4 Generating and Applying Knowledge to Improve Trauma Outcomes
In a continuously learning health system, the collection of data and their use to generate knowledge are processes embedded within the practice of medicine. The capture of patient data in trauma registries and their use in trauma center performance improvement and patient safety programs have been standard practice for decades, driven in part by the trauma center verification requirements of the American College of Surgeons. These combined processes facilitate the measurement of trauma outcomes and system performance that is essential to a continuous learning environment for trauma care. The goal of trauma performance improvement is to decrease variations from the standard of care and promote optimal trauma outcomes for each patient, thereby reducing death and disability among injured individuals. Data from registries and performance improvement processes also can inform trauma research and drive the development of new best practices.
At the start of the wars in Afghanistan and Iraq, the military adopted these civilian-sector trauma system practices in the development of its Central Command (CENTCOM) Joint Theater Trauma System, and these learning processes were later centralized under the Joint Trauma System (JTS). Through the capture, collection, and review of trauma care and patient outcome data in the Department of Defense Trauma Registry (DoDTR) and the dissemination of evidence-based clinical practice guidelines designed to reduce practice variation, the JTS works to support con-
tinuous performance improvement across the continuum of care.1 DoDTR data also support the development of new knowledge through research, identifying clinical needs and providing direction to the military’s combat casualty care research program (Pruitt and Rasmussen, 2014), which in turn informs clinical care, closing the learning loop.
Using the learning trauma care system framework laid out in Chapter 3, this chapter presents an assessment of the cyclical process—in both the military and civilian sectors—by which data are captured, new knowledge is generated and transformed into evidence-based best trauma care practices, and those best practices are disseminated within and across systems.
DIGITAL CAPTURE OF THE TRAUMA PATIENT CARE EXPERIENCE
Trauma professionals have been leaders in the early use of data to improve care at the local, regional, state, and national levels. Data have a long-standing role in improving trauma care, facilitating research and performance improvement, necessitating the incorporation of individuals with appropriate data analytic skills as part of the trauma team (discussed in more detail in Chapter 5). The collection of trauma data across the continuum of care, their accurate capture in a registry, and the ability to share them between registries and across systems is critical to the success of a learning trauma care system.
Digital Capture of Military Trauma Data
The Department of Defense Trauma Registry
Trauma registries have been a standard part of civilian trauma system development for decades; at the start of the wars in Afghanistan and Iraq, however, the U.S. Department of Defense (DoD) (with the notable exception of the 75th Ranger Regiment’s Prehospital Trauma Registry; see Chapter 1) had no registry to capture trauma data. As a result, little in the way of patient data was captured during the early years of the Global War on Terror (Bailey et al., 2013). In 2005, the JTS developed the DoDTR, then known as the Joint Theater Trauma Registry (JTTR), which was modeled after the National Trauma Data Bank (NTDB) (Eastridge et al., 2009; Pruitt and Rasmussen, 2014). The DoDTR is the largest repository of combat
__________________
1 The mission of the JTS is to “improve trauma care delivery and patient outcomes across the continuum of care utilizing continuous performance improvement and evidence-based medicine driven by the concurrent collection and analysis of data maintained in the Department of Defense Trauma Registry” (USAISR, 2016).
injury and trauma management data in history, currently containing more than 130,000 records for nearly 80,000 distinct injury events (Haut et al., 2016).2
Data captured in the DoDTR include injury demographics, anatomic and physiologic parameters, and trauma care processes and outcomes (Blackbourne et al., 2012). These data direct military medical research to areas of greatest need and support continuous performance improvement processes (Bailey et al., 2013; Blackbourne et al., 2012). In addition to these traditional uses of a trauma registry, DoDTR data have been key to communicating battlefield medical issues to medical and line leadership (Eastridge, 2015). A monthly theater trauma surveillance report has guided injury prevention efforts and operational decision making (e.g., aligning trauma team staffing levels and composition, as well as equipment requirements, with battlefield needs) by facilitating the provision of timely advisories to combatant commanders (Blackbourne et al., 2012). The DoDTR also has played a role in policy development. Data from the registry informed then Defense Secretary Gates’s 2009 mandate that medical evacuation times in Afghanistan be reduced to 60 minutes or less (Miles, 2013).
The DoDTR is populated by manual abstraction of data from a multitude of disparate information systems that yield, among other data sources, medical records collected from different points along the continuum of care (Haut et al., 2016). There is no direct link between the electronic medical record systems and the DoDTR; data therefore cannot populate the registry through automated transmission. Although there is redundancy across the different information systems, data from different sources can be inconsistent, necessitating review of each system for optimal comprehensiveness. One noted barrier to trauma data collection has been the reluctance of data source providers to grant access to data for the entire trauma patient population (Spott, 2015). Education on the distinction between the DoDTR and the electronic medical record, as well as the value of the registry, have been necessary to overcome this barrier.
Initially, abstraction of data into the DoDTR was carried out by deployed expert teams of nurses and physicians, but since December 2014, abstraction has occurred at the JTS central site in San Antonio, Texas (Spott, 2015). With the loss of abstraction teams in theater comes the risk of data loss, as registrars cannot have the same kinds of timely discussion with care providers about each case. The abstraction process entails a number of other problems; it is error prone and requires significant financial, human, and technical resources. However, the quality of DoD registry data has been rigorously pursued. Standard data definitions were instituted in 2007 (Spott, 2015), and data integrity has been maintained through stan-
__________________
2 These numbers are accurate as of November 6, 2015.
dard data audits. As resources have diminished with the drawdown of U.S. troops, however, the capacity of the JTS to maintain previous levels of data fidelity has diminished (Eastridge, 2015).
The JTS also has developed specialty modules within the DoDTR that include orthopedics, infectious disease, traumatic brain injury, hearing, and vision. Shared data elements and standard data definitions allow data to be ported automatically into the specialty modules from the higher-level system. However, these modules are underutilized and, more concerning, additional registries are being developed by different subspecialties independently to provide a greater level of data granularity. The committee heard testimony that linkages between registries have not been built into the front-end design of the system and thus will have to be configured on the back end (Eastridge, 2015; Spott, 2015).
Trauma Data Collection Across the Continuum of Care
The military faces unique challenges when collecting data in theater. On the battlefield, medical personnel face hostile, austere, and underresourced environments (Butler and Blackbourne, 2012; Eastridge et al., 2011b). Personnel, logistical, and tactical constraints, in combination with the absence of formal requirements and command leadership ensuring the collection of trauma data across the continuum of care, have resulted in the fragmentation of trauma patient data across multiple data systems (see Figure 4-1).
Collection and integration of prehospital data The challenges of trauma data collection are especially marked in the military prehospital setting, where operational demands hinder documentation of injuries and care delivery by medics. As a result, DoD’s prehospital data collection has been suboptimal, despite evidence that the greatest opportunities for improved outcomes through changes in care are in the prehospital setting (Butler and Blackbourne, 2012; Eastridge et al., 2012). Multiple sources have noted the lack of prehospital data, from point of injury to Role 3 care3 (Butler and Blackbourne, 2012; DHB, 2015; Rotondo et al., 2011). In an analysis of more than 4,000 casualties entered into the DoDTR from 2007 to 2010, a large majority—87 percent— had no documentation of the mechanism and location of injury, prehospital vital signs, or interventions provided prior to arriving at a military treatment facility (MTF) (Eastridge et al., 2011b; Kotwal et al., 2013b). A similar retrospective analysis of DoDTR data entered from 2002 to 2009 showed that only 18.6 percent and 25.4 percent of casualty records for OEF and OIF, respectively, contained prehospital data (Therien et al., 2011).
__________________
3 The echelons of care—Roles 1 through 5—are described in Chapter 2 (see Figure 2-6).
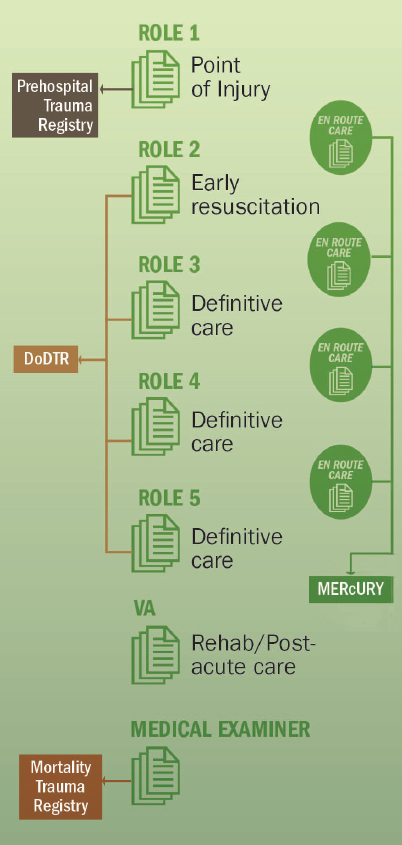
NOTES: DoDTR = Department of Defense Trauma Registry; MERcURY = Military En Route Care Registry. MERcURY is a specialty registry integrated with the DoDTR.
One notable constraint on the collection and sharing of prehospital trauma data is the serious security concern that arises from the potential to link prehospital casualty data with military tactics, techniques, and procedures. As a result, some of these data are designated as sensitive and therefore must be kept in separate classified systems.
Despite the challenges of collecting prehospital data in the combat setting, the 75th Ranger Regiment demonstrated the feasibility and importance of doing so. In 2007, the Committee on Tactical Combat Casualty Care developed a tactical combat casualty care (TCCC) card for prehospital data collection based on the Ranger Casualty Card model (discussed in Chapter 1). The card was updated in 2013 (Kotwal et al., 2013b). Although this form has been endorsed by the Defense Health Board and adopted by DoD as the standard format for documenting prehospital care, it is not used consistently. Information assurance concerns delayed the establishment of the DoD-wide Prehospital Trauma Registry, which was finally fielded in 2013 to collect data on point-of-injury care. As of November 2015, the care records of approximately 750 patients had been entered into the Prehospital Trauma Registry (Haut et al., 2016). Yet despite a 2013 mandate requiring submission of prehospital data (Role 1) to the Prehospital Trauma Registry, documentation of prehospital care remains inconsistent as leadership mandates are not enforced (Kotwal et al., 2013b).
Collection and integration of en route care data In 2014, prehospital transport data, as well as intra- and intertheater transport data, were integrated into the DoDTR. A specialized military en route care registry (MERcURY) was created to document and support analysis of trauma care provided during patient transport by all modes (ground, air, water) (Haut et al., 2016).
Collection and integration of hospital-based acute care data Initially, data were collected for the DoDTR solely from in-theater Role 3 MTFs. Role 4 (Landstuhl Regional Medical Center) data were incorporated starting in 2007, followed by Role 2 (e.g., forward surgical teams) and Role 5 (U.S.-based military hospitals) data in 2008. However, the DoDTR inclusion criteria prior to 2013 required admission to a Role 3 MTF. For those casualties who received care at the Role 2 level and required evacuation to a Role 3 MTF, data on care provided at both the Role 2 and Role 3 facilities were abstracted into the DoDTR, when paper charts from Role 2 were available. Those casualties who did not require evacuation to a Role 3 MTF or who died at a Role 2 facility were therefore not abstracted into the DoDTR; rather, care provided to those patients was abstracted into a separate voluntary Role 2 database, funded as a research study by the U.S. Army Medical Research and Materiel Command (MRMC) (Haut et al., 2016). The U.S. forces commander in Afghanistan mandated prehospital documentation in 2013; however, DoD still has no mandate for Role 2 data collection, so capture of patient data from Role 2 sites has been suboptimal (Kotwal et al., 2016). Such data have been submitted intermittently and on a voluntary basis, primarily by providers who believed
in the value of the registry—a “coalition of the willing” (Spott, 2015). In contrast, the data for Role 3 facilities are nearly complete.
Collection and integration of rehabilitation and post-acute care data For many severely injured military trauma patients, return to duty is not possible, and the patient is discharged from military service. At that point, the patient’s rehabilitative and post-acute care becomes the responsibility of the U.S. Department of Veterans Affairs (VA), a civilian federal agency that operates independently from DoD. Consequently, the vast majority of long-term outcome data for patients treated by the military trauma system resides within the VA. At this time and despite extensive discussion dating to 2006, VA data have not been integrated into the DoDTR. As a result, a paucity of long-term outcome data (including quality-of-life indicators) is available with which to evaluate effects of acute phase interventions and inform performance improvement activities (Spott, 2015).
Collection and integration of Armed Forces Medical Examiner System data For service members killed in action (those who die before reaching an MTF), little to no data may be captured in the DoDTR. Whenever possible, all U.S. combat fatalities are recovered and transferred to the Armed Forces Medical Examiner System (AFMES) for forensic examination. AFMES data are entered into the DoD Mortality Trauma Registry, which enables analysis of service member deaths for trends and modifiable risk factors to inform improvements in equipment, tactics, and casualty care (DHB, 2015). At present, the DoDTR and the Mortality Trauma Registry are and must remain unlinked because of the sensitive nature of mortality data. Although AFMES data have been shared with JTS personnel to enable case-specific studies of preventable trauma deaths (Eastridge et al., 2011a, 2012; Holcomb et al., 2007; Kelly et al., 2008), data sharing is the result of collaborative collegial arrangements, not formal systems for such interface (Spott, 2015). Monthly case reviews of combat trauma deaths by JTS and AFMES personnel have supported performance improvement (DHB, 2015).
Sharing of Patient Data Along the Continuum of Care4
Throughout the wars in Afghanistan and Iraq, patient information from the point of injury to the initial MTF was transmitted primarily by verbal report from the flight medic to the receiving providers. In only 7 percent of cases could a TCCC card be identified in the medical chart. Records
__________________
4 This section draws on information included in the dismounted complex blast injury case study submitted to the committee by DoD (see Appendix A).
from the Role 2 facility were copied and transferred physically with the casualty to the Role 3 MTF. Once the theater of operations became mature, nearly all Role 2 facilities had computers that could link to the Theater Medical Data Store, where medical records are stored. Data entries into the electronic medical record by Role 2 providers were eventually available to providers at Role 3 facilities, where information technology support was much more robust. However, because of bandwidth limitations and periods during which the Internet was blocked as a result of operational security concerns, the use of a hard-copy medical record often remained the most reliable means of ensuring that Role 3 providers received information regarding patient injuries and clinical care previously provided. Hard-copy medical records transferred with the patient were similarly useful to Critical Care Air Transport Teams during the 8- to 10-hour evacuation out of theater.
The reality of patient data sharing and access falls far short of the potential offered by current technology (see Annex 4-1 for an example of optimal data sharing across the continuum of care). Robust systems for both real-time data access and digital capture of care exist and have been implemented successfully in limited cases in the civilian sector. The barrier is one of will.
Digital Capture of Civilian Trauma Data
Civilian-Sector Trauma Registries
In the civilian sector, trauma data are captured in registries and other data repositories at multiple levels. Data roll up from individual trauma centers or emergency medical services (EMS) agencies and ultimately are aggregated in the National EMS Information System (NEMSIS) project’s National EMS Database (prehospital data) and the NTDB (trauma center data).
The NEMSIS project’s National EMS Database, funded by the National Highway Traffic Safety Administration (NHTSA), is the largest collection of prehospital trauma data in the United States. Submission of data to the National EMS Database is voluntary but in 2014, the registry contained nearly 26 million EMS activations submitted by more than 9,600 EMS agencies serving 48 states and territories (Haut et al., 2016). For participating agencies, submission will be automated under the newest version of NEMSIS (Version 3); once a patient care record is complete and has been closed by the local EMS agency, the electronic record is immediately exported to the state repository. A subset of national data elements is then sent from the state repository to the National EMS Database. Data from the National EMS Database are made publically available, and state
and local EMS agencies increasingly use these data to conduct population health investigations and develop performance metrics (Haut et al., 2016). However, the use of these data is limited by the absence of any identifying information in the National EMS Database. While the absence of private information as defined by the Health Insurance Portability and Accountability Act (HIPAA) enables the exchange of information from the local to state to national level, it hinders the ability to associate multiple records over time for the same patient. In addition, the lack of geographic measures makes it impossible to conduct geographic analysis (Haut et al., 2016).
In the hospital setting, all trauma centers utilizing the American College of Surgeons’ trauma center verification process are required to input data into their centers’ trauma registries. As in the military, these trauma registries are populated primarily through manual abstraction of hospital medical records. Submission of hospital trauma registry data to the NTDB also is required for American College of Surgeons trauma center verification. Initiated in 1989 by the American College of Surgeons Committee on Trauma, the NTDB is by far the largest aggregation of civilian hospital trauma registry data, containing more than 6 million records (ACS, 2015). Trauma centers not verified by the American College of Surgeons (i.e., state-designated hospitals) can also submit their data. As of 2015, most Level I (237) and Level II (259) trauma centers were submitting data to the NTDB, although participation of Level III and IV centers was much lower (189) (ACS, 2015; Eastman et al., 2013). A National Trauma Data Standard (NTDS) Data Dictionary helps ensure standard data collection across hospital registries. While the NTDB includes a wealth of information on the acute phase of hospital care for trauma patients, however, it does not include additional data on any delayed complications.
Trauma Data Collection Across the Continuum of Care
The civilian sector faces challenges in aggregating trauma data similar to those experienced by the military, particularly across the phases of care that take place outside the trauma center (see Figure 4-2). As in the military, the civilian sector suffers from missing prehospital data and, with the exception of a few states, its prehospital data do not link to hospital data (Yee, 2015).
Collection and integration of prehospital data In the civilian sector, EMS captures patient data and transmits a record of care to hospital providers using paper or electronic patient care records (Landman et al., 2012). Prior to 2000, little had been accomplished to standardize the collection of prehospital data in the civilian sector. Initiated in 2001, the NEMSIS project
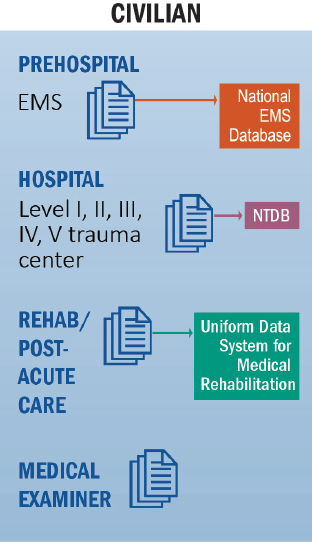
NOTE: EMS = emergency medical services; NTDB = National Trauma Data Bank.
developed a data standard.5 While each local EMS agency has flexibility as to the elements it chooses to collect from the NEMSIS standard, it must include those mandated by the state and required by NEMSIS. Since NEMSIS was initiated, data from an estimated 87 percent of all 911-activated EMS responses have been collected (Haut et al., 2016). However, the NEMSIS project’s National EMS Database does not adequately capture interfacility transfers, critical care transfers, and air medical care.
In addition, registries containing prehospital data are rarely linked to hospital trauma registries at the regional or state level, limiting the extent to which trauma care across the continuum can be analyzed. Although rarely implemented, these linkages are feasible, as demonstrated in a recent study by Seymour and colleagues (2014). In that regional effort, more than 95 percent of EMS records from 33 agencies were successfully linked to hospital electronic medical records within a nine-hospital single system
__________________
5 NEMSIS replaced a smaller prehospital EMS data set (81 elements) developed by the National Highway Traffic Safety Administration in 1994 (NEMSIS, 2013).
in southwestern Pennsylvania, and more than 80 percent of EMS records were linked to long-term outcomes (2-year mortality). This effort offers a scalable model for supporting research and performance improvement at the state/regional level, as well as the eventual testing of financial incentives and reimbursement models for value-based EMS care delivery (Seymour et al., 2014).
Collection and integration of hospital-based data The collection and integration of civilian hospital-based trauma care data improved dramatically with the introduction of the National Trauma Data Standard by the American College of Surgeons Committee on Trauma in 2009. This standard for hospital trauma data collection was developed to avoid variability in data abstraction and reporting among trauma centers, and enables harmonization of data elements common to both NEMSIS and the NTDB. While the NTDB captures trauma data from all American College of Surgeons–verified trauma centers and a number of state-designated trauma centers, it does not capture trauma data from non-trauma center hospitals or state-designated trauma centers not voluntarily submitting their data. This is problematic in that it limits the extent to which it is possible to assess and compare outcomes after injury across all hospitals (trauma and non-trauma center). Furthermore, NTDB data are not linked to outpatient clinic visits, rehabilitation center admission data, or VA data (Haut et al., 2016).
