4
Processes to Enable Appropriate Decision Making Regarding the Return of Individual Research Results
Chapter 3 discusses the importance of the validity of research results that may be returned to participants, but result validity is not the only consideration for investigators deciding whether to return individual research results. This chapter addresses two other key considerations that need to be weighed by investigators on a study-by-study basis—(1) the value of the result to the participant and (2) the feasibility of returning it—and it also provides a conceptual framework to support decision making.
Given the numerous technical and operational considerations associated with the return of individual research results, investigators conducting research on human biospecimens need to consider early in their study design process whether individual results may be returned to participants. Research sponsors and institutions can ensure that such assessments are performed routinely by requiring return-of-results plans in their funding applications and institutional review board (IRB) review processes, respectively, ensuring that value and feasibility have been appropriately considered and that, when relevant, the costs of returning results are included in budgets. The return-of-research plan should take into account not only whether the investigator will offer some or all research results to participants, but also what he or she will do in the case that a participant requests results or if unanticipated results are generated that warrant disclosure. Research institutions and sponsors can also support investigators by connecting them with institutional resources, networks, and training and by engaging participants, patients, and community groups in the development of policy and guidance.
DIMENSIONS TO CONSIDER IN DECISIONS ON RETURNING INDIVIDUAL RESEARCH RESULTS
If results are to be returned to individual participants, the value, risks, and burdens associated with the return will need to be balanced. Below, the committee discusses several dimensions that influence study-specific decisions regarding whether and which individual research results should be returned to participants. These include the value of the research result to the participant and the feasibility of returning individual research results.
Value of the Result to the Participant
Resources for biomedical research—including research funding and investigators’ time—have significant societal value and, accordingly, need to be carefully stewarded. If resources are to be applied to the return of individual research results, the results should have value to the participant, and the benefits of disclosing the results should outweigh the risks. The sections below address different approaches to considering value and discuss steps that can be taken to better understand the value of individual research results from the participant’s perspective.
Defining Value in the Context of Returning Individual Research Results
Decision-making approaches regarding the return of individual research results have traditionally focused on several specific criteria for evaluating the expected value or usefulness of the results to participants. The factors that contribute to value include the analytic and clinical validity, clinical utility, and personal utility1 of the results (Bookman et al., 2006; Ravitsky and Wilfond, 2006).
If analytic validity (AV) and clinical validity (CV)2 have not been established, the results may be misleading, misinterpreted, or have uncertain meaning. This raises questions about the value to the participant of results that lack AV and CV and the appropriateness of returning them, particularly in cases where disclosure is associated with potential risks (e.g., from taking inappropriate actions or failing to take needed actions due to a false sense of security). However, since CV can change over time (IOM, 2016), uncertain meaning should not automatically rule out disclosure. For example, future research may uncover the clinical significance of a genetic variant that, at the time of a study, may have been categorized as a variant of uncertain significance.
___________________
1 Information has personal utility if it can be reasonably used by participants for personal decision making, actions, or self-understanding (Bunnik et al., 2014).
2Analytic validity is the ability of a test to measure what it is designed to measure (NASEM, 2017). Clinical validity is a test’s ability to identify or predict accurately and reliably the clinically defined disorder or final health or medical outcomes of interest in an individual (NASEM, 2017).
As discussed in Chapter 3, it is important to have laboratory quality management systems or peer-review processes in place in order to give stakeholders (investigators, institutions, sponsors, and participants) confidence in the validity of a study’s results before they are returned to participants. In many cases, however, even when the AV and CV of a test have been established, its clinical utility may remain unclear (processes for establishing clinical utility are discussed in Box 4-1). The clinical utility of a test is an indication of whether it can provide information that can be used to inform patient–clinician decisions regarding the prevention, management, or treatment of a disease or health condition—and, in particular, information that would be expected to yield measurable improvement in clinical outcomes (Teutsch et al., 2009). The term is often used interchangeably with “clinical actionability,” which is “the degree to which a result may be used to guide medical or health decisions” (MRCT Center, 2017, p. 11), though there
are subtle differences between the terms since evidence may not yet exist that the clinical action that is taken results in improved outcomes (i.e., clinical utility).
It is important to note that a research result that lacks clear clinical utility may still have personal utility or meaning to a participant. Participants have a variety of motivations for wanting their results, and sometimes the utility and value of the information do not relate to clinical care (SACHRP, 2016). Information has personal utility if it can be reasonably used by participants for personal decision making, actions, or self-understanding (Bunnik et al., 2014). As discussed in Chapter 2, results with personal utility can help in life planning and reproductive decisions or can inform participants on the origins or risk factors for a specific disease or condition, helping patients better understand their disease or risk of disease (Bunnik et al., 2014; PCSBI, 2013). Value can also arise from gaining knowledge about oneself for the sake of knowledge—with no medical or health-related meaning. For a condition like Huntington’s disease, genetic test results have no treatment benefit, but for some individuals with known risk, the results may still have personal utility (Bunnik et al., 2014) by, for example, relieving the anxiety associated with not knowing whether one has the disease genes and by informing decisions to have children. Similarly, lineage information from an ancestry study may have significant personal meaning for an individual who was adopted and seeking information on ethnic identity. Personal utility may also arise from the guidance the test results offer for preventative health decisions; such decisions may fall outside of medical decision making with a clinician. For example, expert panels have concluded that exposures to certain endocrine-disrupting compounds should be reduced as a health precaution—which implies that biological measurements of a person’s exposure to these compounds may have health value, even though they are not now an established part of clinical care (Bergman et al., 2012; Diamanti-Kandarakis et al., 2009).
There is no clear dividing line between clinical and personal utility, and some results may have both characteristics, particularly if clinical utility is broadly defined. In one real-world example, investigators identified genes associated with hypercoagulability, and the results were returned to a participant on the basis that the individual would be able to take preventive action during lengthy air travel (e.g., standing periodically) to reduce the risk of stroke. Additionally, results that provide an end to rounds of diagnostic testing—the so-called “diagnostic odyssey”—can prevent further unnecessary (and potentially invasive) clinical testing and may also have personal value to participants in the form of a sense of relief from a newfound understanding of a health condition (Sawyer et al., 2016).
Despite this overlap between clinical and personal utility, existing frameworks and decision tools suggest a hierarchy among the characteristics of research results that may be returned to participants, and they tend to prioritize clinical utility over personal utility in assessing the value of research results (Fabsitz et al., 2010; Ravitsky and Wilfond, 2006). As discussed in Chapter 2, strong arguments can be made for investigators having an ethical obligation to offer participants results
that are analytically valid, indicative of a substantial health risk to the participant, and actionable (Fabsitz et al., 2010). Except for this kind of scenario, however, the subordination of personal utility may not be appropriate. In a notable departure from the approaches of past expert groups, the committee has chosen to deemphasize the respective influences of clinical and personal utility in decisions regarding the return of individual research results by focusing more inclusively on results that have “value to participants,” with the understanding that the value of a result from the perspective of the participant might entail either clinical utility or personal utility or both and may also arise from the result having personal meaning (see Box 4-2 for a summary of previous expert group recommendations). This participant-centric approach recognizes that the value of a result is not necessarily tied to its use. To clarify, defining value in this way is not meant to imply that each participant needs to be queried regarding which results would be meaningful to him or her, but it does require the investigator to consider value from the participant’s perspective rather than from the more traditional clinical perspective.
The committee also includes as a key determinant of value the benefits of returning a result relative to the risks to the participant. As discussed in Chapter 3, research testing may not be equivalent to clinical testing in purpose or quality.
If the limitations of research results are not adequately conveyed and understood, the research results may be misused by participants or their physicians to make unsupported clinical or personal decisions. Similarly, in a non-clinical setting, such decisions can result in inappropriate personal, life partner, child-bearing, and other life planning decisions. It is particularly important that the communication of results and their meaning and degree of uncertainty is done in such a way that it minimizes confusion and misinterpretation (discussed in more detail in Chapter 5). The weighing of benefits and risks is discussed later in this chapter in the context of a decision-making framework and illustrative examples.
It is important to bear in mind that value is not merely a function of the nature of the test result itself. Contextual factors—such as whether an individual has the condition under study or has an increased risk of developing it—will significantly affect participant perceptions regarding the value of a research result (Cadigan et al., 2011). BRCA1 test results would be expected to have greater value to a participant with an increased risk for hereditary breast cancer than to a participant without such a family history (Bunnik et al., 2014). In the latter case, in fact, the results could provide a false sense of reassurance if they were disclosed without information regarding the incidence of nonhereditary breast cancer in the general population. The study design and the timing for returning individual research results can also influence perceptions of value for some kinds of results. For example, in the case of blinded clinical trials, returning test results may be prohibited until the end of the study in order to maintain the integrity of the research (MRCT Center, 2017). For genetic test results, the informational value to participants may not be diminished if they are returned months or even years later, but some studies may batch test samples at the end of an intervention or observation period, and, for disease states or environmental exposure that may change with time (e.g., cholesterol levels or some volatile environmental contaminants), the timeline for biospecimen collection, testing, and return of results may significantly affect the value of the return.
Ascertaining Participant Needs, Preferences, and Values
Given that the value of a research result to a participant will be influenced by perspective and context, the investigators, institutions, and research sponsors involved in this work need to be cautious about making assumptions regarding the kinds of results that participants may find meaningful. Expert-identified criteria do not always reflect participant preferences and values (Arora and McHorney, 2000; Epstein and Street, 2011; Epstein et al., 2010; Guyatt et al., 2004; Little et al., 2001). For example, recent surveys of participants in three different longitudinal cohort studies found that the participants in one of the cohorts but not the other two expressed a preference for receiving results about gene variants associated with preventability. In fact, for some participants, none of the expert-identified test result characteristics (severity, preventability, disease risk, reproductive implications)
were associated with their preferences for receiving results.3 Another study which examined preferences for return by using hypothetical scenarios found that participant perceptions varied widely concerning the perceived value of results for untreatable conditions or of a finding of unknown significance, with some participants desiring access to all available results and others suggesting that these data could be a burden to a participant (Murphy et al., 2008). In a study of African American parents, participants viewed the return of aggregate results as less preferable than the return of individual results, as the aggregate results failed to provide any perceived personal-level benefit (Halverson and Ross, 2012). Some participants, understanding the trade-off in terms of slowing scientific discovery, have indicated that they would prefer to receive few or even no research results (Bollinger et al., 2014).4 These findings emphasize the importance of working to understand what participants would find to be of value and what their preferences are for receiving results after the benefits, risks, and trade-offs have been discussed.
