5
Risk Assessment: Incorporating Chemical–Microbiome Interactions
Enormous advances have been made in the last several decades in the sciences devoted to understanding the health effects of environmental chemicals, but substantial knowledge gaps still leave large uncertainties in health risk assessments. Studies of chemical–microbiome interactions and their consequences indicate that further research could advance understanding of human health risk posed by exposure to environmental chemicals. Specifically, understanding chemical–microbiome interactions is likely to improve the use of results of studies in epidemiology, toxicology, and exposure science in carrying out risk assessments. Knowledge of chemical–microbiome interactions might also help to explain differences between animal toxicity studies and human responses, to extrapolate research findings from animal studies to humans, and to identify unrecognized health consequences of environmental exposures. The large variation in the microbiome compositions in populations of different life stages, sexes, and ethnicities might inform the extrapolation of findings of studies of laboratory animals to human populations. Epidemiology studies in different populations might sometimes reveal different responses to chemical exposure, and it is possible that those differences might be explained by population variation in microbiome composition. It is reasonable to hypothesize that adequate consideration of the roles of human microbiomes will improve understanding of the health risks posed by exposures to environmental chemicals.
This chapter discusses aspects of the integration of microbiome considerations into risk assessment. First, the risk-assessment process and data sources are briefly reviewed. Next, major risk-assessment issues in chemical–microbiome interactions are identified. Because exposure assessment is a key element of the risk-assessment process, exposure-assessment challenges are discussed in the context of the human microbiome, and several examples are provided to illustrate the challenges. A discussion of research needed to address risk-assessment needs concludes the chapter and sets up the committee’s research strategy described in Chapter 6.
THE RISK-ASSESSMENT PROCESS
Risk assessment used in regulatory programs in the United States and globally has been undergoing considerable reform and advancement in recent years. Much of the reform is aimed at moving from intensive chemical-by-chemical assessment to large-scale assessments that might more easily determine which of the thousands of chemicals used in industry pose health risks that should be assessed in depth. Changes also have been proposed that will improve the usefulness of risk-assessment results for making risk-management decisions (Schaafsma et al. 2009; Krewski et al. 2014).
Science and Decisions: Advancing Risk Assessment (NRC 2009) provides widely accepted guidance on ensuring the scientific adequacy of risk assessments and their utility for decision-making. Effective decisions begin with development of a clear and complete understanding of the problem for which a decision is needed. That initial problem formulation is then used to guide the development of a risk assessment that is certain to be
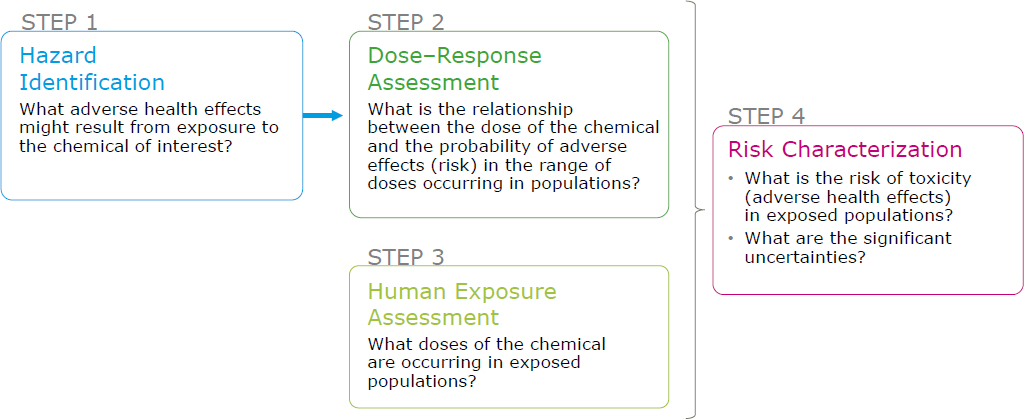
useful for decision-making. The risk assessment is then conducted by using a general framework first proposed in a 1983 National Research Council study (NRC 1983). That framework, illustrated in Figure 5-1, is still considered valid and is used by the US Environmental Protection Agency (EPA) and related agencies. In addition, EPA has developed numerous guidelines for the conduct of risk assessment (EPA 2014a), including guidelines for addressing the recommendations in Science and Decisions. EPA guidelines (EPA 2016) describe the optimal evaluation and use of data that often contain inconsistencies and that require proper treatment of uncertainty in extrapolation of results from animal or human studies of limited scope to policies designed to protect the general public. Other federal agencies have developed guidelines to meet their risk-assessment needs.
It is important to note that the Science and Decisions model can be used to guide the development of a research program of the type outlined in Chapter 6 of the present report. The problem to be addressed—understanding the role of chemical–microbiome interactions in human health risk—leads to the formulation of research questions whose answers make risk assessments that include consideration of the microbiome and its influences feasible.
DATA SOURCES AND REQUIREMENTS FOR RISK ASSESSMENT
Most of the environmental toxicology data used for hazard identification and dose–response assessment (Figure 5-1) are derived from studies of experimental animals. Internationally standardized protocols for animal studies are available to investigate general toxicities and a wide array of effects, including effects on reproduction and development and effects on the immune, nervous, and endocrine systems. Epidemiology studies have contributed valuable information on some important environmental pollutants; often, they are based on exposures in occupational settings. Toxicity data from fundamental research are also used when available. Much of the basic toxicology research has focused on specialized end points and underlying mechanisms of toxic action. Various technologies have become available to study chemical interactions and responses at the molecular and cellular levels, and this knowledge provides information on toxicity mechanisms (NRC 2007; NASEM 2017). Such approaches are being scaled to high-throughput formats for rapid evaluations of large numbers of chemicals, including chemicals that have not been studied previously to any substantial degree (Kavlock and Dix 2010). Environmental toxicology is also changing with the use
of gene-editing technologies that allow rapid probing of the genetic aspects of toxicity mechanisms (Shen et al. 2015).
