. Arterburn, H. Dvory-Sobol, T. Brandt-Sarif, P. S. Pang, J. G. McHutchison, K. R. Reddy, and N. Afdhal. 2015. Ledipasvir and sofosbuvir plus ribavirin for treatment of HCV infection in patients with advanced liver disease. Gastroenterology 149(3):649-659.
Chhatwal, J., F. Kanwal, M. S. Roberts, and M. A. Dunn. 2015. Cost-effectiveness and budget impact of hepatitis C virus treatment with sofosbuvir and ledipasvir in the United States. Annals of Internal Medicine 162(6):397-406.
Cholongitas, E., G. V. Papatheodoridis, M. Vangeli, N. Terreni, D. Patch, and A. K. Burroughs. 2005. Systematic review: The Model for End-stage Liver Disease—should it replace Child-Pugh’s classification for assessing prognosis in cirrhosis? Alimentary Pharmacology and Therapeutics 22(11-12):1079-1089.
Choo, Q. L., G. Kuo, A. J. Weiner, L. R. Overby, D. W. Bradley, and M. Houghton. 1989. Isolation of a cDNA clone derived from a blood-borne non-A, non-B viral hepatitis genome. Science 244(4902):359-362.
Cohen, M. S., Y. Q. Chen, M. McCauley, T. Gamble, M. C. Hosseinipour, N. Kumarasamy, J. G. Hakim, J. Kumwenda, B. Grinsztejn, J. H. S. Pilotto, S. V. Godbole, S. Mehendale, S. Chariyalertsak, B. R. Santos, K. H. Mayer, I. F. Hoffman, S. H. Eshleman, E. PiwowarManning, L. Wang, J. Makhema, L. A. Mills, G. de Bruyn, I. Sanne, J. Eron, J. Gallant, D. Havlir, S. Swindells, H. Ribaudo, V. Elharrar, D. Burns, T. E. Taha, K. Nielsen-Saines, D. Celentano, M. Essex, and T. R. Fleming. 2011. Prevention of HIV-1 infection with early antiretroviral therapy. New England Journal of Medicine 365(6):493-505.
Coleman, K., B. T. Austin, C. Brach, and E. H. Wagner. 2009. Evidence on the chronic care model in the new millennium. Health Affairs 28(1):75-85.
Cox, A. L., D. M. Netski, T. Mosbruger, S. G. Sherman, S. Strathdee, D. Ompad, D. Vlahov, D. Chien, V. Shyamala, S. C. Ray, and D. L. Thomas. 2005. Prospective evaluation of community-acquired acute-phase hepatitis C virus infection. Clinical Infectious Diseases 40:951-958.
Coyle, C., K. Viner, E. Hughes, H. Kwakwa, J. E. Zibbell, C. Vellozzi, and D. Holtzman. 2015. Identification and linkage to care of HCV-infected persons in five health centers—Philadelphia, Pennsylvania, 2012-2014. Morbidity and Mortality Weekly Report 64(17): 459-463.
D’Amico, G., G. Garcia-Tsao, and L. Pagliaro. 2006. Natural history and prognostic indicators of survival in cirrhosis: A systematic review of 118 studies. Journal of Hepatology 44(1):217-231.
Davis, G. L., M. J. Alter, H. El-Serag, T. Poynard, and L. W. Jennings. 2010. Aging of hepatitis C virus (HCV)-infected persons in the United States: A multiple cohort model of HCV prevalence and disease progression. Gastroenterology 138(2):513-521.
Day, C., and L. Topp. 2011. Commentary on Turner et al. (2011): Significant global scale-up of harm reduction interventions and pragmatic research approaches needed to prevent hepatitis C transmission. Addiction 106(11):1989-1990.
de Jong, Y. P., M. Dorner, M. C. Mommersteeg, J. W. Xiao, A. B. Balazs, J. B. Robbins, B. Y. Winer, S. Gerges, K. Vega, R. N. Labitt, B. M. Donovan, E. Giang, A. Krishnan, L. Chiriboga, M. R. Charlton, D. R. Burton, D. Baltimore, M. Law, C. M. Rice, and A. Ploss. 2014. Broadly neutralizing antibodies abrogate established hepatitis C virus infection. Science Translational Medicine 6(254):254ra129.
DeBeck, K., T. Kerr, K. Li, M. J. Milloy, J. Montaner, and E. Wood. 2009. Incarceration and drug use patterns among a cohort of injection drug users. Addiction 104(1):69-76.
DeMaria, A. 2015. Revision of the case definition of hepatitis C for national notification. Council of State and Territorial Epidemiologists: Department of Health and Human Services.
Denniston, M. M., R. M. Klevens, G. M. McQuillan, and R. B. Jiles. 2012. Awareness of infection, knowledge of hepatitis C, and medical follow-up among individuals testing positive for hepatitis C: National health and nutrition examination survey 2001-2008. Hepatology 55(6):1652-1661.
Denniston, M. M., R. B. Jiles, J. Drobeniuc, R. M. Klevens, J. W. Ward, G. M. McQuillan, and S. D. Holmberg. 2014. Chronic hepatitis C virus infection in the United States, National Health and Nutrition Examination Survey 2003 to 2010. Annals of Internal Medicine 160(5):293-300.
Des Jarlais, D. C., T. Perlis, K. Arasteh, L. V. Torian, H. Hagan, S. Beatrice, L. Smith, J. Wethers, J. Milliken, D. Mildvan, S. Yancovitz, and S. R. Friedman. 2005. Reductions in hepatitis C virus and HIV infections among injecting drug users in New York City, 1990-2001. AIDS 19(Suppl 3):S20-S25.
Di Martino, V., P. Rufat, N. Boyer, P. Renard, F. Degos, M. Martinot-Peignoux, S. Matheron, V. Le Moing, F. Vachon, C. Degott, D. Valla, and P. Marcellin. 2001. The influence of human immunodeficiency virus coinfection on chronic hepatitis C in injection drug users: A long-term retrospective cohort study. Hepatology 34(6):1193-1199.
Ditah, I., B. Al Bawardy, H. C. Gonzalez, B. Saberi, C. Ditah, P. S. Kamath, and M. Charlton. 2015. Lack of health insurance limits the benefits of hepatitis C virus screening: Insights from the national health and nutrition examination hepatitis C follow-up study. American Journal of Gastroenterology 110(8):1126-1133.
Dolan, K., B. Moazen, A. Noori, S. Rahimzadeh, F. Farzadfar, and F. Hariga. 2015. People who inject drugs in prison: HIV prevalence, transmission and prevention. International Journal on Drug Policy 26(Suppl 1):S12-S15.
Durham, D. P., L. A. Skrip, R. D. Bruce, S. Vilarinho, E. H. Elbasha, A. P. Galvani, and J. P. Townsend. 2016. The impact of enhanced screening and treatment on hepatitis C in the United States. Clinical Infectious Diseases 62(3):298-304.
Durier, N., C. Nguyen, and L. J. White. 2012. Treatment of hepatitis C as prevention: A modeling case study in Vietnam. PLoS ONE 7(4):e34548.
Edlin, B. R., and E. R. Winkelstein. 2014. Can hepatitis C be eradicated in the United States? Antiviral Research 110:79-93.
Edlin, B. R., T. F. Kresina, D. B. Raymond, M. R. Carden, M. N. Gourevitch, J. D. Rich, L. W. Cheever, and V. A. Cargill. 2005. Overcoming barriers to prevention, care, and treatment of hepatitis C in illicit drug users. Clinical Infectious Diseases 40(Suppl 5):S276-S285.