Ascertaining and incorporating participant needs, preferences, and values into decision-making processes regarding the return of individual research results can be done at the study level but also in the development of policy or guidance. Both actions are critical to advancing a more participant-centric research paradigm and may require the engagement of community members or research participants, or both.
In the context of individual studies, engagement is a bidirectional relationship between stakeholders—the individuals or groups affected by the research—and investigators which informs decision making (e.g., about research selection, design, conduct, or use) (Ahmed and Palermo, 2010; CTSA Community Engagement Key Function Committee Task Force on the Principles of Community Engagement, 2011). There is a range of engagement in research, including consultation, collaboration, and partnership (CTSA Community Engagement Key Function Committee Task Force on the Principles of Community Engagement, 2011). Engagement as a partner may not be necessary for the return of research results. The appropriate level and mechanism will depend on the specific characteristics of each study. In general, the imperative to engage community members in decision making about the return of individual research results increases as the degree of interface with participants during the research increases, and it depends on the potential implications of the research findings for participants or a community. For example, in a community-based participatory research (CBPR) study (see Box 4-3) that is assessing the exposure to potentially hazardous materials,
___________________
3 G. L. Splansky, Preferences for return of genetic results among participants in the Jackson and Framingham Heart Studies. Document provided to the Committee on the Return of Individual-Specific Research Results Generated in Research Laboratories, Washington, DC, October 6, 2017. Available by request through the National Academies’ Public Access Records Office.
4 Testimony of Ellen Wagner of Parent Project Muscular Dystrophy at the public meeting of the Committee on the Return of Individual-Specific Research Results Generated in Research Laboratories on September 6, 2017.
community members may partner with investigators to jointly decide which results should be returned to participants. Such an approach would not make sense for research using stored human biospecimens, where investigators have little or no contact with the contributors of the specimens. However, it should be noted that IRBs may be unfamiliar with CBPR and may require training to understand the value of results to participants and to accommodate the iterative nature of CBPR to prevent unnecessary delays and barriers to return (Brown et al., 2010; Saxton et al., 2015).
Ideally, the level of engagement in a study would be determined in collaboration with community members or with guidance from engagement experts. Study-specific engagement will not be necessary if individual results cannot be returned to participants—e.g., when biospecimens have been de-identified—and may not be required if investigators can reasonably rely on existing documentation of participant needs, preferences, and values in the literature or from past experiences working with community groups.
Table 4-1 describes the range of stakeholder engagement that can be applied in the return of individual research results. The table has the following key messages: (1) a number of approaches have been successfully used to involve stakeholders in research; (2) the number of stakeholders engaged and the extent of engagement should reflect the goals and aims of the research; (3) stakeholder roles range from providing brief, targeted input to highly involved, leadership roles; and (4) the training and experience required varies based on the stakeholders roles. As shown in the table, there is an inverse relationship between the extent of engagement and the number of stakeholders engaged, which leads some researchers to ask whether it is more important to have more engagement or a larger number of stakeholders. Because the best approach aligns with the aims of the study, neither is always more important. Approaches that are poorly aligned are less likely to elicit the stakeholder input needed and may be burdensome. Furthermore, if feasible, it is often useful for research teams to employ more than one approach—to both get the benefit of hearing from a large number of people in the general population affected by the condition being studied and work in an ongoing manner with a small number of participant or community representatives with deeper knowledge of the study process.
The different methods for engaging patient and community stakeholder groups about the return of individual research results (as presented in Table 4-1) can also be used to help determine an appropriate engagement approach to ascertain participant needs, preferences, and values regarding the return of individual research results to the general population being studied. For example, it may be valuable to solicit input from a large group of stakeholders using methods such as surveys, online polling, crowdsourcing, social media, and listening sessions. When more detailed input is required, investigators can use such methods as focus groups, nominal group techniques, Delphi methods, semi-structured interviews, and community engagement studios (Joosten et al., 2015). In some
TABLE 4-1 The Range of Engagement in the Return of Individual Research Results
| STAKEHOLDER ROLE | EXTENT/DEPTH OF ENGAGEMENT | BRIEF DESCRIP TION | EXAMPLES/METHODS | NUMBER OF STAKEHOLDERS IN ROLE |
|---|---|---|---|---|
| Principal Investigator (PI) or Co-PI | Responsible for decision making | Stakeholders drive the research and serve as leaders or co-leaders of the project. They are responsible for all aspects of the study and are directly involved in all decision making | Partnered research (multiple PIs), community-based participatory research, patient powered research network | 1–2 |
| Research Partner or Team Member | Part of team making decisions | Stakeholders are members of the research team and have direct involvement in the design, conduct, and dissemination of research; including helping to understand and make decisions on the return of research results | Community-engaged research, engaged team science | 2–6 |
| Governance or Advisory Group | Provides oversight or guidance to make decisions | Stakeholders consider feedback and findings on participants’ needs, priorities, and values and provide guidance to the research team to make decisions on the return of research results | Advisory boards, councils and committees | 4–25 |
| Consultants, Interviewees, Panelists, Reviewers, etc. | Provides detailed input on participant needs, preferences and values | Stakeholders serve in specific, time-limited roles and provide detailed input on participants’ needs, preferences, and values on the return of research results | Focus groups, semi-structured interviews, Delphi techniques, community engagement studios | 10–100 |
| Knowledge Users and Experiencers (general population affected by condition being studied) | Provides input on participant needs, preferences and values | Stakeholders engage using methods designed to reach a large number of people and provide perspectives on participants’ needs, preferences, and values related to return of research results | Surveys, online polling, crowdsourcing, social media, town hall meetings | 100+ |
cases, participant representatives may participate in oversight or advisory roles—for example to guide institutional policy or as members of bodies that make case-by-case decisions regarding the return of individual research results. Joanne Murabito, clinic director for the Framingham Heart Study (FHS), told the committee during its public workshop that an ethics advisory board comprising participants from all of the study cohorts as well as local physicians, key community leaders, a genetic counselor, and a medical ethicist provides guidance to investigators on results that should be returned to FHS participants.5 Stakeholders in these ongoing roles have effective communication skills and prior leadership experience and may also have research experience. The meaningful engagement of advisory boards and oversight groups requires adequate preparation, clearly defined roles and expectations, bidirectional communication, and numerous opportunities for the stakeholders to provide input (Mott and Crawford, 2008; Newman et al., 2011). Stakeholders may also serve as integral members of the research team, contractually or in a consulting role, or, as in the community-based participatory research model discussed in Box 4-3, as co-principal investigators of the study. Stakeholders across the continuum need appropriate compensation and support to meaningfully engage. Compensation should be commensurate with the stakeholders’ roles, responsibilities, and experience (Black et al., 2013a). It may not be necessary to offer compensation for an engagement that is brief and not burdensome.
For certain kinds of studies, particularly those that will involve recruitment and significant interaction between researchers and participants, obtaining stakeholder input on which individual research results should be returned ideally will be initiated in the study design phase in order to help investigators understand participant preferences, weigh the benefits and risks, and plan for disclosure (e.g., concerning consent and the communication of results). At this early stage, input should be sought from representative community members (i.e., individuals who will be able to represent the prospective participants but may not be those enrolling in the specific research study). The timing of this participant preference assessment relative to applying for funding for the research may vary depending on the extent of this assessment. Extensive engagement processes may be proposed as a part of the research to be funded.
If a decision is made to prospectively offer at least some individual research results, it creates an imperative for investigators to later engage the enrolled study participants in discussions (likely during the consent process) regarding the kinds of results that may be returned and how those results could be communicated to ensure that they are meaningful to participants (as discussed in more detail in Chapter 5). In survey and focus group discussions, participants have expressed a
___________________
5 Testimony of Joanne Murabito of the Framingham Heart Study at the public meeting of the Committee on the Return of Individual-Specific Research Results Generated in Research Laboratories on September 6, 2017.
desire to be given choices and an opportunity to indicate preferences regarding which research results will be offered (Murphy et al., 2008). While the personal value of a result will vary greatly depending on the individual, it would probably not be feasible to tailor plans for returning results according to the individual preferences of each participant. One possible way to accommodate varying participant preferences, depending on the study constraints, would be to offer a tiered disclosure approach where participants are offered a range of options for disclosure (Rothstein, 2006). Investigators can also investigate individual preferences for the return of results over the course of the study (see Chapter 5 on consent) or at the end of a study if a participant requests results that were not offered as part of the study plan.
Many investigators have little training or experience in identifying and convening stakeholders and may be unfamiliar with strategies to implement key principles of engagement such as co-learning and cultivating mutual trust and respect. Therefore, to accomplish this researchers will need to leverage the expertise of community engagement cores, engagement specialists, and engagement scientists to develop plans to engage stakeholders if evidence of their needs, preferences, and values has not previously been captured. Due in part to the infrastructure developed by the Clinical and Translational Science Awards (CTSAs)6 and broader dissemination of engagement methods by the Patient-Centered Outcomes Research Institute (PCORI),7 many research institutions have engagement experts available to assist researchers. The cost, time, and effort of engagement can be minimized by leveraging existing knowledge resources and engagement infrastructure to assess the preferences and needs of communities. Research funding agencies and sponsors can further support investigators by providing guidance and other resources to ensure that engagement needs are not viewed as a barrier to returning individual research results.
Community and patient advocacy organizations, which are often focused on harnessing the power of groups of patients and strategically deploying their assets to drive better outcomes and increased value from research, also have a role. Community organizations and advocacy groups can facilitate communication between those they represent and an institution’s engagement core, a study’s investigators, and research sponsors in order to convey the needs and preferences of those in their community concerning the return of results and engagement in those decisions. Over time, as community engagement experts within institutions (e.g., individuals within community engagement cores at CTSA) and investigators
___________________
6 Community engagement has been a key component of the CTSAs since their initial funding in 2006. Each of the approximately 60 CTSAs (Wilkins et al., 2013) has a community engagement program that may provide useful examples of successful local community engagement models (CTSA Community Engagement Key Function Committee Task Force on the Principles of Community Engagement, 2011; Paberzs et al., 2014; Wilkins et al., 2013).