Collection and integration of rehabilitation and post-acute care data Given that the vast majority of patients who sustain a traumatic injury will survive, maximizing an injured patient’s chances of recovering lost function through timely access to rehabilitation services and post-acute care is essential. Unfortunately in the civilian sector, rehabilitation and post-acute care have been identified as the weakest link in a trauma system (Eastman et al., 2013). Data on rehabilitation and post-acute care are neither comprehensively collected6 nor integrated with prehospital and hospital data, severely limiting the ability to conduct longitudinal analyses of patient outcomes across the continuum of care and to conduct system-wide performance improvement efforts.
Collection and integration of coroner and medical examiner data The NEMSIS project’s National EMS Database and the NTDB provide excellent
__________________
6 The Uniform Data System for Medical Rehabilitation, developed with support from the National Institute on Disability and Rehabilitation Research, represents one example of a database for rehabilitation outcomes. Data are aggregated from rehabilitation hospitals, long-term-care hospitals, skilled nursing facilities, as well as pediatric and outpatient rehabilitation programs (Granger et al., 2010).
sources of aggregated prehospital and hospital trauma data for analysis. There are, however, additional sources of data that can be used to gain a more complete understanding of a patient’s care and outcomes across the trauma care continuum. In the military, all service members who die as a result of battle injuries are autopsied to derive information that can be used to improve clinical performance and prevention measures (e.g., change in personal protective equipment). No comparable effort exists in the civilian sector and as a result, little is known about the timing (phases of care) and predominant causes of preventable civilian trauma deaths. The civilian sector would benefit from additional studies of preventable trauma deaths, particularly in the prehospital setting. One study determined the number of preventable prehospital trauma deaths within a single county (Davis et al., 2014); however, this was a rare effort. A common lexicon also is needed to ensure that such analyses are comparable.
Sharing of Patient Data Along the Continuum of Care
The promise of technology has yet to be fully realized for and globally integrated into trauma care. As a result, data from care delivered earlier in the continuum are not reliably available to downstream providers, either for immediate care of the individual patient or in the aggregate for performance improvement or research purposes. Some EMS agencies have equipped their ambulances with GPS-enabled tablet computers that can capture time-stamped vital signs and procedures. However, these data often cannot be submitted electronically to hospitals en route or downloaded directly into the hospital electronic medical record or trauma registry once EMS providers arrive at a trauma center. The computer-generated documentation may need to be printed, scanned into a nonsearchable medical record at a later date, and then reabstracted by a trauma registrar, a process that compromises data fidelity and fails to provide the trauma team immediate access to the information. There are few federal or state incentives to integrate EMS data into electronic medical records (Haut et al., 2016). Gaps in communication are also evident between trauma centers, for example, during interfacility transfers.
Barriers to Data Sharing Within and Between Military and Civilian Data Systems
Complete and accurate collection of patient-level data across the continuum of trauma care is necessary but insufficient to enable the surveillance, performance improvement, and research required to sustain an effective learning trauma care system. To generate information on the effectiveness, quality, and value of care provided, data must also be shared across infor-
mation systems. Both the military and civilian systems suffer from serious gaps in the extent to which data are, or even can be, integrated and shared across systems. For example, although national-level data repositories for both the prehospital and hospital settings have emerged, these systems are not reliably linked with each other or with the DoDTR (see Table 4-1).
Linkage of Military and Civilian Systems
The DoDTR was intentionally designed using the contemporary standards on which both NEMSIS and the NTDB are based, in many cases employing the same data fields and basic data collection techniques. This alignment was intended to facilitate the transfer of lessons learned from the battlefield to the civilian environment and then back from the civilian environment to the military trauma system (Eastridge, 2015). However, the potential for this transfer of information remains largely unrealized, despite the great potential research benefit of wider access to the wealth of trauma data collected over nearly 15 years of war. Currently, the DoDTR does not link to the civilian prehospital (the NEMSIS project’s National EMS Database) or hospital-based (NTDB) data registry, in part because of operational and security concerns that have limited the DoD’s ability to share its data with the civilian sector (Spott, 2015).
Data sharing between DoD and the VA represents a special case of data sharing between the military and civilian sectors, with the challenge arising from the patient’s movement from the military to a civilian system. The failure to link DoD and VA data has been criticized repeatedly (Maldon et al., 2015), with lack of political will being identified as the most significant barrier to such linkage. To facilitate integration of VA data into the DoDTR, abstractors within the VA system could be given access to the DoDTR to port in patient-level rehabilitation and long-term outcome data, but this issue has not been addressed with any sense of urgency (Spott, 2015). Notably, patient data are transferred from DoD to the VA when patients change veteran status so barriers to data sharing appear to be largely unidirectional in nature.
Barriers to Data Sharing Within and Among Trauma Systems
Multiple, significant barriers impede the sharing of trauma data within and among trauma systems. Political and jurisdictional barriers (e.g., between municipal agencies, organizations, and disciplines) continue to delay data sharing. Technical barriers, although surmountable with existing technology, also play a role. Because data collection processes have generally not been designed with the goal of linking data, after-the-fact methods are
TABLE 4-1 Current Major Trauma-Specific Data Repositories in the United States
| Owner | NTDB | NEMSIS | JTS DoDTR and JTS PHTR |
|---|---|---|---|
| ACS COT | NHTSA | JTS | |
| Time frame | In-hospital only | Prehospital only | Prehospital (Role 1); prehospital and interfacility transport; hospital (Roles 2, 3, and 4) |
| Can data be collected electronically/imported from existing patient care records? | Yes for some elements, no for others | Yes | Yes, has this capability, but is limited by TMDS expeditionary framework |
| How much coverage by percentage of patients? | Moderate (all ACS COT trauma centers); in 2015, 746 hospitals submitted data to the NTDB: 237 Level I, 259 Level II, 189 Level III or IV centers, 36 Level I or II pediatric-only centers (ACS, 2015) | Near complete (includes an estimated 87 percent of all 911-activated EMS responses occurring in the United States) | 100 percent for those admitted to a Role 3 facility that met inclusion criteria of DOA, DOW, transfer, admit of ICD-9 code 800-995 when JTTS teams were in combat theater; near complete for OCONUS combat and noncombat casualties; nominal for CONUS noncombat casualties |
required to link independent data sources, a process that demands significant time and effort.
Effective data sharing requires universally accepted standards that provide consistent formats and meanings for data shared among different information systems. Abstraction allows for data system tailoring but results in nonstandard definitions and hence difficulty with sharing and comparing data across independent systems. Electronic data export is an efficient and technologically advanced process that offers reduced errors and real-time data transmission and availability. With electronic data transmission, information systems can be designed so that data capture is integrated seamlessly into the provider workflow. DoD is currently in the process of implementing a new electronic medical record system, providing an opportunity to design the new system using a bottom-up approach, as described in Chapter 3
| What are the coverage gaps? | Only trauma centers; covers a small percentage of Level III/IV trauma centers and does not cover non-trauma center hospitals; only index hospitalization—does not capture readmissions and long-term outcomes; does not include patients seen and discharged from emergency department; does not link data for transfers (cannot tell it is the same patient who left a Level III center and arrived at a Level I center); no prehospital deaths | Does not adequately capture interfacility transfers, critical care transfers, and air medical care; covers only patients transported by EMS; no data on patients transported by “self” transport mode or police vehicle | 2012: mandate for Role 2 data, retrospective entry occurring now 2012: initiated Trauma Infectious Disease Outcome Study (TIDOS) module 2013: started including prehospital (Role 1) data 2013: initiated military orthopedic trauma registry (MOTR) module 2015: started including rehabilitation facility (VA) data 2015: initiated acoustic module |
NOTE: ACS COT = American College of Surgeons Committee on Trauma; CONUS = continental United States; DOA = dead on arrival; DoDTR = Department of Defense Trauma Registry; DOW = died of wounds; EMS = emergency medical services; ICD = International Classification of Diseases; JTS = Joint Trauma System; JTTS = Joint Theater Trauma System; MOTR = military orthopedic trauma registry; NEMSIS = National EMS Information System; NHTSA = National Highway Traffic Safety Administration; NTDB = National Trauma Data Bank; OCONUS = outside the continental United States; PHTR = Pre-Hospital Trauma Registry; TIDOS = Trauma Infectious Disease Outcome Study; TMDS = Theater Medical Data Store; VA = U.S. Department of Veterans Affairs.
SOURCE: Adapted from Haut et al., 2016.
(Garamone, 2015). Use of this approach would enable seamless data capture, including data export into the DoDTR, and support the integration of data into the provider workflow to support continuous improvement and learning. Although some civilian health systems (e.g., Intermountain Healthcare) utilize this bottom-up design and have developed an electronic data export mechanism to populate disease-specific registries (James and Savitz, 2011), most civilian trauma systems rely on manual data abstraction by registrars. Additionally, federal regulations pose multiple barriers to the use and sharing of registry data, particularly for performance improvement (discussed in more detail later in this chapter). No regulations cover the use of preexisting registry data for operational analysis—for example, to examine workflow or numbers of patients to improve a hospital’s efficiency or plan staffing and budgets. Researchers and institutions often have cited HIPAA as the reason for failing to share data. Yet the committee heard
xstestimony that HIPAA does not limit the ability to share data as long as the appropriate business use agreements are in place (Heide, 2015). It is clear that at present, significant confusion surrounding HIPAA limits the willingness of researchers and institutions to share data, both between and within the military and civilian sectors, and guidance on how to be HIPAA compliant when engaging in both research and performance improvement activities would be of great benefit.
The barriers discussed above—political, operational, technical, and regulatory—all limit the extent to which data can be shared in a learning trauma care system. Inadequate data sharing in turn impedes research and performance improvement, limiting the extent to which new knowledge can be generated and providers/systems can continuously learn from each other to improve care and outcomes for traumatically injured patients. Seymour and colleagues (2014), who successfully linked prehospital and hospital data in southwestern Pennsylvania, propose a number of solutions to address specific policy, technical, and regulatory challenges that may limit the linking of data sets and their subsequent use for research (see Table 4-2).
TABLE 4-2 Barriers and Solutions to Linking Prehospital Data to Hospital Data for Research Use
| OBSERVED BARRIERS | POTENTIAL SOLUTIONS |
|---|---|
| EMS records may not be electronic or uploaded into receiving hospital medical records. | Strive towards NEMSIS compliant, electronic prehospital records across all EMS agencies that are automatically uploaded to the receiving hospital records at completion. |
| Prehospital databases may lack capability to be sorted, cleaned, and queried. | Consider proprietary software or local programmers to develop relational databases suitable for cleaning, queries, and linking. |
| EMS agencies lack Data Use Agreements (DUAs) with researchers to participate in studies. | Multi-disciplinary process across university, health system, EMS agency, and legal to develop local DUAs generalizable for others. |
| Unable to access EMS records for patients outside of health system partnership. | Establish DUAs in advance of research questions, perhaps with regional or statewide mandates. |
NOTE: DUA = data use agreement; EMS = emergency medical services; NEMSIS = National EMS Information System.
SOURCE: Reprinted with permission of John Wiley and Sons, from Seymour et al., 2014.
COORDINATED PERFORMANCE IMPROVEMENT AND RESEARCH TO GENERATE EVIDENCE-BASED BEST TRAUMA CARE PRACTICES
Expanding and improving the supply of knowledge is a critical component of a learning trauma care system. Empirical support for best care practices is essential to such a system, as it is to a system for any other serious disease or condition. Facing an unprecedented burden of injury during the wars in Afghanistan and Iraq, the military adopted a nimble approach to evidence generation as part of the learning cycle, termed focused empiricism (see Box 4-1). Evidence and insights derived from the military’s use of this approach have led to the generation and modification of clinical practice guidelines, the identification of questions requiring further research, and the transfer of innovative care practices to the civilian sector (an example is damage control resuscitation, described in Box 4-2). Focused empiricism is part of a learning process that involves determining whether empirical gaps in care exist, and if so, how best to address them. The concept is an integral and pivotal component of the Military Health System’s requirements-driven approach to trauma care evaluation and improvement, the urgency of which is magnified by the devastating effects of enemy weaponry improvisation on the battlefield.
The Military Health System recognizes the importance of obtaining the highest-quality information possible. When feasible, clinical practice guidelines are supported by rigorously acquired data. Infrastructure developed for the primary purpose of performance improvement (e.g., trauma registries) can enable retrospective, outcome-based analyses to identify best practices in trauma care, often driven by provider experiences and observa-
tions in the field. Flexibility in the types of evidence collected and used to guide clinical care (e.g., trauma registries, retrospective chart reviews, observational studies) is imperative. In some areas of evolving care, however, use of such rigorous evidence is limited by, for example, infrequent injury types or nonstandardized approaches to care due to variable injuries and circumstances.
Focused empiricism is a useful and necessary feature of a learning trauma care system in both military and civilian settings, particularly in urgent circumstances when rapid advances in care are required. However, a balanced approach is important. Unchecked focused empiricism can be risky, and a risk-benefit analysis using best available data is important before implementing changes based on a focused empiricism approach. In
addition, it remains important whenever possible to acquire high-quality evidence more systematically; the history of medicine includes numerous cases in which systematic, formal research has refuted observations and expected outcomes. For example, colon injuries were treated by colostomy for decades based in large part on military experience from World War II (Causey et al., 2012). However, an unblinded randomized trial in 1979 initiated a shift in practice that was later validated by several higher-quality studies in the 1990s showing that primary repair of colon injuries was a safe and effective alternative (Chappuis et al., 1991; Sasaki et al., 1995; Stone and Fabian, 1979). In 1998, the Eastern Association for the Surgery of Trauma changed its practice management guidelines in support of primary repair for nondestructive colon wounds (Pasquale and Fabian, 1998).
The application of focused empiricism in the civilian world could undoubtedly be of great value, but again, it is equally important to develop high-quality data systematically. Hypothesis-driven research remains a necessary source of evidence to complement the experiential learning that characterizes focused empiricism. When it is timely and feasible to design a clinical trial and randomize, researchers need to carefully consider what ought to be included in the control (nonintervention) arm of the study. In the Pragmatic, Randomized Optimal Platelet and Plasma Ratios (PROPPR) trial, for example, researchers were unwilling to compare the previous, lower standard ratio of blood products due to the loss of equipoise from empirical experience with newer, higher ratios of blood products for patients with life-threatening bleeding. As a result, the trial compared only high to very high ratios (Holcomb et al., 2015b).
Experiential Learning
The length of time needed to generate evidence using traditional research methods poses significant problems in the wartime context. In the United States, estimates from some studies suggest that just 10-20 percent of clinical decisions are adequately informed by formal evidence generated by clinical research (Darst et al., 2010; IOM, 1985). The trauma community has long been a leader in the use of registry-driven performance improvement to advance best trauma care practices. In the late 1980s, for example, data from 139 North American hospitals were pooled as part of a large-scale effort to examine outcomes of care for injured patients (Champion et al., 1990). The military, driven by the urgency of reducing substantial numbers of battlefield deaths, has expanded this approach so that observations and experience in the clinical setting drive the generation of new knowledge.
Military Sector
Traditional research methodologies, including randomized controlled trials, are expensive and time-consuming (IOM, 2013). In the context of trauma research, an exception from informed consent would be required in most instances which, while possible, would be difficult, time-consuming, and ethically challenging to achieve in a military trauma setting. Given these challenges, traditional hypothesis-driven research does not provide the urgent solutions needed in a combat setting. Recognition of this fact, combined with the commitment of military providers to deliver the best possible care, encouraged military medicine to adopt a fundamentally different approach to trauma care. It is important that this focused empiricism approach, in
which more flexible and timely methods are used in generating evidence to inform best practices in care, remain a paradigm of military medicine.
This process is often initiated by providers’ experiences in the clinical setting and followed by observational studies or retrospective analysis of registry data to support or refute the evidence generated by those experiences. In this context, data from the DoDTR or hospital registries are at the core of the military’s use of focused empiricism, allowing investigators to mine the trauma registry to identify injury trends and differences in patient outcomes. This approach has produced dozens of peer-reviewed papers (see selected examples in Table 4-3) and led to numerous improvements in care—and even breakthroughs (e.g., damage control resuscitation; see Box 4-2)—over the course of the wars in Afghanistan and Iraq.
Once generated, this evidence is integrated into JTS clinical practice guidelines and performance improvement processes at the local and system levels (Eastridge et al., 2010). These “tight decision-making cycles”—in which the identification of a problem is swiftly followed by data collection and analysis and the dissemination of corrective action—work to continuously refine and improve care practices in the combat setting (DHB, 2015). The military’s experience with hemostatic agents (e.g., QuikClot) provides a useful example of the importance of continuous monitoring of patient data when changes in practice are made using a focused empiricism approach. The zeolite powder form of the QuikClot hemostatic agent was replaced
TABLE 4-3 Examples of Registry-Driven Retrospective and Observational Studies
| Topic | Project | Published Research |
|---|---|---|
| Damage control resuscitation | Retrospective chart review—high ratio (1:1.4) of plasma to red blood cells associated with improved mortality (odds ratio 8.6, 95% confidence interval [CI] 2.1–35.2) (see Box 4-2) | Borgman et al., 2007 |
| Tourniquets | Observational study—tourniquet use in the absence of shock strongly associated with survival (90% versus 10%; p < 0.001) | Kragh et al., 2009 |
| Hypothermia | Retrospective review of registry data from 31st Combat Support Hospital—hypothermia found to be an independent predictor of mortality (odds ratio 3.8, 95% CI 2.1–6.9, p < 0.05) | Arthurs et al., 2006 |
| Fasciotomy | Retrospective chart review—fasciotomy revision associated with a nearly fourfold increase in mortality (20% versus 6%, p < 0.01) and delayed fasciotomies associated with twice the rate of major amputation (31% versus 15%) and a threefold higher mortality (19% versus 5%, p < 0.01) | Ritenour et al., 2008 |
with an alternative combat gauze after a number of case reports associated zeolite use with thermal injury (McManus et al., 2007).
The application of this strategy is further demonstrated in the military’s use of performance improvement initiatives to minimize complications related to traumatic injury. The DoDTR tracks more than 80 complications from point of injury through either death or discharge from an MTF (Palm et al., 2012b). In monitoring common posttraumatic complications, the JTS is able to assess emerging trends related to those complications and drive changes in provider care and system processes to reduce complication-related mortality and morbidity. The impact of these system interventions is then measured and used to identify “potential practices for future [performance improvement]” (Palm et al., 2012b, p. S465). Performance improvement initiatives related to three complications—extremity compartment syndrome, venous thromboembolism in amputation patients, and ventilator-associated pneumonia—highlight the unique features of the military’s performance improvement process (Palm et al., 2012b) (see Box 4-3).
The military has successfully used rapid-cycle performance improvement processes to advance care and improve outcomes related to hypothermia, compartment syndrome, burn care, and hemorrhage (to name a few) (Blackbourne, 2009; Ennis et al., 2008; Palm et al., 2012a). Yet despite these successes, this alternative approach to knowledge generation has not been optimized within the military trauma system. Performance improvement depends upon accurate and complete data collection; as discussed in the previous section; however, data acquisition across the trauma care continuum is neither standardized nor centralized. Moreover, the existing performance improvement process is fragmented. While the JTS can and does support performance improvement activities, it does so in CENTCOM alone. Within CENTCOM, the awareness, implementation, and integration of these processes at the local level and across the various levels of care and services vary extensively (DHB, 2015; Rotondo et al., 2011).
Civilian Sector
Although less acknowledged, experiential learning is utilized in a similar fashion in the civilian sector, particularly when no high-quality evidence is available to guide best practices. This is the case in pediatric trauma, for which focused empiricism must be used to determine best practices for operative techniques and resuscitation because of the lack of clinical trial data. More generally, the publication of case reports and use of mortality and morbidity reviews, for example, represent efforts to learn from anecdotal experiences. However, the civilian sector is not driven by the same sense of urgency that motivates the military’s flexibility in its reliance on experiential learning. Rather, the civilian sector’s use of focused empiricism
is less explicit and conscious, and thus more limited in shaping trauma care practices beyond the provider level.
