4
State of the Science on TMDs
What I want to see going forward is research on just what temporomandibular disorders are—research that validates the safety and effectiveness of every treatment for TMDs. I want research on every aspect of a TMJ device and related patient care. I want better practices and protocols for all stages of TMJ surgical procedures and a formal collaboration with the musculoskeletal branches in medicine. There is no reason we should not have the research on the TM joint that exists on every other joint in the body.
—Lutricia M.
Despite investment in research directly and indirectly related to temporomandibular disorders (TMDs)—most significantly in the field of orofacial pain—researchers have yet to unravel the etiologies and pathophysiologies of TMDs or to translate, in a meaningful way, research findings into improved clinical care practices. Over the past decade the body of research on TMDs centered on the biological mechanisms underlying the development and persistence of orofacial pain and on the structure and function of the joint and its tissues, while more recent research has begun to examine the molecular genetics, biomarkers, and biopsychosocial risk factors of TMDs and common comorbidities. Broadly, the research foundation relating to TMDs, as has been the case with other complex, stigmatized conditions, has suffered from the siloing of disciplines and from a lack of clear direction—thus stunting the potential clinical impact of the research. In the case of TMDs, these difficulties have been heightened by a significant
dental–medical divide that affects both research and clinical care. Fundamentally, the development of safe and effective treatments and therapies for TMDs necessitates the existence of a robust research base spanning multiple disciplines—a foundation that is currently lacking in many areas.
This chapter provides the reader with an overview of recent basic science research related to the pathophysiologies of TMDs and orofacial pain, with a primary focus on the state of the evidence as it relates to the development and growth of temporomandibular joint (TMJ) tissues in health and disease and the mechanisms underpinning pain and tissue dysfunction. This chapter is not intended to be an exhaustive systematic review of all of the scientific evidence available on TMDs or a clinical review; rather, the chapter highlights significant research gaps identified by the committee that need to be addressed to develop a robust evidence base for TMDs. Following an overview of the state of the science and a discussion of overarching research gaps and priorities—primarily focused on the scarcity of multidisciplinary research efforts dedicated to understanding TMDs from the cellular to the societal level—the chapter explores the organizational, financial, and cultural changes within the TMD research enterprise that will be necessary for the development of a research enterprise that prioritizes the needs of the patient. The chapter will conclude by exploring how a patient-centered research framework could be structured to address the short- and long-term research priorities identified throughout this report. Additional evidence and research priorities related to TMD epidemiology, clinical care, and education and training will be covered in greater detail in their respective chapters.
RECENT BIOMECHANICAL AND BIOENGINEERING RESEARCH ON THE TMJ
As detailed in Chapter 2 and Appendix D, the TMJ, or jaw joint, has a unique morphology and function. This complex joint is composed of bone and muscle juxtaposed to cartilaginous tissues that allow for translational and rotational movements of the mandible. The major components of the TMJ include the mandibular condyle, glenoid fossa of the temporal bone, articular disc, synovial fluid encapsulated by the fibrous joint capsule, adjacent ligaments, and retrodiscal tissue (see Figure D-1 in Appendix D). TMJ movement is maintained by the synchronous coordination of a group of bilateral muscles, and TMJ movement is limited by TMJ ligament range. Sensory innervation of the joint is derived from the mandibular branch of the trigeminal nerve, which also provides innervation to the face and mouth (Sessle, 2011). The surrounding tissues are innervated by proprioceptive and mechanosensitive nerve endings, which are important in initiating movement and controlling the mechanics of the joint, as well
as by nociceptive afferents, which are important for transmitting noxious stimuli to the central nervous system (CNS) (Sessle, 2011).
The articular disc within the joint is composed of fibrocartilage—type I and type II collagens—which provides both strength and flexibility. These tissues have only limited regenerative capacity (Roberts and Stocum, 2018); therefore, to support the high cell density in the disc and metabolically demanding joint function, the synovial fluid within the joint provides essential nutrients and lubrication. Without this the increased friction and shear stress would lead to disc degeneration (Wang et al., 2015; Wu et al., 2019). The TMJ is subjected to mechanical loading from use of the jaw, which is critical for cartilage and bone maintenance and ongoing tissue remodeling during growth and repair. However, when the mechanical loads on the TMJ are too large or too frequent, both cartilage and bone tissues may be compromised, and irreversible damage may ensue (Iwasaki et al., 2017; Nickel et al., 2018).
Neuromuscular and musculoskeletal disorders that affect TMJ and jaw function are common conditions (see Chapter 3). Despite this, the TMJ has not been well studied compared to other synovial joint systems, such as the knee, shoulder, and hip joints (Hinton, 2014). The study of the joint is complicated by its complex anatomy, which has a unique fibrocartilage structure and dense neurovascular system (blood vessels and nerves). Together, these complexities impede in vivo experimental measurements of the TMJ components’ articular space, contact forces, stress distribution, and nutrient supply (Nickel et al., 2018). These measurement properties, however, are a key to defining TMJ physiology and pathophysiology, such as wear, fatigue, and degeneration mechanisms.
Developmental Biology and Physiology of the TMJ in Health and Disease
The developmental biology and physiology of the TMJ are different in many ways from those of the limb joints (e.g., knee, shoulder, and hip). Understanding the unique aspects of the joint’s development, postnatal growth, and function are foundational to gaining a comprehensive clinical understanding of the joint and its pathologies. There are several major differences between the TMJ and the other synovial joints. First, the fibrocartilage of the TMJ develops from the neural crest, whereas the hyaline cartilage of the limb joints develops from mesoderm (Somoza et al., 2014). Second, the bony aspects of the TMJ—mandibular condyles and temporal eminences—are formed from secondary cartilage, whereas the limb joints and long bones are formed from primary cartilage (Hinton, 2014). Third, the fibrocartilage of the mandibular condylar and temporal eminence provides the unique and important hybrid physiological functions of both articulation and growth, whereas in the limbs, these two
functions are provided separately—by articular hyaline cartilage on joint surfaces and by the hyaline cartilage of the growth plate in long bones, respectively (Stocum and Roberts, 2018). Fourth, while the mandibular condyle and both sides of limb joints can form de novo, the temporal eminence of the TMJ does not appear de novo (Stocum and Roberts, 2018). Instead, both the mandibular condyle and the temporal eminence require mechanical loading to persist. Fifth, unlike the articular surfaces of limb joints, which are lined by hyaline cartilage, the articular surface of the TMJ consists of fibrocartilage and is unique in that it contains both types I and II collagen. Articular hyaline cartilage does not typically contain type I collagen (Wadhwa and Kapila, 2008). This organization of collagen fiber alignment and type, which provides the TMJ with the functional capacity to withstand tensile loading better than hyaline cartilage, highlights a key distinction in the demands on TMJ function versus that of other joints. Finally, while the TMJ development and growth are regulated by some of the same genes as those in limb joints, other genes have been identified as unique to TMJ development or have different actions (Hinton et al., 2015).
Genetic Influences on TMJ Development, Growth, and Degeneration
The unique origin of the TMJ’s skeletal and connective tissues from neural crest cells rather than from the mesoderm indicates the involvement of different genetic drivers and pathways for the joint’s development and growth (Hinton, 2014). Understanding the role and interactions of these factors in driving morphological variations of the TMJ provides insight into the potential etiologies of certain TMDs and identifies potential future targets for regenerative medicine. The roles of specific genes in driving the growth of mandibular condylar cartilage, the mandibular fossa, and the articular disc and joint cavitation have been studied to varying degrees using animal models, with two comprehensive reviews recently completed by Hinton (2014) and Scariot and collegues (2018). This body of research has identified various genes, such as Runx2, Sox9, BMPr1a, and members of the TGF-β/BMP family, as individual drivers of chondrogenesis (Hinton, 2014). Beyond these potential drivers of chondrogenesis, it is thought that other genes may act as mediators within the morphogenic pathway, such as Indian hedgehog (Ihh), which plays a role in the formation of the articular disc and cavitation (Gu et al., 2014; Hinton, 2014; Hinton et al., 2015), and Osterix (Osx), which may play a part in the regulation of bone formation during postnatal TMJ growth (Jing et al., 2014).
The morphogenesis of the secondary cartilage found in the TMJ has also been a major focus of research in TMJ developmental biology, as it is the most important component of the development hierarchy of the joint tissues (Hinton, 2014). Hinton and colleagues (2015) write, “Recent studies
of the genetic regulation of the TMJ morphogenesis and growth (and lately of its degradation) have identified a patchwork of interacting and hierarchical players that usually but not always resemble those that we know from limb cartilages.” However, the innovation of bioengineering techniques to repair or replace damaged tissues will require additional systematic review of these and additional genes to truly understand the genetic regulation of TMJ development and its role in the health and diseases of the joint (Hinton et al., 2015). Studies using mouse models have indicated that the formation of the fibrocartilage of the mandibular condyle is regulated by Runx2 and Sox9 (Shibata et al., 2004; Oka et al., 2008; Hinton, 2014), with their inactivation resulting in agenesis of condylar cartilage (Mori-Akiyama et al., 2003; Shibata et al., 2004; Wang et al., 2011). Certain members of the TGF-β family are also thought to play a role in the formation of condylar cartilage by interfering with the regulation of Runx2 and Sox9 (Oka et al., 2008). Other significant findings include the potential role of genes like Ihh, Shox2, and Trps in secondary cartilage formation, with studies showing that mice that lack these genes present with delayed or impaired condylar cartilage and disruption in the formation of the articular disc and cavitation (Wang et al., 2011; Gu et al., 2014; Hinton, 2014; Ishizuka et al., 2014). Other genes that have been studied that are involved in the formation of secondary cartilage, mandibular fossa, and the articular disc in the TMJ are listed in Table 4-1.
In the case of TMDs, which tend to appear later in life rather than to present congenitally, a deep understanding of the postnatal roles of genes in TMJ tissue development and the role of genes in regulating degenerative changes will be critical to achieving future clinical applications. Certain genes, such as Ihh, may also play a significant role in postnatal TMJ growth. In a study by Ochiai and colleagues (2010), Ihh knockout mice showed disrupted mandibular condylar architecture and reduced Sox9 and Col2 expression. Additionally, the postnatal interruption of BMPr1a has been shown in mouse models to negatively affect the length of the mandible and the presence of condylar cartilage (Jing et al., 2014). As for the degradation of the articular disc, genes Fgfr3 and Prg4 may play a regulatory role (Yasuda et al., 2012; Koyama et al., 2014; Komori, 2020). Other potentially significant genes and pathways of recent study include fibroblast growth factors, Sprouty genes (Purcell et al., 2012), parathyroid hormone–related protein feedback loops, and BMPr1a, among others (see Table 4-1).
The etiology of TMJ degeneration is complex and involves alterations in the functional environment of the joint (such as changes to mechanical loading or trauma) and in biological mechanisms that trigger responses. The drivers and pathways of TMJ tissue degeneration have been a focus of recent studies using animal models. In one study using osteoarthritic-prone SAMP8 mice, Ihh expression was significantly reduced, as was
TABLE 4-1 Recent Studies Related to Potential Drivers and Pathways of TMJ Tissue Formation
| Driver or Pathway of Study | Author and Year |
|---|---|
| Wnt and Nf-κB pathways | de Sousa et al., 2019 |
| Ihh pathway, MMP-13, caspase-3 | Long et al., 2019 |
| Runx2 | Liao et al., 2019 |
| TNF-β | Yerliyurt et al., 2019 |
| Axin1, Wnt and FGF pathway | Zhou et al., 2019 |
| TGF-β pathway, Col X, Osterix | Zheng et al., 2018 |
| FAM20B | Liu et al., 2018 |
| Ihh pathway/BMP2, TN-C | Stocum and Roberts, 2018 |
| ADAMTS5 | Rogers et al., 2018 |
| Notch signaling | Luo et al., 2018 |
| Ihh pathway | Kurio et al., 2018 |
| HIF-1α and VEGF | Yu et al., 2018 |
| DDR2 | Ge et al., 2018 |
| SMAD signaling | Xiao et al., 2017 |
| VEGF, p-ERK1/2 | Dong et al., 2017 |
lubricin, suggesting that the disruption of certain genes may trigger degenerative outcomes in TMJ tissues (Ishizuka et al., 2014). In mouse models, diminished lubricin production has also been associated with the presence of proteoglycan deficiencies and “significant alterations in all components of the TMJ” (Koyama et al., 2014). Similarly, considerable TMJ tissue degeneration has been seen in models using mice that over-express betacatenin (Wang et al., 2014). Rodent models have shown that the degeneration of the intra-articular cartilage, which is associated with sustained overloading of the TMJ, results in an increased expression of vascular endothelial growth factor (VEGF), hypoxia-inducible factor 1α (HIF-1α), and tumor necrosis factor (TNF) (Tanaka et al., 2005; Kartha et al., 2016) as well as an increased expression of matrix metalloproteinase 3 (Kartha et al., 2016) and damage to the proteins of the extracellular matrix (such as proteoglycans and collagens) (Wang et al., 2015). Sustained expression of these biochemicals and ongoing joint loading and inflammation lead to further degradation of the cartilage and bone, with impacts on the biomechanical function of the joint and enhanced susceptibility to additional injury (Sperry et al., 2017).
Sex Differences of TMJ Formation, Growth, and Function
Significant sex-specific differences exist in TMJ morphology (Iwasaki et al., 2017; Coogan et al., 2018; Coombs et al., 2019). Male condyles are on average larger than female condyles, with complex differences in the microstructural traits. Such sex differences in TMJ morphology, including in the condyle, disc, and fossa, have a direct impact on TMJ biomechanics and, by extension, on the health of these tissues. The available evidence suggests that the initiation of TMJ cartilage degeneration may be associated with repeated mechanical overloading of the TMJ at loads higher than normal physiological conditions (Sperry et al., 2017; Nickel et al., 2018). Such overloading of the TMJ has been associated with oxidative stress and inflammation, which could in turn lead to damage to tissues. In a study by Iwasaki and colleagues (2017), mechanical energy densities imposed on the disc in healthy TMJs were significantly larger in women than in men, which could predispose women to a higher rate of mechanical fatigue of the TMJ disc. Early mechanical fatigue of the disc tissues has been associated with degenerative joint disease of the TMJ (Iwasaki et al., 2017). Similarly, a 2018 study found that, during asymmetrical jaw closing, energy densities were significantly larger in female participants than in male participants, and the authors suggested that this presents a higher risk of mechanical fatigue (Gallo et al., 2018). These findings have led to the implication of sex hormones in playing a role in TMJ remodeling and tissue degeneration; however, this work remains inconclusive and requires additional research. TMJ morphological and biomechanical differences between the sexes need to be identified and explored to develop an understanding of how they may contribute to a female predisposition to tissue dysfunction or chronic orofacial pain.
Biomechanical Function, Joint Tissue Degeneration, and Orofacial Pain Processing
The relationship between biomechanical function, joint tissue degeneration or injury, and pain processing is an expanding field of research that seeks to unravel the complex interactions between the tissues of synovial joints and the nervous system in order to understand their impact on joint function and on the initiation, maintenance, and suppression of pain. From another direction, increasing evidence suggests that regular physical activity and exercise can reduce pain, and treatments are often aimed at improving biomechanical function in this population. However, very few studies have explored the interface among pain, biomechanics, and TMJ function in humans, and little is known about whether the addition or removal of pain results in predictable or sustained changes in joint function or vice versa.
The loss of the integrity of the articulating surfaces of synovial joints is consistent with the classical model of mechanical fatigue (Vazquez et al., 2019). There are analogies that can be made between the mechanical fatigue model of materials and the biopsychosocial model of chronic pain. The fatigue of materials depends on the magnitude and frequency of the mechanical load, with the material properties known as “stress raisers” acting as covariates (Beatty et al., 2008). From a mechanical perspective, stress raisers are entities, such as physical defects, produced during the manufacturing or development of the material or caused by traumatic loads that lead to an increased susceptibility of the material to damage and fatigue failure through normal function (Beatty et al., 2008). Similarly, the biopsychosocial model of pain depends on the magnitude and frequency of the primary neuron afferent barrage to peripheral ganglia and dorsal horn/trigeminal secondary interneurons (Staud, 2011b). From a chronic pain perspective, stress raisers are entities that contribute to neuroinflammation, such as increased sympathetic nervous system input to glial cells (Russo et al., 2018). To date, few studies on the development of chronic pain have quantified the magnitude and frequency of primary afferent input while controlling for co-variable stress raisers. Little is known about whether the removal of the afferent barrage, while controlling co-variable stress raisers, results in predictable or sustained changes in chronic pain and tissue function (Russo et al., 2018). Thus, there is a need to move beyond the separate examination of joint tissue failure and pain by using a biomechano-allostatic model, which combines the important variables in the development of degenerative joint disease with and without chronic pain.
The connection between biomechanical function of the TMJ and orofacial pain has been established in certain animal models (Sperry et al., 2017). TMD models using repeated mouth opening to defined loads in rodents result in hyperalgesia and osteoarthritis-like degeneration of the joint. Rats that experienced higher magnitudes of TMJ loading experienced longer periods of sustained pain (14+ days versus 7 days) than rats with lower levels of loading (Kartha et al., 2016). However, what is less understood is whether the interface of biomechanics and neurobiology, at a mechanistic level, can be manipulated to reduce afferent input and neuroinflammation in ways that attenuate pain signaling and improve tissue integrity. Rodent models have demonstrated that abnormal loading of the TMJ results in inflammatory changes to the cartilage and the upregulation of certain drivers such as VEGF and HIF-1α as well as the upregulation of matrix metalloproteinases, which are associated with degradation found in osteoarthritis of the TMJ (Kartha et al., 2016) and other conditions (Shen et al., 2015; Bechtold et al., 2016). The upregulation of these biochemicals may also mediate the response of nociceptors located in the TMJ tissues (Sperry et al., 2017).
The interface of biomechanical function and pain has been studied in other synovial joints (i.e., the knee and the facet joints of the spine), and it may be possible that the methods and tools used in these studies could be applied similarly to study the TMJ. In some cases, there is a correlation between the biomechanical and functional changes of the joint and pain, yet in many cases, particularly when pain becomes chronic, these relationships may not hold. It is also clear that there are multiple ways to interpret biomechanical function, including strength and endurance testing of muscle. More research is needed to understand how the biomechanical environment of the TMJ relates to joint function and its susceptibility to damage or injury and its relationship to pain signaling. Additionally, there is value in exploring individual-level differences in the biomechanical function of the TMJ and how these differences may affect peripheral and central pain signal processing in light of individual differences in allostasis, the processes by which the body copes with stressors in its efforts to maintain homeostasis.
Animal Models for TMJ Tissue Engineering and Regenerative Medicine Research
Those in the TMJ biomechanics and tissue engineering fields have studied a variety of animal models, including mice, rabbits, dogs, goats, pigs, minipigs, and sheep. Important factors when assessing animal models include anatomy/physiology, mechanical properties, chewing patterns, dietary composition, cost, and ease of surgical access (Donahue et al., 2019). A recent systematic review that examined preclinical TMJ tissue engineering studies noted a lack of standardization regarding animal models (Helgeland et al., 2018), an issue that may be impeding the translation of TMJ bioengineering approaches.
Several studies have examined similarities and differences in temporomandibular and craniofacial anatomy across species including rabbits, pigs, cows, goats, dogs, minipigs, and humans (Herring et al., 2002; Kalpakci et al., 2011; Vapniarsky et al., 2017). The pig is considered to be the gold standard of a non-primate, large-animal TMJ model due to its similarities to human anatomy, function, and tissue material properties (Almarza et al., 2018). Pigs are omnivores, which is one reason their TMJ disc and condyle are similar in structure to those of humans (Bermejo et al., 1993). Recently, the TMJ anatomy, histology, and biomechanics of black Merino sheep have been shown to also be similar to humans, although their diets differ from humans (Angelo et al., 2016). Further comparative studies of TMJ structures in black Merino sheep and pigs will be useful and may help the field reach consensus on the best model system for preclinical studies of TMJ tissue engineering approaches.
A noted challenge in the use of animal models is the difficulty in representing the pathology of the disease in the animal model. However, recent research suggests that some animals, such as dogs, cats, and horses, can develop certain TMJ pathologies such as osteoarthritis, ankylosis, luxation, fracture, and neoplasm (Almarza et al., 2018). Although the anatomic and physiological features of the TMJ may differ between humans and animals, these pathologies may have pathogeneses that are similar to those of TMDs in humans. Specifically, studying TMJ pathologies naturally occurring in animals may explain not only the pathogenesis of TMDs in humans but also the response to treatment.
Additionally, researchers have noted the challenges associated with the lack of established and relevant preclinical animal models for assessing novel approaches for TMJ tissue regeneration, which require larger animal models than those models employed for the study of joint disease (Almarza et al., 2018).
Overview of TMJ Tissue Engineering and Regenerative Medicine Research
In the future, the ability to engineer and regenerate temporomandibular tissues could provide an alternative treatment strategy to address certain TMDs and patients. Disc engineering and regeneration has been a major focus of recent TMJ tissue engineering research (Hunziker et al., 2015; Donahue et al., 2019; Melville et al., 2019). Despite promising results, the ability to develop these engineered constructs for clinical use has been limited by an incomplete understanding of the developmental biology of the TMJ tissues—a limitation that is further compounded by a lack of clinically relevant animal models and challenges with measuring function and forces within the joint space.
It is thought that disc dysfunction can be an early event that may lead to degenerative changes to the TMJ, implying that regeneration of disc tissues would be a valuable target for research. However, a major challenge with developing bioengineered TMJ discs is that they not only need to have similar morphological and histological characteristics to the native disc, but also must achieve the same mechanical function. Developing engineered TMJ disc constructs requires the selection of appropriate cell type or types, a biocompatible scaffold or scaffold-free support structure, and the proper biochemical or mechanical stimuli (Hunziker et al., 2015; Donahue et al., 2019; Melville et al., 2019). In addition, there remain concerns with the attachment of these engineered tissues and their capacity for tolerating early shear and torque that can occur during functional loading of the mandible (Vapniarsky et al., 2018; Almarza et al., 2019). More importantly, patients
will need to be selected carefully, as comorbid conditions could play a significant role in the success of healing.
