2
The Outbreak
Ebola virus was identified in 1976 after simultaneous, highly lethal outbreaks of hemorrhagic fever occurred in northern Zaire (now the Democratic Republic of Congo [DRC]) and southern Sudan, each caused by a different strain of the virus (Muyembe-Tamfum et al., 2012). Ninety percent of these Ebola-Zaire cases, and 50 percent of these Ebola-Sudan cases, resulted in death (CDC, 2014). Sporadic outbreaks of Ebola subsequently arose in Africa, mainly within the Congo and Nile basins. Three additional strains of Ebola virus, also named for the location where they were first isolated, have been identified: Taï Forest, Bundibugyo, and Reston. Ebola-Reston, although first identified among laboratory monkeys in the United States, apparently originated in the Philippines among a single species of macaques from one breeding facility (Miranda and Miranda, 2011). Ebola-Reston is the only known filovirus that apparently does not cause severe disease in humans. It does kill monkeys, however, and it has been recently recovered from infected swine in Southeast Asia.
LESSONS FROM PREVIOUS EBOLA OUTBREAKS
David Heymann of Public Health England and Jean Jacques Muyembe-Tamfum of Kinshasa University in the DRC recounted their firsthand experience with several Ebola outbreaks, including the initial recognized emergence of Ebola-Zaire. Muyembe, one of the first physicians to arrive at the scene of that outbreak, was sent by his country’s ministry of health to determine the etiology of a serious “mysterious disease.” Ebola virus was isolated from blood samples he collected. Nearly two decades later, this experience was brought to bear when
Muyembe correctly suspected that Ebola virus was the cause of a major outbreak of hemorrhagic fever spreading through hospitals in Kikwit, DRC.
Epidemiological observations since gathered from Ebola outbreaks ranging in size from a few individuals to hundreds of cases inform current understanding of disease risk, as described by Heymann:
- Ebola virus is transmitted by blood and other bodily fluids and not, apparently, by airborne means.
- Nosocomial transmission that involves health care workers can drive disease throughout entire communities, but such outbreaks can be prevented.
- Community outbreaks end spontaneously as residents learn how to prevent transmission.
- Humans can be infected through contact with infected nonhuman primates; bats may play a role in viral transmission.
- The virus emerges periodically through cross-species transmission.
Both Heymann and Muyembe emphasized that Ebola outbreaks can be stopped. Indeed, Muyembe observed, all seven Ebola outbreaks that have occurred in the DRC have been geographically confined—and in the two instances where infected people reached the capital city, Kinshasa, few secondary infections occurred.
“When Ebola virus is introduced in the villages, most of the time the outbreak will stop spontaneously,” Muyembe observed. “For example,” he continued, “in 1976 in Yambuku, 55 villages were affected, and in Kikwit 25 villages were affected. Most of these villages reported only less than 10 affected patients. So we think that the village will stop the propagation of the virus.” On the other hand, he noted, “When the virus is introduced in the hospital, there is amplification. The chain of transmission is very long.” This is because, as Heymann pointed out, the initial symptoms of Ebola are relatively mild, and common to several febrile diseases. Thus, early-stage Ebola is difficult to recognize, putting health workers who care for these patients at risk of infection and igniting chains of transmission.
Nevertheless, all Ebola outbreaks can be contained by applying the following basic principles, according to Heymann:
- Patient identification, isolation and protection of health workers, and infection control.
- Surveillance and contact tracing, as well as fever surveillance with rapid diagnosis and isolation.
- Community understanding with safe patient and body transport systems, safe burial, and household and environmental decontamination.
“Those are the strategies that stopped the Kikwit outbreak, and those are the strategies that continue today to be the most effective in dealing with Ebola,” he stated.
DIFFERENCES WITH PREVIOUS EBOLA OUTBREAKS
Daniel Bausch of Tulane University noted that countries in which outbreaks comprising at least 20 cases of Marburg or Ebola have occurred not only in some of the world’s weakest health systems but in areas that are also plagued by ongoing civil unrest (Bausch and Schwarz, 2014). While this is equally true in Central and West Africa, decades of experience with Ebola have taught Central Africans to suspect it when “mysterious” outbreaks occur, and they have allowed the establishment of capacity and procedures for its early identification and control. By comparison, West African countries, lacking experience with Ebola outbreaks, were poorly prepared for its emergence at every level (WHO, 2015d).
This contrast was apparent in a comparison Muyembe made between the West African epidemic and an unrelated Ebola outbreak that began in July 2014 in an isolated village in the DRC (Maganga et al., 2014). The previously described community approach and basic infection control measures were implemented quickly, and the outbreak—which claimed dozens of lives, as compared with thousands in West Africa—was extinguished by November.