Still, performance improvement programs linked to trauma center registries enable learning from outcomes associated with variation in care in civilian trauma centers. The American College of Surgeons’ verification process ensures that every verified trauma center has a comprehensive, systematic trauma performance improvement and patient safety plan and concurrent registry (with patient information being completed within 60 days). Using trauma registry data, system processes are examined for compliance with national standards of care. These reviews include system, clinical, and individual performance measures. Variances from standards of care (identified through audit filters) are then reviewed in detail to determine whether harm occurred, what processes led to the event, and what measures could mitigate or prevent the event from reoccurring. In addition, the verified trauma centers submit data to the NTDB for national analysis and benchmarking comparison. Hospitals participating in the American College of Surgeons Trauma Quality Improvement Program conduct another level of review and analysis addressing trauma outcomes and benchmarking. These efforts align to foster best practice models for trauma care (ACS, 2014).
With the exception of a few systems (e.g., Intermountain Healthcare), the civilian sector is much slower than the military sector to change guidelines and policies based on experiential learning (Haider et al., 2015)—a reflection of the acknowledged risks of focused empiricism. In some instances, however, this more measured approach also limits the extent to which lessons learned in the military through experiential learning can be translated to the civilian sector. For example, while whole-blood collection kits are made available to all prehospital providers in the military, the availability of these kits in civilian EMS is limited (34 percent) because of the lack of high-quality data supporting whole-blood delivery (Camazine et al., 2015).
Hypothesis-Driven Research
Hypothesis-driven research, both clinical and systems based, is an important source of evidence that informs trauma care. Research drives innovation, including the development of new technologies, drugs, devices, and biological therapeutics, and thus plays a critical role in generating knowledge used to change care practices and improve patient outcomes. However, pragmatic, ethical, scientific, regulatory, and resource challenges may arise in the design, implementation, and funding of randomized controlled clinical trials. By their nature such trials require years to complete, which delays the deployment of the new therapeutic and diagnostic modali-
TABLE 4-4 Traditional and Alternative Study Designs
| Design | Definition |
|---|---|
| Traditional clinical trial | A prospective biomedical or behavioral research study that is designed to test the safety and effectiveness of a therapeutic agent or intervention (e.g., drug, vaccine, device) using consenting human subjects. These trials generally evaluate the use of a single treatment relative to the standard of care in a relatively homogeneous patient population. |
| Observational study | A study in which investigators observe one or more groups of subjects and measure characteristics and outcomes of interest about the subjects without assigning treatments to subjects as part of the research. |
| Cluster-randomized trial | A design in which groups (e.g., school, clinic, household) rather than individuals are randomized to a particular treatment or study arm. These trials are useful when individual randomization is unfeasible (IOM, 2010). |
| Randomized withdrawal trial | Experiments in which subjects who respond positively to an intervention are randomized to continue receiving that intervention or to receive a placebo. This trial design minimizes the time subjects spend receiving a placebo (IOM, 2001, p. 40) and focuses the comparison on the subset of subjects who demonstrate a response to treatment, potentially increasing the ability of the trial to demonstrate benefit. |
| Adaptive clinical trial | A clinical trial design that includes a prospectively planned opportunity for modification of one or more specified aspects of the study design (e.g., randomization ratios, sample size) based on analysis of interim data from study subjects (adapted from FDA, 2015). |
| Platform trial | A clinical trial designed to simultaneously evaluate multiple treatments or combinations of treatment. This design offers the possibility that some treatments may be removed from the trial and others may be added over time (Berry et al., 2015). |
| Pragmatic trial | This type of trial measures treatment effectiveness or the benefit the intervention produces in routine clinical practice, and it accurately reflects variation in patient populations and care delivery (Patsopoulos, 2011). |
| Randomized registry trial | A large-scale, randomized experiment based on data collected from registries and patient records. These trials are designed to minimize the burden of data collection, increase external validity, and reduce the time to dissemination when compared with traditional clinical trials (Lauer and D’Agostino, 2013). |
SOURCE: Adapted from IOM, 2015.
ties being investigated. In addition, randomized controlled trials, although considered by many to be the gold standard, are not the most appropriate research methodology for every research question (Berwick, 2008) and have many limitations particular to the context of acute trauma care. Given the heterogeneity and complexity of traumatic injury, conducting a trial in which pure control is achieved is often impractical. In such cases, alternative study designs may be more appropriate (see Table 4-4). As a result of
these challenges, the military’s trauma care practices and clinical guidelines are informed by a diverse evidence base that includes knowledge generated from basic science and large-animal preclinical research, in addition to the retrospective cohort analyses using DoDTR data discussed above and the consensus of subject matter experts (Elster et al., 2013).
On the other hand, formal research serves as an important complement to and check on the more informal and tacit mechanisms for generating knowledge encouraged in a learning trauma care system. While in some cases—for example, how to deal with unexploded ordinance in a casu-
alty (Lein et al., 1999)7—it is simply not possible to generate supporting evidence through formal research, clinical guidelines should to the extent possible be supported by research studies of appropriate design. In the absence of a randomized controlled trial, it can be difficult to determine the safety and efficacy of a care practice that may have emerged from independent provider experience. A well-designed clinical trial can enable
__________________
7 Personal communication, S. Shackelford, U.S. Department of Defense, to A. Downey, the National Academies of Sciences, Engineering, and Medicine, regarding the military’s focused empiricism approach, December 17, 2015.
investigators to detect any harm and either confirm or refute conclusions derived from a focused empiricism approach, encouraging the widespread adoption of a practice thus derived (Borio, 2015; Holcomb and Hoyt, 2015) (see Box 4-4).
A significant challenge for a learning trauma care system is determining when hypothesis-driven research is imperative so that poorly evidenced or untested clinical practice guidelines and therapies are not implemented prematurely. This is not an easy call and may involve trade-offs between strength of evidence and time to implement changes in care. In part, this determination requires intensive assessment of existing evidence from a variety of sources of limited rigor (e.g., literature reviews, expert opinion, clinical case reports, case series, observational data, retrospective studies, clinical research, and querying of trauma registries), as well as evaluation of the feasibility of generating stronger evidence (e.g., randomized controlled trial data) in the face of practical or ethical considerations. The well-being of our service members who sacrifice significantly for our country invokes a sense of passion, urgency, and impatience in finding new therapeutic modalities to treat their horrific and life-altering injuries.
Military Sector
During the wars in Afghanistan and Iraq, the military invested significantly in requirements-driven, programmed trauma research. DoD’s Combat Casualty Care Research Program addresses clinical gaps as well as research questions emerging from clinician experience and performance improvement data in theater. The program thereby serves as a key intermediary in efforts to advance trauma care capabilities, responding to the identification of knowledge gaps and augmenting the evidence base that supports the generation and dissemination of clinical practice guidelines (Rasmussen et al., 2014) (see Figure 4-3). The overall aim of the program is to deliver knowledge and readily deployable solutions to medical providers on the battlefield so as to reduce mortality and morbidity (Rasmussen et al., 2014). The program’s approach to trauma research is organized around the following portfolios, identified as the areas of greatest need for innovative solutions to optimize outcomes for injured service members:
- neurotrauma and traumatic brain injury
- hemorrhage control and resuscitation
- en route care
- photonics and light-based innovation for severe injury (treatments for tissue injury)
- forward surgical care and intensive critical care (Baer, 2015; MRMC and DHA, 2015)
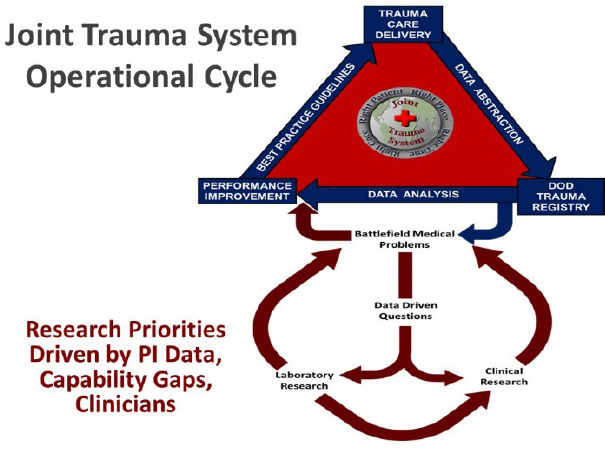
NOTE: DoD = U.S. Department of Defense; PI = performance improvement.
SOURCE: Haut et al., 2016.
The military’s use of a requirements-driven approach in which research is directed toward improving military medical capabilities and closing identified gaps is unique within the broader landscape of federal and private trauma research. In the civilian sector, research is generally investigator driven, and funding opportunities may influence the study topics (Brown, 2015; Rasmussen et al., 2014). DoD’s Combat Casualty Care Research Program reacts directly to battlefield medical problems identified through the DoDTR. In addition, the 2008 DoD report Guidance on Development of the Force specifically identifies 28 gaps relevant to combat casualty care, further directing research priorities (DHB, 2015).
Within DoD, this programmatic research is carried out both individually by each of the services and jointly with funding support from the Defense Health Program (Rasmussen et al., 2014). The Air Force, Army, and Navy each have their own laboratories and facilities for conducting combat casualty care research; an example is the U.S. Army Institute of Surgical Research. The great majority of military research (approximately 80 percent), however, is carried out through the Defense Health Program in collaboration with or at civilian academic centers (Pruitt and Rasmussen, 2014; Rasmussen, 2015).
The development and maturation of the military’s medical research program played a key role in the reduction in the case fatality rate observed over the course of U.S. engagement in Afghanistan and Iraq (Pruitt and Rasmussen, 2014; Rasmussen et al., 2014). At the start of Operation Enduring Freedom and Operation Iraqi Freedom, medical providers lacked basic information and knowledge about how to provide optimal care for trauma patients. Examples of such knowledge gaps included basic questions about resuscitation, such as how much fluid to give, what kind of fluid, and when to stop. The circumstances faced in these wars, including an unprecedented burden of injury and challenging logistical and clinical conditions, necessitated a strongly supported and coordinated military medical research program (Rasmussen et al., 2014). Since the start of these conflicts, military medical research has enabled significant advances in trauma care in such areas as the optimal use of extremity tourniquets, improved outcomes in casualties with traumatic brain injury, damage control resuscitation (see Box 4-2), and burn management (DHB, 2015).
Nonetheless, significant knowledge gaps remain. A 2013 qualitative assessment of the extent to which the gaps identified in DoD’s Guidance on Development of the Force had been closed revealed that the gaps remained less than 50 percent resolved (Rasmussen and Shumacker, 2014). Continued investment in military combat casualty care research is required to achieve further progress and to close existing gaps in trauma care capability.
Civilian Sector
In the civilian sector, a major difference from the military’s approach to research is the absence of a centralized institute dedicated to trauma and emergency care research. A number of research agendas and gap analyses have emerged (EMSC National Resource Center, 2009; NHTSA, 2001; Sayre et al., 2005), aimed at drawing attention to the need for more focused and requirements-driven trauma research in the civilian sector. This need has been highlighted in a number of assessments (see Figure 4-4), a central theme of which is the need for a centralized institute within the National Institutes of Health (NIH) focused on trauma and emergency care (see Table 4-5). NIH has established an Office of Emergency Care Research to coordinate and foster research efforts related to trauma and emergency care within its existing institutes. However, this office is unfunded, which severely limits its influence and ability to direct research to those areas of greatest need (Brown, 2015).
Within the civilian sector, there is some existing infrastructure to support trauma and emergency care research. The Strategies to Innovate
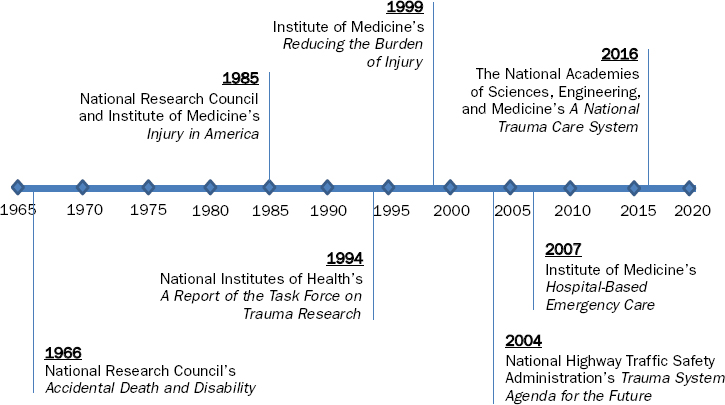
SOURCES: IOM, 1999, 2007b; NHTSA, 2004; NIH, 1994; NRC, 1966; NRC and IOM, 1985.
EmeRgENcy Care Clinical Trials Network8 (SIREN), led by NIH and funded by several institutes as well as DoD, will simultaneously conduct at least four randomized controlled trials focused on patients with neurological, cardiac, pulmonary, hematologic, and traumatic emergencies (Berger, 2016). Proposals for SIREN trials were due in mid 2016, with funds released in early 2017 (NIH, 2016c). In addition, NIH’s National Heart, Lung, and Blood Institute funds the Clinical Trials Network for the Prevention and Early Treatment of Acute Lung Injury. This network conducts randomized controlled trials for acute respiratory distress syndrome, which may be triggered by trauma, infection, or sepsis (PETAL Network, 2015).
While the networks described above are positive examples of trauma and emergency care research under way in the civilian sector, it is important to note that critical systems research may require different kinds of infrastructure (e.g., linked prehospital and hospital databases as described earlier in this chapter) that does not currently exist on a wide scale. Further, no mechanism exists for directing research investments toward identified gaps, despite the repeated recommendations shown in Table 4-5. As highlighted by the Institute of Medicine (IOM, 2007b, p. 12) report Hospital-Based Emergency Care, “The current uncoordinated approach to organizing and
__________________
8 This network replaces the Neurological Emergencies Treatment Trials Network, or NETT, and the Resuscitations Outcomes Consortium, or ROC (Berger, 2016). Among its numerous successes, ROC funded the Pragmatic Randomized Optimal Platelet and Plasma Ratios (PROPPR) trial highlighted in Box 4-2.
| Report | Recommendations |
|---|---|
| NRC, 1966 | “Expansion within the U.S. Public Health Service of research in shock, trauma, and emergency medical conditions, with the goal of establishing a National Institute of Trauma” (p. 34). |
| NRC and IOM, 1985 | “The committee recommends that funding for research on injury be commensurate with the importance of injury as the largest cause of death and disability of children and young adults in the United States” (p. 3). |
| NIH, 1994 | “Creation of an Office of Trauma Research at NIH reporting to the Director of NIH and, in addition, to Congress and the President on an annual basis. The purpose of the Office would be to update the national plan for trauma research and to ensure that this agenda is being addressed and funded” (p. 5). “If the goals and research priorities outlined here are not being substantially addressed . . . then the establishment of a National Institute of Trauma Research should be pursued” (p. 6). |
| IOM, 1999 | “The committee supports a greater focus on trauma research and training at the National Institutes of Health (NIH) and recommends that the National Institute of General Medical Sciences (NIGMS) elevate its existing trauma and burn program to the level of a division” (p. 11). |
| NHTSA, 2004 | “Congress will establish a National Institute for Injury, within the National Institutes of Health” (p. 26). |
| IOM, 2007b | “the Secretary of the Department of Health and Human Services [should] conduct a study to examine the gaps and opportunities in emergency and trauma care research, and recommend a strategy for the optimal organization and funding of the research effort. This study should include . . . improved research coordination through a dedicated center or institute” (p. 12). |
funding emergency and trauma care has been inadequate. There are well-defined emergency and trauma care research questions that would benefit from a coordinated and well-funded research strategy.” Examples of high-priority trauma research needs (clinical and systems research) compiled by the committee, applicable to both the military and civilian sectors, are presented in Table 4-6. As these research needs are consistent across both sectors, most of these questions can be studied in the civilian setting and applied in the military.
Military and Civilian Collaborative Research
DoD has only a single Level I trauma center that treats civilian patients. As a result, patient populations at MTFs are not large enough to support
TABLE 4-6 Examples of High-Priority Trauma Research Needs
| Topic Area | Research Priorities |
|---|---|
| Prevention and epidemiology |
|
| Resuscitation |
|
| Prehospital care |
|
| Burn care |
|
| Pain management |
|
| Topic Area | Research Priorities |
|---|---|
| Traumatic Brain Injury |
|
| Spine |
|
| Critical care |
|
| Nursing |
|
| Topic Area | Research Priorities |
|---|---|
| Orthopedics |
|
| Rehabilitation |
|
| Systems research |
|
SOURCES: The development of this list was informed by preexisting research priority lists, including but not limited to Butler et al., 2015; CDC, NIH, DoD, and VA Leadership Panel, 2013; Helmick et al., 2012; Kotwal et al., 2013a; NCIPC, 2005; NHTSA, 2001; Sauer et al., 2014; Sayre et al., 2005; van Middendorp et al., 2016.
high-quality trauma-related clinical trials and prospective research studies. Civilian partnerships are therefore essential for carrying out the research needed to deliver improvements in combat casualty care. A robust military–civilian research network is particularly relevant during times of low combat activity. Only when patients are pooled across a large number of civilian centers can requisite sample sizes be achieved. While these partnerships are pursued out of necessity, they offer the added benefit of facilitating the exchange of knowledge between the military and civilian sectors. Lessons learned from extramural research are “translatable from the beginning” and serve to improve outcomes for injured patients in both military and civilian settings (Rasmussen, 2015). METRC—the Major Extremity Trauma Research Consortium—is an example of a military–civilian partnership aimed at advancing the development of military best trauma care practices for limb trauma and to promote translation of military lessons learned to the civilian sector (see Box 4-5).
In addition to METRC, there are multiple examples of trauma research carried out through collaboration between the military and civilian sectors, funded in large part by DoD, including
- two clinical trials comparing the military-developed blood transfusion protocol—damage control resuscitation (see Box 4-2)—with a widely used blood transfusion method (the Prospective, Observational, Multicenter, Major Trauma Transfusion [PROMMTT] study [Holcomb et al., 2013] and the Pragmatic, Randomized Optimal Platelet and Plasma Ratios [PROPPR] study [Holcomb et al., 2015b]);
- three ongoing clinical trials evaluating the role, safety, and efficacy of tranexamic acid (ClinicalTrials.gov, 2015a,b,c); and
- multiple studies on the transfusion of plasma in the prehospital setting (Holcomb et al., 2015a).
The National Research Action Plan (discussed further in Box 7-8 in Chapter 7) has facilitated extensive military–civilian collaboration around traumatic brain injury, significantly increasing awareness of this specific trauma injury (DoD et al., 2013). Recently, for example, DoD and NIH collaboratively developed the Federal Interagency Traumatic Brain Injury Research (FITBIR) informatics system to serve as the secure, centralized database for research on traumatic brain injury. Building on current efforts to standardize data elements, FITBIR will serve as the repository for new data, link to current databases, and allow for data comparison across studies (NIH, 2016a).
Barriers to Timely Generation of Evidence to Inform Best Trauma Care Practices
There is no question that the nation’s investment in trauma research has saved lives, but the end of the wars in Afghanistan and Iraq does not signal a time to reduce that investment. As discussed above, numerous gaps in combat casualty care capabilities remain. The military’s experiences in Afghanistan and Iraq have yielded a wealth of experiential knowledge and innovation in trauma care. The potential benefits of translating this knowledge to the civilian sector are significant, but an increased investment in rigorous, collaborative (military and civilian) research will be required to sustain recent advances, encourage further momentum in closing identified gaps in trauma care, and show definitively the safety and efficacy of
new practices and products and their value in civilian trauma care. Yet barriers to research that can generate evidence to inform best trauma care practices—including inadequate funding for trauma research and federal regulations and their interpretation—will have to be overcome.
Inadequate Funding for Trauma Research
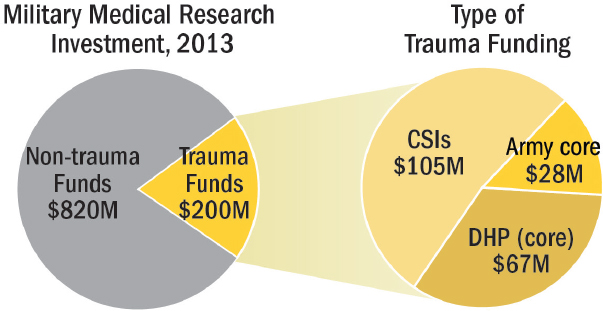
Military-sector trauma research investment There are two major sources of funding for medical research within DoD: the Defense Health Program and Army core funds (Baer, 2015; Rasmussen, 2015). Defense Health Program funding also includes Congressional Special Interest (CSI) funds directed to specific trauma research topics, such as burn care and orthopedic care (Pruitt and Rasmussen, 2014). In 2013, a total of $1.02 billion was allocated for military medical research, $200 million of which was directed toward research on combat casualty care. Of this amount, less than half originated from a core budget (Defense Health Program and Army core funds). The rest was provided by CSIs (Rasmussen, 2015) (see Figure 4-5).