Selection of Cells
In TMJ bioengineering, the selection of cell source remains a critical challenge. Compared to native tissues, bioengineered or regenerated TMJ components not only should have similar morphological and histological characteristics, but must also achieve the same mechanical function. Several cell types have been tested, including native TMJ disc cells (Ronald and Mills, 2016), TMJ fibrocartilage stem cells (Embree et al., 2016), costal chondrocytes (Vapniarsky et al., 2018), co-cultured articular chondrocytes and knee meniscus cells (MacBarb et al., 2013), and mesenchymal stem cells (MSCs) from either autologous or allogeneic sources. Cells obtained from MSC-based approaches have demonstrated similarities to native tissues in terms of morphological and biochemical characteristics, but further research is needed to determine if these MSC-based constructs can withstand the early shear and torque experienced during mandibular functional loading. Further work on progenitor cell populations and their differentiation into TMJ-like cells will be important to move this area of research forward.
Scaffolds and Scaffold-Free Approaches
Scaffolds serve as a surface on which cells can form new tissues and which can carry bioactive molecules that help to influence cell behavior (e.g., migration, stem cell differentiation). Additional research is needed to further refine scaffold fabrication methods such as nanoassembly, a process that builds scaffolds layer by layer, and three-dimensional (3D) printing (Donahue et al., 2019). Scaffold-free approaches are also being developed as a way to reduce complications with scaffold degradation and fabrication byproducts (Donahue et al., 2019).
Biochemical and Mechanical Stimuli
Stimulating bioengineered TMJ discs with growth factors, such as insulin-like growth factor I and TGF-β, has been shown to result in greater levels of collagen synthesis and improved mechanical properties (Detamore and Athanasiou, 2005). In addition to such biochemical stimuli, mechanical stimuli (e.g., hydrostatic pressure) have also been applied, typically to help recapitulate native tissue structure–function relationships in bioengineered TMJ discs (Gunja et al., 2009). Further exploratory research aimed at determining the appropriate combination of biochemical and mechanical stimuli is needed.
Other Tissues: Mandibular Condyle and Condylar Cartilage
Research on regenerative approaches for TMJ tissues beyond the disc is limited. Two areas of focus that have received less attention than disc regeneration include tissue engineering for the mandibular condyle and condylar cartilage. As with TMJ disc engineering, selecting the appropriate cell source has been challenging (Willard et al., 2011). In the case of condylar cartilage, a recent study showed tissue growth and regeneration in goat condylar cartilage using synthetic and natural scaffolds (Chin et al., 2018). Early studies such as these are promising, but further attention to these areas is needed to move the field forward.
Moving Toward Translation
Past and future research breakthroughs in TMJ bioengineering must be examined using orthotropic animal models, and once safety and effectiveness has been established, these approaches can be tested in humans. The location of the TMJ and its proximity to the brain may require more stringent safety requirements for bioengineered products (Donahue et al., 2019). Careful patient selection will be necessary in clinical studies, as it is unknown whether the presence of commonly comorbid conditions such as fibromyalgia could play a significant role in the successful regeneration of damaged tissues. Furthermore, metaplasia (the conversion of one cell type to another type), ossification (bone remodeling), and angiogenesis (development of new blood vessels) may be concerns for specific patients (Detamore et al., 2007).
Regenerative Capacity of Endogenous Cells
Recent work showed that endogenous fibrocartilage stem cells may be useful for regenerating and repairing cartilage in the TMJ condyle (Embree et al., 2016). Terminal differentiation of chondrocytes is thought to drive aspects of osteoarthritis in the TMJ, and another recent study demonstrated that inhibiting Ihh signaling protected chondrocytes (Yang et al., 2019). Additional research is needed on how endogenous cells and the native TMJ microenvironment can be therapeutically exploited to repair or regenerate damaged tissue.
Looking Forward: Research Priorities for Biomechanics and Regenerative Medicine Research on TMDs
More research is needed to better define the unique mechanics of joint and tissue interfaces within the TMJ and the function of the joint in health
and in disease. Too little is known about the function of the genes involved in the development of the TMJ and its tissues, specifically about how variations in TMJ development may predispose the joint to future dysfunction. Such research could lead to improved regeneration methods for damaged tissues. Furthermore, only limited research has been conducted to understand neurological control and pain sensitization in the orofacial region. Of critical importance is the exploration of the relationship between pain and function, specifically an understanding of innervation and its interactions with the many tissues and vascular structure of the TMJ, which can inform methods for joint repair and regeneration. While tissue turnover in and the repair of bone, cartilage, and ligaments of the TMJ have been studied to some extent, a better understanding of these repair processes is needed. Specifically, a better understanding is required of the integration of mechanical cues and pathological joint loading on tissue-remodeling processes. Additionally, there has not been enough research performed on mechanotransduction to allow an understanding of the relationship between joint motion, forces, and the physical properties of tissues and the relation of these to cellular mechanotransduction.
The development of new therapies to repair and prevent joint dysfunction and to regenerate new and functional tissues safely will require a strong foundation of basic and clinical research on the biology of the TMJ in health and in disease. Addressing existing research gaps will demand new approaches and instrumentations for in vivo measurement of joint health, disease, and function. It is clear that there will be value in going beyond static imaging of the joint to examine the dynamic joint function, in using 3D imaging of joint structure and joint quality, and in leveraging imaging technologies and datasets from other disciplines (i.e., cohorts with imaging of brains with neurological disease). Moreover, due to the limitation of direct in vivo measurement, the development of in silico models and the use of virtual human trials would provide significant opportunities to understand TMD risk factors and the etiology of TMD development and progression. Such models might also help explain the increased prevalence of TMDs among women and other known and unknown disparities and musculoskeletal and psychosocial factors. Additionally, researchers could use novel biomedical techniques for modeling the TMJ, such as biocomputing, and molecular imaging for tissues and structures. See Box 4-1 for a list of research priorities.
NEUROBIOLOGY OF OROFACIAL PAIN AND TMDs
The larger field of pain research has worked to define the mechanisms of neuropathic and inflammatory pain, and, as with many complex disorders featuring acute and chronic pain, understanding the pathophysiology
of TMDs requires an in-depth understanding of the mechanisms of pain and their role at different levels, from the cellular to an inter-system level. Despite the significant burden and often life-changing impact of orofacial pain, relatively little is known about the neurobiology of TMJ tissues in health and in disease. This is, in part, due to the highly complex, multidimensional nature of acute and chronic pain and the numerous biopsychosocial underpinnings that mediate the experience and perception of pain. Also, little is known regarding biomechanical dysfunction (see above section on tissue engineering) and how this relates to or contributes directly to orofacial pain. This dearth in knowledge is in part due to the significant siloing of fields of research, with researchers working on tissue bioengineering and those working on pain biology needing to find opportunities to collaborate on TMD research. The following sections will explore the findings of recent research on TMDs and orofacial pain, including:
- Animal models for orofacial pain,
- Overviews of peripheral and central mechanisms of acute and chronic orofacial pain,
- Pain signaling and biomechanics of the TMJ,
- Trigeminal versus extra-cranial pain signaling systems, and
- Commonalities in neuronal pathways and central sensitization within TMDs and other chronic pain conditions.
Current Animal Models of Orofacial Pain
Animal models provide critical opportunities for studying the biological and cellular mechanisms of pain and help advance the development of pharmacological treatments. However, the barriers to translation from studies in experimental animals to clinical drug development are numerous and are a result both of the challenges of using animal models in general and of more systemic issues related to shortcomings in the research ecosystem. Compared to pain experienced in other areas of the body, specific animal models for orofacial pain have been less extensively studied (Krzyzanowska and Avendaño, 2012). While broader pain research findings and methodologies can be applied to the study of orofacial pain, it is essential that mechanisms unique to the orofacial area be explored using anatomically relevant animal models to understand the pathogenesis of inflammatory and neuropathic orofacial pain common to TMDs and to identify effective existing or novel treatment modalities.
In order to study the mechanisms of pain associated with TMDs, commonly used mouse and rat models attempt to mimic inflammatory orofacial pain through the injection of various chemicals or irritants into the masseter muscle or the TMJ (Martínez-García et al., 2019). Models
for inflammatory pain require an initial tissue injury or irritant injected directly into the muscle or joint to trigger a selected area or cell type into activating primary sensory neurons and attract immune response cells that release substances like cytokines, which can then be studied. The methods for triggering an inflammatory pain response range from general chemical irritants (carrageenan and formalin) to agents that directly target specific sensory neurons (capsaicin and mustard oil) (Krzyzanowska and Avendaño, 2012). Alternatively, mechanical trauma such as repeated daily mouth opening, intraoral appliance placement, disc displacement, or ligation of the tendon of the masseter muscle have been used to mimic and study TMJ dysfunction (Guo et al., 2010; Araújo-Filho et al., 2018).
Other animal models designed to explore the pathophysiology of neuropathic orofacial pain involve the constriction, transection, or compression of the distal branches of the trigeminal nerve, such as the infraorbital, inferior alveolar, mandibular, or mental nerves. Of these animal models, the chronic constriction injury (CCI) of the infra-orbital nerve is one of the most commonly used orofacial pain models (Krzyzanowska and Avendaño, 2012; Araújo-Filho et al., 2018; Martínez-García et al., 2019). These models of partial nerve injury attempt to simulate the neuropathic symptoms reported by some patients with a TMD.
Depending on the study, a variety of outcome measures have been used to assess nociceptive responses in awake behaving non-human animals, including behavioral outcome models. Common behavioral outcome measures include evoked withdrawal responses to mechanical, cold, or heat stimuli; non-evoked spontaneous behavior such as face grooming; and other types of operant behavioral assessments (Neubert et al., 2005; Langford et al., 2010; Romero-Reyes et al., 2013). In the TMJ arthritis model, for example, meal pattern analysis (i.e., meal duration, total number of meals, and total time spent eating) is monitored to assess feeding behavior. Observations of feeding behaviors do appear to have value as a behavioral outcome for TMDs. While numerous behavioral outcomes can be used to assess nociceptive responses, these outcomes are difficult to quantify and may lack sensitivity (Krzyzanowska and Avendaño, 2012). These behavioral response studies are in contrast to the study of reflex responses to external stimuli, which do not require the engagement of cerebral processes. It has been argued that spontaneous pain behaviors and complex operant behaviors that involve cortical processing and decision making may be more relevant in providing insights into human orofacial pain conditions.
The pain community has made significant advances in the understanding of pain through the development of diverse animal models used to examine the environmental and biological processes underlying acute and chronic pain. The use of inflammatory and neuropathic pain models has revealed a number of potential mechanisms and pathways involved in painful
TMDs, and it provides useful information for those conditions associated with inflammation and neuropathic pain. However, TMDs are heterogeneous and involve biomechanical and functional alterations, psychosocial factors, and environmental factors and stressors—some factors of which may be difficult to capture and measure in preclinical models. Historically, the field of pain research has focused on factors involved in the transduction of nociceptive stimuli and the induction of acute pain, but more emphasis is needed to understand factors that both promote and prevent the transition from acute to chronic pain and that promote recovery from chronic pain. Recent animal models of musculoskeletal pain have begun to combine negative stress, such as sound or fatigue (Chen et al., 2011; Alvarez et al., 2013; Sluka and Clauw, 2016), with tissue insults, and they show enhanced and more widespread hyperalgesia; the use of these models in TMD research is just emerging (Traub et al., 2014) and requires additional focuses. Furthermore, recent studies in animal models of pain unrelated to TMDs have identified lifestyle factors such as physical activity and biological factors such as anti-inflammatory mediators (e.g., interleukin-10) that can protect both animals and humans from the development of chronic pain (Kavelaars et al., 2011; Leung et al., 2016; Sluka et al., 2018). Unique animal models need to be developed that take into account the complex nature of TMDs, the multiple stressors that can initiate the development and persistence of pain, and the biomechanical and structural findings observed in individuals with TMDs. Collaborations with researchers using other pain models—like post-surgical tissue trauma and burns—and those studying TMJ bioengineering and tissue regeneration could provide potentially valuable new models. Such new models would benefit from the development and use of standard outcome measures for the assessment of orofacial pain and TMJ function.
Overview of Peripheral Mechanisms of Orofacial Pain
As described earlier in the chapter, the orofacial tissues are innervated by the trigeminal nerve and its nociceptive endings, which terminate in the orofacial tissues and can be activated by mechanical stimuli or injury, inflammatory processes, or exposure to an irritant or inhospitable environment. This system of mechanisms is inherently vulnerable to modulation because each component of the system interacts within and is influenced by the complex biochemical environment—endocrine, immune, and other systems—of the human body (Sessle, 2011).
Over the past several decades, research into the central and peripheral mechanisms of acute and chronic orofacial pain has identified a network of chemical mediators, receptors, channels, and interactions that influence the activation and sensitization of nociceptive pathways (Sessle, 2011). The involvement of certain inflammatory mediators—such as prostaglandins
and bradykinins—in activating nociceptive nerve endings in orofacial tissues has been well established (Sessle, 2011). In addition, other peripheral mediators released from mast and immune cells and macrophages—such as 5-hydroxytryptamine (5-HT), histamine, TNF-α, and interleukins (ILs)—act by increasing the excitability of the nociceptive nerve endings at the injury location. This multi-variable mechanistic process of peripheral sensitization is mediated by interactions with the immune, cardiovascular, and endocrine systems (Sessle, 2011). The processes by which these numerous mediators act involve a multitude of receptors and ion channels on the nociceptive endings; they also act via signaling and second messenger systems and through the associated intracellular matrix (Sessle, 2011). During the process of peripheral sensitization, these inflammatory mediators can increase the excitability of stimulated nociceptive endings in the initial site and those adjacent through interactions with certain ion channels and signaling systems (Sessle, 2011). Additionally, exposure to certain inflammatory mediators can result in phenotypic changes within nociceptors, which can fundamentally modify ion channels (e.g., sodium channels) and the expression of certain receptors. Therefore, there is significant value in understanding the role of these mediators and how they act upon the nociceptive pathway to modulate pain response and induce lasting hypersensitivity, because many TMDs feature both acute and chronic inflammatory pain as a primary feature (see Chapter 2).
Overview of Inflammatory Mediators
Certain chemical mediators play a significant role in the activation or sensitization of nociceptors and, by extension, the inflammatory pain characteristic of many TMDs. As such, recent research has focused increasingly on understanding the specific role of these individual mediators and receptor expression on the pathogenesis of orofacial pain. Multiple chemical mediators have been the focus of investigation. Among those, gamma amino butyric acid (GABA) receptor subunit alpha (Gabrα6) has been shown to be expressed by the trigeminal neurons, although the role of this expression on orofacial pain remains unclear. A study in rats by Puri and colleagues (2012) showed that Gabrα6 expression may play a role in hypersensitivity of the TMJ by inhibiting afferents in the trigeminal pathway and reducing inflammatory orofacial nociception. Additionally, a 2019 study suggests that the activation of inflammasomes—via the secretion of IL-1β and IL-18—could play a significant role in the elicitation of an inflammatory response across a range of metabolic, cardiovascular, and neurodegenerative disorders as well as TMDs (Ibi, 2019).
Beyond their role in triggering and maintaining inflammation, the presence of these mediators in orofacial tissues could be an indication of the
health of the tissue and act as diagnostic markers or therapeutic targets. In his 2011 review of central and peripheral mechanisms of orofacial pain, Sessle noted that mediators such as 5-HT, IL-1β, and prostaglandins are not present in the TMJ synovial fluid of healthy subjects, but they are detectable in inflamed TMJ synovial fluid (Sessle, 2011). Sessle also suggested that glutamate levels in the jaw muscles of patients with certain TMDs may indicate the presence of an inflammatory response (Castrillon et al., 2010; Sessle, 2011).
Receptors and Ion Channels
The role of receptors and ion channels, particularly the function of sodium ion channels like Nav1.7, has been and remains a significant focus of orofacial pain research. Recent studies have demonstrated that Nav1.7 signaling plays a significant role in pain processing, with point mutations resulting in either intractable pain or a complete absence of pain through the amplification of otherwise weak stimuli (Cummins et al., 2007). That ion channel may also play a role in visceral pain processing (Hockley et al., 2017) associated with certain chronic overlapping pain conditions, such as irritable bowel syndrome (IBS) (Campaniello et al., 2016). As shown in knockdown and knockout studies in mice, Nav1.7 interferes with mechanical and thermal inflammatory pain responses (Nassar et al., 2004). In the trigeminal nerve, chronic inflammation induced by Freund’s adjuvant injection into the TMJ upregulated Nav1.7 mRNA and protein in neurons innervating the TMJ while blocking the Nav1.7 sodium channel in the trigeminal ganglion, which significantly reduced pain response in the joint (Bi et al., 2013). These findings suggest that Na channel subtypes play a role in the pain response in TMD. Research has also considered the role of calcium-permeable ion channels expressed by trigeminal ganglia sensory neurons, such as TRPV4, in pain behavior. A 2013 study in mice by Chen and colleagues (2013) demonstrated that the expression of TRPV4 is associated with nociceptive response after TMJ inflammation.
In addition to examining the roles that Nav1.7 and TRPV4 play in pain perception within the TMJ, additional research has sought to describe how these channels are regulated and the mechanisms underlying this regulation. As described above, multiple studies have pointed to the regulation of Nav1.7 by pro-inflammatory mediators such as nerve growth factor (NGF), TNF-α, 5-HT, prostaglandins, and cytokines (IL-1β) (Tamura et al., 2014; Isensee et al., 2017) (see section above).
While the etiologies of TMDs remain elusive, the recent explorations of these mechanisms, receptors, and ion channels indicate potential pathways through which chronic inflammation may amplify pain responses. To illustrate such a pathway, a 2017 study by Kobayashi and colleagues demonstrated that synovial cells in samples of human TMJ tissues can release
inflammatory mediators—bioactive peptides called elastin-derived peptides (EDPs)—during the degradation of the extracellular matrix. EDPs promote the upregulation of IL-6 and elastin-degrading matrix metalloproteinase-12 (MMP-12) (Kobayashi et al., 2017). The presence of EDPs was found to be correlated with both the duration of jaw locking and IL-6 expression. These findings suggest an environment within TMJ where the activation of an elastin-binding protein signaling cascade, as a result of harmful mechanical stimuli, triggers a pro-inflammatory cascade and MMP-12 expression, which may create a positive feedback loop of chronic inflammation within the joint (Kobayashi et al., 2017).
Overview of Mechanisms of Neuropathic Pain and TMDs
As described earlier, several models of partial injury to branches of the trigeminal nerve have been used in rodents to study the disabling neuropathic pain observed in a subset of patients with TMDs. The behavioral responses observed in these models include mechanical hyperalgesia, air puff allodynia, and paraesthesias/dysaesthesias. Studies suggest that microglial activation plays an important role in the development and maintenance of central sensitization, as evidenced by the reversal of mechanical allodynia through the suppression of microglial activation (Ma et al., 2012). Expression of phosphorylated extracellular signal-regulated kinase (pERK) in the dorsal horn and its phosphorylation is implicated in pain since pharmacological blockade reduces pain behavior in the infraorbital chronic construction injury (CCI) model. Ito and colleagues (2013) observed an upregulation of P2X(7)R, membrane-bound TNF-α, and soluble TNF-α in the trigeminal sensory nuclear complex after a CCI of the infraorbital nerve. Antagonists of the P2X(7) receptor and inhibitors of the phosphorylated (p)-p38 mitogen-activated protein kinase (MAPK) inhibited the tactile allodynia in this model, suggesting that phosphorylation of p38 MAPK via P2X(7)R may play a critical role in the mechanisms of hypersensitivity. Shibuta and colleagues (2012) reported the presence of hyperactive microglial cells and a large number of pERK-immunoreactive (IR) cells in trigeminal spinal subnucleus caudalis and the upper cervical spinal cord (C1) after infra-orbital CCI. Minocycline significantly reduced the activation of microglial cells and the number of pERK-IR cells at these sites, suggesting that the activation of microglial cells in the trigeminal and upper cervical regions is involved in the increased neuronal excitability associated with the neuropathic pain.
Central Mechanisms of Pain
Beyond the induction of pain in the periphery, peripheral nociceptive stimuli can also affect CNS neurons (Cairns, 2010) via the trigeminal
ganglion to the subnucleus caudalis—an important site for the transmission of information from the periphery to the brain—which contains many CNS pain receptors. Receptors and ligands that may be involved include N-methyl-D-aspartate (NMDA), GABA, 5-HT, glutamate, and certain neuropeptides (Cairns, 2010; Sessle, 2011). Many of the neurons of the C1 and C2 dorsal horns and the subnucleus caudalis receive direct inputs from the peripheral afferents in the surface tissues surrounding the TMJ and play a critical role in the CNS processing of pain in these orofacial tissues (Chichorro et al., 2017). Using injection of glutamate into the TMJ in animal models demonstrates an expansion of the neuronal receptive field and sensitivity in regional tissues via the sensitization of subnucleus caudalis neurons, in addition to the activation of TMJ nociceptors. This suggests the presence of central sensitization (Lam et al., 2009). Neurons of the subnucleus caudalis and the C1–C2 dorsal horns and caudalis/interpolaris transition zone also process nociceptive information from deep tissues—such as facial muscles and the TMJ—which results in complex patterns of convergence in the processing of superficial and deep tissue afferent inputs. These patterns require further study, as these may offer further understanding concerning the localization and expansion of pain in various orofacial pain states (Chichorro et al., 2017).
Evidence of CNS pain processing has also been noted in studies of pain thresholds and electrical stimulation of the TMJ, which found indications of lower pain thresholds among individuals with arthralgia of the TMJ compared with healthy controls (Cairns, 2010). Such findings are highly relevant to understanding the experience of orofacial pain at a mechanistic level, as many individuals with orofacial pain experience increased regional sensitivity in the skin and muscles around the TMJ.
