Summary
Since 1938 and 1941, nutrient intake recommendations have been issued to the public in Canada and the United States, respectively. Currently defined as the Dietary Reference Intakes (DRIs),1 these values are a set of standards established by consensus committees (DRI committees) under the National Academies of Sciences, Engineering, and Medicine (the National Academies) and used for planning and assessing diets of apparently healthy individuals and groups. The Dietary Guidelines for Americans and the Canadian Food Guide, for example, draw partly on the DRI reports. Other important uses are establishing reference points for monitoring dietary intake of populations, evaluating government food assistance
___________________
1 Dietary Reference Intakes are a group of six different standards: (1) Estimated Average Requirement (EAR): The average daily intake of a nutrient that is expected to meet the requirement of half of the healthy individuals in a group defined by sex and life-stage. (2) Recommended Dietary Allowance (RDA): The average daily intake level that is sufficient to meet the nutrient requirements of 97 to 98 percent of healthy individuals in a specified sex and life-stage group. (3) Adequate Intake (AI): When available evidence is not sufficient to determine the EAR for a nutrient, an AI is set. The AI is the average daily nutrient intake observed in apparently healthy individuals in a specified sex and life-stage group. (4) Tolerable Upper Intake Level (UL): The highest average daily nutrient intake level that is likely to pose no risk of adverse effects to nearly all healthy individuals in the specified sex and life-stage sex group. (5) Acceptable Macronutrient Distribution Range (AMDR): A range of usual intakes for a macronutrient that is associated with reduced risk of chronic disease while providing adequate intakes of essential nutrients. An AMDR is expressed as a percentage of total energy intake. (6) Estimated Energy Requirement (EER): A calculated level of energy intake that is estimated to maintain energy balance (or as appropriate, normal growth), that incorporates weight, height, physiological state (i.e., pregnancy), and level of energy expenditure.
programs, planning diets for military personnel, and guiding nationwide health programs.
Traditionally, for each nutrient deemed essential for normal physiological functioning (e.g., vitamins, minerals, protein), the scientific literature is reviewed to determine the most appropriate indicator of adequacy (e.g., calcium balance for adequacy of calcium and vitamin D) and toxicity (e.g., liver damage for excessive copper intake) with the objective to establish standards for 22 groups defined by life-stage and sex. In some cases, associations of nutrients with indicators of chronic diseases have been used to establish DRIs where indicators of adequacy or toxicity were lacking or where the chronic disease considerations were critical. At present, a more general need to explore whether specific levels of nutrients or other food substances (NOFSs) can ameliorate the risk of chronic disease is recognized, given changes in diets and the availability of a larger body of evidence about potential roles of NOFSs in causal pathways leading to chronic diseases. Before chronic disease DRIs are determined, however, various conceptual and methodological differences with traditional DRIs should be considered. A major difference reflects the nature of the goal: while traditional DRIs are required to guide efforts to ensure that populations meet essential nutritional needs for normal physiological functioning, chronic disease DRIs are desirable but not essential. Other differences relate to the slow progression of a chronic disease leading to long latency of any effects, as well as the multi-factorial etiology of a chronic disease, the types of data available, and the analytical methodologies required to evaluate such data. Stakeholders have reflected on approaches for how to address chronic disease DRIs but no agreement yet exists on a process that can be consistently applied. A recent effort to identify and articulate challenges associated with evaluating NOFS-chronic disease relationships occurred under the leadership of a working group sponsored by the Canadian and U.S. governments, resulting in the publication Options for Basing Dietary Reference Intakes (DRIs) on Chronic Disease Endpoints: Report from a Joint US-/Canadian-Sponsored Working Group (i.e., the Options Report) (Yetley et al., 2017, Appendix B). The Options Report is the primary reference resource for this consensus study, which aims to develop guiding principles and recommendations to develop chronic disease DRIs.