The origin of funding directed at military trauma research threatens the sustainability and efficacy of DoD’s Combat Casualty Care Research Program. Unlike core Defense Health Program and Army funds, CSI funds are distributed on a year-to-year basis, at the whim of politicians. Therefore, the military cannot depend on these funds for future, sustained funding (Rasmussen, 2015).
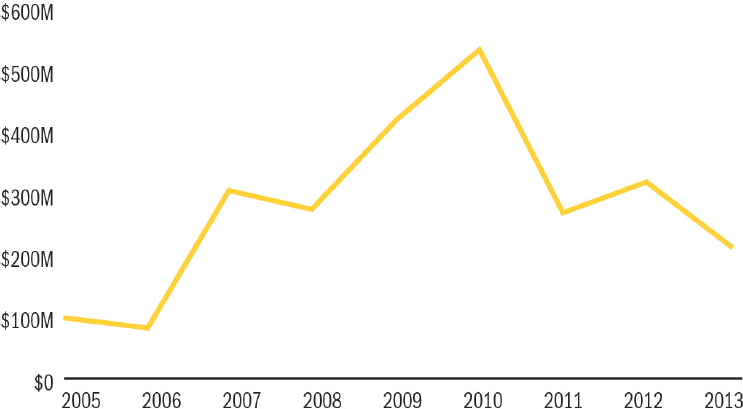
DoD’s investment in research has declined consistently since the drawdown of troops from Afghanistan began (GAO, 2013) (see Figure 4-6). Competing military priorities (e.g., weapon systems) will only further
NOTE: CSI = Congressional Special Interest; DHP = Defense Health Program.
SOURCE: Data from Rasmussen, 2015.
SOURCE: Adapted from GAO (2013, p. 5).
threaten research funding as the transition to an interwar period continues (Di Resta, 2015). Importantly, in contrast with other military research portfolios, such as infectious disease, there is no equivalent to DoD research on combat casualty care in the civilian sector. As a result, military research on combat casualty care has no safety net when DoD investment is cut (Baer, 2015; Rasmussen and Baer, 2014).
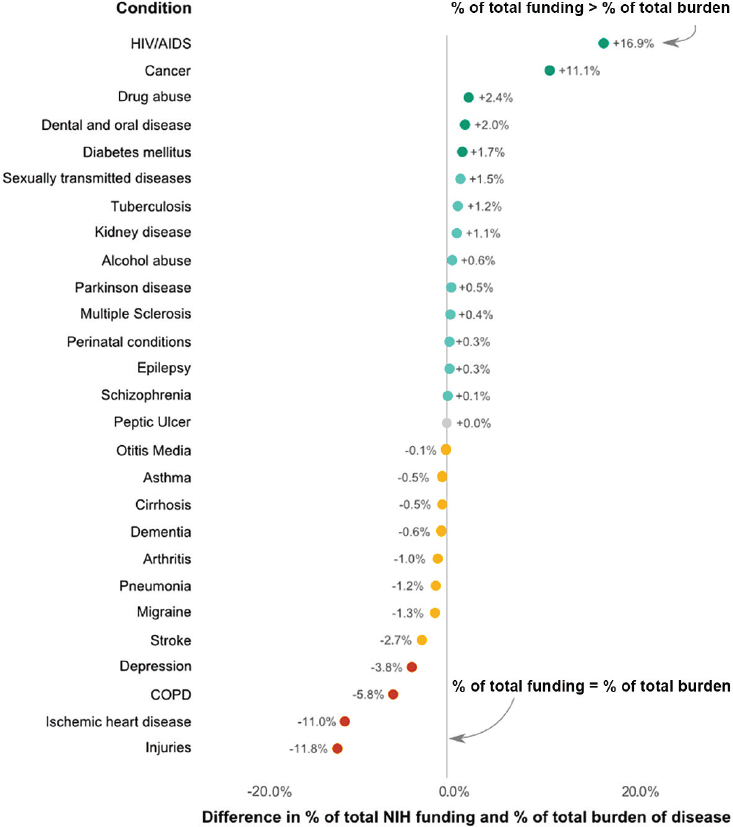
Civilian-sector trauma research investment Exacerbating the reality of dwindling military funds for trauma research is the paucity of civilian-sector investment in trauma research to sustain and further develop advances in trauma care. In 2013, NIH directed just $367 million of its $30 billion budget to trauma (NIH, 2016b), approximately $31.7 million of which was applied to clinical research on the acute phase of trauma care (Brown, 2015; Holcomb and Hoyt, 2015). Despite the significant burden of injury facing the civilian population (as outlined in Chapter 1), federal agencies and private foundations do not dedicate funding to injury research commensurate with its burden to society (Moses et al., 2015; Rhee et al., 2014; Richards, 2015; Trunkey, 1983). In a recent analysis of NIH funding for 27 disease conditions relative to their burden (as measured by disability-adjusted life years [DALYs]), injury was more disproportionately underfunded than any other condition (Moses et al., 2015) (see Figure 4-7). Injury accounts for
NOTE: COPD = chronic obstructive pulmonary disease; HIV/AIDS = human immunodeficiency virus/acquired immunodeficiency syndrome; NIH = National Institutes of Health.
SOURCES: Data from Moses et al., 2015. Created by Catherine A. Richards, Ph.D., M.P.H.
nearly 10 percent of total DALYs in the United States each year9 but receives only about 1 percent of NIH’s biomedical research budget, although the committee acknowledges that proportionality to disease burden is an overly
__________________
9 Injuries accounted for 7,945,100 out of 81,834,600 all-cause DALYs in the United States in 2010 (U.S. Burden of Disease Collaborators, 2013).
simplistic method by which to set research budgets. This disparity between funding and disease burden (as measured by DALYs) may result in part from a lack of patient advocacy and public understanding of trauma and the role of research in addressing gaps in optimal trauma care.
The lack of funding for trauma research highlights a critical limitation of the civilian research enterprise’s investigator-driven approach: “researchers . . . will not pursue clinical problems for which there is little or no funding” (Brown, 2015). As discussed previously, the absence of a designated institute within NIH devoted to trauma research exacerbates this challenge. As expressed to the committee by the director of NIH’s unfunded Office of Emergency Care Research, Dr. Jeremy Brown, “if you are competing with other diseases for which there are institutes, I believe you are always going to lose” (Brown, 2015). Limited funding in the civilian sector results in few high-quality clinical research studies to further advances in trauma care. This is an unfortunate reality, particularly when one considers the history of research on HIV/AIDS, cardiac disease, and cancer, conditions for which increased funding has directly correlated with marked reductions in mortality (Holcomb and Hoyt, 2015).
Federal Regulations and Their Interpretation
The need to protect the rights, safety, and welfare of individual research participants is well accepted. Historical abuses surrounding human research, in particular studies that harmed and willfully exploited participants, gave rise to the federal regulatory landscape in existence today (see Box 4-6 for a brief summary of federal regulations on human subjects protections). In 2005, DoD developed its own human research protection program so that research could be conducted in theater in compliance with all federal regulatory requirements10 (Brosch et al., 2008).
Medical research undoubtedly has clear value to society, particularly in the context of a disease or condition such as trauma for which the stakes are high (i.e., high mortality and morbidity) and there is a paucity of known effective therapies. The protection of individuals who are research subjects also has a clear value, both to the individuals themselves and to society. Trust in the research enterprise is of paramount importance; without it, there would be too few volunteers to sustain clinical research, which would directly and calamitously reduce and retard medical advances. Finding the right balance between providing regulatory protections for research subjects and enabling scientific progress is chal-
__________________
10 Although the military is able to conduct some research in theater, it should be noted that policy, logistical, and ethical issues constrain the conduct of randomized controlled clinical trials on the battlefield.
lenging and, understandably and appropriately, the source of ongoing discussion, scrutiny, and concern.
Through a review of the literature and discussions with researchers and regulatory agency representatives, the committee identified several barriers to research associated with federal regulations, discussed in the sections below. In some cases, the problem lies in ambiguity associated with or misunderstanding or misinterpretation of the regulations. Evolutionary and revolutionary changes in science, technology, and clinical research designs are often accompanied by the development of federal policy statements (i.e., “guidance”), which can reduce problems of ambiguity and misinterpretation, but these frequently can take many years to be issued. In other cases, the problem resides in regulatory silence; that is, the regulations do not address or did not anticipate current concerns. Finally, in some cases, the problem is due to regulations themselves, which can have a static quality and which, in some cases, may have their origin in discussions dating back to the 1970s (e.g., IRB and informed consent regulations). For example, when strong—even overwhelming—evidence has accumulated about the efficacy and safety of a trauma intervention, the inflexibility of the U.S. Food and Drug Administration’s (FDA’s) evidentiary rules has prevented or seriously delayed that real improvement from reaching trauma patients, particularly in the absence of more formal clinical trials. (An example is ketamine’s use as an analgesic, described in Box 4-7.) FDA needs to take a more flexible approach to the nature of the evidence it uses for its determinations, in accordance with the developing epistemology of evidence itself in the scientific community. As discussed in the sections below, continued efforts to streamline regulatory processes would have a major impact on the ability to use limited trauma research funds efficiently and effectively.
Unclear distinction between quality improvement and research At the core of a continuously learning health system as envisioned by the Institute of Medicine is the imperative to learn from the everyday practice of medicine (IOM, 2013). Such learning often takes the form of continuous quality improvement cycles. The generation of knowledge through continuous quality improvement and how it is distinguished from research, however, is a subject of ongoing debate, particularly in light of the multiple federal regulations that govern the conduct of human subjects research (as described in Box 4-6).
The U.S. Department of Health and Human Services (HHS) defines research as “a systematic investigation, including research development, testing and evaluation, designed to develop or contribute to generalizable knowledge.”11 Quality improvement is defined as “systematic data guided
__________________
11 45 CFR § 46.102(d).
activities designed to bring about immediate improvements in health care delivery” (Baily et al., 2006, p. S5). Given these definitions, there are any number of learning activities in which distinctions are blurred, including
- clinical research (controlled trials) versus medical practice,
- quality improvement research versus quality improvement,
- pragmatic clinical trials (comparative effectiveness research in real-life settings) versus medical practice,
- comparative effectiveness research versus medical practice, and
- research on medical practice versus medical practice.
Yet whether a given learning activity is classified as quality improvement or research has significant implications for the need to adhere to the human subjects protections outlined in Box 4-6. The “gray zone” in distinguishing between quality improvement and research slows and even impedes both quality improvement and research activities, limiting the effectiveness and impact of the learning system as a whole.
The ambiguity between quality improvement and research temporarily halted a successful initiative by Pronovost and colleagues (2006) to save lives and eliminate unnecessary costs by decreasing central line-associated bloodstream infections in intensive care units. In this case, HHS’s Office for Human Research Protections disagreed with a Johns Hopkins IRB’s conclusion that this was a quality improvement project and thus did not require
full IRB review or informed consent (Baily, 2008; Faden et al., 2013; Miller and Emanuel, 2008).
Ironically, in many of these scenarios, the activities in question pose low risk to the individuals being studied (Miller and Emanuel, 2008). Clearer guidance is needed from HHS as the blurred distinction between quality improvement and research may impede the dissemination of worthwhile quality improvement activities or relegate those activities to added protections that may not be required. Regardless of this distinction, it is important that the regulatory environment not dampen these learning activities.
Multisite IRB review The federal regulations discussed in Box 4-6 were established in the 1980s and 1990s under a simpler research paradigm in which there was generally one institution conducting research to be reviewed by a single IRB. The current research enterprise is much more complex. Clinical trials and many other kinds of research studies often include many investigators working across multiple institutions. The Common Rule requires prior review and approval of nonexempt human subjects research by an IRB.12 Approval by multiple IRBs is not required for multisite studies and may not be beneficial—for example, in research on public health disasters and rare diseases and in most emergency care research (Goldkind et al., 2014). Yet research has nonetheless been delayed by wary investigators, IRBs, and institutions feeling compelled to obtain IRB approval at each participating site (METRC, 2016). Multiple IRBs can produce conflicting results and require changes to the protocol or informed consent documents that are at cross-purposes. In some cases, the separate reviews occur sequentially, contributing to significant delays in the onset of research, at least locally. Centralized IRBs are one potential means of streamlining this regulatory process (Check et al., 2013; Flynn et al., 2013). Moreover, they can be a means of ensuring the application of scientific or medical expertise that may not exist widely, thus contributing to high-quality reviews and sophisticated science and clinical research designs (Goldkind et al., 2014). NIH has put forth for comment draft policy mandating the use of a single IRB for multisite studies.13 In addition, HHS has published a Notice of Proposed Rulemaking14 that would require, in most cases, the use of a single IRB. This proposal has been reiterated in a number of policy recommendations, including the National Academies of Sciences, Engineering, and Medicine (NASEM, 2015) report Optimizing the Nation’s Investment in
__________________
12 45 CFR Part 46, Subpart A.
13 The NIH draft policy on use of a single IRB for multisite studies is available at http://grants.nih.gov/grants/guide/notice-files/NOT-OD-15-026.html (accessed October 16, 2015).
14 The HHS Notice of Proposed Rulemaking is available at https://www.federalregister.gov/articles/2015/09/08/2015-21756/federal-policy-for-the-protection-of-human-subjects (accessed April 11, 2016).
Academic Research, as well as the 21st Century Cures Act. However, these proposals are still under consideration and not yet reflected in the federal regulations.
Privacy protections for data sharing As discussed earlier in this chapter, HIPAA regulations present barriers to using and sharing trauma patient data across systems for research purposes and care within and between the military and civilian sectors (Baily et al., 2006; Seymour et al., 2014). Box 4-8 provides a catalog of facts that address common misperceptions about HIPAA that can impede research in both sectors.
The use of protected health information for both direct patient care and for research purposes is governed tightly by HIPAA regulations. Provider misperceptions regarding what HIPAA permits for the purposes of direct patient care are a barrier to disclosing patients’ protected health information to those also providing treatment. In fact, HIPAA recognizes a relationship known as an organized health care arrangement, which allows covered entities to disclose protected health information for treatment activities. This arrangement also allows the disclosure of protected health information by covered entities with a legitimate need to reference patient data for the operation of the care delivery system, including quality assurance at the individual case level and quality improvement at the level of a defined process of care. The vast majority of information produced by a trauma registry takes the form of summary reports at the process level, where it is physically impossible to identify individual patients. However, individual clinicians need to see what happens to the patients they treat as those patients move through the complete care process. In this circumstance, a clinician has a legitimate patient–clinician relationship; patients’ explicit consent typically exists for such uses of their data; and this use of patients’ protected health information clearly falls under an organized health care arrangement recognized by the regulations (AMA, 2016; HHS, 2003).
By definition, research utilizing protected health information uses existing data, so the associated risks to patients center on autonomy (patients’ control of their own data) and confidentiality. Deidentified data are not protected health information; as such, they are not protected under HIPAA. There are multiple ways in which covered entities can use and disclose protected health information for research purposes and remain HIPAA compliant. Covered entities may use and disclose protected health information with participants’ written authorization. In addition, protected health information may be used and disclosed in the absence of authorization in a number of circumstances. HIPAA allows for a waiver of research participants’ authorization in those circumstances in which (1) all risks to the patients are minimized; (2) the proposed investigation has the potential to contribute to better care; and (3) it is not possible or feasible to obtain
direct, individual patient consent. Covered entities may also use and disclose protected health information as a limited data set with a data use agreement without an authorization. A data use agreement—entered with the intended recipient of the limited data set—specifies the ways in which the data set may be used and how it will be protected.15
Informed consent challenges for trauma research Trauma research involving human subjects is highly heterogeneous, ranging from minimal-risk epidemiological studies to clinical trials of experimental interventions for life-threatening conditions that must be administered shortly after injury (Goldkind et al., 2014). In the latter case, patients may be unconscious or otherwise incapable of providing informed consent to participate in a study,
__________________
15 45 CFR Part 164.
presenting a significant challenge to the conduct of such research in an ethically appropriate manner. There is general agreement about the degree and types of protections needed for clinical research entailing greater than minimal risk (e.g., an intervention study using an untested therapeutic). Such protections generally include IRB approval and advance informed consent of research participants, although FDA and other HHS regulations permit an exception from informed consent for emergency research under narrow circumstances, in which additional protections are required (e.g., establishment of a data monitoring committee, public disclosure prior to initiation and after completion of the research, community consultation). In the civilian sector, meeting such requirements is challenging. For combat casualty care research that is sponsored, conducted, or supported by DoD, even greater challenges arise in attempting to enroll military personnel in studies. For example, there is ongoing debate regarding the validity of the
community consultation process in a military setting, including concerns about undue influence from higher-ranked members of the military (Perkins et al., 2012).
More controversial, however, is how to protect participants appropriately in minimal-risk research without creating unnecessary barriers to its conduct. Under the Common Rule, minimal-risk trauma research, such as retrospective studies using registry data, can qualify for a waiver of informed consent.16 No provision exists, however, for waiving or altering informed consent processes for FDA-regulated minimal-risk research. Under these conditions, investigators cannot conduct an FDA-regulated clinical investigation, such as a study of a minimal-risk device, if the research offers no direct benefit to participants and does not otherwise qualify for the exception from informed consent under 21 CFR § 50.24 (see Box 4-9).
Human subjects research conducted using any funds appropriated to DoD is further governed and restricted by Title 10 of the United States Code, Section 980 (10 U.S.C. § 980, Limitation on Use of Humans as Experimental Subjects.), which prohibits research studies using human subjects without advance informed consent of the subjects or their legal representatives. The Secretary of Defense or his/her designee17 has the authority under 10 U.S.C. § 980 to waive the requirement for informed consent for a specific research study if it is deemed necessary to the armed forces and may directly benefit the subjects (notably including subjects in control groups). This statute was passed in 1972, prior to DoD’s adoption of the Common Rule,18 which requires both IRB approval and informed consent (with exceptions for minimal-risk research19). Now obsolete, this statute serves as an added barrier to military research, delaying needed trauma research in both military and civilian settings20 by 5 months or more and consuming precious study funds, to the detriment of trauma care delivered to servicemen and -women, injured host nationals, and civilian casualties (Brosch, 2015).
Thus FDA and DoD requirements for informed consent meant to protect individuals enrolled in research can have the untoward effect of impeding research necessary to provide evidentiary standards for ad-
__________________
16 45 CFR § 46.116(d).
17 The Secretary of Defense has delegated this waiver authority to the heads of the DoD services.
18 32 CFR Part 219.
19 10 U.S.C. § 980 does not apply to research that is exempt from the Common Rule (DoD Directive 3216.02, Protection of Human Subjects and Adherence to Ethical Standards in DoD-Supported Research).
20 Research studies conducted at civilian sites but supported by DoD (with funds or supplies) are also subject to 10 U.S.C. § 980.
vances in therapeutic, diagnostic, and preventive clinical interventions. This dilemma is particularly problematic as these requirements ironically make the most innocuous research (from a subject’s perspective) the most difficult to approve. Although a number of changes to these federal regulations are currently under consideration, including revisions to the Common Rule and changes to FDA regulatory processes under the 21st Century Cures Act, it remains to be seen whether these proposed changes will go into effect and ease the regulatory barriers delaying and preventing needed trauma research.
PROCESSES AND TOOLS FOR TIMELY DISSEMINATION OF TRAUMA KNOWLEDGE
Knowledge derived from performance improvement and research efforts will improve medical care and patient outcomes only if it is disseminated and applied in practice. Given the complexity and volume of clinical information available21 and the extraordinary rate at which new knowledge is generated, a broad range of systematic processes and tools are needed to disseminate knowledge and assist front-line care providers in utilizing the best evidence and practices available. In the military, the capture and sharing of knowledge are necessary to sustain and advance trauma care not just in interwar periods but also in wartime, as deployed trauma teams rotate at approximately 6-month to 1-year intervals. The institutionalization of this experiential knowledge and processes for its dissemination ensure that the next team of providers deployed to theater (and their patients) can benefit from the lessons learned by their predecessors. Throughout the wars in Afghanistan and Iraq, the military utilized a number of innovative mechanisms to increase the rate at which new knowledge was disseminated to providers in the challenging battlefield environment (see Table 4-7).