Exposure science, essential for understanding human health risk, has undergone remarkable advances in the last few decades (NRC 2012; NASEM 2017). The science has moved understanding of human exposures to chemicals from simple descriptions of the presence of a chemical in air, water, food, or a consumer product to far more complete depictions of multiple chemical exposures on and in the body and of the variations in these exposures over life stages and in different population groups. The technologies for developing more rapid and complete exposure profiles, from the use of remote and personal sensors to the identification and sampling of key biomarkers, are contributing copious new data for environmental risk assessment. Characterization of animal and human exposure (and effects) has advanced through the use of biomonitoring, biomarkers, and physiologically based pharmacokinetic models (Vandenberg et al. 2010), which facilitate elucidation of the absorption, distribution, metabolism, and excretion of chemicals and have become especially important in informing interspecies extrapolations and characterizing interindividual variability.
EPA usually initiates a risk assessment only when there is sufficient and convincing evidence from whole-animal or epidemiology studies that exposure to a substance is causally related to one or more adverse health effects and when those studies also provide information on dose–response relationships. Research will be needed to develop and test protocols for microbiome health-effects studies that yield dose–response information; current protocols for developing toxicity data do not explicitly take into account a role of the microbiome in affecting outcomes.
MAJOR RISK-ASSESSMENT ISSUES RELATED TO CHEMICAL–MICROBIOME INTERACTIONS
As discussed in Chapter 3, research with pharmaceuticals has shown that some chemicals (for example, antimicrobial pharmaceuticals) can harm or alter human and animal microbiomes. And research with environmental chemicals and pharmaceuticals has shown that the microbiome can alter internal exposures to some chemicals by, for example, transforming a chemical to a more or less toxic form or altering uptake of a chemical. Those types of effects might not be fully evaluated in current risk-assessment practice (Dietert and Silbergeld 2015). As a result, a risk assessment might fail to provide adequate protection of the general population if chemical–microbiome interactions are not incorporated into studies implicitly or are not explicitly addressed, particularly when results from studies in animals or in a specific population are used to characterize risk to another species or population that has a different microbiome composition and function from that of the studied population. The outcome might be a mischaracterization of the nature of a hazard associated with exposure to an environmental chemical—for example, chemical–microbiome interactions might produce a different health effect from the chemical itself—or an overestimation or underestimation of the risk associated with exposure. The following sections consider the implications for each study type used in risk assessment.
Epidemiology Studies
The chemical–microbiome interaction of whatever form and magnitude is presumably integrated into epidemiology studies that are conducted in large populations and include health and exposure assessments throughout the subjects’ lifetimes (or key life stages) of exposure and in a variety of potentially confounding disease states. However, current understanding of the microbiome suggests that the results of such epidemiology studies might be useful only in describing risks to similar populations. An understanding of chemical–microbiome interactions in a population might be critical when using epidemiologic results from studies conducted in populations of different cultures, locations, life stages, and other factors that affect the microbiome. When chemical–microbiome interactions are substantive in modifying exposure or harm-
ing the microbiome, researchers might find that an incomplete understanding of the composition and role of the microbiome has complicated and limited the use of epidemiology studies in risk assessment. If more knowledge of the role of differences in the microbiome among populations in influencing chemical sensitivity were available, the information could indicate whether a study population is more or less sensitive than the general US population and thereby inform decisions regarding the appropriate magnitude of uncertainty factors.
Animal Toxicity Studies
Chemical–microbiome interactions are integrated into whole-animal toxicity studies. However, such studies typically are conducted with a homogeneous, in-bred group of animals that are maintained in standardized laboratory conditions that will affect their microbiomes. Animal studies are rarely carried out from preconception through natural death, so the temporal changes in microbiomes that contribute to risks and benefits over a lifetime might not be seen. They also rarely use experimental designs that control for common variation in microbiome composition between and within animal facilities. Furthermore, housing and test conditions are intended to minimize nonchemical stress, including exposure to pathogens.
If there are chemical–microbiome interactions that affect toxicity, a thorough understanding of the limitations in extrapolating the laboratory-animal results to humans might be necessary. For example, the mode of exposure of research animals could affect the microbiome in ways that influence risk in humans and animals differently; a gavage dose administered as a bolus, even when equivalent in milligrams per kilogram per day, might affect the microbiome of the digestive tract in ways that dietary or environmental exposure would not. Furthermore, the vehicle of administration could influence the microbiome in laboratory animals, and the temporal pattern of exposure could influence the microbiome in a manner that differs between laboratory animals and humans. A wide range of doses from low to high will need to be investigated, and risk assessors will need to know whether current default uncertainty and variability factors that have been used to extrapolate from animals to humans are sufficiently protective of public health. As with epidemiology studies, an understanding of the microbiome in the population targeted for public-health protection will be important throughout all susceptible life stages and disease conditions.
In Vitro Studies
Data derived from in vitro studies and from high-throughput testing alone are not considered a sufficient basis for risk assessment of new chemicals. However, risk assessors are interested in how to use those results in risk assessment, and the data have been recognized as valuable for providing important insights on toxicity mechanisms and setting priorities for in-depth toxicity testing (NRC 2007; NASEM 2017). In the context of the microbiome, however, the challenge of using newer techniques to screen chemicals for toxicity and exposure is exacerbated by the likelihood that the data do not incorporate chemical–microbiome interactions. New methods will be needed to expand in vitro and high-throughput testing to include the effects of the microbiome in mediating toxicity. Some types of in vitro studies might be well suited to testing the direct effect of chemicals on the microbiome and its functions (see Chapter 4).