THE TASK AND APPROACH
The statement of task (see Chapter 1, Box 1-1) directs an ad hoc committee of the National Academies to assess the options (see Table S-1) presented in the Options Report and to determine guiding principles for including chronic disease endpoints for NOFSs that will be used by future National Academies committees to develop DRIs. In the Options Report,
TABLE S-1 Options for Addressing Questions Related to Establishing Chronic Disease (from the Options Report in Appendix B)
| Question/Issue | Options |
|---|---|
| How should chronic disease outcomes be selected? | Option 1: Endpoint (outcome) is the incidence of a chronic disease or a qualified surrogate disease marker. |
| Option 2: Endpoint (outcome) may include nonqualified disease markers. | |
| What are acceptable levels of confidence that the relation is causal? | Option 1: Require a high level of confidence. This level of confidence likely requires at least some evidence from high-quality randomized controlled trials (RCTs) in which the measured outcome is a chronic disease event or qualified surrogate disease marker. |
| Option 2: Use level B evidence. This level of evidence suggests a moderate degree of confidence that the relation of interest is causal, but new findings could change the DRI decision. | |
| Option 3: Use actual level of certainty. | |
| Option 4: Make decisions on a case-by-case basis. | |
| What is the approach to selecting indicators and specifying intake-response relations? | Option 1: Choose a single outcome indicator on the causal pathway. |
| Option 2: Use multiple indicators of a chronic disease. | |
| Option 3: Use of multiple indicators for multiple diseases. This option may be necessary when a single food substance has different intake-response relations with multiple chronic diseases. In this situation, the DRI committee might need to develop criteria for selecting appropriate disease indicators to establish multiple intake-response relations, methods to integrate multiple endpoints, and approaches to account for the inevitable inter-individual variability in the relations of interest. | |
| When should intake-response data be extrapolated? | Option 1: Establish reference intake values only for similar populations. |
| Option 2: Allow extrapolation when sufficient evidence is available. |
| Question/Issue | Options |
|---|---|
| What should be the different types of DRIs associated with benefit? | Option 1: Establish chronic disease risk-reduction intake values, by modifying the traditional EAR/RDA approach to estimate the mean intakes of individuals and the interindividual variability associated with specified disease risk reductions. This option is conceptually very similar to the traditional EAR/RDA approach but uses relative risks and requires knowledge of baseline disease prevalence. |
| Option 2: Identify ranges of beneficial intakes. | |
| What should be the different types of DRIs associated with reduction in chronic disease risk? | Option 1: Base ULs on the traditional threshold model when UL values based on chronic disease endpoints are higher than those based on traditional adverse effects. |
| Option 2: Base ULCD on intakes associated with chronic disease risk. | |
| What are acceptable levels of confidence in the intake-response data? | Option 1: Require a high confidence level by, for example, using RCTs with a chronic disease event or a qualified surrogate disease marker as the outcome measure. |
| Option 2: Accept a moderate confidence level. | |
| Option 3: Piecemeal approach. This option pieces together different relations in which the biomarker of interest is a common factor between the food substance and a chronic disease. | |
| What approaches can be taken to make decisions when benefits and harms overlap? | Option 1: Avoid overlap between beneficial intakes and intakes associated with adverse events. |
| Option 2: Establish criteria related to severity and risk of chronic disease. | |
| Option 3: Describe the nature of the evidence. | |
| What should be the organizational process to set all DRIs? | Option 1: Continue to use a single DRI development process. |
| Option 2: Create 2 separate processes for developing DRIs. | |
| What should be the starting point of chronic disease DRIs? | Option 1: Establish DRIs for individual or small groups of interrelated food substances |
| Option 2: Establish DRIs for multiple food substances on the basis of a chronic disease endpoint. |
SOURCE: Yetley et al., 2017.
key scientific challenges to develop chronic disease DRIs were identified, related to three questions: (1) What are acceptable levels of confidence that the relationship between an NOFS and a chronic disease is causal?, (2) If a causal relationship exists, what are acceptable levels of confidence in the intake-response relationship data and what are the approaches for identifying and characterizing the intake-response relationship and, if appropriate, to recommend DRIs?, and (3) What should be the organizational process for recommending chronic disease DRIs? Although DRI committees will be sensitive to the uses of the DRIs in nutrition policies, this committee recognized that recommendations for tasks that relate to risk management and policy (see Figure S-1), such as formulating the statement of task for a future DRI committee, are outside the scope of this study. In making its recommendations, the committee assumed that the work of future DRI committees will be done within the context of the current DRI process (see Figure S-1), in which a DRI committee will receive a thorough and rigorously implemented systematic review,2 will review the totality of the evidence about questions 1 and 2 above, and will recommend chronic disease DRIs, if appropriate. The committee also assumes that a mechanism will be established for communication between those conducting the systematic review and the DRI committee, which will ensure a moderate level of communication while also protecting against inappropriate influence on the systematic review methods.