Traditional channels for knowledge dissemination, such as publications in peer-reviewed journals and presentations at scientific conferences, are critically important in today’s knowledge landscape. These channels foster increased collaboration between the military and civilian sectors and encourage the bidirectional exchange of information (Pruitt and Rasmussen, 2014). However, additional steps are necessary to promote and accelerate the translation of new evidence into changes in care.
In a learning trauma care system, improvements in trauma care do not depend on the ability of individual providers to discover, assimilate, retain, and put into practice the ever-increasing supply of clinical evidence. Instead, trauma care providers have access to such resources as evidence-based clinical practice guidelines and clinical decision support tools that capture, organize, and disseminate the best available information to guide decision making and reduce variation in care and outcomes (IOM, 2013). In addition, synchronous methods of learning22 offer front-line providers timely access to high-quality tacit knowledge,23 which plays an important role in improving
__________________
21 As an indicator of the increasing volume of clinical information, the number of medical journal articles published in 2010 (more than 750,000) was more than three times the number published in 1970 (IOM, 2013).
22 Synchronous learning occurs in real time through mechanisms that give the teacher and the learner simultaneous access to one another (e.g., one-to-one coaching sessions via phone, classroom teaching), whereas asynchronous methods of learning allow learners to access information whenever they would like, not requiring the presence of an instructor (e.g., websites, guidelines).
23 Tacit knowledge is knowledge that is acquired through practice and application.
TABLE 4-7 Summary of Mechanisms for Knowledge Dissemination Used in the Military Trauma System
| Individual Patient Care | Process Design and Execution | Tacit Knowledge | |
|---|---|---|---|
| Senior Visiting Surgeon Program | |||
| Joint Trauma System weekly teleconference | |||
| Teleconsultation program (e.g., pediatric and severe burn patients) | |||
| Real-time telephone support with an expert (e.g., for burn treatment) | |||
| Full clinical decision support/embedded protocols (e.g., Burn Resuscitation Decision Support System) |
practice in meaningful ways (Dixon, 1994; Nonaka, 1994). Timely access to information allows providers to ask detailed, context-specific questions, rapidly expediting care processes. The exchange of knowledge in real time enables providers to address challenges and questions at the point of care, offering immediate potential improvements to care delivery and patient outcomes. The following sections review four key mechanisms for timely dissemination of trauma knowledge: clinical guidelines, clinical decision support tools, telemedicine, and the Senior Visiting Surgeon Program.
Clinical Guidelines
Clinical guidelines provide a mechanism for changing practice and reducing unwarranted practice variations. Prehospital and hospital clinical practice guidelines can facilitate patient evaluations and help in determining an evidence-based course of action. The strongest guidelines are informed by evidence from hypothesis-driven research and knowledge derived from experiential learning and performance improvement efforts. At the same time, however (as discussed in Chapter 3), guidelines are not followed blindly in a learning system. Rather, optimal learning systems expect and even promote deviation from guidelines. Tracking such deviation and measuring outcomes enables validation and further improvement of the guidelines.
Guidelines in the Military
Codifying lessons learned and best practices into guidelines and developing tools and innovative strategies to assist front-line care providers are particularly important in the military, where brief deployments and the constant rotation of personnel, as well as interwar periods, challenge the ability to sustain institutional knowledge or operational memory. DoD develops and issues multiple sets of guidelines, including TCCC guidelines for the prehospital setting and JTS clinical practice guidelines for the hospital setting. The Defense and Veterans Brain Injury Center also issues clinical recommendations for mild traumatic brain injury. However, DoD does not mandate adherence to any set of guidelines. Military guidelines are more frequently updated and more responsive to advances in knowledge (including those emerging from experiential learning) relative to guidelines in the civilian sector (Gross, 2015).
Tactical combat casualty care (TCCC) guidelines Before the TCCC guidelines were developed in 1996, nominal improvements had been made in the prehospital phase of combat casualty care (Butler et al., 1996). TCCC is a “set of evidence-based, best-practice, prehospital trauma care guidelines customized for use on the battlefield,” developed based on the need to unite good medicine and good tactics to avoid preventable death (Blackbourne et al., 2012; Butler and Blackbourne, 2012; Butler et al., 2015, p. 7). Established in 2001, the Committee on TCCC ensures that new technology, information, and evidence are incorporated into these guidelines on an ongoing basis.
TCCC guidelines are reviewed quarterly and updated as needed. Once approved by a two-thirds majority of voting members of the Committee on TCCC, individual changes are distributed to the committee members and published in the Journal of Special Operations Medicine (DHB, 2015).24 Once per year, all the changes are compiled into an annually updated TCCC curriculum. This curriculum is then posted online, including the web pages of the JTS, the Military Health System, the National Association of Emergency Medical Technicians, the Journal of Special Operations Medicine, and the Special Operations Medical Association. The updated curriculum also is e-mailed to DoD schoolhouses and to the TCCC distribution list (more than 600 individuals across all the services). Included in each change packet are the updated guidelines, the position paper providing the evidence to support the change, and a set of PowerPoint training slides (DHB, 2015).
With the maturation of TCCC, the conflicts in Afghanistan and Iraq
__________________
24 Personal communication, F. K. Butler, Committee on Tactical Combat Casualty Care, U.S. Department of Defense, July 17, 2015.
have seen dramatic changes in prehospital care (Butler and Blackbourne, 2012). The implementation of tourniquet recommendations from the TCCC guidelines, for example, sharply decreased mortality from extremity hemorrhage (Eastridge et al., 2012; Kelly et al., 2008; Kotwal et al., 2011). Despite formal endorsement of TCCC guidelines by medical leaders (e.g., the Defense Health Board, the service surgeons general) and some line leaders (e.g., then COL Stanley McChrystal), however, the implementation and use of updated TCCC guidelines and techniques are incomplete. While combat medics may be very familiar with TCCC, this same level of familiarity is not consistent across other medical personnel and nonmedical leadership (i.e., line leaders), resulting in part from the absence of a DoD-wide requirement for all personnel to receive TCCC training (Kotwal et al., 2013a).
JTS clinical practice guidelines (CPGs) The JTS curates its own set of hospital guidelines, called CPGs. These guidelines are developed by subject matter experts based on battlefield-generated needs and the identification of gaps whose resolution has the potential to drive change and improve performance. As with the prehospital TCCC guidelines, CPGs are evidence-based to the extent possible. Evidence is derived from the scientific literature and analysis of data captured in the DoDTR, supplemented with the consensus of subject matter experts, when necessary. In some instances (e.g., management of an unexploded device in a casualty), high-quality evidence will never be available to inform CPGs, which will be developed based entirely on lessons learned and expert consensus (JTS, 2012b). CPGs are updated by the JTS on an annual basis, with additional updates occurring as needed in response to new evidence or input from subject matter experts. New and updated CPGs are approved by the JTS director (Stockinger, 2015).
Once created or updated, CPGs are disseminated to providers in a combat theater through several mechanisms. They are made widely available online and posted on the U.S. Army Institute of Surgical Research and JTS website25 (JTS, 2012a). In addition, the JTS works with predeployment training centers to incorporate these guidelines into the training curricula.
The DoDTR has developed the capacity to track compliance with many CPGs. Compliance with CPGs has repeatedly demonstrated positive outcomes, for example, in reducing abdominal compartment syndrome mortality (from 36 percent to 18 percent, with 94 percent compliance), hypothermia (from 7 percent to 1 percent, with 84 percent compliance), and mortality in those receiving massive transfusions (from 32 percent to 20 percent, with 85 percent compliance) (Eastridge et al., 2009).
While originally developed for use in CENTCOM alone, the JTS is
__________________
25 Available at http://usaisr.amedd.army.mil/beta/cpgs.html (accessed February 16, 2016).
shifting away from CENTCOM CPGs and is in the process of updating the CPGs so they are no longer specific to any geographic command. These CPGs will then be distributed to each combatant commander for review and approval or modification (Stockinger, 2015). As of this writing, nine CPGs had been developed and approved for use in the Pacific Command (USAISR, 2015).
Guidelines in the Civilian Sector
In the civilian sector, evidence-based guidelines have proliferated in recent years, with the Agency for Healthcare Research and Quality’s National Guidelines Clearinghouse listing more than 2,500 published guidelines. Unfortunately, adherence to clinical practice guidelines varies dramatically (McGlynn et al., 2003). In a recent study conducted at five Level I trauma centers across the United States, compliance with 22 recommended clinical practices ranged from 12 to 94 percent (Shafi et al., 2014). In addition, organizations that develop guidelines often use different methods for evaluating evidence, and the resulting recommendations sometimes conflict with each other (Kerwin et al., 2012). Numerous attempts have been made to address these inconsistencies, including the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) system; the National Prehospital Evidence-Based Guideline Model Process, and the Institute of Medicine (IOM, 2011) report Clinical Practice Guidelines We Can Trust.
In 2007, the IOM recommended that a multidisciplinary panel establish a model for creating evidence-based guidelines for prehospital emergency care, having found that only 4 percent of EMS interventions were based on high-quality evidence, while half of interventions were based on very weak or no evidence (IOM, 2007a; Lang et al., 2012). The resulting National Prehospital Evidence-Based Guideline (EBG) Model Process was a joint project of the National EMS Advisory Council and the Federal Interagency Committee on EMS, with funding from the National Highway Traffic Safety Administration and the Health Resources and Services Administration’s Emergency Medical Services for Children Program. The model that was developed emphasizes evidence-based recommendations rather than expert opinion, and outlines an eight-step process focused on the creation of broad EMS guidelines that can be customized for regional and local needs. This initiative has led to stepwise progress in developing EBGs for EMS, national EMS guidelines, and recommendations for a national consortium to warehouse and coordinate the incorporation of EMS-related EBGs into the EMS community (Martin-Gill et al., 2016). Recent progress includes the development of evidence-based guidelines using the National Prehospital Evidence-Based Guideline Model Process, such as the American College of Surgeons’ hemorrhage control guideline, as well as research-informed
clinical guidelines developed through expert consensus by the National Association of State EMS Officials’ (NASEMSO’s) National Model EMS Clinical Guidelines project.26
In the civilian sector, one of the leaders in the development of hospital-based trauma care guidelines is the Eastern Association for the Surgery of Trauma (EAST). EAST, through membership volunteer efforts, has published more than 60 practice management guidelines, all of which are available on the EAST website27 and in the Journal of Trauma and Acute Care Surgery. The American College of Surgeons also has four trauma guidelines for hospital care (ACS, 2016), and other groups have begun creating trauma care guidelines as well (Bulger et al., 2014).
Military–Civilian Interface and Guideline Development
The development, maintenance, and dissemination of guidelines have offered unique opportunities to facilitate the interface between the military and civilian trauma systems and the transfer of innovations and lessons learned. The military’s Committee on TCCC has an official partnership with the National Association of EMTs. In addition, TCCC guideline changes are posted on several civilian websites, and the Prehospital Trauma Life Support textbook includes multiple chapters on TCCC guidelines (DHB, 2015). The work of the Committee on TCCC also inspired the development of a counterpart civilian committee, the Committee on Tactical Emergency Casualty Care (C-TECC). C-TECC works to expedite the translation of military medical lessons learned into civilian crisis response by adapting TCCC guidelines for civilian use, taking into account differences in operational environments, resources, patient population, and scope of practice (Callaway et al., 2011). In addition, the JTS’s CPGs are developed with input from civilian subject matter experts, and many of the early JTS CPGs were based on existing EAST guidelines. EAST now has its own Military Section, and offers a list of materials and literature relevant to deploying military surgeons (Stockinger, 2015).
Despite these opportunities, however, the dissemination and bidirectional translation of best practices and knowledge between the two sectors is slow and often incomplete. The civilian sector’s adoption of tourniquet use is a case in point. Despite clear evidence of its benefit in preventing death from hemorrhage, uptake of this practice has been variable across the nation. Table 4-8 shows an overall increase in the reported use of tourniquets be-
__________________
26 See https://nasemso.org/Projects/ModelEMSClinicalGuidelines/index.asp (accessed May 23, 2016).
27 See https://www.east.org/education/practice-management-guidelines (accessed May 23, 2016).
TABLE 4-8 Civilian Tourniquet Use in 12 U.S. States, 2010-2014
| STATE | YEAR | |||||
|---|---|---|---|---|---|---|
| 2010 | 2011 | 2012 | 2013 | 2014 | TOTAL | |
| A | 7 | 19 | 13 | 23 | 36 | 98 |
| B | 0 | 0 | 0 | 0 | 20 | 20 |
| C | 0 | 1 | 0 | 0 | 0 | 1 |
| D | 7 | 14 | 27 | 37 | 30 | 115 |
| E | 4 | 10 | 13 | 53 | 63 | 143 |
| F | 2 | 0 | 0 | 0 | 4 | 6 |
| G | 4 | 3 | 278 | 138 | 18 | 441 |
| H | 0 | 2 | 3 | 2 | 13 | 20 |
| I | 10 | 10 | 55 | 98 | 146 | 319 |
| J | 6 | 18 | 36 | 59 | 76 | 195 |
| K | 0 | 7 | 8 | 16 | 29 | 60 |
| L | 30 | 39 | 25 | 19 | 30 | 143 |
| TOTAL | 70 | 123 | 458 | 445 | 465 | 1,561 |
NOTES: These data represent instances of tourniquet usage by 12 states that submitted reliable data to the National EMS Information System (NEMSIS) project’s National EMS Database over the time period 2010-2014. The table excludes states that may now have reliable data, but were not reporting reliable data during this period. Data from the National EMS Database prior to 2010 were not considered reliable enough to include in this analysis.
SOURCE: Data extracted from the National EMS Database.
tween 2010 and 2012, with a subsequent plateau in total reported use (as seen, for example, in State D), but some states (e.g., states C and F) have not yet adopted this practice. In contrast, military tourniquet use increased significantly between 2002 and 2006 and then began to plateau (Kragh et al., 2015), indicating a 6-year lag between the time tourniquet use stabilized in the military and civilian sectors. In some places in the United States, however, adoption occurred earlier. For example, Pennsylvania changed statewide EMS protocols at the end of 2008—5 years before the American College of Surgeons’ hemorrhage control guideline was published (Bulger et al., 2014)—to add a statewide basic life support bleeding control protocol that emphasizes the use of tourniquets “as initial method of bleeding control in severe extremity bleeding” (Pennsylvania Department of Health, Bureau of Emergency Medical Services, 2008). In 2011, Pennsylvania further began to require that every ambulance carry commercial tourniquets instead of relying on improvised versions (PA Bulletin, 2011).
Differences among states may in part explain this variation in uptake of
tourniquet use. In “home rule” states where there is local discretion in the use of protocols, incorporation of a new paradigm for tourniquet use by following national guidelines and changing protocols and training would fall to local EMS agencies and medical directors, likely resulting in variable uptake. In states that use statewide protocols, by contrast, the protocols and training can be altered across the entire state with a single protocol change.
Clinical Decision Support Tools
Clinical decision support tools make knowledge of best trauma care practices, including clinical practice guidelines, available in real time at the point of care. These tools provide patient-specific care recommendations, helping medical providers deliver the best care possible and generate optimal outcomes for injured patients (IOM, 2001, 2013). Some of the most successful decision support tools have been simple, paper-based checklists, such as those used to virtually eliminate central-line-associated bloodstream infections (Pronovost et al., 2006). In other cases, the tools can be stand-alone devices. The military’s Burn Resuscitation Decision Support System (described in Box 4-10), for example, is an FDA-approved device that provides fluid intake recommendations for burn patients. The tool is designed specifically to assist providers who do not routinely provide burn care so as to avoid over- or underresuscitation of patients (Galvan, 2013). Another example of a computerized clinical decision support tool is used in prevention of venous thromboembolism (VTE) (see Box 4-11). As discussed in Chapter 3, however, the integration of these tools into workflow by embedding them in electronic health records represents the pinnacle of real-time access to knowledge based on a learning health system approach.
In the VTE prevention example cited above, the decision support tool assumes that there is little variation among individual patients, and optimal VTE prophylaxis suggestions are based only on clinical and demographic data. However, new data suggest that there is in fact wide variation among individual patients as regards the baseline thrombotic state and response to VTE prevention therapy. Augmenting this decision support tool with laboratory data could bring precision medicine to the bedside, enabling personalized care of injured patients (Haut et al., 2016). To realize the promise of such advances, however, availability of the vast amounts of data of many types that would be required to ensure that patients received optimal care would have to be made a priority.
Overall, while there are isolated examples of successful application of clinical decision support tools to improve real-time access to best practices in trauma care, this remains an area for growth in both military and civilian settings as electronic health records evolve. DoD recently awarded Cerner (in partnership with Leidos and Accenture) a contract to procure Cerner’s
electronic health record system (Garamone, 2015; Monegain, 2015). Intermountain Healthcare, which is in the process of rolling out its own build of the Cerner electronic health record (called iCentra) to all of its hospitals and clinics, is also a strategic partner in this deal and will advise Cerner throughout the project on topics including clinical systems architecture, governance, and workflow (Monegain, 2015). The acquisition of an electronic health record optimized for protocol-based clinical decision support with integrated data capture to feed condition-specific clinical registries may hasten the evolution of the DoD trauma system toward this key feature of a learning system for trauma care.
Telemedicine
Effective communication along the continuum of care is essential to directing the best care, dispatching appropriate transport, and preparing receiving treatment facilities by providing information on the number of incoming casualties and the severity and types of injuries (DHB, 2015).
Such communication among providers also facilitates real-time performance improvement (Pruitt and Rasmussen, 2014). On the battlefield and in operational theaters, the military often works with limited resources in austere and remote locations and lacks specialized medical expertise (McManus et al., 2008). In these challenging conditions, the use of telemedicine improves communication and offers providers access to clinical expertise and advice in near real time.
At the start of the wars in Afghanistan and Iraq, communication between medical providers and receiving treatment facilities and between facilities along the continuum of care was nearly nonexistent. In 2006, the JTS established a weekly teleconference to discuss active patients as they were moved through the system. These system-wide forums link trauma teams working in theater, including medics at the point of injury and evacuation, with providers at Landstuhl Regional Medical Center in Germany and treatment facilities in the continental United States (Bailey et al., 2013). The teleconferences, which still take place and today serve as educational forums, afford providers at all levels the opportunity to discuss patient issues, receive feedback, and better understand care provided at previous facilities (Bailey et al., 2013; Eastridge et al., 2009). From the forum’s initiation to October 15, 2015, 485 teleconferences featuring 3,600 case studies took place (Haut et al., 2016). These teleconferences serve as a conduit for disseminating clinical practice guidelines and system advances in trauma care, optimizing best practices in data collection, and improving the validity of the DoDTR (Pruitt and Rasmussen, 2014). Importantly, the forum facilitates performance improvement through both vertical connections (from the individual facility level to the joint theater trauma system level to the DoD trauma system level) and horizontal connections (e.g., peer learning from colleagues at other facilities as well as international partners).
While deployed, military physicians frequently must perform care outside their area of expertise (Fuenfer et al., 2009). Given this reality, the military made use of an extensive e-mail-based teleconsultation program during the wars in Afghanistan and Iraq (Eastridge et al., 2009; McManus et al., 2008). Developed in 2004, this program is run by the Army Office of the Surgeon General with participation from all services. Deployed providers e-mail consultation requests (which may include questions on diagnoses and treatment options), which are directed to the appropriate specialty group. Recommendations are returned to the requester within 24 hours (McManus et al., 2008). Between 2004 and 2015, the program facilitated more than 12,000 consultations globally and helped to both facilitate and prevent evacuations. Despite the demonstrated value of a teleconsultation program, challenges include lack of provider awareness about the pro-
gram, lack of central funding, and threats to continuity due to inadequate institutionalization.28
As the theater of operations matured over the course of the conflicts in Afghanistan and Iraq, the military increasingly made use of telemedicine to great effect. However, it must be noted that early in a conflict, limited bandwidth may constrain the extent to which it is feasible to utilize more advanced forms of telemedicine. In such an environment, rudimentary forms of telemedicine (e.g., telephone consultation) may be the most reliable.