Bausch broadened this comparison to highlight multiple differences between the West African Ebola epidemic and all previous outbreaks (see Table 2-1). Some of these characteristics convey the magnitude of the West African crisis (e.g., case numbers, duration, cost); some identify contributing factors (e.g., exported cases, involvement of urban areas, community resistance); and, some—such as the number of organizations involved in the response—may reflect both cause and effect, since lack of coordination between agencies proved problematic, he observed.
Focusing on factors that inflamed the Ebola epidemic, Bausch emphasized the connectedness of the affected populations across the borders of their three countries. The prefecture of Guéckédou—site of the initial outbreak—borders both Liberia and Sierra Leone, and its people tend to affiliate more according to regional ethnic groups than by nationality, he reported. A family in this region might live in Guinea, but have their children cross the border to go to school in Liberia. Furthermore, traveling long distances is not unusual. Bausch described how buses and taxis from faraway villages make the long journey to regions’ capitals—cities of thousands of people like Guéckédou—every day, and vice versa.
West Africa in general is characterized by exceptional population mobility across porous borders, driven to a large extent by poverty (WHO, 2015d). These circumstances presented a major—and largely novel—challenge to containing Ebola, Bausch observed. Population mobility impeded the tracing of border-crossing contacts while encouraging the growth of a smoldering epidemic—one
TABLE 2-1 Comparison of Previous Ebola Outbreaks to the 2014 West African Ebola Epidemic
| Aspect | Previous Outbreaks | West Africa 2014–2015 |
|---|---|---|
| Number of cases | Typically 100–200 | ~25,000 |
| Number of responding countries, per outbreak | Usually 1 | 3 (plus ~25 imported) |
| Duration | 2–4 months | > 1 year |
| Exported cases outside area | Extremely rare | Frequent |
| Community cooperation | Occasional resistance | Frequent resistance |
| Organizations responding, per outbreak | 5–10 | > 100 |
| Urban areas | Spared | Heavily involved |
| Number of Ebola treatment units employed, per outbreak | Typically 1–2 | > 50 |
| Number of diagnostic labs involved in response, per outbreak | Typically 1–2 | > 50 |
| Cost of response, per outbreak | < $5 million | Nearing $1 billion |
SOURCE: Bausch presentation, March 24, 2015.
that has been compared to a forest fire. “You can’t put out 98 percent of it. You have to get it all, and if you don’t get it all, then it risks coming back,” he said.
“We had capacity to meet this challenge in the ways that we usually do with a 2- to 4-month outbreak,” Bausch continued. “None of us could have really ever anticipated something this large, including myself. . . . I think if you’d asked [filovirus experts] . . . a year and a half ago ‘Are we going to have 25,000 cases of Ebola in West Africa?,’ most of us would have not said that was a likelihood.”
CHALLENGES WITH EBOLA CONTROL
Muyembe discussed several significant challenges that have arisen during efforts to control Ebola in Central Africa over the past 20 years. Most of these outbreaks occurred in remote locations; thus, logistics were difficult to organize, from transporting responders to the site, to conducting field activities, to pursuing contact follow-up, he noted.
Once on site, medical teams encountered profound sociocultural barriers to disease control, such as resistance to adopting protective burial practices. Such problems were addressed through community engagement and social mobilization, incorporating mass gatherings and messages delivered by posting and radio, Muyembe said—and also by adapting disease control practices to make them more acceptable to local people. For example, after burial protocols used
in Kikwit were rejected, new routines that safely involved family members were instituted by responders to a subsequent outbreak in Isiro, DRC.
Another challenge was providing adequate isolation wards. Often these facilities were distant from patients’ homes and families, at the very least decrepit and sometimes lacking electricity and running water—not places anyone would go willingly, Muyembe observed. His team has worked to improve these circumstances, and thereby, adherence with isolation practices. They now also organize visits by members of the community to Ebola treatment centers to dispel the notion that they are merely “places for dying.” Furthermore, Ebola survivors—whose possessions have been lost to decontamination—are now compensated with mattresses, money, and other necessities.
Public communications have also been improved with experience, Muyembe said. Contradictory messages used in early Marburg and Ebola outbreaks that simultaneously advised treatment for people with symptoms while warning that there was no cure for these diseases have been replaced with messages promoting safe behavior.
In response to these challenges, the DRC has implemented a community approach to Ebola control, encompassing all levels of public health, from the affected villages to a coordinating committee at the national level. “The community approach . . . [promotes] early community engagement and early ownership for implementing Ebola prevention and control measures,” Muyembe said. Its objectives include sensitization of community members to the importance of disease control, prevention and management of community resistance, dialogue and negotiation with members of affected families and the community at large, and promotion of Ebola survivors’ reintegration in their communities. “You cannot organize the fight against Ebola without the community,” he insisted.