Edlin, B. R., B. J. Eckhardt, M. A. Shu, S. D. Holmberg, and T. Swan. 2015. Toward a more accurate estimate of the prevalence of hepatitis C in the United States. Hepatology 62(5):1353-1363.
El-Kamary, S. S., R. Jhaveri, and M. D. Shardell. 2011. All-cause, liver-related, and non–liver-related mortality among HCV-infected individuals in the general US population. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America 53(2):150-157.
Estelle v. Gamble, 429 Henry Putzel, Jr., 97 (US Supreme Court 1976).
FDA (Food and Drug Administration). 2013 (unpublished). Guidance for industry: Chronic hepatitis C virus infection: Developing direct-acting antiviral drugs for treatment. Department of Health and Human Services.
Feld, J. J., K. V. Kowdley, E. Coakley, S. Sigal, D. R. Nelson, D. Crawford, O. Weiland, H. Aguilar, J. Xiong, and T. Pilot-Matias. 2014. Treatment of HCV with abt-450/r-ombitasvir and dasabuvir with ribavirin. New England Journal of Medicine 370(17):1594-1603.
Feld, J. J., I. M. Jacobson, C. Hézode, T. Asselah, P. J. Ruane, N. Gruener, A. Abergel, A. Mangia, C.-L. Lai, H. L. Y. Chan, F. Mazzotta, C. Moreno, E. Yoshida, S. D. Shafran, W. J. Towner, T. T. Tran, J. McNally, A. Osinusi, E. Svarovskaia, Y. Zhu, D. M. Brainard, J. G. McHutchison, K. Agarwal, and S. Zeuzem. 2015. Sofosbuvir and velpatasvir for HCV genotype 1, 2, 4, 5, and 6 infection. New England Journal of Medicine 373(27):2599-2607.
Ferenci, P., D. Bernstein, J. Lalezari, D. Cohen, Y. Luo, C. Cooper, E. Tam, R. T. Marinho, N. Tsai, A. Nyberg, T. D. Box, Z. Younes, P. Enayati, S. Green, Y. Baruch, B. R. Bhandari, F. A. Caruntu, T. Sepe, V. Chulanov, E. Janczewska, G. Rizzardini, J. Gervain, R. Planas, C. Moreno, T. Hassanein, W. Xie, M. King, T. Podsadecki, and K. R. Reddy. 2014. Abt-450/r-ombitasvir and dasabuvir with or without ribavirin for HCV. New England Journal of Medicine 370(21):1983-1992.
Foster, G. R., R. D. Goldin, and H. C. Thomas. 1998. Chronic hepatitis C virus infection causes a significant reduction in quality of life in the absence of cirrhosis. Hepatology 27(1):209-212.
Friedman, S. L. 2015. Hepatic fibrosis: Emerging therapies. Digestive Diseases 33(4):504-507. Galbraith, J. W., R. A. Franco, J. P. Donnelly, J. B. Rodgers, J. M. Morgan, A. F. Viles, E. T.
Overton, M. S. Saag, and H. E. Wang. 2015. Unrecognized chronic hepatitis C virus infection among Baby Boomers in the emergency department. Hepatology 61(3):776-782.
George, S. L., B. R. Bacon, E. M. Brunt, K. L. Mihindukulasuriya, J. Hoffmann, and A. M. Di Bisceglie. 2009. Clinical, virologic, histologic, and biochemical outcomes after successful HCV therapy: A 5 year follow-up of 150 patients. Hepatology (Baltimore, Md.) 49(3):729-738.
Ghany, M. G., D. B. Strader, D. L. Thomas, L. B. Seeff, and D. American Association for the Study of Liver. 2009. Diagnosis, management, and treatment of hepatitis C: An update. Hepatology 49(4):1335-1374.
Ghany, M. G., D. R. Nelson, D. B. Strader, D. L. Thomas, and L. B. Seeff. 2011. An update on treatment of genotype 1 chronic hepatitis C virus infection: 2011 practice guideline by the American Association for the Study of Liver Diseases. Hepatology (Baltimore, Md.) 54(4):1433-1444.
Gidwani, R., P. Barnett, J. Goldhaber-Fiebert, S. Asch, J. Lo, S. Dally, and D. Owens. 2015. Uptake and utilization of directly acting antiviral medications for hepatitis C infection in US veterans. Journal of Viral Hepatitis 22(5):489-495.
Gilead Sciences. 2015a. Gilead sciences announces fourth quarter and full year 2014 financial results.
Gilead Sciences. 2015b. Q1 2015 earning results.
Gilead Sciences. 2015c. Q2 2015 earnings results.
Gilead Sciences. 2015d. Q3 2015 earnings results.
Giovanni, M. L., C. P. Angelo, and S. Mario. 2016 (unpublished). Effective but costly: How to tackle difficult tradeoffs in evaluating health improving technologies in liver diseases.
Glasgow, R. E., C. T. Orleans, and E. H. Wagner. 2001. Does the chronic care model serve also as a template for improving prevention? Milbank Quarterly 79(4): iv-v, 579-612.
Glaze, L. E., and D. Kaeble. 2014. Correctional populations in the United States, 2013. Bureau of Justice Statistics.
Gourlay, K. 2014. Costly hepatitis C drugs threaten to bust prison budgets. In NPR.
Grady, B. P., J. Schinkel, X. V. Thomas, and O. Dalgard. 2013. Hepatitis C virus reinfection following treatment among people who use drugs. Clinical Infectious Diseases 57(Suppl 2):S105-S110.
Grebely, J., and G. J. Dore. 2014. Can hepatitis C virus infection be eradicated in people who inject drugs? Antiviral Research 104:62-72.
Grebely, J., K. A. Genoway, J. D. Raffa, G. Dhadwal, T. Rajan, G. Showler, K. Kalousek, F. Duncan, M. W. Tyndall, C. Fraser, B. Conway, and B. Fischer. 2008. Barriers associated with the treatment of hepatitis C virus infection among illicit drug users. Drug and Alcohol Dependence 93(1-2):141-147.
Grebely, J., M. Prins, M. Hellard, A. L. Cox, W. O. Osburn, G. Lauer, K. Page, A. R. Lloyd, and G. J. Dore. 2012. Hepatitis C virus clearance, reinfection, and persistence, with insights from studies of injecting drug users: Towards a vaccine. Lancet Infectious Diseases 12(5):408-414.
Grebely, J., B. Haire, L. E. Taylor, P. Macneill, A. H. Litwin, T. Swan, J. Byrne, J. Levin, P. Bruggmann, G. J. Dore, and U. International Network for Hepatitis in Substance. 2015. Excluding people who use drugs or alcohol from access to hepatitis C treatments—is this fair, given the available data? Journal of Hepatology 63(4):779-782.
Gross, R., W. B. Bilker, H. M. Friedman, and B. L. Strom. 2001. Effect of adherence to newly initiated antiretroviral therapy on plasma viral load. AIDS 15(16):2109-2117.
Hagan, H., H. Thiede, N. S. Weiss, S. G. Hopkins, J. S. Duchin, and E. R. Alexander. 2001. Sharing of drug preparation equipment as a risk factor for hepatitis C. American Journal of Public Health 91(1):42-46.
Hagan, H., H. Thiede, and D. C. Des Jarlais. 2004. Hepatitis C virus infection among injection drug users: Survival analysis of time to seroconversion. Epidemiology 15(5):543-549.
Hagan, H., E. R. Pouget, and D. C. Des Jarlais. 2011. A systematic review and meta-analysis of interventions to prevent hepatitis C virus infection in people who inject drugs. Journal of Infectious Diseases 204(1):74-83.