ADDRESSING EXPOSURE CHALLENGES
In considering how the interactions between environmental chemicals and the human microbiome might influence human health risk, proper characterization of exposures plays a central role. As defined by Zartarian et al. (2005), exposure constitutes the “contact between an agent and a target. Contact takes place at an exposure surface over an exposure period.” A 2012 National Research Council report, Exposure Science in the 21st Century: A Vision and a Strategy, stated that “exposure science addresses the intensity and duration of contact of humans or other organisms with … chemical, physical, or biologic stressors … and their fate in living systems” (NRC 2012).
A key idea is that to capture its influence on risk, exposure must be characterized both conceptually and quantitatively.
How the human microbiome might mediate health risk associated with exposure to environmental chemicals, however, is barely addressed in the exposure-science literature. For example, NRC (2012) aimed to define the scope of exposure science and stated that “a central theme of this report is the interplay between the external and internal environments and the opportunity for exposure science to exploit novel technologies for assessing biologically active internal exposures from external sources”; the report does not mention the human microbiome. Similarly, exposure science has emphasized the use of information on exposures to environmental chemicals to support quantitative assessments of the associated human health risks (Fenske 2010). An important tool in risk assessment is the EPA Exposure Factors Handbook (EPA 2011); the latest edition, which spans more than 1,500 pages in 19 chapters, contains only brief mentions of microorganisms. It is evident that the exposure-science research community has devoted relatively little attention to microbial exposures of humans; although there is some work defining risk associated with pathogens, the community has been nearly silent on the human microbiome.
The current state presents a challenge and an opportunity. There is a need to expand the scope of exposure science to incorporate the emerging understanding of the roles of the human microbiome as an agent that influences exposures to and risks posed by environmental chemicals. Because knowledge is developing rapidly in this field, there will be a need to refresh the effort on a regular basis.
In the near term, risk assessments will likely continue to incorporate risk factors derived from laboratory studies of animal models. A critical feature for generating accurate risk factors is the proper characterization of exposures in the test animals. Exposure science has made and can continue to make important contributions to such efforts. Doing that well for circumstances in which health risks are influenced by microbiomes will require amendment of some of the core ideas in exposure science. For example, it will be necessary to rethink the concepts of “external” and “internal” in relation to exposure. Traditionally, an external exposure is related to interactions that occur at contact surfaces. Hence, exposure science would seek to quantify the nature and extent of interaction between a chemical and a human receptor at the boundaries that separate the environment from the human body, such as in lung tissues, on skin surfaces, and at the gastrointestinal epithelium. Those ideas could be readily extended to address cases in which a surface-resident microbiota mediates exposure by transforming the chemical or changing the permeability of the epithelium. The alteration of the chemical might influence the associated risk for many reasons, for example, by changing the rate of uptake across the body’s tissues, influencing chemical fate within the body, and changing the toxicity of the agent. In such cases, it could be appropriate to consider the composition and function of the human microbiome as an exposure factor. However, a greater challenge is to incorporate within the framework of exposure science the potential for chemical exposures to alter the human microbiome itself and thereby influence risks. For the specific case of the gastrointestinal tract, it is not clear how to define where the contact surface occurs. How to apply the conceptual differentiation between external and internal exposure is not apparent when the target is a human-associated community of microorganisms that might be influenced by the chemical and interacts with its human host.
The following sections provide examples that illustrate some of the challenges and opportunities in integrating exposure-science principles into studies of how the human microbiome influences risks posed by exposure to environmental chemicals. The examples highlight chemicals that are recognized as environmental health risks and about which there is at least suggestive evidence that microbiome–chemical interactions could modulate their exposure or health risk. One or two examples are provided that pertain to each of the major microbiome sites listed in the committee’s statement of task (gut, skin, and respiratory tract).
Formaldehyde and the Upper Respiratory Tract
Formaldehyde is a widely used industrial chemical. Indoor formaldehyde concentrations can be increased by emissions from indoor sources, most notably urea-formaldehyde resins that are used in the manufacturing of wood-based construction materials, such as plywood (Salthammer et al. 2010). Historically important concerns about formaldehyde exposure have been associated with the use of a spray-foam insulation material in the 1970s (L’Abbé and Hoey 1984) and with trailers used for emergency housing in the aftermath of flooding associated with Hurricane Katrina (Murphy et al. 2013). EPA recently issued regulations limiting emissions of formaldehyde from wood products (81 Fed. Reg. 89674 [2016]).
Formaldehyde has a low molecular mass, high vapor pressure, and high water solubility (Salthammer et al. 2010). Because of its high mobility and strong tendency to partition into aqueous solutions, the primary sites of exposure to formaldehyde are the upper respiratory tract and the eyes. In California, the chronic-exposure reference concentration is 9 µg/m3 (OEHHA 2016), a concentration that is routinely exceeded in indoor environments (Salthammer et al. 2010). Furthermore, the International Agency for Research on Cancer (IARC 2012) concluded that “formaldehyde is carcinogenic to humans,” and the National Toxicology Program (NTP 2016) concluded that formaldehyde is “known to be a human carcinogen.”
Formaldehyde is used as a disinfectant and sterilant. In liquid form, it has a wide range of effectiveness by “alkylating the amino and sulfhydral groups of proteins and ring nitrogen atoms of purine bases” (Rutala et al. 2008). The question is whether inhalation exposure to formaldehyde at high concentrations indoors could disrupt the human microbiome associated with upper airways? If so, would such disruption alter health risks posed by the exposure? The literature contains no clear evidence on that point. However, given the importance of known adverse health effects of formaldehyde, a relatively straightforward exposure pathway, and the antimicrobial properties of formaldehyde, it seems to be a strong candidate for studies to investigate whether and how exposure to an environmental chemical might interact with the microbiota of the upper airways in a manner that influences health risks. What is particularly germane is whether exposures to formaldehyde at concentrations encountered (or potentially encountered) in the environment interact with the microbiota in the upper airways in a manner that materially influences associated health risks, considering both irritancy responses associated with acute exposures and cancer risk associated with cumulative exposures.