Broadly, a DRI committee’s task is to review the evidence and recommend all DRIs for specific nutrients. With respect to chronic disease DRIs, the subject of this study, if enough evidence exists with respect to both the causality and the intake-response relationships questions, the committee’s decisions will include developing chronic disease DRIs along with the adequacy and toxicity DRIs. Making such decisions entails many challenges. Some challenges are universal—measuring the chronic disease clinical outcome directly, or through surrogate markers3 for clinical outcomes, or both, and extrapolating to populations other than those directly studied—and other challenges are unique to nutrition research. An important
___________________
3 A surrogate marker is a type of biomarker that “predicts clinical benefit (or harm or lack of benefit or harm) based on epidemiologic, therapeutic, pathophysiologic, or other scientific evidence. A surrogate disease marker is qualified for its intended purposes” (Yetley et al., 2017, p. 263S).
2 A systematic review “is a scientific investigation that focuses on a specific question and that uses explicit, planned scientific methods to identify, select, assess, and summarize the findings of similar but separate studies. It may or may not include a quantitative synthesis (meta-analysis) of the results from separate studies” (IOM, 2011b, p. 21). A systematic review is typically conducted by a group of experts in the process itself that includes subject matter experts (e.g., a systematic review team) and in consultation with specific subject matter experts (e.g., a technical expert panel).
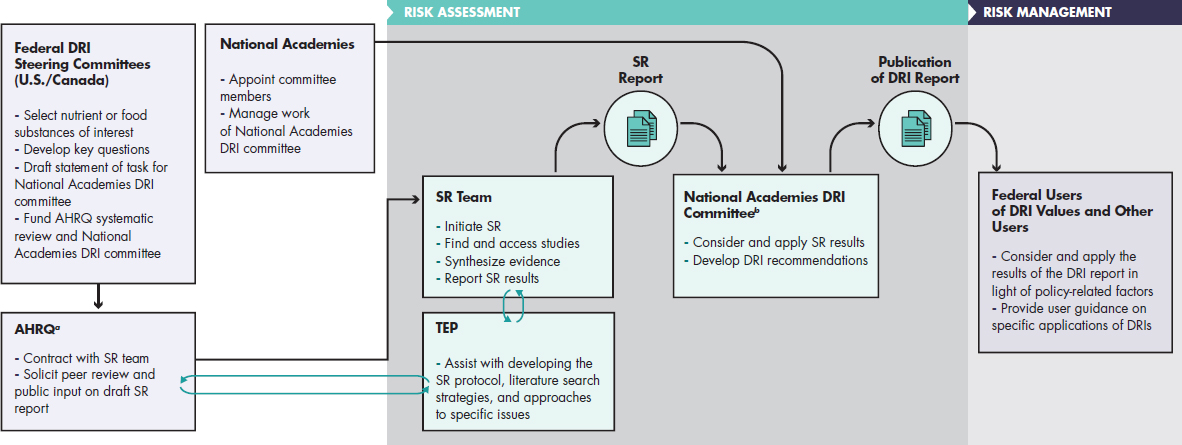
NOTES: AHRQ = Agency for Healthcare Research and Quality; DRI = Dietary Reference Intake; SR = systematic review; TEP = Technical Expert Panel. a The Agency for Healthcare Research and Quality is listed here as the agency that, in the current DRI process, has been responsible for the systematic review aspects of DRI development. b National Academies DRI committees are convened by and positioned with the Food and Nutrition Board of the National Academies of Sciences, Engineering, and Medicine and operate under the National Academies study committee guidelines, which include an external peer review process of the draft DRI report.