Harouaka, D., R. E. Engle, K. Wollenberg, G. Diaz, A. B. Tice, F. Zamboni, S. Govindarajan, H. Alter, D. E. Kleiner, and P. Farci. 2016. Diminished viral replication and compartmentalization of hepatitis C virus in hepatocellular carcinoma tissue. Proceedings of the National Academy of Sciences of the United States of America 113(5):1375-1380.
Harris, D. R., R. Gonin, H. J. Alter, E. C. Wright, Z. J. Buskell, F. B. Hollinger, and L. B. Seeff. 2001. The relationship of acute transfusion-associated hepatitis to the development of cirrhosis in the presence of alcohol abuse. Annals of Internal Medicine 134(2):120-124.
Harvard Law School. 2013. Examining hepatitis C virus treatment access: A review of select state Medicaid fee-for-service managed care programs. Cambridge, MA: Health Law & Policy Clinic.
Hellard, M. E., R. Jenkinson, P. Higgs, M. A. Stoove, R. Sacks-Davis, J. Gold, M. Hickman, P. Vickerman, and N. K. Martin. 2012. Modelling antiviral treatment to prevent hepatitis C infection among people who inject drugs in Victoria, Australia. Medical Journal of Australia 196(10):638-641.
Hellard, M., D. A. Rolls, R. Sacks-Davis, G. Robins, P. Pattison, P. Higgs, C. Aitken, and E. McBryde. 2014. The impact of injecting networks on hepatitis C transmission and treatment in people who inject drugs. Hepatology 60(6):1861-1870.
Hellard, M., E. McBryde, R. Sacks Davis, D. A. Rolls, P. Higgs, C. Aitken, A. Thompson, J. Doyle, P. Pattison, and G. Robins. 2015. Hepatitis C transmission and treatment as prevention—the role of the injecting network. International Journal of Drug Policy 26(10):958-962.
Hepatitis C Online, 2016. Treatment of hepatitis C in a correctional setting. University of Washington.
HHS (Department of Health and Human Services). 2015a. Viral hepatitis—prevention and surveillance. https://www.acf.hhs.gov/hhsgrantsforecast/index.cfm?switch=grant.view&gff_grants_forecastInfoID=100000600 (accessed February 9, 2016).
HHS. 2015b. What is the HIV care continuum? https://www.aids.gov/federal-resources/policies/care-continuum (accessed February 8, 2016).
Hirth, R. A., M. E. Chernew, E. Miller, A. M. Fendrick, and W. G. Weissert. 2000. Willingness to pay for a quality-adjusted life year: In search of a standard. Medical Decision Making 20(3):332-342.
Holmberg, S. D., P. R. Spradling, A. C. Moorman, and M. M. Denniston. 2013. Hepatitis C in the United States. New England Journal of Medicine 368(20):1859-1861.
Honeycutt, A. A., J. L. Harris, O. Khavjou, J. Buffington, T. S. Jones, and D. B. Rein. 2007. The costs and impacts of testing for hepatitis C virus antibody in public STD clinics. Public Health Reports (1974-) 122:55-62.
Hoofnagle, J. H., and A. H. Sherker. 2014. Therapy for hepatitis C—the costs of success. New England Journal of Medicine 370(16):1552-1553.
HRSA (Health Resources and Services Administration). 2016. About the Ryan White HIV/AIDS program. http://hab.hrsa.gov/abouthab/aboutprogram.html (accessed February 9, 2016).
Institute for Clinical and Economic Review. 2015. The comparative clinical effectiveness and value of novel combination therapies for the treatment of patients with genotype 1 chronic hepatitis C infection. Boston, MA: Institute for Clinical and Economic Review.
IOM (Institute of Medicine). 2010. Hepatitis and liver cancer: A national strategy for prevention and control of hepatitis B and C. Washington, DC: The National Academies Press.
Joshi, D., M. Pinzani, I. Carey, and K. Agarwal. 2014. Recurrent HCV after liver transplantation-mechanisms, assessment and therapy. Nature Reviews: Gastroenterology & Hepatology 11(12):710-721.
Kanwal, F., M. S. Schnitzler, B. R. Bacon, T. Hoang, P. M. Buchanan, and S. M. Asch. 2010. Quality of care in patients with chronic hepatitis C virus infection: A cohort study. Annals of Internal Medicine 153(4):231-239.
Kastelic, A., J. Pont, and H. Stöver. 2008. Opioid substitution treatment in custodial settings: A practical guide. United Nations Office on Drugs and Crime, World Health Organization.
Kieffer, T. L., S. De Meyer, D. J. Bartels, J. C. Sullivan, E. Z. Zhang, A. Tigges, I. Dierynck, J. Spanks, J. Dorrian, M. Jiang, B. Adiwijaya, A. Ghys, M. Beumont, R. S. Kauffman, N. Adda, I. M. Jacobson, K. E. Sherman, S. Zeuzem, A. D. Kwong, and G. Picchio. 2012. Hepatitis C viral evolution in genotype 1 treatment-naïve and treatment-experienced patients receiving telaprevir-based therapy in clinical trials. PLoS ONE 7(4):e34372.
Kim, W. R., J. B. Gross, Jr., J. J. Poterucha, G. R. Locke, 3rd, and E. R. Dickson. 2001. Outcome of hospital care of liver disease associated with hepatitis C in the United States. Hepatology 33(1):201-206.
Kirk, G. D., S. H. Mehta, J. Astemborski, N. Galai, J. Washington, Y. Higgins, A. Balagopal, and D. L. Thomas. 2013. HIV, age, and the severity of hepatitis C virus-related liver disease: A cohort study. Annals of Internal Medicine 158(9):658-666.
Klenerman, P., and R. Thimme. 2012. T cell responses in hepatitis C: The good, the bad and the unconventional. Gut 61(8):1226-1234.
Klevens, R. M., S. Liu, H. Roberts, R. B. Jiles, and S. D. Holmberg. 2014. Estimating acute viral hepatitis infections from nationally reported cases. American Journal of Public Health 104(3):482-487.
Kohli, A., S. Kattakuzhy, S. Sidharthan, A. Nelson, M. McLaughlin, C. Seamon, E. Wilson, E. G. Meissner, Z. Sims, R. Silk, C. Gross, E. Akoth, L. Tang, A. Price, T. A. Jolley, B. Emmanuel, M. Proschan, G. Teferi, J. Chavez, S. Abbott, A. Osinusi, H. Mo, M. A. Polis, H. Masur, and S. Kottilil. 2015. Four-week direct-acting antiviral regimens in noncirrhotic patients with hepatitis C virus genotype 1 infection: An open-label, nonrandomized trial. Annals of Internal Medicine 163(12):899-907.
Kowdley, K. V., S. C. Gordon, K. R. Reddy, L. Rossaro, D. E. Bernstein, E. Lawitz, M. L. Shiffman, E. Schiff, R. Ghalib, M. Ryan, V. Rustgi, M. Chojkier, R. Herring, A. M. Di Bisceglie, P. J. Pockros, G. M. Subramanian, D. An, E. Svarovskaia, R. H. Hyland, P. S. Pang, W. T. Symonds, J. G. McHutchison, A. J. Muir, D. Pound, and M. W. Fried. 2014. Ledipasvir and sofosbuvir for 8 or 12 weeks for chronic HCV without cirrhosis. New England Journal of Medicine 370(20):1879-1888.
Krauth, B. 1997. A review of the jail function within state unified corrections systems. Washington, DC: US Department of Justice, National Institute of Corrections.