CRISIS IN WEST AFRICA
Unfortunately, many lessons learned from nearly four decades of Ebola outbreaks in Central Africa did not inform the response when the virus emerged in Guinea in 2013. As noted in a recent report from the WHO, Ebola in West Africa was “an old disease in a new context that favored rapid and initially invisible spread” (WHO, 2015d). Workshop participants discussed numerous factors that fueled the West African Ebola epidemic and which factors distinguished this episode from past outbreaks. Workshop participants also examined knowledge gained from this latest and most devastating encounter with the virus, and considered how to apply it in the future.
The Response: Too Little, Too Late
The international response to the West African Ebola epidemic has been widely characterized as slow, weak, and disorganized (Gostin and Friedman,
2014; MSF, 2015; Nature, 2014; Presidential Commission for the Study of Bioethical Issues, 2015). In the United States, the Presidential Commission for the Study of Bioethical Issues (2015) noted that this is nothing new:
As has been observed throughout history, the beginnings of major epidemics can be slow and confusing and are often only recognized in retrospect. Vital epidemiological and clinical information about Ebola and the western African epidemic was incomplete and rapidly changing. Weaknesses in global and local public health governance and infrastructure also slowed action.
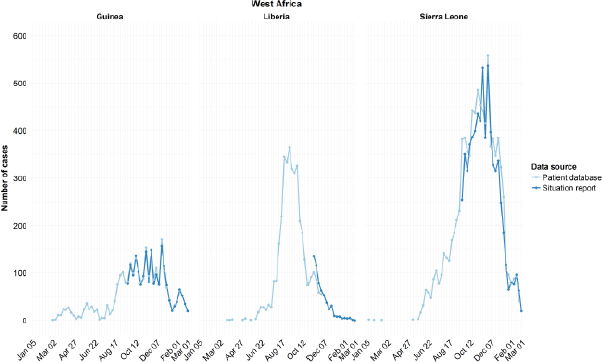
This epidemic comprised many distinct outbreaks, with patterns of cases exhibiting wide variation among the three most affected countries, observed Bausch (see Figure 2-1). “The transmission dynamics are different in each area, and they may be different in each village,” he pointed out. As soon became apparent, he added, a “one-size-fits-all” approach could not be expected to control disease across these diverse contexts. Sadly, it took several precious months for the international community to recognize that the “traditional, core Ebola control package” could not contain Ebola under these circumstances (WHO, 2015a).
On August 8, 2014, the World Health Organization (WHO) declared the epidemic to be a public health emergency of international concern (PHEIC) as defined by the International Health Regulations (IHR);1 however, this did little to mobilize much-needed resources (Gostin and Friedman, 2014). In response to a plea from the Médecins Sans Frontières (MSF) for military support of its operations, U.S. President Barack Obama pledged nearly 3,000 troops to build Ebola treatment centers in Liberia on September 16 (Gostin and Friedman, 2014). By the time the first treatment center opened, the number of cases in Liberia had fallen drastically, in part through efforts by community members to educate others about basic disease control measures (Onishi, 2015).2
“This is a very emotional time for me,” admitted Oyewale Tomori of the Nigerian Academy of Science. West Africa, on the eve of Ebola, was “swimming in this ocean of national apathy, denial, and unpreparedness,” he recalled. That, he
___________________
1 The most recent iteration of the IHR, implemented in June 2007, is a legally binding agreement among its 196 member nation signatories (known as “states parties”) establishing a framework for coordinating and managing threats to international public health. Most participating countries were unable to meet core surveillance and response requirements by 2012 and were granted at least one 2-year extension to this deadline. SOURCE: http://www.who.int/ihr/about/en (accessed November 3, 2016).
2 On May 9, 2015, the WHO announced the official end of the Liberian outbreak. Meanwhile, the epidemic continued to smolder in Sierra Leone and Guinea. By early June, with the onset of the rainy season likely to complicate field operations, the WHO reported that Ebola activity had become more intense and widespread in both countries after reaching a 10-month low in mid-May (WHO, 2015b). As Bausch had predicted at the workshop, in late March, “getting to zero” has proved difficult in these countries, where previously fragile health systems and other public services have collapsed under the strain of the epidemic. Thus, he observed, the task of recovering from Ebola in West Africa may be even more daunting than extinguishing the epidemic.
SOURCE: Bausch presentation, March 24, 2015.
insisted, is why the disease became a humanitarian crisis (Kupferschmidt, 2014), and he proceeded to describe both the causes and the consequences of regional helplessness to avert public health disaster.
Nigeria Stops Ebola
Nigeria was one of several countries that reported Ebola cases during the epidemic, but there, chains of transmission were quickly extinguished (Fasina et al., 2014). Of the 20 Nigerian cases, more than half of them were health care workers; 8 patients died, of whom 5 were health care workers. Several factors contributed to the swift control of the Nigerian outbreak, according to Tomori: the early detection of the index case (a traveler returning from Liberia who developed symptoms during his flight, and who collapsed upon arrival at Lagos airport); the fact that—likely owing to a strike by public hospital workers—he was admitted to a smaller private hospital; and, subsequent intense contact tracing efforts that resulted in the monitoring of nearly 900 people.