7 PCORI’s Engagement in Health Research Literature Explorer contains a searchable list of publications on engagement in health literature (PCORI, 2018).
gain experience with returning individual research results and publish their accumulated knowledge in the literature, the engagement requirements for ascertaining participant needs, preferences, and values may lessen, reducing the burden for investigators.
CONCLUSION: The study-specific engagement of community members and participant representatives in order to ascertain participant needs, preferences, and values related to the return of individual research results will be important for some, but not all, research involving the testing of human biospecimens. Engagement may be limited or unnecessary when biospecimens have been de-identified or if investigators can reasonably rely on the existing documentation of participant needs, preferences, and values in the literature or from past experiences working with community groups.
CONCLUSION: Many investigators will be new to engagement activities and will need to rely on existing models as they develop study protocols. Engagement models, guidance, and numerous informational resources have been developed to guide and support patient and community engagement. Investigators may need to be made aware of their existence or to receive training to effectively engage participants in decision making about the return of individual research results.
CONCLUSION: As the return of individual research results becomes more routine and a body of evidence is built and becomes more accessible to investigators (see Recommendation 11), the process for understanding and considering participant needs, preferences, and values will become less burdensome.
Feasibility of Returning Individual Results to Participants
The return of individual research results requires not just a consideration of the value of the results to the participants, but also of the feasibility of the return. Broadly speaking, feasibility depends on the burden involved in making the returns and the resources available to carry out the returns. There is a small body of literature (mainly from environmental exposure and genetic studies) that addresses the burdens that investigators face in returning individual research results (Bredenoord et al., 2011; Brody et al., 2014; Christensen et al., 2011; Ohayon et al., 2017). Among the burdens described in this literature are communication challenges, maintaining contact with research participants, and logistical and resource constraints (for example, see Box 4-4 which highlights challenges identified by investigators and IRBs) (Ohayon et al., 2017). Depending on the nature of the research and the laboratory used to test biospecimens, the burden can also include getting Clinical Laboratory Improvement Amendments (CLIA) certification.
The burden associated with the return of results varies widely, depending on the context of the study and the communication approaches used. At one extreme, a study might involve a small number of participants who are also patients of the physician–investigator. In this case, neither contacting the participants nor communicating the results effectively is likely to be a significant challenge. At the other extreme, a study might involve an analysis by a basic scientist of a large number of human biospecimens contributed by individuals who are widely distributed in terms of location and the time of specimen acquisition. In this situation, contacting the participants and organizing an effective communication of results by those with appropriate expertise would be a costly and complex endeavor. However, in some situations methods to enable return to geographically distributed participants—e.g., digital methods of communication—can be employed by investigators to make this more feasible (Boronow et al., 2017). Methods for communication of results are discussed in more detail in Chapter 5.
The resources needed to return research results include money, time, expertise, infrastructure, and personnel. The sections below detail some of the major challenges investigators have encountered when returning research results and some of the issues they should consider when making determinations on a case-by-case basis about returning individual research results.
Communicating Results
Research teams may not have the necessary expertise (e.g., being able to understand the test’s meaning in a larger medical context or within the participants’ medical context, being able to communicate the results effectively, or simply having had previous experience with these challenges) to appropriately communicate individual research results, and thus the return of results may require additional training or partnerships with professionals who do have this expertise. In the
case of genetic research, the availability and cost of genetic counselors is a big concern. At the committee’s workshop, Wendy Chung, a professor of pediatrics and medicine at Columbia University, reported that using a genetic counseling service had cost the study approximately $250 per participant, and Jessica B. Langbaum, a principal scientist at Banner Alzheimer’s Institute, said that one barrier to returning results in a national study had been that state licensure laws for genetic counselors varied and that the costs could be prohibitive for a counselor to become licensed in multiple states.
Investigators and IRBs will need to consider various literacies of research participants—e.g., general literacy, health literacy, and relevant aspects of scientific literacy—when planning the return of results, which likely will require new or increased interactions with and the involvement of social scientists, communication experts, the community, and patient advocacy groups. Individuals with different levels of literacy will require different levels of support, which may necessitate varying methods of education and follow-up activities (Terry, 2012). Chapter 5 addresses these communication needs in greater detail.
Maintaining Contact with Participants
Maintaining contact with participants is another frequently cited barrier to the return of results. At the committee’s public workshop, Carolyn Compton, a professor of life sciences at Arizona State University, said that when she worked at the National Cancer Institute it would not have been possible to re-contact trial participants because they did not maintain contact information in their database. “If there were a way that this information could be centralized and made available so that patients could be contacted, that would be an enormous step forward,” she said. Furthermore, contact information may change over time, or research teams may lose contact with participants from transient populations. In the case of one environmental exposure survey (Ohayon, 2017), the researchers involved adopted various strategies to work around these challenges, such as asking for alternative contact information, informing participants when study results would likely be disclosed in the consent process, and “disseminating partial results as they became available to ensure more frequent communication with participants” (Ohayon et al., 2017, p. 145).
Regulatory Requirements
As described in Chapter 3, the Centers for Medicare & Medicaid Services interprets CLIA regulations so as to require that individual results can only be returned if testing is performed in a CLIA-certified laboratory, and getting CLIA certification is a major burden for many investigators who are planning to return
results. At the committee’s workshop, all four investigator panelists8 agreed that CLIA certification would not be practical for many research laboratories, given the nature of the research they conduct (discussed in detail in Chapter 3).9 While investigators could in theory conduct their follow-up testing in CLIA-certified laboratories on the subset of results they plan to return, the ability to perform this follow-up testing might be constrained if investigators are not part of an institution that is affiliated with a CLIA-certified laboratory that performs the specific test or do not have the budget to perform tests again within their institution or via a third-party facility. Chung said that in her case, getting samples retested in a CLIA-certified laboratory tripled the cost of testing per participant. Furthermore, in many cases retesting requires participants to give new biospecimen samples since it cannot be ensured that research laboratories without certification followed quality management processes when collecting, processing, and storing biospecimens. While in the case of genetic testing, getting additional biospecimen samples from participants may be relatively simple (only requiring a saliva swab), in many other types of research, collecting additional samples (e.g., blood, biopsies) would be much more onerous, invasive, or impossible. Thus, if CLIA certification continues to be a requirement for the return of individual research results, it will not be feasible for many investigators to return results without significant additional resources from their institutions and funding agencies, and even then many laboratories would still be left out. If a new quality management system with a tiered approach was developed that was appropriate for research laboratories (Recommendation 2 in Chapter 3), the costs to implement this system would still be significant for many laboratories not familiar with quality processes, but ultimately it would allow a much broader swath of laboratories to return individual research results.
Another regulatory requirement that investigators planning to return research results should take into consideration is the Food and Drug Administration’s (FDA’s) investigational device exemption (IDE). The details of the IDE regulatory process and how it applies to the return of individual research results are discussed in detail in Chapter 6. However, it should be noted that if a study falls under this regulation and does not meet the requirements for exemption, then investigators need to prepare an IDE submission to FDA. The rigorousness of the requirements for submission depends on the risk determination of the study, which is determined by the investigators and IRB. According to a meeting
___________________
8 Wendy Chung, the Kennedy Family Professor of Pediatrics and Medicine, Columbia University; Carolyn Compton, a professor of life sciences, Arizona State University; Jessica B. Langbaum, principal scientist, Banner Alzheimer’s Institute; Lea C. Harty, a biobank biological materials custodian, Pfizer Inc., representative of the Industry Pharmacogenomics Working Group (I-PWG).
9 Testimonies of Wendy Chung of Columbia University, Carolyn Compton of Arizona State University, Jessica B. Langbaum of Banner Alzheimer’s Institute, and Lea C. Harty of Pfizer Inc. at the public meeting of the Committee on the Return of Individual-Specific Research Results Generated in Research Laboratories on September 6, 2017.
summary from a National Human Genome Research Institute (NHGRI) workshop, “If a study is deemed to be SR [significant risk], investigators should be aware of the time and resources that are necessary to prepare an IDE submission and see it through FDA review, as investigators bear much of the responsibility in the IDE process” (NHGRI, 2017a).
Required Resources
The resources needed to return individual research results include funding, time, expertise, personnel, and infrastructure, and finding such resources is an inevitable challenge for researchers who wish to return results. Given the many factors involved, it is very difficult to estimate ahead of time the costs and time that will be required to return results, but it should be acknowledged up front that the return of results will indeed require both time and money. Some documented costs associated with the return of results include verification of the result, genetic or other counseling beyond the disclosure of results, administrative costs, time and labor required to recontact participants, salaries for trained and qualified staff, protocol preparation, creation and printing of educational materials, and technology to enable the storage and protection of identifiable information to enable return to individual participants (Black et al., 2013b; Budin-Ljøsne et al., 2016; Christensen et al., 2011; Fernandez et al., 2004; Heaney et al., 2010; Resnik, 2011). For example, one study that returned CDKN2A research results to melanoma survivors found
Time demands averaged 161 minutes per completed disclosure. An average of 40 minutes was spent on each of the 39 GEM study participants we attempted to re-contact, plus an additional 78 minutes for each of the 19 participants who agreed to receive results. The financial costs associated with our protocol averaged $1,322 per completed disclosure. (Christensen et al., 2011)
The costs of return will become more apparent as there is now a concerted effort among research sponsors, investigators, and institutions to return results and build an evidence base on best practices and to leverage existing resources to carry out returns more efficiently and effectively. The development of a larger evidence base will aid investigators and sponsors in future cost planning and help lower up front costs as the practices becomes more routine—and more routinely documented—and as the infrastructure and expertise get put in place.
The CTSA program is well positioned to facilitate returns at CTSA-affiliated universities. First, CTSAs provide investigators with access to clinical research cores and research nurse support, which includes providing access to CLIA-certified laboratories to facilitate the testing or retesting of specimens when appropriate. Once established, CTSAs could also provide training to investigators to meet the quality management system (see Recommendation 2) or to
ensure that core laboratories follow those quality management system practices so that they can address the diverse needs of the investigators at the institution. Furthermore, CTSA sites can provide training, expertise, and infrastructure that investigators who are planning to return results could use to effectively engage and communicate with participants (NCATS, 2018). Universities that are not connected with a CTSA may be able to form partnerships with CTSAs housed at other universities, local hospitals, or patient and community advocacy groups to provide similar support to their investigators.