Of interest to central sensitization is the process of reversible and non-reversible neuroplastic changes in nociceptive afferents, which has been shown to be initiated by certain neurochemicals of the nociceptive process. For example, glutamate activates NMDA receptors and can result in neuroplastic changes indicative of central sensitization (Chichorro et al., 2017). Chichorro and colleagues (2017) write that this process “underscores the point that the nociceptive pathways and processes in the CNS are not ‘hardwired’ but rather are ‘plastic’ and modifiable by events associated with injury or inflammation in peripheral tissues” (p. 617). Central sensitization involves the prolonged and increased excitability of neurons and increased synapse function in central nociceptive pathways (Woolf, 2011), which can result in pain experienced that may not match or require a noxious stimulus. This process is known to result in pain hypersensitivity and pressure hyperalgesia, and it enhances temporal summation as well as causing secondary changes in brain activity (see section on neuroimaging for brain-based biomarkers of pain) and contributing to diverse pain conditions, such
as fibromyalgia, osteoarthritis, and TMDs, among others (Woolf, 2011). Woolf (2011, p. 4) writes,
Central sensitization introduces another dimension, one where the CNS can change, distort, or amplify pain, increasing its degree, duration, and spatial extent in a manner that no longer directly reflects the specific qualities of peripheral noxious stimuli, but rather the particular functional states of circuits in the CNS…. This does not mean that the pain is not real, just that it is not activated by noxious stimuli.
An understanding of the mechanistic differences and convergence across the many peripheral and central mechanisms underlying acute and chronic pain is critical to the identification of clinically meaningful pain phenotypes within conditions to guide further research and clinical care.
Orofacial Pain Modulation
The modification of pain on an individual level is a product of the convergence of modifiable and non-modifiable biopsychosocial factors. Current research suggests that various interacting mechanisms play a role in the generation, maintenance, and suppression of pain by the CNS, which can, as described above, signal a mismatch between the peripheral nociceptive inputs and the perception of pain by an individual. Hence, the pain experience is highly heterogeneous across individuals and difficult to measure objectively (Harper et al., 2016a). Advances in the understanding of pain signaling mechanisms in both healthy and disease states has led to the concept of pain modulation, where CNS mechanisms can generate and maintain pain. This means that the presence of an injury or inflammation within the peripheral system may not necessarily translate into pain, just as an individual could experience intense pain in the absence of injury and inflammation.
Past studies in humans have compared endogenous pain modulation (pain facilitation or inhibition) in individuals with TMDs versus in individuals without TMDs. Conditioned pain modulation (CPM), an index of pain inhibition, is studied by examining the reduction in pain to a noxious stimulus by a concurrent or prior noxious stimulus at a distant site. Pain facilitation is examined using a temporal stimulation paradigm, where a fixed-intensity noxious stimulus is repeated at frequencies that result in increased pain. Some studies have reported TMD patients feeling greater pain than controls in response to such stimuli. However, these observations have not been consistent across all studies (Greenspan et al., 2011; Moana-Filho et al., 2018), with some findings suggesting impaired CPM effects and others reporting similar CPM in both people with TMDs and healthy
controls (Kothari et al., 2015; Moana-Filho and Herrero Babiloni, 2019). This lack of a significant difference suggests that not all painful TMDs are associated with compromised endogenous pain inhibitory systems and, by extension, that there are likely to be subgroups of individuals with TMDs who feature unique combinations of biopsychosocial mechanisms that contribute to the generation and maintenance of pain (Harper et al., 2016a; Moana-Filho et al., 2018). These findings support the suggestion that pain related to TMDs exists on a spectrum from cases where pain is generated in the periphery through to cases where pain is the result of an exaggerated response in the CNS, with most cases having a combination of peripheral and central changes (Harper et al., 2016a) (see Chapter 2 for the discussion of pain as a chronic disease).
Over the past decade, TMD and chronic pain researchers have become increasingly interested in developing a more complete understanding of the different biopsychosocial mechanisms that are responsible for the generation and maintenance of pain, rather than continuing to rely on exclusively anatomical classifications of pain (i.e., low back pain, jaw pain) (Bair et al., 2016). This approach also allows for further exploration of both the unique and shared mechanisms between TMDs and other commonly comorbid pain conditions, such as IBS, fibromyalgia, and chronic pelvic pain. Clauw writes,
A critical construct is that, within any specific diagnostic category … individual patients may have markedly different peripheral/nociceptive and neural contributions to their pain. Thus, just as low back pain has long been acknowledged to have multiple potential mechanisms, so also is this true of all chronic pain states. (Clauw, 2015, p. 6) (see Table 4-2)
Most notably, as part of the Orofacial Pain Prospective Evaluation and Risk Assessment (OPPERA) study, cluster analysis performed on data from a case–control study (1,031 chronic TMD cases and 3,247 controls) identified three distinct patient subgroups across an array of biopsychosocial factors. Their findings suggest that classification of individuals into clinically relevant and mechanistically based subgroups using biopsychosocial risk factors could provide a better and more personalized approach for understanding orofacial pain etiology and, in the future, for the development and application of more targeted treatments (Bair et al., 2016). The authors suggest that such a method prioritizes mechanistic and etiological distinctions for pain more effectively than grouping by a specific TMD diagnosis as mechanisms within a specific pain diagnosis can vary by individual.
The centralized, multi-focal pain that is characteristic of many patients with TMDs—in addition to many other symptoms, traits, and factors—is a feature of many other overlapping chronic pain conditions (Williams
TABLE 4-2 Mechanistic Characterization of Pain
| Attribute | Nociceptive | Neuropathic | Centralized/Nociplastic |
|---|---|---|---|
| Cause |
|
|
|
| Clinical features |
|
|
|
| Mixed Pain States | |||
| Classic examples |
|
||
NOTE: CNS = central nervous system; GI = gastrointestinal.
SOURCE: Modified from Clauw, 2019.
and Clauw, 2009; Clauw, 2015), suggesting that there may be mechanistic overlap in the pathophysiology in certain subgroups of patients with these disorders. The mechanistic variability across patients and TMDs needs to be parsed out to the pathophysiology of each TMD and for the development of more effective and individualized care strategies for patients.
The use of standardized quantitative sensory testing (QST) to assess the function of sensory nerve fibers in patients with TMDs may help define somatosensory disturbances such as pain sensitivity and endogenous pain modulation. Additionally, QST may provide further insights on the mechanisms of pain in TMDs and help predict which individuals are at higher risk of transitioning from an acute to chronic pain state after injury or inflammation.
Pain and Biomechanical Function of the TMJ
As discussed in a prior section, considerable work needs to be done to understand how the biomechanical function and use of the TMJ affects the health of its tissues and the generation and maintenance of orofacial pain at a mechanistic level. This area of research is complex because of the multitude of known and unknown mechanisms and interactions involved,
including mechanical forces, biochemical mediators, peripheral and CNS interactions, and psychosocial factors affecting the use of the joint and the experience of pain. The relationship between biomechanical control or function and pain has been of interest to researchers in other areas of study, such as low back pain (Hodges and Moseley, 2003) and pain in the hip and knee (Powers, 2010). However, relatively little is known about the mechanisms underlying these relationships and how these relate to the etiologies of TMDs, and there is a lack of consensus in the research regarding a correlation between biomechanical function and pain.
In the case of the TMJ’s biomechanical function and orofacial pain, research has shown that pathological changes to joint tissues, such as the degeneration of tissues resulting from the overloading of the TMJ, alters the biochemical environment of the joint and has some degree of impact on the mediation of peripheral and central signaling processes that initiate and maintain pain (Sperry et al., 2017). However, the presence of tissue degeneration does not equate to a predictable level or the presence of pain, which supports the concept that other biochemical pathways could also be affected by biochemical degenerative changes and play a contributing role (i.e., inflammatory cytokines) in the generation and maintenance of pain (Sperry et al., 2017). Other mediators associated with degeneration (collagenase and the increased expression of pERK) may also play a role in nociception (Gao and Ji, 2009; Adães et al., 2014), although more research is required to understand their role in the sensitization of neurons (Sperry et al., 2017). Furthermore, relatively little is known about the relationship between pain and parafunction of the masticatory muscles. There is some evidence suggesting that TMD pain may be associated with increased protective muscle reflexes, but this requires further study (Cairns, 2010).
This type of research represents an area of significant value for understanding the complexities of TMDs from a biopsychosocial perspective, particularly as researchers continue to elucidate the role of psychosocial factors in relation to the experience of pain and explore pain’s underlying biological mechanisms.
Trigeminal Versus Extra-Cranial Pain Signaling Systems
Pain in the orofacial region is signaled via the trigeminal ganglia (TG), in contrast to pain signaling from the rest of the body, which occurs via the sensory dorsal root ganglia (DRG). Studies in pain-free human subjects indicate regional differences in endogenous pain inhibition, with significantly weaker inhibitory pain modulation in areas innervated by the trigeminal nerve than in other body regions (Levy et al., 2018). Differences in the origins of DRG and TG neurons have been identified. While DRG neurons are derived primarily from the neural crest, TG neurons have a dual origin
and contain cells originating both from the cranial neural crest and from trigeminal ectodermal placodes (Erzurumlu et al., 2010; Lopes et al., 2017).
Recent efforts have attempted to characterize the gene expression profiles of the sensory neurons present in DRG and TG to determine if they have distinct expression profiles and unique molecular fingerprints. Transcriptome analyses of neurons in mice reveal that, although the cells at these two sites are fundamentally similar in gene expression, 24 genes were found exclusively in either of the ganglia and a number of genes were differentially expressed in DRG and TG neurons, including ion channels and genes reportedly involved in pain processing (Lopes et al., 2017). These differences in the expression of inflammatory mediators and pain sensitivity testing need to be explored. Additionally, large-scale population studies would be needed to determine whether there is a unique genetic profile and how this profile may predispose or protect an individual from experiencing trigeminal pain; however, this would require accurate methods of quantifying trigeminal pain.
Commonalities in Neuronal Pathways and Central Sensitization Within TMDs and Other Chronic Pain Conditions
The relationship between TMDs and other overlapping chronic pain conditions, such as primary headaches, cervical spine disease, IBS, and fibromyalgia, imply the existence of common neuronal pathways and central sensitization processes (see the example in Box 4-2). Central sensitization and an impaired descending pain inhibitory system have been postulated as potential shared pathophysiological mechanisms. Other potential mechanisms, such as peripheral sensitization and neuroimmune interactions, have also been considered as mechanisms overlapping TMD and comorbid pain conditions (Costa et al., 2017). Further research is needed to understand these shared mechanisms and to what extent the effective treatment of a comorbid condition would affect the pathophysiology of a co-occurring TMD.
Looking Forward: Priorities for Orofacial Pain Research
As seen in other chronic pain conditions, the contribution of centralized pain mechanisms is often greater than the initial inciting trigger of the pain. As such, these pain syndromes can be difficult to replicate in animal models. The relationship between centralized pain mechanisms and the inciting disease is evident in the clinical research of disorders classified within the Diagnostic Criteria for Temporomandibular Disorders. This classification relies on both Axis I, which provides characterization of the disease in the joint and muscle, and Axis II, which assesses psychosocial and pain-related
disability. The incorporation of pain assessments into TMJ animal models will be critical for understanding the transition from acute to chronic pain and for the improved translation of regenerative medicine research into clinical care. As measures of widespread pain are developed and tested, they should be employed in the study of TMDs. See Box 4-3 for select priorities for orofacial pain research.
NEUROIMMUNE INTERACTIONS AND TMDs
The scientific evidence suggests that while the immune system—which regulates inflammation to protect against threats—and the nervous system—which controls bodily functions through the release of neurotransmitters—are distinct, these two systems coordinate at a molecular and cellular level to maintain tissue function (Chavan et al., 2017; Veiga-Fernandes and Artis, 2018). Beyond these essential interactions necessary for tissue homeostasis and function, there is evidence that neuroimmune interactions could play a role in autoimmune and chronic inflammatory disorders (Hagerty et al., 2019). The role of the immune–nervous system interactions on the pathophysiology of pain and tissue dysfunction found in TMDs remains largely
unclear, although it is a growing area of research because the exploration of neuroimmune interactions could provide critical information on the mechanistic underpinnings of complex disorders (Chavan et al., 2017), such as TMDs and their comorbidities. This section includes a brief overview of the mechanisms of peripheral sensory neuronal function in response to
immune challenges, the neural regulation of immunity and inflammation, and the implications of these findings for treatment.
There are multiple points of interaction and communication between the nervous system and the immune system. Most relevant are the immune cell activation influences on neuronal circuits (such as changes to nociceptive signaling thresholds), which subsequently modulate both innate and adaptive immune responses. This shared molecular signaling—immune cells can produce what are classically defined as neurotransmitters, and neurons and their supporting cells release what are classically defined as cytokines/chemokines—is of particular interest. Different immune–neuronal signaling interfaces might be augmented in tissue environments responding to distinct injuries, and therefore an analysis of the mediators in different subjects with distinct phenotypes might provide opportunities to stratify patients and try novel therapeutic approaches.
Although research in this field directly related to TMDs is limited, the existing studies do shed some light on the potential value of this research. For example, in a mouse model of sustained mouth opening, increased macrophage/microglia activation was observed in the trigeminal subnucleus caudalis (Hawkins and Durham, 2016). Furthermore, inhibiting macrophage and microglial activation prevented the development of orofacial mechanical hypersensitivity.
Furthermore, neuroimmune interactions may play an important role in pain chronicity. Therefore, it may be possible that chronic inflammation and responses to injury, surgery, implants, and devices may play a role in establishing chronic symptoms of TMDs. This is based on the concept that the circuits present in particular patients may favor a greater likelihood of chronic pain and other symptoms than appear in other patients.
To apply neuroimmune interactions in a clinically meaningful way, it is critical that the genetic framework of key inflammatory, immune, and resolution pathways in patients with various subgroups of TMDs be understood. Because there is significant complexity and heterogeneity within these neuroimmune interactions at different sites and for different stimuli, it is therefore important that researchers work toward defining those pathways and circuits that are relevant to TMD in humans to provide a foundational understanding. Despite the various unknowns in this area, enough is known to say that there appears to be potential clinical value for individuals with inflammatory TMDs. Indeed, recent clinical trials using bioelectric devices to modulate the neuroimmune pathways as a treatment strategy for inflammatory diseases such as rheumatoid arthritis and inflammatory bowel disease have demonstrated some success (Brinkman et al., 2019; Payne et al., 2019).
Looking Forward: Future Areas of Neuroimmune Research for TMDs
To date, there has been only limited study of the role of neuroimmune mechanisms in the pathophysiology of TMDs. Neuroimmune approaches have been used in other fields and with other complex diseases (Hagerty et al., 2019), and lessons can be learned both from those findings and from the methodologies employed. The study of neuroimmune interactions represents significant value to the broader field of TMD research, as findings in this area would provide insight into shared mechanisms for inflammation and chronic pain, indicate shared risk factors for autoimmune and inflammatory conditions, and suggest meaningful therapeutic targets needed for the development of more effective treatment and care practices. Several areas of neuroimmune research that could provide promise for TMDs are listed in Box 4-4.
NEUROENDOCRINE INTERACTIONS, STRESS RESPONSE, AND TMDs
The neuroendocrine system is a complex network of neurons, glands, and non-endocrine tissues that generate and interpret a wide variety of neurochemicals, hormones, and other signals that function to regulate physiology or behavior (Levine, 2012). The hypothalamus, anterior pituitary systems, adrenal cortex, and downstream target tissues are key axes in the neuroendocrine system. Regulation of the hypothalamic–pituitary–adrenal (HPA) axis is an essential part of how humans adapt to their environment and is important for the body’s response to stress and to the homeostatic
regulation of the metabolic, cardiovascular, immune, reproductive, and central nervous systems (Smith and Vale, 2006).
Recently there has been growing scientific interest in the role of stress, activation of the HPA axis, and downstream inflammation on the initiation and progression of TMDs. For example, one study showed that individuals with a TMD exhibited higher scores on both the Hospital Anxiety and Depression Scale as well as on “pain-catastrophizing events”—both scores of which may possibly contribute to the upregulation of the HPA axis (Staniszewski et al., 2018). Laboratory studies have also demonstrated the relationship between cortisol and circulating levels of pro- and anti-inflammatory cytokines (e.g., IL-6, TNF-α). Further study is needed to better understand the relationship between stress-related hormones and the onset and severity of TMD symptoms and across subgroups of patients with TMDs.
Other multi-system, chronic pain conditions such as fibromyalgia exhibit altered neuroendocrine and autonomic nervous system function, with both hypo- and hyperactive stress responses having been reported (Adler et al., 2002). Heart rate variability has been used as an indicator of the balance between the parasympathetic and sympathetic nervous system, the two branches of the autonomic nervous system. As measured by 24-hour heart rate monitoring, heart rate variability is lower in persons with a TMD (Chinthakanan et al., 2018), suggesting reduced vagal tone. Additional research on autonomic nervous system function in individuals with a TMD is needed to better understand how that function is altered and if it can be a target of therapeutic interventions.
Chronic pain disorders such as IBS, fibromyalgia, migraine, and interstitial cystitis are present at higher rates in females. In a rat model the estrous cycle and fluctuations in estrogen levels seemed to be linked to pain sensitivity fluctuations (Moloney et al., 2016). There are indications that TMD pain symptoms vary across the menstrual cycle, peaking in the late luteal phase and at menses (LeResche et al., 2003). These phases represent the time of declining or low estrogen levels. Additional research is needed to gain clarity on the effect of estrogen levels and the menstrual cycle on TMD initiation, progression, and response to treatment. The next section covers research on sex differences observed with TMDs in greater detail.
Finally, rigor and reproducibility are two important methodological considerations to take into account with regard to studies on the body’s stress response and its relationship to pain in the context of complex disorders such as TMDs. This is because the stress itself can produce hyperalgesia or analgesia, which raises the question of appropriate comparison groups that consist not only of healthy controls but of healthy controls with similar self-reported stress levels. Furthermore, while there is evidence that patients with a TMD report higher levels of anxiety and depression on standardized
tools, these studies often fail to measure such factors as individual resiliency traits, support networks, and coping strategies that may blunt activation of the HPA axis. Research priorities are highlighted in Box 4-5.
Sex Differences and Painful TMDs
Systematic reviews and meta-analyses indicate that the prevalence of TMDs is more than twice as great in women as in men (odds ratio of 2.24 for combined TMD groups) in all diagnostic groups of the Research Diagnostic Criteria for Temporomandibular Disorders (Axis I: groups I, II, and III) (Bueno et al., 2018), and current evidence suggests that sex hormones may play some role in the pathophysiology of TMDs (see Chapter 3). The increased prevalence of TMDs in women of reproductive age, along with a low prevalence in childhood, suggest that sex hormones such as estrogen may play a role in the pathophysiology of TMD. Studies comparing women with TMDs with controls indicate that the former have a hyperinflammatory phenotype characterized by an increased release of cytokines from circulating monocytes after an inflammatory insult, which was further increased by estrogen (Ribeiro-Dasilva et al., 2017). These preliminary observations suggest that women with a TMD may exhibit an estrogen-induced hyperinflammatory phenotype that may contribute to central sensitization. Early-life stress—an established risk factor for chronic pain—and estrous-cycle estrogen fluctuations have been linked to pain sensitivity and central sensitization of visceral pain in female rats (Moloney et al., 2016). Further research is needed in TMD animal models to determine how estrous cycles and early-life stress mediate pain sensitization. In addition, a recent study employing a TMJ arthritis model in rats reported a greater susceptibility to hypersensitivity and central sensitization in females versus males despite the females receiving a concentration (16.6 mg/mL) of injected monosodium iodoacetate that was only one-fifth
the concentration in the males (80 mg/mL). Female rats developed more widespread pain hypersensitivity following acute inflammation, suggesting that TMJ osteoarthritis may serve as a model for future studies of sex differences (Sannajust et al., 2019).
Additionally, there is some evidence that sex differences exist with regard to estrogen and neuropathic pain signaling. As described above, the voltage-gated sodium channel acts as the threshold channel for the firing of action potentials and is thought to play an important role in pain signaling. The effects of estrogen on the Nav1.7 sodium channel expression in the trigeminal ganglion have been examined in experimental models. In rodents, estradiol-enhanced TMJ inflammation induced the upregulation of Nav1.7 in the trigeminal ganglion (Bi et al., 2017). These observations are consistent with the hypothesis that estrogen enhances hyperalgesia of an inflamed TMJ by modulating the expression or channel threshold of Nav1.7 in the trigeminal ganglion.
There is also evidence of sex differences in spinal cord pain processing mediated by the NMDA receptor, with NMDA antagonism reducing nociceptive responses more in males than in females (Del Rivero et al., 2019). Building off prior research indicating that the blockage of TNF receptor 1 (TNFR1) signaling in mice resulted in a failure to develop neuropathic pain or depressive symptoms following chronic constriction injury, Del Rivero and colleagues (2019) tested the therapeutic efficacy of a drug targeted to inactivate TNF. The results indicated that only male mice experienced accelerated recovery from neuropathic pain following administration of the therapeutic agent, while females did not, suggesting that TFNR1 signaling is associated with pain following nerve injury in males but not in females. The failure of the agent to work in female mice is thought to be due to inhibition of TFNR1 signaling and decreased NMDA receptor levels following injury (Del Rivero et al., 2019). While this specific mechanistic difference is valuable in its own right, this dichotomy in response between the sexes indicates more broadly the importance of conducting research into sex-specific mechanisms of pain as a critical component in the development of effective therapeutics for TMDs.
Looking Forward
Sex differences play a significant role in the mechanisms underlying chronic pain and will require additional focused study if safe and effective treatments targeted for high-risk groups, such as females, are to be developed. Animal models specifically designed to explore sex differences in the development of chronic pain, such as the methodology used by Sannajust and colleagues (2019), are needed for TMDs (see Box 4-6).