Lakdawalla, D., and N. Sood. 2012. Incentives to innovate. In The Oxford handbook of the economics of the biopharmaceutical industry, edited by P. M. Danzon and S. Nicholson. New York: Oxford University Press.
Lambert, S. M., A. N. Page, J. Wittmann, J. S. Hayllar, C. W. Ferndale, T. M. Bain, and G. A. Macdonald. 2011. General practitioner attitudes to prescribing hepatitis C antiviral therapy in a community setting. Australian Journal of Primary Health 17(3):282-287.
Landon, B. E., I. B. Wilson, K. McInnes, M. B. Landrum, L. Hirschhorn, P. V. Marsden, D. Gustafson, and P. D. Cleary. 2004. Effects of a quality improvement collaborative on the outcome of care of patients with HIV infection: The EQHIV study. Annals of Internal Medicine 140(11):887-896.
Larney, S., M. K. Mahowald, N. Scharff, T. P. Flanigan, C. G. Beckwith, and N. D. Zaller. 2014. Epidemiology of hepatitis C virus in Pennsylvania state prisons, 2004-2012: Limitations of 1945-1965 birth cohort screening in correctional settings. American Journal of Public Health 104(6):e69-e74.
Lauer, G. M. 2013. Immune responses to hepatitis C virus (HCV) infection and the prospects for an effective hcv vaccine or immunotherapies. Journal of Infectious Diseases 207(Suppl 1):S7-S12.
Lee, M. H., H. I. Yang, S. N. Lu, C. L. Jen, S. L. You, L. Y. Wang, C. H. Wang, W. J. Chen, and C. J. Chen. 2012. Chronic hepatitis C virus infection increases mortality from hepatic and extrahepatic diseases: A community-based long-term prospective study. Journal of Infectious Diseases 206:469-477.
Link, B. G., and J. C. Phelan. 2006. Stigma and its public health implications. Lancet 367(9509):528-529.
Liu, S., D. Watcha, M. Holodniy, and J. D. Goldhaber-Fiebert. 2014. Sofosbuvir-based treatment regimens for chronic, genotype 1 hepatitis C virus infection in U.S. incarcerated populations: A cost-effectiveness analysis. Annals of Internal Medicine 161(8):546-553.
Lo Re, V., III, V. K. Amorosa, A. R. Localio, R. O’Flynn, V. Teal, Z. Dorey-Stein, J. R. Kostman, and R. Gross. 2009. Adherence to hepatitis C virus therapy and early virologic outcomes. Clinical Infectious Diseases 48(2):186-193.
Lo Re, V., III, V. Teal, A. R. Localio, V. K. Amorosa, D. E. Kaplan, and R. Gross. 2011. Relationship between adherence to hepatitis C virus therapy and virologic outcomes: A cohort study. Annals of Internal Medicine 155(6):353-360.
Lo Re, V., III, V. Teal, A. R. Localio, V. K. Amorosa, D. E. Kaplan, and R. Gross. 2013. Adherence to hepatitis C virus therapy in HIV/hepatitis C-coinfected patients. AIDS and Behavior 17(1):94-103.
Lo Re, V., III, M. J. Kallan, J. P. Tate, A. R. Localio, J. K. Lim, M. B. Goetz, M. B. Klein, D. Rimland, M. C. Rodriguez-Barradas, A. A. Butt, C. L. Gibert, S. T. Brown, L. Park, R. Dubrow, K. R. Reddy, J. R. Kostman, B. L. Strom, and A. C. Justice. 2014. Hepatic decompensation in antiretroviral-treated patients co-infected with HIV and hepatitis C virus compared with hepatitis C virus-monoinfected patients: A cohort study. Annals of Internal Medicine 160(6):369-379.
Loria, P. 2016. This week in pharma: Questions for experts on Merck’s Zepatier, Portola’s betrixaban. http://seekingalpha.com/article/3869636-week-pharma-questions-experts-mercks-zepatier-portolas-betrixaban (accessed March 14, 2016).
Luu, L. 2015. Liver transplants. http://emedicine.medscape.com/article/776313-overview (accessed January 14, 2016).
Ly, K. N., J. Xing, R. M. Klevens, R. B. Jiles, J. W. Ward, and S. D. Holmberg. 2012. The increasing burden of mortality from viral hepatitis in the United States between 1999 and 2007. Annals of Internal Medicine 156(4):271-278.
Marinho, R. T., and D. P. Barreira. 2013. Hepatitis C, stigma and cure. World Journal of Gastroenterology 19(40):6703-6709.
Martin, A. B., M. Hartman, J. Benson, and A. Catlin. 2015. National health spending in 2014: Faster growth driven by coverage expansion and prescription drug spending. Health Affairs 35(1):150-160.
Martin, N. K., P. Vickerman, and M. Hickman. 2011. Mathematical modelling of hepatitis C treatment for injecting drug users. Journal of Theoretical Biology 274(1):58-66.
Martin, N. K., M. Hickman, S. J. Hutchinson, D. J. Goldberg, and P. Vickerman. 2013a. Combination interventions to prevent HCV transmission among people who inject drugs: Modeling the impact of antiviral treatment, needle and syringe programs, and opiate substitution therapy. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America 57(Suppl 2):S39-S45.
Martin, N. K., P. Vickerman, J. Grebely, M. Hellard, S. J. Hutchinson, V. D. Lima, G. R. Foster, J. F. Dillon, D. J. Goldberg, G. J. Dore, and M. Hickman. 2013b. Hepatitis C virus treatment for prevention among people who inject drugs: Modeling treatment scale-up in the age of direct-acting antivirals. Hepatology (Baltimore, Md.) 58(5):1598-1609.
Maylin, S., M. Martinot-Peignoux, M.-P. Ripault, R. Moucari, A. C. Cardoso, N. Boyer, N. Giuily, C. Castelnau, M. Pouteau, T. Asselah, M. H. Nicolas-Chanoine, and P. Marcellin. 2009. Sustained virological response is associated with clearance of hepatitis C virus RNA and a decrease in hepatitis C virus antibody. Liver International 29(4):511-517.
McGowan, C. E., and M. W. Fried. 2012. Barriers to hepatitis C treatment. Liver International 32:151-156.
McNamara, B. C., P. T. Losikoff, L. Huguenin, G. E. Macalino, J. D. Rich, and S. H. Gregory. 2014. Increasing hepatitis C prevalence and associated risk behaviors among incarcerated young adults. Journal of Urban Health 91(2):376-382.
Mehta, S. H., B. L. Genberg, J. Astemborski, R. Kavasery, G. D. Kirk, D. Vlahov, S. A. Strathdee, and D. L. Thomas. 2008. Limited uptake of hepatitis C treatment among injection drug users. Journal of Community Health 33(3):126-133.
Mehta, S. H., J. Astemborski, G. D. Kirk, S. A. Strathdee, K. E. Nelson, D. Vlahov, and D. L. Thomas. 2011. Changes in blood-borne infection risk among injection drug users. The Journal of Infectious Diseases 203(5):587-594.
Mera, J., and D. Drevets. 2015. Cherokee nation health services HCV elimination project. Paper read at Viral Hepatitis Action Coalition Bi-Annual Meeting, November 13, 2015, San Francisco, CA.
Merrill, J. O., L. A. Rhodes, R. A. Deyo, G. A. Marlatt, and K. A. Bradley. 2002. Mutual mistrust in the medical care of drug users: The keys to the “narc” cabinet. Journal of General Internal Medicine 17(5):327-333.