Phthalates and the Transdermal Pathway
Phthalates are a class of semivolatile organic compounds widely used in commercial products, including vinyl flooring and many consumer products. One important application of phthalates is as plasticizers: they are added to polymeric materials to provide flexibility. In that function, the phthalates are not bound to the host polymeric material but instead can migrate into other media. Indoor concentrations of several phthalates are commonly much higher than outdoor concentrations (Rudel et al. 2010), including butyl benzyl phthalate, bis(2ethylhexyl) phthalate (DEHP), diethyl phthalate (DEP), dibutyl phthalate (DBP), and diisobutyl phthalate. Among the health concerns associated with phthalate exposures are reproductive toxicity and developmental toxicity (Kavlock et al. 2006; Lyche et al. 2009; Kay et al. 2014).
Human exposure to phthalates can occur through multiple pathways, including ingestion (dietary and nondietary), inhalation, and transdermal routes (Colacino et al. 2010; Bekö et al. 2013). Recent research has shown that transdermal permeation can make a contribution to human intake of the relatively volatile species DEP and DBP that is quantitatively similar to that of inhalation (Weschler et al. 2015). And clothing has been found to be an important moderator of dermal exposure. Initially clean clothing can inhibit dermal exposure, whereas previously worn clothing exposed to airborne phthalates at high concentrations can be a vector for increased uptake (Morrison et al. 2016).
In the case of DEHP, the effectiveness of uptake has been linked to its chemical conversion to the monoester, mono(2-ethyhexyl) phthalate (MEHP). Lipases are known to play an important role in that process, and the presence of lipases in microorganisms has been noted (Nakamiya et al. 2005; Kavlock et al. 2006). Although, the extent to which such conversion occurs because of gastrointestinal or skin microbiota is unknown, Hopf et al. (2014) have shown that when DEHP is applied to viable skin in aqueous emulsion, the DEHP is converted to MEHP, which can permeate the skin more effectively. Several microbial species have been shown to convert DEHP to MEHP. It appears worthwhile to investigate further whether skin-associated and other microbiomes mediate phthalate uptake and thereby influence risk through chemical conversion of the diesters to monoesters.
Triclosan and the Microbiome
Triclosan presents a potentially important case to consider. It was created as an antimicrobial agent for use in health-care settings. Because of concerns about outbreaks of new diseases, such as severe acute respiratory syndrome, triclosan began to be widely incorporated into a broad array of items, including many cleaning agents and personal-care products. The combination of deliberate, nonspecific antimicrobial action and widespread distribution into the uncontrolled environment raises general and specific concerns. Halden (2014) effectively summarized the issue as follows: “The polychlorinated aromatic antimicrobials triclosan and triclocarban are in widespread use for killing microorganisms indiscriminately, rapidly, and by nonspecific action. While their utility in healthcare settings is undisputed, benefits to users of antimicrobial personal care products are few to none. Yet, these latter, high-volume uses have caused widespread contamination of the environment, wildlife, and human populations.”
Because triclosan is widely used in liquid products applied on the body surface, the general population experiences a high degree of exposure intimacy. On the basis of US production and import data and biomonitoring evidence, Nazaroff et al. (2012) estimated that about 1–2% of all the triclosan used in US commerce enters human bodies and is excreted in urine. Research of Csiszar et al. (2016) substantiates that finding: considering 518 chemicals used in personal-care products, they found that the median product intake fraction was 2% for chemicals in wash-off products and 50% for chemicals in leave-on products. Exposures of the human microbiome as a consequence of inadvertent ingestion (for example, of toothpaste and mouthwash), dermal product use (for example, soaps), and inhalation (Mandin et al. 2016) are certain to occur.
Research is beginning to probe whether exposure to triclosan can disrupt the microbiome. Recent animal studies indicate that triclosan exposure can affect the gut microbiome. For example, exposure of mice to triclosan via drinking water caused an alteration in gut microbial composition that favored the selection of bacteria that had genes related to “triclosan resistance, stress response, antibiotic resistance and heavy metal resistance” (Gao et al. 2017). And exposure of zebrafish to triclosan via the diet altered composition and ecologic dynamics of the gut microbiota (Gaulke et al. 2016).
Human studies have also explored the possible effects of triclosan on the microbiome. Poole et al. (2016) conducted a double-blind crossover study in which 13–16 healthy subjects used household and personal-care products that did or did not contain triclosan and triclocarban for 4-month periods. They concluded that “although there was a significant difference in the amount of triclosan in the urine between the [trial] phases, no differences were found in microbiome composition, metabolic or endocrine markers, or weight.” However, Yee and Gilbert (2016) summarized the evidence about the possible role of triclosan in shaping the human microbiome. They highlight the importance of considering hospitals that provide maternity services and note that more than 98% of infants “are particularly naïve to microbes [and] their microbiota is vulnerable at this developmental stage.” Given widespread human exposure, research to investigate the effects of triclosan on the human mi-
crobiome and to answer such questions as whether early-life exposure to triclosan is predisposing infants to adverse health outcomes appears to be warranted.
Nitrate, Arsenic, and the Gut Microbiome: A Case for Re-evaluation?
As discussed in Chapter 2, the gut is the site with the greatest mass of microbiota, and it is the best studied. A rapidly developing literature describes the many ways in which the gut microbiome influences human health. A smaller literature is emerging on how the gut microbiota mediates health risks posed by exposures to environmental chemicals. Two examples are discussed briefly here to illustrate the nature and significance of how transformations of environmental chemicals that are influenced by the gut microbiome might alter health risk.