BIOMARKERS AND MOLECULAR GENETICS OF TMDs
The role of genetics in TMD pathophysiology is an area of increasing research interest due in part to the growing body of literature on genetic variants associated with similarly multifactorial musculoskeletal and chronic pain conditions (Meloto et al., 2011). In the case of chronic pain, genetic factors are thought to account for approximately half of the variability in pain sensitivity and risk of chronicity (Harper et al., 2016a). However, the contribution of genetics to pain is complex and likely occurs via the interactions of a network of single nucleotide polymorphisms (SNPs), rather than through an association with a single SNP (Meloto et al., 2018). Additionally, a variety of psychosocial and environmental factors and exposures likely affect these polygenetic susceptibilities toward or away from chronic pain, although why and how this occurs is not well established (Meloto et al., 2011; Zorina-Lichtenwalter et al., 2016). Many genes have been investigated to understand their influence on pain processing (see Box 4-7). Of the pain conditions studied, musculoskeletal disorders (TMDs, low back pain, fibromyalgia, and chronic widespread pain) and migraines have received the greatest amount of investigation and have been associated with the greatest number of genetic variants (Zorina-Lichtenwalter et al., 2016). A more complete understanding of the genetic basis of TMDs and overlapping genetic associations with commonly comorbid conditions could provide clarity on etiology, an improved understanding of orofacial pain mechanisms, and ultimately improve clinical care. This area of research is one in which the technologies and methodologies are changing rapidly, and it will be critically important for TMD research to stay on the cutting edge of research advances.
Targeted genotyping studies conducted over the past decade have identified a number of genetic variants that may be associated with TMDs and orofacial pain. A 2016 review of studies of genetic predictors of chronic pain conditions found 36 genes associated with TMDs, including several genes that are also associated with other pain conditions (Zorina-Lichtenwalter et al., 2016). Of those genetic variants thought to play a contributing role in TMD pathophysiology, catechol-O-methyltransferase (COMT) is one that has been well studied as a contributor to chronic pain (Mogil, 2012). Mechanistic studies show that alterations in COMT activity modulate the beta-adrenergic receptors, which in turn stimulate proinflammatory cytokine production (Nackley et al., 2007). Other possible genetic variants identified involve the adrenergic (ADRA2C, ADRA1D) (Smith et al., 2011), estrogenic (ESR1) (Ribeiro-Dasilva et al., 2009), and serotonergic systems (HTR2A, SLC6A4) (Ojima et al., 2007; de Freitas et al., 2013). Additionally, possible associations between a genetic variant in IL-10, a proinflammatory cytokine, and TMD have been suggested (Smith et al., 2011).
The OPPERA study—the largest study to date of the genetic risk factors for TMDs—has also gathered extensive phenotypic information from individuals before and after acute onset of a TMD and also those individuals who transition from acute to chronic pain (see Chapter 3). In their 2011 publication describing findings from the OPPERA case–control study, OPPERA researchers identified several potential genetic risk factors associated with TMDs; specifically, seven SNPs were found to be associated with pain perception, affective processes, and inflammation: COMT, HTR2A, NR3C1, CAMK4, CHRM2, IFRD1, and GRK5 (Smith et al., 2011). The 2013 follow-on study, which was a prospective cohort study, found no SNPs to be significantly associated with the initial onset of a
TMD; however, significant associations were noted in SCN1A and ACE2 with non-painful orofacial symptoms. Mutations in SCN1A and ACE2 have been associated with changes in pain processing (Smith et al., 2013).
Several genetic variants associated with TMDs have also been implicated in targeted genetic association studies for other chronic pain conditions, although the meaning of these overlapping genetic variants has yet to be unraveled. COMT, as mentioned above, has been associated with fibromyalgia (Vargas-Alarcón et al., 2007) and stress-induced chronic pain (McLean, 2011), while ADRB2, an adrenergic mechanism, has been reported in genetic association studies of fibromyalgia (Vargas-Alarcón et al., 2009) and low back pain (Skouen et al., 2012). Further research is necessary to identify and investigate how these polygenetic associations relate to the pathophysiology of TMDs and comorbid conditions.
Studies of genetic associations with TMDs encounter several challenges. Most significant is the lack of studies using large populations. With the exception of OPPERA, most studies have had small participant sizes and used targeted genotyping approaches, making it difficult to identify novel genes associated with TMDs and potential biomarkers. Additional exploratory research looking across the entire genome (e.g., using next-generation sequencing and genome-wide association studies) may facilitate a better understanding of the genetic architecture of TMDs and those of other pain conditions.
Biomarkers for TMDs
Researchers face multiple challenges in developing safe and effective therapies for TMDs. These challenges include the current limited understanding of the mechanisms of TMDs, a lack of and poor translation of preclinical and clinical data, too few robust clinical trials, and a lack of validated biomarkers to predict treatment response and stratify patients into clinically meaningful and mechanistically based subgroups (Harper et al., 2016a; Doshi et al., 2020). (See Box 4-8 for descriptions of biomarkers and their uses.) Other clinical fields, such as oncology and cardiovascular and metabolic diseases, have demonstrated the value of biomarkers in predicting treatment response (Ferber, 2002).
Several studies have targeted potential TMD biomarkers for investigation; however, most of these studies featured very small sample sizes and have not been replicated in other populations. In a case–control study (n=30) of plasma levels of dopamine and serotonin, Dawson and colleagues (2016) found significantly higher levels of dopamine in individuals with a painful TMD than in health controls, as well as a correlation between heightened levels of dopamine and increased pain intensity and perceived stress. However, further research is needed to understand the function of
the dopamine pathway as it relates to TMDs and to determine whether it could be clinically valuable as a biomarker (Dawson et al., 2016).
Cytokines have also been indicated as a potential diagnostic biomarker for TMDs because of the mechanistic role that pro-inflammatory cytokines play in initiating an immune response and pain induction and the suggested involvement of cytokines in the transition from acute to chronic pain.
Furthermore, past research has indicated that cytokine levels are elevated within the TMJ of individuals with TMDs (Kaneyama et al., 2002; Matsumoto et al., 2006; Kobayashi et al., 2017) and points to an association between cytokines and increased pain sensitivity. A heightened production of cytokine levels has also been associated with psychosocial factors, such as perceived stress (Maes et al., 1998) and depression (Maes, 1999), which are associated with painful TMDs. A 2011 case–control study (n=344) found that “localized and anatomically widespread patterns of chronic pain are associated with distinctive profiles of inflammatory biomarkers at protein, transcription factor activity, and gene levels” (Slade et al., 2011, p. 12). Specifically, localized TMDs were associated with an anti-inflammatory cytokine, IL-1RA, and TMDs with widespread palpation tenderness were associated with another cytokine, IL-8. Such findings suggest that cytokines could function as diagnostic biomarkers in the future (Slade et al., 2011; Kobayashi et al., 2017), although additional research is needed. Other inflammatory mediators such as 5-HT, TNFα, and prostaglandins have been explored as potential biomarkers for orofacial pain because of their presence in the synovial fluid of inflamed joints and their absence in healthy individuals, as have certain neuropeptides such as substance P and CGRP (Sessle, 2011).
Unfortunately, the field of TMD research and clinical care does not yet have rigorously validated biomarkers (Nagakura, 2017). It is unlikely that a single biomarker for TMDs will be found due to the complex biopsychosocial nature of TMDs and pain, which cannot be broken down into distinct, independent components. At this point, there are no confirmed biomarkers for TMDs, although there are a number of inflammatory markers that have been suggested as potential biomarkers.
Application of Novel Approaches to the Study of TMDs
The application of omics—genomics, proteomics, transcriptomics, metabolomics, etc.—approaches offers the possibility to collect large amounts of data and map molecular patterns within complex disorders (Hasin et al., 2017) like TMDs and other chronic pain conditions. These data, with proper translation, aid in the ability to identify clinically meaningful biomarkers, stratify patients based on mechanistically relevant factors rather than by diagnoses, and identify therapeutic targets (Gazerani and Vinterhøj, 2016), among others. The clinical value of omics research has been demonstrated. In their 2011 review of genomics research on TMDs, Meloto and colleagues point to pharmacogenomics research that described the outsized role of genetic factors (specifically two genes) on the optimal dosing of the anticoagulant warfarin as an example of similar research that could be conducted for TMDs. Currently, the literature contains few
examinations of blood, synovial fluid, or tissue from human subjects with TMDs that use large-scale, non-targeted approaches in the areas of genomics, epigenetics, proteomics, lipidomics, immune profiling, transcriptomics, metabolomics, and immunophenotyping, despite such approaches having resulted in key scientific breakthroughs in other fields.
Omics approaches are now being applied to the field of pain research through the exploration of DNA, RNA, protein, and metabolic changes in both animal models and in human subjects (Gomez-Varela and Schmidt, 2018); some of the findings of those studies hold potential value for TMD research. Proteomic analysis, which provides data on the expression, function, and regulation of proteins, can provide insights into disease pathophysiology, biomarkers, and treatment response (Gazerani and Vinterhøj, 2016). A proteomic analysis of urine from women with IBS, a chronic pain-related condition that is frequently comorbid with TMD, highlighted several possible protein differences between well-phenotyped subgroups of IBS patients and healthy controls (Goo et al., 2012). This initial analysis found 18 proteins that differed between participants with IBS and healthy controls, and a follow-up study with one of these proteins, trefoil factor 3, which is known to play an important role in gut barrier protection, revealed strong associations between this protein and fecal microbiome taxa (Heitkemper et al., 2018). Similarly, among those with chronic widespread pain, a chronic overlapping pain condition often found in those with fibromyalgia, muscle biopsies identified 17 proteins that were different from those in healthy controls and were associated with metabolism, muscle damage, stress, and inflammation (Olausson et al., 2015). These approaches have also been applied in the Multidisciplinary Approach to the Study of Chronic Pelvic Pain (MAPP) Research Network study, a longitudinal analysis following chronic pelvic pain, which used pain testing and clinical phenotyping to understand the complexities of symptom flares over time (Harte et al., 2019).
Metabolomics is another emerging area of research in the study of chronic pain that could provide insights for TMD research. This field investigates the molecular products of the metabolic process within fluids and tissues and can provide data on cellular states and phenotypes (Gazerani and Vinterhøj, 2016). In their review, Gazerani and Vinterhøj (2016) explain that metabolomics can provide mechanistic insight into the relationship between disease phenotypes and biochemical changes. Metabolomic techniques have been used to identify a marker of neuropathic pain in rats (Patti et al., 2012), an indication of its potential as a future tool to differentiate pain types and inform treatment (Gazerani and Vinterhøj, 2016).
Omics approaches such as those described above would be valuable in the study of TMDs, particularly for understanding shared mechanisms with other chronic overlapping conditions, peripheral and CNS responses, and novel mechanistic and therapeutic targets.
Using these novel methods, researchers could explore how certain genetic loci and non-coding mutations relate to immunoprofiles in patients with clinically defined TMD phenotypes. Through the use of bioinformatics and pathway analyses, polygenic risk scores can also be examined. Furthermore, sophisticated gene-editing techniques (e.g., CRISPR/Cas9), viral vector cell-specific manipulation techniques (e.g., lentivirus with cell-specific promotors), and cell manipulation techniques (e.g., optogenetics, designer receptors exclusively activated by designer drugs) could allow for a more detailed downstream analysis of multiple systems and pathways leading to the generation and maintenance of painful TMDs as well as other pain conditions. Additionally, standardized clinical phenotyping and documentation may allow for the identification of common exposures and stressors, which are not yet well understood.
Looking Forward: Future Areas of Omics Research for TMDs
A greater application of omics approaches—genomics, epigenomics, proteomics, metabolomics, transcriptomics, lipidomics, and immune profiles—to the study of TMDs is needed to understand TMD etiology and identify relevant biomarkers (see Box 4-9). Particularly of value would be the thoughtful integration of a selection of omics approaches to improve identification of patient subgroups and provide a more detailed understanding of potential targets for treatment. Gazerani and Vinterhøj
(2016, p. 262) note that the integration of proteomics and metabolomics into genomics research on pain “enhances [the] quality and validity of big data application in terms of efficacy and safety of approaches taken toward pain and its treatment.” Additionally, because TMDs often present with comorbid pain conditions, a focus on targets beyond TMDs could provide new mechanistic insights and potential therapeutic targets. The committee recommends carrying out non-targeted omics approaches to examine and compare local tissue with more systemic biomarkers in human subjects and animals. Once potential predictor, prognostic, and resilience markers have been identified in human subjects, these should be subsequently validated as therapeutic targets in animal models to aid translational research. Such translational studies act as bridging mechanisms and will be essential to pushing the research forward. These future TMD biomarkers can help drive the discovery of new therapies and define more targeted and personalized approaches to patient care based on a unique TMD “biosignature.” Such future biomarkers will need to address acute, chronic, and high-impact TMDs as well as vulnerability to the development of TMDs, recovery, and treatment outcomes. Combined with appropriate clinical endpoints, these biomarkers could help improve the classification of TMD subtypes and predict TMD progression. Patient stratification biomarkers—which could place individuals into clinically meaningful and mechanistically based subgroups—would be particularly useful in informing the design of clinical trials by improving patient selection and reducing expenses by allowing for more targeted and smaller clinical trials within these subgroups.
NEUROIMAGING OF THE CNS: EXPLORATION OF TMD PHENOTYPES
Neuroimaging has opened a window to the brain for the non-invasive study of both structure and function and has expanded understanding of how pain processing is linked to the CNS, how it is disrupted, and how those disruptions occur with the chronification of pain (Davis and Moayedi, 2013; Nash et al., 2013; Cowen et al., 2015; Martucci and Mackey, 2016, 2018; Martucci et al., 2019; Weber et al., 2018). Furthermore, neuroimaging allows a new perspective and a deeper understanding of the complex nature of chronic pain experience. This has led to the adoption of a whole-brain approach to the study and treatment of chronic pain and the development of novel technologies and analytic techniques, which could have major potential for the development of new diagnostics and more effective therapies. Various neuroimaging modalities have been used, including positron emission tomography (PET), electroencephalograpy (Diers et al., 2007), magnetoencephalography, single-photon emission computed tomography (Harisankar et al., 2012), and magnetic resonance imaging
(MRI). These techniques have been used to study several chronic pain states, including TMDs (Shibukawa et al., 2007; Younger et al., 2010; Gerstner et al., 2011; Moayedi et al., 2012; He et al., 2014, 2018; Lin, 2014; Monaco et al., 2015; Wilcox et al., 2015; Harper et al., 2016b; Mupparapu et al., 2019; Ozdiler et al., 2019), chronic low back pain (Ung et al., 2012), fibromyalgia (Staud, 2011a), osteoarthritis (Howard et al., 2012), complex regional pain syndrome (Schwenkreis et al., 2009; Barad et al., 2014), phantom-limb pain, chronic migraine (Chiapparini et al., 2010), chronic pelvic pain (Farmer et al., 2011; Kairys et al., 2015), and peripheral neuropathy (Moisset and Bouhassira, 2007), among others.
Structural Neuroimaging for TMDs
Researchers have found structural brain differences in gray matter density, gray matter volume, and cortical thickness, among other differences, between people with chronic TMD pain and healthy volunteers (Younger et al., 2010; Moayedi et al., 2011; Lin, 2014; Wilcox et al., 2015). Structural changes have been identified in both the brain (primary somatosensory cortex, cingulate cortex, thalamus, putamen, pallidus, anterior insula) and the brainstem (trigeminal sensory nuclei, medullary dorsal horn) (Younger et al., 2010; Moayedi et al., 2011; Lin, 2014; Wilcox et al., 2015). These findings suggest that there is an underlying structural plasticity within the brain and that changes in cellular composition within the brain may occur in individuals with a TMD. However, the underlying physiological changes that contribute to these differences in gray matter remain unknown. Researchers have hypothesized that changes in gray matter may be the result of changes in gray matter microstructure and the prevalence of glial and other supporting and neuroimmune cells within the brain, in addition to other possible mechanisms (Zatorre et al., 2012). Finally, researchers have investigated white matter abnormalities using diffusion tensor imaging to investigate TMDs. Moayedi and colleagues (2012) found that people with painful TMDs have lower fractional anisotropy in the bilateral trigeminal nerve and diffuse abnormalities in the microstructure of white matter tracts related to sensory, motor, cognitive, and pain functions. These structural MRI findings point to gray and white matter abnormalities in the brain and brainstem systems responsible for the experience and modulation of pain. While these associations have been well established, the causal role of these changes still must be determined. It is important to understand whether the brain changes result from TMDs or whether these differences confer a vulnerability that contributes to the development of a TMD or to the transition from acute to chronic forms of TMDs. Additionally, the reversibility of these changes with therapies is of interest.
Functional Neuroimaging for TMDs
Researchers have used neuroimaging techniques to study functional differences in the brain experiencing chronic pain, including TMDs, versus healthy states. Many of these studies used functional MRI (fMRI) techniques to investigate both abnormalities in brain function in TMDs and also the effect of treatment, specifically functional orthodontic treatments (He et al., 2014, 2018). Collectively, the functional neuroimaging research on pain points toward a heightened responsivity of the CNS to afferent noxious and innocuous stimuli in chronic pain. Additionally, more research with robust participant selection criteria and methodologies is needed to determine the extent that MRI brain-based biomarkers are useful for treatment prediction.
The emergence of resting state fMRI for studying non-evoked brain activity and functional connectivity has allowed many investigations of chronic pain to gain a broader understanding of brain processes, as opposed to only those processes related to noxious stimuli. Several studies applied resting-state fMRI to characterize differences in non-evoked (i.e., resting) brain activity among people with TMDs (He et al., 2018). Participants with TMDs exhibited reduced network functional connectivity, supporting the suggestion that TMDs are associated with reduced functional connectivities in brain corticostriatal networks and that these reduced functional connectivities may underlie motor control deficits, pain processing, and cognition in individuals with TMDs, although additional research is needed.
The Future of Neuroimaging and TMDs
Other advances in the field of pain neuroimaging include combining multiple neuroimaging modalities with large-scale, multi-site investigations. Neuroimaging researchers are increasingly using analytical methods that combine multiple neuroimaging modalities to understand chronic pain. For example, a study by Schrepf and colleagues (2016) used combined PET imaging and fMRI to identify increased µ-opioid receptor availability and evoked pain brain activity (blood oxygenation level dependent) co-occurring in the anterior insula of individuals with fibromyalgia (Schrepf et al., 2016). The neuroimaging of pain is also being included as a major component of large-scale, multi-site investigations that focus on idiopathic chronic pain conditions such as urological chronic pelvic pain (i.e., interstitial cystitis, chronic prostatitis, bladder pain syndrome) (Alger et al., 2016). These collaborative multi-site investigations are also including longitudinal investigations that illustrate changes in brain activity to track symptom profiles over time (Kutch et al., 2017b). Such multimodal, multi-site collaborations offer tremendous opportunities for the study of TMDs.
Future of Brain-Based Biomarkers of Pain and TMDs
MRI has opened a window to the brain by allowing for the noninvasive study of both structure and function and the validation of the role of the CNS in chronic pain. MRI offers a significant opportunity to identify and validate neuroimaging-based biomarkers and surrogate endpoints for pain. Preliminary brain biomarkers have been identified in individuals experiencing acute and chronic pain (Marquand et al., 2010; Brown et al., 2011; Brodersen et al., 2012; Wager et al., 2013; Bagarinao et al., 2014; Woo et al., 2015, 2017; Kutch et al., 2017a,b; Lopez-Sola et al., 2017; Cheng et al., 2018; Zhong et al., 2018). These biomarkers would be of value to clinical and research communities by aiding prognosis (Baliki et al., 2012; Mutso et al., 2014) and understanding pain progression (Mackey, 2014, 2016; Von Korff et al., 2016), predicting response to a treatment, ascertaining a diagnosis, identifying targets for treatment, and defining surrogate endpoints and predicting clinical benefit.
PSYCHOSOCIAL FACTORS UNDERLYING TMDs
The biopsychosocial model seeks to take into account all factors—biological, psychological, and social—that may play a role in the onset and progression of TMDs (see Chapter 2 for an overview of this approach). Unraveling the biological mechanisms underlying the pathophysiologies of TMDs has been the greatest focus of basic research thus far; however, the role of psychosocial factors has increasingly been elevated in importance as the biopsychosocial model is absorbed into research and care philosophies. Despite acknowledgment of the contributing role of psychosocial factors in the experience of pain, this area of research remains out of balance with the comparatively large body of research on biological mechanisms.
How psychosocial factors affect pain and tissue dysfunction associated with TMDs remains unknown, although the significant overlap in psychosocial risk factors between TMDs and other chronic pain conditions (see Chapter 3 for discussion of psychosocial risk factors) suggests that similar underpinnings may be at work. Psychosocial factors (i.e., anxiety, depression, negative affect, and symptom burden) and their impact on the generation, maintenance, suppression, and perception of pain have been studied across overlapping conditions like fibromyalgia (Giesecke et al., 2005), interstitial cystitis (Nickel et al., 2015), IBS, and headache (Kato et al., 2009). This small body of research suggests that, as it relates to the experience of chronic pain, certain psychosocial factors may mediate activity in the various parts of the brain responsible for processing the sensory versus the affective aspects of pain (Giesecke et al., 2005; Harper et al., 2016a).
There is some evidence that psychosocial factors may play a role in the amplification and maintenance of orofacial pain. In a study of 163 individuals with a TMD, those who score high on the pain catastrophizing scale had a six-fold increase in risk of developing persistent pain (Reiter et al., 2015). In another study, orofacial pain response was associated with depression in participants with TMDs (Sherman et al., 2004). The presence of high symptom burden was also associated with increased TMD incidence and decreased improvement in orofacial pain after 5 years (Ohrbach and Dworkin, 1998). These findings for orofacial pain support prior research by Giesecke and colleagues (2005), which demonstrated that individuals with fibromyalgia and depression experience increased pain amplification by the CNS compared with individuals with fibromyalgia but no reported psychological comorbidity.
Findings from the OPPERA study indicate that psychological distress and other somatic symptoms may act as predictive risk factors for developing a painful TMD (Fillingim et al., 2011). The OPPERA study extensively phenotyped individuals based on an array of biopsychosocial risk factors in individuals with and without TMDs (see Chapter 3). The outcome of this phenotyping was the identification of clusters across which symptoms and factors could be compared. Of the three clusters identified—adaptive, pain-sensitive, and global symptoms clusters—individuals in the global symptoms cluster presented with more psychological distress and greater pain sensitivity than the other two clusters. These findings point to the likely presence of multimodal predictors and a range of environmental, physiological, and psychological variables that each contribute to TMDs (Bair et al., 2016). The use of phenotypes that take into account biopsychosocial factors in a clinical care setting could add significant value to the care of patients with TMDs by targeting treatment and predicting outcomes based on the presenting phenotype of the patient; however, more research is required. It should be noted that temporomandibular pain can occur for multiple reasons, including nerve injury and joint conditions such as osteoarthritis, or be non-specific, and thus there may be different underlying mechanisms and combinations of mechanisms for each TMD and across individuals.