Miller, E. R., S. McNally, J. Wallace, and M. Schlichthorst. 2012. The ongoing impacts of hepatitis C—a systematic narrative review of the literature. BMC Public Health 12(1):1-13.
Milloy, M. J., E. Wood, W. Small, M. Tyndall, C. Lai, J. Montaner, and T. Kerr. 2008. Incarceration experiences in a cohort of active injection drug users. Drug and Alcohol Review 27(6):693-699.
Mohd Hanafiah, K., J. Groeger, A. D. Flaxman, and S. T. Wiersma. 2013. Global epidemiology of hepatitis C virus infection: New estimates of age-specific antibody to HCV seroprevalence. Hepatology 57(4):1333-1342.
Moorman, A. C., S. C. Gordon, L. B. Rupp, P. R. Spradling, E. H. Teshale, M. Lu, D. R. Nerenz, C. C. Nakasato, J. A. Boscarino, E. M. Henkle, N. J. Oja-Tebbe, J. Xing, J. W. Ward, and S. D. Holmberg. 2013. Baseline characteristics and mortality among people in care for chronic viral hepatitis: The chronic hepatitis cohort study. Clinical Infectious Diseases 56(1):40-50.
Morano, J. P., A. Zelenev, A. Lombard, R. Marcus, B. A. Gibson, and F. L. Altice. 2014. Strategies for hepatitis C testing and linkage to care for vulnerable populations: Point-of-care and standard HCV testing in a mobile medical clinic. Journal of Community Health 39(5):922-934.
Morgan, R. L., B. Baack, B. D. Smith, A. Yartel, M. Pitasi, and Y. Falck-Ytter. 2013. Eradication of hepatitis C virus infection and the development of hepatocellular carcinoma: A meta-analysis of observational studies. Annals of Internal Medicine 158(5 Pt 1):329-337.
Mosley, J. W., E. A. Operskalski, L. H. Tobler, W. W. Andrews, B. Phelps, J. Dockter, C. Giachetti, and M. P. Busch. 2005. Viral and host factors in early hepatitis C virus infection. Hepatology 42:86-92.
Moyer, V. A. 2013. Screening for hepatitis C virus infection in adults: U.S. Preventive services task force recommendation statement. Annals of Internal Medicine 159(5):349-357.
Nachega, J. B., M. Hislop, D. W. Dowdy, R. E. Chaisson, L. Regensberg, and G. Maartens. 2007. Adherence to nonnucleoside reverse transcriptase inhibitor-based HIV therapy and virologic outcomes. Annals of Internal Medicine 146(8):564-573.
Naggie, S., C. Cooper, M. Saag, K. Workowski, P. Ruane, W. J. Towner, K. Marks, A. Luetkemeyer, R. P. Baden, and P. E. Sax. 2015. Ledipasvir and sofosbuvir for HCV in patients coinfected with HIV-1. New England Journal of Medicine 373(8):705-713.
Najafzadeh, M., K. Andersson, W. H. Shrank, A. A. Krumme, O. S. Matlin, T. Brennan, J. Avorn, and N. K. Choudhry. 2015. Cost-effectiveness of novel regimens for the treatment of hepatitis C virus. Annals of Internal Medicine 162(6):407-419.
The National Center on Addiction and Substance Abuse at Columbia University. 2010. Behind bars ii: Substance abuse and America’s prison population. New York: Columbia University.
Negro, F., D. Forton, A. Craxi, M. S. Sulkowski, J. J. Feld, and M. P. Manns. 2015. Extrahepatic morbidity and mortality of chronic hepatitis C. Gastroenterology 149(6):1345-1360.
Neumann, P. J., J. T. Cohen, and M. C. Weinstein. 2014. Updating cost-effectiveness—the curious resilience of the $50,000-per-qaly threshold. New England Journal of Medicine 371(9):796-797.
Newman, A. I., S. Beckstead, D. Beking, S. Finch, T. Knorr, C. Lynch, M. MacKenzie, D. Mayer, B. Melles, and R. Shore. 2013. Treatment of chronic hepatitis C infection among current and former injection drug users within a multidisciplinary treatment model at a community health centre. Canadian Journal of Gastroenterology 27(4):217-223.
NY DOC (New York Department of Correction). 2016. Rikers Island facilities. http://www.nyc.gov/html/doc/html/about/facilities-overview.shtml (accessed March 15, 2016).
Onofrey, S., D. Church, P. Kludt, A. DeMaria, K. Cranston, G. A. Beckett, S. D. Holmberg, J. W. Ward, and D. Holtzman. 2011. Hepatitis C virus infection among adolescents and young adults: Massachusetts, 2002-2009. Morbidity and Mortality Weekly Report 60(17):537-541.
Onofrey, S., J. Aneja, G. A. Haney, E. H. Nagami, J. A. DeMaria, G. M. Lauer, K. Hills-Evans, K. Barton, S. Kulaga, M. J. Bowen, N. Cocoros, B. H. McGovern, D. R. Church, and A. Y. Kim. 2015. Underascertainment of acute hepatitis C virus infections in the U.S. Surveillance system: A case series and chart review. Annals of Internal Medicine 163(4):254-261.
OPTN/SRTR (Organ Procurement and Transplantation Network/Scientific Registry of Transplant Recipients). 2014. Annual data report, liver chapter. Rockville, MD: Department of Health and Human Services.
Osburn, W. O., B. E. Fisher, K. A. Dowd, G. Urban, L. Liu, S. C. Ray, D. L. Thomas, and A. L. Cox. 2010. Spontaneous control of primary hepatitis C virus infection and immunity against persistent reinfection. Gastroenterology 138:315-324.
Osburn, W. O., A. E. Snider, B. L. Wells, R. Latanich, J. R. Bailey, D. L. Thomas, A. L. Cox, and S. C. Ray. 2014. Clearance of hepatitis C infection is associated with the early appearance of broad neutralizing antibody responses. Hepatology 59(6):2140-2151.
Osilla, K. C., G. Wagner, J. Garnett, B. Ghosh-Dastidar, M. Witt, L. Bhatti, and M. B. Goetz. 2011. Patient and provider characteristics associated with the decision of HIV coinfected patients to start hepatitis C treatment. AIDS Patient Care and STDS 25(9):533-538.
Palmateer, N., J. Kimber, M. Hickman, S. Hutchinson, T. Rhodes, and D. Goldberg. 2010. Evidence for the effectiveness of sterile injecting equipment provision in preventing hepatitis C and human immunodeficiency virus transmission among injecting drug users: A review of reviews. Addiction 105(5):844-859.
Parchman, M. L., J. E. Zeber, R. R. Romero, and J. A. Pugh. 2007. Risk of coronary artery disease in type 2 diabetes and the delivery of care consistent with the chronic care model in primary care settings: A STARNet study. Medical Care 45(12):1129-1134.
Patel, E. U., A. L. Cox, S. H. Mehta, D. Boon, C. E. Mullis, J. Astemborski, W. O. Osburn, J. Quinn, A. D. Redd, G. D. Kirk, D. L. Thomas, T. C. Quinn, and O. Laeyendecker. 2016. Hepatitis C igg antibody avidity as a biomarker to estimate population-level incidence. Journal of Infectious Diseases 1(jiw005).
Paterson, B. L., M. Backmund, G. Hirsch, and C. Yim. 2007. The depiction of stigmatization in research about hepatitis C. International Journal of Drug Policy 18(5):364-373.
Pawlotsky, J. M. 2014. New hepatitis C therapies: The toolbox, strategies, and challenges. Gastroenterology 146(5):1176-1192.