Tomori attributed the success of this contact tracing effort to community involvement. “We must have looked at over 8,000 people we had to contact aggressively, but then it was done by the local government people who were living with the people, and therefore when they see them come into their place it is
nothing new to them,” he explained. This illustrates the importance of educating and involving local people to respond to public health crises, he noted.
Elsewhere, Ebola continued to kill and to affront the living, Tomori observed. “Sorrow, pain, and agony are the traveling companions of Ebola,” he said. The disease “pitches the government against the governed,” he continued, describing conflicts between ill people and their families who had blocked the gates of treatment centers and the roads leading to them, and soldiers sent to move them on. The epidemic orphaned many children, destroyed economies, and overwhelmed—then gutted—health infrastructure in the affected countries. When basic health care collapses, he added, “other things come in their place. There’s fear, there’s denial . . . what people call irrational behavior—but you need to understand where they are coming from,” he insisted.
Owning the Problem
Many have focused on the role of human behavior in allowing Ebola to grow to disastrous proportions in West Africa, Tomori observed, but in focusing on the root causes of the epidemic, he placed emphasis on a different set of circumstances, actions, and ideas—all of which contribute to what he characterized as a high dependency in Africa on external aid.
Tomori introduced his argument with three illustrative stories. When Ebola came to Kikwit in 1995, he recounted, an international team responded, and within 4 weeks, cleared the epidemic—then they left, taking with them the skills and capacity to control the next outbreak. The following year, the U.S. Agency for International Development (USAID) and the U.S. Centers for Disease Control and Prevention (CDC) provided aid to West Africa to help control an outbreak of cerebral spinal meningitis (CSM).3 “They also left,” he remarked, “and then we closed the door, and we lost the key.” However, he observed, in 2014 there was a “glimmer of hope” in the response to Ebola outbreaks in the DRC and Nigeria: these outbreaks were extinguished through a combination of existing national
___________________
3 Cerebral spinal meningitis (CSM) is “most often caused by the bacteria Neisseria meningitides, which infects the meninges, the thin covering of the brain and spinal cord. Symptoms of the disease include stiff neck, high fever, rash, headache, vomiting, and confusion. Even with rapid diagnosis, 5–10 percent of patients typically die within 24–48 hours of symptom onset. Although sometimes fatal, CSM is most often treatable with antibiotics administered upon hospital admission. A preventive vaccine is available, and it is routinely recommended for adolescents 11–18 years of age. However, in the African Meningitis Belt, routine immunization is not practical, as the vaccine typically protects for only 3–5 years. Even with large-scale coverage, vaccination efforts in this part of the world would not provide sufficient herd immunity to protect the population at large. Therefore, control measures used during epidemics, in places such as Nigeria, include enhanced epidemiological surveillance, prompt case management, and mass vaccinations in areas involved in an outbreak.” SOURCE: http://www.healthmap.org/site/diseasedaily/article/cerebral-spinal-meningitis-csm-spreads-nigeria-32609 (accessed November 3, 2016).
capacity to control disease transmission, and “appropriate external assistance,” he observed.
By accepting external aid to cope with infectious disease outbreaks and other health crises, Africa has perpetuated a cycle of dependence, Tomori continued. Such support often benefits the giver more than the receiver, he said; for example, pathogen outbreaks offer foreign experts and health agencies opportunities for both scientific and operational research—of which many workshop participants acknowledged having taken advantage. At the worst, these so-called partnerships leave scientists in resource-poor countries permanently dependent on outsiders, “reduced to mere sample collectors,” he said, and unable to use the experience to control the next outbreak on their own.
To reverse this cycle, Tomori declared, “Each African country must commit its resources to ensuring appropriate surveillance for emerging and reemerging pathogens. Meanwhile, developed countries should ease off on their dominance and control of the processes of global disease surveillance. Only then can developing countries truly ‘own’ these processes,” he concluded. The essentials of Ebola control—as previously described by Heymann and others—are well known to Africans, as well as to the rest of the world, he observed; there is no good reason why Africans cannot implement these measures. Worse, he added, Africa has not only become powerless in the face of health crises, “It has become confused, disoriented, and totally helpless, resorting to international assistance, begging for everything including gloves.”
Unless this situation changes, it will not be long until Africa again calls for assistance, Tomori predicted. “Ebola or some other disease will continue to destroy the very foundations of our socioeconomic structure and orderly development, as it is currently doing in Guinea, Liberia, and Sierra Leone,” he warned. Using considerably more colorful language, Tomori expressed that Ebola had knocked Africa down, but it was not defeated. However, he continued, if the present predicts the future, it is grim: replete with scandal, disease, income inequality, corruption, denial, and inevitably, humanitarian disaster.