APPLICATION OF DATA SCIENCE METHODOLOGIES AND NOVEL TECHNOLOGIES TO TMD RESEARCH
The data science methodologies of machine learning, informatics, and artificial intelligence are increasingly used to unravel complex problems related to etiology, risk, prognosis, and treatment effectiveness. In pain research these methods have been used by researchers to better detect previously unseen patterns in data and to identify subgroups within the data that could inform future research and clinical care (Lötsch and Ultsch, 2018).
The value of these approaches is the ability to identify new clinically relevant and mechanistically based phenotypes within datasets that could indicate or predict response to treatment. Machine learning methods can also be used to identify previously unknown biopsychosocial parameters of complex conditions (Lötsch and Ultsch, 2018) such as TMDs and chronic pain. However, these methods require access to large-scale, high-quality datasets, which are lacking for TMDs (Lötsch and Ultsch, 2018). Useful data could be collected in several ways, including the addition of TMD-related questions to national surveys, the mining of insurance databases and electronic health records and International Classification of Diseases codes, and the creation of national or regional patient registries. The value of these approaches wholly relies on high-quality data inputs, and the research and clinical ecosystems need to be able to manage this. TMD research requires the adoption of these new technologies and approaches, like advanced data analysis, machine learning, artificial intelligence, clustering methods, expression quantitative trait loci (eQTL) analyses, polygenic approaches, and pathway analyses to unravel the complexities of this group of disorders, identify patient subgroups, and develop safe and effective treatments.
TMD RESEARCH FUNDING
Current Funding for TMD Basic Research and Translation
The National Institutes of Health (NIH) provides only about one-third of biomedical research funding in the United States (IOM, 2011), and its impact has a ripple effect that stimulates research interests and training programs across the nation. TMDs are not the primary mission of any NIH institute; however, funding for these disorders primarily falls within the National Institute of Dental and Craniofacial Research (NIDCR). In fiscal year (FY) 2018, NIH awarded approximately $13.7 million for TMD research. Of that amount, approximately $12 million came from NIDCR (NIH, 2019a). The other five institutes (National Institute of General Medical Sciences; National Institute of Neurological Disorders and Stroke; National Institute of Nursing Research; National Heart, Lung, and Blood Institute; and National Institute on Deafness and Other Communication Disorders) contributed remaining funding for TMDs in FY 2018 (NIH, 2019a). While funding estimates for FY 2019 and FY 2020 across NIH indicate a slight upward trend when compared to levels of funding for 2017 and 2018 (see Figure 4-1), recent discussions have highlighted challenges that need to be addressed to move the field forward (MDEpiNet TMJ Patient-Led RoundTable, 2018a,b).
In 2018, NIH’s TMD-targeted funding focused on topics such as cellular and mechanical mechanisms, genetics, emotion dysregulation, and
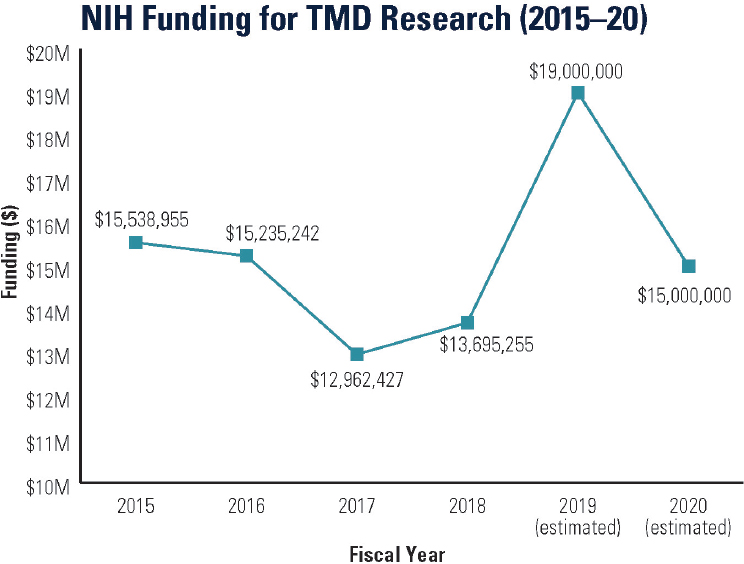
SOURCE: NIH, 2019a.
modulation of the µ-opioid mechanism. While a few projects studied multiple areas of TMD concurrently, most were narrowly focused (NIH, 2019a). The bulk of TMD research funding from NIH is for basic research, with clinical and translational research making up a smaller portion. Of the 40 studies identified under the “TMJD” spending category, 4 were listed as K99 grants (see Box 4-10 on funding mechanisms for research); however, other funding specifically related to training was not identified. The majority of NIH-awarded projects for TMD research in FY 2018 went to dental schools (approximately 40 percent), followed by medical schools (approximately 27.5 percent). However, the actual dollar amount for research was slightly higher for medical schools (NIH, 2019a) (see Figure 4-2). Regarding collaboration among institutions, Allen Cowley highlighted the need for increased collaboration specifically among dental and medical schools in the area of basic research and setting forth a purposefully integrated approach to research for TMDs (Cowley, 2019).
Increased funding for TMDs from across the NIH institutes and other biomedical organizations is essential to address the existing research gaps and to develop safe and effective treatments in the future. Additionally,
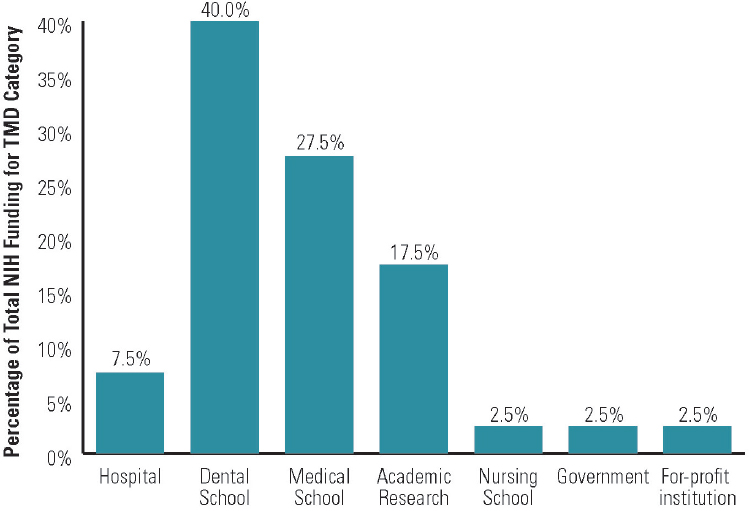
SOURCE: NIH, 2019a.
beyond the direct increase in funding earmarked for the exclusive study of TMDs, the incorporation of aspects of TMD research into the work of other NIH efforts—such as the NIH Pain Consortium—and institutes beyond NIDCR could also help to provide valuable insights into these disorders.
MOVING THE RESEARCH ENTERPRISE FORWARD
Clinicians cannot provide and patients cannot access safe and effective clinical treatment and care without a strong base of scientific evidence—from the basic sciences to implementation research. Despite the work accomplished in the past few decades, there remain significant research gaps and systematic challenges related to translation across the basic, clinical, and epidemiological sciences that are hindering the development of safe and effective treatments for individuals with TMDs and that indicate the need for greater research coordination and translation. Major areas of need, as identified in the State of the Science section above, include the biopsychosocial mechanisms underpinning acute and chronic orofacial pain and tissue dysfunction; the use of new methods and tools, including the application
of molecular and cellular approaches to understand TMD genetics and biomarkers associated with TMD diagnosis, prognosis, treatment outcome, and resilience; and a greater understanding of the tissues of the TMJ joint, its function, and the relationship between function and pain. Additionally, the committee noted the need for sophisticated and consistent outcome measures for assessment of TMD pain and function from preclinical models through to clinical research. It is critically important that pain measures be incorporated into translational models of TMJ degenerative diseases, but such incorporation is currently lacking.
The committee’s overview of the recent literature on TMDs revealed that a concerted effort to develop a more comprehensive foundation of clinically meaningful evidence around the pathophysiologies of TMDs will require a commitment to both the funding and the implementation of multidisciplinary research and to the purposeful transfer of knowledge across the research enterprise. Effectively addressing priority research areas will require a coordinated effort by a diverse group of stakeholders to develop and implement a patient-focused research agenda, cultivate a multidisciplinary research culture, and align system incentives to ensure that novel research findings are transferred from one stage of the research enterprise to the next. Similar efforts to define research priorities for complex disorders, such as the Federal Pain Research Strategy, have also sought to achieve these aims by providing an actionable research framework to guide and prioritize patient-focused research across the research enterprise. A description of the role of the proposed research consortium (see Recommendations 1 and 2 in Chapter 8) and the example research framework for TMD research, which unifies research priorities within patient-centered needs, can be found below.
Conclusion 4-1: The siloed approach to research on temporomandibular disorders (TMDs) has detrimental effects on translation across the research enterprise and limits access to the financial, educational, and intellectual resources needed to cultivate a robust research base. Addressing these challenges will require a multistakeholder collaboration to define patient-focused research priorities and implement systemic change to the conduct of TMD research to improve data quality and comparability, incentivize and support novel and collaborative research, and integrate evidence into clinical care and policy.
Elucidation of Biopsychosocial Mechanisms
The value of identifying and prescribing clinical meaning to biological mechanisms and pathways and understanding how these interact with other
biological, psychological, and environmental factors in relation to TMDs cannot be overstated. However, the value of understanding these mechanistic underpinnings goes beyond the benefit of the scientific knowledge itself; given access to the right tools and data, such an understanding could point to clinically meaningful and mechanistically based subgroups of individuals and inform targeted treatments. At the present time, the current state of our collective understanding of these mechanisms as they relate to subtypes of TMDs is insufficient to provide clinical value. Considerable time and resources have been invested in defining potential mechanisms; however, more research needs to be conducted to confirm the role of these mechanisms across other patient groups and within other TMDs. This includes determining the biopsychosocial mechanisms of resilience to both orofacial pain and tissue dysfunction. Additionally, because preclinical animal models demonstrate that such mechanisms may prove to be meaningful clinical targets, additional studies in the translational and clinical research should be carried out to capitalize on the potential for diagnostic, prognostic, or therapeutic value.
Conclusion 4-2: Elucidating the biopsychosocial mechanisms behind the generation, maintenance, and suppression of temporomandibular disorder (TMD)-related pain and dysfunction will be essential to understanding the complex pathophysiologies within TMDs, identifying clinically meaningful and mechanistically valid patient subgroups, and developing safe, effective, and personalized therapeutics and treatments. Doing so will require expanding current approaches and the use of innovative research methods and tools.
Application of Novel Methods and Tools
The study of TMDs would significantly benefit from the development of novel tools and approaches to understand all aspects of TMDs and from the application of concepts, methods, and tools already being applied in other fields. There exists a multitude of areas where the use of new tools or approaches could provide value. The advancement of our understanding of TMDs would be enhanced by the incorporation of newer molecular and cellular approaches targeting RNA, DNA, and the epigenome, metabolome, and proteome as well as cell-specific activation and silencing techniques. These approaches should be applied to the study of TMDs using relevant animal models and should translate to carefully phenotyped human subjects using tissue and blood samples as appropriate. A better understanding of the mechanisms that lead to development of acute and chronic pain, of the peripheral and CNS responses to nociceptive input, and of long-term changes will be critical. Sophisticated gene-editing techniques (e.g.,
CRISPR/Cas9), viral vector cell-specific manipulation techniques (e.g., lentivirus with cell-specific promotors), and cell manipulation techniques (e.g., optogenetics and designer receptors exclusively activated by designer drugs) will allow for a more detailed analysis of multiple systems and their role in the generation and maintenance of TMD and other pain conditions.
Because TMDs, when chronic, often present with comorbidities and multi-system components, a focus on targets outside the TMD regions or a systemic focus could provide novel mechanistic insights. The use of non-targeted approaches examining and comparing local tissue with more systemic biomarkers using a variety of approaches in human subjects and animals is recommended. These approaches could include genomics, epigenomics, proteomics, transcriptomics, lipidomics, and immune profiles. Potential predictors and resilience markers should be identified in human subjects and subsequently validated as therapeutic targets in animal models. Translational studies bridging mechanisms between animal and human TMD pain mechanisms will be critical to moving the field forward by identifying clinically meaningful and mechanistically based patient subgroups and developing new therapeutic approaches.
Research on TMJ Development, Biomechanical Function, and Relationship with Pain
To advance the understanding of TMJ function in health and disease, contemporary, multidisciplinary research focused on the biology of TMJ tissues, the interactions of these tissues, the innervation of the musculoskeletal component and vascularization of the joint, and the normal developmental processes and disease progression will be key research areas for expansion. Additionally, while bone/cartilage/ligament turnover and repair have been studied to some extent, a better understanding of the TMJ repair processes will be needed to integrate mechanical cues and pathological loading of the joint into bone/cartilage/ligament remodeling activities. Valuable information from these explorations and from the development and use of new in vitro and in vivo models could then inform the design and testing of new scaffolds and materials for regenerative medicine approaches. Most critically and where possible, the focus of these multiple areas of research should be on the translation from small-animal models to large-animal models and ultimately to humans.
A NATIONAL COLLABORATIVE RESEARCH CONSORTIUM AND FRAMEWORK FOR ACTION
Any research study of complex disorders, such as TMDs, must be considered from the perspective of the full biopsychosocial framework.
Consequently, studying TMDs from only a singular research perspective has been insufficient to fully understand the etiology of TMDs and how these disorders affect patients’ lives (MDEpiNet TMD Patient-Led RoundTable, 2018b; Cowley, 2019). The available body of recent laboratory research overviewed in this chapter related to TMDs and orofacial pain illustrates this issue, with many studies focusing on only one aspect of a TMD or conducting highly discipline-specific research. For example, dentists may study TMDs as a jaw issue, psychologists may exclusively consider the mental health comorbidities of TMDs, and medical researchers may study the mechanics of pain modulation, all of them without considering the larger biopsychosocial considerations of TMDs. This is not to say that these individual studies are not valuable—each plays a role in growing the scientific evidence base related to TMDs—but each alone has proven to be insufficient for developing a full understanding of TMD pathophysiologies. The 2011 Institute of Medicine (IOM) report Relieving Pain in America acknowledged the need for collaborative research in the field of pain and concluded that “research is needed to document and assess this full spectrum” of associated problems with acute and chronic pain and that this type of integrated, collaborative research would “enable the development of interventions that would address all aspects of the pain condition” (IOM, 2011, p. 228). In his remarks to the committee, Allen Cowley echoed the need for this approach for research on TMDs and emphasized the paltry research collaborations, particularly in the basic sciences, between dental and medical school researchers, adding that efforts to stimulate such an integrated approach to research on TMDs are lacking. He went on to say that basic research in dental schools would greatly benefit from collaborations with medical school faculty, schools of bioengineering, pharmacy schools, and other related schools (Cowley, 2019).
The 2011 IOM report also noted that research more broadly will increasingly require bioinformatics to aid in the analysis and interpretation of large datasets and that the field requires more scientists from diverse backgrounds (IOM, 2011). This underlines the point that a successful transdisciplinary research agenda would require the cultivation and engagement of both young and established researchers from across dental, medical, bioengineering, neurology, physical therapy, nursing, and psychology who have skills in a broad array of research and data techniques. This will in turn require a lasting financial commitment to research training and funding for research. Several existing funding structures within NIH are well positioned to do this, but funds are not currently earmarked for research on TMDs. (See Box 4-10 for examples of applicable training and research grants for developing a research workforce.)
Additionally, given the significant areas of research overlap between TMDs and other conditions, the TMD research enterprise is well positioned
to align itself within complementary research agendas and initiatives, such as the National Pain Strategy, to access both scientific and clinical knowledge and funding. Funding research on pain is one major area in which NIH can, without significant additional financial cost, expand the evidence base for TMDs by facilitating the transfer of potentially relevant findings from the pain field to researchers engaged in existing TMD research and allowing greater opportunities for researchers involved in pain research to conduct novel research on TMDs. To illustrate this point, in FY 2018 NIH reported funding approximately $605 million within the category of “pain research” and $474 million within the category of “chronic pain” (NIH, 2019a)—as compared to just more than $14 million for TMDs. TMD patients deal with both acute and chronic pain, and the research in this area may benefit patients, even when it is not specifically focused on TMDs (Cowley, 2019).
Conclusion 4-3: Research on temporomandibular disorders (TMDs) would benefit from alignment with an established research agenda with continuity of oversight, financial support, and a collaborative institutional structure that can guide a research agenda and support the integration into clinical practice. A TMD Research Consortium would be well positioned to convene stakeholders and launch a TMD research framework.
Conclusion 4-4: The level and appropriation of funding for research on temporomandibular disorders does not reflect the complexities of these disorders, their prevalence and impact, or the need for transdisciplinary research with a focus on clinical care impacts.
Role of a TMD Research Consortium and Example Research Framework for TMDs
A unified, transdisciplinary research framework for TMDs developed and implemented by a national collaborative research consortium (see Figure 4-3) could address these barriers by embedding a commitment to and the tools needed for the translation of research across disciplines and the research enterprise into the fabric of the framework and drive the development of a research program designed to address patient needs (see Box 4-11). There have already been calls for the implementation of a comprehensive research agenda that addresses the needs of TMD research. The TMJ Patient-Led RoundTable, a public–private partnership within the Food and Drug Administration composed of a variety of stakeholders, has taken the first steps in formalizing a proposed interagency research plan and has called repeatedly for a transformation of TMD research (MDEpiNet TMD Patient-Led RoundTable, 2018a).
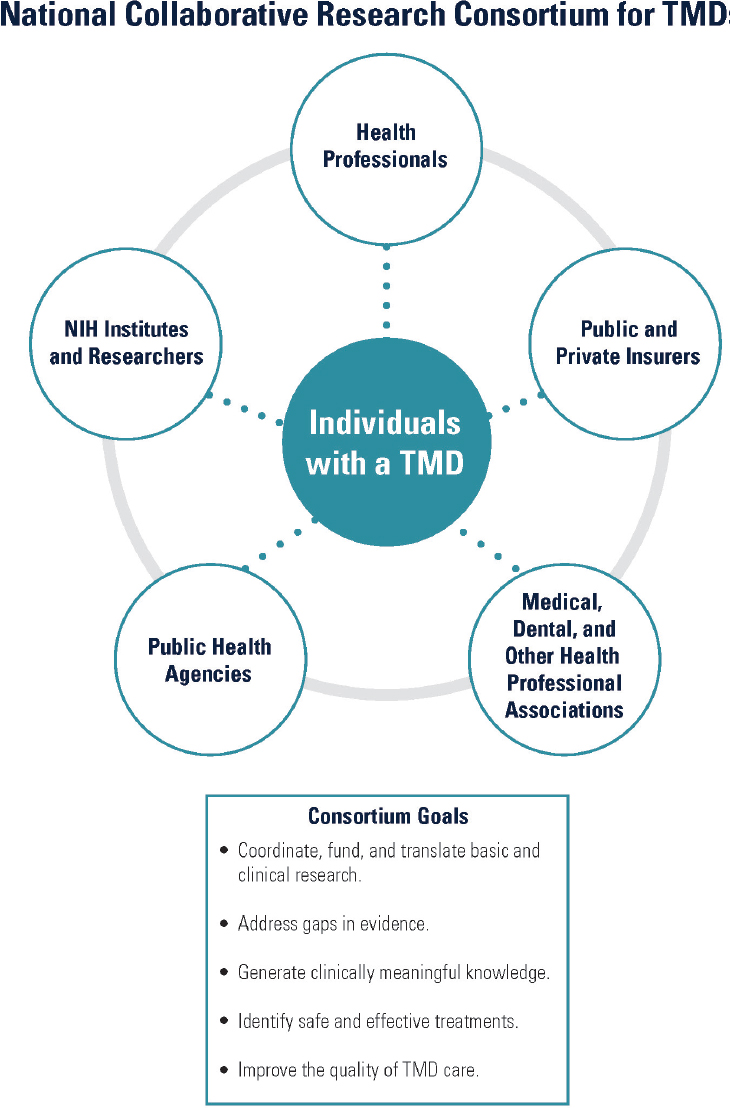
NOTE: NIH = National Institutes of Health; TMD = temporomandibular disorder.
Using the work of the NIH Pain Consortium and the TMJ Patient-Led RoundTable as a foundation, the committee developed an example research framework for TMDs to illustrate the broad range of interrelated research priorities that need to be addressed by the research consortium across the research-to-clinical-care continuum and to illustrate that the potential research overlaps with more broadly funded health concerns such as chronic pain and to emphasize the importance of keeping patient needs central to the process of research. The committee acknowledges that any future research frameworks for TMDs should be developed in collaboration with all essential stakeholders prior to implementation to ensure that the priorities and goals are supported by those stakeholder groups. Additionally, key stakeholder involvement will be needed to establish a realistic timeline, to secure the necessary buy-in as well as formal and informal agreements, to develop incentives, and to confirm that milestones are achievable and relevant.
In developing this example research framework for TMDs (see Box 4-12), the committee identified five broad goals related to access to safe and effective evidence-based treatment and care of those with TMDs. With these patient-focused clinical goals as a guide, the committee identified the essential research priorities—both short and long term—that must be addressed. This example research framework spans the content covered by the report and touches many of the research priorities and conclusions highlighted in the report’s chapters.
CONCLUSIONS
Conclusion 4-1: The siloed approach to research on temporomandibular disorders (TMDs) has detrimental effects on translation across the research enterprise and limits access to the financial, educational, and intellectual resources needed to cultivate a robust research base. Addressing these challenges will require a multistakeholder collaboration to define patient-focused research priorities and implement systemic change to the conduct of TMD research to improve data quality and comparability, incentivize and support novel and collaborative research, and integrate evidence into clinical care and policy.