Pecoraro, A., and G. E. Woody. 2011. Medication-assisted treatment for opioid dependence: Making a difference in prisons. F1000 Medicine Reports 3:1.
Perumalswami, P. V., S. H. Factor, L. Kapelusznik, S. L. Friedman, C. Q. Pan, C. Chang, F. Di Clemente, and D. T. Dieterich. 2013. Hepatitis outreach network: A practical strategy for hepatitis screening with linkage to care in foreign-born communities. Journal of Hepatology 58(5):890-897.
Petta, S., D. Torres, G. Fazio, C. Camma, D. Cabibi, V. Di Marco, A. Licata, G. Marchesini, A. Mazzola, G. Parrinello, S. Novo, G. Licata, and A. Craxi. 2012. Carotid atherosclerosis and chronic hepatitis C: A prospective study of risk associations. Hepatology 55(5):1317-1323.
The Physicians Foundation. 2014. 2014 survey of America’s physicians: Practice patterns & perspectives. Dallas, TX.
Planas, R., B. Ballesté, M. Antonio Álvarez, M. Rivera, S. Montoliu, J. Anton Galeras, J. Santos, S. Coll, R. Maria Morillas, and R. Solà. 2004. Natural history of decompensated hepatitis C virus-related cirrhosis. A study of 200 patients. Journal of Hepatology 40(5):823-830.
Pollack, A. 2015. High cost of sovaldi hepatitis C drug prompts a call to void its patents. The New York Times, May 20, B3.
Poveda, E., D. L. Wyles, A. Mena, J. D. Pedreira, A. Castro-Iglesias, and E. Cachay. 2014. Update on hepatitis C virus resistance to direct-acting antiviral agents. Antiviral Research 108:181-191.
Poynard, T., P. Bedossa, and P. Opolon. 1997. Natural history of liver fibrosis progression in patients with chronic hepatitis C. The OBSVIRC, METAVIR, CLINIVIR, and DOSVIRC groups. Lancet 349(9055):825-832.
Poynard, T., V. Ratziu, F. Charlotte, Z. Goodman, J. McHutchison, and J. Albrecht. 2001. Rates and risk factors of liver fibrosis progression in patients with chronic hepatitis C. Journal of Hepatology 34(5):730-739.
Poynard, T., J. McHutchison, M. Manns, C. Trepo, K. Lindsay, Z. Goodman, M. H. Ling, and J. Albrecht. 2002. Impact of pegylated interferon alfa-2b and ribavirin on liver fibrosis in patients with chronic hepatitis C. Gastroenterology 122(5):1303-1313.
Poynard, T., R. Morra, P. Halfon, L. Castera, V. Ratziu, F. Imbert-Bismut, S. Naveau, D. Thabut, D. Lebrec, F. Zoulim, M. Bourliere, P. Cacoub, D. Messous, M. Munteanu, and V. de Ledinghen. 2007. Meta-analyses of fibrotest diagnostic value in chronic liver disease. BMC Gastroenterology 7:40.
Pybus, O. G., M. A. Charleston, S. Gupta, A. Rambaut, E. C. Holmes, and P. H. Harvey. 2001. The epidemic behavior of the hepatitis C virus. Science 292:2323-2325.
Ray, S. C., and D. L. Thomas. 2015. Hepatitis C. In Principles and practice of infectious diseases. 8th ed, edited by G. L. Mandell, J. E. Bennett and R. Dolin. Philadelphia, PA: Elsevier Saunders. Pp. 1904-1927.
Ray, S. C., J. R. Bailey, and D. L. Thomas. 2013. Hepatitis C virus. In Fields virology. 6th ed. ed, edited by D. M. Knipe and P. M. Howley. Philadelphia, PA: Lippincott, Williams & Wilkins. Pp. 795-824.
Rein, D. B., J. S. Wittenborn, C. M. Weinbaum, M. Sabin, B. D. Smith, and S. B. Lesesne. 2011. Forecasting the morbidity and mortality associated with prevalent cases of pre-cirrhotic chronic hepatitis C in the United States. Digestive and Liver Disease 43(1):66-72.
Rockstroh, J. K., and U. Spengler. 2004. HIV and hepatitis C virus co-infection. Lancet Infectious Diseases 4(7):437-444.
Rotman, Y., and T. J. Liang. 2009. Coinfection with hepatitis C virus and human immunodeficiency virus: Virological, immunological, and clinical outcomes. Journal of Virology 83(15):7366-7374.
Saag, M. S. 2014. Quantum leaps, microeconomics, and the treatment of patients with hepatitis C and HIV coinfection. JAMA 312(4):347-348.
Sacks-Davis, R., G. Daraganova, C. Aitken, P. Higgs, L. Tracy, S. Bowden, R. Jenkinson, D. Rolls, P. Pattison, G. Robins, J. Grebely, A. Barry, and M. Hellard. 2012. Hepatitis C virus phylogenetic clustering is associated with the social-injecting network in a cohort of people who inject drugs. PLoS ONE 7(10):e47335.
Sanger-Katz, M. 2014. Boon for hepatitis C patients, disaster for prison budgets. The New York Times, August 7.
Sanyal, A. J., M. J. Contos, R. K. Sterling, V. A. Luketic, M. L. Shiffman, R. T. Stravitz, and A. S. Mills. 2003. Nonalcoholic fatty liver disease in patients with hepatitis C is associated with features of the metabolic syndrome. American Journal of Gastroenterology 98(9):2064-2071.
Sarrazin, C., and S. Zeuzem. 2010. Resistance to direct antiviral agents in patients with hepatitis C virus infection. Gastroenterology 138(2):447-462.
Selvarajah, S., and M. P. Busch. 2012. Transfusion-transmission of HCV, a long but successful road map to safety. Antiviral Therapy 17(7PtB):1423.
Sengupta, S., B. Banks, D. Jonas, M. S. Miles, and G. C. Smith. 2011. HIV interventions to reduce HIV/AIDS stigma: A systematic review. AIDS and Behavior 15(6):1075-1087.
Simmons, B., J. Saleem, A. Hill, R. D. Riley, and G. S. Cooke. 2016. Risk of late relapse or reinfection with hepatitis C virus after achieving a sustained virological response: A systematic review and meta-analysis. Clinical Infectious Diseases v1(civ948).
Smith, B. D., R. L. Morgan, G. A. Beckett, Y. Falck-Ytter, D. Holtzman, and J. W. Ward. 2012. Hepatitis C virus testing of persons born during 1945-1965: Recommendations from the centers for disease control and prevention. Annals of Internal Medicine 157(11):817-822.
Steinbrook, R., and R. F. Redberg. 2014. The high price of the new hepatitis C virus drugs. JAMA Internal Medicine 174(7):1172.
Stepanova, M., F. Kanwal, H. B. El-Serag, and Z. M. Younossi. 2011. Insurance status and treatment candidacy of hepatitis C patients: Analysis of population-based data from the United States. Hepatology 53(3):737-745.
Subramanian, R., R. Delaney, S. Roberts, N. Fishman, and P. McGarry. 2015. Incarceration’s front door: The misuse of jails in America. New York: Vera Institute of Justice.
Sulkowski, M. S., D. F. Gardiner, M. Rodriguez-Torres, K. R. Reddy, T. Hassanein, I. Jacobson, E. Lawitz, A. S. Lok, F. Hinestrosa, P. J. Thuluvath, H. Schwartz, D. R. Nelson, G. T. Everson, T. Eley, M. Wind-Rotolo, S.-P. Huang, M. Gao, D. Hernandez, F. McPhee, D. Sherman, R. Hindes, W. Symonds, C. Pasquinelli, and D. M. Grasela. 2014. Daclatasvir plus sofosbuvir for previously treated or untreated chronic HCV infection. New England Journal of Medicine 370(3):211-221.