In the context of civilian trauma care, telemedicine has great potential to transform care, costs, and patient visit cycle times.29 For example, telemedicine offers a possible solution to improve communication and the sharing of patient data along the trauma continuum of care, particularly between the prehospital and hospital settings. Technology that enables two-way video and voice communication and data transmission between ambulances and hospitals facilitates collaboration between emergency medical technicians and receiving facility physicians on prehospital care of the patient and also helps the receiving trauma team to better prepare for the patient’s arrival (Latifi et al., 2007). Telemedicine programs also help effectively leverage the time of specialists, particularly in regions (e.g., rural areas) where access to trauma centers and trauma specialists (e.g., trauma surgeons, burn specialists) is limited. In one interhospital teletrauma program (hub and spoke model), centrally located trauma surgeons with video, audio, and data (e.g., vital signs) access to remote sites are able to assist in early survey and care decisions—for example, to help get the patient sufficiently stabilized for transfer to the trauma center (Latifi et al., 2007). In some cases, remote consultations help avoid costly and unnecessary patient transfers (Amadi-Obi et al., 2014; Latifi et al., 2009). The role of telemedicine in trauma care will undoubtedly continue to evolve as advances in technology yield reliable capabilities to support remote care and collaboration such as three-dimensional telepresence (Welch et al., 2009) and teleoperation (Garcia et al., 2009). Beyond real-time assistance with patient care, however, telemedicine technology also offers health care providers an invaluable opportunity to expand their knowledge and expertise through education, training, and peer mentoring (see the description of Project Extension for Community Healthcare Outcomes [ECHO] in Box 3-1). For example, in recent years, the Military Health System has adopted the Project ECHO model in its approach to pain management, working to improve its
__________________
28 Personal communication, C. Lappan, U.S. Department of Defense, to E. Cornett, the National Academies of Sciences, Engineering, and Medicine, December 21, 2015.
29 Cycle time refers to the amount of time that a patient spends at a primary care office visit (IHI, 2016).
providers’ skill sets and capacity to effectively manage and treat complex, chronic pain conditions (discussed further in Chapter 6). Telemedicine, mobile health, and telehealth supports and technologies are rapidly maturing enabling more immediate and more local outreach to patients, and decreasing demand for face-to-face visits. But despite their promise, policy and payment are lagging behind these innovations, although payers are becoming more receptive to these modes of care and more innovative in finding ways to support them.
Senior Visiting Surgeon Program
The Senior Visiting Surgeon (SVS) Program exemplifies how military–civilian collaboration results in bidirectional translation of knowledge and experience, advancing trauma care in both sectors. In the SVS Program, military and senior civilian surgeons worked side by side at a single location, collaboratively providing care to those wounded in combat. The program was developed in 2005 in response to a growing need for experienced, hands-on expertise in providing trauma care during the wars in Afghanistan and Iraq and to such problems as insufficient research infrastructure within the military and a lack of academic mentoring in military trauma research (Knudson and Rasmussen, 2012; Knudson et al., 2014). The program was intended to advance trauma care by bringing experience and expertise in civilian trauma care to the military sector, developing the practices of the military trauma system, providing mentorship for military physicians, and encouraging scientific exchange between military providers and civilian trauma care leaders (Blackbourne et al., 2012).
In the SVS Program, civilian trauma care surgeons spent 2-4 weeks at the military’s Landstuhl Regional Medical Center in Germany. In addition to providing care alongside their military colleagues, visiting surgeons gave grand rounds lectures on various topics, advancing the education of deployed physicians, nurses, and physician assistants (Knudson and Rasmussen, 2012; Moore et al., 2007). Since its induction, the program has contributed to the initiation of research in theater, as well as independent documentation and validation of advances made in combat casualty care achieved under the JTS (Knudson et al., 2014; Moore et al., 2007). While evidently a successful program of value to participating individuals (Knudson et al., 2014), a notable limitation of SVS is that civilian participation was restricted to surgeons alone. Expansion of the program to include other disciplines (e.g., emergency medicine physicians, nurses) could improve cross-fertilization across a broader group of stakeholders.
SUMMARY OF FINDINGS AND CONCLUSIONS
CONCLUSION: In the military and civilian sectors, the failure to collect, integrate, and share trauma data from the entire continuum of care limits the ability to analyze long-term patient outcomes and use that information to improve performance at the front lines of care. The collection and integration of data on the full spectrum of patient care and long-term outcomes using patient-centric, integrated registry systems need to be a priority in both sectors if the full potential of a learning trauma care system is to be realized, and if deaths from survivable injuries are to be reduced and functional outcomes maximized.
Related findings:
- The collection and integration of trauma data across the care continuum is incomplete in both the military and civilian sectors.
- Military and civilian trauma management information systems rely on inefficient and error-prone manual data abstraction to populate registries.
- Existing trauma registries and other data systems are not linked, and sharing data across the military and civilian sectors is impeded by political, operational, technical, regulatory, and security-related barriers.
CONCLUSION: The military uses a flexible and agile approach to guideline development distinct from that used in the civilian sector. This pragmatic, focused empiricism approach enables rapid guideline development using best available data. However, the safety and effectiveness of care practices based on low-quality evidence need to be validated in due time through higher-quality studies carried out in conjunction with the civilian sector. Additionally, more formal processes are needed to encourage joint military–civilian discussion of guidelines so as to enhance bidirectional translation of knowledge and innovation between the two sectors.
Related findings:
- Processes for guideline development in the civilian sector are much more rigid (and rigorous), thorough, and time-consuming compared with military guidelines; therefore, civilian guidelines are less responsive to change and updated less frequently.
- The dissemination and bidirectional translation of knowledge and best practices (e.g., tourniquet use) between the military and civilian sectors is slow and often incomplete.
CONCLUSION: Investment in trauma research is not commensurate with the burden of traumatic injury. To address critical gaps in knowledge of optimal trauma care practices and delivery systems, the United States needs a coordinated trauma research program with defined objectives, a focus on high-priority needs, and adequate resourcing from both the military and civilian sectors.
Related findings:
- Despite the significant burden that trauma places on society, the sustainment of DoD’s trauma research program is threatened, and civilian investment in trauma research is limited.
- Gaps identified in DoD’s Guidance on Development of the Force remain less than 50 percent resolved.
- In the civilian sector, no mechanism exists for directing research investments toward identified gaps, a problem exacerbated by the absence of a centralized institute dedicated to trauma and emergency care research.
CONCLUSION: A learning trauma care system cannot function optimally in the current federal regulatory landscape. Federal regulations or their interpretations often present unnecessary barriers to quality improvement and research activities. Lack of understanding of relevant provisions of the Health Insurance Portability and Accountability Act has resulted in missed opportunities to share data across the continuum of care in both the military and civilian sectors.
Related findings:
- The ambiguity between quality improvement and research slows and even impedes quality improvement and research activities.
- FDA and DoD requirements for informed consent impede needed trauma research; ironically, these regulations make minimal risk research the most difficult to perform.
- Common misperceptions about HIPAA regulations present barriers to using and sharing data across systems for both direct patient care and research purposes.
CONCLUSION: The provision of tacit knowledge improves practice to reduce variation in patient care and outcomes. Yet participation in and support for resourcing of the processes and technologies that provide this knowledge remain limited in both the military and civilian sectors.
Related findings:
- The small size and scope of the Senior Visiting Surgeon Program (192 participants between 2005 and 2012, surgeons only [Knudson et al., 2014]) limits its impact and the exchange of tacit knowledge between military and civilian providers.
- The military’s teleconsultation programs in theater are jeopardized by a lack of funding and institutionalization.
- While best practices in telemedicine exist within the United States (e.g., Project ECHO), its use related to civilian trauma care is limited.
ANNEX 4-1
CASE REPORT: THE OPTIMAL USE OF DATA ACROSS THE CONTINUUM OF CARE30
This real-life case exemplifies how data could be used to improve care of a severely injured military service member. Through optimized data sharing and clinical decision support, the service member receives optimal, timely trauma care along a continuum of multiple levels of medical capability across three continents. Everything shown in italics is currently feasible from an information technology and electronics perspective, although not all systems are currently implemented in practice.
Care Under Fire
A 29-year-old male service member sustains a gunshot wound (GSW) to the head while on patrol following a .50 caliber sniper rifle projectile penetration of the right side of his helmet. A nonmedical but first-responder-trained member of his squad immediately assesses the opportunity for bleeding control, moves the casualty to a hasty casualty collection point (CCP) with cover and concealment, and notifies the squad leader of the casualty and his injury. The squad leader immediately contacts the platoon sergeant, who directs his platoon medic to the casualty to provide additional care.
Tactical Field Care
On arrival, the medic opens his tablet and establishes a linkage with the appropriate medical command center (MCC). At the MCC, a “smart map” identifies and displays the location of the call. An emergency medical communicator (EMC) instructs the medic to identify the potential patient, conduct a primary assessment, convey a “MIST” report (Mechanism of Injury, Injury, Signs/Symptoms, Treatments), and initiate a request for urgent transport and links this information stream to the patient’s existing medical database identifiers in the MCC computer.
__________________
30 This annex was adapted from a paper commissioned by the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, written by Elliott R. Haut, Johns Hopkins University School of Medicine and the Johns Hopkins Bloomberg School of Public Health; N. Clay Mann, University of Utah School of Medicine; and COL (ret) Russ S. Kotwal, Uniformed Services University of the Health Sciences and Texas A&M Health Science Center. The paper in its entirety is available on the study website at nationalacademies.org/TraumaCare.
At the CCP, the patient is noted to have strong carotid pulses, a heart rate (HR) of 130 beats per minute (bpm), and a respiratory rate (RR) of 75 breaths per minute. He is unresponsive and exhibits decorticate posturing. In the meantime, the “smart map” has identified and dispatched the closest acute care response vehicle to the patient. Patient information is linked to an existing health care record, giving the medic access to the patient’s health care database, current health problem list, current medications, allergies, and blood type. As patient care information is collected, it is automatically copied to the responding ground medic’s patient care record, to the responding transport medic’s patient care record, to the closest Role 2 military treatment facility (MTF), to the closest Role 3 MTF, and to the MCC. A real-time clinical decision support tool prompts the field medic to control the patient’s airway because of his depressed Glasgow Coma Scale score. A cricothyroidotomy is performed, and the casualty is placed on a mini-ventilator. The casualty is wrapped in a hypothermia prevention management kit, and an electronic tactical combat casualty care (TCCC) card is completed and transmitted.
Tactical Evacuation
An air transport vehicle arrives within 5 minutes of the evacuation request. The flight paramedic has received and reviewed all patient care documentation prior to arrival. A Role 2 MTF is located 45 minutes away, and a Role 3 MTF with a neurosurgeon is located 60 minutes away. Based on his decision support matrix, the flight paramedic directs the pilots to fly to the Role 3 MTF, provides supplemental oxygen to the casualty, and attaches an all-systems monitor to the casualty’s arm and across his chest. Physiologic data are acquired by the monitor’s computer chip, then analyzed on the scene and transmitted to the medic’s tablet and the MCC collocated with the Role 3 MTF. The flight medics are able to acquire the patient’s medical history electronically and treat accordingly.
Through the medic’s tablet, a video connection is established with the Role 3 MTF and MCC. An MCC emergency medical services (EMS) physician views the patient and his associated vital signs and requests additional Level III monitoring. The patient’s vital signs are blood pressure (BP) 80 mmHg (systolic only), HR 135 bpm, RR 30, Glasgow Coma Scale (GCS) score 3, and oxygen saturation (SpO2) 95 percent. His right pupil is measured at 3 mm and reactive. His left pupil is measured at 4 mm and also reactive. The patient exhibits decorticate posturing. His head wound dressing rapidly becomes soaked with blood. Analysis of all the data and active surveillance of decision support tools by the MCC computer and EMS physician lead to dosing with tranexamic acid, transfusion of 2 units of red blood cells (RBCs), and infusion of 3 percent hypertonic NaCl en
route. Ventilation is maintained by mini-vent, and SpO2 is sustained at 100 percent. An electronic tactical evacuation patient care record is completed and transmitted. The receiving MTF has received all patient vitals and physical exam and care documentation before the patient’s arrival. Accordingly, personnel at the MTF plan for a very brief emergency department assessment, empty the computed tomography (CT) scanner, and prepare a surgical suite with a waiting neurosurgeon.
Receiving Role 3 Military Treatment Facility
Upon arrival at the Role 3 MTF, the patient is noted to have the following vital signs: BP 153/108 mmHg, HR 69 bpm, RR set by ventilator, rectal temperature (T) 99.8°F, and GCS 3T. A cervical spine collar placed on the patient prior to arrival is rapidly removed in accordance with an evidence-based clinical practice guideline that summarizes the medical literature on the topic. A CT scan reveals a GSW to the head at the vertex of the skull with displacement of multiple bone fragments and evidence of diffuse cerebral edema, intraparenchymal hematoma, subarachnoid hemorrhage (SAH), subdural hemorrhage (SDH), and epidural hemorrhage (EDH) at the vertex in association with disruption of the mid third superior sagittal sinus.
The patient is taken to the operating room for decompressive hemicraniectomy, evacuation of subdural hematoma, placement of a right-sided external ventricular drain (EVD), and switch from a 7.5F field cricothyroidotomy tube to placement of an 8.0F tracheostomy. His postoperative CT reveals postsurgical changes, enlargement of right frontal lobe parenchymal hematoma, bilateral enlargement of the lateral ventricular horns, effacement of all basal cisterns, persistent subarachnoid and parafalcine SDH, a 3- to 4-mm midline shift rightward, multiple bone fragments within the brain parenchyma, and extensive scalp edema. He is administered thyroxin on a standard T4 protocol, as well as 3 percent NaCl. A second EVD is placed on the left side because of increasing bifrontal cerebral hemorrhaging reflected in an intracranial pressure (ICP) measurement of 23 mmHg. Following this intervention, ICP improves to 8-10 cmH2O and a cerebral perfusion pressure (CPP) of 60 mmHg. Repeat CT shows transcranial herniation at the site of the craniectomy with evidence of impending tonsillar and uncal herniation, as well as a small amount of right subfalcine herniation. The patient is transfused 2 units of RBCs, 2 units of fresh frozen plasma (FFP), and 1 unit of apheresis platelets. Tranexamic acid is also infused in an effort to control bleeding. Ultimately, the patient’s GCS improves to 7T, with a motor score of 4. He undergoes bronchoscopy to evaluate the effects of the surgical airway procedures and is found to have no evidence of tracheal injury. All medical charting is completed electroni
cally during the patient’s stay and automatically uploaded to the MCC computer. An abbreviated medical status report is exported to the critical care air transport team’s tablet for use during transport care.
Critical Care Air Transport Team: Movement Out of Theater (Postinjury Day #1)
The patient is transported to the flight line without incident. Sedation is increased prior to transport, resulting in a drop in GCS from 6T to GCS 3T. Tele-ICU monitoring is initiated and monitored by a Role 4 MTF. Answers to questions raised by the Tele-ICU team are provided instantaneously and care is modified based on those recommendations. In flight, CPP is maintained at greater than 60 mmHg with low-dose vasopressors, and ICP is maintained at less than 20 mmHg. The patient is transfused two units of RBCs and a 300-mL bolus of normal saline (NS). His hemoglobin increases from 8.2 to 9.2 g/dL. All patient care data collected during the flight are automatically uploaded to the MCC computer. Ventilation is provided via closed-loop ventilation that automatically adjusts the inspired oxygen content and RR in response to changes in oxygenation and end-tidal carbon dioxide monitoring. Upon arrival at Landstuhl Regional Medical Center in Germany, GCS has returned to 6T.
Role 4 Military Treatment Facility: Germany (Arrival Postinjury Day #1)
The patient is hemodynamically stable on arrival and is rapidly weaned off vasopressors. His GCS is initially 5T, with ICP of less than 20 mmHg; brainstem reflexes are intact. He is evaluated by a neurosurgeon, and a repeat CT is performed. The CT reveals intraventricular and large intraparenchymal hemorrhages. His ICP gradually increases to greater than 20 mmHg, and he is taken back to the operating room for evacuation of hematomas and drain placement. ICP improves, and GCS post-op is 4T. Enteral feeding is begun. Prophylaxis for deep venous thrombosis (DVT) and pulmonary embolism (PE) is begun early based on a personalized medicine approach utilizing the patient’s demographics, thromboelastography data, and precision genomics. Baseline duplex ultrasound of the extremities reveals no thrombi.
All medical charting is completed electronically during the patient’s stay and automatically uploaded to the MCC computer. An abbreviated medical status report is exported to the flight care team’s tablet for use during transport care. The patient is evacuated to the United States with a GCS of 4T. The flight is uneventful.
Role 5 Military Treatment Facility: United States
The initial care team in the United States has full access to the patient’s past and current care medical record, including CT images and intraoperative photographs, through the MCC. A patient care conference is held days before the patient arrives, and care plans are prepared and initiated upon his arrival. The patient is hemodynamically stable on admission, but transport trend vital signs suggest a slight but growing fever. The infectious diseases service is available upon the patient’s arrival, and its evaluation reveals pneumonia with a focus in the right lower lobe. Organisms isolated include Hemolytic streptococcus and Serratia rubidaea. The patient is administered antibiotics and antifungals. His GCS improves to 5T and ICP remains within normal range. His anasarca and brain edema improve, and he is able to tolerate enteric feeding.
The patient is transitioned to emerging consciousness rehabilitation after removal of the ventriculostomies on postinjury day 35. His pneumonia has resolved. Long-term follow-up demonstrates dramatically improved mental status. The patient can communicate reliably and is fully oriented. He does exhibit several cognitive deficits, such as reduced attention span and ability to concentrate, reduced visual spatial skills, and left-sided neglect, as well as some memory impairment. His physical status has also improved. He is able to use his right upper extremity and has modest antigravity strength in his right lower extremity. He has little use of his left upper or lower extremities as a result of the site of brain injury. He tolerates sitting for greater than 4 hours per day and can stand for 40 minutes at a time. His tracheostomy is decannulated and he can eat a regular diet.
This case represents an appropriate use of a learning health system to ensure seamless transitions of care between care teams, resulting in the best possible outcome for a severely injured patient.
REFERENCES
ACS (American College of Surgeons). 2014. Resources for optimal care of the injured patient. Chicago, IL: ACS.
ACS. 2015. National Trauma Data Bank 2015: Annual report. Chicago, IL: ACS.
ACS. 2016. ACS TQIP best practice guidelines. https://www.facs.org/quality-programs/trauma/tqip/best-practice (accessed February 16, 2016).
AMA (American Medical Association). 2016. Organized health care arrangement. http://www.ama-assn.org/ama/pub/physician-resources/solutions-managing-your-practice/codingbilling-insurance/hipaahealth-insurance-portability-accountability-act/hipaa-privacy-standards/organized-health-care-arrangement.page (accessed February 29, 2016).
Amadi-Obi, A., P. Gilligan, N. Owens, and C. O’Donnell. 2014. Telemedicine in pre-hospital care: A review of telemedicine applications in the pre-hospital environment. International Journal of Emergency Medicine 7:29.
APHA (American Public Health Association). 2003. Deep-vein thrombosis: Advancing awareness to protect patient lives. White paper. Presented at Public Health Leadership Conference on Deep-Vein Thrombosis, February 26, Washington, DC.
Arthurs, Z., D. Cuadrado, A. Beekley, K. Grathwohl, J. Perkins, R. Rush, and J. Sebesta. 2006. The impact of hypothermia on trauma care at the 31st Combat Support Hospital. American Journal of Surgery 191(5):610-614.
Badjatia, N., N. Carney, T. J. Crocco, M. E. Fallat, H. M. Hennes, A. S. Jagoda, S. Jernigan, P. B. Letarte, E. B. Lerner, T. M. Moriarty, P. T. Pons, S. Sasser, T. Scalea, C. L. Schleien, and D. W. Wright. 2008. Guidelines for prehospital management of traumatic brain injury, 2nd edition. Prehospital Emergency Care 12(Suppl. 1):S1-S52.
Baer, D. G. 2015. Military medical research investment in combat casualty care. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting One, May 18-19, Washington, DC.
Bailey, J. A., J. J. Morrison, and T. E. Rasmussen. 2013. Military trauma system in Afghanistan: Lessons for civil systems? Current Opinion in Critical Care 19(6):569-577.
Baily, M. A. 2008. Harming through protection? New England Journal of Medicine 358(8):768-769.
Baily, M. A., M. Bottrell, J. Lynn, and B. Jennings. 2006. The ethics of using QI methods to improve health care quality and safety. Hastings Center Report 36(4):S1-S40.
Berger, E. 2016. SIREN to replace NETT and ROC. Annals of Emergency Medicine 67(2):A22-A24.