Conclusion 4-2: Elucidating the biopsychosocial mechanisms behind the generation, maintenance, and suppression of temporomandibular disorder (TMD)-related pain and dysfunction will be essential to understanding the complex pathophysiologies within TMDs, identifying clinically meaningful and mechanistically valid patient subgroups, and developing safe, effective, and personalized therapeutics and treatments. Doing so will require expanding current approaches and the use of innovative research methods and tools.
Conclusion 4-3: Research on temporomandibular disorders (TMDs) would benefit from alignment with an established research agenda with continuity of oversight, financial support, and a collaborative institutional structure that can guide a research agenda and support the integration into clinical practice. A TMD Research Consortium would be well positioned to convene stakeholders and launch a TMD research framework.
Conclusion 4-4: The level and appropriation of funding for research on temporomandibular disorders does not reflect the complexities of these disorders, their prevalence and impact, or the need for transdisciplinary research with a focus on clinical care impacts.
REFERENCES
Adães, S., M. Mendonça, T. N. Santos, J. M. Castro-Lopes, J. Ferreira-Gomes, and F. L. Neto. 2014. Intra-articular injection of collagenase in the knee of rats as an alternative model to study nociception associated with osteoarthritis. Arthritis Research & Therapy 16(1):R10.
Adler, G. K., V. F. Manfredsdottir, and K. W. Creskoff. 2002. Neuroendocrine abnormalities in fibromyalgia. Current Pain Headache Reports 6(4):289–298.
Alger, J. R., B. M. Ellingson, C. Ashe-McNalley, D. C. Woodworth, J. S. Labus, M. Farmer, L. Huang, A. V. Apkarian, K. A. Johnson, S. C. Mackey, T. J. Ness, G. Deutsch, R. E. Harris, D. J. Clauw, G. H. Glover, T. B. Parrish, J. Hollander, J. W. Kusek, C. Mullins, E. A. Mayer, and MAPP Research Network Investigators. 2016. Multisite, multimodal neuroimaging of chronic urological pelvic pain: Methodology of the MAPP Research Network. Neuroimage Clincal 12:65–77.
Almarza, A. J., B. N. Brown, B. Arzi, D. F. Angelo, W. Chung, S. F. Badylak, and M. Detamore. 2018. Preclinical animal models for temporomandibular joint tissue engineering. Tissue Engineering—Part B, Reviews 24(3):171–178.
Almarza, A., L. Mercuri, B. Arzi, L. M. Gallo, E. Granquist, S. Kapila, and M. Detamore. 2019. State of TMJ bioengineering: Working together toward improving clinical outcomes. Journal of Biomechanical Engineering [Epub ahead of print].
Alvarez, P., P. G. Green, and J. D. Levine. 2013. Stress in the adult rat exacerbates muscle pain induced by early-life stress. Biological Psychiatry 74(9):688–695.
Amaya, F., H. Wang, M. Costigan, A. J. Allchorne, J. P. Hatcher, J. Egerton, T. Stean, V. Morisset, D. Grose, M. J. Gunthorpe, I. P. Chessell, S. Tate, P. J. Green, and C. J. Woolf. 2006. The voltage-gated sodium channel Na(v)1.9 is an effector of peripheral inflammatory pain hypersensitivity. Journal of Neuroscience 26(50):12852–12860.
Angelo, D. F., P. Morouco, N. Alves, T. Viana, F. Santos, R. Gonzalez, F. Monje, D. Macias, B. Carrapico, R. Sousa, S. Cavaco-Goncalves, F. Salvado, C. Peleteiro, and M. Pinho. 2016. Choosing sheep (Ovis aries) as animal model for temporomandibular joint research: Morphological, histological and biomechanical characterization of the joint disc. Morphologie 100(331):223–233.
Araújo-Filho, H. G., E. W. M. Pereira, A. R. Campos, L. J. Quintans-Júnior, and J. S. S. Quintans. 2018. Chronic orofacial pain animal models—Progress and challenges. Expert Opinion on Drug Discovery 13(10):949–964.
Bagarinao, E., K. A. Johnson, K. T. Martucci, E. Ichesco, M. A. Farmer, J. Labus, T. J. Ness, R. Harris, G. Deutsch, and A. V. Apkarian. 2014. Preliminary structural MRI based brain classification of chronic pelvic pain: A MAPP Network study. Pain 155(12):2502–2509.
Bair, E., S. Gaynor, G. D. Slade, R. Ohrbach, R. B. Fillingim, J. D. Greenspan, R. Dubner, S. B. Smith, L. Diatchenko, and W. Maixner. 2016. Identification of clusters of individuals relevant to temporomandibular disorders and other chronic pain conditions: The OPPERA study. Pain 157(6):1266–1278.
Baliki, M. N., B. Petre, S. Torbey, K. M. Herrmann, L. Huang, T. J. Schnitzer, H. L. Fields, and A. V. Apkarian. 2012. Corticostriatal functional connectivity predicts transition to chronic back pain. Nature Neuroscience 15(8):1117–1119.
Barad, M. J., T. Ueno, J. Younger, N. Chatterjee, and S. Mackey. 2014. Complex regional pain syndrome is associated with structural abnormalities in pain-related regions of the human brain. Journal of Pain 15(2):197–203.
Beatty, M. W., R. H. Hohl, J. C. Nickel, L. R. Iwasaki, and R. M. Pidaparti. 2008. Mode I and mode III fractures in intermediate zone of full-thickness porcine temporomandibular joint discs. Annals Biomedical Engineering 36(5):801–812.
Bechtold, T. E., C. Saunders, R. S. Decker, H.-B. Um, N. Cottingham, I. Salhab, N. Kurio, P. C. Billings, M. Pacifici, H.-D. Nah, and E. Koyama. 2016. Osteophyte formation and matrix mineralization in a TMJ osteoarthritis mouse model are associated with ectopic hedgehog signaling. Matrix Biology 52–54:339–354.
Bermejo, A., O. Gonzalez, and J. M. Gonzalez. 1993. The pig as an animal model for experimentation on the temporomandibular articular complex. Oral Surgery, Oral Medicine, and Oral Pathology 75(1):18–23.
Bi, R. Y., X. X. Kou, Z. Meng, X. D. Wang, Y. Ding, and Y. H. Gan. 2013. Involvement of trigeminal ganglionic Nav1.7 in hyperalgesia of inflamed temporomandibular joint is dependent on ERK1/2 phosphorylation of glial cells in rats. European Journal of Pain 17(7):983–994.
Bi, R. Y., Z. Meng, P. Zhang, X. D. Wang, Y. Ding, and Y. H. Gan. 2017. Estradiol upregulates voltage-gated sodium channel 1.7 in trigeminal ganglion contributing to hyperalgesia of inflamed TMJ. PLoS One 12(6):e0178589.
Brinkman, D. J., A. S. Ten Hove, M. J. Vervoordeldonk, M. D. Luyer, and W. J. de Jonge. 2019. Neuroimmune interactions in the gut and their significance for intestinal immunity. Cells 8(7):670.
Brodersen, K. H., K. Wiech, E. I. Lomakina, C. S. Lin, J. M. Buhmann, U. Bingel, M. Ploner, K. E. Stephan, and I. Tracey. 2012. Decoding the perception of pain from fMRI using multivariate pattern analysis. Neuroimage 63(3):1162–1170.
Brown, J. E., N. Chatterjee, J. Younger, and S. Mackey. 2011. Towards a physiology-based measure of pain: Patterns of human brain activity distinguish painful from non-painful thermal stimulation. PLoS One 6(9):e24124.
Bueno, C. H., D. D. Pereira, M. P. Pattussi, P. K. Grossi, and M. L. Grossi. 2018. Gender differences in temporomandibular disorders in adult populational studies: A systematic review and meta-analysis. Journal of Oral Rehabilitation 45(9):720–729.
Cady, R. J., J. R. Glenn, K. M. Smith, and P. L. Durham. 2011. Calcitonin gene-related peptide promotes cellular changes in trigeminal neurons and glia implicated in peripheral and central sensitization. Molecular Pain 7:1–12.
Cairns, B. E. 2010. Pathophysiology of TMD pain—basic mechanisms and their implications for pharmacotherapy. Journal of Oral Rehabilitation 37(6):391–410.
Campaniello, M. A., A. M. Harrington, C. M. Martin, L. Ashley Blackshaw, S. M. Brierley, and P. A. Hughes. 2016. Activation of colo-rectal high-threshold afferent nerves by Interleukin-2 is tetrodotoxin-sensitive and upregulated in a mouse model of chronic visceral hypersensitivity. Neurogastroenterology & Motility 28(1):54–63.
Castrillon, E. E., M. Ernberg, B. E. Cairns, K. Wang, B. J. Sessle, L. Arendt-Nielsen, and P. Svensson. 2010. Interstitial glutamate concentration is elevated in the masseter muscle of myofascial temporomandibular disorder patients. Journal of Orofacial Pain 24(4):350–360.
Chavan, S. S., V. A. Pavlov, and K. J. Tracey. 2017. Mechanisms and therapeutic relevance of neuro-immune communication. Immunity 46(6):927–942.
Chen, X., P. G. Green, and J. D. Levine. 2011. Stress enhances muscle nociceptor activity in the rat. Neuroscience 185:166–173.
Chen, Y., S. H. Williams, A. L. McNulty, J. H. Hong, S. H. Lee, N. E. Rothfusz, P. K. Parekh, C. Moore, I. R. W. Gereau, A. B. Taylor, F. Wang, F. Guilak, and W. Liedtke. 2013. Temporomandibular joint pain: A critical role for Trpv4 in the trigeminal ganglion. Pain 154(8):1295–1304.
Cheng, J. C., A. Rogachov, K. S. Hemington, A. Kucyi, R. L. Bosma, M. A. Lindquist, R. D. Inman, and K. D. Davis. 2018. Multivariate machine learning distinguishes cross-network dynamic functional connectivity patterns in state and trait neuropathic pain. Pain 159(9):1764–1776.
Chiapparini, L., S. Ferraro, L. Grazzi, and G. Bussone. 2010. Neuroimaging in chronic migraine. Neurological Sciences 31(Suppl 1):S19–S22.
Chichorro, J. G., F. Porreca, and B. Sessle. 2017. Mechanisms of craniofacial pain. Cephalalgia 37(7):613–626.
Chin, A. R., J. Gao, Y. Wang, J. M. Taboas, and A. J. Almarza. 2018. Regenerative potential of various soft polymeric scaffolds in the temporomandibular joint condyle. Journal of Oral and Maxillofacial Surgery 76(9):2019–2026.
Chinthakanan, S., K. Laosuwan, P. Boonyawong, S. Kumfu, N. Chattipakorn, and S. C. Chattipakorn. 2018. Reduced heart rate variability and increased saliva cortisol in patients with TMD. Archives of Oral Biology 90:125–129.
Clauw, D. J. 2015. Diagnosing and treating chronic musculoskeletal pain based on the underlying mechanism(s). Best Practice & Research Clinical Rheumatology 29(1):6–19.
Clauw, D. J. 2019. Defining TMD for clinical care. Presentation at the March 28, 2019, public meeting of the National Academies of Sciences, Engineering, and Medicine’s Committee on Temporomandibular Disorders (TMDs): From Research Discoveries to Clinical Treatment, Washington, DC.
Coogan, J. S., D.-G. Kim, T. L. Bredbenner, and D. P. Nicolella. 2018. Determination of sex differences of human cadaveric mandibular condyles using statistical shape and trait modeling. Bone 106:35–41.
Coombs, M. C., X. She, T. R. Brown, E. H. Slate, J. S. Lee, and H. Yao. 2019. Temporomandibular joint condyle-disc morphometric sexual dimorphisms independent of skull scaling. Journal of Oral and Maxillofacial Surgery 77(11):2245–2257.
Costa, Y. M., P. C. Conti, F. A. de Faria, and L. R. Bonjardim. 2017. Temporomandibular disorders and painful comorbidities: Clinical association and underlying mechanisms. Oral Surgery, Oral Medicine, Oral Pathology, and Oral Radiology 123(3):288–297.
Costigan, M., I. Belfer, R. S. Griffin, F. Dai, L. B. Barrett, G. Coppola, T. Wu, C. Kiselycznyk, M. Poddar, Y. Lu, L. Diatchenko, S. Smith, E. J. Cobos, D. Zaykin, A. Allchorne, E. Gershon, J. Livneh, P. H. Shen, L. Nikolajsen, J. Karppinen, M. Männikkö, A. Kelempisioti, D. Goldman, W. Maixner, D. H. Geschwind, M. B. Max, Z. Seltzer, and C. J. Woolf. 2010. Multiple chronic pain states are associated with a common amino acid-changing allele in KCNS1. Brain 133(9):2519–2527.
Cowen, R., M. K. Stasiowska, H. Laycock, and C. Bantel. 2015. Assessing pain objectively: The use of physiological markers. Anaesthesia 70(7):828–847.
Cowley, A. 2019. TMD research over the years—Gaps, priorities, opportunities. Presentation at the March 28, 2019, public meeting of the National Academies of Sciences, Engineering, and Medicine’s Committee on Temporomandibular Disorders (TMDs): From Research Discoveries to Clinical Treatment, Washington, DC.
Cummins, T. R., P. L. Sheets, and S. G. Waxman. 2007. The roles of sodium channels in nociception: Implications for mechanisms of pain. Pain 131(3):243–257.
Davis, K. D., and M. Moayedi. 2013. Central mechanisms of pain revealed through functional and structural MRI. Journal of Neuroimmune Pharmacology 8(3):518–534.
Dawson, A., N. Stensson, B. Ghafouri, B. Gerdle, T. List, P. Svensson, and M. Ernberg. 2016. Dopamine in plasma—A biomarker for myofascial TMD pain? Journal of Headache and Pain 17(1).
de Freitas, L. V., A. C. Lopes, V. B. Piatto, and J. V. Maniglia. 2013. Association of temporomandibular dysfunction with the 102T-C polymorphism in the serotonin receptor gene in Brazilian patients. Archive of Medical Science 9(6):1013–1018.
de Sousa, L. M., J. M. Dos Santos Alves, C. da Silva Martins, K. M. A. Pereira, P. Goes, and D. V. Gondim. 2019. Immunoexpression of canonical Wnt and NK-κB signaling pathways in the temporomandibular joint of arthritic rats. Inflammation Research 68(10):889–900.
Del Rivero, T., R. Fischer, F. Yang, K. A. Swanson, and J. R. Bethea. 2019. Tumor necrosis factor receptor 1 inhibition is therapeutic for neuropathic pain in males but not in females. Pain 160(4):922–931.
Detamore, M. S., and K. A. Athanasiou. 2005. Evaluation of three growth factors for TMJ disc tissue engineering. Annals of Biomedical Engineering 33(3):383–390.
Detamore, M. S., K. A. Athanasiou, and J. Mao. 2007. A call to action for bioengineers and dental professionals: Directives for the future of TMJ bioengineering. Annals of Biomedical Engineering 35(8):1301–1311.
Diatchenko, L., A. G. Nackley, G. D. Slade, K. Bhalang, I. Belfer, M. B. Max, D. Goldman, and W. Maixner. 2006. Catechol-o-methyltransferase gene polymorphisms are associated with multiple pain-evoking stimuli. Pain 125(3):216–224.
Diers, M., C. Koeppe, E. Diesch, A. M. Stolle, R. Holzl, M. Schiltenwolf, K. van Ackern, and H. Flor. 2007. Central processing of acute muscle pain in chronic low back pain patients: An EEG mapping study. Journal of Clinical Neurophysiology 24(1):76–83.
Donahue, R. P., J. C. Hu, and K. A. Athanasiou. 2019. Remaining hurdles for tissue-engineering the temporomandibular joint disc. Trends in Molecular Medicine 25(3):241–256.
Dong, Y., G. Wu, T. Zhu, H. Chen, Y. Zhu, G. Zhu, F. Han, and H. Zhao. 2017. VEGF promotes cartilage angiogenesis by phospho-ERK1/2 activation of Dll4 signaling in temporomandibular joint osteoarthritis caused by chronic sleep disturbance in Wistar rats. Oncotarget 8(11):17849–17861.
Doshi, T. L., D. R. Nixdorf, C. M. Campbell, and S. N. Raja. 2020. Biomarkers in temporomandibular disorder and trigeminal neuralgia: A conceptual framework for understanding chronic pain. Canadian Journal of Pain 4(1):1–18.
Embree, M. C., M. Chen, S. Pylawka, D. Kong, G. M. Iwaoka, I. Kalajzic, H. Yao, C. Shi, D. Sun, T. J. Sheu, D. A. Koslovsky, A. Koch, and J. J. Mao. 2016. Exploiting endogenous fibrocartilage stem cells to regenerate cartilage and repair joint injury. Nature Communications 7:13073.
Erzurumlu, R. S., Y. Murakami, and F. M. Rijli. 2010. Mapping the face in the somatosensory brainstem. Nature Reviews Neuroscience 11(4):252–263.
Farmer, M. A., M. L. Chanda, E. L. Parks, M. N. Baliki, A. V. Apkarian, and A. J. Schaeffer. 2011. Brain functional and anatomical changes in chronic prostatitis/chronic pelvic pain syndrome. Journal of Urology 186(1):117–124.
FDA-NIH (Food and Drug Administration and National Institutes of Health) Biomarker Working Group. 2018. BEST (Biomarkers, EndpointS, and other Tools) resource. https://www.ncbi.nlm.nih.gov/books/NBK326791/pdf/Bookshelf_NBK326791.pdf (accessed January 9, 2020).
Ferber, G. 2002. Biomarkers and proof of concept. Methods and Findings in Experimental and Clinical Pharmacology 24(Suppl C):35–40.
Fillingim, R. B., R. Ohrbach, J. D. Greenspan, C. Knott, R. Dubner, E. Bair, C. Baraian, G. D. Slade, and W. Maixner. 2011. Potential psychosocial risk factors for chronic TMD: Descriptive data and empirically identified domains from the OPPERA case-control study. Journal of Pain 12(11 Suppl):T46–T60.
Gallo, L. M., N. Fankhauser, Y. M. Gonzalez, H. Liu, Y. Liu, J. C. Nickel, and L. R. Iwasaki. 2018. Jaw closing movement and sex differences in temporomandibular joint energy densities. Journal of Oral Rehabilitation 45(2):97–103.
Gao, Y.-J., and R.-R. Ji. 2009. c-Fos and pERK, which is a better marker for neuronal activation and central sensitization after noxious stimulation and tissue injury? The Open Pain Journal 2:11–17.
Gazerani, P., and H. S. H. Vinterhøj. 2016. “Omics”: An emerging field in pain research and management. Future Neurology 11(4):255–265.
Ge, C., F. Mohamed, A. Binrayes, S. Kapila, and R. T. Franceschi. 2018. Selective role of discoidin domain receptor 2 in murine temporomandibular joint development and aging. Journal of Dental Research 97(3):321–328.
Gerstner, G., E. Ichesco, A. Quintero, and T. Schmidt-Wilcke. 2011. Changes in regional gray and white matter volume in patients with myofascial-type temporomandibular disorders: A voxel-based morphometry study. Journal of Orofacial Pain 25(2):99–106.
Giesecke, T., R. H. Gracely, D. A. Williams, M. E. Geisser, F. W. Petzke, and D. J. Clauw. 2005. The relationship between depression, clinical pain, and experimental pain in a chronic pain cohort. Arthritis and Rheumatism 52(5):1577–1584.
Gomez-Varela, D., and M. Schmidt. 2018. The proteomics and metabolomics of pain—opportunities for systems medicine. In J. N. Wood, ed. Oxford handbook of the neurobiology of pain. Oxford, UK: Oxford University Press.
Goo, Y. A., K. Cain, M. Jarrett, L. Smith, J. Voss, E. Tolentino, J. Tsuji, Y. S. Tsai, A. Panchaud, D. R. Goodlett, R. J. Shulman, and M. Heitkemper. 2012. Urinary proteome analysis of irritable bowel syndrome (IBS) symptom subgroups. Journal of Proteome Research 11(12):5650–5662.
Greenspan, J. D., G. D. Slade, E. Bair, R. Dubner, R. B. Fillingim, R. Ohrbach, C. Knott, F. Mulkey, R. Rothwell, and W. Maixner. 2011. Pain sensitivity risk factors for chronic TMD: Descriptive data and empirically identified domains from the OPPERA case control study. Journal of Pain 12(11 Suppl):T61–T74.
Gu, S., W. Wu, C. Liu, L. Yang, C. Sun, W. Ye, X. Li, J. Chen, F. Long, and Y. Chen. 2014. BMPRIA mediated signaling is essential for temporomandibular joint development in mice. PLoS One 9(8).
Gunja, N. J., R. K. Uthamanthil, and K. A. Athanasiou. 2009. Effects of TGF-β1 and hydrostatic pressure on meniscus cell-seeded scaffolds. Biomaterials 30(4):565–573.
Guo, W., H. Wang, S. Zou, F. Wei, R. Dubner, and K. Ren. 2010. Long lasting pain hypersensitivity following ligation of the tendon of the masseter muscle in rats: A model of myogenic orofacial pain. Molecular Pain 6:40.
Hagerty, S. L., J. M. Ellingson, T. B. Helmuth, L. C. Bidwell, K. E. Hutchison, and A. D. Bryan. 2019. An overview and proposed research framework for studying co-occurring mental- and physical-health dysfunction. Perspectives on Psychological Science 14(4):633–645.
Harisankar, C. N., B. R. Mittal, A. Bhattacharya, P. Singh, and R. Sen. 2012. Utility of single photon emission computed tomography/computed tomography imaging in evaluation of chronic low back pain. Indian Journal of Nuclear Medicine 27(3):156–163.
Harper, D. E., A. Schrepf, and D. J. Clauw. 2016a. Pain mechanisms and centralized pain in temporomandibular disorders. Journal of Dental Research 95(10):1102–1108.
Harper, D. E., Y. Shah, E. Ichesco, G. E. Gerstner, and S. J. Peltier. 2016b. Multivariate classification of pain-evoked brain activity in temporomandibular disorder. Pain Reports 1(3).