SOURCE: Adapted from IOM, 2011a,b; Taylor, 2008.
feature of nutrition studies that explore associations between NOFSs and chronic diseases is their frequent reliance on prospective cohorts, rather than randomized controlled trials (RCTs), as study designs. Reasons for the predominance of observational data include the long-term exposure usually needed to observe an association with the chronic disease, the advantage of following study participants under their usual diet intakes and other living conditions, the ability to observe associations with varying levels of intake of the NOFS of interest, and the considerable cost difference compared to conducting a large RCT. On the other hand, making firm inferences about causal relationships is challenging when observational studies predominate. Two main reasons are the inaccuracy of current methods to measure intake of the NOFS and the potential effect of other factors that, if not accounted for, can lead to erroneous conclusions (e.g., age or other characteristics of the individual, other nutrients in the diet, exposure to other chronic disease factors). Recognizing that causal relationships are inferred mainly from RCTs, observational studies are still critical to inform conclusions about causal relationships and to support evidence on the intake-response relationships.
The committee’s recommendations in the following sections consider the options offered in Table S-1, which addresses the pertinent conceptual and methodological challenges, and are organized based on the three questions above. Two of the challenges, evaluating the dietary intake measure and identifying health outcomes, are central to developing chronic disease DRIs and therefore precede the rest of the recommendations. In addition to providing recommendations, the committee developed guiding principles to support DRI committees as they make decisions about chronic disease DRIs. In implementing the recommendations, communication with users and development of guidance for appropriate application of the recommendations are essential.
MEASURING DIETARY INTAKE AND SELECTING CHRONIC DISEASE OUTCOMES
How Should Dietary Intake Measures Be Evaluated?
The nature and quality of nutrient intake ascertainment are diverse. Self-reported measures may be essential for some purposes but intrinsically suffer from both random errors and systematic biases. Biomarkers of intake, which are more objective, can replace self-report for some purposes but have only been developed for a few nutrients. Currently, no one single approach accurately measures dietary intake in a comprehensive manner for all nutrients. Therefore, each study methodology needs to be assessed on the basis of its own merits.
Recommendation 1. Until better intake assessment methodologies are developed and applied widely, Dietary Reference Intake committees should strive to ensure that random and systematic errors and biases of nutrient or other food substance (NOFS) exposure assessment methodologies are considered in their evidence review. In the long term, research agendas should include accelerated efforts to improve NOFS exposure assessments for application in studies of chronic disease risk.
How Should Chronic Disease Outcomes Be Selected?
Studies exploring relationships between NOFSs and chronic disease outcomes vary in the nature and quality of health outcome measurement. High-quality measures of chronic disease outcomes are ideal when developing chronic disease DRIs. However, outcome measures may be flawed and sufficient data on associations between NOFSs and these outcomes do not always exist. The committee supports a variant of option 1 (see Table S-1), where studies that measure qualified surrogate markers—following the criteria adopted by the committee4—are considered in evaluating the evidence about causal relationships. The committee does not support option 2, using nonqualified intermediate markers, because they could lead to serious misinterpretation of DRIs by users.
Recommendation 2. The ideal outcome used to establish chronic disease Dietary Reference Intakes should be the chronic disease of interest, as defined by accepted diagnostic criteria, including composite endpoints, when applicable. Surrogate markers could be considered with the goal of using the findings as supporting information of results based on the chronic disease of interest. To be considered, surrogate markers should meet the qualification criteria for their purpose. Qualification of surrogate markers must be specific to each nutrient or other food substance, although some surrogates will be applicable to more than one causal pathway.
___________________
4 Qualification criteria for a surrogate marker are (1) analytical validation exists, (2) surrogate marker is on causal pathway in disease pathogenesis, (3) surrogate marker is significantly associated with the disease in target population, (4) surrogate marker consistently changes with health outcome in response to a nutritional intervention, (5) change in the surrogate marker explains a substantial proportion of the change in the disease response to a nutritional intervention, and (6) context of use is defined (Calder et al., 2017; Clarke, 2017; IOM, 2010).
EVALUATING ACCEPTABLE LEVELS OF CONFIDENCE THAT THE RELATION IS CAUSAL
What Are Acceptable Levels of Confidence That the Relation Is Causal?