A DECISION-MAKING FRAMEWORK FOR THE RETURN OF INDIVIDUAL RESEARCH RESULTS
Decisions about the return of individual research results will vary on a study-by-study basis, depending on the characteristics of the research, the results, and the participants. Such decisions will require a significant amount of judgment and several groups have provided flowcharts or frameworks to aid in the decision to return research results (Beskow and Burke, 2010; Haines et al., 2011; Holm et al., 2014; MRCT Center, 2017; Ravitsky and Wilfond, 2006)—the committee offers the conceptual framework in Figure 4-1. Under this framework, the justification for returning results becomes stronger as the potential value of the result to participants increases or as the feasibility of the return increases.
In a small number of well-defined cases there are clear and broadly accepted rationales for when a return should be either obligatory or discouraged (see Box 4-5). However, for the most part decisions will not be so clear cut.
Given the numerous technical and operational considerations inherent in the return of research results, the committee chose to provide examples illustrating how these considerations could be applied in practice. The committee found it helpful to organize the discussion into three scenarios: (1) the research team is prospectively planning to offer results to participants; (2) a participant requests his or her results from the research team or laboratory; and (3) the research team has an unanticipated result that was not part of the study protocol and is considering whether the results should be offered to participants.
Decisions on Individual Research Results That Will Be Offered to Participants
In this first scenario, the research team is considering which individual research results, if any, will be offered to participants in the study. In this case incorporating input from patient and community groups is the most helpful strategy for helping investigators understand which results are likely to be of value to participants and which are not. Additional input from scientific and clinical experts may help elucidate the potential risks and benefits and inform the decision-making process (advisory boards and other such bodies are discussed later in this chapter). Because the decisions are being made prospectively
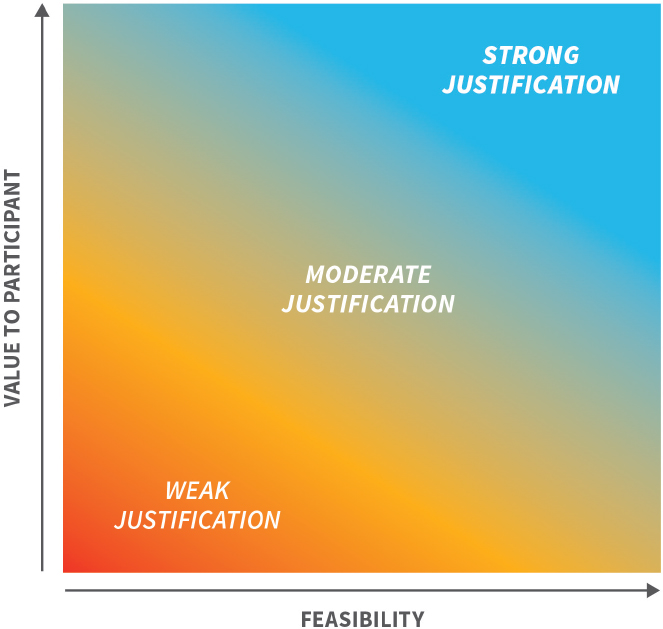
NOTES: This figure demonstrates that as the potential value of the result to participants and the feasibility of return increase, the justification for returning results becomes stronger. Value in this context means the value of a result from the perspective of the participant and might entail clinical utility or personal utility as well as personal meaning. Feasibility is determined by multiple factors, including potential challenges, the costs and burdens of returning results, and whether participants’ biospecimens are linked to the participant identity as well as the resources available to communicate the results effectively and appropriately.
and can be planned for, the research team can ensure that sample tracking and testing processes provide the necessary confidence in the validity of the result (as defined in Recommendation 3) and that the team has the necessary resources and expertise to communicate the results effectively. Thus, in this scenario the investigator should decide on the return of results by carefully weighing the value to participants along with the benefits, risks, and costs of return.
Investigators conducting research testing on human biospecimens may be faced with decisions on whether to offer individual research results for which the strength of the justification spans the full spectrum depicted in Figure 4-1. The committee identified several kinds of research results it would generally encourage investigators to offer. In surveys and focus groups participants have resoundingly expressed interest in receiving results that indicate the presence of or an increased risk for a preventable or treatable disease or health condition (Middleton et al., 2016; Murphy et al., 2008). For example, most members of a focus group indicated that they would want results from a hypothetical asthma study, and such results should, in most cases, be returned (Murphy et al., 2008). Even certain research results that lack clinical utility may still have significant personal value and would be expected to pose little risk. For example, for individuals with a debilitating disease, a biomarker that predicts disease progression may be of great value to a participant and his or her family or caretakers in life planning.10 Furthermore, as long as a lack of resources does not make it impossible, offering the results from routine clinical tests generated in the course of a research study is unlikely to pose a significant risk to the participant and should also be encouraged. When it is possible to do so without impairing the integrity of the study, offering such results in real time is likely to maximize their value to participants. It is important to note that the value of clinical test results may depend on the results (e.g., whether or not they are within the normal range) and the characteristics of the participant. For example, clinical test results within the normal range may have greater value to a research participant with a serious health condition than to a healthy volunteer, and this should weigh into decisions regarding disclosure. Investigators should also keep in mind when considering whether to offer results or to return them only upon request that participants may make assumptions when results are not offered that nothing abnormal was discovered.11
As discussed earlier in this chapter, the return of personal exposure results (e.g., blood levels of an environmental contaminant) may be expected and may even be part of a contract between the investigators and participants engaged in a community-based participatory research study. Then, depending on what is known about the health effects of the contaminant, the questions for investigators may be focused more on such issues as how the results should be returned (considerations for how results should be returned are discussed further in Chapter 5).
When there may be significant risks associated with returning results, decisions about whether to offer results will require more careful consideration. In such cases, the actionability of a result may influence the strength of the
___________________
10 Testimony of Ellen Wagner of Parent Project Muscular Dystrophy at the public meeting of the Committee on the Return of Individual Specific Research Results Generated in Research Laboratories on September 6, 2017.
11 Testimony of John Molina of Native Health at the public session of the Committee on the Return of Individual Specific Research Results Generated in Research Laboratories on December 11, 2017.
justification for its return. Participants tend to be more equivocal about receiving results for a disease or condition that is currently not treatable (e.g., Alzheimer’s disease) (Murphy et al., 2008). As discussed earlier in this chapter, however, investigators must consider in their decision-making process whether withholding the offer of results in the latter case is paternalistic, particularly given the potential personal benefits to participants who opt to receive such results (e.g., life planning, relief from the anxiety of not knowing), and they should pay attention to participant preferences and values.
As a matter of respect for autonomy, Principle 2 of the committee’s guiding principles (see Chapter 2) asserts that participants should have a right to decide whether to receive or share their results with their primary care physician, relatives, an executor, or others. Participants may need to decide whether to share research results with relatives who may be affected by the result while investigators may need to determine the timing of the return and potentially the handoff of the results to a physician if they require further clinical evaluation. While the scientific literature has explored whether an investigator may communicate research results to others, either at the investigator’s initiative or upon request by one of these individuals, there is no formal consensus on this topic (Battistuzzi et al., 2013; Beskow and O’Rourke, 2015; Chan et al., 2012; Tassé, 2011; Wolf et al., 2015). These will not be straightforward decisions, and, in particular, they will be highly context dependent. There are many considerations that will need to be taken into account in order to determine when and with whom a participant’s research results can be shared, and these will require protocol-specific determinations in conjunction with institutional policy. When there is a result that is thought to be clinically significant, the timing of the return relative to the urgency of the finding should be considered when the return is being planned and reviewed by an IRB. Sharing results with physicians, children, or community members may also be a key challenge for investigators and IRBs, particularly if participants do not want their results shared; if the result has meaning for the community, investigators struggled with how to share the data in a way that is valuable for the community while protecting participant privacy (Ohayon et al., 2017). In general, the communication of results to a participants’ family member or proxy should be done with the permission of the participant.
In some research scenarios, there may be challenges to the research team’s ability to solicit, consider, and respect the participant’s preferences for receiving or sharing results because the participant is underage, has reduced decision-making capacity, or is deceased. Offering results to the participants, their caretakers, or their family members in these situations, therefore, involves complex ethical decisions and may require additional oversight mechanisms.
Return of Results for Children, Adults Who Lack Decision-Making Capacity, and Deceased Individuals
The return of research results raises a number of considerations and complexities when the participants are young children, adults or older children who lack decision-making capacity, or individuals who die during the conduct of the study. Each of these scenarios requires special decision-making processes to ensure that sufficient protections are in place. The formulation of such guidance is beyond the scope and ability of this committee, but below we raise key issues that are part of active consideration by the scientific, medical, ethics, and legal communities.
As discussed in Chapter 2, the ethics for the return of results to individuals who lack decision-making capacity differ from the considerations relevant to those who make an informed decision to join a study. The return of results to children or to individuals who lack capacity or are deceased hinges less on considerations such as transparency, reciprocity, or perceived value by the participant and more on considerations of potential benefits to the participant and his or her family members or significant others.
Children In situations where children are participating in research, the return of research results should be addressed during the assent process with the older child and the informed consent process with parents (Anastasova et al., 2013; Avard et al., 2009). All documents and methods of communications used should be adapted to suit the child’s language, age, and sociocultural context (Avard et al., 2009). Older children’s preferences regarding whether and what type of results they would be interested in having returned, including a preference to have no knowledge of their research results (Anastasova et al., 2013), should be taken into account and considered alongside the child’s age and development (Holm et al., 2014). However, the parent or guardian of the child-participant retains the right to know health information that is relevant to the management of the child’s health (Anastasova et al., 2013), although this does not imply a right to all of the child’s research results. In developing their protocols, investigators should anticipate whether the child’s results will have relevance to the health of the child or the health or welfare of other family members. For example, a genetic variant identified in a child is likely to have been inherited from a parent, with potential health implications for that parent, and the variant may be shared by siblings and other “blood” relatives. Similarly, the results of testing for environmental toxins in a child might have importance for that child and family members or others who may share the same exposure. As in other circumstances, the justification for the disclosure of results is strongest when the results have the greatest value to participants or family members and when disclosure is most feasible for investigators. In these situations, results may be offered, or made available upon request, to the parents or legal guardian of the child. Often in such circumstances, the
parents should assist or take primary responsibility for communicating appropriate information to other family members.