“Ebola is Africa’s problem,” Tomori concluded. “The solution must come from Brazzaville, not from Geneva, not from Washington. You may assist, but we must own our problem, and we must seek solutions to it,” he explained. “Out of the debris around me, hope springs from each of you that is here for the work you have done,” he said. “There is hope for an African Renaissance.”
NEW PERSPECTIVES
The effort to control Ebola on a vastly larger scale than previously imagined has raised significant questions and afforded valuable insights on the disease and its treatment, as well as its epidemiology. The topics introduced below are explored in detail in subsequent accounts of workshop presentations and discussion.
Clinical Considerations
Although Ebola is classically termed a hemorrhagic fever, the many case descriptions resulting from the West African epidemic suggest that bleeding is not a major component of the disease, Bausch stated. Instead, volume loss and electrolyte imbalance appear to be the most clinically significant characteristics of the disease. Efforts are currently under way to characterize these observations through systematic data collection, he said; the fact that case fatality rates for Ebola patients evacuated to medical centers in wealthy countries were significantly lower than for those treated in Africa offers hope that more intensive medical care can lower case fatality.
Best practices for fluid and electrolyte support, the use of antidiarrheal agents, and antibiotic prophylaxis remain to be resolved, Bausch noted. Additional approaches under study include blood products and immune therapy, convalescent serum, convalescent blood, and antiviral agents. Ongoing clinical research on these treatments is discussed below.
Public Health Challenges
Nosocomial Transmission
While previous Ebola outbreaks in Central Africa revealed the significant risks of nosocomial transmission, the West African epidemic was tragically notable for the number of health care workers who became infected: more than 850, of whom nearly 500 died, accounting for 3.4 percent of all reported cases, according to Bausch.4 In West Africa, the lack of access to personal protective equipment (PPE) or its appropriate use contributed to the high transmission among health care workers as described by Heymann and Sylvain Aldighieri of the Pan American Health Organization.
Community Resistance
Efforts to control Ebola in West Africa have frequently been thwarted by community resistance by a number of means, including the murder of health care workers (WHO, 2015d). Attempts to explain significantly higher levels of community resistance to Ebola control efforts in West Africa, as compared with Central Africa, have focused on traditional belief systems, low population literacy rates, and shortcomings in outbreak response (WHO, 2015d). Perhaps it is even more true in West Africa than in the DRC that, as Muyembe observed, Ebola cannot be conquered without community support. One year into the West African epidemic, the WHO identified community engagement as “the one factor that
___________________
4 As of February 17, 2015, according to the WHO (Bausch presentation, March 24, 2015).
underlies the success of all other control measures . . . the linchpin for successful control” (WHO, 2015d). The WHO report goes on to note:
Contact tracing, early reporting of symptoms, adherence to recommended protective measures, and safe burials are critically dependent on a cooperative community. Having sufficient facilities and staff in place is not enough. In several areas, communities continued to hide patients in homes and bury bodies secretly even when sufficient treatment beds and burial teams were available. Experience also showed that quarantines will be violated or dissolve into violence if affected communities are given no incentives to comply.
Impact on Other Infectious Diseases
The West African epidemic interrupted important ongoing public health activities, including immunization initiatives against several vaccine-preventable diseases (ECDC, 2015a; Edelstein et al., 2015). Catch-up vaccination campaigns can be organized against such diseases as measles and polio, but they will not be effective unless public trust in health care systems is restored in these vulnerable countries.
Conversely, Heymann pointed out, if hospital infection control is improved overall in response to the threat of Ebola, outbreaks of other infectious diseases are less likely. “While we are looking at complex interventions—drugs, medicines, vaccines, all these different interventions—we’re again neglecting the basics, which are good infection control in hospitals; we’ve seen it stop outbreaks in the past, and it will stop them in the future,” he argued.
In the future, support for ongoing infectious disease control measures should be incorporated into planning for public health crises such as emerging disease outbreaks, suggested Jeffrey Duchin of the Seattle and King County Department of Public Health. Bausch noted that such crises could even be viewed as opportunities to expand capacity for the detection of infectious diseases. Dozens of West African laboratories have responded to Ebola, he reported; they could also contribute to improved diagnosis of infections such as malaria and typhoid fever.
Care for Survivors
Increased attention has also been directed at survivors of Ebola. Again, systematic data collection and analysis are needed to better understand the sequelae of the infection and how to address them. At the time of the workshop, Ebola virus has been shown to persist in the semen of survivors for at least 3 months, Bausch noted, so it is important to explore the potential for its ongoing sexual transmission.5 Bausch continued:
___________________
5 Recent virus persistence studies have demonstrated that Ebola virus can be identified in semen 290 days after symptom onset (Uyeki et al., 2016).