Well-established systems for judging evidence for causal relationships related to a variety of exposures and health outcomes are now available. The committee considered the four options in Table S-1 as well as other evidence review systems related to selecting the levels of confidence about the causality of an association, including the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system. GRADE is an approach to rating the certainty of a body of evidence (i.e., high, moderate, low and/or very low) by using five domains (risk of bias,5 imprecision,6 inconsistency,7 indirectness,8 and publication bias9). Although all the systems reviewed involve similar concepts, GRADE is recommended as the basis for DRI committee judgments about causal relationships between NOFS exposures and chronic diseases for the following reasons: (1) it meets the criteria for an appropriate evidence review tool (e.g., sufficient experience; well-structured protocols, clarity, and transparency; sufficiently detailed; ability to address questions about causal relationships; methods applicable over time), (2) it uses appropriate taxonomy for rating the evidence, and (3) it is in wide use for a variety of health matters. The committee recognizes that any discipline (e.g., public health, environmental health) faces specific challenges that need to be considered when embarking on an evidence review evaluation.
Recommendation 3. The committee recommends that Dietary Reference Intake (DRI) committees use Grading of Recommendations Assessment, Development and Evaluation (GRADE) in assessing the certainty of the evidence related to the causal association between nutrient or other food substances and chronic diseases. Using GRADE, the committee recommends that a decision to pro-
___________________
5 Risk of bias is systematic error due to limitations in the study design or execution (Schunemann et al., 2013).
6 Imprecision is random error that occurs when studies have a small sample size and the number of events is also small; resulting in a wide 95 percent confidence interval around the estimate of the effect (Schunemann et al., 2013).
7 Inconsistency refers to unexplained heterogeneity or variability of study results (Schunemann et al., 2013).
8 Indirectness occurs when a study does not compare the interventions of interest, apply the intervention to the population of interest, or measure the outcomes that are important to patients (Schunemann et al., 2013).
9 Publication bias is a systematic under-estimation or over-estimation of the underlying beneficial or harmful effect due to the selective publication of studies (Schunemann et al., 2013).
ceed with development of chronic disease DRIs be based on at least moderate certainty that a causal relationship exists and on the existence of an intake-response relationship.
APPROACHES TO IDENTIFY AND CHARACTERIZE THE QUANTITATIVE RELATIONSHIP AND DEVELOP DIETARY REFERENCE INTAKES
What Is the Approach to Selecting Indicators and Specifying Intake-Response Relations?
As recommended above, DRI committees should consider only direct measures of disease or surrogate markers deemed appropriate as highly predictive of disease. Characterizing intake-response relationships includes many methodological challenges, including the selection of appropriate models and consideration of confounding factors and interactions. Setting DRIs based on “multiple indicators of a chronic disease” and “multiple indicators for multiple diseases” would require development of multivariate, multi-pathway, intake-response models and, therefore, the committee does not generally recommend those options. The simplest approach of choosing “a single outcome indicator” (option 1) is supported.
Recommendation 4. The committee recommends the use of a single outcome indicator on the causal pathway. However, when a single food substance reduces the risk of more than one chronic disease, reference values could be developed for each chronic disease. The committee, however, does not recommend the use of “multiple indicators of a chronic disease” or “multiple indicators for multiple diseases” unless there is sufficient experience with the use of algorithms or other strong evidence suggesting that multiple indicators point to risk of a chronic disease, due to potential lack of reliability or consistency in the results.
When Should Intake-Response Data Be Extrapolated?
The many factors that influence chronic disease risk in different populations are not well characterized quantitatively, and the likelihood of error is significant. Therefore, the committee supports option 1 in Table S-1. The evidence supporting any departure from this approach should be fully described and should reveal minimal uncertainty.
Recommendation 5. The committee recommends extrapolation of intake-response data for chronic disease Dietary Reference Intakes
only to populations that are similar to studied populations in the underlying factors related to the chronic disease of interest.
What Should Be the Different Types of DRIs Associated with Benefit?
Once intake-response relationships have been identified with acceptable levels of confidence and minimal bias, the challenge is characterizing the relevant intake-response relationships—in terms of their shape and the range of intakes. Several issues complicate translation of intake-response relationships into a DRI, such as the continuous nature of the relationship between nutrients and chronic disease, the multifactorial nature of chronic disease risk, and the diversity of individual baseline risk. Based on this, the committee recommends option 2, which is to recommend DRI ranges at an intake associated with a specified degree of risk reduction or specified benefit but not as single risk-reduction intake value (option 1). The committee also does not recommend developing a family of DRIs for different risk reduction targets for the same chronic disease due to potential difficulties in communicating the uncertainties.