Berry, S. M., J. T. Connor, and R. J. Lewis. 2015. The platform trial: An efficient strategy for evaluating multiple treatments. Journal of the American Medical Association 313(16):1619-1620.
Berwick, D. M. 2008. The science of improvement. Journal of the American Medical Association 299(10):1182-1184.
Black, I. H., and J. McManus. 2009. Pain management in current combat operations. Prehospital Emergency Care 13(2):223-227.
Blackbourne, L. H. 2009. The next generation of combat casualty care. Journal of Trauma 66(Suppl. 4):S27-S28.
Blackbourne, L. H., D. G. Baer, B. J. Eastridge, F. K. Butler, J. C. Wenke, R. G. Hale, R. S. Kotwal, L. R. Brosch, V. S. Bebarta, M. M. Knudson, J. R. Ficke, D. Jenkins, and J. B. Holcomb. 2012. Military medical revolution: Military trauma system. Journal of Trauma and Acute Care Surgery 73(6 Suppl. 5):S388-S394.
Borgman, M. A., P. C. Spinella, J. G. Perkins, K. W. Grathwohl, T. Repine, A. C. Beekley, J. Sebesta, D. Jenkins, C. E. Wade, and J. B. Holcomb. 2007. The ratio of blood products transfused affects mortality in patients receiving massive transfusions at a combat support hospital. Journal of Trauma 63(4):805-813.
Borio, L. 2015. Regulatory perspective on ethical and regulatory issues that impact a learning trauma care system. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting Three, September 16-17, Washington, DC.
Brosch, L. 2015. Regulatory perspective on ethical and regulatory issues that impact a learning trauma care system. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting Three, September 16-17, Washington, DC.
Brosch, L. R., J. B. Holcomb, J. C. Thompson, and P. R. Cordts. 2008. Establishing a human research protection program in a combatant command. Journal of Trauma 64(Suppl. 2):S9-S12.
Brown, J. 2015. Trauma research investment. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting Three, September 16-17, Washington, DC.
BTF (Brain Trauma Foundation). 2007. Guidelines for the management of severe traumatic brain injury. Journal of Neurotrauma 24(Suppl. 1):S1-S106.
Bulger, E. M., D. Snyder, K. Schoelles, C. Gotschall, D. Dawson, E. Lang, N. D. Sanddal, F. K. Butler, M. Fallat, P. Taillac, L. White, J. P. Salomone, W. Seifarth, M. J. Betzner, J. Johannigman, and N. McSwain, Jr. 2014. An evidence-based prehospital guideline for external hemorrhage control: American College of Surgeons Committee on Trauma. Prehospital Emergency Care 18(2):163-173.
Butler, F. K., Jr., and L. H. Blackbourne. 2012. Battlefield trauma care then and now: A decade of tactical combat casualty care. Journal of Trauma and Acute Care Surgery 73(6 Suppl. 5):S395-S402.
Butler, F. K., Jr., J. Haymann, and E. G. Butler. 1996. Tactical combat casualty care in special operations. Military Medicine 161(Suppl. 3):3-16.
Butler, F. K., L. H. Blackbourne, and K. Gross. 2015. The combat medic aid bag: 2025 CoTCCC top 10 recommended battlefield trauma care research, development, and evaluation priorities for 2015. Journal of Special Operations Medicine 15(4):7-19.
Callaway, D. W., E. R. Smith, G. Shapiro, J. S. Cain, S. D. McKay, and R. L. Mabry. 2011. The Committee for Tactical Emergency Casualty Care (C-TECC): Evolution and application of TCCC guidelines to civilian high threat medicine. Journal of Special Operations Medicine 11(2):95-100.
Camazine, M. N., M. R. Hemmila, J. C. Leonard, R. A. Jacobs, J. A. Horst, R. A. Kozar, G. V. Bochicchio, A. B. Nathens, H. M. Cryer, and P. C. Spinella. 2015. Massive transfusion policies at trauma centers participating in the American College of Surgeons Trauma Quality Improvement Program. Journal of Trauma and Acute Care Surgery 78(6 Suppl. 1):S48-S53.
Causey, M. W., D. E. Rivadeneira, and S. R. Steele. 2012. Historical and current trends in colon trauma. Clinics in Colon and Rectal Surgery 25(4):189-199.
CCCRP (Combat Casualty Care Research Program). 2015. U.S. Combat Casualty Care Research Program (CCCRP): Capability gap closure analysis. Washington, DC: Defense Health Agency, U.S. Department of Defense.
CDC (Centers for Disease Control and Prevention), National Institutes of Health (NIH), U.S. Department of Defense (DoD), and VA (U.S. Department of Veterans Affairs) Leadership Panel. 2013. Report to Congress on traumatic brain injury in the United States: Understanding the public health problem among current and former military personnel. Washington, DC: CDC, NIH, DoD, and VA.
Champion, H. R., W. S. Copes, W. J. Sacco, M. M. Lawnick, S. L. Keast, L. W. Bain, M. E. Flanagan, and C. F. Frey. 1990. Major trauma outcome study: Establishing national norms for trauma care. Journal of Trauma 30(11):1356-1365.
Chappuis, C. W., D. J. Frey, C. D. Dietzen, T. P. Panetta, K. J. Buechter, and I. Cohn, Jr. 1991. Management of penetrating colon injuries. A prospective randomized trial. Annals of Surgery 213(5):492-497.
Check, D. K., K. P. Weinfurt, C. B. Dombeck, J. M. Kramer, and K. E. Flynn. 2013. Use of central institutional review boards for multicenter clinical trials in the United States: A review of the literature. Clinical Trials 10(4):560-567.
ClinicalTrials.gov. 2015a. Prehospital tranexamic acid use for traumatic brain injury (TXA). https://clinicaltrials.gov/ct2/show/NCT01990768 (accessed February 16, 2016).
ClinicalTrials.gov. 2015b. Study of Tranexamic Acid during Air Medical Prehospital Transport Trial (STAAMP Trial). https://clinicaltrials.gov/ct2/show/NCT02086500 (accessed February 16, 2016).
ClinicalTrials.gov. 2015c. Tranexamic Acid Mechanisms and Pharmacokinetics in Traumatic Injury (TAMPITI). https://clinicaltrials.gov/ct2/show/NCT02535949 (accessed February 16, 2016).
Cohen, A. T., V. F. Tapson, J.-F. Bergmann, S. Z. Goldhaber, A. K. Kakkar, B. Deslandes, W. Huang, M. Zayaruzny, L. Emery, and F. A. Anderson, Jr. 2008. Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): A multinational cross-sectional study. The Lancet 371(9610):387-394.
Darst, J. R., J. W. Newburger, S. Resch, R. H. Rathod, and J. E. Lock. 2010. Deciding without data. Congenital Heart Disease 5(4):339-342.
Davis, J. S., S. S. Satahoo, F. K. Butler, H. Dermer, D. Naranjo, K. Julien, R. M. Van Haren, N. Namias, L. H. Blackbourne, and C. I. Schulman. 2014. An analysis of prehospital deaths: Who can we save? Journal of Trauma and Acute Care Surgery 77(2):213-218.
DHB (Defense Health Board). 2012. Prehospital use of ketamine in battlefield analgesia 2012. Falls Church, VA: DHB.
DHB. 2015. Combat trauma lessons learned from military operations of 2001-2013. Falls Church, VA: DHB.
Di Resta, N. 2015. Trauma research investment. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting Three, September 16-17, Washington, DC.
Dixon, N. M. 1994. The organizational learning cycle: How can we learn collectively. New York: McGraw-Hill Book Company.
DoD (U.S. Department of Defense), VA (U.S. Department of Veterans Affairs), HHS (U.S. Department of Health and Human Services), and ED (U.S. Department of Education). 2013. National research action plan: Responding to the executive order improving access to mental health services for veterans, service members, and military families (August 31, 2012). Washington, DC: DoD, VA, HHS, and ED.
Dover, M., A. R. Memon, H. Marafi, G. Kelly, and J. F. Quinlan. 2012. Factors associated with persistent sequelae after fasciotomy for acute compartment syndrome. Journal of Orthopaedic Surgery (Hong Kong) 20(3):312-315.
DuBose, J. J., G. Barmparas, K. Inaba, D. M. Stein, T. Scalea, L. C. Cancio, J. Cole, B. Eastridge, and L. Blackbourne. 2011. Isolated severe traumatic brain injuries sustained during combat operations: Demographics, mortality outcomes, and lessons to be learned from contrasts to civilian counterparts. Journal of Trauma 70(1):11-16.
Eastman, A. B., E. J. MacKenzie, and A. B. Nathens. 2013. Sustaining a coordinated, regional approach to trauma and emergency care is critical to patient health care needs. Health Affairs 32(12):2091-2098.
Eastridge, B. 2015. Military trauma system: Learning health system and translation to the civilian sector. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting Two, July 23-24, Washington, DC.
Eastridge, B. J., G. Costanzo, D. Jenkins, M. A. Spott, C. Wade, D. Greydanus, S. Flaherty, J. Rappold, J. Dunne, J. B. Holcomb, and L. H. Blackbourne. 2009. Impact of joint theater trauma system initiatives on battlefield injury outcomes. American Journal of Surgery 198(6):852-857.
Eastridge, B. J., C. E. Wade, M. A. Spott, G. Costanzo, J. Dunne, S. Flaherty, J. B. Holcomb, S. West, A. Apodaca, L. Blackbourne, and S. W. Casscells. 2010. Utilizing a trauma systems approach to benchmark and improve combat casualty care. Journal of Trauma 69(Suppl. 1):S5-S9.
Eastridge, B. J., M. Hardin, J. Cantrell, L. Oetjen-Gerdes, T. Zubko, C. Mallak, C. E. Wade, J. Simmons, J. Mace, and R. Mabry. 2011a. Died of wounds on the battlefield: Causation and implications for improving combat casualty care. Journal of Trauma and Acute Care Surgery 71(1):S4-S8.
Eastridge, B. J., R. Mabry, L. H. Blackbourne, and F. K. Butler. 2011b. We don’t know what we don’t know: Prehospital data in combat casualty care. U.S. Army Medical Department Journal 11-14.
Eastridge, B. J., R. L. Mabry, P. Seguin, J. Cantrell, T. Tops, P. Uribe, O. Mallett, T. Zubko, L. Oetjen-Gerdes, T. E. Rasmussen, F. K. Butler, R. S. Kotwal, J. B. Holcomb, C. Wade, H. Champion, M. Lawnick, L. Moores, and L. H. Blackbourne. 2012. Death on the battlefield (2001-2011): Implications for the future of combat casualty care. Journal of Trauma and Acute Care Surgery 73(6 Suppl. 5):S431-S437.
Elster, E. A., F. K. Butler, and T. E. Rasmussen. 2013. Implications of combat casualty care for mass casualty events. Journal of the American Medical Association 310(5):475-476.
EMSC (Emergency Medical Services for Children) National Resource Center. 2009. Gap analysis of EMS related research. Silver Spring, MD: EMSC.
Ennis, J. L., K. K. Chung, E. M. Renz, D. J. Barillo, M. C. Albrecht, J. A. Jones, L. H. Blackbourne, L. C. Cancio, B. J. Eastridge, S. F. Flaherty, W. C. Dorlac, K. S. Kelleher, C. E. Wade, S. E. Wolf, D. H. Jenkins, and J. B. Holcomb. 2008. Joint theater trauma system implementation of burn resuscitation guidelines improves outcomes in severely burned military casualties. Journal of Trauma 64(Suppl. 2):S146-S151.
Faden, R. R., N. E. Kass, S. N. Goodman, P. Pronovost, S. Tunis, and T. L. Beauchamp. 2013. An ethics framework for a learning health care system: A departure from traditional research ethics and clinical ethics. Hastings Center Report 43(1):S16-S27.
FDA (U.S. Food and Drug Administration). 2015. Draft guidance for industry and Food and Drug Administration staff: Adaptive designs for medical device clinical studies. http://www.fda.gov/ucm/groups/fdagov-public/@fdagov-meddev-gen/documents/document/ucm446729.pdf (accessed June 15, 2015).
Flynn, K. E., C. L. Hahn, J. M. Kramer, D. K. Check, C. B. Dombeck, S. Bang, J. Perlmutter, F. A. Khin-Maung-Gyi, and K. P. Weinfurt. 2013. Using central IRBs for multicenter clinical trials in the United States. PLoS One 8(1):e54999.
Fuenfer, M. M., P. C. Spinella, A. L. Naclerio, and K. M. Creamer. 2009. The U.S. military wartime pediatric trauma mission: How surgeons and pediatricians are adapting the system to address the need. Military Medicine 174(9):887-891.
Galvan, S. 2013. USAISR Burn Center receives FDA clearance for burn resuscitation technology. http://www.army.mil/article/103579/USAISR_Burn_Center_receives_FDA_clearance_for_burn_resuscitation_technology (accessed October 16, 2015).
GAO (U.S. Government Accountability Office). 2013. Defense health: Actions needed to help ensure combat casualty care research achieves goals. Washington, DC: GAO.
Garamone, J. 2015. DoD awards contract for electronic health records. http://www.defense.gov/News-Article-View/Article/612714 (accessed January 7, 2016).
Garcia, P., J. Rosen, C. Kapoor, M. Noakes, G. Elbert, M. Treat, T. Ganous, M. Hanson, J. Manak, C. Hasser, D. Rohler, and R. Satava. 2009. Trauma Pod: A semi-automated telerobotic surgical system. International Journal of Medical Robotics 5(2):136-146.
Goldhaber, S. Z., and V. F. Tapson. 2004. A prospective registry of 5,451 patients with ultrasound-confirmed deep vein thrombosis. American Journal of Cardiology 93(2):259-262.
Goldkind, S. F., L. R. Brosch, M. Biros, R. S. Silbergleit, and G. Sopko. 2014. Centralized IRB models for emergency care research. IRB: Ethics & Human Research 36(2):1-9.
Granger, C. V., S. J. Markello, J. E. Graham, A. Deutsch, T. A. Reistetter, and K. J. Ottenbacher. 2010. The Uniform Data System for Medical Rehabilitation report of patients with traumatic brain injury discharged from rehabilitation programs in 2000-2007. American Journal of Physical Medicine & Rehabilitation 89(4):265-278.
Gross, K. 2015. Joint Trauma System. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting One, May 18-19, Washington, DC.
Hafner, K. 2013. Skull surgery offers perils and potential. New York Times, July 16. http://www.nytimes.com/2013/07/16/science/skull-surgery-offers-perils-and-potential.html?_r=0 (accessed April 29, 2016).
Haider, A. H., L. C. Piper, C. K. Zogg, E. B. Schneider, J. A. Orman, F. K. Butler, R. T. Gerhardt, E. R. Haut, J. P. Mather, and E. J. MacKenzie. 2015. Military-to-civilian translation of battlefield innovations in operative trauma care. Surgery 158(6):1686-1695.
Haut, E. R., B. D. Lau, F. S. Kraenzlin, D. B. Hobson, P. S. Kraus, H. T. Carolan, A. H. Haider, C. G. Holzmueller, D. T. Efron, P. J. Pronovost, and M. B. Streiff. 2012. Improved prophylaxis and decreased rates of preventable harm with the use of a mandatory computerized clinical decision support tool for prophylaxis for venous thromboembolism in trauma. Archives of Surgery 147(10):901-907.
Haut, E. R., N. C. Mann, and R. S. Kotwal. 2016. Military Trauma Care’s Learning Health System: The importance of data-driven decision making. Paper commissioned by the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector. nationalacademies.org/TraumaCare.
Heide, C. 2015. Ethical and regulatory issues. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting Three, September 16-17, Washington, DC.
Helmick, K. L. Baugh, T. Lattimore, and S. Goldman. 2012. Traumatic brain injury: Next steps, research needed, and priority focus areas. Military Medicine 177(8 Suppl.):86-92.
HHS (U.S. Department of Health and Human Services). 2003. Summary of the HIPAA privacy rule (accessed February 29, 2016).
Holcomb, J. B., and D. B. Hoyt. 2015. Comprehensive injury research. Journal of the American Medical Association 313(14):1463-1464.
Holcomb, J. B., N. R. McMullin, L. Pearse, J. Caruso, C. E. Wade, L. Oetjen-Gerdes, H. R. Champion, M. Lawnick, W. Farr, S. Rodriguez, and F. K. Butler. 2007. Causes of death in U.S. Special Operations Forces in the Global War on Terrorism: 2001-2004. Annals of Surgery 245(6):986-991.
Holcomb, J. B., D. J. del Junco, E. E. Fox, C. E. Wade, M. J. Cohen, M. A. Schreiber, L. H. Alarcon, Y. Bai, K. J. Brasel, and E. M. Bulger. 2013. The PRospective, Observational, Multicenter, Major Trauma Transfusion (PROMMTT) study: Comparative effectiveness of a time-varying treatment with competing risks. JAMA Surgery 148(2):127-136.
Holcomb, J. B., D. P. Donathan, B. A. Cotton, D. J. Del Junco, G. Brown, T. V. Wenckstern, J. M. Podbielski, E. A. Camp, R. Hobbs, Y. Bai, M. Brito, E. Hartwell, J. R. Duke, and C. E. Wade. 2015a. Prehospital transfusion of plasma and red blood cells in trauma patients. Prehospital Emergency Care 19(1):1-9.
Holcomb, J. B., B. C. Tilley, S. Baraniuk, E. E. Fox, C. E. Wade, J. M. Podbielski, D. J. del Junco, K. J. Brasel, E. M. Bulger, R. A. Callcut, M. J. Cohen, B. A. Cotton, T. C. Fabian, K. Inaba, J. D. Kerby, P. Muskat, T. O’Keeffe, S. Rizoli, B. R. Robinson, T. M. Scalea, M. A. Schreiber, D. M. Stein, J. A. Weinberg, J. L. Callum, J. R. Hess, N. Matijevic, C. N. Miller, J. F. Pittet, D. B. Hoyt, G. D. Pearson, B. Leroux, G. van Belle, for the PROPPR Study Group. 2015b. Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs. a 1:1:2 ratio and mortality in patients with severe trauma: The PROPPR randomized clinical trial. Journal of the American Medical Association 313(5):471-482.
IHI (Institute for Healthcare Improvement). 2016. Office visit cycle time. http://www.ihi.org/resources/pages/measures/officevisitcycletime.aspx (accessed April 22, 2016).
IOM (Institute of Medicine). 1985. Assessing medical technologies. Washington, DC: National Academy Press.
IOM. 1999. Reducing the burden of injury: Advancing prevention and treatment. Washington, DC: National Academy Press.
IOM. 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
IOM. 2007a. Emergency medical services: At the crossroads. Washington, DC: The National Academies Press.
IOM. 2007b. Hospital-based emergency care: At the breaking point. Washington, DC: The National Academies Press.
IOM. 2010. Redesigning the clinical effectiveness research paradigm: Innovation and practice-based approaches: Workshop summary. Washington, DC: The National Academies Press.
IOM. 2011. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
IOM. 2013. Best care at lower cost: The path to continuously learning health care in America. Washington, DC: The National Academies Press.
IOM. 2015. Strategies to improve cardiac arrest survival: A time to act. Washington, DC: The National Academies Press.
James, B. C., and L. A. Savitz. 2011. How Intermountain trimmed health care costs through robust quality improvement efforts. Health Affairs (Millwood) 30(6):1185-1191.
JTS (Joint Trauma System). 2012a. CENTCOM JTTS CPG development, approval, implementation and monitoring process. San Antonio, TX: JTS.
JTS. 2012b. Clinical practice guideline: Unexploded ordinance management. San Antonio, TX: JTS.
JTS. 2012c. Clinical practice guideline: Ventilator associated pneumonia. San Antonio, TX: JTS.
JTS. 2013a. Clinical practice guideline: Burn care. San Antonio, TX: JTS.
JTS. 2013b. Clinical practice guideline: Management of pain, anxiety and delirium in injured warfighters. San Antonio, TX: JTS.
JTS. 2014. Clinical practice guideline: Management of patients with severe head trauma. San Antonio, TX: JTS.
Kelly, J. F., A. E. Ritenour, D. F. McLaughlin, K. A. Bagg, A. N. Apodaca, C. T. Mallak, L. Pearse, M. M. Lawnick, H. R. Champion, C. E. Wade, and J. B. Holcomb. 2008. Injury severity and causes of death from Operation Iraqi Freedom and Operation Enduring Freedom: 2003-2004 versus 2006. Journal of Trauma 64(Suppl. 2):S21-S26.