Adults Who Lack Decision-Making Capacity Decision-making capacity may never be present in some individuals, or it may be temporarily or permanently lost due to illness or injury. Decision-making capacity is often not simply present or absent but rather should be considered as existing along a spectrum. Furthermore, it can be task specific—that is, an individual may have capacity for some simple decisions but lack capacity for more complicated tasks (SACHRP, 2009). Adults who lack sufficient decision-making capacity for research informed consent can be participants in research with the consent of a legally authorized representative (LAR). The federal regulations governing human research do not specifically address adults without capacity other than to require consent from the participant or a LAR.12 Who qualifies as a LAR is a matter of state law, with some variability across the 50 states and territories. IRBs will be familiar with LAR requirements in the jurisdictions where the research is being conducted. Federal regulations do not require an assent process for adults with limited decision-making capacity, although such measures have been recommended and should be considered by investigators and the IRB (SACHRP, 2009).
Consistent with Recommendation 6, the plan regarding the return of results should be addressed in the consent materials reviewed and signed by the LAR (Wolf et al., 2015), and consideration should be given as to whether the return-of-results plan should be included in assent materials. During the design of the research, all components of the participants’ vulnerabilities should be considered (Lange et al., 2013), and additional support should be made available during the informed consent process, such as supplementary educational activities, the presence of specially trained personnel, and genetic counseling. Researchers, IRBs, and participants’ representatives are responsible for ensuring that this informed consent process sufficiently protects the participant’s autonomy (Groisman et al., 2012). The participant should be an active participant in the informed consent process whenever possible, even if there is a surrogate present, and should be given the information necessary to give or decline assent to participate (Lange et al., 2013).
For adults who are at risk of losing decision-making capacity during the study, Wolf and colleagues recommend that the prospect of sharing the results with family or others be addressed in the consent process and that the participant’s decisions be honored after the participant has lost capacity (Wolf et al., 2015).
Deceased Persons Currently there is no standard practice regarding whether and how to return research results to the relatives of deceased research participants (Wolf et al., 2015). A study by Beskow and O’Rourke found that IRB chairs
___________________
12 45 C.F.R. § 46.116.
did not endorse the return of research results to family members in scenarios where the decedent had stated that he or she did not want the research results shared (Beskow and O’Rourke, 2015). Deceased persons are not considered research subjects under the Common Rule; however, under the Health Insurance Portability and Accountability Act (HIPAA) deceased persons retain certain rights to their medical data. Researchers should be aware of the possibility that family members may want to receive the research results of the decedent (Chan et al., 2012) and should address access to research results by family members following the death of the participant during the informed consent process. If the participant’s preference has not been determined in the consent process in advance of the study, HIPAA may prohibit the return of results to persons who are not the participant’s executor; this is an issue because it has been found that participants may want an individual who is not the executor to be the recipient of any results (Goodman et al., 2017). The privacy of the deceased individual also remains a consideration with respect to the return of results, particularly when the individual did not have an opportunity to decide about disclosing the results to others.
In cases where the result has clinical relevance to family members or to significant others, Chan et al. (2012) recommend that the researcher make an effort to contact the executor of the decedent’s estate or next of kin to assess whether there is interest in receiving the decedent’s results. In contrast, Wolf and colleagues recommend that investigators follow a passive disclosure policy, meaning that results, rather than being offered, would be disclosed to relatives or others only upon their request (Wolf et al., 2015). These authors suggest that a more active disclosure policy may be warranted in rare cases when the results may be critical to avert imminent harm (Wolf et al., 2015).
Decisions on Individual Research Results That Will Be Returned to Participants Upon Request
When a participant requests a result from the research team or laboratory, the calculation of whether or not to return the result is different from the above scenario. By virtue of the request, it can be assumed that the result has value to the participant. There are few data to suggest how common such requests might be (particularly if participants are informed of the option during the consent process; see Recommendation 9), but as long as the burdens or risks to the participant are not prohibitory, the committee believes that investigators should be amenable to returning, upon request, results that they had not previously offered to return. This may include results that investigators might themselves think to be of limited value to participants, such as
- drug or volatile biomarker levels from samples collected during a clinical trial but batch tested months or even years later;
- potential diagnostic biomarkers from an individual who is already aware of his or her condition (e.g., the presence of a protein biomarker in a stroke survivor that may have value in future diagnostic tests to differentiate the kinds of stroke); and
- genetic variants of uncertain significance.
In the case of genetic variants of uncertain significance, it is important to bear in mind that the significance of a result may change over time as scientific knowledge advances. Given that investigators cannot generally be expected to return results after the study funding is gone (Fabsitz et al., 2010), investigators should consider returning such results upon request whenever feasible so that participants have the option of following the state of scientific knowledge and referring back to their individual results as new discoveries are made regarding the significance of variants and other kinds of biomarkers previously not well understood. This would apply to all fields of biomedical research, not just genetics and genomics. The committee agrees with the National Heart, Lung, and Blood Institute working group that investigators do not have the obligation to return results for an indeterminate period; this includes their obligation to reanalyze participant data and follow up, as this would not be feasible. “In practical terms, investigators cannot maintain an open-ended commitment to return results and thus should plan to have the results provided before the end of the operating grant period” (Fabsitz et al., 2010, p. 6). Institutional advisory bodies may be of help to investigators as they consider the return of results with unclear significance, but, as discussed earlier in this chapter, such bodies should include representation of the participant perspective. Perhaps an extreme case of this scenario relates to an individual’s whole-genome sequence. While the committee focus in this report is on the return of results that have meaning to the participant, it recognizes that in this era of next-generation sequencing and precision medicine, the question of whether to return a whole-genome sequence is one that many investigators working in the field of genomics are facing. The return of a whole-genome sequence can be justified in much the same way as returning variants of unclear significance can be, and, in fact, returning whole-genome sequences may be less burdensome for investigators as it requires less analysis. In some cases, investigators may even feel more comfortable providing raw sequencing data upon request, as it puts the onus for seeking interpretation on the participant.13
Although, as noted in Box 4-5, the committee generally encourages the return of individual research results upon request when the resources are available to do so, it discourages the return of results where there is little confidence in the validity of the result (see Recommendation 3). However, if the laboratory that produced the result is a HIPAA-covered entity and the results are part of the
___________________
13 Testimony of Wendy Chung of Columbia University at the public meeting of the Committee on the Return of Individual-Specific Research Results Generated in Research Laboratories on September 6, 2017.
designated record set as defined by the Office of Civil Rights (see Recommendation 12), then the laboratory has a legal obligation to return the requested result.
Decisions on the Offer of Unanticipated Individual Research Results
This scenario is similar to the first one (the prospective offer of results) except that the research team does not have the advantage of planning for disclosure because they did not anticipate the result in question. In the case of findings that can be anticipated, the committee is not commenting on whether there is a duty to hunt for them, as that was outside the scope of this report. Rather, this scenario refers only to those unanticipated results that are organically generated over the course of the study but the investigator was unable to foresee. In considering what can be anticipated, however, investigators have a responsibility to look beyond the primary goal of a test and consider what other results may arise in the conduct of testing.14 In some cases such results may be urgent and should be offered to participants consistent with the ethical obligation related to duty to warn/rescue. This might include, for example, a genetic marker associated with a preventable or treatable life-threatening disease or health condition discovered during a DNA sequencing study for a different medical condition. In other cases, the result may have no clear clinical utility, and the justification for returning it will be weaker. An example could be a sex chromosome anomaly that does not require medical management that was discovered during a study on a sex-linked genetic disease. Given that the research team did not anticipate the result and therefore did not seek input on participant preferences and values during the study design phase, investigators may need to rely on published or otherwise available documentation on potential personal utility and meaning as they carefully weigh the value to the participant and the risks of disclosure.
PLANNING FOR THE RETURN OF INDIVIDUAL RESEARCH RESULTS
The responsible return of individual research results requires careful forethought and preparation (MRCT Center, 2017). Most individual research results that will be generated in the course of a study and that may be expected to have value to participants can be anticipated. Doing so can ameliorate any risks that may arise from hasty decision making and ad hoc processes for disclosure. Incorporating the return-of-results plan into the research protocol fosters transparency and enables appropriate budgeting and allocation of resources, while IRB review
___________________
14 Thoughtful investigators and IRBs can anticipate results outside of the primary study objective. This aligns with the 2013 report of the Presidential Commission for the Study of Bioethical Issues where the commission concluded that there is “a duty on the part of a research investigator to consider what incidental and secondary results might occur from genomic testing, to create a plan for the possible return of results to participants, and to inform research participants of that plan before the tests are conducted” (Weiner, 2014, p. 562).
ensures that the risks and benefits to participants are carefully considered in a peer-review process. Anticipating the consent needs of participants may improve the ability of investigators to communicate their intentions to participants and set appropriate expectations (Ravitsky and Wilfond, 2006). Advance planning also provides investigators with an opportunity to consider community needs and how results can best be returned so as not to perpetuate or deepen existing health disparities and inequities (e.g., by ensuring that factors such as race and ethnicity, language barriers, insurance status, and literacy do not affect whether a participant benefits from the disclosure of individual research results). Thus, developing a plan during the design phase of the study that addresses whether, when, and how results will be offered to participants or provided in response to a participant request can help maximize the benefits and prevent or mitigate the potential harms of returning individual research results.
During the planning process investigators should consider the types of results that might be shared, including results generated in the course of research from routine clinical tests (e.g., cholesterol levels), urgent or unanticipated findings, and primary or secondary endpoints, as well as when during the study life cycle results might be offered and shared without impairing the goals of the research. For some study designs, the sharing of in-study results—for example, those generated in real time during intervention or observation periods—may require more attention to the risks of unblinding or the introduction of bias than the baseline (e.g., used for inclusion/exclusion) and end-of-study test results (SACHRP, 2016). Importantly, the development of the plan should not be viewed as a one-time process. The plan should be dynamic, with feedback potentially informing refinements at multiple points in the study timeline. Such feedback may arise during the IRB review, during the funding application review, and from public and participant engagement (when relevant) prior to the study’s initiation and during the consent process.