Nitrate exposure is linked to the blood disorder methemoglobinemia (blue-baby syndrome), a potentially fatal condition in neonates. EPA’s current health risk assessment of nitrate is based on infant susceptibility (EPA 1991). Although fetal hemoglobin, intestinal pH, and other factors increase susceptibility (Nelson and Hostetler 2003), EPA’s supporting data include a concern that nitrate toxicity appears to be exacerbated by gastrointestinal illness in infants. Research has shown that multiple factors influence bacteria of the infant intestinal tract and nitrate bioactivation (Jones et al. 2015). However, research has not been conducted to characterize and quantify the relationship between the gut microbiome, nitrate exposure, and the risk of methemoglobinemia. Research could be conducted to test the current concern that infant intestinal health status is a key component of nitrate risk assessment. Tools are available to characterize the microbiome in healthy infants and to quantify ex vivo bioactivation of nitrate and nitrite under various conditions. Such research would establish a baseline with which microbiome composition and metabolic capacity of infants who have intestinal illness could be compared. New information on the role of the microbiome in altering susceptibility to nitrate toxicity in infants could be important in refining the outdated assessment that focused on infants or in altering future health risk assessments for nitrate exposure at other life stages.
Arsenic is a ubiquitous contaminant of natural systems with important potential for harming human health. Attaining public drinking-water standards and soil guidance concentrations has been problematic (ATSDR 2007; EPA 2010; NRC 2013; Carlin et al. 2016). Accurate risk assessments of arsenic exposure are important both to protect public health and to ensure that expenditures for water treatment and soil remediation are warranted. Arsenic risk assessments have been based partly on epidemiology studies conducted in multiple countries, including Taiwan, Chile, Argentina, and Bangladesh (EPA 2010; FDA 2016). Those studies are based on populations whose dietary intake can be increased by arsenic in water, rice, and foods cooked in arsenic-contaminated water. As EPA conducts a new arsenic risk assessment, the role of arsenic ingestion will continue to be a key scientific issue (EPA 2014b). Current risk assessments do not incorporate the emerging evidence, described in Chapter 3, that the gut microbiome affects the bioavailability and metabolism of arsenic in animal models and in human microbiome cultures (Diaz-Bone and Van de Wiele 2010; Van de Wiele et al. 2010). New research in mice shows that exposure to arsenic alters the microbiome, perhaps in ways that harm health (Lu et al. 2014). Furthermore, a recently published study found that mice exposed to arsenic at environmentally relevant dietary concentrations had alterations in the gut-microbiome composition and in a variety of important bacterial functional pathways (Chi et al. in press). Characterizing microbiomes in populations from different geographic locations and with different cultural practices (for example, food sources and preparation methods) might reveal different exposure profiles. Differences in the microbiomes of study populations might become as important in interpreting epidemiology study results as measuring arsenic intake in exposed populations.
RESEARCH TO ADDRESS RISK-ASSESSMENT NEEDS AND IMPLICATIONS
Whether interactions between some environmental chemicals and the microbiome have adverse health consequences cannot be known without substantial new research. Accordingly, research is needed to address when the microbiome is a direct target of chemical toxicity and is perturbed by chemical exposures in ways that have measurable adverse health effects on the host, when the microbiome is not itself perturbed in harmful ways but modulates exposure to environmental chemicals, and how variability and variation of the human microbiome influence the consequences of microbiome–environmental-chemical interactions. Research to inform risk assessment could focus on the following elements:
- The extent to which harm to the microbiome is incorporated into or detectable in conventional animal testing.
- The extent to which microbiomes differ substantially among animal strains and species and between humans and animals.
- Characterization of the degree to which microbiomes can recover from insult or adapt to continuing insult.
- How different microbiomes of the body of most relevance to environmental exposures—gut, lungs, and skin—are affected and evaluated.
- Understanding exposure pathways and how physicochemical properties of environmental chemicals influence exposure and mediate uptake.
- How differences among humans in their microbiomes affect their susceptibility or resistance to environmental chemicals.
In addition to the elements listed above, new approaches might be needed to evaluate dose–response relationships that might be affected simultaneously from chemically induced changes in the microbiome, chemically induced toxicity to the host, and microbiome-induced effects on host health. Understanding and integrating the relative effects on the dose–response relationships will likely pose a key risk-assessment challenge. Chapter 6 describes the committee’s research strategy to begin to address the important topics noted.
It is not likely that definitive answers to the important risk-assessment issues will emerge unless a substantial research program is under way; even then, one can expect answers to emerge slowly. Thus, as in all current risk assessment efforts, default assumptions will continue to be used to address data gaps and other uncertainties. Moreover, it might not be possible to develop clear criteria for adverse effects on the microbiome itself, so other targets of toxicity (ranging from intracellular components to organ systems) will remain the subjects of risk assessment even if a chemical has an adverse effect on the human microbiome. Nonetheless, understanding that the microbiome might be adversely affected is important because such knowledge might provide new insights into health effects and human population sensitivities. Uncertainty factors that have traditionally been used in risk assessment should be able to accommodate new knowledge regarding interactions of environmental chemicals and the human microbiome.
IDENTIFYING HEALTH RISK ASSESSMENTS THAT MIGHT NEED RE-EVALUATION
As data on chemical–microbiome interactions emerge and are used in risk assessments, it is likely that some previous risk assessments will be considered outdated and will need to be re-evaluated. The merits of health-risk re-evaluation are well established and based on optimizing public-health benefits, either to provide greater protection from potential health effects or to reduce the expenditure of resources on unnecessary exposure-management actions. Identification of new findings and evaluation of the likelihood that they will alter assessments are activities that are already being pursued in federal and state risk-assessment programs. Indicators of the magnitude of changes in risk that would present opportunities to improve public-health protection have not been uniformly established.