Kerwin, A. J., E. R. Haut, J. B. Burns, J. J. Como, A. Haider, N. Stassen, and P. Dahm. 2012. The Eastern Association of the Surgery of Trauma approach to practice management guideline development using Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) methodology. Journal of Trauma and Acute Care Surgery 73(5 Suppl. 4):S283-S287.
Knudson, M. M., and T. E. Rasmussen. 2012. The Senior Visiting Surgeons Program: A model for sustained military-civilian collaboration in times of war and peace. Journal of Trauma and Acute Care Surgery 73(6 Suppl. 5):S536-S542.
Knudson, M. M., T. W. M. Evans, R. Fang, K. D. Martin, W. Dorlac, D. L. Gillespie, K. J. Cherry, Jr., and T. E. Rasmussen. 2014. A concluding after-action report of the Senior Visiting Surgeon Program with the United States military at Landstuhl Regional Medical Center, Germany. Journal of Trauma and Acute Care Surgery 76(3):878-883.
Kotwal, R. S., H. R. Montgomery, B. M. Kotwal, H. R. Champion, F. K. Butler, Jr., R. L. Mabry, J. S. Cain, L. H. Blackbourne, K. K. Mechler, and J. B. Holcomb. 2011. Eliminating preventable death on the battlefield. Archives of Surgery 146(12):1350-1358.
Kotwal, R. S., F. K. Butler, E. P. Edgar, S. A. Shackelford, D. R. Bennett, and J. A. Bailey. 2013a. Saving lives on the battlefield: A Joint Trauma System review of pre-hospital trauma care in Combined Joint Operating Area-Afghanistan (CJOA-A). https://www.naemt.org/docs/default-source/education-documents/tccc/10-9-15-updates/centcomprehospital-final-report-130130.pdf?sfvrsn=2 (accessed January 13, 2015).
Kotwal, R. S., F. K. Butler, H. R. Montgomery, T. J. Brunstetter, G. Y. Diaz, J. W. Kirkpatrick, N. L. Summers, S. A. Shackelford, J. B. Holcomb, and J. A. Bailey. 2013b. The Tactical Combat Casualty Care Casualty Card TCCC guidelines—proposed change 1301. Journal of Special Operations Medicine 13(2):82-87.
Kotwal, R. S., J. T. Howard, J. A. Orman, B. W. Tarpey, J. A. Bailey, H. R. Champion, R. L. Mabry, J. B. Holcomb, and K. R. Gross. 2016. The effect of a golden hour policy on the morbidity and mortality of combat casualties. JAMA Surgery 151(1):15-24.
Kragh, J. F., Jr., T. J. Walters, D. G. Baer, C. J. Fox, C. E. Wade, J. Salinas, and J. B. Holcomb. 2009. Survival with emergency tourniquet use to stop bleeding in major limb trauma. Annals of Surgery 249(1):1-7.
Kragh, J. F., Jr., M. A. Dubick, J. K. Aden, A. L. McKeague, T. E. Rasmussen, D. G. Baer, and L. H. Blackbourne. 2015. U.S. military use of tourniquets from 2001 to 2010. Prehospital Emergency Care 19(2):184-190.
Landman, A. B., C. H. Lee, C. Sasson, C. M. Van Gelder, and L. A. Curry. 2012. Prehospital electronic patient care report systems: Early experiences from emergency medical services agency leaders. PLoS One 7(3):e32692.
Lang, E. S., D. W. Spaite, Z. J. Oliver, C. S. Gotschall, R. A. Swor, D. E. Dawson, and R. C. Hunt. 2012. A national model for developing, implementing, and evaluating evidence-based guidelines for prehospital care. Academic Emergency Medicine 19(2):201-209.
Latifi, R., R. S. Weinstein, J. M. Porter, M. Ziemba, D. Judkins, D. Ridings, R. Nassi, T. Valenzuela, M. Holcomb, and F Leyva. 2007. Telemedicine and telepresence for trauma and emergency care management. Scandinavian Journal of Surgery 96(4):281-289.
Latifi, R., G. J. Hadeed, P. Rhee, T. O’Keeffe, R. S. Friese, J. L. Wynne, M. L. Ziemba, and D. Judkins. 2009. Initial experiences and outcomes of telepresence in the management of trauma and emergency surgical patients. American Journal of Surgery 198(6):905-910.
Lauer, M. S., and R. B. D’Agostino. 2013. The randomized registry trial—the next disruptive technology in clinical research? New England Journal of Medicine 369(17):1579-1581.
Lein, B., J. Holcomb, S. Brill, S. Hetz, and T. McCrorey. 1999. Removal of unexploded ordnance from patients: A 50-year military experience and current recommendations. Military Medicine 164(3):163-165.
Ling, G., F. Bandak, R. Armonda, G. Grant, and J. Ecklund. 2009. Explosive blast neurotrauma. Journal of Neurotrauma 26(6):815-825.
Malchow, R. J., and I. H. Black. 2008. The evolution of pain management in the critically ill trauma patient: Emerging concepts from the Global War on Terrorism. Critical Care Medicine 36(7):S346-S357.
Maldon, A., L. L. Pressler, S. E. Buyer, D. S. Zakheim, M. R. Higgins, P. W. Chiarelli, E. P. Giambastiani, J. R. Kerrey, and C. P. Carney. 2015. Report of the Military Compensation and Retirement Modernization Commission: Final report. Arlington, VA: Military Compensation and Retirement Modernization Commission.
Markell, K. W., E. M. Renz, C. E. White, M. E. Albrecht, L. H. Blackbourne, M. S. Park, D. A. Barillo, K. K. Chung, R. A. Kozar, J. P. Minei, S. M. Cohn, D. N. Herndon, L. C. Cancio, J. B. Holcomb, and S. E. Wolf. 2009. Abdominal complications after severe burns. Journal of the American College of Surgeons 208(5):940-947.
Martin-Gill, C., J. B. Gaither, B. L. Bigham, J. B. Myers, D. F. Kupas, and D. W. Spaite. 2016. National prehospital evidence-based guidelines strategy: A summary for EMS stakeholders. Prehospital Emergency Care 20(2):175-183.
McGlynn, E. A., S. M. Asch, J. Adams, J. Keesey, J. Hicks, A. DeCristofaro, and E. A. Kerr. 2003. The quality of health care delivered to adults in the United States. New England Journal of Medicine 348(26):2635-2645.
McManus, J., T. Hurtado, A. Pusateri, and K. J. Knoop. 2007. A case series describing thermal injury resulting from zeolite use for hemorrhage control in combat operations. Prehospital Emergency Care 11(1):67-71.
McManus, J., J. Salinas, M. Morton, C. Lappan, and R. Poropatich. 2008. Teleconsultation program for deployed soldiers and healthcare professionals in remote and austere environments. Prehospital and Disaster Medicine 23(3):210-216.
Meador, C. 2014. Burn Resuscitation Decision Support System (BRDSS). Final report. Houston, TX: Arcos, Inc.
METRC (Major Extremity Trauma Research Consortium). 2015. Major Extremity Trauma Research Consortium: Improving outcomes through collaborative research. https://metrc.org (accessed December 22, 2015).
METRC. 2016. Building a clinical research network in trauma orthopaedics: The Major Extremity Trauma Research Consortium (METRC). Journal of Orthopaedic Trauma 30(7):353-361.
Miles, D. 2013. DoD expands trauma registry to include front-line care. http://archive.defense.gov/news/newsarticle.aspx?id=121166 (accessed February 12, 2015).
Miller, F. G., and E. J. Emanuel. 2008. Quality-improvement research and informed consent. New England Journal of Medicine 358:765-767.
Monegain, B. 2015. Cerner rides high with DoD deal. http://www.healthcareitnews.com/news/cerner-rides-high-dod-deal (accessed May 10, 2016).
Moore, E. E., M. M. Knudson, C. W. Schwab, D. D. Trunkey, J. A. Johannigman, and J. B. Holcomb. 2007. Military–civilian collaboration in trauma care and the Senior Visiting Surgeon Program. New England Journal of Medicine 357(26):2723-2727.
Moses, H., 3rd, D. H. Matheson, S. Cairns-Smith, B. P. George, C. Palisch, and E. R. Dorsey. 2015. The anatomy of medical research: US and international comparisons. Journal of the American Medical Association 313(2):174-189.
MRMC (U.S. Army Medical Research and Material Command) and DHA (U.S. Defense Health Agency). 2015. DoD Combat Casualty Care Research Program: Policy review. Fort Detrick, MD: MRMC and DHA.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2015. Optimizing the nation’s investment in academic research: A new regulatory framework for the 21st century: Part 1. Washington, DC: The National Academies Press.
NCIPC (National Center for Injury Prevention and Control). 2005. CDC acute injury care research agenda: Guiding research for the future. Atlanta, GA: Centers for Disease Control and Prevention.
NEMSIS (National EMS Information System). 2013. History of NEMSIS. http://www.nemsis.org/theProject/historyofNemsis.html (accessed February 21, 2016).
NHTSA (National Highway Traffic Safety Administration). 2001. National EMS research agenda. Washington, DC: NHTSA.
NHTSA. 2004. Trauma system agenda for the future. Washington, DC: NHTSA.
NIH (National Institutes of Health). 1994. A report of the Task Force on Trauma Research. Bethesda, MD: NIH.
NIH. 1996. Waiver of informed consent requirements in certain emergency research. Federal Register 61(192, Oct. 2):51531-51533.
NIH. 2016a. About FITBIR. https://fitbir.nih.gov/jsp/about/index.jsp (accessed February 12, 2016).
NIH. 2016b. Estimates of funding for various research, condition, and disease categories (RCDC). https://report.nih.gov/categorical_spending.aspx (accessed February 20, 2016).
NIH. 2016c. Network for Emergency Care Clinical Trials: Strategies to Innovate EmeRgENcy Care Clinical Trials Network (SIREN)—Clinical Coordinating Center (CCC) (U24). http://grants.nih.gov/grants/guide/rfa-files/RFA-NS-16-014.html (accessed July 14, 2016).
Nonaka, I. 1994. A dynamic theory of organizational knowledge creation. Organization Science 5(1):14-37.
NRC (National Research Council). 1966. Accidental death and disability: The neglected disease of modern society. Washington, DC: National Academy Press.
NRC and IOM. 1985. Injury in America: A continuing public health problem. Washington, DC: National Academy Press.
PA Bulletin. 2011. Required ground and air ambulance equipment and supplies, 41 PA.B. 2296. http://pabulletin.com/secure/data/vol41/41-18/739.html (accessed February 29, 2016).
Palm, K., A. Apodaca, D. Spencer, G. Costanzo, J. Bailey, L. H. Blackbourne, M. A. Spott, and B. J. Eastridge. 2012a. Evaluation of military trauma system practices related to damage-control resuscitation. Journal of Trauma and Acute Care Surgery 73(6 Suppl. 5):S459-S464.
Palm, K., A. Apodaca, D. Spencer, G. Costanzo, J. Bailey, G. Fortuna, L. H. Blackbourne, M. A. Spott, and B. J. Eastridge. 2012b. Evaluation of military trauma system practices related to complications after injury. Journal of Trauma and Acute Care Surgery 73(6 Suppl. 5):S465-S471.
Pasquale, M., and T. C. Fabian. 1998. Practice management guidelines for trauma from the Eastern Association for the Surgery of Trauma. Journal of Trauma 44(6):941-956.
Patsopoulos, N. A. 2011. A pragmatic view on pragmatic trials. Dialogues in Clinical NeuroSciences 13(2):217-224.
Pennsylvania Department of Health, Bureau of Emergency Medical Services. 2008. Pennsylvania statewide basic life support protocols. Harrisburg, PA: Pennsylvania Department of Health.
Perkins, J. G., L. R. Brosch, A. C. Beekley, K. L. Warfield, C. E. Wade, and J. B. Holcomb. 2012. Research and analytics in combat trauma care: Converting data and experience to practical guidelines. Surgical Clinics of North America 92(4):1041-1054.
PETAL (Prevention and Early Treatment of Acute Lung Injury) Network. 2015. Who we are. http://petalnet.org (accessed December 22, 2015).
Pronovost, P., D. Needham, S. Berenholtz, D. Sinopoli, H. Chu, S. Cosgrove, B. Sexton, R. Hyzy, R. Welsh, G. Roth, J. Bander, J. Kepros, and C. Goeschel. 2006. An intervention to decrease catheter-related bloodstream infections in the ICU. New England Journal of Medicine 355(26):2725-2732.
Pruitt, B. A., Jr., and T. E. Rasmussen. 2014. Vietnam (1972) to Afghanistan (2014): The state of military trauma care and research, past to present. Journal of Trauma and Acute Care Surgery 77(3 Suppl. 2):S57-S65.
Rasmussen, T. E. 2015. Trauma research investment. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting Three, September 16-17, Washington, DC.
Rasmussen, T. E., and D. G. Baer. 2014. No drift. JAMA Surgery 149(3):221-222.
Rasmussen, T. E., and H. B. Shumacker. 2014. US Combat Casualty Care Research Program: Answering the call. Falls Church, VA: DHB.
Rasmussen, T. E., P. A. Reilly, and D. G. Baer. 2014. Why military medical research? Military Medicine 179(Suppl. 8):1-2.
Rhee, P., B. Joseph, V. Pandit, H. Aziz, G. Vercruysse, N. Kulvatunyou, and R. S. Friese. 2014. Increasing trauma deaths in the United States. Annals of Surgery 260(1):13-21.
Richards, C. 2015. Is the amount of disease-specific NIH funding related to burden of disease? http://dotdata.cc/is-the-amount-of-disease-specific-nih-funding-related-to-burden-of-disease (accessed February 11, 2016).
Ritenour, A. E., W. C. Dorlac, R. Fang, T. Woods, D. H. Jenkins, S. F. Flaherty, C. E. Wade, and J. B. Holcomb. 2008. Complications after fasciotomy revision and delayed compartment release in combat patients. Journal of Trauma 64(Suppl. 2):S153-S161.
Roberts, M. 2011. Treating a gunshot to the head—extreme brain surgery. BBC News, January 10. http://www.bbc.com/news/health-12152501 (accessed April 29, 2016).
Rogers, F. B., M. D. Cipolle, G. Velmahos, G. Rozycki, and F. A. Luchette. 2002. Practice management guidelines for the prevention of venous thromboembolism in trauma patients: The East Practice Management Guidelines Work Group. Journal of Trauma 53(1):142-164.
Rotondo, M., T. Scalea, A. Rizzo, K. Martin, and J. Bailey. 2011. The United States military Joint Trauma System assessment: A report commissioned by the U.S. Central Command Surgeon, sponsored by Air Force Central Command, a strategic document to provide a platform for tactical development. Washington, DC: DoD.
Salinas, J., K. K. Chung, E. A. Mann, L. C. Cancio, G. C. Kramer, M. L. Serio-Melvin, E. M. Renz, C. E. Wade, and S. E. Wolf. 2011. Computerized decision support system improves fluid resuscitation following severe burns: An original study. Critical Care Medicine 39(9):2031-2038.
Sasaki, L. S., R. D. Allaben, R. Golwala, and V. K. Mittal. 1995. Primary repair of colon injuries: A prospective randomized study. Journal of Trauma 39(5):895-901.
Sauer, S. W., J. B. Robinson, M. P. Smith, K. R. Gross, R. S. Kotwal, R. Mabry, F. K. Butler, Z. T. Stockinger, J. A. Bailey, M. E. Mavity, and D. A. Gillies. 2014. Saving lives on the battlefield (Part II)—one year later: A Joint Theater Trauma System & Joint Trauma System review of pre-hospital trauma care in Combined Joint Operating Area-Afghanistan (CJOA-A). http://www.naemsp.org/Documents/E-News%20-%20loaded%20docs%20for%20link/Saving%20Lives%20on%20the%20Battlefield%20II.pdf (accessed January 13, 2015).
Sayre, M. R., L. J. White, L. H. Brown, S. D. McHenry, and National EMS Research Strategic Plan Writing Team. 2005. The National EMS Research Strategic Plan. Prehospital Emergency Care 9(3):255-266.
Schauer, S., J. Robinson, R. Mabry, and J. Howard. 2014. Battlefield analgesia: TCCC guidelines are not being followed. Journal of Special Operations Medicine 15(1):85-89.
Seymour, C. W., J. M. Kahn, C. Martin-Gill, C. W. Callaway, D. C. Angus, and D. M. Yealy. 2014. Creating an infrastructure for comparative effectiveness research in emergency medical services. Academic Emergency Medicine 21(5):599-607.
Shafi, S., S. A. Barnes, N. Rayan, R. Kudyakov, M. Foreman, H. G. Cryer, H. B. Alam, W. Hoff, and J. Holcomb. 2014. Compliance with recommended care at trauma centers: Association with patient outcomes. Journal of the American College of Surgeons 219(2):189-198.
Spinella, P. C., and J. B. Holcomb. 2009. Resuscitation and transfusion principles for traumatic hemorrhagic shock. Blood Reviews 23(6):231-240.
Spott, M. A. 2015. Military trauma data and information systems. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting Two, July 23-24, Washington, DC.
Starr, B. 2011. Military surgeons assist in Giffords’ treatment. CNN, January 11. http://www.cnn.com/2011/US/01/11/arizona.shootings.military (accessed April 29, 2016).
Stockinger, Z. T. 2015. Joint Trauma System Defense Center of Excellence. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting Two, July 23-24, Washington, DC.
Stone, H. H., and T. C. Fabian. 1979. Management of perforating colon trauma: Randomization between primary closure and exteriorization. Annals of Surgery 190(4):430-435.
Streiff, M. B., H. T. Carolan, D. B. Hobson, P. S. Kraus, C. G. Holzmueller, R. Demski, B. D. Lau, P. Biscup-Horn, P. J. Pronovost, and E. R. Haut. 2012. Lessons from the Johns Hopkins Multi-Disciplinary Venous Thromboembolism (VTE) Prevention Collaborative. British Medical Journal 344:e3935.
Therien, S. P., M. E. Nesbitt, A. M. Duran-Stanton, and R. T. Gerhardt. 2011. Prehospital medical documentation in the Joint Theater Trauma Registry: A retrospective study. Journal of Trauma 71(Suppl. 1):S103-S108.
Trunkey, D. D. 1983. Trauma. Scientific American 249(2):28-35.
U.S. Burden of Disease Collaborators. 2013. The state of US health, 1990-2010: Burden of diseases, injuries, and risk factors. Journal of the American Medical Association 310(6):591-608.
USAISR (U.S. Army Institute of Surgical Research). 2015. JTS clinical practice guidelines. http://www.usaisr.amedd.army.mil/cpgs.html (accessed October 22, 2015).
USAISR. 2016. Joint Trauma System. http://www.usaisr.amedd.army.mil/10_jts.html (accessed February 21, 2016).
van Middendorp, J. J., H. C. Allison, S. Ahuja, D. Bracher, C. Dyson, J. Fairbank, A. Gall, A. Glover, L. Gray, W. E. Masri, A. Uttridge, and K. Cowan. 2016. Top ten research priorities for spinal cord injury: The methodology and results of a British priority setting partnership. Spinal Cord 54(5):341-346.
Weisbrod, A. B., C. Rodriguez, R. Bell, C. Neal, R. Armonda, W. Dorlac, M. Schreiber, and J. R. Dunne. 2012. Long-term outcomes of combat casualties sustaining penetrating traumatic brain injury. Journal of Trauma and Acute Care Surgery 73(6):1525-1530.
Welch, G. F., D. H. Sonnenwald, H. Fuchs, B. Cairns, K. Mayer-Patel, H. M. Soderholm, R. Yang, A. State, H. Towles, A. Ilie, M. K. Ampalam, S. Krishnan, V. Noel, M. Noland, and J. E. Manning. 2009. 3D medical collaboration technology to enhance emergency healthcare. Journal of Biomedical Discovery and Collaboration 4:4.
Yee, A. 2015. National Association of EMS Physicians’ perspective on charge to the committee. Paper presented to the Committee on Military Trauma Care’s Learning Health System and Its Translation to the Civilian Sector, Meeting One, May 18-19, Washington, DC.




















































