Suryaprasad, A. G., J. Z. White, F. Xu, B. A. Eichler, J. Hamilton, A. Patel, S. B. Hamdounia, D. R. Church, K. Barton, C. Fisher, K. Macomber, M. Stanley, S. M. Guilfoyle, K. Sweet, S. Liu, K. Iqbal, R. Tohme, U. Sharapov, B. A. Kupronis, J. W. Ward, and S. D. Holmberg. 2014. Emerging epidemic of hepatitis C virus infections among young nonurban persons who inject drugs in the United States, 2006-2012. Clinical Infectious Diseases 59(10):1411-1419.
Tanser, F., T. Bärnighausen, E. Grapsa, J. Zaidi, and M.-L. Newell. 2013. High coverage of art associated with decline in risk of HIV acquisition in rural KwaZulu-Natal, South Africa. Science 339(6122):966-971.
Thein, H. H., Q. Yi, G. J. Dore, and M. D. Krahn. 2008. Estimation of stage-specific fibrosis progression rates in chronic hepatitis C virus infection: A meta-analysis and meta-regression. Hepatology 48(2):418-431.
Thomas, D. L. 2012. Advances in the treatment of hepatitis C virus infection. Topics in Antiviral Medicine 20(1):5-10.
Thomas, D. L., J. Astemborski, R. M. Rai, F. A. Anania, M. Schaeffer, N. Galai, K. Nolt, K. E. Nelson, S. A. Strathdee, L. Johnson, O. Laeyendecker, J. Boitnott, L. E. Wilson, and D. Vlahov. 2000a. The natural history of hepatitis C virus infection: Host, viral, and environmental factors. JAMA 284(4):450-456.
Thomas, D. L., J. Astemborski, D. Vlahov, S. A. Strathdee, S. C. Ray, K. E. Nelson, N. Galai, K. R. Nolt, O. Laeyendecker, and J. A. Todd. 2000b. Determinants of the quantity of hepatitis C virus RNA. Journal of Infectious Diseases 181:844-851.
Tillmann, H. L., M. Wiese, Y. Braun, J. Wiegand, S. Tenckhoff, J. Mossner, M. P. Manns, and K. Weissenborn. 2011. Quality of life in patients with various liver diseases: Patients with HCV show greater mental impairment, while patients with PBC have greater physical impairment. Journal of Viral Hepatitis 18(4):252-261.
Treloar, C., J. Newland, J. Rance, and M. Hopwood. 2010. Uptake and delivery of hepatitis C treatment in opiate substitution treatment: Perceptions of clients and health professionals. Journal of Viral Hepatitis 17(12):839-844.
Treloar, C., J. Rance, and M. Backmund. 2013. Understanding barriers to hepatitis C virus care and stigmatization from a social perspective. Clinical Infectious Diseases 57(Suppl 2):S51-S55.
Trevino, A., C. de Mendoza, P. Parra, C. Rodriguez, A. Madejon, Z. Plaza, J. del Romero, E. Poveda, and V. Soriano. 2011. Natural polymorphisms associated with resistance to new antivirals against HCV in newly diagnosed HIV-HCV-coinfected patients. Antiviral Therapy 16(3):413-416.
Trimoulet, P., C. Belzunce, M. Faure, L. Wittkop, S. Reigadas, M. Dupon, J. M. Ragnaud, H. Fleury, and D. Neau. 2011. Hepatitis C virus (HCV) protease variability and anti-HCV protease inhibitor resistance in HIV/HCV-coinfected patients. HIV Medicine 12(8): 506-509.
Trooskin, S. B., J. Poceta, C. M. Towey, A. Yolken, J. S. Rose, N. L. Luqman, T. W. Preston, P. A. Chan, C. Beckwith, S. C. Feller, H. Lee, and A. S. Nunn. 2015. Results from a geographically focused, community-based HCV screening, linkage-to-care and patient navigation program. Journal of General Internal Medicine 30(7):950-957.
Tseng, F.-C., T. R. O’Brien, M. Zhang, A. H. Kral, B. A. Ortiz-Conde, J. Lorvick, M. P. Busch, and B. R. Edlin. 2007. Seroprevalence of hepatitis C virus and hepatitis B virus among San Francisco injection drug users, 1998 to 2000. Hepatology 46(3):666-671.
Turner, K. M., S. Hutchinson, P. Vickerman, V. Hope, N. Craine, N. Palmateer, M. May, A. Taylor, D. De Angelis, S. Cameron, J. Parry, M. Lyons, D. Goldberg, E. Allen, and M. Hickman. 2011. The impact of needle and syringe provision and opiate substitution therapy on the incidence of hepatitis C virus in injecting drug users: Pooling of UK evidence. Addiction 106(11):1978-1988.
United Health Care. 2015. Clinical pharmacy programs.
University of Washington. 2016. HCV epidemiology in the United States. http://www.hepatitisc.uw.edu/pdf/screening-diagnosis/epidemiology-us/core-concept/all (accessed February 5, 2016).
US Senate Committee on Finance. 2015. Wyden-Grassley sovaldi investigation finds revenue-driven pricing strategy behind $84,000 hepatitis drug. http://www.finance.senate.gov/ranking-members-news/wyden-grassley-sovaldi-investigation-finds-revenue-drivenpricing-strategy-behind-84-000-hepatitis-drug (accessed February, 8, 2016).
USPSTF (US Preventive Services Task Force). 2013. Final recommendation statement: Hepatitis C: Screening. http://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/hepatitis-c-screening#consider (accessed March 9, 2016).
Valdiserri, R. O. 2002. HIV/AIDS stigma: An impediment to public health. American Journal of Public Health 92(3):341-342.
Vallet-Pichard, A., V. Mallet, B. Nalpas, V. Verkarre, A. Nalpas, V. Dhalluin-Venier, H. Fontaine, and S. Pol. 2007. Fib-4: An inexpensive and accurate marker of fibrosis in HCV infection. Comparison with liver biopsy and fibrotest. Hepatology 46(1):32-36.
van de Ven, N., J. Fortunak, B. Simmons, N. Ford, G. S. Cooke, S. Khoo, and A. Hill. 2015. Minimum target prices for production of direct-acting antivirals and associated diagnostics to combat hepatitis C virus. Hepatology 61(4):1174-1182.
Van Nuys, K., R. Brookmeyer, J. W. Chou, D. Dreyfus, D. Dieterich, and D. P. Goldman. 2015a. Broad hepatitis C treatment scenarios return substantial health gains, but capacity is a concern. Health Affairs 34(10):1666-1674.
Van Nuys, K., R. Brookmeyer, J. W. Chou, D. Dreyfus, D. Dieterich, and D. P. Goldman. 2015b (unpublished). Broad hepatitis C treatment scenarios return substantial health gains, but capacity is a concern: Appendix.
Varan, A. K., D. W. Mercer, M. S. Stein, and A. C. Spaulding. 2014. Hepatitis C seroprevalence among prison inmates since 2001: Still high but declining. Public Health Reports 129(2):187-195.
Vargas, R. B., and W. E. Cunningham. 2006. Evolving trends in medical care-coordination for patients with HIV and AIDS. Current HIV/AIDS Reports 3(4):149-153.