Recommendation 6. The committee recommends that Dietary Reference Intakes (DRIs) for chronic disease risk take the form of a range, rather than a single number. Intake-response relationships should be defined as different ranges of the intake-response relationship where risk is at minimum, is decreasing, and/or is increasing (i.e., slope = 0, negative, or positive). When a nutrient or other food substance reduces the risk of more than one chronic disease, DRIs could be developed for each chronic disease, even if the confidence levels for each chronic disease are different. The magnitude of risk slope considered necessary to set a DRI should be decided based on clearly articulated public health goals, such as those previously identified by other authorities (e.g., Healthy People 2020). The committee does not recommend, however, developing a family of DRIs for any one NOFS for different risk reduction targets for the same chronic disease.
What Should Be the Different Types of DRIs Associated with Reduction in Chronic Disease Risk?
The committee supports a variant of options 1 and 2 and notes that the traditional Tolerable Upper Intake Level (UL)10 based on toxicity should
___________________
10 The highest level of daily nutrient intake that is likely to pose no risk of adverse health effects to almost all individuals in the general population.
be retained whether a putative chronic disease DRI is above (option 1) or below the traditional UL value for that substance. If the increase in risk occurs only at intakes greater than the traditional UL, then no chronic disease DRI would be required, because avoiding intakes greater than the UL will also avoid increases in chronic disease risk. In cases in which increased intake is associated with increased chronic disease risks at intakes less than the traditional UL, both the traditional UL and a chronic disease DRI could be retained (a variation of option 2). In this case, the intake-response relationship should be characterized as to whether the range over which increased risk occurs overlaps with the traditional UL (option 2). The rationale for retaining both would be that their meanings are both different and valuable: the UL connotes an intake limit that should not be exceeded and the DRI for chronic disease would imply that chronic disease risks will be increased with increasing and long-term exposure over the DRI range.
Recommendation 7. The committee recommends retaining Tolerable Upper Intake Levels (ULs) based on traditional toxicity endpoints. In addition, if increased intake of a substance has been shown to increase the risk of a chronic disease, such a relationship should be characterized as the range where a decreased intake is beneficial. If the increase in risk only occurs at intakes greater than the traditional UL, no chronic disease Dietary Reference Intake would be required, because avoiding intakes greater than the UL will avoid the chronic disease risk.
What Are Acceptable Levels of Confidence in the Intake-Response Data?
Specifying a DRI involves a continuum of options related to specifying a range; the certainty in the evidence might vary at different nutrient levels. Recommending the chronic disease DRIs involves decisions related to the type of DRIs, acceptable level of confidence in the intake-response data, and balancing health risks and benefits. The committee concluded that factors considered in rating the certainty of evidence delineated by GRADE are also appropriate when evaluating the certainty of the intake-response relationship. The committee supports a variant of option 2, with a number of additional considerations that need to be evaluated on a case-by-case basis. Reliable and accurate intake data are particularly important to develop intake-response relationships.
Recommendation 8. The committee recommends that to develop a chronic disease Dietary Reference Intake, the level of certainty
in the intake-response relationship should generally be the same as the level of certainty for a determination of causality, that is, at least “moderate,” using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE). However, in some cases, for example when a food substance increases chronic disease risk, the level of certainty considered acceptable might be lower. In all cases, a thorough description of the scientific uncertainties is essential in describing quantitative intake-response relationships. Requiring at least “moderate” certainty extends to cases where relationships between intake and a surrogate marker and between the same surrogate marker and the chronic disease are characterized separately, in a piecemeal (i.e., two-stage) approach.
What Approaches Can Be Taken to Make Decisions When Benefits and Harms Overlap?