In recommending that investigators conducting testing on human biospecimens be required to plan for the return of research results, the committee does not mean to imply that individual research results should be returned in all studies. In some situations it may be reasonable for the plan to state that investigators will not offer individual results to research participants. However, in such cases the plan will need to clearly convey the rationale for the investigator’s decision and how that rationale will be explained to participants during the consent process. Moreover, the investigators will still need to address whether the results will be provided upon request—for HIPAA-covered entities, this may be legally required when the results are included in the DRS—and how investigators will handle unanticipated but urgent findings.
CONCLUSION: Sponsors, institutions, IRBs, and investigators will need to balance the needs of each stakeholder and make informed decisions at each phase of the research so as to respect the participants while not hindering the conduct of science.
The Role of Research Sponsors in Supporting Appropriate Planning for the Return of Individual Research Results
The National Bioethics Advisory Commission (NBAC) recommended in 1999 that investigators conducting research on human biospecimens be required to include in their study proposals documentation of whether and how individual research results will be returned to participants (National Bioethics Advisory Commission, 1999). Although there are few data to estimate how common such plans are across all relevant disciplines, one study found that only 30 percent of survey respondents had a formal plan for returning results. More than one-third of the investigators who had no formal plan indicated that they had not considered the need for one (Rigby and Fernandez, 2005). These data, along with the reported variability in investigator and IRB experience with planning for the return of individual research results (Dressler et al., 2012; Williams et al., 2012), suggest that the NBAC recommendation has not been widely adopted. In fact, some researchers have suggested that questions regarding the need to return individual research results and incidental findings should be handled on a case-by-case basis as issues arise, rather than developing policies and plans in advance (Williams et al., 2012). Consequently, it is likely that incentives will be needed to push the field to conduct routine advance planning for the return of individual research results.
Research sponsors and funding agencies (e.g., the National Institutes of Health, PCORI, the Centers for Disease Control and Prevention, and the National Institute of Environmental Health Sciences) have considerable leverage to set
requirements and provide guidance related to planning for the return of individual research results in the studies they fund (Ohayon et al., 2017). Of note, recommendations for funding requirements related to planning for the return of individual research results have been made previously in the context of human biomonitoring studies. In 2006 the National Research Council recommended that research sponsors of human biomonitoring studies and programs require explicit planning for the communication of results in any application for funding (NRC, 2006).
Instituting funding requirements has been an effective mechanism for advancing practices that sponsors believe will benefit research and improve the consistency of practice across institutions and studies. For example, recognizing that effective stakeholder engagement requires careful advance planning, PCORI requires that all funding applications include an engagement plan, which is evaluated in the application review process (PCORI, 2015). Beyond promoting advance planning, the funding application review process provides an important opportunity to ensure that key considerations for the responsible return of individual research results (e.g., laboratory quality systems and approaches to assessing participant needs, preferences, and values) have been attended to and, in the case that investigators plan to return results to participants, that there is an appropriate budget to cover the expected costs. By allowing investigators to budget for the return of individual research results in funding applications, research sponsors can send a powerful message in support of a new research paradigm in which results are more routinely returned to participants.
CONCLUSION: By requiring, reviewing, and supporting return-of-results plans, research sponsors can foster a culture in which the return of individual research results is more routinely considered and conducted.
The Role of IRBs in Reviewing Plans for the Return of Individual Research Results
IRBs play a critical role in human biomedical research. Given their core responsibility within research institutions for protecting the rights and welfare of participants, IRBs are well positioned to help investigators consider the return of individual research results in the development of research protocols (Dressler et al., 2012). In surveys and interviews, IRB professionals have generally held the view that investigators should anticipate the return of results prior to a study’s implementation and that the issue of returning individual research results should be covered in the IRB application and informed consent document for IRB review and approval (Beskow and O’Rourke, 2015; Dressler et al., 2012; Ohayon et al., 2017; Williams et al., 2012). The IRB could then “ensure that an appropriate, ethical process is followed for making decisions and communicating with participants” (Beskow and O’Rourke, 2015, p. 7).
IRBs are more hesitant about being tasked with determining whether specific research results meet the criteria for return to participants and feel that their more appropriate role is to oversee the process for decision making (Beskow and O’Rourke, 2015; Dressler et al., 2012). Many IRB professionals expressed concerns about a lack of scientific (e.g., genomic) and medical knowledge that would be needed to make sound decisions and provide well-informed guidance to investigators based on the expected reliability and value of the result and the potential risks to the participants from returning it (Dressler et al., 2012). The necessary scientific expertise, however, need not always reside within the IRB membership. IRBs may have to rely on additional outside expertise—for example, by developing more involved partnerships with investigators, clinicians, participants, or community or patient advocacy groups or through using scientific review or community advisory boards (see Box 4-6)—to bolster their scientific knowledge base and understanding of participant perspectives so as not to be paternalistic in their decisions. Such institutional advisory bodies can also serve as resources investigators, providing direct guidance concerning the return of individual research results. The IRB will also have to take the context of the study into consideration when making decisions about investigators’ plans for return. This is particularly the case in situations where the investigator is also the physician or caregiver for the participant, including in situations where the investigator is only transiently responsible for care in the context of the research, as this might influence the laboratory environments in which the research tests are conducted.
While recognizing the importance of having policies and procedures in place, few IRBs describe having actual experiences with the return of individual research results (Dressler et al., 2012; Williams et al., 2012), making it challenging to develop institutional policies and procedures. This gap in IRB experience will necessitate additional support for IRBs, including education and training as well as guidance (Beskow and O’Rourke, 2015; Dressler et al., 2012; Fabsitz
et al., 2010). Such guidance should include providing a guiding framework or “ground rules” while preserving the flexibility of IRBs to make decisions based on study and community context (Dressler et al., 2012; Fabsitz et al., 2010). Training exercises could be developed to allow IRBs and researchers to practice making contextual decisions based on case scenarios, and they would also help IRBs, institutions, and investigators better understand and plan for the logistics, infrastructure, and resources needed to support the return of individual research results (Dressler et al., 2012).
In the near term, this lack of experience with the return of individual research results may result in some reservations and a conservative approach (Ohayon et al., 2017). As with similar surveys of investigators, the positions of IRB professionals on returning individual research results have varied (Dressler et al., 2012;
Ohayon et al., 2017). On balance, however, these professionals expressed general support for returning validated individual research results, particularly when such results have clinical significance, are medically actionable, or have personal utility to participants (Dressler et al., 2012). In one survey, IRB chairs noted the “potential to overstate the risks of possible . . . disclosure, and talked of the difficulty of documenting the potential for harm” (Simon et al., 2011, p. 8). As researchers and IRBs gain more experience with returning individual research results, and as a stronger empirical evidence base on the benefits and risks is developed, IRBs may feel more comfortable with providing guidance and may become stronger proponents for expanding the practice of returning research results. In the meantime, interfacing with researchers who have experience returning individual research results may help IRBs develop and consider experience-based perspectives (Ohayon et al., 2017).
CONCLUSION: Some IRBs currently lack sufficient scientific and participant engagement expertise to assess research design and the likely validity, actionability, or utility of research results. IRBs will need additional expertise from an IRB–researcher partnership or outside input from scientific review committees and participant, community, or patient advocacy groups to help inform responsible and feasible institutional approaches to offering and returning research results. IRBs may also require input from engagement specialists and community advisors in order to acquire the expertise necessary to evaluate whether participant preferences have been appropriately considered in planning for the return of individual research results.
CONCLUSION
The return of individual research results will create new demands on the research enterprise. The committee recognizes that many institutions and researchers currently lack the experience and resources to return individual research results appropriately and that balancing the validity of the results (discussed in Chapter 3) with participant preferences, needs, and values will not be an easy task. However, the committee emphasizes the need to consider participant preferences while ensuring the validity of the results so that only high-quality results are returned to participants with the appropriate information. The committee does not expect that the routine and widespread return of individual research results will begin overnight, but it does foresee an evolving set of responsibilities. The recommendations in this report are intended to help stakeholders discuss and prepare for these responsibilities in order to develop the necessary expertise, infrastructure, and resources over time. As that capacity is being developed, a tiered approach to implementation may be prudent for the operational reasons discussed in Chapters 3, 4, and 5 as well as for the legal reasons discussed in Chapter 6. As discussed earlier in this chapter, some institutions (e.g., those with CTSAs) have existing infrastructure and resources that enable them to serve as early adopters. While a tiered approach may support important learning processes, it will be important to ensure that mechanisms are put in place to assist those currently lacking the needed infrastructure and expertise so that this new set of responsibilities is not perceived as an unfunded mandate.
REFERENCES
Ahmed, S. M., and A.-G. S. Palermo. 2010. Community engagement in research: Frameworks for education and peer review. American Journal of Public Health 100(8):1380–1387.
Anastasova, V., A. Mahalatchimy, E. Rial-Sebbag, J. M. Anto Boque, T. Keil, J. Sunyer, J. Bousquet, and A. Cambon-Thomsen. 2013. Communication of results and disclosure of incidental findings in longitudinal paediatric research. Pediatric Allergy and Immunology 24(4):389–394.
Arora, N. K., and C. A. McHorney. 2000. Patient preferences for medical decision making: Who really wants to participate? Medical Care 38(3):335–341.
Avard, D., T. Silverstein, G. Sillon, and Y. Joly. 2009. Researchers’ perceptions of the ethical implications of pharmacogenomics research with children. Public Health Genomics 12(3):191–201.
Battistuzzi, L., R. Ciliberti, W. Bruno, D. Turchetti, L. Varesco, and F. De Stefano. 2013. Communication of clinically useful next-generation sequencing results to at-risk relatives of deceased research participants: Toward active disclosure? Journal of Clinical Oncology 31(32):4164–4165.
Bergman, A., J. J. Heindel, S. Jobling, K. A. Kidd, and R. T. Zoeller. 2012. State of the science of endocrine disrupting chemicals 2012: Summary for decision-makers. Geneva, Switzerland: United Nations Environment Programme and the World Health Organization.
Beskow, L. M., and W. Burke. 2010. Offering individual genetic research results: Context matters. Science Translational Medicine 2(38):1–5.
Beskow, L. M., and P. P. O’Rourke. 2015. Return of genetic research results to participants and families: IRB perspectives and roles. Journal of Law, Medicine & Ethics 43(3):502–513.