We have never actually had a well-documented case of sexual transmission of Ebola, but now we are getting some anecdotal reports of it, and that’s a big issue, even if we think this happens relatively rarely. If you have 12,000 survivors and half of them are men, what do you do? When can you say an outbreak is over? Even if we haven’t had transmission lately, if we still have 6,000 people out there who might still be carrying virus [they] could reintroduce it simply by having unprotected sex with their partners.
The WHO recommends that all survivors of Ebola abstain from all sex acts for at least 3 months after symptom onset, with condom use as a second-best option (WHO, 2015e).6
Ebola virus may also persist in the chambers of the eye, Bausch reported; preliminary data reveals that vision problems are common in survivors of infection. Many also suffer from general arthralgia. People do not necessarily “bounce back” from Ebola, he observed.
Mental health issues are significant for survivors of Ebola, Bausch added. These include dealing with the stigma of having had the disease, as well as posttraumatic stress disorder and depression, which affect both survivors and uninfected people who lost loved ones to Ebola. He noted that the WHO’s Ebola Survivor Network was established to provide resources to care for survivors’ many needs, and, secondarily, to collect data that will inform research and future care.
Potential Directions for Research
Animal Reservoir(s)
While nonhuman primates are susceptible to Ebola and have been confirmed as sources of human infections, the role of bats in Ebola virus transmission—although suspected since the 1976 Sudan outbreak—has yet to be resolved, Heymann noted. The apparently increasing frequency of Ebola outbreaks probably results from more sensitive outbreak detection, Bausch stated.
Clinical Research
As the West African epidemic unfolded, Heymann composed a “wish list” of research questions to be addressed in order to improve future clinical care for Ebola in such areas as maintaining electrolyte balance; the use of convalescent plasma for prophylaxis, as well as for treatment; point-of-care diagnostic testing; antiviral drug treatment; and the use of vaccines for outbreak control and disease prevention in health care workers.
___________________
6 Updated WHO guidelines are available at http://www.who.int/reproductivehealth/topics/rtis/ebola-virus-semen/en (accessed November 3, 2016).
Heymann focused on studies of the feasibility and effectiveness of collecting and using convalescent plasma and serum that currently are being undertaken based on clinical trials conducted during the West African epidemic. “This has been a long time in coming, mainly because it has been felt that it could not be done safely in African countries, [and] that the equipment could not be brought in,” he said. However, he noted, plasma was collected from volunteer survivors in the months after the 1976 Zaire outbreak and stored as a source of convalescent plasma for future outbreaks. It was no longer viable in 1995, when the next outbreak occurred in Kikwit, but previously it had been used in the United Kingdom to treat an infected laboratory worker, who also received interferon and fluid therapy, and who recovered from Ebola. A few Kikwit patients were instead transfused with convalescent blood; all but one survived the disease (Mupapa et al., 1999). If recent analyses demonstrate the effectiveness of convalescent blood interventions, Heymann said, they could be highly sustainable and maintained by African scientists.
Looking Backward, Moving Forward
Days after the 1-year anniversary of the announcement of the West African Ebola epidemic, and with its end plausibly in sight, Keiji Fukuda of the WHO observed that the global public health community had entered into “a period of very intense assessment and questioning.” Now is the time to understand this event in detail, determine what can be done to prevent its recurrence, and consider how to achieve that goal, he stated.
Role of the IHR
Stepping back from the previously discussed factors that contributed to the unprecedented geographic spread, scale, and burden of Ebola in West Africa, both Fukuda and Heymann emphasized the importance of implementing sound global strategy to address threats to international health, as set forth in the IHR. The aims of the IHR are twofold, Fukuda explained: to strengthen each country’s preparedness and capacity to quickly address public health threats, and to provide a legal framework for the coordination of regional and global responses to incidents of international concern. This provides a broad structure for applying effective public health measures, for collecting and analyzing scientific evidence, and for ensuring equal access and treatment for all people and countries.
Responses to global health threats involve a spectrum of entities, including countries, subnational units, communities, the WHO, and other nongovernmental organizations. Among these, countries are central, because sovereignty resides and responsibilities accrue at the national level, Fukuda observed. “In many ways countries remain the first-line responders, and in the ideal situation, they are the main responders,” he said.
Over the past 5 years, the IHR has largely succeeded in preventing and containing threats to public health, including pandemic infections, Fukuda stated. However, he added, significant challenges to implementing the IHR have also arisen: capacity building has proven more difficult than anticipated, even for wealthy countries; lack of enforcement powers compromises compliance; information sharing and equity of access to crucial technology and training remain far from complete; and the global community has faltered when facing complex situations requiring a scaled-up response, as exemplified in West Africa.
“Despite a level of response, which I think was quite vigorous and really in most instances would be enough to address even a complicated Ebola outbreak, the net result is that we ended up in a situation where we had a very large event,” Fukuda recalled—one that required the coordination of resources beyond the affected nations and region, including those of the United Nations and other organizations and countries. “Moving at a real-time speed to do these things when you have very complex situations is difficult,” he observed. To avoid similar delays in addressing future disease threats, he proposed that standing outbreak response teams and funds for their swift deployment be created, but only as an emergency measure to support the primary goal of strengthening each country’s ability to contain infectious outbreaks.