Harte, S. E., A. Schrepf, R. Gallop, G. H. Kruger, H. H. H. Lai, S. Sutcliffe, M. Halvorson, E. Ichesco, B. D. Naliboff, N. Afari, R. E. Harris, J. T. Farrar, F. Tu, J. R. Landis, and D. J. Clauw. 2019. Quantitative assessment of nonpelvic pressure pain sensitivity in urologic chronic pelvic pain syndrome: A MAPP research network study. Pain 160(6):1270–1280.
Hasin, Y., M. Seldin, and A. Lusis. 2017. Multi-omics approaches to disease. Genome Biology 18(1):1–15.
Hawkins, J. L., and P. L. Durham. 2016. Prolonged jaw opening promotes nociception and enhanced cytokine expression. Journal of Oral and Facial Pain and Headache 30(1):34–41.
He, S. S., F. Li, F. Song, S. Wu, J. Y. Chen, N. He, S. J. Zou, X. Q. Huang, S. Lui, Q. Y. Gong, and S. Chen. 2014. Spontaneous neural activity alterations in temporomandibular disorders: A cross-sectional and longitudinal resting-state functional magnetic resonance imaging study. Neuroscience 278:1–10.
He, S., F. Li, T. Gu, H. Ma, X. Li, S. Zou, X. Huang, S. Lui, Q. Gong, and S. Chen. 2018. Reduced corticostriatal functional connectivity in temporomandibular disorders. Human Brain Mapping 39(6):2563–2572.
Heitkemper, M. M., K. C. Cain, R. J. Shulman, R. L. Burr, C. Ko, E. B. Hollister, N. Callen, J. Zia, C. J. Han, and M. E. Jarrett. 2018. Stool and urine trefoil factor 3 levels: Associations with symptoms, intestinal permeability, and microbial diversity in irritable bowel syndrome. Beneficial Microbes 9(3):345–355.
Helgeland, E., S. Shanbhag, T. O. Pedersen, K. Mustafa, and A. Rosen. 2018. Scaffold-based temporomandibular joint tissue regeneration in experimental animal models: A systematic review. Tissue Engineering—Part B: Reviews 24(4):300–316.
Herring, S. W., J. D. Decker, Z. J. Liu, and T. Ma. 2002. Temporomandibular joint in miniature pigs: Anatomy, cell replication, and relation to loading. The Anatomical Record 266(3):152–166.
Hinton, R. J. 2014. Genes that regulate morphogenesis and growth of the temporomandibular joint: A review. Developmental Dynamics 243(7):864–874.
Hinton, R. J., J. Jing, and J. Q. Feng. 2015. Genetic influences on temporomandibular joint development and growth. In Y. Chai, ed. Current topics in developmental biology. Vol. 115. Academic Press. Pp. 85–109.
Hockley, J. R. F., R. González-Cano, S. McMurray, M. A. Tejada-Giraldez, C. McGuire, A. Torres, A. L. Wilbrey, V. Cibert-Goton, F. R. Nieto, T. Pitcher, C. H. Knowles, J. M. Baeyens, J. N. Wood, W. J. Winchester, D. C. Bulmer, C. M. Cendán, and G. McMurray. 2017. Visceral and somatic pain modalities reveal NaV1.7-independent visceral nociceptive pathways. Journal of Physiology 595(8):2661–2679.
Hodges, P. W., and G. L. Moseley. 2003. Pain and motor control of the lumbopelvic region: Effect and possible mechanisms. Journal of Electromyography and Kinesiology 13(4):361–370.
Howard, M. A., D. Sanders, K. Krause, J. O’Muircheartaigh, A. Fotopoulou, F. Zelaya, M. Thacker, N. Massat, J. P. Huggins, W. Vennart, E. Choy, M. Daniels, and S. C. Williams. 2012. Alterations in resting-state regional cerebral blood flow demonstrate ongoing pain in osteoarthritis: An arterial spin-labeled magnetic resonance imaging study. Arthritis and Rheumatism 64(12):3936–3946.
Hunziker, R., N. Lumelsky, and F. Wang. 2015. Editorial: Scaffolds for regenerative medicine: A special issue of the Annals of Biomedical Engineering. Annals of Biomedical Engineering 43(3):487–488.
Ibi, M. 2019. Inflammation and temporomandibular joint derangement. Biological and Pharmaceutical Bulletin 42(4):538–542.
IOM (Institute of Medicine). 2011. Relieving pain in America: A blueprint for transforming prevention, care, education, and research. Washington, DC: The National Academies Press.
Isensee, J., L. Krahé, K. Moeller, V. Pereira, J. E. Sexton, X. Sun, E. Emery, J. N. Wood, and T. Hucho. 2017. Synergistic regulation of serotonin and opioid signaling contributes to pain insensitivity in Nav1.7 knockout mice. Science Signaling 10(461):eaah4874.
Ishizuka, Y., Y. Shibukawa, M. Nagayama, R. Decker, T. Kinumatsu, A. Saito, M. Pacifici, and E. Koyama. 2014. TMJ degeneration in SAMP8 mice is accompanied by deranged Ihh signaling. Journal of Dental Research 93(3):281–287.
Ito, G., Y. Suekawa, M. Watanabe, K. Takahashi, T. Inubushi, K. Murasaki, N. Hirose, S. Hiyama, T. Uchida, and K. Tanne. 2013. P2X7 receptor in the trigeminal sensory nuclear complex contributes to tactile allodynia/hyperalgesia following trigeminal nerve injury. European Journal of Pain 17(2):185–199.
Iwasaki, L. R., Y. M. Gonzalez, Y. Liu, H. Liu, M. Markova, L. M. Gallo, and J. C. Nickel. 2017. TMJ energy densities in healthy men and women. Osteoarthritis and Cartilage 25(6):846–849.
Jing, J., R. J. Hinton, Y. Mishina, Y. Liu, X. Zhou, and J. Q. Feng. 2014. Critical role of BMPr1a in mandibular condyle growth. Connective Tissue Research 55(01):73–78.
Kairys, A. E., T. Schmidt-Wilcke, T. Puiu, E. Ichesco, J. S. Labus, K. Martucci, M. A. Farmer, T. J. Ness, G. Deutsch, E. A. Mayer, S. Mackey, A. V. Apkarian, K. Maravilla, D. J. Clauw, and R. E. Harris. 2015. Increased brain gray matter in the primary somatosensory cortex is associated with increased pain and mood disturbance in interstitial cystitis/painful bladder syndrome patients. Journal of Urology 193(1):131–137.
Kalpakci, K. N., V. P. Willard, M. E. Wong, and K. A. Athanasiou. 2011. An interspecies comparison of the temporomandibular joint disc. Journal of Dental Research 90(2):193–198.
Kaneyama, K., N. Segami, M. Nishimura, T. Suzuki, and J. Sato. 2002. Importance of proinflammatory cytokines in synovial fluid from 121 joints with temporomandibular disorders. British Journal of Oral & Maxillofacial Surgery 40(5):418–423.
Kartha, S., T. Zhou, E. J. Granquist, and B. A. Winkelstein. 2016. Development of a rat model of mechanically induced tunable pain and associated temporomandibular joint responses. Journal of Oral & Maxillofacial Surgery 74(1):54.e1–54.e10.
Kato, K., P. F. Sullivan, B. Evengård, and N. L. Pedersen. 2009. A population-based twin study of functional somatic syndromes. Psychological Medicine 39(3):497–505.
Kavelaars, A., N. Eijkelkamp, H. L. D. M. Willemen, H. Wang, A. G. Carbajal, and C. J. Heijnen. 2011. Microglial GRK2: A novel regulator of transition from acute to chronic pain. Brain, Behavior, and Immunity 25(6):1055–1060.
Kobayashi, K., R. Jokaji, M. Miyazawa-Hira, S. Takatsuka, A. Tanaka, K. Ooi, H. Nakamura, and S. Kawashiri. 2017. Elastin-derived peptides are involved in the processes of human temporomandibular disorder by inducing inflammatory responses in synovial cells. Molecular Medicine Reports 16(3):3147–3154.
Komori, T. 2020. Molecular mechanism of Runx2-dependent bone development. Molecules and Cells 1–8.
Kothari, S. F., L. Baad-Hansen, Y. Oono, and P. Svensson. 2015. Somatosensory assessment and conditioned pain modulation in temporomandibular disorders pain patients. Pain 156(12):2545–2555.
Koyama, E., C. Saunders, I. Salhab, R. S. Decker, I. Chen, H. Um, M. Pacifici, and H. D. Nah. 2014. Lubricin is required for the structural integrity and post-natal maintenance of TMJ. Journal of Dental Research 93(7):663–670.
Krzyzanowska, A., and C. Avendaño. 2012. Behavioral testing in rodent models of orofacial neuropathic and inflammatory pain. Brain and Behavior 2(5):678–697.
Kurio, N., C. Saunders, T. E. Bechtold, I. Salhab, H. D. Nah, S. Sinha, P. C. Billings, M. Pacifici, and E. Koyama. 2018. Roles of Ihh signaling in chondroprogenitor function in postnatal condylar cartilage. Matrix Biology 67:15–31.
Kutch, J. J., E. Ichesco, J. P. Hampson, J. S. Labus, M. A. Farmer, K. T. Martucci, T. J. Ness, G. Deutsch, A. V. Apkarian, S. C. Mackey, D. J. Klumpp, A. J. Schaeffer, L. V. Rodriguez, K. J. Kreder, D. Buchwald, G. L. Andriole, H. H. Lai, C. Mullins, J. W. Kusek, J. R. Landis, E. A. Mayer, J. Q. Clemens, D. J. Clauw, R. E. Harris, and MAPP Research Network. 2017a. Brain signature and functional impact of centralized pain: A multidisciplinary approach to the study of chronic pelvic pain (MAPP) network study. Pain 158(10):1979–1991.
Kutch, J. J., J. S. Labus, R. E. Harris, K. T. Martucci, M. A. Farmer, S. Fenske, C. Fling, E. Ichesco, S. Peltier, B. Petre, W. Guo, X. Hou, A. J. Stephens, C. Mullins, D. J. Clauw, S. C. Mackey, A. V. Apkarian, J. R. Landis, E. A. Mayer, and Mapp Research Network. 2017b. Resting-state functional connectivity predicts longitudinal pain symptom change in urologic chronic pelvic pain syndrome: A MAPP network study. Pain 158(6):1069–1082.
Lam, D. K., B. J. Sessle, and J. W. Hu. 2009. Glutamate and capsaicin effects on trigeminal nociception II: Activation and central sensitization in brainstem neurons with deep craniofacial afferent input. Brain Research 1253:48–59.
Langford, D. J., A. L. Bailey, M. L. Chanda, S. E. Clarke, T. E. Drummond, S. Echols, S. Glick, J. Ingrao, T. Klassen-Ross, M. L. Lacroix-Fralish, L. Matsumiya, R. E. Sorge, S. G. Sotocinal, J. M. Tabaka, D. Wong, A. M. van den Maagdenberg, M. D. Ferrari, K. D. Craig, and J. S. Mogil. 2010. Coding of facial expressions of pain in the laboratory mouse. Nature Methods 7(6):447–449.
LeResche, L., L. Mancl, J. J. Sherman, B. Gandara, and S. F. Dworkin. 2003. Changes in temporomandibular pain and other symptoms across the menstrual cycle. Pain 106(3):253–261.
Leung, A., N. S. Gregory, L. A. Allen, and K. A. Sluka. 2016. Regular physical activity prevents chronic pain by altering resident muscle macrophage phenotype and increasing interleukin-10 in mice. Pain 157(1):70–79.
Levine, J. E. 2012. Chapter 1, an introduction to neuroendocrine systems. In G. Fink, D. W. Pfaff, and J. E. Levine, eds. Handbook of neuroendocrinology. San Diego, CA: Academic Press. Pp. 3–19.
Levy, D., L. Abdian, M. Dekel-Steinkeller, and R. Defrin. 2018. Experimental evidence for weaker endogenous inhibition of trigeminal pain than extra-trigeminal pain in healthy individuals. Cephalalgia 38(7):1307–1315.
Liao, L., S. Zhang, G. Q. Zhou, L. Ye, J. Huang, L. Zhao, and D. Chen. 2019. Deletion of Runx2 in condylar chondrocytes disrupts TMJ tissue homeostasis. Journal of Cellular Physiology 234(4):3436–3444.
Lin, C. S. 2014. Brain signature of chronic orofacial pain: A systematic review and meta-analysis on neuroimaging research of trigeminal neuropathic pain and temporomandibular joint disorders. PLoS One 9(4):e94300.
Liu, X., N. Li, H. Zhang, J. Liu, N. Zhou, C. Ran, X. Chen, Y. Lu, X. Wang, C. Qin, J. Xiao, and C. Liu. 2018. Inactivation of Fam20b in the neural crest-derived mesenchyme of mouse causes multiple craniofacial defects. European Journal of Oral Sciences 126(5):433–436.
Long, H. Q., P. F. Tian, Y. X. Guan, L. X. Liu, X. P. Wu, and B. Li. 2019. Expression of Ihh signaling pathway in condylar cartilage after bite-raising in adult rats. Journal of Molecular Histology 50(5):459–470.
Lopes, D. M., F. Denk, and S. B. McMahon. 2017. The molecular fingerprint of dorsal root and trigeminal ganglion neurons. Frontiers in Molecular Neuroscience 10:1–11.
Lopez-Sola, M., C. W. Woo, J. Pujol, J. Deus, B. J. Harrison, J. Monfort, and T. D. Wager. 2017. Towards a neurophysiological signature for fibromyalgia. Pain 158(1):34–47.
Lötsch, J., and A. Ultsch. 2018. Machine learning in pain research. Pain 159(4):623–630.
Luo, X., Y. Jiang, R. Bi, N. Jiang, and S. Zhu. 2018. Inhibition of notch signaling pathway temporally postpones the cartilage degradation progress of temporomandibular joint arthritis in mice. Journal of Cranio-Maxillofacial Surgery 46(7):1132–1138.
Ma, F., L. Zhang, D. Lyons, and K. N. Westlund. 2012. Orofacial neuropathic pain mouse model induced by trigeminal inflammatory compression (TIC) of the infraorbital nerve. Molecular Brain 5:44.
MacBarb, R. F., A. L. Chen, J. C. Hu, and K. A. Athanasiou. 2013. Engineering functional anisotropy in fibrocartilage neotissues. Biomaterials 34(38):9980–9989.
Mackey, S. 2014. National Pain Strategy Task Force: The strategic plan for the IOM pain report. Pain Medicine 15(7):1070–1071.
Mackey, S. 2016. Future directions for pain management: Lessons from the Institute of Medicine pain report and the National Pain Strategy. Hand Clinics 32(1):91–98.
Maes, M. 1999. Major depression and activation of the inflammatory response system. Advances in Experimental Medicine and Biology 461:25–46.
Maes, M., C. Song, A. Lin, R. De Jongh, A. Van Gastel, G. Kenis, E. Bosmans, I. De Meester, I. Benoy, H. Neels, P. Demedts, A. Janca, S. Scharpe, and R. S. Smith. 1998. The effects of psychological stress on humans: Increased production of pro-inflammatory cytokines and a Th1-like response in stress-induced anxiety. Cytokine 10(4):313–318.
Marquand, A., M. Howard, M. Brammer, C. Chu, S. Coen, and J. Mourao-Miranda. 2010. Quantitative prediction of subjective pain intensity from whole-brain fMRI data using Gaussian processes. Neuroimage 49(3):2178–2189.
Martínez-García, M.-Á., B. C. Migueláñez-Medrán, and C. Goicoechea. 2019. Animal models in the study and treatment of orofacial pain. Journal of Clinical and Experimental Dentistry 11(4):e382–e390.
Martucci, K. T., and S. C. Mackey. 2016. Imaging pain. Anesthesiology Clinics 34(2):255–269.
Martucci, K. T., and S. C. Mackey. 2018. Neuroimaging of pain: Human evidence and clinical relevance of central nervous system processes and modulation. Anesthesiology 128(6):1241–1254.
Martucci, K. T., K. A. Weber, 2nd, and S. C. Mackey. 2019. Altered cervical spinal cord resting-state activity in fibromyalgia. Arthritis & Rheumatology 71(3):441–450.
Matsumoto, K., K. Honda, M. Ohshima, Y. Yamaguchi, I. Nakajima, P. Micke, and K. Otsuka. 2006. Cytokine profile in synovial fluid from patients with internal derangement of the temporomandibular joint: A preliminary study. Dentomaxillofacial Radiology 35(6):432–441.
McLean, S. A. 2011. The potential contribution of stress systems to the transition to chronic whiplash-associated disorders. Spine 36(25 Suppl):S226–S232.
MDEpiNet TMJ Patient-Led RoundTable. 2018a. A national interagency temporomandibular disorder research plan: MDEpiNet TMJ Patient-Led RoundTable. http://mdepinet.org/wp-content/uploads/A-Priority-Listing-of-Research-Recommendations-Agency-Assignments_Final.pdf (accessed January 27, 2020).
MDEpiNet TMJ Patient-Led RoundTable. 2018b. The TMJ Patient-Led RoundTable: A history and summary of work. http://mdepinet.org/wp-content/uploads/TMJ-Patient-RoundTable-Briefing-Report_9_25_18.pdf (accessed January 27, 2020).
Meloto, C. B., P. O. Serrano, M. C. Ribeiro-DaSilva, and C. M. Rizzatti-Barbosa. 2011. Genomics and the new perspectives for temporomandibular disorders. Archives of Oral Biology 56(11):1181–1191.
Meloto, C. B., R. Benavides, R. N. Lichtenwalter, X. Wen, N. Tugarinov, K. Zorina-Lichtenwalter, A.-J. Chabot-Doré, M. H. Piltonen, S. Cattaneo, V. Verma, R. Klares, 3rd, S. Khoury, M. Parisien, and L. Diatchenko. 2018. Human Pain Genetics Database: A resource dedicated to human pain genetics research. Pain 159(4):749–763.
Melville, J. C., V. A. Manon, C. Blackburn, and S. Young. 2019. Current methods of maxillofacial tissue engineering. Oral Maxillofacial Surgery Clinics of North America 31(4):579–591.
Moana-Filho, E. J., and A. Herrero Babiloni. 2019. Endogenous pain modulation in chronic temporomandibular disorders: Derivation of pain modulation profiles and assessment of its relationship with clinical characteristics. Journal of Oral Rehabilitation 46(3):219–232.
Moana-Filho, E. J., A. Herrero Babiloni, and N. R. Theis-Mahon. 2018. Endogenous pain modulation in chronic orofacial pain: A systematic review and meta-analysis. Pain 159(8):1441–1455.
Moayedi, M., I. Weissman-Fogel, A. P. Crawley, M. B. Goldberg, B. V. Freeman, H. C. Tenenbaum, and K. D. Davis. 2011. Contribution of chronic pain and neuroticism to abnormal forebrain gray matter in patients with temporomandibular disorder. Neuroimage 55(1):277–286.
Moayedi, M., I. Weissman-Fogel, T. V. Salomons, A. P. Crawley, M. B. Goldberg, B. V. Freeman, H. C. Tenenbaum, and K. D. Davis. 2012. White matter brain and trigeminal nerve abnormalities in temporomandibular disorder. Pain 153(7):1467–1477.
Mogil, J. S. 2012. Pain genetics: Past, present and future. Trends in Genetics 28(6):258–266.
Moisset, X., and D. Bouhassira. 2007. Brain imaging of neuropathic pain. Neuroimage 37(Suppl 1):S80–S88.
Moloney, R. D., J. Sajjad, T. Foley, V. D. Felice, T. G. Dinan, J. F. Cryan, and S. M. O’Mahony. 2016. Estrous cycle influences excitatory amino acid transport and visceral pain sensitivity in the rat: Effects of early-life stress. Biology of Sex Differences 7:33.
Monaco, A., R. Cattaneo, L. Mesin, E. Ortu, M. Giannoni, and D. Pietropaoli. 2015. Dysregulation of the descending pain system in temporomandibular disorders revealed by low-frequency sensory transcutaneous electrical nerve stimulation: A pupillometric study. PLoS One 10(4):e0122826.
Mori-Akiyama, Y., H. Akiyama, D. H. Rowitch, and B. de Crombrugghe. 2003. Sox9 is required for determination of the chondrogenic cell lineage in the cranial neural crest. Proceedings of the National Academy of Sciences 100(16):9360–9365.
Mupparapu, M., S. Oak, Y. C. Chang, and A. Alavi. 2019. Conventional and functional imaging in the evaluation of temporomandibular joint rheumatoid arthritis: A systematic review. Quintessence International 50(9):742–753.
Mutso, A. A., B. Petre, L. Huang, M. N. Baliki, S. Torbey, K. M. Herrmann, T. J. Schnitzer, and A. V. Apkarian. 2014. Reorganization of hippocampal functional connectivity with transition to chronic back pain. Journal of Neurophysiology 111(5):1065–1076.
Nackley, A. G., K. S. Tan, K. Fecho, P. Flood, L. Diatchenko, and W. Maixner. 2007. CatecholO-methyltransferase inhibition increases pain sensitivity through activation of both beta2- and beta3-adrenergic receptors. Pain 128(3):199–208.
Nagakura, Y. 2017. The need for fundamental reforms in the pain research field to develop innovative drugs. Expert Opinion on Drug Discovery 12(1):39–46.
Nash, P., K. Wiley, J. Brown, R. Shinaman, D. Ludlow, A.-M. Sawyer, G. Glover, and S. Mackey. 2013. Functional magnetic resonance imaging identifies somatotopic organization of nociception in the human spinal cord. Pain 154(6):776–781.
Nassar, M. A., L. C. Stirling, G. Forlani, M. D. Baker, E. A. Matthews, A. H. Dickenson, and J. N. Wood. 2004. Nociceptor-specific gene deletion reveals a major role for Nav1.7 (PN1) in acute and inflammatory pain. Proceedings of the National Academy of Sciences 101(34):12706–12711.
Neubert, J. K., C. G. Widmer, W. Malphurs, H. L. Rossi, C. J. Vierck, Jr., and R. M. Caudle. 2005. Use of a novel thermal operant behavioral assay for characterization of orofacial pain sensitivity. Pain 116(3):386–395.