Findings that might result in a re-evaluation of chemical risk assessments include such outcomes as magnitude of change (lower or higher) in toxicity or exposure and the discovery of a previously unrecognized but highly susceptible population or life stage (that is, one that is highly sensitive to the chemical or is highly exposed). Increased susceptibility might be a consequence of such factors as sex, age, behavior, or health status. As new study protocols that account more completely for chemical–microbiome interactions are developed, the resulting data might reveal previously unknown exposures or health outcomes that are important to consider in applying research results to protect public health. Results from the types of studies described in Chapter 6 could inform current risk-management practices and help to guide priorities for future research on chemical–microbiome interactions relevant to health risk assessment. The work could result in changes in variability and uncertainty factors that could be applied to past risk assessments to adjust for a new understanding of chemical–microbiome interactions. It could also provide important information that would help in setting priorities for retesting or re-evaluation. Classes of chemicals, disease states, life stages, health end points, or other generalizable groupings of data could be identified for re-evaluation because the chemical–microbiome interaction has not been fully included in past studies or the interaction indicates greater exposure and health consequences than previously recognized.
A chemical-specific assessment of risk could be undertaken whenever emerging evidence of toxicity or exposure uniquely related to perturbation of the microbiome becomes available. Such work might already be possible in emerging research on arsenic and on nitrate. Results can be compared with past assessments to begin to gauge the impact that future re-evaluations might have on risk management. On a larger scale, existing risk assessments can be evaluated to determine the extent to which known or possible chemical–microbiome interactions are likely to alter the assessments. A screening evaluation can be used to set priorities for chemicals that warrant reassessment, and new assessments can follow.
Answering the question of whether past assessments of health risk were sufficiently “robust” requires a science-policy finding of the change in health or exposure measures that provides opportunities for public-health protection. A small increment or decrement that is identified in a risk assessment is unlikely to drive wide-scale research in chemical–microbiome interactions or methodologic changes in risk assessment protocols. However, risk managers might support research if the increased risk were found for a highly specific population or site or for an easily regulated chemical.
FINDINGS
- Adequate consideration of the roles of the human microbiome will improve understanding of the health risks posed by exposures to environmental chemicals.
- Data used for hazard identification and dose–response assessment are derived from studies of experimental animals; however, it is not clear that current methods for generating animal data or extrapolating from animals to humans can incorporate the influence of the microbiome on adverse health outcomes properly.
- Characterization of animal and human exposure and health risk has advanced through the use of biomonitoring, biomarkers, and physiologically based pharmacokinetic models. Those methods have not been consistently applied to or do not encompass aspects known to be important for the microbiome, such as life stage, sex, and disease state.
- A risk assessment might fail to provide adequate protection of the general population if chemical–microbiome interactions are not incorporated implicitly into studies or explicitly addressed, particularly when results from studies in animals or in one population are used to characterize risk to another species or population that has a different microbiome composition and function. The outcome might be a mischaracterization of the nature of a hazard associated with exposure to an environmental chemical or an overestimation or underestimation of the risk associated with exposure.
- There is a need to expand the scope of exposure science to incorporate the emerging understanding of the roles of the human microbiome and its components as agents that influence exposures to and risks posed by environmental chemicals.
- Studying how the human microbiome is affected by chemicals requires a clear understanding of the nature and magnitude of change in the microbiome that might result in adverse health effects.
REFERENCES
ATSDR (Agency for Toxic Substances and Disease Registry). 2007. Public Health Statement: Arsenic. August 2007 [online]. Available: https://www.atsdr.cdc.gov/phs/phs.asp?id=18&tid=3 (accessed March 11, 2017).
Bekö, G., C.J. Weschler, S. Langer, M. Callesen, J. Toftum, and G. Clausen. 2013. Children’s phthalate intakes and resultant cumulative exposures estimated from urine compared with estimates from dust ingestion, inhalation and dermal absorption in their homes and daycare centers. PLoS ONE 8(4):e62442.
Carlin, D.J., M.F. Naujokas, K.D. Bradham, J. Cowden, M. Heacock, H.F. Henry, J.S. Lee, D.J. Thomas, C. Thompson, E.J. Tokar, M.P. Waalkes, L.S. Birnbaum, and W.A. Suk. 2016. Arsenic and environmental health: State of the science and future research opportunities. Environ. Health Perspect. 124(7):890-899.
Chi, L., X. Bian, B. Gao, P. Tu, H. Ru, and K. Lu. In press. The effects of an environmentally relevant level of arsenic on the gut microbiome and its functional metagenome. Toxicol. Sci.
Colacino, J.A., T.R. Harris, and A. Schecter. 2010. Dietary intake is associated with phthalate body burden in a nationally representative sample. Environ. Health Perspect. 118(7):998-1003.
Csiszar, S.A., A.S. Ernstoff, P. Fantke, D.E. Meyer, and O. Jolliet. 2016. High-throughput exposure modeling to support prioritization of chemicals in personal care products. Chemosphere 163:490-498.
Diaz-Bone, R.A., and T. Van de Wiele. 2010. Biotransformation of metal(loid)s by intestinal microorganisms. Pure Appl. Chem. 82(2):409-427.
Dietert, R.R., and E.K. Silbergeld. 2015. Biomarkers for the 21st century: Listening to the microbiome. Toxicol. Sci. 144(2):208-216.
EPA (U.S. Environmental Protection Agency). 1991. Nitrate. Chemical Assessment Summary. Integrated Risk Information System [online]. Available: https://cfpub.epa.gov/ncea/iris2/chemicalLanding.cfm?substance_nmbr=76 (accessed July 20, 2017).