The overall strategy expressed in the IHR is sound, Fukuda insisted. The WHO and its organizational partners can facilitate access to funding, expertise, technology, materials, and other necessities for dealing with complex outbreaks, he said. “But it is equally clear that only countries can bring law, sovereignty, legitimacy, and then long-term sustainability to responding to these kinds of events, and this is critical to understand.”
Filling Information Gaps
Workshop participants discussed the consequences of gaps in surveillance and diagnostics, as well as in information sharing through interprocess communication, hospital information systems, and laboratory information systems, during the West African epidemic. They then considered potential strategies to remedy these deficiencies. Jennifer Gardy of the University of British Columbia asked whether such nontraditional citizen-based or digital surveillance methods might be useful where national borders are porous, as in the communities where the West African epidemic began. Fukuda agreed that such approaches are worthy of investigation, especially since they make disease surveillance a community activity (although, he added, formal health departments would still need to oversee such investigations and analyze their results). Heymann noted that better point-of-care “dipstick” diagnostics would further enable disease detection at the community level.
Many innovative, community-level surveillance strategies are currently being touted in response to the Ebola epidemic, noted Julie Pavlin of the Armed
Forces Health Surveillance Center. Even if some might permit the early detection of Ebola, she asked, are they sustainable in places where outbreaks are likely to originate?
All of the affected countries were recently torn by conflict, Muyembe reminded participants; their health systems are in shambles, and the only funding they have received is earmarked to combat Ebola. Once the disease is controlled, he warned, “I think they will come back to the old situation: I mean no water, no disinfectant, no PPE.” That is what happened after the Kikwit outbreak, he recalled. “During the outbreak we had everything: we had PPE, we had gloves,” he said. “After the outbreaks, no PPE, no gloves in the hospital. No water, no electricity. So it is a big problem.”
Mark Kortepeter from the Uniformed Services University of the Health Sciences noted the importance of keeping and sharing medical records to improving clinical care, as well as the dearth of such records from previous Ebola outbreaks. Are electronic medical records being kept in West Africa, he asked?
“There have been various measures addressed to try to move that forward, and it’s varied, depending upon the particular ward and what they have available to them,” Bausch answered. For example, the data could be directly entered into electronic tablets in the wards (although this process might prove unwieldy to medical staff wearing PPE and working under pressure). In some cases, medical charts have been photographed with cell phones and those images distributed, he said; in others, “Someone inside the ward is right next to the fence with someone outside the ward, and just tells them what is going on with the patient, and they can record that.” Overall, medical records were kept in appreciable numbers, establishing a trend that should continue, he concluded. “I think it’s coming, and of course even though it seems like a relatively trivial thing, it’s hugely important in order to get some sort of systematic look at this disease and what works and what doesn’t,” he observed.
In addition to medical records, Heymann noted that considerable genetic data on the Ebola virus was collected during the West African epidemic. Unfortunately, he added, “It has not been shared widely in public databases or in databases where there can be an attempt to see whether or not it is possible, as it is in polio, to determine origins of cases and various issues.” It will take a major effort to ensure open sharing of data from Ebola outbreak investigations, he observed; however, as was demonstrated during the severe acute respiratory syndrome (SARS) outbreak, such data sharing is both feasible and key to infection control.
Toward Appropriate Assistance
In the discussion that followed Tomori’s presentation, several participants responded to his call for African control of its public health response to crises, and for African leadership in establishing basic public health capacity for its citizens. Referring to “one of those forgotten reports” on sustaining global
surveillance for emerging infectious diseases (IOM and NRC, 2009), Gerald Keusch of Boston University noted a key conclusion: donor support for such efforts does not endure. That committee recommended that developing countries develop sustainable support for disease surveillance and outbreak preparedness by taxing “something that’s a repeated activity.” Tomori responded that Africans would first need to be convinced of the value of developing such public health capacity—but “once that realization comes, we’ll be able to find money for it,” he said. “There will be issues we’ll regard as important, and we’ll put tax on things, like tobacco,” he explained. “I think it’s something we need to push for many different countries, but first we need to convince them that they need surveillance.”
Peter Daszak of the EcoHealth Alliance focused on prospects for greater African control over aid allocation. Recognizing that disease emergence is precipitated by rapid development, which frequently encompasses increased agricultural production, deforestation, and land use change, he suggested that when developing countries accept aid for development, a portion of that aid could be set aside to address the public health consequences of environmental destruction. “When we build a road system, shouldn’t we also build a clinic?” he asked.