Nickel, J. C., D. A. Tripp, L. K. Carr, M. Pontari, R. Moldwin, R. Mayer, R. Doggweiler, C. C. Yang, N. Mishra, and J. Nordling. 2015. Clinical and psychological parameters associated with pain pattern phenotypes in women with interstitial cystitis/bladder pain syndrome. Journal of Urology 193(1):138–144.
Nickel, J. C., L. R. Iwasaki, Y. M. Gonzalez, L. M. Gallo, and H. Yao. 2018. Mechanobehavior and ontogenesis of the temporomandibular joint. Journal of Dental Research 97(11):1185–1192.
NIH (National Institutes of Health). 2019a. Estimates of funding for various research, condition, and disease categories (RCDC). https://report.nih.gov/categorical_spending.aspx (accessed January 14, 2020).
NIH. 2019b. Types of grant programs. https://grants.nih.gov/grants/funding/funding_program.htm (accessed January 31, 2020).
Ochiai, T., Y. Shibukawa, M. Nagayama, C. Mundy, T. Yasuda, T. Okabe, K. Shimono, M. Kanyama, H. Hasegawa, Y. Maeda, B. Lanske, M. Pacifici, and E. Koyama. 2010. Indian hedgehog roles in post-natal TMJ development and organization. Journal of Dental Research 89(4):349–354.
Ohrbach, R., and S. F. Dworkin. 1998. Five-year outcomes in TMD: Relationship of changes in pain to changes in physical and psychological variables. Pain 74(2-3):315–326.
Ojima, K., N. Watanabe, N. Narita, and M. Narita. 2007. Temporomandibular disorder is associated with a serotonin transporter gene polymorphism in the Japanese population. Biopsychosocial Medicine 1:3.
Oka, K., S. Oka, R. Hosokawa, P. Bringas, Jr., H. C. Brockhoff, 2nd, K. Nonaka, and Y. Chai. 2008. TGF-beta mediated Dlx5 signaling plays a crucial role in osteo-chondroprogenitor cell lineage determination during mandible development. Developmental Biology 321(2):303–309.
Olausson, P., B. Gerdle, N. Ghafouri, D. Sjöström, E. Blixt, and B. Ghafouri. 2015. Protein alterations in women with chronic widespread pain—An explorative proteomic study of the trapezius muscle. Scientific Reports 5(1):11894.
Ozdiler, O., K. Orhan, E. Cesur, A. Koklu, and O. Algin. 2019. Evaluation of temporomandibular joint, masticatory muscle, and brain cortex activity in patients treated by removable functional appliances: A prospective fMRI study. Dentomaxillofacial Radiology 48(7).
Patti, G. J., O. Yanes, L. P. Shriver, J.-P. Courade, R. Tautenhahn, M. Manchester, and G. Siuzdak. 2012. Metabolomics implicates altered sphingolipids in chronic pain of neuropathic origin. Nature Chemical Biology 8(3):232–234.
Payne, S. C., J. B. Furness, and M. J. Stebbing. 2019. Bioelectric neuromodulation for gastrointestinal disorders: Effectiveness and mechanisms. Nature Reviews Gastroenterology & Hepatology 16(2):89–105.
Powers, C. M. 2010. The influence of abnormal hip mechanics on knee injury: A biomechanical perspective. Journal of Orthopaedic & Sports Physical Therapy 40(2):42–51.
Purcell, P., A. Jheon, M. P. Vivero, H. Rahimi, A. Joo, and O. D. Klein. 2012. Spry1 and Spry2 are essential for development of the temporomandibular joint. Journal of Dental Research 91(4):387–393.
Puri, J., P. Vinothini, J. Reuben, L. L. Bellinger, L. Ailing, Y. B. Peng, and P. R. Kramer. 2012. Reduced GABAA receptor α6 expression in the trigeminal ganglion alters inflammatory TMJ hypersensitivity. Neuroscience 213:179–190.
Reiter, S., A. Emodi-Perlman, C. Goldsmith, P. Friedman-Rubin, and E. Winocur. 2015. Co-morbidity between depression and anxiety in patients with temporomandibular disorders according to the research diagnostic criteria for temporomandibular disorders. Journal of Oral & Facial Pain and Headache 29(2):135–143.
Ribeiro-Dasilva, M. C., S. R. Peres Line, M. C. Leme Godoy dos Santos, M. T. Arthuri, W. Hou, R. B. Fillingim, and C. M. Rizzatti Barbosa. 2009. Estrogen receptor-alpha polymorphisms and predisposition to TMJ disorder. Journal of Pain 10(5):527–533.
Ribeiro-Dasilva, M. C., R. B. Fillingim, and S. M. Wallet. 2017. Estrogen-induced monocytic response correlates with temporomandibular disorder pain. Journal of Dental Research 96(3):285–291.
Roberts, W. E., and D. L. Stocum. 2018. Part II: Temporomandibular joint (TMJ)-regeneration, degeneration, and adaptation. Current Osteoporosis Reports 16(4):369–379.
Rogers, A. W., S. E. Cisewski, and C. B. Kern. 2018. The zonal architecture of the mandibular condyle requires ADAMTS5. Journal of Dental Research 97(12):1383–1390.
Romero-Reyes, M., S. Akerman, E. Nguyen, A. Vijjeswarapu, B. Hom, H.-W. Dong, and A. C. Charles. 2013. Spontaneous behavioral responses in the orofacial region: A model of trigeminal pain in mouse. Headache 53(1):137–151.
Romero-Reyes, M., V. Pardi, and S. Akerman. 2015. A potent and selective calcitonin gene-related peptide (CGRP) receptor antagonist, MK-8825, inhibits responses to nociceptive trigeminal activation: Role of CGRP in orofacial pain. Experimental Neurology 271:95–103.
Ronald, S., and D. K. Mills. 2016. Fibrochondrocyte growth and functionality on TiO2 nanothin films. Journal of Functional Biomaterials 7(2):14.
Russo, M., P. Georgius, and D. M. Santarelli. 2018. A new hypothesis for the pathophysiology of complex regional pain syndrome. Medical Hypotheses 119:41–53.
Sannajust, S., I. Imbert, V. Eaton, T. Henderson, L. Liaw, M. May, M. F. Barbe, and T. King. 2019. Females have greater susceptibility to develop ongoing pain and central sensitization in a rat model of temporomandibular joint pain. Pain 160(9):2036–2049.
Scariot, R., P. Corso, A. M. Sebastiani, and A. R. Vieira. 2018. The many faces of genetic contributions to temporomandibular joint disorder: An updated review. Orthodontics & Craniofacial Research 21(4):186–201.
Schrepf, A., D. E. Harper, S. E. Harte, H. Wang, E. Ichesco, J. P. Hampson, J. K. Zubieta, D. J. Clauw, and R. E. Harris. 2016. Endogenous opioidergic dysregulation of pain in fibromyalgia: A PET and fMRI study. Pain 157(10):2217–2225.
Schwenkreis, P., C. Maier, and M. Tegenthoff. 2009. Functional imaging of central nervous system involvement in complex regional pain syndrome. American Journal of Neuroradiology 30(7):1279–1284.
Sessle, B. J. 2011. Peripheral and central mechanisms of orofacial inflammatory pain. International Review of Neurobiology 97:179–206.
Shen, P., Z. Jiao, J. S. Zheng, W. F. Xu, S. Y. Zhang, A. Qin, and C. Yang. 2015. Injecting vascular endothelial growth factor into the temporomandibular joint induces osteoarthritis in mice. Scientific Reports 5(1):16244.
Sherman, J. J., L. LeResche, K. H. Huggins, L. A. Mancl, J. C. Sage, and S. F. Dworkin. 2004. The relationship of somatization and depression to experimental pain response in women with temporomandibular disorders. Psychosomatic Medicine 66(6):852–860.
Shibata, S., N. Suda, S. Yoda, H. Fukuoka, K. Ohyama, Y. Yamashita, and T. Komori. 2004. Runx2-deficient mice lack mandibular condylar cartilage and have deformed Meckel’s cartilage. Anatomy and Embryology 208(4):273–280.
Shibukawa, Y., T. Ishikawa, Y. Kato, Z. K. Zhang, T. Jiang, M. Shintani, M. Shimono, T. Kumai, T. Suzuki, M. Kato, and Y. Nakamura. 2007. Cerebral cortical dysfunction in patients with temporomandibular disorders in association with jaw movement observation. Pain 128(1–2):180–188.
Shibuta, K., I. Suzuki, M. Shinoda, Y. Tsuboi, K. Honda, N. Shimizu, B. J. Sessle, and K. Iwata. 2012. Organization of hyperactive microglial cells in trigeminal spinal subnucleus caudalis and upper cervical spinal cord associated with orofacial neuropathic pain. Brain Research 1451:74–86.
Skouen, J. S., A. J. Smith, N. M. Warrington, P. B. O’ Sullivan, L. McKenzie, C. E. Pennell, and L. M. Straker. 2012. Genetic variation in the beta-2 adrenergic receptor is associated with chronic musculoskeletal complaints in adolescents. European Journal of Pain 16(9):1232–1242.
Slade, G. D., M. S. Conrad, L. Diatchenko, N. U. Rashid, S. Zhong, S. Smith, J. Rhodes, A. Medvedev, S. Makarov, W. Maixner, and A. G. Nackley. 2011. Cytokine biomarkers and chronic pain: Association of genes, transcription, and circulating proteins with temporomandibular disorders and widespread palpation tenderness. Pain 152(12):2802–2812.
Sluka, K. A., and D. J. Clauw. 2016. Neurobiology of fibromyalgia and chronic widespread pain. Neuroscience 338:114–129.
Sluka, K. A., L. Frey-Law, and M. Hoeger Bement. 2018. Exercise-induced pain and analgesia? Underlying mechanisms and clinical translation. Pain 159(Suppl 1):S91–S97.
Smith, S. B., D. W. Maixner, J. D. Greenspan, R. Dubner, R. B. Fillingim, R. Ohrbach, C. Knott, G. D. Slade, E. Bair, D. G. Gibson, D. V. Zaykin, B. S. Weir, W. Maixner, and L. Diatchenko. 2011. Potential genetic risk factors for chronic TMD: Genetic associations from the OPPERA case control study. Journal of Pain 12(11 Suppl):T92–T101.
Smith, S. B., E. Mir, E. Bair, G. D. Slade, R. Dubner, R. B. Fillingim, J. D. Greenspan, R. Ohrbach, C. Knott, B. Weir, W. Maixner, and L. Diatchenko. 2013. Genetic variants associated with development of TMD and its intermediate phenotypes: The genetic architecture of TMD in the OPPERA prospective cohort study. Journal of Pain 14(12 Suppl):T91–T101.
Smith, S. M., and W. W. Vale. 2006. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues in Clinical Neuroscience 8(4):383–395.
Somoza, R. A., J. F. Welter, D. Correa, and A. I. Caplan. 2014. Chondrogenic differentiation of mesenchymal stem cells: Challenges and unfulfilled expectations. Tissue Engineering—Part B, Reviews 20(6):596–608.
Sperry, M. M., M. E. Ita, S. Kartha, S. Zhang, Y. H. Yu, and B. Winkelstein. 2017. The interface of mechanics and nociception in joint pathophysiology: Insights from the facet and temporomandibular joints. Journal of Biomechanical Engineering 139(2).
Staniszewski, K., H. Lygre, E. Bifulco, S. Kvinnsland, L. Willassen, E. Helgeland, T. Berge, and A. Rosén. 2018. Temporomandibular disorders related to stress and HPA-axis regulation. Pain Research & Management 2018:1–7.
Staud, R. 2011a. Brain imaging in fibromyalgia syndrome. Clinical and Experimental Rheumatology 29(6 Suppl 69):S109–S117.
Staud, R. 2011b. Evidence for shared pain mechanisms in osteoarthritis, low back pain, and fibromyalgia. Current Rheumatology Reports 13(6):513–520.
Stocum, D. L., and W. E. Roberts. 2018. Part I: Development and physiology of the temporomandibular joint. Current Osteoporosis Reports 16(4):360–368.
Tamura, R., T. Nemoto, T. Maruta, S. Onizuka, T. Yanagita, A. Wada, M. Murakami, and I. Tsuneyoshi. 2014. Up-regulation of Nav1.7 sodium channels expression by tumor necrosis factor-α in cultured bovine adrenal chromaffin cells and rat dorsal root ganglion neurons. Anesthesia and Analgesia 118(2):318–324.
Tanaka, E., J. Aoyama, M. Miyauchi, T. Takata, K. Hanaoka, T. Iwabe, and K. Tanne. 2005. Vascular endothelial growth factor plays an important autocrine/paracrine role in the progression of osteoarthritis. Histochemistry and Cell Biology 123(3):275–281.
Tegeder, I., M. Costigan, R. S. Griffin, A. Abele, I. Belfer, H. Schmidt, C. Ehnert, J. Nejim, C. Marian, J. Scholz, T. Wu, A. Allchorne, L. Diatchenko, A. M. Binshtok, D. Goldman, J. Adolph, S. Sama, S. J. Atlas, W. A. Carlezon, A. Parsegian, J. Lotsch, R. B. Fillingim, W. Maixner, G. Geisslinger, M. B. Max, and C. J. Woolf. 2006. GTP cyclohydrolase and tetrahydrobiopterin regulate pain sensitivity and persistence. Nature Medicine 12(11):1269–1277.
Traub, R. J., D.-Y. Cao, J. Karpowicz, S. Pandya, Y. Ji, S. G. Dorsey, and D. Dessem. 2014. A clinically relevant animal model of temporomandibular disorder and irritable bowel syndrome comorbidity. Journal of Pain 15(9):956–966.
Ung, H., J. E. Brown, K. A. Johnson, J. Younger, J. Hush, and S. Mackey. 2012. Multivariate classification of structural MRI data detects chronic low back pain. Cerebral Cortex 24(4):1037–1044.
Vapniarsky, N., A. Aryaei, B. Arzi, D. C. Hatcher, J. C. Hu, and K. A. Athanasiou. 2017. The Yucatan minipig temporomandibular joint disc structure-function relationships support its suitability for human comparative studies. Tissue Engineering—Part C: Methods 23(11):700–709.
Vapniarsky, N., L. W. Huwe, B. Arzi, M. K. Houghton, M. E. Wong, J. W. Wilson, D. C. Hatcher, J. C. Hu, and K. A. Athanasiou. 2018. Tissue engineering toward temporomandibular joint disc regeneration. Science Translational Medicine 10(446):1–10.
Vargas-Alarcón, G., J.-M. Fragoso, D. Cruz-Robles, A. Vargas, A. Vargas, J.-I. Lao-Villadóniga, F. García-Fructuoso, M. Ramos-Kuri, F. Hernández, R. Springall, R. Bojalil, M. Vallejo, and M. Martínez-Lavín. 2007. Catechol-o-methyltransferase gene haplotypes in Mexican and Spanish patients with fibromyalgia. Arthritis Research & Therapy 9(5):1–7.
Vargas-Alarcón, G., J.-M. Fragoso, D. Cruz-Robles, A. Vargas, A. Martinez, J.-I. Lao-Villadóniga, F. García-Fructuoso, M. Vallejo, and M. Martínez-Lavín. 2009. Association of adrenergic receptor gene polymorphisms with different fibromyalgia syndrome domains. Arthritis and Rheumatism 60(7):2169–2173.
Vazquez, K. J., J. T. Andreae, and C. R. Henak. 2019. Cartilage-on-cartilage cyclic loading induces mechanical and structural damage. Journal of the Mechanical Behavior of Biomedical Materials 98:262–267.
Veiga-Fernandes, H., and D. Artis. 2018. Neuronal–immune system cross-talk in homeostasis. Science 359(6383):1465.
Von Korff, M., A. I. Scher, C. Helmick, O. Carter-Pokras, D. W. Dodick, J. Goulet, R. Hamill-Ruth, L. LeResche, L. Porter, R. Tait, G. Terman, C. Veasley, and S. Mackey. 2016. United States National Pain Strategy for population research: Concepts, definitions, and pilot data. Journal of Pain 17(10):1068–1080.
Wadhwa, S., and S. Kapila. 2008. TMJ disorders: Future innovations in diagnostics and therapeutics. Journal of Dental Education 72(8):930–947.
Wager, T. D., L. Y. Atlas, M. A. Lindquist, M. Roy, C. W. Woo, and E. Kross. 2013. An fMRI-based neurologic signature of physical pain. New England Journal of Medicine 368(15):1388–1397.
Wang, M., S. Li, W. Xie, J. Shen, H. J. Im, J. D. Holz, M. Wang, T. G. H. Diekwisch, and D. Chen. 2014. Activation of β-catenin signalling leads to temporomandibular joint defects. European Cells and Materials 28:223–235.
Wang, X. D., J. N. Zhang, Y. H. Gan, and Y. H. Zhou. 2015. Current understanding of pathogenesis and treatment of TMJ osteoarthritis. Journal of Dental Research 94(5):666–673.
Wang, Y., C. Liu, J. Rohr, H. Liu, F. He, J. Yu, C. Sun, L. Li, S. Gu, and Y. Chen. 2011. Tissue interaction is required for glenoid fossa development during temporomandibular joint formation. Developmental Dynamics 240(11):2466–2473.
Weber, K. A., 2nd, A. I. Sentis, O. N. Bernadel-Huey, Y. Chen, X. Wang, T. B. Parrish, and S. Mackey. 2018. Thermal stimulation alters cervical spinal cord functional connectivity in humans. Neuroscience 369:40–50.
Wilcox, S. L., S. M. Gustin, P. M. Macey, C. C. Peck, G. M. Murray, and L. A. Henderson. 2015. Anatomical changes within the medullary dorsal horn in chronic temporomandibular disorder pain. Neuroimage 117:258–266.
Willard, V. P., L. Zhang, and K. A. Athanasiou. 2011. Tissue engineering of the temporomandibular joint. In Comprehensive biomaterials. Vol. 5. Pp. 221–235.
Williams, D. A., and D. J. Clauw. 2009. Understanding fibromyalgia: Lessons from the broader pain research community. Journal of Pain 10(8):777–791.
Woo, C. W., M. Roy, J. T. Buhle, and T. D. Wager. 2015. Distinct brain systems mediate the effects of nociceptive input and self-regulation on pain. PLoS Biology 13(1):e1002036.
Woo, C. W., L. Schmidt, A. Krishnan, M. Jepma, M. Roy, M. A. Lindquist, L. Y. Atlas, and T. D. Wager. 2017. Quantifying cerebral contributions to pain beyond nociception. Nature Communications 8:14211.
Woolf, C. J. 2011. Central sensitization: Implications for the diagnosis and treatment of pain. Pain 152(3 Suppl):S2–S15.
Wu, Y., S. E. Cisewski, M. C. Coombs, M. H. Brown, F. Wei, X. She, M. J. Kern, Y. M. Gonzalez, L. M. Gallo, V. Colombo, L. R. Iwasaki, J. C. Nickel, and H. Yao. 2019. Effect of sustained joint loading on TMJ disc nutrient environment. Journal of Dental Research 98(8):888–895.
Xiao, D., R. Wang, J. Hu, and H. Quan. 2017. Spatial and temporal expression of Smad signaling members during the development of mandibular condylar cartilage. Experimental and Therapeutic Medicine 14(5):4967–4971.
Yang, H., M. Zhang, Q. Liu, H. Zhang, J. Zhang, L. Lu, M. Xie, D. Chen, and M. Wang. 2019. Inhibition of Ihh reverses temporomandibular joint osteoarthritis via a PTH1R signaling dependent mechanism. International Journal of Molecular Sciences 20(15):3797.
Yasuda, T., H. D. Nah, J. Laurita, T. Kinumatsu, Y. Shibukawa, T. Shibutani, N. Minugh-Purvis, M. Pacifici, and E. Koyama. 2012. Muenke syndrome mutation, FgfR3P244R causes TMJ defects. Journal of Dental Research 91(7):683–689.
Yerliyurt, K., A. F. Nursal, A. Tekcan, N. Karakus, M. K. Tumer, and S. Yigit. 2019. Effect of a functional variant of tumor necrosis factor-β gene in temporomandibular disorders: A pilot study. Journal of Clinical Laboratory Analysis 33(1):e22641.
Younger, J. W., Y. F. Shen, G. Goddard, and S. C. Mackey. 2010. Chronic myofascial temporomandibular pain is associated with neural abnormalities in the trigeminal and limbic systems. Pain 149(2):222–228.
Yu, J., F. Liang, H. Huang, P. Pirttiniemi, and D. Yu. 2018. Effects of loading on chondrocyte hypoxia, HIF-1α and VEGF in the mandibular condylar cartilage of young rats. Orthodontics & Craniofacial Research 21(1):41–47.
Zatorre, R. J., R. D. Fields, and H. Johansen-Berg. 2012. Plasticity in gray and white: Neuroimaging changes in brain structure during learning. Nature Neuroscience 15(4):528–536.
Zheng, L., C. Pi, J. Zhang, Y. Fan, C. Cui, Y. Zhou, J. Sun, Q. Yuan, X. Xu, L. Ye, X. Cao, and X. Zhou. 2018. Aberrant activation of latent transforming growth factor-β initiates the onset of temporomandibular joint osteoarthritis. Bone Research 6:26.
Zhong, J., D. Q. Chen, P. S. Hung, D. J. Hayes, K. E. Liang, K. D. Davis, and M. Hodaie. 2018. Multivariate pattern classification of brain white matter connectivity predicts classic trigeminal neuralgia. Pain 159(10):2076–2087.
Zhou, Y., B. Shu, R. Xie, J. Huang, L. Zheng, X. Zhou, G. Xiao, L. Zhao, and D. Chen. 2019. Deletion of Axin1 in condylar chondrocytes leads to osteoarthritis-like phenotype in temporomandibular joint via activation of β-catenin and FGF signaling. Journal of Cellular Physiology 234(2):1720–1729.
Zorina-Lichtenwalter, K., C. B. Meloto, S. Khoury, and L. Diatchenko. 2016. Genetic predictors of human chronic pain conditions. Neuroscience 338:36–62.










































