EPA. 2010. Toxicological Review of Inorganic Arsenic (Cancer) (2010 External Review Draft). EPA/635/R-10/001. U.S. Environmental Protection Agency, Washington, DC [online]. Available: https://cfpub.epa.gov/ncea/iris_drafts/recordisplay.cfm?deid=219111 (accessed July 20, 2017).
EPA. 2011. Exposure Factors Handbook 2011 Edition (Final Report). EPA/600/R-09/052F. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC. September 2011 [online]. Available: https://cfpub.epa.gov/ncea/risk/recordisplay.cfm?deid=236252 (accessed July 20, 2017).
EPA. 2014a. Framework for Human Health Risk Assessment to Inform Decision Making. EPA/100/R-14/001. Office of Science Advisor, Risk Assessment Forum, U.S. Environmental Protection Agency, Washington DC. April 2014 [online]. Available: https://www.epa.gov/sites/production/files/2014-12/documents/hhra-framework-final-2014.pdf (accessed July 20, 2017).
EPA. 2014b. IRIS Bimonthly Public Meeting (Jun 2014) [online]. Available: https://www.epa.gov/iris/iris-bimonthly-public-meeting-jun-2014#ia (accessed March 11, 2017).
EPA. 2016. Risk Assessment Guidelines [online]. Available: https://www.epa.gov/risk/risk-assessment-guidelines [accessed July 20, 2017].
FDA (Food and Drug Administration). 2016. Arsenic in Rice and Rice Products. Risk Assessment Report. March 2016 [online]. Available: https://www.fda.gov/downloads/Food/FoodScienceResearch/RiskSafetyAssessment/UCM486543.pdf [accessed November 9, 2017].
Fenske, R.A. 2010. For good measure: Origins and prospects of exposure science (2007 Wesolowski Award Lecture). J. Expo. Sci. Environ. Epidemiol. 20(6):493-502.
Gao, B., P. Tu, X. Bian, L. Chi, H. Ru, and K. Lu. 2017. Profound perturbation induced by triclosan exposure in mouse gut microbiome: A less resilient microbial community with elevated antibiotic and metal resistomes. BMC Pharmacol. Toxicol. 18(1):46.
Gaulke, C.A., C.L. Barton, S. Proffitt, R.L. Tanguay, and T.J. Sharpton, 2016. Triclosan exposure is associated with rapid restructuring of the microbiome in adult zebrafish. PLoS ONE 11(5):e0154632.
Halden, R.U. 2014. On the need and speed of regulating triclosan and triclocarban in the United States. Environ. Sci. Technol. 48(7):3603-3611.
Hopf, N.B., A. Berthet, D. Vernez, F. Langard, P. Spring, and R. Gaudin. 2014. Skin permeation and metabolism of di(2-ethylhexyl) phthalate (DEHP). Toxicol. Lett. 224(1):47-53.
IARC (International Agency for Research on Cancer). 2012. Formaldehyde. Pp. 401-435 in Chemical Agents and Related Occupations, IARC Monographs on the Evaluation of Carcinogenic Risks to Humans Vol. 100F. Lyon, France: IARC [online]. Available: http://monographs.iarc.fr/ENG/Monographs/vol100F/mono100F-29.pdf (accessed 13 December 2016).
Jones, J.A., A.O. Hopper, G.G. Power, and A.B. Blood. 2015. Dietary intake and bio-activation of nitrite and nitrate in newborn infants. Pediatr. Res. 77(1-2):173-181.
Kavlock, R., and D. Dix. 2010. Computational toxicology as implemented by the U.S. EPA: Providing high throughput decision support tools for screening and assessing chemical exposure, hazard, and risk. J. Toxicol. Environ. Health B Crit. Rev. 13(2-4):197-217.
Kavlock, R., D. Barr, K. Boekelheide, W. Breslin, P. Breysse, R. Chapin, K. Gaido, E. Hodgson, M. Marcus, K. Shea, and P. Williams. 2006. NTP-CERHR Expert Panel update on the reproductive and developmental toxicity of di(2-ethylhexyl) phthalate. Reprod. Toxicol. 22(3):291-399.
Kay, V.R., M.S. Bloom, and W.G. Foster. 2014. Reproductive and developmental effects of phthalate diesters in males. Crit. Rev. Toxicol. 44(6):467-498.
Krewski, D., M. Westphal, M.E. Andersen, G.M. Paoli, W.A. Chiu, M. Al-Zoughool, M.C. Croteau, L.D. Burgoon, and I. Cote. 2014. A framework for the next generation of risk science. Environ. Health Perspect. 122(8):796-805.
L’Abbé, K.A., and J.R. Hoey. 1984. Review of the health effects of urea-formaldehyde foam insulation. Environ. Res. 35(1):246-263.
Lu, K., R.P. Abo, K.A. Schlieper, M.E. Graffam, S. Levine, J.S. Wishnok, J.A. Swenberg, S.R. Tannenbaum, and J.G. Fox. 2014. Arsenic exposure perturbs the gut microbiome and its metabolic profile in mice: An integrated metagenomics and metabolomics analysis. Environ. Health Perspect. 122(3):284-291.
Lyche, J.L., A.C. Gutleb, Å. Bergman, G.S. Eriksen, A.J. Murk, E. Ropstad, M. Saunders, and J.U. Skaare. 2009. Reproductive and developmental toxicity of phthalates. J. Toxicol. Environ. Health B Crit. Rev. 12(4):225-249.
Mandin, C., F. Mercier, O. Ramalho, J.P. Lucas, E. Gilles, O. Blanchard, N. Bonvallot, P. Glorennec, and B. Le Bot. 2016. Semi-volatile organic compounds in the particulate phase in dwellings: A nationwide survey in France. At-mos. Environ. 136:82-94.