Africans themselves need to consider the outcomes associated with development, so when aid is offered, they can define the terms of use, Tomori replied. At the moment, these terms are defined by governments that do not talk to scientists, he lamented. Educated people in the African region should contribute their technical expertise to shape these decisions that highly impact the life of their people, Tomori insisted.
This situation is not being addressed by organizations such as MSF, which tend to invest in infrastructure, rather than in training local people, Joseph Fair of the Fondation Mérieux USA observed, and that does not bode well for the next generation of public health leaders in Africa. They will need more than graduate and medical degrees, he added, they must also learn how to obtain funding, and how to be managers. Also, when agencies do invest in infrastructure, they need simultaneously to invest in local expertise. For example, he asked, “Do we have a Sierra Leonean facilities engineer to manage a new Sierra Leonean lab?”
Fair also emphasized that the design of such a facility should enable its ongoing operation. “It may not be so expensive for us to pay for an 80 watt generator to power a lab, but once we leave here can anyone pay that off a Ministry of Health budget? No, they cannot,” he remarked. Instead, he insisted, facilities should be “appropriate and scalable” to their individual locations.
The Next One
The IHR specify 13 core public health capacities for addressing public health threats, of which 9 directly apply to Ebola preparedness and response, noted Sylvain Aldighieri. However, the West African Ebola epidemic is but one of
several recent emergencies that highlighted gaps in global public health strategy, as recently noted by WHO (2015c):
Gaps in capacity and international support were and continue to be evident in the responses to all manner of emergencies, from the conflict in Syria, to the drought in the Horn of Africa, to the typhoon in the Philippines, and in outbreaks including those of SARS, Middle East respiratory syndrome coronavirus (MERS-CoV), influenza A subtype H1N1, and, most recently, Ebola virus in West Africa. In each case, the response lacked the speed, coordination, clear lines of decision making and dedicated funding needed to optimize implementation, reduce suffering, and save lives. It is critical that countries, and the intergovernmental agencies that support them, identify and internalize the lessons learned from these responses and, in moving forward, adopt an all hazards risk management and response approach to emergencies.
“Today’s outbreak of Ebola is not necessarily the big battle that we will fight tomorrow,” Fukuda emphasized. While most infectious disease outbreaks are less disruptive than Ebola in West Africa (which he characterized as a regional event with profound global implications), it is hardly a worst-case scenario. That position is reserved for pandemic influenza, he argued, because it could directly impact a large number of countries, including those that typically confront global health emergencies. The world must prepare to address the full spectrum of epidemics, from the many less severe than Ebola to the few that could be far more devastating, he concluded.
To meet this challenge, a balance must be struck between “just-in-time” responses to acute, precipitous outbreaks, and investments in basic public health infrastructure to support disease prevention, detection, and control wherever an outbreak might occur—and which are shockingly lacking throughout much of the developing world, as several participants observed. Crises precipitate funding, but what can be done to increase support for basic public health capacity to mitigate outbreaks before they become crises?
This challenge was also explained by Heymann who described three possible scenarios for dealing with infectious outbreaks, Heymann noted. The first, rapid detection and response, is expensive, but attractive to funders. The second, attempting to predict outbreaks before they happen, is not yet technically possible. The third approach, primary prevention—and hospital infection control in particular—is the best choice for Ebola, he argued, because wildlife-to-human emergence cannot yet be effectively prevented. If an Ebola vaccine is developed, it may be effective to vaccinate health workers in the “Ebola belt” to prevent them from transmitting the disease. “I think we have a long way to go to shift that paradigm to primary prevention,” he acknowledged, “but it’s feasible and it’s possible.”
Such long-term, multifactorial investments defy conventional thinking about funding, Bausch said. His suggestion: focus on labor. “I think we need more Jean Jacques Muyembes in West Africa,” he argued. There is a need for smart people
who know what to do with the resources available. If there is no investment in training the workforce, then the response will be just in time or late and will mostly come from outside the country, he added.
Investments in public health, like investments in the stock market, are motivated by self-interest, Fukuda observed. Recognizing that, the case must be made that “what happens in Africa affects your neighborhood, either directly or indirectly,” he insisted. The IHR are founded on that principle, he said, but the world beyond public health remains largely unconvinced of it.
“I am terrified that Ebola is going to evaporate from our collective memories,” Fukuda continued. “We have seen this over and over again. It doesn’t matter how bad something stings us. If it stings us for a period of time and it goes away, then we easily forget about it,” he observed. This, he continued, is why people view cancer differently than infectious diseases: while on the one hand, nearly every person has been touched by cancer, Ebola is to most of the world an event “that happened over there.”
To really make strides against diseases like Ebola, we have to convince people that there is no “over there” for infectious pathogens in a globalized world, Fukuda stated. That Ebola left Africa for the first time provided some traction for that idea, but it remains largely abstract—and so, he predicted, “We are going to be banging our heads for a while.”


