Veldt, B. J., E. J. Heathcote, H. Wedemeyer, J. Reichen, W. P. Hofmann, S. Zeuzem, M. P. Manns, B. E. Hansen, S. W. Schalm, and H. L. Janssen. 2007. Sustained virologic response and clinical outcomes in patients with chronic hepatitis C and advanced fibrosis. Annals of Internal Medicine 147(10):677-684.
Venkatesh, S. K., M. Yin, and R. L. Ehman. 2013. Magnetic resonance elastography of liver: Technique, analysis, and clinical applications. Journal of Magnetic Resonance Imaging 37(3):544-555.
Vernon, G., A. Baranova, and Z. M. Younossi. 2011. Systematic review: The epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Alimentary Pharmacology and Therapeutics 34(3):274-285.
Vickerman, P., N. Martin, K. Turner, and M. Hickman. 2012. Can needle and syringe programmes and opiate substitution therapy achieve substantial reductions in hepatitis C virus prevalence? Model projections for different epidemic settings. Addiction 107(11):1984-1995.
Volk, M. L., R. Tocco, S. Saini, and A. S. F. Lok. 2009. Public health impact of antiviral therapy for hepatitis C in the United States. Hepatology 50(6):1750-1755.
Wagner, E. H., B. T. Austin, C. Davis, M. Hindmarsh, J. Schaefer, and A. Bonomi. 2001. Improving chronic illness care: Translating evidence into action. Health Affairs 20(6):64-78.
Wai, C. T., J. K. Greenson, R. J. Fontana, J. D. Kalbfleisch, J. A. Marrero, H. S. Conjeevaram, and A. S. Lok. 2003. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology 38(2):518-526.
Walmsley, R. 2014. World pre-trial/remand imprisonment list. London: International Centre for Prison Studies 3:2015.
Ward, J. W. 2013. The epidemiology of chronic hepatitis C and one-time hepatitis C virus testing of persons born during 1945 to 1965 in the United States. Clinics in Liver Disease 17(1):1-11.
Ward, J. W., and J. H. Mermin. 2015. Simple, effective, but out of reach? Public health implications of HCV drugs. New England Journal of Medicine 373(27):2678-2680.
Weiss, J. J., N. Brau, A. Stivala, T. Swan, and D. Fishbein. 2009. Review article: Adherence to medication for chronic hepatitis C - building on the model of human immunodeficiency virus antiretroviral adherence research. Alimentary Pharmacology and Therapeutics 30(1):14-27.
Weissenborn, K., J. Krause, M. Bokemeyer, H. Hecker, A. Schuler, J. C. Ennen, B. Ahl, M. P. Manns, and K. W. Boker. 2004. Hepatitis C virus infection affects the brain-evidence from psychometric studies and magnetic resonance spectroscopy. Journal of Hepatology 41(5):845-851.
Wells, K. J., T. A. Battaglia, D. J. Dudley, R. Garcia, A. Greene, E. Calhoun, J. S. Mandelblatt, E. D. Paskett, P. C. Raich, and P. Patient Navigation Research. 2008. Patient navigation: State of the art or is it science? Cancer 113(8):1999-2010.
Wilper, A. P., S. Woolhandler, J. W. Boyd, K. E. Lasser, D. McCormick, D. H. Bor, and D. U. Himmelstein. 2009. The health and health care of US prisoners: Results of a nationwide survey. American Journal of Public Health 99(4):666-672.
Wright, N. M. J., and C. N. E. Tompkins. 2006. How can health services effectively meet the health needs of homeless people? The British Journal of General Practice 56(525): 286-293.
Wyles, D. L. 2013. Antiviral resistance and the future landscape of hepatitis C virus infection therapy. Journal of Infectious Diseases 207(Suppl 1):S33-S39.
Yee, H. S., M. F. Chang, C. Pocha, J. Lim, D. Ross, T. R. Morgan, A. Monto, C. R. C. P. Department of Veterans Affairs Hepatitis, and C. P. O. National Hepatitis. 2012. Update on the management and treatment of hepatitis C virus infection: Recommendations from the Department of Veterans Affairs hepatitis C resource center program and the national hepatitis C program office. American Journal of Gastroenterology 107(5):669-689; quiz 690.
Yehia, B. R., A. J. Schranz, C. A. Umscheid, and V. Lo Re. 2014. The treatment cascade for chronic hepatitis C virus infection in the United States: A systematic review and meta-analysis. PLoS ONE 9(7):e101554.
Yoshida, H., Y. Shiratori, M. Moriyama, Y. Arakawa, T. Ide, M. Sata, O. Inoue, M. Yano, M. Tanaka, S. Fujiyama, S. Nishiguchi, T. Kuroki, F. Imazeki, O. Yokosuka, S. Kinoyama, G. Yamada, and M. Omata. 1999. Interferon therapy reduces the risk for hepatocellular carcinoma: National surveillance program of cirrhotic and noncirrhotic patients with chronic hepatitis C in Japan. IHIT study group. Inhibition of hepatocarcinogenesis by interferon therapy. Annals of Internal Medicine 131(3):174-181.
Younossi, Z. M., M. Stepanova, F. Nader, Z. Younossi, and E. Elsheikh. 2013. Associations of chronic hepatitis C with metabolic and cardiac outcomes. Alimentary Pharmacology and Therapeutics 37(6):647-652.
Younossi, Z. M., H. Park, S. Saab, A. Ahmed, D. Dieterich, and S. C. Gordon. 2015. Cost-effectiveness of all-oral ledipasvir/sofosbuvir regimens in patients with chronic hepatitis C virus genotype 1 infection. Alimentary Pharmacology and Therapeutics 41(6):544-563.
Zeiler, I., T. Langlands, J. M. Murray, and A. Ritter. 2010. Optimal targeting of hepatitis C virus treatment among injecting drug users to those not enrolled in methadone maintenance programs. Drug and Alcohol Dependence 110(3):228-233.
Zeremski, M., J. E. Zibbell, A. D. Martinez, S. Kritz, B. D. Smith, and A. H. Talal. 2013. Hepatitis C virus control among persons who inject drugs requires overcoming barriers to care. World Journal of Gastroenterology 19(44):7846-7851.
Zeuzem, S., G. M. Dusheiko, R. Salupere, A. Mangia, R. Flisiak, R. H. Hyland, A. Illeperuma, E. Svarovskaia, D. M. Brainard, W. T. Symonds, G. M. Subramanian, J. G. McHutchison, O. Weiland, H. W. Reesink, P. Ferenci, C. Hezode, and R. Esteban. 2014. Sofosbuvir and ribavirin in HCV genotypes 2 and 3. New England Journal of Medicine 370(21):1993-2001.
Zibbell, J. E., K. Iqbal, R. C. Patel, A. Suryaprasad, K. J. Sanders, L. Moore-Moravian, J. Serrecchia, S. Blankenship, J. W. Ward, and D. Holtzman. 2015. Increases in hepatitis C virus infection related to injection drug use among persons aged ≤30 years—Kentucky, Tennessee, Virginia, and West Virginia, 2006-2012. Morbidity and Mortality Weekly Report 64(17):453-458.
Zoulim, F., T. J. Liang, A. L. Gerbes, A. Aghemo, S. Deuffic-Burban, G. Dusheiko, M. W. Fried, S. Pol, J. K. Rockstroh, N. A. Terrault, and S. Wiktor. 2015. Hepatitis C virus treatment in the real world: Optimising treatment and access to therapies. Gut 64(11):1824-1833.




















