Morrison, G.C., C.J. Weschler, G. Bekö, H.M. Koch, T. Salthammer, T. Schripp, J. Toftum, and G. Clausen. 2016. Role of clothing in both accelerating and impeding dermal absorption of airborne SVOCs. J. Expo. Sci. Environ. Epidemiol. 26(1):113-118.
Murphy, M.W., J.F. Lando, S.M. Kieszak, M.E. Sutter, G.P. Noonan, J.M. Brunkard, and M.A. McGeehin. 2013. Formaldehyde levels in FEMA-supplied travel trailers, park models, and mobile homes in Louisiana and Mississippi. Indoor Air 23(2):134-141.
Nakamiya, K., H. Takagi, T. Nakayama, H. Ito, H. Tsuruga, J.S. Edmonds, and M. Morita. 2005. Microbial production and vaporization of mono-(2-ethylhexyl) phthalate from di-(2-ethylhexyl) phthalate by microorganisms inside houses. Arch. Environ. Occup. Health 60(6):321-325.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2017. Using 21st Century Science to Improve Risk-Related Evaluations. Washington, DC: The National Academies Press.
Nazaroff, W., C.J. Weschler, J.C. Little, and E.A. Cohen Hubal. 2012. Intake to production ratio: A measure of exposure intimacy for manufactured chemicals. Environ. Health Perspect. 120(12):1678-1683.
Nelson, K.A., and M.A. Hostetler. 2003. An infant with methemoglobinemia. Hosp. Physician 62:31-38.
NRC (National Research Council). 1983. Risk Assessment in the Federal Government: Managing the Process. Washington, DC: National Academy Press.
NRC. 2007. Toxicity Testing in the 21st Century: A Vision and a Strategy. Washington, DC: The National Academies Press.
NRC. 2009. Science and Decisions: Advancing Risk Assessment. Washington, DC: The National Academies Press.
NRC. 2012. Exposure Science in the 21st Century: A Vision and a Strategy. Washington, DC: The National Academies Press.
NRC. 2013. Critical Aspects of EPA’s IRIS Assessment of Inorganic Arsenic: Interim Report. Washington, DC: The National Academies Press.
NTP (National Toxicology Program). 2016. Formaldehyde. Report on Carcinogens, 14th Ed. [online]. Available: https://ntp.niehs.nih.gov/ntp/roc/content/profiles/formaldehyde.pdf [accessed September 19, 2017].
OEHHA (Office of Environmental Health Hazard Assessment). 2016. Formaldehyde. California Environmental Protection Agency [online]. Available: https://oehha.ca.gov/air/chemicals/formaldehyde (accessed July 20, 2017).
Poole, A.C., L. Pischel, C. Ley, G. Suh, J.K. Goodrich, T.D. Haggerty, R.E. Ley, and J. Parsonnet. 2016. Crossover control study of the effect of personal care products containing triclosan on the microbiome. mSphere 1:e00056-15.
Rudel, R.A., R.E. Dodson, L.J. Perovich, R. Morello-Frosch, D.E. Camann, M.M. Zuniga, A.Y. Yau, A.C. Just, and J.G. Brody. 2010. Semivolatile endocrine-disrupting compounds in paired indoor and outdoor air in two northern California communities. Environ. Sci. Technol. 44(17):6583-6590.
Rutala, W.A., D.J. Weber, and Healthcare Infection Control Practices Advisory Committee. 2008. Guideline for Disinfection and Sterilization of Healthcare Facilities, 2008. Centers for Disease Control and Prevention [online]. Available: https://www.cdc.gov/infectioncontrol/pdf/guidelines/disinfection-guidelines.pdf (accessed December 13, 2016).
Salthammer, T., S. Mentese, and R. Marutzky. 2010. Formaldehyde in the indoor environment. Chem. Rev. 110(4):2536-2572.
Schaafsma, G., E.D. Kroese, E.L.J.P. Tielemans, J.J.M. Van de Sandt, and C.J. Van Leeuwen. 2009. REACH, non-testing approaches and the urgent need for a change in mind set. Regul. Toxicol. Pharm. 53(1):70-80.
Shen, H., C.M. McHale, M.T. Smith, and L. Zhang. 2015. Functional genomic screening approaches in mechanistic toxicology and potential future applications of CRISPR-Cas9. Mutat. Res. 764:31-42.
Van de Wiele, T., C.M. Gallawa, K.M. Kubachka, J.T. Creed, N. Basta, E.A. Dayton, S. Whitacre, G. Du Laing, and K. Bradham. 2010. Arsenic metabolism by human gut microbiota upon in vitro digestion of contaminated soils. Environ. Health Perspect. 118(7):1004-1009.
Vandenberg, L.N., I. Chahoud, J.J. Heindel, V. Padmanabhan, F.J.R. Paumgartten, and G. Schoenfelder. 2010. Urinary, circulating, and tissue biomonitoring studies indicate widespread exposure to bisphenol A. Environ. Health Perspect. 118(8):1055-1070.
Weschler, C.J., G. Bekö, H.M. Koch, T. Salthammer, T. Schripp, J. Toftum, and G. Clausen. 2015. Transdermal uptake of diethyl phthalate and di(n-butyl) phthalate directly from air: Experimental verification. Environ. Health Perspect. 123(10):928-934.
Yee, A.L., and J.A. Gilbert. 2016. Is triclosan harming your microbiome? Science 353(6297):348-349.
Zartarian, V., T. Bahadori, and T. McKone. 2005. Adoption of an official ISEA glossary. J. Expo. Anal. Environ. Epidemiol. 15(1):1-5.