Black, K. Z., C. Y. Hardy, M. De Marco, A. S. Ammerman, G. Corbie-Smith, B. Council, D. Ellis, E. Eng, B. Harris, M. Jackson, J. Jean-Baptiste, W. Kearney, M. Legerton, D. Parker, M. Wynn, and A. Lightfoot. 2013a. Beyond incentives for involvement to compensation for consultants: Increasing equity in CBPR approaches. Progress in Community Health Partnerships: Research, Education, and Action 7(3):263–270.
Black, L., D. Avard, M. H. Zawati, B. M. Knoppers, J. Hébert, and G. Sauvageau. 2013b. Funding considerations for the disclosure of genetic incidental findings in biobank research. Clinical Genetics 84(5):397–406.
Bollinger, J. M., J. F. P. Bridges, A. Mohamed, and D. Kaufman. 2014. Public preferences for the return of research results in genetic research: A conjoint analysis. Genetics in Medicine 16(12):932–939.
Bookman, E. B., A. A. Langehorne, J. H. Eckfeldt, K. C. Glass, G. P. Jarvik, M. Klag, G. Koski, A. Motulsky, B. Wilfond, T. A. Manolio, R. R. Fabsitz, and R. V. Luepker. 2006. Reporting genetic results in research studies: Summary and recommendations of an NHLBI working group. American Journal of Medical Genetics, Part A 140(10):1033–1040.
Boronow, K. E., H. P. Susmann, K. Z. Gajos, R. A. Rudel, K. C. Arnold, P. Brown, R. Morello-Frosch, L. Havas, and J. G. Brody. 2017. Derbi: A digital method to help researchers offer “right-to-know” personal exposure results. Environmental Health Perspectives 125(2):A27–A33.
Bredenoord, A. L., H. Y. Kroes, E. Cuppen, M. Parker, and J. J. M. van Delden. 2011. Disclosure of individual genetic data to research participants: The debate reconsidered. Trends in Genetics 27(2):41–47.
Brody, J. G., S. C. Dunagan, R. Morello-Frosch, P. Brown, S. Patton, and R. A. Rudel. 2014. Reporting individual results for biomonitoring and environmental exposures: Lessons learned from environmental communication case studies. Environmental Health 13:40.
Brown, P., R. Morello-Frosch, J. G. Brody, R. G. Altman, R. A. Rudel, L. Senier, C. Pérez, and R. Simpson. 2010. Institutional review board challenges related to community-based participatory research on human exposure to environmental toxins: A case study. Environmental Health 9:39.
Budin-Ljøsne, I., D. Mascalzoni, S. Soini, H. Machado, J. Kaye, H. B. Bentzen, E. Rial-Sebbag, F. D’Abramo, M. Witt, G. Schamps, V. Katic, D. Krajnovic, and J. R. Harris. 2016. Feedback of individual genetic results to research participants: Is it feasible in Europe? Biopreservation and Biobanking 14(3):241–248.
Bunnik, E., A. C. J. W. Janssens, and M. Schermer. 2014. Personal utility in genomic testing: Is there such a thing? Journal of Medical Ethics 41(4):322–326.
Cadigan, R. J., M. Michie, G. Henderson, A. M. Davis, and L. M. Beskow. 2011. The meaning of genetic research results: Reflections from individuals with and without a known genetic disorder. Journal of Empirical Research on Human Research Ethics 6(4):30–40.
Chan, B., F. M. Facio, H. Eidem, S. C. Hull, L. G. Biesecker, and B. E. Berkman. 2012. Genomic inheritances: Disclosing individual research results from whole-exome sequencing to deceased participants’ relatives. American Journal of Bioethics 12(10):1–8.
Christensen, K. D., J. S. Roberts, D. I. Shalowitz, J. N. Everett, S. Y. Kim, L. Raskin, and S. B. Gruber. 2011. Disclosing individual CDKN2A research results to melanoma survivors: Interest, impact, and demands on researchers. Cancer Epidemiology, Biomarkers & Prevention 20(3):522–529.
ClinGen. 2018. ClinGen genome dosage map. https://www.ncbi.nlm.nih.gov/pubmed (accessed May 18, 2018).
CSER (Clinical Sequencing Exploratory Research) Consortium. 2018. Clinical sequencing evidence-generating research. https://cser-consortium.org (accessed May 18, 2018).
CTSA (Clinical and Translational Science Awards) Community Engagement Key Function Committee Task Force on the Principles of Community Engagement. 2011. Principles of community engagement. Bethesda, MD: NIH.
Diamanti-Kandarakis, E., J.-P. Bourguignon, L. C. Giudice, R. Hauser, G. S. Prins, A. M. Soto, R. T. Zoeller, and A. C. Gore. 2009. Endocrine-disrupting chemicals: An Endocrine Society scientific statement. Endocrine Reviews 30(4):293–342.
Dressler, L. G., S. Smolek, R. Ponsaran, J. M. Markey, H. Starks, N. Gerson, S. Lewis, N. Press, E. Juengst, and G. L. Wiesner. 2012. IRB perspectives on the return of individual results from genomic research. Genetics in Medicine 14(2):215–222.
Epstein, R. M., and R. L. Street, Jr. 2011. The values and value of patient-centered care. Annals of Family Medicine 9(2):100–103.
Epstein, R. M., K. Fiscella, C. S. Lesser, and K. C. Stange. 2010. Why the nation needs a policy push on patient-centered health care. Health Affairs (Project Hope) 29(8):1489–1495.
Fabsitz, R. R., A. McGuire, R. R. Sharp, M. Puggal, L. M. Beskow, L. G. Biesecker, E. Bookman, W. Burke, E. G. Burchard, G. Church, E. W. Clayton, J. H. Eckfeldt, C. V. Fernandez, R. Fisher, S. M. Fullerton, S. Gabriel, F. Gachupin, C. James, G. P. Jarvik, R. Kittles, J. R. Leib, C. O’Donnell, P. P. O’Rourke, L. L. Rodriguez, S. D. Schully, A. R. Shuldiner, R. K. Sze, J. V. Thakuria, S. M. Wolf, G. L. Burke, and National Heart, Lung, and Blood Institute Working Group. 2010. Ethical and practical guidelines for reporting genetic research results to study participants: Updated guidelines from a National Heart, Lung, and Blood Institute working group. Circulation: Cardiovascular Genetics 3(6):574–580.
Fernandez, C. V., C. Skedgel, and C. Weijer. 2004. Considerations and costs of disclosing study findings to research participants. Canadian Medical Association Journal 170(9):1417–1419.
Goodman, J. L., L. M. Amendola, M. Horike Pyne, S. B. Trinidad, S. M. Fullerton, W. Burke, and G. P. Jarvik. 2017. Discordance in selected designee for return of genomic findings in the event of participant death and estate executor. Molecular Genetics & Genomic Medicine 5(2):172–176.
Green, R. C., J. S. Berg, W. W. Grody, S. S. Kalia, B. R. Korf, C. L. Martin, A. L. McGuire, R. L. Nussbaum, J. M. O’Daniel, K. E. Ormond, H. L. Rehm, M. S. Watson, M. S. Williams, and L. G. Biesecker. 2013. ACMG recommendations for reporting of incidental findings in clinical exome and genome sequencing. Genetics in Medicine 15(7):565–574.
Groisman, I. J., G. Mathieu, and B. Godard. 2012. Use of next generation sequencing technologies in research and beyond: Are participants with mental health disorders fully protected? BMC Medical Ethics 13:36.
Guyatt, G., V. Montori, P. J. Devereaux, H. Schünemann, and M. Bhandari. 2004. Patients at the center: In our practice, and in our use of language. ACP Journal Club 140(1):A11–A12.
Haines, D. A., T. E. Arbuckle, E. Lye, M. Legrand, M. Fisher, R. Langlois, and W. Fraser. 2011. Reporting results of human biomonitoring of environmental chemicals to study participants: A comparison of approaches followed in two Canadian studies. Journal of Epidemiology and Community Health 65(3):191–198.
Halverson, C. M. E., and L. F. Ross. 2012. Attitudes of African American parents about biobank participation and return of results for themselves and their children. Journal of Medical Ethics 38(9):561–566.
Heaney, C., G. Tindall, J. Lucas, and S. B. Haga. 2010. Researcher practices on returning genetic research results. Genetic Testing & Molecular Biomarkers 14(6):821–827.
Holm, I. A., S. K. Savage, R. C. Green, E. Juengst, A. McGuire, S. Kornetsky, S. J. Brewster, S. Joffe, and P. Taylor. 2014. Guidelines for return of research results from pediatric genomic studies: Deliberations of the Boston Children’s Hospital Gene Partnership Informed Cohort Oversight Board. Genetics in Medicine 16(7):547–552.
IOM (Institute of Medicine). 2016. Biomarker tests for molecularly targeted therapies: Key to unlocking precision medicine. Washington, DC: The National Academies Press.
Israel, B. A., A. J. Schulz, E. A. Parker, and A. B. Becker. 1998. Review of community-based research: Assessing partnership approaches to improve public health. Annual Review of Public Health 19(1):173–202.
Jarvik, G. P., L. M. Amendola, J. S. Berg, K. Brothers, E. W. Clayton, W. Chung, B. J. Evans, J. P. Evans, S. M. Fullerton, C. J. Gallego, N. A. Garrison, S. W. Gray, I. A. Holm, I. J. Kullo, L. S. Lehmann, C. McCarty, C. A. Prows, H. L. Rehm, R. R. Sharp, J. Salama, S. Sanderson, S. L. Van Driest, M. S. Williams, S. M. Wolf, W. A. Wolf, eMERGE Act–ROR Committee, CERC Committee, CSER Act–ROR Working Group, and W. Burke. 2014. Return of genomic results to research participants: The floor, the ceiling, and the choices in between. American Journal of Human Genetics 94(6):818–826.
Joosten, Y. A., T. L. Israel, N. A. Williams, L. R. Boone, D. G. Schlundt, C. P. Mouton, R. S. Dittus, G. R. Bernard, and C. H. Wilkins. 2015. Community engagement studios: A structured approach to obtaining meaningful input from stakeholders to inform research. Academic Medicine 90(12):1646–1650.
Lange, M. M., W. Rogers, and S. Dodds. 2013. Vulnerability in research ethics: A way forward. Bioethics 27(6):333–340.
Little, P., H. Everitt, I. Williamson, G. Warner, M. Moore, C. Gould, K. Ferrier, and S. Payne. 2001. Preferences of patients for patient centred approach to consultation in primary care: Observational study. British Medical Journal 322(7284):468–472.