DRI committees will explore the decisions related to all DRIs (i.e., based on adequacy, toxicity, and chronic diseases) and, in some cases, harms and benefits could overlap. For example, an NOFS that increases the risk of one chronic disease may decrease the risk of another. It might not always be possible to avoid the overlap between benefits and harms (option 1). It also might not be appropriate for DRI committees to establish criteria related to severity and risk (option 2), which is typically the role of risk managers (e.g., federal agencies). Ideally, DRI committees would conduct analyses with existing models for dealing with competing health risks and benefits. If this is not possible, option 3 is recommended.
Recommendation 9. The committee recommends that, if possible, health risk/benefit analyses be conducted and the method to characterize and decide on the balance be made explicit and transparent. Such a decision needs to consider the certainty of evidence for harms and benefits of changing intake and be based on clearly articulated public health goals. If Dietary Reference Intake committees do not perform such risk/benefit analyses, it is still necessary to describe the disease outcomes and their severities, the magnitudes of risk increases and decreases over various ranges of intakes, and other factors that would allow users to make informed decisions.
INTEGRATING CHRONIC DISEASE DRIs IN THE CURRENT PROCESS
What Should Be the Organizational Process to Develop All DRIs?
Current practice is that DRIs for one or more related nutrients are develop by one committee under the auspices of the National Academies. When chronic disease considerations are integrated in the process, all committee members need to exchange ideas and coordinate their recommendations, particularly when harms and benefits overlap. The committee recommends a variation of option 1 such that for each set of NOFSs under review, a single DRI parent committee would be formed. This organization would allow, if necessary, for the formation of two subcommittees—one subcommittee would address DRIs for the prevention of nutrient deficiencies and minimizing toxicities and the other subcommittee would address DRIs for reducing the risk of chronic disease. Creating two separate processes (option 2) would not allow for sufficient exchange of ideas between the two committees.
Recommendation 10. Because of the need for close coordination and exchange of ideas when setting Dietary Reference Intakes (DRIs) based on indicators of adequacy, toxicity, and chronic disease, one single National Academies of Sciences, Engineering, and Medicine parent committee should develop DRIs for the prevention of nutrient deficiencies and toxicities and for reducing the risk of chronic disease. Due to the need for different expertise and different methodological considerations, two subcommittees could be established at the discretion of the parent committee, for reviewing evidence on (1) adequacy and toxicity and (2) chronic disease, respectively.
What Should Be the Starting Point of Chronic Disease DRIs?
The committee concluded that continuing with the current approach of establishing DRIs for individual or small groups of related NOFSs (option 1) has advantages compared to establishing DRIs for multiple food substances that are related to a single chronic disease endpoint (option 2). First, it may be premature to change the current process before additional experience is gained. Second, the scientific literature and study designs tend to explore relationships substance by substance rather than on all substances related to one disease. Finally, balancing of harms and benefits will be more challenging when NOFSs contribute to more than one chronic
disease because there would be more than one DRI committee addressing the same nutrient.
Recommendation 11. When sufficient evidence exists to develop chronic disease Dietary Reference Intakes for one or more nutrient or other food substances (NOFSs) that are interrelated in their causal relationships with one or more chronic diseases, a committee should be convened to review the evidence of their association with all selected diseases. Using a chronic disease as the starting point for the review is not recommended because balancing health risks and benefits for multiple NOFSs that are related to a single chronic disease endpoint will be a challenge in cases where the same NOFSs might be associated with more than one chronic disease.
GUIDING PRINCIPLES FOR THE PROCESS OF ESTABLISHING CHRONIC DISEASE DRIs
The committee developed the following guiding principles as a foundation for a scientifically credible chronic disease DRI process. Although recommendations about integrating chronic disease as a consideration in setting DRIs should be revisited in the future as more practice and knowledge are acquired, these guiding principles are meant to withstand scientific and methodological advances that will occur in the future.
With respect to systematic reviews:
- Well-structured and established protocols that include the question of interest and analytical frameworks are necessary to address multiple major and ancillary scientific issues related to the degree of confidence in evidence for causal associations.
- Protocols should be developed with guidance from a technical expert panel that includes relevant content experts in nutrition science, toxicology, scientific study design and analysis, public health, biostatistics, nutrition epidemiology and chronic disease epidemiology, and disease pathogenesis.
- In consultation with the technical expert panel, systematic reviews should be sufficiently inclusive of all study designs that potentially contribute to evaluation of the causal NOFS-chronic disease relationship of interest and identification of associated intake-response relationships.
- Protocols should include studies that use various dietary assessment approaches, including self-report and biomarkers of intake, while
-
taking the quality of exposure assessment into account when rating study quality.
- Protocols should include studies that document outcomes or surrogates of outcomes of potential importance for assessing benefits and harms, while taking the quality of outcome assessments into account in rating study quality.
- Instruments and analytical methods applied to systematic reviews should be thoughtfully chosen and defensible. Instruments to assess the internal validity of the studies should include considerations that apply to nutrition research and various study designs (observational and intervention studies).
- Results from the systematic review should be clearly presented in study-by-study evidence tables and summary tables of the total evidence for each outcome and study type.
With respect to DRI committee reviews of the totality of the evidence:
- The DRI committees should include content experts and methodologists relevant to the primary scientific issues and to evidence review. DRI committees should be free of significant financial, intellectual, and professional conflicts of interest. In some cases, the required expertise might not be found without some conflicts of interest. In such cases, it is necessary to identify, disclose, and manage any potential conflicts of interest. Mechanisms to allow for interactions between the DRI committee and members representing both the technical expert panel and systematic review team, while also protecting against inappropriate influence on the systematic review methods, are strongly encouraged.
- Particular elements of needed expertise will be guided by the general scientific question(s) and specific questions and will generally include nutrition science, scientific study design and analysis, public health, biostatistics, nutrition and chronic disease epidemiology, disease pathogenesis, and evidence review conduct.
- The evidence review should be sufficiently comprehensive to anticipate the major scientific issues and methods that will likely be a part of the ensuing guideline development process.
- Sufficient documentation, clarity, and transparency in the evidence review process is needed so that others can comprehend and evaluate this process and its activities, methodological considerations, final decisions, and the rationale for decisions about each outcome.
- The review of the evidence and other aspects of the systematic review should be replicable and subject to expert peer review.
- When apparent discrepancies in the evidence exist, DRI commit-
-
tees should attempt to determine whether they can be explained by differences in methodology or conceptualization of diet–disease relationships and, where possible, incorporate such explanations into the process of rating the evidence.
- Where they exist, quantitative intake-response relationships should include a thorough description of the scientific uncertainties associated with them.
REFERENCES
Calder, P. C., A. Boobis, D. Braun, C. L. Champ, L. Dye, S. Einother, A. Greyling, C. Matthys, P. Putz, S. Wopereis, J. V. Woodside, and J. M. Antoine. 2017. Improving selection of markers in nutrition research: Evaluation of the criteria proposed by the ILSI Europe Marker Validation Initiative. Nutr Res Rev 30(1):73-81.
Clarke, R. 2017. Pattern of lipid biomarkers and risk of cardiovascular disease. Presented at the Workshop of the Committee on Development of Guiding Principles for the Inclusion of Chronic Disease Endpoints in Future Dietary Reference Intakes, January 9, 2017, Washington, DC.
IOM (Institute of Medicine). 2010. Evaluation of biomarkers and surrogate endpoints in chronic disease. Washington, DC: The National Academies Press.
IOM. 2011a. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
IOM. 2011b. Finding what works in health care: Standards for systematic reviews. Washington, DC: The National Academies Press.
Schunemann, H., J. Brozek, G. Guyatt, and A. Oxman. 2013. Introduction to GRADE Handbook. http://gdt.guidelinedevelopment.org/app/handbook/handbook.html (accessed May 16, 2017).
Taylor, C. L. 2008. Framework for DRI development: Components “known” and components “to be explored.” Washington, DC.
Yetley, E. A., A. J. MacFarlane, L. S. Greene-Finestone, C. Garza, J. D. Ard, S. A. Atkinson, D. M. Bier, A. L. Carriquiry, W. R. Harlan, D. Hattis, J. C. King, D. Krewski, D. L. O’Connor, R. L. Prentice, J. V. Rodricks, and G. A. Wells. 2017. Options for basing Dietary Reference Intakes (DRIs) on chronic disease endpoints: Report from a joint US-/Canadian-sponsored working group. Am J Clin Nutr 105(1):249S-285S.
This page intentionally left blank.


