Middleton, A., K. I. Morley, E. Bragin, H. V. Firth, M. E. Hurles, C. F. Wright, M. Parker, and Deciphering Developmental Disorders Study. 2016. Attitudes of nearly 7,000 health professionals, genomic researchers and publics toward the return of incidental results from sequencing research. European Journal of Human Genetics 24(1):21–29.
Mjoseth, J. 2012. NHGRI funds return of results studies, forms expert consortium. https://www.genome.gov/27545526/2011-news-feature-nhgri-funds-return-of-results-studies-forms-expertconsortium/2011-news-feature-nhgri-funds-return-of-results-studies-forms-expert-consortium (accessed May 18, 2018).
Mott, L., and E. Crawford. 2008. The role of community advisory boards in project Eban. Journal of Acquired Immune Deficiency Syndromes 49(Suppl 1):S68–S74.
MRCT (Multi-Regional Clinical Trials) Center. 2017. Return of individual results to participants recommendations document. Boston, MA: MRCT Center.
Murphy, J., J. Scott, D. Kaufman, G. Geller, L. LeRoy, and K. Hudson. 2008. Public expectations for return of results from large-cohort genetic research. American Journal of Bioethics 8(11):36–43.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2017. An evidence framework for genetic testing. Washington, DC: The National Academies Press.
National Bioethics Advisory Commission. 1999. Research involving human biological materials: Ethical issues and policy guidance, volume 1. Rockville, MD: U.S. Government Printing Office.
NCATS (National Center for Advancing Translational Science). 2018. About the CTSA program. https://ncats.nih.gov/ctsa/about (accessed May 18, 2018).
NCI (National Cancer Institute). 2016. NCI best practices for biospecimen resources.https://biospecimens.cancer.gov/bestpractices/2016-NCIBestPractices.pdf (accessed May 22, 2018).
Newman, S. D., J. O. Andrews, G. S. Magwood, C. Jenkins, M. J. Cox, and D. C. Williamson. 2011. Community advisory boards in community-based participatory research: A synthesis of best processes. Preventing Chronic Disease 8(3):A70.
NHGRI (National Human Genome Research Institute). 2017a. Points to consider regarding the Food and Drug Administration’s investigational device exemption regulations in the context of genomics research. https://www.genome.gov/27561291/points-to-consider-in-assessing-when-an-investigational-device-exemption-ide-might-be-needed (accessed March 28, 2018).
NHGRI. 2017b. Return of research results. https://www.genome.gov/27569049/return-of-research-results (accessed May 22, 2018).
NRC (National Research Council). 2006. Human biomonitoring for environmental chemicals. Washington, DC: The National Academies Press.
O’Fallon, L. R., and A. Dearry. 2002. Community-based participatory research as a tool to advance environmental health sciences. Environmental Health Perspectives 110(Suppl 2):155–159.
Ohayon, J. L., E. Cousins, P. Brown, R. Morello-Frosch, and J. G. Brody. 2017. Researcher and institutional review board perspectives on the benefits and challenges of reporting back biomonitoring and environmental exposure results. Environmental Research 153:140–149.
Paberzs, A., P. Piechowski, D. Warrick, C. Grawi, C. Choate, G. Sneed, D. Carr, K. Lota, K. Key, V. Alexander, P. Ghosh, and C. Sampselle. 2014. Strengthening community involvement in grant review: Insights from the Community–University Research Partnership (CURES) pilot review process. Clinical and Translational Science 7(2):156–163.
PCORI (Patient-Centered Outcomes Research Institute). 2015. What we mean by engagement. https://www.pcori.org/engagement/what-we-mean-engagement (accessed March 27, 2018).
PCORI. 2018. Engagement in health research literature explorer. https://www.pcori.org/literature/engagement-literature (accessed February 5, 2018).
PCSBI (Presidential Commission for the Study of Bioethical Issues). 2013. Anticipate and communicate ethical management of incidental and secondary findings in the clinical, research, and direct to consumer contexts. Washington, DC: Presidential Commission for the Study of Bioethical Issues.
Ravitsky, V., and B. S. Wilfond. 2006. Disclosing individual genetic results to research participants. American Journal of Bioethics 6(6):8–17.
Resnik, D. B. 2011. Disclosure of individualized research results: A precautionary approach. Accountability in Research 18(6):382–397.
Rigby, H., and C. V. Fernandez. 2005. Providing research results to study participants: Support versus practice of researchers presenting at the American Society of Hematology annual meeting. Blood 106(4):1199–1202.
Rothstein, M. A. 2006. Tiered disclosure options promote the autonomy and well-being of research subjects. The American Journal of Bioethics 6(6):20–21.
SACHRP (Secretary’s Advisory Committee on Human Research Protections). 2009. Attachment: Recommendations regarding research involving individuals with impaired decision-making. https://www.hhs.gov/ohrp/sachrp-committee/recommendations/2009-july-15-letter-attachment/index.html (accessed May 18, 2018).
SACHRP. 2016. Attachment B: Return of individual research results. https://www.hhs.gov/ohrp/sachrp-committee/recommendations/attachment-b-return-individual-research-results/index.html (accessed July 13, 2017).
Sawyer, S. L., T. Hartley, D. A. Dyment, C. L. Beaulieu, J. Schwartzentruber, A. Smith, H. M. Bedford, G. Bernard, F. P. Bernier, B. Brais, D. E. Bulman, J. Warman Chardon, D. Chitayat, J. Deladoëy, B. A. Fernandez, P. Frosk, M. T. Geraghty, B. Gerull, W. Gibson, R. M. Gow, G. E. Graham, J. S. Green, E. Heon, G. Horvath, A. M. Innes, N. Jabado, R. H. Kim, R. K. Koenekoop, A. Khan, O. J. Lehmann, R. Mendoza Londono, J. L. Michaud, S. M. Nikkel, L. S. Penney, C. Polychronakos, J. Richer, G. A. Rouleau, M. E. Samuels, V. M. Siu, O. Suchowersky, M. A. Tarnopolsky, G. Yoon, F. R. Zahir, FORGE Canada Consortium, Care4Rare Canada Consortium, J. Majewski, and K. M. Boycott. 2016. Utility of whole exome sequencing for those near the end of the diagnostic odyssey: Time to address gaps in care. Clinical Genetics 89(3):275–284.
Saxton, D. I., P. Brown, S. Seguinot-Medina, L. Eckstein, D. O. Carpenter, P. Miller, and V. Waghiyi. 2015. Environmental health and justice and the right to research: Institutional review board denials of community-based chemical biomonitoring of breast milk. Environmental Health 14:90.
Simon, C. M., J. K. Williams, L. Shinkunas, D. Brandt, S. Daack-Hirsch, and M. Driessnack. 2011. Informed consent and genomic incidental findings: IRB chair perspectives. Journal of Empirical Research on Human Research Ethics 6(4):53–67.
Tassé, A. M. 2011. The return of results of deceased research participants. The Journal of Law, Medicine & Ethics 39(4):621–630.
Terry, S. F. 2012. The tension between policy and practice in returning research results and incidental findings in genomic biobank research. Minnesota Journal of Law, Science & Technology 13(2):691–736.
Teutsch, S. M., L. A. Bradley, G. E. Palomaki, J. E. Haddow, M. Piper, N. Calonge, W. D. Dotson, M. P. Douglas, and A. O. Berg. 2009. The Evaluation of Genomic Applications in Practice and Prevention (EGAPP) initiative: Methods of the EGAPP working group. Genetics in Medicine 11(1):3–14.
Wallerstein, N. B., and B. Duran. 2006. Using community-based participatory research to address health disparities. Health Promotion Practice 7(3):312–323.
Weiner, C. 2014. Anticipate and communicate: Ethical management of incidental and secondary findings in the clinical, research, and direct-to-consumer contexts (December 2013 report of the Presidential Commission for the Study of Bioethical Issues). American Journal of Epidemiology 180(6):562–564.
Wilkins, C. H., M. Spofford, N. Williams, C. McKeever, S. Allen, J. Brown, J. Opp, A. Richmond, and A. H. Strelnick. 2013. Community representatives’ involvement in clinical and translational science awardee activities. Clinical and Translational Science 6(4):292–296.
Williams, J. K., S. Daack-Hirsch, M. Driessnack, N. Downing, L. Shinkunas, D. Brandt, and C. Simon. 2012. Researcher and institutional review board chair perspectives on incidental findings in genomic research. Genetic Testing and Molecular Biomarkers 16(6):508–513.
Wolf, S. M., F. P. Lawrenz, C. A. Nelson, J. P. Kahn, M. K. Cho, E. W. Clayton, J. G. Fletcher, M. K. Georgieff, D. Hammerschmidt, K. Hudson, J. Illes, V. Kapur, M. A. Keane, B. A. Koenig, B. S. Leroy, E. G. McFarland, J. Paradise, L. S. Parker, S. F. Terry, B. Van Ness, and B. S. Wilfond. 2008. Managing incidental findings in human subjects research: Analysis and recommendations. The Journal of Law, Medicine & Ethics 36(2):219–248.
Wolf, S. M., B. N. Crock, B. Van Ness, F. Lawrenz, J. P. Kahn, L. M. Beskow, M. K. Cho, M. F. Christman, R. C. Green, R. Hall, J. Illes, M. Keane, B. M. Knoppers, B. A. Koenig, I. S. Kohane, B. Leroy, K. J. Maschke, W. McGeveran, P. Ossorio, L. S. Parker, G. M. Petersen, H. S. Richardson, J. A. Scott, S. F. Terry, B. S. Wilfond, and W. A. Wolf. 2012. Managing incidental findings and research results in genomic research involving biobanks and archived data sets. Genetics in Medicine 14(4):361–384.
Wolf, S. M., R. Branum, B. A. Koenig, G. M. Petersen, S. A. Berry, L. M. Beskow, M. B. Daly, C. V. Fernandez, R. C. Green, B. S. LeRoy, N. M. Lindor, P. P. O’Rourke, C. R. Breitkopf, M. A. Rothstein, B. Van Ness, and B. S. Wilfond. 2015. Returning a research participant’s genomic results to relatives: Analysis and recommendations. The Journal of Law, Medicine & Ethics 43(3):440–463.
This page intentionally left blank.










































